Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Effects of olive leaf polyphenols on blood lipid profiles and cardiovascular risk markers in healthy and at-risk populations: a narrative review
Sanne L. M.
van Stratum
 *ab,
Yala
Stevens
*ab,
Yala
Stevens
 ab and
Jogchum
Plat
ab and
Jogchum
Plat
 *a
*a
aDepartment of Nutrition and Movement Sciences, School for Nutrition and Translational Research in Metabolism (NUTRIM), Maastricht University, Maastricht, Netherlands. E-mail: sanne.vanstratum@maastrichtuniversity.nl; j.plat@maastrichtuniversity.nl
bSolabia BV, Gaetano Martinolaan 50, 6229 GS Maastricht, The Netherlands
First published on 16th March 2026
Abstract
Background: cardiovascular disease and metabolic syndrome are major contributors to global morbidity and mortality. Dyslipidaemia plays a central role in their pathogenesis. Olive-derived polyphenols, particularly oleuropein and hydroxytyrosol, have gained attention for their potential lipid-modulating and vascular-protective effects. Objective: This narrative review synthesizes clinical and mechanistic evidence on the effects of olive leaf and olive oil polyphenols on blood lipid profiles and cardiovascular risk markers in both healthy and at-risk populations. Methods: clinical studies using standardized olive leaf extracts (OLE) or olive oil preparations with defined phenolic content were included. Multi-ingredient formulations containing additional bioactive compounds were excluded to isolate the effects of olive-derived polyphenols. Evidence was categorized by population (healthy, cardiovascular risk, overweight/obese, hyperlipidaemic) and complemented by mechanistic insights from animal and cell-based studies. Results: in healthy individuals, olive-derived polyphenols mainly improved oxidative and vascular markers, with little effect on absolute lipid levels. In individuals with elevated cardiovascular risk, reductions in low-density lipoprotein cholesterol, total cholesterol, triglycerides, and oxidised low-density lipoprotein were more consistently observed. Overweight and obese participants showed no lipid changes at the studied doses, while postmenopausal and mildly hyperlipidaemic adults displayed modest short-term improvements and larger lipid reductions with longer supplementation. Mechanistic studies indicate that olive polyphenols influence lipid metabolism and vascular function through AMPK activation, suppression of SREBP-1c, modulation of PPAR pathways, enhancement of antioxidant defences via Nrf2 signalling, and attenuation of inflammatory pathways including NF-κB and MAPK. Conclusion: overall, current evidence indicates that olive leaf and olive oil polyphenols can beneficially modulate lipid parameters, oxidative stress, inflammation, and vascular function. The strongest and most consistent lipid-related effects are observed in individuals with elevated cardiovascular risk and in long-duration interventions in hyperlipidaemic postmenopausal women. While mechanistic studies support multiple pathways relevant to lipid regulation, long-term standardized clinical trials with well-characterized polyphenol compositions are needed to confirm efficacy and identify optimal dosing strategies across different metabolic phenotypes.
1. Introduction
Cardiovascular disease (CVD) and metabolic syndrome represent major global public health challenges. CVD remains the leading cause of mortality worldwide, with projections estimating 35.6 million annual deaths by 2050.1 Metabolic syndrome, often considered a precursor to CVD, is characterized by a cluster of risk factors, including hypertriglyceridemia and reduced high-density lipoprotein cholesterol (HDL-C) concentrations, that increase the risk of developing CVD by 2-fold.2–5 These conditions underscore the clinical relevance of dyslipidaemia, not only in disease management but also in prevention. Maintaining healthy blood lipid levels is therefore important even for individuals without a diagnosed disease.Lifestyle interventions, particularly dietary strategies, offer a practical approach to support regulation of lipid and lipoprotein metabolism and reduce cardiovascular risk across diverse populations.6 Among these, the Mediterranean diet stands out for its cardioprotective effects. Rich in plant-based foods, healthy fats, and moderate intake of animal products, this diet has been shown to improve blood lipid profiles, reduce inflammatory markers, and lower waist-to-hip ratios, ultimately decreasing the risk of coronary heart disease, ischemic stroke, and overall CVD.7–10
Key components of the Mediterranean diet include extra virgin olive oil and olives, which are rich in bioactive polyphenols known for their antioxidant and anti-inflammatory properties. Among these, oleuropein and hydroxytyrosol (HT) are two of the most studied polyphenols. While oleuropein is abundant in olive leaves,11 HT is typically found in higher concentrations in olive-derived byproducts such as pomace and vegetation water.12 In extra virgin olive oil, both compounds occur only at low levels; oleuropein typically ranges from <0.01–1.7 mg kg−1,13 while HT has been reported between 0–25.4 mg kg−1,11,14–16 due to hydrolysis and transformations occurring during olive processing.17 Interestingly, HT can also be obtained through the enzymatic or chemical hydrolysis of oleuropein,18 making olive leaves an indirect but valuable source. As a result, olive leaf extract (OLE) has gained attention not only for its native polyphenol content but also for its potential to serve as a precursor for HT production, with promising implications for cardiovascular and metabolic health.
OLE has been investigated across various health-related domains, including antioxidant activity,19–21 and neuroprotection.22 Cardiometabolic function has also been a focus of both clinical and mechanistic studies, which specifically examine OLE as a concentrated source of olive leaf polyphenols in relation to lipid metabolism, vascular function, and processes such as oxidative stress and inflammation. This research has also explored whether concentrated polyphenol sources might be relevant in populations with low habitual intake of olives or extra virgin olive oil. However, dietary improvement remains the primary strategy to increase polyphenol exposure. Importantly, maintaining healthy blood lipid levels is not only crucial for individuals with existing CVD or metabolic syndrome, but also for the general population as a preventive strategy to reduce long-term cardiovascular risk.
This narrative review aims to synthesize current knowledge on the role of olive-derived polyphenols, oleuropein and HT, particularly from OLE and extra virgin olive oil, in blood lipid regulation and vascular health. The review discusses clinical evidence supporting the lipid-modulating effects of interventions using olive polyphenols, followed by mechanistic insights from animal and cell-based studies. Relevant studies were identified through non-systematic searches in PubMed and Scopus using terms related to olive polyphenols, oleuropein, HT, OLE, lipid metabolism, and cardiovascular health. Studies using extracts with a standardized polyphenol, HT, or oleuropein content, or polyphenol enriched olive oil preparations were included. Interventions involving multi-ingredient formulations containing additional bioactive compounds (e.g., astaxanthin, berberine, curcumin) were excluded to avoid confounding effects and to isolate the impact of olive-derived polyphenols.
2. Olive leaf polyphenols: composition and characteristics
Given the growing interest in natural strategies for blood lipid management, OLE has attracted attention as a concentrated source of polyphenols with potential lipid-modulating effects. This section outlines essential background on olive leaf extract, including its traditional use, production, and the metabolic characteristics of its main bioactive constituents.2.1 Olea europaea: traditional use
Olea europaea, the olive tree, is native to the Mediterranean, parts of Asia, and the Middle East. Cultivated for millennia, it is valued for olives, olive oil, and olive leaves. While olives and olive oil are widely recognized for their nutritional value, particularly in the context of the Mediterranean diet, olive leaves have long been used in traditional medicine for their antipyretic, antimicrobial, and anti-inflammatory properties.23In recent years, scientific interest in olive leaves has increased due to their high content of bioactive polyphenols. These compounds, particularly oleuropein and HT, are being studied for their antioxidant, anti-inflammatory, and cardiometabolic properties. As a result, OLE is now commonly used in dietary supplements and functional foods aimed at supporting cardiovascular and metabolic health.
2.2 Production and standardization of OLE
After harvesting, olive leaves are dried to preserve phenolic compounds and subsequently extracted using either conventional methods, most commonly maceration in organic or aqueous-organic solvents such as methanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water (80
water (80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, v/v), or advanced techniques including ultrasound-assisted extraction (UAE) and microwave-assisted extraction (MAE). These techniques improve polyphenol yield in olive leaves, reduce processing time, and better preserve thermolabile compounds.24 Maceration of olive leaves in methanol
20, v/v), or advanced techniques including ultrasound-assisted extraction (UAE) and microwave-assisted extraction (MAE). These techniques improve polyphenol yield in olive leaves, reduce processing time, and better preserve thermolabile compounds.24 Maceration of olive leaves in methanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water yields very low oleuropein concentrations (<0.13 μg g−1–0.29 mg g−1),25 whereas MAE achieves 11.59 mg g−1 (ref. 26) and UAE reaches 13.39 mg g−1 (ref. 27) providing the highest oleuropein recovery among these methods. HT levels also differ across techniques; MAE of olive leaves yields ∼0.87 mg g−1, while UAE reaches ∼2.19 mg g−1, showing a 2-fold increase in HT concentration by using UAE.28 Lastly, total polyphenol content of olive leaves is also higher with advanced techniques; MAE and UAE deliver ∼79.8 mg g−1 compared to ∼71.1 mg g−1 for conventional heat-reflux extraction, while reducing extraction time from 60 min to only 21 min (UAE) or 2 min (MAE).28
water yields very low oleuropein concentrations (<0.13 μg g−1–0.29 mg g−1),25 whereas MAE achieves 11.59 mg g−1 (ref. 26) and UAE reaches 13.39 mg g−1 (ref. 27) providing the highest oleuropein recovery among these methods. HT levels also differ across techniques; MAE of olive leaves yields ∼0.87 mg g−1, while UAE reaches ∼2.19 mg g−1, showing a 2-fold increase in HT concentration by using UAE.28 Lastly, total polyphenol content of olive leaves is also higher with advanced techniques; MAE and UAE deliver ∼79.8 mg g−1 compared to ∼71.1 mg g−1 for conventional heat-reflux extraction, while reducing extraction time from 60 min to only 21 min (UAE) or 2 min (MAE).28
Although HT is commonly sourced from olive pomace and vegetation water,12 olive leaves are increasingly used as a sustainable alternative, with enzymatic or chemical hydrolysis of oleuropein enabling controlled HT production.18 This approach not only adds value to olive leaf biomass but also offers a controlled and standardized method for obtaining HT, making olive leaves a sustainable alternative source in the development of polyphenol-rich extracts. A broader overview of extraction techniques and their influence on phenolic yield is provided by comparative studies on olive leaf processing methods.24,29,30
Post-extraction steps include filtration, concentration, and spray-drying. To ensure consistency in composition and bioactivity, final products are often standardized for their HT or oleuropein content, which supports reproducibility in research and facilitates regulatory approval.
2.3 Bioactive compounds in OLE
OLE contains a wide range of bioactive compounds, including secoiridoids, flavonoids, simple phenols, triterpenoids, tocopherols, and pigments.31,32 Among these, several phenolic compounds have been identified, including luteolin 7-O-glucoside, apigenin 7-O-glucoside, rutin, tyrosol, and verbascoside.17 While many of these compounds occur in relatively low concentrations, oleuropein and HT are the most abundant and extensively studied. These two compounds are central to the biological activity of OLE and, therefore, form the primary focus of this review.2.4 Oleuropein: abundance and chemical properties
Although often referred to as a polyphenol, oleuropein is more accurately classified as a phenolic secoiridoid glycoside.33 It contributes to the bitter taste of olive leaves and is widely studied for its antioxidant, anti-inflammatory, antimicrobial, and lipid-lowering properties.Oleuropein is unique to the Oleaceae family, especially Olea europaea, and is found in unripe olives and their leaves and, to a lesser extent, in extra virgin olive oil. In general, olive leaves contain a broader and more concentrated phenolic profile than olive fruit or oil. For instance, oleuropein concentrations in olive leaves can exceed 20 mg g−1 extract, and in standardized OLEs, levels typically range between 30 and 57 mg g−1, depending on the olive cultivar and extraction method.34 In contrast, extra virgin olive oil contains only trace amounts of oleuropein (0 to 4.7 × 10−3 mg g−1),35,36 due to degradation during oil processing.34 This makes OLE a particularly rich and consistent source of polyphenols.
Upon hydrolysis, oleuropein yields several biologically relevant derivatives, including oleuropein aglycone, HT and elenolic acid (Fig. 1). The oleuropein-aglycone exhibits distinct biological properties, including reported neuroprotective37 and anti-atherogenic effects.38,39
 | ||
| Fig. 1 Oleuropein metabolism [reproduced from Frumuzachi et al., Antioxidants (Basel), 2024, under CC BY 4.0 license]. | ||
Although “glycosylated oleuropein” and “oleuropein-aglycone” are sometimes used interchangeably in the literature, they represent chemically and functionally different molecules. Glycosylated oleuropein is highly polar and the dominant form in all olive-leaf extracts and purified oleuropein powders used in preclinical and clinical research. In contrast, oleuropein-aglycone and its derivatives (oleacein, oleocanthal) arise predominantly during olive-oil processing through endogenous β-glucosidase activity and they are more lipophilic.40
It is important to note that many mechanistic in vitro studies reported in the broader olive-phenolic literature indeed use oleuropein-aglycone or aglycone-derived secoiridoids. However, in the present review, which focuses specifically on olive-leaf-derived oleuropein, all included in vitro and in vivo studies used glycosylated oleuropein (hereafter referred to as oleuropein). Only (extra) virgin olive oil-based interventions contain aglycone-rich secoiridoid fractions. This distinction is essential for correct mechanistic interpretation, since glycoside-derived oleuropein and oil-derived aglycones differ in polarity, metabolic fate, and biological activity.
2.5 Hydroxytyrosol: abundance and chemical properties
HT is a simple phenolic compound primarily formed through the hydrolysis of oleuropein during olive maturation, oil processing, and digestion (Fig. 1).17 While not exclusive to the Oleaceae family, HT is most abundantly found in olives and their by-products such as pomace and vegetation water.12 In OLE, HT is typically present in lower concentrations than oleuropein, although its levels can vary depending on the olive cultivar, harvest time, and extraction method. Typical HT concentrations in OLE lie around 2.5 to 6.9 mg g−1,41 which is substantially higher than in extra virgin olive oil, where HT concentrations generally range from 1.4 to 5.6 × 10−3 mg g−1.42HT is characterized by an ortho-dihydroxyphenyl group, which is thought to contribute to its notable antioxidant potential.43 It has attracted considerable scientific interest due to its stability and solubility.44
2.6 Metabolism and bioavailability of olive leaf polyphenols
Following ingestion of olive leaf polyphenols, oleuropein and HT undergo a tightly interconnected sequence of absorption, metabolic conversion, systemic distribution and excretion. Oleuropein is only minimally detected in its intact form in human plasma,45 as it is rapidly hydrolysed in the stomach and small intestine into HT and elenolic acid.46 The HT formed is absorbed efficiently in the small intestine. Reported plasma appearance times vary, typically within the first hour, but ranging from ∼15 min to 2 h depending on dose, matrix and analytical sensitivity.47 Furthermore, the fraction of oleuropein that reaches the colon is metabolized by the colonic microbiota, such as Lactobacillus and Bifidobacterium species,48 further increasing the pool of bioavailable HT.Once absorbed, HT undergoes extensive phase II metabolism to glucuronides, sulphates and methylated derivatives.45,49,50 These conjugates appear rapidly in circulation, generally within the first hour after intake,47 and reach peak concentrations shortly thereafter,45 reflecting highly efficient first-pass metabolism. Distribution studies in rodents show that HT and its metabolites reach the liver, red blood cells, kidneys, skeletal muscles, heart, and brain, indicating broad systemic distribution and the ability to cross the blood–brain barrier.51–54
In parallel with their rapid appearance in plasma, urinary excretion of HT-derived conjugates begins within the first few hours after ingestion and typically peaks later in the postprandial period (approximately 4–8 h), with most metabolites eliminated within 24 h.55–57 Human supplement and olive-oil interventions consistently show recovery of HT and its conjugates in urine, confirming efficient absorption, extensive phase II metabolism, and rapid renal clearance, with excretion levels strongly influenced by the food matrix.47,55–57 Polyphenol-enriched olive oils and liquid matrices consistently produce higher circulating metabolite concentrations than refined oils or capsules.47 Lastly, long-term supplementation also influences excretion profiles. After 21 days of supplementation with HT, oleuropein or oleuropein-aglycone extracts, rats excreted substantial quantities of phase I and II metabolites, including HT, HT-sulphate, and elenolic acid derivatives, within the first 24 h.54 The authors pointed out that long-term oleuropein supplementation resulted in higher total excretion of conjugated metabolites compared with chronic HT alone, reflecting the additional formation of HT from oleuropein hydrolysis.54 Collectively, these data demonstrate that the systemic fate of olive leaf phenols is driven less by the absorption of intact oleuropein and more by the rapid formation, distribution and clearance of HT-derived conjugates.
2.7 Safety and toxicological considerations
Toxicological data from animal and human studies suggest that OLE is generally well tolerated across a wide range of doses. A standardized aqueous olive pulp extract, containing 50–70% HT, demonstrated a no observed adverse effect level (NOAEL) of 2000 mg per kg per day in Wistar rats in a 90-day repeated-dose study, with no evidence of systemic toxicity or adverse findings in haematology, clinical chemistry or histopathology.58 Similarly, a standardized water-soluble OLE showed no genotoxicity and a NOAEL of 1000 mg per kg per day in a 90-day Wistar rat study.59 In addition, in a Wistar rat study ethanolic OLE showed no mortality or toxicity acutely after 2000 mg kg−1 and no adverse effects after 28-day exposure up to 400 mg per kg per day, with normal liver and kidney histology and stable biochemical markers.60In humans, OLE has also been well tolerated. Daily doses of 250–1000 mg per day of OLE containing approximately 50–200 mg oleuropein have been administered for periods ranging from several weeks to 12 months without clinically relevant adverse events.45,49,61–64 Overall, available evidence indicates a wide safety margin for oleuropein- and HT-containing preparations when used within commonly studied dose ranges.
2.8 Summary
Oleuropein and HT are the primary phenolic components of OLE, each contributing to the overall phenolic profile through their abundance and chemical structure. Although their individual characteristics differ, they are closely related; oleuropein serves as a key source of HT, and HT forms the basis for most phenolic metabolites detected after ingestion. The next section reviews the clinical evidence examining how these phenolic compounds, as part of OLE, influence blood lipid profiles and cardiovascular risk markers in humans.3. Clinical evidence: effects of oleuropein and hydroxytyrosol on blood lipids
Several clinical studies have explored the effects of olive leaf polyphenols on cardiometabolic health, particularly blood lipid profiles. This section reviews the clinical evidence across different populations, with most studies using olive extracts or oils containing both compounds. Where relevant, the dominant polyphenol is specified. To improve clarity, studies are grouped by cardiometabolic context; healthy populations, populations with elevated cardiovascular risk or established CVD, overweight and obese populations, and hyperlipidaemic populations. While overlap between groups is inevitable (e.g., hyperlipidaemia contributes to cardiovascular risk), studies were categorized based on their primary clinical context to enhance interpretability. To facilitate comparison across studies, all reported blood lipid concentrations were converted to mmol L−1 where necessary, using standard lipid conversion factors.653.1 Healthy populations
Maintaining a healthy lipid profile is not only relevant for individuals with diagnosed metabolic conditions, but also for those who are otherwise healthy. Even in the absence of clinical symptoms, subtle changes in blood lipid levels may contribute to increased cardiovascular risk over time.66 This section reviews clinical evidence on their preventive effects in individuals without overt dyslipidaemia or CVD risk. Table 1 provides an overview of study designs, populations, dosages, and outcomes.| Author, year | Study subjects | Study design | Intervention characteristics | Polyphenol content | Intervention duration | Significant effects |
|---|---|---|---|---|---|---|
| ↑: significant increase; ↓: significant decrease, ↔ no significant effect. Abbreviations: Ab-oxLDL = antibody against oxidised LDL; CAT = catalase; GPx = glutathione peroxidase; HDL-C = high-density lipoprotein cholesterol; LDL-C = low-density lipoprotein; cholesterol; MCP1 = monocyte chemoattractant protein-1; MDA = malondialdehyde; OLEU = oleuropein; oxLDL = oxidised low-density lipoprotein; RCT = randomized controlled trial; SOD/SOD1 = superoxide dismutase/superoxide dismutase 1 gene expression; TAC = total antioxidant capacity; TC = total cholesterol; TG = triglycerides; USF1 = upstream transcription factor 1; VAI = visceral adiposity index. | ||||||
Castañer et al. 2012![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 67 67 |
n = 18 healthy males, age = 20–60 years (mean ∼38 years) | RCT, crossover | 25 mL per day olive oil with low (2.7 mg kg−1) or high (366 mg kg−1) polyphenol content | Low: 0.06 mg per day, high: 7.7 mg per day | 3 weeks per intervention, 2-week washout | ↓ LDL-C: −0.16 mmol L−1, ↓ TC: −0.18 mmol L−1, ↓ oxLDL: −7.3 U L−1, ↓ MCP1: −29 pg mL−1, ↔ HDL-C, TG, glucose |
Covas et al., 2006![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 69 69 |
n = 12 healthy males, age = 20–22 years (mean ∼21 years) | RCT, crossover, 3-armed | 40 mL single dose olive oil with low; 2.7 mg kg−1, medium; 164 mg kg−1, or high; 366 mg kg−1 polyphenol content | Low: 0.09 mg per day medium: 6.0 mg per day high: 13.4 mg per day | Postprandial measurements up to 6 h; 10-day washout | ↓ oxLDL (postprandial): −22.5% vs. +23.2%, ↓ F2-isoprostanes: +11.6% vs. +29.9%, ↑ LDL phenolic content: +57%, ↑ Ab-oxLDL: +22.5% vs. −28.7%, ↔ TG, TC, HDL-C, LDL-C (postprandial) |
Marrugat et al., 2004![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 68 68 |
n = 30 healthy males (non-smoking in religious centre), age = 20–60 years (mean ∼57 years) | RCT, crossover, 3-armed | 25 mL per day of refined, common, or virgin olive oil | Refined: 0 mg per day, common: 1.6 mg per day, virgin: 3.4 mg per day | 3 weeks per intervention, 2-week washout | Virgin olive oil: ↑ HDL-C: +0.08 mmol L−1, ↑ LDL oxidation lag time: +8 min, ↓ oxLDL: −14.5 U L−1, ↔ TC, LDL-C, TG, glucose |
Oliveras-López et al., 2013![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 72 72 |
n = 62 healthy institutionalized elderly, age = 65–96 years (mean ∼82 years) | RCT, placebo-controlled | 50 mL per day polyphenol-rich extra virgin olive oil as only added fat vs. control (maintained dietary habits) | Total polyphenols: 26.79 mg per day, of which 0.09 mg HT | 6 weeks | ↓ TC: −0.40 mmol L−1, ↓ LDL-C: −0.22 mmol L−1, ↑ HDL-C: +0.18 mmol L−1, ↓ TG: −0.16 mmol L−1, ↑ TAC, ↑ CAT, ↓ SOD, ↓ GPx, ↔ glucose |
Perrone et al., 2019![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 73 73 |
n = 22 healthy adults, age = 18–65 years (mean ∼31 years) | RCT, crossover | 25 g polyphenol-rich extra virgin olive oil vs. common olive oil | 11.75 vs. 2.88 mg hydroxyphenyl ethanol and derivatives | Postprandial measurements up to 2 h; 1-week washout | ↓ oxLDL: −37.5%, ↓ MDA: −40.5%, ↓ TG: −11.1%, ↓ TC: −1.8%, ↓ VAI: −10.1%, ↑ SOD1 expression: fold change = 6.2, ↑ CAT expression: fold change = 2.2, ↑ USF1 expression: fold change = 2.4, ↔ HDL-C, LDL-C, glucose (postprandial) |
Pinckaers et al., 2025![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 71 71 |
n = 40 healthy males, age = 50–70 years (mean ∼60 years) | RCT, placebo-controlled | Bonolive®: 100 mg OLEU from 250 mg OLE daily vs. placebo (cellulose) | 100 mg per day OLEU | 36 days daily supplementation (∼5 weeks) | ↓ LDL-C: −0.2 mmol L−1, ↔ TC, HDL-C, TG, glucose, insulin, blood pressure |
Several clinical studies have evaluated the impact of olive leaf polyphenol supplementation on blood lipid profiles in healthy individuals. Despite heterogeneity in intervention type and dose, findings consistently show beneficial effects, particularly reductions in LDL oxidation and improvements in lipid parameters. In a crossover trial, Castañer et al. (2012) found that consuming high-polyphenol olive oil (7.7 mg per day) for three weeks reduced LDL oxidation (−7.3 ± 3.4 U L−1) and downregulated pro-atherogenic gene expression (CD40/CD40L pathway) compared to low-phenol oil (0.06 mg per day).67 Marrugat et al. (2004) demonstrated dose-dependent decreases in oxidised low-density lipoprotein (oxLDL) and increases in HDL-C concentrations following 3 weeks consumption of olive oil with 3.4 mg per day polyphenolic content.68 Covas et al. (2006) showed that olive oil consumption with 13.4 mg polyphenols led to postprandial (up to 6 h) decreases in oxLDL and increases in HDL-C concentrations.69 This indicates that even short-term intake of polyphenol-rich olive oil can confer measurable cardiometabolic benefits in healthy populations. These findings are consistent with the EFSA-approved health claim for HT, which states that it helps protect LDL particles from oxidative damage at intakes of ≥5 mg per day.70
In a more recent study, Pinckaers et al. (2025) investigated the effects of an OLE standardized for 100 mg oleuropein per day for 36 days in healthy older men.71 The intervention led to a statistically significant reduction in low-density lipoprotein cholesterol (LDL-C) concentrations (−0.3 mmol L−1), whereas no changes were observed in other lipid markers or metabolic outcomes. Oliveras-López et al. (2013) studied healthy elderly individuals who consumed polyphenol-rich extra virgin olive oil (containing 26.79 mg total polyphenols per day and 0.0865 mg HT per day) for six weeks.72 They reported significant improvements in total cholesterol (TC) (−0.36 mmol L−1), LDL-C (−0.28 mmol L−1), HDL-C (+0.17 mmol L−1), and triglyceride (TG) concentrations (−0.15 mmol L−1) compared to the control group, alongside enhanced antioxidant status (increase in catalase (CAT) and decrease in superoxide dismutase (SOD) and glutathione peroxidase (GPx)). These findings highlight the potential of olive leaf polyphenols to favourably modulate serum lipid profiles, even in the absence of dyslipidaemia.
Acute postprandial effects were observed by Perrone et al. (2019), who administered 25 g of polyphenol-rich extra virgin olive oil (containing 9.4 mg HT and derivatives) to 22 healthy volunteers.73 Within 2 h, reductions in plasma oxLDL (−1.05 mU L−1), malondialdehyde (MDA) (−0.17 μmol L−1), and TG (−0.08 mmol L−1) concentrations were observed, along with upregulation of antioxidant-related gene expression at the mRNA level (SOD1, CAT, and upstream transcription factor 1 (USF1)).
The clinical evidence reviewed in this section indicates that olive leaf- and olive oil-derived polyphenols may positively influence lipid and lipoprotein metabolism in healthy individuals, with observed effects including reductions in serum LDL-C and oxLDL concentrations, increases in HDL-C concentrations, and improvements in antioxidant status. However, the interpretation of these findings is complicated by the variability in polyphenol composition across studies. Most interventions used olive oils or extracts containing a mixture of compounds, often without standardization or quantification of individual polyphenols. As a result, the specific contributions of oleuropein and HT remain difficult to disentangle. Although several statistically significant changes were reported in ox-LDL and modest shifts in lipid parameters, the absolute lipid changes are small (typically <0.1–0.18 mmol L−1) and therefore unlikely to be clinically meaningful. It should be noted that most interventions were short-term (postprandial up to 6 weeks). At these exposure durations, the interventions are expected to predominantly exert antioxidative rather than lipid-lowering effects.
3.2 Populations with elevated cardiovascular risk or established CVD
Dysregulation of lipid metabolism plays a crucial role in increasing CVD risk and in the pathogenesis of coronary artery syndrome, stroke, and hypertension, making lipid-lowering strategies essential for prevention. This section reviews the limited but emerging evidence on the effects of olive leaf polyphenols in individuals with established CVD or increased CVD risk. Table 2 offers a summary of the clinical trials covered in this section, including study design, participant characteristics, dosage, and outcomes.| Author, year | Study subjects | Study design | Intervention characteristics | Polyphenol content | Intervention duration | Significant effects |
|---|---|---|---|---|---|---|
| ↑: significant increase; ↓: significant decrease, ↔ no significant effect.a Perrinjaquet-Moccetti et al. (2008)63 is listed twice to reflect the two separate sub-studies: one comparing 500 mg vs. control, and one comparing 1000 mg vs. 500 mg. Abbreviations: AUC = area under the curve, BP = blood pressure, CRP = C-reactive protein, DBP = diastolic blood pressure, HDL-C = high-density lipoprotein cholesterol, HT = hydroxytyrosol, IL-8 = interleukin-8, IRH = ischemic reactive hyperaemia, LDL-C = low-density lipoprotein cholesterol, MDA = malondialdehyde, OLE = olive leaf extract, OLEU = oleuropein, oxLDL = oxidised low-density lipoprotein, PAI-1 = plasminogen activator inhibitor-1, PCSK9 = proprotein convertase subtilisin/kexin type 9, RCT = randomized controlled trial, SBP = systolic blood pressure, TC = total cholesterol, TG = triglycerides. | ||||||
Ikonomidis et al., 2023![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 74 74 |
n = 30 patients with chronic coronary artery syndrome, age = 18–70 years (mean ∼62 years) | RCT, crossover, placebo controlled | Capsules with 412.5 mg olive oil enriched with 2.5 mg HT vs. placebo (non-enriched olive oil) | 10 mg per day HT | 1 month, washout period not described | ↓ TG: −0.47 mmol L−1, ↓ oxLDL: −18.9 ng mL−1, ↓ MDA: −0.4 mmol L−1, ↓ CRP: −2.7 mg L−1, ↓ PCSK9: −82.7 ng mL−1, ↔ TC, LDL-C, HDL-C, BP |
Lockyer et al., 2017![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 77 77 |
n = 60 pre-hypertensive males [SBP: 121–140 mmHg; DBP: 81–90 mmHg], age = 24–72 years (mean ∼45 years) | RCT, crossover, placebo controlled | Comvita: OLE ‘extra strength’ vs. polyphenol-free control | Total polyphenols: ∼167 mg per day of which 136 mg OLEU and 6.4 mg HT | 6 weeks, 4 weeks wash-out | ↓ 24 h SBP: −3.3 mmHg, ↓ 24 h DBP: −2.4 mmHg, ↓ TC: −0.32 mmol L−1, ↓ LDL-C: −0.19 mmol L−1, ↓ TG: −0.18 mmol L−1, ↓ IL-8, ↔ HDL-C, oxLDL, CRP |
Naranjo et al., 2024![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75 75 |
n = 8 patients with ischemic stroke, age >18 years (mean ∼69 years) | Exploratory pilot study | Mediteanox®: supplement standardized for 15 mg HT per day vs. placebo (not specified) | 15 mg per day HT | 45 days starting 24 h after stroke, ∼6 weeks | ↓ LDL-C: −1.08 mmol L−1, ↓ TC: −1.27 mmol L−1, ↓ SBP: −26 mmHg, ↓ DBP: −21.2 mmHg, ↔ TG and HDL-C |
Perrinjaquet-Moccetti et al., 2008![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63 63 |
n = 20 borderline hypertensive (SBP >120 or DBP >80 mmHg) monozygotic twins, age = 18–60 years (mean ∼37 years) | Open, controlled, parallel-group, co-twin study | EFLA® 943: OLE 500 mg vs. lifestyle advice | Total polyphenols: 200 mg per day, of which 104 mg OLEU | 8 weeks | ↓ SBP −5 mmHg, ↓ DBP −5 mmHg, ↓ LDL-C −0.5 mmol L−1, ↓ TC −0.9 mmol L−1, ↔ HDL-C |
Perrinjaquet-Moccetti et al., 2008![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63 63 |
n = 20 borderline hypertensive (>120 mmHg SBP or >80 mmHg DBP at rest) monozygotic twins, age = 18–60 years (mean ∼35 years) | Open, controlled, parallel-group, co-twin study | EFLA® 943: OLE 500 mg vs. 1000 mg | Total polyphenols: 400 mg per day of which 208 mg OLEU | 8 weeks | ↓ SBP −11 mmHg, ↓ DBP −4 mmHg; ↓ LDL-C −0.38 mmol L−1, ↓ TC −1.1 mmol L−1, ↔ HDL-C |
Susalit et al., 2011![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 64 64 |
n = 148 stage-1 hypertensive adults, age = 25–60 years (mean ∼50 years) | RCT, active-controlled, parallel | EFLA® 943: OLE 1000 mg vs. active control captopril 12.5–25 mg twice daily | Total polyphenols: 400 mg per day of which ∼200 mg OLEU | 8 weeks | ↓ SBP: −11.5 mmHg, ↓ DBP: −4.8 mmHg, ↓ TC: −0.15 mmol L−1, ↓ TG: −0.13 mmol L−1, ↓ LDL-C: −0.1 mmol L−1, ↔ HDL-C |
Valls et al., 2015![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 76 76 |
n = 13 pre- and stage-1 hypertensive adults, age = 20–75 years (mean ∼51 years) | RCT, crossover, postprandial | Functional (961 mg kg−1 polyphenols) vs. standard (289 mg kg−1) virgin olive oil | ∼29 mg polyphenols (in 30 mL functional virgin olive oil) of which ∼25 mg OLEU and ∼1 mg HT | Postprandial: single dose, 5 h follow-up | ↑ Endothelial function (↑ IRH AUC), ↓ oxLDL (−5.2 U L−1), ↓ PAI-1, ↓ hsCRP, ↔ TC, LDL-C, HDL-C |
Ikonomidis et al. (2023) found that in patients with CCAS, one month of HT-enriched olive oil supplementation (10 mg HT per day) significantly improved blood lipid parameters and other cardiovascular health markers.74 Compared to baseline, consumption of HT-enriched olive oil led to reductions in TG (−0.47 mmol L−1), oxLDL (−18.9 ng mL−1), proprotein convertase subtilisin/kexin type 9 (PCSK9) (−82.7 ng mL−1), and C-reactive protein concentrations (−2.7 mg L−1), with no such changes after placebo. Additionally, compared to baseline, there were significant improvements in vascular function, including increased flow-mediated dilation, improved pulse wave velocity, and enhanced coronary flow reserve, with no such changes after placebo. No significant effects were observed on blood pressure. These findings indicate that HT-enriched olive oil supplementation can improve blood lipid profiles and support cardiovascular health in patients with established coronary artery disease. However, it should be noted that the effects in the group consuming HT-enriched olive oil were not directly compared to those in the placebo group. Therefore, it is not clear whether these effects would remain significant when compared to placebo. Evidence from studies comparing the effects of the intervention to well-defined placebo conditions is needed to draw firm conclusions.
In a pilot clinical study by Naranjo et al. (2024), daily supplementation with an OLE containing 15 mg of HT per day for 45 days, initiated within 24 h post-ischemic stroke, led to a sustained reduction in diastolic blood pressure (−21.2 mmHg).75 TC and LDL-C also decreased substantially (−1.27 mmol L−1 and −1.08 mmol L−1, respectively), pointing to a favourable effect on lipid metabolism. Notably, the rise in nitric oxide, a marker of endothelial activation and oxidative stress, was attenuated in the HT group compared to controls (+3.3 µM vs. +5.4 µM). Proteomic analysis revealed downregulation of apolipoproteins ApoB100, ApoE, and ApoM, indicating potential anti-atherogenic effects. While these findings support a cardiovascular benefit of HT, the small sample size (n = 8) limits generalizability. Larger, controlled trials in stroke patients are needed to confirm these promising effects.
Several randomized controlled trials have explored the lipid-modulating potential of olive-derived polyphenols in hypertensive individuals. In a co-twin study, Perrinjaquet-Moccetti et al. (2008) investigated the effects of 500 and 1000 mg per day of OLE (standardized for 104 and 208 mg oleuropein per day, respectively) over eight weeks in borderline hypertensive monozygotic twins.63 The use of genetically identical twins helped control for interindividual variability, strengthening the evidence for a causal relationship between OLE supplementation and lipid improvements. At the higher dose, significant reductions in TC (−1.1 mmol L−1) and LDL-C (−0.6 mmol L−1) concentrations, alongside reductions in systolic (−11 mmHg) and diastolic (−4 mmHg) blood pressure were found. Interestingly, while the 500 mg dose did not significantly alter mean blood pressure levels, significant within-pair differences were observed, highlighting the sensitivity of the co-twin design to detect subtle effects. These findings underscore the potential of OLE as a complementary strategy for managing dyslipidaemia in hypertensive individuals, particularly when genetic and lifestyle factors are tightly controlled.
Susalit et al. (2011) confirmed these effects in a larger randomized controlled trial comparing 1000 mg per day OLE (standardized for ∼200 mg oleuropein) to an active control (12.5 mg captopril) in patients with stage-1 hypertension.64 After eight weeks, the OLE group showed significant reductions in TG (−0.13 mmol L−1) and TC concentrations (−0.15 mmol L−1) compared to baseline, with lipid-lowering effects most pronounced in participants with elevated baseline levels. Blood pressure reductions in the OLE group were comparable to those achieved with captopril treatment. Systolic blood pressure decreased by 13.8 mmHg with captopril and 11.5 mmHg with OLE, and diastolic blood pressure decreased by 6.5 mmHg and 4.7 mmHg, respectively. These reductions were not significantly different between groups, indicating that OLE was similarly effective to captopril in lowering blood pressure.
In a crossover trial, Valls et al. (2015) examined the acute effects of a single dose of 30 mL polyphenol-enriched virgin olive oil (standardized for ∼25 mg oleuropein and ∼1 mg HT) in pre- and stage-1 hypertensive individuals.76 Postprandial intake led to a 2.8% absolute increase in flow-mediated dilation, corresponding to a 76% relative improvement in endothelial function compared to baseline. In addition, a reduction in oxLDL (−4.9 U L−1) and TG (−0.10 mmol L−1) was found compared to baseline. These effects were not seen in the control group that consumed 30 mL non-enriched virgin olive oil, highlighting the vascular and lipid-modulating potential of polyphenol-rich olive oil formulations. However, it should be noted that the intervention group was not directly compared with the placebo group.
Lockyer et al. (2017) further supported these findings by demonstrating that six weeks of OLE supplementation (standardized for 136 mg oleuropein and 6 mg HT per day) significantly reduced serum TC (−0.32 mmol L−1), LDL-C (−0.19 mmol L−1), and TG (−0.18 mmol L−1) concentrations in prehypertensive men.77 Additionally, plasma IL-8 concentrations decreased (−0.63 pg mL−1), indicating an anti-inflammatory effect. These changes occurred alongside modest reductions in ambulatory blood pressure. No significant effects were observed on HDL-C or oxLDL concentrations.
The emerging clinical evidence supports the idea that olive leaf and olive oil polyphenols may offer lipid-lowering and vascular benefits in individuals with elevated cardiovascular risk, including those with existing coronary artery disease, ischemic stroke, and hypertension. Across studies, improvements in lipid profiles, particularly reductions in serum LDL-C, TG, and oxLDL concentrations, were frequently observed, alongside enhancements in vascular function and inflammatory markers.
Compared to the healthy population studies, the studies discussed in the current section typically used higher doses (HT between 1–15 mg, oleuropein between 25–208 mg) for mostly longer durations (4–8 weeks). Blood lipid improvements across studies range from 0.1 to 1.27 mmol L−1. Although changes at the lower end of this range may appear modest, in populations with existing cardiovascular risk, even small reductions can contribute to improved risk profiles. For example, a reduction of 1 mmol L−1 in LDL-C is associated with approximately a 22–25% lower risk of major CVD events, based on large-scale meta-analyses of statin and non-statin lipid-lowering therapies.78,79 This is reinforced by evidence from Susalit et al. (2011),64 showing that OLE not only equalled captopril in lowering blood pressure but also improved LDL-C and TG, an effect absent in the drug-treated group. Such data highlight the potential of olive leaf polyphenols to offer metabolic benefits in individuals with elevated cardiovascular risk using a high dose (400 mg total polyphenols of which ∼200 mg oleuropein) and long duration (8 weeks).
3.3 Overweight and obese populations
Overweight and obesity are closely linked to disturbances in lipid metabolism and chronic low-grade inflammation. Excess adipose tissue, particularly visceral fat, contributes to dyslipidaemia through increased free fatty acid flux and altered lipoprotein metabolism. Given these metabolic changes, interventions that improve lipid profiles in individuals with overweight or obesity are of particular interest. This section reviews clinical evidence on the effects of olive leaf polyphenols in this population. A summary of the clinical trials covered in this section is shown in Table 3.| Author, year | Study subjects | Study design | Intervention characteristics | Intervention polyphenol content | Intervention duration | Significant effects |
|---|---|---|---|---|---|---|
| ↑: significant increase; ↓: significant decrease, ↔ no significant effect. Abbreviations: BMI = body mass index; HDL-C = high-density lipoprotein cholesterol; HT = hydroxytyrosol; LDL-C = low-density lipoprotein cholesterol; OLE = olive leaf extract; OLEU = oleuropein; oxLDL = oxidised low-density lipoprotein; padj = adjusted p-value; RCT = randomized controlled trial; TC = total cholesterol; TG = triglycerides. | ||||||
de Bock et al., 2013![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49 49 |
n = 46 overweight males, age = 35–55 years (mean ∼46 years), BMI = ∼28 kg m−2 | RCT, crossover, placebo controlled | OLE capsules vs. placebo, both with safflower oil | Total polyphenols: 61.9 mg per day, OLEU: 51.1 mg per day, HT: 9.7 mg per day | 12 weeks, 6-week washout | ↑ Insulin sensitivity (+15%); ↑ β-cell function (+28%); ↔ TC, LDL-C, HDL-C, TG |
Stevens et al., 2021![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80 80 |
n = 77 overweight and obese adults with mildly elevated cholesterol (5–8 mmol L−1), age = 18–70 years (mean ∼56 years), BMI = ∼29 kg m−2 | RCT, placebo controlled | Bonolive® (250 mg OLE) + 100 mg maltodextrin vs. placebo (350 mg maltodextrin) | Standardized for 83.5 mg OLEU per day | 8 weeks | ↓ TG: −0.1 mmol L−1 after 4 weeks (p = 0.03), ↓ TG/HDL-C ratio −0.1 after 4 weeks (p = 0.02), but both not significant after correction for multiple testing (padj = 0.18 and padj = 0.16, respectively); ↔ TC, LDL-C, HDL-C, TG, oxLDL |
Two randomized controlled trials have examined the effects of OLE supplementation on blood lipid profiles in this group. In a double-blind crossover trial, de Bock et al. (2013) supplemented 46 overweight men with OLE (standardized for 51.1 mg oleuropein per day and 9.7 mg HT per day) for 12 weeks.49 While lipid profiles remained unchanged, OLE significantly improved insulin sensitivity (+15%) and pancreatic β-cell responsiveness (+28%), independent of diet, physical activity, or body composition. These effects were accompanied by increases in IGFBP-1 and IGFBP-2, markers of improved glucose regulation. The findings suggest that OLE may enhance glycaemic control in insulin-resistant individuals, even in the absence of lipid-lowering effects.
In contrast, Stevens et al. (2021) evaluated 8-week OLE supplementation (standardized for 83.5 mg oleuropein per day) in 77 overweight and obese adults with mildly elevated cholesterol.80 No significant changes were observed in lipid profiles, oxLDL, blood pressure, glucose, insulin, or liver function. A transient improvement in TG concentrations and the TG-to-HDL-C ratio at 4 weeks was observed, but this did not remain significant after correction for multiple testing. This ratio is considered a more sensitive marker of CV risk in overweight and insulin-resistant populations as it reflects atherogenic dyslipidaemia and correlates more strongly with insulin resistance and cardiovascular events than LDL-C alone.81,82
Together, these studies suggest that while OLE may not improve lipid parameters in overweight individuals, it seems that insulin sensitivity and β-cell function can be enhanced in those with early metabolic impairment. The absence of lipid-lowering effects may reflect differences in intervention duration, polyphenol composition, or metabolic responsiveness. Notably, improvements in lipid parameters have been observed in healthy individuals and those with elevated cardiovascular risk or established CVD, but not in the studied overweight or obese populations. One possible explanation is the relatively low polyphenol dose used in these studies (standardized for 51.1 and 83.5 mg oleuropein per day), which is substantially lower than the 100–200 mg per day of oleuropein used in other trials reporting lipid improvements (Perrinjaquet-Moccetti et al. (2008);63 Lockyer et al. (2017);77 Pinckaers et al. (2025)71). Therefore, at the doses and durations tested, clinically meaningful lipid lowering is unlikely in overweight or obese adults. Factors such as bioavailability, microbiome composition, and metabolic phenotype may also influence individual responses to OLE. These findings raise important questions about the metabolic context and intervention conditions under which olive leaf polyphenols are most effective. Future studies in overweight and metabolically impaired populations should consider using higher doses of polyphenols (∼100–200 mg per day of oleuropein and >10 mg HT), comparable to those used in trials that reported lipid improvements, to better assess therapeutic potential in this specific group.
3.4 Hyperlipidaemic populations
Hyperlipidaemia is a key risk factor for CVD and is defined by elevated serum cholesterol and/or TG concentrations. Certain populations, such as postmenopausal women, are particularly susceptible to lipid imbalances due to hormonal changes that affect lipid metabolism. This section reviews the evidence on the lipid-modulating effects of olive leaf polyphenols in individuals with hyperlipidaemia and in postmenopausal women with elevated lipid levels. A detailed overview of the clinical trials can be found in Table 4.| Author, year | Study subjects | Study design | Intervention characteristics | Polyphenol content | Intervention duration | Significant effects |
|---|---|---|---|---|---|---|
| ↑: significant increase; ↓: significant decrease, ↔ no significant effect. Abbreviations: HDL-C = high-density lipoprotein cholesterol, HT = hydroxytyrosol, LDL-C = low-density lipoprotein cholesterol, OLE = olive leaf extract, OLEU = oleuropein, RCT = randomized controlled trial, TC = total cholesterol, TG = triglycerides, TG/HDL-C = triglyceride to HDL-cholesterol ratio. | ||||||
Filip et al., 2015![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 62 62 |
n = 64 postmenopausal women with osteopenia, age = 49–68 years (mean ∼60 years), baseline TC: ∼6.1–6.4 mmol L−1 | RCT, placebo-controlled, parallel | Bonolive®: OLE 250 mg per day + 1000 mg Ca vs. placebo (1000 mg Calcium) | 100 mg per day OLEU | 12 months | ↓ TC: −0.7 mmol L−1, ↓ LDL-C: −0.9 mmol L−1, ↔ TG and HDL-C |
Imperatrice et al., 2024![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 87 87 |
n = 60 postmenopausal women, age 47–70 years (mean ∼59 years), baseline TC: ∼5.7–6.0 mmol L−1 | RCT, placebo-controlled, parallel | Bonolive®: OLE 250 mg per day vs. placebo (cellulose) | 100 mg per day OLEU | 12 weeks | ↓ TG: −0.1 mmol L−1, ↓ TG/HDL-C ratio: −0.1, ↔ TC, LDL-C, HDL-C |
Lopez-Huertas & Fonolla, 2017![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 83 83 |
n = 14 mild hyperlipidaemic adults, age = 25–42 years (mean ∼34 years), baseline TC: ∼5.2–6.2 mmol L−1 | Single-arm, open-label, before-after | Pure HT 45 mg per day in saline solution | 45 mg per day HT | 8 weeks | ↑ Vitamin C: +24.4 µmol L−1, ↔ TC, LDL-C, HDL-C, TG |
Lopez-Huertas et al. (2017) administered a purified HT supplement (45 mg per day) to mildly hyperlipidaemic volunteers for 8 weeks.83 Despite the relatively high dose, no significant changes were observed in serum TC, LDL-C, HDL-C, or TG concentrations compared to baseline. Interestingly, the intervention led to a two-fold increase in circulating vitamin C levels, indicating a potential antioxidant-sparing effect. These findings indicate that HT alone, at this dose and in this population, may not exert lipid-lowering effects, though it may enhance systemic antioxidant capacity. In addition, it should be noted that this study did not include a control group; therefore, only changes compared to baseline could be investigated.
Although post menopause is a natural physiological stage rather than a disorder, hormonal changes during this period are associated with adverse shifts in lipid metabolism.84 Menopause induces a shift in lipid metabolism, typically resulting in higher serum TC, LDL-C, and TG concentrations, alongside reduced HDL-C concentrations.84 These changes are primarily driven by declining oestrogen levels.84 Oestradiol, the main biologically active oestrogen, is synthesized in the ovaries using LDL-C as a precursor.85 After menopause, this pathway becomes inactive, leading to reduced oestrogen synthesis and a corresponding rise in circulating LDL-C concentrations.86 This mechanism contributes, amongst others, to the increased cardiovascular risk observed in postmenopausal women. As such, postmenopausal women represent a population at increased risk for lipid disorders and cardiovascular disease.
Two randomized controlled trials have investigated the effects of olive leaf polyphenols on lipid metabolism in postmenopausal women. In a 12-week study by Imperatrice et al. (2024), 60 healthy postmenopausal women received 250 mg OLE per day (standardized for 100 mg oleuropein).87 At baseline, these women had mild hyperlipidaemia (average TC between 5.13–6.91 mmol L−1). The intervention led to a significant reduction in TG (−0.1 mmol L−1) and the TG/HDL-C ratio (−0.1) compared to placebo, although no changes were observed in other lipid parameters.
In a longer trial by Filip et al. (2015), postmenopausal women with osteopenia were supplemented with 250 mg OLE per day (standardized for 100 mg oleuropein) for 12 months, alongside 1000 mg of calcium.62 At baseline, these women had mild-to-moderate hyperlipidaemia (average TC between 5.53–7.35 mmol L−1). This study reported significant reductions in TC (−0.68 mmol L−1) and LDL-C (0.90 mmol L−1) concentrations, in addition to improvements in bone markers such as osteocalcin (+32%). TG and HDL concentrations did not change.
Taken together, the available evidence suggests that olive leaf polyphenols exert modest lipid-lowering effects in individuals with (mild) hyperlipidaemia, particularly in postmenopausal women, with a clear distinction between short- and long-term responses. From a clinical perspective, these three trials suggest that the lipid-modulating potential of olive leaf polyphenols in hyperlipidaemic populations is strongly dependent on both formulation and intervention duration. The 8-week study using purified HT alone (Lopez-Huertas & Fonolla, 2017![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 83) did not produce lipid improvements, although the two-fold increase in circulating vitamin C suggests that systemic antioxidant shifts may precede measurable changes in lipid parameters. In contrast, both OLE interventions (Imperatrice et al., 2024;87 Filip et al., 2015
83) did not produce lipid improvements, although the two-fold increase in circulating vitamin C suggests that systemic antioxidant shifts may precede measurable changes in lipid parameters. In contrast, both OLE interventions (Imperatrice et al., 2024;87 Filip et al., 2015![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 62), containing a broader polyphenolic matrix, showed progressively stronger effects with increasing duration; selective reductions in TG and TG/HDL-C ratio after 12 weeks, and clinically meaningful decreases in TC (−0.68 mmol L−1) and LDL-C (−0.90 mmol L−1) after 12 months. These LDL-C reductions fall within a range associated with modest cardiovascular risk reduction, even in mildly hyperlipidaemic individuals. Overall, while promising, the current evidence base remains limited in scale and is heterogeneous, underscoring the need for larger, well-controlled trials to confirm efficacy and clarify optimal dose, formulation, and intervention length.
62), containing a broader polyphenolic matrix, showed progressively stronger effects with increasing duration; selective reductions in TG and TG/HDL-C ratio after 12 weeks, and clinically meaningful decreases in TC (−0.68 mmol L−1) and LDL-C (−0.90 mmol L−1) after 12 months. These LDL-C reductions fall within a range associated with modest cardiovascular risk reduction, even in mildly hyperlipidaemic individuals. Overall, while promising, the current evidence base remains limited in scale and is heterogeneous, underscoring the need for larger, well-controlled trials to confirm efficacy and clarify optimal dose, formulation, and intervention length.
3.5 Summary
Across populations, the lipid-modulating effects of olive leaf and olive oil polyphenols vary depending on baseline metabolic status, polyphenol composition, and intervention duration. In healthy individuals, lipid changes are small (typically <0.2 mmol L−1) and largely attributable to short-term antioxidative effects rather than clinically meaningful lipid lowering. In contrast, individuals with elevated cardiovascular risk, including those with hypertension, chronic coronary artery syndrome, or recent ischemic stroke, display the most consistent and clinically relevant responses, particularly at doses standardized to ≥100 mg per day oleuropein or ≥10–15 mg per day HT administered for 4–8 weeks. In this group, reductions in LDL-C, TG, and oxLDL of up to ∼1 mmol L−1 have been reported, with the strongest evidence coming from higher-dose OLE interventions. Overweight and obese individuals appear least responsive at the doses tested (50–85 mg oleuropein per day), showing improvements in insulin sensitivity rather than lipid profiles, indicating that higher doses may be required to elicit lipid changes in this metabolic context. However, it should be noted that limited evidence is available for this population. In hyperlipidaemic and postmenopausal women with mild dyslipidaemia, effects depend strongly on duration; purified HT for 8 weeks does not alter lipids, whereas OLE providing 100 mg oleuropein per day produces modest TG reductions after 12 weeks and clinically meaningful TC and LDL-C reductions after 12 months. Taken together, the strongest and most clinically relevant lipid-lowering effects are observed in cardiovascular-risk populations and in long-duration hyperlipidaemia trials using OLE formulations standardized for ∼100–200 mg per day oleuropein for ≥8 weeks. To better understand the mechanisms underlying these effects, the following section explores the molecular and physiological pathways through which olive leaf polyphenols may influence lipid metabolism, vascular health, and systemic inflammation.4. Oleuropein and hydroxytyrosol effects on blood lipid management: mechanisms of action
Understanding the mechanisms through which olive leaf polyphenols influence lipid and lipoprotein metabolism and other CVD risk factors is essential for interpreting clinical outcomes and identifying potential therapeutic targets. While numerous studies have demonstrated lipid-lowering effects of OLE and olive polyphenol enriched olive oil, the underlying biochemical pathways remain a key area of interest.4.1 Antioxidant activity of olive leaf polyphenols
Olive leaf-derived polyphenols exert antioxidant effects through several mechanisms, including direct radical scavenging, enhancement of endogenous antioxidant defences, modulation of mitochondrial function, and protection against oxidative lipid and DNA damage. These effects have been demonstrated across in vitro, animal, and ex vivo analyses, supporting their relevance for oxidative balance and vascular–metabolic health.Both oleuropein and HT can directly neutralize reactive oxygen species (ROS) through electron donation, thereby reducing oxidative stress at the cellular level.88,89 In addition to direct scavenging, these compounds upregulate endogenous antioxidant systems. Several studies report increased activity and/or expression of superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) following exposure to olive polyphenols.90–93 Improvements in glutathione homeostasis, such as normalization of total glutathione or restoration of the GSH/GSSG ratio, have also been observed under oxidative challenge.92,94
Olive polyphenols additionally modulate Nrf2 signalling, a key regulator of cellular antioxidant defence. In spontaneously hypertensive rats, oleuropein increased the expression of Nrf2 and its downstream targets, including NAD(P)H quinone dehydrogenase 1 (NQO-1) and heme oxygenase-1 (HO-1), in the paraventricular nucleus of the hypothalamus. This was accompanied by improved mitochondrial biogenesis, reduced superoxide production, and enhanced antioxidant capacity.95 HT similarly supports mitochondrial function by increasing respiratory chain activity, mitochondrial membrane potential, and mtDNA content, mediated through regulators such as PGC-1α, AMPK, Akt, and FOXO3a.92,93,96–101 These mitochondrial effects are relevant to fatty acid oxidation and overall metabolic resilience.
Lipid protection is another consistent finding. In animal models, oleuropein reduced lipid peroxidation markers such as MDA and thiobarbituric acid reactive substances across multiple tissues, including heart, liver, kidney, pancreas, and testes.102–106 HT has been shown to protect LDL particles from oxidative modification, a critical step in atherogenesis. In animal studies, HT reduced circulating oxidised LDL levels,107,108 which was mechanistically linked to an increase in LDL lag-time,109,110 indicating delayed oxidation. In vitro, HT protected LDL from oxidation,89 and its incorporation into LDL particles was associated with reduced oxidation.69,111,112 In addition, HT may enhance aryl esterase activity on HDL, contributing to LDL protection via HDL-mediated antioxidant defence.113,114
Vascular and cellular antioxidant effects have also been reported. Oleuropein lowered ROS levels in platelets stimulated with arachidonic acid via NOX2 inhibition,115 and may counteract endothelial dysfunction by preventing the accumulation of asymmetric dimethylarginine, a known inhibitor of NO synthesis.116 In rat aorta models, HT reduced ROS production,117 and in human endothelial cells and erythrocytes, it showed dose-dependent scavenging activity.93,118,119 HT reduced oxidative burst in macrophages,120 and decreased monocyte adhesion to endothelial cells,121 which suggests that antioxidant effects may translate to reduced early atherogenic signaling.
Finally, olive polyphenols protect cellular integrity under oxidative stress. In bisphenol A-treated rats, oleuropein reduced oxidative DNA damage and improved trolox-equivalent antioxidant capacity in liver and kidney tissue.91,122 Furthermore, polyphenol-rich olive extracts containing oleuropein protected against genotoxicity induced by heterocyclic amines in human peripheral blood mononuclear cells, pointing towards a broader systemic antioxidant role.123 In pancreatic β-cells and skeletal muscle cells, olive polyphenols attenuated ROS-induced cytotoxicity and preserved glucose-responsive function.124–126 These findings suggest that olive leaf polyphenols may indirectly support lipid metabolism by preserving insulin sensitivity and β-cell function under oxidative stress.
Collectively, these data indicate that olive polyphenols, through direct antioxidant actions, reinforcement of endogenous defense pathways, protection of mitochondrial function, and reduction of lipid and DNA oxidation, provide support against oxidative stress. These actions are particularly relevant in the context of atherogenesis, where oxidative stress and lipid peroxidation play a central role in atherosclerosis progression.
4.2 Lipid metabolism modulation of olive leaf polyphenols
Olive leaf polyphenols may influence lipid and lipoprotein regulation through multiple converging mechanisms that affect lipid synthesis, transport, storage, and clearance. Across animal models, supplementation with olive polyphenols consistently reduces serum TG, TC, and LDL-C, and in several studies increases HDL-C concentrations.88,91,124,127 These effects are supported by mechanistic data demonstrating actions at enzymatic, transcriptional, and mitochondrial levels.A first set of mechanisms involves modulation of key enzymes in lipid biosynthesis and oxidation. Oleuropein has been shown to inhibit key enzymes involved in lipid biosynthesis, including acetyl-CoA carboxylase and fatty acid synthase (fatty acid biosynthesis), diacylglycerol acyltransferase (TG synthesis), and HMG-CoA reductase (cholesterol synthesis).128 In addition, oleuropein activated AMPK in adipose tissue, a central metabolic regulator that inhibits lipogenesis and promotes fatty acid oxidation through phosphorylation of downstream targets.124 These enzymatic effects collectively suppress lipid accumulation and promote a metabolic shift toward greater lipid catabolism.
In parallel with these enzymatic effects, olive leaf polyphenols exert transcriptional control over lipid-regulatory pathways. Oleuropein suppressed the expression of key adipogenic and lipogenic genes, including PPARγ, C/EBPα, SREBP-1c, FAS, LPL, FABP-4, and CD36 in preadipocyte cell lines.129–131 This transcriptional suppression limits lipid storage in adipocytes and limits adipose tissue expansion, thereby reducing the flux of free fatty acids and improving systemic lipid handling. HT produces similar anti-adipogenic effects. In vitro, HT reduced TG storage in 3T3-L1 adipocytes,99,130 and increased glycerol release, indicating enhanced lipolysis.130 Additionally, HT inhibited adipocyte differentiation in these 3T3-L1 cells.130 These transcriptional actions help limit free fatty acid spillover from adipose tissue and improve systemic lipid handling.
Olive polyphenols also affect lipid distribution and circulating lipoprotein levels in vivo. HT supplementation significantly reduced TG accumulation in blood, liver, skeletal muscle, and adipose tissue.92 Similar effects were observed in rats,94 and in high-fat diet-fed rats, where serum cholesterol levels were lowered.132 HT also influenced circulating lipoprotein levels. In obese rats, HT lowered both LDL-C and HDL-C,92 while in control rats, 0.03% dietary HT reduced total plasma cholesterol.94 Additionally, HT treatment inhibited epididymal and perirenal fat accumulation and limited liver weight gain in obese mice,92 suggesting systemic effects on lipid storage and distribution.
At the mitochondrial level, HT supports enhanced lipid oxidation. In db/db mice, HT improved mitochondrial function, as evidenced by increased complex I and IV activity in skeletal muscle.92In vitro, HT increased oxygen consumption in adipocytes, suggesting a shift toward oxidative metabolism,92 and upregulated markers of mitochondrial biogenesis and mass.99 HT also modulates regulators such as PGC-1α and UCP-2 expression.98 Beyond fatty acid oxidation, in foam cells HT has been shown to activate the PPARγ/LXRα pathway, leading to upregulation of ABCA1 expression and enhanced cholesterol efflux, thereby reducing intracellular cholesterol accumulation and potentially limiting atherogenesis.133
HT may also inhibit hepatic de novo lipogenesis. In obese mice, HT reduced hepatic SREBP-1c expression,92 suggesting decreased synthesis of fatty acids and cholesterol. Together, these mitochondrial and transcriptional effects reinforce a metabolic phenotype favouring lipid oxidation over lipid accumulation.
It is worth noting that not all findings are consistent. A 2006 study reported that HT supplementation for 10 weeks in ApoE-deficient mice increased plasma cholesterol, very low-density lipoprotein cholesterol (VLDL-C), and LDL-C levels, reduced ApoA-1, and led to a 17.9% increase in atherosclerotic lesion area, alongside enhanced monocyte activation.134 The authors concluded that isolated HT, outside the olive oil matrix, could exert pro-atherogenic effects. However, these findings have since been challenged by more recent research. A 2020 study using a similar ApoE-deficient mouse model found that HT administration over 16 weeks significantly reduced atherosclerotic lesion area by 26.9%, while also lowering serum TG (−17.4%), TC (−15.2%), and LDL-C (−17.9%), and increasing HDL-C (+26.9%) concentrations.135 Given the comparable model and extended intervention period, the more recent findings likely provide a more accurate reflection of the effects of HT, suggesting a predominantly protective role in atherosclerosis.
Although human mechanistic evidence remains limited, one randomized, controlled crossover trial examined in vivo transcriptomic responses in white blood cells following consumption of a single dose of polyphenol-rich olive oil (with 26 mg oleuropein-derived secoiridoids and 1 mg HT). The intervention upregulated genes involved in cholesterol efflux and lipid handling, including ABCA1, SR-B1, CD36, PPARα, PPARBP, PPARγ and PPARδ.136 As the polyphenol profile was dominated by oleuropein-derived secoiridoids rather than glycosylated oleuropein, these findings cannot be directly extrapolated to olive leaf polyphenols. However, the study offers supportive evidence that olive polyphenols can modulate lipid-related pathways in humans.
In summary, olive leaf polyphenols modulate lipid metabolism through suppression of lipogenesis, inhibition of lipid biosynthetic enzymes, promotion of fatty acid oxidation, and enhancement of cholesterol efflux. These actions are mediated by AMPK, PPARγ/LXRα, and SREBP-1c signalling pathways and have been observed across multiple metabolic tissues.
4.3 Inflammatory modulation and vascular protection of olive leaf polyphenols
Chronic low-grade inflammation plays a central role in dyslipidaemia and atherogenesis by impairing lipid and lipoprotein metabolism, promoting foam cell formation, and disrupting endothelial integrity. Olive leaf polyphenols attenuate inflammatory signalling through multiple pathways that are directly relevant to vascular health and lipid regulation.Olive leaf polyphenols consistently reduce the production of key pro-inflammatory cytokines across cell models. In murine macrophages and adipocytes, oleuropein has been shown to suppress the production of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6, primarily via AMPK activation, which inhibits NF-κB signalling.124,137 HT reduced pro-inflammatory cytokine expression in macrophages (THP-1 and RAW264.7 cells), including TNF-α and IL-1β, and inhibited NF-κB and MAPK signalling.138,139 HT also promoted M2 macrophage polarization, which is associated with enhanced cholesterol efflux and resolution of inflammation.140
Anti-inflammatory effects extend beyond immune cells. Oleuropein blocked IL-4-induced transcriptional activity and prevented eosinophil and macrophage infiltration in epithelial cell cultures, reducing inflammation-associated tissue remodelling.141 In gastric ulcer models, high-dose oleuropein reversed HCl/ethanol-induced increases in IL-1β and TNF-α, suggesting systemic anti-inflammatory potential.142 Oleuropein also inhibited 5-lipoxygenase, reduced leukotriene B4 production in leukocyte models,56,143 and suppressed COX-2 and prostaglandin E2 synthesis in cultured endothelial cells, contributing to reduced angiogenic and inflammatory signalling.144 HT exerts anti-inflammatory effects in non-immune cells as well. In senescent MRC5 and NHDF fibroblasts, chronic HT exposure lowered IL-6 and NF-κB activity.145 In keratinocytes, HT inhibited IL-1β-, IL-6-, and IL-8-induced gene expression.146 In HaCaT cells stimulated with a psoriatic cytokine cocktail, HT reduced IL-6, IL-8, and TNF-α.147 In colonic epithelial cells exposed to benzo[a]pyrene, HT suppressed IL-6, IL-8, VEGF, and CXCL13, and inhibited ERK1/2 phosphorylation.148 These effects may indirectly influence lipid metabolism via systemic inflammation and gut–liver axis signalling.
Olive leaf polyphenols also modulate inflammatory pathways in vivo. In a pristane-induced lupus model, oral HT reduced IL-1β and IL-6 in splenocytes and macrophages, and inhibited NF-κB and MAPK signalling in renal tissue.149 In LPS-induced liver injury (C57BL/6 mice), HT shifted macrophage polarization toward M2 and lowered hepatic TNF-α, IL-1β, and IL-6.140 Given the liver's central role in lipid metabolism, this suggests HT may improve lipid handling by reducing hepatic inflammation. In ApoE-deficient mice, HT reduced aortic plaque burden and systemic inflammatory markers (CRP, TNF-α, IL-1β, IL-6), while increasing IL-10.135 This anti-inflammatory profile is associated with improved lipid profiles and reduced atherogenesis. In DSS-induced colitis, HT suppressed IL-6, IL-1β, TNF-α, and NF-κB activation, and modulated gut microbiota composition toward anti-inflammatory profiles, leading to an increased production of short-chain fatty acids.150,151 In neuronal tissue in the hippocampus of mice, oleuropein lowered TNF-α and IL-1β levels, indicating broader systemic anti-inflammatory effects.152 Together, these findings support a systemic anti-inflammatory role that intersects with hepatic lipid handling, vascular inflammation, and immune-metabolic crosstalk.
Inflammation-induced endothelial activation plays a key role in early atherogenesis. Olive leaf polyphenols reduced adhesion molecule expression, including VCAM-1 and ICAM-1, in activated endothelial cells.121,141,144,153,154 This limits monocyte adhesion and infiltration into the vascular wall, helping to prevent foam cell formation and the development of lipid-rich plaques. HT-induced reductions in NF-κB activation in endothelial models further contribute to vascular protection by stabilizing endothelial function under inflammatory stress.154,155 These effects may attenuate foam cell formation and slow the progression of lipid-rich atherosclerotic plaques.
Beyond cytokine modulation, olive leaf polyphenols support vascular homeostasis by enhancing nitric oxide (NO) bioavailability. Studies in animal models and endothelial cells show that oleuropein enhanced NO bioavailability by reducing oxidative stress and preventing accumulation of asymmetric dimethylarginine (ADMA), an endogenous inhibitor of NO synthase.116 This may improve endothelial-dependent vasodilation and reduce LDL oxidation, both relevant to lipid-driven vascular injury.
Olive leaf polyphenols may also influence platelet activation, a key process in atherothrombosis, through their capacity to attenuate redox-sensitive signalling steps involved in collagen- and thrombin-induced platelet reactivity. In vitro studies show that OLE can dose-dependently inhibit platelet aggregation and ATP release, an effect attributed to their strong hydrogen-peroxide-scavenging activity.156 Broader mechanistic work indicates that olive-derived phenolics reduce thromboxane B2 formation, dampen NOX2-mediated ROS generation, and limit the release of pro-atherogenic mediators such as CD40L and PDGF, suggesting that multiple constituents within the olive polyphenol matrix converge on oxidative and eicosanoid pathways central to platelet activation.157 Together, these findings support a plausible mechanistic role for olive polyphenols in modulating platelet-driven processes relevant to atherothrombosis.
Together, these data indicate that olive leaf polyphenols mitigate systemic and vascular inflammation by inhibiting NF-κB/MAPK signalling, modulating macrophage polarization, reducing cytokine production, improving endothelial function, and inhibit platelet aggregation. These mechanisms are relevant to blood lipid regulation because they influence hepatic lipid metabolism, macrophage-driven foam cell formation, and vascular inflammation, which are key processes in dyslipidaemia and atherosclerosis.
4.4 Summary
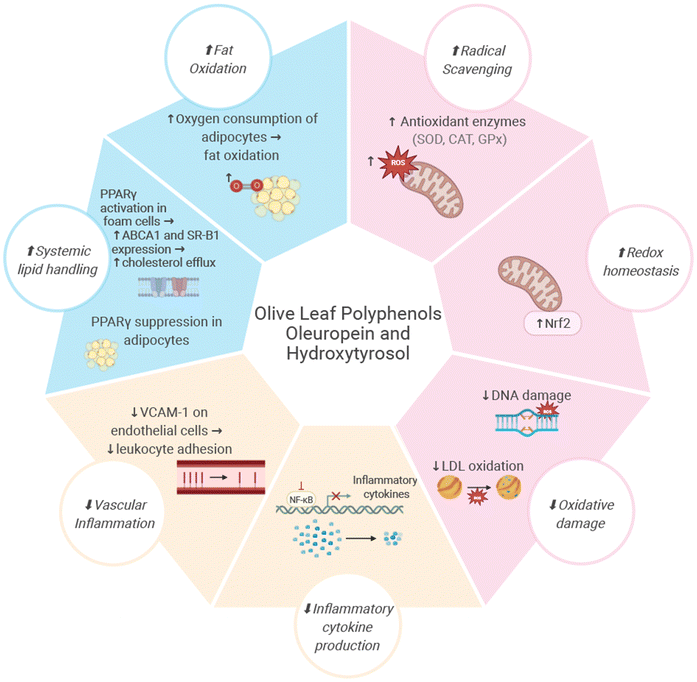
Taken together, the evidence summarized in this section shows that olive leaf polyphenols affect oxidative stress, inflammation, endothelial integrity, and lipid metabolism. Across cellular, animal, and one human study, oleuropein and HT, activate antioxidant responses (Nrf2 signalling), suppress pro-inflammatory cascades (NF-κB/MAPK), improve endothelial function through reduced adhesion molecule expression, and promote lipid homeostasis via AMPK and PPAR-dependent mechanisms. These effects suggest that the biological effects of olive leaf polyphenols should be viewed not in isolation, but as part of an integrated network with relevance to atherogenesis and dyslipidaemia. Fig. 2 provides an overview of these converging pathways and illustrates how olive leaf polyphenols influence key molecular processes implicated in cardiometabolic health.5. Limitations and future directions
Despite the promising findings outlined above, such as improvements in lipid profiles, glycaemic control, antioxidant defences, and vascular function, several limitations constrain the interpretation and generalizability of available clinical evidence on the effect of olive leaf polyphenols on blood lipid profiles and vascular health.A major limitation in the current literature is the heterogeneity in intervention composition. Many studies use OLE or polyphenol-enriched oils with varying and often poorly defined concentrations of total phenols, oleuropein, and HT, complicating dose–response analysis and hindering comparability across studies. In addition to compositional variability, study designs often lack robust control conditions. Several trials do not include a placebo group, while others compare low versus high polyphenolic content without a true control. Furthermore, some studies only report within-group changes despite having a control arm. This limits the interpretability of outcomes and weakens the strength of the evidence. Future studies should use standardized, placebo-controlled designs using olive leaf interventions with fully quantified phenolic profiles to enable meaningful dose–response comparisons and clearer interpretation of mechanistic outcomes. In addition, proper statistical analyses comparing treatment groups to controls, instead of relying on within-group changes from baseline, should be used.
A further limitation is the poor characterization of bioavailability; reported plasma concentrations of oleuropein and HT are typically in the nanomolar range and highly variable, analytical methods for phenol quantification lack standardization, and studies do not account for microbiota-dependent metabolism, despite evidence that gut microbiota markedly affect oleuropein degradation and HT production.158–161 Future studies should incorporate microbiome profiling, as interindividual differences in gut microbial metabolism of oleuropein and HT may influence bioavailability and thereby modify their effect.
Moreover, mechanistic insights derived from olive oil-derived polyphenol studies cannot be directly extrapolated to olive leaf polyphenols, as olive oil contains predominantly secoiridoid aglycones within a lipid-rich matrix, whereas olive leaf extracts primarily provide glycosylated oleuropein and HT in a different compositional and metabolic context.
Finally, mechanistic endpoints are underreported in clinical trials. While several studies demonstrate effects on lipid parameters, few include molecular or cellular markers that could clarify underlying mechanisms in humans. As a result, many mechanistic pathways described in the preclinical literature have not yet been conclusively demonstrated in humans, limiting the translational value of these findings and hampering the ability to bridge in vitro insights with in vivo clinical outcomes. Future clinical studies should incorporate mechanistic endpoints, such as gene expression, inflammatory markers, or ex vivo cellular assays with human samples, to clarify how olive leaf polyphenols exert their effects at the molecular level for the development of more targeted applications.
Together, these gaps highlight the need for well-controlled trials using well-defined standardized OLE interventions with mechanistic endpoints and stratification by metabolic phenotype. Such approaches will enhance the interpretability of findings and allow for more accurate observation of health effects, minimizing confounding factors. Moreover, by integrating microbiota-related analyses, future studies may help identify which individuals are most likely to benefit from olive leaf polyphenol interventions.
6. Conclusion
Olive leaf polyphenols, including oleuropein and HT, can contribute to cardiometabolic health through complementary mechanisms. Clinical evidence suggests that olive leaf polyphenols improve lipid profiles and glycaemic control while offering antioxidant, anti-inflammatory, and vascular-protective effects. Mechanistically, olive leaf polyphenols target pathways involved in lipid metabolism, oxidative stress, and vascular inflammation. They enhance antioxidant defences via Nrf2 activation, inhibit NF-κB-mediated inflammation, and improve endothelial function by downregulating adhesion molecules. Additionally, they can promote fatty acid oxidation, suppress lipogenesis, and upregulate cholesterol efflux transporters such as ABCA1 and SR-B1. Current evidence indicates that olive leaf-derived polyphenols have the potential to influence lipid metabolism and vascular health, but further well-controlled studies with well quantified olive leaf polyphenol interventions are needed before firm recommendations can be made.Author contributions
Conceptualization, S. v. S. and Y. S.; investigation, S. v. S.; writing – original draft preparation, S. v. S., Y. S., and J. P.; writing – review and editing, S. v. S., Y. S., and J. P.; visualization, S. v. S.; supervision, Y. S. and J. P. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
SvS and YS are employees of Solabia BV. YS holds a patent application of Bioactor BV (WO2024079251A1). All potential conflicts of interest have been disclosed in the manuscript. The authors affirm that these affiliations did not influence study selection or interpretation. All other authors declare no conflict of interest.Data availability
This narrative review is based on previously published studies, all of which are cited in the manuscript. No new data were generated or analyzed in this study. Data sharing is not applicable to this article.Acknowledgements
The graphical abstract and Fig. 2 were created with Biorender.com.References
- B. Chong, J. Jayabaskaran, S. M. Jauhari, S. P. Chan, R. Goh and M. T. W. Kueh, et al., Global burden of cardiovascular diseases: projections from 2025 to 2050, Eur. J. Prev. Cardiol., 2024, 32, 1001–1015 CrossRef PubMed.
- P. W. F. Wilson, R. B. D'Agostino, H. Parise, L. Sullivan and J. B. Meigs, Metabolic Syndrome as a Precursor of Cardiovascular Disease and Type 2 Diabetes Mellitus, Circulation, 2005, 112(20), 3066–3072 CrossRef CAS PubMed.
- J. D. Tune, A. G. Goodwill, D. J. Sassoon and K. J. Mather, Cardiovascular consequences of metabolic syndrome, Transl. Res., 2017, 183, 57–70 CrossRef CAS PubMed.
- S. Mottillo, K. B. Filion, J. Genest, L. Joseph, L. Pilote and P. Poirier, et al., The Metabolic Syndrome and Cardiovascular Risk, J. Am. Coll. Cardiol., 2010, 56(14), 1113–1132 CrossRef PubMed.
- S. L. Samson and A. J. Garber, Metabolic Syndrome, Endocrinol. Metab. Clin. North Am., 2014, 43(1), 1–23 CrossRef PubMed.
- D. Le Goff, N. Aerts, M. Odorico, M. Guillou-Landreat, G. Perraud and H. Bastiaens, et al., Practical dietary interventions to prevent cardiovascular disease suitable for implementation in primary care: an ADAPTE-guided systematic review of international clinical guidelines, Int. J. Behav. Nutr. Phys. Act., 2023, 20(1), 93 CrossRef PubMed.
- R. J. Widmer, A. J. Flammer, L. O. Lerman and A. Lerman, The Mediterranean Diet, its Components, and Cardiovascular Disease, Am. J. Med., 2015, 128(3), 229–238 CrossRef PubMed.
- M. A. Martínez-González, A. Gea and M. Ruiz-Canela, The Mediterranean Diet and Cardiovascular Health, Circ. Res., 2019, 124(5), 779–798 CrossRef PubMed.
- M. de Lorgeril, P. Salen, J. L. Martin, I. Monjaud, J. Delaye and N. Mamelle, Mediterranean Diet, Traditional Risk Factors, and the Rate of Cardiovascular Complications After Myocardial Infarction, Circulation, 1999, 99(6), 779–785 CrossRef CAS PubMed.
- V. Dilis, M. Katsoulis, P. Lagiou, D. Trichopoulos, A. Naska and A. Trichopoulou, Mediterranean diet and CHD: the Greek European Prospective Investigation into Cancer and Nutrition cohort, Br. J. Nutr., 2012, 108(4), 699–709 CrossRef CAS PubMed.
- R. Briante, M. Patumi, S. Terenziani, E. Bismuto, F. Febbraio and R. Nucci, Olea europaea L. Leaf Extract and Derivatives: Antioxidant Properties, J. Agric. Food Chem., 2002, 50(17), 4934–4940 CrossRef CAS PubMed.
- S. Granados-Principal, J. L. Quiles, C. L. Ramirez-Tortosa, P. Sanchez-Rovira and M. C. Ramirez-Tortosa, Hydroxytyrosol: from laboratory investigations to future clinical trials, Nutr. Rev., 2010, 68(4), 191–206 CrossRef PubMed.
- J. A. Rothwell, J. Perez-Jimenez, V. Neveu, A. Medina-Remon, N. M'Hiri and P. Garcia-Lobato, et al., Phenol-Explorer 3.0: a major update of the Phenol-Explorer database to incorporate data on the effects of food processing on polyphenol content, Database, 2013, 2013, bat070 CrossRef PubMed.
- T. Mehany, J. M. González-Sáiz and C. Pizarro, Hydroxytyrosol-Infused Extra Virgin Olive Oil: A Key to Minimizing Oxidation, Boosting Antioxidant Potential, and Enhancing Physicochemical Stability During Frying, Antioxidants, 2025, 14(3), 368 CrossRef CAS PubMed.
- M. Brenes, A. García, P. García and A. Garrido, Rapid and Complete Extraction of Phenols from Olive Oil and Determination by Means of a Coulometric Electrode Array System, J. Agric. Food Chem., 2000, 48(11), 5178–5183 CrossRef CAS PubMed.
- J. Fernández-Bolaños, G. Rodríguez, R. Rodríguez, A. Heredia, R. Guillén and A. Jiménez, Production in Large Quantities of Highly Purified Hydroxytyrosol from Liquid−Solid Waste of Two-Phase Olive Oil Processing or “Alperujo”, J. Agric. Food Chem., 2002, 50(23), 6804–6811 CrossRef PubMed.
- O. Frumuzachi, L. I. Gavrilaş, D. C. Vodnar, S. Rohn and A. Mocan, Systemic Health Effects of Oleuropein and Hydroxytyrosol Supplementation: A Systematic Review of Randomized Controlled Trials, Antioxidants, 2024, 13(9), 1040 CrossRef CAS PubMed.
- C. S. Papageorgiou, P. Lyri, I. Xintaropoulou, I. Diamantopoulos, D. P. Zagklis and C. A. Paraskeva, High-Yield Production of a Rich-in-Hydroxytyrosol Extract from Olive (Olea europaea) Leaves, Antioxidants, 2022, 11(6), 1042 CrossRef CAS PubMed.
- D. Borjan, M. Leitgeb, Ž Knez and M. K. Hrnčič, Microbiological and Antioxidant Activity of Phenolic Compounds in Olive Leaf Extract, Molecules, 2020, 25(24), 5946 CrossRef CAS PubMed.
- M. Moudache, M. Colon, C. Nerín and F. Zaidi, Phenolic content and antioxidant activity of olive by-products and antioxidant film containing olive leaf extract, Food Chem., 2016, 212, 521–527 CrossRef CAS PubMed.
- P. G. Lins, S. Marina Piccoli Pugine, A. M. Scatolini and M. P. de Melo, In vitro antioxidant activity of olive leaf extract (Olea europaea L.) and its protective effect on oxidative damage in human erythrocytes, Heliyon, 2018, 4(9), e00805 CrossRef PubMed.
- T. Grubić Kezele and B. Ćurko-Cofek, Neuroprotective Panel of Olive Polyphenols: Mechanisms of Action, Anti-Demyelination, and Anti-Stroke Properties, Nutrients, 2022, 14(21), 4533 CrossRef PubMed.
- A. H. Muhammad, K. Afsar, H. Muhammad, F. Umar and P. Shagufta, Traditional Uses, Phytochemistry, and Pharmacology of Olea europaea (Olive), J. Evidence-Based Complementary Altern. Med., 2015, 541591 Search PubMed.
- M. L. Clodoveo, P. Crupi, A. Annunziato and F. Corbo, Innovative Extraction Technologies for Development of Functional Ingredients Based on Polyphenols from Olive Leaves, Foods, 2021, 11(1), 103 CrossRef PubMed.
- E. Kritikou, N. P. Kalogiouri, L. Kolyvira and N. S. Thomaidis, Target and Suspect HRMS Metabolomics for the Determination of Functional Ingredients in 13 Varieties of Olive Leaves and Drupes from Greece, Molecules, 2020, 25(21), 4889 CrossRef CAS PubMed.
- E. Debs, A. M. Abi-Khattar, H. N. Rajha, R. M. Abdel-Massih, J. C. Assaf and M. Koubaa, et al., Valorization of Olive Leaves through Polyphenol Recovery Using Innovative Pretreatments and Extraction Techniques, An Updated Review, Separations, 2023, 10(12), 587 CrossRef CAS.
- J. Giacometti, G. Žauhar and M. Žuvić, Optimization of Ultrasonic-Assisted Extraction of Major Phenolic Compounds from Olive Leaves (Olea europaea L.) Using Response Surface Methodology, Foods, 2018, 7(9), 149 CrossRef PubMed.
- A. Dobrinčić, M. Repajić, I. E. Garofulić, L. Tuđen, V. Dragović-Uzelac and B. Levaj, Comparison of Different Extraction Methods for the Recovery of Olive Leaves Polyphenols, Processes, 2020, 8(9), 1008 CrossRef.
- B. Soldo, T. Bilušić, J. Giacometti, I. Ljubenkov, V. Čikeš Čulić and A. Bratanić, et al., A Comparative Study of Oleuropein Extraction from Wild Olive Leaves (Olea europea subsp. oleaster, Hoffmanns. & Link), Its Gastrointestinal Stability, and Biological Potential, Appl. Sci., 2024, 14(2), 869 CrossRef CAS.
- F. S. Markhali, J. A. Teixeira and C. M. R. Rocha, Olive Tree Leaves—A Source of Valuable Active Compounds, Processes, 2020, 8(9), 1177 CrossRef CAS.
- K. Kiritsakis, M. G. Kontominas, C. Kontogiorgis, D. Hadjipavlou-Litina, A. Moustakas and A. Kiritsakis, Composition and Antioxidant Activity of Olive Leaf Extracts from Greek Olive Cultivars, J. Am. Oil Chem. Soc., 2010, 87(4), 369–376 CrossRef CAS.
- S. Selim, M. Albqmi, M. M. Al-Sanea, T. S. Alnusaire, M. S. Almuhayawi and H. AbdElgawad, et al., Valorizing the usage of olive leaves, bioactive compounds, biological activities, and food applications: A comprehensive review, Front. Nutr., 2022, 9 Search PubMed.
- I. Hassen, H. Casabianca and K. Hosni, Biological activities of the natural antioxidant oleuropein: Exceeding the expectation – A mini-review, J. Funct. Foods, 2015, 18, 926–940 CrossRef CAS.
- M. M. Özcan and B. Matthäus, A review: benefit and bioactive properties of olive (Olea europaea L.) leaves, Eur. Food Res. Technol., 2017, 243(1), 89–99 CrossRef.
- F. Caponio, V. Alloggio and T. Gomes, Phenolic compounds of virgin olive oil: influence of paste preparation techniques, Food Chem., 1999, 64(2), 203–209 CrossRef CAS.
- C. I. G. Tuberoso, A. Kowalczyk, E. Sarritzu and P. Cabras, Determination of antioxidant compounds and antioxidant activity in commercial oilseeds for food use, Food Chem., 2007, 103(4), 1494–1501 CrossRef CAS.
- M. Leri, A. Natalello, E. Bruzzone, M. Stefani and M. Bucciantini, Oleuropein aglycone and hydroxytyrosol interfere differently with toxic Aβ1–42 aggregation, Food Chem. Toxicol., 2019, 129, 1–12 CrossRef CAS PubMed.
- S. Antonopoulou and C. A. Demopoulos, Protective Effect of Olive Oil Microconstituents in Atherosclerosis: Emphasis on PAF Implicated Atherosclerosis Theory, Biomolecules, 2023, 13(4), 700 CrossRef CAS PubMed.
- A. D. Marrero, A. R. Quesada, B. Martínez-Poveda and M.Á Medina, Anti-Cancer, Anti-Angiogenic, and Anti-Atherogenic Potential of Key Phenolic Compounds from Virgin Olive Oil, Nutrients, 2024, 16(9), 1283 CrossRef CAS PubMed.
- F. Xu, Y. Li, M. Zheng, X. Xi, X. Zhang and C. Han, Structure Properties, Acquisition Protocols, and Biological Activities of Oleuropein Aglycone, Front. Chem., 2018, 6 Search PubMed.
- I. Khelouf, I. J. Karoui, A. Lakoud, M. Hammami and M. Abderrabba, Comparative chemical composition and antioxidant activity of olive leaves Olea europaea L. of Tunisian and Algerian varieties, Heliyon, 2023, 9(12), e22217 CrossRef CAS PubMed.
- K. L. Tuck and P. J. Hayball, Major phenolic compounds in olive oil: metabolism and health effects, J. Nutr. Biochem., 2002, 13(11), 636–644 CrossRef CAS PubMed.
- F. D. Ragione, V. Cucciolla, A. Borriello, V. D. Pietra, G. Pontoni and L. Racioppi, et al., Hydroxytyrosol, a Natural Molecule Occurring in Olive Oil, Induces Cytochrome c-Dependent Apoptosis, Biochem. Biophys. Res. Commun., 2000, 278(3), 733–739 CrossRef CAS PubMed.
- C. Bender, S. Strassmann and C. Golz, Oral Bioavailability and Metabolism of Hydroxytyrosol from Food Supplements, Nutrients, 2023, 15(2), 325 CrossRef CAS PubMed.
- R. García-Villalba, M. Larrosa, S. Possemiers, F. A. Tomás-Barberán and J. C. Espín, Bioavailability of phenolics from an oleuropein-rich olive (Olea europaea) leaf extract and its acute effect on plasma antioxidant status: comparison between pre- and postmenopausal women, Eur. J. Nutr., 2014, 53(4), 1015–1027 CrossRef PubMed.
- P. L. Huang, P. L. Huang and S. Lee-Huang, Oleuropein and Related Compounds Reduce Atherosclerosis, Open Conf. Proc. J., 2010, 1(1), 81–86 CAS.
- S. Galmés, B. Reynés, M. Palou, A. Palou-March and A. Palou, Absorption, Distribution, Metabolism, and Excretion of the Main Olive Tree Phenols and Polyphenols: A Literature Review, J. Agric. Food Chem., 2021, 69(18), 5281–5296 CrossRef PubMed.
- M. M. Santos, C. Piccirillo, P. M. L. Castro, N. Kalogerakis and M. E. Pintado, Bioconversion of oleuropein to hydroxytyrosol by lactic acid bacteria, World J. Microbiol. Biotechnol., 2012, 28(6), 2435–2440 CrossRef CAS PubMed.
- M. de Bock, J. G. B. Derraik, C. M. Brennan, J. B. Biggs, P. E. Morgan and S. C. Hodgkinson, et al., Olive (Olea europaea L.) Leaf Polyphenols Improve Insulin Sensitivity in Middle-Aged Overweight Men: A Randomized, Placebo-Controlled, Crossover Trial, PLoS One, 2013, 8(3), e57622 CrossRef CAS PubMed , Nerurkar P V., editor.
- H. Tan, K. Tuck, I. Stupans and P. Hayball, Simultaneous determination of oleuropein and hydroxytyrosol in rat plasma using liquid chromatography with fluorescence detection, J. Chromatogr. B, 2003, 785(1), 187–191 Search PubMed.
- S. D'Angelo, C. Manna, V. Migliardi, O. Mazzoni, P. Morrica and G. Capasso, et al., Pharmacokinetics and metabolism of hydroxytyrosol, a natural antioxidant from olive oil, Drug Metab. Dispos., 2001, 29(11), 1492–1498 Search PubMed.
- A. Serra, L. Rubió, X. Borràs, A. Macià, M. Romero and M. Motilva, Distribution of olive oil phenolic compounds in rat tissues after administration of a phenolic extract from olive cake, Mol. Nutr. Food Res., 2012, 56(3), 486–496 CrossRef CAS PubMed.
- L. Rubió, A. Serra, A. Macià, C. Piñol, M. P. Romero and M. J. Motilva, In vivo distribution and deconjugation of hydroxytyrosol phase II metabolites in red blood cells: A potential new target for hydroxytyrosol, J. Funct. Foods, 2014, 10, 139–143 CrossRef.
- M. López de las Hazas, L. Rubió, A. Kotronoulas, R. de la Torre, R. Solà and M. Motilva, Dose effect on the uptake and accumulation of hydroxytyrosol and its metabolites in target tissues in rats, Mol. Nutr. Food Res., 2015, 59(7), 1395–1399 CrossRef PubMed.
- M. N. Vissers, P. L. Zock, A. J. C. Roodenburg, R. Leenen and M. B. Katan, Olive Oil Phenols Are Absorbed in Humans, J. Nutr., 2002, 132(3), 409–417 CrossRef CAS PubMed.
- S. Lockyer, G. Corona, P. Yaqoob, J. P. E. Spencer and I. Rowland, Secoiridoids delivered as olive leaf extract induce acute improvements in human vascular function and reduction of an inflammatory cytokine: a randomised, double-blind, placebo-controlled, cross-over trial, Br. J. Nutr., 2015, 114(1), 75–83 Search PubMed.
- O. Khymenets, M. C. Crespo, O. Dangles, N. Rakotomanomana, C. Andres-Lacueva and F. Visioli, Human hydroxytyrosol's absorption and excretion from a nutraceutical, J. Funct. Foods, 2016, 23, 278–282 Search PubMed.
- M. G. Soni, G. A. Burdock, M. S. Christian, C. M. Bitler and R. Crea, Safety assessment of aqueous olive pulp extract as an antioxidant or antimicrobial agent in foods, Food Chem. Toxicol., 2006, 44(7), 903–915 CrossRef CAS PubMed.
- A. E. Clewell, E. Béres, A. Vértesi, R. Glávits, G. Hirka and J. R. Endres, et al., A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (BonoliveTM), Int. J. Toxicol., 2016, 35(2), 208–221 CrossRef CAS PubMed.
- C. G. Guex, F. Z. Reginato, K. C. Figueredo, A. R. H. da Silva, F. B. da Pires and R. da S. Jesus, et al., Safety assessment of ethanolic extract of Olea europaea L. leaves after acute and subacute administration to Wistar rats, Regul. Toxicol. Pharmacol., 2018, 95, 395–399 CrossRef CAS PubMed.
- M. J. Leach and I. Breakspear, Efficacy and safety of olive leaf extract (Olea europaea L.) for glycaemic control in adults with type 2 diabetes mellitus (ESOLED): A pilot randomised controlled trial, Complement Ther. Clin. Pract., 2025, 59, 101949 Search PubMed.
- R. Filip, S. Possemiers, A. Heyerick, I. Pinheiro, G. Raszewski and M. J. Davicco, et al., Twelve-month consumption of a polyphenol extract from olive (Olea europaea) in a double blind, randomized trial increases serum total osteocalcin levels and improves serum lipid profiles in postmenopausal women with osteopenia, J. Nutr., Health Aging, 2015, 19(1), 77–86 CrossRef CAS PubMed.
- T. Perrinjaquet-Moccetti, A. Busjahn, C. Schmidlin, A. Schmidt, B. Bradl and C. Aydogan, Food supplementation with an olive (Olea europaea L.) leaf extract reduces blood pressure in borderline hypertensive monozygotic twins, Phytother. Res., 2008, 22(9), 1239–1242 CrossRef PubMed.
- E. Susalit, N. Agus, I. Effendi, R. R. Tjandrawinata, D. Nofiarny and T. Perrinjaquet-Moccetti, et al., Olive (Olea europaea) leaf extract effective in patients with stage-1 hypertension: Comparison with Captopril, Phytomedicine, 2011, 18(4), 251–258 CrossRef CAS PubMed.
- B. Rugge, H. Balshem, R. Sehgal, R. Relevo, P. Gorman and M. Helfand, Screening and Treatment of Subclinical Hypothyroidism or Hyperthyroidism [Internet], 2011 Search PubMed.
- Y. Zhang, M. J. Pletcher, E. Vittinghoff, A. M. Clemons, D. R. Jacobs and N. B. Allen, et al., Association Between Cumulative Low-Density Lipoprotein Cholesterol Exposure During Young Adulthood and Middle Age and Risk of Cardiovascular Events, JAMA Cardiol., 2021, 6(12), 1406 CrossRef PubMed.
- O. Castañer, M. I. Covas, O. Khymenets, K. Nyyssonen, V. Konstantinidou and H. F. Zunft, et al., Protection of LDL from oxidation by olive oil polyphenols is associated with a downregulation of CD40-ligand expression and its downstream products in vivo in humans, Am. J. Clin. Nutr., 2012, 95(5), 1238–1244 CrossRef PubMed.
- J. Marrugat, M. I. Covas, M. Fitó, H. Schröder, E. Miró-Casas and E. Gimeno, et al., Effects of differing phenolic content in dietary olive oils on lipids and LDL oxidation, Eur. J. Nutr., 2004, 43(3), 140–147 CrossRef CAS PubMed.
- M. I. Covas, K. de la Torre, M. Farré-Albaladejo, J. Kaikkonen, M. Fitó and C. López-Sabater, et al., Postprandial LDL phenolic content and LDL oxidation are modulated by olive oil phenolic compounds in humans, Free Radical Biol. Med., 2006, 40(4), 608–616 CrossRef CAS PubMed.
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA), Scientific Opinion on the substantiation of health claims related to polyphenols in olive and protection of LDL particles from oxidative damage (ID 1333, 1638, 1639, 1696, 2865), maintenance of normal blood HDL cholesterol concentrations (ID 1639), mainte, EFSA J., 2011, 9(4), 2033 CrossRef.
- P. J. Pinckaers, H. L. Petrick, A. M. Horstman, A. Moreno-Asso, U. De Marchi and F. K. Hendriks, et al., Oleuropein Supplementation Increases Resting Skeletal Muscle Fractional Pyruvate Dehydrogenase Activity but Does Not Influence Whole-Body Metabolism: A Randomized, Double-Blind, and Placebo-Controlled Trial in Healthy, Older Males, J. Nutr., 2025, 155(5), 1373–1386 CrossRef CAS PubMed.
- M. J. Oliveras-López, J. J. M. Molina, M. V. Mir, E. F. Rey, F. Martín and H. L. G. de la Serrana, Extra virgin olive oil (EVOO) consumption and antioxidant status in healthy institutionalized elderly humans, Arch. Gerontol. Geriatr., 2013, 57(2), 234–242 CrossRef PubMed.
- M. A. Perrone, P. Gualtieri, S. Gratteri, W. Ali, D. Sergi and S. Muscoli, et al., Effects of postprandial hydroxytyrosol and derivates on oxidation of LDL, cardiometabolic state and gene expression: a nutrigenomic approach for cardiovascular prevention, J. Cardiovasc. Med., 2019, 20(7), 419–426 CrossRef CAS PubMed.
- I. Ikonomidis, K. Katogiannis, C. Chania, N. Iakovis, M. Tsoumani and A. Christodoulou, et al., Association of hydroxytyrosol enriched olive oil with vascular function in chronic coronary disease, Eur. J. Clin. Invest., 2023, 53(7), e13983 CrossRef CAS PubMed.
- Á. Naranjo, M. J. Álvarez-Soria, P. Aranda-Villalobos, A. M. Martínez-Rodríguez, E. Martínez-Lara and E. Siles, Hydroxytyrosol, a Promising Supplement in the Management of Human Stroke: An Exploratory Study, Int. J. Mol. Sci., 2024, 25(9), 4799 Search PubMed.
- R. M. Valls, M. Farràs, M. Suárez, S. Fernández-Castillejo, M. Fitó and V. Konstantinidou, et al., Effects of functional olive oil enriched with its own phenolic compounds on endothelial function in hypertensive patients. A randomised controlled trial, Food Chem., 2015, 167, 30–35 CrossRef CAS PubMed.
- S. Lockyer, I. Rowland, J. P. E. Spencer, P. Yaqoob and W. Stonehouse, Impact of phenolic-rich olive leaf extract on blood pressure, plasma lipids and inflammatory markers: a randomised controlled trial, Eur. J. Nutr., 2017, 56(4), 1421–1432 CrossRef CAS PubMed.
- M. G. Silverman, B. A. Ference, K. Im, S. D. Wiviott, R. P. Giugliano and S. M. Grundy, et al., Association Between Lowering LDL-C and Cardiovascular Risk Reduction Among Different Therapeutic Interventions, J. Am. Med. Assoc., 2016, 316(12), 1289 Search PubMed.
- P. M. Burger, J. A. N. Dorresteijn, S. Koudstaal, J. Holtrop, J. J. P. Kastelein and J. W. Jukema, et al., Course of the effects of LDL-cholesterol reduction on cardiovascular risk over time: A meta-analysis of 60 randomized controlled trials, Atherosclerosis, 2024, 396, 118540 CrossRef CAS PubMed.
- Y. Stevens, B. Winkens, D. Jonkers and A. Masclee, The effect of olive leaf extract on cardiovascular health markers: a randomized placebo-controlled clinical trial, Eur. J. Nutr., 2021, 60(4), 2111–2120 CrossRef CAS PubMed.
- C. E. Kosmas, S. Rodriguez Polanco, M. D. Bousvarou, E. J. Papakonstantinou, E. Peña Genao and E. Guzman, et al., The Triglyceride/High-Density Lipoprotein Cholesterol (TG/HDL-C) Ratio as a Risk Marker for Metabolic Syndrome and Cardiovascular Disease, Diagnostics, 2023, 13(5), 929 CrossRef CAS PubMed.
- M. P. Hermans, S. A. Ahn and M. F. Rousseau, The atherogenic dyslipidemia ratio [log(TG)/HDL-C] is associated with residual vascular risk, beta-cell function loss and microangiopathy in type 2 diabetes females, Lipids Health Dis., 2012, 11(1), 132 CrossRef CAS PubMed.
- E. Lopez-Huertas and J. Fonolla, Hydroxytyrosol supplementation increases vitamin C levels in vivo. A human volunteer trial, Redox Biol., 2017, 11, 384–389 CrossRef CAS PubMed.
- I. A. Sobenin, V. A. Myasoedova and A. N. Orekhov, Phytoestrogen-Rich Dietary Supplements in Anti-Atherosclerotic Therapy in Postmenopausal Women, Curr. Pharm. Des., 2015, 22(2), 152–163 CrossRef CAS PubMed.
- S. H. Ko and H. S. Kim, Menopause-Associated Lipid Metabolic Disorders and Foods Beneficial for Postmenopausal Women, Nutrients, 2020, 12(1), 202 CrossRef CAS PubMed.
- J. J. Thaung Zaw, P. R. C. Howe and R. H. X. Wong, Postmenopausal health interventions: Time to move on from the Women's Health Initiative?, Ageing Res. Rev., 2018, 48, 79–86 CrossRef PubMed.
- M. Imperatrice, A. Lasfar, C. A. J. van Kalkeren and F. Troost, Olive Leaf Extract Supplementation Improves Postmenopausal Symptoms: A Randomized, Double-Blind, Placebo-Controlled Parallel Study on Postmenopausal Women, Nutrients, 2024, 16(22), 3879 CrossRef CAS PubMed.
- H. Jemai, A. El Feki and S. Sayadi, Antidiabetic and Antioxidant Effects of Hydroxytyrosol and Oleuropein from Olive Leaves in Alloxan-Diabetic Rats, J. Agric. Food Chem., 2009, 57(19), 8798–8804 CrossRef CAS PubMed.
- R. Briante, F. Febbraio and R. Nucci, Antioxidant/Prooxidant Effects of Dietary Non–Flavonoid Phenols on the Cu 2+ –Induced Oxidation of Human Low–Density Lipoprotein (LDL), Chem. Biodivers., 2004, 1(11), 1716–1729 CrossRef CAS PubMed.
- M. Ivanov, U. J. Vajic, N. Mihailovic-Stanojevic, Z. Miloradovic, D. Jovovic and J. Grujic-Milanovic, et al., Highly potent antioxidant Olea europaea L. leaf extract affects carotid and renal haemodynamics in experimental hypertension: The role of oleuropein, EXCLI J., 2018, 17, 29–44 Search PubMed.
- A. Mahmoudi, F. Hadrich, I. Feki, H. Ghorbel, Z. Bouallagui and R. Marrekchi, et al., Oleuropein and hydroxytyrosol rich extracts from olive leaves attenuate liver injury and lipid metabolism disturbance in bisphenol A-treated rats, Food Funct., 2018, 9(6), 3220–3234 RSC.
- K. Cao, J. Xu, X. Zou, Y. Li, C. Chen and A. Zheng, et al., Hydroxytyrosol prevents diet-induced metabolic syndrome and attenuates mitochondrial abnormalities in obese mice, Free Radical Biol. Med., 2014, 67, 396–407 CrossRef CAS PubMed.
- H. Zrelli, M. Matsuoka, S. Kitazaki, M. Zarrouk and H. Miyazaki, Hydroxytyrosol reduces intracellular reactive oxygen species levels in vascular endothelial cells by upregulating catalase expression through the AMPK–FOXO3a pathway, Eur. J. Pharmacol., 2011, 660(2–3), 275–282 CrossRef CAS PubMed.
- E. Giordano, A. Dávalos and F. Visioli, Chronic hydroxytyrosol feeding modulates glutathione-mediated oxido-reduction pathways in adipose tissue: A nutrigenomic study, Nutr. Metab. Cardiovasc. Dis., 2014, 24(10), 1144–1150 CrossRef CAS PubMed.
- W. Sun, X. Wang, C. Hou, L. Yang, H. Li and J. Guo, et al., Oleuropein improves mitochondrial function to attenuate oxidative stress by activating the Nrf2 pathway in the hypothalamic paraventricular nucleus of spontaneously hypertensive rats, Neuropharmacology, 2017, 113, 556–566 CrossRef CAS PubMed.
- A. Zheng, H. Li, J. Xu, K. Cao, H. Li and W. Pu, et al., Hydroxytyrosol improves mitochondrial function and reduces oxidative stress in the brain of db/db mice: role of AMP-activated protein kinase activation, Br. J. Nutr., 2015, 113(11), 1667–1676 CrossRef CAS PubMed.
- S. Granados-Principal, N. El-azem, R. Pamplona, C. Ramirez-Tortosa, M. Pulido-Moran and L. Vera-Ramirez, et al., Hydroxytyrosol ameliorates oxidative stress and mitochondrial dysfunction in doxorubicin-induced cardiotoxicity in rats with breast cancer, Biochem. Pharmacol., 2014, 90(1), 25–33 CrossRef CAS PubMed.
- L. Zhu, Z. Liu, Z. Feng, J. Hao, W. Shen and X. Li, et al., Hydroxytyrosol protects against oxidative damage by simultaneous activation of mitochondrial biogenesis and phase II detoxifying enzyme systems in retinal pigment epithelial cells, J. Nutr. Biochem., 2010, 21(11), 1089–1098 CrossRef CAS PubMed.
- J. Hao, W. Shen, G. Yu, H. Jia, X. Li and Z. Feng, et al., Hydroxytyrosol promotes mitochondrial biogenesis and mitochondrial function in 3T3-L1 adipocytes, J. Nutr. Biochem., 2010, 21(7), 634–644 CrossRef CAS PubMed.
- E. Bali, V. Ergin, L. Rackova, O. Bayraktar, N. Küçükboyacı and Ç. Karasu, Olive Leaf Extracts Protect Cardiomyocytes against 4-Hydroxynonenal-Induced Toxicity In Vitro: Comparison with Oleuropein, Hydroxytyrosol, and Quercetin, Planta Med., 2014, 80(12), 984–992 CrossRef CAS PubMed.
- M. A. Martín, S. Ramos, A. B. Granado-Serrano, I. Rodríguez-Ramiro, M. Trujillo and L. Bravo, et al., Hydroxytyrosol induces antioxidant/detoxificant enzymes and Nrf2 translocation via extracellular regulated kinases and phosphatidylinositol-3-kinase/protein kinase B pathways in HepG2 cells, Mol. Nutr. Food Res., 2010, 54(7), 956–966 Search PubMed.
- M. Bakir, F. Geyikoglu, K. Koc and S. Cerig, Therapeutic effects of oleuropein on cisplatin-induced pancreas injury in rats, J. Cancer Res. Ther., 2018, 14(3), 671–678 CrossRef CAS PubMed.
- M. Alirezaei, A. Kheradmand, R. Heydari, N. Tanideh, S. Neamati and M. Rashidipour, Oleuropein protects against ethanol-induced oxidative stress and modulates sperm quality in the rat testis, Med. J. Nutr. Metab., 2012, 5(3), 205–211 Search PubMed.
- F. Geyikoğlu, S. Çolak, H. Türkez, M. Bakır, K. Koç and M. K. Hosseinigouzdagani, et al., Oleuropein Ameliorates Cisplatin-induced Hematological Damages Via Restraining Oxidative Stress and DNA Injury, Indian J. Hematol. Blood Transfus., 2017, 33(3), 348–354 CrossRef PubMed.
- F. Geyikoglu, H. Isikgoz, H. Onalan, S. Colak, S. Cerig and M. Bakir, et al., Impact of high-dose oleuropein on cisplatin-induced oxidative stress, genotoxicity and pathological changes in rat stomach and lung, J. Asian Nat. Prod. Res., 2017, 19(12), 1214–1231 CrossRef CAS PubMed.
- F. Geyikoglu, M. Emir, S. Colak, K. Koc, H. Turkez and M. Bakir, et al., Effect of oleuropein against chemotherapy drug-induced histological changes, oxidative stress, and DNA damages in rat kidney injury, J. Food Drug Anal., 2017, 25(2), 447–459 CrossRef CAS PubMed.
- M. Tabernero, B. Sarriá, C. Largo, S. Martínez-López, A. Madrona and J. L. Espartero, et al., Comparative evaluation of the metabolic effects of hydroxytyrosol and its lipophilic derivatives (hydroxytyrosyl acetate and ethyl hydroxytyrosyl ether) in hypercholesterolemic rats, Food Funct., 2014, 5(7), 1556–1563 Search PubMed.
- I. Fki, Z. Sahnoun and S. Sayadi, Hypocholesterolemic Effects of Phenolic Extracts and Purified Hydroxytyrosol Recovered from Olive Mill Wastewater in Rats Fed a Cholesterol-Rich Diet, J. Agric. Food Chem., 2007, 55(3), 624–631 CrossRef CAS PubMed.
- F. Visioli, G. Bellomo, G. Montedoro and C. Galli, Low density lipoprotein oxidation is inhibited in vitro by olive oil constituents, Atherosclerosis, 1995, 117(1), 25–32 Search PubMed.
- M. Fitó, M. I. Covas, R. M. Lamuela-Raventós, J. Vila, J. Torrents and C. de la Torre, et al., Protective effect of olive oil and its phenolic compounds against low density lipoprotein oxidation, Lipids, 2000, 35(6), 633–638 Search PubMed.
- K. de la Torre-Carbot, J. L. Chávez-Servín, O. Jaúregui, A. I. Castellote, R. M. Lamuela-Raventós and M. Fitó, et al., Presence of virgin olive oil phenolic metabolites in human low density lipoprotein fraction: Determination by high-performance liquid chromatography–electrospray ionization tandem mass spectrometry, Anal. Chim. Acta, 2007, 583(2), 402–410 Search PubMed.
- M. González-Santiago, E. Martín-Bautista, J. J. Carrero, J. Fonollá, L. Baró and M. V. Bartolomé, et al., One-month administration of hydroxytyrosol, a phenolic antioxidant present in olive oil, to hyperlipemic rabbits improves blood lipid profile, antioxidant status and reduces atherosclerosis development, Atherosclerosis, 2006, 188(1), 35–42 CrossRef PubMed.
- M. Mackness, S. Arrol, C. Abbott and P. Durrington, Protection of low-density lipoprotein against oxidative modification by high-density lipoprotein associated paraoxonase, Atherosclerosis, 1993, 104(1–2), 129–135 Search PubMed.
- M. I. Mackness and P. N. Durrington, HDL, its enzymes and its potential to influence lipid peroxidation, Atherosclerosis, 1995, 115(2), 243–253 Search PubMed.
- R. Carnevale, P. Pignatelli, C. Nocella, L. Loffredo, D. Pastori and T. Vicario, et al., Extra virgin olive oil blunt post-prandial oxidative stress via NOX2 down-regulation, Atherosclerosis, 2014, 235(2), 649–658 CrossRef CAS PubMed.
- J. P. Cooke, Asymmetrical Dimethylarginine, Circulation, 2004, 109(15), 1813–1818 Search PubMed.
- S. J. Rietjens, A. Bast, J. de Vente and G. R. M. M. Haenen, The olive oil antioxidant hydroxytyrosol efficiently protects against the oxidative stress-induced impairment of the NO•response of isolated rat aorta, Am. J. Physiol.: Heart Circ. Physiol., 2007, 292(4), H1931–H1936 CrossRef CAS PubMed.
- M. Deiana, A. Incani, A. Rosa, A. Atzeri, D. Loru and B. Cabboi, et al., Hydroxytyrosol glucuronides protect renal tubular epithelial cells against H2O2 induced oxidative damage, Chem.-Biol. Interact., 2011, 193(3), 232–239 Search PubMed.
- F. Paiva-Martins, A. Silva, V. Almeida, M. Carvalheira, C. Serra and J. E. Rodrígues-Borges, et al., Protective Activity of Hydroxytyrosol Metabolites on Erythrocyte Oxidative-Induced Hemolysis, J. Agric. Food Chem., 2013, 61(27), 6636–6642 Search PubMed.
- Y. O′Dowd, F. Driss, P. M. C. Dang, C. Elbim, M. A. Gougerot-Pocidalo and C. Pasquier, et al., Antioxidant effect of hydroxytyrosol, a polyphenol from olive oil: scavenging of hydrogen peroxide but not superoxide anion produced by human neutrophils, Biochem. Pharmacol., 2004, 68(10), 2003–2008 CrossRef PubMed.
- C. Manna, D. Napoli, G. Cacciapuoti, M. Porcelli and V. Zappia, Olive Oil Phenolic Compounds Inhibit Homocysteine-Induced Endothelial Cell Adhesion Regardless of Their Different Antioxidant Activity, J. Agric. Food Chem., 2009, 57(9), 3478–3482 CrossRef CAS PubMed.
- A. Mahmoudi, H. Ghorbel, Z. Bouallegui, R. Marrekchi, H. Isoda and S. Sayadi, Oleuropein and hydroxytyrosol protect from bisphenol A effects in livers and kidneys of lactating mother rats and their pups’, Exp. Toxicol. Pathol., 2015, 67(7–8), 413–425 CrossRef CAS PubMed.
- R. Fuccelli, P. Rosignoli, M. Servili, G. Veneziani, A. Taticchi and R. Fabiani, Genotoxicity of heterocyclic amines (HCAs) on freshly isolated human peripheral blood mononuclear cells (PBMC) and prevention by phenolic extracts derived from olive, olive oil and olive leaves, Food Chem. Toxicol., 2018, 122, 234–241 CrossRef CAS PubMed.
- F. Hadrich, M. Garcia, A. Maalej, M. Moldes, H. Isoda and B. Feve, et al., Oleuropein activated AMPK and induced insulin sensitivity in C2C12 muscle cells, Life Sci., 2016, 151, 167–173 CrossRef CAS PubMed.
- A. Cumaoğlu, L. Rackova, M. Stefek, M. Kartal, P. Maechler and C. Karasu, Effects of olive leaf polyphenols against H2O2 toxicity in insulin secreting β-cells, Acta Biochim. Pol., 2011, 58(1), 45–50 CrossRef.
- H. Kaneto, T.-a. Matsuoka, N. Katakami, D. Kawamori, T. Miyatsuka and K. Yoshiuchi, et al., Oxidative Stress and the JNK Pathway are Involved in the Development of Type 1 and Type 2 Diabetes, Curr. Mol. Med., 2007, 7(7), 674–686 CrossRef CAS PubMed.
- A. Khalili, A. A. Nekooeian and M. B. Khosravi, Oleuropein improves glucose tolerance and lipid profile in rats with simultaneous renovascular hypertension and type 2 diabetes, J. Asian Nat. Prod. Res., 2017, 19(10), 1011–1021 CrossRef CAS PubMed.
- P. Priore, L. Siculella and G. V. Gnoni, Extra virgin olive oil phenols down-regulate lipid synthesis in primary-cultured rat-hepatocytes, J. Nutr. Biochem., 2014, 25(7), 683–691 CrossRef CAS PubMed.
- R. Santiago-Mora, A. Casado-Díaz, M. D. De Castro and J. M. Quesada-Gómez, Oleuropein enhances osteoblastogenesis and inhibits adipogenesis: the effect on differentiation in stem cells derived from bone marrow, Osteoporosis Int., 2011, 22(2), 675–684 CrossRef CAS PubMed.
- R. Drira, S. Chen and K. Sakamoto, Oleuropein and hydroxytyrosol inhibit adipocyte differentiation in 3 T3-L1 cells, Life Sci., 2011, 89(19–20), 708–716 CrossRef CAS PubMed.
- M. Svobodova, I. Andreadou, A. L. Skaltsounis, J. Kopecky and P. Flachs, Oleuropein as an inhibitor of peroxisome proliferator-activated receptor gamma, Genes Nutr., 2014, 9(1), 376 CrossRef PubMed.
- C. Pirozzi, A. Lama, R. Simeoli, O. Paciello, T. B. Pagano and M. P. Mollica, et al., Hydroxytyrosol prevents metabolic impairment reducing hepatic inflammation and restoring duodenal integrity in a rat model of NAFLD, J. Nutr. Biochem., 2016, 30, 108–115 CrossRef CAS PubMed.
- S. Franceschelli, F. De Cecco, M. Pesce, P. Ripari, M. T. Guagnano and A. B. Nuevo, et al., Hydroxytyrosol Reduces Foam Cell Formation and Endothelial Inflammation Regulating the PPARγ/LXRα/ABCA1 Pathway, Int. J. Mol. Sci., 2023, 24(3), 2057 CrossRef CAS PubMed.
- S. Acín, M. A. Navarro, J. M. Arbonés-Mainar, N. Guillén, A. J. Sarría and R. Carnicer, et al., Hydroxytyrosol Administration Enhances Atherosclerotic Lesion Development in Apo E Deficient Mice, J. Biochem., 2006, 140(3), 383–391 CrossRef PubMed.
- X. Zhang, Y. Qin, X. Wan, H. Liu, C. Iv and W. Ruan, et al., Hydroxytyrosol Plays Antiatherosclerotic Effects through Regulating Lipid Metabolism via Inhibiting the p38 Signal Pathway, BioMed Res. Int., 2020, 2020(1), 5036572 CrossRef PubMed.
- M. Farràs, R. M. Valls, S. Fernández-Castillejo, M. Giralt, R. Solà and I. Subirana, et al., Olive oil polyphenols enhance the expression of cholesterol efflux related genes in vivo in humans. A randomized controlled trial, J. Nutr. Biochem., 2013, 24(7), 1334–1339 CrossRef PubMed.
- I. P. Salt and T. M. Palmer, Exploiting the anti-inflammatory effects of AMP-activated protein kinase activation, Expert Opin. Invest. Drugs, 2012, 21(8), 1155–1167 CrossRef CAS PubMed.
- G. Serra, M. Deiana, J. P. E. Spencer and G. Corona, Olive Oil Phenolics Prevent Oxysterol–Induced Proinflammatory Cytokine Secretion and Reactive Oxygen Species Production in Human Peripheral Blood Mononuclear Cells, Through Modulation of p38 and JNK Pathways, Mol. Nutr. Food Res., 2017, 61(12), 1700283 CrossRef PubMed.
- Y. Yonezawa, T. Miyashita, H. Nejishima, Y. Takeda, K. Imai and H. Ogawa, Anti-inflammatory effects of olive-derived hydroxytyrosol on lipopolysaccharide-induced inflammation in RAW264.7 cells, J. Vet. Med. Sci., 2018, 80(12), 1801–1807 CrossRef CAS PubMed.
- Y. B. Yu, H. Z. Zhuang, X. J. Ji, L. Dong and M. L. Duan, Hydroxytyrosol suppresses LPS-induced intrahepatic inflammatory responses via inhibition of ERK signaling pathway activation in acute liver injury, Eur. Rev. Med. Pharmacol. Sci., 2020, 24(11), 6455–6462 Search PubMed.
- Y. H. Kim, Y. J. Choi, M. K. Kang, E. J. Lee, D. Y. Kim and H. Oh, et al., Oleuropein Curtails Pulmonary Inflammation and Tissue Destruction in Models of Experimental Asthma and Emphysema, J. Agric. Food Chem., 2018, 66(29), 7643–7654 CrossRef CAS PubMed.
- S. Al-Quraishy, M. S. Othman, M. A. Dkhil and A. E. Abdel Moneim, Olive (Olea europaea) leaf methanolic extract prevents HCl/ethanol-induced gastritis in rats by attenuating inflammation and augmenting antioxidant enzyme activities, Biomed. Pharmacother., 2017, 91, 338–349 CrossRef CAS PubMed.
- R. de la Puerta, V. R. Gutierrez and J. R. S. Hoult, Inhibition of leukocyte 5-lipoxygenase by phenolics from virgin olive oil, Biochem. Pharmacol., 1999, 57(4), 445–449 CrossRef CAS PubMed.
- E. Scoditti, N. Calabriso, M. Massaro, M. Pellegrino, C. Storelli and G. Martines, et al., Mediterranean diet polyphenols reduce inflammatory angiogenesis through MMP-9 and COX-2 inhibition in human vascular endothelial cells: A potentially protective mechanism in atherosclerotic vascular disease and cancer, Arch. Biochem. Biophys., 2012, 527(2), 81–89 CrossRef CAS PubMed.
- B. Menicacci, C. Cipriani, F. Margheri, A. Mocali and L. Giovannelli, Modulation of the Senescence-Associated Inflammatory Phenotype in Human Fibroblasts by Olive Phenols, Int. J. Mol. Sci., 2017, 18(11), 2275 CrossRef PubMed.
- M. Aparicio-Soto, D. Redhu, M. Sánchez-Hidalgo, J. G. Fernández-Bolaños, C. Alarcón-de-la-Lastra and M. Worm, et al., Olive–Oil–Derived Polyphenols Effectively Attenuate Inflammatory Responses of Human Keratinocytes by Interfering with the NF–κB Pathway, Mol. Nutr. Food Res., 2019, 63(21), 1900019 CrossRef CAS PubMed.
- C. Chen, L. Chen, J. Zhou, R. Cai, Z. Ye and D. Zhang, Anti-psoriasis activities of hydroxytyrosol on HaCaT cells under psoriatic inflammation in vitro, Immunopharmacol. Immunotoxicol., 2023, 45(3), 328–333 CrossRef CAS PubMed.
- R. Santarelli, C. Pompili, M. S. Gilardini Montani, L. Evangelista, R. Gonnella and M. Cirone, 3,4-Dihydroxyphenylethanol (DPE or Hydroxytyrosol) Counteracts ERK1/2 and mTOR Activation, Pro-Inflammatory Cytokine Release, Autophagy and Mitophagy Reduction Mediated by Benzo[a]pyrene in Primary Human Colonic Epithelial Cells, Pharmaceutics, 2022, 14(3), 663 CrossRef CAS PubMed.
- M. Aparicio-Soto, M. Sánchez-Hidalgo, A. Cárdeno, A. González-Benjumea, J. G. Fernández-Bolaños and C. Alarcón-de-la-Lastra, Dietary hydroxytyrosol and hydroxytyrosyl acetate supplementation prevent pristane-induced systemic lupus erythematous in mice, J. Funct. Foods, 2017, 29, 84–92 CrossRef CAS.
- Q. Wang, C. Wang, Abdullah, W. Tian, Z. Qiu and M. Song, et al., Hydroxytyrosol Alleviates Dextran Sulfate Sodium-Induced Colitis by Modulating Inflammatory Responses, Intestinal Barrier, and Microbiome, J. Agric. Food Chem., 2022, 70(7), 2241–2252 CrossRef CAS PubMed.
- F. Miao, Hydroxytyrosol alleviates dextran sodium sulfate–induced colitis by inhibiting NLRP3 inflammasome activation and modulating gut microbiota in vivo, Nutrition, 2022, 97, 111579 CrossRef CAS PubMed.
- B. Lee, I. Shim, H. Lee and D. H. Hahm, Effect of oleuropein on cognitive deficits and changes in hippocampal brain-derived neurotrophic factor and cytokine expression in a rat model of post-traumatic stress disorder, J. Nat. Med., 2018, 72(1), 44–56 CrossRef CAS PubMed.
- M. Dell'Agli, R. Fagnani, N. Mitro, S. Scurati, M. Masciadri and L. Mussoni, et al., Minor Components of Olive Oil Modulate Proatherogenic Adhesion Molecules Involved in Endothelial Activation, J. Agric. Food Chem., 2006, 54(9), 3259–3264 CrossRef PubMed.
- M. A. Carluccio, L. Siculella, M. A. Ancora, M. Massaro, E. Scoditti and C. Storelli, et al., Olive Oil and Red Wine Antioxidant Polyphenols Inhibit Endothelial Activation, Arterioscler., Thromb., Vasc. Biol., 2003, 23(4), 622–629 CrossRef CAS PubMed.
- J. Salas-Salvadó, A. Garcia-Arellano, R. Estruch, F. Marquez-Sandoval, D. Corella and M. Fiol, et al., Components of the mediterranean-type food pattern and serum inflammatory markers among patients at high risk for cardiovascular disease, Eur. J. Clin. Nutr., 2008, 62(5), 651–659 CrossRef PubMed.
- I. Singh, M. Mok, A. M. Christensen, A. H. Turner and J. A. Hawley, The effects of polyphenols in olive leaves on platelet function, Nutr. Metab. Cardiovasc. Dis., 2008, 18(2), 127–132 CrossRef CAS PubMed.
- M. E. Katsa and T. Nomikos, Olive Oil Phenolics and Platelets—From Molecular Mechanisms to Human Studies, Rev. Cardiovasc. Med., 2022, 23(8), 255 CrossRef PubMed.
- M. Deiana, G. Serra and G. Corona, Modulation of intestinal epithelium homeostasis by extra virgin olive oil phenolic compounds, Food Funct., 2018, 9(8), 4085–4099 RSC.
- I. Prieto, M. Hidalgo, A. B. Segarra, A. M. Martínez-Rodríguez, A. Cobo and M. Ramírez, et al., Influence of a diet enriched with virgin olive oil or butter on mouse gut microbiota and its correlation to physiological and biochemical parameters related to metabolic syndrome, PLoS One, 2018, 13(1), e0190368 CrossRef PubMed , Gonzalez-Bulnes A, editor.
- Z. Liu, N. Wang, Y. Ma and D. Wen, Hydroxytyrosol Improves Obesity and Insulin Resistance by Modulating Gut Microbiota in High-Fat Diet-Induced Obese Mice, Front. Microbiol., 2019, 10 CAS.
- G. Marcelino, P. A. Hiane, K de C. Freitas, L. F. Santana, A. Pott and J. R. Donadon, et al., Effects of Olive Oil and Its Minor Components on Cardiovascular Diseases, Inflammation, and Gut Microbiota, Nutrients, 2019, 11(8), 1826 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |