 Open Access Article
Open Access ArticleDietary β-glucan modulates sucrose preference via gut–brain neuropeptide signaling in a sex-dependent manner in Drosophila
Jiaojiao
Li†
 a,
Wenyun
Li†
a,
Wenyun
Li†
 b,
Yuanchao
Li
a,
Jiawen
Liang
a,
Yuwei
Liu
b,
Yuanchao
Li
a,
Jiawen
Liang
a,
Yuwei
Liu
 *a and
Gengsheng
He
*a
*a and
Gengsheng
He
*a
aSchool of Public Health/Key Laboratory of Public Health Safety of Ministry of Education, Fudan University, Shanghai 200032, China. E-mail: ywliu@fudan.edu.cn; gshe@shmu.edu.cn
bDepartment of Clinical Nutrition, Zhongshan Hospital, Fudan University, Shanghai 200032, China
First published on 28th April 2026
Abstract
This study investigated the role of soluble dietary fiber, specifically β-glucan, as a modulator of nutrient-specific food choice and metabolic function using Drosophila melanogaster as a model. Adult males and mated females were assigned to a blank group (B), a corn starch control group (C), or groups fed diets supplemented with β-glucan at 5% (LBG), 10% (MBG), or 20% (HBG), or arabinoxylan (AX; 10%) for 4 days. Short-term food preference was quantified using dye-based dual-choice assays, and long-term sucrose preference was assessed using the capillary feeder assay. Circulating glucose, whole-body triglycerides (TG), whole-body Drosulfakinin (DSK), and midgut Neuropeptide F (NPF) were measured by ELISA, complemented by immunofluorescence analyses in gut and brain tissues. Dietary β-glucan modulated sucrose-related feeding behavior in a sex- and dose-dependent manner. In females, neither short- nor long-term sucrose preference differed across interventions (p > 0.05). In males, β-glucan produced a non-linear response: in the sucrose-yeast assay, MBG reduced the preference index relative to C (0.50 ± 0.04 versus 0.63 ± 0.07, p = 0.014), whereas HBG increased it (0.77 ± 0.07 versus 0.63 ± 0.07, p = 0.006). In the high–low sucrose assay, HBG showed a nominal trend toward reduced short-term preference (p = 0.094). Long-term sucrose preference was significantly lower in males receiving β-glucan compared with the control (−0.09 ± 0.38 versus 0.45 ± 0.56, p = 0.022). AX did not significantly affect sucrose preference. TG levels increased in females at MBG and HBG but decreased in males at the same doses. DSK levels were higher in females (LBG and HBG, p < 0.001) and in males (LBG, MBG, HBG and AX, p < 0.001), while NPF concentrations increased in multiple intervention groups in both sexes (p < 0.001). Immunofluorescence analyses revealed broadly comparable neuropeptide distributions across groups. These findings demonstrate that dietary β-glucan modulates sucrose-related feeding behavior in a sex- and dose-dependent manner, with males exhibiting a non-linear response and reduced long-term sucrose preference. The metabolic and neuropeptide changes support a gut–brain-metabolic framework through which dietary fiber may shape sugar preference, highlighting a behavioral dimension of dietary fiber function.
Introduction
Dietary sugar preference is a fundamental feeding behavior, as sugars provide a rapid and efficient source of energy.1 However, a persistent bias toward sugar-rich foods can promote overconsumption and has been closely linked to metabolic outcomes,2 such as dysregulated glucose metabolism, abnormal lipid storage, and increased risk of obesity.3 Accumulating evidence suggests that what organisms preferentially choose to eat represents an important, and potentially modifiable, behavioral determinant of long-term metabolic health.Sugar preference is not a fixed trait; rather, it is dynamically regulated by internal nutritional state and endocrine signals. In Drosophila, gut-derived signals play a central role in communicating nutritional status to the brain and guiding food choice. Enteroendocrine cells in the adult midgut can sense specific dietary components and release neuropeptides, such as Neuropeptide F (NPF), which suppresses sugar appetite while promoting protein-rich food intake under defined physiological contexts.4 In parallel, Drosulfakinin (DSK), a functional homolog of mammalian cholecystokinin, has been implicated in satiety-related processes and feeding regulation in flies.5,6 Together, these gut–brain signaling pathways provide a flexible neuroendocrine framework through which dietary inputs can influence nutrient-directed feeding behavior.
Dietary fiber is increasingly recognized as an active modulator of metabolic and endocrine functions rather than a passive agent.7 Soluble fibers, including β-glucan as a glucose polymer abundant in cereals and fungi, have been widely studied for their roles in glycemic regulation, lipid metabolism and gut physiology.8,9 However, despite the growing interest in fiber–gut interactions, whether dietary fiber can directly influence nutrient-specific feeding behaviors, such as sucrose preference, remains poorly understood. In particular, it is unclear whether such behavioral changes are dose-dependent or sexually dimorphic or how these behavioral outcomes relate to coordinated metabolic and neuroendocrine responses.
In this study, we used Drosophila melanogaster as a model to investigate how dietary β-glucan modulates sucrose preference across various doses and both sexes. By integrating short-term and long-term behavioral assays with metabolic measurements and quantitative analyses of key neuropeptides, we aimed to characterize the relationship between β-glucan intake, endocrine responses, and feeding behavior. This integrative approach allows us to assess the behavioral dimension of dietary fiber function and to place these observed changes in sucrose preference within a broader physiological context.
Materials and methods
Drosophila stocks and dietary interventions
Wild-type w1118Drosophila melanogaster were used for the experiments. Adult male flies and mated female flies were purchased from Fungene Biology (Beijing, China) and were randomly assigned to dietary intervention groups. Female flies were obtained from stock populations in which males and females were co-housed under standard laboratory conditions, allowing natural mating prior to experimental assignment. A blank group (B) was fed a basal diet without supplementation. A corn starch control group (C) received the basal diet supplemented with 10% corn starch, serving as the primary reference for fiber-specific effects. Dietary fiber intervention groups included oat β-glucan at 5% (LBG), 10% (MBG), or 20% (HBG), as well as an arabinoxylan group (AX) supplemented at 10%, which was comparable to MBG.The basal medium consisted of 90 g L−1 sucrose, 80 g L−1 yeast, 10 g L−1 agar, 2 g L−1 potassium sorbate, and 15 mL L−1 methylparaben. Flies were maintained on their assigned diets for 4 days at 25 °C under 60–70% relative humidity with a 12 h light/dark cycle. Prior to behavioral assays, flies were starved on 1% agar-water medium for 15–24 h to standardize hunger state and promote feeding motivation.
Chemicals
β-Glucan (purity ≥80%, derived from oats) was purchased from Wuhan Huaxiang Kejie Biotechnology Co., Ltd (Wuhan, China). Wheat AX was obtained from Megazyme (P-WAXYL, Bray, Ireland). Corn starch was obtained from Solarbio (S9840, Beijing, China). Sucrose, yeast extract, agar, potassium sorbate, and methylparaben (Nipagin) were supplied by Fangjing Biotechnology (Guangzhou, China). Amaranth (A6620) and Brilliant Blue (E8500) dyes used for preference assays were obtained from Solarbio (Beijing, China).Short-term sucrose preference and yeast choice assays
Short-term feeding behavior was quantified using a spectrophotometric dye ingestion assay adapted from previously described protocols.10 After starvation, flies were transferred to dual-choice feeding plates containing alternating droplets of either high-sucrose medium (10% sucrose) versus low-sucrose medium (1% sucrose) or sucrose medium (10% sucrose) versus yeast medium (10% yeast). The two food sources were labeled with 0.5% (w/v) amaranth or 0.5% (w/v) brilliant blue, respectively.Flies were allowed to feed for 1 h in darkness to minimize potential visual bias. Individual flies were then homogenized in 200 μL phosphate-buffered saline (PBS). The homogenates were centrifuged at 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g for 5 min to remove tissue debris and lipids that could interfere with optical measurements. The absorbance of the supernatant was measured at 540 nm (amaranth) and 629 nm (brilliant blue) using a microplate reader (Bio-Rad Laboratories, California, USA). Age-matched flies fed on non-dyed medium were processed in parallel to determine baseline absorbance. Food intake was quantified using standard curves generated from serial dilutions of each dye.
000g for 5 min to remove tissue debris and lipids that could interfere with optical measurements. The absorbance of the supernatant was measured at 540 nm (amaranth) and 629 nm (brilliant blue) using a microplate reader (Bio-Rad Laboratories, California, USA). Age-matched flies fed on non-dyed medium were processed in parallel to determine baseline absorbance. Food intake was quantified using standard curves generated from serial dilutions of each dye.
Long-term sucrose preference assay
Long-term feeding intake was measured using the capillary feeder (CAFE) assay, adapted from a previously validated protocol.11 Individual flies were housed in standard Drosophila vials equipped with two 5 μL microcapillaries containing 10 g L−1 and 100 g L−1 sucrose solutions, respectively. The flies were allowed to feed continuously in a controlled incubator environment (25 °C, 60–70% relative humidity). Liquid consumption was recorded after 6 h by measuring capillary volume displacement using a high-resolution ruler. Evaporation was controlled by including parallel capillaries without flies, and corrected intake values were used for analysis.Quantification of circulating glucose and triglycerides
To assess metabolic responses, whole-fly homogenates were prepared following dietary intervention and starvation. Circulating glucose and triglyceride (TG) levels were quantified using commercial enzymatic assay kits (Stanbio Laboratory, Boerne, TX, USA) according to the manufacturer's instructions.Enzyme-linked immunosorbent assay
Systemic levels of Drosulfakinin (DSK) and midgut-derived Neuropeptide F (NPF) were quantified using enzyme-linked immunosorbent assays. For DSK measurements, whole flies were homogenized in PBS. For NPF measurements, midgut tissues were dissected prior to homogenization. Samples were centrifuged at 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g for 5 min at 4 °C, and supernatants were collected for analysis. DSK and NPF concentrations were measured using Drosophila-specific ELISA kits (Fantai Biologicals, Shanghai, China) following the manufacturer's protocols.
000g for 5 min at 4 °C, and supernatants were collected for analysis. DSK and NPF concentrations were measured using Drosophila-specific ELISA kits (Fantai Biologicals, Shanghai, China) following the manufacturer's protocols.
Immunofluorescence analysis
Immunofluorescence analyses were conducted to assess the spatial distribution of DSK and NPF in gut and brain tissues. Whole flies were fixed in 4% paraformaldehyde, paraffin-embedded, and sectioned at 4 μm thickness. After deparaffinization, heat-induced antigen retrieval was performed in citrate buffer (pH 6.0). Sections were blocked with 3% bovine serum albumin (BSA) and incubated overnight at 4 °C with rabbit anti-NPF (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500; RayBiotech, Inc., Georgia, USA) and rabbit anti-DSK (1
500; RayBiotech, Inc., Georgia, USA) and rabbit anti-DSK (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100; GenScript Biotech Corporation, Nanjing, China). Fluorescence-conjugated secondary antibodies were applied for 50 min at room temperature in the dark. Nuclei were counterstained with DAPI, and autofluorescence was minimized using a commercial quencher (Servicebio Technology Co., Hubei, China). Images were acquired using a Pannoramic MIDI digital slide scanner. Quantification of fluorescence intensity was performed within predefined regions of interest.
100; GenScript Biotech Corporation, Nanjing, China). Fluorescence-conjugated secondary antibodies were applied for 50 min at room temperature in the dark. Nuclei were counterstained with DAPI, and autofluorescence was minimized using a commercial quencher (Servicebio Technology Co., Hubei, China). Images were acquired using a Pannoramic MIDI digital slide scanner. Quantification of fluorescence intensity was performed within predefined regions of interest.
Statistical analysis
Data are presented as mean ± standard deviation unless otherwise indicated. For behavioral assays, metabolic markers, and neuropeptide measurements, pairwise comparisons were performed using Student's t-tests. C was compared with B to evaluate vehicle effects, and each fiber intervention group (LBG, MBG, HBG, and AX) was compared individually with C to assess fiber-specific effects. The preference index (PI) was calculated as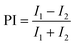 , where I1 and I2 represent the intake of the two food sources. Intake was estimated either from dye absorbance or from capillary volume displacement.
, where I1 and I2 represent the intake of the two food sources. Intake was estimated either from dye absorbance or from capillary volume displacement.
Immunofluorescence data were analyzed using linear mixed-effects models to account for variability across tissue sections and regions. The treatment group was included as a fixed effect, and region-level variation was modeled as a random effect. Given the exploratory nature of the study and predefined group comparisons, no formal correction for multiple testing was applied. All statistical analyses were conducted using R software (version 4.2.2). A two-sided p-value <0.05 was considered statistically significant.
Results
Short-term changes in sucrose preference
Short-term sucrose preference was evaluated using both a high–low sucrose choice assay and a sucrose-yeast choice assay. In the high–low sucrose assay, female flies did not show significant differences in PI among dietary intervention groups compared with C (p > 0.05; Fig. 1A and SI Fig. S1A). In males, the PI showed a progressive decrease with increasing β-glucan doses, although no significant differences were observed between LBG or MBG and the control (p > 0.05). HBG exhibited a nominal trend toward a lower PI compared with the control (0.26 ± 0.41 versus 0.64 ± 0.30, p = 0.094; Fig. 1B). Supplementation with AX did not significantly affect short-term sucrose preference in either sex (SI Fig. S1B).In the sucrose-yeast choice assay, female flies again showed no significant differences in PI across dietary groups relative to C (p > 0.05; Fig. 1C and SI Fig. S1C). In contrast, male flies exhibited distinct responses to dietary interventions (Fig. 1D). Compared with the control, LBG showed a nominal trend toward a lower PI (p = 0.053), while MBG exhibited a significantly reduced preference for sucrose relative to yeast (0.50 ± 0.04 versus 0.63 ± 0.07, p = 0.014). In contrast, HBG showed a significantly higher PI than the control (0.77 ± 0.07 versus 0.63 ± 0.07, p = 0.006). Compared with B, C showed a nominally lower PI (p = 0.050). The AX group exhibited a nominal trend toward a reduced PI relative to C (p = 0.068; SI Fig. S1D).
Long-term sucrose preference
Long-term sucrose preference was assessed using the high–low sucrose preference assay. In females, the PI did not differ significantly among dietary groups compared with C (p > 0.05, Fig. 2A and SI Fig. S2A). In contrast, male flies receiving β-glucan exhibited a significantly lower long-term sucrose PI compared with C (−0.09 ± 0.38 versus 0.45 ± 0.56, p = 0.022, Fig. 2B). No significant difference was observed between the AX and control groups in males (p > 0.05, SI Fig. S2B).Sex-specific metabolic responses to β-glucan
Circulating metabolic markers were measured to assess physiological responses associated with dietary β-glucan intakes (Fig. 3). In female flies, circulating glucose levels were higher at increased β-glucan doses relative to C (Fig. 3A). Specifically, glucose levels were elevated in the MBG (17.38 ± 5.27 versus 4.27 ± 1.78 μg per fly, p < 0.001) and HBG groups (15.03 ± 2.31 versus 4.27 ± 1.78 μg per fly, p < 0.001), whereas no significant differences were observed in the LBG or AX groups (p = 0.210 and 0.934, respectively). In males, circulating glucose levels were significantly higher in LBG (10.95 ± 2.89 versus 3.99 ± 2.38 μg per fly, p < 0.001, Fig. 3B), HBG (10.05 ± 2.88 versus 3.99 ± 2.38 μg per fly, p < 0.001), and AX (p = 0.002, SI Fig. S3B) compared with C, whereas no significant difference was observed in the MBG group (p = 0.601).Whole-body triglyceride (TG) levels exhibited distinct sex- and dose-dependent patterns (Fig. 3C and D). In females, TG levels were significantly higher in MBG (10.18 ± 4.51 versus 4.89 ± 2.02 μg per fly, p = 0.002) and HBG (8.32 ± 1.52 versus 4.89 ± 2.02 μg per fly, p < 0.001), whereas no significant differences were observed in the LBG or AX groups (p = 0.403 and 0.361, respectively). In males, TG levels were lower in the MBG (5.10 ± 4.06 versus 8.36 ± 2.57 μg per fly, p = 0.038, Fig. 3D) and HBG groups (5.65 ± 1.36 versus 8.36 ± 2.57 μg per fly, p = 0.003) relative to C, while no significant differences were observed in the LBG or AX groups (p = 0.466 and 0.875, respectively, SI Fig. S3B).
Hormonal responses to β-glucan
Systemic and gut-derived neuropeptide levels were quantified to characterize endocrine responses associated with dietary intervention (Fig. 4 and SI Fig. S4). In female flies, whole-body DSK levels were significantly higher in LBG (18.16 ± 1.49 versus 6.41 ± 0.40 pg per fly, p < 0.001) and HBG (16.72 ± 0.74 versus 6.41 ± 0.40 pg per fly, p < 0.001) compared with C, whereas no significant differences were observed in MBG or AX (p = 0.273 and 0.967, respectively; Fig. 4A and SI Fig. S4A). In male flies, DSK levels were elevated across all dietary fiber interventions, including LBG (17.34 ± 1.82 versus 6.02 ± 0.48 pg per fly, p < 0.001), MBG (6.93 ± 0.34 versus 6.02 ± 0.48 pg per fly, p < 0.001), HBG (17.87 ± 1.21 versus 6.02 ± 0.48 pg per fly, p < 0.001, Fig. 4B), and AX (19.14 ± 0.81 versus 6.02 ± 0.48 pg per fly, p < 0.001) relative to C (Fig. 4B and SI Fig. S4B).Midgut-derived NPF levels were quantified to further characterize endocrine responses associated with dietary intervention. In females, NPF levels were significantly lower in C than in B (9.48 ± 0.68 versus 10.70 ± 0.75 pg per fly, p < 0.001). Relative to C, NPF levels were significantly higher in LBG (18.36 ± 0.93 versus 9.48 ± 0.68 pg per fly, p < 0.001), HBG (18.69 ± 1.70 versus 9.48 ± 0.68 pg per fly, p < 0.001), and AX (10.87 ± 0.42 versus 9.48 ± 0.68 pg per fly, p < 0.001), whereas no significant difference was observed in MBG (p = 0.140, Fig. 4C and SI Fig. S4C). In males, NPF levels did not differ between C and B (p = 0.271), but were significantly higher in LBG (18.44 ± 1.11 versus 9.64 ± 0.83 pg per fly, p < 0.001), MBG (11.82 ± 1.13 versus 9.64 ± 0.83 pg per fly, p < 0.001), HBG (17.32 ± 0.80 versus 9.64 ± 0.83 pg per fly, p < 0.001), and AX (18.85 ± 0.85 versus 9.64 ± 0.83 pg per fly, p < 0.001) compared with C.
Immunofluorescence analysis of DSK and NPF
Immunofluorescence analyses were performed to evaluate the tissue distribution and staining intensity of DSK and NPF in the gut and brain. Representative staining images are shown in SI Fig. S5.In the midgut, DSK immunofluorescence intensity exhibited subtle, sex-dependent differences (SI Fig. S6A and B). In females, the gut DSK signal tended to be lower in LBG and MBG relative to C (p < 0.1), whereas HBG did not differ significantly from the control group (p > 0.05, SI Fig. S6A). In males, no significant differences in midgut DSK immunofluorescence intensity were observed among the control and β-glucan intervention groups (p > 0.05, SI Fig. S6B). In brain tissue, DSK immunofluorescence did not differ significantly among dietary groups in either sex (p > 0.05, SI Fig. S7A and B).
For NPF, immunofluorescence signals in both gut and brain tissues were comparable across the control, LBG, MBG, and HBG in females and males (p > 0.05; SI Fig. S6C, D and S7C, D).
Discussion
This study demonstrates that dietary β-glucan can modulate sucrose preference in Drosophila in a sex-dependent and dose-dependent manner. By combining short-term and long-term behavioral paradigms, we identify a distinct behavioral sensitivity to β-glucan in males, characterized by a non-linear response across doses, whereas mated females remain behaviorally stable despite pronounced physiological alterations. These findings extend the functional scope of dietary fiber beyond metabolic regulation and highlight its potential role in influencing sucrose preference.A key observation of this study is the non-linear modulation of sucrose preference observed exclusively in males. In the short-term sucrose-yeast choice assay, an intermediate dose of β-glucan reduced sucrose preference, whereas the high dose elicited an opposite shift in food choices. This non-monotonic pattern is consistent with previous reports showing that feeding behavior is regulated by integrated nutritional and endocrine signals that often exhibit dose–response relationships and act through gut–brain communication pathways.12,13 Previous studies have also shown that intermediate levels of nutrient-derived signaling often optimally engage feeding circuits, while excessive stimulation can recruit compensatory or counter-regulatory mechanisms that alter behavioral outcomes.14,15 Importantly, the suppression of sucrose preference in males persisted in the long-term feeding assay, suggesting that this effect reflects not only acute sensory-driven decisions but also sustained feeding patterns. Such behavioral plasticity may reflect adaptive adjustments to dietary composition rather than simple changes in caloric intake.
In contrast to males, female flies displayed largely stable sucrose preference across all dietary interventions. This behavioral stability occurred despite marked alterations in circulating glucose, triglyceride levels, and neuropeptide concentrations, indicating a partial uncoupling between physiological responses and overt feeding behavior in females. This observation aligns with previous studies demonstrating that the feeding decisions in females are highly dependent on the physiological state, reproductive status, and internal nutrient demands,16,17 which may buffer behavioral outputs against dietary perturbations. Our findings are consistent with this notion and suggest a sex-specific regulatory framework that shapes how dietary fiber intake is translated into feeding behavior.
To be specific, dietary β-glucan induced pronounced, yet sex- and dose-dependent metabolic responses. In males, β-glucan exposure was associated with reduced TG levels at medium and high doses, whereas females displayed a non-monotonic TG response. A high β-glucan exposure in males was associated with both elevated glucose levels and increased sucrose preference, but not with intermediate β-glucan doses. Meanwhile, females displayed elevated glucose at higher β-glucan doses, yet without accompanying changes in sucrose preference. These divergent metabolic outcomes suggest that circulating TG and glucose levels are unlikely to be solely driven by changes in sucrose preference and instead point to sex-specific downstream handling of dietary components and energy storage, as well as integrated physiological states,18–20 which is consistent with previous studies21 and highlights the multi-layered regulation of feeding and metabolism by dietary fiber.
Neuroendocrine analyses revealed substantial but sex-dependent alterations in DSK and NPF signaling following β-glucan intervention, providing a gut–brain signaling framework. Notably, in females, changes in whole-body DSK and midgut-derived NPF levels did not uniformly predict behavioral outcomes across doses, although previous reports have suggested that gut-derived NPF suppresses sugar appetite in adult female flies, particularly in mated females.4 In contrast, males displayed a coordinated, non-linear neuroendocrine response in which DSK and NPF levels followed a U-shaped pattern that paralleled the behavioral phenotype. These findings suggest that, in males, DSK and NPF signaling exert bidirectional influences on feeding behavior, with moderate activation favoring sugar. Such non-monotonic gut–brain hormonal regulation has been proposed as a general feature of nutrient-responsive systems governing feeding and reward.22,23 Immunofluorescence analyses further indicated that the spatial distribution of these neuropeptides in the gut and brain remained largely comparable across dietary groups. Together, these observations suggest that dietary β-glucan primarily modulates neuroendocrine signaling at a systemic or functional level rather than inducing major anatomical changes and that behavioral outcomes likely emerge from the integrated action of multiple physiological signals.
To examine dose-dependent and non-linear effects, dietary β-glucan was provided across a range of exposure levels in Drosophila. In humans, dietary fiber intake is commonly discussed as absolute daily intake, with approximately 25–29 g day−1 considered nutritionally relevant.24 The dose range used here was therefore intended for mechanistic interpretation rather than direct translation to habitual human diets.
From a functional nutrition perspective, our findings highlight an underexplored behavioral dimension of dietary fiber action. While β-glucan has been extensively studied for its effects on glycemic control and lipid metabolism, its potential to shape food choice has received comparatively little attention. The sex- and dose-dependent behavioral effects observed here underscore the importance of considering behavioral endpoints when evaluating the functional impact of dietary components. Several limitations should also be acknowledged. Firstly, although we identified coordinated changes in feeding behavior, metabolic markers, and neuropeptide levels, the present analyses do not establish direct causal relationships between specific neuroendocrine signals and behavioral outcomes. Secondly, measurements of circulating metabolites represent integrated physiological states and cannot distinguish primary drivers from downstream consequences. Thirdly, although both males and mated females were included to examine sex-related differences, unmated females were not evaluated. Because mating status can influence feeding behavior and metabolic physiology in Drosophila, future studies may further distinguish the relative contributions of sex and reproductive state to dietary fiber-induced behavioral responses. Finally, while Drosophila melanogaster provides a powerful model for investigating diet-behavior interactions and nutrient-sensing mechanisms, with substantial conservation of gut–brain signaling pathways across species,25,26 validation in mammalian systems will be important to determine whether similar regulatory mechanisms operate in higher organisms. In particular, the neuropeptides examined in this study, DSK and NPF, are functionally related to mammalian CCK and NPY, respectively,27 supporting the relevance of this model while also highlighting the need for cross-species validation.
Conclusion
In conclusion, this study provides functional evidence that dietary β-glucan can influence sucrose preference in Drosophila in a sex- and dose-dependent manner. By integrating behavioral, metabolic and neuroendocrine measurements, our findings support a gut–brain–metabolic framework through which dietary fiber may reduce sucrose preference. These results broaden the functional understanding of dietary fiber and highlight the value of incorporating behavioral outcomes into nutritional research.Author contributions
Conceptualization: Jiaojiao Li, Wenyun Li, Yuwei Liu, and Gengsheng He; investigation: Jiaojiao Li, Wenyun Li, Yuanchao Li, and Jiawen Liang; formal analysis: Jiaojiao Li and Wenyun Li; visualization: Jiaojiao Li; writing – original draft: Jiaojiao Li and Wenyun Li; writing – review & editing: Jiaojiao Li, Wenyun Li, Yuwei Liu, and Gengsheng He.Conflicts of interest
There are no conflicts to declare.Data availability
The data are available from the corresponding author upon reasonable request.Supplementary information (SI) is available and includes additional figures. See DOI: https://doi.org/10.1039/d6fo00187d.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82373555).References
- H. E. Tan, A. C. Sisti, H. Jin, M. Vignovich, M. Villavicencio, K. S. Tsang, Y. Goffer and C. S. Zuker, The gut-brain axis mediates sugar preference, Nature, 2020, 580, 511–516 CrossRef CAS PubMed.
- P. K. Olszewski, E. L. Wood, A. Klockars and A. S. Levine, Excessive Consumption of Sugar: an Insatiable Drive for Reward, Curr. Nutr. Rep., 2019, 8, 120–128 CrossRef CAS PubMed.
- B. Merino, C. M. Fernandez-Diaz, I. Cozar-Castellano and G. Perdomo, Intestinal Fructose and Glucose Metabolism in Health and Disease, Nutrients, 2019, 12, 94 CrossRef PubMed.
- A. Malita, O. Kubrak, T. Koyama, N. Ahrentlov, M. J. Texada, S. Nagy, K. V. Halberg and K. Rewitz, A gut-derived hormone suppresses sugar appetite and regulates food choice in Drosophila, Nat. Metab., 2022, 4, 1532–1550 CrossRef CAS PubMed.
- D. R. Nassel and M. J. Williams, Cholecystokinin-Like Peptide (DSK) in Drosophila, Not Only for Satiety Signaling, Front. Endocrinol., 2014, 5, 219 Search PubMed.
- T. Y. Fedina, E. T. Cummins, D. E. L. Promislow and S. D. Pletcher, The neuropeptide drosulfakinin enhances choosiness and protects males from the aging effects of social perception, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2308305120 CrossRef CAS PubMed.
- X. Gao, S. Hu, Y. Liu, S. De Alwis, Y. Yu, Z. Li, Z. Wang and J. Liu, Dietary Fiber as Prebiotics: A Mitigation Strategy for Metabolic Diseases, Foods, 2025, 14, 2670 CrossRef CAS PubMed.
- M. Schmidt, Cereal beta-glucans: an underutilized health endorsing food ingredient, Crit. Rev. Food Sci. Nutr., 2022, 62, 3281–3300 CrossRef CAS PubMed.
- E. J. Murphy, E. Rezoagli, I. Major, N. J. Rowan and J. G. Laffey, beta-Glucan Metabolic and Immunomodulatory Properties and Potential for Clinical Application, J. Fungi, 2020, 6, 356 CrossRef CAS PubMed.
- G. B. Carvalho, P. Kapahi, D. J. Anderson and S. Benzer, Allocrine modulation of feeding behavior by the Sex Peptide of Drosophila, Curr. Biol., 2006, 16, 692–696 CrossRef CAS PubMed.
- W. W. Ja, G. B. Carvalho, E. M. Mak, N. N. de la Rosa, A. Y. Fang, J. C. Liong, T. Brummel and S. Benzer, Prandiology of Drosophila and the CAFE assay, Proc. Natl. Acad. Sci. U. S. A., 2007, 104, 8253–8256 CrossRef CAS PubMed.
- R. Gutierrez, E. Fonseca and S. A. Simon, The neuroscience of sugars in taste, gut-reward, feeding circuits, and obesity, Cell. Mol. Life Sci., 2020, 77, 3469–3502 CrossRef CAS PubMed.
- A. Sclafani, Gut-brain nutrient signaling. Appetition vs. satiation, Appetite, 2013, 71, 454–458 CrossRef PubMed.
- A. H. Pool, P. Kvello, K. Mann, S. K. Cheung, M. D. Gordon, L. Wang and K. Scott, Four GABAergic interneurons impose feeding restraint in Drosophila, Neuron, 2014, 83, 164–177 CrossRef CAS PubMed.
- C. Y. Su and J. W. Wang, Modulation of neural circuits: how stimulus context shapes innate behavior in Drosophila, Curr. Opin. Neurobiol., 2014, 29, 9–16 CrossRef CAS PubMed.
- S. J. Cheriyamkunnel, S. Rose, P. F. Jacob, L. A. Blackburn, S. Glasgow, J. Moorse, M. Winstanley, P. J. Moynihan, S. Waddell and C. Rezaval, A neuronal mechanism controlling the choice between feeding and sexual behaviors in Drosophila, Curr. Biol., 2021, 31, 4231–4245 CrossRef CAS PubMed.
- D. R. Nassel and A. M. Winther, Drosophila neuropeptides in regulation of physiology and behavior, Prog. Neurobiol., 2010, 92, 42–104 CrossRef PubMed.
- S. E. la Fleur and M. J. Serlie, The interaction between nutrition and the brain and its consequences for body weight gain and metabolism; studies in rodents and men, Best Pract. Res. Clin. Endocrinol. Metab., 2014, 28, 649–659 CrossRef CAS PubMed.
- S. Choi, D. S. Lim and J. Chung, Feeding and Fasting Signals Converge on the LKB1-SIK3 Pathway to Regulate Lipid Metabolism in Drosophila, PLoS Genet., 2015, 11, e1005263 CrossRef PubMed.
- O. Strilbytska, U. Semaniuk, V. Bubalo, K. B. Storey and O. Lushchak, Dietary Choice Reshapes Metabolism in Drosophila by Affecting Consumption of Macronutrients, Biomolecules, 2022, 12, 1201 CrossRef CAS PubMed.
- L. Zhang, W. Han, C. Lin, F. Li and I. E. de Araujo, Sugar Metabolism Regulates Flavor Preferences and Portal Glucose Sensing, Front. Integr. Neurosci., 2018, 12, 57 CrossRef CAS PubMed.
- X. Li, M. J. Qu, Y. Zhang, J. W. Li and T. X. Liu, Expression of Neuropeptide F Gene and Its Regulation of Feeding Behavior in the Pea Aphid, Acyrthosiphon pisum, Front. Physiol., 2018, 9, 87 CrossRef CAS PubMed.
- A. Klockars, A. S. Levine, M. A. Head, C. E. Perez-Leighton, C. M. Kotz and P. K. Olszewski, Impact of Gut and Metabolic Hormones on Feeding Reward, Compr. Physiol., 2021, 11, 1425–1447 CrossRef PubMed.
- A. Reynolds, J. Mann, J. Cummings, N. Winter, E. Mete and L. T. Morenga, Carbohydrate quality and human health: a series of systematic reviews and meta-analyses, Lancet, 2019, 393, 434–445 CrossRef CAS PubMed.
- A. H. Pool and K. Scott, Feeding regulation in Drosophila, Curr. Opin. Neurobiol., 2014, 29, 57–63 CrossRef CAS PubMed.
- I. Miguel-Aliaga, H. Jasper and B. Lemaitre, Anatomy and Physiology of the Digestive Tract of Drosophila melanogaster, Genetics, 2018, 210, 357–396 CrossRef CAS PubMed.
- P. Agrawal, D. Kao, P. Chung and L. L. Looger, The neuropeptide Drosulfakinin regulates social isolation-induced aggression in Drosophila, J. Exp. Biol., 2020, 223, jeb.207407 CrossRef PubMed.
Footnote |
| † Jiaojiao Li and Wenyun Li contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |




