 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Evaluation of a lab-scale ionic liquid synthesis using life cycle assessment
Rhea
Mathew
a,
Sirui
Chen
 a,
Agnieszka
Brandt-Talbot
a,
Agnieszka
Brandt-Talbot
 a,
Jacqueline S.
Edge
a,
Jacqueline S.
Edge
 b and
Tom
Welton
b and
Tom
Welton
 *a
*a
aDepartment of Chemistry, Molecular Sciences Research Hub, Imperial College, White City, London W12 0BZ, UK. E-mail: t.welton@imperial.ac.uk
bSchool of Metallurgy & Materials, University of Birmingham, Edgbaston, Birmingham, B15 2TT, UK
First published on 3rd March 2026
Abstract
This study presents the life cycle assessment (LCA) for the synthesis of 1-ethyl-3-methylimidazolium acetate, [C2C1im][OAc], an ionic liquid (IL) that is often studied at lab-scale. Three pathways were evaluated using SimaPro® LCA software. At a 3–4 g scale, the anion-exchange route had the lowest environmental impact (99.8 Pt), followed closely by the dimethyl carbonate route (102.0 Pt), while the silver acetate route had an increased impact score (133.5 Pt). Scaling up the dimethyl carbonate route using a high-pressure benchtop reactor considerably improved its sustainability (14.8 Pt). Hotspots were identified and recommendations for improvements are given. Comparison is made with several green metrics. The investigation acts as a guide for sustainable ionic liquid synthesis in the laboratory, promoting critical evaluation of established synthetic work-flows using methodologies such as LCA, and the holistic implementation of green chemistry principles in daily working practices.
Green foundation1. The work establishes evidence-based guidelines to improve the synthesis of a commonly synthesised aprotic IL, or chemicals with similar synthetic pathways. It provides actionable and targeted recommendations to reduce the impact of laboratory IL research.2. The advantages of operating on a larger scale under high-pressure conditions were sufficient that this was the preferred method by both LCA and the Green Metrics E-factor, Process Mass Intensity and atom economy. However, when this route is not available, the study reveals discrepancies among the results of the individual Green Metrics analysis (E-factor, Effective Mass Yield, Process Mass Intensity, Atom Economy) and between these and the results of the LCA, establishing LCA as a methodology that more precisely evaluates the environmental impact of a chemical reaction. 3. Future work includes establishing modified synthesis procedures for ionic liquids in the lab based on the findings, such as improved reactor insulation and larger batch processing. Expanding the LCA to include toxicological data of the IL would provide a more comprehensive environmental assessment. |
Introduction
As scientists race to develop solutions to environmental and other societal challenges, their laboratories contribute to depletion of the earth's finite resources and to global warming. Laboratories can consume 5 to 10 times more energy than office spaces and may consume up to 60% of the total freshwater used by universities.1 Frameworks towards achieving sustainable laboratories such as the Laboratory Efficiency Assessment Framework2 (LEAF) and My Green Lab3 are being adopted worldwide by academics concerned about the impact of their research activities on the environment. While these programs encourage systematic monitoring and reduction of waste generation in laboratories, they do not target individual chemical reactions. Each of these reactions requires distinct reagents and reaction conditions, with different classes of chemicals having unique degradation pathways and interactions with the environment. The use of hazardous reagents and resource-intensive purification steps needs a case-by-case assessment and targeted modification to advance truly sustainable practices.4,5The field of Green Chemistry started in the 1990s, introducing fundamental principles and green metrics for evaluating the ‘greenness’ of a chemical process.6 Mass-based green metrics, which include Atom Economy and Effective Mass Yield (EMY), were developed based on the principle that chemical reactions are greener when they produce either no by-products or ones with lower molar weight.7 Another metric, the E-factor, the mass ratio of waste to product, is closely related to Process Mass Intensity (PMI) and measures the mass ratio of total waste to total product, while PMI measures the ratio of total input materials to product (E-factor = PMI − 1).8 The ACS Green Chemistry Institute Pharmaceutical Roundtable adopted PMI as a benchmark green metric, due to its broad coverage of resource utilisation and easy availability of data on inputs.9 These metrics, however, fail to capture environmental, health, and safety concerns that are associated with specific types of inputs and outputs.10 The relevance of mass-based metrics can change because some materials have a larger or smaller environmental impact than their weight alone indicates, and fail to cover more recent developments in sustainability practice, such as the Safe and Sustainable by Design concept.11 However, their strength lies in the limited information required to calculate them and easy mathematical manipulations, which allows application to small-scale syntheses that are carried out infrequently or even only as a one-off.
For products that are made at a larger scale and/or more frequently, a broader and more detailed evaluation can be carried out through methodologies such as Life Cycle Assessment (LCA).12 LCA assesses resources consumed and waste emitted during an item's production or the delivery of a service, and associates these with measurable environmental and health impacts. Unlike green metrics, it has the potential to consider all aspects of a product's (or service's) life cycle and evaluates the impacts based on the intended function, not just product weight. LCA can also detect the unintended shifting of the environmental burden to elsewhere in a product's life cycle.13 A ‘cradle-to-grave’ LCA examines the extraction of raw materials, production, transportation, distribution, use, and final disposal of the product. A ‘cradle-to-gate’ LCA considers only the extraction and production stages and is adequate for comparing different manufacturing routes that generate the same product.
LCA is usually carried out by government bodies or industries but also has applications in academic research settings. While LCA can be time-consuming and labour-intensive, interest has grown in applying it to small scale chemical reactions that are conducted repeatedly, such as in university teaching laboratories. For example, mass-based green metrics combined with LCA were used to evaluate the environmental impact of an organic dye synthesis, comparing two proposed synthetic routes to a classic Suzuki–Miyaura cross-coupling.14 The classic route's main impact areas were solvent waste, metals, and energy required for multiple purification steps. While the new routes proposed in the study had a lower overall impact, their eco-toxicity score was higher due to the use of n-butyllithium and tin-containing reagents. A LCA comparison of two methods for synthesizing (E)-stilbenes found that the method traditionally taught in an undergraduate course had a significantly higher acidification potential, and utilised volatile organic compounds with higher global warming potential.15 Another study used the DOZN 2.0 platform to analyse the laboratory synthesis of hexane nitrile from hexanoic acid and benzophenone from benzhydrol, identifying organic reagent and solvent use as environmental hotspots.16 Given these reactions are conducted by many students every year, modifying them to reduce environmental impact at a small scale can lead to substantial cumulative benefits over time.
This study focuses on the synthesis of ionic liquids (ILs) which are liquids with melting points below or near ambient temperature and are composed entirely of cations and anions.17 They have garnered interest as promising alternatives to organic solvents, which are often flammable, toxic by inhalation, and have limited opportunity for specialised design.18 ILs are made up of charged species and hence are highly conductive, have low vapour pressures and can have strong interactions with solutes through coulombic attractions and hydrogen bonding. Since a substantial number of cation and anion combinations are available to form low melting salts, ILs are regarded to have customisable physicochemical properties, and are being investigated extensively for various applications where these characteristics may provide advantages.19,20
This study compares the environmental impact of three synthesis pathways for the IL 1-ethyl-3-methylimidazolium acetate or [C2C1im][OAc] (Fig. 1) using LCA and Green Metrics. [C2C1im][OAc] has been studied as a solvent in making biopolymer films21,22 and fibres,23 and as a sorbent in carbon capture.24 [C2C1im][OAc] was selected for this study because it is widely used by researchers and is frequently synthesised in individual laboratories. The LCA of [C2C1im][OAc] synthesis can also serve as a model for the synthesis of other hydrophilic aprotic ionic liquids, which are generally made using similar methods.
Synthesis of [C2C1im][OAc]
[C2C1im][OAc] is typically synthesised by generating the cation through the alkylation of an uncharged 1-alkylimidazole. The anion of some aprotic ILs can directly derive from the alkylating agent. Since this is not possible for [C2C1im][OAc], as methyl acetate and ethyl acetate are not sufficiently active alkylating agents, a suitable aprotic IL precursor containing the [C2C1im]+ cation is generated and the desired anion is subsequently introduced through anion exchange, a process also known as metathesis.25 A common route for synthesising aprotic imidazolium ILs is via a halide precursor, such as 1-ethyl-3-methylimidazolium bromide ([C2C1im]Br), synthesised from 1-methylimidazole and bromoethane via an SN2 mechanism (Scheme 1). The unsubstituted nitrogen atom in 1-methylimidazole acts as a nucleophile and attacks the electrophilic carbon atom on bromoethane. This leads to the formation of a charged imidazolium cation while bromide forms the anion of the precursor IL.26 This reaction is often carried out over 2–3 days at room temperature if a colourless product is the target, however, a raised reaction temperature can accelerate the rate of reaction.27 Since [C2C1im]Br is a precursor to many ionic liquids, a sustainability analysis of its synthesis is generally useful.If a metathesis reaction to generate the final IL is required, an anion exchange is needed between the precursor IL and another source that can provide the target anion. Target anions that have limited hydrogen-bond basicity can be introduced using Li+ or Na+ salts while dissolved in an organic solvent, with left-over metal halide preferentially extracted from the organic solvent phase with deionised water. This approach does not work for halide precursors and hydrophilic target ILs such as [C2C1im][OAc], as both the halide and acetate are removed into the water phase. Therefore, alternative metathesis reactions that quantitatively remove the precursor anion need to be used. Several strategies have been developed to produce 1,3-dialkylimidazolium acetate ILs. Silver salts can help generate highly water-miscible ILs from halide precursors, as silver halides such as AgBr and AgCl are sparingly soluble in water and form solids that can be removed by filtration.27 Silver(I) acetate has been used to synthesise [C2C1im][OAc] from bromide precursors in aqueous solution (Scheme 2(a)). The acetate anion and [C2C1im]+ cation remain dissolved in water, which is then removed by evaporation. The reaction takes place at room temperature and the precipitation of AgX (X = Cl or Br) drives the reaction, ensuring complete exchange of anions and a high yield. In terms of practical limitations, the silver salt metathesis is fast, but it must take place in a dark environment, as silver acetate exhibits photodecomposition,28 resulting in silver oxide contamination in the product. Another drawback is the potential for formation of submicron particles of silver halide, which are challenging to remove.29 A variation has been proposed where silver acetate is replaced by lead(II) acetate. While lead has lower cost and higher stability, the use of lead is strongly discouraged, due to its persistent human and environmental toxicity, and should not be utilised in any laboratory environment, if lead-free methods are available.30 Hence, this alternative was not analysed here.
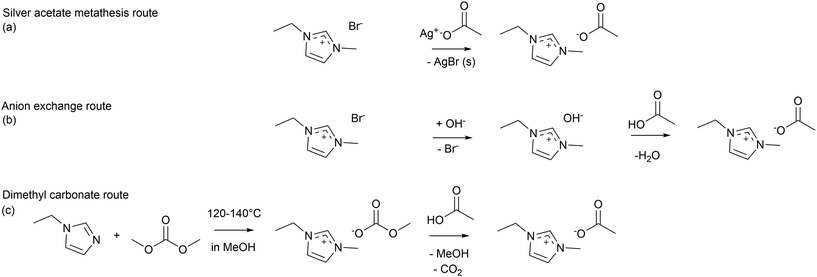 | ||
| Scheme 2 Synthesis of [C2C1im][OAc] via (a) silver acetate metathesis, (b) anion-exchange route, (c) dimethyl carbonate routes. | ||
Anion metathesis for hydrophilic ILs can also be carried out by employing an anion-exchange resin, which is called the anion-exchange column method. An appropriate anion is coordinated to a positively charged stationary phase in large excess, while the cation is present in solution as part of the precursor IL. Anion exchange is reversible in principle, so the resin must be chosen to provide strong preference for the anion of the precursor IL.29 Cationic resins loaded with hydroxide (OH−) ions are particularly useful in this application, as they generate hydroxide salts (Scheme 2(b)), which enable a subsequent Brønsted acid–base neutralisation to introduce the desired IL anion, with water as the by-product. Direct loading of weakly basic anions such as acetate onto the strongly acidic resin is inefficient because the resin's positive sites have a lower selectivity for [AcO]− than for OH− or Br−.31 To produce [C2C1im][OAc], the halide precursor [C2C1im]X [X = Cl or Br] dissolved in water is contacted with an anion-exchange resin loaded with OH− ions provided in a column. The resulting hydroxide precursor [C2C1im][OH], which is maintained in solution, is then reacted with an equimolar amount of acetic acid. A concentrated solution of [C2C1im]X and a column of sufficient length ensures complete anion exchange, while contamination can be prevented by ensuring that the resin has been rinsed according to the manufacturer's guidelines.25 In DuPont AmberLyst™ resin columns, the acid sites can be contaminated by exchanged cations, amine-containing organics that neutralize charge, nitriles and formamide which hydrolyse to amines, and dialkyl sulfides that react with sulfonic acid groups. Fouling can also occur by adsorption and steric blockage from higher molecular weight organics, such as dienes and aldehydes, that accumulate in the resin pores.32 While anion-exchange resins can be reused, they have a limited lifespan, as contaminants build up over time and, hence, they must be replaced periodically, adding to resource consumption.31
An alternative four-step method that combines precipitation followed by acid base neutralisation has been reported using 1-ethyl-3-methylimidazolium hydrogen sulfate, [C2C1im] [HSO4], as an initial precursor.33 1-Ethylimidazole is alkylated with dimethyl sulfate to generate 1-ethyl-3-methylimolium methyl sulfate, followed by hydrolysis to yield the [HSO4]− anion. The anion metathesis step can be carried out with calcium hydroxide in water, which converts the sulfate precursor into the second hydroxide precursor, taking advantage of the insolubility of calcium sulfate in water, followed by equimolar reaction of the dissolved [C2C1im][OH] with acetic acid, similar to the anion-exchange method.33 The advantage is that calcium hydroxide is photostable and has lower cost than silver(I) acetate while the use of an ion-exchange column is omitted, however, the approach was not included in the assessment, due to the use of a proven carcinogenic alkylating reagent.
Dialkylimidazolium cations can also be formed by employing dimethyl carbonate (DMC) as an alkylating agent (Scheme 2(c)), with the advantage that methyl carbonate can be decomposed by sufficiently strong Brønsted acids. The 1-ethylimidazole is heated with an excess of DMC in methanol at elevated pressure and temperature, and the resulting precursor 1-ethyl-3-methylimidazolium methyl carbonate [C2C1im][MeCO3] is combined with acetic acid to yield [C2C1im][OAc] with methanol and CO2 as by-products. The cation and methyl carbonate can undergo a reaction, leading to formation of the zwitterion 1-ethyl-3-methylimidazolium-2-carboxylate. However, upon addition of acetic acid, both the main precursor and the precursor by-product form [C2C1im][OAc].34
Experimental
Life cycle assessment
The proposed methodology (Fig. 2) for carrying out this assessment was adapted from previous state-of-the-art life cycle studies of ionic liquids.35–40 The LCA was conducted as per the ISO 14040/44 standards41,42 and modelling was carried out on SimaPro 9.643 in four stages: (i) goal and scope definition, (ii) inventory analysis, (iii) impact assessment, and (iv) interpretation of results.Ionic liquid synthesis. 1H and 13C NMR spectra were obtained using a Bruker-DRX-400 spectrometer, with DMSO-d6 as the solvent. The spectra were analysed using the MESTRELAB MestReNova software (SI Fig. S1–S10) The water content of the ILs was measured using Mettler Toledo Karl–Fischer titrators V20 and C20. 1-Methylimidazole (C4H6N2, >99%), 1-ethylimidazole (C5H8N2, >99.0%), 1-bromoethane (C2H5Br, 98%), acetic acid (CH3COOH, 99.8%), and dimethyl sulfate (DMSO, 99.8%) were purchased from Sigma Aldrich. Acetonitrile (C2H3N, ≥99.9%) and ethyl acetate (CH3COOC2H5, ≥99.5%) were purchased from VWR Chemicals. Silver acetate (AgC2H3O2, 99%) was purchased from Flurochem. DuPont Amberlyst™ anion-exchange resin was obtained from ACROS Organics.
The synthetic procedures are detailed in this section. Additional quantitative measurements such as yields and the measurements of energy consumption are detailed in the SI. We should point out that measurements were made using equipment available in the Welton Group laboratories at Imperial College London and that results could vary if other apparatus were used.
Synthesis of 1-ethyl-3-methylimidazolium bromide (∼90 g batch). 1-Methylimidazole and bromoethane were purified by distillation shortly before the alkylation. 1-Methylimidazole (41.2 g) and potassium hydroxide (5 g) were added into a 150 mL round bottom flask, and the mixture was stirred overnight (18 h). The yellow liquid was distilled under vacuum for 1 h at 70 °C to obtain purified, colourless 1-methylimidazole. Bromoethane (147 g) in a separating funnel was extracted with concentrated H2SO4 (25 mL), followed by washing with deionized water, extraction with aqueous NaHCO3, and finally washing with deionized water until pH 7 was reached. It was subsequently mixed with 5 g solid MgSO4 in a 250 mL round bottom flask and distilled at 55 °C, which took 2 h. 1-Methylimidazole (40 mL) was dissolved in ethyl acetate (150 mL) in a round bottom flask, to which bromoethane (100 mL) was added dropwise. The reaction was stirred vigorously at room temperature for 72 h. The temperature was increased to 35 °C to drive the reaction forward, and the mixture was stirred for an additional 20 h at this temperature. Upon cooling, the solid was recrystallized by dissolving in acetonitrile (156 g) and pouring into cold ethyl acetate (1939 g) (MeCN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc should be in a 1
EtOAc should be in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 ratio). The sample was first dried using a rotary evaporator, followed by overnight drying on the Schlenk line under vacuum, with an external trap to reduce the water content to <0.5% and a yield of 87.3 g.
10 ratio). The sample was first dried using a rotary evaporator, followed by overnight drying on the Schlenk line under vacuum, with an external trap to reduce the water content to <0.5% and a yield of 87.3 g.
Characterisation: 1H NMR (400 MHz, DMSO-d6, ppm): δ 9.14 (1H, s), 7.81 (1H, t, J = 2 Hz), 7.72 (1H, t, J = 2 Hz), 4.19 (2H, q, J = 7 Hz), 3.85 (3H, s), 1.41 (3H, t, J = 7 Hz). 13C {1H} NMR (100 MHz, DMSO-d6, ppm): δ 136.87, 124.18, 122.60, 44.73, 36.34, 15.77.
Synthesis of [C 2 C 1 im][OAc] via silver(I) acetate metathesis from the halide precursor. 1-Ethyl-3-methylimidazolium bromide was dried thoroughly overnight (18 h) under vacuum on a Schlenk line at room temperature and weighed (4.92 g). The 1-ethyl-3-methylimidazolium bromide was dissolved in a minimal amount of deionised water, i.e. water was added dropwise until the IL completely dissolved. Silver(I) acetate (4.32 g) was added to a 100 mL round bottom flask covered in aluminium foil to prevent its photodegradation. The silver(I) acetate and 1-ethyl-3-methylimidazolium bromide were stirred vigorously for approximately 70 h in the dark. The solution was filtered under vacuum, and a sample of the filtrate was tested for silver with 1 M HCl. Another sample was tested for bromide ions with a solution of 0.3 M AgNO3 in 1.0 M HNO3. An additional 0.5 g of silver acetate had to be added to obtain a negative result for both tests. The ionic liquid was first dried using a rotary evaporator for 2 h, followed by overnight drying under vacuum on a Schlenk line with an external trap with a yield of 3.61 g.
Characterisation: 1H NMR (400 MHz, DMSO-d6, ppm): δ 10.16 (1H, s), 7.94 (1H, t, J = 2 Hz), 7.84 (1H, t, J = 2 Hz), 4.21 (2H, q, J = 8 Hz), 3.87 (3H, s), 1.57 (3H, s), 1.38 (3H, t, J = 8 Hz). 13C {1H} NMR (100 MHz, DMSO-d6, ppm): δ 173.99, 138.09, 123.97, 122.44, 44.33, 35.86, 26.58, 15.68.
Synthesis of [C 2 C 1 im][OAc] via the anion-exchange method from the halide precursor. A glass column (inner diameter 4 cm, length 25 cm) was packed with Amberlite™ IRN-78 in its OH-form, and water was flushed through the resin until the eluent reached pH 7. 1-Ethyl-3-methylimidazolium bromide (5 g) was dissolved in a minimal amount of water and the solution was passed through the anion-exchange column using deionised water as the eluent, with the emerging liquid collected in a flask. The eluent was allowed to flow at a rate of approximately 1 drop per 30 s. The column was washed twice with distilled water. The pH of the eluent increased to pH 14, showing that 1-ethyl-3-methylimidazolium hydroxide was eluting. The collection was stopped once pH 7 was measured, indicating that all the 1-ethyl-3-methylimidazolium hydroxide had been collected. The eluent was tested for the presence of halide ions with a solution of 0.3 M AgNO3 in 1.0 M HNO3. The eluent containing the 1-ethyl-3-methylimidazolium hydroxide was combined with acetic acid (1.57 g) in a 1 L round bottom flask to generate aqueous [C2C1im][OAc]. The water was removed using a rotary evaporator for 4 h and the ionic liquid was further dried under vacuum for 2–3 days on the Schlenk line with an external trap, with a yield of 3.52 g.
Characterisation: 1H NMR (400 MHz, DMSO-d6, ppm): δ 9.90 (1H, s), 7.85 (1H, t, J = 2 Hz), 7.76 (1H, t, J = 2 Hz), 4.22 (2H, q, J = 8 Hz), 3.86 (3H, s), 1.55 (3H, s), 1.40 (3H, t, J = 8 Hz). 13C {1H} NMR (100 MHz, DMSO-d6, ppm): δ 172.91, 137.32, 123.41, 121.86, 43.86, 35.42, 25.62, 15.12.
Synthesis of [C 2 C 1 im][OAc] via the dimethyl carbonate route using a pressure tube. 1-Ethylimidazole was purified by stirring overnight (18 h) with potassium hydroxide (5 g) and distilling under vacuum at 80 °C. 1-Ethylimidazole (2 mL), dimethyl carbonate (4.1 mL), and methanol (8 mL) were added into a pressure tube (Ace Glass, 35 mL, 25.4 mm, 17.8 cm long, with #15 back-seal plug, rated to 10.34 bar). The tube was sealed and heated at 120 °C with an oil bath on a standard heating plate. After 115 h (4 and a half days), the tube was cooled to room temperature, before acetic acid was added dropwise to the reaction mixture with stirring. The mixture was dried on the rotary evaporator for 30 min to remove excess MeOH and DMC, then transferred to a Schlenk line and dried under vacuum for 1 h, with a yield of 3.78 g.
Characterisation: 1H NMR (400 MHz, DMSO-d6, ppm): δ 9.90 (1H, s), 7.85 (1H, t, J = 2 Hz), 7.75 (1H, t, J = 2 Hz), 4.20 (2H, q, J = 8 Hz), 3.80 (3H, s), 160 (3H, s), 1.40 (3H, t, J = 8 Hz). 13C {1H} NMR (100 MHz, DMSO-d6, ppm): δ 174.16, 138.35, 123.38, 121.36, 42.90, 35.45, 26.32, 15.60.
Synthesis of [C 2 C 1 im][OAc] via the dimethyl carbonate route using a high-pressure reactor. 1-Ethylimidazole was first purified by stirring overnight with potassium hydroxide (5 g) and distilling under vacuum at 80 °C. The vessel of a Benchtop Parr 4520 Reactor was charged with 1-ethylimidazole (85 mL), dimethyl carbonate (185 mL) and methanol (330 mL). The reaction was carried out for 2 days at 140 °C with a blast shield installed for safety. The pressure rose from 10 bar on the first day to approximately 15 bar on the second day. After decanting the reaction mixture into a round bottom flask, acetic acid (51 mL) was added and the mixture stirred for 3 h at room temperature. The product was treated with 5 g activated charcoal overnight. The suspension was filtered with filter paper (VWR, quantitative, 5–13 micrometres), syringe filter (Fisher Scientific, PTFE 0.2 micrometre) and Biotage ISOLUTE C18. The ionic liquid was dried using the rotary evaporator for 1–2 h, with a yield of 145 g.
Characterisation: 1H NMR (400 MHz, DMSO-d6, ppm): δ 10.09 (1H, s), 7.88 (1H, t, J = 2 Hz), 7.76 (1H, t, J = 2 Hz), 4.12 (2H, q, J = 8 Hz), 3.88 (3H, s), 1.50 (3H, s), 1.26 (3H, t, J = 8 Hz). 13C {1H} NMR (100 MHz, DMSO-d6, ppm): δ 173.38, 137.53, 123.37, 121.85, 43.54, 34.88, 25.48, 15.05.
Material inventory. The material inputs and outputs were measured manually using standard laboratory apparatus. The results were normalised to account for experimental yield and converted to g per g yield of IL. The reference flow included reagents, solvents, anti-solvents, purifying agents, cooling agents, and washing solvents. Waste scenarios for the discarded materials and by-products were considered: water used for washing was assigned as wastewater treatment. Sublimated dry ice and evaporated liquid nitrogen were regarded as indoor air emissions; and hazardous organic waste was assumed to undergo incineration. Consumable items such as personal protective equipment, the life cycle of instrumentation, and chemicals utilised in analytical procedures such as NMR spectroscopy were outside the scope of this investigation.
Precursor materials used for IL synthesis often lack comprehensive life cycle data, due to their limited applications and complex molecular structures. The Life Cycle Tree methodology was implemented to trace these materials back to precursors with available data in the EcoInvent database, using prevailing commercial synthesis routes from literature and patents (Fig. 4). Stoichiometric estimations were made excluding auxiliaries and assuming a 100% yield. 1-Methylimidazole synthesis was assumed to proceed via acid-catalysed methylation of imidazole using methanol.44 Similarly, 1-ethylimidazole synthesis was assumed to occur via the same mechanism, through the Debus–Radziszewski reaction from glyoxal, formaldehyde, and ammonia in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ratio.45 For silver(I) acetate, due to the absence of silver salts in EcoInvent, the Life Cycle Tree was traced back to silver metal. The synthesis pathway involved the conversion of silver metal to silver nitrate,46 followed by silver carbonate,47 and finally to silver acetate.48 The life cycle inventory of the precursor materials and the conducted experiments can be found in the SI.
2 ratio.45 For silver(I) acetate, due to the absence of silver salts in EcoInvent, the Life Cycle Tree was traced back to silver metal. The synthesis pathway involved the conversion of silver metal to silver nitrate,46 followed by silver carbonate,47 and finally to silver acetate.48 The life cycle inventory of the precursor materials and the conducted experiments can be found in the SI.
Energy inventory. Electricity was measured using Energenie-ENER007 energy monitoring devices that are placed between the device plug and the power socket. The energy consumed by the high-pressure reactor for the dimethyl carbonate route could not be measured directly, as its power rating exceeded the safe operating limit of the available energy monitors, therefore the energy requirement for this pathway was calculated using the power rating and operation time. For the rotary evaporator, the energy meters measured the electricity consumed by the vacuum pump and water bath individually.
| Electricity (W h) = power (W) × time (h) |
The energy requirements of the precursors were calculated based on the methods outlined by Felder and Rousseau.49
| Q − W = ΔH + ΔEk + ΔEp | (i) |
| ΔH = ∑(n × Ĥ)output − ∑(n × Ĥ)input | (ii) |
 | (iii) |
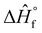 is their heat of formation, and Cp is their calorific value. We assume no work (W) is done, and the kinetic (ΔEk) and potential (ΔEp) energies are zero. Additionally, we assume that the temperature of the reactants is the same as the reference temperature. Thus, T1 = T2 = 298.15 K, rendering the integral term zero. The resulting equations are as follows:
is their heat of formation, and Cp is their calorific value. We assume no work (W) is done, and the kinetic (ΔEk) and potential (ΔEp) energies are zero. Additionally, we assume that the temperature of the reactants is the same as the reference temperature. Thus, T1 = T2 = 298.15 K, rendering the integral term zero. The resulting equations are as follows:| Q = ΔH | (iv) |
| ΔH = ∑(n × Ĥ)output − ∑(n × Ĥ)input | (v) |
 | (vi) |
Only the energy consumption for the heating and cooling of each step was considered, as the specifics of other process operations, such as pumping and separation, are not reported by manufacturers. Assuming that the reagents were manufactured commercially with practical inefficiencies, conversion factors were applied based on the methodology outlined by previous IL life cycle studies. The theoretical heat requirements for endothermic reactions were multiplied by a factor of 4.2, assuming natural gas as the heat source. Similarly, the theoretical heat generated by exothermic reactions was converted to actual cooling electricity requirements, using a factor of 3.2.37,38 The energy input selected while modelling the SimaPro process was electricity available on the high-voltage grid in the United Kingdom (GB). The energy inventory of the precursor materials and the conducted experiments can be found in the SI. Instrument-specific LCI data is also available.
Results and discussion
Base case scenario
The base case scenario compares the impact of the anion-exchange route, the dimethyl carbonate pressure tube route, and the silver acetate route using life cycle assessment methodology (Fig. 5). The total environmental load of each endpoint category is expressed as a single score ‘Pt’, the relative magnitude of damage. The assessment found that the anion-exchange route had the lowest impact score of 99.8 Pt, followed closely by the dimethyl carbonate route at 102.0 Pt, and finally the silver acetate route at 133.5 Pt. The human health category dominated the impact overall score, as weighting factors are determined by policy and monetisation targets which tend to prioritise human well-being, that is, stakeholders and policymakers are expected to invest in preventing damage to human health over conserving the biosphere and resources.52 | ||
| Fig. 5 Comparison of endpoint single scores (in Pt) of the three methods expressed as their weighted impact on human health, ecosystems, and resources. | ||
Midpoint categories can help identify specific sources of environmental load (Fig. 6). Despite the silver acetate route having the highest overall damage score, the anion-exchange and dimethyl carbonate routes had higher contributions to the global warming categories due to the energy intensity of these routes, discussed in detail later. The global warming scores of the anion-exchange route and dimethyl carbonate route were 45% and 42% higher than the silver acetate route, respectively. The values for the midpoint categories discussed in the ‘hotspot analysis’ section can be found in the SI (Fig. S11–S15).
 | ||
| Fig. 6 Impact in selected midpoint categories for the three [C2C1im][OAc] synthesis routes evaluated in this study, expressed as a percentage of the highest impact in each category. | ||
Ionic liquid toxicity and life cycle data
It should be noted that there is a lack of life cycle data for dialkylimidazolium ILs, and ILs in general, as the manufacture of ILs and their precursors is still an emerging industry. The data required to accurately capture industrial production, associated energy, transportation, and waste treatment steps will take several years to establish. Toxicological data on ILs and their precursors is scarce. UK and EU REACH regulations do not require mandatory testing of small-scale chemical production (less than 1 tonne per year) and require only a basic level of testing for production of up to 100 tonnes per year.53,54 While [C2C1im][OAc] is predicted to be of lower toxicity amongst ILs, a study on its effect on the yeast S. cerevisiae indicated that it inhibited their respiration activity.55 Toxicity to microorganisms, which are foundational species, can impact ecosystems. Reports suggest that dialkylimidazolium ILs are already persistent in aqueous samples, food products, and the human body.56 The environmental profile of [C4C1im]Br was compared to that of toluene in the synthesis of acetylsalicylic acid or aspirin, which demonstrated that the IL had higher ecotoxicity scores than the organic solvent it was replacing.39Hotspot identification and analysis
The hotspot analysis revealed that the production of the precursor [C2C1im]Br contributes significantly to the impact of the final ionic liquid synthesis. Ethyl acetate, used in the recrystallisation, was the main contributor (33.6%) to the precursor damage score (Fig. 7, SI Fig. S11). Ethyl acetate is a Category 2 eye irritant and can be acutely toxic to internal organ systems, causing loss of respiratory and heart function.57 Acetonitrile, also used in the recrystallisation, is classified as an Environmental Protection Agency (EPA) Category 4 acute toxin and is highly flammable.58 While some solvent selection guides deem acetonitrile as relatively green, its production releases fine particulate matter and carbon emissions that increase the damage score of the reaction. The use of solvents in precursor production and their subsequent incineration accounts for the 60.7% contribution to human non-carcinogenic toxicity, as well as a 63.4% contribution to human carcinogenic toxicity. Neither acetonitrile nor ethyl acetate are classified carcinogens,57,58 so the contribution to carcinogenic toxicity in the LCA may stem from the production of these chemicals. In the case of ethyl acetate, it is made via Fisher esterification which is catalysed by sulfuric acid, and occupational exposure to its mists has been linked to increased risk of laryngeal and lung cancers in factory workers.59 Bromoethane used in the precursor production step is suspected of causing cancer on direct exposure.60Electricity consumed by the equipment was found to be another deciding factor for overall environmental damage score of the precursor synthesis, silver acetate route, and anion-exchange route. The rate of electricity consumed by the rotary evaporator to remove the bulk of the solvent was 0.33 kW. After this stage, the product contained trace amounts of solvent and was dried overnight on the Schlenk line since the rotary vane vacuum pump (Oerlikon Leybold Vacuum Pump) attached to the Schlenk line can reach lower pressures than the membrane vacuum pump associated with the rotary evaporator (Heidolph Hei-VAP Rotary Evaporator). However, the energy consumed by the Schlenk line vacuum pump is higher than the rotary evaporator. The specifications of the equipment are given in the SI. This extended overnight drying using the Schlenk line vacuum system accounts for the majority of the overall electricity used in the synthesis of [C2C1im]Br.
After the reaction takes place in the resin column during the anion-exchange route, the [C2C1im][OAc] product is present in a very dilute solution (∼3.5 g product in 500 mL water). The production of dry ice and subsequent release as carbon dioxide in the solvent removal by rotary evaporator is a hotspot for this process, contributing to its high global warming impact score (Fig. 8, SI Fig. S13), along with the impact from using [C2C1im]Br as the precursor. Dry ice also contributed 72.4% to terrestrial ecotoxicity, 72.0% to marine ecotoxicity, and 73.4% to freshwater ecotoxicity for the anion exchange route. The production of dry ice uses refrigerants such as hydrochlorofluorocarbons (HCFCs) and nitrous oxide, which explain its high impact on stratospheric ozone depletion.61,62 Ethanol, used to clean the apparatus after use, contributed 64.1% to the stratospheric ozone depletion score, as bulk chemical industries use similar refrigerants during the manufacturing process.40
 | ||
| Fig. 8 Single score contributions for material flows for [C2C1im][OAc] synthesis via the anion-exchange route. The hotspots are dry use for drying and contributions from the precursor. | ||
The primary impact of the silver acetate route arose from the use of silver(I) acetate salt, contributing 55.4% of total damage score (Fig. 9, SI Fig. S14). The extraction and processing of silver metal was the source of environmental damage, not the multi-step synthesis of silver acetate. The mineral resource scarcity score of silver acetate is 16.2 USD2013, higher than for all other chemicals in this study (all have scores below 1.0 USD2013) as silver is both an expensive and scarce substance in comparison. According to the EcoInvent database, the silver metal extraction and production consume a substantial amount of water, is linked to the production of fine particulate matter and emissions that are carcinogenic. The processing of silver emits volatile arsenic (As) and sulphur dioxide (SO2) from the copper smelters. Large-scale silver mining is associated with socio-cultural conflicts, such as displacement of local communities and occupational hazards faced by mineworkers.63 Thus, the contribution of silver(I) acetate to the single damage score of this pathway is 72.5%. Beyond just the LCA results, the social costs of silver mining are not aligned with the United Nations Sustainable Development Goals of reduced inequalities, good health and well-being, and decent work.
 | ||
| Fig. 9 Single score contribution for material flows for [C2C1im][OAc] synthesis via the silver acetate route. The hotspot is silver acetate production and use. | ||
This route also has practical drawbacks as silver salt is expensive (£12.8 per gram) and photodegrades quickly. It was also observed that silver(I) acetate stored in the lab cupboard failed to produce the desired product, as some of it had oxidised, increasing the waste generated. Even with new, pure reagent, achieving the correct stoichiometric ratio of silver(I) acetate to [C2C1im]Br was challenging, requiring additional reagent to be added until the solution tested negative for Ag+ and Br− ions, and prolonging the reaction by several days.
The hotspot of the dimethyl carbonate route (Fig. 10, SI Fig. S15) was electricity (54.4%) and dry ice (25.4%), with small direct impact from the reagents, including the alkylating agent dimethyl carbonate (DMC). DMC has been classified as a sustainable solvent, meeting the safety, health, and environmental criteria set by the CHEM21 consortium.64 Unlike the other alkylating agents discussed for IL production, it is low in mutagenic toxicity and readily biodegradable. It can be synthesised from captured carbon dioxide and methanol, thereby contributing to a closed-loop carbon cycle.65 The electricity consumption was due the prolonged, high-temperature heating (120 °C) of the pressure tube over a hot plate for 115 hours during the synthesis step. Significant energy is lost to the environment as the hot plate is not insulated, necessitating continuous power input to maintain reaction temperature. Using methanol as the solvent, whose latent heat of vaporisation (∼1100 kJ kg−1) is about half that of water (2260 kJ kg−1), reduced the energy required to remove the solvent compared to water used in the other two synthetic routes.
 | ||
| Fig. 10 Single score contribution of each material flow for [C2C1im][OAc] synthesis via the dimethyl carbonate route in a pressure tube, expressed as Pt. | ||
The impact of electricity on the sustainability of IL synthesis depends on the energy mix of that country's electricity supply. Great Britain's National Grid (the high-voltage power transmission network that supports the country's electricity market) electricity mix contributes to the ionising radiation and global warming impact categories, that can be attributed to electricity generated from nuclear power (19.9% of the National Grid) and natural gas (14.7% of the National Grid), respectively.66
Sensitivity analysis
 | ||
| Fig. 11 Impact of a larger scale synthesis of [C2C1im][OAc] via the dimethyl carbonate route utilising a high-pressure reactor compared with the base case scenarios. | ||
Evaluating the contribution of individual inputs, electricity consumption contributed 90% (13.4 Pt) of the total single score impact (Fig. 12). The electricity use of the DMC route with high-pressure reactor was notably high at 1699 Wh per g of IL, primarily because temperature and pressure in the reactor needed to be maintained continuously over two days. The pressure reactor can be operated at 140 °C, while the pressure tube reaction can only be operated safely up to 120 °C due to a lower pressure rating, resulting in a longer reaction time and hence consumption of more electricity per g IL produced in the pressure tube. The electricity use of the DMC route with the pressure tube was estimated as 7046 Wh per g of IL.
The power rating of the reactor (Parr 4520 Benchtop Reactor) exceeded the safe limit of the energy meter, leading to the use of a standard formula for energy calculation (details in SI). This approach may overestimate energy input, since the reactor was operated well below its maximum capacity, hence a sensitivity analysis, adjusting the energy consumption to 75% and 50% of the maximum was performed (Fig. 13). Results indicated that reducing the energy input to three-quarters led to a 15% decrease in impact, while a reduction by half resulted in a 39% decrease, suggesting potential for significant energy savings. The adjustment in electricity consumption affected all midpoint categories in a similar manner.
Recommendations
Instrument choice can also bring down the impact score. Heidolph Hei-VAP Rotary Evaporator was the model used throughout this work for solvent removal. This was compared to a Buchi Rotavapor® with a recirculatory cooling system after the study was completed (Fig. 15). When removing 50.0 mL water from 3.5 g [C2C1im][OAc], the dry ice model consumed only 332 W on average, while the recirculatory model used about 950 W, due to cooling the solvent to −10 °C, while using a similar drying time of approximately 45 min. However, the recirculatory system's higher energy consumption was offset by eliminating dry ice from the process, lowering the damage score substantially from 99.8 Pt to 61.6 Pt. LEAF guidelines advocate for the use of closed cooling systems such as air-cooled or liquid-cooled condensers (with low boiling point solvents instead of water) for rotary evaporators over single pass cooling systems, and for instruments to be upgraded and repaired, if the funding allows.2
 | ||
| Fig. 15 Reduction in damage score for the ion-exchange route, after incorporating recommended changes in the drying protocol. | ||
While liquid nitrogen has a lower greenhouse gas potential than dry ice, replacing it in the Schlenk line trap might not be feasible due to the fast evaporation of liquid nitrogen, which prevents safe overnight drying. Experimental instructions giving poorly defined ranges of time like ‘overnight’ likely expend resources unnecessarily and need to be critically examined in future work.
While [C2C1im][OAc] cannot be synthesised using sodium acetate, less water-soluble ionic liquids may benefit from using sodium salt instead. Using sodium acetate instead of silver acetate demonstrated a lower environmental impact than the silver acetate and ion-exchange methods, although the dimethyl carbonate high pressure reactor route remains the most sustainable option (Fig. 16). In cases where the use of an alternative salt to silver is feasible, the metathesis approach could be greener than using the ion-exchange column. However, it should be noted that such syntheses often require purification steps involving the use of disfavoured organic solvents,21 which would need to be taken into account.
For larger-scale production or when multiple experiments collectively require substantial quantities, the high-pressure benchtop reactor DMC route is recommended. With good planning, a large batch can be prepared for multiple experiments, thereby reducing the environmental impact per g of IL. As a guideline, the impact of chemicals use can be improved by making experiment-specific reagent purchases at the correct volume and potentially reducing physical experiments through comprehensive computational and literature studies.3
Comparing LCA scores to green metrics
Green metrics for the four reaction routes are presented in Table 1, with detailed calculations available in the SI. Comparing the metrics with results from the LCA is intended to help identify green metrics that might provide guidance for synthetic chemists on the sustainability of a reaction without the need for conducting a comprehensive LCA, which requires additional time and resources such as access to a life cycle inventory data base and energy meter readings. The silver acetate route had the highest LCA score (133.5 Pt), followed by the DMC pressure tube method (102.0 Pt) which had similar score of the anion-exchange method (99.8 Pt). Given the sensitivities discussed earlier (Fig. 13), small changes in experimental conditions (such as modifying dry ice usage) can easily reverse this order for the latter two.| Green metrics | Silver acetate metathesis | Anion-exchange method | Dimethyl carbonate reaction (pressure tube) | Dimethyl carbonate reaction (high-pressure reactor) | Favourable score |
|---|---|---|---|---|---|
| Yield | 81.1% | 79.0% | 91.0% | 96.2% | 100% |
| Atom economy | 47.6% | 63.5% | 91.4% | 91.4% | 100% |
| EMY | 36.9% | 53.9% | 48.1% | 43.1% | 100% |
| E-Factor | 1348 | 4068 | 1463 | 48 | 0 |
| PMI | 1349 | 4069 | 1464 | 49 | 1 |
| Electricity (Wh per g IL) | 413 | 707 | 7046 | 1699 | Low |
| LCA score (Pt) | 133.5 | 99.8 | 102.0 | 14.8 | Low |
In terms of atom economy, the dimethyl carbonate route (91.4%) performed better than the anion-exchange (63.5%) and silver acetate (47.6%) routes, but this did match the pattern observed for the LCA scores. This is ascribed to the lack of accounting for electricity or solvent use, which can be major contributors to life cycle burdens. The Process Mass Intensity (PMI) and E-factor, which are both based on the total bulk mass of materials used per mass of product and take into account solvent use, showed high values across most routes: silver acetate (PMI: 1349, E-factor: 1348), dimethyl carbonate pressure tube (PMI: 1464, E-factor: 1463), and anion-exchange (PMI: 4069, E-factor: 4068). The values reflect the decision to include all materials inventoried during the LCA study such as cooling agents and washing solvents. This is not always done in literature, where PMI and E-factor often only include the reagents and perhaps major solvent use during purification. We encourage other researchers to adopt this more comprehensive approach, as it provides a clearer and more accurate picture of process material demands.
The Effective Mass Yield (EMY) considers the mass of the desired product relative to all non-benign (or hazardous) inputs and parallels the LCA results well. EMY was highest for the ion-exchange method (53.9%), followed by the DMC pressure tube route (48.1%) and lowest for silver acetate (36.9%). Water is typically excluded from EMY calculations, which matches impact assignments in LCA, since non-hazardous compounds are less weighted in LCA scoring.
The dimethyl carbonate route using the high-pressure benchtop reactor stands out as the least impactful synthesis route across the evaluated green metrics. The high-pressure benchtop reactor achieved the highest yield and atom economy, as well as the lowest E-factor and PMI, indicating minimal waste. Its LCA score (14.8 Pt) is much lower than all other methods. The two DMC methods had similar yields, atom economies, and effective mass yield while they vary vastly in PMI and E-factor, where solvent and auxiliary use are included. Taken together, these results indicate that the environmental impact of ionic liquid synthesis arises more from solvents and auxiliary materials than from the core reactants and that reaction scale can generate reducing in use of auxiliaries and solvents if planned correctly.
Energy consumption measured as electricity use per kilogram of ionic liquid (Wh g−1 IL) is the highest at 7046 for the dimethyl carbonate reaction in a pressure tube and lowest for 413 for silver acetate metathesis. Although these energy values do not directly correlate with LCA scores, it can identify process hotspots discussed in earlier sections in a relatively accessible manner, since energy meters are affordable and easy to use.
It should be noted that the results should do not imply that one metric is inherently superior to another. Rather, it is good practice to use multiple metrics in parallel and interpret them collectively to develop a more nuanced understanding of the environmental impacts of the processes being compared.
Conclusion
This study assessed the environmental impact of synthesising the ionic liquid [C2C1im][OAc] on a laboratory scale. The findings presented are pertinent to the synthesis of all aprotic ionic liquids made by similar methods. Through comparative computational LCA, we evaluated the relative sustainability of three laboratory procedures for synthesizing [C2C1im][OAc]. The LCA results demonstrate that for synthesis at scales under 5 g, the ion-exchange route offers the lowest environmental impact (99.8 Pt), and we recommend this method for exploratory studies. The dimethyl carbonate route using a pressure tube showed similar impact (102.0 Pt), while the silver acetate route exhibited the highest environmental burden (133.5 Pt). When researchers need to prepare ionic liquids in greater amounts, the dimethyl carbonate route using a high-pressure benchtop reactor proves significantly more sustainable (14.8 Pt).Through hotspot analysis, we identified that solvents and auxiliary materials drive environmental impacts rather than reactants themselves. Specifically, ethyl acetate and acetonitrile used in precursor recrystallisation contribute substantially to human toxicity categories, while dry ice use significantly impacts global warming and ecotoxicity scores. Electricity consumption serves as a deciding factor for overall environmental damage across all routes, with the energy-intensive DMC pressure tube method consuming the most electricity per kilogram of ionic liquid produced. The silver mining process makes silver acetate particularly damaging, contributing substantially to mineral resource scarcity in addition to practical drawbacks including high cost, photodegradation, and difficult stoichiometry. We recommend specific protocol modifications to improve sustainability: researchers should extend rotary evaporation before Schlenk line drying to reduce energy consumption, use rotary evaporators with closed cooling systems for solvent removal, minimise overnight Schlenk line drying, avoid silver salts where possible, and plan synthesis quantities based on actual research needs.
Among the green metrics we evaluated, Effective Mass Yield (EMY) most closely paralleled the LCA results. Our findings indicate that no single green metric proves inherently superior. Calculating and comparing multiple metrics collectively provides a more comprehensive assessment when LCA is not feasible. We also identified electricity consumption as an easy-to-measure parameter that researchers can monitor using affordable standard electricity meters to identify process hotspots.
It should be noted that toxicological and life cycle data for ionic liquids and their precursors remain scarce, as current regulations do not require comprehensive testing for small-scale chemical production. As the ionic liquid industry matures and more environmental data becomes available, future LCA studies may provide increasingly accurate assessments.
Finally, we have demonstrated the value of LCA for laboratory-scale reactions, particularly for syntheses that are repeated frequently and have a choice of synthetic route. This study provides a framework for evaluating the environmental sustainability of chemical synthesis at the laboratory scale and identifies actionable modifications that researchers can implement immediately. Moving forward, we encourage chemists to conduct environmental assessments when feasible and include energy monitoring in their experiments.
Author contributions
Rhea Mathew contributed to conceptualisation, formal analysis, investigation, data curation, methodology, software, validation, visualisation, and writing – original draft. Sirui Chen contributed to the experimental investigation. Agnieszka Brandt-Talbot contributed to conceptualisation, funding acquisition, methodology, resources, supervision, writing – review & editing. Jacqueline S. Edge contributed to conceptualisation, funding acquisition, methodology, resources, supervision, writing – review & editing. Tom Welton contributed to conceptualisation, methodology, funding acquisition, project administration, resources, supervision, writing – review & editing. These author contributions follow the CRediT (https://credit.niso.org/) taxonomy (Contributor Roles Taxonomy).Conflicts of interest
There are no conflicts to declare.Data availability
Data supporting this article have been included as part of the supplementary information (SI). Supplementary information: details of the experimental and modelling work such as Nuclear Magnetic (NMR) Resonance spectra, Life Cycle Inventory (LCI) data, precursor LCI estimation, instrumentation details and energy required per instrument, and calculations of green metrics. See DOI: https://doi.org/10.1039/d5gc06385j.Data files with detailed LCA data have also been deposited on https://doi.org/10.6084/m9.figshare.31435540.
Acknowledgements
The authors extend their gratitude to Diego Freire Ordonez of Imperial College London for useful discussions regarding life cycle assessment.References
- J. Woolliams, M. Lloyd and J. D. Spengler, The case for sustainable laboratories: first steps at Harvard University, Int. J. Sustainability Higher Educ., 2005, 6, 363–382 CrossRef.
- LEAF – Laboratory Efficiency Assessment Framework, UCL.
- T. Freese, R. Kat, S. D. Lanooij, T. C. Böllersen, C. M. De Roo, N. Elzinga, M. Beatty, B. Setz, R. R. Weber and I. Malta, A guidebook for sustainability in laboratories, 2024 Search PubMed.
- T. Freese, N. Elzinga, M. Heinemann, M. M. Lerch and B. L. Feringa, The relevance of sustainable laboratory practices, RSC Sustainability, 2024, 2, 1300–1336 RSC.
- N. Winter, R. Marchand, C. Lehmann, L. Nehlin, R. Trapannone, D. Rokvić and J. Dobbelaere, The paradox of the life sciences: How to address climate change in the lab, EMBO Rep., 2023, 24, e56683 CrossRef CAS PubMed.
- P. T. Anastas and J. C. Warner, Green chemistry: theory and practice, Oxford university press, 2000 Search PubMed.
- C. Jiménez-González, D. J. Constable and C. S. Ponder, Evaluating the “Greenness” of chemical processes and products in the pharmaceutical industry—a green metrics primer, Chem. Soc. Rev., 2012, 41, 1485–1498 RSC.
- R. A. Sheldon, M. L. Bode and S. G. Akakios, Metrics of green chemistry: Waste minimization, Curr. Opin. Green Sustain. Chem., 2022, 33, 100569 CrossRef CAS.
- S. M. Kernaghan, T. Coady, M. Kinsella and C. M. Lennon, A tutorial review for research laboratories to support the vital path toward inherently sustainable and green synthetic chemistry, RSC Sustainability, 2024, 2, 578–607 RSC.
- J. Martínez, J. F. Cortés and R. Miranda, Green chemistry metrics, a review, Processes, 2022, 10, 1274 CrossRef.
- C. Apel, A. Sudheshwar, K. Kümmerer, B. Nowack, K. Midander, E. Strömberg and L. G. Soeteman-Hernández, Safe-and-sustainable-by-design roadmap: identifying research, competencies, and knowledge sharing needs, RSC Sustainability, 2024, 2, 2833–2838 RSC.
- E. Lucas, A. J. Martín, S. Mitchell, A. Nabera, L. F. Santos, J. Pérez-Ramírez and G. Guillén-Gosálbez, The need to integrate mass-and energy-based metrics with life cycle impacts for sustainable chemicals manufacture, Green Chem., 2024, 26, 9300–9309 RSC.
- S. Shaked, P. Crettaz, M. Saade-Sbeih, O. Jolliet and A. Jolliet, Environmental life cycle assessment, CRC Press, 2015 Search PubMed.
- M. L. Parisi, A. Dessì, L. Zani, S. Maranghi, S. Mohammadpourasl, M. Calamante, A. Mordini, R. Basosi, G. Reginato and A. Sinicropi, Combined LCA and green metrics approach for the sustainability assessment of an organic dye synthesis on lab scale, Front. Chem., 2020, 8, 214 CrossRef CAS PubMed.
- M. Mooney, A. J. Vreugdenhil and S. Shetranjiwalla, A toolkit of green chemistry and life-cycle analysis for comparative assessment in undergraduate organic chemistry experiments: synthesis of (E)-stilbene, J. Chem. Educ., 2020, 97, 1336–1344 CrossRef CAS.
- K. M. Reyes, K. Bruce and S. Shetranjiwalla, Green Chemistry, Life Cycle Assessment, and Systems Thinking: An Integrated Comparative-Complementary Chemical Decision-Making Approach, J. Chem. Educ., 2022, 100, 209–220 CrossRef.
- T. Welton, Room-temperature ionic liquids. Solvents for synthesis and catalysis, Chem. Rev., 1999, 99, 2071–2084 CrossRef CAS PubMed.
- E. L. Baker, A review of recent research on health effects of human occupational exposure to organic solvents: a critical review, J. Occup. Med., 1994, 1079–1092 CrossRef CAS PubMed.
- T. Welton, Ionic liquids in catalysis, Coord. Chem. Rev., 2004, 248, 2459–2477 CrossRef CAS.
- S. K. Singh and A. W. Savoy, Ionic liquids synthesis and applications: An overview, J. Mol. Liq., 2020, 297, 112038 CrossRef CAS.
- A. Brandt, J. Gräsvik, J. P. Hallett and T. Welton, Deconstruction of lignocellulosic biomass with ionic liquids, Green Chem., 2013, 15, 550–583 RSC.
- C. Verma, A. Mishra, S. Chauhan, P. Verma, V. Srivastava, M. A. Quraishi and E. E. Ebenso, Dissolution of cellulose in ionic liquids and their mixed cosolvents: A review, Sustainable Chem. Pharm., 2019, 13, 100162 CrossRef.
- A. Bengtsson, J. Bengtsson, K. Jedvert, M. Kakkonen, O. TanhuanpäÄ, E. Brännvall and M. Sedin, Continuous stabilization and carbonization of a lignin–cellulose precursor to carbon fiber, ACS Omega, 2022, 7, 16793–16802 CrossRef CAS PubMed.
- F. Zareiekordshouli, A. Lashanizadehgan and P. Darvishi, Study on the use of an imidazolium-based acetate ionic liquid for CO2 capture from flue gas in absorber/stripper packed columns: Experimental and modeling, Int. J. Greenhouse Gas Control, 2018, 70, 178–192 CrossRef CAS.
- S. Koutsoukos, J. Becker, A. Dobre, Z. Fan, F. Othman, F. Philippi, G. J. Smith and T. Welton, Synthesis of aprotic ionic liquids, Nat. Rev. Methods Primers, 2022, 2, 49 CrossRef CAS.
- J. C. Schleicher and A. M. Scurto, Kinetics and solvent effects in the synthesis of ionic liquids: imidazolium, Green Chem., 2009, 11, 694–703 RSC.
- A. Aupoix, B. Pégot and G. Vo-Thanh, Synthesis of imidazolium and pyridinium-based ionic liquids and application of 1-alkyl-3-methylimidazolium salts as pre-catalysts for the benzoin condensation using solvent-free and microwave activation, Tetrahedron, 2010, 66, 1352–1356 CrossRef CAS.
- E. K. Fields and S. Meyerson, Thermal and photochemical decomposition of silver carboxylates, J. Org. Chem., 1976, 41, 916–920 CrossRef CAS.
- Ionic Liquids, ed. B. Clare, A. Sirwardana and D. R. MacFarlane, 2010 Search PubMed.
- H. X. Zhu, J. S. Li, R. Xu and S. Y. Yang, An environmental friendly approach for the synthesis of the ionic liquid 1-ethyl-3-methylimidazolium acetate and its dissolubility to 1, 3, 5-triamino-2, 4, 6-trinitrobenzene, J. Mol. Liq., 2012, 165, 173–176 CrossRef CAS.
- Anonymous DuPont™ Ion Exchange Resins Selectivity Tech Fact.
- DuPont™ AmberLyst™ Fouling Limits Recommendations, 2022.
- J. L. Ferguson, J. D. Holbrey, S. Ng, N. V. Plechkova, K. R. Seddon, A. A. Tomaszowska and D. F. Wassell, A greener, halide-free approach to ionic liquid synthesis, Pure Appl. Chem., 2011, 84, 723–744 Search PubMed.
- M. Tiano, R. Clark, L. Bourgeois and M. C. Gomes, The dialkylcarbonate route to ionic liquids: purer, safer, greener?, Green Chem., 2023, 25, 2541–2558 RSC.
- R. M. Cuéllar-Franca, P. García-Gutiérrez, J. P. Hallett and N. Mac Dowell, A life cycle approach to solvent design: challenges and opportunities for ionic liquids – application to CO2 capture, React. Chem. Eng., 2021, 6, 258–278 RSC.
- V. G. Maciel, D. J. Wales, M. Seferin, C. M. L. Ugaya and V. Sans, State-of-the-art and limitations in the life cycle assessment of ionic liquids, J. Cleaner Prod., 2019, 217, 844–858 CrossRef CAS.
- R. M. Cuéllar-Franca, P. García-Gutiérrez, S. F. R. Taylor, C. Hardacre and A. Azapagic, A novel methodology for assessing the environmental sustainability of ionic liquids used for CO2 capture, Faraday Discuss., 2016, 192, 283–301 RSC.
- A. Mehrkesh and A. T. Karunanithi, Energetic ionic materials: How green are they? A comparative life cycle assessment study, ACS Sustainable Chem. Eng., 2013, 1, 448–455 CrossRef CAS.
- P. L. A. Alviz and A. J. Alvarez, Comparative life cycle assessment of the use of an ionic liquid ([Bmim] Br) versus a volatile organic solvent in the production of acetylsalicylic acid, J. Cleaner Prod., 2017, 168, 1614–1624 CrossRef.
- H. Baaqel, I. Díaz, V. Tulus, B. Chachuat, G. Guillén-Gosálbez and J. P. Hallett, Role of life-cycle externalities in the valuation of protic ionic liquids–a case study in biomass pretreatment solvents, Green Chem., 2020, 22, 3132–3140 RSC.
- G. International Organization for Standardization Switzerland, ISO 14044 (2006) Environmental management—Life cycle assessment—Requirements and guidelines, 2006.
- G. International Organization for Standardization Switzerland , ISO 14040 (2006) Environmental management—Life cycle assessment—Principles and framework, 2006.
- PRé Sustainability, SimaPro 9.5.0.2, 2023 Search PubMed.
- K. Ebel, H. Koehler, A. O. Gamer and R. Jäckh, in Ullmann's Encyclopedia of Industrial Chemistry, 2000 Search PubMed.
- S. Saxer, C. Marestin, R. Mercier and J. Dupuy, The multicomponent Debus–Radziszewski reaction in macromolecular chemistry, Polym. Chem., 2018, 9, 1927–1933 RSC.
- L. Rosate, S. Monette and M. Blais, Process for making a silver nitrate solution, EP0568259A1, 1993.
- W. Jiang, Y. Zeng, X. Wang, X. Yue, S. Yuan, H. Lu and B. Liang, Preparation of Silver Carbonate and its Application as Visible Light-driven Photocatalyst Without Sacrificial Reagent, Photochem. Photobiol., 2015, 91, 1315–1323 CrossRef CAS PubMed.
- V. Logvinenko, O. Polunina, Y. Mikhailov, K. Mikhailov and B. Bokhonov, Study of thermal decomposition of silver acetate, J. Therm. Anal. Calorim., 2007, 90, 813–816 CrossRef CAS.
- R. M. Felder, R. W. Rousseau and L. G. Bullard, Elementary principles of chemical processes, John Wiley & Sons, 2020 Search PubMed.
- M. A. J. Huijbregts, Z. J. N. Steinmann and P. M. F. Elshout, ReCiPe2016: a harmonised life cycle impact assessment method at midpoint and endpoint level, Int. J. Life Cycle Assess., 2017, 22, 138–147 CrossRef.
- M. Goedkoop, M. Oele, A. de Schryver, M. Vieira and S. Hegger, SimaPro database manual methods library, PRé Consultants, The Netherlands, 2008, pp. 22–25 Search PubMed.
- F. Verones, J. Bare, C. Bulle, R. Frischknecht, M. Hauschild, S. Hellweg, A. Henderson, O. Jolliet, A. Laurent and X. Liao, LCIA framework and cross-cutting issues guidance within the UNEP-SETAC Life Cycle Initiative, J. Cleaner Prod., 2017, 161, 957–967 CrossRef PubMed.
- REACH Regulation EC 1907/2006, 2007.
- UK REACH: Article 1, 2022.
- N. Mehmood, E. Husson, C. Jacquard, S. Wewetzer, J. Büchs, C. Sarazin and I. Gosselin, Impact of two ionic liquids, 1-ethyl-3-methylimidazolium acetate and 1-ethyl-3-methylimidazolium methylphosphonate, on Saccharomyces cerevisiae: metabolic, physiologic, and morphological investigations, Biotechnol. Biofuels, 2015, 8, 1–14 Search PubMed.
- J. Maculewicz, K. Świacka, P. Stepnowski, J. Dołżonek and A. Białk-Bielińska, Ionic liquids as potentially hazardous pollutants: Evidences of their presence in the environment and recent analytical developments, J. Hazard. Mater., 2022, 437, 129353 CrossRef CAS PubMed.
- Sigma-Aldrich. Ethyl Acetate Safety Data Sheet.
- Sigma-Aldrich. Acetonitrile Safety Data Sheet.
- National Toxicology Program, Strong Inorganic Acid Mists Containing Sulfuric Acid, 15th Report on Carcinogens, 2021.
- Sigma-Aldrich. Ethyl Bromide Safety Data Sheet.
- M. K. Vollmer, J. Mühle, S. Henne, D. Young, M. Rigby, B. Mitrevski, S. Park, C. R. Lunder, T. S. Rhee and C. M. Harth, Unexpected nascent atmospheric emissions of three ozone-depleting hydrochlorofluorocarbons, Proc. Natl. Acad. Sci. U. S. A., 2021, 118, e2010914118 CrossRef CAS PubMed.
- F. Graziosi, J. Arduini, F. Furlani, U. Giostra, L. J. Kuijpers, S. A. Montzka, B. R. Miller, S. J. O'Doherty, A. Stohl and P. Bonasoni, European emissions of HCFC-22 based on eleven years of high frequency atmospheric measurements and a Bayesian inversion method, Atmos. Environ., 2015, 112, 196–207 CrossRef CAS.
- M. J. Eckelman and T. E. Graedel, Silver emissions and their environmental impacts: a multilevel assessment, Environ. Sci. Technol., 2007, 41, 6283–6289 CrossRef CAS PubMed.
- D. Prat, A. Wells, J. Hayler, H. Sneddon, C. R. McElroy, S. Abou-Shehada and P. J. Dunn, CHEM21 selection guide of classical-and less classical-solvents, Green Chem., 2016, 18, 288–296 RSC.
- A. Raza, M. Ikram, S. Guo, A. Baiker and G. Li, Green synthesis of dimethyl carbonate from CO2 and methanol: new strategies and industrial perspective, Adv. Sustainable Syst., 2022, 6, 2200087 CrossRef CAS.
- National Grid Live, (accessed Aug 11 2025).
- A. R. N. Pontillo, S. Chen, D. F. Ordóñez, N. Mac Dowell, K. Lee and T. Welton, Fabricating lignocellulosic films as potential biobased plastics, RSC Sustainability, 2025, 3, 2301–2310 RSC.
- A. Brandt, J. P. Hallett, D. J. Leak, R. J. Murphy and T. Welton, The effect of the ionic liquid anion in the pretreatment of pine wood chips, Green Chem., 2010, 12, 672–679 RSC.
| This journal is © The Royal Society of Chemistry 2026 |










