Open Access Article
Open Access ArticleTwo-step production of a furfural-derived biosolvent in a single reactor: an economic and life cycle assessment study
C. Yezeguelianb and
M. Pera-Titus *a
*a
aCardiff Catalysis Institute, School of Chemistry, Cardiff University, Cardiff CF10 3AT, UK. E-mail: peratitusm@cardiff.ac.uk
bSyensqo, Research & Innovation Centre of Lyon, 85 Avenue des Frères Perret, 69192 Saint Fons, France
First published on 6th March 2026
Abstract
In this study, we investigated the credentials of furfural as a building block for producing 5-methyl-1-(tetrahydrofuran-2-yl)hexan-3-one (THF-ALD-1), a prospective biosolvent. The process consists of two steps: (1) aldol condensation–crotonisation of furfural with methyl isobutyl ketone (MIBK) to generate (E)-1-(furan-2-yl)-5-methylhex-1-en-3-one (ALD-1) and (2) selective hydrogenation of ALD-1 to THF-ALD-1 in MIBK. We conducted a comparative techno-economic analysis (TEA) and life cycle assessment (LCA) to evaluate the potential benefits of process intensification in a single reactor using a bifunctional 5%Pd/Al2O3 catalyst compared to a two-reactor process employing two catalysts—Al2O3 for the aldol condensation–crotonisation step and 5%Pd/Al2O3 for ALD-1 hydrogenation. A sensitivity analysis was performed to identify the key contributing factors and propose levers to reduce the cost and environmental footprint of THF-ALD-1 production. Our study demonstrates that a single-reactor process can offer an 11% and 7.5% reduction in the full manufacturing cost and minimum selling point, respectively, alongside reductions in environmental impact in most categories, with statistically significant improvements observed for acidification potential (21%), photochemical oxidant creation potential (22%), freshwater ecotoxicity potential (19%), and terrestrial ecotoxicity potential (46%). Directional trends toward lower impacts were also observed in the remaining categories. The furfural and MIBK market price fluctuations, overall yield, MIBK/furfural molar ratio, and the number of Pd/Al2O3 catalyst regeneration cycles are critical levers that influence fresh Pd demand, waste generation, and heat requirements for distillation. Optimisation of these parameters could yield potential cost benefits up to 20% for each process and mitigate by 32–36% the global warming potential, non-renewable energy use, and abiotic and ozone depletion potential, with full statistical significance according to Monte Carlo analysis.
Green foundation1. This work provides a combined TEA and LCA to evaluate the benefits of process intensification in a single reactor compared to a two-step reactor process for manufacturing 5-methyl-1-(tetrahydrofuran-2-yl)hexan-3-one (THF-ALD-1), a potential biosolvent from the aldol condensation–crotonization of furfural with MIBK, followed by hydrogenation.2. A single-reactor process can offer an 11% cost reduction and lower environmental impact in terrestrial ecotoxicity potential (46%), renewable energy (25%), photochemical oxidant creation potential (22%), and acidification potential (21%). 3. A sensitivity analysis identified the key levers to reduce the cost and environmental footprint of THF-ALD-1 production. Process optimisation could yield potential cost benefits up to 20% and mitigate by 32–36% the global warming potential, non-renewable energy, and abiotic/ozone depletion potential. |
Introduction
The depletion of fossil resources and the environmental shortcomings associated with their use are driving the development of innovative, more sustainable technologies capable of reducing oil dependency. Biomass represents a vast reservoir of renewable carbon that can provide a large variety of building blocks for the manufacture of fuels and chemicals.1 To this end, it is necessary to design new chemical processes starting from renewable feedstocks, offering economically feasible and environmentally friendly routes to target products across a broad variety of markets and scenarios.2 This necessitates a critical and rigorous analysis of the key parameters and variables affecting the processes and their boundaries.3Furanic compounds, produced from the hydrolysis of carbohydrates derived from lignocellulose, have attracted great interest for manufacturing commodities, specialty chemicals, and fuels.4 The aldol condensation reaction of furfural (FF) with a ketone, followed by a sequential reaction, has been proposed as a strategy to access biofuels and biosolvents with controllable carbon molecularity. The overall reaction involves two steps, which can be carried out either in two catalytic reactors or in a single reactor combining two catalysts or a bifunctional catalyst. Compared to the two-reactor process, a single reactor offers greater process intensification and avoids the need to separate the aldol intermediate. For instance, biofuels, biosolvents and monomer precursors have been prepared in a single reactor by combining an aldol condensation step of FF with acetone, methyl isobutyl ketone (MIBK) or cyclopentanone followed by hydrodeoxygenation/hydrogenation catalysed by Pd or Pt supported over acid/base supports,5 Pd supported over hierarchical zeolites or propylamine-grafted mesoporous silicas6 and Pt encapsulated in acid zeolites.7 A key challenge in these developments is the compatibility of the different active centres, which can exhibit antagonistic behaviour under varying conditions across the reaction steps.
Herein we explore the potential of FF as a building block for the production of 5-methyl-1-(tetrahydrofuran-2-yl)hexan-3-one (THF-ALD-1), a prospective biosolvent for applications requiring a high boiling point (>200 °C), medium polarity and strong solvency for polymers and resins, low vapour pressure and VOC compliance, and biodegradability and renewable carbon content. Target sectors include coatings and paints, polymer processing and resin formulation, agrochemical co-solvents, and industrial cleansing and electronic-grade solvents. The process for THF-ALD-1 production comprises two steps (Scheme 1A): (1) aldol condensation–crotonisation of FF with MIBK to generate (E)-1-(furan-2-yl)-5-methylhex-1-en-3-one (ALD-1) using MIBK as excess solvent and (2) selective hydrogenation of ALD-1 to THF-ALD-1 in MIBK. The single-reactor and two-reactor processes generate different by-products with comparable overall THF-ALD-1 yields (86% and 84%, respectively) (Scheme 1B; see ref. 8 for more details).
We conducted a comparative technoeconomic analysis (TEA) and life cycle assessment (LCA) study of two alternative process configurations to produce THF-ALD-1—single-reactor and two-reactor routes—with the objective of quantifying the potential benefits and trade-offs associated with process intensification using a bifunctional 5%Pd/Al2O3 catalyst. This study aims to identify the key contributors to the cost and environmental impact of THF-ALD-1 production and propose levers using a reasonable set of simplifying assumptions regarding system boundaries and allocation methods for attributing environmental impacts.9
Methodology
Metrics
Our combined TEA and LCA followed five steps in accordance with the ISO 14040:2006 and ISO 14044:2006 standards:10 (i) scope and functional unit definition, (ii) life-cycle inventory analysis, (iii) life cycle impact assessment (LCIA), (iv) cost assessment, and (v) interpretation. The inventory analysis and impact assessment were performed using SimaPro 9.1 software and the Ecoinvent database versions 3.4 and 3.6, applying the cut-off modelling approach, with version 3.4 specifically adopted for palladium and maize production inventories. Unit costs for raw materials were obtained from the references indicated in Table 1.| Variables | Base casea | Ref. | |
|---|---|---|---|
| a In LCA + economics, values correspond to single-reactor/two-reactor processes.b Excess H2 over stoichiometric requirement.c THF-ALD-1 purification yield of 98%. References: (a) Business Analyst, Q4 2018 (ref. 11); (b) Business Analyst, Q4 2018 (ref. 12); (c) price for green H2 (ref. 13); (d) from Zibo Anquan Chemical Co., Ltd (China) (ref. 14); (e) based on internal Solvay (Syensqo) data; (f) Johnson Matthey Pd prices, Q2 2018 (ref. 15); (g) data from ref. 8 (lab-scale optimised reaction conditions); (h) process assumption; (i) “yes”: on site valorisation of organic wastes by combustion in a boiler for steam generation (steam credit); “no”: wastes burned outside. | |||
| Economics | FF, € per ton | 1300 | (a) |
| MIBK, € per ton | 1250 | (b) | |
| H2, € per ton | 2000 | (c) | |
| Ethanol 95%, € per ton | 1000 | (d) | |
| Al2O3, € per ton | 9000 | (e) | |
| Pd/Al2O3, € per ton (fresh) | 744![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
(f) | |
| LCA + economics | FF conversion, % | 93/92 | (g) |
| FF → ALD-1 selectivity, % | 92.5/100 | (g) | |
| FF → ALD-2 selectivity, % | 7.5/— | (g) | |
| ALD-1 conversion, % | 100/100 | (g) | |
| ALD-1 → THF-ALD-1 selectivity,c % | 100/92 | (g) | |
| Overall THF-ALD-1 yield, % | 86.0/84.0 | (g) | |
| Al2O3 loading, wt% | —/5 | (h) | |
| Pd/Al2O3 loading, wt% | 5/5 | (h) | |
| H2 consumption,b % | +10/+10 | (h) | |
| MIBK recycling rate, % | 98/98 | (h) | |
| Ethanol recycling rate, % | 95/95 | (h) | |
| Pd/Al2O3 loss after each batch, % | 0.05/0.05 | (h) | |
| Pd/Al2O3 regeneration/recycling, % | 90/90 | (h) | |
| Aqueous/organic waste | Yes | (i) | |
| Landfill | Yes | — | |
Our LCA methodology is not limited to the carbon footprint (climate change), which is often presented as the sole environmental indicator for evaluating the sustainability of biomass-derived chemicals. Instead, a broader panel of indicators was adopted to account for pollution transfer across different impact categories within the three main areas of protection: toxicity, ecosystem energy and quality, and resource depletion.
Scope and functional unit definition
Transportation of reagents (e.g., furfural, MIBK, and catalysts) was excluded from the system boundary, as preliminary sensitivity checks using representative European transport distances (100–500 km by truck) and typical carbon footprint of trucks (∼1 kgCO2 km−1) revealed a contribution of <1–2% to the global warming potential of the processes.
The consistent scaling framework and harmonised data sources enabled the following key objectives:
➢ Evaluate the FMC and SPM costs of each process;
➢ Identify major cost and environmental drivers through sensitivity analysis;
➢ Quantify the performance improvements associated with process intensification; and
➢ Provide a refined comparison of the environmental impact of both process routes.
Life cycle inventory (LCI)
Production scale assumptions. To extrapolate to the industrial scale, a production rate of 3300 t per year of THF-ALD-1 was assumed, corresponding to continuous reactor operation over 8000 h per year. This scale represents a mid-tonnage specialty chemical intermediate, above typical niche batch volumes but below commodity-scale production. At this scale, continuous reactors are a feasible configuration, whereas smaller production volumes would typically rely on batch or toll-manufacturing processes. These assumptions provide context for interpreting the LCA results, rather than serving as a definitive techno-economic assessment.
Market context and applications. THF-ALD-1 is prospectively used as a biosolvent, which is generally a niche market with limited publicly available production data. The present study does not include a detailed market analysis, as the main objective is to evaluate the environmental performance of alternative synthetic routes rather than assess commercial feasibility. The production scale assumption informs the interpretation of material and energy flows and supports a realistic industrial extrapolation of laboratory-scale results.
Inventory development and scaling. All inventory data were obtained from primary laboratory experiments,8 ensuring a consistent and directly measured dataset. No secondary data from literature sources were used for the core process inventory. The experimental results were collected under controlled laboratory conditions and reflect the actual material and energy inputs measured during operation. To extrapolate the findings to a larger (industrial) scale, a scaling framework based on process engineering principles was applied, maintaining key mass and energy balance relationships while adjusting for expected efficiencies and operational parameters.
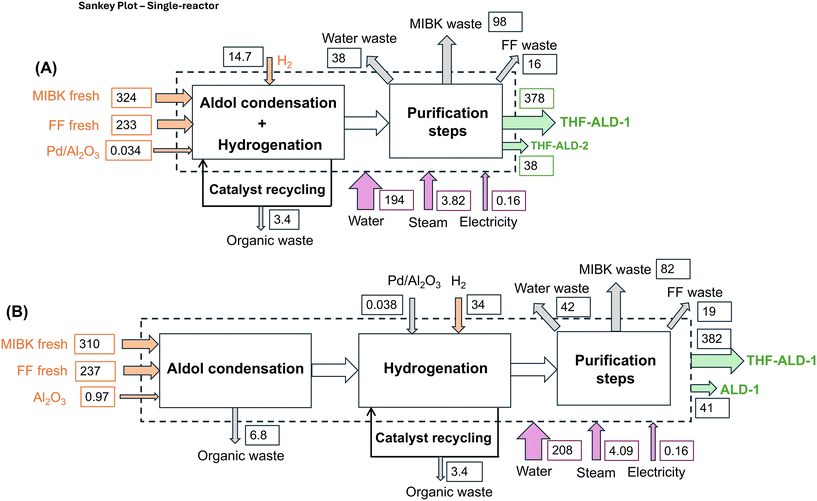
Table 1 lists the key variables influencing cost and environmental footprint for both processes. Table S1 lists the physical properties of all compounds used in the calculations. Mass and energy balances were developed for both processes to construct the inventory presented in Table S2, with the process assumptions in Table S3. Flowsheet diagrams are provided in Fig. S1 and S2. Sankey plots are provided in Scheme 2.
Key energy-related datasets were harmonized across all inventories to ensure comparability and avoid artificial variability. Specifically, steam demand was modelled using a relatively efficient European industrial natural-gas boiler (heat, district or industrial, natural gas {Europe without Switzerland} |heat production, natural gas, at boiler condensing modulating >100 kW| cut-off, U), representative of generic chemical-industry steam production, while electricity consumption was modelled using a European medium-voltage market mix (electricity, medium voltage {RER} |market| cut-off, U).
Feedstock- and catalyst-related datasets include (Table S4): maize (GLO) and palladium (GLO) (Ecoinvent v3.4); alumina HP Ziegler process (RER) (Ecoinvent v3.6, European proxy); MIBK (RER), electricity (RER), steam/heat (Europe without Switzerland), process water (RER) and H2 (GLO) (Ecoinvent v3.6).
These choices allow for reproducibility of the life cycle assessment while reflecting realistic environmental burdens associated with the supply of materials and energy.
LCI of Pd and PGM systems17. he LCI of Pd was updated using 2010–2019 market data (superseding the older 1993–2002 data in Ecoinvent 3.4, {GLO} |market for update 2019| cut-off, U). The share between primary (70%) and secondary (30%) Pd was revised, with primary Pd sourced from South Africa and Russia and secondary Pd from automotive recycling (20% Europe, 80% rest of the world) based on Johnson Matthey data.18 The production processes in South Africa and Russia were treated separately and implemented in two different LCIs. Allocations were carried out by revenue using prices of the period 2010–2019.18 South African and Russian production were modelled separately and integrated into a global LCI weighted by market share. For Europe (Germany), automotive recycling was divided into three steps: collection, beneficiation and refining. Emissions and energy use in the final step were approximated using primary production values. Other recycling streams (e.g., electronics, jewellery) were excluded.
The revised impact values for Pd were found to be 1.7 times higher than those in Ecoinvent 3.4 (e.g., 8252 kgCO2-eq. per kg vs. 4900 kgCO2-eq. per kg) (Table S5). These values align with recent studies on metal-supported catalysts.19
LCI of γ-Al2O3. The environmental profile of γ-Al2O3 was derived from the Ziegler process using aluminium alkoxides in Germany, with 20
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80 economic allocation between alumina and co-produced fatty alcohols (Ecoinvent 3.6, GLO).20 This reflects the production of a grade like Puralox Scca-5/170 (154 m2 g−1) used in laboratory-scale tests. The cost of γ-Al2O3 was estimated at €9000 per ton, based on internal Solvay data.
80 economic allocation between alumina and co-produced fatty alcohols (Ecoinvent 3.6, GLO).20 This reflects the production of a grade like Puralox Scca-5/170 (154 m2 g−1) used in laboratory-scale tests. The cost of γ-Al2O3 was estimated at €9000 per ton, based on internal Solvay data.
LCI of 5 wt%Pd/γ-Al2O3. Catalyst production was modelled using a simplified synthesis pathway for the Pd precursor Pd(NO3)2, excluding the acetic acid step described in ref. 21 and based on (eqn 1):
| Pd(0) + 4HNO3 → Pd(NO3)2 + 2NO2 + 2H2O | (1) |
Reaction conditions:
○ HNO3/Pd ratio: 0.073 w/w;
○ Temperature: 118 °C (reflux);
○ Time: 30 min; and
○ Yield: 100% (vs. 92% in ref. 22).
Alumina was assumed to be roughly shaped by extrusion instead of being kept in powder form for better reflecting common industrial practices. Dry impregnation was assumed, with a pore volume of 1 cm3 g−1.23 The full inventory is provided in Table S6, with the flowsheet diagram in Fig. S4. Environmental impacts were obtained for Ecoinvent 3.6 (GLO) are listed in Table S7, and a breakdown of component contributions is shown in Fig. S5. The main impact on Pd/Al2O3 manufacture is attributed to Pd(0) and scales linearly with Pd loading (Fig. S6).
Pd/Al2O3 catalyst regeneration. The Pd/Al2O3 catalyst underwent internal regeneration (see Fig. S1 and S2). Catalyst loss during each regeneration cycle was assumed to be externally recovered and recycled, with at least 90% Pd recovery efficiency.24 A recycling cost of €20
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 per ton of Pd was applied (electronic grade).25 The required amount of fresh Pd as a function of the number of regeneration cycles (C) is presented in Table S8 and was used in the sensitivity analysis.
000 per ton of Pd was applied (electronic grade).25 The required amount of fresh Pd as a function of the number of regeneration cycles (C) is presented in Table S8 and was used in the sensitivity analysis.
LCI of FF. The LCI of FF was based on a furfuryl alcohol plant in Henan province, China (capacity: 60 kton per year).27 The process achieves:
○ Overall FF yield: 45%;
○ Energy consumption: 200 kWh per ton of FF; and
○ Energy recovery: 19.8 tons from 20.41 tons of steam generated from residues.
Inventory details are provided in Table S9, based on the flowsheet in Fig. S7.
LCI of corncob. A maize inventory from the Agri-footprint database was adapted to Ecoinvent 3.4, with the following allocation of farming burdens: grains (43%), corncob (5%) and stove (52%). The price of corncob was set at USD70 per ton.
Life cycle impact assessment (LCIA) methods
1. Climate change (kg CO2-eq. per kg): assessed using global warming potential over a 100-year horizon [GWP 100a], corrected for biogenic CO2 uptake as a carbon sink (eqn 2):
 | (2) |
2. Energy indicators (primary energy demand) (MJ kg−1):
2.1. Non-Renewable Energy [NRE]: from nuclear materials [uranium], coal [coal], crude oil [oil], and natural gas [gas].
2.2. Renewable Energy [Renew]: from hydropower [hydroelectric], wind [wind], solar [solar], geothermal power [geothermal], H2 power [hydrogen], and biomass [biomass + forestry].
3. Toxicity indicators (kg 1,4-DB-eq. per kg):
3.1. Human Toxicity Potential [HTP]
3.2. Freshwater Aquatic Ecotoxicity Potential [FAETP]
3.3. Marine Aquatic Ecotoxicity Potential [MAETP]
3.4. Terrestrial Ecotoxicity Potential [TETP]
4. Water Use [Water] (m3)
5. Abiotic Depletion Potential [ADP] [kg Sb-eq. kg−1]
6. Quantitative land use [Land Use] (m2 a kg−1)
7. Acidification potential [AP] (kg SO2-eq. per kg)
8. Eutrophication Potential [EP] (kg PO4-eq. per kg)
9. Ozone Layer Depletion Potential [OLDP] over a time horizon of 100 years (kg CFC-11-eq. per kg)
10. Photochemical Oxidant Creation Potential [POCP] (kg C2H4-eq. per kg).
1. H2: consumption during ALD-1 hydrogenation and Pd/Al2O3 reduction;
2. Waste treatment: incineration of organic waste (biogenic or fossil based);
3. Heat: energy used for reactor and distillation columns;
4. Electricity: electrical power use;
5. Chemicals: FF and MIBK production;
6. Ethanol: used in catalyst regeneration;
7. Catalyst: manufacture of Al2O3 and Pd/Al2O3, and recycling of Pd/Al2O3; and
8. Process: total process-related contributions.
For each impact category j, the total impact was computed as follows (eqn 3):
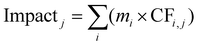 | (3) |
1. Pd/Al2O3 catalyst regeneration cycles (C): best case assumed three regenerations per year (C = 3);
2. Overall THF-ALD-1 yield: best case scenario at 100% yield; and
3. MIBK usage: best case reduction of the MIBK/FF molar ratio from 18.2 to 10.2 at 100% overall yield.
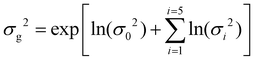 | (4) |
A statistical test was carried out for each impact factor to assess any statistically significant difference between both processes. For each iteration, all input parameters were randomly sampled from their respective distributions taking the same MC sampling sets, and the impacts were recalculated. To obtain representative results, 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 MC runs were executed in each simulation. The test was performed by counting the number of MC runs in which the single-reactor process showed higher/lower impact than the 2-step process for each impact category. In our calculations, we assumed that if at least 70% of the runs were favourable for one path, then the difference might be significant.
000 MC runs were executed in each simulation. The test was performed by counting the number of MC runs in which the single-reactor process showed higher/lower impact than the 2-step process for each impact category. In our calculations, we assumed that if at least 70% of the runs were favourable for one path, then the difference might be significant.
For each impact category, the 95% confidence interval (CI) was determined empirically from the MC output distribution, using the 2.5th and 97.5th percentiles as the lower and upper limits, respectively. This non-parametric method was chosen because LCA results typically exhibit non-normal distributions. Therefore, the reported intervals represent the range within which the true mean impact is expected to lie with 95% confidence.
 | (5) |
○ Fixed costs include labour and maintenance, estimated using an internal Solvay (Syensqo) methodology;
○ Depreciation covers capital expenditure amortised over the service life of the facility; and
○ The facility was assumed to be a new production line installed at an existing European industrial site (add-in-facility).
Capital cost estimation (CAPEX). CAPEX costs were estimated using the Taylor ‘process step scoring’ method.31 This method assigns a complexity score to each step based on factors such as dilution, reaction time, pressure, temperature, equipment materials, and safety. The scores are used to derive the cost index, which is then related to capacity and capital cost via empirical correlations.
Variable cost estimation. Variable cost per ton of product was calculated as (eqn 6):
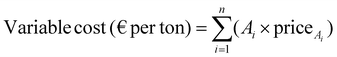 | (6) |
Table 1 lists the key input prices. All cost estimations were based on 2018 price values. This assumption maintains internal consistency for comparative purposes and avoids the effect of price volatility. The cost of Pd/Al2O3 was based on the market Q2-2018 Pd price (€24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 430 per kg Pd). The cost of γ-Al2O3 was set at €9000 per ton, using internal Solvay (Syensqo) data. For both processes, four initial catalyst charges were assumed: one for the reaction, one for internal regeneration, one for external recycling, and one spare for loading. Pd/Al2O3 consumption and Pd recovery were included in the cost calculations.
430 per kg Pd). The cost of γ-Al2O3 was set at €9000 per ton, using internal Solvay (Syensqo) data. For both processes, four initial catalyst charges were assumed: one for the reaction, one for internal regeneration, one for external recycling, and one spare for loading. Pd/Al2O3 consumption and Pd recovery were included in the cost calculations.
Annual net cash flows (CFt) were calculated as (eqn 7)
| CFt = Rt − Copex,t | (7) |
 | (8) |
The minimum selling price (MSP) was defined as the product price required to achieve a target IRR and was calculated iteratively using (eqn 9):
 | (9) |
(1) Operational vulnerability, representing the environmental footprint of production, and
(2) Market alignment, representing the extent to which the product supports or conflicts with sustainability trends.32
The operational vulnerability was determined from the cradle-to-gate LCA assessment. Environmental impacts were monetised using ‘shadow costs’, theoretical prices reflecting the social cost of environmental damage.33 These values were derived from avoidance or repair cost approaches.34 For instance, Solvay applies a shadow cost of €75 per ton CO2 for climate impacts. The aggregated monetised impact yields an environmental cost intensity (€ per kg of THF-ALD-1), which is compared to the product value to determine its vulnerability.35
The market alignment assessment complements the above analysis by evaluating regulatory trends, customer demand, and potential sustainability benefits or risks.36 Combining both axes produces a heat map that classifies products as solutions, neutrals, or challengers, supporting portfolio steering and innovation priorities.
Results and discussion
Production costs
Table 2 lists the FMC of THF-ALD-1 for the single-reactor and two-reactor processes, based on the prices and inventories in Table S2. The FMC is 11% lower in the single-reactor process (€4015 vs. €4461 per ton). The costs are distributed across the three main categories as follows (single vs. two-reactor):○ Variable costs: €2591 vs. €2738 per ton
○ Fixed costs: €862 vs. €1050 per ton
○ Depreciation: €561 vs. €650 per ton.
| Single reactor | Two reactors | Difference (%) | |
|---|---|---|---|
| a Production = 3300 tons per year of THF-ALD-1, operation = 8000 h. | |||
| Variable cost | 2591 (64%) | 2727 (61%) | −5.2 |
| Fixed cost | 862 (22%) | 1050 (24%) | −22 |
| Depreciation | 561 (14%) | 650 (15%) | −16 |
| FMC cost | 4015 | 4428 | −10 |
| MSP | 5225 | 5609 | −7.5 |
The capital expenditure (CAPEX) is 10% higher in the two-reactor process (€19.0 million) than in the single-reactor process (€16.7 million) due to differences in equipment and catalyst usage. The major contributors to CAPEX are:
○ ISBL (main process equipment): €10.9M (single) vs. €12.8M (two-reactor);
○ OSBL (supporting equipment): €5.0M vs. €5.5M; and
○ Pd catalyst charges (4 loads): €0.83M vs. €0.72M.
For reactor sizing, the single-reactor process assumes a total residence time of 6 h (3 h for each reaction), while the two-reactor process assumes a 3 h contact time for each reactor.
The reactor(s) is(are) the primary contributor(s) to fixed costs. In the two-reactor process, the aldol condensation and hydrogenation reactors account for 44% of fixed costs, respectively, while the intensified reactor in the single-reactor process alone accounts for 45% of fixed costs. This apparent parity, despite process intensification, is explained by the higher dilution of FF needed in the single reactor to suppress side-product formation and the higher-pressure rating required for hydrogenation, which increases material and construction costs.
○ Slightly lower overall yield (86% vs. 84%), increasing FF and MIBK consumption;
○ Use of two catalysts, rising catalyst costs from €87 per ton to €117 per ton, broken down as: €23 per ton for γ-Al2O3 and €7 per ton for increased Pd/Al2O3 usage due to higher catalyst mass loading (34% vs. 20%); and
○ MIBK usage is marginally reduced in the two-reactor process due to the higher selectivity of γ-Al2O3 for ALD-1 formation, while the single-reactor generates 7.5% ALD-2 as a side product.
○ Fluctuation of reactant costs: the market prices of FF and MIBK showed high fluctuations in the period 2018–2025 due to changes in the economic cycles and geopolitical situation (Fig. S9). This translates into a fluctuation of the THF-ALD-1 cost between −5% and +29% for the single-reactor process and −9.5% and +28% for the two-step process.
○ THF-ALD-1 yield: increasing the yield to 100% results in a 17% reduction in variable costs and a 12% reduction in the FMC, covering essentially variable costs, with a limited effect on fixed costs and depreciation (Fig. S10).
○ MIBK/FF molar ratio: at 100% yield, every 2-point reduction in the MIBK/FF molar ratio leads to 1% and 2% decreases in the variable and FMC, respectively (Fig. S10).
When these factors are optimised simultaneously for a 100% overall yield, an MIBK/FF molar ratio of 10.2, and the lowest market prices for reagents, both processes achieve about a 20% cost reduction down to €2140 per ton and €2207 per ton for the single-reactor and two-reactor processes, respectively, with best-case FMC values of €3563 per ton and €3907 per ton. Overall, the single-reactor process offers notable advantages: a 10% reduction in FMC, 10–15% lower capital expenditure (CAPEX), and a 4.6% reduction in environmental costs.
Besides, the FMC is affected by the number of regeneration cycles (C) of the Pd/Al2O3 catalyst. Assuming 90% Pd recovery during catalyst unloading and treatment, the FMC increases sharply at lower catalyst lifetimes. Minimising Pd losses and maximising catalyst regeneration frequency are crucial for cost reduction. A maximum of three regeneration cycles per year (C = 3) is recommended to control costs effectively (Fig. S11).
Contribution to environmental impacts
○ A higher contribution from energy (utilities), mainly due to the increased steam consumption required for distillation—75% of the total steam and cooling water are used for MIBK distillation; and
○ A lower contribution from catalysts, as it is assumed that 90% of palladium (Pd) lost in each cycle (0.05% of total Pd used) is regenerated with no associated environmental impact.
The total SPM costs are comparable between the two processes: €2745 per ton for the single-reactor process and €2792 per ton for the two-reactor process. The primary contributors to the environmental costs are FF and MIBK, consistent with their contributions to manufacturing costs. Accordingly, the same levers—feedstock price, yield optimisation, and reduction of the MIBK/FF ratio—are relevant for reducing both the manufacturing and environmental footprints.
 | ||
| Fig. 3 Relative impacts between the single-reactor and two-reactor processes (base case). The different impact categories for each reference (from 1 to 10) are listed in Table 3. | ||
| No. | Impact category | Unit | Single-reactor | Two-reactor |
|---|---|---|---|---|
| a Calculations assuming C = 3 Pd/Al2O3 catalyst regeneration cycles per year.b See Table S16 for a breakdown of NRE (nuclear materials, coal, crude oil, natural gas). | ||||
| 1 | GWP 100a | kg CO2-eq. per kg | 6.482 | 6.560 |
| Trapped CO2 [Biogenic CO2] | kg CO2-eq. per kg | −1.19 | −1.19 | |
| 2.1 | Non-renewable energy [NRE] | MJ primary per kg | 118 | 120 |
| 2.2 | Renewable energy [Renew]b | MJ kg−1 | 3.75 | 5.03 |
| 3.1 | Human toxicity potential [HTP] | kg 1,4-DB-eq. per kg | 2.05 | 2.21 |
| 3.2 | Freshwater ecotoxicity potential [FAETP] | kg 1,4-DB-eq. per kg | 1.45 | 1.71 |
| 3.3 | Marine ecotoxicity potential [MAETP] | kg 1,4-DB-eq. per kg | 1.469 | 1.646 |
| 3.4 | Terrestrial ecotoxicity potential [TETP] | kg 1,4-DB-eq. per kg | 0.0571 | 0.1076 |
| 4 | Water consumption [Water] | m3 kg−1 | 0.1412 | 0.1510 |
| 5 | Abiotic depletion potential [ADP] | kg Sb-eq. per kg | 0.055 | 0.057 |
| 6 | Quantitative land use [Land Use] | m2 a kg−1 | 1.62 | 1.65 |
| 7 | Acidification potential [AP] | kg SO2-eq. per kg | 0.035 | 0.044 |
| 8 | Eutrophication potential [EDP] | kg PO4-eq. per kg | 0.012 | 0.012 |
| 9 | Ozone depletion potential [OLDP] | kg CFC11-eq. per kg | 8.94 × 10−7 | 8.80 × 10−7 |
| 10 | Photochemical oxidation creation potential [POCP] | kg C2H4-eq. per kg | 0.0018 | 0.0024 |
Both the single-reactor and two-reactor processes show comparable environmental impacts in terms of global warming potential [GWP 100a] (6.482 vs. 6.560 kg CO2-eq. per kg of THF-ALD-1) and non-renewable energy [NRE] (118 vs. 120 MJ per kg of THF-ALD-1) (Table 3). The impact of the biobased origin of FF is accounted for via a carbon sink of 1.19 kg CO2-eq. per kg THF-ALD-1, reducing the net GWP to 5.292 and 5.370 kg CO2-eq. per kg of THF-ALD-1 for the single-reactor and two-reactor processes, respectively. For both processes, the main contributors to GWP are MIBK (40%), FF (20%), heat (i.e. steam, cooling water) for distillation units (20%), and waste (aqueous and organic) (18%) (Fig. 4, left). The main contributors to NRE are MIBK (60%) and heat (20%) (Fig. 4, right). These results highlight that although the single-reactor process offers slight improvements in GWP and NRE, the difference between the two configurations is not significant in these specific categories.
 | ||
| Fig. 4 Impact score breakdown for Global Warming Potential [GWP 100a] and Non-Renewable Energy [NRE] for (1) single-reactor and (2) two-reactor processes. | ||
The single-reactor process demonstrates about a 7–16% reduction in most of the toxicity-related environmental impacts (except [TETP]) compared to the two-reactor process (Table 3): 2.05 vs. 2.21 kg 1,4-DB-eq. per kg of THF-ALD-1 for human toxicity potential [HTP], 1.45 vs. 2.21 kg 1,4-DB-eq. per kg of THF-ALD-1 for freshwater aquatic ecotoxicity potential [FAETP], 1.47 vs. 1.65 kg 1,4-DB-eq. per kg of THF-ALD-1 for marine aquatic ecotoxicity potential [MAETP] and 0.0571 vs. 0.1706 kg 1,4-DB-eq. per kg of THF-ALD-1 for terrestrial ecotoxicity potential [TETP]. The main contributor to HTP is FF (62%) and, to a lesser extent, MIBK (13%) and waste (8%) (Fig. 5). The main contributors to [HTP], [FAETP] and [MAETP] are catalysts (11%), FF (59%) and MIBK (10%); catalysts (12%), FF (52%) and waste (81%); and catalysts (13%), FF (38%), electricity (12%) and waste (28%), respectively.
The remaining indicators show values of 0.14 vs. 0.15 m3 per kg of THF-ALD-1 [Water], 0.055 vs. 0.057 kg Sb-eq. per kg of THF-ALD-1 [ADP], 1.62 vs. 1.65 m2 a per kg of THF-ALD-1 [Land Use], 0.035 vs. 0.044 kg SO2-eq. per kg of THF-ALD-1 [AP], 0.012 vs. 0.012 kg PO4-eq. per kg of THF-ALD-1 [EP], 8.94 × 10−7 vs. 8.80 × 10−7 kg CFC-11-eq. per kg of THF-ALD-1 [OLDP], and 0.0018 vs. 0.0024 kg C2H4 per kg of THF-ALD-1 [POCP]. These indicators exhibit diverging contributors (Fig. 6). The main contributor to [Water], [Land Use] and [EP] is FF at levels of 70%, 98% and 72%, respectively. ADP and OLDP are strongly affected by MIBK at levels of 58% and 56%, respectively, together with heat (21%) [ADP] and waste (11%) [OLDP]. Finally, the main contributors to [ADP] and [POCP] are catalysts (35%) and FF (40%) and catalysts (25%) and waste (30%), respectively. Details on the statistical significance of these results are provided later (see Uncertainly analysis).
Increasing the number of regeneration cycles has a significant effect on the environmental impacts, especially in toxicity and ecotoxicity categories (see the list of results in Tables S17–S19). Pd/Al2O3 catalyst regeneration affects all impact categories, especially [GWP 100a], [Water], [ADP], [EP], and [TETP] (Fig. 7–11). Increasing the number of regeneration cycles from C = 3 to C = 30 increases these impact categories by 77%, 49%, 417%, 81% and 76%, respectively, for the single-reactor process, and this increase is even higher for the two-reactor process with values of 105%, 2400%, 384%, 103% and 89%, respectively. Switching C from 30 to 300 increases almost exponentially the impact for all these categories, making both processes unfeasible. All these differences are statistically significant for a 95% confidence interval. The potential impact for the other categories is also very high, but lower than 200% at C = 30 for both processes (Fig. S11–S18). These results illustrate the strong impact of the number of Pd/Al2O3 regeneration cycles which should be kept below C = 3 to make both processes viable. From these results, we retained C = 3 and we performed a sensitivity analysis to evaluate the effect of the overall yield and the MIBK/FF molar ratio (i.e. flow rate of recirculated MIBK) on the impact scores for the single-reactor and two-reactor processes.
For both processes, an increase of the overall yield to 100% decreases the amount of waste generated by 33% and 47% for the single-reactor and two-reactor processes, respectively, and increases the H2 consumption by 7.5%. Reducing the MIBK/FF molar ratio from 18.2 to 10.2 at 100% overall yield decreases the flow rate of MIBK recirculated from 12% to 47% (streams 17 and 22 in the flowsheet diagrams for single-reactor and two-reactor processes, Fig. S1). Lower MIBK/FF molar ratios reduce the heat demand (i.e. steam, cooling water) for distillation columns from 9% to 38%. Incorporating process optimisations, i.e. increased reaction yield and reduced MIBK/FF molar ratio, into the environmental assessment reveals significant reductions in midpoint category impacts, particularly for the single-reactor process. The highest reductions compared to the base case (up to 25–30%) are observed for [GWP 100a], [NRE], [OLDP], [POCP], and [MAETP] (Fig. 12–16). These reductions are on average 10–15% lower for the two-reactor process compared to the single-reactor process. Reductions of about 20–25% compared to the base case are observed for [ADP], [HTP], [FAETP], and [TETP] (Fig. S19–S22), which are also 10–15% higher for the single reactor process. Finally, the potential reductions for the other categories are about 15–20% and are similar for both processes (Fig. S23–S26).
Overall, these results emphasise that process intensification—embodied in the single-reactor configuration—results in a consistently lower environmental impact. Maximising the overall reaction yield and reducing the MIBK/FF molar ratio significantly improve sustainability outcomes. At a controlled catalyst regeneration cycle count (C = 3), these optimisations provide meaningful environmental benefits that complement the FMC reduction. Thus, process optimisation alongside smart reactor design plays a crucial role in minimising the environmental footprint of THF-ALD-1 production.
When comparing the results from the sensitivity analysis for the above four impact categories, it can be observed that [AP] and [POCP] do not show any statistical difference for both processes (Fig. 17). Similarly, [FAETP] and [TETP] do not show statistically significant differences when increasing the overall reaction yield and decreasing the MIBK/FF molar ratio. In contrast, increasing the number of catalyst regeneration steps from C = 3 to C = 300 favours the single-reactor process for [FAETP] with statistical robustness, while slightly disadvantaging it for [TETP]. Furthermore, a comparison of the sensitivity analysis results within each process (i.e. base case vs. best case) shows that more than 70% of Monte Carlo runs favour the best-case scenario (Fig. 18), supporting the robustness of the simulation outcomes.
In contrast, regions in the Americas, such as Canada and Brazil, could reduce energy-related impacts by 50–80%, potentially diminishing the relative environmental advantage of the single-reactor configuration. Regions with moderate fossil energy shares (e.g., USA, Japan, Mexico) are expected to show impacts comparable to or slightly higher than Europe. Notably, impacts related to feedstock and catalysts remain largely independent of the energy mix.
These results highlight that the choice of location for implementing the processes can meaningfully affect the environmental profile, particularly in energy-intensive steps such as distillation, and should be considered in both process design and sustainability assessments.
Conclusions
This study assessed the potential of process intensification for producing a biobased biosolvent, THF-ALD-1, via the aldol condensation–crotonisation of furfural and MIBK, followed by hydrogenation. A comparative analysis between a conventional two-reactor process and an intensified single-reactor process was conducted from both economic and environmental perspectives, using life cycle assessment (LCA) and techno-economic analysis (TEA) at an early technology readiness level (TRL).The intensified single-reactor process demonstrated superior economic performance, achieving a full manufacturing cost of €4015 per ton, representing an 11% reduction compared to the two-reactor configuration. Sensitivity analysis identified Pd/Al2O3 catalyst recycling, the overall reaction yield, and the MIBK/FF molar ratio as the most influential levers for cost reduction. Optimisation of these parameters can yield cost benefits of up to 20%.
From an environmental perspective, the single-reactor process showed reductions in most midpoint categories. Statistically significant improvements were observed for the acidification potential (21%), photochemical oxidant creation potential (22%), freshwater ecotoxicity potential (19%), and terrestrial ecotoxicity potential (46%), as confirmed by Monte Carlo analysis. These improvements are primarily driven by slightly higher overall yields and reduced ethanol consumption, as the single-reactor process avoids γ-Al2O3 catalyst washing prior to regeneration. Directional trends toward lower impacts were also observed in other categories, although not all reached statistical significance.
Further environmental gains were achieved by limiting the Pd/Al2O3 catalyst regeneration cycles to three (C = 3), which substantially reduced impacts in ecotoxicity, acidification, and photochemical oxidant creation potential categories. Minimising Pd catalyst losses—through internal regeneration or external metal recovery—is therefore essential for sustainable operation. Process optimisation also showed that increasing the overall yield to 100% decreases waste generation by 32%, with only a minor increase in H2 consumption (+7.5%). Additionally, reducing the MIBK/FF molar ratio from 18.2 to 10.2 at full yield led to impact reductions of 32–36% (single-reactor) and 31–34% (two-reactor) in global warming potential, non-renewable energy, and abiotic and ozone depletion categories, all statistically significant according to Monte Carlo analysis.
The study also considers production scale and market context, assuming mid-tonnage continuous production of 3300 t per year. While THF-ALD-1 is a specialty chemical with limited publicly available market data, this scale is representative of plausible industrial deployment. At this stage, assumptions regarding process configuration, reactor design, and downstream purification are preliminary; a detailed pioneer-plant techno-economic analysis was considered outside the scope of this work.
Despite these limitations, the findings provide positive indications of the benefits of process intensification using a bifunctional Pd/Al2O3 catalyst, both economically and environmentally. These results support further development, including multi-site organic–inorganic hybrid catalysts and process optimisation, to enhance efficiency, reduce environmental burden, and inform future scale-up and commercialisation efforts of biobased chemicals.
Abbreviations
Variables
| Ai | Item contributing to the inventory in eqn (6) (variable unit) |
| C | Number of regeneration cycles of the Pd/Al2O3 catalyst (—) |
| Copex,t | Annual operating cost |
| CI | Confidence interval (variable unit) |
| CFt | Annual net cash flow (€) |
| CF | Characterisation factor of substance i in eqn (3) (variable unit) |
| Impactj | Impact attributed to variable j in eqn (3) (variable unit) |
| IRR | Internal return rate (%) |
| mi | Emission of substance i (—) |
| MSP | Minimum selling price (€) |
| NPV | Net present value (€) |
| PriceAi | Price of item Ai in eqn (6) |
| Rt | Annual revenue (€) |
Greek symbols
| σ | Uncertainty factor (—) |
Acronyms
| [AP] | Acidification potential |
| [ADP] | Abiotic depletion potential |
| ALD-1 | (E)-1-(Furan-2-yl)-5-methylhex-1-en-3-one |
| ALD-2 | 6-(Furan-2-yl)-2,10-dimethylundecane-4,8-dione |
| CAPEX | Capital expenditure |
| CED | Cumulative energy demand |
| [EP] | Eutrophication potential |
| FF | Furfural |
| FMC | Full manufacturing cost |
| F-ALD-1 | 1-(Furan-2-yl)-5-methylhexan-3-one |
| [FAETP] | Freshwater aqueous ecotoxicity potential |
| [GWP] | Global warming potential |
| [HTP] | Human toxicity potential |
| IPA | International Platinum Group Metal Association |
| ISBL | Inside battery limits |
| [Land Use] | Quantitative land use |
| LCI | Life cycle inventory |
| LCIA | Life cycle inventory analysis |
| LCA | Life cycle assessment |
| [MAETP] | Marine ecotoxicity potential |
| MIBK | Methyl isobutyl ketone |
| MC | Monte Carlo |
| [NRE] | Non-renewable energy |
| [OLDP] | Ozone layer depletion potential |
| OPEX | Operational expenditure |
| OSBL | Outside battery limits |
| [Renew] | Renewable energy |
| PGM | Platinum group metals |
| [POCP] | Photochemical oxidant creation potential |
| SPM | Solvay portfolio management |
| TEA | Techno-economic assessment |
| [TETP] | Terrestrial ecotoxicity potential |
| THF-ALD-1 | 5-Methyl-1-(tetrahydrofuran-2-yl)hexan-3-one |
| THF-ALD-2 | 2,10-Dimethyl-6-(tetrahydrofuran-2-yl)undecane-4,8-dione |
| VOC | Volatile organic compound |
| [Water] | Water use |
Author contributions
M. P.-T. conceived the study. C. Y. performed the calculations, data analysis and figure drawing. Both authors contributed to writing and revising the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
All relevant data supporting the findings of this study are available within the article and its supplementary information (SI). Flowsheets for single-reactor and two-reactor processes; Market price of PGM catalysts; Flowsheet for preparing Pd/Al2O3 catalyst; Contributions to environmental impact for the manufacture of Pd/Al2O3 and effect of Pd loading; Flowsheet for manufacturing FF from corn; Market price of FF, MIBK, H2 and ethanol; Effect of overall yield and MIBK/FF molar ratio on FMC for both processes; Breakdown of costs for both processes; Effect of number of regeneration cycles for Pd/Al2O3 on [NRE], [Land Use], [AP], [OLDP], [POCP], [HTP], [FAETP], [MAETP]; Effect of the overall yield and MIBK/FF molar ratio on [ADP], [HTP], [FAETP], [TETP], [Water], [Land Use], [AP], [EP]; Uncertainty analysis for the different impact categories; Log-normal plots from MC analysis for [GWP 100], [AP], [POCP], [FAETP], [TETP]; Physicochemical properties of chemicals for THF-ALD-1 production; Process inventory for THF-ALD-1 production; Summary of process assumptions; Key background datasets in LCIs; Revised Pd inventories; Process inventory for Pd/Al2O3; Environmental impacts for Pd/Al2O3; Amount of Pd reused and cost as a function of the number of regeneration cycles (C); Process inventory for the manufacture of FF; Pedigree matrix and scores for both processes; Breakdown of fixed and variable (base case) costs; Sensitivity analysis for FMC and environmental impacts for both processes; Breakdown of NRE for both processes; Definition of environmental impacts. See DOI: https://doi.org/10.1039/d5gc06149k.Additional raw data are available from the corresponding author upon reasonable request.
Acknowledgements
This project has received funding from the European Union's Horizon 2020 Research and Innovation Program under grant agreement no. 720783-MULTI2HYCAT.References
- (a) A. Koutinas, C. Du, R. H. Wang and C. Webb, Introduction to Chemicals from Biomass, Wiley, Hoboken, 2008, p. 77 Search PubMed; (b) D. M. Alonso, J. Q. Bond and J. A. Dumesic, Green Chem., 2010, 12, 1493 RSC; (c) J. A. Melero, J. Iglesias and A. Garcia, Energy Environ. Sci., 2012, 6, 7393 RSC; (d) M. Dusselier, M. Mascal and B. F. Sels, Top. Curr. Chem., 2014, 353, 1 CrossRef CAS PubMed.
- (a) J. F. Jenck, F. Agterberg and M. J. Droescher, Green Chem., 2004, 6, 544 RSC; (b) J. J. Bozell and G. R. Petersen, Green Chem., 2010, 12, 539 RSC.
- S. A. Miller, A. E. Landis and T. L. Theis, Environ. Sci. Technol., 2007, 41, 5176 CrossRef CAS PubMed.
- (a) H. E. Hoydonckx, W. M. van Rhijn, W. van Rhijn, D. E. de Vos and P. A. Jacobs, Furfural and Derivatives, in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, 2007 Search PubMed; (b) J. N. Chheda and J. A. Dumesic, Catal. Today, 2007, 123, 59 CrossRef CAS; (c) R. J. van Putten, J. C. van der Waal, E. de Jong, C. B. Rasrendra, H. J. Heeres and J. G. de Vries, Chem. Rev., 2013, 113, 1499 CrossRef CAS PubMed; (d) R. Mariscal, P. Maireles-Torres, M. Ojeda, I. Sádaba and M. López Granados, Energy Environ. Sci., 2016, 9, 1144 RSC.
- (a) C. J. Barrett, J. N. Chheda, G. W. Huber and J. A. Dumesic, Appl. Catal., B, 2006, 66, 111 CrossRef CAS; (b) W. Dedsuksophon, K. Faungnawakij, V. Champreda and N. Laosiripojana, Bioresour. Technol., 2011, 102, 2040 CrossRef CAS PubMed; (c) C. Ki, D. Ding, Q. Xia and Y. Wang, ChemSusChem, 2016, 9, 1712 CrossRef PubMed; (d) M. Gu, Q. Xia, X. Liu, Y. Guo and Y. Wang, ChemSusChem, 2017, 10, 4102 CrossRef CAS PubMed; (e) L. Faba, E. Diaz and S. Ordonez, Catal. Today, 2011, 164, 451 CrossRef CAS; (f) J. Yang, N. Li, G. Li, W. Wang, A. Wang, X. Wang, Y. Cong and T. Zhang, ChemSusChem, 2013, 6, 1149 CrossRef CAS PubMed; (g) M. Li, X. Xu, Y. Gong, Z. Wei, Z. Hou, H. Li and Y. Wang, Green Chem., 2014, 16, 4371 RSC; (h) L. Faba, E. Diaz and S. Ordonez, ChemSusChem, 2014, 7, 2816 CrossRef CAS PubMed.
- (a) L. Gao, I. Miletto, C. Ivaldi, G. Paul, L. Marchese, S. Coluccia, F. Jiang, E. Gianotti and M. Pera-Titus, J. Catal., 2021, 397, 75 CrossRef CAS; (b) M. C. Hernandez-Soto, C. Segarra, F. Rey, U. Diaz and M. Pera-Titus, Appl. Catal., A, 2022, 643, 118710 CrossRef CAS.
- (a) H. J. Cho, D. Kim, J. Li, D. Su and B. Xu, J. Am. Chem. Soc., 2018, 140, 13514 CrossRef CAS PubMed; (b) H. J. Cho, D. Kim and B. Xu, ACS Catal., 2020, 10, 4770 CrossRef CAS.
- L. Gao, Z. Jiang, I. Miletto, E. Gianotti, E. Rebmann, L. Baussaron, F. Jiang and M. Pera-Titus, Chem. Eng. J., 2023, 473, 145021 CrossRef CAS.
- (a) L. Luo, E. van der Voet, G. Huppes and H. A. Udo de Haes, Int. J. Life Cycle Assess., 2009, 14, 529 CrossRef CAS; (b) A. L. Borrion, M. C. McManus and G. P. Hammond, Renewable Sustainable Energy Rev., 2012, 16, 4638 CrossRef.
- International Organization for Standardization, ISO 14040:2006(E) Environmental management – Life cycle assessment – Principles and framework; Geneva, 2006; International Organization for Standardization, ISO 14044:2006(E) Environmental management.
- https://businessanalytiq.com/procurementanalytics/index/furfural-price-index/ (Accessed 6th June 2025).
- https://businessanalytiq.com/procurementanalytics/index/methyl-isobutyl-ketone-mibk-price-index/ (Accessed 6th June 2025).
- https://chinafinechemicals.en.made-in-china.com/product-group/lqITBtnCszpY/Ethanol-1.html (Accessed 6th June 2025).
- https://chinafinechemicals.en.made-in-china.com/product-group/lqITBtnCszpY/Ethanol-1.html (Accessed 6th June 2025).
- https://matthey.com/products-and-markets/pgms-and-circularity/pgm-management (Accessed 6th June 2025).
- The Environmental Profile of Platinum Group Metals, https://ipa-news.com/index/sustainability/ (Accessed 6th June 2025).
- Database Ecoinvent report – Part V Platinum Group Metals version 2.1, 2009 Search PubMed.
- T. Bossi and J. Gediga, The Environmental Profile of Platinum Group Metals, Johnson Matthey Technol. Rev., 2017, 61, 111.
- (a) R. S. Frazier, E. Jin and A. Kumar, Energies, 2014, 8, 621 CrossRef; (b) P. T. Benavides, D. C. Cronauer, F. Adoma, Z. Wang and J. B. Dunn, Sustainable Mater. Technol., 2017, 11, 53 CrossRef CAS.
- K. Noweck and W. Grafahrend, Fatty Alcohols, in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2006, p. 117 Search PubMed.
- H. Greenberg, J. S. Foot, M. G. Banwell and D. S. Roman, Palladium(II) Acetate, in Encyclopedia of Reagents for Organic Synthesis, Wiley-VCH, Weinheim, 2001, p. 1 Search PubMed.
- Y.-D. Zang, C.-J. Li, X.-Y. Song, J. Ma, J.-Z. Yang, N.-H. Chen and D.-M. Zhang, J. Asian Nat. Prod. Res., 2017, 19, 623 CrossRef CAS PubMed.
- Pro-Catalyse, Patent FR, 8309251, 1983 Search PubMed.
- Council of Scientific and Industrial Research (CSIR), Patent US, 7473406B2, 2003 Search PubMed.
- PW Consulting Chemical & Energy Research Center, Electronic grade Palladium Market, 2025, retrieved from https://pmarketresearch.com/chemi/electronic-grade-palladium-market/ (Accessed 10th November 2025) Search PubMed.
- J. Hong, J. Zhou and J. Hong, Int. J. Life Cycle Assess., 2015, 20, 623 CrossRef CAS.
- IHS Chemical, Furfural – CEH report, March 2016.
- J. B. Guinee, M. Gorree, R. Heijungs, G. Huppes, R. Kleijn, A. de Koning, L. van Oers, A. Wegener Sleeswijk, S. Suh, H. A. Udo de Haes, H. de Bruijn, R. van Duin, M. A. J. Huijbregts, E. Lindeijer, A. A. H. Roorda, B. L. van der Ven and B. P. Weidema, in Life Cycle Assessment. An Operation Guide to the ISO Standards, ed. J. B. Guinee (final editor), Centrum voor Milieukunde Leiden (CML), 2001 Search PubMed.
- Dübendorf: Swiss Centre for Life Cycle Inventories, Ecoinvent Database v2.2, 2010 Search PubMed.
- B. Weidema and M. S. Wesnæs, J. Cleaner Prod., 1996, 4, 167 CrossRef.
- J. H. Taylor, Eng. Process Econ., 1977, 2, 259 CrossRef.
- (a) Solvay's Sustainable Portfolio Management (SPM) Guide, Version 1.0, Solvay S.A, Brussels, 2021. Retrieved from https://www.solvay.com/sites/g/files/srpend221/files/2021-01/SolvaySPM-v10.pdf (Accessed 10th November 2025) Search PubMed; (b) World Business Council for Sustainable Development (WBCSD), 2017. Retrieved fromhttps://archive.wbcsd.org/Archive/Assess-and-Manage-Performance/Measuring-and-valuing-impact-business-examples/Solvay-Sustainable-Portfolio-Management (Accessed 10th November 2025).
- S. Morel, M. Traverso and P. Preiss, Discussion Panel—Assessment of Externalities: Monetisation and Social LCA, in Designing Sustainable Technologies, Products and Policies. From Science to Innovation, Luxemburg Institute of Science and Technology (LIST), Springer Open, 2017, p. 391 Search PubMed.
- Quantis, Solvay Case Study: Integrating Sustainability Metrics into Business Decisions, 2020. Retrieved from https://quantis.com/case-studies/solvay (Accessed 10th November 2025) Search PubMed.
- A. Bednarz, J. Beier, T. Grünenwald, B. Himmelreich, B. Hundt, F. A. Jaeger, M. Kirchner, S. Krinke, U. Létinois, C. Merz, L. Mohr, D. Morris, N. Otte, G. Rebitzer, P. Saling, U. W. Schenker, D. Schowanek, G. Vollmer, N. von der Assen and A. Wathelet, Life Cycle Management in Industry—Supporting Business with Life Cycle Based Assessments, in Designing Sustainable Technologies, Products and Policies. From Science to Innovation, Luxemburg Institute of Science and Technology (LIST), Springer Open, 2017, p. 351 Search PubMed.
- International Institute for Management Development (IMD), Are you ready for sustainable value creation?, 2022. Retrieved from https://www.imd.org/ibyimd/audio-articles/are-you-ready-for-sustainable-value-creation (Accessed 10th November 2025) Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |