Structure-enabled liquid manipulation: bioinspired control across all dimensions
Siqi
Sun
a,
Liqiu
Wang
 *ab and
Yiyuan
Zhang
*ab and
Yiyuan
Zhang
 *a
*a
aDepartment of Mechanical Engineering, The Hong Kong Polytechnic University, Hong Kong SAR, PR China. E-mail: liqiu.wang@polyu.edu.hk; yi-yuan.zhang@polyu.edu.hk
bDepartment of Biomedical Engineering, The Hong Kong Polytechnic University, Hong Kong SAR, PR China
First published on 7th January 2026
Abstract
Directional liquid manipulation underpins critical processes across nature and engineering, where targeted functionality demands precise control over fluid behaviour. While fundamental theories for liquid manipulation are well-established, optimizing control along application-specific minimal-path trajectories remains a significant challenge. This review discusses recent advances in bioinspired strategies and engineered manipulators enabling superior liquid directional control across dimensional frameworks: 1D trajectories for targeted delivery, 2D planes for complex transport, and 3D spaces for programmable interfaces. Drawing on nature's energy-efficient principles, from Laplace pressure gradients to capillary effects, we decode evolutionary-optimized liquid manipulation mechanisms and their translation into dimension-specific artificial systems. These manipulators achieve precise liquid guidance through simplified asymmetric architectures, enhancing liquid utilization efficiency. Finally, we outline design paradigms for next-generation on-demand liquid control systems, bridging interfacial phenomena with microfluidic, thermal, and environmental technologies.
1. Introduction
Liquids are indispensable carriers of mass and energy in biological and industrial systems,1–7 mediating vital transfer processes through interfacial interactions with gases, solids, or other liquids.8–12 However, spatial separation between liquid sources and target phases often impedes direct exchange, necessitating directional spreading to bridge this gap. Uncontrolled isotropic spreading, driven by surface energy minimization, leads to significant resource loss. In addition, rapid vaporization under extreme conditions further limits liquid availability. Nature has evolved elegant solutions to these challenges: specialized structures in plants and animals enable directional transport,13,14 efficient condensation,15–17 and retention18,19 of liquids with minimal energy consumption,20,21 inspiring advanced liquid manipulation technologies.Controlling liquid movement requires guiding it along defined one-dimensional (1D) trajectories under unbalanced forces (Fig. 1).22 Natural systems have established specialised strategies for liquid directional control,23–25 exemplified through asymmetric cones (Sarracenia,26 cacti,27 and pine needles28), capillary architectures (mouthparts of honeybees,29,30 bats,31,32 butterfly,33 and hummingbirds34), and curvature morphology-facilitated deposition (A. macrorrhiza35). These enable horizontal, vertical upward, or vertical downward transport, which are fundamental directions that form the basis for complex pathways. Mimicking such strategies offers efficient solutions for fog harvesting,36,37 oil–water separation,38 water purification,39 and precision irrigation,40 minimizing residual loss via shortest-path transport.
 | ||
| Fig. 1 Overview of primary controllable liquid behaviours learned from biological strategies, categorized as controlled motion in 1D trajectories, 2D planes, and 3D space, as well as their relationships and suitable application scenarios. The foundational control of liquid behaviour is to achieve unidirectional motion, termed controllable 1D flow, with primary directions including horizontal, vertical upward, and vertical downward. Representative biological structures enabling motion in these directions are Sarracenia trichomes (horizontal), honeybee mouthparts (vertical upward), and A. macrorrhiza leaves (vertical downward). Controllable 1D flow is ideal for applications requiring directional liquid transport along the shortest trajectories to minimize residual loss, such as fog collection, water–oil separation, water purification and irrigation. Controllable 1D flow constructs multi-directional liquid surface deformations in 2D planes and 3D space. Typical plants that enable macro multi-directional liquid transport and apply multi-directional forces to liquid surfaces in 2D planes are Araucaria and lotus leaves. Such controllable 2D flow is suitable for applications demanding adequate mass and energy exchange. 3D liquid manipulation achieved in nature is through specialized plant structures like T. capitata, which enables 3D liquid transport trajectories and physiological processes such as transpiration that facilitates complex liquid–solid–gas interface formation. These controllable 3D flow systems are particularly valuable for applications requiring sophisticated liquid–environment interaction. The Sarracenia image is reprinted with permission from ref. 26. Copyright 2018, Springer Nature. The honey bee image is reprinted with permission from ref. 153. Copyright 2023, The American Association for the Advancement of Science. The A. macrorrhiza image is reprinted with permission from ref. 35. Copyright 2020, National Academy of Sciences. The Araucaria image is reprinted with permission from ref. 42. Copyright 2021, The American Association for the Advancement of Science. The lotus leaf image is reproduced under CC BY 4.0. The T. capitata image is reprinted with permission from ref. 45. Copyright 2025, National Academy of Sciences. The transpiration image is reprinted with permission from ref. 197. Copyright 2021, Springer Nature. | ||
Besides 1D, liquid manipulation extends to two-dimensional (2D) planes and three-dimensional (3D) space,41 enabling sophisticated surface deformations and interfacial morphologies (Fig. 1). In 2D, liquids achieve multidirectional transport (e.g., Araucaria42 and Crassula muscosa43) or form controlled wetting patterns (e.g., lotus leaf44). In 3D, structures like T. capitata45 or plant vasculature46 facilitate programmable transport and complex liquid–gas–solid interfaces, enhancing applications in biomedicine, thermal management, and antifouling.47–53
While existing reviews have extensively covered fundamental liquid control mechanisms54–60 and external-field actuation (e.g., electric, light, magnetic, thermal, acoustic, or pneumatic),61–77 systematic optimization of transport pathways for application-specific efficiency remains relatively underexplored. Structure-guided strategies, energy-efficient and inherently scalable, offer distinct advantages for tailored directional control:78 simplified 1D trajectories prove optimal for separation processes, whereas multidimensional (2D/3D) manipulation maximizes interfacial exchange in biochemical reactions,79,80 thermal cooling,81 or scenarios demanding precise interface regulation. To bridge this gap, our review first establishes the theoretical prerequisites for structural liquid control, and then lists bioinspired strategies and their underlying mechanisms evolved in nature. We subsequently highlight recent advances in structurally guided liquid manipulation across complexity scales—from foundational 1D trajectories to 2D open-channel planes and 3D spatial architectures—and conclude by addressing current limitations and future directions to advance application-driven liquid manipulator design.
2. Basic principles of liquid manipulation
Effective liquid manipulation requires understanding of the interaction between liquid properties, solid surface characteristics, and the ambient environment.82,83 This section outlines the fundamental mechanisms governing the liquid surface morphology and wetting dynamics, providing a theoretical foundation for designing solid structures that precisely control liquid behaviour under specific physicochemical conditions.The cornerstone of modern wettability theory is Young's equation84 (1805), which describes the equilibrium contact angle (θ) formed at the solid–liquid–vapor interface (Fig. 2a):85
γLG![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ = γSG − γSL θ = γSG − γSL | (1) |
Surface roughness fundamentally modulates wettability. Wenzel86 (1936) and Cassie–Baxter87 (1944) established models for rough surfaces (Fig. 2b and c). In the Wenzel state, liquid fully impregnates surface asperities:
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θA = r θA = r![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ θ | (2) |
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θA = σSL θA = σSL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ + (1 − σSL) cos θ + (1 − σSL) cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 180° 180° | (3) |
Liquid dynamics manifest as two key modes: (i) liquid-over-solid wetting: governed by contact angle hysteresis (CAH, Δθ), quantified via the Furmidge model92 using advancing (θa) and receding (θr) angles on inclined surfaces93 (Fig. 2d):
| Δθ = θa − θr | (4) |
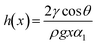 | (5) |
Interfacial energy minimization governs liquid transfer between immiscible phases (e.g., reagent aspiration from storage vials into reaction chambers).100 Consider a cubic manipulator immersed in liquid 1 (Fig. 2f). System energy (E1) depends on interfacial areas and tensions (σij):
| E1 = SSσ13 + Sσ12 | (6) |
| E2 = SSσ23 + Sσ12 | (7) |
| E3 = S23σ23 + Sσ12 + S13σ13 + S12σ12 | (8) |
3. Bioinspired liquid manipulation: evolutionary strategies in nature
Through evolutionary adaptation, organisms have developed sophisticated micro/nanostructures that optimize wettability and enable precise liquid control-essential adaptations for survival in diverse environments.3.1. Wettability engineering for survival
Organisms precisely tailor surface wettability to address ecological challenges. Mobile animals typically evolve surfaces for locomotion and interaction, while sessile plants develop defensive architectures against water loss, pathogens, or solar damage. Each solution reflects exquisite adaptation to specific biological constraints.Tree frogs (Fig. 3a) leverage hydrophilic hierarchical toe pads to retain secreted liquid, establishing complete wetting on varied substrates. This generates robust adhesion for arboreal locomotion.101 Roses (Fig. 3b) achieve paradoxical “sticky superhydrophobicity” through micropapillae with nanofolds.102,103 While high static angles (>150°) minimize pathogen contact, nanoscale cavities create strong contact angle hysteresis (CAH), pinning droplets even when inverted (the “petal effect”).104,105 This reconciles disease resistance with water acquisition needs. Lotus leaves (Fig. 3c) employ superhydrophobicity for complete repellency.106–108 Wax-coated microcapillaries and nanotubules109,110 create a composite air–liquid interface, enabling near-spherical droplets with ultra-low CAH. This prevents water adhesion—critical for avoiding sunlight-focused thermal damage (“lensing effect”) in aquatic habitats. Morpho butterfly wings (Fig. 3d) dynamically regulate wettability. Anisotropic micro/nanostructures enable reversible switching: outward tilting reduces solid–liquid contact for easy roll-off, while inward positioning engages flexible nanotips to increase adhesion—an active defense against rain/dew in tropical climates.111
 | ||
| Fig. 3 Overview and mechanism schematic of typical natural organisms with specialized liquid manipulation strategies. (a) Tree frog. (b) Rose petal. (c) Lotus leaf. (d) Butterfly wing. (e) Fish scales. (f) Dessert beetle surface. The tree frog optical image and SEM image of (a) are reprinted with permission from ref. 101. Copyright 2015, Wiley. The rose petal optical image of (b) is reprinted with permission from ref. 24. Copyright 2025, Elsevier. The rose petal SEM image of (b) is reprinted with permission from ref. 104. Copyright 2008, American Chemical Society. The lotus leaf optical image of (c) is reproduced under CC BY 4.0. The lotus leaf SEM image of (c) is reprinted with permission from ref. 82. Copyright 2017, Springer Nature. The butterfly optical image and SEM image of (d) are reprinted with permission from ref. 33. Copyright 2005, The Royal Society of Chemistry. The fish optical image of (e) is reprinted with permission from ref. 24. Copyright 2025, Elsevier. The fish SEM image of (e) is reprinted with permission from ref. 114. Copyright 2013, Wiley. The desert beetle optical image and SEM image of (f) are reprinted with permission from ref. 115. Copyright 2001, Springer Nature. | ||
Aquatic and arid specialists further demonstrate nature's ingenuity: fish skin (Fig. 3e) exhibits unique submerged amphiphilicity. Hierarchical scales of hydrophilic calcium proteins, phosphates, and mucus112 form directional micropillars. In water, trapped interfacial layers create an oil–water–solid composite interface, yielding superoleophobicity for drag reduction and contaminant repellence (“underwater superoleophobicity”).113,114 Desert beetles (Fig. 3f) harvest water via spatially patterned wettability.115 Their back features hydrophilic (wax-free, smooth bumps) and superhydrophobic (wax-coated, microstructured valleys) regions. In head-standing posture, fog nucleates on hydrophilic zones, grows, and rolls down hydrophobic channels to the mouth—enabling survival under hyperarid conditions.
These adaptations showcase nature's mastery in tailoring surface wettability for targeted liquid control. Besides surface phenomena, biological systems have also evolved sophisticated internal strategies for precise liquid management. For instance, plant leaves116 exhibit structurally optimized vascular networks that prevent gas embolism during drought, thereby ensuring continuous water supply. This mechanism represents a distinct strategy for efficient water utilization in harsh environments. Together with surface-wettability-based control, these strategies collectively provide a rich design lexicon for advanced bioinspired technologies.
3.2. Biological asymmetric surface structures for directional liquid control
Asymmetric surface architectures represent nature's sophisticated solution for directional liquid transport, leveraging structural anisotropies across scales to break interfacial symmetry. These evolved geometries harness energy gradient, Laplace pressure gradient, the capillary effect, and the contact line pinning effect to guide liquid transport along predetermined pathways with remarkable efficiency.Spider silk (Fig. 4a) exemplifies this principle through its periodic spindle-knot structures. Its complex hierarchical architecture comprises layered hydrophilic flagelliform proteins exhibiting humidity sensitivity.117–123 The alternating roughness between knots and joints creates diameter variations of spider silk and establishes a surface energy gradient. Rougher spindle knots possess higher surface energy (lower contact angles) compared to joints. When droplets condense on the silk, an imbalance in water–substrate interfacial tension across the three-phase contact line generates a net driving force (F) that propels droplets toward the more wettable spindle knots:120
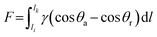 | (9) |
 | ||
| Fig. 4 Overview and mechanism schematic of directional liquid manipulation by organisms with asymmetric structures. (a) Spider silk. (b) Cactus spine. (c) Nepenthes alata. (d) Texas horned lizard. (e) Tillandsia capitata. (f) Crassula muscosa. (g) Araucaria. The spider silk image of (a) is reprinted with permission from ref. 123. Copyright 2010, Springer Nature. The cactus spine image of (b) is reprinted with permission from ref. 27. Copyright 2012, Springer Nature. The Nepenthes alata image of (c) is reprinted with permission from ref. 128. Copyright 2016, Springer Nature. The Texas horned lizard image of (d) is reprinted with permission from ref. 129. Copyright 2015, The Royal Society. The Tillandsia capitata image of (e) is reprinted with permission from ref. 45. Copyright 2025, National Academy of Sciences. The Crassula muscosa image of (f) is reprinted with permission from ref. 130. Copyright 2024, The American Association for the Advancement of Science. The Araucaria image of (g) is reprinted with permission from ref. 42. Copyright 2021, The American Association for the Advancement of Science. | ||
Cactus is a highly drought-resistant tropical plant that has evolved specialized morphological adaptations to thrive in arid environments (Fig. 4b). This plant efficiently harvests water from fog through an integrated system of hierarchical surface structures, featuring a regular array of needle-like spines and hair-like trichome clusters. Each spine has three functionally distinct regions: a barbed tip for droplet nucleation, a grooved mid-section with a gradient morphology for directional transport, and a base with belt-structured trichomes for water absorption. This sophisticated architecture enables fog collection through Laplace pressure gradients,126 where the conical geometry of the spines creates a pressure difference (ΔP) across adhering droplets (Fig. 4b). The resulting pressure imbalance drives directional water transport, as described by the following relationship:127
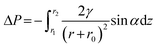 | (10) |
Nepenthes alata integrates two complementary fluid regulatory effects (Fig. 4c), i.e., forward capillary-driven fluid propagation and posterior edge-pinning stabilization. The forward transport is enabled by sharp wedge-shaped microcavities formed between hydrophilic arched structures and microgroove bases, which generate a capillary hysteresis force, and the force can be modelled as capillary elevation (Fig. 2e). The natural system exhibits an evolved gradient morphology where the wedge angle progressively contracts from α1 at the base to α2 at the apex. The resulting gradient capillary rise hN(x) along the transport path follows this relationship:128
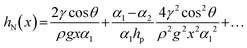 | (11) |
The Texas horned lizard (Fig. 4d) harnesses capillary action, synergistically combining with Laplace pressure gradient, to direct water toward its snout through its evolved semi-open capillary channels.129 The transport dynamics arise from the channels' saw-tooth periodicity, which creates asymmetric liquid surface curvature and consequently unbalanced Laplace pressures that preferentially drive forward flow twice as fast as reverse. Specifically, the lizard's skin features an interconnected network of shortened lateral capillaries that create continuous transport pathways. At capillary junctions, abrupt channel widening (e.g., at capillary I) causes the advancing liquid meniscus curvature radius to approach infinity, temporarily halting forward motion. When liquid from adjacent capillary II reaches these junctions, it spontaneously redistributes into capillary I, propelling the liquid front forward until it encounters the next capillary II segment. This self-sustaining cycle of capillary refilling and meniscus regeneration reflects a critical adaptation for desert hydration.
Tillandsia capitata leaves integrate macroscale curvature with microscale trichomes for efficient directional liquid transport and spontaneous redistribution (Fig. 4e).45 The transversely curved lanceolate macroscale morphology establishes fundamental liquid navigation pathways, and microscale hydrophilic trichomes provide capillary-driven water absorption. The density gradient distribution of trichomes generates a macroscopic wettability gradient from the leaf tip to base, which drives liquid directional transport via the surface energy gradient. Remarkably, the trichomes' continuously varying morphology achieves rapid liquid propagation through reduced interwing spacing, creating an apparent wetting gradient that operates even on hydrophobic substrates.
Crassula muscosa shoots (Fig. 4f) exhibit a unique capability for selectively directional liquid transport through the asymmetric reentrant leaf geometry.130 Variations in reentrant angles induce heterogeneous meniscus formation, where partial pinning at the lower fin edge in the negative direction enhanced the heterogeneity. As the advancing meniscus front reaches an adjacent fin, the capillary action at the corners accelerates liquid transfer across the fin, achieving velocities nearly 20 times faster than inter-fin propagation, while flow in other directions is suppressed. Upon crossing, a new meniscus forms and spreads directionally toward the next fin, establishing a self-sustaining cycle of selective liquid transport along the shoot surface.
Araucaria leaves have evolved ratchet structures with dual curvature that enable surface-tension-dependent liquid transport (Fig. 4g).42 With periodic spines tilted toward the leaf tip, the unique geometry exhibits an opposite liquid manipulation direction. Specifically, low-surface-tension liquids (e.g., ethanol) follow the ratchet inclination, while high-surface-tension liquids (e.g., water) move oppositely. The mechanism of this bidirectional liquid manipulation is asymmetric contact line pinning induced by the reentrant curvatures.
These natural organisms demonstrate evolutionarily refined structural anisotropies that enable effective liquid control strategies. By synergistically integrating directional wettability gradients, unbalanced Laplace pressures, and capillary dynamics, biologically optimized architectures achieve remarkable liquid manipulation.
The universal biological strategies of surface wettability and asymmetric structures establish a powerful design platform. This platform, in turn, guides the development of engineering liquid manipulators capable of precisely controlling liquid behaviours to meet diverse application requirements.
4. On-demand control of liquid surface deformation by engineered manipulators
By learning from nature's wisdom in surface engineering, researchers have created advanced liquid manipulators that enable on-demand control of liquid transport or surface deformation across 1D, 2D, and 3D trajectories. The core control strategies of these manipulators rely on proper surface wettability and well-designed structures to direct liquid motion or regulate liquid surface shape. These manipulators transform fundamental physical principles into platforms that merge operational simplicity with control accuracy, designed to meet diverse application needs, from guiding microfluidic flows in lab-on-a-chip diagnostics to enhancing droplet transport in atmospheric water collectors and facilitating selective chemical separation in industrial settings.131,1324.1. Liquid movement along 1D trajectories
Controllable liquid motion relies on applying unbalanced forces, orthogonally decomposable into three fundamental components: horizontal (Fig. 5a(i)), vertically upward (Fig. 5b(i)), and vertically downward (Fig. 5c(i)). Through the comprehensive investigation of liquid transport dynamics under these unbalanced forces, we could understand controllable liquid behaviour under arbitrary directional force effects. Gravity exhibits orientation-specific influences, becoming negligible in horizontal manipulation where it acts perpendicular to motion, resistive in upward vertical transport where it opposes the driving force, and assistive in downward vertical flow where it augments the applied force. This analytical approach reveals how solid manipulators, decorated with asymmetric microstructures, can generate the requisite forces to overcome or harness gravity, enabling precise control of the liquid morphology and transport pathways in engineered systems. | ||
| Fig. 5 Schematic, design, capability and applications of controllable 1D liquid movement along (a(i)–(iii)) horizontal, (b(i)–(iii)) vertical upward, and (c(i)–(iii)) vertical downward directions. The spider silk images of (a(ii)) and (a(iii)) are reprinted with permission from ref. 133. Copyright 2017, Springer Nature. The cone-tip structure image of (a(ii)) is reprinted with permission from ref. 139. Copyright 2013, Wiley. The oil–water separation figure of (a(iii)) is reprinted with permission from ref. 143. Copyright 2013, Springer Nature. The figure of (b(ii)) is reprinted with permission from ref. 146. Copyright 2025, Institute of Physics Publishing. The figure of (b(iii)) is reprinted with permission from ref. 148. Copyright 2022, Wiley. The figure of (c(ii)) is reprinted with permission from ref. 35. Copyright 2020, National Academy of Sciences. The figure of (c(iii)) is reprinted with permission from ref. 150. Copyright 2023, Springer Nature. | ||
Fiber-like structures with asymmetric topography, exemplified by spider silk-inspired spindle knots and cactus spine-derived conical designs, enable efficient horizontal liquid transport. These systems efficiently harvest discrete airborne droplets in humid environments and direct them along predefined paths (Fig. 5a(ii)).133 For instance, cavity-microfibers can absorb ambient moisture. Randomly condensed microdroplets subsequently grow via continuous vapor deposition and directionally migrate toward spindle knots due to the combined influence of surface energy gradients and Laplace pressure differences between knots and joints. This directional transport leads to progressive droplet coalescence at knots.
The system's maximum water collection capacity per single cavity-fiber is ultimately determined by the equilibrium between droplet adhesion and gravitational forces, with droplets detaching once exceeding the critical volume. Theoretical analysis yields the maximum sustainable droplet volume as (V):
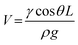 | (12) |
The cactus spine-inspired conical structure can be precisely engineered with tailored wettability, ranging from uniform hydrophobic/hydrophilic to spatially gradient patterns, to achieve directional liquid transport. Take water droplet movement on the hydrophilic surface as an example (Fig. 5a(ii)).139 The water droplet grows on the tip of the conical structure and moves toward the base part. During this transport, the droplet undergoes a distinct morphological transition from a barrel to a clamshell configuration, which fundamentally alters droplet dynamics.140 Common fabrication approaches like electrochemical corrosion and chemical modification enable wettability programming by creating micro/nanoscale features and functional group distributions.
As shown in Fig. 5a(iii), these fiber-like structures are ideal for sustainable fog harvesting applications, as an eco-friendly water recovery strategy.141,142 Due to different wettability responses to distinct liquid, these solid structures can selectively control target liquids (e.g., oil collection) while rejecting others (e.g., water repulsion), thereby achieving highly efficient oil–water separation.143–145
Vertically upward liquid transport typically relies on narrow tubes with capillary effects, as shown in Fig. 5b(i). In nature, plants like young pumpkin stems and Nepenthes alata have evolved far more sophisticated microstructural solutions for liquid transport. Inspired by these biological designs, researchers have developed structure-decorated microchannels using 3D printing, such as helicoidally patterned146 and peristome-mimetic structures,147 achieving approximately twice the liquid lifting height of similarly sized smooth microchannels (Fig. 5b(ii)) due to the enhanced capillary effect. Take the helicoidal microstructures as an example. They promote additional meniscus formation, reducing the radius of curvature and significantly increasing capillary pressure. Additionally, the patterned microstructures provide partial fluid support, further improving microfluidic performance. This biomimetic approach demonstrates how nature-inspired designs can outperform conventional methods in capillary-driven liquid transport. A key application requiring vertical liquid transport is water purification, as illustrated in Fig. 5b(iii).148 This system utilizes solar energy to purify wastewater and seawater. Microchannels first absorb the contaminated water, then filter impurities through adsorption along their walls, and finally collect purified water vapor generated by solar evaporation.
The typical purpose of controlling liquid vertically downward is to control the falling volume of droplets from the solid surface (Fig. 5c(i)). In general, on a non-hysteretic substrate, a droplet can only detach and fall when gravity (FG) overcomes the surface's adhesive forces, i.e., a balance governed by both gravitational pull and liquid–solid adhesion. For instance, on an inclined surface with tilt angle β (Fig. 5c(ii)), the primary adhesive force stems from capillary effects (FC), and droplet detachment occurs when:149
FG = ρgV![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) β > FC = γW β > FC = γW![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ θ | (13) |
WC/VC ∼ ρg![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) β/|γ β/|γ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ| θ| | (14) |
Consequently, a steeply inclined leaf surface (small β) enhances the gravitational component, reducing VC. When β is fixed, the bending position along the inclined surface further influences VC. As illustrated in Fig. 5c(ii), threshold droplets maintain a stable triple contact line (TCL) at a specific contact width W on a flat incline. If the surface bends precisely at the threshold contact line, the droplet remains stable. In contrast, bending the surface above the TCL disrupts equilibrium, increasing the gravitational force component while reducing capillary resistance, thus triggering droplet instability. This force redistribution causes the droplet to detach at a smaller volume than would be required on a uniformly inclined surface. The decreased volume of falling droplets allows effective agricultural drip irrigation (Fig. 5c(iii)), benefiting water saving, fast sprouting, and upright growth of crop seedlings. When the irrigation surface is placed vertically (β = 0), the falling droplet volume is smallest unless further optimization, such as surface front curvature150 and microstructure decoration of surface.151
As the manipulated liquid volume is further reduced (such as microliters or even nanoliters), gravity, which is a kind of body force, has little effect on such small-volume droplets. Therefore, to achieve controllable droplet release, it is necessary to introduce a stronger force to resist adhesion, such as mechanical force.152 In addition, combining the upward and downward forces to control the liquid can achieve liquid transfer.153
These capabilities in unidirectional liquid control represent a major step forward in fluid manipulation technology, with far-reaching implications for applications requiring directed liquid transport from the source to the target,154 such as precision agriculture, water source collection and industrial water processing. Harnessing these fundamental control mechanisms enables breakthrough liquid management capabilities, paving the way for more efficient, accurate, and sustainable liquid resource utilization across science and engineering.
4.2. Liquid transport/patterning on 2D planes
Applying forces to liquids within a 2D plane can be categorized into two distinct scenarios. The first involves sequential application of forces along multiple directions, enabling liquid transport along complex trajectories. The second scenario employs simultaneous application of forces along multiple directions to spatially confine liquids into predetermined shapes or restricted areas, with extreme confinement (as seen on superhydrophobic surfaces) producing quasi-spherical droplet formations. This dimensional expansion of fluid control fundamentally transforms liquid handling paradigms. While 1D manipulation remains essential for basic directional transport and many practical applications, 2D control unlocks significantly more advanced capabilities, such as innovative microfluidic technologies and anti-fouling superhydrophobic surfaces. The ability to precisely control both the position and morphology of liquids in 2D space represents a major breakthrough in microfluidics, enabling sophisticated liquid-based systems that integrate engineering functionality at unprecedented scales.As illustrated in Fig. 6a(i), pre-designed asymmetric surface structures enable precise control of complex liquid transport trajectories. For example, a flat superhydrophilic surface integrated with 2D barriers, which induce anisotropic resistance, controls liquid transport along the preferential direction (Fig. 6a(ii)).155 These barriers provide a simple yet robust solution for long-range liquid manipulation on open surfaces. Through adjusting the tilt angle, the transport direction can be regulated, and steeper angles reduce spreading resistance, allowing sequential directional control. Controllable liquid transport in 2D planes has important applications in biochemical analysis and high-throughput drug screening in both air and liquid environments,156–160 where samples can be precisely positioned and retrieved at any point within the microfluidic network. A notable implementation is the open microfluidic chip shown in Fig. 6a(iii), which enables autonomous liquid absorption, propulsion, and complete mixing.161 When the manipulated fluid is a functional liquid such as a coolant, its performance can be significantly enhanced on surfaces engineered with specific asymmetric structures, enabling applications like highly efficient steel pipe cooling.162 More innovatively, liquid transport trajectories can be the medium for information transmission.163 For example, the surface is designed only to permit directional transport for liquids with specific liquid–solid interfacial energy relationships (quantified by the ratio ζ = γS/γL, where γS is the surface free energy and γL is the liquid surface tension) (Fig. 6a(iv)).164 This forms the basis for a novel liquid-based encryption system, where information is encoded in surface patterns and is only decipherable using liquids with precise ζ values acting as decryption keys. The system demonstrates three distinct regimes: at low ζ values, liquids accumulate near injection points without revealing information; at high ζ values, uncontrolled bidirectional transport displays incorrect information; only within the appropriate ζ range does the liquid properly convey the intended message, creating a sophisticated platform for secure information transfer through controlled fluid dynamics.
 | ||
| Fig. 6 Schematic, design, capability and applications of multi-directional liquid transport. (a(i)–(iv)) The structure of the solid surface induced complex 2D liquid spreading trajectories. (b(i)–(iv)) The liquid intrinsic properties determined specific spreading directions. The figure of (a(ii)) is reprinted with permission from ref. 155. Copyright 2014, The Royal Society of Chemistry. The figure of (a(iii)) is reprinted with permission from ref. 161. Copyright 2023, Wiley. The figure of (a(iv)) is reprinted with permission from ref. 164. Copyright 2025, American Chemical Society. The figure of (b(ii)) is reprinted with permission from ref. 165. Copyright 2018, American Chemical Society. The figure of (b(iii)) is reprinted with permission from ref. 166. Copyright 2023, Springer Nature. The figure of (b(iv)) is reprinted with permission from ref. 168. Copyright 2024, Wiley. | ||
Direction-specific manipulation tailored to liquid properties enables advanced functionalities (Fig. 6b(i)), which is typically achieved through submillimeter to millimeter-scale asymmetric structures fabricated via 3D printing, leveraging Laplace pressure differences to directionally transport different liquids. For instance, ethanol (low surface tension) flows right while water (high surface tension) moves left (Fig. 6b(ii)).165 Such property-specific directional control has critical application in liquid separation systems, exemplified by the gear system that effectively separates oil–water emulsions (Fig. 6b(iii)).166 This system combines surface superwettability with complementary topological structures to achieve continuous separation: water and oil rapidly form distinct, mutually repelling films on their respective preferential gear surfaces, while the gears' rotational motion and overflow design maintain high separation flux by preventing fouling-induced performance degradation, which is an important improvement over oil–water separators based on 1D droplet moving trajectories. Moreover, this principle enables innovative liquid characterization methods.167 As shown in Fig. 6b(iv), surfaces are decorated with structured units, which have bottom-up multi-curvature features, thus directing liquids of different surface tensions into five distinct spreading modes under Laplace pressure gradients.168 By fabricating surfaces with progressively increasing unit heights (h1 < h2 < h3 < h4), they offer a portable surface tension indicator that refines measurement accuracy through mode-based classification. More significantly, this technology holds transformative potential as a versatile characterization tool capable of quantifying any physicochemical parameter that influences liquid surface tension, including solvent composition changes, temperature effects, or the presence of surface-active contaminants. The approach establishes a generalized paradigm for liquid property analysis through interfacial behaviour observation.
Multi-directional control of liquid transport trajectories further extends to thermal management applications through phase-change-enhanced heat dissipation, where controlled liquid generates vapor phases with high surface-to-volume ratios and latent heat benefits.169 A notable implementation is a thermal regulator combining Tesla valves with engineered capillary structures to rectify chaotic two-phase flow into directional transport, significantly boosting heat transfer coefficients and critical heat flux in its activated state.170 Conversely, thermal fields can also actively drive liquid motion through mechanisms such as the Leidenfrost effect, where localized vaporization creates self-propulsion of droplets, demonstrating another dimension of thermo-fluidic coupling.171,172
Besides the aforementioned control of the fluid transport direction through the design and fabrication of specific structures on fully open surfaces, the geometric shaping of surface channels, including their parameters and internal structural features, enables further regulation of additional liquid properties, such as flow resistance. Specifically, the design of channels with specialized shapes (e.g., spiral forms173) or the incorporation of asymmetric structures embedded within channels can modulate liquid behaviour, such as vortex dynamics and flow velocity.174,175 Such control is essential for high-efficiency particle separation, including polymer particles and cells, and holds significant value for both laboratory and commercial development of microfluidic systems in life and materials sciences.176–178
In practical applications of channel-based microfluidic systems, the intrusion of gas into water-filled microchannels often complicates liquid transport dynamics. This phenomenon can lead to gas blockages that severely disrupt continuous flow within microfluidic chips. Therefore, it is essential to design channel geometries and dimensional parameters that facilitate thorough investigation of gas-induced flow obstruction dynamics.179 Such a fundamental understanding is critical for developing stable and robust microfluidic devices.
Simultaneous application of forces along multiple directions to liquids enables precise spatial confinement of liquid surfaces, as demonstrated in Fig. 7a(i), where the droplet stabilized between four pillars forms well-defined liquid–environment interfaces, creating functional modular units capable of independent operation or programmed assembly. Such a liquid confinement strategy allows the construction of reconfigurable liquid devices with complex geometries and precisely controlled liquid–liquid interfaces (Fig. 7a(ii)),180 achieved through a 3D-printed pillar array substrate, immersed in silicone oil that facilitates in situ liquid unit formation via surface tension effects. The system exhibits distinct assembly behaviours where miscible liquids spontaneously merge into homogeneous structures while immiscible liquids maintain predetermined interfacial configurations. The underlying mechanism of maintaining the liquid unit arises from the resistance caused by Laplace pressure from convex water–oil interfaces, preventing liquid spreading, and thus creating mechanically stable liquid modules. The reconfigurable characteristic of such liquid units enables rapid connection, separation, and modification of liquid architectures, dramatically simplifying complex experimental workflows by integrating multiple processing steps, including alternating reaction and washing steps, into a single device. Fig. 7a(iii) shows the successful execution of all eight steps in human IL-4 protein ELISA using liquid unit constructed microfluidic channels. The system's dynamic reconfigurability provides unique advantages. When channels are disconnected, isolated microreactors form for specific reaction steps (2, 4, 6, and 8), preventing reagent waste in flow paths and connected equipment; when reconnected, integrated channels enable efficient washing steps (1, 3, 5, and 7) via simple syringe pump operation. Notably, the open architecture of this chip design permits sampling from any region at any processing stage without bubble disturbance, which is a critical advantage unachievable in conventional microfluidic systems. This innovative approach establishes a new paradigm for modular liquid manipulation that combines the precision of microfluidics with the flexibility of macroscopic fluid handling.
 | ||
| Fig. 7 Schematic, design, capability and applications of liquid surface shape manipulation by liquid–solid interactions. (a(i)–(iii)) Multi-directional confinement applied on a liquid unit. (b(i)–(iv)) Omni-directional confinement of small liquid volumes. Figures of (a(ii)) and (a(iii)) are reprinted with permission from ref. 180. Copyright 2024, Springer Nature. Figures of (b(ii)) and (b(iii)) are reprinted with permission from ref. 181. Copyright 2017, Springer Nature. The figure of (b(iv)) is reprinted with permission from ref. 182. Copyright 2019, The American Association for the Advancement of Science. | ||
The extreme case of multi-directional liquid manipulation involves applying forces along omni-directions to liquids within a 2D plane, as demonstrated in Fig. 7b(i). This is typically achieved through liquid-repellent surfaces that exert uniform forces in all directions, causing the liquid–solid contact area to form a circular interface while the droplet itself assumes a near-spherical shape. A breakthrough example shown in Fig. 7b(ii)181 presents a durable omniphobic membrane fabricated via microfluidic emulsion templating, featuring interconnected honeycomb microstructures with precisely engineered re-entrant geometries that provide exceptional liquid repellency. The membrane's flexible polyvinyl alcohol (PVA) composition allows for conformal coating on diverse substrates, including glass tubes and paper, as illustrated in Fig. 7b(iii). Besides solid surfaces, an alternative approach utilizes immiscible liquid–liquid interfaces to achieve omnidirectional confinement. Fig. 7b(iv) demonstrates how standard cell culture dishes can be transformed into reconfigurable microfluidic platforms by strategically manipulating liquid–liquid interfaces around adherent cells.182 This innovative technique, employing immiscible liquids as structural elements, enables critical biological applications including single-cell cloning, selective cell isolation, drug screening, and wound healing studies. The method's biological compatibility and operational simplicity address longstanding barriers to microfluidic adoption in life sciences,183,184 potentially accelerating the translation of microfluidic technologies from engineering prototypes to mainstream biological research tools that finally realize their transformative potential in biomedical applications.
In certain applications, control of liquid multidirectional transport and confinement of liquid surface shape have demonstrated exceptional performance. For example, anisotropic microstructured surfaces enable directional liquid sheeting, effectively removing low-surface-tension contaminants and small particles.185,186 2D liquid control systems outperform their 1D counterparts in droplet collection, demonstrating accelerated droplet growth rates, faster nucleation onset, higher steady-state turnover efficiency, and greater total collected volumes. This enhancement is achieved through optimized structural designs, including convex features that improve vapor diffusion flux, widening slope structures that promote downward droplet transport before the growth rate decline, and specialized nano coatings that facilitate synergistic interactions between coalescence-driven growth and capillary-assisted motion.187 Additionally, precise control over the three-phase contact line (TCL) retreat allows the fabrication of complex 3D structures from a single resin droplet.188 Moreover, retaining specific liquids within surface microstructures enables the transport of immiscible droplets,189 drag reduction,190 and friction noise suppression.191 These applications highlight the advantages of multi-directional liquid manipulation in 2D space: by simultaneously regulating interfacial forces along multiple directions, it enables complex fluid behaviours, opening new possibilities in surface engineering, microfluidics, and functional material design.
4.3. Programmable liquid manipulation in 3D space
3D liquid manipulation can be categorized into two primary approaches: controlling the 3D flow trajectories of liquids and programming the liquid surface morphology within 3D space. These strategies not only enable more sophisticated liquid control but also allow the creation of unprecedented complex liquid interfaces with tailored functionalities. Remarkably, besides maintaining the ability to handle liquids as conveniently as solids (permitting precise pickup, transportation, and deposition), the 3D programming of liquid surfaces maximizes the interfacial contact area between the liquid and its environment. This enhanced interface facilitates optimal mass and energy exchange, making the technology particularly valuable for applications in advanced microfluidics, biochemical reactors, and smart manufacturing systems where precise liquid control and efficient interfacial interactions are critical.A highly effective strategy for achieving 3D liquid transport trajectories is fabricating flexible liquid manipulators (Fig. 8a(i)). This approach enables on-demand adjustment of liquid pathways through material deformation. A prime example is the flexible liquid diode microtube fabricated via pulsed microfluidics, which achieves directional liquid transport through asymmetric pinning effects and unbalanced Laplace pressure.192,193 The microtube's pliable nature allows versatile configurations. Specifically, it can be coiled around cylindrical structures, freely suspended, or placed in surface-contact arrangements (Fig. 8a(ii)). Such flexible 3D liquid manipulators are particularly valuable for portable and wearable fluidic devices.194 For example, they can be integrated into electrolysis systems to create compact and wearable water-splitting microchips. A practical implementation could involve using in cold, polluted regions, where solar-powered hydrogen production provides heating fuel while simultaneously generating breathable oxygen (Fig. 8a(iii)).195 The ability to control flexible 3D liquid trajectories enables adaptive liquid manipulation in constrained space, which is crucial for portable energy systems and wearable medical devices.
 | ||
| Fig. 8 Schematic, design, capability and applications of controllable 3D complex liquid behaviours. (a(i)–(iii)) Soft liquid manipulators guided 3D flexible spreading trajectories. (b(i)–(iv)) The 3D liquid manipulation framework governed programmable liquid–environment interfaces. The figure of (a(ii)) is reprinted with permission from ref. 192. Copyright 2024, National Academy of Sciences. The figure of (a(iii)) is reprinted with permission from ref. 195. Copyright 2024, Elsevier. The figure of (b(ii)) is reprinted with permission from ref. 196. Copyright 2024, Springer Nature. The figure of (b(iii)) is reprinted with permission from ref. 197. Copyright 2021, Springer Nature. The figure of (b(iv)) is reprinted with permission from ref. 198. Copyright 2025, Wiley. | ||
Programming the 3D liquid surface morphology through solid frameworks enables omnidirectional liquid manipulation within 3D space (Fig. 8b(i)), where controlled liquid capture and release are achieved through capillary effects and drainage channels formed by liquid films. The underlying mechanism involves single-rod-connected frames that function as liquid capture units, while double-rod-connected frames create interconnecting liquid films that bridge the framed liquid with the bulk reservoir below, enabling programmable release, constructing a system capable of generating complex 3D liquid patterns (Fig. 8b(ii)).196 This solid-framework-based liquid programming technique significantly enhances liquid–environment interactions by creating optimized interfacial architectures. In gas absorption applications (Fig. 8b(iii)), the engineered frameworks dramatically increase liquid–gas contact areas, substantially improving processes like CO2 capture through enhanced transport of gaseous molecules into liquid sorbents.197 Similarly, for thermal management (Fig. 8b(iv)), these frameworks provide unprecedented control over continuous flow while maintaining open interfaces, eliminating the heat transfer limitations imposed by conventional pipe walls and enabling the development of highly efficient wall-less heat exchangers.198 The ability to program complex 3D liquid surfaces using solid frameworks represents a paradigm shift in fluid manipulation, as it combines the precision of engineered structures with the dynamic adaptability of liquids, enabling tailored interfacial geometries that optimize mass and energy transfer processes while maintaining the fundamental transport advantages of liquid systems. This approach opens new possibilities in chemical processing, energy systems, and thermal management, where enclosed fluid handling systems face fundamental limitations.
Extending the concept of manipulating liquids within 3D space to surfactant-mediated systems introduces a dynamic temporal dimension to spatial control. For example, surfactant solutions can autonomously navigate complex 2D mazes through exogenous–endogenous surfactant interactions.199,200 The incorporation of surfactants creates responsive liquid–solid interfaces where Marangoni stresses can be programmed to evolve spatially and temporally, enabling autonomous reconfiguration of liquid morphologies within predefined trajectories. This synergistic combination allows solid matrices to define static morphological boundaries while surfactant gradients dictate dynamic flow pathways, achieving coordinated liquid manipulation.
The engineered systems can be artificially designed to meet precise application requirements. This is achieved by recognizing fluidic processes as specific dimensional tasks, controlling liquid motion along 1D paths, across 2D surfaces, or within 3D spaces. By focusing on the specific behavioural requirements of liquids in a targeted dimension, researchers can develop specialized manipulators that apply the most appropriate control mechanisms, whether through tailored surface wettability, and optimized geometric features. This engineering approaches offer superior flexibility and tailorability for liquid manipulation, extending capabilities beyond what is commonly observed in the natural world.
5. Conclusions and outlook
Fluidity provides unparalleled advantages for energy and mass transfer,201 enabling liquids as indispensable media for diverse applications that demand tuneable physical properties or matter exchange, such as thermal management,202 soft robotics,203,204 biomolecular analysis,205,206 chemical reactions,207 and functional material synthesis.208,209 A challenge is that no single liquid manipulator can optimally serve all these distinct applications due to their divergent requirements. Instead, the solution lies in application-specific design, where liquid manipulators are tailored by considering the primary liquid morphology and its interaction dynamics with surrounding phases in each application. This targeted approach enables on-demand liquid control, with liquid macro behaviours categorized as controlled motion in 1D trajectories, 2D planes, and 3D space. By constructing dimension-specific (1D, 2D, or 3D) liquid manipulators designed with asymmetric structures, we can achieve efficient liquid control through simplified architectures and enhanced liquid utilization efficiency, mirroring nature's elegant, energy-minimizing strategies observed in biological systems.Each liquid manipulation strategy in different dimensions is suitable for specific liquid–solid–gas interaction. 1D trajectory-based liquid manipulation excels in targeted liquid collection from the source to destination (Fig. 9a). Typical scenarios include directional transport of gaseous-phase liquids on solid surfaces through designed wettability gradients and micro/nanostructures, and selective extraction and drainage of discrete liquid volumes from bulk liquids or immiscible mixtures via surface-mediated adsorption/expulsion mechanisms. These 1D liquid manipulators overcome intrinsic liquid mobility challenges, enabling rapid liquid movement, though they face practical constraints including volume limitations and long-term efficacy issues such as drainage delays or residual contamination. Arranging 1D asymmetric structures into 2D arrays unlocks complex planar liquid control (Fig. 9b). This approach enables thorough liquid–liquid mixing and reactions, and liquid–solid energy/matter transfer (exemplified by surface cooling and contaminant removal) via long-range and intricate transport pathways, and liquid morphology confinement. Current 2D surfaces, however, remain constrained to single predefined trajectories for one kind of liquid, necessitating complete device redesign for pathway modification, which is a significant barrier to flexible implementation.
3D structural manipulation achieves programmable liquid morphologies with partial liquid–solid contact and direct liquid–environment interaction (Fig. 9c), offering distinct advantages. Maximized specific surface area enhances gas absorption and release, and direct interfacial contact accelerates reaction kinetics, which is integrable with conventional systems (e.g., heat exchangers) and enables novel performance. However, like 1D liquid manipulators, these architectures remain susceptible to liquid retention on high-energy solid surfaces, limiting operational reusability.
Future progress demands deeper investigation of dynamic interphase phenomena. Engineering precursor film propagation could harness liquid–liquid interaction forces to achieve ultrafast transport beyond current limits. Integrating post-wetting behaviours, such as meniscus oscillation and coalescence dynamics, into structural designs may resolve drainage constraints. Furthermore, 3D nanoconfinement architectures show promise for stabilizing liquid films on surfaces, potentially overcoming depletion issues in slip surfaces. These frontiers underscore the field's development toward precisely orchestrated multiphase interactions across scales.
Author contributions
Siqi Sun: investigation, writing – original draft, writing – review & editing. Liqiu Wang: supervision, funding acquisition, writing – review & editing. Yiyuan Zhang: supervision, funding acquisition, investigation, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included, and no new data were generated or analysed as part of this review.Acknowledgements
The financial support from the Research Grants Council of Hong Kong (GRF 15309825, 17213823, and 17205421) and the Hong Kong Polytechnic University (SHS Chair Professor: P0045687; SHS Assistant Professor: P0058250 and P0048083) is acknowledged.Notes and references
- C. W. Lo, Y. C. Chu, M. H. Yen and M. C. Lu, Joule, 2019, 3, 2806–2823 CrossRef.
- L. Mohapatra and S. H. Yoo, Chem. Eng. J., 2024, 481, 148537 CrossRef.
- B. Wang, X. Luo, Y. Feng, L. Yang, C. Zhang, Z. Dong, L. Jiang and H. Dai, Adv. Sci., 2022, 9, 2204244 CrossRef.
- Q. Wang, X. Yao, H. Liu, D. Quéré and L. Jiang, Proc. Natl. Acad. Sci. U. S. A., 2015, 112, 9247–9252 CrossRef CAS PubMed.
- M. Prakash, D. Quéré and J. W. M. Bush, Science, 2008, 320, 931–934 CrossRef CAS.
- H. F. Bohn and W. Federle, Proc. Natl. Acad. Sci. U. S. A., 2004, 101, 14138–14143 CrossRef CAS.
- C. Zhang, S. Guo, X. Liu, Z. Guo, C. Yu, Y. Ning, K. Liu and L. Jiang, Appl. Phys. Rev., 2024, 11, 021316 CAS.
- W. Li, Z. Wang, F. Yang, T. Alam, M. Jiang, X. Qu, F. Kong, A. S. Khan, M. Liu, M. Alwazzan, Y. Tong and C. Li, Adv. Mater., 2019, 32, 1905117 CrossRef.
- C. Zhang, M. Cao, H. Ma, C. Yu, K. Li, C. Yu and L. Jiang, Adv. Funct. Mater., 2017, 27, 1702020 CrossRef.
- R. Wen, Q. Li, W. Wang, B. Latour, C. H. Li, C. Li, Y. C. Lee and R. Yang, Nano Energy, 2017, 38, 59–65 CrossRef CAS.
- M. A. Shannon, P. W. Bohn, M. Elimelech, J. G. Georgiadis, B. J. Mariñas and A. M. Mayes, Nature, 2008, 452, 301–310 CrossRef CAS PubMed.
- J. Miao, T. Zhang, G. Li, W. Shang and Y. Shen, Adv. Eng. Mater., 2022, 24, 2101399 CrossRef.
- I. J. Wright, et al. , Science, 2017, 357, 917–921 CrossRef CAS.
- H. Dai, Z. Dong and L. Jiang, Sci. Adv., 2020, 6, eabb5528 CrossRef CAS.
- M. Zhang, Z. Zheng, Y. Zhu, Z. Zhu, T. Si and R. X. Xu, Chem. Eng. J., 2022, 433, 134495 CrossRef CAS.
- H. Yue, Q. Zeng, J. Huang, Z. Guo and W. Liu, Adv. Colloid Interface Sci., 2022, 300, 102583 CrossRef CAS PubMed.
- S. R. Begum and A. Chandrasekhar, iScience, 2024, 27, 108878 CrossRef CAS.
- H. V. Moradi and J. M. Floryan, Procedia IUTAM, 2015, 14, 147–151 CrossRef.
- P. Motta, M. L. Habegger, A. Lang, R. Hueter and J. Davis, J. Morphol., 2012, 273, 1096–1110 CrossRef.
- A. K. Geim, S. V. Dubonos, I. V. Grigorieva, K. S. Novoselov, A. A. Zhukov and S. Y. Shapoval, Nat. Mater., 2003, 2, 461–463 CrossRef CAS PubMed.
- H. Lee, B. P. Lee and P. B. Messersmith, Nature, 2007, 448, 338–341 CrossRef CAS.
- J. Ju, Y. Zheng and L. Jiang, Acc. Chem. Res., 2014, 47, 2342–2352 CrossRef CAS.
- C. Zhang, S. Guo, X. Liu, Z. Guo, C. Yu, Y. Ning, K. Liu and L. Jiang, Appl. Phys. Rev., 2024, 11, 021316 CAS.
- C. Zhang, L. Wang, C. R. Crick and Y. Lu, Prog. Mater. Sci., 2025, 147, 101358 CrossRef.
- J. Li, J. Li, J. Sun, S. Feng and Z. Wang, Adv. Mater., 2019, 31, 1806501 CrossRef.
- H. Chen, T. Ran, Y. Gan, J. Zhou, Y. Zhang, L. Zhang, D. Zhang and L. Jiang, Nat. Mater., 2018, 17, 935–942 CrossRef CAS.
- J. Ju, H. Bai, Y. Zheng, T. Zhao, R. Fang and L. Jiang, Nat. Commun., 2012, 3, 1247 CrossRef.
- S. Feng, J. Delannoy, A. Malod, H. Zheng, D. Quéré and Z. Wang, Sci. Adv., 2020, 6, eabb4540 CrossRef.
- H. Yang, J. Wu and S. Yan, Appl. Phys. Lett., 2014, 104, 263701 CrossRef.
- A. Lechantre, et al. , Proc. Natl. Acad. Sci. U. S. A., 2021, 118, e2025513118 CrossRef CAS.
- C. J. Harper, S. M. Swartz and E. L. Brainerd, Proc. Natl. Acad. Sci. U. S. A., 2013, 110, 8852–8857 CrossRef CAS.
- Y. Winter and O. V. Helversen, J. Mammal., 2003, 84, 886–896 CrossRef.
- Y. Zheng, X. Gao and L. Jiang, Soft Matter, 2007, 3, 178–182 RSC.
- E. Siefert, et al. , Nat. Commun., 2025, 16, 4913 CrossRef CAS PubMed.
- T. Wang, Y. Si, H. Dai, C. Li, C. Gao, Z. Dong and L. Jiang, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 1890–1894 CrossRef.
- Q. Wang, Y. He, X. Geng, Y. Hou and Y. Zheng, ACS Appl. Mater. Interfaces, 2021, 13, 48292–48300 CrossRef PubMed.
- H. Dong, N. Wang, L. Wang, H. Bai, J. Wu, Y. Zheng, Y. Zhao and L. Jiang, ChemPhysChem, 2012, 13, 1153–1156 CrossRef PubMed.
- B. Wang, H. Dai, C. Zhang, Z. Dong, K. Li and L. Jiang, Adv. Mater. Interfaces, 2022, 9, 2102311 CrossRef.
- K. Dutta and S. De, J. Mater. Chem. A, 2017, 5, 22095–22112 RSC.
- B. Bhushan, Philos. Trans. R. Soc., A, 2009, 367, 1445–1486 CrossRef PubMed.
- T. Shen, N. Li, S. Liu, C. Yu, C. Zhang, K. Yang, X. Li, R. Fang, L. Jiang and Z. Dong, Int. J. Extreme Manuf., 2024, 6, 035502 CrossRef.
- S. Feng, P. Zhu, H. Zheng, H. Zhan, C. Chen, J. Li, L. Wang, X. Yao, Y. Liu and Z. Wang, Science, 2021, 373, 1344–1348 CrossRef.
- L. Yang, W. Li, J. Lian, H. Zhu, Q. Deng, Y. Zhang, J. Li, X. Yin and L. Wang, Science, 2024, 384, 1344–1349 CrossRef PubMed.
- W. Barthlott and C. Neinhuis, Planta, 1997, 202, 1–8 CrossRef.
- J. Lian, W. Li, L. Yang, H. Li, Q. Deng, H. Zhu, Y. Zhang, N. X. Fang and L. Wang, Proc. Natl. Acad. Sci. U. S. A., 2025, 122, e2421589122 CrossRef PubMed.
- T. D. Wheeler and A. D. Stroock, Nature, 2008, 455, 208–212 CrossRef PubMed.
- M. Filippi, T. Buchner, O. Yasa, S. Weirich and R. K. Katzschmann, Adv. Mater., 2022, 34, 2108427 Search PubMed.
- R. Zilionis, J. Nainys, A. Veres, V. Savova, D. Zemmour, A. M. Klein and L. Mazutis, Nat. Protoc., 2017, 12, 44–73 CrossRef PubMed.
- S. Xu, et al. , Science, 2014, 344, 70–74 CrossRef PubMed.
- F. Bian, L. Sun, L. Cai, Y. Wang, Y. Wang and Y. Zhao, Small, 2020, 16, 1903931 CrossRef PubMed.
- Y. Zheng, Y. Shao and J. Fu, Nat. Protoc., 2020, 16, 309–326 CrossRef.
- N. J. Cira, A. Benusiglio and M. Prakash, Nature, 2015, 519, 446–450 CrossRef.
- M. Jiang, et al. , Nature, 2022, 601, 568–572 CrossRef PubMed.
- Z. Liu, Y. Si, C. Yu, L. Jiang and Z. Dong, Chem. Soc. Rev., 2024, 53, 10012–10043 RSC.
- S. Zhou, L. Jiang and Z. Dong, Chem. Rev., 2023, 123, 2276–2310 CrossRef.
- L. Hou, et al. , Innovation, 2023, 4, 100508 Search PubMed.
- J. Xu, S. Xiu, Z. Lian, H. Yu and J. Cao, Droplet, 2022, 1, 11–37 CrossRef.
- M. Li, C. Li, B. R. K. Blackman and S. Eduardo, Int. Mater. Rev., 2021, 67, 658–681 CrossRef.
- H. Bai, T. Zhao and M. Cao, Chem. Soc. Rev., 2025, 54, 1733–1784 RSC.
- C. Li, B. Kim, J. Yoon, S. Sett and J. Oh, Adv. Funct. Mater., 2023, 34, 2308265 CrossRef.
- J. Li, N. S. Ha, T. Liu, R. M. Dam and C. J. Kim, Nature, 2019, 572, 507–510 CrossRef.
- M. G. Pollack, A. D. Shenderov and R. B. Fair, Lab Chip, 2002, 2, 96–101 RSC.
- W. Wang, H. Vahabi, A. Taassob, S. Pillai and A. K. Kota, Adv. Sci., 2024, 11, 2308101 CrossRef.
- S. Jiang, et al. , Nat. Commun., 2023, 14, 5455 CrossRef.
- J. Miao and A. C. H. Tsang, Adv. Sci., 2024, 11, 2405641 CrossRef PubMed.
- B. A. Grzybowski, H. A. Stone and G. M. Whitesides, Nature, 2000, 405, 1033–1036 CrossRef.
- J. Miao, Sens. Actuators, A, 2024, 368, 115104 CrossRef.
- J. Miao, S. Sun, T. Zhang, G. Li, H. Ren and Y. Shen, ACS Appl. Mater. Interfaces, 2022, 14, 50296–50307 CrossRef PubMed.
- K. Ichimura, S. K. Oh and M. Nakagawa, Science, 2000, 288, 1624–1626 CrossRef CAS PubMed.
- G. Kwon, et al. , Nat. Commun., 2017, 8, 14968 CrossRef PubMed.
- J. Lv, et al. , Nature, 2016, 537, 179–184 CrossRef CAS.
- J. Miao, T. Zhang, G. Li, D. Guo, S. Sun, R. Tan, J. Shi and Y. Shen, Engineering, 2023, 23, 170–180 CrossRef.
- S. Liu, et al. , Nat. Phys., 2025, 21, 808–816 Search PubMed.
- H. Zhu, et al. , Sci. Adv., 2025, 11, eadv6314 Search PubMed.
- Y. Xue, J. Markmann, H. Duan, J. Weissmüller and P. Huber, Nat. Commun., 2014, 5, 4237 Search PubMed.
- M. K. Chaudhury, A. Chakrabarti and S. Daniel, Langmuir, 2015, 31, 9266–9281 CrossRef CAS PubMed.
- J. Miao, J. Li and A. C. H. Tsang, Nano Lett., 2025, 25, 12495–12502 CrossRef CAS.
- H. Zhang, F. Wang and Z. Guo, Adv. Colloid Interface Sci., 2024, 325, 103097 CrossRef CAS PubMed.
- Y. Zhang, et al. , Adv. Mater., 2021, 34, 2108567 CrossRef.
- D. Wu, et al. , Adv. Mater., 2020, 32, 2005039 CrossRef CAS.
- C. Li, C. Yu, D. Hao, L. Wu, Z. Dong and L. Jiang, Adv. Funct. Mater., 2018, 28, 1707490 CrossRef.
- M. Liu, S. Wang and L. Jiang, Nat. Rev. Mater., 2017, 2, 17036 CrossRef CAS.
- W. Miao, Y. Tian and L. Jiang, Acc. Chem. Res., 2022, 55, 1467–1479 CrossRef CAS.
- T. Young, Philos. Trans. R. Soc. London, 1805, 95, 65–87 CrossRef.
- D. Bonn, J. Eggers, J. Indekeu, J. Meunier and E. Rolley, Rev. Mod. Phys., 2009, 81, 739 CrossRef CAS.
- R. N. Wenzel, Ind. Eng. Chem., 1936, 28, 988–994 CrossRef CAS.
- A. B. D. Cassie and S. Baxter, Trans. Faraday Soc., 1944, 40, 546–551 RSC.
- X. Zhang, Y. Lei, C. Li, G. Sun and B. You, Adv. Funct. Mater., 2022, 32, 2110830 CrossRef CAS.
- O. Tricinci, F. Pignatelli and V. Mattoli, Adv. Funct. Mater., 2023, 33, 2206946 CrossRef CAS.
- Y. Si, Z. Dong and L. Jiang, ACS Cent. Sci., 2018, 4, 1102–1112 CrossRef CAS PubMed.
- Y. Xiang, et al. , Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 2282–2287 CrossRef CAS.
- C. G. L. Furmidge, J. Colloid Sci., 1962, 17, 309–324 CrossRef CAS.
- H. J. Butt, J. Liu, K. Koynov, B. Straub, C. Hinduja, I. Roismann, R. Berger, X. Li, D. Vollmer, W. Steffen and M. Kappl, Curr. Opin. Colloid Interface Sci., 2022, 59, 101574 CrossRef CAS.
- A. Ponomarenko, D. Queue and C. Clanet, J. Fluid Mech., 2011, 666, 146–154 CrossRef.
- F. J. Higuera, A. Medina and A. Liñán, Phys. Fluids, 2008, 20, 102102 CrossRef.
- A. L. Ramos and R. L. Cerro, Chem. Eng. Sci., 1994, 49, 2395–2398 CrossRef.
- Q. Wang, Y. He, X. Geng, Y. Hou and Y. Zheng, ACS Appl. Mater. Interfaces, 2021, 13, 48292–48300 CrossRef CAS.
- K. Luan, M. He, B. Xu, P. Wang, J. Zhou, B. Hu, L. Jiang and H. Liu, Adv. Funct. Mater., 2021, 31, 2010634 CrossRef CAS.
- M. Lee, J. Oh, H. Lim and J. Lee, Adv. Funct. Mater., 2021, 31, 2011288 CrossRef CAS.
- Y. Zhang, et al. , Sci. Adv., 2021, 7, eabi7498 CrossRef CAS.
- J. Iturri, et al. , Adv. Mater., 2015, 25, 1499–1505 CAS.
- M. Chakraborty, J. A. Weibel, J. A. Schaber and S. V. Garimella, Adv. Mater. Interfaces, 2019, 6, 1900652 CrossRef.
- E. Bormashenko, T. Stein, R. Pogreb and D. Aurbach, J. Phys. Chem. C, 2009, 113, 5568–5572 CrossRef.
- L. Feng, Y. Zhang, J. Xi, Y. Zhu, N. Wang, F. Xia and L. Jiang, Langmuir, 2008, 24, 4114–4119 CrossRef PubMed.
- S. Ben, T. Zhou, H. Ma, J. Yao, Y. Ning, D. Tian, K. Liu and L. Jiang, Adv. Sci., 2019, 6, 1900834 Search PubMed.
- D. Wang, J. Huang and Z. Guo, Chem. Eng. J., 2020, 400, 125883 CrossRef.
- W. Barthlott and C. Neinhuis, Planta, 1997, 202, 1–8 Search PubMed.
- L. Feng, S. Li, Y. Li, H. Li, L. Zhang, J. Zhai, Y. Song, B. Liu, L. Jiang and D. Zhu, Adv. Mater., 2002, 14, 1857–1860 CrossRef.
- P. Wang, T. Zhao, R. Bian, G. Wang and H. Liu, ACS Nano, 2017, 11, 12385–12391 Search PubMed.
- C. Chen, M. Liu, L. Zhang, Y. Hou, M. Yu and S. Fu, ACS Appl. Mater. Interfaces, 2019, 11, 7431–7440 CrossRef PubMed.
- C. Liu, J. Ju, Y. Zheng and L. Jiang, ACS Nano, 2014, 8, 1321–1329 CrossRef PubMed.
- M. Liu, S. Wang, Z. Wei, Y. Song and L. Jiang, Adv. Mater., 2009, 21, 665–669 CrossRef.
- W. Rong, H. Zhang, T. Zhang, Z. Mao, X. Liu and K. Song, Adv. Eng. Mater., 2021, 23, 2000821 CrossRef.
- Y. Cai, et al. , Adv. Funct. Mater., 2013, 24, 809–816 CrossRef.
- A. R. Parker and C. R. Lawrence, Nature, 2001, 414, 33–34 CrossRef PubMed.
- T. J. Brodribb, D. Bienaimé and P. Marmottant, Proc. Natl. Acad. Sci. U. S. A., 2016, 113, 4865–4869 Search PubMed.
- F. Vollrath and D. T. Edmonds, Nature, 1989, 340, 305–307 Search PubMed.
- F. Vollrath, Rev. Mol. Biotechnol., 2000, 74, 67–83 Search PubMed.
- Y. Liu, Z. Shao and F. Vollrath, Nat. Mater., 2005, 4, 901–905 CrossRef PubMed.
- F. Vollrath, W. J. Fairbrother, R. J. P. Williams, E. K. Tillinghast, D. T. Bernstein, K. S. Gallagher and M. A. Townley, Nature, 1990, 345, 526–528 CrossRef.
- F. Vollrath and D. Porter, Soft Matter, 2006, 2, 377–385 RSC.
- E. Doblhofer, A. Heidebrecht and T. Scheibel, Appl. Microbiol. Biotechnol., 2015, 99, 9361–9380 CrossRef.
- Y. Zheng, et al. , Nature, 2010, 463, 640–643 Search PubMed.
- M. K. Chaudhury and G. M. Whitesides, Science, 1992, 256, 1539–1541 Search PubMed.
- S. Daniel, M. K. Chaudhury and J. C. Chen, Science, 2001, 291, 633–636 CrossRef.
- X. Yu, H. Lai, H. Kang, Y. Liu, Y. Wang and Z. Cheng, ACS Appl. Mater. Interfaces, 2022, 14, 6274–6282 CrossRef PubMed.
- E. Lorenceau and D. Queue, J. Fluid Mech., 2004, 510, 29–45 CrossRef.
- H. Chen, et al. , Nature, 2016, 532, 85–89 Search PubMed.
- P. Comanns, et al. , J. R. Soc., Interface, 2015, 12, 20150415 CrossRef.
- L. Yang, et al. , Science, 2024, 384, 1344–1349 CrossRef PubMed.
- L. Shang, Y. Cheng and Y. Zhao, Chem. Rev., 2017, 117, 7964–8040 CrossRef PubMed.
- Y. Si, C. Li, J. Hu, C. Zhang and Z. Dong, Adv. Funct. Mater., 2023, 33, 2301017 CrossRef.
- Y. Tian, et al. , Nat. Commun., 2017, 8, 1080 CrossRef PubMed.
- M. Du, Y. Zhao, Y. Tian, K. Li and L. Jiang, Small, 2016, 12, 1000–1005 CrossRef.
- A. Greiner and J. H. Wendorff, Angew. Chem., 2007, 119, 5770–5805 CrossRef.
- H. Bai, J. Ju, R. Sun, Y. Chen, Y. Zheng and L. Jiang, Adv. Mater., 2011, 23, 3708–3711 Search PubMed.
- H. Dong, Y. Zheng, N. Wang, H. Bai, L. Wang, J. Wu, Y. Zhao and L. Jiang, Adv. Mater. Interfaces, 2016, 3, 1500831 CrossRef.
- X. He, et al. , ACS Appl. Mater. Interfaces, 2015, 7, 17471–17481 CrossRef.
- J. Ju, K. Xiao, X. Yao, H. Bai and L. Jiang, Adv. Mater., 2013, 25, 5937–5942 CrossRef.
- J. Van Hulle, et al. , Phys. Rev. Fluids, 2021, 6, 024501 Search PubMed.
- M. Cao, J. Ju, K. Li, S. Dou, K. Liu and L. Jiang, Adv. Funct. Mater., 2014, 24, 3235–3240 CrossRef.
- J. Ju, X. Yao, S. Yang, L. Wang, R. Sun, Y. He and L. Jiang, Adv. Funct. Mater., 2014, 24, 6933–6938 CrossRef.
- K. Li, J. Ju, Z. Xue, J. Ma, L. Feng, S. Gao and Lei Jiang, Nat. Commun., 2013, 4, 2276 CrossRef.
- Z. Cui, et al. , J. Mater. Chem. A, 2025, 13, 18870 Search PubMed.
- Y. Li, et al. , Adv. Funct. Mater., 2022, 32, 2201035 CrossRef.
- Z. Wang, et al. , Int. J. Extreme Manuf., 2025, 7, 025505 CrossRef.
- C. Li, H. Dai, C. Gao, T. Wang, Z. Dong and L. Jiang, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 12704–12709 CrossRef PubMed.
- Z. Wang, Z. Zhan, L. Chen, G. Duan, P. Cheng, H. Kong, Y. Chen and H. Duan, Sol. RRL, 2022, 6, 2101063 Search PubMed.
- T. Wang, Y. Si, H. Dai, C. Li, C. Gao, Z. Dong and L. Jiang, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 1890–1894 Search PubMed.
- S. Liu, C. Zhang, T. Shen, Z. Zhan, J. Peng, C. Yu, L. Jiang and Z. Dong, Nat. Commun., 2023, 14, 5934 Search PubMed.
- S. Liu, Z. Zhan, Y. Si, C. Yu, L. Jiang and Z. Dong, Adv. Funct. Mater., 2023, 33, 2212485 Search PubMed.
- X. Tang, P. Zhu, Y. Tian, X. Zhou, T. Kong and L. Wang, Nat. Commun., 2017, 8, 14831 Search PubMed.
- Z. Cheng, C. Li, C. Gao, C. Zhang, L. Jiang and Z. Dong, Sci. Adv., 2023, 9, eadi5990 CrossRef PubMed.
- S. Zhang, M. Chi, J. Mo, T. Liu, Y. Liu, Q. Fu, J. Wang, B. Luo, Y. Qin, S. Wang and S. Nie, Nat. Commun., 2022, 13, 4168 CrossRef PubMed.
- H. Geng, H. Bai, Y. Fan, S. Wang, T. Ba, C. Yu, M. Cao and L. Jiang, Mater. Horiz., 2018, 5, 303–308 Search PubMed.
- B. Zhao, J. S. Moore and D. J. Beebe, Science, 2001, 291, 1023–1026 CrossRef PubMed.
- A. I. Shallan, Y. Tavares, M. N. Kashani, M. C. Breadmore and C. Priest, Angew. Chem., 2021, 133, 2686–2689 Search PubMed.
- D. J. Guckenberger, T. E. Groot, A. M. D. Wan, D. J. Beebea and E. W. K. Young, Lab Chip, 2015, 15, 2364–2378 Search PubMed.
- J. Yu, et al. , Nat. Biomed. Eng., 2019, 3, 830–841 CrossRef.
- Q. Zhang, S. Feng, L. Lin, S. Mao and J. M. Lin, Chem. Soc. Rev., 2021, 50, 5333–5348 RSC.
- X. Liu, B. Li, Z. Gu and K. Zhou, Small, 2023, 19, 2207640 CrossRef.
- U. Hermens, et al. , Appl. Surf. Sci., 2017, 418, 499–507 Search PubMed.
- M. Xie, et al. , Small, 2023, 19, 2300047 CrossRef.
- J. Miao and A. C. H. Tsang, ACS Nano, 2025, 19(5), 5829–5838 CrossRef.
- Y. Si, T. Wang, C. Li, C. Yu, N. Li, C. Gao, Z. Dong and L. Jiang, ACS Nano, 2018, 12, 9214–9222 CrossRef PubMed.
- Z. Liu, Z. Zhan, T. Shen, N. Li, C. Zhang, C. Yu, C. Li, Y. Si, L. Jiang and Z. Dong, Nat. Commun., 2023, 14, 4128 CrossRef.
- S. Sun, J. Miao, R. Tan, T. Zhang, G. Li and Y. Shen, Adv. Funct. Mater., 2022, 33, 2209769 Search PubMed.
- S. Sun, Y. Zhang, S. Wu and L. Wang, Adv. Mater., 2024, 36, 2209769 Search PubMed.
- R. Erp, R. Soleimanzadeh, L. Nela, G. Kampitsis and E. Matioli, Nature, 2020, 585, 211–216 CrossRef PubMed.
- W. Li, S. Yang, Y. Chen, C. Li and Z. Wang, Nat. Commun., 2023, 14, 3996 CrossRef.
- G. Dupeux, et al. , Sci. Rep., 2014, 4, 5280 CrossRef PubMed.
- A. Bouillant, et al. , Nat. Phys., 2018, 14, 1188–1192 Search PubMed.
- S. Shen, et al. , Lab Chip, 2017, 17, 3578–3591 RSC.
- M. Saito, et al. , Proc. Natl. Acad. Sci. U. S. A., 2024, 121, e2306182121 CrossRef PubMed.
- A. Groisman, et al. , Phys. Rev. Lett., 2004, 92, 094501 Search PubMed.
- E. K. Sackmann, A. L. Fulton and D. J. Beebe, Nature, 2014, 507, 181–189 Search PubMed.
- Y. Yu, H. Wen, J. Ma, S. Lykkemark, H. Xu and J. Qin, Adv. Mater., 2014, 26, 2494–2499 Search PubMed.
- Y. Ma, J. Z. Pan, S. P. Zhao, Q. Lou, Y. Zhu and Q. Fang, Lab Chip, 2016, 16, 4658–4665 RSC.
- L. Keiser, P. Marmottant and B. Dollet, J. Fluid Mech., 2022, 948, A52 CrossRef.
- Y. Zeng, et al. , Nat. Chem. Eng., 2024, 1, 149–158 CrossRef.
- P. Zhu, T. Kong, X. Tang and L. Wang, Nat. Commun., 2017, 8, 15823 Search PubMed.
- C. Soitu, A. Feuerborn, C. Deroy, A. A. Castrejón-Pita, P. R. Cook and E. J. Walsh, Sci. Adv., 2019, 5, eaav8002 CrossRef PubMed.
- E. J. Walsh, et al. , Nat. Commun., 2017, 8, 816 CrossRef.
- C. Li, et al. , Sci. Adv., 2020, 6, eaay9919 CrossRef PubMed.
- K. Luan, M. He, B. Xu, P. Wang, J. Zhou, B. Hu, L. Jiang and H. Liu, Adv. Funct. Mater., 2021, 31, 2010634 CrossRef.
- Y. J. Chen, et al. , ACS Appl. Mater. Interfaces, 2024, 16, 36840–36850 CrossRef PubMed.
- K. C. Park, P. Kim, A. Grinthal, N. He, D. Fox, J. C. Weaver and J. Aizenberg, Nature, 2016, 531, 78–82 CrossRef.
- Y. Zhang, Z. Dong, C. Li, H. Du, N. X. Fang, L. Wu and Y. Song, Nat. Commun., 2020, 11, 4685 CrossRef PubMed.
- C. Yu, L. Zhang, Y. Ru, N. Li, C. Li, C. Gao, Z. Dong and L. Jiang, ACS Nano, 2018, 12, 11307–11315 CrossRef.
- K. Yang, et al. , Adv. Mater., 2025, 37, 2417337 CrossRef.
- B. Zhang, K. Yang, X. Yu, J. Zhang, Z. Dong, R. Fang and L. Jiang, Adv. Funct. Mater., 2025, 35, 2422552 Search PubMed.
- C. Yang, W. Li, Y. Zhao and L. Shang, Proc. Natl. Acad. Sci. U. S. A., 2024, 121, e2402331121 CrossRef PubMed.
- C. Yang, Y. Yu, L. Shang and Y. Zhao, Nat. Chem. Eng., 2024, 1, 87–96 CrossRef.
- B. Zhang, et al. , Nature, 2024, 628, 84–92 CrossRef PubMed.
- M. Cao, et al. , Matter, 2024, 7, 3053–3068 CrossRef.
- Y. Zhang, et al. , Nat. Chem. Eng., 2024, 1, 472–482 CrossRef.
- N. A. Dudukovic, et al. , Nature, 2021, 595, 58–65 CrossRef.
- S. Wu, S. Sun, J. Ye, L. Wang and Y. Zhang, Adv. Mater., 2025, 2503840 Search PubMed.
- R. Mcnair, et al. , Phys. Rev. Lett., 2025, 134, 034001 Search PubMed.
- F. T. Coleto, et al. , Phys. Rev. Fluids, 2018, 3, 100507 CrossRef.
- H. Zhao, et al. , Adv. Funct. Mater., 2024, 34, 2407654 Search PubMed.
- X. Lin, H. Wang, Y. Wu and S. He, ACS Nano, 2025, 19, 25986–25998 Search PubMed.
- C. Zhang, et al. , Nano Lett., 2025, 25, 11618–11625 CrossRef CAS PubMed.
- J. Miao and S. Sun, J. Magn. Magn. Mater., 2023, 586, 171160 CrossRef.
- D. Mark, S. Haeberle, G. Roth, F. Stetten and R. Zengerle, Chem. Soc. Rev., 2010, 39, 1153–1182 RSC.
- E. K. Sackmann, A. L. Fulton and D. J. Beebe, Nature, 2014, 507, 181–189 CrossRef PubMed.
- S. Wu, et al. , Appl. Phys. Lett., 2023, 122, 174101 CrossRef.
- P. Zhu and L. Wang, Lab Chip, 2017, 17, 34–75 RSC.
- Q. Deng, et al. , Nat. Commun., 2025, 16, 4601 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |





