Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Comment on “Plant-based diet and risk of arthritis: a nationwide cohort study of the Chinese elderly population” by H. Zhang, J. Liang, Y. Han, J. Tian, Y. Tu, R. Fan, W. Zhu, Z. Zhang and H. Zhao, Food Func., 2025, 16, 6576
Tong Xin ab,
Che Wangc,
Cuiting Gongab,
Jingjing Heab,
Xiaoping Wangab and
Aijing Liu*abd
ab,
Che Wangc,
Cuiting Gongab,
Jingjing Heab,
Xiaoping Wangab and
Aijing Liu*abd
aDepartment of Rheumatology and Immunology, The Second Hospital of Hebei Medical University, Shijiazhuang, 050000, Hebei Province, P.R. China. E-mail: ajliu@hebmu.edu.cn
bHebei International Joint Research Center on Rheumatic Diseases, Shijiazhuang, 050000, Hebei Province, P.R. China
cDepartment of Medical and Pharmaceutical Informatics, Hebei Medical University, Shijiazhuang, 050017, Hebei Province, P.R. China
dHebei Key Laboratory of Laboratory Medicine, Shijiazhuang, 050000, Hebei Province, P.R. China
First published on 24th February 2026
Abstract
A stratified perspective is essential to understand the divergent impacts of plant-based diets on osteoarthritis risk in older populations.
We read with great interest the nationwide cohort study by Zhang et al.,1 which reported that higher adherence to a plant-based diet was associated with a reduced risk of arthritis in older Chinese adults. The authors concluded that promoting plant-based dietary patterns may help delay arthritis onset and support healthy aging. While these findings are important, it should be noted that “arthritis” encompasses heterogeneous conditions, mainly including rheumatoid arthritis (RA) and osteoarthritis (OA).2 RA patients may benefit from the anti-inflammatory components of plant-based diets,3 but the effects in OA-specific populations remain unclear.
Using data from 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 125 Chinese adults aged ≥65 years without baseline arthritis from the Chinese Longitudinal Healthy Longevity Survey (CLHLS, 2008–2018), we excluded participants with self-reported or clinically suspected RA to focus on OA-specific risk (SI Fig. S1). Dietary intake was assessed using a simplified 16-item food frequency questionnaire, and plant-based diet indices (PDI)—overall PDI, healthful PDI (hPDI), and unhealthful PDI (uPDI)—were calculated according to established scoring methods. Associations between PDIs and incident OA were estimated using Poisson regression and restricted cubic splines, adjusting for age, sex, residence, education, lifestyle factors, body mass index, hypertension, and vitamin supplement use (SI Tables S1–S3). Participants with higher PDI scores were generally younger, more likely male, and had higher BMI and family income (SI Table S1). Other baseline characteristics followed similar trends across hPDI and uPDI (SI Tables S2 and S3).
125 Chinese adults aged ≥65 years without baseline arthritis from the Chinese Longitudinal Healthy Longevity Survey (CLHLS, 2008–2018), we excluded participants with self-reported or clinically suspected RA to focus on OA-specific risk (SI Fig. S1). Dietary intake was assessed using a simplified 16-item food frequency questionnaire, and plant-based diet indices (PDI)—overall PDI, healthful PDI (hPDI), and unhealthful PDI (uPDI)—were calculated according to established scoring methods. Associations between PDIs and incident OA were estimated using Poisson regression and restricted cubic splines, adjusting for age, sex, residence, education, lifestyle factors, body mass index, hypertension, and vitamin supplement use (SI Tables S1–S3). Participants with higher PDI scores were generally younger, more likely male, and had higher BMI and family income (SI Table S1). Other baseline characteristics followed similar trends across hPDI and uPDI (SI Tables S2 and S3).
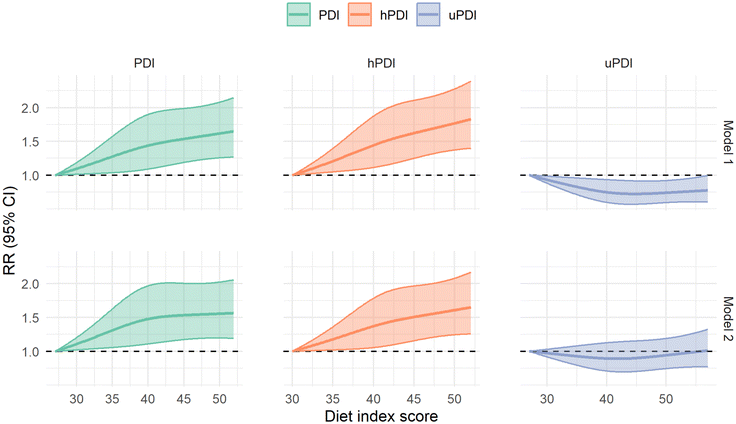
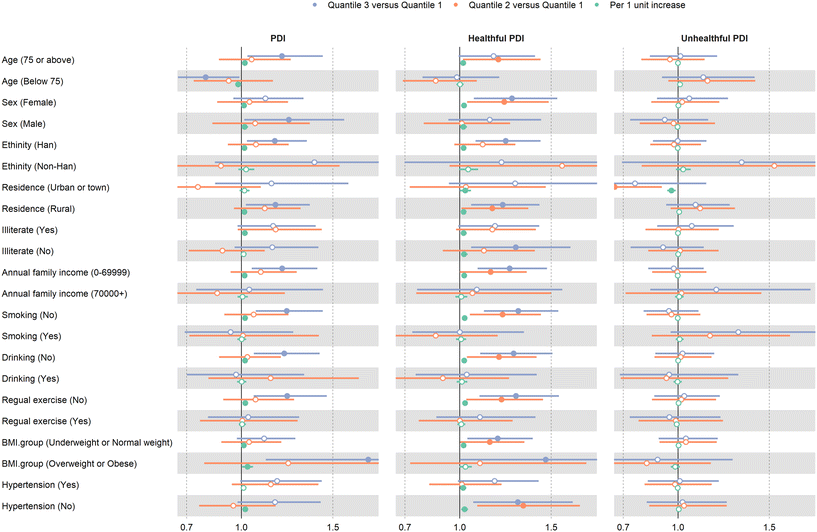
Over a median follow-up of 3.2 years, 1199 participants developed OA (incidence rate 9.9 per 1000 person-years). Higher PDI and hPDI were associated with increased OA risk. Compared with participants in the lowest tertile, the relative risk (RR) for OA was 1.19 (95% CI 1.04–1.36) for the highest tertile of overall PDI and 1.25 (95% CI 1.09–1.43) for the highest tertile of hPDI (Fig. 1). No significant association was observed for the uPDI. Dose–response analyses using restricted cubic splines indicated a near-linear increase in OA risk with higher PDI and hPDI scores (Fig. 2). Subgroup analyses revealed that the associations were more pronounced among adults aged ≥75 years, whereas other stratified factors showed generally consistent patterns (Fig. 3).
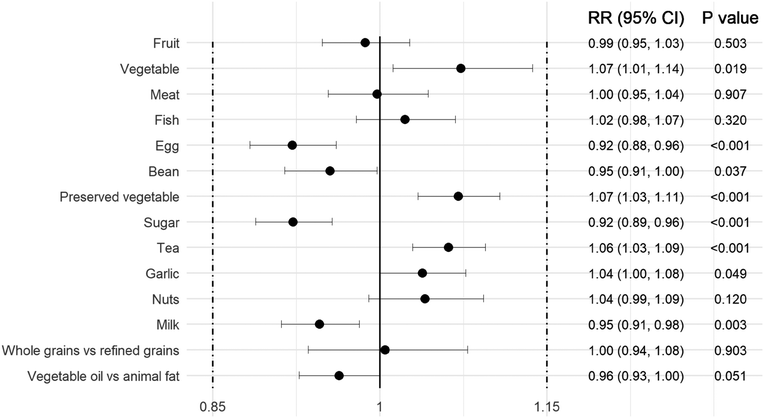
Food-item specific analyses revealed that higher intakes of vegetables, preserved vegetables, tea, and garlic were linked to increased OA risk, whereas frequent consumption of egg, milk, beans, and sugar was associated with lower risk (Fig. 4). These patterns suggest that the overall protective effect of plant-based diets observed in broad arthritis populations may not apply uniformly to OA-specific populations.
The observed heterogeneity in food-level associations suggests that the impact of plant-based diets on OA in the elderly is complex and likely driven by a combination of specific nutrient density, such as sodium and calcium content, co-occurring dietary habits, and potential residual confounding, rather than the plant source alone. Strict or predominantly plant-based diets in older adults have been associated with lower intakes or reduced bioavailability of calcium, high-quality protein, vitamin D, and vitamin B12,4 all of which are essential for cartilage matrix maintenance, subchondral bone integrity, and periarticular muscle support.5,6 This risk may be further exacerbated by age-related reductions in gastric acidity and digestive efficiency, as well as by antinutritional factors in plant foods, such as phytates and oxalates, that impair mineral absorption.7 Consistent with this hypothesis, our item-level analyses showed inverse associations between OA risk and consumption of eggs,8 milk,9 and beans,10 which provide bioavailable protein, calcium, vitamin D, magnesium, and anti-inflammatory bioactive compounds that support joint and bone health. In contrast, higher intakes of vegetables,11 preserved vegetables,12 and tea13 were associated with increased OA risk, which may be related to excessive sodium exposure, altered oxidative balance, or context-dependent pro-inflammatory effects of certain phytochemicals when consumed frequently in susceptible populations. The observed associations for sugar and garlic likely reflect dietary habits of Chinese older adults, with sugar intake (often used in cooking rather than as sugary beverages) serving as a proxy for dietary variety, and garlic intake (partly from preserved or processed garlic) unlikely to directly represent the biological effects of fresh garlic itself. Together, these findings suggest that the health effects of plant-based diets on OA are heterogeneous and are strongly influenced by specific food composition rather than by dietary pattern alone.
In this cohort, we identified 1199 incident OA cases and excluded 133 participants with a history of rheumatism. Although the excluded group was small, removing these potential inflammatory cases is critical. RA typically responds robustly to anti-inflammatory plant-based diets, with studies showing clinical improvements in as few as 7 days.14 In combined analyses, this strong and rapid protective signal can disproportionately skew the overall risk estimate, masking the specific associations observed in degenerative OA. Therefore, excluding rheumatism prevents ‘inflammatory bias’ from obscuring the true risk profile of the OA-dominant population.
Our findings highlight that the impact of plant-based diets may differ across arthritis subtypes. Contrary to its known anti-inflammatory benefits in RA, higher adherence to plant-based diets may not confer protective effects in OA, potentially due to nutrient deficiencies or biomechanical factors in elderly populations.15 While anti-inflammatory benefits may drive protective effects in RA, OA risk may be influenced by other factors, such as nutrient composition, joint load, and metabolic processes. Therefore, clinicians and policymakers should prioritize a disease-subtype specific approach. Rather than assuming uniform benefits, dietary guidance for OA prevention in older adults should explicitly emphasize optimizing overall dietary quality and nutrient density to balance musculoskeletal support with the management of specific dietary risks. Future research is warranted to explore the mechanistic basis of these differences and to refine dietary guidance for OA prevention.
There are several limitations that should be acknowledged when interpreting our findings. First, arthritis subtypes were based on self-reported information, and OA was approximated by excluding participants reporting RA. Some misclassification may remain, although OA accounts for the vast majority of arthritis cases in older Chinese adults, and any residual bias would likely attenuate the observed associations. Second, dietary intake was assessed only at baseline via a simplified food frequency questionnaire, precluding the analysis of changes over time and introducing potential measurement error. Third, although we adjusted for numerous sociodemographic, lifestyle, and clinical factors, residual confounding by unmeasured variables, such as medication use or comorbidities not recorded in the survey, cannot be excluded. Fourth, the study population consisted of older Chinese adults, which may limit the generalizability of our findings to younger populations or those from other regions. Finally, it is important to note that this study represents a post-hoc exploratory analysis of an existing dataset. Consequently, the observed associations should be interpreted with caution and considered hypothesis-generating, requiring further verification in prospective cohorts with pre-specified endpoints.
Abbreviations
| OA | Osteoarthritis |
| RA | Rheumatoid arthritis |
| PDI | Plant-based diet index |
| hPDI | Healthful plant-based diet index |
| uPDI | Unhealthful plant-based diet index |
| CLHLS | Chinese longitudinal healthy lifespan survey |
| RR | Relative risk |
| CI | Confidence interval |
| BMI | Body mass index |
Author contributions
Conceptualization: T. X., C. W., A. L. Data curation: T. X., C. G., C. W. Writing – original draft: T. X. Formal analysis: T. X., C. G., J. H. Software: J. H., X. W. Funding acquisition: A. L. Methodology: T. X., C. W., C. G., J. H., A. L. Validation: A. L., J. H., X. W. Visualization: T. X., C. W. Writing – review & editing: T. X., A. L. Supervision: A. L. All authors read and approved the final manuscript.Conflicts of interest
None of the authors has any potential conflict of interest.Data availability
The data used in this study are from the Chinese Longitudinal Healthy Longevity Survey (CLHLS), which are available from the Peking University Open Research Data Platform for qualified researchers. Access to the data requires an application and approval process. The dataset can be accessed via the following link: https://doi.org/10.18170/DVN/WBO7LK.Supplementary information (SI) is available, including the participant selection flow chart (Fig. S1) and baseline characteristics of the study population stratified by PDI, hPDI, and uPDI quantiles (Tables S1–S3). See DOI: https://doi.org/10.1039/d5fo05090a.
Acknowledgements
We express our gratitude to all members of the Chinese Longitudinal Healthy Lifespan Survey research team for their dedicated efforts. This work was supported by the Medical Science and Research Program of Hebei Province (NO.20240127), the Government Foundation of Excellent Clinical Medicine Talent Program of Hebei (NO.2025100), and the Opening Foundation of the State Key Laboratory of Neurology and Oncology Drug Development (No. SKLSIM-20250996).References
- H. Zhang, J. Liang, Y. Han, J. Tian, Y. Tu, R. Fan, W. Zhu, Z. Zhang and H. Zhao, Plant-based diet and risk of arthritis: a nationwide cohort study of the Chinese elderly population, Food Funct., 2025, 16, 6576–6585 RSC.
- A. L. Foster, M. A. Boring, T. D. Lites, J. E. Croft, E. L. Odom and E. A. Fallon, Distribution of Arthritis Subtypes Among Adults With Arthritis in the United States, 2017-March 2020, Prev. Chronic Dis., 2025, 22, E28 Search PubMed.
- J. Alwarith, H. Kahleova, E. Rembert, W. Yonas, S. Dort, M. Calcagno, N. Burgess, L. Crosby and N. D. Barnard, Nutrition Interventions in Rheumatoid Arthritis: The Potential Use of Plant-Based Diets. A Review, Front. Nutr., 2019, 6, 141 CrossRef PubMed.
- G. Kent, L. Kehoe, A. Flynn and J. Walton, Plant-based diets: a review of the definitions and nutritional role in the adult diet, Proc. Nutr. Soc., 2022, 81, 62–74 CrossRef CAS PubMed.
- R. M. Merrill and S. G. Aldana, Consequences of a plant-based diet with low dairy consumption on intake of bone-relevant nutrients, J. Womens Health, 2009, 18, 691–698 CrossRef PubMed.
- B. M. Tang, G. D. Eslick, C. Nowson, C. Smith and A. Bensoussan, Use of calcium or calcium in combination with vitamin D supplementation to prevent fractures and bone loss in people aged 50 years and older: a meta-analysis, Lancet, 2007, 370, 657–666 CrossRef CAS.
- A. Zayed, G. M. Adly and M. A. Farag, Management Strategies for the Anti-nutrient Oxalic Acid in Foods: A Comprehensive Overview of Its Dietary Sources, Roles, Metabolism, and Processing, Food Bioprocess Technol., 2025, 18, 4280–4300 CrossRef CAS.
- M. Rondanelli, M. A. Faliva, G. C. Barrile, A. Cavioni, F. Mansueto, G. Mazzola, L. Oberto, Z. Patelli, M. Pirola, A. Tartara, A. Riva, G. Petrangolini and G. Peroni, Nutrition, Physical Activity, and Dietary Supplementation to Prevent Bone Mineral Density Loss: A Food Pyramid, Nutrients, 2021, 14, 74 CrossRef PubMed.
- D. A. Savaiano and R. W. Hutkins, Yogurt, cultured fermented milk, and health: a systematic review, Nutr. Rev., 2021, 79, 599–614 CrossRef PubMed.
- Y. Y. Leung, A. Jin, K. B. Tan, L. W. Ang, J. M. Yuan and W. P. Koh, Food sources of dietary fibre and risk of total knee replacement related to severe osteoarthritis, the Singapore Chinese Health Study, RMD Open, 2021, 7, e001602 CrossRef.
- H. D. Nguyen, H. Oh and M. S. Kim, An increased intake of nutrients, fruits, and green vegetables was negatively related to the risk of arthritis and osteoarthritis development in the aging population, Nutr. Res., 2022, 99, 51–65 CrossRef CAS PubMed.
- P. Zhuang, F. Wu, X. Liu, F. Zhu, Y. Li, J. Jiao and Y. Zhang, Preserved vegetable consumption and its association with mortality among 440,415 people in the China Kadoorie Biobank, BMC Med., 2023, 21, 135 CrossRef PubMed.
- Z. Chen, M. B. Pettinger, C. Ritenbaugh, A. Z. LaCroix, J. Robbins, B. J. Caan, D. H. Barad and I. A. Hakim, Habitual tea consumption and risk of osteoporosis: a prospective study in the women's health initiative observational cohort, Am. J. Epidemiol., 2003, 158, 772–781 CrossRef CAS.
- A. M. Hartmann, M. Dell'Oro, M. Spoo, J. M. Fischer, N. Steckhan, M. Jeitler, T. Haupl, F. I. Kandil, A. Michalsen, D. A. Koppold-Liebscher and C. S. Kessler, To eat or not to eat-an exploratory randomized controlled trial on fasting and plant-based diet in rheumatoid arthritis (NutriFast-Study), Front. Nutr., 2022, 9, 1030380 CrossRef PubMed.
- Y. Zheng, J. Wang, Y. Wang, K. Xu and X. Chen, The Hidden Dangers of Plant-Based Diets Affecting Bone Health: A Cross-Sectional Study with U.S. National Health and Nutrition Examination Survey (NHANES) Data from 2005–2018, Nutrients, 2023, 15, 1794 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |