A multi-distance shared calibration curve for quantitative analysis of manganese in high-manganese steel based on a portable remote LIBS instrument
Yue
Tang
a,
Haodong
Wu
a,
Kaiming
Sha
a,
Zixin
Zhang
a,
Mengjiao
Zhu
a,
Linjie
Luo
a,
Adil
Shahbaz
a,
Guanghui
Niu
b,
Qingyu
Lin
 *a and
Yixiang
Duan
*a and
Yixiang
Duan
 a
a
aResearch Center of Analytical Instrumentation, School of Mechanical Engineering, Sichuan University, No. 24, South Section 1, First Ring Road, 610064 Chengdu, Sichuan, China. E-mail: qylin@scu.edu.cn; Tel: +86-028-85403687
bBaowu Equipment Intelligent Technology Co., Ltd, Research and Development Center, 201900 Shanghai, China
First published on 27th November 2025
Abstract
Remote laser-induced breakdown spectroscopy (LIBS) has attracted significant research attention in the field of metallurgy due to its ability to enable rapid and remote elemental analysis. However, maintaining the stability of remote analysis and achieving reliable results across varying distances remain challenging. Here, an innovative portable LIBS instrument has been developed, with a modular design to reduce complexity and increase usability. The portable remote LIBS was employed to simultaneously detect multiple elements in steel samples at different standoff distances. A multi-distance shared calibration curve was developed to provide a simple and accurate quantitative method for different standoff distances. This method allows quantitative analysis at different standoff distances with only one calibration curve. The coefficient of determination of the multi-distance shared calibration curve is 0.9641 within the standoff distance range of 2000 mm to 2400 mm, with the lowest mean relative error being 0.67% for Mn quantification at the 2000 mm distance. In field environments, the relative standard deviation of Mn element spectral line intensity decreased from 9.89% to 7.48% and that of Cr element spectral line intensity dropped from 10.34% to 8.25%. This method has shown potential usefulness as analytical quality control for remote LIBS instruments. It may be further optimized for use in production processes requiring speed and stability analysis.
1 Introduction
High-manganese steel is a type of corrosion-resistant steel intended for use in heavy industry and its performance is directly linked to its constituent elements.1–3 Manganese levels determine the hardness and toughness of high-manganese steel, carbon affects strength and silicon affects resistance to oxidation. In addition, alloying elements such as chromium, molybdenum, and tungsten further control the performance, including corrosion resistance and high-temperature stability. Thus, rapid quantitative analysis of elements is essential for quality control in steel production lines, as these elements together determine the steel quality.The primary methods currently employed for analyzing the elemental composition of steel are X-ray fluorescence,4 inductively coupled plasma-atomic emission spectroscopy,5 and spark atomic emission spectroscopy.6 However, X-ray fluorescence spectroscopy requires contact measurements, while the other two methods require a complex sample preparation step, which makes it difficult to achieve on-line detection in the metallurgical industry. Laser-induced breakdown spectroscopy (LIBS or LIPS) is an atomic emission spectroscopy technique used for quantifying elements in materials. The main advantages of LIBS techniques are as follows: no or minimal sample preparation,7 rapid response,8 remote analysis9 and simultaneous measurement of multiple elements.10 Presently, LIBS is widely applied in various fields, including bioanalysis,11 space science,12 geology,13 environmental science14 and metallurgical analysis.15–17 Specifically, remote LIBS has attracted significant research attention in the field of metallurgy due to its ability to enable rapid and remote elemental analysis.18–20
Building on the advantages of LIBS mentioned above, early studies laid the foundation for the application of LIBS systems and the integration of quantitative algorithms in the practice of metallurgical analysis. López-Moreno et al. applied remote LIBS combined with a transfer function algorithm to analyze high-temperature steel slag samples, enabling quantitative analysis using room-temperature calibration curves, which supported real-time remote analysis in metallurgy.21 Similarly, Sturm et al. employed LIBS with multiple pulse excitation, demonstrating its potential in on-line analysis of light elements.22 In addition, automated LIBS systems with a 10–30 cm working distance could identify steel grades in rolling mills.23 Gonzaga et al. coupled the ultraviolet spectral region of a collinear quartz acousto-optical tunable filter (AOTF) to a photomultiplier and applied it to the determination of manganese in steel.24
Research on steel production scenarios has advanced further in the past few years. To address the classification of industrial steel, researchers used LIBS to investigate the effect of feature engineering on the robustness of the model. It was found that using spectral intensity ratios as input features significantly enhances the classification reliability.25 A coaxial remote LIBS system has been developed, which is combined with the k-nearest neighbor (kNN) algorithm, to achieve a 100% classification accuracy for unprocessed aerospace alloys.26 Zaytsev et al. proposed a multivariate calibration strategy based on Principal Component Regression (PCR) for the quantitative analysis of Si, Mn, Cr, and Ni in high-alloy stainless steels using LIBS,27 and the relative root mean square error of cross-validation (RMSECV) values were 2.8% for Mn, 3.1% for Cr, 5% for Ni, and 8% for Si, respectively. Luna et al. balanced detection accuracy and cost control, developed a self-made low-cost LIBS system and compared three multivariate regression models (full-spectrum PLS, VIP-PLS, and iPLS),28 thereby achieving direct quantitative analysis of Mn, Cr and Ni in steel, and the relative standard error of prediction (RSEP) values were 3.81% for Cr, 6.22% for Ni, and 10.32% for Mn, respectively.
To enhance the precision of remote LIBS for online elemental monitoring of steel manufacturing processes, Wang et al. proposed a pioneering coaxial laser beam measurement system that integrates LIBS and 3D contouring.29 This system is capable of automatically adjusting the focus according to the target contour. Cui et al. developed an open-path remote LIBS system with enhanced positional tolerance (±15 mm) for integration into steel production lines.30 This all-optical configuration enabled in situ manganese analysis in steel samples at elevated temperatures, demonstrating a 2.3-fold signal enhancement at 500 °C compared to ambient conditions. The system achieved 3.1% mean prediction error with 7.7% RSD under high-temperature industrial environments.
However, despite the significant progress achieved with remote LIBS, the main challenge remains the effective implementation of dynamic steel production scenarios. It is crucial to ensure the stability of the non-contact detection of steel and the reliability of quantitative results when the standoff distance fluctuates. Although various algorithms are generally used in LIBS, simplifying the calibration process is more suitable in real-world applications. Importantly, developing portable remote LIBS instrumentation is helpful in complex plant situations, as it offers greater flexibility and easier operation.
Herein, an innovative portable remote LIBS instrument has been developed. The portable instrument was designed with a modular approach in order to reduce complexity and increase its usability. In the meantime, the multi-distance shared calibration curve method, combined with an internal standard, has been optimized for the quantitative analysis of elements in high manganese steel. Field functional verification was carried out at the factory towards the roller in service. The results showed that the use of the internal standard method of multi-distance shared curves for steel element analysis can meet remote analysis requirements using portable LIBS instrument.
2 Materials and methods
2.1 Portable remote LIBS instrumentation
A schematic representation of the remote LIBS instrumentation is shown in Fig. 1(a). The excitation source is a Nd:YAG laser with a wavelength of 1064 nm, a pulse width of 6 ns, and a repetition frequency of 20 Hz. The single pulse energy is 10 mJ. The laser beam enters the Cassegrain telescope through a reflector and creates plasma on the sample. Plasma emissions are collected by the Cassegrain telescope. A spectrometer (AvaSpec ULS2048, Avantes) with a single channel is employed to detect the plasma emissions. The wavelength region of the spectrometer is 395–646 nm with a resolution of ∼0.15 nm. The integration time is set at 500 ms for each laser detection. The remote telemetry system adopts a Cassegrain reflective optical structure, composed of a parabolic primary mirror and a hyperbolic secondary mirror. The aperture of the telescope objective is 140 mm, the reflective telescope is coated with aluminum, the focal length of the primary mirror is 600 mm, and the magnification is 50 times. The secondary mirror is actuated by a stepper motor to alter the focal length, enabling the laser focusing point to be adjusted at different distances. | ||
| Fig. 1 Instrumentation of the remote LIBS. (a) Schematic of the remote LIBS, (b) remote LIBS design diagram, and (c) photograph of the portable remote LIBS instrumentation. | ||
To achieve industrial field measurements, reduce the complexity of the system and increase its practicality, a modular design approach is adopted. All modules of the remote LIBS were integrated in one unit. As shown in Fig. 1(b), this unit consists of a laser module, a host computer communication module, a spectrometer module, a laser ranging module, an optical path module, and a camera module. After assembly and packaging, the dimensions of the unit are around 536 × 303 × 508 mm, and the weight is about 13 kg. When assembled with a mechanical pan and tripod, it forms a complete set of portable remote LIBS instrument (see Fig. 1(c)).
2.2 Sample details and preparation
Eight certified reference materials of manganese steel were procured from the Research Institution of Mechanical Engineering (Zhengzhou, China). The performance indicators and technical requirements are strictly in line with the Chinese national standard (GB/T 5680-2023 High Manganese Steel Castings). The elemental composition of the samples was determined using the spark-discharge method. Samples (S1, S2, S3, S5, S7, and S8) were utilized to construct the calibration curves, while samples S4 and S6 were employed to assess the performance of the analysis method. The elemental composition of the samples is given in Table 1. Before measurement of the LIBS, the steel sample was subjected to a fine polish process with abrasive paper to remove any residual oxides. To perform the statistical analysis, each spectrum was obtained by averaging the signals of 10 ablation events on different sites.| Sample no. | High-manganese steel | Fe | Mn | Cr | Ni | Mo | W | C | Si |
|---|---|---|---|---|---|---|---|---|---|
| S1 | ZGMn7Cr1 | 90.26 | 6.02 | 0.94 | 0.58 | 0.74 | 0.31 | 0.73 | 0.25 |
| S2 | ZGMn10Cr2 | 83.64 | 9.84 | 1.56 | 1.62 | 1.08 | 0.49 | 0.78 | 0.60 |
| S3 | ZGMn13Cr2 | 84.78 | 11.83 | 1.51 | 0.02 | 0.00 | 0.00 | 1.21 | 0.54 |
| S4 | ZGMn13Cr2 | 77.17 | 13.97 | 1.91 | 2.43 | 1.39 | 0.76 | 1.07 | 0.77 |
| S5 | ZGMn18Cr2 | 77.95 | 16.90 | 2.20 | 0.20 | 0.18 | 0.09 | 1.20 | 0.66 |
| S6 | ZGMn18Cr2 | 76.22 | 18.70 | 2.00 | 0.59 | 0.25 | 0.08 | 1.30 | 0.46 |
| S7 | ZGMn22Cr2 | 71.63 | 22.39 | 1.23 | 1.69 | 0.58 | 0.49 | 1.10 | 0.67 |
| S8 | ZGMn24Cr2 | 69.53 | 24.20 | 1.86 | 1.65 | 1.01 | 0.04 | 1.20 | 0.35 |
2.3 Analytical method
The internal standard (IS) method was employed for quantitative analysis in this work. The IS calibration curve is built by plotting the analyte concentration on the x-axis, with the analyte/IS signal ratio on the y-axis.31 The process can be further clearly understood by analyzing eqn (1),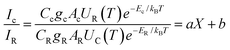 | (1) |
In order to find the best IS combination, each spectral line of the analytical element has been paired with all spectral lines of the Fe element in this work. Linear fitting was done for each pair using the least squares method and the corresponding R2 was calculated. By comparing all R2 values, the pair with the highest value was selected as the optimal IS combination, and its quantitative curve was defined as the optimal calibration curve.
The evaluation parameters of the quantitative model are the coefficient of determination (R2), the mean relative error (MRE), and the relative standard deviation (RSD). The parameters are expressed as follows:
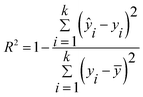 | (2) |
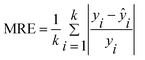 | (3) |
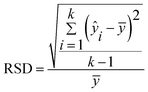 | (4) |
3 Results and discussion
3.1 Quantitative for portable remote LIBS instrumentation
Fig. 2 shows the LIBS spectra obtained at 2000 mm (with a distance accuracy of ±1 mm) from a sample of high-manganese steel. Emission spectral lines corresponding to Mn, Fe, Cr, Mo, and W are shown clearly. These spectral lines are summarized in Table 2. To achieve the quantitative analysis of elements in high-manganese steel, the spectra of six standard samples of high-manganese steel (S1, S2, S3, S5, S7, and S8) were used to construct the calibration curves and the remaining two samples (S4 and S6) were used to evaluate the analytical performance of the curve.| Line type | Wavelength (nm) | ||||
|---|---|---|---|---|---|
| Mn(I) | 403.07 | 403.30 | 404.13 | 475.40 | 476.23 |
| 476.65 | 478.34 | ||||
| Fe(I) | 404.58 | 406.35 | 430.79 | 432.57 | 438.35 |
| 440.47 | 516.74 | 526.95 | 532.85 | ||
| Cr(I) | 425.43 | 520.60 | 520.84 | ||
| Mo(I) | 550.65 | 553.30 | |||
| W(I) | 429.46 | ||||
The high-manganese steel sample was analysed at three different standoff distances (2000 mm ± 1 mm, 2200 mm ± 1 mm and 2400 mm ± 1 mm). The spectrum of S7 is shown in Fig. 3(a). The observed behavior is that the intensity of the element spectral line decreases as the distance increases (Fig. 3(b)). This suggests that the LIBS signals are apparently affected by changes in the standoff distance.
 | ||
| Fig. 3 LIBS spectra of the high-manganese steel (S7) at different standoff distances. (a) Wavelengths from 395 nm to 646 nm and (b) element lines of Mn and Cr. | ||
For quantitative analysis using elemental emission line intensities, a calibration curve shall be determined for each standoff distance. For the quantification of Mn content in the samples at different standoff distances, the Mn spectral line at 475.40 nm was used as the analytical spectral line. The correlation between Mn content and peak intensity at different standoff distances is shown in Fig. 4(a). Similarly, the Cr spectral line at 520.60 nm was used to quantify Cr content at different standoff distances, with the corresponding correlation between Cr content and peak intensity as shown in Fig. 4(b). It is noted that the R2 value of the calibration curves decreases with increasing standoff distance, affecting the quantitative performance. A separate calibration curve is required for each distance condition, making field analysis vulnerable to distance variations. The internal standard method could be used to solve this problem for elemental quantification.
 | ||
| Fig. 4 Calibration curves between element content and spectral line intensity at different standoff distances. (a) Mn and (b) Cr. | ||
Therefore, to achieve the optimal quantitative analysis, we further compared the combinations of different spectral lines of the target elements and different internal standard lines of Fe. The performance of the model was evaluated using R2, and the results are shown in Table 3. For Mn and Cr, the internal standard method can improve quantitative accuracy, and the optimal combinations for the IS series are Mn(I) 475.40 nm and Fe(I) 516.74 nm for Mn quantitative analysis, while Cr(I) 520.60 nm and Fe(I) 532.85 nm are used for Cr quantitative analysis. It should be noted that R2 for Mo did not improve after the use of the internal standard method and W content was relatively low, thus leading to poor quantitative performance. Therefore, these two elements will not be analysed further in the forthcoming studies.
| Element | Spectral lines (nm) | R 2 | Optimal internal standard line (nm) | R 2 |
|---|---|---|---|---|
| Mn | 403.07 | 0.9363 | 438.35 | 0.9985 |
| 403.30 | 0.9447 | 438.35 | 0.9983 | |
| 404.13 | 0.9491 | 438.35 | 0.9993 | |
| 475.40 | 0.9773 | 516.74 | 0.9999 | |
| 476.23 | 0.9759 | 438.35 | 0.9997 | |
| 476.65 | 0.9792 | 438.35 | 0.9998 | |
| 478.34 | 0.9733 | 438.35 | 0.9995 | |
| Cr | 425.43 | 0.5408 | 532.85 | 0.8104 |
| 520.60 | 0.7096 | 532.85 | 0.8979 | |
| 520.84 | 0.6597 | 532.85 | 0.8901 | |
| Mo | 550.65 | 0.8390 | 532.85 | 0.7445 |
| 553.30 | 0.7280 | 532.85 | 0.6200 | |
| W | 429.46 | 0.0459 | 532.85 | 0.3562 |
A better rationalization for IS selection and application would be highly useful in routine analyses. In remote LIBS measurements, after adopting the internal standard method, the influence caused by distance variation can be reduced, and the stability of the quantitative results can be improved. However, a series of calibration curves still need to be prepared for each standoff distance. Consequently, it is complex and impractical for real-world quantitative analysis. To enable quantitative analysis with the same calibration curve across different distances, we integrated the data collected at each distance and established a shared calibration curve coupled with the internal standard line. These multi-distance shared quantitative curves are shown in Fig. 5(a) and (b) for Mn and Cr, respectively.
To test the adequacy of the multi-distance shared calibration curve for Mn and Cr quantification at three different distances, the results of quantitative analysis for Sample 4 (S4) and Sample 6 (S6) at different standoff distances were obtained. The Mn and Cr true contents of S4 are 13.97% and 1.91%, respectively, while those of S6 are 18.70% and 2.00%. Five parallel samples were analysed under optimal experimental conditions. The predicted Mn and Cr contents using the calibration curve method based on peak intensity and the multi-distance shared method coupled with an IS are shown in Table 4.
| Sample | Distances | Mn | Cr | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Calibration curves | Multi-distance shared | Calibration curves | Multi-distance shared | ||||||
| Measure | MRE | Measure | MRE | Measure | MRE | Measure | MRE% | ||
| S4 | 2000 | 14.92 | 6.78 | 14.06 | 0.67 | 2.69 | 41.02 | 2.33 | 21.99 |
| 2200 | 15.56 | 11.41 | 14.30 | 2.39 | 2.92 | 52.80 | 2.63 | 37.69 | |
| 2400 | 12.20 | 12.69 | 14.33 | 2.56 | 2.16 | 13.19 | 2.48 | 29.84 | |
| S6 | 2000 | 17.12 | 8.45 | 18.55 | 0.79 | 2.20 | 9.99 | 2.38 | 19.23 |
| 2200 | 19.86 | 6.23 | 19.48 | 4.16 | 2.51 | 25.51 | 2.40 | 20.44 | |
| 2400 | 20.86 | 11.56 | 17.80 | 4.80 | 2.65 | 32.48 | 2.19 | 9.66 | |
The accuracy of each curve was evaluated according to the MRE values obtained with the two test samples. The lowest MRE for Mn was 6.23% obtained with the calibration curves based on peak intensity at a 2200 mm distance toward S6. However, the lowest MRE was 0.67% obtained with the multi-distance shared curve at a 2000 mm distance toward S4. It is suggested that the multi-distance shared curve method could provide an apparent improvement in quantitative accuracy. For Cr, the lowest MRE was 9.99% with the calibration curves based on peak intensity and 9.96% with the multi-distance shared curve. Although the multi-distance shared curve method shows limited improvement in Cr quantitative results due to the low Cr content of the samples, the results still demonstrate that it is possible to obtain quantitative results at different distances using the same curve.
3.2 Field validation for portable remote LIBS instrumentation
In order to perform field functional verification of the portable remote LIBS instrumentation, steel rolls from a Shanghai rolling plant were used as test samples (see Fig. 6(a)). The ambient temperature was room temperature, and the rolls were just taken off the production line with a smooth surface free of oxide layers. The standoff distance was set at 2000 mm. Different points were selected on the roll surface for LIBS analysis. As shown in Fig. 6(b), the instrumentation can effectively detect the target elements (Mn, Cr, and Fe) in the rolls. The spectral intensity variations of Mn and Cr are shown in Fig. 6(c), where the RSD value of Mn spectral line intensity is 9.89% and the RSD for Cr spectral line intensity is 10.34%. In addition, by taking Fe as an internal standard factor, the RSD values of Mn and Cr relative to Fe intensities are reduced to 7.48% and 8.25%, respectively (see Fig. 6(d)). The results showed that the use of the multi-distance shared curve internal standard method for the analysis of elements in steel can meet the requirements for remote analysis with the use of the portable LIBS instrumentation. However, further optimization is necessary to enhance its reliability in high-precision application scenarios.4 Conclusions
In conclusion, this work involved the design and construction of a portable remote LIBS instrument, used for standoff distance in situ analysis of steel samples. It successfully detected Fe, Mn, Cr, Mo, and W. Based on the internal standard method, a multi-distance shared calibration curve method was proposed to quantitatively analyze Mn and Cr in steel by identifying the optimal internal standard combination. Compared with calibration curves based on peak intensity, this innovative method only requires a single calibration curve to achieve quantification at different distances, with improved accuracy and stability. Additionally, on-site verification of the instrument and the method was conducted.This work provides a feasible approach for rapid elemental analysis in the production process of high-manganese steel. By realizing the rapid determination of manganese content at different distances, the portable remote instrument can optimize the LIBS detection process and provide support for the development of the steel industry. However, further research is still needed on the stability of analytical results under the influence of matrix movement and temperature changes.
Author contributions
Yue Tang: conceptualization, methodology, and investigation; Haodong Wu: data curation and conceptualization; Kaiming Sha: validation and software; Zixin Zhang: project administration and formal analysis; Mengjiao Zhu: formal analysis and visualization; Linjie Luo: investigation and writing – original draft; Adil Shahbaz: writing – review & editing; Guanghui Niu: investigation and resources; Qingyu Lin: supervision, writing – review & editing and funding acquisition; Yixiang Duan: project administration and resources.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.Acknowledgements
The authors are grateful for financial support from the National Natural Science Foundation of China (62475174) and Continuous-Support Basic Scientific Research Project (18BJ010261224900).Notes and references
- Y. K. Park, K. Y. Shin, K. Y. Lee and W. Lee, Steel Res. Int., 2024, 95, 2300492, DOI:10.1002/srin.202300492.
- M. Morawiec, J. Opara, C. Garcia-Mateo, J. A. Jimenez and A. Grajcar, J. Therm. Anal. Calorim., 2023, 148, 1567–1576, DOI:10.1007/s10973-022-11664-2.
- J. Chu, Y. Nian, L. Zhang, Y. Bao, N. Ali, C. Zhang and H. Zhou, J. Mater. Res. Technol., 2023, 22, 1505–1521, DOI:10.1016/j.jmrt.2022.12.023.
- K. Sanyal and S. Dhara, Anal. Sci., 2022, 38, 665–673, DOI:10.1007/s44211-022-00082-x.
- A. A. Belozerova, A. V. Mayorova, N. Y. Kalinina and M. N. Bardina, J. Anal. Chem., 2025, 80, 96–103, DOI:10.1134/S1061934824701570.
- A. Golestani, S. Sadeghi, H. Pourmahmoudi, M. Sohrabnejad, M. Feyzbakhshian, M. Zadsadeghi, M. Arabnejad and A. Bahaaldini, Anal. Sci., 2023, 39, 389–394, DOI:10.1007/s44211-022-00264-7.
- Q. Lin and Y. Duan, Chin. J. Anal. Chem., 2017, 45, 1405–1414, DOI:10.1016/S1872-2040(17)61040-5.
- H. B. Andrews, M. Z. Martin, A. M. Wymore and U. C. Kalluri, Plant Soil, 2024, 495, 3–12, DOI:10.1007/s11104-023-05988-7.
- S. Liu, C. Li, Q. He, H. Wu, X. Hu, B. Men, D. Wu, R. Hai, X. Wu and H. Ding, J. Anal. At. Spectrom., 2024, 39, 2363–2373, 10.1039/D4JA00208C.
- Q. Lin, P. Yin, Y. Duan, Y. Wang, L. Zhang and X. Wang, Opt. Express, 2022, 28, 14198–14208, DOI:10.1364/OE.387858.
- E. J. Blanchette, E. A. Tracey, A. Baughan, G. E. Johnson, H. Malik, C. N. Alionte, I. G. Arthur, M. E. S. Pontoni and S. J. Rehse, Spectrochim. Acta, Part B, 2024, 215, 106911, DOI:10.1016/j.sab.2024.106911.
- F. Chen, C. Sun, S. Qu, B. Zhang, Y. Rao, T. Sun, Y. S. Zhao and J. Yu, Spectrochim. Acta, Part B, 2024, 214, 106887, DOI:10.1016/j.sab.2024.106887.
- D. V. Babos, A. M. Tadini, C. P. D. Morais, B. B. Barreto, M. A. R. Carvalho, A. C. C. Bernardi, P. P. A. Oliveira, J. R. M. Pezzopane, D. M. B. P. Milori and L. Martin-Neto, Catena, 2024, 239, 107914, DOI:10.1016/j.catena.2024.107914.
- Y. Chen, J. Si, X. Wan, Y. Wang, A. Chen and M. Jin, Spectrochim. Acta, Part B, 2024, 213, 106880, DOI:10.1016/j.sab.2024.106880.
- M. Cui, G. Shi, L. Deng, H. Guo, S. Xiong, L. Tan, C. Yao, D. Zhanga and Y. Deguchi, J. Anal. At. Spectrom., 2024, 39, 1361–1374, 10.1039/D3JA00453H.
- K. K. Herrera, E. Tognoni, N. Omenetto, B. W. Smitha and J. D. Winefordner, J. Anal. At. Spectrom., 2009, 24, 413–425, 10.1039/B820493D.
- D. Diaz and D. W. Hahn, Spectrochim. Acta, Part B, 2020, 166, 105795, DOI:10.1016/j.sab.2020.105800.
- X. Cao, Y. Luo, Y. Zhao, J. Zhang and N. Li, Chin. J. Lasers, 2025, 52, 0611002, DOI:10.3788/CJL241126.
- T. Chen, L. Sun, H. Yu, L. Qi, P. Zhang and H. Dong, Analyst, 2024, 149, 4407–4417, 10.1039/D4AN00631C.
- Y. He, Y. Zhang, C. Ke, Q. Wen, S. Liu, D. Zhao, M. Xu, T. Xu and Y. Zhao, IEEE Trans. Instrum. Meas., 2023, 72, 1–8, DOI:10.1109/tim.2023.3300425.
- C. López-Moreno, S. Palanco and J. J. Laserna, Spectrochim. Acta, Part B, 2005, 60, 1034–1039, DOI:10.1016/j.sab.2005.05.037.
- V. Sturm, J. Vrenegor, R. Nolla and M. Hemmerlin, J. Anal. At. Spectrom., 2004, 19, 451–456, 10.1039/B315637K.
- R. Noll, C. Fricke-Begemann, S. Connemann, C. Meinhardt and V. Sturm, J. Anal. At. Spectrom., 2018, 33, 945–956, 10.1039/C8JA00076J.
- F. B. Gonzaga and C. Pasquini, Spectrochim. Acta, Part B, 2008, 63, 1268–1273, DOI:10.1016/j.sab.2008.09.014.
- G. Jeon, H. Keum, H. Lee, K. Oh and J. Choi, Spectrochim. Acta, Part B, 2024, 212, 106857, DOI:10.1016/j.sab.2024.106857.
- X. Liu, D. Zhang, Z. Feng, J. Ding, R. Yang, S. Sun, K. Wang, J. Zhu and Z. Wei, Acta Photonica Sin., 2021, 50, 1030003, DOI:10.3788/gzxb20215010.1030003.
- S. M. Zaytsev, A. M. Popov, E. V. Chernykh, R. D. Voronina, N. B. Zorova and T. A. Labutin, J. Anal. At. Spectrom., 2014, 29, 1417–1424, 10.1039/C3JA50389E.
- A. S. Luna, F. B. Gonzaga, W. F. C. da Rocha and I. C. A. Lima, Spectrochim. Acta, Part B, 2018, 139, 20–26, DOI:10.1016/j.sab.2017.10.016.
- Z. Wang, Y. Deguchi, F. Shiou, S. Tanaka, M. Cui, K. Rong and J. Yan, ISIJ Int., 2020, 60, 971–978, DOI:10.2355/isijinternational.ISIJINT-2019-317.
- M. Cui, Y. Deguchi, Z. Wang, S. Tanaka, M. Jeon, Y. Fujita and S. Zhao, Plasma Sci. Technol., 2019, 21, 034007, DOI:10.1088/2058-6272/aaeba7.
- N. B. Zorov, A. A. Gorbatenko, T. A. Labutin and A. M. Popov, Spectrochim. Acta, Part B, 2010, 65, 642–657, DOI:10.1016/j.sab.2010.04.009.
| This journal is © The Royal Society of Chemistry 2026 |



