Open Access Article
Open Access ArticleThe effect of hemp product consumption on blood fatty acid profiles and cardiovascular disease risk factors: results of a randomized, double-blind, crossover clinical trial
Yongbo
She†
 abc,
Kaizhen
Jia†
abc,
Kaizhen
Jia†
 de,
Adrianne
Cardillo
ab,
Maryam
Shamloo
ab,
Alejandra Serrano
León
ab,
Xin
Liu
e,
Rebecca
Mollard
af and
Dylan S.
Mackay
de,
Adrianne
Cardillo
ab,
Maryam
Shamloo
ab,
Alejandra Serrano
León
ab,
Xin
Liu
e,
Rebecca
Mollard
af and
Dylan S.
Mackay
 *abg
*abg
aRichardson Centre for Food Technology and Research, University of Manitoba, Winnipeg, Manitoba R3T 2E1, Canada. E-mail: dylan.mackay@umanitoba.ca
bDepartment of Food and Human Nutritional Sciences, University of Manitoba, Winnipeg, Manitoba R3T 6C5, Canada
cDivision of Food, Nutrition and Health, College of Food Science and Engineering, Northwest A&F University, Yangling, Shaanxi, China
dXi'an Center for Disease Control and Prevention, 599 Xiying Road, Xi'an, Shaanxi, China
eDepartment of Epidemiology and Biostatistics, School of Public Health, Global Health Institute, Xi'an Jiaotong University Health Science Center, Xi'an, Shaanxi, China
fChronic Disease Innovation Centre, Seven Oaks General Hospital, Winnipeg, Manitoba R2V 4W1, Canada
gDepartment of Internal Medicine (Endocrinology), Rady Faculty of Health Sciences, University of Manitoba, Winnipeg, Manitoba R3E 0T6, Canada
First published on 24th February 2026
Abstract
Hemp seeds are high in polyunsaturated fatty acids (PUFAs) including gamma linolenic acid (GLA), stearidonic acid (SDA), alpha linolenic acid (ALA) and linoleic acid (LA). To date, limited evidence is available on hemp product consumption and particularly hemp seeds and oil in humans and its relation to cardiometabolic risk factors. The objective of present study was to examine the effects of hemp product consumption versus similar controls on circulating fatty acid profiles and cardiovascular disease (CVD) risk factors. A randomized, double-blinded, crossover trial with 30 normoglycemic adults (18–65 years) within a BMI range of 25–35 kg m−2 were included. Participants consumed both hemp products and controlled products over the course of 4 weeks each. As expected, ALA (18:3 n-3), GLA (18:3 n-6) and dihomo-γ-linolenic acid (DGLA, 20:3 n-6) were elevated after the hemp treatment than controls. Similarly, ALA, DGLA as well as eicosapentaenoic acid (EPA) levels were elevated after the hemp treatment than controls. No differences in serum lipid levels, glucose and insulin concentrations, blood pressure, or body composition were observed between treatments. Overall, consumption of hemp products modulated plasma and RBC fatty acids levels in a way which reflected the fatty acids these products are enriched in, without showing differences in major cardiometabolic risk factors. The present study demonstrated the human fatty acids profile response to consuming hemp products, novel functional foods rich in polyunsaturated fatty acids. The clinical trial registry number is NCT02400203 (https://clinicaltrials.gov).
Introduction
Cannabis sativa L. is a herbaceous plant native to Central Asia and is broadly classified into drug-type (marijuana) and non-drug-type (hemp) varieties based on Δ9-tetrahydrocannabinol content.1 In recent years, hemp seeds and hemp-derived products have attracted increasing attention as functional food due to their distinctive and favorable fatty acid composition. Hemp seed oil is particularly rich in polyunsaturated fatty acids (PUFAs), providing substantial amounts of linoleic acid (LA, 18:2 n-6) and α-linolenic acid (ALA, 18:3 n-3), as well as smaller but physiologically relevant quantities of γ-linolenic acid (GLA, 18:3 n-6) and stearidonic acid (SDA, 18:4 n-3), fatty acids that are uncommon in most commonly consumed vegetable oils.2–4 In addition, hemp seed oil contains tocopherols and polyphenolic compounds with antioxidant potential, further supporting its emerging role as a cardioprotective dietary fat source.Cardiovascular disease (CVD) remains the leading cause of mortality worldwide, accounting for approximately one third of global deaths.5 Extensive epidemiological and clinical evidence demonstrates that dietary fat quality, rather than total fat intake alone, plays a critical role in modulating CVD risks. For instance, high dietary intake and circulating levels of LA and ALA are associated with improved cardiometabolic health and reduced risk of thrombotic and ischemic events,6,7 while SDA intake may further counteract some CVD risk factors through its efficient conversion to long-chain n-3 fatty acids.8 In contrast, diets characterized by high saturated fatty acid (SFA) content and low PUFA intake are consistently associated with increased CVD risk.9,10 Moreover, an imbalanced dietary n-6/n-3 PUFA ratio has been linked to adverse cardiometabolic outcomes, whereas increased n-3 PUFA intake has been shown to reduce the risk of sudden cardiac death.11 Beyond dietary intake, circulating fatty acid profiles measured in plasma as well as erythrocytes serve as promising biomarkers of fatty acid exposure, metabolism, and tissue incorporation, and are increasingly recognized as integrative indicators of cardiometabolic perturbations. Plasma fatty acids reflect more recent dietary intake, whereas erythrocyte fatty acid composition provides insight into longer-term fatty acid bioavailability and incorporation into cell membranes. Alterations in these fatty acid pools have been linked to established CVD risk factors, including dyslipidemia, hypertension, insulin resistance, and obesity based on findings from numerous human cohort studies and including those from our own group.12–16
Preclinical studies suggest that hemp seed and hemp oil consumption can beneficially modify fatty acid metabolism. Supplementation with hemp seed oil in maternal sows resulted in an altered n-6/n-3 PUFA profile with increased circulating ALA and SDA concentrations and EPA/DPA in piglets,17 while feeding hemp seeds to laying hens increased hepatic LA, ALA, GLA, arachidonic acid (AA), and docosahexaenoic acid (DHA) levels and decreased the n-6/n-3 ratio.18 Despite the growing popularity of hemp-based foods, well-controlled human intervention studies examining the effects of hemp product consumption on circulating fatty acid profiles and CVD risk factors remain limited. In particular, there is a lack of randomized clinical trials evaluating both plasma and erythrocyte fatty acid responses to hemp products in populations at elevated cardiometabolic risk.
Hence, the objective of the present randomized, double-blind, crossover clinical trial was to investigate the effects of hemp product consumption on circulating fatty acid profiles in plasma and erythrocytes, as well as on established cardiovascular disease risk factors, in adults with overweight or obesity. This study aimed to provide controlled human evidence to clarify the cardiometabolic relevance of hemp-derived fatty acids and to establish circulating fatty acid signatures associated with hemp product intake.
Methods
Study design
This study was a randomized, double-blinded, two period cross-over design. Each treatment was 28 days followed by a 4-week wash-out period. The primary outcome was to determine changes in plasma as well as erythrocytes fatty acids profile and the secondary outcomes were to determine the changes in major CVD risk factors including lipids profile, blood pressure and arterial thickness, and fasting glucose and insulin. BMI, waist circumference and body composition were also measured.Diet and treatment
The control treatment included 27 g of hulled sesame seeds with 3 g of lentil flakes incorporated to mimic the appearance of hulled hempseed. The control treatment salad dressings base oil was soybean oil. The hemp treatment included 30 g of hulled hempseed and the salad dressing base oil was hemp oil. Treatment products were produced by RCFTR staff outside the trial team and given to trial staff in a blinded fashion. Table 1 shows the recipe used for the control and the hemp treatment salad dressings. Hulled seeds were given in opaque sachets and salad dressings in plastic containers. Participants were required to consume two 30 g sachets of hulled seeds and a single 51.8 g serving of salad dressing daily, which contained 30 g of oil. Labelled cups were provided to ensure the proper amount was distributed daily. The salad dressing recipe was selected for flavor and color that would help mask any differences in treatment oils appearance and taste. During the participants’ weekly visits, they were asked if there were any issues arising from consuming the treatment products and if they needed any further assistance and/or advise in ways to incorporate the treatment products into their diets. The fatty acid profiles of the soybean and hempseed oil used in the salad were determined by using the direct transesterification method and processed on the gas chromatograph (GC) using flame ionization.19 Results of the fatty acid profile in the soybean and hempseed oils are presented in Table 2. The individual fatty acid concentration is calculated as peak area of the fatty acid divided by total area identified and expressed as g per 100 g. The sesame seeds and hempseed were sent to a third-party lab (Covance Lab. Madison, WI) to determine the macronutrient contents presented in Table 3.| Ingredients | Hemp | Control |
|---|---|---|
| The recipe is presented in daily portions. | ||
| Balsamic vinegar (g) | 11 | 11 |
| Chopped, dried parsley flakes (g) | 0.5 | 0.5 |
| Fresh lemon juice (g) | 6 | 6 |
| Chopped, fresh garlic cloves (g) | 2.5 | 2.5 |
| Crumbled, dried basil (g) | 0.5 | 0.5 |
| Dried oregano (g) | 0.5 | 0.5 |
| Salt (g) | 0.5 | 0.5 |
| Pepper (g) | 0.3 | 0.3 |
| Hempseed (g) | 30 | NA |
| Soybean oil (g) | NA | 30 |
| Total (g) | 51.8 | 51.8 |
| Hemp | Soy | |
|---|---|---|
| Fatty acid percentages are based on averages between two samples. ND = non-detectable. | ||
| SFA (%) | 9.93 | 15.4 |
| C16:0 | 6.5 | 10.8 |
| C18:0 | 1.96 | 3.78 |
| C20:0 | 0.86 | 0.32 |
| MUFA (%) | 9.61 | 23.9 |
| C16:1n-7 | 0.12 | 0.1 |
| C17:1n-7 | 0.02 | 0.06 |
| C18:1n-9 | 9.02 | 23.5 |
| C20:1n-11 | 0.43 | 0.25 |
| PUFA (%) | 79 | 60.7 |
| C18:2n-6 | 55.7 | 53 |
| C18:3n-6 | 4.53 | 0.561 |
| C18:3n-3 | 18.2 | 6.89 |
| C18:4n-3 | 1.5 | ND |
| C20:2n-6 | 0.07 | 0.03 |
| n-6 PUFA (%) | 60.93 | 53.6 |
| n-3 PUFA (%) | 19.7 | 6.89 |
n-6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) n-3 ratio n-3 ratio
|
3.09 | 7.7 |
| Hulled hempseed | Sesame seeds with lentil flakes | |
|---|---|---|
| The values are presented per 30 g of serving. | ||
| Calories (kcal) | 172 | 191 |
| Calories from fat (kcal) | 119 | 144 |
| Fat (g) | 13.3 | 16 |
| Total carbohydrates (g) | 3.54 | 4.2 |
| Total dietary fiber (g) | 0.78 | 2.45 |
| Protein (g) | 9.69 | 7.5 |
BMI was calculated using the participants’ weight and height measurements. An electric scale was used to measure body weight of the participants. Individuals removed shoes and heavy cloths prior to weighing. For height measurements individuals stood with their scapula, buttocks and heels against a wall, the neck was held in a natural non-stretched position and the head was held straight. Participants’ systolic and diastolic blood pressures were measured in triplicate, on the non-dominant arm in a sitting position using a validated oscillometric blood pressure monitor (BP760CAN, Omron, Burlington, Ontario, Canada). Participants were required to rest for 5–10 min before taking the measurement. Pulse wave velocity and augmentation index were measured on the non-dominant arm in a sitting position using a Mobil-O-Graph PWA Monitor and the HMS Client Server Software (IEM GmbH, Stolberg, Germany) according to the manufacturer's protocol. Body composition including body fat percentage, and visceral adipose tissue mass were assessed using DEXA (Lunar Prodigy Advance, GE Healthcare, Mississauga, Ontario, Canada). After day 1, the participants were required to drop in weekly to pick up the treatments for the following week, which continued during both study periods. During the washout period, participants were asked to follow the same n-3 fatty acids food restrictions discussed prior to commencing the study and asked to maintain regular dietary habits.
Plasma insulin, lipid, and glucose analysis. Plasma insulin was measured by radioimmunoassay (RIA) in duplicate using Millipore Human Insulin Specific RIA Kits (125 I-Insulin). Plasma total cholesterol (TC), HDL cholesterol, triglycerides, and glucose concentrations were determined by automated enzymatic methods on a Vitros-350 chemistry analyzer (ortho-Clinical Diagnostics). LDL-cholesterol concentrations were calculated by the Friedewald equation.20
Fatty acid profile analysis. Plasma and RBC fatty acids were extracted and methylated using a direct transesterification method.19 Briefly, 1.6 ml methanol was initially added into the sample (500 μl plasma or 0.5 g RBC) followed by adding 75 μl of 1 mg ml−1 methyl cis-10-heptadecenoate (Sigma Aldrich, USA) as the internal standard. Then, adding 400 μl toluene and 200 μl acetyl chloride into the sample while mixed on a vortex. A ten-second nitrogen flash was also applied to each sample followed by incubation at 80 °C for 1 hour. After cooling to room temperature, 5 ml of 6% K2CO3 was added into the tube and followed by centrifugation at 2500 rpm for 5 minutes. The top layer was transferred into the GC vial and stored at −80 °C for further analysis.
Methylated fatty acid samples were analyzed by gas chromatography (GC, Varian 430) by using a 30 m × 0.25 mm column (Agilent Technologies, Inc). Fatty acids in samples were identified based on the retention time of internal standard and a mix of standard known fatty acids. Concentrations of each fatty acid were expressed as percentage of total identified fatty acids. The RBC long chain n-3 fatty acid content was determined by combining the amount of EPA, DPA, and DHA present in the RBC.
Statistical analysis
Sample size was determined using a power calculation performed using PROC POWER (SAS 9.2, SAS Inc). Estimated changes in erythrocyte EPA fatty acid was selected as primary outcome and LDL-C was selected as secondary outcome based on our previous study,21 a trial involving ALA supplementation from flax oil, a n = 30 had a power = 0.996 to detect a difference in EPA fatty acids of 0.56% and power = 0.611 to detect a difference of 0.22 mmol L−1 in LDL-C, with α = 0.05 and correlation factor of repeated measures set at 0.8 using a 2-sided test. The estimated changes in fatty acids and LDL-C were taken as half the effect of the canola/flax treatment which provided a dose of 20 g d−1 ALA (the current trial would delivered 12.27 g d−1 ALA and 0.56 g d−1 SDA). The highest standard deviations for the fatty acid and cholesterol treatments from the previous study were selected as the estimates of variance for the power calculation. This sample size also provided moderate power (power = 0.611) to detect a clinically relevant difference of 0.22 mmol L−1 in LDL-C as a secondary outcome. To account for potential attrition, 37 participants were enrolled, resulting in 30 participants completing the study, which met the predefined sample size requirement for the primary outcome.The outcomes are reported as means ± standard error (SE). Effects of dietary treatments compared the endpoint outcome value of each treatment period using a mixed model analysis of variance procedure (PROC MIXED, SAS 9.2, SAS Inc), with repeat measures. Treatment, sex, sequence, treatment × sex, treatment × sequence were assigned as fixed factors and participant ID was a repeated factor. Normality of data was observed and checked visually based on the plot of residuals. Statistical significance was set at P < 0.05 for all outcome measurements.
Results
Participant characteristics
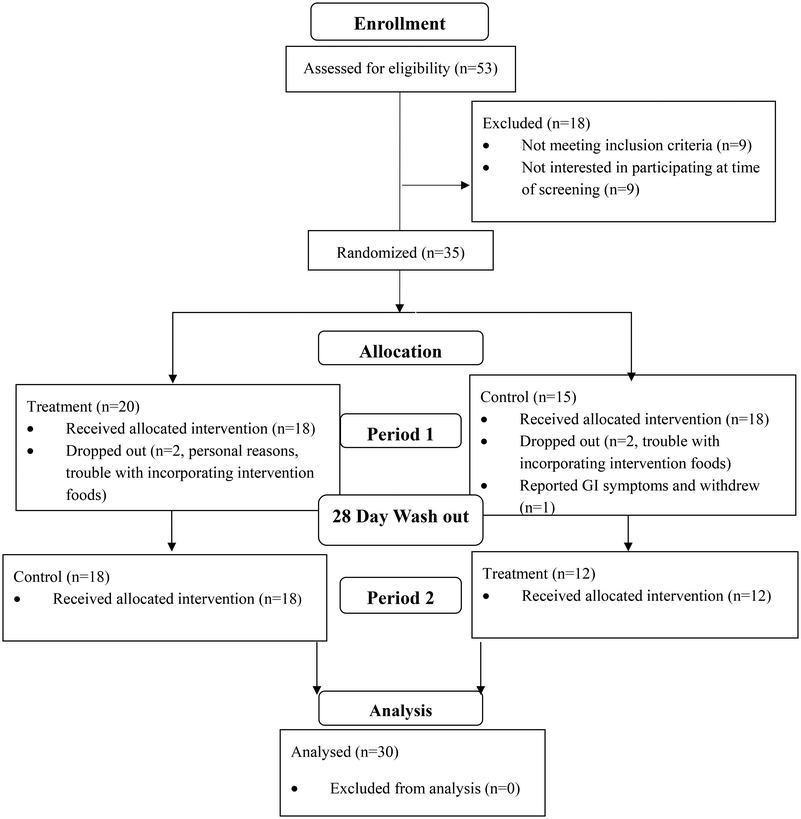
Baseline demographic characteristics of participants are presented in Table 4. The trial procedures are outlined in Fig. 1. Overall, 37 participants were enrolled in the study and 30 participants completed with 13 males and 17 females. A total of seven participants dropped out of the study. Four participants had trouble incorporating the treatments into their daily diet; one participant was worried about the increase in fat intake from the intervention products: one participant had to leave the study for personal reasons, and one participant reported gastrointestinal symptoms related to the salad dressing during the control treatment period.| Characteristic | Mean |
|---|---|
| Values are expressed as mean ± SD. | |
| Age (year) | 49.23 ± 14.52 |
| Weight (kg) | 84.27 ± 12.92 |
| BMI (kg m−2) | 28.94 ± 2.9 |
| Waist circumference (cm) | 97.97 ± 12.8 |
| Systolic pressure (mmHg) | 120.37 ± 16.28 |
| Diastolic pressure (mmHg) | 78.60 ± 9.48 |
| Glucose (mmol L−1) | 5.43 ± 0.56 |
| Cholesterol (mmol L−1) | 5.14 ± 1.08 |
| Triglycerides (mmol L−1) | 1.31 ± 0.65 |
| dHDL (mmol L−1) | 1.41 ± 0.33 |
| LDL (mmol L−1) | 3.14 ± 0.95 |
Plasma and RBC fatty acids profile
Results for plasma fatty acid concentrations is presented in Table 5. There were no differences in total SFA and monosaturated fatty acids (MUFA) between hemp and control treatments. As expected, total PUFA was increased after hemp treatment compared to the control (P = 0.01). Specifically, GLA (C18:3n-6), ALA (C18:3n-3), DGLA (C20:3n-6) concentrations were found to be higher in hemp treatment than in control (all P < 0.05). The results of the RBC fatty acid concentrations are presented in Table 6. Similar to what we observed in plasma, there were no major differences in SFA, MUFA between the treatments. In terms of individual PUFAs, ALA (C18:3n-3, P = 0.01), EPA (C20:5n-3, P = 0.02) as well as DGLA (C20:3n-6, P < 0.05) concentrations were found to be higher in hemp treatment than in control.| Measure | Hemp | Control | P-Value |
|---|---|---|---|
| Values are expressed as mean ± SE. | |||
| SFA (%) | 43.73 ± 0.84 | 45.14 ± 0.84 | 0.21 |
| C10:0 | 0.11 ± 0.04 | 0.06 ± 0.04 | 0.4 |
| C12:0 | 0.07 ± 0.01 | 0.08 ± 0.01 | 0.43 |
| C14:0 | 0.52 ± 0.03 | 0.54 ± 0.03 | 0.65 |
| C15:0 | 0.22 ± 0.01 | 0.21 ± 0.01 | 0.31 |
| C16:0 | 28.85 ± 0.82 | 30.6 ± 0.82 | 0.09 |
| C18:0 | 13.78 ± 0.31 | 13.52 ± 0.31 | 0.48 |
| C20:0 | 0.07 ± 0.01 | 0.06 ± 0.01 | 0.18 |
| MUFA (%) | 14.61 ± 0.96 | 14.46 ± 0.96 | 0.9 |
| C14:1n-9 | 0.08 ± 0.03 | 0.04 ± 0.03 | 0.38 |
| C16:1n-7 | 1.92 ± 0.77 | 0.88 ± 0.77 | 0.35 |
| C17:1n-7 | 0.06 ± 0 | 0.06 ± 0 | 0.36 |
| C18:1n-9 | 12.42 ± 0.42 | 13.31 ± 0.42 | 0.08 |
| C20:1n-11 | 0.11 ± 0.07 | 0.19 ± 0.07 | 0.45 |
| C22:1n-13 | 0.7 ± 0.06 | 0.56 ± 0.06 | 0.08 |
| PUFA (%) | 41.79 ± 0.45 | 40.48 ± 0.45 | 0.01 |
| C18:2n-6 | 24.59 ± 0.46 | 24.63 ± 0.46 | 0.95 |
| C18:3n-6 | 0.2 ± 0.02 | 0.08 ± 0.02 | <0.05 |
| C18:3n-3 | 0.7 ± 0.05 | 0.49 ± 0.05 | <0.05 |
| C18:4n-3 | 0.05 ± 0 | 0.05 ± 0 | 0.41 |
| C20:2n-6 | 0.19 ± 0.01 | 0.2 ± 0.01 | 0.81 |
| C20:3n-6 | 3.26 ± 0.13 | 2.46 ± 0.13 | <0.05 |
| C20:4n-6 | 9.22 ± 0.29 | 8.74 ± 0.29 | 0.06 |
| C20:5n-3 | 0.34 ± 0.32 | 0.18 ± 0.3 | 0.76 |
| C22:4n-6 | 0.16 ± 0.02 | 0.2 ± 0.02 | 0.08 |
| C22:5n-3 | 0.61 ± 0.05 | 0.61 ± 0.05 | 0.98 |
| C22:6n-3 | 2.16 ± 0.13 | 2.38 ± 0.13 | 0.17 |
| n-6 PUFA (%) | 37.62 ± 0.44 | 36.52 ± 0.44 | 0.03 |
| n-3 PUFA (%) | 4.17 ± 0.19 | 3.96 ± 0.19 | 0.39 |
n-6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) n-3 ratio n-3 ratio |
10.68 ± 1.39 | 10.26 ± 1.39 | 0.84 |
| Measure | Hemp | Control | P-Value |
|---|---|---|---|
| Values are expressed as mean ± SE. | |||
| SFA (%) | 42.6 ± 0.29 | 42.9 ± 0.29 | 0.5 |
| C10:0 | 0.03 ± 0.01 | 0.05 ± 0.01 | 0.53 |
| C12:0 | 0.07 ± 0.04 | 0.02 ± 0.04 | 0.35 |
| C14:0 | 0.37 ± 0.07 | 0.42 ± 0.07 | 0.64 |
| C15:0 | 0.27 ± 0.03 | 0.35 ± 0.03 | 0.01 |
| C16:0 | 23.7 ± 0.23 | 23.9 ± 0.23 | 0.69 |
| C18:0 | 16.1 ± 0.23 | 16 ± 0.23 | 0.57 |
| C20:0 | 0.18 ± 0.02 | 0.2 ± 0.02 | 0.41 |
| C22:0 | 0.8 ± 0.05 | 0.79 ± 0.05 | 0.81 |
| C24:0 | 1.04 ± 0.09 | 1.28 ± 0.09 | 0.01 |
| MUFA (%) | 16.6 ± 0.27 | 17 ± 0.27 | 0.19 |
| C14:1n-9 | 0.12 ± 0.02 | 0.1 ± 0.02 | 0.4 |
| C16:1n-7 | 0.78 ± 0.08 | 0.74 ± 0.08 | 0.72 |
| C17:1n-7 | 0.5 ± 0.06 | 0.5 ± 0.06 | 0.98 |
| C18:1n-9 | 12.8 ± 0.17 | 12.9 ± 0.17 | 0.35 |
| C18:1n-7 | 1.17 ± 0.04 | 1.16 ± 0.04 | 0.76 |
| C22:1n-13 | 0.03 ± 0 | 0.03 ± 0 | 0.96 |
| PUFA (%) | 40.8 ± 0.3 | 40 ± 0.3 | 0.08 |
| C18:2n-6 | 12.3 ± 0.21 | 12.5 ± 0.21 | 0.28 |
| C18:3n-6 | 0.09 ± 0.01 | 0.07 ± 0.01 | 0.17 |
| C18:3n-3 | 0.36 ± 0.02 | 0.27 ± 0.02 | 0.01 |
| C18:4n-3 | 0.59 ± 0.07 | 0.55 ± 0.07 | 0.62 |
| C20:2n-6 | 0.23 ± 0.01 | 0.23 ± 0.01 | 0.83 |
| C20:3n-6 | 1.83 ± 0.06 | 1.59 ± 0.06 | <0.05 |
| C20:4n-6 | 14.9 ± 0.27 | 14.4 ± 0.27 | 0.11 |
| C20:5n-3 | 0.78 ± 0.05 | 0.7 ± 0.05 | 0.02 |
| C22:4n-6 | 2.94 ± 0.09 | 2.8 ± 0.09 | 0.12 |
| C22:5n-6 | 0.38 ± 0.02 | 0.399 ± 0.02 | 0.38 |
| C22:5n-3 | 2.47 ± 0.1 | 2.36 ± 0.1 | 0.13 |
| C22:6n-3 | 3.81 ± 0.18 | 3.99 ± 0.18 | 0.26 |
| n-6 PUFA (%) | 32.7 ± 0.34 | 32 ± 0.34 | 0.09 |
| n-3 PUFA (%) | 8.08 ± 0.21 | 8.01 ± 0.21 | 0.78 |
n-6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) n-3 ratio n-3 ratio |
4.13 ± 0.13 | 4.09 ± 0.13 | 0.77 |
Blood pressure, arterial stiffness and DEXA measures
Blood pressure measurement were not different between the hemp and the control treatments for either systolic or diastolic blood pressure. The PWV and the augmentation index showed no differences after consumption of hemp treatment compared to control. No differences were seen in visceral fat, body fat percentage, or BMI across treatments. Results are presented in Table 7.| Measures | Hemp | Soy | P-Value |
|---|---|---|---|
| Values are expressed as mean ± SE. | |||
| BMI (kg m−2) | 29.29 ± 0.54 | 29.07 ± 0.54 | 0.13 |
| Systolic blood pressure (mmHg) | 120.77 ± 3.16 | 124.51 ± 3.16 | 0.28 |
| Diastolic blood pressure (mmHg) | 80.29 ± 1.62 | 80.2 ± 1.62 | 0.95 |
| Augmentation index (%) | 17.87 ± 2.68 | 14.95 ± 2.69 | 0.25 |
| Pulse wave velocity (m s−1) | 9.33 ± 1.57 | 7.29 ± 1.58 | 0.38 |
| Visceral fat (lbs) | 2.94 ± 0.28 | 2.87 ± 0.28 | 0.57 |
| Body fat (%) | 36.5 ± 0.92 | 36.2 ± 0.92 | 0.31 |
Lipids, glucose, and insulin concentrations
The fasting glucose, insulin, and lipid concentrations are presented in Table 8. No differences were seen between hemp and control treatments in total cholesterol, triglyceride, HDL-C, or LDL-C concentrations. There were no changes in glucose and insulin concentrations measured across periods.| Hemp | Soy | P-Value | |
|---|---|---|---|
| Values are expressed as mean ± SE. | |||
| Glucose (mmol L−1) | 5.4 ± 0.11 | 5.31 ± 0.11 | 0.42 |
| Fasting insulin (µU ml−1) | 17.83 ± 1.36 | 18.1 ± 1.36 | 0.78 |
| Total cholesterol (mmol L−1) | 5.03 ± 0.2 | 5.13 ± 0.2 | 0.24 |
| Triglyceride (mmol L−1) | 1.27 ± 0.14 | 1.16 ± 0.14 | 0.15 |
| HDL (mmol L−1) | 1.39 ± 0.05 | 1.43 ± 0.05 | 0.06 |
| LDL (mmol L−1) | 3.06 ± 0.17 | 3.17 ± 0.17 | 0.11 |
Discussion
This randomized, double-blind, crossover clinical trial investigated the effects of hemp product, specifically the hemp seeds and hemp oil consumption on circulating fatty acid profiles and major cardiovascular disease (CVD) risk factors in adults with overweight or obesity. The principal finding of this study is that daily consumption of hulled hempseed and hempseed oil for four weeks significantly increased the relative abundance of some beneficial fatty acids, namely ALA, GLA, DGLA, and EPA in plasma and erythrocyte membranes respectively, without adversely modulating major cardiometabolic risk markers, including blood lipids, blood pressure, arterial stiffness and/or body composition.The observed enrichment of plasma as well as RBC ALA, GLA, DGLA, and EPA following hemp product consumption indicates effective absorption, metabolism, and incorporation of hemp-derived polyunsaturated fatty acids into cell membranes. Erythrocyte fatty acid composition reflects longer-term dietary exposure and tissue bioavailability rather than short-term intake,22,23 these findings provide robust evidence that hemp products can favorably modify systemic fatty acid pool. In particular, the increase in EPA is noteworthy, as EPA has well-established anti-inflammatory and cardioprotective properties and has been associated with reduced obesity-related metabolic dysfunction and cardiovascular mortality.8,24 In our present study, the elevation of EPA observed is likely attributable to, at least in part, the unique fatty acid composition of hemp products, characterized by relatively high ALA and GLA content and a lower LA/ALA ratio compared with many conventional vegetable oils. This fatty acid milieu may facilitate endogenous conversion of ALA to EPA by reducing competitive inhibition from excessive linoleic acid.25,26 In contrast, DHA levels remained unchanged, which is consistent with prior evidence demonstrating limited conversion of ALA to DHA in humans, particularly over short intervention periods. DGLA and EPA are known precursors to anti-inflammatory eicosanoids and series-1 prostaglandins, which are implicated in improved vascular function and reduced inflammatory tone.27 Elevated circulating or membrane-bound DGLA has been associated with lower triglyceride concentrations and reduced cardiovascular mortality in observational studies,24,28 while EPA has demonstrated cardiovascular benefits independent of DHA in both epidemiological and interventional settings. Thus, the fatty acid shifts observed in this study are directionally consistent with a more favorable cardiometabolic profile, even in the absence of measurable short-term changes in clinical endpoints.
Despite these biologically meaningful changes in both plasma and erythrocyte fatty acids, no significant effects were observed on BMI, body composition, blood pressure, arterial stiffness, or plasma lipid concentrations. This is not unexpected since structural vascular changes, lipid remodeling, and body composition adaptations typically require longer intervention durations, larger sample sizes, or populations with overt metabolic dysregulation. Consistent with this interpretation, major anthropometric parameters, hs-CRP, blood pressure, fasting glucose and insulin concentrations, and HOMA-IR were also not significantly altered following MUFA- or PUFA-rich dietary interventions relative to high-carbohydrate or high-dairy-fat diets in our previous full-diet randomized controlled crossover trial.12
Similarly, several randomized controlled trials have reported that hemp seed and/or hempseed oil supplementation does not significantly modify circulating lipid parameters, including total cholesterol, triglycerides, HDL-C, and LDL-C,29–31 findings that are concordant with the present results. In addition, population-based evidence suggests that short-term (≥2 weeks) supplementation with n-3 PUFAs does not substantially affect total cholesterol levels per se.32 In contrast, some pre-clinical studies have reported improvements in atherosclerosis or insulin sensitivity following hemp product supplementation in animal models,33–35 while reductions in total and LDL cholesterol have also been observed in selected human cohorts.30 Discrepancies across studies likely reflect differences in species, baseline metabolic status, intervention dose, duration, and overall dietary context. Importantly, we emphasize that no single food or isolated dietary component is expected to induce substantial improvements in cardiometabolic health in isolation, particularly in the absence of broader dietary pattern optimization and lifestyle modification. Cardiovascular risk is shaped by the integrated effects of food matrix interactions, habitual diet quality, physical activity, and metabolic health. In this context, the absence of short-term changes in clinical risk markers does not diminish the biological relevance of the observed shifts in circulating fatty acid profiles, which may represent early and necessary metabolic adaptations that precede longer-term cardiometabolic benefits.
To the best of our knowledge, this was first human clinical study that directly investigate the beneficial effects of hemp seeds and hemp oil on major cardiometabolic risk factors in overweight and obese patients. A key strength of this study is the use of erythrocyte fatty acid composition as a primary outcome, providing an objective and biologically relevant marker of fatty acid exposure and incorporation. Exclusion of participants who had recently consumed n-3 supplements minimized background variability and strengthened attribution of observed changes to the intervention. However, we also admit that limitations should be acknowledged. Specifically, the intervention duration may have been insufficient to detect changes in downstream cardiometabolic outcomes. Dietary intake outside the intervention products was not quantitatively controlled, which may have attenuated detectable effects. Additionally, the relatively modest sample size limited statistical power to detect small but potentially meaningful changes in secondary outcomes. Finally, the study population consisted of overweight or obese adults from a single geographic region, which may limit generalizability.
Overall, the present study showed that 4-week consumption of hulled hempseed and hemp oil in overweight individuals increased ALA, GLA, DGLA and EPA relative percentages in plasma and RBC respectively, demonstrating effective incorporation of hemp-derived polyunsaturated fatty acids into long-term lipid pools. These changes occurred without adverse effects on lipid metabolism, vascular function, and/or body composition. Collectively, these findings support the metabolic safety of hemp products and highlight their potential utility as dietary sources of polyunsaturated fatty acids for improving circulating fatty acid profiles. Longer-term studies are warranted to determine whether these favorable changes translate into clinically meaningful cardiovascular benefits.
Author contributions
Y. S., K. J., D. M. prepared the manuscript. A. C. and A. S. L. conducted the clinical trial and analysis and contributed to the manuscript. R. M., D. M. designed the study protocol, selection criteria of participants, sought funding, ethical approval and reviewed the manuscript. Y. S., X. L. and D. M. revised the manuscript. R. M. and D. M. supervised the staff and conduct of the clinical trial. All authors contributed to, have read, made critical revisions, and approved the final manuscript before submission.Conflicts of interest
The authors declare no conflict of interests.Data availability
The data for this article is primarily based on analyses from human randomized controlled trial and securely stored at the University of Manitoba with ethical implications. Deidentified data will be made available upon request to the corresponding author.Acknowledgements
This project was funded by the Canada and Manitoba Governments through Growing Forward 2, a federal-provincial-territorial initiative. Matching funds were provided by Mitacs Canada Accelerate Program, Manitoba Harvest, Hempro Int. GmbH & Co. KG, and Hemp Oil Canada. Firstly, the authors acknowledge the volunteers that participated in the study. Additionally, the authors thank the RCFTR clinical unit staff; including, Haizhou Wang and Jeann Buenafe, for all their help.References
- E. Small and A. Cronquist, A Practical and Natural Taxonomy for Cannabis, Taxon, 1976, 25(4), 405–435 CrossRef.
- J. C. Callaway, Hempseed as a nutritional resource: An overview, Euphytica, 2004, 140, 65–71 CrossRef.
- B. D. Oomah, M. Busson, D. V. Godfrey and J. C. G. Drover, Characteristics of hemp (Cannabis sativa L.) seed oil, Food Chem., 2002, 76(1), 33–43 CrossRef CAS.
- C. Leizer, D. M. Ribnicky, A. Poulev, D. Dushenkov and I. Raskin, The composition of hemp seed oil and its potential as an important source of nutrition, J. Nutraceuticals, Funct. Med. Foods, 2000, 2(4), 35–53 CrossRef.
- WHO, WHO Cardiovascular diseases (CVDs) fact sheets, 2016 Search PubMed.
- K. Yamagishi, H. Iso, H. Yatsuya, N. Tanabe, C. Date and S. Kikuchi, et al., Dietary intake of saturated fatty acids and mortality from cardiovascular disease in Japanese: The Japan Collaborative Cohort Study for Evaluation of Cancer Risk (JACC) study, Am. J. Clin. Nutr., 2010, 92(4), 759–765 CrossRef CAS PubMed.
- M. S. Farvid, M. Ding, A. Pan, Q. Sun, S. E. Chiuve and L. M. Steffen, et al., Dietary linoleic acid and risk of coronary heart disease: A systematic review and meta-analysis of prospective cohort studies, Circulation, 2014, 130(18), 1568–1578 CrossRef CAS PubMed.
- W. S. Harris, M. Miller, A. P. Tighe, M. H. Davidson and E. J. Schaefer, Omega-3 fatty acids and coronary heart disease risk: Clinical and mechanistic perspectives, Atherosclerosis, 2008, 197(1), 12–24 CrossRef CAS PubMed.
- R. P. Mensink, P. L. Zock, A. D. M. Kester and M. B. Katan, Effects of dietary fatty acids and carbohydrates on the ratio of serum total to HDL cholesterol and on serum lipids and apolipoproteins: A meta-analysis of 60 controlled trials, Am. J. Clin. Nutr., 2003, 77(5), 1146–1155 CrossRef CAS PubMed.
- P. W. Siri-Tarino, Q. Sun, F. B. Hu and R. M. Krauss, Meta-analysis of prospective cohort studies evaluating the association of saturated fat with cardiovascular disease, Am. J. Clin. Nutr., 2010, 91(3), 535–546 CrossRef CAS PubMed.
- C. M. Albert, H. Campos, M. J. Stampfer, P. M. Ridker, J. E. Manson and W. C. Willett, et al., Blood Levels of Long-Chain n–3 Fatty Acids and the Risk of Sudden Death, N. Engl. J. Med., 2002, 346(15), 1113–1118 CrossRef CAS.
- D. Brassard, M. Tessier-Grenier, J. Allaire, E. Rajendiran, Y. She and V. Ramprasath, et al., Comparison of the impact of SFAs from cheese and butter on cardiometabolic risk factors: A randomized controlled trial, Am. J. Clin. Nutr., 2017, 105(4), 800–809 CrossRef CAS PubMed.
- P. M. Kris-Etherton and J. A. Fleming, Emerging Nutrition Science on Fatty Acids and Cardiovascular Disease: Nutritionists’ Perspectives, Adv. Nutr., 2015, 6(3), 326S–337S CrossRef CAS PubMed.
- T. M. Powell-Wiley, P. Poirier, L. E. Burke, J. P. Després, P. Gordon-Larsen and C. J. Lavie, et al., Obesity and Cardiovascular Disease: A Scientific Statement from the American Heart Association, Circulation, 2021, 43(21), e984–e1010 Search PubMed.
- P. J. H. Jones, D. S. MacKay, V. K. Senanayake, S. Pu, D. J. A. Jenkins and P. W. Connelly, et al., High-oleic canola oil consumption enriches LDL particle cholesteryl oleate content and reduces LDL proteoglycan binding in humans, Atherosclerosis, 2015, 238(2), 231–238 CrossRef CAS PubMed.
- K. M. Davis, K. S. Petersen, K. J. Bowen, P. J. H. Jones, C. G. Taylor and P. Zahradka, et al., Effects of Diets Enriched with Conventional or High-Oleic Canola Oils on Vascular Endothelial Function: A Sub-Study of the Canola Oil Multi-Centre Intervention Trial 2 (COMIT-2), a Randomized Crossover Controlled Feeding Study, Nutrients, 2022, 14(16), 3404 CrossRef CAS.
- D. Vodolazska and C. Lauridsen, Effects of dietary hemp seed oil to sows on fatty acid profiles, nutritional and immune status of piglets, J. Anim. Sci. Biotechnol., 2020, 11(1), 28 CrossRef CAS PubMed.
- N. Gakhar, E. Goldberg, M. Jing, R. Gibson and J. D. House, Effect of feeding hemp seed and hemp seed oil on laying hen performance and egg yolk fatty acid content: Evidence of their safety and efficacy for laying hen diets, Poult. Sci., 2012, 91(3), 701–711 CrossRef CAS PubMed.
- B. B. Albert, J. G. B. Derraik, C. M. Brennan, J. B. Biggs, M. L. Garg and D. Cameron-Smith, et al., Supplementation with a blend of krill and salmon oil is associated with increased metabolic risk in overweight men, Am. J. Clin. Nutr., 2015, 102(1), 49–57 CrossRef CAS PubMed.
- M. E. O'Reilly, Y. M. Lenighan, E. Dillon, S. Kajani, S. Curley and R. Bruen, et al., Conjugated Linoleic Acid and Alpha Linolenic Acid Improve Cholesterol Homeostasis in Obesity by Modulating Distinct Hepatic Protein Pathways, Mol. Nutr. Food Res., 2020, 64(7), 1900599 CrossRef PubMed.
- L. G. Gillingham and P. J. Jones, Consumption of a novel flaxseed and canola oil blend favourably improves serum lipid profiles in hyperlipidemic subjects, FASEB J., 2009, 23(S1), 722.1 CrossRef.
- W. S. Harris and C. Von Schacky, The Omega-3 Index: A new risk factor for death from coronary heart disease?, Prev. Med., 2004, 39(1), 212–220 CrossRef CAS PubMed.
- V. R. Ramprasath, I. Eyal, S. Zchut and P. J. Jones, Enhanced increase of omega-3 index in healthy individuals with response to 4-week n-3 fatty acid supplementation from krill oil versus fish oil, Lipids Health Dis., 2013, 12(1), 178 CrossRef PubMed.
- P. C. Calder, Omega-3 fatty acids and inflammatory processes: From molecules to man., Biochem. Soc. Trans., 2017, 45(5), 1105–1115 CrossRef CAS PubMed.
- J. T. Brenna, N. Salem, A. J. Sinclair and S. C. Cunnane, α-Linolenic acid supplementation and conversion to n-3 long-chain polyunsaturated fatty acids in humans, Prostaglandins, Leukotrienes Essent. Fatty Acids, 2009, 80(2–3), 85–91 CrossRef CAS PubMed.
- M. Plourde and S. C. Cunnane, Extremely limited synthesis of long chain polyunsaturates in adults: Implications for their dietary essentiality and use as supplements, Appl. Physiol., Nutr., Metab., 2007, 32(4), 619–634 CrossRef CAS PubMed.
- B. Strandvik, E. Svensson and H. W. Seyberth, Prostanoid biosynthesis in patients with cystic fibrosis, Prostaglandins, Leukotrienes Essent. Fatty Acids, 1996, 55(6), 419–425 CrossRef CAS PubMed.
- P. C. Calder, Omega-3 fatty acids and inflammatory processes, Nutrients, 2010, 2(3), 355–374 CrossRef CAS PubMed.
- N. Kaul, R. Kreml, J. A. Austria, M. N. Richard, A. L. Edel and E. Dibrov, et al., A Comparison of Fish Oil, Flaxseed Oil and Hempseed Oil Supplementation on Selected Parameters of Cardiovascular Health in Healthy Volunteers, J. Am. Coll. Nutr., 2008, 27(1), 51–58 CrossRef CAS PubMed.
- A. Mohammadrezaei, A. Kavakeb, M. Abbasalizad-Farhangi and M. Mesgari-Abbasi, Effects of hemp seed alone and combined with aerobic exercise on metabolic parameters, oxidative stress, and neurotrophic factors in young sedentary men, J. Food Biochem., 2022, 46(12), e14417 CrossRef CAS PubMed.
- U. S. Schwab, J. C. Callaway, A. T. Erkkilä, J. Gynther, M. I. J. Uusitupa and T. Järvinen, Effects of hempseed and flaxseed oils on the profile of serum lipids, serum total and lipoprotein lipid concentrations and haemostatic factors, Eur. J. Nutr., 2006, 45(8), 470–477 CrossRef CAS PubMed.
- W. S. Harris, n-3 Fatty acids and serum lipoproteins: Human studies, Am. J. Clin. Nutr., 1997, 65(5 Suppl), 1645S–1654S CrossRef CAS PubMed.
- M. Majewski and A. Jurgoński, The effect of hemp (Cannabis sativa l.) seeds and hemp seed oil on vascular dysfunction in obese male zucker rats, Nutrients, 2021, 13(8), 2575 CrossRef CAS PubMed.
- P. M. Opyd, A. Jurgoński, B. Fotschki and J. Juśkiewicz, Dietary Hemp Seeds More Effectively Attenuate Disorders in Genetically Obese Rats than Their Lipid Fraction, J. Nutr., 2020, 150(6), 1425–1433 CrossRef PubMed.
- X. Y. Li, Y. H. Liu, B. Wang, C. Y. Chen, H. M. Zhang and J. X. Kang, Identification of a sustainable two-plant diet that effectively prevents age-related metabolic syndrome and extends lifespan in aged mice, J. Nutr. Biochem., 2018, 51, 16–26 Search PubMed.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |