Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
In situ photocatalytic formation of carbon quantum dots from corn stover via interfacial hydrogen peroxide generation
Xuejing Kanga,
Qian Yangb,
Sean Xiang Tanga,
Zhe Chena,
Samir Budhathokia,
Ramhari Panerua,
Soyoung Kim g,
Yan Bai
g,
Yan Bai c,
Qian Lic,
Zhongbing Chen
c,
Qian Lic,
Zhongbing Chen d,
Alexander Goroncy
d,
Alexander Goroncy e,
Richard N. Zare
e,
Richard N. Zare *f and
Maohong Fan
*f and
Maohong Fan *gh
*gh
aDepartment of Energy & Petroleum Engineering, University of Wyoming, Laramie, WY 82071, USA
bCenter for Advanced Scientific Instrumentation, University of Wyoming, Laramie, WY 82071, USA
cCollege of Chemistry and Chemical Engineering, Henan University, Kaifeng, Henan 475004, P.R. China
dDepartment of Applied Ecology, Faculty of Environmental Sciences, Czech University of Life Sciences Prague, Kamýcká 129, 16500 Praha-Suchdol, Czech Republic
eDepartment of Chemistry, University of Wyoming, Laramie, WY 82071, USA
fDepartment of Chemistry, Stanford University, Stanford, CA 94305, USA. E-mail: zare@stanford.edu
gCollege of Engineering and Physical Sciences, and School of Energy Resources, University of Wyoming, Laramie, WY 82071, USA. E-mail: mfan@uwyo.edu
hCollege of Engineering, Georgia Institute of Technology, Atlanta, GA 30332, USA
First published on 12th March 2026
Abstract
We present a sustainable photocatalytic approach for synthesizing carbon quantum dots (CQDs) from renewable corn stover under simulated solar irradiation. In this process, hydrogen peroxide (H2O2) is spontaneously and continuously generated at the gas–water microdroplet interface. Naturally occurring ferrous (Fe2+) and other multivalent ions in the corn stover catalyze the decomposition of H2O2, producing highly reactive hydroxyl radicals (˙OH) that drive the oxidation and fragmentation of lignocellulosic components, ultimately leading to CQD formation. An oxygen-rich environment further facilitates the reaction. Compared to conventional methods, this approach enhances H2O2 utilization efficiency while avoiding excessive oxidation associated with high doses of external oxidants (e.g., H2O2 and HNO3). The resulting CQDs (4.6 ± 0.8 nm) exhibit intense blue fluorescence with a quantum yield of up to 20.1 ± 0.8%. Structural analyses (XRD, FTIR, Raman, NMR, and XPS) confirm their amorphous graphitic nature with nitrogen self-doping. Uniform CQDs with an average size of 3.1 ± 1.4 nm and high purity were obtained through centrifugation and dialysis purification. This low-cost, scalable method, which integrates in situ H2O2 generation at the microdroplet interface, offers a promising pathway for environmentally friendly CQD production.
Green foundation1. We demonstrate a low-energy, catalyst-free, and renewable feedstock-based strategy to produce high-value nanomaterials, namely carbon quantum dots (CQDs), from agricultural waste (corn stover) using water microdroplets.2. CQDs of 3–5 nm size with quantum yields of up to 20.1% were produced without strong acids or concentrated oxidants. The method avoids liters of 1–10 M HNO3 or >30% H2O2 solutions typically required in conventional routes. 3. This work can be made greener and elevated by further research on replacing simulated solar light with direct sunlight, further reducing energy demand, on employing recycled or wastewater streams to minimize freshwater consumption, and by investigating whether residual lignin/cellulose fractions after CQD formation can be valorized, achieving a zero-waste biorefinery model. |
1. Introduction
Carbon Quantum Dots (CQDs) are carbon-based nanoparticles, typically less than 10 nm in size, renowned for their exceptional optical, electrical, and chemical properties.1–4 Since their discovery in 2004,5 CQDs have received much interest because of their diverse applications in bioimaging, drug delivery, environmental sensing, and clean energy production.6–9 Their intrinsic photoluminescence, chemical stability, and biocompatibility position them as a promising, nontoxic alternative to traditional semiconductor quantum dots, particularly for sustainable applications.10–13 Moreover, CQDs offer an eco-friendly nanomaterial solution, potentially reducing costs associated with CO2 emission control.14–16CQDs have been synthesized through top-down methods, such as laser ablation, arc discharge, and electrochemical exfoliation, which break down larger carbon structures, or bottom-up methods, including chemical oxidation, microwave irradiation, hydrothermal, and pyrolysis techniques, which assemble CQDs from molecular precursors.3,17,18 Conventional carbon sources include graphite and graphene in top-down approaches, and citric acid or glucose in bottom-up approaches. A promising and sustainable alternative involves synthesizing CQDs from renewable agricultural biomass,19 promoting a circular economy. However, current biomass-based synthesis methods often rely on concentrated acids, strong oxidants (e.g., H3PO4, H2SO4, and HNO3), and high-temperature reaction conditions, which limit their environmental and economic viability.
Hydrogen peroxide (H2O2) is an environmentally friendly oxidizing agent that decomposes into water, making it attractive for various applications, including bleaching, sterilization, and organic reactions.20 While some studies have employed Fenton reagents (acidic H2O2/Fe2+–Fe3+) or H2O2/W18O49 systems for carbon nanodot synthesis,21–23 Zare and colleagues recently demonstrated a novel method for in situ H2O2 generation using mechanical energy-driven microdroplets at water–gas and water–solid interfaces.24–28
Although microdroplet chemistry has advanced rapidly, prior studies have centered on pure water or simple dissolved molecules, leaving their potential for the green, low-energy conversion of renewable biomass largely unexplored. Here, we report a facile photocatalytic approach for biomass-derived CQD synthesis utilizing the spontaneous H2O-to-H2O2 conversion strategy. This method operates without external catalysts, strong oxidants, or acids. Specifically, when corn stover is mixed with deionized water and exposed to simulated sunlight, H2O2 forms in situ, enabling the direct synthesis of CQDs. This environmentally benign process provides a mild, scalable, and sustainable route for producing high-quality CQDs from raw lignocellulosic biomass and establishes a foundation for integrating microdroplet reactivity with solar-driven carbon transformation.
2. Methods
2.1 Materials
The corn stover was purchased from a research field at the University of Wyoming. Quinine sulfate dihydrate, cellulose, hemicellulose (from Aspergillus), lignin (alkali-treated), 5,5-dimethyl-1-pyrroline N-oxide (DMPO), quinine sulfate dihydrate (C20H24N2O2·H2SO4·2H2O) and ferrous sulfate heptahydrate (FeSO4·7H2O) were purchased from Sigma-Aldrich. O2 (99.999%) and N2 (99.999%) were purchased from US Welding Inc. Deionized (DI) water was used throughout this work.A Power Research Xenon lamp (ozone-free, 1000 W) for simulated solar irradiation was purchased from Newport Corporation (USA). A standard test sieve (Cole-Parmer Essentials Full Height Sieve, No. 18, 1.00 mm, USA) was used for particle control. Hydrogen peroxide H2O2 test strips (low level, 0–100 ppm, Bartovation, USA) were applied to determine the concentration of H2O2 in aqueous samples. A hotplate magnetic stirrer (Thermo Scientific/IKA C-MAG HS 7, USA) was used to stir and/or heat samples.
2.2 Fabrication of corn stover-derived CQDs
CQDs were synthesized from corn stover (including stalk, leaf, cob, and husk) using an experimental setup under the illumination of an Xe lamp. A schematic illustration for the experiment is shown in the SI Appendix (Fig. S1). Dried and cleaned corn stover was used as the sole precursor, which was first ground well using a coffee grinder and then sieved through a 1 mm mesh. Corn stover particles with sizes smaller than 1 mm were collected and used for CQD preparation. 0.5 g of corn stover powder was mixed with 100 mL of DI water and transferred into a Teflon-lined stainless-steel autoclave equipped with a quartz window. The reactor was placed on a magnetic stirrer plate and stirred the samples vigorously throughout the reaction. A controlled flow rate of pure oxygen gas was introduced into the reactor, and the reaction temperature was continuously recorded. Vacuum filtration with a 0.22 µm membrane filter was employed to remove the unreacted corn stover powder after the reaction. The residue underwent multiple rinses (2–3 times) using DI water during the filtration process. Subsequently, the liquid sample was subjected to evaporation in a VWR 1330FM mechanical oven (Marshall Scientific, USA) maintained at 50 °C, while maintaining a final CQD solution volume of 100 mL. The obtained CQD solutions were then stored under refrigeration (4 °C) for subsequent experimental applications.2.3 Characterization of CQDs
Three-dimensional excitation emission matrix (3D-EEM) fluorescence was performed using a spectrofluorometer (Fluorolog-3, Horiba, USA), and the photoluminescence property of the CQDs was determined by shifting the excitation wavelengths within 200–600 nm (slits: 4 nm) with an increase of emission wavelengths from 200 to 700 nm (slits: 2 nm). Ultraviolet-visible (UV-vis) absorption spectroscopy was done using a UV-spectrophotometer (Shimadzu). 1H-NMR and 13C-NMR spectra were recorded using a Bruker Avance III 600 MHz spectrometer with deuterium oxide (D2O) as the solvent. Fourier transform infrared (FTIR) spectrum analysis was performed using a spectrophotometer instrument with non-destructive attenuated total reflectance (ATR) technology (Nicolet iS50, Thermo Scientific, USA). The prepared CQDs were scanned 64 times at a resolution of 16 cm−1, with a wavenumber range of 4000–400 cm−1. X-ray diffraction (XRD) analysis was conducted using an X-ray diffractometer (Rigaku, Japan) with a scanning rate of 4° min−1, ranging (2θ) from 10° to 90°. FTIR and XRD were employed to determine the structural characteristics of CQDs. X-ray photoelectron spectroscopy (XPS, Kraros Axis Ultra DLD) was used to determine the surface chemical compositions. ESR spectra were recorded with a Bruker EMX EPR spectrometer (X-band, ∼9.87 GHz) using DMPO as a spin trap to detect the measurements (SI Appendix, text S7).Morphological characterization was carried out using high-resolution transmission electron microscopy (HR-TEM, JEOL JEM-2100F, Japan) with ultra-thin amorphous film-supported copper grids. The elemental composition of the corn stover was determined using a vario MACRO cube elemental analyzer (Elementar), with the oxygen content estimated by the subtraction method (SI Appendix, Table S2). Raman spectra were recorded using a DXR2 Raman spectrometer with a 532 nm excitation laser in the range of 1000–2000 cm−1 to analyze the structural characteristics of CQDs. The thermal stability of the samples was determined using a simultaneous thermal analyzer (DSC/TGA SDT Q600). Liquid chromatography-mass spectrometry (LC-MS) analysis was performed using an Agilent 1260 Infinity HPLC system coupled with a 6120 Quadrupole MS equipped with a Hi-Plex H column. Inductively coupled plasma mass spectrometry (ICP-MS) and ion chromatography (IC) analyses were performed using a Thermo iCAP RQ single quadrupole mass spectrometer and a Thermo ICS 6000 system, respectively. A pH meter (Mettler Toledo SevenCompact S220, Switzerland) was used to measure the pH values of the aqueous CQD samples.
2.4 Quantum yield measurements
The fluorescence quantum yield (QY) is the ratio of fluorescence photons emitted to excitation photons absorbed. The QYs of the prepared CQDs were evaluated by comparing their integrated photoluminescence (PL) intensities under 340 nm excitation. Quinine sulfate dihydrate in 0.1 M H2SO4, with a refractive index (η) of 1.33 and a quantum yield of 0.54 at 350 nm excitation, was used as the standard reference. The CQDs were dissolved in deionized water (refractive index η = 1.33) and evaluated at a specific concentration, ensuring that their absorbance value remained below 0.1 at 350 nm. The absorbance at the excitation wavelength of 350 nm was measured using a UV-Vis spectrophotometer (Shimadzu). The photoluminescence emission spectra of the solutions were recorded using a fluorescence spectrometer (Fluorolog-3, Horiba, USA) at an excitation wavelength of 350 nm, with both excitation and emission slit widths set to 2 nm. The integrated PL intensity, representing the total emission within the wavelength range of 300–700 nm, was determined as the area under the PL curve. The QY was calculated using the following equation:29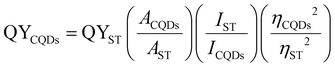 | (1) |
Here, QY denotes the fluorescence quantum yield, “A” represents the measured integrated fluorescence intensity and absorbance, “I” denotes the absorbance value, and “η” refers to the refractive index of the solvent. The subscripts “CQDs” and “ST” denote the aqueous CQD solutions and the standard solution of QS, respectively.
3. Results and discussion
3.1 Factors affecting CQD synthesis without external O2 introduction
Initial experiments (SI Appendix, Table S1) were conducted to evaluate the feasibility of generating hydrogen peroxide (H2O2) from water under solar irradiation without the introduction of external oxygen (O2). As shown in SI Appendix (Fig. S1e), H2O2 accumulated to approximately 50–100 ppm under simulated solar light (>260 nm) within five hours, primarily due to the condensation of water vapor.26Fluorescence and UV-vis spectroscopy analyses (SI Appendix, Fig. S2) revealed that the highest fluorescence intensity was achieved at a light intensity of 834 mW cm−2. Further optimization of the reaction duration at this light intensity (SI Appendix, Fig. S3) revealed that 96 h was the optimal duration. The highest-performing sample (Sample 6) exhibited emission peaks at excitation wavelengths of 420–470 nm and 320–370 nm, as characterized by its 3D-EEM fluorescence spectrum (Fig. S3d). The quantum yield of Sample 6 was evaluated as 13.7% (SI Appendix, Table S4).
HRTEM images of Sample 6 revealed CQDs with an average size of 29.8 ± 13.5 nm (Fig. 1a and b). Additionally, larger CQDs (50–100 nm) and irregular carbon sheets with graphite-like structures were observed (Fig. 1c) in the sample. Under UV irradiation (365 nm), the CQDs exhibited blue-green fluorescence (Fig. 1b), despite their size nonuniformity. Their crystal lattice spacing was found to be 0.21 nm, corresponding to the (1![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 00) lattice planes of graphitic carbon, in agreement with prior studies.3 The UV-vis spectrum (SI Appendix, Fig. S3f) displayed peaks at 232 nm and 278 nm, associated with the π → π* transition of C
00) lattice planes of graphitic carbon, in agreement with prior studies.3 The UV-vis spectrum (SI Appendix, Fig. S3f) displayed peaks at 232 nm and 278 nm, associated with the π → π* transition of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds and the n → π* transition of carbonyl groups (C
C bonds and the n → π* transition of carbonyl groups (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), respectively, consistent with previously reported CQDs.
O), respectively, consistent with previously reported CQDs.
The temperature of the aqueous Sample 6 solution reached ∼50 °C at an irradiation intensity of 834 mW cm−2 (SI Appendix, Fig. S3f). To distinguish photothermal effects from photocatalytic contributions, a control experiment (SI Appendix, Table S1, Sample 8) was conducted by heating the solution to 50 °C for 96 h in the absence of light (SI Appendix, Fig. S1c). The HRTEM image (Fig. 1d) revealed that Sample 8 contained larger CQDs (69.4 ± 20.5 nm) (Fig. 1e) without graphite-like structures. Although its UV-vis spectrum (Fig. 1f) showed absorption peaks at 232 nm and 280 nm similar to those of Sample 6, its 3D-EEM fluorescence spectrum (Fig. 1g) exhibited significantly lower fluorescence performance than that of Sample 6 (SI Appendix, Fig. S3d). The CQDs from Sample 8 emitted green fluorescence (Fig. 1e), which is attributed to increased particle size heterogeneity. The quantum yield of Sample 8 was 9.0% (SI Appendix, Table S4). These findings suggest that both thermal effects and photocatalytic reactions, particularly H2O-to-H2O2 conversion, are crucial for CQD formation and optical enhancement.
To further decouple the effects of illumination and oxygen availability, additional control experiments were conducted under dark conditions with O2 (Sample 18) and light conditions with N2 (Sample 19), which are described in Text S6 and Fig. S7. Through their 3D-EEM results, almost no fluorescence (Fig. S7a) was observed in Sample 18, demonstrating that oxygen alone—without illumination-driven interfacial activation—is difficult to generate detectable amounts of CQDs. Conversely, illumination in the absence of a continuous oxygen supply (light with N2, Sample 19) produced CQDs with quite weak fluorescence (Fig. S7b) due to the O2-deficient environment. When considered together with light with the atmospheric environment (Sample 6), light with the O2-rich environment (Sample 10), and dark with external heating (Sample 8) experiments, these results confirm that both illumination and oxygen are simultaneously required for high efficiency of CQD formation.
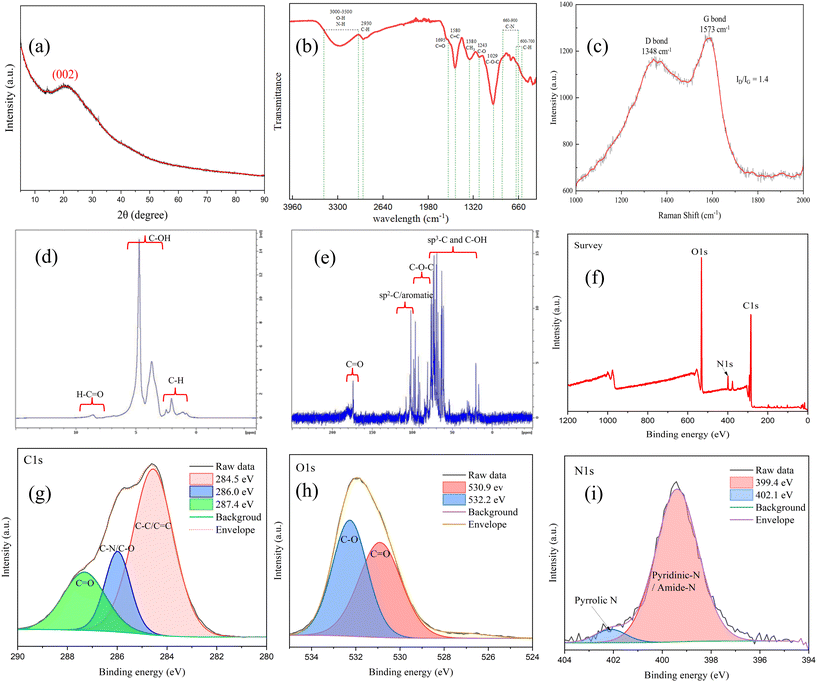
3.2 Factors affecting CQD synthesis with external O2 introduction
Additional experiments were conducted with O2 introduction to synthesize uniform, high-performance CQDs. The effect of O2 flow rate on CQD synthesis was investigated (SI Appendix, Table S1, Samples 9–11). The results (SI Appendix, Fig. S4) show that Sample 10, synthesized at an O2 flow rate of 28 mL min−1, exhibited optimal fluorescence performance. HRTEM images of Sample 10 (Fig. 1h and i) revealed a monodisperse CQD population with an average diameter of 4.6 ± 0.8 nm. In addition, graphite sheets and some graphite-like quantum dots (5–10 nm) were observed with a lattice spacing of 0.21 nm (Fig. 1j), consistent with those observed in Sample 6 (Fig. 1c). Notably, the quantum yield of Sample 10 reached 20.1 ± 0.8% (SI Appendix, Table S4), which is more than double that of Sample 8 (9.0%) synthesized under dark conditions with external heating at the same temperature.The structural characterization of CQDs (Sample 10) synthesized under O2-rich conditions was performed using XRD, ATR-FTIR, Raman spectroscopy, NMR, and XPS. The XRD pattern (Fig. 2a) exhibits a broad diffraction peak at 21°, corresponding to the (002) plane of amorphous graphite (JCPDS: 41-1487), indicating low graphitization.30 ATR-FTIR spectra (Fig. 2b) showed characteristic functional groups, including O–H/N–H (3500–3000 cm−1), C–H (2930 cm−1), C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C (1580 cm−1), C–O (1243 cm−1), and C
C (1580 cm−1), C–O (1243 cm−1), and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (1690–1740 cm−1).31–34 The obtained Raman spectrum (Fig. 2c) was baseline-corrected and fitted using Gauss functions, revealing the characteristic D band (∼1348 cm−1) and G band (∼1573 cm−1) associated with disordered and graphitic sp2 carbon, respectively. An ID/IG ratio of 1.4, calculated based on the integrated peak areas of the fitted bands, indicates a high level of structural disorder relative to the graphitic content.35
O (1690–1740 cm−1).31–34 The obtained Raman spectrum (Fig. 2c) was baseline-corrected and fitted using Gauss functions, revealing the characteristic D band (∼1348 cm−1) and G band (∼1573 cm−1) associated with disordered and graphitic sp2 carbon, respectively. An ID/IG ratio of 1.4, calculated based on the integrated peak areas of the fitted bands, indicates a high level of structural disorder relative to the graphitic content.35
NMR spectroscopy (Fig. 2d and e) shows proton signals at 1–3 ppm (sp3 C–H), 3–6 ppm (oxygenated groups), and 8–10 ppm (aldehydic protons). The 13C NMR spectrum displays signals at 20–80 ppm (sp3 carbon), 80–100 ppm (ether-bound carbon), 100–120 ppm (aromatic carbon), and 170–185 ppm (carboxyl or ester C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O).36,37 XPS analysis (Fig. 2f–i) indicates peaks corresponding to C–C/C
O).36,37 XPS analysis (Fig. 2f–i) indicates peaks corresponding to C–C/C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, C–O/C–N, and C
C, C–O/C–N, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in the C 1s spectrum, while O 1s/N 1s spectra confirmed the presence of oxygen-containing functionalities and pyridinic-N/amide-N species.
O in the C 1s spectrum, while O 1s/N 1s spectra confirmed the presence of oxygen-containing functionalities and pyridinic-N/amide-N species.
These results highlight the synergistic roles of O2 in photothermal and photocatalytic processes, leading to the formation of smaller, more uniform CQDs with enhanced fluorescence. The self-doping of nitrogen from corn stover contributed to a high quantum yield (20.1 ± 0.8%), demonstrating an efficient and sustainable approach to CQD synthesis.12
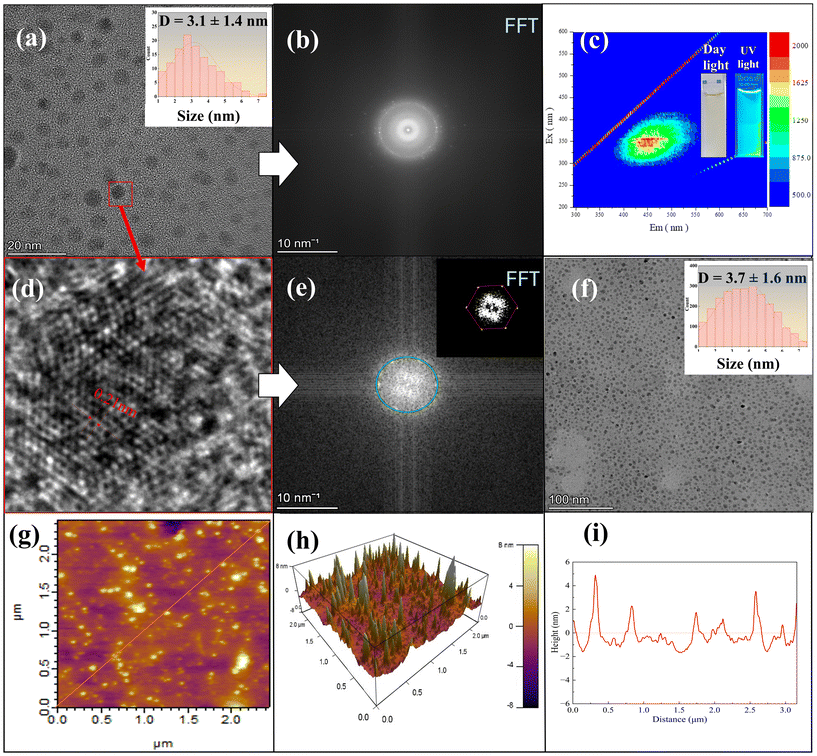
To obtain high-purity CQDs, centrifugation and dialysis were employed (Sample 10) as described in SI Appendix (Text S10). The purity of the CQDs was then evaluated using multiple analytical techniques, including LC-MS, ICP-MS, and IC to detect residual small molecules, metal ions, and anionic species (SI Appendix, Text S11). Fig. 3 presents the comprehensive characterization of CQDs after purification by centrifugation and dialysis. HRTEM images (Fig. 3a and f) reveal that the CQDs are uniformly dispersed, with average diameters of 3.1 ± 1.4 nm and 3.7 ± 1.6 nm, respectively, confirming good size uniformity and monodispersity. The high-resolution image in Fig. 3d shows clear lattice fringes with an interplanar spacing of 0.21 nm, corresponding to the (100) plane of graphitic carbon,2 further supported by the FFT patterns in Fig. 3b and e, indicating partial crystallinity. The 3D-EEM spectrum (Fig. 3c) and accompanying photographs demonstrate strong fluorescence and excellent optical properties of the CQDs under UV light. AFM analysis (Fig. 3g–i) further confirms the nanoscale size and uniform height distribution of individual CQDs, with a height range of ∼3–4 nm and no visible aggregation. TEM analysis (SI Appendix, Fig. S12) of multiple regions confirms that the carbon quantum dots (CQDs) exhibit diameters ranging from approximately 3 to 4 nm, demonstrating that dialysis successfully yielded well-dispersed individual nanoparticles with no observable aggregation. These results confirm that centrifugation and dialysis effectively remove impurities while preserving the structural integrity, size uniformity, and fluorescence properties of the CQDs.
The key synthesis conditions, particle sizes, and quantum yields of representative samples are summarized in Table 1. To clarify how structural features govern photoluminescence (PL), we quantitatively compared particle size, surface chemistry, and graphitic domain structure across samples. TEM analysis (Fig. 1 and 2d) shows that CQDs synthesized under light + rich-O2 (Sample 10) are the smallest (4.6 ± 0.8 nm before purification; 3.1 ± 0.4 nm after dialysis), whereas the oxygen-limited condition (Sample 6) produces much larger particles (29.8 ± 13.5 nm), and the dark–thermal condition (Sample 8) yields even larger amorphous structures (69.4 ± 20.5 nm). Correspondingly, Sample 10 exhibits the strongest PL emission (Fig. S2–S5), consistent with quantum confinement, while samples forming larger particles show weaker and broader emission. FTIR and XPS analyses (Fig. 2b and f–i) indicate that Sample 10 contains abundant oxygen-containing functional groups (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, C–OH, and epoxy), which are known to create radiative surface states that can enhance PL. Raman spectroscopy (Fig. 2c) further shows an ID/IG ratio of 1.4 for Sample 10, indicating defect-rich sp2 nanodomains that are also associated with emissive sites. Together, these results demonstrate that bright PL arises from the combined effects of ultrasmall particle size, oxygen-rich surface states, and defect-engineered sp2 domains formed uniquely under simultaneous illumination and O2 supply.
O, C–OH, and epoxy), which are known to create radiative surface states that can enhance PL. Raman spectroscopy (Fig. 2c) further shows an ID/IG ratio of 1.4 for Sample 10, indicating defect-rich sp2 nanodomains that are also associated with emissive sites. Together, these results demonstrate that bright PL arises from the combined effects of ultrasmall particle size, oxygen-rich surface states, and defect-engineered sp2 domains formed uniquely under simultaneous illumination and O2 supply.
| Sample | Key conditions | Particle size (nm) | Quantum yield (%) |
|---|---|---|---|
| Sample 6 | Light: 834 mW cm−2; atmospheric environment; 96 h | 29.8 ± 13.5 | 13.7 |
| Sample 8 | Dark; heating to 50 °C; atmospheric environment; 96 h | 69.4 ± 20.5 | 9.0 |
| Sample 10 | Light: 834 mW cm−2; rich-O2; 96 h | 4.6 ± 0.8 (after dialysis: 3.1 ± 0.4) | 20.1 ± 0.8 |
3.3 Formation of CQDs from corn stover
The mechanism of CQD formation from natural biomass remains incompletely established.38 In this study, we propose a pathway involving photothermal and photocatalytic oxidation of lignin, hemicellulose, and cellulose in corn stover, primarily driven by Fenton and Fenton-like reactions. This reaction employs in situ-generated H2O2 (SI Appendix, Fig. S1e) and naturally occurring metal ions (SI Appendix, Table S3). The system utilizes an ozone-free xenon lamp, ensuring that unwanted UV wavelengths (<260 nm) are filtered out to prevent ozone formation and associated side reactions.| H2O → H+ + OH− | (R1) |
| OH− → ˙OH + e− | (R2) |
| ˙OH + ˙OH → H2O2 | (R3) |
Another mechanism is electron transfer from OH− to H+ at the interface, followed by ˙OH radical recombination, while both ˙OH and ˙H have been detected experimentally using electron spin resonance (ESR) spectroscopy.41 In addition, contact electrification at the water–solid interface42,43 has been reported to contribute to H2O2 formation.
In the present system, elevated solution temperatures (∼50 °C) under illumination lead to partial water vaporization, followed by continuous condensation at the water–air interface, enabling continuous H2O2 generation (Fig. 4a). H2O2 formation is further enhanced by UV radiation,44 as confirmed using H2O2 test strips (0–100 ppm) (SI Appendix, Fig. S1d).
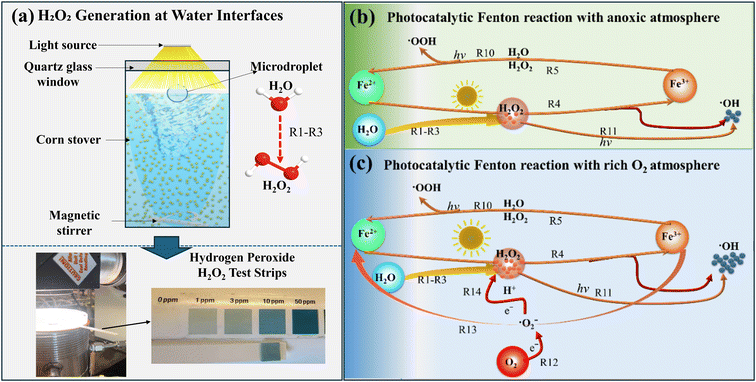 | ||
| Fig. 4 Proposed mechanism of peroxide generation and localized radical activation under illumination conditions. | ||
Building on the foundational discovery that H2O at the hydrophilic–hydrophobic interface yields H2O2,24,25 our approach further integrates simulated solar light to further enhance H2O2 production through photochemical pathways. This strategy synergistically couples interfacial electrochemical processes with photochemical activation, enabling sustained H2O2 generation without the need for external catalysts or energy-intensive UV sources, supporting a scalable and sustainable reaction pathway.
| Fe2+ + H2O2 → Fe3+ + ˙OH + OH− | (R4) |
| Fe3+ + H2O2 → Fe2+ + ˙OOH + H+ | (R5) |
| Fe2+ + ˙OH → Fe3+ + OH− | (R6) |
| H2O2 + ˙OH → ˙OOH + H2O | (R7) |
| Fe2+ + ˙OOH + H+ → Fe3+ + H2O2 | (R8) |
| Fe3+ + ˙OOH → Fe2+ + O2 + H+ | (R9) |
Hydroxyl radicals (˙OH) generated through these reactions oxidatively degrade organic molecules in corn stover, facilitating CQD formation. In addition to iron, other multivalent metal ions (e.g., Co, Cr, Cu, Mn, Mo, Ru, Ti, W, and V), detected by ICP-MS (SI Appendix, Table S3), may participate in Fenton-like reactions, further contributing to reactive oxygen species generation.46
Simulated solar irradiation enhances the Fenton process through photo-induced reactions (Fig. 4b),47 as described using the following equations:
| Fe3+ + H2O + hv → Fe2+ + ˙OH + H+ | (R10) |
| H2O2 + hv → ˙OH + ˙OH | (R11) |
These reactions continuously regenerate Fe2+ and produce additional ˙OH radicals, accelerating oxidative degradation. Experimental results confirm that UV light (250–430 nm) plays a crucial role in CQD synthesis, as evidenced by increased fluorescence intensity and reduced pH, indicating enhanced formation of organic acids (–COOH) during biomass degradation (SI Appendix, Fig. S5 and Table S1).
Molecular oxygen further promotes Fe3+ reduction and reactive oxygen species (ROS) generation,48 as illustrated by the following reactions:
| e− + O2 → ˙O2− | (R12) |
| Fe3+ + ˙O2− → Fe2+ + O2 | (R13) |
| ˙O2− + e− + 2H+ → H2O2 | (R14) |
The resulting superoxide radicals (˙O2−) react with H2O to form additional H2O2, accelerating the photo-Fenton cycle.49 An O2-rich atmosphere consequently enhances the oxidative degradation of hemicellulose, cellulose, and lignin, boosting CQD yield (Fig. 4c). In parallel, organic radical intermediates may undergo peroxo-organic transformations,50,51 further amplifying oxidative pathways and promoting CQD formation.
ESR spin-trapping experiments were carried out to detect ROS formation and peroxide accumulation using DMPO (SI Appendix, Fig. S8). Fig. S8a exhibits a four-line ESR pattern of H2O2 standards measured with Fe2+ triggering. In biomass-free controls (Fig. S8b), illumination and O2 flow induce discernible ESR signals absent under dark conditions, indicating interfacial photochemical radical generation, although the spectra are broadened by overlapping ROS adducts. In the corn-stover-containing system (Fig. S8c), ESR signals are weak and broadened due to the rapid radical consumption and heterogeneous scavenging by biomass-derived organic species and redox-active sites. Notably, Fe2+ addition at extended reaction time (60 min) markedly enhances the ESR response, supporting the accumulation of peroxide species and their involvement in localized, transient radical activation during CQD formation.
3.4 Corn stover conversion to CQDs
Corn stover primarily consists of cellulose (35–40%), hemicellulose (20–25%), lignin (19–22%) and trace metals (e.g., Fe2+).52 Control experiments using isolated cellulose, hemicellulose, and lignin as individual precursors yielded markedly weaker fluorescence under identical reaction conditions (SI Appendix, Fig. S6), displaying that hemicellulose and lignin can be easily degraded and contribute to CQD production.Hemicellulose, a heteropolymer of sugars (xylan and glucan), and lignin, a complex phenylpropanoid network, undergo preferential oxidative degradation in this system, while the proposed pathway of CQD production is shown in Fig. 5. Hydroxyl radicals (˙OH) cleave glycosidic (C–O–C) linkages in polysaccharides and β-O-4 ether bonds, fragmenting these macromolecules into monomeric and oligomeric sugars as well as phenolic compounds (e.g., vanillin, syringol, and guaiacol).53,54 Notably, as-formed CQDs may enhance Fenton reactions by acting as photocatalysts, promoting ˙OH radical production.55–57 Their surface carboxyl functional groups can lower the solution pH, thereby optimizing the reaction efficiency.54 Consistent with this interpretation, the solution pH decreased with prolonged reaction time and higher O2 flow rates, reaching 4.09 under O2-rich conditions (SI Appendix, Table S1, Sample 10), indicating enhanced organic acid formation and controlled H2O2 consumption.
 | ||
| Fig. 5 Proposed pathway for CQD formation from corn stover via peroxide-mediated oxidative depolymerization. | ||
3.5 Comparison with conventional CQD synthesis methods
For comparison, Tables S6–S8 systematically evaluate the reported CQD synthesis methods in terms of raw materials, reaction conditions, CQD sizes, quantum yields, and environmental considerations.Table S6 shows a comparison of CQD synthesis routes involving high-temperature pyrolysis (≥300 °C), two-step carbonization, or hydrothermal methods with added chemicals. These methods often require elevated temperatures (300–700 °C) or strong acids, bases, or oxidants, which can complicate purification and introduce secondary environmental burdens, despite achieving high quantum yields in some cases. Table S7 focuses on one-step hydrothermal and photothermal CQD synthesis using only biomass and water. Compared with hydrothermal processes reported by other groups, the present photothermal method operates at substantially lower temperatures, requires less stringent equipment, and produces fewer liquid organic byproducts, resulting in easier CQD separation and higher product purity. Although several hydrothermal studies report higher quantum yields, these typically rely on higher temperatures or fresh biomass sources rich in soluble carbon, which are more readily degraded than solid lignocellulosic feedstocks. Table S8 shows a comparison of CQD synthesis strategies based on externally added H2O2 and catalysts with those relying on internally generated H2O2. In contrast to methods requiring large H2O2 dosages and added catalysts (e.g., Fe2+/Fe3+ or W18O49), the present approach generates H2O2 in situ without external oxidants or catalysts. This eliminates the need for costly reagents, reduces overoxidation of carbon precursors, enables smaller CQD sizes, and lowers overall energy demand due to the mild reaction temperature.
Overall, this work demonstrates a green and economically attractive CQD synthesis route that combines renewable biomass, internally generated oxidants, ambient-pressure operation, and low thermal input, offering clear advantages over conventional high-temperature or chemically intensive methods.
4. Conclusions
This CQD synthesis strategy minimizes byproducts, simplifies purification, and eliminates harsh acids (e.g., H2SO4 and HNO3). By leveraging corn stover as both a carbon source and a natural catalytic reservoir, it efficiently integrates spontaneous H2O-to-H2O2 conversion under solar irradiation, providing an environmentally sustainable route for high-purity CQD production. These results extend microdroplet chemistry into the realm of biomass valorization and highlight a sustainable, external-catalyst-free strategy for converting raw biomass into high-quality nanocarbon materials.Author contributions
M. Fan, X. Kang, and R. N. Zare designed the research concepts; X. Kang performed the major research tasks, including tests and data analyses, and prepared the initial draft of the manuscript. Q. Yang, S. X. Tang, Z. Chen, S. Budhathoki, R. Paneru, S. Kim and A. Goroncy assisted X. Kang in performing the research tasks. Y. Bai, Qian Li, and Z. Chen assisted M. Fan, X. Kang, and R. N. Zare in developing the research concepts. X. Kang, M. Fan, and R. N. Zare worked together to finalize the manuscript.Conflicts of interest
The authors declare no conflicts of interest.Data availability
All data supporting the findings of this study are available within the paper and its supplementary information (SI). Supplementary information: Text S1–S11, Fig. S1–S12 and Tables S1–S8 provide additional experimental details and analyses, including the effects of light intensity, reaction time, oxygen flow, light wavelength ranges, and separated corn stover components, as well as descriptions of Fenton and Fenton-like reactions, thermogravimetric analysis, ESR spectra, and purification protocols with purity evaluation. Characterization data (TEM, 3D-EEM, TGA, LC-MS, ICP-MS, IC, and pH), together with tables containing comparisons to conventional CQD synthesis methods as well as their reported quantum yields of CQDs, are also provided. See DOI: https://doi.org/10.1039/d5gc04545b.Acknowledgements
We would like to thank Zhaojie Zhang for the help with fluorescence microscopy measurements. This work was supported by the University of Wyoming and the Czech University of Life Sciences Prague.References
- S. Y. Lim, W. Shen and Z. Gao, Chem. Soc. Rev., 2015, 44, 362–381 RSC.
- W. Li, W. Zhou, Z. Zhou, H. Zhang, X. Zhang, J. Zhuang, Y. Liu, B. Lei and C. Hu, Angew. Chem., Int. Ed., 2019, 131, 7356–7361 CrossRef.
- L. Wang, W. Li, L. Yin, Y. Liu, H. Guo, J. Lai, Y. Han, G. Li, M. Li, J. Zhang, R. Vajtai, P. M. Ajayan and M. Wu, Sci. Adv., 2020, 6, eabb6772 CrossRef CAS PubMed.
- Y. Li, X. Zhang, W. Hou, Z. Zhou, S. Zhang, H. Guo, J. Zhang, J. Xu and L. Wang, Appl. Catal., B, 2026, 380, 125758 CrossRef CAS.
- X. Xu, R. Ray, Y. Gu, H. J. Ploehn, L. Gearheart, K. Raker and W. A. Scrivens, J. Am. Chem. Soc., 2004, 126, 12736–12737 CrossRef CAS PubMed.
- R. Liu, D. Wu, S. Liu, K. Koynov, W. Knoll and Q. Li, Angew. Chem., Int. Ed., 2009, 48, 4598–4601 CrossRef CAS PubMed.
- L. Cao, X. Wang, M. J. Meziani, F. Lu, H. Wang, P. G. Luo, Y. Lin, B. A. Harruff, L. M. Veca, D. Murray, S. Y. Xie and Y. P. Sun, J. Am. Chem. Soc., 2007, 129, 11318–11319 CrossRef CAS PubMed.
- A. Xu, G. Wang, Y. Li, H. Dong, S. Yang, P. He and G. Ding, Small, 2020, 16, 2004621 CrossRef CAS PubMed.
- S. Chen, S. Z. Zhang and H. Jiang, ACS ES&T Water, 2024, 4, 531–542 Search PubMed.
- S. N. Baker and G. A. Baker, Angew. Chem., Int. Ed., 2010, 49, 6726–6744 CrossRef CAS PubMed.
- S. Chung, R. A. Revia and M. Zhang, Adv. Mater., 2021, 33, 1904362 CrossRef CAS PubMed.
- R. Paneru, X. Kang, S. Budhathoki, Z. Chen, Q. Yang, S. T. Tjeng, Q. Dai, W. Wang, J. Tang and M. Fan, J. Environ. Sci., 2025, 154, 590–601 CrossRef CAS PubMed.
- H. Ding, S. B. Yu, J. S. Wei and H. M. Xiong, ACS Nano, 2016, 10, 484–491 CrossRef CAS PubMed.
- B. Dutcher, M. Fan, B. Leonard, M. D. Dyar, J. Tang, E. A. Speicher, P. Liu and Y. Zhang, J. Phys. Chem. C, 2011, 115, 15532–15544 CrossRef CAS.
- M. Yang, L. Guo, G. Hu, X. Hu, J. Chen, S. Shen, W. Dai and M. Fan, Ind. Eng. Chem. Res., 2016, 55, 757–765 CrossRef CAS.
- P. M. Gawal and A. K. Golder, Colloids Surf., A, 2024, 683, 133068 CrossRef CAS.
- T. J. Pillar-Little, N. Wanninayake, L. Nease, D. K. Heidary, E. C. Glazer and D. Y. Kim, Carbon, 2018, 140, 616–623 CrossRef CAS.
- D. Xu, C. Yu, X. Peng, H. Yan and Y. Zhang, Res. Chem. Intermed., 2024, 50, 4597–4617 CrossRef CAS.
- V. G. Matveeva and L. M. Bronstein, Prog. Mater. Sci., 2022, 130, 100999 CrossRef CAS.
- S. C. Perry, D. Pangotra, L. Vieira, L. I. Csepei, V. Sieber, L. Wang, C. Ponce de León and F. C. Walsh, Nat. Rev. Chem., 2019, 3, 442–458 CrossRef CAS.
- X. Zhou, Y. Zhang, C. Wang, X. Wu, Y. Yang, B. Zheng, H. Wu, S. Guo and J. Zhang, ACS Nano, 2012, 6, 6592–6599 CrossRef CAS PubMed.
- C. Zhu, S. Yang, G. Wang, R. Mo, P. He, J. Sun, Z. Di, Z. Kang, N. Yuan, J. Ding, G. Ding and X. Xie, J. Mater. Chem. B, 2015, 3, 6871–6876 RSC.
- D. Y. Lee, Z. Haider, S. K. Krishnan, T. Kanagaraj, S. H. Son, J. Jae, J. R. Kim, P. S. M. Kumar and H. I. Kim, Chemosphere, 2024, 361, 142330 CrossRef CAS PubMed.
- M. A. Mehrgardi, M. Mofidfar and R. N. Zare, J. Am. Chem. Soc., 2022, 144, 7606–7609 CrossRef CAS PubMed.
- J. K. Lee, K. L. Walker, H. S. Han, J. Kang, F. B. Prinz, R. M. Waymouth, H. G. Nam and R. N. Zare, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 19294–19298 CrossRef CAS PubMed.
- J. K. Lee, H. S. Han, S. Chaikasetsin, D. P. Marron, R. M. Waymouth, F. B. Prinz and R. N. Zare, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 30934–30941 CrossRef CAS PubMed.
- X. Song, C. Basheer, J. Xu and R. N. Zare, Sci. Adv., 2024, 10, 4443 CrossRef PubMed.
- Y. Meng, Y. Xia, J. Xu and R. N. Zare, Sci. Adv., 2025, 11, 8979 CrossRef PubMed.
- H. Kalita, J. Mohapatra, L. Pradhan, A. Mitra, D. Bahadur and M. Aslam, RSC Adv., 2016, 6, 23518–23524 RSC.
- A. Afzalalghom, A. Beitollahi, S. M. Mirkazemi, M. Maleki and H. Sarpoolaky, Small, 2024, 20, 2308082 CrossRef CAS PubMed.
- B. Al-Hashimi, K. M. Omer and H. S. Rahman, Arabian J. Chem., 2020, 13, 5151–5159 CrossRef CAS.
- N. B. Mandava, in Handbook of Natural Pesticides: Methods, CRC Press, 2019, pp. 219–256 Search PubMed.
- H. Nie, M. Li, Q. Li, S. Liang, Y. Tan, L. Sheng, W. Shi and S. X. A. Zhang, Chem. Mater., 2014, 26, 3104–3112 CrossRef CAS.
- S. Moonrinta, B. Kwon, I. In, S. Kladsomboon, W. Sajomsang and P. Paoprasert, Opt. Mater., 2018, 81, 93–101 CrossRef CAS.
- A. C. Ferrari and D. M. Basko, Nat. Nanotechnol., 2013, 8, 235–246 CrossRef CAS PubMed.
- S. Zhu, Q. Meng, L. Wang, J. Zhang, Y. Song, H. Jin, K. Zhang, H. Sun, H. Wang and B. Yang, Angew. Chem., Int. Ed., 2013, 52, 4045–4049 CrossRef.
- B. De and N. Karak, RSC Adv., 2013, 3, 8286–8290 RSC.
- M. Si, J. Zhang, Y. He, Z. Yang, X. Yan, M. Liu, S. Zhuo, S. Wang, X. Min, C. Gao, L. Chai and Y. Shi, Green Chem., 2018, 20, 3414–3419 RSC.
- C. Pan, G. Bian, Y. Zhang, Y. Lou, Y. Zhang, Y. Dong, J. Xu and Y. Zhu, Appl. Catal., B, 2022, 316, 121675 CrossRef CAS.
- T. Liu, Z. Pan, J. J. M. Vequizo, K. Kato, B. Wu, A. Yamakata, K. Katayama, B. Chen, C. Chu and K. Domen, Nat. Commun., 2022, 13, 1034 CrossRef CAS PubMed.
- J. Li, Y. Xia, X. Song, B. Chen and R. N. Zare, Proc. Natl. Acad. Sci. U. S. A., 2024, 121, e2318408121 CrossRef CAS PubMed.
- A. Gallo Jr., N. H. Musskopf, X. Liu, Z. Yang, J. Petry, P. Zhang, S. Thoroddsen, H. Im and H. Mishra, Chem. Sci., 2022, 13, 2574–2583 RSC.
- B. Chen, Y. Xia, R. He, H. Sang, W. Zhang, J. Li, L. Chen, P. Wang, S. Guo, Y. Yin, L. Hu, M. Song, Y. Liang, Y. Wang, G. Jiang and R. N. Zare, Proc. Natl. Acad. Sci. U. S. A., 2022, 119, e2209056119 CrossRef CAS PubMed.
- B. C. Faust, C. Anastasio, J. M. Allen and T. Arakaki, Science, 1993, 260, 73–75 CrossRef CAS PubMed.
- J. De Laat and H. Gallard, Environ. Sci. Technol., 1999, 33, 2726–2732 CrossRef CAS.
- Y. Liu and J. Wang, Chem. Eng. J., 2023, 466, 143147 CrossRef CAS.
- J. Kiwi, C. Pulgarin and P. Peringer, Appl. Catal., B, 1994, 3, 143147 CrossRef.
- S. Chen, Y. Chen, Y. Zhang, X. Kuang, Y. Liu, M. Guo, L. Ma, D. Zhang and Q. Li, Front. Neurosci., 2020, 14, 601193 CrossRef PubMed.
- P. Zhou, J. Xu, J. Guo, X. Hou, L. Dai, X. Xiao and K. Huo, Green Chem., 2024, 26, 6005–6018 RSC.
- O. Legrini, E. Oliveros and A. M. Braun, Chem. Rev., 1993, 93, 671–698 CrossRef CAS.
- B. Utset, J. Garcia, J. Casado, X. Domènech and J. Peral, Chemosphere, 2000, 41, 1187–1192 CrossRef CAS PubMed.
- A. H. Rony, L. Kong, W. Lu, M. Dejam, H. Adidharma, K. A. M. Gasem, Y. Zheng, U. Norton and M. Fan, Bioresour. Technol., 2019, 284, 466–473 CrossRef CAS PubMed.
- D. Van Eylen, F. van Dongen, M. Kabel and J. de Bont, Bioresour. Technol., 2011, 102, 5995–6004 CrossRef CAS PubMed.
- E. Mnich, N. Bjarnholt, A. Eudes, J. Harholt, C. Holland, B. Jørgensen, F. H. Larsen, M. Liu, R. Manat, A. S. Meyer, J. D. Mikkelsen, M. S. Motawia, J. Muschiol, B. L. Møller, S. R. Møller, A. Perzon, B. L. Petersen, J. L. Ravn and P. Ulvskov, Nat. Prod. Rep., 2020, 37, 919–961 RSC.
- T. Zhang, Y. Wen, Z. Pan, Y. Kuwahara, K. Mori, H. Yamashita, Y. Zhao and X. Qian, Environ. Sci. Technol., 2022, 56, 2617–2625 CrossRef CAS PubMed.
- Y. Yang, Q. Guo, Q. Li, L. Guo, H. Chu, L. Liao, X. Wang, Z. Li and W. Zhou, Adv. Funct. Mater., 2024, 34, 2400612 CrossRef CAS.
- W. Han, H. Zhang, D. Li, W. Qin, X. Zhang, S. Wang and X. Duan, Appl. Catal., B, 2024, 350, 123918 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |