Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Well-controlled synthesis of biodegradable polyesters using edible catalysts
Toshiki
Miwa
a,
Ryota
Suzuki
 b,
Tianle
Gao
b,
Takuya
Yamamoto
b,
Tianle
Gao
b,
Takuya
Yamamoto
 b,
Feng
Li
b,
Feng
Li
 *b,
Takuya
Isono
*b,
Takuya
Isono
 b and
Toshifumi
Satoh
b and
Toshifumi
Satoh
 *bcd
*bcd
aGraduate School of Chemical Sciences and Engineering, Hokkaido University, Sapporo, 060-8628, Japan
bDivision of Applied Chemistry, Faculty of Engineering, Hokkaido University, Sapporo 060-8628, Japan. E-mail: feng.li@eng.hokudai.ac.jp; satoh@eng.hokudai.ac.jp
cList Sustainable Digital Transformation Catalyst Collaboration Research Platform, Institute for Chemical Reaction Design and Discovery, Hokkaido University, Sapporo, 001-0021 Japan
dDepartment of Chemical & Materials Engineering, National Central University, Taoyuan 320317, Taiwan
First published on 2nd February 2026
Abstract
Biodegradable polyesters, represented by polylactic acid and poly(ε-caprolactone), are regarded as not only environmentally friendly alternatives to current nondegradable polyolefins but also important polymer materials for biomedical applications. The ring-opening polymerization (ROP) of the corresponding cyclic esters is a typical approach for synthesizing these polyesters in a well-controlled and efficient manner. A wide variety of organometallics and organocatalysts have been developed for this polymerization process. Organocatalysts are considered safer than organometallic catalysts because they do not release toxic metal residues, but not all of them are necessarily harmless to humans or the environment. Some truly safe and edible catalysts, including sodium acetate and vitamin C, have been reported to catalyze the ROP of lactide or ε-caprolactone. In this study, we explore the concept of “edible catalysts” by screening the structures of various organic acids and their sodium or potassium salts as catalysts for the ROP of cyclic esters. We demonstrate the suitability of various edible compounds for different cyclic ester monomers. The synthesized polyesters had a relatively narrow dispersity (Đ = 1.1–1.3) with a well-controlled polymer chain-end structure, identical to those obtained using conventional catalysts. Moreover, by mixing different edible catalysts, random and block copolymers can be synthesized in a well-controlled manner. These findings demonstrate that unassuming edible compounds in our daily lives possess significant potential as safe and effective catalysts for the synthesis of biodegradable polymer materials.
Green foundation1. In this work, we established a well-controlled ROP of cyclic esters to synthesize biodegradable polyesters, such as polylactic acid (PLA) and poly(ε-caprolactone) (PCL), by screening edible compounds comprised of organic acids and their alkali metal salts.2. Homopolymers and copolymers, including random-like copolymer PLA-stat-PCL and block copolymer PLA-b-PCL, were successfully synthesized by mixing different types of edible catalysts. 3. The use of truly safe and edible catalysts can eliminate the costly process of residual catalyst removal, thereby benefiting polymer production, particularly for biomedical applications. |
1. Introduction
With the increasing concern about environmental issues caused by nondegradable polymer materials, the development of (bio)degradable polymers is becoming urgent. Aliphatic polyesters (APEs) have emerged as prominent candidates owing to their widely tunable material properties and (bio)degradability.1–3 Polycondensation is a widely applied method for synthesizing these types of polymers; however, precisely controlling their molecular weights and dispersities is difficult. Ring-opening polymerization (ROP) of cyclic esters or cyclic carbonate monomers is another well-known approach for synthesizing APEs and aliphatic polycarbonates in a well-controlled manner, including controlling the molecular weight, distribution, chain-end functionality, and synthesis of block copolymers. Polylactic acid (PLA), poly(ε-caprolactone) (PCL), and poly(trimethylene carbonate) (PTMC) are among the most important and widely used biodegradable, biocompatible, and bioresorbable polymers, usually synthesized via ROP of their corresponding cyclic monomers: lactide (LA), ε-caprolactone (ε-CL), and trimethylene carbonate (TMC), respectively. To date, numerous catalysts, including organometallic, enzymatic, and organocatalysts, have been developed for ROP processes.4–7Since the landmark study by Hedrick et al., who accomplished the ROP of LA catalyzed by 4-dimethylaminopyridine (DMAP) in 2001,8 numerous reports on organocatalyzed polymerizations, particularly the ROP of the above-mentioned cyclic esters, have been published.9–13 Organocatalysts are generally recognized as more environmentally friendly and safer than organometallic catalysts because they are free of toxic metal residues. However, this claim is not necessarily valid. For example, DMAP is categorized as “Dangerous” in the materials safety data sheet, and it is toxic when placed in contact with skin or swallowed. Recent studies have demonstrated that phosphazene bases are cytotoxic.14 By contrast, Sn(Oct)2, a widely used organometallic catalyst for the ROP of cyclic esters,15 has been approved by the United States Food and Drug Administration, with an upper limit of 1 wt% tin in products that do not directly contact food. However, for biomedical applications such as bioresorbable sutures, implants, and drug delivery carriers, higher standards need to be met. For example, Evonik Industries AG launched RESORMER ZERO® products by cleanly removing the tin catalyst residues (<1 ppm) after polymerization. Therefore, truly safe polymer synthesis catalysts that do not require costly catalyst removal processes are needed.
Some truly safe and edible acid compounds (e.g., amino acids, citric acid (CA), tartaric acid) have been examined in the ROP of ε-CL.16 Our group has reported that alkali metal carboxylates (AMCs), some of which are safe as food additives, and a safe natural compound, trimethyl glycine, can catalyze the ROP of L-LA and TMC in a well-controlled manner.17–19 Other safe catalysts, including sodium or potassium carbonates, ascorbic acid (vitamin C), and its sodium salt, have also been reported to catalyze the abovementioned ROPs.20,21 Not only homopolymerization, but also random copolymerization of L-LA and ε-CL—a challenging yet useful task for the synthesis of biodegradable polymers with desired mechanical properties—have also been realized by benzoic acid, a commonly used preservative.22,23
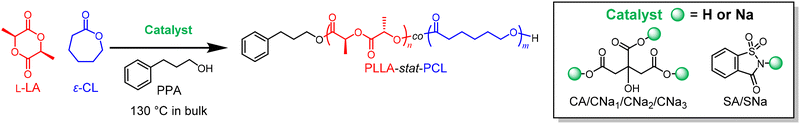
These scattered successes inspired us to further investigate the potential of truly safe and edible catalysts for the abovementioned ROPs. Here, we report our evaluation of safe catalysts selected from the food additive list or those rich in edible natural products, such as fruits, for the synthesis of biodegradable, biocompatible, and bioresorbable polymers via ROPs, with the goals of efficient catalytic performance, better controllability, and the synthesis of copolymers with various monomer sequences (Fig. 1).
 | ||
| Fig. 1 (A) Edible catalyst for the ROP of various cyclic esters; synthesis of copolymers of PLLA and PCL using mixed edible catalysts (B) or sequential addition of edible catalysts and monomers (C). | ||
2. Experimental
2.1. Chemicals
L-Lactide (L-LA; >98.0%, Tokyo Kasei Kogyo (TCI)) was purified twice via recrystallization from dry toluene. Trimethylene carbonate (TMC, >98.0%, TCI) was purified by recrystallization from dry tetrahydrofuran and diethyl ether. ε-Caprolactone (ε-CL; >99.0%, TCI), δ-valerolactone (δ-VL; >99%, Sigma-Aldrich), and 3-phenyl-1-propanol (PPA; >98.0%, TCI) were distilled over CaH2 under reduced pressure. 1,4-Benzenedimethanol (BDM; >99.0%, TCI) was dried under high vacuum prior to use. CA (>98.0%, TCI), sodium dihydrogen citrate anhydrous equivalent (CNa1; ≥97.0%, Nacalai Tesque), disodium dihydrogen citrate 1.5 hydrate (CNa2; ≥98.0%, TCI) trisodium citrate (CNa3; ≥98.0%, Sigma-Aldrich), potassium dihydrogen citrate (CK1; 95%, Fujifilm Wako Pure Chemicals), tripotassium citrate monohydrate (CK3; ≥99.0%, Kanto Chemical), saccharin (SA; >99.0%, TCI), saccharin sodium salt dihydrate (SNa; >98.0%, TCI), and tetrasodium ethylenediaminetetraacetate tetrahydrate (EDTANa4; >98.0%, TCI) were dried by heating at 100 °C under high vacuum for at least 72 h prior to use. Malonic acid (MA; >99.0%, TCI), disodium malonate (MNa2; >99.0%, TCI), and ethylenediaminetetraacetic acid (EDTA; >98.0%, Kanto Chemical) were used as received.Chloroform-d (CDCl3; >99.8%), methanol (>99.8%, Sigma-Aldrich), dichloromethane (CH2Cl2; >98.0%, JUNSEI CHEMICAL), tetrahydrofuran (THF; >99.0%, JUNSEI CHEMICAL), and dry toluene (>99.5%, water content <0.001%, KANTO CHEMICAL) were used as received.
2.2. Instruments
Polymerization was set up in a MBRAUN stainless steel glovebox equipped with a gas purification system (molecular sieves and copper catalyst) in a dry argon atmosphere (H2O, O2 < 0.1 ppm). MB-MO-SE 1 and MB-OX-SE 1 were used to monitor the moisture and oxygen contents in the glovebox, respectively.1H and 13C NMR spectra were recorded at 25 °C on JEOL JNM-ECS 400, JEOL JNMECX 400 (400 MHz), and JNM-ECZ600R (600 MHz) instruments using chloroform-d as the solvent, and chemical shifts were referenced to an internal standard.
The Fourier transform infrared spectroscopy (FT-IR) spectra were obtained using a PerkinElmer Frontier MIR spectrometer equipped with a single reflection diamond universal attenuated total reflection (ATR) accessory.
Size exclusion chromatography (SEC) was conducted at 40 °C in THF (flow rate, 1.0 mL min−1) using a Jusco high-performance liquid chromatography system (PU-4180 HPLC pump, AS-4550 auto sampler and CO-4060 column oven) equipped with a Shodex K-800D guard column (8.0 mm × 100 mm; particle size, 10 µm) and two Shodex columns (K-806L and K-804L; linear, 8.0 mm × 100 mm; particle size, 10 µm). A polystyrene standard curve ranging from 2170 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 320
320![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 was used for calibration to determine the molecular weights (Mn, SEC) and polydispersity indices (Ð) of the polymers.
000 was used for calibration to determine the molecular weights (Mn, SEC) and polydispersity indices (Ð) of the polymers.
Matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS) was performed using a Bruker Daltonics (Germany) Ultraflex MALDI TOF/TOF system equipped with a 355 nm Nd:YAG laser. Five hundred shots were accumulated for the spectra, and the acceleration voltage was adjusted between 10 and 30 kV, depending on the molecular weight and nature of each analyzed compound in reflector mode. The poly(L-lactide) (PLLA) and PTMC samples for MALDI-TOF MS were prepared by depositing a mixture of the polymer (4.0 mg mL−1, 0.5 μL) and a matrix (2,5-dihydroxybenzoic acid, 60 mg mL−1, 0.5 μL) in THF on a sample plate, which was coated by an acetone solution (1.0 mmol L−1, 1.0 μL) of NaI as the cationic agent. For poly(ε-caprolactone) (PCL) and poly(δ-valerolactone) (PVL) samples, only the polymer (1.0 mg mL−1) and matrix (15 mg mL−1) in THF were changed.
2.3. Polymerization
M
n, NMR = 8930; Mn, SEC = 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200; Đ = 1.13.
200; Đ = 1.13.
M n, NMR = 4340; Mn, SEC = 4500; Đ = 1.24.
M n, NMR = 9400; Mn, SEC = 9800; Đ = 1.46.
M
n, NMR = 7800; Mn, SEC = 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000; Đ = 1.18.
000; Đ = 1.18.
3. Results and discussion
3.1. Catalyst Screening
Carboxylic acids and their corresponding alkali metal salts have been demonstrated to be active catalysts for the ROP of cyclic esters, according to our previous research and other researchers. We examined a list of food additives and natural products to select safe catalysts.Saccharin is a non-nutritive artificial sweetener with a moderately strong acidity similar to that of diphenyl phosphate (DPP), a well-known organocatalyst for the ROP of ε-caprolactone (ε-CL); the pKa values of saccharin and DPP are 4.0 and 3.88 in DMSO, respectively.24–26 Saccharins and their derivatives have been used as catalysts in various organic reactions.27 Notably, the DMAP–saccharin (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) salt has been reported to catalyze the ROP of L-LA.28 Therefore, we anticipated that saccharin and its sodium or potassium salts could be safe catalysts for ROP of cyclic esters, such as L-LA and ε-CL.
1) salt has been reported to catalyze the ROP of L-LA.28 Therefore, we anticipated that saccharin and its sodium or potassium salts could be safe catalysts for ROP of cyclic esters, such as L-LA and ε-CL.
From the viewpoint of chemical structure, saccharin is a monofunctional Brønsted acid. The integration of two or more identical functional groups into one molecule, producing bifunctional or multifunctional catalysts, is a common strategy to improve catalytic performance, which is attributed to their synergistic effect.29 Therefore, we investigated safe di-, tri-, and tetracarboxylic acids and their corresponding alkali metal salts.30 With this consideration in mind, malonic acid (MA), citric acid (CA), ethylenediaminetetraacetic acid (EDTA), and their sodium salts were selected as safe catalysts in the ROP of L-LA and ε-CL (Fig. 2 and Table S1). The initial screening for ROP was conducted using BDM as an initiator at a [monomer]0/[BDM]0/[catalyst] ratio of 100/1/5 for 24 h at 100 °C in bulk.
 | ||
| Fig. 2 Bar graph illustrating monomer conversion (conv.) in the ROP of L-LA (left) and ε-CL (right) using various catalysts. | ||
In the ROP of L-LA using acid catalysts, the monomer conversion was less than 10%, and the formation of PLLA was not confirmed (runs 1–4 in Table S1). By contrast, employing sodium salts as catalysts led to significantly improved monomer conversion, and white powdered solid PLLA was obtained as the product for all catalysts (runs 5–8 in Table S1). When ε-CL was employed as the monomer, significant polymerization progress was observed only for saccharin and CA (runs 9–12 in Table S1). By contrast, their sodium salts showed considerably low catalytic activities in the ROP of ε-CL (runs 13–16 in Table S1). These results indicate that sodium salts are effective catalysts for the ROP of L-LA, while organic acid catalysts are effective for the ROP of ε-CL. This trend aligns with previous reports indicating that base catalysts are more active for L-LA and acid catalysts for the ROP of ε-CL.17,30,31
Based on a rough catalyst screening that considered performance in the acid-catalyzed ROP of ε-CL and the corresponding sodium salt-catalyzed ROP of L-LA, CA and citrate were selected as optimal catalysts. Therefore, we applied them in a more detailed investigation of the polymerization conditions, including the catalyst loading, monomer-to-initiator ratio, alkali metal species, and polymerization in the presence or absence of solvent. The investigation commenced with the bulk polymerization of L-LA at 100 °C. 3-Phenyl-1-propanol (PPA) was used as the initiator, and polymerization was conducted at a [L-LA]0/[PPA]0/[CNa3] = 50/1/1 (run 1 in Table 1). After 66.5 h of polymerization initiation, the monomer conversion reached 76.9%. The viscosity of the reaction mixture increased progressively during the reaction, indicating PLLA formation. The number-average molecular weight (Mn, NMR) determined from 1H NMR measurements was 6050, which was in reasonable agreement with the theoretical value (Mn, th. = 5680), calculated based on the initial monomer-to-initiator ratio and monomer conversion. The dispersity of the obtained PLLA was as narrow as 1.13. By increasing the catalyst loading to [L-LA]0/[PPA]0/[CNa3] = 50/1/5, the polymerization rate increased, with a result similar to that obtained for PLLA (run 2 in Table 1 and Fig. 3a). When the targeted degree of polymerization (DP) was increased to 150, viz. [L-LA]0/[PPA]0/[CNa3] = 150/1/5, the molecular weight of the obtained PLLA increased accordingly, without sacrificing controllability (run 3 in Table 1 and Fig. 3b). When tripotassium citrate (CK3) was employed as a catalyst, 91.1% of the monomer was converted 18.5 h after the onset of polymerization, indicating a faster progression of polymerization compared with that using sodium citrate (run 1 vs. 4 in Table 1). This trend is the same as that observed in our previous study on the counter-cation effect of AMCs in the ROP of L-LA.17,19 Given that CK3 exhibited higher reactivity than CNa3, the targeted DP was increased to 150, and the polymerization as scaled up to 5 g to evaluate its scalability (run 5 in Table 1 and Fig. S1a). The polymerization proceeded at a reasonably fast rate while maintaining good control over the polymerization, indicating the promising potential of this system for scale-up. The 1H NMR spectrum of the obtained PLLA showed signals from both the PLLA main chain and 3-phenyl-1-propoxy group (Fig. 3a). In the MALDI-TOF mass spectrum of PLLA, two series of peaks were apparent, corresponding to PPA as the major peak and water at the initiating end as the minor peak (Fig. 4).
 | ||
| Fig. 3 (a) 1H NMR spectrum of the PLLA obtained from run 2 in Table 1 (solvent, CDCl3; 400 MHz), and (b) SEC traces of the PLLAs obtained from runs 2 and 3 in Table 1 (eluent, THF; flow rate, 1.0 mL min−1). | ||
 | ||
| Fig. 4 (a) MALDI-TOF mass spectrum of PLLA (run 2 in Table 1), (b) expanded spectrum (ranging from 2000 to 6500), and (c) theoretical values. | ||
| Run | Monomer | Catalyst | [Monomer]0/[PPA]0/[catalyst] | Time (h) | Conv.b (%) |
M
n, th.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c (g mol−1) c (g mol−1) |
M
n, NMR![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (g mol−1) b (g mol−1) |
M
n, SEC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d (g mol−1) d (g mol−1) |
Đ |
|---|---|---|---|---|---|---|---|---|---|
| a Polymerization conditions: atmosphere, Ar; initiator, PPA; temperature, 100 °C. b Determined using 1H NMR spectroscopy. c Calculated from [monomer]0/[PPA]0 × conv. × (M.W. of the monomer) + (M.W. of PPA). d Determined using SEC in THF with a polystyrene standard. e 5 g scale polymerization. f Polymerization was conducted in the presence of toluene (ε-CL/toluene = 25 mmol/1 mL). | |||||||||
| 1 | L-LA | CNa3 | 50/1/1 | 66.5 | 76.9 | 5680 | 6050 | 8000 | 1.13 |
| 2 | L-LA | CNa3 | 50/1/5 | 39.0 | 80.8 | 5960 | 6050 | 7700 | 1.12 |
| 3 | L-LA | CNa3 | 150/1/5 | 134.0 | 75.3 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
1.19 |
| 4 | L-LA | CK3 | 50/1/1 | 18.5 | 91.1 | 6700 | 6620 | 7200 | 1.13 |
| 5e | L-LA | CK3 | 150/1/1 | 48.0 | 78.8 | 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
1.15 |
| 6 | ε-CL | CA | 50/1/1 | 12.5 | 72.9 | 4300 | 4360 | 7600 | 1.11 |
| 7f | ε-CL | CA | 150/1/1 | 103.0 | 79.0 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
1.29 |
| 8e,f | ε-CL | CA | 150/1/1 | 120.0 | 76.8 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
1.27 |
A detailed investigation of the ROP of ε-CL using CA as the catalyst was initially conducted in a [ε-CL]0/[PPA]0/[CNa3] ratio of 50/1/1 (run 6 in Table 1). The monomer conversion was 72.9% after 12.5 h. The obtained PCL showed a narrow dispersity, Đ = 1.11, and Mn, NMR = 4360 is nearly the same as Mn, th. = 4300 (Fig. 5a). When the target DP was increased to 150, the polymerization proceeded well, with a slightly broader molecular weight distribution (run 7 in Table 1 and Fig. 5b). A similar result was obtained when the polymerization was scaled up 5 g (run 8 in Table 1 and Fig. S1a). Regarding the role of CA in the polymerization of ε-CL, CA has been used either as a catalyst or initiator.16,32 Therefore, we aimed to clarify the role of CA in our study. The 1H-NMR spectrum and MALDI-TOF mass spectra of the synthesized PCL were obtained (Fig. 5a and 6). Signals corresponding to PPA were observed in the 1H-NMR spectrum. Moreover, the MALDI spectrum showed only one series of peaks, which were assumed to correspond to the ones with PPA structure. Therefore, in this polymerization system, CA functions only as a catalyst, whereas PPA acts as an initiator. Notably, the level of dispersity control achieved in this study is comparable to, or even superior to, that reported in representative works employing benzoic acid, vitamin C, or its sodium salt, while simultaneously exhibiting significantly higher catalytic activity. In contrast, those studies typically require substantially higher reaction temperatures (155–170 °C).20,22
 | ||
| Fig. 5 (a) 1H NMR spectrum of the PCL obtained from run 6 in Table 1 (solvent, CDCl3; 400 MHz), and (b) SEC traces of the PCLs obtained from runs 6 and 7 in Table 1 (eluent, THF; flow rate, 1.0 mL min−1). | ||
 | ||
| Fig. 6 (a) MALDI-TOF mass spectrum of PCL (run 6 in Table 1), and (b) expanded spectrum (ranging from 4750 to 4070), and (c) theoretical values. | ||
3.2. Monomer scope
To further investigate the catalytic activity in the ROP of other cyclic monomers, TMC and δ-VL were polymerized (Scheme 1). Each ROP was performed in bulk at 100 °C with a [monomer]0/[PPA]0/[catalyst] ratio of 50/1/5 (Table 2). For the ROP of TMC using CA as a catalyst, the monomer conversion reached 56.9% 9.5 h after polymerization initiation, confirming polymer formation (run 1 in Table 2). By contrast, when CNa3 was used as a catalyst, the conversion reached 86.0% after only 1.5 h, demonstrating a much higher catalytic activity than that of run 1, involving an acidic catalyst (run 2 in Table 2). However, the molecular weight distribution was slightly broad (Đ = 1.30) (Fig. S2a). The 1H NMR spectrum of the obtained PTMC showed signals attributed to both the PTMC main-chain and PPA residues (Fig. S2b). In the MALDI-TOF mass spectrum, small subpeaks corresponding to transesterification and 1,3-propanediol initiation were observed (Fig. S3). This may explain the slightly broader dispersion observed. When the targeted DP was increased to 150 with a decreased CNa3 catalyst loading, viz. [TMC]0/[PPA]0/[CNa3] = 150/1/1, the molecular weight of the resulting PTMC increased accordingly, accompanied by a slightly higher dispersity (run 3 in Table 1 and Fig. S2c). These results demonstrated that CNa3 could function as a catalyst for the controlled synthesis of PTMC, although, upon comparison with our previous finding, it did not outperform sodium acetate.33| Run | Monomer | Catalyst | [Monomer]0/[PPA]0/[catalyst] | Time (h) | Conv.b (%) |
M
n, th.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c (g mol−1) c (g mol−1) |
M
n, NMR![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (g mol−1) b (g mol−1) |
M
n, SEC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d (g mol−1) d (g mol−1) |
Đ |
|---|---|---|---|---|---|---|---|---|---|
| a Polymerization conditions: atmosphere, Ar; initiator, PPA; temperature, 100 °C. b Determined from 1H NMR spectrum of the obtained polymer in CDCl3. c Calculated from [monomer]0/[PPA]0 × conv. × (M.W. of the monomer) + (M.W. of PPA). d Determined using SEC in THF with a polystyrene standard. e Temperature, 70 °C. f Polymerization was conducted in the presence of toluene (δ-VL/toluene = 25 mmol/1 mL). (n.d = not determined). | |||||||||
| 1 | TMC | CA | 50/1/5 | 9.5 | 56.9 | 3040 | 4320 | 3800 | 1.20 |
| 2 | TMC | CNa3 | 50/1/5 | 1.5 | 86.0 | 4530 | 4830 | 6400 | 1.30 |
| 3e | TMC | CNa3 | 150/1/1 | 15.0 | 74.2 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 900 |
15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
1.38 |
| 4 | δ-VL | CA | 50/1/5 | 2.0 | 88.5 | 4570 | 4340 | 4500 | 1.24 |
| 5 | δ-VL | CNa3 | 50/1/5 | 24.0 | 11.0 | n.d. | n.d. | n.d. | n.d. |
| 6f | δ-VL | CA | 150/1/1 | 30.0 | 82.3 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
1.13 |
The ROP of δ-VL yielded similar results to the ROP of ε-CL. When CA was used as the catalyst, 88.5% of the monomer was converted within 2 h and produced PVL with fairly good control of the molecular weight dispersity (run 4 in Table 2 and Fig. S4a). Good polymerization control also reflects the good agreement between Mn, NMR and Mn, th., together with the 1H NMR and MALDI-TOF mass spectra (Fig. S4b and S5). By contrast, the ROP of δ-VL was sluggish when using CNa3 as the catalyst: only 11% of the monomer was consumed after 24 h (run 5 in Table 2). This result is consistent with our previous study on AMC-catalyzed ROP of δ-VL, in which a low polymerization rate was observed.17 Given that CA exhibited higher catalytic activity in the ROP of δ-VL, polymerization targeting a DP of 150 using CA catalyst was conducted. As a result, PVL with narrow dispersity (Đ = 1.13) and correspondingly higher molecular weight was obtained (run 6 in Table 2 and Fig. S4c). These investigations demonstrate that the ROP of cyclic esters and carbonate monomers with drastically different reactivities can be accomplished by screening the edible catalysts, particularly acids with sufficient acidity and their corresponding alkali metal salts.
3.3. Copolymerization of L-LA and ε-CL
Except for the polyester and polycarbonate homopolymers mentioned above, copolymers including statistical and block copolymers could further tune the material properties to meet broader application demands. In particular, the copolymers of LA and ε-CL are the most widely applied.The statistic copolymers of LA and ε-CL are mostly synthesized via copolymerization of LA and ε-CL using organometallic catalysts such as Sn(Oct)2 and alumina-based catalysts. In most cases, LA is consumed faster than ε-CL, thus resulting in a monomer sequence gradient.34–38 By carefully tuning the structure of ligands in the Salen complex of aluminum, Nomura et al. realized a nearly perfect random copolymerization of L-LA and ε-CL.39 More recently, Taton et al. used simple and safe benzoic acid as a catalyst for the random copolymerization of L-LA and ε-CL, although a reaction temperature of as high as 155 °C was required.22,23 Pang et al. used sodium ascorbate to catalyze the copolymerization of L-LA and ε-CL under 170 °C, claiming to obtain the random copolymer based on an analysis of the copolymer; however, they did not analyze the polymerization kinetics.20
In this study, we have already demonstrated that the ROP of ε-CL and L-LA can proceed efficiently and in a well-controlled manner using the edible catalysts CA and CNa3, respectively. Between CA and CNa3, there are medium states, viz. monosodium citrate (CNa1) and disodium citrate (CNa2). These can be regarded as mixtures of CA and CNa3. As a preliminary experiment, the homopolymerizations of L-LA and ε-CL conducted using CNa1, CNa2, CK1, and CK3 as a catalyst (Scheme S1). These results confirmed that CNa1 and CNa2 exhibited catalytic behaviors intermediate to those of CA and CNa3 (Tables S2, S3 and Fig. S6). We wondered if CNa1 and CNa2 could catalyze the copolymerization of ε-CL and L-LA in the fashion between CA and CNa3, thus resulting in the copolymers having near-random monomer sequences. With this idea in mind, the copolymerization of L-LA and ε-CL was conducted using CA, CNa1, CNa2, and CNa3, respectively (Scheme 2 and runs 1–4 in Table 3). The copolymerizations were conducted in the ratio of [L-LA]0/[ε-CL]0/[PPA]0/[catalyst] = 50/50/1/1 under 130 °C in bulk. When CA was utilized, after 80 h, the conversion of L-LA and ε-CL reached 49.1% and 78.0%, respectively (run 1 in Table 3). By contrast, when CNa3 was used as the catalyst, the copolymerization demonstrated the opposite polymerization kinetics, with 72.5% and 4.9% consumption of L-LA and ε-CL, respectively, after 25 h (run 2 in Table 3). These results align with our previous results for the homopolymerization of L-LA and ε-CL using these two catalysts, respectively, although the difference in their reactivity in the copolymerization is not as obvious as that in their homopolymerization, when CA is used at the catalyst (Fig. 2). When CNa1 and CNa2 were applied as catalysts under the same polymerization condition, to our surprise, they exhibited similar reactivities, with 91%–92% and 18%–20% conversion of L-LA and ε-CL, respectively, after 22.5 h (runs 3 and 4 in Table 3). In both cases, L-LA was consumed faster than ε-CL, whose catalytic kinetics were closer to the CNa3-catalyzed polymerization. These results indicate that in the catalytic system using CNa1 and CNa2, the catalyst forms are in equilibrium among the four possible states (CA, CNa1, CNa2, and CNa3), among which CNa3 may have the highest catalytic reactivity (Scheme S2). The obtained copolymers under these four copolymerization conditions have relatively broad molecular weight distributions, Đ = 1.40–1.61 (Fig. S7–S10). The Mn, SEC values of the obtained four copolymers are obviously lower than Mn, th. compared with the results in homopolymerizations (Table 1). This presumably indicates an undesired initiation from a species other than the PPA initiator.
| Run | Catalyst | Time (h) | Conv.L-LA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d (%) d (%) |
Conv.ε-CL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d (%) d (%) |
L-LA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ε-CL in copolymerd ε-CL in copolymerd |
M
n, th.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e e |
M
n, NMR![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d d |
M
n, SEC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) f f |
Đ |
|---|---|---|---|---|---|---|---|---|---|
| a Polymerization conditions: atmosphere, Ar; initiator, PPA; temperature, 130 °C. b [L-LA]0/[ε-CL]0/[PPA]0/[catalyst] = 50/50/1/1. c [L-LA]0/[ε-CL]0/[PPA]0/[catalyst] = 50/50/1/5. d Determined by 1H NMR spectroscopy of the obtained polymer in CDCl3. e Calculated from [L-LA]0/[PPA]0 × conv.L-LA × (M.W. of L-LA) + [ε-CL]0/[PPA]0 × conv.ε-CL × (M.W. of ε-CL) + (M.W. of PPA). f Determined by SEC in THF using a polystyrene standard. g Determined from the crude product. h Temperature, 160 °C. | |||||||||
| 1b | CA | 80.0 | 49.1 | 78.0 | n.d. | 8130 | n.d. | 2800g | 1.40g |
| 2b | CNa3 | 25.0 | 72.5 | 4.9 | n.d. | 5640 | n.d. | 3700g | 1.55g |
| 3b | CNa1 | 22.5 | 91.6 | 19.7 | n.d. | 7860 | n.d. | 4100g | 1.62g |
| 4b | CNa2 | 22.5 | 91.3 | 18.0 | n.d. | 7740 | n.d. | 4100g | 1.61g |
| 5c | SA | 164.0 | 22.6 | 78.3 | 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75 75 |
6220 | 6090 | 6600 | 1.39 |
| 6c | SNa | 50.0 | 85.9 | 10.1 | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
6900 | 5200 | 6800 | 1.22 |
| 7c | SA/SNa = (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) 4) |
168.0 | 83.8 | 53.4 | 62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 38 38 |
9220 | 8900 | 7900 | 1.30 |
| 8c | SA/SNa = (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
305.0 | 73.0 | 73.8 | 49![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 51 51 |
9610 | 9400 | 9800 | 1.46 |
| 9c,h | SA/SNa = (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
48.0 | 96.0 | 83.0 | 55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45 45 |
11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600 |
6700 | 1.62 |
The possible equilibrium between the four states in the CA and sodium citrate systems was too complicated to analyze. To simplify the system, we focused on a saccharin/saccharin sodium salt (SA/SNa) system, in which the catalysts have only two possible states. When SA was used as the catalyst for copolymerization, 22.6% of L-LA and 78.3% ε-CL were converted, respectively, after 164 h (run 5 in Table 3). By contrast, when using SNa, the consumption of L-LA and ε-CL were 85.9% and 10.1%, respectively, after 50 h (run 6 in Table 3). The selectivity of the two monomers was in good agreement with their homopolymerization results, although decreased reactivity was observed during copolymerization. When SA and SNa were mixed at a molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, L-LA was still consumed faster than ε-CL (83.8% vs. 53.4% conversion, respectively); however, the difference in consumption rates was smaller than that observed when using SNa alone as the catalyst (run 7 vs. run 6 in Table 3 and Fig. S11). When mixing SA and SNa in a molar ratio of 3
4, L-LA was still consumed faster than ε-CL (83.8% vs. 53.4% conversion, respectively); however, the difference in consumption rates was smaller than that observed when using SNa alone as the catalyst (run 7 vs. run 6 in Table 3 and Fig. S11). When mixing SA and SNa in a molar ratio of 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, L-LA and ε-CL were consumed at almost the same rate, but the reaction time was long (run 8 in Table 3, Fig. 7 and S12). In terms of the copolymers obtained using the SA/SNa catalytic system, although the dispersity index was still slightly broad (Đ = 1.22–1.46), Mn, NMR and Mn, SEC were in good agreement with Mn, th., indicating fairly good polymerization control (runs 5–8 in Table 3 and Fig. S13–S16). Increasing the polymerization temperature to 160 °C indeed shortened the reaction time; however, a broader dispersity (Đ = 1.62) and a lower Mn, SEC compared with Mn, NMR were observed, indicating a reduced level of polymerization control (run 9 in Table 3, Fig. S17 and S18). The obtained PLLA-stat-PCL copolymer was characterized using 1H-NMR and quantitative 13C-NMR (run 8 in Table 3, Fig. 8 and S19). In particular, the 13C NMR spectrum revealed multiple peaks arising from the complex arrangement of L-LA and ε-CL units, confirming the formation of a statistical copolymer. Notably, the absence of peaks corresponding to the CLC sequence at ∼171 ppm (C = ε-CL, L = lactic acid) indicates that transesterification does not occur in this copolymerization.40 The average sequence lengths of L-LA and ε-CL units in the copolymer were calculated to be LL-LA = 2.7 and Lε-CL = 2.9, respectively (Fig. S20).41 Although these values are slightly higher than those for an ideal random copolymer (LL-LA = Lε-CL = 2.0), they demonstrate that the obtained copolymer exhibits random-like characteristics.
2, L-LA and ε-CL were consumed at almost the same rate, but the reaction time was long (run 8 in Table 3, Fig. 7 and S12). In terms of the copolymers obtained using the SA/SNa catalytic system, although the dispersity index was still slightly broad (Đ = 1.22–1.46), Mn, NMR and Mn, SEC were in good agreement with Mn, th., indicating fairly good polymerization control (runs 5–8 in Table 3 and Fig. S13–S16). Increasing the polymerization temperature to 160 °C indeed shortened the reaction time; however, a broader dispersity (Đ = 1.62) and a lower Mn, SEC compared with Mn, NMR were observed, indicating a reduced level of polymerization control (run 9 in Table 3, Fig. S17 and S18). The obtained PLLA-stat-PCL copolymer was characterized using 1H-NMR and quantitative 13C-NMR (run 8 in Table 3, Fig. 8 and S19). In particular, the 13C NMR spectrum revealed multiple peaks arising from the complex arrangement of L-LA and ε-CL units, confirming the formation of a statistical copolymer. Notably, the absence of peaks corresponding to the CLC sequence at ∼171 ppm (C = ε-CL, L = lactic acid) indicates that transesterification does not occur in this copolymerization.40 The average sequence lengths of L-LA and ε-CL units in the copolymer were calculated to be LL-LA = 2.7 and Lε-CL = 2.9, respectively (Fig. S20).41 Although these values are slightly higher than those for an ideal random copolymer (LL-LA = Lε-CL = 2.0), they demonstrate that the obtained copolymer exhibits random-like characteristics.
 | ||
| Fig. 7 Plots of monomer conversion vs. time (runs 5, 6, and 8 in Table 3). | ||
 | ||
| Fig. 8 13C NMR spectrum of the PLLA-stat-PCL obtained from run 8 in Table 3 (solvent, CDCl3; 100 MHz). | ||
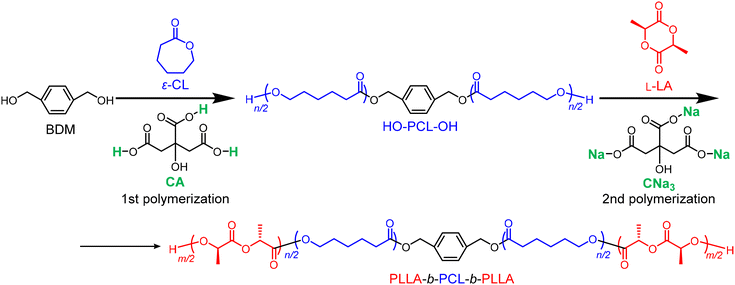
Block copolymers made from PLA and PCL, including ABA-type triblock and multiblock copolymers, are biodegradable and bioresorbable thermoplastic elastomers used in various biomedical applications. Jikei et al. synthesized multiblock copolymers via self-polycondensation of PLA-b-PCL diblock copolymers and demonstrated their effectiveness as adhesion barrier films in surgeries.42 Liu et al. reported that star-shaped PCL-b-PLLA block copolymers are promising candidates for cardiovascular tissue engineering and absorbable medical devices.43 However, in both cases, Sn(Oct)2 was used as the catalyst.
In our previous study on AMCs, we successfully synthesized a PTMC-b-PLLA block copolymer using sodium acetate as a catalyst.33 However, this remains a challenge for PLA-b-PCL block copolymers. Using benzoic acid, Taton et al. synthesized a PLLA-b-PCL-b-PLLA triblock copolymer via the sequential addition of monomers in a one-pot/two-step fashion; however, the ROP of L-LA required harsh conditions and a long reaction time.22
Owing to the drastically different characteristics of ε-CL and L-LA in terms of their polymerization conditions when using organocatalysts, a two-step synthesis involving a catalyst change is often needed.44–46 In this study, we have already demonstrated that the ROP of ε-CL and L-LA can proceed efficiently and in a well-controlled manner when catalyzed by edible CA and sodium citrate, respectively. We wondered whether these two catalysts could be used to synthesize PCL-b-PLLA block copolymers in a one-pot/two-step fashion. As shown in Scheme 3, from the bifunctional initiator BDM, the ROP of ε-CL was first performed using CA as the catalyst. When ε-CL was almost completely consumed, L-LA together with CNa3 were added to synthesize the subsequent PLLA block. Mixing CA and CNa3 generates a complex equilibrium of four species, including two new species: CNa1 and CNa2. According to the performance of CA, CNa1, CNa2, and CNa3 in the copolymerization of L-LA and ε-CL (runs 1–4 in Table 3), the catalyst generated by mixing CA and CNa3 should be able to catalyze L-LA with sufficient reactivity and selectivity. Following this hypothesis, the synthesis of the inner PCL block was first performed in a [ε-CL]0/[BDM]0/[CA] ratio of 50/1/1 with a small amount of toluene (ε-CL/toluene = 1 mmol/40 µL) at 100 °C. After reacting for 8 h, the conversion of ε-CL reached 84.5%. At this moment, L-LA, CNa3, and a small amount of toluene ([L-LA]0/[BDM]0/[CNa3] = 50/1/5, L-LA/toluene = 1 mmol/40 µL) were added without purification. The reaction mixture was heated to 130 °C for 21 h. A 74.3% conversion of L-LA and 89.8% final conversion of ε-CL were obtained. The SEC trace of the obtained copolymers exhibited a good shift to the high-molecular-weight region while maintaining a narrow dispersity, that is, from Mn, SEC = 9000 g mol−1, Đ = 1.09 (PCL) to Mn, SEC = 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1, Đ = 1.18 (PLA-b-PCL-b-PLA) (Fig. 9a). Comparing the 1H-NMR spectra before and after the chain extension of PLLA, the peak representing the chain-end hydroxyl groups of PCL disappeared and the peak corresponding to PLLA appeared (Fig. 9b). These experimental results demonstrate that using CA and CNa3 as catalysts, PLLA-b-PCL-b-PLLA was successfully synthesized in a one-pot, two-step fashion, albeit with an almost negligible amount of the CL monomer incorporated into the PLLA block (Table S4 and Fig. S21).
000 g mol−1, Đ = 1.18 (PLA-b-PCL-b-PLA) (Fig. 9a). Comparing the 1H-NMR spectra before and after the chain extension of PLLA, the peak representing the chain-end hydroxyl groups of PCL disappeared and the peak corresponding to PLLA appeared (Fig. 9b). These experimental results demonstrate that using CA and CNa3 as catalysts, PLLA-b-PCL-b-PLLA was successfully synthesized in a one-pot, two-step fashion, albeit with an almost negligible amount of the CL monomer incorporated into the PLLA block (Table S4 and Fig. S21).
4. Conclusions
In this study, a series of edible compounds, specifically organic acids and their sodium or potassium salts, were systematically examined as safe catalysts for the ROP of cyclic esters and cyclic carbonates. We confirmed that the appropriate selection of organic acids and alkali metal salt catalysts enabled the synthesis of biodegradable, biocompatible, and bioresorbable polyesters (PLLA, PCL, and PVL) and polycarbonates (PTMC). The obtained polymers exhibited narrow distributions (Đ = 1.1–1.3) and well-defined chain-end structures. Copolymers with different sequence structures can be synthesized using binary catalytic systems comprising a mixture of edible organic acids and sodium salts. Random-like PLLA-stat-PCL was synthesized using a saccharin/sodium-saccharin mixed catalyst system. The PLLA-b-PCL-b-PLLA triblock copolymer was synthesized using a one-pot, two-step method using CA/CNa3. These examples represent innovative approaches that yield high-value-added biodegradable polymers through the simple technique of mixing two types of edible catalysts. Therefore, unassuming edible compounds in our daily lives have the potential to be safe and effective catalysts that replace conventional metal catalysts and organocatalysts, thereby contributing to the practical application of biodegradable polymer materials.Author contributions
Toshiki Miwa: writing – original draft, methodology, visualization, and data curation. Ryota Suzuki: conceptualization, methodology, data curation, and funding acquisition. Tianle Gao: supervision, writing – review and editing, and funding acquisition. Takuya Yamamoto: supervision. Feng Li: supervision, conceptualization, methodology, writing – original draft, writing – review and editing, and funding acquisition. Takuya Isono: conceptualization, supervision, and writing – review and editing. Toshifumi Satoh: supervision, conceptualization, writing – review and editing, and funding acquisition.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5gc05933j.Acknowledgements
This work was financially supported by the Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for Scientific Research (A) (24H00766), Grant-in-Aid for Early-Career Scientists (24K17719), Grant-in-Aid for JSPS Research Fellows, the Frontier Chemistry Center (Hokkaido University), the Photo-Excitonic Project (Hokkaido University), the List Sustainable Digital Transformation Catalyst Collaboration Research Platform (List-PF, Hokkaido University), the Project of Junior Scientist Promotion (Hokkaido University), Co-Creation Core for Soft Materials Aspiring Research & Translation (C3-SMART), the JSPS Program for Forming Japan's Peak Research Universities (J-PEAKS; No. JPJS00420230001) “Soft Materials Platform Aspiring the Unique Properties from Natural Polymers”, and the Japan Food Chemical Research Foundation. R.S. and T.G. gratefully acknowledge the JSPS Fellowship for Young Scientists.References
- M. Vert, S. M. Li, G. Spenlehauer and P. Guerin, J. Mater. Sci.: Mater. Med., 1992, 3, 432–446 CrossRef CAS.
- R. P. Brannigan and A. P. Dove, Biomater. Sci., 2017, 5, 9–21 RSC.
- N. A. Asri, N. A. A. Sezali, H. L. Ong, M. H. M. Pisal, Y. H. Lim and J. Fang, Macromol. Rapid Commun., 2024, 45, 2400475 CrossRef CAS PubMed.
- B. J. O'Keefe, M. A. Hillmyer and W. B. Tolman, J. Chem. Soc., Dalton Trans., 2001, 2215–2224 RSC.
- J. Wu, T.-L. Yu, C.-T. Chen and C.-C. Lin, Coord. Chem. Rev., 2006, 250, 602–626 CrossRef CAS.
- A.-C. Albertsson and R. K. Srivastava, Adv. Drug Delivery Rev., 2008, 60, 1077–1093 CrossRef CAS PubMed.
- J. Xu, X. Wang, J. Liu, X. Feng, Y. Gnanou and N. Hadjichristidis, Prog. Polym. Sci., 2022, 125, 101484 CrossRef CAS.
- F. Nederberg, E. F. Connor, M. Möller, T. Glauser and J. L. Hedrick, Angew. Chem., Int. Ed., 2001, 40, 2712–2715 CrossRef CAS PubMed.
- N. E. Kamber, W. Jeong, R. M. Waymouth, R. C. Pratt, B. G. G. Lohmeijer and J. L. Hedrick, Chem. Rev., 2007, 107, 5813–5840 CrossRef CAS PubMed.
- M. K. Kiesewetter, E. J. Shin, J. L. Hedrick and R. M. Waymouth, Macromolecules, 2010, 43, 2093–2107 CrossRef CAS.
- O. Coulembier, B. Martin-Vaca, D. Bourissou, K. V. Fastnacht, P. P. Datta, M. K. Kiesewetter, S. Naumann, P. Lecomte, C. Jérôme, S. M. Guillaume and K. Fukushima, in Organic Catalysis for Polymerisation, ed. A. Dove, H. Sardon and S. Naumann, Royal Society of Chemistry, Cambridge, 2018, pp. 1–327 Search PubMed.
- K. Fukushima and K. Nozaki, Macromolecules, 2020, 53, 5018–5022 CrossRef CAS.
- F. Li, T. Isono and T. Satoh, in Organocatalysts in Polymer Chemistry: Synthesis and Applications, ed. Z. Li, WILEY-VCH, Weinheim, 2025, pp. 1–43 Search PubMed.
- Y. Xia, J. Shen, H. Alamri, N. Hadjichristidis, J. Zhao, Y. Wang and G. Zhang, Biomacromolecules, 2017, 18, 3233–3237 CrossRef CAS PubMed.
- H. R. Kricheldorf and S. M. Weidner, Polym. Chem., 2022, 13, 1618–1647 RSC.
- J. Casas, P. V. Persson, T. Iversen and A. Córdova, Adv. Synth. Catal., 2004, 346, 1087–1089 CrossRef CAS.
- T. Saito, Y. Aizawa, T. Yamamoto, K. Tajima, T. Isono and T. Satoh, Macromolecules, 2018, 51, 689–696 CrossRef CAS.
- T. Saito, K. Takojima, T. Oyama, S. Hatanaka, T. Konno, T. Yamamoto, K. Tajima, T. Isono and T. Satoh, ACS Sustainable Chem. Eng., 2019, 7, 8868–8875 CrossRef CAS.
- F. Li, R. Suzuki, T. Gao, X. Xia, T. Isono and T. Satoh, Bull. Chem. Soc. Jpn., 2023, 96, 1003–1018 CrossRef CAS.
- Q. Zhang, C. Hu, R. Duan, Y. Huang, X. Li, Z. Sun, X. Pang and X. Chen, Green Chem., 2022, 24, 9282–9289 Search PubMed.
- H. Xie, L. Zheng, J. Feng, X. Wang, S. Kuang, L. Zhou, J. Jiang, Y. Xu, Y. Zhao and Z. Xu, Polym. Chem., 2023, 14, 1630–1638 RSC.
- L. Mezzasalma, J. D. Winter, D. Taton and O. Coulembier, Green Chem., 2018, 20, 5385–5396 RSC.
- L. Mezzasalma, S. Harrisson, S. Saba, P. Loyer, O. Coulembier and D. Taton, Biomacromolecules, 2019, 20, 1965–1974 Search PubMed.
- K. Makiguchi, T. Satoh and T. Kakuchi, Macromolecules, 2011, 44, 1999–2005 CrossRef CAS.
- P. Christ, A. G. Lindsay, S. S. Vormittag, J.-M. Neudörfl, A. Berkessel and A. C. O'Donoghue, Chem. – Eur. J., 2011, 17, 8524–8528 CrossRef CAS PubMed.
- A. Kütt, S. Selberg, I. Kaljurand, S. Tshepelevitsh, A. Heering, A. Darnell, K. Kaupmees, M. Piirsalu and I. Leito, Tetrahedron Lett., 2018, 59, 3738–3748 CrossRef.
- K. Kaur and S. Srivastava, RSC Adv., 2020, 10, 36571–36608 RSC.
- F. Wei, H. Zhu, Z. Li, H. Wang, Y. Zhu, L. Zhang, Z. Yao, Z. Luo, C. Zhang and K. Guo, Adv. Synth. Catal., 2019, 361, 1335–1347 CrossRef CAS.
- K. V. Fastnacht, S. S. Spink, N. U. Dharmaratne, J. U. Pothupitiya, P. P. Datta, E. T. Kiesewetter and M. K. Kiesewetter, ACS Macro Lett., 2016, 5, 982–986 CrossRef CAS PubMed.
- Just before our submission, a study on the ROP of ε-caprolactone and L-lactide using carboxylic acids with mono-, di-, and trifunctionality was reported RSC; J. P. Aldaba-Ramos, M. P. Barrera-Nava, J. B. Cruz, A. Aparicio-Saguilán, A. Ramírez-Hernández and J. E. Báez, RSC Adv., 2025, 15, 43106–43119 RSC.
- T. Saito, Y. Aizawa, K. Tajima, T. Isono and T. Satoh, Polym. Chem., 2015, 6, 4374–4384 RSC.
- W. Limwanich, P. Meepowpan, M. Dumklang, W. Funfuenha, P. Rithchumpon and W. Punyodom, Thermochim. Acta, 2024, 736, 179734 CrossRef CAS.
- K. Takojima, T. Saito, C. Vevert, V. Ladelta, P. Bilalis, J. Watanabe, S. Hatanaka, T. Konno, T. Yamamoto, K. Tajima, N. Hadjichristidis, T. Isono and T. Satoh, Polym. J., 2020, 52, 103–110 CrossRef CAS.
- B. G. G. Lohmeijer, R. C. Pratt, F. Leibfarth, J. W. Logan, D. A. Long, A. P. Dove, F. Nederberg, J. Choi, C. Wade, R. M. Waymouth and J. L. Hedrick, Macromolecules, 2006, 39, 8574–8583 CrossRef CAS.
- M. Baśko and P. Kubisa, J. Polym. Sci., Part A: Polym. Chem., 2006, 44, 7071–7081 CrossRef.
- H. Alamri, J. Zhao, D. Pahovnik and N. Hadjichristidis, Polym. Chem., 2014, 5, 5471–5478 RSC.
- Y. Wang, J. Niu, L. Jiang, Y. Niu and L. Zhang, J. Macromol. Sci., Part A: Pure Appl. Chem., 2016, 53, 374–381 CrossRef CAS.
- E. Stirling, Y. Champouret and M. Visseaux, Polym. Chem., 2018, 9, 2517–2531 RSC.
- N. Nomura, A. Akita, R. Ishii and M. Mizuno, J. Am. Chem. Soc., 2010, 132, 1750–1751 CrossRef CAS PubMed.
- J. Kasperczyk and M. Bero, Makromol. Chem., 1991, 192, 1777–1787 CrossRef CAS.
- J. Kasperczyk and M. Bero, Makromol. Chem., 1993, 194, 913–925 CrossRef CAS.
- M. Jikei, Y. Takeyama, Y. Yamadoi, N. Shinbo, K. Matsumoto, M. Motokawa, K. Ishibashi and F. Yamamoto, Polym. J., 2015, 47, 657–665 CrossRef CAS.
- Z. Yu, C. Zhu, H. Zhang, C. Liu, R. Wei, R. Duan, Y. Liu, X. Bian, H. Piao, W. Wang, K. Liu and X. Chen, Int. J. Biol. Macromol., 2025, 311, 144072 CrossRef CAS PubMed.
- K. Makiguchi, S. Kikuchi, K. Yanai, Y. Ogasawara, S. Sato, T. Satoh and T. Kakuchi, J. Polym. Sci., Part A: Polym. Chem., 2014, 52, 1047–1054 CrossRef CAS.
- X. Wang, J. Liu, S. Xu, J. Xu, X. Pan, J. Liu, S. Cui, Z. Li and K. Guo, Polym. Chem., 2016, 7, 6297–6308 RSC.
- M. D. Deokar, B. Garnaik and S. Sivaram, ACS Omega, 2022, 7, 9118–9129 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |