Open Access Article
Open Access ArticleElectrocatalytic CO2 reduction coupled with biocatalysis for high-value products
Weili Qiao
a,
Yuyan Zhanga,
Boyang Wua,
Canyu Zhanga,
Wen-Yong Lou ab and
Yufei Cao
ab and
Yufei Cao
 *ab
*ab
aLab of Applied Biocatalysis, School of Food Science and Engineering, South China University of Technology, Guangzhou 510640, Guangdong, China. E-mail: yufeicao@scut.edu.cn
bGuangdong Province Key Laboratory for Green Processing of Natural Products and Product Safety, South China University of Technology, Guangzhou 510640, Guangdong, China
First published on 19th March 2026
Abstract
The rising concentration of carbon dioxide (CO2) has created an urgent need for technologies that can both mitigate emissions and supply sustainable carbon feedstocks. Electrocatalytic carbon dioxide reduction utilizes renewable electricity to convert CO2 into C1–C3 products at high rates under mild conditions, but it remains challenging to produce longer-chain molecules with high selectivity and efficiency. Biocatalytic systems are notable for forging C–C bonds and assembling complex products. However, most organisms fix CO2 inefficiently and purely biological routes are constrained by thermodynamics and enzyme kinetics. This review examines how integrating electrocatalysis and biocatalysis in cascade systems combines the strengths of both approaches. We discuss the historical development and motivation for these hybrids, summarize recent progress in electrocatalytic CO2/CO conversion to C1–C3 intermediates, and outline biocatalytic strategies that extend short-chain feedstocks to higher-value Cn products. We further examine challenges in catalyst stability, pathway and strain engineering, and process scale-up, and propose directions for advancing electro-biosynthetic manufacturing. Together, these developments point toward a circular carbon economy in which CO2 becomes a practical starting point for complex carbon-based products.
Keywords: Electrocatalytic CO2 reduction; Biocatalysis; Hybrid electro-biocatalytic systems; Multi-enzyme cascades; Metabolic engineering.
1 Introduction
The conversion of carbon dioxide (CO2) into value-added chemicals is both a major challenge and an important opportunity for sustainable technologies. CO2 is an abundant but low-energy carbon source, and its utilization could help mitigate greenhouse gas emissions while providing renewable feedstocks for fuels, materials, and food ingredients.1–3 In nature, photosynthetic organisms fix CO2 into biomass. However, this route is constrained by slow enzyme kinetics and low efficiency, which is typically well below 1%.4 Conventional industrial routes for CO2 utilization, such as thermocatalytic hydrogenation exemplified by Fischer–Tropsch synthesis of hydrocarbons from CO/H2, operate at high temperatures and pressures and often yield broad, poorly defined product mixtures.5,6 Over the past decade, electrocatalytic CO2 reduction (CO2RR) has emerged as a promising alternative that uses renewable electricity to convert CO2 under relatively mild conditions. Electrocatalytic systems can generate C1 and C2 products, such as carbon monoxide, formate, methane, ethylene, ethanol, and acetic acid at high reaction rates.7–9 However, as the desired carbon chain length increases (for example, C3–C5 products), purely electrocatalytic approaches face significant limitations in both selectivity and overall efficiency.10–12In contrast, biocatalytic processes such as microbial fermentation and multienzyme cascades are highly effective at converting simple carbon sources into complex and high-value products, with precise control over structure and stereochemistry.13,14 Cells and enzymes can form C–C bonds and generate long-chain or highly chiral molecules that are often difficult to obtain through conventional electrocatalytic synthetic methods. However, most organisms cannot fix CO2 at high rates, because CO2 is thermodynamically stable and key CO2-fixing enzymes such as RuBisCO and formate dehydrogenases have intrinsically low activities.15,16 Many natural CO2 fixation pathways operate near equilibrium and have slow turnover rates, which limits the efficiency of purely biological CO2 conversion.17–19
This contrast points to a clear strategy: integrating electrocatalysis and biocatalysis in a sequence of orchestrated chemoenzymatic cascades (Fig. 1). In such hybrid systems, an electrocatalytic module first reduces CO2 to simple C1–C3 intermediates, which are subsequently elongated and assembled into larger, higher-value products by biocatalysis. In this configuration, the high driving force and rapid kinetics of electrochemical conversion are harnessed while the selectivity and synthetic versatility of biological catalysis are maintained. As a result, integrated electro-biocatalytic schemes are attracting increasing attention as routes to convert waste CO2, under renewable energy input, into fuels, chemicals and even food products.
Many existing reviews discuss electrocatalytic CO2/CO reduction and the biotransformation of C1–C3 substrates in depth, but fewer offer a fully integrated, system-level perspective on electro-biocatalytic cascades. Here, we provide a system-level synthesis that explicitly connects electrocatalysis with downstream biocatalysis through the lens of intermediate selection, interface compatibility, and coupling constraints. We distinguish the biocatalytic stage into two complementary modules—cell-free enzyme cascades and metabolic engineering-based bio-conversion—and clarify how each interfaces with electrocatalytic outputs. We begin by placing the field in its historical context and outlining the motivation for coupling electrocatalytic and biocatalytic platforms, with particular emphasis on the conceptual and practical advantages of cascade architectures. We then discuss the current state of electrocatalytic CO2 reduction, focusing on recent progress in catalyst development, reactor engineering and performance metrics. Next, we discuss biocatalytic strategies for carbon chain elongation, including in vitro enzyme cascades and microbial metabolic engineering, and examine how these systems use C1 and C2 feedstocks. Representative case studies of integrated electro-biocatalytic systems are presented to illustrate the conversion of CO2 into high-value products, ranging from fuels and bulk chemicals to biopolymers and food ingredients, and to highlight the design and optimization principles that govern each module. Finally, we identify key challenges at the interface between electrocatalysis and biocatalysis and propose directions for future research.
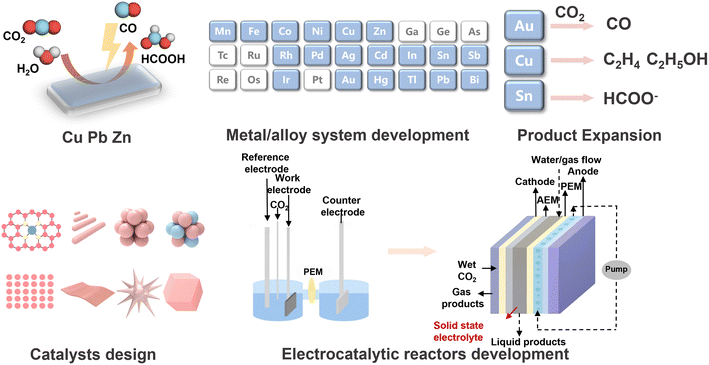
2 Background and early developments in CO2 conversion
Using CO2 as a carbon feedstock has long been viewed as a key goal on the path to carbon neutrality. Traditional strategies include high-temperature thermochemical processes, such as reforming and Fischer–Tropsch synthesis, and biological CO2 fixation through natural photosynthesis or microbial fermentation driven by hydrogen or other energy sources.1,20–23 However, these routes either demand intensive energy input or suffer from low efficiencies. Research on electrocatalytic CO2 reduction (CO2RR) has a long history and has undergone continuous development and refinement (Fig. 2). Early studies date back to the nineteenth century, when CO2 was reduced to CO at Zn cathodes. Interest increased in the 1980s, after the oil crises of the 1970s, when systematic work on Cu, Zn and lead electrodes identified formate as a dominant product. Based on these results, CO2RR was recognized by the end of the 20th century as a promising way to convert CO2 into high-value products, ideally from renewable sources, under ambient conditions. Collectively, this work marked the beginning of systematic metal and alloy systems development for CO2RR. Pioneering work by Hori and collaborators in the 1980s and 1990s further showed that Cu electrodes can reduce CO2 to multi-carbon products such as ethylene and ethanol, although only at modest yields. In contrast, metals such as Au and Ag mainly produce CO, and metals such as Sn tend to form formate.24–26 These studies clarified the fundamental reaction pathways and established the unique ability of Cu to promote C–C bond formation. By the 2010s, advances in catalyst design and cell architecture had significantly increased reaction rates and selectivity, bringing CO2RR much closer to practical application.27–30 For example, gas diffusion electrode (GDE) reactors have enabled current densities that are orders of magnitude higher than those in conventional H cells by alleviating limitations in CO2 mass transfer.31On the biocatalysis side, the concept of microbial electrosynthesis began to emerge around 2010. Several groups reported that acetogenic bacteria growing on cathodes could directly accept electrons, or hydrogen generated in situ, and reduce CO2 to acetate and other small organic molecules.32–34 This direct coupling of electrical current to living cells showed that bio-based CO2 reduction is feasible, but precise control of the process and achievement of high rates remains challenging. Indirect coupling through soluble intermediates emerged in 2012, when Li et al. reported an integrated electro-microbial process that converted CO2 into liquid fuels using a two-step strategy.35 In that study, CO2 was first reduced electrochemically to formate, and an engineered strain of Ralstonia eutropha, a natural autotroph, was then used to produce isobutanol and 3-methyl-1-butanol. The strain was engineered with a synthetic isobutanol biosynthetic pathway, thereby enabling electrical energy to be stored in liquid fuel molecules. Formate served as a clean energy carrier that circumvented the handling and solubility challenges associated with gaseous hydrogen in fermentation, while the biocatalyst directed carbon into a specific, high-energy-density fuel. This work provided a clear proof of concept that combining an electrolyzer with a bioreactor can help offset the limitations of each unit. The success of Li et al. stimulated many subsequent efforts to integrate electrocatalysis with fermentation.35 Given the close interplay between microbial and enzymatic systems, electro-enzymatic CO2 reduction emerged around the same time and largely in parallel with microbial electrosynthesis.36 In 2014, Srikanth et al. presented an approach for electrochemical CO2 reduction to formate catalyzed by formate dehydrogenase immobilized on an electrode.37 Besides CO2 reduction, NADH is regenerated in the electrochemical system at the same time. Also, Cai et al. have realized the potential of integrating enzymes with electrocatalysis. In their work, they show the electrochemical addressing of dehydrogenase enzymes without the requirement of any cofactor. In this electro-enzymatic system, CO2 is reduced to ethylene (C2H4) and propene (C3H6) by a single VFe nitrogenase.38
Over the 2010s, many studies employed electrochemically produced small molecules as substrates for microbial production of ethanol, butanol, fatty acids, and bioplastics.35,39–42 Multi-enzymatic cascade reaction provides a new avenue for C–C coupling directly from CO2 under mild conditions.43 A representative example of electro-driven, C–C coupling from CO2 in vitro is the four-enzyme cascade developed for ethylene glycol (EG) synthesis. In this design, formate dehydrogenase (PaFDH) and formaldehyde dehydrogenase (BmFADH) generate C1 intermediates from CO2, which are then coupled via glycolaldehyde synthase (PpGALS) to form a C2 backbone, followed by alcohol dehydrogenase (GoADH) to yield EG. To continuously supply reducing equivalents under mild conditions, the authors integrated a rhodium-based electrochemical NADH-regeneration electrode, achieving a Faradaic efficiency of 82.9% at ∼0.6 V (vs. Ag/AgCl) and an NADH productivity of 0.737 mM h−1.36 These efforts fit into a broader vision of artificial photosynthesis, in which human-engineered systems aim to match and even exceed the efficiency of natural photosynthesis by separating the light-harvesting step from carbon fixation. Rather than relying on chlorophyll, these schemes employ photovoltaic devices or electrocatalysts for the light-harvesting stage, and microbes or isolated enzymes for the biosynthetic stage.44,45 A representative example of artificial photosynthesis was reported by Hann et al.46 Their team designed a spatially separated, solar-driven process to convert CO2 into food. The electrochemical module first fixes CO2 into a simple carbon substrate and then supplies this intermediate to cultures of photosynthetic algae or other microorganisms, which in turn produce starch and edible biomass. In both early and recent studies, inorganic catalysts are used for CO2 activation, whereas biological systems provide flexible and selective product formation. Over time, the scope of the field has expanded from formate-based systems to those employing multiple intermediates, and from fuel alcohols to a broad range of products, including polymers, pharmaceuticals, and food ingredients.47–49 The following sections review recent examples of each component of this tandem strategy and the current state of their integration.
2.1 Electrocatalytic conversion of CO2 to C1–C3 compounds
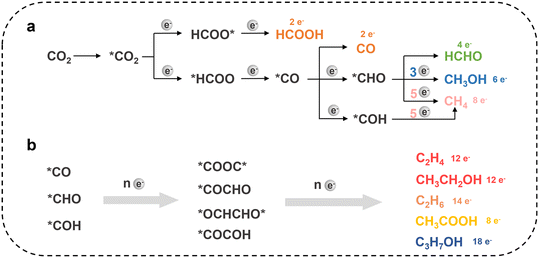
CO2RR proceeds through a sequence of coupled proton and electron transfer steps at the electrode surface, which progressively convert CO2 into more reduced carbon-containing products. The associated reaction network is highly complex (Fig. 3). The number of electrons and protons transferred determines whether CO2 is converted into a two-electron product, a six-electron product or a twelve-electron product.50–53 Each major product class follows a distinct pathway with its own characteristic surface-bound intermediates. Faradaic efficiency (FE) is a key metric for evaluating electrocatalytic performance and is influenced by the catalyst type, operating potential, current density, reactor configuration and mass-transport conditions.CO is typically generated via a two-electron reduction pathway. CO2 first adsorbs on the catalyst and forms a *COOH intermediate, and then loses an oxygen atom to produce CO. The rate-limiting step is often the formation of *COOH and the subsequent desorption of CO. Catalysts such as Au and Ag bind the *COOH strongly enough to form it but weakly enough to release CO, which makes them highly selective for CO.54,55 As a result, FE above 90% on Ag and Au are common, particularly in flow reactors.56,57 Formate/formic acid is another two-electron product that forms via a different intermediate, HCOO*. HCOO* is a bidentate species in which both oxygen atoms bind to the catalyst surface. Metals such as tin (Sn), bismuth (Bi), indium (In) and lead (Pb) are known to favour this pathway.58–61 In optimized systems, formate selectivities are achieved above 90%. Moreover, its direct generation in the liquid phase, typically as a dissolved anion, facilitates coupling to downstream biological processes.57 From an industrial perspective, formate and formic acid are currently among the most practical products, with some continuous flow systems delivering high current densities together with overall energy efficiencies >50%.62 Methane and methanol are also C1 products that form through a multi-electron reduction pathway.52,63,64 Methane formation proceeds via surface-bound *COOH, *CO and *COH, whereas CH3OH likely follows a route involving *COOH or dissolved hydrocarbon-like species.65–67 Transition-metal catalysts such as cobalt phthalocyanines have also been tuned to methanol, although with low yields.52,68,69 Achieving high selectivity for methanol is challenging due to competition from the hydrogen evolution reaction and the behaviour of key intermediates, which either bind too strongly and poison the surface or are further reduced to methane. In addition, maintaining the stability of the catalysts used for methanol production remains another challenge.
Cu is unique among pure metals in its ability to promote C–C bond formation, giving products such as ethylene, ethanol and even small amounts of n-propanol.70–74 Formation of multi-carbon products generally begins with adsorbed *CO or *COH on the surface, followed by coupling between two CO-derived intermediates. If CO binds too weakly, it desorbs before C–C coupling can occur, whereas if it binds too strongly, it poisons the surface. The intermediate affinity of Cu for CO is widely believed to underlie its ability to deliver appreciable yields of C2 products. Research has shown that the presence of mixed oxidation states of Cu, such as Cu0 and Cu+, and the presence of high index facets and grain boundaries can enhance C2+ formation by stabilizing key intermediates.75 The most advanced Cu-based catalysts reported so far achieve about 50–90% combined FE for C2+ products. But these C2+ products are typically obtained as mixtures of ethylene, ethanol, acetate and related species, so the selectivity for any single product remains modest.76,77 Controlling the distribution of multi-carbon products and selectively favouring one target remains difficult because the underlying reaction network contains many competing branching pathways.
A key performance metric for electrocatalysis is the overall energy efficiency (EE), defined as the fraction of electrical energy input that is stored in the chemical products. Recent progress in catalyst development and reactor engineering has substantially increased overall EE.78–81 CO and formate can reach >50%, whereas multi-carbon products usually remain at 20–60% because their formation requires higher overpotentials and competes more strongly with side reactions. To make CO2 electrolysis economically viable, efficiencies must be further increased by reducing voltage losses and enhancing product selectivity.
For the reactor, the field has evolved beyond simple H-type cells, in which two chambers are separated by a membrane, to continuous flow systems that handle gaseous CO2 more effectively. Flow cells equipped with gas diffusion electrodes (GDEs) deliver CO2 directly to the catalyst interface and can sustain high reaction rates,82 for example, fluorine-modified Cu GDE achieved a C2+ (mainly ethylene and ethanol) formation rate exceeding 4.0 mmol h−1 cm−2, with 80% FE.83 In such devices, current densities of hundreds mA cm−2, relevant to industrial operation, are routinely achieved, in contrast to the few mA cm−2 typically obtained in H-cells. One major challenge is the management of the liquid electrolyte, since flooding of the GDEs can block gas transport. Hydrophobic treatments such as polytetrafluoroethylene (PTFE) coatings are commonly used to extend GDEs' lifetimes. Existing studies have shown that periodically recoating the GDE with PTFE enables stable CO2 electrolysis at 600 mA cm−2 for about 5200 hours, corresponding to more than 7 months of continuous operation.84–86 Such durability is encouraging for scale up, as it suggests that degradation modes such as electrode flooding and catalyst deactivation can be mitigated through engineering strategies. Another important development is the membrane electrode assembly (MEA) configuration, similar to that used in fuel cells, in which humidified CO2 gas is supplied directly to the catalyst layer without a liquid catholyte.85,86 MEA reduces diffusion resistance and eliminates the need to dissolve CO2, thereby reaching higher current and efficiency. However, these systems require careful water management and typically an ion-exchange membrane to conduct ions. Recently, solid-state electrolyte reactors have attracted increasing attention. In these systems, ionic charge such as H+ or HCOO− is transported through a solid polymer or inorganic conductor, and the products can be collected as an almost pure liquid stream without mixing with electrolyte salts.87,88 Xia and co-workers demonstrated a solid-state cell that continuously converted CO2 into a 12 M formic acid solution, close to neat formic acid.88 Likewise, Zheng and co-workers used a porous solid electrolyte reactor to produce purified acetic acid. Their configuration employed thick anion exchange membranes to conduct hydroxide and suppress crossover, achieving ∼97 wt% pure acetic acid directly from the electrochemical cell.89 This is a significant advance for integration with bioprocesses, since it eliminates the need for costly downstream separation of the product from supporting electrolytes. Together, these reactors and process innovations provide a much more favourable platform for CO2 electrolysis, but the achievable rates and product distributions are still governed by the properties of the catalyst surface.
In parallel with these engineering advances, the search for more active and selective electrocatalysts has intensified. Beyond the conventional metals described above, current work focuses on alloys, nanostructured copper and well-defined molecular systems.26,90 For example, single-atom catalysts (SACs) have shown exceptional activity for some reactions. Zheng et al. employed a Ni-based single-atom catalyst on a nitrogen-doped carbon (Ni–N–C) to convert CO2 to CO with nearly 100% efficiency,89 and then used CO to drive acetate formation on a Cu catalyst. Nano-structuring Cu to create grain boundary-rich surfaces has also been reported to enhance acetate production, reaching 52% FE for acetate in CO reduction.75 Studies have also shown that oxide-derived Cu obtained by pretreating Cu2O favours C2 products such as ethylene and ethanol by maintaining a dynamic mixture of Cu+/Cu0 surface sites during operation. Bimetallic catalysts such as Cu–Ag and Cu–Sn aim to combine complementary functions, for example, using Ag domains to generate CO in situ on a Cu surface and thereby facilitate C–C coupling.91,92 For example, a Cu–Ag GDE has been reported to allow the distribution of C1, C2 and even C3 products to be tuned by adjusting the surface composition.93
One of the most challenging product classes under ambient CO2/CO electroreduction conditions is still C3+. Early reports showed only trace FE for n-propanol, allyl alcohol and related C3 products on modified Cu electrodes, and sub-percent yields for C4 species such as n-butane or 1-butanol.75,94 Recent studies have improved the performance, with systems now reaching C3+ FE of ∼56%.95,96 These advances demonstrate that selective C3+ formation is indeed feasible, although broadly extending such high efficiencies across different systems remains challenging. As the carbon chain length of the target molecule increases, achieving its efficient synthesis by electrocatalysis becomes increasingly challenging.97–99
To push beyond this limitation, some researchers have taken inspiration from thermocatalysis.100 For example, Cao et al. employed Ru nanoparticles for electrocatalytic CO reduction under elevated temperature and elevated pressure, essentially creating an electrochemical analogue of Fischer–Tropsch synthesis.101 In that work, hydrocarbons up to C21 were produced, with a 32% FE for C5+ products. Although this strategy yields long-chain hydrocarbons, it relies on conditions far from ambient and on a specialised high-pressure cell, which blurs the boundary between electrochemical and thermochemical catalysis. Under mild aqueous electrolysis conditions, extending product distributions much beyond C2 or C3 appears to be intrinsically constrained by sluggish kinetics and competition from side reactions. This is where handing off the task to biocatalysis becomes attractive.
In summary, electrocatalysis can now generate a range of C1–C3 building blocks from CO2/CO. CO and formate can be produced with high EE values, while ethanol, acetate and ethylene can be obtained in appreciable yields. Advances in reactor design have also enabled these processes to operate at industrially relevant current densities with improved stability. In some cases, they can run continuously for thousands of hours. However, the selectivity challenge intensifies for each additional carbon added electrochemically. The cascade strategies address this limitation by ending the electrosynthetic step at an optimal intermediate, such as formate or acetate, which can be produced in a relatively clean form, and then using biological systems to perform the downstream C–C bond-forming steps with high fidelity.
2.2 Biocatalytic carbon chain elongation from C1/C2 to Cn products
Biological systems are adept at C–C bond formation and complex molecular synthesis, as illustrated by the diversity of natural metabolites ranging from sugars and fatty acids to polyketides.102,103 Currently, two broad biocatalytic platforms can be employed, including multi-enzyme cascades in vitro and engineered microorganisms operating in vivo. Each offers distinct advantages. Cell-free enzymatic systems offer a defined environment free of cellular regulation, allowing the use of non-natural pathways and potentially higher fluxes. Microbial systems benefit from self-replication, long-term stability and the broad catalytic repertoire of living metabolism.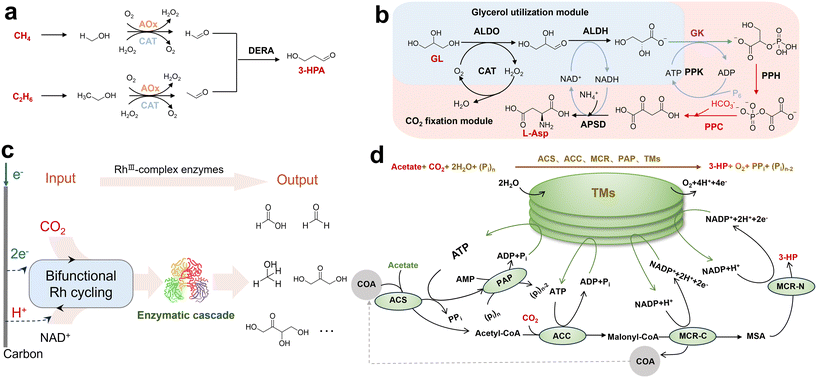 | ||
| Fig. 4 Synthesis via multi-enzyme cascades. (a) Multi-enzyme conversion of C1/C2 alcohols to 3-HPA. Methanol and ethanol are first oxidized by alcohol oxidase to formaldehyde and acetaldehyde, respectively. The resulting C1/C2 aldehydes are then condensed to 3-HPA by a C1/C2 carboligase (DERA), while the hydrogen peroxide formed during alcohol oxidation is decomposed by catalase. Reprinted with permission from ref. 107. Copyright 2025, American Chemical Society; (b) in vitro pathway for L-aspartate (L-Asp) production, comprising a glycerol utilization module coupled to a CO2 fixation module. Reprinted with permission from ref. 110. Copyright 2025, Elsevier; (c) bifunctional Rh(III) complex-driven CO2 reduction and NADH regeneration enabling direct bio-electrosynthesis of C2+ products. The metal complex [Cp*Rh(bpy)Cl]2+ (Rh(III) complex, [Rh(III)–H2O]2+) simultaneously catalyzes electrochemical CO2 reduction to formate and the reduction of NAD+ to NADH. Formate is subsequently upgraded to methanol (C1), dihydroxyacetone (C3) and L-erythrulose (C4) via tandem enzymatic cascades ACS–ACDH–ADH, ACS–ACDH–FLS and ACS–ACDH–FLS–FSAA129S. Reprinted with permission from ref. 118. Copyright 2024, American Chemical Society; (d) light-powered in vitro synthetic enzymatic biosystem (ivSEB) for 3-HP production from acetate and CO2. The cascade involves ACS, ACC, MCR-C, MCR-N, PAP, AMP and MSA to achieve stepwise conversion of acetate to 3-HP. Reprinted with permission from ref. 119. Copyright 2024, American Chemical Society. | ||
The power of computation and enzyme engineering has greatly expanded the realm of possible cascades. Researchers use pathway design algorithms to identify theoretical routes for target molecules, then assemble and optimize these pathways with available enzymes or engineered ones.108,109 A prime example is the recently reported artificial biosynthesis pathway, which converts CO2 and glycerol to L-aspartate in one pot using eight enzymes and produces 18.6 mM aspartate within 2 h (Fig. 4b).110 In another case, Ding et al. obtained lactic acid titers of 2.2–2.8 g L−1 from 100 mM methanol by carefully optimizing enzyme loadings and cofactor supply.111 These studies demonstrate that with the appropriate enzyme catalysts, C1 substrates can be channelled into central metabolites and organic acids.
Many enzymatic reductions (FDH, ADH) require NAD(P)H as electron donors, and some ligase or kinase steps require ATP.104,112,113 These cofactors are expensive and must be regenerated in situ for the process to be sustainable. Inexpensive phosphites can be oxidized to phosphates by adding enzymes such as phosphite dehydrogenase (PTDH), simultaneously reducing NADP+ to NADPH.104,105,112 Alternatively, NADH can be obtained directly at the electrode or through redox media (such as viologen or quinones) by electrochemical cofactor regeneration.114–116 This effectively links an electrical energy input to drive the biochemistry, similar to how nature uses the photosynthetic electron transport chain to generate NADPH. Researchers have constructed bio-electrocatalytic cells in which one electrode continuously supplies NADH to an enzyme cascade. For example, Wu et al. employed an electrochemical system to sustain a reductive glycine pathway (rGlyP), producing 0.81 mM glycine from CO2 and NH3.117 Li et al. developed a bifunctional Rh(III) complex that simultaneously catalyzed the reduction of CO2 to formate and the electrochemical regeneration of NADH at the electrode (Fig. 4c).118 Coupled with two downstream enzymes, this one-pot system produced dihydroxyacetone and L-erythrulose from CO2. Although the yields were low (2.63 mM DHA and 1.93 mM erythrulose), this work demonstrated a practical all-in-one route: from electrical energy to a formate intermediate, then to enzymatic C–C coupling and ultimately to higher polyols. Photochemical regeneration of cofactors offers another attractive strategy.119 By harnessing light, cofactor recycling can be driven by photoredox systems or by whole thylakoid membranes isolated from plants. Ning et al. used chloroplast thylakoid membranes to supply ATP and NADPH for the CO2-to-3-hydroxypropionate (3-HP) pathway, achieving a 92% yield (Fig. 4d).119 This approach integrates concepts from natural and artificial photosynthesis by using light-harvesting proteins to power a synthetic cycle. And several systems have eliminated specific cofactor requirements through pathway redesign. Liu et al. recently reported a chemoenzymatic route to synthesize amino acids and pyruvate from CO2 and methanol without external NADH/NADPH or ATP.120 Normally, the glycine to serine conversion requires the NAD-dependent protein. They replaced that step by using a chemical reducing agent, dithiothreitol (DTT), to keep the key enzyme in a reduced state, effectively substituting DTT for NADH in that step. With this innovation, they coupled methanol oxidation and CO2 incorporation to produce glycine, which was then converted to serine and pyruvate, achieving titers in the g L−1 range for those products.
In summary, multi-enzyme cascades offer a modular and tunable platform for upgrading C1–C2 building blocks to larger products through protein engineering, pathway design, and use of chemical or electrochemical energy inputs. These cascades are a powerful complement to electrocatalysis. Enzymatic biocatalysis enables precise control, but it often relies on purified enzymes and externally supplied cofactors or recycling systems, leading to higher cost pressure during scale-up.121 Scaling up these cascades and maintaining their efficiency outside the laboratory remains a significant challenge. Integrating them with electrochemical processes that provide a continuous driving force is one promising way to address this limitation.
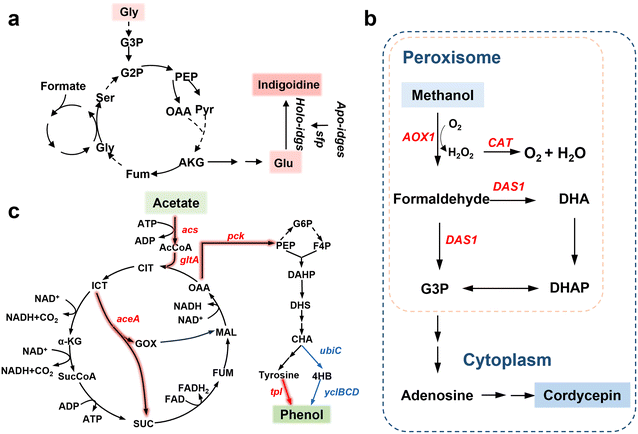 | ||
| Fig. 5 Microbial routes to product formation. (a) Engineered biosynthetic pathway for the microbial production of indigoidine. Reprinted with permission from ref. 132. Copyright 2023, Springer Nature; (b) schematic of enhanced methanol assimilation in peroxisomes and the resulting changes in cordycepin yield achieved through enzyme fusion engineering. Reprinted with permission from ref. 139. Copyright 2023, Elsevier; (c) engineering of a microbial pathway for phenol production from acetate, illustrating the underlying genetic modification strategies. Reprinted with permission from ref. 143. Copyright 2025, American Chemical Society. | ||
Methanol is an attractive substrate for biotechnology. It is a liquid and energy-dense substrate that can be produced via syngas-based routes or electrochemical methods.134–136 Pichia pastoris is widely used industrially for recombinant protein production on methanol-based feed.133 Synthetic biology has enabled even non-methylotrophic organisms to utilize methanol by transplanting or imitating these pathways.137,138 E. coli and S. cerevisiae have been engineered to co-consume methanol and sugars by expressing methanol dehydrogenases together with downstream assimilation enzymes, although robust growth on methanol as the sole carbon source remains difficult to achieve. In parallel, methylotrophic yeasts have been engineered to convert methanol into a variety of valuable chemicals. Zhao et al. reprogrammed Pichia pastoris to produce cordycepin, a nucleoside drug precursor, achieving titers of 1.55 g L−1 in shake flasks and 8.11 g L−1 in fed-batch fermentation (Fig. 5b).139 Similarly, Niu et al. engineered Pichia pastoris to produce a complex plant secondary metabolite.140 These examples show that methanol-fed bioproduction can reach g L−1 levels for high-value products. A significant metabolic challenge is the toxicity of formaldehyde, a key intermediate in methanol metabolism. Cells must keep the formation and consumption of formaldehyde in a tight balance. When engineering methanol/formate-utilization pathways, it is therefore often necessary to increase downstream flux so that formaldehyde is rapidly converted into product to enhance detoxification and tolerance mechanisms to prevent its accumulation.
CO and CH4 are gaseous substrates that some microbes can utilize.141,142 Their low solubility in water poses a practical challenge for bioreactor operation. To address this, innovative reactor designs are being developed to enhance gas–liquid mass transfer. Ma et al. constructed a hollow-fiber membrane bioreactor that efficiently delivers CH4 to methanotrophic cultures, enabling high cell densities and single-cell protein production from methane with a yield of 1.36 g biomass per g methane and a protein content of 67% w/w.142 Such designs could likewise facilitate CO utilization by providing a large surface area for gas exchange. Acetate and ethanol are liquid substrates that are broadly compatible with microbial metabolism. Acetate can be taken up and converted to acetyl-CoA, although high concentrations of acetic acid can become inhibitory by lowering the pH.128,143–146 Ethanol is similarly converted to acetyl-CoA via alcohol dehydrogenase and acetaldehyde dehydrogenase in many organisms. Because of their simplicity in metabolism, a wide range of products can be made with appropriate metabolic engineering. Feng et al. engineered E. coli to produce phenol from acetate by introducing the tyrosine-to-phenol pathway (Fig. 5c).143 Phenol is an aromatic chemical normally derived from petroleum, but here they achieved up to 2.01 g L−1 phenol using fed-batch culture with in situ extraction. And Qian et al. reassembled a plant flavonoid pathway in Pichia pastoris, enabling the yeast to convert simple carbon sources into 4′-deoxyflavones.128 They engineered synthetic regulatory circuits to balance the pathway, achieving high-level production of baicalein. While that particular demonstration used glucose as the carbon source in the published work, the authors noted the host could also run on methanol, illustrating the potential to produce complex natural products from C1 feeds in the future.
In all of these microbial systems, metabolic engineering is critical. It involves introducing new enzymes, deleting competing pathways and installing dynamic control circuits. In a representative study on acetate-to-flavone biosynthesis, the researchers divided the pathway into modules and used synthetic transcription factors to control each module, ensuring that precursors were supplied with appropriate ratios and timing for efficient synthesis.128 Comparable levels of control are likely to be necessary when integrating unfamiliar feedstocks into hosts that do not naturally metabolize them. In electro-bio cascades, microbes act as versatile biocatalysts that take the relatively small set of molecules produced by electrolyzers and convert them into a wide variety of products through their metabolic networks.147 They also enable chain elongation beyond C3/C4 via polymerization or iterative pathways, giving access to biopolymers and larger fuels that are difficult to obtain by electrocatalysis alone.148–151 Recent work has shown that yeast can convert acetate into sugars and even starch polymers, and that bacteria can upgrade formate into C4 products.152,153
3 Design of the coupled electro-biocatalysis cascade system
Integrating an electrochemical reactor with a bioprocess is non-trivial. The two systems have very different operational requirements, and direct interfacing can lead to mutual interference if not properly managed. Here, we outline key design considerations for effective coupling (Fig. 6) and then highlight illustrative case studies where these principles are applied.3.1 Key factors for effective coupling
Choosing an appropriate bridging intermediate, the electrocatalytic product that feeds the biocatalytic step, is critical. Intermediate selection in electro-biocascades should be tailored to the target product and the downstream module, and should be made by jointly considering electrolyzer performance, interface compatibility, and biocatalytic assimilation requirements. This intermediate should be produced at a high rate and efficiency by the electrocatalytic module and be readily and selectively consumed by the biological module. Generally, liquid products are preferred over gaseous products (CO and H2). From a technological maturity standpoint, CO2RR to formate/formic acid is currently among the most industrially feasible routes, largely because it offers markedly higher selectivity and formation rates than C2+ products. By contrast, liquid products such as methanol, ethanol, and acetate have higher energy density and are more readily utilized as carbon feedstocks for downstream bioconversion, yet further improvements in their selectivity and yields remain necessary.Catalyst and reactor stability are also critical. A typical fermentation might run for a few hours to several days.132 Therefore, the electrocatalyst must be able to operate stably over comparable durations. Many laboratory-scale CO2 reduction catalysts, particularly nanostructured materials, lose activity within tens of hours because of sintering, poisoning and carbonate formation in the electrolyte. For practical integrated systems, catalysts need to maintain high activity and selectivity over extended periods. Strategies to enhance stability include using more robust materials, modifying the catalyst surface, forming alloys, and continuously regenerating the active surface, for example, by applying potential pulses or adding ions that promote surface healing.60,154–156
In a single reactor where microbes and electrodes co-exist, the reaction conditions must suit both. This is challenging because many electrocatalysts perform best in strongly alkaline electrolytes such as KOH, whereas most cells require near-neutral pH and cannot tolerate highly alkaline media. Likewise, catalysts may depend on some metal ions but might be inhibited by others. Ca2+ and Mg2+, common components of growth media, can precipitate on electrodes or even deactivate the catalyst. Conversely, the electric field and local high potentials near the electrode can harm delicate biological components or cause unwanted reactions of metabolites. Meanwhile, impurities introduced or generated in the electrolyte—such as metal ions leached from the electrodes and partially oxidized byproducts—may also inhibit enzyme activity or cell growth and disrupt the redox balance.157,158 In one-pot configurations where the culture medium also serves as the electrolyte, these problems often arise. To avoid such cross-interference, researchers use compartmentalization, in which membranes separate the electrochemical chamber from the bioreactor and allow only the intermediate to pass, or temporal separation, in which the electrolyzer is run first and its product is then fed to the bioreactor.46,89,151 This avoids direct exposure of cells to electrochemical conditions and allows each part to be optimized independently.
On the biological side, substrate tolerance and affinity are critical. Formate concentrations >100 g L−1 can inhibit many microorganisms due to acid stress and high ionic strength, high methanol levels are toxic to non-adapted cells, and acetate concentrations >10 g L−1 often slow bacterial growth. Enzymes also tend to exhibit poor tolerance to organic solvents. Advances in immobilization technologies have expanded the feasibility of electroenzymatic cascade catalysis. For example, co-immobilizing enzymes or cells on electrodes to construct bioelectrodes can effectively improve the compatibility between the electrochemical and biocatalytic modules.159 Protein engineering to enhance robustness can therefore make enzyme cascades more resilient at higher substrate loadings. Alternatively, intermediates can be fed in a stepwise or fed-batch manner to keep their concentrations within a non-toxic window. Similarly, feeding intermediates at excessively low concentrations into a bioreactor can cause substantial volumetric dilution, limiting achievable product titers and increasing the energy demand for downstream separation. Efficient conversion of intermediates into products typically requires metabolic rewiring of the host. For example, transcriptional reprogramming and pathway engineering in Saccharomyces cerevisiae can enable strains to assimilate acetate and channel it into glucose biosynthesis.89 In addition, metabolic engineering must secure efficient cofactor regeneration by coupling to auxiliary pathways or introducing transhydrogenases, so that assimilation and product formation proceed without stalling.160
3.2 Studies on electro-biocatalysis cascade systems
Biocatalytic conversion of CO2 into value-added chemicals is an exciting research direction with the potential to transform biomanufacturing processes. Table 1 summarizes representative studies and key performance metrics in CO2 electro-biocatalysis cascade systems. In general, these strategies first electrochemically reduce CO2 to well-defined C1–C3 intermediates (e.g., CO, formate, acetate/ethanol), which are subsequently upgraded via enzymatic catalysis or microbial metabolism through carbon-chain extension and functionalization, yielding more energy-dense, higher-carbon-number long-chain products.| Substrate | Intermediate | Products | Yield | Ref. |
|---|---|---|---|---|
| CO2 | CO | Butyrate (C4H8O2) | 2.1 mmol h−1 | 161 |
| Butanol (C4H10O) | 1.68 mmol h−1 | |||
| Hexanol (C6H14O) | 0.8 mmol h−1 | |||
| CO2 | Formate (HCOO−) | Polyhydroxybutyrate (PHB) (C6+) | 11.5 mg h−1 | 151 |
| CO2 | Formate (HCOO−) | Poly(3-hydroxybutyrate) (C6+) | 75.6 mg L−1 per day | 162 |
| CO2 | Formate (HCOO−) | Mesaconate (C5H6O4) | 151.6 mg L−1 per day | 163 |
| 2S-Methylsuccinate (C5H6O4) | 220.2 mg L−1 per day | |||
| CO2 | Formate (HCOO−) | Isobutanol (C4H10O) | 28 mg L−1 per day | 35 |
| CO2 | Acetate (CH3COO−) | Food or food products (Cn) | — | 46 |
| CO2 | Acetate (CH3COO−) | Glucose (C6) | 1.81 g L−1 | 89 |
| Fatty acids (C6+) | ∼500 mg L−1 | |||
| CO2 | Acetate (CH3COO−) | PHB (C6+) | 118 mg L−1 per day | 164 |
| CO2 | Acetate (CH3COO−) | PHB (C6+) | 153.3 mg L−1 per day | 165 |
| CO2 | Ethanol (CH3CH2OH) | Polyhydroxyalkanoates (PHA) (C6+) | 566.2 mg L−1 per day | 166 |
| CO2 | Ethanol (CH3CH2OH) | Lactol (C6) | 712–752 mg L−1 per day | 43 |
Li et al. integrated an electrocatalytic formate generator with an engineered Ralstonia eutropha strain that produced isobutanol and 3-methyl-1-butanol (Fig. 7a).35 Powered by solar electricity, the electrolyzer generated formate at high yield. The bacterium was engineered with a heterologous pathway that redirected acetyl-CoA from CO2 fixation into isobutanol and 3-methyl-1-butanol. The integrated system achieved an electrical-to-alcohol energy conversion efficiency of about 50%. Although product titers were still modest, on the order of a few g L−1, and the setup was a batch proof of concept, the study demonstrated that electricity and CO2 can be converted into liquid fuel in a single integrated process. Zheng et al. developed a synthetic pathway for glucose production from CO2. They used a Ni single-atom catalyst in a membrane assembly to convert CO2 to CO, and then a nanostructured Cu catalyst rich in grain boundaries to reduce CO to acetate, achieving ∼52% FE.89 To avoid salt contamination, they built a custom reactor, where acetate was formed as an acetic acid solution of about 97% purity. The acetate stream was continuously collected and fed to an engineered yeast Saccharomyces cerevisiae strain. With these modifications, the yeast grew on acetate and produced an average glucose titer of 1.81 g L−1. The related engineered strain was further adapted to produce free fatty acids (C8–C18), reaching about 0.5 g L−1. This study represented a breakthrough by demonstrating a carbon-negative route to sugars, producing them directly from CO2 rather than from plants and without relying on photosynthesis. Initial glucose yields were low, but subsequent optimizations by the same research team and related groups significantly increased production to tens of grams per liter, far above the initial 1.8 g L−1.152 Together, these advances outline a compelling concept of a fully renewable sugar refinery in which CO2 is converted, using only electricity and engineered microbes, into sugars and other biochemicals at industrially relevant concentrations.
 | ||
| Fig. 7 Electro-biocatalytic synthesis. (a) An integrated electro-microbial process to convert CO2 to higher alcohols. Electricity powered the electrochemical CO2 reduction on the cathode to produce formate, which is converted to isobutanol and 3 MB by the engineered R. eutropha. Reprinted with permission from ref. 35. Copyright 2012, American Association for the Advancement of Science; (b) gaseous CO2 is reduced to formate in the electrolyzer, and a single catholyte, formulated to be compatible with both electrochemical CO2 reduction and microbial growth, is continuously circulated to a fermenter where cells transform formate into PHB. Reprinted with permission from ref. 151. Copyright 2012, National Academy of Sciences; (c) hybrid electro-enzymatic CO2 valorization process. Waste CO2 is electrochemically reduced to ethanol on a Cu electrocatalyst (1), and the ethanol is subsequently upgraded in a two-enzyme cascade: ADH (with NAD+/NADH) oxidizes ethanol to acetaldehyde (2), and DERA catalyzes tandem aldol condensations to form cyclic statin-precursor molecules (3, 4). Reprinted with permission from ref. 43. Copyright 2022, American Chemical Society. | ||
Beyond fuels and sugars, tandem electro-bio systems have also been applied to make polymeric materials directly from CO2. Lim et al. integrated CO2 electrolysis with fermentation to produce the biopolymer poly-3-hydroxybutyrate (PHB) (Fig. 7b).151 In this system, a Sn-based GDE catalyst converted CO2 to formate with high efficiency, and the formate-containing electrolyte was circulated through a bioreactor holding Cupriavidus necator. The researchers optimized a nutrient-containing phosphate-buffered catholyte so that it supported both efficient electrochemistry and robust bacterial growth. The bacteria accumulated PHB up to 83% of their dry cell weight, yielding 1.38 g of PHB in a small reactor with a 4 cm2 electrode. The PHB content is comparable to that achieved in PHB processes fed with sugar. They also demonstrated continuous operation by continuously withdrawing broth rich in PHB and adding fresh cells. In this way, they maintained a steady state in which the electrolyzer supplied formate and the culture produced PHB for more than 17 days. This setup addresses two key integration issues. It directly couples electrolysis and fermentation through a circulating loop, and it can run continuously, which is important for scale-up. PHB is usually produced from plant-derived sugars, so directly making gram-level PHB from CO2 in this study is a significant advance. Studies have shown that PHB productivity can reach up to 118 mg L−1 per day.164 These results show that CO2 electro-biosystems can produce materials, not just simple chemicals, at practically relevant scales, thereby providing a solid process foundation and strong prospects for renewable electricity-driven biomanufacturing of materials.
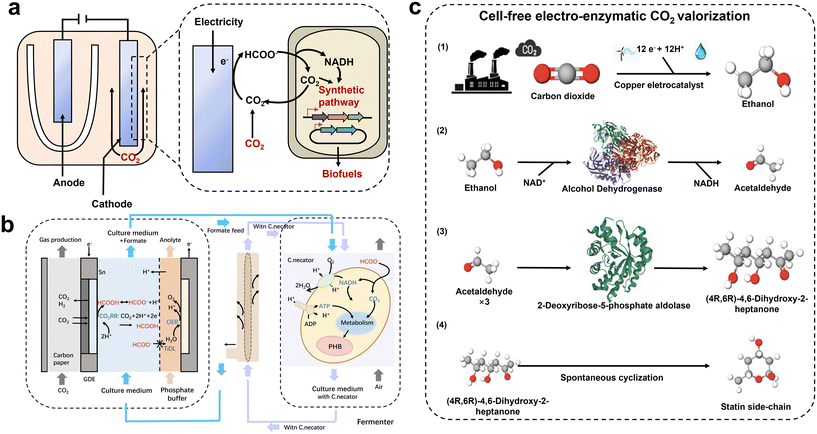
The healthcare sector is carbon intensive and urgently requires decarbonization. Jack and co-workers proposed and validated an end-to-end hybrid electrochemical-enzymatic route that upgrades waste CO2 into high-value C6 pharmaceutical precursors (Fig. 7c).43 In this platform, a three-chamber electrolyzer equipped with a Cu-based GDE efficiently converts gaseous CO2 to ethanol at a current density of 40–60 mA cm−2, achieving a selectivity of 43–81 mol% and a production rate of 368–428 mg L−1 h−1. The electrolytically produced ethanol is then separated, purified, and fed into an enzymatic reactor, where alcohol dehydrogenase (ADH) and 2-deoxyribose-5-phosphate aldolase (DERA) further construct C6 statin-precursor molecules via an acetaldehyde intermediate, delivering overall yields of 29–35%. The process attains competitive C6 lactol synthesis performance, with rates of 4.7–5.7 mM per day and titers of 712–752 mg L−1, demonstrating the feasibility of electro-enzyme cascades for manufacturing high-value molecules. Given that the C6 lactol product can be readily converted into statins, this work offers a new pathway for CO2 valorization to pharmaceutical-value chemicals.
More recently, Chen et al. developed an “electro-biodiesel” platform to synthesize lipid (fatty acid) biodiesel precursors from CO2.167 They co-designed the catalyst and the microbe. The Zn–Cu catalyst was tailored to favour C2 products. The genetically engineered Rhodococcus jostii strain can efficiently consume acetate and ethanol and store the carbon in the form of lipids. The resulting system reached a solar-to-lipid energy efficiency of 4.5%, greatly exceeding crop-based biodiesel in land use and energy efficiency. This work showcases a fully integrated design from catalyst to microorganism, where the catalyst is tuned to produce the C2 intermediates preferred by the microbe and the microbe is tuned to efficiently assimilate the catalyst's product stream, yielding one of the most efficient CO2 to fuel processes reported so far.
Taken together, these case studies employ different intermediates and operating modes, illustrating multiple integration strategies that couple electrocatalytic CO2 reduction with biocatalysis to produce multi-carbon products. The overall performance of integrated electro-biocascade systems is jointly constrained by electrolyzer losses (e.g., mass-transport limitations and overpotentials) and the ATP/NAD(P)H demands of downstream bioconversion. Thus, even when individual modules perform well, system-level energy and carbon efficiencies can remain limited. By contrast, conventional thermochemical CO2 to methanol synthesis can achieve a thermal efficiency of 62%,168 and traditional fermentation for PHB production could reach peak productivities of 1.73 g L−1 h−1,169 which are substantially higher than those of most electro-biocascades. Because evaluation metrics and system boundaries differ, direct quantitative comparisons between these routes remain scarce. Nonetheless, recent demonstrations show rapid progress: CO2 electrolysis coupled with gas fermentation has operated continuously for >1200 h at 300 mA cm−2 and achieved near-quantitative Faradaic utilization during higher-alcohol production.161 Meanwhile, formate-mediated biohybrid platforms have also demonstrated stable electrolysis at ∼120 mA cm−2, highlighting the strong development potential of electro-biocascades.151
4 Challenges and future directions
Despite exciting progress, the field of CO2 electro-biocatalytic conversion is still in its early stages. There are scientific and engineering challenges to address before these hybrid systems can be scaled up and widely implemented. Here we discuss key challenges and suggest directions for future research.Improving overall energy efficiency
Despite substantial progress at the module level, overall efficiencies for converting CO2 to products remain modest once all losses are taken into account. The overall energy efficiency of existing electricity-driven microbial electrosynthesis systems varies widely across reactor configurations and product pathways. Reported systems can reach efficiencies on the order of the teens to several tens of percent, yet further improvements are generally needed, particularly for end-to-end integrated operation and long-duration runs.89,167,170 Future efforts must focus on lowering electrocatalytic overpotentials, sharpening product selectivity, minimizing side reactions and redesigning biological pathways to reduce ATP and NAD(P)H demand. Whole process models that couple electrolysis with cellular metabolism will be essential to identify dominant energy sinks and guide targeted optimization.Scaling up and redesigning reactors
Moving from electrodes on the centimetre scale and bioreactors with volumes from milliliters to liters to industrial plants will amplify challenges in mixing, mass transfer, gas handling and product purification. Large area electrodes and high currents are already used in water electrolysis and in plants that convert CO2 to CO, but maintaining high purity liquid products and managing large CO2 and O2 flows at industrial scale remain difficult.171 Modular “scale-out” architectures, with many electro-bioreactors operated in parallel and co-located with cheap renewable power and CO2 point sources, are likely to be a practical route to deployment.Engineering biocatalysts and intermediate selection
On the biological side, a key goal is to build strains that consume C1 and C2 feeds rapidly and channel carbon into products at high titer, rate and yield. Strategies include adapting existing production strains that currently run on sugars, exploiting non-model organisms that natively fix CO2 or C1 substrate, and designing co-cultures that divide labor between substrate assimilation and product formation. Continued development of genetic tools for acetogens, methylotrophs and other specialist microbes will expand the design space for electro-biocatalytic cascade systems. In parallel, establishing target-oriented, quantitative criteria for selecting bridging C1–C3 intermediates (e.g., achievable titer and stability, separation/transport penalties, and biocompatibility/assimilation constraints) is an important next step to guide the rational design of electro-bio cascades.Integrating control, CO2 sourcing and sustainability
Although electrocatalytic CO2 reduction has shown promise for scale-up, achieving large-scale and stable industrial production remains challenging once it is cascade-coupled with biocatalysis, particularly in terms of long-term operational stability, mass-transport and impurity tolerance, and system-level energy efficiency and cost control. Robust operation of coupled systems will depend on dynamic control of the relative rates of electrolysis and bioconversion. These processes must also be coupled to realistic CO2 sources (for example, flue gas or direct air capture streams) and validated through rigorous techno-economic analysis and life-cycle assessment to demonstrate climate benefits and cost competitiveness.172,173 Future cascade research should be oriented toward industrial deployment by strengthening integrated technical-economic-application evaluation and optimization. And quantitatively evaluate the environmental benefits and economic costs to provide more intuitive data support. Utilizing waste or air-derived CO2, alongside low-cost renewable energy sources, is one strategy that significantly improves economic efficiency.In conclusion, electro-biocatalysis cascades represent a powerful synthesis paradigm for a sustainable future. Fast and modular energy input supplied by electrochemistry is coupled to the complex and carbon-efficient chemistry of biological systems. In recent years, striking proof-of-concept studies have been reported in which fuels, plastics and even food precursors are produced from CO2 and renewable electricity. The field is evolving rapidly, with continuous improvements in catalysts, metabolic engineering strategies and integrated process designs. Advances in materials science will be needed to deliver stable, selective catalysts and practical reactor architectures. Progress in synthetic biology will be required to create robust cell factories and enzyme systems. Contributions from systems and process engineering will be essential to integrate and scale these hybrid platforms. Together, these efforts point toward a new industrial paradigm in which carbon waste becomes a valuable feedstock, powered by green energy, to yield the products society needs—from fuels and fertilizers to pharmaceuticals. Over the coming decade, both incremental improvements and step-change innovations, such as substantial gains in overall energy and carbon efficiency, are expected to further strengthen the technical and economic feasibility of this approach.
Conflicts of interest
The authors declare no conflict of interest.Data availability
This review article does not involve original data. All analyzed information is from published sources.Acknowledgements
This work was supported by the National Natural Science Foundation of China (22408113) and the Fundamental Research Funds for the Central Universities (2024ZYGXZR078).References
- J. Artz, T. E. Müller, K. Thenert, J. Kleinekorte, R. Meys, A. Sternberg, A. Bardow and W. Leitner, Sustainable conversion of carbon dioxide: An integrated review of catalysis and life cycle assessment, Chem. Rev., 2018, 118, 434–504 CrossRef CAS PubMed.
- J. Klankermayer, S. Wesselbaum, K. Beydoun and W. Leitner, Selective catalytic synthesis using the combination of carbon dioxide and hydrogen: Catalytic chess at the interface of energy and chemistry, Angew. Chem., Int. Ed., 2016, 55, 7296–7343 CrossRef CAS PubMed.
- R. Gao, X. X. Xu, Z. M. Wu, L. G. Xu, H. Kuang and C. L. Xu, The potential of converting carbon dioxide to food compounds asymmetric catalysis, Nanoscale Adv., 2023, 5, 2865–2872 RSC.
- R. Croce, E. Carmo-Silva, Y. B. Cho, M. Ermakova, J. Harbinson, T. Lawson, A. J. McCormick, K. K. Niyogi, D. R. Ort, D. Patel-Tupper, P. Pesaresi, C. Raines, A. P. M. Weber and X. G. Zhu, Perspectives on improving photosynthesis to increase crop yield, Plant Cell, 2024, 36, 3944–3973 CrossRef CAS PubMed.
- S. W. Lee, M. L. Luna, N. Berdunov, W. M. Wan, S. Kunze, S. Shaikhutdinov and B. R. Cuenya, Unraveling surface structures of gallium promoted transition metal catalysts in CO2 hydrogenation, Nat. Commun., 2023, 14, 4649 CrossRef CAS PubMed.
- H. K. Wu, F. Zhang, J. Y. Li, Z. R. Tang and Y. J. Xu, Photo-driven Fischer-Tropsch synthesis, J. Mater. Chem. A, 2020, 8, 24253–24266 RSC.
- J. Lin, S. L. Yan, C. X. Zhang, Q. Hu and Z. M. Cheng, Electroreduction of CO2 toward high current density, Processes, 2022, 10, 826 CrossRef CAS.
- M. Serafini, F. Mariani, F. Basile, E. Scavetta and D. Tonelli, From traditional to new benchmark catalysts for CO2 electroreduction, Nanomaterials, 2023, 13, 5090 CrossRef PubMed.
- Z. X. Gu, H. Shen, Z. Chen, Y. Y. Yang, C. Yang, Y. L. Ji, Y. H. Wang, C. Zhu, J. L. Liu, J. Li, T. K. Sham, X. Xu and G. F. Zheng, Efficient electrocatalytic CO2 reduction to C2+ alcohols at defect-site-rich cu surface, Joule, 2021, 5, 429–440 CrossRef CAS.
- M. M. Abdelnaby, K. L. Liu, K. Hassanein and Z. Y. Yin, Photo/electrochemical carbon dioxide conversion into C3+ hydrocarbons: Reactivity and selectivity, ChemNanoMat, 2021, 7, 969–981 CrossRef CAS.
- Y. Y. Birdja, E. Pérez-Gallent, M. C. Figueiredo, A. J. Göttle, F. Calle-Vallejo and M. T. M. Koper, Advances and challenges in understanding the electrocatalytic conversion of carbon dioxide to fuels, Nat. Energy, 2019, 4, 732–745 CrossRef CAS.
- Y. J. Chen, X. Y. Wang, X. Y. Li, R. K. Miao, J. C. Dong, Z. L. Zhao, C. H. Liu, J. E. Huang, J. H. Wu, S. L. Chu, W. Y. Ni, Z. M. Guo, Y. Xu, P. F. Ou, B. J. Xu, Y. Hou, D. Sinton and E. H. Sargent, Electrified synthesis of n-propanol using a dilute alloy catalyst, Nat. Catal., 2025, 8, 239–247 CrossRef CAS.
- R. Siedentop, C. Claassen, D. Rother, S. Luetz and K. Rosenthal, Getting the most out of enzyme cascades: Strategies to optimize multi-enzymatic reactions, Catalysts, 2021, 11, 1183 CrossRef CAS.
- Y. Y. Qiao, W. Y. Ma, S. J. Zhang, F. Guo, K. Liu, Y. J. Jiang, Y. X. Wang, F. X. Xin, W. M. Zhang and M. Jiang, Artificial multi-enzyme cascades and whole-cell transformation for bioconversion of C1 compounds: Advances, challenge and perspectives, Synth. Syst. Biotechnol., 2023, 8, 578–583 CrossRef CAS PubMed.
- I. Bernhardsgrütter, G. M. M. Stoffel, T. E. Miller and T. J. Erb, CO2-converting enzymes for sustainable biotechnology: From mechanisms to application, Curr. Opin. Biotechnol., 2021, 67, 80–87 CrossRef PubMed.
- D. W. Mulder, J. W. Peters and S. Raugei, Catalytic bias in oxidation-reduction catalysis, Chem. Commun., 2021, 57, 713–720 RSC.
- S. Bierbaumer, M. Nattermann, L. Schulz, R. Zschoche, T. J. Erb, C. K. Winkler, M. Tinzl and S. M. Glueck, Enzymatic conversion of CO2: From natural to artificial utilization, Chem. Rev., 2023, 123, 5702–5754 CrossRef CAS PubMed.
- X. J. Yu, D. Niks, A. Mulchandani and R. Hille, Efficient reduction of CO2 by the molybdenum-containing formate dehydrogenase from Cupriavidus, J. Biol. Chem., 2017, 292, 16872–16879 CrossRef CAS PubMed.
- A. Bassegoda, C. Madden, D. W. Wakerley, E. Reisner and J. Hirst, Reversible interconversion of CO2 and formate by a molybdenum-containing formate dehydrogenase, J. Am. Chem. Soc., 2015, 137, 4592–4592 CrossRef CAS PubMed.
- S. Roy, A. Cherevotan and S. C. Peter, Thermochemical CO2 hydrogenation to single carbon products: Scientific and technological challenges, ACS Energy Lett., 2018, 3, 1938–1966 CrossRef CAS.
- X. G. Zhu, S. P. Long and D. R. Ort, Improving photosynthetic efficiency for greater yield, Annu. Rev. Plant Biol., 2010, 61, 235–261 CrossRef CAS PubMed.
- K. Im-orb, L. Simasatitkul and A. Arpornwichanop, Performance analysis and optimization of the biomass gasification and fischer-tropsch integrated process for green fuel productions, Comput.-Aided Chem. Eng., 2015, 37, 275–280 CAS.
- F. Liew, M. E. Martin, R. C. Tappel, B. D. Heijstra, C. Mihalcea and M. Köpke, Gas fermentation a flexible platform for commercial scale production of low-carbon-fuels and chemicals from waste and renewable feedstocks, Front. Microbiol., 2016, 7, 694 Search PubMed.
- Y. Hori, CO2-reduction, catalyzed by metal electrodes, in Handbook of Fuel Cells, 2010, DOI:10.1002/9780470974001.f207055.
- Y. Hori, A. Murata, K. Kikuchi and S. Suzuki, Electrochemical reduction of carbon-dioxide to carbon-monoxide at a gold electrode in aqueous potassium hydrogen carbonate, J. Chem. Soc., Chem. Commun., 1987, 728–729 RSC.
- S. Nitopi, E. Bertheussen, S. B. Scott, X. Y. Liu, A. K. Engstfeld, S. Horch, B. Seger, I. E. L. Stephens, K. Chan, C. Hahn, J. K. Norskov, T. F. Jaramillo and I. Chorkendorff, Progress and perspectives of electrochemical CO2 reduction on copper in aqueous electrolyte, Chem. Rev., 2019, 119, 7610–7672 CrossRef CAS PubMed.
- S. C. Ma, M. Sadakiyo, R. Luo, M. Heima, M. Yamauchi and P. J. A. Kenis, One-step electrosynthesis of ethylene and ethanol from CO2 in an alkaline electrolyzer, J. Power Sources, 2016, 301, 219–228 CrossRef CAS.
- C. T. Dinh, F. P. G. de Arquer, D. Sinton and E. H. Sargent, High rate, selective, and stable electroreduction of CO2 to CO in basic and neutral media, ACS Energy Lett., 2018, 3, 2835–2840 CrossRef CAS.
- R. A. Tufa, D. Chanda, M. Ma, D. Aili, T. B. Demissie, J. Vaes, Q. F. Li, S. H. Liu and D. Pant, Towards highly efficient electrochemical CO2 reduction: Cell designs, membranes and electrocatalysts, Appl. Energy, 2020, 277, 115557 CrossRef CAS.
- C. B. Li, M. M. Guo, B. Yang, Y. Ji, J. Zhang, L. J. Zhou, C. X. Liu, H. Y. Wang, J. W. Li, W. Q. Xue, X. Y. Zhang, H. L. Zeng, Y. J. Wang, D. H. Zhao, K. X. Zhong, S. S. Pi, M. Z. Hei, X. Li, Q. Jiang, T. T. Zheng, X. Gao and C. Xia, Efficient and scalable upcycling of oceanic carbon sources into bioplastic monomers, Nat. Catal., 2025, 8, 1023–1037 CrossRef CAS.
- L. C. Weng, A. T. Bell and A. Z. Weber, Modeling gas-diffusion electrodes for CO2 reduction, Phys. Chem. Chem. Phys., 2018, 20, 16973–16984 RSC.
- K. P. Nevin, T. L. Woodard, A. E. Franks, Z. M. Summers and D. R. Lovley, Microbial electrosynthesis: Feeding microbes electricity to convert carbon dioxide and water to multicarbon extracellular organic compounds, MBio, 2010, 1, e00103–e00110 CrossRef PubMed.
- K. P. Nevin, S. A. Hensley, A. E. Franks, Z. M. Summers, J. H. Ou, T. L. Woodard, O. L. Snoeyenbos-West and D. R. Lovley, Electrosynthesis of organic compounds from carbon dioxide is catalyzed by a diversity of acetogenic microorganisms, Appl. Environ. Microbiol., 2011, 77, 2882–2886 CrossRef CAS PubMed.
- K. Rabaey and R. A. Rozendal, Microbial electrosynthesis-revisiting the electrical route for microbial production, Nat. Rev. Microbiol., 2010, 8, 706–716 CrossRef CAS PubMed.
- H. Li, P. H. Opgenorth, D. G. Wernick, S. Rogers, T. Y. Wu, W. Higashide, P. Malati, Y. X. Huo, K. M. Cho and J. C. Liao, Integrated electromicrobial conversion of CO2 to higher alcohols, Science, 2012, 335, 1596–1596 CrossRef CAS PubMed.
- L. Luan, Y. Zhang, X. Ji, B. Guo, S. Song, Y. Huang and S. Zhang, Electro-driven multi-enzymatic cascade conversion of CO2 to ethylene glycol in nano-reactor, Adv. Sci., 2024, 11, e2407204 CrossRef PubMed.
- S. Srikanth, M. Maesen, X. Dominguez-Benetton, K. Vanbroekhoven and D. Pant, Enzymatic electrosynthesis of formate through CO2 sequestration/reduction in a bioelectrochemical system (BES), Bioresour. Technol., 2014, 165, 350–354 CrossRef CAS PubMed.
- R. Cai, R. D. Milton, S. Abdellaoui, T. Park, J. Patel, B. Alkotaini and S. D. Minteer, Electroenzymatic C-C bond formation from CO2, J. Am. Chem. Soc., 2018, 140, 5041–5044 CrossRef CAS PubMed.
- D. R. Lovley and K. P. Nevin, Electrobiocommodities: Powering microbial production of fuels and commodity chemicals from carbon dioxide with electricity, Curr. Opin. Biotechnol., 2013, 24, 385–390 CrossRef CAS PubMed.
- A. S. Hawkins, Y. J. Han, H. Lian, A. J. Loder, A. L. Menon, I. J. Iwuchukwu, M. Keller, T. T. Leuko, M. W. W. Adams and R. M. Kelly, Extremely thermophilic routes to microbial electrofuels, ACS Catal., 2011, 1, 1043–1050 CrossRef CAS.
- W. O. Khunjar, A. Sahin, A. C. West, K. Chandran and S. Banta, Biomass production from electricity using ammonia as an electron carrier in a reverse microbial fuel cell, PLoS One, 2012, 7, e44846 CrossRef CAS PubMed.
- H. M. Woo, Solar-to-chemical and solar-to-fuel production from CO2 by metabolically engineered microorganisms, Curr. Opin. Biotechnol., 2017, 45, 1–7 CrossRef CAS PubMed.
- J. Jack, H. Fu, A. Leininger, T. K. Hyster and Z. J. Ren, Cell-free CO2 valorization to C6 pharmaceutical precursors via a novel electro-enzymatic process, ACS Sustainable Chem. Eng., 2022, 10, 4114–4121 CrossRef CAS.
- T. Bouwens, S. J. Cobb, C. W. S. Yeung, Y. Liu, G. Martins, I. A. C. Pereira and E. Reisner, Semiartificial photoelectrochemistry for CO2-mediated enantioselective organic synthesis, J. Am. Chem. Soc., 2025, 147, 13114–13119 CrossRef CAS PubMed.
- C. W. S. Yeung, Y. Liu, D. M. Vahey, S. J. Cobb, V. Andrei, A. M. Coito, R. R. Manuel, I. A. C. Pereira and E. Reisner, Semi-artificial leaf interfacing organic semiconductors and enzymes for solar chemical synthesis, Joule, 2025, 9, 102165 CrossRef CAS.
- E. C. Hann, S. Overa, M. Harland-Dunaway, A. F. Narvaez, D. N. Le, M. L. Orozco-Cárdenas, F. Jiao and R. E. Jinkerson, A hybrid inorganic-biological artificial photosynthesis system for energy-efficient food production, Nat. Food, 2022, 3, 461–471 CrossRef CAS PubMed.
- S. Jeevanandham, S. Ramasundaram, N. Vijay, T. H. Oh and S. T. Selvan, Recent progress in designing nanomaterial biohybrids for artificial photosynthesis, Nanomaterials, 2025, 15, 730 CrossRef CAS PubMed.
- W. Yu, M. V. Pavliuk, A. J. Liu, Y. Zeng, S. P. Xia, Y. M. Huang, H. T. Bai, F. T. Lv, H. N. Tian and S. Wang, Photosynthetic polymer dots-bacteria biohybrid system based on transmembrane electron transport for fixing CO2 into poly-3-hydroxybutyrate, ACS Appl. Mater. Interfaces, 2023, 15, 2183–2191 CrossRef CAS PubMed.
- C. Q. Zhang, C. Ottenheim, M. Weingarten and L. H. Ji, Microbial utilization of next-generation feedstocks for the biomanufacturing of value-added chemicals and food ingredients, Front. Bioeng. Biotechnol., 2022, 10, 874612 CrossRef PubMed.
- H. J. Cui, Y. B. Guo, L. M. Guo, L. Wang, Z. Zhou and Z. Q. Peng, Heteroatom-doped carbon materials and their composites as electrocatalysts for CO2 reduction, J. Mater. Chem. A, 2018, 6, 18782–18793 RSC.
- C. Liu, R. T. Guo, H. W. Zhu, H. F. Cui, M. Y. Liu and W. G. Pan, Cu2O-based catalysts applied for electrocatalytic CO2 reduction: A review, J. Mater. Chem. A, 2024, 12, 31769–31796 RSC.
- C. L. Rooney, M. Lyons, Y. S. Wu, G. F. Hu, M. Y. Wang, C. Choi, Y. Z. Gao, C. W. Chang, G. W. Brudvig, Z. X. Feng and H. L. Wang, Active sites of cobalt phthalocyanine in electrocatalytic CO2 reduction to methanol, Angew. Chem., Int. Ed., 2024, 63, e202310623 CrossRef CAS PubMed.
- S. Yu, H. Yamauchi, S. Wang, A. Aggarwal, J. Kim, K. Gordiz, B. T. Huang, H. B. Xu, D. J. Zheng, X. Wang, H. Iriawan, D. Menga and Y. Shao-Horn, CO2-to-methanol electroconversion on a molecular cobalt catalyst facilitated by acidic cations, Nat. Catal., 2024, 7, 1000–1009 CrossRef CAS.
- M. Z. Iqbal, S. Imteyaz, C. Ghanty and S. Sarkar, A review on electrochemical conversion of CO2 to CO: Ag-based electrocatalyst and cell configuration for industrial application, J. Ind. Eng. Chem., 2022, 113, 15–31 CrossRef CAS.
- M. L. Souza and F. H. B. Lima, Dibenzyldithiocarbamate-functionalized small gold nanoparticles as selective catalysts for the electrochemical reduction of CO2 to CO, ACS Catal., 2021, 11, 12208–12219 CrossRef CAS.
- S. Jin, Z. M. Hao, K. Zhang, Z. H. Yan and J. Chen, Advances and challenges for the electrochemical reduction of CO2 to CO: From fundamentals to industrialization, Angew. Chem., Int. Ed., 2021, 60, 20627–20648 CrossRef CAS PubMed.
- N. Ye, K. Wang, Y. Tan, Z. Qian, H. Guo, C. Shang, Z. Lin, Q. Huang, Y. Liu, L. Li, Y. Gu, Y. Han, C. Zhou, M. Luo and S. Guo, Industrial-level CO2 to formate conversion on turing-structured electrocatalysts, Nat. Synth., 2025, 4, 799–807 CrossRef CAS.
- P. X. Lei, S. Q. Liu, Q. R. Wen, J. Y. Wu, S. W. Wu, X. X. Wei, R. F. Feng, X. Z. Fu and J. L. Luo, Integrated “two-in-one” strategy for high-rate electrocatalytic CO2 reduction to formate, Angew. Chem., Int. Ed., 2025, 64, e202415726 CrossRef CAS PubMed.
- P. F. Li, F. Q. Yang, J. Li, Q. Zhu, J. W. Xu, X. J. Loh, K. W. Huang, W. P. Hu and J. Lu, Nanoscale engineering of p-block metal-based catalysts toward industrial-scale electrochemical reduction of CO2, Adv. Energy Mater., 2023, 13, 2301597 CrossRef CAS.
- W. B. Li, C. Yu, X. Y. Tan, Y. W. Ren, Y. F. Zhang, S. Cui, Y. Yang and J. S. Qiu, Beyond leverage in activity and stability toward CO2 electroreduction to formate over a bismuth catalyst, ACS Catal., 2024, 14, 8050–8061 CrossRef CAS.
- J. Xue, X. M. Fu, S. Geng, K. L. Wang, Z. W. Li and M. Li, Boosting electrochemical CO2 reduction via valence state and oxygen vacancy controllable Bi-Sn/CeO2 nanorod, J. Environ. Manage., 2023, 342, 118354 CrossRef CAS PubMed.
- H. Cui, X. Wang and L. Zhang, Coupling electrocatalysis and biotransformation for CO2-based biomanufacturing, Synth. Biol. Eng., 2025, 3, 10010 CAS.
- Q. S. Zhu, C. L. Rooney, H. Shema, C. S. A. Zeng, J. A. Panetier, E. Gross, H. L. Wang and L. R. Baker, The solvation environment of molecularly dispersed cobalt phthalocyanine determines methanol selectivity during electrocatalytic CO2 reduction, Nat. Catal., 2024, 7, 987–999 CrossRef CAS.
- J. Cai, Q. Zhao, W. Y. Hsu, C. Choi, Y. Liu, J. M. P. Martirez, C. Chen, J. Huang, E. A. Carter and Y. Huang, Highly selective electrochemical reduction of CO2 into methane on nanotwinned Cu, J. Am. Chem. Soc., 2023, 145, 9136–9143 CrossRef CAS PubMed.
- H. Y. Wang, J. Xue, C. X. Liu, Z. Y. Chen, C. B. Li, X. Li, T. T. Zheng, Q. Jiang and C. Xia, CO2 electrolysis toward acetate: A review, Curr. Opin. Electrochem., 2023, 39, 101253 CrossRef CAS.
- X. W. Nie, W. J. Luo, M. J. Janik and A. Asthagiri, Reaction mechanisms of CO2 electrochemical reduction on Cu(111) determined with density functional theory, J. Catal., 2014, 312, 108–122 CrossRef CAS.
- Y. H. Shi, Y. Yang, A. N. Xu, K. N. Hui, F. W. Li and J. Zeng, Immobilized azole layer tunes interfacial hydrogen source for CO2 electroreduction in strong acid, J. Am. Chem. Soc., 2025, 147, 35698–35704 CrossRef CAS PubMed.
- Y. S. Wu, Z. Jiang, X. Lu, Y. Y. Liang and H. L. Wang, Domino electroreduction of CO2 to methanol on a molecular catalyst, Nature, 2019, 575, 639–642 CrossRef CAS PubMed.
- J. Li, Q. S. Zhu, A. L. Chang, S. Cheon, Y. Z. Gao, B. Shang, H. Li, C. L. Rooney, L. T. Ren, Z. Jiang, Y. Y. Liang, Z. X. Feng, S. Z. Yang, L. R. Baker and H. L. Wang, Molecular-scale CO spillover on a dual-site electrocatalyst enhances methanol production from CO2 reduction, Nat. Nanotechnol., 2025, 20, 515–522 CrossRef CAS PubMed.
- K. P. Kuhl, E. R. Cave, D. N. Abram and T. F. Jaramillo, New insights into the electrochemical reduction of carbon dioxide on metallic copper surfaces, Energy Environ. Sci., 2012, 5, 7050–7059 RSC.
- Z. M. Li, Z. Liu, S. J. Li, Y. H. Pei, D. Li, J. L. Mao, R. Zhou, C. T. Qiu, Y. Y. Lu and B. Zhang, Modulating the localized electronic distribution of Cu species during reconstruction for enhanced electrochemical CO2 reduction to C2+ products, J. Mater. Chem. A, 2024, 12, 15082–15089 RSC.
- X. S. Ma, C. Fang, M. Ding, Y. Zuo, X. Y. Sun and S. X. Wang, Atomic-level elucidation of lattice-hydrogens in copper catalysts for selective CO2 electrochemical conversion toward C2 Products, Angew. Chem., Int. Ed., 2025, 64, e202500191 CrossRef CAS PubMed.
- L. D. Sun, X. L. Zheng, Y. R. Li, M. R. Lin, X. L. Zeng, J. Yu, Z. X. Song and L. Zhang, Nanoconfinement and tandem catalysis over yolk-shell catalysts towards electrochemical reduction of CO2 to multi-carbon products, J. Colloid Interface Sci., 2025, 687, 733–741 CrossRef CAS PubMed.
- M. J. Wei, J. W. Li, J. K. Zhao, S. P. Hu, Y. Ji, W. Q. Xue, Y. Z. Dai, H. Y. Wang, X. Y. Zhang, K. N. Hui, X. Li, C. Xia, T. T. Zheng and J. Zeng, Stabilization of oxidized Cu species via CeOx nano-islands for enhanced CO2 reduction to C2+ products, Natl. Sci. Rev., 2025, 12, 351 CrossRef PubMed.
- Z. Q. Liang, T. T. Zhuang, A. Seifitokaldani, J. Li, C. W. Huang, C. S. Tan, Y. Li, P. De Luna, C. T. Dinh, Y. F. Hu, Q. F. Xiao, P. L. Hsieh, Y. H. Wang, F. W. Li, R. Quintero-Bermudez, Y. S. Zhou, P. N. Chen, Y. J. Pang, S. C. Lo, L. J. Chen, H. R. Tan, Z. Xu, S. L. Zhao, D. Sinton and E. H. Sargent, Copper-on-nitride enhances the stable electrosynthesis of multi-carbon products from CO2, Nat. Commun., 2018, 9, 3828 CrossRef PubMed.
- X. Dong, X. F. Sun, S. Q. Jia, S. T. Han, D. W. Zhou, T. Yao, M. Wang, M. H. Fang, H. H. Wu and B. X. Han, Electrochemical CO2 reduction to C2+ products with ampere-level current on carbon-modified copper catalysts, Acta Phys.-Chim. Sin., 2025, 41, 100024 CrossRef.
- L. Y. Q. Xie, Y. J. Jiang, W. L. Zhu, S. C. Ding, Y. Zhou and J. J. Zhu, Cu-based catalyst designs in CO2 electroreduction: Precise modulation of reaction intermediates for high-value chemical generation, Chem. Sci., 2023, 14, 13629–13660 RSC.
- C. S. Cao, D. D. Ma, J. F. Gu, X. Y. Xie, G. Zeng, X. F. Li, S. G. Han, Q. L. Zhu, X. T. Wu and Q. Xu, Metal-organic layers leading to atomically thin bismuthene for efficient carbon dioxide electroreduction to liquid fuel, Angew. Chem., Int. Ed., 2020, 59, 15014–15020 CrossRef CAS PubMed.
- J. P. Edwards, Y. Xu, C. M. Gabardo, C. T. Dinh, J. Li, Z. B. Qi, A. Ozden, E. H. Sargent and D. Sinton, Efficient electrocatalytic conversion of carbon dioxide in a low-resistance pressurized alkaline electrolyzer, Appl. Energy, 2020, 261, 114305 CrossRef CAS.
- C. P. O'Brien, R. K. Miao, A. S. Zeraati, G. H. Lee, E. H. Sargent and D. Sinton, CO2 electrolyzers, Chem. Rev., 2024, 124, 3648–3693 CrossRef PubMed.
- Y. T. Wang, Y. H. Li, J. Z. Liu, C. X. Dong, C. Q. Xiao, L. Cheng, H. L. Jiang, H. Jiang and C. Z. Li, BiPO4-derived 2D nanosheets for efficient electrocatalytic reduction of CO2 to liquid fuel, Angew. Chem., Int. Ed., 2021, 60, 7681–7685 CrossRef CAS PubMed.
- D. Ma, T. Jin, K. Y. Xie and H. T. Huang, An overview of flow cell architecture design and optimization for electrochemical CO2 reduction, J. Mater. Chem. A, 2021, 9, 20897–20918 RSC.
- W. C. Ma, S. J. Xie, T. T. Liu, Q. Y. Fan, J. Y. Ye, F. F. Sun, Z. Jiang, Q. H. Zhang, J. Cheng and Y. Wang, Electrocatalytic reduction of CO2 to ethylene and ethanol through hydrogen-assisted C–C coupling over fluorine-modified copper, Nat. Catal., 2020, 3, 478–487 CrossRef CAS.
- W. S. Fang, W. Guo, R. H. Lu, Y. Yan, X. K. Liu, D. Wu, F. M. Li, Y. S. Zhou, C. H. He, C. F. Xia, H. T. Niu, S. C. Wang, Y. W. Liu, Y. Mao, C. Y. Zhang, B. You, Y. J. Pang, L. L. Duan, X. Yang, F. Song, T. Y. Zhai, G. X. Wang, X. P. Guo, B. Tan, T. Yao, Z. Y. Wang and B. Y. Xia, Durable CO2 conversion in the proton-exchange membrane system, Nature, 2024, 627, 86–91 CrossRef PubMed.
- L. Ge, H. Rabiee, M. R. Li, S. Subramanian, Y. Zheng, J. H. Lee, T. Burdyny and H. Wang, Electrochemical CO2 reduction in membrane-electrode assemblies, Chem, 2022, 8, 663–692 CAS.
- L. M. Hu, J. A. Wrubel, C. M. Baez-Cotto, F. Intia, J. H. Park, A. J. Kropf, N. Kariuki, Z. Huang, A. Farghaly, L. Amichi, P. Saha, L. Tao, D. A. Cullen, D. J. Myers, M. S. Ferrandon and K. C. Neyerlin, A scalable membrane electrode assembly architecture for efficient electrochemical conversion of CO2 to formic acid, Nat. Commun., 2023, 14, 7605 CrossRef CAS PubMed.
- A. Luthfiah and C. W. Lee, Solid-state electrolyte-based electrochemical conversion of carbon dioxide: Progress and opportunities, ChemCatChem, 2023, 15, e202300702 CrossRef CAS.
- C. Xia, P. Zhu, Q. Jiang, Y. Pan, W. T. Liang, E. Stavitski, H. N. Alshareef and H. T. Wang, Continuous production of pure liquid fuel solutions via electrocatalytic CO2 reduction using solid-electrolyte devices, Nat. Energy, 2020, 4, 776–785 CrossRef.
- T. T. Zheng, M. L. Zhang, L. H. Wu, S. Y. Guo, X. J. Liu, J. K. Zhao, W. Q. Xue, J. W. Li, C. X. Liu, X. Li, Q. Jiang, J. Bao, J. Zeng, T. Yu and C. Xia, Upcycling CO2 into energy-rich long-chain compounds via electrochemical and metabolic engineering, Nat. Catal., 2022, 5, 388–396 CrossRef CAS.
- W. J. Wu, J. Long and J. P. Xiao, Recent advances in electrochemical CO2 reduction catalyzed by single-atom alloys, ChemCatChem, 2025, 17, 202401785 CrossRef.
- T. T. H. Hoang, S. Verma, S. C. Ma, T. T. Fister, J. Timoshenko, A. I. Frenkel, P. J. A. Kenis and A. A. Gewirth, Nanoporous copper-silver alloys by additive-controlled electrodeposition for the selective electroreduction of CO2 to ethylene and ethanol, J. Am. Chem. Soc., 2018, 140, 5791–5797 CrossRef CAS PubMed.
- N. Rehman, Y. Wang, X. Tan, X. Fan, X. Li, W. Shi, A. W. Robertson, J. Texter, U. Aiman and Z. Sun, Electrocatalytic CO2 reduction to multi-carbon products on non-copper-based catalysts: Reaction pathways, enhancement strategies, and future challenges, Carbon Hydrogen, 2025, 27, 102–127 CrossRef CAS.
- C. B. Chen, Y. F. Li, S. Yu, S. Louisia, J. B. Jin, M. F. Li, M. B. Ross and P. D. Yang, Cu-Ag tandem catalysts for high-rate CO2 electrolysis toward multicarbons, Joule, 2020, 4, 1688–1699 CrossRef CAS.
- S. Lee, D. Kim and J. Lee, Electrocatalytic production of C3-C4 compounds by conversion of CO2 on a chloride-induced bi-phasic Cu2O-Cu catalyst, Angew. Chem., Int. Ed., 2015, 54, 14701–14705 CrossRef CAS PubMed.
- K. U. D. Calvinho, A. B. Laursen, K. M. K. Yap, T. A. Goetjen, S. Hwang, N. Murali, B. Mejia-Sosa, A. Lubarski, K. M. Teeluck, E. S. Hall, E. Garfunkel, M. Greenblatt and G. C. Dismukes, Selective CO2 reduction to C3 and C4 oxyhydrocarbons on nickel phosphides at overpotentials as low as 10 mV, Energy Environ. Sci., 2018, 11, 2550–2559 RSC.
- M. Choi, S. Bae, Y. Kim, Y. Lee, M. Cho, S. Kang and J. Lee, Selective formaldehyde condensation on phosphorus-rich copper catalyst to produce liquid C3+ chemicals in electrocatalytic CO2 reduction, Nat. Catal., 2025, 8, 476–486 CrossRef CAS.
- Y. Li, X. Wang, K. D. Chen, Z. K. Zhuang, H. T. Tang, T. Yu and W. B. Cao, Efficient production of 2′-fucosyllactose in Pichia pastoris through metabolic engineering and constructing an orthogonal energy supply system, Synth. Syst. Biotechnol., 2025, 10, 807–815 CrossRef CAS PubMed.
- H. L. Wu, H. J. Pan, Z. J. Li, T. F. Liu, F. L. Liu, S. Y. Xiu, J. Wang, H. Q. Wang, Y. Hou, B. Yang, L. C. Lei and J. Z. Lian, Efficient production of lycopene from CO2 via microbial electrosynthesis, Chem. Eng. J., 2022, 430, 132943 CrossRef CAS.
- C. X. Liu, J. K. Zhao, H. T. Tang, J. Xue, W. Q. Xue, X. Li, H. L. Li, Q. Jiang, T. T. Zheng, T. Yu, J. Zeng and C. Xia, Upcycling surplus acetone into long-chain chemicals using a tandem electro-biosystem, Nat. Sustain., 2025, 8, 806–817 CrossRef.
- M. G. Lee, X. Y. Li, A. Ozden, J. Wicks, P. F. Ou, Y. H. Li, R. Dorakhan, J. K. Y. Lee, H. K. Park, J. W. Yang, B. Chen, J. Abed, R. dos Reis, G. H. Lee, J. E. Huang, T. Peng, Y. H. Chin, D. Sinton and E. H. Sargent, Selective synthesis of butane from carbon monoxide using cascade electrolysis and thermocatalysis at ambient conditions, Nat. Catal., 2023, 6, 310–318 CrossRef CAS.
- B. Cao, F. Z. Li, S. B. Han, Q. Xu and J. Gu, Electrocatalytic CO reduction to produce long-chain products through fischer-tropsch pathway, ChemElectroChem, 2025, 12, e202400595 CrossRef CAS.
- C. Hertweck, The biosynthetic logic of polyketide diversity, Angew. Chem., Int. Ed., 2009, 48, 4688–4716 CrossRef CAS PubMed.
- C. T. Walsh and M. A. Fischbach, Natural products version 2.0: Connecting genes to molecules, J. Am. Chem. Soc., 2010, 132, 2469–2493 CrossRef CAS PubMed.
- D. L. Zhu, S. S. Ao, H. H. Deng, M. Wang, C. Q. Qin, J. Zhang, Y. R. Jia, P. Ye and H. G. Ni, Ordered coimmobilization of a multienzyme cascade system with a metal organic framework in a membrane: Reduction of CO2 to methanol, ACS Appl. Mater. Interfaces, 2019, 11, 33581–33588 CrossRef CAS PubMed.
- R. Pei, J. Liu, C. Y. Jing and M. Zhang, A multienzyme cascade pathway immobilized in a hydrogen-bonded organic framework for the conversion of CO2, Small, 2024, 20, 2306117 CrossRef CAS PubMed.
- J. B. Siegel, A. L. Smith, S. Poust, A. J. Wargacki, A. Bar-Even, C. Louw, B. W. Shen, C. B. Eiben, H. M. Tran, E. Noor, J. L. Gallaher, J. Bale, Y. Yoshikuni, M. H. Gelb, J. D. Keasling, B. L. Stoddard, M. E. Lidstrom and D. Baker, Computational protein design enables a novel one-carbon assimilation pathway, Proc. Natl. Acad. Sci. U. S. A., 2015, 112, 3704–3709 CrossRef CAS PubMed.
- M. J. Seo, Y. J. Jeong, S. B. Ju, D. M. Kim, H. R. Seo, H. F. Son, B. H. Sung, J. B. Park and S. J. Yeom, Enzymatic cascade transformation of renewable C1 and C2 alcohols into 3-hydroxypropionaldehyde with a high conversion rate, ACS Sustainable Chem. Eng., 2025, 13, 2694–2705 CrossRef CAS.
- T. Cai, H. B. Sun, J. Qiao, L. L. Zhu, F. Zhang, J. Zhang, Z. J. Tang, X. L. Wei, J. G. Yang, Q. Q. Yuan, W. Y. Wang, X. Yang, H. Y. Chu, Q. Wang, C. You, H. W. Ma, Y. X. Sun, Y. Li, C. Li, H. F. Jiang, Q. H. Wang and Y. H. Ma, Cell-free chemoenzymatic starch synthesis from carbon dioxide, Science, 2021, 373, 1523–1527 CrossRef CAS PubMed.
- J. A. Yang, W. Song, T. Cai, Y. Y. Wang, X. W. Zhang, W. Y. Wang, P. Chen, Y. Zeng, C. Li, Y. X. Sun and Y. H. Ma, De novo artificial synthesis of hexoses from carbon dioxide, Sci. Bull., 2023, 68, 2370–2381 CrossRef CAS PubMed.
- L. W. Lou, F. Y. Cheng, Z. L. Li and Z. M. Li, Constructing an artificial in vitro multi-enzyme cascade pathway to convert glycerol and CO2 into L-aspartic acid, Bioresour. Technol., 2024, 411, 131350 CrossRef CAS PubMed.
- X. W. Ding, J. Rong, Z. P. Pan, X. X. Zhu, Z. Y. Zhu, Q. Chen, Z. J. Zhang, J. H. Xu, C. X. Li and G. W. Zheng, Multienzyme synthetic pathways for lactic acid production, ACS Catal., 2024, 14, 4665–4674 CrossRef CAS.
- R. K. Singh, R. Singh, D. Sivakumar, S. Kondaveeti, T. Kim, J. L. Li, B. H. Sung, B. K. Cho, D. R. Kim, S. C. Kim, V. C. Kalia, Y. H. P. J. Zhang, H. M. Zhao, Y. C. Kang and J. K. Lee, Insights into cell-free conversion of CO2 to chemicals by a multienzyme cascade reaction, ACS Catal., 2018, 8, 11085–11093 CrossRef CAS.
- Y. Sheng, F. Guo, B. Guo, N. Wang, Y. Sun, H. Liu, X. Feng, Q. Han, Y. Yu and C. Li, Light-driven CO2 reduction with a surface-displayed enzyme cascade-C3N4 hybrid, ACS Synth. Biol., 2023, 12, 2715–2724 CrossRef CAS PubMed.
- L. K. Luan, Y. F. Zhang, X. L. Ji, B. X. Guo, S. Y. Song, Y. H. Huang and S. J. Zhang, Electro-driven multi-enzymatic cascade conversion of CO2 to ethylene glycol in nano-reactor, Adv. Sci., 2024, 11, 2407204 CrossRef CAS PubMed.
- P. P. Wang, X. Wang, S. Chandra, A. Lielpetere, T. Quast, F. Conzuelo and W. Schuhmann, Hybrid enzyme-electrocatalyst cascade modified gas-diffusion electrodes for methanol formation from carbon dioxide, Angew. Chem., Int. Ed., 2025, 64, e202422882 CrossRef CAS PubMed.
- Y. S. Lee, R. Gerulskis and S. D. Minteer, Advances in electrochemical cofactor regeneration: Enzymatic and non-enzymatic approaches, Curr. Opin. Biotechnol., 2022, 73, 14–21 CrossRef CAS PubMed.
- R. R. Wu, F. Li, X. Y. Cui, Z. H. Li, C. L. Ma, H. F. Jiang, L. L. Zhang, Y. H. P. J. Zhang, T. X. Zhao, Y. P. Zhang, Y. Li, H. Chen and Z. G. Zhu, Enzymatic electrosynthesis of glycine from CO2 and NH3, Angew. Chem., Int. Ed., 2023, 62, e202218387 CrossRef CAS PubMed.
- H. L. Li, Y. Z. Wu, Y. X. Wang, K. Zhang, J. Zhu, Y. Ji, T. Gu, W. X. Nie, L. C. Sun and Y. J. Wang, Bifunctional RhIII-complex-catalyzed CO2 reduction and NADH regeneration for direct bioelectrochemical synthesis of C3 and C4, ACS Catal., 2024, 14, 17201–17208 CrossRef CAS.
- X. Ning, F. Li, X. L. Wei, Z. G. Zhu and C. You, A light-powered synthetic enzymatic biosystem for the synthesis of 3-hydroxypropionic acid via CO2 fixation, ACS Synth. Biol., 2024, 13, 2611–2620 CrossRef CAS PubMed.
- J. M. Liu, H. Zhang, Y. Y. Xu, H. Meng and A. P. Zeng, Turn air-captured CO2 with methanol into amino acid and pyruvate in an ATP/NAD(P)H-free chemoenzymatic system, Nat. Commun., 2023, 14, 2772 CrossRef CAS PubMed.
- N. J. Claassens, S. Burgener, B. Vogeli, T. J. Erb and A. Bar-Even, A critical comparison of cellular and cell-free bioproduction systems, Curr. Opin. Biotechnol., 2019, 60, 221–229 CrossRef CAS PubMed.
- K. K. Sahoo, G. Goswami and D. Das, Biotransformation of methane and carbon dioxide into high-value products by methanotrophs: Current state of art and future prospects, Front. Microbiol., 2021, 12, 636486 CrossRef PubMed.
- J. D. Semrau, A. A. DiSpirito and J. C. Murrell, Life in the extreme: Thermoacidophilic methanotrophy, Trends Microbiol., 2008, 16, 190–193 CrossRef CAS PubMed.
- K. Schuchmann and V. Müller, Autotrophy at the thermodynamic limit of life: A model for energy conservation in acetogenic bacteria, Nat. Rev. Microbiol., 2014, 12, 809–821 CrossRef CAS PubMed.
- F. Y. H. Chen, H. W. Jung, C. Y. Tsuei and J. C. Liao, Converting Escherichia coli to a synthetic methylotroph growing solely on methanol, Cell, 2020, 182, 933–946 CrossRef CAS PubMed.
- M. A. Reiter, T. Bradley, L. A. Buechel, P. Keller, E. Hegedis, T. Gassler and J. A. Vorholt, A synthetic methylotrophic Escherichia coli as a chassis for bioproduction from methanol, Nat. Catal., 2024, 7, 560–573 CrossRef CAS PubMed.
- U. M. Vasudevan, L. Soussan, A. Fernández-Castané, S. Noack and A. D. Goddard, Yeast as C1 cell factory: Transforming methanol and formate into high-value compounds, Biotechnol. Adv., 2025, 83, 108641 CrossRef CAS PubMed.
- Z. L. Qian, J. H. Yu, X. J. Chen, Y. J. Kang, Y. N. Ren, Q. Liu, J. Lu, Q. Zhao and M. H. Cai, De novo production of plant 4′-deoxyflavones baicalein and oroxylin A from ethanol in crabtree-negative yeast, ACS Synth. Biol., 2022, 11, 1600–1612 CrossRef CAS PubMed.
- J. G. de la Cruz, F. Machens, K. Messerschmidt and A. Bar-Even, Core catalysis of the reductive glycine pathway demonstrated in yeast, ACS Synth. Biol., 2019, 8, 911–917 CrossRef PubMed.
- I. Sánchez-Andrea, I. A. Guedes, B. Hornung, S. Boeren, C. E. Lawson, D. Z. Sousa, A. Bar-Even, N. J. Claassens and A. J. M. Stams, The reductive glycine pathway allows autotrophic growth of desulfovibrio desulfuricans, Nat. Commun., 2020, 11, 5090 CrossRef PubMed.
- O. Yishai, M. Bouzon, V. Döring and A. Bar-Even, In vivo assimilation of one-carbon via a synthetic reductive glycine pathway in Escherichia coli, ACS Synth. Biol., 2018, 7, 2023–2028 CrossRef CAS PubMed.
- J. Z. Tian, W. S. Y. Deng, Z. W. Zhang, J. Q. Xu, G. L. Yang, G. P. Zhao, S. Yang, W. H. Jiang and Y. Gu, Discovery and remodeling of vibrio natriegens as a microbial platform for efficient formic acid biorefinery, Nat. Commun., 2023, 14, 7758 CrossRef CAS PubMed.
- B. M. Mitic, C. Troyer, L. Lutz, M. Baumschabl, S. Hann and D. Mattanovich, The oxygen-tolerant reductive glycine pathway assimilates methanol, formate and CO2 in the yeast Komagataella phaffii, Nat. Commun., 2023, 14, 7754 CrossRef CAS PubMed.
- Y. Gan, X. Meng, C. Gao, W. Song, L. Liu and X. Chen, Metabolic engineering strategies for microbial utilization of methanol, Eng. Microbiol., 2023, 3, 845–855 Search PubMed.
- W. Jiang, W. Newell, J. J. Liu, L. Coppens, K. B. Slater, H. D. Peng, D. Bell, L. Liu, V. Haritos and R. Ledesma-Amaro, Insights into the methanol utilization capacity of Y. lipolytica and improvements through metabolic engineering, Metab. Eng., 2025, 91, 170 CrossRef CAS PubMed.
- S. X. Wang, J. Y. Fang, M. Y. Wang, S. J. Yu, Y. Xia, G. X. Liu, Y. P. Zhang, Y. Li and T. C. Zhu, Rewiring the methanol assimilation pathway in the methylotrophic yeast Pichia pastoris for high-level production of erythritol, Bioresour. Technol., 2025, 427, 132430 CrossRef CAS PubMed.
- M. S. Jia, M. G. Liu, J. W. Li, W. K. Jiang, F. X. Xin, W. M. Zhang, Y. J. Jiang and M. Jiang, Formaldehyde: An essential intermediate for C1 metabolism and bioconversion, ACS Synth. Biol., 2024, 13, 3507–3522 CrossRef CAS PubMed.
- T. Wu, P. A. Gómez-Coronado, A. Kubis, S. N. Lindner, P. Marlière, T. J. Erb, A. Bar-Even and H. He, Engineering a synthetic energy-efficient formaldehyde assimilation cycle in Escherichia coli, Nat. Commun., 2023, 14, 8490 CrossRef CAS PubMed.
- B. J. Zhao, Y. Li, Y. Zhang, M. X. Pan, G. S. Zhao and Y. B. Guo, Low-carbon and overproduction of cordycepin from methanol using engineered Pichia pastoris cell factory, Bioresour. Technol., 2024, 413, 131446 CrossRef CAS PubMed.
- T. T. Niu, X. G. Yan, J. R. Wang, H. J. Song, Y. Cui, X. P. Cai, A. K. Chang, J. J. Qiao, M. Z. Wen and J. W. He, Engineering of pichia pastoris for the de novo synthesis of the sesquiterpene zealexin A1 from methanol, ACS Sustainable Chem. Eng., 2024, 12, 12786–12794 CrossRef CAS.
- J. Xu, J. Wang, C. L. Ma, Z. X. Wei, Y. D. Zhai, N. Tian, Z. G. Zhu, M. Xue and D. M. Li, Embracing a low-carbon future by the production and marketing of C1 gas protein, Biotechnol. Adv., 2023, 63, 108096 CrossRef CAS PubMed.
- Y. C. Ma, T. Liu, Z. G. Yuan and J. H. Guo, Single cell protein production from methane in a gas-delivery membrane bioreactor, Water Res., 2024, 259, 121820 CrossRef CAS PubMed.
- J. Feng, Y. Zhao, Z. D. Liu, X. Wang, S. Xu and K. Q. Chen, Engineered biosynthesis of phenol using acetate as the carbon source in Escherichia coli, ACS Sustainable Chem. Eng., 2025, 13, 1657–1666 CrossRef CAS.
- D. Kiefer, L. R. Tadele, L. Lilge, M. Henkel and R. Hausmann, High-level recombinant protein production with using acetate as carbon source, Microb. Biotechnol., 2022, 15, 2744–2757 CrossRef CAS PubMed.
- R. Kutscha and S. Pflügl, Microbial upgrading of acetate into value-added products-examining microbial diversity, bioenergetic constraints and metabolic engineering approaches, Int. J. Mol. Sci., 2020, 21, 8777 CrossRef CAS PubMed.
- P. Wang, B. Q. Li, B. Y. Li, J. Yang, X. R. Xu, S. T. Yang and X. Zou, Carbon-economic biosynthesis of poly-2-hydrobutanedioic acid driven by nonfermentable substrate ethanol, Green Chem., 2022, 24, 6599–6612 RSC.
- N. J. Claassens, C. A. R. Cotton, D. Kopljar and A. Bar-Even, Making quantitative sense of electromicrobial production, Nat. Catal., 2019, 2, 437–447 CrossRef CAS.
- G. Q. Chen, A microbial polyhydroxyalkanoates (PHA) based bio- and materials industry, Chem. Soc. Rev., 2009, 38, 2434–2446 RSC.
- S. Y. Lee, Deciphering bioplastic production, Nat. Biotechnol., 2006, 24, 1227–1229 CrossRef CAS PubMed.
- P. P. Peralta-Yahya, F. Z. Zhang, S. B. del Cardayre and J. D. Keasling, Microbial engineering for the production of advanced biofuels, Nature, 2012, 488, 320–328 CrossRef CAS PubMed.
- J. Lim, S. Y. Choi, J. W. Lee, S. Y. Lee and H. Lee, Biohybrid CO2 electrolysis for the direct synthesis of polyesters from CO2, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, 2221438120 CrossRef PubMed.
- H. T. Tang, L. H. Wu, S. Y. Guo, W. B. Cao, W. H. Ma, X. Wang, J. F. Shen, M. L. Wang, Q. N. Zhang, M. T. Huang, X. Z. Luo, J. Zeng, J. D. Keasling and T. Yu, Metabolic engineering of yeast for the production of carbohydrate-derived foods and chemicals from C1–3 molecules, Nat. Catal., 2024, 7, 21–34 CrossRef CAS.
- J. Bang, J. H. Ahn, J. A. Lee, C. H. Hwang, G. B. Kim, J. Lee and S. Y. Lee, Synthetic formatotrophs for one-carbon biorefinery, Adv. Sci., 2021, 8, 2100199 CrossRef CAS PubMed.
- U. Contaldo, M. Curtil, J. Pérard, C. Cavazza and A. Le Goff, A pyrene-triazacyclononane anchor affords high operational stability for CO2RR by a CNT-supported histidine-tagged CODH, Angew. Chem., Int. Ed., 2022, 61, 202117212 CrossRef PubMed.
- S. W. Wang, T. Y. Kou, J. B. Varley, S. A. Akhade, S. E. Weitzner, S. E. Baker, E. B. Duoss and Y. Li, Cu2O/CuS nanocomposites show excellent selectivity and stability for formate generation via electrochemical reduction of carbon dioxide, ACS Mater. Lett., 2021, 3, 100–109 CrossRef CAS.
- H. Wu, S.-J. Zhang, J. Vongsvivut, M. Jaroniec, J. Hao and S.-Z. Qiao, Aqueous zinc-iodine batteries with ultra-high loading and advanced performance, Joule, 2025, 9, 102000 CrossRef CAS.
- R. Mirea, E. Popescu and T. Zaharescu, Microbial electrosynthesis: The future of next-generation biofuel production–A review, Energies, 2025, 18, 5187 CrossRef CAS.
- A. Ronen, W. Duan, I. Wheeldon, S. Walker and D. Jassby, Microbial attachment inhibition through low-voltage electrochemical reactions on electrically conducting membranes, Environ. Sci. Technol., 2015, 49, 12741–12750 CrossRef CAS PubMed.
- M. Z. d. V. Gomes, G. Masdeu, P. Eiring, A. Kuhlemann, M. Sauer, B. Åkerman and A. E. C. Palmqvist, Improved biocatalytic cascade conversion of CO2 to methanol by enzymes Co-immobilized in tailored siliceous mesostructured cellular foams, Catal. Sci. Technol., 2021, 11, 6952–6959 RSC.
- J. Jan, I. Martinez, Y. P. Wang, G. N. Bennett and K. Y. San, Metabolic engineering and transhydrogenase effects on NADPH availability in Escherichia coli, Biotechnol. Prog., 2013, 29, 1124–1130 CrossRef CAS PubMed.
- T. Haas, R. Krause, R. Weber, M. Demler and G. Schmid, Technical photosynthesis involving CO2 electrolysis and fermentation, Nat. Catal., 2018, 1, 32–39 CrossRef CAS.
- I. S. Al Rowaihi, A. Paillier, S. Rasul, R. Karan, S. W. Grotzinger, K. Takanabe and J. Eppinger, Poly(3-hydroxybutyrate) production in an integrated electromicrobial setup: Investigation under stress-inducing conditions, PLoS One, 2018, 13, e0196079 CrossRef PubMed.
- R. Hegner, K. Neubert, C. Kroner, D. Holtmann and F. Harnisch, Coupled electrochemical and microbial catalysis for the production of polymer bricks, ChemSusChem, 2020, 13, 5295–5300 CrossRef CAS PubMed.
- I. S. Al Rowaihi, B. Kick, S. W. Grötzinger, C. Burger, R. Karan, D. Weuster-Botz, J. Eppinger and S. T. Arold, A two-stage biological gas to liquid transfer process to convert carbon dioxide into bioplastic, Bioresour. Technol. Rep., 2018, 1, 61–68 CrossRef.
- K. Zhang, Y. Zhou, T. Song and J. Xie, Bioplastic production from the microbial electrosynthesis of acetate through CO2 reduction, Energy Fuels, 2021, 35, 15978–15986 CrossRef CAS.
- P. Zhang, K. Chen, B. Xu, J. Li, C. Hu, J. S. Yuan and S. Y. Dai, Chem-bio interface design for rapid conversion of CO2 to bioplastics in an integrated system, Chem, 2022, 8, 3363–3381 CAS.
- K. A. Chen, P. Zhang, Y. Y. Chen, C. C. Fei, J. L. Yu, J. H. Zhou, Y. H. Liang, W. W. Li, S. S. Xiang, S. Y. Dai and J. S. Yuan, Electro-biodiesel empowered by co-design of microorganism and electrocatalysis, Joule, 2025, 9, 101769 CrossRef CAS.
- Y. Khojasteh-Salkuyeh, O. Ashrafi, E. Mostafavi and P. Navarri, CO2 utilization for methanol production; Part I: Process design and life cycle GHG assessment of different pathways, J. CO2 Util., 2021, 50, 101608 CrossRef CAS.
- T. T. Loan, D. T. Q. Trang, P. Q. Huy, P. X. Ninh and D. Van Thuoc, A fermentation process for the production of poly(3-hydroxybutyrate) using waste cooking oil or waste fish oil as inexpensive carbon substrate, Biotechnol. Rep., 2022, 33, e00700 CrossRef CAS PubMed.
- G. Y. Liu, Y. Zhong, Z. H. Liu, G. Wang, F. Gao, C. Zhang, Y. J. Wang, H. W. Zhang, J. Ma, Y. G. Hu, A. B. Chen, J. Y. Pan, Y. Z. Min, Z. Y. Tang, C. Gao and Y. J. Xiong, Solar-driven sugar production directly from CO2 via a customizable electrocatalytic–biocatalytic flow system, Nat. Commun., 2024, 15, 2636 CrossRef CAS PubMed.
- C. Minke, M. Suermann, B. Bensmann and R. Hanke-Rauschenbach, Is iridium demand a potential bottleneck in the realization of large-scale PEM water electrolysis?, Int. J. Hydrogen Energy, 2021, 46, 23581–23590 CrossRef CAS.
- R. Villa, S. Nieto, A. Donaire and P. Lozano, Direct biocatalytic processes for CO2 capture as a green tool to produce value-added chemicals, Molecules, 2023, 28, 5520 CrossRef CAS PubMed.
- L. Huang, Z. Y. Fu, H. Y. Yin, C. Xia, S. Cao, X. L. Weng, L. Shi and Z. B. Wu, Electrocatalytic utilization of air pollutants and GHGs: Fundamentals, electrode materials, and reactors, Environ. Sci. Technol., 2025, 59, 24177–24196 CrossRef CAS PubMed.
| This journal is © Institute of Process Engineering of CAS 2026 |