Open Access Article
Open Access ArticleSynergistic sleep-promoting effects of magnesium and apigenin in normal and insomnia mouse models
Wing-Yan
Wong
ab,
Brandon Dow
Chan
 ab,
Hengshen
Zhou
ab,
Tsz Chun
Leung
ab and
William Chi-Shing
Tai
ab,
Hengshen
Zhou
ab,
Tsz Chun
Leung
ab and
William Chi-Shing
Tai
 *abc
*abc
aDepartment of Applied Biology and Chemical Technology, The Hong Kong Polytechnic University, Hung Hom, Hong Kong S.A.R., China. E-mail: william-cs.tai@polyu.edu.hk
bThe Laboratory for Probiotic and Prebiotic Research in Human Health, The Hong Kong Polytechnic University, Hung Hom, Hong Kong S.A.R., China
cState Key Laboratory of Chinese Medicine and Molecular Pharmacology (Incubation), Shenzhen Research Institute, The Hong Kong Polytechnic University, Shenzhen, China
First published on 17th February 2026
Abstract
Insomnia is a prevalent condition affecting sleep quality and overall health. While apigenin and magnesium individually exhibit sleep-promoting properties, their combined potential remains unexplored. This study investigates the combinatory effects of apigenin and magnesium (A + M) on sleep. Results demonstrated that A + M significantly enhanced sleep duration in normal mice (44%, p < 0.005), caffeine-induced sleep disturbed mice (32%, p < 0.001), and p-chlorophenylalanine (PCPA)-induced insomnia mice (37%, p < 0.05), surpassing individual treatments of apigenin or magnesium. Mechanistically, A + M treatment suppressed systemic and hypothalamic TNFα levels in PCPA-induced mice. We further demonstrated A + M treatment could inhibit microglial activation, potentially via suppressing NFκB signaling. These findings demonstrate the combinatory efficacy of A + M in restoring sleep architecture and suggest its potential as a natural, effective intervention for insomnia management.
1. Introduction
Insomnia is a sleep disorder characterized by difficulties in initiating sleep, maintaining sleep, as well as early morning awakening. To qualify, such symptoms must occur at least three times per week for at least three months.1 Despite affecting 28.3% of adults in the Asian population, insomnia is not immediately life-threatening and may therefore be under-diagnosed and under-treated.2 Moreover, insomnia rarely exists in isolation and frequently appears alongside metabolic dysfunctions, including cardiovascular disease, obesity, and diabetes,3 as well as psychological conditions including anxiety and depression.4Currently, subjective complaints about sleep quality and duration contribute significantly to disease prognosis.5 Even with the aid of polysomnographic assessment, which provides a more objective evaluation of sleep impairment, high costs of time and equipment, as well as brain wave variations across individuals remain significant barriers to insomnia diagnosis. For clinical management, cognitive behavioural therapy for insomnia (CBT-I) remains the first-line treatment due to its non-invasive nature. When CBT-I proves insufficient, medications such as benzodiazepine, gamma-aminobutyric acid (GABA), and melatonin receptor agonists are used in conjunction with CBT to enhance sleep outcomes.6
Increasing evidence suggests that there is bi-directional communication between sleep and the immune system, linking insomnia with neuroinflammation.7 Increased pro-inflammatory cytokine levels have been identified in patients with chronic insomnia,8 and insomnia may increase the risk of inflammatory diseases.9,10 Treatment of inflammatory diseases with anti-inflammatory therapeutics has led to improved sleep duration and quality,11–13 hinting at the developmental potential of anti-inflammatory therapeutics for insomnia.
Apigenin, a naturally occurring flavonoid found abundantly in chamomile, exhibits antidepressant effects;14,15 it has also been shown to improve insomnia in mouse models of p-chlorophenylalanine (PCPA)-induced sleep disorders.16 In addition to its ability to improve sleep, apigenin has also exhibited anti-inflammatory effects in vivo.17 Together, these indicate the high potential of apigenin for development as a hypnotic agent.
Magnesium is the fourth most abundant ion in the body. As it participates in numerous enzymatic reactions, magnesium is important for proper bodily function, including synaptic activity.18 In addition, magnesium has been suggested to play an important role in sleep, by regulating neurotransmitters such as GABA and the enzyme N-acetyltransferase, which converts 5-hydroxytryptamine (5-HT) into N-acetyl-5-hydroxytryptamine and then to melatonin.19,20 Low GABA and melatonin levels have been implicated in sleep disorders,21,22 and magnesium supplementation can lead to improved sleep outcomes, including increased sleep duration and better overall sleep quality.23 Collectively, these findings underline magnesium's key role in sleep regulation and its therapeutic value in addressing sleep deficiencies.
Here, we aimed to evaluate the sleep-promoting effects of apigenin and magnesium in combination. Using multiple mouse models of sleep disturbance, we assessed its impact on sleep duration and investigated the underlying mechanisms involved.
2. Materials and methods
2.1 Animals
Male C57BL/6J wild-type mice were purchased from the Jackson Laboratory (Bar Harbor, ME, USA). An inbred colony was maintained in the Centralised Animal Facilities at The Hong Kong Polytechnic University. Mice were kept under constant temperature and humidity (22 ± 2 °C; 65 ± 5%), and on a 12-hour light/dark cycle. Food and water were provided ad libitum. All animal experiments were approved by the Animal Subjects Ethics Sub-Committee (ASESC) of The Hong Kong Polytechnic University and adhered to the guidelines of Institutional Guidelines and the Animal Ordinance of the Department of Health, Hong Kong S.A.R.2.2 Animal treatment and pentobarbital-induced testing
8 weeks-old male C57BL/6J mice were randomly assigned to control (water), vehicle (water) apigenin (A; 4 mg kg−1), magnesium (M; 40 mg kg−1), low-dose apigenin + magnesium (A + M (LD); 10 mg kg−1 magnesium + 1 mg kg−1 apigenin), medium-dose apigenin + magnesium (A + M (MD); 20 mg kg−1 magnesium + 2 mg kg−1 apigenin), high-dose apigenin + magnesium (A + M (HD); 40 mg kg−1 magnesium + 4 mg kg−1 apigenin) and diphenhydramine hydrochloride (DIPH; 20 mg kg−1) treatment groups. The chemicals were gifts from Telford International Company Ltd, Hong Kong.For the pentobarbital-induced sleep test, mice were fasted 24 hours prior to sleep testing, unless specified otherwise. 40 mg kg−1 sodium pentobarbital was administered intraperitoneally 30 minutes after treatments, as previously described.24 After induction, mice were transferred to individual cages for observation. Sleep duration (the period of time from falling asleep to exhibiting stereotactic reflection) was measured as an indication of sleep status. Mice that did not exhibit any signs of stereotactic reflection loss 15 minutes after induction with pentobarbital were excluded from the test.25 On the day after the sleep test, mice were sacrificed and blood was collected by cardiac puncture. Mouse organs, whole brains, and the hypothalamus were removed and weighed. Plasma and hypothalamus samples were stored at −80 °C for further analysis.
2.3 Bliss independence model analysis
Bliss independence was calculated as previously reported by Petraitis et al. and Duarte et al., using the equation EIND = EA + EM − EA × EM, where EIND is the expected effect of A + M (HD), and EA and EM are respectively the promoting effects of apigenin and magnesium treatment on mouse sleep duration.27,28 The interaction between apigenin and magnesium is represented by the difference between the expected effect and experimentally observed effect of A + M (HD) (EOBS, ΔE = EOBS − EIND). If ΔE and its 95% confidence interval (CI) were <1, synergy was concluded.2.4 ELISA
TNFα, IL-1β, and IL-6 levels in plasma were measured by commercially available ELISA kits (BioLegend, Cambridge, UK). Procedures were performed according to the manufacturer's instructions.Briefly, diluted standards and plasma samples were added to 96-well plates coated with TNFα, IL-1β, or IL-6 antibodies and incubated overnight. After incubation, detection antibodies and Avidin-HRP solution were added, and the colour intensity was measured using a Multiskan SkyHigh Microplate Spectrophotometer (ThermoFisher Scientific, USA). The concentrations of TNFα, IL-1β, and IL-6 were calculated relative to their corresponding standard curves.
2.5 Quantitative polymerase chain reaction (qPCR) assay
DNA was extracted from approximately 10 mg of hypothalamic tissues using the TGuide Smart Blood/Cell/Tissue RNA kit (TIANGEN BIOTECH, Beijing, China), according to manufacturer's instructions. Extracted DNA was quantified using a Nanodrop One spectrophotometer (Thermo Scientific) and subsequently stored at −80 °C until use in qPCR analysis.qPCR reaction mixtures were prepared as follows: 10 μl of 2× PowerUp SYBR Green Master Mix (Applied Biosystems, USA), 400 nM of each forward and reverse primer (Table 1), and 2 μl of sample DNA at a concentration of 2.5 ng μl−1, resulting in a total reaction volume of 20 μl. Amplification was performed on a QuantStudio 7 system (Applied Biosystems) under the following conditions: an initial incubation at 50 °C for 2 minutes, followed by an activation step at 95 °C for 2 minutes, and then 40 cycles of denaturation at 95 °C for 1 second and annealing/extension at 60 °C for 30 seconds. Relative expression of each target was calculated using the 2−ΔΔCT method.
| Target | Forward primer 5′–3′ | Reverse primer 5′–3′ |
|---|---|---|
| TNFα | GGTGAGGAGCACGTAGTCGG | TCCCAGGTTCTCTTCAAGGGA |
| GAPDH | ATGGGGAAGGTGAAGGTCG | GGGGTCATTGATGGCAACAATA |
2.6 Western blotting
Western blot analysis was conducted as previously described.29 In brief, hypothalamus samples were lysed in RIPA buffer and cellular debris removed by centrifugation. Protein concentration of supernatants was quantified using the DC protein assay (Bio-Rad, Hercules, CA, USA) and samples were then solubilized in Laemmli sample buffer. Equal protein amounts were resolved by SDS-PAGE and transferred onto PVDF membranes (Advansta Inc., CA, USA). Blots were then blocked in 5% non-fat skim milk and probed with primary antibodies followed by their corresponding secondary antibodies. Protein bands were visualized using WesternBright ECL HRP substrate (Advansta Inc.) and a ChemiDoc Imaging System (Bio-Rad). Protein expression levels were quantified using Image Lab software (Bio-Rad). Antibodies used were as follows: β-actin (Santa Cruz Biotechnology, Santa Cruz, CA, USA), Iba1 (Fujifilm, Osaka, Japan), NF-κB, and phospho-NF-κB (Cell Signaling Technology, Danvers, MA, USA).2.7 Statistical analysis
Data were analyzed using GraphPad Prism 10.3 (GraphPad Software, USA). Results are presented as mean ± SD and were analyzed by either one-way ANOVA with Dunnett's multiple comparison test or two-way ANOVA with Tukey's comparison test. P values < 0.05 were considered statistically significant.3. Results
3.1 Apigenin and magnesium treatment exhibited hypnotic effects in mice
Previous studies have shown that individually, apigenin and magnesium exhibit sleep improving properties,30,31 however, their combinatory effects have yet to be explored. Therefore, we adopted a pentobarbital-induced sleep mouse model to evaluate their potential combined hypnotic effect. Mice were fed orally with the corresponding treatments 60 minutes before pentobarbital-induced sleep testing. The treatment scheme is illustrated in Fig. 1A.As shown in Fig. 1B, mice receiving treatment with either apigenin or magnesium alone showed no improvement in sleep duration when compared to control, however, a dose-dependent hypnotic effect was observed when they were used in combination. A + M (MD) and A + M (HD) significantly increased the sleep duration of pentobarbital-induced mice by 16% (p < 0.05) and 44% (p < 0.01) respectively. Notably, the increase in sleep duration after A + M (HD) treatment was comparable to DIPH, an antihistamine drug commonly used as a sleep aid32 and here employed as a positive control, which exhibited a 48% (p < 0.001) increase in sleep duration after administration. Thus, we identified a potential dose-dependent hypnotic effect for the combination of A + M. The high-dose (HD) group showed the greatest sleep improving efficacy and was therefore selected for downstream experiments.
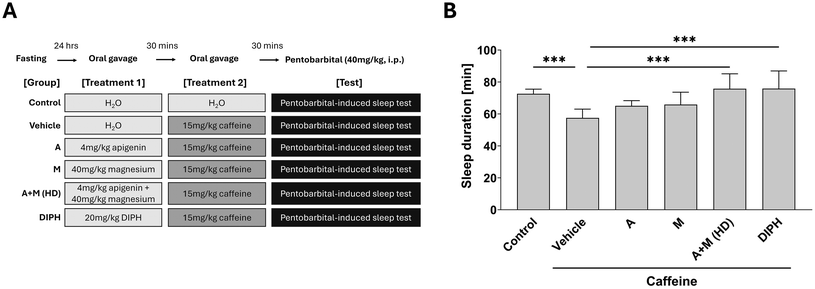
3.2 Apigenin and magnesium treatment ameliorated caffeine-induced sleep disturbance in mice
Nocturnal use of caffeine may lead to sleep disruption and sleeplessness, as caffeine antagonizes adenosine receptors in the brain and interferes with the sleep–wake cycle.33,34 To further investigate the sleep improving properties of A + M, the caffeine-induced sleep disturbance mice model was adopted. Mice were fed with the corresponding treatments 30 minutes before caffeine induction, and pentobarbital-induced sleep tests were performed 30 minutes after caffeine induction. The treatment scheme is shown in Fig. 2A.In comparison with uninduced control, sleep duration was significantly reduced by 21% (p < 0.001) in caffeine-induced mice, indicating successful induction of sleep disturbance (Fig. 2B). Apigenin or magnesium alone did not significantly improve sleep duration, but A + M (HD) treatment lead to a significant increase in sleep duration. A + M (HD)-treated caffeine-induced mice showed a 32% increase in sleep duration when compared to vehicle control (p < 0.001). Again, the improvement in sleep duration of A + M (HD)-treated mice was comparable to DIPH, which also exhibited a significant increase of 32% (p < 0.001) when compared to vehicle control. In addition, we identified a synergistic effect of A + M (HD) in caffeine-induced mice (Table 2). These results further supported the potential hypnotic effect of the apigenin and magnesium combination.
3.3 Apigenin and magnesium treatment exhibited hypnotic and anti-inflammatory effects in mice
To further explore its potential as a dietary sleep supplement, we tested if A + M induced effects of sleep improvement after continuous treatment. As shown in Fig. 3A, mice received 21 consecutive days of treatment, and pentobarbital-induced sleep tests were performed to evaluate the hypnotic effects of continuous treatment.Daily treatment of apigenin, magnesium, or A + M (HD) did not alter the body weight, or food and water consumption of mice when compared to control (Fig. 3B–D, SI Fig. 1). Next, we examined the sleep duration of mice. In order to mimic actual conditions, we did not fast the mice in this experiment. As shown in Fig. 3E, mice exhibited comparable sleep durations before treatment in the pentobarbital-induced sleep test, and control mice did not show alterations in sleep duration after the treatment period. No sleep improving effect was observed in mice treated with apigenin or magnesium, but significant sleep improving effects were observed in the A + M (HD) group. After 21 days of treatment, a significant increase in sleep duration of 22% (p < 0.05) was observed in mice treated with A + M (HD) when compared with control mice.
Mice were terminated after the experiment, and vital organs including kidneys, liver, lungs, heart, spleen, thymus, and brain were removed, weighed, and organ coefficients calculated (SI Fig. 2). No changes in organ gross morphology (data not shown) or organ coefficients were observed in treatment groups when compared to control.
As both apigenin and magnesium have been reported to exhibit anti-inflammatory effects,35,36 and previous studies have linked sleeplessness to increased inflammatory status,37,38 we hypothesized that the sleep improving effects of A + M (HD) may be mediated via anti-inflammatory mechanisms. Therefore, we investigated the effect of A + M (HD) treatment on systemic levels of the pro-inflammatory cytokines TNFα, IL-1β, and IL-6. As shown in Fig. 4A and C, 21-day treatment of A + M (HD) significantly suppressed TNFα and IL-6 levels by 27% (p < 0.05) and 34% (p < 0.001) respectively when compared with control. In addition, apigenin and magnesium treatment alone also suppressed IL-6 levels by 29% (p < 0.001) and 28% (p < 0.001) respectively when compared with control. However, we did not observe any alterations in IL-1β in mice after the various treatments. Thus, our results indicated that the sleep improving effect of A + M (HD) was potentially mediated via anti-inflammatory activity.
3.4 Apigenin and magnesium treatment improved sleep duration in PCPA-induced insomnia mice
To further understand the potential of A + M on sleep improvement, we adopted a commonly used insomnia mouse model to investigate the effects of A + M. PCPA is a selective inhibitor of tryptophan hydroxylase (TPH), the key and rate-limiting enzyme in 5-HT biosynthesis.39 Administration of PCPA leads to suppression of 5-HT production, subsequent disturbance of sleep rhythms, and insomnia-like symptoms.26,40 To induce insomnia, mice were injected with PCPA for two consecutive days before daily treatment with apigenin, magnesium, A + M (HD), or water for 7 consecutive days (Fig. 5A). As shown in Fig. 5B–D and SI Fig. 3, no alterations in mouse body weight, or food and water consumption were identified when comparing treatment groups with vehicle during the experimental period.At the end of the treatment period, pentobarbital-induced sleep testing was performed to evaluate the potential sleep-improving effect of the treatments. In comparison with control, sleep duration was reduced by 33% (p < 0.001) in vehicle-treated mice, indicating successful induction of insomnia after PCPA induction (Fig. 5E). Although no improvements in sleep were observed in PCPA-induced mice receiving apigenin or magnesium treatments alone, A + M (HD) treatment significantly increased sleep duration in mice by 37% (p < 0.05). A synergistic effect was observed in PCPA-induced mice treated with A + M (HD) (Table 3).
At termination, no significant alterations were observed among organ coefficients (kidney, liver, lung, heart, spleen, thymus, and brain) of mice in treatment groups (SI Fig. 4).
3.5 Apigenin and magnesium treatment exhibited anti-inflammatory effects in PCPA-induced insomnia mice
As we observed anti-inflammatory effects of apigenin, magnesium, and A + M (HD) in the previous mouse models, we next examined whether the treatments also exhibited anti-inflammatory effects in PCPA-induced mice. As shown in Fig. 6A–C, plasma levels of TNFα, IL-1β, and IL-6 were significantly increased in PCPA-induced mice when compared with control mice, confirming an inflammatory state. No anti-inflammatory effects were observed in PCPA-induced mice treated with magnesium alone, whereas apigenin treatment in PCPA-induced mice led to a significant suppression of plasma IL-6 levels by 21% (p < 0.05). Treatment with A + M (HD) significantly suppressed the plasma levels of TNFα, IL-1β, and IL-6 by 37% (p < 0.01), 31% (p < 0.05), and 23% (p < 0.01) respectively when compared with vehicle.The hypothalamus plays a critical role in regulating various aspects of the sleep–wake cycle, and sleep deprivation can lead to neuroinflammation.41,42 Since A + M (HD) treatment reduced plasma levels of pro-inflammatory cytokines in PCPA-induced mice, we asked if the treatment also exhibited anti-neuroinflammatory effects. Based on the importance of the hypothalamus in sleep cycle regulation, we focused on this brain region in subsequent experiments. Previous studies have suggested that hypothalamus size may be associated with sleep disturbance, as reductions in hypothalamic volume were observed in neurodegenerative subjects with sleep dysfunction.43 Therefore, we examined the hypothalamic weights of mice. As shown in Fig. 6D, PCPA-induced mice exhibited a significant reduction of 44% (p < 0.001) in hypothalamus coefficient when compared with control. A + M (HD) treatment rescued this hypothalamic deficiency by 63% (p < 0.01) when compared with vehicle-treated mice. No significant effects were identified in mice treated with apigenin or magnesium alone. Next, we examined the expression of TNFα, IL-1β, and IL-6 in the hypothalamus. A significant increase in hypothalamic TNFα expression was observed in PCPA-induced mice when compared with control mice (Fig. 6E). Notably, mice treated with apigenin, magnesium, or A + M (HD) showed significant suppression of TNFα. We did not observe alteration of IL-1β or IL-6 expression in the hypothalamuses of PCPA-induced mice (data not shown).
Given that microglial activation can lead to increased inflammatory cytokine expression in the hypothalamus, and that PCPA-induced insomnia leads to microglial activation,44–46 we examined if the treatments could alter the activation of microglia, resulting in the reduced expression of inflammatory cytokines. As shown in Fig. 7A, a significant increase in the expression of Iba1, a commonly used microglial activation marker, was observed in the hypothalamus of PCPA-induced mice when compared with control, indicating microglia activation. However, apigenin, magnesium, and A + M treatments significantly suppressed the expression levels of Iba1. It has been shown that activation of NFκB can lead to increased expression of pro-inflammatory cytokines, including TNFα, in microglial cells.47 To further explore the anti-inflammatory mechanism of the treatments, we evaluated the activation of NFκB in the hypothalamic tissues of the mice. We observed activation of NFκB in PCPA-induced mice when compared with control, while A + M (HD) could significantly suppress this activation (Fig. 7B). In summary, we showed that the hypnotic effect of A + M (HD) was potentially mediated via suppression of NFκB and microglial activation.
4. Discussion
CBT-I is widely used in conjunction with sleep medications for the treatment of insomnia.6 However, long-term usage of hypnotic agents has been linked to the development of tolerance and reduction of therapeutic efficacy,48–50 and studies have demonstrated that prolonged administration of pharmacological treatments for insomnia is associated with a range of adverse effects, including impairments in psychomotor performance, cognitive function, and memory. DIPH is a common antihistamine used to treat allergic reactions, motion sickness, and insomnia. It acts as an inverse agonist by binding to histamine 1 (H1) receptors in the central nervous system (CNS), thereby inhibiting the effects of histamine, an excitatory neurotransmitter that primarily facilitates alertness and wakefulness. Hence, blocking H1 receptors leads to increased drowsiness and sleep promotion.51 Mice with histamine-deficiency exhibit obesity, increased visceral adiposity, hyperleptinemia, and hyperinsulinemia when fed a high-fat diet.52,53 Notably, we did not observe similar effects in apigenin, magnesium, or A + M (HD)-treated mice, highlighting their advantage as sleep aids.The naturally occurring flavonoid apigenin has been previously demonstrated to have sleep-promoting effects.54 The anti-inflammatory and TNFα-suppressing effects of apigenin have also been demonstrated in vitro and in vivo in different animal models.55–59 Apigenin has been shown to suppress activated NFκB in LPS-stimulated macrophages, as well as in microglia cells.55,60 Mechanistically, apigenin has been shown to inhibit the phosphorylation and degradation of IκB, which functions to sequester NFκB in the cytoplasm and prevents it from entering the nucleus and regulating the expression of pro-inflammatory genes.60 On the other hand, magnesium is an essential ion required for proper bodily functioning as well as regulation of immunity.18 Magnesium deficiency has been linked to activation of the immune system and increased risk of various diseases including Alzheimer's disease, cardiovascular disease, stroke, and type 2 diabetes mellitus.61,62 In endothelial cells, low magnesium leads to redox imbalance, increased production of free radicals, and upregulation of the NFκB pathway.63 Similar effects were also observed in immune cells, where neutrophils and macrophages produced higher levels of free radicals when they were maintained in low magnesium conditions.62 Further, a previous study has demonstrated the anti-inflammatory effect of magnesium, where treatment of LPS-activated microglia cells with magnesium sulfate could suppress the activity of NFκB.64 In addition to the inflammatory response, magnesium is also involved in the regulation of circadian rhythms, and magnesium intake has been correlated with improved sleep quality and increased sleep duration,23,65 indicating its potential as a supplement for sleep. Therefore, the combination of apigenin and magnesium may simultaneously target IκB and NFκB of the NFκB signaling pathway, providing a more substantial suppressive effect on NFκB and downstream TNFα expression and offering a more comprehensive approach to improving sleep. Moreover, the effective dose used in our study, equivalent to 19.4 mg apigenin and 194.4 mg magnesium for a 60 kg human, is well below doses previously shown to be well-tolerated in clinical trials (23.4 mg and 225 mg for apigenin and magnesium respectively),66,67 underscoring the translational potential of this combination.
Based on the biopharmaceutics classification system, apigenin has been classified as a class II drug, indicating it possesses poor aqueous solubility but high intestinal permeability.70 Though apigenin has been shown to cross the blood–brain barrier and act on the CNS through the circulatory system,71 the oral bioavailability of apigenin remains low due to its low water solubility,72,73 hindering its widespread use as a functional food. In order to improve the aqueous solubility, dissolution, and in vivo bioavailability of apigenin, different nanoformulations have been proposed, including apigenin-phospholipid phytosome, apigenin-loaded polylactic-co-glycolic-acid meso-2,3-dimercaptosuccinic acid nanoparticles, and hydrogel encapsulated apigenin.74–76 However, these nanoformulations of apigenin are in the experimental stage and require further development for practical application. In contrast, different forms of magnesium compounds are currently available as dietary supplements, though comparisons of their bioavailability and efficacy are limited. A previous report by Uysal et al. showed that among five tested magnesium compounds (magnesium sulfate, magnesium oxide, magnesium acetyl taurate, magnesium citrate, and magnesium malate), magnesium acetyl taurate was the most rapidly absorbed into the brain and exhibited considerable effects on anxious behaviours.68 In another study, Kappeler et al. demonstrated that magnesium citrate exhibited higher bioavailability than magnesium oxide in humans.69 Although the selection of magnesium compounds varies across different studies, these findings may provide some insight into the selection of magnesium compound when composing the A + M formulation.
The hypothalamus plays a vital role in regulating sleep functions through its complex interactions with various neurotransmitter systems and neuropeptides. It is central to both homeostatic and circadian regulation of sleep, integrating signals that influence sleep–wake cycles.41,77 The hypothalamus is a key component of the hypothalamic–pituitary–adrenal (HPA) axis, which modulates stress responses that can affect sleep quality. Activation of the HPA axis typically results in lighter sleep and increased nocturnal awakenings, while insufficient sleep can heighten HPA activity, creating a feedback loop that exacerbates insomnia.78 Hypothalamic dysfunction, such as that induced by PCPA, disrupts the balance of sleep–wake regulation, destabilizing the HPA axis and amplifying stress–sleep feedback loops.
Chronic sleep deprivation or insomnia may contribute to increased pro-inflammatory cytokine activity through hyperactivation of the HPA axis and sympathetic nervous system pathways.79 TNFα, a major pro-inflammatory cytokine, has been implicated in sleep regulation. While previous animal studies have demonstrated increased TNFα levels in the brains of PCPA-induced insomnia rodents,46 most human studies are restricted to systemic TNFα levels. Various diseases with elevated levels of TNFα have been reported to be associated with sleeplessness, and treatment with TNFα inhibitors not only alleviated disease symptoms, but also restored sleep patterns.80 Although it is still unclear how systemic TNFα communicates with the brain in humans, it has been shown that systemic TNFα can induce brain expression of TNFα mRNA in animal studies.80 In our study, we found increased plasma and hypothalamic levels of TNFα in PCPA-induced mice, indicative of an inflammatory status. However, A + M (HD) treatment suppressed TNFα levels in both the plasma and hypothalamus, an effect that could not be achieved by treatment with apigenin or magnesium alone, and which may contribute to the enhanced sleep-promoting effect of the combination.
The significance of reducing pro-inflammatory cytokines extends beyond sleep regulation; TNFα, and IL-6 which we demonstrated to be downregulated by A + M treatment, are also established mediators of stress, anxiety, and depression. A dysregulated inflammatory response is a core feature linking these conditions and studies have consistently identified increased levels of TNFα and IL-6 in anxious and depressed subjects when compared to healthy controls.81,82 This creates a mechanistic bridge to the well-documented, bidirectional relationship between sleep disturbance and mood disorders, where poor sleep exacerbates inflammatory signaling and emotional dysregulation, while anxiety and depression drive sleep-disrupting hyperarousal.83 The capacity of A + M to suppress TNFα and IL-6 thus positions it as an intervention capable of targeting this shared neuroinflammatory pathway. This is supported by evidence showing that inhibiting these cytokines has mood-stabilizing effects: inhibition of TNFα in mouse models ameliorates depressive-like behaviours,84 and patients with inflammatory diseases treated with TNFα or IL-6 inhibitors show significant improvements in depressive and emotional measures.85,86 Thus, the intake of apigenin and magnesium could potentially alleviate anxiety and depression not only via restoration of sleep but also through direct anti-inflammatory activity.
Microglia are the resident phagocytes in the CNS, playing a critical role in maintaining homeostasis and defence against invasion. Microglial activation is a hallmark of neuroinflammatory responses and has been implicated in sleep regulation through modulation of cytokine release.46 Since chronic sleep deprivation has been shown to activate microglia,87 the observed reduction of Iba1 in the hypothalamus of PCPA-induced mice after A + M (HD) treatment indicates suppression of microglial activation, further supporting the anti-inflammatory effect of the combination. Collectively, these findings support a model in which the sleep-promoting effects of apigenin and magnesium are mediated, at least in part, by the suppression of neuroinflammation, as evidenced by the reduction in microglial activation within the hypothalamus.
Our study primarily utilized pentobarbital-induced sleep tests, a well-established model commonly used for research on sleep-promoting therapies. While the model measures sleep duration via the behavioural endpoint of loss of righting reflex, it cannot provide direct information on changes in the architecture of natural sleep such as transitions between wakefulness, rapid eye movement (REM), and non-REM (NREM) sleep stages. Furthermore, our study focussed on pro-inflammatory mediators in circulation and in the brain rather than sleep-associated neuropeptides. For example, changes in the levels of orexin affect the sleep–wake cycle through its promotion of wakefulness, while melatonin is a well-known regulator of the circadian rhythm.88,89 Future studies employing electroencephalography and electromyography assessments would enable the quantification of specific sleep parameters such as REM and NREM sleep latency, duration, bout continuity and fragmentation.90 Measurement of neuropeptides and inflammatory markers such as orexin, melatonin, and cortisol in the cerebrospinal fluid would provide further insight into the potential involvement of central nervous system signaling and central inflammation. Together, this multimodal-approach would provide a deeper understanding of the sleep-promoting mechanism of A + M.
5. Conclusion
In this study we showed that the A + M combinatory treatment led to improvements in sleep duration under normal sleep conditions and in insomnia models. Mechanistic analyses showed that A + M modulated systemic and hypothalamic TNFα levels and suppressed activation of microglia in the hypothalamus through suppression of NFκB. These findings highlight the potential of A + M as a natural intervention to restore sleep architecture and mitigate neurophysiological deficits associated with insomnia, positioning it as a promising alternative to pharmacological agents.Author contributions
Wing-Yan Wong: writing – original draft, writing – review & editing, investigation, conceptualization. Brandon Dow Chan: writing – original draft, writing – review & editing, investigation. Hengshen Zhou: investigation. Tsz Chun Leung: investigation. William Chi-Shing Tai: conceptualization, writing – review & editing.Conflicts of interest
The authors declare no conflicts of interests in the work reported in this paper.Data availability
All data generated or analyzed during this study are included in the published article.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo04538j.
Acknowledgements
This research was supported in part by funding from The Hong Kong Polytechnic University to WCST (ZDFT and BE8Z). The authors would like to thank the University Research Facility in Life Sciences (ULS) of the Hong Kong Polytechnic University for technical and equipment support.References
- C. M. Morin and R. Benca, Chronic insomnia, Lancet, 2012, 379, 1129–1141 CrossRef PubMed.
- C. M. Morin and D. C. Jarrin, Epidemiology of insomnia: prevalence, course, risk factors, and public health burden, Sleep Med. Clin., 2022, 17, 173–191 CrossRef PubMed.
- D. Duan, L. J. Kim, J. C. Jun and V. Y. Polotsky, Connecting insufficient sleep and insomnia with metabolic dysfunction, Ann. N. Y. Acad. Sci., 2023, 1519, 94–117 CrossRef PubMed.
- N. J. Pearson, L. L. Johnson and R. L. Nahin, Insomnia, trouble sleeping, and complementary and alternative medicine: analysis of the 2002 national health interview survey data, Arch. Intern. Med., 2006, 166, 1775–1782 CrossRef PubMed.
- C. M. Morin, D. C. Jarrin, H. Ivers, C. Mérette, M. LeBlanc and J. Savard, Incidence, persistence, and remission rates of insomnia over 5 years, JAMA Netw. Open, 2020, 3, e2018782 CrossRef PubMed.
- M. T. Liu, Current and emerging therapies for insomnia, Am. J. Manage. Care, 2020, 26, S85–S90 Search PubMed.
- M. R. Zielinski and J. M. Krueger, Sleep and innate immunity, Front. Biosci., Scholar Ed., 2011, 3, 632–642 Search PubMed.
- Z. Aghelan, S. Karima, S. H. Abtahi, H. Khazaie, Y. Salimi and R. Khodarahmi, The imbalance of circulating pro-inflammatory cytokines interleukin-12 and interferon gamma and anti-inflammatory cytokine interleukin-1ra in chronic insomnia disorder, Sleep Med., 2023, 112, 194–196 Search PubMed.
- M. Ditmer, A. Gabryelska, S. Turkiewicz, P. Białasiewicz, E. Małecka-Wojciesko and M. Sochal, Sleep Problems in Chronic Inflammatory Diseases: Prevalence, Treatment, and New Perspectives: A Narrative Review, J. Clin. Med., 2021, 11, 67 CrossRef PubMed.
- X. Cao, J. Zhao, M. Zhang, S. Lou and Q. Cao, P1276 Persistent Insomnia Ignites the Flames of Crohn’s Disease by Elevating Systemic Inflammation, J. Crohn’s Colitis, 2025, 19, i2304 Search PubMed.
- M. Sochal, M. Ditmer, A. Binienda, A. Gabryelska, P. Białasiewicz, R. Talar-Wojnarowska, J. Fichna and E. Małecka-Wojciesko, Relation between Selected Sleep Parameters, Depression, Anti-Tumor Necrosis Factor Therapy, and the Brain-Derived Neurotrophic Factor Pathway in Inflammatory Bowel Disease, Metabolites, 2023, 13, 450 CrossRef CAS PubMed.
- M. Rudwaleit, K. Gooch, B. Michel, M. Herold, Å. Thörner, R. Wong, M. Kron, N. Chen and H. Kupper, Adalimumab improves sleep and sleep quality in patients with active ankylosing spondylitis, J. Rheumatol., 2011, 38, 79–86 Search PubMed.
- A. Deodhar, J. Braun, R. D. Inman, M. Mack, S. Parasuraman, J. Buchanan, B. Hsu, T. Gathany and D. Van Der Heijde, Golimumab reduces sleep disturbance in patients with active ankylosing spondylitis: results from a randomized, placebo–controlled trial, Arthritis Care Res., 2010, 62, 1266–1271 Search PubMed.
- L. Zhang, R. R. Lu, R. H. Xu, H. H. Wang, W. S. Feng and X. K. Zheng, Naringenin and apigenin ameliorates corticosterone-induced depressive behaviors, Heliyon, 2023, 9, e15618 Search PubMed.
- L.-T. Yi, J.-M. Li, Y.-C. Li, Y. Pan, Q. Xu and L.-D. Kong, Antidepressant-like behavioral and neurochemical effects of the citrus-associated chemical apigenin, Life Sci., 2008, 82, 741–751 Search PubMed.
- R.-F. Zhong, C.-Y. Shen, K.-X. Hao and J.-G. Jiang, Flavonoids from Polygoni Multiflori Caulis alleviates p-chlorophenylalanine-induced sleep disorders in mice, Ind. Crops Prod., 2024, 218, 119002 Search PubMed.
- R. Fu, L. Wang, Y. Meng, W. Xue, J. Liang, Z. Peng, J. Meng and M. Zhang, Apigenin remodels the gut microbiota to ameliorate ulcerative colitis, Front. Nutr., 2022, 9, 1062961 Search PubMed.
- I. Slutsky, N. Abumaria, L. J. Wu, C. Huang, L. Zhang, B. Li, X. Zhao, A. Govindarajan, M. G. Zhao, M. Zhuo, S. Tonegawa and G. Liu, Enhancement of learning and memory by elevating brain magnesium, Neuron, 2010, 65, 165–177 Search PubMed.
- K. Peuhkuri, N. Sihvola and R. Korpela, Diet promotes sleep duration and quality, Nutr. Res., 2012, 32, 309–319 CrossRef CAS PubMed.
- N. Górska, J. Słupski, P. Szałach Ł, A. Włodarczyk, J. Szarmach, K. Jakuszkowiak-Wojten, M. Gałuszko-Węgielnik, A. Wilkowska, M. S. Wiglusz and W. J. Cubała, Magnesium and ketamine in the treatment of depression, Psychiatr. Danubina, 2019, 31, 549–553 Search PubMed.
- J. J. Poza, M. Pujol, J. J. Ortega-Albás and O. Romero, Melatonin in sleep disorders, Neurología (Engl. Ed.), 2022, 37, 575–585 Search PubMed.
- S. Park, I. Kang, R. A. E. Edden, E. Namgung, J. Kim and J. Kim, Shorter sleep duration is associated with lower GABA levels in the anterior cingulate cortex, Sleep Med., 2020, 71, 1–7 Search PubMed.
- Y. Zhang, C. Chen, L. Lu, K. L. Knutson, M. R. Carnethon, A. D. Fly, J. Luo, D. M. Haas, J. M. Shikany and K. Kahe, Association of magnesium intake with sleep duration and sleep quality: findings from the CARDIA study, Sleep, 2021, 45, zsab276 Search PubMed.
- A. Lin, C. T. Shih, C. L. Huang, C. C. Wu, C. T. Lin and Y. C. Tsai, Hypnotic Effects of Lactobacillus fermentum PS150(TM) on Pentobarbital-Induced Sleep in Mice, Nutrients, 2019, 11, 2409 CrossRef CAS PubMed.
- H. Yang, M. Yoon, M. Y. Um, J. Lee, J. Jung, C. Lee, Y. T. Kim, S. Kwon, B. Kim and S. Cho, Sleep-Promoting Effects and Possible Mechanisms of Action Associated with a Standardized Rice Bran Supplement, Nutrients, 2017, 9, 512 Search PubMed.
- Q. Tang, J. Xiong, J. Wang, Z. Cao, S. Liao, Y. Xiao, W. Tian and J. Guo, Queen bee larva consumption improves sleep disorder and regulates gut microbiota in mice with PCPA-induced insomnia, Food Biosci., 2021, 43, 101256 CrossRef CAS.
- V. Petraitis, R. Petraitiene, W. W. Hope, J. Meletiadis, D. Mickiene, J. E. Hughes, M. P. Cotton, T. Stergiopoulou, M. Kasai, A. Francesconi, R. L. Schaufele, T. Sein, N. A. Avila, J. Bacher and T. J. Walsh, Combination therapy in treatment of experimental pulmonary aspergillosis: in vitro and in vivo correlations of the concentration- and dose- dependent interactions between anidulafungin and voriconazole by Bliss independence drug interaction analysis, Antimicrob. Agents Chemother., 2009, 53, 2382–2391 CrossRef CAS PubMed.
- D. Duarte and N. Vale, Evaluation of synergism in drug combinations and reference models for future orientations in oncology, Curr. Res. Pharmacol. Drug Discovery, 2022, 3, 100110 CrossRef PubMed.
- W. Y. Wong, M. M. Lee, B. D. Chan, R. K. Kam, G. Zhang, A. P. Lu and W. C. Tai, Proteomic profiling of dextran sulfate sodium induced acute ulcerative colitis mice serum exosomes and their immunomodulatory impact on macrophages, Proteomics, 2016, 16, 1131–1145 Search PubMed.
- N. K. Chow, M. Fretz, M. Hamburger and V. Butterweck, Telemetry as a tool to measure sedative effects of a valerian root extract and its single constituents in mice, Planta Med., 2011, 77, 795–803 CrossRef CAS PubMed.
- J. Mah and T. Pitre, Oral magnesium supplementation for insomnia in older adults: a Systematic Review & Meta-Analysis, BMC Complementary Med. Ther., 2021, 21, 125 Search PubMed.
- Y. Q. Wang, Y. Takata, R. Li, Z. Zhang, M. Q. Zhang, Y. Urade, W. M. Qu and Z. L. Huang, Doxepin and diphenhydramine increased non-rapid eye movement sleep through blockade of histamine H1 receptors, Pharmacol., Biochem. Behav., 2015, 129, 56–64 CrossRef CAS PubMed.
- N. S. Chaudhary, M. A. Grandner, N. J. Jackson and S. Chakravorty, Caffeine consumption, insomnia, and sleep duration: Results from a nationally representative sample, Nutrition, 2016, 32, 1193–1199 CrossRef CAS PubMed.
- N. Olini, S. Kurth and R. Huber, The effects of caffeine on sleep and maturational markers in the rat, PLoS One, 2013, 8, e72539 Search PubMed.
- X. Feng, D. Weng, F. Zhou, Y. D. Owen, H. Qin, J. Zhao, W. Yu, Y. Huang, J. Chen, H. Fu, N. Yang, D. Chen, J. Li, R. Tan and P. Shen, Activation of PPARγ by a Natural Flavonoid Modulator, Apigenin Ameliorates Obesity-Related Inflammation Via Regulation of Macrophage Polarization, eBioMedicine, 2016, 9, 61–76 CrossRef PubMed.
- M. Mazidi, P. Rezaie and M. Banach, Effect of magnesium supplements on serum C-reactive protein: a systematic review and meta-analysis, Arch. Med. Sci., 2018, 14, 707–716 CrossRef PubMed.
- A. N. Vgontzas, E. Zoumakis, E. O. Bixler, H. M. Lin, H. Follett, A. Kales and G. P. Chrousos, Adverse effects of modest sleep restriction on sleepiness, performance, and inflammatory cytokines, J. Clin. Endocrinol. Metab., 2004, 89, 2119–2126 CrossRef CAS PubMed.
- M. Haack, E. Sanchez and J. M. Mullington, Elevated inflammatory markers in response to prolonged sleep restriction are associated with increased pain experience in healthy volunteers, Sleep, 2007, 30, 1145–1152 CrossRef PubMed.
- A. A. Borbély, H. U. Neuhaus and I. Tobler, Effect of p-chlorophenylalanine and tryptophan on sleep, EEG and motor activity in the rat, Behav. Brain Res., 1981, 2, 1–22 CrossRef PubMed.
- Y. Si, L. Wang, J. Lan, H. Li, T. Guo, X. Chen, C. Dong, Z. Ouyang and S. Q. Chen, Lilium davidii extract alleviates p-chlorophenylalanine-induced insomnia in rats through modification of the hypothalamic-related neurotransmitters, melatonin and homeostasis of the hypothalamic-pituitary-adrenal axis, Pharm. Biol., 2020, 58, 915–924 Search PubMed.
- D. Ono and A. Yamanaka, Hypothalamic regulation of the sleep/wake cycle, Neurosci. Res., 2017, 118, 74–81 CrossRef CAS PubMed.
- D. Cao, Y. Zhao, Y. Wang, D. Wei, M. Yan, S. Su, H. Pan and Q. Wang, Effects of sleep deprivation on anxiety-depressive-like behavior and neuroinflammation, Brain Res., 2024, 1836, 148916 Search PubMed.
- P. T. Best, M. Bocchetta, J. D. Rohrer, J. B. Rowe, B. Borroni, D. Galimberti, P. Tiraboschi, M. Masellis, M. C. Trataglia, E. C. Finger, J. C. van Swieten, H. Seelaar, L. C. Jiskoot, S. Sorbi, C. R. Butler, C. Graff, A. Gerhard, T. Langheinrich, R. J. Laforce, R. Sánchez-Valle, A. d. Mendonça, F. Moreno, M. Synofzik, R. Vandenberghe, I. L. Ber, J. Levin, A. Danek, M. Otto, F. Pasquier, I. Santana, M. L. Schroeter, A. M. Remes, M. L. Waldö, Y. A. L. Pijnenburg, J. E. Nielsen, T. van Langenhove, M. Chakravarty and S. Ducharme, Hypothalamic volumes predict sleep dysfunction in genetic frontotemporal dementia, Alzheimer’s Dementia, 2023, 19, e061536 CrossRef.
- B. García-Bueno, J. Serrats and P. E. Sawchenko, Cerebrovascular cyclooxygenase-1 expression, regulation, and role in hypothalamic-pituitary-adrenal axis activation by inflammatory stimuli, J. Neurosci., 2009, 29, 12970–12981 Search PubMed.
- M. Valdearcos, J. D. Douglass, M. M. Robblee, M. D. Dorfman, D. R. Stifler, M. L. Bennett, I. Gerritse, R. Fasnacht, B. A. Barres, J. P. Thaler and S. K. Koliwad, Microglial Inflammatory Signaling Orchestrates the Hypothalamic Immune Response to Dietary Excess and Mediates Obesity Susceptibility, Cell Metab., 2017, 26, 185–197 CrossRef CAS PubMed.
- M. Shi, J. Yang, Y. Liu, H. Zhao, M. Li, D. Yang and Q. Xie, Huanglian Wendan Decoction Improves Insomnia in Rats by Regulating BDNF/TrkB Signaling Pathway Through Gut Microbiota-Mediated SCFAs and Affecting Microglia Polarization, Mol. Neurobiol., 2025, 62, 1047–1066 CrossRef CAS PubMed.
- M. Zusso, V. Lunardi, D. Franceschini, A. Pagetta, R. Lo, S. Stifani, A. C. Frigo, P. Giusti and S. Moro, Ciprofloxacin and levofloxacin attenuate microglia inflammatory response via TLR4/NF-kB pathway, J. Neuroinflammation, 2019, 16, 148 CrossRef PubMed.
- M. Soyka, I. Wild, B. Caulet, C. Leontiou, F. Lugoboni and G. Hajak, Long-term use of benzodiazepines in chronic insomnia: a European perspective, Front. Psychiatry, 2023, 14, 1212028 CrossRef PubMed.
- D. F. Kripke, Chronic hypnotic use: deadly risks, doubtful benefit, Sleep Med. Rev., 2000, 4, 5–20 CrossRef PubMed.
- F. Guo, L. Yi, W. Zhang, Z. J. Bian and Y. B. Zhang, Association Between Z Drugs Use and Risk of Cognitive Impairment in Middle-Aged and Older Patients With Chronic Insomnia, Front. Hum. Neurosci., 2021, 15, 775144 Search PubMed.
- S. Nuutinen and P. Panula, Histamine in neurotransmission and brain diseases, Histamine Inflammation, 2010, 95–107 Search PubMed.
- A. s. K. Fülöp, A. Földes, E. Buzás, K. Hegyi, I. H. Miklós, L. s. Romics, M. Kleiber, A. s. Nagy, A. s. Falus and K. J. Kovács, Hyperleptinemia, visceral adiposity, and decreased glucose tolerance in mice with a targeted disruption of the histidine decarboxylase gene, Endocrinology, 2003, 144, 4306–4314 Search PubMed.
- E. A. Jørgensen, T. W. Vogelsang, U. Knigge, T. Watanabe, J. Warberg and A. Kjaer, Increased susceptibility to diet-induced obesity in histamine-deficient mice, Neuroendocrinology, 2006, 83, 289–294 CrossRef PubMed.
- D. J. Kramer and A. A. Johnson, Apigenin: a natural molecule at the intersection of sleep and aging, Front. Nutr., 2024, 11, 1359176 CrossRef PubMed.
- X. Zhang, G. Wang, E. C. Gurley and H. Zhou, Flavonoid apigenin inhibits lipopolysaccharide-induced inflammatory response through multiple mechanisms in macrophages, PLoS One, 2014, 9, e107072 Search PubMed.
- M. Palacz-Wrobel, P. Borkowska, M. Paul-Samojedny, M. Kowalczyk, A. Fila-Danilow, R. Suchanek-Raif and J. Kowalski, Effect of apigenin, kaempferol and resveratrol on the gene expression and protein secretion of tumor necrosis factor alpha (TNF-α) and interleukin-10 (IL-10) in RAW-264.7 macrophages, Biomed. Pharmacother., 2017, 93, 1205–1212 Search PubMed.
- S. Malik, K. Suchal, S. I. Khan, J. Bhatia, K. Kishore, A. K. Dinda and D. S. Arya, Apigenin ameliorates streptozotocin-induced diabetic nephropathy in rats via MAPK-NF-κB-TNF-α and TGF-β1-MAPK-fibronectin pathways, Am. J. Physiol.: Renal Physiol., 2017, 313, F414–f422 CrossRef CAS PubMed.
- J. Wang, Y.-T. Liu, L. Xiao, L. Zhu, Q. Wang and T. Yan, Anti-Inflammatory Effects of Apigenin in Lipopolysaccharide-Induced Inflammatory in Acute Lung Injury by Suppressing COX-2 and NF-kB Pathway, Inflammation, 2014, 37, 2085–2090 Search PubMed.
- A. Charalabopoulos, S. Davakis, M. Lambropoulou, A. Papalois, C. Simopoulos and A. Tsaroucha, Apigenin Exerts Anti-inflammatory Effects in an Experimental Model of Acute Pancreatitis by Down-regulating TNF-alpha, In Vivo, 2019, 33, 1133–1141 Search PubMed.
- D. N. Che, B. O. Cho, J.-s. Kim, J. Y. Shin, H. J. Kang and S. I. Jang, Effect of Luteolin and Apigenin on the Production of Il-31 and Il-33 in Lipopolysaccharides-Activated Microglia Cells and Their Mechanism of Action, Nutrients, 2020, 12, 811 CrossRef CAS PubMed.
- S. L. Volpe, Magnesium in Disease Prevention and Overall Health, Adv. Nutr., 2013, 4, 378S–383S Search PubMed.
- J. A. Maier, S. Castiglioni, L. Locatelli, M. Zocchi and A. Mazur, Magnesium and inflammation: Advances and perspectives, Semin. Cell Dev. Biol., 2021, 115, 37–44 CrossRef CAS PubMed.
- S. Ferrè, E. Baldoli, M. Leidi and J. A. M. Maier, Magnesium deficiency promotes a pro-atherogenic phenotype in cultured human endothelial cells via activation of NFkB, Biochim. Biophys. Acta, Mol. Basis Dis., 2010, 1802, 952–958 Search PubMed.
- F. Gao, B. Ding, L. Zhou, X. Gao, H. Guo and H. Xu, Magnesium sulfate provides neuroprotection in lipopolysaccharide-activated primary microglia by inhibiting NF-κB pathway, J. Surg. Res., 2013, 184, 944–950 Search PubMed.
- K. A. Feeney, L. L. Hansen, M. Putker, C. Olivares-Yañez, J. Day, L. J. Eades, L. F. Larrondo, N. P. Hoyle, J. S. O'Neill and G. van Ooijen, Daily magnesium fluxes regulate cellular timekeeping and energy balance, Nature, 2016, 532, 375–379 CrossRef CAS PubMed.
- S. M. Zick, B. D. Wright, A. Sen and J. T. Arnedt, Preliminary examination of the efficacy and safety of a standardized chamomile extract for chronic primary insomnia: A randomized placebo-controlled pilot study, BMC Complementary Altern. Med., 2011, 11, 78 CrossRef PubMed.
- M. Rondanelli, A. Opizzi, F. Monteferrario, N. Antoniello, R. Manni and C. Klersy, The effect of melatonin, magnesium, and zinc on primary insomnia in long-term care facility residents in Italy: a double-blind, placebo-controlled clinical trial, J. Am. Geriatr. Soc., 2011, 59, 82–90 Search PubMed.
- N. Uysal, S. Kizildag, Z. Yuce, G. Guvendi, S. Kandis, B. Koc, A. Karakilic, U. M. Camsari and M. Ates, Timeline (Bioavailability) of Magnesium Compounds in Hours: Which Magnesium Compound Works Best?, Biol. Trace Elem. Res., 2019, 187, 128–136 CrossRef CAS PubMed.
- D. Kappeler, I. Heimbeck, C. Herpich, N. Naue, J. Höfler, W. Timmer and B. Michalke, Higher bioavailability of magnesium citrate as compared to magnesium oxide shown by evaluation of urinary excretion and serum levels after single-dose administration in a randomized cross-over study, BMC Nutr., 2017, 3, 7 Search PubMed.
- J. Zhang, D. Liu, Y. Huang, Y. Gao and S. Qian, Biopharmaceutics classification and intestinal absorption study of apigenin, Int. J. Pharm., 2012, 436, 311–317 CrossRef CAS PubMed.
- B. Salehi, A. Venditti, M. Sharifi-Rad, D. Kręgiel, J. Sharifi-Rad, A. Durazzo, M. Lucarini, A. Santini, E. B. Souto, E. Novellino, H. Antolak, E. Azzini, W. N. Setzer and N. Martins, The Therapeutic Potential of Apigenin, Int. J. Mol. Sci., 2019, 20, 1305 CrossRef CAS PubMed.
- M. Xiao, Y. Shao, W. Yan and Z. Zhang, Measurement and correlation of solubilities of apigenin and apigenin 7-O-rhamnosylglucoside in seven solvents at different temperatures, J. Chem. Thermodyn., 2011, 43, 240–243 Search PubMed.
- J. Zhang, Y. Huang, D. Liu, Y. Gao and S. Qian, Preparation of apigenin nanocrystals using supercritical antisolvent process for dissolution and bioavailability enhancement, Eur. J. Pharm. Sci., 2013, 48, 740–747 Search PubMed.
- D. R. Telange, A. T. Patil, A. M. Pethe, H. Fegade, S. Anand and V. S. Dave, Formulation and characterization of an apigenin-phospholipid phytosome (APLC) for improved solubility, in vivo bioavailability, and antioxidant potential, Eur. J. Pharm. Sci., 2017, 108, 36–49 CrossRef CAS PubMed.
- R. Sen, S. Ganguly, S. Ganguly, M. C. Debnath, S. Chakraborty, B. Mukherjee and D. Chattopadhyay, Apigenin-Loaded PLGA-DMSA Nanoparticles: A Novel Strategy to Treat Melanoma Lung Metastasis, Mol. Pharm., 2021, 18, 1920–1938 CrossRef CAS PubMed.
- X. Zhao and Z. Wang, A pH-sensitive microemulsion-filled gellan gum hydrogel encapsulated apigenin: Characterization and in vitro release kinetics, Colloids Surf., B, 2019, 178, 245–252 Search PubMed.
- R. Van Drunen and K. Eckel-Mahan, Circadian Rhythms of the Hypothalamus: From Function to Physiology, Clocks Sleep, 2021, 3, 189–226 CrossRef PubMed.
- V. Lo Martire, D. Caruso, L. Palagini, G. Zoccoli and S. Bastianini, Stress & sleep: A relationship lasting a lifetime, Neurosci. Biobehav. Rev., 2020, 117, 65–77 CrossRef PubMed.
- L. Palagini, P. A. Geoffroy, M. Miniati, G. Perugi, G. Biggio, D. Marazziti and D. Riemann, Insomnia, sleep loss, and circadian sleep disturbances in mood disorders: a pathway toward neurodegeneration and neuroprogression? A theoretical review, CNS Spectrums, 2022, 27, 298–308 CrossRef PubMed.
- M. D. Rockstrom, L. Chen, P. Taishi, J. T. Nguyen, C. M. Gibbons, S. C. Veasey and J. M. Krueger, Tumor necrosis factor alpha in sleep regulation, Sleep Med. Rev., 2018, 40, 69–78 CrossRef PubMed.
- S. Hassamal, Chronic stress, neuroinflammation, and depression: an overview of pathophysiological mechanisms and emerging anti-inflammatories, Front. Psychiatry, 2023, 14, 1130989 CrossRef PubMed.
- Y. Yin, T. Ju, D. Zeng, F. Duan, Y. Zhu, J. Liu, Y. Li and W. Lu, “Inflamed” depression: A review of the interactions between depression and inflammation and current anti-inflammatory strategies for depression, Pharmacol. Res., 2024, 207, 107322 CrossRef CAS PubMed.
- D. Freeman, B. Sheaves, F. Waite, A. G. Harvey and P. J. Harrison, Sleep disturbance and psychiatric disorders, Lancet Psychiatry, 2020, 7, 628–637 CrossRef PubMed.
- Y. N. Liu, Y. L. Peng, L. Liu, T. Y. Wu, Y. Zhang, Y. J. Lian, Y. Y. Yang, K. W. Kelley, C. L. Jiang and Y. X. Wang, TNFα mediates stress-induced depression by upregulating indoleamine 2,3-dioxygenase in a mouse model of unpredictable chronic mild stress, Eur. Cytokine Network, 2015, 26, 15–25 CAS.
- H. Siuchnińska, A. Minarowska and E. Wasilewska, Beyond Joints: Neuropsychiatric Benefits of TNF-α and IL-6 Inhibitors in Rheumatoid Arthritis-Narrative Review, Int. J. Mol. Sci., 2025, 26, 8361 CrossRef PubMed.
- G. M. Wittenberg, A. Stylianou, Y. Zhang, Y. Sun, A. Gupta, P. S. Jagannatha, D. Wang, B. Hsu, M. E. Curran, S. Khan, G. Chen, E. T. Bullmore and W. C. Drevets, Effects of immunomodulatory drugs on depressive symptoms: A mega-analysis of randomized, placebo-controlled clinical trials in inflammatory disorders, Mol. Psychiatry, 2020, 25, 1275–1285 CrossRef CAS PubMed.
- M. Bellesi, L. de Vivo, M. Chini, F. Gilli, G. Tononi and C. Cirelli, Sleep Loss Promotes Astrocytic Phagocytosis and Microglial Activation in Mouse Cerebral Cortex, J. Neurosci., 2017, 37, 5263–5273 CrossRef CAS PubMed.
- Y. C. Shen, X. Sun, L. Li, H. Y. Zhang, Z. L. Huang and Y. Q. Wang, Roles of Neuropeptides in Sleep-Wake Regulation, Int. J. Mol. Sci., 2022, 23, 4599 CrossRef CAS PubMed.
- S. Koop and H. Oster, Eat, sleep, repeat - endocrine regulation of behavioural circadian rhythms, FEBS J., 2022, 289, 6543–6558 CrossRef CAS PubMed.
- G. M. Mang and P. Franken, Sleep and EEG Phenotyping in Mice, Curr. Protoc. Mouse Biol., 2012, 2, 55–74 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |