Open Access Article
Open Access ArticleTurning light into carbinols: a metal-free radical strategy for pyridine based triaryl scaffolds
Matteo
Leone
 * and
Maurizio
Fagnoni
* and
Maurizio
Fagnoni
 *
*
PhotoGreen Lab, Department of Chemistry, University of Pavia, Viale Taramelli 12, 27100 Pavia, Italy. E-mail: maurizio.fagnoni@unipv.it
First published on 18th February 2026
Abstract
Triaryl carbinols are key building blocks in pharmaceuticals, agrochemicals, and functional materials, yet their synthesis typically relies on hazardous organometallic reagents or costly transition-metal catalysts. Here we report a metal-free, light-driven strategy for their preparation under mild and sustainable conditions. The method exploits the dual role of 2-propanol, which acts as a hydrogen donor in the photomediated HAT process with benzophenone, while the resulting ketyl radical selectively reduces 4-cyanopyridines via electron transfer, enabling a radical–radical coupling that furnishes triaryl carbinols in a single step. The protocol delivers excellent yields (up to 99%) across a broad substrate scope, encompassing diverse benzophenones and pyridines. Its robustness was demonstrated in batch, flow, and under simulated sunlight, and the methodology enabled the efficient synthesis of analogues of bioactive molecules of pharmaceutical relevance. Notably, the optimized protocol scores >80 on the EcoScale (indicating excellent sustainability in terms of reagent safety, energy input, and workup efficiency). This work establishes a versatile and greener platform for the synthesis of triaryl carbinols with strong potential for industrial and medicinal applications.
Green foundation1. This work advances green chemistry by introducing a metal-free, visible-light-driven synthesis of triaryl carbinols eliminating the need for hazardous organometallic reagents and costly transition-metal catalysts. The reaction proceeds under mild conditions, achieving excellent atom economy. Its scalability further underscores its practicality and low environmental impact. The preparation of analogues of bioactive compounds shows excellent results providing an efficient and user-friendly method.2. Highly efficient (up to 99% yield) and atom-economical (AE = 93%) synthesis of triaryl carbinols was achieved from inexpensive and readily available starting materials. The protocol employs green solvents and harnesses light as a safe energy source. A high EcoScale score (86) underscores the superior sustainability of this method compared to traditional metal based protocols. 3. Triaryl carbinols obtained, particularly on a large scale, could be purified by simple crystallization, eliminating the need for extensive solvent use. |
Introduction
The pursuit of greener methodologies for the synthesis of chemical products has been a central objective in recent decades.1 In particular, the pharmaceutical and fine chemical sectors aim to reduce their carbon footprint by adopting lower-carbon fuels, improving energy efficiency, and employing catalytic processes.2 Yet, many established transformations still rely heavily on organometallic reagents, which are hazardous, when handled in large quantities, but also raise significant environmental concerns due to the challenges associated with their disposal.3 In this context, open-shell reactions offer valuable opportunities to access new reactivity modes and improve existing methods,4 thereby contributing to more efficient synthesis of fine chemicals. Among these, light-mediated processes have emerged as powerful tools, promoting a wide range of transformations under mild conditions by harnessing photon energy as a green and traceless reagent.5 Over the past decades, the scope of such reactions has expanded beyond academia, demonstrating considerable potential for application on an industrial scale.6 Within this framework, we turned our attention to the synthesis of triaryl carbinols (TACs), a valuable class of compounds widely employed as key building blocks in pharmaceuticals, dyes, and functional materials.7 Recent studies have underscored their broad potential, as TAC derivatives display significant biological activities, including antifungal (e.g. fenarimol),8 anticancer,9 and antiviral properties,10 while also serving as essential intermediates in the preparation of commercially available drugs (Fig. 1a).11 Their synthesis often makes use of benzophenones as reagents and typically relies on a limited number of strategies: (i) Grignard reactions of aryl bromides with benzophenones;12 (ii) metal-mediated reduction of cyanoarenes in the presence of benzophenones;13 and (iii) transition-metal catalyzed addition of arylboronates to benzophenones (Fig. 1b).14 To broaden the structural diversity and enhance the potential biological relevance of TACs, recent efforts have focused on incorporating heteroaromatic rings into their framework.15 The introduction of such motifs is known to significantly influence physicochemical and pharmacological properties, often leading to improved bioactivity.16 Developing an efficient and general strategy for their direct incorporation therefore remains highly desirable. Among the approaches reported for introducing heteroaromatic units,17 heteroaromatic nitriles have emerged as particularly versatile building blocks in modern organic synthesis. Over the past decades, remarkable progress has been achieved in the decyanative cross-coupling of cyanoarenes, enabling the construction of a wide array of C(sp2)–C and C(sp2)–X (X = B, N, P, O, Si, S) bonds.18 In this context, cyanopyridines have found extensive application in photocatalytic transformations, acting as optimal electron acceptors.19 Of special interest is the well-established use of benzophenones as photocatalysts, acting as hydrogen-atom abstractors to generate nucleophilic radicals via C–H bond activation.20 The corresponding benzophenone ketyl radical can then selectively reduce 4-cyanopyridines through a highly favorable proton-coupled electron transfer (PCET)21 process, ultimately enabling efficient radical–radical coupling.22 This reactivity has been largely restricted to couplings between benzylic radicals and cyanopyridines (Fig. 1c). To date, no approaches take advantage of benzophenones as both substrates and photoactive species. Building on these precedents, we envisioned that the intrinsic reactivity of benzophenones and cyanopyridines could be harnessed to develop a light-mediated radical–radical coupling between these two classes of building blocks (Fig. 1d). Surprisingly, no examples have been reported on the selective generation of TAC libraries under photo-mediated conditions. Although radical addition of 4-cyanopyridines to aromatic ketones has been described,23 its broader application has been limited by competing side reactions such as benzopinacol formation24 and undesired addition of competitive nucleophilic radicals in the presence of multiple C–H activation sites, which significantly reduce efficiency and practicality.25 We envisioned that we could overcome these limitations choosing a suitable hydrogen donor capable of fulfilling multiple roles: (i) selective generating the benzophenone ketyl radical by HAT process (ii) reducing the 4-cyanopyridine by PCET or ET and (iii) avoid participating in undesired radical–radical coupling. Consequently, the choice of hydrogen donor plays a crucial role, since it is expected to suppress undesired side processes, thereby enabling selective access to the targeted TACs. This methodology not only would provide straightforward entry to a broad library of TACs but also holds promise for the scalable production of targeted compounds under mild, sustainable conditions, thereby reducing the environmental impact of such transformations. Moreover, the incorporation of pyridines, one of the most prevalent structural motifs in natural products and pharmaceuticals,26 further underscores the synthetic relevance of this approach, possibly generating libraries of medicinally relevant compounds. | ||
| Fig. 1 (a) Bio-activity of various TACs (b) Polar approaches for the synthesis of TACs (c) application of benzophenone (BP) as photocatalyst for 4-pyridination of C(sp3)–H bonds. (d) Our work. | ||
Results and discussion
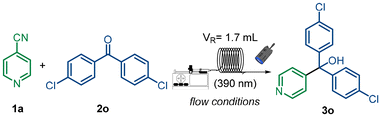
Benzophenone has been widely employed as an efficient photocatalyst in net-reductive transformations.27 In its excited state, it readily abstracts a hydrogen atom from C–H bonds even with high bond dissociation energies (BDEs), generating the corresponding ketyl radical.28 Owing to its oxidation potential (Eox = –0.25 V vs. SCE),29 this ketyl radical is capable of reducing 4-cyanopyridine (Ered = –0.67 V vs. SCE)30 through a proton-coupled electron transfer (PCET) process. This strategy has been successfully exploited in a variety of transformations for the synthesis of substituted pyridines.22 For our purposes, however, the coexistence of both the benzophenone-derived ketyl radical and the reduced cyanopyridine is essential to promote the desired radical–radical coupling. We therefore considered the possibility of generating the reduced cyanopyridine via electron transfer mediated by the radical adduct of the hydrogen donor. An ideal candidate fulfilling these requirements is 2-propanol: it features a relatively low BDE (91 kcal mol−1)31 and can donate a hydrogen atom to excited benzophenone through a HAT process,32 while the resulting α-hydroxyalkyl radical is sufficiently reducing (Eox = –0.61 V vs. SCE)33 to activate the cyanopyridine. Guided by these considerations and our previous experience,34 we initiated optimization studies on the model reaction by screening different conditions (Table 1). Firstly, we screened different candidates as H-donors with low bond dissociation energies (BDEs)31 and whose corresponding radicals are not highly nucleophilic (entries 1 and 2).35 EtOAc was chosen as green media and despite obtaining promising results with γ-terpinene and M3N–BH3, they did not prove to be as efficient as 2-propanol (45% yield entry 3). With 2-propanol identified as the optimal donor, fine-tuning the stoichiometry of 1a, as limiting reagent, and 2a revealed that a slight excess of 2a minimized waste while maintaining excellent reactivity (entry 4). Next, increasing the stochiometric amount of 2-propanol to 10 equivalents afforded the desired product 3a in 90% isolated yield (entry 5). We also evaluated other green solvents,36 including MeCN, H2O, and acetone, which delivered 3a in good to excellent yields (entries 6–8). In contrast, when 2-MeTHF was employed as the solvent, a competing addition onto 1a was observed, resulting in a diminished yield of 3a. Control experiments confirmed that no reactivity occurred in the absence of either light or the hydrogen donor recovering mostly starting materials (entries 10 and 11). To further enhance reaction efficiency, we likewise tested continuous-flow conditions. Compared to batch, flow offers superior heat and mass transfer, precise control over reaction parameters, and uniform light penetration, thereby enabling shorter reaction times, improved selectivity, and greater scalability under energy-efficient conditions.37 With the aim of synthesizing drug analogues bearing chloro substituents, we considered optimizing the flow process using 2o (Table 2). At a flow rate of 1.8 mL h−1, complete conversion of the starting material was achieved, affording 3o in 95% yield (90% in batch entry 2). Remarkably, the reaction could also be performed under more concentrated conditions (0.2 M EtOAc), which required a lower flow rate to ensure full conversion of 1a (entry 4). These conditions not only enhanced the molar productivity but also reduced solvent consumption. With the optimized conditions in hand, we next investigated the radical addition of substituted benzophenones to 4-cyanopyridine (Fig. 2). The substrate scope was primarily examined in batch, with selected cases also evaluated under flow conditions. Guided by the results obtained for 3a and 3o, we first explored mono-substituted methylbenzophenones. para- (2b) and meta-substituted (2c) derivatives afforded the desired products 3b e 3c quantitatively, whereas the ortho analogue 2r yielded only trace amounts, likely due to intramolecular quenching of the carbonyl excited state.25 Comparable reactivity was observed for 3b under flow conditions. We then examined para-substituted benzophenones with different electronic properties. Both electron-rich and electron-poor derivatives provided the corresponding products in good to excellent yields (3d–3f). Benzophenone 2f showed a partial reduction to diaryl methanol, therefore for better reaction performances 2 equivalents of benzophenone were used. We screened also a free carboxylic acid which was remarkably well tolerated, affording 3g in near-quantitative yield. Slightly diminished yields were obtained in flow, presumably due to the formation of insoluble salts in more concentrated reaction medium. Building on this robustness, the ester derivative 2h furnished 3h almost quantitatively. Importantly, the methodology also enabled late-stage functionalization of complex scaffolds, as demonstrated by the successful modification of cholesterol and cedrol derivatives, delivering 3i and 3j up to 70% yield. It is worth noting that, despite the presence of several C–H bonds susceptible to HAT in both substrates, the abstraction process was selectively directed to the chosen H-donor, thereby suppressing undesired side reactions. Next, we investigated bis-substituted benzophenones. The symmetric 4,4′-dimethoxybenzophenone 2k afforded 3k in moderate yield under both batch and flow conditions, whereas unsymmetrical derivatives (2l–2n) gave the desired products (3l–3n) in good to excellent yields. Interestingly, ortho-fluoro substitution slightly reduced reactivity, while 3o was obtained in excellent yield both in batch and flow conditions. Finally, the feasibility of incorporating heteroaromatic scaffolds was demonstrated, with derivative 2p affording 3p in 82% yield. Encouraged by these results, we next investigated the reactivity of benzophenone 2o with differently substituted cyanopyridines (Fig. 3). To the best of our knowledge, no systematic studies have explored the behavior of polysubstituted cyanopyridines in the presence of benzophenones;22 previous reports have largely focused on the addition of nucleophilic radicals to 4-cyanopyridines. We attributed this limited exploration of cyanopyridine derivatization to two main factors: (i) the possible un-matching of redox potential of benzophenones, which may not align with that of substituted cyanopyridines;38 and (ii) variations in spin density across the pyridine scaffold, which could lead to divergent reactivity at different C-positions and, consequently, to unpredictable outcomes.39 We began our study with 2-cyanopyridine, which initially furnished only trace amounts of the desired product 4, accompanied by significant formation of the corresponding diphenylmethanol derived from 2o. This observation suggests that ipso-substitution at C-2 is less favorable than at C-4, rendering 2o more prone to direct reduction.40 Guided by this insight, we undertook reaction optimization (see SI section 4), ultimately isolating the desired product 4 in 45% yield. Notably, the addition of small amounts of water and performing the reaction under mildly acidic conditions significantly promoted the ipso-substitution pathway, by protonation of pyridine, thereby enhancing product formation.41 We then evaluated 2,6-dicyanopyridine, whose electron-deficient core proved more reactive, readily undergoing ET to give the mono-substituted carbinol 5 in 52% yield showing better results using 2 equivalents of 2o. Building on this, we investigated 2,4-dicyanopyridine and we were able to isolate the disubstituted product 6, bearing carbinol units at both C-2 and C-4. Varying the equivalents of 2o (0.5, 1.0, and 2.0 equiv.) consistently furnished 6 as the sole product. A similar outcome was observed with 3-fluoroisonicotinonitrile, which underwent double addition of 2o at C-2 and C-4, affording 7 as the only detectable product. These findings indicate that, while ipso-substitution at C-4 is typically favored, in certain cases radical addition at C-2 can proceed with comparable activation barriers, leading to the formation of doubly substituted pyridines.42 Next, we investigated the reactivity of a pyridine bearing a cyano group at C-2 and a good leaving group at C-4, such as a halide. As anticipated, ipso-substitution occurred preferentially at the para position, affording product 8 in 63% yield. This result highlights the potential to design pyridines with leaving groups beyond cyano, thereby enabling selective radical additions at C-2, C-4, or both positions depending on the substitution pattern. We then turned our attention to ortho-substituted 4-cyanopyridines. Pyridine 1g, bearing an acetophenone moiety, underwent substitution with concurrent reduction of the ketone, mediated by 2o itself.43 Monitoring the reaction by 1H NMR revealed that the addition of 2o was accompanied by concomitant formation of the corresponding benzyl alcohol. Finally, C-2-substituted pyridines bearing either aryl groups or protected amines delivered the desired products in good yields (≈80%), underscoring the versatility of this methodology for diversifying pyridine scaffolds. To further demonstrate the practicality of our methodology, we scaled up the synthesis of 3o to the gram scale using both batch and flow setups (Fig. 4a). In both cases, the reaction proceeded efficiently with no noticeable loss in yield, underscoring the robustness of the strategy and its suitability for scalable synthesis under mild conditions. Notably, compared to previously reported procedures,15 this approach offers significant improvements in atom economy (AE) and Eco scale,44 establishing it as a more sustainable and synthetically efficient alternative to conventional polar methodologies (see SI, section 10). Moreover, the reaction could be carried out under simulated solar irradiation (SolarBOX), affording 3o in good yields (81% yield) and highlighting the potential of sunlight as a sustainable energy source (Fig. 4a).45 We next explored the utility of this approach for the preparation of analogues of pharmaceutically and agrochemically relevant compounds. As a first example, we targeted fenarimol, a widely used fungicide commercialized under various tradenames and mostly applied for the control of powdery mildew.46 Its analogue was obtained in a single step and in excellent yields in up to one millimolar scale, while avoiding the use of highly reactive n-BuLi and cryogenic conditions (−95 °C) required in traditional syntheses (Fig. 4b).47 In a similar vein, we prepared an analogue of clotrimazole, a major antifungal active ingredient used in commercial formulations to treat vaginal yeast infections.48| Entry | 1a (equiv.) | 2a (equiv.) | H-Donor (equiv.) | Solvent (0.1 M) | Yielda |
|---|---|---|---|---|---|
| Reaction conditions: reaction run on 0.2 mmol scale in 0.1 M of solvent under N2. Reactions were irradiated with a 40 W Kessil lamp (390 nm) for 16 h.a Yield determined by GC-FID with the addition of biphenyl as an internal standard.b Isolated yield.c Competitive addition of the solvent onto cyanopyridine.d No light. | |||||
| 1 | 3 | 1 | γ-Terpinene (10) | EtOAc | 16% |
| 2 | 3 | 1 | Me3N-BH3 (2) | EtOAc | 40% |
| 3 | 3 | 1 | 2-Propanol (5) | EtOAc | 45% |
| 4 | 1 | 1.2 | 2-Propanol (5) | EtOAc | 70% |
| 5 | 1 | 1.2 | 2-Propanol (10) | EtOAc | 92% (90%)b |
| 6 | 1 | 1.2 | 2-Propanol (10) | MeCN | 85% |
| 7 | 1 | 1.2 | 2-Propanol (10) | MeCN/H2O 9/1 | 72% |
| 8 | 1 | 1.2 | 2-Propanol (10) | Acetone | 61% |
| 9c | 1 | 1.2 | 2-Propanol (10) | 2-MeTHF | 10% |
| 10d | 1 | 1.2 | 2-Propanol (10) | EtOAc | 0% |
| 11 | 1 | 1.2 | — | EtOAc | 0% |
 | ||
| Fig. 4 (a) Grame scale reaction performed in batch, flow and solar box conditions. (b) Light-mediated synthesis of Fenarimol's analogue (c) light-mediated synthesis of clotrimazole's analogue. | ||
| Entry | Flow rate | Solvent | RTDa (min) | Yieldb | Productivity (mmol d−1) |
|---|---|---|---|---|---|
| Reaction conditions: 1a (0.2 mmol 1.0 equiv.), 2o (0.24 mmol, 1.2 equiv.), 2-propanol (2 mmol, 10 equiv.), EtOAc as solvent. Reactions were irradiated with two 40 W Kessil lamp (390 nm).a Ritention time distribution.b Yield determined by GC-FID with the addition of biphenyl as an internal standard.c Isolated yield for batch conditions.d Isolated yield. | |||||
| 1 | 2 mL h−1 | EtOAc (0.1 M) | 51 | 80% | 3.8 |
| 2 | 1.8 mL h−1 | EtOAc (0.1 M) | 56 | 95% (90%)c | 4.1 |
| 3 | 1.5 mL h−1 | EtOAc (0.2 M) | 68 | 90% | 6.5 |
| 4 | 1.2 mL h−1 | EtOAc (0.2 M) | 85 | 99% (98%)d | 5.8 |
In 2016, Canesten-brand clotrimazole was among the top-selling over-the-counter medications in Great Britain, with sales of £39.2 million.49 Classical routes to clotrimazole involve the synthesis of the triaryl carbinol intermediate, typically employing organolithium reagents, followed by chlorination with SOCl2 and subsequent nucleophilic substitution with imidazole.50 In contrast, our strategy provided the target analogue under significantly milder conditions, delivering an overall yield above 80% over two steps on both small and millimolar scale (Fig. 4c).
Finally, to gain mechanistic insight, we carried out the reaction in the presence of TEMPO as a radical scavenger. The reaction was completely suppressed, and the formation of a TEMPO-2-propanol adduct was observed by LC-MS analysis, consistent with the involvement of a radical pathway (see SI). We then sought to investigate the fate of the ketyl radical generated from 2-propanol. To this end, the reaction was performed under the general conditions using CD3CN as solvent, enabling direct NMR analysis of the crude mixture. This revealed the formation of acetone, supporting a possible electron transfer (ET) event between the cyanopyridine and the ketyl radical derived from 2-propanol. Further insight into the reaction pathway was obtained by determining the quantum yield for the consumption of 1a, which gave ϕ = 1.71. This value indicates that both the benzophenone- and 2-propanol-derived ketyl radicals contribute to the reduction of cyanopyridine.
Therefore, based on these observations and previous reports, we propose the following mechanism (Fig. 5). Upon irradiation, benzophenone (2) is excited to its triplet state (2*), it abstracts a hydrogen atom from 2-propanol (II).31 This process generates the corresponding ketyl radicals (I and III), which are capable of reducing cyanopyridine 1 to the persistent radical (V).51 The coexistence of radicals I and V in solution facilitates radical–radical coupling, and subsequent extrusion of HCN furnishes the triaryl carbinol (3) from intermediate VI. In addition, 2-propanol may play an auxiliary role, as it has been reported, to directly reduce benzophenone (2) to ketyl radical (I), although the associated rate constant (k = 7 × 104 M−1 s−1) is relatively low.33a
Conclusions
In summary, we have developed a metal-free, light-mediated strategy for the synthesis of triaryl carbinols from benzophenones and cyanopyridines under mild and sustainable conditions. This approach avoids the use of hazardous organometallic reagents and costly transition-metal catalysts, while operating efficiently in green solvents. Central to the reactivity is the dual role of 2-propanol: as H-donor in a HAT process with benzophenone, while its corresponding radical reduces cyanopyridines, thus enabling the key radical–radical coupling that affords the target products.The methodology grants straightforward access to a broad library of triaryl carbinols, and its robustness was demonstrated across different operational modes, including batch, flow, and gram-scale syntheses, as well as under solar-simulated irradiation. Furthermore, we showcased its applicability to the preparation of analogues of bioactive compounds of pharmaceutical and agrochemical relevance, highlighting its potential as a practical alternative to classical methods that rely on highly reactive organolithium reagents and harsh conditions.
Overall, this work establishes a versatile and scalable platform for the synthesis of pyridine based triaryl carbinols, with significant promise for future applications in industrial settings and medicinal chemistry.
Author contributions
This work was conceptualized by M. L., experimentation was performed by M. L. The first draft of the manuscript was prepared by M. L. and the final version was edited and revised by M. L. and M. F.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5gc05657h.Acknowledgements
We acknowledge support from UniPV and MUR through the program ‘Departments of Excellence’ (2023-2027). We thank Elisa Girardi for preliminary experiments.References
- (a) P. T. Anastas, J. C. Warner and J. C. Warner, Green Chemistry: Theory and Practice, Oxford University Press, Oxford, 2000. 1. paperback CrossRef; (b) Encyclopedia of Green Chemistry, ed. B.Török, Elsevier, Amsterdam Boston Heidelberg London New York Oxford Paris San Diego San Francisco Singapore Sydney Tokyo, 2025 Search PubMed.
- (a) R. S. Treptow, J. Chem. Educ., 2010, 87, 168–171 Search PubMed; (b) D.-S. Chang, L.-T. Yeh and W. Liu, Clean Technol. Environ. Policy, 2015, 17, 359–371 Search PubMed; (c) J. Rissman, C. Bataille, E. Masanet, N. Aden, W. R. Morrow, N. Zhou, N. Elliott, R. Dell, N. Heeren, B. Huckestein, J. Cresko, S. A. Miller, J. Roy, P. Fennell, B. Cremmins, T. Koch Blank, D. Hone, E. D. Williams, S. De La Rue Du Can, B. Sisson, M. Williams, J. Katzenberger, D. Burtraw, G. Sethi, H. Ping, D. Danielson, H. Lu, T. Lorber, J. Dinkel and J. Helseth, Appl. Energy, 2020, 266, 114848 CrossRef CAS; (d) A. Estevez-Torres, F. Gauffre, G. Gouget, C. Grazon and P. Loubet, Green Chem., 2024, 26, 2613–2622 Search PubMed.
- (a) E. Dopp, L. M. Hartmann, A.-M. Florea, A. W. Rettenmeier and A. V. Hirner, Crit. Rev. Toxicol., 2004, 34, 301–333 CrossRef CAS PubMed; (b) A. Majumdar, M. K. Upadhyay, M. Ojha, R. Biswas, S. Dey, S. Sarkar, D. Moulick, N. K. Niazi, J. Rinklebe, J.-H. Huang and T. Roychowdhury, Sci. Total Environ., 2024, 951, 175531 CrossRef CAS PubMed.
- (a) B. Halliwell and J. M. C. Gutteridge, Free Radicals in Biology and Medicine, Oxford University Press, 2015 CrossRef; (b) S. Z. Zard, Radical Reactions in Organic Synthesis, Oxford University PressOxford, 2003 CrossRef; (c) A. Studer, Adv. Synth. Catal., 2020, 362, 2074 Search PubMed; (d) Z. X. Chen, Y. Li and F. Huang, Chem, 2021, 7, 288–332 CrossRef CAS; (e) D. P. Curran, B. Giese and N. A. Porter, Stereochemistry of radical reactions: concepts, guidelines, and synthetic applications, VCH, Weinheim New York, 1996 Search PubMed; (f) M. Inoue, L. R. Malins and H. Renata, Org. Lett., 2024, 26, 7775–7777 CrossRef CAS PubMed; (g) V. T. Perchyonok, I. N. Lykakis and K. L. Tuck, Green Chem., 2008, 10, 153–163 RSC.
- (a) S. Crespi and M. Fagnoni, Chem. Rev., 2020, 120, 9790–9833 CrossRef CAS PubMed; (b) M. D. Kärkäs, B. S. Matsuura and C. R. J. Stephenson, Science, 2015, 349, 1285–1286 CrossRef PubMed; (c) D. Ravelli, S. Protti and M. Fagnoni, Acc. Chem. Res., 2016, 49, 2232–2242 CrossRef CAS PubMed; (d) C. K. Prier, D. A. Rankic and D. W. C. MacMillan, Chem. Rev., 2013, 113, 5322–5363 CrossRef CAS PubMed; (e) C. Michelin and N. Hoffmann, Curr. Opin. Green Sustainable Chem., 2018, 10, 40–45 CrossRef; (f) A. Quintavalla, D. Carboni and M. Lombardo, ChemCatChem, 2024, 16, e202301225 CrossRef CAS.
- (a) L. Candish, K. D. Collins, G. C. Cook, J. J. Douglas, A. Gómez-Suárez, A. Jolit and S. Keess, Chem. Rev., 2022, 122, 2907–2980 CrossRef CAS PubMed; (b) A. A. Adesina, Catal. Surv. Asia, 2004, 8, 265–273 CrossRef CAS; (c) M. Devi, S. Praharaj and D. Rout, in Nanostructured Materials for Visible Light Photocatalysis, Elsevier, 2022, pp. 535–567 Search PubMed; (d) D. Lahiri, M. Nag and R. R. Ray, in Photocatalytic Degradation of Dyes, Elsevier, 2021, pp. 467–488 Search PubMed; (e) P. Li, J. A. Terrett and J. R. Zbieg, ACS Med. Chem. Lett., 2020, 11, 2120–2130 CrossRef CAS PubMed.
- (a) S. Mondal and G. Panda, RSC Adv., 2014, 4, 28317–28358 RSC; (b) S. K. Sen Gupta and S. Mishra, J. Phys. Chem. A, 2011, 115, 4616–4623 CrossRef CAS PubMed; (c) S. Alhamed, J. Alnakhli, W. Boadi and R. Beni, Open J. Med. Chem., 2019, 09, 49–62 CAS; (d) J. De Boer, in Reviews of Environmental Contamination and Toxicology, ed. G. W. Ware, Springer New York, New York, NY, 1997, vol. 150, pp. 95–106 Search PubMed; (e) R. Beni, W. Boadi, K. Karim, J. Alnakhli and S. Alhamed, Open J. Med. Chem., 2019, 09, 37–47 CAS; (f) E. Kelderman, W. A. J. Starmans, J. P. M. Van Duynhoven, W. Verboom, J. F. J. Engbersen, D. N. Reinhoudt, L. Derhaeg, T. Verbiest, K. Clays and A. Persoons, Chem. Mater., 1994, 6, 412–417 CrossRef CAS; (g) K. G. Rutherford, J. L. H. Batiste and J. M. Prokipcak, Can. J. Chem., 1969, 47, 4073–4076 CrossRef CAS; (h) A. Bacchi, E. Bosetti, M. Carcelli, P. Pelagatti, G. Pelizzi and D. Rogolino, CrystEngComm, 2006, 8, 233 RSC; (i) Y. Gong and F. F. Mulks, Synthesis, 2025, 1542–1550 CAS.
- K. Oh, T. Matsumoto, A. Yamagami, T. Hoshi, T. Nakano and Y. Yoshizawa, Int. J. Mol. Sci., 2015, 16, 17273–17288 CrossRef CAS PubMed.
- M. R. Saberi, T. K. Vinh, S. W. Yee, B. J. N. Griffiths, P. J. Evans and C. Simons, J. Med. Chem., 2006, 49, 1016–1022 CrossRef CAS PubMed.
- S. Gemma, G. Campiani, S. Butini, G. Kukreja, B. P. Joshi, M. Persico, B. Catalanotti, E. Novellino, E. Fattorusso, V. Nacci, L. Savini, D. Taramelli, N. Basilico, G. Morace, V. Yardley and C. Fattorusso, J. Med. Chem., 2007, 50, 595–598 CrossRef CAS PubMed.
- (a) G. A. McNaughton-Smith, J. F. Burns, J. W. Stocker, G. C. Rigdon, C. Creech, S. Arrington, T. Shelton and L. De Franceschi, J. Med. Chem., 2008, 51, 976–982 CrossRef CAS PubMed; (b) M. Keenan, M. J. Abbott, P. W. Alexander, T. Armstrong, W. M. Best, B. Berven, A. Botero, J. H. Chaplin, S. A. Charman, E. Chatelain, T. W. Von Geldern, M. Kerfoot, A. Khong, T. Nguyen, J. D. McManus, J. Morizzi, E. Ryan, I. Scandale, R. A. Thompson, S. Z. Wang and K. L. White, J. Med. Chem., 2012, 55, 4189–4204 CrossRef CAS PubMed; (c) E. Zeiman, C. L. Greenblatt, S. Elgavish, I. Khozin-Goldberg and J. Golenser, J. Parasitol., 2008, 94, 280–286 CrossRef CAS PubMed.
- (a) J. E. Baldwin, R. M. Adlington, J. C. Bottaro, J. N. Kolhe, I. M. Newington and M. W. D. Perry, Tetrahedron, 1986, 42, 4235–4246 CrossRef CAS; (b) G. Cravotto, A. Procopio, M. Oliverio, L. Orio and D. Carnaroglio, Green Chem., 2011, 13, 2806 RSC; (c) M. Singh, J. T. Schott, M. A. Leon, R. T. Granata, H. K. Dhah, J. A. Welles, M. A. Boyce, H. S. Oseni-Olalemi, C. E. Mordaunt, A. J. Vargas, N. V. Patel and S. Maitra, Bioorg. Med. Chem. Lett., 2012, 22, 6252–6255 Search PubMed; (d) Y. Wen, G. Chen, S. Huang, Y. Tang, J. Yang and Y. Zhang, Adv. Synth. Catal., 2016, 358, 947–957 CrossRef CAS; (e) A. F. Quivelli, G. D'Addato, P. Vitale, J. García-Álvarez, F. M. Perna and V. Capriati, Tetrahedron, 2021, 81, 131898 CrossRef CAS.
- (a) J. P. Wibaut and L. G. Heeringa, Recl. Trav. Chim. Pays-Bas, 1955, 74, 1003–1020 CrossRef CAS; (b) D. Seyferth, W.-L. Wang and R. C. Hui, Tetrahedron Lett., 1984, 25, 1651–1654 CrossRef CAS; (c) D. Seyferth, R. C. Hui and W. L. Wang, J. Org. Chem., 1993, 58, 5843–5845 CrossRef CAS; (d) J. A. D. Good, F. Wang, O. Rath, H. Y. K. Kaan, S. K. Talapatra, D. Podgórski, S. P. MacKay and F. Kozielski, J. Med. Chem., 2013, 56, 1878–1893 CrossRef CAS PubMed; (e) X. Zeng, J. Cai and Y. Gu, Tetrahedron Lett., 1995, 36, 7275–7276 Search PubMed; (f) M. Watanabe, M. Date, K. Kawanishi, M. Tsukazaki and S. Furukawa, Chem. Pharm. Bull., 1989, 37, 2564–2566 CrossRef CAS.
- (a) T. Maekawa, H. Sekizawa and K. Itami, Angew. Chem., Int. Ed., 2011, 50, 7022–7026 CrossRef CAS PubMed; (b) C. Berini and O. Navarro, Chem. Commun., 2012, 48, 1538–1540 RSC; (c) Y.-X. Liao, C.-H. Xing and Q.-S. Hu, Org. Lett., 2012, 14, 1544–1547 CrossRef CAS PubMed; (d) R. Akiyama, M. Sugaya, H. Shinozaki and T. Yamamoto, Synth. Commun., 2019, 49, 1193–1201 CrossRef CAS; (e) Y. Okuda, M. Nagaoka and T. Yamamoto, ChemCatChem, 2020, 12, 6291–6300 CrossRef CAS; (f) J. Huang, X. Yan and Y. Xia, Angew. Chem., Int. Ed., 2022, 61, e202211080 CrossRef CAS PubMed; (g) D. Lichte, N. Pirkl, G. Heinrich, S. Dutta, J. F. Goebel, D. Koley and L. J. Gooßen, Angew. Chem., Int. Ed., 2022, 61, e202210009 CrossRef CAS PubMed; (h) S. Wu, X. Ye, Z. Wang and S. Shi, ChemCatChem, 2024, 16, e202400633 CrossRef CAS.
- C. D. Jones, M. A. Winter, K. S. Hirsch, N. Stamm, H. M. Taylor, H. E. Holden, J. D. Davenport, E. V. Krumkalns and R. G. Suhr, J. Med. Chem., 1990, 33, 416–429 CrossRef CAS PubMed.
- (a) E. Kabir and M. Uzzaman, Results Chem., 2022, 4, 100606 CrossRef CAS; (b) P. Ertl, S. Jelfs, J. Mühlbacher, A. Schuffenhauer and P. Selzer, J. Med. Chem., 2006, 49, 4568–4573 CrossRef CAS PubMed; (c) H. V. Bossche, J. Steroid Biochem. Mol. Biol., 1992, 43, 1003–1021 CrossRef PubMed; (d) L. Banting, in Progress in Medicinal Chemistry, Elsevier, 1996, vol. 33, pp. 147–184 Search PubMed; (e) I. A. S. De Borba, J. B. Peripolli, A. R. Joaquim and F. Fumagalli, Organics, 2025, 6, 23 CrossRef CAS.
- (a) F. Minisci, R. Bernardi, F. Bertini, R. Galli and M. Perchinummo, Tetrahedron, 1971, 27, 3575–3579 CrossRef CAS; (b) C. Friedel and J. M. Crafts, C. R. Chim., 1877, 84, 1392 Search PubMed; (c) N. Miyaura, K. Yamada and A. Suzuki, Tetrahedron Lett., 1979, 20, 3437–3440 CrossRef; (d) H. Nakahara, R. Shirai, Y. Nishimoto, D. Yokogawa and J. Yamaguchi, Nat. Commun., 2025, 16, 8998 CrossRef CAS PubMed.
- (a) S. Tong, K. Li, X. Ouyang, R. Song and J. Li, Green Synth. Catal., 2021, 2, 145–155 Search PubMed; (b) R. Y. Peshkov, E. V. Panteleeva, W. Chunyan, E. V. Tretyakov and V. D. Shteingarts, Beilstein J. Org. Chem., 2016, 12, 1577–1584 CrossRef CAS PubMed; (c) C. Berini and O. Navarro, Chem. Commun., 2012, 48, 1538–1540 RSC; (d) Y.-Y. Jiang, H.-Z. Yu and Y. Fu, Organometallics, 2013, 32, 926–936 CrossRef CAS; (e) E. C. Keske, T. H. West and G. C. Lloyd-Jones, ACS Catal., 2018, 8, 8932–8940 Search PubMed; (f) J.-S. Zhang, T. Chen, Y. Zhou, S.-F. Yin and L.-B. Han, Org. Lett., 2018, 20, 6746–6749 CrossRef CAS PubMed.
- (a) A. McNally, C. K. Prier and D. W. C. MacMillan, Science, 2011, 334, 1114–1117 CrossRef CAS PubMed; (b) Z. Zuo, D. T. Ahneman, L. Chu, J. A. Terrett, A. G. Doyle and D. W. C. MacMillan, Science, 2014, 345, 437–440 CrossRef CAS PubMed; (c) D. Liu, C. Liu, H. Li and A. W. Lei, Angew. Chem., Int. Ed., 2013, 52, 4453–4456 CrossRef CAS PubMed; (d) C. Yan, L. Li, Y. Liu and Q. Wang, Org. Lett., 2016, 18, 4686–4689 CrossRef CAS PubMed; (e) M. M. Walker, B. Koronkiewicz, S. Chen, K. N. Houk, J. M. Mayer and J. A. Ellman, J. Am. Chem. Soc., 2020, 142, 8194–8202 CrossRef CAS PubMed; (f) L.-J. Zhong, H.-Y. Wang, X.-H. Ouyang, J.-H. Li and D.-L. An, Chem. Commun., 2020, 56, 8671–8674 RSC; (g) G. Wang, J. Cao, L. Gao, W. Chen, W. Huang, X. Cheng and S. Li, J. Am. Chem. Soc., 2017, 139, 3904–3910 CrossRef CAS PubMed.
- (a) A. Beckett and G. Porter, Trans. Faraday Soc., 1963, 59, 2038–2050 RSC; (b) L. Capaldo, D. Ravelli and M. Fagnoni, Chem. Rev., 2022, 122, 1875–1924 CrossRef CAS PubMed; (c) A. M. González-Cameno, M. Mella, M. Fagnoni and A. Albini, J. Org. Chem., 2000, 65, 297–303 CrossRef PubMed; (d) G. Campari, M. Fagnoni, M. Mella and A. Albini, Tetrahedron: Asymmetry, 2000, 11, 1891–1906 CrossRef CAS; (e) I. K. Sideri, E. Voutyritsa and C. G. Kokotos, Org. Biomol. Chem., 2018, 16, 4596–4614 RSC; (f) A. Pulcinella, S. Bonciolini, F. Lukas, A. Sorato and T. Noël, Angew. Chem., Int. Ed., 2023, 62, e202215374 CrossRef CAS PubMed.
- (a) E. C. Gentry and R. R. Knowles, Acc. Chem. Res., 2016, 49, 1546–1556 CrossRef CAS PubMed; (b) R. Tyburski, T. Liu, S. D. Glover and L. Hammarström, J. Am. Chem. Soc., 2021, 143, 560–576 CrossRef CAS PubMed; (c) M. H. V. Huynh and T. J. Meyer, Chem. Rev., 2007, 107, 5004–5064 CrossRef CAS PubMed; (d) D. Shukla, W. G. Ahearn and S. Farid, Photochem. Photobiol., 2006, 82, 146 CrossRef CAS PubMed; (e) R. I. Cukier and D. G. Nocera, Annu. Rev. Phys. Chem., 1998, 49, 337–369 CrossRef CAS PubMed.
- (a) T. Hoshikawa and M. Inoue, Chem. Sci., 2013, 4, 3118 RSC; (b) J. Xu, Y. Zhou and B. Liu, J. Org. Chem., 2024, 89, 15877–15883 CrossRef CAS PubMed; (c) J. Xu and B. Liu, Chem. – Eur. J., 2024, 30, e202400612 CrossRef CAS PubMed; (d) A. Bisoyi, A. Behera, A. R. Tripathy, V. K. Simhadri and V. R. Yatham, J. Org. Chem., 2024, 89, 17818–17823 CrossRef CAS PubMed; (e) Y. Zhang and B. Liu, Synlett, 2025, 1064–1068 Search PubMed.
- B. M. Vittimberga, F. Minisci and S. Morrocchi, J. Am. Chem. Soc., 1975, 97, 4397–4398 CrossRef CAS.
- W. E. Bachmann, Org. Synth., 1934, 14, 8 CrossRef CAS.
- R. Bernardi, T. Caronna, S. Morrocchi and M. Ursini, J. Heterocycl. Chem., 1996, 33, 1137–1142 CrossRef CAS.
- (a) C. M. Marshall, J. G. Federice, C. N. Bell, P. B. Cox and J. T. Njardarson, J. Med. Chem., 2024, 67, 11622–11655 CrossRef CAS PubMed; (b) S. De, A. Kumar, S. K. Ashok Kumar, S. K. Shah, S. Kazi, N. Sarkar, S. Banerjee and S. Dey, RSC Adv., 2022, 12, 15385–15406 RSC; (c) A. R. Dwivedi, S. Jaiswal, D. Kukkar, R. Kumar, T. G. Singh, M. P. Singh, A. M. Gaidhane, S. Lakhanpal, K. N. Prasad and B. Kumar, RSC Med. Chem., 2025, 16, 12–36 RSC.
- (a) R. Mosca, M. Fagnoni, M. Mella and A. Albini, Tetrahedron, 2001, 57, 10319–10328 CrossRef CAS; (b) S. Wang, P. S. Singh and D. H. Evans, J. Phys. Chem. C, 2009, 113, 16686–16693 CrossRef CAS; (c) P. R. D. Murray, J. H. Cox, N. D. Chiappini, C. B. Roos, E. A. McLoughlin, B. G. Hejna, S. T. Nguyen, H. H. Ripberger, J. M. Ganley, E. Tsui, N. Y. Shin, B. Koronkiewicz, G. Qiu and R. R. Knowles, Chem. Rev., 2022, 122, 2017–2291 CrossRef CAS PubMed; (d) A. Luridiana, D. Mazzarella, L. Capaldo, J. A. Rincón, P. García-Losada, C. Mateos, M. O. Frederick, M. Nuño, W. J. Buma and T. Noël, ACS Catal., 2022, 12, 11216–11225 CrossRef CAS PubMed; (e) J. Lyu, M. Leone, A. Claraz, C. Allain, L. Neuville and G. Masson, RSC Adv., 2021, 11, 36663–36669 RSC.
- A. M. Cardarelli, M. Fagnoni, M. Mella and A. Albini, J. Org. Chem., 2001, 66, 7320–7327 CrossRef CAS PubMed.
- P. Neta and L. K. Patterson, J. Phys. Chem., 1974, 78, 2211–2217 CrossRef CAS.
- U. Bruehlmann and E. Hayon, J. Am. Chem. Soc., 1974, 96, 6169–6175 CrossRef CAS PubMed.
- Y.-R. Luo, Comprehensive Handbook of Chemical Bond Energies, 1st edn, CRC Press, 2007 Search PubMed.
- G. O. Schenck, G. Koltzenburg and H. Grossmann, Angew. Chem., 1957, 69, 177–178 CrossRef CAS.
- (a) T. Lund, D. D. M. Wayner, M. Jonsson, A. G. Larsen and K. Daasbjerg, J. Am. Chem. Soc., 2001, 123, 12590–12595 CrossRef CAS PubMed; (b) J. Lilie, G. Beck and A. Henglein, Ber. Bunsenges. Phys. Chem., 1971, 75, 458–465 CrossRef CAS.
- (a) M. Leone, D. Arnaldi and M. Fagnoni, Org. Chem. Front., 2025, 12, 4970–4979 RSC; (b) C. Manfrotto, M. Mella, M. Freccero, M. Fagnoni and A. Albini, J. Org. Chem., 1999, 64, 5024–5028 CrossRef CAS PubMed.
- (a) W. Tantawy and H. Zipse, Eur. J. Org. Chem., 2007, 2007, 5817–5820 CrossRef; (b) D. J. Van Hoomissen and S. Vyas, J. Org. Chem., 2017, 82, 5731–5742 CrossRef CAS PubMed; (c) Y. Fu, Z. Wang, Y. Zhang, G. Shen and X. Zhu, ChemistrySelect, 2022, 7, e202202625 CrossRef CAS; (d) A. Gansäuer, L. Shi, M. Otte, I. Huth, A. Rosales, I. Sancho-Sanz, N. M. Padial and J. E. Oltra, in Radicals in Synthesis III, ed. M. Heinrich and A. Gansäuer, Springer Berlin Heidelberg, Berlin, Heidelberg, 2011, vol. 320, pp. 93–120 Search PubMed; (e) M. Salamone, M. Galeotti, E. Romero-Montalvo, J. A. Van Santen, B. D. Groff, J. M. Mayer, G. A. DiLabio and M. Bietti, J. Am. Chem. Soc., 2021, 143, 11759–11776 CrossRef CAS PubMed.
- F. P. Byrne, S. Jin, G. Paggiola, T. H. M. Petchey, J. H. Clark, T. J. Farmer, A. J. Hunt, C. R. McElroy and J. Sherwood, Sustainable Chem. Processes, 2016, 4, 7 CrossRef.
- (a) L. Buglioni, F. Raymenants, A. Slattery, S. D. A. Zondag and T. Noël, Chem. Rev., 2022, 122, 2752–2906 CrossRef CAS PubMed; (b) T. H. Rehm, Chem. – Eur. J., 2020, 26, 16952–16974 CrossRef CAS PubMed; (c) A. I. Alfano, S. Pelliccia, G. Rossino, O. Chianese, V. Summa, S. Collina and M. Brindisi, ACS Med. Chem. Lett., 2023, 14, 672–680 CrossRef CAS PubMed; (d) L. Capaldo, Z. Wen and T. Noël, Chem. Sci., 2023, 14, 4230–4247 RSC.
- (a) W. J. Leigh, D. R. Arnold, R. W. R. Humphreys and P. C. Wong, Can. J. Chem., 1980, 58, 2537–2549 CrossRef CAS; (b) A. Dewanji, P. E. Krach and M. Rueping, Angew. Chem., Int. Ed., 2019, 58, 3566–3570 CrossRef CAS PubMed.
- (a) E. Le Saux, E. Georgiou, I. A. Dmitriev, W. C. Hartley and P. Melchiorre, J. Am. Chem. Soc., 2023, 145, 47–52 CrossRef CAS PubMed; (b) J. Zhang, G. Wu, Y. Zhao, X. Liu, H. Zhu, A. Studer, X. Qi and Q. Cheng, Nat. Synth, 2026, 5, 27–35 CrossRef CAS; (c) E. Georgiou, D. Spinnato, K. Chen, P. Melchiorre and K. Muñiz, Chem. Sci., 2022, 13, 8060–8064 RSC.
- (a) W. D. Cohen and J. Böeseken, Recl. Trav. Chim. Pays-Bas, 1920, 39, 243–279 CrossRef CAS; (b) N. Filipescu and F. L. Minn, J. Am. Chem. Soc., 1968, 90(6), 1544–1547 Search PubMed.
- T. Caronna, S. Morrocchi and B. M. Vittimberga, J. Heterocycl. Chem., 1980, 17, 399–400 Search PubMed.
- (a) L. Gao, G. Wang, J. Cao, H. Chen, Y. Gu, X. Liu, X. Cheng, J. Ma and S. Li, ACS Catal., 2019, 9, 10142–10151 Search PubMed; (b) E. Pu, Y. Zhu, S. Yang, P. Jiang, X. Li, H. Zhang and J. Chen, Org. Lett., 2025, 27, 10855–10861 CrossRef CAS PubMed.
- (a) S. Wang and B. König, Angew. Chem., Int. Ed., 2021, 60, 21624–21634 CrossRef CAS PubMed; (b) C. Boeke, A. Mellott, G. Lahiri, H. Y. K. Huang and J. S. Cannon, Tetrahedron Lett., 2024, 152, 155334 CrossRef CAS PubMed; (c) B. A. Sandoval, S. I. Kurtoic, M. M. Chung, K. F. Biegasiewicz and T. K. Hyster, Angew. Chem., Int. Ed., 2019, 58, 8714–8718 CrossRef CAS PubMed.
- K. Van Aken, L. Strekowski and L. Patiny, Beilstein J. Org. Chem., 2006, 2, 3 Search PubMed.
- (a) S. Protti and M. Fagnoni, Photochem. Photobiol. Sci., 2009, 8, 1499–1516 Search PubMed; (b) S. Choudhury, J.-O. Baeg, N.-J. Park and R. K. Yadav, Green Chem., 2014, 16, 4389 RSC; (c) S. Protti, D. Ravelli, M. Fagnoni and A. Albini, Chem. Commun., 2009, 7351 Search PubMed.
- K. A. Lewis, J. Tzilivakis, D. J. Warner and A. Green, Hum. Ecol. Risk Assess., 2016, 22, 1050–1064 Search PubMed.
- H. M. Taylor, C. D. Jones, J. D. Davenport, K. S. Hirsch, T. J. Kress and D. Weaver, J. Med. Chem., 1987, 30, 1359–1365 CrossRef CAS PubMed.
- R. J. Holt, Drugs, 1975, 9, 401–405 CrossRef CAS PubMed.
- D. Connelly, Pharm. J., 2017, 298, 7900 Search PubMed.
- J. Bartrolí, M. Algueró, E. Boncompte and J. Forn, Arzneimittelforschung, 1992, 42, 832–835 Search PubMed.
- D. Leifert and A. Studer, Angew. Chem., Int. Ed., 2020, 59, 74–108 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |




![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
: