 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Quantification of spacecraft heatshield contaminants seen in reentry shock layer emissions using calibration-free LIBS
Keira J. Leistikowa,
Noshin Nawar b,
Justin I. Borrero-Negrón
b,
Justin I. Borrero-Negrón c,
Marat Kulakhmetovd,
Paolo Valentini
c,
Marat Kulakhmetovd,
Paolo Valentini b and
Ashwin P. Rao
b and
Ashwin P. Rao *b
*b
aUniversity of California San Diego, La Jolla, CA, USA
bSpace Vehicles Directorate, Air Force Research Laboratory, Kirtland Air Force Base, NM, USA. E-mail: ashwin.rao.1@spaceforce.mil
cNuclear Engineering Program, University of Florida, Gainesville, FL, USA
dVarda Space Industries, El Segundo, CA, USA
First published on 6th February 2026
Abstract
Shock layer thermochemistry during atmospheric reentry of spacecraft is strongly influenced by the composition of thermal protection system (TPS) materials. In this study, we implement calibration-free laser-induced breakdown spectroscopy (CF-LIBS) to profile cross-sections of phenolic impregnated carbon ablator (PICA) and room temperature vulcanizing (RTV) silicone from the heatshield of a commercial reentry capsule. CF-LIBS measurements determine that these materials contain alkali and alkaline earth contaminants (e.g., Na, Ca) at levels up to 10 parts-per-million (ppm) and heavier metals like Fe at 103 ppm. Contaminant concentrations are linked to observed strong atomic radiators identified from in situ shock layer emissions taken during reentry, demonstrating the value of CF-LIBS for interpreting complex shock-layer emission data from reentry spacecraft.
1 Introduction
Spacecraft thermal protection systems (TPS) must withstand extreme aerothermodynamic conditions during atmospheric reentry. The bow shocks ahead of reentry bodies heat the surrounding air to temperatures in excess of 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 K and create high-enthalpy flowfields with thermochemical nonequilibrium.1,2 The combined radiative and convective heating from these flowfields can exceed 1 kW cm−2 on the surface of the spacecraft.3 Ablative TPS, such as phenolic impregnated carbon ablator (PICA), mitigate this thermal loading by absorbing thermal energy through endothermic processes, which decompose the heatshield into high temperature volatile species.4 These gaseous products pass through the TPS into the shock layer, dissipating heat from the spacecraft surface into the surrounding air flow. These molecules undergo further complex chemical interactions with shock-heated air species, influencing the evolution of shock layer chemistry and radiative emissions during reentry.5,6
000 K and create high-enthalpy flowfields with thermochemical nonequilibrium.1,2 The combined radiative and convective heating from these flowfields can exceed 1 kW cm−2 on the surface of the spacecraft.3 Ablative TPS, such as phenolic impregnated carbon ablator (PICA), mitigate this thermal loading by absorbing thermal energy through endothermic processes, which decompose the heatshield into high temperature volatile species.4 These gaseous products pass through the TPS into the shock layer, dissipating heat from the spacecraft surface into the surrounding air flow. These molecules undergo further complex chemical interactions with shock-heated air species, influencing the evolution of shock layer chemistry and radiative emissions during reentry.5,6
Varda Space Industries, an American space manufacturing startup, has developed a ballistic reentry capsule to enable commercial orbital manufacturing needs and serve as a standard hypersonic testbed for the research community. The testbed helps develop advanced heat shields, flight sensors, and communication systems, while providing an avenue for data collection to advance state-of-the-art modeling of hypersonic flows. On the 28th of February 2025, the Varda's W-2 capsule successfully executed an orbital reentry flight test into Koonibba Test Range, South Australia. W-2 housed an optical emission spectroscopy (OES) payload developed by the U.S. Air Force Research Laboratory called: Optical Sensing of Plasmas in the ReEntry Environment (OSPREE).7,8 The OSPREE payload captured Vis-NIR (340–800 nm) in situ spectral emission data of the reentry shock layer at speeds in excess of Mach 25, providing critical data needed for the development and validation of ab initio hypersonic air chemistry models.9 Interestingly, OSPREE detected strongly radiating atomic emissions of metallic elements such as Na and Fe in the reentry shock layer, which were not expected from the predicted heatshield ablation chemistry. The capsule forebody TPS chars and pyrolyzes in flight, injecting material from the heatshield into the flowfield. Thus, it became important to characterize the constituents of the capsule TPS to aid post-flight data analysis and support designs of future flight experiments on the Varda testbed.
Chemical makeup of the Varda capsule forebody heatshield is analyzed using laser-induced breakdown spectroscopy (LIBS). LIBS has emerged as a powerful method for in situ material characterization, requiring little to no sample preparation and providing rapid, efficient, and sensitive field analyses.10,11 Focusing a pulsed laser with moderate to high irradiance levels (∼GW cm−2) onto the surface of a target ablates the sample surface, and generates a laser-produced plasma (LPP). In this technique, the LPP typically exhibits extreme temperatures greater than 103 K and less than 105 K and high electron densities greater than 1020 m−3 up to approximately 1025 m−3 over a broad timescale relative to the laser pulse duration (e.g., ns-, ps-, or fs-pulsed laser).12 The resultant luminescent plasma is composed of excited atoms and molecules, which produce optical emission signatures at specific wavelengths characteristic of the target's elemental composition, making LIBS a viable method to analyze chemical composition of various materials. In prior literature, LIBS has proven to be a versatile chemical analysis tool with applications in geochemistry,13,14 nuclear science,15–19 metallurgy,20 and environmental science.21,22 LIBS has also been used to study chemical kinetics of high-temperature materials, such as carbon composites, in plasma environments.23
A method called calibration-free LIBS (CF-LIBS) was introduced by Ciucci et al.24 to overcome the limitations of LIBS due to quantitative uncertainties arising from emission signal dependencies on the sample matrix in which an element is embedded, or “matrix effects”. CF-LIBS removes the need to perform measurements of a standard sample set and relate changes in LIBS spectral emission intensity to known changes in analyte quantity. Instead, CF-LIBS proposes using analytical equations with measured line intensities and tabulated transition parameters to determine elemental concentrations from measured spectra.25 CF-LIBS has been widely reported as a useful analytical method across a wide range of materials.26–30 Detection limits below the single part-per-million have been reported, although limit of detection in CF-LIBS appears to vary between studies due to dependent on experimental parameters such as laser-produced plasma emitter densities and plasma temperature.31–33 Nevertheless, CF-LIBS presents a promising avenue for investigating elemental contaminants in spacecraft heatshields where a calibration standard sample set is not available.
We present the implementation of CF-LIBS to identify and quantify metallic elemental contaminants in materials comprising the forebody heatshield of the W-2 reentry capsule. The identified spectral emissions and calculated concentrations of these elements are compared directly to OES data obtained during atmospheric reentry. The laboratory measurements are used to explain the source of these metallic emissions seen in shock layer OES data. The aerodynamics of the vehicle and gas–surface interactions during reentry are discussed to explain the physical mechanisms introducing these metals into the reentry plasma and their effects on shock layer thermochemistry.
2 In situ OES observations during atmospheric reentry
The OSPREE OES payload used an Ocean Optics HR2 spectrometer to obtain Vis-NIR (340–800 nm) in situ spectra of the shock layer during an atmospheric reentry mission. Optical emissions were obtained from a line of sight (LOS) on the shoulder of the vehicle through an optical window on the capsule backshell. Fig. 1 provides a fluid dynamics solution contour plot of the vehicle showing the exact LOS geometry from the vehicle aft dome into the shock layer. Full details of the design of the payload and mission can be found in Rao et al. (2025),7 while comprehensive results of the mission including a full analysis of collected OES data can be read in ref. 34. Fig. 2(a) shows a raw spectrum taken during flight at 94.2 km. | ||
| Fig. 1 Flowfield translational temperature contour plot of W-2 cross section with annotated optical line of sight into the shock layer used for emission collection. | ||
The spectrum exhibits many molecular emission features (CN, CH, C2) resulting from breakdown and interaction of PICA pyrolysis products with the surrounding shock heated air, as well as two strong atomic emissions at 589 nm and 623.3 nm identified as neutral Na and Fe, respectively. Multiple smaller emissions of Fe I are seen at 487.3 nm, 529.5 nm, and 552.6 nm. Further investigation of weaker emissions in the data set revealed the presence of the Ca I emission at 422.6 nm. The presence of strong atomic emissions of alkali, alkaline earth, and other metallic species in the flight data was unexpected, and analysis of the measured spectral radiance over time indicated continuous injection of these elemental contaminants into the shock layer during flight. Fig. 2(b) shows the spectral radiance of Fe I (623.3 nm), Na I (589 nm), and Ca I (422.6 nm) from 620–810 s into reentry. This data reflects strong spectral radiance from Fe and Na peaking around 80 km, with minor contributions from Ca throughout reentry. Based on the time-evolving radiance trends, it was hypothesized that these atomic emissions stem from contaminants in the vehicle forebody heatshield which are introduced into the shock layer through the various chemical processes accompanying pyrolysis, ablation, and heatshield decomposition during flight.
The presence of metallic impurities in the vehicle TPS has significant effects on shock layer chemistry and radiative heating experienced by the capsule during reentry. Alkali and alkaline earth metals, such as Na, are readily ionized and exhibit large electron-impact cross sections.35 Bortner 1964 is one of the sole reports detailing the effects of Na on hypervelocity flow chemistry.36 Two representative reactions for Na impact ionization and Na/NO charge exchange are shown below.
| Na + e− → Na+ + e− + e− |
| NO+ + Na → NO + Na+ |
The presence of Na in hypervelocity flows can increase shock layer electron density as a result of such reactions.36,37 Augmented electron density values during reentry have major implications on radiofrequency communications. Because the plasma cutoff frequency is directly related to electron density, electromagnetic waves below the plasma frequency attempting to penetrate a plasma layer will be attenuated.38 Thus, contributions of alkali metals to shock layer chemistry can have non-trivial second order effects. Na is easily excitable due to its electronic structure, and the D1/D2 doublet presents as a strong radiator even when Na is only present in trace quantities.39 This feature was observed in ex situ spectral observations of the Stardust40 and Hayabusa2 (ref. 41) reentries. Transition metals such as Fe also present as strong radiators due to their dense line emission across the UV-Vis-NIR. Lewis et al. observed this in graphite ablation experiments in the X-2 expansion tunnel, noting that Fe contamination in measured emission spectra contributed between 20–40% of the total radiance in the short-wavelength visible band.42,43 Because radiative emission strength is directly related to incident radiative heat flux on the surface, the presence of strong radiators in the shock layer can increase the radiative heat load experienced during reentry, and their effects must be accounted for in vehicle design. The appearance of these emissions in the OSPREE flight data has sparked great interest in characterizing the W-2 forebody TPS to fully assess the sources of these metallic species and quantify their presence in the heatshield.
3 Experimental methodology
3.1 Materials
In this study, pristine samples of two TPS materials used on the W-2 forebody were investigated: PICA and RTV silicone. This was done to best mimic the state of the heatshield immediately prior to reentry and understand the true native concentrations of metallic impurities in a newly assembled vehicle. PICA is a legacy NASA material created by impregnating a carbon fiber preform with phenolic resole resin.44 With its low density and high ablative performance, PICA and its variants have been used in several planetary entry vehicles that include: Stardust, Mars Science Laboratory, OSIRIS-REx, Dragon, and Dragonfly.45,46 The W-series capsule forebodies are assembled in tiled sections rather than as a monolithic shell. A high temperature RTV silicone is used as a gap-filler between PICA tile seams. Fig. 3(a) and (b) show the cross sections of the PICA and RTV samples investigated in this study. The particular PICA sample analyzed in this experiment contains a waterproof coating, roughly 12 mm in depth, to mitigate moisture absorption.3.2 CF-LIBS setup
The experimental setup, shown in Fig. 4, used a New Wave GEMINI 1064 nm Nd:YAG laser with a pulse width of 10 ns and an energy output of 80 mJ per pulse at a repetition rate of 10 Hz to induce breakdown in and ablate the PICA and RTV samples. The 5 mm diameter beam is focused into a 6 × 6 × 6 in. vacuum cube with a plano-convex lens with f = +110 mm, yielding a 30 µm spot size on the sample. This process was performed under vacuum at a pressure of 80 mTorr to decrease the presence of molecular emissions (e.g., CN, C2) resulting from reactions between air molecules and atomic constituents of the samples produced from ablation. Furthermore, conducting LIBS at lower pressure or vacuum conditions has been shown to reduce or eliminate the effects of self-absorption, improving quantitative accuracy.47–50 LIBS emissions were captured using a secondary lens with f = +70 mm and a 74-VIS Ocean Optics collimator. An Echelle spectrograph (Catalina Scientific OWL 325/65) with an ICCD camera (Raptor Photonics Condor) set to 1 ms exposure time, 10 µs gate width, 250 ns gate delay, and MCP gain of 1500 were used for spectral emission capture from 200 nm to 800 nm with Δλ = 0.01 nm. It is standard to use a larger gate width in minor element CF-LIBS quantification to improve signal-to-noise ratio and thus better resolve weak emissions.51 A digital delay generator (DDG) synchronized the triggering of the ICCD with each laser pulse. An automatic background subtraction was performed in the camera software with the acquisition of each spectrum. A 1-D raster of the ablation point through the length of the sample cross section was conducted using a tri-axis translation stage plate made for the vacuum cube. The micrometer dial was turned to shift the measurement point 0.159 mm per step. 1570 spectra were collected from the PICA sample with 10 shots taken at 157 total locations through the cross section. 320 spectra were collected from the RTV sample with 10 shots taken at 32 total locations. Each set of 10 shots were averaged at every location of the depth profile of both samples, as is standard when using LIBS for quantification of minor elements.51 | ||
| Fig. 4 Diagram of LIBS measurement setup implemented to obtain cross-sectional profile measurements of TPS samples. | ||
4 Spectral line identification
Fig. 5–8 show the collection of averaged spectra from each location on the PICA sample and Fig. 9–11 show the averaged RTV spectra broken up into various wavelength ranges, highlighting the distinct spectral features observed in each sample. Atomic emission lines were identified using data tabulated in the NIST Atomic Spectral Database.39The PICA spectrum is characterized by several Si I emissions, along with C I, Ca II, and Al I lines in the UV (Fig. 5 and 6). Further into the Vis-NIR, Hα (656 nm), the Na D1/D2 doublet (588/589 nm), Si II, and O I at 777 nm are observed (Fig. 7 and 8). The presence of C, H, and O emissions stem from the atomic constituents of the phenolic resin. Si, Na, Ca, and Al are not expected in PICA, and were thus labeled as elemental impurities.
In the RTV spectra, Si I, C I, Ca II, and Al I emission lines are identified in the UV along with several Fe I and Fe II emissions (Fig. 9 and 10). In the visible range, the RTV spectra exhibit Na I and Si II lines along with Hα (Fig. 11). K I and O I emissions lines are also seen in the NIR (Fig. 11). The molecular structure of RTV silicone accounts for the presence of Si, C, O, and H, while iron oxide additives are included in the RTV for high-temperature applications account for the presence of Fe. Thus, elemental contaminants found within the RTV are identified as Ca, Al, Na, and K. The spectral line identification of the LIBS data yields insight into the unexpected presence of several strong radiative emission lines observed in the shock layer OES data obtained by OSPREE, indicating that these elements are introduced into the flowfield by the TPS. Since OSPREE's spectral bandwidth did not extend into the UV, the numerous Si emissions seen in the LIBS data were not observed in flight. Al and K were not directly observed during reentry, as emissions from these elements were likely occluded by nearby molecular emission bands from N2 and O2. Thus, the initial elemental identification indicates that the Fe, Na, and Ca see in the flight OES data from W-2 stem from constituents of the capsule TPS. Table 1 summarizes the main species emissions identified in the LIBS spectra selected for CF-LIBS analysis.
| Species | λ (nm) | gkAki (s−1) | Ei (eV) | Ek (eV) | Sample |
|---|---|---|---|---|---|
| Si I | 288.14 | 6.51 × 108 | 0.7810 | 5.0823 | PICA, RTV |
| Si II | 634.68 | 2.34 × 108 | 8.1210 | 10.0739 | RTV |
| Na I | 588.96 | 2.46 × 108 | 0.0000 | 2.1044 | PICA, RTV |
| Ca I | 422.67 | 6.54 × 108 | 0.0000 | 2.9325 | PICA, RTV |
| Ca II | 393.36 | 5.88 × 108 | 0.0000 | 3.1509 | PICA, RTV |
| Al I | 396.14 | 1.97 × 108 | 0.0139 | 3.1427 | PICA, RTV |
| C I | 247.85 | 8.40 × 107 | 2.6840 | 7.6848 | PICA, RTV |
| H I | 656.19 | 7.93 × 108 | 10.1988 | 12.0875 | PICA, RTV |
| O I | 777.20 | 2.58 × 108 | 9.1461 | 10.7409 | PICA, RTV |
| Fe I | 404.55 | 7.76 × 108 | 1.4849 | 4.5485 | RTV |
| Fe II | 259.93 | 2.35 × 109 | 0.0000 | 4.7683 | RTV |
| K I | 766.48 | 1.51 × 108 | 0.0000 | 1.6171 | RTV |
5 CF-LIBS analysis and results
5.1 Establishing pLTE conditions
In LIBS, the use of quantitative analysis methodologies dependent on Boltzmann statistics is valid if the LPP is determined to be in partial local thermodynamic equilibrium (pLTE). The criterion shown in eqn (1), developed by Fujimoto and McWhirter,52 is implemented to determine if pLTE conditions are met.
 | (1) |
In this relation, T refers to the plasma temperature (K) and ΔEki represents the maximum energy difference between the two consecutive excited states (eV) of the spectral lines used. If the electron density (ne) exceeds the right-hand side (RHS) of the equation then the LPP meets the pLTE criterion. Plasma temperature is determined using the Boltzmann method per eqn (2),
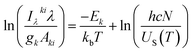 | (2) |
 | (3) |
 , independent of the y-intercept, and therefore the total species population N. The species concentrations were determined from this calculated temperature using the measured line intensities in conjunction with the calculated partition function, tabulated transition parameters, and the evaluated temperature.
, independent of the y-intercept, and therefore the total species population N. The species concentrations were determined from this calculated temperature using the measured line intensities in conjunction with the calculated partition function, tabulated transition parameters, and the evaluated temperature.
An abundance of Si I emissions listed in Table 2, were identified in the spectra of both samples and used to calculate temperature for each measurement location. Because the Boltzmann method extracts the plasma temperature from the slope of a linear regression fit dependent on upper-level energies of the emission lines, a reliable temperature determination generally requires emission lines spanning a broad range of excitation energies.53 In this study, due to the wider gate width employed in the LIBS measurements to improve SNR, we employed the selection criteria proposed by De Giacomo et al. for calculating temperature by excluding (i) resonant lines, (ii) lines with a transition probability less than 2 × 106 s−2, (iii) ionic lines, and (iv) lines with NIST tabulated relative intensities >3000.54 These selection criteria, along with demonstrating the validity of the McWhirter criterion, provide sufficient confidence of the LTE conditions and the validity of Boltzmann statistics to use CF-LIBS for elemental quantification. The Saha–Boltzmann approach is also known to be more accurate than the traditional Boltzmann method due to the broader range of energies from neutral and ionized species transitions,55 but it was not feasible to use in this study as too few ionized lines were observed or easily resolved support a reliable fit. Fig. 12(a and b) show selected Boltzmann plot fits parameters, and uncertainty metrics for selected measurement locations on both samples.
| λ (nm) | gkAki (s−1) | Ei (eV) | Ek (eV) |
|---|---|---|---|
| 212.41 | 2.08 × 109 | 0.7810 | 6.6161 |
| 221.65 | 3.18 × 108 | 0.0277 | 5.6192 |
| 243.49 | 2.22 × 108 | 0.7810 | 5.8708 |
| 250.68 | 2.74 × 108 | 0.0096 | 4.9548 |
| 251.60 | 8.40 × 108 | 0.0277 | 4.9538 |
| 251.93 | 1.65 × 108 | 0.0096 | 4.9296 |
| 252.41 | 2.22 × 108 | 0.0096 | 4.9201 |
| 252.86 | 2.71 × 108 | 0.0277 | 4.9296 |
| 263.11 | 3.18 × 108 | 1.9087 | 6.6191 |
| 288.14 | 6.51 × 108 | 0.7810 | 5.0823 |
| 390.52 | 3.99 × 107 | 1.9087 | 5.0823 |
 | ||
| Fig. 12 Boltzmann temperature plot with fit error bounds, fit equation and uncertainties, and temperature value with error for (a) PICA measurement location 157 and (b) RTV measurement location 10. | ||
Using the calculated plasma temperatures, the electron density of the LPP at each measurement location of both samples was calculated from the Stark broadening of the Si I emission at 288.4 nm using eqn (4), where Δλ is the measured broadening of the line and w is the impact width parameter tabulated in Griem.56
 | (4) |
Using the McWhirter criterion with the calculated temperatures and densities at each shot location, the LHS of eqn (1) ranged from 6.65 × 1016 ± 1.79 × 1015 to 7.58 × 1016 cm−3 ± 1.16 × 1016 for PICA and 6.65 × 1016 ± 3.19 × 1015 to 6.94 × 1016 ± 1.25 × 1015 cm−3 for the RTV shot locations. The RHS ranged from 2.15 × 1016 ± 1.08 × 1015 to 2.48 × 1016 ± 1.02 × 1016 cm−3 for PICA and 2.58 × 1016 ± 3.44 × 1015 to 2.76 × 1016 ± 2.54 × 1015 cm−3 for RTV. In this experiment, given that the calculated McWhirter criterion for all PICA and RTV locations is always lower than the computed electron density, the pLTE condition is validated for all data points. This is visually presented in Fig. 13(a and b) which shows the LHS and RHS quantity of the criterion calculated for each measurement location on both samples. Thus, the plasma temperatures at each measurement location calculated via the Boltzmann method with the Si I lines can be used to describe the LPP temperature for each respective location.
 | ||
| Fig. 13 Comparison of the LHS and RHS of the McWhirter criterion computed for each measurement location of (a) PICA and (b) RTV samples, visually demonstrating validity of eqn (1). | ||
It should be noted that, in this study, a large gate width of 10 µs was selected primarily to discriminate against the continuum emission for minor and trace element detection. This is a standard practice57–59 in traditional LIBS setups. Although the closure relation (i.e., ∑CS = 1) discussed in Section 5.2 has an effect on composition estimates for minor and trace elements due to error differences with major (abundant) elements, these errors related to insufficient signal-to-noise ratio are more pronounced.60
To address the issue related to the LTE assumption and sufficiently large gate for high SNR, De Giacomo et al.54 proposed the following mitigation. The excitation temperature obtained from the Boltzmann plot can be treated as an average across time if the observed emissions are approximately constant during the selected interval of detection. Given the long integration forced by a 10 µs gate for sufficient SNR, this necessitates the following selection criteria:
(1) Emission lines involving the ground state were excluded to minimize resonance effects. From the original and revised manuscripts and all analyses, Si I at 220.8 nm and 251.41 nm were removed.54
(2) Transitions with spontaneous emission rates (Aki) < 2 × 106 s−1 were removed such that all emission times are comparable to the associated time for transient plasma variations.54,61
(3) Due to deviations which can result from the Saha equation for ionic emissions related to recombination during expansion, only atomic (neutral) emissions were considered. This is inherent to the Boltzmann plot method.54,61
(4) All emission lines with tabulated relative intensities >3000 were excluded to filter out intensity values affected by noise and ensure sufficiently high SNR to ensure less significant effect on temperature calculations. Emissions with high intensity values are recommended to be excluded because they may be affected by broadening and saturation effects.53,54
In addition to the pLTE conditions confirmed through the McWhirter criterion, these mitigation methods ensure that LTE conditions are met, and the Boltzmann method applied is valid.
5.2 CF-LIBS methodology
Assuming a LPP in partial local thermodynamic equilibrium that is optically thin, the intensity of an emitted line (Iλki) is expressed by eqn (5),24
 | (5) |
 | (6) |
 | (7) |
Determining F from eqn (6) using the parameters for every identified emission in the spectrum then allows the calculation of the relative concentration of each individual species from eqn (5). At each shot location, the temperature determined from the Si I lines in Section 5.1 was used for the calculation of the partition function of each species and in eqn (5). This method is valid due to the fulfillment of the criterion for pLTE demonstrated in the previous section. Thus, a Boltzmann plot for each species was not required, and the parameters from the emission lines in Table 1 were used with eqn (5) to determine each relative species concentration. This method is demonstrated in literature in several prior studies, also referred to as one-line CF-LIBS.62–64 Cavalcanti et al.65 specifically implements this methodology and cites the McWhirter criterion52 to justify the assumption of a single temperature and the use of eqn (5) to determine species concentrations. The associated line parameters necessary for this calculation (e.g., upper level energy, transition probability) are found using the NIST database. This analytical process was repeated for the PICA and RTV samples separately at each location along the depth profile due to variations in sample composition.
Where applicable, the contributions of different ionization states (e.g., Si I and Si II) of an element are added together to determine a total relative concentration for the individual species (CTOTSi = C(I)Si + C(II)Si).24 The validity of this approach relies on the widely adopted, first-order approximation for CF-LIBS that the observed emission spectrum captures the complete sample composition.66 It should also be noted that, while resonance lines are generally prone to self-absorption effects (e.g., Na I and K I in Table 1), this issue can become negligible at sufficiently low pressures, and is thus not accounted for in the CF-LIBS algorithm of this study due to the vacuum conditions under which measurements were taken.67
5.3 Cross-section profiles of PICA and RTV
Fig. 14(a) and (b) show the concentrations in parts-per-million (ppm) (Cs) of each main species listed in Table 1 calculated using eqn (5) as a function of cross-sectional distance through each sample. Error bars are determined using shot-to-shot standard deviation of peak area at each location, propagated according to basic statistical error formulae. As expected, PICA is mainly composed of C, H, and O with respective concentration ranges of 6.2 × 103 ± 7.7 × 102–2.0 × 105 ± 1.3 × 105 ppm (C), 5.5 × 105 ± 9.1 × 104–9.1 × 105 ± 3.2 × 105 ppm (H), and 7.0 × 104 ± 3.0 × 104–3.2 × 105 ± 3.8 × 104 ppm (O) across the sample. The calculations reveal an interesting result in the unexpectedly high concentration of Si ranging from 4.21 ± 2.32 to 230 ± 46.0 ppm across the sample. The Si concentration presents at a higher concentration on the order of 102 ppm from 0–12 mm into the cross section, and then drops to 101 ppm across the remainder of the profile. The inverse of this trend is seen in the C concentration, which rises from ∼104 to 105 ppm after 12 mm. We attribute this trend to the use of a proprietary waterproof coating used on the PICA tiles prior to assembly of the heatshield. The coating likely penetrates into the tile through the layers of carbon fibers, causing the higher Si concentrations observed in the first 12 mm of the sample. The root cause of the presence of Si in the uncoated PICA is still unknown and will be the subject of further investigation into these materials. The remaining identified elemental contaminants are Ca, Al, and Na, present in trace concentrations of 4 × 10−5 ± 6 × 10−4–0.01 ± 0.05 ppm (Ca), 6.5 × 10−3 ± 6.0 × 10−4–0.91 ± 0.14 ppm (Al), and 1 × 10−5 ± 9 × 10−5–0.010 ± 0.019 ppm. The larger error bars in the calculated quantities of these three elements are a direct result of higher shot-to-shot variation in the emission line intensity of these minor analytes across the sample. These results reflect previous reporting of lower accuracy in trace level analyte quantification using CF-LIBS, particularly when the analyte concentration is below the single part-per-million level.31–33 Although the consistent appearance of the emissions of these elements in the recorded spectra provides confidence of their presence across the sample, the larger the uncertainty of their calculated concentration is critical to note when performing quantitative analysis. If truly high fidelity quantification is required, these results should be compared to concentrations determined from robust mass spectrometry methods as a validation. | ||
| Fig. 14 Species concentrations across linear cross sections of samples of (a) PICA and (b) RTV silicone investigated in this experiment. | ||
The RTV linear profile composition depicted in Fig. 14(b) indicates the sample is composed primarily of H, O, C, and Si. This is expected as the molecular structure of silicone consists of a siloxane backbone (Si–O–Si–O) and attached organic groups such as methyl (CH3). The concentrations of the 4 major elemental constituents are relatively constant across the measurement, presenting average values of 6.2 × 105 ± 1.8 × 105 ppm (H), 2.7 × 105 ± 2.2 × 104 ppm (O), 8.4 × 104 ± 3.2 × 104 ppm (C), and 3.0 × 104 ± 2.6 × 103 ppm (Si). The variant of RTV silicone used in this study contains iron oxide as an additive for high temperature applications. It is assumed that iron oxide is mixed during manufacturing such that it is homogeneous in the final material. A pronounced presence of Fe, averaging 2.7 × 103 ± 5.9 × 102 ppm, was measured, consistent with the radiant emission of Fe observed in the shock layer during reentry. While Fe is not an intrinsic contaminant of the base material, it is designated as a such due to its anomalous radiative emission in the context of this experiment. Multiple alkali and alkaline earth (Ca, Na, K) metals as well as Al are present through the sample. Al and Ca appear with average concentrations of 21.0 ± 13.0 and 11.0 ± 6.0 ppm, respectively. Na presents with an average concentration of 7.1 ± 2.4 ppm, while K is seen at the lowest average concentration of 0.71 ± 0.7 ppm. The average concentration of Na calculated in the RTV sample is 2 orders of magnitude greater than that in the PICA, helping to explain the persistent radiance of the Na doublet reflected in Fig. 2. Similarly, Ca is found in the RTV at concentrations 3 orders of magnitude higher than those of PICA. Based on these observations, we posit that much of the apparent radiance of these metallic species observed in the flight data originates from shock-heated air interacting with the RTV silicone, with minor contributions from the release of trace metal impurities during pyrolysis and ablation of the PICA matrix. These measurements reflect the utility of the CF-LIBS method and the value in performing benchtop analyses of TPS material composition. A full understanding of the TPS sample chemistry elucidates the causes of the unexpected spectral emissions seen in the flight data, helping to better correlate the effects of these impurities on shock layer thermochemistry. A comprehensive tabulation of the measured elemental concentrations at each location for both samples is provided in the SI file for this manuscript. To further investigate the physical phenomena corresponding to the release of these species during flight, we examine the fundamental chemical properties of these elements as well as the aerodynamics of the vehicle.
6 Discussion on aerothermodynamic phenomena and effects of metallic contaminants in the shock layer
The Varda capsule forebody is comprised of three tiled sections: one nose tile, followed by concentric rows of midsection and shoulder tiles. Fig. 15(a) shows the forebody of W-2. The middle section tile gap in this image was directly in line with the collimating lens of OSPREE placed behind the optical window above the shoulder section. Thus, it is highly likely that ablated material from the PICA tiles and the RTV between the tiles was observed in the recorded shock layer emissions. The thermal decomposition of the PICA tiles, through processes such as pyrolysis and ablation, introduce C, H, O, and trace metallic species into the shock layer. These species in turn undergo chemical reactions with shock heated air molecules, as well as collisional and radiative excitation and ionization. These processes contribute to the carbon molecular emission signatures seen in Fig. 2(a), in addition to some of the early Ca, Fe, and Na atomic emission lines. Since the nose tile of the capsule experiences the highest heat flux (300 W cm−2) at the stagnation point, it is expected to contribute to most of the early shock layer trace species emissions.In addition to heatshield decomposition from thermal loading, aerodynamic effects in gaps between the forebody tiles contribute to the presence of metallic species emissions in the spectral data. The high enthalpy flow penetrates the tile gaps, which consequently expand into cavities, thus exposing the RTV to high heat fluxes. This decomposes the first few mm of the RTV and introduces gaseous RTV constituents (Si, O, C, H, Fe) into the shock layer. The higher concentration of minor metals (Ca) in the RTV compared to the PICA likely contributes to the initial rise in Ca radiance seen in Fig. 2(b), as the erosion in the tile gap would introduce these minor metals into the shock layer. A coarse mesh computational fluid dynamics (CFD) simulation of this cavity flow is shown in Fig. 15(b). The simulation indicates that the cavities initially entrain flow into themselves, resulting in higher heating at the leading edge of the gaps. Subsequently, the mid-row cavities establish a vortex towards the center of the cavity that ejects flow from the cavity back into the shock layer. These vortices help reradiate heat from the vehicle back into the shock layer. Thus, the RTV at the leading edge of the cavity is expected to decompose and erode into the shock layer. Since the collimating lens of OSPREE was directly upstream and in line from a midsection tile gap, we conclude that the recorded Fe emissions and the persistent presence of Na and Ca in the data are a direct result of the flow-induced decomposition of the RTV in this midsection tile gap. Proper understanding of how aerothermodynamic phenomena and gas–surface interactions drive the radiance of metallic TPS impurities in the shock layer is key to assessing TPS performance and optimizing reentry spacecraft design. An improved understanding of the chemical makeup of common TPS materials can aid in identifying the sources of metallic contaminants during manufacturing to reduce or eliminate them from the material entirely. Subsequently, using TPS materials with less of these contaminants in spacecraft design would yield overall lower radiative heating, reducing the total heat load experienced by the vehicle. This would enable a reduced total mass of TPS needed to ensure vehicle survivability, boosting both available payload mass and aerodynamic performance as a result.
Furthermore, knowing the amounts of trace contaminants in the TPS, especially alkali metals, can enable better fluid dynamics modeling predictions of the flowfield chemistry and analysis of other physical effects caused by the presence of such species. Fig. 16 shows results from the fluid dynamics simulation of a commonly simulated historical test vehicle (NASA RAM C-II)68 where a small mole fraction of Na was added to the simulation. The electron density along a line of sight corresponding to the location of a receiving antenna is shown. The addition of 0.01 mole fraction of Na increases the maximum electron density by a factor of 3.2, largely due to the impact ionization reaction discussed in Section 2. The plasma frequency (ωpe), given by
 | (8) |
7 Conclusions
We report the chemical characterization of PICA and RTV using CF-LIBS and the identification of several metallic contaminants such as Si, Na, Ca, K, Al, and Fe in these TPS materials. Cross-section profiles of each material using CF-LIBS are presented, characterizing depth-dependent concentration of the varying metallic elements. It is determined that PICA contains trace amounts (0.001–1 ppm) of Na, Ca, and Al, in addition to Si on the order of 102 ppm due to the application of a waterproof coating. The RTV silicone is characterized by higher levels of alkali/alkaline earth metals (101 ppm), along with a large amount of Fe (103 ppm) as an additive for high temperature applications. Pyrolysis and ablation of PICA during reentry, as well as decomposition and erosion of the RTV silicone between tile gaps due to the high enthalpy cavity flow, introduce these metallic elements into the shock layer. The subsequent excitation and radiation of these metals explains the strong spectral radiance of Na and Fe observed in shock layer OES measurements of the Varda W-2 reentry capsule. Quantification of these contaminants will enable higher fidelity modeling of the TPS-shock layer reaction chemistry to account for enhanced radiative heating and electron generation, contributing to the development of higher-performing future reentry spacecraft.Conflicts of interest
There are no conflicts to declare.Data availability
Data are available upon request from the authors.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ja00374a.
Acknowledgements
Distribution unlimited; approved for public release. AFRL Public Affairs Release #AFRL-2025-4812. This work was funded by the Air Force Office of Scientific Research (AFOSR), Lab Task RVCOR005, through the High-Speed Aerodynamics Portfolio (Dr Amanda Chou). The W-2 flight test was funded under SPACEWERX STRATFI contract FA-9453-23-C-A035. Development of OSPREE was funded through contract FA-9453-18-D-0042. The authors acknowledge funding from the Universities Space Research Association and the AFRL Scholars Program. The authors would like to thank their colleagues at NASA Ames Research Center, Thermal Protection Materials and Aerothermodynamics branches, for their thoughtful discussions on these results which contributed to the analysis. The views expressed are those of the authors and do not necessarily reflect the official policy or position of the Department of the Air Force, the Department of Defense, or the U.S. government.References
- C. Park, Nonequilibrium Hypersonic Aerothermodynamics, Wiley, 1990 Search PubMed.
- J. J. Bertin and R. M. Cummings, Annu. Rev. Fluid. Mech., 2006, 38, 129–157 CrossRef.
- B. K. Bessire, S. A. Lahankar and T. K. Minton, ACS Appl. Mater. Interfaces, 2015, 7, 1383–1395 Search PubMed.
- B. K. Bessire and T. K. Minton, ACS Appl. Mater. Interfaces, 2017, 9, 21422–21437 CrossRef CAS PubMed.
- M. W. Winter, 19th AIAA International Space Planes and Hypersonic Systems and Technologies Conference, Atlanta, GA, 2014 Search PubMed.
- M. Winter, B. Butler, P. M. Danehy, S. Splinter, Z. Diao, F. Panerai, A. Martin and S. Bailey, 46th AIAA Thermophysics Conference, 2016, p. 3233 Search PubMed.
- A. P. Rao, J. D. Crespo, P. Valentini, J. B. Taylor, V. J. Murray, E. I. Vaughan, R. Alviani and M. Kulakhmetov, J. Spacecr. Rockets, 2025, 62, 1486–1495 Search PubMed.
- M. F. Kulakhmetov, R. Alviani, A. Rao, V. Murray, J. B. Taylor, J. Seik and E. Vaughan, AIAA Aviation Forum and Ascend 2024, 2024, p. 4560 Search PubMed.
- M. Kulakhmetov, M. Gallis and A. Alexeenko, J. Chem. Phys., 2016, 144, 1–15 CrossRef PubMed.
- D. W. Hahn and N. Omenetto, Appl. Spectrosc., 2012, 66, 347–419 CrossRef CAS PubMed.
- S. K. H. Shah, J. Iqbal, P. Ahmad, M. U. Khandaker, S. Haq and M. Naeem, Radiat. Phys. Chem., 2020, 170(33), 108666 CrossRef.
- D. A. Cremers and L. J. Radziemski, Handbook of Laser-Induced Breakdown Spectroscopy, John Wiley & Sons, 2013 Search PubMed.
- R. S. Harmon, J. Remus, N. J. McMillan, C. McManus, L. Collins, J. L. Gottfried Jr, F. C. DeLucia and A. W. Miziolek, Appl. Geochem., 2009, 24, 1125–1141 Search PubMed.
- R. Anderson, T. Gabriel, O. Forni, J. Manrique, P. Gasda, D. Vogt, A. Ollila et al., 54th Lunar and Planetary Science Conference 2023, 2023, p. 2182 Search PubMed.
- K. C. Hartig, I. Ghebregziabher and I. Jovanovic, Sci. Rep., 2017, 7, 43852 Search PubMed.
- M. B. Shattan, M. Gragston, Z. Zhang, I. J. D. Auxier, K. G. McIntosh and C. G. Parigger, Appl. Spectrosc., 2019, 73, 591–600 CrossRef CAS PubMed.
- A. P. Rao, P. R. Jenkins, J. D. Auxier, M. B. Shattan and A. K. Patnaik, Appl. Opt., 2022, 61, D30–D38 Search PubMed.
- P. E. Anderson, J. I. Braun, J. I. Borrero Negrón, K. C. Hartig and A. P. Rao, Appl. Opt., 2025, 64, D122–D132 Search PubMed.
- Z. B. Kitzhaber, D. Orea, J. McFarlane, B. T. Manard and H. B. Andrews, ACS Omega, 2025, 10(33), 37889–37897 Search PubMed.
- R. Noll, C. Fricke-Begemann, S. Connemann, C. Meinhardt and V. Sturm, J. Anal. At. Spectrom., 2018, 33, 945–956 Search PubMed.
- M. Dell'Aglio, R. Gaudiuso, G. S. Senesi, A. De Giacomo, C. Zaccone, T. M. Miano and O. De Pascale, J. Environ. Monit., 2011, 13, 1422–1426 RSC.
- Y. Zhang, T. Zhang and H. Li, Spectrochim. Acta, Part B, 2021, 181, 106218 CrossRef CAS.
- G. Radhakrishnan, P. Adams and L. Bernstein, J. Appl. Phys., 2023, 134(1), 013303 Search PubMed.
- A. Ciucci, M. Corsi, V. Palleschi, S. Rastelli, A. Salvetti and E. Tognoni, Appl. Spectrosc., 1999, 53, 960–964 CrossRef CAS.
- N. Zhang, T. Ou, M. Wang, Z. Lin, C. Lv, Y. Qin, J. Li, H. Yang, N. Zhao and Q. Zhang, Front. Phys., 2022, 10, 887171 CrossRef.
- J. Yang, X. Li, J. Xu and X. Ma, Appl. Spectrosc., 2018, 72, 129–140 Search PubMed.
- R. Pamu, S. A. Davari, D. Darbar, E. C. Self, J. Nanda and D. Mukherjee, ACS Appl. Energy Mater., 2021, 4, 7259–7267 Search PubMed.
- L. C. L. Borduchi, D. M. B. P. Milori, M. C. Meyer and P. R. Villas-Boas, Spectrochim. Acta, Part B, 2022, 198, 106561 CrossRef CAS.
- E. Tognoni, G. Cristoforetti, S. Legnaioli and V. Palleschi, Spectrochim. Acta, Part B, 2010, 65, 1–14 CrossRef.
- J. Hermann, C. Gerhard, M. Burger, V. Craciun and F. Pelascini, Spectrochim. Acta, Part B, 2023, 200, 106595 Search PubMed.
- P. R. Villas-Boas and L. C. L. Borduchi, Spectrochim. Acta, Part B, 2023, 205, 106690 CrossRef CAS.
- S. Pandhija, N. Rai, A. K. Rai and S. N. Thakur, Appl. Phys. B, 2010, 98, 231–241 CrossRef CAS.
- M. Shah, A. Pulhani, G. Gupta and B. Suri, Appl. Opt., 2012, 51, 4612–4621 Search PubMed.
- A. P. Rao, J. D. Crespo, P. Valentini, J. B. Taylor, V. J. Murray, E. I. Vaughan, R. Alviani and M. Kulakhmetov, J. Spacecr. Rockets, 2025, 1–15 Search PubMed.
- K.-P. Schneider and C. Park, Phys. Fluids, 1975, 18, 969–981 CrossRef CAS.
- M. H. Bortner, The Chemical Kinetics of Sodium in Re-entry, General electric technical report, 1964 Search PubMed.
- M. Lennon, K. Bell, H. Gilbody, J. Hughes, A. Kingston, M. Murray and F. Smith, J. Phys. Chem. Ref. Data, 1988, 17, 1285–1363 CrossRef CAS.
- M. Jung, H. Kihara, K.-i. Abe and Y. Takahashi, 47th AIAA Fluid Dynamics Conference, 2017, p. 3308 Search PubMed.
- Y. Ralchenko, Mem. Soc. Astron. Ital., 2005, 8, 96 Search PubMed.
- J. Zhong and D. A. Levin, J. Spacecr. Rockets, 2010, 47, 757–764 CrossRef CAS.
- C. O. Johnston, AIAA Aviation 2022 Forum, 2022, p. 3799 Search PubMed.
- S. W. Lewis, R. G. Morgan, T. J. McIntyre, C. R. Alba and R. B. Greendyke, J. Spacecr. Rockets, 2016, 53, 887–899 CrossRef.
- S. W. Lewis, C. M. James, R. G. Morgan, T. J. McIntyre, C. R. Alba and R. B. Greendyke, J. Thermophys. Heat Transfer, 2017, 31, 193–204 CrossRef CAS.
- H. Tran, C. Johnson, D. Rasky, F. Hui, M.-T. Hsu and Y. Chen, 31st Thermophysics Conference, 1996, p. 1911 Search PubMed.
- M. Gasch, K. Skokova, M. Stackpoole, E. Venkatapathy, D. Ellerby, F. Milos, K. Peterson, D. Prabhu, G. Gonzales, S. Violette et al., Development of Domestic Lyocell Based Phenolic Impregnated Carbon Ablator (PICA-D) for Future NASA Missions, National aeronautics and space administration technical report, 2019 Search PubMed.
- M. M. Stackpoole, M. J. Gasch, E. Venkatapathy and S. Violette, National Space and Missile Materials Symposium (NSMMS), 2019 Search PubMed.
- I. Karnadi, M. Pardede, I. Tanra, R. Hedwig, A. M. Marpaung, Z. S. Lie, E. Jobiliong, D. Kwaria, M. M. Suliyanti, M. Ramli, K. Lahna, T. J. Lie, H. Suyanto, K. H. Kurniawan and K. Kagawa, Sci. Rep., 2020, 10, 13278 CrossRef CAS PubMed.
- J. A. J. Effenberger and J. R. Scott, Sensors, 2010, 10, 4907–4925 Search PubMed.
- Z. Q. Hao, L. Liu, M. Shen, X. Y. Yang, K. H. Li, L. B. Guo, X. Y. Li, Y. F. Lu and X. Y. Zeng, Opt. Express, 2016, 24, 26521–26528 Search PubMed.
- N. Karim, M. P. Polek, A. M. Casella, D. J. Senor, S. S. Harilal and E. J. Kautz, Spectrochim. Acta, Part B, 2025, 223, 107081 Search PubMed.
- M. Dell'Aglio, M. López-Claros, J. Laserna, S. Longo and A. De Giacomo, Spectrochim. Acta, Part B, 2018, 147, 87–92 Search PubMed.
- T. Fujimoto and R. McWhirter, Phys. Rev. A, 1990, 42, 6588 Search PubMed.
- B. Bousquet, V. Gardette, V. M. Ros, R. Gaudiuso, M. Dell'Aglio and A. De Giacomo, Spectrochim. Acta, Part B, 2023, 204, 106686 Search PubMed.
- A. De Giacomo, M. Dell'Aglio, O. De Pascale, S. Longo and M. Capitelli, Spectrochim. Acta, Part B, 2007, 62, 1606–1611 Search PubMed.
- T. Völker and I. B. Gornushkin, J. Anal. At. Spectrom., 2022, 37, 1972–1974 RSC.
- H. Griem, Spectral Line Broadening by Plasmas, Elsevier, 2012 Search PubMed.
- M. Sabsabi and P. Cielo, Appl. Spectrosc., 1995, 49, 499–507 CrossRef CAS.
- E. Tognoni, V. Palleschi, M. Corsi and G. Cristoforetti, Spectrochim. Acta, Part B, 2002, 57, 1115–1130 CrossRef.
- C. Aragon, J. A. Aguilera and F. Penalba, Appl. Spectrosc., 1999, 53, 1259–1267 CrossRef CAS.
- J. Hermann, Laser Induced Breakdown Spectroscopy (LIBS) Concepts, Instrumentation, Data Analysis and Applications, 2023, vol. 1, pp. 89–121 Search PubMed.
- A. De Giacomo, Spectrochim. Acta, Part B, 2003, 58, 71–83 CrossRef.
- G. H. Cavalcanti, D. N. Stratis and C. Parigger, Appl. Opt., 2012, 51, 264–271 Search PubMed.
- Y. Fu, J. Liu, X. Li and Y. Zhang, Appl. Spectrosc., 2014, 68, 201–209 Search PubMed.
- M. Fahad, W. A. Farooq, A. Qayyum, S. A. Buzdar and A. Q. Malik, AIP Adv., 2019, 9, 095127 Search PubMed.
- G. Cavalcanti, D. Teixeira, S. Legnaioli, G. Lorenzetti, L. Pardini and V. Palleschi, Spectrochim. Acta, Part B, 2013, 87, 51–56 Search PubMed.
- F. Poggialini, B. Campanella, B. Cocciaro, G. Lorenzetti, V. Palleschi and S. Legnaioli, J. Anal. At. Spectrom., 2023, 38, 1751–1771 Search PubMed.
- F. Rezaei, G. Cristoforetti, E. Tognoni, S. Legnaioli, V. Palleschi and A. Safi, Spectrochim. Acta, Part B, 2020, 169, 105878 CrossRef CAS.
- W. L. Jones and A. E. Cross, Electrostatic-probe Measurements of Plasma Parameters for Two Reentry Flight Experiments at 25000 Feet Per Second, National Aeronautics and Space Administration, 1972, vol. 6617 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |











