Open Access Article
Open Access ArticlePlant proteins for human health: the current status and future needs
Kun Gaoab,
Shiyu Zhoub,
Jiajia Rao b and
Bingcan Chen
b and
Bingcan Chen *b
*b
aKey Laboratory of Jianghuai Agricultural Product Fine Processing and Resource Utilization of Ministry of Agriculture and Rural Affairs, School of Food and Nutrition, Anhui Agricultural University, Hefei, 230036, China
bDepartment of Plant Sciences, North Dakota State University, Fargo, ND 58108, USA. E-mail: bingcan.chen@ndsu.edu
First published on 21st April 2026
Abstract
Plant based protein consumption is increasingly recognized for its therapeutic potential in managing metabolic health and preventing chronic diseases. This review provides a comprehensive analysis of the physiological impact of plant proteins, including their roles in satiety regulation and weight management via the modulation of appetite regulating hormones. We examine how plant proteins optimize lipid metabolism and reinforce gut homeostasis by promoting diverse microbiota and increasing the production of short chain fatty acids. Furthermore, we dissect the mechanisms through which plant proteins and their digestion derived peptides attenuate the pathogenesis of cardiovascular disease, type 2 diabetes, and chronic kidney disease. Specific attention is given to the modulation of intracellular signaling pathways such as PI3K-Akt and the regulation of the renin angiotensin system. The review also highlights that the health efficacy of plant proteins is highly dependent on the food matrix, where synergistic interactions between proteins, fiber, and phytochemicals are critical. Finally, the impact of food processing on peptide bioaccessibility is examined, and a process–matrix function paradigm is proposed for future research. In conclusion, these insights underscore the role of plant proteins as functional components that are essential for developing sustainable and precise nutritional strategies to mitigate the global burden of non-communicable diseases.
1. Introduction
Proteins are indispensable macronutrients that provide essential amino acids and serve as structural and functional components of human tissues, supporting growth, repair, and metabolic homeostasis. The 2025–2030 Dietary Guidelines for Americans has significantly increased the recommended protein intake from the previous 0.8 g kg−1 standard to 1.2 to 1.6 grams of protein per kg of body weight.1 In general, humans can obtain dietary proteins from a wide range of sources, including animal, dairy, plant, microbial, marine, and insect origins. With increasing environmental and sustainability concerns, a marked shift in consumer preferences toward proteins derived from plants has been observed. Traditionally, plant proteins are central to the food structure and functionality, governing the formation, stability, and sensory properties of many food systems. For instance, wheat gluten confers viscoelasticity to dough systems, whereas soy proteins exhibit strong gelation and water-holding capacity, properties that are widely exploited in structured plant-based foods.2 More recently, the structural potential of plant proteins has been extended to advanced colloidal system development, such as hydrogels, oleogels, and hybrid bi-gels, which enable precise modulation of the texture, fat structuring, and digestion behavior.3 These plant protein-based networks hold significant promise for developing healthier fat-reduced foods and designing next-generation plant-based products with tailored functional properties.Plant proteins are derived from a wide range of botanical sources, which are commonly classified into four major categories: legumes, cereals, oilseeds, and emerging novel sources. Legume proteins, such as those from soy, pea, and lentil, are among the most extensively studied due to their relatively high protein content and distinctive amino acid composition. Cereal proteins, including wheat, rice, and maize, constitute a major proportion of global dietary protein intake and are closely associated with staple food matrices that determine both technological functionality and nutritional outcomes.4 Oilseed proteins, obtained primarily from defatted meals of crops such as peanut, rapeseed, and sunflower, represent an underutilized yet protein-rich resource with diverse structural characteristics.5 Beyond these traditional sources, increasing attention has been directed toward novel plant proteins from leafy biomass, reflecting the growing interest in expanding the diversity and sustainability of plant-based protein supplies. These protein sources exhibit substantial heterogeneity in the composition, structure, and functionality, underscoring the need to consider the protein origin when evaluating their nutritional and health-related roles.6
Plant proteins, particularly those derived from legumes such as soy and peas, provide broad health-promoting benefits primarily mediated by their unique amino acid composition and synergistic food matrix effects.7 At the protein level, plant proteins provide diverse amino acid profiles and encrypted bioactive sequences that have been associated with improved metabolic regulation, cardiometabolic risk factors, and inflammatory status. Substitution of animal protein with plant protein sources has consistently been linked to favorable outcomes in body weight control, blood lipid profiles, and glycemic regulation, supporting their relevance in the prevention and management of chronic diseases such as cardiovascular disease, diabetes mellitus, and chronic kidney disease.8 In addition to the protein fraction itself, increasing attention has been directed toward the synergistic role of the plant food matrix, in which proteins coexist with dietary fiber, phytochemicals, and other bioactive constituents. For instance, in soy-based foods, dietary fiber and associated phytochemicals such as isoflavones work together to regulate satiety signals, manage energy balance, and modulate the gut microbiota composition through prebiotic activities. Together, current evidence suggests that the health impact of plant proteins is multifactorial and context-dependent, highlighting the importance of considering both the protein source and food matrix when evaluating their role in human nutrition.
This review aims to consolidate current knowledge on the relationships between plant proteins and human health. In particular, the roles of plant proteins in satiety, weight management, and muscle protein synthesis, as well as their effects on lipid metabolism and gut health are systematically discussed. Evidence regarding the potential of plant proteins in the prevention and management of non-communicable diseases, particularly cardiovascular disease, diabetes mellitus, and chronic kidney disease, is critically evaluated. Finally, this review highlights key research priorities, including the comparative efficacy of different plant protein sources, the role of co-existing constituents within plant food matrices, and the bioactivity of protein-derived peptides released during digestion.
2. Satiety and weight management
Foods rich in fat, dietary fiber, and protein are generally associated with prolonged satiety compared with those rich in rapidly digestible carbohydrates. Among these macronutrients, dietary protein plays a particularly prominent role in appetite regulation, with consistent effects observed across acute feeding studies as well as short- to medium-term dietary interventions. As illustrated in Fig. 1, protein-induced satiety, including that by plant proteins, has been widely attributed to coordinated changes in gastrointestinal and endocrine signaling, including increased secretion of anorexigenic hormones such as glucagon-like peptide-1 (GLP-1), cholecystokinin (CCK), and peptide YY (PYY), alongside suppression of the orexigenic hormone ghrelin.9 These hormonal responses are thought to contribute to reduced hunger perception and enhanced postprandial fullness, although their translation into subsequent energy intake remains variable.Energy expenditure has also been proposed as a contributor to protein-induced satiety, particularly under sustained high-protein dietary conditions, and to a lesser extent following single high-protein meals. While it was previously thought that this mechanism might be less pronounced for plant-based sources, recent evidence suggests plant proteins can effectively modulate energy intake without necessarily altering systemic thermogenesis. For instance, a 50 g dose of soy protein was found to significantly enhance subjective satiety and reduce subsequent energy intake, despite no significant differences in diet-induced thermogenesis compared to that of carbohydrate controls.10 Crucially, the role of plant proteins in supporting muscle protein synthesis is a vital but often overlooked component of weight management. By providing the essential amino acid substrates to maintain metabolically active lean mass, plant proteins help prevent the decline in the basal metabolic rate typically observed during weight loss, thereby supporting long-term energy homeostasis.
The satiety-inducing capacity of plant proteins is further influenced by postprandial metabolites and specific amino acid profiles. High-protein soy-based meals (25% of energy) have been shown to elicit greater satiety ratings compared to their normal-protein counterparts, an effect associated with elevated concentrations of metabolites like taurine and increased insulin responses.11 However, the satiety capacity is not uniform across all botanical sources. Specific intervention studies highlighted that pea protein (e.g., NUTRALYS®) often elicits stronger acute appetite suppression and more pronounced hormonal responses (e.g., CCK and PYY) compared to soy protein. This superiority in acute satiety is likely driven by its distinct amino acid profile and its tendency to form transient aggregates during gastric digestion, which delays nutrient emptying more effectively than the relatively faster-digesting soy protein.12 Furthermore, fortifying foods with specific amino acids, such as L-arginine in legume-based cookies, have been demonstrated to strengthen insulinemic responses and satiety hormone release, highlighting the potential for targeted amino acid modulation to enhance the functional properties of plant-based foods.13
Protein digestibility is another key determinant of physiological function. Many plant proteins exhibit slower digestion kinetics due to their inherent globular structures and the presence of antinutritional factors. This leads to a more sustained release of amino acids, as exemplified by pea protein that forms transient aggregates during gastric digestion. This results in prolonged aminoacidemia, extending the activation of central nervous system pathways involved in appetite control.14 From an anabolic perspective, while the “slow” nature of some plant proteins might result in a lower peak in muscle protein synthesis compared to rapidly digested whey, the sustained amino acid supply may be advantageous for maintaining net protein balance over a longer postprandial period. Moreover, food processing, such as extrusion, can decouple the relationship between digestibility and satiety. Texturized vegetable protein (TVP) has been shown to induce lower energy intake than meat, despite lower in vitro digestibility, suggesting that the physical structure created during processing may prolong gastric retention.15
A critical factor that has long been underestimated is the impact of the food matrix which may ultimately be more crucial than the origin of protein alone. Dietary fiber, a major constituent of plant protein, contributes to satiety through gastric distention, prolonged CCK secretion, and the production of short-chain fatty acids (SCFAs) via colonic fermentation. Legume-based meals (beans/peas) have been shown to induce 12–13% lower subsequent energy intake compared to isoenergetic animal-based meals, largely due to this fiber-protein synergy.16 Additionally, certain antinutritional compounds like lectins and trypsin inhibitors may stimulate CCK secretion though their retention must be balanced against their potential to limit the bioavailability of amino acids needed for muscle protein synthesis. Optimized processing, such as the combination of pea protein and fiber in extruded cereals, can effectively manage glycemic and insulinemic responses while preserving the satiety-enhancing components of the matrix.17
In short, plant proteins facilitate weight management through a multi-pathway approach involving the regulation of energy intake via acute satiety signaling and the support of metabolic health through muscle mass preservation (Fig. 1). The existing evidence highlights a complex relationship between internal satiety signals and actual eating behavior, which suggests that future weight management strategies should move beyond acute hormonal responses. A critical area for advancement lies in optimizing food processing techniques to balance protein digestibility with the retention of bioactive matrix components like dietary fiber and the elimination of antinutrients, particularly those that reduce the digestibility of plant protein. By tailoring the physical structure of plant-based foods to prolong gastric retention or modulate nutrient release, it may be possible to develop functional plant protein-based foods that provide more sustained appetite suppression. Furthermore, integrating these mechanistic insights into long-term dietary interventions will be essential to validate the efficacy of plant proteins in achieving sustainable weight loss and improving body composition in diverse populations. Understanding these integrated relationships is fundamental for the rational design of next-generation plant protein-based products that can effectively address the global challenges of obesity.
3. Lipid metabolism
Lipid metabolism encompasses the complex processes governing the digestion, absorption, transport, and utilization of fats within the body. Plant proteins influence these pathways through a dual mechanism involving the modulation of initial lipid bioaccessibility in the gastrointestinal tract and the direct biochemical regulation of systemic lipid synthesis and catabolism.18 These complementary pathways, supported by principles of food structure science and molecular biology, position plant protein consumption as a key dietary intervention for improving lipid profiles and metabolic health. The effects of different plant protein sources and forms on lipid metabolism together with their proposed mechanisms and experimental evidence are summarized in Table 1.| Plant protein source | Form | Evidence types | Key lipid metabolic pathway | Lipid outcome | Ref. |
|---|---|---|---|---|---|
| Pea and lupin protein | Emulsion-stabilizing proteins | In vitro digestion model | Protein-stabilized interfacial layers hinder lipase adsorption and lipid hydrolysis | Decreased lipid digestibility and bioaccessibility | 18 |
| Soy protein | Intact protein isolate | Meta-analysis of 46 RCTs in adults | Increased bile acid excretion, reduced cholesterol absorption, altered hepatic cholesterol metabolism | Decreased LDL-C and total cholesterol | 25 |
| Chickpea protein; lentil protein | Purified intact protein | Wistar rats (28-day feeding) | Reduced hepatic TG synthesis and VLDL export; decreased adipose tissue LPL activity; increased hepatic lipase activity | Decreased plasma TG, VLDL particle number and TG content, liver TG and cholesterol | 20 |
| Soy protein | Intact dietary proteins | High-fat diet-fed mice | Regulation of AMPK/mTOR signaling and gut microbiota-lipid metabolism interactions | Improved lipid profile and insulin sensitivity | 33 |
| Red bean protein concentrate | Protein concentrate (with associated phytochemicals) | In vitro antioxidant assays | Antioxidant activity limiting oxidative damage to lipids | Decreased lipid peroxidation | 22 |
| Rapeseed meal protein | Enzymatic hydrolysates (bioactive peptides) | In vitro antioxidant and lipid oxidation models | Metal chelation and radical scavenging by low-molecular-weight peptides | Decreased lipid oxidation in model systems | 23 |
| Rapeseed protein | Purified bioactive peptide (EFLELL) | Cell-based assays + molecular docking | Modulation of LDLR-PCSK9 signaling; inhibition of PCSK9-LDLR interaction | Decreased TC, TG, and LDL-C | 21 |
| Pea protein | Protein hydrolysate | GDM mouse model | Inhibition of PI3K/AKT/mTOR/PPARγ signaling; restoration of PC, PE, and FAHFA species | Decreased TG, TC, LDL-C, hepatic and placental lipid accumulation; improved placental lipid profile | 17 |
| Pea protein | Albumin | OA-induced LO2 hepatocyte model | Inhibition of lipid synthesis; activation of triglyceride catabolism and fatty acid oxidation pathways | Decreased intracellular TG and TC, lipid droplet accumulation, ALT, AST, MDA; improved GSH-Px | 26 |
Among them, a primary pathway is the ability of plant proteins to physically govern lipid digestion and absorption. Their functionality as emulsifiers and gelation agents within the food matrix actively determines lipid bioaccessibility including the fraction of digested lipids available for intestinal uptake. For instance, Keuleyan et al. found that lipid bioaccessibility was significantly lower in emulsions stabilized with lupin protein ingredients compared to pea protein isolates, while their lipolysis rates were similar.19 Beyond acting as a physical barrier, the molecular interactions between plant proteins and lipid species further dictate metabolic outcomes through the binding of cholesterol and bile acids. Mechanistic disparities are also evident among cereal proteins. While wheat proteins are primarily valued for their techno-functional roles in food matrices, oat-derived protein hydrolysates exhibit a significant biochemical capacity to bind bile acids such as taurocholate, with values reaching 46.3% following digestion. This sequestration of bile acids disrupts enterohepatic circulation and promotes hepatic cholesterol conversion, a mechanism that is far more pronounced in oats than in traditional wheat-based proteins.20 By promoting the fecal excretion of these bile acids, plant proteins disrupted enterohepatic circulation and stimulated the hepatic conversion of cholesterol into new bile acids.
Furthermore, plant proteins provide essential protection against lipid oxidation during the complex process of digestion. The gastric environment can promote the formation of pro-oxidant species that lead to the generation of toxic lipid hydroperoxides and aldehydes. Many plant-derived proteins and their hydrolyzed peptides possess inherent antioxidant properties such as radical scavenging and metal chelating activities.21,22 Lucrecia et al. demonstrates that while the intact red bean protein concentrate showed limited activity, their gastric and duodenal digests significantly inhibited lipid peroxidation. Specifically, red bean protein concentrate digests was able to inhibit lipid oxidation by up to 93% in vitro and maintained high efficacy in vivo using zebrafish models.23 Similar protective mechanisms have been reported for rapeseed meal protein hydrolysates produced by controlled enzymatic proteolysis, which exhibited strong reducing power and pronounced metal-chelating activity, particularly in Prolyve-derived (Bacillus licheniformis proteinases) peptide fractions, thereby effectively limiting lipid oxidation in model systems.24 These findings were also complemented by studies on oat bran protein hydrolysates that similarly protected human low-density lipoprotein (LDL) against copper-mediated lipid oxidation by reducing the concentration of lipid hydroperoxides.20
Beyond intestinal protection, plant proteins can be engineered into delivery systems that temporally control lipid release. For example, Browning et al. have developed stable microcapsules from pea protein isolate that protect lipid cargo (e.g., oils) under gastric conditions and release it in the intestine.25 This strategy can delay and attenuate postprandial lipid absorption to smooth the metabolic load. By reducing the rate and extent of lipid absorption, plant protein-based matrices can lower chylomicron output and alleviate the postprandial lipemic burden on the liver. The consistent reduction in LDL cholesterol observed in human trials by substituting animal protein with plant protein is congruent with a mechanism that reduces the efficient absorption of dietary cholesterol and saturated fats.26,27
In addition to their intrinsic nutritional value, plant proteins exhibit important synergistic interactions with unsaturated fatty acids (UFAs), particularly in the regulation of lipid digestion and postprandial lipid metabolism. Plant proteins, especially in their native or mildly processed forms, can act as natural emulsifiers and adsorb at the oil–water interface to form viscoelastic interfacial layers around lipid droplets. These protein-stabilized interfaces create steric and electrostatic barriers that hinder the adsorption and activity of pancreatic lipase and bile salts, thereby slowing the hydrolysis of triglycerides and reducing lipid bioaccessibility.28,29 This interfacial modulation mechanism leads to a more gradual release of free fatty acids during digestion, which may contribute to the attenuation of postprandial lipemia and improved lipid metabolic responses. In addition, the structural properties of plant proteins, including their aggregation state, flexibility, and surface hydrophobicity, can further influence emulsion stability and digestion kinetics, highlighting the importance of protein physicochemical characteristics in determining lipid digestion behavior.
Parallel to physical modulation in the gut, plant proteins and their derivatives enact direct biochemical effects on endogenous lipid metabolism, primarily in the liver. Upon digestion, plant proteins release bioactive peptides that function as endogenous metabolic regulators. A well-documented mechanism is the inhibition of hepatic 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA) reductase, the rate-limiting enzyme in cholesterol synthesis, by peptides derived from soy, quinoa, and beans.30,31 Concurrently, certain peptides have been shown to upregulate the hepatic LDL receptor expression, enhancing the clearance of circulating LDL cholesterol.26 These peptides thus operate dually, i.e., suppressing de novo cholesterol production while accelerating its removal.
The benefits of plant proteins are further amplified by the gut microbiome through the gut-liver axis. Fermentation of co-existing dietary fiber produces short-chain fatty acids (SCFAs) like propionate, which function as signaling molecules. In hepatic cells, SCFAs activate the AMP-activated protein kinase (AMPK) pathway. This leads to the upregulation of peroxisome proliferator-activated receptor alpha (PPARα), a master regulator that enhances fatty acid β-oxidation and suppresses lipogenesis.32 This gut-liver axis represents a critical indirect pathway for plant proteins to improve lipid homeostasis. For instance, Ji and coworkers found that supplementation with soy protein isolate (SPI) in mice fed with a high-fat diet not only activated hepatic AMPK signaling but also improved systemic insulin sensitivity and lipid metabolism. Furthermore, SPI positively remodeled the gut microbiota by increasing its diversity and reducing potentially harmful genera, which demonstrated how a single plant protein intervention can simultaneously engage both the hepatic AMPK pathway and the gut microbial ecosystem to restore metabolic homeostasis.33
Recent advances in lipidomics have provided deeper insight into how dietary patterns including the consumption of plant proteins modulate lipid metabolism at the molecular level. A large-scale lipidomic study in 2860 adults demonstrated that higher protein intake was inversely associated with multiple sphingolipid subclasses, including ceramides, hexosylceramides, and sphingomyelins. However, the opposite trend was observed in a higher saturated fat intake which increased several sphingolipid species and the associated cardiometabolic risk.34 These findings imply that plant protein-rich diets may induce favorable modifications in both classical lipid markers and deeper molecular lipid species. However, despite these promising associations, current understanding remains constrained by several key limitations. In particular, interventional studies combining plant protein dietary shifts with comprehensive lipidomics profiling remain scarce, and most mechanistic evidence concerning bioactive peptides and gut microbiota-derived metabolites (e.g., SCFAs) is still derived from in vitro animal models, with the human data and clear quantification of individual variability in response notably lacking. Future research must therefore prioritize well-controlled human interventions that integrate multi-omics approaches to bridge these gaps, confirm causality, and translate the compelling mechanistic insights into validated, personalized plant protein based dietary strategies for effective lipid management.4
4. Gut health
The human gut microbiota is a highly diverse and dynamic ecosystem that plays an essential role in host digestion, immune regulation, and metabolic homeostasis. Plant proteins possess a distinctive capacity to interact with this microbial community, which is partly due to their generally lower digestibility in the small intestine compared to animal-derived proteins. Consequently, a greater proportion of undigested proteins and peptides reaches the colon, where they serve as substrates for microbial fermentation.35 The extent of this interaction is shaped by the intrinsic structural traits of plant protein source, such as the high hydrophobicity and trypsin inhibitor content found in soy protein.To improve the gastrointestinal fate of plant proteins and modulate their impact on the gut health, a variety of processing strategies have been developed to reduce structural barriers, inactivate anti-nutritional factors, and enhance upper gastrointestinal digestibility. Thermal treatments like extrusion and biological strategies such as fermentation are widely applied for this purpose.36 Comparative studies have indicated that while fermentation primarily improves sensory profiles and reduces antinutrients, its impact on digestibility can be limited compared to thermomechanical methods. Specifically, high-moisture extrusion (HME) has been shown to increase the degree of protein hydrolysis in sunflower-pea blends to 52.8%, significantly outperforming mild heating. This is because HME's combined thermal and mechanical shear more effectively uncoils globular proteins, though excessive heat may still lead to insoluble aggregates that increase the load of resistant protein in the colon.37 While fermentation may differ in its impact on initial hydrolysis, it remains a powerful tool for improving nutritional availability. For example, solid-state fermentation of red kidney beans has been shown to significantly elevate free amino acid levels and increase peptide production during subsequent digestion.38 A mechanistic in vitro study by Kim et al. further demonstrated that probiotic fermentation of pea protein reduced poorly digestible β-sheet structures by 53.92% and improved the overall digestion by 22.50%.39 Meanwhile, in vivo evidence also aligns with these findings, as Jäger et al. reported that consuming 20 g of pea protein together with five billion colony-forming units each of Lactobacillus paracasei LP-DG (CNCM I-1572) and LPC-S01 (DSM 26760) for two weeks enhanced systemic amino acid availability, with the increments for methionine, histidine, and branched-chain amino acids being 16.3%, 49.2%, and 26.8%, respectively.40 These improvements are critical because they limit the substrate available for putrefactive fermentation. Unlike animal proteins, plant-based diets often feature a lower ratio of sulfur-containing amino acids, which results in reduced production of hydrogen sulfide. This is beneficial because high concentrations of hydrogen sulfide can impair mitochondrial respiration in colonocytes and compromise the mucosal barrier. However, overprocessing can lead to unintended consequences for gastrointestinal physiology. Extensive heat treatment can induce the formation of advanced Maillard reaction products and insoluble protein aggregates. These modifications diminish enzymatic digestibility and increase the load of resistant protein substrates available for microbial proteolysis.41 Such alterations may shift colonic fermentation toward pathways associated with mucosal irritation.
Additionally, the synergistic interaction between plant proteins and probiotics plays a pivotal role in shaping the gut ecosystem. Probiotics enhance the nutritional quality of plant proteins from sources such as soybean, pea, and rice by liberating bioactive peptides and diversifying microbial metabolites. While fiber fermentation produces beneficial short-chain fatty acids (SCFAs), certain plant proteins are rich in tryptophan that serves as a precursor for microbial-derived indoles like indole-3-propionic acid (IPA). These metabolites act as signaling ligands for the aryl hydrocarbon receptor (AhR) in the gut mucosa to promote immune homeostasis and strengthen the intestinal barrier. This synergy is evidenced by research using aging senescence-accelerated mouse prone 8 (SAMP8) mouse models where a low-protein diet supplemented with soy and pea protein isolates and probiotics significantly improved protein utilization and muscle strength. As reported by Han et al.,42 these functional benefits were accompanied by increased abundances of beneficial bacterial taxa such as Bifidobacterium and Roseburia, along with elevated levels of butyrate and IPA. Importantly, these metabolic shifts were associated with reduced circulating inflammatory cytokines and the modulation of immune-related genes, which highlights a functional link between plant protein and probiotic co-intervention in regulating host health through the gut–muscle axis.
In vivo evidence further indicates that plant proteins influence colonic microbial metabolism in a manner that differs from animal-derived proteins. Diets based on soybean or cottonseed protein have been shown to produce significantly lower concentrations of proteolytic products like ammonia compared to animal-protein diets.43 This suggests that plant proteins, possibly through their fiber-associated matrix and lower rates of small intestinal escape, reduce the availability of undigested substrates in the hindgut and moderate microbial putrefaction. Human studies support this observation, as legume-rich dietary patterns have been found to increase the abundance of beneficial bacterial groups like Bifidobacterium and Roseburia. These microbial changes are often accompanied by reductions in inflammatory biomarkers and improved lipid profiles.44
5. Non-communicable diseases
The relationship between dietary protein sources and the development and progression of non-communicable diseases (NCDs) has emerged as a critical area of nutritional epidemiology and clinical research. While the fundamental role of adequate protein intake in human health is undisputed, and contemporary science is increasingly focusing on how the source of dietary protein specifically the shift from animal-based to plant-based origins modulates disease risk through distinct physiological mechanisms. This section will examine the evidence linking plant protein consumption to three major NCDs: cardiovascular disease, diabetes mellitus, and chronic kidney disease. The analysis will explore not only the observed epidemiological associations but also the underlying molecular and metabolic pathways that explain these relationships, while acknowledging areas where evidence remains limited or conflicting. The clinical efficacy of plant proteins is highly dependent on tailored dosage strategies that account for the recipient's life stage and health status. As summarized in Table 2, these recommendations range from general metabolic support in healthy adults to high-purity protein-probiotic co-interventions for the elderly, and stage-specific protein source substitution for patients with chronic diseases.| Category | Target group/stage | Evidence-based dosage/strategy | Key clinical outcome | Ref. |
|---|---|---|---|---|
| Age groups | General healthy adults | 1.2–1.6 g per kg body weight per day | Maintain metabolic homeostasis and tissue repair | 1 |
| Elderly/sarcopenia | 20 g peas protein + probiotics (e.g., L. paracasei) | Improved protein utilization, AA availability, and muscle strength | 40 and 42 | |
| Physiological states | Weight management (Satiety) | 50 g soy protein dose or high-protein meals (25% energy) | Increased satiety perception and reduced subsequent energy intake | 10 and 11 |
| Cardiovascular health | ≥25 g day−1 soy protein | Significant reduction in systolic and diastolic blood pressure | 49 | |
| Disease stages | Diabetes (T2DM prevention) | Replace 1 daily serving of red meat with legumes/nuts | 11%–18% reduction in T2DM risk; improved insulin sensitivity | 61 and 63 |
| Early-stage CKD (stages 3 and 4) | Replace animal protein with 70% plant-derived protein | Slower eGFR decline, reduced proteinuria, and lower phosphorus load | 73 | |
| Advanced CKD/dialysis | Restricted total protein (0.6–0.8 g kg−1) with high plant protein ratio | Attenuated generation of uremic toxins and reduced multi-organ burden | 69, 72 and 75 |
5.1 Cardiovascular disease (CVD)
As summarized in Fig. 2, the cardioprotective efficacy of plant proteins is primarily mediated through three interconnected mechanisms: lipid profile optimization, blood pressure regulation, and anti-inflammatory signaling. Among these, the lipid-modifying properties of specific plant proteins have been most extensively documented. A comprehensive meta-analysis of 112 randomized controlled trials demonstrated that the isocaloric substitution of plant protein for animal protein significantly reduced established lipid targets for cardiovascular risk, including low-density lipoprotein cholesterol (LDL-C), non-high-density lipoprotein cholesterol (non-HDL-C), and apolipoprotein B (ApoB).45 Among various plant proteins, soy protein has been the most intensively studied. The bioactivity is largely attributed to globulin fractions, particularly β-conglycinin (7S) and glycinin (11S). These components are thought to upregulate hepatic LDL receptors, thereby accelerating the clearance of circulating LDL-C.46 Notably, the efficacy of these plant proteins is also influenced by their genetic background and subunit composition. For instance, soy protein isolates (SPIs) rich in the 7S fraction, such as the 7S (α′ + α)-null genotype, exhibited superior potency in elevating HDL-C levels in hypercholesterolemic models.47 It is noteworthy that the cardiovascular benefits of the soy matrix are augmented by co-occurring bioactive compounds. Isoflavones, in particular, appear to act synergistically with the soy protein fractions to modulate lipid metabolism and further improve total cholesterol profiles.48In addition to lipid metabolism, plant-derived proteins exhibit significant vasoprotective potential through the regulation of blood pressure. Clinical meta-analyses indicate that soy protein supplementation (≥25 g day−1) effectively reduced both systolic and diastolic blood pressure, an effect partially attributed to the vasodilatory properties of isoflavones.49 Beyond soy, dietary pulses such as lentils and chickpeas also confer beneficial effects on vascular function and pressure regulation, suggesting that antihypertensive properties are a shared attribute among diverse legume species.50 Recent advancements in bioactive peptide research have further elucidated the molecular mechanisms behind these observations. For instance, Moringa oleifera (MO) leaf protein has emerged as a potent source of antihypertensive peptides. Hydrolysates from MO leaves, specifically those with a molecular weight <1 kDa, demonstrated the ability to exert dual inhibitory effects on both angiotensin-converting enzyme (ACE) and renin in the renin–angiotensin system (RAS). Identified peptide sequences, such as Leu–Gly–Phe–Phe (LGF) and Gly–Leu–Phe–Phe (GLFF), not only significantly lowered blood pressure in spontaneously hypertensive rats (SHRs) but also demonstrated remarkable stability against gastrointestinal digestion.51 Despite these promising molecular findings, the application of leafy biomass proteins is currently constrained by low bioaccessibility and a lack of long-term human intervention studies to validate their efficacy and safety. Another broader meta-analysis reported that while substituting protein for carbohydrates lowers blood pressure, the effects of plant and animal protein were broadly similar, suggesting that the benefit in such comparisons may be partly attributed to protein per se, and the definitive superiority of plant protein for blood pressure requires further confirmation.52
Plant proteins also significantly attenuate systemic chronic inflammation, a pathological hallmark of CVD. This anti-inflammatory efficacy is believed to arise from a synergistic interplay between bioactive peptides and co-passengers within the plant matrix. At the molecular level, soy-derived tripeptides such as Leu–Ser–Trp (LSW) have been shown to suppress vascular inflammation by downregulating COX-2 and angiotensin II Type 1 receptor (AT1R) expressions via the SRC/ERK signaling pathway.53 This protective effect extends to systemic markers. For instance, chickpea protein hydrolysates was reported to not only reduce the C-reactive protein in hypercholesterolemic models but also uniquely enhance the antioxidant capacity of HDL, outperforming animal-derived proteins like casein.54 Furthermore, the matrix effect of plant-based foods integrates the actions of associated bioactive constituents. The intrinsic isoflavones and other polyphenols in plant protein isolates were shown to interfere with pro-inflammatory cytokine cascades, thereby stabilizing the vasculature.55 Crucially, the inclusion of dietary fiber facilitates a gut-heart axis mechanism. A study reported that supplementation with soy-based matrices restored the gut microbiota eubiosis by increasing the production of SCFAs like butyrate. This, in turn, upregulated colonic anti-inflammatory markers (e.g., IL-10 and Foxp3) and reinforced the intestinal barrier integrity via zonula occludens protein-1 (ZO-1) and the membrane linker protein occludin, preventing the translocation of pro-inflammatory endotoxins into the circulation.56
Despite the robust evidence supporting the cardiometabolic benefits of plant proteins, clinical efficacy exhibits significant heterogeneity depending on intervention parameters. For instance, a rigorous six-month trial in postmenopausal women with prediabetes found that daily supplementation with 15 g of soy protein (plus 100 mg of isoflavones) failed to significantly alter inflammatory or lipid markers compared to a milk protein control.57 Such discrepancies underscore that the efficacy of plant proteins is modulated by a complex constellation of factors, including the participant baseline metabolic status, intervention dosage, duration, and the specific dietary matrix used when these variables are optimized to ensure meaningful dietary substitution. Furthermore, emerging evidence suggests that the benefits of plant proteins may extend to improving the functional quality of protective molecules. A notable example is the effect of rice endosperm protein which enhanced the intrinsic anti-inflammatory capacity of high-density lipoprotein (HDL) in murine models. This functional improvement in HDL was directly correlated with reduced atherosclerotic lesion formation, revealing a protective pathway that complements traditional lipid-lowering and anti-inflammatory mechanisms.58
5.2 Diabetes mellitus
Type 2 diabetes mellitus (T2DM) is characterized by a complex interplay of insulin resistance, β-cell dysfunction, and chronic systemic inflammation. Plant protein-rich foods, including legumes, cereals, nuts, and oilseeds, differ from animal protein foods in the nutrient matrix, typically providing more unsaturated fatty acids, polyphenols, and dietary fiber when consumed in their minimally processed form.59 Prospective cohort analyses suggest that replacing red and processed meats with legumes, nuts, whole grains, or isolated plant protein reduces diabetes incidence, largely through improvements in lipid profiles and reduced exposure to saturated fat, heme iron, and advanced glycation end products. For example, a meta-analysis of 11 cohort studies reported that higher total and animal protein intake were associated with an increased risk of T2DM, whereas plant protein intake showed a neutral to protective association, particularly in women.60 Specifically, modeling the replacement of one daily serving of red meat with legumes or nuts was associated with an 11% to 18% reduction in T2DM risk.61The amino acid composition of plant proteins may further contribute to their metabolic advantages in glycemic regulation. Compared with many animal-derived proteins, plant proteins generally contain lower levels of branched-chain amino acids, methionine, and aromatic amino acids, which are associated with impaired insulin sensitivity when consumed in excess.62 Ji et al. demonstrated that the soy protein isolate was more effective than the whey protein isolate in reducing fasting insulin concentrations and enhancing the whole-body insulin sensitivity, as assessed by the Homeostatic Model Assessment for Insulin Resistance (HOMA-IR), in obese and insulin-resistant mouse model.63 Such metabolic improvements may reflect slower gastric emptying, altered amino acid absorption kinetics, and activation of nutrient-sensing pathways, which collectively enhance insulin sensitivity.62
Beyond fundamental amino acid profiles, the digestion kinetics of plant proteins and the subsequent liberation of bioactive peptides play a decisive role in glycemic regulation. Specific plant-derived oligopeptides have been shown to enhance insulin signaling through the modulation of PI3K-Akt signaling pathways and the attenuation of oxidative stress in insulin-sensitive tissues. For instance, pea-derived peptides restored IRS-1-Akt signaling in insulin-resistant HepG2 cells, while peptides from algae and chickpeas exhibited potent inhibitory activity against dipeptidyl peptidase-IV (DPP-4).64 Specifically, the identified short-chain peptides from chickpea hydrolysates were the key contributors to this enzymatic inhibition, which served to prolong the half-life of incretin hormones and improved postprandial glucose disposal.65,66 Moreover, plant proteins typically coexist with intact dietary fiber in whole-food matrices, which delays glucose absorption and contributes to attenuated postprandial glycemia.67 Nevertheless, the metabolic efficacy of these plant proteins is highly sensitive to technological interventions. While fermentation and controlled enzymatic hydrolysis can enhance the bioaccessibility of antioxidant and anti-inflammatory fragments, excessive thermal treatments or advanced Maillard reactions may impair peptide formation and reduce the overall glycemic benefits of plant proteins. These observations indicate that the technological context of consumption is a critical determinant of the functional properties of plant proteins in diabetes management.68
5.3 Chronic kidney disease
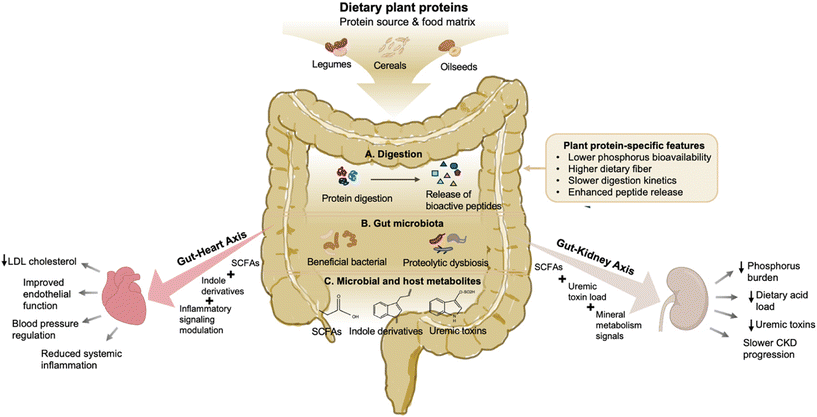
Chronic kidney disease (CKD) is characterized by a progressive decline in the glomerular filtration rate and the accumulation of uremic toxins. While traditional management emphasized a restricted protein intake to minimize the renal workload, contemporary research identifies the protein source as a critical modulator of disease progression. Prospective cohort analyses revealed that a higher intake of plant-derived proteins, particularly as a replacement for red and processed meats, was associated with a significantly slower decline in the estimated glomerular filtration rate (eGFR) and reduced proteinuria. A recent meta-analysis further demonstrated a consistent dose–response relationship between plant-based dietary patterns and reduced CKD incidence.69 Notably, NHANES III data indicated that a higher proportion of the dietary plant protein was associated with lower all-cause mortality for individuals with an eGFR below 60 mL min−1 per 1.73 m2.70The modulation of the gut–heart–kidney axis is increasingly recognized as another key benefit (Fig. 2). Plant proteins facilitate a healthier gut microbiota profile, leading to the attenuated generation of gut-derived uremic toxins such as p-cresyl sulfate and indoxyl sulfate.71 Clinical trials, such as the one conducted by Azadbakht et al., demonstrated that partially replacing animal protein with soy protein in patients with diabetic nephropathy resulted in significant reductions in proteinuria and urinary urea nitrogen. These improvements are often accompanied by optimized lipid profiles and reduced markers of systemic inflammation, collectively alleviating the multi-organ burden characteristic of CKD.72 Phosphorus bioavailability represents another critical distinction between plant and animal protein sources. Phosphorus in plant-based foods is largely bound to phytate, leading to substantially lower intestinal absorption compared with the highly bioavailable inorganic and animal-derived phosphorus abundant in meat and processed foods. In patients with stage 3–4 CKD, a controlled dietary intervention replacing animal protein with a diet containing 70% plant-derived protein for four weeks significantly reduced 24-hour urinary phosphorus excretion and dietary acid load without adverse effects on muscle mass or functional status.73
Nevertheless, the application of plant proteins must be tailored to the specific stage of CKD and individual metabolic needs. According to the NHANES-based analyses, the association between plant protein intake and renal outcomes is not linear but follows a stage-specific pattern. While moderate to high plant protein ratios are favorable in non-CKD and early CKD stages, these associations can become attenuated or non-significant in advanced stages. Dose–response analyses even revealed U-shaped or inverted U-shaped relationships, suggesting that the optimal protein balance may shift as renal function declines.74 In early-stage CKD, whole-food plant sources like legumes and whole grains provide essential fiber and a low acid load that support renal longevity. For patients with advanced CKD or those requiring dialysis, practical concerns regarding potassium burden and phosphorus management may necessitate more structured dietary oversight, indicating that the technological form and processing of plant proteins remain critical determinants of their clinical utility in end-stage renal care.75
6. Future needs
Despite the accumulating evidence supporting the preventive and/or therapeutic potential of plant proteins, several critical knowledge gaps must be addressed to translate these findings into precise clinical guidelines. The following sections outline the essential research priorities required to advance our understanding of plant protein based nutrition.6.1 Diversity and specificity of plant protein sources
While plant protein intake is broadly linked to favorable outcomes, there is an urgent need to disentangle the specific roles of different origins. Plant proteins are often treated as a homogeneous dietary category in observational studies and dietary guidelines, despite substantial heterogeneity in amino acid composition, protein structure, digestibility, and food matrix interactions across different origins.5 Legume-derived proteins (e.g., soy, pea, lentil, and chickpea), cereal proteins (e.g., wheat, rice, and oats), and proteins from oilseeds or pseudocereals differ markedly in their proportions of essential amino acids, sulfur-containing amino acids, and branched-chain amino acids, as well as in their association with fiber, phytate, and polyphenols. Beyond traditional crops, novel plant proteins such as leafy biomass (e.g., RuBisCO from alfalfa or duckweed) represent a sustainable alternative with a balanced essential amino acid profile. However, their practical application is currently constrained by significant technical and sensory limitations. The intensive biorefinery processes required to remove chlorophyll and phenolic compounds can lead to protein denaturation, while persistent green off-notes and bitter flavors remain primary barriers to consumer acceptance.76 These intrinsic differences and technological challenges are likely to influence postprandial metabolism, nitrogen utilization efficiency, acid load, and gut microbial fermentation, and yet remain insufficiently addressed in current research. Therefore, the extent to which observed health benefits attributed to plant protein are driven by specific sources rather than plant-based diets as a whole remains unclear.Future research should prioritize controlled comparisons among distinct plant protein sources to clarify their differential metabolic and clinical effects. While soy protein has been extensively studied and often serves as a reference plant protein, its dominance in the literature may limit generalizability to other increasingly consumed proteins such as pea, faba bean, lentil, and cereal-derived proteins. Moreover, future studies can prioritize controlled, head-to-head comparisons of major and emerging plant protein sources (e.g., legume vs. cereal vs. oilseed proteins) to delineate their distinct impacts on human health. Emerging dietary patterns and food technologies have rapidly expanded the use of non-soy plant proteins in both whole-food and processed formats, yet long-term human intervention studies evaluating their effects on glycemic control, lipid metabolism, renal function, and mineral homeostasis are scarce. Additionally, the physiological effects of plant proteins are further shaped by their form of utilization, including whole-food matrices, plant protein concentrates, isolates, hydrolysates, and derived bioactive peptides. Processing and fractionation can markedly alter the protein structure, digestion kinetics, and interactions with other dietary components, thereby modulating postprandial amino acid availability, lipid handling, and gut microbial responses. For example, protein isolates and hydrolysates may exhibit enhanced digestibility or bioactivity compared with intact proteins, yet they lack the complex food matrix present in whole legumes or cereals, which can slow digestion and influence metabolic outcomes. Consequently, evidence derived from isolated or hydrolyzed plant proteins may not be directly extrapolated to whole-food sources, even when derived from the same origin. Distinguishing between the protein source and the protein form as well as understanding their interaction is therefore essential for interpreting heterogeneous findings across studies and for translating plant protein research into dietary recommendations.
6.2 The food matrix and synergistic interactions
Plant proteins are inherently consumed within complex food matrices rich in dietary fiber, resistant starch, and diverse polysaccharides. These constituents can modulate nutrient absorption, gastrointestinal transit, and microbial metabolism, thereby influencing the physiological effects of plant proteins. A key research imperative is to move beyond studying isolated protein effects and to quantify the synergistic interactions between plant proteins and these co-passengers. We must elucidate how fibers from legumes, cereals, and oilseeds jointly modulate gastric emptying, nutrient absorption kinetics, and gut microbial metabolism to produce the attenuated postprandial glycemic responses and enhanced SCFA production observed with whole foods. Understanding these matrix-driven synergies is critical to explain why health outcomes from whole plant foods may not be fully replicable by isolated plant protein ingredients.Plant proteins often coexist with polyphenols, flavonoids, saponins, and other secondary metabolites that can further enhance metabolic and health outcomes. These bioactive phytochemicals exert antioxidant and anti-inflammatory effects, potentially complementing the benefits of protein-derived peptides in insulin sensitivity, lipid regulation, and renal function. For example, isoflavones in soy and phenolic compounds in legumes have been reported to reduce oxidative stress in vitro and in animal models, and to modulate enzymes involved in carbohydrate and lipid metabolism.77 The interaction between these phytochemicals and plant protein may potentiate the formation or activity of bioactive peptides during gastrointestinal digestion, suggesting a cooperative effect that extends beyond the protein itself. Future research can move beyond observing additive effects to mechanistically dissect these interactions and quantify the relative contribution of protein versus phytochemicals to observed health outcomes in humans.
However, plant protein matrices are often rich in phytate, an antinutritional factor that can chelate essential minerals such as iron, zinc, and calcium, thereby reducing their bioavailability. This presents a potential limitation in plant protein-based diets, particularly in populations with marginal micronutrient intake. To mitigate these effects, various processing strategies such as fermentation, germination, enzymatic hydrolysis, and phytase treatment have been shown to effectively degrade phytate and enhance mineral bioaccessibility.78 By reducing antinutritional compounds and improving peptide release, such approaches enable the concurrent delivery of high-quality amino acids and functional co-factors. The efficacy of plant protein modification is highly sensitive to optimized processing parameters. For HME, maintaining a moisture content of 60–65% and specific temperature profiles is critical to achieving a meat-like fibrillar texture without inducing excessive protein cross-linking that might hinder enzyme accessibility.37 Furthermore, processing technologies such as fermentation and extrusion exert synergistic effects on the entire food matrix by modulating interactions between proteins, fibers, and phytochemicals. Combining fermentation with a neutral pH shift prior to extrusion can effectively remove intensive off-odors; however, the impact on the matrix integrity must be carefully managed. Research on rapeseed protein indicated that fermentation-induced proteolysis can release short peptides that may form complexes with fiber-derived polysaccharides and phytochemical hydrolysis products (e.g., from glucosinolates), which potentially hinders the formation of the fibrous structure required for meat analogues.79 Future research can systematically establish quantitative relationships between specific processing parameters (e.g., fermentation strains, enzymatic hydrolysis conditions, and extrusion temperature) and the resultant modifications in the food matrix. Key outcomes to measure include the reduction of antinutritional factors, the preservation or enhancement of bioactive phytochemicals, the improvement of protein digestibility and amino acid release kinetics, and the consequent metabolic responses in vivo. This relationship among processing, matrix, and function is essential for rationally designing next-generation plant-protein ingredients and foods that maximize nutritional quality and deliver targeted health benefits beyond basic protein supplementation.
Ultimately, emerging evidence has highlighted the synergistic potential of plant proteins and probiotics in modulating gut health and preventing non-communicable diseases (NCDs). Within the food matrix, plant protein digestion products serve as critical substrates for the gut microbiota, promoting the production of short-chain fatty acids (SCFAs) and beneficial metabolites like indole-3-propionic acid. When combined with specific probiotic strains, these proteins enhance microbial diversity and reinforce the intestinal barrier, thereby reducing systemic inflammation.80 This mechanistic potential is increasingly supported by clinical application evidence. For instance, a randomized clinical trial (RCT) involving 100 patients with Type 2 diabetes mellitus (T2DM) demonstrated that a six-week intervention with soymilk and probiotics significantly improved cardiovascular risk profiles-evidenced by reductions in diastolic blood pressure, triglycerides, and total cholesterol-alongside improved insulin sensitivity.81 While statistical synergy compared to individual components may vary across study designs, the observed metabolic improvements underscore the practical efficacy of integrated dietary strategies for multi-target management of chronic diseases. This perspective emphasizes that plant proteins are not merely individual nutrients but integral modulators within a complex diet–microbiota-host interaction network, where the food matrix ultimately governs the physiological outcome.82
6.3 Peptidomics and gastrointestinal digestion as a functional gateway
Gastrointestinal digestion is increasingly recognized not merely as a degradative process, but as a critical phase for the generation and functional exposure of bioactive peptides from dietary proteins. Proteolysis by gastric and pancreatic enzymes can release encrypted peptide sequences that are inactive within the native protein structure but acquire biological activity upon digestion. A comprehensive review has demonstrated that digestive conditions including enzyme specificity, pH gradients, and food matrix interactions strongly determined the peptide profiles generated from food proteins and, consequently, their physiological relevance.83 From this perspective, protein digestibility does not solely dictate the amino acid availability, but also governs the emergence of peptide-mediated regulatory effects on host metabolism.Accumulating experimental evidence has indicated that plant protein-derived peptides can directly modulate glucose homeostasis through effects on insulin signaling and carbohydrate metabolism. In cellular models of insulin resistance, oligopeptides derived from pea protein digestion have been shown to restore IRS-1 and Akt phosphorylation, suppress oxidative stress-related pathways, and enhance glucose uptake, thereby improving insulin responsiveness in hepatocytes.64 Similar mechanisms have been reported for peptides released from soy and lupin proteins, which influenced PI3K-Akt signaling cascades and attenuated stress-activated kinases implicated in insulin resistance. However, most available evidence is derived from simulated digestion systems and in vitro cellular models, and key uncertainties remain regarding peptide stability, intestinal permeability, and effective concentrations under physiological dietary conditions. Addressing these gaps will require integrated approaches combining dynamic digestion models, intestinal transport studies, and well-designed human interventions to establish the true relevance of digestion-derived plant protein peptides for chronic disease prevention.
The kinetics of peptide release and persistence during digestion further distinguish plant proteins from many animal-derived counterparts. The relatively slower digestion rates and heterogeneous protein structures of legumes and cereals promote a more gradual liberation of peptides and amino acids, which may favor sustained metabolic signaling rather than rapid postprandial excursions. Importantly, food processing strategies strongly influence these outcomes. Fermentation and controlled enzymatic hydrolysis can enhance the generation of functional peptides with antioxidant, enzyme-inhibitory, or anti-inflammatory properties, whereas excessive thermal treatment may impair peptide release or reduce bioactivity. Future studies should move beyond identifying these inhibitory effects and instead focus on quantifying how specific processing parameters determine the structural integrity and physiological potency of plant-derived peptides.
Current evidence supports a conceptual framework in which gastrointestinal digestion serves as a functional gateway that transforms plant proteins into a diverse repertoire of bioactive peptides capable of modulating host metabolic pathways. Elucidating the stability, absorption, and target specificity of these peptides in vivo remains an important challenge, particularly in distinguishing direct peptide effects from secondary metabolic adaptations. Nevertheless, advances in peptidomics and digestion models continue to strengthen the link between plant protein digestion and peptide-mediated regulation of glucose metabolism and metabolic health.
7. Conclusions
Plant proteins represent more than mere alternative dietary protein sources, as they exert multifaceted effects on human health through mechanisms that extend far beyond protein quantity and basic amino acid composition. Evidence synthesized in this review indicates that plant proteins influence satiety regulation, lipid metabolism, and gut health, while also modulating the risk and progression of major non-communicable diseases, including cardiovascular disease, diabetes mellitus, and chronic kidney disease. These health outcomes are primarily driven by the integrated effects of digestion kinetics, food matrix interactions, and the presence of bioactive compounds. Importantly, the physiological impact of plant protein intake is highly context-dependent, being shaped by the specific protein source, the methods of processing, and co-existing constituents such as fiber and phytochemicals. Emerging data further suggest that peptides and microbial metabolites derived from plant proteins can directly regulate host metabolic, inflammatory, and immune pathways, highlighting mechanisms that are distinct from those traditionally associated with animal proteins. Despite growing interest, comparative evidence across diverse plant protein sources remains limited, and long-term human intervention studies are essential to clarify source-specific efficacy as well as dose and response relationships across different disease stages. Addressing these gaps will be critical for translating these mechanistic insights into practical dietary recommendations and for guiding the rational development of plant protein based foods with targeted health functions.Author contributions
Kun Gao: conceptualization and writing – original draft. Shiyu Zhou: writing – review & editing. Jiajia Rao: writing – review & editing. Bingcan Chen: conceptualization, project administration, funding acquisition, and writing – review & editing.Conflicts of interest
There are no conflicts of interest to declare.Data availability
All data supporting this systematic review were extracted from previous publications and are available within this review.Acknowledgements
This work is partially supported by the USDA-ARS Pulse Crop Health Initiative (FAR0037153).References
- U.S. Department of Agriculture and U.S. Department of Health and Human Services, Prioritize whole, health, and nutritious foods with the new Dietary Guidelines for Americans, Dietary Guidelines for Americans, https://www.dietaryguidelines.gov.
- J. A. Delcour, I. J. Joye, B. Pareyt, E. Wilderjans, K. Brijs and B. Lagrain, Wheat gluten functionality as a quality determinant in cereal-based food products, Annu. Rev. Food Sci. Technol., 2012, 3, 469–492 CrossRef CAS PubMed.
- R. F. Gonçalves, H. Zhou, A. A. Vicente, A. C. Pinheiro and D. J. McClements, Plant-based bigels for delivery of bioactive compounds: Influence of hydrogel: oleogel ratio and protein concentration on their physicochemical properties, Food Hydrocoll., 2024, 150, 109721 CrossRef.
- K. Gao and B. Chen, The application of advanced analytical techniques in ensuring quality, nutrition, and safety of cereal-based food, Adv. Food Nutr. Res., 2025, 117, 265–302 Search PubMed.
- R. T. Ahnen, S. S. Jonnalagadda and J. L. Slavin, Role of plant protein in nutrition, wellness, and health, Nutr. Rev., 2019, 77, 735–747 CrossRef PubMed.
- K. Gao, J. Rao and B. Chen, Plant protein solubility: A challenge or insurmountable obstacle, Adv. Colloid Interface Sci., 2024, 324, 103074 CrossRef CAS PubMed.
- C. K. Richter, A. C. Skulas-Ray, C. M. Champagne and P. M. Kris-Etherton, Plant protein and animal proteins: do they differentially affect cardiovascular disease risk?, Adv. Nutr., 2015, 6, 712–728 CrossRef CAS PubMed.
- M. Zahir, V. Fogliano and E. Capuano, Food matrix and processing modulate in vitro protein digestibility in soybeans, Food Funct., 2018, 9, 6326–6336 RSC.
- J. King, S. Y. Leong, M. Alpos, C. Johnson, S. Mcleod, M. Peng, K. Sutton and I. Oey, Role of food processing and incorporating legumes in food products to increase protein intake and enhance satiety, Trends Food Sci. Technol., 2024, 147, 104466 CrossRef CAS.
- S. Nepocatych, C. E. Melson, T. A. Madzima and G. Balilionis, Comparison of the effects of a liquid breakfast meal with varying doses of plant-based soy protein on appetite profile, energy metabolism and intake, Appetite, 2019, 141, 104322 CrossRef PubMed.
- M. A. Veldhorst, A. G. Nieuwenhuizen, A. Hochstenbach-Waelen, K. R. Westerterp, M. P. Engelen, R.-J. M. Brummer, N. E. Deutz and M. S. Westerterp-Plantenga, Effects of high and normal soyprotein breakfasts on satiety and subsequent energy intake, including amino acid and ‘satiety'hormone responses, Eur. J. Nutr., 2009, 48, 92–100 CrossRef CAS PubMed.
- M. L. Braden, J. A. Gwin and H. J. Leidy, Protein source influences acute appetite and satiety but not subsequent food intake in healthy adults, J. Nutr., 2023, 153, 1825–1833 CrossRef PubMed.
- A. E. Yanni, A. Kokkinos, P. Binou, V. Papaioannou, M. Halabalaki, P. Konstantopoulos, S. Simati and V. T. Karathanos, Postprandial glucose and Gastrointestinal hormone responses of healthy subjects to wheat biscuits enriched with l-arginine or branched-chain amino acids of plant origin, Nutrients, 2022, 14, 4381 CrossRef CAS PubMed.
- J. Overduin, L. Guérin-Deremaux, D. Wils and T. T. Lambers, NUTRALYS® pea protein: characterization of in vitro gastric digestion and in vivo gastrointestinal peptide responses relevant to satiety, Food Nutr. Res., 2015, 59, 25622 CrossRef PubMed.
- M. S. Martin, A. W. Dynesen, B. Petersen, I. L. Petersen, P. Duque-Estrada and M. D. Aaslyng, The Satiating Effect of Extruded Plant Protein Compared with Native Plant and Meat Protein in a Ragú “Bolognaise” Meal: A Randomized Cross-Over Study, Nutrients, 2024, 16, 3407 CrossRef CAS PubMed.
- M. D. Kristensen, N. T. Bendsen, S. M. Christensen, A. Astrup and A. Raben, Meals based on vegetable protein sources (beans and peas) are more satiating than meals based on animal protein sources (veal and pork)–a randomized cross-over meal test study, Food Nutr. Res., 2016, 60, 32634 CrossRef PubMed.
- A. J. Johnston, R. C. Mollard, D. Dandeneau, D. S. MacKay, N. Ames, J. Curran, D. R. Bouchard and P. J. Jones, Acute effects of extruded pea fractions on glycemic response, insulin, appetite, and food intake in healthy young adults, results of a double-blind, randomized crossover trial, Appl. Physiol., Nutr., Metab., 2021, 46, 1126–1132 CrossRef CAS PubMed.
- T. Yu, N. Shang, L. Chen, K. Lu, H. Xia, L. Yang, S. Wang, G. Sun and W. Liao, The Regulatory Effects of Pea Protein Hydrolysate on Lipid Metabolism in Gestational Diabetic Mice, Mol. Nutr. Food Res., 2025, 69, e70178 CrossRef CAS PubMed.
- E. Keuleyan, S. Laurent, A. Riaublanc, C. Berton-Carabin and A. Meynier, Digestive fate of emulsions formulated with pea and lupin protein ingredients: Modulation of lipid digestibility and bioaccessibility by the interfacial composition?, Food Res. Int., 2025, 202, 115749 CrossRef CAS PubMed.
- G. Y. Campos Espinosa, Inhibitory effects of hydrolyzed oat bran proteins on human LDL oxidation and their bile acid binding capacity. Carleton University, 2019 Search PubMed.
- A. Boualga, J. Prost, D. Taleb-Senouci, D. Krouf, O. Kharoubi, M. Lamri-Senhadji, J. Belleville and M. Bouchenak, Purified chickpea or lentil proteins impair VLDL metabolism and lipoprotein lipase activity in epididymal fat, but not in muscle, compared to casein, in growing rats, Eur. J. Nutr., 2009, 48, 162–169 CrossRef CAS PubMed.
- F. Yang, J. Huang, H. He, X. Ju, Y. Ji, F. Deng, Z. Wang and R. He, Study on the hypolipidemic activity of rapeseed protein-derived peptides, Food Chem., 2023, 423, 136315 CrossRef CAS PubMed.
- M. L. Piñuel, E. Vilcacundo, P. Boeri, D. A. Barrio, D. Morales, A. Pinto, R. Moran, I. Samaniego and W. Carrillo, Extraction of protein concentrate from red bean (Phaseolus vulgaris L.): Antioxidant activity and inhibition of lipid peroxidation, J. Appl. Pharm. Sci., 2019, 9, 1–14 Search PubMed.
- E. Durand, S. Beaubier, F. Fine, P. Villeneuve and R. Kapel, High metal chelating properties from rapeseed meal proteins to counteract lipid oxidation in foods: controlled proteolysis and characterization, Eur. J. Lipid Sci. Technol., 2021, 123, 2000380 CrossRef CAS.
- L. W. Browning, H. Wang, J. W. Taylor, P. Wilde, M. Rodriguez-Garcia, L. A. M. Holland and T. P. Knowles, Digestibility and enteric release achieved with microencapsulates made from emulsion-templated plant proteins, Sustainable Food Technol., 2025, 3, 689–699 RSC.
- S. Blanco Mejia, M. Messina, S. S. Li, E. Viguiliouk, L. Chiavaroli, T. A. Khan, K. Srichaikul, A. Mirrahimi, J. L. Sievenpiper and P. Kris-Etherton, A meta-analysis of 46 studies identified by the FDA demonstrates that soy protein decreases circulating LDL and total cholesterol concentrations in adults, J. Nutr., 2019, 149, 968–981 CrossRef PubMed.
- B. Fang, J. Luo, Z. Cui, R. Liu, P. Wang and J. Zhang, Pea albumin alleviates oleic acid-induced lipid accumulation in LO2 cells through modulating lipid metabolism and fatty acid oxidation pathways, Foods, 2024, 13, 3482 CrossRef CAS PubMed.
- S. Sandra, E. A. Decker and D. J. McClements, Effect of interfacial protein cross-linking on the in vitro digestibility of emulsified corn oil by pancreatic lipase, J. Agric. Food Chem., 2008, 56, 7488–7494 CrossRef CAS PubMed.
- M. Golding and T. J. Wooster, The influence of emulsion structure and stability on lipid digestion, Curr. Opin. Colloid Interface Sci., 2010, 15, 90–101 CrossRef CAS.
- R. Boachie, S. Yao and C. C. Udenigwe, Molecular mechanisms of cholesterol-lowering peptides derived from food proteins, Curr. Opin. Food Sci., 2018, 20, 58–63 CrossRef.
- M. Zhao, K. Qiao, L. Zhang, L. Liang, S. Chen, L. Chen and Y. Zhang, Research Progress on Anti-Hyperlipidemia Peptides Derived from Foods, Nutrients, 2025, 17, 1181 CrossRef CAS PubMed.
- J. Gao, Q. Mang, Y. Sun and G. Xu, Short-Chain Fatty Acids (SCFAs) Modulate the Hepatic Glucose and Lipid Metabolism of Coilia nasus via the FFAR/AMPK Signaling Pathway In Vitro, Int. J. Mol. Sci., 2025, 26, 3654 CrossRef CAS PubMed.
- A. Ji, W. Chen, C. Liu, T. Zhang, R. Shi, X. Wang, H. Xu and D. Li, Soy protein compared with whey protein ameliorates insulin resistance by regulating lipid metabolism, AMPK/mTOR pathway and gut microbiota in high-fat diet-fed mice, Food Funct., 2023, 14, 5752–5767 RSC.
- J. Y. H. Seah, W. S. Chew, F. Torta, C. M. Khoo, M. R. Wenk, D. R. Herr, E. S. Tai and R. M. van Dam, Dietary fat and protein intake in relation to plasma sphingolipids as determined by a large-scale lipidomic analysis, Metabolites, 2021, 11, 93 CrossRef CAS PubMed.
- J. Jia, A. Dell'Olio, D. Izquierdo-Sandoval, E. Capuano, X. Liu, X. Duan and J. Rubert, Exploiting the interactions between plant proteins and gut microbiota to promote intestinal health, Trends Food Sci. Technol., 2024, 153, 104749 CrossRef CAS.
- I. Panapparambil Sooraj, J. Leech, T. F. O'Callaghan and O. McAuliffe, Application and Utilisation of Fermentation as a Processing Tool to Mitigate Protein Putrefaction in Plant-Based Diets, Front. Microbiol., 2025, 16, 1638378 CrossRef CAS PubMed.
- Z. Öztürk, M. Lille, N. Rosa-Sibakov and N. Sozer, Impact of heat treatment and high moisture extrusion on the in vitro protein digestibility of sunflower and pea protein ingredients, LWT – Food Sci. Technol., 2024, 214, 117133 CrossRef.
- W. Sun, J. He, H. Wang, Q. Zhang, W. Li and X. Rui, Solid-state fermentation alters the fate of red kidney bean protein during buccal and gastrointestinal digestion: Relationship with cotyledon cell wall integrity, Food Chem., 2023, 410, 135370 CrossRef CAS PubMed.
- H. D. Kim, K. S. Lee, K. E. Lee, H. J. Suh and B.-Y. Kim, Improved digestibility and bioavailability of pea protein following enzymatic treatment and fermentation by lactic acid bacteria, Food Sci. Biotechnol., 2024, 33, 607–615 CrossRef CAS PubMed.
- R. Jäger, J. Zaragoza, M. Purpura, S. Iametti, M. Marengo, G. M. Tinsley, A. J. Anzalone, J. M. Oliver, W. Fiore and A. Biffi, Probiotic administration increases amino acid absorption from plant protein: a placebo-controlled, randomized, double-blind, multicenter, crossover study, Probiotics Antimicrob. Proteins, 2020, 12, 1330–1339 CrossRef PubMed.
- J. Zhang, J. Wang, M. Li, S. Guo and Y. Lv, Effects of heat treatment on protein molecular structure and in vitro digestion in whole soybeans with different moisture content, Food Res. Int., 2022, 155, 111115 CrossRef CAS PubMed.
- R. Han, L. Ouyang, C. Yin, L. Cai, Q. Wu, L. Chen, J. Du, X. Li, Z. Zhu and Y. Pi, Supplementation of low-protein diets with plant protein and probiotics enhances muscle health by regulating gut microbiota and metabolomic profiles in SAMP8 mice, Food Funct., 2025, 16, 4575–4592 RSC.
- R. Li, L. Chang, G. Hou, Z. Song, Z. Fan, X. He and D.-X. Hou, Colonic microbiota and metabolites response to different dietary protein sources in a piglet model, Front. Nutr., 2019, 6, 151 CrossRef PubMed.
- X. Wu, A. S. Tjahyo, V. S. B. Volchanskaya, L. H. Wong, X. Lai, Y. N. Yong, F. Osman, S. L. Tay, P. Govindharajulu and S. Ponnalagu, A legume-enriched diet improves metabolic health in prediabetes mediated through gut microbiome: a randomized controlled trial, Nat. Commun., 2025, 16, 942 CrossRef CAS PubMed.
- S. S. Li, S. Blanco Mejia, L. Lytvyn, S. E. Stewart, E. Viguiliouk, V. Ha, R. J. de Souza, L. A. Leiter, C. W. Kendall and D. J. Jenkins, Effect of plant protein on blood lipids: a systematic review and meta–analysis of randomized controlled trials, J. Am. Heart Assoc., 2017, 6, e006659 CrossRef PubMed.
- P. G. Fassini, R. W. Noda, E. S. Ferreira, M. A. Silva, V. A. Neves and A. Demonte, Soybean glycinin improves HDL-C and suppresses the effects of rosuvastatin on hypercholesterolemic rats, Lipids Health Dis., 2011, 10, 165 CrossRef CAS PubMed.
- S. Liu, T. Luo, Y. Song, H. Ren, Z. Qiu, C. Ma, Y. Tian, Q. Wu, F. Wang and H. B. Krishnan, Hypocholesterolemic effects of soy protein isolates from soybeans differing in 7S and 11S globulin subunits vary in rats fed a high cholesterol diet, J. Funct. Foods, 2022, 99, 105347 CrossRef CAS.
- A. Barańska, A. Błaszczuk, W. Kanadys, B. Baczewska, M. Jędrych, E. Wawryk-Gawda and M. Polz-Dacewicz, Effects of soy protein containing of isoflavones and isoflavones extract on plasma lipid profile in postmenopausal women as a potential prevention factor in cardiovascular diseases: systematic review and meta-analysis of randomized controlled trials, Nutrients, 2021, 13, 2531 CrossRef PubMed.
- T. Kou, Q. Wang, J. Cai, J. Song, B. Du, K. Zhao, Y. Ma, B. Geng, Y. Zhang and X. Han, Effect of soybean protein on blood pressure in postmenopausal women: a meta-analysis of randomized controlled trials, Food Funct., 2017, 8, 2663–2671 RSC.
- O. G. Figueroa-Salcido, J. G. Arámburo-Gálvez, J. A. Mora-Melgem, D. L. Camacho-Cervantes, M. H. Gracia-Valenzuela, E. O. Cuevas-Rodríguez and N. Ontiveros, Alcalase-Based Chickpea (Cicer arietinum L.) Protein Hydrolysates Efficiently Reduce Systolic Blood Pressure in Spontaneously Hypertensive Rats, Foods, 2024, 13, 1216 CrossRef CAS PubMed.
- K. Ma, Y. Wang, M. Wang, Z. Wang, X. Wang, X. Ju and R. He, Antihypertensive activity of the ACE–renin inhibitory peptide derived from Moringa oleifera protein, Food Funct., 2021, 12, 8994–9006 RSC.
- S. M. Tielemans, M. Engberink, E. Brink, M. van Baak, S. Bakker and J. Geleijnse, Intake of total protein, plant protein and animal protein in relation to blood pressure: a meta-analysis of observational and intervention studies, J. Hum. Hypertens., 2013, 27, 564–571 CAS.
- Q. Lin, W. Liao, J. Bai, W. Wu and J. Wu, Soy protein-derived ACE-inhibitory peptide LSW (Leu-Ser-Trp) shows anti-inflammatory activity on vascular smooth muscle cells, J. Funct. Foods, 2017, 34, 248–253 CrossRef CAS.
- F. Dehiba, A. Allaoui, S. Benomar, S. Yahia, N. Guillén, M. J. Rodríguez-Yoldi, J. Osada and A. Boualga, Protective properties of sardine and chickpea protein hydrolysates against lipoprotein oxidative damages and some inflammation markers in hypercholesterolemic rats, Mediterr. J. Nutr. Metab., 2021, 14, 439–452 Search PubMed.
- Y. Cao, The protective effects of soy isoflavones on the cardiovascular system, Proceedings of the 4th International Conference on Biomedical and Intelligent Systems, 2025, 428–432 Search PubMed.
- S.-M. Lee, H. W. Han and S. Y. Yim, Beneficial effects of soy milk and fiber on high cholesterol diet-induced alteration of gut microbiota and inflammatory gene expression in rats, Food Funct., 2015, 6, 492–500 RSC.
- Z.-M. Liu, S. Ho, Y.-M. Chen and Y. Ho, The effects of isoflavones combined with soy protein on lipid profiles, C-reactive protein and cardiovascular risk among postmenopausal Chinese women, Nutr., Metab. Cardiovasc. Dis., 2012, 22, 712–719 CrossRef CAS PubMed.
- R. Kaseda, M. Hosojima, S. Kuwahara, H. Kabasawa, H. Aoki, Y. Higuchi, V. Kon, I. Narita and A. Saito, Rice Endosperm Protein Improves the Anti-Inflammatory Effects of High-Density Lipoprotein and Produces Lower Atherosclerotic Lesion Accelerated by the Renal Mass Reduction than Casein in a Mouse Model, J. Am. Nutr. Assoc., 2022, 41, 668–678 CAS.
- M. McMacken and S. Shah, A plant-based diet for the prevention and treatment of type 2 diabetes, J. Geriatr. Cardiol., 2017, 14, 342 CAS.
- S. Tian, Q. Xu, R. Jiang, T. Han, C. Sun and L. Na, Dietary protein consumption and the risk of type 2 diabetes: a systematic review and meta-analysis of cohort studies, Nutrients, 2017, 9, 982 CrossRef PubMed.
- A. M. L. Würtz, M. U. Jakobsen, M. L. Bertoia, T. Hou, E. B. Schmidt, W. C. Willett, K. Overvad, Q. Sun, J. E. Manson and F. B. Hu, Replacing the consumption of red meat with other major dietary protein sources and risk of type 2 diabetes mellitus: a prospective cohort study, Am. J. Clin. Nutr., 2021, 113, 612–621 CrossRef PubMed.
- J.-P. D. Bandt, X. Coumoul and R. Barouki, Branched-chain amino acids and insulin resistance, from protein supply to diet-induced obesity, Nutrients, 2022, 15, 68 CrossRef PubMed.
- A. Ji, Y. Qi, K. Zhao, J. Niu, R. Shi, Z. Qi, L. Zhou, C. Zhao and D. Li, Soy Protein Outperforms Whey Protein in Ameliorating Insulin Resistance but Not Obesity in High-Fat Diet-Induced Obese Mice, Nutrients, 2025, 17, 3427 CrossRef CAS PubMed.
- Y. Zhu, H. Zhang, Y. Wei, M. Cai, R. Gu, Y. Wang, Y. Ma and L. Chen, Pea–derived peptides, VLP, LLP, VA, and LL, improve insulin resistance in HepG2 cells via activating IRS–1/PI3K/AKT and blocking ROS–mediated p38MAPK signaling, J. Food Biochem., 2020, 44, e13454 CAS.
- K. A. A. Martínez and E. G. Mejia, Comparison of five chickpea varieties, optimization of hydrolysates production and evaluation of biomarkers for type 2 diabetes, Food Res. Int., 2021, 147, 110572 CrossRef PubMed.
- C. J. Radlowski, Chickpea (Cicer arietinum) protein hydrolysate ameliorates the metabolic effect of feeding a high-fat-diet-induced obesity in mice by modulating inflammation, University of Illinois at Urbana-Champaign, 2022 Search PubMed.
- C. G. Forde and D. Bolhuis, Interrelations between food form, texture, and matrix influence energy intake and metabolic responses, Curr. Nutr. Rep., 2022, 11, 124–132 CrossRef PubMed.
- W. Wu, J. Qiu, A. Wang and Z. Li, Impact of whole cereals and processing on type 2 diabetes mellitus: a review, Crit. Rev. Food Sci. Nutr., 2020, 60, 1447–1474 CrossRef CAS PubMed.
- Z. Dang, Y. He, R. Xie, P. Chen and F. Dong, Plant-based diet and chronic kidney disease: a systematic review and meta-analysis, J. Renal Nutr., 2025, 35, 517–530 CrossRef CAS PubMed.
- X. Chen, G. Wei, T. Jalili, J. Metos, A. Giri, M. E. Cho, R. Boucher, T. Greene and S. Beddhu, The associations of plant protein intake with all-cause mortality in CKD, Am. J. Kidney Dis., 2016, 67, 423–430 CrossRef CAS PubMed.
- J. J. Scialla and C. A. Anderson, Dietary acid load: a novel nutritional target in chronic kidney disease?, Adv. Chronic Kidney Dis., 2013, 20, 141–149 CrossRef PubMed.
- L. Azadbakht, R. Shakerhosseini, S. Atabak, M. Jamshidian, Y. Mehrabi and A. Esmaill-Zadeh, Beneficiary effect of dietary soy protein on lowering plasma levels of lipid and improving kidney function in type II diabetes with nephropathy, Eur. J. Clin. Nutr., 2003, 57, 1292–1294 CrossRef CAS PubMed.
- R. N. Moorthi, C. L. Armstrong, K. Janda, K. Ponsler-Sipes, J. R. Asplin and S. M. Moe, The effect of a diet containing 70% protein from plants on mineral metabolism and musculoskeletal health in chronic kidney disease, Am. J. Nephrol., 2015, 40, 582–591 CrossRef PubMed.
- G. Wang, Q. Yi, X. Zhang, M. Zhou, J. Zhao, H. Lu, J. Li, D. Meng, Y. Xu and K. Wang, Differential effects of plant protein ratio on renal function and mortality across CKD stages, Front. Nutr., 2025, 12, 1596836 CrossRef PubMed.
- S. M. Moe, M. P. Zidehsarai, M. A. Chambers, L. A. Jackman, J. S. Radcliffe, L. L. Trevino, S. E. Donahue and J. R. Asplin, Vegetarian compared with meat dietary protein source and phosphorus homeostasis in chronic kidney disease, Clin. J. Am. Soc. Nephrol., 2011, 6, 257–264 CrossRef CAS PubMed.
- A. Anoop, P. K. Pillai, M. Nickerson and K. Ragavan, Plant leaf proteins for food applications: Opportunities and challenges, Compr. Rev. Food Sci. Food Saf., 2023, 22, 473–501 CrossRef CAS PubMed.
- I.-S. Kim, Current perspectives on the beneficial effects of soybean isoflavones and their metabolites for humans, Antioxidants, 2021, 10, 1064 CrossRef CAS PubMed.
- J. Auer, M. Alminger, M. Marinea, M. Johansson, G. Zamaratskaia, A. Högberg and M. Langton, Assessing the digestibility and estimated bioavailability/bioaccessibility of plant-based proteins and minerals from soy, pea, and faba bean ingredients, LWT – Food Sci. Technol., 2024, 197, 115893 CrossRef CAS.
- A. Nisov, A. Valtonen, H. Aisala, A. Spaccasassi, C. Walser, C. Dawid and N. Sozer, Effect of peptide formation during rapeseed fermentation on meat analogue structure and sensory properties at different pH conditions, Food Res. Int., 2024, 180, 114070 CrossRef CAS PubMed.
- Q. Wu, J. Kan, Z. Cui, Y. Ma, X. Liu, R. Dong, D. Huang, L. Chen, J. Du and C. Fu, Understanding the nutritional benefits through plant proteins-probiotics interactions: mechanisms, challenges, and perspectives, Crit. Rev. Food Sci. Nutr., 2025, 65, 3569–3587 CrossRef CAS PubMed.
- A. Hasanpour, S. Babajafari, S. M. Mazloomi and M. Shams, The effects of soymilk plus probiotics supplementation on cardiovascular risk factors in patients with type 2 diabetes mellitus: a randomized clinical trial, BMC Endocr. Disord., 2023, 23, 36 CrossRef CAS PubMed.
- A. Rathi, T. Gaonkar, D. Dhar, G. Kallapura and S. Jadhav, Study of amino acids absorption and gut microbiome on consumption of pea protein blended with enzymes-probiotics supplement, Front. Nutr., 2024, 11, 1307734 CrossRef PubMed.
- S. Fernández-Tomé and B. Hernández-Ledesma, Gastrointestinal digestion of food proteins under the effects of released bioactive peptides on digestive health, Mol. Nutr. Food Res., 2020, 64, 2000401 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |