Open Access Article
Open Access ArticleSolvent-free/scalable mechanochemical-assisted addition of phosphine oxides to aldehydes: sustainable protocol for one-pot tandem P–C bond formation
Marcos López-Aguilar a,
Javier F. Reynes
a,
Javier F. Reynes b,
Luciana Cicco
b,
Luciana Cicco ac,
Alba Suárez-Fernándeza,
Vito Capriati
ac,
Alba Suárez-Fernándeza,
Vito Capriati c,
Alejandro Presa Soto
c,
Alejandro Presa Soto *a,
Felipe García
*a,
Felipe García *bd and
Joaquín García-Álvarez
*bd and
Joaquín García-Álvarez *a
*a
aLaboratorio de Química Sintética Sostenible (QuimSinSos), Departamento de Química Orgánica e Inorgánica, (IUQOEM), Centro de Innovación en Química Avanzada (ORFEO-CINQA), Facultad de Química, Universidad de Oviedo, E33071 Oviedo, Spain. E-mail: garciajoaquin@uniovi.es
bDepartamento de Química Orgánica e Inorgánica, Facultad de Química, Universidad de Oviedo, E33071 Oviedo, Spain. E-mail: garciafelipe@uniovi.es
cDipartimento di Farmacia–Scienze del Farmaco, Università di Bari “Aldo Moro”, Consorzio C.I.N.M.P.I.S., Via E. Orabona 4, I-70125 Bari, Italy
dSchool of Chemistry, Monash University, Clayton, Victoria 3800, Australia. E-mail: Felipe.Garcia@monash.edu
First published on 5th May 2026
Abstract
The development of greener and more efficient synthetic methodologies is a central goal in modern organic and main-group chemistry, particularly in the context of reducing solvent use, energy consumption and environmental impact. Mechanochemistry is introduced here as an efficient, cost-effective, solvent- and catalyst-free, and scalable synthetic tool for the chemoselective formation of P–C bonds with a markedly reduced environmental footprint. Using an atom-economical ball-milled protocol, the addition of a broad range of phosphine oxides [R2P(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)H] to aromatic, heteroaromatic, and aliphatic aldehydes cleanly affords the corresponding α-hydroxyphosphine oxides in excellent yields and with high functional-group tolerance, all performed at room temperature and under air/moisture without the need for an inert atmosphere. In addition, we have developed an unprecedented mechanochemical one-pot tandem protocol that enables the chemoselective synthesis of α-hydroxyphosphine oxides directly from primary alcohols, further expanding the efficiency and synthetic versatility of this solvent-free approach. These results highlight the potential of mechanochemistry to streamline main-group synthetic transformations and pave the way for future solvent-free strategies in sustainable phosphorus chemistry.
O)H] to aromatic, heteroaromatic, and aliphatic aldehydes cleanly affords the corresponding α-hydroxyphosphine oxides in excellent yields and with high functional-group tolerance, all performed at room temperature and under air/moisture without the need for an inert atmosphere. In addition, we have developed an unprecedented mechanochemical one-pot tandem protocol that enables the chemoselective synthesis of α-hydroxyphosphine oxides directly from primary alcohols, further expanding the efficiency and synthetic versatility of this solvent-free approach. These results highlight the potential of mechanochemistry to streamline main-group synthetic transformations and pave the way for future solvent-free strategies in sustainable phosphorus chemistry.
Green foundation1. Our work advances green chemistry by delivering a solvent- and catalyst-free mechanochemical protocol for chemoselective P–C bond formation and related tandem transformations under ambient conditions. It demonstrates that α-hydroxyphosphine oxides can be prepared at scale without VOCs, inert atmospheres, multistep solvent handling or energy-intensive purification, establishing a solid-state platform for sustainable main-group synthesis.2. The methodology exhibits markedly improved green metrics versus electrochemical, DES-based and solution-state routes, including lower E-factors, high atom economy, enhanced mass efficiency and energy reductions of up to 98.2%. Excellent isolated yields (up to 95%) are achieved without chromatography and scalability from 0.5 to 15 mmol is shown without loss of performance. 3. The process could be made even greener by improving jar material sustainability and expanding solvent-free tandem strategies toward continuous mechanochemical processing. |
Introduction
Organophosphorus compounds play a pivotal role in life-essential structures, being omnipresent across a wide range of biomolecules, from information-encoding biopolymers (e.g., DNA or RNA) to mediators for energy transfer in multiple metabolic pathways (e.g., ATP).1 Beyond their fundamental biological relevance, organophosphorus derivatives hold enormous importance in applied chemical disciplines, including pharmaceutical chemistry, the industrial production of pesticides and flame retardants, organic catalysis and materials science.2 Given this broad impact, the development of robust, efficient and sustainable synthetic methods for phosphorus–carbon (P–C) bond formation remains a long-standing challenge of both academic and industrial significance.Historically, synthetic strategies for constructing P–C bonds have prioritised chemoselectivity, functional-group tolerance, substrate scope and productivity; typically expressed through high yields and product purity.3 While these approaches have driven major advances in organophosphorus chemistry, they often overlook wider sustainability considerations. The introduction of Green Chemistry Principles has therefore prompted a paradigm shift in the field, promoting synthetic processes that: (i) operate under ambient, aerobic conditions with high energy efficiency; (ii) maximise atom economy; (iii) ensure safety for both humans and the environment; and (iv) avoid the use of non-renewable and hazardous volatile organic solvents (VOCs).4
Within this context, the addition of secondary phosphine oxides [R2P(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)H] to aldehydes (formally inserting the carbonyl moiety into the P–H bond) stands out as an ideal atom-economic transformation, delivering α-hydroxyphosphine oxides with complete mass efficiency.5 However, despite its conceptual simplicity, this reaction typically requires long reaction times (up to 24 h), high temperatures (up to 200 °C), acidic or basic catalysts, and harmful organic solvents such as benzene or dichloromethane, alongside purification steps involving significant solvent and energy use.5 These limitations highlight the need for methodologies that deliver the same transformation under milder, safer and more sustainable conditions (Scheme 1).
O)H] to aldehydes (formally inserting the carbonyl moiety into the P–H bond) stands out as an ideal atom-economic transformation, delivering α-hydroxyphosphine oxides with complete mass efficiency.5 However, despite its conceptual simplicity, this reaction typically requires long reaction times (up to 24 h), high temperatures (up to 200 °C), acidic or basic catalysts, and harmful organic solvents such as benzene or dichloromethane, alongside purification steps involving significant solvent and energy use.5 These limitations highlight the need for methodologies that deliver the same transformation under milder, safer and more sustainable conditions (Scheme 1).
 | ||
Scheme 1 Schematic comparison of previously reported solution based5i (a), microwave6b (b), electrochemical6c (c) and neat7b (d) approaches with the mechanochemical-assisted formation of P–C bonds via: (i) addition of secondary phosphine oxides [R2P(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)H] into aldehydes [route (e)]; or (ii) design of a one-pot tandem protocol which start from primary alcohols [route (f)]. O)H] into aldehydes [route (e)]; or (ii) design of a one-pot tandem protocol which start from primary alcohols [route (f)]. | ||
Several greener alternatives have recently emerged, including non-thermal activation strategies (ultrasound,6a microwave,6b or electrochemical oxidation6c) and methods employing environmentally friendly reaction media such as Deep Eutectic Solvents (DESs)7a or neat conditions.7b Although these approaches represent meaningful progress, many still rely on external energy input, catalysts or specific liquid media, and do not fully eliminate (or drastically reduce) the environmental burdens associated with solvent-based synthetic and purification operations.
In this landscape, mechanochemistry has rapidly emerged as a powerful, inherently sustainable synthetic platform.8 By enabling chemical transformations under mild, bench-top conditions and with little to no solvent, mechanochemistry offers substantial reductions in solvent waste, energy consumption and purification requirements.9 Thus, and over the past two decades, mechanochemistry has become an increasingly robust alternative to conventional solution-based thermochemistry across a wide range of chemical disciplines.9–17 Despite this progress, the application of mechanochemistry in p-block chemistry (particularly for P–C bond formation) has remained comparatively limited until recently.18 However, a growing body of work now highlights the rapid expansion of mechanochemistry in this area, although current studies are primarily focused on transformations such as deoxygenation or phosphorylation,19 rather than direct P–C bond formation via aldehyde addition. Thus, and to the best of our knowledge, no mechanochemical protocol has been reported for the direct, chemoselective addition of secondary phosphine oxides to aldehydes to afford α-hydroxyphosphine oxides under solvent- and catalyst-free conditions.18o
Building on our previous work involving: (i) the fast and chemoselective addition of in situ-generated lithium phosphides (LiPR2) to aldehydes and epoxides in Deep Eutectic Solvents under air at room temperature;20 and (ii) the synthesis of cyclodiphosphazane macrocycles and PNP ligands in both solution and the solid state;18b–e,21 we sought to extend our main-group and mechanochemical expertise into a new area of sustainable P–C bond formation.
Herein, we report the mechanochemical, chemoselective and atom-economic addition of phosphine oxides R2P(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)H to aldehydes, performed under neat conditions, at room temperature and in the absence of catalysts or protective atmospheres. This operationally simple protocol delivers α-hydroxyphosphine oxides in excellent yields (up to 95%). Moreover, we have evaluated its green chemistry metrics and, importantly, demonstrated its scalability to 15 mmol, reinforcing its sustainability and its readiness for preparative scales.
O)H to aldehydes, performed under neat conditions, at room temperature and in the absence of catalysts or protective atmospheres. This operationally simple protocol delivers α-hydroxyphosphine oxides in excellent yields (up to 95%). Moreover, we have evaluated its green chemistry metrics and, importantly, demonstrated its scalability to 15 mmol, reinforcing its sustainability and its readiness for preparative scales.
In addition, capitalising on the mildness and compatibility of this solvent-free protocol, we have developed an unprecedented mechanochemical one-pot tandem procedure that integrates P–C bond formation with the chemoselective Cu-catalysed oxidation of primary alcohols to aldehydes using aerial O2 as co-oxidant. This tandem process requires no intermediate purification and significantly reduces reaction time and energy consumption, and broaden the synthetic versatility of our approach.22a,b Considering the wide range of applications of R2P(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)-containing scaffolds, including: (i) phase-transfer catalysts;23 (ii) organocatalysts;24 (iii) ligands in metal-catalysed transformations;25 (iv) precursors to phosphorus-containing polymers;26 and (v) functional motifs in medicinal chemistry;27 the development of solvent-free, high-yielding and scalable routes to α-hydroxyphosphine oxides represents a timely and valuable contribution to sustainable main-group synthetic methodology.
O)-containing scaffolds, including: (i) phase-transfer catalysts;23 (ii) organocatalysts;24 (iii) ligands in metal-catalysed transformations;25 (iv) precursors to phosphorus-containing polymers;26 and (v) functional motifs in medicinal chemistry;27 the development of solvent-free, high-yielding and scalable routes to α-hydroxyphosphine oxides represents a timely and valuable contribution to sustainable main-group synthetic methodology.
Results and discussion
Mechanochemical-assisted formation of P–C bonds via chemoselective and atom-economic addition of secondary phosphine oxides R2P(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/h3_char_e001.gif) O)H (1a–e) into aldehydes (2a–p)
O)H (1a–e) into aldehydes (2a–p)
Taking into account all the aforementioned precedents, we decided to start our investigations by taking, as a model reaction, the operationally simple and mechanochemical-assisted direct reaction between equimolecular amounts of two commercially-available solids, namely diphenylphosphine oxide [Ph2P(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)H, 1a] and solid p-chlorobenzaldehyde (2a), working: (i) at 30 Hz; (ii) at room temperature; (iii) in the presence of air/moisture (no protecting atmosphere is needed); and (iv) in the absence of any external VOC solvent (neat conditions) or catalyst (see Table 1). Under these ball-milled conditions and by using both stainless-steel jar (5 ml) and ball (10 mm), the formation of the desired α-hydroxyphosphine oxide 3a occurs chemoselectively with no side products observed in the crude reaction mixture, as only unreacted starting materials 1a and 2a were detected by 1H or 31P{1H}-NMR. This total atom-economic mechanochemical-assisted addition reaction reaches good conversion after only 10 minutes (81%, entry 1, Table 1), requiring 60 minutes to increase the conversion up to 93% (entries 2 and 3, Table 1). The use of longer reaction times (70 minutes, entry 4, Table 1) did not improve the conversion into the desired α-hydroxyphosphine oxide 3a. Remarkably, the well-known over-oxidation of the starting aldehyde 2a into the corresponding p-chlorobenzoic acid, due to the presence of atmospheric O2 in the jar, was not observed under our ball-milled conditions.
O)H, 1a] and solid p-chlorobenzaldehyde (2a), working: (i) at 30 Hz; (ii) at room temperature; (iii) in the presence of air/moisture (no protecting atmosphere is needed); and (iv) in the absence of any external VOC solvent (neat conditions) or catalyst (see Table 1). Under these ball-milled conditions and by using both stainless-steel jar (5 ml) and ball (10 mm), the formation of the desired α-hydroxyphosphine oxide 3a occurs chemoselectively with no side products observed in the crude reaction mixture, as only unreacted starting materials 1a and 2a were detected by 1H or 31P{1H}-NMR. This total atom-economic mechanochemical-assisted addition reaction reaches good conversion after only 10 minutes (81%, entry 1, Table 1), requiring 60 minutes to increase the conversion up to 93% (entries 2 and 3, Table 1). The use of longer reaction times (70 minutes, entry 4, Table 1) did not improve the conversion into the desired α-hydroxyphosphine oxide 3a. Remarkably, the well-known over-oxidation of the starting aldehyde 2a into the corresponding p-chlorobenzoic acid, due to the presence of atmospheric O2 in the jar, was not observed under our ball-milled conditions.
| Entry | Hz | Jar (vol in mL) | Ball (size mm) | T (min) | Cb (%) 3a |
|---|---|---|---|---|---|
| a General conditions: reactions performed under air/moisture, at the desired frequency (30 to 15 Hz) in a Retsch MM400 ball mill, using 0.5 mmol of phosphine oxide 1a and p-chlorobenzaldehyde (2a).b Conversion determined by 31P{1H}-NMR. | |||||
| 1 | 30 | SS (5) | 10 | 10 | 81 |
| 2 | 30 | SS (5) | 10 | 30 | 88 |
| 3 | 30 | SS (5) | 10 | 60 | 93 |
| 4 | 30 | SS (5) | 10 | 70 | 93 |
| 5 | 30 | SS (10) | 10 | 60 | 71 |
| 6 | 30 | SS (10) | 12 | 60 | 49 |
| 7 | 30 | ZrO2 (10) | 10 | 60 | 96 |
| 8 | 30 | ZrO2 (10) | 12 | 60 | 98 |
| 9 | 30 | ZrO2 (10) | 12 (×2) | 60 | 98 |
| 10 | 30 | WC-SS (10) | 10 | 60 | 93 |
| 11 | 30 | WC-SS (10) | 12 | 60 | 98 |
| 12 | 25 | ZrO2 (10) | 12 | 60 | 92 |
| 13 | 20 | ZrO2 (10) | 12 | 60 | 91 |
| 14 | 15 | ZrO2 (10) | 12 | 60 | 88 |
Next, and taking into account previous examples in all-milled-assisted organic synthesis which demonstrated the pivotal role of both the nature and the size of the jar and the ball employed,9–17 we decided to extend our parameter optimisation studies to larger jars and balls, by using different combinations of jars (5–10 mL) and balls (10–12 mm; entries 5 and 6, Table 1). In both cases, we observed a clear erosion of the conversion into the final α-hydroxyphosphine oxide 3a (from 93 to 71–49%, respectively) when increasing both the volume of the jar and the size of the ball. Next, the chemical composition of the jar was also investigated, finding almost quantitative conversion into α-hydroxyphosphine oxide 3a (96–98%, entries 7 and 8, Table 1) when using a 10 mL zirconium dioxide jar and 10–12 mm ball. At this point, we should mention that similar results were obtained when using: (i) two balls in the mechanochemical synthesis (entry 9, Table 1); or (ii) a tungsten-carbide-steel-based jar (entries 10 and 11, Table 1). In addition, a series of control experiments were conducted to clarify the role of mechanochemical activation. Performing the reaction under identical conditions but in the absence of a milling ball resulted in poor mixing and the formation of a heterogeneous mixture of solids, leading to significantly diminished conversion (44% by 31P NMR), thus highlighting the importance of efficient mechanical energy transfer. In contrast, short-time manual grinding using a mortar and pestle (10 min) led to high conversion (89% by 31P NMR), confirming that manual mechanical activation is responsible for promoting the transformation, although this approach suffers from limited reproducibility and poor control over energy input. Finally, the gradual reduction of the ball-mill frequency (from 30 to 15 Hz) produces the expected concomitant diminution of conversion into the desired α-hydroxyphosphine oxide 3a (compare entries 12–14, Table 1). Overall, these experiments support that efficient mechanochemical activation (controlled impact and mixing) is key to achieving high conversion.
Based on the aforementioned reactivity studies, a plausible mechanism can be proposed. Thus, the reaction most likely proceeds via a classical Pudovik-type hydrophosphinylation, in which the starting secondary phosphine oxide Ph2P(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)H [1a; P(V)] is in equilibrium with its phosphinous acid tautomer Ph2P–OH [P(III)], the latter acting as the nucleophilic species that adds to the aldehyde, followed by proton transfer to afford the α-hydroxyphosphine oxide 3a. Under solvent-free mechanochemical conditions, this process is expected to be facilitated by efficient mixing, particle size reduction and the continuous generation of fresh reactive interfaces. Moreover, the role of the milling media should also be considered. In this sense, Colacino and co-workers have previously proposed, for related mechanochemical transformations involving imines and secondary phosphine oxides,18o that zirconium oxide surfaces may contribute through a surface-mediated or Lewis-acid-type activation, in addition to purely mechanical effects. Although our system involves direct addition to an aldehyde rather than an imine, this precedent provides a useful conceptual framework to rationalise the influence of mechanochemical activation and milling media in the present transformation. The absence of detectable side products and the clean conversion observed by NMR further support a direct nucleophilic addition pathway rather than alternative radical or oxidative mechanisms.
O)H [1a; P(V)] is in equilibrium with its phosphinous acid tautomer Ph2P–OH [P(III)], the latter acting as the nucleophilic species that adds to the aldehyde, followed by proton transfer to afford the α-hydroxyphosphine oxide 3a. Under solvent-free mechanochemical conditions, this process is expected to be facilitated by efficient mixing, particle size reduction and the continuous generation of fresh reactive interfaces. Moreover, the role of the milling media should also be considered. In this sense, Colacino and co-workers have previously proposed, for related mechanochemical transformations involving imines and secondary phosphine oxides,18o that zirconium oxide surfaces may contribute through a surface-mediated or Lewis-acid-type activation, in addition to purely mechanical effects. Although our system involves direct addition to an aldehyde rather than an imine, this precedent provides a useful conceptual framework to rationalise the influence of mechanochemical activation and milling media in the present transformation. The absence of detectable side products and the clean conversion observed by NMR further support a direct nucleophilic addition pathway rather than alternative radical or oxidative mechanisms.
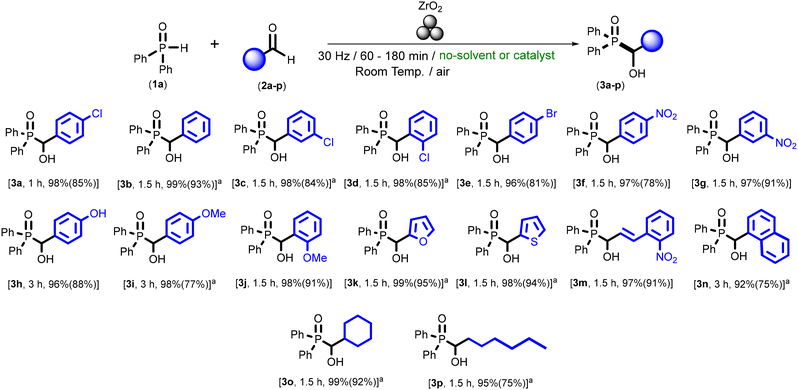
After setting up the best conditions for the mechanochemical-assisted addition of phosphine oxide 1a into solid p-chlorobenzaldehyde (2a; 30 Hz, rt, under air/moisture and using a ZrO2-based jar), we decided to extend our studies to a wide family of different aldehydes 2a–p (see Scheme 2). Firstly, and for the specific case of aromatic aldehydes 2a–j, we observed experimentally that our protocol is able to cope with: (i) the three possible different aromatic substitution (para- in 2a,e,f,h,i; meta- in 2c,g; or ortho- in 2d,j); and (ii) the presence of either electron-withdrawing [like, Cl (2a,c-d), Br (2e) or NO2 (2f,g)] or electron-donating-groups (OCH3, 2i,j). Furthermore, broad substrate compatibility is demonstrated with: (i) α,β-unsaturated systems (2m); (ii) heteroaromatic substrates bearing either furyl (2k) or thienyl (2l) substituents; and (iii) polycyclic aromatic hydrocarbons (e.g., 1-naphthaldehyde, 2n). Finally, it is important to mention that our system tolerates the use of purely aliphatic aldehydes, being compatible with both cyclic (2o) or linear (2p) backbones, thus clearly demonstrating the broad applicability of our methodology.
Moreover, it is worth highlighting that all the desired α-hydroxyphosphine oxides 3a–p were obtained with: (i) good to excellent yields (up to 95%); and (ii) in a total chemoselective manner, as no side products (aside from the unreacted phosphine oxide 1a and the employed aldehyde 2a–p) were observed when using our ball-milled protocol. Finally, we should mention that our mechanochemical assisted formation of P–C bond is also capable of working with liquid aldehydes 2b–d,i,k-l,n-p just by simple addition of NaCl as a solid grinding auxiliary.28 In these cases of liquid or low-melting aldehydes, the role of NaCl in this transformation goes beyond a simple additive, as it significantly influences the physical properties of the reaction medium. In this sense, NaCl acts as an inert solid dispersing agent, improving mixing and ensuring a more homogeneous distribution of the reactants throughout the milling process. By maintaining a free-flowing solid mixture, NaCl prevents the formation of agglomerated or paste-like phases that would otherwise dampen mechanical energy transfer, indicating that the additive improves the efficiency of mechanochemical activation rather than altering the intrinsic chemical reactivity. These effects are in line with the well-established role of inorganic grinding auxiliaries in mechanochemistry,29 where they primarily modulate rheology and energy transfer rather than directly affecting intrinsic chemical reactivity. Concerning product isolation, and in those cases where NaCl was used as a grinding auxiliary, the resulting α-hydroxyphosphine oxides were readily isolated and purified by a simple aqueous washing step followed by filtration to remove the NaCl additive, affording the desired compounds cleanly, supporting the high chemoselectivity of the transformation.30 In contrast, for solid products no purification was required, as the α-hydroxyphosphine oxides were obtained directly from the milling jar in high purity.
Next, and trying to have a full picture of the influence of the two components involved in our mechanochemical-assisted P–C bond formation protocol, we decided to evaluate the use of other phosphine oxides under our previously optimised reaction conditions (30 Hz, room temperature, under air/moisture and using a ZrO2-based jar; see Scheme 3).
Gratifyingly, we observed that mono-substituted (1b) aromatic phosphine oxides could be chemoselectively added (no by-products apart from the unreacted starting materials were observed) into different aromatic aldehydes, containing both electron-withdrawing (Cl, 2a) or electron-donating groups (MeO, 2i), thus giving access to the corresponding α-hydroxyphosphine oxides 3q–s in good to almost quantitative isolated yields (89–95%), although longer reaction times are needed. Nevertheless, di-substituted aromatic phosphines were tested, but mixtures were obtained after more than 12 hours of reaction time, and the final α-hydroxyphosphine oxides could not be isolated. In contrast, our system exhibits good tolerance towards purely aliphatic phosphine oxides (e.g., n-dibutyl phosphine oxide, 1c), delivering the desired α-hydroxyphosphine oxide 3t in 74% isolated yield.
Finally, to further expand the scope of our study, we explored the reactivity of other diphenylphosphine chalcogenides [Ph2(P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S)H (1d) and Ph2(P
S)H (1d) and Ph2(P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Se)H (1e) derivatives] under the same optimised mechanochemical conditions. In these cases, the reactions proceeded smoothly with p-chloro- (2a) and m-nitrobenzaldehyde (2g), both bearing electron-withdrawing substituents, as well as with the α,β-unsaturated aldehyde (2m) in the case of sulfur. In all instances, the corresponding α-hydroxyphosphine sulfides (3u–w) and α-hydroxyphosphine selenides (3x,y) were obtained in good to excellent isolated yields (66–90%), demonstrating that our mechanochemical P–C bond formation is also applicable to other chalcogen-containing phosphine oxides beyond oxygen.
Se)H (1e) derivatives] under the same optimised mechanochemical conditions. In these cases, the reactions proceeded smoothly with p-chloro- (2a) and m-nitrobenzaldehyde (2g), both bearing electron-withdrawing substituents, as well as with the α,β-unsaturated aldehyde (2m) in the case of sulfur. In all instances, the corresponding α-hydroxyphosphine sulfides (3u–w) and α-hydroxyphosphine selenides (3x,y) were obtained in good to excellent isolated yields (66–90%), demonstrating that our mechanochemical P–C bond formation is also applicable to other chalcogen-containing phosphine oxides beyond oxygen.
Comparative assessment of the environmental footprint
As previously mentioned, sustainability and environmental factors have become increasingly important in designing new synthetic protocols, which were traditionally planned solely to maximise strictly synthetic parameters (selectivity and functional group tolerance) or productivity.4 In this sense, the use of Green Chemistry Metrics (GCM)31 is nowadays mandatory to clearly determine which is the real green and sustainable advance of a new synthetic protocol. Moreover, in the specific case of main-group synthesis, the number of previously reported examples that provide a detailed comparison between mechanochemical and conventional solution-based methodologies is scarce.18n,32 Green Chemistry Metrics (GCM) were calculated for the α-hydroxyphosphine oxide compounds 3a–h,j-l,o. It should be noted that these calculations were restricted exclusively to the aldehyde-to-α-hydroxybenzyl step of the previously mentioned α-hydroxyphosphine oxide compounds, since extending the analysis from the alcohol (tandem protocol) would introduce significant methodological variability, thus hindering the interpretation of results and undermining the primary purpose of this section, which is to provide an illustrative and coherent analysis of GCM application in the context of this study.For the calculation of the GCM, we decided to compare (see Fig. 1) our new mechanochemical-assisted P–C formation protocol with a recently reported (2023) electrochemically oxidative phosphating of aldehydes and ketones to generate α-hydroxyphosphine oxides in solution,6c except for compounds 3c and 3f,h,j, that were not reported by this method and were compared using a DES-assisted (Deep Eutectic Solvent) addition protocol20 and a conventional solution-based method, respectively,5i notably demonstrating that our synthetic approach consistently delivers improved sustainability metrics, under the conditions evaluated for all calculated GCM. At this point, it is also worth noting that the crude products obtained from the electrochemical oxidative process6c were purified by column chromatography (petroleum ether/ethyl acetate = 2/1, v/v). In contrast, those from the conventional solution-based reaction5i were purified by recrystallisation in THF/methanol. These purification steps were not included in the GCM calculations because the authors did not provide quantitative data for the amounts used. However, it is important to emphasise that the actual GCM values for those processes would likely be even less favourable if purification were considered.
Although the environmental impact of a synthetic method is a key criterion, a meaningful sustainability assessment must also account for energy demand and operational costs. To complement the aforementioned mass-based Green Chemistry Metrics, we decided to also compare the energy efficiency and cost implications of our mechanochemical P–C bond formation with those of the corresponding conventional protocols. This provides a more complete picture of the practical advantages associated with the solvent-free, room-temperature mechanochemical process.
Energy usage was also quantified by combining the power rating of each device with the corresponding operation time, assuming negligible consumption during idle periods (see SI). On a 0.1 g scale, the mechanochemical protocol employed for the synthesis of 3a–l,o requires 2250 kW h kg−1. In contrast, the electrochemically oxidative route consumes 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 kW h kg−1, while the DES protocol for 3c and the conventional solution-based methods for 3f, 3h and 3j require 4000 kW h kg−1 and 90
400 kW h kg−1, while the DES protocol for 3c and the conventional solution-based methods for 3f, 3h and 3j require 4000 kW h kg−1 and 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 960–122
960–122![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 kW h kg−1, respectively (Table 2 and SI Table S1).
000 kW h kg−1, respectively (Table 2 and SI Table S1).
| Compound | Energy consumptionb kW h kg−1 (MJ kg−1) | Energy savedc (%) | Approx. electricity cost in averaged (€ kg−1) | |
|---|---|---|---|---|
| Household consumer | Non-household consumer | |||
| a To complement the mass-based green chemistry metrics, a quantitative assessment of the energy demand associated with each synthetic methodology was carried out.b The energy consumption was estimated using a standardized approach based on the nominal power of the equipment and the corresponding operation time, assuming negligible energy consumption during idle periods (see SI for full details). The total energy input was calculated according to: E = P·t where P is the power rating of the device (kW) and t is the reaction time (h). To enable comparison between different methodologies, the energy consumption was normalized to the mass of product obtained using the formula: Enorm = (P·t)/mproduct. The normalized values were converted into MJ kg−1 using the relationship: 1 kWh = 3.6 MJ.c The percentage of energy savings for the mechanochemical protocol relative to alternative methods was calculated as: % Energy saved = [(Ebenchmark − Emechanochemical)/Ebenchmark] × 100.d The associated electricity cost was estimated by: cost = Enorm·(electricity tariff) where average European electricity prices were used (see SI for full details). All calculations were derived from laboratory-scale experiments and extrapolated to a per-kilogram basis, providing a consistent comparative framework for evaluating the energy efficiency of the different synthetic routes. | ||||
| Mechanochemistry method | ||||
| 3a–3l, 3o | 2250 (8100) | 593.6 | 315.7 | |
| Electrochemically oxidative method | ||||
| 3a–3b, 3d–3e, 3g, 3i, 3k, 3l, 3o | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 (73 400 (73![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 440) 440) |
88.9 | 5382.3 | 2862.1 |
| Deep Eutectic Solvent (DES) method | ||||
| 3c | 4000 (14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400) 400) |
43.7 | 1042.9 | 561.2 |
| Conventional solution-based method | ||||
| 3f | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 960 (327 960 (327![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 456) 456) |
97.5 | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 557.9 557.9 |
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 761.8 761.8 |
| 3h | 96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 640 (347 640 (347![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 904) 904) |
97.7 | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 497.3 497.3 |
13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 558.6 558.6 |
| 3j | 122![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 (439 000 (439![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200) 200) |
98.2 | 32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 188.2 188.2 |
17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 116.6 116.6 |
These values translate into energy savings of 88.9% for most electrochemical comparisons (3a–3b, 3d–3e, 3g, 3i, 3k, 3l, 3o), 43.7% versus the DES protocol for 3c, and 97.5–98.15% with respect to the conventional solution-based syntheses of 3f, 3h and 3j (Table 2), indicating that the mechanochemical route is intrinsically less energy intensive on a per-kilogram basis. Although these values are extrapolated from small-scale experiments, the consistent methodology applied across all systems ensures meaningful comparative outcomes.
To assess the economic implications of these differences, approximate electricity costs were estimated using average 2021–2022 EU tariffs for medium-sized household and non-household consumers (Table 2 and SI Tables S2–S4). For household tariffs, the projected energy cost of producing 1 kg of 3a–l,o by mechanochemistry ranges from 440 to 720 €, depending on the country, whereas the corresponding conventional solution-based routes lie in the range from 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 to 30
000 to 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 € kg−1 (SI Table S3). For non-household tariffs, mechanochemical production is estimated between 190 and 350 € kg−1, compared to the 7700–19
000 € kg−1 (SI Table S3). For non-household tariffs, mechanochemical production is estimated between 190 and 350 € kg−1, compared to the 7700–19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 € kg−1 for the solution-based protocols (SI Table S4). Although these projections constitute a proof of concept derived from scaling up 0.1 g laboratory data, they clearly show that the solvent-free mechanochemical process affords a substantial reduction in energy-related operating costs relative to the benchmark methods. Nevertheless, as our estimations represent only a proof of concept based solely on energy consumption, additional costs associated with solvent handling, transport, recycling, or disposal have not been considered.
000 € kg−1 for the solution-based protocols (SI Table S4). Although these projections constitute a proof of concept derived from scaling up 0.1 g laboratory data, they clearly show that the solvent-free mechanochemical process affords a substantial reduction in energy-related operating costs relative to the benchmark methods. Nevertheless, as our estimations represent only a proof of concept based solely on energy consumption, additional costs associated with solvent handling, transport, recycling, or disposal have not been considered.
Upscaled synthesis of α-hydroxyphosphine oxide 3a
Nowadays, one of the major challenges to solve in the field of mechanochemical synthetic methodologies is the possibility to be implemented at large preparative scales,33 which would enlarge the synthetic applicability of ball-milled protocols, especially in the field related with main-group chemistry, in which the upscaling of synthetic protocols, thanks to the implementation of mechanochemically-assisted synthetic tools, is still in its infancy.18n,32 Thus, and based on this premise, we decided to set up an upscaled synthetic route to compound 3a. Therefore, a 25 mL jar (equipped with two 12 mm balls) was used to produce 3a (in a 15 mmol scale, 4.73 g) by milling solid diphenylphosphine oxide [Ph2P(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)H, 1a, 3.03 g] and solid p-chlorobenzaldehyde (2a, 2.10 g), in the absence of any external VOC solvents or co-catalysts, and working at 30 Hz, in the presence of air/moisture and at room temperature (Fig. 2). To our delight, this scale-up reaction (from 0.5 to 15 mmol; 30 times larger) was successfully completed after only 90 minutes, with no increase in reaction time compared to the 0.5 mmol scale. Under these conditions, the desired α-hydroxyphosphine oxide 3a was obtained in a remarkable 92% isolated yield, even surpassing the yield achieved at smaller scale. This result clearly highlights the robustness, scalability and practical applicability of the developed mechanochemical protocol.
O)H, 1a, 3.03 g] and solid p-chlorobenzaldehyde (2a, 2.10 g), in the absence of any external VOC solvents or co-catalysts, and working at 30 Hz, in the presence of air/moisture and at room temperature (Fig. 2). To our delight, this scale-up reaction (from 0.5 to 15 mmol; 30 times larger) was successfully completed after only 90 minutes, with no increase in reaction time compared to the 0.5 mmol scale. Under these conditions, the desired α-hydroxyphosphine oxide 3a was obtained in a remarkable 92% isolated yield, even surpassing the yield achieved at smaller scale. This result clearly highlights the robustness, scalability and practical applicability of the developed mechanochemical protocol.
 | ||
| Fig. 2 Milling jars used: (Left) Restch 10 mL ZrO2 jar for 0.5 mmol scale. (Right) Restch 25 mL ZrO2 jar for 15 mmol scale. | ||
Design of one-pot tandem transformation based on the mechanochemical-assisted and chemoselective formation of P–C bonds through the addition of phosphine oxides into aldehydes
During the last decade, there has been a notable increase of interest within the synthetic chemistry community towards the development of multistep one-pot tandem protocols,22a,b since the implementation of these methodologies avoids the need for halfway isolation/purification steps of reaction intermediates, which are typically required in tedious and traditional step-by-step synthetic procedures, allowing moreover to: (i) reduce the chemical waste and energy/time costs (in clear consonance with the Principles of Green Chemistry);4 and (ii) simplify the practical aspects of the desired synthetic methodology. While these tandem protocols have proven successful in generating a diverse array of natural products and intricate molecular structures under traditional thermal conditions,34 in the field of main-group chemistry there are currently only a limited number of mechanochemical-assisted variants that operate without VOC solvents or catalysts and can be performed under simple bench-type reaction conditions (room temperature, open to air and moisture).22c Thus, and taking into account our previous experience in the design of one-pot tandem protocols involving main-group reagents,35 and our knowledge in the chemoselective Cu(II)-catalysed oxidation of terminal alcohols into aldehydes,35g,36 we decided to design (as a proof-of concept), a new mechanochemical hybrid one-pot tandem protocol37 for the direct, straightforward and chemoselective conversion of primary alcohols 4a,c-d,f,i,l into the desired α-hydroxyphosphine oxide 3a,c-d,f,i,l,q under ball-milled conditions (30 Hz) working at room temperature and in the presence of air/moisture (see Scheme 4). Here, it is important to note that, to the best of our knowledge, the combination of transition-metal catalysis and main-group chemistry under mechanochemical conditions has not been reported.Thus, we firstly study the catalytic activity of the oxidative system CuCl2/TMEDA/TEMPO [TMEDA = N,N,N′,N′-tetramethylethylenediamine; TEMPO = (2,2,6,6-tetramethylpiperidin-1-yl)oxyl] to promote the chemoselective oxidation of p-chlorobenzyl alcohol 4a into the desired aldehyde 2a, under conditions parametrised, that is working: (i) under solvent-free conditions; (ii) at 20 Hz and at room temperature; and (iii) in the presence of air/moisture (see Scheme 4). Pleasingly, we observed quantitative and chemoselective conversion (the formation of the corresponding p-chlorobenzoic acid was not detected) into aldehyde 2a after 2 hours of reaction by using simple aerial O2 as external co-oxidant, thus generating H2O as the sole by-product of the oxidation protocol.38 Importantly, our oxidative system is also able to cope with benzylic alcohols containing electron-donating groups (4i, OMe) or even heteroaromatic primary alcohol (4l), observing in all cases quantitative and chemoselective conversion into corresponding aldehydes 2a,c-d,f,i,l (1H NMR analysis). Once the conversions of primary alcohols 4a,c-d,f,i,l into the desired aldehydes 2a,c-d,f,i,l was completed, the desired solid phosphine oxide (1a–b) was directly added to the jar under air (no isolation/purification of aldehydes 2a,c-d,f,i,l was needed), which was subsequently ball-milled (30 Hz, room temperature and in the absence of VOC solvents) for 180 minutes. Remarkably, and by using our unprecedented hybrid one-pot tandem protocol, the starting primary alcohols 4a,c-d,f,i,l could be chemoselectively and directly converted into the desired α-hydroxyphosphine oxide 3a,c-d,f,i,l,q in yields up to 75% (Scheme 4).
Conclusions
In summary, we present a pioneering mechanochemical approach for the atom-economic, chemoselective and environmentally friendly synthesis of α-hydroxyphosphine oxides, operating under solvent- and catalyst-free conditions at room temperature and in the presence of air/moisture. The calculation of Green Chemistry Metrics (GCMs) and scalability experiments clearly demonstrate the sustainable advantages of our P–C bond-forming protocol. Importantly, we have also designed an unprecedented mechanochemical one-pot tandem process that combines this P–C bond formation with the Cu-catalysed oxidation of primary alcohols to aldehydes using aerial O2 as co-oxidant.Finally, it is important to highlight that our solvent-free P–C bond-forming methodologies surpass traditional thermal and solution-based approaches reported over the past decades, delivering rapid, high-yielding and environmentally benign transformations that represent a significant milestone in the development of mechanochemical methods within main-group chemistry. Importantly, the combined reduction in solvent use, energy demand and purification requirements positions this methodology as a practical and scalable alternative for sustainable P–C bond formation.
We hope that this work encourages the development of further main-group-mediated green synthetic protocols through the broader adoption of mechanochemistry as a superior tool in organic synthesis. Looking ahead, we hope that these results stimulate new solvent-free strategies and promote deeper integration of mechanochemical techniques into sustainable phosphorus chemistry, main group chemistry and beyond.
Conflicts of interest
There are no conflicts to declareData availability
The data supporting this article have been included as part of the supplementary information (SI). The Supplementary Information contains detailed experimental procedures, characterisation data (1H, 13C and 31P NMR spectra), additional optimisation studies, green chemistry metrics calculations, and energy consumption analyses. Supplementary information is available. See DOI: https://doi.org/10.1039/d5gc06683b.Acknowledgements
ML-A, JFR, ASF, APS, FG and JGA thank financial support from: (i) MCIN/AEI/10.13039/501100011033 (project numbers PID2020-113473GB-100, PID2021-122763NB-I00 and PID2023-148663NB-I00); and (ii) “Programa de Subvenciones para grupos de investigación de organismos del Principado de Asturias” [Project Química Inorgánica y Catálisis (QUIMINORCAT); ref.: IDE/2024/000727. MLA acknowledges FICyT for funding (grant PA-22-BP21-088). VC gratefully acknowledges the Ministero dell'Università e della Ricerca (MUR) for supporting the National PRIN2022 project MENDELEEV (Grant No. 2022KMS84P), funded by the European Union – NextGenerationEU under the PNRR Mission 4, Component 2, Investment 1.1 (CUP H53D23004580006). Additional support from the University of Bari Aldo Moro and the CINMPIS Consortium is also warmly acknowledged. LC thanks the University of Bari Aldo Moro for awarding an Erasmus fellowship to conduct research at the University of Oviedo. FG thanks Monash University for financial support. The authors also acknowledge the technical support provided by SCTIs from Universidad de Oviedo.References
- (a) D. Watson and F. H. C. Crick, Nature, 1953, 171, 737–738 CrossRef PubMed; (b) E. Gajewski, D. K. Steckler and R. N. Goldberg, J. Biol. Chem., 1986, 261, 12733–12737 CrossRef CAS PubMed.
- For books/reviews covering this topic, see: (a) M. Peruzzini and L. Gonsalvi, Phosphorus Compounds Advanced Tools in Catalysis and Material Sciences, in Catalysis by Metal Complexes Series, 37, ed. C. Bianchini, D. J. Cole-Hamilton and P. W. N. M. van Leeuwen, Springer, Dordrecht, Heidelberg, London, New York, 2011 CrossRef; (b) J. B. Rodríguez and C. Gallo-Rodríguez, ChemMedChem, 2019, 14, 190–216 CrossRef PubMed; (c) C. Xie, A. J. Smaligo, X.-R. Song and O. Kwon, ACS Cent. Sci., 2021, 7, 536–558 CrossRef CAS PubMed; (d) S. P.-M. Ung and C.-H. Li, RSC Sustainability, 2023, 1, 11–37 RSC.
- (a) R. Engel and J. I. Coen, Synthesis of Carbon-Phosphorus Bonds, CRC Press, Boca Raton, 2nd edn, 2004 Search PubMed; (b) A. L. Schwan, Chem. Soc. Rev., 2004, 33, 218–224 RSC; (c) J. E. Borger, A. W. Ehlers, J. C. Slootweg and K. Lammertsma, Chem. – Eur. J., 2017, 23, 11738–11746 CrossRef CAS PubMed; (d) D. S. Gluek, J. Org. Chem., 2020, 85, 14276–14285 CrossRef PubMed.
- (a) P. T. Anastas and J. C. Warner, Green Chemistry Theory and Practice, Oxford University Press, Oxford, 1998 Search PubMed; (b) A. S. Matlack, Introduction to Green Chemistry, Marcel Dekker, New York, 2001 Search PubMed; (c) M. Poliakoff, J. M. Fitzpatrick, T. R. Farren and P. T. Anastas, Science, 2002, 297, 807 CrossRef CAS PubMed; (d) M. Lancaster, Green Chemistry: An Introductory Text, RSC Publishing, Cambridge, 2002 RSC“United Nations Sustainable Development Goals” can be found under https://sdgs.un.org/goals. “Chemistry & Sustainable Development Goals” can be found under https://www.acs.org/content/acs/en/sustainability/chemistry-sustainable-development-goals.html.
- (a) N. Gusarova, A. M. Reutskaya, N. I. Ivanova, A. S. Medvedeva, M. M. Demina, P. S. Novopashin, A. V. Afonin, A. I. Albanov and B. A. Trofimov, J. Organomet. Chem., 2002, 659, 172–175 CrossRef CAS; (b) N. K. Gusarova, S. N. Arbuzova, A. M. Reutskaya, N. I. Ivanova, L. V. Baikalova, L. M. Sinegovskaya, N. N. Chipanina, A. V. Afonin and I. A. Zyryanova, Chem. Heterocycl. Compd., 2002, 38, 65–70 CrossRef CAS; (c) N. I. Ivanova, A. M. Reutskaya, N. K. Gusarova, S. A. Medvedeva, A. V. Afonin, I. A. Ushakov, A. A. Tatarinova and B. A. Trofimov, Russ. J. Gen. Chem., 2003, 73, 1354–1357 CrossRef CAS; (d) N. I. Ivanova, N. K. Gusarova, E. A. Nikitina, A. I. Albanov, L. M. Sinegovskaya, M. V. Nikitin, N. A. Konovalova and B. A. Trofimov, Phosphorus, Sulfur Silicon Relat. Elem., 2004, 179, 7–18 CrossRef CAS; (e) N. I. Ivanova, N. K. Gusarova, E. A. Nikitina, S. A. Medvedeva, M. L. Al'pert and B. A. Trofimov, Chem. Heterocycl. Compd., 2004, 40, 426–429 CrossRef CAS; (f) S. Priya, M. S. Balakrishna and S. M. Mobin, Polyhedron, 2005, 24, 1641–1650 CrossRef CAS; (g) N. I. Ivanova, P. A. Volkov, N. K. Gusarova, L. I. Larina and B. A. Trofimov, J. Gen. Chem., 2011, 81, 315–321 CrossRef CAS; (h) X. Luo, Z. Zhou, X. Li, X. Liang and J. Ye, RSC Adv., 2011, 1, 698–705 RSC; (i) J. Zheng, X. Feng, Y. Yu, X. Zhen and Y. Zhao, Phosphorus, Sulfur Silicon Relat. Elem., 2013, 188, 1080–1087 CrossRef CAS; (j) D. Das and D. Seidel, Org. Lett., 2013, 15, 4358–4361 CrossRef CAS PubMed; (k) F. A. Kortmann, M.-C. Chang, E. Otten, E. P. A. Couzijn, M. Lutz and A. J. Minnaard, Chem. Sci., 2014, 5, 1322–1327 RSC.
- (a) M. Xia and Y. D. Lu, Ultrason. Sonochem., 2007, 14, 235–240 CrossRef CAS PubMed; (b) G. Keglevich, V. R. Tóth and L. Drahos, Heteroat. Chem., 2011, 22, 15–17 CrossRef CAS; (c) Y. Ma, X. Zhang, C. Ma, W. Xia, L. Hu, X. Dong and Y. Xiong, J. Org. Chem., 2023, 88, 4264–4272 CrossRef CAS PubMed.
- (a) S. Mandal, R. Narvariya, S. L. Sunar, I. Paul, A. Jain and T. K. Panda, Green Chem., 2023, 25, 8266–8272 RSC; (b) N. K. Gusarova, N. I. Ivanova, P. A. Volkov, K. O. Khrapova, L. I. Larina, V. I. Smirnov, T. N. Borodina and B. A. Trofimov, Synthesis, 2015, 1611–1622 CAS.
- Mechanochemistry and Emerging Technologies for Sustainable Chemical Manufacturing, ed. E. Colacino and F. García, CRC Press, Boca Raton, 1st Ed., 2023 Search PubMed.
- (a) S. L. James, C. J. Adams, C. Bolm, D. Braga, P. Collier, T. Friščić, F. Grepioni, K. D. Harris, G. Hyett and W. Jones, Chem. Soc. Rev., 2012, 41, 413–447 RSC; (b) J.-L. Do and T. Friščić, ACS Cent. Sci., 2017, 3, 13–19 CrossRef CAS PubMed; (c) T. Friščić, C. Mottillo and H. M. Titi, Angew. Chem., Int. Ed., 2020, 59, 1030–1041 CrossRef.
- (a) V. Declerck, P. Nun, J. Martinez and F. Lamaty, Angew. Chem., Int. Ed., 2009, 48, 9318–9321 CrossRef CAS PubMed; (b) J. G. Hernández and C. Bolm, J. Org. Chem., 2017, 82, 4007–4019 CrossRef PubMed; (c) D. Virieux, F. Delogu, A. Porcheddu, F. García and E. Colacino, J. Org. Chem., 2021, 86, 13885–13894 CrossRef CAS PubMed; (d) F. Cuccu, L. De Luca, F. Delogu, E. Colacino, N. Solin, R. Mocci and A. Porcheddu, ChemSusChem, 2022, 15, e202200362 CrossRef CAS PubMed.
- (a) L. Konnert, B. Reneaud, R. M. de Figueiredo, J.-M. Campagne, F. Lamaty, J. Martinez and E. Colacino, J. Org. Chem., 2014, 79, 10132–10142 CrossRef CAS PubMed; (b) D. Tan, L. Loots and T. Friščić, Chem. Commun., 2016, 52, 7760–7781 RSC; (c) P. Ying, J. Yu and W. Su, Adv. Synth. Catal., 2021, 363, 1246–1271 CrossRef CAS; (d) O. Galant, G. Cerfeda, A. S. McCalmont, S. L. James, A. Porcheddu, F. Delogu, D. E. Crawford, E. Colacino and S. Spatari, ACS Sustainable Chem. Eng., 2022, 10, 1430–1439 CrossRef CAS.
- (a) N. R. Rightmire and T. P. Hanusa, Dalton Trans., 2016, 45, 2352–2362 RSC; (b) F. Leon and F. Garcia, Comprehensive Coordination Chemistry III, 2021, vol. 9, pp. 620–679 Search PubMed.
- (a) A. A. Gečiauskaitė and F. García, Beilstein J. Org. Chem., 2017, 13, 2068–2077 CrossRef PubMed; (b) D. Tan and F. García, Chem. Soc. Rev., 2019, 48, 2274–2292 RSC.
- (a) T. Friščić, J. Mater. Chem., 2010, 20, 7599–7605 RSC; (b) P. A. Julien, C. Mottillo and T. Friščić, Green Chem., 2017, 19, 2729–2747 RSC; (c) D. Chen, J. Zhao, P. Zhang and S. Dai, Polyhedron, 2019, 162, 59–64 CrossRef CAS; (d) T. Stolar and K. Užarevíc, CrystEngComm, 2020, 22, 4511–4525 RSC.
- V. K. Portnoi, V. A. Leonov, S. E. Filippova, A. I. Logacheva, A. G. Beresnev and I. M. Razumovskii, Inorg. Mater., 2016, 52, 895–901 CrossRef CAS.
- (a) T. F. Grigorieva, A. P. Barinova and N. Z. Lyakhov, J. Nanopart. Res., 2003, 5, 439–453 CrossRef CAS; (b) B. Szczesniak, S. Borysiuk, J. Choma and M. Jaroniec, Mater. Horiz., 2020, 7, 1457–1473 RSC.
- (a) D. Braga, L. Maini and F. Grepioni, Chem. Soc. Rev., 2013, 42, 7638–7648 RSC; (b) M. M. Haskins and M. J. Zaworotko, Cryst. Growth Des., 2021, 21, 4141–4150 CrossRef CAS PubMed; (c) Z. X. Ng, D. Tan, W. L. Teo, F. Leon, X.-Y. Shi, Y. Sim, Y. Li, R. Ganguly, Z. Yanli, M. Sharmarke and F. García, Angew. Chem., Int. Ed., 2021, 60, 17481–17490 CrossRef CAS PubMed.
- For recent examples on the use of mechanochemistry in the field of main-group chemistry, see: (a) N. R. Rightmire, T. P. Hanusa and A. L. Rheingold, Organometallics, 2014, 33, 5952–5955 CrossRef CAS; (b) J. Wang, R. Ganguly, L. Yongxin, J. Díaz, H. S. Soo and F. García, Dalton Trans., 2016, 45, 7941–7946 RSC; (c) Y.-X. Shi, K. Xu, J. Clegg, R. Ganguly, H. Hirao, T. Friščić and F. García, Angew. Chem., Int. Ed., 2016, 55, 12736–12740 CrossRef CAS PubMed; (d) Y. Sim, Y.-X. Shi, R. Ganguly, Y. Li and F. García, Chem. – Eur. J., 2017, 47, 11279–11285 CrossRef PubMed; (e) R. F. Koby, T. P. Hanusa and N. D. Schley, J. Am. Chem. Soc., 2018, 140, 15934–15942 CrossRef CAS PubMed; (f) Y. Sim, D. Tan, R. Ganguly, Y. Li and F. García, Chem. Commun., 2018, 54, 6800–6803 RSC; (g) I. R. Speight, S. C. Chmely, T. P. Hanusa and A. L. Rheingold, Chem. Commun., 2019, 55, 2202–2205 RSC; (h) I. R. Speight and T. P. Hanusa, Molecules, 2020, 25, 570 CrossRef CAS PubMed; (i) R. F. Koby, A. M. Doerr, N. R. Rightmire, N. D. Schley, B. K. Long and T. P. Hanusa, Angew. Chem., Int. Ed., 2020, 59, 9542–9548 CrossRef CAS PubMed; (j) R. Takahashi, A. Hu, P. Gao, Y. Gao, Y. Pang, T. Seo, J. Jiang, S. Maeda, H. Takaya, K. Kubota and H. Ito, Nat. Commun., 2021, 12, 6691 CrossRef CAS PubMed; (k) R. F. Koby, A. M. Doerr, N. R. Rightmire, N. D. Schley, W. W. Brennessel, B. K. Long and T. P. Hanusa, Chem. – Eur. J., 2021, 27, 8195–8202 CrossRef CAS PubMed; (l) R. F. Koby, N. D. Schley and T. P. Hanusa, Angew. Chem., Int. Ed., 2021, 60, 21174–21178 CrossRef CAS PubMed; (m) D. Jedrzkiewicz, J. Mai, J. Langer, Z. Mathe, N. Patel, S. DeBeer and S. Harder, Angew. Chem., Int. Ed., 2022, 61, e202200511 CrossRef CAS PubMed; (n) P. Gao, J. Jiang, S. Maeda, K. Kubota and H. Ito, Angew. Chem., Int. Ed., 2022, 61, e202207118 CrossRef CAS PubMed; (o) J. A. Cabeza, J. F. Reynes, F. García, P. García-Álvarez and R. García-Soriano, Chem. Sci., 2023, 14, 12477–12483 RSC. For a recent and elegant mechanochemical-promoted Kabachnik–Fields domino reaction which allows the synthesis of α-aminophosphonates or α-aminophosphine oxides, see: (p) C. Fiore, I. Sovic, S. Lukin, I. Halasz, K. Martina, F. Delogu, P. C. Ricci, A. Porcheddu, O. Shemchuk, D. Braga, J.-L. Pirat, D. Virieux and E. Colacino, ACS Sustainable Chem. Eng., 2020, 8, 18889–18902 CrossRef CAS.
- (a) J. F. Reynes, F. León and F. García, ACS Org. Inorg. Au, 2024, 4, 432–470 CrossRef CAS PubMed; (b) K. Kubota, R. Hisazumi, T. Seo and H. Ito, RSC Mechanochem., 2024, 1, 250–254 RSC; (c) X. Gao, L. Zhang, M. Cui, W. Qi, H. L. Lam, R. Huang and R. Su, Chem. Eng. J., 2025, 511, 162260 CrossRef CAS.
- L. Cicco, A. Fombona-Pascual, A. Sánchez-Condado, G. A. Carriedo, F. M. Perna, V. Capriati, A. Presa Soto and J. García-Álvarez, ChemSusChem, 2020, 13, 4967–4973 CrossRef CAS PubMed.
- (a) D. Tan, Z. Xuan, Y. Sim, R. Ganguly and F. García, CrystEngComm, 2018, 20, 5998–6004 RSC; (b) Y. X. Shi, K. A. Martin, R. Z. Liang, D. G. Star, Y. Li, R. Ganguly, Y. Sim, D. Tan, J. Díaz and F. García, Inorg. Chem., 2018, 57, 10993–11004 CrossRef CAS PubMed; (c) X. Shi, F. León, Y. Sim, S. Quek, G. Hum, Y. Xin, J. Khoo, Z. X. Ng, M. Yang Par, H. Chee Ong, V. K. Singh, R. Ganguly, J. K. Clegg, J. Díaz and F. García, Angew. Chem., Int. Ed., 2020, 59, 22100–22108 CrossRef CAS PubMed; (d) X. Shi, F. Leon, H. C. Ong, R. Ganguly, J. Díaz and F. García, Commun. Chem., 2021, 4, 21 CrossRef CAS PubMed; (e) Q. Lo, D. Pye, S. Gesslbauer, Y. Sim, F. García, A. J. P. White and G. J. P. Britovsek, Catal. Sci. Technol., 2022, 12, 4544–4551 RSC; (f) Y. Sim, F. Leon, G. Hum, S. J. I. Phang, H. C. Ong, R. Ganguly, J. Díaz, J. K. Clegg and F. García, Commun. Chem., 2022, 5, 59 CrossRef CAS PubMed.
- For general reviews which cover the advantages of one-pot tandem protocols, see: (a) Y. Hayashi, Chem. Sci., 2016, 7, 866–880 RSC; (b) Y. Hayashi, Acc. Chem. Res., 2021, 54, 1385–1398 CrossRef CAS PubMed. For an up-to-date and thorough overview of mechanochemical-assisted tandem transformations, see: (c) N. Biedermann and M. Schnürch, Chem. – Eur. J., 2025, 31, e202500798 CrossRef CAS PubMed.
- O. Herd, A. Hebler, M. Hingst, P. Machnitzki, M. Terrer and O. Stelzer, Catal. Today, 1998, 42, 413–420 CrossRef CAS.
- (a) S. Kotani and M. Nakajima, Tetrahedron Lett., 2020, 61, 151421 CrossRef CAS; (b) T. Ayad, A. Gernet, J.-L. Pirat and D. Virieux, Tetrahedron, 2019, 75, 4385–4418 CrossRef CAS; (c) M. Banaglia and S. Rossi, Org. Biomol. Chem., 2010, 8, 3824–3830 RSC.
- (a) A. Bader and E. Lindner, Coord. Chem. Rev., 1991, 108, 27–110 CrossRef CAS; (b) T. M. Shaikh, C.-M. Weng and F.-E. Hong, Coord. Chem. Rev., 2012, 256, 771–803 CrossRef CAS; (c) P. Štěpnička and F. Horký, Eur. J. Inorg. Chem., 2022, e202200276 CrossRef.
- N. I. Ivanova, P. A. Volkov, K. O. Khrapova, L. I. Larina, I. Y. Bagryanskaya, N. K. Gusarova and B. A. Trofimov, Russ. J. Org. Chem., 2016, 52, 772–776 CrossRef CAS.
- (a) T. O'Hare, R. Pollock, E. P. Stoffregen, J. A. Keats, O. M. Abdullah, E. M. Moseson, V. M. Rivera, H. Tang, C. A. Metcalf III, R. S. Bohacek, Y. Wang, R. Sundaramoorthi, W. C. Shakespeare, D. Dalgarno, T. Clackson, T. K. Sawyer, M. W. Deininger and B. J. Druker, Blood, 2004, 104, 2532–2539 CrossRef PubMed; (b) S. Demkowicz, J. Rachon, M. Daśkoa and W. Kozak, RSC Adv., 2016, 6, 7101–7112 RSC; (c) W.-S. Huang, S. Liu, D. Zou, M. Thomas, Y. Wang, T. Zhou, J. Romero, A. Kohlmann, F. Li, J. Qi, L. Cai, T. A. Dwight, Y. Xu, R. Xu, R. Dodd, A. Toms, L. Parillon, X. Lu, R. Anjum, S. Zhang, F. Wang, J. Keats, S. D. Wardwell, Y. Ning, Q. Xu, L. E. Moran, Q. K. Mohemmad, H. G. Jang, T. Clackson, N. I. Narasimhan, V. M. Rivera, X. Zhu, D. Dalgarno and W. C. Shakespeare, J. Med. Chem., 2016, 59, 4948–4964 CrossRef CAS PubMed.
- NaCl has been previously reported as a solid solvent to assist both the mechanochemical synthesis and the post-synthesis. See for example RSC; J. Yang, X. Feng, G. Lu, Y. Li, C. Mao, Z. Wena and W. Yuan, Dalton Trans., 2018, 47, 5065–5071 RSC.
- J. Breinsperger, N. Podlesnik, F. Mele and M. Schnürch, Chem. – Eur. J., 2026, 32, e202503536 CrossRef PubMed.
- Product purity was assessed by NMR spectroscopy (1H and 31P), with no significant side products detected (see SI).
- A. Lapkin and D. J. C. Constable, Green Chemistry Metrics: Measuring and Monitoring Sustainable Processes, 2008, Print ISBN: 9781405159685, Online ISBN: 9781444305432 Search PubMed.
- (a) V. K. Singh, A. Chamberlain-Clay, H. C. Ong, F. León, G. Hum, M. Y. Par, P. Daley-Dee and F. García, ACS Sustainable Chem. Eng., 2021, 9, 1152–1160 CrossRef CAS; (b) F. Leon, C. Li, J. F. Reynes, V. K. Singh, X. Lian, H. C. Ong, G. Hum, H. Sun and F. García, Faraday Discuss., 2023, 241, 63–78 RSC; (c) N. Fantozzi, J.-N. Volle, A. Porcheddu, D. Virieux, F. García and E. Colacino, Chem. Soc. Rev., 2023, 52, 6680–6714 RSC.
- (a) E. Colacino, V. Isoni, D. Crawford and F. García, Trends Chem., 2021, 3, 335–339 CrossRef CAS; (b) J. F. Reynes, V. Isoni and F. García, Angew. Chem., Int. Ed., 2023, 62, e202300819 CrossRef CAS PubMed.
- (a) P. J. Parsons, C. S. Penkett and A. J. Shell, Chem. Rev., 1996, 96, 195–206 CrossRef CAS PubMed; (b) M. J. Climent, A. Corma and S. Iborra, Chem. Rev., 2011, 111, 1072–1133 CrossRef CAS PubMed; (c) H. Pellissier, Chem. Rev., 2013, 113, 442–524 CrossRef CAS PubMed; (d) U. Wille, Chem. Rev., 2013, 113, 813–853 CrossRef CAS PubMed; (e) X. Zeng, Chem. Rev., 2013, 113, 6864–6900 CrossRef CAS PubMed; (f) C. M. R. Volla, I. Atodiresei and M. Rueping, Chem. Rev., 2014, 114, 2390–243125 CrossRef CAS PubMed; (g) X. Ma and W. Zhang, iScience, 2022, 25, 105005 CrossRef CAS PubMed.
- (a) L. Cicco, M. J. Rodríguez-Álvarez, F. M. Perna, J. García-Álvarez and V. Capriati, Green Chem., 2017, 19, 3069–3077 RSC; (b) G. Dilauro, M. Dell’Aera, P. Vitale, V. Capriati and F. M. Perna, Angew. Chem., Int. Ed., 2017, 56, 10200–10203 CrossRef CAS PubMed; (c) S. Ghinato, G. Dilauro, F. M. Perna, V. Capriati, M. Blangetti and C. Prandi, Chem. Commun., 2019, 55, 7741–7744 RSC; (d) D. Elorriaga, M. J. Rodríguez-Álvarez, N. Ríos-Lombardía, F. Morís, A. Presa Soto, J. González-Sabín, E. Hevia and J. García-Álvarez, Chem. Commun., 2020, 56, 8932–8935 RSC; (e) D. Elorriaga, F. de la Cruz, M. J. Rodríguez-Álvarez, A. Lara-Sánchez, J. A. Castro-Osma and J. García-Álvarez, ChemSusChem, 2021, 14, 2084–2092 CrossRef CAS PubMed; (f) M. Ramos-Martín, R. Lecuna, L. Cicco, P. Vitale, V. Capriati, N. Ríos-Lombardía, J. González-Sabín, A. Presa-Soto and J. García-Álvarez, Chem. Commun., 2021, 57, 13534–13537 RSC; (g) L. Cicco, M. Roggio, M. López-Aguilar, M. Ramos-Martín, F. M. Perna, J. García-Álvarez, P. Vitale and V. Capriati, ChemistryOpen, 2022, 11, e202200160 CrossRef CAS PubMed; (h) D. Elorriaga, B. Parra-Cadenas, A. Antiñolo, F. Carrillo-Hermosilla and J. García-Álvarez, ChemSusChem, 2022, 15, e202201348 CrossRef CAS PubMed; (i) D. Arnodo, M. Ramos-Martín, L. Cicco, V. Capriati, N. Ríos-Lombardía, J. González-Sabín, A. Presa Soto and J. García-Álvarez, Org. Biomol. Chem., 2023, 21, 4414–4421 RSC; (j) N. Ríos-Lombardía, G. Morís-Menéndez, I. Lavandera, V. Gotor-Fernández and J. García-Álvarez, Adv. Synth. Catal., 2024, 366, 3144–3152 CrossRef.
- M. López-Aguilar, M. Ramos-Martín, N. Ríos-Lombardía, L. Cicco, J. García-Álvarez, C. Concellón and V. del Amo, ChemCatChem, 2024, 16, e202400437 CrossRef.
- Hybrid one-pot tandem protocols rely on the combination of different synthetic organic tools (transition metals, organocatalysts, enzymes or main-group reagents) in the same reaction media CrossRef; R. Ye, J. Zhao, B. B. Wickmeyer, F. D. Toste and G. A. Somorjai, Nat. Catal., 2018, 1, 318–325 CrossRef.
- Under these conditions, a TON of 8.1 and a TOF of 4.05 h−1 were determined (2 h, 10 mol% catalyst). Full details are provided in the SI (section III).
| This journal is © The Royal Society of Chemistry 2026 |