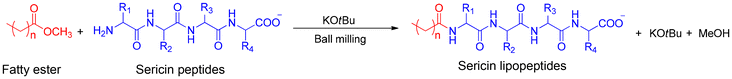Open Access Article
Open Access ArticleInnovative mechanochemical synthesis of lipopeptide surfactants through direct amidation of fatty esters: application to silk-derived amphiphilic lipopeptides
Lara
Wehbe
,
Erwann
Guénin†
 and
Alla
Nesterenko†
and
Alla
Nesterenko†
 *
*
Université de technologie de Compiègne, ESCOM, TIMR (Integrated Transformations of Renewable Matter), Centre de recherche Royallieu, CS 60 319, 60 203 Compiègne Cedex, France. E-mail: alla.nesterenko@utc.fr
First published on 20th March 2026
Abstract
The present work describes the development of a novel methodology for producing lipopeptides from fatty acid methyl esters and amino acids or peptides using mechanochemistry in basic conditions, and its use to prepare biobased surfactants from silk sericin protein. The experimental protocol was optimized with variable molar ratios and reaction times, on model amino acids, glycine, leucine, and serine. Lipoamino acids resulted in a significantly improved conversion rate, up to 99%, after an increase in the base and fatty ester ratio. The structures of the synthesized molecules were characterized with NMR and FTIR. The optimal experimental conditions for high conversion with no excess fatty acid ester were applied to produce sericin lipopeptides with three different lengths of hydrophobic part: 10, 12 and 14 carbons. The acylation rates of sericin peptides of 40–49% were obtained depending on fatty acid size. The surface activity of the silk lipopeptides produced was evaluated and showed a significant reduction in surface tension until ∼24 mN m−1. The results of this study provide an efficient new strategy for the amidation of amino acids and peptides using mechanochemistry, presenting a promising alternative to the conventional Schotten–Baumann acylation reaction. The results also demonstrate the effectiveness of silk sericin lipopeptides for reduced surface tension, indicating their good potential as substitutes for conventional petroleum derived surfactants in different applications.
Green foundation1. This work proposes a novel solventless methodology able to replace current industrially applied conditions to produce lipopeptide or lipoamino-acid surfactants and without the use of acyl chloride. The methodology was developed on model amino-acid and applied to a peptide hydrolysate from a protein considered as waste in the textile industry (silk sericin).2. The described new procedure for surfactants synthesis allows high conversion rates: up to 99% for lipoamino acids and 49% for silk lipopeptides comparable to conventional Schotten–Baumann in solution. It showed a significant improvement of green metrics compared with this reaction, from 23.8 to 0.2 for E-factor, from 24.8 to 1.2 for process mass intensity (PMI). And finally, the lipopeptides obtained from silk sericin show great promises as bio-based surfactants. 3. The proposed efficient and eco-friendlier approach provides a valuable reference for research on lipoamino acids and the development of similar surfactants, and could be extended to synthesize amino acid and lipopeptide surfactants from other protein sources for greener pharmaceuticals, cosmetics and detergence applications. |
1 Introduction
Surfactants, or surface-active agents, are among the most widely consumed chemical products globally, owing to their diverse functionalities, including detergency, wetting, emulsifying, solubilizing, dispersing, and foaming. Increasingly, both manufacturers and consumers seek novel surfactants that are biodegradable, biocompatible, and derived from renewable resources, produced using clean and sustainable technologies. Proteins, such as renewable polymers, have significant potential for developing sustainable, biodegradable, and eco-friendly surfactants with low toxicity. In light of this, lipopeptides emerge as promising candidates due to their dual functionality. They not only serve as effective surfactants but also exhibit biological activities, such as antimicrobial properties, anti-wrinkle effects, and nutrient-like roles, further broadening their applicability in various fields.1–4 Natural-based lipopeptides are mainly derived from agricultural biomass, composed of proteins, sugars, starches and oils.5–8 However, the current challenge lies in identifying new non-food resources to address increasing global demand. In this context, silk proteins are a promising alternative as non-food source of amino acids for amphiphilic molecule production. Silk from Bombyx mori cocoons is a natural polymer which consists of two primary proteins: fibroin, which constitutes 70–80% of the silk cocoon and is extensively used in the textile industry and in biomaterials such as FDA-approved polymer, and sericin, which surrounds the fibroin fibers. Sericin, is primarily a by-product of textile effluents, with a composition rich in serine (25–40%), aspartic acid (13–19%), and glycine (10–18%).9,10 Recently, sericin has attracted significant attention from researchers for its potential in the valorization of silk biomass. Its unique properties offer promising applications in cosmetics, food industries, and biomedicine, making it a valuable resource for sustainable innovation.11–15Lipopeptides and lipo-amino acids surfactants are among the molecules that have an amide moiety within their structure. Through this bond, a hydrophobic moiety of a non-polar long chain is linked to an amino acid or peptide group considered the hydrophilic moiety, resulting in an amphiphilic molecule with surface-active properties.1 The reference method for grafting the hydrophobic group onto the protein-derived polar parts for large-scale amide production is the Schotten–Baumann reaction. This reaction consists of the formation of an amide bond between the carboxyl group of fatty acid chloride and the amine group of an amino acid, peptide or protein in water under basic conditions. This method has several advantages, such as using water as a solvent, high reaction yield, and good selectivity. However, this reaction suffers from several significant drawbacks. Notably, it involves using of fatty acid chlorides, which are classified as toxic compounds and therefore require stringent safety and handling procedures, particularly when used on an industrial scale. Moreover, these reagents are typically prepared from precursors that are themselves classified as hazardous substances. In addition, the reaction inevitably generates side products, such as alkali salts formed during the hydrolysis step. The formation of these by-products negatively impacts the greenness of the chemical process. Considering modern policies that encourage the application of sustainable development strategies for environmentally-friendly chemicals, it has been a major challenge for the chemical industry to find an amidation reaction that constructs amide bonds while respecting the principles of green chemistry.16 These principles include preventing waste, maximizing the atom economy of the final product, avoiding the use of harmful solvents and chemicals, promoting solventless reactions and using of renewable feedstocks.17 Mechanochemistry has emerged as a promising approach to satisfy these requirements.18–20
Amide or peptide bonds are ubiquitous in the majority of both natural and synthetic molecules.21,22 Due to its interesting properties, this bond has become invaluable in numerous research fields.23 Recently, the development of novel amidation methodologies using mechanochemistry has seen significant progress. Most studies have been dedicated to synthesizing protected peptides,24,25 amino esters25,26 and amides.27,28 The first approach to amidation involved using of carboxylic acids activated by coupling agents, to replace the hydroxyl group –OH of the carboxylic acid function with a better leaving group. The following coupling agents, such as 1,1-carbonyldiimidazole (CDI),27–29 2,4,6-trichloro-1,3,5-triazine (TCT) and triphenylphosphine (PPh3),30N-ethyl-N′-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDC)31,32 are commonly used in peptide bond synthesis protocols in mechanochemistry. For example, Lamaty et al. explored the synthesis of peptides involving a Boc-protected α-amino acid N-hydroxysuccinimide ester (Boc-AA-OSu) and alkyl ester salts of α-amino acids.33 This was conducted in the presence of sodium bicarbonate (NaHCO3) and using ethyl acetate or tert-butyl acetate as liquid-assisted grinding (LAG) solvents, employing a vibrational ball mill. Under optimal conditions, a yield of 95% is obtained. Until recently no mechanical approaches were used to produce lipopeptides. We propose a mechanochemical approach to amino acid and peptide N-acylation for lipopeptide production. This process uses a one-pot, two-step method with 1,1′-carbonyldiimidazole (CDI) fatty acid activation, followed by acylation.34 The procedure was optimized on model amino acids such as leucine, glycine, serine, and aspartic acid using a vibrational ball mill and a planetary ball mill. High conversion rates of 99% were obtained for amino acid acylation under optimal conditions. Moreover, the methodology was extended to the grafting of hydrophobic chains onto silk sericin hydrolysates with an acylation rate of 48–51%. Lipopeptides obtained using the mechanochemical methodology showed good surface activity and reduced surface tension.
The mechanochemical approach thus not only makes the process more sustainable, but also reduces costs, simplifies the synthesis of peptides and amides, and makes it more suitable for larger-scale production. Despite all these advantages, some of the coupling reactants that can be used for amide bond formation are considered harmful products due to their non-green nature (EDC, DMAP, PPh3).35 Additionally, the method requires stoichiometric quantities of the coupling agent, which reduces the atom economy of the syntheses applied.
Few studies described the mechanochemical amidation approach without use of coupling agents. Stolar et al. described a thermo-mechanical method for directly coupling carboxylic acids and amines applied to synthesizing the active pharmaceutical ingredient with a conversion of up to 98%.36 Bil et al. investigated the mechanochemical synthesis of aldoamines, which have notable properties attributed to the presence of the amide group.37 The approach involves the aminolysis of lactones using mechanochemistry water as a LAG, and achieves a yield of 90% for N-dodecyl-galactonamide within just 5 minutes. Nicholson et al. established a method for amide synthesis from esters and amines using a vibrational ball mill.38 They demonstrated that direct amidation could be achieved without using a solvent, although a base, potassium tert-butoxide, was necessary. Their approach was applied to various amino acids and esters and generated an extensive collection of amides, achieving moderate to excellent yields (11–99%) under solventless conditions.
Mechanical amidation approaches are thus increasingly diverse, and show very promising results that address the growing needs across various applications for the amide bond. Considering the more recent scientific progress, it would thus be interesting to study the reaction of fatty acid grafting onto amino acids using a mechanochemical approach without activating agent.
The aim of this work is to develop a new strategy for lipopeptide synthesis through mechanochemistry without acyl chlorides. It focuses on producing lipopeptide amphiphilic molecules derived from amino acids and peptides, adapting the methodology proposed by Nicholson et al. using fatty esters in the presence of potassium tert-butoxide as the base. The amidation reaction was firstly optimized using model amino acids such as glycine, leucine, and serine. The conversion rates were quantified using NMR and UV/Vis spectroscopy. The optimized procedure was subsequently applied to peptides of silk sericin, as a natural, non-food biomass, with potential applications in cosmetics, detergents, and pharmaceuticals. The physico-chemical properties of sericin lipopeptides, such as air/water surface tension, aggregate size and zeta-potential were analyzed. Finally, green metrics were evaluated to compare the environmental impact of this method with conventional synthesis approaches in solution.
2 Experimental methodology
2.1 Chemical reactants
Bombyx mori silk cocoons were purchased from the company Au fil d'Emma, France. Alcalase from Bacillus licheniformis (2.964 U mL−1) was purchased from Merck Millipore (Billerica, MA, USA). Potassium tert-butoxide (KOtBu), methyl myristate (C14OCH3), methyl laurate (C12OCH3), methyl decanoate (C10OCH3), O-phthaldialdehyde 97% (OPA), β-mercaptoethanol, deuterated chloroform (CDCl3) 99.5%, glycine, leucine and serine were purchased from Sigma-Aldrich (Saint-Quentin Fallavier, France). Deuterium oxide 99.9% (D2O), and NaOH solutions (1 mol L−1) were purchased from VWR. Demineralized water at 18 MΩ was produced using an Aquadem demineralization system (Veolia, Water STI, France).2.2 Synthesis of lipoamino acids
The methodology for the direct amidation of esters using ball milling was originally described by Nicholson et al.38 and is used here with some changes. The mechanochemical reactions were performed in the ball mill equipment: a planetary ball mill PM100 (Retsch GmbH, Germany) using a 50 mL zirconium oxide grinding jar (167 zirconium oxide balls of 5 mm of diameter). The amidation reaction was optimized on model amino acids: glycine, leucine and serine in terms of stoichiometry of reactants amino acid, base and fatty ester and reaction time. The amino acids glycine and leucine were pretreated with an equimolar amount of sodium hydroxide (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio) and freeze dried, the resulting amino acid in its aminocarboxylate form was used without any further purification. The amidation reaction was then performed following a one-pot one-step method using KOtBu as a base. For serine, the same protocol could not be used because as it had been freeze-dried, a significant amount of residual water was still present, leading to unsuitable degradation of the base during the amidation reaction. Therefore, the basification was carried out with mechanochemistry before the amidation step using one equivalent of KOtBu as the base and a second equivalent was added along with the fatty ester for the amidation reaction.
1 molar ratio) and freeze dried, the resulting amino acid in its aminocarboxylate form was used without any further purification. The amidation reaction was then performed following a one-pot one-step method using KOtBu as a base. For serine, the same protocol could not be used because as it had been freeze-dried, a significant amount of residual water was still present, leading to unsuitable degradation of the base during the amidation reaction. Therefore, the basification was carried out with mechanochemistry before the amidation step using one equivalent of KOtBu as the base and a second equivalent was added along with the fatty ester for the amidation reaction.
The amidation reaction consists of adding the pretreated amino acid and the fatty ester to the grinding jar in the presence of the base KOtBu, then the reaction mixture was ground during different time periods at 650 rpm. The lower rotation speed of the milling jar was previously evaluated, and systematically resulted in reduced conversions.34 The reaction time varied from 60 to 90 and 120 minutes, and the molar ratios of the base and ester were optimized. For the base, the addition of 0.85, 1, and 1.5 equivalents were tested, and then the addition of 1, 1.2, and 2 equivalents of the ester were evaluated to obtain the best conversion. At the end of the reaction, the mixture was removed from the jar then stored in a closed bottle at 20 °C for further analysis. The conversion of the amino acid into N-acylamino acid was estimated from the 1H NMR spectrum by comparing integration of the peaks corresponding to the –CHα of the remaining amino acid and the –CHα of the acylated amino acid.
All the NMR measurements were performed using a Bruker Avance III 400 MHz spectrometer. NMR samples were prepared in D2O. About 20 mg of sample was dissolved in D2O and introduced into an NMR tube. The 1H NMR spectra were acquired at 400 MHz and recorded in ppm using the solvent residual HOD peak as reference (4.79 ppm).
2.3 Synthesis of sericin lipopeptides using mechanochemistry
Developing silk sericin lipopeptides consists in three steps: extracting the sericin from cocoons, hydrolyzing the proteins to obtain peptides, and grafting the hydrophobic moiety using mechanochemistry.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 w/w cocoon-to-water ratio at 120 °C for 20 min, as described previously.39 Silk sericin solution was separated from the fibroin fibers and recovered via filtration. The sericin solution obtained at a concentration of 1% w/w was used directly in the hydrolysis process.
25 w/w cocoon-to-water ratio at 120 °C for 20 min, as described previously.39 Silk sericin solution was separated from the fibroin fibers and recovered via filtration. The sericin solution obtained at a concentration of 1% w/w was used directly in the hydrolysis process.
Sericin peptides were produced by enzymatic hydrolysis using an Alcalase enzyme according to the method described in our previous studies.34,40 Sericin solution was treated with 1% of an enzyme/protein ratio at 50 °C for 30 min. Following the reaction period, the enzyme was deactivated by heating to 95 °C for 10 min. The hydrolysate solution was then adjusted to pH 11 by adding 1 M NaOH solution. The purpose of this basification is to improve the nucleophilicity of the terminal amine group in sericin peptides, resulting in both better reactivity in the acylation reaction and higher conversion. Next, the peptide mixture was freeze-dried and stored at 4 °C. The degree of hydrolysis (DH) was evaluated using the o-phthaldialdehyde method (OPA) based on UV/Vis spectroscopy using a UV/Vis spectrometer Lambda 12 (PerkinElmer, United States).41 The DH is defined as follows:
 | (1) |
 | (2) |
The formation of the amide bond between sericin peptides and the fatty chain was confirmed from an FTIR analysis recorded with a Nicolet iS5 FTIR spectrometer (Thermo Scientific, United States) equipped with a transmission accessory for KBr pellets. A background was recorded in ambient conditions with no sample. All samples were scanned 16 times with a wavenumber ranging from 400 to 4000 cm−1 with a resolution of 4 cm−1.
2.4 Characterizing the lipopeptide solutions
The sericin lipopeptide products obtained consisted predominantly of lipopeptides, as well as non-functionalized peptides and residual fatty chains. In line with industrial practice, and to avoid using organic solvents, these mixtures were characterized without further purification. Aqueous solutions of lipopeptides were prepared using demineralized water to analyze surface tension and physicochemical properties. The resulting lipopeptide solutions had a slightly alkali pH depending on the concentration. The pH of the samples analyzed was thus adjusted to 7 using a 1 M NaOH solution. Surface activity of sericin peptides and sericin lipopeptides was measured using a Tracker tensiometer from TECLIS (Teclis Instruments, Civrieux d'Azergues, France) equipped with an automatic injection system and a camera. The pendant drop method was used for the analysis. A 250 μL syringe was used to take a sample of the solution and a straight G18 needle was fixed on the syringe tip to form the droplet. Calibration with pure deionized water was performed before each series of measurements. At the lowest concentration (1 g L−1), sericin lipopeptides were easily soluble in water, forming clear and translucent solutions. For the higher concentrations, 5 and 10 g L−1, the solutions obtained were turbid. It can be assumed that this turbidity came from the residual unreacted fatty chain in our samples. A droplet volume of between 6 and 13 μL was chosen, depending on the lipopeptide sample and concentration, to obtain the maximal deformation in the drop. For each measurement, the surface area and volume of the pendant drop were fixed throughout the analysis. The equilibrium surface tension for lipopeptides was considered at 1 hour for all samples.The presence of lipopeptide aggregates in solution was analyzed with dynamic light scattering (DLS) by determining the mean hydrodynamic diameter at pH 7. Measurements were performed using a Zetasizer Ultra (Malvern Panalytical, UK) with detection angles set at 173°, 90°, and 13°. Intensity-weighted hydrodynamic diameter distributions were obtained using ZS Xplorer software. Lipopeptide solutions were analyzed at room temperature in polystyrene cuvettes. The ζ-potential of the lipopeptide aggregates was measured using the same instrument at pH2, 7 and 10. Prior to the ζ-potential measurements, lipopeptide solutions were filtered through a 0.22 μm PVDF membrane directly into the measurement cuvette.
2.5 Green metrics calculation
Green metrics were calculated for sericin lipopeptides obtained using the amidation approach in solvent-free conditions and compared to the sericin lipopeptides produced by a conventional Schotten–Baumann reaction in aqueous solution40 (SI, Table S1). The atom economy (AE) was calculated as the ratio of molecular weight of the target product Mw (lipopeptide) to the sum of molecular weights of all the products in a stoichiometric equation of reaction42–44 following eqn (3): | (3) |
The environmental E-factor was expressed as the ratio between waste mass and product mass.44 The waste mass can be expressed as the difference between the mass of raw materials entering the reactor and the product mass (eqn (4)).
 | (4) |
The process mass intensity (PMI) was estimated as the ratio between the total mass of reactants in the process and the mass of product of interest as mentioned in eqn (5). This factor also indicates the “greenness” of a process based on the total mass of materials required to produce a specific mass of product.
 | (5) |
3 Results and discussion
Mechanochemical synthesis by direct ester amidation in the presence of a KOtBu base contributes to the formation of an amide bond between the terminal amine function of an amino acid or peptide and the fatty ester. The approach developed leads to promising results in the synthesis of lipopeptides, thereby avoiding the use of acyl chlorides applied in the traditional Schotten–Baumann reaction for lipopeptide synthesis. Our team has previously studied the mechanosynthesis of lipopeptides using 1,1′-carbonyldiimidazoles as a coupling agent to activate the fatty acid, giving acylation rates up to 50%. However, this route of direct amidation of esters seems more advantageous, because it requires using a base, KOtBu, which can be easily removed from the medium unlike the by-products of CDI, imidazoles, which are harmless but remain in the final product.Glycine and methyl laurate were chosen as the model to set the experimental parameters and the optimal quantities of reactants. The optimized reaction was then applied to leucine and serine (Scheme 1).
3.1 Optimizing N-acyl-glycine synthesis
The reaction was obtained using a planetary mill zirconia grinding jar. The protocol used was inspired by the work of Nicholson et al. This protocol involves a one-pot one-step reaction and consists of mixing 1 equivalent of glycine (previously lyophilized in its basic form) with 1.2 equivalents of methyl laurate in the presence of 0.85 equivalents of KOtBu. After a milling time of 1 hour, the reaction achieved a conversion of 47% (Table 1, entry 1). The conversion was determined from 1H NMR spectra of glycine and N-acyl-glycine by comparing the integration of the peaks corresponding to the –CHα of glycine and the –CHα of N-acyl-glycine (Fig. 1). | ||
| Fig. 1 1H NMR spectra from top to bottom of glycinate, free fatty carboxylate and crude mixture of N-acyl-glycine reaction corresponding to entry 6 in Table 1 carried out in HOD. | ||
| Entry | Amino acidb (x eq.) | Ester (x eq.) | Base (x eq.) | Reaction time (min) | Conversionc (%) |
|---|---|---|---|---|---|
| a Reaction conditions: planetary ball mill, 50 mL zirconia jar and 167 zirconia balls (5 mm diameter) at 650 rpm. b Glycine pre-treated with 1 eq. of NaOH. c Estimated with 1H NMR. d Glycine basified in situ with KOtBu. e Glycine basified with KOtBu for 20 min before the amidation reaction. | |||||
| 1 | Glycine (1 eq.) | C12OCH3 (1.2 eq.) | KOtBu (0.85 eq.) | 60 | 47 |
| 2 | Glycine (1 eq.) | C12OCH3 (1.2 eq.) | KOtBu (1 eq.) | 60 | 86 |
| 3 | Glycine (1 eq.) | C12OCH3 (1.2 eq.) | KOtBu (1.5 eq.) | 60 | 86 |
| 4 | Glycine (1 eq.) | C12OCH3 (1 eq.) | KOtBu (1.5 eq.) | 60 | 66 |
| 5 | Glycine (1 eq.) | C12OCH3 (2 eq.) | KOtBu (1.5 eq.) | 60 | 99 |
| 6 | Glycine (1 eq.) | C12OCH3 (1 eq.) | KOtBu (1.5 eq.) | 90 | 85 |
| 7 | Glycine (1 eq.) | C12OCH3 (1 eq.) | KOtBu (1.5 eq.) | 120 | 81 |
| 8 | Glycine (1 eq.) | C10OCH3 (1 eq.) | KOtBu (1.5 eq.) | 90 | 78 |
| 9 | Glycine (1 eq.) | C14OCH3 (1 eq.) | KOtBu (1.5 eq.) | 90 | 79 |
| 10 | Glycined (1 eq.) | C12OCH3 (1 eq.) | KOtBu (2.5 eq.) | 90 | 47 |
| 11 | Glycinee (1 eq.) | C12OCH3 (1 eq.) | KOtBu (2.5 eq.) | 20 + 90 | 62 |
Glycine has one characteristic peak at 3.14 ppm that corresponds to the two protons in the alpha position present in its structure (Fig. 1, top spectrum). By analyzing the 1H NMR spectrum corresponding to N-acyl-glycine (Fig. 1, bottom spectrum), it is possible to identify the peaks corresponding to the acylated product and the unreacted reactants, glycine and fatty chain. The peak at 3.61 ppm was ascribed to the –CH2α of N-acyl-glycine (in blue in Fig. 1). This peak is shifted compared to the –CH2α of glycinate (in green in Fig. 1) because the chemical environment of the α protons had changed. When conversion was not total the presence of unacylated glycine was therefore confirmed by the peak at 3.06 ppm ascribed to the –CH2α of glycinate. The conversion was determined on the N-acyl-glycine spectrum by comparing the integration of the two peaks. The peaks at 2.17, 1.48, 1.17 and 0.75 ppm were ascribed respectively to the αCH2, βCH2 (a and b signals highlighted in violet in Fig. 1) and all the other –CH2 and the terminal –CH3 (signals highlighted in yellow on Fig. 1) in the fatty alkyl chain. The peaks at 2.04 and 1.45 ppm (a and b signal highlighted in red on Fig. 1) were attributed to the αCH2 and the βCH2 of the unreacted fatty alkyl chain.
To improve the amidation reaction and increase conversion, the molar equivalence of the base and ester was optimized. Increasing the amount of base from 0.85 to 1 and 1.5 equivalents made in possible to increase the conversion from 47 to 86% (Table 1, entries 1–3). In the study carried out by Nicholson et al. 0.85 equivalents of the base were the optimum for the reaction between cyclic and secondary amines and heteroaromatic and heterocyclic esters in mechanochemical conditions.38 When compared to other studies conducted by Kim et al. and Slavchev et al., up to 2.5 equivalents of KOtBu are added for the direct amidation of esters under ambient and green conditions, in solution using THF and DMSO as solvents.45,46 In this study, a linear fatty ester with a different structure was used for lipopeptide mechanosynthesis. As KOtBu is sensitive to humidity and can easily degrade a slight excess of KOtBu (1.5 eq.) was needed to achieve the best yields.
To evaluate the influence of the amount of ester, it was found that the increase in equivalents from 1 to 1.2 then 2 equivalents made it possible to increase conversion from 66 to 86 and 99% respectively (Table 1, entries 3–5). Based on these results two equivalents of the ester made possible complete conversion of the amide in one hour. However, the excess of the ester was not preferred, because the final product will be used without further purification to align the process with industry practices. The presence of a significant amount of residual fatty acid is undesired, as it impacts the physico-chemical properties of the lipopeptide mixture. To avoid excess of the unreacted hydrophobic chains in the final product, 1 equivalent of the ester was set and the reaction time was optimized.
Increasing the reaction time from 60 to 90 min increased the glycine conversion from 66 to 85% (Table 1, entries 4 & 6). Nevertheless, a reaction time of 120 min led to a conversion of 81% only (Table 1, entry 7). Therefore, all the following amidation reactions were run for 90 min as even after a prolonged reaction time (120 min), the amino acid had not been fully converted.
With the optimal conditions established, the amidation of two fatty esters with chain lengths of 10 and 14 carbons was explored using glycine as the amine component. The conversions obtained with both methyl decanoate and methyl myristate were 78 and 79%, respectively. These results indicate that there is no significant difference in conversion for esters with variable chain lengths (Table 1, entries 6–9). The same tendency was observed in our previous work, on the mechanosynthesis of lipoamino acids and lipopeptides with pre-activation of the fatty acid by CDI.34
As mentioned previously, glycine was pre-treated with 1 equivalent of sodium hydroxide. This pre-treatment with NaOH enhanced the nucleophilicity of the glycine's terminal amino group, resulting in better reactivity in the acylation reaction and higher conversion. To circumvent the pre-treatment step followed by freeze-drying, the basification of glycine was tested through ball milling, using the same base, KOtBu. It was assumed that 1 equivalent of the base would be used for the basification reaction, and 1.5 equivalents would be used for the amidation reaction. For this reason, 2.5 equivalents of the base were added in one pot one step with glycine and ester. These conditions resulted in a conversion rate of 47% (Table 1, entry 10). This lower conversion could be due to the direct competition between the basification reaction of glycine and its amidation reaction with the ester, or to the presence of moisture in the glycine powder, which was not dry before the reaction.
Another experiment was conducted by basifying glycine before the amidation reaction using the one pot two step approach. One equivalent of KOtBu was added to one equivalent of glycine, and this mixture was treated for 20 min in the ball mill. Subsequently, 1 equivalent of the ester and 1.5 equivalents of the base were introduced to the milling jar, and the amidation reaction was carried out for 90 min. This produced conversion of 62% (Table 1, entry 11).
3.2 N-Acylation of amino acids using the amidation approach
After optimizing the amidation reaction on glycine, the optimal conditions were applied to leucine and serine amino acids, to evaluate the acylation potential of other amino acids.1H NMR spectra were used to determine the conversion rates of leucine and serine by comparing integration of the –CHα free amino acid group with that of the –CHα of the acylated amino acid (SI, Fig. S2 and S3) as previously detailed for N-acyl-glycine. Leucine and serine pre-treated with one equivalent of NaOH were used for the amidation reaction. The low conversion rate achieved for N-acyl-leucine could be attributed to its larger and hydrophobic lateral chain, resulting in conversion of 8 and 21% (Table 2, entries 1 and 3 respectively). The steric effects created by the leucine lateral chain may prevent easy access of the amine to the methyl laurate, thus reducing the efficiency of the reaction. To push the reaction further, another experiment was conducted with leucine by adding 1.2 equivalents of the ester (Table 2, entry 2). The conversion improved significantly, reaching 51%. The amidation reaction between serine pre-treated with 1 equivalent of NaOH and methyl laurate achieved a conversion of only 16% (Table 2, entry 4). This result might be caused by the presence of a significant amount of moisture after freeze drying the treated serine, which is incompatible with KOtBu as it is sensitive to water. Water molecules remain strongly bound to the serine, which may hinder the reaction. To prevent this problem, serine was basified with KOtBu for 20 min before the amidation reaction. The conversion of the product obtained increased to 57% (Table 2, entry 5). Increasing the ester quantity from 1 to 1.2 equivalents raised the conversion to 79% (Table 2, entry 6). It should be noted that no esterification reaction with the hydroxyl function on the serine side chain was detected during this reaction as can be shown on the NMR spectra (SI, Fig. S3) which shows that this amidation protocol is highly selective. Based on these findings, it was noticed that the reaction conversions largely depended on the structure of the amine component and the presence of water. The differences in conversion rates could be attributed to the structural variations of the lateral group of each amino acid. Glycine, with only a single hydrogen atom (H) as its side chain, exhibited the highest acylation rate. This can be explained by its minimal steric hindrance, facilitating the accessibility of the fatty chain to the amino group, thereby enhancing the amidation reaction. Leucine has a bulky side group (CH2CH(CH3)2), which can cause significant steric hindrance during the amidation reaction. This hinders the accessibility of the fatty chain to the amino group, thereby reducing the reaction efficiency and explaining the lower conversion rate observed. Serine has a hydroxyl function on its side chain (CH2OH). This group may have a moderate steric effect on the amidation reaction, which explains the good conversion rate achieved. Nicholson et al. also observed variations in yield when applying the mechanochemical amidation protocol using KOtBu to a wide range of ester and amine substrates.38
| Entry | Amino acid (x eq.) | Ester (x eq.) | Base (x eq.) | Reaction time (min) | Conversiond (%) |
|---|---|---|---|---|---|
| a Reaction conditions: planetary ball mill, 50 mL zirconia jar and 167 zirconia balls (5 mm diameter) at 650 rpm. b Amino acid pre-treated with 1 eq. of NaOH. c Serine basified with KOtBu for 20 min before the amidation reaction. d Estimated by 1H NMR. | |||||
| 1 | Leucineb (1 eq.) | C12OCH3 (1 eq.) | KOtBu (1.5 eq.) | 90 | 21 |
| 2 | Leucineb (1 eq.) | C12OCH3 (1.2 eq.) | KOtBu (1.5 eq.) | 90 | 51 |
| 3 | Leucineb (1 eq.) | C12OCH3 (1 eq.) | KOtBu (2.5 eq.) | 20 + 90 | 8 |
| 4 | Serineb (1 eq.) | C12OCH3 (1 eq.) | KOtBu (1.5 eq.) | 90 | 16 |
| 5 | Serinec (1 eq.) | C12OCH3 (1 eq.) | KOtBu (2.5 eq.) | 20 + 90 | 57 |
| 6 | Serinec (1 eq.) | C12OCH3 (1.2 eq.) | KOtBu (2.5 eq.) | 20 + 90 | 79 |
In this study, a base-promoted system for the direct amidation of esters was used to synthesize lipoamino acids, yielding good to high conversion rates ranging from 51 to 99% for three different amino acids. These results are promising for synthesizing of our target molecules. The conversion rates obtained are comparable to those reported in the introduction, such as the 11 to 99% yields achieved by Nicholson et al. with a similar system.38
3.3 Synthesis of sericin lipopeptides
Lipopeptide synthesis is conventionally performed under Schotten–Baumann conditions.5,40,47 With the progress made in amide bond synthesis under solvent-free conditions, producing lipopeptides via this route appears feasible. The direct amidation of esters using KOtBu was selected for developing silk lipopeptide surfactants due to its alignment with green chemistry principles. As previously mentioned, silk sericin is predominantly considered waste from the textile industry. Therefore, its use as a raw material for the hydrophilic part of lipopeptides fits within the green chemistry framework.Developing sericin lipopeptides using a Schotten–Baumann reaction or mechanochemical coupling with CDI activation was previously described by our team.34,40 The Schotten–Baumann reaction was carried out in an aqueous solution at pH 9.0 for 3 hours, and sericin peptides were functionalized with a fatty acid chloride. The resulting acylation rates varied from 27% to 41%. The mechanochemical coupling with CDI was carried out on two different sericin hydrolysates with an acylation rate of 48–51% using three fatty acids with chain lengths of 10, 12, and 14. The objective of the present work is to produce similar sericin lipopeptides using the amidation approach in solventless conditions with fatty esters and KOtBu in the planetary ball mill (Scheme 2).
First, sericin peptides were produced through enzymatic hydrolysis of the protein to constitute the hydrophilic part of the lipopeptides. Sericin peptides with a DH of 66 ± 2% were used for lipopeptide synthesis. Sericin peptides can also be characterized by a Peptide Chain Length (PCL) that corresponds to the average number of amino acids and is calculated from the inverse of the DH.7 In our peptide mixture, we were able to estimate that the peptides produced had an average peptide chain length (PCL) of 1.5 amino acids. Tsericin lipopeptides were then acylated in the optimal conditions obtained on the model amino acids. In the planetary ball mill, sericin peptides, fatty ester and KOtBu were added to the milling jar at a molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5, and the amidation reaction was maintained for 90 min. After this period, the acylation rate (AR) of the sericin peptides was estimated with the OPA dosage as the quantitative discrimination of peptides and lipopeptides using NMR could not be carried out. It could only be observed that fatty alkyl chains were present in the mixture by visualizing the –CH2 of the chain and the terminal –CH3 (Fig. 2). It is important to note that even after further purification to eliminate any unreacted fatty chain, the lipopeptide mixture still exhibited these signals on the spectra.
1.5, and the amidation reaction was maintained for 90 min. After this period, the acylation rate (AR) of the sericin peptides was estimated with the OPA dosage as the quantitative discrimination of peptides and lipopeptides using NMR could not be carried out. It could only be observed that fatty alkyl chains were present in the mixture by visualizing the –CH2 of the chain and the terminal –CH3 (Fig. 2). It is important to note that even after further purification to eliminate any unreacted fatty chain, the lipopeptide mixture still exhibited these signals on the spectra.
 | ||
| Fig. 2 NMR spectra of sericin peptides (DH = 66 ± 2%) and sericin lipopeptides mixtures LP-C14 carried out in HOD. | ||
The structure of the sericin lipopeptides following the amidation reaction was analyzed using FTIR spectroscopy of both the lipopeptides and non-grafted peptides. As can be seen in the results in Fig. 3, silk sericin exhibited characteristic vibrational bands of proteins in IR spectra, in the range of 1630 to 1650 cm−1 for amide I, 1540 cm−1 to 1520 cm−1 for amide II and 1270 cm−1 to 1230 cm−1 for amide III.48 These specific vibration bands correspond to sericin proteins reported previously.39 By analyzing the spectrum of the sericin lipopeptides (Fig. 3), the same spectra shape was obtained with additional bands that showed the formation of amide bonds between the N-terminal and the carbonyl group of the fatty ester. The band at 1560 cm−1 was associated with the N–H bending vibration combined with the partial contribution of C–N stretching (amide II band). The band observed between 2850 and 3000 cm−1 primarily corresponded to the C–H stretching vibrations associated with the –CH3 and –CH2 groups present in the long fatty chains.
To examine the influence of the size of the hydrophobic part on the acylation reaction, three fatty esters of varying chain lengths (10, 12 and 14) were grafted onto sericin peptides. The AR of sericin lipopeptides obtained with the amidation approach using mechanochemistry are presented in Table 3.
An acylation rate of 40 ± 1% was obtained with methyl decanoate, while this rate reached 47 ± 1% with methyl laurate and 49 ± 1% with methyl myristate. These results indicate a slight increase in AR for the reaction with C12–C14 fatty methyl esters. This trend can be compared with previously reported acylation rates for sericin peptides. Under conventional Schotten–Baumann conditions in an aqueous medium, the AR of sericin peptides (DH = 45%) remained similar for the three corresponding fatty acyl chlorides (chain lengths of 10, 12 or 14 carbons), ranging between 38 and 41%.40 The moderate improvement in AR achieved with the mechanochemical amidation approach, compared to the Schotten–Baumann reaction, could be related to the higher DH of the sericin peptides used in the present study (66% vs. 45%). Even at a fixed peptide –NH2/fatty acid ester molar ratio, the acylation reaction proceeded more efficiently when a greater amount of terminal –NH2 functions was available for reaction with the fatty chain. Additionally, during the Schotten–Baumann reaction in solution, a competing secondary reaction occurs, in which the acid chloride hydrolyzes to form sodium alkanoate. On the other hand, the silk sericin peptides (DH = 64%) produced with a mechanochemical approach using a CDI pre-activation strategy, led to an AR of about 47–51% for fatty acids with the same chain length.34 These findings demonstrate that both mechanochemical procedures, direct amidation and CDI pre-activation, make comparable AR of sericin peptides possible, which is highly promising for this type of molecule in the absence of a solvent.
3.4 Characterizing lipopeptides
The results of surface tension measurements for sericin peptides and the lipopeptides mixture LP-C14, LP-C12, LP-C10 produced via the amidation approach are presented in Fig. 4. It is important to note that the products obtained were mainly composed of lipopeptides, but also contained non-functionalized peptides and residual fatty chains. Aligning the process with industry practices, the adsorption properties of these mixtures were analyzed without further purification. Based on the results, acylation enhanced the amphiphilic nature of the sericin peptides. The adsorption of the lipopeptides at the air/water interface became more efficient resulting in lower surface tension values. Increasing the lipopeptide concentration from 1 to 5 g L−1 accelerated the decrease in surface tension and led to lower equilibrium values for all samples. However, no significant change in surface tension values was observed for the solutions at 5 and 10 g L−1, which was due to interface saturation and the formation of self-assembled lipopeptide structures in solution (data not shown). For instance, the equilibrium surface tension of the LP-C14 lipopeptide mixture was 41.5 ± 1, 23.6 ± 1 and 25 ± 1 mN m−1 for solutions at 1, 5 and 10 g L−1 respectively. The LP-C12 and LP-C10 samples were less efficient in reducing surface tension and exhibited higher surface tension values, ranging from 59 to 35 mN m−1 depending on concentration. It can be assumed that the adsorption of sericin-derived lipopeptides was more efficient with increasing hydrophobic chain length.
 | ||
| Fig. 4 The equilibrium surface tension of the sericin peptides and lipopeptide mixtures LP-C14, LP-C12 and LP-C10 at t = 1 h (pH = 10). | ||
Our previous work on the acylation of sericin peptides using the CDI pre-activation approach reported the surface tension of lipopeptide solutions ranging from 25.9 to 35.5 mN m−1 at 5 g L−1.34 Variations in surface tension values compared to the results of the present study are probably due to differences in the AR and the presence of synthesis byproducts (imidazole) for CDI activation synthesis. Other studies have also reported the surface properties of silk peptide-based surfactants. For example, silk lipopeptides with a C12 fatty chain synthesized using a Schotten–Baumann reaction in a water/acetone mixture achieved a surface tension of 36.4 mN m−1 at 1.24 g L−1.49 Similarly, Wu et al. produced lipopeptides derived from silkworm pupae, which exhibited a surface tension of 28.04 mN m−1 at 2.57 g L−1.50 All these findings indicate that the silk sericin lipopeptide surfactants developed in the present work showed promising surface activity compared with other peptide- or silk-based surfactants. However, it is essential to consider that all the constituents of the mixture could participate in this reduction, including the lipopeptide, the non-grafted peptides and the rest of the fatty chain. Although it is difficult to identify the real individual contribution or potential synergistic effects of each species, it should be noted that the lipopeptide was the main component of the mixture. Given these initial results, sericin-derived lipopeptides produced via the solvent-free amidation methodology appear to be promising candidates for the potential replacement of petroleum-based amphiphiles.
It is important to mention that lipopeptides consist of a mixture. They mainly contain sericin lipopeptides, but also unreacted peptides and residual fatty acids. Fig. 5A shows the results of zeta potential measurements across a range pf pH values for three sericin lipopeptides. At a neutral pH of 7, lipopeptides are predominantly negatively charged due to the deprotonation of the terminal carboxylic group, as the pKa of –COOH function is lower than 7. Considering that silk sericin is rich in serine, aspartic acid and glycine,51 the terminal groups of peptides produced corresponded more frequently to one of these amino acids. The non-acylated peptides at neutral pH had a zwitterionic character, with a protonated amino terminus (–NH3+) and a deprotonated carboxylate group (–COO−), in agreement with reported amino acid pKa values.52 Free fatty acids present in the mixture were likewise deprotonated and contributed additional negative charges to the system. At pH 12, all lipopeptides showed negative values for zeta potential. In these conditions, the acidic groups of the lipopeptides were fully deprotonated (–COO−), non-acylated peptides displayed a negative charge, featuring a carboxylate terminus (–COO−), an unprotonated amine terminus (–NH2), and free fatty acids in their deprotonated carboxylate form. Under acidic conditions, the zeta potential shifted toward positive values for all lipopeptide mixtures because the acidic groups of lipopeptides, non-acylated peptides, and fatty acids were protonated (–COOH) and thus neutral, whereas the amine groups of the non-acylated peptides were in their protonated form (–NH3+). Across the entire pH range, sericin lipopeptides synthesized by mechanochemistry displayed zeta potentials comparable to those reported for sericin lipopeptides prepared using Schotten–Baumann reactions in solution.40
 | ||
| Fig. 5 A: Zeta potential of sericin lipopeptides as a function of pH, B: size distributions of aggregates formed by lipopeptides. | ||
The aggregate size of the sericin lipopeptides was evaluated using dynamic light scattering (DLS), and the resulting size distributions are presented in Fig. 5B. The lipopeptide mixtures LP-C14, LP-C12 and LP-C10 formed aggregates of comparable size, with average diameters of 312 ± 14 nm, 336 ± 35 nm and 296 ± 25 nm respectively. A second minor population of smaller particles (∼90 nm), likely corresponding to monomers or small assemblies of lipopeptides, was detected for the LPF-C12 sample. Similar results have been previously reported for lipopeptides derived from cotton proteins.53 On the other hand, common synthetic surfactants such as SDS and CTAB form smaller micelles, with hydrodynamic diameters between 2 and 10 nm.54 The ability of protein-derived amphiphilic molecules to form larger aggregates arises from their bulky hydrophilic peptide head groups, which make possible additional intermolecular interactions such as hydrogen bonding. Variations in the length and composition of this peptide moiety led to less symmetric and more heterogeneous assemblies. In contrast to low-molecular-weight surfactants, which form uniform spherical micelles, lipopeptide assemblies are characterized by less regular hydrophobic cores and hydrophilic coronas.55
3.5 Calculating green metrics
As an emerging technology, mechanochemistry aligns with the principles of green chemistry by seeking to develop environmentally friendly processes while adopting safer approaches, without solvents, and with a minimum number of reagents. Green chemistry metrics are essential for assessing the environmental impact and efficiency of these processes.56 For the lipopeptide synthesis protocol, green metrics made it possible to evaluate synthesis via a mechanochemical amidation approach compared to the conventional Schotten–Baumann synthesis in solution. The green chemistry metrics for the conventional acylation reaction in aqueous solution and the novel mechanochemical amidation reaction are summarized in Table 4.| Green metrics & energy consumption | Ideal value | Conventional acylation reaction40 | CDI pre-activation by mechanochemistry34 | Amidation reaction by mechanochemistry |
|---|---|---|---|---|
| AR (%) | — | 41 ± 3 | 48 ± 1 | 49 ± 1 |
| E-Factor | 0 | 54.9 | 1.2 | 1.6 |
| PMI | 1 | 56.0 | 2.3 | 2.6 |
| AE (%) | 100 | 78.4 | 68.9 | 66.6 |
| Time reaction | — | 180 | 35 | 90 |
| Energy consumption (kWh) | — | 3.0 | 0.73 | 1.88 |
The atom economy (AE) assesses the efficiency of the synthesis and determines the number of atoms incorporated into the final product of interest. The closer the AE value is to 100%, the better the process is. The AE for Schotten–Baumann acylation in solution is 78.4%, compared to 68.9% and 66.6% for the CDI activation and mechanochemical amidation approaches respectively. This difference could be attributed to the use of CDI (1 equivalent, MW = 162.2 g mol−1) or KOtBu (1.5 equivalent, MW = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 122 g mol−1) for the mechanochemical reaction, while NaOH (MW = 40 g mol−1) was used for pH regulation in the aqueous solution during conventional acylation. Furthermore, the E-factor is an important parameter for estimating the amount of waste generated at the end of the synthesis reaction. In contract to the AE where 100% is the optimal reaction yield, the E-factor considers the chemical yield. The optimal E-factor is zero, reflecting a reaction that generates no waste. The results show a significant reduction in the E-factor to 1.2 for the CDI activation approach and 1.6 for the amidation reaction, compared to 54.9 for the conventional reaction in solution respectively. This significant enhancement is due to the elimination of the solvent (water) from the synthesis process, made possible by the solventless methods. Although both mechanochemical strategies exhibit similar E-factor values, they differ in the nature of the coproduct formed, with an imidazolium salt for the CDI-based approach and base for the amidation method. Process mass intensity (PMI) is another environmental process waste metric that we also considered to compare the greenness of the lipopeptide synthesis process. The main source of waste in chemical reactions comes from solvent losses. Conducting solvent-free reactions helps address this issue and reduces the environmental impact. Therefore, PMI was significantly decreased from 56.0 for the reaction in solvent to 2.3 and 2.6 for the mechanochemical reactions. These metrics indicate that the mechanochemical reaction generates less waste and the solvent-free reaction is greener than the synthesis in solution. An improvement in reaction time was achieved in this study, reducing it by half, from 180 minutes for acylation in aqueous solution to 90 minutes for amidation using mechanochemistry, while producing the same product of interest. Finally, the estimated energy consumption shows that both mechanochemical approaches require less energy for the production of lipopeptides than conventional solution-phase synthesis. All these parameters demonstrate that the amidation reaction via mechanochemistry has a high potential for replacing conventional synthesis, as it is effective and generates no waste, adhering to the principles of green chemistry.
122 g mol−1) for the mechanochemical reaction, while NaOH (MW = 40 g mol−1) was used for pH regulation in the aqueous solution during conventional acylation. Furthermore, the E-factor is an important parameter for estimating the amount of waste generated at the end of the synthesis reaction. In contract to the AE where 100% is the optimal reaction yield, the E-factor considers the chemical yield. The optimal E-factor is zero, reflecting a reaction that generates no waste. The results show a significant reduction in the E-factor to 1.2 for the CDI activation approach and 1.6 for the amidation reaction, compared to 54.9 for the conventional reaction in solution respectively. This significant enhancement is due to the elimination of the solvent (water) from the synthesis process, made possible by the solventless methods. Although both mechanochemical strategies exhibit similar E-factor values, they differ in the nature of the coproduct formed, with an imidazolium salt for the CDI-based approach and base for the amidation method. Process mass intensity (PMI) is another environmental process waste metric that we also considered to compare the greenness of the lipopeptide synthesis process. The main source of waste in chemical reactions comes from solvent losses. Conducting solvent-free reactions helps address this issue and reduces the environmental impact. Therefore, PMI was significantly decreased from 56.0 for the reaction in solvent to 2.3 and 2.6 for the mechanochemical reactions. These metrics indicate that the mechanochemical reaction generates less waste and the solvent-free reaction is greener than the synthesis in solution. An improvement in reaction time was achieved in this study, reducing it by half, from 180 minutes for acylation in aqueous solution to 90 minutes for amidation using mechanochemistry, while producing the same product of interest. Finally, the estimated energy consumption shows that both mechanochemical approaches require less energy for the production of lipopeptides than conventional solution-phase synthesis. All these parameters demonstrate that the amidation reaction via mechanochemistry has a high potential for replacing conventional synthesis, as it is effective and generates no waste, adhering to the principles of green chemistry.
4 Conclusions
This work demonstrated an efficient new method for the amidation of amino acids and silk peptides in solventless basic conditions for the development of lipoamino acids and lipopeptides. The experimental conditions for amidation were optimized on model amino acids. The molecular structure of the lipoamino acids synthesized was characterized using FTIR and 1H NMR, confirming the efficient conversion. The highest conversion rate of 99% was obtained for glycine in excess of the fatty ester and base. The reaction developed was then used to produce silk lipopeptides. A series of sericin-derived lipopeptides was developed in three steps: sericin extraction, sericin enzymatic hydrolysis to produce peptides, and grafting of the hydrophobic part through acylation with fatty esters of varying carbon chain lengths. The acylation rates of 40–49% were comparable to the results obtained for the acylation of sericin peptides using the conventional Schotten–Baumann reaction in aqueous solution40 or mechanochemical acylation using CDI.34 The surface activity of sericin-derived lipopeptides was evaluated using the pendant drop method. A significant decrease in surface tension of up to 24 mN m−1 was observed for long hydrophobic chain of 14 carbons. The approach proposed makes it possible to recover the sericin protein, a waste by-product of the silk industry, as a source of renewable peptides. This solventless methodology is a valuable reference for research on lipoamino acids and the development of similar surfactants, and could be extended to synthesizing amino acid surfactants from other protein sources.Author contributions
Lara Wehbe: conceptualization, formal analysis, investigation, methodology, writing – original draft, visualization; Erwann Guénin: conceptualization, methodology, supervision, writing – review & editing; Alla Nesterenko: conceptualization, supervision, methodology, writing – review & editing, project administration, funding acquisition.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: Table S1 with the detail of Green Metrics calculation and Fig. S1–S8 with NMR spectra of obtained lipoamino acids. See DOI: https://doi.org/10.1039/d5gc06742a.Acknowledgements
This work was supported by the French National Research Agency (Agence Nationale de la Recherche-ANR), LIPOSILK project under grant number ANR-22-CE43-0007-01. The authors would like to thank the Service d'Analyse Physico-Chimique (SAPC) at the Université de technologie de Compiègne for the use of the FTIR and NMR facilities.References
- R. Bordes and K. Holmberg, Amino acid-based surfactants – do they deserve more attention?, Adv. Colloid Interface Sci., 2015, 222, 79–91 CrossRef CAS PubMed.
- M. R. Infante, L. Pérez, A. Pinazo, P. Clapés, M. C. Morán, M. Angelet, M. T. García and M. P. Vinardell, Amino acid-based surfactants, C. R. Chim., 2004, 7, 583–592 CrossRef CAS.
- D. B. Tripathy, A. Mishra, J. Clark and T. Farmer, Synthesis, chemistry, physicochemical properties and industrial applications of amino acid surfactants: A review, C. R. Chim., 2018, 21, 112–130 CrossRef CAS.
- X. Zhao, F. Pan, H. Xu, M. Yaseen, H. Shan, C. A. E. Hauser, S. Zhang and J. R. Lu, Molecular self-assembly and applications of designer peptide amphiphiles, Chem. Soc. Rev., 2010, 39, 3480–3498 RSC.
- C. Rondel, B. Portet, I. Alric, Z. Mouloungui, J.-F. Blanco and F. Silvestre, Green Production of Anionic Surfactant Obtained from Pea Protein, J. Surfactants Deterg., 2011, 14, 535–544 CrossRef CAS.
- C. C. Ruiz, Sugar-based ionic surfactants: Syntheses, solution properties, and applications, CRC Press, Boca Raton, 2008 Search PubMed.
- R. Sánchez-Vioque, C. L. Bagger, C. Rabiller and J. Guéguen, Foaming Properties of Acylated Rapeseed (Brassica napus L.) Hydrolysates, J. Colloid Interface Sci., 2001, 244, 386–393 CrossRef.
- A. O. Matemu, H. Kayahara, H. Murasawa, S. Katayama and S. Nakamura, Improved emulsifying properties of soy proteins by acylation with saturated fatty acids, Food Chem., 2011, 124, 596–602 CrossRef CAS.
- C. Vepari and D. L. Kaplan, Silk as a biomaterial, Prog. Polym. Sci., 2007, 32, 991–1007 CrossRef CAS PubMed.
- C. Belda Marín, V. Fitzpatrick, D. L. Kaplan, J. Landoulsi, E. Guénin and C. Egles, Silk Polymers and Nanoparticles: A Powerful Combination for the Design of Versatile Biomaterials, Front. Chem., 2020, 8, 604398 CrossRef PubMed.
- K. Tarangini, P. Kavi and K. J. Rao, Application of sericin-based edible coating material for postharvest shelf-life extension and preservation of tomatoes, Food, 2022, 3, 36 Search PubMed.
- I. M. Matran, C. Matran and M. Tarcea, Sustainable Prebiotic Dessert with Sericin Produced by Bombyx mori Worms, Sustainability, 2023, 15, 110 CrossRef CAS.
- J. P. Kumar and B. B. Mandal, Antioxidant potential of mulberry and non-mulberry silk sericin and its implications in biomedicine, Free Radical Biol. Med., 2017, 108, 803–818 CrossRef CAS PubMed.
- B. B. Mandal and S. C. Kundu, Self-assembled silk sericin/poloxamer nanoparticles as nanocarriers of hydrophobic and hydrophilic drugs for targeted delivery, Nanotechnology, 2009, 20, 355101 CrossRef PubMed.
- R. Patel and M. Modasiya, Sericin: Pharmaceutical Applications, Int. J. Res. Pharm. Biomed. Sci., 2011, 2, 913–917 Search PubMed.
- J. C. Anastas and P. T. Warner, Green Chemistry: Theory and Practice, Oxford University Press, Oxford U.K., 1998 Search PubMed.
- P. Anastas and N. Eghbali, Green Chemistry: Principles and Practice, Chem. Soc. Rev., 2009, 39, 301–312 RSC.
- G. Cave, C. Raston and J. Scott, Recent advances in solventless organic reactions: towards benign synthesis with remarkable versatility, Chem. Commun., 2001, 21, 2159–2169 RSC.
- A. A. Gečiauskaitė and F. García, Main group mechanochemistry, Beilstein J. Org. Chem., 2017, 13, 2068–2077 CrossRef PubMed.
- J. Andersen and J. Mack, Mechanochemistry and organic synthesis: From mystical to practical, Green Chem., 2018, 20, 1435–1443 RSC.
- E. Massolo, M. Pirola and M. Benaglia, Amide Bond Formation Strategies: Latest Advances on a Dateless Transformation, Eur. J. Org. Chem., 2020, 4641–4651 CrossRef CAS.
- E. Valeur and M. Bradley, Amide bond formation: beyond the myth of coupling reagents, Chem. Soc. Rev., 2009, 38, 606–631 RSC.
- J. G. Hernández, M. Turberg, I. Schiffers and C. Bolm, Mechanochemical Strecker Reaction: Access to α-Aminonitriles and Tetrahydroisoquinolines under Ball-Milling Conditions, Chem. – Eur. J., 2016, 22, 14513–14517 CrossRef PubMed.
- V. Declerck, P. Nun, J. Martinez and F. Lamaty, Solvent-Free Synthesis of Peptides, Angew. Chem., Int. Ed., 2009, 48, 9318–9321 CrossRef CAS PubMed.
- L. Konnert, A. Gauliard, F. Lamaty, J. Martinez and E. Colacino, Solventless Synthesis of N-Protected Amino Acids in a Ball Mill, ACS Sustainable Chem. Eng., 2013, 1, 1186–1191 CrossRef CAS.
- L. Konnert, F. Lamaty, J. Martinez and E. Colacino, Solventless Mechanosynthesis of N-Protected Amino Esters, J. Org. Chem., 2014, 79, 4008–4017 CrossRef CAS PubMed.
- T.-X. Métro, J. Bonnamour, T. Reidon, J. Sarpoulet, J. Martinez and F. Lamaty, Mechanosynthesis of amides in the total absence of organic solvent from reaction to product recovery, Chem. Commun., 2012, 48, 11781–11783 RSC.
- T.-X. Métro, J. Bonnamour, T. Reidon, A. Duprez, J. Sarpoulet, J. Martinez and F. Lamaty, Comprehensive Study of the Organic-Solvent-Free CDI-Mediated Acylation of Various Nucleophiles by Mechanochemistry, Chem. – Eur. J., 2015, 21, 12787–12796 CrossRef PubMed.
- T.-X. Métro, J. Martinez and F. Lamaty, 1,1′-Carbonyldiimidazole and Mechanochemistry: A Shining Green Combination, ACS Sustainable Chem. Eng., 2017, 5, 9599–9602 CrossRef.
- C. Duangkamol, S. Jaita, S. Wangngae, W. Phakhodee and M. Pattarawarapan, An efficient mechanochemical synthesis of amides and dipeptides using 2,4,6-trichloro-1,3,5-triazine and PPh3, RSC Adv., 2015, 5, 52624–52628 RSC.
- V. Porte, M. Thioloy, T. Pigoux, T.-X. Métro, J. Martinez and F. Lamaty, Peptide Mechanosynthesis by Direct Coupling of N-Protected α-Amino Acids with Amino Esters, Eur. J. Org. Chem., 2016, 3505–3508 CrossRef CAS.
- V. Štrukil, B. Bartolec, T. Portada, I. Đilović, I. Halasz and D. Margetić, One-pot mechanosynthesis of aromatic amides and dipeptides from carboxylic acids and amines, Chem. Commun., 2012, 48, 12100–12102 RSC.
- J. Bonnamour, T.-X. Métro, J. Martinez and F. Lamaty, Environmentally benign peptide synthesis using liquid-assisted ball-milling: application to the synthesis of Leu-enkephalin, Green Chem., 2013, 15, 1116–1120 RSC.
- L. Wehbe, R. Bascou, A. Nesterenko and E. Guénin, Green Strategy for Sustainable Silk Lipopeptide Production using Mechanochemistry, ACS Sustainable Chem. Eng., 2025, 13, 1882–1893 CrossRef CAS.
- J. Adams, C. Alder, I. Andrews, A. Bullion, M. Campbell-Crawford, M. Darcy, J. Hayler, R. Henderson, C. Oare, I. Pendrak, A. Redman, L. Shuster, H. Sneddon and M. Walker, Development of GSK's reagent guides – embedding sustainability into reagent selection, Green Chem., 2013, 15, 1542–1549 RSC.
- T. Stolar, J. Alić, G. Talajić, N. Cindro, M. Rubčić, K. Molčanov, K. Užarević and J. G. Hernández, Supramolecular intermediates in thermo-mechanochemical direct amidations, Chem. Commun., 2023, 59, 13490–13493 RSC.
- A. Bil, B. Abdellahi, G. Pourceau and A. Wadouachi, Fast and Efficient Mechanosynthesis of Aldonamides by Aminolysis of Unprotected Sugar Lactones, Sustainable Chem., 2022, 3, 300–311 CrossRef CAS.
- W. I. Nicholson, F. Barreteau, J. A. Leitch, R. Payne, I. Priestley, E. Godineau, C. Battilocchio and D. L. Browne, Direct Amidation of Esters by Ball Milling, Angew. Chem., Int. Ed., 2021, 60, 21868–21874 CrossRef CAS PubMed.
- R. Bascou, J. Hardouin, M. A. Ben Mlouka, E. Guénin and A. Nesterenko, Detailed investigation on new chemical-free methods for silk sericin extraction, Mater. Today Commun., 2022, 33, 104491 CrossRef CAS.
- R. Bascou, A. Flick, E. Guénin and A. Nesterenko, Development of lipopeptide surfactants from silk sericin and evaluation of their surface active properties, Colloids Surf., A, 2023, 678, 132460 CrossRef CAS.
- C. C. Goodno, H. E. Swaisgood and G. L. Catignani, A fluorimetric assay for available lysine in proteins, Anal. Biochem., 1981, 115, 203–211 CrossRef CAS PubMed.
- S. Fadlallah, P. S. Roy, G. Garnier, K. Saito and F. Allais, Are lignin-derived monomers and polymers truly sustainable? An in-depth green metrics calculations approach, Green Chem., 2021, 23, 1495–1535 RSC.
- G. Purohit and D. S. Rawat, Hierarchically Porous Mixed Oxide Sheetlike Copper–Aluminum Nanocatalyzed Synthesis of 2-Alkynyl Pyrrolidines/Piperidines and Their Ideal Green Chemistry Metrics, ACS Sustainable Chem. Eng., 2019, 7, 19235–19245 CrossRef CAS.
- R. A. Sheldon, The E factor 25 years on: the rise of green chemistry and sustainability, Green Chem., 2017, 19, 18–43 RSC.
- I. Slavchev, J. S. Ward, K. Rissanen, G. M. Dobrikov and S. Simeonov, Base-promoted direct amidation of esters: beyond the current scope and practical applications, RSC Adv., 2022, 12, 20555–20562 RSC.
- B. Kim, H.-G. Lee, S.-B. Kang, G. Sung, J.-J. Kim, J. Park, S.-G. Lee and Y.-J. Yoon, tert-Butoxide-Assisted Amidation of Esters under Green Conditions, Synthesis, 2012, 42–50 CAS.
- C. Rondel, I. Alric, Z. Mouloungui, J.-F. Blanco and F. Silvestre, Synthesis and Properties of Lipoamino Acid–Fatty Acid Mixtures: Influence of the Amphiphilic Structure, J. Surfactants Deterg., 2009, 12, 269–275 CrossRef CAS.
- A. Barth, Infrared spectroscopy of proteins, Biochim. Biophys. Acta, Bioenerg., 2007, 1767, 1073–1101 CrossRef CAS PubMed.
- Y. Zhang, Y. N. Feng and X. R. Wang, Study on Synthesis and Surface Activities of Silk Peptide-Based Surfactants, Adv. Mater. Res., 2011, 175–176, 755–759 CAS.
- M.-H. Wu, L.-Z. Wan and Y.-Q. Zhang, A novel sodium N-fatty acyl amino acid surfactant using silkworm pupae as stock material, Sci. Rep., 2014, 4, 4428 CrossRef PubMed.
- M. S. Zafar and K. H. Al-Samadani, Potential use of natural silk for bio-dental applications, J. Taibah Univ. Med. Sci., 2014, 9, 171–177 Search PubMed.
- D. L. Nelson and M. M. Cox, Lehninger Principles of Biochemistry, W. H. Freeman & Company, 4th edn, 2004, ch. 3 Search PubMed.
- J. Xia, J. Qian and I. A. Nnanna, Synthesis and Surface Properties of Amino Acid Surfactants from Industrial Waste Proteins, J. Agric. Food Chem., 1996, 44, 975–979 CrossRef CAS.
- A. Kadiri, T. Fergoug, K. O. Sebakhy, Y. Bouhadda, R. Aribi, F. Yssaad, Z. Daikh, M. E. H. El Nokab and P. H. M. Van Steenberge, Insights into the Characterization of the Self-Assembly of Different Types of Amphiphilic Molecules Using Dynamic Light Scattering, ACS Omega, 2023, 8, 47714–47722 CrossRef CAS PubMed.
- I. W. Hamley, A. Adak and V. Castelletto, Influence of chirality and sequence in lysine-rich lipopeptide biosurfactants and micellar model colloid systems, Nat. Commun., 2024, 15, 6785 CrossRef CAS PubMed.
- N. Fantozzi, J.-N. Volle, A. Porcheddu, D. Virieux, F. García and E. Colacino, Green metrics in mechanochemistry, Chem. Soc. Rev., 2023, 52, 6680–6714 RSC.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |