 Open Access Article
Open Access ArticleAdvanced photocatalysis enabled by water-state-driven interface design
Qian Zhang†
a,
Dongmin Liu†
a,
Bin Yanga,
Hao Liua,
Guangfu Liao*b,
Anna Lipovkad,
Raul D. Rodriguez
*d,
Jun Li *c and
Xin Jia
*c and
Xin Jia
 *a
*a
aSchool of Chemistry and Chemical Engineering, Shihezi University, Shihezi, 832003, P. R. China. E-mail: jiaxin@shzu.edu.cn
bNational Forestry and Grassland Administration Key Laboratory of Plant Fiber Functional Materials, College of Materials Engineering, Fujian Agriculture and Forestry University, Fuzhou 350002, China. E-mail: liaogf@fafu.edu.cn
cHenan Institute of Advanced Technology, Zhengzhou University, Zhengzhou, 450052, P. R. China. E-mail: junli2019@zzu.edu.cn
dTomsk Polytechnic University, 30 Lenin Avenue, 634050 Tomsk, Russia. E-mail: raul@tpu.ru
First published on 9th April 2026
Abstract
Photocatalytic conversion of abundant gaseous small molecules on Earth (such as CO2, N2, and O2) into high-value chemicals is a promising strategy for renewable fuel production and environmental remediation. However, conventional gas–liquid photocatalytic interfaces face three unavoidable bottlenecks. Poor utilization of solar energy, rapid charge carrier recombination, and sluggish mass transfer limit the efficiencies of solar-to-fuel processes and the widespread application of photocatalysis in industry. Recent advances in water-state interface engineering, namely utilizing liquid, microdroplet, and vapor phases, have demonstrated unprecedented performance enhancements for earth-abundant gas conversions. This review critically analyzes mechanistic principles of phase-tailored photocatalyst design, elucidates interfacial charge and mass transfer dynamics, and discusses structure–activity relationships in CO2 reduction, N2 fixation, and H2O2 synthesis. Supported by recent experimental data, we highlight emerging opportunities in metastable interface engineering, offering actionable insights to overcome limitations in bi-phase systems. These innovations are critical for scalable solar chemical production, advancing the industrialization of photocatalytic technologies.
Keywords: Photocatalysis; Phase interface; Liquid water; Microdroplets; Water vapor.
1 Introduction
The escalating global energy and environmental crisis, exacerbated by the unsustainable consumption of fossil fuels, have spurred an urgent demand for advanced sustainable energy conversion technologies. Emerging solutions, including solar photovoltaics, wind energy, electrocatalysis, hydropower, and carbon capture and utilization, hold significant promise for mitigating the detrimental impacts of fossil fuel dependence and facilitating the transition toward carbon neutrality goals.1 Among these technologies, photocatalysis has emerged as a particularly promising approach due to its unique capability to directly utilize solar energy for the conversion of ubiquitous atmospheric small molecules (e.g., H2O, CO2, N2, O2) into high-value chemicals, such as renewable fuels and fertilizers.2,3Since the pioneering discovery of photocatalysis by Fujishima and Honda in 1972,4 this environmentally friendly and cost-effective technology has demonstrated substantial potential for real-world implementation. A notable example is the work by Domen's group, who developed a 100 m2 panel reactor based on aluminum-doped strontium titanate, achieving a solar-to-hydrogen (STH) efficiency of 0.76% with operational stability exceeding one year.5 Inspired by natural photosynthesis, Li et al. introduced the concept of a “hydrogen farm” based on suspended photocatalyst nanoparticles for large-scale solar-driven water splitting. This system achieved a remarkable STH efficiency of over 1.8%, which is the highest reported value for overall water splitting to date.6 Huang et al., in turn, utilized the intrinsic polarization properties of polar crystals to induce enhanced internal electric fields, which promote photogenerated charge separation. This strategy allowed the use of these crystals as photocatalytic materials with enhanced performance.7–9
Despite their conceptual simplicity, conventional solid–liquid bi-phase photocatalytic systems, where chemical reactions occur at the interface between the solid photocatalyst and an aqueous medium, face significant challenges that limit their industrial applications. Three interrelated bottlenecks persist: (i) inefficient solar spectrum utilization due to limited light absorption, (ii) rapid recombination of photogenerated electron–hole pairs before interfacial charge transfer, and (iii) constrained mass transport of reactants to active sites. Despite significant advances in photocatalyst design and strong theoretical support, these inherent limitations continue to hinder the practical efficiency of biphasic systems below industrially viable thresholds.
Emerging phase-interface modulation strategies, achieved by controlling the states of water (namely, liquid water, microdroplets, and water vapor), can dramatically enhance photocatalytic performance by simultaneously addressing all three fundamental limitations. The development of innovative interfacial architectures, particularly gas–liquid–solid triphase and gas–solid biphasic systems, has enabled (i) broadband solar energy harvesting through enhanced light–matter interactions, (ii) efficient charge separation via interfacial electric fields or synergistic photothermal/hydrovoltaic effects, and (iii) optimal mass transport by circumventing traditional solubility and diffusion constraints for gaseous reactants. These tailored interfaces represent a paradigm shift in photocatalytic system design, offering new pathways to overcome the efficiency bottlenecks of conventional approaches.
The growing research interest in phase-interface engineering for photocatalysis is reflected in publication trends. The Web of Science search using keywords such as “three-phase photocatalysis”, “gas–liquid–solid interface”, “microdroplet photocatalysis”, or “gas-phase photocatalysis” revealed over 1750 publications from 2015 to 2025, with approximately 1170 articles (67%) appearing in the most recent five-year period (2020–2025), as shown in Fig. 1. This trend highlights the swiftly growing interest and substantial advances in utilizing water-state modulation to design more efficient photocatalytic interfaces.
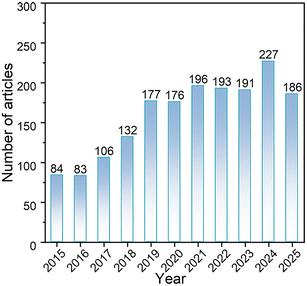 | ||
| Fig. 1 Publication trends for phase-interface modulation in photocatalysis (2015–2025). Data retrieved from Web of Science. | ||
This review article aims to systematically analyze how phase-interface modulation, enabled by controlling water states, enhances photocatalytic efficiency. The article is organized into four main sections:
Section 2 explains how three water states – liquid water, microdroplets, and water vapor – enhance photocatalytic efficiency. We explore the underlying mechanisms, highlight recent state-of-the-art developments, and identify current limitations and future directions for each approach.
Section 3 focuses on specific applications of the discussed phase-interface modulations in CO2 reduction, H2O2 synthesis, and N2 fixation. Through detailed examples and performance metrics, we evaluate the potential to overcome traditional limitations of photocatalysis.
In section 4, we summarize the key experimental and theoretical methods employed to unravel the structure–activity relationships and dynamic mechanisms at these advanced interfaces, providing a toolbox for future research in this field.
In section 5, we provide a summary and outlook highlighting the key advantages and practical implementations relevant to experts in materials science, photocatalysis, and energy conversion. We discuss open questions and research opportunities that could accelerate photocatalytic approaches toward industrial-scale sustainable energy and environmental solutions.
2 Phase-interface modulation via water state control
As stated in the introduction, conventional solid–liquid biphasic photocatalytic systems face fundamental challenges such as poor light utilization, rapid charge recombination, and inefficient mass transfer, which limit their practical application in industry. In this section, we systematically analyze three water-state-based interfacial systems, explaining how they enhance light harvesting, charge carrier dynamics, and reactant accessibility.2.1 Liquid water
In conventional solid–liquid bi-phase photocatalytic systems, reactions occur at the interface between a solid photocatalyst and an aqueous medium. In contrast, gas–liquid–solid tri-phase interfaces in liquid water include gas as a third phase, positioning the photocatalyst at the water–gas boundary to allow direct sunlight exposure and access to gaseous reactants, thus overcoming the limitations of conventional systems. There is already solid evidence supporting the efficiency of such systems, as discussed in several studies.10–18Mainly, such tri-phase interfaces are achieved through two configurations: (1) photocatalysts are immobilized on lightweight, hydrophobic carriers that float on water, or (2) modified photocatalysts with induced hydrophobicity are suspended in water. These interactions create a gas–liquid–solid interface that promotes photocatalytic reactions.
For case (1), photocatalysts like TiO2 or g-C3N4 are loaded onto carriers such as carbon fiber, polystyrene,19 melamine sponges,15 polytetrafluoroethylene fiber films,14,20,21 or hydrogels12,18 via electrostatic assembly or in situ growth, allowing direct contact with gas reactants. In traditional solid–liquid two-phase systems, gaseous reactants (such as CO2) need to dissolve in the aqueous phase before diffusing to the catalyst surface. Thus, this two-step process is fundamentally limited by the low solubility of gases in water (for CO2-1.45 g L−1 at room temperature), resulting in limited mass transfer. In contrast, in the gas–liquid–solid three-phase system, the catalyst is deliberately positioned at the gas–water interface, enabling gaseous reactants to adsorb directly onto the catalyst surface from the gas phase, significantly increasing the local concentration of reactants and the mass transfer rate.18,22
For instance, Sheng et al. showed immobilizing TiO2 on a superhydrophobic PTFE-treated carbon fiber substrate (Fig. 2a). This interface promoted a 10-fold increase (compared to a conventional system) in pollutant degradation rates under UV light by using O2 as an electron scavenger to minimize electron–hole recombination and produce abundant reactive oxygen free radicals.22 Similarly, Lee et al. reported a floatable elastomer–hydrogel nanocomposite photocatalytic platform, doubling H2 production rates (163 mmol h−1 m−2) compared to submerged systems by avoiding light attenuation and facilitating H2 gas separation (Fig. 2b).18
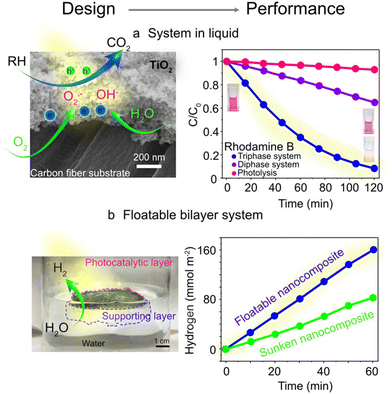 | ||
| Fig. 2 Design and performance of photocatalytic platforms, exploiting gas–liquid–solid tri-phase interfaces in liquid water. (a) Scanning electron microscopy image of a tri-phase system together with the concept of photocatalytic reactions, implementation in rhodamine B degradation. Reprinted with permission from ref. 22. Copyright 2017, American Chemical Society; (b) floating nanocomposite promoting solar hydrogen production: photo, concept, and H2 evolution rate. Reprinted with permission from ref. 18. Copyright 2023, Springer Nature. | ||
In the second case, photocatalysts are modified with organic molecules like alkyl acids with different carbon chain lengths23 or (3-aminopropyl) trimethoxysilane24 by chemical bonding. This modification increases hydrophobicity and allows photocatalysts to suspend at the water–gas interface, enhancing O2 adsorption and charge transfer. The three-phase systems mainly regulate the interface microenvironment by suppressing competitive side reactions (such as the hydrogen evolution reaction, HER)10 and promoting the detachment of products. In traditional solid–liquid systems where the catalyst is fully immersed in water, the catalyst surface is saturated with water molecules, creating a high local proton concentration that favors the HER. In contrast, positioning the catalyst at the gas–water interface in a three-phase system spatially separates the catalyst from water, reducing the availability of protons at catalytic sites while maintaining direct access to gas. Additionally, at the gas–liquid–solid interface, the accessibility of the gas phase provides a thermodynamic driving force for volatile products (such as CO, CH4, or H2) to desorb rapidly into the gas phase rather than remaining adsorbed in the aqueous environment.22,24
Both approaches propose several advantages, including:
A. Enhanced solar light utilization because of the direct photocatalyst exposure to light;
B. Improved reactant accessibility caused by the facilitated adsorption and direct contact between the reactant and the photocatalyst;
C. Increased selectivity to the target product by suppressing side reactions and product decomposition;
D. Simplified recycling of the catalysts compared to solid–liquid systems.
The key consideration is how these tri-phase systems will function in real-world applications. Floating photocatalysts in the form of nets are ideal for in situ degradation of hydrophobic organic pollutants, such as polycyclic aromatic hydrocarbons, algal toxins, or pesticides. These photocatalytic nets do not sink, do not require mixing, and are easily recoverable.25 Suspended photocatalysts, on the other hand, perform exceptionally well in continuous flow reactors. Such reactors can synthesize hydrogen peroxide in situ on a large scale for antibacterial and Fenton-like reactions, and can also produce liquid nitrogen fertilizer through nitrogen fixation to promote crop growth. Such systems take advantage of a large contact interface with dissolved pollutants and oxygen in the water.26
It is worth noting that these interfaces are relatively new, and challenges still exist in their implementation. For instance, the pending issues include weak binding between photocatalysts and hydrophobic carriers, which can lead to detachment during extended use; and difficulties in regulating hydrophilicity to optimize contact and photocatalytic activity. To address these issues, the current and future research should focus on developing robust carrier–photocatalyst interfaces and elucidating the relationship between hydrophobic microenvironments and photocatalytic performance to enable industrial-scale applications.
2.2 Microdroplets
Microdroplets are familiar to us as mist near waterfalls, clouds forming on mountains in the morning, or vapor from humidifiers. They are produced under the combined effects of surface tension and hydrodynamic forces, when liquid water breaks into tiny droplets (1–20 μm) under external forces like shear or electric fields, storing energy as surface tension. Unlike bulk water, microdroplets possess a high specific surface area, creating many gas–liquid interfaces with unique thermodynamic and kinetic properties that accelerate chemical reactions and enable interfacial catalysis.27–29 These properties make microdroplets a promising platform for enhancing photocatalytic efficiency in gas–liquid–solid tri-phase systems. This section explores the surface electric fields of microdroplets and their coupling with photocatalysis, highlighting their mechanisms, applications in environmental pollutant management and clean energy production, and the challenges for industrial applications.For example, Xie et al. demonstrated that strong microdroplet electric fields reduce energy barriers in Menshutkin reactions, enhancing reaction rates.40 This concept is shown in Fig. 3a. In another study, Wang et al. utilized Raman spectroscopy to study photochemical oxidation of organic matter with Fe(III)-oxalate in microdroplets, demonstrating that the surface electric field activates oxidants to generate free radicals and remove pollutants as visualized with rhodamine dye degradation.41
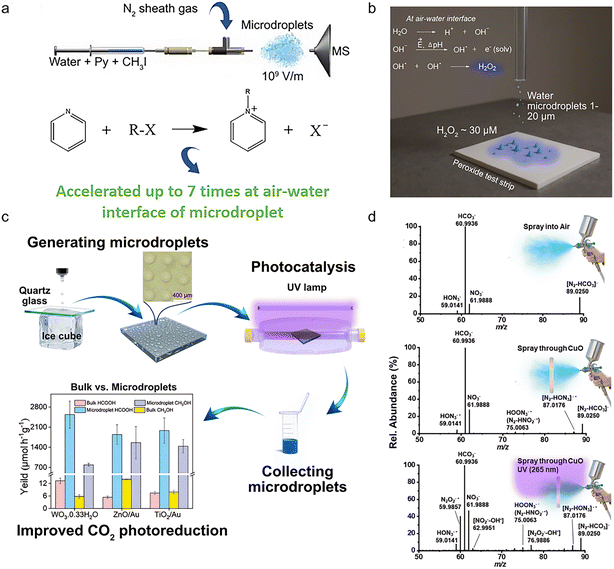 | ||
| Fig. 3 Photocatalysis using microdroplets systems. (a) Reducing energy barriers in Menshutkin reactions. Reprinted with permission from ref. 40. Copyright 2023, American Chemical Society; (b) spontaneous production of H2O2 using microdroplets of pure water. Reprinted with permission from ref. 39. Copyright 2019, PNAS; (c) the most efficient CO2 photoreduction using microdroplets: concept and performance. Reprinted with permission from ref. 52. Copyright 2023, Wiley-VCH GmbH; (d) real-time monitoring of the urea generation process by spraying water microdroplets and the CuBi2O4 catalyst. Reprinted with permission from ref. 54. Copyright 2023, American Chemical Society. | ||
It has been reported that high electric fields within the microdroplets' surface can catalyze chemical reactions and break water molecules, leading to the production of strongly reducing free electrons and hydroxyl radicals.42–45 In 2019, Zare et al. revealed for the first time that microdroplets of pure water spontaneously produce hydrogen peroxide (H2O2) at their surfaces.39 This concept is illustrated in Fig. 3b where the proposed mechanism involves ·OH recombination at the interface, which are generated by the loss of electrons from OH− at the surface of the water microdroplets.
These electric field-driven properties make microdroplets a versatile platform for accelerating reaction rates in applications like organic synthesis, pollutant degradation and H2O2 synthesis. However, these reactions currently suffer from low efficiency due to limitations in scalability and droplet stability control. Therefore, it is essential to integrate these methods with other technologies.
For example, Zare et al. significantly enhanced the stereoselective reduction of ketones to chiral alcohols by using enzymes, photosensitizers, cofactors, and triethanolamine in microdroplet form. Switching to microdroplets from the bulk state contributed to reducing the optical path of transmitted light, resulting in increased light intensity that promoted photocatalysis, reaching yields 1.5–5.6 times higher than in bi-phase systems.50
Using microdroplets indeed can significantly boost photocatalysis because of several configuration advantages:51,52
A. The strong gas–liquid interfacial electric field within microdroplets generates a powerful driving force for the separation of photogenerated charge carriers.
B. Microdroplets improve the adsorption of gaseous reactants, promoting their enrichment at the interface, and enabling more efficient interactions and mass transfer.
C. The unique shape and refractive properties of microdroplets enhance local light absorption, thus improving the photocatalytic activity.
D. Additionally, the unique phase interface of microdroplets can lower the reaction energy barrier and improve selectivity.
The aforementioned advantages, particularly the enhanced adsorption of gaseous reactants (B), stem from the multiple effects of the droplet system: firstly, the extremely high specific surface area provides sufficient contact interfaces; secondly, the strong self-generated electric field at the interface (∼109 V m−1) can polarize the reactant molecules, promoting their enrichment and activation through electrostatic interactions; thirdly, the thermodynamic surface enrichment effect caused by the curved interface further enhances the local concentration of reactants in the interface region; finally, the droplets' micrometer-scale size reduces the gas transport resistance significantly. The synergy of these effects collectively surpasses traditional bulk or conventional two-phase systems, achieving a significant enhancement in the adsorption and transport of gaseous reactants.27–32,35–38
Technically, there are two ways to couple microdroplets with photocatalysis:
(1) Dispersing photocatalyst powder in liquid water and generating microdroplets of the suspension using a microdroplet generator to enhance the contact area between the photocatalyst and the reactants, improving reaction efficiency51,52 (see Fig. 3c).
(2) Spraying microdroplets of water directly onto a surface coated with the photocatalyst for the reaction to occur.53 For instance, this approach was used for urea synthesis54 (see Fig. 3d).
Both methods are viable, each with its pros and cons, and the choice depends on the specific requirements of the application. For instance, spraying water microdroplets (2) makes the recycling and reuse of the photocatalysts easier. However, as the reaction proceeds, the hydrophilic surface of the catalyst tends to become saturated with water, potentially hindering effective interaction with fresh microdroplets and reducing efficiency.
The proposed tri-phase interfaces in microdroplets could be adapted to several practical applications. For instance, in industrial exhaust gas treatment, particularly from petrochemicals, printing, painting, and semiconductor fabrication, microdroplets can capture volatile organic compounds (VOCs). These VOCs accumulate at the droplet surface, where loaded photocatalysts generate reactive radicals under UV illumination, leading to the mineralization of VOCs into CO2 and H2O.55 Another demonstrated industrial application is high-value chemical synthesis. Microdroplet reactors act as continuous flow platforms, dispersing reactants and photocatalysts to accelerate light-driven reactions like oxidation, C–H activation, asymmetric synthesis, hydrogenation, and dehydrogenation.56–58 Lastly, these interfaces play a crucial role in photocatalytic CO2 utilization, H2O2 production and N2 fixation. Microdroplets enhance these photocatalytic reactions by improving gas solubility and reaction kinetics, supporting sustainable fuel production.
In recent years, microdroplet-based interfaces have shown remarkable potential for enhancing photocatalytic efficiency. However, these systems remain relatively rare, which results in limited exploration of the full range of photocatalytic reactions and materials compatible with microdroplet environments. To unlock their full potential in energy and environmental applications, we believe that future research should prioritize the development of stable photocatalyst-microdroplet systems, the investigation of new reaction types, and the improved design of scalable microdroplet reactors.
2.3 Water vapor
A further reduction of microdroplets in size leads to another water state useful for photocatalysis: water vapor. This medium is environmentally friendly and sustainable, offering several advantages in heterogeneous catalytic systems. It's low viscosity, high polarity, and dynamic adsorption properties can enhance dynamic interactions between gaseous reactants and the photocatalyst's active sites.59 This section explores the effects and mechanisms of water vapor systems in clean energy production and environmental remediation, including applications in water decomposition, CO2 or N2 reduction, and challenges associated with practical implementation.Water vapor is used in solid–gas bi-phase interfaces to significantly enhance photocatalytic activity. For instance, Suguro et al. demonstrated efficient overall water splitting using Al-doped SrTiO3 photocatalysts coated with TiOx or TaOx nanomembranes under vapor feeding. The system maintained a STH efficiency of 0.4% over 100 hours, even with using seawater as the vapor source. This approach minimizes the reverse O2 reduction to H2O, a common issue in solid–liquid systems.60
There are two primary approaches for achieving water vapor-based interfaces. The first approach involves directly introducing water vapor into the reactor. Zhang et al. used this approach to tackle the issue of O2 reverse reduction to H2O and developed three COF-based photocatalysts which are denoted as Pt@Tp-TAPB-COF (Tp = 1,3,5-triformylphloroglucinol, TAPB = 1,3,5-tris(4-aminophenyl)benzene), Pt@Tp-TAPT-COF (TAPT = 2,4,6-tris(4-aminophenyl)-1,3,5-triazine), and Pt@Tp-TAPyT-COF (TAPyT = tripyridyltriazine).
Among them, Pt@Tp-TAPyT-COF exhibits the best photocatalytic overall water splitting activity (Fig. 4a). The core reason behind this superior performance originates from several synergistic reasons. First of all, the unique composite structure provides abundant active sites; second, the in situ deposited Pt nanoparticles function both as active sites for the HER and as electron-withdrawing centers that create strong electronic coupling with the COF framework, facilitating efficient charge separation; third, the pyridine-N atoms in the catalyst skeleton reduce the Gibbs free energy barrier of the OER, accelerating O2 generation and promoting its rapid desorption before photogenerated electrons can reduce it back to water. Unlike a traditional solid–liquid reaction system, the catalyst in the gas–solid reaction system has no direct contact with liquid water. This spatial separation prevents dissolved O2 from accumulating photogenerated electrons on the catalyst, fundamentally suppressing the competing O2 reduction pathway and enabling efficient overall water splitting.61
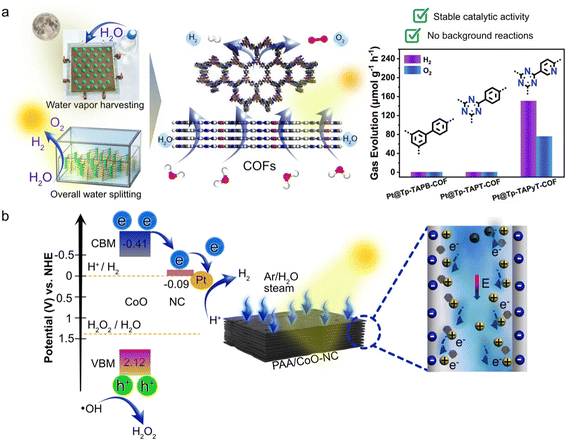 | ||
| Fig. 4 Water vapor systems in photocatalysis. (a) Schematic diagram of COF photocatalysts for overall water vapor splitting and gas evolution metrics. Reprinted with permission from ref. 61. Copyright 2025, Wiley-VCH GmbH; (b) the band structure and schematic illustration of hydrovoltaic effect-enhanced photocatalytic water splitting. Reprinted with permission from ref. 65. Copyright 2023, Springer Nature. | ||
Notably, the gas–solid bi-phase interfaces formed between water vapor and the photocatalyst could also exhibit photothermal or hydrovoltaic effects. These effects optimize charge carrier separation and proton migration, thus enhancing solar energy conversion efficiency. The second approach involves loading photocatalysts with photothermal materials to generate water vapor in situ through photothermal water evaporation. This photothermal effect enhances reaction efficiency by converting sunlight, especially in the visible and near-infrared range, into thermal energy, which facilitates water vapor generation and accelerates different photocatalytic processes. In addition, the heat generated by the photothermal base can be radiated to accelerate carrier separation and the desorption of gaseous products, further promoting the photocatalytic reaction.
In this context, Li et al. developed a photothermal/photocatalytic Z-scheme heterojunction by combining ZnIn2S4-WO3 with carbonized wood (C-wood), achieving a STH efficiency of 1.52% for overall water splitting. The C-wood serves as a bridge to enhance charge carrier separation and facilitate the transfer of photogenerated carriers, making this photothermal approach promising for increasing efficiency.62 Additionally, the photothermal effect allows for simultaneous freshwater collection, as demonstrated by Bu et al. with a biomimetic COF aerogel photocatalyst that produces H2O2 while desalinating water.63
Besides the photothermal effect, the photovoltaic effect also plays a significant role. The hydrovoltaic effect refers to the direct conversion of kinetic energy from water molecules into electrical energy through interactions between functional materials and water.64 In photocatalysis, this effect occurs at gas–solid interfaces, generating a potential gradient from interfacial water flow (e.g., evaporation, capillarity, or ionic diffusion), which forms a built-in electric field that promotes the directional separation of photogenerated electrons and holes.
For instance, Xin et al. developed a PAA/CoO-NC photocatalyst by combining poly(acrylic acid) with cobalt oxide-modified nitrogen-doped carbon (NC) to simultaneously produce H2 and H2O2 via photocatalytic water splitting65 (Fig. 4b). The generation of hydrovoltaic potential in the PAA/CoO-NC system is mainly attributed to the directional migration and charge separation of H+ induced by water vapor diffusion. This hydrovoltaic effect strengthens interactions between protons and reaction centers of photocatalysts, improving electron transport and water splitting efficiency. The catalyst design involves several key benefits: (1) the high density of carboxylic groups in PAA renders the composite surface negatively charged, facilitating H+ enrichment and directional migration; (2) the porous structure enables continuous vapor flux, maintains the contact between the hydrovoltaic PAA and photocatalytic CoO-NC components, and sustains the humidity gradient required for H+ migration; (3) the hydrovoltaic effect reduces the Schottky barrier at the CoO-NC interface by 33%, significantly promoting charge separation. This combined mechanism of water vapor generating electrostatic potential (hydrovoltaic effect) while light generates photovoltaic potential is the reason for the superior water splitting efficiency of this system. The hydrovoltaic effect, driven by H+ diffusion, strengthens interactions between protons and reaction centers of photocatalysts, improving electron transport and water splitting efficiency. Similarly, Yang et al. integrated photocatalytic H2 evolution with the hydrovoltaic effect in a water generator, reconfiguring ion gradients for continuous current output, thereby enhancing hydrovaporation performance.64
The number of research reports using the hydrovoltaic effect in photocatalysis is limited, with only a few studies reported on this. To advance practical applications in clean energy and environmental remediation, future research should focus on developing materials that exhibit a strong hydrovoltaic effect as well as on investigating interfacial dynamics of water molecules to enhance photocatalytic efficiency.
Despite these advantages, several challenges remain. For example, water molecules have strong polarity and hydrogen bonding ability, which leads to competitive adsorption at active sites, hindering reactant (e.g., organic pollutant molecules, CO2, O2) access as well as product desorption. Additionally, high water vapor concentrations can compete for photogenerated charges during gaseous reactants, promoting unwanted side reactions. In the future, in response to the aforementioned challenges, we should focus on designing specific photocatalysts and reactors to optimize the interface between water vapor, catalysts and gaseous reactants, thereby enhancing the photocatalytic efficiency.
2.4 Summary
Section 2 has systematically elucidated the “water-state-driven” paradigm for interface design in photocatalysis. The core idea behind this concept is that water acts not merely as a solvent or reactant, but its physical state can be leveraged as a primary driver for optimized reaction microenvironments. Specifically:Liquid water drives the creation of gas–liquid–solid triphase interfaces through buoyancy and tailored wettability. This design directly addresses the limitations of gas solubility and light penetration by positioning the catalyst at the water–gas boundary.
Microdroplets drive the formation of high-curvature gas–liquid interfaces characterized by an immense specific surface area and a spontaneously generated intense electric field. These properties concurrently facilitate enhanced local reactant concentration and dramatically accelerated charge separation.
Water vapor drives the establishment of dynamic gas–solid biphasic interfaces. Its low viscosity results in rapid mass transfer, while its interaction with functional materials can induce synergistic photothermal or hydrovoltaic effects, further promoting efficient proton transfer and product desorption.
In each case, the transition from a passive aqueous medium to an actively engineered water-state-based interface provides a targeted solution to the fundamental bottlenecks plaguing conventional photocatalysis.
3 Photocatalytic applications
The modulation of phase interfaces via water state control has been widely applied to enhance the photocatalytic conversion of Earth's abundant small molecules into valuable chemicals. This approach demonstrates superior catalytic activity and selectivity compared to conventional solid–liquid bi-phase interfaces in reactions such as CO2 reduction, H2O2 production, and N2 fixation.22,66,67 In this section, we summarize the progress in phase-interface modulation for these three reactions utilizing liquid water, microdroplets, and water vapor. We highlight the advantages of phase-interface modulation, the rationality behind catalyst design, the underlying mechanisms, and compare catalytic performance.3.1 Photocatalytic CO2 reduction
The excessive greenhouse gas emissions, primarily CO2, due to the overconsumption of fossil fuels, have raised significant concerns. The photocatalytic CO2 reduction reaction (CO2RR) offers a sustainable solution by converting CO2 into valuable carbon derivatives, such as CO, CH4, CH3OH, and C2H4. These reactions are particularly interesting for researchers and engineers because they can occur under mild conditions.Over the past decade, efforts have largely focused on designing photocatalysts for conventional bi-phase interfaces to enhance the efficiency of CO2RR. However, the low solubility and diffusion of CO2 in water at these interfaces significantly hinder photocatalytic efficiency. This subsection discusses successful examples of three different interfaces that have been developed to improve CO2RR. To highlight the advantages of the novel interfaces, we also summarized the CO2RR activity under a water-state-driven interface in Table 1.
| Water state | Material design | Light source | Reaction conditions | Product rate (μmol g−1 h−1) | Selectivity for CO2RR | Enhancement factor | Ref. |
|---|---|---|---|---|---|---|---|
| Liquid water | Ag–TiO2-GDL | 300 W Xe lamp (λ ≤ 400 nm) | Three-phase interface; catalyst floated onto the surface of the water | CO: 220CH4: 100 | 97.8% | 8 times | 68 |
| NAL-MRF | Not available | Three-phase interface; hydrophobic catalyst suspended in water | CH3OH: 31.41 | 93.62% | 4.2 times | 75 | |
| Pt/o-PCN | Visible-light irradiation | Three-phase interface; hydrophobic catalyst suspended in water | CO: 286CH4: 136 | 87.9% | 34 times | 69 | |
| Cu2O–Ag–TiO2/PTFE | Two-sided illumination was implemented from both the top and bottom with uniform light intensities | Three-phase interface; catalyst floated onto the surface of the water | CO: 347.4CH4: 25.1 | 96.2% | 120 times | 14 | |
| g-C3N4/CuFe2O4/ZnIn2S4 | Visible-light irradiation | Three-phase interface; catalyst suspended in water | CH4: 267.4 | 96.8% | 2 times | 76 | |
| Try-CD/CN | 300 W Xe lamp (λ ≥ 420 nm) | Three-phase interface; hydrophobic catalyst suspended in water | CH4: 17.1 | 97% | 6.7 times | 10 | |
| Microdroplets | WO3·0.33H2O | 365 nm UV lamp | Gas–liquid interface; catalysts presented in microdroplets | HCOOH: 2536 | Not available | 195 times | 52 |
| Water vapour | CD/VOR-BMO-1.2 | Not available | The catalyst is in an atmosphere of water vapor and CO2 | CO: 60 | 100% | Not available | 72 |
| CeO2 | 300 W Xe lamp (AM 1.5 G) | The catalyst is in an atmosphere of water vapor and CO2 | CH4: 192.75 | 100% | Not available | 73 | |
| TiO2/SrTiO3 | 300 W Xe lamp (100 mW cm−2) | The catalyst is in an atmosphere of water vapor and CO2 | CO: 100CH4: 47 | 100% | Not available | 70 |
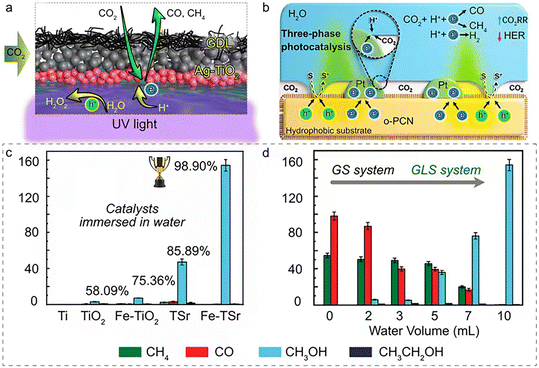 | ||
| Fig. 5 (a) Schematic illustration of the triphase photocatalytic CO2RR system based on Ag-TiO2. Reprinted with permission from ref. 68. Copyright 2022, Wiley-VCH GmbH; (b) scheme of the three-phase photocatalyst Pt/o-PCN and the mechanism of three-phase photocatalysis. Reprinted with permission from ref. 69. Copyright 2019, Wiley-VCH GmbH; (c) CO2 photoreduction for 5 h on different samples in 10 mL pure water; (d) CO2 photoreduction on Fe-TSr with different volumes of water. Reprinted with permission from ref. 70. Copyright 2023, Wiley-VCH GmbH. | ||
The theoretical potential of photocatalytic CO2RR is comparable to that of the hydrogen evolution reaction (HER), making the HER the primary competing process affecting CO2RR selectivity and efficiency. While protons (H+) are abundant in aqueous solutions because of water ionization, CO2 availability at the photocatalyst surface is limited by its low solubility and slow diffusion. Therefore, enhancing CO2 accessibility while reducing proton concentration is critical to suppressing the HER. Recent studies demonstrate that solid–liquid–gas tri-phase interfaces in liquid water effectively mitigate the HER and enhance CO2RR. For example, Antonietti et al. developed a hydrophobic photocatalyst surface that facilitates direct contact between gas-phase CO2, liquid water, and the solid catalyst. This design successfully overcomes CO2 mass transfer limitations, maintaining an impressive 87.9% selectivity for CO2RR, even when using a platinum co-catalyst, a well-known HER-favoring material, as shown in Fig. 5b.69
A typical CO2RR involves both CO2 reduction and water oxidation, which require different environments: gas–solid for CO2 reduction and solid–liquid for water oxidation. To overcome this challenge, Xie et al. designed a photocatalyst by subsequently depositing Cu2O, Ag, and TiO2 layers on a floatable polytetrafluoroethylene (PTFE) membrane. While the system floats on water under illumination, it enables two simultaneous processes: 1) photogenerated electrons get transferred to the gas-exposed Cu2O side for CO2 reduction, and 2) holes are directed to the water-immersed TiO2 side for water oxidation. This innovative configuration leads to high selectivity for CO2RR and H2O2 production while suppressing the HER.14,70
Conventional photocatalytic CO2RR faces several challenges, including limited CO2 accessibility and competition with the HER, which have been effectively addressed in previous studies, as cited above. However, a significant issue is poor product selectivity, partially attributed to interfacial wettability. Hydrophilic surfaces easily bind water molecules and favor the HER, while hydrophobic surfaces enhance CO2 adsorption and promote CO2RR. This means that the distribution of intermediates and the reaction mechanism can be changed by altering the wettability. Huang et al. experimentally indicated this by modifying C3N4 with hydrophobic carbonaceous co-catalysts (Try-CDs), constructing a tri-phase interface between CO2, the catalyst, and water. This Try-CD/C3N4 design allowed 97% selectivity for CH4 with a yield of 17.1 μmol g−1 h−1, which is 6.7 times higher than that of pristine C3N4.10
In this context, Huang et al. developed a Fe@TiO2/SrTiO3 heterostructured nanotube array photocatalyst with excellent CO2RR activity and stability.70 By regulating the water state, the researchers achieved selective synthesis: submerged catalysts in liquid water led to selective CO2RR to prepare CH3OH with over 98.9% selectivity, while CO2RR under water vapor yielded syngas (CO/CH4) with over 99.9% selectivity. The mechanism revealed that in liquid water, abundant ·OH radicals and H2O molecules around monatomic Fe inhibit the dehydration reaction (1), favoring CH3OH formation.
| ·COH + H+ + e− → ·C + H2O | (1) |
In contrast, reduced ·OH and H2O levels in water vapor facilitate this dehydration, leading to the production of ·CH, ·CH2, ·CH3, and CH4. This study provides a valuable reference for controlling water states to achieve selective regulation of photocatalytic CO2RR.
3.2 Photocatalytic H2O2 synthesis
H2O2 is a green chemical, playing a vital role in energy, environmental applications, and chemical synthesis. However, the growing market demands, as well as the limitations of conventional anthraquinone technology have contributed to the need for sustainable H2O2 synthesis approaches. Photocatalysis is indeed a promising eco-friendly strategy, which utilizes only sunlight, H2O and O2 to produce H2O2.For the purpose of H2O2 synthesis, conventional solid–liquid bi-phase interfaces involve immersing powdered photocatalysts in water. The whole process mainly takes place via two possible pathways:
(1) Two-step O2 reduction, following reactions (2) and (3):
| O2 + e− → ·O2− | (2) |
| ·O2− + e− + 2H+ → H2O2 | (3) |
(2) Direct one-step two-electron O2 reduction, following reaction (4):
| O2 + 2e− + 2H+ → H2O2 | (4) |
However, the efficiency of H2O2 synthesis is limited by low solubility (0.9 mM, 298 K, 1 atm) and slow diffusion (∼2.1 × 10−5 cm2 s−1) of O2 in water. These transport issues restrict O2 accessibility to the photocatalyst, limiting the overall efficiency of photocatalytic activity using conventional solid–liquid bi-phase interfaces. Additionally, such systems often suffer from poor product selectivity control, H2O2 decomposition, catalyst recovery difficulties, agglomeration, and limited light harvesting.
Gaseous O2, with its higher diffusion coefficient and concentration, can replace dissolved O2 in water to solve the problem of O2 accessibility to the catalyst. The introduction of the new phase within the control of water state is expected to address all the discussed pending issues and significantly increase the efficiency of H2O2 production16 (Table 2).
| Water state | Material design | Light source | Reaction conditions | Product rate (μmol g−1 h−1) | Apparent quantum efficiency (%) | Enhancement factor | Ref. |
|---|---|---|---|---|---|---|---|
| Liquid water | 2 L-mCN/F-Naf | 300 W Xe lamp (λ ≤ 400 nm) | Three-phase interface; catalyst floated onto the surface of the aqueous ethanol solution | 5380 | 2.1 at 400 nm | Not available | 21 |
| M-SBP | 100 W LED lamp (λ ≥ 420 nm) | Three-phase interface; catalyst floated onto the surface of the water | 717.5 | 2.9 at 420 nm | 3.3 times | 77 | |
| TPB-DMTP-COF | 300 W Xe lamp (λ ≥ 420 nm) | Three-phase interface; catalyst floated onto the surface of the water | 2882 | 18.4 at 420 nm | 15 times | 16 | |
| TiO2/Pt/Co POM | 5 W lamp (λ = 365 nm) | Three-phase interface; catalyst floated onto the surface of the aqueous methanol solution | 1395 | 1.09 at 420 nm | 5 times | 79 | |
| CTTP | 300 W Xe lamp (AM1.5G filter) | Three-phase interface; catalyst floated onto the surface of the water | 1858 | 1.08 at 420 nm | 14 times | 13 | |
| Cv-PCN | 300 W Xe lamp (AM1.5G filter) | Three-phase interface; catalyst floated onto the surface of the water | 2063 | 26.8 at 400 nm | 10 times | 17 | |
| Pd/A/BiVO4 | 300 W Xe lamp (λ ≥ 420 nm) | Three-phase interface; catalyst suspended in water | 805.9 | 6.02 at 400 nm | Not available | 24 | |
| TBO40 | 300 W Xe lamp (780 ≥ λ ≥ 350 nm) | Three-phase interface; catalyst floated onto the surface of furfuryl alcohol aqueous solution | 57![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
1.25 at 365 nm | 6 times | 19 | |
| Microdroplets | g-C3N4 | 365 nm UV lamp (2.32 mW cm−2) | Gas–liquid interface; catalysts presented in microdroplets with Triethanolamine | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600 |
Not available | 75 times | 51 |
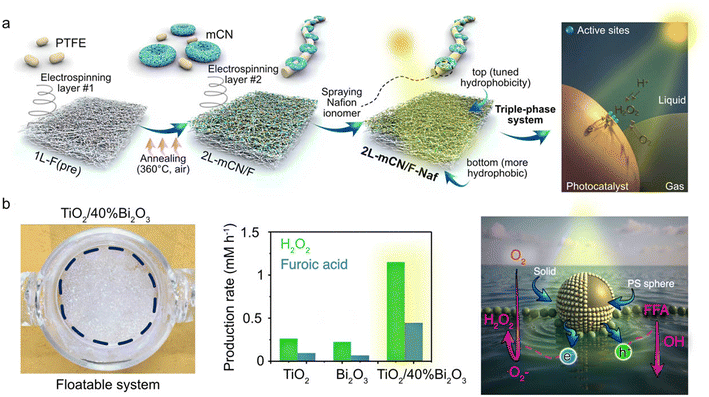 | ||
| Fig. 6 (a) Schematic illustration of the synthesis route for the Janus 2 L-mCN/F-Naf fiber membrane and photocatalytic O2 reduction to H2O2. Reprinted with permission from ref. 21. Copyright 2024, American Chemical Society; (b) floatable S-scheme TiO2/Bi2O3 photocatalyst, production rates of H2O2 and FA for 12 hours, schematic illustration of the photoredox reaction in the proposed system. Reprinted with permission from ref. 19. Copyright 2022, Wiley-VCH GmbH. | ||
Yu's et al. fixed the hydrophobic TiO2 and Bi2O3 on lightweight polystyrene (PS) spheres using hydrothermal and photodeposition methods.19 By constructing an S-scheme (step-scheme) heterojunction, they achieved efficient spatial separation of photogenerated carriers, while retaining the electrons with strong reducing ability (located in the conduction band of Bi2O3) for the two-electron reduction of O2 to H2O2, and the holes with strong oxidizing ability (located in the valence band of TiO2) for the oxidation of organic substrates. This floating structure built a stable gas–liquid–solid three-phase interface on the solution surface, enabling the direct and rapid diffusion of gaseous O2 to the catalyst active sites. This combination results in overcoming the bottleneck of low O2 solubility and slow diffusion in traditional suspension systems, and improving the light utilization efficiency. The floatable S-scheme TiO2/Bi2O3 photocatalyst is transparent to sunlight, achieving a significantly improved H2O2 yield of 1.15 mM h−1, while realizing the conversion of furfuryl alcohol to furfuryl acid, as shown in Fig. 6b.
Later, the same group designed a ternary TiO2/Pt/CoPOM floatable photocatalyst.79 In this design, lightweight PS spheres ensured the photocatalyst floating on the liquid surface, improving light utilization and contact with the gaseous reactants. Pt and Co polyoxometalate (CoPOM) acted as a channel for electron transfer to the Pt sites through ligand networks. This system resulted in a H2O2 yield of 0.93 mM h−1 along with a HCOOH yield of 0.46 mM h−1.
Li et al. developed a tri-phase photocatalytic system comprising air–liquid–solid phases using polymerized carbon nitride (PCN) doped with pyrrole and cyano groups. This development boosts O2 activation and spatially separates the HOMO and LUMO orbitals,17 resulting in improved carrier separation efficiency and selective H2O2 generation. The system achieved an impressive H2O2 generation rate of 2063 μmol g−1 h−1 in pure water, which is about 10 times higher than that of the conventional powder photocatalyst systems.
Building on this, Kang et al. engineered a dual Pd/A/BiVO4 photocatalyst with superhydrophobic and super-oxygenophilic properties to further enhance selectivity and suppress H2O2 decomposition. This tri-phase interface enabled efficient one-step two-electron O2 reduction directly from air, significantly minimizing H2O2 degradation and achieving high product concentrations.24
Zhang's group pioneered this approach by using inkjet-printed water microdroplets containing g-C3N4 nanosheet photocatalysts and triethanolamine as photo-generated hole scavengers.51 The research revealed a dramatic increase in H2O2 generation reaching up to 20.6 mmol g−1 h−1, which is nearly two orders of magnitude higher than conventional biphasic systems. The study also showed a volcano-type dependence on droplet size, suggesting an optimal microdroplet diameter for H2O2 formation. This synergistic effect, resulting from increased O2 access, enhanced photocarrier separation, and interfacial electric fields, enabled efficient H2O2 production. Moreover, the team demonstrated in situ generation of H2O2 using electrospray, opening the door to portable or on-demand applications. As of now, no further studies have been reported in this area, indicating untapped potential for future experimental work.
(1) The thermodynamic instability of H2O2 in gaseous environments, which leads to spontaneous decomposition and unsustainable production (eqn (5))
| H2O2 → 2H2O + O2, ΔG < 0 | (5) |
(2) The challenge of collecting or utilizing gaseous H2O2 in situ, attributed to its high diffusivity and low concentration.
These factors, combined with the relative novelty of this approach, indicate that it remains an underexplored yet intriguing frontier in photocatalytic research.
3.3 Photocatalytic N2 fixation
Photocatalytic N2 fixation involves converting atmospheric N2 into plant-available ammonia (NH3), nitrate, or urea, contributing to renewable nitrogen energy production. By harnessing sunlight, this process demonstrates high efficiency and sustainability, which can enhance crop growth and reduce reliance on chemical fertilizers. However, conventional solid–liquid bi-phase photocatalytic systems, where the photocatalyst surface is fully immersed in water, face two significant challenges:(1) The poor solubility and slow diffusion rate of N2 severely restrict its N2 availability at the catalyst surface.
(2) The two-electron HER reactions compete for photogenerated electrons, which undermines the multi-electron transfer required for effective N2 fixation.
To overcome these challenges, recent studies have been focusing on engineering gas–liquid–solid interfaces. In this section, we discuss the positive results in photocatalytic N2 fixation under three different water states.
For example, to address the low solubility of N2 in water and the liquid-phase diffusion coefficient, Li et al. designed a floating photocatalyst (Bi/CC) by loading metallic bismuth (Bi) onto carbon cloth. During the synthesis, polyvinylpyrrolidone (PVP) was implemented as a surfactant and structure-directing agent, performing several functions, which regulated Bi crystallinity and morphology. At an optimal amount of 1.0 g, it forms regular hollow spheres with high crystallinity, the highest specific surface area, and the strongest N2 adsorption capacity. The wettability of the carbon cloth substrate is intrinsically determined by its hydrophilic or hydrophobic nature, enabling the construction of a stable gas–liquid–solid triphase interface for direct N2 supply to catalytic sites when using hydrophilic carbon cloth. While placing the photocatalyst directly in an N2 atmosphere, the carbon cloth substrate enhances photothermal heating, promoting N2 activation at the interface, and yielding an impressive rate of 2.85 mmol L−1 h−1 g−1 for NH3, which is four times higher than what is achievable in a conventional biphasic system.80 The comparison of the results from diphase and triphase systems is presented in Fig. 7a.
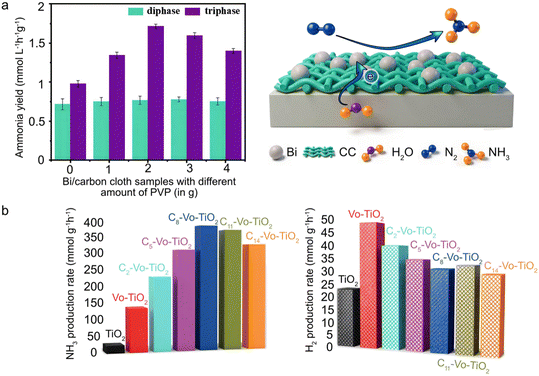 | ||
| Fig. 7 Demonstrations of photocatalytic N2 fixation in liquid water. (a) Comparison of ammonia yields from bi/carbon cloth using diphase and triphase systems, along with a schematic illustration of the photocatalytic N2 reduction reaction at a gas–liquid–solid interface. Reprinted with permission from ref. 80. Copyright 2022, Royal Society of Chemistry; (b) NH3 and H2 production rates achieved using alkyl-acid-modified TiO2 photocatalysts, highlighting the influence of carbon chain length on selectivity and activity. Reprinted with permission from ref. 23. Copyright 2023, Tsinghua University Press. | ||
Liu's team developed a hydrophilic–hydrophobic structure by depositing hydrophilic Bi4O5Br2 onto a hydrophobic ZIF-8 surface. This tri-phase interface, ensured by the wettability contrast, promoted a rapid N2 supply and effective capture of photogenerated electrons, resulting in 327 μmol L−1 h−1 g−1 of NH3, which is 3.6 times greater than that of Bi4O5Br2 alone.81
In a separate study, Guan et al. modified the surface of defective TiO2 by using alkyl acids with different carbon chain lengths. This optimization enhanced the catalyst's wettability and established a gas–liquid–solid tri-phase interface increasing N2 adsorption at oxygen-vacancy sites while suppressing the HER. The NH3 production rate for n-octanoic acid-deficient TiO2 reached 392 μmol g−1 h−1.23 The influence of wettability adjustment on the production yields and selectivity is summarized in Fig. 7b.
For example, Zare's group used an ultrasonic spray to produce microdroplets of water mixed with N2 and CO2 over a solid-phase graphite carrier network loaded with CuBi2O4 for urea synthesis. They achieved 2.8 ± 0.3 μM concentration detected by real-time mass spectrometry.54 In this system, the microdroplets act as both a transport medium for gaseous reactants and as proton/electron donors for urea synthesis. By replacing CuBi2O4 with an N2-selective photocatalyst under illumination, it is possible to reach similar or even enhanced N2 fixation efficiency. Moreover, we hypothesize that N2 fixation could also be realized by using a similar system to the one developed by Zhang et al. for microdroplet-coupled photocatalytic H2O2 synthesis or CO2RR.51
Following this development, Meng's group constructed a tri-phase interfacial system for photocatalytic NH3 production by immobilizing TiO2 on hydrophilic carbon paper, and exposing it to N2.82 This system achieved an NH3 production rate of 360.37 μmol g−1 h−1, which is 21 times higher than the yield from a conventional bi-phase interfacial system. During outdoor sunlight tests, the system produced NH3 at a rate of 73.65 μmol g−1 h−1 demonstrating the potential for large-scale photocatalytic N2 fixation. Using a related strategy, Lu's group coupled metal-phthalocyanine-derived porous organic frameworks (POFs) with a novel gas-dominated interface.83 This system overturns the traditional “gas in solvent” three-phase reaction mode and creates a “solvent in gas” system, with the reaction gas (such as N2) as the dominant environment. Specifically: the photocatalyst is directly placed in the flowing nitrogen gas, while the proton source (such as water/methanol) is ultrasonically atomized and uniformly suspended in the gas atmosphere in the form of micro-nano droplets. This design brings two core advantages. On the one hand, enhanced mass transfer: the diffusion coefficient of nitrogen gas in the gas phase is nearly three orders of magnitude higher than in the liquid phase, enabling the porous framework to efficiently adsorb and enrich high-concentration N2 molecules; on the other hand, inhibition of side reactions: the contact between the catalyst surface and the bulk solvent is significantly limited, effectively suppressing the competitive hydrogen evolution reaction caused by excess water. This strategy increases the ammonia production rate of photocatalysis to over 1820.7 μmol g−1 h−1, which is more than eight times higher than the traditional system, and the apparent quantum efficiency at 400 nm is 0.5%. These impressive results are shown in Fig. 8.
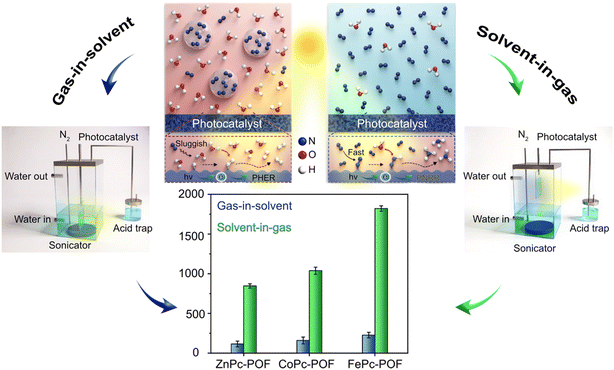 | ||
| Fig. 8 Schematic illustration of experimental setups and reaction mechanisms using conventional gas-in-solid and advanced solid-to-gas systems, indicating the significant increase in photocatalytic NH3 synthesis. Reprinted with permission from ref. 83. Copyright 2023, Wiley-VCH GmbH. | ||
3.4 Summary
As systematically demonstrated in section 3, phase-interface engineering through the control of water's physical state is a practically-oriented strategy to overcome the mass-transport, selectivity, and charge-dynamics challenges of conventional photocatalysis. Through rational interface design and optimized material combinations, researchers have achieved significant breakthroughs in three critical application areas.In CO2RR, tri-phase interfaces in aqueous systems dramatically boost CO2 availability at active sites while simultaneously suppressing the HER and steering selectivity toward valuable products. Microdroplet reactors offer dual advantages: (1) local CO2 enrichment and (2) intense interfacial electric fields that promote charge separation. Remarkably, gas–solid interfaces under humid conditions enable ultra-selective synthesis of target chemicals with ultra-high selectivity, demonstrating the transformative potential of phase engineering.
In H2O2 synthesis, phase-engineered systems, including floating photocatalysts, microdroplets, or vapor interfaces raise local O2 concentration, which inhibits H2O2 decomposition. These advantages improve selectivity, increase yields, suppress the HER, and optimize charge-carrier dynamics, moving photocatalytic H2O2 synthesis closer to large-scale practical applications.
For N2 fixation, floating and wettability-tuned catalysts in liquid water create tri-phase interfaces that facilitate direct delivery of N2 from the gas phase, while suppressing the HER. Vapor-phase interfaces further leverage photothermal and hydrovoltaic effects to dramatically improve charge separation efficiency. While microdroplet-mediated N2 fixation remains unexplored, its inherent ability to concentrate gaseous reactants and amplify interfacial fields presents an exciting research frontier. Notably, the photocatalytic synthesis of higher-value nitrogen compounds (urea, nitrates) through phase interface regulation remains completely unreported – a promising area for future research aimed at advancing photocatalytic N2 fixation.
Across all these reactions, phase-interface modulation consistently improves photocatalytic performance by (1) ensuring effective gas-phase supply of CO2, O2, or N2; (2) suppressing the HER; (3) regulating product selectivity; and (4) enhancing photogenerated charge separation and utilization.
4 Characterization methods for probing mechanisms at phase interfaces
Understanding the reasons behind the enhanced photocatalytic performance at water-state-modulated phase interfaces requires moving beyond conventional solid–liquid catalyst characterization. The unique interfacial environment conditions, including intense spontaneous electric fields in microdroplets, localized reactant enrichment at triphase boundaries, and synergistic photothermal/hydrovoltaic effects at gas–solid interfaces, demand the choice of adequate characterization techniques. This section provides a systematic overview of the experimental and theoretical methods that have proven to be the most powerful for resolving structure–activity relationships and dynamic mechanisms at these advanced interfaces, providing a toolbox for future research in this field.46–484.1 Probing interfacial structure and microenvironment
First of all, it is important to reveal the morphology and physicochemical properties of the interfaces. Scanning electron microscopy (SEM) and transmission electron microscopy (TEM) remain the standards for visualizing the microstructure, including the morphology of floatable PS-sphere-supported composites and the distribution of active sites.15,19 X-ray photoelectron spectroscopy (XPS) complements morphological analysis by resolving the oxidation states and chemical bonding environments of surface species. Contact angle measurements, in turn, quantitatively define the wettability of catalyst surfaces, directly correlating hydrophobicity/hydrophilicity with the ability to form stable gas–liquid–solid triphase interfaces, as demonstrated in studies on alkyl-acid-modified TiO2 (ref. 23) and Janus fiber membranes.21Brunauer–Emmett–Teller (BET) surface area analysis evaluates the porosity of scaffold materials (e.g., carbon cloth, porous frameworks), which governs gaseous reactant adsorption and diffusion in vapor-phase or “solvent-in-gas” (SIG) systems.83 For microdroplet systems specifically, dynamic light scattering (DLS) is a commonly used tool to determine the droplet size distribution, which is a key parameter influencing interfacial curvature and field strength.52,55
4.2 Elucidating charge carrier dynamics
Prior to the investigation of the charge behavior, it is critical to understand whether an interfacial electric field exists and to quantify its magnitude, since such fields are the primary drivers of enhanced separation in microdroplet systems. This could be evaluated by Vibrational Stark effect spectroscopy, particularly using surface-enhanced Raman scattering (SERS) with molecular probes (e.g., 4-nitrobenzenethiol or SCN−). This is a powerful tool to directly measure and map the immense electric field (∼108–109 V m−1) at the air–water interface of microdroplets.30Steady-state and time-resolved photoluminescence (PL) spectroscopy are great methods to reveal the efficiency of electron–hole pair separation and their recombination lifetimes. Significant PL quenching and prolonged lifetimes often indicate effective charge separation facilitated by interfacial fields or heterojunctions, as could be seen in S-scheme TiO2/Bi2O3 systems.19 Transient photocurrent response and electrochemical impedance spectroscopy (EIS) performed on electrode-immobilized catalysts provide insights into charge transfer resistance and conductivity at the phase interface.65,79
For processes occurring on the ultrafast timescale, femtosecond transient absorption (fs-TA) spectroscopy is indispensable. By tracking the ultrafast kinetics of photogenerated charge carriers on picosecond to nanosecond timescales, fs-TA promotes the understanding of the initial separation events driven by interfacial electric fields in microdroplet systems.19,61
4.3 Monitoring reaction dynamics and intermediates
Identifying the molecular intermediates that are formed and transformed at the phase interface under operating conditions is essential for establishing the mechanisms. In situ Fourier-transform infrared (FTIR) or diffuse reflectance infrared Fourier-transform spectroscopy (DRIFTS) both allow real-time observation of adsorbed reactants and key reaction intermediates (e.g., *COOH for CO2 reduction, *N2H for N2 fixation) on the catalyst surface.19,83 Isotope tracer experiments (e.g., using 13CO2 or 15N2) coupled with mass spectrometry (MS) or nuclear magnetic resonance (NMR) are definitive methods to confirm the origin of products, ruling out carbon or nitrogen sources from contaminants, as rigorously applied in studies on CO2 reduction and N2 fixation.52,83Experimental observations are most powerfully interpreted when supported by theoretical modeling. Thus, density functional theory (DFT) calculations simulate adsorption energies, reaction pathways, and activation barriers at model interfaces. DFT is essential for verifying the role of interfacial fields in lowering energy barriers and predicting catalyst design principles.19,51,71
4.4 Quantifying macroscopic system effects
The unique energy conversion pathways in water-state-driven systems (particularly photothermal heating and hydrovoltaic potential generation) require dedicated macroscopic characterization tools. Infrared thermal imaging, in this regard, provides spatially resolved temperature maps of catalyst surfaces under illumination, directly quantifying the photothermal contribution in systems such as Bi/carbon cloth and enabling its correlation with catalytic activity.80 Open-circuit voltage and short-circuit current measurements, in turn, directly quantify the electrical output from hydrovoltaic or evaporation-induced potential generation, as exemplified in PAA/CoO-NC and related systems.64,65Reliable quantification of catalytic output requires the appropriate analysis for each target product. Gas chromatography (GC), high-performance liquid chromatography (HPLC), and ion chromatography (IC) represent the standard techniques for quantifying gaseous and liquid products (e.g., CO, CH4, H2O2, NH4+).51,83 Long-term cyclic tests coupled with post-reaction characterizations (XRD, XPS) assess the stability and extended cycling tests of the interfacial systems.19,51
Together, this integrated multi-modal characterization framework provides the analytical foundation to bridge the gap between macroscopic performance metrics and microscopic interfacial mechanisms, guiding the rational design of next-generation phase-engineered photocatalytic systems in Fig. 9.
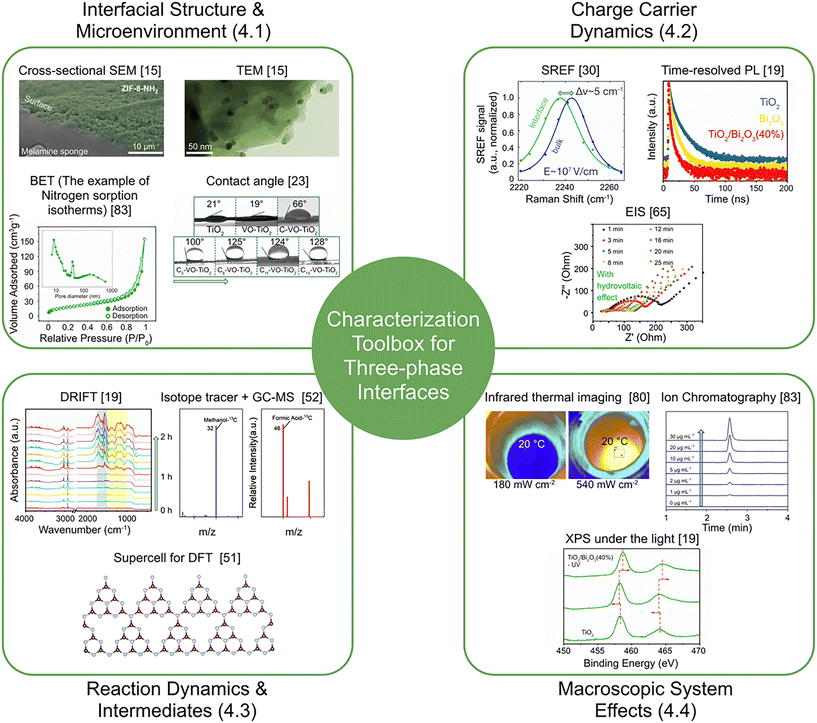 | ||
| Fig. 9 Multi-modal characterization framework for probing mechanisms at water-state-modulated phase interfaces, summarizing the methods introduced in subsections. Reprinted with permission from ref. 15. Copyright 2025, Springer Nature; reprinted with permission from ref. 19. Copyright 2022, Wiley-VCH GmbH; reprinted with permission from ref. 23. Copyright 2023, Tsinghua University Press; reprinted with permission from ref. 30. Copyright 2020, American Chemical Society; reprinted with permission from ref. 51. Copyright 2023, The Royal Society of Chemistry; reprinted with permission from ref. 52. Copyright 2023, Wiley-VCH GmbH; reprinted with permission from ref. 65. Copyright 2023, Springer Nature; reprinted with permission from ref. 80. Copyright 2022, The Royal Society of Chemistry; reprinted with permission from ref. 83. Copyright 2023, Wiley-VCH GmbH. | ||
5 Summary and outlook
5.1 Summary
In this review article, we demonstrate that controlling water states to modulate phase interfaces is an effective strategy for enhancing the efficiency of photocatalytic reactions involving Earth's abundant small gaseous molecules.In liquid water, designing floating photocatalysts and adjusting their hydrophobicity creates gas–liquid–solid tri-phase interfaces. This approach addresses challenges such as light shielding, scattering, and the low solubility and slow diffusion rates of gaseous molecules in conventional biphasic systems.
For microdroplets, dispersing photocatalysts within droplets or spraying them onto carriers leverages the intense electric field at the droplet surface. This approach drives photogenerated charge separation overcoming the limitations of conventional interfaces.
In water vapor, photocatalysts are often combined with photothermal materials that float on the water surface to generate vapor through the photothermal effect. The can also be suspended in a vapor atmosphere with reactive small molecules.
These innovative solutions provide a robust platform for optimizing photocatalytic reaction kinetics, opening new opportunities for industrial applications in the environmental and energy sectors.
5.2 Future outlook
Despite significant progress, the implementation of novel interfaces for photocatalytic applications remains challenging. Below are key issues and future perspectives:Conflicts of interest
The authors declare no conflict of interest.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This study was mainly supported by the National Natural Science Foundation of China (Grant No. 22308223), the High-level Talents launching Project (RCZK202328), and Xinjiang Uygur Autonomous Region Tianchi Talent Introduction Program (Young Doctor) for Bin Yang.References
- D. Cheng, Solar and wind, Nat. Clim. Change, 2023, 13, 1020–1020 CrossRef.
- R. Li, Photocatalytic nitrogen fixation: An attractive approach for artificial photocatalysis, Chin. J. Catal., 2018, 39, 1180–1188 Search PubMed.
- H. Jiao, C. Wang, L. Xiong and J. Tang, Insights on carbon neutrality by photocatalytic conversion of small molecules into value-added chemicals or fuels, Acc. Mater. Res., 2022, 3, 1206–1219 Search PubMed.
- A. Fujishima and K. Honda, Electrochemical photolysis of water at a semiconductor electrode, Nature, 1972, 238, 37–38 Search PubMed.
- H. Nishiyama, T. Yamada, M. Nakabayashi, Y. Maehara, M. Yamaguchi, Y. Kuromiya, Y. Nagatsuma, H. Tokudome, S. Akiyama, T. Watanabe, R. Narushima, S. Okunaka, N. Shibata, T. Takata, T. Hisatomi and K. Domen, Photocatalytic solar hydrogen production from water on a 100-m2 scale, Nature, 2021, 598, 304–307 CrossRef CAS PubMed.
- Y. Zhao, C. Ding, J. Zhu, W. Qin, X. Tao, F. Fan, R. Li and C. Li, A hydrogen farm strategy for scalable solar hydrogen production with particulate photocatalysts, Angew. Chem., Int. Ed., 2020, 59, 9653–9658 CrossRef CAS PubMed.
- L. Liu, J. Hu, Z. Ma, Z. Zhu, B. He, F. Chen, Y. Lu, R. Xu, Y. Zhang, T. Ma, M. Sui and H. Huang, One-dimensional single atom arrays on ferroelectric nanosheets for enhanced CO2 photoreduction, Nat. Commun., 2024, 15, 305 Search PubMed.
- H. Li, Y. Zhang, J. Li, Q. Liu, X. Zhang, Y. Zhang and H. Huang, Boosting H2O2 evolution of CdS via constructing a ternary photocatalyst with earth-abundant halloysite nanotubes and NiS co-catalyst, Chin. J. Catal., 2025, 69, 111–122 CrossRef.
- Z. Zhu, J. Hu, C. Hu, Y. Lu, S. Chu, F. Chen, Y. Zhang and H. Huang, Oriented crystal polarization tuning bulk charge and single-site chemical state for exceptional hydrogen photo-production, Adv. Mater., 2024, 36, 2411339 CrossRef CAS PubMed.
- Q.-S. Huang, C. Chu, Q. Li, Q. Liu, X. Liu, J. Sun, B.-J. Ni and S. Mao, Three-phase interface construction on hydrophobic carbonaceous catalysts for highly active and selective photocatalytic CO2 conversion, ACS Catal., 2023, 13, 11232–11243 CrossRef CAS.
- H. Zhou, X. Sheng, Z. Ding, X. Chen, X. Zhang, X. Feng and L. Jiang, Liquid–liquid–solid triphase interface microenvironment mediates efficient photocatalysis, ACS Catal., 2022, 12, 13690–13696 CrossRef CAS.
- C. He, L. Shang, H. Zhu, L. Yu, L. Wang and J. Zhang, Photocatalytic conversion of methane to ethanol at a three-phase interface with concentration-matched hydroxyl and methyl radicals, J. Am. Chem. Soc., 2024, 146, 11968–11977 CrossRef CAS PubMed.
- Y. Guo, Y. Dong, B. Liu, B. Ni, C. Pan, J. Zhang, H. Zhao, G. Wang and Y. Zhu, Effective H2O2 photosynthesis in gas–liquid–solid triphase system with self-floating conjugated organic polymers, Adv. Funct. Mater., 2024, 34, 2402920 CrossRef CAS.
- Y. Xie, M. Wang, Q. Huang, Q. Huang, B. Sheng, W. Song, H. Sheng and J. Zhao, A floatable photocatalyst to synergistically promote CO2 reduction and water oxidation by creating oriented charge separation across a tri-phase interface, Energy Environ. Sci., 2024, 17, 4725–4734 RSC.
- J. Wang, J. Zhang, Y. Li, X. Xia, H. Yang, J.-H. Kim and W. Zhang, Silver single atoms and nanoparticles on floatable monolithic photocatalysts for synergistic solar water disinfection, Nat. Commun., 2025, 16, 981 CrossRef CAS PubMed.
- L. Li, L. Xu, Z. Hu and J. C. Yu, Enhanced mass transfer of oxygen through a gas–liquid–solid interface for photocatalytic hydrogen peroxide production, Adv. Funct. Mater., 2021, 31, 2106120 CrossRef CAS.
- S. Yan, Y. Li, X. Yang, X. Jia, J. Xu and H. Song, Photocatalytic H2O2 generation reaction with a Benchmark Rate at air-liquid-solid joint interfaces, Adv. Mater., 2024, 36, 2307967 CrossRef CAS PubMed.
- W. H. Lee, C. W. Lee, G. D. Cha, B.-H. Lee, J. H. Jeong, H. Park, J. Heo, M. S. Bootharaju, S.-H. Sunwoo, J. H. Kim, K. H. Ahn, D.-H. Kim and T. Hyeon, Floatable photocatalytic hydrogel nanocomposites for large-scale solar hydrogen production, Nat. Nanotechnol., 2023, 18, 754–762 CrossRef CAS PubMed.
- B. He, Z. Wang, P. Xiao, T. Chen, J. Yu and L. Zhang, Cooperative coupling of H2O2 production and organic synthesis over a floatable polystyrene-sphere-supported TiO2/Bi2O3 S-scheme photocatalyst, Adv. Mater., 2022, 34, 2203225 Search PubMed.
- Z. Zhang, Y. Wang, Y. Xie, T. Tsukamoto, Q. Zhao, Q. Huang, X. Huang, B. Zhang, W. Song, C. Chen, H. Sheng and J. Zhao, Floatable artificial leaf to couple oxygen-tolerant CO2 conversion with water purification, Nat. Commun., 2025, 16, 274 CrossRef CAS PubMed.
- Y. Li, Z. Pei, D. Luan and X. W. D. Lou, Triple-phase photocatalytic H2O2 production on a Janus fiber membrane with asymmetric hydrophobicity, J. Am. Chem. Soc., 2024, 146, 3343–3351 CrossRef CAS PubMed.
- X. Sheng, Z. Liu, R. Zeng, L. Chen, X. Feng and L. Jiang, Enhanced photocatalytic reaction at air–liquid–solid joint interfaces, J. Am. Chem. Soc., 2017, 139, 12402–12405 CrossRef CAS PubMed.
- R. Guan, X. Cheng, Y. Chen, Z. Wu, Z. Zhao, Q. Shang, Y. Sun and Z. Sun, Wettability control of defective TiO2 with alkyl acid for highly efficient photocatalytic ammonia synthesis, Nano Res., 2023, 16, 10770–10778 CrossRef CAS.
- M. Sun, X. Wang, Y. Li, H. Pan, M. Murugananthan, Y. Han, J. Wu, M. Zhang, Y. Zhang and Z. Kang, Bifunctional Pd-Ox center at the liquid–solid–gas triphase interface for H2O2 photosynthesis, ACS Catal., 2022, 12, 2138–2149 Search PubMed.
- Y. Zhang, H. Zhang, F. Chang, P. Xie, Q. Liu, L. Duan, H. Wu, X. Zhang, W. Peng, F. Liu and L. Xu, In-situ responses of phytoplankton to graphene photocatalysis in the eutrophic lake Xingyun, southwestern China, Chemosphere, 2021, 278, 130489 Search PubMed.
- Z. Chen, H. Weng, C. Chu, D. Yao, Q. Li, C. Zhang and S. Mao, Nitrogen heterocyclic covalent organic frameworks for efficient H2O2 photosynthesis and in situ water treatment, Nat. Commun., 2025, 16, 6943 Search PubMed.
- M. Li, C. Boothby, R. E. Continetti and V. H. Grassian, Size-dependent sigmoidal reaction kinetics for pyruvic acid condensation at the air–water interface in aqueous microdroplets, J. Am. Chem. Soc., 2023, 145, 22317–22321 CrossRef CAS PubMed.
- K. Gong, J. Ao, K. Li, L. Liu, Y. Liu, G. Xu, T. Wang, H. Cheng, Z. Wang, X. Zhang, H. Wei, C. George, A. Mellouki, H. Herrmann, L. Wang, J. Chen, M. Ji, L. Zhang and J. S. Francisco, Imaging of pH distribution inside individual microdroplet by stimulated Raman microscopy, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2219588120 CrossRef CAS PubMed.
- H. Hao, I. Leven and T. Head-Gordon, Can electric fields drive chemistry for an aqueous microdroplet?, Nat. Commun., 2022, 13, 280 CrossRef CAS PubMed.
- H. Xiong, J. K. Lee, R. N. Zare and W. Min, Strong electric field observed at the interface of aqueous microdroplets, J. Phys. Chem. Lett., 2020, 11, 7423–7428 CrossRef CAS PubMed.
- Q. Liang, C. Zhu and J. Yang, Water charge transfer accelerates criegee intermediate reaction with H2O – radical anion at the aqueous interface, J. Am. Chem. Soc., 2023, 145, 10159–10166 CrossRef CAS PubMed.
- Y. Liu, Q. Ge, T. Wang, R. Zhang, K. Li, K. Gong, L. Xie, W. Wang, L. Wang, W. You, X. Ruan, Z. Shi, J. Han, R. Wang, H. Fu, J. Chen, C. K. Chan and L. Zhang, Strong electric field force at the air/water interface drives fast sulfate production in the atmosphere, Chem, 2024, 10, 330–351 CAS.
- S. Shaik, R. Ramanan, D. Danovich and D. Mandal, Structure and reactivity/selectivity control by oriented-external electric fields, Chem. Soc. Rev., 2018, 47, 5125–5145 RSC.
- V. V. Welborn, L. R. Pestana and T. Head-Gordon, Computational optimization of electric fields for better catalysis design, Nat. Catal., 2018, 1, 649–655 CrossRef.
- L. Xu, E. I. Izgorodina and M. L. Coote, Ordered solvents and ionic liquids can be harnessed for electrostatic catalysis, J. Am. Chem. Soc., 2020, 142, 12826–12833 CrossRef CAS PubMed.
- K. Gong, Y. Meng, R. N. Zare and J. Xie, Molecular mechanism for converting carbon dioxide surrounding water microdroplets containing 1,2,3-triazole to formic acid, J. Am. Chem. Soc., 2024, 146, 8576–8584 CrossRef CAS PubMed.
- J. Zhou, Q. Wang, G. Cheng, W. Shen, R. N. Zare and X. Sun, Charged water microdroplets enable dissociation of surrounding dioxygen, J. Am. Chem. Soc., 2025, 147, 10916–10924 CrossRef CAS PubMed.
- K. Li, B. Zhang, D. Xia, Z. Ye, Y. Pan, J. S. Francisco and Z. Mi, Room-temperature catalyst-free ammonia decomposition for hydrogen production on water microdroplets, J. Am. Chem. Soc., 2025, 147, 20417–20425 CrossRef PubMed.
- J. K. Lee, K. L. Walker, H. S. Han, J. Kang, F. B. Prinz, R. M. Waymouth, H. G. Nam and R. N. Zare, Spontaneous generation of hydrogen peroxide from aqueous microdroplets, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 19294–19298 Search PubMed.
- Z. Song, C. Liang, K. Gong, S. Zhao, X. Yuan, X. Zhang and J. Xie, Harnessing the high interfacial electric fields on water microdroplets to accelerate Menshutkin reactions, J. Am. Chem. Soc., 2023, 145, 26003–26008 CrossRef CAS PubMed.
- J. Wang, D. Huang, F. Chen, J. Chen, H. Jiang, Y. Zhu, C. Chen and J. Zhao, Rapid redox cycling of Fe(II)/Fe(III) in microdroplets during iron–citric acid photochemistry, Environ. Sci. Technol., 2023, 57, 4434–4442 Search PubMed.
- C. Gong, D. Li, X. Li, D. Zhang, D. Xing, L. Zhao, X. Yuan and X. Zhang, Spontaneous reduction-induced degradation of viologen compounds in water microdroplets and its inhibition by host–guest complexation, J. Am. Chem. Soc., 2022, 144, 3510–3516 CrossRef CAS PubMed.
- L. Zhao, X. Song, C. Gong, D. Zhang, R. Wang, R. N. Zare and X. Zhang, Sprayed water microdroplets containing dissolved pyridine spontaneously generate pyridyl anions, Proc. Natl. Acad. Sci. U. S. A., 2022, 119, e2200991119 CrossRef CAS PubMed.
- X. Yuan, D. Zhang, C. Liang and X. Zhang, Spontaneous reduction of transition metal ions by one electron in water microdroplets and the atmospheric implications, J. Am. Chem. Soc., 2023, 145, 2800–2805 CrossRef CAS PubMed.
- H. Chen, R. Wang, J. Xu, X. Yuan, D. Zhang, Z. Zhu, M. Marshall, K. Bowen and X. Zhang, Spontaneous reduction by one electron on water microdroplets facilitates direct carboxylation with CO2, J. Am. Chem. Soc., 2023, 145, 2647–2652 CrossRef CAS PubMed.
- C. Han, C. Li, J. A. Yuwono, Z. Liu, K. Sun, K. Wang, G. He, J. Huang, P. V. Kumar, J. Vongsvivut, J. Cong, H. Mehrvarz, Z. Han, X. Lu, J. Pan, X. Hao and R. Amal, Nanostructured hybrid catalysts empower the artificial leaf for solar-driven ammonia production from nitrate, Energy Environ. Sci., 2024, 17, 5653–5665 Search PubMed.
- B. Yang, J. Zhao, Y. Xiong, C. Li, M. Zhang, R. D. Rodriguez and X. Jia, Vacancies engineering in ultrathin porous g-C3N4 tubes for enhanced photocatalytic PMS activation for imidacloprid degradation, Chem. Eng. J., 2024, 498, 155117 CrossRef CAS.
- Z. Wang, G. Ding, H. Huang, J. Zhang, Q. Lv, L. Shuai, Y. Ni and G. Liao, Unraveling the dipole field in ultrathin, porous, and defective carbon nitride nanosheets for record-high piezo-photocatalytic H2O2 production, eScience, 2025, 5, 100370 CrossRef.
- B. Dai, J. Guo, C. Gao, H. Yin, Y. Xie and Z. Lin, Recent advances in efficient photocatalysis via modulation of electric and magnetic fields and reactive phase control, Adv. Mater., 2023, 35, 2210914 CrossRef CAS PubMed.
- Y. Bai, P. Luan, Y. Bai, R. N. Zare and J. Ge, Enzyme-photo-coupled catalysis in gas-sprayed microdroplets, Chem. Sci., 2022, 13, 8341–8348 RSC.
- K. Li, Q. Ge, Y. Liu, L. Wang, K. Gong, J. Liu, L. Xie, W. Wang, X. Ruan and L. Zhang, Highly efficient photocatalytic H2O2 production in microdroplets: Accelerated charge separation and transfer at interfaces, Energy Environ. Sci., 2023, 16, 1135–1145 RSC.
- Q. Ge, Y. Liu, K. Li, L. Xie, X. Ruan, W. Wang, L. Wang, T. Wang, W. You and L. Zhang, Significant acceleration of photocatalytic CO2 reduction at the gas-liquid interface of microdroplets, Angew. Chem., Int. Ed., 2023, 62, e202304189 Search PubMed.
- X. Song, C. Basheer, Y. Xia and R. N. Zare, Oxidation of ammonia in water microdroplets produces nitrate and molecular hydrogen, Environ. Sci. Technol., 2024, 58, 16196–16203 CrossRef CAS PubMed.
- X. Song, C. Basheer, Y. Xia, J. Li, I. Abdulazeez, A. A. Al-Saadi, M. Mofidfar, M. A. Suliman and R. N. Zare, One-step formation of urea from carbon dioxide and nitrogen using water microdroplets, J. Am. Chem. Soc., 2023, 145, 25910–25916 CrossRef CAS PubMed.
- Y. Chen, L. Zhang, S. Chen, S. Sun, H. Cheng, S. Li, J. Yu, B. Ding and J. Yan, Synthesis of heteromorphic Bi2WO6 films with an interpenetrate 1D/2D network structure for efficient and stable photocatalytic degradation of VOCs, Adv. Mater., 2024, 36, 2407400 CrossRef CAS PubMed.
- X. Zheng, Y.-G. Fang, M. Tan, B. Chen, J. S. Francisco, C. Zhu and C. Chu, Accelerated methane photo-oxidation at the air - water interface, J. Am. Chem. Soc., 2025, 147, 26635–26642 Search PubMed.
- J. Li, J. Xu, Q. Song, X. Zhang, Y. Xia and R. N. Zare, Methane C(sp3)–H bond activation by water microbubbles, Chem. Sci., 2024, 15, 17026–17031 RSC.
- Y. Wang, H. Shi, Y. Sun, X. Zhang, Y. Li and H. Yang, Locating organocatalysts at pickering droplet interfaces enables promotion of their catalytic enantioselectivity, J. Am. Chem. Soc., 2025, 147, 31049–31059 CrossRef CAS PubMed.
- D. Yu, F. Zhang, S. Gao, L. Wang, C. Zhang, X. Chen, X. Zhang, S. Chen, X. Fan, X. Yu and Z. Zhao, La-modified manganese-based oxides with amorphous structure: Facile synthesis and mechanistic study for boosting catalytic soot combustion, Appl. Catal., B, 2025, 377, 125516 CrossRef CAS.
- T. Suguro, F. Kishimoto, N. Kariya, T. Fukui, M. Nakabayashi, N. Shibata, T. Takata, K. Domen and K. Takanabe, A hygroscopic nano-membrane coating achieves efficient vapor-fed photocatalytic water splitting, Nat. Commun., 2022, 13, 5698 CrossRef CAS PubMed.
- J.-D. Qu, Y. Wang, T.-T. Sun, X.-Y. Chu, Y.-X. Jiang, N.-N. Zhang, Z.-H. Zhao, H. Dong, Y.-Q. Lan and F.-M. Zhang, Engineering covalent organic frameworks for photocatalytic overall water vapor splitting, Angew. Chem., 2025, 64, e202502821 Search PubMed.
- Y. Wang, W. Huang, S. Guo, X. Xin, Y. Zhang, P. Guo, S. Tang and X. Li, Sulfur-deficient ZnIn2S4/oxygen-deficient WO3 hybrids with carbon layer bridges as a novel photothermal/photocatalytic integrated system for Z-scheme overall water splitting, Adv. Energy Mater., 2021, 11, 2102452 CrossRef CAS.
- Y. Xu, S. Ai, T. Wu, C. Zhou, Q. Huang, B. Li, D. Tian and X.-H. Bu, Bioinspired photo-thermal catalytic system using covalent organic framework-based aerogel for synchronous seawater desalination and H2O2 production, Angew. Chem., 2025, 64, e202421990 CrossRef CAS PubMed.
- P. Duan, C. Wang, Y. Huang, C. Fu, X. Lu, Y. Zhang, Y. Yao, L. Chen, Q.-C. He, L. Qian and T. Yang, Moisture-based green energy harvesting over 600 hours via photocatalysis-enhanced hydrovoltaic effect, Nat. Commun., 2025, 16, 239 CrossRef CAS PubMed.
- X. Xin, Y. Zhang, R. Wang, Y. Wang, P. Guo and X. Li, Hydrovoltaic effect-enhanced photocatalysis by polyacrylic acid/cobaltous oxide–nitrogen doped carbon system for efficient photocatalytic water splitting, Nat. Commun., 2023, 14, 1759 CrossRef CAS PubMed.
- X. Xiong, Z. Wang, Y. Zhang, Z. Li, R. Shi and T. Zhang, Wettability controlled photocatalytic reactive oxygen generation and Klebsiella pneumoniae inactivation over triphase systems, Appl. Catal., B, 2020, 264, 118518 CrossRef CAS.
- Y. Tang, Z. Qin, Y. Zhong, S. Yin, S. Liang and H. Sun, Three-phase interface photocatalysis for the enhanced degradation and antibacterial property, J. Colloid Interface Sci., 2022, 612, 194–202 Search PubMed.
- H. Huang, R. Shi, Z. Li, J. Zhao, C. Su and T. Zhang, Triphase photocatalytic CO2 reduction over silver-decorated titanium oxide at a gas–water boundary, Angew. Chem., Int. Ed., 2022, 61, e202200802 CrossRef CAS PubMed.
- A. Li, Q. Cao, G. Zhou, B. V. K. J. Schmidt, W. Zhu, X. Yuan, H. Huo, J. Gong and M. Antonietti, Three-phase photocatalysis for the enhanced selectivity and activity of CO2 reduction on a hydrophobic surface, Angew. Chem., Int. Ed., 2019, 58, 14549–14555 Search PubMed.
- J.-R. Huang, W.-X. Shi, S.-Y. Xu, H. Luo, J. Zhang, T.-B. Lu and Z.-M. Zhang, Water-mediated selectivity control of CH3OH versus CO/CH4 in CO2 photoreduction on single-atom implanted nanotube arrays, Adv. Mater., 2024, 36, 2306906 CrossRef CAS PubMed.
- Y. Zhang, CO2 conversion in water microdroplets, Adv. Synth. Catal., 2025, 367, e70008 CrossRef CAS.
- J. Lin, J. He, Q. Huang, Y. Luo, Y. Zhang, W. Li, G. Zhou, J. Hu, Z. Yang and Y. Zhou, Interfacial Bi-O-C bonds and rich oxygen vacancies synergistically endow carbon quantum dot/Bi2MoO6 with prominent photocatalytic CO2 reduction into CO, Appl. Catal., B, 2025, 362, 124747 CrossRef CAS.
- Y. Ren, Y. Fu, N. Li, C. You, J. Huang, K. Huang, Z. Sun, J. Zhou, Y. Si, Y. Zhu, W. Chen, L. Duan and M. Liu, Concentrated solar CO2 reduction in H2O vapour with >1% energy conversion efficiency, Nat. Commun., 2024, 15, 4675 CrossRef CAS PubMed.
- N. Li, B. Wang, Y. Si, F. Xue, J. Zhou, Y. Lu and M. Liu, Toward high-value hydrocarbon generation by photocatalytic reduction of CO2 in water vapor, ACS Catal., 2019, 9, 5590–5602 CrossRef CAS.
- R. Wang, M. Zhang, S. Zhang, J. Zheng, Y. Zeng, Y. Yang, J. Ding, X. Wu and Q. Zhong, Self-supporting triphase photocatalytic CO2 reduction to CH3OH on controllable core–shell structure with tunable interfacial wettability, ACS Nano, 2023, 17, 24363–24373 CrossRef CAS PubMed.
- D. Liu, L. Jiang, D. Chen, Z. Hao, B. Deng, Y. Sun, X. Liu, B. Jia, L. Chen and H. Liu, Twin S-scheme g-C3N4/CuFe2O4/ZnIn2S4 heterojunction with a self-supporting three-phase system for photocatalytic CO2 reduction: Mechanism insight and DFT calculations, ACS Catal., 2024, 14, 5326–5343 CrossRef CAS.
- D. Yao, Z. Chen, C. Chu, X. Ran, X. Liu and S. Mao, Positionally isomeric conjugated polymers for photosynthesis of hydrogen peroxide through triphase interface reaction, Appl. Catal., B, 2025, 367, 125071 CrossRef CAS.
- R. He, D. Xu and X. Li, Floatable S-scheme photocatalyst for H2O2 production and organic synthesis, J. Mater. Sci. Technol., 2023, 138, 256–258 CrossRef CAS.
- B. He, C. Luo, Z. Wang, L. Zhang and J. Yu, Synergistic enhancement of solar H2O2 and HCOOH production over TiO2 by dual co-catalyst loading in a tri-phase system, Appl. Catal., B, 2023, 323, 122200 CrossRef CAS.
- Y. Li, J. Liu, Z. Sun, R. Li, L. Guo, X. Zhang, Y. Wang, Y. Wang, Z. Yu and C. Fan, Enhanced photocatalytic ammonia synthesis over a Bi/carbon cloth float: Triphase reaction system-assisted N2 supply and photothermal co-activation, Green Chem., 2022, 24, 9253–9262 RSC.
- J. Liu, R. Li, X. Zu, X. Zhang, Y. Wang, Y. Wang and C. Fan, Photocatalytic conversion of nitrogen to ammonia with water on triphase interfaces of hydrophilic-hydrophobic composite Bi4O5Br2/ZIF-8, Chem. Eng. J., 2019, 371, 796–803 CrossRef CAS.
- H.-Y. Yang, Y. Zhao, K.-L. Huang and X.-C. Meng, Photothermally catalytic fixation of N2 over TiO2 loaded onto carbon paper by fast Joule heating, Rare Met., 2025, 44, 3206–3217 CrossRef CAS.
- S. Liu, M. Wang, H. Ji, L. Zhang, J. Ni, N. Li, T. Qian, C. Yan and J. Lu, Solvent-in-gas system for promoted photocatalytic ammonia synthesis on porous framework materials, Adv. Mater., 2023, 35, 2211730 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © Institute of Process Engineering of CAS 2026 |




