Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Scalable mechanochemical synthesis of amides using bead milling technology
Pietro
Caboni
 ab,
Andrea
Porcheddu
ab,
Andrea
Porcheddu
 b,
Sándor B.
Ötvös
b,
Sándor B.
Ötvös
 *ac and
C. Oliver
Kappe
*ac and
C. Oliver
Kappe
 *ac
*ac
aInstitute of Chemistry, University of Graz, NAWI Graz, Heinrichstrasse 28, A-8010 Graz, Austria. E-mail: sandor.oetvoes@rcpe.at; oliver.kappe@uni-graz.at
bDipartimento di Scienze Chimiche e Geologiche, Università degli Studi di Cagliari, Cittadella Universitaria Monserrato, 09042 Cagliari, Italy
cCenter for Continuous Flow Synthesis and Processing (CCFLOW), Research Center Pharmaceutical Engineering GmbH (RCPE), Inffeldgasse 13, A-8010 Graz, Austria
First published on 7th January 2026
Abstract
Amide bonds are among the most ubiquitous linkages in pharmaceuticals, agrochemicals, and materials, yet their synthesis is still dominated by solvent-intensive protocols. Mechanochemistry offers a sustainable alternative, but its scalability has remained a critical challenge. Here we demonstrate that amide bond formation can be translated into an industrially relevant process using a standard agitator bead mill. Through systematic optimization, we achieved efficient amidations over a broad substrate scope under liquid-assisted grinding with only minimal ethyl acetate. A 20-fold scale-up delivered productivities of up to 2.89 kg h−1, without excess reagents, added base, or bulk solvent, substantially reducing waste. The use of commercially available equipment that is available from lab to manufacturing-scale establishes bead milling as a practical, and environmentally responsible platform for scalable amide synthesis.
Green foundation1. Our work advances green chemistry by establishing bead milling as a sustainable platform for mechanochemical amide bond formation offering high efficiency and straightforward scalability.2. Key green chemistry advantages include: (i) efficient reactions without the need for excess reagents or added base; (ii) short reaction times under liquid-assisted grinding conditions using minimal amounts of ethyl acetate, an environmentally benign additive; (iii) high-yielding amidations across a broad substrate scope (isolated yields up to 96%); (iv) successful scale-up achieving productivities of up to 2.89 kg h−1; (v) product isolation without the use of organic solvents, relying solely on water for purification; and (vi) minimal waste generation (E-factors as low as 1.5 and process mass intensities as low as 2.5). 3. Further research will focus on exploiting larger-scale bead milling reactors to facilitate continuous mechanochemical amide synthesis with on-demand batch sizes suitable for sustainable pharmaceutical manufacturing. |
Introduction
Amide bonds are vital structural motifs due to their exceptional stability and bioavailability, making them essential in both natural and synthetic biologically active molecules, particularly in pharmaceuticals.1 Despite their central role in synthesis, conventional strategies for amide bond formation typically depend on hazardous solvents, large quantities of toxic coupling reagents, and additional bases or auxiliaries.2 As a result, amidation remains one of the most environmentally burdensome transformations in organic chemistry. Among these drawbacks, the reliance on problematic solvents such as N,N-dimethylformamide (DMF) and N-methyl-2-pyrrolidone (NMP) is especially concerning.3 Due to their well-documented health hazards and environmental persistence, these solvents are increasingly subject to strict regulatory limitations,4 underscoring the pressing need for greener and safer alternatives.A promising strategy involves replacing organic solvents with water, often in combination with surfactants or other enabling additives. This strategy provides significant environmental, safety, and economic benefits, as highlighted in numerous recent studies.5 However, the process water together with the surface-active additives ultimately ends up as waste, necessitating complex wastewater treatment or disposal protocols.6 In contrast, mechanochemistry techniques employ mechanical force to initiate chemical reactions under completely solvent-free or solvent-minimized conditions,7 thereby further reducing waste generation and environmental impact.8
The earliest documented example of mechanochemical amide bond formation dates back to 2009, when Lamaty and co-workers reported the solvent-free synthesis of di- and tripeptides via the reaction of urethane-protected α-amino acid N-carboxyanhydride derivatives with amino esters, driven solely by mechanical forces.9 Since this pioneering study, the field has rapidly expanded, and numerous protocols have emerged. These include amidation of esters, direct couplings of carboxylic acids with amines, reactions involving carboxyanhydrides, peptide assembly through fragment condensation or protected amino acid couplings, and even mechanoenzymatic strategies for oligopeptide synthesis.10 Notably, mechanochemical amidation has been extensively applied to the synthesis of various active pharmaceutical ingredients (APIs), and other biologically relevant structures.11 Initially, such transformations were carried out using small-scale milling devices, such as mixer or planetary ball mills, which apply mechanical force through the movement of grinding balls.10b–d,f–j,11a,d–f These instruments typically produce material on a gram scale, while larger scale versions remain scarce, particularly for applications in synthetic chemistry. More recently, mechanochemical amidations have been demonstrated using extruders, which continuously force reagent mixtures through confined spaces, applying shear and compression forces.10a,e,11b,c Unlike ball milling, reactive extrusion enables continuous operation and offers flexible production up to manufacturing scale, although experience with its implementation in commercial settings remains very limited.12 While adopting reactive extrusion as a scale-up strategy for ball milling might appear practical, translating conditions from ball mills to extruders is often not straightforward, due to substantial technical differences and the fundamentally distinct operating parameters that define them (e.g., ball size/weight and milling frequency vs. screw configuration and speed).12
In mechanochemical transformations, small amounts of liquid additives, typically organic solvents, are often employed to enhance or control both reactivity and processability.13 This approach, known as liquid-assisted grinding (LAG), can improve mixing, facilitate mass transfer, and accelerate reaction rates through complex interactions.14 In many cases, the use of LAG additives has proven essential in fine-tuning mechanochemical amidations that are not optimal under neat grinding conditions.10b–e,g–i,11a–c
Another related approach that shows considerable promise for the mechanochemical activation of chemical transformations is bead milling.15 This technique employs a large number of microbeads in a grinding chamber agitated by intensive rotation, thereby delivering high energy input. Compared with ball mills, a key advantage of bead mills is their straightforward scalability and flexibility to operate in either continuous or batch mode. Traditionally, bead milling has been applied to particle size reduction in areas such as pigment dispersion, pharmaceutical and nutritional processing, mineral grinding, and nanoparticle fabrication.16 Despite these well-established applications, and in contrast to their apparent technical resemblance to small-scale ball mills, the potential of bead mills to promote mechanochemical transformations has only recently begun to be explored. Reported applications include, Beckmann rearrangements, nitrations using a bench-stable nitrating reagent, synthesis of deep eutectic solvents and also biomass valorization.17
Given the critical importance of scalable, solvent-minimized approaches to amide bond formation, we set out to investigate bead milling as a potential enabling technology, which to date has not been applied to mechanochemical amidations. For this purpose, we employed a commercially available agitator bead mill, the Dyno®-Mill Research Lab, which features a proprietary high-speed grinding system capable of delivering intense mechanical energy.18 We anticipated that this laboratory-scale device could serve as a proof of concept for promoting mechanochemical amide couplings via bead milling, while the commercial availability of industrial-scale versions of the same system supports the feasibility of further scale-up without significant modifications to process characteristics.19
Results and discussion
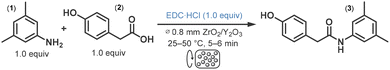
The amidation of 4-hydroxyphenylacetic acid (2) with 3,5-dimethylaniline (1) was chosen as a model reaction for process optimization. This transformation affords amide 3, an advanced intermediate of efaproxiral, a radiation-sensitizing agent developed primarily for cancer treatment.20 1-Ethyl-3-(3-(dimethylamino)propyl)-carbodiimide hydrochloride (EDC·HCl) was used as coupling agent, as its water solubility allows for easy removal of the urea byproduct thus facilitating product isolation without any organic solvent needed.5c The carboxylic acid, the aniline derivative, and the coupling agent were used in equimolar amounts to maximize atom economy and simplify work-up and purification. Among these components, 3,5-dimethylaniline was the only liquid, while both the carboxylic acid and coupling agent were solids.The Dyno®-Mill Research Lab employs bead milling technology to deliver high energy input, ensuring efficient mechanochemical reactions (Fig. 1; see also Fig. S1 in the SI).17,18 It features a cylindrical silicon carbide grinding chamber and a specialized rotational grinding system, the Dyno®-Accelerator, mounted on a rotating shaft. The reaction mixture is continuously fed into the chamber via a feed screw, where it combines with a large number of miniature grinding beads (∅ 0.05–1.0 mm). The high rotational speed of the accelerator imparts motion to the grinding beads and creates a multitude of impacts, providing mechanochemical activation via shock, pressure, shear, friction, impact, and torsion, while constantly renewing the surface of the reactants. Additionally, strong turbulence further promotes excellent mixing and effective mass transfer within the reactor zone. The fast rotational movement continuously drives the reaction mixture toward the reactor outlet, where a sieve plate retains the grinding beads. The system can be operated either in continuous single-pass mode or in recirculation mode, the latter achieved by redirecting the outlet flow back to the inlet funnel.
Owing to their excellent mechanical properties and exceptional hardness (Mohs scale 9), yttria-stabilized zirconia beads (ZrO2/Y2O3; ∅ 0.8 mm) were selected as the grinding medium. The bead filling level in the grinding chamber was maintained at 60 v/v%, and the reactor was operated in continuous flow single-pass mode at accelerator speeds of 1500–6000 rpm on a 50 mmol scale. Under these conditions, the entire reaction mixture was discharged from the reactor within 5–6 min, allowing for short processing times and rapid parameter optimization.
We initiated our study by evaluating the reaction in the presence of added bases. Guided by our earlier findings,5c pyridine was first tested, affording a promising 83% yield at 50 °C and an accelerator speed of 3800 rpm (Table 1, entry 1). Recognizing that small quantities of liquid additives can facilitate mechanochemical processes, we next explored LAG conditions using a small amount of EtOH (η = 0.5 μL mg−1), a solvent broadly compatible with the reagents.21 Remarkably, the addition of EtOH improved the yield to 97% while allowing the reaction to proceed efficiently at room temperature (25 °C; Table 1, entry 2). In contrast, replacing pyridine with triethylamine (TEA) gave a significantly lower yield of 57% (Table 1, entry 3). Notably, potassium hydroxide, a cost-effective and non-toxic alternative to pyridine, delivered a yield of 83% (Table 1, entry 4).
| #a,b | LAG | Basec | Rotor speed [rpm] | Scale (mmol) | T [°C] | Yieldd [%] | |
|---|---|---|---|---|---|---|---|
| Solvent | η [μL mg−1] | ||||||
| a Reactions were performed in continuous flow single-pass mode using the Dyno®-Mill Research Lab reactor. b In all experiments, 187 g of ZrO2/Y2O3 beads were used, corresponding to a reactor filling degree of 60 v/v%. c 1.0 equiv. of base. d Determined by HPLC-UV at 215 nm. e Sticky reaction mixture, complex to remove from the reactor. | |||||||
| 1e | — | — | Pyridine | 3800 | 50 | 50 | 83 |
| 2 | EtOH | 0.5 | Pyridine | 3800 | 50 | 25 | 97 |
| 3 | EtOH | 0.5 | TEA | 3800 | 50 | 25 | 57 |
| 4 | EtOH | 0.5 | KOH | 3800 | 50 | 25 | 83 |
| 5 | EtOAc | 0.5 | — | 3800 | 50 | 25 | 84 |
| 6 | EtOAc | 0.5 | — | 3800 | 200 | 25 | 86 |
| 7 | EtOAc | 0.5 | — | 6000 | 50 | 25 | 95 |
| 8 | EtOAc | 0.5 | — | 1500 | 50 | 25 | 51 |
| 9 | EtOAc | 0.34 | — | 3800 | 50 | 25 | 96 |
| 10 | EtOAc | 0.25 | — | 3800 | 50 | 25 | 82 |
| 11 | DMSO | 0.34 | — | 3800 | 50 | 25 | 29 |
| 12 | 2MeTHF | 0.34 | — | 3800 | 50 | 25 | 97 |
However, when EtOH was used as the LAG additive, ester formation was observed as a side reaction in some of the experiments. This prompted us to explore ethyl acetate (EtOAc) as an alternative.10c,h,i,11a,b To our delight, EtOAc performed very well, affording a yield of 84% under the same conditions as EtOH, but without the need for any added base (Table 1, entry 5). Eliminating the base is very beneficial, not only because of the toxicity of pyridine, but it also improves the overall atom economy of the transformation. When increasing the reaction scale from 50 mmol to 200 mmol, a practically identical yield was obtained (Table 1, entry 5 vs. entry 6), providing encouraging preliminary evidence for the scalability of the process. To fine-tune the amount of mechanical energy input, we next investigated the effect of Dyno®-Accelerator rotation speed. As expected, increasing the agitator speed to the maximum of 6000 rpm led to a higher yield (95%), whereas reducing it to 1500 rpm resulted in a substantial drop to 51% (Table 1, entry 5 vs. entries 7 and 8). These results clearly underscore the crucial role of mechanical energy in this process. Despite the very good result at 6000 rpm rotation speed, we selected 3800 rpm for further experimentation to avoid running the system at its maximum performance.
The amount of LAG additive proved to be a critical parameter. Compared to the initially applied 0.5 μL mg−1 (Table 1, entry 5), reducing the amount to 0.34 μL mg−1 improved the outcome, delivering an excellent yield of 96% under otherwise identical conditions (Table 1, entry 9). However, a further reduction to 0.25 μL mg−1 proved suboptimal, resulting in a lower yield of 82% (Table 1, entry 10). Finally, DMSO and 2MeTHF were also tested as alternative LAG additives. Interestingly, DMSO caused a pronounced decrease in yield to 29%, whereas 2MeTHF performed comparably to EtOAc (Table 1, entries 11 and 12). Given its lower cost and more favorable environmental profile,22 EtOAc was chosen as the LAG additive for subsequent process development.
A series of control experiments were next performed to evaluate the impact of mechanochemical activation in comparison with conventional mixing strategies. As a baseline, a control experiment was conducted by combining equimolar amounts of aniline 1, carboxylic acid 2, and EDC·HCl with EtOAc (η = 0.34 μL mg−1) in a flask, which was left to stand at 25 °C for 5 min without mixing or agitation. As expected, this resulted in a low yield of 15% (Table 2, entry 1). In contrast, manually stirring the same reaction mixture with a spatula for 5 min at 25 °C increased the yield to 51% (Table 2, entry 2). A comparable result was obtained when the mixture was agitated using a horizontal shaker (Table 2, entry 3). To further investigate the influence of mechanochemical conditions on the model reaction, the same procedure was next carried out using a mixer ball mill (Retsch MM400) operating at 30 Hz for the same duration. Highlighting the effectiveness of mechanochemical activation, this resulted in a yield of 75%, outperforming conventional agitation but falling somewhat short compared to the bead mill (Table 2, entry 4 vs. entries 2, 3 and 5). To better assess the alignment between these mechanochemical approaches, several experiments initially performed in the Dyno®-Mill during parameter optimization (Table 2) were repeated in the mixer ball mill. The observed trends under varying conditions were highly consistent, although yields from ball milling were generally slightly lower (see Table S1 in the SI), likely due to the higher mechanical energy input provided by the bead mill. These findings indicate that reaction conditions are readily transferable between these setups across different scales. Moreover, the enhanced energy input in bead milling may allow for shorter reaction times, although ball milling parameters such as frequency, ball size, and material were not fully optimized in this study. Bead milling thus emerges as a promising scale-up strategy, combining the efficacy of traditional ball milling with the advantages of continuous operation in systems like the Dyno®-Mill.
| # | Experimental conditions | Yielda [%] |
|---|---|---|
| a Determined by HPLC-UV at 215 nm. b 1.0 mmol scale. c Using a Retsch MM400 mixer mill operating at 30 Hz with ∅ 0.4 cm stainless steel balls (×20). | ||
| 1b | Non-mixed control reaction | 15 |
| 2b | Mixed with a spatula in a beaker | 51 |
| 3b | Agitated using a horizontal shaker | 50 |
| 4b | Performed in a mixer ball millc | 75 |
| 5 | Performed in Dyno®-Mill (Table 2, entry 9) | 96 |
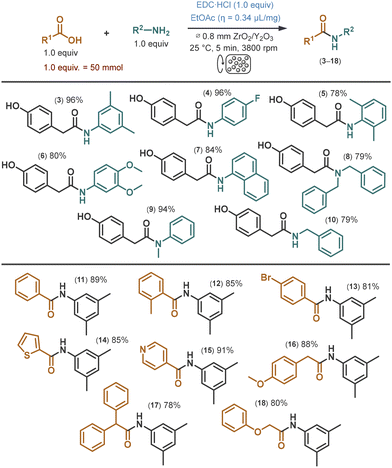
To demonstrate the scope and generality of the process, a series of amidations involving various carboxylic acids and amines was performed in the Dyno®-Mill reactor (Scheme 1). All experiments were carried out at the 50 mmol scale under the previously optimized conditions using continuous flow single-pass mode. A range of amines was reacted with 4-hydroxyphenylacetic acid (2) as the common coupling partner. Aniline derivatives with diverse substitution patterns, as well as 1-naphthylamine, delivered the corresponding amides (3–7) in yields ranging from 78% to 96%. Among these, the sterically hindered 2,6-dimethylaniline exhibited the lowest reactivity, yielding 78% of the product (5). Secondary amines such as dibenzylamine and N-methylaniline were also well tolerated, furnishing tertiary amides 8 and 9 in 79% and 94% yields, respectively. Benzylamine similarly reacted smoothly, providing amide 10 in 79% yield. Unlike most of the amines explored, 1-naphthylamine and 3,4-dimethoxyaniline are solids; therefore, their amidation with 4-hydroxyphenylacetic acid involved exclusively solid components apart from the LAG additive. The scope of carboxylic acid derivatives was evaluated using 3,5-dimethylaniline (1) as the coupling partner. Benzoic acid and its substituted derivatives gave the corresponding amides (11–13) in good yields of up to 89%. Heteroaromatic carboxylic acids, such as thiophene-2-carboxylic acid and isonicotinic acid also reacted efficiently, affording amides 14 and 15 in excellent yields of 85% and 91%, respectively. The process also proved effective for the amidation of substituted phenylacetic acid derivatives as well as 2-phenoxyacetic acid, furnishing amides 16–18 in yields of up to 88%. Notably, the amide products were isolated at ≥90% purity without the use of any organic solvents, following a straightforward work-up procedure involving precipitation with cold water, filtration, washing with water, and drying. This not only minimizes environmental impact but also highlights the practicality of the method, underscoring its potential to be extended to larger-scale syntheses under sustainable conditions.
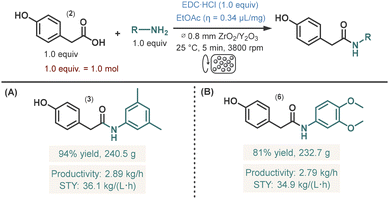
Finally, to assess the scalability of the process, we conducted scale-up experiments using two model reactions: the amidation of 4-hydroxyphenylacetic acid (2) with either 3,5-dimethylaniline (1) or 3,4-dimethoxyaniline (Scheme 2). The latter reaction involved only solid reagents and EtOAc as a LAG additive, whereas 3,5-dimethylaniline in the first reaction is a liquid (see SI for photographs taken during the experiments). Both reactions were performed on a 1.0 mol scale, representing a 20-fold increase compared to the initial 50 mmol experiments. Reaction conditions were directly taken from the small-scale experiments, with the sole modification being the use of recirculation mode to accommodate the larger reagent quantities within the comparatively limited reactor volume. The accelerator speed was maintained at 3800 rpm, and recirculation was continued for 5 min to match the reaction time of the small-scale experiments. Gratifyingly, the 1.0 mol reactions delivered yields comparable to those obtained at the smaller scale (94% and 81% for amides 3 and 6, Scheme 2A and B, respectively. Within just 5 min, the experiments furnished 240.5 g and 232.7 g of amide products 3 and 6, respectively following precipitation, filtration, washing with water, and drying. These results correspond to productivities of 2.89 kg h−1 and 2.79 kg h−1, and space–time yields (STY) of 36.1 kg (L h)−1 and 34.1 kg (L h)−1, respectively. Importantly, the consistency of results from model reactions with both solid–liquid and solid–solid mixtures demonstrate that process efficiency is not governed by the rheological properties of the mixture, underscoring the robustness and broad applicability of the methodology.
Importantly, these reactions proceeded without requiring reagent excess, added bases, or large volumes of organic solvents, thereby minimizing waste generation. This is reflected in the low E-factors of 1.5 and 1.7 and the corresponding process mass intensities (PMIs) of 2.5 and 2.8 for the synthesis of amides 3 and 6, respectively (Table 3). The calculated reaction mass efficiency (RME), atom economy (AE), and optimum efficiency (OE) further highlight the environmental credentials of the methodology.23 For amide 3, the green metrics were directly compared with those obtained in our previously reported slurry flow process employing water as the reaction medium and hydroxypropyl methylcellulose (HPMC) as an enabling additive.5c The superior performance of the Dyno(R)-Mill protocol, reflected by lower E-factor and PMI values together with improved RME, AE, and OE, can be attributed to the elimination of pyridine and HPMC, both major contributors to waste in the slurry process. Additionally, the markedly higher productivity achieved under mechanochemical conditions constitutes a further significant advantage.
Conclusions
In summary, we have established a robust and scalable mechanochemical protocol for amide bond formation using bead milling in a Dyno®-Mill Research Lab reactor. Employing EDC·HCl as the coupling agent under strictly equimolar conditions, the reactions proceeded efficiently without reagent excess or additional base, addressing important limitations of conventional amidations. The process was further enhanced under LAG conditions, with EtOAc identified as the optimal additive owing to its high efficacy, low toxicity, and favorable environmental profile. Crucially, product isolation required no organic solvents, relying solely on precipitation, filtration, and washing with water. This simplified downstream processing not only reduced environmental impact but also underscores the practicality of the protocol for larger-scale operations. Optimization of key mechanochemical parameters (accelerator speed, additive volume) enabled high-yielding amidations across a broad substrate scope, highlighting the versatility of the method. Control experiments in a mixer ball mill demonstrated strong consistency across platforms and scales providing compelling evidence that bead milling offers a viable route for translating and scaling ball milling methodologies. Using optimized conditions, a 20-fold scale-up was successfully achieved, delivering productivities up to 2.89 kg h−1 with minimal waste, as reflected in low E-factor and PMI values. Importantly, continuous operation was demonstrated up to the 200 mmol scale in single-pass mode, while the 1.0 mol scale was realized in recirculation mode, with further scalability anticipated in larger reactors.Taken together, these results establish bead milling as a sustainable and industrially relevant platform for continuous amide bond formation, combining high efficiency, minimal waste generation, and straightforward scalability. Ongoing studies are directed toward exploiting larger-scale bead milling reactors to enable fully continuous mechanochemical amidations with on-demand batch sizes suitable for pharmaceutical and fine chemical manufacturing.
Experimental section
Experiments were carried out using a Dyno®-Mill Research Lab agitator bead mill (WAB-GROUP®, Switzerland), equipped with an 80 mL grinding chamber featuring the proprietary Dyno®-Accelerator rotational grinding system.18 The chamber was loaded with 187 g of ZrO2/Y2O3 grinding beads (∅ 0.8 mm), corresponding to a reactor filling degree of 60% v/v. The reaction mixture was fed via a feed screw. Temperature control was maintained using an external thermostat (Ministat 240, Huber, Germany), with a 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 water/ethylene glycol mixture as the cooling medium.
1 water/ethylene glycol mixture as the cooling medium.
For reactions at 50 mmol and 200 mmol scales, the reactor was operated in continuous flow single-pass mode at rotation speeds of 1500, 3800, or 6000 rpm. Scale-up experiments at the 1.0 mol scale were conducted in recirculation mode at a rotation speed of 3800 rpm. In the smaller scale experiments, the carboxylic acid component and EDC·HCl were introduced into the inlet hopper along with the LAG additive, followed by the corresponding amine derivative. For the 1.0 mol scale experiments, 4-hydroxyphenylacetic acid (2) and EDC·HCl were premixed with EtOAc (LAG additive) in a beaker until a uniform paste was formed, then transferred to the inlet hopper. The respective aniline derivative was subsequently added, then recirculation was maintained for 5 min while maintaining the reactor temperature at 25 °C.
After completion, the crude amide products were isolated by precipitation with cold water, followed by filtration under reduced pressure, washing with water and drying. For HPLC analysis, samples were diluted with 1 mL of MeCN prior to injection. The isolated compounds were characterized by 1H and 13C NMR spectroscopy and HRMS.
Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: further details on instrumentation, reaction setups as well as general experimental procedures. See DOI: https://doi.org/10.1039/d5gc04764a.Acknowledgements
The Research Center Pharmaceutical Engineering (RCPE) is funded within the framework of COMET – Competence Centers for Excellent Technologies by BMK, Bundesministerium für Digitalisierung und Wirtschaftsstandort, Land Steiermark and Steirische Wirtschaftsförderungsgesellschaft. The COMET program is managed by the FFG. We would like to thank Willy A. Bachofen AG (WAB-GROUP®) for the generous loan of the Dyno®-Mill Research Lab reactor used in this study. We also thank Dr Philippe Roth for his valuable support, as well as Dr Heidrun Gruber-Woelfler for her assistance during the initial ball-milling tests.References
- (a) K. Sharma, K. K. Sharma, A. Sharma and R. Jain, Drug Discovery Today, 2023, 28, 103464 Search PubMed; (b) J. Pitzer and K. Steiner, J. Biotechnol., 2016, 235, 32–46 Search PubMed.
- (a) I. Kekessie, K. Wegner, I. Martinez, M. E. Kopach, T. D. White, J. K. Tom, M. N. Kenworthy, F. Gallou, J. Lopez, S. G. Koenig, P. R. Payne, S. Eissler, B. Arumugam, C. Li, S. Mukherjee, A. Isidro-Llobet, O. Ludemann-Hombourger, P. Richardson, J. Kittelmann, D. Sejer Pedersen and L. J. van den Bos, J. Org. Chem., 2024, 89, 4261–4282 CrossRef CAS PubMed; (b) L. Ferrazzano, M. Catani, A. Cavazzini, G. Martelli, D. Corbisiero, P. Cantelmi, T. Fantoni, A. Mattellone, C. De Luca, S. Felletti, W. Cabri and A. Tolomelli, Green Chem., 2022, 24, 975–1020 RSC.
- (a) P. Acosta-Guzmán, A. Ojeda-Porras and D. Gamba-Sánchez, Adv. Synth. Catal., 2023, 365, 4359–4391 CrossRef; (b) E. Massolo, M. Pirola and M. Benaglia, Eur. J. Org. Chem., 2020, 4641–4651 Search PubMed.
- (a) J. Sherwood, F. Albericio and B. G. de la Torre, ChemSusChem, 2024, 17, e202301639 Search PubMed; (b) J. Sherwood, T. J. Farmer and J. H. Clark, Chem, 2018, 4, 2010–2012 Search PubMed.
- For recent reviews, see: (a) B. H. Lipshutz, Green Chem., 2024, 26, 739–752 Search PubMed; (b) S. Hazra, F. Gallou and S. Handa, ACS Sustainable Chem. Eng., 2022, 10, 5299–5306 CrossRef CAS. For selected examples, see: (c) S. H. A. Rajendran, S. Kogler, P. Kögl, W. M. Braje, S. B. Ötvös and C. O. Kappe, ACS Sustainable Chem. Eng., 2025, 13, 6423–6432 Search PubMed; (d) D. Petkova, N. Borlinghaus, S. Sharma, J. Kaschel, T. Lindner, J. Klee, A. Jolit, V. Haller, S. Heitz, K. Britze, J. Dietrich, W. M. Braje and S. Handa, ACS Sustainable Chem. Eng., 2020, 8, 12612–12617 CrossRef CAS; (e) S. Sharma, N. W. Buchbinder, W. M. Braje and S. Handa, Org. Lett., 2020, 22, 5737–5740 CrossRef CAS PubMed; (f) C. M. Gabriel, M. Keener, F. Gallou and B. H. Lipshutz, Org. Lett., 2015, 17, 3968–3971 CrossRef CAS.
- N. Fleck, F. Roschangar and A. M. Haydl, Org. Process Res. Dev., 2023, 27, 822–830 CrossRef CAS.
- For recent reviews, see: (a) L. Dong, L. Li, H. Chen, Y. Cao and H. Lei, Adv. Sci., 2025, 12, 2403949 Search PubMed; (b) J. F. Reynes, F. Leon and F. García, ACS Org. Inorg. Au, 2024, 4, 432–470 CrossRef PubMed; (c) R. S. Atapalkar and A. A. Kulkarni, React. Chem. Eng., 2024, 9, 10–25 RSC; (d) F. Cuccu, L. De Luca, F. Delogu, E. Colacino, N. Solin, R. Mocci and A. Porcheddu, ChemSusChem, 2022, 15, e202200362 CrossRef; (e) O. Bento, F. Luttringer, T. Mohy El Dine, N. Pétry, X. Bantreil and F. Lamaty, Eur. J. Org. Chem., 2022, e202101516 Search PubMed; (f) T. Friščić, C. Mottillo and H. M. Titi, Angew. Chem., Int. Ed., 2020, 59, 1018–1029 Search PubMed; (g) J. L. Howard, Q. Cao and D. L. Browne, Chem. Sci., 2018, 9, 3080–3094 RSC; (h) J. Andersen and J. Mack, Green Chem., 2018, 20, 1435–1443 RSC.
- (a) E. P. T. Leitão, RSC Sustainability, 2024, 2, 3655–3668 RSC; (b) J. Alić, M.-C. Schlegel, F. Emmerling and T. Stolar, Angew. Chem., Int. Ed., 2024, 63, e202414745 CrossRef; (c) N. Fantozzi, J.-N. Volle, A. Porcheddu, D. Virieux, F. García and E. Colacino, Chem. Soc. Rev., 2023, 52, 6680–6714 RSC; (d) K. J. Ardila-Fierro and J. G. Hernández, ChemSusChem, 2021, 14, 2145–2162 Search PubMed.
- V. Declerck, P. Nun, J. Martinez and F. Lamaty, Angew. Chem., Int. Ed., 2009, 48, 9318–9321 Search PubMed.
- (a) R. R. A. Bolt, H. R. Smallman, J. A. Leitch, G. W. Bluck, F. Barreteau, A. V. Iosub, D. Constable, O. Dapremont, P. Richardson and D. L. Browne, Angew. Chem., Int. Ed., 2024, 63, e202408315 CrossRef; (b) Y. Yeboue, N. Rguioueg, G. Subra, J. Martinez, F. Lamaty and T.-X. Métro, Eur. J. Org. Chem., 2022, e202100839 CrossRef; (c) Y. Yeboue, M. Jean, G. Subra, J. Martinez, F. Lamaty and T.-X. Métro, Org. Lett., 2021, 23, 631–635 CrossRef PubMed; (d) T. Dalidovich, K. A. Mishra, T. Shalima, M. Kudrjašova, D. G. Kananovich and R. Aav, ACS Sustainable Chem. Eng., 2020, 8, 15703–15715 CrossRef; (e) Y. Yeboue, B. Gallard, N. Le Moigne, M. Jean, F. Lamaty, J. Martinez and T.-X. Métro, ACS Sustainable Chem. Eng., 2018, 6, 16001–16004 CrossRef; (f) J. G. Hernández, K. J. Ardila-Fierro, D. Crawford, S. L. James and C. Bolm, Green Chem., 2017, 19, 2620–2625 Search PubMed; (g) L. Gonnet, T. Tintillier, N. Venturini, L. Konnert, J.-F. Hernandez, F. Lamaty, G. Laconde, J. Martinez and E. Colacino, ACS Sustainable Chem. Eng., 2017, 5, 2936–2941 CrossRef; (h) V. Porte, M. Thioloy, T. Pigoux, T.-X. Métro, J. Martinez and F. Lamaty, Eur. J. Org. Chem., 2016, 3505–3508 CrossRef; (i) J. Bonnamour, T.-X. Métro, J. Martinez and F. Lamaty, Green Chem., 2013, 15, 1116–1120 Search PubMed; (j) J. G. Hernández and E. Juaristi, J. Org. Chem., 2010, 75, 7107–7111 CrossRef.
- (a) T. Nikonovich, T. Jarg, J. Martõnova, A. Kudrjašov, D. Merzhyievskyi, M. Kudrjašova, F. Gallou, R. Aav and D. Kananovich, RSC Mechanochem., 2024, 1, 189–195 RSC; (b) M. Lavayssiere and F. Lamaty, Chem. Commun., 2023, 59, 3439–3442 RSC; (c) R. S. Atapalkar and A. A. Kulkarni, Chem. Commun., 2023, 59, 9231–9234 RSC; (d) W. I. Nicholson, F. Barreteau, J. A. Leitch, R. Payne, I. Priestley, E. Godineau, C. Battilocchio and D. L. Browne, Angew. Chem., Int. Ed., 2021, 60, 21868–21874 CrossRef PubMed; (e) T.-X. Métro, J. Bonnamour, T. Reidon, A. Duprez, J. Sarpoulet, J. Martinez and F. Lamaty, Chem. – Eur. J., 2015, 21, 12787–12796 CrossRef; (f) T.-X. Métro, J. Bonnamour, T. Reidon, J. Sarpoulet, J. Martinez and F. Lamaty, Chem. Commun., 2012, 48, 11781–11783 RSC.
- (a) J. F. Reynes, V. Isoni and F. García, Angew. Chem., Int. Ed., 2023, 62, e202300819 CrossRef PubMed; (b) R. R. A. Bolt, J. A. Leitch, A. C. Jones, W. I. Nicholson and D. L. Browne, Chem. Soc. Rev., 2022, 51, 4243–4260 RSC.
- L. E. Wenger and T. P. Hanusa, Chem. Commun., 2023, 59, 14210–14222 RSC.
- (a) S. A. Salami, M. H. Manyeruke, C. I. Ezekiel, U. N. Ndagano, J. B. Safari, S. O. Amusat and R. W. M. Krause, Results Chem., 2025, 18, 102675 CrossRef; (b) P. Ying, J. Yu and W. Su, Adv. Synth. Catal., 2021, 363, 1246–1271 CrossRef.
- (a) L. Little, A. N. Mainza, M. Becker and J. Wiese, Miner. Eng., 2017, 111, 148–157 CrossRef; (b) F. Shi, R. Morrison, A. Cervellin, F. Burns and F. Musa, Miner. Eng., 2009, 22, 673–680 CrossRef.
- (a) S. Arfelis, I. Malpartida, A. Bala, V. Lair, R. Xifré, R. Aguado, M. Delgado-Aguilar, J. Parduhn, I. Sazdovski and P. Fullana-i-Palmer, ACS Sustainable Chem. Eng., 2024, 12, 5105–5116 CrossRef; (b) G. Guner, M. Mehaj, N. Seetharaman, S. Elashri, H. F. Yao, D. J. Clancy and E. Bilgili, Pharmaceutics, 2023, 15, 2213 CrossRef PubMed; (c) H. Tanaka, Y. Ochii, Y. Moroto, T. Ibaraki and K.-i. Ogawara, Chem. Pharm. Bull., 2021, 69, 81–85 CrossRef; (d) C. Knieke, C. Steinborn, S. Romeis, W. Peukert, S. Breitung-Faes and A. Kwade, Chem. Eng. Technol., 2010, 33, 1401–1411 CrossRef.
- (a) V. Valsamidou, S. Patra, B. Kadriu, M. G. Metzger, L. Gremaud and D. Katayev, Green Chem., 2025, 27, 7122–7128 Search PubMed; (b) R. Nguyen, S. Halloumi, I. Malpartida and C. Len, Org. Process Res. Dev., 2025, 29, 769–777 CrossRef; (c) R. Geib, E. Colacino and L. Gremaud, ChemSusChem, 2024, 17, e202301921 CrossRef; (d) R. Nguyen, A. Auvigne, A. M. Pérez Merchán, I. Malpartida and C. Len, Org. Process Res. Dev., 2024, 28, 3560–3569 Search PubMed; (e) A. I. Martín-Perales, D. Rodríguez-Padrón, A. M. Balu, S. Halloumi, I. Malpartida and R. Luque, Ind. Eng. Chem. Res., 2023, 62, 17545–17552 CrossRef.
- https://www.wab-group.com/en/products/dyno-mill-research-lab/ .
- https://www.wab-group.com/en/products/dyno-mill-ecm-ap/ .
- J. H. Suh, Expert Opin. Invest. Drugs, 2004, 13, 543–550 Search PubMed.
- (a) J. Gómez-Carpintero, J. D. Sánchez, J. F. González and J. C. Menéndez, J. Org. Chem., 2021, 86, 14232–14237 CrossRef; (b) E. Broumidis, M. C. Jones, F. Vilela and G. O. Lloyd, ChemPlusChem, 2020, 85, 1754–1761 CrossRef PubMed.
- D. Prat, A. Wells, J. Hayler, H. Sneddon, C. R. McElroy, S. Abou-Shehada and P. J. Dunn, Green Chem., 2016, 18, 288–296 Search PubMed.
- C. R. McElroy, A. Constantinou, L. C. Jones, L. Summerton and J. H. Clark, Green Chem., 2015, 17, 3111–3121 RSC.
| This journal is © The Royal Society of Chemistry 2026 |