Open Access Article
Open Access ArticleImmunoprogramming by maternal fibre and polyphenol intake enhances humoral systemic and mucosal immunity in adult offspring
Sergi Casanova-Crespo ab,
Daniela Ceballos-Sánchez
ab,
Daniela Ceballos-Sánchez ab,
Maria José Rodríguez-Lagunas
ab,
Maria José Rodríguez-Lagunas ab,
Malén Massot-Cladera
ab,
Malén Massot-Cladera ab,
Margarida Castell
ab,
Margarida Castell *abc and
Francisco José Pérez-Cano
*abc and
Francisco José Pérez-Cano ab
ab
aDepartment of Biochemistry and Physiology, Faculty of Pharmacy and Food Science, University of Barcelona, Barcelona, Spain. E-mail: margaridacastell@ub.edu
bInstitute of Research in Nutrition and Food Safety (INSA-UB), Santa Coloma de Gramenet, Spain
cCentro de Investigación Biomédica en Red de Fisiopatología de la Obesidad y la Nutrición (CIBEROBN), Instituto de Salud Carlos III, 28029 Madrid, Spain
First published on 30th April 2026
Abstract
Maternal diet is an element of the exposome that could shape the immune development in mothers’ offspring. Although diets containing fibre and polyphenols are known for their direct immunomodulatory properties, their influence on the immune status of the adult offspring of mothers following such diets remains unclear. This preclinical study aimed to establish whether maternal consumption of a fibre- and polyphenol-enriched (FP) diet during a period including pregestation, gestation, and lactation modulates the immune response to a specific antigenic challenge in the offspring later in life. For this purpose, female Wistar rats were fed either a reference or an FP diet across the above three periods. Their offspring, fed a standard diet after weaning, were immunized with ovalbumin (OVA) at the age of seven weeks. Offspring of FP-fed mothers exhibited a markedly enhanced humoral immune response, characterized by elevated levels of anti-OVA antibodies in both systemic and mucosal compartments. No significant differences were observed in the delayed-type hypersensitivity (DTH) response. Moreover, FP offspring displayed differential immunoglobulin (Ig) profiles in plasma and mesenteric lymph nodes. In parallel, an FP maternal diet changed the lymphoid composition in both the spleen and mesenteric lymph nodes of the offspring. In particular, there was a relatively increased proportion of CD8+ cells in TCRαβ+ and NKT subsets. These findings provide evidence that a maternal diet enriched with fibre and polyphenols can durably program offspring immunity, enhancing their capacity to mount more efficient humoral systemic and mucosal responses in adulthood.
1. Introduction
The exposome includes the full range of environmental exposures that a person experiences from conception onward, all of which can influence their health.1 Most studies focus on long-term chemical exposure in adult life, while evidence shows that some conditions during preconception, gestation, and lactation can influence the development and health of offspring once they have reached adulthood.2The exposome influences multiple physiological systems, including metabolic, cardiovascular, neurological, and immune systems, through diverse molecular pathways.3 Among them, epigenetic mechanisms, such as DNA methylation and histone modifications, play a central role in translating environmental signals into long-lasting biological effects.4,5 Although much attention has been paid to their influence on the metabolic and endocrine systems,6,7 growing evidence highlights the immune system as a key target of exposome-driven modulation.8 Among the environmental factors included in the exposome, diet stands out as a key modifiable determinant of health.6,9
Diets rich in fibre and polyphenols have been associated with beneficial effects on metabolic regulation, immune homeostasis, and inflammation control.10,11 Fibre, which is primarily composed of nondigestible carbohydrates, promotes gut health by modulating microbiota composition and increasing the production of short-chain fatty acids (SCFAs), which supports mucosal immunity, reduces systemic inflammation, and promotes regulatory T cell populations.12–14 Clinical studies have shown that a high fibre intake correlates with lower levels of systemic inflammatory markers and improved vaccine responses.15 Polyphenols, a diverse group of plant-derived compounds, including flavonoids, phenolic acids, and lignans, exhibit antioxidant, anti-inflammatory, and immunomodulatory properties,16 and some of them can also act as prebiotics by fostering beneficial microbiota and influencing the production of immunologically active microbial metabolites.17,18 Preclinical studies have demonstrated that certain polyphenol-rich diets can modulate dendritic cell activation, promote a regulatory immune environment, and reduce allergic sensitization.19,20 In addition, clinical studies report improvements in the outcomes of allergic diseases, such as asthma and allergic rhinitis.19,20 These findings consolidate the important role of dietary fibre and polyphenols in modulating immune responses, inflammatory status, and the capacity to mount effective and regulated antigen-specific immune reactions.
Nowadays, the critical role of maternal nutrition during sensitive periods such as gestation and lactation in shaping both the mother's and the offspring's metabolic and immune health is clear.21 For instance, the maternal diet during gestation can influence foetal growth, adiposity, glucose metabolism, and microbiota development, while nutrition during lactation may affect the composition of breast milk, the transfer of bioactive molecules, and early microbial colonization of the infant gut, all of which contribute to immunological and metabolic outcomes.22–24 However, despite increasing interest, fewer studies have investigated the impact of nutrition during the preconception period, a crucial but underexplored window of developmental plasticity. Preliminary evidence suggests that dietary exposures during pregestation can influence oocyte quality, uterine immune readiness, and epigenetic programming, which may persist through gestation and impact the health of offspring later in life.25
It has been shown that maternal intake of fibre, polyphenols, or both during gestation and lactation modifies gene expression and the gut microbiota composition and enhances the quality of breast milk.26–28 For example, preclinical studies have demonstrated that maternal fibre supplementation during pregnancy increases the abundance of SCFA-producing bacteria, promoting regulatory immune responses and protecting offspring from infant allergic diseases.29 The maternal influence on the microbiota and breast milk composition extends to the functionality of the neonatal immune system.30,31 Similarly, polyphenol intake during pregnancy and lactation has been linked to enhanced mucosal immunity and the modulation of immune cell differentiation in offspring.32 In a clinical context, maternal adherence to a diet rich in fibre and polyphenols during pregnancy was associated with beneficial Ig and cytokine profiles in cord blood, suggesting a programming effect on the neonatal immune system.33 Together, these findings support the concept of maternal immunonutrition as a strategy for shaping immunoprogramming in offspring.
However, despite evidence of the benefits of a maternal diet in terms of early immune development, the extent to which a maternal diet enriched in fibre and polyphenols can influence specific immune challenges in offspring later in life remains unclear. In the present study, we hypothesize that maternal intake of fibre and polyphenols beginning before gestation and lasting until the end of the lactation period may exert long-term immunoprogramming effects on offspring that persist into adulthood. Therefore, the aim of this study was to ascertain, in a preclinical model, whether such a maternal dietary intervention influences the specific immune response of offspring to an antigenic challenge later in life at both systemic and mucosal immune sites.
2. Experimental
2.1. Diets
A standard diet (REF diet) based on the AIN-93G formulation34 (Envigo, Indianapolis, IN, USA) and a fibre and polyphenol-enriched diet (FP diet) were used (Table 1). The FP diet contained 8% inulin (Fibruline™, derived from chicory roots, Cosucra, Warcoing, Belgium) and 1% pectin (PE21006 from citrus fruits, Gojira Fine Chemicals, LLC, Mundelein, IL, USA) as additional fibre sources, and 0.5% polyphenols including catechin (C1251), epicatechin (E1753), hesperidin (H5254), naringenin (N5893), and quercetin (Q4951) (all sourced from Millipore Sigma, Madison, WI, USA). The pelleted diets were produced vacuum-packed to prevent oxidation and contamination by Envigo and stored at 4 °C until use. The types of fibre and polyphenols in the FP diet were established in accordance with the content of a healthy dietary pattern in a gestating/lactating Spanish Mediterranean cohort,35 and the doses of these components were established based on previous preclinical approaches.36| Components | REF diet (g kg−1) | FP diet (g kg−1) |
|---|---|---|
| Casein | 200 | 200 |
| L-Cysteine | 3 | 3 |
| Cornstarch flour | 379.186 | 289.186 |
| Inulin | 0 | 80 |
| Pectin | 0 | 10 |
| Maltodextrin | 132 | 132 |
| Sucrose | 100 | 100 |
| Cellulose | 50 | 50 |
| Soybean oil | 70 | 70 |
| Mineral mix (TD94049) | 48 | 48 |
| Ferric citrate | 0.3 | 0.3 |
| Vitamin mix (TD94047) | 15 | 15 |
| Choline bitartrate | 2.5 | 2.5 |
| Tertiary butylhydroquinone | 0.014 | 0.014 |
| Polyphenols | 0 | 5 |
| Catechin | 0 | 1 |
| Epicatechin | 0 | 1 |
| Hesperidin | 0 | 1.5 |
| Naringenin | 0 | 0.75 |
| Quercetin | 0 | 0.75 |
2.2. Animals: mothers and offspring
Six-week-old female and male Wistar rats were obtained from Janvier Labs (Saint-Berthevin, France) and housed in the experimental animal facility of the Diagonal Campus at the University of Barcelona (UB). Female rats (n = 6 females per group) were randomly assigned to the REF group, fed the REF diet, or to the FP group, fed the FP diet. The diets were provided before gestation (3 weeks), during mating with a male (4 days), during gestation (3 weeks) and during lactation (3 weeks). The pups were born naturally, and on the same birthday, each litter was standardized to include 4 female and 4 male pups. Until day 21, the pups had unrestricted access to their mother's milk for nursing. On weaning day, 3–4 pups per dam were randomly selected from each litter (6 litters per diet), resulting in 18–24 offspring animals per group with a balanced sex distribution. All selected pups were kept on the REF diet until the end of the study. The required sample size was determined using the Appraising Project Office's program from the Universidad Miguel Hernández de Elche (Alicante, Spain). This calculation, based on intestinal IgA as a variable, among others, assumed no dropout rate and a two-sided type I error of 0.05. Additionally, the sample size was adjusted in accordance with the University Ethical Committee guidelines, to have animals from more than 5 different litters, adhering to the three Rs principle for animal experimentation.The animals were maintained under controlled environmental conditions (50–55% humidity, 21 ± 2 °C, and a 12 h light–dark cycle), and they had ad libitum access to food and water. The offspring body weight was monitored throughout the study from their birthday to their last day.
All animal procedures were approved by the Ethics Committee for Animal Experimentation of the University of Barcelona (CEEA/UB, ref. 240/19) and the Generalitat de Catalunya (DAAM10933).
2.3. Immunization with ovalbumin
At seven weeks of age, 19 animals born from mothers fed the REF diet and 20 born from mothers fed the FP diet were distributed into two subgroups: 12 were immunized (named the REF OVA group and the FP OVA group, according to their mothers’ diet) and 7–8 served as nonimmunized controls (named the REF group and the FP group, again depending on their mother's diet). Immunization was carried out with ovalbumin (OVA, grade V, Sigma-Aldrich, Madrid, Spain) emulsified in alum (Pierce, IL, USA) via the intraperitoneal (i.p.) route (500 µL per rat containing 50 µg of OVA and 20 mg of alum). Nonimmunized controls received the same volume of phosphate-buffered saline (PBS) via the i.p. route. After this single injection, immunization was followed over a four-week period, during which all animals (immunized and controls) were monitored, and blood samples were collected, from a saphenous vein at weeks 0, 2, 3, and 4 post-immunization as previously described.372.4. Delayed-type hypersensitivity (DTH) response
After 4 weeks of immunization, 24 h before euthanasia, the thickness of both ears of each animal was measured using a 7309 pocket thickness gauge (Mitutoyo, Hampshire, UK) to establish baseline values. The animals were immediately anesthetized with isoflurane (Abbott Laboratories, Berkshire, UK), and 50 µL of 1 mg mL−1 OVA solution in sterile PBS were injected into the right ear. The same volume of PBS was then injected into the left ear. After 24 h, ear thickness was measured again. In addition, biopsies from the ears of anesthetized rats were excised using a fixed-diameter punch, enabling the collection of ear samples with identical circular areas, which were immediately weighed. The results are expressed in terms of both the thickness and weight of the OVA and PBS ears of rats.2.5. Sample collection
After 4 weeks of immunization, animals were anesthetized using ketamine (90 mg kg−1) and xylazine (10 mg kg−1) (Bayer A.G., Leverkusen, Germany). Measures for calculating the body mass index (BMI) (weight/length2 (g cm−2)) and the Lee index ((weight0.33/length) × 1000 (g0.33 cm−1)) were carried out.Cardiac exsanguination was conducted to collect blood samples. EDTA-treated blood samples were centrifuged to obtain plasma. Mesenteric lymph nodes (MLNs), thymus, spleen, liver, brain, heart, kidney, submaxillary salivary gland and small and large intestines were collected. Some samples were weighed and, in some cases, immediately processed or stored at −20 °C or −80 °C for future analysis.
A central section of the small intestine (SI) was longitudinally opened, weighed and incubated with PBS while shaking at 37 °C for 10 min to collect the gut wash (GW) as in previous studies.38
The spleen, some MLNs, and thymus were processed as previously described to obtain lymphocytes.39 Briefly, tissues were ground using a 40 µm mesh cell strainer (Thermo Fisher Scientific, Barcelona, Spain) and Roswell Park Memorial Institute (RPMI) 1640 medium (Sigma-Aldrich, Madrid, Spain), enriched with 10% foetal bovine serum (FBS, Sigma-Aldrich), 100 IU mL−1 streptomycin–penicillin (Sigma-Aldrich), 2 mM L-glutamine (Sigma-Aldrich), and 0.05 mM 2-β-mercaptoethanol (Merck Millipore, Darmstadt, Germany). For lymphocyte isolation from the spleen, erythrocytes were removed via an additional osmotic lysis step.40 Cell count and viability were subsequently assessed using the Countess™ Automated Cell Counter (Invitrogen™, Thermo Fisher Scientific, Waltham, MA, USA).
Other MLNs were stored at −20 °C and later homogenized for Ig quantification as previously described.41
2.6. Lymphocytic composition in the spleen, MLNs, and thymus
The spleen, MLN and thymus lymphocyte subsets were characterized by flow cytometry analysis using mouse anti-rat monoclonal antibodies (mAbs) conjugated to fluorochromes, as previously described.40 The following mAbs were used: CD4 (OX-35), CD8α (OX-8), TCRαβ (R73), and CD161a (10/78) from eBioscience (Frankfurt, Germany); CD8β (341), TCRγδ (V65), CD25 (OX-39), CD44 (OX-49), and CD103 (OX-62) from BD Biosciences (San Diego, CA, USA); and CD45RA (OX-33) from Caltag (Burlingame, CA, USA). These mAbs were conjugated to one of the following fluorochromes: fluorescein isothiocyanate (FITC), phycoerythrin (PE), peridinin chlorophyll protein (PerCP), allophycocyanin (APC), or brilliant violet (BV) 421, BV 605, or BV786.Negative control staining using isotype-matched control mAbs was included in all samples. Additionally, a pool of cells was stained with each single fluorochrome-labelled mAb as a positive control. Data were acquired using an Aurora Cytek spectral flow cytometer (Cytek, Fremont, CA, USA) of the Flow Cytometry Unit (FCU) in the Cytometry Service of the Scientific and Technological Centres of the University of Barcelona (CCiT-UB, Barcelona, Spain) and analysed using FlowJo v.10 software (Tree Star, Inc., Ashland, OR, USA). Results are expressed as percentages of positive cells within the total lymphocyte population, gated based on forward- and side-scatter characteristics (FSC and SSC), or within a specific subpopulation.
2.7. Quantification of plasma and mucosal Ig concentrations
For plasma and homogenized MLNs, a ProcartaPlex™ Multiplex immunoassay was used to quantify the concentrations of IgA, IgM, IgG1, IgG2a, IgG2b, and IgG2c. The assay was carried out following the manufacturer's guidelines and as previously described.41 Results were obtained using a MAGPIX® analyser (Luminex Corporation, Austin, TX, USA) at the CCiT-UB. The sensitivity of the assay was as follows: 0.15 ng mL−1 for IgM; 1.16 ng mL−1 for IgG1; 2.08 ng mL−1 for IgG2a; 2.68 ng mL−1 for IgG2b; 4.21 ng mL−1 for IgG2c and 0.46 ng mL−1 for IgA.2.8. Quantification of plasma and mucosal anti-OVA antibodies
To determine the levels of specific anti-OVA antibodies (Abs) in plasma, GW, and homogenized MLNs, an indirect ELISA was performed as previously described.42 In addition, the concentrations of specific anti-OVA IgG1, IgG2a, IgG2b, and IgG2c Abs were also determined.43Briefly, plates were coated with OVA (10 µg mL−1 in PBS, overnight) and blocked with 0.5% gelatine in PBS (1 h). After washing, appropriately diluted samples and standards were added and incubated for 3 h. Thereafter, peroxidase-conjugated anti-rat Ig (Dako Cytomation, Glostrup, Denmark) was applied for 2 h for total anti-OVA Ab quantification, while biotin-conjugated anti-rat IgG1, IgG2a, IgG2b or IgG2c (BD Biosciences, San Diego, CA, USA) was added to assess the concentration of anti-OVA Abs from these isotypes. In this case, an additional incubation with peroxidase-conjugated ExtrAvidin (Sigma-Aldrich, Madrid, Spain) was needed. Bound peroxidase was evinced using H2O2 and a solution of o-phenylenediamine dihydrochloride as the chromogenic substrate. Absorbance was measured at 492 nm using a microplate photometer (Labsystems Multiskan, Helsinki, Finland), and results were expressed as arbitrary units (A.U.) relative to a standard constituted by a pool of OVA-immunized rat plasmas from a previous study,43 assigned a value of 800 A.U. mL−1 for total anti-OVA Abs and 100 A.U. mL−1 for each anti-OVA IgG isotype.
2.9. Statistical analysis
Statistical analysis was conducted using SPSS Statistics 22.0 software (SPSS Inc., Chicago, IL, USA). Prior to the main analysis, potential effects of sex and immunization status were assessed to determine whether data could be pooled. If no significant differences were observed between sexes, data were combined for further analysis. Normality and homogeneity of variances were evaluated using the Shapiro–Wilk and Levene tests, respectively. When data followed a normal distribution with homogeneous variance, a one-way ANOVA was performed, followed by Tukey's post hoc test for multiple comparisons. In cases where these assumptions were not met, the Kruskal–Wallis test was applied to assess significant differences (p < 0.05).Additionally, nonmetric multidimensional scaling (NMDS) was employed in RStudio (version 4.4.2, R Foundation for Statistical Computing, Vienna, Austria) using the ‘vegan’ package to identify clusters of sample similarities based on immune factor composition. The ‘envfit’ function was used to assess the association of factors with sample ordination in the NMDS plot. Statistical significance was defined as p < 0.05.
The effect of the maternal diet and OVA immunization was evaluated in offspring lymphocytes from the spleen, MLNs and thymus and Ig from plasma, MLNs and GW. In all cases, results for males and females in the same group did not differ; thus, data were gathered.
3. Results
3.1. Morphometric variables
The animals’ growth was monitored daily from day 1 until the last day of the study (week 11 of their life). The pups weighed between 5 and 7 g at birth and about 59–60 g at the end of the suckling period (week 3). By the end of the study, female rats weighed between 232 and 261 g, and males between 345 and 382 g. No differences in body weight during the study were observed due to the maternal diet or due to immunization at week 7 of life (Fig. 1).At the end of the study, BMI and the Lee index were calculated, and organ weights were recorded, with most showing no differences between groups (SI Table S1).
3.2. Lymphoid composition of the spleen, mesenteric lymph nodes and thymus
With regard to the spleen, no significant differences were observed in the proportions of TCRαβ+, TCRγδ+, NK, and NKT cells of the offspring due to the maternal FP diet or OVA immunization (Table 2). Nevertheless, B cell proportion increased in REF OVA animals with respect to the nonimmunized REF group. Within TCRαβ+ lymphocytes, the proportions of Th (CD4+) and Tc (CD8+) cells changed in the OVA group with the maternal FP diet, inducing a relatively higher CD8+ cell proportion and, consequently, a lower CD4+ cell percentage and CD4/CD8 ratio than in the REF OVA group. Similar results were found for the CD4+ and CD8+ subsets in the NKT population.| REF | FP | REF OVA | FP OVA | |
|---|---|---|---|---|
| The percentages of the main lymphocyte populations are expressed as cell proportions within the total lymphocyte gate, whereas the percentages of subsets are expressed as cell proportions within the particular indicated cells. Results are expressed as a percentage and mean ± SEM (n = 7–12). REF groups consisted of those born from mothers fed the reference diet; FP groups comprised those born from mothers fed a fiber- and polyphenol-enriched diet; and OVA groups were those that were immunized at week 7 of life. Statistical differences: *p < 0.05 vs. reference diet under the same immunized conditions; #p < 0.05 vs. not immunized under the same diet conditions. | ||||
| Main lymphocyte populations | ||||
| B cells (CD45RA+) | 31.03 ± 1.80 | 34.11 ± 2.59 | 40.15 ± 2.14# | 40.77 ± 3.96 |
| TCRαβ+ cells (TCRαβ+ NK−) | 42.10 ± 1.25 | 39.55 ± 1.98 | 37.79 ± 1.44 | 37.13 ± 2.49 |
| TCRγδ+ cells | 3.29 ± 0.31 | 3.05 ± 0.16 | 2.46 ± 0.15 | 2.42 ± 0.17 |
| NKT cells (TCRαβ+ NK+) | 13.85 ± 1.02 | 13.46 ± 0.85 | 11.69 ± 0.56 | 11.83 ± 1.31 |
| NK cells (TCRαβ− NK+) | 9.69 ± 0.94 | 10.53 ± 1.23 | 8.39 ± 0.56 | 7.92 ± 0.60 |
| Subsets within particular cells | ||||
| CD4+ in TCRαβ+ cells | 68.45 ± 1.55 | 67.98 ± 1.11 | 72.01 ± 1.14 | 66.95 ± 1.44* |
| CD8+ in TCRαβ+ cells | 29.78 ± 1.60 | 29.23 ± 1.70 | 26.00 ± 1.11 | 31.02 ± 1.35* |
| Ratio CD4/CD8 in TCRαβ+ cells | 2.33 ± 0.17 | 2.45 ± 0.12 | 2.84 ± 0.17 | 2.21 ± 0.14* |
| CD8+ in TCRγδ+ cells | 91.12 ± 1.05 | 90.94 ± 0.67 | 91.44 ± 0.58 | 91.46 ± 0.81 |
| CD4+ in NKT cells | 47.00 ± 0.96 | 47.95 ± 1.40 | 51.25 ± 1.39 | 45.16 ± 1.84* |
| CD8+ in NKT cells | 50.20 ± 1.04 | 49.35 ± 1.34 | 46.17 ± 1.31 | 52.02 ± 2.11* |
| CD8+ in NK cells | 37.38 ± 1.98 | 38.50 ± 1.83 | 29.97 ± 2.60# | 27.27 ± 2.57# |
When considering CD8+ cells in NK lymphocytes, the percentage decreased in both OVA-immunized groups with respect to their nonimmunized counterparts (Table 2).
With respect to the MLNs, the results are quite similar to those obtained for the spleen. Thus, no significant differences were found in the major lymphocyte populations, including B cells, TCRαβ+, NK, and NKT cells, among the experimental groups (Table 3). However, the immunization led to a lower percentage of TCRγδ+ cells in the REF OVA group than in the nonimmunized group (REF group). Furthermore, as also noted in the spleen, the CD8+ cell proportion within the TCRαβ+ population was higher and the CD4/CD8 ratio was lower in the FP OVA-immunized group than in the REF OVA-immunized group. Similarly, in NKT cells, OVA immunization caused a relatively higher proportion of CD8+ cells (and reciprocally a lower CD4+ cell percentage) in animals from FP mothers (FP OVA group vs. FP group).
| REF | FP | REF OVA | FP OVA | |
|---|---|---|---|---|
| The percentages of the main lymphocyte populations are expressed as cell proportions within the total lymphocyte gate, whereas the percentages of subsets are expressed as cell proportions within the particular indicated cells. Results are expressed as a percentage and mean ± SEM (n = 7–12). REF groups consisted of those born from mothers fed the reference diet; FP groups comprised those born from mothers fed a fiber- and polyphenol-enriched diet; and OVA groups were those that were immunized at week 7 of life. Statistical differences: *p < 0.05 vs. the reference diet under the same immunized conditions; #p < 0.05 vs. not immunized under the same diet conditions. | ||||
| Main lymphocyte populations | ||||
| B cells (CD45RA+) | 24.73 ± 1.08 | 28.63 ± 2.63 | 28.86 ± 1.93 | 30.70 ± 3.14 |
| TCRαβ+ cells (TCRαβ+ NK−) | 67.93 ± 2.01 | 63.54 ± 2.91 | 62.51 ± 1.92 | 61.07 ± 3.00 |
| TCRγδ+ cells | 1.70 ± 0.12 | 1.44 ± 0.13 | 1.21 ± 0.10# | 1.26 ± 0.05 |
| NKT cells (TCRαβ+ NK+) | 5.01 ± 0.84 | 6.06 ± 0.50 | 6.87 ± 0.42 | 6.50 ± 0.76 |
| NK cells (TCRαβ− NK+) | 2.34 ± 0.59 | 2.99 ± 0.40 | 2.89 ± 0.45 | 2.84 ± 0.36 |
| Subsets within particular cells | ||||
| CD4+ in TCRαβ+ cells | 71.50 ± 1.04 | 72.44 ± 0.83 | 72.87 ± 0.96 | 70.79 ± 1.16 |
| CD8+ in TCRαβ+ cells | 26.90 ± 0.89 | 26.05 ± 0.83 | 24.18 ± 0.97 | 27.60 ± 1.09* |
| Ratio CD4/CD8 in TCRαβ+ cells | 2.67 ± 0.13 | 2.81 ± 0.12 | 3.11 ± 0.17 | 2.60 ± 0.13* |
| CD8+ in TCRγδ+ cells | 93.24 ± 0.52 | 92.59 ± 1.32 | 93.20 ± 0.62 | 93.36 ± 0.66 |
| CD4+ in NKT cells | 64.55 ± 1.29 | 59.80 ± 1.60 | 58.38 ± 2.28 | 55.70 ± 2.38# |
| CD8+ in NKT cells | 36.38 ± 1.43 | 32.53 ± 1.32 | 38.79 ± 2.31 | 41.31 ± 2.11# |
| CD8+ in NK cells | 15.68 ± 1.00 | 10.62 ± 0.74* | 9.60 ± 0.92# | 10.43 ± 0.94 |
In NK cells, the percentage of CD8+ cells was lower due to the maternal FP diet in nonimmunized groups (FP group vs. REF group) and it was also lower after OVA immunization in animals from REF mothers (REF OVA group vs. REF group).
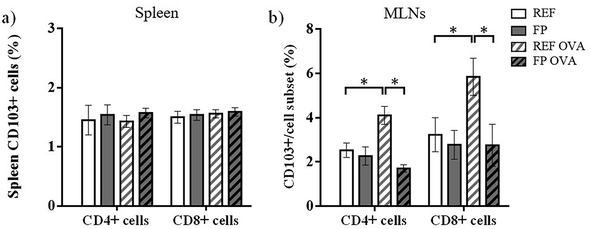
The proportion of CD103+ cells was also assessed in the T cell subsets of spleen and MLN cells. No changes were observed in CD103+ cells within T CD4+ and T CD8+ cell populations in the spleen (Fig. 2). In contrast, in the MLNs, OVA immunization in REF animals (REF OVA group) led to an increase in CD103+ cell proportions in T CD4+ and T CD8+ lymphocytes. However, this increase was not observed in the offspring of mothers fed the FP diet.
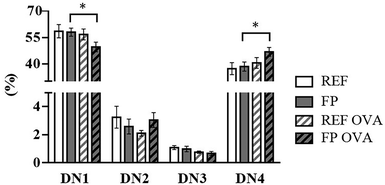
In the thymus, T cell maturation was assessed by analysing the proportions of CD4+ and CD8+ subsets (SI Fig. S1). No differences were observed between groups in the proportions of cells with a different degree of maturation, i.e., CD4−CD8− (DN), CD4+CD8+ (DP), CD4+CD8− (CD4+ SP), and CD4−CD8+ (CD8+ SP) thymocytes. Within the DN subset, the proportion of CD25+ and CD44+ cells was also measured (Fig. 3). The proportion of DN1 cells (CD44+CD25−) was reduced in OVA-immunized FP animals compared to the nonimmunized FP group, whereas DN4 cell percentage (CD44−CD25−) was reciprocally increased. No significant changes were found in the DN2 cell proportion (CD44+CD25+) and DN3 cell proportion (CD44−CD25−) due to the maternal diet or OVA immunization.
3.3. Immunoglobulin concentration in plasma and mesenteric lymph nodes
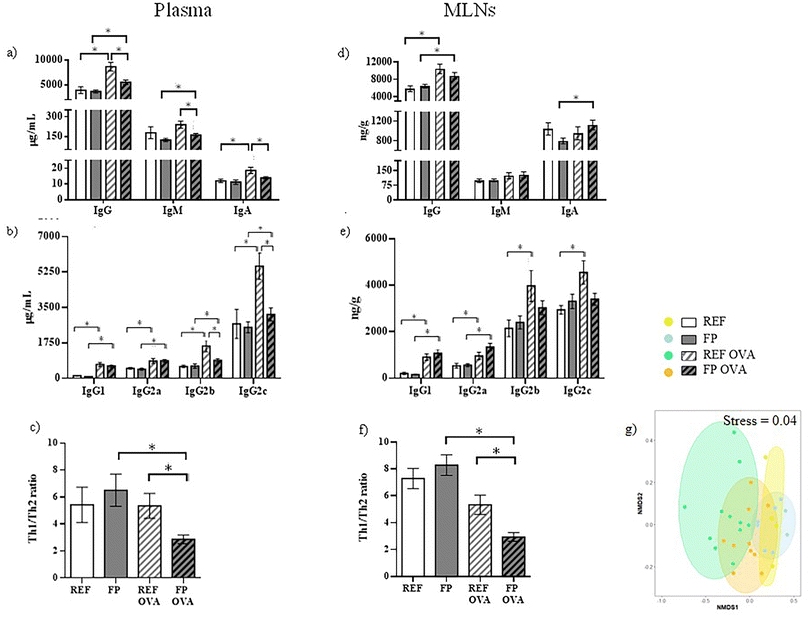
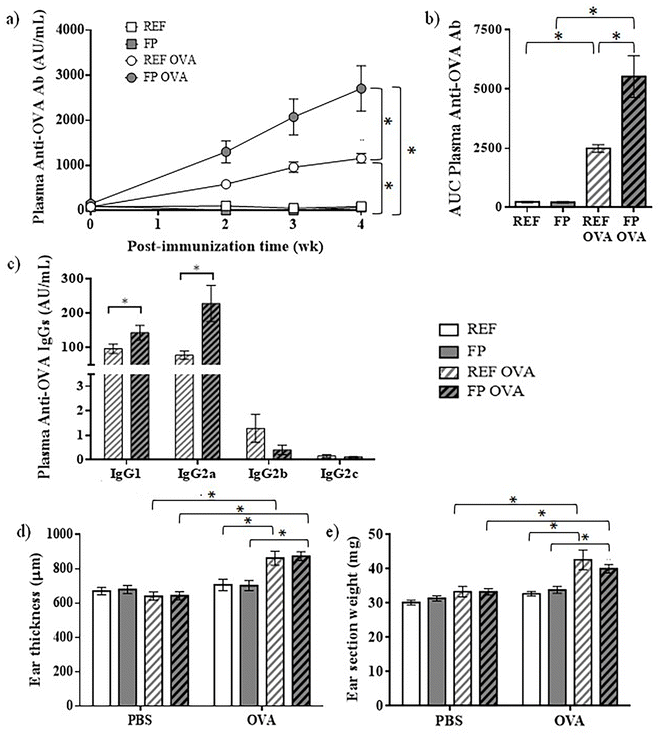
To explore the effect of an FP maternal diet and OVA immunization on the overall systemic humoral response, we first measured the plasma concentrations of IgG, IgM, and IgA, as well as IgG isotypes (IgG1, IgG2a, IgG2b and IgG2c) (Fig. 4a–c).In plasma, there was a significant increase in total IgG concentrations in both REF and FP OVA-immunized groups compared to their respective nonimmunized groups (Fig. 4a). This increase was due to elevated levels of all IgG subclasses (Fig. 4b), and was less pronounced in the FP OVA group, mainly due to lower concentrations of IgG2b and IgG2c compared to the REF OVA group. Given that, in rats, IgG2b and IgG2c are associated with a Th1-type response, and IgG1 and IgG2a with a Th2-type response,43–46 we calculated the ratio of IgG2b + IgG2c to IgG1 + IgG2a concentrations. The results indicated a lower ratio in the FP OVA group (Fig. 4c).
Regarding plasma IgM and IgA concentrations, higher levels of IgM were observed in FP OVA animals than in FP nonimmunized rats, while IgA increased in the REF OVA group with respect to the REF group but not in those animals receiving the FP diet (Fig. 4a).
A similar pattern was observed in the mucosal compartment estimated by Ig levels in MLNs, with both the REF OVA and FP OVA groups exhibiting a higher total IgG concentration than their nonimmunized counterparts (Fig. 4d), due to increased concentrations of IgG1 and IgG2a in both groups and IgG2b and IgG2c just in the REF OVA group (Fig. 4e). Similar to plasma, the ratio of Ig associated with Th1/Th2 immune responses also revealed a lower Th1/Th2 ratio in FP OVA animals in this compartment (Fig. 4f).
On the other hand, the low levels of IgM in MLNs did not change in response to the maternal diet or immunization, but the IgA concentration increased after immunization in those animals with the maternal FP diet with respect to the nonimmunized FP group (Fig. 4d).
In line with the above findings, the NMDS ordination analysis based on Ig concentrations in plasma and MLNs further supported the presence of group-specific Ig profiles. Pairwise PERMANOVA comparisons indicated no significant difference between the REF and FP non-immunized groups (p = 0.18), in line with their overlapping ordination. In contrast, the comparison between the REF OVA and FP OVA groups revealed a clear and statistically significant separation (p = 0.01). Likewise, REF animals immunized with OVA clustered apart from their non-immunized counterparts (REF vs. REF OVA, p = 0.001), and a similar pattern was observed within the FP background (FP vs. FP OVA, p = 0.04), indicating a clear impact of immunization on the overall Ig profile.
3.4. Humoral and cellular systemic immune responses against OVA
To assess the impact of the maternal diet in their adult progeny on the OVA-specific humoral immune response, total anti-OVA Abs were measured in offspring plasma over a period of 4 weeks (Fig. 5a and b).Immunized rats developed anti-OVA Abs, which were detected after 2 weeks of immunization and increased progressively until the end of the study (Fig. 5a). Interestingly, immunized offspring born from mothers fed the FP diet (FP OVA) exhibited consistently higher concentrations of anti-OVA Abs than the REF immunized group (REF OVA). This enhanced humoral response in the FP group was confirmed by a significantly greater AUC of the overall Ab response pattern (Fig. 5b). In addition, anti-OVA IgG isotypes were quantified in plasma at the final time point (Fig. 5c). Notably, the concentrations of IgG1 and IgG2a were higher in the FP OVA group than in the REF OVA group.
To assess the specific cellular response against OVA, a DTH reaction to OVA was carried out (Fig. 5d and e). Both immunized groups (REF OVA and FP OVA groups) showed a significant DTH response at the OVA-injected ear, with significantly increased ear thickness and section weight compared to their respective PBS-injected control ears and to nonimmunized siblings. In this case, no significant influence of the maternal diet was observed in the DTH response in the adult offspring.
3.5. Mucosal Ab response against OVA
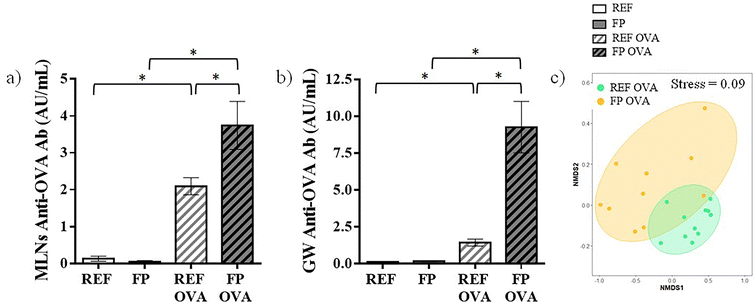
In addition to systemic responses, anti-OVA Ab levels were also assessed at the mucosal level at the final time point. Specifically, anti-OVA Ab concentration was measured in MLNs and GW (Fig. 6a and b). Similarly to the plasma findings, offspring from the FP OVA group exhibited higher anti-OVA Ab levels in both MLNs and GW than those from the REF OVA group. In line with these findings, the NMDS ordination analysis based on anti-OVA Ab concentrations (Fig. 6c) demonstrated a clear separation between the REF OVA and FP OVA groups. This separation was statistically significant (PERMANOVA, p = 0.0001), indicating that the overall Ab response profiles differed markedly between groups.4. Discussion
This study addresses the need to understand the influence of the maternal diet during pregestation, gestation, and lactation on the long-term immune system function of offspring, in particular, in shaping the specific immune response against an antigenic challenge in adult offspring. Here, we demonstrate that a maternal diet enriched in fibre and polyphenols (FP) during that perinatal period is able to enhance the immune response to an external challenge with ovalbumin (OVA) in adult offspring, highlighting the immunoprogramming effect of this particular diet.Enriching the maternal diet with fibre—specifically inulin and pectin—and polyphenols, including quercetin, naringenin, hesperidin, catechin, and epicatechin, was chosen for two reasons. First, these components reflect the composition of a healthy dietary pattern in a gestating/lactating Spanish Mediterranean cohort,35 ensuring physiological relevance and translational validity. Second, both dietary fibre and flavonoids, administered independently, have been documented as providing immunomodulatory benefits in both the mother and the offspring.47,48 For example, a GOS/inulin mixture administered during gestation and lactation in mice reduced IgE levels and clinical allergic responses in the offspring, promoting a more regulatory immune environment.47 Similarly, exposure to quercetin during gestation and lactation has been shown to modulate maternal–foetal interface immune cells, reducing inflammation, supporting T-cell homeostasis, and maintaining immune tolerance.48 Moreover, it has been stated that the direct intake of flavonoids can modulate the immune system by influencing immune cell activation, cytokine production, and Ab responses.49 Nevertheless, the use of dietary fibre and polyphenols together in dams and the effect on their offspring later in adulthood, particularly in the acquired immune response, have not been established before.
We have quantified the amount of specific anti-OVA Abs developed after 4 weeks of a single immunization. The offspring from FP-fed mothers exhibited higher levels of anti-OVA Abs in plasma than the offspring from REF-fed mothers. This enhanced immunity in adult offspring, despite no direct exposure to the FP diet, indicates that the maternal diet enriched in fibre and polyphenols, beginning before pregnancy and maintained throughout the entire pregnancy and lactation periods, caused immunological programming in the offspring. To the best of our knowledge, this is the first evidence indicating that such a diet can exert this type of long-term immunoprogramming effect. These findings, which demonstrate the potential of the maternal diet, are in line with the sharp contrast observed with those from maternal Western-style diet (WSD) interventions [reviewed in ref. 50].
The isotype of anti-OVA Abs was established at the end of the study. The levels of specific anti-OVA Abs belonging to IgG1 and IgG2a were markedly higher in all OVA-immunized animals compared to Abs belonging to IgG2b and IgG2c isotypes, which were scarcely detectable. This can be attributed to the use of alum as an adjuvant in the immunization, as it preferentially promotes a Th2-type immune response,51 which is usually responsible for fighting against parasites and extracellular infections.52,53 We found that the offspring born from FP-fed mothers exhibited even higher levels of anti-OVA IgG1 and IgG2a than immunized animals from REF-fed mothers. These findings are consistent with a study showing that intranasal immunization of mice with OVA plus polymerized caffeic acid, ferulic acid or p-coumaric acid elevated OVA-specific plasma IgG, particularly IgG1, showing the reinforcement of the immune response.54 In summary, a maternal diet enriched in fibre and polyphenols fed before gestation and maintained until the end of the lactation period enhances the specific humoral immune response boost with an adjuvant that stimulates Th2 immune responses. It remains to be determined whether such a maternal diet is also able to modulate Th1 immune responses using a suitable and specific adjuvant. Despite the enhanced specific humoral immunity, no differences were observed in the cellular response against OVA established by the DTH response depending on the maternal diet. This is consistent with previous reports indicating that the maternal diet tends to have a limited or modest impact on cellular immune responses such as DTH in both animal models and human populations.55,56
Besides the higher levels of specific anti-OVA Abs quantified in the plasmas of animals born from mothers consuming an FP diet, we also found enhanced levels of anti-OVA Abs in the MLNs and in the GW from the same animals. These findings indicate that the enhancement in the immune response to OVA also affects the mucosal compartment, in particular, the gut-associated lymphoid tissue (GALT). This compartmental analysis was further supported by NMDS ordination, which revealed a clear clustering of anti-OVA Ab profiles across plasma, MLNs, and GW, highlighting the coordinated enhancement of systemic and mucosal humoral responses induced by the maternal FP diet. Due to the connection of the GALT to other mucosal-associated lymphoid tissues,57 such as those present in the respiratory tract, boosting the immune system through the maternal diet could be very important and could have significant implications for fighting against the most common infections. Further studies must focus on establishing the effect of FP maternal diet on respiratory infection to demonstrate the potential of such a diet also at this level. The results obtained here are in line with studies that reported enhanced mucosal Ab responses in neonates after maternal fibre supplementation.58,59 However, no studies have focused on the effect of a maternal diet enriched in polyphenols in the mucosal immune system of the offspring.
In addition to the enhanced specific Ab response in systemic and mucosal compartments in the FP group. OVA immunization in both FP and REF groups led to an increase in the plasma and MLN concentrations of IgG1 and IgG2a. However, the content of IgG2b and IgG2c in plasma and MLNs was increased only in the REF group. The IgG isotypes made it possible to estimate the ratio of Th1-related isotypes (i.e., IgG2b and IgG2c) and Th2-related isotypes (i.e., IgG1 and IgG2b) in rats.43 The resulting ratio suggests that FP offspring had a Th2 enhanced immune response, which is consistent with the results concerning specific anti-OVA Abs in plasma and mucosal compartments and the Th2 promoting effect of the adjuvant used. Overall, the results obtained for specific and total Igs indicate that a maternal FP diet induces a more efficient immune response, which, in this case, is based on a higher Th2 immune response, and conversely, modulates the Th1 immune response that promotes inflammatory responses. Nevertheless, although this suggestion is in line with results obtained from a direct diet enriched in some polyphenols,60,61 as stated before, it remains to be determined what would happen with an immune challenge specific to a Th1 immune response.
The mechanisms involved in the immunoprogramming effect on the development of humoral immune response were not established here, but we can suggest some insights based on previous results at both preclinical and clinical levels. On the one hand, the maternal diet influences the placental transfer of bioactive compounds, such as Ig and cytokines.62 Moreover, after birth, maternal diet affects milk immune composition,63,64 and therefore it can modulate the offspring's immune system development. Additionally, the immunoprogramming effect could be a consequence of changes in the breast milk microbiota that are transferred from the mother to the infant, enhancing infant immunity among other functions as described.65 Lastly, progeny could undergo epigenetic changes due to maternal diet. It has been described that early life constitutes a “window of epigenetic plasticity”, during which direct or indirect dietary exposures can permanently alter gene expression patterns and influence long-term immunity. Both DNA methylation and microRNA changes, among other mechanisms, have been ascribed to polyphenols and other dietary components.66 Once the impact of the maternal dietary intervention on the offspring immunity has been demonstrated, further studies will focus on the mechanisms involved.
In addition to the impact of the maternal diet on humoral immune response, its influence on thymus, as primary lymphoid tissue, on the spleen, as secondary systemic lymphoid tissue, and on the MLNs, as secondary mucosal lymphoid tissue, was also established. The higher proportion of CD8+ cells within TCRαβ+ and NKT lymphocytes both in the spleen and in the MLNs of FP OVA-immunized offspring are in line with those obtained in a murine model, in which maternal supplementation of a strain of the probiotic Lactobacillus rhamnosus during gestation and lactation promoted the development of virus-specific CD8+ T cells in the offspring.67 On the other hand, we found no changes in the proportion of cells expressing CD103 in spleen T CD4+ and T CD8+ lymphocytes, indicating that maternal diet did not influence tissue-resident memory cells or regulatory T cells.68,69 Nevertheless, the proportion of CD103+ cells increased in MLNs after OVA immunization in REF animals but not in the FP group, indicating that the maternal FP diet affects these cells. Further studies must clarify the meaning of such an effect.
In the thymus, the lymphocyte maturation from double negative or DN cells (with DN1–DN4 stages), to double positive or DP cells (CD4+CD8+ cells), and, finally, single positive or SP cells (CD4+ cells and CD8+ cells) remained unchanged by the maternal diet. However, FP-OVA offspring showed a reduced proportion of DN1 cells and an increased percentage of DN4 cells with respect to their nonimmunized counterparts. This could indicate that OVA immunization in the offspring of FP mothers accelerates the thymocyte maturation in this particular early stage. Although more maturated thymic cells, such as CD8+ SP cells, were not affected by the maternal diet or immunization, the increase in DN4 cells could indicate that the maternal diet influences early T-cell development, potentially biasing the peripheral immune profile toward a cytotoxic CD8+ phenotype as observed in the spleen and MLNs. Further studies focused on the function of CD8+ cells might confirm such a hypothesis.
The influence of polyphenols and fibre present in the maternal diet on the lymphoid tissues of adult offspring could be attributed to the same mechanisms suggested before. Firstly, they could be derived from the presence of polyphenolic metabolites during gestation or lactation, through the placenta or breast milk, respectively, as it has been described.62,70 In addition, the maternal FP diet impacted its microbiota composition and, consequently, that from the litter in early life, leading to a more effective immune development.71 Finally, both SCFAs, derived from dietary fibre, and some polyphenols have epigenetic potential in terms of DNA acetylation/methylation or miRNA modulation.71,72 Consequently, future studies should focus in establishing the precise mechanisms involved in the effect of the maternal diet on lymphoid tissue composition and function.
Finally, it is important to emphasize that a maternal diet enriched in fibre and polyphenols during pregestation, gestation, and lactation did not influence the growth of the offspring. Minor, sex-dependent changes in relative organ weights were observed, suggesting subtle physiological effects without impacting overall somatic growth. It is relevant to explore whether a maternal diet influences offspring body weight, as maternal obesity and nutritional status have been shown to impact metabolic and immune outcomes in the progeny. Maternal obesity can predispose offspring to altered immune responses and increased risk of metabolic disorders, highlighting the importance of maternal nutrition beyond immune programming.73
5. Conclusions
This study demonstrates that maternal consumption of a diet enriched in fibre and polyphenols, from pregestation through lactation, enhances both systemic and mucosal humoral immune responses against an antigenic challenge in adult offspring. Our findings provide novel evidence that such maternal dietary patterns can induce long-term immunoprogramming effects, improving the capacity of offspring to mount effective specific immune responses.Although mechanistic studies are needed, this research may contribute to understanding the role of maternal nutrition in the long-term modulation of immune function and provide insights into preventive strategies for immune-related diseases.
Author contributions
Sergi Casanova-Crespo: writing – original draft, methodology, and formal analysis. Daniela Ceballos-Sánchez: investigation and methodology. Maria José Rodríguez-Lagunas: writing – review & editing and investigation. Malén Massot-Cladera: writing – review & editing, investigation, and methodology. Margarida Castell: writing – review & editing, investigation, and supervision. Francisco José Pérez-Cano: writing – review & editing, investigation, project administration, funding acquisition, and conceptualization.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available from the corresponding author, MC, upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo04563k.
Acknowledgements
This study was supported by grants PID2020-119602RB-I00 and PID2023-151804OB-I00 funded by MCIN/AEI/10.13039/501100011033, and the INSA-UB Maria de Maeztu Unit of Excellence (Grant CEX2021-001234-M) and funded by MICIN/AEI/FEDER, UE. SCC is a recipient of a doctoral fellowship from Ministry of Science, Innovation and Universities (PRE2022-102924). The authors would like to thank Nuria Rodríguez Martinez, who assisted in the animal handling, and Candela Veloso de Simón, who assisted in the flow cytometry analysis and performing ELISAs.References
- R. Vermeulen, E. L. Schymanski, A. L. Barabási and G. W. Miller, The exposome and health: Where chemistry meets biology, Science, 2020, 367(6476), 392–396, DOI:10.1126/science.aay3164 , PubMed PMID: 31974245.
- R. O. Wright, Environment, susceptibility windows, development and child health, Curr. Opin. Pediatr., 2017, 29(2), 211, DOI:10.1097/MOP.0000000000000465 , PubMed PMID: 28107208.
- K. K. Dennis, S. S. Auerbach, D. M. Balshaw, Y. Cui, M. D. Fallin and M. T. Smith, et al., The Importance of the Biological Impact of Exposure to the Concept of the Exposome, Environ. Health Perspect., 2016, 124(10), 1504–1510, DOI:10.1289/EHP140 , PubMed PMID: 27258438.
- I. Kimura, A. Ichimura, R. Ohue-Kitano and M. Igarashi, Free fatty acid receptors in health and disease, Physiol. Rev., 2020, 100(1), 171–210, DOI:10.1152/physrev.00041.2018 , PubMed PMID: 31487233.
- N. Acevedo, B. Alashkar Alhamwe, L. Caraballo, M. Ding, A. Ferrante and H. Garn, et al., Perinatal and Early-Life Nutrition, Epigenetics, and Allergy, Nutrients, 2021, 13(3), 724, DOI:10.3390/nu13030724 , PubMed PMID: 33668787; PubMed Central PMCID: PMC7996340.
- N. Haddad, X. D. Andrianou and K. C. Makris, A Scoping Review on the Characteristics of Human Exposome Studies, Curr. Pollut. Rep., 2019, 5(4), 378–393, DOI:10.1007/s40726-019-00130-7.
- J. Marín-Sáez, M. Hernández-Mesa, G. Cano-Sancho and A. M. García-Campaña, Analytical challenges and opportunities in the study of endocrine disrupting chemicals within an exposomics framework, Talanta, 2024, 279, 126616, DOI:10.1016/J.TALANTA.2024.126616 , PubMed PMID: 39067205.
- R. R. Dietert, Developmental immunotoxicology: Focus on health risks, Chem. Res. Toxicol., 2009, 22(1), 17–23, DOI:10.1021/tx800198m , PubMed PMID: 18783253.
- C. Selman, The dietary exposome: a brief history of diet, longevity, and age-related health in rodents, Clin. Sci., 2024, 138(21), 1343–1356, DOI:10.1042/CS20241248 , PubMed PMID: 39444221.
- C. A. Monteiro, G. Cannon, R. B. Levy, J. C. Moubarac, M. L. C. Louzada and F. Rauber, et al., Ultra-processed foods: what they are and how to identify them, Public Health Nutr., 2019, 22(5), 936, DOI:10.1017/S1368980018003762 , PubMed PMID: 30744710.
- A. Tresserra-Rimbau, Dietary Polyphenols and Human Health, Nutrients, 2020, 12(9), 2893, DOI:10.3390/NU12092893 , PubMed PMID: 32971874.
- N. Arpaia, C. Campbell, X. Fan, S. Dikiy, J. Van Der Veeken and P. Deroos, et al., Metabolites produced by commensal bacteria promote peripheral regulatory T cell generation, Nature, 2013, 504(7480), 451, DOI:10.1038/NATURE12726 , PubMed PMID: 24226773.
- Y. Q. Qin, L. Y. Wang, X. Y. Yang, Y. J. Xu, G. Fan and Y. G. Fan, et al., Inulin: properties and health benefits, Food Funct., 2023, 14(7), 2948–2968, 10.1039/D2FO01096H , PubMed PMID: 36876591.
- A. Koh, F. De Vadder, P. Kovatcheva-Datchary and F. Bäckhed, From dietary fiber to host physiology: Short-chain fatty acids as key bacterial metabolites, Cell, 2016, 165(6), 1332–1345, DOI:10.1016/j.cell.2016.05.041 , PubMed PMID: 27259147.
- J. L. S. Donadio, J. P. Fabi, M. B. Sztein and R. Salerno-Gonçalves, Dietary fiber pectin: challenges and potential anti-inflammatory benefits for preterms and newborns, Front. Nutr., 2024, 10, 1286138, DOI:10.3389/fnut.2023.1286138 , PubMed PMID: 38283907; PubMed Central PMCID: PMC10811139.
- H. Shakoor, J. Feehan, V. Apostolopoulos, C. Platat, A. S. A. Dhaheri and H. I. Ali, et al., Immunomodulatory Effects of Dietary Polyphenols, Nutrients, 2021, 13(3), 1–18, DOI:10.3390/NU13030728 , PubMed PMID: 33668814.
- R. Pei, X. Liu and B. Bolling, Flavonoids and gut health, Curr. Opin. Biotechnol, 2020, 61, 153–159, DOI:10.1016/J.COPBIO.2019.12.018 , PubMed PMID: 31954357.
- M. Massot-Cladera, À. Franch, F. J. Pérez-Cano and M. Castell, Cocoa and cocoa fibre differentially modulate IgA and IgM production at mucosal sites, Br. J. Nutr., 2016, 115(9), 1539–1546, DOI:10.1017/S000711451600074X , PubMed PMID: 26975903.
- A. Rauf, M. Imran, T. Abu-Izneid, Iahtisham-Ul-Haq, S. Patel and X. Pan, et al., Proanthocyanidins: A comprehensive review, Biomed. Pharmacother., 2019, 116, 108999, DOI:10.1016/j.biopha.2019.108999.
- T. B. Emran, T. A. Eva, M. Zehravi, F. Islam, J. Khan and S. Kareemulla, et al., Polyphenols as Therapeutics in Respiratory Diseases: Moving from Preclinical Evidence to Potential Clinical Applications, Int. J. Biol. Sci., 2024, 20(8), 3236–3256, DOI:10.7150/IJBS.93875 , PubMed PMID: 38904027.
- B. Koletzko, B. Brands, L. Poston, K. Godfrey and H. Demmelmair, Early nutrition programming of long-term health, Proc. Nutr. Soc., 2012, 71(3), 371–378, DOI:10.1017/S0029665112000596 , PubMed PMID: 22703585.
- E. C. Davis, V. P. Castagna, D. A. Sela, M. A. Hillard, S. Lindberg and N. J. Mantis, et al., Gut microbiome and breast-feeding: Implications for early immune development, J. Allergy Clin. Immunol., 2022, 150(3), 523–534, DOI:10.1016/J.JACI.2022.07.014 , PubMed PMID: 36075638.
- A. P. Starling, J. T. Brinton, D. H. Glueck, A. L. Shapiro, C. S. Harrod and A. M. Lynch, et al., Associations of maternal BMI and gestational weight gain with neonatal adiposity in the Healthy Start study1, Am. J. Clin. Nutr., 2014, 101(2), 302, DOI:10.3945/AJCN.114.094946 , PubMed PMID: 25646327.
- B. Caffé, A. Blackwell, B. D. Fehrenkamp, J. E. Williams, R. M. Pace and K. A. Lackey, et al., Human milk immune factors, maternal nutritional status, and infant sex: The INSPIRE Study, Am. J. Hum. Biol., 2023, 35(11), e23943, DOI:10.1002/ajhb.23943 , PubMed PMID: 37358306; PubMed Central PMCID: PMC10749986.
- M. del Campo Giménez, A. Fernández Bosch, C. Martínez de la Torre, M. Minuesa García, J. López-Torres Hidalgo and S. Córcoles García, et al., Impact of pregnancy on women's health-related lifestyle habits: Diet, physical activity, alcohol consumption, and smoking, Eur. J. Obstet. Gynecol. Reprod. Biol., 2024, 299, 182–187, DOI:10.1016/J.EJOGRB.2024.06.021 , PubMed PMID: 38878522.
- H. Szyller, K. Antosz, J. Batko, A. Mytych, M. Dziedziak and M. Wrześniewska, et al., Bioactive Components of Human Milk and Their Impact on Child's Health and Development, Literature Review, Nutrients, 2024, 16(10), 1487, DOI:10.3390/NU16101487 , PubMed PMID: 38794725.
- D. Ceballos-Sánchez, L. Sáez-Fuertes, S. Casanova-Crespo, M. J. Rodríguez-Lagunas, M. Castell and F. J. Pérez-Cano, et al., Influence of Dietary Fiber and Polyphenols During Pre-Gestation, Gestation, or Lactation on Intestinal Gene Expression, Nutrients, 2025, 17(2), 341, DOI:10.3390/nu17020341 , PubMed PMID: 39861471; PubMed Central PMCID: PMC11767784.
- J. Zhou, Y. Ren, J. Yu, Y. Zeng, J. Ren and Y. Wu, et al., The effect of maternal dietary polyphenol consumption on offspring metabolism, Crit. Rev. Food Sci. Nutr., 2024, 1(19), 1–18, DOI:10.1080/10408398.2024.2442539 , PubMed PMID: 39698806.
- R. A. Pretorius, M. Bodinier, S. L. Prescott and D. J. Palmer, Maternal Fiber Dietary Intakes during Pregnancy and Infant Allergic Disease, Nutrients, 2019, 11(8), 1767, DOI:10.3390/nu11081767 , PubMed PMID: 31374861; PubMed Central PMCID: PMC6722741.
- E. Cortes-Macías, M. Selma-Royo, I. García-Mantrana, M. Calatayud, S. González and C. Martínez-Costa, et al., Maternal Diet Shapes the Breast Milk Microbiota Composition and Diversity: Impact of Mode of Delivery and Antibiotic Exposure, J. Nutr., 2020, 151(2), 330, DOI:10.1093/JN/NXAA310 , PubMed PMID: 33188413.
- K. Rio-Aige, I. Azagra-Boronat, M. Massot-Cladera, M. Selma-Royo, A. Parra-Llorca and S. González, et al., Association of Maternal Microbiota and Diet in Cord Blood Cytokine and Immunoglobulin Profiles, Int. J. Mol. Sci., 2021, 22(4), 1778, DOI:10.3390/ijms22041778 , PubMed PMID: 33579027; PubMed Central PMCID: PMC7916816.
- C. A. Morais, L. M. Oyama, R. de Moura Conrado, V. V. de Rosso, C. O. do Nascimento and L. P. Pisani, Polyphenols-rich fruit in maternal diet modulates inflammatory markers and the gut microbiota and improves colonic expression of ZO-1 in offspring, Food Res. Int., 2015, 77, 186–193, DOI:10.1016/J.FOODRES.2015.06.043.
- T. Sasaki, M. Kawamura, C. Okuno, K. Lau, J. Riel and M. J. Lee, et al., Impact of Maternal Mediterranean-Type Diet Adherence on Microbiota Composition and Epigenetic Programming of Offspring, Nutrients, 2023, 16(1), 47, DOI:10.3390/nu16010047 , PubMed PMID: 38201877; PubMed Central PMCID: PMC10780434.
- P. G. Reeves, F. H. Nielsen and G. C. Fahey, AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet, J. Nutr., 1993, 123(11), 1939–1951, DOI:10.1093/jn/123.11.1939 , PubMed PMID: 8229312.
- I. García-Mantrana, C. Alcántara, M. Selma-Royo, A. Boix-Amorós, M. Dzidic and J. Gimeno-Alcañiz, et al., MAMI: a birth cohort focused on maternal-infant microbiota during early life, BMC Pediatr., 2019, 19, 140, DOI:10.1186/s12887-019-1502-y , PubMed PMID: 31053102; PubMed Central PMCID: PMC6498642.
- K. Rio-Aige, M. Selma-Royo, M. Massot-Cladera, A. Everard, M. Castell and M. J. Rodríguez-Lagunas, et al., A diet rich in fibre and vegetable protein during gestation and lactation shapes maternal immunity, intestinal microbiota and lipid metabolism, eBioMedicine, 2025, 117, 105784, DOI:10.1016/j.ebiom.2025.105784.
- M. Camps-Bossacoma, À. Franch, F. J. Pérez-Cano and M. Castell, Influence of Hesperidin on the Systemic and Intestinal Rat Immune Response, Nutrients, 2017, 9(6), 580, DOI:10.3390/nu9060580 , PubMed PMID: 28587283; PubMed Central PMCID: PMC5490559.
- M. d. M. Rigo-Adrover, K. van Limpt, K. Knipping, J. Garssen, J. Knol and A. Costabile, et al., Preventive Effect of a Synbiotic Combination of Galacto- and Fructooligosaccharides Mixture With Bifidobacterium breve M-16V in a Model of Multiple Rotavirus Infections, Front. Immunol., 2018, 9, 1318, DOI:10.3389/fimmu.2018.01318 , PubMed PMID: 29942312; PubMed Central PMCID: PMC6004411.
- M. Massot-Cladera, M. Abril-Gil, S. Torres, À. Franch, M. Castell and F. J. Pérez-Cano, Impact of cocoa polyphenol extracts on the immune system and microbiota in two strains of young rats, Br. J. Nutr., 2014, 112(12), 1944–1954, DOI:10.1017/S0007114514003080 , PubMed PMID: 25345541.
- S. Estruel-Amades, P. Ruiz-Iglesias, M. Périz, À. Franch, F. J. Pérez-Cano and M. Camps-Bossacoma, et al., Changes in Lymphocyte Composition and Functionality After Intensive Training and Exhausting Exercise in Rats, Front. Physiol., 2019, 10, 1491, DOI:10.3389/fphys.2019.01491 , PubMed PMID: 31920698; PubMed Central PMCID: PMC6928120.
- P. Ruiz-Iglesias, M. Massot-Cladera, M. J. Rodríguez-Lagunas, À. Franch, M. Camps-Bossacoma and M. Castell, et al., A Cocoa Diet Can Partially Attenuate the Alterations in Microbiota and Mucosal Immunity Induced by a Single Session of Intensive Exercise in Rats, Front. Nutr., 2022, 9, 861533, DOI:10.3389/fnut.2022.861533 , PubMed PMID: 35479747; PubMed Central PMCID: PMC9036086.
- M. Abril-Gil, A. Garcia-Just, F. J. Pérez-Cano, À. Franch and M. Castell, Development and characterization of an effective food allergy model in Brown Norway rats, PLoS One, 2015, 10(4), e0125314, DOI:10.1371/journal.pone.0125314 , PubMed PMID: 25923134; PubMed Central PMCID: PMC4414460.
- T. Pérez-Berezo, E. Ramiro-Puig, F. J. Pérez-Cano, C. Castellote, J. Permanyer and À. Franch, et al., Influence of a cocoa-enriched diet on specific immune response in ovalbumin-sensitized rats, Mol. Nutr. Food Res., 2009, 53(3), 389–397, DOI:10.1002/mnfr.200700396.
- J. Binder, E. Graser, W. W. Hancock, B. Wasowska, M. H. Sayegh and H. D. Volk, et al., Downregulation of intragraft IFN-gamma expression correlates with increased IgG1 alloantibody response following intrathymic immunomodulation of sensitized rat recipients, Transplantation, 1995, 60(12), 1516–1524, DOI:10.1097/00007890-199560120-00025 , PubMed PMID: 8545884.
- B. W. Bridle, B. N. Wilkie, A. M. Jevnikar and B. A. Mallard, Deviation of xenogeneic immune response and bystander suppression in rats fed porcine blood mononuclear cells, Transpl. Immunol., 2007, 17(4), 262–270, DOI:10.1016/j.trim.2007.01.010.
- A. Saoudi, I. Bernard, A. Hoedemaekers, B. Cautain, K. Martinez and P. Druet, et al., Experimental autoimmune myasthenia gravis may occur in the context of a polarized Th1- or Th2-type immune response in rats, J. Immunol. (Baltimore Md.: 1950), 1999, 162(12), 7189–7197 CAS . PubMed PMID: 10358165.
- G. Bouchaud, L. Castan, J. Chesné, F. Braza, P. Aubert and M. Neunlist, et al., Maternal exposure to GOS/inulin mixture prevents food allergies and promotes tolerance in offspring in mice, Allergy, 2016, 71(1), 68–76, DOI:10.1111/all.12777.
- W. Liu, Y. Zhou, Y. Qin, L. Yu, R. Li and Y. Chen, et al., Quercetin Intervention Alleviates Offspring's Oxidative Stress, Inflammation, and Tight Junction Damage in the Colon Induced by Maternal Fine Particulate Matter (PM2.5) Exposure through the Reduction of Bacteroides, Nutrients, 2020, 12(10), 3095, DOI:10.3390/nu12103095 , PubMed PMID: 33050645; PubMed Central PMCID: PMC7601264.
- G. Martínez, M. R. Mijares and J. B. D. Sanctis, Effects of Flavonoids and Its Derivatives on Immune Cell Responses, Recent Pat. Inflammation Allergy Drug Discovery, 13(2), 84–104, DOI:10.2174/1872213X13666190426164124.
- B. N. Nelson and J. E. Friedman, Developmental Programming of the Fetal Immune System by Maternal Western-Style Diet: Mechanisms and Implications for Disease Pathways in the Offspring, Int. J. Mol. Sci., 2024, 25(11), 11, DOI:10.3390/ijms25115951.
- D. H. Cribbs, A. Ghochikyan, V. Vasilevko, M. Tran, I. Petrushina and N. Sadzikava, et al., Adjuvant–dependent modulation of Th1 and Th2 responses to immunization with β–amyloid, Int. Immunol., 2003, 15(4), 505–514, DOI:10.1093/intimm/dxg049.
- H. Nakano, K. L. Lin, M. Yanagita, C. Charbonneau, D. N. Cook and T. Kakiuchi, et al., Blood-derived inflammatory dendritic cells in lymph nodes stimulate acute TH1 immune responses, Nat. Immunol., 2009, 10(4), 394–402, DOI:10.1038/ni.1707 , PubMed PMID: 19252492; PubMed Central PMCID: PMC2668134.
- W. E. Paul and J. Zhu, How are TH2-type immune responses initiated and amplified?, Nat. Rev. Immunol., 2010, 10(4), 225–235, DOI:10.1038/nri2735.
- R. Tada, M. Ogasawara, D. Yamanaka, Y. Sakurai, Y. Negishi and H. Kiyono, et al., Enzymatically polymerised polyphenols prepared from various precursors potentiate antigen-specific immune responses in both mucosal and systemic compartments in mice, PLoS One, 2021, 16(2), e0246422, DOI:10.1371/journal.pone.0246422 , PubMed PMID: 33556119; PubMed Central PMCID: PMC7870002.
- C. Venter, B. Pereira, K. Voigt, J. Grundy, C. B. Clayton and B. Higgins, et al., Factors associated with maternal dietary intake, feeding and weaning practices, and the development of food hypersensitivity in the infant, Pediatr. Allergy Immunol., 2009, 20(4), 320–327, DOI:10.1111/j.1399-3038.2008.00832.x.
- N. H. A. Suaini, Q. Y. Koh, J. Y. Toh, V. X. Soriano, M. T. Colega and C. Riggioni, et al., Maternal and Infant Dietary Patterns Are Not Related to Food Allergy Risk in Singapore Children: GUSTO Cohort Study, J. Nutr., 2024, 154(7), 2157–2166, DOI:10.1016/j.tjnut.2024.05.002.
- U. M. Mörbe, P. B. Jørgensen, T. M. Fenton, N. von Burg, L. B. Riis and J. Spencer, et al., Human gut-associated lymphoid tissues (GALT); diversity, structure, and function, Mucosal Immunol., 2021, 14(4), 793–802, DOI:10.1038/s41385-021-00389-4.
- M. Gomez de Agüero, S. C. Ganal-Vonarburg, T. Fuhrer, S. Rupp, Y. Uchimura and H. Li, et al., The maternal microbiota drives early postnatal innate immune development, Science, 2016, 351(6279), 1296–1302, DOI:10.1126/science.aad2571 , PubMed PMID: 26989247.
- Q. Li, S. Yang, X. Zhang, X. Liu, Z. Wu and Y. Qi, et al., Maternal Nutrition During Late Gestation and Lactation: Association With Immunity and the Inflammatory Response in the Offspring, Front. Immunol., 2022, 12, 758525, DOI:10.3389/fimmu.2021.758525 , PubMed PMID: 35126349; PubMed Central PMCID: PMC8814630.
- Y. Ysrafil, Z. Sapiun, N. S. Slamet, F. Mohamad, H. Hartati and S. A. Damiti, et al., Anti-inflammatory activities of flavonoid derivates, ADMET DMPK, 2023, 11(3), 331–359, DOI:10.5599/admet.1918 , PubMed PMID: 37829324; PubMed Central PMCID: PMC10567070.
- J. M. Al-Khayri, G. R. Sahana, P. Nagella, B. V. Joseph, F. M. Alessa and M. Q. Al-Mssallem, Flavonoids as Potential Anti-Inflammatory Molecules, A Review, Molecules, 2022, 27(9), 9, DOI:10.3390/molecules27092901.
- K. Rio-Aige, I. Azagra-Boronat, M. Massot-Cladera, M. Selma-Royo, A. Parra-Llorca and S. González, et al., Association of Maternal Microbiota and Diet in Cord Blood Cytokine and Immunoglobulin Profiles, Int. J. Mol. Sci., 2021, 22(4), 1778, DOI:10.3390/ijms22041778.
- K. Rio-Aige, M. Selma-Royo, M. Massot-Cladera, A. Everard, M. Castell and M. J. Rodríguez-Lagunas, et al., A diet rich in fibre and vegetable protein during gestation and lactation shapes maternal immunity, intestinal microbiota and lipid metabolism, eBioMedicine, 2025, 117, 105784, DOI:10.1016/j.ebiom.2025.105784.
- K. Rio-Aige, M. Selma-Royo, R. Cabrera-Rubio, S. González, C. Martínez-Costa and M. Castell, et al., Maternal diet shapes infant microbiota and defensive capacity against infections in early life via differential human milk composition, eBioMedicine, 2025, 118, 105850, DOI:10.1016/j.ebiom.2025.105850.
- M. LaTuga, A. Stuebe and P. Seed, A Review of the Source and Function of Microbiota in Breast Milk, Semin. Reprod. Med., 2014, 32(01), 068–073, DOI:10.1055/s-0033-1361824.
- R. M. Swanson, B. J. Davila Ruiz, J. L. Hurlbert, W. J. S. Diniz, P. Banerjee and C. Dahlen, et al., Livestock models of maternal nutrition and developmental programming, J. Dev. Orig. Health Dis., 2026, 17, e14, DOI:10.1017/S2040174426100452.
- C. Valentin, P. Brito Rodrigues, M. Verce, S. Delbauve, L. La Palombara and F. Demaret, et al., Maternal probiotic exposure enhances CD8 T cell protective neonatal immunity and modulates offspring metabolome to control influenza virus infection, Gut Microbes, 2025, 17(1), 2442526, DOI:10.1080/19490976.2024.2442526.
- X. Zhang, X. Ouyang, Z. Xu, J. Chen, Q. Huang and Y. Liu, et al., CD8+CD103+ iTregs Inhibit Chronic Graft-versus-Host Disease with Lupus Nephritis by the Increased Expression of CD39, Mol. Ther., 2019, 27(11), 1963–1973, DOI:10.1016/j.ymthe.2019.07.014.
- X. Pan, F. Shi, S. Tang, M. Liu, L. Pan and G. Liang, et al., Characteristics of CD103+CD8+ T cells in the spleen of Plasmodium yoelii NSM-infected mice, Front. Cell. Infect. Microbiol., 2025, 15, 1668438, DOI:10.3389/fcimb.2025.1668438.
- A. López-Yerena, B. Grases-Pintó, S. Zhan-Dai, F. J. Pérez-Cano, R. M. Lamuela-Raventos and M. J. Rodríguez-Lagunas, et al., Nutrition during pregnancy and lactation: New evidence for the vertical transmission of extra virgin olive oil phenolic compounds in rats, Food Chem., 2022, 391, 133211, DOI:10.1016/j.foodchem.2022.133211.
- N. C. Rubas, A. Torres and A. K. Maunakea, The Gut Microbiome and Epigenomic Reprogramming: Mechanisms, Interactions, and Implications for Human Health and Disease, Int. J. Mol. Sci., 2025, 26(17), 8658, DOI:10.3390/ijms26178658.
- W. Andonotopo, M. A. Bachnas, J. Dewantiningrum, M. B. Adi Pramono, S. Sulistyowati and I. N. Hariyasa Sanjaya, et al., Nutriepigenomics in perinatal medicine: maternal nutrition as a modulator of fetal gene expression and long-term health, J. Perinat. Med., 2025, 54(2), 244–257, DOI:10.1515/jpm-2025-0289.
- P. M. Catalano and K. Shankar, Obesity and pregnancy: mechanisms of short term and long term adverse consequences for mother and child, Br. Med. J., 2017, 356, j1, DOI:10.1136/bmj.j1 , PubMed PMID: 28179267.
| This journal is © The Royal Society of Chemistry 2026 |