Open Access Article
Open Access ArticleFrom wild plant to functional ingredient: phytochemical insights and neuroprotective activity of Melissa officinalis subsp. altissima
Donatella
Ambroselli
ab,
Fabrizio
Masciulli
 c,
Manal
El Ali
d,
Annalisa
Chiavaroli
c,
Manal
El Ali
d,
Annalisa
Chiavaroli
 c,
Claudio
Ferrante
c,
Simonetta Cristina
Di Simone
c,
Cinzia
Ingallina
c,
Claudio
Ferrante
c,
Simonetta Cristina
Di Simone
c,
Cinzia
Ingallina
 *ab,
Luisa
Mannina
ab and
Alain
Muselli
*ab,
Luisa
Mannina
ab and
Alain
Muselli
 d
d
aFood Chemistry Lab, Department of Chemistry and Technology of Drugs, Sapienza University of Rome, P. le Aldo Moro 5, 00185 Rome, Italy. E-mail: cinzia.ingallina@uniroma1.it
bNMR Lab, Sapienza University of Rome, Piazzale Aldo Moro 5, 00185 Rome, Italy
cDepartment of Pharmacy, G. d'Annunzio University of Chieti-Pescara, Chieti, Italy
dNatural Products Chemistry Laboratory, University of Corsica, UMR CNRS 6134, Campus Grimaldi, BP 52, Corté, FR-20250, France
First published on 17th February 2026
Abstract
Melissa officinalis (lemon balm) is a culinary and medicinal herb traditionally used across diverse cultural systems for its cognitive-enhancing and neuroprotective effects. In light of its historical use in herbal teas, dietary supplements, and functional foods, this study investigates the bioactive potential of Melissa officinalis subsp. altissima, a wild subspecies of growing interest in the food and nutraceutical sectors. Wild specimens collected in Corsica (France) were investigated through an integrated metabolomic approach, combining a comprehensive phytochemical characterization of different extractive fractions and essential oils with a targeted biological evaluation performed on a hydroalcoholic extract selected as a representative model of the whole phytocomplex relevant to functional nutrition. Phytochemical profiling using high-resolution NMR spectroscopy identified a range of metabolites, including rosmarinic, chlorogenic, caffeic, and caftaric acids. GC-MS analyses were performed in parallel on the hydrodistilled essential oils and the volatiles obtained via HS-SPME, revealing a rich profile of bioactive volatile compounds such as germacrene-D, α,τ-cadinol, β-caryophyllene, and β-ocimene, which are known for their antioxidant and antimicrobial activities. Biological assays of the hydroalcoholic extracts demonstrated significant antioxidant capacity against hydrogen peroxide-induced oxidative stress in rat hypothalamic cells, as well as anti-inflammatory and neuroprotective effects in ex vivo murine assays. Notably, the extracts modulated the gene expression of key mediators such as TNF-α, NOS-2, IL-6, BDNF, and AChE suggesting a potential role in supporting brain health. These findings reinforce the potential of this lemon balm subspecies as a valuable natural ingredient for the formulation of functional foods and nutraceuticals with antioxidant, neuroprotective, and preservative properties. Moreover, the study underscores the broader significance of native aromatic plants as sustainable resources for health-oriented food innovation.
1. Introduction
Lemon balm, also known as Melissa officinalis, is a perennial shrubby plant from the Lamiaceae family. While it grows worldwide, its exact origins are not well-defined, though it's believed to have originated in the Eastern Mediterranean, Western Asia, Southern Europe, Caucasus, and Northern Iran. Italy and France are the major producers of lemon balm.1 In Europe, two main subspecies of lemon balm have been identified: Melissa officinalis subsp. officinalis and Melissa officinalis subsp. altissima (Sm.), which are distinguished by the indumentum of the leaves and the shape of the fruiting calyx.2 The Melissa officinalis subsp. altissima is also referred to as Melissa romana MILLER or wild lemon balm.3The Lamiaceae family is important as a source of medicinal, aromatic, and edible plants. Indeed, the lemon balm plant is rich in biologically active components, including terpenes, phenolic acids, and flavonoids, and has been cited for significant pharmacological activities, as antibacterial, antifungal, antiviral, anti-inflammatory, and antioxidant properties.4
The growing preference for a healthy lifestyle and nutrition has led to increased consumption of natural and functional food products. As a result, there has been a significant expansion in the use of plants and their derivatives in the food industry and for nutraceutical applications.5 The use of lemon balm leaf products for mild stress symptoms, as sleep aid, and for mild digestive disorders has been approved by the HMPC (Committee on Herbal Medicinal Products) of the EMA (European Medicines Agency) based on traditional use. Although there aren't enough clinical studies, the efficacy of these herbal medicines is well-founded, and evidence shows their safe use in these ways for at least thirty years. Ethnopharmacological studies have reported that lemon balm is used in many countries to treat various disorders, mainly in the pharmaceutical forms of essential oil, infusion, and decoction.6
In addition to this, there are studies in the literature discussing the potential positive effects of this medicinal plant on memory formation and neuroprotection.7 Through mechanisms involving rosmarinic acid, its primary polyphenol, lemon balm appears to inhibit the formation of β-amyloid fibrils (Aβ1-40 and Aβ1-42).8–10
In this framework, plant metabolomics—dedicated to detecting bioactive chemicals responsible for therapeutic outcomes—has become increasingly prominent.11 The vegetable metabolome consists of primary and secondary metabolites with different physicochemical properties, including some that are species-specific. Therefore, it is essential to gather as much information as possible about the composition of an aromatic plant, and thus the metabolites extracted, the characterization of the volatile component, and the biological properties that are related to it.
In our previous study,12 metabolomic profiling via NMR was performed on three officinal plant species, including Melissa officinalis subsp. officinalis. The investigation evaluated the influence of both pedoclimatic factors and cultivation practices by comparing three ecotypes: Land Spontaneous Ecotype (LSE), Mountain Spontaneous Ecotype (MSE), and Organic Ecotype (OE). The goal was to map their metabolic diversity and assess how altitude, growing conditions (wild vs. organic cultivation), and ecotype typology affect the abundance and identity of metabolites.
In the present study, Melissa officinalis subsp. altissima (MOA), a wild-growing species from Corsica, was analysed using a multi-methodological analytical protocol to obtain a complete metabolomic profile. In particular, high-resolution Nuclear Magnetic Resonance (NMR) spectroscopy was applied to investigate the hydroalcoholic and organic metabolites. Moreover, the chemical compositions of the essential oils obtained from the vegetative parts of the plant were analysed by Gas Chromatography coupled to Mass Spectrometry (GC-MS).
The aim of this research was to determine the phytochemical composition of extracts and essential oils in relation to their antioxidant and antimicrobial activities, respectively. In addition, the effects on general inflammation and neuroinflammation were evaluated by biological assays. In this freamwork, the metabolomic characterisation of the non-polar fraction was performed exclusively for chemical profiling purposes, to provide a comprehensive description of the phytochemical composition of Melissa officinalis subsp. altissima. No biological assays were conducted on the non-polar extract, and therefore no direct functional conclusions can be drawn for this fraction. Specifically, the biocompatibility of hydroalcoholic MOA extracts was assessed in rat hypothalamic cells (HypoE22). The same cell line was tested to evaluate the antioxidant activity exerted by MOA hydroalcoholic extracts against cell damage induced by hydrogen peroxide (H2O2). Then, the gene expression of inflammatory and neurotrophic mediators such as TNF-α (Tumour necrosis factor), NOS-2 (Nitric oxide synthase-2), and BDNF (Brain-derived neurotrophic factor) in brain samples from C57/BL6 mice (prefrontal cortex and hypothalamus) treated with MOA hydroalcoholic extracts was evaluated. The cortical tissues of mice were also exposed to β-amyloid to subsequently assess the expression of IL-6 (Interleukin-6) and AChE (Acetylcholinesterase) genes during treatment with hydroalcoholic extracts of MOA.
An effective strategy to enhance specific activities—such as antioxidant, antimicrobial, anti-inflammatory, or neuroprotective effects—is to combine compounds that possess the desired properties. The resulting synergistic effect can significantly increase the targeted activity, even at low concentrations.
2. Materials and methods
2.1. Plant material
Melissa officinalis subsp. altissima aerial parts were harvested in central Corsica (Corte, France, 42°17′56.5″ N, 9°10′13.0″ E), during the flowering stage in July 2023. The botanical determination of the plants was carried out according to the characteristics summarised in Flora Corsica.132.2. Chemicals and reagents
Ethanol CH3CH2OH (HPLC-grade), methanol CH3OH (HPLC-grade), chloroform CHCl3 (HPLC-grade), and diethyl ether (C2H5)2O were obtained from Merck (Milan, Italy).Double-distilled water was purchased using a Millipore Milli-Q Plus water treatment system (Millipore Bedford Corp., Bedford, MA, USA).
EDTA deuterated was purchased from Cambridge Isotope laboratories, Inc. (Andover, USA). Monobasic potassium phosphate (KH2PO4) and dibasic potassium phosphate (K2HPO4) were purchased from Aldrich-Fluka-Sigma S.r.l. (Milan, Italy). Deuterated water (D2O), methanol-D4 (CD3OD), and chloroform-D (CDCl3) were purchased from Euriso-Top (Saclay, France).
3-(Trimethylsilyl)-propionic-2,2,3,3-d4 acid sodium salt (TSP) and Dimethyl sulfoxide (DMSO) were acquired from Merck (Milan, Italy). TRI reagent was obtained from Sigma-Aldrich (St Louis, MO, USA). All murine genes detailed below, PCR primers, TaqMan probes, and High-Capacity cDNA Reverse Transcription Kit were sourced from Thermo Fisher Scientific (Walthman, Massachusetts, USA). The genes included: GAPDH, TNF-α, NOS-2, IL-6, AChE and BDNF. β-Amyloid (1–42), (rat/mouse) (TFA) (95%) was acquired from MedChemTronica (The European Branch of MedChemExpress LLC, Sweden).
2.3. NMR analysis
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) mixture, sonicated (thermostat ultrasonic bath ARGOLAB DU-100 (Rome, Italy)) for 10 minutes at room temperature, then partitioned with CHCl3 and water to form a biphasic system. After centrifugation (Eppendorf Centrifuge 5430 R (Milan, Italy)), the hydroalcoholic and organic phases were separated. The extraction was repeated twice on the pellet, and combined extracts were dried under nitrogen. Triplicate analyses were performed.
1, v/v) mixture, sonicated (thermostat ultrasonic bath ARGOLAB DU-100 (Rome, Italy)) for 10 minutes at room temperature, then partitioned with CHCl3 and water to form a biphasic system. After centrifugation (Eppendorf Centrifuge 5430 R (Milan, Italy)), the hydroalcoholic and organic phases were separated. The extraction was repeated twice on the pellet, and combined extracts were dried under nitrogen. Triplicate analyses were performed.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v).
1 v/v).
NMR analyses were conducted using a Jeol JNM-ECZ 600R instrument operating at a proton frequency of 600.17 MHz and equipped with a Jeol 5 mm FG/RO DIGITAL AUTOTUNE probe. Spectra processing and signal integration were performed using JEOL Delta software version 5.3.1. Additionally, two-dimensional experiments (1H–1H TOCSY, 1H–13C HMBC, and 1H–13C HSQC), and the quantifications of the hydroalcoholic and organic portions were performed following the experimental protocols previously reported.12
2.4. GC-MS analysis
The hydrosol was obtained by collecting the initial 300 mL (co-coating) from the Clevenger device during hydrodistillation. Then, the hydrosol underwent a liquid–liquid extraction procedure. A total of 300 mL was extracted in three portions of 50 mL each using diethyl ether. The organic phase was washed with 50 mL of water saturated with NaCl, dried over anhydrous Na2SO4, and filtered. The resulting product was concentrated to yield the hydrosol extract (HYD). The obtained HYD was colorless, with a yield of 0.06% (w/dw).
2.5. Biological assays
For biological assays, phytocomplex-related biological effects were evaluated using a green hydroalcoholic extraction protocol previously described.20 In particular, 100 mg of lyophilized sample was added to 2 mL of a H2O/EtOH mixture (30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70% v/v) with a 1
70% v/v) with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 ratio of plant matrix to extraction solvent. The obtained system was sonicated (ARGOLAB DU-100 ultrasonic thermostat bath (Rome, Italy)) at 30 °C for 10 min and then centrifuged (Eppendorf 5430 R centrifuge (Milan, Italy)) for 5 min (30 °C, 7745g). The hydroalcoholic extract was separated, and the residual pellet was extracted twice using the same protocol. The collected supernatants were pooled and filtered through 0.45 μm syringe filters and then brought to dryness under a constant flow of nitrogen at room temperature. The dried hydroalcoholic extract was dissolved in a DMSO/Milli-Q water mixture using a total solvent volume of 6 mL to maintain the appropriate matrix/solvent ratio. In addition, to avoid the potential toxicity of MOA extract, the maximum phytochemical concentration tested was 200 μg mL−1, with less than 1% DMSO. Therefore, the stock solution for cell treatment or tissue incubation was prepared by adding a 50
20 ratio of plant matrix to extraction solvent. The obtained system was sonicated (ARGOLAB DU-100 ultrasonic thermostat bath (Rome, Italy)) at 30 °C for 10 min and then centrifuged (Eppendorf 5430 R centrifuge (Milan, Italy)) for 5 min (30 °C, 7745g). The hydroalcoholic extract was separated, and the residual pellet was extracted twice using the same protocol. The collected supernatants were pooled and filtered through 0.45 μm syringe filters and then brought to dryness under a constant flow of nitrogen at room temperature. The dried hydroalcoholic extract was dissolved in a DMSO/Milli-Q water mixture using a total solvent volume of 6 mL to maintain the appropriate matrix/solvent ratio. In addition, to avoid the potential toxicity of MOA extract, the maximum phytochemical concentration tested was 200 μg mL−1, with less than 1% DMSO. Therefore, the stock solution for cell treatment or tissue incubation was prepared by adding a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50% v/v DMSO/H2O mixture to the dried hydroalcoholic extract. The system was sonicated for 10 min at room temperature and filtered through 0.22 μm syringe filters to obtain a sterile solution. As shown in Table 1, the stock solution was used to create subsequent dilutions for in vitro and ex vivo assays.
50% v/v DMSO/H2O mixture to the dried hydroalcoholic extract. The system was sonicated for 10 min at room temperature and filtered through 0.22 μm syringe filters to obtain a sterile solution. As shown in Table 1, the stock solution was used to create subsequent dilutions for in vitro and ex vivo assays.
| Phytochemical concentration | DMSO % | |
|---|---|---|
| a This percentage is higher than that tolerated by the cells, which is why the effects on cell viability were evaluated in comparison to two control lines (Ctrl) in 96-well culture plates (one line was at this DMSO concentration). | ||
| 80× | 16 mg mL−1 | 50 |
| 10× | 2 mg mL−1 | 6.25 |
| Highest tested | 500 μg mL−1 | 1.56a |
| Higher Int. tested | 200 μg mL−1 | 0.625 |
| Lower Int. tested | 100 μg mL−1 | 0.312 |
| Lowest tested | 50 μg mL−1 | 0.156 |
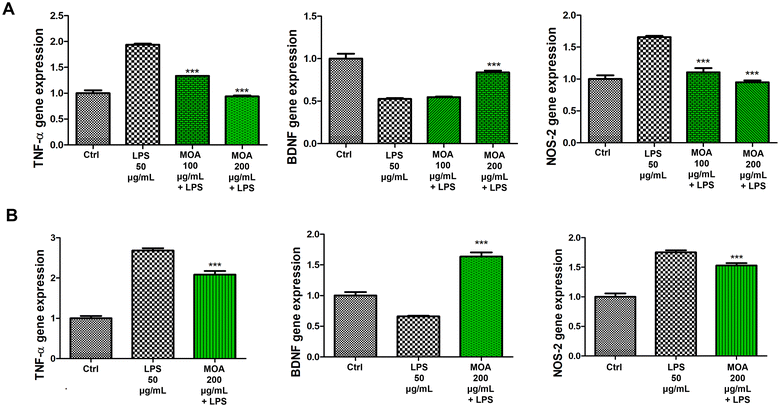
For the LPS stimulus, brain samples were cut to obtain sections of the prefrontal cortex (split in two, n = 16) and the hypothalamus (n = 8). Next, brain tissue samples were placed in 6-well plates in RPMI buffer by adding bacterial LPS (E. coli) (50 μg mL−1) to each well, except for the control (Ctrl). The prefrontal cortex samples were treated with hydroalcoholic extract of MOA at concentrations of 100 and 200 μg mL−1. The hypothalamic sections were treated only at the highest concentrations of MOA extract, 200 μg mL−1.
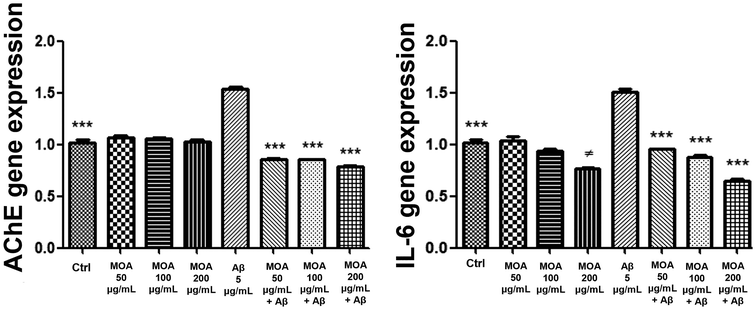
For stimulation with β-amyloid, brain samples were cut to obtain prefrontal cortex sections divided into two, with n = 10. Thus, the brain tissue samples were placed in 10-well plates containing RPMI buffer, two of which were used as controls (Ctrl). Prefrontal cortex samples were treated with hydroalcoholic extract of basal MOA at concentrations of 50, 100, and 200 μg mL−1, and with hydroalcoholic MOA extract (50, 100, and 200 μg mL−1) after stimulation with β-amyloid (C = 0.05 mg mL−1). Two prefrontal cortex samples were subjected to β-amyloid (C = 0.05 mg mL−1) stimulus treatment alone.
After 4 h of incubation at 37 °C, all samples were collected and stored at −80 °C until RNA extraction.
Total RNA was extracted from the brain tissues using TRI reagent, according to the manufacturer's protocol, and 1 μg of total RNA from each sample in a 20 μL reaction volume was reverse transcribed using High-Capacity cDNA Reverse Transcription Kit. Gene expression of TNF-α, BDNF, NOS-2, IL-6, and AChE was evaluated by quantitative real-time PCR using TaqMan probe-based chemistry, as previously reported.21 GADPH (Glyceraldehyde-3-phosphate dehydrogenase) was used as the housekeeping gene. Real-time PCR was performed in triplicate for each cDNA sample with respect to the selected genes. The elaboration of data was carried out with the Sequence Detection System (SDS) software version 2.3 (Thermo Fischer Scientific). Relative quantification of gene ex-pression was performed by the comparative 2−ΔΔCt method.22
For statistical analysis one-way ANOVA, followed by Tukey's multiple comparison test, was applied to underline, among genes expression, significant differences for each MOA hydroalcoholic extract concentration vs. LPS (Fig. 3) or vs. β-amyloid (Fig. 4) stimuli.
3. Results and discussion
3.1. NMR-based detection of hydrophilic metabolites
Assignments of 1H NMR spectra of water-soluble MOA extracts in D2O/phosphate buffer, Table 2, were based on 2D NMR experiments (1H–1H TOCSY, 1H–13C HSQC, and 1H–13C HMBC) and a previous NMR study.12| Compound | Assignment | 1H (ppm) | Multiplicity [J (Hz)] | 13C (ppm) | Mean ± SD (mg 100 per g) |
|---|---|---|---|---|---|
| Sugars | |||||
| α-D-Fructofuranose | C-2 | 105.9 | |||
| CH-3 | 4.11 | 82.3 | |||
| β-D-Fructofuranose | C-2 | 102.6 | 167.61 ± 1.10 | ||
| CH-4 | 4.12* | 75.6 | |||
| β-D-Fructopyranose | C-2 | 99.3 | |||
| β-Galactose | CH-1 | 4.60* | d [7.9] | 97.3 | 51.38 ± 1.15 |
| CH-2 | 3.51 | ||||
| CH-3 | 3.67 | ||||
| CH-4 | 3.95 | ||||
| CH-5 | 4.05 | ||||
| CH-6 | 3.78 | ||||
| α-Glucose | CH-1 | 5.23* | d [3.8] | 93.3 | 362.84 ± 2.90 |
| CH-2 | 3.54 | 72.6 | |||
| CH-3 | 3.72 | 73.3 | |||
| CH-4 | 3.41 | 70.8 | |||
| CH-5 | 3.84 | 72.5 | |||
| β-Glucose | CH-1 | 4.66* | d [7.9] | 97.0 | 229.40 ± 1.83 |
| CH-2 | 3.25 | 75.3 | |||
| Inulin | CH-1 (Glc) | 5.44* | 93.9 | 738.19 ± 3.88 | |
| CH-2 | 3.57 | 72.1 | |||
| CH-3 | 3.78 | 73.6 | |||
| CH-4 | 3.48 | 70.2 | |||
| CH-5 | 3.85 | 73.4 | |||
| CH2-6 | 3.83 | 61.1 | |||
| CH2-1′ (Fru) | 3.75; 3.89 | ||||
| CH-3′a | 4.21 | 77.5 | |||
| CH-3′b | 4.26 | ||||
| CH-4′a | 4.05 | 74.9 | |||
| CH-5′ | 3.89 | 82.2 | |||
| Myo-inositol | CH-2,5 | 3.56 | |||
| CH-3,6 | 3.65 | ||||
| CH-4 | 3.28* | 75.0 | 628.52 ± 2.06 | ||
| Sucrose | CH-1 (Glc) | 5.42* | d [3.9] | 93.1 | 1736.28± 4.20 |
| CH-2 | 3.53 | 71.8 | |||
| CH-3 | 3.72 | 73.6 | |||
| CH-4 | 3.42 | 70.2 | |||
| CH-5 | 3.84 | 73.5 | |||
| C2′ (Fru) | 104.8 | ||||
| CH-3′ | 4.23 | 77.4 | |||
| CH-5′ | 3.92 | 82.4 | |||
| CH-4′ | 3.83 | 61.2 | |||
| Organic acids | |||||
| Acetate | CH3 | 1.92* | s | 24.4 | 13.22 ± 1.12 |
| COOH | 184.4 | ||||
| Citrate | α,γ-CH | 2.56* | d [16.0] | 46.0 | 63.80 ± 1.64 |
| α′,γ′-CH | 2.68 | 46.0 | |||
| β-C | 73.2 | ||||
| 1,5-COOH | 180.2 | ||||
| 6-COOH | 183.0 | ||||
| Formate | HCOOH | 8.46* | s | ||
| Lactate | β-CH3 | 1.34 | d [7.1] | 21.4 | 4.66 ± 0.28 |
| Malate | α-CH | 4.31 | dd [9.8; 3.3] | 71.0 | 470.89 ± 1.41 |
| β-CH | 2.68 | dd [15.5; 3.3] | 43.5 | ||
| β’-CH | 2.41 | dd [15.5; 9.8] | 43.5 | ||
| Quinate | CH2-2,2′ | 1.89, 2.06* | dd [10.8;13.4], m | 41.8 | 365.84 ± 1.30 |
| CH-3 | 4.16 | 71.5 | |||
| CH-4 | 3.56 | 76.3 | |||
| CH-5 | 4.03 | 68.0 | |||
| Succinate | α,β-CH2 | 2.42* | s | 35.2 | 95.34 ± 1.87 |
| Tartrate | CH(OH)COOH | 4.37* | s | 75.3 | 287.50 ± 2.75 |
| Amino acids | |||||
| Alanine | α-CH | 3.80 | 51.0 | 23.61 ± 0.08 | |
| β-CH3 | 1.49* | d [7.2] | 17.3 | ||
| COOH | 178.6 | ||||
| GABA | α-CH2 | 2.30* | t [7.4] | 35.4 | 38.48 ± 0.00 |
| β-CH2 | 1.90 | 24.9 | |||
| γ-CH2 | 3.01 | t [7.5] | 40.4 | ||
| Glutamate | α-CH | 3.78 | 55.6 | 117.37 ± 1.51 | |
| β,β′-CH2 | 2.07; 2.14* | 28.0 | |||
| γ-CH2 | 2.36 | m | 34.8 | ||
| Glutamine | α-CH | 3.76 | 55.3 | ||
| β,β′-CH2 | 2.14 | m | 27.5 | ||
| γ-CH | 2.45* | m | 31.9 | ||
| Isoleucine | α-CH | 1.97 | 38.0 | 43.43 ± 1.53 | |
| β-CH | 1.27 | 29.4 | |||
| γ-CH3 | 1.01* | d [7.1] | 15.7 | ||
| δ-CH3 | 0.89 | d [7.4] | |||
| Leucine | β-CH2 | 1.74 | 41.0 | 13.10 ± 0.87 | |
| γ-CH | 1.71 | ||||
| δ-CH3 | 0.97* | d [6.4] | 23.1 | ||
| δ′-CH3 | 0.96 | 22.1 | |||
| CH-4 | 7.38 | ||||
| CH-3,5 | 7.43 | m | 130.6 | ||
| Proline | α-CH | 4.13 | 62.5 | 251.67 ± 2.18 | |
| β,β′-CH2 | 2.07, 2.33 | 30.0 | |||
| γ-CH3 | 2.01* | m | 25.0 | ||
| δ,δ′-CH3 | 3.33, 3.41 | 47.4 | |||
| Threonine | α-CH | 3.59 | 62.3 | 21.40 ± 0.53 | |
| γ-CH3 | 1,33* | d [6.7] | 20.71 | ||
| Tyrosine | CH-2, 6 ring | 7.22* | d [8.2] | 132.0 | 728.15 ± 2.60 |
| CH-3, 5 ring | 6.96 | d [8.5] | 117.0 | ||
| Valine | α-CH | 3.60 | 20.00 ± 0.15 | ||
| β-CH | 2.27 | ||||
| γ-CH3 | 1.10 | d [7.1] | 17.7 | ||
| γ′-CH3 | 1.04* | d [7.1] | 19.0 | ||
| Polyphenols | |||||
| Caffeic acid | α-CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH CH |
6.36* | d [16.00] | 116.0 | 145.07 ± 1.47 |
β-CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH CH |
7.29 | d [16.00] | 147.2 | ||
| Caftaric acid | CH-10 | 5.33 | d [2.2] | 454.49 ± 12.62 | |
| CH-11 | 4.57 | d [2.2] | |||
| CH-2 | 6.49* | d [16.0] | 115.4 | ||
| CH-3 | 7.72 | d [16.0] | 148.0 | ||
| CH-8 ring | 6.96 | d [8.2] | 117.6 | ||
| CH-9 ring | 7.17 | dd [2.0; 8.2] | 124.2 | ||
| CH-5 ring | 7.25 | d [2.0] | 117.5 | ||
| COOH | 171.4 | ||||
| Chlorogenic acid | CH-2 | 6.39* | d [16.0] | 115.8 | 525.78 ± 3.0 |
| CH-3 | 7.37 | d [16.0] | 146.8 | ||
| CH2-2′ | 1.89; 2.10 | m | 41.7 | ||
| CH-4′ | 3.90 | m | 70.8 | ||
| CH2-6′ | 1.99; 2.06 | m | 38.6 | ||
| Rosmarinic acid | CH-2 | 6.22* | d [16.0] | 115.8 | 1352.01 ± 11.86 |
| CH-3 | 7.37 | d [16.0] | 146.8 | ||
| CH-1a | 5.01 | m | 77.5 | ||
| CH2-2a | 2.85; 2.93 | m | 37.9 | ||
Five sugars (galactose, glucose, fructose, sucrose, and inulin), a polyol (myo-inositol), eight organic acids (acetate, citrate, lactate, quinate, formate, malate, succinate, and tartrate), ten amino acids (alanine, GABA, glutamate, glutamine, isoleucine, leucine, proline, threonine, tyrosine, and valine), and four polyphenols (caftaric, chlorogenic, caffeic, and rosmarinic acids) were identified.
Metabolic diversity is a key indicator of the physiological and adaptive flexibility of plants, often influenced by genetic and ecological differentiation at the subspecies level.23 Subspecies may show distinct metabolic footprints due to variations in gene expression and environmental pressures. This biochemical heterogeneity affects both primary metabolites, which were essential for growth, and secondary compounds involved in stress response and ecological interactions.24
To the best of our knowledge, the assignment and quantification of a 1H NMR spectrum of an extract of Melissa officinalis subsp. altissima has not been reported previously.
Here, the metabolites identified in the MOA samples were quantified by integrating specific 1H NMR signals and the results are shown in Table 2.
The following sections provide a separate discussion for each class of compounds, reflecting increased metabolic activity and indicating specific physiological states of the plant.
3.2. NMR-based detection of lipophilic metabolites
The identification of lipophilic metabolites was based on a combination of 1D and 2D NMR experiments, comparing literature data on complex lipid mixtures,12,54 and is reported in Table 3.| ppm | Group | Metabolite | Mean ± SD (mg 100 per g) | |
|---|---|---|---|---|
| I β-Sit | 0.65 | CH3-18 | β-Sitosterol | 91.07 ± 5.37 |
| I FA | 2.30 | CH2-11 | Totally fatty acids | 8.15 ± 1.0 |
| I DUFA | 2.73 | CH2-11 | Linoleic acid | 0.37 ± 0.05 |
| I TUFA | 2.77 | CH2-11,14 | Linolenic acid | 1.58 ± 0.38 |
| I PCG | 3.23 | N(CH3)3 | Glyceroyl-phosphatidylcholine | 0.10 ± 0.00 |
| I DGG | 4.87 | CH-1 | Glyceroyl-digalactose | 0.54 ± 0.01 |
| I PHEO | 11.15 | CH-5 | Pheophytin | 0.10 ± 0.00 |
| I CHL | 11.13 | CH-5 | Chlorophyll | 0.05 ± 0.01 |
In line with an untargeted NMR-based metabolomic approach, the analysis allowed the comprehensive detection of the main classes of lipophilic constituents present in the organic extracts, including fatty acids, sterols, chlorophyll derivatives, phospholipids and galactolipids. Quantitative evaluation was subsequently focused on selected representative metabolites showing well-resolved and diagnostic NMR signals, while additional minor components were annotated at the qualitative level to define the overall lipophilic fingerprint. Quantitative analysis of the lipophilic portion revealed that MOA contained significantly lower amounts of total fatty acids and unsaturated fatty acids than M. o. officinalis, whereas presenting an exceptionally high percentage of β-sitosterol (91.07 ± 5.37 mg per 100 g),12Table 3. These values suggest an active biosynthetic pathway for phytosterol and a possible role in membrane stabilization under environmental stress conditions, as reported for other plant species.55,56 High levels of β-sitosterol have been linked to increased tolerance to abiotic stress and to the health properties of plant extracts.57 Among sterols, although stigmasterol was found in the previously found,12 it was undetected in MOA. Besides sterols and fatty acids, the untargeted NMR analysis also revealed the presence of chlorophyll and pheophytin, detected through their characteristic downfield resonances (δ ∼11.1 to 11.2 ppm), which are commonly observed in plant lipophilic fractions and reflect the photosynthetic pigment pool. Although present at low concentrations, these compounds contribute to defining the lipophilic metabolomic signature of MOA and are indicative of chloroplast-derived components. MOA has a significantly reduced lipid profile compared to the other study,12Table 3. In all fatty acid classes, the levels detected in MOA were drastically lower than those reported for the officinalis subspecies. DUFAs occurred at roughly one-twenty-third of the levels reported by Ambroselli et al. (2024),12 whereas TUFAs and MUFAs reached only about one-seventeenth of those values, see Table 3. Even SFAs reached only about a quarter of the concentrations previously detected. Signals attributable to glycerol-based lipids, such as glycerophosphocholine and digalactosyldiacylglycerols, were also observed in the 2D NMR spectra, supporting the presence of membrane-related lipid components, although their low abundance prevented an extensive quantitative evaluation. Total fatty acids in MOA amount to about one-tenth and total unsaturated fatty acids to about one-eighteenth of the levels found in officinalis ecotypes.12 This widespread depletion is particularly pronounced in the unsaturated fractions, showing a fundamentally different membrane lipid composition.58 Compared with published data on Melissa o. officinalis, this divergence is even more evident. Studies on Tunisian and Moroccan populations of this subspecies consistently report lipid fractions dominated by unsaturated fatty acids—especially linoleic acid, which typically accounts for 60–75% of total fatty acids—accompanied by moderate amounts of saturated fatty acids.53,59 These works also identify a sterol profile composed of β-sitosterol, stigmasterol, and campesterol, confirming the presence of multiple sterol species within the lipophilic matrix of Melissa o. officinalis. In contrast, MOA exhibits a drastic depletion across all fatty-acid classes and a near-exclusive accumulation of β-sitosterol, with stigmasterol completely absent. Furthermore, comprehensive GC/MS analyses such as those reported by Ieri et al. describe Melissa o. officinalis as chemically rich in lipophilic components, reinforcing that a lipid-poor and sterol-skewed profile such as that observed in MOA is not typical of the officinalis subspecies.60
3.3. GC-MS analysis of volatile compounds
The chemical compositions of the essential oil and the volatile fraction are listed in Table 4. In the essential oil MOA, 38 components were identified, constituting 90.5% of the oil. In the volatile fraction 28 components were identified. The main components of the oil were hydrocarbon compounds (70.7%), with germacrene D (44.2%) as the dominant sesquiterpene. The oxygen compounds included 11 sesquiterpenes (17.3%), one diterpene (0.7%), one monoterpene (0.4%) and one non-terpenic compound (1.2%). Of the 54 individual compounds identified from the essential oil and volatile fraction, 51 were matched using EI-mass spectra and library retention indices. The main oil components besides germacrene D were α-cadinol (7.4%), β-caryophyllene (6.9%) and τ-cadinol (5.9%). Few studies on the volatile component of MOA have been found in the literature. The chemical characterization of essential oil from this subspecies (formerly known as M. officinalis L. var. romana) has been reported by Dawson et al.61 In a study,3 the composition of MOA samples from different stations in Sardinia (Italy) and in different growing seasons was analysed. Subsequently, Miceli et al. reported the essential oil composition of samples collected in southern Italy.62 A comprehensive survey of essential oils extracted from Melissa officinalis L. ssp altissima collected in Tarquinia (Italy) was carried out, showing a variability in the relative amounts of the main compounds depending on the extraction time and harvest period.63 In particular, although some of the main compounds have already been reported, their relative quantities vary depending on when they are extracted, harvested and, most importantly, the place where the plant grows. In the first cited study,61 the major compound was β-cubebene (39%) and the minority β-caryophyllene (4%), totally disagreeing with Usai et al.,3 showing a high concentration of terpenes such as β-caryophyllene (17.3–27.5%), α-muurolene (17.5–38.8%) and caryophyllene oxide (3.3–7.9%). The EO obtained from the flowering tops of plants, in Italy, showed the predominance of caryophyllene (15.8%), cumene (14%), β-pinene (12%) and citral (12%), whereas other constituents presented in smaller quantities were menthol (6.5%), α-pinene (5.1%), spathulenol (4.8%) and γ-terpinene (4.0%).62 Comparing these results with the oils from Tarquinia, in particular with the August material, significant differences in the percentages of caryophyllene, β-pinene and spathulenol are observed.63 Depending on the harvest period, chemical diversity was observed; GC/MS analysis showed that cis-caryophyllene and its oxide were the main constituents of EO. Other compounds were predominant only in certain months, such as germacrene D in July and September, and terpinene-4-ol in August and September. However, our results are in line with those from Sardinia.3 Variations in the amounts of the compounds reflect regional and seasonal differences, as some OE constituents are characteristic of specific phenological phases. In contrast, others are only associated with the reproductive period of the plant, as observed in the case of the samples from Italy. In the volatile fraction analyzed by HS-SPME, described for the first time, hydrocarbon compounds are dominant, accounting for 62.1% of the total composition, with 37.8% monoterpenes. The main components identified were (E)-β-ocimene (20%) and germacrene D (16.7%). Also detected was the presence of oxygenated molecules (1–11), mainly aliphatic aldehydes with relatively short linear chains, including isovaleraldehyde (4.4%), hexanal (5.2%) and 2-methylbutanal (3.3%), which were not found in the essential oil. Significant quantitative and qualitative differences were observed between OE and VF analysis for oxygenated and hydrocarbon compounds. Establishing a direct correlation between the HS-SMPE and hydrodistillation techniques proved difficult. The former is governed by phase equilibrium, whereas the latter involves the near-complete extraction of plant volatiles.| No.a | Compounds | RIalittb | RIaexpc | RIpexpd | % Identification | Identificatione | |
|---|---|---|---|---|---|---|---|
| EO | VF | ||||||
| a Compounds are listed in order of their elution from non-polar Rtx-1 column. b RIalitt: Retention indices for a non-polar column (R. P. Adams, 2007). c RIaexp: Retention indices determined experimentally on the non-polar Rtx-1 column. d RIpexp: Retention indices determined experimentally on the polar Rtx-wax column. e The contents were determined on the non-polar column Rtx-1 column. EO: essential oil, VF: volatiles sampled by SPME, Ref: compounds identified from commercial libraries. | |||||||
| 1 | Butyraldehyde | 605 | 607 | 880 | — | 1.1 | MS, Ref |
| 2 | 2-Methyl 1-propanol | 605 | 605 | 1081 | — | 0.5 | MS |
| 3 | Isovaleraldehyde | 632 | 631 | 1082 | — | 4.3 | MS |
| 4 | 2-Methylbutanal | 644 | 643 | 909 | — | 3.3 | MS, Ref |
| 5 | Penten-3-ol | 662 | 662 | 1144 | — | 1 | MS |
| 6 | Furan-2-ethyl | 691 | 690 | 960 | — | 0.5 | MS, Ref |
| 7 | 3-Methylbutanol | 719 | 716 | 1177 | — | 2.1 | MS |
| 8 | 2-Methylbutanol | 727 | 717 | 1178 | — | 1.8 | MS |
| 9 | Hexanal | 777 | 770 | 1308 | — | 5.2 | MS |
| 10 | (E) Hex-3-en-1-ol | 814 | 810 | 1360 | — | 1.3 | MS |
| 11 | (Z)-Hex-3-en-1-ol | 836 | 831 | 1380 | — | 2.8 | MS |
| 12 | α-Thujene | 922 | 922 | 1405 | — | 1.6 | MS |
| 13 | Benzaldehyde | 930 | 929 | 1536 | 0.2 | — | MS |
| 14 | α-Pinene | 931 | 931 | 1023 | — | 0.4 | MS |
| 15 | Oct-1-en-3-ol | 962 | 959 | 1447 | 1.2 | 3.5 | MS |
| 16 | Sabinene | 973 | 963 | 1124 | 0.4 | 3 | MS |
| 17 | β-Pinene | 978 | 969 | 1114 | 0.4 | 6.6 | MS |
| 18 | Myrcene | 978 | 976 | 1159 | — | 0.4 | MS |
| 19 | P-Cymene | 1015 | 1010 | 1272 | 0.1 | 0.8 | MS |
| 20 | 1,8-Cineole | 1020 | 1020 | 1209 | — | 0.1 | MS |
| 21 | (Z)-β-Ocimene | 1029 | 1023 | 1234 | 0.7 | 5 | MS |
| 22 | (E)-β-Ocimene | 1040 | 1035 | 1252 | 3 | 20 | MS |
| 23 | Terpinen-4-ol | 1161 | 1161 | 1599 | 0.4 | — | MS |
| 24 | α-Copaene | 1379 | 1376 | 1490 | 1.4 | — | MS |
| 25 | β-Bourbonene | 1386 | 1379 | 1516 | 0.1 | 0.7 | MS |
| 26 | β-Cubebene | 1388 | 1383 | 1593 | 0.4 | — | MS |
| 27 | β-Elemene | 1388 | 1388 | 1590 | 2.6 | 1 | MS |
| 28 | (E)-β-Caryophyllene | 1421 | 1419 | 1597 | 6.9 | 3.2 | MS |
| 29 | β-Copaene | 1430 | 1426 | 1571 | 0.5 | — | MS |
| 30 | α-Humulene | 1455 | 1450 | 1666 | 1 | — | MS |
| 31 | Alloaromadendrene | 1462 | 1458 | 1642 | 1 | 1.8 | MS |
| 32 | γ-Muurolene | 1474 | 1471 | 1681 | 0.8 | — | MS |
| 33 | Germacrene D | 1479 | 1480 | 1716 | 44.2 | 16.7 | MS |
| 34 | γ-Gurjunene | 1472 | 1480 | 1704 | — | 0.3 | MS |
| 35 | Ledene | 1494 | 1492 | 1695 | 0.5 | — | MS |
| 36 | α-Muurolene | 1496 | 1488 | 1723 | 0.8 | — | MS |
| 37 | Valencene | 1494 | 1496 | 1719 | 0.2 | — | MS |
| 38 | γ-Cadinene | 1507 | 1507 | 1752 | 1.1 | — | MS |
| 39 | Cis/trans calamenene | 1512 | 1510 | 1816 | 0.2 | — | MS |
| 40 | δ-Cadinene | 1520 | 1516 | 1748 | 3.9 | — | MS |
| 41 | trans-Cadina-1,4 Diene | 1523 | 1524 | 1763 | 0.1 | — | MS |
| 42 | α-Cadinene | 1534 | 1530 | 1748 | 0.4 | 0.6 | MS |
| 43 | E-Nerolidol | 1553 | 1547 | 2038 | 0.5 | — | MS |
| 44 | trans-Hydrate-sesquisabinene | 1543 | 1550 | 1990 | 0.8 | — | MS |
| 45 | Caryophyllenoxide | 1578 | 1575 | 1975 | 0.4 | — | MS |
| 46 | Zingiberenol | 1606 | 1608 | 2108 | 0.7 | — | MS |
| 47 | Zingiberen-2-ol | 1613 | 1615 | 2192 | 0.4 | — | MS |
| 48 | Epicubenol | 1623 | 1622 | 2051 | 0.3 | — | MS |
| 49 | τ-Cadinol | 1633 | 1628 | 2185 | 5.9 | — | MS |
| 50 | α-Cadinol | 1643 | 1642 | 2227 | 7.4 | — | MS |
| 51 | Oplopenol 1 | 1644 | 1644 | 2205 | 0.4 | — | MS |
| 52 | 14-Hydroxy-9-epi-E-caryophyllene | 1658 | 1657 | 2300 | 0.2 | — | MS |
| 53 | Eudesma-4(15)-7-dien-1-β-ol | 1671 | 1666 | 2348 | 0.3 | — | MS |
| 54 | E-Phytol | 2096 | 2098 | 2605 | 0.7 | — | MS |
| Total identification % | 90.5 | 89.6 | |||||
| Hydrocarbon compounds | 70.7 | 62.1 | |||||
| Oxygenated compounds | 19.8 | 27.5 | |||||
| Hydrocarbon monoterpenes | 4.6 | 37.8 | |||||
| Oxygenated sesquiterpenes | 17.3 | 0 | |||||
| Hydrocarbon sesquiterpenes | 66.1 | 24.3 | |||||
| Other oxygenated compounds | 2.5 | 27.5 | |||||
Thus, germacrene D and β-ocimene were the main volatile compounds identified in MOA. Germacrene D is a sesquiterpene, a volatile organic compound belonging to the terpene class, known to occur in numerous aromatic plants, derived from the mevalonate metabolic pathway common in terpenoid synthesis.64 This compound is well-known for its antimicrobial, anti-inflammatory, and antioxidant properties.65 It also plays a role in the defence of plants against pests and herbivores due to its ability to inhibit the development of pathogenic microorganisms. In cosmetics and pharmaceuticals, it is exploited for its skin-soothing and protective properties. β-Ocimene is a monoterpene, a volatile aromatic compound found in many plants, flowers, and fruits.66 It imparts a fresh, floral aroma to many natural essences and displays antifungal, antimicrobial, and antioxidant activities, which support its application in aromatherapy and the formulation of natural fragrances. Moreover, this terpene contributes to plant signalling, enhancing pollinator attraction while deterring herbivorous insects.66 In cosmetics and pharmaceuticals, β-ocimene is also valued for its soothing and protective qualities.
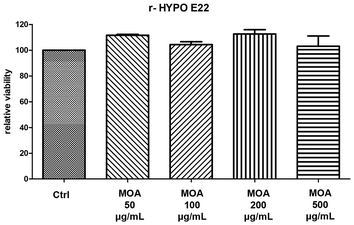
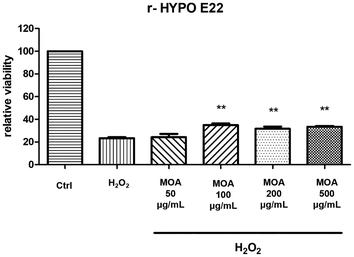
3.4. Antioxidant and neuroprotective activity of MOA hydroalcoholic extract
Throughout this study, biological interpretations are strictly limited to the results obtained with the hydroalcoholic extract, whereas the analysis of the non-polar fraction is intended solely to provide a comprehensive chemical and comparative characterisation of the species.
4. Conclusion
This study provides the first comprehensive metabolomic and biological characterization of Melissa officinalis subsp. altissima (MOA), integrating high-resolution NMR, GC-MS, and a range of in vitro and ex vivo assays. MOA exhibited a distinctive phytochemical profile highlighted by exceptionally high levels of rosmarinic acid along with abundant chlorogenic and caftaric acids, compounds well recognized for their potent antioxidant and neuroprotective properties. Remarkably, the lipophilic fraction was notable for a high content of β-sitosterol, coupled with a marked depletion of total and unsaturated fatty acids, pointing to a unique membrane-stabilizing phytosterol profile rarely reported in lemon balm species. The volatile fraction was dominated by germacrene and (E)-β-ocimene, molecules linked to antimicrobial and anti-inflammatory activities. Biological assays confirmed that the hydroalcoholic extract, characterised by a complex phytochemical composition, translates into functional antioxidant and neuroprotective effects. Hydroalcoholic extracts demonstrated a strong antioxidant capacity by protecting hypothalamic cells from hydrogen-peroxide–induced cytotoxicity, significantly down-regulating the pro-inflammatory mediators TNF-α and NOS-2, and increasing BDNF levels beyond control values. In cortical tissue challenged with β-amyloid, the extracts suppressed IL-6 and AChE expression, thus indicating a neuroprotective effect that may be related, at least in part, to the content of hydroxycinnamic acids and other bioactive constituents found in MOA acting within a complex phytochemical matrix, likely through synergistic mechanisms rather than the action of single compounds. Taken together, these findings highlight MOA as a promising source of bioactive compounds with antioxidant, anti-inflammatory, and neuroprotective properties. Its unique metabolic signature, particularly the enrichment in phytosterols and the distinctive volatile profile, supports its potential use as a functional ingredient for nutraceutical, pharmaceutical, and food applications, while underscoring the value of wild aromatic plants as reservoirs of health-promoting metabolites.However, this study has some limitations that should be acknowledged. Notably, the findings are based on in vitro and ex vivo assays, and no in vivo validation or bioavailability assessment has been performed. Therefore, while the data support potential functional benefits, causal relationships between individual compounds and the observed biological effects cannot be definitively established within the scope of the present study and should be further investigated in future studies with a dedicated bioactivity-guided fractionation and in vivo validation to confirm the physiological relevance and therapeutic potential of MOA.
Author contributions
Conceptualisation, D. A., and C. I.; data curation, D. A., F. M., and C. I.; formal analysis, D. A. and M. E. A.; funding acquisition, L. M., A. M. and C. I.; investigation, D. A., F. M., M. E. A., A. C., and C. D. S.; methodology, D. A. and C. I.; project administration, C. I., L. M., and A. M.; resources, L. M., C. F., and C. I.; supervision, C. I., and A. M.; validation, C. I., A. C., L. M., C. F.; visualisation, D. A., F. M., and C. I.; writing – original draft, D. A. and F. M.; writing – review and editing, D. A., and C. I. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data that support the findings of the study are available from the corresponding author upon reasonable request. For further information or to request access to the datasets, please contact cinzia.ingallina@uniroma1.it.Acknowledgements
This work was funded by the European Union—NextGenerationEU (the National Recovery and Resilience Plan (NRRP), Mission 4 Component 2 Investment 1.3—Call for tender no. 341 of 15 March 2022 of Italian Ministry of University and Research; Award Number: Project code PE00000003, Concession Decree No. 1550 of 11 October 2022 adopted by the Italian Ministry of University and Research, CUP D93C22000890001), Project title “ON Foods—Research and Innovation Network on Food and Nutrition Sustainability, Safety, and Security—Working ON Foods” and by the European Union—NextGenerationEU project within the Agritech National Research Center (the National Recovery and Resilience Plan (NRRP), Mission 4 Component 2 Investment 1.4—D.D. 1032 17/06/2022, CN00000022). This manuscript reflects only the authors’ views and opinions; neither the European Union nor the European Commission can be considered responsible for them. Health Operational Plan – Trajectory 5 “Nutraceuticals, Nutrigenomics, and Functional Foods” Action Line 5.1 “Development of an action program to combat all forms of malnutrition and to promote the principles of the Mediterranean Diet” Mediterranean Diet for Human Health Lab (MeDiHealthLab), Project code T5-AN-07, funded by the Italian Ministry of Health (Sapienza University of Rome, CUP B83C22005580006). PROGETTI MEDI ATENEO 2024—RM12419112B744FD.References
- G. Sabet Teymouri and M. Sabet Teimouri, The comparative effect of hydro alcoholic and hydro distillation extracts of Melissa officinalis on acne and pimple, Int. J. Pharmacol., Phytochem. Ethnomed., 2019, 12, 35, DOI:10.18052/www.scipress.com/ijppe.12.35.
- R. Chizzola, U. Lohwasser and C. Franz, Biodiversity within Melissa officinalis: Variability of bioactive compounds in a cultivated collection, Molecules, 2018, 23, 294, DOI:10.3390/molecules23020294.
- M. Usai, A. D. Atzei and M. Marchetti, A comparative study on essential oil intraspecific and seasonal variations: Melissa romana Mill. and Melissa officinalis L. from Sardinia, Chem. Biodivers., 2016, 13, e1500507, DOI:10.1002/cbdv.201500507.
- G. Petrisor, L. Motelica, L. N. Craciun, O. C. Oprea, D. Ficai and A. Ficai, Melissa officinalis: Composition, pharmacological effects and derived release systems—A review, Int. J. Mol. Sci., 2022, 23, 3591, DOI:10.3390/ijms23073591.
- S. G. Deans and G. Ritchie, Antibacterial properties of Melissa officinalis essential oil, Int. J. Food Microbiol., 1987, 5, 219–229, DOI:10.1016/0168-1605(87)90034-1.
- A. Shakeri, A. Sahebkar and B. Javadi, Melissa officinalis L. – A review of its traditional uses, phytochemistry and pharmacology, J. Ethnopharmacol., 2016, 188, 204–211, DOI:10.1016/j.jep.2016.05.010.
- M. Eivani and N. Khosronezhad, Melissa officinalis: A memory enhancer remedy, Physiol. Pharmacol., 2020, 24, 10, DOI:10.32598/ppj.24.3.10.
- M. Noguchi-Shinohara, K. Ono, T. Hamaguchi, T. Nagai, S. Kobayashi, J. Komatsu, M. Samuraki-Yokohama, K. Iwasa, K. Yokoyama, H. Nakamura and M. Yamada, Safety and efficacy of Melissa officinalis extract containing rosmarinic acid in the prevention of Alzheimer's disease progression, Sci. Rep., 2020, 10, 13792, DOI:10.1038/s41598-020-73729-2.
- K. Ono, K. Hasegawa, H. Naiki and M. Yamada, Curcumin has potent anti-amyloidogenic effects for Alzheimer's beta-amyloid fibrils in vitro, J. Neurosci. Res., 2004, 75, 742–750, DOI:10.1002/jnr.20025.
- F. Yang, G. P. Lim, A. N. Begum, O. J. Ubeda, M. R. Simmons, S. S. Ambegaokar, P. Chen, R. Kayed, C. G. Glabe, S. A. Frautschy and G. M. Cole, Curcumin inhibits formation of amyloid-β oligomers and fibrils, binds plaques, and reduces amyloid in vivo, J. Biol. Chem., 2005, 280, 5892–5901, DOI:10.1074/jbc.M404751200.
- Y. Liu, L. Zhang, Q. Li, T. Xue, F. Qin and Z. Xiong, Discovery of bioactive-chemical Q-markers of Acanthopanax sessiliflorus leaves: An integrated strategy of plant metabolomics, fingerprint and spectrum-efficacy relationship research, J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2024, 124009, DOI:10.1016/j.jchromb.2024.124009.
- D. Ambroselli, F. Masciulli, E. Romano, R. Guerrini, C. Ingallina, M. Spano and L. Mannina, NMR Metabolomics of Arctium lappa L., Taraxacum officinale and Melissa officinalis: A Comparison of Spontaneous and Organic Ecotypes, Foods, 2022, 13, 1642, DOI:10.3390/foods13111642.
- D. Jeanmonod and J. Gamisans, Flora of Corsica, 2007, p. 1 Search PubMed.
- E. G. Bligh and W. J. Dyer, A rapid method of total lipid extraction and purification, Can. J. Biochem. Physiol., 1959, 37, 911–917, DOI:10.1139/o59-099.
- H. van den Dool and P. D. Kratz, A generalization of the retention index system including linear temperature programmed gas–liquid partition chromatography, J. Chromatogr. A, 1963, 11, 463–471, DOI:10.1016/s0021-9673(01)80947-x.
- Y. Tine, A. Diallo, I. Ndoye, C. Gaye, B. Ndiaye, A. Diop, J. Costa, C. S. Bouh Boye, A. Wélé and J. Paolini, Chemical Variability and Antibacterial Activity of Eucalyptus, camaldulensis Essential Oils from Senegal, Int. J. Org. Chem., 2022, 12, 4014, DOI:10.4236/ijoc.2022.124014.
- R. P. Adams, Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th edn, Allured Publishing, Carol Stream, 2007, vol. 8 Search PubMed.
- NIST PC Version of the NIST/EPA/NIH Mass Spectral Library, 2008 Search PubMed.
- NIST, Spectral Database for Organic Compounds, 2008 Search PubMed.
- F. Masciulli, D. Ambroselli, M. L. Libero, A. Acquaviva, S. C. Di Simone, A. Chiavaroli, L. Menghini, C. Ferrante, G. Orlando, L. Mannina and C. Ingallina, Phytochemical composition and bioactivity of edible Taraxacum officinale: potential as an ingredient in brain health-oriented functional foods, Food Funct., 2025, 16, 7217–7229 RSC.
- A. Chiavaroli, F. Masciulli, C. Ingallina, L. Mannina, M. L. Libero, S. C. Di Simone, A. Acquaviva, Nilofar, L. Recinella, S. Leone, L. Brunetti, S. Carradori, L. Cantò, G. Orlando, G. Zengin, A. Ibrahim Uba, U. Cakilcioğlu, M. Mukemre, O. Elkiran, M. Di Vito, L. Menghini and C. Ferrante, Comprehensive metabolite and biological profile of “Sulmona Red Garlic” ecotype’s aerial bulbils, Food Res. Int., 2023, 164, 113654, DOI:10.1016/j.foodres.2023.113654.
- K. J. Livak and T. D. Schmittgen, Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method, Methods, 2001, 25, 402–408, DOI:10.1006/meth.2001.1262.
- S. Kleessen, C. Antonio, R. Sulpice, R. Laitinen, A. R. Fernie, M. Stitt and Z. Nikoloski, Structured patterns in geographic variability of metabolic phenotypes in Arabidopsis thaliana, Nat. Commun., 2013, 4, 2333, DOI:10.1038/ncomms2333.
- Z. Feng, L. Sun, M. Dong, S. Fan, K. Shi, Y. Qu, L. Zhu, J. Shi, W. Wang, Y. Liu, L. Song, Y. Weng, X. Liu and H. Ren, Novel players in organogenesis and flavonoid biosynthesis in cucumber glandular trichomes, Plant Physiol., 2023, 192, 1234–1248, DOI:10.1093/plphys/kiad236.
- S. Kim, M. H. Shin, M. A. Hossain, E. J. Yun, H. Lee and K. H. Kim, Metabolite profiling of sucrose effect on the metabolism of Melissa officinalis by gas chromatography-mass spectrometry, Anal. Bioanal. Chem., 2012, 404, 2103–2112, DOI:10.1007/s00216-011-4693-0.
- M. Kikowska, I. Kedziora, A. Krawczyk and B. Thiem, Methyl jasmonate, yeast extract and sucrose stimulate phenolic acids accumulation in Eryngium planum L. shoot cultures, Acta Biochim. Pol., 2014, 61, 395–401, DOI:10.18388/abp.2014_880.
- R. S. Payyavula, R. Shakya, V. G. Sengoda, J. E. Munyaneza, P. Swamy and D. A. Navarre, Synthesis and regulation of chlorogenic acid in potato: Rerouting phenylpropanoid flux in HQT-silenced lines, Plant Biotechnol. J., 2015, 13, 1234–1243, DOI:10.1111/pbi.12280.
- S. Kim, J. Kim, N. Kim, D. Lee, H. Lee, D. Y. Lee and K. H. Kim, Metabolomic Elucidation of the Effect of Sucrose on the Secondary Metabolite Profiles in Melissa officinalis by Ultraperformance Liquid Chromatography–Mass Spectrometry, ACS Omega, 2020, 5, 30322–30331, DOI:10.1021/acsomega.0c04745.
- C. Ceccanti, T. C. Finimundy and L. Barros, Nutritional Value of Wild and Domesticated Sanguisorba minor Scop. Plant, Horticulturae, 2023, 9, 560, DOI:10.3390/horticulturae9050560.
- S. A. Petropoulos, Â. Fernandes, M. I. Dias, C. Pereira, R. Calhelha, F. Di Gioia, N. Tzortzakis, M. Ivanov, M. Sokovic, L. Barros and I. C. F. R. Ferreira, Bioactive compounds and antioxidant activity of Melissa officinalis, Antioxidants, 2020, 9, 314, DOI:10.3390/antiox9040314.
- I. de Oliveira, A. Chrysargyris, S. A. Heleno, M. Carocho, R. C. Calhelha, M. I. Dias, J. Petrović, M. Soković, S. A. Petropoulos, C. Santos-Buelga, N. Tzortzakis and L. Barros, Wild and Cultivated Centaurea raphanina subsp. mixta: A Valuable Source of Bioactive Compounds, Food Res. Int., 2023, 164, 113044, DOI:10.1016/j.foodres.2023.113044.
- F. H. Awlqadr, A. B. Altemimi, S. A. Qadir, O. A. Mohammed, M. N. Saeed, M. A. Hesarinejad and N. Lakhssassi, Bioactive Compounds, Medicinal Benefits, and Contemporary Extraction Methods for Lemon Balm (Melissa officinalis), Food Sci. Nutr., 2025, 13, 70864, DOI:10.1002/fsn3.70864.
- J. J. Xu, X. Fang, C. Y. Li, L. Yang and X. Y. Chen, General and specialized tyrosine metabolism pathways in plants, BioMed. Res. Int., 2020, 2020, 123456, DOI:10.1007/s42994-019-00006-w.
- C. Chen, H. Naveed and K. Chen, Research progress on branched-chain amino acid aminotransferases, Front. Genet., 2023, 14, 1233669, DOI:10.3389/fgene.2023.1233669.
- M. M. Hasan, N. M. Alabdallah, B. M. Alharbi, M. Waseem, G. Yao, X. D. Liu, H. G. A. El-gawad, A. A. El-yazied, M. F. M. Ibrahim, M. S. Jahan and X. W. Fang, GABA: A Key Player in Drought Stress Resistance in Plants, Int. J. Mol. Sci., 2021, 22, 10136, DOI:10.3390/ijms221810136.
- V. Mohasseli, F. Farbood and A. Moradi, Antioxidant defense and metabolic responses of lemon balm (Melissa officinalis L.) to Fe-nano-particles under reduced irrigation regimes, Ind. Crops Prod., 2020, 147, 112338, DOI:10.1016/j.indcrop.2020.112338.
- S. Miraj, S. Rafieian-Kopaei and S. Kiani, Melissa officinalis L: A Review Study With an Antioxidant Prospective, J. Herb. Med., 2017, 10, 1–9, DOI:10.1177/2156587216663433.
- A. Binello, G. Cravotto, L. Boffa, L. Stevanato, M. Bellumori, M. Innocenti and N. Mulinacci, Efficient and selective green extraction of polyphenols from lemon balm, C. R. Chim., 2017, 20, 727–734, DOI:10.1016/j.crci.2017.06.003.
- K. Dastmalchi, H. J. D. Dorman, P. P. Oinonen, Y. Darwis, I. Laakso and R. Hiltunen, Chemical composition and in vitro antioxidative activity of a lemon balm (Melissa officinalis L.) extract, LWT, 2007, 41, 374–379, DOI:10.1016/j.lwt.2007.03.007.
- J. J. Ordaz, J. M. Hernández, J. Ramírez-Godínez, A. Castañeda-Ovando, L. G. González-Olivares and E. Contreras-López, Bioactive compounds in aqueous extracts of lemon balm (Melissa officinalis) cultivated in Mexico, Arch. Latinoam. Nutr., 2018, 68, 209–217, DOI:10.37527/2018.68.3.009.
- H. Guan, W. Luo, B. Bao, Y. Cao, F. Cheng, S. Yu, Q. Fan, L. Zhang, Q. Wu and M. Shan, A Comprehensive Review of Rosmarinic Acid: From Phytochemistry to Pharmacology and Its New Insight, Molecules, 2022, 27, 3292, DOI:10.3390/molecules27103292.
- A. Kola, G. Vigni, S. Lamponi and D. Valensin, Antioxidant and neuroprotective effects of Melissa officinalis extracts, Antioxidants, 2024, 13, 1419, DOI:10.3390/antiox13111419.
- A. Farah, M. Monteiro, C. M. Donangelo and S. Lafay, Chlorogenic acids from green coffee extract are highly bioavailable in humans, J. Nutr., 2008, 138, 2309–2315, DOI:10.3945/jn.108.095554.
- J. Zhong, Q. Ran, Y. Han, L. Gan and C. Dong, Biosynthetic Mechanisms of Plant Chlorogenic Acid from a Microbiological Perspective, Microorganisms, 2025, 13, 1114, DOI:10.3390/microorganisms13051114.
- M. Miao and L. Xiang, Pharmacological action and potential targets of chlorogenic acid, Adv. Pharmacol., 2020, 87, 105–130, DOI:10.1016/bs.apha.2019.12.002.
- M. Su, F. Liu, Z. Luo, H. Wu, X. Zhang, D. Wang, Y. Zhu, Z. Sun, W. Xu and Y. Miao, The Antibacterial Activity and Mechanism of Chlorogenic Acid Against Foodborne Pathogen Pseudomonas aeruginosa, Foodborne Pathog. Dis., 2020, 17, 545–554, DOI:10.1089/fpd.2019.2678.
- Z. Sun, X. Zhang, H. Wu, H. Wang, H. Bian, Y. Zhu, W. Xu, F. Liu, D. Wang and L. Fu, Antibacterial activity and action mode of chlorogenic acid against Salmonella Enteritidis, a foodborne pathogen in chilled fresh chicken, World J. Microbiol. Biotechnol., 2020, 36, 112, DOI:10.1007/s11274-020-2799-2.
- D. Wang, J. Hou, J. Wan, Y. Yang, S. Liu, X. Li, W. Li, X. Dai, P. Zhou, W. Liu and P. Wang, Dietary chlorogenic acid ameliorates oxidative stress and improves endothelial function in diabetic mice via Nrf2 activation, J. Int. Med. Res., 2020, 48, 1–12, DOI:10.1177/0300060520985363.
- N. Liang and D. D. Kitts, Role of Chlorogenic Acids in Controlling Oxidative and Inflammatory Stress Conditions, Nutrients, 2015, 8, 16, DOI:10.3390/nu8010016.
- Y. Yan, Q. Li, L. Shen, K. Guo and X. Zhou, Chlorogenic acid improves glucose tolerance, lipid metabolism, inflammation and microbiota composition in diabetic db/db mice, Front. Endocrinol., 2022, 13, 1042044, DOI:10.3389/fendo.2022.1042044.
- M. Yu, I. Gouvinhas, J. Rocha and A. I. R. N. A. Barros, Phytochemical and antioxidant analysis of medicinal and food plants towards bioactive food and pharmaceutical resources, Sci. Rep., 2021, 11, 89437, DOI:10.1038/s41598-021-89437-4.
- A. Arceusz and M. Wesolowski, Quality consistency evaluation of Melissa officinalis L. commercial herbs by HPLC fingerprint and quantitation of selected phenolic acids, J. Pharm. Biomed. Anal., 2013, 76, 162–170, DOI:10.1016/j.jpba.2013.05.020.
- F. Khallouki, A. Breuer, M. Akdad, F. E. Laassri, M. Attaleb, B. Elmoualij, M. Mzibri, L. Benbacer and R. W. Owen, Cytotoxic activity of Moroccan Melissa officinalis leaf extracts and HPLC-ESI-MS analysis of its phytoconstituents, Future J. Pharm. Sci., 2020, 6, 37, DOI:10.1186/s43094-020-00037-x.
- L. Goppa, M. Spano, R. M. Baiguera, M. Cartabia, P. Rossi, L. Mannina and E. Savino, NMR-Based Characterization of Wood Decay Fungi as Promising Novel Foods: Abortiporus biennis, Fomitopsis iberica and Stereum hirsutum Mycelia as Case Studies, Foods, 2023, 12, 2507, DOI:10.3390/foods12132507.
- H. Schaller, The role of sterols in plant growth and development, Phytochemistry, 2003, 63, 325–332, DOI:10.1016/S0163-7827(02)00047-4..
- J. N. Valitova, A. G. Sulkarnayeva and F. V. Minibayeva, Plant sterols: Diversity, biosynthesis, and physiological functions, Biochemistry, 2016, 81, 1234–1240, DOI:10.1134/S0006297916080046.
- Z. Li, B. Cheng, B. Yong, T. Liu, Y. Peng, X. Zhang, X. Ma, L. Huang, W. Liu and G. Nie, Metabolomics and physiological analyses reveal β-sitosterol as an important plant growth regulator inducing tolerance to water stress in white clover, Planta, 2019, 250, 1127–1137, DOI:10.1007/s00425-019-03277-1.
- E. Reszczyńska and A. Hanaka, Lipids Composition in Plant Membranes, Cell Biochem. Biophys., 2020, 38, 102–111, DOI:10.1007/s12013-020-00947-w.
- M. Souihi, R. Ben Ayed, I. Trabelsi, M. Khammassi, N. Ben Brahim and M. Annabi, Plant Extract Valorization of Melissa officinalis L. for Agroindustrial Purposes through Their Biochemical Properties and Biological Activities, J. Chem., 2020, 9728093, DOI:10.1155/2020/9728093.
- F. Ieri, L. Cecchi, P. Vignolini, M. F. Belcaro and A. Romani, HPLC/DAD, GC/MS and GC/GC/TOF analysis of Lemon balm (Melissa officinalis L.) sample as standarlized raw material for food and nutraceutical uses, Adv. Hortic. Sci., 2017, 31, 141–147, DOI:10.13128/ahs-21091.
- B. S. W. Dawson, R. A. Franich and R. Meder, Essential oil of Melissa officinalis L. subsp. altissima (Sibthr. et Smith) Arcang, Flavour Fragrance J., 1988, 3, 167–176, DOI:10.1002/ffj.2730030406.
- A. Miceli, C. Negro and L. Tommasi, Essential Oil of Melissa romana (Miller) Grown in Southern Apulia (Italy), J. Essent. Oil Res., 2006, 18, 300–305, DOI:10.1080/10412905.2006.9699144.
- M. Božović, S. Garzoli, A. Baldisserotto, C. Romagnoli, F. Pepi, S. Cesa, S. Vertuani, S. Manfredini and R. Ragno, Melissa officinalis L. subsp. altissima (Sibth. & Sm.) Arcang. essential oil: Chemical composition and preliminary antimicrobial investigation of samples obtained at different harvesting periods and by fractionated extractions, Ind. Crops Prod., 2018, 117, 358–366, DOI:10.1016/j.indcrop.2018.03.018.
- P. Steliopoulos, M. Wüst, K. P. Adam and A. Mosandl, Biosynthesis of the sesquiterpene germacrene D in Solidago canadensis: 13C and 2H labeling studies, Phytochemistry, 2002, 60, 643–651, DOI:10.1016/S0031-9422(02)00068-7.
- K. Noge and J. X. Becerra, Germacrene D, a common sesquiterpene in the genus Bursera (Burseraceae), Molecules, 2009, 14, 5289–5301, DOI:10.3390/molecules14125289.
- G. Farré-Armengol, I. Filella, J. Llusià and J. Peñuelas, β-Ocimene, a Key Floral and Foliar Volatile Involved in Multiple Interactions between Plants and Other Organisms, Molecules, 2017, 22, 1148, DOI:10.3390/molecules22071148.
- L. Safaeian, S. E. Sajjadi, S. H. Javanmard, H. Montazeri and F. Samani, Protective effect of Melissa officinalis extract against H2O2-induced oxidative stress in human vascular endothelial cells, Res. Pharm. Sci., 2018, 11, 523–534, DOI:10.4103/1735-5362.192488.
- S. Sipos, E. A. Moacă, I. Z. Pavel, Ş. Avram, O. M. Creţu, D. Coricovac, R. M. Racoviceanu, R. Ghiulai, R. D. Pană, C. M. Şoica, F. Borcan, C. A. Dehelean and Z. Crăiniceanu, Melissa officinalis L. Aqueous Extract Exerts Antioxidant and Antiangiogenic Effects and Improves Physiological Skin Parameters, Molecules, 2021, 26, 2369, DOI:10.3390/molecules26082369.
- D. B. Magalhães, I. Castro, V. Lopes-Rodrigues, J. M. Pereira, L. Barros, I. C. F. R. Ferreira, C. P. R. Xavier and M. H. Vasconcelos, Melissa officinalis L. ethanolic extract inhibits the growth of a lung cancer cell line by interfering with the cell cycle and inducing apoptosis, Food Funct., 2018, 9, 2657–2667, 10.1039/c8fo00446c.
- T. T. Kuo, H. Y. Chang, T. Y. Chen, B. C. Liu, H. Y. Chen, Y. C. Hsiung, S. M. Hsia, C. J. Chang and T. C. Huang, Melissa officinalis Extract Induces Apoptosis and Inhibits Migration in Human Colorectal Cancer Cells, ACS Omega, 2020, 5, 22315–22323, DOI:10.1021/acsomega.0c04489.
- A. Jahanban-Esfahlan, S. Modaeinama, M. Abasi, M. M. Abbasi and R. Jahanban-Esfahlan, Anti Proliferative Properties of Melissa officinalis in Different Human Cancer Cells, Asian Pac. J. Cancer Prev., 2015, 16, 5703–5711, DOI:10.7314/APJCP.2015.16.14.5703.
- P. I. Costea, F. Hildebrand, A. Manimozhiyan, F. Bäckhed, M. J. Blaser, F. D. Bushman, W. M. De Vos, S. D. Ehrlich, C. M. Fraser, M. Hattori, C. Huttenhower, I. B. Jeffery, D. Knights, J. D. Lewis, R. E. Ley, H. Ochman, P. W. O'Toole, C. Quince, D. A. Relman, F. Shanahan, S. Sunagawa, J. Wang, G. M. Weinstock, G. D. Wu, G. Zeller, L. Zhao, J. Raes, R. Knight and P. Bork, Enterotypes in the landscape of gut microbial community composition, Nat. Microbiol., 2018, 3, 8–16, DOI:10.1038/s41564-017-0072-8.
- C. Nathan and A. Cunningham-Bussel, Beyond oxidative stress: an immunologist's guide to reactive oxygen species, Nat. Rev. Immunol., 2013, 13, 349–361, DOI:10.1038/nri3423.
- K. A. Frankola, N. H. Greig, W. Luo and D. Tweedie, Targeting TNF-α to elucidate and ameliorate neuroinflammation in neurodegenerative diseases, CNS Neurol. Disord.: Drug Targets, 2011, 10, 391–403, DOI:10.2174/187152711794653751.
- O. M. Iova, G. E. Marin, I. Lazar, I. Stanescu, G. Dogaru, C. A. Nicula and A. E. Bulboacă, Nitric Oxide/Nitric Oxide Synthase System in the Pathogenesis of Neurodegenerative Disorders—An Overview, Antioxidants, 2023, 12, 753, DOI:10.3390/antiox12030753.
- N. Draginic, V. Jakovljevic, M. Andjic, J. Jeremic, I. Srejovic, M. Rankovic, M. Tomovic, T. Nikolic Turnic, A. Svistunov, S. Bolevich and I. Milosavljevic, Melissa officinalis L. as a Nutritional Strategy for Cardioprotection, Front. Physiol., 2021, 12, 661778, DOI:10.3389/fphys.2021.661778.
- M. Kara, S. Sahin, F. Rabbani, E. Oztas, G. Hasbal-Celikok, E. Kanımdan, A. Kocyigit, A. Kanwal, U. Wade, A. Yakunina, F. Di Pierro and A. Khan, An in vitro analysis of an innovative standardized phospholipid carrier-based Melissa officinalis L. extract as a potential neuromodulator for emotional distress and related conditions, Front. Mol. Biosci., 2024, 11, 1359177, DOI:10.3389/fmolb.2024.1359177.
- S. Peng, J. Wuu, E. J. Mufson and M. Fahnestock, Precursor form of brain-derived neurotrophic factor and mature brain-derived neurotrophic factor are decreased in the pre-clinical stages of Alzheimer's disease, J. Neurochem., 2005, 93, 1412–1421, DOI:10.1111/j.1471-4159.2005.03135.x.
- J. Siuda, M. Patalong-Ogiewa, W. Żmuda, M. Targosz-Gajniak, E. Niewiadomska, I. Matuszek, H. Jędrzejowska-Szypułka and M. Rudzińska-Bar, Cognitive impairment and BDNF serum levels, Neurol. Neurochir. Pol., 2016, 51, 24–32, DOI:10.1016/j.pjnns.2016.10.001.
- M. Ozarowski, P. L. Mikolajczak, A. Piasecka, P. Kachlicki, R. Kujawski, A. Bogacz, J. Bartkowiak-Wieczorek, M. Szulc, E. Kaminska, M. Kujawska, J. Jodynis-Liebert, A. Gryszczynska, B. Opala, Z. Lowicki, A. Seremak-Mrozikiewicz and B. Czerny, Influence of the Melissa officinalis Leaf Extract on Long-Term Memory in Scopolamine Animal Model with Assessment of Mechanism of Action, J. Evidence-Based Complementary Altern. Med., 2016, 9729818, DOI:10.1155/2016/9729818.
- F. J. Mirza, S. Amber, Sumera, D. Hassan, T. Ahmed and S. Zahid, Rosmarinic acid and ursolic acid alleviate deficits in cognition, synaptic regulation and adult hippocampal neurogenesis in an Aβ1-42-induced mouse model of Alzheimer’s disease, Phytomedicine, 2021, 83, 153490, DOI:10.1016/j.phymed.2021.153490.
- F. J. Mirza and S. Zahid, Ursolic acid and rosmarinic acid ameliorate alterations in hippocampal neurogenesis and social memory induced by amyloid beta in mouse model of Alzheimer's disease, Front. Pharmacol., 2022, 13, 1058358, DOI:10.3389/fphar.2022.1058358.
- K. Dastmalchi, V. Ollilainen, P. Lackman, G. B. af Gennäs, H. J. D. Dorman, P. P. Järvinen, J. Yli-Kauhaluoma and R. Hiltunen, Acetylcholinesterase inhibitory guided fractionation of Melissa officinalis L, Bioorg. Med. Chem., 2009, 17, 6816–6824, DOI:10.1016/j.bmc.2008.11.034.
- N. M. Lyra e Silva, R. A. Gonçalves, T. A. Pascoal, R. A. S. Lima-Filho, E. de P. F. Resende, E. L. M. Vieira, A. L. Teixeira, L. C. de Souza, J. A. Peny, J. T. S. Fortuna, I. C. Furigo, D. Hashiguchi, V. S. Miya-Coreixas, J. R. Clarke, J. F. Abisambra, B. M. Longo, J. Donato, P. E. Fraser, P. Rosa-Neto, P. Caramelli, S. T. Ferreira and F. G. De Felice, Pro-inflammatory interleukin-6 signaling links cognitive impairments and peripheral metabolic alterations in Alzheimer's disease, Transl. Psychiatry, 2021, 11, 251, DOI:10.1038/s41398-021-01349-z.
- T. Hase, S. Shishido, S. Yamamoto, R. Yamashita, H. Nukima, S. Taira, T. Toyoda, K. Abe, T. Hamaguchi, K. Ono, M. Noguchi-Shinohara, M. Yamada and S. Kobayashi, Rosmarinic acid suppresses Alzheimer's disease development by reducing amyloid β aggregation by increasing monoamine secretion, Sci. Rep., 2019, 9, 45168, DOI:10.1038/s41598-019-45168-1.
- R. A. Quintanilla, D. I. Orellana, C. González-Billault and R. B. Maccioni, Interleukin-6 induces Alzheimer-type phosphorylation of tau protein by deregulating the cdk5/p35 pathway, Exp. Cell Res., 2004, 295, 245–257, DOI:10.1016/j.yexcr.2004.01.002.
- C. Thingore, V. Kshirsagar and A. Juvekar, Amelioration of oxidative stress and neuroinflammation in lipopolysaccharide-induced memory impairment using Rosmarinic acid in mice, Metab. Brain Dis., 2020, 36, 299–313, DOI:10.1007/s11011-020-00629-9.
- B. J. Jongkees, B. Hommel, S. Kühn and L. S. Colzato, Neurocognitive effects of lemon balm supplementation in humans, J. Psychiatr. Res., 2015, 69, 1–7, DOI:10.1016/j.jpsychires.2015.08.014.
- C. A. Schenck and H. A. Maeda, Tyrosine biosynthesis, metabolism, and catabolism in plants, Phytochemistry, 2018, 149, 82–102, DOI:10.1016/j.phytochem.2018.02.003.
- P. Sengupta, R. Das, P. Majumder and D. Mukhopadhyay, Connecting the ends: signaling via receptor tyrosine kinases and cytoskeletal degradation in neurodegeneration, Explor. Foods, 2024, 2, 183–194, DOI:10.37349/en.2024.00033.
- E. Romano, G. Domínguez-Rodríguez, L. Mannina, A. Cifuentes and E. Ibáñez, Sequential Obtention of Blood–Brain Barrier-Permeable Non-Polar and Polar Compounds from Salvia officinalis L. and Eucalyptus globulus Labill. with Neuroprotective Purposes, Int. J. Mol. Sci., 2025, 26, 601, DOI:10.3390/ijms26020601.
- M. Koga, S. Nakagawa, A. Kato and I. Kusumi, Caffeic acid reduces oxidative stress and microglial activation in the mouse hippocampus, Tissue Cell, 2019, 60, 14–20, DOI:10.1016/j.tice.2019.07.006.
- M. A. Shah, J. B. Kang and P. O. Koh, Chlorogenic acid modulates the ubiquitin-proteasome system in stroke animal model, Lab. Anim. Res., 2022, 38, 41, DOI:10.1186/s42826-022-00151-2.
- J. D. Sanchez-Martinez, M. Bueno, G. Alvarez-Rivera, J. Tudela, E. Ibanez and A. Cifuentes, In vitro neuroprotective potential of terpenes from industrial orange juice by-products, Food Funct., 2020, 12, 302–314, 10.1039/d0fo02809f.
- A. Kola, G. Vigni, S. Lamponi and D. Valensin, Protective Contribution of Rosmarinic Acid in Rosemary Extract Against Copper-Induced Oxidative Stress, Antioxidants, 2020, 13, 1419, DOI:10.3390/antiox13111419.
- M. Ayaz, A. Wadood, A. Sadiq, F. Ullah, O. Anichkina and M. Ghufran, In-silico evaluations of the isolated phytosterols from polygonum hydropiper L against BACE1 and MAO drug targets, J. Biomol. Struct. Dyn., 2021, 40, 10230–10238, DOI:10.1080/07391102.2021.1940286.
- N. Sharma, M. A. Tan and S. S. A. An, Phytosterols: Potential Metabolic Modulators in Neurodegenerative Diseases, Int. J. Mol. Sci., 2021, 22, 12255, DOI:10.3390/ijms222212255.
- C. Shi, F. Wu, X. Zhu and J. Xu, Incorporation of beta-sitosterol into the membrane increases resistance to oxidative stress and lipid peroxidation via estrogen receptor-mediated PI3K/GSK3beta signaling, Biochem, Biophys. Acta, 2013, 1830, 2538–2544, DOI:10.1016/j.bbagen.2012.12.012.
| This journal is © The Royal Society of Chemistry 2026 |