Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Reactivity under mechanochemical conditions: a new and more sustainable era for hypervalent iodine?
Pernilla
Öberg
 a,
Sayad
Doobary
a,
Sayad
Doobary
 *ab and
Berit
Olofsson
*ab and
Berit
Olofsson
 *ab
*ab
aDepartment of Chemistry, Stockholm University, 106 91 Stockholm, Sweden. E-mail: berit.olofsson@su.se
bStockholm University Center for Circular and Sustainable Systems (SUCCeSS), Stockholm University, 106 91 Stockholm, Sweden
First published on 10th March 2026
Abstract
Hypervalent iodine reagents can mediate or catalyze a wide variety of organic reactions, and have hence become important tools in the synthetic toolbox of organic chemists. Recent developments in the field include reactions under mechanochemical conditions, ranging from reports on similar or improved reactivity compared to the solution-phase, to papers describing novel reaction pathways. As mechanochemistry has the potential to improve the sustainability profile of reactions by, for instance, avoiding reaction solvents and shortening reaction times, this critical review provides a sustainability comparison of mechanochemical and solution-phase reactions with iodine(III) compounds. The sustainability profiles were evaluated by comparing reaction yields, solvent properties and calculated E-factors, based on data available in the literature. Overall, the comparison provides strong support that mechanochemical hypervalent iodine reactions have improved sustainability compared to solution-phase, often with more than ten-fold decrease in waste production, and avoid a range of solvents with major issues according to the GSK solvent guide.
Green foundation1. Hypervalent iodine reactions are popular in synthesis, as their reactions often display high selectivity under mild reaction conditions, without the need for inert conditions or transition metal catalysts. The low-toxicity and stable nature of these compounds is attractive, but the sustainability profile is hampered by the need for solvents that are often classed as highly hazardous. In this critical review, we evaluate their reactivity under mechanochemical conditions, which avoid the use of solvents.2. The review describes the reaction performance, as well as the waste profile, of selected mechanochemical iodine(III) reactions in comparison to the corresponding solution-phase reactions, highlighting sustainability benefits as well as novel reactivity, providing guidance to readers on which reaction setups are more sustainable for each type of reaction. 3. The growing field of mechanochemistry has the potential to make organic synthesis more sustainable, and the green chemistry analysis used in this review should be applicable beyond iodine(III) reactions, providing further insight into sustainable organic synthesis. |
1 Introduction
Hypervalent iodine chemistry has in recent decades developed into a flourishing field of research, with a wide variety of application areas ranging from organic synthesis to polymer chemistry.1 Hypervalent iodine reagents are generally bench-stable, display low toxicity, and do not require inert reaction conditions, which are beneficial properties from a sustainability point of view. Drawbacks of this type of chemistry include the production of stoichiometric iodoarene (ArI) waste, and the common use of hazardous solvents both for reagent synthesis and use.1a,bWhile the stoichiometric ArI waste can in some cases be avoided through the development of catalytic reactions,2 the solvent use remains problematic. Due to the polarized structure of common hypervalent iodine reagents (Fig. 1), solubility can be challenging.3 Beyond the polymeric structures PhIO and 2-iodoxybenzoic acid (IBX), also (diacetoxyiodo)benzene (DIB), [hydroxy(tosyloxy)iodo]benzene (HTIB), many iodonium salts, and benziodoxolones are sparingly soluble in most aprotic organic solvents. DMP and the fluorinated reagents [bis(trifluoroacetoxy)iodo]benzene (PIFA) and benziodoxoles display better solubility. The synthesis and application of hypervalent iodine reagents are frequently executed in the halogenated solvents dichloromethane (DCM), dichloroethane (DCE), chloroform, trifluoroethanol (TFE) and hexafluoroisopropanol (HFIP). Other solvents are dimethylformamide (DMF), tetrahydrofuran (THF) and rarely diethyl ether.1a Common solvent sustainability guides categorize all of these as “Red – major issues/hazardous”,4 except for HFIP, which has not been evaluated in the guides but has a toxicity profile similar to that of TFE.5
The increasing use of acetonitrile (MeCN) and toluene, both listed as “Amber – some issues/problematic” in the solvent guides,4 is an important step in the right direction. However, solvent-free methodology for synthesis and applications of hypervalent iodine compounds would be very attractive from a sustainability point of view.
Pioneering studies combining mechanochemistry with synthetic organic chemistry have demonstrated that mechanochemical organic reactions alleviate the need for a reaction solvent, can be high-yielding and time-efficient, can be less sensitive to air and moisture, and can sometimes give different reaction selectivity.6 The use of liquid-assisted grinding (LAG), i.e., the addition of a minimal amount of a liquid to the reaction vessel, can often facilitate mechanochemical reactions. While common solvents are often utilized as LAG additives, the volumes are considerably smaller and less of a sustainability concern.
Early reports on solvent-free reactions with hypervalent iodine reagents highlight that reactivity can be achieved through simple stirring under neat conditions,7 and the combination with mechanochemical activation through grinding with a pestle and mortar or ball-milling is becoming more common. While many papers focus on reproducing reactions described in solution, the potential of combining mechanochemistry with hypervalent iodine chemistry is illustrated by several recent reports focusing on novel reactions that are unreported in solution.
2 Methodology and literature selection
2.1 Methods
In this critical review, we describe the synthesis and use of hypervalent iodine compounds in mechanochemical reactions. For a selected set of iodine(III) reactions, we have performed a sustainability analysis, comparing the mechanochemical reactions to the corresponding solution-phase methods. The latter were selected to be as similar as possible to the mechanochemical reactions and refer only to transformations using hypervalent iodine reagents. The comparison includes yields for selected products, a sustainability assessment of the toxicity of the reaction solvent vs. LAG using the GSK solvent guide,4a and waste formation quantified using E-factors.8 As few literature reports contain such data, we have calculated the E-factors for the transformations that have sufficient data to do so. We decided to calculate the E-factors without purification,5 both to focus on the reactivity differences between mechanochemistry and solution-phase, and because of insufficient data on workup and purification in the reviewed papers. Solution-phase reactions performed on a much larger scale than the mechanochemical versions are presented without E-factors, as such comparisons are of limited use.Many methods have been demonstrated across a broader substrate range in solution, and later with a smaller mechanochemical scope. It is unclear whether this difference reflects an inherent advantage of the solvent-based method, or merely the extent of the substrates investigated, and so we have refrained from in-depth discussions of the scope and limitations of the methods.
We have limited the solution-phase comparison and sustainability analysis to reactions utilizing the unique reactivity of the iodine(III) core to form new bonds, beyond simple oxidation. Other mechanochemical reactions with hypervalent iodine compounds are briefly described below.
2.2 Mechanochemical iodine(III) reactions
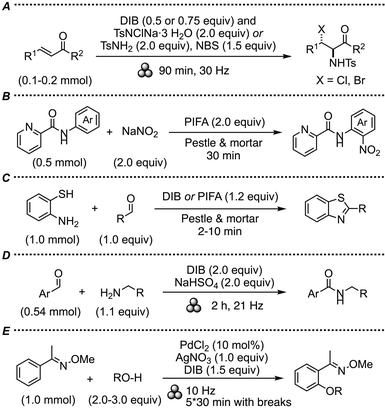
DIB is often utilized both as a substrate in the synthesis of more complex iodine(III) reagents, and as a potent oxidant.1a,b In this fashion, the synthesis of oligomeric iodosobenzene sulfate (PhIO)3·SO3 was initiated by grinding DIB with NaHSO4 in a mortar, followed by stirring in water.9 Early reports from Ando and co-workers detailed that pulverization of iodosobenzene and HCl-treated silica gel in a mortar provided an oxidative chlorinating reagent, presumably PhI(OH)Cl, which was utilized in situ for reactions with alkenes and sulfides.10DIB was also utilized in halogenation of arenes through grinding with NaCl, NaBr or I2,11 and in the synthesis of oxadiazoles from acylhydrazones.12 Wang and co-workers described aminohalogenation of the alkene moiety of α,β-unsaturated ketones through ball-milling with DIB in the presence of chloramine-T (TsNClNa) or N-bromosuccinimide (NBS, Scheme 1A).13 Furthermore, fused N-heterocycles could be halogenated or thiocyanated by grinding with DIB and the corresponding alkali metal or ammonium salt.14
PIFA is a fluorinated and often more reactive analogue of DIB. A regioselective nitration of anilides was achieved through grinding with PIFA and sodium nitrite, likely through a radical reaction (Scheme 1B).15 A series of papers details oxidative transformations with DIB, PIFA, or HTIB to yield heterocycles by grinding with a pestle and mortar. In this fashion, the oxidative aromatization of 1,4-dihydropyridines, as well as an oxidative cyclization of 2-pyridyl- or 2-quinolylhydrazones, were realized.16 The DIB-mediated cyclization of chalcone oximes gave diarylisoxazoles through grinding with DIB,17 and benzothiazoles were synthesized from 2-aminothiophenols and aldehydes, using DIB or PIFA (Scheme 1C).18
Mal and co-workers reported a DIB-mediated oxidative amidation of aryl aldehydes and benzylamines, with sodium bisulfate added to control the reactivity of this otherwise explosive combination (Scheme 1D).19 Alkynyl- and vinyl selenides were synthesized by grinding DIB with carboxylic acids and diaryldiselenides, resulting in a selenodicarboxylation.20
There are several reports on multicomponent reactions utilizing an iodine(III) reagent under mechanochemical conditions. Wang and co-workers demonstrated an elegant synthesis of dihydropyrroles through a sequential ball-milling reaction of amines, alkyne esters and chalcones, promoted by iodine and DIB.21 Likewise, a multicomponent reaction with dihydrazinopyridazines and aromatic aldehydes yielded bistriazolopyridazines through grinding with DIB.22 Also, the one-pot synthesis of triazoles from hydralazines and aldehydes, through oxidative cyclization of the corresponding hydrazones with DIB or PIFA, could be achieved in a planetary ball-mill.23 The authors highlighted that the sustainability metrics were considerably improved compared to the corresponding solution-phase reaction.
Transition metal-catalyzed reactions with DIB as oxidant are common in solution1a,b and mechanochemical versions of such reactions indicate that inert conditions are not required. A Pd-catalyzed ortho-C–H etherification of aryl oximes with a range of aliphatic alcohols proceeded well in the presence of DIB under ball-milling conditions (Scheme 1E).24 In a DIB-mediated, Rh-catalyzed C–H amination with sulfamides, the catalyst was found to be much more stable than in solution-phase reactions.25 Finally, a recent paper utilized DIB as a Lewis acid in a pestle and mortar-activated Pictet–Spengler-type reactions.26
Diaryliodonium salts (Ar2IX) are frequently used either as efficient arylation reagents27 (see Section 5.1) or as aryl radical initiators in polymerization reactions.1c Recently, Golder and co-workers demonstrated that Ph2IPF6 could be used under mechanoredox catalysis with the piezoelectric catalyst BaTiO3 for free-radical polymerization (FRP) or reversible addition–fragmentation chain transfer (RAFT) polymerization to provide poly(meth)acrylates.28
2.3 Mechanochemical iodine(V) reactions
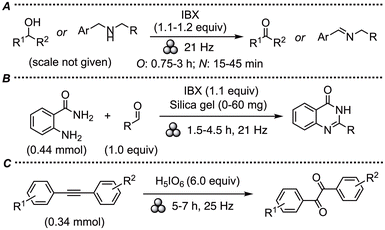
In 2014, Mal and co-workers performed an interesting study of the oxidative power of IBX under ball-milling conditions compared to reported solution-phase reactions.29 Mechanochemical oxidation was efficient in a variety of reactions, including alcohol and amine oxidations to the corresponding aldehydes, ketones, or imines, as well as the one-pot synthesis of benzimidazoles, dithiane deprotection, and α-haloketone synthesis from olefins (Scheme 2A).29 Furthermore, the stoichiometric iodine(III) waste, 2-iodosobenzoic acid (IBA), could be re-oxidized in situ with Oxone to IBX, highlighting that iodine(V) compounds can both be formed and used under ball-milling conditions without explosions.Another study demonstrated that while mechanochemical reactions between anilines and IBX caused explosions, the corresponding 2-aminobenzamides could be reacted with aldehydes and IBX to form quinazolinones (Scheme 2B).30 IBX was also found to be the best oxidant in a mechanochemical synthesis of azo-based polymers.31
2.4 Mechanochemical iodine(VII) reactions
The inorganic iodine(VII) reagent periodic acid (H5IO6) was demonstrated to be an efficient and divergent oxidant for polycyclic aromatic hydrocarbons, providing C–H oxidation under mechanochemical conditions and C–H iodination in solution.32 Furthermore, it was utilized in the oxidation of internal alkynes to provide a large scope of 1,2-diketones under ball-milling conditions (Scheme 2C).33The remainder of this review is focused on highlighting novel mechanochemical transformations with iodine(III) compounds, as well as providing a critical sustainability comparison with the corresponding solution-phase methodologies, where they exist.
3 Ligand exchange reactions
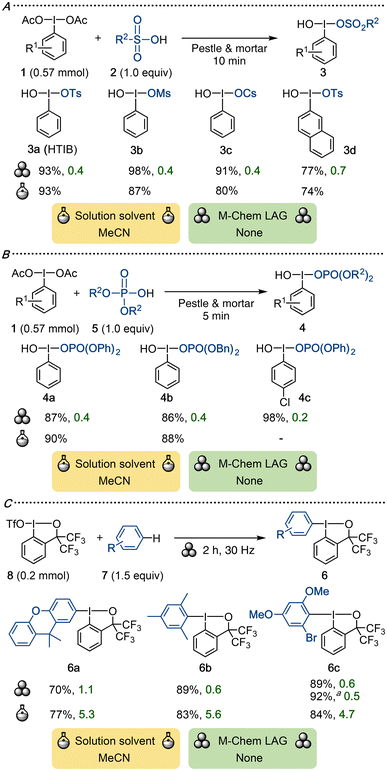
Reactions whereby hypervalent iodine compounds exchange an iodine ligand are called ligand exchanges. Such reactions are generally straightforward with heteroatom ligands, whereas introduction of carbon ligands can be more difficult.1aDIB and HTIB are often used as substrates in ligand exchanges to reach more complex iodine(III) reagents in solution.1a,b In one of the early reports combining mechanochemistry with hypervalent iodine reagents, Yusubov and Wirth described the synthesis of HTIB derivatives by reacting (diacetoxyiodo)arenes 1 and sulfonic acid 2via grinding with a pestle and mortar (Scheme 3A).34 Evaporation of the liberated acetic acid afforded products 3 in good to excellent yields and with excellent waste profiles (E-factors < 1). In solution, the same transformation typically utilizes acetonitrile,1a classified as “Amber” by the GSK solvent guide, and proceeds in similarly high yields.35 As the reported reaction scales are much larger than the mechanochemical ones, E-factor comparisons offer limited insight.5 Eliminating the need for a problematic solvent enables a greener approach to these species, albeit reported here only on a small scale. By contrast, multi-gram syntheses of HTIB are well established in solution and are of commercial relevance.
 | ||
| Scheme 3 Mechanochemical ligand exchange reactions. a2 mmol scale. Ms = methanesulfonyl; Cs = (1R)-10-camphorsulfonyl; Tf = trifluoromethylsulfonyl. | ||
[Hydroxy(phosphoryloxy)iodo]arenes 4 are structurally similar to HTIB, bearing a phosphate ligand in lieu of a tosylate. They were conveniently prepared in a solvent-free manner by grinding equimolar quantities of DIB 1 and phosphate ester 5 in a pestle and mortar. This efficiently provided the desired products 4 in high to excellent yields (Scheme 3B).36 The corresponding reaction can be performed in aqueous acetonitrile with similar efficiency.37 While direct comparison of E-factors is precluded due to the differences in reported reaction scale,5 the mechanochemical reaction has impressively low E-factors, reflecting the minimal waste generated in the absence of solvent.
Yoshikai and co-workers reported a regioselective synthesis of arylbenziodoxoles 6 from arenes 7 and benziodoxole triflate 8 under both solution-phase and ball-milling conditions (Scheme 3C).38 Reactions in acetonitrile for 24 h were performed in a glove-box and applied to a broad scope of arenes in high yields. While the mechanochemical reaction was only demonstrated across four substrates, the authors highlighted the utility of the method through a gram-scale synthesis of 6c in 92% yield. The mechanochemical setup is attractive due to both the shorter reaction times and the excellent E-factors, which are 5–9 times lower than in solution.
4 Oxidative reactions
Iodine(III) compounds with two heteroatom ligands on iodine are widely employed oxidants under transition metal-free or transition metal-catalyzed solution-phase conditions.1a,b Reactions commonly employ DIB (1a), PIFA (9) or HTIB (3a), and sometimes involve direct ligand transfer from iodine(III) to the substrate. Section 4 describes selected mechanochemical methods, including sustainability comparisons with the corresponding solution-phase methods.4.1 Tosyloxylation
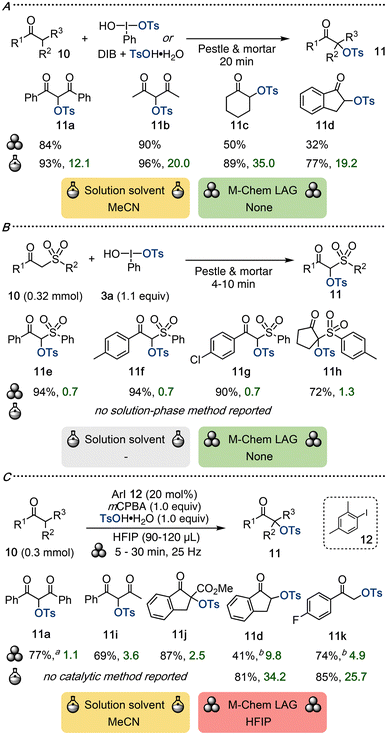
Carbonyl compounds readily undergo α-oxidation upon treatment with various iodine(III) compounds, with α-tosyloxylations using HTIB as the most well-known transformation. This reactivity is well-established under inert conditions in solution, either utilizing stoichiometric amounts of iodine(III) or combining catalytic amounts of iodoarene with a stoichiometric oxidant such as mCPBA.1a,39In 2005, Yusubov and Wirth demonstrated the first mechanochemical α-sulfonyloxylation of carbonyl compounds 10 to yield products 11 (Scheme 4A).34 The reactions were performed in a pestle and mortar, either using isolated HTIB or in situ formed reagent from DIB and tosic acid. Tosyloxylation of dicarbonyl substrates proceeded in good yields, whereas reactions with ketones or transfer of alternative sulfonyl groups gave reduced yields. Reactions performed in acetonitrile required increased temperatures to access such products,40 which is unfeasible through simple grinding, highlighting an inherent limitation of this methodology. Unfortunately, the E-factors could not be calculated for the mechanochemical setup due to a lack of experimental data.
 | ||
| Scheme 4 α-Tosyloxylation of carbonyl compounds. aNo HFIP added. b1.5 equiv. mCPBA and tosic acid, preheated vessel. | ||
Varma and co-workers subsequently extended the mechanochemical tosyloxylation scope to include the facile synthesis of α-tosyloxy β-keto sulfones 11e–11h from HTIB (Scheme 4B).41 Grinding of the substrates in a pestle and mortar produced a scope of nine products in good to excellent yields. A range of electronics, including aryl and cyclic alkyl substituents, were well tolerated. This singular mechanochemical procedure constitutes the only synthesis of α-tosyloxy β-keto sulfones, and the low E-factors demonstrate a minimal environmental footprint.
Recently, our group investigated the efficiency of a range of iodine(III) reactions under ball-milling conditions. Within that study, we carried out tosyloxylations of carbonyl compounds 10 to give products 11 using a catalytic amount of 2,4-dimethyliodobenzene (12), with mCPBA and tosic acid as stoichiometric reagents and HFIP as LAG (Scheme 4C).42 While catalytic iodine(III) reactions are prevalent in solution, this report constitutes the first example of a catalytic system with hypervalent iodine under mechanochemical conditions, highlighting that fast oxidation from iodine(I) to iodine(III) is feasible through ball-milling. The mechanochemical yields of 11d and 11k could be improved by preheating the ball-milling vessel and increasing the reagent amounts to partially overcome the limitations observed with less reactive substrates in the pestle and mortar reaction in Scheme 4A.
Comparison to solution-phase results for the catalytic reaction can only be performed for 11d and 11k, due to differing product scopes. Both products showcase considerably lower E-factors in the mechanochemical protocol, despite lower yields being obtained with that method. While the need for toxic HFIP as LAG lowers the sustainability profile, the minimal volume needed (0.3–0.4 mL mmol−1) should be compared to solution-phase reactions in acetonitrile (10 mL mmol−1), which is also a problematic solvent. Furthermore, the synthesis of 11a proceeded efficiently without use of HFIP.
To conclude, sulfonyloxylations of carbonyl compounds proceed well under both mechanochemical and solution-phase conditions. The catalytic method in Scheme 4C gives similar yields to the stoichiometric iodine(III) method in Scheme 4A, as illustrated by products 11a (77% vs. 84%) and 11c (41% vs. 32%). Though the E-factors of individual tosyloxylations may be lower for reactions using stoichiometric iodine(III), the waste generated during reagent synthesis is not considered in such calculations. In addition to the consistently lower E-factors achieved with mechanochemistry, most examples proceed without need for any toxic additive. However, large-scale applications of the mechanochemical approach remain unreported.
4.2 Oxidative rearrangement
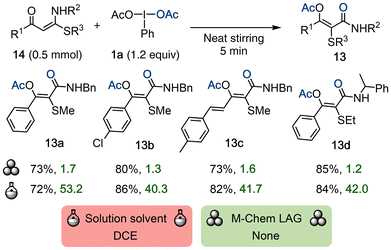
Yu and co-workers reported the synthesis of α,β-unsaturated amides 13 through a DIB-mediated oxidation and alkylthio migration of alkylthio-functionalized enaminones 14 (Scheme 5).43 The reaction was initiated by manually stirring the substrates with a glass rod in a glass vial for 5 minutes, and the resultant liquid was left under ambient conditions for an additional two hours until complete conversion of 14 was observed. This highlights that, while mechanochemical activation of the reaction was necessary, the continued reaction took place without it. The target compounds 13 were isolated in moderate to good yields across ten substrates of varying electronics. The authors also reported a solution-phase version with a substrate scope of 42 amides, which required the use of DCE, designated “Red – highly hazardous” by the GSK solvent guide4 due to its toxicity and probable carcinogenicity. Furthermore, the DIB loading in solution was increased from 1.2 to 1.5 equiv. The solvent-free route is thus more favorable from a green chemistry perspective, both based on E-factor score and solvent toxicity.4.3 Oxidative functionalization of alkenes
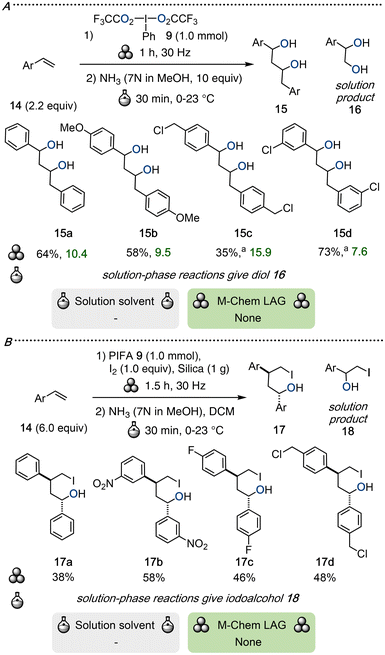
Mechanosynthesis can proceed via pathways distinct from conventional solution-phase methods, giving access to products with different regio- or stereoselectivity. The groups of Yeung and Ke demonstrated that the mechanochemical dioxygenation of styrenes 14 with PIFA yielded dimeric 1,3-diol 15 as the major product and monomeric 1,2-diol 16 as the side-product (Scheme 6A).44 The reaction was conducted in a horizontal ball-mill and the in situ formed diester was subsequently treated with ammonia in methanol to give the corresponding diol 15. In contrast, a one-pot reaction performed in dichloromethane (DCM) under reflux afforded the 1,2-diol 16 as sole product.45 Mechanistic studies suggest that strong mechanical impact forces may induce dimerization of PIFA, thereby reducing the concentration of the active monomeric species and promoting an intramolecular cascade reaction between olefins to furnish the 1,3-disubstituted product. In solution, dissociated trifluoroacetate anions may instead selectively favor a direct substitution mechanism, thus following the conventional dioxygenation pathway to selectively give 1,2-diols.44The calculated E-factors are relatively poor, which stem from side-product formation and the need for treatment with ammonia in methanol. Nonetheless, this method provides a novel synthetic route to 1,3-diols through the direct dioxygenation of styrenes, whereas conventional solution-phase routes to 1,3-diols require several steps.46
A mechanochemical 1,4-iodohydroxylation strategy was also developed by treating styrenes 14 with PIFA and molecular iodine under ball-milling conditions with silica as solid-assisted grinding agent (SAG).44 Quenching with ammonia in methanol furnished the corresponding 1,4-iodoalcohols 17 as single diastereomers in moderate yields (Scheme 6B). The same mechanistic rationale as above was employed to explain the observed selectivity in favor of 17 over the solution-phase product 18. Though insufficient details are provided to accurately calculate E-factors, this protocol remains the only reported route to access these novel products.
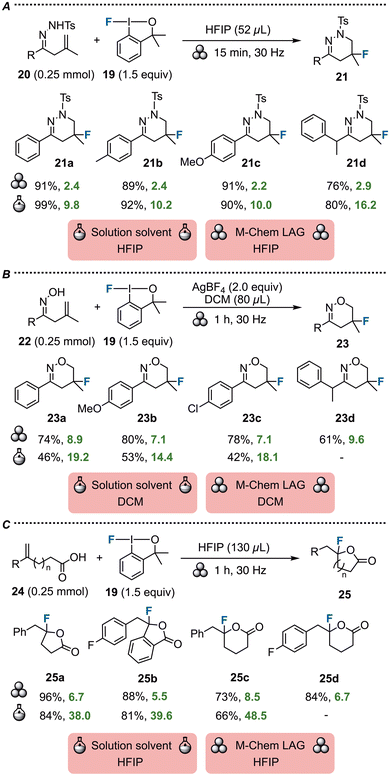
Hypervalent iodine reagents are frequently employed in fluorinations to reach pharmaceutically relevant characteristics, such as increased metabolic stability and potency in medicines. In this vein, the groups of Stuart and Browne reported a mechanochemical fluorocyclization of functionalized alkenes via ball-milling.47 Fluoroiodoxole 19 was employed as a fluorine source in reactions with alkenyl hydrazones 20 to yield 5-fluoro-tetrahydropyridazines 21 in high yields when activated by the use of HFIP as LAG (Scheme 7A). While the reaction could also be performed in similar yields with HFIP as solvent under inert conditions, the mechanochemical protocol affords much-improved E-factors through an 8-fold reduction of toxic HFIP.
In a similar fashion, treatment of alkenyl oximes 22 with fluoroiodoxole 19 produced 5-fluorodihydrooxazines 23 with DCM as LAG (Scheme 7B).47 Notably, mechanochemical activation afforded higher yields than conventional synthesis in DCM due to suppressed side-product formation.47 The mechanochemical E-factors are comparatively high due to the use of excess AgBF4 as reaction additive. However, the use of DCM is markedly reduced, along with the environmental footprint of the reaction.
Finally, the authors reported an intramolecular fluorolactonization protocol under ball-milling conditions, using HFIP as LAG (Scheme 7C).47 Fluoroiodoxole 19 was applied to unsaturated carboxylic acids 24 to furnish γ- and δ-lactones 25.
Compared to their previously reported solution-based method in HFIP solvent,48 higher yields were obtained. Furthermore, the volume of toxic HFIP was reduced by a factor of 6, thereby leading to much-improved E-factor scores indicative of a more sustainable process by mechanochemistry.
4.4 Oxidative sulfenylation
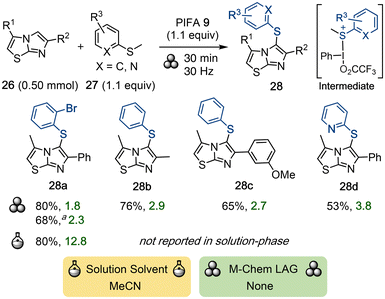
Sulfenylation of imidazothiazoles and related scaffolds is frequently copper-catalyzed and often involves harmful solvents such as DMF, elevated temperatures, and long reaction times.49 In 2025, Hamze and co-workers developed a PIFA-mediated mechanochemical route towards the regioselective C5-sulfenylation of imidazothiazoles.50 For this purpose, imidazo[2,1-b]thiazoles 26 were ground with aryl methyl sulfides 27 and PIFA in a vibrational ball-mill to produce a broad scope of arylthiolated products 28 (Scheme 8). The procedure was also upscaled to afford 1.36 g of the desired product 28a after recrystallization. A mechanistic investigation indicated an ionic pathway with a highly electrophilic iodine(III)–S(Me)Ar intermediate that is attacked by the imidazothiazole, whereas related methods proposed radical mechanisms.51 The authors compared the environmental impact of their method to performing the synthesis of 28a in acetonitrile at 50 °C. The E-factor of the solution-based reaction is 100 times higher due to the need for excess reagents, solvent waste, and an aqueous/DCM workup to quench residual PIFA.4.5 Reactions with iodonium sulfonimides
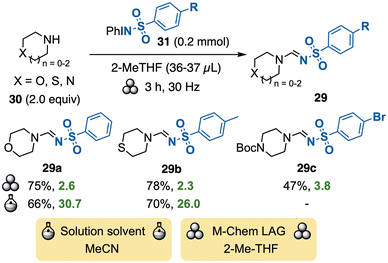
Iodonium sulfonimides (PhINSO2R) are iodine(III) reagents commonly used in nitrogen-transfer reactions, e.g., imination or aziridination reactions.1a They were recently applied in a metal-free imination to provide sulfondiimines, where mechanochemical conditions proved less efficient than reactions in MeCN.52 The same year, Sen & co-workers reported a synthesis of N-sulfonyl amidines 29 by ball-milling cyclic amines 30 with the iodine(III) reagents N-sulfonyl iminoiodinanes 31 and 2-methyl-THF as LAG (Scheme 9).53 The substrate scope included several types of heterocyclic amines, giving C–N coupled products 29 in moderate to good yields. The proposed mechanism is complex and involves a ring contraction step to provide the added carbon in 29, motivating the need for 2 equiv. of amines 30. The method can be compared with the previously reported DIB-mediated transformation in acetonitrile, which has a large scope and similar yields.54 While the E-factors of the mechanochemical reactions are considerably lower, they do not show the waste formed in the synthesis of iminoiodinanes 31 from sulfonamides, DIB, and KOH in methanol, which is not required in the solution-phase reaction. A fair comparison would be based upon the entire two-step process, but the E-factors for the synthesis of iminoiodinanes 31 cannot be calculated due to a lack of experimental details.5 Transfer of carbon ligands
Iodine(III) reagents with two carbon ligands are efficient reagents for the transfer of a carbon ligand to a nucleophile under metal-free or transition metal-catalyzed conditions. The most common reagents are diaryliodonium salts, alkynyliodonium salts, and vinyliodonium salts, as well as the cyclic reagents ethynylbenziodoxolones (EBX), vinylbenziodoxolones (VBX) and trifluoromethylbenziodoxol(on)es (called Togni's reagents).1a5.1 Arylations
Diaryliodonium salts (Ar2IX) are efficient electrophilic arylation reagents under both transition metal-catalyzed and metal-free conditions, and have found broad applications in solution-phase reactions.27 In 2020, Li and co-workers disclosed the first mechanochemical reactions with Ar2IX, in which arylation of anilines 30 with diaryliodonium triflates 31-OTf was achieved by ball-milling either under copper-catalyzed or metal-free conditions to provide diarylamines 32 (Scheme 10A, methods A & B).55 Interestingly, both reaction setups utilized silica gel as SAG, and the metal-free method also employed water as LAG. Method A delivered slightly higher yields than method B and could also be applied for the arylation of heterocyclic amines in moderate yields. Aliphatic amines proved to be poor substrates with both methods. Method A is also superior in terms of E-factors, as method B requires excess reagent usage and LAG.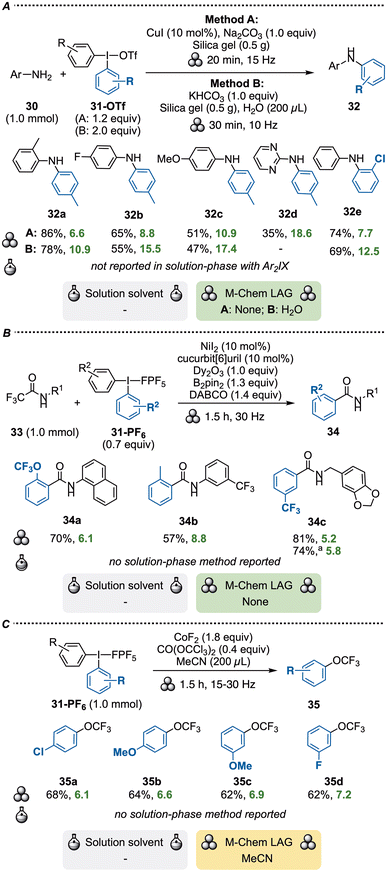 | ||
| Scheme 10 Arylations with diaryliodonium salts. a10 mmol scale. DABCO = (1,4-diazabicyclo[2.2.2]octane). | ||
While there are reports with limited scope for metal-free arylation of anilines with Ar2IX in DMF or toluene,56 as well as a Cu-catalyzed method in DCM/DMSO,57 those products do not overlap with Li's product scope,55 making a direct comparison unfeasible.
The groups of Iaroshenko and Mkrtchyan reported the mechanochemical activation of trifluoroacetamides 33 to achieve defluorinative arylation to amides 34 (Scheme 10B).58 Several aryl sources were suitable for this transformation under ball-milling conditions, ranging from boronic acids and siloxanes to dimethylsulfonium salts and Ar2IPF631-PF6. The arylation required nickel-catalysis combined with stoichiometric amounts of lanthanide additive and B2pin2 to furnish the products in moderate to high yields. A range of electronics and sterics, as well as aliphatic amides, were tolerated. A large-scale reaction was also demonstrated, providing 2.39 g of 34c. Considering the many additives required for the reaction, the associated E-factors are quite low. We did not find any equivalent reactions in solution, and the authors state that the corresponding solution-phase reactions failed to produce the target product. On the other hand, conventional amide coupling methodologies to amides 34 may be more accessible than the use of substrates 33.
Iaroshenko and co-workers subsequently developed a trifluoromethoxylation protocol utilizing a combination of CoF2 and triphosgene to produce an OCF3 source in situ under ball-milling conditions. This system was applied to a number of different aryl sources, including Ar2IPF631-PF6 to produce 4 examples of products 35 (Scheme 10C).59 While there is no solution-phase methodology for this reaction with hypervalent iodine reagents to allow for comparison, there is a plethora of methodologies to conduct this transformation.60
In 2019, Ito and co-workers pioneered the use of piezoelectric materials to carry out mechanoredox methodologies with redox-sensitive compounds, where aryldiazonium salts were utilized as an aryl radical source for arylation and borylation reactions.61 In this sense, mechanical force from milling causes a polarity within the material, similar to an excited state of a photo-excitable material.6a Li and co-workers subsequently showed that diaryliodonium triflates 31-OTf could be used as an alternative aryl source in C–H arylation of quinoxalines 36 through a mechanoredox reaction with the piezoelectric material BaTiO3 (Scheme 11A).62 Ball-milling efficiently delivered products 37 in moderate to good yields.
The authors extended this strategy towards quinoxalinones 38, which delivered the corresponding products 39 in similar yields (Scheme 11B).62 The E-factors for these reactions are higher than for previous arylation examples due to the required excess of piezoelectric material and triethylamine. However, the BaTiO3 could be reused several times without marked loss of activity. As there is no solution-phase method to generate the targeted products, a comparison cannot be made.
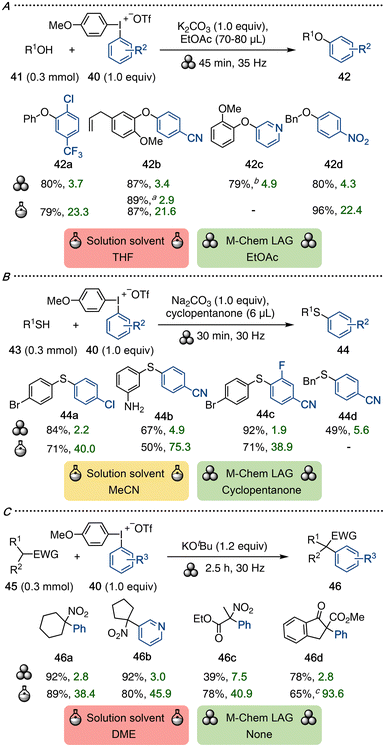
Our group recently reported mechanochemical arylations of a range of O-, S- and C-nucleophiles (Scheme 12).42 Using known solution-phase methods as a starting point,63 we investigated the reactivity under ball-milling conditions in the presence of LAGs that were selected based on the GSK solvent guide. Unsymmetric diaryliodonium salts (Ar1Ar2IOTf, 40) were utilized to chemoselectively transfer the more electron-poor aryl moiety, giving iodoanisole as waste.64
A LAG was imperative for arylations of phenols and alcohols 41, and ethyl acetate was hence used as a LAG in the synthesis of aryl ethers 42 (Scheme 12A). The scope included a large-scale synthesis of 42b in 89% yield. Arylation of S-nucleophiles 43 to products 44 required minimal amounts of a LAG, and the green solvent cyclopentanone was selected for this purpose (Scheme 12B). C-Arylations of nitroalkenes and other alkanes with EWG (44) proceeded well without a LAG (Scheme 12C).
A range of electronics and sterics were tolerated on the explored nucleophiles. Aryl groups decorated with electron-withdrawing groups (EWGs) and/or ortho-substituents were efficiently transferred, whereas electron-rich aryl groups were more challenging.42 The article also contained a benchmarking section where reported solution-phase methodology under inert conditions63 was utilized to synthesize selected products in the mechanochemical scope. Interestingly, yields were similar under standard reaction conditions, whereas more concentrated or neat reactions gave considerably lower yields, indicating the importance of mechanochemical activation.42
The sustainability analysis highlights that the problematic solvents THF, dimethoxyethane (DME) and MeCN were avoided through the ball-milling approach, which instead used small amounts of green solvents as LAGs. Furthermore, the mechanochemical E-factors were consistently lower, ranging from a 5- to 20-fold reduction in waste, signifying a much-improved environmental footprint.
5.2 Alkynylations
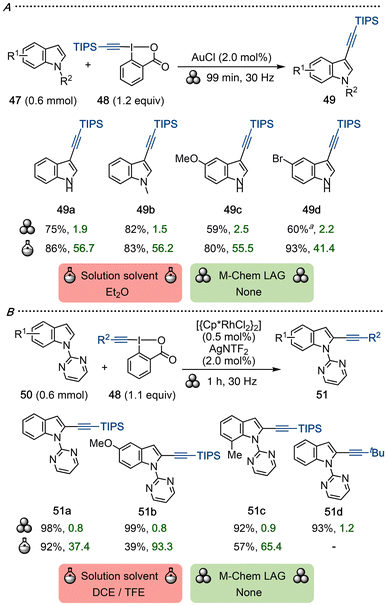
Ethynylbenziodoxolones (EBX) are commonly used as electrophilic alkynylation reagents, often allowing for more chemoselective and predictive reactions than alkynyl(aryl)iodo-nium salts due to the stabilizing benziodoxolone core.1a Waser and co-workers have popularized EBX chemistry under both transition metal-catalyzed and metal-free conditions, including the gold-catalyzed C–H alkynylation of indole derivatives.65 The C2-selective alkynylation of indoles with EBX has been reported under Rh- or Co-catalyzed inert conditions.66Bolm and co-workers developed mechanochemical versions of these transformations using two different transition metal-catalyzed systems to achieve C3- or C2-selective alkynylation under ball-milling conditions.67 Following Waser's precedent in solution-phase,65a indoles 47 were regioselectively alkynylated with TIPS-EBX 48 to yield products 49 using AuCl as catalyst (Scheme 13A). The reaction proceeded well with both NH- and N-alkyl indoles, and a C3-methylated substrate was instead alkynylated in the C2 position, following Waser's observations.
To reach a general method for C2 alkynylation, a Rh-catalyzed method was developed for N-pyrimidyl indoles 50 (Scheme 13B). The reaction occurred within one hour, and the scope of 51 includes a variety of substituted indoles, although EWG-decorated indoles were formed in lower yield. Silylated EBX as well as alkyl-EBX reagents were tolerated in the reaction.
While solution-based methods work well for both types of alkynylation,65a,66 the catalyst loadings and the reaction times were reduced in the mechanochemical setup. Furthermore, it avoids the handling of the hazardous solvents diethyl ether, DCE and TFE, and does not require inert conditions. The calculated E-factors for the ball-milling method are excellent and dramatically improved compared to the solution-phase reactions, signifying a greener and more resource-efficient process.
5.3 Synthesis of alkenes
Vinylbenziodoxolones (VBX) and vinylbenziodoxoles (VBO) were recently demonstrated as electrophilic vinylation reagents under both transition metal-free and transition metal-catalyzed conditions.68 They offer more tunable reactivity than acyclic vinyl(aryl)iodonium salts, and the three reagent types can be screened to fine-tune the reactivity in vinylations.We investigated the vinylation of S- and C-nucleophiles under ball-milling conditions, and found that VBX 52 was suitable for the S-vinylation of thiols to give products 54 (Scheme 14A). Vinyliodonium salts 53 were preferred in the C-vinylation of nitroalkenes and β-ketoesters to yield products 55, here exemplified with the synthesis of 55a on both small and large scales (Scheme 14B).42
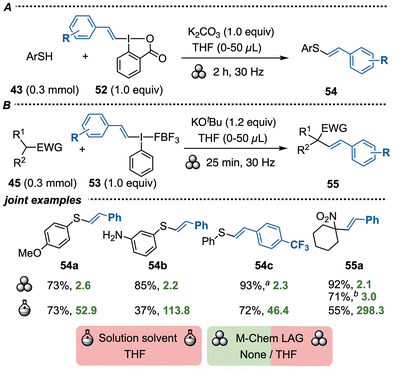 | ||
| Scheme 14 Transition metal-free vinylations with VBX. a0.2 mmol scale with LAG. b3.0 mmol scale, milling time: 15 + 10 min. | ||
The corresponding solution-phase vinylations require inert conditions in anhydrous THF and give similar or lower yields.68a,69 The E-factors in solution are very high, which further highlights the benefits of the mechanochemical transformation.
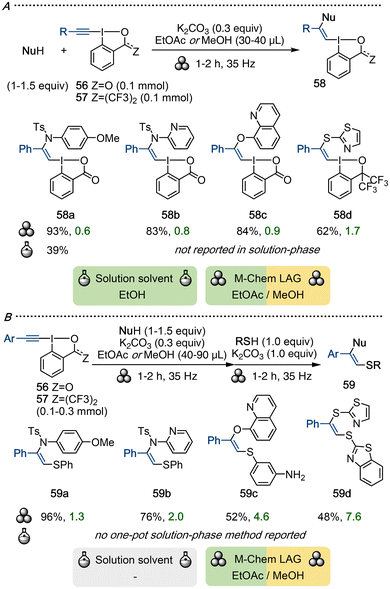
VBX compounds with a β-heteroatom substituent (X-VBX) are formed through β-addition of various heteroatom nucleophiles to EBX. This transformation was demonstrated in solution-phase by Waser and co-workers,70 and Yoshikai's group developed the corresponding transformation from ethynylbenziodoxoles (EBO) to yield X-VBO.71 This type of reactivity was unexplored under mechanochemical conditions, which inspired our group to embark upon the investigation depicted in Scheme 15A.72 A wide variety of N- and O-nucleophiles underwent efficient addition to EBX 56 under ball-milling conditions when ethyl acetate was utilized as LAG. Reactions with electron-rich nucleophiles proceeded smoothly in a 1.5 mL vessel, whereas weaker nucleophiles required a 5 mL vessel with a larger ball to ensure sufficient reactivity. S-Nucleophiles were too reactive in reactions with EBX, resulting in byproduct formation. Reactions with EBO 57 were, on the other hand, selective for mono-addition, and yielded a range of S-VBO products (exemplified by 58d) with methanol as LAG.
A one-pot reaction combining the synthesis of X-VBX/VBO 58 with a subsequent S-vinylation to reach bis(hetero)-substituted Z-alkenes 59 was subsequently developed (Scheme 15B).72 A large scope was demonstrated by variation of the first nucleophile and the thiol, and moderate to excellent yields of 59 were obtained within 2–4 hours of ball-milling.
Comparing the first method with solution-phase methodology69 highlights that improved yields and E-factors were obtained under mechanochemical conditions, while the major influence on the solution-phase E-factor originates from the use of sustainable solvent ethanol. The one-pot reaction for the synthesis of alkenes 59 was also evaluated in the solution-phase, but failed under a range of conditions, which is likely due to the mismatch of solvents reported for the separate steps (EtOH vs. dry, degassed THF). To the contrary, the ball-milling method is fast, clean, and high yielding, illustrating the utility of mechanochemistry in organic synthesis.
6 Conclusions and outlook
The combination of mechanochemistry with hypervalent iodine chemistry has been investigated in a wide range of reactions. In this critical review, we highlighted several mechanochemical iodine(III) reactions where solution-phase reactions either fail or give different types of products. We also compared the reactivity between mechanochemical and solution-phase reactions when possible, and evaluated the methods from a sustainability perspective. We found that the calculated E-factors for solution-phase reactions were often dominated by solvent contributions, and moderate to great improvements in the waste profiles were observed under mechanochemical conditions. Furthermore, mechanochemical reactions avoid a range of hazardous reaction solvents and inert conditions that are required in the solution phase.Mechanochemical activation of hypervalent iodine compounds holds the potential to open a new and more sustainable era of hypervalent iodine research, with novel reactivity to be discovered. Challenges of this growing research field include that the high energy environment can lead to leaching of trace metals from the reaction vessels, requiring careful control over reaction materials.73 While transfer of the crude reaction from the reaction vessel can be difficult and require solvent in small scale reactions, such issues can be better handled at large scale. The development of large-scale or industrial processes could also allow alternative workup and purification procedures, further amplifying sustainability benefits. We have highlighted a handful of up-scaled mechanochemical reactions in the review, but further investigations are needed, especially considering that up-scaling of hypervalent iodine reactions needs to be done with care both in solution and in the solid phase.74
To conclude, we believe that the demonstrated possibilities to reach different reaction pathways, or to develop one-pot reactions for systems that cannot be combined in solution, can be key to new discoveries in hypervalent iodine chemistry as well as in other parts of organic synthesis and catalysis.
Author contributions
Conceptualization: SD, BO; investigation: PÖ, SD, BO; visualization: PÖ, SD, BO; project administration: BO; funding acquisition: BO; writing – original draft: PÖ, SD, BO; writing – review & editing: PÖ, BO.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: method description and E-factor calculations. See DOI: https://doi.org/10.1039/d6gc00367b.Acknowledgements
This work was generously funded by Olle Engkvist Foundation (224-0058) and the Swedish Research Council (2023-04519).References
- (a) A. Yoshimura and V. V. Zhdankin, Chem. Rev., 2024, 124, 11108 CrossRef; (b) The Chemistry of Hypervalent Halogen Compounds, ed. B. Olofsson, I. Marek and Z. Rappoport, Wiley, 2019 Search PubMed; (c) A. D. Asandei, Chem. Rev., 2016, 116, 2244 CrossRef PubMed.
- (a) K. Ishihara and K. Muniz, in Iodine Catalysis in Organic Synthesis, Wiley-VCH, Weinheim, Germany, 2022 CrossRef; (b) F. V. Singh, S. E. Shetgaonkar, M. Krishnan and T. Wirth, Chem. Soc. Rev., 2022, 51, 8102 RSC.
- V. V. Zhdankin and J. D. Protasiewicz, Coord. Chem. Rev., 2014, 275, 54 CrossRef.
- (a) C. M. Alder, J. D. Hayler, R. K. Henderson, A. M. Redman, L. Shukla, L. E. Shuster and H. F. Sneddon, Green Chem., 2016, 18, 3879 RSC; (b) D. Prat, A. Wells, J. Hayler, H. Sneddon, C. R. McElroy, S. Abou-Shehada and P. J. Dunn, Green Chem., 2016, 18, 288 RSC.
- See the SI for details.
- (a) J. L. Howard, Q. Cao and D. L. Browne, Chem. Sci., 2018, 9, 3080 RSC; (b) J.-L. Do and T. Friščić, ACS Cent. Sci., 2017, 3, 13 CrossRef; (c) V. Martinez, T. Stolar, B. Karadeniz, I. Brekalo and K. Užarević, Nat. Rev. Chem., 2023, 7, 51 CrossRef; (d) J. G. Hernández and C. Bolm, J. Org. Chem., 2017, 82, 4007 CrossRef.
- O. Prakash and V. Sharma, Indian J. Chem., 1999, 38B, 229 CAS.
- R. A. Sheldon, Green Chem., 2023, 25, 1704 RSC.
- A. Y. Koposov, B. C. Netzel, M. S. Yusubov, V. N. Nemykin, A. Y. Nazarenko and V. V. Zhdankin, Eur. J. Org. Chem., 2007, 4475 CrossRef CAS.
- (a) H. Sohmiya, T. Kimura, P. Bauchat, M. Fujita and T. Ando, Chem. Lett., 1991, 20, 1391 CrossRef; (b) H. Sohmiya, T. Kimura, M. Fujita and T. Ando, Chem. Lett., 1992, 21, 891 CrossRef; (c) H. Sohmiya, T. Kimura, M. Fujita and T. Ando, Tetrahedron, 1998, 54, 13737 CrossRef CAS.
- N. N. Karade, G. B. Tiwari, D. B. Huple and T. A. J. Siddiqui, J. Chem. Res., 2006, 2006, 366 CrossRef.
- V. S. Rao and K. V. G. Chandra Sekhar, Synth. Commun., 2004, 34, 2153 CrossRef CAS.
- (a) G.-W. Wang and X.-L. Wu, Adv. Synth. Catal., 2007, 349, 1977 CrossRef CAS; (b) X.-L. Wu, J.-J. Xia and G.-W. Wang, Org. Biomol. Chem., 2008, 6, 548 RSC.
- D. R. Indukuri, G. R. Potuganti and M. Alla, Synlett, 2019, 1573 CAS.
- D. An, W. Song, Z. Peng, Y. Zhang and W. Dong, ChemistrySelect, 2018, 3, 12946 CrossRef CAS.
- (a) P. Kumar, Chin. J. Chem., 2009, 27, 1487 CrossRef CAS; (b) P. Kumar, Chem. Heterocycl. Compd., 2012, 47, 1237 CrossRef CAS.
- K. V. G. Chandra Sekhar, T. V. N. V. T. Sasank, H. N. Nagesh, N. Suresh, K. M. Naidu and A. Suresh, Chin. Chem. Lett., 2013, 24, 1045 CrossRef.
- P. Kumar, Meenakshi, S. Kumar, A. Kumar, K. Hussain and S. Kumar, J. Heterocycl. Chem., 2012, 49, 1243 CrossRef.
- T. Kumar Achar and P. Mal, Adv. Synth. Catal., 2015, 357, 3977 CrossRef.
- J. P. Das, U. K. Roy and S. Roy, Organometallics, 2005, 24, 6136 CrossRef.
- H. Xu, H.-W. Liu, K. Chen and G.-W. Wang, J. Org. Chem., 2018, 83, 6035 CrossRef PubMed.
- R. Aggarwal, Mamta and G. Sumran, ARKIVOC, 2019, 2019, 190 Search PubMed.
- L. Gonnet, C. André-Barrès, B. Guidetti, A. Chamayou, C. Menendez, M. Baron, R. Calvet and M. Baltas, ACS Sustainable Chem. Eng., 2020, 8, 3114 Search PubMed.
- K. Zhou, H.-Y. Hao, Y.-J. Mao, Q.-Z. Wu, L. Chen, S. Wang, W. Jin, Z.-Y. Xu, S.-J. Lou and D.-Q. Xu, ACS Sustainable Chem. Eng., 2021, 9, 4433 Search PubMed.
- X. Lu, Y. Bai, J. Qin, N. Wang, Y. Wu and F. Zhong, ACS Sustainable Chem. Eng., 2021, 9, 1684 Search PubMed.
- M. Panda, A. Bera, D. Dandapat, P. Barik, S. Dhara, M. Ikbal, K. Pramanik, S. Pramanik and S. Samanta, Asian J. Org. Chem., 2025, 14, e00118 CrossRef.
- (a) E. A. Merritt and B. Olofsson, Angew. Chem., Int. Ed., 2009, 48, 9052 CrossRef PubMed; (b) P. Villo and B. Olofsson, in Patai's Chemistry of Functional Groups: The Chemistry of Hypervalent Halogen Compounds, ed. B. Olofsson, I. Marek and Z. Rappoport, 2019, p. 461 Search PubMed.
- (a) P. Chakma, S. M. Zeitler, F. Baum, J. Yu, W. Shindy, L. D. Pozzo and M. R. Golder, Angew. Chem., Int. Ed., 2023, 62, e202215733 CrossRef PubMed; (b) S. M. Zeitler, P. Chakma and M. R. Golder, Chem. Sci., 2022, 13, 4131 RSC.
- T. K. Achar, S. Maiti and P. Mal, RSC Adv., 2014, 4, 12834 RSC.
- M. T. Alam, S. Maiti and P. Mal, Beilstein J. Org. Chem., 2018, 14, 2396 CrossRef.
- S. Hou, J. Xu, J. Wang, H. Wang and P. Zhang, Chem. – Eur. J., 2024, 30, e202303126 CrossRef.
- H. Luo, F.-Z. Liu, Y. Liu, Z. Chu and K. Yan, J. Am. Chem. Soc., 2023, 145, 15118 CrossRef PubMed.
- P. Nayek and P. Mal, Chem. – Eur. J., 2024, 30, e202401027 CrossRef PubMed.
- M. S. Yusubov and T. Wirth, Org. Lett., 2005, 7, 519 CrossRef PubMed.
- G. F. Koser and R. H. Wettach, J. Org. Chem., 1977, 42, 1476 CrossRef.
- M. Zhu, C. G. Cai, W. Ke and J. Shao, Synth. Commun., 2010, 40, 1371 CrossRef.
- G. F. Koser, J. S. Lodaya, D. G. Ray and P. B. Kokil, J. Am. Chem. Soc., 1988, 110, 2987 CrossRef.
- W. Ding, C. Wang, J. R. Tan, C. C. Ho, F. León, F. García and N. Yoshikai, Chem. Sci., 2020, 11, 7356 RSC.
- G. F. Koser, Aldrichimica Acta, 2001, 34, 89 Search PubMed.
- (a) M. S. Yusubov, T. V. Funk, R. Y. Yusubova, G. Zholobova, A. Kirschning, J. Y. Park and K.-W. Chi, Synth. Commun., 2009, 39, 3772 CrossRef; (b) S. T. Handy and M. Okello, Synlett, 2002, 489 CrossRef; (c) J. S. Lodaya and G. F. Koser, J. Org. Chem., 1988, 53, 210 CrossRef.
- D. Kumar, M. S. Sundaree, G. Patel, V. S. Rao and R. S. Varma, Tetrahedron Lett., 2006, 47, 8239 CrossRef.
- S. Doobary, M. M. de Vries Ibáñez and B. Olofsson, Green Chem., 2024, 26, 11518 RSC.
- Z. Liu, F. Huang, P. Wu, Q. Wang and Z. Yu, J. Org. Chem., 2018, 83, 5731 CrossRef PubMed.
- L. Pan, L. Zheng, Y. Chen, Z. Ke and Y.-Y. Yeung, Angew. Chem., Int. Ed., 2022, 61, e202207926 CrossRef.
- M. Çelik, C. Alp, B. Coşkun, M. S. Gültekin and M. Balci, Tetrahedron Lett., 2006, 47, 3659 CrossRef.
- J. Cai, Y. Wen, W. Sheng, X. Huang, Y. Zheng, C. Song and J. Li, Green Chem., 2023, 25, 6618 RSC.
- W. Riley, A. C. Jones, K. Singh, D. L. Browne and A. M. Stuart, Chem. Commun., 2021, 57, 7406 RSC.
- H. K. Minhas, W. Riley, A. M. Stuart and M. Urbonaite, Org. Biomol. Chem., 2018, 16, 7170 RSC.
- I. P. Beletskaya and V. P. Ananikov, Chem. Rev., 2022, 122, 16110 CrossRef.
- X. Liu, V. Dorokhov, O. Provot, C. Tran, P. Retailleau, J.-F. Soulé and A. Hamze, ChemSusChem, 2025, 18, e202500320 CrossRef.
- M. Liu, K. Yan, J. Wen, N. Zhang, X. Chen, X. Li and X. Wang, Asian J. Org. Chem., 2022, 11, e202200381 CrossRef.
- M. T. Passia, N. Bormann, J. S. Ward, K. Rissanen and C. Bolm, Angew. Chem., Int. Ed., 2023, 62, e202305703 CrossRef.
- S. Guha, S. Maheshwari, M. K. Ravva, J. M. Jacob, S. Yadav and S. Sen, Asian J. Org. Chem., 2023, 12, e202300348 CrossRef.
- C. S. Nishad, K. K. Haldar and B. Banerjee, J. Org. Chem., 2022, 87, 11644 CrossRef PubMed.
- J. Jiang and J. Li, ChemistrySelect, 2020, 5, 542 CrossRef.
- (a) M. A. Carroll and R. A. Wood, Tetrahedron, 2007, 63, 11349 CrossRef; (b) N. Purkait, G. Kervefors, E. Linde and B. Olofsson, Angew. Chem., Int. Ed., 2018, 57, 11427 CrossRef PubMed.
- I. Sokolovs, D. Lubriks and E. Suna, J. Am. Chem. Soc., 2014, 136, 6920 CrossRef.
- S. Mkrtchyan, M. Shkoor, M. Phanindrudu, M. Medved′, O. Sevastyanova and V. O. Iaroshenko, J. Org. Chem., 2023, 88, 863 CrossRef PubMed.
- S. Mkrtchyan, V. B. Purohit, J. Zapletal, O. Shalimov, J. Nociarová, G. Addová, J. Filo, M. G. Garcia, E. Kupcová, B. Benická and V. O. Iaroshenko, Cell Rep. Phys. Sci., 2024, 5, 102118 CrossRef CAS.
- T. Liang, C. N. Neumann and T. Ritter, Angew. Chem., Int. Ed., 2013, 52, 8214 CrossRef CAS PubMed.
- K. Kubota, Y. Pang, A. Miura and H. Ito, Science, 2019, 366, 1500 CrossRef CAS.
- J. Jiang, S. Song, J. Guo, J. Zhou and J. Li, Tetrahedron Lett., 2022, 98, 153820 CrossRef CAS.
- (a) N. Jalalian, E. E. Ishikawa, L. F. Silva and B. Olofsson, Org. Lett., 2011, 13, 1552 CrossRef CAS; (b) S. Sarkar, N. Wojciechowska, A. A. Rajkiewicz and M. Kalek, Eur. J. Org. Chem., 2022, e202101408 CrossRef CAS; (c) C. Dey, E. Lindstedt and B. Olofsson, Org. Lett., 2015, 17, 4554 CrossRef CAS PubMed.
- J. Malmgren, S. Santoro, N. Jalalian, F. Himo and B. Olofsson, Chem. – Eur. J., 2013, 19, 10334 CrossRef CAS.
- (a) J. P. Brand, J. Charpentier and J. Waser, Angew. Chem., Int. Ed., 2009, 48, 9346 CrossRef CAS; (b) E. Le Du and J. Waser, Chem. Commun., 2023, 59, 1589 RSC.
- (a) F. Xie, Z. Qi, S. Yu and X. Li, J. Am. Chem. Soc., 2014, 136, 4780 CrossRef CAS PubMed; (b) Z.-Z. Zhang, B. Liu, C.-Y. Wang and B.-F. Shi, Org. Lett., 2015, 17, 4094 CrossRef CAS.
- G. N. Hermann, M. T. Unruh, S.-H. Jung, M. Krings and C. Bolm, Angew. Chem., Int. Ed., 2018, 57, 10723 CrossRef CAS.
- (a) E. Stridfeldt, A. Seemann, M. J. Bouma, C. Dey, A. Ertan and B. Olofsson, Chem. – Eur. J., 2016, 22, 16066 CrossRef CAS PubMed; (b) N. Declas, G. Pisella and J. Waser, Helv. Chim. Acta, 2020, 103, e2000191 CrossRef.
- L. Castoldi, E. M. Di Tommaso, M. Reitti, B. Gräfen and B. Olofsson, Angew. Chem., Int. Ed., 2020, 59, 15512 CrossRef PubMed.
- (a) P. Caramenti, N. Declas, R. Tessier, M. D. Wodrich and J. Waser, Chem. Sci., 2019, 10, 3223 RSC; (b) T. Ura, D. Shimbo, M. Yudasaka, N. Tada and A. Itoh, Chem. – Asian J., 2020, 15, 4000 CrossRef PubMed; (c) J. Li, C. Zhou, L.-M. Chen, X.-Y. Chen, X.-Q. Guo and T.-R. Kang, Synlett, 2024, 307 Search PubMed.
- (a) J. Wu, X. Deng, H. Hirao and N. Yoshikai, J. Am. Chem. Soc., 2016, 138, 9105 CrossRef; (b) J. Wu, K. Xu, H. Hirao and N. Yoshikai, Chem. – Eur. J., 2017, 23, 1521 CrossRef PubMed.
- S. Doobary, J. Braunreuther, A. K. Inge and B. Olofsson, Angew. Chem., Int. Ed., 2026, 65, e19049 CrossRef PubMed.
- F. Basoccu, P. Caboni and A. Porcheddu, ChemSusChem, 2025, 18, e202402547 CrossRef PubMed.
- R. Obermüller, H. Tobisch, L. Stockhammer and M. Waser, Org. Process Res. Dev., 2024, 28, 3735 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |