 Open Access Article
Open Access ArticleDetermination of absolute isotope ratios of vanadium by internal standardisation
D.
Malinovsky
 *,
P. J. H.
Dunn
*,
P. J. H.
Dunn
 ,
B. A.
Engin
and
H.
Goenaga-Infante
,
B. A.
Engin
and
H.
Goenaga-Infante

National Measurement Laboratory, LGC, 10 Priestley Road, Guildford, GU2 7XY, UK. E-mail: dmitriy.malinovskiy@lgcgroup.com
First published on 29th April 2026
Abstract
Due to the prohibitively high cost of materials enriched in the 50V isotope, internal standardisation is the only viable method for the determination of absolute vanadium isotope ratios by mass spectrometry. This work describes methodology that applies, for the first time, internal standardisation to the determination of n(51V)/n(50V) isotope ratios by multi-collector inductively coupled plasma mass spectrometry (MC-ICP-MS). To achieve this, solutions of vanadium standard NIST SRM 3165 were doped with iron standard IRMM-014 with certified Fe isotope amount ratios, which was used as a calibrant in the measurements. A comparison of the performance of the established models to correct for instrumental mass fractionation showed that although the mean n(51V)/n(50V) values obtained by the regression model and the exponential model were statistically indistinguishable (399.8 ± 12.6 and 397.3 ± 1.7, Uexpanded, k = 2, respectively), their uncertainties were remarkably different. High uncertainty of the n(51V)/n(50V) value by the regression model was attributed to bias arising due to heterogeneity of variance of measured data on the log scale in a situation when isotope ratios of an analyte and an internal standard are significantly different. By its nature, the regression model offered an important advantage of assumption-free calibration, and, following its unsatisfactory performance, a new calibration method that does not rely on a priori assumed functional form of instrumental isotope fractionation was developed in this study. The new method is based on extracting a calibration factor for measured 51V/50V ratios from the relationship constructed between calibration factors obtained for measured isotope ratios of the internal standard and ratios of relative atomic masses making up these ratios (56Fe/54Fe, 57Fe/54Fe and 57Fe/56Fe). The data obtained allowed us to re-determine the atomic weight of vanadium in NIST SRM 3165, to get Ar(V) = 50.94146 ± 0.00003 (Uexpanded, k = 2), with the uncertainties improved by a factor of four as compared to the last estimate made in 1977.
Introduction
Vanadium has two stable isotopes, namely 50V and 51V, with natural abundances of ∼0.25% and ∼99.75%, respectively. Variations in the abundances of vanadium isotopes were shown to be useful markers in geochemistry and environmental science.1–10 They help discriminate the origin of vanadium containing materials and can provide insight into migration pathways of the element. Recent studies documented an increase in global anthropogenic releases of the element to soil and water that led to naming vanadium a re-emerging environmental hazard.11 Given the potential offered by vanadium isotope data in the above areas, further studies employing vanadium isotopes are warranted.Vanadium isotope ratio data were reported previously almost exclusively as delta values. Despite being a convenient way of reporting results, the delta value notation has an important drawback of poor data comparability in the situation when an isotope standard used is not certified for absolute isotope ratios. This is precisely the situation for vanadium isotope ratio measurements as there is no vanadium material with certified n(51V)/n(50V) ratios.
The determination of absolute isotope ratios of vanadium with high accuracy and precision is a very non-trivial task. The traditional method for the determination of absolute isotope ratios is based on the use of synthetic isotope mixtures to calibrate measurements. These calibration mixtures need to be prepared from parent materials highly enriched in 50V and 51V isotopes. However, the cost of 50V isotope materials, such as vanadium oxides, with the required degree of enrichment is prohibitively high, starting from more than five thousand US dollars for a milligram of vanadium pentoxide enriched in 50V isotope.12
An alternative calibration method is internal standardisation. Using this approach, an internal standard with a known amount of a substance, different from the analyte of interest, is added to samples to correct for fluctuations in instrument response during analysis and as an aid for calibration.13,14 It has been shown that normalisation to an internal standard with certified isotope abundances by using the so-called regression method is the metrologically rigorous method for the determination of isotope amount ratios.15–17 Empirical models based on a priori assumed functional relationships between measured isotope ratios of an analyte and internal standard are also a popular choice, albeit it is known that the accuracy of results obtained by these models is often poor.18
In this study, the performance of the existing approaches for the determination of the n(51V)/n(50V) isotope ratio of vanadium standard solution NIST SRM 3165 by normalisation to the internal standard with certified Fe isotope abundances has been systematically evaluated. Following initial results, a novel method for obtaining a calibration factor that relates true and measured isotope ratios of vanadium was developed and validated. The isotope ratio data obtained using the new method have enabled re-determination of the atomic weight of vanadium with a four-fold improvement in measurement uncertainty relative to the last IUPAC estimate in the year 1977.
Experimental
Materials and reagents
Single element NIST Standard Reference Material 3165, a vanadium standard solution (Lot No. 992706, NIST, USA) certified only for concentration of vanadium, was used as the sample in this study. IRMM-014 Fe solution with certified Fe isotope abundances was used as the internal standard in the measurements (JRC, Belgium). High purity deionised water was obtained using an ELGA water purification system (Veolia Water, Marlow, UK). Ultrapure nitric acid was purchased from Romil (Cambridge, UK) and used as supplied by the manufacturer.Mass spectrometry and uncertainty estimation
Vanadium and iron isotope ratios were measured by multicollector ICP-MS (Neptune, Thermo Scientific, Germany). A stable introduction system consisting of a peristaltic pump, a 50 µl min−1 microconcentric PFA nebulizer, and a tandem quartz spray chamber arrangement (cyclone + Scott double pass) was used for sample introduction. 50V+, 51V+, 52Cr+, 53Cr+, 54Fe+, 56Fe+ and 57Fe+ isotopic ions were collected simultaneously in static mode at Low 3, Low 2, Low 1, Central, High 1, High 2 and High 4 Faraday cup positions, respectively. Signals on 52Cr and 53Cr isotopes were monitored in the case of a potential presence of chromium which also has a minor 50Cr isotope which, if present, interferes with the 50V isotope. The least abundant 58Fe isotope was not measured because it was not possible to achieve a well-aligned static Faraday cup configuration that included 50V and 58Fe isotopes due to the limitations on ion beam dispersion in MC-ICP-MS and the fact that an ion counter was installed on the Low 4 Faraday cup.The NIST SRM 3165 standard solution was in most cases diluted to a vanadium concentration of ∼2 mg l−1 and spiked with the IRMM-014 Fe standard at ∼2 mg l−1. In the beginning of each measurement session, the instrument was carefully tuned to maximise the intensity of the vanadium signal by adjusting the torch position, gas flow rates, and lens voltages. Typical operating conditions for MC-ICPMS are shown in Table 1. All analyses were conducted in high mass resolution mode of the instrument (R ∼ 9000). Typical sensitivities for 51V+ and 56Fe+ isotopic ions were ∼6 volts and ∼5 volts per mg l−1, respectively.
| RF power, W | 1100 | ||||||
|---|---|---|---|---|---|---|---|
| Sample and skimmer cones | Nickel, 1.1 mm and 0.8 mm orifice diameter, respectively; H-type skimmer cone | ||||||
| Argon gas flow rate, l min−1 | |||||||
| Cool/plasma | 15.4 | ||||||
| Auxiliary | ∼0.9 | ||||||
| Nebuliser | ∼1.05 | ||||||
| Sample uptake rate, ml min−1 | ∼0.10–0.12 | ||||||
| Mass resolution, R(5–95%) | ∼9000 | ||||||
| Ion lens settings | Optimised for maximum intensity of the vanadium signal | ||||||
| Typical 51V+ sensitivity, V mg l−1 | ∼6.4 | ||||||
| Detector configuration | |||||||
| Isotope m/z | 50V | 51V | 52Cr | 53Cr | 54Fe | 56Fe | 57Fe |
| Faraday cup | L3 | L2 | L1 | Centre | H1 | H2 | H3 |
| Cup position, mm | 74.300 | 47.100 | 21.080 | 0.000 | 22.540 | 68.340 | 90.310 |
With the aim of producing a larger extent of variations in instrumental mass bias, the measurement protocol involved a series of single measurements, each with an incremental change in the RF power away from the maximum sensitivity setting at ∼1100 W with steps ranging from 10 W to 40 W.16,17 Each measurement consisted of 100 cycles, with each cycle having an 8 second duration. It was always verified in the on-line scan window prior to starting the next measurement that the measurement position was in the middle of the interference-free plateau of the 50V+ and 56Fe+ signals.
Vanadium and iron ion current intensities were corrected for procedural blank. The blank signal on the least abundant 50V isotope was always lower than 0.1% and lower than 0.05% for 51V and all Fe isotopes. n(51V)/n(50V) isotope ratio calculations were performed off-line using a template made in MS Excel program. Combined standard uncertainties of measurement results were obtained by propagating the uncertainty of individual components according to the ISO/GUM Guide (see details in the SI).
Results and discussion
Calibration of n(51V)/n(50V) isotope ratios by using the regression model
The regression model is based on correlated temporal drift between simultaneously measured isotope ratios of the analyte element and internal standard element. The rationale of the model was detailed in previous works.15–17 It can be shown that when using the 56Fe/54Fe ratio of isotopically certified iron as the internal standard as a calibrator, mass bias corrected isotope ratios of vanadium, n(51V)/n(50V), can be calculated as follows: | (1) |
A notable advantage of using iron as an internal standard is that the element has four stable isotopes, namely 54Fe, 56Fe, 57Fe, and 58Fe. The abundances of these isotopes were certified in the IRMM-014 isotope standard and allow us to use 56Fe/54Fe, 57Fe/54Fe, and 57Fe/56Fe isotope ratios to calibrate the measured 51V/50V ratios of the NIST SRM 3165 material. Therefore, equations analogous to eqn (2) can be written when 57Fe/54Fe and 57Fe/56Fe isotope ratios are used for calibration. This also implies that each measurement yields a vanadium isotope ratio that represents a grand mean of three 51V/50V ratio values calculated by normalisation to 56Fe/54Fe, 57Fe/54Fe, and 57Fe/56Fe isotope ratios of the admixed internal standard, respectively.
Another point to consider is that the slope and intercept of regression can be calculated by using either an ordinary linear regression (OLR), that does not account for measurement errors in variables X and Y, or an errors-in-variables model that does account for measurement errors. The use of the latter can help achieve higher accuracy in estimates of slopes and intercepts but often at the cost of higher uncertainties associated with their values. A weighted linear regression (WLR) as described by MacTaggert and Farwell (1992)19 was used in this work as one of the variants of the errors-in-variables model.
n(51V)/n(50V) isotope ratios obtained by using the regression model in 12 independent measurement sessions are shown in Fig. 1. After examining these data, two observations can be made. First, there is a good agreement between values obtained by OLR and WLR. Second, there is significant dispersion in n(51V)/n(50V) isotope ratios between measurement sessions, resulting in high uncertainty of the grand mean. The OLR approach yields an n(51V)/n(50V) isotope ratio of NIST SRM 3165 equal to 399.8 ± 12.6 (Uexpanded, k = 2). The WLR approach yields an n(51V)/n(50V) isotope ratio of the same solution equal to 401.9 ± 23 (Uexpanded, k = 2). In relative terms, these uncertainties are 3.1% and 5.7%, respectively. In comparison to previous studies which used the regression model for calibration of isotope ratios of medium and heavy mass elements, the results obtained for vanadium are poor.
Interestingly, when measured 51V/50V and 56Fe/54Fe isotope ratios without mass bias correction are presented with the same statistical treatment as the data in Fig. 1, we can see that they are much more repeatable. As can be seen from Fig. 2, the mean value for all measured 51V/50V isotope ratios is equal to 408.6 ± 1.0 (Uexpanded, k = 2), and the mean value for all measured 56Fe/54Fe isotope ratios is equal to 16.510 ± 0.071 (Uexpanded, k = 2). In relative terms, these uncertainties are 0.24% and 0.43%, respectively.
Comparison of the data shown in Fig. 1 and 2 reveals that the main reason for poor repeatability of vanadium isotope ratios calibrated by the regression model is the lack of accuracy in the determination of regression parameters. Difficulties in obtaining an accurate slope and intercept of linear regression through V and Fe isotope ratio data plotted in ln–ln space are largely driven by the inherent properties of the natural logarithmic function.
A logarithmic curve, defined by the equation y = ln(x), is characterised by a decreasing rate of change that levels off as the input variable, x, grows (Fig. 3A). As a consequence, the vertical variability in a plot of logarithmic data may depend on where you are on the horizontal scale, with variance of the measurement data in the region of Fe isotope ratios (e.g., 56Fe/54Fe ≈ 16.5 and ln(56Fe/54Fe) ≈ 2.8) being slightly different to variance of the data for a large isotope ratio, such as that of vanadium (51V/50V ≈ 408 and ln(51V/50V) ≈ 6.01).
 | ||
| Fig. 3 An illustration of a potential cause for unequal variance of the log transformed Fe and V isotope ratios. Part (A): the natural logarithmic function, corresponding to the range of isotope ratios from 0.023 to 411 considered in this study, is a non-linear curve. The rate of change in the Y axis values of this curve is dependent on the position on the horizontal scale. Part (B): a plot of residuals vs. fit values for all measurements of 51V/50V ratios made in this study. The vertical distance between any one measured data point, yi, and its value, yi, predicted by using regression coefficients is known as “residual”: ei = yi − yi. A funnel like pattern of the data on such plots is commonly interpreted as an indicator of unequal variance21 (see the text for details). | ||
This problem of unequal variance, also known as heterogeneity of variance or heteroscedasticity, was discussed previously for the log transformed data as it can lead to bias in estimating regression coefficients.20–22 A plot of residuals versus predicted values of ln(51V/50V) used in identifying heteroscedasticity of the data is shown in Fig. 3B. As can be seen from this figure, the scatter of data points forms a funnel shape pattern which is commonly interpreted as an indicator of unequal variance. More rigorous techniques can be used to confirm heteroscedasticity of the data, including Welch's t-test. However, to obtain strong evidence, one will need to consider a wider than natural range of ln(51V/50V) values, which can only be obtained using synthetically prepared synthetic isotope mixtures of enriched vanadium isotopes. Yet, as noted above, the prohibitively high cost of the enriched 50V isotope does not allow easy implementation of such tests.
It is also worth noting that previous studies that determined isotope amount ratios of Ni, Mo, Pb and other elements with high accuracy and precision by using the regression model worked with more favourable ranges of isotope ratios of analytes and internal standards.15–17,23–28 When plotted on a ln–ln scale, the isotope ratios of analytes and internal standards in these studies were not as significantly different as in the case of vanadium.
Calibration by using the exponential model and robustness of the measurements
The measurement procedure with incremental changes in RF power offers a good opportunity to test the robustness of measurements with internal standardisation. The robustness or ruggedness of an analytical method means its capacity to remain unaffected by small variations in the operating parameters and produce consistent and repeatable results.29 The exponential model is well suited for testing the repeatability of results obtained under slightly changed measurement conditions because the calibration factor can be calculated from a single measurement of isotope ratios of an analyte and an internal standard.Mass bias corrected vanadium isotope ratios were calculated using the approach by Russell et al. (1978),18,30 written as follows in the case of the 56Fe/54Fe ratio of the internal standard:
| R51V/50V = K·r51V/50V | (2) |
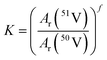 | (3) |
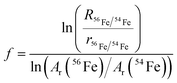 | (4) |
The n(51V)/n(50V) isotope ratios of NIST SRM 3165 obtained by using the exponential model are shown in Fig. 4. These ratios are very reproducible, with the grand mean of 397.3 ± 1.7 (Uexpanded, k = 2). In relative terms, the expanded uncertainty of the result obtained by the exponential model is equal to 0.42% and that is much lower than the expanded uncertainties of the results of the regression model discussed above.
New method of calibration using internal standardisation
The new method exploits the fact that three isotope ratios of the internal standard, namely 56Fe/54Fe, 57Fe/54Fe and 57Fe/54Fe, were measured simultaneously with the 51V/50V isotope ratio. Since Fe isotope abundances in the internal standard were certified, it is possible to construct a relationship between calibration factors obtained using the isotope ratios of the internal standard and ratios of relative atomic masses of 54Fe, 56Fe, and 57Fe isotopes, making up these ratios. Fig. 5 shows such a relationship based on the results of a single measurement session. This relationship can be plotted on both a linear scale and a ln–ln scale. The lines passing through the data points in Fig. 5 closely approximate the distribution of calibration factors as a function of relative atomic mass ratios over the narrow range of values defined by Fe isotopes.At the same time, the relative atomic mass ratio of 51V and 50V isotopes, that is equal to 1.020, sits within the range defined by the relative atomic mass ratios of Fe isotopes. A calibration factor for the measured 51V/50V isotope ratio can therefore be calculated using parameters of regression through the data obtained for the internal standard. The n(51V)/n(50V) isotope ratios obtained by this approach, with extracting calibration factors from the Fe isotope data plotted on a linear scale, are shown in Fig. 6. The grand mean value of these n(51V)/n(50V) isotope ratios is 397.59 ± 2.04 (Uexpanded, k = 2). A nearly identical grand mean value of 397.60 ± 2.04 (Uexpanded, k = 2) was obtained when calibration factors for vanadium isotope ratios were extracted from the Fe isotope data plotted on a ln–ln scale.
The combined standard uncertainties of n(51V)/n(50V) isotope ratios were estimated by propagating uncertainty through the calculations with the aid of the numerical method of differentiation by Kragten (1994).31 The largest contribution to total uncertainty budget of the new method comes from uncertainty in determining the K factor from Fe isotope ratios of the internal standard that, on average, accounts for 70% of total uncertainty. Contribution from measurements of the analyte, 51V/50V isotope ratio, accounts for the remaining 30% of total uncertainty.
To compare the results of the new calibration method with results of the exponential and regression models, the n(51V)/n(50V) isotope ratio values of NIST SRM 3165 and their uncertainties obtained in this study are shown in Fig. 7. As can be seen from this figure, the result of the new method is in excellent agreement with the result of the exponential model. A closer look at the procedure by which the abundances of Fe isotopes were determined in the standard IRMM-014 reveals why this agreement is so good. Only the n(56Fe)/n(54Fe) isotope ratio of the IRMM-014 standard was calibrated using the gravimetrically prepared mixtures of enriched 56Fe and 54Fe isotopes; other isotope ratios were obtained using an internal normalisation procedure.32 This implies that once an internal standard with independently calibrated Fe isotope ratios is used, the accuracy of n(51V)/n(50V) isotope ratio measurements by the new method can further be improved. Likewise, uncertainties of the measurements can also be improved provided that isotope amount ratios of a new Fe isotope standard have lower uncertainties than those of the IRMM-014 material.
 | ||
| Fig. 7 n(51V)/n(50V) isotope ratios of the NIST SRM 3165 vanadium standard determined by different techniques of internal standardisation. Uncertainty bars are expanded uncertainties (k = 2). | ||
It is also worth noting that calculating a calibration factor for measured vanadium isotope ratios from the relationship between calibration factors and relative atomic mass ratios of Fe isotopes implies mass-dependent instrumental isotope fractionation in the mass spectrometer. As vanadium has only two stable isotopes, there is no way of identifying a potential mass-independent component in instrumental fractionation of vanadium isotopes from measured ratios (a three-isotope plot can be used for this purpose in other elements). Deviations from the mass-dependent distribution in measured isotope ratios can be caused by a spread in the ion energy during transmission of ions in the mass spectrometer.33 However, measured Fe isotope ratios of the internal standard are consistent with the mass-dependent pattern, ruling out such a spread as the cause. Mass-independent isotope fractionation can also potentially occur due to the differences in nuclear properties of isotopes. However, a careful examination of research on the topic shows that it is very unlikely that any detectable mass-independent vanadium isotope fractionation can occur during measurements by MC-ICP-MS. Of the two known mechanisms, the nuclear volume effect was predicted to be negligible for naturally occurring vanadium species.6 Another mechanism, the magnetic isotope effect, requires a solvent cage to manifest itself.34 Yet it is unrealistic for a solvent cage to be formed in the plasma and the plasma interface region of the ICP-MS.34,35
The isotope abundances and atomic weight of vanadium in NIST SRM 3165
The atomic weight of an element is the weighted sum of the relative masses of its isotopes, Ar(iE), and the abundance of each isotope, x(iE):36| Ar(E) = ∑[x(iE)·Ar(iE)] | (5) |
The abundances of 50V and 51V isotopes were calculated from the n(51V)/n(50V) isotope ratio, R, obtained for the NIST SRM 3165 standard as follows:
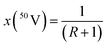 | (6) |
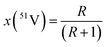 | (7) |
Conclusions
When assessing the performance of the existing calibration approaches, we observed that the so-called regression model was unable to determine the n(51V)/n(50V) isotope ratios of the NIST SRM 3165 vanadium solution with sufficiently low uncertainty. Difficulties in obtaining accurate and precise parameters of regression seem to be connected with the pattern of measurement data distribution in ln–ln space and the effect of reducing the scatter for large isotope ratios, such as a 51V/50V ratio of ∼408. This observation will need to be considered when the regression model is applied for the determination of large isotope ratios of other elements, e.g., in characterisation of highly enriched isotopic materials.Interestingly, the exponential model based on the functional form known as the Russell's equation demonstrated reproducible and robust performance, with the grand mean of the mass bias corrected n(51V)/n(50V) isotope ratios of 397.3 ± 1.7 (Uexpanded, k = 2).
The possibility of using three isotope ratios of the internal standard, 56Fe/54Fe, 57Fe/54Fe, and 57Fe/56Fe, to calibrate the measured 51V/50V isotope ratio led to the development of a new method based on internal standardisation that does not rely on a priori assumed functional form of instrumental isotope fractionation. It involves calculation of a calibration factor for measured vanadium isotopes from a relationship of calibration factors versus relative atomic mass ratios obtained for Fe isotope ratios of the internal standard. By using the newly developed method, the n(51V)/n(50V) isotope ratio of the NIST SRM 3165 vanadium solution was determined to be 397.6 ± 2.0 (Uexpanded, k = 2). This has enabled re-determination of the abundances of 50V and 51V isotopes and the atomic weight of a reference sample of vanadium (NIST SRM 3165), the uncertainties of which have been improved by a factor of four as compared to the last estimate made in 1977. The developed method has good potential for isotopic characterisation of vanadium reference materials.
This work has also highlighted that further improvement in accuracy and precision of n(51V)/n(50V) isotope ratio measurements by internal standardisation can be achieved by using a standard with independently calibrated Fe isotope amount ratios, which is currently unavailable on the market.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: Table S1 – Isotope ratio data obtained in this study and further experimental details. See DOI: https://doi.org/10.1039/d5ja00509d.Acknowledgements
The work described in this paper was funded in part by the UK Government Department for Science, Innovation & Technology (DSIT) through the National Measurement System Chemical and Biological Metrology Programme and also received funding from the European Partnership on Metrology (Funder ID: 10.13039/100019599, Grant number: 21GRD09 MetroPOEM).References
- P. I. Premović, I. R. Tonsa, L. López, M. S. Pavlović, O. M. Nešović, S. LoMonaco, D. M. Đorđević and M. V. Veljkovic, J. Inorg. Biochem., 2000, 80, 153–155 CrossRef PubMed.
- S. G. Nielsen, J. Prytulak and A. N. Halliday, Geostand. Geoanal. Res., 2011, 35, 293–306 CrossRef CAS.
- J. Prytulak, S. G. Nielsen, D. A. Ionov, A. N. Halliday, J. Harvey, K. A. Kelley, Y. L. Niu, D. W. Peate, K. Shimizu and K. W. W. Sims, Earth Planet. Sci. Lett., 2013, 365, 177–189 CrossRef CAS.
- D. Malinovsky and N. Kashulin, Anal. Methods, 2016, 8, 5921–5929 RSC.
- M. A. Stow, J. Prytulak, M. C. S. Humphreys, S. J. Hammond and G. M. Nowell, Earth Planet. Sci. Lett, Earth Planet. Sci. Lett., 2024, 643, 118825 CrossRef CAS.
- T. Fujii, C. Kato, N. Wada, A. Uehara, P. Sossi and F. Moynier, ACS Earth Space Chem., 2023, 7(4), 912–925 CrossRef CAS.
- L. H. Dong, W. Wei, C. L. Yu, Z. H. Hou, Z. Zeng, T. Chen and F. Huang, Anal. Chem., 2021, 93(19), 7172–7179 CrossRef CAS PubMed.
- W. Yan, B. Zhang, Y. Li, J. Lu, Y. Fei, S. Zhou, H. Dong and F. Huang, Engineering, 2025, 46, 257–266 CrossRef CAS.
- S. Schuth, A. Brüske, S. V. Hohl, S. Y. Jiang, A. K. Meinhardt, D. D. Gregory, S. Viehmann and S. Weyer, Chem. Geol., 2019, 528, 119261 CrossRef CAS.
- Y. Huang, Z. Long, D. Zhou, L. Wang, P. He, G. Zhang, S. S. Hughes, H. Yu and F. Huang, Sci. Total Environ., 2021, 791, 148240 CrossRef CAS PubMed.
- J. A. J. Watt, I. T. Burke, R. A. Edwards, H. M. Malcolm, W. M. Mayes, J. P. Olszewska, G. Pan, M. C. Graham, K. V. Heal, N. L. Rose, S. D. Turner and B. M. Spears, Environ. Sci. Technol., 2018, 52, 11973–11974 CrossRef CAS PubMed.
- C. Cinder, Purchase quotation from Oak Ridge National Laboratories, 2014 Search PubMed.
- D. A. Skoog, F. J. Holler and S. R. Crouch, Principles of Instrumental Analysis, Cengage Learning, 7th edn, 2018 Search PubMed.
- D. T. Burns and M. J. Walker, Anal. Bioanal. Chem., 2019, 411, 2749–2753 CrossRef CAS PubMed.
- J. Meija, L. Yang, R. E. Sturgeon and Z. Mester, J. Anal. At. Spectrom., 2010, 25, 384–389 RSC.
- D. Malinovsky, P. J. H. Dunn and H. Goenaga-Infante, J. Anal. At. Spectrom., 2016, 31, 1978–1988 RSC.
- S. Tong, J. Meija, L. Zhou, B. Methven, Z. Mester and L. Yang, Anal. Chem., 2019, 91(6), 4164–4171 CrossRef CAS PubMed.
- D. C. Baxter, I. Rodushkin and E. Engström, J. Anal. At. Spectrom., 2012, 27, 1355–1381 RSC.
- D. L. MacTaggert and S. O. Farwell, J. AOAC Int., 1992, 75, 608–614 CrossRef.
- P. Kennedy, Oxf Bull Econ Stat, 1983, 45(4), 389–392 CrossRef.
- W. G. Manning, J. Health Econ., 1998, 17, 283–295 CrossRef CAS PubMed.
- R. Richardson, H. D. Tolley, W. E. Evenson and B. M. Lunt, PLoS One, 2018, 13(5), e0197222 CrossRef PubMed.
- Z. Zhu, J. Meija, A. Zheng, Z. Mester and L. Yang, Anal. Chem., 2017, 89(17), 9375–9382 CrossRef CAS PubMed.
- R. Zhang, J. Meija, Y. Huang, X. Pei, Z. Mester and L. Yang, Anal. Chim. Acta, 2019, 48(4), 19–24 CrossRef PubMed.
- J. He, J. Meija, X. Hou, C. Zheng, Z. Mester and L. Yang, Anal. Bioanal. Chem., 2020, 412(24), 6257–6263 CrossRef CAS PubMed.
- J. He, L. Yang, X. Hou, Z. Mester and J. Meija, Anal. Chem., 2020, 92(8), 6103–6110 CrossRef CAS PubMed.
- D. Malinovsky, P. J. H. Dunn and H. Goenaga-Infante, J. Anal. At. Spectrom., 2020, 35, 2723–2731 RSC.
- J. Meija, J. He, B. Methven, Z. Mester and L. Yang, Geostand. Geoanal. Res., 2024, 48(4), 795–805 CrossRef CAS.
- Y. Vander Heyden, A. Nijhuis, J. Smeyers-Verbeke, B. G. M. Vandeginste and D. L. Massart, J. Pharm. Biomed. Anal., 2001, 24, 723–753 CrossRef CAS PubMed.
- W. A. Russell, D. A. Papanastassiou and T. A. Tombrello, Geochim. Cosmochim. Acta, 1978, 42, 1075–1090 CrossRef CAS.
- J. Kragten, Analyst, 1994, 119, 2161–2165 RSC.
- P. D. P. Taylor, R. Maeck and P. De Bièvre, Int. J. Mass Spectrom. Ion Process., 1992, 121, 111–125 CrossRef CAS.
- F. Albarède, P. Telouk, J. Blichert-Toft, M. Boyet, A. Agranier and B. Nelson, Geochim. Cosmochim. Acta, 2004, 68, 2725–2744 CrossRef.
- A. Buchachenko, J. Phys. Chem. B, 2013, 117, 2231–2238 CrossRef CAS PubMed.
- D. Malinovsky and F. Vanhaecke, Anal. Bioanal. Chem., 2011, 400, 1619–1624 CrossRef CAS PubMed.
- T. Prohaska, J. Irrgeher, J. Benefield, J. K. Böhlke, L. A. Chesson, T. B. Coplen, T. Ding, P. J. H. Dunn, M. Gröning, N. E. Holden, H. A. J. Meijer, H. Moossen, A. Possolo, Y. Takahashi, J. Vogl, T. Walczyk, J. Wang, M. E. Wieser, S. Yoneda, X.-K. Zhu and J. Meija, Pure Appl. Chem., 2022, 94(5), 573–600 CrossRef CAS.
- J. R. De Laeter, J. K. Boehlke, P. De Biever, H. Hidaka, H. S. Peiser, K. J. R. Rosman and P. D. P. Taylor, Pure Appl. Chem., 2003, 75, 683–800 CAS.
| This journal is © The Royal Society of Chemistry 2026 |





