Open Access Article
Open Access ArticlePrecision health targeting TMAO in postmenopausal women: polyphenol effects modulated by urolithin A and equol metabotypes in a randomised, placebo-controlled crossover trial
María Paula
Jarrín-Orozco†
a,
María
García-Nicolás†
a,
María
Romo-Vaquero
 a,
Concepción
Carrascosa
b,
José
Berná
a,
Concepción
Carrascosa
b,
José
Berná
 c,
Julio
Puigcerver
c,
Adrián
Saura-Sanmartín
c,
Julio
Puigcerver
c,
Adrián
Saura-Sanmartín
 c,
María Ángeles
Ávila-Gálvez
*a and
Juan Carlos
Espín
c,
María Ángeles
Ávila-Gálvez
*a and
Juan Carlos
Espín
 *a
*a
aLaboratory of Food & Health, Research Group on Quality, Safety, and Bioactivity of Plant Foods, CEBAS-CSIC, 30100 Campus de Espinardo, Murcia, Spain. E-mail: mavila@cebas.csic.es; jcespin@cebas.csic.es
bDepartment of Obstetrics and Gynecology, Virgen de La Arrixaca University Hospital, El Palmar, 30120, Murcia, Spain
cDepartment of Organic Chemistry, Faculty of Chemistry, University of Murcia, 30100 Murcia, Spain
First published on 2nd February 2026
Abstract
Menopause increases cardiometabolic risk, partly by reducing the protective effects of estrogens and inducing gut microbiota dysbiosis, which can promote the production of atherogenic metabolites such as trimethylamine N-oxide (TMAO). Polyphenols may reduce TMAO levels, though interindividual variability limits reproducibility. We compared urinary and serum TMAO levels, and urinary trimethylamine (TMA) and dimethylamine (DMA) levels between healthy women of reproductive age (Pre-M, n = 120) and non-medicated postmenopausal women (Post-M, n = 90) using UPLC-QqQ-MS/MS. In Post-M women, we conducted a randomised, placebo-controlled crossover study to evaluate the effects of a polyphenol-rich extract mixture containing pomegranate, Polygonum cuspidatum, and red clover (sources of ellagitannins, resveratrol, and isoflavones) on TMAO, TMA, and DMA in the whole group and after metabotyping. Because medication is common in Post-M women due to age and cardiometabolic risk, trials in non-medicated participants are challenging, yet avoiding drug-diet interactions allows clearer attribution of dietary effects. Urinary TMAO and DMA levels were higher in Post-M than in Pre-M. No changes were observed in serum TMAO. However, the intervention reduced urinary TMAO and DMA versus baseline and placebo. The effects varied by metabotype. TMAO reduction was significant in urolithin A metabotype (UMA), equol producers (EP), and lunularin non-producers (LNP). Reductions and effect sizes were most pronounced in the metabotype clusters MC3 (UMA + EP + LP) and MC7 (UMA + EP + LNP), which represented 39% of participants. DMA decreased selectively in UMA. No correlations were found between TMAO or DMA changes and BMI, age at menopause onset, or years since menopause. These findings show that polyphenol supplementation reduces urinary TMAO in a metabotype-dependent manner and support metabotyping as a precision-health strategy to mitigate cardiometabolic risk after menopause.
Introduction
Menopause is defined as the permanent cessation of menstruation resulting from declining ovarian function and reduced production of estrogen and progesterone. It typically occurs between the ages of 50 and 52 and represents a complex biological transition that extends beyond reproductive ageing. The decline in estrogen levels triggers a cascade of physiological, psychological, and metabolic changes that affect women's health and well-being.1 This hormonal shift contributes to vasomotor symptoms, disturbances in lipid and glucose metabolism, and increased vulnerability to mood disorders. As a result, postmenopausal women face a higher risk of cardiovascular disease (CVD), osteoporosis, type 2 diabetes, cognitive decline, and affective disorders, all of which can substantially impair health-related quality of life.2,3Polyphenols are recognised as modulators of human physiology and potential agents for the prevention of chronic diseases. Experimental and clinical evidence links polyphenol consumption with improvements in oxidative stress, inflammation, lipid metabolism, and mitochondrial function.4–6 However, despite a large body of literature suggesting beneficial effects, most human trials have yielded inconsistent or modest outcomes. This lack of reproducibility has led regulatory authorities, including the European Food Safety Authority (EFSA), to reject the majority of health claims related to polyphenols, mainly due to pronounced interindividual variability in their metabolism and biological responses.7
This variability is especially evident in studies involving postmenopausal women. Several meta-analyses support the benefits of isoflavones in alleviating menopausal symptoms and improving cardiometabolic health,8 but other analyses report inconsistent findings or significant heterogeneity among participants.9 The health effects of other polyphenols, such as resveratrol, have been less frequently studied in postmenopausal women, and the available evidence remains inconclusive.10 These discrepancies likely stem from differences in gut microbial metabolism, since polyphenols are extensively transformed in the colon before absorption.11,12 Thus, these microbial transformations critically shape the bioactivity and systemic actions of polyphenols.
Gut microbial metabolites, such as urolithins, equol, lunularin, and various phenolic acids, exhibit higher bioavailability and distinct biological activities than their precursors. Subsequent phase II metabolism regulates their circulation and excretion, while enterohepatic recycling prolongs systemic exposure. Many of the physiological effects attributed to polyphenols are now believed to arise primarily from these gut microbiota-derived metabolites.7 Equol and urolithin A are among the best characterised examples, both exerting antioxidant, anti-inflammatory, mitophagic, and cardioprotective actions.5
Variation in the capacity to produce these metabolites among individuals has led to the identification of specific gut microbiota metabotypes associated with polyphenol metabolism. Individuals can be classified according to their ability to generate equol from daidzein, urolithins from ellagic acid, or lunularin from resveratrol.5 Combinations of these metabotypes, known as metabotype clusters (MCs), may coexist in the same individual, generating up to 12 possible profiles with distinct biological potentials.13 Recent findings in postmenopausal women showed that clinically meaningful improvements in quality of life after supplementation with a mixture of polyphenol-rich plant extracts (pomegranate, resveratrol, and red clover) were mainly observed in equol producers (EP) and in certain MCs.14 These results indicate that the efficacy of polyphenol interventions can depend on the individual's capacity to generate specific bioactive metabolites, providing a mechanistic basis for the inconsistent outcomes often observed in intervention trials and supporting the use of metabotyping as a tool in precision health approaches.
Among the many metabolites produced by the gut microbiota, trimethylamine N-oxide (TMAO) has received particular attention because of its role in CVD.15,16 TMAO is generated in the liver from trimethylamine (TMA), a compound produced by gut bacteria from dietary precursors such as choline, carnitine, and betaine, which are abundant in red meat, fish, eggs, and some vegetables. Despite some discrepancies between observational and intervention studies, elevated TMAO levels have been associated with vascular inflammation, endothelial dysfunction, foam cell formation, and platelet hyperreactivity, processes that contribute to the development of atherosclerosis and adverse cardiovascular events.17 Lifestyle and dietary factors that modulate the gut microbiota, such as increased intake of fibre-rich or plant-based foods, can lower circulating TMAO levels and improve cardiovascular outcomes.18,19
Accumulating evidence suggests that polyphenols may attenuate TMAO-related CVD risk by reshaping gut microbial composition and reducing the abundance of taxa involved in TMA production and/or modulating hepatic flavin monooxygenase 3 (FMO3) activity.15 This interaction provides an additional explanation for the cardiovascular benefits of polyphenol-rich diets and highlights the potential of targeting host-microbiota co-metabolism as a strategy to improve health in postmenopausal women.
Based on the above, we hypothesise that the effects of certain polyphenols on TMAO levels may be modulated by the gut microbiota's metabolism of these compounds, leading to distinct metabolites with varying impacts on TMAO levels. Therefore, this study aims to determine whether a mixture of plant extracts rich in ellagitannins, resveratrol, and isoflavones can influence serum and/or urinary TMAO levels compared with a placebo in non-medicated postmenopausal women, depending on their gut microbiota metabotype.
Materials and methods
Chemicals and reagents
Trimethylamine N-oxide (TMAO; Ref. 317594), trimethylamine (TMA; Ref. 021758-01), dimethylamine (DMA; Ref. 011755-04), and creatinine anhydrous (≥98%, Ref. C4255) were obtained from Sigma-Aldrich (St Louis, MO, USA). trans-Resveratrol (resveratrol), ellagic acid, daidzein, genistein, formononetin, biochanin A, equol, and hesperetin were purchased from Sigma-Aldrich. O-Demethylangolensin (ODMA) and equol 7-O-glucuronide were purchased from LGC Standards (Barcelona, Spain). Resveratrol 3-O-glucuronide, dihydroresveratrol 3-O-glucuronide, resveratrol 3-O-sulfate, resveratrol 4′-O-sulfate, Urolithin A (Uro-A), B (Uro-B), Isourolithin A (IsoUro-A), Uro-A 3-O-glucuronide, Uro-A 8-O-glucuronide, Uro-B glucuronide, Uro-B sulfate, IsoUro-A 3-O-glucuronide, IsoUro-A 9-O-glucuronide, lunularin, 4-hydroxydibenzyl (4HDB) were obtained as reported elsewhere.20 Uro-A sulfate, lunularin-O-glucuronide regioisomers, lunularin-O-sulfate regioisomers, 4HDB glucuronide and 4HDB sulfate were obtained as described in the SI (Fig. S1–S14). All the reagents and metabolites were 97% pure or higher. Ammonium acetate was obtained from PanReac AppliChem (Barcelona, Spain). Formic acid was purchased from Sigma-Aldrich. LC-MS grade acetonitrile, dimethyl sulfoxide, and methanol were supplied by Scharlab (Barcelona, Spain). Ultrapure water was produced using a Milli-Q purification system (Millipore, Bedford, MA, USA) and was used for all experiments and mobile phases.Polyphenol-rich mixture (PPs)
The polyphenol-rich mixture (PPs), comprising resveratrol, pomegranate, and red clover extracts, was prepared as previously described.21 Capsules (700 mg) contained Polygonum cuspidatun extract (50 mg, containing 98% purity resveratrol), punicalagin-rich pomegranate extract (320 mg, 20% punicalagin), EA-rich pomegranate extract (80 mg, 40% EA), and red clover extract (250 mg, 20% isoflavones) (Laboratorios Admira S.L., Alcantarilla, Spain). The extracts were further analysed by HPLC-ESI-IT-MS/MS.21 Each PPs capsule contained 103.7 ± 10.3 mg of ellagitannins and ellagic acid, 44.4 ± 3.4 mg of resveratrol, and 55.5 ± 9.1 mg of isoflavones (Table S1). Placebo capsules contained microcrystalline cellulose (700 mg) and were identical in appearance to the PPs capsules.Subjects and study design
The study protocol was approved by the Bioethics Committees of the Spanish National Research Council (CSIC, Madrid; ref. 249/2023), IMDEA-Food (Madrid; ref. PI-065), and Virgen de La Arrixaca University Hospital (Murcia; ref. 2023-4-9-HCUVA), within the framework of the PolyPAUSE Project (PID2022-136419OB-I00; MICIU, Spain). The trial was conducted in accordance with the Declaration of Helsinki (1975, as revised) and registered at clinicaltrials.gov (NCT07182370). Participant recruitment took place between September 2023 and January 2025. All participants provided written informed consent prior to inclusion in the study.The experimental design and general procedures have been described in detail elsewhere.14 The trial comprised two studies: Study 1 included healthy premenopausal women (Pre-M), defined as women in their reproductive years (aged 30–48 years), who were recruited for comparative purposes; and Study 2 involved postmenopausal women (Post-M, aged 45–59 years), enrolled in an 8-week intervention to assess the effects of PPs on urinary and serum levels of TMAO, TMA, and DMA.
In Study 1, Pre-M participants consumed 3 capsules of PPs daily for 3 days to standardise metabotype determination using first-morning urine samples. Urine samples were collected before and after intake.
In Study 2, healthy, non-medicated Post-M women were randomised to receive either PPs or the placebo for 8 weeks, followed by a 1-month washout and crossover to the alternate treatment (Fig. 1). Participants followed a controlled diet, supervised by a nutritionist throughout the trial, with several menu options. Although not excessively strict given the five-month duration of the study, the diet was low in polyphenols and regulated intake of choline and carnitine sources (red meat, eggs, fish, and some legumes such as lentils and chickpeas).14 Blood and urine samples, and adherence records were collected at each time point. Refer to SI for additional information.
The sample size for this two-period, two-sequence crossover study was determined following established guidelines for crossover designs in nutrition research and methodological considerations for continuous outcomes in clinical nutrition; i.e., a parallel design requires about five times as many participants as a crossover to achieve similar power and α.22 We performed an a priori sample size justification for a 2 × 2 crossover design with urinary and serum TMAO as the primary endpoint. Assuming a moderate within-subject standardised effect size (paired Cohen's d ≈ 0.5), α = 0.05, and 80% power, crossover trials require fewer participants than parallel designs because power is driven by the within-subject variance of treatment differences. Using a conservative efficiency factor (R ≈ 5), a parallel design achieving significance with n ≈ 30–40 per comparison23,24 would require approximately 10 participants in a crossover design to achieve comparable power. However, given our planned metabotyping and clustering (MCs), which fragment the cohort and reduce per-group sample sizes with uncertain prevalences, we prospectively increased recruitment to n = 90 to ensure adequate power for the overall analysis and for the most prevalent MCs. We further incorporated an anticipated 15% drop-out and accounted for phase and sequence in the mixed/repeated measures analysis, with a washout period to minimise carryover. This conservative, subgroup-oriented planning ensured robust inference for both the primary endpoint and metabotype-specific comparisons. Baseline assessments were performed before any supplementation to ensure unsupplemented reference values.
Sampling procedure
Urine samples were centrifuged at 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g for 10 min at room temperature. A volume of 500 µL of the supernatant was collected and filtered through 13 mm polystyrene syringe filters (PES) with a 0.22 µm pore size (Agilent Technologies, Santa Clara, CA, USA). The filtered urine was subsequently diluted 1
000g for 10 min at room temperature. A volume of 500 µL of the supernatant was collected and filtered through 13 mm polystyrene syringe filters (PES) with a 0.22 µm pore size (Agilent Technologies, Santa Clara, CA, USA). The filtered urine was subsequently diluted 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 to a final volume of 500 µL. In some samples, a 1
100 to a final volume of 500 µL. In some samples, a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 dilution was required to ensure linearity within the calibration range. Concentrations of DMA, TMA, TMAO, and creatinine (used to normalise urinary metabolite concentrations) were determined.
200 dilution was required to ensure linearity within the calibration range. Concentrations of DMA, TMA, TMAO, and creatinine (used to normalise urinary metabolite concentrations) were determined.
Serum samples were processed following the extraction procedure described elsewhere,25 with minor adaptations. Briefly, 20 µL of serum was mixed with 80 µL of MeOH. Proteins were precipitated by vortexing for 1 min, and samples were then centrifuged at 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g for 10 min at 4 °C. The resulting supernatant was carefully recovered and further filtered using 0.22 µm PES syringe filters. A 30 µL aliquot was transferred into a vial insert for injection. After analysing the three metabolites in a representative subset of samples, only TMAO was measured in all the samples.
000g for 10 min at 4 °C. The resulting supernatant was carefully recovered and further filtered using 0.22 µm PES syringe filters. A 30 µL aliquot was transferred into a vial insert for injection. After analysing the three metabolites in a representative subset of samples, only TMAO was measured in all the samples.
Matrix effects were evaluated by comparing the TMAO response in post-extraction spiked serum samples with that obtained from standards prepared in MeOH at equivalent concentrations. As the analyte is endogenously present in serum, background signals from blank serum samples were subtracted from all spiked samples prior to matrix effect calculation. Matrix effects were calculated as the percentage ratio of background-corrected responses in serum and MeOH. In accordance with commonly accepted bioanalytical criteria, matrix effects within ±15% were considered negligible. Under these conditions, the observed matrix effect was below 5%, indicating minimal ion suppression or enhancement and confirming that matrix effects did not compromise quantitative performance; therefore, no correction was required.
Thyroid, hepatic, and renal functions were determined at the hospital's facilities. All metabolites were analysed by UPLC–QqQ using multiple reaction monitoring (MRM), as described below.
UPLC-QqQ-MS/MS analysis
The separation and quantification of TMAO, TMA, DMA, and urinary creatinine in urine and serum samples were performed using a 6460 QqQ-MS/MS (Agilent) equipped with an electrospray ionisation (ESI) source. Chromatographic separation was achieved on an ACQUITY Premier BEH Amide column (2.1 × 100 mm, 1.7 µm; Waters, Germany) maintained at 30 °C. The injection volume was 1 µL, and the flow rate was set at 0.3 mL min−1. The mobile phase consisted of solvent A (20 mM ammonium acetate with 0.1% formic acid in water) and solvent B (100% acetonitrile). The chromatographic run began at 90% B. The proportion of solvent B was then reduced to 60% at 4.6 min and subsequently increased to 90% at 6.0 min. The system was finally returned to the initial conditions and equilibrated for an additional 0.5 min before the next injection.The mass spectrometer was operated in multiple reaction monitoring (MRM) mode. TMAO, TMA, DMA, and creatinine were detected in positive ion mode. Optimised precursor and product ion transitions (Q1 → Q3) and the corresponding collision energies are summarised in Table S2. Data acquisition and integration of chromatographic peaks were carried out using MassHunter Quantitative Analysis software (Agilent Technologies). Calibration curves were constructed using serial dilutions of authentic standards for each analyte prepared in water, and linearity was assessed by the correlation coefficient (R2) exceeding 0.99.
UPLC-ESI-QTOF-MS for metabotype determination
Urinary (poly)phenol-derived metabolites were analysed by Ultra-Performance Liquid Chromatography coupled to Electrospray Ionisation Quadrupole Time-of-Flight Mass Spectrometry (UPLC-ESI-QTOF-MS) using an Agilent 1290 Infinity UPLC system coupled to a 6550 accurate-mass QTOF mass spectrometer (Agilent Technologies, Waldronn, Germany). This analytical platform was used exclusively for metabotype determination. Chromatographic separation and ESI parameters were those recently described.14 Data were processed using Mass Hunter Qualitative Analysis software (version B.08.00, Agilent).Phenolic-derived metabolites were identified by direct comparison with available analytical standards whenever possible. Spectral characteristics such as molecular mass and fragmentation patterns were evaluated to confirm compound assignments. Quantification was performed using extracted ion chromatograms (EICs) with a narrow mass window to minimise peak overlap. Metabolites used for metabotype classification, including the corresponding phase-II metabolites of equol, urolithin A, isourolithin A, urolithin B, and lunularin, were measured using a validated targeted metabolomics method under consistent analytical conditions.14 These measurements ensured accurate detection of phase II conjugates of equol, urolithin, and lunularin, as well as the assignment of participants to their respective metabotype clusters (MCs).
For metabolites without commercial standards, such as isourolithin A sulfate, the identification was based on accurate mass and isotopic distribution; inclusion required a signal-to-noise ratio >10.
Statistical analyses
The effects of polyphenol (PPs) intake on TMAO, TMA, and DMA were examined using a two-way repeated-measures ANOVA, which evaluated within- and between-group variations in the dependent variables across time and treatment conditions (SigmaPlot v16.0, Systat Software, San Jose, CA, USA; jamovi v2.6.26, The jamovi project 2025, https://www.jamovi.org, Sydney, Australia). This approach allowed the assessment of main effects for each factor and their potential interactions. Data normality was verified through the Shapiro–Wilk test. When overall differences reached statistical significance, pairwise comparisons were subsequently explored using the Bonferroni post hoc test.Analyses were carried out considering the entire group as a single population and, additionally, stratifying by individual metabotypes and their corresponding clusters (MCs). Changes in TMAO, TMA, and DMA were estimated using least-squares means (LSM) derived from the two-way repeated-measures ANOVA model that accounted for intra-subject variability and the crossover design of the trial. In cases where the assumption of sphericity was not met, degrees of freedom were corrected using the Greenhouse-Geisser adjustment, yielding non-integer residual values. This procedure ensured reliable inference of treatment effects with appropriate error control.
Cohen's d was calculated for each within-subject comparison (pre- vs. post-intervention) to estimate the effect size of PPs intake on TMAO, TMA, and DMA. Effect sizes were interpreted according to conventional thresholds (small: 0.2 ≤ d < 0.5; medium: 0.5 ≤ d < 0.8; and large: d ≥ 0.8), thereby facilitating the evaluation of clinical relevance across metabotype-defined subgroups.26
Baseline values of TMAO, TMA, and DMA were compared between the two arms at the start of each intervention phase (T1 and T3; Fig. 1) to rule out carryover effects between treatment periods.
Variations in TMAO (post-treatment vs. baseline) were correlated with BMI, chronological age, age at menopause onset, years since menopause, PREDIMED questionnaire, and IPAQ scores to assess potential confounding effects. Depending on data distribution, relationships were tested using multiple linear regression or by calculating Pearson's or Spearman's correlation coefficients. Comparisons between two independent groups (e.g., Pre-M vs. Post-M women at baseline) were performed using independent Student's t-tests for normally distributed data, or Mann–Whitney U tests otherwise. For comparisons between two dependent conditions (e.g., pre- vs. post-intervention with PPs or placebo within each arm, before and after washout, and crossover), paired t-tests were applied for parametric data and Wilcoxon signed-rank tests for nonparametric distributions.
Participants were classified into low, medium, and high producers using unsupervised K-means clustering (k = 3) based on creatinine-normalised metabolite intensities as described elsewhere.20 For each metabotype, the sum of the specific metabolites was considered to define the production groups. Data were log-transformed and autoscaled before analysis in MetaboAnalyst 5.0 (https://www.metaboanalyst.ca). Clustering was performed using the Hartigan–Wong algorithm with Euclidean distance. The thresholds defining the three clusters correspond to the centroid values automatically determined by the algorithm. Data plots were performed using GraphPad Prism 10.4.1 software (GraphPad Software, San Diego, CA, USA) and SigmaPlot v16.0. Statistical significance was set at *p < 0.05, **p < 0.01, and ***p < 0.001.
Results
Baseline characteristics of participants, metabotypes, and metabotype clusters distribution
Baseline characteristics and the distribution of individual metabotypes and metabotype clusters (MCs) are shown in Table 1 and were previously reported in detail.14 In Pre-M, 160 eligible participants were contacted, and 120 agreed to participate (Study 1). Among Post-M participants, 185 were contacted, and 90 were enrolled in Study 2 (Table 1; Fig. 1). The prevalence of equol producers (EP), urolithin A metabotype (UMA, i.e., producing only Uro-A), and lunularin producers (LP, i.e., reducing resveratrol to dihydroresveratrol and subsequently dehydroxylating it to lunularin and 4HDB) was comparable between groups, with only minor differences associated with menopausal status (Table 1). UMA was the most frequent metabotype in both groups. The distribution of MCs showed moderate variation, with cluster MC3 (UMA + EP + LP; 21.8%) predominating in Pre-M women and cluster MC7 (UMA + EP + LNP; 22.5%) in Post-M participants. Clusters MC2 (UMA + ENP + LP; 15.7%), MC3 (15.7%), and MC5 (UMA + ENP + LNP; 18%) were consistently prevalent, whereas clusters MC9 to MC12 (all containing UM0) were not detected in Post-M women (Table 1).| Characteristics | Description | Pre-M (n = 120) | Post-M (n = 90) |
|---|---|---|---|
| Table 1 has been adapted from Jarrín-Orozco et al.14 The numbering of MCs corresponds to the order established in a previous report, based on prevalence rates.13 PREDIMED adherence categories were calculated from the 14-item Mediterranean diet questionnaire (score range 0–14), classified as low (≤5), moderate (6–9), and high (≥10) adherence.27 IPAQ categories were derived from the International Physical Activity Questionnaire-Short Form,28 and were normalised to a 0–100 scale for readability and classified as low (0–33), moderate (34–66), and high (67–100) physical activity levels. | |||
| Age (years) | Mean ± SD; median (range) | 40.2 ± 4.0; 41.0 (30–48) | 53.2 ± 3.3; 54.0 (45–59) |
| BMI (kg m−2) | Mean ± SD; median (range) | 23.7 ± 3.8; 23.0 (18.3–36.2) | 26.0 ± 4.3; 25.1 (19.0–37.4) |
| BMI categories (%) | Normal/overweight/obese | 70.8/22.5/6.7 | 50.0/35.6/14.4 |
| Adherence to the Mediterranean diet (PREDIMED) (%) | Low/medium/high | — | 15.7/74.2/10.1 |
| IPAQ score (%) | Low/medium/high | — | 61.1/32.2/6.7 |
| Menopause onset (years) | Mean ± SD (range) | — | 49.8 ± 3.7 (40–56) |
| Postmenopausal duration (years) | Mean ± SD (range) | — | 3.9 ± 3.5 (0.3–14) |
| Equol metabotype (%) | EP/ENP | 49.6/50.4 | 50.6/49.4 |
| Urolithin metabotypes (%) | UMA/UMB/UM0 | 64.7/32.8/2.5 | 71.9/28.1/0 |
| Lunularin metabotype (%) | LP/LNP | 60.5/39.5 | 50.6/49.4 |
| Metabotype clusters (MC1–MC12) | Percentage per cluster | MC1: 10.9%; MC2: 14.3%; MC3: 21.8%; MC4: 12.6%; MC5: 18.5%; MC6: 6.0%; MC7: 10.1%; MC8: 3.4%; MC9: 0.8%; MC10: 0.8%; MC11: 0.8%; MC12: 0% | MC1: 10.1%; MC2: 15.7%; MC3: 15.7%; MC4: 9.0%; MC5: 18.0%; MC6: 5.6%; MC7: 22.5%; MC8: 3.4%; MC9–MC12: 0% |
TMAO levels are influenced by menopausal status
Urinary TMAO levels were significantly higher in Post-M women compared with Pre-M women (Fig. 2).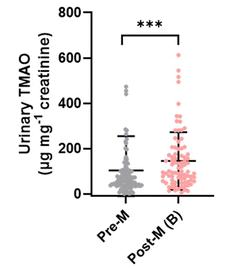 | ||
| Fig. 2 Comparison of urinary TMAO levels between pre-menopausal (Pre-M; n = 120) and post-menopausal women at baseline (Post-M (B); n = 90) ***p < 0.001. | ||
Excreted TMAO levels did not correlate with BMI or age in either Pre-M or Post-M groups (results not shown). In Post-M women, TMAO concentrations were likewise unrelated to age at menopause onset, years since menopause, IPAQ score (which had a low mean, Table 1), or adherence to the Mediterranean diet (PREDIMED), which was moderate in the majority (75%, Table 1) of participants.
Moreover, differences in TMAO between Pre-M and Post-M did not vary when stratified by metabotypes or MCs, indicating that menopausal status influences TMAO concentration independently of other variables.
TMA levels did not differ between Pre-M and Post-M groups (results not shown). In contrast, DMA levels were significantly higher in Post-M than in Pre-M (Fig. S15). Similar to TMAO, TMA and DMA values did not depend on BMI, age at menopause onset, years since menopause, or IPAQ and PREDIMED scores.
Metabotype and cluster shifts following polyphenol-rich extract intake in postmenopausal women
In Study 2, 78 Post-M participants completed the five-month intervention. Adherence to the low-polyphenol diet and capsule intake protocol was high, with full submission of dietary records and minimal deviations in capsule consumption. A few participants reported minor gastrointestinal symptoms, but these were transient and did not affect study completion.After 8 weeks of PPs intake, modest shifts in individual metabotypes were observed. The proportion of UMB increased, while UMA decreased slightly. Both EP and LP metabotypes showed moderate increases, and the overall distribution remained comparable to baseline and to that of Pre-M women.14 MC frequencies also changed moderately. MC3 became more prevalent (20%) and surpassed MC7 (19.2%), which had dominated at baseline. Clusters MC1 and MC4 increased (12.8% and 14.1%, respectively), and MC2 declined (9%). Although the distribution in Post-M women continued to differ modestly from that in Pre-M participants, PPs intake led to partial convergence in MC profiles without substantial reconfiguration.14
Consumption of polyphenol-rich extracts reduces TMAO levels in a metabotype-dependent manner compared with placebo
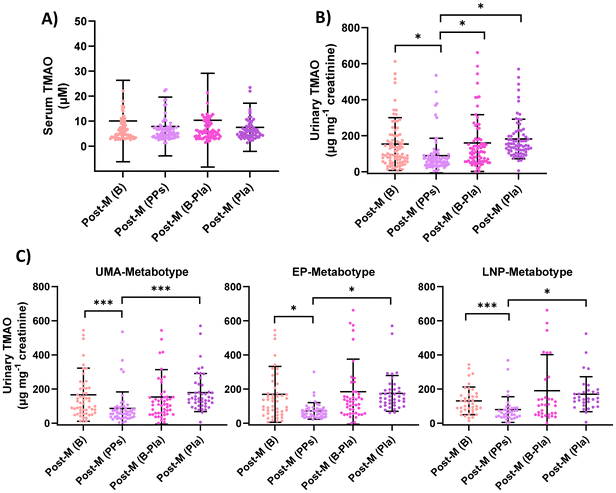
In a first attempt, we determined TMAO in serum. However, no statistically significant changes were observed as a consequence of PPs intake (Fig. 3A). Serum is not a reservoir, and circulating metabolites are strongly conditioned by their turnover in the organism, which can limit the detection of changes in methylamine-related metabolites when using fasting serum. For this reason, we analysed TMAO and related metabolites in urine, normalising diuresis with excreted creatinine. Both serum and urinary TMAO are affected by recent dietary intake, but they reflect different time windows. Urine provides a more integrated measure of TMAO production over time. In contrast, fasting serum represents a single moment and is more sensitive to the rapid turnover of this metabolite. This helps explain the absence of detectable changes in fasting serum despite the clear differences observed in urine. All participants were healthy women with normal renal function, so the use of urinary metabolites was appropriate in this context.In Post-M women, without stratification by metabotypes or MCs, PPs intervention significantly reduced urinary TMAO levels compared with baseline and placebo, with effects of small to moderate magnitude (Table 2 and Fig. 3B). After stratification by metabotypes, the greatest decreases in TMAO, significantly different from both baseline and placebo, were observed in UMA, followed by EP and LNP, also with small to moderate effects, probably influenced by the reduction of the sample size (Table 2 and Fig. 3C). No significant differences in TMAO levels were detected in UMB, ENP, or LP (Fig. S16). The effects of PPs intervention were even more pronounced in MC3 (UMA + EP + LP) and MC7 (UMA + EP + LNP), where PPs intake significantly reduced TMAO with large or very large effects (Table 2 and Fig. 4). Nevertheless, the low n warrants caution, although it exceeded the minimum estimated for the crossover trial design. No significant differences in TMAO levels were found in the remaining MCs (Fig. S17).
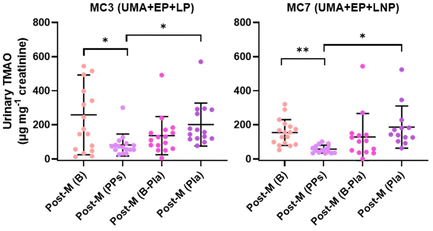 | ||
| Fig. 4 Urinary TMAO changes after polyphenol supplementation (PPs) in postmenopausal (Post-M) women stratified by MCs (MC3 and MC7). Only cases with statistically significant differences are shown. Post-M (B), baseline; Post-M (PPs), after PPs intervention; Post-M (B-Pla), placebo baseline; and Post-M (Pla), after placebo consumption. *p < 0.05, **p < 0.01. Details are provided in Table 2. | ||
| Groups | TMAO difference (LSM, μg mg−1 creatinine) | 95% CI LSM | p-Value | Cohen's d |
|---|---|---|---|---|
| a Only individual metabotypes and MCs with statistically significant differences are shown. Statistically significant differences are boldfaced. Values are expressed as least squares mean (LSM) differences in urinary TMAO, derived from a two-factor (time × treatment) repeated measures ANOVA adjusted for interindividual variability, washout period, and crossover. UMA, Uro-A producers; EP, equol producers; LNP, lunularin non-producers (i.e., resveratrol is reduced to dihydroresveratrol, with no lunularin formation); LP, lunularin producers (i.e., dihydroresveratrol undergoes dehydroxylation to yield lunularin and 4HDB). PPs and Pla indicate within-group changes from baseline to post-intervention. PPs – Pla indicates the LSM difference between PPs and placebo post-intervention. Negative values denote decreases. Positive values denote increases. Cohen's d values quantify the magnitude and direction of group differences, with negative and positive values indicating reductions and increases, respectively. Effect sizes were interpreted using conventional thresholds: small: 0.2 ≤ d < 0.5; medium: 0.5 ≤ d < 0.8; and large: d ≥ 0.8.26 | ||||
| All participants (n = 78) | ||||
| PPs | −70.58 | [−118.5, −22.7] | 0.018 | −0.25 |
| Pla | 18.08 | [−28.5, 64.7] | 0.543 | 0.13 |
| PPs–Pla | −85.97 | [−136.0, −43.0] | 0.013 | −0.39 |
| Metabotypes | ||||
| UMA (n = 49) | ||||
| PPs | −84.30 | [−137.4, −31.2] | 0.005 | −0.38 |
| Pla | 15.73 | [−42.8, 74.2] | 0.543 | 0.07 |
| PPs–Pla | −100.01 | [−156.1, −44.3] | 0.001 | −0.45 |
| EP (n = 46) | ||||
| PPs | −69.40 | [−136.3, −3.5] | 0.033 | −0.21 |
| Pla | −49.77 | [−126.7, 27.1] | 0.543 | −0.19 |
| PPs–Pla | −16.63 | [−120.2, −7.2] | 0.032 | −0.11 |
| LNP (n = 44) | ||||
| PPs | −55.10 | [−108.5, −1.8] | 0.009 | −0.26 |
| Pla | −24.42 | [−72.5, 23.7] | 0.507 | −0.14 |
| PPs–Pla | −30.72 | [−89.1, 27.6] | 0.011 | −0.14 |
| Metabotype clusters (MCs) | ||||
| MC3 (UMA + EP + LP) (n = 16) | ||||
| PPs | −139.10 | [−249.1, −41] | 0.013 | −0.71 |
| Pla | −39.00 | [−13.2, 35.2] | 0.464 | −0.25 |
| PPs–Pla | −100.10 | [−178.2, −22.0] | 0.044 | −0.52 |
| MC7 (UMA + EP + LNP) (n = 16) | ||||
| PPs | −111.41 | [−188.4, −34.4] | 0.005 | −0.71 |
| Pla | 40.39 | [−45.6, 126.4] | 0.328 | 0.23 |
| PPs–Pla | −162.56 | [−252.9, −72.3] | 0.023 | −1.58 |
Regarding TMA, no differences were observed in the entire group or after stratification by metabotypes (results not shown). In contrast, urinary DMA values decreased significantly after PPs intervention, but only in the UMA metabotype (Fig. S18).
The change in urinary TMAO induced by PPs intake did not correlate with BMI, age at menopause onset, or years since menopause. Likewise, no carryover effect was observed. Remarkably, no significant effect was detected after placebo intake in any case (Table 2, Fig. 3 and 4).
Discussion
This study demonstrates, for the first time, that supplementation with selected dietary polyphenols reduces TMAO levels in non-medicated Post-M women compared with placebo, with effects strongly conditioned by gut microbiota metabotypes. The greatest reductions were observed in the urolithin A metabotype (UMA), equol producers (EP), and lunularin non-producers (LNP), particularly in the metabotype clusters MC3 and MC7, which together accounted for 39% of participants.Most studies examining the association between TMAO and cardiovascular risk have used serum or plasma measurements, particularly in clinical populations with elevated risk. Our study focuses on healthy women in a controlled dietary intervention, in which urinary TMAO is commonly used to monitor changes in microbial-derived production over the intervention period.
TMAO is recognised as an independent cardiovascular risk factor, promoting atherosclerosis through foam cell formation, impaired cholesterol efflux, endothelial inflammation, and enhanced platelet reactivity.17 However, causal evidence is primarily supported by preclinical studies, and in humans, intervention trials are limited and often yield mostly associative findings. The relationship between diet and circulating TMAO has been debated for nearly a decade. Adherence to a Mediterranean diet has been inversely associated with urinary TMAO levels, underscoring the role of dietary patterns in shaping the gut microbiota and metabolome.29 In contrast, other reports have shown that TMAO increased after a fibre-enriched Mediterranean diet30 and after the consumption of diets rich in fish-derived n-3 fatty acids and whole-grain cereals (foods generally considered cardioprotective). These findings suggest that TMAO is not a universally valid biomarker of cardiovascular risk independent of dietary background.31
Against this backdrop, several trials have explored the role of polyphenols as possible modulators of TMAO. For example, Annunziata et al. (2019) observed a marked reduction in TMAO in young adults consuming a grape polyphenol extract, despite its very low resveratrol content (135.7 ± 0.64 μg g−1 extract), suggesting synergistic actions of the polyphenolic matrix.32 However, no effect of a much higher resveratrol dose (0.5–1 g) combined with exercise in older adults at high cardiovascular risk was recently reported, suggesting that resveratrol alone might be insufficient.33 Consuming flavanol-rich cocoa improved endothelial function and decreased serum TMAO concentrations in healthy ageing adults.23 Also, a polyphenol-rich tomato extract reduced fasting TMAO levels, with effects more evident in urine than in plasma, and was accompanied by microbial shifts, including decreases in the genera Bacteroides and Ruminococcus and increases in the genus Alistipes.34 Recently, pomegranate polyphenols were shown to inhibit the microbial conversion of choline and carnitine to TMA in vitro.35 Within this evolving evidence, our trial in non-medicated Post-M women adds a new dimension, i.e., polyphenol supplementation significantly reduced TMAO levels compared with placebo, with effects strongly dependent on gut microbiota metabotypes.
A central feature of our study is the inclusion of non-medicated Post-M women. In most previous trials, Post-M participants were either medicated or this information was not specified, and therapies such as hormone replacement or other drugs can alter gut microbiota composition or hepatic metabolism of amines, including FMO3 activity. These confounders make it difficult to attribute net effects to dietary constituents. Although restricting recruitment to non-medicated women is challenging, given the high prevalence of treatments in this population segment, it allows a less confounded assessment of the impact of polyphenols.
Comparison with women of reproductive age (Pre-M) provides additional context. Post-M women showed higher urinary levels of TMAO and DMA than premenopausal women, suggesting that menopause favours a more atherogenic methylamine profile, independent of diet or physical activity. In Pre-M women, lower TMAO levels may reflect both hormonal protection and a more balanced gut microbiota. The reduction of TMAO following polyphenol supplementation in Post-M women supports the hypothesis that diet can partially counteract this unfavourable profile.15 However, the inter-individual variability, as captured by metabotype in our study, appears to modulate the magnitude of the effects. In the present study, gut microbiota analysis was not included, as it is currently ongoing. We focused instead on endpoint changes, particularly TMAO, consistent with other studies and recent trials.23,35 Gut microbiota analysis will complement these findings in the near future, although the most widely accepted mechanism for TMAO reduction is through the gut microbiota.36 Other mechanisms have also been proposed, including downregulation of hepatic FMO3 expression (limiting TMA to TMAO conversion), reduced intestinal absorption of dietary precursors (e.g., choline and carnitine), systemic anti-inflammatory and antioxidant actions (e.g., NFκB pathway attenuation), and improved renal clearance. However, non-microbiota mechanisms remain primarily supported by preclinical evidence.37,38
Although FMO3 expression and/or activity could not be assessed in the participants since it is a hepatic enzyme, its involvement remains a plausible hypothesis. Our metabotypes and MCs analysis indicate that the effect of polyphenols is not uniform. UMA, EP, and LNP women showed greater reductions in TMAO. Importantly, the magnitude of urolithin A, equol, and dihydroresveratrol production did not directly correlate with the decrease in TMAO. This reflects that causality is elusive when linking single-time-point measurements to processes modulated over weeks. It also suggests that benefits may not derive exclusively from the metabolites themselves, but from gut microbial ecosystems capable of producing them, which may simultaneously suppress TMA-generating bacteria, favour competing microbial routes, or activate alternative pathways in the host.39
Our findings on DMA (dimethylamine) are particularly novel. Very few studies have investigated DMA in postmenopausal women, even fewer have addressed its dietary modulation, and none to date have examined its modulation by polyphenols. The higher baseline DMA levels compared with those of Pre-M women, and their selective reduction in UMA, suggest that DMA may serve as a marker of alternative microbial routes in methylamine metabolism. Although its physiological role is not clearly defined, DMA is mostly endogenously produced and is the most abundant short-chain aliphatic amine in human and animal urine,40,41 with elevated levels in renal dysfunction.42 Although certain functions begin to be compromised during menopause, renal, thyroid, and hepatic function values remained within normal reference ranges and were not affected by PPs consumption (results not shown). Therefore, the slightly higher DMA levels observed in Post-M compared with Pre-M cannot be directly attributed to renal dysfunction in our study. DMA is not a direct precursor of TMAO. Instead, TMA can be demethylated by hepatic demethylases and gut microbial pathways to generate DMA.43 Thus, monitoring DMA may provide insights into the complexity of methylamine flux and its dietary responsiveness, an emerging field.
Taken together, our results reinforce the concept of precision health. Stratification of Post-M women by metabotype and MCs allows identification of those most likely to benefit from polyphenol supplementation. This approach addresses variability that has limited reproducibility of previous trials and provides a mechanistic basis for personalised interventions in Post-M women, a group at elevated CVD risk where medication often introduces confounding.
We acknowledge some limitations. The relatively small sizes of some subgroups after clustering limit generalisation and the precision of estimates. Longer studies are needed to assess the persistence of polyphenol effects on TMAO and their possible clinical translation, including vascular inflammation, endothelial function, and platelet activity. Direct evaluation of FMO3 expression and activity in humans remains a challenge. Further research is required to identify possible bioactive intermediates and to resolve the apparent paradox between metabolite production and TMAO reduction.
Conclusion
Polyphenol supplementation in non-medicated Post-M women significantly reduced urinary TMAO and DMA excretion in a metabotype-dependent manner. Compared with fertile (Pre-M) women, menopause was associated with a more atherogenic methylamine profile, which the dietary intervention partially counteracted. In a context where most previous studies included medicated Post-M women, our trial provides a less biased view of polyphenol action.These results support the idea that identifying responsive metabotypes highlights the need to integrate metabolic stratification into precision intervention strategies. Thus, using metabotyping in Post-M women could predict the benefit of polyphenol supplementation, converting interindividual variability into an opportunity for personalised interventions.
(Poly)phenols emerge as dietary modulators and catalysts of precision health, capable of rebalancing methylamine metabolism at a critical stage in women's lives. Overall, these findings provide a mechanistic basis for understanding the heterogeneous outcomes observed in previous trials and reinforce metabotyping as a strategy to mitigate cardiometabolic risk after menopause.
Author contributions
J. C. E.: conceptualisation, funding acquisition, project administration, supervision, writing – original draft, writing – review & editing. M. A. A.-G.: conceptualisation, formal analysis, validation, methodology, investigation, writing – original draft, writing – review & editing; M. P. J.-O., M. G.-N., M. R.-V.: formal analysis, methodology, data curation, investigation, writing – review & editing. J. B., A. S.-S., J. P., C. C.: methodology, investigation, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The datasets generated and analysed during the current study cannot be made publicly available due to General Data Protection Regulation (GDPR) requirements and Ethics Committee restrictions, as full anonymisation of individual-level health data cannot be ensured. Anonymised data may be shared upon reasonable request to the corresponding sponsor of the trial (J.C.E.), in accordance with institutional and ethical requirements. The Supplementary Information provides extended methodological details, synthetic procedures for urolithin and lunularin metabolites, full 1H/13C NMR spectra, additional statistical analyses, and supplementary figures and tables supporting the main findings.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo05461c.
Acknowledgements
This work has been funded by the Projects PID2022-136419OB-100 (Ministerio de Ciencia, Innovación y Universidades, MICIU, AEI/10.13039/501100011033, Spain) with co-funding from the EU “ERDF A way of making Europe” and the AGROALNEXT programme (PRTR-C17.I1, MICIU, Spain) with co-funding from the NextGenerationEU and Fundación Séneca (Comunidad Autónoma Región de Murcia, Spain). M. G.-N. acknowledges funding from a Juan de la Cierva Postdoctoral Contract (JDC2023-051288-I) funded by MCIU/AEI/10.13039/501100011033 and the FSE+.References
- S. R. Davis, J. Pinkerton, N. Santoro and T. Simoncini, Menopause-Biology, consequences, supportive care, and therapeutic options, Cell, 2023, 186, 4038–4058 Search PubMed.
- E. R. Uddenberg, N. Safwan, M. Saadedine, M. D. Hurtado, S. S. Faubion and C. L. Shufelt, Menopause transition and cardiovascular disease risk, Maturitas, 2024, 185, 107974 CrossRef CAS PubMed.
- C. Gatenby and P. Simpson, Menopause: Physiology, definitions, and symptoms, Best Pract. Res., Clin. Endocrinol. Metab., 2024, 38, 101855 Search PubMed.
- S. Wang, Q. Du, X. Meng and Y. Zhang, Natural polyphenols: a potential prevention and treatment strategy for metabolic syndrome, Food Funct., 2022, 13, 9734–9753 Search PubMed.
- J. C. Espín, M. P. Jarrín-Orozco, L. Osuna-Galisteo, M. Á. Ávila-Gálvez, M. Romo-Vaquero and M. V. Selma, Perspective on the Coevolutionary Role of Host and Gut Microbiota in Polyphenol Health Effects: Metabotypes and Precision Health, Mol. Nutr. Food Res., 2024, 68, e2400526 CrossRef PubMed.
- M. Villalva, E. García-Díez, M. D. C. López de Las Hazas, O. Lo Iacono, J. I. Vicente-Díez, S. García-Cabrera, M. Alonso-Bernáldez, A. Dávalos, M. Á. Martín, S. Ramos and J. Pérez-Jiménez, Cocoa-carob blend acute intake modifies miRNAs related to insulin sensitivity in type 2 diabetic subjects: a randomised controlled nutritional trial, Food Funct., 2025, 16, 3211–3226 Search PubMed.
- C. E. Iglesias-Aguirre, A. Cortés-Martín, M. Á. Ávila-Gálvez, J. A. Giménez-Bastida, M. V. Selma, A. González-Sarrías and J. C. Espín, Main drivers of (poly)phenol effects on human health: metabolite production and/or gut microbiota-associated metabotypes?, Food Funct., 2021, 12, 10324–10355 Search PubMed.
- J. Bajerska, K. Łagowska, M. Mori, J. Reguła, A. Skoczek-Rubińska, T. Toda, N. Mizuno and Y. Yamori, A Meta-Analysis of Randomized Controlled Trials of the Effects of Soy Intake on Inflammatory Markers in Postmenopausal Women, J. Nutr., 2022, 152, 5–15 Search PubMed.
- M. Abshirini, M. Omidian and H. Kord-Varkaneh, Effect of soy protein containing isoflavones on endothelial and vascular function in postmenopausal women: a systematic review and meta-analysis of randomised controlled trials, Menopause, 2020, 27, 1425–1433 Search PubMed.
- M. L. Rodrigues Uggioni, L. Ronsani, S. Motta, J. C. Denoni Júnior, F. Marçal, S. Dagostin Ferraz, M. I. Rosa and T. Colonetti, Effects of resveratrol on the lipid profile of post-menopause women: Systematic review and meta-analysis, Nutr. Metab. Cardiovasc. Dis., 2025, 35, 103827 CrossRef CAS PubMed.
- C. Manach, A. Scalbert, C. Morand, C. Rémésy and L. Jiménez, Polyphenols: food sources and bioavailability, Am. J. Clin. Nutr., 2004, 79, 727–747 Search PubMed.
- L. Lavefve, L. R. Howard and F. Carbonero, Berry polyphenols metabolism and impact on human gut microbiota and health, Food Funct., 2020, 11, 45–65 RSC.
- E. C. Iglesias-Aguirre, M. Romo-Vaquero, M. V. Selma and J. C. Espín, Unveiling metabotype clustering in resveratrol, daidzein, and ellagic acid metabolism: Prevalence, associated gut microbiomes, and their distinctive microbial networks, Food Res. Int., 2023, 173, 113470 CrossRef PubMed.
- M. P. Jarrín-Orozco, M. Romo-Vaquero, C. Carrascosa, M. Pertegal, J. Berná, J. Puigcerver, A. Saura-Sanmartín, I. Espinosa-Salinas, M. García-Nicolás, M. Á. Ávila-Gálvez and J. C. Espín, Polyphenol-Related Gut Metabotype Signatures Linked to Quality of Life in Postmenopausal Women: A Randomised, Placebo-Controlled Crossover Trial, Nutrients, 2025, 17, 3572 CrossRef PubMed.
- C. Simó and V. García-Cañas, Dietary bioactive ingredients to modulate the gut microbiota-derived metabolite TMAO, New opportunities for functional food development, Food Funct., 2020, 11, 6745–6776 RSC.
- M. He, C. P. Tan and Y. Liu, Gut Microbiota-Derived Metabolites and Cardiovascular Disease: Focus on Trimethylamine-N-oxide, J. Agric. Food Chem., 2025, 73, 30569–30581 Search PubMed.
- R. A. Koeth, Z. Wang, B. S. Levison, J. A. Buffa, E. Org, B. T. Sheehy, E. B. Britt, X. Fu, Y. Wu, L. Li, J. D. Smith, J. A. DiDonato, J. Chen, H. Li, G. D. Wu, J. D. Lewis, M. Warrier, J. M. Brown, R. M. Krauss, W. H. Tang, F. D. Bushman, A. J. Lusis and S. L. Hazen, Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis, Nat. Med., 2013, 19, 576–585 CrossRef CAS PubMed.
- Z. S. DiMattia, J. Zhao, F. Hao, S. Koshkin, J. E. Bisanz, A. D. Patterson, J. A. Fleming, P. M. Kris-Etherton and K. S. Petersen, Effect of Varying Quantities of Lean Beef as Part of a Mediterranean-Style Dietary Pattern on Gut Microbiota and Plasma, Fecal, and Urinary Metabolites: A Randomized Crossover Controlled Feeding Trial, J. Am. Heart Assoc., 2025, 14, e041063 Search PubMed.
- M. Ş. Deniz and M. Baş, Short-Term Mediterranean Dietary Intervention Reduces Plasma Trimethylamine-N-Oxide Levels in Healthy Individuals, Nutrients, 2025, 17, 3135 Search PubMed.
- C. E. Iglesias-Aguirre, F. Vallejo, D. Beltrán, E. Aguilar-Aguilar, J. Puigcerver, M. Alajarín, J. Berná, M. V. Selma and J. C. Espín, Lunularin Producers versus Non-producers: Novel Human Metabotypes Associated with the Metabolism of Resveratrol by the Gut Microbiota, J. Agric. Food Chem., 2022, 70, 10521–10531 Search PubMed.
- M. Á. Ávila-Gálvez, R. García-Villalba, F. Martínez-Díaz, B. Ocaña-Castillo, T. Monedero-Saiz, A. Torrecillas-Sánchez, B. Abellán, A. González-Sarrías and J. C. Espín, Metabolic Profiling of Dietary Polyphenols and Methylxanthines in Normal and Malignant Mammary Tissues from Breast Cancer Patients, Mol. Nutr. Food Res., 2019, 63, e1801239 Search PubMed.
- J. E. Harris and H. A. Raynor, Crossover Designs in Nutrition and Dietetics Research, J. Acad. Nutr. Diet., 2017, 117, 1023–1030 Search PubMed.
- J. García-Cordero, A. Martínez, C. Blanco-Valverde, A. Pino, V. Puertas-Martín, R. San Román and S. de Pascual-Teresa, Regular Consumption of Cocoa and Red Berries as a Strategy to Improve Cardiovascular Biomarkers via Modulation of Microbiota Metabolism in Healthy Aging Adults, Nutrients, 2023, 15, 2299 Search PubMed.
- B. Amato, E. Novellino, D. Morlando, C. Vanoli, E. Vanoli, F. Ferrara, R. Difruscolo, V. M. Goffredo, R. Compagna, G. C. Tenore, N. Stornaiuolo, M. Fordellone and E. Caradonna, Benefits of Taurisolo in Diabetic Patients with Peripheral Artery Disease, J. Cardiovasc. Dev. Dis., 2024, 11, 174 Search PubMed.
- Z. Wang, N. Bergeron, B. S. Levison, X. S. Li, S. Chiu, X. Jia, R. A. Koeth, L. Li, Y. Wu, W. H. W. Tang, R. M. Krauss and S. L. Hazen, Impact of chronic dietary red meat, white meat, or non-meat protein on trimethylamine N-oxide metabolism and renal excretion in healthy men and women, Eur. Heart J., 2019, 40, 583–594 CrossRef CAS PubMed.
- J. Cohen, Statistical Power Analysis for the Behavioral Sciences, (2nd ed.), Lawrence Erlbaum Associates, Hillsdale (NJ), 1988 Search PubMed.
- M. A. Martínez-González, E. Fernández-Jarne, M. Serrano-Martínez, M. Wright and E. Gómez-Gracia, Development of a short dietary intake questionnaire for the quantitative estimation of adherence to a cardioprotective Mediterranean diet, Eur. J. Clin. Nutr., 2004, 58, 1550–1552 Search PubMed.
- C. L. Craig, A. L. Marshall, M. Sjöström, A. E. Bauman, M. L. Booth, B. E. Ainsworth, M. Pratt, U. Ekelund, A. Yngve, J. F. Sallis and P. Oja, International physical activity questionnaire: 12-country reliability and validity, Med. Sci. Sports Exercise, 2003, 35, 1381–1395 Search PubMed.
- F. De Filippis, N. Pellegrini, L. Vannini, I. B. Jeffery, A. La Storia, L. Laghi, D. I. Serrazanetti, R. Di Cagno, I. Ferrocino, C. Lazzi, S. Turroni, L. Cocolin, P. Brigidi, E. Neviani, M. Gobbetti, P. W. O'Toole and D. Ercolini, High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome, Gut, 2016, 65, 1812–1821 CrossRef CAS PubMed.
- C. Barber, M. Mego, C. Sabater, F. Vallejo, R. A. Bendezu, M. Masihy, F. Guarner, J. C. Espín, A. Margolles and F. Azpiroz, Differential Effects of Western and Mediterranean-Type Diets on Gut Microbiota: A Metagenomics and Metabolomics Approach, Nutrients, 2021, 13, 2638 Search PubMed.
- G. Costabile, C. Vetrani, L. Bozzetto, R. Giacco, L. Bresciani, D. Del Rio, M. Vitale, G. Della Pepa, F. Brighenti, G. Riccardi, A. A. Rivellese and G. Annuzzi, Plasma TMAO increase after healthy diets: results from 2 randomised controlled trials with dietary fish, polyphenols, and whole-grain cereals, Am. J. Clin. Nutr., 2021, 114, 1342–1350 Search PubMed.
- G. Annunziata, M. Maisto, C. Schisano, R. Ciampaglia, V. Narciso, G. C. Tenore and E. Novellino, Effects of Grape Pomace Polyphenolic Extract (Taurisolo) in Reducing TMAO Serum Levels in Humans: Preliminary Results from a Randomized, Placebo-Controlled, Crossover Study, Nutrients, 2019, 11, 139 CrossRef CAS PubMed.
- L. C. Baptista, L. Wilson, S. Barnes, S. D. Anton and T. W. Buford, Effects of resveratrol on changes in trimethylamine-N-oxide and circulating cardiovascular factors following exercise training among older adults, Exp. Gerontol., 2024, 194, 112479 CrossRef CAS PubMed.
- A. Rehman, S. M. Tyree, S. Fehlbaum, G. DunnGalvin, C. G. Panagos, B. Guy, S. Patel, T. G. Dinan, A. K. Duttaroy, R. Duss and R. E. Steinert, A water-soluble tomato extract rich in secondary plant metabolites lowers trimethylamine-N-oxide and modulates gut microbiota: a randomised, double-blind, placebo-controlled crossover study in overweight and obese adults, J. Nutr., 2023, 153, 96–105 Search PubMed.
- J. E. Haarhuis, P. Day-Walsh, E. Shehata, G. M. Savva, B. Peck, M. Philo and P. A. Kroon, A Pomegranate Polyphenol Extract Suppresses the Microbial Production of Proatherogenic Trimethylamine (TMA) in an In Vitro Human Colon Model, Mol. Nutr. Food Res., 2025, 69, e70166 CrossRef CAS PubMed.
- Z. Wang, E. Klipfell, B. J. Bennett, R. Koeth, B. S. Levison, B. Dugar, A. E. Feldstein, E. B. Britt, X. Fu, Y. M. Chung, Y. Wu, P. Schauer, J. D. Smith, H. Allayee, W. H. Tang, J. A. DiDonato, A. J. Lusis and S. L. Hazen, Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease, Nature, 2011, 472, 57–63 CrossRef CAS PubMed.
- D. Li, Y. Li, S. Yang, X. Zhang, Y. Cao, R. Zhao, Y. Zhao, X. Jin, J. Lu, X. Wang, Q. Wang, L. Liu and M. Wu, Polydatin combined with hawthorn flavonoids alleviate high fat diet induced atherosclerosis by remodeling the gut microbiota and glycolipid metabolism, Front. Pharmacol., 2025, 16, 1515485 Search PubMed.
- M. Canyelles, M. Tondo, L. Cedó, M. Farràs, J. C. Escolà-Gil and F. Blanco-Vaca, Trimethylamine N-Oxide: A Link among Diet, Gut Microbiota, Gene Regulation of Liver and Intestine Cholesterol Homeostasis and HDL Function, Int. J. Mol. Sci., 2018, 19, 3228 CrossRef PubMed.
- A. Cortés-Martín, M. V. Selma, F. A. Tomás-Barberán, A. González-Sarrías and J. C. Espín, Where to Look into the Puzzle of Polyphenols and Health? The Postbiotics and Gut Microbiota Associated with Human Metabotypes, Mol. Nutr. Food Res., 2020, 64, e1900952 Search PubMed.
- A. Q. Zhang, S. C. Mitchell and R. L. Smith, Dimethylamine in human urine, Clin. Chim. Acta, 1995, 233, 81–88 Search PubMed.
- D. Tsikas, Urinary Dimethylamine (DMA) and Its Precursor Asymmetric Dimethylarginine (ADMA) in Clinical Medicine, in the Context of Nitric Oxide (NO) and Beyond, J. Clin. Med., 2020, 9, 1843 Search PubMed.
- D. Tsikas, Analysis, biology and significance of dimethylamine, trimethylamine and trimethylamine N-oxide in humans and in the marine ecosystem, J. Chromatogr. B:Anal. Technol. Biomed. Life Sci., 2025, 1258, 124602 Search PubMed.
- I. Gut and A. H. Conney, The FAD-containing monooxygenase-catalysed N-oxidation and demethylation of trimethylamine in rat liver microsomes, Drug Metabol. Drug Interact., 1991, 9, 201–208 Search PubMed.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |