Open Access Article
Open Access ArticleVascularizing organoids-on-chip for perfused and personalized models
Bianca
Menzani†
,
Priscille
De Gea†
 ,
Xavier
Gidrol
,
Xavier
Gidrol
 * and
Emily
Tubbs
* and
Emily
Tubbs
 *
*
University Grenoble Alpes, CEA, Inserm, IRIG, UA13 BGE, Biomics, 38000 Grenoble, France. E-mail: xavier.gidrol@cea.fr; emily.tubbs@cea.fr
First published on 27th February 2026
Abstract
Organoids represent one of the most advanced in vitro models for studying human physiology, development and disease. Their potential is very important and they have broad applications, but their impact is currently limited by persistent challenges such as incomplete maturation, batch variability, restricted long-range interactions and, critically, the absence of functional and perfusable vasculature. Integrating organoids into microfluidic platforms offers a way to overcome some of these constraints by providing a controlled and dynamic microenvironment with precisely tuned physical and biochemical cues. Among emerging strategies, vascularization stands out as a critical step toward improving organoid physiology and relevance: establishing stable, lumenized and perfusable networks within the 3D structure enables direct delivery of oxygen and nutrients, facilitates metabolic waste removal and promotes their maturation beyond embryonic stages. Achieving such models will require the combined expertise of stem cell biology, microfluidics, and biomaterials engineering to generate devices with organ-specific endothelial and stromal components, physiological flow profiles, and bidirectional anastomosis between endogenous and exogenous vascular compartments. This review discusses the biological rationale, current strategies, and technical considerations for vascularizing organoids-on-chip, highlighting their potential to improve physiological relevance, functional performance, personalization and translational applicability.
1. Introduction
Organoids are 3D self-assembled cell structures derived from embryonic stem cells (ESCs), adult stem cells (ASCs), or pluripotent stem cells (PSCs), that can represent a single organ or multiple organs, and are models enabling numerous readouts (morphological architecture, gene expression signatures, physiology) in comparison with 2D methods.1,2 Through aggregation and self-organization, the aim is to recapitulate the structure and function of the in vivo counterparts, thereby providing physiologically relevant models for: studying organ development, disease modelling, and exploring transplantation strategies. Advances in the field have rapidly expanded the repertoire of available organoid types, driven by the need for more predictive in vitro systems. Their complexity can be tailored by: adjusting the number and type of constituent cell populations, the starting stage of maturation, while culture duration and conditions strongly influence maturation and functionality.3 The global adoption of organoid technology has led to diverse methodologies—and equally diverse challenges. Indeed, as the technology continues to develop and spread, the scientific community is tasked with clearly defining these limitations and working together to identify viable solutions for patient applications.4 Four major hurdles consistently arise in organoid research: incomplete maturation, insufficient or inadequate cell–cell and cell–matrix interactions, and the absence of vascularization.2,3 The advent of microphysiological systems has enabled the development of dynamic culture, controlled flow dynamics, and continuous nutrient replenishment in small-volume environments.5 Organoids-on-chip have gained increasing relevance due to their ability to precisely regulate physical and mechanical cues and to incorporate real-time monitoring with integrated sensors (e.g., oxygen, temperature). Among the possibilities offered by microfluidic devices, a key focus is to establish a vascular network capable of supplying and draining the organoid. Indeed, hypoxic cores and necrosis develop because nutrient and oxygen delivery in organoids is diffusion-limited when diameters exceed ∼200 μm.6 Overcoming this diffusion-limit by introducing perfusable vascular networks has become a central objective in the field. Such active convection of nutrients and oxygen, in contrast to passive diffusion, holds the potential to markedly enhance organoid growth, viability, and functionality.7In this review, we first outline the major limitations of current organoid models and then examine the techniques available to tackle the absence of a functional vascularization in organoids-on-chip systems, providing a critical viewpoint informed by recent work from our group.8
2. Organoids in static culture: a limited model
2.1. Maturity
Organoid formation recapitulates key aspects of embryonic development, meaning that cells are inherently immature at the outset and must acquire organ-specific functions as the organoid develops.3 Two main approaches are used: either (i) co-differentiating all stem cells within a single 3D culture from the start, or (ii) pre-differentiating individual cell types in 2D monoculture to a defined developmental stage before assembling into an organoid.4Maturity in this context encompasses transcriptional, structural, epigenetic, and functional hallmarks, many of which remain fetal-like in vitro. Most organoid models show incomplete maturation despite achieving structural organization and partial functional features of the target organ. Transcriptional analysis of kidney organoids, for instance, reveal profiles resembling first-trimester tissue, with expression of early developmental genes even though regional specificity of nephron structures was reached.9 When considering the chromatin accessibility as a feature of epigenetic maturation of the kidney organoids, similar observation were made of foetal-stage maturation of cells, with differences even more pronounced in some nephron regions.10 In intestinal organoids, maturation toward an adult-like state is markedly hindered in vitro compared with in-tissue development, even in models derived from adult stem cells.11 Pancreatic organoids cultured in vitro exhibit altered metabolic activity (glucose response, respiratory activity) – a key functional readout for this model type.12 For brain organoids, functional maturation depends on neuronal connectivity between distinct regions to establish physiological circuits, a process that remains challenging in vitro.13
Multiple studies have shown that transplantation into host animals can promote functional, morphological, and transcriptional maturation, as well as the emergence of additional cell types, highlighting the role of a physiological environment in advancing organoid development.12,14,15 These findings underline the need to optimize in vitro culture system as conventional static methods often achieve high levels of complexity and function only after in vivo transplantation. Creating a supportive microenvironment will be essential to drive maturation without leaving the dish. In static culture, however, the surrounding matrix remains synthetic and reductionist, offering only a narrow subset of the biochemical and mechanical signals present in vivo. This dependence on artificial matrices represents a second major barrier, as these scaffolds only partially recapitulate native microenvironmental cues.
2.2. Microenvironment interaction
The importance of recreating an extracellular matrix (ECM)-like environment for organoid formation has long been recognized.1 A common strategy is to embed organoids in a hydrogel containing matrix proteins, often supported by fibroblasts that secrete collagen, fibronectin, and laminin, rapidly assembling into a scaffold.16 Many protocols still rely on Matrigel – a laminin- and collagen-rich hydrogel derived from mouse sarcoma – which remains one of the most widely used ECM-based matrices in vitro.17 However, its tumour-derived origin introduces unwanted factors and significant lot-to-lot variability, potentially affecting experimental reproducibility.18 Alternative approaches include supplementing natural or synthetic hydrogels with defined ECM proteins (e.g., laminin, collagen, fibrin) or using scaffolds from decellularized tissues (dECM). dECM offers biochemical and mechanical properties closely matching native tissue and has been increasingly recognized for enhancing 3D organization, functional maturation, and phenotypic stability of organoids.18,19 For instance, brain dECM, which closely mirrors the native tissue protein composition, has demonstrated greater batch-to-batch consistency than Matrigel,20 while pancreatic dECM has been shown to enhance structural integrity compared with collagen matrices.21 Nonetheless, dECM variability may still arise due to differences in tissue source20 or heterogeneous ECM distribution within an organ, underscoring the potential need for standardized protocols.22,23Beyond biochemical composition, the mechanical properties of hydrogels – stiffness, viscoelasticity, and tunability – also play a decisive role in organoid behaviour.24,25 Mechanical parameters can be standardized through controlled fabrication protocols,26 or quantitatively characterized by techniques such as atomic force microscopy, which probes the elastic modulus of the material at the microscale or rheometry, which provides bulk measurements of viscoelastic behaviour.27 As matrix physical properties influence processes including stem cell fate decisions, cytoskeletal organization, and morphogenetic patterning, precise definition and reproducibility of hydrogel mechanical properties are critical for organoid culture.
In contrast to these structural cues, the biochemical composition of the ECM influences lineage commitment and functional behaviour through molecular signalling pathways. Both soluble factors (cytokines, growth factors) and insoluble ECM components, such as collagen and fibronectin assemblies, engage integrin receptors triggering signalling cascades that regulate gene expression and cell fate.28,29 Incorporating additional support cells into the hydrogel can further enhance the microenvironment: fibroblasts contribute to ECM production and direct cell–cell contacts, while endothelial, immune, stromal, or cancer stem cells can be added depending on the research focus.28,30–32 To reproduce more complex or spatially organized interactions, engineering approaches such as bioprinting now allow spatial arrangement of these supporting cells, for example controlled positioning endothelial cells (EC) around organoids to better replicate physiological interactions and functions.21 While these strategies successfully capture local microenvironmental cues, they remain insufficient for modelling interactions that depend on long-range connectivity, such as circuit formation between distant brain regions. To overcome this limitation, assembloids – generated by fusing two or more region-specific organoids – provide a complementary approach that enable the study of developmental biology, inter-organ interactions, innervation, or vascularization by combining organoids from anatomically distant but functionally connected regions.13,33–36 A recent study, for example, combined four hPSC-derived brain organoids – somatosensory, spinal, thalamic, and cortical neural organoids – to reconstruct the spinothalamic pathway, producing an interconnected system with synchronized electrical activity.37
Together, these engineering-driven advances – including defined hydrogels, dECM, bioprinting, mosaic organoids, and assembloids – are rapidly expanding the scope and physiological relevance of organoid models.38
2.3. Lack of vasculature
Nutrient renewal and oxygen distribution within the 3D structure also influence variability and growth. In static cultures, limited diffusion to the organoid core can induce central necrosis, and while continuous agitation can partially improve flow, it seems to remain insufficient to sustain long-term viability.383. A need for functional vascularization of organoids on-chip
Despite significant progress in organoids technology, achieving functional vascularization and sustained perfusion often remains reliant upon in vivo transplantation into animal models.38,58,59 However, host–graft immune incompatibility, inter-species variability, ethical considerations and high costs limit the scalability and interpretability of such approaches.6,60 Organs-on-chip systems offer a scalable, precisely controlled and ethically sound alternative, providing dynamic biochemical and biophysical cues that promote vascular integration and maturation in vitro.61Organoids and organs-on-chip constitute two fundamentally different yet complementary strategies for modelling the complexity of human physiology and pathology in vitro. Organoids excel at recapitulating developmental self-organization and multilineage architectures, whereas organs-on-chip provide precise control over cell–cell and cell-microenvironment interactions, nutrient perfusion and external mechanical and electrical stimuli, offering functional advantages over static culture systems.62,63 However, organ-on-chip models generally rely on combining pre-differentiated cells (often cell lines) to emulate native tissues through 3D matrix remodelling, and therefore frequently lack the cellular heterogeneity, phenotypic fidelity and physiologically relevant structural complexity characteristic of organoids.61 Conversely, organoids remain limited in their ability to achieve functional vascularization and to integrate effectively within dynamic flow environments.
The combination of these two approaches offers a route to overcome the limitations inherent to each individually and to unlock a synergistic strategy for developing more faithful and functional tissue models for applications in regenerative and precision medicine. As suggested previously by Takebe et al., balancing the complementary strengths of organoids and organs-on-chip – and identifying the optimal integration point – may provide a more effective strategy for generating high-fidelity, stem cell-derived in vitro organ models.2 By combining organoid self-organization and cellular complexity with the dynamic microenvironment of microfluidic platforms, hybrid systems can more faithfully reproduce human tissue physiology while also providing a scalable alternative for complementing and potentially reducing animal studies for preclinical assessments.64–66
4. Vessels-on-chip
In order to obtain vascularized organoids on-chip, the development and understanding of techniques for endothelial and vascular systems on-chip is required. These systems are commonly referred to as vessels-on-chip, indicating vessel-like culture models implemented in microfluidics, with the ultimate goal of obtaining an in vivo relevant level of vascular organization and complexity.4.1. Strategies for engineering vessels-on-chip
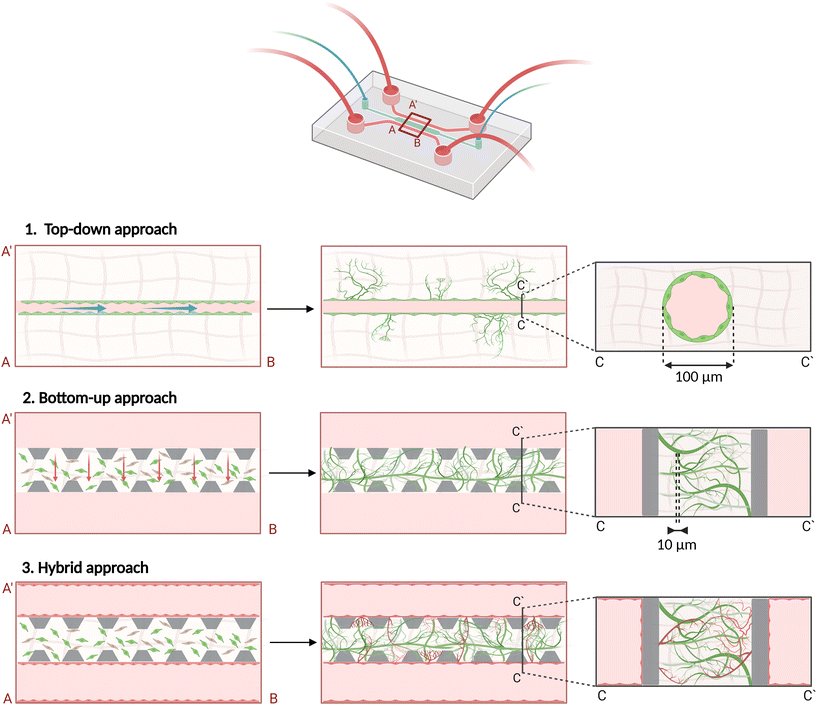
Two complementary engineering strategies have emerged to introduce a perfusable vasculature into organ-on-chip platforms: (i) a top-down, lumen-based approach in which pre-patterned conduits are endothelial-lined, and (ii) a bottom-up, self-assembly approach that lets endothelial and stromal cells build capillaries de novo,67,68 as shown in Fig. 2.The lumen patterning strategy begins by pre-defining micro-conduits whose geometry, branching and diameter are dictated by the fabrication technique. Hollow channels can be carved mechanically through microneedle withdrawal from soft biomaterials, moulded with sacrificial templates immersed in uncured hydrogel solution that are dissolved after gelation (e.g., gelatine, alginate, Pluronic, carbohydrate glass), etched by viscous-finger patterning by pumping a less viscous fluid through an uncured hydrogel, or laid down additively by 3D printing and coaxial extrusion to build multi-plane, hierarchical bifurcating trees. Once the lumen is obtained, an ECs suspension is loaded to coat with a monolayer the inner walls of the conduit, generating an instantly perfusable “macro-vessel” whose shear profile and flow rate can be predicted from the channel dimensions and the fluid viscosity.69–73 Technical advantages include precise and consistent control over vessel size and density, immediate perfusion, predictable intraluminal flow, and straightforward integration of sensors or pumps; practical constraints are (i) lumen diameters that rarely fall below 100 μm (small arteriole-scale vessels), (ii) non-circular cross-sections that disturb endothelial monolayers and create in vitro non-physiological artifacts, (iii) inconsistency in endothelializing small-sized or complex channels, (iv) limited capacity for adaptive remodelling (the vessel shape is fixed by fabrication) and (v) limited throughput due to technical challenges and manual labour.
Recent reviews catalogue rod-based luminal patterning as the archetypal “engineering-directed” strategy: geometry, branching and flow are fully determined by the artificially designed shapes and dimensions of the microchannels. This enables precise control over the morphology of the vasculature and predictable intraluminal shear stress, at the cost of a reduced biologic realism and limited micro-scale resolution.67,68
An alternative approach is biologically-driven and mimics the mechanisms of in vivo developmental vasculogenesis, the de novo formation of blood vessels from endothelial progenitor cells.74 In the self-assembled microvascular networks strategy, ECs are co-embedded with stromal cells in what is initially a random spatial distribution inside a permissive hydrogel (collagen, fibrin, Matrigel, synthetic PEG/GelMA). Guided by intrinsic programmes and extrinsic cues – interstitial and intraluminal flow, vascular endothelial growth factor (VEGF) gradients, matrix stiffness – cells migrate, align and lumenize, generating microvessel networks with lumen diameters typically in the 15–50 μm range. Although larger than physiological capillaries (<10 μm), these structures are generally described as “capillary-like” meshes in microvascular-on-chip systems.75–84 Within 3–7 days, the forming vessels can intertwine with one another and anastomose with adjacent medium channels, establishing perfusable microvascular beds that mirror key features of embryonic vasculogenesis. These networks exhibit continuous VE-cadherin junction belts, physiologically relevant permeability and dynamic responsiveness to inflammatory or metabolic stimuli. Together, these functional properties reflect the intrinsic strengths of the self-assembly approach – including robust self-organization, basement membrane deposition and ECM remodelling – while limitations include: (i) stochastic geometry and topology, (ii) reliance on mural-cell recruitment for long-term stability, (iii) gel compaction and (iv) less predictable shear stress distributions.
Together, these two strategies – and their hybrid combinations – provide a toolkit that spans macro-scale conduit precision to micro-scale capillary fidelity, enabling researchers to choose or blend methods according to the required vessel diameter range, flow control and fabrication resources. To bridge length scales, top-down arteriole-sized conduits are combined with bottom-up capillary beds in hybrid macro–micro architectures, in which self-assembled capillaries sprout in a hydrogel-filled chamber starting from endothelialized lateral medium channels.85–90 The sprouting is governed by the biological process of angiogenesis, that refers to the formation of new blood vessels from existing ones. The process is orchestrated by the migration and proliferation of ECs promoted by angiogenic signals (e.g., VEGF, Fibroblast Growth Factor FGF), degradation and remodelling of ECM by matrix metalloproteinases, formation and fusion of vascular sprouts, recruitment of support cells enhanced by angiopoietins (e.g., Platelet-derived Growth Factor PDGF) and finally vascular maturation.91 With the assistance of VEGF gradients, researchers can simulate the process of ECs degrading and invading the hydrogel chambers, yielding perfusable multi-scale networks encompassing artificially patterned macro-vessels anastomosed to self-assembled capillaries, better matching physiological hierarchy and flow distribution.85,92 These hybrid macro–micro designs offer the most promising compromise to leverage the strengths of the two approaches, and should become the default rather than the exception as they likely represent the most versatile route for engineering perfusable vasculature on-chip. In vivo, vascular beds span a broad hierarchy – from large-calibre arteries to micro-capillaries – and exhibit organ-specific endothelial cell population and mural cell functionality. While current vessel-on-chip systems can only reproduce selected aspects of this diversity, hybrid architectures still enable the controlled formation of lumenized endothelial structures and early features of microvascular specialization, providing a functional basis for modelling key components of vascular physiology in vitro.
4.2. Vessels-on-chip limitations
Building on the supportive role of fibroblasts, the addition of pericytes further enhances vascular maturation by promoting vessel stabilization, inhibiting regression, and refining lumen structure and function. Without mural support, vessels display elevated permeability, rapid regression and poor barrier protein expression, mirroring the in vivo observation that stable vasculature is always a composite of an inner endothelial tube and an outer mural layer.95 Pericytes envelop capillaries and small venules; their PDGFR-β-mediated recruitment promotes basement-membrane deposition, tight-junction maturation and long-term patency, while their contractile phenotype fine-tunes microvascular diameter and flow resistance.96,97 In the microfluidic setting, several studies co-culturing pericytes with ECs showed an overall enhancement in the barrier function.98,99 Kim et al. demonstrated that inclusion of pericytes into their biomimetic vasculature increased numbers of junctions and branches yet greatly decreased vascular permeability as well as the vascular diameter as opposed to the EC monoculture.99
Smooth-muscle cells, instead, form concentric layers around larger arterioles and veins, providing the elastic strength and vasomotor control needed to withstand pulsatile pressure and actively regulate tissue perfusion, but they are not found in capillaries in vivo. In practice, coculturing ECs with pericytes, smooth-muscle cells or mesenchymal stromal cells is essential to produce differentiated, leak-resistant and functionally responsive vascular networks that approximate native physiology of the vessel of choice.100–103
Beyond cellular composition, it is also important to note that vascular phenotypes vary substantially across organs. ECs acquire organ-specific transcriptional, metabolic and barrier properties, and mural cell diversity further contributes to tissue-specific specializations such as blood–brain barrier function, sinusoidal permeability in the liver, or fenestrated capillaries in endocrine tissues.39 Current vessel-on-chip models typically recapitulate early microvascular traits – such as lumen formation, barrier function and flow responsiveness – but only partially capture these organ-specific endothelial and perivascular identities. This incomplete specialization is partly attributable to the use of non-tissue-specific endothelial sources, such as HUVECs, which do not fully reproduce organ-dependent vascular phenotypes. Transitioning toward primary or hiPSC-derived endothelial and mural cells may therefore be necessary to achieve more faithful organ-specific microvasculature on-chip.
In addition to luminal shear stress, interstitial flow – the slow movement of fluid leaking through the ECM – plays a key role in providing directional cues that guide angiogenic sprouting and endothelial migration. Physiological interstitial flow rates (typically ranging between 0.1–10 μm s−1), beyond aiding nutrient distribution and waste removal, exert low-magnitude mechanical forces that provide directional cues, promote ECM remodelling, and enhance capillary branching and elongation.67,92,106,107 These effects arise from the activation of mechanosensory receptors such as PIEZO1, and complexes including PECAM, VE-cadherin, integrins, and VEGFR2. This activation triggers signal transduction pathways, such as the FAK pathway, which not only modulate cell phenotype but also influence the expression of key regulators of the surrounding microenvironment, such as the Matrix Metalloproteinase 1 (MMP1).6,108–111
Therefore, recreating physiological flow dynamics in microfluidic systems is essential for developing predictive and biomimetic vascularized organ-on-chip models. Flow can be introduced using various methods, including gravity-driven systems and motorized pumps. Among these, hydrostatic pressure differences or bidirectional rocking platforms are commonly used due to their simplicity, but they often generate irregular and direction-changing shear, which diverges from physiological in vivo waveforms. While under laminar flow ECs elongate and form tight junctions associated with reduced vascular permeability, turbulent flow can lead to weakened junctions and increased proinflammatory expression levels.112 More advanced approaches, such as pressure-driven, syringe-driven or peristaltic pumps have been shown to provide a more developed and unidirectional flow, allowing finer control on the flow rate and shear stress applied.113
Perfusion of fluorescent dextrans (typically 3–150 kDa) or polystyrene microbeads through the vessel network enables live imaging of tracer transit, velocity fields, and bead trajectories, confirming the presence of continuous lumens and anastomosis with inlet channels.79,85,89,116,117 These read-outs are widely accepted as gold-standard benchmarks for network perfusability; however, they lack standardization in tracer size or molecular weight, observation window duration, and data processing, limiting reproducibility and cross-study comparability.
Complementing these, diffusional permeability measurements using fluorescent tracers driven intraluminally provide a quantitative readout of barrier integrity, reflecting contributions from tight junctions, glycocalyx and mural cells interactions.115,118–120 By tracking the accumulation of fluorescence in the surrounding hydrogel over time, the effective permeability coefficient (Pe) can be calculated, though it may be overestimated due to the neglect of convective transport. Notably, Pe values measured in 3D perfusable networks have been reported up to two orders of magnitude lower than those measured in transwell systems using the same cells, and to more closely match in vivo benchmarks.4,79,83,116,121 This demonstrates that dynamic 3D conditions significantly enhance endothelial barrier performance, bringing it closer to physiological reality. Additionally, barrier transport of large macromolecules, such as albumin and therapeutic antibodies, has been assessed with paracellular permeability and transcytosis assays, providing insights into leakiness and selective transport properties.121–124 For real-time, label-free monitoring of barrier tightness, trans-endothelial electrical resistance (TEER) could be measured using integrated electrodes flanking the vessel, offering a sensitive surrogate for endothelial integrity assessment.125,126
Ultimately, a combination of both structural and functional readouts provides a comprehensive and quantitative assessment of microvascular networks maturation and barrier function.
5. Vascularization of organoids-on-chip
5.1. Considerations and approaches
Current co-culture methods begin by seeding organoids on top of a pre-established vascular bed or by embedding them in the hydrogel precursor solution (Fig. 3). The latter may contain ECs and stromal cells to exploit a vasculogenesis-like self-assembly approach, or just the organoid, to obtain vascularization through angiogenic sprouting of ECs from a pre-lined, pre-patterned lumen (Fig. 3(1)).127 Endothelial tip cells are either guided by VEGF gradients or chemokines to extrude through the matrix where the organoid is embedded in, extending towards it and potentially anastomosing with its endogenous vasculature.4,39 Vascularization of organoids-on-chip and functional anastomosis could be potentially achieved by allowing the obtained perfusable vascular network to further sprout into the organoid. A proof of concept was demonstrated in numerous studies in which perfusable vascularization was achieved in spheroid models through vascular invasion.128,129 Nashimoto et al. reported a vascularized cancer-on-a-chip model obtained by embedding a heterotypic spheroid comprising tumour cells, fibroblasts and ECs in a hydrogel solution seeded in the central chamber of a microfluidic chip.128 Under the stimulation of angiogenic factors secreted by the tumour cells, ECs lining the lateral channels sprouted through the hydrogel growing toward the spheroid. After invading into the tumour spheroids, the vessels growing from both sides anastomosed, increasing the proliferative activity of tumour cells and reducing cell death in the spheroids. Remarkably, these functional anastomoses led to a fully perfusable network across the central gel, including the spheroid, as evidenced by successful perfusion of fluorescent dextran dye into the core of the spheroid.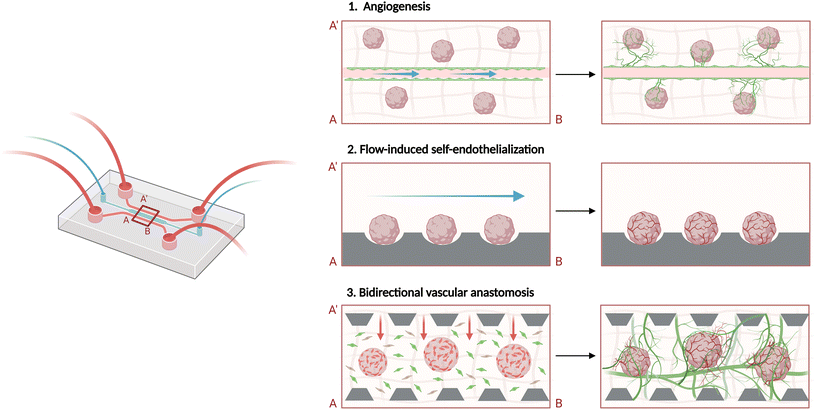 | ||
| Fig. 3 Mechanisms enabling vascular integration of organoids on-chip. (1) Angiogenesis: organoids embedded in a hydrogel adjacent to an endothelialized channel can be vascularized through angiogenic sprouting. Guided by angiogenic cues such as VEGF gradients, endothelial tip cells sprout and migrate through the matrix toward the organoid and may invade and anastomose with it, as demonstrated in vascularized tumour spheroid models.128 (2) Flow-induced self-endothelialization: controlled fluid shear stress applied on organoids can activate their endogenous endothelial precursors, stimulating the emergence of primitive intramural vessels that could grow outward once embedded into a permissive matrix.130 (3) Bidirectional vascular anastomosis: when pre-vascularized organoids are co-cultured with self-assembled microvascular networks, endothelial cells can sprout from both the organoid and the surrounding vasculature, forming reciprocal connections that generate a continuous perfusable network. Although full intramural vascular integration remains rare in lineage-specific organoids, these mechanisms illustrate the strategies currently explored to achieve functional anastomosis in organoid-on-chip systems.131 Created with Biorender. | ||
Although achieving a fully perfusable vascular system connecting an external vascular network with an internal one has been demonstrated solely in vascularized tumour spheroids, recent research indicates that organoids derived from iPSCs can also secrete VEGF, suggesting potential for vascular integration beyond tumour models.132,133 Examples of similar attempts at vascularization on-chip of parenchymal organoids have proved the vascular network to reach the vicinity of the organoid, sometimes enveloping it, but rarely extending deep into its core.131,134–136 In a recent study, Shaji et al. employed a microfluidic chip to simulate neurovascular interactions, aiming at the vascularization of hiPSC-derived cerebral organoids. The study identified the VEGF-HIF1α-AKT signalling pathway through transcriptomic analysis as a key driver of angiogenesis within the system. While vascularization was initiated, it was inhibited after 10 days of co-culture, revealing critical insights into the limitations of in vitro vascularization.131 Similarly, Schulla et al.135 developed a co-culture model to address the vascularization of human small intestinal organoids (hSIO). By integrating organoids with ECs and fibroblasts in a fibrin/Matrigel matrix, the study demonstrated that co-culture significantly enhances organoid stemness and survival compared to monoculture, even though the vascular network did not seem to vascularize the organoid itself.135 In a comparable setup, Salmon et al.136 designed a chip with spatial separation between the endothelial and organoid compartments, requiring endothelial sprouting to reach hiPSC-derived brain organoids. While ECs successfully infiltrated the hydrogel chamber and perfusion was achieved, full vascular integration into the organoid remained limited.136 Despite these major advancements, the vasculature in current organoids-on-chip models often exhibits primarily structural features and still faces challenges in achieving full functionality, such as sustained perfusion. This undermines the anticipated advantages of using vascularized organoid-on-a-chip as in vitro models for screening, given that the delivery of drugs or immune cells hinges on their transport through physiologically relevant vascular networks, mimicking the in vivo context. Nevertheless, recent developments in microfluidic design, co-culture protocols, and matrix engineering continue to push the field toward realizing this goal, and underscore the importance of addressing these challenges in future research.
In addition to supporting perfusion, the inclusion of endothelial components in on-chip models, even in the absence of a fully assembled vascular network, has also been shown to promote organoid maturation and functional refinement. Several recent studies demonstrated that their integration in co-culture systems enhances both functional and transcriptional maturation, underscoring the potential of vascular elements to drive developmental progression and improve organoid-based assays.137–139 Landau et al. notably reported that co-culturing iPSC-derived cardiomyocytes with HUVECs, dental pulp stromal cells, and primitive macrophages enhanced both vascular stabilization and cardiomyocyte contractility.137 In a gut model involving disaggregated intestinal organoids-on-chip, Ballerini et al. found that even without direct contact, the presence of EC influenced epithelial protein expression and improved tight junctions, suggesting a paracrine effect on barrier integrity.138 In another study on hiPSC-derived kidney organoids on-chip, Homan et al. demonstrated that dynamic stimulation expanded their endogenous endothelial progenitor population and led to the formation of vascular sprouts within the organoid.139 The emergence of these vascular structures supported epithelial polarity and ciliogenesis, with changes in protein expression toward mature tubule identity. Notably, the disruption of the endogenous VEGF gradient, either by VEGF addition or blockade, led to a loss of this mature phenotype, underscoring the critical role of epithelial–endothelial cross-talk. Interestingly, the interaction appears bidirectional: organoids can also enhance endothelial maturation. Co-culture systems have been shown to increase EC number, migratory capacity, and vascular markers expression. In a brain organoid model, endothelial and pericyte markers expression was upregulated compared to monoculture vascular systems,136 while in bone organoids, the stage of organoid maturity influenced vascularization through the secretion of pro- or anti- angiogenic factors.130 Similarly, Bas-Cristóbal Menéndez et al. reported that the co-culture of kidney organoids with HUVECs enhanced endothelial migration and lumen formation, yielding a higher proportion of mature (PECAM+) ECs compared to transwell conditions.140
These findings support the view that vascularization not only enables perfusion but also contributes, through both paracrine signalling and the flow-mediated biomechanical cues, to the biological relevance and maturation of organoids.
5.2. Challenges of organoids inclusion on-chip
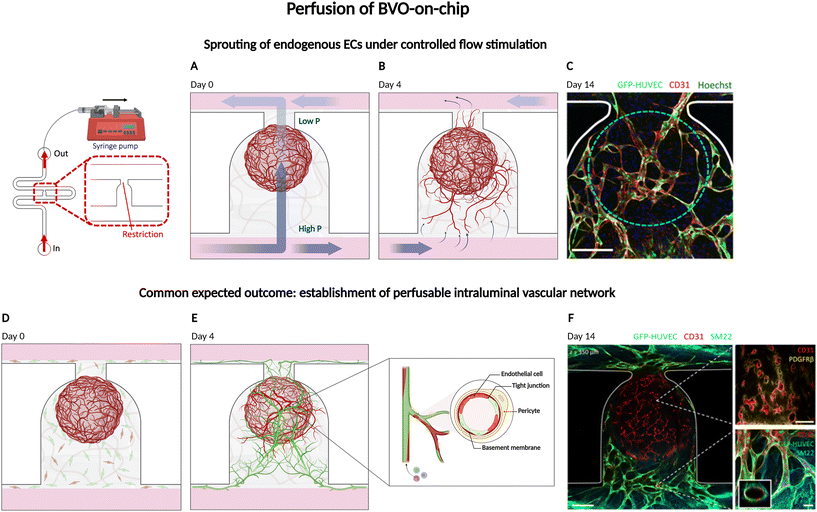 | ||
| Fig. 4 Perfusion and vascular integration of blood vessel organoids (BVOs) on-chip. A schematic view of the microfluidic device with its U-shaped hydrodynamic trap used for passive organoid positioning, is included to contextualize the organoid position and perfusion setup used in this model. (A, B, D and E) Schematic representation of BVO (in red) on U-shapped serpentine chip and (C and F) confocal images from Quintard et al. illustrating two complementary modes of vascularization. (A) Under pressure-driven interstitial flow, endogenous endothelial cells (red) sprout from the BVO core and extend into the surrounding matrix; (B and C) once established, these nascent vessels are subjected to intraluminal shear stress, which promotes remodeling and functional maturation. (D) Schematic representation of BVO seeded on-chip together with HUVECs-GFP (green) and fibroblasts (brown) for the formation of an external network. (E and F) Anastomosis of BVOs with the external endothelial network enables the generation of continuous, lumenized vessels composed of endothelial cells (CD31+, red) and perivascular support cells (SM22+/PDGFRβ+, blue). Remarkably, the hierarchical structure of vascularized BVO-on-chip emulates the arteriole–capillary–venule transition observed in vivo, underscoring their physiological relevance and exemplifying the concept of hybrid macro–micro vasculature models-on-chip. Adapted from ref. 103, created with Biorender. | ||
6. Bidirectional functional anastomosis to achieve perfusion
6.1. Perfusable mesoderm-derived organoids
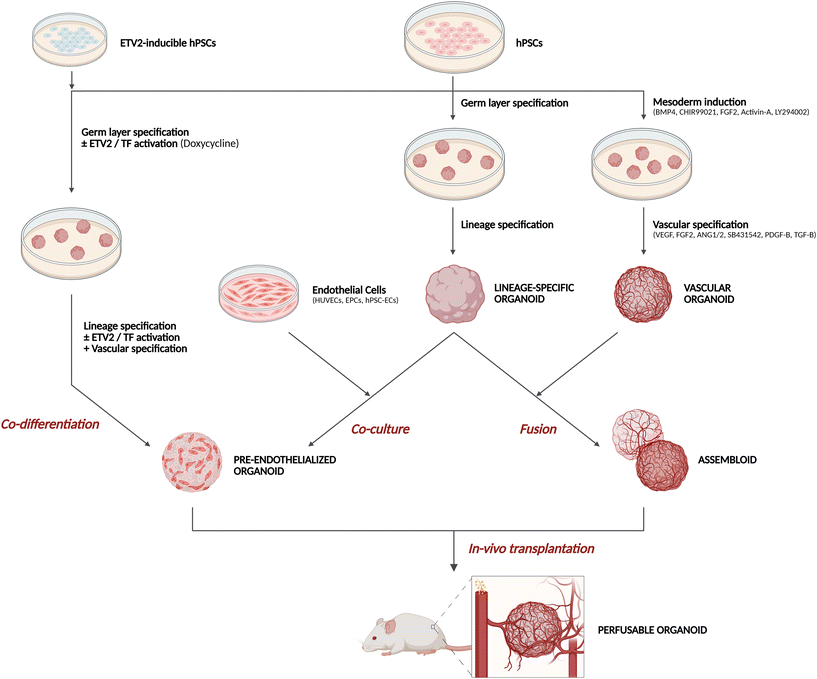
To obtain perfusion within the organoid, two complementary strategies should be combined: an inside-out mechanism should stimulate endogenous endothelial precursors to develop an internal vascular structure that sprouts outwards, while an outside-in mechanism driven by angiogenesis should promote the invasion of the organoid by the external exogenous vascular network.4 Such mechanism closely mimics the process that occurs after in vivo transplantation of pre-vascularized organoids, where host vasculature invades the graft establishing a vascularized organoid through functional connections.38,58,59 This mechanism is activated by pro-angiogenic factors, such as VEGF, secreted by the parenchymal cells within the organoid, as a natural response to the hypoxic conditions found in the core – a process that has also been observed in PSC-derived organoids.60,61,76 These endogenous signals not only promote the ingrowth of external vessels but may also enhance the success of vascularization strategies such as co-culture with ECs (primary or iPSC-derived) or the co-differentiation of vascular lineages within the organoid itself.6 The possibility to engineer the sprouting of endogenous ECs on-chip has been demonstrated by Homan et al., who showed vascularization in hPSCs-derived kidney organoids under controlled wall shear stress (Fig. 3(2)).139 Fluid stimulation and growth factors activated endogenous vascular development pathways in the organoid, stimulating the generation of ECs, thereby promoting the organoids` functional maturation and luminal structure formation. While they demonstrated that the vasculature within the organoid could embed and invade the surrounding ECM, these primordial vascular sprouts were not connected to an external vasculature for the establishment of a functional perfusable network. A follow-up study from the same group further advanced this concept by engineering a platform capable of supporting true intraluminal perfusion of kidney organoids in vitro.154 In the dual-channel device, Kroll et al. embedded two parallel lumens: an upper one loaded with kidney organoids at day 10–11 of differentiation, and a lower one endothelialized with HUVECs to generate a bioengineered perfused macrovessel. Under controlled shear stress, endogenous ECs within the organoids expanded and migrated through the ECM toward the macrovessel. A key advance of this system was the demonstration of lumen-to-lumen anastomosis between organoid-derived microvessels and the artificial HUVEC-lined channel, enabling controlled perfusion of dextran and red blood cells through the organoid microvasculature. This model therefore represents the first in vitro demonstration of a fully perfusable kidney organoid, overcoming the major limitation of the earlier Homan et al. system, where endogenous endothelial sprouts lacked connections to an external vasculature and therefore could not sustain intraluminal flow.139,154A conceptually related example of vascular integration in a mesoderm-derived system was reported by Arslan et al., with a vascularized hiPSC-derived cardiac microtissue-on-chip (Fig. 3(3)).134 In contrast to the kidney organoids described by Homan et al., where ECs arise endogenously when stimulated by shear stress to expand and sprout, the cardiac model is generated through the engineered assembly of multiple hiPSC-derived lineages – cardiomyocytes, ECs and fibroblasts – thereby incorporating a pre-existing endothelial compartment. When these pre-vascularized microtissues were embedded within a vessel-on-chip platform containing an external network formed by hiPSC-derived ECs and pericytes, the two vascular compartments underwent bidirectional integration, with both outside-in invasion of the microtissue and inside-out extension of its internal vessels into the surrounding network, giving rise to lumenized hybrid vessels that supported particle perfusion under flow. Continuous perfusion further increased vessel density and the extent of anastomosis, and the presence of a connected vascular compartment modulated cardiac contractile dynamics and inflammatory responses via endothelial–cardiomyocyte crosstalk. Although these engineered microtissues do not fulfil organoid criteria of being assembled from pre-differentiated cells rather than generated through self-organizing developmental programs, they offer a compelling demonstration of coordinated anastomosis between a pre-vascularized hiPSC-derived 3D microtissue and an external, self-organized vascular network. A complementary approach to achieve directional functional anastomosis has been demonstrated using BVOs (Fig. 4D and F).8 In the study by Quintard et al., pre-formed hiPSC-derived BVOs connected to a self-assembled external vascular bed in a microfluidic device under hemodynamic flow, thereby permitting direct tracer perfusion through the organoid's internal capillary system, indicating that intraluminal perfusion could be established upon continuity between the two vascular compartments achieved.8 Control conditions lacking the external microvascular bed did not exhibit intraluminal tracer flow, underscoring the requirement for a bidirectional connection to sustain perfusion. Extended perfusion culture for up to 30 days supported progressive vascular remodelling within the organoid, including increased network complexity, reduced hypoxia and ECM reorganization, features consistent with vascular maturation and stabilization. Transcriptomic analyses further indicated upregulation of endothelial identity and matrix-interaction pathways, while also showing downregulation of anti-angiogenic and immature endothelial markers, compared to non-perfused static controls. Although BVOs inherently contain vascular structures and therefore differ from lineage-specific organoids, this model shows that establishing physiological flow across a connected internal-external vascular interface can support the stabilization and functional maturation of organoid-derived microvessels. As such, this approach complements the mesoderm-derived models described above by providing a proof-of-principle for perfusion through a fully endothelialized organoid lumen, and also highlights the potential of combining vascular organoids with tissue-specific constructs as a route toward functional vascular integration.
Taken together, these mesoderm-derived models illustrate that functional intraluminal perfusion can be achieved when organoid-intrinsic endothelial compartments – whether endogenously specified, engineered during tissue assembly, or provided through vascular organoids – are able to establish stable bidirectional anastomoses with an external vascular network. However, lineage-specific organoids derived from endodermal or ectodermal lineages generally lack such endothelial precursors, making these mechanisms more challenging to access.
6.2. Assembloid-on-chip to vascularize lineage-specific organoids
While mesoderm-derived organoids can generate endothelial precursors that support the inside-out and outside-in mechanisms described above, most epithelial and neuroectodermal organoids lack endogenous vascular lineages.To address this limitation, several approaches have been explored in vitro. One strategy relies on co-culturing with primary or iPSC-derived EC, though integration often remains superficial and limited in depth.4,58,155,156 Alternatively, the co-differentiation of ECs demonstrated in mesoderm-derived models has proven challenging in endodermal or ectodermal due to their distinct developmental trajectories.48,157,158 Moreover, inducible genetic engineering strategies, with “reset” EC triggered by the expression of the transcription factor ETV2, can lead to the formation of vascular networks with patent lumens, yet the resulting networks typically lack the hierarchical architecture and stabilizing perivascular support required for sustained perfusion.159–161 Finally, one promising approach involves the co-culture of lineage-specific organoids with BVO, which supply a self-organizing endothelial–pericyte compartment capable of sprouting toward and ultimately penetrating the target one.57,162–165
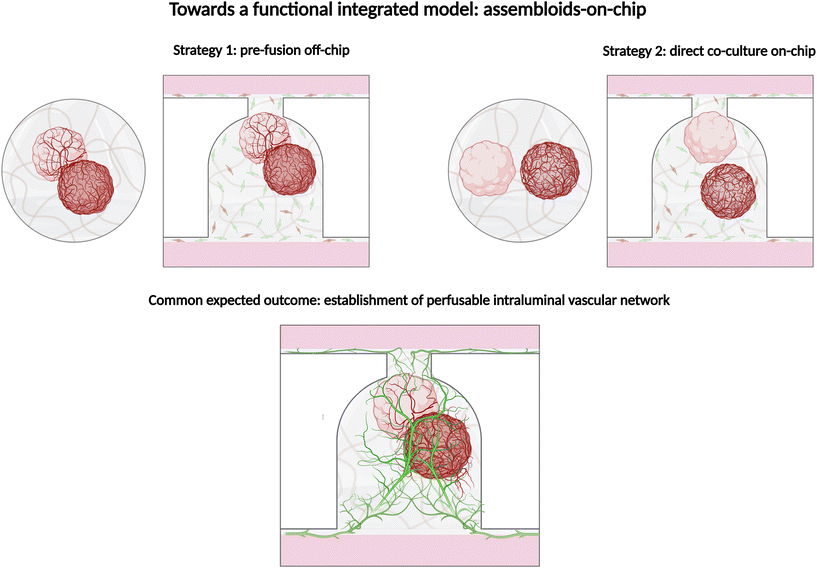
Building on these advances, we suggest that the employment of assembloids-on-chip could bridge the gap between vascular engineering in static biotechnology and the development of physiologically relevant, perfused tissue models-on-chip (Fig. 5). While lineage-specific organoids vascularization has been pursued through various in vitro strategies, their functional integration with dynamic flow environments remains limited. In contrast, Quintard et al. study8 has demonstrated that BVOs are capable of establishing functional anastomosis with external endothelial networks under flow, enabling intravascular perfusion through the organoid. This precedent provides evidence that assembloids could be successfully integrated into microfluidic systems to achieve perfusion. Extending this concept, we propose that co-culturing the target organoid with a BVO within a microfluidic platform offers a promising modular route to overcome the intrinsic vascular limitations of epithelial organoids. Two complementary implementation strategies could be envisioned. In the pre-fusion (ex-chip) approach, the BVO and target organoid are allowed to integrate prior to seeding into the chip. This ensures that internal capillaries are already contiguous with the parenchyma at the time of loading, thereby simplifying spatial control and reducing the delay in establishing perfusion, which is advantageous for minimizing hypoxic injury during early culture stages. In contrast, the direct on-chip co-culture strategy involves introducing the parenchymal organoid, the BVO and a suspension of endothelial and stromal cells into a shared hydrogel channel. Under continuous perfusion, angiogenic sprouts from both vascular components can grow toward and inosculate with the organoid, guided by the biomechanical cues of the microfluidic environment. While both approaches aim to create a continuous intraluminal connection between the organoid and the external vascular network, they differ in practical logistics: pre-fusion provides immediate vascular proximity and earlier perfusion, whereas the on-chip strategy capitalizes on microenvironmental guidance and dynamic remodelling. Depending on the organoid type, required maturation stage and experimental constraints, assembloids-on-chip offer a flexible and adaptable framework to extend perfusion beyond mesoderm-derived models and to vascularize epithelial or neuroectodermal organoids that cannot autonomously generate vasculature.
7. Discussion
Despite rapid evolution of organoid technology, a major unmet need in achieving in vivo-like functionality has been the lack of mature structural organization, which is a direct consequence of absent functional vasculature. Without perfusable vessels, organoids rapidly develop necrotic cores, as metabolic demands outpace passive diffusion. Thus, incorporating functional vasculature for active convection of nutrients and oxygen seems indispensable for organoids growth and maturation beyond their embryonic stages. To date, perfusable vasculature within organoids has only been demonstrated through transplantation into host animals, where native vasculature either invades into the ectopic implant or anastomoses with pre-vascularized organoids.58,59 Indeed, no in vitro approach has yet successfully demonstrated intravascular perfusion within lineage-specific organoids-on-chip, as most studies still rely on static diffusion or side-channel perfusion that fail to penetrate the core of the organoid. Achieving continuous and convective intraluminal flow with defined shear stress and stable barrier integrity therefore remains a major challenge, as is maintaining vascular maturity over time. Without physiological shear stimuli and cellular heterogeneity, in vitro vasculature tends to regress toward a foetal gene expression and leaky, immature phenotype. Additionally, the complex vascular architecture hinders control over continuous, directional flow both around and within the organoid.In most vascularization strategies, HUVECs remain the EC type of choice due to their accessibility and compatibility with the foetal-like phenotype of hPSC-derived organoids. Yet, their primary origin, donor age, culture history (e.g. cell passage), and donor-to-donor variability limit their suitability for long-term modelling and tissue-specific maturation.166,167 The next step toward greater physiological relevance and complexity should involve replacing primary vascular cells with iPSC-derived ECs, fibroblasts, pericytes, smooth muscle cells, and other relevant stromal components, tailored to both the organ-specific phenotype and patient genotype.168 iPSC-derived ECs offer a renewable and customizable source of vascular and mural cells, and their use would enable the creation of fully autologous, vascularized organoid systems, advancing in vitro disease modelling and drug testing in the context of precision medicine.169,170 Current personalization efforts are often limited to the epithelial compartment, as most parenchymal organoids are derived from patient-specific iPSCs.171 However, for a truly integrated personalized model, the vascular, stromal, and immune compartments should also match the patient's genotype.
To meet these personalization goals while also addressing the limitations of avascular endodermal or ectodermal organoids, vascularized assembloids-on-chip composed of patient-derived components offer a promising solution. By integrating genetically matched endothelial and stromal components into perfusable assembloids, these models may achieve both vascular fidelity and immune compatibility. Importantly, such systems extend beyond modelling vascular pathologies and are well suited to investigate disease processes in which microvascular dysfunction plays a mechanistic role, including diabetic microangiopathy (for which BVOs were originally developed), neurovascular dysfunction and blood–brain barrier impairment, tumour vascularization and metastatic extravasation.51,172–174 They could also provide a generalizable strategy to support the maturation, metabolic stability and functional performance of a wide range of lineage-specific organoids – including neural, hepatic, renal, pancreatic, intestinal and cardiac models – where sustained perfusion, controlled microenvironmental cues and vascular-parenchymal cross-talk are essential. This also opens the door to studying how organ-specific vasculature shapes tissue development, barrier properties, differentiation trajectories and drug responsiveness in a fully human context. When implemented on-chip, such platforms not only enable sustained perfusion, but also open the door to functional studies that require a dynamic vascular interface like immune cell trafficking, leukocyte adhesion and extravasation, endothelial activation under defined shear stress, and directional inter-organ communication mediated by physiologically relevant flow. They would also support a wide range of quantitative assessment of both vascular and tissue-level readouts: barrier integrity and selective permeability, angiogenic and matrix-remodelling dynamics, inflammatory and cytokine signalling, flow-dependent metabolic exchanges, endothelial mechano-transduction, and shear-induced modulation of organoid maturation and function; parameters that are difficult to access in vivo and highly relevant for disease modelling and therapeutic testing. In addition, such systems enable controlled delivery of therapeutics, nanoparticles or cytokines, allowing assessment of drug penetration, vascular transport, accumulation and toxicity in a fully human context, capabilities that are highly relevant for preclinical screening and mechanistic disease modelling. Looking ahead, moving toward interconnected and interdependent systems, multi-organoid-on-chip platforms linked by perfusable vasculature could more faithfully replicate human systemic physiology. Indeed, organoids naturally and intrinsically recapitulate much of the cellular diversity and intercellular cross-talk found in native organs, yet modelling higher-order dynamic interactions remains a significant limitation of current organoid systems. Currently, most multi-organoids-on-chip models rely on microfluidic arrays of isolated compartments connected by recirculating media rather than by functional vascular bridges.175–179 This constrains inter-organ communication to paracrine signalling (e.g., cytokine-mediated cross-talk), and fails to replicate the directional distribution of soluble factors, immune cells, and therapeutics via physiologically meaningful perfusion routes. Introducing vascular interconnections between organoids would enable circulation-like flow, essential for modelling ADME processes and systemic immune responses across multiple tissues.180 Functional vasculature would thus serve not only as a conduit for perfusion, but also as a dynamic bridge for drug delivery, immune cell trafficking, and inter-organ signalling, ultimately improving the accuracy of toxicity and efficacy assessments of personalized therapy approaches. Within the framework of the 3Rs, these platforms currently contribute primarily to reduction and refinement, by supporting mechanistic studies on human-relevant pathology and decreasing reliance on animal models, and may progressively enable partial replacement in disease areas where human-specific responses cannot be adequately recapitulated in vivo. Notably, the translational relevance of human vascularized organoid systems is beginning to be recognized at the regulatory level. Recent reports indicate that efficacy data generated using human vascularized organoid models have contributed to the first FDA Investigational New Drug (IND) acceptance supported by an in vitro vascularized organoid platform, underscoring their potential to complement or, in specific contexts, partially replace animal studies during early-stage therapeutic development.181
Realizing this vision will require compatible media formulations, optimized co-culture conditions, and strategies to mitigate metabolic waste accumulation between connected compartments, advancing the translational readiness of vascularized organoids-on-chip for future patient-specific clinical applications.182
Conclusions
Vascularised organoid-on-chip systems have passed the stage of conceptual novelty, as progress over the past years has lifted vascularized organoid technology from proof-of-concept to a promising micro-physiological test-bed, yet the field still confronts fundamental hurdles that limit fidelity and translational value. There is now a clear need for rigorous standardisation, organ-specific and patient-matched cell sources, and mechanically active microenvironments to fulfil their promise as predictive human surrogates. The next leap will come not from a single fabrication technique, but from the coordinated integration these elements into cohesive, user-friendly platforms that can exit the engineering lab and enter routine biological, pharmacological and regulatory workflows.Author contributions
BM wrote the original manuscript, and prepared the graphics. PDG wrote the introduction and provided critical input throughout the manuscript. BM, PDG, XG and ET conceptualized, edited and revised it. The work was fulfilled under XG and ET supervision.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was supported by ANR-JCJC “ATOP1” (ANR-24-CE52-7278-01), ANR MAGIC (ANR-24-EXME-0003), the CEA “OOC inflexion” and “Focus organoids”.Notes and references
- M. A. Lancaster and J. A. Knoblich, Organogenesis in a dish: Modeling development and disease using organoid technologies, Science, 2014, 345(6194), 1247125 CrossRef PubMed.
- T. Takebe, B. Zhang and M. Radisic, Synergistic Engineering: Organoids Meet Organs-on-a-Chip, Cell Stem Cell, 2017, 21(3), 297–300 CrossRef CAS PubMed.
- G. Rossi, A. Manfrin and M. P. Lutolf, Progress and potential in organoid research, Nat. Rev. Genet., 2018, 19(11), 671–687 CrossRef CAS PubMed.
- S. Zhang, Z. Wan and R. D. Kamm, Vascularized organoids on a chip: strategies for engineering organoids with functional vasculature, Lab Chip, 2021, 21(3), 473–488 RSC.
- T. Chauhdari, S. A. Zaidi, J. Su and Y. Ding, Organoids meet microfluidics: recent advancements, challenges, and future of organoids-on-chip, In Vitro Models, 2025, 4(1), 71–88 CrossRef PubMed.
- P. N. Nwokoye and O. J. Abilez, Bioengineering methods for vascularizing organoids, Cells Rep. Methods, 2024, 4(6), 100779 CrossRef CAS PubMed.
- M. Vila Cuenca, M. Bulut, C. L. Mummery and V. V. Orlova, Vascularization of organoid microenvironments: Perfusable networks for organoid growth and maturation, Curr. Opin. Biomed. Eng., 2025, 34, 100586 CrossRef CAS.
- C. Quintard, E. Tubbs, G. Jonsson, J. Jiao, J. Wang and N. Werschler, et al., A microfluidic platform integrating functional vascularized organoids-on-chip, Nat. Commun., 2024, 15(1), 1452 CrossRef CAS PubMed.
- M. Takasato, P. X. Er, H. S. Chiu, B. Maier, G. J. Baillie and C. Ferguson, et al., Kidney organoids from human iPS cells contain multiple lineages and model human nephrogenesis, Nature, 2015, 526(7574), 564–568 CrossRef CAS PubMed.
- Y. Yoshimura, Y. Muto, N. Ledru, H. Wu, K. Omachi and J. H. Miner, et al., A single-cell multiomic analysis of kidney organoid differentiation, Proc. Natl. Acad. Sci. U. S. A., 2023, 120(20), e2219699120 CrossRef CAS PubMed.
- M. Navis, T. Martins Garcia, I. B. Renes, J. L. Vermeulen, S. Meisner and M. E. Wildenberg, et al., Mouse fetal intestinal organoids: new model to study epithelial maturation from suckling to weaning, EMBO Rep., 2019, 20(2), e46221 CrossRef PubMed.
- D. Balboa, T. Barsby, V. Lithovius, J. Saarimäki-Vire, M. Omar-Hmeadi and O. Dyachok, et al., Functional, metabolic and transcriptional maturation of human pancreatic islets derived from stem cells, Nat. Biotechnol., 2022, 40(7), 1042–1055 CrossRef CAS PubMed.
- M. G. Andrews and A. R. Kriegstein, Challenges of Organoid Research, Annu. Rev. Neurosci., 2022, 45, 23–39 CrossRef CAS PubMed.
- O. Revah, F. Gore, K. W. Kelley, J. Andersen, N. Sakai and X. Chen, et al., Maturation and circuit integration of transplanted human cortical organoids, Nature, 2022, 610(7931), 319–326 CrossRef CAS PubMed.
- C. J. Childs, H. M. Poling, K. Chen, Y. H. Tsai, A. Wu and A. Vallie, et al., Coordinated differentiation of human intestinal organoids with functional enteric neurons and vasculature, Cell Stem Cell, 2025, 32(4), 640–651.e9 CrossRef CAS PubMed.
- A. Malakpour-Permlid, I. Buzzi, C. Hegardt, F. Johansson and S. Oredsson, Identification of extracellular matrix proteins secreted by human dermal fibroblasts cultured in 3D electrospun scaffolds, Sci. Rep., 2021, 11(1), 6655 CrossRef CAS PubMed.
- C. S. Hughes, L. M. Postovit and G. A. Lajoie, Matrigel: A complex protein mixture required for optimal growth of cell culture, Proteomics, 2010, 10(9), 1886–1890 CrossRef CAS PubMed.
- J. H. Heo, D. Kang, S. J. Seo and Y. Jin, Engineering the Extracellular Matrix for Organoid Culture, Int. J. Stem Cells, 2022, 15(1), 60–69 CrossRef CAS PubMed.
- C. Li, N. An, Q. Song, Y. Hu, W. Yin and Q. Wang, et al., Enhancing organoid culture: harnessing the potential of decellularized extracellular matrix hydrogels for mimicking microenvironments, J. Biomed. Sci., 2024, 31(1), 96 CrossRef PubMed.
- A. N. Cho, Y. Jin, Y. An, J. Kim, Y. S. Choi and J. S. Lee, et al., Microfluidic device with brain extracellular matrix promotes structural and functional maturation of human brain organoids, Nat. Commun., 2021, 12(1), 4730 CrossRef CAS PubMed.
- M. Kim, S. Cho, D. G. Hwang, I. K. Shim, S. C. Kim and J. Jang, et al., Bioprinting of bespoke islet-specific niches to promote maturation of stem cell-derived islets, Nat. Commun., 2025, 16(1), 1430 CrossRef CAS PubMed.
- S. Dauth, T. Grevesse, H. Pantazopoulos, P. H. Campbell, B. M. Maoz and S. Berretta, et al., Extracellular Matrix Protein Expression Is Brain Region Dependent, J. Comp. Neurol., 2016, 524(7), 1309–1336 CrossRef PubMed.
- D. Reginensi, D. A. Ortiz, B. Denis, S. Castillo, A. Burillo and N. Khoury, et al., Region-specific brain decellularized extracellular matrix promotes cell recovery in an in vitro model of stroke, Sci. Rep., 2025, 15(1), 11921 CrossRef CAS PubMed.
- W. Lai, H. Geliang, X. Bin and W. Wang, Effects of hydrogel stiffness and viscoelasticity on organoid culture: a comprehensive review, Mol. Med., 2025, 31(1), 83 CAS.
- X. Xie, X. Chen, J. Zhou, T. Wang, G. Yang and F. Han, et al., Dynamic Hydrogels with Tunable Mechanics for 3D Organoid Derivation, Small, 2025, 21(29), 2501862 CrossRef CAS PubMed.
- I. Krupa, N. J. Treacy, S. Clerkin, J. L. Davis, A. F. Miller and A. Saiani, et al., Protocol for the Growth and Maturation of hiPSC-Derived Kidney Organoids using Mechanically Defined Hydrogels, Curr. Protoc., 2024, 4(7), e1096 CrossRef CAS PubMed.
- M. D. A. Norman, S. A. Ferreira, G. M. Jowett, L. Bozec and E. Gentleman, Measuring the elastic modulus of soft culture surfaces and 3D hydrogels using atomic force microscopy, Nat. Protoc., 2021, 16(5), 2418–2449 CrossRef CAS PubMed.
- A. Bhattacharya, K. Alam, N. S. Roy, K. Kaur, S. Kaity and V. Ravichandiran, et al., Exploring the interaction between extracellular matrix components in a 3D organoid disease model to replicate the pathophysiology of breast cancer, J. Exp. Clin. Cancer Res., 2023, 42(1), 343 CrossRef PubMed.
- Y. Jun, K. V. Nguyen-Ngoc, S. Sai, R. H. F. Bender, W. Gong, V. Kravets, H. Zhu, C. J. Hatch, M. Schlichting, R. Gaetani, M. Mallick, S. J. Hachey, K. L. Christman, S. C. George, C. C. W. Hughes and M. Sander, Engineered vasculature induces functional maturation of pluripotent stem cell-derived islet organoids, Dev. Cell, 2025, 60(18), 2455–2469.e7 CrossRef CAS PubMed.
- L. Elomaa, M. Lindner, R. Leben, R. Niesner and M. Weinhart, In vitro vascularization of hydrogel-based tissue constructs via a combined approach of cell sheet engineering and dynamic perfusion cell culture, Biofabrication, 2022, 15(1) DOI:10.1088/1758-5090/ac9433.
- K. K. Dijkstra, C. M. Cattaneo, F. Weeber, M. Chalabi, J. van de Haar and L. F. Fanchi, et al., Generation of Tumor-Reactive T Cells by Co-culture of Peripheral Blood Lymphocytes and Tumor Organoids, Cell, 2018, 174(6), 1586–1598.e12 CrossRef CAS PubMed.
- E. Fiorini, L. Veghini and V. Corbo, Modeling Cell Communication in Cancer With Organoids: Making the Complex Simple, Front. Cell Dev. Biol., 2020, 8, 166 CrossRef PubMed.
- M. M. Onesto, J. Kim and S. P. Pasca, Assembloid models of cell-cell interaction to study tissue and disease biology, Cell Stem Cell, 2024, 31(11), 1563–1573 CrossRef CAS PubMed.
- H. Koike, K. Iwasawa, R. Ouchi, M. Maezawa, M. Kimura and A. Kodaka, et al., Engineering human hepato-biliary-pancreatic organoids from pluripotent stem cells, Nat. Protoc., 2021, 16(2), 919 CrossRef CAS PubMed.
- L. Dao, Z. You, L. Lu, T. Xu, A. K. Sarkar and H. Zhu, et al., Modeling blood-brain barrier formation and cerebral cavernous malformations in human PSC-derived organoids, Cell Stem Cell, 2024, 31(6), 818–833.e11 CrossRef CAS PubMed.
- L. Rossi, B. Auletta, L. Sartore, M. La Placa, G. Cecconi and P. Chiolerio, et al., Engineering Assembloids to Mimic Graft-Host Skeletal Muscle Interaction, Adv. Healthcare Mater., 2025, 2404111 CrossRef CAS PubMed.
- J. il Kim, K. Imaizumi, O. Jurjuţ, K. W. Kelley, D. Wang and M. V. Thete, et al., Human assembloid model of the ascending neural sensory pathway, Nature, 2025, 1–11 Search PubMed.
- E. Garreta, R. D. Kamm, C. de Sousa, S. M. Lopes, M. A. Lancaster, R. Weiss and X. Trepat, et al., Rethinking organoid technology through bioengineering, Nat. Mater., 2021, 20(2), 145–155 CrossRef CAS PubMed.
- E. Trimm and K. Red-Horse, Vascular endothelial cell development and diversity, Nat. Rev. Cardiol., 2023, 20(3), 197–210 CrossRef PubMed.
- R. Craig-Schapiro, G. Li, K. Chen, J. M. Gomez-Salinero, R. Nachman and A. Kopacz, et al., Single-cell atlas of human pancreatic islet and acinar endothelial cells in health and diabetes, Nat. Commun., 2025, 16(1), 1338 CrossRef CAS PubMed.
- Y. Shi, L. Sun, M. Wang, J. Liu, S. Zhong and R. Li, et al., Vascularized human cortical organoids (vOrganoids) model cortical development in vivo, PLoS Biol., 2020, 18(5), e3000705 CrossRef CAS PubMed.
- H. A. Strobel, S. M. Moss and J. B. Hoying, Vascularized Tissue Organoids, Bioengineering, 2023, 10(2), 124 CrossRef CAS PubMed.
- S. Lee, C. Park, J. W. Han, J. Y. Kim, K. Cho and E. J. Kim, et al., Direct Reprogramming of Human Dermal Fibroblasts Into Endothelial Cells Using ER71/ETV2, Circ. Res., 2017, 120(5), 848–861 CrossRef CAS PubMed.
- B. Palikuqi, D. H. T. Nguyen, G. Li, R. Schreiner, A. F. Pellegata and Y. Liu, et al., Adaptable haemodynamic endothelial cells for organogenesis and tumorigenesis, Nature, 2020, 585(7825), 426–432 CrossRef CAS PubMed.
- S. Park and S. W. Cho, Bioengineering toolkits for potentiating organoid therapeutics, Adv. Drug Delivery Rev., 2024, 208, 115238 CrossRef CAS PubMed.
- G. Li, R. Craig-Schapiro, D. Redmond, K. Chen, Y. Lin and F. Geng, et al., Vascularization of human islets by adaptable endothelium for durable and functional subcutaneous engraftment, Sci. Adv., 2025, 11(5), eadq5302 CrossRef CAS PubMed.
- O. J. Abilez, H. Yang, Y. Guan, M. Shen, Z. Yildirim and Y. Zhuge, et al., Gastruloids enable modeling of the earliest stages of human cardiac and hepatic vascularization, Science, 2025, 388(6751), eadu9375 CrossRef CAS PubMed.
- Y. Miao, N. M. Pek, C. Tan, C. Jiang, Z. Yu and K. Iwasawa, et al., Co-development of mesoderm and endoderm enables organotypic vascularization in lung and gut organoids, Cell, 2025, 188(16), 4295–4313.e27 CrossRef CAS PubMed.
- S. Mallapaty, Mini hearts, lungs and livers made in lab now grow their own blood vessels, Nature, 2025, 643(8073), 892–892 CrossRef CAS PubMed.
- N. Werschler and J. Penninger, Generation of Human Blood Vessel Organoids from Pluripotent Stem Cells, J. Visualized Exp., 2023, 191, 64715 Search PubMed.
- R. A. Wimmer, A. Leopoldi, M. Aichinger, N. Wick, B. Hantusch and M. Novatchkova, et al., Human blood vessel organoids as a model of diabetic vasculopathy, Nature, 2019, 565(7740), 505–510 CrossRef CAS PubMed.
- R. A. Wimmer, A. Leopoldi, M. Aichinger, D. Kerjaschki and J. M. Penninger, Generation of blood vessel organoids from human pluripotent stem cells, Nat. Protoc., 2019, 14(11), 3082–3100 CrossRef CAS PubMed.
- M. T. Nikolova, Z. He, M. Seimiya, G. Jonsson, W. Cao and R. Okuda, et al., Fate and state transitions during human blood vessel organoid development, Cell, 2025, 188(12), 3329–3348.e31 CrossRef CAS PubMed.
- P. Wörsdörfer, N. Dalda, A. Kern, S. Krüger, N. Wagner and C. K. Kwok, et al., Generation of complex human organoid models including vascular networks by incorporation of mesodermal progenitor cells, Sci. Rep., 2019, 9(1), 15663 CrossRef PubMed.
- E. Kim, S. Choi, B. Kang, J. Kong, Y. Kim and W. H. Yoon, et al., Creation of bladder assembloids mimicking tissue regeneration and cancer, Nature, 2020, 588(7839), 664–669 CrossRef CAS PubMed.
- L. Nalbach, L. P. Roma, B. M. Schmitt, V. Becker, C. Körbel and S. Wrublewsky, et al., Improvement of islet transplantation by the fusion of islet cells with functional blood vessels, EMBO Mol. Med., 2021, 13(1), e12616 CrossRef CAS PubMed.
- E. Tubbs, M. Mehanović, M. Lopes, C. Quintard, S. Combe and C. Pirlian, et al., Human assembloid of human blood vessel organoids with pancreatic islets improves insulin secretion over time ex vivo, Cell Rep., 2025, 44(10), 116378 CrossRef CAS PubMed.
- T. Takebe, K. Sekine, M. Enomura, H. Koike, M. Kimura and T. Ogaeri, et al., Vascularized and functional human liver from an iPSC-derived organ bud transplant, Nature, 2013, 499(7459), 481–484 CrossRef CAS PubMed.
- A. A. Mansour, J. T. Gonçalves, C. W. Bloyd, H. Li, S. Fernandes and D. Quang, et al., An in vivo model of functional and vascularized human brain organoids, Nat. Biotechnol., 2018, 36(5), 432–441 CrossRef CAS PubMed.
- O. J. Abilez, Developing advanced organoids: challenges, progress, and outlook, BioTechniques, 2024, 76(12), 575–580 CrossRef CAS PubMed.
- S. N. Bhatia and D. E. Ingber, Microfluidic organs-on-chips, Nat. Biotechnol., 2014, 32(8), 760–772 CrossRef CAS PubMed.
- M. Hofer and M. P. Lutolf, Engineering organoids, Nat. Rev. Mater., 2021, 6(5), 402–420 CrossRef CAS PubMed.
- T. Takebe and J. M. Wells, Organoids by design, Science, 2019, 364(6444), 956–959 CrossRef CAS PubMed.
- A. Polini, L. Prodanov, N. S. Bhise, V. Manoharan, M. R. Dokmeci and A. Khademhosseini, Organs-on-a-chip: a new tool for drug discovery, Expert Opin. Drug Discovery, 2014, 9(4), 335–352 CrossRef CAS PubMed.
- E. W. Esch, A. Bahinski and D. Huh, Organs-on-chips at the frontiers of drug discovery, Nat. Rev. Drug Discovery, 2015, 14(4), 248–260 CrossRef CAS PubMed.
- D. Huh, G. A. Hamilton and D. E. Ingber, From 3D cell culture to organs-on-chips, Trends Cell Biol., 2011, 21(12), 745–754 CrossRef CAS PubMed.
- V. S. Shirure, C. C. W. Hughes and S. C. George, Engineering Vascularized Organoid-on-a-Chip Models, Annu. Rev. Biomed. Eng., 2021, 23(1), 141–167 CrossRef CAS PubMed.
- Z. Li, D. Yu, C. Zhou, F. Wang, K. Lu, Y. Liu, J. Xu, L. Xuan and X. Wang, Engineering vascularised organoid-on-a-chip: strategies, advances and future perspectives, Biomater. Transl., 2024, 5(1), 21–32 CrossRef PubMed.
- K. M. Chrobak, D. R. Potter and J. Tien, Formation of perfused, functional microvascular tubes in vitro, Microvasc. Res., 2006, 71(3), 185–196 CrossRef CAS PubMed.
- Y. Zheng, J. Chen, M. Craven, N. W. Choi, S. Totorica and A. Diaz-Santana, et al., In vitro microvessels for the study of angiogenesis and thrombosis, Proc. Natl. Acad. Sci. U. S. A., 2012, 109(24), 9342–9347 CrossRef CAS PubMed.
- L. L. Bischel, E. W. K. Young, B. R. Mader and D. J. Beebe, Tubeless microfluidic angiogenesis assay with three-dimensional endothelial-lined microvessels, Biomaterials, 2013, 34(5), 1471–1477 CrossRef CAS PubMed.
- J. S. Miller, K. R. Stevens, M. T. Yang, B. M. Baker, D. H. T. Nguyen and D. M. Cohen, et al., Rapid casting of patterned vascular networks for perfusable engineered three-dimensional tissues, Nat. Mater., 2012, 11(9), 768–774 CrossRef CAS PubMed.
- S. S. Verbridge, A. Chakrabarti, P. DelNero, B. Kwee, J. D. Varner and A. D. Stroock, et al., Physicochemical regulation of endothelial sprouting in a 3D microfluidic angiogenesis model, J. Biomed. Mater. Res., Part A, 2013, 101(10), 2948–2956 CrossRef PubMed.
- W. Risau and I. Flamme, Vasculogenesis, Annu. Rev. Cell Dev. Biol., 1995, 11(1), 73–91 CrossRef CAS PubMed.
- A. Sobrino, D. T. T. Phan, R. Datta, X. Wang, S. J. Hachey and M. Romero-López, et al., 3D microtumors in vitro supported by perfused vascular networks, Sci. Rep., 2016, 6(1), 31589 CrossRef CAS PubMed.
- V. S. Shirure, Y. Bi, M. B. Curtis, A. Lezia, M. M. Goedegebuure and S. P. Goedegebuure, et al., Tumor-on-a-chip platform to investigate progression and drug sensitivity in cell lines and patient-derived organoids, Lab Chip, 2018, 18(23), 3687–3702 RSC.
- M. L. Moya, Y. H. Hsu, A. P. Lee, C. C. W. Hughes and S. C. George, In Vitro Perfused Human Capillary Networks, Tissue Eng., Part C, 2013, 19(9), 730–737 CrossRef CAS PubMed.
- J. S. Jeon, S. Bersini, M. Gilardi, G. Dubini, J. L. Charest and M. Moretti, et al., Human 3D vascularized organotypic microfluidic assays to study breast cancer cell extravasation, Proc. Natl. Acad. Sci. U. S. A., 2015, 112(1), 214–219 CrossRef CAS PubMed.
- S. Kim, H. Lee, M. Chung and N. L. Jeon, Engineering of functional, perfusable 3D microvascular networks on a chip, Lab Chip, 2013, 13(8), 1489 RSC.
- T. Osaki, V. Sivathanu and R. D. Kamm, Engineered 3D vascular and neuronal networks in a microfluidic platform, Sci. Rep., 2018, 8(1), 5168 CrossRef PubMed.
- M. B. Chen, J. A. Whisler, J. Fröse, C. Yu, Y. Shin and R. D. Kamm, On-chip human microvasculature assay for visualization and quantification of tumor cell extravasation dynamics, Nat. Protoc., 2017, 12(5), 865–880 CrossRef CAS PubMed.
- E. Sano, C. Mori, Y. Nashimoto, R. Yokokawa, H. Kotera and Y.-S. Torisawa, Engineering of vascularized 3D cell constructs to model cellular interactions through a vascular network, Biomicrofluidics, 2018, 12(4), 042204 CrossRef PubMed.
- M. B. Chen, J. A. Whisler, J. S. Jeon and R. D. Kamm, Mechanisms of tumor cell extravasation in an in vitro microvascular network platform, Integr. Biol., 2013, 5(10), 1262 CrossRef CAS PubMed.
- J. A. Whisler, M. B. Chen and R. D. Kamm, Control of Perfusable Microvascular Network Morphology Using a Multiculture Microfluidic System, Tissue Eng., Part C, 2014, 20(7), 543–552 CrossRef CAS PubMed.
- S. W. Chen, A. Blazeski, S. Zhang, S. E. Shelton, G. S. Offeddu and R. D. Kamm, Development of a perfusable, hierarchical microvasculature-on-a-chip model, Lab Chip, 2023, 23(20), 4552–4564 RSC.
- J. Paek, S. E. Park, Q. Lu, K. T. Park, M. Cho and J. M. Oh, et al., Microphysiological Engineering of Self-Assembled and Perfusable Microvascular Beds for the Production of Vascularized Three-Dimensional Human Microtissues, ACS Nano, 2019, 13(7), 7627–7643 CrossRef CAS PubMed.
- D. H. T. Nguyen, S. C. Stapleton, M. T. Yang, S. S. Cha, C. K. Choi and P. A. Galie, et al., Biomimetic model to reconstitute angiogenic sprouting morphogenesis in vitro, Proc. Natl. Acad. Sci. U. S. A., 2013, 110(17), 6712–6717 CrossRef CAS PubMed.
- V. K. Lee, A. M. Lanzi, H. Ngo, S. S. Yoo, P. A. Vincent and G. Dai, Generation of Multi-scale Vascular Network System Within 3D Hydrogel Using 3D Bio-printing Technology, Cell. Mol. Bioeng., 2014, 7(3), 460–472 CrossRef CAS PubMed.
- X. Wang, D. T. T. Phan, A. Sobrino, S. C. George, C. C. W. Hughes and A. P. Lee, Engineering anastomosis between living capillary networks and endothelial cell-lined microfluidic channels, Lab Chip, 2016, 16(2), 282–290 RSC.
- H. G. Song, A. Lammers, S. Sundaram, L. Rubio, A. X. Chen and L. Li, et al., Transient Support from Fibroblasts is Sufficient to Drive Functional Vascularization in Engineered Tissues, Adv. Funct. Mater., 2020, 30(48), 2003777 CrossRef CAS PubMed.
- P. Carmeliet and R. K. Jain, Molecular mechanisms and clinical applications of angiogenesis, Nature, 2011, 473(7347), 298–307 CrossRef CAS PubMed.
- S. Kim, M. Chung, J. Ahn, S. Lee and N. L. Jeon, Interstitial flow regulates the angiogenic response and phenotype of endothelial cells in a 3D culture model, Lab Chip, 2016, 16(21), 4189–4199 RSC.
- A. C. Newman, M. N. Nakatsu, W. Chou, P. D. Gershon and C. C. W. Hughes, The requirement for fibroblasts in angiogenesis: fibroblast-derived matrix proteins are essential for endothelial cell lumen formation, Mol. Biol. Cell, 2011, 22(20), 3791–3800 CrossRef CAS PubMed.
- C. C. Hughes, Endothelial-stromal interactions in angiogenesis, Curr. Opin. Hematol., 2008, 15(3), 204–209 CrossRef PubMed.
- A. N. Stratman and G. E. Davis, Endothelial Cell-Pericyte Interactions Stimulate Basement Membrane Matrix Assembly: Influence on Vascular Tube Remodeling, Maturation, and Stabilization, Microsc. Microanal., 2012, 18(1), 68–80 CrossRef CAS PubMed.
- M. Sweeney and G. Foldes, It Takes Two: Endothelial-Perivascular Cell Cross-Talk in Vascular Development and Disease, Front. Cardiovasc. Med., 2018, 5, 154 CrossRef CAS PubMed.
- A. L. Gonzales, N. R. Klug, A. Moshkforoush, J. C. Lee, F. K. Lee and B. Shui, et al., Contractile pericytes determine the direction of blood flow at capillary junctions, Proc. Natl. Acad. Sci. U. S. A., 2020, 117(43), 27022–27033 CrossRef CAS PubMed.
- K. Fujimoto, S. Erickson, M. Nakayama, H. Ihara, K. Sugihara and Y. Nashimoto, et al., Pericytes and shear stress each alter the shape of a self-assembled vascular network, Lab Chip, 2023, 23(2), 306–317 RSC.
- J. Kim, M. Chung, S. Kim, D. H. Jo, J. H. Kim and N. L. Jeon, Engineering of a Biomimetic Pericyte-Covered 3D Microvascular Network, PLoS One, 2015, 10(7), e0133880 Search PubMed.
- N. Takakura, Role of intimate interactions between endothelial cells and the surrounding accessory cells in the maturation of blood vessels, J. Thromb. Haemostasis, 2011, 9, 144–150 CrossRef CAS PubMed.
- N. Koike, D. Fukumura, O. Gralla, P. Au, J. S. Schechner and R. K. Jain, Creation of long-lasting blood vessels, Nature, 2004, 428(6979), 138–139 CrossRef CAS PubMed.
- B. Andrée, H. Ichanti, S. Kalies, A. Heisterkamp, S. Strauß and P. M. Vogt, et al., Formation of three-dimensional tubular endothelial cell networks under defined serum-free cell culture conditions in human collagen hydrogels, Sci. Rep., 2019, 9(1), 5437 CrossRef PubMed.
- T. Takebe, N. Koike, K. Sekine, M. Enomura, Y. Chiba and Y. Ueno, et al. Generation of Functional Human Vascular Network, Transplant. Proc., 2012, 44(4), 1130–1133 CrossRef CAS PubMed.
- K. Haase and R. D. Kamm, Advances in On-Chip Vascularization, Regener. Med., 2017, 12(3), 285–302 CrossRef CAS PubMed.
- S. S. S. Aye, Z. Fang, M. C. L. Wu, K. S. Lim and L. A. Ju, Integrating microfluidics, hydrogels, and 3D bioprinting for personalized vessel-on-a-chip platforms, Biomater. Sci., 2025, 13(5), 1131–1160 RSC.
- V. S. Shirure, A. Lezia, A. Tao, L. F. Alonzo and S. C. George, Low levels of physiological interstitial flow eliminate morphogen gradients and guide angiogenesis, Angiogenesis, 2017, 20(4), 493–504 CrossRef CAS PubMed.
- Y. H. Hsu, M. L. Moya, P. Abiri, C. C. W. Hughes, S. C. George and A. P. Lee, Full range physiological mass transport control in 3D tissue cultures, Lab Chip, 2013, 13(1), 81–89 RSC.
- P. A. Galie, D. H. T. Nguyen, C. K. Choi, D. M. Cohen, P. A. Janmey and C. S. Chen, Fluid shear stress threshold regulates angiogenic sprouting, Proc. Natl. Acad. Sci. U. S. A., 2014, 111(22), 7968–7973 CrossRef CAS PubMed.
- W. J. Polacheck, A. E. German, A. Mammoto, D. E. Ingber and R. D. Kamm, Mechanotransduction of fluid stresses governs 3D cell migration, Proc. Natl. Acad. Sci. U. S. A., 2014, 111(7), 2447–2452 CrossRef CAS PubMed.
- A. Lai, C. D. Cox, N. Chandra Sekar, P. Thurgood, A. Jaworowski and K. Peter, et al., Mechanosensing by Piezo1 and its implications for physiology and various pathologies, Biol. Rev., 2022, 97(2), 604–614 CrossRef CAS PubMed.
- T. Fu, D. P. Sullivan, A. M. Gonzalez, M. E. Haynes, P. J. Dalal and N. S. Rutledge, et al. Mechanotransduction via endothelial adhesion molecule CD31 initiates transmigration and reveals a role for VEGFR2 in diapedesis, Immunity, 2023, 56(10), 2311–2324.e6 CrossRef CAS PubMed.
- C. J. Mandrycky, C. C. Howard, S. G. Rayner, Y. J. Shin and Y. Zheng, Organ-on-a-chip systems for vascular biology, J. Mol. Cell. Cardiol., 2021, 159, 1–13 CrossRef CAS PubMed.
- A. Martier, Z. Chen, H. Schaps, M. J. Mondrinos and J. S. Fang, Capturing physiological hemodynamic flow and mechanosensitive cell signaling in vessel-on-a-chip platforms, Front. Physiol., 2024, 15, 1425618 CrossRef CAS PubMed.
- Y. Xiao, C. Liu, Z. Chen, M. R. Blatchley, D. Kim and J. Zhou, et al., Senescent Cells with Augmented Cytokine Production for Microvascular Bioengineering and Tissue Repairs, Adv. Biosyst., 2019, 3(8), 1900089 CrossRef PubMed.
- M. Campisi, Y. Shin, T. Osaki, C. Hajal, V. Chiono and R. D. Kamm, 3D self-organized microvascular model of the human blood-brain barrier with endothelial cells, pericytes and astrocytes, Biomaterials, 2018, 180, 117–129 CrossRef CAS PubMed.
- C. Hajal, G. S. Offeddu, Y. Shin, S. Zhang, O. Morozova and D. Hickman, et al., Engineered human blood–brain barrier microfluidic model for vascular permeability analyses, Nat. Protoc., 2022, 17(1), 95–128 Search PubMed.
- Z. Wan, A. X. Zhong, S. Zhang, G. Pavlou, M. F. Coughlin and S. E. Shelton, et al., A Robust Method for Perfusable Microvascular Network Formation In Vitro, Small Methods, 2022, 6(6), 2200143 Search PubMed.
- J. Song, H. Choi, S. K. Koh, D. Park, J. Yu and H. Kang, et al., High-Throughput 3D In Vitro Tumor Vasculature Model for Real-Time Monitoring of Immune Cell Infiltration and Cytotoxicity, Front. Immunol., 2021, 12, 733317 CrossRef CAS PubMed.
- C. G. M. Van Dijk, M. M. Brandt, N. Poulis, J. Anten, M. Van Der Moolen and L. Kramer, et al., A new microfluidic model that allows monitoring of complex vascular structures and cell interactions in a 3D biological matrix, Lab Chip, 2020, 20(10), 1827–1844 Search PubMed.
- S. J. Grainger and A. J. Putnam, Assessing the Permeability of Engineered Capillary Networks in a 3D Culture, PLoS One, 2011, 6(7), e22086 CrossRef CAS PubMed.
- G. S. Offeddu, K. Haase, M. R. Gillrie, R. Li, O. Morozova and D. Hickman, et al., An on-chip model of protein paracellular and transcellular permeability in the microcirculation, Biomaterials, 2019, 212, 115–125 CrossRef CAS PubMed.
- G. S. Offeddu, L. Possenti, J. T. Loessberg-Zahl, P. Zunino, J. Roberts and X. Han, et al., Application of Transmural Flow Across In Vitro Microvasculature Enables Direct Sampling of Interstitial Therapeutic Molecule Distribution, Small, 2019, 15(46), 1902393 CrossRef CAS PubMed.
- G. Offeddu, et al., Microphysiological endothelial models to characterize subcutaneous drug absorption, ALTEX, 2023, 40(2) DOI:10.14573/altex.2207131.
- M. B. Chen, J. M. Lamar, R. Li, R. O. Hynes and R. D. Kamm, Elucidation of the Roles of Tumor Integrin β1 in the Extravasation Stage of the Metastasis Cascade, Cancer Res., 2016, 76(9), 2513–2524 CrossRef CAS PubMed.
- P. Zoio, S. Lopes-Ventura and A. Oliva, Barrier-on-a-Chip with a Modular Architecture and Integrated Sensors for Real-Time Measurement of Biological Barrier Function, Micromachines, 2021, 12(7), 816 CrossRef PubMed.
- M. W. Van Der Helm, M. Odijk, J. P. Frimat, A. D. Van Der Meer, J. C. T. Eijkel and A. Van Den Berg, et al., Direct quantification of transendothelial electrical resistance in organs-on-chips, Biosens. Bioelectron., 2016, 85, 924–929 CrossRef CAS PubMed.
- T. J. Kwak and E. Lee, In vitro modeling of solid tumor interactions with perfused blood vessels, Sci. Rep., 2020, 10(1), 20142 CrossRef CAS PubMed.
- Y. Nashimoto, R. Okada, S. Hanada, Y. Arima, K. Nishiyama and T. Miura, et al., Vascularized cancer on a chip: The effect of perfusion on growth and drug delivery of tumor spheroid, Biomaterials, 2020, 229, 119547 CrossRef CAS PubMed.
- Y. Nashimoto, T. Hayashi, I. Kunita, A. Nakamasu, Y.-S. Torisawa and M. Nakayama, et al. Integrating perfusable vascular networks with a three-dimensional tissue in a microfluidic device, Integr. Biol., 2017, 9(6), 506–518 Search PubMed.
- I. T. Whelan, R. Burdis, S. Shahreza, E. Moeendarbary, D. A. Hoey and D. J. Kelly, A microphysiological model of bone development and regeneration, Biofabrication, 2023, 15(3), 034103 CrossRef CAS PubMed.
- M. Shaji, A. Tamada, K. Fujimoto, K. Muguruma, S. L. Karsten and R. Yokokawa, Deciphering potential vascularization factors of on-chip co-cultured hiPSC-derived cerebral organoids, Lab Chip, 2024, 24(4), 680–696 RSC.
- C. W. Van Den Berg, L. Ritsma, M. C. Avramut, L. E. Wiersma, B. M. Van Den Berg and D. G. Leuning, et al., Renal Subcapsular Transplantation of PSC-Derived Kidney Organoids Induces Neo-vasculogenesis and Significant Glomerular and Tubular Maturation In Vivo, Stem Cell Rep., 2018, 10(3), 751–765 CrossRef PubMed.
- E. Garreta, D. Moya-Rull, A. Marco, G. Amato, A. Ullate-Agote and C. Tarantino, et al., Natural Hydrogels Support Kidney Organoid Generation and Promote In Vitro Angiogenesis, Adv. Mater., 2024, 36(34), 2400306 Search PubMed.
- U. Arslan, F. E. Van Den Hil, C. L. Mummery and V. Orlova, Generation and Characterization of hiPSC-Derived Vascularized-, Perfusable Cardiac Microtissues-on-Chip, Curr. Protoc., 2024, 4(7), e1097 CrossRef CAS PubMed.
- L. S. Schulla, E. D. Alupoaie, L. De Silva, D. Gawlitta, S. Middendorp and P. J. Coffer, et al., Development of a Novel Microfluidic Co-culture model to study Organoid Vascularization, bioRxiv, 2022, preprint, DOI:10.1101/2022.03.25.485813.
- I. Salmon, S. Grebenyuk, A. R. Abdel Fattah, G. Rustandi, T. Pilkington and C. Verfaillie, et al., Engineering neurovascular organoids with 3D printed microfluidic chips, Lab Chip, 2022, 22(8), 1615–1629 RSC.
- S. Landau, Y. Zhao, H. Hamidzada, G. M. Kent, S. Okhovatian and R. X. Z. Lu, et al., Primitive macrophages enable long-term vascularization of human heart-on-a-chip platforms, Cell Stem Cell, 2024, 31(8), 1222–1238.e10 CrossRef CAS PubMed.
- M. Ballerini, S. Galiè, P. Tyagi, C. Catozzi, H. Raji and A. Nabinejad, et al., A gut-on-a-chip incorporating human faecal samples and peristalsis predicts responses to immune checkpoint inhibitors for melanoma, Nat. Biomed. Eng., 2025, 9(6), 967–984 CrossRef PubMed.
- K. A. Homan, N. Gupta, K. T. Kroll, D. B. Kolesky, M. Skylar-Scott and T. Miyoshi, et al., Flow-enhanced vascularization and maturation of kidney organoids in vitro, Nat. Methods, 2019, 16(3), 255–262 Search PubMed.
- A. Bas-Cristóbal Menéndez, Z. Du, T. P. P. Van Den Bosch, A. Othman, N. Gaio and C. Silvestri, et al., Creating a kidney organoid-vasculature interaction model using a novel organ-on-chip system, Sci. Rep., 2022, 12(1), 20699 Search PubMed.
- L. Y. Wu, D. Di Carlo and L. P. Lee, Microfluidic self-assembly of tumor spheroids for anticancer drug discovery, Biomed. Microdevices, 2008, 10(2), 197–202 Search PubMed.
- Q. Luan, C. Macaraniag, J. Zhou and I. Papautsky, Microfluidic systems for hydrodynamic trapping of cells and clusters, Biomicrofluidics, 2020, 14(3), 031502 Search PubMed.
- Y. Chen, D. Gao, Y. Wang, S. Lin and Y. Jiang, A novel 3D breast-cancer-on-chip platform for therapeutic evaluation of drug delivery systems, Anal. Chim. Acta, 2018, 1036, 97–106 Search PubMed.
- L. Yu, M. C. W. Chen and K. C. Cheung, Droplet-based microfluidic system for multicellular tumor spheroid formation and anticancer drug testing, Lab Chip, 2010, 10(18), 2424 RSC.
- L. A. Hetzel, A. Ali, V. Corbo and T. Hankemeier, Microfluidics and Organoids, the Power Couple of Developmental Biology and Oncology Studies, Int. J. Mol. Sci., 2023, 24(13), 10882 CrossRef CAS PubMed.
- A. Markoski, I. Y. Wong and J. T. Borenstein, 3D Printed Monolithic Device for the Microfluidic Capture, Perfusion, and Analysis of Multicellular Spheroids, Front. Med. Technol., 2021, 3, 646441 CrossRef PubMed.
- C. P. Huang, J. Lu, H. Seon, A. P. Lee, L. A. Flanagan and H. Y. Kim, et al., Engineering microscale cellular niches for three-dimensional multicellular co-cultures, Lab Chip, 2009, 9(12), 1740 RSC.
- H. Cho, H. Y. Kim, J. Y. Kang and T. S. Kim, How the capillary burst microvalve works, J. Colloid Interface Sci., 2007, 306(2), 379–385 CrossRef CAS PubMed.
- H. Liu, Y. Wang, K. Cui, Y. Guo, X. Zhang and J. Qin, Advances in Hydrogels in Organoids and Organs-on-a-Chip, Adv. Mater., 2019, 31(50), 1902042 CrossRef CAS PubMed.
- S. Rajasekar, D. S. Y. Lin, L. Abdul, A. Liu, A. Sotra and F. Zhang, et al., IFlowPlate—A Customized 384-Well Plate for the Culture of Perfusable Vascularized Colon Organoids, Adv. Mater., 2020, 32(46), 2002974 CrossRef CAS PubMed.
- G. E. Davis and D. R. Senger, Endothelial Extracellular Matrix: Biosynthesis, Remodeling, and Functions During Vascular Morphogenesis and Neovessel Stabilization, Circ. Res., 2005, 97(11), 1093–1107 CrossRef CAS PubMed.
- Y. Li, Q. M. Pi, P. C. Wang, L. J. Liu, Z. G. Han and Y. Shao, et al., Functional human 3D microvascular networks on a chip to study the procoagulant effects of ambient fine particulate matter, RSC Adv., 2017, 7(88), 56108–56116 Search PubMed.
- Y. K. Park, T. Y. Tu, S. H. Lim, I. J. M. Clement, S. Y. Yang and R. D. Kamm, In Vitro Microvessel Growth and Remodeling within a Three-Dimensional Microfluidic Environment, Cell. Mol. Bioeng., 2014, 7(1), 15–25 Search PubMed.
- K. T. Kroll, K. A. Homan, S. G. M. Uzel, M. M. Mata, K. J. Wolf and J. E. Rubins, et al., A perfusable, vascularized kidney organoid-on-chip model, Biofabrication, 2024, 16(4), 045003 CrossRef CAS PubMed.
- K. K. Yap, Y. W. Gerrand, A. M. Dingle, G. C. Yeoh, W. A. Morrison and G. M. Mitchell, Liver sinusoidal endothelial cells promote the differentiation and survival of mouse vascularised hepatobiliary organoids, Biomaterials, 2020, 251, 120091 Search PubMed.
- H. J. Kim, G. Kim, K. Y. Chi and J. H. Kim, In Vitro Generation of Luminal Vasculature in Liver Organoids: From Basic Vascular Biology to Vascularized Hepatic Organoids, Int. J. Stem Cells, 2023, 16(1), 1–15 CrossRef CAS PubMed.
- E. M. Holloway, J. H. Wu, M. Czerwinski, C. W. Sweet, A. Wu and Y. H. Tsai, et al., Differentiation of Human Intestinal Organoids with Endogenous Vascular Endothelial Cells, Dev. Cell, 2020, 54(4), 516–528.e7 Search PubMed.
- F. Wu, D. Wu, Y. Ren, Y. Huang, B. Feng and N. Zhao, et al., Generation of hepatobiliary organoids from human induced pluripotent stem cells, J. Hepatol., 2019, 70(6), 1145–1158 CrossRef PubMed.
- J. C. Maggiore, R. LeGraw, A. Przepiorski, J. Velazquez, C. Chaney and T. Vanichapol, et al., A genetically inducible endothelial niche enables vascularization of human kidney organoids with multilineage maturation and emergence of renin expressing cells, Kidney Int., 2024, 106(6), 1086–1100 Search PubMed.
- B. Cakir, Y. Xiang, Y. Tanaka, M. H. Kural, M. Parent and Y. J. Kang, et al., Engineering of human brain organoids with a functional vascular-like system, Nat. Methods, 2019, 16(11), 1169–1175 CrossRef CAS PubMed.
- K. Wang, R. Z. Lin, X. Hong, A. H. Ng, C. N. Lee and J. Neumeyer, et al., Robust differentiation of human pluripotent stem cells into endothelial cells via temporal modulation of ETV2 with modified mRNA, Sci. Adv., 2020, 6(30), eaba7606 CrossRef CAS PubMed.
- X. Y. Sun, X. C. Ju, Y. Li, P. M. Zeng, J. Wu and Y. Y. Zhou, et al., Generation of vascularized brain organoids to study neurovascular interactions, eLife, 2022, 11, e76707 Search PubMed.
- L. Song, X. Yuan, Z. Jones, K. Griffin, Y. Zhou and T. Ma, et al., Assembly of Human Stem Cell-Derived Cortical Spheroids and Vascular Spheroids to Model 3-D Brain-like Tissues, Sci. Rep., 2019, 9(1), 5977 CrossRef PubMed.
- D. Kong, K. H. Park, D. H. Kim, N. G. Kim, S. E. Lee and N. Shin, et al., Cortical-blood vessel assembloids exhibit Alzheimer's disease phenotypes by activating glia after SARS-CoV-2 infection, Cell Death Discovery, 2023, 9(1), 32 Search PubMed.
- N. Werschler, C. Quintard, S. Nguyen and J. Penninger, Engineering next generation vascularized organoids, Atherosclerosis, 2024, 398, 118529 Search PubMed.
- L. M. Becker, S. H. Chen, J. Rodor, L. P. M. H. de Rooij, A. H. Baker and P. Carmeliet, Deciphering endothelial heterogeneity in health and disease at single-cell resolution: progress and perspectives, Cardiovasc. Res., 2022, 119(1), 6–27 Search PubMed.
- S. Dobner, L. Kleissl, F. Tóth, R. Paxton, I. Yordanova and K. Brazdilova, et al., Uncovering the transcriptional hallmarks of endothelial cell aging via integrated single-cell analysis, bioRxiv, 2025, p. 2025.08.18.669055, preprint, DOI:10.1101/2025.08.18.669055v1.
- C. Praça, S. C. Rosa, E. Sevin, R. Cecchelli, M. P. Dehouck and L. S. Ferreira, Derivation of Brain Capillary-like Endothelial Cells from Human Pluripotent Stem Cell-Derived Endothelial Progenitor Cells, Stem Cell Rep., 2019, 13(4), 599–611 CrossRef PubMed.
- R. Samuel, L. Daheron, S. Liao, T. Vardam, W. S. Kamoun and A. Batista, et al., Generation of functionally competent and durable engineered blood vessels from human induced pluripotent stem cells, Proc. Natl. Acad. Sci. U. S. A., 2013, 110(31), 12774–12779 CrossRef CAS PubMed.
- C. Patsch, L. Challet-Meylan, E. C. Thoma, E. Urich, T. Heckel and J. F. O'Sullivan, et al., Generation of vascular endothelial and smooth muscle cells from human pluripotent stem cells, Nat. Cell Biol., 2015, 17(8), 994–1003 Search PubMed.
- Y. Li, P. Tang, S. Cai, J. Peng and G. Hua, Organoid based personalized medicine: from bench to bedside, Cell Regener., 2020, 9(1), 21 CrossRef PubMed.
- K. Salewskij and J. M. Penninger, Blood Vessel Organoids for Development and Disease, Circ. Res., 2023, 132(4), 498–510 Search PubMed.
- D. Skowronek, R. A. Pilz, V. V. Saenko, L. Mellinger, D. Singer and S. Ribback, et al., High-throughput differentiation of human blood vessel organoids reveals overlapping and distinct functions of the cerebral cavernous malformation proteins, Angiogenesis, 2025, 28(3), 32 CrossRef CAS PubMed.
- S. G. Romeo, I. Secco, E. Schneider, C. M. Reumiller, C. X. C. Santos and A. Zoccarato, et al., Human blood vessel organoids reveal a critical role for CTGF in maintaining microvascular integrity, Nat. Commun., 2023, 14(1), 5552 CrossRef CAS PubMed.
- S. E. Park, A. Georgescu and D. Huh, Organoids-on-a-chip, Science, 2019, 364(6444), 960–965 CrossRef CAS PubMed.
- K. Ronaldson-Bouchard, D. Teles, K. Yeager, D. N. Tavakol, Y. Zhao and A. Chramiec, et al., A multi-organ chip with matured tissue niches linked by vascular flow, Nat. Biomed. Eng., 2022, 6(4), 351–371 CrossRef CAS PubMed.
- Y. Jin, J. Kim, J. S. Lee, S. Min, S. Kim and D. Ahn, et al. Drug Screening: Vascularized Liver Organoids Generated Using Induced Hepatic Tissue and Dynamic Liver-Specific Microenvironment as a Drug Testing Platform (Adv. Funct. Mater. 37/2018), Adv. Funct. Mater., 2018, 28(37), 1870266 CrossRef.
- A. Skardal, S. V. Murphy, M. Devarasetty, I. Mead, H. W. Kang and Y. J. Seol, et al., Multi-tissue interactions in an integrated three-tissue organ-on-a-chip platform, Sci. Rep., 2017, 7(1), 8837 CrossRef PubMed.
- D. E. Ingber, Human organs-on-chips for disease modelling, drug development and personalized medicine, Nat. Rev. Genet., 2022, 23(8), 467–491 CrossRef CAS PubMed.
- Q. Ramadan, R. S. Fardous, R. Hazaymeh, S. Alshmmari and M. Zourob, Pharmacokinetics-On-a-Chip: In Vitro Microphysiological Models for Emulating of Drugs ADME, Adv. Biol., 2021, 5(9), 2100775 CrossRef PubMed.
- Qureator, First FDA IND Milestone Achieved Using Human Vascularized Organoid Efficacy Data, FierceBiotech, 2025, Available from: https://www.fiercebiotech.com/sponsored/first-fda-ind-milestone-achieved-using-human-vascularized-organoid-efficacy-data Search PubMed.
- X. Zhao, Z. Xu, L. Xiao, T. Shi, H. Xiao and Y. Wang, et al., Review on the Vascularization of Organoids and Organoids-on-a-Chip, Front. Bioeng. Biotechnol., 2021, 9, 637048 CrossRef PubMed.
Footnote |
| † Authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |