Open Access Article
Open Access ArticleKey factors influencing direct CO2 capture from air
Ao-Chuan Zheng
a,
Hao-Sheng Xua,
Lin Du a,
Yan Li
*a and
Xin-Ming Hu
a,
Yan Li
*a and
Xin-Ming Hu
 *abc
*abc
aEnvironment Research Institute, Shandong University, Qingdao 266237, China. E-mail: liyan2024@sdu.edu.cn; huxm@sdu.edu.cn
bShandong Key Laboratory of Environmental Processes and Health, Shandong University, Qingdao 266237, China
cState Key Laboratory of Microbial Technology, Shandong University, Qingdao 266237, China
First published on 11th March 2026
Abstract
Excessive CO2 emission into the atmosphere has caused serious climate change that draws global concerns. Direct air capture (DAC) plays a crucial role in reducing atmospheric CO2 concentration and mitigating climate change. In this review, we summarize the latest advances and emerging opportunities in DAC through three key aspects. First, we introduce two different types of air contactors (adsorption- and absorption-based) and their functions, unveiling their critical roles in DAC. Second, we discuss a number of effective capture agents for DAC, including solid adsorbent materials and liquid absorbent solutions, with emphasis on the capture mechanism and efficiency. Third, we present three typical methods for CO2 release and capture agent regeneration that match DAC effectively, including temperature swing, precipitation-phase separation, and electrolysis, focusing on the CO2 release process and energy consumption. In the end, we provide insights into the existing challenges, potential solutions, and future directions for advancing DAC technologies.
Keywords: CO2 capture; Direct air capture; Air contactor; Capture agent; Regeneration method.
1 Introduction
The CO2 concentration in the atmosphere has risen to 420 ppm, bringing a significant crisis to the Earth's climate system, such as global warming.1 In response to this challenge, the historic Paris Agreement was adopted at the 2015 Conference of the Parties to the United Nations Framework Convention on Climate Change, which emphasizes the significance of reducing greenhouse gas emissions and calls for global cooperation to limit temperature rise to <2 °C, with efforts to strive for a limit of 1.5 °C.2 The agreement has spurred increasing efforts to reduce CO2 emissions in many countries and regions by setting carbon neutrality targets, which is defined as equilibrating CO2 emissions with removal and represents a state of net-zero emissions essential for climate stabilization.3–7Carbon capture, utilization, and storage (CCUS) is a crucial technology for achieving carbon neutrality by reducing CO2 emissions and enhancing carbon sinks across various industrial sections, offering both environmental benefits and economic opportunities.8,9 CO2 capture is the first step in CCUS, laying the foundation for subsequent processes of CO2 storage and utilization. At present, research on CO2 capture is mainly focused on point sources (e.g., power, petrochemical, and steel plants) due to their enormous and manageable CO2 emissions. However, about 1000 gigatons (Gt) of CO2 must be removed from the atmosphere by 2100 to meet the target set by the Paris Agreement, which sounds like an impossible task if only relying on conventional CCUS technologies.10 In addition to point sources, emissions from decentralized sources such as transportation, residence, and agriculture account for 36% of total annual CO2 emissions.11 This portion of CO2 is almost entirely emitted into the atmosphere, constituting another important emission source. Uncontrolled CO2 emissions could lead to a global temperature increase exceeding 1.5 °C within 10 years.12 Gür proposed a vivid picture for CO2 removal: offsetting just 1 Gt atmospheric CO2 would require reforesting an area of 900![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 km2 (nearly the total land area of Germany and France).13 Therefore, it is imperative to explore new technologies for capturing CO2 from air.
000 km2 (nearly the total land area of Germany and France).13 Therefore, it is imperative to explore new technologies for capturing CO2 from air.
Direct air capture (DAC) emerges as a new technology aimed at reducing atmospheric CO2 concentrations. Unlike carbon capture from flue gas which targets specific emission sources, DAC removes CO2 directly from ambient air. This capacity makes it particularly suitable for sectors where controlling CO2 emissions is challenging, such as transportation, residence, agriculture, and construction. Moreover, DAC technology can be integrated with other emerging technologies to create a more comprehensive strategy for addressing climate change.14 For example, powering DAC systems with renewable energy such as solar and wind power can significantly reduce their carbon footprint during operation, and converting the captured CO2 into valuable fuels and chemicals enables carbon recycling.15–19 Thus, the implementation of DAC not only offers a technological means for mitigating climate change but also holds the potential to promote the development of emerging industries and green economy.
Despite of the great prospect, DAC is currently in its early stages of development. Fig. 1a summarizes the reported projects for DAC and point source CO2 capture all over the world, according to the data from the International Energy Agency, April 2025. It shows that the number of DAC projects is very limited, and only a few of them (5) are in operation. Most DAC initiatives are still under construction (5) or in the planning (45) stages. In contrast, there are already 53 operational point-source CO2 capture projects, 36 under construction, and 337 planned, which all outnumbers DAC dramatically. The total capacity of operational CO2 capture plants suggests that the 53 point source plants can capture approximately 50.9 million tons CO2 per year, while the DAC plants only capture 0.017 million tons CO2 annually (Fig. 1b).
 | ||
| Fig. 1 (a) Reported plants for CO2 capture which are either in operation, under construction, or planned; (b) total annual CO2 capture capacity of operational carbon capture plants. Data from International Energy Agency.20 | ||
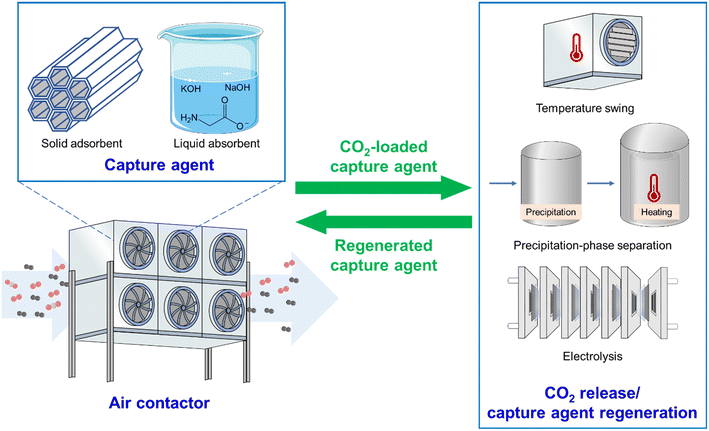
The slow development of DAC is primarily due to the large energy consumption and low efficiency of extracting low concentration of CO2 from air. Consequently, developing efficient DAC systems is crucial to advance this emerging technology and making it viable for practical applications. A complete DAC system can be divided into three major components: air contactor, capture agent, and CO2 release/capture agent regeneration unit. Each component critically influences the overall efficiency of the DAC process. Previous research has generated substantial knowledge and progress into these key aspects, paving the way for practical applications of the DAC technologies.21,22 Recently, a handful of reviews have summarized the important progress of DAC technologies, primarily focusing on either the development of efficient sorbent materials,23,24 the system's water management,25 or mobile application scenarios.26 However, a review that explicitly links the design and optimization of air contactors, capture agents, and regeneration methods remains absent in the literature. Addressing this gap through a systematic examination of the entire DAC process would significantly benefit the DAC community.
This review is structured around the three key factors influencing the DAC efficiency, including air contactors, capture agents, and CO2 release/capture agent regeneration approaches (Fig. 2). We start by introducing the types and functions of the core setup for DAC, i.e., the air contactors. Following this, we discuss two types of capture agents for DAC, i.e., solid adsorbent materials and liquid absorbent solutions, focusing on their mechanism and efficiency for CO2 capture. Finally, we present three primary approaches for CO2 release and capture agent regeneration that match DAC. Additionally, we outline the challenges faced by current DAC technologies and propose potential solutions in conjunction with our perspectives on the future directions of DAC.
2 Air contactors
The air contactor is the core unit of the DAC system, responsible for maintaining substantial airflow and facilitating the interaction between ad/absorbents and CO2 molecules. It typically comprises axial fans and packing layers. The fan system draws in air, while the packing layer, made from lightweight and corrosion-resistant materials such as polyvinyl chloride (PVC), is designed with numerous pores and channels to maximize the surface area.Air contactors can be categorized into two types based on CO2 capture modes: adsorption and absorption. In general, adsorption-based air contactors employ solid adsorbents for CO2 capture, whereas absorption-based air contactors utilize liquid solutions. However, it is important to note that some CO2 capture systems may involve hybrid mechanisms. For example, Liu et al. reported a slurry-based sorbent consisting of the metal–organic framework (MOF), ion liquid, amine, and membrane, which achieved efficient CO2 capture through a hybrid adsorption–absorption mechanism that leverages the advantages of both solid and liquid sorbents.27 To provide conceptual clarity, we classify DAC systems as either adsorption- or absorption-based types according to the sorbent phase and the dominant CO2 capture mechanism.
2.1 Adsorption-based air contactors
An adsorption-based air contactor is usually equipped with a solid adsorbent on the packing layer and connected with an in situ system for CO2 release and adsorbent regeneration (Fig. 3a). The operational process is straightforward: (i) air is drawn into the contactor via the fan system and brought into contact with the solid adsorbent, where CO2 is adsorbed; (ii) after the capture process, the contactor is sealed, and a temperature/pressure swing program is initiated to release CO2 and regenerate the adsorbent. Most reported DAC systems rely on adsorption-based air contactors, including those from Climeworks, Heirloom, and CarbonCapture Inc.28–30 These adsorption-based air contactors integrate all functions for CO2 capture, release, and adsorbent regeneration, showing significant advantages such as small space occupation and flexible modularity for placement in any location. Notably, the size of the air contactors determines the scale of CO2 capture. In 2021, Climeworks launched its Orca facility in Iceland, which was the world's largest adsorption-based DAC plant at the time, achieving an annual CO2 capture capacity of 4000 tons.31 Building on this milestone, the company launched its Mammoth facility in 2024, with a capacity of 36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons CO2 per year, nearly ten times that of Orca. This scale-up marks a significant step toward megaton-scale DAC deployment.32
000 tons CO2 per year, nearly ten times that of Orca. This scale-up marks a significant step toward megaton-scale DAC deployment.32
 | ||
| Fig. 3 Different configurations of adsorption-based air contactors: (a) the common fixed-bed configuration with in situ temperature/pressure swing for CO2 release; (b) the rotating-bed configuration with steam-assisted CO2 release. Reprinted with permission from ref. 35. Copyright 2026 Wiley; different configurations of packing layers: (c) helical packing structure. Reprinted with permission from ref. 36. Copyright 2023 Elsevier; (d) wash-coated honeycomb monolithic configuration. Reprinted with permission from ref. 37. Copyright 2023 American Chemical Society. | ||
The common fixed-bed reactors can serve as air contactors for DAC. However, they impose stringent requirements on the size of adsorbent particles, which limits adsorbent selection and makes them susceptible to attrition.33 Moreover, multiple fixed beds must operate in parallel to maintain continuous CO2 capture, significantly increasing system complexity.34 To address these challenges, Wu et al. recently developed a rotating-bed contactor (Fig. 3b).35 In this design, powdered capture agents are fabricated into structured sorption modules and loaded into a rotatable reaction bed, which is divided into two functional zones for adsorption and desorption. In the adsorption zone, air flows through the module, where CO2 is captured by the adsorbent. In the desorption zone, CO2 is desorbed with hot steam. A motor drives the bed to rotate slowly and continuously, enabling each sorption module to cycle periodically between adsorption and desorption. This configuration allows for continuous CO2 capture from air.
The packings inside the adsorption-based air contactor can be of various cross-sectional shapes, with the adsorbent material loaded on the walls of the channels. As air flows through the contactor, CO2 diffuses through the channel walls, where it is captured by the adsorbent. Enhancing the mass transport of CO2 to the adsorbent is crucial for improving CO2 capture efficiency and reducing operational cost. Therefore, optimizing the structure of the packing layer is very important. Tegeler et al. presented a high-surface-area packing with helical channels instead of straight ones, significantly enhancing the transport of CO2 to the adsorbent (Fig. 3c).36 The air contactor with such a packing configuration reduced the overall DAC cost by about 30%. The research by Grossmann et al. indicated that the structure of the adsorbent loaded onto the packing layer also affected CO2 capture efficiency (Fig. 3d).37 They evaluated two adsorbents: one with a pellet structure and the other with a wash-coated honeycomb monolith structure, and found that the latter enhanced mass transport kinetics and reduced pressure drop, making it a more promising option for DAC contactors.
It is important to note that temperature and vacuum are key driving forces for the regeneration of adsorbents in adsorption-based air contactors, necessitating the integration of control valves, vacuum pumps, and heat sources.38 This makes the internal piping and control processes of such air contactors relatively intricate, as a single contactor incorporates too many subsystems. Moreover, given the need to release high-purity CO2, the sealing integrity of the air contactor deserves special attention.
2.2 Absorption-based air contactors
An absorption-based air contactor shares a similar structure with the adsorption-based counterpart but has completely different CO2 capture and release processes. In this type of air contactor, the liquid absorbent flows uniformly through the packing layer, ensuring sufficient contact with CO2 in the air stream to form a CO2-loaded solution, which is then transported to a separate unit for CO2 desorption (Fig. 4a). In this system, CO2 release is decoupled from the capture process, enabling on-demand adjustment of the release strategy to accommodate actual needs and varying operating conditions. To our knowledge, Carbon Engineering is the only company in the world using absorption-based air contactors, which employ a calcium loop system involving precipitation, phase separation, and calcination for CO2 release and KOH absorbent solution regeneration (Fig. 4a).39 The first commercial facility using Carbon Engineering's technology is under construction in the USA and is designed to capture 500![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ton CO2 per year.40 Beyond conventional absorption-based air contactors, the hollow fiber membrane contactor (HFMC) offers a compelling alternative by uniquely integrating CO2 absorption in solutions with membrane-based gas separation (Fig. 4b). While still under development, the modular design and stable phase interface of HFMC render it a promising platform for DAC applications.41
000 ton CO2 per year.40 Beyond conventional absorption-based air contactors, the hollow fiber membrane contactor (HFMC) offers a compelling alternative by uniquely integrating CO2 absorption in solutions with membrane-based gas separation (Fig. 4b). While still under development, the modular design and stable phase interface of HFMC render it a promising platform for DAC applications.41
 | ||
| Fig. 4 (a) The common absorption-based air contactor with separate precipitation, phase separation, and calcination steps for CO2 release and capture agent regeneration; (b) the HFMC-type contactor. Reprinted with permission from ref. 41. Copyright 2024 Elsevier; (c) packings with 3D printed triply periodic minimal surfaces. Reprinted with permission from ref. 43. Copyright 2024 Wiley. | ||
The packing is also the core component of the absorption-based air contactor, directly determining the system's mass transport efficiency and CO2 capture performance. The packing used in traditional absorption towers for flue gas capture is well established, and recent research has been directed to optimizing the packing layer structure with advanced technologies such as 3D printing.42 However, due to the significant difference in CO2 concentration between air and flue gas, using the same packing from absorption towers is not effective for DAC, necessitating the development of special packing structures tailored to DAC applications. For example, Kasturi et al. developed a hybrid packing structure utilizing two specialized materials, structured packing made of PVC and random packing made of stainless steel, to optimize mass transport and enhance carbon capture performance.21 Such a packing layer exhibits a relatively high specific surface area (885 m2 m−3), achieving a CO2 absorption efficiency of 75% and a CO2 capture rate of 550 g per day, highlighting the critical role of packing materials in high-throughput contactors. An et al. proposed a packing geometry known as triply periodic minimal surfaces (TPMS), which consisted of two interpenetrating fluid domains separated by thin walls and exhibited periodicity in three dimensions (Fig. 4c).43 Such TPMS geometry enhanced mass transport efficiency by 49–61% and increased the effective gas–liquid interfacial area by 91–140% compared to the conventional packing layer.
Since absorption-based air contactors are inevitably exposed to strong alkaline absorbents, the corrosion resistance of the contactor material (e.g., steel) is particularly pivotal.44,45 Furthermore, prolonged gas–liquid contact generally leads to significant water loss via evaporation. For instance, An et al. reported that the water evaporation for per ton of CO2 captured was 6.6 tons under the conditions of 21 °C and 69% RH (relative humidity), while the water evaporation reported by Keith was 2–8 ton H2O for each ton CO2 (20 °C, 35–75% RH).39,46 Therefore, it is essential to develop efficient water management strategies tailored to the local climate conditions for the absorption-based air contactor.
No matter which type of air contactor is used, the low concentration of atmospheric CO2 necessitates a vast number of large-size contactors to maintain adequate air intake for large-scale DAC, which increases construction and operational costs and demands substantial land use, thereby eroding its practical benefits.47 Previous estimation indicates that the theoretical energy consumption required for DAC is about twice as high as or even more than that of flue gas capture.48 Therefore, it is essential to develop efficient and cost-effective air contactors or explore alternative solutions.
2.3 Air contactors retrofitted from existing facilities
Integrating DAC with existing buildings or facilities can accomplish efficient CO2 capture and meanwhile effectively reduce construction and operational costs for air contactors.49 For example, panels made from Ca(OH)2 can serve as replaceable components of buildings for DAC, with estimated CO2 capture cost of as low as $102 per tonCO2−1, comparable to CO2 capture from flue gases.50 These replaceable panels can be easily affixed to building surfaces, addressing the large land use issue of similarly sized DAC systems.Recently, Bachman et al. proposed a concept of combining DAC with railcars (Fig. 5a).51 This specialized train system is equipped with a CO2 capture system, a battery array, compression equipment, and auxiliary devices, eliminating the need for traditional fan array-based air contactors. They estimated the scaling potential of the DAC-railcar system, indicating that each railcar could capture approximately 3000 tons of CO2 annually, and potentially 0.45 gigatons of CO2 per year by 2030. Alternatively, Zheng et al. demonstrated the proof-of-concept of integrating DAC with industrial cooling towers that are operated worldwide, which could be retrofitted to capture CO2 from air by adding a suitable absorbent into the circulating water (Fig. 5b).52 Based on the experimental data, they projected that the existing cooling towers in the North American region alone could achieve an annual CO2 capture capacity of ∼4 gigatons if they could all be retrofitted with DAC function. These examples reveal the great potential of retrofitting existing facilities for DAC.
 | ||
| Fig. 5 Integrating DAC with existing facilities: (a) railcar. Reprinted with permission from ref. 51. Copyright 2022 Elsevier; (b) cooling tower. Reprinted with permission from ref. 52. Copyright 2025 Wiley. | ||
The air contactor is a key determinant of DAC's practical feasibility, governing both capital expenditure and CO2 capture efficiency. As summarized in Table 1, current contactor designs fall into two broad categories, adsorption-based and absorption-based systems, each with distinct trade-offs. Among adsorption-based configurations, traditional fixed bed contactors with structured packings offer high modularity and scalability, whereas the emerging rotating-bed contactors enhance mass transfer within a compact footprint. In comparison, conventional absorption-based contactors employing structured packings and sprays leverage mature gas–liquid contacting technologies and decouple CO2 capture from absorbent regeneration, offering operational flexibility for industrial scaling. However, they remain susceptible to water loss and corrosion. Membrane contactors present a promising alternative by providing a stable, high-surface-area interface, albeit at higher material costs.
| Type | Configuration | Key advantages | Challenges/limitations | Ref. |
|---|---|---|---|---|
| Adsorption-based | Static structured packings | Highly modular | Batch/cyclic operation | 36, 37 |
| Scalable | ||||
| Rotating bed | Intensified mass transfer | Mechanical complexity | 35 | |
| Compact footprint | Extra energy consumption for rotation | |||
| Continuous capture/release | ||||
| Building | Using existing urban surfaces | Low capture flux | 50 | |
| Significant capital expenditure saving | Uncontrolled capture rate | |||
| Railcar | Using “free” high-speed airflow | Influence on railcar | 51 | |
| Absorption-based | Static structured packings/spray | Mature gas–liquid contact technology | Water loss by evaporation | 39, 42, 43 |
| Decoupled capture and regeneration units | Corrosion | |||
| Cost-effective absorbent solutions | ||||
| Membrane | Stable gas–liquid interface | High membrane cost | 41 | |
| High surface-to-volume ratio | Low capture flux | |||
| Cooling tower | Using existing industrial facilities | Balancing DAC operation with cooling function | 52 | |
| Significant capital expenditure saving |
Retrofitting existing facilities into adsorption- or absorption-based air contactors represents a promising approach to reduce DAC costs. However, its practical implementation faces several engineering challenges. For instance, although the laboratory study has demonstrated the feasibility of integrating DAC into industrial cooling towers, large-scale deployment may compromise the primary heat dissipation function of such towers, necessitating a comprehensive assessment. These challenges highlight that the success of retrofitting strategies hinges not only on the capture technology itself but also on optimized integration with the host infrastructure.
3 Capture agents
The choice of capture agent is of great importance in DAC, as it directly governs the efficiency of the entire process, from initial CO2 capture to subsequent CO2 release and its own regeneration. This section examines the characteristics of two typical types of capture agents, solid adsorbent materials and liquid absorbent solutions, discussing their advantages and limitations for DAC applications.3.1 Solid adsorbents
Adsorbents can be classified into physical and chemical types based on their properties and mechanisms for CO2 capture. Physical adsorption is driven by intermolecular attraction with weaker binding forces, resulting in lower heat enthalpy and relatively higher rates for both CO2 adsorption and desorption. Common physical adsorbents include zeolites, activated carbons, alumina, MOFs, and covalent organic frameworks (COFs), which are porous, possess large surface areas, and can be modified and functionalized.53 At present, physical adsorbents are primarily utilized for post-combustion CO2 capture from flue gases, with limited applications in DAC.54 This limitation is largely due to their low selectivity toward CO2 whose concentration is low in air. Additionally, air humidity can lead to competitive adsorption between water and CO2 molecules on physical adsorbents, significantly impacting CO2 adsorption performance.Accordingly, a major focus of recent research on physical adsorbents is to understand and alleviate the detrimental effects of water co-adsorption. For example, Hu et al. systematically compared the CO2 and water adsorption behaviour of two typical zeolites, FAU and MFI, and evaluated their CO2 capture performance under high relative humidity (75%).55 They show that increasing the Si/Al ratio of the zeolite slightly reduces its CO2 uptake but markedly enhances hydrophobicity, enabling the material to retain relatively high CO2 capacity under humid conditions. More importantly, they developed an advanced zeolite adsorbent that achieves high CO2 capacity, fast adsorption kinetics, and excellent humidity resistance by repairing structural defects and eliminating hydrophilic silanol groups of the MFI zeolite.
In comparison, chemical adsorbents exhibit greater tolerance to moisture and show a stronger affinity toward CO2. Thus, DAC studies mainly utilized chemical adsorbents. Metal oxides, such as CaO, capture CO2 via carbonation reactions to form the corresponding carbonates (i.e., CaCO3). The adsorbent can then be regenerated with CO2 released through high-temperature calcination. However, the inherently low specific surface area of metal oxides limits the availability of their active sites for CO2 adsorption. A widely adopted strategy to overcome this limitation is to disperse metal oxide onto a highly porous support. For instance, Huang et al. developed a composite adsorbent for DAC by loading CaO onto acid-modified attapulgite (HcATP).56 They revealed that the CO2 uptake of the composite arises from a synergistic effect of physical and chemical adsorption. Specifically, the HcATP support alone shows a CO2 uptake of 212.4 μmol g−1 through physisorption, while the loaded CaO provides additional adsorption via carbonation, significantly increasing the total CO2 uptake to 426.7 μmol g−1, underscoring the important contribution of chemisorption. However, it should be noted that the CO2 adsorption capacity of this inorganic adsorbent is relatively low, while the regeneration temperatures for adsorbents are usually high (carbonate calcination occurs at >800 °C).
To date, amine-functionalized porous materials are the most extensively studied chemical adsorbents for DAC. These amine-based adsorbents utilize traditional physical adsorbents as supports whose porous structure facilitates the dispersion of amines, while the amines serve as the adsorption sites for CO2 capture (Fig. 6). Thus, selecting the appropriate amine, support material, and method to link them together is crucial for optimizing the performance of the resulting adsorbent.
 | ||
| Fig. 6 Preparation methods and key components of amine-functionalized adsorbents. (a) Two typical methods for immobilizing amines onto support materials; common (b) amines and (c) support materials for preparing amine-functionalized adsorbents. The alumina in (c) is reprinted with permission from ref. 57. Copyright 2020 Elsevier; the MOF in (c) is reprinted with permission from ref. 58. Copyright 2024 American Chemical Society; the COF in (c) is reprinted with permission from ref. 59. Copyright 2022 American Chemical Society. | ||
Amine-based adsorbents are categorized into two classes according to their preparation methods: physical impregnation and covalent binding (Fig. 6a). Amine-impregnated adsorbents involve the physical impregnation of amine oligomers or polymers onto porous support materials by stirring the support materials with the desired amines in an organic solvent. The advantages of easy preparation and good performance make amine-impregnated adsorbents attract intensive investigation. A variety of amine oligomers or polymers can be used to prepare amine-impregnated adsorbents, including polyethyleneimine (PEI), poly(propyleneimine) (PPI), tetraethylenepentamine (TEPA) (Fig. 6b); and the support can be any type of porous material, such as silica, alumina, and MOFs, and COFs (Fig. 6c). Table 2 summarizes a variety of amine-based adsorbents and their performance for DAC applications.
| Type | Amine | Support | Capture conditionsa | RHb | CO2 uptake (mmol g−1) | Ref. |
|---|---|---|---|---|---|---|
| a Simulated air is used for all the studies listed.b Relative humidity.c TEPA is tetraethylenepentamine, DEA is diethanolamine.d PEI is polyethyleneimine.e AEEA is N-(2-aminoethyl)ethanolamine.f TCPB is 1,3,5-tris(4-cyanomethylphenyl)benzene, BPDA-N3 is 3,3′-bis[(6-azidohexyl)oxy]4,4′-biphenyl-dicarbaldehyde.g TFTDA is 2′,3,′5′,6′-tetrafluoro-[1,1′:4′,1″terphenyl]-4,4″-dicarbaldehyde, TAPPy is 1,3,6,8-tetrakis(4-aminophenyl)pyrene. | ||||||
| Physical impregnation | 40 wt% TEPA & 10 wt% DEAc | SBA-15 | 400 ppm, 25 °C | Dry | 1.93 | 60 |
| 50 wt% PEId | Commercial resin particles | 400 ppm, 25 °C | 25% | 2.95 | 61 | |
| 70 wt% PEI | SBA-15 | 400 ppm, 35 °C | 78% | 3.48 | 62 | |
| 40 wt% PEI, 40 wt% TEPA | Mesoporous γ-Al2O3 | 400 ppm, 25 °C | Dry | 0.9, 1.8 | 63 | |
| 30–50 wt% TEPA | MOF MIL-101(Cr) | 400 ppm, −20 °C | Dry | 0.72–1.11 | 64 | |
| 45 wt% PEI | Mg–Al-CO3 layered double hydroxide | 400 ppm, 25 °C | Dry | 1.82 | 65 | |
| 50 wt% PEI | Mesoporous cellular foam | 400 ppm, 25 °C | Dry-80% | 0.41–0.71 | 66 | |
| 40 wt% PEI | Commercial resins | 400 ppm, 28 °C | 50% | 1.47–1.60 | 67 | |
| 25 wt% DEA & 25 wt% PEI | SBA-15 | 400 ppm, −5 to 25 °C | 20% | 1.29 | 68 | |
| 30 wt% PEI | MgxAl-CO3 layered double hydroxides & MgxAl-O mixed metal oxides | 400 ppm, −20 to 25 °C | 70% | 2.0–3.2 | 69 | |
| 70 wt% TEPA | Hierarchical silica | 420 ppm, 30 °C | 50 ± 3% | 5.2 | 70 | |
| 71 wt% TEPA | Meso-macroporous melamine formaldehyde | 420 ppm,30 °C | Dry | 2.65 | 71 | |
| 70 wt% PEI | Zeolite 13X & MOF MIL-101(Cr) | 400 ppm, −20 °C | Dry-70% | 1.05–1.43 | 72 | |
| Covalent binding | AEEAe | MOF MIL-100 (Cr) | 400 ppm, −25 to 0 °C | 1% | 1.91–2.42 | 73 |
| N1-(3-Trimethoxysilylpropyl)-diethylenetriamine | Hierarchical bimodal mesoporous silica | 415 ppm, 25 °C | Dry | 1.04 | 74 | |
| 47 wt% PEI | Mesoporous silica foam | 400 ppm, 25 °C | 65% | 1.50 | 75 | |
| SH-bPEI | COF (synthesized by TFTDA and TAPPy)g | 400 ppm, 25 °C | Dry-75% | 0.48–1.24 | 76 | |
| Aziridine | COF (synthesized by TCPB and BPDA-N3)f | 400 ppm, 25 °C | Dry-50% | 0.96–2.05 | 77 | |
PEI has been widely utilized to prepare amine-impregnated adsorbents for its low degree of polymerization and large abundance of primary amine groups (Table 2). PEI has two typical types of structures, including linear chains with amine groups mostly secondary and dendritic structure with tertiary amines serving as branching points (Fig. 6b). The desorption enthalpy of linear PEI (74–75 kJ mol−1) is slightly lower than that of branched PEI (77–80 kJ mol−1), giving linear PEI a higher CO2 desorption rate.78 A significant limitation of PEI is its high susceptibility to oxidative degradation in high-temperature, oxygen-rich environments. Consequently, the regeneration of PEI requires a vacuum or inert atmosphere, which raises process complexity and energy consumption.79
PPI demonstrates better oxidation resistance compared to PEI. Yang et al. compared the performance of amine-impregnated adsorbents with 20 wt% PEI and 20 wt% PPI supported on a MOF (UiO-67(Zr)) under ambient air conditions (∼400 ppm CO2).80 The PPI-based adsorbent exhibited a higher CO2 capture capacity (0.42 mmol g−1) than the PEI counterpart (0.38 mmol g−1). After exposing both adsorbents to ambient air for 7 days, PPI remained fresh, while PEI experienced a 22% loss in amine efficiency. Rosu et al. conducted the prolonged aging of PPI (about 2 years) and studied its performance for DAC.81 Adsorbents made from aged PPI showed a slight decline in CO2 capture performance (approximately 20%) compared to the fresh polymer, but the overall CO2 cycling performance remained excellent (the number of cycles is 20, the average CO2 capacity is 0.96 mmol g−1). However, the synthesis of the PPI monomer is relatively complex, involving the use of various acid initiators (i.e., HBr, HClO4, HCl, CH3SO3H).82
The molecular weight of the amine significantly influences the performance of amine-functionalized adsorbents. While high molecular weight PEI can increase the content of amine groups, it may impede CO2 diffusion within the support structure, resulting in reduced CO2 uptake. Smaller amines such as TEPA are reported to improve DAC owing to their high nitrogen content and reduced stacking tendency, which enhances CO2 mass transport within porous supports.83
The performance of amine-impregnated adsorbents can be influenced by the structure of support materials, because the porous structure of the support governs amine dispersion and CO2 accessibility. Rim et al. demonstrated this by comparing TEPA impregnated on two types of support materials, MIL-101(Cr) and γ-Al2O3 (Fig. 7a and b).84 In situ IR spectroscopy revealed that weak chemisorption (forming carbamic acid) was the dominant pathway for CO2 capture over MIL-101(Cr)-supported TEPA, whereas strong chemisorption (forming carbamate) prevailed on γ-Al2O3-supported TEPA. This mechanistic divergence was attributed to the stronger amine–support interaction in MIL-101(Cr), which possessed a significantly higher surface area to pore volume ratio than γ-Al2O3. This strong interaction likely stabilized the carbamic acid intermediate via surface hydroxyls or water. In contrast, the weaker interaction of amine with γ-Al2O3 allowed for greater amine–amine interaction, facilitating the formation of the ammonium carbamate pair. Therefore, the CO2 capture pathway of impregnated amines can be engineered by modulating the amine–support interaction, highlighting the important effect of the support materials.
 | ||
| Fig. 7 The influence of different support materials on CO2 capture. (a) MIL-101(Cr) and (b) γ-Al2O3. Reprinted with permission from ref. 84. Copyright 2023 American Chemical Society; (c) synthesis of PEI-grafted COF-709, (d) CO2 uptake performance under varying RH conditions, and (e) stability over multiple CO2 adsorption–desorption cycles using PEI-grafted COF-709 for DAC. Reprinted with permission from ref. 76. Copyright 2024 American Chemical Society. | ||
In practical DAC applications, environmental conditions vary with seasons. Understanding the effect of air temperature and humidity is important to accomplish optimal performance of amine-impregnated adsorbents for DAC. Priyadarshini et al. studied a series of TEPA-impregnated γ-Al2O3 with varying amine loadings (10, 20, and 40 wt%) for CO2 capture under ambient (25 °C) and sub-ambient (−20 °C) conditions.63 At 25 °C, the CO2 capacity of these adsorbents increased with amine loading, with 40 wt% TEPA-impregnated γ-Al2O3 showing the highest CO2 uptake capacity (1.8 mmol g−1). At −20 °C, however, the CO2 uptake increased for the adsorbent with 10 wt% TEPA but slightly decreased for the adsorbents with 20 and 40 wt% TEPA. This was because high pore filling and restricted mobility of amine chains arising from high amine loadings caused pore blocking of the adsorbents at sub-zero temperatures. This hindered CO2 diffusion, leading to reduced capacity for CO2 capture. A similar phenomenon was observed in TEPA and PEI-impregnated MOFs.64
Humidity is another critical factor influencing CO2 adsorption, typically leading to higher CO2 capture capacity under high humidity conditions. Under dry conditions, CO2 reacts with two amine molecules to form ammonium carbamate ion pairs (i.e., RNH3+COO− and R1R2NH2+COO− for primary and secondary amines, respectively). This reaction limits the capacity to 0.5 mol CO2 per mole of amine and contributes to cross-linking within or between amine chains, which significantly impedes CO2 diffusion. In contrast, under moist conditions, water-stabilized ions (e.g., ammonium bicarbonate or hydronium carbamate) are thought to bind through a single amino group.85 This monodentate binding can promote CO2 diffusion and enhance CO2 capture capacity. The humidity also affects the durability of adsorbents. Carneiro et al. studied the role of water in the degradation of amine-based adsorbents using elemental analysis, solid-state NMR, and in situ infrared spectroscopy.86 They established the cleavage of terminal C–N bonds as the dominant deactivation mechanism in PEI-based amine adsorbents, and the presence of highly reactive oxygen species (e.g., OH·) in water exacerbates such cleavage. In consequence, drier air minimizes the impact of adsorbed water on amines, while low temperature leading to reduced absolute humidity enhances adsorbent durability.
A significant issue of amine-impregnated adsorbents is that the physical impregnation of amines leads to disordered amine stacking, which has adverse effects on CO2 diffusion. Additionally, the weak interaction between the amine and support makes these adsorbents more susceptible to detach. In contrast, amine-grafted adsorbents through covalent binding are more stable since their amines are chemically bonded to the support materials.
Amine-grafted adsorbents are typically prepared via two primary methods: one is through the silanization reaction between a silica support and aminosilanes; the other is through C–O/C–S bond formation (Fig. 6a). Anyanwu et al. grafted N1-(3-trimethoxysilylpropyl)diethylenetriamine onto mesoporous silica under anhydrous (dry-grafting) and hydrous (wet-grafting) conditions. A significantly higher amine loading was accomplished in the wet-grafting adsorbents (49%) than in the dry-grafting ones (28%).74 This was because the presence of water enhanced the hydrolysis of unreacted alkoxyl groups and introduced additional hydroxyl groups, thereby increasing amine loading in wet-grafting. The higher amine loading allowed the amine-grafted silica to achieve a CO2 uptake of 1.04 mmol g−1 at 415 ppm of CO2.
Beyond silica, a variety of other support materials can also be functionalized with amines to fabricate efficient DAC adsorbents. A notable example is the PEI-grafted COF (COF-709) developed by Li et al. via an aromatic nucleophilic substitution reaction, forming C–S bonds between PEI and COF (Fig. 7c).76 When tested with 400 ppm CO2 at 25 °C, COF-709 exhibited a remarkable CO2 uptake that was highly dependent on RH, increasing significantly from 0.48 mmol g−1 under dry conditions to 1.24 mmol g−1 at 75% RH (Fig. 7d), similar to amine-impregnated adsorbents.85,86 13C NMR spectroscopy revealed that this enhancement was due to a shift of the adsorption products: while carbamate and carbamic acid formed under dry conditions, the presence of moisture promotes the formation of bicarbonate along with carbamate, increasing CO2 capture capacity. Furthermore, the C–S bonds used to anchor PEI within the COF pores provided exceptional chemical stability, preventing amine loss (Fig. 7e). As a result, COF-709 exhibited excellent cycling stability, maintaining its CO2 uptake without degradation over 10 consecutive adsorption–desorption cycles under simulated DAC conditions. In another work, the same group developed a polyamine-functionalized COF through a ring-opening polymerization reaction with aziridine, which exhibited similarly good performance for DAC.77 These studies confirm the effectiveness and durability of amine-grafted COFs for efficient CO2 capture in an open air environment.
In addition to amine-based adsorbents, Li et al. recently reported a new class of adsorbents known as “charged sorbents”. Such adsorbents (also named as PCS-OH) were synthesized by electrochemically inserting reactive hydroxide ions into a porous carbon (Fig. 8a).87 The hydroxide ions present in PCS-OH serve as reactive sites for selective CO2 capture from air, forming (bi)carbonate species (Fig. 8b). PCS-OH was evaluated for DAC under simulated dry air with 400 ppm CO2 at 30 °C, showing a stable CO2 capacity of ∼0.2 mmol g−1 over 10 repeated adsorption and desorption cycles (Fig. 8c). It should be noted that the CO2 capacity of PCS-OH is generally lower than amine-based adsorbents due to its limited hydroxide content, but this can be improved by optimizing the synthesis procedure to increase the hydroxide content in the adsorbents. This study lays the foundation for developing a new family of effective and low-cost adsorbents for DAC.
 | ||
| Fig. 8 Charged sorbents for DAC. (a) The preparation steps, (b) proposed mechanism for CO2 capture, and (c) cycling capacities for ten CO2 adsorption–desorption cycles over the charged sorbents. Reprinted with permission from ref. 87. Copyright 2024 Springer Nature. | ||
3.2 Liquid absorbents
In CO2 capture by absorbent solutions, CO2 first comes into contact and reacts with the absorbent, getting separated from air. Then, the CO2-rich absorbent solution is transported to the regeneration unit to release CO2 and regenerate the absorbent, where the absorbent effluent can be reused for the next cycle of CO2 capture. The absorption of CO2 depends on an interfacial equilibrium between air and absorbent solutions. Thus, the capture capacity of CO2 in the absorbent solution is influenced by the absorbent property as well as the air temperature and pressure. Common absorbents used in DAC are alkaline solutions, including inorganic and organic bases dissolved in water (Fig. 9a and b). | ||
| Fig. 9 Two common types of liquid absorbents: (a) inorganic alkaline solutions and (b) organic amine and amino acid salt solutions; (c) the CO2 loading capacity of amino acid salt and MEA solutions at 313 K. Reprinted with permission from ref. 88. Copyright 2022 De Gruyter. | ||
Hydroxide aqueous solutions (e.g., NaOH, KOH) have been widely utilized to eliminate CO2 from air in closed spaces like submarines and spacecraft. They can easily react with CO2 to form carbonate, enabling selective CO2 capture from air (eqn (1) and (2)). These alkaline solutions are extremely attractive as CO2 absorbents because of their excellent stability and affordable price. However, there are some drawbacks to be addressed when using them for large-scale applications. For instance, when Ca(OH)2 comes into contact with CO2, it generates CaCO3, which may plug the packing and piping components of the air contactor, posing challenges for long-term operation. Furthermore, the limited solubility of Ca(OH)2 in water results in a low CO2 capture capacity. As for other hydroxide solutions (e.g., NaOH and KOH), the regeneration process requires extremely high temperatures (e.g., >340 °C for Na2CO3 solution), leading to the complete boiling of the aqueous solution.89 Thus, the NaOH and KOH absorbent solutions after CO2 capture are typically subjected to reaction with Ca(OH)2 to regenerate the absorbent and form CaCO3, which is then calcined to release CO2. It should be emphasized that this calcination process occurs at even higher temperatures (≥700 °C), resulting in large energy penalty.90 The mixture of carbonate and hydroxide solutions (e.g., K2CO3/KOH, Na2CO3/NaOH) has also been used for DAC, because mixing carbonate and hydroxide solutions creates a synergistic system, in which hydroxides ensure fast CO2 capture kinetics, while carbonates offer low corrosivity.91 However, the mixture also encounters challenges similar to those associated with pure hydroxide solutions.92,93
| 2OH− + CO2 ⇌ CO32− + H2O | (1) |
| CO32− + CO2 + H2O ⇌ 2HCO3− | (2) |
Amine solutions are well-established absorbents for CO2 capture mostly from flue gas. Compared to hydroxide solutions, the amine regeneration process is milder, usually at lower temperatures (i.e., 80–120 °C), thereby featuring lower energy consumption. Commonly used amines include monoethanolamine (MEA), ethylenediamine, triethylamine, etc.94 CO2 absorption by amine occurs via the zwitterion mechanism, where the amine reacts with CO2 to form protonated amine and carbamate/bicarbonate. Both primary and secondary amines undergo similar reactions to form carbamate (eqn (3), (4), (6) and (7)), with a maximum theoretical CO2 uptake of 0.5 mol CO2 per mol amine. However, a certain carbamate can undergo hydrolysis in the presence of water to form bicarbonate (eqn (5) and (8)), which can slightly increase the CO2 uptake. In contrast, tertiary amines have a much higher theoretical CO2 loading (1.0 mol CO2 per mol amine) because they cannot directly capture CO2 but instead act as catalysts to promote the hydrolysis reaction of CO2 (eqn (9)). Thus, the performance of amines for CO2 capture is highly dependent on their chemical structures. Barzagli et al. carried out a screening study on the performance of different aqueous amine solutions for DAC.95 They found that unhindered primary amines, such as MEA and isopropanolamine, are the most effective absorbents for DAC, as efficient as aqueous hydroxides. This high efficiency was attributed to the formation of a high-concentration carbamate. In contrast, tertiary amines show poorer performance as they cannot form carbamate, but instead rely on the slower kinetics of bicarbonate formation.96 Interestingly, aqueous 2-amino-2-methyl-1-propanol (AMP), an efficient flue gas absorbent, was found unsuitable for capturing extremely diluted CO2, highlighting the differences between DAC and conventional CO2 capture. Therefore, the optimal absorbents for point-source capture are not necessarily the best choice for DAC.
| RNH2 + CO2 ⇌ RNH2+COO− | (3) |
| RNH2+COO− + RNH2 ⇌ RNH3+ + RNHCOO− | (4) |
| RNHCOO− + H2O ⇌ RNH2 + HCO3− | (5) |
| R1R2NH + CO2 ⇌ R1R2NH+COO− | (6) |
| R1R2NH+COO− + R1R2NH ⇌ R1R2NH2+ + R1R2NCOO− | (7) |
| R1R2NCOO− + H2O ⇌ R1R2NH + HCO3− | (8) |
| R1R2R3N + H2O + CO2 ⇌ R1R2R3NH+ + HCO3− | (9) |
Indeed, many amine-based absorbents are unsuitable for DAC due to their oxidative degradation in the capture of low concentration CO2. On the one hand, amine degradation compromises carbon capture stability, particularly in the presence of high concentration of oxygen.86 On the other hand, amine degradation products are directly released into the atmosphere.97 In DAC systems with large air flow, these emissions may lead to severe environmental impacts. Consequently, most amine-based absorption solvents are primarily utilized in flue gas capture rather than DAC.
Amino acid salts (AAS) are promising alternative absorbents to traditional amines, showing multiple advantages such as low toxicity, low volatility, antioxidant properties, resistance to thermal degradation, and fast reaction kinetics with CO2.98,99 Currently, the most extensively studied AAS for DAC applications include glycine (Gly), lysine, sarcosine, leucine, valine, phenylalanine, and arginine.100–103 AAS absorbent solutions are typically prepared by mixing amino acids with a hydroxide solution like NaOH or KOH.104 Similar reactions occur when using AAS for DAC (eqn (10) and (11)), but AAS solutions are slightly more alkaline than common amine solutions due to the presence of both amino and carboxylate groups. Fig. 9c shows that many AAS absorbent solutions, such as lysinate, glycinate, histidinate, and glutaminate, exhibit superior CO2 capture capacity compared to the conventional MEA solution.88 In a following study, McQuillan et al. found that K-Gly exhibited comparable CO2 uptake to MEA in DAC but showed lower energy consumption for regeneration.105 More recently, Xiao et al. compared the stability of AAS and amine absorbents in air.106 After exposure in air for 7 days, 3 M K-Gly showed a degradation rate of 16.7 mmol per mole K-Gly, while the degradation rate of common amines (3 M MEA and AMP) approximately doubled (e.g., 29.2 mmol per mol MEA and 31.6 mmol per mol AMP). For these advantages, increasing efforts are directed to exploring AAS for DAC.
| 2NH2RCOO− + CO2 ⇌ −OOCNHRCOO− + NH3+RCOO− | (10) |
| −OOCNHRCOO− + H2O ⇌ NH2RCOO− + HCO3− | (11) |
4 CO2 release and capture agent regeneration
CO2 release accompanied by adsorbent/absorbent regeneration is a critical step in continuous DAC processes. In general, the methods used for CO2 release and adsorbent/absorbent regeneration in DAC show some similarity to those in flue gas carbon capture. However, differences also exist due to the significantly lower CO2 uptake in DAC-based systems as a result of low atmospheric CO2 concentration. Currently, the main approaches to release CO2 from DAC systems are temperature swing and precipitation-phase separation, which are deployed by two representative DAC companies, Climeworks and Carbon Engineering, respectively. With the growing emphasis on sustainable energy, electrochemical methods are emerging as a compelling option for CO2 release due to their advantages of using renewable electricity, controllable reaction rates, and mild operation conditions. In this section, we will scrutinize these regeneration methods.4.1 Temperature swing
Thermal desorption based on temperature swing is a common method for CO2 release and regeneration of solid adsorbents. At elevated temperatures, the bond energy between CO2 and the adsorbent is weakened, facilitating the release of CO2 gas. In temperature swing adsorption (TSA) systems, CO2 capture occurs under ambient conditions, while CO2 release operates at elevated temperatures (100–120 °C).107 The desorption temperature is the most critical parameter in TSA, directly influencing the energy consumption of the systems. Generally, CO2 release at higher temperature leads to higher energy consumption (≥6.0 GJ tonCO2−1).38To lower the operation temperature, TSA can be integrated with vacuum systems, which is known as temperature vacuum swing adsorption (TVSA).108 As a result, TVSA shows reduced energy consumption and improved stability for cyclic CO2 capture. A good application of the TVSA process in DAC is demonstrated by Climeworks. Their system operates at temperatures below 100 °C with an energy consumption of 5.40–7.20 GJ tonCO2−1.109 Similar energy consumption (4.20–8.60 GJ tonCO2−1) for TVSA was also reported in the literature.110 Recently, the company has introduced a new generation of DAC technology, which incorporates novel filter materials and is reported to reduce energy consumption by roughly 50% compared to earlier systems.111 This upgraded system is planned for deployment in the United States, aiming at reducing the DAC cost to $250–300 per tonCO2−1 by 2030.
It has to be noted that the TSA/TVSA system requires closing the inlet and outlet during CO2 release and heating the entire interior space of the air contactor, leading to unnecessary heat loss. To further optimize the system, researchers have developed a novel electrically driven temperature swing adsorption (e-TSA) method, which can minimize the heat loss as it employees joule heating (also known as resistive heating) to generate thermal energy only on the conductor or the material itself to release CO2.112,113 This method allows the heat to be transferred directly from the inside to the outside of the adsorbent, thereby enhancing the energy efficiency of the temperature swing. For example, Lee et al. developed a DAC module made of a PEI-impregnated adsorbent-coated carbon fiber, which reached the CO2 release temperature within one minute when a 7 V voltage was applied (Fig. 10a).114 This DAC module released CO2 and regenerated the adsorbent six times faster than the traditional TSA thermal desorption, with an energy consumption of 7.20 GJ tonCO2−1 and only 7% of the total heating lost to the surrounding environment. Later, Li et al. employed joule heating at a voltage of 7–8 V to release CO2 from “charged sorbents” at low temperatures of 90–100 °C, showing an energy consumption of 6.50 GJ tonCO2−1 under 11% RH.87 More recently, Lee et al. established an electrothermal-vacuum swing adsorption (e-TVSA) process by integrating joule heating with a vacuum system to release CO2 from an ethylenediamine-grafted Y zeolite (EDA-Y) adsorbent.115 In this case, a significantly reduced voltage of 3 V was sufficient to release CO2 and regenerate the adsorbent, resulting a lower energy consumption of 6.1 GJ tonCO2−1.
 | ||
| Fig. 10 (a) An electrically driven temperature swing adsorption module for DAC. Reprinted with permission from ref. 114. Copyright 2023 Elsevier; (b) solar-driven CO2 release coupled with thermal adsorbent regeneration. Reprinted with permission from ref. 116. Copyright 2024 Springer Nature; (c) solar-assisted DAC system enabling in situ CO2 conversion. Reprinted with permission from ref. 117. Copyright 2025 Springer Nature. | ||
Solar concentrators offer a way of direct use of solar energy for thermal regeneration of adsorbents. A solar-driven DAC system developed by Wang et al. accomplished near-complete CO2 release (∼100%) in 50 minutes under 500–700 W m−2 solar irradiance with 63.1% photothermal conversion efficiency (Fig. 10b).116 Kar et al. reported a similar CO2 release method, compatible with most commercial adsorbents regenerable at 80–100 °C.117 Additionally, they explored in situ CO2 conversion with the DAC system, showing a maximum yield of ∼24 μmol gTiO2−1 for syngas production via solar photocatalysis (Fig. 10c). These two studies demonstrate the feasibility of integrating renewable energy into the process of CO2 release and adsorbent regeneration. Solar-driven CO2 release/adsorbent regeneration systems enable decentralized deployment in remote locations without extensive thermal infrastructure, positioning them as a promising DAC technology.
For the regeneration of liquid absorbents, direct heating of the CO2-loaded absorbent solutions is commonly applied to organic amines but is unsuitable for inorganic hydroxide solutions due to the high temperatures required.118,119 Although organic amine solutions have been widely deployed in flue gas CO2 capture, they are prone to oxidative degradation in air, which explains the scarce examples of their applications in DAC. AAS solutions are more stable and employed as promising absorbents for CO2 capture from air. After CO2 capture, AAS solutions can be regenerated by heating them to a certain temperature (70–90 °C) at a lowered pressure (21 kPa).41 However, the direct heating of absorbent solutions faces challenges such as solvent evaporation, oxidative degradation, and the need to heat large volumes of liquid.120,121 Therefore, new technologies have been developed to regenerate liquid absorbents in DAC (vide infra).
4.2 Precipitation-phase separation
DAC systems based on aqueous sodium/potassium hydroxide solutions usually rely on an additional calcium cycle to release the captured CO2 and regenerate the sodium/potassium hydroxide solutions. This is a typical precipitation-phase separation method that precipitates the CO2 captured, significantly reducing the volume or mass of substance for follow-up thermal treatment.Fig. 11a shows a potassium–calcium loop for CO2 release in the DAC process developed by Carbon Engineering.39 In this process, a K2CO3 solution formed after CO2 capture by KOH solutions reacts with Ca(OH)2 to regenerate KOH and yield the CaCO3 precipitate, which is separated and calcined at 900 °C to release gaseous CO2 and produce solid CaO. Ca(OH)2 is then regenerated through hydration of CaO. The major challenge of the potassium–calcium loop is its high energy intensity and complex operation steps. The regeneration energy of this process is estimated to be 5.25–8.81 GJ tonCO2−1, which represents 80% of the total energy consumption in the DAC system.
 | ||
| Fig. 11 CO2 release and absorbent regeneration by precipitation-phase separation. (a) Potassium–calcium loop; (b) AAS-guanidine loop. | ||
In order to reduce the energy consumption, Brethome et al. developed a new precipitation-phase separation system relying on the reaction between CO2-loaded AAS aqueous solution (glycinate and sarcosinate) and a guanidine compound (2,6-pyridinebis(iminoguanidine), PyBIG), which forms insoluble carbonate crystals and regenerates the AAS absorbent (Fig. 11b).122 The carbonate crystals can decompose at mild temperatures (80–120 °C) to release CO2 and regenerate PyBIG at a percentage of 92%. Liu and co-workers uncovered the structural evolution and decomposition mechanism of the GBIG-bicarbonate (GBIGH2(CO3)2(H2O)2) crystal formed by the reaction of CO2-loaded AAS with glyoxal-bis(iminoguanidine) (GBIG) during the CO2 release process using in situ wide-angle X-ray scattering, attenuated total reflectance-Fourier transform infrared spectroscopy, and other measurements.123 Their findings indicate that CO2 and water molecules are released simultaneously from the GBIG-bicarbonate crystal during the thermal regeneration process at 97–134 °C.
The guanidine compound can also be utilized directly to capture CO2 without the use of AAS. For instance, Cai et al. reported a trichelating iminoguanidine (BTIG) for DAC.124 Unlike previous works, this guanidine can directly and strongly bind with CO2 to form insoluble carbonate crystals without the aid of AAS. In the reversible process, BTIG-CO2 crystals release CO2 and regenerate the guanidine at mild temperatures (60–150 °C), showing much lower energy consumption (2.27 GJ tonCO2−1) than CaCO3 calcination.
4.3 Electrolysis
In recent years, electrolysis has shown significant potential in CO2 release, as it avoids external heating or vacuum conditions. Electrolysis exhibits excellent compatibility with absorbents that are sensitive to heating and can be applied in various scenarios for CO2 removal, particularly in DAC. There are two reasons for this. Firstly, the modular design of electrolyzers allows them to be deployed in diverse environments, including urban, industrial, and remote areas, which aligns with the geographically flexible nature of the DAC technologies. Secondly, the electrochemical equipment is easy to maintain and can be connected to renewable energy sources, making the process of CO2 release and absorbent regeneration more sustainable and cost-effective. The methods for electrochemical CO2 release mainly include pH swing and redox mediation.| 2H2O − 4e− → 4H+ + O2(g) | (12) |
| 4H2O + 4e− → 4OH− + 2H2(g) | (13) |
The pH swing performance can be significantly improved using bipolar membrane electrodialysis (BPMED). The core of this system is the bipolar membrane (BPM), which is constituted by a cation- and an anion-exchange layer. When applying a potential, the BPM dissociates water into H+ and OH−, generating a sharp pH gradient across the membrane (Fig. 12a). A typical BPMED stack comprises multiple BPMs alternating with anion exchange membranes (AEMs), with acidification and basification chambers in between. During operation, carbonate/bicarbonate ions migrate through the AEM into the acidification chamber, where H+ triggers CO2 release. Simultaneously, OH− in the basification chamber regenerates the absorbent solution.
In an early work, Eisaman et al. reported a BPMED stack for releasing CO2 from potassium carbonate and bicarbonate solutions, which allows for CO2 release and absorbent regeneration at the energy of 100 to 200 kJ molCO2−1 (i.e., 2.27 to 4.54 GJ tonCO2−1) at the low current density (5 mA cm−2).127 This work utilized an electrolyzer stack composed of seven unit cells, with the AEM reaching 0.14 m2, demonstrating the scalability potential of this technology. Recently, Castaño et al. systematically investigated the influence of parameters such as current density on the performance of a BPMED.128 Among the tested current densities, 100 mA cm−2 showed the highest CO2 desorption efficiency but also the highest energy consumption, whereas 25 mA cm−2 exhibited the lowest energy consumption (8.8 GJ tonCO2−1) but lower CO2 desorption. These results reveal that the current density is a key factor governing the overall performance of BPMED, affecting both CO2 release efficiency and energy consumption.
The main challenge of BPMED technology is its high energy consumption and high expense of ion exchange membranes. Sabatino et al. conducted a techno-economic analysis of BPMED and proposed that future development should be aimed at enhancing the electrical conductivity and overall stability of ion exchange membranes.129 With more durable and cost-effective membranes, the total cost of BPMED could potentially fall below $250 per tonCO2−1.
Fig. 12b shows another efficient system for electrochemical CO2 release and absorbent regeneration, called three-chamber electrolysis system comprising cathode, acidification, and anode chambers.130 In this system, the anode oxidizes H2 to produce H+, which is transferred through the ion exchange membrane to the acidification chamber, while the cathode produces OH− by water reduction to regenerate the absorbent solution and H2. Notably, this system avoids the generation of O2 from water reduction and instead utilizes the hydrogen generated at the cathode for proton supply. In the chemical industry, the storage and transportation of H2 often incur more costs and safety risks. This system addresses these challenges by effectively utilizing H2, making it particularly promising for application.
For example, Shu et al. employed amine-functionalized ion-exchange resins for DAC and utilized a three-chamber electrolytic cell for CO2 release.131 This study reported a very high energy consumption of 537 ± 33 kJ molCO2−1 (i.e., 12.20 ± 0.75 GJ tonCO2−1) at a current density of 20 mA cm−2, which was attributed to the large electrode overpotential of the cell. Zheng et al. also employed a similar three-chamber electrolytic cell to release CO2 with a lower energy consumption of 5.20–8.08 GJ tonCO2−1 and a high CO2 purity of ≥98% from a CO2-loaded AAS solution.52 It was found that reducing the thickness of the acidification chamber from 5 mm to 1.5 mm led to an approximately 70% reduction in energy consumption. Recently, Zhang et al. proposed a different three-chamber electrolysis system for CO2 release from 0.5 M NaHCO3 solution.132 This system showed a very low energy consumption of 118 kJ molCO2−1 (i.e., 2.68 GJ tonCO2−1) at 100 mA cm−2 due to the use of a thinner solid electrolyte, instead of the acidification chamber. The solid electrolyte allowed the cell voltage to remain stable below 2 V when the current density was doubled from 100 to 200 mA cm−2. Without this solid interlayer, the voltage surged beyond 8 V even at current densities below 100 mA cm−2.
Xu et al. reported an alternative three-chamber electrolytic system that switches between the electrolytic cell and the fuel cell in the same middle chamber by applying voltage alternately.133 This system released CO2 from Li2CO3 solution, with an energy consumption of 6.40 GJ tonCO2−1 at a current density of 100 mA cm−2. To optimize the system, Liu et al. combined two electrolytic cells into a three-chamber structure, utilizing cyclic viologen electrocatalysis to facilitate CO2 release.134 This system could operate stably for 200 hours with a reduced energy consumption of 3.80 GJ tonCO2−1.
It is important to note the distinct structural and functional differences between the two and three-chamber electrolytic cells. In the first configuration, the anode and cathode are physically separated by two ion-exchange membranes with a central chamber between them, allowing the chambers to operate independently (Fig. 12c).52,132 This configuration is well-suited for continuous-flow systems, offering benefits in operational stability and simplicity. In contrast, the second design employs current switching to control all chambers, enabling operational transitions between electrolysis and fuel cell modes (Fig. 12d).133,134 This approach necessitates a more complex voltage control system, and its practical stability remains unclear and requires further validation.
Quinones are the most widely studied compounds for electrochemical CO2 capture and release. The mechanism of quinone in CO2 capture and release is shown in Fig. 13a. Generally, quinone undergoes a proton coupled electron transfer reaction in a neutral aprotic electrolyte. At reduction potentials, quinone is reduced to semiquinone and radical anions. As the reduction potential becomes more negative, the radical anions are reduced further to form hydroquinone and divalent radical anions, which are capable of binding CO2. In contrast, during the oxidation, CO2-loaded quinones release CO2 to regenerate hydroquinone and divalent radical anions. The capture and release of CO2 occur upon reduction and oxidation, respectively, thereby constituting a complete electrochemical carbon capture cycle.
In a protic medium (e.g., water), the formation of hydroquinone becomes more favorable than CO2 binding, making CO2 capture inefficient. To address this issue, Liu et al. proposed to use salt-concentrated aqueous media.135 In this electrolyte, the mass and volume of dissolved salts exceed those of water, resulting in the absence of free water, which effectively suppresses water molecule activity and extends the electrochemical stability window. Compared to conventional diluted quinones, this approach demonstrates enhanced thermodynamic reactivity toward CO2 in salt-concentrated electrolytes, achieving a high CO2 capture uptake (8.9 mmol g−1) and low regeneration energy consumption of 56 kJ molCO2−1 (i.e., 1.27 GJ tonCO2−1). Voskian and co-workers demonstrated that quinones can also be made into electrode materials for DAC.136 The adsorbent consisting of polyanthraquinone and carbon nanotubes captures CO2 during charging while releasing it during discharging. A preliminary economic analysis revealed the high potential of this system, with costs ranging from $50–100 per tonCO2−1.
Phenazine is an alternative redox mediator for CO2 capture and release. During reduction (Fig. 13b), phenazine is hydrogenated by protons from water, producing OH− that captures CO2, forming HCO3− and CO32−. In the oxidation process, the hydrogenated phenazine is oxidized to produce H+, which then reacts with HCO3− and CO32− to release gaseous CO2 and regenerate phenazine. Xie et al. developed a CO2 capture/release cell utilizing a sulfonated phenazine derivative, which changed pH in aqueous NaHCO3/Na2CO3 electrolyte to facilitate the release of CO2 and the regeneration of the absorbent solution.137 The system showed a faradaic efficiency of 95.8% at 10 mA cm−2, with an electrolytic energy consumption as low as 21.6 kJ molCO2−1 (i.e., 0.49 GJ tonCO2−1). Pang et al. reported an electrochemical cell based on 2,2′-(phenazine-1,8-diyl)bis(ethane-1-sulfonate) for CO2 capture and release, achieving an energy consumption of 36–55 kJ molCO2−1 (i.e., 0.82–1.25 GJ tonCO2−1).138 Notably, this system exhibits excellent charge and discharge capabilities, with a remarkably low degradation rate (less than 0.01% per day), enabling flexible adjustment of the capture and release time in response to fluctuations in the electricity market. In principle, the phenazine mediated electrolysis for CO2 capture/release is similar to the pH swing, but its energy consumption is significantly reduced as a result of the phenazine mediation.
We make a table to have a better comparison of the presented methods for CO2 release and capture agent regeneration (Table 3). As a mature technology validated in large-scale DAC applications, TSA/TVSA can operate under mild conditions (∼100 °C), but it still features moderate to high energy consumption, which is primarily attributed to the thermal energy required for desorption. As an emerging alternative, e-TSA/e-TVSA shows great promise for adsorbent regeneration due to its high efficiency and rapid response, yet it currently exhibits relatively high overall energy consumption (6.10–11.40 GJ tonCO2−1, see the entries for temperature swing in Table 3). Another alternative is solar-driven TSA, which can directly utilize renewable solar energy. However, its operational stability is highly dependent on weather conditions, posing a major challenge for maintaining continuous and stable performance at scale.
| Methods | Specific types | Capture agent | Working temperature (°C) | Energy consumption (GJ tonCO2−1) | Ref. |
|---|---|---|---|---|---|
| a A commercial amine-functionalized weakly basic anion exchange resin.b Using a simulated CO2-captured solution (e.g., NaHCO3/Na2CO3).c Room temperature.d The differences in electrolytic cell configuration,e The middle acidification chamber thickness, andf The reaction type lead to the variations in energy consumption. | |||||
| Temperature swing | TSA/TVSA | Amine loaded adsorbents | 80–100 | 5.40–7.20 | 109 |
| e-TSA/TVSA | PEI loaded silica | 80–120 | 7.20 | 114 | |
| Hydroxide-functionalized porous carbon | 90–100 | 6.50–11.40 | 87 | ||
| EDA-Y zeolite/cellulose acetate fiber sorbent | 110 | 6.10 | 115 | ||
| Solar-driven TSA | Lewatit VP OC 1065 (Lanxess)a | 110 | No data available | 116 | |
| PEI-loaded SBA-15 | 80–100 | No data available | 117 | ||
| Precipitation-phase separation | Potassium–calcium loop | KOH/K2CO3 | 900 | 5.25–8.81 | 39 |
| Amino acid salt-guanidine loop | K-Gly/K-Sar | 80–120 | No data available | 122 | |
| BTIG | 60–150 | 2.27 | 124 | ||
| Electrolysis | pH swing with BPMED | K2CO3/KHCO3b | RTc | 2.27–4.54d | 127 |
| K2CO3/KHCO3b | RT | 8.80d | 128 | ||
| pH swing with three-chamber reactors | Na2CO3/NaHCO3b | RT | 8.49 | 130 | |
| NaOH | RT | 12.19 ± 0.75e | 131 | ||
| NaOH | RT | 2.68e | 132 | ||
| Na-Gly/Gly/NaCl | RT | 5.20–8.08 | 52 | ||
| LiOH | RT | 6.40f | 133 | ||
| LiOH | RT | 3.80f | 134 | ||
| Redox mediation by quinones | 1,4-Napthoquinone and 1-ethyl-3-methylimidazolium tricyanomethanide | RT | 0.91–2.04 | 136 | |
| Hydroquinone/quinone | RT | 1.27 | 135 | ||
| Anthraquinone | RT | 2.57 | 139 | ||
| Poly(vinylanthraquinone) | RT | 0.48 | 140 | ||
| 2,3,5,6-Tetrachloro-p-benzoquinone | RT | 1.09–3.18 | 141 | ||
| Redox mediation by phenazines | Na2CO3/7,8-dihydroxyphenazine-2-sulfonic acid | RT | 0.49 | 137 | |
| 1,8-ESP | RT | 0.82–1.25 | 138 | ||
| Cyclic poly(phenazine sulfide) | RT | 2.36 | 142 | ||
The precipitation-phase separation method allows the transformation of captured CO2 in the solution into a solid form, thereby avoiding the treatment of large volumes of absorbent solutions. This makes the method particularly suitable for large-scale DAC systems. The separated CO2 capture and release units can also provide greater flexibility and controllability, enabling the accumulation of sufficient quantities of carbonate solid from multiple capture cycles before heating treatment. The precipitation-phase separation method through the calcium loop has a relatively high energy consumption due to high-temperature calcination (see the entries for precipitation-phase separation in Table 3). In comparison, the CO2 release through the guanidine loop opens up new avenues for reducing the heating temperature required (60–150 °C), significantly reducing the energy consumption (2.27 GJ tonCO2−1) and also making solar heating a viable option. However, while specific guanidine compounds have shown good performance in previous studies, their potential for scaled-up synthesis remains uncertain.
The traditional electrochemical pH swing usually results in the generation of byproducts such as O2, which necessitates an additional separation step to obtain pure CO2. BPMED and the three-chamber electrolytic system offer alternative approaches for releasing CO2 with high purity and moderate to high energy consumption (2.68–12.19 GJ tonCO2−1, see the entries for electrolysis in Table 3). The thickness of the central acidification chamber dictates the electrode spacing, while the electrolyte ion concentration governs the system's conductivity. These two parameters are key factors leading to the variations in energy consumption reported across different studies. Replacing the central acidification chamber with the solid electrolyte layer can increase the current density significantly without increasing the cell voltage, representing a promising direction for electrolytic CO2 release.
Using redox mediators for electrolysis exhibits the lowest energy consumption across all systems, making it a highly promising approach (0.48–3.18 GJ tonCO2−1, see the entries for electrolysis–redox mediation in Table 3). The energy consumption of the redox-mediated approach has reached close to the theoretical minimum for CO2 release (0.43 GJ tonCO2−1).143 However, the complex synthesis process for the organic mediators remains the primary limitation for its scale-up.
5 Summary and outlook
5.1 Summary
DAC has shown great potential in promoting carbon recycling and achieving carbon neutrality, garnering increasing attention in recent years. In this work, we discuss three key factors influencing its efficiency and development, which are summarized in Table 4.| Key factors | Types | Key benefits | Key challenges |
|---|---|---|---|
| Air contactor | Adsorption-based | Compact and small-size devices | Operational complexity from cyclic switching |
| CO2 capture and release on one site | |||
| Modular scale-out | |||
| Absorption-based | Modular scale-out & centralized scale-up | Water evaporation | |
| Compatible with various CO2 release technologies | Corrosion risk of facility | ||
| Capture agent | Solid adsorbents | Capable of modularization | Competing with H2O adsorption |
| Wide adaptability to environmental temperature | Poor stability | ||
| Liquid absorbents | Relatively low cost and easy to scale up | Corrosion to the air contactor/pipeline | |
| High CO2 selectivity | Water evaporation | ||
| CO2 release/capture agent regeneration | Temperature swing | Waste heat can be utilized | Low thermal energy efficiency |
| Degradation of capture agent | |||
| Precipitation-phase separation | Treating a small amount of carbonate solid | Involving multiple steps | |
| High operating temperature | |||
| Electrolysis | Compact equipment | Relying on precious electrocatalysts, mediators, and ion-exchange membranes | |
| Controllable reaction rate | |||
| Coupling with renewable energy |
(1) The air contactor is an indispensable component of the DAC system which guarantees air flow and CO2 supply. The adsorption-based air contactor allows for CO2 capture and release in a compact design. However, this integrated functionality requires a more complex piping and process design for the DAC system. Thus, the system can achieve large CO2 capacities primarily through modular scale-out (i.e., running many modular units in parallel). In contrast, the absorption-based air contactor operates independently from the CO2 release unit and shows high compatibility with a wide range of CO2 release technologies. Accordingly, it offers greater flexibility for scaling, via both modular scale-out and centralized scale-up (i.e., expanding a single plant). For example, Carbon Engineering is leading the front-end engineering and design for a DAC facility with a CO2 capture capacity of 500![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons per year.144 However, it is important to note that the absorption-based air contactor usually utilizes a strongly alkaline absorbent solution, which requires careful consideration of water evaporation and corrosion risk to the contactor and piping framework, as well as the potential for precipitate formation.
000 tons per year.144 However, it is important to note that the absorption-based air contactor usually utilizes a strongly alkaline absorbent solution, which requires careful consideration of water evaporation and corrosion risk to the contactor and piping framework, as well as the potential for precipitate formation.
(2) The capture agent determines the CO2 capture capacity. There are two typical types of capture agents, including solid adsorbents and liquid absorbents, which are used in adsorption- and absorption-based air contactors, respectively. Amine-functionalized porous materials are the most important solid adsorbents, and their performance for DAC is influenced by a number of factors such as the type of amine, amine loading, support material, and environmental temperature and humidity. However, the high production cost and limited lifespan of adsorbent materials hinder their widespread use in DAC. Alkaline hydroxide and amine solutions are both efficient and cost-effective absorbents for CO2 capture. Alkaline hydroxides have found practical applications in the absorption-based DAC facility. However, the use of amine compounds for DAC is limited, because of the volatility and degradation issues. Thus, the stability of organic amines under atmospheric conditions requires further improvement, for example, by developing new amine compounds with low volatility and resistant to oxidative degradation. AAS, as easily accessible and environmentally friendly absorbents with rapid reaction kinetics for CO2 capture, hold significant potential for DAC.
(3) The process of CO2 release and capture agent regeneration contributes to most energy consumption in DAC. There are several types of effective methods to release the captured CO2. Temperature swing, which usually operates at elevated temperatures of 80–120 °C and under vacuum conditions, is primarily used to release CO2 from adsorption-based DAC systems. This technology enables the utilization of waste heat, offering potential sustainability benefits.47 However, the degradation of capture agents at high regeneration temperature leads to decreased CO2 capture performance. The precipitation-phase separation method separates CO2 capture and release steps. This method converts captured CO2 in liquid solution into a solid precipitate, significantly reducing the volume/mass of substance for processing. However, the high calcination temperature required for CO2 release from CaCO3 remains the primary limitation of this method. Additionally, the multiple processing steps lead to operational complexity. The electrochemical approach for CO2 release provides distinct advantages including (i) mild reaction conditions, (ii) electrochemically tunable reaction rates, (iii) compatibility with renewable electricity, and (iv) low energy consumption when mediated by redox couples. To date, these developments have been primarily limited to lab-scale prototypes, lacking industrial-scale implementation cases. Scaling it up remains challenging, as it depends on stable and costly electrocatalysts and ion exchange membranes to achieve high efficiency and durability.
Overall, existing research has largely focused on the development and optimization of individual DAC components, such as the air contactor, capture agent, or regeneration unit, under idealized conditions. In practice, however, these three components form a tightly connected system, where the choice of air contactors determines the choice of capture agents, which in turn dictates the feasible regeneration approaches. The lack of a system-level study on the entire DAC process poses a major obstacle for achieving efficient and large-scale DAC implementation.
5.2 Outlook
Despite the challenges and issues mentioned above, the DAC technology is highly promising for its great prospect of removing CO2 from the atmosphere directly. To overcome the issues and promote the development and practical applications of DAC technology, research efforts may be directed to the following aspects.Notably, machine learning, leveraging datasets of atomic and molecular properties, is becoming a powerful tool for CO2 capture studies.152,153 While numerous efficient ad/absorbents have already been identified, future efforts can focus on constructing comprehensive databases and employing text mining to extract insights from previous reports. Building on this, developing robust machine learning algorithms will be crucial for guiding the rational design of new capture agents. The ultimate goal is to discover new capture agents that deliver both high CO2 uptake and long-term stability across a range of contactor configurations and operating environments.
Looking forward, future research should shift from studying individual components toward a system-level paradigm. The air contactor, capture agent, and regeneration method are not independent but tightly connected components, whose collective performance determines overall DAC efficiency. For instance, a highly efficient sorbent cannot realize its potential unless paired with an air contactor and a regeneration process enabling effective mass and energy transfer. Research efforts on linking the three components as a single system are expected to unlock synergistic gains in enhancing DAC efficiency for practical applications.
Author contributions
All authors contributed to the manuscript's writing, reviewing, and revising. X.-M. Hu coordinated and supervised the overall project.Conflicts of interest
The authors declare no conflict of interest.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was financially supported by National Natural Science Foundation of China (22376120, 22509114), Taishan Scholars program from Shandong Province (tsqn202103021, tsqn202507032), and Shandong Provincial Natural Science Foundation (ZR2025QC1317).References
- National Oceanic & Atmospheric Administration, Global Monitoring Laboratory: Trends in atmospheric carbon dioxide, http://gml.noaa.gov/ccgg/trends, (accessed October 2025).
- J. Rogelj, M. den Elzen, N. Hoehne, T. Fransen, H. Fekete, H. Winkler, R. S. Chaeffer, F. Ha, K. Riahi and M. Meinshausen, Paris Agreement climate proposals need a boost to keep warming well below 2 °C, Nature, 2016, 534, 631–639 CrossRef CAS PubMed.
- Z. Liu, Z. Deng, G. He, H. Wang, X. Zhang, J. Lin, Y. Qi and X. Liang, Challenges and opportunities for carbon neutrality in China, Nat. Rev. Earth Environ., 2022, 3, 141–155 CrossRef.
- Z. Wu, X. Huang, R. Chen, X. Mao and X. Qi, The United States and China on the paths and policies to carbon neutrality, J. Environ. Manage., 2022, 320, 115785 CrossRef CAS PubMed.
- A. Ozawa, T. Tsani and Y. Kudoh, Japan's pathways to achieve carbon neutrality by 2050 – Scenario analysis using an energy modeling methodology, Renewable Sustainable Energy Rev., 2022, 169, 112943 CrossRef.
- J. Krause, M. Yugo, Z. Samaras, S. Edwards, G. Fontaras, R. Dauphin, P. Prenninger and S. Neugebauer, Well-to-wheels scenarios for 2050 carbon-neutral road transport in the EU, J. Cleaner Prod., 2024, 443, 141084 CrossRef.
- F. Zhang, K. S. Gallagher, M. Deng, H. Liu, R. Orvis and X. Xuan, Assessing the policy gaps for achieving China's carbon neutrality target, Environ. Sci. Technol., 2025, 59, 18124–18133 CrossRef CAS PubMed.
- W. Gao, S. Liang, R. Wang, Q. Jiang, Y. Zhang, Q. Zheng, B. Xie, C. Y. Toe, X. Zhu, J. Wang, L. Huang, Y. Gao, Z. Wang, C. Jo, Q. Wang, L. Wang, Y. Liu, B. Louis, J. Scott, A.-C. Roger, R. Amal, H. He and S.-E. Park, Industrial carbon dioxide capture and utilization: State of the art and future challenges, Chem. Soc. Rev., 2020, 49, 8584–8686 RSC.
- B. Liang, C. Chen, C. Jia, C. Wang, X. Wang, Y. Zha, R. Wang, Z. Meng and H. Wang, Carbon capture, utilization and storage (CCUS) in oil and gas reservoirs in China: Status, opportunities and challenges, Fuel, 2024, 375, 132353 CrossRef CAS.
- S. Cobo, A. Galan-Martin, V. Tulus, M. A. J. Huijbregts and G. Guillen-Gosalbez, Human and planetary health implications of negative emissions technologies, Nat. Commun., 2022, 13, 2535 CrossRef CAS PubMed.
- Z. Deng, B. Zhu, S. J. Davis, P. Ciais, D. Guan, P. Gong and Z. Liu, Global carbon emissions and decarbonization in 2024, Nat. Rev. Earth Environ., 2025, 6, 231–233 CrossRef.
- H. D. Matthews and S. Wynes, Current global efforts are insufficient to limit warming to 1.5°C, Science, 2022, 376, 1404–1409 CrossRef CAS PubMed.
- T. M. Gür, Carbon dioxide emissions, capture, storage and utilization: Review of materials, processes and technologies, Prog. Energy Combust. Sci., 2022, 89, 100965 CrossRef.
- G. J. Fulham, P. V. Mendoza-Moreno and E. J. Marek, Managing intermittency of renewable power in sustainable production of methanol, coupled with direct air capture, Energy Environ. Sci., 2024, 17, 4594–4621 RSC.
- D. U. Nielsen, X.-M. Hu, K. Daasbjerg and T. Skrydstrup, Chemically and electrochemically catalysed conversion of CO2 to CO with follow-up utilization to value-added chemicals, Nat. Catal., 2018, 1, 244–254 CrossRef CAS.
- S. Li, S. Zhao, X. Lu, M. Ceccato, X.-M. Hu, A. Roldan, J. Catalano, M. Liu, T. Skrydstrup and K. Daasbjerg, Low-valence Znδ+ (0<δ<2) single-atom material as highly efficient electrocatalyst for CO2 reduction, Angew. Chem., Int. Ed., 2021, 60, 22826–22832 CrossRef CAS PubMed.
- B. Lux, N. Schneck, B. Pfluger, W. Männer and F. Sensfuß, Potentials of direct air capture and storage in a greenhouse gas-neutral European energy system, Energy Strategy Rev., 2023, 45, 101012 CrossRef.
- Z. Yang, R. Chen, L. Zhang, Y. Li and C. Li, Recent progress in nickel single-atom catalysts for the electroreduction of CO2 to CO, Ind. Chem. Mater., 2024, 2, 533–555 RSC.
- Y. Ma, T. Xiao, K. Zhu, D. Zhao and W. Li, Cu nanoparticles embedded in fluorinated mesoporous carbon for enhanced CO2 electroreduction to C2 products, Carbon Future, 2025, 2, 9200034 CrossRef.
- International Energy Agency, CCUS Projects Database: Covers all large-scale CO2 capture, transport, storage, and utilisation projects commissioned or in planning worldwide, https://www.iea.org/data-and-statistics/data-product/ccus-projects-database, (accessed October 2025).
- A. Kasturi, G. Gug Jang, A. Dona-Tella Akin, A. Jackson, J. Jun, D. Stamberga, R. Custelcean, D. S. Sholl, S. Yiacoumi and C. Tsouris, An effective air–liquid contactor for CO2 direct air capture using aqueous solvents, Sep. Purif. Technol., 2023, 324, 124398 CrossRef CAS.
- P. Zhu, Z.-Y. Wu, A. Elgazzar, C. Dong, T.-U. Wi, F.-Y. Chen, Y. Xia, Y. Feng, M. Shakouri, J. Y. Kim, Z. Fang, T. A. Hatton and H. Wang, Continuous carbon capture in an electrochemical solid-electrolyte reactor, Nature, 2023, 618, 959–966 CrossRef CAS.
- S. Bose, D. Sengupta, T. M. Rayder, X. Wang, K. O. Kirlikovali, A. K. Sekizkardes, T. Islamoglu and O. K. Farha, Challenges and opportunities: Metal-organic frameworks for direct air capture, Adv. Funct. Mater., 2024, 34, 2307478 CrossRef CAS.
- H. Akaya, S. Lamnini, H. Sehaqui and J. Jacquemin, Amine-functionalized cellulose as promising materials for direct CO2 capture: A review, ACS Appl. Mater. Interfaces, 2025, 17, 16380–16395 CrossRef CAS PubMed.
- H. E. Holmes, M. J. Realff and R. P. Lively, Water management and heat integration in direct air capture systems, Nat. Chem. Eng., 2024, 1, 208–215 CrossRef CAS.
- S. Li, Y. Feng, Y. Li, S. Deng, X. E. Cao, K. B. Lee and J. Wang, Critical review on mobile direct air capture: Concept expansion, characteristic description, and performance evaluation, Matter, 2024, 7, 889–933 CrossRef CAS.
- H. Liu, B. Liu, L.-C. Lin, G. Chen, Y. Wu, J. Wang, X. Gao, Y. Lv, Y. Pan, X. Zhang, X. Zhang, L. Yang, C. Sun, B. Smit and W. Wang, A hybrid absorption–adsorption method to efficiently capture carbon, Nat. Commun., 2014, 5, 5147 CrossRef CAS PubMed.
- Official website for Climeworks, http://www.climeworks.com, (accessed February 2026).
- Official website for Heirloomcarbon, http://www.heirloomcarbon.com, (accessed February 2026).
- Official website for Carboncapture Inc, https://www.carboncapture.com/, (accessed February 2026).
- Climeworks, Climeworks begins operations of Orca, the world's largest direct air capture and CO2 storage plant, https://climeworks.com/press-release/climeworks-launches-orca, (accessed October 2025).
- Climeworks, Climeworks switches on world's largest direct air capture plant, https://climeworks.com/press-release/climeworks-switches-on-worlds-largest-direct-air-capture-plant-mammoth, (accessed October 2025).
- J. Chladek, C. K. Jayarathna, B. M. E. Moldestad and L.-A. Tokheim, Fluidized bed classification of particles of different size and density, Chem. Eng. Sci., 2018, 177, 151–162 CrossRef CAS.
- M.-Y. Low, L. V. Barton, R. Pini and C. Petit, Analytical review of the current state of knowledge of adsorption materials and processes for direct air capture, Chem. Eng. Res. Des., 2023, 189, 745–767 CrossRef CAS.
- J. Wu, Y. Chen, K. Wang, Y. Huo, Y. Chen, Q. Pan and T. Ge, Rapid steam-assisted temperature swing adsorption for direct air capture using a rotary adsorber, Adv. Sci., 2026, e21499 CrossRef PubMed.
- E. Tegeler, Y. Cui, M. Masoudi, A. M. Bahmanpour, T. Colbert, J. Hensel and V. Balakotaiah, A novel contactor for reducing the cost of direct air capture of CO2, Chem. Eng. Sci., 2023, 281, 119107 CrossRef CAS.
- Q. Grossmann, V. Stampi-Bombelli, A. Yakimov, S. Docherty, C. Copéret and M. Mazzotti, Developing versatile contactors for direct air capture of CO2 through amine grafting onto alumina pellets and alumina wash-coated monoliths, Ind. Eng. Chem. Res., 2023, 62, 13594–13611 CrossRef CAS PubMed.
- F. Sabatino, A. Grimm, F. Gallucci, M. van Sint Annaland, G. J. Kramer and M. J. J. Gazzani, A comparative energy and costs assessment and optimization for direct air capture technologies, Joule, 2021, 5, 2047–2076 CrossRef CAS.
- D. W. Keith, G. Holmes, D. S. Angelo and K. Heidel, A process for capturing CO2 from the atmosphere, Joule, 2018, 2, 1573–1594 CrossRef CAS.
- Carbon engineering, Carbon Engineering to invest in technology development and research facilities in Squamish, B.C, https://carbonengineering.com/news-updates/carbon-engineering-to-invest-in-technology-development-and-research-facilities/, (accessed October 2025).
- A. Momeni, R. V. McQuillan, M. S. Alivand, A. Zavabeti, G. W. Stevens and K. A. Mumford, Direct air capture of CO2 using green amino acid salts, Chem. Eng. J., 2024, 480, 147934 CrossRef CAS.
- N. C. Ellebracht, P. Roy, T. Moore, A. E. Gongora, D. I. Oyarzun, J. K. Stolaroff and D. T. Nguyen, 3D printed triply periodic minimal surfaces as advanced structured packings for solvent-based CO2 capture, Energy Environ. Sci., 2023, 16, 1752–1762 RSC.
- K. An, K. Li, C.-M. Yang, J. Brechtl, D. Stamberga, M. Zhang and K. Nawaz, Direct air capture with amino acid solvent: Operational optimization using a crossflow air-liquid contactor, AIChE J., 2024, 70, e18429 CrossRef CAS.
- F. Zhao, C. Cui, S. Dong, X. Xu and H. Liu, An overview on the corrosion mechanisms and inhibition techniques for amine-based post-combustion carbon capture process, Sep. Purif. Technol., 2023, 304, 122091 CrossRef CAS.
- Y. Zhou, F. Xie, D. Wang, Y. Wang and M. Wu, Carbon capture, utilization and storage (CCUS) pipeline steel corrosion failure analysis: A review, Eng. Failure Anal., 2024, 155, 107745 CrossRef.
- K. An, A. Farooqui and S. T. McCoy, The impact of climate on solvent-based direct air capture systems, Appl. Energy, 2022, 325, 119895 CrossRef CAS.
- S. Deutz and A. Bardow, Life-cycle assessment of an industrial direct air capture process based on temperature-vacuum swing adsorption, Nat. Energy, 2021, 6, 203–213 CrossRef CAS.
- K. S. Lackner and H. Azarabadi, Buying down the cost of direct air capture, Ind. Eng. Chem. Res., 2021, 60, 8196–8208 CrossRef CAS.
- M. Erans, E. S. Sanz-Pérez, D. P. Hanak, Z. Clulow, D. M. Reiner and G. A. Mutch, Direct air capture: Process technology, techno-economic and socio-political challenges, Energy Environ. Sci., 2022, 15, 1360–1405 RSC.
- J. Carlos Abanades, Y. A. Criado and J. Ramon Fernandez, An air CO2 capture system based on the passive carbonation of large Ca(OH)2 structures, Sustainable Energy Fuels, 2020, 4, 3409–3417 RSC.
- E. Bachman, A. Tavasoli, T. A. Hatton, C. T. Maravelias, E. Haites, P. Styring, A. Aspuru-Guzik, J. MacIntosh and G. Ozin, Rail-based direct air carbon capture, Joule, 2022, 6, 1368–1381 CrossRef CAS.
- A.-C. Zheng, Y.-B. Zou, L. Du, Q. Zhang, K. Daasbjerg and X.-M. Hu, Efficient direct air capture in industrial cooling towers mediated by electrochemical CO2 release, Angew. Chem., Int. Ed., 2025, 64, e202412697 CrossRef CAS PubMed.
- C. Y. Chuah, Y. L. Ho, A. M. H. Syed, K. G. K. Thivyalakshmi, E. Yang, K. Johari, Y. Yang and W. C. Poon, Applicability of adsorbents in direct air capture (DAC): Recent progress and future perspectives, Ind. Eng. Chem. Res., 2025, 64, 4117–4147 CrossRef CAS.
- R.-S. Liu, X.-D. Shi, C.-T. Wang, Y.-Z. Gao, S. Xu, G.-P. Hao, S. Chen and A.-H. Lu, Advances in post-combustion CO2 capture by physical adsorption: From materials innovation to separation practice, ChemSusChem, 2021, 14, 1428–1471 CrossRef CAS PubMed.
- P. Hu, R. Oishi, H. Ya, Y. Yonezawa, M. Matsukura, K. Iyoki, T. Okubo and T. Wakihara, Development of zeolite adsorbent with low water sensitivity for CO2 capture, Chem. Eng. J., 2025, 508, 161054 CrossRef CAS.
- L. Huang, J. Ma, F. Wang, G. Xu and H. Zhao, Design of alkali metal oxide adsorbent for direct air capture: Identification of physicochemical adsorption and analysis of regeneration mechanism, Carbon Capture Sci. Technol., 2024, 13, 100268 CAS.
- R. Prins, On the structure of γ-Al2O3, J. Catal., 2020, 392, 336–346 CrossRef CAS.
- O. I.-F. Chen, C.-H. Liu, K. Wang, E. Borrego-Marin, H. Li, A. H. Alawadhi, J. A. R. Navarro and O. M. Yaghi, Water-enhanced direct air capture of carbon dioxide in metal–organic frameworks, J. Am. Chem. Soc., 2024, 146, 2835–2844 CrossRef CAS PubMed.
- H. Lyu, H. Li, N. Hanikel, K. Wang and O. M. Yaghi, Covalent organic frameworks for carbon dioxide capture from air, J. Am. Chem. Soc., 2022, 144, 12989–12995 CrossRef CAS PubMed.
- Z. He, Y. Wang, Y. Miao, H. Wang, X. Zhu and J. Li, Mixed polyamines promotes CO2 adsorption from air, J. Environ. Chem. Eng., 2022, 10, 107239 CrossRef CAS.
- J. Hua, X. Shen, X. Jiao, H. Lin, G. Li, X. Sun, F. Yan, H. Wu and Z. Zhang, Direct air capture of CO2 by amine-impregnated resin: The effect of resin pore structure and humid conditions, Carbon Capture Sci. Technol., 2024, 12, 100237 CAS.
- X. Jia, Z. Wu, H. Shi, Y. Fan, R. Zheng and C. Wang, Direct air CO2 capture using coal fly ash derived SBA-15 supported polyethylenimine, Carbon Capture Sci. Technol., 2024, 10, 100167 CAS.
- P. Priyadarshini, G. Rim, C. Rosu, M. Song and C. W. Jones, Direct air capture of CO2 using amine/alumina sorbents at cold temperature, ACS Environ. Au, 2023, 3, 295–307 CrossRef CAS PubMed.
- G. Rim, F. Kong, M. Song, C. Rosu, P. Priyadarshini, R. P. Lively and C. W. Jones, Sub-ambient temperature direct air capture of CO2 using amine-impregnated MIL-101(Cr) enables ambient temperature CO2 recovery, JACS Au, 2022, 2, 380–393 CrossRef CAS PubMed.
- Q. Shao, Z. Gan, B. Ge, X. Liu, C. Chen, D. O'Hare and X. Zhu, 3D printing of poly(ethyleneimine)-functionalized Mg-Al mixed metal oxide monoliths for direct air capture of CO2, J. Energy Chem., 2024, 96, 491–500 CrossRef CAS.
- J. Wu, Y. Chen, Y. Xu, S. Chen, H. Lv, Z. Gan, X. Zhu, R. Wang, C.-H. Wang and T. Ge, Facile synthesis of structured adsorbent with enhanced hydrophobicity and low energy consumption for CO2 capture from the air, Matter, 2024, 7, 123–139 CrossRef CAS.
- M. Yang, S. Wang and L. Xu, Hydrophobic functionalized amine-impregnated resin for CO2 capture in humid air, Sep. Purif. Technol., 2023, 315, 123606 CrossRef CAS.
- Y. Miao, Y. Wang, B. Ge, Z. He, X. Zhu, J. Li, S. Liu and L. Yu, Mixed diethanolamine and polyethyleneimine with enhanced CO2 capture cpacity from air, Adv. Sci., 2023, 10, 2207253 CrossRef CAS PubMed.
- G. Rim, M. Song, L. Proaño, O. Ghaffari Nik, S. Parker, R. P. Lively and C. W. Jones, Humidity effects on sub-ambient direct air capture of CO2 with amine functionalized Mg-Al LDHs and MMOs, ACS ES T Eng., 2025, 5, 204–214 CrossRef CAS.
- V. Kulkarni, D. Panda and S. K. Singh, Direct air capture of CO2 over amine-modified hierarchical silica, Ind. Eng. Chem. Res., 2023, 62, 3800–3811 CrossRef CAS.
- M. S. Alivand, U. Habiba, M. Ghasemian, S. Askari and P. A. Webley, Amine-functionalized meso-macroporous polymers for efficient CO2 capture from ambient air, ACS Appl. Mater. Interfaces, 2024, 16, 17411–17421 CrossRef PubMed.
- Y. Wang, G. Rim, M. Song, H. E. Holmes, C. W. Jones and R. P. Lively, Cold temperature direct air CO2 capture with amine-loaded metal–organic framework monoliths, ACS Appl. Mater. Interfaces, 2024, 16, 1404–1415 CrossRef CAS PubMed.
- F. Liu, T. Wang, H. Dong and W. Liu, Modified metal–organic framework by a novel coordinatively unsaturated amine grafting mechanism for direct air capture of CO2, Chem. Eng. J., 2023, 454, 140431 CrossRef CAS.
- J.-T. Anyanwu, Y. Wang and R. T. Yang, CO2 capture (including direct air capture) and natural gas desulfurization of amine-grafted hierarchical bimodal silica, Chem. Eng. J., 2022, 427, 131561 CrossRef CAS.
- A. A. Al-Absi, M. Mohamedali, A. Domin, A. M. Benneker and N. Mahinpey, Development of in situ polymerized amines into mesoporous silica for direct air CO2 capture, Chem. Eng. J., 2022, 447, 137465 CrossRef CAS.
- H. Li, Z. Zhou, T. Ma, K. Wang, H. Zhang, A. H. Alawadhi and O. M. Yaghi, Bonding of polyethylenimine in covalent organic frameworks for CO2 capture from air, J. Am. Chem. Soc., 2024, 146, 35486–35492 CrossRef CAS PubMed.
- Z. Zhou, T. Ma, H. Zhang, S. Chheda, H. Li, K. Wang, S. Ehrling, R. Giovine, C. Li, A. H. Alawadhi, M. M. Abduljawad, M. O. Alawad, L. Gagliardi, J. Sauer and O. M. Yaghi, Carbon dioxide capture from open air using covalent organic frameworks, Nature, 2024, 635, 96–101 CrossRef CAS PubMed.
- K. Li, J. Jiang, F. Yan, S. Tian and X. Chen, The influence of polyethyleneimine type and molecular weight on the CO2 capture performance of PEI-nano silica adsorbents, Appl. Energy, 2014, 136, 750–755 CrossRef CAS.
- S. H. Pang, L.-C. Lee, M. A. Sakwa-Novak, R. P. Lively and C. W. Jones, Design of aminopolymer structure to enhance performance and stability of CO2 sorbents: Poly(propylenimine) vs poly(ethylenimine), J. Am. Chem. Soc., 2017, 139, 3627–3630 CrossRef CAS PubMed.
- R. A. Yang, D. R. Ganza, M. R. Smith, S. Cho, J. A. Vandermel, E. Jiang and M. L. Sarazen, Investigation of storage environments on aminopolymer stabilization within UiO-67(Zr) for CO2 capture, Mater. Adv., 2023, 4, 901–909 RSC.
- C. Rosu, S. H. Pang, A. R. Sujan, M. A. Sakwa-Novak, E. W. Ping and C. W. Jones, Effect of extended aging and oxidation on linear poly(propylenimine)-mesoporous silica composites for CO2 capture from simulated air and flue gas streams, ACS Appl. Mater. Interfaces, 2020, 12, 38085–38097 CrossRef CAS PubMed.
- M. L. Sarazen, M. A. Sakwa-Novak, E. W. Ping and C. W. Jones, Effect of different acid initiators on branched poly(propylenimine) synthesis and CO2 sorption performance, ACS Sustainable Chem. Eng., 2019, 7, 7338–7345 CrossRef CAS.
- R. Surkatti, Y. M. Abdullatif, R. Muhammad, A. Sodiq, K. Mroue, T. Al-Ansari and A. I. Amhamed, Comparative analysis of amine-functionalized silica for direct air capture (DAC): Material characterization, performance, and thermodynamic efficiency, Sep. Purif. Technol., 2025, 354, 128641 CrossRef CAS.
- G. Rim, P. Priyadarshini, M. Song, Y. Wang, A. Bai, M. J. Realff, R. P. Lively and C. W. Jones, Support pore structure and composition strongly influence the direct air capture of CO2 on supported amines, J. Am. Chem. Soc., 2023, 145, 7190–7204 CrossRef CAS PubMed.
- G. A. Russell-Parks, N. Leick, M. A. T. Marple, N. A. Strange, B. G. Trewyn, S. H. Pang and W. A. Braunecker, Fundamental insight into humid CO2 uptake in direct air capture nanocomposites using fluorescence and portable NMR relaxometry, J. Phys. Chem. C, 2023, 127, 15363–15374 CrossRef CAS.
- J. S. A. Carneiro, G. Innocenti, H. J. Moon, Y. Guta, L. Proaño, C. Sievers, M. A. Sakwa-Novak, E. W. Ping and C. W. Jones, Insights into the oxidative degradation mechanism of solid amine sorbents for CO2 capture from air: Roles of atmospheric water, Angew. Chem., Int. Ed., 2023, 62, e202302887 CrossRef CAS PubMed.
- H. Li, M. E. Zick, T. Trisukhon, M. Signorile, X. Liu, H. Eastmond, S. Sharma, T. L. Spreng, J. Taylor, J. W. Gittins, C. Farrow, S. A. Lim, V. Crocella, P. J. Milner and A. C. Forse, Capturing carbon dioxide from air with charged-sorbents, Nature, 2024, 630, 654–659 CrossRef CAS PubMed.
- R. Ramezani, S. Mazinani and R. Di Felice, State-of-the-art of CO2 capture with amino acid salt solutions, Rev. Chem. Eng., 2022, 38, 273–299 CrossRef CAS.
- R. Custelcean, Direct air capture of CO2 using solvents, Annu. Rev. Chem. Biomol. Eng., 2022, 13, 217–234 CrossRef CAS PubMed.
- R. Han, Y. Wang, S. Xing, C. Pang, Y. Hao, C. Song and Q. Liu, Progress in reducing calcination reaction temperature of Calcium-Looping CO2 capture technology: A critical review, Chem. Eng. J., 2022, 450, 137952 CrossRef CAS.
- A. Rouxhet, S.-m. Kim and G. Léonard, Study of direct air capture (DAC) using a KOH/K2CO3 absorbing solution for CO2 capture, Proceedings of the 16th Greenhouse Gas Control Technologies Conference (GHGT-16), 2022, pp. 23–24.
- R. Ragipani, K. Sreenivasan, R. P. Anex, H. Zhai and B. Wang, Direct air capture and sequestration of CO2 by accelerated indirect aqueous mineral carbonation under ambient conditions, ACS Sustainable Chem. Eng., 2022, 10, 7852–7861 CrossRef CAS.
- Z. Zolfaghari, A. Aslani, R. Zahedi and S. Kazzazi, Simulation of carbon dioxide direct air capture plant using potassium hydroxide aqueous solution: Energy optimization and CO2 purity enhancement, Energy Convers. Manag.: X, 2024, 21, 100489 CAS.
- F. Meng, Y. Meng, T. Ju, S. Han, L. Lin and J. Jiang, Research progress of aqueous amine solution for CO2 capture: A review, Renewable Sustainable Energy Rev., 2022, 168, 112902 CrossRef CAS.
- F. Barzagli, C. Giorgi, F. Mani and M. Peruzzini, Screening study of different amine-based solutions as sorbents for direct CO2 capture from air, ACS Sustainable Chem. Eng., 2020, 8, 14013–14021 CrossRef CAS.
- F. A. Chowdhury, H. Yamada, T. Higashii, K. Goto and M. Onoda, CO2 capture by tertiary amine absorbents: A performance comparison study, Ind. Eng. Chem. Res., 2013, 52, 8323–8331 CrossRef CAS.
- G. T. Rochelle, Air pollution impacts of amine scrubbing for CO2 capture, Carbon Capture Sci. Technol., 2024, 11, 100192 CAS.
- F. O. Ochedi, J. Andresen and M. van der Spek, Oxidative degradation of glycine in aqueous KOH/K2CO3 solutions for CO2 capture, Greenhouse Gases:Sci. Technol., 2024, 14, 1025–1036 CrossRef CAS.
- A. Kiani, W. Conway, M. H. Abdellah, G. Puxty, A.-J. Minor, G. Kluivers, R. Bennett and P. Feron, A study on degradation and CO2 capture performance of aqueous amino acid salts for direct air capture applications, Greenhouse Gases:Sci. Technol., 2024, 14, 859–870 CrossRef CAS.
- R. Custelcean, K. A. Garrabrant, P. Agullo and N. J. Williams, Direct air capture of CO2 with aqueous peptides and crystalline guanidines, Cell Rep. Phys. Sci., 2021, 2, 100385 CrossRef CAS.
- U. I. Premadasa, D. Dong, D. Stamberga, R. Custelcean, S. Roy, Y.-Z. Ma, V. Bocharova, V. S. Bryantsev and B. Doughty, Chemical feedback in the self-assembly and function of air–liquid interfaces: insight into the bottlenecks of CO2 direct air capture, ACS Appl. Mater. Interfaces, 2023, 15, 19634–19645 CrossRef CAS PubMed.
- M. Modayil Korah, S. Ly, T. Stangherlin Barbosa, R. Nile, K. Jin, K. S. Lackner and M. D. Green, Electrospun poly(vinyl alcohol)–l-arginine nanofiber composites for direct air capture of CO2, ACS ES T Eng., 2023, 3, 373–386 CrossRef CAS.
- A. Kasturi, J. Gabitto, G. Gug Jang, J. A. Thompson, D. Stamberga, J. Seo, D. S. Sholl, S. Yiacoumi, R. Custelcean and C. Tsouris, Sub-ambient performance of potassium sarcosinate for direct air capture applications: CO2 flux and viscosity measurements, Sep. Purif. Technol., 2025, 357, 130026 CrossRef CAS.
- B. K. Mondal and A. N. Samanta, Equilibrium solubility and kinetics of CO2 absorption in hexamethylenediamine activated aqueous sodium glycinate solvent, Chem. Eng. J., 2020, 386, 121462 CrossRef CAS.
- R. V. McQuillan, A. Momeni, M. S. Alivand, G. W. Stevens and K. A. Mumford, Evaluation of potassium glycinate as a green solvent for direct air capture and modelling its performance in hollow fiber membrane contactors, Chem. Eng. J., 2024, 481, 148764 CrossRef CAS.
- Y. C. Xiao, S. S. Sun, Y. Zhao, R. K. Miao, M. Fan, G. Lee, Y. Chen, C. M. Gabardo, Y. Yu, C. Qiu, Z. Guo, X. Wang, P. Papangelakis, J. E. Huang, F. Li, C. P. O'Brien, J. Kim, K. Han, P. J. Corbett, J. Y. Howe, E. H. Sargent and D. Sinton, Reactive capture of CO2 via amino acid, Nat. Commun., 2024, 15, 7849 CrossRef CAS PubMed.
- K. An, K. Li, C.-M. Yang, J. Brechtl and K. Nawaz, A comprehensive review on regeneration strategies for direct air capture, J. CO2 Util., 2023, 76, 102587 CrossRef CAS.
- W. Liu, J. K. Wu, M. Yu, X. J. Zhang, T. Wang, M. X. Fang and L. Jiang, Thermodynamic analysis of adsorption carbon capture from limiting cycle to heat pump assisted cycle, Energy, 2024, 291, 130299 CrossRef CAS.
- Climeworks, UN Climate Change Conference: Climeworks informs about negative emission technologies, https://climeworks.com/news/un-climate-change-conference-climeworks-informs-about, (accessed October 2025).
- J. Elfving, J. Kauppinen, M. Jegoroff, V. Ruuskanen, L. Jarvinen and T. Sainio, Experimental comparison of regeneration methods for CO2 concentration from air using amine-based adsorbent, Chem. Eng. J., 2021, 404, 126337 CrossRef CAS.
- Climeworks, Next generation tech powers Climeworks' megaton leap, https://climeworks.com/press-release/next-gen-tech-powers-climeworks-megaton-leap, (accessed October 2025).
- S. Zhao, Y. Zhang, Y. Ning, L. Li, J. Lu, X. Wang, F. Wang, Z. Wei, Z. Huang and H. Lin, Electrically conductive amine-functionalized adsorbents driven by joule heating for direct air capture, ACS Sustainable Chem. Eng., 2025, 13, 14949–14960 CrossRef CAS.
- K. Petzall, B. J. Rhodes, Y. Wang, T. Trisukhon, H. Kukreja, S. A. Lim, P. J. Milner and A. C. Forse, Developing joule-heating regeneration of carbon-based adsorbents for direct air capture, Chem. Eng. J., 2025, 524, 169247 CrossRef CAS.
- W. H. Lee, X. Zhang, S. Banerjee, C. W. Jones, M. J. Realff and R. P. Lively, Sorbent-coated carbon fibers for direct air capture using electrically driven temperature swing adsorption, Joule, 2023, 7, 1241–1259 CrossRef CAS.
- Y. H. Lee, J. H. Lee, H. Joo, M. Massen-Hane, I. Park, S. Rho, A. Jamal, T. Alan Hatton and D.-Y. Koh, Design of electrified fiber sorbents for direct air capture with electrically-driven temperature vacuum swing adsorption, Adv. Mater., 2025, 37, e04542 Search PubMed.
- Y. Wang, L. Qu, H. Ding, P. Webley and G. K. Li, Distributed direct air capture of carbon dioxide by synergistic water harvesting, Nat. Commun., 2024, 15, 9745 CrossRef CAS PubMed.
- S. Kar, D. Kim, A. Bin Mohamad Annuar, B. B. Sarma, M. Stanton, E. Lam, S. Bhattacharjee, S. Karak, H. F. Greer and E. Reisner, Direct air capture of CO2 for solar fuel production in flow, Nat. Energy, 2025, 10, 448–459 Search PubMed.
- A. M. Thomas Jr, Thermal decomposition of sodium carbonate solutions, J. Chem. Eng. Data, 1963, 8, 51–54 Search PubMed.
- M. Hartman, K. Svoboda, M. Pohořelý and M. Šyc, Thermal decomposition of sodium hydrogen carbonate and textural features of its calcines, Ind. Eng. Chem. Res., 2013, 52, 10619–10626 CrossRef CAS.
- R. Wang, H. Zhao, X. Yang, C. Qi, H. Zhao, S. Zhang, Q. Li, P. Li and L. Wang, Energy-efficient non-aqueous biphasic solvent for carbon capture: Absorption mechanism, phase evolution process, and non-corrosiveness, Energy, 2023, 281, 128353 CrossRef CAS.
- S. J. Vevelstad, V. Buvik, H. K. Knuutila, A. Grimstvedt and E. F. da Silva, Important aspects regarding the chemical stability of aqueous amine solvents for CO2 capture, Ind. Eng. Chem. Res., 2022, 61, 15737–15753 Search PubMed.
- F. M. Brethome, N. J. Williams, C. A. Seipp, M. K. Kidder and R. Custelcean, Direct air capture of CO2 via aqueous-phase absorption and crystalline-phase release using concentrated solar power, Nat. Energy, 2018, 3, 553–559 Search PubMed.
- M. Liu, R. Custelcean, S. Seifert, I. Kuzmenko and G. Gadikota, Hybrid absorption-crystallization strategies for the direct air capture of CO2 using phase-changing guanidium bases: Insights from in operando X-ray scattering and infrared spectroscopy measurements, Ind. Eng. Chem. Res., 2020, 59, 20953–20959 Search PubMed.
- H. Cai, X. Zhang, L. Lei and C. Xiao, Direct CO2 capture from air via crystallization with a trichelating iminoguanidine ligand, ACS Omega, 2020, 5, 20428–20437 Search PubMed.
- S. Kim, M. Choi, J. S. Kang, H. Joo, B. H. Park, Y.-E. Sung and J. Yoon, Electrochemical recovery of LiOH from used CO2 adsorbents, Catal. Today, 2021, 359, 83–89 Search PubMed.
- L. D. Ellis, A. F. Badel, M. L. Chiang, R. J. Y. Park and Y.-M. Chiang, Toward electrochemical synthesis of cement-an electrolyzer-based process for decarbonating CaCO3 while producing useful gas streams, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 12584–12591 Search PubMed.
- M. D. Eisaman, L. Alvarado, D. Larner, P. Wang, B. Garg and K. A. Littau, CO2 separation using bipolar membrane electrodialysis, Energy Environ. Sci., 2011, 4, 1319–1328 Search PubMed.
- S. V. Castaño, Q. Shu, M. Shi, R. Blauw, P. L. Fosbol, P. Kuntke, M. Tedesco and H. V. M. Hamelers, Optimizing alkaline solvent regeneration through bipolar membrane electrodialysis for carbon capture, Chem. Eng. J., 2024, 488, 150870 Search PubMed.
- F. Sabatino, M. Gazzani, F. Gallucci and M. v. S. Annaland, Modeling, optimization, and techno-economic analysis of bipolar membrane electrodialysis for direct air capture processes, Ind. Eng. Chem. Res., 2022, 61, 12668–12679 Search PubMed.
- Q. Shu, L. Legrand, P. Kuntke, M. Tedesco and H. V. M. Hamelers, Electrochemical regeneration of spent alkaline absorbent from direct air capture, Environ. Sci. Technol., 2020, 54, 8990–8998 Search PubMed.
- Q. Shu, M. Haug, M. Tedesco, P. Kuntke and H. V. M. Hamelers, Direct air capture using electrochemically regenerated anion exchange resins, Environ. Sci. Technol., 2022, 56, 11559–11566 Search PubMed.
- X. Zhang, Z. Fang, P. Zhu, Y. Xia and H. Wang, Electrochemical regeneration of high-purity CO2 from (bi)carbonates in a porous solid electrolyte reactor for efficient carbon capture, Nat. Energy, 2025, 10, 55–65 Search PubMed.
- Y. Xu, S. Liu, J. P. Edwards, Y. C. Xiao, Y. Zhao, R. K. Miao, M. Fan, Y. Chen, J. E. Huang, E. H. Sargent and D. Sinton, Regeneration of direct air CO2 capture liquid via alternating electrocatalysis, Joule, 2023, 7, 2107–2117 Search PubMed.
- S. Liu, J. Zhang, F. Li, J. P. Edwards, Y. C. Xiao, D. Kim, P. Papangelakis, J. Kim, D. Elder, P. De Luna, M. Fan, G. Lee, R. K. Miao, T. Ghosh, Y. Yan, Y. Chen, Y. Zhao, Z. Guo, C. Tian, P. Li, Y. Xu, E. H. Sargent and D. Sinton, Direct air capture of CO2 via cyclic viologen electrocatalysis, Energy Environ. Sci., 2024, 17, 1266–1278 Search PubMed.
- Y. Liu, H.-Z. Ye, K. M. Diederichsen, T. Van Voorhis and T. A. Hatton, Electrochemically mediated carbon dioxide separation with quinone chemistry in salt-concentrated aqueous media, Nat. Commun., 2020, 11, 2278 Search PubMed.
- S. Voskian and T. A. Hatton, Faradaic electro-swing reactive adsorption for CO2 capture, Energy Environ. Sci., 2019, 12, 3530–3547 Search PubMed.
- H. Xie, Y. Wu, T. Liu, F. Wang, B. Chen and B. Liang, Low-energy-consumption electrochemical CO2 capture driven by biomimetic phenazine derivatives redox medium, Appl. Energy, 2020, 259, 114119 Search PubMed.
- S. Pang, S. Jin, F. Yang, M. Alberts, L. Li, D. Xi, R. G. Gordon, P. Wang, M. J. Aziz and Y. Ji, A phenazine-based high-capacity and high-stability electrochemical CO2 capture cell with coupled electricity storage, Nat. Energy, 2023, 8, 1126–1136 Search PubMed.
- A. Hemmatifar, J. S. Kang, N. Ozbek, K.-J. Tan and T. A. Hatton, Electrochemically mediated direct CO2 capture by a stackable bipolar cell, ChemSusChem, 2022, 15, e202102533 Search PubMed.
- J. M. Barlow and J. Y. Yang, Oxygen-stable electrochemical CO2 capture and concentration with quinones using alcohol additives, J. Am. Chem. Soc., 2022, 144, 14161–14169 Search PubMed.
- Y. Jing, K. Amini, D. Xi, S. Jin, A. M. Alfaraidi, E. F. Kerr, R. G. Gordon and M. J. Aziz, Electrochemically induced CO2 capture enabled by aqueous quinone flow chemistry, ACS Energy Lett., 2024, 9, 3526–3535 Search PubMed.
- X. Jin, S. Jin, L. Li, R. G. Gordon, P. Wang, M. J. Aziz and Y. Ji, Direct air capture of CO2 in an electrochemical hybrid flow cell with a spatially isolated phenazine electrode, Nat. Energy, 2025, 10, 1146–1154 Search PubMed.
- S. Jin, M. Wu, R. G. Gordon, M. J. Aziz and D. G. Kwabi, pH swing cycle for CO2 capture electrochemically driven through proton-coupled electron transfer, Energy Environ. Sci., 2020, 13, 3706–3722 Search PubMed.
- Carbon Engineering, 1PointFive and TD Announce One of the Finance Industry's Largest Purchases of Direct Air Capture Carbon Removal Credits, https://carbonengineering.com/news-updates/1pointfive-and-td-announce-one-of-the-finance-industrys-largest-purchases-of-direct-air-capture-carbon-removal-credits/, (accessed October 2025).
- X. Yang, Z. Chen, C. Xiang, H. Shan and R. Wang, Enhanced continuous atmospheric water harvesting with scalable hygroscopic gel driven by natural sunlight and wind, Nat. Commun., 2024, 15, 7678 Search PubMed.
- A. Ghaffarkhah, M. Panahi-Sarmad, S. Rostami, O. Zaremba, L. A. Bauman, S. A. Hashemi, S. Dutta, P. Yang, T. Guo, F. Jiang, S. Wuttke, M. Arjmand and O. J. Rojas, Ambient-dried MOF/Cellulose-based aerogels for atmospheric water harvesting and sustainable water management in agriculture, Adv. Funct. Mater., 2025, 35, 2506427 Search PubMed.
- W. Guan, Y. Zhao, C. Lei, Y. Wang, K. Wu and G. Yu, Molecularly functionalized biomass hydrogels for sustainable atmospheric water harvesting, Adv. Mater., 2025, 37, 2420319 Search PubMed.
- H. Lin, Y. Yang, Y.-C. Hsu, J. Zhang, C. Welton, I. Afolabi, M. Loo and H.-C. Zhou, Metal–organic frameworks for water harvesting and concurrent carbon capture: A review for hygroscopic materials, Adv. Mater., 2024, 36, 2209073 Search PubMed.
- M. Shen, L. Ji, D. Cheng, Z. Wang, Q. Xue, S. Feng, Y. Luo, S. Chen, J. Wang, H. Zheng, X. Wang, P. Sautet and J. Zhu, Hierarchical design enables sufficient activated CO2 for efficient electrolysis of bicarbonate to CO, Joule, 2024, 8, 1999–2015 CrossRef CAS.
- Y. Kim, E. W. Lees, C. Donde, A. M. L. Jewlal, C. E. B. Waizenegger, B. M. W. de Hepcée, G. L. Simpson, A. Valji and C. P. Berlinguette, Integrated CO2 capture and conversion to form syngas, Joule, 2024, 8, 3106–3125 Search PubMed.
- N. Kumar, V. S. Bryantsev and S. Roy, The role of nonequilibrium solvent effects in enhancing direct CO2 capture at the air–aqueous amino acid interface, J. Am. Chem. Soc., 2025, 147, 1411–1415 Search PubMed.
- H. Mashhadimoslem, M. A. Abdol, P. Karimi, K. Zanganeh, A. Shafeen, A. Elkamel and M. Kamkar, Computational and machine learning methods for CO2 capture using metal–organic frameworks, ACS Nano, 2024, 18, 23842–23875 Search PubMed.
- Y. Lim, H. Park, A. Walsh and J. Kim, Accelerating CO2 direct air capture screening for metal-organic frameworks with a transferable machine learning force field, Matter, 2025, 8, 102203 Search PubMed.
- C. Brady, M. E. Davis and B. Xu, Integration of thermochemical water splitting with CO2 direct air capture, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 25001–25007 CrossRef CAS PubMed.
- A. Datta and R. Krishnamoorti, Analysis of direct air capture integrated with wind energy and enhanced oil recovery, Environ. Sci. Technol., 2023, 57, 2084–2092 Search PubMed.
- E. Prats-Salvado, N. Jagtap, N. Monnerie and C. Sattler, Solar-powered direct air capture: Techno-economic and environmental assessment, Environ. Sci. Technol., 2024, 58, 2282–2292 Search PubMed.
- X.-M. Hu, H.-Q. Liang, A. Rosas-Hernández and K. Daasbjerg, Electrochemical valorization of captured CO2: Recent advances and future perspectives, Chem. Soc. Rev., 2025, 54, 1216–1250 Search PubMed.
- M. Namdari, Y. Kim, D. J. D. Pimlott, A. M. L. Jewlal and C. P. Berlinguette, Reactive carbon capture using electrochemical reactors, Chem. Soc. Rev., 2025, 54, 590–600 Search PubMed.
- L. Li, S. Miyazaki, Z. Wu, T. Toyao, R. Selyanchyn, Z. Maeno, S. Fujikawa and K.-i. Shimizu, Continuous direct air capture and methanation using combined system of membrane-based CO2 capture and Ni-Ca based dual functional materials, Appl. Catal., B, 2023, 339, 123151 CrossRef CAS.
- T. Tatsumichi, R. Okuno, H. Hashimoto, N. Namiki and Z. Maeno, Direct capture of low-concentration CO2 and selective hydrogenation to CH4 over Al2O3-supported Ni–La dual functional materials, Green Chem., 2024, 26, 10842–10850 Search PubMed.
- Y. Tian, R. Wang, S. Deng, Y. Tao, W. Dai, Q. Zheng, C. Huang, C. Xie, Q. Zeng, J. Lin and H. Chen, Coupling direct atmospheric CO2 capture with photocatalytic CO2 reduction for highly efficient C2H6 production, Nano Lett., 2023, 23, 10914–10921 CrossRef CAS PubMed.
- Y.-O. He, W.-Y. Zheng, Y. Liu, W.-D. Zhang, H. Pang, J. Zhang, W.-K. Han and Z.-G. Gu, Direct air capture and photoconversion of CO2 to ethylene by defect-tailored Cu3-based metal–organic frameworks, Angew. Chem., Int. Ed., 2026, 65, e16438 CrossRef CAS PubMed.
- S. Ma, Y. Kim, Z. Zhang, S. Ren, C. Donde, L. Melo, A. S. R. Williams, M. Stolar, E. R. Grant and C. P. Berlinguette, Cathode surface pH modulates multicarbon product selectivity during the electrochemical conversion of CO2 capture solutions, ACS Energy Lett., 2024, 9, 2326–2332 CrossRef CAS.
- Y.-B. Zou, A.-C. Zheng, L.-H. Feng, L. Du, K. Daasbjerg and X.-M. Hu, Integrated capture and electrochemical conversion of CO2 from flue gas mediated by carbonate/bicarbonate cycle, J. Mater. Chem. A, 2025, 13, 36191–36201 RSC.
- Y.-B. Zou, A.-C. Zheng, C.-Q. Yin, L. Du, K. Daasbjerg and X.-M. Hu, Simultaneous CO2 capture and conversion from flue gas using bicarbonate electrolysis over a nickel single-atom catalyst, Small, 2026, 22, e09800 CrossRef CAS PubMed.
| This journal is © Institute of Process Engineering of CAS 2026 |