Open Access Article
Open Access ArticleImpact of the orange juice fruit matrix on postprandial glycemia: a crossover randomized trial in healthy young men with post hoc analysis of interindividual response variability
María Teresa
García-Conesa
 *a,
Rocío
García-Villalba
*a,
Rocío
García-Villalba
 a,
María Dolores
Frutos-Lisón
a,
Carlos Javier
García
a,
Javier
Marhuenda
b,
Pilar
Zafrilla
a,
María Dolores
Frutos-Lisón
a,
Carlos Javier
García
a,
Javier
Marhuenda
b,
Pilar
Zafrilla
 b,
Juan Antonio
Tudela
c,
Marta Ruiz
Arráez
d,
Gary
Williamson
b,
Juan Antonio
Tudela
c,
Marta Ruiz
Arráez
d,
Gary
Williamson
 e,
Carrie
Ruxton
f and
Francisco A.
Tomás-Barberán
e,
Carrie
Ruxton
f and
Francisco A.
Tomás-Barberán
 a
a
aResearch Group on Quality, Safety and Bioactivity of Plant-Derived Foods, Centro de Edafología y Biología Aplicada del Segura-Consejo Superior de Investigaciones Científicas (CEBAS-CSIC), 30100 Murcia, Spain. E-mail: mtconesa@cebas.csic.es; Tel: +34 968 396200, Ext: 445476
bFaculty of Pharmacy and Nutrition, Campus de los Jerónimos, Guadalupe, Universidad Católica de Murcia (UCAM), 30107 Murcia, Spain
cResearch Group on Microbiology and Quality of Fruit and Vegetables, Centro de Edafología y Biología Aplicada del Segura-Consejo Superior de Investigaciones Científicas (CEBAS-CSIC), 30100 Murcia, Spain
dFruit & Tech., Carretera Madrid-Cartagena 390, 30100 Murcia, Spain
eSchool of Biological Sciences, Queen's University Belfast, University Road, Belfast, BT7 1NN, UK
fNutrition Communications, East Road, Cupar KY15 4HQ, UK
First published on 11th February 2026
Abstract
The impact of fruit juices on postprandial glucose response (PPGR) remains controversial due to their free sugar content. The aim of this study was to investigate the effect of the fruit matrix in 100% orange juice (OJ) on PPGR. In this randomized crossover trial, we compared the intake of 300 mL of 100% OJ to sugar-matched drinks with reduced (50% OJ) or no (0% OJ) fruit matrix and a glucose control (25 g sugar) in healthy young males. We characterised the juices for nutrients and (poly)phenols and measured glucose and insulin over 2 hours. We also analysed interindividual variability and performed a post-hoc cluster analysis of the participants based on the different responses to the drinks. Metabolomics was used to further explore plasma differences between clusters. Differences in incremental area under the curve (iAUC) with the 100% OJ and 50% OJ against the 0% OJ did not reach significance, but the 100% OJ significantly lowered the glucose peak (Cmax) compared to 0% OJ and reduced glucose levels at 15 minutes (C15 min) compared to 50% OJ and 0% OJ. High interindividual variability was expressed as ‘high responders’ with larger differences in Cmax between 100% OJ and 0% OJ than ‘low responders’ as well as differences in the 60 min plasma metabolomes, particularly in OJ-derived metabolites like dihydroferulic acid glucuronide. The OJ fruit matrix attenuates postprandial glucose peaks and rate of glucose rise in healthy young males. The occurrence of responder groups with differentiated plasma metabolomes underscores the importance of considering both the food matrix and individual physiological differences when assessing the glycemic impact of fruit juices.
Introduction
Type 2 diabetes mellitus (T2DM) is characterized by impaired glucose metabolism.1 Maintaining normal glycemia (70 mg dL−1 (3.9 mmol L−1)–100 mg dL−1 (5.6 mmol L−1)),2 and avoiding postprandial hyperglycemia (which begins when plasma glucose rises above 140 mg dL−1 (7.77 mmol L−1))3 are important for the prevention of T2DM. Lifestyle and dietary habits, in particular, the quantity and type of carbohydrate intake, are key factors contributing to the postprandial glucose response (PPGR).4,5 The PPGR refers to the changes in blood glucose concentration following the consumption of a meal. It is characterized by measuring glucose levels at different time points over a two-hour period to generate a response curve. The overall glycemic impact is commonly quantified by the incremental area under the curve (iAUC) (area above the individual's pre-meal (fasting) baseline glucose level). Other metrics such as the magnitude and time of the glucose spike (Cmax and Tmax)6 as well as the rate of the rise of blood glucose (related to the concentration at 15 min: C15 min)7 are also important for understanding of the response dynamics.The classification of specific dietary sugars as ‘free’ was proposed by the World Health Organization (WHO) in 20038 and was adopted by the European Food Safety Authority (EFSA) and several countries but not the United States (US) where added sugars are used. The free sugars classification includes added sugars plus those naturally present in honey, syrups, fruit and vegetable juices, and juice concentrates. Since sugar molecules are chemically and biologically indistinguishable by source,9 any physiologic differentiation between free and other sugars must arise mainly from the effects of the food matrix, defined as the complex structural and chemical environment of a food, its components (e.g., nutrients and non-nutrients), and how they interact.10
The International Diabetes Federation advises against sugar-sweetened beverages (SSB) and fruit juices because these can cause spikes in blood glucose levels.11 All free sugars, whether from fruit juices or added sugars are assumed by the WHO to have potential negative health effects leading to inconsistent dietary guidelines, with some countries discouraging their consumption while others equate a maximum of one daily serving of fruit juice to a portion of fruit.12 Nevertheless, the impact of fruit juice consumption on glucose metabolism remains unclear and, whereas some observational studies suggest that each additional serving of fruit juice is associated with a higher risk of T2DM,13 several meta-analyses conclude that fruit juice has no impact on glucose and insulin regulation nor the risk of T2DM.14–16 Regarding orange juice (OJ), the effects on glucose and insulin are also inconsistent.17–19 One problem with the assessment of fruit juices is the indiscriminate combination of data on 100% fruit juices and juice-type drinks which could contain added sugars and a lower proportion of fruit matrix (i.e. micronutrients (vitamin C, minerals), pectin, bioactives (flavonoids)).20 Only a few studies have compared the intake of 100% OJ with carbohydrate-matched sweetened orange drinks but the results are insufficiently conclusive.21–23
The main aim of the present study was to investigate the effect of the fruit matrix on the postprandial glycemic and insulin response by comparing a well-characterized OJ (100% OJ) with two sugar-matched drinks with 50% or 0% of the fruit matrix in healthy young male adults. We hypothesized that the 100% OJ would have a lower response than the drink containing only sugars. If free sugars are equally detrimental, as judged by WHO, we would expect to see no statistically significant difference in the glycemic responses following the three test beverages. We also analyzed interindividual variability and explored distinct responder subgroups using a post-hoc clustering analysis.
Materials and methods
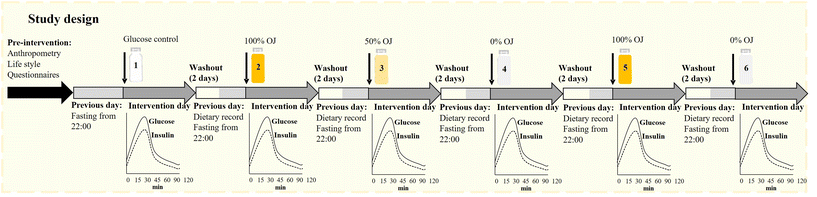
Four beverages were manufactured, bottled (300 mL) and supplied by AMC Natural Drinks, (Murcia, Spain): (i) a 100% OJ, (ii) a 50% OJ formulated to contain the same qualitative and quantitative composition of sugars (sucrose, glucose and fructose) as in the 100% OJ but half the content of fruit matrix, (iii) a drink formulated to contain only the sugars (sucrose, glucose and fructose) as in the 100% OJ (0% OJ), and (iv) a control solution containing only the equivalent total quantity of glucose (25 g). The 100% OJ was obtained by directly squeezing Navel and Valencia orange varieties. Washed fruit was processed using extractor cups, which separate the juice and pulp from the peel oil and rinds. The fresh juice was then transferred to a holding tank for bottling and subsequent High-Pressure Processing (HPP) to preserve its ‘freshly squeezed’ characteristics. For the 50% OJ and the 0% OJ, the ingredients were blended directly in the holding tank and packaged under identical conditions to the 100% juice. No additives, preservatives, or additional nutrients were introduced into any of the beverages. Despite a most commonly recommended quantity of 50 g, to avoid the intake of an excessive amount of juice, the quantity of total sugars was reduced to 25 g. All drinks were kept under refrigeration (4 °C) during the study period.Characterization of the drinks
The filtered samples were injected into an Agilent 1100 HPLC system equipped with a photodiode array detector G1315D (Agilent Technologies, Waldbronn, Germany) and coupled in series to a High Trap Capacity (HCT) ion trap mass spectrometer (MS) (Bruker Daltonics, Bremen, Germany) through an electrospray ionization (ESI) interface (HPLC-DAD-ESI-MS/MS (IT)). The chromatographic separation was achieved using a reversed-phase C18 Poroshell column (100 × 3 mm, 2.7 μm particle size, Agilent Technologies). The method used was a binary gradient, A (water/formic acid, 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v)) and B (acetonitrile), settled in the following gradients: 0 min, 5% B; 7 min, 18% B; 17 min, 28% B; 22 min, 50% B; 27 min, 90% B; the initial conditions were re-established at 29 min and kept under isocratic conditions up to 35 min. The flow rate was 0.5 mL min−1, the injection volume was 5 μL, and the column temperature was settled at 25 °C. The UV-Visible spectra were acquired in the range of 200 to 600 nm and the chromatograms were registered at 320, 330 and 360 nm. In the mass spectrometer, nitrogen was used as the drying, nebulizing and collision gas. The ESI parameters were: nebulizer pressure 65 psi, dry gas flow 11 L min−1 and dry gas temperature 350 °C. The capillary voltage was set at 4 kV and the spectra were acquired in negative and positive ionization mode in the range of m/z 100–1200. Automatic MS/MS mode was applied with fragmentation amplitude 1 V and number of parents, 3.
1 (v/v)) and B (acetonitrile), settled in the following gradients: 0 min, 5% B; 7 min, 18% B; 17 min, 28% B; 22 min, 50% B; 27 min, 90% B; the initial conditions were re-established at 29 min and kept under isocratic conditions up to 35 min. The flow rate was 0.5 mL min−1, the injection volume was 5 μL, and the column temperature was settled at 25 °C. The UV-Visible spectra were acquired in the range of 200 to 600 nm and the chromatograms were registered at 320, 330 and 360 nm. In the mass spectrometer, nitrogen was used as the drying, nebulizing and collision gas. The ESI parameters were: nebulizer pressure 65 psi, dry gas flow 11 L min−1 and dry gas temperature 350 °C. The capillary voltage was set at 4 kV and the spectra were acquired in negative and positive ionization mode in the range of m/z 100–1200. Automatic MS/MS mode was applied with fragmentation amplitude 1 V and number of parents, 3.
Polyphenols were identified by their UV spectra, retention time, molecular weight and MS/MS fragmentation pattern and were quantified in UV using calibration curves of the authentic standards: p-coumaric acid (ref. 89498,) and sinensetin (ref. 89278) from PhytoLab phyproof (Germany); isosinensetin (ref. 36009) from Cayman Chemicals (Michigan, USA); hesperidin (ref. 1126S), apigenin (ref. 1102S), eriodictyol (ref. 1111S-10 mg), naringin (ref. 1129S-10 mg), and isorhamnetin (ref. 1120S) from Extrasynthèse (Geney, France); quercetin (17799-1MG-F) was from Sigma-Aldrich (Madrid, Spain), and nobiletin (ref. 1467848) was from USP-Reference (Rockville, USA). Hydroxycinnamic acids were quantified with the calibration curve of p-coumaric acid, glycosylated derivatives of flavonoids with the calibration curve of their corresponding aglycones except for hesperetin and naringenin derivatives that were quantified with the calibration curve of hesperidin and naringin, and polymethoxyflavones were quantified with their corresponding standards except for heptamethoxyflavone, hexamethylquercetagetin, demethoxytangeretin and artemitin that were quantified with the nobiletin calibration curve. The limits of detection (LOD) and limits of quantification (LOQ) for the different compounds are as follows: Apigenin: LOD 0.42 µM and LOQ 1.41 µM; Quercetin: LOD 0.45 µM and LOQ 1.50 µM; Isorhamnetin: LOD 0.50 µM and LOQ 1.66 µM; Naringin: LOD 1.50 µM and LOQ 5.00 µM; Eriodictyol: LOD 1.50 µM and LOQ 5.00 µM; Hesperidin: LOD 1.58 µM and LOQ 5.26 µM; p-coumaric acid: LOD 0.18 µM and LOQ 0.61 µM; Isosinensetin: LOD 0.25 µM and LOQ 0.84 µM; Sinensetin: LOD 0.19 µM and LOQ 0.63 µM and Nobiletin: LOD 0.20 µM and LOQ 0.65 µM. Analyses of (poly)phenols were carried out in triplicate.
Clinical trial
During the first visit, the participants completed a series of questionnaires to assess their dietary habits and lifestyle, and anthropometric measurements were obtained by the UCAM study personnel prior to the initial blood extraction. For each session (test drinks), participants also completed a paper 24 h dietary record documenting all meals taken during the previous day (food items, cooking procedure, drinks, and estimated quantities). A total of 5 food diaries was collected. Separation between food diaries was at least 2 days, and Sundays were included when a test drink was being consumed on a Monday.
On the day of the intervention, the participants arrived at the study unit after a 10–12 h fast – excepting only a small amount of water taken up to 1 h before arrival. After compliance with the overnight fasting requirements was verified, the 0 min (baseline levels) blood sample was obtained. The volunteers were then given the assigned test drink and instructed to drink the full volume (300 mL) within 5 minutes. Following beverage consumption, blood samples were drawn at 15, 30, 45, 60, 90, and 120 minutes, via venous canulation using a single-use butterfly needle and standard venipuncture procedures. All blood samples were collected from the antecubital vein by trained nurses. During the whole intervention, the participants remained all in the same room under the same conditions, and under the direct care of the nurses who supervised each participant to minimize potential harms associated to the blood sampling (pain, bruising) or signs of discomfort following the intake of the test drink (nausea, dizziness, headache). The nurses provided immediate assistance and ensured the participants were comfortable during the study.
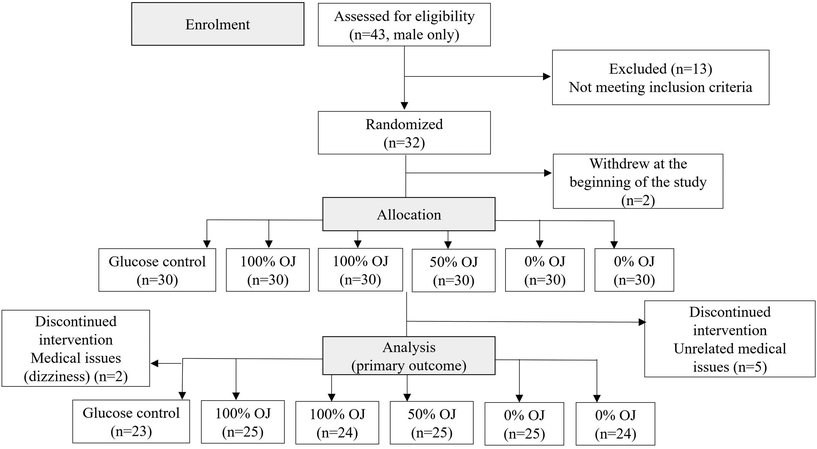
Volunteers from the area local to the UCAM were recruited during September and October 2024. Recruitment was conducted via online classified advertisements, social media, and word-of-mouth. Women were excluded to control for hormonal fluctuations during the menstrual cycle that affect glucose levels.25 Eligible criteria were: healthy adult males aged 18 to 45 years, non-smokers, habitual breakfast consumers, and be willing to consume the study beverages. Individuals were excluded if they were following a therapeutic diet or had food allergies, sensitivities, or an aversion to the beverages investigated in the study, had a BMI ≥30.0 or significant weight fluctuations within the past six months, or had been diagnosed with diabetes, pre-diabetes, gastrointestinal disease, liver disease, kidney disease, or any metabolic disorder. Exclusion criteria were based on self-reporting. A flow diagram (Fig. 2) indicates the number of participants recruited and assessed for eligibility, randomly allocated to the test sessions, and finally analyzed. All participants were informed about the protocol, signed a consent form before entering into the study and received a compensation payment after their participation was completed. The study protocol was approved by the UCAM Ethical Committee (ref: CE092408) and registered at Clinicaltrials.gov (NCT06638190).
Glucose and insulin analyses
Blood samples were collected in standard serum separator tubes (SST) without anticoagulant. After clotting at room temperature, the samples were centrifuged at 1200–1500g for 10 minutes at 4 °C to obtain the serum. Serum aliquots were immediately stored at −80 °C until analysis of glucose and insulin concentrations.The Glucose Oxidase (GOD) Activity Assay Kit (E-BC-K520-M) (Elabscience, Texas, USA) was used for the quantitative determination of blood glucose.26 This method utilizes a reagent that consists of GOD, peroxidase (POD), and a chromogenic system (comprising 4-aminophenazone and phenol) prepared in accordance with the manufacturer's instructions. A volume of 10 µL of the serum samples (diluted if necessary) was mixed with 1 mL of the GOD-POD reagent, incubated at 37 °C for 10 minutes, and absorbance measured at 500 nm with a Synergy HT multi detect microplate reader BioTek Instruments, Inc. (Winooski, VT, USA). The glucose concentration was determined by comparison with a calibration curve prepared using serial dilutions of a glucose standard solution. This method has a detection range of 1.0–400 mg dL−1. All samples fell within this range. No samples exceeded these limits or required imputation.
To measure insulin, we applied a highly sensitive Human Insulin ELISA Kit (E-EL-H2665) (Elabscience, Texas, USA).27 Briefly, all samples, reagents, calibrators and controls were prepared at room temperature. Next, a capture reagent composed of insulin-specific monoclonal antibodies labelled with magnetic particles was added to the wells, followed by the addition of 50–100 µL of sample, calibrators, or controls according to the manufacturer's instructions. The mixture was incubated at 30–37 °C for 40 min. Subsequently, the magnetic particles were washed to remove any unbound debris. After the wash step, a chemiluminescent reagent that reacted with the antibody–insulin complex was added and incubated for 40 min. The emitted light signal was measured with a Synergy HT multi detect microplate reader BioTek Instruments, Inc. (Winooski, VT, USA) and the insulin concentration in each sample calculated using a calibration curve generated from standards. This method has a detection range of 0.156–10 µU mL−1. Samples exceeding this range were diluted and re-analyzed according to the manufacturer's protocol.
All samples were measured in duplicate, and results with a coefficient of variation (CV%) > 10% between replicates were re-analyzed. Calibration curves were constructed for each assay batch using kit standards. Internal controls (low, medium, and high concentration) were included in each run to monitor assay performance.
The glucose and insulin results at the different time points (0–120 min) were used to calculate the iAUC using the trapezoidal method (areas where concentrations dropped below fasting were treated as zero).
Untargeted metabolomics analysis of plasma samples by UPLC-ESI-QTOF-MS
The plasma samples (200 µL) obtained from the volunteers at time 0 and 60 min after the intervention with 100% OJ and 0% OJ were extracted with 600 µL acetonitrile + 1% formic acid by vortexing for 2 min. The mixture was centrifuged at 4000g for 10 min, and the supernatant was reduced to dryness using a speed vacuum concentrator (Savant SPD121P, ThermoScientific, Alcobendas, Spain). The dried samples were re-suspended in 200 µL of methanol, chrysin (Sigma Aldrich, St Louis, MO, USA) 0.1 µM added as internal standard, and filtered through a 0.22 µm polyvinylidene fluoride (PVDF) filter before analysis.An untargeted metabolomics approach was performed using an ultra-high-performance liquid chromatography system (UHPLC Infinity 1290, Agilent) connected to a high-resolution quadrupole time-of-flight mass spectrometer (Agilent 6550 iFunnel Q-TOF) featuring an Agilent Jet Stream (AJS) electrospray ionization (ESI) source. The MS was operated in negative ionization mode with the following settings: gas temperature at 280 °C, drying gas flow rate of 11 L min−1, nebulizer pressure at 45 psi, sheath gas maintained at 400 °C with a flow of 12 L min−1, capillary voltage set to 3500 V, fragmentor voltage at 100 V, and octopole RF voltage at 750 V. Data were acquired over a m/z range of 100 to 1100 with a scan speed of 3 spectra per second, applying continuous mass calibration using reference ions at m/z 112.9855 and 1033.9881.
Chromatographic separation was achieved using a reversed-phase C18 column (Poroshell 120, 3 × 100 mm, 2.7 µm) held at a constant temperature of 20 °C. The mobile phases were composed of water containing 0.1% formic acid (Solvent A) and acetonitrile with 0.1% formic acid (Solvent B), delivered at a flow rate of 0.4 mL min−1. The gradient program was as follows: starting at 1% B, ramping to 28% B at 10 min, increasing to 50% B at 16 min and to 95% at 24 min, maintained at 95% until 26 min, followed by a drop to 1% B at 27 min, and re-equilibrated to 1% B from 27 to 32 min.
Spectral data were collected in both centroid and profile modes. The raw files were converted to .abf format and analyzed using MS-DIAL version 5.1.2 (prime.psc.riken.jp/compms) for feature detection and data matrix generation. Parameter settings for peak detection were adjusted to ensure broad metabolome coverage, with an MS1 tolerance of 0.01 Da. Peaks were detected using a minimum intensity threshold of 1000 and a mass slice width of 0.1 Da. A linear weighted moving average with a smoothing factor of three scans was applied for noise reduction.
Statistical analysis
We explored the sources of variability in the Cmax glucose response by conducting a variance analysis using a more complex LMM that includes an additional random intercept for each combination of participant and treatment (Cmax ∼ treatment + (1|participant) + (1|participant: treatment)). This model allows to estimate the intra- and interindividual variability as well as the variance attributable to subject-by-treatment interaction, which represents the degree of consistent individual differences in treatment response. This model was applied only to duplicate treatments, 0% OJ and 100% OJ. Results are presented as variance (SD).
All analyses were performed in R 4.5.033 using dplyr34 for data processing and ggplot235 for visualization.
Results
Nutrient and phytochemical composition of the test drinks
The pH and nutrient composition of the test drinks are shown in Table 1. The 100% OJ and 50% OJ were slightly more acidic than the 0% OJ and the glucose solution. The four drinks contained a similar quantity of total sugars (∼25 g per bottle) which remained constant during the study period. The 50% OJ and the 0% OJ contained the same qualitative and quantitative composition in sugars (glucose, fructose, sucrose) as the 100% OJ. The analyses confirmed that the 50% OJ contained approximately half the quantity of the other main nutrients (complex carbohydrates, protein, minerals, fiber, fat and vitamin C) present in the 100% OJ. Concerning the minerals, the 50% OJ also contained approximately half the amount of the main macro-minerals (K, P, Ca, Mg, S, Si) per bottle than the 100% OJ whereas the other two drinks (0% OJ and glucose control) had only trace quantities of these minerals. Na was present at similar levels in the 100% OJ and in the 50% OJ. Micro minerals (Mn, Fe, B, Sr, Al, Rb, Ni, Zn, Cu, Mo, Ti) were also detected in the 100% OJ (0.5–0.05 mg per bottle), and the 50% OJ (0.25–0.02 mg per bottle) and they were present only at trace quantities (<0.01 mg per bottle) in the 0% OJ and the glucose control solution.| 100% OJ | 50% OJ | 0% OJ | Glucose control | |
|---|---|---|---|---|
| The results are presented as the mean ± SD (n = 2). Abbreviations: CHOs: carbohydrates; OJ, orange juice.a Complex CHOs were estimated as the difference between total CHOs and total sugars. | ||||
| pH | 3.5 | 3.4 | 4.2 | 5.9 |
| Nutrients (g per bottle) | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD |
| Total sugars | 25.0 ± 0.6 | 24.6 ± 1.2 | 25.0 ± 0.2 | 24.9 ± 0.4 |
| Glucose | 6.5 ± 0.4 | 6.4 ± 0.1 | 6.3 ± 0.1 | 24.5 ± 0.4 |
| Fructose | 6.5 ± 0.1 | 6.4 ± 0.4 | 6.8 ± 0.1 | — |
| Sucrose | 12.1 ± 0.1 | 11.9 ± 0.6 | 11.9 ± 0.0 | — |
| Total CHOs | 29.4 ± 0.2 | 26.7 ± 0.4 | — | — |
| Complex CHOsa | 4.4 ± 0.8 | 2.1 ± 0.7 | — | — |
| Fiber | 0.7 ± 0.2 | 0.6 ± 0.0 | — | — |
| Protein | 2.1 ± 0.0 | 1.1 ± 0.2 | — | — |
| Fat | 0.7 ± 0.2 | 0.9 ± 0.0 | — | — |
| Ashes | 1.2 ± 0.0 | 0.6 ± 0.0 | — | — |
| Vitamin C | 0.2 ± 0.2 | <0.04 | — | — |
| kcal | 134.2 ± 2.1 | 121.5 ± 2.1 | 97.3 ± 0.7 | 97.0 ± 1.7 |
| Minerals (mg per bottle) | ||||
| K | 599.9 ± 102.2 | 337.6 ± 26.6 | <0.003 | <0.003 |
| P | 61.3 ± 10.8 | 35.3 ± 1.0 | 0.1 ± 0.0 | 0.02 ± 0.00 |
| Ca | 42.2 ± 8.5 | 25.1 ± 1.8 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| Mg | 35.9 ± 8.2 | 21.2 ± 1.9 | 0.07 ± 0.00 | 0.05 ± 0.01 |
| S | 19.3 ± 3.2 | 11.0 ± 0.2 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| Na | 3.6 ± 0.7 | 3.5 ± 0.2 | 1.1 ± 0.0 | 0.6 ± 0.0 |
| Si | 3.7 ± 2.9 | 1.6 ± 0.6 | 0.05 ± 0.00 | 0.04 ± 0.00 |
The total quantity of (poly)phenols in the 100% OJ and 50% OJ were 218.3 ± 6.9 and 115.2 ± 0.6 mg per bottle, respectively (Table 2). The main families of (poly)phenols present in these two drinks were flavanones (84.3% of the total (poly)phenols), followed by hydroxycinnamic acids (8.2%) and flavones (6.2%). Flavonols (0.6%) and polymethoxyflavones (0.6%) were less abundant. The specific identified compounds were: glycosylated derivatives of hesperetin, naringenin, isosakuranetin and eriodyctiol (flavanones), hexoside derivatives of sinapic and ferulic acid (hydroxycinnamic acids), apigenin-6,8-C-dihexoside (flavone) and glycosylated derivatives of quercetin and isorhamnetin (flavonols). Regarding the polymethoxyflavones, nobiletin, sinensetin and heptamethoxyflavone were the most abundant ones.
| Compounds | Rt (min) | Absmax (nm) | [M − H]− | MS2 ions (m/z) | 50% OJ (mg per bottle) | 100% OJ (mg per bottle) |
|---|---|---|---|---|---|---|
| The results are presented as the mean ± SD (n = 3). Abbreviations: OJ, orange juice; Rt. retention time; Abs, absorbance; MS, mass spectrometry; n.d., not detected. | ||||||
| Flavanones | Mean ± SD | Mean ± SD | ||||
| Hesperetin-7-O-rutinoside (Hesperidin) | 14.1 | 330 | 609 | 301 | 67.01 ± 0.60 | 122.4 ± 6.9 |
| Naringenin-7-O-rutinoside (Narirutin) | 12.9 | 330 | 579 | 271 | 13.83 ± 0.09 | 25.97 ± 0.40 |
| Isosakuranetin-7-O-rutinoside (Dydimin) | 19.0 | 330 | 593 | 285 | 2.75 ± 0.15 | 5.20 ± 0.14 |
| Hesperitin-7-O-(rha, glu)-glu (I) | 7.7 | 330 | 771 | 609; 301 | 5.59 ± 0.29 | 11.20 ± 0.22 |
| Eriodyctiol-O-rutinoside (Eriocitrin) | 11.1 | 330 | 595 | 449; 287 | 5.28 ± 0.05 | 11.80 ± 0.16 |
| Hesperitin-7-O-(rha, glu)-glu (II) | 10.7 | 330 | 771 | 609; 463; 301 | 3.39 ± 0.29 | 6.37 ± 0.74 |
| Total | 97.84 ± 0.80 | 182.92 ± 6.70 | ||||
| Hydroxycinnamic acids | ||||||
| Sinapic acid hexoside (III) | 8.2 | 320 | 385 | 191 | 1.22 ± 0.04 | 2.56 ± 0.02 |
| Hydroxyferulic acid deoxyhexoside | 7.9 | 320 | 355 | 193 | 2.45 ± 0.12 | 4.49 ± 0.01 |
| Sinapic acid hexoside (I) | 6.3 | 320 | 385 | 191 | 2.56 ± 0.29 | 5.56 ± 0.03 |
| Ferulic acid hexoside | 7.3 | 320 | 355 | 337; 191 | 1.97 ± 0.14 | 3.88 ± 0.01 |
| Sinapic acid hexoside (II) | 6.5 | 320 | 385 | 191 | 0.96 ± 0.06 | 1.94 ± 0.02 |
| Total | 9.17 ± 0.48 | 18.43 ± 0.09 | ||||
| Flavones | ||||||
| Apigenin-6,8-C-dihexoside | 8.9 | 330 | 593 | 503; 473 | 6.87 ± 0.05 | 14.11 ± 0.06 |
| Total | 6.87 ± 0.05 | 14.11 ± 0.06 | ||||
| Flavonols | ||||||
| Isorhamnetin-3-O-rutinoside | 13.2 | 360 | 623 | 315 | 0.54 ± 0.06 | 1.14 ± 0.06 |
| Quercetin 3-O-rutinoside (Rutin) | 11.3 | 360 | 609 | 301 | 0.06 ± 0.00 | 0.12 ± 0.05 |
| Total | 0.61 ± 0.05 | 1.26 ± 0.07 | ||||
| Polymethoxyflavones | ||||||
| Nobiletin | 18.1 | 330 | 403 | 373, 388, 342 | 0.25 ± 0.01 | 0.60 ± 0.24 |
| Sinensetin | 16.8 | 330 | 373 | 312, 329, 343, 358, 297 | 0.20 ± 0.00 | 0.45 ± 0.14 |
| Heptamethoxyflavone | 18.9 | 330 | 433 | 403, 418, 385 | 0.12 ± 0.01 | 0.27 ± 0.11 |
| Demethoxytangeretin | 18.2 | 330 | 343 | 282, 299, 313, 328 | 0.09 ± 0.00 | 0.19 ± 0.01 |
| Hexamethylquercetagetin | 17.5 | 330 | 403 | 373, 387, 355, 339, 327, 314, 296 | 0.02 ± 0.00 | 0.05 ± 0.00 |
| Artemitin | 18.4 | 330 | 389 | 356, 374, 331, 313 | n.d. | 0.03 ± 0.00 |
| Isosinensetin | 15.6 | 330 | 373 | 343, 329, 358 | n.d. | 0.02 ± 0.00 |
| Total | 0.67 ± 0.01 | 1.60 ± 0.48 | ||||
| Total polyphenols | 115.15 ± 0.55 | 218.32 ± 6.97 | ||||
General characteristics of the study participants
| Mean ± SD | CV% | |
|---|---|---|
| The results are presented as the Mean ± SD (n = 25) and the CV% (coefficient of variation (percentage) = (SD/Mean) × 100) for continuous variables. Categories for some of the variables are also indicated with N (%): number and percentage of responses within a category for category. Abbreviations: BMI, body mass index; WC, waist circumference. | ||
| Age | 22.7 ± 2.7 | 11.9% |
| Anthropometric characteristics | ||
| BMI (kg m−2) | 24.1 ± 2.0 | 8.3% |
| BMI category | N (%) | |
| Healthy individuals (18.5–25.0 kg m−2) | 19 (76.0%) | |
| Individuals with overweight (25.0–30.0 kg m−2) | 6 (24.0%) | |
| Individuals with obesity (>30.0 kg m−2) | None | |
| WC (cm) | 81.6 ± 8.6 | 10.5% |
| WC category | N (%) | |
| No abdominal obesity (≤94 cm) | 23 (92.0%) | |
| Risk abdominal obesity (>94 cm) | 2 (8.0%) | |
| Increased risk abdominal obesity (>102 cm) | None | |
| % muscle mass | 49.9 ± 1.9 | 3.8% |
| % fat mass | 15.1 ± 1.3 | 8.6% |
| Baseline characteristics | ||
| Baseline glucose (mg dL−1) | 79.4 ± 4.5 | 5.7% |
| (mmol L−1) | (4.4 ± 0.3) | |
| Baseline insulin (µU mL−1) | 7.5 ± 2.6 | 34.0% |
Baseline glucose and insulin levels did not correlate with each other, nor did they correlate with any of the anthropometric and lifestyle variables.
| Glucose | Insulin | |||||
|---|---|---|---|---|---|---|
| Response metric | Treatment | n | Estimated Mean ± SEb (mg dL−1) | Estimated Mean ± SEb (mmol L−1) | n | Estimated Mean ± SEb (µU mL−1) |
| a Sample size (n) indicate the number of participants who completed the test. Data for glucose control and 50% OJ are from one replicate; data for 0% OJ and 100% OJ are from two replicates. b Data are the estimated means ± SE calculated using a LMM with treatment as a fixed effect and participant as a random effect. Within each metric, means in the same column that do not share a common superscript letter (a, b, c) are significantly different from each other (Tukey's post-hoc test, p < 0.05). Abbreviations: iAUC, incremental area under the curve; Cmax, maximum concentration; C15 min, concentration at 15 minutes; LMM, linear mixed model; OJ, orange juice. | ||||||
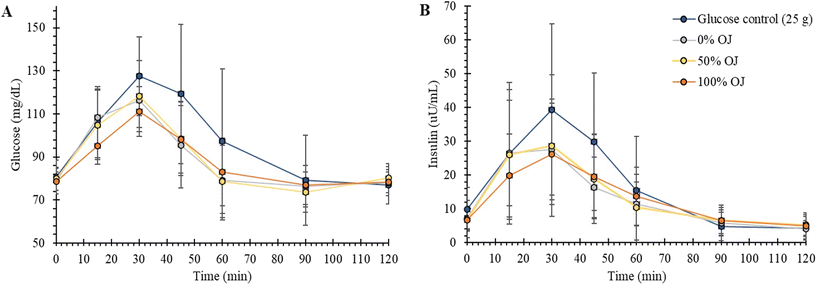
| iAUC (120 min) | Glucose | 23 | 2387 ± 213a | 133 ± 11.8a | 23 | 1208 ± 131a |
| 0% OJ | 49 | 1564 ± 178b | 87 ± 9.9b | 48 | 924 ± 101a | |
| 50% OJ | 25 | 1363 ± 207b | 77 ± 11.5b | 25 | 981 ± 126a | |
| 100% OJ | 49 | 1396 ± 178b | 78 ± 9.9b | 49 | 940 ± 100a | |
| C max | Glucose | 23 | 134.6 ± 3.1a | 7.5 ± 0.2a | 23 | 47.0 ± 3.7a |
| 0% OJ | 49 | 121.6 ± 2.5b | 6.8 ± 0.1b | 49 | 37.0 ± 3.0b | |
| 50% OJ | 25 | 119.3 ± 3.0bc | 6.6 ± 0.2bc | 25 | 34.9 ± 3.6b | |
| 100% OJ | 49 | 113.8 ± 2.5c | 6.3 ± 0.1c | 49 | 33.4 ± 3.0b | |
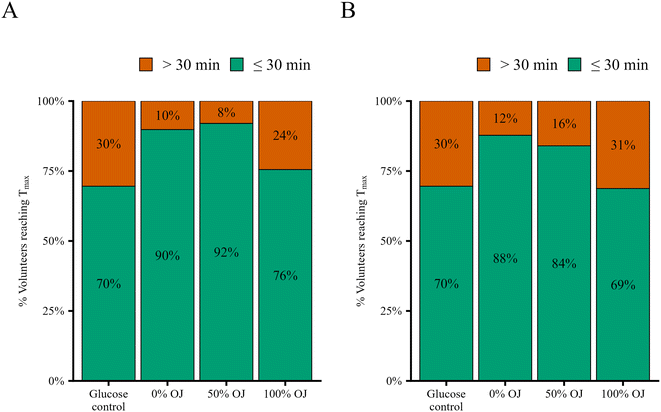
| C 15 min | Glucose | 23 | 107.0 ± 3.0a | 6.0 ± 0.2a | 22 | 27.2 ± 4.0a |
| 0% OJ | 49 | 108.7 ± 2.4a | 6.0 ± 0.1a | 49 | 27.0 ± 3.0a | |
| 50% OJ | 23 | 104.6 ± 3.0a | 5.8 ± 0.2a | 22 | 26.6 ± 4.0a | |
| 100% OJ | 49 | 95.9 ± 2.4b | 5.3 ± 0.1b | 47 | 20.3 ± 3.1a | |
We did not find any correlations between the glucose and insulin responses and any of the anthropometric and lifestyle variables included in this study.
Variability in the glucose Cmax responses to the drinks
The glycemic responses of each of the volunteers to the glucose control solution showed a high variability between the participants in the response to glucose (from participants where the Cmax did not go beyond 110 mg dL−1 to those where the Cmax increased up to nearly 180 mg dL−1, SI Fig. 1). The LMM analysis of the sources of variation in the glucose Cmax response confirmed a substantial inter-individual variance = 61.0 (SD 7.8 mg dL−1) as well as intra-individual variance = 84.9 (SD 9.2 mg dL−1). In addition, the model quantified the degree to which the effect of 100% OJ (relative to 0% OJ) varied from person to person yielding a variance = 16.2 (SD 4.0 mg dL−1). This value accounted for approximately 10% of the total variance. Overall, these results supported a complex structure of the variance with a high and stable intra- and inter-individual variability as well as a moderate heterogeneity in the glucose Cmax response to treatment (100% OJ against 0% OJ).Clustering analysis of the PPGR
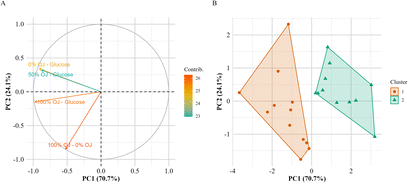
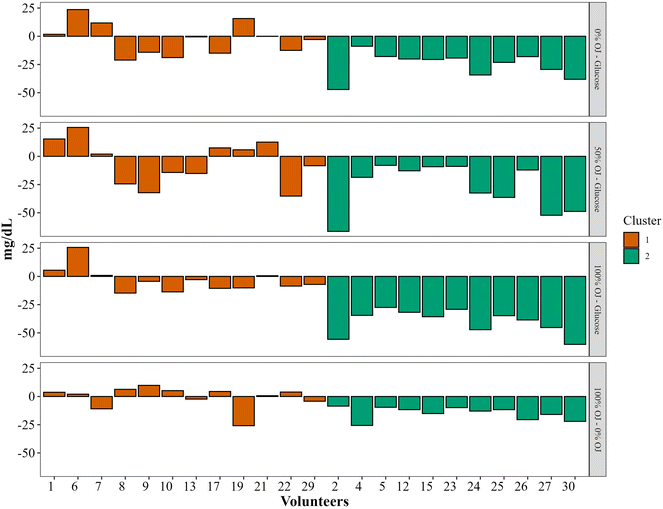
K-means cluster analysis of the participants based on the significant Cmax differences between the four drinks led to two well-separated clusters. PCA analysis was used to help identifying which differences were more important in distinguishing these two groups (Fig. 6A). The analysis revealed that 94.8% of the differences were explained by two principal components (Fig. 6B): the first principal component (PC1) explained 70.7% of the variance and represented the changes between the three juices and the glucose control, and the second principal component (PC2) explained 24.1% of the variance and represented principally the differences between the 100% OJ and the 0% OJ. The Cmax differences between the tested drinks for each volunteer in cluster 1 and cluster 2 are shown in Fig. 7. Cluster 1 grouped volunteers (n = 12) that showed variable differences between the responses to the four drinks (both peak increases and reductions) whereas the volunteers grouped in cluster 2 (n = 11) displayed consistent reductions in the glucose peaks when comparing the three juices against the control solution and when comparing the 100% OJ against the 0% OJ.The results of the comparison between the PPGR profiles of the two clusters is presented in Table 5. In addition to the significant differences in the Cmax differences between treatments, the volunteers in cluster 2 also displayed higher iAUC values than in cluster 1 which were significant for the control glucose solution and the 0% OJ. The Cmax values were also significantly higher in cluster 2 for the control glucose solution and slightly higher for the 0% OJ and 50% OJ. We also detected a lower C15 min value in cluster 2 following the intake of the 50% OJ and of the 100% OJ (p-value = 0.01).
| Cluster 1 (n = 12) | Cluster 2 (n = 11) | ||
|---|---|---|---|
| Mean ± SD | Mean ± SD | p-Value | |
| The results are the mean ± SD of (n = 12) for cluster 1 and (n = 11) for cluster 2. Clustering analysis was performed using the k-means clustering algorithm based on the Cmax differences between responses to the four test drinks. Further comparison between clusters was carried out using the Student's t test (level of significance p-value < 0.05). Statistical analyses were performed in R 4.5.0 (R Core Team, 2025). Abbreviations: Cmax, maximum (or peak) concentration; C15 min, concentration at time 15 min of the curve after ingestion of the test drink; iAUC, incremental area under the curve; n.s., not significant; OJ, orange juice. | |||
| Mean Cmax difference mg dL−1 (0% OJ – control glucose) | −2.7 ± 14.3 | −25.2 ± 11.0 | <0.001 |
| Mean Cmax difference mg dL−1 (50% OJ – control glucose) | −5.1 ± 19.6 | −27.8 ± 20.7 | 0.01 |
| Mean Cmax difference mg dL−1 (100% OJ – control glucose) | −3.3 ± 11.02 | −40.0 ± 10.7 | <0.001 |
| Mean Cmax difference mg dL−1 (100% OJ – 0% OJ) | −0.6 ± 9.7 | −14.8 ± 5.6 | <0.001 |
| iAUC (mg dL−1 120 min) (control glucose) | 1480 ± 912 | 3334 ± 1875 | 0.006 |
| iAUC (mg dL−1 120 min) (0% OJ) | 1142 ± 426 | 1923 ± 889 | 0.01 |
| iAUC (mg dL−1 120 min) (50% OJ) | 1046 ± 752 | 1622 ± 838 | 0.09 |
| iAUC (mg dL−1 120 min) (100% OJ) | 1337 ± 747 | 1468 ± 802 | n.s. |
| Mean Cmax mg dL−1 (control glucose) | 119.4 ± 16.8 | 150.0 ± 16.7 | <0.001 |
| Mean Cmax mg dL−1 (0% OJ) | 117.0 ± 10.2 | 125.0 ± 8.6 | 0.06 |
| Mean Cmax mg dL−1 (50% OJ) | 114.0 ± 16.4 | 122.0 ± 11.6 | n.s. |
| Mean Cmax mg dL−1 (100% OJ) | 116.6 ± 12.0 | 110.0 ± 8.6 | n.s. |
| Mean C15 min mg dL−1 (control glucose) | 102.5 ± 13.0 | 110.3 ± 19.4 | n.s. |
| Mean C15 min mg dL−1 (0% OJ) | 107.9 ± 13.5 | 108.0 ± 12.6 | n.s. |
| Mean C15 min mg dL−1 (50% OJ) | 106.0 ± 14.9 | 98.3 ± 13.4 | n.s. |
| Mean C15 min mg dL−1 (100% OJ) | 98.8 ± 7.3 | 90.6 ± 6.8 | 0.01 |
We further analyzed the differences between the two clusters for the insulin responses as well as for all the anthropometric and lifestyle characteristics investigated in this study. Most values were not significantly different between the clusters but we detected lower C15 min in cluster 2 following the intake of the 50% OJ (p-value = 0.04) and of the 100% OJ (not significant) (SI Table 5).
Multivariate analysis and metabolite tentative assessment
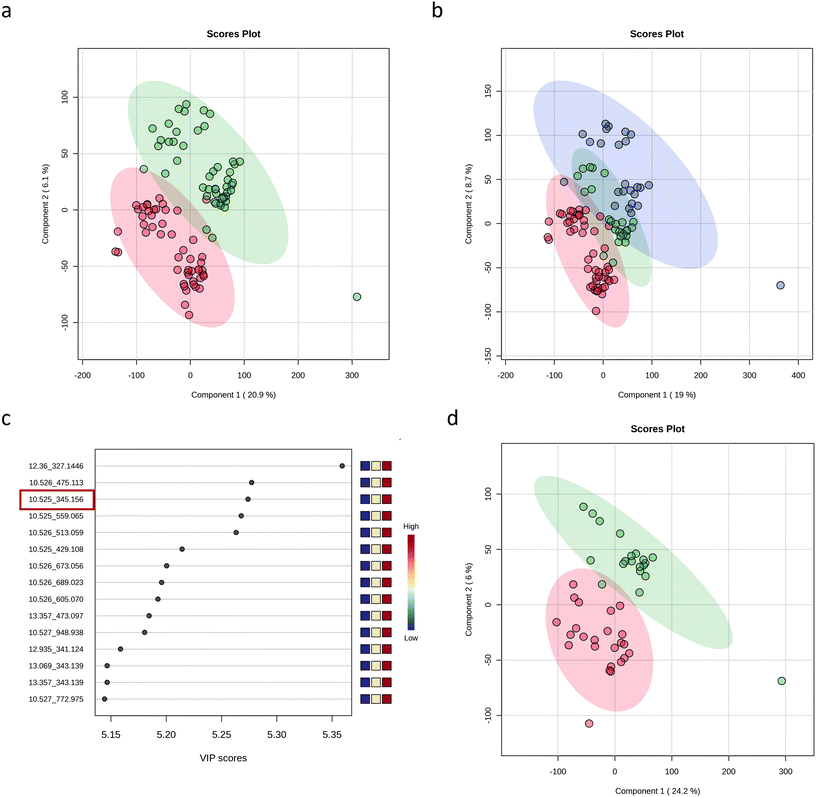
After preprocessing the raw data, a total of 38![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 878 entities were aligned and used to create the data matrix. The created data matrix was exported to MetaboAnalyst for its evaluation. For the first metabolome inspection, we created a PLS-DA model of the full data based only on the sampling time, i.e. 0 min compared to 60 min following the intake of the two drinks (100% OJ and 0% OJ) (Fig. 8a). The results showed a clear separation and expected discrimination between the two time points with the two first components explaining a total of 27% of the variability of the data.
878 entities were aligned and used to create the data matrix. The created data matrix was exported to MetaboAnalyst for its evaluation. For the first metabolome inspection, we created a PLS-DA model of the full data based only on the sampling time, i.e. 0 min compared to 60 min following the intake of the two drinks (100% OJ and 0% OJ) (Fig. 8a). The results showed a clear separation and expected discrimination between the two time points with the two first components explaining a total of 27% of the variability of the data.
On a second inspection, we created the PLS-DA model of the full data based on the sampling time and including the intake of the 100% OJ and 0% OJ as independent variables. In addition to the separation by time (0 min and 60 min), we were able to observe the separation at 60 min between the plasma metabolomes of the two drinks (Fig. 8b: 0% OJ (green dots) and 100% OJ (blue dots)) implying a general difference in the plasma metabolome of the volunteers after the intake of each of the two drinks. The VIP score plot of the PLS-DA displayed the discriminant entities responsible for the separation between the three groups. The most discriminating (VIP > 5) entities with higher concentrations 60 min after the intake of 100% OJ are depicted in (Fig. 8c). At the upper part of the list, we were able to identify limonene-8,9-diol-glucuronide (uroterpenol-glucuronide) with m/z 345.156 (SI Table 6). Additional discriminating compounds (VIP > 1) found at higher concentrations in the plasma of the volunteers 60 min after drinking the 100% OJ were tentatively identified and classified as terpenoids, phenolic acids, and polymethoxyflavones (SI Table 6). Correlation analyses between these detected plasma metabolites and the glucose iAUC, Cmax and C15 min individual responses associated with the intake of the 100% OJ detected significant positive associations between the Cmax values and dihydroferulic acid glucuronide (r = 0.49, p-value = 0.02) and perillic acid-8,9-diol glucuronide (r = 0.47, p-value = 0.03).
Finally, a PLS-DA model was developed based on the full dataset collected at 60 min after the intake of the 100% OJ and the 0% OJ but, this time, including the two clusters as independent variables. Samples were clearly separated into two groups independently of the type of juice ingested (100% OJ or 0% OJ) (Fig. 8d). These plasma metabolome-based differences strengthen the two responsive clusters and suggest that the main discriminant metabolites between the groups are not directly associated with the different composition of the two drinks (e.g. fruit matrix components like (poly)phenols). Nonetheless, we searched for metabolites that appeared only as a result of the 100% OJ intake (those absent at time 0 min and at time 60 min after the intake of 0% OJ) and that were discriminant between the two clusters by applying a biomarker analysis by univariate receiving operating characteristic (ROC). Following the evaluation of the candidates, we confirmed the presence of dihydroferulic acid glucuronide and perillic acid-8,9-diol glucuronide (SI Table 6) at a significantly higher intensity in volunteers of cluster 1 than in those of cluster 2 (SI Fig. 2).
Discussion
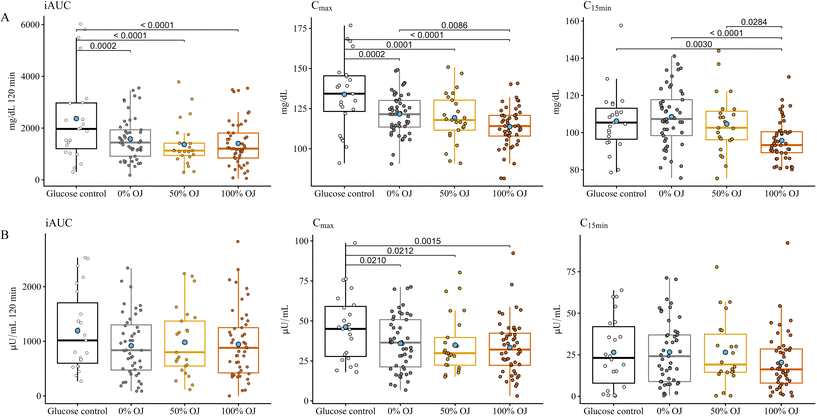
In the present study, we provide further evidence of the improving effect of the fruit matrix in the glycemic response to the consumption of orange juice. As expected, the overall response (iAUC) of the three test juices was significantly lower than that of the pure glucose control due to the different metabolic fate of fructose and sucrose.36,37 Since the three tested juices had the same total sugar composition and very small differences in the quantity of fruit matrix, we did not detect a change in iAUC in response to the various treatments (100% OJ or 50% OJ against the 0% OJ). Of note, the 100% OJ which contained the highest proportion of fruit matrix had a significant reducing effect on the peak glucose response (Cmax) compared with the 0% OJ containing only sugars. This effect might be beneficial since postprandial glucose spikes have been considered a risk factor for cardiovascular diseases.3,38,39 Gastric emptying has a major impact on the glycemic profile and small increases in the rate of gastric emptying can cause significant increases in the glucose levels even at early time points.40 The small but significant delay observed in the C15 min glucose levels following the intake of the 100% OJ might be related to a small delay in the gastric emptying and can contribute to attenuate the early rate of rise of glucose. Various components of the fruit matrix are able to influence the intestinal absorption of glucose. The addition of fiber-enriched products to the orange juice attenuates the PPGR in humans.41–43 Minerals like Na, K, P, Ca, and Mg are involved in the correct functioning of the Na+/K+-ATPase pump coupled to the SGLT1 active transport of glucose from the intestinal lumen.44–48 It is also plausible that the differences in the pH49 between the drinks can affect the transport system of sugars. The oxidized form of vitamin C (dehydroascorbic acid) has also been reported to interfere with the GLUT2 glucose transporter.50 In addition, the 100% OJ contained substantial quantities of (poly)phenols, mostly flavanones like hesperidin (∼122 mg) and narirutin (∼26 mg), as well as some hexosides of ferulic and sinapic acids (Table 2). Several in vitro and animal studies have shown that narirutin can reduce glucose absorption51 and that naringenin, the aglycone of narirutin, has the potential to inhibit the intestinal Na+-glucose cotransporter.52 Studies in animals also show that ferulic and sinapic acids have the potential to attenuate glucose levels by regulating carbohydrate digestive and glucose metabolizing enzymes, and promoting the uptake of glucose in muscles.53 In humans, hesperidin has been reported to reduce peak plasma glucose in healthy volunteers following the intake of a diluted orange juice with added hesperidin (49 mg per 200 ml portion). This effect was partially attributed to the inhibition of intestinal absorption by glucose transporters.54 All this evidence supports the hypothesis that the reduction in the glucose response observed in our study following the intake the 100% OJ against the 0% OJ is likely due to the combined action of the fruit matrix components. Our results are in line with those of Robayo et al.23 and of de Paiva et al.21 who examined 100% OJ but with different comparators and in overweight/obese individuals, although Papandreou et al.22 found no differences in the PPGR between 100% fresh OJ and a nectar-sweetened orange juice in healthy young females.PPGR is known to display a large intra- and inter-individual variability in healthy subjects.55 Our study confirmed a considerable interindividual variability in PPGR of the young normoglycemic male participants following the intake of the reference glucose but also in the Cmax responses to the three test juices. We successfully classified the participants into two subgroups, cluster 1 characterized by small differences in peak glucose across the three test drinks (designated as ‘low responders’) and, cluster 2 defined by more consistent and higher differences in peak responses between the drinks (designated as ‘high responders’). These results are in agreement with previous efforts to categorize individuals into groups with different responses and sensitivity to the intake of different types and quantities of carbohydrates.4,5,56 Recently, the differences in the PPGR were partially associated to the insulin sensitiveness or resistance and beta-cell function of the individuals, and metabolic phenotypes.56 Our study also corroborated a very high interindividual variability in the insulin responses of the participants but neither insulin nor any of the other characteristics investigated in this study (sex, age, BMI, diet, PA, and sleep patterns) differed significantly between the two groups of responders suggesting that, other factors such as the genetic, microbiome, and metabolomic profiles might be contributing to the observed interindividual variations in the PPGR.56
Several studies have already investigated the plasma metabolome during the 2 h oral glucose response and have identified a large number of important discriminatory metabolites including molecules like fatty acids, bile acids, amino acids, etc. The majority of these metabolites were investigated between 30 min and 60 min of the glucose response.57,58 Our metabolomic strategy corroborated a clear difference in the metabolome with time (0 min compared to 60 min) as well as between the postprandial response to 100% OJ and to 0% OJ. Some of the discriminatory metabolites identified can be associated with compounds present in the 100% OJ (sinapic, ferulic, polymethoxyflavones) as well as various terpenoids phase I and phase II (sulfates, glucuronides) derivatives. Some of these metabolites, i.e. ferulic acid sulfate, hydroxy-trimethoxyflavone-sulfate, hydroxy-tetramethoxyflavone-sulfate limonene-8,9-diol-glucuronide (uroterpenol-glucuronide), and perillic acid-8,9-diol glucuronide have been previously identified in humans as urinary biomarkers of orange juice consumption.59 Previous in vitro and animal model studies have also reported the potential effects of orange phytochemicals, i.e. terpenoids as limonene, phenolic acids as ferulic acid hexoside, and flavonoids as hesperidin and polymethoxyflavones (sinensetin, nobiletin and tangeretin) on glucose homeostasis.60–62 However, no clinical studies have yet been carried out to translate these preclinical results into valid clinical evidence. Our metabolomic analysis showed that some of the 100% OJ derived phytochemicals (dihydroferulic acid glucuronide and perillic acid-8,9-diol glucuronide) were positively correlated with the glucose Cmax response to the 100% OJ and were also present at higher levels in the plasma of the volunteers of cluster 1 than in those of cluster 2 (SI Fig. 2) supporting differences in the capacity to metabolize and absorb these compounds between the two groups of responders. Ferulic acid and limonene have been reported to exert beneficial effects on glucose metabolism through mechanisms such as enhanced cellular uptake and the regulation of key regulatory enzymes.63,64 We hypothesize that the conversion of these compounds into metabolites – such as dihydroferulic acid glucuronide and perillic acid-8,9-diol glucuronide – may reduce these effects. This loss of bioactivity aligns with our results which showed a correlation between higher plasma metabolite levels and increased glucose Cmax following the intake of the 100% OJ. Consistent with this, Cluster 1 displayed significantly higher levels of these metabolites alongside slightly higher glucose Cmax values following the 100% OJ intake.
Interindividual variability in glycemic response challenges one-size-fits-all dietary recommendations for fruit juices. Our classification into ‘low’ and ‘high’ responders has practical implications: for ‘low responders’, the glycemic impact of 100% OJ was minimal and similar to the sugar-matched drink. In contrast, ‘high responders’ saw a significant reduction in glucose peaks with 100% OJ, making it a more beneficial choice for them. The modulating effect of the fruit matrix was most evident in this high-responder group. Our study supports the need to stratify analyses by responder type as well as to further characterize the associated metabolic phenotype to develop more accurate, personalized dietary advice.56 The health implications of the OJ intake can be influenced by the lifestyle and background diet, e.g. total sugar and polyphenol intake. In our study, many participants had a high level of PA and consumed < 50 g of sugar daily and thus, the intake of 100% OJ may have a different impact than in individuals with higher-sugar diets and lower activity. Also, a 300 mL serving of 100% OJ provides substantial (poly)phenols (∼240 mg). Around 44% of the study participants consumed less than 650 mg day−1 and the 100% OJ can contribute toward reaching levels associated with health benefits (500–1000 mg day−1).65–67
Strengths and limitations
Strengths of the current study are: (i) a randomized crossover design with repeated measures, (ii) the comparison of well-characterized drinks with the same sugar profiles to determine the effect of the fruit matrix, and (iii) a detailed characterization of the participants which allowed us to have a fairly homogenous sample comprised of healthy young males with very similar anthropometric and lifestyle characteristics. Limitations of the study: (i) the results of our study cannot be generalized to other population groups and thus this research should be extended to females, older people, and individuals with metabolic disorders such as overweight/obesity, and pre-diabetes/T2DM, (ii) this study was an acute postprandial study that does not provide information on the effects of the habitual consumption of OJ on long-term glycemic control, (iii) the use of venous blood instead of capillary blood that offers a more immediate reflection of postprandial glucose fluctuations (this was, however, necessary to obtain the larger sample volume required for insulin and metabolomics analysis, and iv) although adequate for the primary purpose of this study, the data from our group of n = 25 should be considered as an exploratory investigation into interindividual variation. Proper characterization of the responder and non-responder clusters would require validation in larger more diverse populations with additional data on the biological factors (genetics, microbiome, metabolome) involved in the glucose response regulation to identify predictors of response. This approach should be extended to future studies to see how widely applicable the interindividual variation is across populations.Conclusions
We hypothesized a similar impact on glycemic response given that the ‘free sugars’ classification applies equally to natural sugars in fruit juices and sugars added to beverages. However, 100% orange juice attenuates the acute PPGR compared with its sugar components alone in healthy normoglycemic young males. This beneficial effect seems attributable to the fruit matrix components, although further research is required to test which ones are responsible. The glycemic responses were highly variable between individuals and participants could be classified into ‘low’ and ‘high’ responder phenotypes based on the magnitude of the differences in the reductions of post-prandial glucose spikes. This research reinforces the complexity of dietary responses to carbohydrates and emphasizes the need for larger studies to validate these responder metabolic phenotypes, elucidate the contributing mechanisms, and translate the findings into personalized, dietary recommendations concerning fruit juice intake.Abbreviations
| iAUC | Incremental area under the curve |
| JS | Jet stream |
| BMI | Body mass index |
| C max | Maximum (or peak) concentration |
| C 15 min | Concentration at time 15 min of the curve after ingestion of the test drink |
| ESI | Electrospray ionization |
| GLUT2 | Glucose transporter 2 |
| GOD | Glucose oxidase |
| GPAQ | Global physical activity questionnaire |
| LMM | Linear mixed model |
| MD | Mediterranean diet |
| MEDAS | Mediterranean diet adherence score |
| MEQ | Morning-evening questionnaire |
| MET | Metabolic equivalent of task |
| OJ | Orange juice |
| PA | Physical activity |
| PCA | Principal component analysis |
| PLS-DA | Partial least squares discriminant analyses PODperoxidase |
| PPGR | Postprandial glucose response |
| PSQI | Pittsburgh sleep quality index |
| PVDF | Polyvinylidene fluoride |
| Q-TOF LC/MS | Quadrupole time-of-flight liquid chromatography mass spectrometer |
| ROC | Receiving operating characteristic |
| SGLT1 | Sodium-glucose linked transporter 1 |
| SSB | Sugar-sweetened beverages |
| SST | Serum separator tubes |
| T max | Time to reach maximum concentration |
| T2DM | Type 2 diabetes mellitus |
| UHPLC | Ultra-high-performance liquid chromatography system |
| VIP | Variable importance in projection |
| WC | Waist circumference |
Author contributions
Conceptualization: M. T. García-Conesa, R. García-Villalba, C. Ruxton, G. Williamson, F. Tomás-Barberán. Methodology: M. T. García-Conesa, R. García-Villalba, C. Ruxton, G. Williamson, F. Tomás-Barberán, M. R. Arráez, J. Marhuenda, P. Zafrilla. Software: J. A. Tudela. Formal analysis: M. T. García-Conesa, R. García-Villalba, M. D. Frutos-Lisón, C. J. García, J. A. Tudela, J. Marhuenda, P. Zafrilla, F. Tomás-Barberán. Investigation: M. T. García-Conesa, R. García-Villalba, G. Williamson, F. Tomás-Barberán. Data curation: M. T. García-Conesa, J. A. Tudela. Writing – original draft: M. T. García-Conesa. Writing – review and editing: M. T. García-Conesa, R. García-Villalba, C. J. García, J. A. Tudela, C. Ruxton, G. Williamson, F. Tomás-Barberán. Supervision: M. T. García-Conesa, R. García-Villalba, F. Tomás-Barberán. Project administration: F. Tomás-Barberán. Funding acquisition: M. T. García-Conesa, R. García-Villalba, F. Tomás-Barberán.Conflicts of interest
CR has received fees for providing independent scientific advice to the Fruit Juice Science Centre, British Egg Industry Consortium, UK Tea and Infusions Association, The Proprietary Association of Great Britain, General Mills, Yoplait, Danone, Holland & Barrett, Tate & Lyle, Reading University and INRAE. She is also a board director of Quality Meat Scotland. GW is on the Scientific Advisory Board for Nutrilite, USA, and receives research funding from them, as well as from The Product Makers, Australia.All other authors report no conflicts of interest and all authors participated sufficiently in the work and take public responsibility for the content of the paper. The final manuscript has been seen and approved by all authors.
Data availability
Data described in the manuscript and analytic code are publicly and freely available without restriction at https://zenodo.org/records/15977192.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo04536c.
Acknowledgements
This study was funded by a grant from the Fruit Juice Science Centre, a project of the European Fruit Juice Association (AIJN). No employee or member of AIJN had any role in the design, execution, or interpretation of the research. AIJN did not restrict publication or dissemination of the study. This research was also supported by the Seneca Foundation from Murcia, Spain (FSRM/10.13039/100007801) through the Funding Program for Research Groups of Excellence (Grant reference: 23023/GERM/25). We would like to dedicate this article to the memory of Dr Mari Cruz Arcas Minarro who was instrumental in the development of the study concept. We also would like to express our sincere gratitude to the nursing staff at the UCAM involved in this study. We deeply appreciate their support in participant management and care, sample collection, and overall coordination throughout the intervention period. Last but not least, we are indebted to all the subjects who volunteered in the clinical trial.References
- D. Vejrazkova, M. Vankova, P. Lukasova, M. Hill, J. Vcelak and A. Tura, et al., The glycemic curve during the oral glucose tolerance test: is it only indicative of glycoregulation?, Biomedicines, 2023, 11, 1278 CrossRef CAS PubMed.
- T. K. Mathew, M. Zubair and P. Tadi, Blood Glucose Monitoring. [Updated 2023 Apr 23], in StatPearls, StatPearls Publishing, Treasure Island (FL), 2025 Search PubMed.
- P. R. E. Jarvis, J. L. Cardin, P. M. Nisevich-Bede and J. P. McCarter, Continuous glucose monitoring in a healthy population: understanding the post-prandial glycemic response in individuals without diabetes mellitus, Metabolism, 2023, 146, 155640 CrossRef CAS PubMed.
- D. Zeevi, T. Korem, N. Zmora, D. Israeli, D. Rothschild and A. Weinberger, et al., Personalized nutrition by prediction of glycemic responses, Cell, 2015, 163, 1079–1094 CrossRef CAS PubMed.
- J. Song, T. J. Oh and Y. Song, Individual postprandial glycemic responses to meal types by different carbohydrate levels and their associations with glycemic variability using continuous glucose monitoring, Nutrients, 2023, 15, 3571 CrossRef CAS PubMed.
- B. J. Narang, G. Atkinson, J. T. Gonzalez and J. A. Betts, A Tool to Explore Discrete-Time Data: The Time Series Response Analyser, Int. J. Sport Nutr. Exercise Metab., 2020, 30, 374–381 CAS.
- D. J. Jenkins, T. M. Wolever, R. H. Taylor, H. Ghafari, A. L. Jenkins, H. Barker and M. J. Jenkins, Rate of digestion of foods and postprandial glycaemia in normal and diabetic subjects, Br. Med. J., 1980, 281, 14–17 CrossRef CAS PubMed.
- World Health Organization (WHO), Diet Nutrition and the Prevention of Chronic Diseases. Report of a Joint WHO/FAO Expert Consultation. WHO Technical Report Series 916, 2003, Geneva.
- D. J. Mela and E. M. Woolner, Perspective: Total, Added, or Free? What Kind of Sugars Should We Be Talking About?, Adv. Nutr., 2018, 9, 63–69 CrossRef PubMed.
- C. M. Weaver and D. I. Givens, Overview: the food matrix and its role in the diet, Crit. Rev. Food Sci. Nutr., 2025, 4, 1–18 Search PubMed.
- International Diabetes Federation, Diabetes and healthy nutrition, Available on line: https://idf.org/about-diabetes/diabetes-management/healthy-nutrition/(accessed the 24th of March 2025).
- C. H. S. Ruxton and M. Myers, Fruit juices: are they helpful or harmful? An evidence review, Nutrients, 2021, 13, 1815 CrossRef CAS PubMed.
- K. A. Della Corte, T. Bosler, C. McClure, A. E. Buyken, J. D. LeCheminant and L. Schwingshackl, et al., Dietary sugar intake and incident type 2 diabetes risk: a systematic review and dose-response meta-analysis of prospective cohort studies, Adv. Nutr., 2025, 16, 100413 CrossRef CAS PubMed.
- L. D'Elia, M. Dinu, F. Sofi, M. Volpe and P. Strazzullo (SINU Working Group), 100% Fruit juice intake and cardiovascular risk: a systematic review and meta-analysis of prospective and randomized controlled studies, Eur. J. Nutr., 2021, 60, 2449–2467 CrossRef PubMed.
- M. M. Murphy, E. C. Barrett, K. A. Bresnahan and L. M. Barraj, 100% Fruit juice and measures of glucose control and insulin sensitivity: a systematic review and meta-analysis of randomized controlled trials, J. Nutr. Sci., 2017, 6, e59 CrossRef PubMed.
- B. Wang, K. Liu, M. Mi and J. Wang, Effect of fruit juice on glucose control and insulin sensitivity in adults: a meta-analysis of 12 randomized controlled trials, PLoS One, 2014, 9, e95323 CrossRef PubMed.
- H. Alhabeeb, M. H. Sohouli, A. Lari, S. Fatahi, F. Shidfar and O. Alomar, et al., Impact of orange juice consumption on cardiovascular disease risk factors: a systematic review and meta-analysis of randomized-controlled trials, Crit. Rev. Food Sci. Nutr., 2022, 62, 3389–3402 CrossRef CAS PubMed.
- L. Li, N. Jin, K. Ji, Y. He, H. Li and X. Liu, Does chronic consumption of orange juice improve cardiovascular risk factors in overweight and obese adults? A systematic review and meta-analysis of randomized controlled trials, Food Funct., 2022, 13, 11945–11953 RSC.
- M. Motallaei, N. Ramezani-Jolfaie, M. Mohammadi, S. Shams-Rad, A. S. Jahanlou and A. Salehi-Abargouei, Effects of orange juice intake on cardiovascular risk factors: A systematic review and meta-analysis of randomized controlled clinical trials, Phytother. Res., 2021, 35, 5427–5439 CrossRef PubMed.
- V. Chen, T. A. Khan, L. Chiavaroli, A. Ahmed, D. Lee and C. W. C. Kendall, et al., Relation of fruit juice with adiposity and diabetes depends on how fruit juice is defined: a re-analysis of the EFSA draft scientific opinion on the tolerable upper intake level for dietary sugars, Eur. J. Clin. Nutr., 2023, 77, 699–704 CrossRef PubMed.
- A. de Paiva, D. Gonçalves, P. Ferreira, E. Baldwin and T. Cesar, Postprandial effect of fresh and processed orange juice on the glucose metabolism, antioxidant activity and prospective food intake, J. Funct. Foods, 2019, 52, 302–309 CrossRef CAS.
- D. Papandreou, E. Magriplis, M. Abboud, Z. Taha, E. Karavolia and C. Karavolias, et al., Consumption of raw orange, 100% fresh orange juice, and nectar- sweetened orange juice-effects on blood glucose and insulin levels on healthy subjects, Nutrients, 2019, 11, 2171 CrossRef CAS PubMed.
- S. Robayo, M. Kucab, S. E. Walker, K. Suitor, K. D'Aversa and O. Morello, et al., Effect of 100% orange juice and a volume-matched sugar-sweetened drink on subjective appetite, food intake, and glycemic response in adults, Nutrients, 2024, 16, 242 Search PubMed.
- H. Kang, Sample size determination and power analysis using the G*Power software, J. Educ. Eval. Health Prof., 2021, 18, 17 CrossRef PubMed.
- G. Lin, R. Siddiqui, Z. Lin, J. M. Blodgett, S. N. Patel and K. N. Truong, et al., Blood glucose variance measured by continuous glucose monitors across the menstrual cycle, NPJ Digit. Med., 2023, 6, 140 CrossRef PubMed.
- M. Bhatt, N. Rai, S. Pokhrel, P. Acharya, S. Marhatta and D. Khanal, et al., Standardization of visible kinetic assay for the estimation of plasma glucose by glucose oxidase and peroxidase method, J. Manmohan Mem. Inst. Health Sci., 2021, 7, 49–59 CrossRef.
- Y. Shen, W. Prinyawiwatkul and Z. Xu, Insulin: a review of analytical methods, Analyst, 2019, 144, 4139–4148 RSC.
- A. Kassambara, rstatix: Pipe-Friendly Framework for Basic Statistical Tests. R package version 0.7.2, 2023 Search PubMed.
- G. Hesselmann, Applying Linear Mixed Effects Models (LMMs) in Within-Participant Designs with Subjective Trial-Based Assessments of Awareness-a Caveat, Front. Psychol., 2018, 25, 788 CrossRef PubMed.
- D. Bates, M. Mächler, B. Bolker and S. Walker, Fitting Linear Mixed-Effects Models Using lme4, J. Stat. Softw., 2015, 67, 1–48 Search PubMed.
- R. Lenth, emmeans: Estimated Marginal Means, aka Least-Squares Means. R package version 1.11.2-80001, 2025 Search PubMed.
- A. Kassambara and F. Mundt, Factoextra: Extract and Visualize the Results of Multivariate Data Analyses. R Package Version 1.0.7, 2020 Search PubMed.
- R Core Team, R: A Language and Environment for Statistical Computing (Version 4.5.0) [Computer Software], R Foundation for Statistical Computing, 2025 Search PubMed.
- H. Wickham, R. François, L. Henry, K. Müller and D. Vaughan, dplyr: A Grammar of Data Manipulation. R package version 1.1.2, 2023 Search PubMed.
- H. Wickham, ggplot2: Elegant Graphics for Data Analysis, Springer-Verlag New York, 2016. ISBN 978-3-319-24277-4 Search PubMed.
- S. J. Dholariya and J. A. Orrick, Biochemistry, Fructose Metabolism. [Updated 2022 Oct 17], in StatPearls, StatPearls Publishing, Treasure Island (FL), 2025 Search PubMed.
- J. T. Gonzalez, Are all sugars equal? Role of the food source in physiological responses to sugars with an emphasis on fruit and fruit juice, Eur. J. Nutr., 2024, 63, 1435–1451 Search PubMed.
- E. Bonora, Postprandial peaks as a risk factor for cardiovascular disease: epidemiological perspectives, Int. J. Clin. Pract., 2002, 129, 5–11 Search PubMed.
- N. M. J. Hanssen, M. J. Kraakman, M. C. Flynn, P. R. Nagareddy, C. G. Schalkwijk and A. J. Murphy, Postprandial Glucose Spikes, an Important Contributor to Cardiovascular Disease in Diabetes?, Front. Cardiovasc. Med., 2020, 18, 570553 Search PubMed.
- C. S. Marathe, C. K. Rayner, K. L. Jones and M. Horowitz, Relationships between gastric emptying, postprandial glycemia, and incretin hormones, Diabetes Care, 2013, 36, 1396–1405 Search PubMed.
- H. Dong, C. Rendeiro, A. Kristek, L. J. Sargent, C. Saunders and L. Harkness, et al., Addition of orange pomace to orange juice attenuates the increases in peak glucose and insulin concentrations after sequential meal ingestion in men with elevated cardiometabolic risk, J. Nutr., 2016, 146, 1197–1203 CrossRef CAS PubMed.
- N. Bosch-Sierra, R. Marqués-Cardete, A. Gurrea-Martínez, C. Grau-Del Valle, C. Morillas and A. Hernández-Mijares, et al., Effect of fiber-enriched orange juice on postprandial glycemic response and satiety in healthy individuals: an acute, randomized, placebo-controlled, double-blind, crossover study, Nutrients, 2019, 11, 3014 CrossRef CAS PubMed.
- G. Guzman, D. Xiao, D. Liska, E. Mah, K. Sanoshy and L. Mantilla, et al., Addition of orange pomace attenuates the acute glycemic response to orange juice in healthy adults, J. Nutr., 2021, 151, 1436–1442 CrossRef PubMed.
- A. G. Therien and R. Blostein, Mechanisms of sodium pump regulation, Am. J. Physiol.: Cell Physiol., 2000, 279, C541–C566 CrossRef CAS PubMed.
- C. Du, S. Chen, H. Wan, L. Chen, L. Li and H. Guo, et al., Different functional roles for K+ channel subtypes in regulating small intestinal glucose and ion transport, Biol. Open, 2019, 8, bio042200 CrossRef CAS PubMed.
- M. Barbagallo and L. J. Dominguez, Magnesium and type 2 diabetes, World J. Diabetes, 2015, 6, 1152–1157 Search PubMed.
- P. L. Jorgensen, K. O. Hakansson and S. J. Karlish, Structure and mechanism of Na,K-ATPase: functional sites and their interactions, Annu. Rev. Physiol., 2003, 65, 817–849 Search PubMed.
- L. Chen, B. Tuo and H. Dong, Regulation of intestinal glucose absorption by ion channels and transporters, Nutrients, 2016, 8, 43 CrossRef PubMed.
- M. Ortiz, M. Lluch and F. Ponz, Effect of the pH on intestinal absorption of sugars in vivo, Rev. Esp. Fisiol., 1979, 35, 239–242 Search PubMed.
- C. P. Corpe, P. Eck, J. Wang, H. Al-Hasani and M. Levine, Intestinal dehydroascorbic acid (DHA) transport mediated by the sodium-dependent glucose transporter (SGLT) 1, PLoS One, 2013, 8, e74119 CrossRef PubMed.
- S. Mitra, M. S. Lami, T. M. Uddin, R. Das, F. Islam and J. Anjum, et al., Prospective multifunctional roles and pharmacological potential of dietary flavonoid narirutin, Biomed. Pharmacother., 2022, 150, 112932 CrossRef CAS PubMed.
- J. M. Li, C. T. Che, C. B. Lau, P. S. Leung and C. H. Cheng, Inhibition of intestinal and renal Na+-glucose cotransporter by naringenin, Int. J. Biochem. Cell Biol., 2006, 38, 985–995 CrossRef CAS PubMed.
- V. F. Salau, O. L. Erukainure, N. A. Koorbanally and M. S. Islam, Ferulic acid promotes muscle glucose uptake and modulate dysregulated redox balance and metabolic pathways in ferric-induced pancreatic oxidative injury, J. Food Biochem., 2022, 46, e13641 CrossRef CAS PubMed.
- A. Kerimi, J. S. Gauer, S. Crabbe, J. W. Cheah, J. Lau and R. Walsh, et al., Effect of the flavonoid hesperidin on glucose and fructose transport, sucrase activity and glycemic response to orange juice in a crossover trial on healthy volunteers, Br. J. Nutr., 2019, 121, 782–792 CrossRef CAS PubMed.
- S. Hirsch, G. Barrera, L. Leiva, M. P. de la Maza and D. Bunout, Variability of glycemic and insulin response to a standard meal, within and between healthy subjects, Nutr. Hosp., 2013, 28, 541–544 CAS.
- Y. Wu, B. Ehlert, A. A. Metwally, D. Perelman, H. Park and A. W. Brooks, et al., Individual variations in glycemic responses to carbohydrates and underlying metabolic physiology, Nat. Med., 2025, 31, 2232–2243 CrossRef PubMed.
- C. Wildberg, A. Masuch, K. Budde, G. Kastenmüller, A. Artati and W. Rathmann, et al., Plasma metabolomics to identify and stratify patients with impaired glucose tolerance, J. Clin. Endocrinol. Metab., 2019, 104, 6357–6370 Search PubMed.
- X. Zhao, A. Peter, J. Fritsche, M. Elcnerova, A. Fritsche and H. U. Häring, et al., Changes of the plasma metabolome during an oral glucose tolerance test: is there more than glucose to look at?, Am. J. Physiol. Endocrinol. Metab., 2009, 296, E384–E393 CrossRef CAS PubMed.
- M. Tomás-Navarro, J. L. Navarro, F. Vallejo and F. A. Tomás-Barberán, Novel urinary biomarkers of orange juice consumption, interindividual variability, and differences with processing methods, J. Agric. Food Chem., 2021, 69, 4006–4017 Search PubMed.
- M. Bacanlı, H. G. Anlar, S. Aydın, T. Çal, N. Arı and Ü. Ündeğer Bucurgat, et al., d-Limonene ameliorates diabetes and its complications in streptozotocin-induced diabetic rats, Food Chem. Toxicol., 2017, 110, 434–442 CrossRef PubMed.
- F. Kaviani, I. Baratpour and S. Ghasemi, The antidiabetic mechanisms of hesperidin: hesperidin nanocarriers as promising therapeutic options for diabetes, Curr. Mol. Med., 2024, 24, 1483–1493 CrossRef CAS PubMed.
- X. Li, J. Wu, F. Xu, C. Chu, X. Li and X. Shi, et al., Use of ferulic acid in the management of diabetes mellitus and its complications, Molecules, 2022, 27, 6010 CrossRef CAS PubMed.
- D. A. Jacobo-Velázquez, Ferulic Acid: Mechanistic Insights and Multifaceted Applications in Metabolic Syndrome, Food Preservation, and Cosmetics, Molecules, 2025, 30, 3716 CrossRef PubMed.
- L. T. Al Kury, A. Abdoh, K. Ikbariah, B. Sadek and M. Mahgoub, In Vitro and In Vivo Antidiabetic Potential of Monoterpenoids: An Update, Molecules, 2021, 27, 182 Search PubMed.
- G. Williamson and B. Holst, Dietary reference intake (DRI) value for dietary (poly)phenols: are we heading in the right direction?, Br. J. Nutr., 2008, 99, S55–S58 Search PubMed.
- A. Tresserra-Rimbau, E. B. Rimm, A. Medina-Remón, M. A. Martínez-González, R. de la Torre and D. Corella, et al. (PREDIMED Study Investigators), Inverse association between habitual polyphenol intake and incidence of cardiovascular events in the PREDIMED study, Nutr. Metab. Cardiovasc. Dis., 2014, 24, 639–647 CrossRef CAS PubMed.
- R. Zamora-Ros, M. Rabassa, A. Cherubini, M. Urpí-Sardà, S. Bandinelli and L. Ferrucci, et al., High concentrations of a urinary biomarker of polyphenol intake are associated with decreased mortality in older adults, J. Nutr., 2013, 143, 1445–1450 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |