Open Access Article
Open Access ArticleLinking zeolite structures to reactivity in the carbonylation of dimethyl ether
Dong Jae Jeonga,
Xu Wang†
 b,
Namjun Parka,
Donggyu Leea,
Yongkyu Leea,
Jong Wook Bae
b,
Namjun Parka,
Donggyu Leea,
Yongkyu Leea,
Jong Wook Bae *b,
Won Bo Lee
*b,
Won Bo Lee *a and
Jong Hun Kang
*a and
Jong Hun Kang *a
*a
aSchool of Chemical and Biological Engineering and the Institute of Chemical Processes, Seoul National University, Seoul 08826, Republic of Korea. E-mail: wblee@snu.ac.kr; jonghunkang@snu.ac.kr
bSchool of Chemical Engineering, Sungkyunkwan University (SKKU), 2066 Seobu-ro, Jangan-gu, Suwon, Gyeonggi-do 16419, Republic of Korea. E-mail: finejw@skku.edu
First published on 3rd January 2026
Abstract
Carbonylation of dimethyl ether (DME) to methyl acetate (MA) is one of the key reactions for converting syngas into valuable chemicals and renewable energy sources. The reaction is predominantly catalyzed at Brønsted acid sites (BASs) on the 8-membered ring (8-MR) of the zeolite, which stabilizes the transient compound [Z–CH3–CO]‡ of the rate-determining step (RDS). Although many studies focus on local descriptors, such as the RDS activation energy and enthalpy or BAS site geometry, these models often fail to fully describe the complex interplay among BAS distribution, pore connectivity, and the three-dimensional frameworks. To develop a more globalized approach for clarifying the structure-reactivity correlation, we synthesized various 8-MR-possessing zeolites, including CHA, FER, LTA, ERI, AEI, and STI with different Si/Al ratios and evaluated their activity (normalized per acid site) in the DME carbonylation reaction. PXRD, SEM(EDS), and BET characterization confirmed the successful synthesis of each zeolite, and NH3-TPD and pyridine-IR analyses quantified their acid properties. Density functional theory (DFT) calculations show that the activation barrier of the RDS and the stability of the most abundant surface intermediate vary depending on the zeolite topology and active site environment. In this work, it was confirmed that “pocket-like” structures connecting two 8-MRs consecutively, which make 8-MR channels, have the most influential effect on catalytic behavior. Moreover, an adsorption isovolume-based descriptor, namely DF, which captures the 3-dimensional distribution of BAS and the accessible reaction space, is introduced based on thorough geometry analyses and correlates specifically with the transition state stabilization in the RDS. It is noteworthy that MOR and FER exhibited the highest turnover frequency, correlated with their unique BAS arrangement and favorable channel geometry for transition state stabilization, as predicted by the isovolume-based descriptor proposed in this work. Consequently, extracting 3-dimensional structural descriptors that correlate with the RDS is essential for accurate prediction of structure–reactivity relationships. This contribution provides an effective design principle for untested zeolite topologies and may guide the rational design of advanced catalysts for DME carbonylation and related shape-selective processes.
Keywords: Dimethyl ether carbonylation; Zeolites; Brønsted acid site (BAS); Isovolume; 3-Dimensional structural descriptors.
1 Introduction
Acetic acid is an essential chemical in the production of adhesives, paints, and synthetic fabrics like rayon, latex, and nylon.1–4 Generally, it is produced through catalytic methanol carbonylation processes such as the Monsanto and Cativa processes, which rely on Rh or Ir organometallic catalysts and iodide promoters. However, these processes face critical drawbacks such as the high cost of precious metal catalysts, high-pressure operating conditions (30–60 bar), and environmental challenges related to halide treatment.5–7In response to these issues, acidic zeolite catalysts are considered promising alternatives for dimethyl ether (DME) carbonylation yielding methyl acetate (MA). Not only by eliminating the need for precious metals or halides but also by allowing operation under milder conditions, zeolites improve both the economic feasibility and sustainability. Moreover, converting syngas (CO + H2) from waste-derived sources, such as biomass gasification, plastic pyrolysis, or CO2 recycling, into value-added MA, not only provides environmental benefits but also marks the beginning of its role as a platform intermediate for the synthesis of a broad range of key industrial chemicals, such as methanol, ethanol, acetic acid, methyl iodide, vinyl acetate, and ketenes, etc.8–10 In particular, acetic acid can be produced from methyl acetate (MA) via simple hydrolysis, followed by hydrogenation to yield ethanol. This sequential conversion presents a promising example of carbon capture and utilization (CCU) technology, enabling the valorization of carbon-containing by-products generated by the existing chemical industry.11
Several studies indicate that this DME carbonylation reaction typically takes place at Brønsted acid sites (BASs) on the 8-membered ring (8-MR) pores of zeolites, where the transition states of adsorbed reactants are effectively stabilized.8,12–17 Notably, mordenite (MOR) and ferrierite (FER), which both contain 8-MR pore openings in their channel systems, exhibit superior reactivity. Despite considerable research into the correlation between DME carbonylation reactivity and the 8-MR structures,18–20 there have been a limited number of comparative studies to evaluate whether these findings hold true for other 8-MR-possessing zeolites.
More in-depth studies have focused on elucidating the intrinsic role of the 8-MR structure. These investigations use density functional theory (DFT) calculations to analyze the reaction energies associated with BASs located within 8-MR pores.21,22 By calculating energy levels and activation energies for a sequence of elementary reactions during the DME carbonylation cycle, researchers have explained why MOR and FER show favorable reactivity. The DFT calculations commonly target two key elementary steps, based on the DME carbonylation mechanism (Fig. 1): the methylation step and the CO addition step.
The methylation step involves DME or methanol interacting with BASs within the zeolite micropores, leading to hydrogenolysis and substitution of the acid site by a methyl group (eqn (1) and (2)), where Z denotes the zeolite framework.
| CH3OCH3 + [Z–O(H)] ⇄ CH3OH + [Z–O(CH3)] | (1) |
| CH3OH + [Z–O(H)] ⇄ H2O + [Z–O(CH3)] | (2) |
CO addition, the key step in DME carbonylation, converts SMGs into surface acetyl groups (SAGs), which are subsequently esterified by additional DME to produce the primary product, MA (eqn (3) and (4)).
| [Z–O(CH3)] + CO → [Z–O(CH3CO)] | (3) |
| [Z–O(CH3CO)] + CH3OCH3 → CH3COOCH3 + [Z–O(CH3)] | (4) |
Boronat et al.14 confirmed the most active BAS for DME carbonylation in MOR by comparing DFT-predicted activation energies for the RDS and the geometric characteristics of the transient species. In this study, we adopt a similar approach but designate certain ‘effective’ sites (see section S1, Criteria for selecting effective Brønsted acid sites for DFT calculations in the SI), based on the crystallographic information file (cif) data from the International Zeolite Association (IZA) database (Fig. 2a).23 Zeolites possess frameworks of repeating tetrahedral sites (T-sites) connected by bridging oxygen atoms (O-sites), each labelled as TxOy (where x = 1, 2, 3…; y = 1, 2, 3…). Also, it is well-known that several T- and O-sites within a single zeolite framework are crystallographically equivalent. Based on this nomenclature, Boronat et al. emphasizes that the T3O8 sites of MOR are the most reactive active sites (Fig. 2c).
 | ||
| Fig. 2 MOR and FER frameworks and their substructures. (a) MOR channel system, (b) FER channel system, (c) detailed graphic of 8-MR side pockets of MOR and a series of surface methyl groups (SMGs) substituted for Brønsted acid on T3O8 sites. The reactant gases can diffuse through the cyan space, and especially in the side pocket of MOR, and can easily be added to central carbon of the SMG in the orthogonal direction (nucleophilic backside attack, see also Fig. 6); (d) schematic representation of t-dah natural tiling and staggered 8-MR pockets, which are stacked along the a-axis, and (e) schematic representation of t-fer natural tiling and the FER cage. The labels of T4O7 and T2O5 sites in FER are colored black. [Atom color] brown: carbon, white: hydrogen, gray: tetrahedral site (silicon or aluminum), red: oxygen. | ||
The T3O8 sites are located within the 8-MR side pockets of MOR, arranged in a staggered configuration along the c-axis, continuously distributed along the channel (Fig. 2d). CO addition at these sites generally experiences a lower activation energy barrier than competing pathways involving other molecules such as CH3OH, H2O, or DME. Thus, a more favorable pathway for CO is provided when the DME carbonylation occurs at these T3O8 BAS sites. Furthermore, the orthogonal bonding of CO to the SMG ([Z–O(CH3)] in eqn (4)) at the MOR T3O8 site facilitates the nucleophilic attack required to form acetyl species (Fig. 2c). In MOR synthesized with a low Si/Al ratio, aluminum tends to be concentrated at the T3 and T4 sites, with protons (H+) favoring O8 or O10, further enhancing the catalytic reactivity of MOR.24
Feng et al.15 used a similar approach to analyze FER, identifying T4O7 and T2O5 (following the site numbering convention of the IZA cif files)23 on the 6-MR unit of the 8-MR side channel as the most active sites for DME carbonylation in the FER-type zeolites (Fig. 2b and e). During the methylation step, they investigated the correlation between reaction enthalpy and the distance between the surface methyl group central carbon (CSMG) and the framework oxygen (OFER), noting a generally inverse relationship. However, T4O7 and T2O5 did not follow this trend, showing deviation from the lowest methylation enthalpy.
In the subsequent CO addition step, Feng et al. observed that all TxOy sites in FER require higher activation energies for CO addition compared to competing compounds, including CH3OH, H2O, or DME, yet T4O7 and T2O5 show the lowest CO activation energy. Notably, the CO addition activation energy for FER (0.829–1.762 eV) determined in this study15 is lower than that of MOR (1.451–1.866 eV).14 In comparison, MOR has a far lower methylation enthalpy (0.008–0.363 eV)14 than FER (0.083–0.933 eV).15 These corroborate with Feng's conclusion that the superior reactivity of MOR is due to its lower methylation energy, suggesting a milestone for comparing the reactivity of different 8-MR zeolites.
In this study, we attempted to derive the generalized relationship between the 8-MR structure and the DME carbonylation reactivity by synthesizing not only ZSM-35 (FER) but also SSZ-13 (CHA), zeolite A (LTA), UZM-12 (ERI), SSZ-39 (AEI), and TNU-10 (STI) within the practically feasible synthesizable range of Si/Al (note: MOR was tested using a commercial catalyst, CMOR). Details of the synthesis procedures are provided in section S2 in the SI with their representative organic structure directing agents (OSDAs) (see Fig. S3). All samples were characterized by PXRD (Fig. S4–S9), SEM(EDS) (Fig. S10), and BET analyses (Fig. S11 and Table S1) to confirm successful synthesis. NH3-TPD and pyridine-IR (Fig. S12–S20 and Table S2) analyses were conducted for acid site quantification, and representative NH3-TPD profiles for zeolites with the investigated topologies are provided in the cited references.25–31 The results of characterization analyses are presented in section S3 in the SI. All samples were tested in a lab-scale tubular packed-bed reactor under the same controlled reaction atmosphere. We obtained the turnover frequency (TOF) of each sample as a normalized reactivity where the methyl acetate (MA) generation rate was divided by the density of 8-MR BASs extracted from NH3-TPD and pyridine-IR measurements. The long-term reactivity data for the whole reaction time on stream (40 h) are presented in section S4 in the SI (Fig. S21–S26). Then we systematically correlated them with some descriptors of density functional theory (DFT), e.g., the CO addition activation energy and reaction enthalpy, side reactions with H2O and CO (only addressed in section S6 of the SI), and geometric configuration of a transient compound [Z–CH3–CO]‡, all of which were adopted in many previous studies for connecting to reactivity.14–16,32,33 However, it was confirmed that these descriptors ultimately fail to explain the DME carbonylation reactivity trends of extended 8-MR-possessing zeolites. Thus, we further considered the 3-dimensional distribution of BASs, proposing an “isovolume-based descriptor, (DHe, DCO, and DF)” highly correlated with the TOF evidenced from actual DME carbonylation of extended 8-MR zeolites, which can provide insightful information of the rational design of structurally diverse 8-MR zeolites.
2 Results and discussion
2.1 DME carbonylation reactivity over 8-MR zeolites
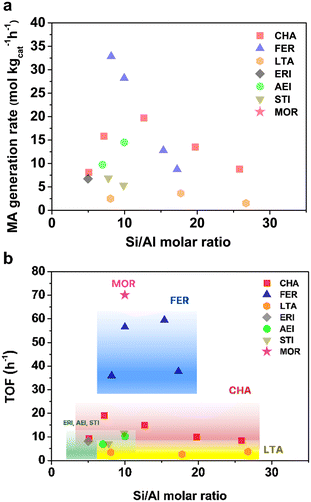
The results of the DME carbonylation reactivity test performed on the seven 8-MR zeolites selected in this study are summarized in Fig. 3. To investigate the correlation between zeolite topology and DME carbonylation reactivity, we selected Si/Al ratios ranging from 5 to 30 for each topology within the synthesis limitations available in the literature.31,34–40 All data points represent average reactivity evaluated over a 5-hour period (30–34 h), after an initial stabilization period, which was defined as a steady state, kinetic regime with negligible deactivation. Under mild reaction conditions, which are a strict kinetic regime (T = 165 °C, DME/CO/N2 = 4.5/90/5.5 mol%, P = 2.0 MPa), the product selectivity of each zeolite was limited to MA and unconverted DME. Consequently, DME conversions served as the primary measure and were correlated directly with zeolite topology.The BASs of the selected zeolites were evaluated using NH3-TPD and pyridine IR analyses. Pyridine IR was particularly effective for FER and MOR zeolites, where the focus was placed on 8-MR acid sites while minimizing the contributions from channels wider than 10-MR. Since a pyridine molecule (∼5.7 Å, in kinetic diameter) is larger than the pore size of 8-MR pore channels (∼3.5 × 4.9 Å2), pyridine selectively adsorbs on BASs within wider channels (see Fig. S1), thereby allowing to identify the influence from non-8-MR BASs at a wavenumber of 1540 cm−1 (see Table S2 and Fig. S18). Conclusively, the turnover frequency (TOF) for each zeolite is presented in Fig. 3b, calculated as: TOF (h−1) = MA generation rate (mmol gcat−1 h−1)/8-MR BAS density (mmol gcat−1). Here, the 8-MR BAS density denotes an estimated concentration of strong BASs located in 8-MR pockets, obtained from the high-temperature NH3-TPD component, and for MOR and FER, the values after subtracting the pyridine-accessible Brønsted acid sites in 10-MR or wider channels were determined by Py-IR at 165 °C. This procedure provides an approximate but internally consistent measure of carbonylation-relevant 8-MR BASs across all frameworks. The possibility of falsified kinetics by intracrystalline diffusion was ruled out by verifying that two CHA samples, with similar Si/Al ratios but significantly different crystal sizes, exhibited comparable apparent TOF values (see Fig. S2), despite CHA having the narrowest pores [3.6 × 3.6 Å] and thus being the most diffusion-sensitive framework among the zeolites studied.
Most zeolites had maximum MA generation rate values in the range of Si/Al elemental ratios within 7–10, consistent with prior investigations on CHA16 and FER26 regarding DME carbonylation. This is because this range strikes a good balance between keeping enough Brønsted acidity and not overcrowding the micropores too much.41–43 The apparent 8-MR BAS density (in particular for CHA) shows a “volcano-type” dependence on Si/Al, because at very low Si/Al, Al-site pairing and increased “NH3-philicity” reduce the fraction of sites expressed as strong, well-isolated 8-MR Brønsted acids in the high-temperature NH3-TPD component, whereas at very high Si/Al the total number of framework Al (and hence BAS) decreases, so the population of strong 8-MR BASs seems to be maximized at intermediate Si/Al. In addition, 7–10 is effectively the only Si/Al window in which phase-pure CHA, FER, AEI, STI, LTA, and MOR could be reproducibly synthesized with their respective OSDAs in this work, so the cross-topology TOF comparison is restricted to catalysts within this range. ERI could be obtained reproducibly only at lower Si/Al (∼5) under our synthesis conditions and is therefore represented by a single composition in Fig. 3. ERI (with an Si/Al ratio of ca. 5) exhibited a TOF of 8.1 h−1, whereas AEI (7.0) and STI (7.8) showed TOFs of 6.9 h−1 and 7.1 h−1, respectively. This shows that, despite having only one data point due to the synthetic constraint, ERI will follow a TOF trend similar to AEI and STI. According to Fig. 3b, even though all the zeolites share the same 8-MR structure, their TOFs vary depending on the topology. Within the Si/Al ratio range of 5 to 10, the reactivity order was found to be MOR > FER > CHA > ERI ≈ AEI ≈ STI > LTA.
By considering the 8-MR BAS density, the acid sites responsible for DME carbonylation were fully accounted for. However, since the catalysts still show different TOFs, it is clear that not only the acid site properties but also the intrinsic framework structures play a crucial role in determining the DME carbonylation reactivity. To gain in-depth insights into the relationship between structure and reactivity, as mentioned in the final part of section 1, this study applied two main approaches—1. DFT calculations and 2. geometric structures of a transient compound [Z–CH3–CO]‡—used in previous research to explain different reactivity orders. Sections 2.2 to 2.3 contain critical results showing that these conventional methods were inadequate to explain the reactivity order of investigated zeolite samples. Section 2.4 will finally present and explain the ‘key descriptor’ influencing the DME carbonylation reactivity of different zeolites possessing diverse 8-MRs, demonstrating that this descriptor best accounts for the reactivity observed in the zeolites investigated in this study.
2.2 DFT analysis of the rate-determining transition state
Numerous DFT calculations have been conducted to determine the acid sites with the highest reactivity by comparing the activation energy of the RDS.13–16,32,33 Although the details vary among these studies, they commonly evaluate both the activation energy and reaction enthalpy of the RDS at the TxOy sites of each zeolite. Overall, these findings conclude that the CO addition, which takes place primarily in and around the 8-MR pores, is the most crucial step influencing the DME carbonylation, albeit with varying levels of significance.Guided by these results, we focused on the specific TxOy sites located in or near 8-MR (referred to here as effective aluminum tetrahedral sites, or Al T sites) for all investigated topologies, as summarized in Table 2. Selected additional sites, essential for verification and extended discussion, are marked with a (see SI section S1). Table 2 also includes the results of the DFT calculation for both the methylation and CO addition steps across the examined TxOy sites, which are further supported by energy diagrams of each step (methylation and CO addition steps) in the SI section S5 (Fig. S27–S45). In particular, Fig. S41–S45 illustrate the whole sequential process from the initial BAS-mediated methylation step through CO addition, ultimately leading to the formation of the transient compound [Z–CH3–CO]‡. To help readers clearly understand which elementary steps contribute to the energies summarized in Table 2, we even address one of these diagrams (CHA T1O4 case) in Fig. 4 as a representative.
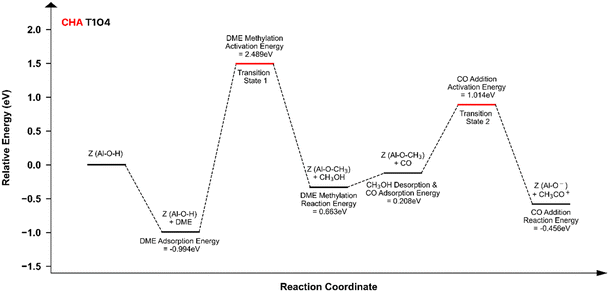 | ||
| Fig. 4 Full scheme of two key steps on the DME carbonylation (methylation and CO addition step) energy diagram for CHA T1O4. | ||
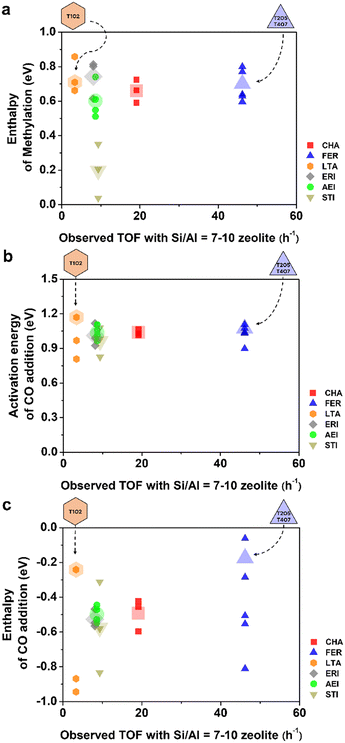
Fig. 5 depicts the relationship between DFT-calculated energy values and the average TOF for each topology. To facilitate a quantitative comparison of DME carbonylation reactivity, we correlated the average TOF values, which were acquired from catalysts with Si/Al ratios ranging from 7–10 (the appropriate Si/Al ratio range that allows comparison between different zeolite topologies), with (a) the methylation enthalpy, (b) the CO addition activation energy, and (c) the CO addition enthalpy of the analyzed TxOy sites. Furthermore, the average values for each topology are represented by larger and lighter markers to illustrate their representative values (see Fig. 5). For the FER and LTA, the data points were calculated by considering the T sites highlighted with a in Table 2, identified as the most likely TxOy sites for these frameworks (see Fig. 6 and SI section S1). In other topologies, the effective TxOy sites were evenly distributed throughout the 8-MR pores, without a preference for any specific region; these are represented as larger and lighter markers, calculated as the average values in Table 2.
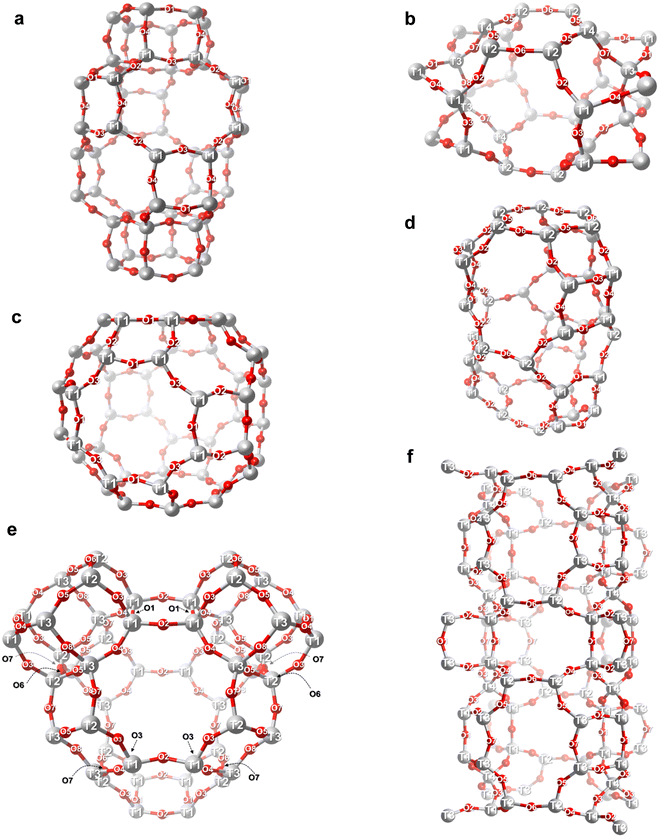 | ||
| Fig. 6 Detailed TxOy site locations of the zeolites examined in this study. The number of distinct tetrahedral sites (T-sites) and bridging oxygens (O-sites) for each zeolite was determined based on structural data from the IZA database.23 (a) CHA; (b) FER; (c) LTA; (d) ERI; (e) AEI; (f) STI. [Atom color] gray: tetrahedral site (T-site), red: bridging oxygen. | ||
As shown Fig. 5a, FER shows the highest TOF among the zeolites investigated. However, its methylation enthalpy does not appear to be more favorable than those of other topologies. Even the methylation energy at the most effective TxOy site within the FER framework (the T4O7 site, as identified by Feng et al.) is not more advantageous than other T sites within FER in this study. Fig. 5a shows this phenomenon more distinctively, corroborating previous observations.15
In conclusion, although lower methylation enthalpy facilitates the framework hydroxyl group in the subsequent CO addition step, it does not serve as a reliable metric for comparing different zeolite topologies. This result also contradicts the explanations given by Feng et al. for the performance differences between MOR and FER. Likewise, the DFT calculation of activation energy and enthalpy for the CO addition step fails to account for the significant TOF differences, as shown in Fig. 5b and c, where no discernible trend emerges. Therefore, while the DFT results for TxOy sites may be valid for evaluating the reactivity of different TxOy sites within a single topology, they appear insufficient when extended to comparisons across multiple zeolite topologies. Although these calculations consider each framework as a periodic model, the computational approach seems to overlook several critical factors.
2.3 Structural specificity of the TxOy site
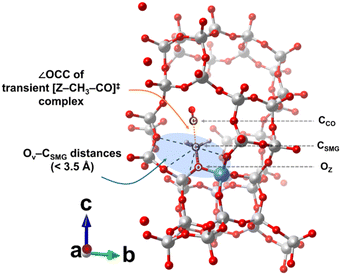
Boronat et al. identified the T3-O33 site (here referred to as T3O8) in the MOR framework as a key contributor to the high DME carbonylation reactivity, noting that a particular orientation of the SMG is necessary for efficient DME carbonylation within the channel structure.14 This specific orientation emerges because CO diffuses toward the surface methyl group (SMG), which is formed when a proton at a given TxOy site is replaced, and preferentially attaches orthogonally to the carbon atom of the SMG. The information of the zeolite T and O sites and the orientation of the SMG at each TxOy site is listed in Table S3 with the additional information in Fig. S46–S51 in section S5 of the SI for deeper understanding through graphical illustration. Geometrically, this can be related to the central atom angle ∠OCC of the transient [Z–CH3–CO]‡ complex during the CO addition step, where O refers to the oxygen of the TxOy site that anchors the SMG, the first C is the center carbon of the SMG, and the second C is the carbon of the reacting CO molecule (see Fig. 7). As previously explained with Fig. 2c, the reason MOR shows excellent reactivity is because the reactant gas CO can be easily added to the SMG in the orthogonal direction. Therefore, the closer the ∠OCC is to 180°, where ‘nucleophilic backside attack’ of CO on the center carbon of SMG readily occurs, the easier it is to form the transient [Z–CH3–CO]‡ complex, so the CO addition activation energy is relatively low, facilitating subsequent reaction steps.14–16,44,45 Although diffusion effects can influence DME carbonylation at elevated temperatures,46 the present study was conducted at 165 °C and 2.0 MPa CO, conditions chosen to suppress side reactions and ensure kinetic control by the CO addition step. The observed TOFs thus reflect intrinsic structural effects rather than diffusion or transport limitations.To determine the ∠OCC bond angle, the DFT calculations at the most stable transition state energy level can be utilized. However, exhaustively evaluating all TxOy sites across the zeolites featured in this study would be computationally expensive. Therefore, we calculated the ∠OCC bond angles, along with the OZ–CSMG and CSMG–CCO distances in the transition state (see Fig. 7), for only the effective TxOy sites in each topology along with the associated CO addition activation energies (see Table 3). Here, OZ represents the zeolite framework oxygen, CSMG refers to the carbon of the surface methoxy group, and CCO refers to the carbon in reacting CO molecule.
The ∠OCC angle of the transient compound [Z–CH3–CO]‡ shows a good correlation with catalytic performance. Several effective TxOy sites in MOR, FER, and CHA—zeolites known for relatively high reactivity—exhibit an ∠OCC angle close to 180°. A closer inspection reveals that the ∠OCC angle at the MOR T3O8 site (177.8°) is slightly farther from 180° than that at the FER T4O7 site (179.5°). Nevertheless, MOR is widely known to exhibit much higher reactivity than FER (although with rapid deactivation). This discrepancy implies that reactivity cannot be fully explained by the bond angle of the transient complex alone. The ∠OCC angle becomes even more pivotal when comparing zeolites with 3-dimensional pore networks. For instance, the notably higher reactivity of CHA compared to the other 3-dimensional pore-type zeolites (such as LTA, ERI, and AEI) can be partly attributed to the ∠OCC angle (177.8°) at the CHA T1O2 site. Zeolites with 8-MR TxOy sites with ∠OCC angles below 177° exhibit significantly lower TOFs than CHA, as seen in Fig. 3b.
Moreover, as the Brønsted acid site (BAS) occupancy at a TxOy site rises, where the ∠OCC angle is around 180°, the validity of the association between this angle descriptor and reactivity can be clarified. While the distribution of BASs is likely influenced by the synthesis procedure of each zeolite, the exact nature of this relationship remains unclear and even varies across zeolite types. This site preference is mainly discussed in SI section S1 about criteria for selecting effective Brønsted acid sites for DFT. A precise measurement of proton preference at each TxOy site would be ideal but remains challenging due to the myriad of environmental factors (e.g., reaction temperature, calcination, and pretreatment) affecting the sample. In zeolites with multiple crystallographically unique T sites, such as FER and STI, even state-of-the-art computational techniques demand substantial resources for accurate analysis. Consequently, prior studies have generally provided only approximate estimates of BAS preferences in diverse zeolite topologies.38,47–55 Accordingly, this study presents relative occupancy levels of effective TxOy sites instead of exact distribution, thereby underscoring the usefulness of the ∠OCC angle as a key descriptor for assessing reactivity.
In the case of CHA, at high Si/Al ratios, most protons occupy the T1O1 site. However, as Al substitution for tetrahedral Si increases, protons gradually populate the T1O2 site, which has an ∠OCC angle of 177.8°, favorable for CO addition. Likewise, the proton occupancy in FER is predominantly concentrated at the T4O7 site (∠OCC = 179.5°), facilitating the attack of CO on the methoxy group at this site. On the other hand, for LTA, most protons prefer the T1O2 site, which has an ∠OCC angle of 170.1°, the third lowest among the effective TxOy sites listed in Table 3. While LTA possesses the T1O3 site with a more favorable ∠OCC angle of 176.7° for CO addition, the lowest TOF of LTA among the zeolites studied can be adequately explained by the relative scarcity of proton occupancy at T1O3. In conclusion, if a substantial proportion of actual BASs occupy the TxOy site with an ∠OCC angle favorable for CO addition, high catalytic performance can be linked more directly to these geometric factors.
The case of ERI is also well explained by this correlation. Previous studies38,55 indicate that low-silica ERI prefers Al substitution at T2 sites (T2O2 and T2O6), which together contribute 75% of the 8-MR TxOy sites. With ∠OCC angles of 175.6° and 176.3°, these T2 sites favor CO addition and suggest that ERI could be relatively reactive among other small-pore zeolites. However, experimentally verifying this has been challenging due to difficulties in synthesizing high-silica ERI with Si/Al > 10. Hence, limited reaction data exist for ERI to confirm this conclusion. Nonetheless, as can be seen from Fig. 3b, with a Si/Al ratio of 5, ERI exhibits slightly higher reactivity than AEI, STI, and LTA with Si/Al ratios of 7–8. This trend suggests that ERI likely surpasses other small-pore zeolites in reactivity, supporting the notion that ERI has promising characteristics for DME carbonylation.
For AEI, prior research does not specify a TxOy site preference for Brønsted acid sites; however, each 8-MR TxOy site in AEI exhibits an ∠OCC angle of 175° or lower, which is somewhat suboptimal for CO addition. This observation explains the comparatively lower TOF of AEI than CHA, even though the two zeolites differ only in their stacking order of double-6-ring (d6r) units. By contrast, this geometric metric does not fully account for the notably low TOF of STI. The ∠OCC angles for T2O5 and T3O7 sites within the 8-MR of STI are 175.1° and 176.5°, respectively. Although these angles are not ideal (180°) for CO addition via ‘nucleophilic backside attack’ on the SMG, these angles should not severely hinder CO addition. However, Fig. 3b indicates that the TOF of STI is nearly the same as that of AEI, suggesting similarly low reactivity. Despite prior expectations, which are based on the relatively low methylation energy and CO addition activation energy of STI, that STI would outperform other reference zeolites, experimental findings show otherwise. This discrepancy indicates that factors beyond the ∠OCC angle or DFT-calculated values for specific TxOy sites must be considered to fully account for the reactivity differences.
Additionally, examining the OZ–CSMG and CSMG–CCO distances for each TxOy site reveals that they are mostly around 2 Å, showing no significant variation. Because the CO addition step occurs in the narrowly confined zeolite pore spaces, evaluating which TxOy site better stabilizes the transient compound [Z–CH3–CO]‡ cannot rely solely on these distance metrics. Rather, effective stabilization of the [Z–CH3–CO]‡ charge density is crucial for facilitating CO addition. Oxygen atoms within ca. 3.5 Å of the central carbon of SMGs have the greatest impact on its transition-state stabilization, as they readily donate electrons through delocalization. Consequently, the proximity of the central carbon of SMGs to nearby framework oxygens has a more pronounced influence on reactivity than simple interatomic distances within the transient compounds. To quantify these effects, we introduced the descriptor NO,CC, defined as the number of vicinal framework oxygens (OV) within 3.5 Å of CSMG, along with the average OV–CSMG distance. These values for each TxOy site in each topology are listed in Table 3.
Fig. 8 shows that the average OV–CSMG distance (<3.5 Å) correlates reasonably with the CO addition activation energy identified in section 2.2. Although some differences exist in the details, the overall similarity suggests that the stabilization of CO addition activation energy is largely influenced by close-contact oxygen atoms. These minor differences may stem from using an averaged OV–CSMG distance, which does not fully distinguish between oxygens directly adjacent to the Al site and those slightly farther away.16 Nevertheless, it is clear that the DFT calculation results presented in section 2.2 are not fully aligned, or proportional with the TOF trends shown in Fig. 3b. Overall, the observed discrepancy between the CO addition activation energies and average OV–CSMG distances among different TxOy sites reflects the limitations in applying simple geometrical descriptors in predicting catalytic performance.
 | ||
| Fig. 8 Average OV–CSMG distance (<3.5 Å) and CO addition activation energy at effective TxOy sites of selected zeolites. | ||
Those outcomes in section 2.2–2.3 suggested that conventional approaches, which are relying on examining only one effective TxOy site in isolation and investigating a single active TxOy site as the sole determinant of reactivity, are insufficient to explain the reactivity variation among zeolites. This underscores the necessity in building better descriptors that capture the multi-faceted, complex nature in zeolite reactivity, as addressed in the following sections.
2.4 Channel dimensionality of zeolite topology
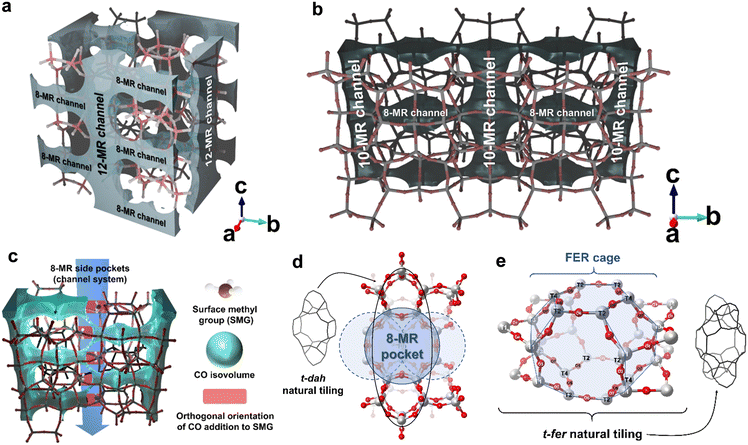
As detailed in section 1 on the DME carbonylation mechanism (eqn (1)–(4)), the surface acetyl group (SAG) created during the CO addition step ultimately forms the final product, MA, through DME adsorption. During this esterification process, the adsorbed MA leaves the BAS, which is then re-substituted by a SMG. It is worth noting that DME not only creates the active methyl group, which is known as the most abundant reaction intermediate (MARI), but also participates in the esterification that follows CO addition. Even if the partial pressure of DME is relatively low, it still sufficiently saturates the BAS of the zeolite to maintain a continuous supply of SMGs, thus sustaining the reaction cycle.17,33,56,57 Meanwhile, the high CO partial pressure (ca. 1.8 MPa) further increases the rate of reaction. This mechanism implies that, once formed, the SMG will not necessarily remain on a single acid site; instead, it may migrate to other sites during the esterification, driven by CO addition, a phenomenon sometimes called as “remethylation”. Thus, the average distance between BASs (for example, estimated by the ratio of accessible volume to the number of BASs) likely contributes significantly to overall reactivity of DME carbonylation.Based on this insight, we propose the descriptor, “molecule adsorption isovolume.”. In Fig. 9a, the gray and cyan regions depict the volumetric zones where critical portions of the reaction, especially interactions at BASs, predominantly occur within each zeolite, determined using helium (He) and CO as internal probe molecules, respectively. The isovolume region extends beyond the sum of the kinetic diameter of each probe molecule and the characteristic van der Waals radius (1.35 Å) of the framework atoms (oxygens), thereby defining where these probe molecules can diffuse and where the reaction takes place. This descriptor serves as a precise method for analyzing how active sites are distributed.
The isovolume is calculated exclusively on the basis of van der Waals radii, eliminating the effect of external energy that can promote molecular diffusion via vibration and rotation (i.e., hard-sphere model). To address this limitation under a controllable criterion, He, which is one of the smallest probe molecules, with a kinetic diameter of 2.60 Å, was employed to calculate the “He isovolume” (light gray). The cyan zone represents the CO-accessible volume, highlighting regions where DME carbonylation can occur via CO addition. Table 4 summarizes the numerical values of the corresponding isovolumes (VHe and VCO) shown in Fig. 9a.
Channel dimensionality can be assessed directly by examining isovolume data graphically. Although all zeolites in this study share an 8-MR pore system, they differ fundamentally in properties such as unit cell shape and channel dimensionality. Among these, CHA, LTA, ERI, and AEI are classified as 3-dimensional frameworks; each contains a repeating cage structure that connected with six neighboring cages through 8-MR pores, forming an interconnected 3-dimensional network. The isovolume values for these 3-dimensional type zeolites are calculated based on their cage structures.
In contrast, MOR and FER stand out for their distinct 2-dimensional frameworks, which many previous studies have linked to their superior performance. This enhanced reactivity stems from this cross-connected channel structure, where two intersecting channels of different diameters operate synergistically.12–15,18–21,32,33,58,59 The effectiveness of these channel systems lies in their “role division structure”: t-dah (natural tiling of MOR) or t-fer (natural tiling of FER) as the primary active site, while the larger channel promotes smooth product diffusion.
Further evidence for this phenomenon comes from Monte Carlo and dynamic diffusion simulations, which measure CO aggregation and MA transport, enabling systematic comparisons across various zeolite types.59,60 Nevertheless, these computational studies often exhibit certain limitations, such as overlooking the actual distribution of BASs (particularly those that dominate in real zeolites) and not explicitly applying the DME carbonylation mechanism at those sites. Relying predominantly on computational predictions over empirical reaction data may thus result in incomplete conclusions.
The isovolume graphical data also reveal notable structural distinctions between FER and MOR shown in Fig. 9b. Using CO as a probe molecule, a small cage (highlighted in cyan) appears within the FER cage (part of t-fer) and t-dah (MOR) natural tilings (green-colored box) whereas using helium as a probe uncovers staggered interconnections in MOR, depicted in gray fin-like shape. This distinctive 8-MR side-pocket structure helps clarify why the CO addition at the MOR T3O8 site is particularly favorable (see also Fig. 2c).
This side-pocket runs parallel to the c-axis (see Fig. 9b MOR, light blue-colored cylinder). Although its diameter (∼5.5 Å) is somewhat narrow for MA, it allows the CH3+ (methyl cation) released from DME during esterification to traverse the pocket. Thus, the T3O8-site-rich side pocket offers a short diffusion path for methyl groups to be rapidly substituted. Moreover, the favorable OCC angle (177.8°) supports surface acetyl group (SAG) formation through CO addition at the acid site, contributing to the higher reactivity of MOR compared to FER.
MA, the final product of DME carbonylation, can then diffuse through the main 12-MR channel, creating a well-defined role division structure that promotes overall reactivity. However, this structure also has a drawback: when acid sites are distributed in large channels like the 12-MR, dehydrogenation can occur, producing hydrocarbons that hinder MA diffusion, accelerate coking, and degrade catalyst performance.
STI, although structurally classified as a 2-dimensional zeolite, is widely recognized as functionally 1-dimensional in terms of molecular diffusion behavior. This transport confinement holds for most molecules involved in chemical reactions, including CO, which serves as the probe molecule in the cyan isovolume shown in Fig. 9. In STI, the primary 10-MR channels are linked through 8-MR pores, yet it lacks the role division structure (such as t-fer of FER or t-dah of MOR) found in the 2-dimensional zeolites MOR and FER. Because STI lacks the repeating cage structure found in cage-type zeolites (e.g., CHA, AEI, ERI, etc.), the unit cell volume was used to calculate its repeating structural unit; this shape is also depicted in Fig. 9a.
Although the specific approach to isovolume-based dimensional classification may differ, we ultimately assume a minimum repeating unit of a 3-dimensional structure for each topology in this study. Given the focus on the DME carbonylation mechanism, particular attention is paid to the effective TxOy sites contributing to the reaction. Accordingly, the isovolume concept aims to show how narrower internal volume (where reactions occur) and a higher density of BASs within that volume can result in proportionally higher TOF. This correlation provides a quantitative measure of how readily BASs can migrate across the other sites, reflecting the frequency of the DME carbonylation cycle.
Table 4 lists the values needed to calculate these frequencies, including He and CO isovolumes (VHe and VCO), the number of isovolumes per unit cell (NI) for each topology, and the count of effective oxygen sites (NEO) critical to the reaction. Oxygen-site numbering follows the IZA database23 and the criteria for selecting effective oxygen sites across tested zeolites are given in SI section S1. We define three descriptors (DHe, DCO, and DF) as follows:
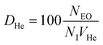 | (5) |
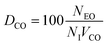 | (6) |
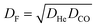 | (7) |
Fig. 10 shows the values of DHe, DCO, and DF for each topology, corresponding to the observed TOFs for samples with Si/Al ratios of 7–10 (the appropriate Si/Al ratio range that allows comparison between different zeolite topologies in this study). The graphs include trend lines for data both excluding MOR (solid line) and including MOR (dashed line), along with associated R2 values or slope standard error (SSE) values.
Fig. 10a and b show a linear correlation for the 8-MR zeolites (excluding MOR). However, when MOR data are included, the R2 value for DHe decreases from 0.96 to 0.91, and the SSE for DCO increases from 0.10 to 0.12. These changes suggest that DHe and DCO alone cannot fully capture the behavior of MOR, with its 8-MR side pocket and unique internal channel structure.
In contrast, Fig. 10c shows that using DF as a descriptor reveals a more robust linear correlation with TOF, even when MOR is included (R2 increases 0.96 to 0.98, SSE decreases from 0.07 to 0.04). These findings underscore the importance of considering VHe and VCO isovolumes when analyzing reactivity. They significantly support the notion that, in addition to CO addition, the “remethylation” step during subsequent esterification critically influences catalytic performance.
In conclusion, previously proposed descriptors for local BASs have shown limitation in explaining the reactivity differences across various zeolites. However, once the essential elementary steps in the reaction mechanism are clearly identified, it becomes possible to derive a 3-dimensional structural descriptor that strongly correlates with catalytic performance, based solely on the intrinsic framework geometry without the need for extensive computational analysis.
3 Conclusions
It is widely accepted that investigating the influence of 8-MR zeolites on DME carbonylation requires both thorough experimental and theoretical approaches. In this study, we systematically investigated multiple 8-MR zeolites and proposed a universal descriptor that captures the correlation between zeolite structure and DME carbonylation reactivity. It is emphasized that the findings of this work clearly indicate that conventional parameters, such as DFT-calculated energies of RDSs (CO addition and methylation steps), and local geometric descriptors of the transition state (e.g., ∠OCC angle or average distance of OV–CSMG < 3.5 Å), fully capture the noticeable variations in TOF resulting from different topologies.It should be noted that these conventional approaches were confined to evaluating the contribution of individual TxOy sites, rather than considering the broader 3-dimensional configuration of the zeolites. In order to fill this gap, we introduced two structural descriptors, DHe and DCO, which quantify how many effective oxygen sites exist within pre-defined isovolumes (VHe and VCO) linked to key steps in DME carbonylation. By using He as a probe for methyl cation pathways and CO as a probe for the CO addition pathway, each descriptor captures unique yet complementary information on the catalysts' internal environment. We highlight that each descriptor (DHe and DCO) shows strong correlation with TOF across multiple 8-MR zeolites, and taking their geometric mean (DF) gives an even better correlation, especially when MOR is included.
We underscore that this observation demonstrates the prominence of CO addition and remethylation steps in controlling DME carbonylation reactivity, given that the proposed descriptor DF explicitly includes both VHe and VCO, representing the two key steps, respectively. These results highlight two primary insights. First, zeolite structure and reactivity cannot be captured by focusing solely on local descriptors within the framework. Nonetheless, detailed investigation of DFT-calculated energies, interatomic distances, and conformation angles of the transition state remain critical, as they help define the three-dimensional isovolume more systematically for explaining reactions based on the zeolite framework and pore structures.
It should also be noted that DF was constructed based on the mechanistic features of DME carbonylation. Although the concept may be extendable, its applicability to other 8-MR reactions has not been validated and should be examined in future studies. Finally, the considerations of this work may advance the foundation for zeolite catalyst design by relating reactivity to 3-dimensional structural descriptors, providing both theoretical insights and practical guidance for optimizing performance in commercial applications.
4 Experimental
4.1 Synthesis
Complete details of the zeolite synthesis methods are detailed in section S2 of the SI.4.2 Characterization
Powder X-ray diffraction (PXRD) patterns were examined for each zeolite sample on a SMARTLAB X-ray diffractometer using Cu Kα radiation in the range of 2θ from 5° to 40° to confirm the crystallinity of the zeolites.NH3-temperature-programmed desorption (NH3-TPD) was conducted with a BELCAT-M instrument to examine the total Brønsted acid site (BAS) concentration. Prior to the main TPD analysis, the sample (∼50 mg) was pretreated under a flow of He (30 ml min−1) at 500 °C for 1 h. Then, the sample was cooled to 100 °C, after which NH3 was adsorbed for 1 h. Subsequently, the sample was heated from 100 to 800 °C at a ramping rate of 10 °C min−1, and the TCD signals were simultaneously acquired. The resulting profiles were deconvoluted into three Gaussian peaks, which can be typically interpreted respectively as weak, moderate, and strong acid sites. The higher the temperature of ammonia desorption, the stronger the acid sites. Generally, the weak and moderate acid sites are considered as Lewis acid sites or defected acid sites from extra-framework species in the zeolite. On the other hand, the strong acid sites are normally assigned to BASs.
Fourier-transform infrared spectroscopy (FT-IR) analysis was conducted on a PerkinElmer Frontier with a liquid N2-cooled mercury–cadmium-telluride (MCT) detector to measure Brønsted and Lewis acid sites distributed on a larger than 10-membered ring (10-MR) framework of the MOR and FER through an in situ adsorption of pyridine probe molecules. In more detail, the sample was pretreated at 500 °C for 1 h under a N2 flow and cooled down to 165 °C by measuring the respective background spectra. After the adsorption of probe pyridine for 1 h, the physisorbed surface pyridine was removed under vacuum conditions for 30 minutes, and the Py-IR spectra were obtained at the desorption temperature of 165 °C.
The results of characterization, calculated energy diagrams, framework geometries of the surface methyl group (SMG) for the TxOy sites of the investigated zeolites are listed in SI section S3.
4.3 Reaction and activity measurement
The gas-phase DME carbonylation reaction was evaluated by using a fixed-bed tubular reactor with an inner diameter of 7 mm. Before the carbonylation test, 0.3 g of each zeolite sample was pretreated under an N2 flow to remove any contaminants at 1 bar and 500 °C at a ramping rate of 10 °C min−1 for 1 h. Following pretreatment, the main reaction test was conducted at 165 °C and 2.0 MPa, with a space velocity (SV) of 2000 L kgcat−1 h−1 and a feed consisting of DME/CO/N2 = 4.5/90/5.5 vol%. The effluent stream was analyzed online by using a gas chromatograph (Younglin GC, YL6100) with a flame ionization detector (FID) and an HP-PLOT-U capillary column to identify and quantify DME, methanol (CH3OH), methyl acetate (MA), and other light hydrocarbons.Comprehensive numerical values for reactivity, 8-MR BAS density, and the essential rings and channel dimensions23 relevant to each topology are listed in Table 1.
| Catalystsa | 8-MR Brønsted acid site densityb (mmol g−1) | Essential ringsc | Channel dimensionality | MA generation rate, SSe (mol kgcat−1 h−1) | TOF, SSe (h−1) |
|---|---|---|---|---|---|
| a The number in parentheses indicates the Si/Al ratio. All tested catalysts fall within the range of Si/Al ratios that are feasible for synthesis.b The 8-MR Brønsted acid site (BAS) density was calculated by using an NH3-TPD curve, which was deconvoluted by Gaussian distribution using a 3-distribution model (GN(DLS) method) with damping set at 0.7. Among the 3 deconvoluted peaks, the peak in the highest temperature range distribution was chosen to represent the 8-MR BAS.25–31 Pyridine-IR was also used to estimate the Brønsted acid sites that can be adsorbed in the channel larger than 8-MR, consisting of FER and MOR, especially in this study. In these cases, the 8-MR BAS density was calculated by subtracting the BAS density derived by pyridine-IR from the strong acid site density estimated by NH3-TPD.c Essential rings, derived from face symbols of natural tiles, are ‘strong’ rings that cannot be reduced to sums of smaller rings, distinguishing them as key structural elements in zeolite channels.23d In STI, 8-MR pores connect parallel 10-MR channels, which are oriented in the [100] direction, but when CO is used as a probe molecule, van der Waals forces can cause the 8-MR pores to pucker, blocking the connection between the 10-MR channel (see SI section S1-STI).e Approximation to steady state (SS), which is the average value in the range of 30–34 h. However, CHA (79.5) is an exception, showing low reactivity, and is represented as an average value corresponding to the range of 24–28 h. | |||||
| CHA (5.1) | 0.864 | [81·61·42] | 3-Dimensional | 8.0 | 9.2 |
| CHA (7.2) | 0.822 | 15.7 | 19.1 | ||
| CHA (12.7) | 1.314 | 19.6 | 14.9 | ||
| CHA (19.8) | 1.346 | 13.4 | 10.0 | ||
| CHA (25.9) | 1.030 | 8.7 | 8.4 | ||
| CHA (50.8) | 0.416 | 2.1 | 5.2 | ||
| CHA (67.5) | 0.333 | 1.3 | 4.1 | ||
| CHA (79.5) | 0.231 | 0.7 | 3.1 | ||
| FER (8.2) | 0.914 | [101·81·61·54] | 2-Dimensional | 32.8 | 35.9 |
| FER (10.0) | 0.498 | 28.1 | 56.5 | ||
| FER (15.4) | 0.213 | 12.7 | 59.5 | ||
| FER (17.3) | 0.229 | 8.6 | 37.7 | ||
| LTA (8.1) | 0.694 | [81·61·42] | 3-Dimensional | 2.3 | 3.4 |
| LTA (17.8) | 0.544 | 3.5 | 2.6 | ||
| LTA (26.8) | 0.368 | 1.4 | 3.7 | ||
| LTA (44.0) | 0.189 | 0.5 | 2.4 | ||
| ERI (∼5) | 0.819 | [81·63·42] | 3-Dimensional | 6.6 | 8.0 |
| AEI (7.0) | 1.389 | [82·61·44] | 3-Dimensional | 9.6 | 6.9 |
| AEI (10.0) | 1.394 | 14.4 | 10.3 | ||
| STI (7.8) | 0.956 | [101·82·61·51·42]d | Functionallyd | 6.8 | 7.1 |
| STI (9.9) | 0.448 | 1-Dimensional | 5.2 | 11.6 | |
| CMOR (10.0) | 1.369 | [121·83·55·41] | 2-Dimensional | 96.0 | 70.1 |
| Zeolite | Site | Methylation | CO addition | ||
|---|---|---|---|---|---|
| ΔE (eV) | Average ΔE (eV) | Ea (eV) | ΔE (eV) | ||
| a Some TxOy sites that are not present in 8-MR but required for discussion. See SI section S1.b MOR T3O8 site is introduced in this paper due to its unique site position in mordenite for the selective DME carbonylation process. | |||||
| CHA | T1O2 | 0.590 | 0.659 | 1.028 | −0.422 |
| T1O3 | 0.725 | 1.065 | −0.597 | ||
| T1O4 | 0.663 | 1.014 | −0.456 | ||
| FER | T1O3 | 0.638 | 0.686 | 1.031 | −0.507 |
| T2O2 | 0.596 | 1.072 | −0.554 | ||
| T2O5a | 0.629 | 1.106 | −0.063 | ||
| T2O6 | 0.799 | 0.897 | −0.811 | ||
| T4O7a | 0.770 | 1.037 | −0.286 | ||
| LTA | T1O1 | 0.661 | 0.742 | 0.968 | −0.868 |
| T1O2a | 0.709 | 1.169 | −0.242 | ||
| T1O3 | 0.857 | 0.807 | −0.944 | ||
| ERI | T1O4 | 0.613 | 0.742 | 1.116 | −0.468 |
| T2O2 | 0.812 | 0.989 | −0.547 | ||
| T2O6 | 0.801 | 0.924 | −0.565 | ||
| AEI | T1O2 | 0.610 | 0.602 | 1.105 | −0.445 |
| T1O4 | 0.740 | 0.965 | −0.527 | ||
| T2O3 | 0.548 | 1.057 | −0.547 | ||
| T3O7 | 0.511 | 1.015 | −0.471 | ||
| STI | T2O5 | 0.350 | 0.198 | 0.982 | −0.580 |
| T2O6 | 0.206 | 0.825 | −0.834 | ||
| T3O7 | 0.038 | 1.080 | −0.312 | ||
| MOR | T3O8b | −0.247 | — | 0.890 | −0.442 |
| Zeolite | Site | [Z–CH3–CO]‡ | NO,CC | Average distance of OV–CSMG (<3.5 Å) | ||
|---|---|---|---|---|---|---|
| ∠OCC (°) | OZ–CSMG (Å) | CSMG–CCO (Å) | ||||
| a Some TxOy sites that are not present in 8-MR but required for discussion. See SI section S1.b MOR T3O8 site is introduced in this paper due to its unique site position in mordenite for selective DME carbonylation process. | ||||||
| CHA | T1O2 | 177.8 | 2.024 | 1.989 | 3 | 3.049 |
| T1O3 | 172.3 | 2.028 | 2.018 | 4 | 3.151 | |
| T1O4 | 175.5 | 2.012 | 2.007 | 4 | 3.163 | |
| FER | T1O3 | 171.3 | 2.034 | 1.975 | 3 | 3.062 |
| T2O2 | 134.8 | 2.035 | 1.995 | 3 | 3.076 | |
| T2O5a | 174.7 | 2.034 | 1.968 | 4 | 3.125 | |
| T2O6 | 171.5 | 2.011 | 1.999 | 4 | 3.073 | |
| T4O7a | 179.5 | 2.023 | 1.998 | 3 | 3.088 | |
| LTA | T1O1 | 172.9 | 2.015 | 2.013 | 3 | 2.986 |
| T1O2a | 170.1 | 2.004 | 1.966 | 6 | 3.258 | |
| T1O3 | 176.7 | 2.022 | 2.031 | 2 | 2.835 | |
| ERI | T1O4 | 172.1 | 2.004 | 1.990 | 4 | 3.220 |
| T2O2 | 175.6 | 2.022 | 2.010 | 3 | 3.017 | |
| T2O6 | 176.3 | 2.031 | 2.001 | 4 | 3.011 | |
| AEI | T1O2 | 172.5 | 2.026 | 1.965 | 4 | 3.150 |
| T1O4 | 174.6 | 2.030 | 1.980 | 4 | 3.263 | |
| T2O3 | 174.4 | 2.002 | 1.956 | 4 | 3.206 | |
| T3O7 | 172.1 | 1.989 | 1.952 | 4 | 3.177 | |
| STI | T2O5 | 175.1 | 2.016 | 1.953 | 5 | 3.204 |
| T2O6 | 169.5 | 2.031 | 2.043 | 4 | 2.998 | |
| T3O7 | 176.5 | 2.041 | 2.014 | 2 | 2.948 | |
| MOR | T3O8b | 177.8 | 2.009 | 2.003 | 4 | 3.162 |
| Zeolite type | Number of T site per unit cell | VHe (Å3) | VCO (Å3) | Effective oxygen site | NI | NEO | DHe (Å−3) | DCO (Å−3) | DF (Å−3) |
|---|---|---|---|---|---|---|---|---|---|
| a Similar to other zeolites, the effective oxygen sites of the LTA-type framework (those contributing to the reaction) are located on the 8-MR. However, during synthesis, these sites predominantly occupy the O2 site on the d6r unit. Therefore, values corresponding to actual cases are indicated in parentheses for clarity. See SI section S1. | |||||||||
| CHA | 36 | 210.7 | 102.2 | O2, O3, O4 | 3 | 54 | 8.54 | 17.6 | 12.3 |
| FER | 36 | 80.4 | 38.0 | O2, O3, O5, O6, O7 | 2 | 44 | 27.4 | 58.0 | 39.8 |
| LTA | 24 | 385.8 | 246.6 | O1, O3, (O2) | 1 | 36 | 9.33 (3.11)a | 14.6 (4.87)a | 3.89 |
| ERI | 36 | 250.5 | 124.0 | O2, O4, O6 | 2 | 42 | 8.38 | 16.9 | 11.9 |
| AEI | 48 | 179.3 | 101.0 | O2, O3, O4, O7 | 4 | 56 | 7.81 | 13.9 | 10.4 |
| STI | 72 | 246.5 | 133.6 | O5, O6, O7 | 4 | 32 | 3.25 | 5.99 | 4.41 |
| MOR | 48 | 111.0 | 22.7 | O2, O3, O5, O7, O8, O10 | 2 | 56 | 25.2 | 123 | 55.8 |
4.4 Computational methods for DFT calculations
Density functional theory (DFT) calculations were performed to validate the reaction enthalpies and activation energies for the key steps—methylation and CO addition—in DME carbonylation at various BASs within small-pore zeolite frameworks. The choice of BASs was informed by representative studies identifying locations where these sites are most densely populated.The VASP code, GGA-PBE XC-functionals, and zero damping D3-Grimme's correction for van der Waals dispersion are employed for the computation.61 A cutoff energy of 400 eV was used, and all structures are relaxed.62–64 The coordinates of reagents and products were optimized until the maximum force on each atom was below 0.05 eV Å−1,65,66 and the energy was converged to within a 1 × 10−6 eV threshold. Transition states were initially located via the NEB method, using a convergence criterion of 0.3 eV Å−1,67,68 and then refined with the dimer method, which reached a force convergence of 0.05 eV Å−1.69 During the dimer method, only the CH3CO+ moiety was relaxed.
All DFT calculations were carried out under a static framework approximation at 0 K, without including thermal vibrations or dynamic solvent effects. While our calculations neglect finite-temperature effects and vibrational dynamics, this static DFT approach is commonly used and sufficient for capturing qualitative trends in zeolite-catalyzed reactions.
Author contributions
D. J. Jeong: conceptualization, writing – original draft, review, editing and visualization, design experimental work, zeolite synthesis, data analysis, characterization, reaction and calculation of 3-dimension related data. X. Wang: data analysis, characterization, reaction. N. Park: DFT calculation. D. Lee: zeolite synthesis. Y. Lee: figuration of the DFT energy diagram in the SI. J. W. Bae: writing – review & editing, supervision. W. B. Lee: writing – review & editing, supervision. J. H. Kang: writing – review & editing, supervision, conceptualization. D. J. Jeong, X. W., and N. P. contributed equally to this work.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included in the main text and as part of the supporting information (SI). Any further details will be available upon request. Supplementary information is available. See DOI: https://doi.org/10.1039/d5im00276a.Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) funded by the Korea government (MSIT) (grant numbers RS-2024-00405818 and RS-2024-00343871). The authors thank the staff of the National Center for Inter-University Research Facilities (NCIRF).References
- P. Kalck, C. Le Berre and P. Serp, Recent advances in the methanol carbonylation reaction into acetic acid, Coord. Chem. Rev., 2020, 402, 213078 Search PubMed.
- P. Pal and J. Nayak, Acetic acid production and purification: Critical review towards process intensification, Sep. Purif. Rev., 2017, 46, 44–61 Search PubMed.
- S. K. Bhatia and Y. H. Yang, Microbial production of volatile fatty acids: Current status and future perspectives, Rev. Environ. Sci. Bio/Technol., 2017, 16, 327–345 CrossRef CAS.
- N. Yoneda, S. Kusano, M. Yasui, P. Pujado and S. Wilcher, Recent advances in processes and catalysts for the production of acetic acid, Appl. Catal., A, 2001, 221, 253–265 Search PubMed.
- P. M. Maitlis, A. Haynes, G. J. Sunley and M. J. Howard, Methanol carbonylation revisited: Thirty years on, J. Chem. Soc., Dalton Trans., 1996, 2187–2196 Search PubMed.
- A. Haynes, in Advances in Catalysis, ed. B. C. Gates and H. Knözinger, Elsevier, Amsterdam, 2010, ch. 1, vol. 53, pp. 1–45 Search PubMed.
- C. M. Thomas and G. Süss-Fink, Ligand effects in the rhodium-catalyzed carbonylation of methanol, Coord. Chem. Rev., 2003, 243, 125–142 Search PubMed.
- P. Cheung, A. Bhan, G. J. Sunley and E. Iglesia, Selective carbonylation of dimethyl ether to methyl acetate catalyzed by acidic zeolites, Angew. Chem., Int. Ed., 2006, 45, 1617–1620 Search PubMed.
- D. Mao, W. Yang, J. Xia, B. Zhang, Q. Song and Q. Chen, Highly effective hybrid catalyst for the direct synthesis of dimethyl ether from syngas with magnesium oxide-modified HZSM-5 as a dehydration component, J. Catal., 2005, 230, 140–149 Search PubMed.
- T. Pöpken, L. Götze and J. Gmehling, Reaction kinetics and chemical equilibrium of homogeneously and heterogeneously catalyzed acetic acid esterification with methanol and methyl acetate hydrolysis, Ind. Eng. Chem. Res., 2000, 39, 2601–2611 Search PubMed.
- H. Ham, H. S. Jung, H. S. Kim, J. Kim, S. J. Cho, W. B. Lee, M. J. Park and J. W. Bae, Gas-phase carbonylation of dimethyl ether on the stable seed-derived ferrierite, ACS Catal., 2020, 10, 5135–5146 Search PubMed.
- A. Bhan, A. D. Allian, G. J. Sunley, D. J. Law and E. Iglesia, Specificity of sites within eight-membered ring zeolite channels for carbonylation of methyls to acetyls, J. Am. Chem. Soc., 2007, 129, 4919–4924 CrossRef CAS PubMed.
- D. B. Rasmussen, J. M. Christensen, B. Temel, F. Studt, P. G. Moses, J. Rossmeisl, A. Riisager and A. D. Jensen, Reaction mechanism of dimethyl ether carbonylation to methyl acetate over mordenite – A combined DFT/experimental study, Catal. Sci. Technol., 2017, 7, 1141–1152 Search PubMed.
- M. Boronat, C. Martínez-Sánchez, D. Law and A. Corma, Enzyme-like specificity in zeolites: A unique site position in mordenite for selective carbonylation of methanol and dimethyl ether with CO, J. Am. Chem. Soc., 2008, 130, 16316–16323 Search PubMed.
- P. Feng, G. Zhang, X. Chen, K. Zang, X. Li and L. Xu, Specific zone within 8-membered ring channel as catalytic center for carbonylation of dimethyl ether and methanol over FER zeolite, Appl. Catal., A, 2018, 557, 119–124 Search PubMed.
- M. Lusardi, T. T. Chen, M. Kale, J. H. Kang, M. Neurock and M. E. Davis, Carbonylation of dimethyl ether to methyl acetate over SSZ-13, ACS Catal., 2019, 10, 842–851 Search PubMed.
- P. Cheung, A. Bhan, G. Sunley, D. Law and E. Iglesia, Site requirements and elementary steps in dimethyl ether carbonylation catalyzed by acidic zeolites, J. Catal., 2007, 245, 110–123 CrossRef CAS.
- Y. Chu, A. Y. Lo, C. Wang and F. Deng, Origin of high selectivity of dimethyl ether carbonylation in the 8-membered ring channel of mordenite zeolite, J. Phys. Chem. C, 2019, 123, 15503–15512 Search PubMed.
- X. B. Feng, F. Chen, Z. M. He, X. Y. Zhao and J. P. Cao, Comparison of dimethyl ether carbonylation performance over some zeolites containing 8-member ring pores, Energy Fuels, 2022, 36, 14341–14348 Search PubMed.
- S. Liu, X. Fang, Y. Liu, H. Liu, X. Ma, W. Zhu and Z. Liu, Dimethyl ether Carbonylation over Mordenite zeolite modified by Alkyimidazolium ions, Catal. Commun., 2020, 147, 106161 Search PubMed.
- A. Bhan and E. Iglesia, A link between reactivity and local structure in acid catalysis on zeolites, Acc. Chem. Res., 2008, 41, 559–567 Search PubMed.
- Y. Chu, B. Han, A. Zheng, X. Yi and F. Deng, Pore selectivity for olefin protonation reactions confined inside mordenite zeolite: A theoretical calculation study, J. Phys. Chem. C, 2013, 117, 2194–2202 Search PubMed.
- IZA Structure Commission, https://america.iza-structure.org/IZA-SC/ftc_table.php, (accessed December 28, 2025).
- S. Yuan, J. Wang, Y. Li and S. Peng, Siting of B, Al, Ga or Zn and bridging hydroxyl groups in mordenite: An ab initio study, J. Mol. Catal. A: Chem., 2001, 175, 131–138 Search PubMed.
- Y. Li, R. Liu, Q. Guo, H. Bian, A. Lan, X. Li, P. Han and T. Dou, Efficient synthesis of high silica SSZ-13 zeolite via a steam-assisted crystallization process, J. Porous Mater., 2019, 26, 1879–1888 CrossRef CAS.
- S. Y. Park, C. H. Shin and J. W. Bae, Selective carbonylation of dimethyl ether to methyl acetate on Ferrierite, Catal. Commun., 2016, 75, 28–31 CrossRef CAS.
- N. E. Gordina, T. N. Borisova, K. S. Kiyagina, I. A. Astrakhantseva, A. A. Ilyin and R. N. Rumyantsev, Investigation of NH3 desorption kinetics on the LTA and SOD zeolite membranes, Membranes, 2022, 12, 147 Search PubMed.
- P. Xiao, Y. Wang, L. Wang, H. Toyoda, k. Nakamura, S. Bekhti, Y. Lu, J. Huang, H. Gies and T. Yokoi, Understanding the effect of spatially separated Cu and acid sites in zeolite catalysts on oxidation of methane, Nat. Commun., 2024, 15, 2718 CrossRef CAS PubMed.
- J. Zhu, K. Muraoka, T. Ohnishi, Y. Yanaba, M. Ogura, A. Nakayama, T. Wakihara, Z. Liu and T. Okubo, Synthesis and structural analysis of high-silica ERI zeolite with spatially-biased Al distribution as a promising NH3-SCR catalyst, Adv. Sci., 2024, 11, 2307674 CrossRef CAS PubMed.
- K. Cai, S. Huang, Y. Li, Z. Cheng, J. Lv and X. Ma, Influence of acid strength on the reactivity of dimethyl ether carbonylation over H-MOR, ACS Sustainable Chem. Eng., 2018, 7, 2027–2034 CrossRef.
- S. B. Hong, E. G. Lear, P. A. Wright, W. Zhou, P. A. Cox, C. H. Shin, J. H. Park and I. S. Nam, Synthesis, structure solution, characterization, and catalytic properties of TNU-10: A high-silica zeolite with the STI topology, J. Am. Chem. Soc., 2004, 126, 5817–5826 CrossRef CAS PubMed.
- M. Boronat, C. Martinez and A. Corma, Mechanistic differences between methanol and dimethyl ether carbonylation in side pockets and large channels of mordenite, Phys. Chem. Chem. Phys., 2011, 13, 2603–2612 Search PubMed.
- D. B. Rasmussen, J. M. Christensen, B. Temel, F. Studt, P. G. Moses, J. Rossmeisl, A. Riisager and A. D. Jensen, Ketene as a reaction intermediate in the carbonylation of dimethyl ether to methyl acetate over mordenite, Angew. Chem., Int. Ed., 2015, 54, 7261–7264 Search PubMed.
- N. Tsunoji, D. Shimono, K. Tsuchiya, M. Sadakane and T. Sano, Formation pathway of AEI zeolites as a basis for a streamlined synthesis, Chem. Mater., 2019, 32, 60–74 CrossRef.
- B. W. Boal, J. E. Schmidt, M. A. Deimund, M. W. Deem, L. M. Henling, S. K. Brand, S. I. Zones and M. E. Davis, Facile synthesis and catalysis of pure-silica and heteroatom LTA, Chem. Mater., 2015, 27, 7774–7779 CrossRef CAS.
- D. Jo, T. Ryu, G. T. Park, P. S. Kim, C. H. Kim, I. S. Nam and S. B. Hong, Synthesis of high-silica LTA and UFI zeolites and NH3–SCR performance of their copper-exchanged form, ACS Catal., 2016, 6, 2443–2447 CrossRef CAS.
- R. Shannon, R. Staley, A. Vega, R. Fischer, W. Baur and A. Auroux, Nature of the framework hydroxyl groups in steamed H-RHO and H-erionite zeolites, J. Phys. Chem., 1989, 93, 2019–2027 CrossRef CAS.
- K. Lillerud, Aluminium distribution in the zeolites offretite and erionite, Zeolites, 1987, 7, 14–17 Search PubMed.
- F. Roessner, K. H. Steinberg, A. Rudolf and B. Staudte, Infrared spectroscopic study on the cation distribution in calcium-exchanged erionite, Zeolites, 1989, 9, 371–376 Search PubMed.
- J. Antúnez-García, D. H. Galván, V. Petranovskii, F. N. Murrieta-Rico, R. I. Yocupicio-Gaxiola and S. Fuentes-Moyado, Theoretical study of the effect of isomorphous substitution by Al3+ and/or Fe3+ cations to tetrahedral positions in the framework of a zeolite with erionite topology, J. Mater. Sci., 2019, 54, 13190–13199 CrossRef.
- J. Li, M. Gao, W. Yan and J. Yu, Regulation of the Si/Al ratios and Al distributions of zeolites and their impact on properties, Chem. Sci., 2023, 14, 1935–1959 Search PubMed.
- Z. Wang, Y. Jiang, O. Lafon, J. Trébosc, K. D. Kim, C. Stampfl, A. Baiker, J. P. Amoureux and J. Huang, Brønsted acid sites based on penta-coordinated aluminum species, Nat. Commun., 2016, 7, 13820 Search PubMed.
- P. Bräuer, P. L. Ng, O. Stiumorang, I. Hitchcock and C. D'Agostino, Effect of Al content on number and location of hydroxyl acid species in zeolites: A DRIFTS quantitative protocol without the need for molar extinction coefficients, RSC Adv., 2017, 7, 52604–52613 Search PubMed.
- M. Boronat and A. Corma, Factors controlling the acidity of zeolites, Catal. Lett., 2015, 145, 162–172 Search PubMed.
- S. Wang, S. Li, L. Zhang, Z. Qin, Y. Chen, M. Dong, J. Li, W. Fan and J. Wang, Mechanistic insights into the catalytic role of various acid sites on ZSM-5 zeolite in the carbonylation of methanol and dimethyl ether, Catal. Sci. Technol., 2018, 8, 3193–3204 Search PubMed.
- Y. Liu, N. Zhao, H. Xian, Q. Cheng, Y. Tan, N. Tsubaki and X. Li, Facilely synthesized H-mordenite nanosheet assembly for carbonylation of dimethyl ether, ACS Appl. Mater. Interfaces, 2015, 7, 8398–8403 Search PubMed.
- M. Bocus, R. Goeminne, A. Lamaire, M. Cools-Ceuppens, T. Verstraelen and V. Van Speybroeck, Nuclear quantum effects on zeolite proton hopping kinetics explored with machine learning potentials and path integral molecular dynamics, Nat. Commun., 2023, 14, 1008 CrossRef CAS PubMed.
- Z. Zhao, Y. Xing, S. Li, X. Meng, F. Xiao, R. McGuire, A. Parvulescu, U. Müller and W. Zhang, Mapping Al distributions in SSZ-13 zeolites from 23Na solid-state NMR spectroscopy and DFT calculations, J. Phys. Chem. C, 2018, 122, 9973–9979 Search PubMed.
- A. Martucci, A. Alberti, G. Cruciani, P. Radaelli, P. Ciambelli and M. Rapacciulo, Location of Brønsted sites in D-ferrierite by neutron powder diffraction, Microporous Mesoporous Mater., 1999, 30, 95–101 CrossRef CAS.
- T. Lemishko, S. Valencia, F. Rey, M. Jiménez-Ruiz and G. Sastre, Inelastic neutron scattering study on the location of Brønsted acid sites in high silica LTA zeolite, J. Phys. Chem. C, 2016, 120, 24904–24909 Search PubMed.
- C. Beauvais, X. Guerrault, F. Coudert, A. Boutin and A. Fuchs, Distribution of sodium cations in faujasite-type zeolite: Acanonical parallel tempering simulation study, J. Phys. Chem. B, 2004, 108, 399–404 Search PubMed.
- J. Sauer, Sodium doped sodium sodalite: Magnetic coupling between F centers and hyperfine interactions with framework atoms, Phys. Chem. Chem. Phys., 1999, 1, 4505–4514 RSC.
- H. Trill, H. Eckert and V. Srdanov, Topotactic transformations of sodalite cages: Synthesis and NMR study of mixed salt-free and salt-bearing sodalites, J. Am. Chem. Soc., 2002, 124, 8361–8370 Search PubMed.
- R. Fletcher, S. Ling and B. Slater, Violation of Löwenstein's rule in zeolites, Chem. Sci., 2017, 8, 7483–7491 RSC.
- G. Engelhardt and D. Michel, High-resolution solid-state NMR of silicates and zeolites, Wiley, New York, 1987 Search PubMed.
- W. Chen, K. A. Tarach, K. Góra-Marek and A. Zheng, Confinement driving mechanism of surface methoxy species formation in mordenite zeolite: An interplay of different molecular factors, Appl. Catal., B, 2024, 357, 124306 CrossRef CAS.
- Y. Jiang, M. Hunger and W. Wang, On the reactivity of surface methoxy species in acidic zeolites, J. Am. Chem. Soc., 2006, 128, 11679–11692 Search PubMed.
- W. Chen, Z. Liu, X. Yi and A. Zheng, Confinement-driven dimethyl ether carbonylation in mordenite zeolite as an ultramicroscopic reactor, Acc. Chem. Res., 2024, 57, 2804–2815 CrossRef CAS PubMed.
- Z. Liu, X. Yi, G. Wang, X. Tang, G. Li, L. Huang and A. Zheng, Roles of 8-ring and 12-ring channels in mordenite for carbonylation reaction: From the perspective of molecular adsorption and diffusion, J. Catal., 2019, 369, 335–344 Search PubMed.
- W. Wang, W. Qian, H. Ma, W. Ying and H. Zhang, A theoretical study on the feed ratio of dimethyl ether carbonylation on H-MOR zeolites, Mol. Phys., 2021, 119, e1896044 Search PubMed.
- A. M. Ukpong, First principles study of van der Waals heterobilayers, Comput. Condens. Matter, 2015, 2, 1–10 CrossRef.
- A. Kiejna, T. Pabisiak and S. Gao, The energetics and structure of rutile TiO2 (110), J. Phys. Condens. Matter, 2006, 18, 4207 Search PubMed.
- X. Ma, Y. Wu, Y. Lu and Y. Zhu, Correlation effects on lattice relaxation and electronic structure of ZnO within the GGA+ U formalism, J. Phys. Chem. C, 2013, 117, 26029–26039 Search PubMed.
- J. Sun, R. C. Remsing, Y. Zhang, Z. Sun, A. Ruzsinszky, H. Peng, Z. Yang, A. Paul, U. Waghmare and X. Wu, Accurate first-principles structures and energies of diversely bonded systems from an efficient density functional, Nat. Chem., 2016, 8, 831–836 Search PubMed.
- P. Maragakis, S. A. Andreev, Y. Brumer, D. R. Reichman and E. Kaxiras, Adaptive nudged elastic band approach for transition state calculation, J. Chem. Phys., 2002, 117, 4651–4658 Search PubMed.
- J. A. Garrido Torres, P. C. Jennings, M. H. Hansen, J. R. Boes and T. Bligaard, Low-scaling algorithm for nudged elastic band calculations using a surrogate machine learning model, Phys. Rev. Lett., 2019, 122, 156001 CrossRef CAS PubMed.
- H. Aljama and F. Abild-Pedersen, Accessing the C–C transition state energy on transition metals, Phys. Chem. Chem. Phys., 2019, 21, 25328–25333 Search PubMed.
- R. Kronberg, H. Lappalainen and K. Laasonen, Revisiting the Volmer–Heyrovský mechanism of hydrogen evolution on a nitrogen doped carbon nanotube: Constrained molecular dynamics versus the nudged elastic band method, Phys. Chem. Chem. Phys., 2020, 22, 10536–10549 RSC.
- K. Sun, Y. Zhao, H. Y. Su and W. X. Li, Force reversed method for locating transition states, Theor. Chem. Acc., 2012, 131, 1–10 Search PubMed.
Footnote |
| † Present address: Institute of Advanced Study, Chengdu University, Chengdu 610106, China. |
| This journal is © Institute of Process Engineering of CAS 2026 |