Open Access Article
Open Access ArticleInvestigating the SO2 absorption behavior of pyrimidine-based deep eutectic solvents via a dual-site thermodynamic model
Yiru Zouac,
Xiaoxiao Xinga,
Chao Wang c,
Duanjian Tao
c,
Duanjian Tao *d,
Haiyan Jic,
Peiwen Wu
*d,
Haiyan Jic,
Peiwen Wu *c,
Yanhong Chaoa and
Wenshuai Zhu
*c,
Yanhong Chaoa and
Wenshuai Zhu *abc
*abc
aCollege of Chemical Engineering and Environment, State Key Laboratory of Heavy Oil Processing, College of Science, China University of Petroleum-Beijing, Beijing 102249, PR China. E-mail: zhuws@cup.edu.cn
bShandong Key Laboratory of Green Electricity & Hydrogen Science and Technology, Shandong Institute of Petroleum and Chemical Technology, Dongying 257061, PR China
cSchool of Chemistry and Chemical Engineering, School of the Environment and Safety Engineering, School of Materials Science and Engineering, Jiangsu University, Zhenjiang 212013, PR China
dCollege of Chemistry and Chemical Engineering, Jiangxi Normal University, Nanchang 330022, PR China
First published on 14th January 2026
Abstract
Deep eutectic solvents (DESs), composed of hydrogen bond acceptors (HBAs) and hydrogen bond donors (HBDs), are widely used in flue gas desulfurization due to their excellent SO2 absorption properties. In this work, 14 DESs with pyrimidine derivatives and their isomers (including 2-aminopyrimidine (AmPyr), 2-chloropyrimidine, 2-bromopyrimidine, 4-amino-2-hydroxypyrimidine, 2,4-dihydroxypyrimidine, 4,6-diaminopyrimidene, 2-aminopyrazine, and 3-aminopyridazine, respectively) as HBDs and 1-ethyl-3-methylimidazolium chloride (C2mimCl) and 1-ethyl-3-methylimidazolium bromide as HBAs have been successfully prepared and used for SO2 absorption. Among them, C2mimCl-7 + AmPyr exhibited the highest SO2 absorption (19.032 mol kg−1, at 298.15 K and 1.0 bar), rapid gas–liquid equilibrium within 40 s, and an exceptional ideal selectivity of 528.7 for SO2/CO2. After 30 cycles of absorption–desorption, the SO2 absorption capacity remained as high as 18.265 mol kg−1. A dual-site reaction equilibrium thermodynamic model (DS-RETM) was established for absorption behavior analysis. Using C2mimCl-7 + AmPyr as a case study, Henry's constant, equilibrium constants, and other thermodynamic parameters were determined. DS-RETM fitting further enabled visualization of the potential absorption behavior of each DES component, facilitating comparison of HBA and HBD effects on SO2 absorption. This study offers new insights into the development of high-performance flue gas desulfurization absorbents and introduces a novel model for thermodynamic analysis of SO2 absorption in DESs.
Keywords: Deep eutectic solvents; SO2 absorption; Pyrimidine; Thermodynamic; Selectivity.
1 Introduction
The various fuels burned in factories during the production process, such as coal, oil, natural gas, etc., will produce a large amount of flue gas.1 The harmful gases contained therein, especially sulfur dioxide (SO2), cause serious damage to the ecological environment and pose a significant threat to human health.2 In the atmosphere, SO2 oxidizes into sulfuric acid mist or sulfate aerosols, which are important precursors for environmental acidification. The content of SO2 exceeding 0.5 ppm has potential impacts on human health, while when its concentration exceeds 1.5 ppm, healthy individuals may suffer from chronic bronchitis, laryngitis, and severe respiratory infections.3,4 Thus, it is necessary to remove SO2 before it is released into the atmosphere. Flue gas desulfurization (FGD) is the process of separating SO2 from flue gas through various technologies and converting it into harmless or usable substances.5 According to the state of the absorbent and product during the reaction process, it can be divided into wet, dry, and semi-dry desulfurization. Wet desulfurization technology is relatively mature, efficient, and easy to operate, and is the most widely used desulfurization method, including limestone gypsum method, ammonia absorption method, etc. However, these traditional absorbents have drawbacks such as irreversibility, volatility, and secondary environmental pollution.6 Developing efficient, selective, and renewable absorbents is one of the key challenges in FGD technology.Among all reported efficient absorbents, deep eutectic solvents (DESs) have been regarded as a promising one, because of their high selectivity, remarkable absorption capacity, green characteristics, etc.7 DESs were first proposed by Abbott in 2003 and are widely regarded as one of the green solvents.8,9 They are formed by the interaction between hydrogen bond donors (HBDs) and hydrogen bond acceptors (HBAs) through hydrogen bonds, halogen bonds, or van der Waals forces, and have advantages such as easy synthesis, recyclability, designability, etc.10–12 Due to the wide variety of HBDs and HBAs, theoretically, there could be at least 106 types of DESs, which can be designed with specific functions based on application requirements.13 Since Han et al.14 first used choline chloride-glycerol deep eutectic solvents to absorb SO2 in 2013, various DESs have been developed for flue gas desulfurization. Among them, DESs using nitrogen-containing heterocyclic organic compounds as HBDs or HBAs, including imidazole,15–18 triazole,19 tetrazole,20 benzimidazole,21 pyrazole,22 pyridine,23–26 etc., have been reported successively.
Pyrimidine, a six-membered nitrogen-containing heterocyclic organic compound, differs from pyridine in that it contains two alternating N atoms and has lower toxicity. Schröder et al.1 reported that the N atom of the pyrimidine ring can form dipole–dipole interactions with the O atom in S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, indicating that pyrimidine-based absorbents are excellent candidates for SO2 absorption. However, the physical interaction formed by electrostatic attraction between polar molecules is relatively weak, which limits the ability of materials to remove SO2. Notably, DESs prepared from pyridine derivatives containing functional groups such as –NH2, –OH, and –COOH exhibit excellent SO2 absorption capacity.25,26 Thus, to prepare pyrimidine-based DESs with high SO2 absorption performance, pyrimidine derivatives containing functional groups such as –NH2, C
O, indicating that pyrimidine-based absorbents are excellent candidates for SO2 absorption. However, the physical interaction formed by electrostatic attraction between polar molecules is relatively weak, which limits the ability of materials to remove SO2. Notably, DESs prepared from pyridine derivatives containing functional groups such as –NH2, –OH, and –COOH exhibit excellent SO2 absorption capacity.25,26 Thus, to prepare pyrimidine-based DESs with high SO2 absorption performance, pyrimidine derivatives containing functional groups such as –NH2, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, –Cl, and –Br have been inferred to enhance their interactions with SO2.
O, –Cl, and –Br have been inferred to enhance their interactions with SO2.
Ulteriorly, to investigate the absorption behavior, the SO2 absorption process of DESs was analyzed through a thermodynamic model. Zhang et al.27 obtained the physical and chemical absorption enthalpy changes (ΔrHm) of acidic protonic ionic liquid (APIL) based DESs through a reaction equilibrium thermodynamic model (RETM). In our previous work,28 we also successfully used a RETM to demonstrate that purine-based DESs exhibit chemical absorption in the low-pressure region and physical absorption in the high-pressure region, and obtained a series of thermodynamic parameters. The absorption behavior of different DESs can be observed through the RETM, but how to gain more comprehensive insight into the absorption capacity of each component still needs further exploration.
In this work, a series of DESs with pyrimidine derivatives (one of 2-aminopyrimidine, 2-chloropyrimidine, 2-bromopyrimidine, 4-amino-2-hydroxypyrimidine, 2,4-dihydroxypyrimidine, and 4,6-diaminopyrimidene) or isomers of 2-aminopyrimidine (2-aminopyrazine or 3-aminopyridazine) as HBDs with 1-ethyl-3-methylimidazolium chloride or 1-ethyl-3-methylimidazolium bromide as HBAs have been successfully prepared. The relationship between temperature and the densities as well as viscosities of the prepared DESs was analyzed, and the thermal decomposition temperature (Td) was further determined through thermogravimetric analysis. Subsequently, the SO2 absorption performance of the prepared DESs was measured to investigate the effects of the molar ratio (HBAs![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HBDs), HBD type, pressure, and temperature. Moreover, the time-dependent SO2 absorption, SO2/CO2 selectivity and regeneration performance of pyrimidine-based DESs were also explored. Specifically, the physical and chemical absorption behavior of the prepared DESs was investigated using a dual-site reaction equilibrium thermodynamic model (DS-RETM), which can further elucidate the influence of HBAs and HBDs on SO2 absorption and thermodynamic parameters. Finally, the possible interaction between pyrimidine-based DESs and SO2 was further elucidated. Our findings offer a novel strategy for the design of high-performance, high-selectivity SO2 absorbents and introduce a new thermodynamic model for SO2 absorption in DESs.
HBDs), HBD type, pressure, and temperature. Moreover, the time-dependent SO2 absorption, SO2/CO2 selectivity and regeneration performance of pyrimidine-based DESs were also explored. Specifically, the physical and chemical absorption behavior of the prepared DESs was investigated using a dual-site reaction equilibrium thermodynamic model (DS-RETM), which can further elucidate the influence of HBAs and HBDs on SO2 absorption and thermodynamic parameters. Finally, the possible interaction between pyrimidine-based DESs and SO2 was further elucidated. Our findings offer a novel strategy for the design of high-performance, high-selectivity SO2 absorbents and introduce a new thermodynamic model for SO2 absorption in DESs.
2 Results and discussion
2.1 Characterization of pyrimidine-based DESs
The densities of the as-prepared DESs with C2mimCl as a HBA were measured at different temperatures, as shown in Fig. 1a and b (Tables S1 and S2). Obviously, as the molar ratio increases from 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 7
1 to 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the density of C2mimCl-n + AmPyr DESs gradually decreases from 1.164 g cm−3 to 1.151 g cm−3 at 298.15 K. Thus, the increase of C2mimCl is beneficial to decrease the density of C2mimCl-n + AmPyr DESs. As for the DESs of C2mimCl-7 + HBDs, the order of density (arranged by HBDs) at the same temperature is: AmPz < AmPd ≈ AmPyr < ChPyr < DAmPyr < Cyt < Ura < BrPyr. Considering that the molar ratio of C2mimCl to HBDs remains unchanged, there exists a significant difference in density, indicating that the molecular structure of HBDs will affect the density of DESs significantly.29 The density of DESs was fitted to the temperature (T) (ρDESs = α + βT, detailed parameters are provided in Tables S3 and S4). The results show that R12 and R22 are 0.999, revealing a linear relationship between densities and temperatures.
1, the density of C2mimCl-n + AmPyr DESs gradually decreases from 1.164 g cm−3 to 1.151 g cm−3 at 298.15 K. Thus, the increase of C2mimCl is beneficial to decrease the density of C2mimCl-n + AmPyr DESs. As for the DESs of C2mimCl-7 + HBDs, the order of density (arranged by HBDs) at the same temperature is: AmPz < AmPd ≈ AmPyr < ChPyr < DAmPyr < Cyt < Ura < BrPyr. Considering that the molar ratio of C2mimCl to HBDs remains unchanged, there exists a significant difference in density, indicating that the molecular structure of HBDs will affect the density of DESs significantly.29 The density of DESs was fitted to the temperature (T) (ρDESs = α + βT, detailed parameters are provided in Tables S3 and S4). The results show that R12 and R22 are 0.999, revealing a linear relationship between densities and temperatures.
 | ||
| Fig. 1 The densities of (a) C2mimCl-n + AmPyr (n = 2, 3, 4, 5, 6, 7) and (b) C2mimCl-7 + HBDs; the viscosities of (c) C2mimCl-n + AmPyr (n = 2, 3, 4, 5, 6, 7) and (d) HBAs-7 + HBDs. | ||
Subsequently, the viscosities of pyrimidine-based DESs were measured at different temperatures, as shown in Fig. 1c and d (Tables S5 and S6). The results in Fig. 1c are similar to the effect of temperature on densities, indicating that increasing C2mimCl is beneficial for reducing its viscosities, which may be due to the formation of more intramolecular hydrogen bonds.30,31 In Fig. 1d, the order of viscosities (at 298.15 K) for different C2mimCl-7 + HBDs DESs is: C2mimCl-7 + BrPyr < C2mimCl-7 + ChPyr < C2mimCl-7 + AmPz < C2mimCl-7 + Ura < C2mimCl-7 + AmPd < C2mimCl-7 + AmPyr < C2mimCl-7 + DAmPyr < C2mimCl-7 + Cyt. Owing to the different types, positions, and numbers of functional groups in HBDs, their ability to form intramolecular hydrogen bonds with C2mimCl differs from one another, demonstrating that the structures of HBAs and HBDs significantly alter the viscosities of DESs.29 In addition, the viscosity of C2mimCl-7 + AmPyr is higher than that of C2mimBr-7 + AmPyr, which may be due to the different anions of HBAs, leading to the weaker ability of C2mimBr and AmPyr to form intermolecular hydrogen bonds.32 The viscosities were fitted to the temperature (T) (VFT equation: η = η0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(t/(T − T0)),33 and the detailed parameter data are shown in Tables S7 and 1). The results indicate that R32 and R42 are 0.999, suggesting an exponential relationship between viscosities and temperatures.
exp(t/(T − T0)),33 and the detailed parameter data are shown in Tables S7 and 1). The results indicate that R32 and R42 are 0.999, suggesting an exponential relationship between viscosities and temperatures.
| Parameters | HBDs | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Cyt | AmPyr | DAmPyr | AmPd | Ura | AmPz | ChPyr | AmPyr* | BrPyr | |
| AmPyr*: C2mimBr-7 + AmPyr. | |||||||||
| η0 × 104 (Pa s) | 6.591 ± 3.095 | 5.847 ± 2.319 | 4.233 ± 1.846 | 9.167 ± 3.933 | 13.50 ± 4.797 | 15.90 ± 3.597 | 12.20 ± 2.858 | 14.00 ± 8.586 | 18.60 ± 4.826 |
| t2 (K) | 696.7 ± 83.61 | 601.2 ± 59.98 | 800.0 ± 85.88 | 563.1 ± 66.87 | 522.0 ± 58.61 | 448.2 ± 35.34 | 492.1 ± 41.15 | 495.7 ± 115.5 | 396.2 ± 46.08 |
| T0 (K) | 208.9 ± 5.231 | 220.7 ± 3.644 | 200.0 ± 5.216 | 219.7 ± 4.482 | 217.2 ± 4.495 | 222.0 ± 2.981 | 214.0 ± 3.567 | 209.1 ± 10.69 | 215.4 ± 5.018 |
| R42 | 0.999 | 0.999 | 0.999 | 0.999 | 0.999 | 0.999 | 0.999 | 0.999 | 0.999 |
| Td (K) | 508 | 443 | 503 | 514 | 503 | 519 | 464 | 444 | 393 |
To investigate the thermal stability of pyrimidine-based DESs, HBAs-7 + HBDs were analyzed by thermogravimetry (TG), as shown in Fig. S1 and Table 1. Thermal decomposition temperature (Td) can be used to judge the absorption temperature of absorbent and desorption temperature during regeneration, which is generally defined as the temperature at which the mass loss exceeds 5%. The maximum Td value is 519 K (C2mimCl-7 + AmPz), and the minimum value is 393 K (C2mimCl-7 + BrPyr), indicating that pyrimidine-based DESs have excellent thermal stability during absorption–desorption processes and have the potential for flue gas desulfurization at high temperature.
2.2 SO2 absorption of pyrimidine-based DESs
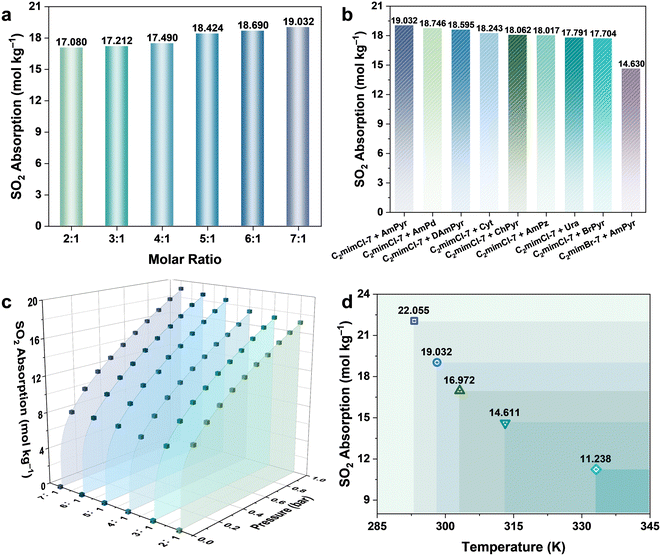
To explore the optimal molar ratio of HBAs to HBDs, the SO2 absorption capacity of C2mimCl-n + AmPyr was investigated. Fig. 2a shows the SO2 absorption of C2mimCl-n + AmPyr at 298.15 K and 1.0 bar. As the molar ratio of C2mimCl to AmPyr increases, the SO2 absorption of C2mimCl-n + AmPyr increases gradually, reaching a maximum of 19.032 mol kg−1. With the increase of C2mimCl, the viscosity of C2mimCl-n + AmPyr decreases, which is more beneficial to mass transfer;34 nevertheless, the charge-transfer interaction between the highly electronegative Cl− in C2mimCl and the S atoms in SO2 (Cl−⋯SO2) is beneficial for improving SO2 absorption.35,36 According to the above analysis, the optimal molar ratio of C2mimCl to AmPyr is 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.
1.
Furthermore, a series of HBAs-7 + HBDs DESs were synthesized, and their SO2 absorption capacity is shown in Fig. 2b. Besides, Table S8 shows the SO2 absorption capacity of synthesized DESs compared to other materials, which are superior to many solid absorbents. For DESs with different HBAs, the absorption capacity of C2mimBr-7 + AmPyr (14.630 mol kg−1) is significantly lower compared to C2mimCl-7 + AmPyr (19.032 mol kg−1), indicating that anions have a greater influence on SO2 absorption capacity. In other words, the greater the electronegativity, the higher the absorption capacity of SO2.29,32 As isomers of AmPyr, AmPd and AmPz were also used to form DESs with C2mimCl. The SO2 absorption capacities of C2mimCl-7 + AmPz and C2mimCl-7 + AmPd are 18.017 mol kg−1 and 18.746 mol kg−1, respectively. These values, all lower than that of C2mimCl-7 + AmPyr (19.032 mol kg−1), demonstrate that the position of the nitrogen atoms within the heterocyclic ring structure of the HBDs also significantly impacts the performance of DESs, and the pyrimidine ring is more conducive to SO2 absorption.
Hence, to develop pyrimidine-based DESs with superior performance, the SO2 absorption of DESs using pyrimidine derivatives with different functional groups as HBDs was investigated. Firstly, with BrPyr, ChPyr, and AmPyr as HBDs, the SO2 absorption capacity is C2mimCl-7 + BrPyr < C2mimCl-7 + ChPyr < C2mimCl-7 + AmPyr. Typically, the higher the viscosity, the smaller the absorption capacity. Here, the main reason that affects the absorption capacity is that AmPyr contains weak basic functional groups (–NH2), which may have acid–base interaction with SO2 molecules.37 Then, the SO2 absorption of DESs with multifunctional pyrimidine derivatives (Ura, Cyt, and DAmPyr) as HBDs was further investigated. The SO2 absorption of the three DESs is C2mimCl-7 + Ura (17.791 mol kg−1) < C2mimCl-7 + Cyt (18.243 mol kg−1) < C2mimCl-7 + DAmPyr (18.595 mol kg−1). Thus, the pyrimidine-based DESs with optimal SO2 absorption is C2mimCl-7 + AmPyr.
It is universally known that pressure plays a key role in affecting the gas absorption capacity of DESs.38 Fig. 2c shows the SO2 absorption of pyrimidine-based DESs at 298.15 K and 0–1.0 bar. The SO2 absorption capacity of C2mimCl-n + AmPyr increases nonlinearly with the increase of pressure. Taking C2mimCl-7 + AmPyr with the highest absorption capacity as an example, with the pressure increasing from 0.1 bar to 1.0 bar, the absorption capacity of SO2 increases from 7.911 mol kg−1 to 19.032 mol kg−1. This shows that increasing the pressure can promote the absorption of SO2. Conversely, reducing the pressure can be considered to desorb SO2.39,40 Additionally, the SO2 absorption increases rapidly between 0 and 0.1 bar, probably owing to the chemical interaction between the absorbent and SO2 molecules.41 However, the growth rate of absorption capacity decreases between 0.1 and 1.0 bar, which may be due to the gradual occupation of absorption sites, so rapid absorption has been completed, resulting in the process being dominated by physical absorption. Therefore, it is speculated that the absorption process of SO2 via pyrimidine-based DESs includes chemical absorption and physical absorption.42
According to the existing literature,6,22,43–46 temperature is another important factor affecting the absorption capacity of DESs. Hence, C2mimCl-7 + AmPyr was selected to investigate the SO2 absorption performance at different temperatures, as shown in Fig. 2d. With the increase of temperature, the SO2 absorption capacity of C2mimCl-7 + AmPyr decreases continuously, and the absorption capacities are 22.055, 19.032, 16.972, 14.611 and 11.238 mol kg−1, respectively. This result shows that increasing the temperature is beneficial to the regeneration of absorbent. Furthermore, it should be noted that C2mimCl-7 + AmPyr still has a high absorption capacity at high temperatures, indicating that pyrimidine-based DESs could be used as candidate absorbents for high-temperature flue gas desulfurization.29
2.3 Analysis of SO2 absorption behavior for pyrimidine-based DESs
A DS-RETM equation was established for fitting the SO2 absorption.47 It is assumed that the absorption of SO2 by C2mimCl-7 + AmPyr DESs involves the following steps:| SO2 (g) → SO2 (l) | (1) |
| SO2 (g) + C2mimCl (l) → C2mimCl–SO2 (l) | (2) |
| SO2 (g) + AmPyr (l) → AmPyr–SO2 (l) | (3) |
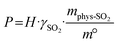 | (4) |
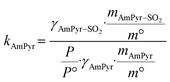 | (5) |
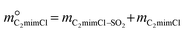 | (6) |
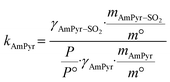 | (7) |
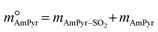 | (8) |
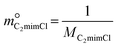 | (9) |
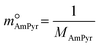 | (10) |
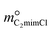 and
and 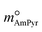 are constants (mol kg−1) that could be calculated using eqn (9) and (10); MC2mimCl and MAmPyr are the molar mass of C2mimCl and AmPyr, respectively, in g mol−1.
are constants (mol kg−1) that could be calculated using eqn (9) and (10); MC2mimCl and MAmPyr are the molar mass of C2mimCl and AmPyr, respectively, in g mol−1.
The values of γSO2, γC2mimCl–SO2, γC2mimCl, γAmPyr–SO2, and γAmPyr are all regarded as 1.0.28,48 Thus, the DS-RETM could be expressed by eqn (11) and (12):
| mt = mphys-SO2 + mCmimCl–SO2 + mAmPyr–SO2 | (11) |
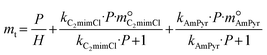 | (12) |
The SO2 absorption of C2mimCl-7 + AmPyr at different temperatures and pressures was correlated according to eqn (12), and fitted lines are shown in Fig. 3a. Notably, the fitting curve matches well with the experimental data, with R52 values greater than 0.999, indicating that the established thermodynamic model has extremely high reliability. The corresponding fitting parameters were calculated and revealed in Table 2. The Henry's law constants keep an increasing trend, while the equilibrium constants keep a decreasing trend, which is consistent with the negative dependence of SO2 absorption on temperature.
 | ||
| Fig. 3 (a) DS-DETM equation fitting of C2mimCl-7 + AmPyr for SO2 absorption; the calculated (b) physical and (c) chemical absorption of SO2 by C2mimCl-7 + AmPyr. | ||
| Parameters | 293.15 K | 303.15 K | 313.15 K | 333.15 K |
|---|---|---|---|---|
| H (bar) | 0.10814 ± 0.00279 | 0.21585 ± 0.00688 | 0.30983 ± 0.01596 | 0.66939 ± 0.16117 |
| kC2mimCl | 173.03801 ± 72.62040 | 99.15970 ± 14.25614 | 36.28764 ± 2.19755 | 15.10983 ± 0.82593 |
| kAmPyr | 1.27796 ± 0.09255 | 1.11052 ± 0.05036 | 0.80265 ± 0.04400 | 0.46775 ± 0.06859 |
| R52 | 0.999 | 0.999 | 0.999 | 0.999 |
| ΔphysHm (kJ mol−1) | −35.993 | −35.993 | −35.993 | −35.993 |
| ΔphysGm (kJ mol−1) | −5.421 | −3.864 | −3.051 | −1.112 |
| ΔphysSm (J mol K−1) | −104.287 | −105.983 | −105.197 | −104.701 |
| ΔchemHm (kJ mol−1) | −72.131 | −72.131 | −72.131 | −72.131 |
| ΔchemGm (kJ mol−1) | −13.158 | −11.850 | −8.779 | −5.416 |
| ΔchemSm (J mol K−1) | −201.169 | −198.85 | −202.308 | −200.254 |
| ΔrHm (kJ mol−1) | −108.124 | −108.124 | −108.124 | −108.124 |
| ΔrGm (kJ mol−1) | −18.579 | −15.714 | −11.829 | −6.528 |
| ΔrSm (J mol K−1) | −305.457 | −304.833 | −307.505 | −304.955 |
Based on the H, kC2mimCl, and kAmPyr, the mphys-SO2, mC2mimCl–SO2 and mAmPyr–SO2 could be obtained. As shown in Fig. 3b and c, both physical absorption and chemical absorption decrease with increasing temperature. The mphys-SO2 is most significantly controlled by temperature, followed by mAmPyr–SO2, and mC2mimCl–SO2 is least affected, indicating that the interaction between C2mimCl and SO2 is stronger than AmPyr, and the physical interaction is the weakest. Besides, the physical absorption capacity increases linearly with increasing pressure, while the chemical absorption capacity increases nonlinearly and gradually approaches equilibrium. Thus, the absorption capacity of DESs for SO2 gradually increases linearly with the increase of pressure.
According to the relationship of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H and ln
H and ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k to 1/T (Fig. 4a), the parameters of enthalpy change (ΔHm), Gibbs free energy change (ΔGm), and entropy change (ΔSm) could be obtained by the following equations:
k to 1/T (Fig. 4a), the parameters of enthalpy change (ΔHm), Gibbs free energy change (ΔGm), and entropy change (ΔSm) could be obtained by the following equations:
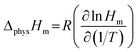 | (13) |
ΔphysGm = RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Hm Hm
| (14) |
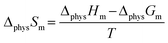 | (15) |
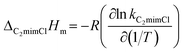 | (16) |
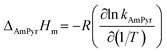 | (17) |
| ΔchemHm = ΔC2mimClHm + ΔAmPyrHm | (18) |
ΔchemGm = −RT(ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) kC2mimCl + ln kC2mimCl + ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) kAmPyr) kAmPyr)
| (19) |
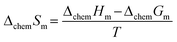 | (20) |
 | ||
Fig. 4 (a) The fitting results of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H and ln H and ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k to 1/T of C2mimCl-7 + AmPyr; (b) the calculated SO2 absorption of C2mimCl-7 + AmPyr at 298.15 K. k to 1/T of C2mimCl-7 + AmPyr; (b) the calculated SO2 absorption of C2mimCl-7 + AmPyr at 298.15 K. | ||
Here, ΔphysHm, ΔphysGm, and ΔphysSm correspond to the physical process; ΔC2mimClHm, ΔAmPyrHm, ΔchemHm, ΔchemGm, and ΔchemSm correspond to the chemical process (Table 2). As can be seen, the values of ΔHm, ΔGm, and ΔSm are always less than 0, which indicate the absorption process is exothermic, spontaneous, and with increased orderliness.
Besides, the ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H, ln
H, ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) kC2mimCl, and ln
kC2mimCl, and ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) kAmPyr at 298.15 K can be calculated, and the SO2 absorption of mphys, mC2mimCl, mAmPyr, and mft could be further obtained. Fig. 4b shows that the calculated SO2 absorption remains consistent with the experimental values (such as 19.051 mol kg−1 vs. 19.032 mol kg−1 at 1.0 bar), indicating the high reliability of the DS-RETM equation.
kAmPyr at 298.15 K can be calculated, and the SO2 absorption of mphys, mC2mimCl, mAmPyr, and mft could be further obtained. Fig. 4b shows that the calculated SO2 absorption remains consistent with the experimental values (such as 19.051 mol kg−1 vs. 19.032 mol kg−1 at 1.0 bar), indicating the high reliability of the DS-RETM equation.
To further investigate the absorption behavior of different pyrimidine-based DESs, 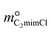 and
and 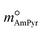 in eqn (12) were replaced with the initial molality of the corresponding HBAs and HBDs (
in eqn (12) were replaced with the initial molality of the corresponding HBAs and HBDs (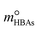 and
and 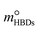 ), resulting in DS-RETM equations for different HBAs-7 + HBDs DESs. Subsequently, the SO2 absorption of different pyrimidine-based DESs at 298.15 K was fitted, and the detailed results are shown in Fig. S2 and Table S9. Based on the obtained H, kHBAs, and kHBDs, the mphys, mHBAs, and mHBDs of corresponding HBAs-7 + HBDs DESs could be calculated separately, which is of great significance for insight exploration of the absorption behavior of each component in DESs (Fig. S3). Compared with C2mimBr-7 + AmPyr, the mHBDs, mHBAs, and mphys of C2mimCl-7 + AmPyr were significantly increased, indicating that Cl− has a key impact on the SO2 absorption of DESs. Obviously, for DESs using the same HBAs (C2mimCl-7 + HBDs), there is not much difference in mHBAs (also known as mC2mimCl) (Fig. S3a and b), attributed to the same molar mass of C2mimCl, which forms a strong charge transfer interaction between Cl− and the S atom of SO2 (Cl−⋯SO2). However, significant differences in mHBDs and mphys between C2mimCl-7 + HBDs DESs could be observed. Notably, the order of mHBDs is not the same as the SO2 absorption of their corresponding DESs (Fig. S3c and d). Among them, mAmPd and mDAmPyr are both greater than mAmPyr, but the SO2 absorption of C2mimCl-7 + AmPyr is greater than that of C2mimCl-7 + AmPd and C2mimCl-7 + DAmPyr. Interestingly, for the physical absorption (mphys) of C2mimCl-7 + HBDs (Fig. S3e and f), there is even a phenomenon opposite to the SO2 absorption of the corresponding DESs. For instance, C2mimCl-7 + BrPyr has the minimum SO2 absorption but the maximum mphys. To sum up, the types and numbers of functional groups in HBDs determine their varying abilities to absorb SO2, while also affecting the physical absorption properties of the corresponding DESs. Hence, DS-RETM fitting could visualize these potential absorption behaviors, which is more conducive to exploring the physical and chemical absorption processes of the prepared DESs.
), resulting in DS-RETM equations for different HBAs-7 + HBDs DESs. Subsequently, the SO2 absorption of different pyrimidine-based DESs at 298.15 K was fitted, and the detailed results are shown in Fig. S2 and Table S9. Based on the obtained H, kHBAs, and kHBDs, the mphys, mHBAs, and mHBDs of corresponding HBAs-7 + HBDs DESs could be calculated separately, which is of great significance for insight exploration of the absorption behavior of each component in DESs (Fig. S3). Compared with C2mimBr-7 + AmPyr, the mHBDs, mHBAs, and mphys of C2mimCl-7 + AmPyr were significantly increased, indicating that Cl− has a key impact on the SO2 absorption of DESs. Obviously, for DESs using the same HBAs (C2mimCl-7 + HBDs), there is not much difference in mHBAs (also known as mC2mimCl) (Fig. S3a and b), attributed to the same molar mass of C2mimCl, which forms a strong charge transfer interaction between Cl− and the S atom of SO2 (Cl−⋯SO2). However, significant differences in mHBDs and mphys between C2mimCl-7 + HBDs DESs could be observed. Notably, the order of mHBDs is not the same as the SO2 absorption of their corresponding DESs (Fig. S3c and d). Among them, mAmPd and mDAmPyr are both greater than mAmPyr, but the SO2 absorption of C2mimCl-7 + AmPyr is greater than that of C2mimCl-7 + AmPd and C2mimCl-7 + DAmPyr. Interestingly, for the physical absorption (mphys) of C2mimCl-7 + HBDs (Fig. S3e and f), there is even a phenomenon opposite to the SO2 absorption of the corresponding DESs. For instance, C2mimCl-7 + BrPyr has the minimum SO2 absorption but the maximum mphys. To sum up, the types and numbers of functional groups in HBDs determine their varying abilities to absorb SO2, while also affecting the physical absorption properties of the corresponding DESs. Hence, DS-RETM fitting could visualize these potential absorption behaviors, which is more conducive to exploring the physical and chemical absorption processes of the prepared DESs.
2.4 Kinetic analysis of SO2 absorption for pyrimidine-based DESs
A typical time-dependent SO2 absorption process involves taking a certain amount of DESs (∼0.10 g) and observing the change in SO2 absorption capacity over time at an initial pressure of ∼1.0 bar. The time-dependent SO2 absorption capacity of C2mimCl-7 + HBDs at 298.15 K is shown in Fig. 5a. Interestingly, the as-prepared DESs can absorb SO2 rapidly, which suggests that the absorption process is mainly dominated by chemical absorption. To further observe the SO2 absorption rate, Boltzmann equation (eqn (21)) fitting was performed on the experimental results,32 and the first derivative of the curve is the absorption rate of the as-prepared pyrimidine-based DESs (Fig. 5b). Clearly, C2mimCl-7 + AmPyr achieved the maximum absorption rate in the shortest possible time. Moreover, the as-prepared DESs can rapidly absorb SO2 and basically achieve absorption equilibrium within 40 s, which is superior to numerous reported DESs.35,49–51 This result shows that pyrimidine-based DESs have potential for flue gas desulfurization.
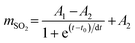 | (21) |
 | ||
| Fig. 5 (a) The time-dependent SO2 absorption and (b) SO2 absorption rate of pyrimidine-based DESs; T = 298.15 K. | ||
2.5 SO2/CO2 absorption selectivity of pyrimidine-based DESs
Owing to the complex composition of actual flue gas, it generally contains 0.04–15% CO2.52 Thus, the designed absorbent for flue gas desulfurization should have high SO2/CO2 selectivity. The CO2 and SO2 absorption capacities of C2mimCl-7 + HBDs were compared at 298.15 K and 1.0 bar, as shown in Fig. 6. Obviously, pyrimidine-based DESs show a negative absorption of CO2. Generally, the selectivity of SO2/CO2 keeps the ratio of SO2 absorption capacity to CO2 absorption capacity under given conditions (SDESs = mSO2/mCO2).29 The comparison of SO2 absorption capacity and selectivity with literature data is shown in Table S10. Accordingly, the absorption selectivity of pyrimidine-based DESs is C2mimCl-7 + ChPyr (451.6), C2mimCl-7 + Ura (494.2), C2mimCl-7 + AmPyr (528.7), C2mimCl-7 + BrPyr (536.5), C2mimCl-7 + DAmPyr (808.5), C2mimCl-7 + Cyt (1073.1), respectively. The minimum value of SDESs has reached up to 451.6, indicating that pyrimidine-based DESs have excellent SO2/CO2 selective absorption performance and could be used as a candidate for selective absorption of SO2 from actual flue gas.2.6 SO2 absorption mechanism of pyrimidine-based DESs
To explore the mechanism of DES formation and SO2 absorption, C2mimCl-7 + AmPyr with the highest absorption capacity was selected for FT-IR and 1H NMR analysis. In Fig. 7a, the peaks appearing at 3325 and 3161 cm−1 are attributed to the stretching vibration of –N(g)H2, the peak around 1645 cm−1 is attributed to the bending vibration of –N(g)H2, and two peaks at 1575 and 1556 cm−1 are attributed to the stretching vibration of the pyrimidine ring.53 In DES (C2mimCl-7 + AmPyr), the peak of –N(g)H2 around 1645 cm−1 is red shifted to 1628 cm−1, indicating that AmPyr had hydrogen bond interactions with C2mimCl (N(g)–H⋯Cl−). Besides, the peak of C(2)–H in C2mimCl around 3056 cm−1 shifts to 3046 cm−1,51 which may be due to Cl− forming intramolecular hydrogen bonds (C(2)–H⋯Cl−), leading to a decrease in force constant and vibrational energy, and a red shift in functional groups.23,29,54For the spectrum after SO2 absorption, four new peaks attributed to SO2 could be observed. Among them, three peaks are attributed to the bending vibration (529 cm−1), symmetric stretching vibration (1124 cm−1), and asymmetric stretching vibration (1281 cm−1) of S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, respectively.6,22,32 Compared with free SO2, a significant red shift was observed in the stretching vibration of S
O, respectively.6,22,32 Compared with free SO2, a significant red shift was observed in the stretching vibration of S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, indicating that Cl− in C2mimCl may form a charge transfer interaction with S atoms in SO2 (Cl−⋯SO2).55,56 Hence, the original hydrogen bonds in DESs get weakened significantly, leading to the peaks of –C(8)H3, C(2)–H and C(4+5)–H shifting from 2976, 3046 and 3140 cm−1 to 2984, 3088 and 3145 cm−1, respectively.32,49 The fourth peak around 836 cm−1 is attributed to the bending vibration of S–O, indicating that there exists chemical absorption of SO2.28,57 Owing to the bending vibration of –N(g)H2 shifting and splitting into two peaks around 1693 and 1680 cm−1, it is inferred that the acid–base interactions might be formed between AmPyr and SO2.23 Contrary to SO2 absorption, the FT-IR spectrum after CO2 absorption showed no significant changes compared to before absorption, indicating that there is almost no interaction between the as-prepared DESs and CO2, thus exhibiting extremely high SO2/CO2 selectivity.
O, indicating that Cl− in C2mimCl may form a charge transfer interaction with S atoms in SO2 (Cl−⋯SO2).55,56 Hence, the original hydrogen bonds in DESs get weakened significantly, leading to the peaks of –C(8)H3, C(2)–H and C(4+5)–H shifting from 2976, 3046 and 3140 cm−1 to 2984, 3088 and 3145 cm−1, respectively.32,49 The fourth peak around 836 cm−1 is attributed to the bending vibration of S–O, indicating that there exists chemical absorption of SO2.28,57 Owing to the bending vibration of –N(g)H2 shifting and splitting into two peaks around 1693 and 1680 cm−1, it is inferred that the acid–base interactions might be formed between AmPyr and SO2.23 Contrary to SO2 absorption, the FT-IR spectrum after CO2 absorption showed no significant changes compared to before absorption, indicating that there is almost no interaction between the as-prepared DESs and CO2, thus exhibiting extremely high SO2/CO2 selectivity.
Fig. 7b shows the 1H NMR spectra of AmPyr, C2mimCl, C2mimCl-7 + AmPyr, and C2mimCl-7 + AmPyr after SO2 absorption. Notably, the peak of –N(g)H2 shifts from 6.59 ppm in AmPyr to 6.66 ppm in DES, and the peak of C(2)–H shifts from 9.70 ppm in C2mimCl to 9.78 ppm in DES. These down-field shifts are attributed to the formation of N(g)–H⋯Cl− and C(2)–H⋯Cl−,23 respectively, which would reduce electron cloud density (de-shielding effect).25,58 Concurrently, the peak of C(4+5)–H shifts to down-field, while the peaks of C(d+f)–H and C(e)–H shift to up-field. In contrast, the peaks of C(2)–H and –N(g)H2 shift to up-field after SO2 absorption. The former is attributed to the charge transfer between the Cl− and S atoms (Cl−⋯SO2), while the latter could be ascribed to the acid–base interaction between –N(g)H2 and SO2.25,59 The shift of other C–H signals in C2mimCl and AmPyr indicates that the original hydrogen bonds in DES have been disrupted by SO2.
2.7 Regeneration of pyrimidine-based DESs
In order to avoid secondary pollution caused by a large number of waste absorbents, it is usually required that the absorbents have good regeneration performance. As shown in Fig. 8, C2mimCl-7 + AmPyr maintains the SO2 absorption capacity of 18.265 mol kg−1 after 30 times of absorption–desorption. This represents excellent regeneration performance compared with the reported DESs for absorbing SO2.16,22,23,38,60 To investigate whether the structure of pyrimidine-based DESs is stable and maintains the original absorption performance, the SO2 absorption capacity of C2mimCl-7 + AmPyr was measured every 7 days under the same conditions (Fig. S4). The results showed that C2mimCl-7 + AmPyr remained liquid after 4 weeks, and maintained excellent SO2 absorption performance.3 Conclusions
In this work, 14 different DESs were successfully developed and used for SO2 absorption. The density and viscosity of the as-prepared DESs show a negative correlation with temperature, but the former is linear and the latter is exponential. As the molar ratio of C2mimCl to AmPyr increases, the SO2 absorption capacity of C2mimCl-n + AmPyr gradually increases, ultimately reaching up to 19.032 mol kg−1 at 298.15 K and 1.0 bar. The thermodynamic analysis of SO2 absorption by C2mimCl-7 + AmPyr at different temperatures was conducted using the DS-RETM equation, which not only yielded a series of thermodynamic parameters, but also successfully calculated the SO2 absorption capacity (19.051 mol kg−1) that is highly consistent with the experimental values at 298.15 K, indicating that this model has extremely high reliability. Subsequently, it was further applied to analyze the absorption behavior of C2mimCl-7 + HBDs DESs, and the corresponding mphys, mC2mimCl, and mHBDs were obtained, demonstrating the impact of different HBDs on SO2 absorption. Dynamics analysis shows that the as-prepared DESs could reach gas–liquid equilibrium in 40 s. Besides, the present DESs exhibit remarkable SO2/CO2 selectivity, with C2mimCl-7 + AmPyr achieving a value as high as 528.7. After 30 cycles of absorption–desorption, the SO2 absorption of the regenerated C2mimCl-7 + AmPyr still reached 18.265 mol kg−1. Finally, combining thermodynamic analysis, FT-IR, and 1H NMR results, it is indicated that the SO2 absorption process involves physical absorption and chemical absorption.4 Experimental section
Preparation of DESs: C2mimCl-n + AmPyr DESs (n = 2, 3, 4, 5, 6, and 7) were prepared by combining C2mimCl and AmPyr at specified molar ratios (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 3
1, 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 4
1, 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 5
1, 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 6
1, 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 7
1, 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). The mixtures underwent heating at 353.15 K for 180 min with constant stirring. Subsequent cooling to ambient temperature yielded homogeneous liquids (Scheme S2).
1). The mixtures underwent heating at 353.15 K for 180 min with constant stirring. Subsequent cooling to ambient temperature yielded homogeneous liquids (Scheme S2).
C2mimCl-7 + ChPyr, C2mimCl-7 + BrPyr, C2mimCl-7 + Cyt, C2mimCl-7 + DAmPyr, C2mimCl-7 + Ura, C2mimCl-7 + AmPd and C2mimCl-7 + AmPz were prepared using 2-chloropyrimidine, 2-bromopyrimidine, 4-amino-2-hydroxypyrimidine, 4,6-diaminopyrimidene, 2,4-dihydroxypyrimidine, 3-aminopyridazine and 2-aminopyrazine as HBDs, respectively.
C2mimBr-7 + AmPyr was prepared using C2mimBr as a HBA and AmPyr as a HBD.
At a molar ratio of 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 353.15 K, with C2mimCl as a HBA and TAmPyr as a HBD, DESs could not be formed after vigorous stirring at 800 rpm for 180 min.
1 and 353.15 K, with C2mimCl as a HBA and TAmPyr as a HBD, DESs could not be formed after vigorous stirring at 800 rpm for 180 min.
For other details not described, please refer to the SI.
Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5im00317b.
Acknowledgements
All authors appreciate the financial support from the National Science Foundation for Distinguished Young Scholars (No. 22425808), the National Natural Science Foundation of China (No. 22178154, 22578187), the Science Foundation of China University of Petroleum, Beijing (No. 2462022YJRC002, 2462022YJRC003), and the Carbon Neutrality Research Institute Fund (No. CNIF20240103).References
- W. Li, J. Li, T. D. Duong, S. A. Sapchenko, X. Han, J. D. Humby, G. F. S. Whitehead, I. J. Victórica-Yrezábal, I. da Silva, P. Manuel, M. D. Frogley, G. Cinque, M. Schröder and S. Yang, Adsorption of sulfur dioxide in Cu(II)-carboxylate framework materials: The role of ligand functionalization and open metal sites, J. Am. Chem. Soc., 2022, 144, 13196–13204 CrossRef CAS PubMed.
- X. Bie, R. Wu, B. Yu, X. Quan, S. Zhang, Q. Li, Y. Zhang and H. Zhou, Deactivation mechanisms of Cu–Zn–Al2O3 in CO2 hydrogenation induced by SO2 exposure, Ind. Chem. Mater., 2025, 3, 710–722 Search PubMed.
- X. Cao, Y. Liu, Q. Huang, Z. Chen, J. Sun, J. Sun, S. Pang, P. Liu, W. Wang, Y. Zhang and M. Ge, Single droplet tweezer revealing the reaction mechanism of Mn(II)-catalyzed SO2 oxidation, Environ. Sci. Technol., 2024, 58, 5068–5078 CrossRef CAS PubMed.
- W. Zhao, J. L. Obeso, V. B. López-Cervantes, M. Bahri, E. Sánchez-González, Y. A. Amador-Sánchez, J. Ren, N. D. Browning, R. A. Peralta, G. Barcaro, S. Monti, D. Solis-Ibarra, I. A. Ibarra and D. Zhao, Achieving sub-ppm sensitivity in SO2 detection with a chemically stable covalent organic framework, Angew. Chem., Int. Ed., 2024, 64, e202415088 Search PubMed.
- W. Gong, P. Gao, Y. Gao, Y. Xie, J. Zhang, X. Tang, K. Wang, X. Wang, X. Han, Z. Chen, J. Dong and Y. Cui, Modulator-directed counterintuitive catenation control for crafting highly porous and robust metal–organic frameworks with record high SO2 uptake capacity, J. Am. Chem. Soc., 2024, 146, 31807–31815 Search PubMed.
- Q. Zhu, C. Wang, J. Yin, H. Li, W. Jiang, J. Liu, P. Li, Q. Zhang, Z. Chen and W. Zhu, Efficient and remarkable SO2 capture: A discovery of imidazole-based ternary deep eutectic solvents, J. Mol. Liq., 2021, 330, 115595 Search PubMed.
- Z. Yan, S. Y. Lai, C. L. Ngan, H. Li and A. R. Mohamed, Recent advances in energy-efficient and regenerative SO2 absorption over deep eutectic solvents, J. Environ. Chem. Eng., 2022, 10, 108967 Search PubMed.
- A. P. Abbott, G. Capper, D. L. Davies, R. K. Rasheed and V. Tambyrajah, Novel solvent properties of choline chloride/urea mixtures, Chem. Commun., 2003, 70–71 Search PubMed.
- Y. Chen and Z. Yu, Low-melting mixture solvents: Extension of deep eutectic solvents and ionic liquids for broadening green solvents and green chemistry, Green Chem. Eng., 2024, 5, 409–417 CrossRef CAS.
- M. Hu, B. Han, L. Xie, B. Lu, D. Bai, N. Shi, Y. Liao, Y. Wang, L. Liu, S. Wu, R. Lan, X. Lei, C. Shi, D. Huang, Y. Li, L. Lin and J. Zhang, Ultrasonic assisted natural deep eutectic solvents as a green and efficient approach for extraction of hydroxytyrosol from olive leaves, Ind. Chem. Mater., 2024, 2, 309–320 RSC.
- Y. Chen, G. Zhao, J. Dong, J. Wang, D. Dong, Z. Li, M. Zhao, Z. Shi and Z. Niu, Green recovery of all-solid-state sodium-ion batteries/lithium-ion batteries by ionic liquids, deep eutectic solvents and low-melting mixture solvents, Ind. Chem. Mater., 2025, 3, 464–474 RSC.
- E. L. Smith, A. P. Abbott and K. S. Ryder, Deep eutectic solvents (DESs) and their applications, Chem. Rev., 2014, 114, 11060–11082 CrossRef CAS PubMed.
- M. Francisco, A. van den Bruinhorst and M. C. Kroon, Low-transition-temperature mixtures (LTTMs): A new generation of designer solvents, Angew. Chem., Int. Ed., 2013, 52, 3074–3085 Search PubMed.
- D. Yang, M. Hou, H. Ning, J. Zhang, J. Ma, G. Yang and B. Han, Efficient SO2 absorption by renewable choline chloride–glycerol deep eutectic solvents, Green Chem., 2013, 15, 2261 Search PubMed.
- D. Deng, X. Liu and B. Gao, Physicochemical properties and investigation of azole-based deep eutectic solvents as efficient and reversible SO2 absorbents, Ind. Eng. Chem. Res., 2017, 56, 13850–13856 CrossRef CAS.
- K. Zhang, S. Ren, X. Yang, Y. Hou, W. Wu and Y. Bao, Efficient absorption of low-concentration SO2 in simulated flue gas by functional deep eutectic solvents based on imidazole and its derivatives, Chem. Eng. J., 2017, 327, 128–134 CrossRef CAS.
- D. Lee, W. Y. Choi, K. Jang, J. Park and Y. Yoo, Functionalized imidazole–alkanolamine deep eutectic solvents with remarkable performance for low-concentration SO2 absorption, Sep. Purif. Technol., 2023, 307, 122782 CrossRef CAS.
- X. Li, L. Meng, F. Yang, Z. Yang, J. Li, Y. Chen and X. Ji, Tuning the structure of N-methyldiethanolamine-based deep eutectic solvents for efficient and reversible SO2 capture, Chem. Commun., 2024, 60, 10560–10563 Search PubMed.
- G. Long, C. Yang, X. Yang, T. Zhao, F. Liu and J. Cao, Bisazole-based deep eutectic solvents for efficient SO2 absorption and conversion without any additives, ACS Sustainable Chem. Eng., 2020, 8, 2608–2613 CrossRef CAS.
- L. Zhang, H. Ma, G. Wei, B. Jiang, Y. Sun, X. Tantai, Z. Huang and Y. Chen, Efficient and reversible nitric oxide absorption by low-viscosity, azole-derived deep eutectic solvents, J. Chem. Eng. Data, 2019, 64, 3068–3077 CrossRef CAS.
- Y. Chen, B. Jiang, H. Dou, L. Zhang, X. Tantai, Y. Sun and H. Zhang, Highly efficient and reversible capture of low partial pressure SO2 by functional deep eutectic solvents, Energy Fuels, 2018, 32, 10737–10744 CrossRef CAS.
- P. Zhang, G. Xu, M. Shi, Z. Wang, Z. Tu, X. Hu, X. Zhang and Y. Wu, Unexpectedly efficient absorption of low-concentration SO2 with phase-transition mechanism using deep eutectic solvent consisting of tetraethylammonium chloride and imidazole, Sep. Purif. Technol., 2022, 286, 120489 CrossRef CAS.
- Q. Wang, H. Wu, T. Zhang, Y. Fan, W. Zhang and K. He, Efficient absorption of low partial pressure SO2 by deep eutectic solvents based on pyridine derivatives, Chem. Eng. Res. Des., 2022, 177, 36–44 Search PubMed.
- Z. Wang and D. Yang, SO2 capture by 2-pyridineethanol through the formation of a zwitterionic liquid, Chem. Commun., 2022, 58, 6212–6214 Search PubMed.
- C. Wang, H. Wu, J. Li, J. Zhang, J. Zhang, J. Ding, H. Li, H. Li and W. Zhu, Surpassingly efficient, selective, and reversible absorption of SO2 through pyridine based deep eutectic solvents, Chem. Eng. J., 2023, 471, 144394 CrossRef CAS.
- T. Yang, Y. Wang, Z. Huang, F. Liu, Q. Liao and T. Zhao, Deep eutectic solvents composed of 1-methyl-3-ethylimidazole halides and pyridine derivatives for efficient absorption and conversion of SO2 into cyclic sulfites under ambient conditions, Sep. Purif. Technol., 2025, 361, 131475 CrossRef CAS.
- P. Zhang, Z. Tu, X. Zhang, X. Hu and Y. Wu, Acidic protic ionic liquid-based deep eutectic solvents capturing SO2 with low enthalpy changes, AIChE J., 2023, 69, e18145 CrossRef CAS.
- Y. Zou, C. Wang, H. Ji, P. Wu, Y. Chao, X. Yu, Z. Yu, H. Liu, Z. Liu and W. Zhu, Rapid, selectivity, and reversibility absorption of SO2 via purine-based deep eutectic solvents and thermodynamic analysis, Green Chem. Eng., 2026, 7, 180–190 CrossRef.
- P. Li, X. Wang, T. Zhao, C. Yang, X. Wang and F. Liu, Deep eutectic solvents formed by EmimCl plus lactams: Effective SO2 capture and conversion into sulphur via DESs-mediated claus process, Chem. Eng. J., 2021, 422, 130033 CrossRef CAS.
- X. Y. Luo, X. Fan, G. L. Shi, H. R. Li and C. M. Wang, Decreasing the viscosity in CO2 capture by amino-functionalized ionic liquids through the formation of intramolecular hydrogen bond, J. Phys. Chem. B, 2016, 120, 2807–2813 CrossRef CAS PubMed.
- M. Pan, Y. Zhao, X. Zeng and J. Zou, Efficient absorption of CO2 by introduction of intramolecular hydrogen bonding in chiral amino acid ionic liquids, Energy Fuels, 2018, 32, 6130–6135 CrossRef CAS.
- K. Sheng, D. Li and Y. Kang, Unexpectedly promoted SO2 capture in novel ionic liquid-based eutectic solvents: The synergistic interactions, J. Mol. Liq., 2021, 337, 116432 CrossRef CAS.
- Y. Cao, J. Zhang, Y. Ma, W. Wu, K. Huang and L. Jiang, Designing low-viscosity deep eutectic solvents with multiple weak-acidic groups for ammonia separation, ACS Sustainable Chem. Eng., 2021, 9, 7352–7360 CrossRef CAS.
- G. Long, C. Yang, X. Yang, T. Zhao and M. Xu, Deep eutectic solvents consisting of 1-ethyl-3-methylimidazolium chloride and glycerol derivatives for highly efficient and reversible SO2 capture, J. Mol. Liq., 2020, 302, 112538 CrossRef CAS.
- B. Jiang, H. Zhang, L. Zhang, N. Zhang, Z. Huang, Y. Chen, Y. Sun and X. Tantai, Novel deep eutectic solvents for highly efficient and reversible absorption of SO2 by preorganization strategy, ACS Sustainable Chem. Eng., 2019, 7, 8347–8357 CrossRef CAS.
- X. Wu, R. Guan, W. Zheng and K. Huang, New deep eutectic solvents formed by 1-ethyl-3-methylimidazolium chloride and dicyandiamide: Physiochemical properties and SO2 absorption performance, J. Taiwan Inst. Chem. Eng., 2021, 119, 45–51 Search PubMed.
- D. Deng, G. Han and Y. Jiang, Investigation of a deep eutectic solvent formed by levulinic acid with quaternary ammonium salt as an efficient SO2 absorbent, New J. Chem., 2015, 39, 8158–8164 Search PubMed.
- C. Wang, G. Cui, X. Luo, Y. Xu, H. Li and S. Dai, Highly efficient and reversible SO2 capture by tunable azole-based ionic liquids through multiple-site chemical absorption, J. Am. Chem. Soc., 2011, 133, 11916–11919 CrossRef CAS PubMed.
- Y. Jiang, X. Liu and D. Deng, Solubility and thermodynamic properties of SO2 in three low volatile urea derivatives, J. Chem. Thermodyn., 2016, 101, 12–18 Search PubMed.
- D. Yang, Y. Han, H. Qi, Y. Wang and S. Dai, Efficient absorption of SO2 by EmimCl-EG deep eutectic solvents, ACS Sustainable Chem. Eng., 2017, 5, 6382–6386 CrossRef CAS.
- K. Zhang, S. Ren, Y. Hou and W. Wu, Efficient absorption of SO2 with low-partial pressures by environmentally benign functional deep eutectic solvents, J. Hazard. Mater., 2017, 324, 457–463 Search PubMed.
- G. Cui, J. Liu, S. Lyu, H. Wang, Z. Li and J. Wang, Efficient and reversible SO2 absorption by environmentally friendly task-specific deep eutectic solvents of PPZBr + Gly, ACS Sustainable Chem. Eng., 2019, 7, 14236–14246 Search PubMed.
- Z.-M. Li, W.-Q. Gong, J.-F. Li, S.-X. Zhu, D.-J. Tao and Y. Zhou, Efficient and selective absorption of SO2 by low-viscosity matrine-based deep eutectic solvents, J. Mol. Liq., 2022, 367, 120521 Search PubMed.
- Y. Zhao, J. Dou, R. Dai, H. Bai, S. Khoshk Rish, X. Xiao and J. Yu, Superefficient absorption of low-concentration SO2 in flue gas using new ternary imidazole-based deep eutectic solvents: Mechanism, thermodynamics, and process analysis, Energy Fuels, 2022, 36, 8351–8359 CrossRef CAS.
- Y. Zhao, J. Dou, H. Li, R. Dai, H. Bai, S. Khoshk Rish, X. Chen, X. Xiao and J. Yu, Low-cost Na2S-EG-MTPB deep eutectic solvents absorb SO2 effectively at a high temperature in flue gas, Sep. Purif. Technol., 2022, 303, 122283 CrossRef CAS.
- T. Zhou, Y. Zhao, X. Xiao, Y. Liu, H. Bai, X. Chen, J. Dou and J. Yu, Effective absorption mechanism of SO2 and NO2 in the flue gas by ammonium-bromide-based deep eutectic solvents, ACS Omega, 2022, 7, 29171–29180 CrossRef CAS PubMed.
- W. J. Jiang, F. Y. Zhong, L. S. Zhou, H. L. Peng, J. P. Fan and K. Huang, Chemical dual-site capture of NH3 by unprecedentedly low-viscosity deep eutectic solvents, Chem. Commun., 2020, 56, 2399–2402 RSC.
- P. Liu, K. Cai, X. Zhang, X. Wang, M. Xu, F. Liu and T. Zhao, Rich ether-based protic ionic liquids with low viscosity for selective absorption of SO2 through multisite interaction, Ind. Eng. Chem. Res., 2022, 61, 5971–5983 CrossRef CAS.
- D. Yang, S. Zhang, D. E. Jiang and S. Dai, SO2 absorption in EmimCl-TEG deep eutectic solvents, Phys. Chem. Chem. Phys., 2018, 20, 15168–15173 RSC.
- T. Zhao, J. Liang, Y. Zhang, Y. Wu and X. Hu, Unexpectedly efficient SO2 capture and conversion to sulfur in novel imidazole-based deep eutectic solvents, Chem. Commun., 2018, 54, 8964–8967 RSC.
- M. Lv, D. Yang and J. Chen, Deep eutectic solvents consisting of 1-ethyl-3-methylimidazolium chloride and biobased 2-pyrrolidone for reversible SO2 capture, ChemistrySelect, 2020, 5, 7142–7147 Search PubMed.
- H. Wang, P. Wu, C. Li, J. Zhang and R. Deng, Reversible and efficient absorption of SO2 with natural amino acid aqueous solutions: Performance and mechanism, ACS Sustainable Chem. Eng., 2022, 10, 4451–4461 CrossRef CAS.
- S. Akyüz and T. Akyüz, FT-IR spectroscopic investigations of surface and intercalated 2-aminopyrimidine adsorbed on sepiolite and montmorillonite from Anatolia, J. Mol. Struct., 2003, 651–653, 205–210 CrossRef.
- C. Wang, Q. Bi, Y. Huo, Z. Zhang, D. Tao, Y. Shen, Q. Zhu, Z. Chen, H. Li and W. Zhu, Investigation of amine-based ternary deep eutectic solvents for efficient, rapid, and reversible SO2 absorption, Energy Fuels, 2021, 35, 20406–20410 CrossRef CAS.
- X. Yang, Y. Zhang, F. Liu, P. Chen, T. Zhao and Y. Wu, Deep eutectic solvents consisting of EmimCl and amides: Highly efficient SO2 absorption and conversion, Sep. Purif. Technol., 2020, 250, 117273 Search PubMed.
- K.-Y. Lee, C.-S. Kim, H.-G. Kim, M.-S. Cheong, D. K. Mukherjee and K.-D. Jung, Effects of halide anions to absorb SO2 in ionic liquids, Bull. Korean Chem. Soc., 2010, 31, 1937–1940 CrossRef CAS.
- Z. L. Li, L. S. Zhou, Y. H. Wei, H. L. Peng and K. Huang, Highly efficient, reversible, and selective absorption of SO2 in 1-ethyl-3-methylimidazolium chloride plus imidazole deep eutectic solvents, Ind. Eng. Chem. Res., 2020, 59, 13696–13705 CrossRef CAS.
- W. S. Ham, H. Choi, J. Zhang, D. Kim and S. Chang, C2-selective, functional-group-divergent amination of pyrimidines by enthalpy-controlled nucleophilic functionalization, J. Am. Chem. Soc., 2022, 144, 2885–2892 Search PubMed.
- C. Wang, J. Zhang, Y. Zou, J. Ding, H. Li, M. Zhang, H. Li and W. Zhu, Effects of attractive electrostatic interactions on sulfur dioxide capture by functionalized deep eutectic solvents with abundant negative sites, Chem. Eng. J., 2025, 519, 165030 Search PubMed.
- S. Sun, Y. Niu, Q. Xu, Z. Sun and X. Wei, Efficient SO2 absorptions by four kinds of deep eutectic solvents based on choline chloride, Ind. Eng. Chem. Res., 2015, 54, 8019–8024 Search PubMed.
| This journal is © Institute of Process Engineering of CAS 2026 |