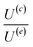Open Access Article
Open Access ArticleExperimental diffusiophoresis of porous and non-porous silica particles in dead-end pore microchannel geometry
Mansoureh Rashidi,
Matina Nooryani,
Giovanniantonio Natale * and
Anne M. Benneker
* and
Anne M. Benneker *
*
Department of Chemical and Petroleum Engineering, Schulich School of Engineering, University of Calgary, AB T2N 1N4, Canada. E-mail: gnatale@ucalgary.ca; anne.benneker@ucalgary.ca
First published on 1st May 2026
Abstract
The movement of colloids in response to a concentration gradient of solutes, known as diffusiophoresis (DP), plays a crucial role in various applications, including separations, sorting and reactant transport. In this study, we experimentally investigate DP of porous and non-porous silica particles in different electrolyte solutions using a microfluidic system with a dead-end pore geometry. Experimental results are linked to a theoretical analysis, which reveals that the direction of particle motion is governed by the interplay between electrophoresis (EP) and chemiphoresis (CP), influenced by effects such as double layer polarization (DLP) and electroosmotic flow (EOF). The type of electrolyte has a significant impact on the overall behavior, as different ions have significantly different interactions with the particles and each other. The findings provide valuable insights into the DP behavior of porous particles, which can be leveraged to optimize particle transport in biomedical and environmental applications.
1. Introduction and background
Diffusiophoresis (DP) is the movement of colloidal particles driven by a solute concentration gradient. In drug delivery, DP helps direct nanoparticles to specific target sites, increasing treatment efficiency and accuracy.1,2 In microfluidics, it allows for precise manipulation and sorting of particles, which is crucial for the development of lab-on-a-chip devices.3,4 DP was initially observed in non-electrolytes by Derjaguin et al. in the 1940s,42 and then was expanded to include electrolytes in the 1980s.5,6 DP in non-electrolyte systems arises from particle–solute interactions within a diffuse interfacial layer surrounding the particles. These interactions, whether repulsive or attractive, create an osmotic pressure gradient along the particle surface as a result of local solute concentration differences. The osmotic pressure imbalance across the particle surface drives a flow, known as chemi-osmosis (CO). This flow exerts a net force on the particle, causing chemiphoresis (CP) toward higher or lower solute concentrations, depending on whether interactions are attractive or repulsive.7–9 In electrolyte solutions, there is an additional contribution to DP; electrophoresis (EP) resulting from an induced electric field.10 EP (in DP) is driven by the electric field induced by the electrolyte concentration gradient, which arises because of differences in the ionic diffusivities of the solute. Initially, ions with higher diffusivity migrate faster, but Coulomb repulsion balances their migration rates, maintaining local electroneutrality within the solution.6,11 The ratio of cation (D+) and anion (D−) diffusivities is described by the diffusion potential (β) defined as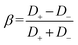 ,12 and indicates the direction of the developed electric field. CP in electrolyte systems results from localized double-layer polarization (DLP) caused by the concentration gradient around the particle surface.10 An electric double layer (EDL) with a thickness (λ) that is dependent on the local electrolyte concentration arises at the charged particle, where lower electrolyte concentrations result in larger EDLs. In a concentration gradient, the EDL thickness will change along the particle, and osmotic pressure differences will arise.
,12 and indicates the direction of the developed electric field. CP in electrolyte systems results from localized double-layer polarization (DLP) caused by the concentration gradient around the particle surface.10 An electric double layer (EDL) with a thickness (λ) that is dependent on the local electrolyte concentration arises at the charged particle, where lower electrolyte concentrations result in larger EDLs. In a concentration gradient, the EDL thickness will change along the particle, and osmotic pressure differences will arise.
DP of non-porous particles has been extensively studied through theoretical,13,14 numerical,15,16 and experimental approaches.17,18 In the literature, the DP velocity of a non-porous particle in a Newtonian fluid is defined as:
UDP = μDP ∇![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) C(x,t) C(x,t)
| (1) |
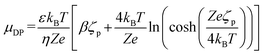 | (2) |
DP of porous particles has been examined only through a limited number of theoretical and numerical studies.21–23 Theoretical work derived the DP velocity of porous particles (presented in eqn (3)),21 while numerical studies have investigated the impact of different parameters such as EDL thickness, surface charges, diffusion potential (β) of different electrolytes, and porosity on DP velocity of porous particles.21,22,24
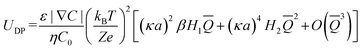 | (3) |
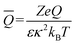 is the dimensionless fixed charge density of the porous particle. The first and second terms in eqn (3) represent the EP and CP contributions to the DP motion of the porous particle, respectively. The terms H1 and H2 are functions of porous particle's permeability, EDL thickness, and the diffusion coefficient of the ionic species both outside and inside the porous particle.21 The last term represents the equilibrium solution for the linearized Poisson–Boltzmann equation, which is applicable for small electric potential values (the Debye–Hückel approximation).21 From the available numerical and theoretical work, porous particles are expected to migrate faster than non-porous particles because (1) the interconnected pores in porous particles reduce the overall hydrodynamic drag force when compared to non-porous particles and (2) the fixed charges on the surface, distributed throughout the interior pore area, create a larger electric driving force that attracts more counter-ions into the particle interior, influencing the nominal charge of porous particles.26–28
is the dimensionless fixed charge density of the porous particle. The first and second terms in eqn (3) represent the EP and CP contributions to the DP motion of the porous particle, respectively. The terms H1 and H2 are functions of porous particle's permeability, EDL thickness, and the diffusion coefficient of the ionic species both outside and inside the porous particle.21 The last term represents the equilibrium solution for the linearized Poisson–Boltzmann equation, which is applicable for small electric potential values (the Debye–Hückel approximation).21 From the available numerical and theoretical work, porous particles are expected to migrate faster than non-porous particles because (1) the interconnected pores in porous particles reduce the overall hydrodynamic drag force when compared to non-porous particles and (2) the fixed charges on the surface, distributed throughout the interior pore area, create a larger electric driving force that attracts more counter-ions into the particle interior, influencing the nominal charge of porous particles.26–28
To the best of our knowledge, there are no experimental studies on how porous particles respond to an electrolyte concentration gradient. In this research, we investigate the behavior of porous particles with two different pore sizes under the influence of a temporary concentration gradient of various electrolytes and compare them with non-porous particles, using three different electrolyte compositions that have different values for β and pH.
2. Experimental methods
2.1. Experimental setup and sample preparation
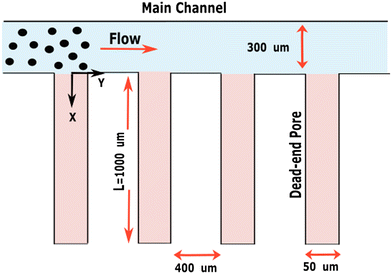
Non-porous and porous particles are analyzed under an imposed concentration gradient using different electrolytes (HCl, NaOH and NaCl) in a microfluidic dead-end pore geometry, as used prior.29 Fig. 1 illustrates a schematic of the chip, consisting of two sections; (1) the main channel with width 300 μm and height 90 μm and (2) pores of 1000 μm long, 50 μm wide and 30 μm in height. This narrow, long pore geometry allows us to neglect the pressure gradient in y-direction across a single pore.30 The height of the dead-end pores is thinner than that of the main channel to minimize disturbances from the main channel flow within the pores.31 Consequently, this design can be considered to exhibit one-dimensional motion of colloidal particles in the x-direction under the influence of a transient concentration gradient, while external effects as a result of the flow in the main channel are minimized. Chips were fabricated by mixing PDMS and a curing agent (SYLGARD 184) in a 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 weight ratio, then pouring the mixture onto a silicon wafer mold (DRIE etched at Nanofab Co, Edmonton Alberta). After curing, PDMS was bonded to a microscope glass slide (75 × 25 × 1 mm, VWR) using a plasma cleaner (HARRICK PLASMA, Ithaca, NY, USA). Chips were left overnight prior to use to minimize the impact of changes in PDMS surface properties after fabrication.
1 weight ratio, then pouring the mixture onto a silicon wafer mold (DRIE etched at Nanofab Co, Edmonton Alberta). After curing, PDMS was bonded to a microscope glass slide (75 × 25 × 1 mm, VWR) using a plasma cleaner (HARRICK PLASMA, Ithaca, NY, USA). Chips were left overnight prior to use to minimize the impact of changes in PDMS surface properties after fabrication.
Electrolyte solutions used in the experiments were prepared at a concentration of 10 mM, utilizing HCl (>37% purity, Sigma Aldrich), NaOH (>98% purity, Sigma Aldrich), and NaCl (>99.5% purity, Sigma Aldrich). The experiments employed 1 μm non-porous silica particles and 1 μm porous silica particles (all SOLADTM particles, Glantreo) with two different pore sizes: 2 nm and 10 nm. Table 1 summarizes the characteristics of the porous particles used in this study. Silica particles were suspended in deionized (DI) water using a bath sonicator for 30 min at a concentration of 2.6 × 10−4 by volume, which is in the dilute regime to avoid particle–particle interactions. The EP mobility of silica particles (both non-porous and porous) was measured using a Dynamic Light Scattering instrument (Malvern Zetasizer Nano ZS) at various pH levels to observe EP mobility changes during DP experiments in different electrolyte concentration gradients. EP mobility (μEP) was measured instead of ζ-potential because, for porous particles, the validity of the Smoluchowski approximation at high ionic strengths (κa ≥ 1) is debated due to their distinct electrokinetic behavior compared to non-porous particles.
| Property | Porous particle (2 nm pores) | Porous particle (10 nm pores) |
|---|---|---|
| BET (cm2 g−1) | 624.60 | 196.24 |
| Pore volume (cm3 g−1) | 0.566 | 0.517 |
| Porosity | 36.8% | 34.2% |
| Tortuosity | 1.65 | 1.71 |
For each experiment, the 10 mM electrolyte solution was injected into the microfluidic channel using a syringe pump (Harvard Apparatus, Pump 11 Elite). Once the main channel was filled, the outlet was blocked using a microfluidic valve to pressurize and fill the side channels. After ensuring that all channels were filled with electrolyte, the outlet blockage was removed and the injection of electrolyte continued for approximately 10–15 min to allow the system to reach equilibrium. Subsequently, the main channel was purged of electrolyte by introducing an air plug. The particle suspension was then injected into the main channel at a flow rate of 250 μl h−1. Upon reaching the entrance of the first pore, the injection rate was reduced to 30 μl h−1. Particle motion was observed using an optical microscope (ECHO, RVL-100-G) at a magnification of 20×, with images recorded at a frame rate of 8.3 frames per second. The motion of the particles under the influence of the electrolyte concentration gradient was recorded for approximately 600–700 s. All experiments were conducted at room temperature (21 ± 1 °C). After recording particle motion in the presence of the electrolyte concentration gradient, ImageJ software (a Java-based image processing program)32 was used to determine the maximum distance traveled by the particles within or away from the dead-end pores, denoted as L (μm). This value corresponds to the particle that reached the greatest distance from the pore entrance. The reported L values represent measurements obtained from two dead-end pores within the microchannel.
3. Results and discussion
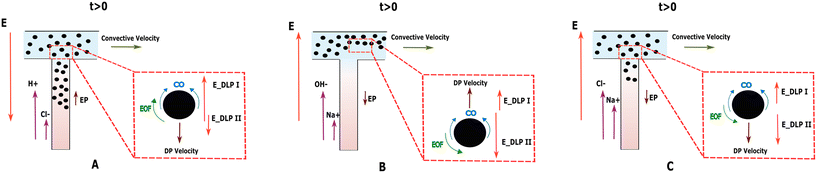
In electrolyte solutions, there are two types of double layer polarization leading to chemiphoresis.8,33 The electrostatic attraction between the charged particle and counter-ions accumulated in the EDL on the higher-concentration generates a local electric field around the particle affecting particle motion. This type of polarization, called DLP type I, drives the particle towards the higher electrolyte concentration. DLP type II, is generated due to the electrostatic repulsive force between the charged particle and co-ions accumulated adjacent to the EDL. This creates another local electric field around the particle, driving it towards the lower electrolyte concentration.6,8,33 Thus, the directions of these two induced electric fields are opposite to each other, as shown in Fig. 2. Both the counter and co-ions move under the influence of these microscopic localized electric fields, resulting in electroosmotic flow (EOF) near the particle surface.8,34 This EOF in turn contributes to the overall movement of the particles as a pressure gradient is induced. Depending on which DLP dominates, the movement as a result of CP can be in either direction.To explain the observed behavior of particles in a temporary concentration gradients, it is important to consider that there is a higher ionic concentration near the particle on the side of higher electrolyte concentration. Consequently, the thickness of the EDL near that side of the particle is thin when compared to the lower concentration side, which is true for all our experimental configurations.
Here, we separately present the behavior of particles in three different electrolytes, the characteristics of which are described in Table 2. We define a positive direction of DP motion as particle movement towards the higher solute concentration (towards and into the dead-end pores) and a negative direction as particle movement towards the lower solute concentration (away from the dead-end pores). For all experiments, the effective diffusivity of the particles, expressed as 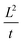 , is plotted for motion within the microfluidic chip in both directions (towards or away from the dead-end pores).
, is plotted for motion within the microfluidic chip in both directions (towards or away from the dead-end pores).
| HCl | NaOH | NaCl | |
|---|---|---|---|
| D+ (m2 s−1) | 9.3 × 10−9 | 1.33 × 10−9 | 1.33 × 10−9 |
| D− (m2 s−1) | 2.032 × 10−9 | 5.27 × 10−9 | 2.032 × 10−9 |
| β | 0.642 | −0.596 | −0.207 |
In the absence of a concentration gradient, particles penetrate only a short distance into the dead-end pore, approximately equal to the entrance length (Le).19 Le represents the distance from the pore inlet where the local velocity, typically reaches 99% of its fully developed value.35 Le is calculated using an empirical equation (eqn (4)) applicable to rectangular cross-sections.36 Here, Re is the Reynolds number defined as 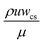 for a rectangular channel, where wcs is the width of the cross-section. Additionally, ρ and μ represent the fluid's density and viscosity, respectively. Since the electrolytes used in this study have density and viscosity values close to those of water, we used the properties of water at 20 °C in our calculations. Specifically, we assumed μ = 1.0016 mPa s and ρ = 998.2 kg m−3 to compute Le, which was found to be 27.5 μm using eqn (4).
for a rectangular channel, where wcs is the width of the cross-section. Additionally, ρ and μ represent the fluid's density and viscosity, respectively. Since the electrolytes used in this study have density and viscosity values close to those of water, we used the properties of water at 20 °C in our calculations. Specifically, we assumed μ = 1.0016 mPa s and ρ = 998.2 kg m−3 to compute Le, which was found to be 27.5 μm using eqn (4).
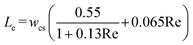 | (4) |
3.1. Particles in HCl
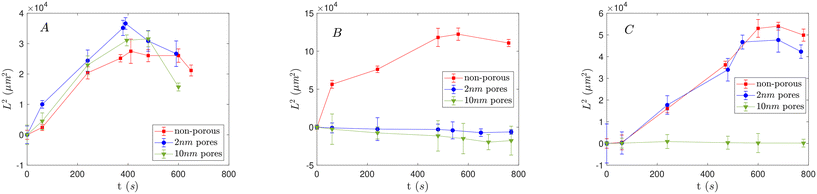
Fig. 3A depicts the effective diffusivity of porous and non-porous particles under a temporary imposed HCl concentration gradient. When particles reach the inlet of the first dead-end pore, all different types of particles move into the pore towards the higher electrolyte concentration as shown in Fig. 4. This indicates that CP dominates over EP, as schematically depicted in Fig. 2A.β is positive for HCl (see Table 2), which indicates that the background macroscopic electric field induced by the electrolyte concentration gradient, is directed towards the higher concentration of the electrolyte, as illustrated in Fig. 2A. Initially, the EP mobility (and thus ζ-potential) of the particles suspended in DI water pH = 7.14, is negative (Fig. 7). With time, as the pH around the particles decreases, their absolute EP mobility decreases, but stays negative. In this case, as ζβ < 0, EP is expected out of the channel in negative direction.
For HCl the faster diffusion of H+ ions (in comparison to Cl−) into the main channel and the attraction between the negatively charged particles and protons leads to their adsorption on the particle surface, reducing the effective surface charge of particles and subsequently lowering the absolute EP mobility. This results in a more pronounced establishment of DLP type I but a weaker DLP type II, consistent with previous reports.8,33 Furthermore, as illustrated in Fig. 2A, this DLP results in local EOF around the particles in the same direction as the developed chemi-osmosis (CO). This enhances the ion transport near the particles' surface and propels them toward the higher electrolyte concentration.
Overall, at low pH, as in the case of HCl, the particles exhibit a small absolute EP mobility, indicating a weakened EP response when compared to the CP response. This results in CP to dominate and drive both particle types toward the higher electrolyte concentration end.
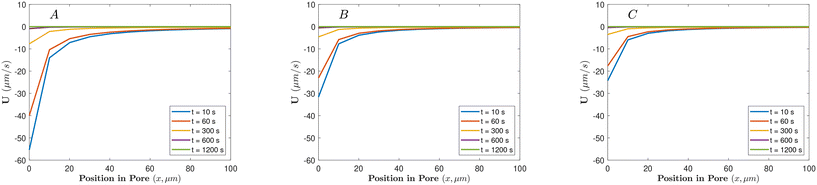
Fig. 3A shows that the effective diffusivity of porous particles is greater than that of non-porous particles. Such behavior indicates faster effective transport and is consistent with a higher DP velocity compared to non-porous particles. The DP motion of porous particles is generally faster than that of non-porous particles due to the more fixed charges distributed throughout the interior region of the particle, combined with the reduced hydrodynamic drag force26–28 measured experimentally in this study (see Fig. 1, Appendix A in the SI). Particles with a pore size of 2 nm travel further into the dead-end pore compared to particles with a pore size of 10 nm. The “hindrance effect”21,22 occurs when there is resistance to the diffusion of ions inside the porous particle. A smaller pore size can result in a more directed or focused movement of ions into the particle, leading to a stronger concentration gradient across the particle, and therefore, higher DP velocity.21,22 The characterization data in Table 1 further support this idea; the smaller tortuosity value for the 2 nm pore particles suggests a less obstructed, more direct path for ion transport compared to the 10 nm pore particles, which directly contributes to their enhanced migration speed.
Numerical studies have shown that pore size, porosity and surface heterogeneity significantly influence the overall ζ-potential of porous particles,38,39 influencing their EP mobility. Particle heterogeneity causes deviations from traditional flat surface charge predictions seen in non-porous particles, where surface charge density decreases due to EDL overlap in the “valleys” and increases due to curvature effects on the “hills”. The average ζ-potential and EP mobility is influenced by which effect is more dominant.39 Additionally, the larger surface area and ion penetration into the pores of porous particles can enhance electrostatic screening, thereby reducing their effective surface potential. Consequently, the measured EP mobility of porous particles, which represents their ζ-potential, differs from that of non-porous particles within the same pH range, as shown in Fig. 7. However, the measurements indicate that with reduced EDL overlap (at very low or very high pH), the EP mobility of porous and non-porous particles converges. Studies support that ion penetration, internal EDL overlap, and internal CP mechanisms can play an important role in porous particle transport.21,24 In HCl solution, because counter-ion condensation reduces the effective surface charge, the strength of EP is weakened. CP therefore dominates, propelling porous particles toward regions of higher electrolyte concentration, in agreement with experimental observations.
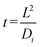 , where L is the length of the dead-end pore and Di is the diffusivity of the electrolyte ions, based on the ion with the smaller diffusivity (Cl−). As can be inferred from Fig. 3A, non-porous particles start leaving the dead-end pore (reduction of effective diffusivity) around 8.2 min, which corresponds closely to the calculated time for the disappearance of the electrolyte concentration gradient.
, where L is the length of the dead-end pore and Di is the diffusivity of the electrolyte ions, based on the ion with the smaller diffusivity (Cl−). As can be inferred from Fig. 3A, non-porous particles start leaving the dead-end pore (reduction of effective diffusivity) around 8.2 min, which corresponds closely to the calculated time for the disappearance of the electrolyte concentration gradient.According to Fig. 3A, the sequence of departure time for different particles under a temporary HCl concentration gradient is as follows: porous particles with 2 nm pore size leave first, followed by porous particles with 10 nm pore size, and finally, non-porous particles. We hypothesize that over time, the phenomenon of “counter-ion condensation” or “solidification” reduces the influence of CP, allowing EP to become the dominant mechanism driving the particles out of the dead-end pores.1,11,22 This effect is more pronounced in porous particles due to ion diffusion into their internal structure, which accelerates the charge neutralization process and leads to earlier departure compared to non-porous particles. Furthermore, previous studies have shown that increased permeability; as in porous particles with 10 nm pores; slows the rate of charge neutralization or solidification.27 This explains why 10 nm porous particles exit the channel later than those with 2 nm pores, despite both leaving earlier than their non-porous counterparts. Moreover, as the electrolyte within the dead-end pores is gradually replaced by DI water over time, the local pH increases. This rise in pH leads to an increase in the absolute value of the surface potential of the particles and consequently, their EP mobility (as shown in Fig. 7), thereby enhancing the EP effect and further promoting particle migration out of the dead-end pores.
3.2. Particles in NaOH
Fig. 3B shows the effective diffusivity of particles under the concentration gradient of NaOH in the microchannel. Non-porous particles and porous particles with 2 and 10 nm pore sizes demonstrate distinct behavior, moving in different directions. In contrast to the case of HCl, in NaOH the anion moves faster and the local pH of both solutions are different, influencing the particles' EP mobility and ζ-potential.The diffusion potential β is negative for NaOH (Table 2) indicates a background electric field towards the lower electrolyte concentration exists, as shown in Fig. 2B. The negatively charged particles are thus expected to move towards the higher electrolyte concentration due to EP (since ζβ > 0). This is experimentally observed for non-porous particles (red squares).
As shown in Fig. 7, EP mobility and consequently, surface potential remains negative across the pH range of 7–12 for DI water and NaOH solution. As pH changes, the electrolyte concentration, EP mobility of the particles (representative of ζ-potential), and κa (or EDL thickness) also vary during the experiment. The absolute value of EP mobility for both particle types is greater in the NaOH solution compared to the HCl solution (Fig. 7). When EP mobility is high, indicating a high particle surface potential, a strong repulsive force arises between particles and the co-ions (in this case, OH−), which increases the amount of co-ions near the outer region of the high-concentration side of the EDL.6,15,33 Due to the relatively slow diffusion of Na+, lower concentration of counter-ions is present in the EDL at the top of the dead end channel, making DLP type II stronger than DLP type I. Therefore, co-ions have a greater influence on particle motion than counter-ions.8,28 Due to the stronger induced electric field by DLP type II, an EOF in the opposite direction of the CO along the particle surface is created. This EOF could overcomes CO, reversing the ions diffusion direction within the EDL, and consequently, the CP direction, thereby driving particles toward the lower electrolyte concentration. This indicates that EP dominates over CP for non-porous particles in NaOH.
The exclusion zone thickness formed in the case of porous particles with a 2 nm pore size is smaller compared to those with a 10 nm pore size (smaller absolute magnitude of effective diffusivity in Fig. 5). As a result, particles with 2 nm pores move less far away from the pores (see “hindrance effect” in section 3).
Our observations reveal that the exclusion zone thickness decreases over time for both types of porous particles. Conversely, non-porous particles gradually exit the dead-end pores after a specific duration under the temporary NaOH concentration gradient, as depicted in Fig. 3B. The concentration gradient within the dead-end pores is expected to dissipate within 12.53 min, calculated based the ion with the smaller diffusivity (Na+). This suggests that the transient behavior observed for both porous and non-porous particles are likely influenced by the dissipation of the concentration gradient in the system, as was the case for HCl.
3.3. Particles in NaCl
The observation of DP motion of porous and non-porous particles in a temporary NACl NaCl concentration gradient shows that porous particles with 10 nm pore size move to the dead-end pores up to the entrance length described earlier. Non-porous particles and particles with small 2 nm pores, move into the dead-end pores (Fig. 3C and 6). For NaCl, negative β (Table 2) creates a background electric field towards the lower electrolyte concentration as shown in Fig. 2C, similar to that of the NaOH case. As mentioned, the particles are initially negatively charged in DI water, and since ζβ > 0, the background electric field drives negatively charged particles toward the higher electrolyte concentration.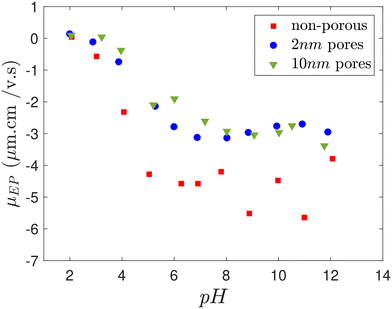 | ||
| Fig. 7 EP mobility measurement of 1 μm non-porous silica particles and 1 μm porous silica particles with 2 and 10 nm pore sizes at different pH. | ||
Due to the higher diffusion rate of co-ions (DCl−) and the repulsive force between the particles and co-ions, the resulting DLP type II is stronger than DLP type I, similarly to the NaOH case. Consequently, EOF in the opposite direction of CO forms along the particles surface. The close diffusivity magnitudes of Na+ and Cl− suggests that DLP type I and DLP type II compete, resulting in a weaker EOF along the particles surface compared to the NaOH case, where there is a larger difference in ion diffusivity. Although the EOF opposes the CO effect on the particle surface, it is not strong enough to reverse the direction of ion diffusion on the particle's surface. Therefore, the net effect of CP remains in the same direction as EP for particles in NaCl, and both mechanisms act cooperatively to drive particles toward the higher electrolyte concentration side. Since βNaCl < βNaOH, the background electric field and EP strength is weaker in NaCl compared to NaOH. This results in a smaller effective diffusivity into the dead-end pores under the imposed electrolyte concentration gradient, as shown in Fig. 3B and C.
In experiments with non-porous and porous particles exposed to NaCl concentration gradient, both types of particles started to leave the dead-end pores as time progresses, similar to the behavior seen with HCl and NaOH. The concentration gradient within the dead-end pores is expected to dissipate within approximately 12.53 minutes, driven by the slower diffusion of Na+ ions. This is linked to the dissipation of the concentration gradient within the system (Fig. 3), as was the case in the other electrolytes.
3.4. Comparison to theoretical predictions
The velocity equations developed by Prieve et al.,6 shown in eqn (5) and (6) are used to evaluate the relative significance of CP over EP. These equations are valid under the condition of κa ≥ 1 (small EDL thickness) and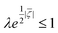 . This allows the use of eqn (7) to estimate the velocity ratio of
. This allows the use of eqn (7) to estimate the velocity ratio of 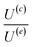 , quantifying the contribution of CP relative to EP when particles are driven by a transient concentration gradient. In these expressions, γ is a dimensionless surface parameter defined as
, quantifying the contribution of CP relative to EP when particles are driven by a transient concentration gradient. In these expressions, γ is a dimensionless surface parameter defined as 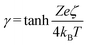 ,
, ![[small zeta, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0c7.gif) is the dimensionless form of the ζ-potential, and C∞ is the bulk concentration of electrolyte. To obtain the ζ-potential values used in these equations, the experimentally measured EP mobility (from DLS) was converted to ζ-potential using the Smoluchowski approximation.
is the dimensionless form of the ζ-potential, and C∞ is the bulk concentration of electrolyte. To obtain the ζ-potential values used in these equations, the experimentally measured EP mobility (from DLS) was converted to ζ-potential using the Smoluchowski approximation.
In our system, generally, the required assumptions hold true, and calculated values of 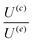 across a specific range of pH values that represent the potential pH variations under the different concentration gradients are summarized in Table 3. Empty cells in the table indicate pH conditions where the underlying assumptions are not valid and the velocity ratio cannot be reliably calculated.
across a specific range of pH values that represent the potential pH variations under the different concentration gradients are summarized in Table 3. Empty cells in the table indicate pH conditions where the underlying assumptions are not valid and the velocity ratio cannot be reliably calculated.
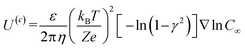 | (5) |
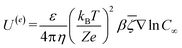 | (6) |
 | (7) |
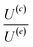 is smaller than 1 for all particles in the HCl system, indicating that in all cases EP velocity dominates over CP velocity, and this becomes more pronounced at lower pH. This is true for the gradients of NaOH and NaCl as well (see Tables 1 and 2 in Appendix B in the SI). According to this, for HCl particles should migrate toward regions of lower electrolyte concentration, contradicting our experimental observations. For NaOH and NaCl these values indicate that particles are expected to migrate toward the region of higher electrolyte concentration. Only for NaCl, our experimental observations fully align with these estimations.
is smaller than 1 for all particles in the HCl system, indicating that in all cases EP velocity dominates over CP velocity, and this becomes more pronounced at lower pH. This is true for the gradients of NaOH and NaCl as well (see Tables 1 and 2 in Appendix B in the SI). According to this, for HCl particles should migrate toward regions of lower electrolyte concentration, contradicting our experimental observations. For NaOH and NaCl these values indicate that particles are expected to migrate toward the region of higher electrolyte concentration. Only for NaCl, our experimental observations fully align with these estimations.
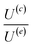 for non-porous particles and porous particles with 2 nm and 10 nm pore sizes under the transient HCl concentration gradient. The experiment captures particle behavior across the pH range of 2 to 7
for non-porous particles and porous particles with 2 nm and 10 nm pore sizes under the transient HCl concentration gradient. The experiment captures particle behavior across the pH range of 2 to 7
A possible explanation for these discrepancies is that in deriving the expressions for CP and EP, Prieve et al.6 assumed a uniform electric field outside the EDL and did not account for microscale electric fields induced by ions accumulation around the particles. These local electric fields give rise to DLP type I and II, which significantly influence particles motion, especially for porous particles. Additionally, these expressions were derived for non-porous particles and are likely not valid for porous particles, where more complex phenomena can occur, such as ion penetration into the particle, formation and overlap of internal EDLs,38 and even potential EOF within the porous matrix can all affect particle motion.21 Theoretical studies on DP velocity of porous particles21 have shown that the DP transport of this type of particles should be treated as a volumetric phenomenon rather than a surface-driven one. When the fluid moves through the porous particle, it interacts directly with the fixed charge density distributed throughout the internal volume, rather than just a thin surface double layer. This means that the macroscopic EP and the CP, exert a force on every charged segment within the particle's interior. This internal coupling often causes the CP component to become more dominant. Consequently, the porosity and permeability of the particle do not just modify the velocity magnitude; they can fundamentally dictate the direction of motion by re-balancing the competition between the EP and the internal CP.21 Another potential source of discrepancy for porous particles arises from the use of the Smoluchowski approximation to calculate the ζ-potential, which was used in estimating the ratio of CP to EP velocities. As mentioned earlier, the validity of this approximation for porous particles is not validated. Although in the theoretical model the effects of DLP type I and DLP type II are ignored, the close diffusivity of Na+ and Cl− likely leads to competing induced electric fields. This balance appears to validate the theoretical prediction in the NaCl case, in contrast to the electrolytes with large diffusivity differences.
To rule out the effect of pH variation in the channel on particle's motion in the system playing a dominant role, we numerically examined how changes in pH influence particle DP velocity. To analyze this process, the one-dimensional transient concentration gradient of H+ was numerically solved using the equations presented in Appendix C.30,41 As discussed previously, the long pore geometry allows the pressure gradient in the y-direction to be neglected30 and the pores are designed to be thinner than the main channel to minimize disturbances from the main channel flow.
The results, shown in Fig. 8 for HCl and Appendix C for NaOH, indicate that both non-porous and porous particles are predicted to have a negative velocity in HCl based on pH variations alone, while in the case of NaOH all types of particles are predicted to migrate toward the region of higher electrolyte concentration. Again, this is in contradiction to the experimental observations for most of the particles. Furthermore, the variation in pH does not induce a sign change of the particle ζ-potential, indicating that the particle movement reversal is not a result of the pH variation. Overall, this analysis of the effect of pH-variation cannot explain the experimentally observed particle behavior, supporting our hypothesis that the direction of particle motion is influenced by the particle structure and the balance between EP and CP and is not dictated by the pH variation in the system.
4. Conclusions
In this experimental work, diffusiophoresis of porous and non-porous silica particles in dead-end microchannels under different electrolyte concentration gradients is investigated. The results show that electrolyte composition plays a key role in determining particle migration behavior, while the internal structure of porous particles further modifies their response. Overall, the study reveals a complex interplay between particle structure, interfacial electrokinetic phenomena, and electrolyte diffusivity.In an acidic environment (HCl), where higher diffusivity of H+ ions within the dead-end pores prevails, both porous and non-porous particles migrate towards regions of higher electrolyte concentration. Due to the attractive force between the negatively charged particle and H+ ions, the DLP type I is stronger than DLP type II in this case. Consequently, the resulting EOF and the CO (diffusion of the ions on the particle surface) contributing to CP align, dominating over the weaker EP force, driving the particles towards the higher electrolyte concentration. Conversely, in a basic NaOH electrolyte, stronger DLP type II induces opposing EOF and CO contributions to CP, which can result in a reversal of the direction of the ionic diffusion flow on the particle's surface, thereby altering the CP direction toward the region of lower electrolyte concentration. Additionally, due to the smaller absolute value of the EP mobility of porous particles compared to non-porous particles, they are driven towards regions of lower electrolyte concentration, creating an exclusion zone at the entrance of the pores. In NaCl electrolyte, the close diffusivity of Cl− and Na+ ions result in the compensation of the two types of DLP. As a result, EP becomes dominant in driving both negatively charged porous and non-porous particles towards regions of higher electrolyte concentration. Notably, our study also highlights the significant reduction in drag forces experienced by porous silica particles compared to non-porous ones, a phenomenon influenced by pore size and permeability as confirmed through optical tweezers measurements.
To validate the experimental results, a theoretical velocity ratio of CP to EP was used to evaluate the relative contribution of each mechanism in driving particle motion. While the model successfully predicts particle behavior in the NaCl system, likely due to the balanced effects of DLP type I and type II, its predictions deviate from the experimental observations in the HCl and NaOH cases. This discrepancy is hypothesized to arise as the model does not account for microscale induced electric fields around the particle (associated with DLP type I and type II). In addition, it does not incorporate the complex dynamics involved in the DP of porous particles, such as ion penetration into the pores, overlap of internal EDLs, and EDL relaxation effects. The absence of these physical mechanisms limits the model's ability to accurately predict DP behavior in more complex systems. A numerical study was also conducted to examine the effect of pH variation in the system on the DP behavior of the particles. The results indicate that local pH variations do not alter the direction of motion of particles, further supporting the proposed mechanism and the physical interpretation of the observed DP behavior of porous particles.
Overall, our results demonstrate that DP transport in confined geometries such as dead-end pores is governed by both electrolyte properties and particle internal structure. There is a strong interplay between electrolyte composition and particle structure in determining both the direction and magnitude of particle migration. These findings provide practical design insights for microfluidic systems by showing that particle accumulation, exclusion, or penetration depth can be tuned through appropriate selection of electrolyte type and particle pore size. Such control over particle transport is relevant for applications including targeted particle delivery, sensing, passive separation, and controlled trapping of colloids in microfluidic and porous environments.
Conflicts of interest
The authors declared no conflict of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Raw data and additional information can be obtained from the corresponding authors.Supplementary information is available. See DOI: https://doi.org/10.1039/d5lc01020a.
Acknowledgements
The authors acknowledge the financial support from Canada First Research Excellence Fund (CFREF).Notes and references
- O. Annunziata, D. Buzatu and J. G. Albright, J. Phys. Chem. B, 2012, 116, 12694–12705 CrossRef CAS PubMed.
- M. S. McAfee and O. Annunziata, Langmuir, 2015, 31, 1353–1361 CrossRef CAS PubMed.
- S. V. Hartman, B. Božič and J. Derganc, New Biotechnol., 2018, 47, 60–66 CrossRef PubMed.
- M. Seo, S. Park, D. Lee, H. Lee and S. J. Kim, Lab Chip, 2020, 20, 4118–4127 RSC.
- J. L. Anderson, Annu. Rev. Fluid Mech., 1989, 21, 61–99 CrossRef.
- D. Prieve, J. Anderson, J. Ebel and M. Lowell, J. Fluid Mech., 1984, 148, 247–269 CrossRef CAS.
- P. O. Staffeld and J. A. Quinn, J. Colloid Interface Sci., 1989, 130, 88–100 CrossRef CAS.
- J.-P. Hsu, W.-L. Hsu and Z.-S. Chen, Langmuir, 2009, 25, 1772–1784 CrossRef CAS PubMed.
- J. Anderson, M. Lowell and D. Prieve, J. Fluid Mech., 1982, 117, 107–121 CrossRef CAS.
- D. Velegol, A. Garg, R. Guha, A. Kar and M. Kumar, Soft Matter, 2016, 12, 4686–4703 RSC.
- E. Lee, Interface Science and Technology, Elsevier, 2019, vol. 26, pp. 323–358 Search PubMed.
- T.-Y. Chiang and D. Velegol, J. Colloid Interface Sci., 2014, 424, 120–123 CrossRef CAS PubMed.
- H. J. Keh and Y. K. Wei, Langmuir, 2000, 16, 5289–5294 CrossRef CAS.
- A. S. Khair, J. Fluid Mech., 2013, 731, 64–94 CrossRef CAS.
- D. C. Prieve and R. Roman, J. Chem. Soc., Faraday Trans. 2, 1987, 83, 1287–1306 RSC.
- S. Shim, M. Baskaran, E. H. Thai and H. A. Stone, Lab Chip, 2021, 21, 3387–3400 RSC.
- S. Shin, Phys. Fluids, 2020, 32, 101302 CrossRef CAS.
- S. Shim, J. K. Nunes, G. Chen and H. A. Stone, Phys. Rev. Fluids, 2022, 7, 110513 CrossRef.
- S. Battat, J. T. Ault, S. Shin, S. Khodaparast and H. A. Stone, Soft Matter, 2019, 15, 3879–3885 RSC.
- D. Li, Electrokinetics in microfluidics, Elsevier, 2004 Search PubMed.
- Y. K. Wei and H. J. Keh, J. Colloid Interface Sci., 2004, 269, 240–250 CrossRef CAS PubMed.
- H. Y. Huang and H. J. Keh, J. Phys. Chem. B, 2015, 119, 2040–2050 CrossRef CAS PubMed.
- S. Marbach, H. Yoshida and L. Bocquet, J. Fluid Mech., 2020, 892, A6 CrossRef CAS PubMed.
- S.-C. Tsai and E. Lee, Langmuir, 2019, 35, 3143–3155 CrossRef CAS PubMed.
- M. v. Smoluchowski, Band II, Barth-Verlag, Leipzig, 1921, pp. 366–427 Search PubMed.
- R. Wu and D. Lee, Chem. Eng. Sci., 1998, 53, 3571–3578 CrossRef CAS.
- P. P. Gopmandal, S. Bhattacharyya and B. Barman, Eur. Phys. J. E: Soft Matter Biol. Phys., 2014, 37, 1–12 CAS.
- E. Lee, Interface Science and Technology, Elsevier, 2019, vol. 26, pp. 385–409 Search PubMed.
- M. Nooryani, A. M. Benneker and G. Natale, Lab Chip, 2023, 23, 2122–2130 RSC.
- B. M. Alessio, S. Shim, E. Mintah, A. Gupta and H. A. Stone, Phys. Rev. Fluids, 2021, 6, 054201 CrossRef.
- S. Shin, E. Um, B. Sabass, J. T. Ault, M. Rahimi, P. B. Warren and H. A. Stone, Proc. Natl. Acad. Sci. U. S. A., 2016, 113, 257–261 CrossRef CAS PubMed.
- J. Schindelin, C. T. Rueden, M. C. Hiner and K. W. Eliceiri, Mol. Reprod. Dev., 2015, 82, 518–529 CrossRef CAS PubMed.
- Y. Pawar, Y. E. Solomentsev and J. L. Anderson, J. Colloid Interface Sci., 1993, 155, 488–498 CrossRef CAS.
- H. J. Keh and H. C. Ma, Langmuir, 2005, 21, 5461–5467 CrossRef CAS PubMed.
- R. Shah, Laminar flow forced convection in ducts, Supplement 1 to Advances in Heat Transfer, Academic Press, New York, 1978, pp. 153–195 Search PubMed.
- T. Ahmad and I. Hassan, J. Fluids Eng., 2010, 132(4), 041102 CrossRef.
- L.-H. Yeh and J.-P. Hsu, Soft Matter, 2011, 7, 396–411 RSC.
- F. E. Yakin, M. Barisik and T. Sen, J. Phys. Chem. C, 2020, 124, 19579–19587 CrossRef CAS.
- B. O. Alan, M. Barisik and H. G. Ozcelik, J. Phys. Chem. C, 2020, 124, 7274–7286 CrossRef CAS.
- W. Fang and E. Lee, J. Colloid Interface Sci., 2015, 459, 273–283 CrossRef CAS PubMed.
- E. Coleman and A. Gupta, Phys. Rev. Fluids, 2025, 10, 103701 CrossRef.
- B. V. Derjaguin, G. P. Sidorenkov, E. A. Zubashchenkov and E. V. Kiseleva, Kolloid-Z., 1947, 9, 335–347 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |