Open Access Article
Open Access ArticleFramework-stabilized metal nanostructures for next-generation photocatalysis
Bikash Chandra
Dhal†
a,
Debadutta
Samal†
a,
Debashis
Acharya
a,
Subrat
Swain
a,
Basudeb
Dutta
 a,
Srikanta
Palei
b,
A. K.
Tyagi
a,
Srikanta
Palei
b,
A. K.
Tyagi
 c,
Puspanjali
Sahu
*d and
Rojalin
Sahu
c,
Puspanjali
Sahu
*d and
Rojalin
Sahu
 *a
*a
aFuture Materials Laboratory, Department of Chemistry, School of Applied Sciences, Kalinga Institute of Industrial Technology (KIIT) Deemed to be University, Bhubaneswar, Odisha 751024, India. E-mail: rsahufch@kiit.ac.in
bCentral Institute of Petrochemicals Engineering and Technology (CIPET): Institute of Petrochemicals Technology (IPT), B-25, CNI Complex, Patia, Bhubaneswar-751024, Odisha, India
cHomi Bhabha National Institute, Anushaktinagar, Mumbai, 400094, India
dBhadrak (Autonomous) College, Bhadrak-756100, Odisha, India. E-mail: psahu.chem@gmail.com
First published on 17th February 2026
Abstract
Framework-stabilized metal nanostructures have emerged as a powerful class of catalysts, offering excellent activity, selectivity, and long-term stability. Encapsulation of nanoparticles (NPs) within porous crystalline frameworks such as metal–organic frameworks (MOFs) and covalent organic frameworks (COFs) effectively prevents aggregation, ensures accessibility of active sites, and enables efficient charge separation. This review provides a comprehensive analysis of synthesis protocols: in situ, post-synthetic, and one-pot methods, keeping in mind the crucial factors that dictate structural architectures. The role of reducing agents, solvents, functional groups, temperature, and framework nanoparticle interactions acts as the pivotal role in the structure–activity relationship. The stimulating role of such a catalyst system can be employed in the field of clean energy and sustainable development: hydrogen evolution reaction (HER), CO2 reduction (CO2RR), selective oxidation, and organic transformations are critically discussed to establish active materials in such applications. In particular, the challenges related to uniform encapsulation, framework stabilisation under reaction conditions, scalability, and recyclability are significantly accessed, together with future prospects in defect engineering, hybrid bimetallic systems, and enhanced light-harvesting abilities. Therefore, this review aims to provide an overview of catalysis, energy conversion, and materials science with a consolidated perspective on the fabrication of next-generation hierarchical frameworks.
1. Introduction
Nanostructures received significant attention from researchers owing to their catalytic efficiency.1–5 However, owing to their high surface area, these structures exhibit thermodynamic instability and are prone to aggregation. The high surface energy of these nanostructures can be reduced by covering their surface with capping agents.6,7 However, such surface coverings restrict surface accessibility and consequently reduce the catalytic efficiency of the nanostructures. Aggregation of nanostructures can also be prevented by confining them within supports such as zeolites and silica or making composites with graphene, MXene, etc.8–10 However, this approach suffers from limitations such as uncontrolled growth of nanostructures and poor stability under harsh catalytic conditions.Recently developed frameworks offer an efficient alternative to encapsulate nanostructures. These frameworks are an exclusive class of crystalline materials constructed through strong chemical bonds between organic molecules or organic molecules with metal ions.2,11–18 Due to their captivating attributes, comprising varied chemical compositions, flexibility, large surface areas, and precisely defined pore architectures, frameworks have showcased significant potential as catalysts. Frameworks are predominantly used for various applications like storage and separation, carbon dioxide capture, drug delivery, sensing, and catalysis, marking them as a promising avenue for multilayered applications.19–22 In particular, catalysts constructed by integrating nanoparticles within frameworks have garnered substantial interest within the catalysis community.23–28 Encapsulation of NPs within frameworks blends the versatility of specially created organic matrices with the complexity of nanoscale engineering. This cooperative approach has paved the way for the development of catalysts that exhibit exceptional activity, stability and selectivity. The frameworks provide remarkable control over NP size, shape, and stability.
The overall theme of the metal nanoparticle-confined framework material is depicted in Scheme 1.
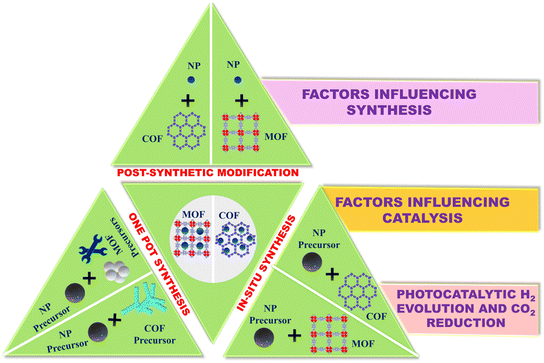
Further, these frameworks possess distinctive attributes that make them exceptionally well suited as hosts and substrates for NPs, resulting in a synergistic amplification of catalytic activity. Moreover, the existence of libraries of frameworks and NPs can provide a wide variety of catalysts suitable for a wide range of applications.29,30 The past decade has seen notable advancements in the development of NP-encapsulated frameworks.24,31–35 This review offers a comprehensive analysis of the variables influencing the development of these catalysts (NP-encapsulated frameworks). It also sheds light on the synergic influences of frameworks and NPs on the catalytic processes. Additionally, the current challenges associated with these encapsulated structures are discussed, as illustrated in Fig. 1.
 | ||
| Fig. 1 Factors influencing the synthesis, catalytic efficiencies, and challenges associated with nanostructure-encapsulated frameworks. | ||
Judicious analysis of Table 1 revealed a significant gap in correlating efficient catalytic activity with the role of framework-stabilized confinement effects. The synthesis and significant stability of metal clusters confined in frameworks remain challenging for broader aspects. Therefore, we aimed to establish strategies for nanostructure stabilization with reticular materials and to elucidate their role in photocatalysis.
| Sl no. | Material feature | Highlights | Ref. |
|---|---|---|---|
| 1 | MOF-on-MOF heterostructures | Explores interfacial engineering strategies to enhance the catalytic performance of MOF-on-MOF heterostructures | 36 |
| 2 | COF/MOF hybrids | Reviews progress in hybridizing COFs with MOFs to combine structural and functional advantages | 37 |
| 3 | MOF-derived nanocarbons and oxides | Surveys strategies for transforming MOFs into nanocarbons and metal oxide derivatives for enhanced photocatalysis | 38 |
| 4 | MOF–COF composites | Reviews design, synthesis, and mechanisms of MOF–COF composites for photocatalytic applications | 39 |
| 5 | MOFs and COFs | Provides a comprehensive overview of the status, challenges, and future prospects of MOF and COF applications | 40 |
| 6 | COFs | Discusses alternative synthetic methods for COFs beyond conventional solvothermal approaches | 41 |
| 7 | COFs for biomedical therapy | Explores COFs as rationally designed materials for targeted cancer therapy | 42 |
| 8 | MOFs (ML-guided discovery/computational screening) | Demonstrates machine learning for screening MOFs, rapidly identifying candidates for photocatalytic oxygen evolution reaction (OER) | 43 |
| 9 | Photocatalytic MOFs | Reviews MOFs as photocatalysts for water splitting and CO2 reduction | 44 |
| 10 | MOF heterojunctions | Highlights design strategies for MOF-based heterojunctions to improve charge separation and photocatalytic efficiency | 45 |
| 11 | COF photocatalysts | Summarises the design, linkers, morphologies, and applications of COF-based photocatalysts in energy and environment remediation | 46 |
| 12 | MOF-derived photocatalysts | Discusses heteroatom-doped MOF-derived materials as efficient photocatalysts for CO2 reduction | 47 |
| 13 | MOF photocatalysts for CO2 reduction | Reviews MOF-based catalysts for CO2 reduction, focusing on mechanisms, product selectivity (CO, CH4, C1–C2+), and design strategies | 48 |
| 14 | Zirconium-based MOFs | Highlights defect engineering and active-site optimization in Zr-MOFs for efficient photocatalytic CO2 reduction | 49 |
| 15 | MOFs, COFs, and heterojunctions | Reviews progress on MOF/COF-based photocatalysts for H2O2 production | 50 |
| 16 | COF photocatalysts | Examines COFs as tunable photocatalysts for H2O2 production, focusing on linker design, heterojunction, and stability | 51 |
| 17 | Porphyrin-based MOFs and COFs | Reviews visible-light-driven solar fuel production using nanosized porphyrin-based frameworks | 52 |
| 18 | MOF photocatalysts | Discusses design principles and applications of MOFs for efficient solar-driven photocatalytic water splitting | 53 |
| 19 | Porous organic frameworks | Demonstrates MOFs, COFs, and HOFs as multifunctional separators in rechargeable lithium metal batteries | 54 |
| 20 | Conductive MOFs for Zn batteries | Reviews conductive MOFs as promising electrodes for high-performance zinc-based batteries | 55 |
| 21 | MOF-based composites | Discusses advances in MOF composites, linking dimensionality and functionality to supercapacitor applications | 56 |
| 22 | COFs for biomedical implants | Reports COFs enabling NO release to enhance the multi-modal therapeutic performance of PEEK implants | 57 |
| 23 | Aptamer-modified MOFs | Reviews aptamer@MOF systems for efficient bacterial pathogen detection | 58 |
| 24 | MOFs as oxidase mimics | Discusses design and modification of MOFs to improve oxidase-mimic sensing applications | 59 |
| 25 | Uric acid-sensing MOFs | Reviews MOFs for uric acid detection and explores future integration with machine learning | 60 |
| 26 | Porphyrin MOFs and COFs | Reviews their use of heterogeneous photosensitizers for singlet oxygen-based antimicrobial nanodevices | 61 |
| 27 | Multiscale COF films | Develops multiscale COF films for selective sensing of multicomponent gases | 62 |
| 28 | Two-dimensional COFs | Reviews the design and application of 2D COFs for membrane separation | 63 |
| 29 | MOF/COF heterostructured hybrids | Reviews design, synthesis, and applications of MOF–COF heterostructures for advanced functions | 64 |
| 30 | MOF composites for photocatalysis | Reviews classifications and synergistic design of MOF composites, including metal/MOF systems | 65 |
| 31 | Noble-metal/MOF photocatalysts | Summarises synergistic effects of noble metal nanoparticles in MOF photocatalysis | 66 |
| 32 | Encapsulated metal NPs in porous hosts | Summarises advances in the synthesis and catalysis of encapsulated metal NPs in porous supports, emphasizing stability, selectivity, and metal–support interactions | 67 |
| 33 | MOF-based photocatalysts for CO2 reduction | Summarises recent developments in MOF-based photocatalysts including NP-loaded systems | 68 |
| 34 | Metal nanocluster-confined reticular materials | This review critically analyzes various factors influencing synthesis strategies, stabilization of metallic nanostructures and discusses the synergistic effects of nanoparticles and organic frameworks on photocatalytic applications. To the best of our knowledge, this is the only review that critically analyses and correlates factors influencing synthesis and activity | This review |
2. Encapsulation techniques for the nanostructures within frameworks
The well-designed structures of frameworks enable the effective encapsulation of a wide range of nanostructures. Methodologies developed so far can be divided into three broad categories. First, in the in situ synthesis method, metal ions are anchored on the skeleton of frameworks, and their successive reduction leads to the formation of nanostructures inside the pores of frameworks. Second, in a co-synthesis strategy, precursors of frameworks and nanoparticles can be mixed and used as starting materials to generate framework-encapsulated nanostructures. Finally, nanostructures can also be introduced into the pore of frameworks through post-synthetic modification. Together, these three methodologies provide complementary approaches to achieve encapsulation, as illustrated schematically in Fig. 2.2.1. In situ synthesis approach
It is possible to pre-embed metal ions into the pores of frameworks by connecting with framework constituent units or by using certain functional groups. Afterwards, certain conventional reducing agents can reduce those metal ions to nanoscale particles. With these in-situ synthesis techniques, the native crystal structure and porosity of the frameworks can be preserved.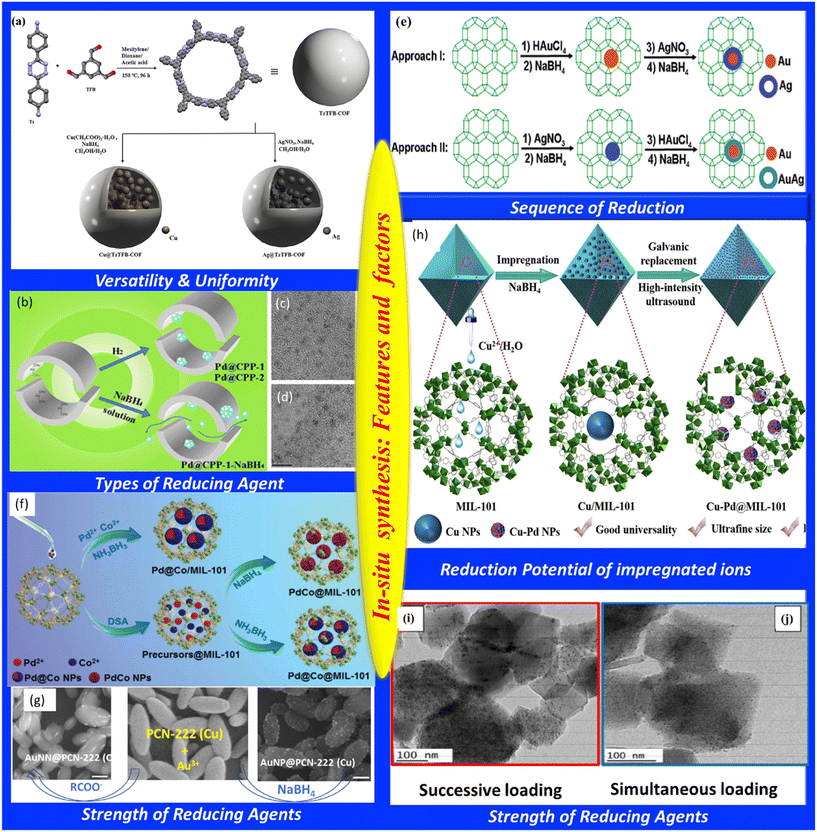 | ||
| Fig. 3 (a) Diagrammatic illustration of TzTFB-COF formation and functionalization with Ag and Cu NPs. Adapted from ref. 69, with permission from the American Chemical Society, copyright 2023. (b) Diagrammatic illustration of the synthesis of Pd@CPP with different reducing agents. (c and d) TEM images of Pd@CPP obtained by reduction with H2 and NaBH4 respectively. Adapted from ref. 72, with permission from the American Chemical Society, copyright 2014. (e) Schematic of synthesis procedure of Au@Ag@ZIF-8 and Au@AuAg@ZIF-8. Adapted from ref. 78, with permission from the American Chemical Society, copyright 2011. (f) Synthesis of Pd@Co@MIL-101, Pd@Co/MIL-101, and PdCo@MIL-10 catalysts by different procedures and reducing agents. Adapted from ref. 79, with permission from Wiley, copyright 2014. (g) TEM images of PCN-222 (Cu), AuNN@PCN-222 (Cu) and AuNP@PCN-222 (Cu). Adapted from ref. 80, with permission from Nature, copyright 2022. (h) Schematic of synthesis procedure of Cu–Pd NPs@MIL-101.82 Adapted from ref. 82, with permission from the American Chemical Society, copyright 2019. (i and j) TEM images of Pd2.5Ni2.5@MIL-101. Slightly bigger metal NPs are obtained by successive loading of Pd and Ni. Simultaneous loading results in smaller NPs within MIL-101. Adapted from ref. 81, with permission from Wiley, copyright 2012. | ||
The use of an external strong reducing agent, however, has a significant influence on the crystallinity and porosity of frameworks. To overcome this issue, other reducing agents have also been explored. Li et al. have demonstrated the efficiency of hydrogen as a reducing agent in controlling the size and position of NPs in click-based porous organic polymers (CPPs).72 Here, NaBH4 was used as a reducing agent, where Pd2+@CPP-1 was converted into PdNP@CPP-1 with dual size distribution of Pd NPs, as shown in Fig. 3b and d. The interior of CPP-1 was decorated with 1.4 nm Pd NPs, while the exterior part was decorated with 3.9 nm Pd NPs. It is assumed that due to the hydrophobic nature of the polymer, aqueous NaBH4 could not penetrate efficiently into the pores of CPP, and hence, some Pd ions diffuse out of the pores, leading to the formation of Pd NPs at the exterior of the pore. In contrast, when H2 was used as a reducing agent, nucleation of Pd nanoparticles occurred within the nucleus. Growth of the nanoparticles was restricted by the polymer, resulting in uniformly distributed smaller-sized particles (∼1.4 nm) as shown in Fig. 3b and c.
Beyond chemical reduction, alternative energy-assisted methods can also generate nanoparticles within frameworks. For instance, NPs can be generated inside frameworks by pyrolysis, UV radiation, or microwave irradiation. In this direction, Bhadra et al. developed an in situ approach in which Pd metal was inserted into the precursor of the COF.73 The synthesis of Pd@TpBpy was performed by the reaction of 1,3,5-triformylphloroglucinol (Tp) with 2,2′-bipyridine-5,5′-diamine palladium chloride (Bpy-PdCl2) in the presence of aqueous acetic acid and solvents. The system was heated at 90 °C, and the Pd-NPs were formed by the breakage of the Pd–N bond. Additionally, Kalidindi et al. demonstrated uniform distribution of smaller Pd nanoparticles (2.4 nm) by the reduction of Pd-loaded COF-102 under UV irradiation.74
Together, these studies highlight the importance of carefully selecting reducing conditions to control nanoparticle formation and preserve framework integrity. Similarly, innovative strategies have combined nanoparticle formation with conductive coatings. Chen et al. developed a unique method to form a coating of ultra-small Pd nanoparticles along with a conductive layer of poly(3,4-ethylenedioxythiophene) (PEDOT) on a hydrazone-linked COF (COF-42).75 First, a Pd2+-ions-loaded COF was synthesized by dispersing COF-42 in the H2PdCl4 solution. Subsequent addition of EDOT (the monomer unit of PEDOT) changed the color of the mixture from ginger brown to dark brown, indicating oxidative polymerization of EDOT monomers to PEDOT polymer and formation of Pd nanoparticles. Here, PdCl42− acted as an oxidant and was reduced to Pd NPs. The coordinating sites present in the PEDOT polymer further attracted Pd ions to deposit on the surface, resulting in uniformly distributed small-sized nanoparticles on the COF surface.
Electrochemical methods also provide alternative in-situ reduction routes. Pan et al. demonstrated an electrochemical reduction technique for the synthesis of Au nanoflower incorporated thiolated COF.76 For this synthesis, the COF was initially modified with –SH groups. A mixture of COF-SH and carbon nanotubes was deposited on a glassy electrode, which was then dipped in a 1 mM solution of HAuCl4. A nanoflower-decorated COF-SH/CNT was formed at a potential of −0.2 V for 60 s.
Interestingly, the sequence of reduction also influences the final structure of bimetallic NP-encapsulated frameworks. Pan et al. showed that simultaneous reduction of HAuCl4 and H2PtCl6 led to randomly distributed Pt and Au nanoparticles inside UiO-66 MOF.77 However, sequence addition produced different architectures: when HAuCl4 was added to Pt@UiO-66, followed by NaBH4 reduction, a composite structure formed with Pt nanoparticles at the core and Au nanoparticles distributed inside the MOFs. Similarly, when Ag+ ions were impregnated and reduced in Au@ZIF-8, it led to the formation of Au@Ag@ZIF-8. However, reversing the order, i.e., addition of Au+ ions to Ag@ZIF-8, followed by reduction, yielded Au@AuAg@ZIF-8. This difference arises from the reduction potentials of metals. Compared to Ag+/Ag, Au3+/Au has a greater reduction potential and hence when Au3+ is added to Ag@ZIF-8, part of the Ag metal is utilized in the reduction process of Au3+ ions, and the remaining Ag+ ions and Au3+ ions form an AuAg alloy shell via co-reduction, as shown in Fig. 3e.78
The strength of the reducing agents also plays a critical role in determining NP structure. Chen et al. demonstrated this using Pd- and Co-ion-impregnated MIL-101. Interestingly, the use of a strong reducing agent such as NaBH4 led to the formation of PdCo alloy nanoparticles encapsulated in MIL-101. In contrast, using aminoborane (a weaker reducing agent) led to the formation of Pd@Co core–shell nanoparticles inside MIL-101, as shown in Fig. 3f.79 A similar strategy was adopted by Xie et al. to prepare an Au nanoneedle@zirconium-based porous coordination network (AuNN@PCN-222).80 Here, Au3+ ions were reduced by carboxylate ions present in the framework. However, reduction with NaBH4 resulted in the formation of Au NPs on the surface of PCN-222, as shown in Fig. 3g. This was attributed to slower reduction with carboxylate ions, which allowed crystallization of Au nanoneedles (NNs), while rapid reduction by NaBH4 triggered multiple nucleation events leading to the formation of nanoparticles. The larger diameter of Au NNs compared to the pore channel indicated partial framework erosion due to node–linker linkage.
Difference in reduction potential can also be exploited to develop a series of bimetallic alloy NPs inside the frameworks. According to Chen et al., under high-intensity ultrasonic irradiation, transition-metal NPs (Cu, Co, Ni) can undergo galvanic exchange with noble-metal ions (Pd, Ru, Pt), producing ultrafine alloy NPs within MIL-101, as illustrated in Fig. 3h. Ultrasonic irradiation initiated galvanic replacement reactions, yielding alloys such as CuPd, CuRu, CuPt, CoPd, CoRu, CoPt, NiPd, NiRu, and NiPt were formed using this protocol.20,82
Additionally, the precursor addition procedure influences final NP morphology. Hermannsdörfer et al. showed that bimetallic Ni/Pd nanoparticles could be formed by reducing Ni and Pd precursors loaded into an MIL-101 MOF. When both precursors were loaded simultaneously, smaller nanostructures resulted, as shown in Fig. 3j. Conversely, larger Pd nanostructures were formed when Ni precursor was loaded after Pd nanostructure formation, as shown in Fig. 3i.81 In this case, aminoborane reduced Pd2+ (with higher reduction potential than Co2+), while Co2+ ions were subsequently reduced by M–H species generated during the hydrolysis of the reducing agent.
 | ||
| Fig. 4 (a) Zn-MeIM and Co-MeIM coordination mode in ZIF-8 and ZIF-67, where the tetrahedrons represent primary building units in ZIFs. Left: change in crystal structure during the process of constructing the yolk–shell structure. Adapted from ref. 83, with permission from Elsevier, copyright 2021. Schematic drawings of crystal structure transformation, morphology changes (SEM, TEM, scanning TEM, or optical microscope images) of (b) ZIF-67@ZIF-8, (c) Cu-HKUST-1@Zn-HKUST-1, (d) Al-MIL-53@Fe-MIL-53 (scale bars: 300 nm), (e) Zn/Cu-HKUST-1 (scale bars: 50 mm), (f) Cu/Zn-ZIF-L (scale bars: 400 nm), (g) Cu/Zn-ZIF-8 (scale bars: 100 nm), (h) Zn/Pd-HKUST-1, and (i) Cu/Zr-UiO-66-NH2. Adapted from ref. 83, with permission from Elsevier, copyright 2021. (j) Schematic representation of the synthesis of the Cu2WS4 nano-homojunction@COF hybrid material.100 Adapted from ref. 100, with permission from the American Chemical Society, copyright 2022. (k) Synthesis of CuZn@UiO-bpy through in situ reduction of post-synthetically metalated UiO-bpy. Cu2+ ions were coordinated to the bpy groups while Zn2+ ions were attached to the SBUs. Adapted from ref. 93, with permission from the American Chemical Society, copyright 2017. (l) The 6-anionic PCC-2b hardly encapsulates metal cluster precursors, by showing bulky and aggregated nanoclusters. The highly reactive Co nanoclusters encapsulated by PCC-2a, show considerable activity improvement for the hydrolysis of ammonia borane. (m and n) TEM images of Co NCs@PCC-2a (red cycles indicate single NCs) and Co NCs/PCC-2b.101 Adapted from ref. 101, with permission from Wiley, copyright 2018. | ||
The duration of reduction is another important factor. Timely reduction of Cu/Zn bimetallic MOFs results in the reduction of Cu clusters in the outer layer, producing a quasi-core–shell structure (Fig. 4e). Similarly, selective reduction of Cu ions in evenly distributed bimetallic MOFs such as Cu/Zn-ZIF-L and Cu/Zn-ZIF-8 leads to the formation of evenly dispersed Cu nanostructures encapsulated within MOFs, as shown in Fig. 4f. These examples confirm the general utility of site-selective strategies for forming metal nanostructures enclosed in MOFs. Reduction efficiency and the resultant structure also depend on the strength of the reducing agent, which can be tuned by the use of different solvents. For example, site-selective reduction of ZIF-67@ZIF-8 with NaBH4 dissolved in an ethanol–water mixture produced intact MOF–metal composites. However, when NaBH4 was used in water alone, it acted as a powerful reducing agent, completely disintegrating the ZIF-8 shell and leaving only the Co–B core. However, the mixture of water with NaBH4 controlled the reduction process.
For instance, Tao et al. investigated solvent effects on the growth of Pd NPs inside a triangular bipyramid covalent organic cage (Phos-cage) containing two triphenylphosphine and three diamine moieties. They dispersed Phos-cage and K2PdCl4 in solvents such as pyridine, THF, and methanol and reduced with NaBH4. In pyridine, larger Pd nanoparticles (∼3.39 nm) were obtained compared to THF (∼1.19 nm) and methanol (∼0.85 nm). Pyridine has a stronger affinity towards Pd, and faster ligand exchange between Cl− of [PdCl4]2− occurs, leading to the formation of a large pyridine–Pd complex. The complex could not enter the pore of the core, and successive reduction leads to the formation of Pd nanoparticles outside the core. Methanol has low coordination ability with Pd and does not affect the entry of [PdCl4]2− inside the cage. Hence, smaller NPs were formed inside the cage after reduction.87
Nitrogen functionalization can also stabilize NPs. For instance, Jiang et al. reported that nitrogen-rich covalent organic frameworks can control the local Pd nanoparticle environment to achieve selective semi-hydrogenation of alkynes.88 In this work, high Pd–N coordination and framework confinement were used to control Pd dispersion and electronic structure. Lan et al. showed that exfoliation of CTFs exposed more nitrogen groups, which stabilized Ag NPs.89 Ultrafine Ag NPs were embedded by adding AgNO3 precursor to exfoliated CTF nanosheets, followed by reduction with NaBH4. XPS analysis confirmed N–Ag coordination as the stabilizing mechanism.
Similarly, sulfur-based functionalization enables ultrafine NP growth. Lu et al. proposed an innovative strategy utilizing COFs as templates for the controlled synthesis of ultrafine, stable, and well-dispersed metal NPs. They designed and synthesized a thioether-functionalized COF to confine the growth of metal NPs. The uniform distribution of thioether groups within the ordered framework facilitated the formation of platinum (Pt) and palladium (Pd) nanoparticles averaging 1.7 ± 0.2 nm in size, exhibiting a narrow size distribution. This was achieved by impregnating the COF with K2PtCl4 and K2PdCl4, followed by reduction using NaBH4. A similar process with the same crystalline COF but without thioether groups leads to the formation of both small and aggregated Pt/Pd NPs inside frameworks.90 Further, Mishra et al. have developed a novel thiol-functionalized MOF as a template for the controlled synthesis of highly dispersed ultrafine yet stable ruthenium (Ru) nanoparticles. They have shown that the Ru NPs of 1.5 nm size are well stabilized with thiol-functionalized Ni-MOF.91
Beyond stabilization, functional groups can direct precursor incorporation. Chen et al. incorporated Pd ions into UiO-67 using 2,2-bipyridine-5,5′-dicarboxylic acid as a ligand, which acted as an anchoring site due to its structural similarity to biphenyl dicarboxylic acid (the native UiO-67 linker). Reduction with H2 yielded Pd NPs.92 Similarly, Wang et al. synthesized defect-rich Cu2WS4@COF by pre-anchoring W ions in COF-LZU1, followed by hydrothermal treatment with CuCl2 and thioacetamide. The synthesis procedure is shown in Fig. 4j.35,100
Bimetallic nanostructures within frameworks are formed through a strong interaction between metal ions and the functional group of frameworks. Lin et al. demonstrated this by synthesizing ultra-small Cu/ZnOx NPs on preassembled bpy and Zr6(μ3-O)4(μ3-OH)4 sites in UiO-bpy.93 Here, for the formation of Cu/ZnOx NPs, Cu2+ ions were first coordinated onto the bpy sites present in MOF by post-synthetic metalation with CuCl2, followed by the introduction of Zn2+ ions by reaction with ZnEt2 with the μ3-OH sites on the SBUs. Reduction of these ions with H2 at 250 °C leads to the formation of ultra-small Cu/ZnOx NPs (1 nm) within the MOF, as shown in Fig. 4k. The robust interactions between Cu/ZnOx NPs and the bpy groups of the ligands as well as Zr6 SBUs stabilize these ultra-small NPs, preventing their agglomeration and phase separation between Cu and ZnOx.
Similarly, Ding et al. have shown that diamine-containing UiO-66 (UiO-66-(NH2)2) can effectively coordinate with the precursor of Pd/Au NPs during the preparation and hence result in smaller-sized particles.94 These UiO-66-(NH2)2 also efficiently stabilized the PdAu NPs during the catalysis process compared to UiO-66-NH2, thereby significantly accelerating the dehydrogenation of formic acid.
| Nanostructure precursor | Framework | Resultant structure | Approach adopted | Important observation | Ref. |
|---|---|---|---|---|---|
| HAuCl4 | Cu-MOF | Au–Pd decorated Cu-MOF | Reduction with NaBH4 | Cu2+ ions present in the MOF are not reduced | 102 |
| PdCl2 | |||||
| AgNO3 | CTFs | Ag0@CTF nanosheets | Exfoliation of CTFs followed by reduction with NaBH4 | Exfoliation of CTFs enhances | 89 |
| HAuCl4 | COF | Au nanoflower/SH–COF/CNTs | Electrochemical reduction | Functional group influences the resultant structure | 76 |
| Co–Mn metal within MOF | CoMn-MOF-74 | Co/MnOx nanoparticle-encapsulated quasi-MOF | Controlled pyrolysis | By carefully deligandating CoMn-MOF-74, the Co/MnOx NPs were in situ enclosed within quasi-MOF-74 | 98 |
| Cu present within MOF | CuCo-MOF | Cu nanowire-encapsulated quasi-MOF | Controlled pyrolysis (300 °C, N2 atmosphere) | Mild pyrolysis retains framework integrity while forming in situ Cu nanowires, enhancing catalytic oxidation efficiency | 99 |
| Pd present in 2UiO-67 framework | UiO-67 | Pt NPs@UiO-67 | Reduction by solvent molecules at high temperature (130 °C) | N atoms present in the OF stabilize the Pd NPs | 103 |
| Copper(II) nitrate hydrate and palladium(II) nitrate hydrate | MIL-101 | 2–3 nm Cu, Pd, and Cu–Pd alloy nanoparticles are encapsulated in MIL-101 | Microwave irradiation | Microwave irradiation simultaneously activates the pores of MIL-101 and reduces metal ions | 104 |
| Chloroauric acid | UiO-66 | UiO-66 having uniform distribution of Pt- and Au-nanoparticles | Simultaneous and sequential reduction with NaBH4 | The sequence of nanoparticle addition and reduction influences the final structure | 77 |
| H2PtCl6 | |||||
| UiO-66 having Pt-NPs at the center and Au-NP distributed inside MOF | |||||
| AuCl3, PdCl2, Pd (OAc)2 | MOF-253 | Au–Pd@MOF-253 | Sequential deposition and reduction (with H2) of Au and Pd on MOF-253 | Selectivity for the carbonylation of amines was obtained by Au–Pd/MOF catalysts through adjusting the molar ratio of Au and Pd | 105 |
| K2PtCl4 | UiO-66-NH2 | Pt–Sn alloy encapsulated within UiO-66-NH2 | Sequential impregnation of Pt and Sn, followed by reduction with H2 | Control over Pt and Sn concentration can be achieved by sequential impregnation | 106 |
| SnCl2·2H2O | |||||
| Pd(NO3)2·2H2O | MIL-101 | PdCo alloy@MIL-101 | Reduction with aminoborane | Electrochemical reduction potential difference between two metals leads to the formation of a core–shell structure with a weak reducing agent | 79 |
| CoCl2·6H2O | |||||
| Pd@Co core–shell@MIL-101 | Reduction with sodium borohydride | ||||
| CuCl2·2H2O | MIL-101 | CuPd, CuRu, CuPt, CoPd, CoRu, CoPt, NiPd, NiRu, and NiPt@MIL-101 | Galvanic replacement | Electrochemical reduction potential difference between two metals | 82 |
| PdCl2 | |||||
| RuCl3·xH2O | |||||
| Pt(NO3)2 | |||||
| Pt(NO3)2·2H2O | MIL-101 | PdAg@MIL-101 | Double-solvent approach | Very small PdAg alloy NPs (∼1.5 nm) have been encapsulated into MIL-101 | 86 |
| AgNO3 | |||||
| Et3PAuCl | NU-1000 | Au@NU-1000 | Modification of MOF to bind specific Au(I) phosphine, followed by condensed phase reduction | Pore size of MOF controls the size of NPs | 2 |
| CuCl2 | UiO-bpy | CuZnOx@UiO-bpy | In situ reduction during CO2 hydrogenation | Strong interactions between Cu/ZnOx NPs and the bpy groups of the ligands and Zr6 SBUs stabilize these very small NPs | 93 |
| ZnEt2 | |||||
| HAuCl4 | PCN-222 (Cu) | Au nanoneedle@PCN-222 (Cu) | Impregnation of Au3+ ions followed by electronic activation of N sites within the Cu-porphyrin MOF | Reduction with –RCOO− leads to the formation of AuNN@PCN-222 (Cu), and reduction with NaBH4 leads to the formation of AuNPs on the surface of PCN-222 (Cu) | 80 |
2.2. Post-synthesis approach
Compared to the in situ synthesis method, this approach avoids the issues associated with nanostructure formation on the external surface of the frameworks and the structural damage of frameworks arising from impregnation reduction. This method offers the benefit of incorporating metal nanostructures with different sizes, shapes, and compositions. For example, ZIF-8 MOFs can be grown on the hollow TiO2 NP surface using a sonochemical approach.107However, this approach often relies on trial-and-error optimization. Harsh conditions, such as high temperature and low pH, can accelerate aggregation of nanostructures. Moreover, it is difficult to place nanoparticles uniformly inside each framework, resulting in a low yield of nanostructure-encapsulated frameworks in self-assembled structures. This approach has been adopted for confining nanostructures within different types of frameworks such as MOFs and COFs. Additionally, it has also been extended to organic molecular cages (OMCs). For instance, Inomata et al. recently demonstrated that thiolate-protected Au55 clusters, Grubbs's second-generation ruthenium catalysts, and zinc meso-tetraaryl porphyrins may be directly mixed to encapsulate Au55 clusters in hexaporphyrin cages in a controlled manner.54 Through the cage pores, tiny substrates can reach the Au55 cluster cores.
However, this principle is not universal. Some nanostructures require a specific number of ligands for stability, and their dimensionality also depends strongly on ligand concentration. Yang et al. showed that altering ligand concentration can even affect the location of metal NPs.109 When Au nanoparticles anchored on ZnO nanorods were dispersed in DMF/H2O containing 2-methylimidazole (Hmim) and kept at 70 °C for 12 h, the formation of ZnO@Au@ZIF-8 resulted. At low concentrations, Hmim reacted with Zn2+ ions at the surface of ZnO, producing ZnO@Au@ZIF-8 with Au NPs located on the surface. At high concentrations of Hmim, Au NPs were embedded inside the MOF shell.
This ligand concentration-dependent positioning of metal nanoparticles can be attributed to two distinct formation mechanisms.110 At high ligand concentrations, the dissolution–precipitation mechanism, as shown in Fig. 5a, is operative, wherein parent oxides dissolve to yield metal ions into solution, followed by the nucleation and growth of MOFs. During this process, metal nanoparticles become immobilised at the oxide–MOF interface and are subsequently embedded within the MOF matrix as crystallisation proceeds. Conversely, under the localised conversion mechanism, as shown in Fig. 5b, where insoluble oxide or hydroxide precursors are directly converted into MOFs through acid–base interactions, with no ion release into solution. In this case, metal nanoparticles are positioned near the MOF surface rather than being entrapped within the framework. Time-dependent structural evolution corresponding to each mechanism is shown in Fig. 5c–h and Fig. 5i and j, respectively.
 | ||
| Fig. 5 (a and b) Illustration of two mechanisms for the position-regulated encapsulation of MNPs into ZIF-8. (c–h) TEM images of the product synthesized at high Hmim concentration for various reaction times of (c) 0.5 h, (d) 1 h and (e) 5 h and with low Hmim concentration at the reaction times of (f) 0.5 h, (g) 1 h and (h) 5 h. (i) Concentration of Zn2+versus reaction time and (j) pH versus reaction time of the ZnO@Au@ZIF-8 at low (black line) and high (red line) concentration of Hmim, respectively. Scale bar: 100 nm. (Variation with pH needs to be explained.) Adapted from ref. 109, with permission from Nature, copyright 2017. (k) Oligomer evolution during COF shell formation on ligand-modified (PVP, PEI, PVP + PEI) and unmodified SiO2 nanoparticles, based on the LaMer model. C1–C3 indicate critical heterogeneous nucleation concentrations on PEI-, PVP + PEI-, and PVP-modified SiO2, respectively, while C4 and C5 denote critical concentrations for homogeneous and heterogeneous nucleation on unmodified SiO2. (l) Schematic diagram showing the interaction between the dual ligands and monomers. (m) Correlation between the PVP concentration and the thickness of the COF shell in SiO2@COF core–shell nanoparticles synthesized using the dual-ligand-assisted method. Scale bars: 50 nm. Adapted from ref. 111, with permission from Nature, copyright 2021. (n–q) Schematic and TEM images of the coordination-assisted oxidative etching process. (n) Initial solid Pt–Ni polyhedral. (o) Pt–Ni frame@MOF intermediates I. (p) Pt–Ni frame@MOF intermediates II. (q) Final Pt–Ni frame@MOF. Scale bars: 50 nm (insets are the magnified TEM images. Scale bars: 5 nm).117 Adapted from ref. 117, with permission from Nature, copyright 2015. (r) Tracking the localized surface plasmon resonance (LSPR) shift of the aluminum (Al) core over time during the growth of the MIL-53(Al) MOF shell, performed with varying initial concentrations of acetic acid (HOAc) in the reaction mixture. The extent of the LSPR shift is categorized into regions (i) and (ii), corresponding to the dissolution and shell growth phases, respectively. (s) Representative TEM images of Al NC@MOF heterostructures synthesized without acetic acid (orange) and with 5 mM (blue), 15 mM (red), and 30 mM (green) acetic acid. Increasing the HOAc concentration in the starting mixture progressively enhances the dissolution rate of the oxide layer, resulting in more pronounced shrinkage of the Al core and the formation of a thicker, denser MOF shell. Adapted from ref. 115, with permission from American Association for the Advancement of Science, copyright 2019. (t) TEM images of LoP@UiO-66 and time-dependent studies of the synthesis of LoP@UiO-66. (u) TEM images of HiP/UiO-66 and time-dependent studies of the synthesis stages of HiP/UiO-66. A high concentration of capping agents on Pt NPs leads to a decrease in the surface energy. Adapted from ref. 108, with permission from Chemical Science, copyright 2023. | ||
Building on this, Chen et al. have demonstrated a dual-ligand strategy to create NP-COF heterostructures (Fig. 5k–m).111 SiO2 NPs were first modified with PEI and PVP, followed by addition of COF precursors, 1,3,5-triformylbenzene (TFB) and 1,4-phenylenediamine (PDA). Two competitive reactions occurred: (i) homogenous nucleation, leading to COF NP formation, and ii) heterogeneous nucleation, forming SiO2@COF. The critical concentration for homogeneous nucleation (C5) is fixed for a particular condition, but for heterogeneous nucleation depends on the surface properties. Critical concentration for heterogeneous nucleation (C4) on a naked SiO2 surface is the same or even higher than that of homogeneous nucleation. When NPs were modified with PEI, Schiff base reaction accelerated the deposition of COF precursors on the surfaces of NPs, lowering the critical nucleation concentration. However, too-fast growth of the COF resulted in a non-uniform shell. When NPs' surfaces were coated with PVP, COF precursors prefer to deposit on the surfaces of NPs because of the H-bonding. This process is much slower than with PEI. The concentration of PVP and PEI can be varied to control the rate of the reaction and, hence, the shell thickness and uniformity.
The positioning of metal NPs within the MOF layers can be controlled by adjusting the crystallization process of the MOFs through varying the concentration of organic ligands. Acharya et al. developed a unique strategy to create a skin-like thin layer of COF on the surface of plasmonic Ag nanoparticles coated with hollow silica.4 Hollow silica helped to keep Ag nanocrystals separated, which made the surface of these nanocrystals accessible. These h-SiO2-capped Ag nanocrystals were immersed in a solution containing diol (DAL, 1,4-benzenedimethanol) and triamine [TAE, 1,3,5-tris(4-aminophenyl)benzene]. Exposure of this solution to a blue laser of 405 nm for 1 h led to the formation of a thin coating of COF on the surface of Ag nanocrystals. With the help of 1H NMR, it was proven that the surface of Ag nanoparticles helped in the oxidation of DAL to dialdehyde upon laser irradiation, and a COF layer was formed by the aldehyde–amine condensation-crosslinking reactions. However, direct condensation of aldehyde and amine on the Ag@h-SiO2 surface led to the formation of a bulk COF layer around Ag@h-SiO2. Replacement of light with heat also does not lead to the formation of a thin layer.
Coordination of ligands with metals also controls the NP–framework heterostructure. Li et al. used a Pt–Ni frame as a sacrificial template for the growth of Pt–Ni@Ni-MOF-74. Ni2+ ions formed after reaction of Ni0 with oxygen diffused outward and were captured by MOF precursors, forming Pt-enriched Pt–Ni@Ni-MOF-74, as shown in Fig. 5n–q. Similarly, Chen et al. demonstrated that selective etching of Co2+ from an alloy by ligands results in a ternary alloy encapsulated by different MOFs.112 RhCoNi nanoflowers (NFs) were used as a sacrificial component. Solvothermal treatment of RhCoNi NFs with 2-methylimidazole (HmIM) led to preferential etching of Co2+ ions. These ions reacted with HmIM to form ZIF-67 (Co) MOF. In a subsequent step, repetition of the same process with 2,5-dihydroxyterephthalic acid (DOT) resulted in the formation of MOF-74(Ni)-encapsulated RhCoNi NFs owing to the stronger coordination ability of Co2+ ions with DOT.
In addition to etching strategies, organic modulators can also be employed to tune NP@MOF growth. Organic molecules act as modulators and strongly influence the size and uniformity of NP@MOF. Wang et al. demonstrated that controlling the nucleation rate using modulators can regulate the crystal size of MOFs. Specifically, they synthesized Pt- and Pd-encapsulated MOFs using acetic acid as a modulator.113 Acetic acid competed with dicarboxylic linkers to react with zirconium ions (Zr4+) during the nucleation and growth of NP@UiO-66, thereby providing control over particle size.
In a related strategy, synthesis was initiated by solution-phase binding of metal ions using poly(amidoamine) (PAMAM) dendrimers. The positively charged dendrimers were incorporated into the negatively charged pores of Zr-based MOFs (NU-1000). The extent of electrostatic interactions, and hence NP size and distribution, could be tuned by adjusting the solution pH.
In addition to ligand addition rate, the availability of metal precursors also plays a critical role in nanoparticle growth. H. Robatjazi et al. showed that in post-synthetic approaches, the availability of metal precursors can be modulated by adjusting reaction conditions, which in turn influences nanoparticle growth.115 For example, Al nanoparticles were converted into Al@MIL-53 by the addition of 1,4-benzenedicarboxylic acid (H2BDC) ligands. As no external source of Al precursor was supplied, it was proposed that Al3+ ions present in the surface aluminium oxide layer were dissolved and react with H2BDC to produce MIL-53 around Al cores. This transformation follows a dissolution-growth mechanism. Evidence was provided by the increase in MOF shell thickness with rising pH (controlled by adding acetic acid). At higher pH, the rate of Al dissolution increased, while MIL-53 nucleation was suppressed due to reduced linker deprotonation. As the reaction proceeded, the solution's pH gradually rose, increasing the concentration of deprotonated linkers, which then initiated MOF growth and simultaneously inhibited further dissolution of the Al nanoparticle core.
The dissolution-growth process was monitored using the sensitivity of the localized surface plasmon resonance (LSPR) of Al@MIL-53, which depends on the size of the Al core and the refractive index of the surrounding medium. Time-dependent changes in LSPR spectra, along with TEM images for different acid concentrations, are shown in Fig. 5r. The analysis revealed two distinct stages. The blue region (region I) indicated that the reaction was dominated by Al dissolution, while in region II, MOF growth became the dominant process. As shown in Fig. 5s, the magnitude of the LSPR blue shift (region I) increased with higher acid concentrations, indicating greater dissolution of Al ions. This, in turn, facilitated the subsequent growth of the surrounding MIL-53 layer.
| Nanostructure | OF | Resultant structure | Importance | Ref. |
|---|---|---|---|---|
| PVP stabilized Pt NPs | UiO-66 (MOF) | Pt@UiO-66 core–shell | Surface energy mediated encapsulation | 108 |
| PVP stabilized Au nanorods/Au@Pt nanorods/Au@Pt nanotubes | ZIF-67 and ZIF-8 | Multiple Au nanorods inside ZIF | • Multicore NRs@OF can be synthesized | 118 |
| • The size of OFs can be controlled by controlling ligands | ||||
| Au@Pt nanorods inside ZIF | ||||
| Au@Pt nanotubes inside ZIF | ||||
| Thiolated poly(ethylene glycol) stabilized Au nanorods | NU-901 | Au NRs @NU-901 and Au NRs @NU-1000 | • Interaction between thiolated PEG and MOF precursor promotes encapsulation | 114 |
| • Increase in concentration of Au nanorods decreases the size of the crystallite | ||||
| Pt and Pd nanoparticles | MOFs (nano-UiO-66, nano-UiO-66-NH2, nano- MOF-801) | Pt@nano-MOFs | • Nucleation rate of MOFs on the nanoparticles can be controlled by the addition of modulators | 113 |
| Pd@nano-MOFs | ||||
| • Slow nucleation of MOF on the nanoparticles leads to NP@nano-MOF formation | ||||
| Ag@h-SiO2 nanostructure | COF | Ultrathin COF layer on the surface of Ag nanostructures | • Surface of Ag nanocrystals helps in the oxidation of alcohol to aldehyde upon irradiation of light | 4 |
| • The formation process is specific to light | ||||
| ZnO NRs@Au NPs | ZIF-8 | ZnO NRs@Au@ZIF-8 composites with Au NPs at different locations | • At high concentration of ligand, dissolution–precipitation mechanism occurs | 109 |
| • At low concentrations of ligands, a localized conversion mechanism occurs | ||||
| SiO2 NP, upconversion NPs, semiconductor NPs | COF | NPs@COF | • Bowl-shaped, yolk@shell, core@satellites@shell nanostructures of several NP@COFs can also be fabricated using dual-ligand strategy | 111 |
| TiO2 hollow nanosphere | ZIF-8 | Double-shell TiO2@ZIF-8 | • A double-shell TiO2@ZIF-8 structure is successfully obtained via carboxylation and sonocrystallization process | 107 |
| PVP-stabilized Au@AgPt nanorattles | ZIF-8 | Au@AgPt encapsulated within ZIF-8 | • PVP plays an important role in the encapsulation process | 119 |
| Au55 clusters | Hexaporphyrin cages | Au55 | • Small substrates can enter the caged cluster through the apertures because there is an interstitial gap between the core and the porphyrin shell | 120 |
| Pt/CeO2 NPs | UiO-66-NH2 | Pt/CeO2@UiO-66-NH2 | • Coating of Pt/CeO2 with polystyrene sulfonate facilitates electrostatic interaction | 121 |
| • Use of microwave leads to uniform growth of MOFs on the surfaces of NPs | ||||
| CdS NRs | Cu-TCPP | Core–shell CdS@Cu-TCPP NRs | • With CdS NRs, Cu-TCPP has a sheetlike structure, but when grown on CdS NRs, Cu-TCPP forms around the NRs | 122 |
| Pt NPs | Porphyrinic MOF (PCN-224-RT) | Pt@PCN-222-RT | • A facile stirring method at RT | 123 |
| • To obtain Pt@PCN-222 within 24 hours |
2.3. One-pot synthesis
The “one-pot” approach refers to directly mixing precursors of nanostructures with precursors of frameworks. This direct combination leads to the formation of NP@framework structures via a self-assembly process. It is a simple and efficient strategy, yet still relatively unexplored for preparing nanostructures. Additionally, using this approach, the diffusion resistance between the outer and the inner surfaces of the frameworks can be efficiently avoided.In one of the earlier studies, Li et al. demonstrated the encapsulation of Pd NPs within UiO-67 MOF functionalized with platinum bipyridine coordination complexes by using a one-pot strategy.124 Similarly, Wang et al. developed a capping and reducing-agent-free strategy to obtain Pt@MOF-s-T3.125 The synthesis procedure is shown in Fig. 6a. In this case, uniform and well-dispersed Pt NPs formed inside the cavities of MOFs, attributed to the strong complexation of Pt ions with dipyridyl groups and the stabilization provided by dipyridyl ligands.
 | ||
| Fig. 6 (a) Schematic illustration of in situ one-step synthesis of Pt@MOFs-T3. Adapted from ref. 125, with permission from the American Chemical Society, copyright 2020. (b) Preparation of Au NPs@COF with two different methods. Adapted from ref. 126, with permission from the American Chemical Society, copyright 2024. (c) Schematic representation of the incorporation of PdNi NPs in UiO-67. The assembly of MOFs and the incorporation of metal precursors are performed in a single step. Adapted from ref. 127, with permission from Wiley, copyright 2015. (d) Diagrammatic representation of the Ag–AgMOM one-step in situ solvothermal synthesis. (e) Enlarged TEM images of Ag–AgMOM; the lattice of AgMOM was observed. Adapted from ref. 128, with permission from Nature, copyright 2023. | ||
The advantage of the one-pot approach over conventional in situ synthesis was further highlighted by Gamraoui et al., who achieved the encapsulation of ultra-small Au NPs inside a COF.126 They used N-heterocyclic carbene (NHC)-functionalized Au(I) complexes as Au NP precursors. When these NHC–Au(I) complexes were added during COF precursor assembly, they became incorporated inside the framework pores. Reduction of these NHC–Au(I)@COFs with NaBH4 led to the formation of ultra-small Au NPs inside the COF. However, addition of NHC–Au(I) complexes to preformed COF, followed by reduction, led to the formation of bigger-sized agglomerated Au NPs, as shown in Fig. 6b. This comparison clearly shows the efficiency of the one-pot method.
Reaction temperature also plays a decisive role in one-pot synthesis. Chen et al. demonstrated that by carefully controlling the reaction temperature, Pd nanoparticles could be encapsulated inside UiO-67 without the addition of any external reducing agent.103 Mixing PdCl2(CH3CN)2 with a solution of MOF precursors, i.e., ZrCl2 and 2,2′-bipyridine-5,5′-dicarboxylate (H2bpydc), in DMF at 80 °C led to the incorporation of Pd ions in UiO-67 crystals. The chelating ability of nitrogen atoms in the bipyridyl groups facilitated the incorporation of Pd ions into the UiO-67 crystals. Pd ions present in the UiO-67 framework were reduced by the solvent molecules at high temperature (130 °C). The formed nanoparticles are stabilized by the bipyridyl groups.
This strategy can also be extended to fabricate bimetallic NP@framework structures. For instance, ultrafine Pd, Ni, and PdNi alloy NPs were synthesized inside pores of MOFs using the same approach, as shown in Fig. 6c.127 Beyond MOFs, Liu et al. reported the creation of Ag–AgMOMs, hybrid structures consisting of Ag NPs and an Ag-porphyrin metal–organic matrix (MOM), in a single step, as shown in Fig. 6d. The in situ growth enabled by one-pot synthesis allowed silver atoms to be shared at the interface, thereby facilitating efficient electron transmission between Ag NPs and AgMOM (Fig. 6e).128
Finally, microwave irradiation significantly reduces the time needed for the one-pot synthesis process. Zhang et al. showed that Au NPs could be immobilized within MOD-199 within 2 min using microwave irradiation, achieving Au loadings as high as 5.13%.129
The above discussion demonstrates that various factors influence each synthesis procedure in distinct ways. These factors and their corresponding impacts are summarized in Scheme 2.
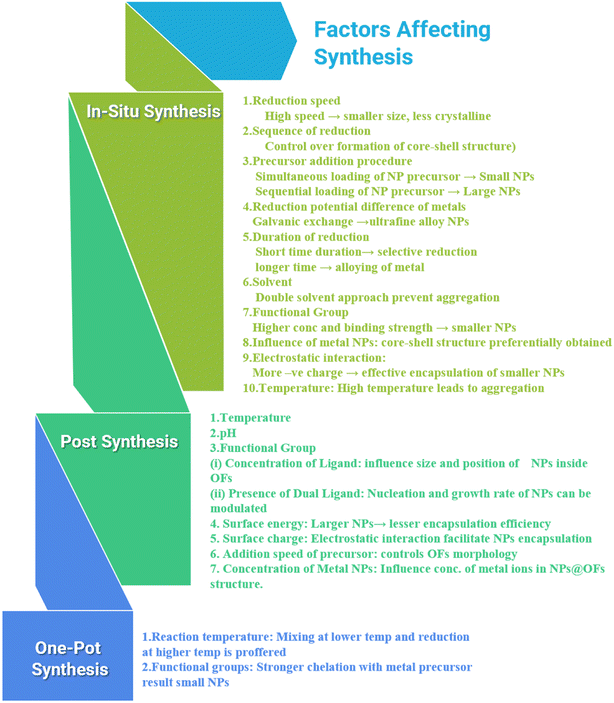 | ||
| Scheme 2 Impact of different factors on the NP@organic frameworks system obtained using different synthesis strategies | ||
3. Nanoparticle encapsulation and enhanced stability of the hybrid structures
The encapsulation of nanoparticles within organic frameworks significantly enhances the thermal and chemical stability of both nanoparticles and the host frameworks. This enhancement of stability is primarily due to synergistic electronic interactions between the NPs and the functional groups of the framework as well as from the vibrational restrictions. The structural rigidity and appropriate interactions of metal with the organic counterpart can promote the structural consistency. Ding et al. developed Pd–Au nanoparticles supported on a diamine-functionalized UiO-66 framework, which exhibited marked resistance to aggregation and could be recycled seven times during formic acid dehydrogenation.94 High stability was correlated to the enhanced density of –NH2 groups in MOFs, thereby effectively stabilizing ultra-small PdAu NPs. Similarly, in the case of amine functionality, improved stability was obtained in Pd NPs when combined with Cr-MIL-101.130Muhamed et al. showed that the strong binding affinity of Ag and the thiol group can be used to synthesise Ag NP-modified NU-1000. In this process, the NU-1000 MOF was successfully functionalized with –SH groups using the solvent-assisted ligand incorporation method. These –SH groups chelate with Ag(I) ions while simultaneously causing their reduction, resulting in the formation of Ag NPs/NU-1000 nanohybrids.131 Hamoud et al. found that the ligand-driven in situ reconstruction of the UiO-66-(COOH)2-Cu hybrid structure provides additional stability to UiO-66-(COOH)2-Cu and enhances its catalytic efficiency. Under solar radiation, the UiO-66-(COOH)2-Cu structure transforms to an anhydride analogue structure UiO-66-(COO)2-Cu. This intra-framework cross-linking via anhydride linkage provides additional stability to the framework structures.132 Zhang et al. reported the synthesis of a unique porous organic polymer (POP) containing thiazolo[5,4-d] thiazole units, namely TzPOP. The resulting TzPOP–OH has three –OH groups as binding sites in addition to N and S atoms. However, the N and S binding sites can be easily protonated under highly acidic conditions. Nevertheless, the –OH binding sites can effectively bind to Pd(II) ions in a strong acid environment. The importance of strong binding between nanoparticles and the binding groups of the organic frameworks in stabilisation of the overall system has thus been emphasised in the above-mentioned studies.
The incorporation of nanoparticles in the framework structure also limits the vibrational flexibility of the framework structure. In this regard, Wang et al. synthesised Au25@M-MOFs (M = Mg, Co, Zn, Ni) by nucleating MOFs on the surface of Au25(Cys)18 nanoclusters.133 The Au25@M-MOF exhibits enhanced photoluminescence (PL) intensity compared to Au25(Cys)18, upon excitation at 420 nm. This observation is attributed to coordination between the metal oxo chain of MOFs and the free carbonyl groups of Cys of Au nanoclusters. This imposes constraints on the vibrations and rotations of Cys ligands, leading to an increase in PL intensity. In addition to the above-mentioned factors, when frameworks are grown on the surface of NPs, a uniform, defect-controlled coating of OFs over the NP surface is observed, due to which the structure becomes more stable.
4. Factors that regulate catalytic activities
The effectiveness of metal nanoparticles in catalytic processes is influenced by several factors, such as size, morphology, surface roughness, presence of defects, surface protective agents, and the nature of the exposed crystal facets. Certain metal NPs, such as Ag, Au, and Cu, possess excellent light absorption capabilities due to their surface plasmon resonance (SPR), which significantly influences the photocatalysis process. Frameworks themselves also serve as excellent catalysts, with their performance depending on structure, dimensionality, types of metal centers, and organic linkers. Importantly, heterostructures comprising both metal NPs and frameworks do not behave solely as a function of either component. Their integration changes the intrinsic properties of both the NPs and the frameworks. Below, key factors influencing the catalytic efficiency of NP@framework structures are outlined.(i) Structure of the organic framework
The structural characteristics of frameworks strongly influence the catalytic activity of the catalyst. Wang et al. reported that Pt@ZIF-7, having varying sizes of ZIF-7, could be synthesized by controlling the nucleation and growth rate of ZIF on preformed Pt NPs.113 These Pt@nano-ZIF structures effectively catalyzed the hydrogenation of n-hexene. Pt@nano-ZIF, having 40 nm ZIF, exhibited better conversion efficiency than those with 850 nm and 1.5 μm ZIF. This is attributed to the shorter diffusion path from the MOF surface to the active sites.Strain in the frameworks also influences the catalytic activity of NP@frameworks. The incorporation of NPs can induce strain, which modifies the catalytic activity of NP@framework systems. For instance, in the in situ synthesis, excessive loading of metal precursors may deform frameworks with small pores, and reaction conditions may further accelerate framework degradation. Nevertheless, these quasi-NP@quasi-framework structures often develop strong interactions between NPs and frameworks. Tsumori et al. prepared an Au/MIL-101 structure via a controlled deligandation process. These Au NP-quasi-MIL-101 structures have stronger interactions between Au NPs and Cr–O metal nodes. These interactions activated Cr–O sites for O2 uptake and activation, thereby enhancing CO oxidation.134 Similarly, Yang et al. highlighted that strain induced during NP incorporation impacts electrocatalytic efficiency. Using the vapor-phase infiltration (VPI) method, they introduced Zn coordination sites into MOFs. Prolonged VPI treatment converted these sites into ZnO NPs, introducing strain in the system.135 This strain enhanced the electrocatalytic reduction efficiency and selectivity of the MOF.
(ii) Size of the nanoparticles
The size of NPs directly influences their surface area, electronic structure, and thus catalytic activity. Wang et al. revealed that by modulating the ligand, the pore size of the frameworks could be adjusted, which in turn controlled NP size, as shown in Fig. 7a. They further reported that the size of Pt NPs influenced the efficiencies of the photocatalytic hydrogen evolution reaction (HER).136 Specifically, Pt@MOF structures having 5–6 nm Pt NPs were shown to have better catalytic activity than Pt@MOF structures having 2–3 nm Pt NPs. The enhanced catalytic efficiency was attributed to higher light absorbance (Fig. 7b) and more efficient electron transfer from [Ir3+(ppy)2(bpy˙−)] species to Pt NPs. A contradiction was observed by Mian et al.137 They found that Cu NPs having ∼1.5 nm placed in NU-907 exhibited better catalytic activity than ∼0.9 nm sized Cu NPs placed in NU-901 for the selective hydrogenation of acetylene to ethylene. Theoretical calculations explained this behavior: smaller, highly curved particles required higher activation energy for the reaction compared to larger, flatter surfaces, thereby reducing catalytic efficiency. | ||
| Fig. 7 (a) Synthesis procedure of Zr-carboxylate MOFs (1 and 2) followed by photoreduction of K2PtCl4 for Pt NP loading within the MOF cavities to form Pt@1 and Pt@2 structures. (b) Diffuse reflectance spectra of 1 (red), Pt@1 (black), 2 (purple), and Pt@2 (blue). A photograph of suspensions of these samples is shown in the inset. Adapted from ref. 136, with permission from the American Chemical Society, copyright 2012. (c) Synergetic influence of SPR of Ag NPs and –NH2 group in dehydrogenation of HCOOH. Adapted from ref. 143, with permission from the American Chemical Society, copyright 2017. (d) Comparative XPS spectra of Pd (3d) in pCOL-Pd/AuNC@h-SiO2 and Pd/AuNC@h-SiO2. Adapted from ref. 4, with permission from Nature, copyright 2023. (e and f) Typical TEM images of (e) Pt@UiO-66-NH2 and (f) Pt/UiO-66-NH2. (g) Photocatalytic hydrogen-production rates of UiO-66-NH2, Pt@UiO-66-NH2, and Pt/UiO-66-NH2. Adapted from ref. 145, with permission from Wiley, copyright 2016. | ||
(iii) Plasmonic effect of nanoparticles
Certain NPs, such as Ag, Au, and Cu, strongly absorb solar electromagnetic radiation because of the collective oscillation of free electrons upon interaction with light. This phenomenon is known as surface plasmon resonance (SPR).138,139 Integration of plasmonic NPs with frameworks enhances their applicability.140,141 For instance, Qin et al. reported that plasmonic Au NPs@MIL-100(Fe) displayed improved photocatalytic performance for the oxidation of benzyl alcohol when illuminated under visible light.142 This improved photocatalytic performance was attributed to the migration of SPR-excited hot electrons from plasmonic Au NPs to MIL-100(Fe), which facilitated the generation of more active O2˙− radicals.SPR can also help in the activation of non-plasmonic catalysts. Wen et al. presented a unique core–shell ZIF-8@Pd2Ag1@ZIF-67, which acted as an excellent catalyst for photocatalytic selective dehydrogenation of formic acid (HCOOH).143 Here, the catalytic activity originated from Pd sites electronically promoted under visible light due to the Ag plasmon resonance, combined with the favorable core–shell MOF architecture, as shown in Fig. 7c. In another example, Wen et al. established that plasmonic Au@Pd nanoparticles supported on titanium-doped, amine-functionalized zirconium-based MOFs (UiO-66(Zr100−xTix)) exhibited synergistic activity, significantly enhancing room-temperature hydrogen production through the photocatalytic dehydrogenation of formic acid.71 Among the tested catalysts, Au@Pd-UiO-66(Zr85Ti15) outperformed both Au-UiO-66(Zr85Ti15) and Pd-UiO-66(Zr85Ti15). This superior activity was attributed to the generation of electron-rich Pd sites through electron transfer from Au NPs to Pd. Since formic acid is electron-deficient, it preferentially adsorbs on Pd, thereby facilitating the electron transfer and catalysis.
(iv) Change in electronic environment
Integration of NPs with frameworks also modifies the electronic charge distribution of both components, with electron transfer occurring either from NP to framework or vice versa. Acharya et al. confirmed that coating of Pd/Au NPs with a thin layer of COFs altered the electronic state of both NPs and frameworks. Density functional theory (DFT)-based Bader charge analysis and XPS measurements provided evidence that coating Pd NPs with a thin layer of COFs made them more electron-rich, as shown in Fig. 7d.4 Similarly, Shen et al. unveiled the role of metal–O clusters in regulating the electronic environment of composite catalysts.144 Fe–O cluster-based MOFs decreased the electron density of encapsulated Pt nanoparticles, thereby suppressing the complete hydrogenation of phenylacetylene and enabling the selective semi-hydrogenation of phenylacetylene to styrene with ∼80% selectivity. Conversely, MOFs containing Cu–O clusters promoted the formation of highly oxidized Pt NPs and released Cu2+ ions, which jointly contributed to the complete deactivation of Pt NPs during hydrogenation.(v) Position of nanoparticles inside the MOF
The spatial location of NPs within frameworks critically affects catalytic behaviour. Xiao et al. synthesized Pt NPs inside UiO-66-NH2, termed as Pt@UiO-66-NH2, and compared them to Pt NPs outside the framework, as illustrated in Fig. 7e and f.145 Pt@UiO-66-NH2 showed superior H2 production efficiency because the shorter electron transfer pathway facilitated faster charge migration, as shown in Fig. 7g. Consistently, other studies have highlighted that NPs positioned near framework surfaces often exhibit higher catalytic activity than those deeply confined within the MOF matrix.109Beyond these key factors, catalytic efficiency is also influenced by structural changes during catalysis, reaction conditions, and the concentration of NPs within frameworks. Each of these parameters can alter stability, accessibility of active sites, and overall catalytic performance.
5. Photocatalytic applications
Integrating nanostructures with frameworks offers several key advantages: (i) the formation of a Schottky junction, which enhances charge separation and transfer; (ii) localized surface plasmon resonance (LSPR), which broadens the light absorption range and facilitates the excitation of active charge carriers; and (iii) suppression of nanoparticle aggregation.The overall photocatalytic activity can be described through the following schematic diagram (Scheme 3). Electron–hole pairs are generated when NP@OFs are illuminated with light. The electrons are used for the reduction process, leading to the formation of H2 from water and value-added chemicals from CO2. Similarly, the oxidation process leads to the formation of oxygen. To have maximum photocatalytic efficiency, electron–hole recombination should be avoided.
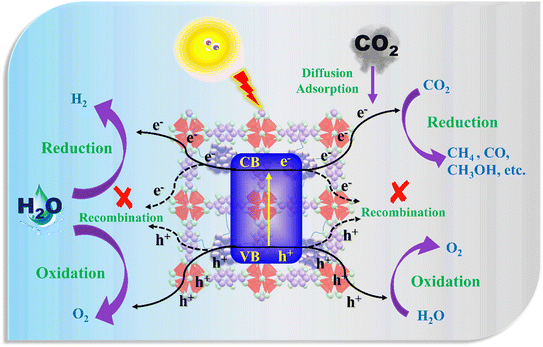 | ||
| Scheme 3 Proposed mechanism of photocatalytic CO2 reduction process and H2 production from water splitting. | ||
H2 production
Given the global green demand for green and sustainable energy, photocatalytic hydrogen evolution reactions (HERs) have gained significant attention. Semiconductor NPs are often used as catalysts for photocatalytic HER. Shi et al. reported that the photocatalytic activity of CdS was greatly enhanced when incorporated within a porphyrin framework (4,4′,4″,4‴-porphine-5,10,15,20-tetrayl)tetrakis(benzoic acid) (TCPP).122 They prepared CdS@Cu-TCPP-Pt nanorod catalysts with an H2 production rate of 20.75 mmol g−1 h−1. The superior photocatalytic performance was attributed to a favorable energy-level alignment of CdS and Cu-TCPP and their interfacial interaction, which promoted efficient separation of photogenerated carriers.Similarly, Li et al. have constructed a core–shell Pt NPs–upconversion NPs@MOF/Au structure, which can absorb a broad spectrum of solar light, as shown in Fig. 8a. In this system, UV light excites the MOF, transferring electrons to Pt, which acts as the active site for hydrogen production. Meanwhile, the SPR effect of Au enhances absorption of visible light, generating hot electrons that transfer first to the LUMO of the MOF and then to Pt NPs. Under NIR irradiation, the energy transfer from Yb3+ causes excitation of Tm3+ and Er3+ ions in UCNPs. The MOF absorbs the upconverted UV emission from Tm3+, generating electron–hole pairs whose electrons migrate to Pt for hydrogen production. Concurrently, the upconverted green emission from Er3+ couples with Au SPR, producing hot electrons that further transfer into the MOF, as shown in Fig. 8b. These dual charge-transfer pathways synergistically improve NIR harvesting and overall photocatalytic activity.
 | ||
| Fig. 8 (a) Schematic of the synthetic process for UCNPs-Pt@MOF/Au. (b) Mechanism involved in photocatalytic H2 production. Adapted from ref. 122, with permission from the American Chemical Society, copyright 2024. (c) Mechanism for H2 generation from FA over the Pd0.8Au0.2/UiO-66-(NH2)2 catalyst. Adapted from ref. 94, with permission from the American Chemical Society, copyright 2021. (d) Hydrogen evolution profiles of EY–MIL-101, EY–Ni@MIL-101, EY–Mo@MIL-101, and EY–NiMo@MIL-101 photocatalysts over time. Adapted from ref. 146, with permission from the American Chemical Society, copyright 2016. (e) Mechanism of the hydrogen evolution reaction (HER) over eosin Y-sensitized NiMo@MIL-101 under visible light irradiation. Adapted from ref. 146, with permission from the American Chemical Society, copyright 2016. | ||
Shi et al. further highlighted that integrating Cu-TCPP with CdS nanorods (NRs) expanded the light absorption range of CdS but also reduced the rate of recombination of electron–hole pairs.122 Subsequent incorporation of these structures with Pt NPs further suppressed e–h recombination and hence boosted the rate of H2 evolution from selective oxidation of benzylamine.
The SPR effect has also been exploited in Ag-based systems. A similar SPR influence on the catalytic efficiency of Ag NP–Ag porphyrin metal–organic matrix (Ag–AgMOM) on the H2 evolution reaction from water was demonstrated by Liu et al.128 The uniqueness of this structure lies in the shared Ag atoms at the Ag NP/AgMOM interface, which enable efficient interfacial charge transfer. Under visible and full-spectrum light irradiation using a full-spectrum 300 W Xe lamp, the Ag–AgMOM exhibited H2 production rates of up to 1025 μmol h−1 g−1 and 3153 μmol h−1 g−1, respectively. Additionally, Ag NPs contribute to stabilizing the otherwise unstable Ag node-based porphyrin MOM by sharing Ag atoms while their SPR effect broadens light harvesting. The presence of a small amount of poly(vinylpyrrolidone) (PVP) at the Ag/AgMOM interface permits efficient charge transfer. All these factors collectively make Ag–AgMOM an efficient catalyst.
Furthermore, the functionalities present in the framework compound play an important role in the catalytic activity by facilitating substrate adsorption followed by chemical transformation. Ding et al. synthesized a PdAu NP-loaded UiO-66-(NH2)2 MOF using a double-solvent (H2O/n-hexane) method followed by NaBH4 reduction.94 The PdAu-loaded UiO-66-(NH2)2 catalyst exhibited superior formic acid dehydrogenation performance compared to PdAu@UiO-66-NH2, with enhanced stability over a wide temperature range. This improvement was attributed to the stronger stabilization effect of the diamine-functionalized UiO-66-(NH2)2 framework, as shown in Fig. 8c.
In general, bimetallic NPs incorporated within frameworks show enhanced activity compared to their single-metal counterparts. For instance, Wen et al. reported that Au@Pd NPs, when integrated with titanium-doped zirconium-based amine-functionalized MOF (UIO-66(TixZr100−x)), act as an excellent catalyst for H2 production from formic acid.143 Zhen et al. revealed that NiMo@MIL-101 outperforms both Mo@MIL-101 and Ni@MIL-101 for H2 generation.146 Photocatalytic activity was tested under 420 nm irradiation in triethanolamine (TEOA) and H2O solution and in the presence of eosin Y (EY), as shown in Fig. 8d.
The mechanism of the H2 evolution process is depicted in Fig. 8e, where electrons generated by EY are transferred to NiMo sites via the highly conductive MIL-101. Hydrogen adsorbed on NiMo is subsequently converted into H2 molecules. Density functional theory (DFT) calculations further confirmed that the hydrogen adsorption free energy (ΔGH) of MoNi4–Hads (458 kJ mol−1) was found to be lower than that of Ni–Hads (537 kJ mol−1), which explains the superior catalytic activity of NiMo catalysts.
Beyond bimetallic effects, the light-trapping efficiency of frameworks can also be tuned to boost the photocatalytic performance. Liu et al. synthesized a series of donor–acceptor network-type COFs that confine PtCo2 nanoclusters.147 Under visible light irradiation, these PtCo2@COFs act as an efficient photocatalyst with a turnover frequency (TOF) of 486 min−1. This effect is ascribed to the strong electronic interactions between the COF and PtCo2 nanoclusters. Furthermore, the D–A type COF significantly improves light-trapping capability by adjusting the electron-acceptor units within the framework, while also facilitating the transfer of photogenerated electrons from the D–A type COF to PtCo2 nanoclusters, which aids in the adsorption and activation of H2O. Additionally, PtCo2@COF demonstrated exceptional stability, attributed to its enhanced resistance to NC aggregation, a result of the nanopore confinement effect within the D–A type COF that prevents nanocluster aggregation.
CO2 reduction
Combining frameworks with encapsulated metals, such as metal NPs and NCs, leverages the unique qualities of each component to create a synergistic effect.21,148 It is worth noting that a series of recent studies have demonstrated that the coexistence of single-site metal active centers and metal nanoclusters in photosensitised MOFs can remarkably activate and convert CO2 through the direction–orientation charge-transfer mechanism and effective photoluminescence, achieving complex catalytic reactions like CO2 carbonylation.21,149 Among them, Cu NPs and NCs are widely recognized as efficient catalysts for CO2 reduction.150–152 Confinement of metal nanostructures within frameworks restricts their agglomeration and regulates their electronic and geometric structures, thereby enhancing their catalytic activities.For example, Zhang et al. developed confined Cu nanoclusters within CuPc (2,3,9,10,16,17,23,24-octahydroxyphthalocyaninato copper) and HHTP (2,3,6,7,10,11-hexahydroxytriphenylene) COFs, as shown in Fig. 9a.153 The linear sweep voltammetry curve in Fig. 9b indicated that the efficiency of the catalyst for the conversion of CO2 to CH4 follows the order CuNC@CuPc > CuNC > CuNC@HTTP. In particular, Cu-NC@CuPc-COF achieved a faradaic efficiency of approximately 74 ± 3% (at ∼1.0 V vs. RHE), along with a peak current density of 538 ± 31 mA cm−2 (at ∼1.2 V vs. RHE). This high efficiency of CuNC@CuPc-COF is attributed to the synergistic effect between Cu NCs and the CuPc-COF. The CO2-to-CH4 conversion proceeds via two reaction pathways, as illustrated in Fig. 9c. DFT calculations confirmed that the free energy barriers for COOH and CO radical formation are significantly reduced, thereby facilitating the overall conversion. Expanding on Cu-based systems, Kung et al. reported a Cu NP-embedded MOF (NU-1000) prepared by solvothermal deposition of Cu2+ ions into NU-1000, followed by electrochemical reduction of Cu2+ ions.154 The resulting Cu NPs exhibited promising electrochemical activity for CO2 reduction. However, the products included CH4, C2H4, and CO with poor selectivity. This limitation was attributed to variations in particle size, degree of agglomeration, and local CO2 concentration. To improve selectivity, bimetallic interfaces are often utilized. It is well known that metal–semiconductor interfaces such as Cu/ZrO2 and Cu/ZnO play critical roles in the hydrogenation of CH3OH from CO2. However, during the catalysis process, particle growth and structural reorganization significantly reduce the activity of these interfaces. An et al. revealed that confinement of Cu/ZnOx NPs in pre-assembled bpy and Zr6(μ3-O)4(μ3-OH)4 sites in UiO-bpy MOFs prevents aggregation of NPs and preserves high activity.93 Other metal NPs, besides Cu, also influence CO2 reduction. For instance, H. Robatjazi et al. established that Al@MIL-53 can be used as a catalyst for the decomposition of CO2.115 Under illumination, CO2 was selectively converted into CO, driven by the plasmonic effect of Al NPs. Likewise, Chen et al. highlighted the influence of Ag NPs on the photoreduction of CO2.155 They showed that Ag@Co-ZIF-9 acted as an effective photocatalyst for CO2-to-CO conversion, with selectivity of the catalyst improving with an increase in Ag content. Similarly, plasmonic Ag nanocube-coated Ren-MOF (Ag⊂Ren-MOF) structures acted as photocatalysts for the conversion of CO2 to CO.156 The catalytic activity of these structures was attributed to the cooperative effect between spatially confined photoactive Re centers and the intensified near-surface electric fields at the surface of Ag nanocubes, as shown in Fig. 9d. Cooperatively enhanced photocatalytic activity in Ren-MOFs required a delicate balance of closeness between photoactive centers. In comparison to the Re3-MOF under visible light, the improved Re3-MOF on silver nanocubes exhibited a sevenfold increase in CO evolution rate (Fig. 9e). Additionally, compared to molecular H2ReTC, the Ag⊂Re3-MOF structure showed long-term stability of up to 48 hours, and after 48 hours, the CO generated from Ag⊂Re3-MOF was nearly twice that of H2ReTC. Further, noble metal-incorporated frameworks also showed tunable selectivity. Zirconium-based PCN-222 MOFs containing metalloporphyrin Cu centers with embedded Au nanoneedles acted as efficient catalysts for CO2 reduction.80 AuNN@PCN-222(Cu) selectively reduced CO2 to ethylene, whereas AuNP@PCN-222(Cu) and PCN-222(Cu) primarily produced CO and H2, respectively. The high ethylene selectivity of AuNN@PCN-222(Cu) was attributed to an energetically favorable path for CO–CHO formation, as illustrated in Fig. 9f.
 | ||
| Fig. 9 (a and b) Schematic of the synthesis process of Cu NC@CuPc and Cu NC@HHTP, and the LSV curve showing CO2-to-CH4 conversion, respectively. (c) Proposed mechanism for the CO2 reduction electrochemically. Adapted from ref. 153, with permission from Elsevier, copyright 2023. (d) Ren-MOF coated on a Ag nanocube for enhanced photocatalytic conversion of CO2. Adapted from ref. 156, with permission from the American Chemical Society, copyright 2017. (e) Photocatalytic CO2-to-CO conversion activity of Ren-MOFs, Ag⊂Re0-MOF, Cu⊂Re2-MOF, and Ag⊂Re3-MOFs with MOF thickness of 16 nm and 33 nm. Adapted from ref. 156, with permission from the American Chemical Society, copyright 2017. (f) The formation energy of key intermediates along with the reaction coordinates for TCPP(Cu) and TCPP(Cu)–Au12. Adapted from ref. 80, with permission from Nature, copyright 2022. (g) Illustration depicting the assembly of Au@Pd@MOF-74, Pt/MOF-74, and Pt/Au@Pd@MOF-74 catalysts, along with their functions. (h) and (i) represent CO and CH4 yield for MOF-74, Pt/MOF-74, Au@Pd@MOF-74, and Pt/Au@Pd@MOF-74, respectively. Adapted from ref. 157, with permission from Elsevier, copyright 2019. | ||
In addition, Han et al. demonstrated that multi-metallic NPs supported on MOF-74 serve as an excellent photocatalyst for CO2 reduction.157 They fabricated a Pt/Au@Pd@MOF-74 catalyst by growing MOF-74 on pre-formed Au@Pd NPs and subsequently adding Pt NPs, as shown in Fig. 9g. In photocatalytic conversion of CO2, Au@Pd@MOF-74 selectively generated CO from CO2, whereas Pt/Au@Pd@MOF-74 favored CH4 with 84% selectivity, as seen in Fig. 9h and i. Since CO2-to-CO conversion is a two-electron process, while CO2 to CH4 requires four electrons, Pt's ability to absorb and transfer more electrons explains its effectiveness in promoting CH4 formation.
Su et al. reported that the addition of ZIF-8 precursor to PVP-capped Au@AgPt nanorattles (NRTs) leads to the formation of Au@AgPt@ZIF-8 nanostructures.119 These nanostructures served as highly efficient catalysts for the conversion of CO2 to CH4. Building on structural factors, Yang et al. revealed that internal strain within MOFs can enhance their electrocatalytic CO2 reduction efficiency and selectivity.135 Specifically, growth of ZnO NPs inside MOFs via the VPI method created 2.8% additional internal strain, which improved CO2 to CO reduction efficiency by a factor of four compared to unstrained MOFs. Semiconductor NP embedded in OFs also acts as an efficient catalyst for CO2 reduction. Yao et al. have shown that BiVO4 nanocluster-embedded MIL act as an excellent catalyst for the CO2 to urea conversion process in a mixed O2/air/N2 atmosphere.158 Reaction conditions also play a crucial role. CO2 reduction under acidic conditions improves the carbon utilization rate compared to alkaline conditions. However, under acidic conditions, the hydrogen evolution reaction (HER) competes strongly with CO2 reduction. To address this limitation, Wan et al. established that conductive 2D NiPc-based MCOF (NiPc-MTAA) frameworks, coupled with metal tetraaza[14]-annulene linkages, act as efficient electrocatalysts for CO2 reduction in acidic electrolytes. In comparison to 2D NiPc-based MOFs (NiPc-MN4), these MCOFs demonstrated superior electrical conductivity and stability, attributed to full in-plane π-delocalization.159 Moreover, the NiTAA node optimized the electron distribution of 3d orbitals by shifting the d-band center closer to the Fermi level, thereby facilitating easier activation of CO2.
6. Conclusion and outlook
The encapsulation of nanoparticles within porous frameworks, such as MOFs and COFs, has emerged as a rapidly growing and dynamic area of research. These hybrid materials combine the unique physicochemical properties of nanoparticles with the structural tunability, high surface area, and functional versatility of the host frameworks. This synergy opens new possibilities for applications in catalysis, sensing, energy storage, drug delivery, and environmental remediation. In this review, we present a comprehensive overview of the most significant recent advancements in the design, synthesis, and application of NP-encapsulated frameworks, highlighting key strategies, challenges, and future directions in the field. NPs can be grown on pre-formed MOFs (in situ approach), or MOFs can be grown on pre-formed NPs (post-synthetic approach). This review provides a critical analysis of the underlying factors that influence the encapsulation process, with emphasis on extended stability and on-demand functionality for catalysis. Importantly, frameworks and NPs do not act as individual entities in NP@framework structures but instead function synergistically. Frameworks restrict NP aggregation, thereby enhancing the stability of NPs, which in turn improves their catalytic performance under harsh conditions. Conversely, NPs also provide stability to organic frameworks by imposing restrictions on their flexibility. In post-synthesis approaches, the nucleation of organic frameworks on nanoparticle surfaces often leads to the formation of defect-controlled, uniform, and stable structures. Both frameworks and NPs mutually influence the electronic environment of each other, which strongly impacts catalytic performance. In addition, other factors such as the size and morphology of NPs, their electronic environment, surface-active agents, framework dimensions, and functional groups also influence catalytic activity. A detailed analysis of the factors is provided in this review. The advantages of these structures, mentioned below, highlight future possibilities that need exploration.(i) The advantages of NPs@frameworks structures lie in the ability to generate a vast library of materials owing to the diverse availability of both NPs and frameworks. Since significant property changes can be achieved with slight structural modifications, there is a strong potential to expand the existing library. Recent developments in complex heterostructures further broaden this scope. Dimensionality also impacts catalytic efficiency. Yang et al. have demonstrated that a reduction in the thickness of 3D MOFs enhances their catalytic efficiency for electrochemical CO2 reduction.160 They showed that compared to 3D MOFs, monolayer MOFs with a thickness of 1.8 nm exhibited a higher faradaic efficiency for CO production (93% compared to 51% for 3D MOFs). Crystal engineering of 2D framework nanosheets has been shown to significantly influence photocatalytic performance. Hence, these structures represent promising future catalysts for diverse reactions. Extensive studies are still required to develop new catalyst systems. For instance, yolk–shell and mull architectures, as well as NP incorporation into complex structures such as MOF@MOF, MOF@COF, and MOF/COF composites, warrant further exploration. A recent study by Chu et al. showed that control over the internal electric field in MOF/COF heterojunctions can be achieved by engineering crystal facets.161 Similarly, the influence of the crystal facet of NPs on catalytic processes is well evidenced. However, strategies to control the crystal facets of both NPs and organic frameworks as well as their interfacial connections need to be developed to achieve improved catalytic efficiencies.
(ii) Frameworks can effectively stabilize small-sized NPs. However, since catalytic reactions are often carried out under harsh conditions, frameworks must be designed with improved robustness. In addition, scalable synthesis strategies are essential to enable the commercial-scale production of these materials. A seed-mediated method developed by Han et al. operates at lower temperatures and facilitates the production of highly crystalline MOFs, which can be readily scaled up.162
(iii) As discussed earlier, frameworks act as transporters of charge carriers during photocatalysis. However, their conductivity depends on several factors, including the functionality of metal centers and bridging ligands, the nature of the framework, and its dimensionality. Therefore, detailed investigations are needed to clarify how these parameters influence integrated NP@framework systems.
(iv) The strong synergic effects between NPs and frameworks are well recognized. However, the extent and precise nature of these interactions as well as their influence on catalytic behavior remain unclear. In-depth mechanistic studies using in situ techniques are required to unravel the underlying principles.
(v) These materials often face intrinsic limitations in their structural tunability, which restricts adaptability to evolving application requirements. Such constraints can hinder the optimization of their physicochemical properties, ultimately limiting their overall performance.
(vi) Since the properties of NPs are strongly influenced by their size and shape, further research is required to successfully incorporate NPs with different morphologies into various frameworks. Uniformly distributed NPs show enhanced catalytic efficiencies compared to non-uniformly distributed ones. In this study, we analyse the factors that influence the spatial distribution of NPs inside framework structures. However, further studies are required to fully understand the synergy between these factors.
(vii) Defect engineering plays a critical role in catalysis.163 For example, Fei et al. designed a new type of defect-rich thulium(III) metal–organic framework that acted as a superior CO2 fixation catalyst.164 However, the low integration of NPs inside OFs influences the defect of OFs, which is still unexplored and needs research. Photo responsivity and multilevel electron transfer of cluster-included framework material plays a vital role in the enhancement of the lifetime of electrons for photocatalytic applications.
In summary, the lack of precise control over morphology, pore size, surface functionality, and framework flexibility can impede effective interactions with guest molecules or nanoparticles, thereby reducing efficiency in targeted applications. Overcoming these challenges is essential to unlock the full potential of framework-stabilized nanostructures and to develop versatile, high-performing materials tailored to specific technological needs. Considerable research efforts have already demonstrated the remarkable catalytic efficiencies of NP@frameworks across diverse reactions. Nevertheless, extended and focused investigations are required, particularly on synthesis strategies, stability under harsh conditions, mechanistic insights, and semiconductor-based systems, to advance these materials into practical applications. With continued progress, NP@frameworks hold strong promise as the “materials of a sustainable society”.
Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software, or code have been included, and no new data were generated or analysed as part of this review.Acknowledgements
RS gratefully acknowledges the BRNS-funded project (Sanction No. 58/14/04/2025-BRNS) supported by the Department of Atomic Energy (DAE), Government of India. PS acknowledges extra mural research funding under MRIP 2024-Chemistry (24EM/CH/27) from the Department of Higher Education, Odisha.References
- Z. Qin, Nanomaterials, 2023, 13, 1980 CrossRef CAS PubMed.
- S. Goswami, H. Noh, L. R. Redfern, K.-i. Otake, C.-W. Kung, Y. Cui, K. W. Chapman, O. K. Farha and J. T. Hupp, Chem. Mater., 2019, 31, 1485–1490 CrossRef CAS.
- L. Liu and A. Corma, Chem. Rev., 2018, 118, 4981–5079 CrossRef CAS PubMed.
- A. Acharya, T. B. Mete, N. Kumari, Y. Yoon, H. Jeong, T. Jang, B. Song, H. C. Choi, J. W. Han, Y. Pang, Y. Yun, A. Kumar and I. S. Lee, Nat. Commun., 2023, 14, 7667 Search PubMed.
- A. Kumar and L. Kumar, Res. Chem. Intermed., 2024, 50, 195–217 CrossRef CAS.
- P. Sahu, J. Shimpi and B. Prasad, in Molecular Materials, CRC Press, 2017, pp. 189–212 Search PubMed.
- R. Javed, M. Zia, S. Naz, S. O. Aisida, N. u. Ain and Q. Ao, J. Nanobiotechnol., 2020, 18, 172 CrossRef PubMed.
- Q. Yu, J. Pan, J. Li, C. Su, Y. Huang, S. Bi, J. Jiang and N. Chen, J. Mater. Chem. C, 2022, 10, 13143–13156 RSC.
- E. Eom, M. Song, J.-C. Kim, D.-i. Kwon, D. N. Rainer, K. Gołąbek, S. C. Nam, R. Ryoo, M. Mazur and C. Jo, JACS Au, 2022, 2, 2327–2338 CrossRef CAS PubMed.
- A. M. Jauhar, Z. Ma, M. Xiao, G. Jiang, S. Sy, S. Li, A. Yu and Z. Chen, J. Power Sources, 2020, 473, 228607 CrossRef CAS.
- V. F. Yusuf, N. I. Malek and S. K. Kailasa, ACS Omega, 2022, 7, 44507–44531 CrossRef CAS PubMed.
- A. E. Baumann, D. A. Burns, B. Liu and V. S. Thoi, Commun. Chem., 2019, 2, 86 CrossRef.
- S. M. Moosavi, A. Nandy, K. M. Jablonka, D. Ongari, J. P. Janet, P. G. Boyd, Y. Lee, B. Smit and H. J. Kulik, Nat. Commun., 2020, 11, 4068 CrossRef CAS PubMed.
- H. Furukawa, K. E. Cordova, M. O'Keeffe and O. M. Yaghi, Science, 2013, 341, 1230444 CrossRef PubMed.
- D. Li, A. Yadav, H. Zhou, K. Roy, P. Thanasekaran and C. Lee, Glob. Chall., 2024, 8, 2300244 CrossRef PubMed.
- I. Abánades Lázaro, X. Chen, M. Ding, A. Eskandari, D. Fairen-Jimenez, M. Giménez-Marqués, R. Gref, W. Lin, T. Luo and R. S. Forgan, Nat. Rev. Methods Primers, 2024, 4, 42 CrossRef.
- A. M. Wright, M. T. Kapelewski, S. Marx, O. K. Farha and W. Morris, Nat. Mater., 2025, 24, 178–187 CrossRef CAS PubMed.
- Y. Qian and H.-L. Jiang, Acc. Chem. Res., 2024, 57, 1214–1226 CrossRef CAS PubMed.
- Y. Sakamaki, M. Tsuji, Z. Heidrick, O. Watson, J. Durchman, C. Salmon, S. R. Burgin and H. Beyzavi, J. Chem. Educ., 2020, 97, 1109–1116 CrossRef CAS PubMed.
- S. Bommakanti, A. Puthukkudi, M. Samal, P. Sahu and B. P. Biswal, Cryst. Growth Des., 2023, 23, 6172–6200 CrossRef CAS.
- P. Sahu, S. Palei and U. Manju, in Electrochemical Applications of Metal-Organic Frameworks, ed. S. Dave, R. Sahu and B. C. Tripathy, Elsevier, 2022, pp. 95–116, DOI:10.1016/B978-0-323-90784-2.00003-4.
- P. Z. Moghadam, Y. G. Chung and R. Q. Snurr, Nat. Energy, 2024, 9, 121–133 CrossRef CAS.
- F. Zheng, S. Cao, Z. Yang, Y. Sun, Z. Shen, Y. Wang and H. Pang, Energy Fuels, 2024, 38, 11494–11520 CrossRef CAS.
- G. Li, S. Zhao, Y. Zhang and Z. Tang, Adv. Mater., 2018, 30, 1800702 CrossRef PubMed.
- M. Xu, C. Lai, X. Liu, B. Li, M. Zhang, F. Xu, S. Liu, L. Li, L. Qin, H. Yi and Y. Fu, J. Mater. Chem. A, 2021, 9, 24148–24174 RSC.
- M. Duan, L. Jiang, G. Zeng, D. Wang, W. Tang, J. Liang, H. Wang, D. He, Z. Liu and L. Tang, Appl. Mater. Today, 2020, 19, 100564 CrossRef.
- W. Xiang, Y. Zhang, H. Lin and C.-j. Liu, Molecules, 2017, 22, 2103 CrossRef PubMed.
- T. Zhang, T. Li, M. Gao, W. Lu, Z. Chen, W. L. Ong, A. S. W. Wong, L. Yang, S. Kawi and G. W. Ho, Adv. Energy Mater., 2024, 14, 2400388 CrossRef CAS.
- M. Chalermnon, S. R. Thomas, J. M. Chin and M. R. Reithofer, Inorg. Chem. Front., 2025, 12, 6435–6459 RSC.
- X. L. Ma, W. X. Shi, S. Guo, Q. P. Zhao, W. Lin, T. B. Lu and Z. M. Zhang, Adv. Mater., 2025, 37, 2506133 CrossRef CAS PubMed.
- X. Yang and Q. Xu, Trends Chem., 2020, 2, 214–226 CrossRef CAS.
- F. Ke, J. Yuan, C. Zhang, S. Ye, K. Ramachandraiah and H. Pang, Coord. Chem. Rev., 2024, 518, 216116 CrossRef CAS.
- T. Zorlu, I. B. Becerril-Castro, A. Sousa-Castillo, B. Puértolas, L. V. Besteiro, Z. Wang, A. Govorov, M. A. Correa-Duarte and R. A. Alvarez-Puebla, Adv. Funct. Mater., 2024, 34, 2410352 CrossRef CAS.
- R. Abazari, N. Ghorbani, J. Shariati, R. S. Varma and J. Qian, Inorg. Chem., 2024, 63, 12667–12680 CrossRef CAS PubMed.
- S. Sanati, R. S. Varma, M. Liu and R. Abazari, Energy Environ. Sci., 2025, 18(16), 7733–7755 RSC.
- L. Mao and J. Qian, Small, 2024, 20, 2308732 CrossRef CAS PubMed.
- Y. Deng, Y. Wang, X. Xiao, B. J. Saucedo, Z. Zhu, M. Xie, X. Xu, K. Yao, Y. Zhai and Z. Zhang, Small, 2022, 18, 2202928 CrossRef CAS PubMed.
- J. Q. Ye, S. Y. Xu, Q. Liang, Y. Z. Dai and M. Y. He, Chem. – Asian J., 2024, 19, e202400161 CrossRef CAS PubMed.
- G. Yuan, L. Tan, P. Wang, Y. Wang, C. Wang, H. Yan and Y.-Y. Wang, Cryst. Growth Des., 2021, 22, 893–908 CrossRef.
- R. Freund, O. Zaremba, G. Arnauts, R. Ameloot, G. Skorupskii, M. Dincă, A. Bavykina, J. Gascon, A. Ejsmont and J. Goscianska, Angew. Chem., Int. Ed., 2021, 60, 23975–24001 CrossRef CAS PubMed.
- J. Hu, Z. Huang and Y. Liu, Angew. Chem., Int. Ed., 2023, 62, e202306999 CrossRef CAS PubMed.
- L. L. Zhou, Q. Guan and Y. B. Dong, Angew. Chem., 2024, 136, e202314763 CrossRef.
- J. Park, H. Kim, Y. Kang, Y. Lim and J. Kim, JACS Au, 2024, 4, 3727–3743 CrossRef CAS PubMed.
- K. Sun, Y. Qian and H. L. Jiang, Angew. Chem., 2023, 135, e202217565 CrossRef.
- A. Dhakshinamoorthy, Z. Li, S. Yang and H. Garcia, Chem. Soc. Rev., 2024, 53, 3002–3035 RSC.
- H. Wang, H. Wang, Z. Wang, L. Tang, G. Zeng, P. Xu, M. Chen, T. Xiong, C. Zhou and X. Li, Chem. Soc. Rev., 2020, 49, 4135–4165 RSC.
- Z. Wang, H. Fei and Y. n. Wu, ChemSusChem, 2024, 17, e202400504 CrossRef CAS PubMed.
- T. Li, P. Wang, M. He, T. Zhang, C. Yang and Z. Li, Coord. Chem. Rev., 2024, 521, 216179 CrossRef CAS.
- M. Li, H. Zhang, C. Li, F. Lang, S.-W. Yao, J. Pang and X.-H. Bu, Precis. Chem., 2025, 3, 424–450 CrossRef CAS PubMed.
- Y. An, T. Cai, W. Jiang, T. Lei and H. Pang, Green Chem., 2025, 27, 10478–10509 RSC.
- Y. Ou, Y. Zhang, W. Luo, Y. Wu and Y. Wang, Macromol. Rapid Commun., 2025, 46, 2401149 CrossRef CAS PubMed.
- P. Asselin and P. D. Harvey, ACS Appl. Nano Mater., 2022, 5, 6055–6082 CrossRef CAS.
- H. L. Nguyen, Sol. RRL, 2021, 5, 2100198 CrossRef CAS.
- Y. Yang, Z. Sun, Y. Wu, Z. Liang, F. Li, M. Zhu and J. Liu, Small, 2024, 20, 2401457 CrossRef CAS PubMed.
- Q. Zhang, S. Jiang, T. Lv, Y. Peng and H. Pang, Adv. Mater., 2023, 35, 2305532 CrossRef CAS PubMed.
- T. Shaikh, S. Pise, R. Bhosale, M. Vadiyar, K.-W. Nam and S. Kolekar, Energy Fuels, 2025, 39, 2396–2421 CrossRef CAS.
- L. Huang, M. Du, D. Sun, M. He, Z. Liu, R. Wu, Y. Jiang, L. Qi, J. Wang and C. Zhu, Small, 2024, 20, 2306508 CrossRef CAS PubMed.
- S. A. Fatah and K. M. Omer, ACS Appl. Mater. Interfaces, 2025, 17, 11578–11594 CrossRef CAS PubMed.
- S. S. Mohammed Ameen, A. Bedair, M. Hamed, F. R. Mansour and K. M. Omer, ACS Appl. Mater. Interfaces, 2024, 17, 110–129 CrossRef PubMed.
- P. Hazra, S. Vadnere, S. Mishra, S. Halder, S. Mandal and P. Ghosh, ACS Appl. Mater. Interfaces, 2023, 15, 52065–52082 CAS.
- A. Schlachter, P. Asselin and P. D. Harvey, ACS Appl. Mater. Interfaces, 2021, 13, 26651–26672 CrossRef CAS PubMed.
- C. Yu, L. Xu, F. Zhao, L. Kong, Y. Chen, L. Li, Z. Zhu and L. Jiang, ACS Mater. Lett., 2024, 6, 5454–5478 CrossRef CAS.
- J. Li, X. Zhou, J. Wang and X. Li, Ind. Eng. Chem. Res., 2019, 58, 15394–15406 CrossRef CAS.
- C. Guo, F. Duan, S. Zhang, L. He, M. Wang, J. Chen, J. Zhang, Q. Jia, Z. Zhang and M. Du, J. Mater. Chem. A, 2022, 10, 475–507 RSC.
- C. Zhang, Y. Wu, D. Li and H.-L. Jiang, Chem. Sci., 2025, 16, 13149–13172 RSC.
- X. C. Qi, F. Lang, C. Li, M. W. Liu, Y. F. Wang and J. Pang, ChemPlusChem, 2024, 89, e202400158 Search PubMed.
- C. Gao, F. Lyu and Y. Yin, Chem. Rev., 2020, 121, 834–881 CrossRef PubMed.
- C. Liu, W. Wang, B. Liu, J. Qiao, L. Lv, X. Gao, X. Zhang, D. Xu, W. Liu and J. Liu, Catalysts, 2019, 9, 658 Search PubMed.
- S. Kumar, B. Z. Dholakiya and R. Jangir, ACS Appl. Mater. Interfaces, 2023, 16, 1553–1563 CrossRef PubMed.
- G. Ye, G. Shi, R. Huang, X. Liu, J. Zhou, S. Wen and Q. Zhang, Inorg. Chem., 2025, 64(15), 7806–7817 CrossRef CAS PubMed.
- S. E. Alavi, S. F. Alavi, M. Koohi, A. Raza and H. Ebrahimi Shahmabadi, J. Pharm. Invest., 2024, 54, 751–783 CrossRef.
- L. Li, H. Zhao, J. Wang and R. Wang, ACS Nano, 2014, 8, 5352–5364 Search PubMed.
- M. Bhadra, H. S. Sasmal, A. Basu, S. P. Midya, S. Kandambeth, P. Pachfule, E. Balaraman and R. Banerjee, ACS Appl. Mater. Interfaces, 2017, 9, 13785–13792 CrossRef CAS PubMed.
- S. B. Kalidindi, H. Oh, M. Hirscher, D. Esken, C. Wiktor, S. Turner, G. Van Tendeloo and R. A. Fischer, Chem. – Eur. J., 2012, 18, 10848–10856 CrossRef CAS PubMed.
- Y. Chen, Q. Zhou and J. Zheng, ACS Sustainable Chem. Eng., 2022, 10, 1961–1971 CrossRef CAS.
- F. Pan, C. Tong, N. Wang, Y. Wang, D. Pan and R. Zhu, ACS Sustainable Chem. Eng., 2022, 10, 16027–16036 CrossRef CAS.
- T. Pan, I. E. Khalil, Z. Xu, H. Li, X. Zhang, G. Xiao, W. Zhang, Y. Shen and F. Huo, Nano Res., 2022, 15, 1178–1182 CrossRef CAS.
- H.-L. Jiang, T. Akita, T. Ishida, M. Haruta and Q. Xu, J. Am. Chem. Soc., 2011, 133, 1304–1306 Search PubMed.
- Y.-Z. Chen, Q. Xu, S.-H. Yu and H.-L. Jiang, Small, 2015, 11, 71–76 CrossRef CAS PubMed.
- X. Xie, X. Zhang, M. Xie, L. Xiong, H. Sun, Y. Lu, Q. Mu, M. H. Rummeli, J. Xu, S. Li, J. Zhong, Z. Deng, B. Ma, T. Cheng, W. A. Goddard and Y. Peng, Nat. Commun., 2022, 13, 63 CrossRef CAS PubMed.
- J. Hermannsdörfer, M. Friedrich, N. Miyajima, R. Q. Albuquerque, S. Kümmel and R. Kempe, Angew. Chem., Int. Ed., 2012, 51, 11473–11477 CrossRef PubMed.
- F. Chen, K. Shen, J. Chen, X. Yang, J. Cui and Y. Li, ACS Cent. Sci., 2019, 5, 176–185 CrossRef CAS PubMed.
- X.-J. Bai, X. Zhai, L.-Y. Zhang, Y. Fu and W. Qi, Matter, 2021, 4, 2919–2935 CrossRef CAS.
- A. Aijaz, A. Karkamkar, Y. J. Choi, N. Tsumori, E. Rönnebro, T. Autrey, H. Shioyama and Q. Xu, J. Am. Chem. Soc., 2012, 134, 13926–13929 CrossRef CAS PubMed.
- Q.-L. Zhu, J. Li and Q. Xu, J. Am. Chem. Soc., 2013, 135, 10210–10213 CrossRef CAS PubMed.
- Y.-Z. Chen, Y.-X. Zhou, H. Wang, J. Lu, T. Uchida, Q. Xu, S.-H. Yu and H.-L. Jiang, ACS Catal., 2015, 5, 2062–2069 CrossRef CAS.
- R. Tao, K. Kang, X. Li, R. Li, R. Huang, Y. Jin, L. Qiu and W. Zhang, Inorg. Chem., 2021, 60, 12517–12525 CrossRef CAS PubMed.
- M. Guo, Q. Meng, W. Chen, Z. Meng, M. L. Gao, Q. Li, X. Duan and H. L. Jiang, Angew. Chem., Int. Ed., 2023, 62, e202305212 CrossRef CAS PubMed.
- X. Lan, C. Du, L. Cao, T. She, Y. Li and G. Bai, ACS Appl. Mater. Interfaces, 2018, 10, 38953–38962 CrossRef CAS PubMed.
- S. Lu, Y. Hu, S. Wan, R. McCaffrey, Y. Jin, H. Gu and W. Zhang, J. Am. Chem. Soc., 2017, 139, 17082–17088 CrossRef CAS PubMed.
- B. Mishra, S. Biswal and B. P. Tripathi, ACS Appl. Nano Mater., 2024, 7, 5317–5328 CrossRef CAS.
- L. Chen, H. Chen, R. Luque and Y. Li, Chem. Sci., 2014, 5, 3708–3714 RSC.
- B. An, J. Zhang, K. Cheng, P. Ji, C. Wang and W. Lin, J. Am. Chem. Soc., 2017, 139, 3834–3840 CrossRef CAS PubMed.
- R.-D. Ding, Y.-L. Li, F. Leng, M.-J. Jia, J.-H. Yu, X.-F. Hao and J.-Q. Xu, ACS Appl. Nano Mater., 2021, 4, 9790–9798 CrossRef CAS.
- L. Chen, B. Huang, X. Qiu, X. Wang, R. Luque and Y. Li, Chem. Sci., 2016, 7, 228–233 RSC.
- W. Qin, W. Cao, H. Liu, Z. Li and Y. Li, RSC Adv., 2014, 4, 2414–2420 RSC.
- Y. Yang, H. Noh, Q. Ma, R. Wang, Z. Chen, N. M. Schweitzer, J. Liu, K. W. Chapman and J. T. Hupp, ACS Appl. Mater. Interfaces, 2021, 13, 36232–36239 CrossRef CAS PubMed.
- W.-G. Cui, Y.-T. Li, H. Zhang, Z.-C. Wei, B.-H. Gao, J.-J. Dai and T.-L. Hu, Appl. Catal., A, 2020, 278, 119262 CrossRef CAS.
- Y. Shen, L.-W. Bao, F.-Z. Sun and T.-L. Hu, Mater. Chem. Front., 2019, 3, 2363–2373 RSC.
- L. Wang, K. Zhang, J. Li, X. Shen, N. Yan, H.-Z. Zhao and Z. Qu, Environ. Sci. Technol., 2022, 56, 16240–16248 CrossRef CAS PubMed.
- Y. Fang, Z. Xiao, J. Li, C. Lollar, L. Liu, X. Lian, S. Yuan, S. Banerjee, P. Zhang and H.-C. Zhou, Angew. Chem., Int. Ed., 2018, 57, 5283–5287 CrossRef CAS PubMed.
- M. Zhong, S. Zhang, A. Dong, Z. Sui, L. Feng and Q. Chen, J. Mater. Sci., 2020, 55, 10388–10398 CrossRef CAS.
- L. Chen, X. Chen, H. Liu, C. Bai and Y. Li, J. Mater. Chem. A, 2015, 3, 15259–15264 RSC.
- M. S. El-Shall, V. Abdelsayed, A. E. R. S. Khder, H. M. A. Hassan, H. M. El-Kaderi and T. E. Reich, J. Mater. Chem., 2009, 19, 7625–7631 RSC.
- H. Duan, Y. Zeng, X. Yao, P. Xing, J. Liu and Y. Zhao, Chem. Mater., 2017, 29, 3671–3677 CrossRef CAS.
- B. Zhang, Y. Pei, R. V. Maligal-Ganesh, X. Li, A. Cruz, R. J. Spurling, M. Chen, J. Yu, X. Wu and W. Huang, Ind. Eng. Chem. Res., 2020, 59, 17495–17501 CrossRef CAS.
- M. Zhang, Q. Shang, Y. Wan, Q. Cheng, G. Liao and Z. Pan, Appl. Catal., B, 2019, 241, 149–158 CrossRef CAS.
- L. Zhou, Y. An, J. Ma, G. Hao, Z. Li, J. Chen and L.-Y. Chou, Chem. Sci., 2023, 14, 13126–13133 RSC.
- Q. Yang, W. Liu, B. Wang, W. Zhang, X. Zeng, C. Zhang, Y. Qin, X. Sun, T. Wu, J. Liu, F. Huo and J. Lu, Nat. Commun., 2017, 8, 14429 CrossRef CAS PubMed.
- Q. Yang, W. Liu, B. Wang, W. Zhang, X. Zeng, C. Zhang, Y. Qin, X. Sun, T. Wu and J. Liu, Nat. Commun., 2017, 8, 14429 CrossRef CAS PubMed.
- L. Chen, W. Wang, J. Tian, F. Bu, T. Zhao, M. Liu, R. Lin, F. Zhang, M. Lee, D. Zhao and X. Li, Nat. Commun., 2021, 12, 4556 CrossRef CAS PubMed.
- L.-N. Chen, H.-Q. Li, M.-W. Yan, C.-F. Yuan, W.-W. Zhan, Y.-Q. Jiang, Z.-X. Xie, Q. Kuang and L.-S. Zheng, Small, 2017, 13, 1700683 CrossRef PubMed.
- B. Wang, W. Liu, W. Zhang and J. Liu, Nano Res., 2017, 10, 3826–3835 CrossRef CAS.
- J. W. M. Osterrieth, D. Wright, H. Noh, C.-W. Kung, D. Vulpe, A. Li, J. E. Park, R. P. Van Duyne, P. Z. Moghadam, J. J. Baumberg, O. K. Farha and D. Fairen-Jimenez, J. Am. Chem. Soc., 2019, 141, 3893–3900 CrossRef CAS PubMed.
- H. Robatjazi, D. Weinberg, D. F. Swearer, C. Jacobson, M. Zhang, S. Tian, L. Zhou, P. Nordlander and N. J. Halas, Sci. Adv., 2019, 5, eaav5340 CrossRef CAS PubMed.
- K. Nakatsuka, T. Yoshii, Y. Kuwahara, K. Mori and H. Yamashita, Chem. – Eur. J., 2018, 24, 898–905 CrossRef CAS PubMed.
- Z. Li, R. Yu, J. Huang, Y. Shi, D. Zhang, X. Zhong, D. Wang, Y. Wu and Y. Li, Nat. Commun., 2015, 6, 8248 CrossRef CAS PubMed.
- J. Wang, H. Xu, C. Ao, X. Pan, X. Luo, S. Wei, Z. Li, L. Zhang, Z.-l. Xu and Y. Li, iScience, 2020, 23, 101233 CrossRef CAS PubMed.
- Y. Su, H. Xu, J. Wang, X. Luo, Z.-l. Xu, K. Wang and W. Wang, Nano Res., 2019, 12, 625–630 CrossRef CAS.
- T. Inomata and K. Konishi, Chem. Commun., 2003,(11), 1282–1283 RSC.
- Y. Long, S. Song, J. Li, L. Wu, Q. Wang, Y. Liu, R. Jin and H. Zhang, ACS Catal., 2018, 8, 8506–8512 CrossRef CAS.
- M. Shi, D. Luo, R. Liu, J. Wei, S. Guo, Z. Lu and Y. Ni, Inorg. Chem., 2024, 63, 18233–18241 CrossRef CAS PubMed.
- H. He, L. Li, Y. Liu, M. Kassymova, D. Li, L. Zhang and H.-L. Jiang, Nano Res., 2021, 14, 444–449 CrossRef CAS.
- L. Chen, H. Chen and Y. Li, Chem. Commun., 2014, 50, 14752–14755 RSC.
- K. Wang, W. Zhao, Q. Zhang, H. Li and F. Zhang, ACS Omega, 2020, 5, 16183–16188 CrossRef CAS PubMed.
- H. Gamraoui, A. Khojastehnezhad, M. Bélanger-Bouliga, A. Nazemi and M. Siaj, ACS Appl. Nano Mater., 2024, 7, 22570–22580 CrossRef CAS.
- L. Chen, X. Chen, H. Liu and Y. Li, Small, 2015, 11, 2642–2648 CrossRef CAS PubMed.
- Y. Liu, C.-H. Liu, T. Debnath, Y. Wang, D. Pohl, L. V. Besteiro, D. M. Meira, S. Huang, F. Yang, B. Rellinghaus, M. Chaker, D. F. Perepichka and D. Ma, Nat. Commun., 2023, 14, 541 CrossRef CAS PubMed.
- Y. Jiang, X. Zhang, X. Dai, W. Zhang, Q. Sheng, H. Zhuo, Y. Xiao and H. Wang, Nano Res., 2017, 10, 876–889 CrossRef CAS.
- H. Alamgholiloo, S. Rostamnia, A. Hassankhani, X. Liu, A. Eftekhari, A. Hasanzadeh, K. Zhang, H. Karimi-Maleh, S. Khaksar and R. S. Varma, J. Colloid Interface Sci., 2020, 567, 126–135 CrossRef CAS PubMed.
- S. Muhamed, R. K. Aparna, A. Karmakar, S. Kundu and S. Mandal, Nanoscale, 2022, 14, 17345–17353 RSC.
- H. Issa Hamoud, P. Damacet, D. Fan, N. Assaad, O. I. Lebedev, A. Krystianiak, A. Gouda, O. Heintz, M. Daturi and G. Maurin, J. Am. Chem. Soc., 2022, 144, 16433–16446 CrossRef CAS PubMed.
- H. Wang, X. Liu, Y. Zhao, Z. Sun, Y. Lin, T. Yao and H.-L. Jiang, Natl. Sci. Rev., 2024, 11, nwae252 CrossRef CAS PubMed.
- N. Tsumori, L. Chen, Q. Wang, Q.-L. Zhu, M. Kitta and Q. Xu, Chem, 2018, 4, 845–856 CAS.
- F. Yang, W. Hu, C. Yang, M. Patrick, A. L. Cooksy, J. Zhang, J. A. Aguiar, C. Fang, Y. Zhou, Y. S. Meng, J. Huang and J. Gu, Angew. Chem., Int. Ed., 2020, 59, 4572–4580 CrossRef CAS PubMed.
- C. Wang, K. E. deKrafft and W. Lin, J. Am. Chem. Soc., 2012, 134, 7211–7214 CrossRef CAS PubMed.
- M. R. Mian, L. R. Redfern, S. M. Pratik, D. Ray, J. Liu, K. B. Idrees, T. Islamoglu, L. Gagliardi and O. K. Farha, Chem. Mater., 2020, 32, 3078–3086 CrossRef CAS.
- C. Zhan, M. Moskovits and Z.-Q. Tian, Matter, 2020, 3, 42–56 CrossRef.
- S. Ezendam, M. Herran, L. Nan, C. Gruber, Y. Kang, F. Gröbmeyer, R. Lin, J. Gargiulo, A. Sousa-Castillo and E. Cortés, ACS Energy Lett., 2022, 7, 778–815 CrossRef CAS PubMed.
- C. S. L. Koh, H. Y. F. Sim, S. X. Leong, S. K. Boong, C. Chong and X. Y. Ling, ACS Mater. Lett., 2021, 3, 557–573 CrossRef CAS.
- A. Amirjani, N. B. Amlashi and Z. S. Ahmadiani, ACS Appl. Nano Mater., 2023, 6, 9085–9123 CrossRef CAS.
- Y. Qin, M. Hao, J. Wang, R. Yuan and Z. Li, ACS Appl. Mater. Interfaces, 2022, 14, 56930–56937 CrossRef CAS PubMed.
- M. Wen, K. Mori, Y. Kuwahara and H. Yamashita, ACS Energy Lett., 2017, 2, 1–7 CrossRef CAS.
- Y. Shen, T. Pan, P. Wu, J. Huang, H. Li, I. E. Khalil, S. Li, B. Zheng, J. Wu, Q. Wang, W. Zhang, W. D. Wei and F. Huo, CCS Chem., 2021, 3, 1607–1614 CrossRef CAS.
- J.-D. Xiao, Q. Shang, Y. Xiong, Q. Zhang, Y. Luo, S.-H. Yu and H.-L. Jiang, Angew. Chem., Int. Ed., 2016, 55, 9389–9393 CrossRef CAS PubMed.
- W. Zhen, H. Gao, B. Tian, J. Ma and G. Lu, ACS Appl. Mater. Interfaces, 2016, 8, 10808–10819 CrossRef CAS PubMed.
- Y. Liu, Y. Shi, H. Wang and S. Zhang, Nano Res., 2024, 17, 5835–5844 CrossRef CAS.
- F. Kong and W. Chen, Nanomaterials, 2024, 14, 1340 CrossRef CAS PubMed.
- S. Fu, S. Yao, S. Guo, G.-C. Guo, W. Yuan, T.-B. Lu and Z.-M. Zhang, J. Am. Chem. Soc., 2021, 143, 20792–20801 CrossRef CAS PubMed.
- S. Nitopi, E. Bertheussen, S. B. Scott, X. Liu, A. K. Engstfeld, S. Horch, B. Seger, I. E. L. Stephens, K. Chan, C. Hahn, J. K. Nørskov, T. F. Jaramillo and I. Chorkendorff, Chem. Rev., 2019, 119, 7610–7672 CrossRef CAS PubMed.
- T. Kawawaki, T. Okada, K. Takemae, S. Tomihari and Y. Negishi, ChemNanoMat, 2024, 10, e202300575 CrossRef CAS.
- Q.-J. Wu, D.-H. Si, P.-P. Sun, Y.-L. Dong, S. Zheng, Q. Chen, S.-H. Ye, D. Sun, R. Cao and Y.-B. Huang, Angew. Chem., Int. Ed., 2023, 62, e202306822 CrossRef CAS PubMed.
- M. Zhang, M. Lu, M.-Y. Yang, J.-P. Liao, Y.-F. Liu, H.-J. Yan, J.-N. Chang, T.-Y. Yu, S.-L. Li and Y.-Q. Lan, eScience, 2023, 3, 100116 CrossRef.
- C.-W. Kung, C. O. Audu, A. W. Peters, H. Noh, O. K. Farha and J. T. Hupp, ACS Energy Lett., 2017, 2, 2394–2401 CrossRef CAS.
- M. Chen, L. Han, J. Zhou, C. Sun, C. Hu, X. Wang and Z. Su, Nanotechnology, 2018, 29, 284003 CrossRef PubMed.
- K. M. Choi, D. Kim, B. Rungtaweevoranit, C. A. Trickett, J. T. D. Barmanbek, A. S. Alshammari, P. Yang and O. M. Yaghi, J. Am. Chem. Soc., 2017, 139, 356–362 CrossRef CAS PubMed.
- Y. Han, H. Xu, Y. Su, Z.-l. Xu, K. Wang and W. Wang, J. Catal., 2019, 370, 70–78 CrossRef CAS.
- S. Yao, S. Y. Jiang, B. F. Wang, H. Q. Yin, X. Y. Xiang, Z. Tang, C. H. An, T. B. Lu and Z. M. Zhang, Am. Ethnol., 2025, 137, e202418637 Search PubMed.
- C.-P. Wan, H. Guo, D.-H. Si, S.-Y. Gao, R. Cao and Y.-B. Huang, JACS Au, 2024, 4, 2514–2522 CrossRef CAS PubMed.
- Z. Yang, A. Li, H. Li, G. Lai, Y. Fu, Y. Zhang, K. Wang, S. Zeng, L. Xie and M. Li, Angew. Chem., Int. Ed., 2025, 64(19), e202505399 CAS.
- X. Chu, S. Liu, B. B. Luan, Y. Zhang, Y. Xi, L. H. Shao, F. M. Zhang and Y. Q. Lan, Am. Ethnol., 2025, 137, e202422940 CrossRef.
- W. Han, M. Shi and H. L. Jiang, Angew. Chem., Int. Ed., 2025, 64, e202421942 CrossRef CAS PubMed.
- Y. Q. Zhang, M. Liu, L. T. Zhang, N. Lu, X. Wang, Z. G. Li, X. H. Zhang, N. Li and X. H. Bu, Adv. Funct. Mater., 2025, 35, 2412406 CrossRef CAS.
- Y. Fei, R. Abazari, M. Ren, X. Wang and X. Zhang, Inorg. Chem., 2024, 63, 18914–18923 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |