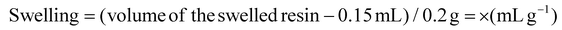Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Solvent recycling in solid-phase peptide synthesis (SPPS): combination of a swellable macroporous polystyrene (SMPS) resin and green binary solvents
Mani Kaushala,
Sikabwe Nokiab,
Yuxing Yangc,
Chaoyang Wangc,
Cheng Zhangc,
Alesandra Bassoc,
Simona Serbanc,
Dan Wuc,
Yanjun Lic,
Xiaokang Kouc,
Ashish Kumara,
Anamika Sharmaa,
Beatriz G. de la Torre *b and
Fernando Albericio
*b and
Fernando Albericio *ad
*ad
aPeptide Science Laboratory, School of Chemistry and Physics, University of KwaZulu-Natal, Durban 4001, South Africa. E-mail: albericio@ukzn.ac.za
bSchool of Laboratory Medicine and Medical Sciences, College of Health Sciences, University of KwaZulu-Natal, Durban 4041, South Africa. E-mail: Garciadelatorreb@ukzn.ac.za
cSunresin New Materials, Sunresin Park, No. 135 Jinye Rd, Xi'an Hi-tech Industrial Development Zone, Shaanxi, 710076, China
dDepartment of Inorganic and Organic Chemistry, University of Barcelona, Martí i Franqués 1-11, 08028-Barcelona, Spain
First published on 13th May 2026
Abstract
Solid-phase peptide synthesis (SPPS) relies heavily on hazardous solvents, such as N,N′-dimethylformamide (DMF), whose substitution by green solvents is proving more challenging than expected. Currently, binary mixtures of dimethyl sulfoxide (DMSO) or N-butylpyrrolidone (NBP) with other solvents are rendering the best results. These solvent mixtures are difficult to recycle due to the high boiling points of DMSO and NBP. Ideally, the solvent or solvent mixture used in SPPS should have a boiling point that allows its recycling without excessive energy use. Herein, we have developed a new swellable macroporous polystyrene (SMPS) resin that enables SPPS in greener, more recyclable solvent systems. The SMPS resin, characterized by a 6% divinylbenzene crosslinking degree and macroporous architecture, exhibited good swelling behavior across a broad range of single and binary solvents, including acetonitrile (MeCN), ethyl acetate (EtOAc), 2-methyltetrahydrofuran (2-MeTHF), and mixtures of EtOAc with MeCN, triethyl phosphate (TEP), and toluene (Tol). Its performance was assessed through the synthesis of three model peptides of increasing complexity: Leu-enkephalin, a decapeptide analogue, and an afamelanotide analogue. At 50 °C, several alternative solvent systems matched or outperformed DMF, with MeCN and EtOAc giving excellent results for shorter sequences, while binary mixtures proved superior for longer peptides. In particular, EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) afforded the best crude purity for the afamelanotide analogue and, after optimization of deprotection and coupling conditions, reached 91% purity. Importantly, this solvent mixture could be recovered by distillation, reducing the process mass intensity (PMI) from 3206 to 1137, corresponding to an approximately 65% decrease in solvent contribution. These results demonstrate that combining SMPS resin with recyclable binary solvents is a promising strategy to improve the sustainability of SPPS without compromising synthetic efficiency.
1) afforded the best crude purity for the afamelanotide analogue and, after optimization of deprotection and coupling conditions, reached 91% purity. Importantly, this solvent mixture could be recovered by distillation, reducing the process mass intensity (PMI) from 3206 to 1137, corresponding to an approximately 65% decrease in solvent contribution. These results demonstrate that combining SMPS resin with recyclable binary solvents is a promising strategy to improve the sustainability of SPPS without compromising synthetic efficiency.
Green foundation1. We are tackling one of the most important unmet requirements in green peptide synthesis, the reduction of the solvent consumption2. By a combination of a new concept of resin (solid support) and a binary solvent, we are able to recycle the solvent and reduce the PMI by 60% 3. This strategy could be carried out in both research and industrial modes on the tonnes scale with the consequence of saving solvent |
Introduction
Over the past few years, peptides have evolved from being primarily biochemical tools to becoming one of the most important classes of active pharmaceutical ingredients (APIs).1,2 From a chemical standpoint, three major classes of therapeutic peptides can be identified: (i) long peptides containing lipophilic side chains, mainly targeting metabolic disorders; (ii) (bi-tri)cyclic peptides containing unnatural amino acids, predominantly developed for oncology; and (iii) disulfide-bridged cyclic peptides used as radioligands. Driven largely by the first class, global peptide production has increased to levels that were unimaginable only a few years ago. At the same time, growing societal awareness and increasingly stringent regulatory requirements have intensified the demand for more sustainable manufacturing processes.3–5 This convergence has fueled the need for peptide synthesis methodologies that rely on more benign reagents, minimize solvent consumption, and enable solvent recovery and reuse within a circular-economy framework.6–12 The urgent need for green peptide synthesis has led to the widespread adoption of quantitative sustainability metrics. Among these, Process Mass Intensity (PMI), defined in eqn (1), is the most commonly used parameter for evaluating the material footprint and environmental impact of synthetic processes.13,14 However, solvent recovery efforts have so far been limited. This is partly because the high boiling points of commonly used solvents make energy-intensive recovery, which can offset its environmental benefits. Nevertheless, when solvent recovery is implemented, the PMI should be adjusted accordingly, as shown in eqn (2).15–17 PMI is calculated as the total mass of all input materials (reagents, solvents, and auxiliary substances) divided by the mass of isolated product. In addition to PMI, a recovery-adjusted metric (PMIr) was employed, in which the mass of recovered solvent is subtracted from the total input prior to normalization.
 | (1) |
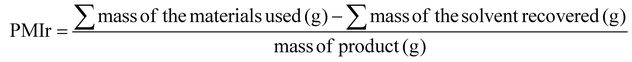 | (2) |
The search for new solvents has primarily focused on those compatible with gel-type polystyrene (PS) resins, as they are the most common on the market, offer good loading, and are affordable. The use of other resins, such as those based on polyethylene glycol (PG), which are more adaptable to a wider range of solvents, has the disadvantage of low functionalization and high price. In this context, only N-butylpyrrolidone (NBP) can represent an alternative to N,N′-dimethylformamide (DMF) as sole solvent.18–20 However, NBP is also an amide base solvent and it is expected to suffer some regulatory restrictions in a near future.21
After evaluating numerous single-solvent systems,22–32 it became apparent that binary solvent mixtures were required to achieve acceptable performance in SPPS.33 Subsequent extensive screening of solvent combinations6,34–40 with conventional polystyrene (PS) resins made typically using a cross-linking of 1 or 2% DVB led to the widespread adoption of two binary systems: ethyl acetate (EtOAc) combined with either dimethyl sulfoxide (DMSO)41,42 or N-butylpyrrolidone (NBP).43,44 However, both DMSO and NBP possess high boiling points and are not readily recyclable, limiting their sustainability.
The development of alternative solvents or solvent systems therefore necessitates the evaluation of different solid supports. In previous work, we introduced two rigid, non-swellable supports—a macroporous polyacrylate resin45 and a silica-based resin46—which enabled the use of alternative solvents while simultaneously reducing solvent consumption due to their structural properties.
In the present study, we propose the use of a Swellable Macroporous PolyStyrene (SMPS) resin as a complementary strategy to enable SPPS in recyclable solvent systems, thereby moving away from the reliance on high-boiling polar aprotic solvents such as DMSO and NBP.
Results and discussion
SMPS resin was developed using a new proprietary technology (patent applied) with the target of decreasing swelling in DMF while allowing swelling in other solvents not compatible with the standard SPPS resins (Fig. 1). These are based on 1 or 2% DVB cross-linking and are typically manufactured according to Fig. 1, where a mixture of styrene and DVB are mixed, followed by a chloromethylation step. Chloromethylation is a step that introduces the functional group in the resin. The Cl group is then transformed into amino groups by Gabriel reaction. The resin that is formed is characterized by very high swelling and is called gel type resin. | ||
| Fig. 1 Comparison of manufacturing process of a typical 1% crosslinked gel-type polystyrene resin and 6% crosslinked SMPS-resin functionalised with Fmoc-RinkAmide linker. | ||
The newly developed SMPS resins were developed using 3 different monomers (Fig. 1) as styrene, divinylbenzene and 4-vinylbenzyl chloride. To achieve a defined porosity and a lower swelling, the amount of DVB was increased to 6% of divinylbenzene and a porogen was used. The function of the porogen is to provide porosity and accessibility of functional groups. The porogen is washed after the suspension polymerization. The obtained polymer was further transformed by introduced a spacer arm, introducing some hydrophilicity and capping the unreacted groups in the non-accessible microporosity of the resin. The so obtained amino methyl (AM) resin with a target of 0.62 mmol g−1 can then be used for the Fmoc-RinkAmide based strategy used in this study. This proprietary technology is flexible, and different spacers can be used, as aliphatic (C2 or C6), phenyl or PEG to make different types of hydrophobicity. For the present study we have used aliphatic C6 spacer.
The advantage of having a macroporous structure is also the increased chemical stability so that the application in organic solvents is becoming broader, including green ones, because SMPS can show some swelling also these (Fig. 2). The most classical gel-type PS resin (1–2% divinylbenzene) exhibits a smooth, non-porous structure that can change depending on the solvent used, whereas the SMPS resin shows macroporous structure characterised by connecting channels as shown in the scanning electron microscopy (SEM) images (Fig. 2a). Macroporous resins are characterized by a Gaussian distribution of porosity as shown in Fig. 2b. In the case of SMPS most pores lays in the range 25–160 nm range, as showed the pore size distribution curves of mercury intrusion (Fig. 2b).
Swelling
The swelling of AM-SMPS-resin was evaluated in different solvents, including green solvents and their binary mixtures,3 with DMF used as a reference. The AM-SMPS-resin swells well in all the solvents and mixtures tested. With the exception of acetonitrile (MeCN), the swelling values range from 4.5 to 7.0 mL g−1. The swelling in DMF was 6.2 mL g−1, whereas the gel-type AM-PS resin swells to around 4.5 mL g−1. In addition to DMF, higher swelling was observed in less polar solvents such as 2-MeTHF and toluene (Tol). This confirms the good diffusion of solvents within the SMPS-resin beads as shown in Fig. 3 (also see SI S1). AM-SMPS-resin was further evaluated in mixture of solvents with EtOAc as one of the choices. For these three mixtures with MeCN, triethylphosphate (TEP), and Tol showed fair swelling properties. The choice of this ratio was made to avoid the formation of azeotropes. This is important as the azeotropic mixtures cannot be distilled. Among the three combinations EtOAc![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Tol showed much better swelling properties and hence was used for further experiments.
Tol showed much better swelling properties and hence was used for further experiments.
Application of AM-SMPS-resin SPPS
First, to initiate the synthesis, the Fmoc-RinkAmide-linker was attached to the AM-SMPS-resin using a standard SPPS protocol. After the incorporation of the linker, the loading resulted in 0.43 mmol g−1, which is very close to the theoretical loading (0.47 mmol g−1) from the nominal loading of 0.62 mmol g−1.To establish a reliable synthetic strategy, we first evaluated the performance of different solvents during Fmoc-based SPPS using a short model pentapeptide, H-YGGFL-NH2. Syntheses were carried out using standard protocol at room temperature on Fmoc-RinkAmide-AM-SMPS-resin (0.043 mmol scale) using the following solvents for coupling, Fmoc removal, and washings: DMF (as reference), MeCN, EtOAc, TEP, acetone, 2-MeTHF, DMSO, Tol and EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) (Scheme 1).
1) (Scheme 1).
 | ||
Scheme 1 Protocol used for the synthesis of H-YGGFL-NH2 using different solvents on Fmoc-RinkAmide-AM-SMPS-resin. Solvents: DMF, MeCN, EtOAc, TEP, acetone, 2-MeTHF, DMSO, Tol and EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). 1). | ||
The results shown in Table 1, Fig. 4, and S2 clearly demonstrate that SMPS-resin is suitable for SPPS. Interestingly, its performance in MeCN (#2, Table 1) is very similar to that observed in DMF (#1, for LC-MS, also see SI S2). Furthermore, it corroborates our previous work showing that MeCN, in combination with swellable resins such as poly(ethylene glycol)-polystyrene (PEG-PS), is a highly effective solvent system for SPPS.47 The main impurities observed are the des-Leu peptide, which is frequently encountered in SPPS, and the protected peptide with Fmoc on. The former can be attributed either to partial Fmoc removal from the RinkAmide-AM-SMPS resin or to inefficient incorporation of the first protected amino acid (Fmoc-Leu-OH). In this case, because an acetylation step was performed after incorporation of Fmoc-Leu-OH, the des-Leu impurity is most likely due to partial removal of the Fmoc group from the RinkAmide moiety. This issue could be mitigated by increasing the resin swelling time and/or extending the Fmoc-removal step. If these two side products are excluded, as they are readily avoidable, syntheses performed in 2-MeTHF (#5), EtOAc (#7), and the EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) mixture (#9) also afford excellent yields. Tol alone (#8) gave acceptable results, whereas acetone (#3), TEP (#4), and DMSO (#6) did not provide satisfactory outcomes. In the case of acetone, an important impurity is the imine terminated peptide (Fig. 4).
1) mixture (#9) also afford excellent yields. Tol alone (#8) gave acceptable results, whereas acetone (#3), TEP (#4), and DMSO (#6) did not provide satisfactory outcomes. In the case of acetone, an important impurity is the imine terminated peptide (Fig. 4).
 | ||
| Fig. 4 Synthesis of H-YGGFL-NH2 at room temperature in different solvents. Gradient: 5–60% B into A in 15 min; flow rate: 1 mL min−1; detection at 220 nm. | ||
| # | Solvents | H-YGGFL-NH2 | Des F | Des Y | Des L | Des G | Fmoc-YGGFL-NH2 | Others | |
|---|---|---|---|---|---|---|---|---|---|
| A | B | C | D | E | F | G | A + D + F | ||
| 1 | DMF | 95.4 | 0.4 | 0.6 | 0.9 | 0.8 | — | 1.9 | 96.3 |
| 2 | MeCN | 95.2 | — | 1.1 | 1.4 | 1.2 | — | 1.1 | 96.6 |
| 3 | Acetone | 78.1 | 0.4 | 0.1 | 1.4 | 1.9 | — | 18.1 | 79.5 |
| 4 | TEP | 25.9 | — | — | 38.8 | — | — | 35.3 | 64.7 |
| 5 | 2-MeTHF | 78.3 | 0.3 | — | 7.0 | 2.1 | 10.9 | 1.4 | 96.2 |
| 6 | DMSO | 72.0 | 5.0 | 3.5 | 0.5 | 14.0 | — | 5.0 | 72.5 |
| 7 | EtOAc | 52.4 | 0.3 | 1.1 | 6.2 | 0.3 | 37.9 | 1.8 | 96.5 |
| 8 | Tol | 71.7 | 1.2 | 2.9 | 9.6 | 7.5 | 6.4 | 0.7 | 87.7 |
| 9 | EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
60.4 | — | 0.4 | 4.0 | 0.7 | 33.5 | 1 | 97.9 |
Synthesis of model peptide YGGFL at 50 °C
Based on these observations, the same solvents, except acetone because at higher temperature, the imine impurity should be more important, were further evaluated at 50 °C in the same peptide synthesis to determine whether heating could overcome the observed limitations by enhancing reaction kinetics, improving resin swelling, and accelerating diffusion of reagents into the polymer matrix. TEP was included despite its low performance at room temperature, as it is a green solvent and has been successfully employed in our previous work,31,37 making it important to assess its behaviour under thermally enhanced conditions. Under these conditions, most of the selected solvents, DMF (#1), MeCN (#2), TEP (#4), EtOAc (#6), and the EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) mixture (#8), afforded HPLC purities above 90%. In addition, 2-MeTHF (#4) and DMSO (#5) showed improved performance at elevated temperature, yielding peptide purities of 84%. In contrast, Tol (#7) remained inefficient, unexpectedly providing only 34% purity (Fig. 5 and Table 2).
1) mixture (#8), afforded HPLC purities above 90%. In addition, 2-MeTHF (#4) and DMSO (#5) showed improved performance at elevated temperature, yielding peptide purities of 84%. In contrast, Tol (#7) remained inefficient, unexpectedly providing only 34% purity (Fig. 5 and Table 2).
 | ||
| Fig. 5 Synthesis of H-YGGFL-NH2 at 50 °C in different solvents. Gradient: 5–60% B into A in 15 min; flow rate: 1 mL min−1; detection at 220 nm. | ||
| # | Solvents | H-YGGFL-NH2 | Des F | Des Y | Des L | Des G | Fmoc-YGGFL-NH2 | Others |
|---|---|---|---|---|---|---|---|---|
| 1 | DMF | 95.9 | 0.9 | 1.4 | 1.2 | 0.6 | — | — |
| 2 | MeCN | 99.1 | — | — | — | — | — | 0.9 |
| 3 | TEP | 95.7 | — | — | 1.3 | 0.6 | — | 2.4 |
| 4 | 2-MeTHF | 83.6 | 4.4 | 1.8 | 0.4 | 4.5 | 1.7 | 3.6 |
| 5 | DMSO | 84.1 | 3.9 | 3.7 | 0.7 | 3.8 | — | 3.8 |
| 6 | EtOAc | 99.3 | 0.3 | — | — | — | — | 0.4 |
| 7 | Tol | 33.6 | 14.4 | 5.1 | 0.1 | 23.9 | 1.4 | 21.5 |
| 8 | EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
92.6 | — | — | 0.7 | 0.3 | 5.0 | 1.4 |
Synthesis of model peptide H-(YGGFL)2-NH2 at 50 °C
As the results for the synthesis of H-YGGFL-NH2 improved at 50 °C, the synthesis of the corresponding decapeptide, H-YGGFLYGGFL-NH2 (twice the length of the pentapeptide YGGFL), was subsequently carried out under the same temperature conditions. At this stage, DMF was used as a reference solvent, and syntheses were performed in MeCN, EtOAc, and TEP, as well as in the solvent mixtures EtOAc–MeCN, EtOAc–TEP, and EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), to assess whether the enhanced efficiency observed at elevated temperature could be maintained during longer peptide assembly (Fig. 6, Table 3 and Fig. S3). Interestingly, the poorest results were obtained with DMF (#1, for LC-MS, also see SI S3). Even TEP (#4), which had shown the weakest performance at room temperature, afforded slightly better results than DMF under these conditions. Once again, MeCN (#2) delivered the best performance, closely followed by EtOAc (#3). The three EtOAc-based mixtures with MeCN (#5), TEP (#6), and Tol (#7) also provided more than acceptable results, particularly considering that des-Leu is a major impurity in all of these syntheses. Overall, these results confirm that the use of simple solvents such as EtOAc, MeCN, and Tol, as well as their mixtures, enables efficient solid-phase peptide synthesis when an appropriate resin is employed.
1), to assess whether the enhanced efficiency observed at elevated temperature could be maintained during longer peptide assembly (Fig. 6, Table 3 and Fig. S3). Interestingly, the poorest results were obtained with DMF (#1, for LC-MS, also see SI S3). Even TEP (#4), which had shown the weakest performance at room temperature, afforded slightly better results than DMF under these conditions. Once again, MeCN (#2) delivered the best performance, closely followed by EtOAc (#3). The three EtOAc-based mixtures with MeCN (#5), TEP (#6), and Tol (#7) also provided more than acceptable results, particularly considering that des-Leu is a major impurity in all of these syntheses. Overall, these results confirm that the use of simple solvents such as EtOAc, MeCN, and Tol, as well as their mixtures, enables efficient solid-phase peptide synthesis when an appropriate resin is employed.
 | ||
| Fig. 6 Synthesis of H-(YGGFL)2-NH2 at 50 °C in different solvents. Gradient: 5–60% B into A in 15 min; flow rate: 1 mL min−1; detection at 220 nm. | ||
| # | Solvent | H-(YGGFL)2-NH2 | Des F | Des L | Des G | Others |
|---|---|---|---|---|---|---|
| 1 | DMF | 77.0 | 4.0 | 0.8 | 5.2 | 13.0 |
| 2 | MeCN | 90.9 | — | 0.7 | — | 8.4 |
| 3 | EtOAc | 89.9 | — | 3.6 | — | 6.5 |
| 4 | TEP | 79.0 | — | 3.2 | — | 17.8 |
| 5 | EtOAc–MeCN (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
90.0 | — | 2.0 | 1.0 | 7.0 |
| 6 | EtOAc–TEP (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
84.7 | — | 3.6 | 2.6 | 9.1 |
| 7 | EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
84.7 | — | 8.3 | 1.6 | 5.4 |
Synthesis of an afamelanotide analogue (13-mer peptide) at 50 °C
Finally, the performance of these solvent systems was evaluated in the synthesis of an analogue of afamelanotide, H-Ser-Tyr-Ser-Leu-Glu-His-Phe-Arg-Trp-Gly-Lys-Pro-Val-NH2, carried out at 50 °C. In this model system, the two major impurities observed in all syntheses were des-Lys and des-Ser (Fig. 7, Table 4 and Fig. S4). Single-solvent systems using DMF (#1), MeCN (#2), and EtOAc (#3) afforded comparable results, yielding crude peptide purities of approximately 66–70%. In contrast, TEP (#4) again showed the poorest performance, providing less than 40% purity. As observed for the decapeptide synthesis, the three binary mixtures of EtOAc with MeCN (#5), TEP (#6), and Tol (#7) delivered the highest purities, all exceeding 75%. Among them, the EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) mixture gave the best result, affording a crude peptide purity of 81% (Fig. 7, for LC-MS, see Fig. S4). Overall, while single-solvent systems produced only moderate purities, the use of mixed solvent systems led to markedly improved outcomes, with purities in the range of 77–81%. The superior performance of the EtOAc–Tol (3
1) mixture gave the best result, affording a crude peptide purity of 81% (Fig. 7, for LC-MS, see Fig. S4). Overall, while single-solvent systems produced only moderate purities, the use of mixed solvent systems led to markedly improved outcomes, with purities in the range of 77–81%. The superior performance of the EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) mixture is likely due to an optimal balance between polarity and hydrophobicity, enhanced mass transfer, and reduced aggregation-induced side reactions. Consequently, this solvent mixture was identified as the most effective system for further optimization.
1) mixture is likely due to an optimal balance between polarity and hydrophobicity, enhanced mass transfer, and reduced aggregation-induced side reactions. Consequently, this solvent mixture was identified as the most effective system for further optimization.
 | ||
| Fig. 7 Synthesis of afamelanotide analogue at 50 °C in different solvents. Gradient: 5–60% B into A in 15 min; flow rate: 1 mL min−1; detection at 220 nm. | ||
| # | Solvents | Afamelanotide analogue | Des S | Des K | Others |
|---|---|---|---|---|---|
| 1 | DMF | 79.1 | 13.3 | 1.1 | 6.5 |
| 2 | MeCN | 72.4 | 14.9 | 0.9 | 11.8 |
| 3 | EtOAc | 77.5 | 13.0 | 1.5 | 8.0 |
| 4 | TEP | 42.0 | 10.9 | 1.7 | 45.4 |
| 5 | EtOAc–MeCN (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
77.7 | 13.0 | 3.8 | 5.5 |
| 6 | EtOAc–TEP (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
78.4 | 12.0 | 1.6 | 8.0 |
| 7 | EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
81.2 | 10.0 | 5.6 | 3.2 |
Optimization in the synthesis of afamelanotide analogue (13-mer peptide)
In the previous section, it was demonstrated that the afamelanotide analogue can be synthesized with reasonably good yields using SMPS resin in combination with binary solvent mixtures of EtOAc with MeCN, TEP, or Tol. Among the conditions screened, the EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) mixture afforded the highest crude purity (81%). Based on this result, and considering that EtOAc, Tol, and DMSO are classified as “yellow” (i.e., presenting some concerns) in the GSK solvent selection guide,48 this binary system was selected for further optimization. Detailed LC-MS analysis of the crude peptide revealed the presence of des-Ser and des-Leu deletion sequences, indicating incomplete coupling of these residues and/or insufficient Fmoc removal of the preceding amino acids. To address these issues, two complementary optimization strategies were implemented. First, the Fmoc-removal time was increased from 15 to 30 min following the incorporation of Leu, Tyr, Pro, and His-residues identified as critical points in the sequence where incomplete deprotection could adversely affect subsequent coupling efficiency. Second, the coupling protocol was strengthened by the addition of an extra 3 eq. of DIC after 1 h, followed by an additional 30 min coupling period, to drive the reaction to completion. These modifications significantly reduced the formation of deletion sequences, demonstrating that extended Fmoc removal and reinforced coupling conditions are effective strategies for improving the crude purity of the afamelanotide analogue, which reached 91% in the EtOAc–Tol binary solvent system (Fig. 8, for LC-MS, see Fig. S5).
1) mixture afforded the highest crude purity (81%). Based on this result, and considering that EtOAc, Tol, and DMSO are classified as “yellow” (i.e., presenting some concerns) in the GSK solvent selection guide,48 this binary system was selected for further optimization. Detailed LC-MS analysis of the crude peptide revealed the presence of des-Ser and des-Leu deletion sequences, indicating incomplete coupling of these residues and/or insufficient Fmoc removal of the preceding amino acids. To address these issues, two complementary optimization strategies were implemented. First, the Fmoc-removal time was increased from 15 to 30 min following the incorporation of Leu, Tyr, Pro, and His-residues identified as critical points in the sequence where incomplete deprotection could adversely affect subsequent coupling efficiency. Second, the coupling protocol was strengthened by the addition of an extra 3 eq. of DIC after 1 h, followed by an additional 30 min coupling period, to drive the reaction to completion. These modifications significantly reduced the formation of deletion sequences, demonstrating that extended Fmoc removal and reinforced coupling conditions are effective strategies for improving the crude purity of the afamelanotide analogue, which reached 91% in the EtOAc–Tol binary solvent system (Fig. 8, for LC-MS, see Fig. S5).
 | ||
| Fig. 8 Synthesis of afamelanotide analogue at 50 °C after optimization. Gradient: 5–60% B into A in 15 min; flow rate: 1 mL min−1; detection at 220 nm. | ||
Distillation of solvent, EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) 1) and process mass intensity (PMI)
1) and process mass intensity (PMI)
In peptide synthesis, sustainability is commonly addressed either by reducing solvent consumption or by implementing solvent recovery through distillation. In industrial manufacture of APIs, however, solvent distillation is not easy to apply, as the collected waste streams from coupling, washing, and Fmoc removal steps typically contain piperidine, excess amino acids, and coupling reagents, all of which complicate solvent recovery. Consequently, several parameters must be carefully considered before distillation can be applied and still be cost-effective. To evaluate the feasibility and impact of solvent recovery, the synthesis of Afamelanotide analogue was selected as a representative case study and its process mass intensity (PMI) was first calculated without solvent recovery. Under these conditions, a PMI of 3206 was obtained in agreement with other SPPS (in case of polystyrene resin and with DMF as solvent PMI for same peptide is 2242).49 The solvent system employed for the synthesis, a mixture of ethyl acetate and Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), was collected separately. Notably, this solvent combination is reported not to form an azeotrope,50 rendering it amenable to distillation.
1), was collected separately. Notably, this solvent combination is reported not to form an azeotrope,50 rendering it amenable to distillation.
After afamelanotide analogue synthesis, the total waste was treated with 0.1 N HCl to remove excess unreacted piperidine. Upon extraction, a total of 160 mL of solvent waste was collected and distillation was performed. After distillation successfully 116 mL of solvent mixture was recovered. The obtained distilled mixture was further confirmed by 1H NMR (Fig. 9). Incorporation of this solvent recovery into the PMI calculation (eqn (2)) resulted in a substantially reduced PMI (PMIr) of 1137. This corresponds to an approximate 65% decrease in solvent contribution, clearly demonstrating the significant impact of solvent recovery on improving the sustainability profile of the peptide synthesis.
The solvent recovered can be again defined to be in the same ratio of EtOAc![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Tol (3
Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and, after an adjustment, it can be reused as solvent in the further synthesis, making the process more sustainable. The methodology is also amenable to translation into automated peptide synthesizers, supporting its robustness and practical applicability. Furthermore, the recovered solvent stream may be repurposed for use in other industrial applications (e.g., paints and varnishes), contributing to waste minimization and resource efficiency.
1) and, after an adjustment, it can be reused as solvent in the further synthesis, making the process more sustainable. The methodology is also amenable to translation into automated peptide synthesizers, supporting its robustness and practical applicability. Furthermore, the recovered solvent stream may be repurposed for use in other industrial applications (e.g., paints and varnishes), contributing to waste minimization and resource efficiency.
Experimental
Materials, reagents and methods
For SPPS, a syringe with a polypropylene filter was used and procured from Chemetrix. Macroporous polystyrene resin (SMPS) polymerized with 6% of divinylbenzene (0.62 mmol g−1) were manufactured using a priority method by Sunresin (Xi'an, China). The solvents used for synthesis DMF, acetonitrile (MeCN), ethyl acetate (EtOAc), acetone, toluene (Tol), DMSO and 2-methyltetrahydrofuran (2-MeTHF) were purchased from Honeywell. Another solvent used, triethyl phosphate (TEP) and reagents, N,N″-diisopropylcarbodiimide (DIC) and OxymaPure were generous gifts from Luxembourg Bio Technologies (Nes Ziona, Israel). The remaining reagents, diisopropylethylamine (DIEA), acetic anhydride (Ac2O), trifluoroacetic acid (TFA), triisopropylsilane (TIS) and solvents were from diverse commercially available sources. Piperidine (PIP) was purchased from Sigma-Aldrich. Fmoc-amino acids (Fmoc-AA-OH) were sourced from Iris Biotech (Marktredwitz, Germany). Additional organic solvents and HPLC-grade MeCN were obtained from Merck. Milli-Q water was used for reverse-phase High Performance Liquid Chromatography (RP-HPLC) analyses on Shimadzu-LC-2050 equipped with Phenomenex Aeris™C18 (3.6 μm, 4.6 × 150 mm) column. The separations were carried out at a flow rate of 1.0 mL min−1 with UV detection at 220 nm and data was processed using LabSolutions software. The mobile phase consisted of buffer A: 0.1% TFA in H2O; buffer B: 0.1% TFA in MeCN. LC-MS analyses were conducted on Thermo Scientific Dionex UltiMate 3000 with a Phenomenex Aeris™C18 (3.6 μm, 4.6 × 150 mm) column. The mobile phase comprised buffer A (0.1% formic acid in water) and buffer B (0.1% formic acid in MeCN).Solid-phase peptide synthesis
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1) (sol. A). 400 mL of sol A was prepared. 300 mL of EtOAc was placed in a 500 mL measuring cylinder and the volume was made up to 400 mL with Tol. Similarly, EtOAc–MeCN (3
1) (sol. A). 400 mL of sol A was prepared. 300 mL of EtOAc was placed in a 500 mL measuring cylinder and the volume was made up to 400 mL with Tol. Similarly, EtOAc–MeCN (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and EtOAc–TEP (3
1) and EtOAc–TEP (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) were prepared.
1) were prepared.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 20% PIP/EtOAc–TEP (3
1), 20% PIP/EtOAc–TEP (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 20% PIP/EtOAc–Tol (3
1), 20% PIP/EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) were prepared.
1) were prepared.Swelling of AM-SMPS-resin
SMPS resin (200 mg) was loaded into a 5 mL polypropylene fritted syringe and swollen with solvent (10 vol) at 25 °C on a shaker for 30 min. The resin was then compressed, and the solvent was removed by filtration. The piston was slowly pulled to allow the resin to reach its maximum swollen volume, which was recorded. A void volume of 0.15 mL (syringe tip and frit) was subtracted from the measured value.The swelling was calculated using the following formula:
Fmoc-RinkAmide linker incorporation on AM-SMPS-resin
Firstly, Fmoc-RinkAmide linker was incorporated on AM-SMPS-resin using standard protocol in DMF, Fmoc-RinkAmide-OH linker (3.34 g, 5.0 eq.) and OxymaPure (881 mg, 5.0 eq.) were weighed and dissolved in DMF (20 mL, 10 vol), and then DIC was added (966.0 µL, 5.0 eq.) into the solution. After 1 min pre-activation, the solution was transferred to swelled AM-SMPS-resin in a syringe reaction vessel and kept on a shaker for 2 h at RT. The resin was filtered after 2 h and washed with DMF (10 vol × 3). To cap the non-reacted sites, acetylation was done using Ac2O (703.0 µL, 10.0 eq.) and DIEA (4.3 mL, 20.0 eq.) dissolved in DMF (10 vol) and then transferred to the syringe reaction vessel and kept for 30 min on a shaker at RT. Finally, the resin was filtered and washed with DMF (10 vol × 3) + DCM (10 vol × 3) and fully dried under vacuum to render 2.64 g of the target resin. Yield = 99.6%.Loading quantification
The loading (substitution) of Fmoc-RinkAmide-AM-SMPS-resin was determined spectrophotometrically. Fmoc-RinkAmide-AM-SMPS-resin (10 mg) was treated with 20% PIP/DMF (2.5 mL) in a volumetric flask. This mixture was then kept on a shaker for 30 min at RT. Next, MeOH was added to the flask, and the volume was made up to 50 mL. Similarly, blank solution was prepared in another volumetric flask. The absorbance of this solution was measured in a spectrophotometer at 300 nm using a quartz cuvette. Resin loading was calculated using the following equation:| L = (abs × vol)/ε × l × m |
Synthesis of peptides
The model pentapeptide Leu-enkephalin (YGGFL), decapeptide (YGGFLYGGFL) and afamelanotide analogue (13 mer peptide) were synthesized on Fmoc-RinkAmide-AM-SMPS-resin using standard Fmoc-based SPPS in various single and binary solvent systems. The synthesis of YGGFL was carried out at both 25 °C and 50 °C, whereas decapeptide and afamelanotide analogue were synthesized at 50 °C only. The general procedure for synthesis is described below.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5
2.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5) (10 vol) was used for cleaving peptide from resin for 1 hour at RT. The crude peptide was precipitated by the addition of cold methyl tert-butylether (MTBE) (10 vol) and then collected by centrifugation. The supernatant was discarded and the precipitation step was repeated twice. The residue was evaporated to dryness and then dissolved in water and acetonitrile for HPLC analysis, monitored at 220 nm.
2.5) (10 vol) was used for cleaving peptide from resin for 1 hour at RT. The crude peptide was precipitated by the addition of cold methyl tert-butylether (MTBE) (10 vol) and then collected by centrifugation. The supernatant was discarded and the precipitation step was repeated twice. The residue was evaporated to dryness and then dissolved in water and acetonitrile for HPLC analysis, monitored at 220 nm.Distillation of EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) 1)
1)
Distillation was carried out to assess the feasibility of recovering EtOAc, Tol or a mixture from the solvent waste produced during the afamelanotide analogue synthesis in EtOAc–Tol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) solvent system. The distillation setup comprised an oil-bath heater for controlled heating of the solution, a Vigreux fractionating column equipped with a temperature probe, a condenser, and a receiver where the distillate was collected.
1) solvent system. The distillation setup comprised an oil-bath heater for controlled heating of the solution, a Vigreux fractionating column equipped with a temperature probe, a condenser, and a receiver where the distillate was collected.
Conclusions
The work described in this manuscript confirms that improving sustainability in SPPS requires a multifactorial approach. Traditionally, our group and others have focused on modifying a single parameter, most often the solvent, while keeping all other variables (resin, coupling reagents, and protocols) unchanged. As a consequence, with the notable exception of NBP, no alternative solvent has been able to fully replace DMF when gel-type polystyrene resins (1–2% divinylbenzene) are used as solid support. This limitation has driven the exploration of binary solvent mixtures, with the most widely adopted systems combining DMSO or NBP with other solvents, such as EtOAc (the most common), tBuOAc, TEP, or IPA. However, both DMSO and NBP are classified as “yellow” solvents and possess very high boiling points (189 °C for DMSO and 240 °C for NBP), which preclude efficient recovery by distillation, as DMF (bp 153 °C). In this context, the investigation of solvent systems composed of more benign components and with boiling points compatible with solvent recycling becomes particularly attractive.Herein, we demonstrate that the use of a swellable macroporous polystyrene (SMPS) resin enables efficient SPPS in alternative solvents such as MeCN, EtOAc, and 2-MeTHF, as well as their mixtures, without the need for high-boiling dipolar aprotic solvents such as DMSO or NBP. As a first example, we propose the EtOAc–Tol binary mixture; although we acknowledge that Tol is less benign than DMF and therefore not an ideal green solvent, this system serves as a proof of concept.
This work complements our previous studies, in which the use of rigid resins allowed a significant reduction in solvent consumption.45,46 Ongoing efforts in our laboratories are focused on combining both approaches, minimizing solvent use while enabling solvent recycling, to further advance the sustainability of SPPS.
Author contributions
M. K., S. B., and A. K. performed the swelling experiments and the peptide synthesis; M. K. prepared the first draft of the manuscript; Sunresin researchers designed and prepared the SMPS resin; A. S. was responsible for PMI and NMR analysis. B. G. T. and F. A. supervised the research work at UKZN and prepared the last version of the manuscript; A. S. and A. K. contributed to the preparation of the first draft; the final version of the manuscript got the consent of all the authors.Conflicts of interest
Sunresin researchers declare that Sunresin is selling resins for SPPS. The rest of authors declare no conflict of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. HPLC and Mass data is provided in supplementary information. See DOI: https://doi.org/10.1039/d6gc01716a.Acknowledgements
We thank Yoav Luxembourg for extending support for this work.References
- M. Muttenthaler, G. F. King, D. J. Adams and P. F. Alewood, Nat. Rev. Drug Discovery, 2021, 20, 309–325 CrossRef CAS.
- D. AlShaer, O. Al Musaimi, F. Albericio and B. G. de la Torre, Pharmaceuticals, 2026, 19, 244 CrossRef PubMed.
- O. Al Musaimi, B. G. de la Torre and F. Albericio, Green Chem., 2020, 22, 996–1018 RSC.
- L. Ferrazzano, M. Catani, A. Cavazzini, G. Martelli, D. Corbisiero, P. Cantelmi, T. Fantoni, A. Mattellone, C. De Luca, S. Felletti, W. Cabri and A. Tolomelli, Green Chem., 2022, 24, 975–1020 RSC.
- B. G. de la Torre and F. Albericio, Nat. Sustainability, 2026, 9, 484–485 CrossRef.
- J. Pawlas and J. H. Rasmussen, ChemSusChem, 2021, 14, 3231–3236 CrossRef CAS PubMed.
- I. Kekessie, K. Wegner, I. Martinez, M. E. Kopach, T. D. White, J. K. Tom, M. N. Kenworthy, F. Gallou, J. Lopez, S. G. Koenig, P. R. Payne, S. Eissler, B. Arumugam, C. Li, S. Mukherjee, A. Isidro-Llobet, O. Ludemann-Hombourger, P. Richardson, J. Kittelmann, D. Sejer Pedersen and L. J. van den Bos, J. Org. Chem., 2024, 89, 4261–4282 CrossRef CAS.
- S. B. H. Kent, J. Pept. Sci., 2025, 31, e70013 CrossRef CAS.
- M. U. Luescher, A. Valotta, J. Haber and F. Gallou, Chimia, 2025, 79, 606–613 CrossRef CAS.
- P. Richardson, M. C. Bryan, L. Diorazio, F. Gallou, S. Keily, I. Martinez, S. Plummer, A. B. Reed, R. Sheppard, J. Shields, G. Wrigley, C. Yeung and Á. Enríquez-García, J. Med. Chem., 2025, 68, 25625–25664 CrossRef CAS.
- Z. Cai, L. Liu, C. Zou, X. Jiang, G. Wang, X. He, W. Zhuang, C. Yang, Y. Yao, X. Sun, C. Ye and W. Hu, Biofabrication, 2026, 18, 012001 CrossRef CAS.
- N. Maarouf-Mesli, J. Desjardins-Michaud, Z. Imani, K. Ghosh, F. Polyak and W. D. Lubell, Green Chem., 2026, 28, 264–276 RSC.
- R. A. Sheldon, M. L. Bode and S. G. Akakios, Curr. Opin. Green Sustainable Chem., 2022, 33, 100569 CrossRef CAS.
- M. Ferrara, F. Della Negra and M. Verzini, Org. Process Res. Dev., 2025, 29, 1110–1124 CrossRef CAS.
- G. Martelli, P. Cantelmi, C. Palladino, A. Mattellone, D. Corbisiero, T. Fantoni, A. Tolomelli, M. Macis, A. Ricci, W. Cabri and L. Ferrazzano, Green Chem., 2021, 23, 8096–8107 RSC.
- T. Fantoni, A. Tolomelli and W. Cabri, Catal. Today, 2022, 397–399, 265–271 CrossRef CAS.
- C. Palladino, T. Fantoni, L. Ferrazzano, A. Tolomelli and W. Cabri, ACS Sustainable Chem. Eng., 2023, 11, 15994–16004 CrossRef CAS.
- J. Lopez, S. Pletscher, A. Aemissegger, C. Bucher and F. Gallou, Org. Process Res. Dev., 2018, 22, 494–503 CrossRef CAS.
- B. G. de la Torre, A. Kumar, M. Alhassan, C. Bucher, F. Albericio and J. Lopez, Green Chem., 2020, 22, 3162–3169 RSC.
- M. A. Malone, M. De Francisco Craparotta, K. I. M. Arnott, A. G. Jamieson and N. Wade, Org. Biomol. Chem., 2026, 24, 1496–1502 RSC.
- J. Sherwood, F. Albericio and B. G. de la Torre, ChemSusChem, 2024, 17, e202301639 CrossRef CAS.
- Y. E. Jad, G. A. Acosta, S. N. Khattab, B. G. de la Torre, T. Govender, H. G. Kruger, A. El-Faham and F. Albericio, Amino Acids, 2016, 48, 419–426 CrossRef CAS.
- G. Vivenzio, M. C. Scala, P. Marino, M. Manfra, P. Campiglia and M. Sala, Pharmaceutics, 2023, 15, 1773 CrossRef CAS PubMed.
- A. Kumar, Y. E. Jad, J. M. Collins, F. Albericio and B. G. de la Torre, ACS Sustainable Chem. Eng., 2018, 6, 8034–8039 CrossRef CAS.
- Y. E. Jad, G. A. Acosta, T. Govender, H. G. Kruger, A. El-Faham, B. G. de la Torre and F. Albericio, ACS Sustainable Chem. Eng., 2016, 4, 6809–6814 CrossRef CAS.
- A. Kumar, Y. E. Jad, A. El-Faham, B. G. de la Torre and F. Albericio, Tetrahedron Lett., 2017, 58, 2986–2988 CrossRef CAS.
- G. Martelli, P. Cantelmi, A. Tolomelli, D. Corbisiero, A. Mattellone, A. Ricci, T. Fantoni, W. Cabri, F. Vacondio, F. Ferlenghi, M. Mor and L. Ferrazzano, Green Chem., 2021, 23, 4095–4106 RSC.
- S. B. Lawrenson, R. Arav and M. North, Green Chem., 2017, 19, 1685–1691 RSC.
- N. Varró, B. Mándityné Huszka, E. Erdei, A. Mándoki and I. M. Mándity, Chem.: Methods, 2025, 5, 2500010 Search PubMed.
- A. Kumar, A. Sharma, B. G. de la Torre and F. Albericio, Green Chem. Lett. Rev., 2021, 14, 545–550 CrossRef CAS.
- K. P. Nandhini, N. Cele, B. G. de la Torre and F. Albericio, Green Chem. Lett. Rev., 2024, 17, 2330639 CrossRef.
- D. A. Wellings, J. Greenwood, I. Thomas, C. Hughes, W. Li, F. Lin, M. A. Hossain, A. Lanza, M. Meldal and J. D. Wade, Nat. Sustainability, 2026, 9, 565–574 CrossRef.
- L. Ferrazzano, D. Corbisiero, G. Martelli, A. Tolomelli, A. Viola, A. Ricci and W. Cabri, ACS Sustainable Chem. Eng., 2019, 7, 12867–12877 CrossRef CAS.
- G. Vivenzio, M. C. Scala, G. Auriemma, C. Sardo, P. Campiglia and M. Sala, Green Chem. Lett. Rev., 2024, 17, 2404234 CrossRef.
- L. Pacini, M. Muthyala, L. Aguiar, R. Zitterbart, P. Rovero and A. M. Papini, J. Pept. Sci., 2024, 30, e3605 CrossRef CAS.
- L. Pacini, M. K. Muthyala, R. Zitterbart, O. Marder, P. Rovero and A. M. Papini, Int. J. Pept. Res. Ther., 2025, 31, 43 CrossRef CAS.
- S. Noki, L. Lastella, S. Serban, A. Basso, G. Orosz, B. G. de la Torre and F. Albericio, Green Chem. Lett. Rev., 2025, 18, 2438068 CrossRef.
- L. Lastella, M. Zecca, P. Centomo, K. Jeřábek, F. Formaggio, I. Guryanov, A. Ricci and B. Biondi, J. Pept. Sci., 2025, 31, e70038 CrossRef CAS PubMed.
- J. Pawlas, T. Qvist and L. M. Haugaard-Kedström, J. Org. Chem., 2025, 90, 15909–15915 CrossRef CAS.
- A. Phungula, A. Kumar, M. Kaushal, C. Tucker, L. Chen, B. G. de la Torre and F. Albericio, ACS Sustainable Chem. Eng., 2025, 13, 19833–19848 CrossRef CAS.
- V. Martin, S. Jadhav, P. H. G. Egelund, R. Liffert, H. J. Castro, T. Krüger, K. F. Haselmann, S. T. Le Quement, F. Albericio, F. Dettner, C. Lechner, R. Schönleber and D. S. Pedersen, Green Chem., 2021, 23, 3295–3311 RSC.
- S. Jadhav, V. Martin, P. H. G. Egelund, H. J. Castro, T. Krüger, F. Richner, S. T. Le Quement, F. Albericio, F. Dettner, C. Lechner, R. Schönleber and D. S. Pedersen, Green Chem., 2021, 23, 3312–3321 RSC.
- M. Erny, M. Lundqvist, J. H. Rasmussen, O. Ludemann-Hombourger, F. Bihel and J. Pawlas, Org. Process Res. Dev., 2020, 24, 1341–1349 CrossRef CAS.
- J. Pawlas, B. Antonic, M. Lundqvist, T. Svensson, J. Finnman and J. H. Rasmussen, Green Chem., 2019, 21, 2594–2600 RSC.
- S. Noki, A. Kumar, C. Zhang, A. Basso, S. Serban, X. Kou, Y. Li, A. Sharma, B. de la Torre and F. Albericio, ChemSusChem, 2026, 8936857 Search PubMed.
- N. Nkwanyana, A. Kumar, A. Chakraborty, S. Potvin, G. Thibaut-Koumba, B. Nardone, F. Beland, A. Sharma, B. G. de la Torre and F. Albericio, J. Pept. Sci., 2025, 31, e70052 CrossRef CAS PubMed.
- G. A. Acosta, M. del Fresno, M. Paradis-Bas, M. Rigau-DeLlobet, S. Côté, M. Royo and F. Albericio, J. Pept. Sci., 2009, 15, 629–633 CrossRef CAS.
- R. K. Henderson, C. Jiménez-González, D. J. C. Constable, S. R. Alston, G. G. A. Inglis, G. Fisher, J. Sherwood, S. P. Binks and A. D. Curzons, Green Chem., 2011, 13, 854–862 RSC.
- A. Kumar, A. Sharma, B. G. de la Torre and F. Albericio, Green Chem., 2022, 24, 4887–4896 RSC.
- F. Albericio, Curr. Chem. Biol., 2017, 11, 1 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |