Open Access Article
Open Access ArticleUrinary track health benefits and phytochemical characterization based on a narrative review on cranberry and its innovative formulation in phospholipids
Francesco Scaglione
School of Clinical Pharmacology and Toxicology, University of Milan, Milan, Italy. E-mail: francesco.scaglione@unimi.it
First published on 9th April 2026
Abstract
Cranberry is rich in nutritional components and numerous bioactive compounds with properties useful in a variety of physiological and clinical conditions. Both the American (Vaccinium macrocarpon) and European (Vaccinium oxycoccus) species are rich in (poly)phenols, including flavonoids and phenolic acids (anthocyanins, flavonoids, and polyphenols such as phenolic acids). Cranberry is one of the few fruits with a high content of type A proanthocyanidins, associated with numerous health benefits, such as reducing recurrent urinary tract infections (UTIs). This review analyses information on the chemical composition of cranberry, the beneficial impact on human health, and disease prevention after cranberry consumption. Specifically, the mechanism of action of its benefits against urinary tract infections (UTIs), the second most common type of infection worldwide, is explored. Finally, the review focuses on the characteristics of new formulation technologies to increase bioavailability, such as new phospholipid-based delivery systems capable of dispersing cranberry extract in sunflower lecithin, thus improving its stability, solubility, absorption, and biological efficacy at lower doses. In conclusion, the available literature suggests that cranberry products are widely supported for reducing recurrent urinary tract infections, especially in women and children. Their mechanisms include anti-adhesion, anti-biofilm, and antimicrobial effects. They are generally safe and well-tolerated, although some restrictions apply (drug interactions, pre-existing conditions). Innovative delivery systems (such as phospholipids, microcapsules, gel formulations) optimize application, allowing for lower effective doses and lasting benefits, primarily improving safety. Further large-scale, high-quality studies are needed, especially for uses in non-urological areas, to confirm broader health impacts.
A brief history of cranberry
Cranberries belong to the Ericaceae family and are primarily represented by two species: the American cranberry (Vaccinium macrocarpon) and the European cranberry (Vaccinium oxycoccos). The “large” American cranberry is mainly cultivated in North America, especially in the northern United States and Canadian provinces, as well as in Chile and in commercial farms in parts of Europe.1,2 The American variety is a low-growing evergreen shrub with slender stems and small, elliptical leaves. Its berries ripen from pale pink to dark purple, typically from late summer through early autumn. In contrast, the European cranberry is smaller, grows wild across parts of Europe and Asia, and is better adapted to colder climates.2,3 The term “cranberry” is thought to have originated from early observations by Indigenous peoples of North America, who noted the resemblance of the plant's floral parts to a crane's neck and beak.1 Because of their intense tartness, only about 5% of cranberries are sold fresh; the vast majority are processed into juices, sauces, and dried products. In addition to their culinary uses, cranberries have a longstanding history as both a food and medicinal plant in North America, Europe, and Asia.Chemical composition
Cranberries have a complex and rich chemical composition, with essential nutrients alongside a variety of bioactive compounds. These chemical constituents can vary significantly between species and cultivars, influenced by both genetic and environmental factors.Water is the predominant component of the fruit, comprising approximately more than 80% of the mass. Carbohydrates make up around 12%, primarily in the form of simple sugars such as glucose, fructose, and sucrose, with glucose typically being the most abundant.2 Large-fruited cranberries tend to contain higher concentrations of both monosaccharides (ranging from 3% to 7%) and sucrose (3.9% to 5.3%) compared to their smaller European counterparts (Vaccinium oxycoccos), which exhibit lower sugar levels (2% to 6% for monosaccharides and 0.01% to 0.5% for sucrose).1,4 Oligosaccharides represent an important component that could be responsible for some health benefits through prebiotic, anti-adhesive, anti-inflammatory and antioxidant activity.5 The perceived sweetness of cranberries is largely attributed to these sugars, though it remains tempered by the fruit's high organic acid content. Among these organic acids, citric, malic, and quinic acids are predominant, contributing to the fruit's notable acidity. Titratable acidity, primarily from citric acid, ranges from 2.1% to 4.9% in smaller cranberries and is generally lower in larger varieties (1.9% to 2.4%).1 These levels are influenced by a variety of factors, including cultivar, ripeness, climate, and soil conditions.
Cranberries are also recognized for their vitaminic profile, particularly their high content of vitamins C, E, and K, which underlie many of their antioxidant properties. The American cranberry contains higher levels of ascorbic acid, with reported values reaching up to 47.5%, compared to 15.3% to 30% in European species. Additionally, although present in small quantities, omega-3 and omega-6 fatty acids are also found in cranberry fruit, contributing further to its nutritional value.1,2 Mineral content in cranberries is similarly notable. Potassium is the most abundant micronutrient in both major species, but trace elements such as manganese and boron can also be found.6
Beyond their nutritional constituents, cranberries are particularly rich in phytochemicals, many of which have been associated with potential health effects. Among the most prominent are phenolic acids, triterpenoids, and flavonoids.7,8 Phenolic acids in cranberries include both hydroxybenzoic and hydroxycinnamic acids. Benzoic acid is especially abundant, accompanied by smaller quantities of caffeic, ferulic, p-coumaric, and chlorogenic acids.9 Triterpenoids, another important class of cranberry phytochemicals, are found mainly in the skin. These include ursolic acid, oleanolic acid, and betulinic acid, with ursolic acid in particular garnering attention for its anti-inflammatory activity.8,10
Flavonoids are a major group of compounds found in cranberries, classified into subgroups including anthocyanins, flavonols, and proanthocyanidins (also known as flavan-3-ols when referring to their monomeric forms).4,8 Anthocyanins, the pigments responsible for cranberries’ vivid red coloration, are found in much higher concentrations in the skin than in the pulp. Their levels increase markedly as the berries ripen. Major anthocyanins include cyanidin-3-galactoside, cyanidin-3-glucoside, cyanidin-3-arabinoside, peonidin-3-galactoside, and peonidin-3-arabinoside.2,11 Flavonols, primarily quercetin and myricetin derivatives, are present throughout ripening and show a modest increase as the fruit matures. These include compounds such as quercetin-3-O-galactoside and myricetin-3-O-galactoside, which contribute to the fruit's antioxidant capacity.9,10 Among the most studied cranberry flavonoids are flavan-3-ols and their polymeric forms, proanthocyanidins (PACs). These compounds play critical roles in plant defence and are widely recognized for their antimicrobial, antioxidant, and anti-inflammatory properties.9 Catechin and epicatechin are the principal flavan-3-ols identified in cranberries, with epicatechin being a dominant constituent of PACs. The concentration of these compounds tends to peak in early ripening stages and diminishes as the fruit overripens. Notably, American cranberries generally contain higher levels of A-type PACs compared to European varieties.2,7,8 It is known that A-type PACs and oligosaccharides could act in a synergistic way to enhance their beneficial properties.5
Cranberry health benefits
This rich source of bioactive compounds has been investigated in studies as a potential supplementation for different urological and even non-urological applications. Overall, cranberries have also been shown health benefits as following: inhibit progression of degenerative unhealthy conditions and loss of functionality as neural degeneration; promotion of cardiovascular health; promotion of gastrointestinal health; oral benefits linked also to antiviral potentials; prevention and inhibition of cancer; modulation of inflammatory responses, antioxidant properties.2,12 A diagram showed a summary of potential cranberry benefits (Fig. 1).The presence of antioxidants like flavonoids, anthocyanins, and tannins, as well as vitamins C, E, and K has raised interest for their activity in scavenging free radicals and neutralizing reactive oxygen species, helping to protect cells from oxidative damage.13 On the contrary, (poly)phenol antioxidant action derives more from an indirect modulation of cellular signaling pathways related to antioxidant defence.14 Their anti-inflammatory properties are also well documented and largely attributed to (poly)phenols and triterpenoids such as ursolic acid, which can modulate inflammatory pathways by downregulating pro-inflammatory cytokines like TNF-α, IL-1β, and IL-6, while promoting anti-inflammatory mediators.15
Cranberries may further exert cardioprotective effects by improving lipid profiles,16 and lowering both systolic and diastolic blood pressure, possibly through nitric oxide-mediated vasodilation.17 Their positive effect on glycemia modulation potential has been demonstrated through improvements in insulin sensitivity, as well as other positive effects on metabolism in diet-induced obesity in mice.18–20
In the gastrointestinal tract, cranberry (poly)phenols modulate the gut microbiota by promoting growth of beneficial species like Lactobacillus and Bifidobacterium, suppressing harmful bacteria and exerting prebiotic and anti-inflammatory effects.21,22
Additionally, cranberry-derived phytochemicals, particularly proanthocyanidins (PACs) and flavonoids, have shown in vitro anticancer activity through inhibition of tumor cell proliferation, invasion, and metastasis, and by inducing apoptosis in various cancer cell lines.23,24
Neuroprotective properties have also been observed, with evidence suggesting cranberry intake may protect against neurodegenerative processes seen in disease models.25,26
Collectively, these diverse bioactivities underscore the broad potential of cranberries in supporting human health against chronic disease.
Focus on urinary tract: mechanism of action
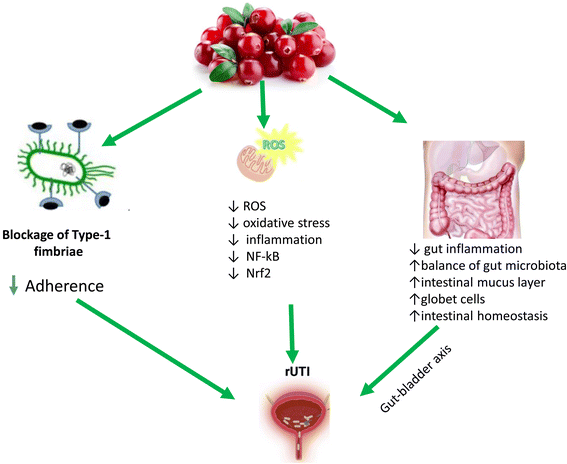
Among the various applications of cranberry, its most well-known and extensively studied role is in the management of urinary tract infections (UTIs). This activity is attributed to several complementary mechanisms involving different aforementioned bioactive compounds.A-type proanthocyanidins (PACs) are unique polyphenols containing double linkages (A-type) that inhibit the adhesion of P-fimbriated Escherichia coli (E. coli) to urinary tract cells, helping to prevent urinary tract infections. These potent compounds also exhibit antioxidative, anti-inflammatory, and potential anti-cancer activities.27 By preventing bacterial adhesion, cranberries interfere with the critical first step of infection development and progression.28,29 A previous clinical study in healthy volunteers showed that administration of PAC-standardized cranberry powder (72 mg of PAC per day) may offer some protection against bacterial adhesion and virulence in the urinary tract.30 These data were confirmed in a study where the phenyl-γ-valerolactone cranberry metabolites and their conjugated forms (sulphated) were able to inhibit the adherence of uropathogenic Escherichia coli in bladder T24 epithelial cells.31
Second, in addition to preventing adhesion, cranberry compounds also demonstrate anti-biofilm properties. Flavonols and (poly)phenols have been shown to contribute to biofilm inhibition, with some showing synergistic effects with A-Type PACs.32 (Poly)phenol-rich cranberry extracts have been shown to reduce the formation and stability of polymicrobial biofilms by decreasing biomass, acidogenicity, and the ratio of exopolysaccharide to microbial biovolume, as well as colony-forming unit counts. These effects target bacterial adhesion and biofilm development in its early stages.5,33,34
Third, cranberries possess broader antimicrobial activity, including bacteriostatic and bactericidal effects against various human pathogens.35–38 This is partly reinforced by the presence of organic acids in cranberry juice, such as quinic, malic, shikimic, and citric acid, which may act synergistically. Some studies have suggested that combinations of these acids improve antibacterial efficacy.5,39
Furthermore, cranberry urinary metabolites [5-(3′,4′-dihydroxyphenyl)-γ-valerolactone and 4-hydroxybenzoic acid (4-HBA)] showed inhibitory activity of Candida albicans biofilm early-phase formation, possibly preventing genitourinary Candida infections.33,40
Lastly, cranberries may also impact microbial communities beyond the urinary tract. Intake of cranberry juice has been associated with a reduction in Proteobacteria, including E. coli, and an increase in beneficial Firmicutes within the urinary microbiome of children.41 Similarly, cranberry juice has been observed to lower levels of pathogenic Streptococcus species and increase commensal bacteria in the vaginal microbiota, suggesting a broader modulatory role in maintaining urogenital health.42
As summarized in Fig. 2, it is well known that cranberry contributes to UTIs prevention. Additionally, due to the biological effects of its phytochemicals, cranberry may act on an impaired Nrf2/NF-κB system, mitochondrial dysfunctions, and gut microbiota imbalance, alleviating oxidative stress and inflammation present in individuals suffering from recurrent UTIs (rUTI).
Clinical use
EMA monography
Interestingly, the European Medicines Agency (EMA) has developed a monograph for Vaccinii macrocarpi fructus as a traditional herbal medicinal product.43 It recognizes the traditional use of a preparation made from the expressed juice of fresh cranberry fruits, administered orally in liquid form. This herbal preparation is indicated for two uses: first, for the relief of symptoms of mild, recurrent lower urinary tract infections in women, such as dysuria and/or pollakiuria, after serious conditions have been excluded by a medical doctor; and second, for the prevention of recurrent, uncomplicated lower urinary tract infections in women, again following the exclusion of serious underlying conditions by a physician. Importantly, in both cases, the product is classified as a traditional herbal medicinal product, and its use is supported exclusively by long-standing tradition, without the requirement for pharmacokinetic or pharmacodynamic, in accordance with Directive 2001/83/EC (Directive – 2001/83 – EN – EUR-Lex). According to the monograph, the recommended dosage for adult and elderly women is 50–60 mL of expressed juice, taken two to four times daily for symptomatic relief, and 30 mL once daily for prophylaxis.Use in children and adolescents is not recommended. Contraindications include hypersensitivity to cranberry and use in patients with kidney disorders who have experienced recurrent urinary tract infections, as these individuals require medical supervision. Concomitant use with warfarin or tacrolimus is also contraindicated;43 in fact, cranberry juice has been reported to reduce serum levels of tacrolimus,44 and to potentiate the effect of warfarin, increasing the risk of bleeding45,46 although this last interaction has been discussed.47
The use of cranberry preparations in men and in pregnant women is not actually recommended, as lower urinary tract symptoms in these populations require clinical evaluation. Furthermore, cranberry concentrate has a high oxalate content, raising concerns about the potential risk of stone formation in patients with a history of urolithiasis. Although no severe toxicity has been reported, mild adverse effects such as gastrointestinal discomfort, nausea, diarrhea, and dyspepsia, as well as hypersensitivity reactions like urticaria and rash, may occur.43
Clinical evidence from meta-analysis
| Author | Title | Inclusion criteria | Exclusion criteria | N studies included | N participants | Population | Intervention | Comparator | Outcome | Setting | Findings |
|---|---|---|---|---|---|---|---|---|---|---|---|
| BP: blood pressure; CI: confidence interval; CVD: cardiovascular disease; DBP: diastolic blood pressure; FBG: fasting blood glucose; HbA1c: glycated hemoglobin; HDL-C: high-density lipoprotein cholesterol; HOMA-I: homeostatic model assessment of insulin resistance; LDL-C: low-density lipoprotein cholesterol; MD: mean difference; OR: odds ratio; RCT: randomized controlled trial; RR: relative risk; SBP: systolic blood pressure; TC: total cholesterol; TG: triglycerides. CI: confidence interval; MD: mean difference; PAC: proanthocyanidin; RCT: randomized controlled trial; RR: relative risk; UTI: urinary tract infection. | |||||||||||
| Panel A | |||||||||||
| X. Li et al., (2024)82 | The effects of cranberry consumption on glycemic and lipid profiles in humans: a systematic review and meta-analysis of randomized controlled trials | - RCTs (parallel or cross-over) | - In vitro, animal, non-interventional or non-RCTs | 16 | 708 | Adults | Cranberry supplements | Placebo or none | Blood lipid and glucose-related indicators | RCTs | - Significant reduction in TC/HDL-C ratio (4 studies, mean difference MD = −0.24; 95% CI: −0.45, −0.04; peffect = 0.02) and HOMA-IR (5 studies, MD = −0.59; 95% CI: −1.05, −0.14; peffect = 0.01) |
| - Participants >18 years | - Duplicates | - No effect on TC, HDL-C, LDL-C, TG, FBG, HbA1c, or fasting insulin overall | |||||||||
| - Cranberry supplementation used | - Incomplete data on lipids or glucose | - Subgroup analysis: dried form (capsules, powder, tablets) reduced fasting insulin (3 studies, 165 sixty-five participants, MD = −2.16; 95% CI: −4.24, −0.07; p effect = 0.04) | |||||||||
| - Reported blood lipid/glucose indicators (TC, TG, HDL-C, LDL-C, FBG, insulin, HbA1c, HOMA-IR) | - Cranberry combined with other antioxidants (e.g., omega-3, L-citrulline) | ||||||||||
| - Intervention <2 weeks | |||||||||||
| Delpino et al., (2024)83 | The effects of blueberry and cranberry supplementation on blood pressure in patients with cardiovascular diseases: a systematic review and meta-analysis of randomized clinical trials | - Original RCTs (cross-over or parallel) | - Healthy individuals | 17 (8 with cranberry) | 835 (cranberry 373) | Adults with cardiometabolic disease | Cranberry or blueberry (any form) | Placebo or any intervention | SBP, DBP | RCTs | - No significant effect on SBP (MD = −0.81 mmHg, 95%CI −2.26–0.63) or DBP (MD = −0.15 mmHg, 95%CI −1.36–1.05) therefore blueberry/cranberry supplementation had neutral effect on BP in cardiometabolic disease patients |
| - Intervention ≥1 week | - In vitro or animal studies | ||||||||||
| - Participants ≥18 years | - No baseline or endpoint data | ||||||||||
| - Used blueberry or cranberry (any form) | - Non-controlled trials | ||||||||||
| - Reported SBP and DBP at baseline and endpoint | - Cranberry combined with other interventions without control comparison | ||||||||||
| - Participants had cardiometabolic diseases | - Unpublished data, abstracts, patents, theses | ||||||||||
| - Published in English, Portuguese, or Spanish | - No significant results for cranberry alone after stratification (SBP/DBP MD = −1.39 mmHg) | ||||||||||
| Wang et al., (2023)84 | Effect of polyphenol compounds on Helicobacter pylori eradication: a systematic review with meta-analysis | - Published in English or Chinese | - Duplicate articles or same samples | 12 (4 cranberry) | 1251 (699 cranberry) | Adults with H. pylori infection | (Poly)phenol compounds alone or in addition to other treatments | Placebo/eradication therapy | H. pylori eradication rate | RCTs | - Polyphenol compounds significantly increased H. pylori eradication (RR = 1.19; 95% CI: 1.03–1.38; p = 0.02) |
| - Adults with diagnosed H. pylori | - Observational, narrative, basic research, meta-analyses, retrospective, case reports, conference presentations | - No significant difference in side effects between groups (RR = 1.47; 95%CI 0.83–2.58, p = 0.18) | |||||||||
| - Intervention: polyphenols (curcumin, cranberry, garlic, liquorice, broccoli) alone or with eradication therapy | - Antibiotic/PPI/H2-antagonist use in prior 2 weeks | - Cranberry subgroup eradication rate: 54.4% (vs. 45.4% in control); not statistically significant (RR = 1.24, 95%CI 0.94–1.64 p = 0.13) | |||||||||
| - Control: placebo or eradication therapy | - Polyphenols from non-cranberry sources (e.g., tea, coffee, wine) | ||||||||||
| - Reported eradication rates and/or side effects | |||||||||||
| - RCTs only | |||||||||||
| Delpino et al., (2022)85 | Effects of blueberry and cranberry on type 2 diabetes parameters in individuals with or without diabetes: a systematic review and meta-analysis of randomized clinical trials | - RCTs comparing blueberry or cranberry on T2DM parameters | - Observational studies | 22 (9 cranberry) | 993 (cranberry studies: 404) | Diabetic or non-diabetic adults and older adults | Blueberry or cranberry (capsule, juice, or other single-substance preparation) | Placebo usual care (non-pharmacological or pharmacological) | Fasting blood glucose, insulin resistance, glycated hemoglobin (HbA1c) | RCTs | - Cranberry/blueberry significantly reduced fasting blood glucose (MD = −17.72 mg dL−1,95% CI: −29.62, −5.82) and HbA1c (MD = −0.32%, 95% CI: −0.57, −0.07), but not insulin resistance |
| - Adult or older adult population (with or without diabetes) | - Non-human studies | - Analyses stratified comparing blueberry vs. cranberry showed a small significant effect on insulin resistance (MD = −0.24, 95%CI −0.48, −0.01) | |||||||||
| - Cranberry/blueberry used in any form | - Non-RCTs | ||||||||||
| - Control group included | - Grey literature | ||||||||||
| - Data reported before and after intervention | |||||||||||
| Nikbazm et al., (2022)86 | The effect of cranberry supplementation on Helicobacter pylori eradication in H. pylori positive subjects: a systematic review and meta-analysis of randomised controlled trials | - Parallel or crossover RCTs | - Duplicate data | 4 | 1935 | Adults with H. pylori | Cranberry | Placebo or non-placebo control | H. pylori eradication or suppression | RCTs | - Cranberry showed a non-significant increased odds of H. pylori eradication (OR = 1.27, 95% CI: 0.63–2.58) |
| - H. pylori-positive by accepted diagnostic tests | - Cohort, cross-sectional, case-control, review, and ecological studies | - Evidence inconclusive due to small number of studies and moderate heterogeneity | |||||||||
| - Intervention: cranberry juice/extract/powder vs. placebo or control | |||||||||||
| - Duration ≥1 week | |||||||||||
| - Outcome: H. pylori suppression/eradication | |||||||||||
| - Adult participants (>18 years) | |||||||||||
| - Multi-arm trials treated as separate studies | |||||||||||
| Pourmasoumi et al., (2020)17 | The effects of cranberry on cardiovascular metabolic risk factors: a systematic review and meta-analysis | - Randomized clinical trials (RCTs) only | - Duplicate data | 12 | 496 | Adults | Cranberry supplements | Any | CVD metabolic risk factors (lipid profile, glycemic status, BP, BMI, inflammatory markers) | RCTs | - Cranberry significantly reduced systolic blood pressure (SBP) and body mass index (BMI) |
| - Cranberry used as a supplement | - Cranberry combined with other supplements | - No significant changes in triacylglycerol, total cholesterol, LDL, HDL, fasting glucose, insulin, HOMA-IR, DBP, waist circumference, CRP, or ICAM | |||||||||
| - Adult participants (≥18 years) | - Treatment duration <2 weeks | - Greater SBP reduction in studies with mean age ≥50 | |||||||||
| - Reported CVD risk factors as outcomes | - Postprandial studies | ||||||||||
| - Unclear information and no author feedback | - Subgroup analysis: increased HDL in <50 mean age group; increased triacylglycerol with juice form cranberry | ||||||||||
| Panel B | |||||||||||
| Han et al., (2025)48 | Nonantibiotic prophylaxis for urinary tract infections: a network meta-analysis of randomized controlled trials | - History or risk factors for UTIs | - Cohort, case-control, animal studies | 50 (cranberry 18) | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 495 (cranberry 3262) 495 (cranberry 3262) |
Individuals with a history of UTIs or UTI risk factors | Non-antibiotic intervention | Any other | Incidence of UTIs | RCTs | - Cranberry significantly reduced UTI incidence vs. placebo (RR = 0.72, 95%CI 0.60–0.87) |
| - Investigated ≥1 non-antibiotic UTI prevention intervention | - Conference abstracts, editorials, letters | - Triple therapy (cranberry + probiotics + vitamin A) most effective (RR = 0.27, 95%CI 0.09–0.87) | |||||||||
| - Reported UTI incidence | - No usable outcomes | - Cranberry vs. placebo: 16 studies, 2990 patients (87% female), RR = 0.80 (95%CI 0.69–0.92, p = 0.002) | |||||||||
| - RCT design | - Sample size <10 per group | - Cranberry vs. antibiotics: 2 studies, 272 patients (100% female), RR = 1.38 (95%CI 0.82, 2.33, p = 0.220) | |||||||||
| - Antibiotics allowed as comparator | |||||||||||
| Xiong et al., (2024)87 | Preventive effect of cranberries with high dose of proanthocyanidins on urinary tract infections: a meta-analysis and systematic review | - RCTs analysing cranberry and UTIs | - Animal studies, case studies, systematic reviews | 10 | 2468 | Any population | Cranberry with known PAC dose | Placebo or other treatment | - Number of UTI patients in treatment vs. control groups at study end | RCTs | - When daily intake of PACs ≥36 mg/day: 18% UTI risk reduction (RR = 0.82, 95% CI = 0.69–0.98, p = 0.03) |
| - Compared cranberry-containing products to placebo or non-placebo control | - Trials with incomplete evidence | - No significant effect for PACs <36 mg day−1 | |||||||||
| - PAC content reported | - Subgroup analysis: significant UTI reduction with 12–24 weeks of cranberry use (RR = 0.75, 95% CI = 0.61–0.91, p = 0.004) | ||||||||||
| - Outcome: number of participants experiencing UTIs | - UTI risk reduction only in female-only subgroups (RR = 0.84, 95% CI = 0.71–0.98, p = 0.02) | ||||||||||
| Gkiourtzis et al., (2024)50 | Prophylaxis options in children with a history of recurrent urinary tract infections: a systematic review | - RCTs only | - Non-English language studies | 23 (8 with cranberry) | 3335 (871 randomised in a trial with cranberry) | Pediatric population | Antibiotics and dietary supplements as UTI prophylaxis | Other antibiotics or supplements | - Primary: incidence of symptomatic UTI in children with RUTI during prophylaxis | RCTs | - Cranberry and nitrofurantoin reduced symptomatic UTI odds vs. control and antibiotics |
| - Investigated antibiotics or dietary supplements as prophylaxis | - Included patients with congenital urogenital anomalies | - Secondary: febrile UTI episodes, kidney scarring, adverse events | - Nitrofurantoin may be the best option for UTI incidence | ||||||||
| - Pediatric patients with recurrent UTI (≥2 episodes) | - Cranberry OR = 0.41 (95% CI: 0.23–0.74, p < 0.01) | ||||||||||
| Moro et al., (2024)49 | Cranberry juice, cranberry tablets, or liquid therapies for urinary tract infection: a systematic review and network meta-analysis | - Individuals of any age/gender at risk of UTI | - Catheterised participants (unless data for non-catheterised participants reported separately) | 20 | 3091 | Individuals of any age and gender at risk for UTIs | Cranberry non-liquid (tablet, capsule, fruit) cranberry liquid (juice, cocktail, concentrate) | Placebo/no treatment/other cranberry formulation | - Primary: number of UTIs per group | RCTs and non-randomised controlled clinical trials | 18/20 studies highlighted cranberry juice reduced UTI rate by 54% vs. no treatment and 27% vs. placebo |
| - Compared at least two interventions, including cranberry (liquid/non-liquid), other liquids, or no treatment | - Secondary: UTI symptoms (e.g., urgency, dysuria, frequency, pain), antimicrobial use | - Antibiotic use reduced by 49% vs. placebo and 59% vs. no treatment | |||||||||
| - Symptom prevalence reduced | |||||||||||
| Subgroup analyses: | |||||||||||
| - Cranberry tablet vs. comparator: RR = 5.22 (95%CI 1.26–21.55; 3 studies, 85 patients) | |||||||||||
| - Cranberry juice vs. placebo: RR = 0.74 (95%CI 0.74 95%CI 0.60–0.91 p = 0.004; 14 RCTs, 2400 participants) | |||||||||||
| - Cranberry juice vs. no treatment: RR = 0.31 (95%CI 0.18–0.54, p < 0.000; 13 RCTs + 1 CCT, 234 participants) | |||||||||||
| Bolgarina et al., (2023)52 | Cranberry supplements for urinary tract infection prophylaxis in pregnant women: a systematic review of clinical trials and observational studies on efficacy, acceptability, outcomes measurement methods, and studies’ feasibility | - English-language primary studies | Non-primary studies | 3 | 1156 | Pregnant women | Cranberry supplements | Any comparator | - UTI prevention efficacy | RCTs and observational | - UTI reduction trend noted |
| - Pregnant women receiving cranberry supplements | - Tolerance: side effects, pregnancy/neonatal safety, compliance | - Cranberry juice caused GI intolerance in 23% (44/188) | |||||||||
| - Included data on efficacy, acceptability, outcome methods, and feasibility | - Feasibility: recruitment and retention rates | - Capsules reduced side effects (2%, 1/49) | |||||||||
| - Human studies only | - Methodology: study design and data collection | - Recruitment and retention issues: 33% and 59% dropout respectively | |||||||||
| Xia et al., (2021)55 | Consumption of cranberry as adjuvant therapy for urinary tract infections in susceptible populations: a systematic review and meta-analysis with trial sequential analysis | - RCTs only | - Cranberry combined with other bioactive compounds | 23 | 3979 | Various susceptible populations | Cranberry-containing products | Placebo or non-placebo control | - Number of participants experiencing a UTI | RCTs | - Cranberry significantly reduced UTI incidence in susceptible populations (RR = 0.70; 95% CI: 0.59–0.83) |
| - Compared cranberry products to placebo or control | - Did not meet inclusion criteria | - Relative risk reduction: 32% in women with recurrent UTIs(RR = 0.68; 95% CI: 0.56–0.81), 45% in children (RR = 0.55; 95% CI: 0.31–0.97), 51% in catheter users (RR = 0.49; 95% CI: 0.33–0.73) | |||||||||
| - Reported outcomes as number of UTI cases | - Animal studies, case reports, reviews, conference papers, editorials | - Juice form had greater effect than capsules/tablets (RR = 0.65; 95% CI: 0.54–0.77) | |||||||||
| - Populations included: recurrent UTIs, elderly, pregnant women, children, catheterized, neuropathic bladder | - Insufficient data | ||||||||||
| Meena et al., (2021)51 | Non-antibiotic interventions for prevention of urinary tract infections in children: a systematic review and meta-analysis of randomized controlled trials | - Children <18 years | - Included adults or kidney transplant patients | 16 (9 cranberry) | 1426 (769 cranberry studies) | Children (<18 years) | Any non-antibiotic interventions | Placebo or antibiotics | Recurrence of UTI | RCTs | - Cranberry was as effective as antibiotics prophylaxis (RR = 0.92; 95% CI: 0.56–1.50) but better than placebo/no therapy (RR = 0.48, 95% CI: 0.28–0.80) in reducing UTI recurrence |
| - Non-antibiotic interventions for UTI prophylaxis | - Did not report UTI recurrence as outcome | - In children with normal urinary tracts, cranberry significantly reduced UTI recurrence over 6–12 months when compared to placebo/no therapy (5 trials, 466 participants, RR: 0.48; 95% CI: 0.28–0.80) | |||||||||
| - Reported outcome: UTI recurrence | |||||||||||
| Gbinigie et al., (2020)88 | Cranberry extract for symptoms of acute, uncomplicated urinary tract infection: a systematic review | - RCTs (blinded/open-label) comparing cranberry extract vs. any treatment for acute uncomplicated UTIs | - Exclusively complicated UTI (e.g., catheterised, spinal cord injury, renal tract abnormalities, male UTIs, pyelonephritis) | 3 | 688 | Adults | Cranberry supplements | Any treatment or placebo | Participant symptoms, clinical cure, microbiological/biochemical outcomes, and wellbeing | RCTs | - One RCT (n = 309) found no effect of cranberry juice on UTI symptoms or antibiotic use vs. water (MD: −0.01, 95% CI: −0.37 to 0.34) |
| - Adults (≥18 years) | - Animal studies, case reports, systematic reviews | - One RCT (n = 319) found no added benefit from cranberry juice with immediate antibiotics for acute UTI | |||||||||
| - Cranberry used orally (juice, fruit, capsule, etc.) | - One RCT (n = 60) showed within-group symptom and E. coli load improvement with cranberry extract, not seen in controls | ||||||||||
| - If combined with other interventions, cranberry effect needed to be isolatable | |||||||||||
| - Reported at least one primary or secondary outcome (symptoms, antibiotic use, clinical cure, microbiological/biochemical outcomes, harms) | |||||||||||
| Raguzzini et al., (2020)54 | Cranberry for bacteriuria in individuals with spinal cord injury: a systematic review and meta-analysis | - Peer-reviewed studies | - Subjects with spina bifida or myelomeningocele | 6 | 477 | Patients with spinal cord injury | Cranberry supplements | Placebo or no treatment | Bacteriuria or UTI prevention | Observational and interventional studies | - No significant difference in bacteriuria prevention (RR = 0.85; 95% CI: 0.60–1.21) |
| - Published in English | - Low study quality; unable to recommend or exclude cranberry for individuals with spinal cord injury | ||||||||||
| - Focused on cranberry's effect on bacteriuria or UTI vs. control | |||||||||||
| - Included regardless of cranberry bioactive source/dose or study design | |||||||||||
| Fu et al., (2017)53 | Cranberry reduces the risk of urinary tract infection recurrence in otherwise healthy women: a systematic review and meta-analysis | - RCTs in healthy, nonpregnant women (≥18 years) with UTI history | - Not peer-reviewed full-text articles | 7 | 1498 | Generally healthy nonpregnant women ≥18 years with history of UTI | Cranberry supplements | Placebo or control | - Number of participants experiencing a UTI | RCTs | - Cranberry reduced UTI risk by 26% (RR = 0.74; 95% CI: 0.55–0.98) |
| - Compared cranberry to placebo or no treatment | - Special populations (elderly, children, pregnant women) | - Risk of bias in 2 studies due to high attrition or selective reporting | |||||||||
| - Reported outcome: number of UTI cases | - Institutionalized subjects | - Only 2 studies had >300 participants | |||||||||
| - Complicated UTIs (e.g., renal impairment, MS, dementia, spinal injury, abnormal urinary tract, neuropathic bladder, catheter, stones) | - Cranberry may help prevent UTI recurrence in healthy women; larger trials needed | ||||||||||
Regarding H. pylori, some reviews reported a trend toward improved eradication rates with cranberry supplementation, although statistical significance was not consistently reached. Overall, these findings suggest that standard cranberry formulations/products may provide limited but potentially clinically relevant effects in specific subgroups or outcomes. This warrants further investigation using standardized formulations and adequately powered trials.
While some systematic reviews reported modest benefits for certain outcomes, such as reductions in HOMA-IR, fasting blood glucose, or systolic blood pressure, results are not always consistent across different reviews. For some endpoints (e.g., LDL-C, TG), statistically significant positive effects have never been observed.
Overall, cranberries show: inhibited progression of degenerative unhealthy conditions and loss of functionality as neural degeneration; promotion of cardiovascular health; promotion of gastrointestinal health; oral benefits linked also to antiviral potentials; prevention and inhibition of cancer; modulation of inflammatory responses, antioxidant properties.2,12,24 However, further well-designed clinical studies are needed to clearly define the non-urological effects of cranberry.
Overall, the reviews consistently show a benefit across different populations, though the magnitude and certainty of effect vary. In adults with prior UTIs or predisposing risk factors, meta-analyses report significant risk reductions of UTI's rate with cranberry.48,49 Paediatric populations also appear to benefit: two reviews50,51 found cranberry to be as effective as antibiotics or superior to placebo for preventing recurrent UTIs. Both pregnant women52 and non-pregnant women53 have been studied, with positive results.
Finally, reviews in other high-risk groups, such as catheter users, patients with spinal cord injury,54 and including also those with neurogenic bladder,55 suggest subgroup-specific efficacy.
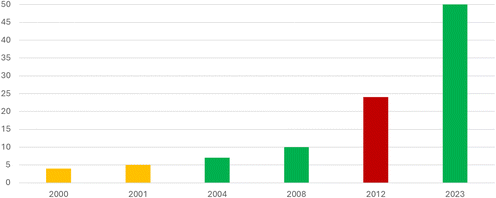
Very useful and interesting are the Cochrane meta-analyses, updated on the assessments of the published clinical experiences. The evolution of the Cochrane meta-analyses on cranberry products for UTIs prevention is particularly illustrative, given its striking “up–down–up” trajectory (Fig. 3).
The first Cochrane review in 200056 included four RCTs assessing outcomes such as symptomatic and asymptomatic UTIs, side effects, and adherence. Data from three trials suggested some benefit for at least one outcome, but the review concluded that the small number and poor quality of trials provided no reliable evidence. High dropout rates indicated poor long-term acceptability of cranberry juice, though capsules might be more acceptable; cranberry juice could not be recommended for prevention. The 2001 update,57 which added a fifth trial, reached the same conclusion.
In 2004,58 the review included seven trials, with two good-quality RCTs showing a significant reduction in UTI incidence at 12 months with cranberry products in women (RR 0.61, 95% CI 0.40–0.91). The authors concluded there was some evidence that cranberry juice may reduce symptomatic UTIs over 12 months in women, though its effect in children, older adults, and men was unclear; dropout rates again raised concerns about long-term use. By 2008,59 10 studies (n = 1049) were included, showing that cranberry products reduced UTI incidence at 12 months (RR 0.65, 95% CI 0.46–0.90) compared with placebo/control, with greater benefit in women with recurrent UTIs than in elderly adults or catheter users. Evidence for other groups remained uncertain, and dropout rates persisted.
The 2012 update changed this perspective.60 This version expanded to 24 studies (n = 4473) and found no significant reduction in symptomatic UTIs overall (RR 0.86, 95% CI 0.71–1.04) or in any subgroup, including women with recurrent UTIs, older adults, pregnant women, children, cancer patients, or people with neuropathic bladder/spinal injury. The addition of 14 new studies suggested cranberry juice was less effective than previously thought and given high dropout rates, mainly due to poor acceptability over long periods, and the small observed benefit, the authors concluded it could not be recommended.
By contrast, the 2023 update,61 which is the last available version, included 50 studies (n = 8857) and found moderate-certainty evidence that cranberry products reduced UTI risk overall (6211 participants: RR 0.70, 95% CI 0.58–0.84; I2 = 69%). Subgroup analyses suggested benefit in women with recurrent UTIs (8 studies, n = 1555: RR 0.74, 95% CI 0.55–0.99; I2 = 54%), children (5 studies, n = 504: RR 0.46, 95% CI 0.32–0.68; I2 = 21%), and people susceptible due to an intervention (6 studies, n = 1434: RR 0.47, 95% CI 0.37–0.61; I2 = 0%), but no benefit in elderly populations, those with bladder emptying problems, or pregnant women. The authors concluded cranberry products probably reduce the risk of symptomatic, cell culture-verified UTIs in specific groups. Cochrane has also addressed cranberries for UTIs. The 2000 review62 found no RCTs or quasi-RCTs meeting inclusion criteria, and the 2023 update63 reached the same conclusion, highlighting the persistent lack of high-quality, double-blind, placebo-controlled trials assessing cranberry products for this indication.
In summary, Cochrane's stance on cranberries for UTI prevention has evolved from no reliable evidence (2000–2001), to some benefit in women with recurrent UTIs (2004–2008), to no significant effect (2012), and back to probable benefit in select groups (2023), while for acute UTIs, robust RCT evidence remains entirely absent. These findings are in line with European Association of Urology 2025 Guidelines on Urological Infections, which report that a highly standardised cranberry extract formulated with phospholipids and high-dose proanthocyanidins appears effective in preventing cystitis recurrence episodes.64
Cranberry formulations
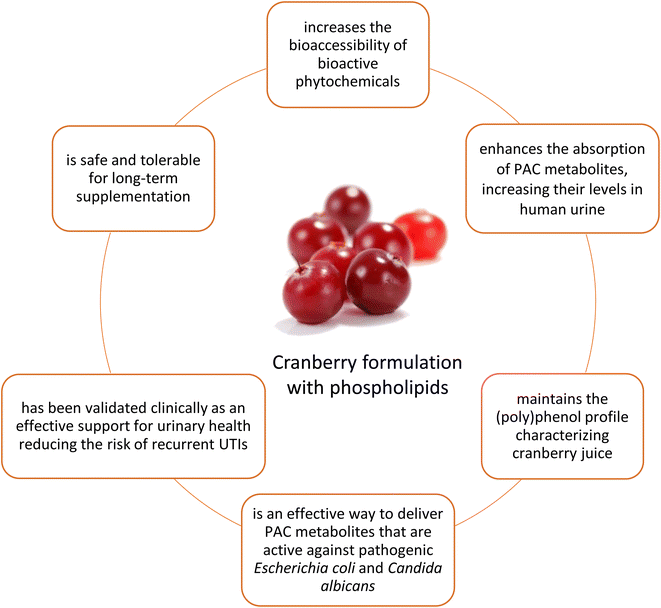
Several delivery systems are nowadays available. The most studied in literature were developed by the need to improve the bioavailability of active compounds related to cranberry activities. The most common are represented by: microencapsulation using Whey Protein Isolate (WPI) developed to improve bioavailability, liposomal/bile salt formulations where cranberry extract loaded into bile salt activated liposomes (BSALs) shows a sustained release pattern (61%–93% release); chitosan-based gels or carboxymethylcellulose formulations for a sustained and slower release of anthocyanins; phospholipids formulations.65–69 Cranberry extract was also microencapsulated in poly(lactide-co-glycolide) PLGA, to generate formulations able to inhibit salivary biofilm.70 More research has been performed on cranberry phospholipids (Anthocran™ Phytosome™) which is a rational formulation of cranberry extract based on lecithin using Phytosome™ technology, an innovative food-grade delivery system, which optimizes the solubility, the absorption and biological properties of nutraceuticals, such as cranberry extracts, by creating a solid dispersion of the botanical ingredient within a sunflower lecithin (phospholipid)-based matrix. This technology has been used for various bioactive molecules and botanical extracts, including quercetin, Berberis aristata extract, Coenzyme Q10, Palmitoylethanolamide (PEA), bergamot, Melissa officinalis, green tea from Camelia sinensis, Boswellia serrata extract and curcuminoids from Curcuma longa L.71–78Natural compounds, and botanical extracts in general, often have low solubility, poor stability, and complex chemical structures that limit their diffusion through intestinal membranes. Formulation with the phospholipids solid dispersion was reported to prevent self-aggregation of phytochemicals during gastrointestinal transit, to promote dispersion and absorption in the small intestine, to optimize their tolerability and interaction with biological membranes and the intestinal microbiota.
This could pose an interesting rational, as a better absorption could lead to similar results by administering a lower dosage, with consequent minimization of potential off-target adverse events.
Regarding specifically cranberry formulated in phospholipids, the rational of the formulation was mostly linked to the target organ, thanks to the delivery of most of the active metabolites in urine after cranberry intake. Cranberry phospholipids contained the full bouquet of (poly)phenols presented in natural cranberry juice, thanks to a rigorous supply chain from fruits harvest to manufacturing of the final health food ingredient.33 From PACs to anthocyanins and organic acids, cranberry in phospholipids mimics the composition of natural cranberry juice that was the original ingredient consumed in the traditional medicine of Native American for urinary health and later investigated by modern science and ethnopharmacology. This cranberry formulation is standardized to contain 6–9% of PACs as measured by spectrophotometry using the DMAC method. Its pharmacokinetic profile was studied in urines rather than human plasma, as urines are the target for Vaccinium macrocarpon ingredient benefits for urinary health (Baron et al., 202033). This in-depth investigation provided new insights into the absorption, metabolism, and excretion of cranberry compounds when formulated with phospholipids compared to the standard cranberry extract. In-depth, that investigation allowed detection of up to 42 compounds in human urines after supplementation with cranberry formulated with phospholipids, confirming that PACs are almost absent in the urine after cranberry intake; on the other hand, PACs metabolites were detected and identified.33 The study tested urine samples from individuals who consumed standardized cranberry extract formulated with phospholipids and compared them to those who took unformulated cranberry extract, focusing on antimicrobial activity against Candida albicans, which is key in determining infection outcomes. The analysis revealed that urine fractions richer in proanthocyanidins metabolites, derived from consumption of cranberry formulated with phospholipids, were the most effective in inhibiting Candida albicans adhesion and biofilm formation, attributing the health benefits to PACs metabolites rather than the parent PACs. This research contradicted the belief that PACs themselves are the main active compounds for health benefits, showing instead that intestinal microbiota converts PACs into beneficial and active metabolites with significant antimicrobial activity. Remarkably, the study showed that cranberry phospholipids, containing ∼9 mg of PACs in a 120 mg dose, demonstrated bioequivalence to 120 mg of unformulated cranberry extract (∼36 mg PACs) in terms of inhibiting Candida albicans adhesion. This highlights the equivalence efficacy of cranberry phospholipids formulation to cranberry extracts at 36% PACs, as it achieves the same effect with a significantly lower PACs content, thus proving its optimized bioavailability and effectiveness with high antimicrobial activity, firstly described in human urines. Moreover, cranberry formulated with phospholipids has been reported to allow to reach active concentrations of phenyl-γ-valerolactone in urine, the active PACs metabolites, with a lower variability in absorption and an improved organ target accessibility, when compared to unformulated cranberry extract.33 Furthermore, the concentrations of phenyl-γ-valerolactones in urine permitted the inhibition of the adhesion and the biofilm formation of Candida albicans and are in line with inhibitory concentrations of Escherichia coli.31,33 Human studies have demonstrated that cranberry formulated with phospholipids is effective in alleviating symptoms associated with urinary tract infections (UTIs), such as urinary frequency, urgency, dysuria, nocturia, ache, and haematuria, and it has been shown to reduce the recurrence of UTIs at dosages starting from 120 mg per day.79 A randomized, double-blind, placebo-controlled study in postmenopausal women taking SGLT2 inhibitors (antidiabetic drugs linked to increased UTI risk) further confirms the efficacy of cranberry formulated with phospholipids in reducing UTI recurrence, positioning it as a beneficial supplement for those at high risk for UTIs80 and included this as a valuable botanical support in the European Association of Urology guidelines on urological infections64 (2025). Furthermore, cranberry formulated with phospholipids has been extensively studied and proven to be safe for long-term use, without any serious side effects reported.33,79,80 This formulation also respects the natural balance of the intestinal microbiota, as demonstrated in ex vivo studies. It maintains the physiological transformation of (poly)phenols during microbial metabolism, supporting the production of biologically active colonic metabolites like phenyl-γ-valerolactones.81 Hence, cranberry formulated with phospholipids stands out for its dual advantage of supporting urinary health and maintaining intestinal health without disrupting the natural balance of gut microbiota. Taking into account all the above evidence, the main features of cranberry formulated with phospholipids are summarized in Fig. 4.
Conclusion
Urologists are increasingly recommending cranberry products like juice or supplements for the prevention of recurrent urinary tract infections (UTIs), particularly in women. Cranberry is a rich source of bioactive compounds that exert anti-adhesion, anti-biofilm, and antimicrobial effects against uropathogenic E. coli, supporting its traditional and evidence-based role in preventing UTIs. meta-Analyses indicate consistent, although variable, efficacy across diverse populations, including women, children, and high-risk groups, in preventing UTIs. Beyond urology, modest effects on glycaemic control, blood pressure, Helicobacter pylori eradication, neural protection, antiviral potentials, cancer prevention, have been reported, but evidence remains inconsistent and further studies are needed.Cranberry is generally well tolerated, with gastrointestinal discomfort the most common adverse event, though caution is warranted in patients on warfarin, tacrolimus, or with a history of oxalate stones. Cranberry extracts have shown to be effective against recurrent urinary tract infections and in addition, their lecithin-based formulation with an ameliorated urinary absorption, may present potential advantages compared to traditional non-formulated extracts. Of note, cranberry formulated with phospholipids is one of the few standardized cranberry ingredients with a rigorous characterization.33,81 Nonetheless, large, well-designed randomized controlled trials of cranberry preparations (including phospholipids-based formulation), are essential to clarify its full potential on health benefits.
Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.References
- L. Česonienė and R. Daubaras, Phytochemical Composition of the Large Cranberry (Vaccinium macrocarpon) and the Small Cranberry (V accinium oxycoccos), in Nutritional Composition of Fruit Cultivars, Simmonds MJS & Preedy V, 2016, pp. 173–194 Search PubMed.
- B. V. Nemzer, F. Al-Taher, A. Yashin, I. Revelsky and Y. Yashin, Cranberry: Chemical Composition, Antioxidant Activity and Impact on Human Health: Overview, Molecules, 2022, 27, 1503 CrossRef CAS PubMed.
- M. Majeed, K. Nagabhushanam and S. Arumugam, et al., Cranberry seed fibre: a promising prebiotic fibre and its fermentation by the probiotic Bacillus coagulans MTCC 5856, Int. J. Food Sci. Technol., 2018, 53, 1640–1647 CrossRef CAS.
- J. Oszmiański, J. Kolniak-Ostek, S. Lachowicz, J. Gorzelany and N. Matłok, Phytochemical Compounds and Antioxidant Activity in Different Cultivars of Cranberry (Vaccinium Macrocarpon L), J. Food Sci., 2017, 82, 2569–2575 CrossRef PubMed.
- J. Sun, J. P. Marais and C. Khoo, et al., Cranberry (Vaccinium macrocarpon) oligosaccharides decrease biofilm formation by uropathogenic Escherichia coli., J. Funct. Foods, 2015, 17, 235–242 CrossRef CAS PubMed.
- A. Karlsons, A. Osvalde, G. Čekstere and J. Pormale, Research on the mineral composition of cultivated and wild blueberries and cranberries., Agron. Res., 2018, 16(2), 454463 Search PubMed.
- N. Karim, A. K. Rashwan, S. Liu, J. Tangpong, T. Lin and W. Chen, An updated review on chemical compositions, biological capabilities, and clinical benefits of cranberries, Food Biosci., 2023, 54, 102877 CrossRef CAS.
- J. Oszmiański, S. Lachowicz, J. Gorzelany and N. Matłok, The effect of different maturity stages on phytochemical composition and antioxidant capacity of cranberry cultivars, Eur. Food Res. Technol., 2018, 244, 705–719 CrossRef.
- J. B. Blumberg, T. A. Camesano and A. Cassidy, et al., Cranberries and their bioactive constituents in human health, Adv. Nutr., 2013, 4, 618–632 CrossRef CAS PubMed.
- T. Jurikova, S. Skrovankova, J. Mlcek, S. Balla and L. Snopek, Bioactive Compounds, Antioxidant Activity, and Biological Effects of European Cranberry, Molecules, 2018, 24, 24 CrossRef PubMed.
- S. C. Debnath and D. An, Antioxidant properties and structured biodiversity in a diverse set of wild cranberry clones, Heliyon, 2019, 5, e01493 CrossRef CAS PubMed.
- E. Pappas and K. M. Schaich, Phytochemicals of cranberries and cranberry products: characterization, potential health effects, and processing stability, Crit. Rev. Food Sci. Nutr., 2009, 49, 741–781 CrossRef CAS PubMed.
- M. C. Denis, Y. Desjardins and A. Furtos, et al., Prevention of oxidative stress, inflammation and mitochondrial dysfunction in the intestine by different cranberry phenolic fractions, Clin. Sci., 2015, 128, 197–212 CrossRef CAS PubMed.
- J. Treml, P. Večeřová, P. Herczogová and K. Šmejkal, Direct and Indirect Antioxidant Effects of Selected Plant Phenolics in Cell-Based Assays, Molecules, 2021, 26, 2534 CrossRef CAS PubMed.
- Y. Huang, D. Nikolic, S. Pendland, B. J. Doyle, T. D. Locklear and G. B. Mahady, Effects of cranberry extracts and ursolic acid derivatives on P-fimbriated Escherichia coli, COX-2 activity, pro-inflammatory cytokine release and the NF-kappabeta transcriptional response in vitro, Pharm. Biol., 2009, 47, 18–25 CrossRef CAS PubMed.
- M. J. Kim, H. N. Jung, K. N. Kim and H. K. Kwak, Effects of cranberry powder on serum lipid profiles and biomarkers of oxidative stress in rats fed an atherogenic diet, Nutr. Res. Pract., 2008, 2, 158–164 CrossRef CAS PubMed.
- M. Pourmasoumi, A. Hadi, A. Najafgholizadeh, F. Joukar and F. Mansour-Ghanaei, The effects of cranberry on cardiovascular metabolic risk factors: A systematic review and meta-analysis, Clin. Nutr., 2020, 39, 774–788 CrossRef CAS PubMed.
- F. F. Anhê, D. Roy and G. Pilon, et al., A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. population in the gut microbiota of mice, Gut, 2015, 64, 872–883 CrossRef PubMed.
- F. F. Anhê, R. T. Nachbar and T. V. Varin, et al., A polyphenol-rich cranberry extract reverses insulin resistance and hepatic steatosis independently of body weight loss, Mol. Metab., 2017, 6, 1563–1573 CrossRef PubMed.
- K. Kowalska and A. Olejnik, Beneficial effects of cranberry in the prevention of obesity and related complications: Metabolic syndrome and diabetes – A review, J. Funct. Foods, 2016, 20, 171–181 CrossRef CAS.
- X. Cai, Y. Han and M. Gu, et al., Dietary cranberry suppressed colonic inflammation and alleviated gut microbiota dysbiosis in dextran sodium sulfate-treated mice, Food Funct., 2019, 10, 6331–6341 RSC.
- T. Gao, M. Hou and B. Zhang, et al., Effects of cranberry beverages on oxidative stress and gut microbiota in subjects with Helicobacter pylori infection: A randomized, double-blind, placebo-controlled trial, Food Funct., 2021, 12, 6878–6888 RSC.
- C. C. Neto, Cranberry and its phytochemicals: a review of in vitro anticancer studies, J. Nutr., 2007, 137, 186S–193S CrossRef CAS PubMed.
- R. Šedbarė, V. Janulis, A. Pavilonis and V. Petrikaite, Comparative In Vitro Study: Assessing Phytochemical, Antioxidant, Antimicrobial, and Anticancer Properties of, Pharmaceutics, 2024, 16, 735 CrossRef PubMed.
- L. Tao, D. Kumari and S. K. Badam, et al., Evaluation of Cranberry as a Novel Therapeutic Strategy for Intracerebroventricular (ICV) Quinolinic Acid-induced Cognitive Impairment in Rats, Comb. Chem. High Throughput Screen., 2026, 29, 1059–1072, DOI:10.2174/0113862073375293250520050009.
- Ł. Witucki, M. Kurpik and H. Jakubowski, et al., Neuroprotective Effects of Cranberry Juice Treatment in a Rat Model of Parkinson’s Disease, Nutrients, 2022, 14, 2014 CrossRef PubMed.
- L. M. Bystrom, D. P. Bezerra and H. T. Hsu, et al., Cranberry A-type proanthocyanidins selectively target acute myeloid leukemia cells, Blood Adv., 2019, 3, 3261–3265 CrossRef CAS PubMed.
- G. Ermel, S. Georgeault, C. Inisan and M. Besnard, Inhibition of adhesion of uropathogenic Escherichia coli bacteria to uroepithelial cells by extracts from cranberry, J. Med. Food, 2012, 15, 126–134 CrossRef CAS PubMed.
- S. Li, S. Zhao, L. M. Christman, T. L. Washington and L. Gu, Anti-adhesion capacities of selected cranberry polyphenols and metabolites against P-type and Type-1 fimbriated uropathogenic E. coli using a fluorometric method, Food Biosci., 2022, 49, 101960 CrossRef CAS.
- A. B. Howell, H. Botto and C. Combescure, et al., Dosage effect on uropathogenic Escherichia coli anti-adhesion activity in urine following consumption of cranberry powder standardized for proanthocyanidin content: a multicentric randomized double blind study, BMC Infect. Dis., 2010, 10, 94 CrossRef PubMed.
- P. Mena, D. G. de Llano and N. Brindani, et al., 5-(3′,4′-Dihydroxyphenyl)-γ-valerolactone and its sulphate conjugates, representative circulating metabolites of flavan-3-ols, exhibit anti-adhesive activity against uropathogenic Escherichia coli in bladder epithelial cells, J. Funct. Foods, 2017, 29, 275–280 CrossRef CAS.
- R. K. Ulrey, S. M. Barksdale, W. Zhou and M. L. van Hoek, Cranberry proanthocyanidins have anti-biofilm properties against Pseudomonas aeruginosa, BMC Complementary Altern. Med., 2014, 14, 499 CrossRef PubMed.
- G. Baron, A. Altomare and L. Regazzoni, et al., Profiling Vaccinium macrocarpon components and metabolites in human urine and the urine ex vivo effect on Candida albicans adhesion and biofilm-formation, Biochem. Pharmacol., 2020, 173, 113726 CrossRef CAS PubMed.
- M. C. Sánchez, H. Ribeiro-Vidal and B. Bartolomé, et al., New Evidences of Antibacterial Effects of Cranberry Against Periodontal Pathogens, Foods, 2020, 9, 246 CrossRef PubMed.
- J. S. Castellanos, D. E. Betancourt, D. Díaz-Báez and P. A. Baldión, Effect of flavonoids from grape seed and cranberry extracts on the microbiological activity of Streptococcus mutans: a systematic review of in vitro studies, BMC Oral Health, 2024, 24, 662 CrossRef CAS PubMed.
- A. Lacombe, C. McGivney, S. Tadepalli, X. Sun and V. C. Wu, The effect of American cranberry (Vaccinium macrocarpon) constituents on the growth inhibition, membrane integrity, and injury of Escherichia coli O157:H7 and Listeria monocytogenes in comparison to Lactobacillus rhamnosus, Food Microbiol., 2013, 34, 352–359 CrossRef CAS PubMed.
- P. Y. Lian, T. Maseko, M. Rhee and K. Ng, The antimicrobial effects of cranberry against Staphylococcus aureus, Food Sci. Technol. Int., 2012, 18, 179–186 CrossRef CAS PubMed.
- R. Singhal, P. Patil, M. Siddibhavi, A. V. Ankola, R. Sankeshwari and V. Kumar, Antimicrobial and Antibiofilm Effect of Cranberry Extract on, Int. J. Clin. Pediatr. Dent., 2020, 13, 11–15 CrossRef PubMed.
- H. D. Jensen, C. Struve, S. B. Christensen and K. A. Krogfelt, Cranberry Juice and Combinations of Its Organic Acids Are Effective against Experimental Urinary Tract Infection, Front. Microbiol., 2017, 8, 542 Search PubMed.
- E. Ottaviano, G. Baron and L. Fumagalli, et al., Candida albicans Biofilm Inhibition by Two Vaccinium macrocarpon (Cranberry) Urinary Metabolites: 5-(3′,4′-DihydroxyPhenyl)-γ-Valerolactone and 4-Hydroxybenzoic Acid, Microorganisms, 2021, 9, 1492 CrossRef CAS PubMed.
- M. Hakkola, P. Vehviläinen and J. Muotka, et al., Cranberry-lingonberry juice affects the gut and urinary microbiome in children - a randomized controlled trial, APMIS, 2023, 131, 112–124 CrossRef CAS PubMed.
- A. Al Othaim, D. Marasini and F. Carbonero, Impact of cranberry juice consumption on gut and vaginal microbiota in postmenopausal women, Food Front., 2021, 2, 282–293 CrossRef CAS.
- The European Medicines Agency (EMA), Vaccinii Macrocarpi Fructus – Herbal Medicinal Product. See https://www.ema.europa.eu/en/medicines/herbal/vaccinii-macrocarpi-fructus for further details.
- A. A. Dave and J. Samuel, Suspected Interaction of Cranberry Juice Extracts and Tacrolimus Serum Levels: A Case Report, Cureus, 2016, 8, e610 Search PubMed.
- G. L. Hamann, J. D. Campbell and C. M. George, Warfarin-cranberry juice interaction, Ann. Pharmacother., 2011, 45, e17 CrossRef PubMed.
- C. H. Paeng, M. Sprague and C. A. Jackevicius, Interaction between warfarin and cranberry juice, Clin. Ther., 2007, 29, 1730–1735 CrossRef PubMed.
- J. Zikria, R. Goldman and J. Ansell, Cranberry juice and warfarin: when bad publicity trumps science, Am. J. Med., 2010, 123, 384–392 CrossRef CAS PubMed.
- Z. Han, X. Yi, J. Li, D. Liao and J. Ai, Nonantibiotic prophylaxis for urinary tract infections: a network meta-analysis of randomized controlled trials, Infection, 2025, 53, 535–546 CrossRef CAS PubMed.
- C. Moro, C. Phelps and V. Veer, et al., Cranberry Juice, Cranberry Tablets, or Liquid Therapies for Urinary Tract Infection: A Systematic Review and Network Meta-analysis, Eur. Urol. Focus, 2024, 10, 947–957 CrossRef PubMed.
- N. Gkiourtzis, A. Stoimeni and A. Glava, et al., Prophylaxis Options in Children With a History of Recurrent Urinary Tract Infections: A Systematic Review, Pediatrics, 2024, 154, e2024066758 CrossRef PubMed.
- J. Meena, C. C. Thomas, J. Kumar, S. Raut and P. Hari, Non-antibiotic interventions for prevention of urinary tract infections in children: a systematic review and meta-analysis of randomized controlled trials, Eur. J. Pediatr., 2021, 180, 3535–3545 CrossRef PubMed.
- Z. Bolgarina, L. F. Gonzalez-Gonzalez, G. V. Rodroiguez and A. Camacho, Cranberry Supplements for Urinary Tract Infection Prophylaxis in Pregnant Women: A Systematic Review of Clinical Trials and Observational Studies on Efficacy, Acceptability, Outcomes Measurement Methods, and Studies’ Feasibility, Cureus, 2023, 15, e46738 Search PubMed.
- Z. Fu, D. Liska, D. Talan and M. Chung, Cranberry Reduces the Risk of Urinary Tract Infection Recurrence in Otherwise Healthy Women: A Systematic Review and Meta-Analysis, J. Nutr., 2017, 147, 2282–2288 CrossRef CAS PubMed.
- A. Raguzzini, E. Toti, T. Sciarra, A. L. Fedullo and I. Peluso, Cranberry for Bacteriuria in Individuals with Spinal Cord Injury: A Systematic Review and Meta-Analysis, Oxid. Med. Cell. Longevity, 2020, 9869851 CAS.
- J. Y. Xia, C. Yang, D. F. Xu, H. Xia, L. G. Yang and G. J. Sun, Consumption of cranberry as adjuvant therapy for urinary tract infections in susceptible populations: A systematic review and meta-analysis with trial sequential analysis, PLoS One, 2021, 16, e0256992 CrossRef CAS PubMed.
- R. G. Jepson, L. Mihaljevic and J. Craig, Cranberries for preventing urinary tract infections, Cochrane Database Syst. Rev., 2000, CD001321 CAS.
- R. G. Jepson, L. Mihaljevic and J. Craig, Cranberries for preventing urinary tract infections, Cochrane Database Syst. Rev., 2001, CD001321 CAS.
- R. G. Jepson, L. Mihaljevic and J. Craig, Cranberries for preventing urinary tract infections, Cochrane Database Syst. Rev., 2004, CD001321 Search PubMed.
- R. G. Jepson and J. C. Craig, Cranberries for preventing urinary tract infections, Cochrane Database Syst. Rev., 2008, CD001321 CAS.
- R. G. Jepson, G. Williams and J. C. Craig, Cranberries for preventing urinary tract infections, Cochrane Database Syst. Rev., 2012, 10, CD001321 Search PubMed.
- G. Williams, C. I. Stothart, D. Hahn, J. H. Stephens, J. C. Craig and E. M. Hodson, Cranberries for preventing urinary tract infections, Cochrane Database Syst. Rev., 2023, 11, CD001321 Search PubMed.
- R. G. Jepson, L. Mihaljevic and J. Craig, Cranberries for treating urinary tract infections, Cochrane Database Syst. Rev., 2000, 1998, CD001322 Search PubMed.
- R. G. Jepson, L. Mihaljevic and J. C. Craig, Cranberries for treating urinary tract infections, Cochrane Database Syst. Rev., 2023, 12, CD001322 Search PubMed.
- The European Association of Urology, EAU Guidelines on Urological Infections—Uroweb. See https://uroweb.org/guidelines/urological-infections for further details.
- N. Żurek, J. Kaliciak, M. Świeca and I. Kapusta, Microencapsulation of the polyphenolic fraction of cranberry fruit (Vaccinium oxycoccos) with coatings based on protein isolates: in vitro assessment of the bioavailability of polyphenolic compounds, Food Bioprod. Process., 2025, 154, 1–12 CrossRef.
- M. S. Soliman, S. Mosallam, M. A. Mamdouh, M. A. Hussein and M. S. Abd El-Halim, Design and optimization of cranberry extract loaded bile salt augmented liposomes for targeting of MCP-1/STAT3/VEGF signaling pathway in DMN-intoxicated liver in rats, Drug Delivery, 2022, 29, 427–439 CrossRef PubMed.
- R. Šedbarė, V. Janulis and K. Ramanauskiene, Formulation and Biopharmaceutical Evaluation of Capsules Containing Freeze-Dried Cranberry Fruit Powder, Plants, 2023, 12, 1397 CrossRef PubMed.
- J. Xu, C. Li and W. Wu, et al., Cranberry Research Progress: A Systematic Review of Chemical Composition, Pharmacological Mechanisms, Clinical Applications, and Nutritional Significance, Int. J. Mol. Sci., 2025, 26, 9707 CrossRef CAS PubMed.
- Y. Wang, P. B. Harrington and P. Chen, Analysis of Phenolic Compositions in Cranberry Dietary Supplements using UHPLC-HRMS, J. Food Compos. Anal., 2020, 86, 103362 CrossRef CAS PubMed.
- A. C. Greene, A. P. Acharya, S. B. Lee, R. Gottardi, E. Zaleski and S. R. Little, Cranberry extract-based formulations for preventing bacterial biofilms, Drug Delivery Transl. Res., 2021, 11, 1144–1155 CrossRef CAS PubMed.
- A. Riva, M. Ronchi, G. Petrangolini, S. Bosisio and P. Allegrini, Improved Oral Absorption of Quercetin from Quercetin Phytosome®, a New Delivery System Based on Food Grade Lecithin, Eur. J. Drug Metab. Pharmacokinet., 2019, 44, 169–177 CrossRef CAS PubMed.
- G. Petrangolini, F. Corti, M. Ronchi, L. Arnoldi, P. Allegrini and A. Riva, Development of an Innovative Berberine Food-Grade Formulation with an Ameliorated Absorption: In Vitro Evidence Confirmed by Healthy Human Volunteers Pharmacokinetic Study, J. Evidence-Based Complementary Altern. Med., 2021, 2021, 7563889 Search PubMed.
- G. Petrangolini, M. Ronchi, E. Frattini, E. De Combarieu, P. Allegrini and A. Riva, A New Food-grade Coenzyme Q10 Formulation Improves Bioavailability: Single and Repeated Pharmacokinetic Studies in Healthy Volunteers, Curr. Drug Delivery, 2019, 16, 759–767 CrossRef CAS PubMed.
- V. Mollace, M. Scicchitano and S. Paone, et al., Hypoglycemic and Hypolipemic Effects of a New Lecithin Formulation of Bergamot Polyphenolic Fraction: A Double Blind, Randomized, Placebo- Controlled Study, Endocr. Metab. Immune Disord. Drug Targets, 2019, 19, 136–143 CrossRef CAS PubMed.
- A. Bano, P. Hepsomali and F. Rabbani, et al., The possible “calming effect” of subchronic supplementation of a standardised phospholipid carrier-based, Front. Pharmacol., 2023, 14, 1250560 CrossRef CAS PubMed.
- P. Pietta, P. Simonetti, C. Gardana, A. Brusamolino, P. Morazzoni and E. Bombardelli, Relationship between rate and extent of catechin absorption and plasma antioxidant status, Biochem. Mol. Biol. Int., 1998, 46, 895–903 CAS.
- A. Riva, P. Morazzoni and C. Artaria, et al., A single-dose, randomized, cross-over, two-way, open-label study for comparing the absorption of boswellic acids and its lecithin formulation, Phytomedicine, 2016, 23, 1375–1382 CrossRef CAS PubMed.
- J. Cuomo, G. Appendino and A. S. Dern, et al., Comparative absorption of a standardized curcuminoid mixture and its lecithin formulation, J. Nat. Prod., 2011, 74, 664–669 CrossRef CAS PubMed.
- R. Cotellese, A. Ledda and G. Belcaro, et al., Anthocran® Phytosome®: Prevention of Recurring Urinary Infections and Symptoms after Catheterization, J. Diet. Suppl., 2023, 20, 55–67 CrossRef CAS PubMed.
- M. Rondanelli, F. Mansueto, C. Gasparri, S. B. Solerte, P. Misiano and S. Perna, Supplementation with Highly Standardized Cranberry Extract Phytosome Achieved the Modulation of Urinary Tract Infection Episodes in Diabetic Postmenopausal Women Taking SGLT-2 Inhibitors: A RCT Study, Nutrients, 2024, 16, 2113 CrossRef CAS PubMed.
- L. Bresciani, G. Di Pede and C. Favari, et al., In vitro (poly)phenol catabolism of unformulated- and phytosome-formulated cranberry (Vaccinium macrocarpon) extracts, Food Res. Int., 2021, 141, 110137 CrossRef CAS PubMed.
- X. Li, W. Chen, J. Xia and G. Sun, The Effects of Cranberry Consumption on Glycemic and Lipid Profiles in Humans: A Systematic Review and Meta-Analysis of Randomized Controlled Trials, Nutrients, 2024, 16, 782 CrossRef CAS PubMed.
- F. M. Delpino, F. S. Dos Santos, T. R. Flores, H. S. Cerqueira and H. O. Santos, The effects of blueberry and cranberry supplementation on blood pressure in patients with cardiovascular diseases: A systematic review and meta-analysis of randomized clinical trials, Phytother. Res., 2024, 38, 646–661 CrossRef PubMed.
- Q. Wang, C. Yao, Y. Li, L. Luo, F. Xie, Q. Xiong and P. Feng, Effect of polyphenol compounds on Helicobacter pylori eradication: a systematic review with meta-analysis, BMJ Open., 2023, 13, e062932 CrossRef PubMed.
- F. M. Delpino, L. M. Figueiredo, T. Gonçalves da Silva and T. R. Flores, Effects of blueberry and cranberry on type 2 diabetes parameters in individuals with or without diabetes: A systematic review and meta-analysis of randomized clinical trials, Nutr., Metab. Cardiovasc. Dis., 2022, 32, 1093–1109 CrossRef CAS PubMed.
- R. Nikbazm, Z. Rahimi, Y. Moradi, M. Alipour and F. Shidfar, The effect of cranberry supplementation on Helicobacter pylori eradication in H. pylori positive subjects: a systematic review and meta-analysis of randomised controlled trials, Br. J. Nutr., 2022, 128, 1090–1099 CrossRef CAS PubMed.
- Z. Xiong, Y. Gao, C. Yuan, Z. Jian and X. Wei, Preventive effect of cranberries with high dose of proanthocyanidins on urinary tract infections: a meta-analysis and systematic review, Front. Nutr., 2024, 11, 1422121 CrossRef PubMed.
- O. A. Gbinigie, E. A. Spencer, C. J. Heneghan, J. J. Lee and C. C. Butler, Cranberry Extract for Symptoms of Acute, Uncomplicated Urinary Tract Infection: A Systematic Review, Antibiotics (Basel), 2020, 10, 12 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |