Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Visible-light-induced carbonylative atom transfer radical addition of alkenes: straightforward preparation of branched unsaturated thioesters
Ren-Guan Miaoab,
Zhi-Peng Baoab,
Yuanrui Wangab,
Chang-Sheng Kuai ab and
Xiao-Feng Wu
ab and
Xiao-Feng Wu *ab
*ab
aDalian National Laboratory for Clean Energy, Dalian Institute of Chemical Physics, Chinese Academy of Sciences, 116023 Dalian, Liaoning, China. E-mail: xwu2020@dicp.ac.cn
bLeibniz-Institut für Katalyse e.V, Albert-Einstein-Straße 29a, 18059 Rostock, Germany
First published on 4th February 2026
Abstract
Carbonylation is a straightforward methodology for the preparation of carbonyl-containing compounds, which are one of the core classes of compounds in pharmaceutical chemistry and industrial production. Among the developed carbonylation procedures, methods for branched unsaturated compounds are much less studied compared with their linear derivatives. However, their unique chemical structure and properties make them irreplaceable in numerous fields. Herein, we report a new visible-light-induced photocatalytic carbonylative atom transfer radical addition reaction of alkenes. The reaction proceeds effectively with 100% atom efficiency and produced various thioesters in good yields. Then, the product also led to efficient synthesis of branched unsaturated thioesters by simple base-mediated desulfonylation with recyclable arylsulfinic acid eliminated. Remarkably, besides their own importance, thioesters can also be used as acyl donors to produce important compounds such as ketones, aldehydes, esters, amides, and acyl silicon reagents.
Keywords: Carbonylation; Alkene; Unsaturated thioester; Radical addition; Photochemistry.
1 Introduction
Carbonyl-containing compounds are among the most important chemicals in pharmaceutical chemistry and advanced materials. Their unique chemical structure gives them a wide range of transformation possibilities, making them essential in basic organic synthesis, materials science and pharmaceutical chemistry.1–8 Carbonylation is an ideal methodology for the preparation of carbonyl-containing compounds and is advantageous due to its easily scalable production with carbon monoxide as a readily available C1 source. Catalyzed by a suitable catalyst (Pd, Rh, Co, Ni or Cu), carbon monoxide can react with various substrates, such as alkenes, alcohols, amines and halogenated hydrocarbons, to give the corresponding carbonylated products. Numerous carbonyl-containing organic compounds can be readily generated with high atom economy, including aldehydes, carboxylic acids, amides, ketones, esters, thioesters and so on (Fig. 1a).9–15 Among them, branched unsaturated carboxylic acid derivatives are a key class of carbonyl-containing compounds extensively applied in industry. Their unique chemical structures and properties make them invaluable in many fields, including coatings and adhesives, healthcare, textiles and leather, electronics and optics, and papermaking. They are also important synthetic intermediates in organic chemistry and have been used to construct complex organic molecular skeletons through reactions such as addition, cyclisation and coupling.16–29 They are also used to synthesize drug molecules, natural products and functional materials, such as methacrylic acid compounds including MMA, EMA, BMA, HEMA, MACl and MAPA. Among them, methyl methacrylate (MMA) is a key synthetic raw material for polymethyl methacrylate (PMMA), with an annual global market size as high as 6.4 million tonnes (Fig. 1b),30 and plays an irreplaceable role in many fields due to its active chemical properties and excellent processing performance. In industry, MMA is produced by metal-catalyzed carbonylation of ethylene and then condensation with formamide (known as the Lucite alpha process). In comparison, procedures for branched unsaturated compounds are much less studied compared with their linear derivatives.The efficient synthesis of methacrylic acid compounds has long been an attractive area of research for chemists. Decades of development have resulted in a variety of synthetic pathways, each with significant differences in terms of the cost of raw materials, environmental friendliness and process complexity. The ACH method was the first industrialized MMA synthesis route and has been widely used due to the low cost and availability of its raw materials (acetone and hydrocyanic acid), accounting for 60% of global annual production. However, the high toxicity of the raw material hydrocyanic acid and the environmental pollution caused by its by-products as the main drawbacks make this process outdated technology. In the 1980s, the application of the isobutylene oxidation method gradually expanded due to the increasing availability of C4 fractions in the refining industry and growing environmental protection requirements. It currently accounts for around 35% of global production capacity. Isobutylene or tert-butyl alcohol as its raw materials are relatively non-toxic and environmentally friendly, as well as highly atom-economical. However, this method suffers from complex production steps, high catalyst requirements and high reaction temperature. The synthesis of MMA by ethylene carbonylation is an important production process. It boasts advantages such as inexpensive and readily available raw materials, a short production process and low equipment investment costs, making it a promising technology. Nevertheless, expensive catalysts such as rhodium or palladium are compulsory (Fig. 1c).31–33 Therefore, exploring efficient and environmentally friendly synthesis methods remains in high demand.
Alkenes are one of the most important carbon-based resources in chemical industry, and their carbonylative difunctionalization reactions have been widely explored.34–40 However, the direct preparation of branched unsaturated carboxylic acid derivatives is still relatively rare and challenging. Considering the rationality of the reaction design, we found that sulfonyl radicals can readily eliminate and reconstruct the double bond when a carbonyl group is present in the β-position.41 Given the high oxidation potential of unactivated aliphatic alkenes, we envisaged visible-light-induced photocatalytic carbonylation of thiosulfonates via homolytic addition to alkene as a feasible route for preparing branched unsaturated thioesters. Furthermore, thioesters are often used as acyl donors to obtain important compounds such as ketones, aldehydes, esters, amides, and acylsilyl groups due to their excellent stability in air and poor orbital overlap compared to other ester analogues.42–49 Moreover, the eliminated arylsulfinic acid can be recovered in an easy manner, which increases the overall atomic utilization (Fig. 1d).
2 Results and discussion
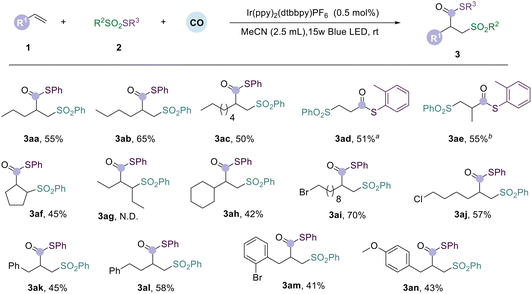
To implement the above synthetic method, we selected S-phenyl benzenesulfonothioate (2a) and hex-1-ene (1a) as the template substrates for systematic studies. After a series of reaction condition explorations, we found that the above design was feasible, and the target product was obtained successfully. After extensive research on the reaction parameters, the optimal conditions were obtained. In acetonitrile solution in the presence of [Ir(dF(CF3)ppy)2(dtbbpy)]PF6 (0.5 mol%), hex-1-ene (1a) (2.0 equiv.) was reacted with S-phenyl benzenesulfonothioate (2a) (1.0 equiv.) at room temperature for 48 hours to afford compound 3ab in 65% yield. Compound 3ab was then desulfonylated with DBU to give compound 4ad in 55% yield (Fig. 2a). Other metal photocatalysts, such as ruthenium and iridium, could also produce the target compound, but with relatively low yields (Fig. 2b). We also screened the CO pressure required; the yield of product 3ab was decreased to 31% when the pressure of CO was reduced from 60 bar CO to 40 bar. The yield of 3ab decreased to 9–20% when the reaction was performed in other solvents. In the absence of a photocatalyst, the reaction yielded only 7% of 3ab, and in the absence of light, the target compound was not obtained, even at evaluated reaction temperature. It is worth mentioning that a non-carbonylation product was the main side product detectable during the optimization process.2.1 Evaluating the reaction scope
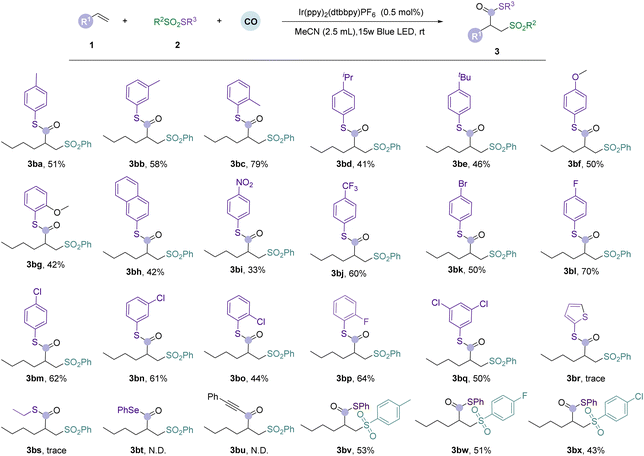
After determining the optimal conditions for the visible-light-induced carbonylation of alkenes, we tested the versatility of various substrates (Table 1). First, we examined the effects of various unactivated alkene substrates. For linear unactivated alkenes with varying lengths, from ethylene to oct-1-ene, we found that the target products (3aa–3ae) were all obtained in moderate to good yields under standard conditions. Notably, when using ethylene or propylene as the substrate, we achieved smooth conversion using a mixture of 10 bar of alkene gas and 50 bar of carbon monoxide, demonstrating the excellent applicability of this reaction with gaseous alkenes. Then, internal alkenes (3af and 3ag) were checked under the standard conditions; the cyclic internal alkene reacted smoothly and gave the desired product in 45% yield, but no target compound was detected with the linear internal alkene. These phenomena might be due to the reason that the cyclic internal alkene is strained and activated while the linear internal alkene is not. We then examined substrates containing various substituents on the linear alkene chain. Cyclohexyl-substituted alkenes yielded only 42% (3ah), possibly due to its steric hindrance. Halogen-substituted alkenes also gave moderate to good yields of the desired products successfully (3ai and 3aj). To further demonstrate the influence of substituents, we further investigated allylbenzene and its aryl variations, as well as but-3-en-1-ylbenzene (3ak–3an). The desired products can be obtained in moderate yields without any problem, even with ortho-bromo substituted and para-methoxy substituted substrates. However, no desired product was detected when styrene or butyl acrylate was tested.Next, we examined the substrate versatility of thiosulfonates under optimal conditions (Table 2). We first investigated thiosulfonates containing electron-rich groups (including methyl, isopropylene, tert-butyl, and methoxy), which afforded the desired products in moderate to good yields (3ba–3bg). The naphthalene substituted substrate can also give the corresponding product in 42% yield (3bh). Notably, thiosulfonates substituted with an ortho-methyl group resulted in the highest yield compared with those substituted with meta- and para-methyl groups (3bc), likely due to steric hindrance. However, when ortho-methoxy substituted thiosulfonate was tested under the same conditions, the reaction outcome did not meet the expectation (3bg). We then investigated the compatibility of thiosulfonates substituted with electron-withdrawing groups (nitro, trifluoromethyl, chloride, bromide, and fluoride), which were successfully transformed to the desired products in moderate to good yields (3bi–3bq). We found that with fluoride- and chloride-substituted thiosulfonates, the yields of ortho- and para-substituted thiosulfonates were lower. When testing thiophene- and alkyl-substituted substrates (3br and 3bs), only a small amount of the target compound was detected. When using Se-phenyl benzenesulfonoselenoate and ((phenylethynyl)sulfonyl)benzene as the substrates, no expected products (3bt–3bu) were detected. We then tested sulfonyl-substituted substrates, including para-substituted methyl, fluorine, and chlorine (3bv–3bx), which afforded the corresponding products in moderate yields in all cases.
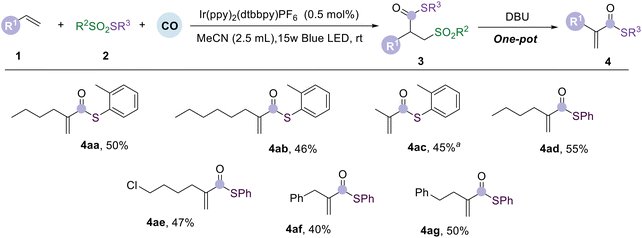
Based on the above discussed substrate studies, we attempted a one-pot, two-step approach to obtain branched unsaturated thioesters (Table 3). We examined alkene substrates of varying chain lengths and found that they could be obtained in moderate yields (4aa–4ad). Subsequently, we explored chlorine- and phenyl-substituted alkenes, and also afforded the desired unsaturated thioesters in moderate yields (4ae–4ag). This demonstrates that this reaction can successfully provide branched unsaturated thioesters conveniently in a one-pot, two-step approach.
| General conditions: step 1: 1 (0.2 mmol), 2 (0.1 mmol), Ir(ppy)2(dtbbpy)PF6 (0.5 mol%), in MeCN (2.5 mL) at rt for 48 h under CO (60 bar), irradiated by 15 W blue LEDs. Step 2: DBU (0.6 equiv.), 10 min, rt. All yields are isolated yields.a Propylene (10 bar), CO (50 bar). |
|---|
 |
Under optimal conditions, we also attempted to scale up the substrate reaction, and we still obtained the target product in 51% yield with a 1 mmol scale (Fig. 3a). Furthermore, we attempted to polymerize S-phenyl 2-methylprop-2-enethioate; a colorless solid was obtained successfully with promising molecule weight (Fig. 3b). The obtained unsaturated thioester can also be converted to the corresponding unsaturated ester easily (Fig. 3c). Furthermore, 44% yield of the desired product can be produced even in the absence of a photosensitizer by replacing light with 365 nm LEDs (Fig. 3d).
2.2 Mechanism studies
To further explore the mechanistic pathway of this reaction, we conducted a series of control experiments, as shown in Fig. 4. We attempted to verify the involvement of radical intermediates by adding radical inhibitors, such as TEMPO and BHT. First, no target product could be detected after 3 equivalents of TEMPO were added under standard conditions (Fig. 4a). Additionally, only a small amount of the target product was produced when 3 equivalents of BHT were added (Fig. 4b). These results suggest that the reaction may undergo a radical mechanism. Furthermore, we attempted a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 reaction of S-phenyl benzenesulfonothioate and S-(4-chlorophenyl) 4-chlorobenzenesulfonothioate under standard conditions. We detected the formation of both S-(4-chlorophenyl)benzenesulfonothioate and S-phenyl 4-chlorobenzenesulfonothioate, further confirming the possibility of radical involvement (Fig. 4c). However, in the absence of light, no reaction occurred, leading us to speculate that the thiosulfonate reacts via homolytic cleavage. When we added 1,2-bis(4-chlorophenyl)disulfane to the template reaction, two mixed products were detected (Fig. 4d). The reaction with only 1,2-bis(4-chlorophenyl)disulfane, in the absence of thiosulfonate, will lead to β-thiopropionate thioester formation.50 To verify whether sulfinic acid was produced after desulfonylation, we tried adding NaOH and water for extraction after the desulfonylation step. We detected sodium benzenesulfinate salt, and the resulting mixture could be used directly as a synthetic raw material for substate preparation (Fig. 4e).
1 reaction of S-phenyl benzenesulfonothioate and S-(4-chlorophenyl) 4-chlorobenzenesulfonothioate under standard conditions. We detected the formation of both S-(4-chlorophenyl)benzenesulfonothioate and S-phenyl 4-chlorobenzenesulfonothioate, further confirming the possibility of radical involvement (Fig. 4c). However, in the absence of light, no reaction occurred, leading us to speculate that the thiosulfonate reacts via homolytic cleavage. When we added 1,2-bis(4-chlorophenyl)disulfane to the template reaction, two mixed products were detected (Fig. 4d). The reaction with only 1,2-bis(4-chlorophenyl)disulfane, in the absence of thiosulfonate, will lead to β-thiopropionate thioester formation.50 To verify whether sulfinic acid was produced after desulfonylation, we tried adding NaOH and water for extraction after the desulfonylation step. We detected sodium benzenesulfinate salt, and the resulting mixture could be used directly as a synthetic raw material for substate preparation (Fig. 4e).
2.3 Possible mechanism
Based on the above experiments and results, we proposed a possible reaction mechanism (Fig. 5). First, compound B is excited by the excited photosensitizer and homolytically splits into a sulfonyl radical and a phenylthiol radical. The sulfonyl radical is then added to the unactivated alkene A to form intermediate C. Under carbon monoxide pressure, intermediate C captures carbon monoxide to form acyl radical intermediate D. In the presence of a phenylthiol radical or disulfide, product E is obtained finally. Product E can then be desulfonylated by DBU to produce product F.3 Conclusions
In summary, we have developed a novel visible-light-induced carbonylation method for the synthesis of branched unsaturated thioesters. This reaction involves homolytic addition of thiosulfonates to unactivated alkenes, followed by carbonylative difunctionalization under the action of carbon monoxide to afford thioesters. This reaction, while milder than previous methods for synthesizing α,β-unsaturated carboxylic acid derivatives, afforded a variety of valuable branched unsaturated thioesters in moderate to good yields. The reaction also exhibited good tolerance for a wide range of functional groups. Furthermore, the difunctionalized carbonylation of thiosulfonates via homolytic cleavage demonstrated high atom utilization up to 100%.4 Methods
1 (0.2 mmol), 2 (0.1 mmol), Ir(ppy)2(dtbbpy)PF6 (0.5 mol%), and an oven-dried stir bar were placed in a 4 mL screw-capped vial. The vial was sealed with a Teflon septum and cap and vented to the atmosphere via a needle. MeCN (2.5 mL) was then added via a syringe under a nitrogen atmosphere. The sealed autoclave was flushed twice with nitrogen (10 bar) and filled with 60 bar of CO. The reaction mixture was stirred at room temperature under 15 W blue LEDs (450–460 nm) for 48 hours. After the reaction was complete, the pressure was carefully released. DBU (0.6 equiv.) was added to the reaction solution and stirred at room temperature under air for 10 minutes. The mixture was concentrated in vacuo. The crude product was purified by silica gel column chromatography (PE/EA = 50/1) to obtain the corresponding product. It is important to note that unsaturated thioesters readily decompose in DBU, so the product should be handled promptly after the reaction.Conflicts of interest
The authors declare no conflict of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI).Supplementary information is available. See DOI: https://doi.org/10.1039/d5im00399g.
Acknowledgements
We are thankful for the financial support provided by the National Key R&D Program of China (2023YFA1507500), the National Natural Science Foundation of China (22571291, 22302198, 22572190), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB1530000), and the Chinese Academy of Sciences Dalian Institute of Chemical Physics (DICP).References
- B. Gabriele, Carbon monoxide in organic synthesis – carbonylation chemistry, Wiley, 2022 Search PubMed.
- B. Liu, F. Hu and B.-F. Shi, Recent advances on ester synthesis via transition-metal catalyzed C−H functionalization, ACS Catal., 2015, 5, 1863–1881 CrossRef CAS.
- A. Das and P. Theato, Activated ester containing polymers: Opportunities and challenges for the design of functional macromolecules, Chem. Rev., 2016, 116, 1434–1495 CrossRef CAS PubMed.
- P. A. Dub, R. J. Batrice, J. C. Gordon, B. L. Scott, Y. Minko, J. G. Schmidt and R. F. Williams, Engineering catalysts for selective ester hydrogenation, Org. Process Res. Dev., 2020, 24, 415–442 CrossRef CAS.
- J. S. Carey and E. McCann, Lipase-catalyzed regioselective ester hydrolysis as a key step in an alternative synthesis of a buprenorphine pro-drug, Org. Process Res. Dev., 2019, 23, 771–774 CrossRef CAS.
- A. Greenberg, C. M. Breneman and J. F. Liebman, The amide linkage: Structural significance in chemistry, biochemistry, and materials science, John, Wiley & Sons, 2000 Search PubMed.
- J. W. Clader, The discovery of ezetimibe: A view from outside the receptor, J. Med. Chem., 2004, 47, 1–9 CrossRef CAS PubMed.
- L. Crespo, G. Sanclimens, M. Pons, E. Giralt, M. Royo and F. Albericio, Peptide and amide bond-containing dendrimers, Chem. Rev., 2005, 105, 1663–1682 CrossRef CAS PubMed.
- W. Gao, S. Liu, Z. Wang, J. Peng, Y. Zhang, X. Yuan, X. Zhang, Y. Li and Y. Pan, Outlook of cobalt-based catalysts for heterogeneous hydroformylation of olefins: From nanostructures to single atoms, Energy Fuels, 2024, 38, 2526–2547 CrossRef CAS.
- M. Chen, Y. Li and Z.-H. Guan, Palladium-catalyzed hydroalkoxycarbonylation and hydroxycarbonylation of cyclopent-3-en-1-ols: Divergent synthesis of bridged cyclic lactones and β,γ-unsaturated carboxylic acids, Org. Lett., 2023, 25, 2571–2576 CrossRef CAS PubMed.
- Y. Li, K. Dong, F. Zhu, Z. Wang and X.-F. Wu, Copper-catalyzed carbonylative coupling of cycloalkanes and amides, Angew. Chem., Int. Ed., 2016, 55, 7227–7230 Search PubMed.
- T. Morimoto, J. Jia, Y. Yamaguchi, H. Tanimoto and K. Kakiuchi, Cationic rhodium(I)-catalyzed carbonylative [2+2+1] cycloaddition of diynes, Asian J. Org. Chem., 2020, 9, 1778–1782 CrossRef CAS.
- M. Luo, Z. Liu, H. Chen, H. Fu, R. Li and X. Zheng, Bifunctional diphosphine ligand-enabled cobalt catalyzedbis-alkoxycarbonylation of alkynes, J. Catal., 2024, 433, 115459 CrossRef CAS.
- H.-J. Ai, W. Lu and X.-F. Wu, Palladium catalysis ligand-controlled regiodivergent thiocarbonylation of alkynestoward linear and branched α,β-unsaturated thioesters, Angew. Chem., 2021, 60, 17178–17184 CrossRef CAS PubMed.
- J.-X. Xu, C.-S. Kuai and X.-F. Wu, Cobalt-catalyzed four-component carbonylation of methylareneswith ethylene and alcohols, J. Org. Chem., 2022, 87, 6371–6377 CrossRef CAS PubMed.
- S. Zhang, H. Neumann and M. Beller, Synthesis of α,β-unsaturated carbonyl compounds by carbonylation reactions, Chem. Soc. Rev., 2020, 49, 3187–3210 RSC.
- Z. Shi, C. Grohmann and F. Glorius, Mild Rhodium(III)-catalyzed cyclization of amides with α,β-unsaturated aldehydes and ketones to azepinones: Application to the synthesis of the homoprotoberberine framework, Angew. Chem., Int. Ed., 2013, 52, 5393–5397 CrossRef CAS PubMed.
- S. Gnaim, J. C. Vantourout, F. Serpier, P.-G. Echeverria and P. S. Baran, Carbonyl desaturation: Where does catalysis stand?, ACS Catal., 2021, 11, 883–892 CrossRef CAS.
- M. Chen and G. Dong, Copper-catalyzed desaturation of lactones, lactams, and ketones under pH-neutral conditions, J. Am. Chem. Soc., 2019, 141, 14889–14897 CrossRef CAS PubMed.
- S. Gnaim, Y. Takahira, H. R. Wilke, Z. Yao, J. Li, D. Delbrayelle, P.-G. Echeverria, J. C. Vantourout and P. S. Baran, Electrochemically driven desaturation of carbonyl compounds, Nat. Chem., 2021, 13, 367–372 CrossRef CAS PubMed.
- B. Hu, T. X. Liu, P. Zhang, Q. Liu, J. Bi, L. Shi, Z. Zhang and G. Zhang, N-heterocyclic carbene-catalyzed α,β-unsaturated aldehydes umpolung in fullerene chemistry: Construction of [60]fullerene-fused cyclopentan-1-ones and cyclohex-2-en-1-ones, Org. Lett., 2018, 20, 4801–4805 CrossRef CAS PubMed.
- M. Schroeder, M. Mathys, N. Ehrensperger and M. Büchel, γ-Unsaturated aldehydes as potential lilial replacers, Chem. Biodiversity, 2014, 11, 1651–1673 CrossRef CAS PubMed.
- A. J. von Wangelin, H. Neumann, D. Gördes, S. Klaus, D. Strübing and M. Beller, Multicomponent coupling reactions for organic synthesis: chemoselective reactions with amide–aldehyde mixtures, Chem. – Eur. J., 2003, 9, 4286–4294 CrossRef CAS PubMed.
- S. Hübner, H. Jiao, D. Michalik, H. Neumann, S. Klaus, D. Strübing, A. Spannenberg and M. Beller, In situ generation of chiral N-dienyl lactams in a multicomponent reaction: An efficient and highly selective way to asymmetric amidocyclohexenes, Chem. – Asian J., 2007, 2, 720–733 CrossRef PubMed.
- L. V. Reddy, V. Kumar, R. Sagar and A. K. Shaw, Glycal-derived δ-hydroxy α,β-unsaturated aldehydes (perlin aldehydes): Versatile building blocks in organic synthesis, Chem. Rev., 2013, 113, 3605–3631 CrossRef CAS PubMed.
- L. Hu, X. Luand and L. Deng, Catalytic enantioselective peroxidation of α,β-unsaturated aldehydes for the asymmetric synthesis of biologically important chiral endoperoxides, J. Am. Chem. Soc., 2015, 137, 8400–8403 Search PubMed.
- M. Breuer, K. Ditrich, T. Habicher, B. Hauer, M. Kesseler, R. Stürmer and T. Zelinski, Industrial methods for the production of optically active intermediates, Angew. Chem., Int. Ed., 2004, 43, 788–824 Search PubMed.
- S. Wu, N. Yang, Y. Liu, J. Cao, H. Hu, Y. Sun and J. Liu, Optically active helical polymer from radical polymerization of menthyl vinyl ketone, J. Polym. Sci., Part A: Polym. Chem., 2011, 49, 293–299 Search PubMed.
- R. Nazir, T. T. Meiling, P. J. Cywinski and D. T. Gryko, Synthesis and optical properties of α,β-unsaturated ketones bearing a benzofuran moiety, Asian J. Org. Chem., 2015, 4, 929–935 Search PubMed.
- W. Bauer, Methacrylic acid and derivatives, in Ullmann's encyclopedia of industrial chemistry, Wiley, 2011 Search PubMed.
- J. Liu, Q. Chen, Y. Liu, M. Gao and X. Zhao, Research progress in synthesis technologies of methyl methacrylate, Zhongguo Suliao, 2022, 36, 178–183 Search PubMed.
- K.-Y. Chen, Z. Yang, F.-Z. Chen, J.-G. Li, Y.-L. Chen and N.-W. Zhou, Analysis of methyl methacrylate supply and demand status and development prospect, Guangzhou Huagong, 2022, 50, 36–38 Search PubMed.
- J. Liu, K. Dong, R. Franke, H. Neumann, R. Jackstella and M. Beller, Development of efficient palladium catalysts for alkoxycarbonylation of alkenes, Chem. Commun., 2018, 54, 12238–12241 Search PubMed.
- L. Zeng, H. Li, J. Hu, D. Zhang, J. Hu, P. Peng, S. Wang, R. J. Peng, C.-W. Pao, J.-L. Chen, J.-F. Lee, H. Zhang, Y.-H. Chen and A. Lei, Electrochemical oxidative aminocarbonylation of terminal alkynes, Nat. Catal., 2020, 3, 438–445 CrossRef CAS.
- M. Yang, Y. Liu, X. Qi, Y. Zhao and X.-F. Wu, Carbonylative transformation of aryl halides and strong bonds via cheap metal catalysts and sustainable technologies, Green Synth. Catal., 2024, 5, 211–269 CAS.
- Y. Zhang, B.-H. Teng and X.-F. Wu, Copper-catalyzed trichloromethylative carbonylation of ethylene, Chem. Sci., 2024, 15, 1418–1423 RSC.
- R.-G. Miao, Y. Wang, Z.-P. Bao and X.-F. Wu, Trifluoromethylthiolation carbonylation of unactivated alkenes via distal migration, Org. Lett., 2024, 26, 10189–10194 CrossRef CAS PubMed.
- R.-G. Miao, Y. Wang and X.-F. Wu, Visible-light-promoted phosphorylation carbonylation of unactivated alkenes, J. Catal., 2025, 442, 115933 CrossRef CAS.
- Q. Li, L.-C. Wang and X.-F. Wu, Photo-promoted carbonylative difunctionalization of alkenes toward β-aminoketones, J. Catal., 2025, 443, 115935 CrossRef CAS.
- Z.-P. Bao, Y. Zhang, L.-C. Wang and X.-F. Wu, Difluoroalkylative carbonylation of alkenes to access carbonyl difluoro-containing heterocycles: Convenient synthesis of gemigliptin, Sci. China: Chem., 2023, 66, 139–146 CrossRef CAS.
- C. Nájera, B. Mancheño and M. Yus, (E)-N-isopropyl-3-tosylacrylamide: A new α-acylvinyl cation: Equivalent in the synthesis of α-substituted acrylamides, Tetrahedron Lett., 1989, 30, 6085–6088 CrossRef.
- I. A. Os'kina and V. M. Vlasov, Activation parameters of the reactions of 4-nitrophenyl benzoates and S-phenyl benzothioate with 4-chlorophenol in dimethylformamide in the presence of potassium carbonate, Russ. J. Org. Chem., 2009, 45, 523–527 CrossRef.
- T. Miyazaki, Y. Han-ya, H. Tokuyama and T. Fukuyama, New odorless protocols for the synthesis of aldehydes and ketones from thiol esters, Synlett, 2004, 3, 477–480 Search PubMed.
- H. Tokuyama, S. Yokoshima, T. Yamashita and T. Fukuyama, A novel ketone synthesis by a palladium-catalyzed reaction of thiol esters and organozinc reagents, Tetrahedron Lett., 1998, 39, 3189–3192 CrossRef CAS.
- L. S. Liebeskind and J. Srogl, Thiol ester-boronic acid coupling. A mechanistically unprecedented and general ketone synthesis, J. Am. Chem. Soc., 2000, 122, 11260–11261 CrossRef CAS.
- J. M. Villalobos, J. Srogl and L. S. Liebeskind, A new paradigm for carbon-carbon bond formation: Aerobic, copper-templated cross-coupling, J. Am. Chem. Soc., 2007, 129, 15734–15735 CrossRef CAS PubMed.
- F. Sun, M. Li, C. He, B. Wang, B. Li, X. Sui and Z. Gu, Cleavage of the C(O)-S bond of thioesters by palladium/norbornene/copper cooperative catalysis: An efficient synthesis of 2-(arylthio)aryl ketones, J. Am. Chem. Soc., 2016, 138, 7456–7459 Search PubMed.
- M. Ueda, K. Seki and Y. Imai, S- and N-acyl derivatives of 2-mercaptobenzoxazole; New, highly reactive acylating agents for synthesis of amides and esters, Synthesis, 1981, 12, 991–993 CrossRef.
- M. N. Burhardt, R. H. Taaning and T. Skrydstrup, Pd-catalyzed thiocarbonylation with stoichiometric carbon monoxide: Scope and applications, Org. Lett., 2013, 15, 948–951 CrossRef CAS PubMed.
- J.-X. Xu, Z.-P. Bao and X.-F. Wu, Palladium-catalyzed carbonylation of disulfides and ethylene: Synthesis of β-thiopropionate thioesters, Org. Lett., 2022, 24, 1848–1852 CrossRef CAS PubMed.
| This journal is © Institute of Process Engineering of CAS 2026 |