Open Access Article
Open Access ArticleDietary (poly)phenols, the gut-brain axis, and menopause: a perspective on an overlooked biological crossroad
María
García-Nicolás†
a,
María Paula
Jarrín-Orozco†
a,
María
Romo-Vaquero
 a,
María Elena
Martínez-Nortes
b,
María Ángeles
Ávila-Gálvez
*a and
Juan Carlos
Espín
a,
María Elena
Martínez-Nortes
b,
María Ángeles
Ávila-Gálvez
*a and
Juan Carlos
Espín
 *a
*a
aLaboratory of Food & Health, Research Group on Quality, Safety, and Bioactivity of Plant Foods, CEBAS-CSIC, 30100 Campus de Espinardo, Murcia, Spain. E-mail: mavila@cebas.csic.es; jcespin@cebas.csic.es
bDepartment of Obstetrics and Gynaecology, Virgen de La Arrixaca University Hospital, El Palmar, 30120, Murcia, Spain
First published on 14th April 2026
Abstract
Hormonal decline, chronic low-grade inflammation, metabolic alterations and polypharmacy shape the postmenopausal period. These factors remodel the gut microbiota and influence the production of microbial metabolites that modulate immune, endocrine, and neural communication. The gut-brain axis provides a framework for understanding how microbial activity affects cognition, mood, stress responses, neuroinflammation, and gastrointestinal function. Dietary (poly)phenols depend on gut microbial transformation to generate metabolites with distinct biological activity targeting mechanisms, such as intestinal and blood–brain barrier integrity, inflammatory signalling, redox balance, neurotransmitter synthesis, tryptophan metabolism, short-chain fatty acid production, and bile acid remodelling. These pathways are sensitive to hormonal decline, inflammaging, and polypharmacy, which modify microbial metabolism, host conjugation processes, enterohepatic cycling, and physiological response to dietary compounds. Despite this mechanistic basis, no human intervention study has examined these interactions in postmenopausal women. This perspective integrates three dimensions that are usually addressed separately: the physiological and pharmacological characteristics of postmenopause, the communication pathways of the gut-brain axis, and the gut microbial transformation of dietary (poly)phenols. We review human trials assessing (poly)phenols and gut-brain outcomes and highlight the scarcity of mechanistic endpoints, including microbial metabolites, barrier markers, neuroimmune mediators, and bile acid profiles. We also highlight how chronic medications reshape microbial composition and functionality, and how host targets of (poly)phenols, together with interindividual variability in polyphenol-related microbiota metabotypes, such as equol- and urolithin-producing metabotypes, influence biological responses and support personalised strategies. By identifying gaps and research priorities, this perspective provides a conceptual basis for developing precision health for postmenopausal women.
1. Introduction
1.1. Menopause as a multifactorial stage with pharmacological implications
Menopause is a complex biological transition that affects women across physiological, psychological, and social domains. It is diagnosed retrospectively after 12 consecutive months of amenorrhea in women with previously regular menstrual cycles. Ovarian hormone production declines gradually or abruptly. This hormonal shift triggers a cascade of changes that extend far beyond reproductive ageing. Estrogen deficiency contributes to neurovascular instability, altered lipid and glucose metabolism, and increased vulnerability to mood disturbances.1 These alterations are closely linked to a higher prevalence of cardiovascular disease, osteoporosis, type 2 diabetes (T2D), cognitive decline, and affective disorders.2The impact of menopause also extends into the social and economic spheres. Many women experience reduced work capacity, increased healthcare costs, and unequal access to specialised care, particularly in resource-limited settings. The psychological burden of menopausal symptoms can affect interpersonal relationships, professional performance, and overall quality of life, while cultural perceptions of ageing and femininity shape how symptoms are experienced and managed, influencing both help-seeking behaviour and treatment adherence.3
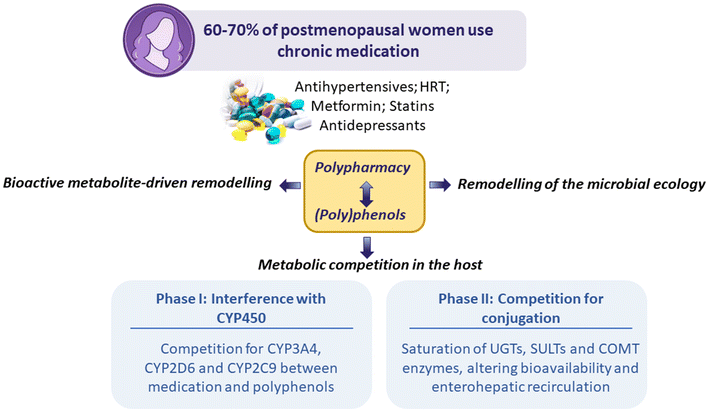
Menopause should not be understood solely as a hormonal transition but also as the onset of a pharmacologically complex stage in a woman's life. The accumulation of comorbidities such as hypertension, dyslipidemia, insulin resistance, osteoporosis, and depression often requires sustained pharmacological treatment.4 Consequently, polypharmacy becomes common, with therapeutic regimens that may include hormone replacement therapy (HRT), lipid-lowering drugs such as statins, antihypertensive agents, antidiabetic drugs such as metformin, and antidepressants. These treatments are essential for managing symptoms and preventing long-term complications. Still, their interactions with age-related physiological changes and individual variability can alter the metabolism of dietary bioactive compounds, such as (poly)phenols and their microbial-derived metabolites, drugs, as well as the therapeutic response, and the risk of adverse effects.5–9
These pharmacological and metabolic shifts, together with changes in the gut microbiota, may influence brain function, cognitive resilience, and mood regulation. Understanding these interactions highlights the importance of the gut-brain axis in postmenopausal health, a dimension that remains underexplored (Fig. 1). Notably, a recent review highlights this gap despite clear evidence of menopause-related changes in brain structure and function.10
1.2. The gut-brain axis
The gut-brain axis is a bidirectional communication system linking the gastrointestinal tract and the central nervous system through neural, immune, and endocrine pathways, with the vagus nerve, the intestinal barrier, the blood–brain barrier (BBB), and the gut microbiota as principal components.11–13 Gut microbes support digestion, immune regulation, and metabolic balance, producing metabolites such as short-chain fatty acids (SCFAs), neurotransmitter precursors, modified bile acids, and phenolic derivatives that can reach the systemic circulation or act via neural pathways to influence cognition, mood, and metabolic homeostasis,14–16 as also reviewed in the context of ageing and neurodegeneration.17Communication along the gut-brain axis is bidirectional because the brain regulates motility, secretion, and immune tone, which shape gut microbial composition and function, and microbial changes feedback on neuroendocrine and inflammatory pathways that modulate behaviour and systemic metabolism (Fig. 1).18
Age-related low-grade inflammation, or inflammaging, compromises gut barrier integrity and perturbs microbial ecology, increasing systemic inflammatory load and vulnerability to cognitive impairment and mood disturbances. A resilient gut microbiota helps preserve barrier function and limits bacterial translocation, thereby buffering inflammaging.19,20 Gut microbial composition also affects BBB development and maintenance. This effect is partly reversible by restoring the conventional microbiota or by administering microbial metabolites such as butyrate, which support BBB integrity and neuroprotection.21–23 Importantly, gut-derived molecules can exert context-dependent outcomes.
Trimethylamine N-oxide (TMAO), commonly associated with cardiovascular risk,24 has also been reported, under specific conditions, to support BBB stability and cognitive function. This highlights that concentration, microbial background and metabolic status determine the net physiological effects.25 In postmenopausal women, the decline in sex hormones shifts immune and metabolic set points, remodels the gut microbiota and its metabolite profile, and thereby alters susceptibility to cognitive decline and mood disorders, as well as the response to dietary or microbiota-targeted interventions for brain health.26–28
Given that microbial activity shapes the production of bioactive molecules, (poly)phenols may be particularly relevant candidates to modulate gut-brain communication in postmenopausal women, whose gut microbiota composition and pharmacological exposures may critically shape the production and bioactivity of microbial metabolites.
1.3. Dietary (poly)phenols
Phenolic compounds, commonly referred to as (poly)phenols, comprise a diverse family of plant-derived molecules abundant in fruits, vegetables, cereals, and foods such as tea, coffee, olive oil, and red wine, historically considered xenobiotics but now associated with modulation of chronic diseases, including metabolic, cardiovascular, and neurodegenerative disorders through effects on inflammation, redox balance, lipid metabolism, and mitophagy.17,29–32 These mechanisms are particularly relevant for postmenopausal women, in whom vascular dysfunction, metabolic disturbances, and neuroinflammation are prevalent and often coexist with polypharmacy. Overall, low systemic bioavailability, large structural diversity, and complex host metabolism impede the precision of dietary recommendations, so health outcomes attributed to (poly)phenols must be interpreted in the context of host conjugation, enterohepatic recycling, gut microbial metabolism, and interindividual variability (Fig. 1).9,33Most ingested (poly)phenols escape small-intestinal absorption and reach the colon, where microbial enzymes such as hydrolases, reductases, and dehydroxylases convert glycosides and oligomers into low-molecular-weight metabolites such as phenolic acids, urolithins, enterolignans, lunularin, equol, and others, which frequently show enhanced absorption and distinct bioactivity compared with their precursors. Host phase II metabolism produces conjugates, such as glucuronides and sulfates, that circulate and are excreted, and enterohepatic cycling prolongs exposure.34,35
Remarkably, some gut microbial metabolites influence systemic physiology both directly and indirectly, often considered as the primary drivers of the health effects observed upon consuming the parent (poly)phenol compound.33,36–38 For example, this is the case of equol and urolithin A, which are produced by the gut microbiota from isoflavones and ellagic acid, respectively. These metabolites have been reported to exert pleiotropic effects on chronic diseases by impacting metabolic and cellular homeostasis.9,32,39–41 In addition, microbial shifts induced by (poly)phenols alter SCFA production and the bile acid pool, thereby affecting gut barrier integrity, immune tone, metabolic signalling via FXR/TGR5, and gut-brain communication.36,42,43 Host genetics, sex, age, dietary matrix, and lifestyle determine which metabolites are formed and the magnitude of their effects, supporting a personalised approach to exploit (poly)phenol-microbiota interactions for health.9
These considerations highlight the need to contextualise (poly)phenol metabolism and bioactivity within the specific physiological and pharmacological landscape of postmenopausal women.
However, although the gut-brain axis and dietary (poly)phenols have received increasing attention, the intersection of hormonal decline, pharmacological treatments, and microbial metabolism in postmenopausal women remains largely unexplored. Recent work highlights that even the characterisation of gut-microbiota-brain interactions during menopause remains incomplete,10 underscoring the need for integrative frameworks such as the one proposed here. Thus, in this perspective, we aim to address this overlooked crossroads by integrating three critical dimensions: (i) the multifactorial, pharmacologically complex nature of menopause, (ii) bidirectional communication between the gut and brain, and (iii) the modulatory potential of dietary (poly)phenols. By examining how these factors converge to shape gut microbiota composition, metabolite production, and their impact on cognitive, mood, and metabolic health, we propose a conceptual framework that identifies research gaps and opportunities for precision nutrition and personalised interventions to support long-term well-being in postmenopausal women. This mechanistic overview sets the stage for exploring how (poly)phenols and microbial metabolism converge to influence the gut-brain axis.
2. Dietary (poly)phenols and the gut-brain axis: mechanistic overview
2.1. Direct influence of (poly)phenols on the gut-brain axis
Dietary (poly)phenols influence the gut-brain axis through a wide array of molecular and complementary mechanisms, as also described in ageing and neurodegeneration.17 These compounds exert pleiotropic effects as protective agents of the neurovascular unit, including anti-inflammatory actions, antioxidant and free-radical scavenging properties, and regulators of numerous signalling pathways (e.g., GABA, serotonin, cannabinoid receptors) that collectively act at multiple mechanistic levels.44,45 These mechanisms are particularly relevant in postmenopausal women, in whom hormonal decline exacerbates vascular dysfunction, inflammation, and barrier fragility.1,2,26–28A substantial body of evidence indicates that (poly)phenols contribute to the maintenance of vascular elasticity and the preservation of both intestinal and brain barrier integrity.46,47 These compounds are potent vasodilators that enhance endothelial nitric oxide synthase (eNOS) activity and NO bioavailability, thereby promoting anti-atherogenic and endothelial-protective gene expression profiles.48 Dietary (poly)phenols have been widely reported to inhibit iNOS expression and activity, reduce NO overproduction, suppress COX-2, and attenuate pro-inflammatory cytokines, including IL-1β, IL-6, and TNF-α, while modulating anti-inflammatory mediators, including IL-10. Plant-derived (poly)phenols traditionally used for menopausal symptoms have shown neurovascular and neuroimmune effects relevant to gut-brain communication.49
Beyond vascular effects, (poly)phenols also strengthen epithelial and endothelial barriers by upregulating key tight junction proteins, supporting the structure of the intestinal epithelium43 and the blood–brain barrier.50 Preservation of both barriers is essential for limiting peripheral inflammatory signals from reaching the brain.
Inflammation represents a central innate immune response to tissue injury, infection, and metabolic stress, and is tightly regulated by key signalling pathways, including nuclear factor κB (NF-κB), nuclear factor erythroid 2-related factor 2 (Nrf2), and mitogen-activated protein kinases (MAPKs). In the context of (poly)phenols, extensive preclinical evidence indicates that these compounds suppress key inflammatory mediators, including NO, iNOS, COX-2, and TNF-α following LPS or receptor activator of NF-κB ligand (RANKL) stimulation, indicating upstream inhibition of NF-κB activation.51–54 Moreover, prevention of NF-κB nuclear translocation by phenolic compounds has been consistently reported in models of both intestinal and brain disorders, highlighting a shared anti-inflammatory mechanism relevant to gut-brain axis regulation.55–57
Direct evidence linking (poly)phenol intake to modulation of the gut-brain axis has been demonstrated in aged, menopause-related animal models. In a 12-week supplementation intervention, tea extract attenuated cognitive decline by inhibiting the TLR4/NF-κB inflammatory cascade triggered by gut dysbiosis.58
Tryptophan metabolism represents another critical pathway at the interface of dietary (poly)phenols and the gut-brain axis. Tryptophan is an essential amino acid involved in protein synthesis and serves as a precursor for several neuroactive metabolites. Through the hepatic kynurenine pathway, tryptophan is converted into kynurenic acid. This neuroprotective compound antagonises excitatory amino acid receptors and has been implicated in modulating cognitive and psychiatric functions.59 In addition, tryptophan is the unique precursor of the neurotransmitter serotonin, which is vital for the processing of emotions, hunger, sleep, and pain, as well as colonic motility and secretory activity in the gut.60 In this context, dietary (poly)phenols have been shown to dampen inflammatory cascades and influence tryptophan metabolism, thereby shaping serotonin availability and downstream neurophysiological responses.61
Supporting this, anthocyanins and flavanols have been reported to mitigate oxidative stress in high-fat diet models by increasing SOD and GSH-Px activity, while elevating hippocampal 5-HT and dopamine levels.62 These neurochemical improvements were accompanied by increased abundances of several beneficial taxa, including Bifidobacterium, Lactobacillus, Roseburia, Faecalibaculum, Parabacteroides, and Ruminiclostridium, and by decreased abundances of Staphylococcus, a genus associated with gut inflammation. Collectively, these findings underscore the central role of tryptophan metabolism and redox balance in the pathogenesis of neurological and psychiatric disorders and highlight the capacity of dietary (poly)phenols to modulate these pathways through both direct and indirect mechanisms.
Finally, it is important to consider that many of the molecular targets influenced by (poly)phenols overlap with those affected by commonly prescribed pharmacological treatments. Given the high prevalence of chronic medication use in postmenopausal women, potential interactions at the level of shared receptors, enzymes, and signalling pathways may modify the efficacy and outcomes of dietary interventions.
2.2. Gut microbiota-mediated influence on the gut-brain axis
The gut microbiota modulates the gut-brain axis through multiple interacting mechanisms that converge on barrier integrity, neuroimmune signalling, neural pathways, and systemic metabolism.63,64 These mechanisms acquire particular relevance in postmenopausal women, whose microbial composition and metabolic capacity are shaped by hormonal decline and chronic medication use. Notably, the gut-microbiota-brain axis in menopause has not yet been characterised despite clear evidence of menopause-related changes in brain connectivity and function.10Microbial metabolites, including SCFAs, TMA derivatives, neurotransmitter precursors, and polyphenol-derived compounds, act on enteric neurons, enteroendocrine cells, and immune cells in the intestinal mucosa, then reach the circulation or the lymphatic system to influence distant targets.37,65
SCFAs, primarily acetate, propionate, and butyrate, exert multiple effects on the brain by regulating BBB integrity, modulating microglial activation, and controlling key neuroinflammatory pathways. These microbial metabolites are key players in altering paracellular diffusion and transcytosis at neurovascular interfaces.17,66 In neurodegenerative diseases, SCFA dysregulation is common. For example, patients with Parkinson's disease show reduced faecal acetate, propionate and butyrate, whereas Alzheimer's disease is characterised by decreased butyrate and increased acetate and valerate, potentially promoting inflammation and BBB dysfunction.67,68
Microbe-driven modification of tryptophan and aromatic amino acid pathways generates ligands that influence central serotonin and catecholamine synthesis and vagal afferent signalling, providing a direct neurochemical route from gut to brain circuits.65,69 Moreover, the gut microbiota metabolises tryptophan into a diverse array of indole and indole-derived compounds (e.g., indole-3-propionic acid and indole-3-aldehyde). These microbial indoles act as signalling molecules at the intestinal and systemic levels, primarily by activating the aryl hydrocarbon receptor (AHR), thereby modulating intestinal barrier integrity, immune homeostasis, and neuroinflammatory responses.70 Notably, bacteria capable of indole production increase their activity in response to dietary substrates, and both (poly)phenols and specific dietary fibres have been shown to promote indole-generating microbial pathways by selectively enriching indole-producing taxa.71 This route provides a critical, microbiota-dependent link between diet, tryptophan metabolism, and gut-brain communication, complementing host kynurenine and serotonin pathways.
In addition, the gut microbiota transforms dietary (poly)phenols into smaller molecules, such as low-molecular-weight phenolics, urolithins, equol, enterolignans, lunularin, 8-prenylnaringenin, and others. By modulating neuroimmune signalling, oxidative stress, and estrogen pathways, these compounds may enhance cognitive and emotional resilience in postmenopausal women. These microbial derivatives frequently display superior bioavailability and exert distinct biological effects, often considered the main drivers of health outcomes. Their formation depends strongly on gut microbial composition and the individual metabotype, as well as on the (poly)phenol ingested.9,29,33,37
Changes in gut microbial community structure induced by (poly)phenols also affect the production of SCFAs, the remodelling of bile acids, and the generation of signalling molecules that regulate gut barrier integrity, vagal tone, and systemic inflammation. These shifts may help counteract the low-grade inflammation associated with menopause and support balanced communication along the gut-brain axis. However, the extent of these effects varies depending on host physiology and microbial background.33,37,42
Dysbiosis or increased gut microbial translocation elevates exposure to bacterial products such as lipopolysaccharide, driving low-grade systemic inflammation and altering microglial reactivity.66 Additionally, the microbiota regulates intestinal vascular and lymphatic function through microbial factors, such as VEGF-C, and by modulating lipid and chylomicron handling, thereby affecting the absorption and trafficking of lipophilic neuroactive compounds.72 The net effect of these mechanisms is context-dependent, with microbial composition, host age, barrier status, immune tone, and lymphatic function collectively determining whether microbiota-derived signals support homeostasis or promote neuroinflammation, metabolic dysregulation, and behavioural changes.64,72,73 Significantly, chronic pharmacological treatments, common in postmenopausal women, may further modulate microbial composition and function, altering metabolite profiles and barrier dynamics. These drug-induced shifts can interact with dietary bioactives and microbial signalling, potentially amplifying or attenuating gut-brain communication. Understanding how medication influences microbiota-derived effects is essential for interpreting intervention outcomes and designing personalised strategies.
This overlap suggests that chronic medication use, which is highly prevalent in postmenopausal women, may significantly modulate the physiological response to dietary (poly)phenols and should therefore be considered when interpreting gut-brain axis outcomes.
3. Medications and potential interactions with (poly)phenol effects: the often overlooked variable
3.1. Effects of common chronic medications on the gut microbiota
Medication use is increasingly recognised as an important, yet often overlooked, determinant of gut microbiota composition and function. The profound microbiome-disrupting effects of antibiotics are well documented. However, accumulating evidence shows that many non-antibiotic drugs commonly used in midlife and older adults also exert measurable impacts on microbial communities.74–76 Importantly, medication exposure is particularly prevalent among postmenopausal women, a population characterised by an increased burden of chronic conditions and sustained use of long-term pharmacological treatments.Epidemiological data indicate that approximately 60–70% of women in the menopausal transition and postmenopausal period regularly use at least one chronic medication, and a substantial proportion are exposed to polypharmacy.77 HRT, antidepressants, anxiolytics, sleep medications, antihypertensives, lipid-lowering agents, and antidiabetic drugs are among the most frequently prescribed treatments in this demographic group, collectively shaping a pharmacologically driven microbial environment.
Among non-antibiotic drugs, antidiabetic agents are the most extensively studied for their capacity to modulate the gut microbiota. Metformin, in particular, has been consistently shown to induce marked and reproducible shifts in microbial composition across both diabetic and non-diabetic cohorts. These changes include increased abundance of Escherichia and Bifidobacterium spp., enrichment of mucin-degrading taxa such as Akkermansia muciniphila, and stimulation of SCFA-producing bacteria.78,79 These shifts are accompanied by functional changes affecting microbial genes involved in carbohydrate metabolism and SCFA biosynthesis. GLP-1 receptor agonists alter the Bacillota (Firmicutes)/Bacteroidota (Bacteroidetes) ratio, increase the abundance of SCFA-producing taxa such as Lachnospiraceae, and modulate immune populations, reducing the frequency of Th1 lymphocytes.80,81 Although findings for SGLT2 inhibitors are more heterogeneous, several studies report increases in A. muciniphila abundance and shifts in the Bacillota/Bacteroidota ratio.82 Collectively, these microbiota alterations overlap with key mechanisms implicated in gut-brain axis signalling, including barrier integrity, immune modulation, and neuroinflammatory control.
Lipid-lowering agents, particularly statins, represent another major drug class with documented effects on the gut microbiome. Statin therapy has been associated with changes in microbial diversity, enrichment of bile acid-transforming bacteria, and alterations in microbial bile acid profiles, which in turn may influence gut barrier function, systemic inflammation, and central nervous system signalling. Long-term use of statins may impact gut microbiota diversity, potentially leading to dysbiosis.83 Paradoxically, statin therapy has also been associated with shifts in gut microbial ecology, characterised by an enrichment of health-associated taxa, including A. muciniphila and Lactobacillus spp., together with a reduction in bacterial groups linked to pro-inflammatory states.84,85 These alterations in the gut microbiota might influence intestinal barrier function by modulating tight-junction architecture and epithelial integrity, thereby affecting gut permeability and host–microbe interactions.
Similarly, antihypertensive drugs, including beta-blockers, angiotensin-converting enzyme (ACE) inhibitors, angiotensin receptor blockers, and calcium channel blockers, have been linked to distinct microbial signatures. These medications may affect microbial diversity, SCFA production, and microbial pathways involved in neurotransmitter synthesis and immune regulation.75,86 In these interventions, an increased relative abundance of butyrate-producing genera such as Roseburia has been reported,75 alongside a reduction in Faecalibacterium.86 However, mechanistic insights remain fragmented, and their potential implications for gut-brain axis function are rarely addressed explicitly.
HRT remains the most effective pharmacological approach for alleviating menopausal symptoms. Beyond its systemic endocrine effects, HRT may be considered as an important modulator of gut microbiota composition and function. Estrogens and estrogen-containing therapies influence the so-called estrobolome, a subset of gut microbial genes involved in estrogen deconjugation and recirculation, thereby shaping systemic estrogen exposure and signalling pathways.87,88
In this context, dietary (poly)phenols with estrogenic activity, especially the isoflavones daidzein and genistein, add another layer of complexity. These compounds share some structural similarity with 17β-estradiol and exhibit selective estrogen receptor modulator activity. Notably, the microbial conversion of daidzein into equol determines the biological response, such that only equol-producing individuals, i.e., the equol producers metabotype, derive maximal benefit, underscoring the central role of microbial composition in shaping hormonal and gut-brain axis outcomes.9,89 Recent evidence supports a microbiota-mediated contribution to the metabolic benefits associated with HRT. In postmenopausal women receiving hormone therapy, higher circulating estradiol and progesterone levels were accompanied by lower fasting glucose and a distinct microbial signature characterised by increased relative abundance of Prevotella and reduced levels of Escherichia, Klebsiella, and Lactobacillus, taxa previously associated with cardiometabolic risk.90
In general, despite the recognition that medications profoundly shape the gut microbiota, most evidence derives from mixed-sex cohorts or male-dominated populations, with limited stratification by menopausal status.87 As a result, the specific gut microbiome signatures associated with long-term pharmacological treatments in postmenopausal women, and their consequences for gut-brain axis signalling, remain poorly characterised. Addressing medication as a core variable is therefore essential for accurately interpreting gut-brain axis research and for developing personalised nutritional strategies in postmenopausal women.87,91 Therefore, these drug-induced alterations in microbial composition and metabolic capacity are likely to influence the gut-brain signalling, as well as the absorption, transformation, and systemic activity of dietary (poly)phenols.
3.2. Interactions between medications and the bioactivity of (poly)phenols
Medication-bioactive interactions can occur at multiple, interconnected levels, influencing the systemic exposure to dietary (poly)phenols and their biological effects on gut-brain axis pathways. Most chronic drugs commonly used in postmenopausal women, including antihypertensives, lipid-lowering agents, HRT, and sleep medications, modulate metabolic and transport processes that overlap with those governing (poly)phenol transformation, circulation, and tissue distribution. Consequently, polypharmacy represents an overlooked confounder in nutritional intervention studies and a potential modifier of the efficacy of (poly)phenol-rich diets or supplements (Fig. 2).For instance, CYP2C19 and CYP2D6 have been described as important isoforms in the metabolism of antidepressants.93 Interestingly, a wider range of dietary (poly)phenols and various gut microbiota-derived metabolites have been shown to inhibit CYP2D6 enzyme activity.94 Other examples include lipid-lowering agents, such as the statins simvastatin, lovastatin, and atorvastatin, which are highly susceptible to drug interactions, as potent CYP3A4 inhibitors markedly increase their plasma concentrations, thereby elevating the risk of toxicity.95 In this regard, (poly)phenol-rich foods and flavonoids have shown a specific inhibitory capacity against CYP3A4 in preclinical and clinical studies.96,97
Antihypertensive drugs are another major group of drugs prescribed to middle-aged women. Their metabolism involves several CYP450 isoforms, including CYP2D6, CYP2C9, CYP2D19 and CYP3A4, which metabolise most antihypertensive drugs.98 Notably, resveratrol intervention was found to inhibit the phenotypic activity of CYP3A4, CYP2D6, and CYP2C9, but induced CYP1A2 activity in healthy volunteers (Fig. 2).99
Beyond CYP-mediated metabolism, modulation of drug transporters represents an additional clinically relevant mechanism. For example, green tea-derived catechins have been shown to inhibit intestinal uptake transporters, leading to reduced plasma concentrations of specific drugs, such as nadolol, in human studies.100
Importantly, although the vast majority of these interactions have been characterised at the mechanistic level, clinical evidence supports their relevance in specific contexts. A well-established example is the interaction between polyphenol-rich grapefruit juice and drugs metabolised by CYP3A4, where inhibition of intestinal CYP3A4 leads to increased systemic drug exposure and potential toxicity.101 In addition, clinical pharmacokinetic studies have also demonstrated that catechins can significantly reduce the bioavailability of certain drugs, through inhibition of intestinal uptake transporters, including organic anion-transporting polypeptides (OATP).102
Collectively, these findings indicate that phase I-mediated interactions may meaningfully reshape the metabolic landscape in which dietary (poly)phenols and chronic medications coexist, a prevalent scenario in postmenopausal women.
Available evidence indicates that polymorphisms in UGTs, SULTs, and transporters can meaningfully influence drug exposure, efficacy, and toxicity.103,104 A similar paradigm applies to dietary (poly)phenols, for which interindividual differences in conjugation capacity shape metabolite patterns and biological responses.105
Importantly, many drugs chronically prescribed to postmenopausal women rely on the same enzymatic systems, creating potential competition at the level of phase II metabolism. As a consequence, competitive substrate interactions at the level of phase II enzymes may alter both drug disposition and (poly)phenol metabolism.106,107 Saturation or inhibition of UGTs and SULTs can shift the relative abundance of glucuronidated, sulfated, or methylated metabolites, thereby modifying their biological activity, tissue distribution, and capacity to engage gut-brain axis signalling.
Although the available clinical evidence supporting these interactions remains limited, heterogeneous, and highly compound-specific, both the studies included in this review and additional human data suggest that certain dietary (poly)phenols can modulate drug-metabolising enzymes and transport processes, supporting their potential relevance in specific contexts. Taken together, these interactions indicate that the pharmacological landscape typical of postmenopausal women may substantially modify the physiological and neurobiological effects attributed to dietary (poly)phenols, underscoring the need to interpret gut-brain outcomes within this broader context (Fig. 2).
4. Polyphenols and the gut-brain axis in postmenopausal health: an area yet to be investigated
Research on (poly)phenols to improve vasomotor symptoms, cardiometabolic parameters, and bone health during postmenopause has long attracted attention. In this context, the majority of (poly)phenol intervention studies in postmenopausal women have focused on isoflavone intake, reflecting their prominent role in this stage of women's health research. This emphasis is attributable, at least in part, to their moderate estrogenic activity and to accumulating evidence indicating that their efficacy is closely linked to intestinal biotransformation and the generation of bioactive microbial metabolites, primarily (S)-equol.108,109 This well-established example illustrates how microbial metabolism determines the physiological relevance of dietary (poly)phenols, extending its implications to pathways directly involved in gut-brain communication. However, despite this mechanistic plausibility, the implications of (poly)phenol-derived microbial metabolites for gut-brain communication in postmenopausal women remain unexamined.Despite extensive research in postmenopausal women, clinical trials have predominantly focused on peripheral endpoints, leaving the role of (poly)phenols in modulating the gut-brain axis insufficiently explored. To date, no human intervention study has investigated how dietary (poly)phenols influence gut-brain axis processes specifically in postmenopausal women.
As commented, the gut-brain axis has emerged as a central conceptual framework for understanding the bidirectional interplay between the gut microbiota, host metabolism, immune function and the central nervous system, with direct repercussions for cognition, mood regulation, stress responsiveness, and functional gastrointestinal disorders.16,61 Understanding this gap requires revisiting how the gut-brain axis operates and why postmenopause represents a uniquely sensitive context. Within this landscape, (poly)phenols have been proposed as potential modulators of gut-brain communication, exerting biological effects directly or indirectly via their gut microbial biotransformation into neuroactive and immunomodulatory metabolites. This gap is striking given that hormonal decline, inflammaging, and polypharmacy directly modulate both microbial metabolism of (poly)phenols and gut-brain signalling. However, a critical review of the existing literature on human interventions reveals that the evidence supporting these interactions is currently fragmented and lacks a clear connection to the postmenopausal context, despite the profound hormonal, immunometabolic and inflammatory changes that characterise this life stage and are known to influence gut-brain axis function.110,111
As shown in Table 1, this perspective provides a concise overview of existing human intervention studies investigating associations between (poly)phenols or (poly)phenol-rich dietary products and markers or outcomes relevant to the gut-brain axis. Notably, the lack of studies identified in this context underscores that the intersection of menopause, (poly)phenol metabolism, and gut-brain communication has not yet been addressed in human intervention research. The following targeted PubMed and Scopus search string was used: “(polyphenol OR flavan* OR flavon* OR procyan* OR ellagi* OR isoflav* OR genist* or daidz* OR catech* OR epicate* OR resveratrol OR curcum* OR hydroxycinn* OR caffeic OR anthocyan*) AND gut-brain AND (patient* OR women OR female OR volunteer* OR trial OR intervention) NOT (rat OR mouse OR mice OR animal model OR cell* OR in vitro) NOT (review or metaanalysis)”. Importantly, adding the term “menopause” yielded no results, highlighting a complete lack of human intervention studies addressing the gut-brain axis specifically in postmenopausal women.
| (Poly)phenol source | Population | Dose | Duration | Primary outcomes | Medication | Key limitations | Type of study | References |
|---|---|---|---|---|---|---|---|---|
| Abbreviations: ASD: autism spectrum disorder; EEG: electroencephalography; FP: fibre and polyphenol; FGID: functional gastrointestinal disorders; GIQLI: gastrointestinal quality of life index; GM-CSF: granulocyte-macrophage colony-stimulating factor; GSRS: gastrointestinal clinical rating scale; IBS: irritable bowel syndrome; IBS-C: irritable bowel syndrome-constipation; IBS-M: irritable bowel syndrome-mixed type; IBS-D: irritable bowel syndrome-into diarrhoea; IBS-SSS: irritable bowel syndrome-severity scoring system; IFN-γ: interferon-γ; MIP-1β, macrophage inflammatory protein-1β (CCL4); OQ45.2 (outcome questionnaire 45.2); PB: peanut butter; PHGG (partially hydrolysed guar gum); QoL: quality of life; SCFAs: short chain fatty acids; SRP: skin roasted peanuts; TNF: tumor necrosis factor; VLSCFAs: very low short chain fatty acids. | ||||||||
| Resveratrol and coumaric acid-rich SRP and PB | 63 healthy adults (age 18–33) | SRP 25 g day−1 or PB 32 g day−1 or control butter 32 g day−1 | 6 months | SRP and PB improved immediate memory; PB improved the delayed and total memory; anxiety scores reduced (SRP vs. control); ↑faecal SCFAs (acetic, propionic, butyric) and total SCFAs; ↓urinary cortisol | No medication | Limited sample size; lack of blinding; control butter still contained VLCSFAs; limited statistical power after dropouts; no microbiota data | Three-arm parallel-group randomised controlled trial | Parilli-Moser et al. (2021)112 |
| Polyphenols via an “anti-inflammatory” diet (NeuroGutPlus: fruits, vegetables, olive oil, nuts, seeds) | 30 children with ASD (age 6–17) + 12 neurotypical controls | Food package or 1 dose per day multi-strain probiotics (16 Lactobacillus/Bifidobacterium strains) | 12 weeks | ↓IFN-γ with diet; probiotics ↑IL-8 and MIP-1β, but ↓IFN-γ; immune profile stabilisation with diet | No antibiotics or immunosuppressives within the last month; stable behavioural therapy ≥3 months; no structured dietary support before trial | Limited sample size; no microbiota data; lack of microbial metabolite profiling; behavioural outcomes only; limited mechanistic insight | Randomised controlled nutritional trial | Naranjo-Galvis et al. (2025)119 |
| Quebracho (Schinopsis lorentzii) and Chestnut (Castanea sativa) tannins (ellagitannins, profisetinidins) | 156 adults with IBS (age 18–70) | 480 mg day−1 (two capsules) | 56 days | Significant improvement in IBS severity score (IBS-SSS), quality of life (IBS-QoL, GIQLI), and reduction of bloating, abdominal pain and flatulence; improved bowel movement frequency in IBS-C and IBS-D | Excluded patients on opioids or drugs affecting intestinal function; excluded recent antibiotics (last 4–6 weeks); no concurrent probiotics or supplements | Single-centre design; no microbiota data; limiting mechanistic interpretation of polyphenol effects; absence of microbial metabolite profiling | Single-centre, randomised, double-blind, placebo-controlled clinical trial | Molino et al. (2025)117 |
| Blueberries (anthocyanins; total phenolics ∼32 mg g−1; polyphenol-rich fruit) | 47 adults with FGID (IBS and/or functional dyspepsia); (age 18–60) | 30 g day−1 freeze-dried blueberry powder (2 × 15 g), equivalent to ∼180 g fresh | 14–16 weeks. 6 weeks per period (2 periods) and 2–4 weeks of wash-out | Higher proportion with abdominal symptom relief vs. placebo (53% vs. 30%, p = 0.03); improved OQ45.2 well-being/function; GSRS total/pain trends but NS; stool consistency trending to normal | Excluded antibiotics or probiotics within 2 weeks before or during the trial; no initiation of new medications during study; background meds otherwise stable/not reported | Limited sample size; no microbiota data; carry-over effects; limited mechanistic gut-brain axis biomarkers | Randomised double-blind placebo-controlled, crossover clinical trial | Wilder-Smith et al. (2023)116 |
| Extracts from Aronia melanocarpa (black chokeberry) and Sambucus nigra (elderberry) (Fenactive® blend: 25% total polyphenols; 15% anthocyanins) | 47 adults with IBS (age 18–65) | 200 mg day−1 combination of probiotics, PHGG 5 g day−1 and (poly)phenol blend | 2 months | QoL improved most in the full synbiotic + polyphenol group (most significant dysphoria decrease); ↓TNF-α and ↓GM-CSF (Groups II/III); ↑total SCFAs (group III vs. placebo); stool consistency improved (IBS-D firmer; IBS-C softer) | Concomitant medications not detailed | Limited sample size; placebo maltodextrin may not be inert; no microbiota data | Randomised double-blind placebo-controlled clinical trial | Wierzbicka et al. (2025)118 |
| Blend of fermentable fibres (oligofructose-enriched inulin, Bimuno® GOS, resistant starch) + polyphenols (cocoa, green tea, blueberry, cranberry extracts) in snack bars | 26 young adults (age 17–39) | 2 or 4 bars per day | 14 days | Altitude increased mood disturbance; FP did not improve cognition; FP attenuated altitude-induced anger but increased confusion | Excluded if regular use of medication | Limited sample size; no microbiota data; lack of microbial metabolite profiling | Randomised, double-blind, placebo-controlled crossover trial | Beckner et al. (2025)114 |
| Blackcurrant anthocyanins (151 mg) + pine bark proanthocyanidins (part of 308 mg total polyphenols) | 40 healthy females (age 18–45) | 300 mL day−1 | 4 weeks | No effect on stress reactivity; secondary improvements in working memory (letter retrieval) and mood (tension/anxiety; anger/hostility) | Excluded if medication interferes with digestive processes (e.g., laxatives) | Limited sample size; lack of microbial metabolite profiling | Randomised, double-blind, placebo-controlled crossover trial | Gillies et al. (2024)113 |
| Anthocyanin-rich extract from black carrot (Daucus carota) | 50 older adults with dementia | 300 mg anthocyanins daily (125 mL water) | 12 weeks | ↑EEG connectivity; no significant change in overall gut microbiota diversity; genus-level trends: ↑Alistipes, Streptococcus thermophilus, Flavonifractor, and ↓Hungatella | Cognitive enhancers permitted (Donepezil, Rivastigmine, Memantine, Ginkgo biloba extract, or Piracetam) | Lack of a placebo-controlled design; small microbiota subsample; and absence of microbial metabolite profiling | Single-arm pre-post exploratory pilot | Muduroglu-Kirmizibekmez et al. (2025)120 |
| Astragaloside IV extract from Astragalus membranaceus plant | 20 healthy adults (age 19–27) | Single dose 150 mg | 1 day | ↑EEG connectivity:↓delta, theta, beta and gamma bands and ↑alpha band, ↑alpha/beta, ↑theta/beta ↓delta/alpha | Excluded if regular use of medication | Lack of a placebo-controlled design; limited sample size; lack of microbiota profiling; and absence of microbial metabolite profiling | Single-arm pre-post exploratory pilot | Muduroglu-Kirmizibekmez et al. (2025)115 |
4.1. Human intervention studies exploring the gut-brain axis and (poly)phenols
As outlined in Table 1, human intervention studies evaluating the relationship between (poly)phenols and outcomes relevant to the gut-brain axis have been conducted in heterogeneous study populations, including healthy young adults,112–115 patients with disorders of gut-brain interaction, such as irritable bowel syndrome (IBS) and other functional gastrointestinal disorders (FGID),116–118 as well as specific clinical populations, such as children with autism spectrum disorder (ASD)119 or older adults with dementia.120Trials conducted in healthy young populations allow exploration of the possible effects of (poly)phenols on cognition, mood, and stress under relatively stable physiological conditions, characterised by low inflammation and the absence of polypharmacy.112,113, These studies have reported improvements in specific cognitive domains, including immediate or working memory, as well as reductions in anxiety and anger. These populations are particularly suited for detecting subtle behavioural effects in the absence of major confounding factors. However, these findings cannot be extrapolated to postmenopausal women, whose endocrine and inflammatory milieu differs profoundly.
In contrast, studies in IBS and FGID, in which gut-brain dysregulation is already established, demonstrate consistent improvements in symptom severity, quality of life, and dysphoria following intervention with tannins, blueberries, or (poly)phenol-enriched symbiotic combinations.116–118 These outcomes reinforce the relevance of (poly)phenols as modulators of gut-brain signalling in populations exhibiting heightened visceral sensitivity and altered microbiota composition. However, these pathophysiological contexts differ fundamentally from postmenopause, where gut-brain alterations arise from endocrine and metabolic transitions rather than visceral hypersensitivity.
In parallel, the incorporation of immune profiles119 and neurophysiological measures such as electroencephalogram (EEG) connectivity115,120 has broadened the range of biological indicators explored across different clinical contexts. Existing models have primarily been developed in pathophysiological or life-stage contexts that differ substantially from the climacteric stage of postmenopause. For instance, Gillies et al.113 conducted a study in women aged 18–45 years, representing a hormonal and metabolic context that is not representative of the postmenopausal physiological environment, where estrogens decline, low-grade inflammation increases, and polypharmacy is frequent, all of which may affect both the bioavailability and bioactivity of (poly)phenols. Thus, these differences underscore the need for dedicated studies in postmenopausal women.
4.2. Gut-brain axis outcomes assessed
A thorough examination of the outcomes enumerated in Table 1 shows that gut-brain axis outcomes have been assessed using a broad range of functional, psychological, immunological, and neurophysiological endpoints, with notable variability across populations and intervention designs.In healthy adult populations, outcomes of the gut-brain axis are predominantly evaluated using cognitive and affective measures. Interventions based on (poly)phenol-rich foods or extracts have been associated with improvements in immediate, delayed, or working memory, as well as reductions in anxiety, anger, or tension-related mood states.112–114 However, these endpoints have never been assessed in postmenopausal women, despite their heightened vulnerability to cognitive and emotional changes. Among these studies, Parilli-Moser et al. (2021) provide one of the most integrative assessments of the axis, linking cognitive and emotional improvements to changes in microbial-derived metabolites. Specifically, increased faecal concentrations of SCFAs, including acetate, propionate, and butyrate, were observed following peanut-based interventions, alongside reductions in urinary cortisol levels, supporting a microbiota- or gut-brain interaction pathway (Table 1). In contrast, other trials in healthy individuals report behavioural or mood-related outcomes without accompanying microbial or metabolic data.113,114
In populations with IBS and FGID, outcome assessment has primarily relied on validated clinical questionnaires capturing gastrointestinal symptom severity, abdominal pain, bloating, bowel habits, and health-related quality of life.116–118 These endpoints are highly relevant for reflecting the functional expression of gut-brain dysregulation, although they provide limited insight into the underlying biological mechanisms. Notably, a symbiotic intervention combining probiotics, fermentable fibres, and polyphenols reported improvements in quality of life and dysphoria, and also increased total SCFAs levels, positioning microbial metabolites as potential mediators of symptom improvement in this context.118 However, these outcomes do not capture the endocrine-immune interactions characteristic of postmenopause.
Beyond functional and psychological outcomes, some studies extend gut-brain axis assessment to immune and neurophysiological domains. In children with ASD, (poly)phenol intake through an anti-inflammatory diet was associated with modulation of systemic immune markers, including reductions in the pro-inflammatory cytokine IFN-γ and a significant increase in IL-8 and MIP-1β, suggesting an immune-mediated gut-brain pathway.119 Similarly, in both older adults with dementia and healthy adults, supplementation with an anthocyanin-rich black carrot extract and Astragaloside IV was linked to increased EEG connectivity across multiple frequency bands, indicating central nervous system responsiveness to dietary (poly)phenols.115–120 These neurophysiological changes occurred without major shifts in overall gut microbiota diversity and without concurrent assessment of SCFA levels, highlighting both the potential and the current mechanistic limitations of such approaches. Such neurophysiological markers could be highly informative in postmenopausal research, but they remain entirely unexplored in this population. Nevertheless, compositional changes at the genus level were observed in the Muduroglu-Kirmizibekmez et al. (2025)120 study, including increases in Alistipes, Streptococcus thermophilus, and Flavonifractor, accompanied by a reduction in Hungatella, suggesting potential links with SCFA metabolism and inflammatory regulation.
Taken together, the results presented in Table 1 show that the existing clinical evidence primarily captures the downstream or functional manifestations of the gut-brain axis, including symptoms, mood, cognition, immune signalling and brain connectivity. However, only a limited number of studies incorporate microbial-derived metabolites, particularly SCFAs, as intermediate biomarkers linking gut and brain responses. Inconsistent inclusion of microbial, metabolic and central endpoints within the same experimental framework is a major gap in the field, limiting our comprehensive understanding of how (poly)phenols modulate gut-brain communication. This gap is especially limiting in postmenopausal women, whose gut-brain axis is shaped by hormonal decline, inflammaging and medication use.
4.3. Gut microbiota and microbial metabolites: a critical mechanistic gap
The gut microbiota and its metabolites are central mediators of gut-brain axis communication, as outlined in sections 1.2 and 2.2.11,14,63,64 Nevertheless, the clinical trials summarised in Table 1 reveal that these mechanisms are rarely addressed in human (poly)phenol interventions despite their established relevance. This omission is particularly limiting for postmenopausal women, whose gut microbial metabolism of (poly)phenols is shaped by hormonal decline and medication use.91Across the studies included in Table 1, gut-brain axis outcomes are predominantly assessed through functional or symptomatic endpoints. In contrast, microbial and metabolic intermediates are largely overlooked. Only Parilli-Moser et al. (2021) directly quantified microbial-derived metabolites, reporting increased faecal SCFAs.112 These findings align with evidence that SCFAs support intestinal and blood–brain barrier integrity and modulate neuroinflammatory and stress-related pathways, yet microbiota composition was not assessed, limiting mechanistic resolution.21,66,67 In studies involving IBS and FGID, microbial metabolites, gut microbial composition and functionality were not evaluated, except for the symbiotic intervention, which reported increased total SCFAs and reduced inflammatory cytokines.118 Given that SCFA production declines with age and is sensitive to medication, their omission in postmenopausal research represents a major blind spot.
Beyond SCFAs, none of the trials assessed other microbiota-derived molecules known to influence the gut-brain axis, such as tryptophan metabolites, modified bile acids, or low-molecular-weight phenolic derivatives generated from dietary (poly)phenols.9,33,65 This omission is particularly relevant in postmenopausal health, where hormonal decline, inflammaging, and frequent medication use reshape microbial metabolism and may alter responsiveness to dietary interventions.19,26,27,87
Overall, there is a significant gap in our understanding of the mechanisms by which gut microbiota-derived metabolites regulate the gut-brain axis, particularly given their limited assessment in current human (poly)phenol trials. Integrating analyses of gut microbiota composition and functionality, along with its derived metabolite profiling, is crucial for elucidating causal pathways and advancing targeted dietary strategies for gut-brain health. Notably, this gap becomes even more evident in postmenopausal women, for whom no clinical research is currently available in this area. Addressing this mechanistic gap is essential for developing precision nutrition strategies tailored to the physiological and pharmacological landscape of postmenopausal women.
5. Conclusions, research gaps, and perspectives
Menopause is a distinct biological stage in women in which hormonal decline, chronic low-grade inflammation, and metabolic alterations increase cardiometabolic risk, anxiety, and vasomotor symptoms, thereby contributing to the frequent polypharmacy observed during this stage. Collectively, these changes remodel gut microbiota composition and metabolic activity, shaping immune, endocrine, and neural communication pathways that are central to the gut-brain axis function. Within this altered physiological landscape, the gut microbiota transforms dietary (poly)phenols into distinct bioactive metabolites (e.g., urolithin A or equol) that may influence cognitive, affective, and cardiometabolic resilience. Although this mechanistic plausibility is strong, the hypothesis linking (poly)phenols, the gut-brain axis, and menopause remains untested in targeted human intervention studies.The still-limited evidence on (poly)phenols and the gut-brain axis comes primarily from mixed, unhealthy populations or younger adults, and only a minority of studies include detailed profiling of gut microbial composition, metabolite characterisation, or functional pathway analysis. Greater mechanistic resolution is needed to identify (poly)phenol-related gut microbial metabotypes that determine individual responsiveness to dietary phenolic precursors and to interpret intervention outcomes with precision. A further critical gap is the limited consideration of pharmacotherapy. Chronic medications commonly prescribed during postmenopause substantially modify gut microbial ecology and host molecular targets shared with dietary (poly)phenols, yet most dietary interventions do not systematically report or control for medication use. Incorporating pharmacological profiling is therefore essential for understanding gut microbial metabolic variability, identifying potential interactions, and accurately assessing the effects of dietary interventions.
Future research should integrate dietary (poly)phenols, gut microbiota, and pharmacology within the specific physiological context of postmenopause. Stratification based on (poly)phenol-related microbial metabotypes, such as equol or urolithin A producers, together with medication exposure, will be necessary to advance personalised health strategies. Clinical trials should adopt multi-omics approaches to characterise microbial composition, microbial-derived metabolites, immune and neuroimmune mediators, bile acid signatures, and cognitive and emotional outcomes, explicitly evaluating how (poly)phenol-derived metabolites modulate gut-brain axis pathways in postmenopausal women. These approaches can be strengthened by incorporating repeated sampling across the menopausal transition or by using crossover interventions, thereby helping to capture steady-state production of microbial-derived phenolics. Multi-omics analyses may also benefit from biomarker panels that combine circulating microbial-derived phenolics, bile acid profiles, short-chain fatty acids, and markers of epithelial integrity and inflammation, thereby improving mechanistic resolution. Given the relevance of estrogen-dependent microbial pathways in this population, incorporating measures of estrobolome activity may help clarify how estrogen metabolism interacts with (poly)phenol biotransformation and downstream neuroendocrine responses. Including neurophysiological endpoints, such as markers of blood–brain barrier integrity, neuroinflammation, or functional connectivity, would further strengthen the link between microbial and metabolic changes and brain-level mechanisms. Finally, longitudinal designs may help clarify how the menopausal transition reshapes gut microbial function and modulates responsiveness to (poly)phenol-based interventions, paving the way for precision strategies to support long-term health in postmenopausal women.
Author contributions
J. C. E.: conceptualisation, supervision, funding acquisition, writing original draft, and validation. M. A. A.-G.: conceptualisation, writing original draft, and validation. M. G.-N.: writing original draft, methodology, and investigation. M. P. J.-O., M. E. M.-N., and M. R.-V.: formal analysis, investigation, and methodology. All authors: writing, review, and editing.Conflicts of interest
The authors declare no conflict of interest.Data availability
No primary research results, software, or codes have been included, and no new data were generated or analysed as part of this review.Acknowledgements
This work has been funded by the Project PID2022-136419OB-100 (Ministerio de Ciencia, Innovación y Universidades, MICIU, AEI/10.13039/501100011033, Spain), with co-funding from the European Union through “ERDF A way of making Europe”. M.G.-N. acknowledges support from a Juan de la Cierva Postdoctoral Contract (JDC2023-051288-I) funded by MCIU/AEI/10.13039/501100011033 and the ESF+.References
- N. Santoro, C. Roeca, B. A. Peters and G. Neal-Perry, The Menopause Transition: Signs, Symptoms, and Management Options, J. Clin. Endocrinol. Metab., 2021, 106, 1–15 CrossRef PubMed.
- M. Fasero and P. J. Coronado, Cardiovascular Disease Risk in Women with Menopause, J. Clin. Med., 2025, 14, 3663 CrossRef CAS.
- M. Peate, T. L. Johnson, N. E. Avis and M. Hickey, Addressing sociodemographic, socioeconomic, and gendered disparities for equity in menopause care, Cell Rep. Med., 2024, 5, 101616 CrossRef PubMed.
- S. Young-Moss, A. Lee and J. A. Simon, Advances in Pharmacotherapy for Menopausal Vasomotor Symptoms, Drugs, 2025, 85, 1363–1379 CrossRef CAS PubMed.
- N. F. Col, The impact of risk status, preexisting morbidity, and polypharmacy on treatment decisions concerning menopausal symptoms, Am. J. Med., 2005, 118, 155–162 CrossRef.
- P. Gardiner, M. B. Stargrove and T. L. Dog, Concomitant use of prescription medications and dietary supplements in menopausal women: an approach to provider preparedness, Maturitas, 2011, 68, 251–255 CrossRef PubMed.
- N. Koppel, V. Maini Rekdal and E. P. Balskus, Chemical transformation of xenobiotics by the human gut microbiota, Science, 2017, 356, eaag2770 CrossRef PubMed.
- K. R. Trepka, C. A. Olson, V. Upadhyay, C. Zhang and P. J. Turnbaugh, Pharma[e]cology: How the Gut Microbiome Contributes to Variations in Drug Response, Annu. Rev. Pharmacol. Toxicol., 2025, 65, 355–373 CrossRef CAS.
- J. C. Espín, M. P. Jarrín-Orozco, L. Osuna-Galisteo, M. Á. Ávila-Gálvez, M. Romo-Vaquero and M. V. Selma, Perspective on the Coevolutionary Role of Host and Gut Microbiota in Polyphenol Health Effects: Metabotypes and Precision Health, Mol. Nutr. Food Res., 2024, 68, e2400526 CrossRef PubMed.
- M. Liaquat, A. M. Minihane, D. Vauzour and M. G. Pontifex, The gut microbiota in menopause: Is there a role for prebiotic and probiotic solutions?, Post Reprod. Health, 2025, 31, 105–114 CrossRef PubMed.
- M. Schaeffer, D. J. Hodson and P. Mollard, The blood-brain barrier as a regulator of the gut-brain axis, Front. Horm. Res., 2014, 42, 29–49 Search PubMed.
- K. Rathore, N. Shukla, S. Naik, K. Sambhav, K. Dange, D. Bhuyan and Q. M. I. Haq, The Bidirectional Relationship Between the Gut Microbiome and Mental Health: A Comprehensive Review, Cureus, 2025, 17, e80810 Search PubMed.
- N. Faraji, B. Payami, N. Ebadpour and A. Gorji, Vagus nerve stimulation and gut microbiota interactions: A novel therapeutic avenue for neuropsychiatric disorders, Neurosci. Biobehav. Rev., 2025, 169, 105990 CrossRef CAS PubMed.
- X. Su, Y. Gao and R. Yang, Gut microbiota derived bile acid metabolites maintain the homeostasis of gut and systemic immunity, Front. Immunol., 2023, 14, 1127743 CrossRef CAS PubMed.
- J. Wang, N. Zhu, X. Su, Y. Gao and R. Yang, Gut-Microbiota-Derived Metabolites Maintain Gut and Systemic Immune Homeostasis, Cells, 2023, 12, 793 CrossRef CAS.
- N. M. Swer, B. S. Venkidesh, T. S. Murali and K. D. Mumbrekar, Gut microbiota-derived metabolites and their importance in neurological disorders, Mol. Biol. Rep., 2023, 50, 1663–1675 CrossRef CAS PubMed.
- L. Láng, S. McArthur, A. S. Lazar, L. Pourtau, D. Gaudout, M. G. Pontifex, M. Müller and D. Vauzour, Dietary (Poly)phenols and the Gut-Brain Axis in Ageing, Nutrients, 2024, 16, 1500 CrossRef PubMed.
- S. M. Jabbari-Shiadeh, W. K. Chan, S. Rasmusson, N. Hassan, S. Joca, L. Westberg, A. Elfvin, C. Mallard and M. Ardalan, Bidirectional crosstalk between the gut microbiota and cellular compartments of brain: Implications for neurodevelopmental and neuropsychiatric disorders, Transl. Psychiatry, 2025, 15, 278 CrossRef.
- P. C. Calder, N. Bosco, R. Bourdet-Sicard, L. Capuron, N. Delzenne, J. Doré, C. Franceschi, M. J. Lehtinen, T. Recker, S. Salvioli and F. Visioli, Health relevance of the modification of low grade inflammation in ageing (inflammageing) and the role of nutrition, Ageing Res. Rev., 2017, 40, 95–119 CrossRef CAS PubMed.
- T. Fulop, A. Larbi, G. Pawelec, A. Khalil, A. A. Cohen, K. Hirokawa, J. M. Witkowski and C. Franceschi, Immunology of Aging: the Birth of Inflammaging, Clin. Rev. Allergy Immunol., 2023, 64, 109–122 CrossRef CAS PubMed.
- V. Braniste, M. Al-Asmakh, C. Kowal, F. Anuar, A. Abbaspour, M. Tóth, A. Korecka, N. Bakocevic, L. G. Ng, P. Kundu, B. Gulyás, C. Halldin, K. Hultenby, H. Nilsson, H. Hebert, B. T. Volpe, B. Diamond and S. Pettersson, The gut microbiota influences blood-brain barrier permeability in mice, Sci. Transl. Med., 2014, 6, 263ra158 Search PubMed.
- N. Bosco and M. Noti, The aging gut microbiome and its impact on host immunity, Genes Immun., 2021, 22, 289–303 CrossRef PubMed.
- E. Fock and R. Parnova, Mechanisms of Blood-Brain Barrier Protection by Microbiota-Derived Short-Chain Fatty Acids, Cells, 2023, 12, 657 CrossRef CAS PubMed.
- Q. A. Khan, M. Asad, A. H. Ali, A. M. Farrukh, U. Naseem, B. Semakieh, Y. Levin Carrion and M. Afzal, Gut microbiota metabolites and risk of major adverse cardiovascular events and death: A systematic review and meta-analysis, Medicine, 2024, 103, e37825 CrossRef CAS PubMed.
- L. Hoyles, M. G. Pontifex, I. Rodriguez-Ramiro, M. A. Anis-Alavi, K. S. Jelane, T. Snelling, E. Solito, S. Fonseca, A. L. Carvalho, S. R. Carding, M. Müller, R. C. Glen, D. Vauzour and S. McArthur, Regulation of blood-brain barrier integrity by microbiome-associated methylamines and cognition by trimethylamine N-oxide, Microbiome, 2021, 9, 235 CrossRef CAS.
- X. Xie, J. Song, Y. Wu, M. Li, W. Guo, S. Li and Y. Li, Study on gut microbiota and metabolomics in postmenopausal women, BMC Womens. Health, 2024, 24, 608 CrossRef CAS PubMed.
- G. Marano, S. Rossi, S. Sfratta, G. Traversi, F. M. Lisci, M. B. Anesini, R. Pola, A. Gasbarrini, E. Gaetani and M. Mazza, Gut Microbiota: A New Challenge in Mood Disorder Research, Life, 2025, 15, 593 CrossRef CAS.
- H. Wang, F. Shi, L. Zheng, W. Zhou, B. Mi, S. Wu and X. Feng, Gut microbiota has the potential to improve health of menopausal women by regulating estrogen, Front. Endocrinol., 2025, 16, 1562332 CrossRef.
- G. Williamson, C. D. Kay and A. Crozier, The Bioavailability, Transport, and Bioactivity of Dietary Flavonoids: A Review from a Historical Perspective, Compr. Rev. Food Sci. Food Saf., 2018, 17, 1054–1112 CrossRef PubMed.
- A. Sanches-Silva, L. Testai, S. F. Nabavi, M. Battino, K. Pandima Devi, S. Tejada, A. Sureda, S. Xu, B. Yousefi, M. Majidinia, G. L. Russo, T. Efferth, S. M. Nabavi and M. H. Farzaei, Therapeutic potential of polyphenols in cardiovascular diseases: Regulation of mTOR signaling pathway, Pharmacol. Res., 2020, 152, 104626 CrossRef CAS.
- L. E. Monfoulet, T. Ruskovska, V. Ajdžanović, J. Havlik, D. Vauzour, B. Bayram, I. Krga, K. F. Corral-Jara, E. Kistanova, D. Abadjieva, M. Massaro, E. Scoditti, E. Deligiannidou, C. Kontogiorgis, A. Arola-Arnal, E. M. van Schothorst, C. Morand and D. Milenkovic, Molecular Determinants of the Cardiometabolic Improvements of Dietary Flavanols Identified by an Integrative Analysis of Nutrigenomic Data from a Systematic Review of Animal Studies, Mol. Nutr. Food Res., 2021, 65, e2100227 CrossRef.
- R. García-Villalba, F. A. Tomás-Barberán, C. E. Iglesias-Aguirre, J. A. Giménez-Bastida, A. González-Sarrías, M. V. Selma and J. C. Espín, Ellagitannins, urolithins, and neuroprotection: Human evidence and the possible link to the gut microbiota, Mol. Aspects Med., 2023, 89, 101109 CrossRef.
- C. E. Iglesias-Aguirre, A. Cortés-Martín, M. Á. Ávila-Gálvez, J. A. Giménez-Bastida, M. V. Selma, A. González-Sarrías and J. C. Espín, Main drivers of (poly)phenol effects on human health: metabolite production and/or gut microbiota-associated metabotypes?, Food Funct., 2021, 12, 10324–10355 RSC.
- C. Manach, A. Scalbert, C. Morand, C. Rémésy and L. Jiménez, Polyphenols: food sources and bioavailability, Am. J. Clin. Nutr., 2004, 79, 727–747 CrossRef CAS PubMed.
- J. C. Espín, A. González-Sarrías and F. A. Tomás-Barberán, The gut microbiota: A key factor in the therapeutic effects of (poly)phenols, Biochem. Pharmacol., 2017, 139, 82–93 CrossRef.
- A. Cortés-Martín, M. V. Selma, F. A. Tomás-Barberán, A. González-Sarrías and J. C. Espín, Where to Look into the Puzzle of Polyphenols and Health? The Postbiotics and Gut Microbiota Associated with Human Metabotypes, Mol. Nutr. Food Res., 2020, 64, e1900952 CrossRef.
- H. Cheng, D. Zhang, J. Wu, J. Liu, Y. Zhou, Y. Tan, W. Feng and C. Peng, Interactions between gut microbiota and polyphenols: A mechanistic and metabolomic review, Phytomedicine, 2023, 119, 154979 CrossRef CAS PubMed.
- Q. Li and T. Van de Wiele, Gut microbiota as a driver of the interindividual variability of cardiometabolic effects from tea polyphenols, Crit. Rev. Food Sci. Nutr., 2023, 63, 1500–1526 CrossRef CAS.
- K. D. Setchell and C. Clerici, Equol: history, chemistry, and formation, J. Nutr., 2010, 140, 1355S–1362S CrossRef CAS.
- J. W. Daily, B. S. Ko, J. Ryuk, M. Liu, W. Zhang and S. Park, Equol Decreases Hot Flashes in Postmenopausal Women: A Systematic Review and Meta-Analysis of Randomised Clinical Trials, J. Med. Food, 2019, 22, 127–139 CrossRef PubMed.
- J. I. Jiménez-Loygorri, B. Villarejo-Zori, Á. Viedma-Poyatos, J. Zapata-Muñoz, R. Benítez-Fernández, M. D. Frutos-Lisón, F. A. Tomás-Barberán, J. C. Espín, E. Area-Gómez, A. Gomez-Duran and P. Boya, Mitophagy curtails cytosolic mtDNA-dependent activation of cGAS/STING inflammation during aging, Nat. Commun., 2024, 15, 830 CrossRef PubMed.
- R. G. Pushpass, S. Alzoufairi, K. G. Jackson and J. A. Lovegrove, Circulating bile acids as a link between the gut microbiota and cardiovascular health: impact of prebiotics, probiotics and polyphenol-rich foods, Nutr. Res. Rev., 2022, 35, 161–180 CrossRef CAS PubMed.
- S. Bernardi, C. Del Bo’, M. Marino, G. Gargari, A. Cherubini, C. Andrés-Lacueva, N. Hidalgo-Liberona, G. Peron, R. González-Dominguez, P. Kroon, B. Kirkup, M. Porrini, S. Guglielmetti and P. Riso, Polyphenols and Intestinal Permeability: Rationale and Future Perspectives, J. Agric. Food Chem., 2020, 68, 1816–1829 CrossRef CAS.
- J. Godos, A. Micek, P. Mena, D. Del Rio, F. Galvano, S. Castellano and G. Grosso, Dietary (Poly)phenols and Cognitive Decline: A Systematic Review and Meta-Analysis of Observational Studies, Mol. Nutr. Food Res., 2024, 68, e2300472 CrossRef PubMed.
- S. R. Kopalli, T. Behl, A. Kyada, M. M. Rekha, M. Kundlas, P. Rani, D. Nathiya, K. Satyam Naidu, M. Gulati, M. Bhise, P. Gupta, P. Wal, M. Fareed, S. Ramniwas, S. Koppula and A. Gasmi, Synaptic plasticity and neuroprotection: The molecular impact of flavonoids on neurodegenerative disease progression, Neuroscience, 2025, 569, 161–183 CrossRef CAS PubMed.
- I. Grabska-Kobyłecka, P. Szpakowski, A. Król, D. Książek-Winiarek, A. Kobyłecki, A. Głąbiński and D. Nowak, Polyphenols and Their Impact on the Prevention of Neurodegenerative Diseases and Development, Nutrients, 2023, 15, 3454 CrossRef PubMed.
- S. Dobani, L. K. Pourshahidi, N. G. Ternan, G. J. McDougall, G. Pereira-Caro, L. Bresciani, P. Mena, T. M. Almutairi, A. Crozier, K. M. Tuohy, D. Del Rio and C. I. R. Gill, A review on the effects of flavan-3-ols, their metabolites, and their dietary sources on gut barrier integrity, Food Funct., 2025, 16, 815–830 RSC.
- G. Serrelli and M. Deiana, Role of Dietary Polyphenols in the Activity and Expression of Nitric Oxide Synthases: A Review, Antioxidants, 2023, 12, 147 CrossRef PubMed.
- S. Jeong, S. Lee, W. J. Choi, U. D. Sohn and W. Kim, The Effect of Polyphenols Isolated from Cynanchi wilfordii Radix with Anti-inflammatory, Antioxidant, and Anti-bacterial Activity, Korean J. Physiol. Pharmacol., 2015, 19, 151–158 CrossRef CAS PubMed.
- J. Feng, Y. Zheng, M. Guo, I. Ares, M. Martínez, B. Lopez-Torres, M. R. Martínez-Larrañaga, X. Wang, A. Anadón and M. A. Martínez, Oxidative stress, the blood-brain barrier and neurodegenerative diseases: The critical beneficial role of dietary antioxidants, Acta Pharm. Sin. B, 2023, 13, 3988–4024 CrossRef CAS PubMed.
- S. Y. Bu, M. Lerner, B. J. Stoecker, E. Boldrin, D. J. Brackett, E. A. Lucas and B. J. Smith, Dried plum polyphenols inhibit osteoclastogenesis by downregulating NFATc1 and inflammatory mediators, Calcif. Tissue Int., 2008, 82, 475–488 CrossRef CAS PubMed.
- S. Hooshmand, A. Kumar, J. Y. Zhang, S. A. Johnson, S. C. Chai and B. H. Arjmandi, Evidence for anti-inflammatory and antioxidative properties of dried plum polyphenols in macrophage RAW 264.7 cells, Food Funct., 2015, 6, 1719–1725 RSC.
- J. L. Graef, E. Rendina-Ruedy, E. K. Crockett, P. Ouyang, L. Wu, J. B. King, R. C. Cichewicz, D. Lin, E. A. Lucas and B. J. Smith, Osteoclast Differentiation is Downregulated by Select Polyphenolic Fractions from Dried Plum via Suppression of MAPKs and Nfatc1 in Mouse C57BL/6 Primary Bone Marrow Cells, Curr. Dev. Nutr., 2017, 1, e000406 CrossRef PubMed.
- F. Mirza, J. Lorenzo, H. Drissi, F. Y. Lee and D. Y. Soung, Dried plum alleviates symptoms of inflammatory arthritis in TNF transgenic mice, J. Nutr. Biochem., 2018, 52, 54–61 CrossRef CAS PubMed.
- F. Biasi, M. Astegiano, M. Maina, G. Leonarduzzi and P. Poli, Polyphenol supplementation as a complementary medicinal approach to treating inflammatory bowel disease, Curr. Med. Chem., 2011, 18, 4851–4865 CrossRef CAS PubMed.
- M. S. Uddin, S. Hasana, J. Ahmad, M. F. Hossain, M. M. Rahman, T. Behl, A. Rauf, A. Ahmad, A. Hafeez, A. Perveen and G. M. Ashraf, Anti-Neuroinflammatory Potential of Polyphenols by Inhibiting NF-κB to Halt Alzheimer's Disease, Curr. Pharm. Des., 2021, 27, 402–414 CAS.
- A. A. Mamun, C. Shao, P. Geng, S. Wang and J. Xiao, Polyphenols Targeting NF-κB Pathway in Neurological Disorders: What We Know So Far?, Int. J. Biol. Sci., 2024, 20, 1332–1355 CrossRef PubMed.
- C. Song, Y. Zhang, L. Cheng, M. Shi, X. Li, L. Zhang and H. Zhao, Tea polyphenols ameliorates memory decline in aging model rats by inhibiting brain TLR4/NF-κB inflammatory signaling pathway caused by intestinal flora dysbiosis, Exp. Gerontol., 2021, 153, 111476 CrossRef CAS PubMed.
- H. Q. Wu, E. F. R. Pereira, J. P. Bruno, R. Pellicciari and E. X. Albuquerque, The Astrocyte-Derived α7 Nicotinic Receptor Antagonist Kynurenic Acid Controls Extracellular Glutamate Levels in the Prefrontal Cortex, J. Mol. Neurosci., 2010, 40, 204–210 CrossRef CAS PubMed.
- W. Roth, K. Zadeh, R. Vekariya, Y. Ge and M. Mohamadzadeh, Tryptophan Metabolism and Gut-Brain Homeostasis, Int. J. Mol. Sci., 2021, 22, 2973 CrossRef CAS.
- Q. Wang, H. Huang, Y. Yang, X. Yang, X. Li, W. Zhong, B. Wen, F. He and J. Li, Reinventing gut health: leveraging dietary bioactive compounds for the prevention and treatment of diseases, Front. Nutr., 2024, 11, 1491821 CrossRef PubMed.
- X. Si, J. Bi, Q. Chen, H. Cui, Y. Bao, J. Tian, C. Shu, Y. Wang, H. Tan and W. Zhang, et al., Effect of Blueberry Anthocyanin-Rich Extracts on Peripheral and Hippocampal Antioxidant Defensiveness: The Analysis of the Serum Fatty Acid Species and Gut Microbiota Profile, J. Agric. Food Chem., 2021, 69, 3658–3666 CrossRef CAS PubMed.
- J. F. Cryan, K. J. O'Riordan, C. S. M. Cowan, K. V. Sandhu, T. F. S. Bastiaanssen, M. Boehme, M. G. Codagnone, S. Cussotto, C. Fulling and A. V. Golubeva, et al., The Microbiota-Gut-Brain Axis, Physiol. Rev., 2019, 99, 1877–2013 CrossRef CAS PubMed.
- K. J. O'Riordan, G. M. Moloney, L. Keane, G. Clarke and J. F. Cryan, The gut microbiota-immune-brain axis: Therapeutic implications, Cell Rep. Med., 2025, 6, 101982 CrossRef PubMed.
- Y. P. Silva, A. Bernardi and R. L. Frozza, The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication, Front. Endocrinol., 2020, 11, 25 CrossRef PubMed.
- Q. Cao, M. Shen, R. Li, Y. Liu, Z. Zeng, J. Zhou, D. Niu, Q. Zhang, R. Wang, J. Yao and G. Zhang, Elucidating the specific mechanisms of the gut-brain axis: the short-chain fatty acids-microglia pathway, J. Neuroinflammation, 2025, 22, 133 CrossRef PubMed.
- M. Marizzoni, A. Cattaneo, P. Mirabelli, C. Festari, N. Lopizzo, V. Nicolosi, E. Mombelli, M. Mazzelli, D. Luongo and D. Naviglio, Short-Chain Fatty Acids and Lipopolysaccharide as Mediators Between Gut Dysbiosis and Amyloid Pathology in Alzheimer's Disease, J. Alzheimer's Dis., 2020, 78, 683–697 CAS.
- T. Shen, Y. Yue, T. He, C. Huang, B. Qu, W. Lv and H. Y. Lai, The Association Between the Gut Microbiota and Parkinson's Disease, a Meta-Analysis, Front. Aging Neurosci., 2021, 13, 636545 CrossRef CAS PubMed.
- A. Parker, S. Fonseca and S. R. Carding, Gut microbes and metabolites as modulators of blood-brain barrier integrity and brain health, Gut Microbes, 2020, 11, 135–157 CrossRef PubMed.
- C. J. G. Pinto, M. Á. Ávila-Gálvez, Y. Lian, P. Moura-Alves and C. Nunes dos Santos, Targeting the aryl hydrocarbon receptor by gut phenolic metabolites: A strategy towards gut inflammation, Redox Biol., 2023, 61, 102622 CrossRef CAS PubMed.
- Z. Huang, J. M. Wells, V. Fogliano and E. Capuano, Microbial tryptophan catabolism as an actionable target via diet–microbiome interactions, Crit. Rev. Food Sci. Nutr., 2024, 64, 3650–3664 Search PubMed.
- M. Zhuang, X. Zhang and J. Cai, Microbiota-gut-brain axis: interplay between microbiota, barrier function and lymphatic system, Gut Microbes, 2024, 16, 2387800 CrossRef PubMed.
- N. S. Ortiz-Samur, A. K. Vijaya, A. Burokas and V. Mela, Exploring the Role of Microglial Cells in the Gut-Brain Axis Communication: A Systematic Review, J. Neurochem., 2025, 169, e70154 CrossRef PubMed.
- L. Maier, M. Pruteanu and M. Kuhn, et al., Extensive impact of non-antibiotic drugs on human gut bacteria, Nature, 2018, 555, 623–628 CrossRef CAS.
- V. Vich Vila, V. Collij, S. Sanna, T. Sinha, F. Imhann, A. R. Bourgonje, Z. Mujagic, D. M. A. E. Jonkers, A. A. M. Masclee and J. Fu, et al., Impact of commonly used drugs on the composition and metabolic function of the gut microbiota, Nat. Commun., 2020, 11, 362 CrossRef PubMed.
- A. Cortés-Martín, C. E. Iglesias-Aguirre, A. Meoro, M. V. Selma and J. C. Espín, Pharmacological Therapy Determines the Gut Microbiota Modulation by a Pomegranate Extract Nutraceutical in Metabolic Syndrome: A randomised Clinical Trial, Mol. Nutr. Food Res., 2021, 65, e2001048 CrossRef PubMed.
- A. V. G. de Moraes, L. Costa-Paiva, J. F. Lui-Filho and A. O. Pedro, Medication use and climacteric syndrome: a cross-sectional population-based study, Menopause, 2019, 26, 1133–1140 CrossRef PubMed.
- K. Forslund, F. Hildebrand, T. Nielsen, G. Falony, E. Le Chatelier, S. Sunagawa, E. Prifti, S. Vieira-Silva, V. Gudmundsdottir and H. K. Pedersen, et al., Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota, Nature, 2015, 528, 262–266 CrossRef CAS PubMed.
- H. Wu, E. Esteve, V. Tremaroli, M. T. Khan, R. Caesar and L. Mannerås-Holm, et al., Metformin Alters the Gut Microbiome of Individuals With Treatment-Naive Type 2 Diabetes, Contributing to the Therapeutic Effects of the Drug, Nat. Med., 2017, 23, 850–858 CrossRef CAS PubMed.
- L. Zhao, Y. Chen, F. Xia, B. Abudukerimu, W. Zhang and Y. Guo, et al., A Glucagon-Like Peptide-1 Receptor Agonist Lowers Weight by Modulating the Structure of Gut Microbiota, Front. Endocrinol., 2018, 9, 233 CrossRef PubMed.
- J. Charpentier, F. Briand, B. Lelouvier, F. Servant, A. Puel and A. Puel, et al.Liraglutide Targets the Gut Microbiota and the Intestinal Immune System to Regulate Insulin Secretion, Acta Diabetol., 2021, 58, 881–897 CrossRef CAS PubMed.
- N. M. Mindrescu, C. Guja, V. Jinga, S. Ispas, A. Curici, R. E. Danciulescu Miulescu, A. Nelson Twakor and A. M. Pantea Stoian, SGLT-2 Inhibitors and Metabolic Outcomes: A Primary Data Study Exploring the Microbiota-Diabetes Connection, Metabolites, 2025, 15, 411 CrossRef CAS PubMed.
- H. H. T. Ko, R. R. Lareu, B. R. Dix and J. D. Hughes, Statins: antimicrobial resistance breakers or makers?, PeerJ, 2017, 5, e3952 CrossRef PubMed.
- L. Sun, C. Xie and G. Wang, et al., Gut microbiota and intestinal FXR mediate the clinical benefits of metformin, Nat. Med., 2018, 24, 1919–1929 CrossRef CAS PubMed.
- J. She, L. Sun, Y. Yu, H. Fan, X. Li, X. Zhang, X. Zhuo, M. Guo, J. Liu and P. Liu, et al., A gut feeling of statin, Gut Microbes, 2024, 16, 2415487 CrossRef PubMed.
- S. K. Forslund, R. Chakaroun and M. Zimmermann-Kogadeeva, et al., Combinatorial, additive and dose-dependent drug–microbiome associations, Nature, 2021, 600, 500–505 CrossRef CAS PubMed.
- M. E. Martínez-Nortes, M. Ávila-Gálvez, M. Romo-Vaquero and J. C. Espín, Impact of long-term medication on estrobolome-associated β-glucuronidase and sulfatase activities: Implications for estrogen homeostasis in postmenopausal women, Maturitas, 2026, 206, 108830 CrossRef PubMed.
- M. I. Dothard, S. M. Allard and J. A. Gilbert, The effects of hormone replacement therapy on the microbiomes of postmenopausal women, Climacteric, 2023, 26, 182–192 CrossRef CAS PubMed.
- A. Intharuksa, W. Arunotayanun, M. Na Takuathung, S. Chaichit, A. Prasansuklab, K. Chaikhong, B. Sirichanchuen, S. Chupradit and N. Koonrungsesomboon, Daidzein and Genistein: Natural Phytoestrogens with Potential Applications in Hormone Replacement Therapy, Int. J. Mol. Sci., 2025, 26, 6973 CrossRef CAS PubMed.
- G. Leite, G. M. Barlow, G. Parodi, M. L. Pimentel, C. Chang, A. Hosseini, J. Wang, M. Pimentel and R. Mathur, Duodenal microbiome changes in postmenopausal women: effects of hormone therapy and implications for cardiovascular risk, Menopause, 2022, 29, 264–275 CrossRef PubMed.
- M. P. Jarrín-Orozco, M. Romo-Vaquero, C. Carrascosa, M. Pertegal, J. Berná, J. Puigcerver, A. Saura-Sanmartín, I. Espinosa-Salinas, M. García-Nicolás, M. Á. Ávila-Gálvez and J. C. Espín, Polyphenol-Related Gut Metabotype Signatures Linked to Quality of Life in Postmenopausal Women: A Randomised, Placebo-Controlled Crossover Trial, Nutrients, 2025, 17, 3572 CrossRef PubMed.
- U. J. Lee, J. K. Sohng, B. G. Kim and K. Y. Choi, Recent trends in the modification of polyphenolic compounds using hydroxylation and glycosylation, Curr. Opin. Biotechnol., 2023, 80, 102914 CrossRef CAS PubMed.
- D. Li, O. Pain, C. Fabbri, W. L. E. Wong, C. W. H. Lo, S. Ripke, A. Cattaneo, D. Souery, M. Z. Dernovsek, N. Henigsberg, J. Hauser, G. Lewis, O. Mors, N. Perroud, M. Rietschel, R. Uher, W. Maier, B. T. Baune, J. M. Biernacka, G. Bondolfi, K. Domschke, M. Kato, Y. L. Liu, A. Serretti, S. J. Tsai, R. Weinshilboum, GSRD Consortium, the Major Depressive Disorder Working Group of the Psychiatric Genomics Consortium, A. M. McIntosh and C. M. Lewis, Metabolic activity of CYP2C19 and CYP2D6 on antidepressant response from 13 clinical studies using genotype imputation: a meta-analysis, Transl. Psychiatry, 2024, 14, 296 CrossRef CAS PubMed.
- E. Fliszár-Nyúl, V. Mohos, R. Csepregi, P. Mladěnka and P. Poór, Inhibitory effects of polyphenols and their colonic metabolites on CYP2D6 enzyme using two different substrates, Biomed. Pharmacother., 2020, 131, 110732 CrossRef PubMed.
- K. An, R. Huang, S. Tian, D. Guo, J. Wang, H. Lin and S. Wang, Statins significantly reduce mortality in patients receiving clopidogrel without affecting platelet activation and aggregation: a systematic review and meta-analysis, Lipids Health Dis., 2019, 18, 121 CrossRef PubMed.
- K. Mansoor, R. Bardees, B. Alkhawaja, E. Mallah, L. AbuQatouseh, M. Schmidt and K. Matalka, Impact of Pomegranate Juice on the Pharmacokinetics of CYP3A4- and CYP2C9-Mediated Drugs Metabolism: A Preclinical and Clinical Review, Molecules, 2023, 28, 2117 CrossRef CAS PubMed.
- M. Kondža, I. Brizić and S. Jokić, Flavonoids as CYP3A4 Inhibitors In Vitro, Biomedicines, 2024, 12, 644 CrossRef PubMed.
- M. J. Rodríguez Arcas, E. García-Jiménez, F. Martínez-Martínez and P. Conesa-Zamora, Role of CYP450 in pharmacokinetics and pharmacogenetics of antihypertensive drugs, Farm. Hosp., 2011, 35, 84–92 CrossRef PubMed.
- H. H. Chow, L. L. Garland, C. H. Hsu, D. R. Vining, W. M. Chew, J. A. Miller, M. Perloff, J. A. Crowell and D. S. Alberts, Resveratrol modulates drug- and carcinogen-metabolising enzymes in a healthy volunteer study, Cancer Prev. Res., 2010, 3, 1168–1175 CrossRef CAS PubMed.
- M. J. Hanley, P. Cancalon, W. W. Widmer and D. J. Greenblatt, The effect of grapefruit juice on drug disposition, Expert Opin. Drug Metab. Toxicol., 2011, 7, 267–286 CrossRef CAS PubMed.
- S. Misaka, O. Abe, T. Ono, Y. Ono, H. Ogata, I. Miura, Y. Shikama, M. F. Fromm, H. Yabe and K. Shimomura, Effects of single green tea ingestion on pharmacokinetics of nadolol in healthy volunteers, Br. J. Clin. Pharmacol., 2020, 86, 2314–2318 CrossRef CAS PubMed.
- N. M. Kyriacou, A. S. Gross and A. J. McLachlan, Green tea catechins as perpetrators of drug pharmacokinetic interactions, Clin. Pharmacol. Ther., 2025, 118, 45–61 CrossRef CAS PubMed.
- N. Yang, R. Sun, X. Liao, J. Aa and G. Wang, UDP-glucuronosyltransferases (UGTs) and their related metabolic cross-talk with internal homeostasis: A systematic review of UGT isoforms for precision medicine, Pharmacol. Res., 2017, 121, 169–183 CrossRef CAS PubMed.
- K. Kurogi, M. I. Rasool, F. A. Alherz, A. A. El Daibani, A. F. Bairam, M. S. Abunnaja, S. Yasuda, L. J. Wilson, Y. Hui and M. C. Liu, SULT genetic polymorphisms: physiological, pharmacological and clinical implications, Expert Opin. Drug Metab. Toxicol., 2021, 17, 767–784 CrossRef CAS PubMed.
- B. Wu, K. Kulkarni, S. Basu, S. Zhang and M. Hu, First-pass metabolism via UDP-glucuronosyltransferase: a barrier to oral bioavailability of phenolics, J. Pharm. Sci., 2011, 100, 3655–3681 CrossRef CAS PubMed.
- K. W. Bock, Selective contribution of human UDP-glucuronosyltransferases UGT1A1, 1A8, 1A9, and 2B7 to the hepatic and intestinal glucuronidation of the flavonoids, Biochem. Pharmacol., 2012, 84, 1205–1212 CrossRef PubMed.
- J. Chen, H. Zhang, X. Hu, M. Xu, Y. Su, C. Zhang, Y. Yue, X. Zhang, X. Wang, W. Cui, Z. Zhao and X. Li, Phloretin exhibits potential food-drug interactions by inhibiting human UDP-glucuronosyltransferases in vitro, Toxicol. In Vitro, 2022, 84, 105447 CrossRef CAS PubMed.
- G. Mainini, S. Ercolano, R. De Simone, I. Iavarone, R. Lizza and M. Passaro, Dietary supplementation of myo-inositol, cocoa polyphenols, and soy isoflavones improves vasomotor symptoms and metabolic profile in menopausal women with metabolic syndrome: A retrospective clinical study, Medicina, 2024, 60, 598 CrossRef PubMed.
- L. Guadamuro, M. A. Azcárate-Peril, R. Tojo, B. Mayo and S. Delgado, Impact of dietary isoflavone supplementation on the fecal microbiota and its metabolites in postmenopausal women, Int. J. Environ. Res. Public Health, 2021, 18, 7939 CrossRef CAS PubMed.
- D. Serra, L. M. Almeida and T. C. Dinis, The impact of chronic intestinal inflammation on brain disorders: The microbiota-gut-brain axis, Mol. Neurobiol., 2019, 56, 6941–6951 CrossRef CAS PubMed.
- L. J. Sun, J. N. Li and Y. Z. Nie, Gut hormones in microbiota-gut-brain cross-talk, Chin. Med. J., 2020, 133, 826–833 CrossRef CAS PubMed.
- I. Parilli-Moser, I. Domínguez-López and M. Trius-Soler, et al., Consumption of peanut products improves memory and stress response in healthy adults from the ARISTOTLE study: a 6-month randomised controlled trial, Clin. Nutr., 2021, 40, 5556–5567 CrossRef CAS PubMed.
- N. A. Gillies, B. C. Wilson, J. R. Miller, N. C. Roy, A. Scholey and A. J. Braakhuis, Effects of a flavonoid-rich blackcurrant beverage on markers of the gut-brain axis in healthy females: secondary findings from a 4-week randomised crossover control trial, Curr. Dev. Nutr., 2024, 8, 102158 CrossRef CAS PubMed.
- M. E. Beckner, H. S. Fagnant, P. J. Niro, G. E. Giles, H. R. Lieberman and J. P. Karl, Effects of fermentable fiber and polyphenol supplementation on mood and cognition in adults during hypobaric hypoxia exposure, Physiol. Rep., 2025, 13, e70541 CrossRef CAS PubMed.
- A. Muduroglu-Kirmizibekmez, M. Y. Özdemir, A. Önder, C. Çatı and İ. Kara, A Pilot EEG Study on the Acute Neurophysiological Effects of Single-Dose Astragaloside IV in Healthy Young Adults, Nutrients, 2025, 17, 2425 CrossRef PubMed.
- C. H. Wilder-Smith, A. Materna and S. S. Olesen, Blueberries Improve Abdominal Symptoms, Well-Being and Functioning in Patients with Functional Gastrointestinal Disorders, Nutrients, 2023, 15, 2396 CrossRef CAS PubMed.
- S. Molino, L. F. De Lellis and M. V. Morone, et al., Improving Irritable Bowel Syndrome (IBS) Symptoms and Quality of Life with Quebracho and Chestnut Tannin-Based Supplementation: A Single-Centre, Randomised, Double-Blind, Placebo-Controlled Clinical Trial, Nutrients, 2025, 17, 552 CrossRef CAS PubMed.
- A. Wierzbicka, B. Khaidakov and O. Zakerska-Banaszak, et al., Effects of a polyphenol-rich extract blend, probiotics, and hydrolysed fiber on quality of life and gut health markers in patients with irritable bowel syndrome-A randomised, double-blind, placebo-controlled trial, Front. Nutr., 2025, 12, 1603011 CrossRef PubMed.
- C. A. Naranjo-Galvis, D. M. Trejos-Gallego and C. Correa-Salazar, et al., Anti-inflammatory diet and probiotic supplementation as strategies to modulate immune dysregulation in autism Spectrum disorder, Nutrients, 2025, 17, 2664 CrossRef CAS PubMed.
- A. Muduroglu-Kirmizibekmez, A. Onder and M. Y. Ozdemir, et al., Feeding the mind: preliminary insights into the effects of Anthocyanin-Rich Extract from black carrots on brain activity and gut microbiota in patients with cognitive impairments, Sci. Rep., 2025, 15, 45450 CrossRef CAS PubMed.
Footnote |
| † These authors have contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |