Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Enzymatic polymerization of various biobased and biodegradable BHMTHF-based (co)polyesters
Cornelis
Post
 ab,
George
Karchilakis
ab,
George
Karchilakis
 ab,
Dina
Maniar
ab,
Dina
Maniar
 a,
Vincent S. D.
Voet
a,
Vincent S. D.
Voet
 b,
Rudy
Folkersma
b and
Katja
Loos
b,
Rudy
Folkersma
b and
Katja
Loos
 *a
*a
aMacromolecular Chemistry & New Polymeric Materials, Zernike Institute for Advanced Materials, University of Groningen, Nijenborgh 3, 9747 AG Groningen, the Netherlands. E-mail: k.u.loos@rug.nl
bCircular Plastics, Academy Tech & Design, NHL Stenden University of Applied Sciences, Van Schaikweg 94, 7811 KL Emmen, the Netherlands
First published on 5th February 2026
Abstract
2,5-Bishydroxymethyltetrahydrofuran (BHMTHF), also known as 2,5-tetrahydrofurandimethanol (THFDM), is a sugar-derived monomer containing a rigid alicyclic tetrahydrofuran structure. In this study, three series of BHMTHF-based (co)polyesters were enzymatically synthesized using various aliphatic comonomers and the furanic comonomers 2,5-bishydroxymethylfuran (BHMF) and 2,5-furandicarboxylic acid (FDCA). The length of the aliphatic comonomers had a significant effect on the Tg and complex viscosity. Similarly, a higher furan content, either BHMF or FDCA, in the copolyesters increased the Tg values and complex viscosities. Biodegradation experiments in activated sludge demonstrated that all aliphatic-BHMTHF-based polyesters were biodegradable, with rates ranging from 60 to 89% in 28 days. In addition, the furan-BHMTHF-based copolyesters also demonstrated a biodegradable behavior over time, without a significant influence of either the BHMF or FDCA content. Finally, the greenness of the enzymatic synthesis process was assessed based on atom economy (AE), reaction mass efficiency (RME), overall efficiency (OE), E-factor, and EcoScale. This work supports the tuneability of the thermal and rheological properties of BHMTHF-based (co)polyesters and reveals the biodegradability of these materials. This emphasizes that BHMTHF is a very promising biobased rigid monomer for the production of sustainable (co)polyesters.
Green foundation1. The conventional plastic economy relies largely on fossil-based polymers, bio-based polymers are renewable and more sustainable alternatives. Sugar-derived monomers, including BHMTHF, are considered key materials for the development of green polymers.2. The design and application of biodegradable polymers are important to reduce (micro)plastic pollution and accumulation in the environment. BHMTHF-based (co)polyesters are demonstrated to be biodegradable and have tunable properties, which makes BHMTHF a promising monomer for the production of environmentally friendly polyesters. 3. The traditional synthesis process of polyesters involves high temperature and toxic metal-based catalysts. Enzymatic polymerization is an efficient, selective, and low-temperature synthesis process, and is suitable for a wide range of monomers. BHMTHF-based (co)polyesters could be successfully synthesized via a sustainable enzymatic bulk polymerization process in relatively high molecular weight. |
Introduction
Sugar-derived monomers are key materials for producing renewable polymers due to their sustainable character, inexpensiveness, abundance and chemical diversity, and align with the principles of Green Chemistry.1 An important sugar-based intermediate is the biobased platform chemical 5-(hydroxymethyl)furfural (HMF), which can be converted into numerous different monomers that can be used for the synthesis of polyesters, polyamides and polyurethanes.2 The most famous HMF-derived monomer is the oxidized product 2,5-furandicarboxylic acid (FDCA), which is close to being produced at a large scale by the Dutch company Avantium.3Furthermore, HMF can also be reduced to yield monomers such as 2,5-bishydroxymethylfuran (BHMF), 2,5-bishydroxymethyltetrahydrofuran (BHMTHF), 1,6-hexanediol (HDO), and 1,2,6-hexanetriol (HT).4 Among these, BHMTHF, also reported as 2,5-tetrahydrofurandimethanol (THFDM) and 2,5-dihydroxymethyltetrahydrofuran (DHMTHF), is particularly attractive. Its alicyclic tetrahydrofuran structure in combination with two primary hydroxyl groups makes it structurally similar to BHMF, while providing the additional benefit of a remarkably higher thermal stability compared to its furan-based counterpart.5
This makes BHMTHF, in contrast to BHMF, suitable for high temperature bulk polymerization for the synthesis of diverse polyesters with unique properties. The group of Wei synthesized a variety of BHMTHF-based (co)polyesters via melt polymerization using an L-scale reactor. They synthesized poly(ethylene-co-tetrahydrofurandimethanol terephthalate) (PETT) wherein BHMTHF (THFDM) was demonstrated to be a suitable alternative to replace 1,4-cyclohexanedimethanol (CHDM), which is currently used in the industrial production of poly(ethylene terephthalate-co-cyclohexanedimethanol terephthalate) (PETG).6 In addition, they (co)polymerized BHMTHF with terephthalic acid (TA), FDCA, succinic acid, and adipic acid, which resulted in a polymeric material with a wide range of properties.7–9 The copolyester PETT is popular for fiber applications and was also synthesized by other groups to study melt spinning, electrospinning, crystallization behavior and mechanical performances.10–12 In general, the incorporation of BHMTHF units in copolyesters results in reduced crystallinity or even fully amorphous materials, relatively high Tg values and increased hydrophilicity.13–15 These features are attributed to the rigid tetrahydrofuran ring structure, which contains an electronegative ether group. These unique characteristics of the BHMTHF ring are also recognized in polyurethanes, which demonstrated self-healing behavior due to strong hydrogen bonding between the urethane groups and the oxygen functionality in the BHMTHF units.16–18
Although BHMTHF-based polymers are not extensively studied, it is mainly (co)polymerized with either FDCA or TA, while only succinic acid and adipic acid are used as aliphatic comonomers. Hence, it is important to polymerize BHMTHF with biobased aliphatic comonomers of various sizes to expand the library. In addition, conventional melt polycondensation is currently the only reported synthesis route. Enzymatic polymerization is demonstrated to be a sustainable, efficient and selective tool to produce various biobased polyesters.19–22 In addition, the use of toxic metal-based catalysts and high temperatures are avoided, which follows the Green Chemistry principles about efficient catalysis and less hazardous chemical syntheses. Due to BHMTHF's structural similarity to BHMF, it is expected that enzymatic polymerization could be a viable synthesis route as well.23,24 Furthermore, poly(butylene-co-tetrahydrofurandimethylene succinate) is shown to be prone to enzymatic degradation, which is another indication that enzymatic polymerization might be a promising alternative.9 These results indicate that BHMTHF-based polyesters are prone to biodegradation. Although enzymatic degradation is not the same as biodegradation, where polymers degrade by microorganisms to water, minerals, biomass, and CO2.25 The produced CO2 returns in the carbon cycle, or can be a valuable input for the chemical industry.26
In this work, BHMTHF is enzymatically polymerized with aliphatic comonomers that contain a varying number of methylene units. In addition, the polyester derived from BHMTHF and dimethyl adipate (poly(tetrahydrofurandimethylene adipate)) was copolymerized with either BHMF or diethyl FDCA in varying ratios. The impact of aliphatic spacer length and the furanic content (BHMF or FDCA) on the thermal characteristics, rheology and biodegradability are studied. The results give a broad perspective on the structure–property relationship of BHMTHF units in (co)polyesters.
Experimental
Materials
2,5-Bishydroxymethyltetrahydrofuran (BHMTHF, >98%, 90.6% cis, 9.4% trans) was purchased from AKOS GmbH (Germany). 2,5-Bis(hydroxymethyl)furan (BHMF, >97%) and 2,5-furan dicarboxylic acid (FDCA, >97%) were both obtained from Apollo Scientific. Dimethyl succinate (DMSuc, 98%), dimethyl glutarate (DMGlu, 99%), dimethyl adipate (DMAd, >99%), dimethyl pimelate (DMPim, 99%), dimethyl suberate (DMSub, 99%), chloroform (amylene stabilized, HPLC grade, >99.8%), and lipase acrylic resin (Candida antartica lipase B (iCALB), 5000 U g−1, recombinant, expressed in Aspergillus niger) were all purchased from Sigma-Aldrich. Dimethyl azelate (DMAze, >98%) and dimethyl sebacate (DMSeb, >98%) were both obtained from TCI EUROPE. Diethyl ether was purchased from Honeywell Research Chemicals and ethanol (absolute) was obtained from J.T. Baker. Ethyl acetate was purchased from Macron Fine Chemicals and sulfuric acid (96–98%) was obtained from Boom B.V. All chemicals were used as received. The wastewater treatment facility in Glimmen (the Netherlands) kindly supplied the activated sludge.Synthesis of diethyl furan-2,5-dicarboxylate
FDCA was converted into diethyl furan-2,5-dicarboxylate (DEFDCA) via esterification with ethanol.27 A round bottom flask was filled with 10.0 g (64 mmol) FDCA, 300 mL of ethanol and 0.8 mL concentrated sulfuric acid as a catalyst. The mixture was allowed to reflux for 24 h by using an oil bath. Then, the ethanol was removed under reduced pressure, and 200 mL of ethyl acetate was added to the reaction product. The organic layer was washed several times with water and once with concentrated NaHCO3. Anhydrous MgSO4 was used to remove water traces and the ethyl acetate was removed under reduced pressure. The obtained DEFDCA appeared as white crystals in a yield of 81% and its structure was confirmed by 1H-NMR: (600 MHz, DMSO-d6), δ (ppm): 7.39 (s, 2H, CH-furan), 4.32 (m, 4H, CH2-ethyl group), 1.29 (t, 6H, CH3-ethyl group).Synthesis of BHMTHF-based (co)polyesters
All polyesters were synthesized via the same procedure, only the ratios and type of monomers varied. The reactions were performed in a 25 mL three-necked round bottom flask, which was magnetically stirred and connected to a distillation setup and Schlenk line. The BHMTHF-aliphatic polyesters were produced on a 2.50 g scale, at a BHMTHF![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) dimethyl ester molar ratio of 1.00
dimethyl ester molar ratio of 1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
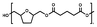
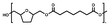
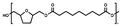
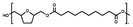
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.00, and 10% w/w iCALB was added as a catalyst (Fig. 1). The same scale and catalyst loadings were used for the synthesis of the copolyesters poly(tetrahydrofurandimethylene adipate-co-tetrahydrofurandimethylene 2,5-furandicarboxylate) (PTAd-TF) and poly(tetrahydrofurandimethylene adipate-co-2,5-furandimethylene adipate) (PTAd-FAd)(Fig. 2), and the feed ratios are provided in Table 2.
1.00, and 10% w/w iCALB was added as a catalyst (Fig. 1). The same scale and catalyst loadings were used for the synthesis of the copolyesters poly(tetrahydrofurandimethylene adipate-co-tetrahydrofurandimethylene 2,5-furandicarboxylate) (PTAd-TF) and poly(tetrahydrofurandimethylene adipate-co-2,5-furandimethylene adipate) (PTAd-FAd)(Fig. 2), and the feed ratios are provided in Table 2.
 | ||
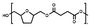
| Fig. 1 Enzymatic bulk polymerization of BHMTHF and aliphatic dimethyl esters with a varying number of methylene units x. | ||
The reaction mixture was first maintained at 100 °C for 24 h under a mild argon flow. Then, the temperature was increased to 120 °C, and the system was gently switched to vacuum (2 × 10−2 mbar) and maintained for another 24 h. Finally, the temperature was increased to 130 °C for the last 7 h of the experiment. The obtained polymer was dissolved in chloroform, separated from the enzyme using a needle and syringe, precipitated in diethyl ether, centrifuged (10 min, 4500 rpm) and dried in the vacuum oven at 80 °C for 24 h. Organic solvents were required for purification and product recovery, but could be eliminated at larger scales, where polymers can be directly extruded from the reactor. All synthesized polyesters appeared as a viscous colorless liquid except PTAd-FAd_100, which appeared as a solid. The detailed NMR data of all synthesized polyesters are provided in the SI Fig. S1–S19.
Structural characterization
A Bruker AVANCE NEO 600 MHz NMR spectrometer, equipped with a BBFO probe and DMSO-d6 as the solvent, was used for proton nuclear magnetic resonance (1H-NMR) measurements. Carbon-13 nuclear magnetic resonance (13C-NMR), Heteronuclear Single Quantum Coherence (HSQC), Heteronuclear Multiple Bond Correlation (HMBC) and Correlated Spectroscopy (COSY) were performed at the same device and used to identify all signals of BHMTHF and its derived polymers.The number average molecular weight ( ) and weight average molecular weight (
) and weight average molecular weight ( ) were determined via gel permeation chromatography (GPC). The analyses were performed on a Hewlett Packard 1100 device with three MIXED-E columns (Agilent Technologies PL, 300 × 7.5 mm 3 μm) equipped with a GBC 1240 refractive index detector. THF, stabilized with BHT, served as the solvent and eluent at a flow rate of 1 mL min−1 at 40 °C. The polymer samples were prepared at a concentration of 5 mg mL−1, and toluene acted as an internal standard. A conventional calibration curve based on monodispersed polystyrene (PS) standards was used to calculate the relative molecular weights.
) were determined via gel permeation chromatography (GPC). The analyses were performed on a Hewlett Packard 1100 device with three MIXED-E columns (Agilent Technologies PL, 300 × 7.5 mm 3 μm) equipped with a GBC 1240 refractive index detector. THF, stabilized with BHT, served as the solvent and eluent at a flow rate of 1 mL min−1 at 40 °C. The polymer samples were prepared at a concentration of 5 mg mL−1, and toluene acted as an internal standard. A conventional calibration curve based on monodispersed polystyrene (PS) standards was used to calculate the relative molecular weights.
The degree of polymerization (DP) of the copolyesters was determined as follows:
 | (1) |
Thermal stability analysis
The thermal stability of the (co)polyesters was investigated via thermogravimetric analysis (TGA) on a TA-Instruments Discovery TGA 5500. To eliminate solvent residues, the samples were first heated to 80 °C for 1 h in a nitrogen environment. The samples were then heated from room temperature to 700 °C at a steady rate of 10 °C min−1.The glass transition temperatures (Tg) and the melting points (Tm) of the polymers were determined by modulated differential scanning calorimetry (mDSC) analysis. A TA-Instruments Q1000 DSC was used to subject the samples (5–6 mg) to a heat-cool-heating cycle ranging from −70 to 150 °C under a nitrogen atmosphere. The average heating rate was set to 3 °C min−1 with a temperature modulation period of 60 s and an amplitude of 0.50 °C.
Rheological analysis
The rheology of the polyesters was analyzed on a TA-Instruments HDR20 rheometer, equipped with an 8 mm plate–plate geometry. The experiments were performed at 40 °C, well above all Tg values, under a nitrogen atmosphere and a constant gap size, ranging from 0.5 to 1.0 mm between the different samples. A dynamic strain sweep experiment was conducted to determine the linear viscoelastic regime at a constant oscillation frequency (100 rad s−1), while the strain ranged from 0.1 to 10%. Subsequently, a dynamic frequency sweep was performed at a constant strain of 1%, well within the linear viscoelastic regime, while the frequency ranged from 0.01–100 rad s−1.Biodegradability examination
A Lovibond® Water testing BOD-System BD 600 setup was used to study the biodegradability of the BHMTHF-based polyesters in activated sludge. The method was based on the OECD 301F manometric respirometry test guidelines and is described in detail in our previous work.28,29 Briefly, the experiments are based on the biochemical oxygen demand (BOD) and the theoretical oxygen demand (ThOD) of the tested sample. The BOD ranges from 0 to 400 mg L−1 oxygen and the ThOD is used to determine the mass of each sample that is required to reach the theoretical maximum BOD value of 400 mg L−1 oxygen. The activated sludge, obtained from a wastewater facility, was aerated for 3 days prior to use, diluted with water and provided with nutrient salts. The experiments were performed for 28 days and each sample was measured in duplicate. A reference (sodium acetate) and a control (no sample) were also included in duplicate to verify the activity of the sludge and the measurement range. The percentage of biodegradation was based on the averaged values and corrected for the average BOD value of the control experiment: | (2) |
Green metrics calculations
The greenness of the polymerization protocols was evaluated using a set of complementary green metrics, namely atom economy (AE), reaction mass efficiency (RME), overall efficiency (OE), E-factor, and EcoScale.30,31 These metrics were calculated at the level of the polymerization step, from diester and diol to isolated (co)polyester, following established definitions used for synthetic organic and polymer chemistry.32AE was determined from the reaction stoichiometry as the ratio between the molecular weight of the ideal repeating unit and the total molecular weight of all reactants entering the polymer backbone, assuming methanol or ethanol to be the only stoichiometric by-products (eqn (3)). RME was obtained by dividing the mass of the isolated polymer by the total mass of all reactants, and incorporates, therefore, the yield and non-stoichiometric excess of BHMTHF or diester(eqn (4)). OE is defined as the ratio between RME and AE and was used as an integrated measure of the transformation efficiency (eqn (5)).
 | (3) |
 | (4) |
 | (5) |
The E-factor was calculated as the mass ratio of all waste generated to the mass of isolated (co)polyester (eqn (6)). In accordance with common practice, water was excluded from the E-factor calculations, whereas all organic solvents, immobilized enzyme, and other auxiliaries used during the polymerization and work-up were counted as waste.33 Hence, the reported E-factors represent conservative laboratory-scale values that do not account for solvent recycling or enzyme reuse.
 | (6) |
EcoScale scores were determined using the semi-quantitative methodology proposed by Van Aken et al., in which an ideal score of 100 is reduced by penalty points associated with low yields, hazardous or expensive reagents and catalysts, non-preferred solvents, high temperatures, long reaction times, and demanding work-up.31 The input parameters for the EcoScale calculations (yields, reagents, solvent types, temperature, and reaction time) were taken directly from the experimental procedures used for each polymerization.
Results and discussion
Synthesis and structural confirmation
Three different series of BHMTHF-based polyesters were successfully polymerized in bulk by using the enzyme iCALB as a catalyst. In the first series, BHMTHF was reacted with aliphatic dimethyl esters of various sizes, and the results are provided in Table 1. The use of diesters instead of dicarboxylic acids violates the principles of Green Chemistry about atom economy and reducing derivatives. However, the reaction kinetics of dimethyl esters are higher, which improves the efficiency and reduces overall energy consumption, while the produced methanol could be recycled. ), weight average molecular weight (
), weight average molecular weight ( ), polydispersity (Đ), degree of polymerization (DP), glass transition temperature (Tg), and 5% and 50% decomposition temperatures (Td5%, Td50%)
), polydispersity (Đ), degree of polymerization (DP), glass transition temperature (Tg), and 5% and 50% decomposition temperatures (Td5%, Td50%)
The number average molecular weight ( ) ranged from 5500 to 11
) ranged from 5500 to 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 g mol−1, corresponding to a degree of polymerization (DP) ranging from 26 to 41. The use of DMSuc and DMPim as comonomers resulted in slightly lower
200 g mol−1, corresponding to a degree of polymerization (DP) ranging from 26 to 41. The use of DMSuc and DMPim as comonomers resulted in slightly lower  and DP values, expected due to lower reaction kinetics, which was also observed in our previous work about BHMF-based polyesters.29
and DP values, expected due to lower reaction kinetics, which was also observed in our previous work about BHMF-based polyesters.29
The second series involves copolyesters consisting of BHMTHF, BHMF, and DMAd, wherein the BHMTHF content was gradually increased at the cost of BHMF content (Table 2a). The  values varied from 6600 to 11
values varied from 6600 to 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1 and there was no significant effect of the BHMTHF/BHMF content on the molecular weight of the copolyesters. This implies a similar reactivity of BHMTHF and BHMF toward DMAd during enzymatic polymerization with iCALB. However, the polyester consisting of only BHMF (PTAd-FAd_100) demonstrated a lower
000 g mol−1 and there was no significant effect of the BHMTHF/BHMF content on the molecular weight of the copolyesters. This implies a similar reactivity of BHMTHF and BHMF toward DMAd during enzymatic polymerization with iCALB. However, the polyester consisting of only BHMF (PTAd-FAd_100) demonstrated a lower  compared to the pure polyester from BHMTHF and DMAd (PTAd-FAd_00, or PTAd). Noteworthily, the polymerization reactions started at 100 °C, while the synthesis of BHMF-based homopolyesters was initiated at 70 °C in our previous work due to BHMF's limited thermal stability.29,34 The possibility of starting the polymerization at a higher temperature was attributed to a higher purity of the BHMF used in this study. BHMF is commonly derived from the thermally less stable intermediate HMF, and trace amounts might influence the overall thermal stability.35
compared to the pure polyester from BHMTHF and DMAd (PTAd-FAd_00, or PTAd). Noteworthily, the polymerization reactions started at 100 °C, while the synthesis of BHMF-based homopolyesters was initiated at 70 °C in our previous work due to BHMF's limited thermal stability.29,34 The possibility of starting the polymerization at a higher temperature was attributed to a higher purity of the BHMF used in this study. BHMF is commonly derived from the thermally less stable intermediate HMF, and trace amounts might influence the overall thermal stability.35
 ), weight average molecular weight (
), weight average molecular weight ( ), polydispersity (Đ), degree of polymerization (DP), glass transition temperature (Tg), melting temperature (Tm), and 5% and 50% decomposition temperatures (Td5%, Td50%)
), polydispersity (Đ), degree of polymerization (DP), glass transition temperature (Tg), melting temperature (Tm), and 5% and 50% decomposition temperatures (Td5%, Td50%)
| (a) Polymer | BHMF feed (%) | BHMF polymer (%) | (g mol−1) | (g mol−1) | Đ | DP | T g (°C) | T m (°C) | T d5% (°C) | T d50% (°C) |
|---|---|---|---|---|---|---|---|---|---|---|
| PTAd-FAd_100 | 100 | 100 | 6600 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
1.6 | 28 | −18.8 | 65.5 | 271 | 293 |
| PTAd-FAd_75 | 75 | 74 | 9800 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
1.5 | 41 | −22.0 | 43.6 | 276 | 305 |
| PTAd-FAd_50 | 50 | 50 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
1.7 | 44 | −23.3 | — | 276 | 370 |
| PTAd-FAd_25 | 25 | 24 | 8600 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600 |
1.9 | 36 | −24.8 | — | 298 | 375 |
| PTAd-FAd_00 | 0 | 0 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 900 |
1.6 | 45 | −27.5 | — | 303 | 365 |
| (b) Polymer | FDCA feed (%) | FDCA polymer (%) | (g mol−1) | (g mol−1) | Đ | DP | T g (°C) | T m (°C) | T d5% (°C) | T d50% (°C) |
|---|---|---|---|---|---|---|---|---|---|---|
| PTAd-TF_100 | 100 | — | <500 | <500 | — | — | — | — | — | — |
| PTAd-TF_75 | 75 | 69 | 1000 | 1300 | 1.3 | 4 | −3.0 | — | 267 | 385 |
| PTAd-TF_50 | 50 | 53 | 4600 | 7100 | 1.6 | 18 | −4.5 | — | 332 | 392 |
| PTAd-TF_25 | 25 | 29 | 8500 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
1.8 | 35 | −9.0 | — | 338 | 394 |
| PTAd-TF_00 | 0 | 0 | 9100 | 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
1.9 | 38 | −29.9 | — | 322 | 393 |
A different phenomenon was observed in the third series, which covered copolyesters based on BHMTHF, DEFDCA, and DMAd (Table 2b). The  values decreased significantly for higher contents of DEFDCA in the feed. In addition, the enzymatic polymerization of only BHMTHF and DEFDCA was unsuccessful since only oligomer formation was observed. This is attributed to the limited enzymatic activity towards polymerization of two cyclic monomers, e.g., FDCA and BHMTHF. This substrate limitation effect of the active site of iCALB was also observed for copolyesters based on dimethyl FDCA, BHMF, and aliphatic diols.21
values decreased significantly for higher contents of DEFDCA in the feed. In addition, the enzymatic polymerization of only BHMTHF and DEFDCA was unsuccessful since only oligomer formation was observed. This is attributed to the limited enzymatic activity towards polymerization of two cyclic monomers, e.g., FDCA and BHMTHF. This substrate limitation effect of the active site of iCALB was also observed for copolyesters based on dimethyl FDCA, BHMF, and aliphatic diols.21
It is worth noticing that iCALB easily polymerizes BHMTHF with DMAd, but FDCA with BHMTHF does not even result in oligomers under the same conditions, while BHMTFH and FDCA contain both a five-membered ring structure. This lipase consists of a Ser-His-Asp catalytic triad, which is surrounded by a relatively large hydrophobic pocket to allow absorption on hydrophobic surfaces, a mechanism called interfacial activation.36,37 The catalytic transesterification mechanism involves first the incorporation and activation of the carbonyl group in the active site and subsequently the liberation of the alcohol group, as described in detail by Hevilla et al. (2021).38 This is followed by the nucleophilic attack of another primary alcohol group and the release of the product with the newly formed ester bond.20,22 This means that the chemical structure and bulkiness of the diacid, dimethyl, or diethyl ester is key to interacting and fitting in the active site and thus the possibility of polymerization. In addition, the results in this work demonstrate that the bulkiness of the diol, i.e., BHMTHF or BHMF, has no significant limitation.
Although iCALB has difficulties with polymerization of two cyclic monomers, it proved to be a very selective and efficient catalyst at relatively low temperature, which makes it an advantageous tool for monomers with limited thermal and chemical stability, such as BHMF, and avoids the use of toxic metal-based catalysts and solvents. This aligns with the principles of Green Chemistry related to less hazardous chemical syntheses, designing safer chemicals, and efficient catalysis.
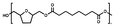
The polymer structures were confirmed by NMR analysis. The 1H-NMR spectrum of PTAd, named as PTAd-TF_00 and PTAd-FAd_00 in the two copolyester series, is provided as a representative example and depicted in Fig. 3. The spectra of the other BHMTHF-based polyesters are provided in the SI (Fig. S1–S6). The 13C-NMR, COSY, HSQC, and HMBC spectra (SI, Fig. S7–S10) were used to assign all protons to their corresponding signals in the 1H-NMR spectrum, due to overlapping signals of the protons “3” and “4” of the tetrahydrofuran ring, caused by the presence of the chiral carbons. The polymerization was confirmed by the substantial decrease of BHMTHF hydroxyl groups (“1”) and methoxy groups of DMAd (“7”), and the appearance of the protons next to the formed BHMTHF ester group (“3” and “4”).
 | ||
| Fig. 3 1H-NMR spectrum of poly(tetrahydrofurandimethylene adipate) (PTAd, PTAd-TF_00, PTAd-FAd_00) in DMSO-d6. | ||
The furan content of the copolyesters, either BHMF or FDCA content, was derived from 1H-NMR spectra as well, which are provided in the SI (Fig. S11–S19). In case of the BHMF-based copolyesters, the integrated signals of the CH2 groups of the furan next to the ester bond (“8”) and the similar peaks of BHMTHF units (“3” and “4”) were used to determine the actual furan content in the copolymers (eqn (7)). The FDCA content was derived from the integrated BHMTHF peaks “3” and “4” for the BHMTHF-DMAd segments, and similar signals at slightly higher ppm for the BHMTHF-FDCA segments (“8” and “9”) (eqn (8)).
The results are provided in Table 2a and b, and reveal that the feed ratios are similar to the final ratios in the case of the BHMF-BHMTHF-based and FDCA-BHMTHF-based copolyesters. The oligomer PTAd-TF_75 yielded a furan ratio of 69%, however this value is underestimated due to the presence of relatively large DEFDCA end groups that overlap with the BHMTHF ester bonds.
 | (7) |
 | (8) |
Thermal behavior
The thermal stability of the BHMTHF-based polyesters was determined by TGA analysis and the results of the first series are given in Table 1, while the curves are depicted in the SI (Fig. S20). All polyesters demonstrated a high thermal stability with Td5% and Td50% values ranging from 285 to 342 °C and 387 to 420 °C, respectively. This illustrates that BHMTHF-based polyesters have enhanced thermal stability compared to their BHMF-based polyester counterparts.24,29 The size of the aliphatic spacer had no significant influence on the thermal degradation behavior, attributed to their chemical similarities.The thermal behavior of the polyesters was analyzed by modulated DSC experiments. The Tg values of the BHMTHF-based polyesters from the first series were obtained from the second reversing heating ramp to prevent interference with the relaxation enthalpy. Fig. 4 demonstrates the decreasing quadratic behavior of the Tg values as a function of the aliphatic spacer, i.e., the number of methylene units in the aliphatic comonomer. A longer aliphatic spacer length increases chain flexibility and mobility, while steric hindrance decreases due to the relatively lower BHMTHF content for a higher number of methylene units. This thermal behavior is very similar to the BHMF-based polyesters that are synthesized with the same aliphatic comonomers; only the absolute Tg values of the BHMF-based polyesters are approximately 20 °C higher. This difference is attributed to the planar and aromatic structure of BHMF, which is absent for BHMTHF.
 | ||
| Fig. 4 T g values of BHMTHF- and BHMF-based polyesters containing different numbers of methylene units in the comonomer segment, and the structural representations of the polyesters. Data of the BHMF-based polyesters is obtained from previous work.29 | ||
All BHMTHF-based polymers from the first series lack a melting endotherm and are therefore completely amorphous. This corresponds well with the literature, for PTSuc and PTAd, respectively, and their appearance as a viscous liquid.8 The lack of crystallinity was attributed to the rigidity of the ring structure of BHMTHF, which disrupts interactions between the chains.15
The Td5% and Td50% of the two series of copolyesters are given in Table 2a and b for the BHMF and FDCA-based copolyesters, respectively. The thermal stability of the BHMF-based copolyesters (PTAd-FAd) increased for higher BHMTHF content, as represented by the Td5% values. In addition, these copolyesters demonstrated a twostep degradation profile (Fig. S21), which corresponded well with the degradation profiles of the pure polyesters and could be linked to the composition of the copolyesters.
The thermal stabilities of the FDCA-based copolyesters were all relatively high and similar in magnitude. However, the oligomer PTAd-TF_75 demonstrated a lower Td5% value, which is attributed to its significantly lower molecular weight.
The thermal behavior of the two series of BHMTHF-based copolyesters are depicted in Fig. 5. The Tg values of the BHMF-BHMTHF-based copolyesters increased linearly with a higher furan content as a result of increased structural rigidity. In other words, the Tg values of the copolyesters align well between the values of the two homopolyesters PTAd-FAd_100 (−19 °C) and PTAd-FAd_00 (−28 °C), and correspond well with literature values of the same homopolyesters, −13 and −33 °C, respectively.8,29
The Tg values of the FDCA-BHMTHF-based copolyesters increased more drastically for a higher furan content. This behavior is expected due to the high Tg value of the homopolyester PTAd-TF_100 (poly(2,5-tetrahydrofurandimethanol 2,5-furandicarboxylate) (PTF)), which could not be enzymatically synthesized, but literature reported values around 75 °C.7,13 The Tg value of the oligomer PTAd-TF_75 is included, however, is expected to be on the low side due to the limited molecular weight.
The BHMF-based polyester PFAd, synthesized in our previous work, is known to be a semicrystalline material with a relatively low Tm (63 °C).29 This Tm (65 °C) was also observed in the first heating ramp of the same polyester (PTAd-FAd_00) in this work (Fig. S26). In addition, the copolyester PTAd-FAd_25 also demonstrated a Tm (44 °C), and was considerably lower in magnitude, which indicated a significant reduction in degree of crystallinity. None of the other copolyesters, from both series, demonstrated a melting endotherm, attributed to BHMTHF's ability to hinder crystallization.7
The modulated DSC curves of the second reversing heating ramps of all BHMTHF-based (co)polyesters are provided in the SI (Fig. S23–S25).
Rheology
The mechanical behavior of the BHMTHF-based polyesters was analyzed by rheology. A strain sweep was initially performed to determine the linear viscoelastic behavior at 40 °C, well above all Tg values. Subsequently, a frequency sweep was applied to study the behavior at various shear stresses and the results of the complex viscosity versus frequency of the first series BHMTHF-based homopolyesters are shown in Fig. 6a. In all cases, the amorphous polyesters behaved like low viscous Newtonian fluids, since the complex viscosity was hardly influenced by the frequency. Only a minor shear-thinning behavior was observed for the polymers PTPim, PTSub, PTAze, and PTSeb at higher frequencies. The low frequency complex viscosity (1 rad s−1) of each polyester was plotted versus the number of methylene units in the aliphatic comonomer (Fig. 6b). This demonstrated a decreasing viscosity for BHMTHF-based polyesters containing a longer aliphatic segment. This phenomenon was also observed for BHMF-based polyesters obtained from the same aliphatic comonomers.29The same rheology experiments were performed on the two series of copolyesters. The low frequency complex viscosities were derived from the frequency sweeps (Fig. S27 and S28), and are plotted here versus the furan content, either BHMF or FDCA. Fig. 6c visualizes that the incorporation of FDCA results generally in higher strength materials with respect to the BHMF-based copolyesters, which aligns with the Tg results. In addition, in both cases, a higher furan content resulted in higher viscosities. The copolyester PTAd-TF_75 is an outlier in this trend; however, the low strength was attributed to its low molecular weight  . The polymers PTAd-TF_100 and PTAd-FAd_100 demonstrated a very similar viscosity, which is expected given that they are structurally identical polyesters (PTAd) with similar
. The polymers PTAd-TF_100 and PTAd-FAd_100 demonstrated a very similar viscosity, which is expected given that they are structurally identical polyesters (PTAd) with similar  . The polymer PTAd-FAd_100 was not subjected to the rheology measurements, since its Tm (65 °C) is higher than the experimental temperature (40 °C). PTAd-FAd_75 was also observed as a semi-crystalline material (Tm = 44 °C), but its melting endotherm was very low in magnitude and the material behaved like a viscoelastic material during the strain and frequency sweeps. Therefore, this data point is still included in Fig. 6c, but could have been influenced by minor crystalline phases in the material during the measurement.
. The polymer PTAd-FAd_100 was not subjected to the rheology measurements, since its Tm (65 °C) is higher than the experimental temperature (40 °C). PTAd-FAd_75 was also observed as a semi-crystalline material (Tm = 44 °C), but its melting endotherm was very low in magnitude and the material behaved like a viscoelastic material during the strain and frequency sweeps. Therefore, this data point is still included in Fig. 6c, but could have been influenced by minor crystalline phases in the material during the measurement.
In summary, the rheological behavior of the BHMTHF-based (co)polyesters was demonstrated to be tunable by varying the number of methylene units or the furan content. In addition, it indicates their behavior during (re)processing and confirms their recyclability to prevent waste and increase their sustainability.
Biodegradation
The biodegradability of the BHMTHF-based polyesters was tested in activated sludge, which consists of various naturally occurring microorganisms, and is the most common inoculum, and is seen as a reliable method to study the biological degradation of polymers.39 Although the biodegradability of polymers is dependent on multiple parameters, for example, environmental conditions and the type of microorganisms, the polymeric properties play an important role as well. First of all, the amorphous parts of semi-crystalline polymers are more prone to biodegradation than the crystalline phases.40 Furthermore, aliphatic polyesters generally have a higher rate of biodegradation compared to aliphatic–aromatic polyesters.41,42Fig. 7a demonstrates the percentage of biodegradation versus time for the first series of BHMTHF-based polyesters, and supports this theory. All these polyesters demonstrated a clear biodegradation behavior over time in activated sludge. The percentage of biodegradation of days ranged from 60 to 89%, while there was no discernible relationship observed between the chemical structure and degree of biodegradation.
The biodegradation behavior of the BHMF-BHMTHF-based copolyesters is depicted in Fig. 7b. All copolyesters were partially degraded in the active sludge, and the percentage of biodegradation ranged from 60 to 78%. Similar results are obtained from the FDCA-BHMTHF-based polyesters, which resulted in biodegradation rates ranging from 44 to 81% (Fig. 7c). In both series, the copolyester composition did not influence the biodegradation performances significantly. This is attributed to the amorphous morphology and low Tg values of all these copolymers, implying a similar physical appearance.
The pure BHMF-based polyester PTAd-FAd_100 (PFAd) demonstrated a remarkably lower degree of biodegradation (8%) compared to the copolyesters, while the biodegradation result of the same polyester in our previous work was also higher (22%).29 The difference regarding the BHMF-BHMTHF-based copolyesters is attributed to the differences in crystallinity. PTAd-FAd_100 demonstrated a clear melting peak, while PTAd-FAd_75 showed only a small melting endotherm and the other copolyesters were completely amorphous (Table 2). The lower percentage of biodegradation with respect to our previous result of the same polyester is explained by a difference in surface area. In this work, all viscous polymers were attached to the stirring bar at the start of the experiments, while PTAd-FAd_100 was first melted and subsequently also attached to the stirring bar for consistency in surface area. In our previous work, all polyesters were added as a fine powder, implying a higher surface area and enhanced biodegradation.
All polymers are measured in duplicate, except for the polymers highlighted (*) due to technical failure of the setup. However, the simplex data of these polymers are in line with expectations and the other polymers tested in duplicate, and are therefore still considered to give a good estimation of the biodegradation behavior. The minor fluctuations in the obtained BOD values and calculated percentages of biodegradation are attributed to relatively small variations in the surrounding temperature during the experiments.
In summary, the percentage of biodegradation after 28 days ranged from 44 to 89% for all BHMTHF-based (co)polyesters, and can therefore be considered biodegradable. This is in line with expectations due to their similarities with biodegradable BHMF-based polyesters and their amorphous morphology.29 The biodegradability of these polymers prevents the formation and accumulation of microplastics, which aligns with the principles of Green Chemistry about waste prevention, safer chemicals, design of degradation, and pollution prevention.
Green characteristic analysis
The greenness of the enzymatic bulk polymerizations was quantified using AE, RME, OE, E-factor and EcoScale (Table 3) in line with recent recommendations on multi-metric assessment of green chemistry processes.43 For the BHMTHF homopolyesters (PTSuc–PTSeb), AE values are in a narrow range of approximately 77 to 82%, reflecting the favorable stoichiometry of diester–diol polycondensation with only methanol as byproduct. RME values between roughly 48 and 60% and OE values between about 60 and 77% indicate that the mass efficiency of these bulk polymerizations is mainly limited by the isolated yield rather than by intrinsic stoichiometric constraints. The corresponding E-factors (67–84) are in the range of pharmaceuticals at an industrial scale, due to the use of solvent for purification and product collection.33 However, the use of solvents could be limited or eliminated in the case of upscaling, where the polymer product is commonly extruded directly from the reactor, which would drastically reduce the E-factor for all synthesized polymers. EcoScale scores (63–75) place all BHMTHF-based homopolyester syntheses firmly in the “acceptable” region (75–50), with PTGlu and PTSeb approaching the threshold for “excellent” protocols (>75), mainly attributed to their higher yields.| Polymer | AE (%) | RME (%) | OE (%) | E-factor (kg/kg) | EcoScale |
|---|---|---|---|---|---|
| (a) | |||||
| PTSuc | 77 | 48 | 62 | 84 | 67 |
| PTGlu | 78 | 60 | 77 | 67 | 75 |
| PTAd | 79 | 58 | 73 | 69 | 73 |
| PTPim | 80 | 50 | 62 | 81 | 64 |
| PTSub | 81 | 49 | 61 | 82 | 64 |
| PTAze | 82 | 49 | 60 | 82 | 63 |
| PTSeb | 82 | 54 | 66 | 74 | 69 |
| (b) | |||||
| PTAd-FAd_100 | 79 | 56 | 71 | 73 | 72 |
| PTAd-FAd_75 | 79 | 56 | 71 | 73 | 71 |
| PTAd-FAd_50 | 79 | 56 | 71 | 72 | 72 |
| PTAd-FAd_25 | 79 | 48 | 61 | 85 | 66 |
| PTAd-FAd_00 | 79 | 55 | 70 | 74 | 71 |
| (c) | |||||
| PTAd-TF_100 | 73 | — | — | — | — |
| PTAd-TF_75 | 75 | 49 | 66 | 82 | 61 |
| PTAd-TF_50 | 76 | 49 | 64 | 83 | 60 |
| PTAd-TF_25 | 78 | 53 | 69 | 76 | 65 |
| PTAd-TF_00 | 79 | 59 | 74 | 69 | 68 |
A similar picture emerges for the BHMF-BHMTHF-based copolyesters (PTAd-FAd series). Introduction of BHMF resulted in similar AE (79%) relative to the purely aliphatic PTAd, as expected from the similarities in molecular weight between BHMF and BHMTHF. In addition, RME (48–56%) and OE (61–71%) remain comparable to those of the homopolyesters due to similarities in yield. The E-factors ranged from 72 to 84 and were of similar magnitude as the aliphatic homopolymers due to an identical experimental procedure. EcoScale scores of 66 to 72 again classify these syntheses as “acceptable”, with no significant influence of the BHMF content.
For the FDCA-BHMTHF-based copolyesters (PTAd-TF series), the metrics highlight both strengths and clear opportunities for further improvement. In the case of the FDCA-BHMTHF polymer (PTAd-TF_100), only the AE is presented since the enzymatic polymerization was unsuccessful for these two cyclic monomers, and the obtained oligomers would represent unrealistic values for the other green metrics.
All other copolyesters in this series (PTAd-TF_75–PTAd-TF_00) show AE values of roughly 75 to 79%, RME values of 49 to 59% and OE in the range of 64 to 74%, with E-factors of 69 to 83 and EcoScale scores of 60 to 68. This shows that partial substitution of adipate by FDCA, enabling higher glass transition temperatures, complex viscosities, and expected improved barrier properties as reported for related furanic copolyesters,44 can be achieved without a significant penalty in the reaction-level green metrics.
This highlights that the distribution of AE, RME, OE, E-factors, and EcoScale values across the PTAd-TF and PTAd-FAd series becomes essentially indistinguishable from that of the BHMTHF homopolyesters. This underlines that the entire BHMTHF-based (co)polyester family can be accessed via protocols that are consistently in the “acceptable” EcoScale range and comparable in performance.
These values are in line with, and in several cases slightly better than, those reported recently for enzymatic bulk polycondensation of BHMF-based polyesters using the same immobilized CALB catalyst and very similar work-up procedures. This resulted in AE values around 77 to 82%, E-factors on the order of 98 to 161 and EcoScale scores between 62 and 73 for the synthesis of BHMF-based polyesters.29
A meaningful benchmark for the green advance of the present work is provided by the state-of-the-art preparation of BHMTHF-based polyesters via conventional melt polycondensation. Jin and co-workers demonstrated the synthesis of various BHMTHF-based polyesters at the kilogram scale using a titanium catalyst up to 240 °C under high vacuum and with multiple steps for pre-polymer formation and removal of low-molecular-weight by-products.8 Although green metrics were not reported, the use of metal catalysts, high reaction temperatures, extended reaction times, and additional purification steps inherently increases energy consumption and the mass of auxiliaries, and would be expected to result in significantly higher E-factors and lower EcoScale scores than those obtained for the mild enzymatic bulk polymerizations described here.45 In contrast, the present method operates at substantially lower temperatures, avoids any metal catalyst, and relies on a recyclable biocatalyst in a solvent-free reaction medium, which directly addresses several of the “Measuring Green Chemistry: Methods, Models, and Metrics” criteria highlighted for greener polymer syntheses.43
At the same time, the metrics analysis clearly identifies the main hotspots of the current laboratory procedure. Across all polymerizations, the E-factor is dominated by the organic solvents used for precipitation, washing, and enzyme removal, rather than by the reagents themselves. This is consistent with quantitative studies showing that solvent use and work-up typically contribute the majority of the mass intensity and environmental burden in polymer synthesis.29 In an industrial context, replacing chlorinated solvents with greener alternatives, minimizing the number of work-up steps and implementing solvent recycling or direct melt processing (for example, via extrusion of the crude polymer) would therefore be expected to reduce the effective E-factor by approximately an order of magnitude and move the EcoScale scores towards the “excellent” regime.
This work aligns with key Green Chemistry principles: the use of renewable feedstocks (7) such as sugar-derived monomers BHMTHF, BHMF, and FDCA; enzymatic catalysis (9) replacing metal catalysts; and energy-efficient, solvent-free bulk polymerization (6) under mild conditions. The resulting biodegradable materials (10) support circularity and environmental compatibility. Minor deviations occur in atom economy (2) due to methanol/ethanol release from ester monomers and in auxiliaries (5) through limited use of chloroform and diethyl ether for purification. Overall, the study exemplifies a renewable, catalytic, and degradable polymer synthesis consistent with the Green Chemistry framework.
Conclusion
BHMTHF, a rigid biobased building block containing a ring structure, was used in a cis![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) trans ratio of 90.6
trans ratio of 90.6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9.4 for the enzymatic polymerization of various (co)polyesters. In the first series, BHMTHF was reacted with various aliphatic comonomers to obtain amorphous materials with a relatively high molecular weight, having a
9.4 for the enzymatic polymerization of various (co)polyesters. In the first series, BHMTHF was reacted with various aliphatic comonomers to obtain amorphous materials with a relatively high molecular weight, having a  ranging from 5500 to 11
ranging from 5500 to 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 g mol−1. The second series consisted of copolyesters derived from BHMTHF, DMAd, and BHMF, wherein the BHMF-BHMTHF ratio was varied. Similarly, the third series consisted of copolyesters derived from BHMTHF, DMAd, and FDCA units, wherein the DMAd-FDCA ratio was altered. The BHMF-based copolyesters reached relatively high
200 g mol−1. The second series consisted of copolyesters derived from BHMTHF, DMAd, and BHMF, wherein the BHMF-BHMTHF ratio was varied. Similarly, the third series consisted of copolyesters derived from BHMTHF, DMAd, and FDCA units, wherein the DMAd-FDCA ratio was altered. The BHMF-based copolyesters reached relatively high  values (6600–11
values (6600–11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1), while the enzyme iCALB had difficulties catalyzing the high FDCA content copolyesters, leading to lower
000 g mol−1), while the enzyme iCALB had difficulties catalyzing the high FDCA content copolyesters, leading to lower  ranging from 1000–9100 g mol−1.
ranging from 1000–9100 g mol−1.
DSC analysis demonstrated that a higher aliphatic spacer length in the first series resulted in a quadratic decrease in Tg values from −8 to −49 °C. Furthermore, a higher BHMF or FDCA content increased the Tg values of the copolyesters, this phenomenon was more pronounced in the FDCA-BHMTHF-based copolyesters.
Rheological analysis demonstrated that polyesters from the first series behave like Newtonian fluids, and the low shear viscosity decreased with an increase in aliphatic spacer length. The low shear viscosity increased with a higher furan content, which indicated that both BHMF and FDCA increased the strength of these materials.
Finally, biodegradation tests revealed that all BHMTHF-based (co)polyesters are prone to biodegradation in activated sludge. The aliphatic spacing length, BHMF, or FDCA content had no significant influence on the biodegradation performances due to their amorphous morphology and chemical similarities.
The greenness of the enzymatic synthesis of all BHMTHF-based (co)polyesters was evaluated based on AE, RME, OE, E-factor, and EcoScale. The polymer composition had no significant effect on the green characteristic metrics of these copolyesters, which were all assessed as “acceptable” according to the EcoScale criteria.
This study demonstrates the compatibility of BHMTHF to be used for various (co)polyesters featuring unique and tunable thermal and rheological properties. This paves the way for BHMTHF to be further explored and utilized as a renewable, rigid biobased monomer in a sustainable plastic economy.
Author contributions
C. Post: investigation, conceptualization, methodology, formal analysis, data curation, visualization, writing – original draft. G. Karchilakis: investigation, formal analysis, data curation, writing – review & editing. D. Maniar: supervision, writing – review & editing. V. S. D. Voet, R. Folkersma: supervision, funding acquisition, writing – review & editing. K. Loos: supervision, resources, project administration, funding acquisition, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary Information: 1H-NMR data and spectra, 2D-NMR spectra, TGA curves, DSC curves, and frequency sweeps of the BHMTHF-based (co)polyesters. Supplementary information is available. See DOI: https://doi.org/10.1039/d5gc06368j.Acknowledgements
This work was supported by Greenwise Campus, Regio Deals, the Province of Drenthe, and the Municipality of Emmen. Furthermore, the authors acknowledge the financial support from the European Commission's Horizon ACTPAC program.In addition, we want to acknowledge Hero Jan Heeres and Léon Rohrbach, from the Engineering and Technology Institute Groningen (ENTEG), University of Groningen, The Netherlands, for allowing us to use their GPC facilities.
References
- J. A. Galbis, M. D. G. García-Martín, M. V. De Paz and E. Galbis, Synthetic Polymers from Sugar-Based Monomers, Chem. Rev., 2016, 116(3), 1600–1636, DOI:10.1021/acs.chemrev.5b00242.
- A. Gandini and T. M. Lacerda, Monomers and macromolecular materials from renewable resources: State of the art and perspectives, Molecules, 2022, 27(1), 1–26, DOI:10.3390/molecules27010159.
- E. de Jong, H. A. Visser, A. S. Dias, C. Harvey and G. J. M. Gruter, The Road to Bring FDCA and PEF to the Market, Polymers, 2022, 14(5), 1–32, DOI:10.3390/polym14050943.
- A. A. Turkin, E. V. Makshina and B. F. Sels, Catalytic Hydroconversion of 5–HMF to Value–Added Chemicals: Insights into the Role of Catalyst Properties and Feedstock Purity, ChemSusChem, 2022, 15(13), e202200412, DOI:10.1002/cssc.202200412.
- C. Chien Truong, D. Kumar Mishra, S. Hyeok Ko, Y. Jin Kim and Y. W. Suh, Sustainable Catalytic Transformation of Biomass-Derived 5-Hydroxymethylfurfural to 2,5-Bis(hydroxymethyl)tetrahydrofuran, ChemSusChem, 2022, 15(13), e202200178, DOI:10.1002/cssc.202200178.
- C. Jin, F. Ma, C. Li, Z. Tu, H. Wang and Z. Wei, Kilogram-scale preparation of sustainable PETG modified with a biobased cyclic diol derived from 5-hydroxymethylfurfural: From synthesis to properties, Eur. Polym. J., 2021, 161, 110832, DOI:10.1016/j.eurpolymj.2021.110832.
- C. Jin, W. Tian, Z. Tu and Z. Wei, Kilogram-Scale Production of Sustainable PCF Copolyesters Based on Novel Cyclic Diol THFDM Derived from 5-Hydroxymethylfurfural: Trade-Off between the THFDM Structure and Various Properties of Copolyesters, ACS Sustainable Chem. Eng., 2021, 9(39), 13287–13302, DOI:10.1021/acssuschemeng.1c04574.
- C. Jin, L. Liu, Z. Tu, B. Wang, P. Wang and Z. Wei, Melt polycondensation of 2,5-tetrahydrofurandimethanol with various dicarboxylic acids towards a variety of biobased polyesters, Polym. Chem., 2022, 13, 5718–5729, 10.1039/d2py00975g.
- C. Jin, B. Wang, L. Liu, Z. Tu and Z. Wei, Biodegradable Poly(butylene succinate) Copolyesters Modified by Bioresoured 2,5-Tetrahydrofurandimethanol, ACS Sustainable Chem. Eng., 2022, 10(34), 11203–11214, DOI:10.1021/acssuschemeng.2c02842.
- Y. Liu, B. Zheng, Z. Chen, X. Pu and S. Zhang, 2,5-Tetrahydrofurandimethanol as a sustainable comonomer for modification of polyethylene terephthalate: Liter-level pilot test and non-isothermal crystallization kinetics, J. Thermoplast. Compos. Mater., 2024, 37(4), 1364–1384, DOI:10.1177/08927057231191466.
- Y. L. Chen, et al., New bio-based polyester with excellent spinning performance: poly(tetrahydrofuran dimethanol-co-ethylene terephthalate), RSC Adv., 2022, 12(45), 29516–29524, 10.1039/d2ra04484f.
- D. Wang, et al., Bio-Based Copolyester PEIFT: Enhanced Hydrophilicity, Rigidity, and Applications in Nanofibers, Macromol. Rapid Commun., 2024, 45(12), 1–7, DOI:10.1002/marc.202300715.
- Y. Hu, H. Zhang, X. Pu and L. Yang, New Bio-Based Copolyesters Derived from Novel Cyclic Diol 2,5-Tetrahydrofurandimethanol, 2,5-Furardicarboxylic acid and 1,3-Propanediol: Synthesis, Thermal Behavior and Rheological Properties, Polym. Sci., Ser. B, 2023, 65(4), 429–437, DOI:10.1134/S1560090423701166.
- X. Cai, Y. Yang, H. Yu, D. Jiao, L. Xu and D. Zhang, A Sustainable Poly(butylene furandicarboxylate) (PBF)-Based Copolyester Containing a Cyclic Ether Structure: Integrated with High Thermal, Mechanical, and Hydrolytic Degradable Properties, ACS Sustainable Chem. Eng., 2025, 13(1), 658–668, DOI:10.1021/acssuschemeng.4c09157.
- C. Post, D. Maniar, R. Folkersma, V. S. D. Voet and K. Loos, Panoramic view of biobased BHMTHF-based polymers, Polym. Chem., 2025, 16(32), 3587–3596, 10.1039/d5py00544b.
- J. Kim, et al., A heterocyclic polyurethane with enhanced self-healing efficiency and outstanding recovery of mechanical properties, Polymers, 2020, 12(4), 968, DOI:10.3390/POLYM12040968.
- T. Chen, H. Wu, W. Zhang, J. Li, F. Liu and E. H. Han, A sustainable body-temperature self-healing polyurethane nanocomposite coating with excellent strength and toughness through optimal hydrogen-bonding interactions, Prog. Org. Coat., 2023, 185, 107876, DOI:10.1016/j.porgcoat.2023.107876.
- T. Chen, W. Zhang, Y. Li, F. Liu and E. H. Han, A self-healing sustainable halloysite nanocomposite polyurethane coating with ultrastrong mechanical properties based on reversible intermolecular interactions, Appl. Surf. Sci., 2024, 667, 160399, DOI:10.1016/j.apsusc.2024.160399.
- A. Douka, S. Vouyiouka, L. M. Papaspyridi and C. D. Papaspyrides, A review on enzymatic polymerization to produce polycondensation polymers: The case of aliphatic polyesters, polyamides and polyesteramides, Prog. Polym. Sci., 2018, 79, 1–25, DOI:10.1016/j.progpolymsci.2017.10.001.
- F. Silvianti, D. Maniar, L. Boetje and K. Loos, Green Pathways for the Enzymatic Synthesis of Furan-Based Polyesters and Polyamides, in Sustainability & Green Polymer Chemistry Volume 2: Biocatalysis and Biobased Polymers, American Chemical Society, 2020, vol. 2, pp. 3–29. DOI:10.1021/bk-2020-1373.ch001.
- D. Maniar, Y. Jiang, A. J. J. Woortman, J. van Dijken and K. Loos, Furan-Based Copolyesters from Renewable Resources: Enzymatic Synthesis and Properties, ChemSusChem, 2019, 12(5), 990–999, DOI:10.1002/cssc.201802867.
- C. I. Gkountela and S. N. Vouyiouka, Enzymatic Polymerization as a Green Approach to Synthesizing Bio-Based Polyesters, Macromol, 2022, 2(1), 30–57, DOI:10.3390/macromol2010003.
- A. Pellis, S. Weinberger, M. Gigli, G. M. Guebitz and T. J. Farmer, Enzymatic synthesis of biobased polyesters utilizing aromatic diols as the rigid component, Eur. Polym. J., 2020, 130, 109680, DOI:10.1016/j.eurpolymj.2020.109680.
- Y. Jiang, A. J. J. Woortman, G. O. R. Alberda Van Ekenstein, D. M. Petrović and K. Loos, Enzymatic synthesis of biobased polyesters using 2,5-bis(hydroxymethyl) furan as the building block, Biomacromolecules, 2014, 15(7), 2482–2493, DOI:10.1021/bm500340w.
- T. P. Haider, C. Völker, J. Kramm, K. Landfester and F. R. Wurm, Plastics of the Future? The Impact of Biodegradable Polymers on the Environment and on Society, Angew. Chem., Int. Ed., 2019, 58(1), 50–62, DOI:10.1002/anie.201805766.
- B. C. A. de Jong, et al., Sustainable Fuels from CO2-Rich Synthesis Gas via Fischer-Tropsch Technology, ACS Catal., 2025, 15(12), 10946–10956, DOI:10.1021/acscatal.5c03024.
- J. K. Ogunjobi, et al., Synthesis of Biobased Diethyl Terephthalate via Diels-Alder Addition of Ethylene to 2,5-Furandicarboxylic Acid Diethyl Ester: An Alternative Route to 100% Biobased Poly(ethylene terephthalate), ACS Sustainable Chem. Eng., 2019, 7(9), 8183–8194, DOI:10.1021/acssuschemeng.8b06196.
- OECD Guideline for testing of chemicals, OECD 301 – Ready Biodegradability, OECD Guidel. Test. Chem., 1992.
- C. Post, et al., Enzymatic bulk synthesis, characterization, rheology, and biodegradability of biobased 2,5-bis(hydroxymethyl)furan polyesters, Green Chem., 2024, 26(15), 8744–8757, 10.1039/d4gc01512f.
- C. R. McElroy, A. Constantinou, L. C. Jones, L. Summerton and J. H. Clark, Towards a holistic approach to metrics for the 21st century pharmaceutical industry, Green Chem., 2015, 17(5), 3111–3121, 10.1039/c5gc00340g.
- K. Van Aken, L. Strekowski and L. Patiny, EcoScale, a semi-quantitative tool to select an organic preparation based on economical and ecological parameters, Beilstein J. Org. Chem., 2006, 2(1), 3, DOI:10.1186/1860-5397-2-3.
- R. A. Sheldon, Metrics of Green Chemistry and Sustainability: Past, Present, and Future, ACS Sustainable Chem. Eng., 2018, 6(1), 32–48, DOI:10.1021/acssuschemeng.7b03505.
- R. A. Sheldon, The E factor at 30: a passion for pollution prevention, Green Chem., 2023, 25(5), 1704–1728, 10.1039/d2gc04747k.
- E. H. Choi, J. Lee, S. U. Son and C. Song, Biomass-derived furanic polycarbonates: Mild synthesis and control of the glass transition temperature, J. Polym. Sci., Part A: Polym. Chem., 2019, 57(17), 1796–1800, DOI:10.1002/pola.29448.
- F. Aricò, Synthetic approaches to 2,5-bis(hydroxymethyl)furan (BHMF): a stable bio-based diol, Pure Appl. Chem., 2021, 93(5), 551–560, DOI:10.1515/pac-2021-0117.
- C. Ortiz, et al., Novozym 435: The ‘perfect’ lipase immobilized biocatalyst?, Catal. Sci. Technol., 2019, 9(10), 2380–2420, 10.1039/c9cy00415g.
- E. G. Dixon, L. Ciano, A. Pordea, K. Alvey and V. Taresco, Harnessing enzymes for greener polymerisations: advances in chain and step growth processes, Polym. Chem., 2025, 16(26), 2997–3029, 10.1039/d5py00223k.
- V. Hevilla, A. Sonseca, C. Echeverría, A. Muñoz-Bonilla and M. Fernández-García, Enzymatic Synthesis of Polyesters and Their Bioapplications: Recent Advances and Perspectives, Macromol. Biosci., 2021, 21(10), 1–28, DOI:10.1002/mabi.202100156.
- G. A. Vázquez-Rodríguez, R. I. Beltrán-Hernández, C. Coronel-Olivares and J. L. Rols, Standardization of activated sludge for biodegradation tests, Anal. Bioanal. Chem., 2011, 401(4), 1127–1137, DOI:10.1007/s00216-011-5212-z.
- A. Bher, P. C. Mayekar, R. A. Auras and C. E. Schvezov, Biodegradation of Biodegradable Polymers in Mesophilic Aerobic Environments, Int. J. Mol. Sci., 2022, 23(20), 12165, DOI:10.3390/ijms232012165.
- A. A. Shah, S. Kato, N. Shintani, N. R. Kamini and T. Nakajima-Kambe, Microbial degradation of aliphatic and aliphatic-aromatic co-polyesters, Appl. Microbiol. Biotechnol., 2014, 98(8), 3437–3447, DOI:10.1007/s00253-014-5558-1.
- Y. Chen, L. Tan, L. Chen, Y. Yang and X. Wang, Study on biodegradable aromatic/aliphatic copolyesters, Braz. J. Chem. Eng., 2008, 25(2), 321–335, DOI:10.1590/S0104-66322008000200011.
- A. Bardow, J. Pérez-Ramírez, S. Sala and L. Vaccaro, Measuring green chemistry: methods, models, and metrics, Green Chem., 2024, 26(22), 11016–11018, 10.1039/d4gc90118e.
- Q. Wang, et al., Synthesis of 2,5-furandicarboxylic acid-based biodegradable copolyesters with excellent gas barrier properties composed of various aliphatic diols, Eur. Polym. J., 2022, 181, 111677, DOI:10.1016/j.eurpolymj.2022.111677.
- S. Fadlallah and F. Allais, Sustainability Appraisal of Polymer Chemistry Using E-Factor: Opportunities, Limitations and Future Directions, ACS Symp. Ser., 2023, 1451, 3–30, DOI:10.1021/bk-2023-1451.ch001.
| This journal is © The Royal Society of Chemistry 2026 |