Open Access Article
Open Access ArticlePrevention of colorectal cancer and modulation of murine gut microbiota using functional meats
Patrick L.
McAlpine
abc,
Javier
Borondo
d,
Mar
Grande
d,
Lidia
Gil-Martínez
e,
Ana
Falcón-Piñeiro
e,
Alberto
Baños
e,
Estefanía
Crespo Losada
f,
Francisco M.
Reyes-Sosa
f,
Miriam
Caro Ruiz
g,
Isabel
Uribe Oyarbide
g,
María
Álvarez Gil
h,
Jesús Fidel
Delgado Ramallo
h,
Ana Martín
Atienza
i,
Diego
García Miguélez
i,
Fernando
Santamaría-Martos
j,
Jaume Palau
Clivillé
j,
Claudio J.
Villar
abc,
Javier
Fernández
*abc and
Felipe
Lombó
 *abc
*abc
aResearch Group BIONUC (Biotechnology of Nutraceuticals and Bioactive Compounds), Departamento de Biología Funcional, Área de Microbiología, Universidad de Oviedo, 33006, Oviedo, Spain. E-mail: fernandezfjavier@uniovi.es; lombofelipe@uniovi.es
bIUOPA (Cancer Research Institute of the Principality of Asturias), 33006 Oviedo, Spain
cISPA (Health Research Institute of the Principality of Asturias), 33011 Oviedo, Spain
dAGrowingData, 04002 Almería, Spain
eDMC Research Center, 18620 Alhendín, Spain
fBDI-Biotechnology Development for Industry SL, 47151 Boecillo, Spain
gFUDin, 26315 Alesón, Spain
hNeoalgae, 33211 Gijón, Spain
iBTSA, 28805 Alcalá de Henares, Spain
jARGAL, 25424 Miralcamp, Spain
First published on 23rd February 2026
Abstract
Extensive studies associate processed meat products with a higher risk of colorectal cancer, while consumption of plant nutraceuticals has the opposite effect, via the reduction of oxidative stress and mucosal inflammation, gut microbiota modification, and direct effects upon tumor cells. In this work, five different processed meat products have been formulated, containing two carotenoids, two omega-3 fatty acids, catechins, carnosic acid, propyl propane thiosulfonate (PTSO), a postbiotic from Lactiplantibacillus plantarum, and the vitamins α-tocopherol, and ascorbyl palmitate. These functional meat products have been tested in a murine model of colorectal cancer. Tumor development, inflammatory biomarkers, and the impact upon the gut microbiota were quantified. PTSO plus the L. plantarum postbiotic exerted an anti-inflammatory effect. The carotenoids exhibited a potent prebiotic-like effect. The functional meat product containing all nutraceutical compounds used in this study reduced the development of macroscopic colorectal tumors, indicating that a synergy between all these compounds is necessary to achieve potent antitumor effects. These results demonstrate that functional meat products can function as an effective system to deliver nutraceutical compounds to the host and promote disease prevention.
1. Introduction
Colorectal cancer (CRC) is one of the most common types of neoplasia worldwide. Specifically, it is the third most common cancer for both men and women, with 1.8 million new cases in 2018. It is also the second leading cause of cancer related mortality, with 880![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 deaths in 2018.1 While hereditary risk factors for CRC exist, most cases are sporadic and associated with environmental and lifestyle patterns.2 Amongst these lifestyle patterns, dietary consumption has been found to be particularly important, as several studies have highlighted the protective effects of healthy dietary patterns,3,4 emphasizing the consumption of fruits and vegetables, dietary fiber, calcium, and vitamins.5
000 deaths in 2018.1 While hereditary risk factors for CRC exist, most cases are sporadic and associated with environmental and lifestyle patterns.2 Amongst these lifestyle patterns, dietary consumption has been found to be particularly important, as several studies have highlighted the protective effects of healthy dietary patterns,3,4 emphasizing the consumption of fruits and vegetables, dietary fiber, calcium, and vitamins.5
Among the lifestyle factors that promote CRC, the evidence around processed and red meat consumption have been declared convincing and probable, respectively.5 Similarly, the International Agency for Research on Cancer (IARC) has declared processed meats as a group 1 carcinogen. Processed meats are classified as meats that are transformed through salting, smoking, curing, or fermentation.1 Numerous mechanisms have been proposed to explain the associations of red and processed meats with CRC, including the formation of N-nitroso compounds and heterocyclic aromatic amines, as well as the high abundance of heme iron.6 Both red and processed meat are major ingredients in the Western style diet, but also in numerous traditional local diets.7 It has even been proposed that increasing rates of meat consumption in non-Western countries may partially explain the parallel increases in CRC rates in these countries.1
While the ideal strategy to reduce the rising number of CRC cases globally could be to shift dietary habits away from high meat consumption and towards non-processed and plant-based diets, initiatives to promote this switch have been poorly accepted by the public, as evidenced by an increasing meat consumption worldwide.8 An alternative approach may be to reformulate processed meat products into more healthy alternatives, such as functional meat products. Studies by our research group have demonstrated that the addition of plant compounds such as prebiotic fibers9 and polyphenols10 to processed meat products is a highly effective strategy to reduce CRC development in murine models for this disease. The effectiveness of these previous functional meat products was due, at least in part, to their gut microbiota-modulating and anti-inflammatory properties.
The gut microbiota has been found to play a central role in CRC development.11 A dysbiotic gut microbiota can reduce the intestinal mucus lining, damage the colon's epithelial integrity, promote the passage of bacterial antigens and lipopolysaccharides through the intestinal epithelium, and induce chronic mucosal inflammation.12 In severe cases, this inflammation can develop into irritable bowel syndrome (IBS) and promote the development of CRC.13 In contrast, a healthy gut microbiota is better adapted to the fermentation of dietary plant derived carbohydrates (prebiotic fibers) rather than host mucins. This healthy gut microbiota fermentation of prebiotic fibers produces short chain fatty acids (SCFAs) such as acetate, propionate, and butyrate, in greater quantities, promoting intestinal integrity (higher expression of tight junctions proteins) and reducing inflammation.14
In addition to the previous work done by our group, research has demonstrated the excellent capacity of sausage-like formulations to retain highly diverse compounds. The mixture of lipids, water, starches, fibers, and salts in sausages results in a highly complex mixture with affinities for a potentially wide variety of compounds. The presence of saturated lipids from the meat itself and unsaturated lipids from the addition of vegetable oils creates an environment that could be highly capable of retaining hydrophobic compounds. Similarly, the water content, and the ability of the starches and fibers to retain this water, create an environment that could be highly capable of retaining hydrophilic compounds. It has been demonstrated that the mixture of these ingredients forms gel-like matrixes that are highly capable of retaining exogenous lipids and polyphenols.15–18
In this study, we have formulated five different functional meat products and tested them in a chemically induced murine model of CRC (Rattus norvegicus). These meat products were formulated as cooked sausages and contained various mixtures of β-carotene, astaxanthin, α-linolenic acid (ALA), docosahexaenoic acid (DHA), catechins, carnosic acid, propyl propane thiosulfonate (PTSO), a heat inactivated postbiotic strain of Lactiplantibacillus plantarum DMC-26, α-tocopherol (vitamin E), and ascorbyl palmitate (a lipophilic version of vitamin C).
β-Carotene and astaxanthin are both carotenoids that are readily found in the human diet. β-Carotene is present in orange vegetables such as carrots, pumpkin, oranges and squash.19 Astaxanthin is primarily found in seafood such as salmon and shrimp.20 Both are regarded as potent lipophilic antioxidants, although astaxanthin is superior to β-carotene in free radical scavenging activity. In addition to their antioxidant properties, β-carotene has also been found to reduce the capacity for self-renewal in CRC stem cells,21 inhibit the polarization of macrophages, and inhibit the activation of fibroblasts in the tumor microenvironment.22 Similarly, astaxanthin has been found to inhibit colorectal carcinogenesis due to anti-proliferative,23,24 anti-metastatic,25 antioxidant,26 and anti-inflammatory27 activities.
ALA and DHA are both essential omega-3 fatty acids. ALA is primarily found in seeds such as flax and chia,28 while DHA is primarily found in seafood.29 Both compounds have been found to have CRC preventing activities,30–33 with proposed mechanism including a reduction in inflammation and increased oxidative stress in tumor tissues.34 ALA has been found to promote apoptosis due to mitochondrial stress.35 DHA has been found to increase tumor TNF-α secretion,36 increase p53 expression, and decrease survivin expression in stem cell-like CRC cells.37
Green tea derived catechins (a class of flavonoids) and rosmarin derived carnosic acid (a diterpenoid) exhibit a wide range of bioactivities. Several studies have demonstrated that treatment with flavonoids reduces colitis-associated inflammation38 and CRC tumors development,39 while similar results have also been found for green tea catechins40–42 and carnosic acid.43,44 The mechanisms by which flavonoids can reduce CRC development include depletion of reactive oxygen species, reduction of intestinal inflammation, and inhibition of tumor associated epigenetic modifications.45 (−)-Epigallocatechin gallate (EGCG), which is the most studied catechin from green tea, has been shown to inhibit the DNA methyltransferase activity and therefore reverse the silencing of genes in cancer cells,46 inhibit the JAK/STAT pathway,47 and reduce the expression of insulin like growth factor-1 (IGF-1).48
PTSO is a relatively poorly studied organosulfur compound derived from onion.49 Organosulfur compounds are a diverse set of compounds that are primarily produced by vegetables in the Allium genus and in the Brassicaceae family which have well demonstrated activities against colorectal tumors.50 Although CRC preventing activities have been found for numerous organosulfur compounds, to our knowledge, the impact of PTSO has never directly been investigated. Until now, PTSO has been found to be non-toxic,51 antioxidant, antimicrobial,52 anti-inflammatory,53–55 and broadly antitumoral,56 where it has been shown to be highly toxic to tumor cells while being largely harmless to healthy immune cells. PTSO is a chemically stable, saturated thiosulfonate compound biosynthesized from S-propyl cysteine sulfoxide (propiin), a sulfur-containing cysteine sulfoxide that accumulates in Allium cepa. Although structurally distinct from allicin (diallyl thiosulfinate), the major bioactive compound in Allium sativum, PTSO exhibits comparable bioactivities. Allicin has been reported to alleviate colitis symptoms, increase intratumor oxidative stress and inhibit the JAK/STAT pathway in CRC studies.50
Lactiplantibacillus plantarum (previously Lactobacillus plantarum), is a very well described probiotic bacterial species known for its production of lactic acid.57 Its use as a live probiotic has been shown to reduce CRC development by inducing apoptosis and reducing angiogenesis.58 In this study, L. plantarum DMC-26 cells have been cooked along with the rest of the meat product and thus will be present as a postbiotic rather than as a live probiotic in the final functional meat product. L. plantarum exopolysaccharides alone are sufficient to induce apoptosis in CRC cells59 and potentiate the antitumor immune response.60 Unpublished data from the manufacturer confirms that Lactiplantibacillus plantarum DMC-26 produces bioactive exopolysaccharides.
α-Tocopherol is a common form of vitamin E and ascorbyl palmitate is a liposoluble form of vitamin C. Both are liposoluble antioxidants, but neither has been found to have effects against CRC on their own.61,62 However, α-tocopherol is known to prevent lipid peroxidation of cell membranes and promote endothelial integrity,63 while ascorbyl palmitate is used in the food industry to preserve lipids, but its pharmaceutical activities are poorly investigated.64
Thus, our previous studies have demonstrated, on a preclinical scale, the feasibility of functional meat products to retain nutraceutical compounds, deliver them to the host, and induce health promoting effects. In this study, we set out to demonstrate this feasibility on a broader preclinical scale, using a wide range of nutraceutical compounds with diverse chemical and physiological properties. Furthermore, we tested the potential of mixing numerous compounds into the same functional meat product to maximize their synergistic effects.
2. Methods
2.1. Generation of functional meat products
Seven dietary groups were generated in the rats cohorts. Group control (C), which consumed only standard rat feed, was formulated to have no dietary prebiotic fiber. For this, a formula with 14.6% protein, 53.6% carbohydrate, 6% lipid, 20% cellulose (0% prebiotic fiber), and 3.26 kcal g−1 (18% from proteins, 66% from carbohydrates, 16% from lipids) was designed, based on the 2014 Teklad Global 14% standard diet (Research Diets, New Jersey, USA).Group control meat (CM) consumed a mixture of 50% modified rat feed (50% daily intake; with 10.8% protein, 58.2% carbohydrate, 4.2% lipid and 20.6% cellulose (0% prebiotic fiber)) and 50% control meat product with no added nutraceuticals (18.22% protein, 6.48% carbohydrate, 7.4% lipid, 5.69% non-prebiotic fiber, and 1.56 kcal g−1). This modified feed was formulated to provide, together with the control meat, the same macro- and micro-nutrients as the control feed, to ensure that all the rats had similar intakes of these nutrients.
Group functional meat 1 (FM1) consumed the modified rat feed along with a functional meat product containing a carotenoid mixture consisting of β-carotene and astaxanthin, an omega-3 fatty acid mixture consisting of ALA and DHA, plus catechins, carnosic acid, an antioxidant mixture consisting of α-tocopherol and ascorbyl palmitate, PTSO, and L. plantarum DMC-26 (pasteurized during the meat manufacturing, therefore as a postbiotic).
Group functional meat 2 (FM2) consumed the modified rat feed along with a functional meat product containing PTSO, L. plantarum DMC-26, catechins, and carnosic acid.
Group functional meat 3 (FM3) consumed the modified rat feed along with a functional meet product containing PTSO, L. plantarum DMC-26, and the carotenoid mixture.
Group functional meat 4 (FM4) consumed the modified rat feed along with a functional meat product containing PTSO, L. plantarum DMC-26, and the omega-3 fatty acid mixture.
Group functional meat 5 (FM5) consumed the modified rat feed along with a functional meat product containing PTSO and L. plantarum DMC-26.
The target concentration for each nutraceutical was the following: 200 mg kg−1 of β-carotene and astaxanthin, 5 g kg−1 of ALA and DHA, 100 mg kg−1 catechins, 200 mg kg−1 carnosic acid, 100 mg kg−1 α-tocopherol, 500 mg kg−1 ascorbyl palmitate, 200 mg kg−1 PTSO, and 109 CFU g−1 of L. plantarum DMC-26 (pasteurized). The nutraceuticals used in each group, as well as their empirical concentrations, are summarized in Table 1. The concentrations of each nutraceutical were selected based on demonstrated bioactivities in the literature,22,27,30,40,44,51,61,65–67 in consultation with the European Food Safety Authority (EFSA) dietary guidelines and including technological and organoleptic limitations during the elaboration of the meat products (thermal stability, color, taste, etc.).
| Cohort | Meat | β-Carotene | Astaxanthin | α-Linolenic acid | Docosahexaenoic acid | α-Tocopherol | Catechins | Ascorbyl palmitate | Carnosic acid | Propyl propane thiosulfonate | Lactobacillus plantarum DMC-26 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Control (C) | |||||||||||
| Control Meat (CM) | X | ||||||||||
| Functional Meat 1 (FM1) | X | X | X | X | X | X | X | X | X | X | X |
| Functional Meat 2 (FM2) | X | X | X | X | X | ||||||
| Functional Meat 3 (FM3) | X | X | X | X | X | ||||||
| Functional Meat 4 (FM4) | X | X | X | X | X | ||||||
| Functional Meat 5 (FM5) | X | X | X | ||||||||
| Concentration in meat: | 50 mg kg−1 | 50 mg kg−1 | 5 g kg−1 | 5 g kg−1 | 100 mg kg−1 | 100 mg kg−1 | 500 mg kg−1 | 200 mg kg−1 | 200 mg kg−1 | 109 CFU g−1 (pasteurized) |
The meat products were elaborated by the research center FUDin (Alesón, Spain) using the manufacturers standard protocol for mixture and pasteurization. They were supplied in a sausage-like consistency, and contained 79.2% pork loin, 3.5% whole wheat flour, 7.1% water, 3.5% starch, 2.0% fiber, 3.0% vegetable oil, and 1.6% salt. The concentration of each nutraceutical was confirmed by high performance liquid chromatography (HPLC) or gas chromatography (GC). Pork meat was supplied by ARGAL S.A. (Miralcamp, Spain) and Hijo de Martinez Somalo (Baños de Río Tobía, Spain). The β-carotene and astaxanthin were supplied by Neoalgae (Gijón, Spain). The ALA, DHA, α-tocopherol, ascorbyl palmitate, catechins, and carnosic acid were supplied by BTSA S.L. (Alcalá de Henares, Spain). The PTSO and L. plantarum DMC-26 were supplied by DMC Research Center (Alhendín, Spain).
2.2. Murine model for CRC
All animal experiments were conducted in the Animal Facilities at the University of Oviedo (authorized facility no. ES330440003591) in accordance with relevant guidelines and regulations (Federation of European Laboratory Animal Science Associations, FELASA). All experiments were approved by the Ethics Committee of the Principality of Asturias (authorization code PROAE 14/2022).The murine model for CRC was conducted using previously established protocols.9,10,39,68–70A total of 70 males 4 weeks old Fischer 344 rats were purchased from Charles River Laboratories (Lyon, France). The rats were given a 1 week adaptation period before beginning with their respective diets. The rats were then split into 7 groups of 10 rats each, separated into individual cages, and given 1 week to adapt to their new diets. Group C was fed 100% feed ad libitum. All the meat-eating groups (CM, FM1, FM2, FM3, FM4, FM5) were given 20 g of 50% adjusted feed and 50% their respective meat product per day. We checked daily to ensure that the majority of the food was being eaten each day to ensure that there were no differences in dietary consumption between the groups. The meat products were well tolerated and even preferred to the feed. These respective diets were maintained during the whole study (20 weeks).
After the dietary adaptation period, the CRC induction began. In week one, 8 rats from each group were assigned to cancer induction and given an intraperitoneal injection of azoxymethane (Merck, Madrid, Spain) dissolved in sterile saline (0.9% NaCl) at a concentration of 2 mg mL−1 (Braun, Barcelona, Spain), corresponding to a dose of 10 mg kg−1 of rat body weight. This injection was repeated in week 2. In week 3, dextran sodium sulphate (DSS) with a molecular weight of 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1 (VWR, Barcelona, Spain) was added to the drinking water at a 3% concentration, ad libitum during that week. This process was repeated in week 13, but the DSS was then administered at 2% concentration. Two rats in each group were used as absolute healthy controls (no CRC induction), receiving injections of sterile saline and receiving no DSS treatment.
000 g mol−1 (VWR, Barcelona, Spain) was added to the drinking water at a 3% concentration, ad libitum during that week. This process was repeated in week 13, but the DSS was then administered at 2% concentration. Two rats in each group were used as absolute healthy controls (no CRC induction), receiving injections of sterile saline and receiving no DSS treatment.
At the end of 20 weeks, the rats were anesthetized with vaporized isoflurane (Zoetis, NJ, USA) and then euthanized via pneumothorax. During euthanasia, blood samples were collected, Peyer's patches in the small intestine were visually quantified, the cecum was removed and weighed fresh, and the colon was removed, opened longitudinally, and fixed in 4% paraformaldehyde (Thermo Scientific, Madrid, Spain). The number of macroscopic tumors in the fixed colons was visually quantified, and the circumference of each tumor was measured with a caliper (Bricody, Madrid, Spain). The tumors were classified as either flat, rounded, peduncular, or fully spherical. The area of each tumor was calculated based on its shape (L × W for flat, πr2 for rounded, 2πr2 for peduncular, and 4πr2 for fully spherical). The area of each tumor was then summed to calculate the total tumoral area for each rat.
2.3. Metataxonomic analysis
200 mg of cecal matter was removed from the center of each cecum to ensure spatial homogeneity, and then, genomic DNA was extracted and purified using the E.Z.N.A.® DNA Stool Kit (ref. D4015-02, VWR, Madrid, Spain). The DNA was quantified using a Qubit fluorometer (Thermo Fisher Scientific, Madrid, Spain). 16S rRNA gene amplification and sequencing was done by Novogene (Shanghai, China). The V3/V4 regions of the 16S rRNA gene were amplified by PCR using the standard 515F-806R primers. The PCR products were purified by magnetic beads and agarose gel electrophoresis. PCR products from each sample were pooled, end-repaired, A-tailed, and further ligated with Illumina adapters. DNA concentrations were normalized by quantitative PCR and the libraries were sequenced on a paired-end Illumina Novaseq platform.After obtaining the raw sequences, the barcodes and primers were removed in silico, and the forward and reverse sequences were merged using FLASH (version 1.2.1).71 Raw sequences without primers and barcodes were uploaded to NCBI SRA under BioProject ID: PRJNA1338236. Merged reads underwent quality control using fastp (version 0.23),72 and high-quality sequences were compared to the SILVA database using V-Search (version 2.16.0)73 to remove known chimeras. Finally, the remaining sequences were trimmed to 400 bp, and amplicon sequence variants (ASVs) were called using DADA2.74
Within the Qiime2 environment (version amplicon-2023.9)75,76 the generated ASVs were then searched against the Greengenes2 2024.09 database77 using the q2-vsearch script73 to generate taxonomy and phylogenetic placements. Alpha diversity was calculated as Shannon entropy78 and beta diversity distances were calculated based on Bray Curtis dissimilarity.79 Beta diversity was visualized using principal coordinate analysis,80 and the emperor visualizer.81 The significance between group distances was calculated by PERMANOVA.82 The significance of alpha diversity differences was tested using the Kruskal–Wallis nonparametric test. For alpha and beta diversity analysis, the data was rarefied to a sampling depth of 38![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 781.
781.
Differential abundance of taxa between groups was calculated using the lowest possible taxonomic assignment for each ASV. Differential taxa were identified using Maaslin2.83 Count data was normalized to relative abundance by total sum scaling (TSS) followed by log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 transformation. Metadata were converted into Z scores and differential abundance was tested using linear models (LM) with a 0.05% false discovery rate (FDR) cutoff, which was calculated with the Benjamini–Hochberg method. Prevalence filter was set up as 10% of rats. Groups were used as fixed effects, and no random effects were considered.
2 transformation. Metadata were converted into Z scores and differential abundance was tested using linear models (LM) with a 0.05% false discovery rate (FDR) cutoff, which was calculated with the Benjamini–Hochberg method. Prevalence filter was set up as 10% of rats. Groups were used as fixed effects, and no random effects were considered.
2.4. Other statistics
All other statistics not described in section 2.3 were conducted in the GraphPad Prism software. Outliers were identified using the ROUT test with a 1% Q-value. Each variable was tested for normality using the Shapiro–Wilk test. Changes in body weight due to the two transient colitis episodes were tested using nonparametric Kruskal–Wallis tests. Differences in final body weight were tested by Brown–Forsythe ANOVA. Tumor numbers and total tumoral areas were tested by ordinary ANOVA. Differences in Payer's patches were tested by ordinary ANOVA. Relative cecal weight was tested by Brown–Forsythe ANOVA. Tukey's test was used for multiple hypothesis correction of parametric tests, while Dunn's test was used for non-parametric tests. Adjusted p-values below 0.05 were considered significant. In all figures, only significant differences are labeled. A summary of all significant differences, with mean differences, p values, and 95% confidence intervals can be found in SI Table S1.3. Results
3.1 Integration of nutraceutical compounds into the functional meat products
An initial assessment was done to assess the stability of the nutraceutical compounds in the functional meat products after cooking. The retention rates for β-carotene, astaxanthin, and DHA were 25%, 68%, and 50% respectively. All the remaining nutraceutical compounds experienced only minor losses below 10%. By adjusting for these losses, the target concentrations were reached for each nutraceutical compound except for β-carotene. Because of its poor retention, the maximum concentration of β-carotene that could be achieved was approximately 0.5 mg kg−1 of functional meat product. The target concentration for each nutraceutical compound is shown in the methods section, while the actual concentrations are shown in Table 1.3.2. Effect of meat food products on colitis and death
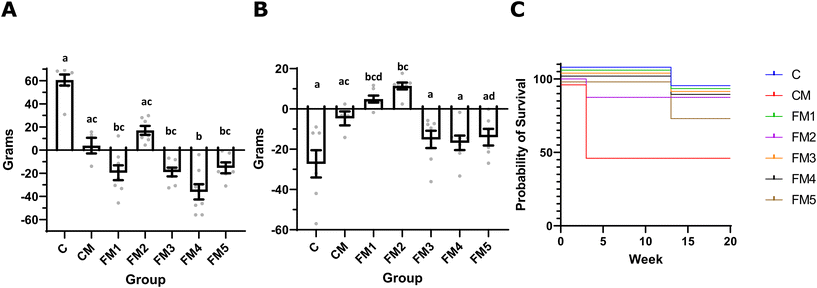
The first parameter analyzed was the changes in body weight due to each colitis period. During the first colitis period (week 3) tumors have yet to develop, but 3% dextran sodium sulfate is sufficient to induce severe colitis symptoms at this timepoint. We observed that group C, and to a lesser extent group FM2, continued to gain weight during this period, while groups FM1, FM3, FM4, and FM5 lost weight (Fig. 1A). Group CM appears to have maintained the same weight, but this result is likely biased. 4 of the 8 rats in group CM developed such severe colitis that they had to be sacrificed before the end of that week. Thus, the results of the weight change for group CM only represent the 4 rats that were least affected. The differences in body weight changes were significantly different when comparing group C and groups FM1, FM3, FM4, and FM5, and when comparing groups FM2 and FM4.In the second colitis period, during week 13, tumors have already begun to develop. Thus, although a lower concentration of dextran sodium sulfate was used (only 2%), symptoms can vary depending on the level of tumor development. During this period, we observed significant weight loss in all the groups except for FM1 and FM2 (Fig. 1B). Significant differences in body weight changes were seen when comparing group FM to groups C, FM3, and FM4, and comparing group FM3 to groups C, FM3, FM4, and FM5.
All the rats that were sacrificed prior to the end of the experiment were sacrificed due to the development of excessively severe colitis symptoms. Group C lost one rat during the second colitis period. Group CM lost 4 rats during the first colitis period. Groups FM1, FM3, and FM4 all lost 1 rat during the second colitis period. Group FM2 lost one rat during the first colitis period, and group FM5 lost 2 rats during the second colitis period (Fig. 1C). Thus, the numbers of rats in each group at the end of the study were C: 7, CM: 4, FM1: 7, FM2: 7, FM3: 7, FM4: 7, FM5: 6.
3.3. Effect on body weight for cancer and control animals
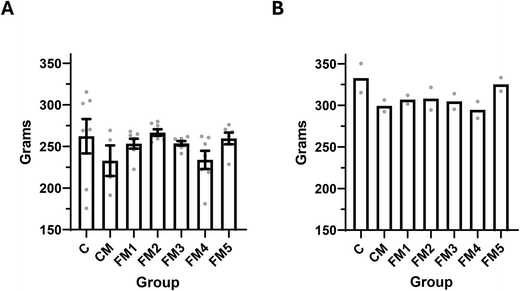
We next compared the body weight of each surviving animal at the end of the experiment, at week 20. We observed no significant differences in body weight between the groups, either for the cancer induced rats (Fig. 2A), or for the healthy control rats (Fig. 2B). There was a general reduction in body weight of more than 50 g, on average for each group, when comparing the cancer-induced rats to their healthy controls. This pattern was the same for all the groups.3.4. Tumor reduction
At the time of euthanasia, we collected the colon of each rat, fixed it, and then quantified both the number of macroscopic tumors as well as the total tumoral area. 100% of the CRC induced rats in all the groups developed colorectal tumors. Comparing the average number of tumors in each group, we see that there are no significant differences between groups C, CM, FM2, FM3, FM4, and FM5. FM1 has fewer tumors than all the other groups, although this reduction only reaches significance when comparing with FM2, FM3, and FM4 (Fig. 3A). As it was expected, the average total tumoral area of each group follows the same trend, with no significant differences between groups C, CM, FM2, FM3, FM4, and FM5. FM1 has less total tumoral area than the other groups, although this reduction only reaches significance when compared with group CM (Fig. 3B).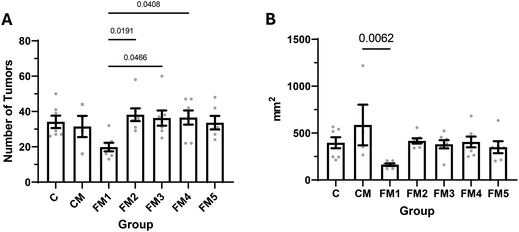 | ||
| Fig. 3 (A) Average number of tumors in each group. (B) Average total tumoral area in each group. Error bars represent standard error. | ||
3.5. Reduction in number of Peyer's patches
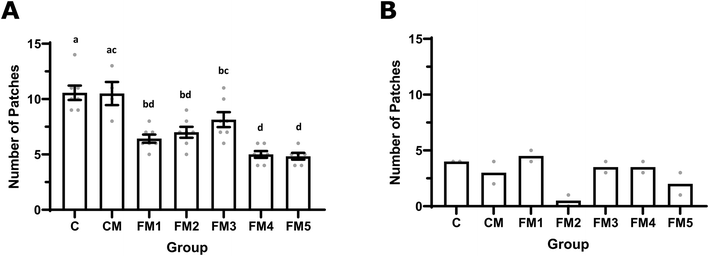
At the time of sacrifice, we extracted the small intestine from each rat and quantified the number of hyperplastic Peyer's patches. In the cancer-induced rats we observed that all the FM groups had fewer Peyer's patches than both C and CM, while no difference was observed between C and CM. The reduction in Peyer's patches reached significance between C and all the FM groups, while the differences between CM and the FM groups only reached significance between CM and FM1, FM2, FM4, and FM5. The lowest numbers of Peyer's patches were achieved at FM4 and FM5, while FM3 had significantly more Peyer's patches than both (Fig. 4A). Each group of healthy rats had on average less than 5 Peyer's patches, less than any of the cancer-induced groups (Fig. 4B).3.6. Effects on cecum weight and gut microbiota
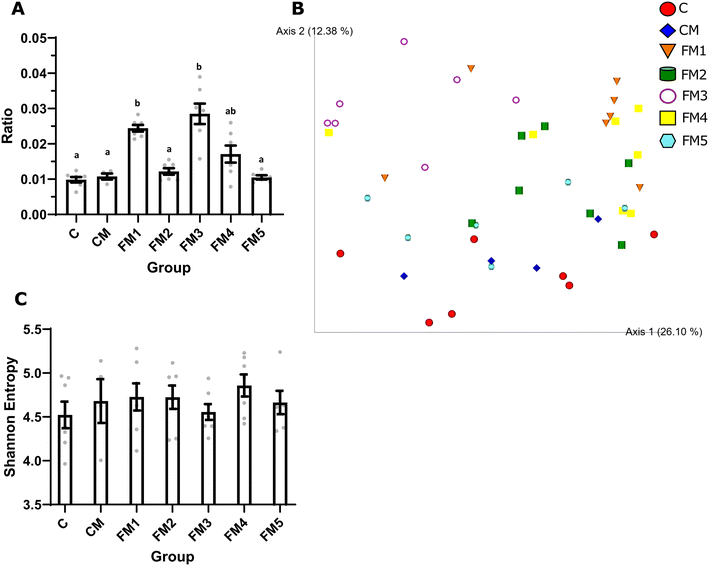
At the time of euthanasia, we recorded the wet weight of each rat's cecum. We observed that groups FM1 and FM3 had on average, heavier cecum than the other groups. This difference was significant compared to C, CM, FM3, and FM5. FM4 had an intermediate cecum weight which was not significantly different from any of the other groups (Fig. 5A).The cecal microbiota composition of each rat was then characterized. An observation of the Bray Curtis dissimilarity between each rat showed that the different diets significantly modulated the cecal microbiota (Fig. 5B). The shift from control feed (C) to feed with control meat (CM) had only a minimal impact, with the distance between the two groups not reaching significance. In contrast, all the FM groups were significantly different from both control groups. FM5 was the most similar FM group to the controls, with a pseudo-F value of 2.11 and 2.09 from C and CM respectively. The next most distant from the controls was FM2, with pseudo-F scores of 2.64 and 2.24 from C and CM, respectively. Next was group FM4, with pseudo-F scores of 2.63 and 2.61 from C and CM, respectively. Next was FM1, with pseudo-F scores of 3.46 and 3.16 from C and CM, respectively. Finally, the most distant group from the controls was group FM3, with pseudo-F scores of 4.44 and 4.46 from C and CM, respectively. Amongst the FM groups, FM1 was significantly different from groups FM3 and FM5, with pseudo F scores of 4.221 and 3.007 respectively, and FM3 was significantly different from all the other FM groups. Thus, FM3 was not only the most distant from the controls, but also highly unique from all the other FM groups. The pseudo-F scores and significance values between each group are shown in Table 2.
| Group 1 | Group 2 | Sample size | Permutations | Pseudo-F | p-Value | q-Value |
|---|---|---|---|---|---|---|
| C | CM | 11 | 999 | 1.149 | 0.261 | 0.274 |
| C | FM1 | 14 | 999 | 3.456 | 0.002 | 0.008 |
| C | FM2 | 14 | 999 | 2.635 | 0.004 | 0.012 |
| C | FM3 | 14 | 999 | 4.439 | 0.001 | 0.008 |
| C | FM4 | 14 | 999 | 2.627 | 0.019 | 0.031 |
| C | FM5 | 13 | 999 | 2.111 | 0.027 | 0.041 |
| CM | FM1 | 11 | 999 | 3.160 | 0.007 | 0.018 |
| CM | FM2 | 11 | 999 | 2.242 | 0.004 | 0.012 |
| CM | FM3 | 11 | 999 | 4.462 | 0.002 | 0.008 |
| CM | FM4 | 11 | 999 | 2.610 | 0.013 | 0.025 |
| CM | FM5 | 10 | 999 | 2.091 | 0.041 | 0.054 |
| FM1 | FM2 | 14 | 999 | 1.340 | 0.198 | 0.223 |
| FM1 | FM3 | 14 | 999 | 4.221 | 0.012 | 0.025 |
| FM1 | FM4 | 14 | 999 | 0.654 | 0.774 | 0.774 |
| FM1 | FM5 | 13 | 999 | 3.007 | 0.016 | 0.028 |
| FM2 | FM3 | 14 | 999 | 5.092 | 0.002 | 0.008 |
| FM2 | FM4 | 14 | 999 | 1.269 | 0.202 | 0.223 |
| FM2 | FM5 | 13 | 999 | 1.352 | 0.173 | 0.214 |
| FM3 | FM4 | 14 | 999 | 4.211 | 0.009 | 0.021 |
| FM3 | FM5 | 13 | 999 | 4.061 | 0.001 | 0.008 |
| FM4 | FM5 | 13 | 999 | 2.158 | 0.038 | 0.053 |
Alpha diversity was measured as Shannon entropy scores, in which a higher score represents greater diversity. Despite the significant differences in cecal weight and the significant shifts in microbiota composition, no significant differences were observed in Shannon entropy (Fig. 5C).
3.7. Effects of each nutraceutical on microbiota composition
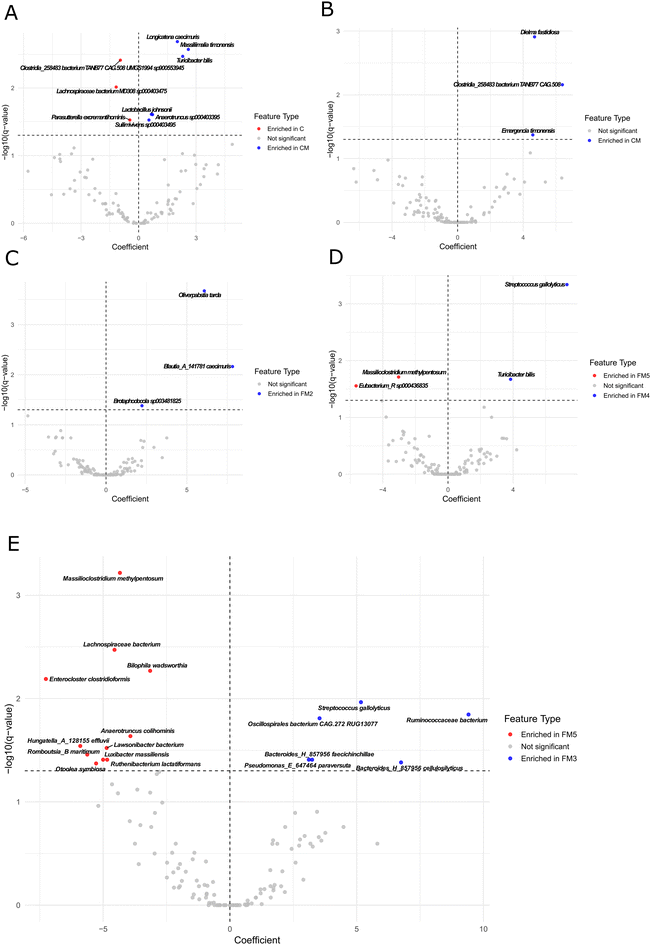
Once establishing that the different functional meats significantly altered the cecal microbiota composition, we next compared the relative abundance of ASVs grouped at the lowest possible taxonomy. The study was started by comparing groups C and CM to identify the impact of meat consumption. In this case, 9 taxa were identified as being significantly different between the two groups with an FDR cutoff of 0.05%. The differential taxa are shown in Fig. 6A, while all the significantly differential taxa with an FDR cutoff of 0.25 can be found in SI Table S2. The most significant changes due to the addition of 50% meat to the diet were decreases in Clostridia_258483 bacterium TANB77 CAG.508 UMGS1994 sp900553945, Lachnospiraceae bacterium MD308 sp000403475, and Parasutterella excrementihominis, as well as increases in the species Longicatena caecimuris, Massiliimalia timonensis, Turicibacter bilis, Lactobacillus johnsonii, Anaerotruncus sp000403395, and Suilimivivens sp000403495.Then, the impact of the PTSO/L. plantarum mixture was studied by comparing the groups CM and FM5. Three taxa were identified as being significantly different between the two groups with an FDR cutoff of 0.05%. The differential taxa are shown in Fig. 6B, while all the significantly differential taxa with an FDR cutoff of 0.25 can be found in SI Table S3. The most significant changes due to the addition of PTSO/L. plantarum included decreases in Dielma fastidiosa, Clostridia_258483 bacterium TANB77 CAG.508, and Emergencia timonensis.
The impact of the addition of the catechin and carnosic acid mixture was studied by comparing the groups FM5 and FM2. Three taxa were identified as being significantly different between the two groups with an FDR cutoff of 0.05%. All the differential taxa are shown in Fig. 6C and all differential taxa with an FDR cutoff of 0.25 are shown in SI Table S4. The significant changes due to the addition of catechins and carnosic acid included increase in Oliverpabstia tarda, Blautia_A_141781 caecimuris, and Brotaphodocola sp003481825.
The impact of the omega-3 fatty acid mixture of ALA and DHA was analyzed by comparing groups FM5 and FM4. Four taxa were identified as being significantly different between the two groups with an FDR cutoff of 0.05%. All the differential taxa are shown in Fig. 6D and all differential taxa with an FDR cutoff of 0.25 are shown in SI Table S5. The significant changes due to the addition of ALA and DHA included increases in Streptococcus gallolyticus and Turicibacter bilis, as well as decreases in Massilioclostridium methylpentosum and Eubacterium_R sp000436835.
Finally, the impact of the carotenoid mixture of β-carotene and astaxanthin was analyzed by comparing the groups FM5 and FM3. Here, 17 taxa were identified as being significantly different between the two groups with an FDR cutoff of 0.05%. The differential taxa in each direction are shown in Fig. 6E, while all the significantly differential taxa with an FDR cutoff of 0.25 can be found in SI Table S6. The most significant changes due to the addition of β-carotene and astaxanthin included increases in Ruminococcaceae bacterium, Streptococcus gallolyticus, Oscillospirales bacterium CAG.272 RUG13077, Bacteroides_H_857956 faecichinchillae, Pseudomonas_E_647464 paraversuta, and Bacteroides_H_857956 cellulosilyticus, as well as decreases in Massilioclostridium methylpentosum, Lachnospiraceae bacterium, Bilophila wadsworthia, Enterocloster clostridioformis, Anaerotruncus colihominis, Hungatella_A_128155 effluvii, Lawsonibacter bacterium, Romboutsia_B maritimum, Luxibacter massiliensis, Ruthenibacterium lactatiformans, and Otoolea symbiosa. The relative abundance of all the significantly different taxa across each group can be seen in Fig. 7.
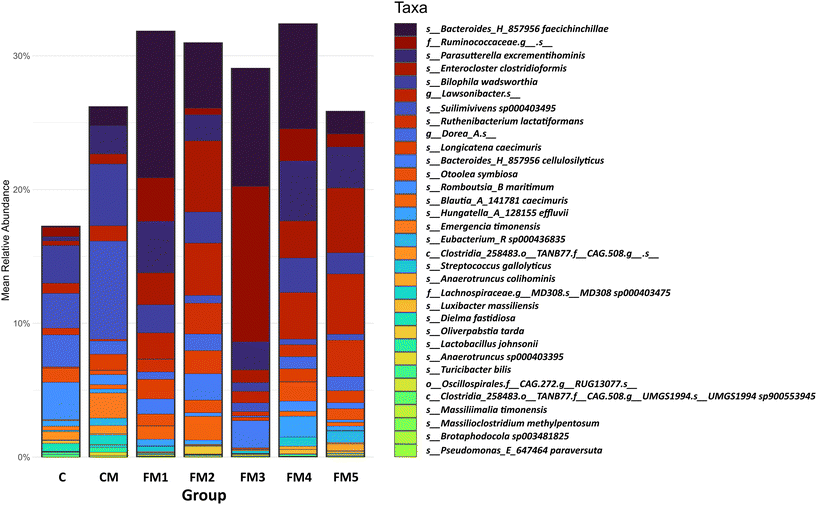 | ||
| Fig. 7 Bar plot displaying average relative abundance of taxa per group. Only taxa that were significantly different in the previously discussed comparisons are displayed. | ||
3.8. Gut microbiota taxons possibly involved in tumor reduction
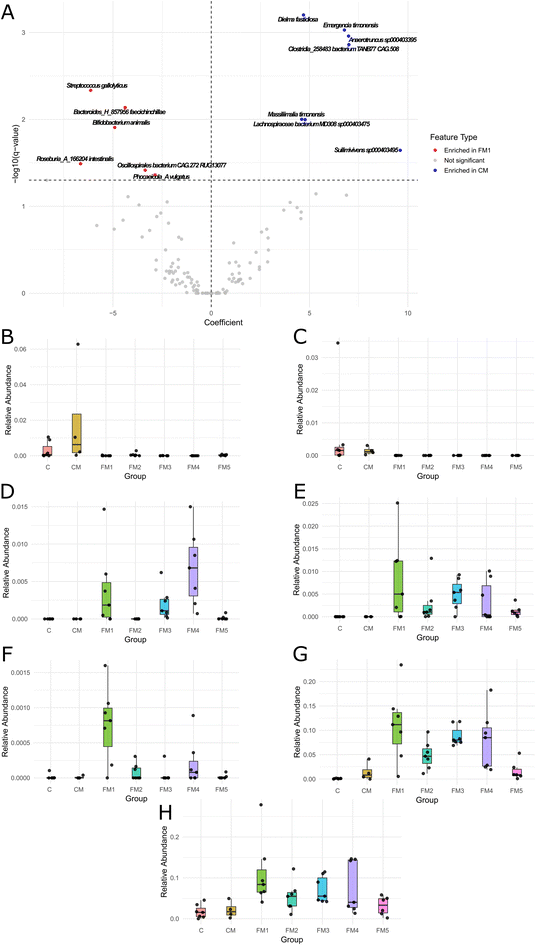
Since group FM1 was the only group to experience a significant reduction in tumor development, and this group had a highly dissimilar gut microbiota composition compared to the C and CM controls, we set out to identify differential taxa that could make FM1 unique. After comparing FM1 to CM, 13 differential taxa were identified between the two groups with an FDR cutoff of 0.05%. The differential taxa are shown in Fig. 8A, while all the significantly differential taxa with an FDR cutoff of 0.25 can be found in SI Table S7. The significant changes due to the addition of the complete nutraceutical mixture included increases in Streptococcus gallolyticus, Bacteroides_H_857956 faecichinchillae, Bifidobacterium animalis, Roseburia_A_166204 intestinalis, Oscillospirales bacterium CAG.272 RUG13077, and Phocaeicola_A vulgatus, as well as decreases in Dielma fastidiosa, Emergencia timonensis, Anaerotruncus sp000403395, Clostridia_258483 bacterium TANB77 CAG.508, Massiliimalia timonensis, Lachnospiraceae bacterium MD308 sp000403475, and Suilimivivens sp000403495.The relative abundances across groups for species that have previously been shown to be involved in physiological processes that could be related to CRC development were tracked. First, the relative abundance of Emergencia timonensis was tracked (Fig. 8B). This species existed at relatively high relative abundance in group and CM and was significantly reduced in groups FM1 and FM5 compared to CM. It was largely absent from groups FM1–FM5, while the statistical differences between group CM and groups FM2, FM3, and FM4 were not assessed. Dielma Fastidiosa (Fig. 8C) was almost exclusively observed in groups C and CM, while being significantly reduced in groups FM1 and FM5 compared to CM. This taxon was also largely absent from groups FM1–FM5, while the statistical differences between group CM and groups FM2, FM3, and FM4 were not assessed. Streptococcus gallolyticus (Fig. 8D) was largely absent from groups C, CM, FM2, and FM5, and was significantly more abundant when comparing group FM1 to CM and group FM4 to FM5. Roseburia intestinalis (Fig. 8E) was practically absent from both groups C and CM, and was significantly more abundant in group FM1 compared to group CM. Bifidobacterium animalis (Fig. 8F), existed in very low abundance in all of the groups except for FM1, and was significantly more abundant in group FM1 compared to group CM. Bacteroides faecichinchillae (Fig. 8G) was significantly more abundant in group FM1 compared to CM, and in group FM3 compared to group FM5. Phocaeicola vulgatus (Fig. 8H) was significantly more abundant in group FM1 compared to group CM only.
4. Discussion
Since a reduction in tumor development was only seen in group FM1, it is clear that the synergistic effects of all the nutraceutical compounds together are required to achieve this reduction. These findings indicate that tumor suppression requires the simultaneous engagement of multiple biological mechanisms, which is only achieved by the complete functional meat formulation. Throughout this study, anti-inflammatory, prebiotic, and microbiota modulating effects are observed. Other mechanisms may also contribute to these effects, although they were not directly assessed in this study.Since processed meat consumption is associated with a greater incidence of CRC, we hypothesized that meat consumption in our rat model would increase tumor development. Our results indicate that this is not the case, but it is possible that our model is so effective in inducing tumor development that we are already at the upper limit of what the rat subjects are able to tolerate. The fact that 50% of the rats in group CM did not make it past the first period of ulcerative colitis indicates that meat consumption not only worsened colitis symptoms but also pushed those symptoms beyond the limit that was intended (inflammation as a triggering factor for tumor development) as part of a CRC development model. Also, since group CM lost the 50% of subjects that were most negatively affected, it is highly likely that the results of this group are biased toward those of healthier rats (the survivors).
Still, our data clearly indicates that meat consumption significantly worsened colitis-associated weight loss during the early period induced with 3% DSS since all the groups except for C and FM2 lost weight (Fig. 1A and B). The second colitis period was less extreme, with only 2% DSS used, but since tumor development has likely already begun by this point, the colitis associated weight loss is likely more representative of early tumor load. During this second colitis period we clearly see that all the groups are affected except for groups FM1 and FM2. FM2 seems to be protected from colitis-associated weight loss during both periods, indicating a protective effect from the catechin/carnosic acid mixture. FM1 is only protected from colitis associated weight loss during the second period, which could be due to a lower tumor load by that point in just these animals. It should be noted that this model is not intended to be an in-depth study into colitis development. A true study into colitis development would require collection of numerous specific parameters and histopathological diagnosis of ulcerative colitis, as has been done in numerous studies.84,85 Still, weight loss during the colitis period may serve as a proxy for colitis symptoms.
Previous studies have found that both green tea (Camellia sinensis) derived catechins86–88 and rosmarin (Salvia rosmarinus) derived carnosic acid89–91 can exhibit significant protective effects against models of ulcerative colitis due to their antioxidant and anti-inflammatory properties. Numerous epidemiological and case control studies have found associations between meat consumption and ulcerative colitis,92–95 while a murine model of DSS induced colitis found that high meat consumption worsened colitis symptoms and impaired intestinal barrier integrity.96 Thus, both the anti-colitis effects of the catechin/carnosic acid mixture, and pro-colitis effects of meat consumption that we observe in this study are supported by the literature.
The fact that the number of Peyer's patches was significantly reduced in all the FM groups indicates that this effect is likely due to the PTSO and L. plantarum mixture, which are the only two nutraceuticals present in all FM animal groups (Fig. 4 and Table 1). PTSO has been shown to have immune modulation activity. Treating Caco-2 colon epithelial adenocarcinoma cells with PTSO significantly reduced the secretion of IL-8, while treating lipopolysaccharide activated THP-1 monocytes with PTSO reduced the secretion of IL-1β, IL-6, and TNF-α.97 IL-8 is known to stimulate the migration of immune cells from circulation to the colon mucosa.98 Similarly, the inhibitory effects upon monocyte cytokine secretion may indicate that PTSO inhibits these cells from being activated by lipopolysaccharides. Furthermore, it has been shown that PTSO supplementation in murine models of metabolic syndrome restores healthy immune cell profiles in adipose and hepatic tissues.99 Studies on the use of inactivated L. plantarum indicate similar results, such as reductions in IL-18 secretion,100 and the inhibition of NF-kB.101 Thus, the literature has identified strong immune modulating properties for both PTSO and L. plantarum postbiotics, supporting our findings.
In addition to the demonstrated directly anti-inflammatory activities of both nutraceuticals, our data also proposes that their observed impacts may be in part due to their Emergencia timonensis-modulating effects. E. timonensis is known to be involved in trimethylamine oxide (TMAO) biosynthesis. Numerous studies have demonstrated the mechanisms by which dietary L-carnitine is converted to γ-butyrobetaine by members of the human gut microbiota, which is then converted to trimethylamine (TMA) by E. timonensis via a unique set of anaerobic enzymes.102–104 TMA from the gut microbiota is then transported to the liver, where it is oxidized to TMAO by flavin-containing monooxygenase 3 (FMO3), a liver cytochrome.105,106 Association studies have shown that E. timonensis is central to predicting blood TMAO concentrations from an oral L-carnitine challenge.107 TMAO is commonly associated with numerous aspects of cardiovascular disease,105 but studies have also identified a role for TMAO in CRC development.108 Mechanistically, TMAO has been proposed to promote tumor cell proliferation and tumor angiogenesis.109L-Carnitine is abundant in meat products and studies have found that the abundance of genes involved in TMA biosynthesis from γ-butyrobetaine are significantly more abundant in people who eat diets rich in red meat.102 Our data indicates that the relative abundance of E. timonensis is less than 1% in group C, rises to almost 2% in group CM, and decreases to less than 0.1% in all the FM groups. Thus, E. timonensis reaches its highest relative abundance in control meat eating rats, in corroboration with these previous studies, while the PTSO/L. plantarum mixture seems to reduce its abundance.
Since groups FM1 and FM3 had significantly enlarged cecum compared to the rest of the groups, we concluded that this was due to the presence of β-carotene and astaxanthin in these groups, as these two groups share the presence of these carotenoids (Fig. 5A and Table 1). Cecum weight is usually associated with greater bacterial biomass or greater crypt depth, and it is thus a marker of cecum health. Numerous studies have demonstrated that dietary supplementation with prebiotic fibers significantly increases cecal weight, intestinal mucus secretion, and bacterial SCFA biosynthesis.69,110–115 Previous studies have shown that dietary supplementation with both β-carotene and astaxanthin modulate the composition of the murine gut microbiome,116–118 with astaxanthin even being shown to promote intestinal mucus secretion. Furthermore, in vitro fermentation studies have shown that the addition of both β-carotene and astaxanthin increases SCFA biosynthesis and shifts the community composition towards greater abundance of the genera Roseburia and Ruminococcus, in line with our results,119 while also increasing alpha diversity and acetic acid biosynthesis.120 Our results indicate that dietary supplementation with the carotenoid mixture yields similar effects to what has been observed in the literature. Thus, we may be able to attribute prebiotic-like effects to these two compounds.
Adding further weight to the prebiotic properties of our carotenoid mixture is the significant shift in cecal microbiota composition that it induced. Of all the nutraceutical combinations that were studied, the addition of our carotenoid mixture caused the largest shift in microbiota composition. This is confirmed by group FM3 having the greatest pseudo-F value from the controls (Fig. 5B and Table 2).
The observation that some taxa, including Roseburia intestinalis and Bifidobacterium animalis, were exclusively enriched in group FM1 indicated that these taxa were enriched by the consumption of the complex mixture of nutraceuticals, rather than being responsive to any single nutraceutical group (Fig. 7A). Furthermore, since this group was the only one to achieve a significant reduction in tumor development (Fig. 3A), the potential impacts of these taxa cannot be ignored. R. intestinalis is a very well characterized butyrate producing bacteria. It has been associated with improved intestinal health and reduced inflammation.121 Association studies have also found it to be significantly reduced in patients with Crohn's disease122 and CRC.123,124 Mechanistic studies have shown that R. intestinalis sensitizes colorectal tumors to radiation stress,125 activates CD8+ T cells, and improves intestinal barrier integrity.126 All these mechanisms are attributed to the butyrate which R. intestinalis produces. B. animalis has been shown to exhibit beneficial effects upon intestinal integrity and inflammation.127 Metabolites from B. animalis have been shown to induce apoptosis in CRC cells by modifying the expression of genes involved in apoptosis regulation128 while rodent models have shown that B. animalis supplementation has a protective effect against oxidative stress.129
One result that seems contradictory is the pattern of abundance of Streptococcus gallolyticus. It is an opportunistic pathogen that is heavily associated with CRC development.130 Studies have demonstrated that it can be a significant member of the gut microbiome of herbivores due to its unusual ability to metabolize tannins.131 Our data indicates that it is enriched in groups FM1 and FM4. This indicates that S. gallolyticus may proliferate due to the addition of our omega-3 fatty acid mixture (Fig. 6D and E). The role of S. gallolyticus in our study and its impact upon the observed tumor development is not abundantly clear.
Although the concentrations of nutraceuticals that were used in this study were under safe consumption limits, we needed to make sure that the mixtures of compounds used in this study did not induce negative effects upon the health of the animals. For this reason, two animals from each group were used as dietary controls with no cancer induction (absolute healthy control animals). All the healthy control rats reached comparable body weights, and none developed tumors, elevated numbers of Peyer's patches, or any other signs of negative health impacts. Thus, we can confirm that the nutraceutical combinations that were used in this study had no observed negative effects on the health of the rodent subjects, nor did they affect the parameters that were measured in this study without the induction of CRC.
It should also be noted that since group FM1 is the only group to contain α-tocopherol and ascorbyl palmitate, the roles of these antioxidant compounds cannot be discounted. α-Tocopherol has been proposed to have antitumoral effects due to its antioxidant properties,132 but clinical trials have not supported this.62 Similarly, studies with ascorbyl palmitate have not identified any protective activity against CRC.61 Thus, it seems unlikely that the tumor reducing effects observed in group FM1 are directly due to the addition of α-tocopherol or ascorbyl palmitate. On the other hand, it is known that omega-3 fatty acids can become oxidized during the cooking process.133 It is possible that the addition of fat-soluble antioxidants, such as α-tocopherol, ascorbyl palmitate, carotenoids, or even catechins, could protect our omega-3 fatty acid mixture from oxidation and improve its antitumoral properties in group FM1.
Taken together, our data supports a multifactorial mechanistic model in which tumor suppression in group FM1 arises from the convergence of microbiota modulation, inflammatory control, and likely other uninvestigated pathways. The PTSO/L. plantarum mixture appears to attenuate mucosal immune activation, potentially through both direct cytokine suppression and reduced abundance of the TMAO-producing bacterium E. timonensis, thereby limiting pro-tumorigenic inflammatory signaling. In parallel, the carotenoid mixture induces prebiotic-like effects, increasing cecal biomass and selectively enriching butyrate-producing taxa such as R. intestinalis, which are known to enhance epithelial barrier integrity and exert anti-inflammatory and anti-tumoral effects. In addition, the catechin/carnosic acid mixture protects against the damage caused by ulcerative colitis. These microbiota-driven changes likely reduce the inflammatory and metabolic microenvironment that supports tumor initiation and progression. Although each nutraceutical mixture exerts measurable biological effects in isolation, only their combined action simultaneously modulates inflammation, microbial composition, and colitis symptoms, resulting in a significant reduction in colorectal tumor burden.
It is important to note that this study was conducted in a murine model of chemically induced colorectal cancer. Precautions should always be taken when extrapolating results from animal models to humans without further investigation. Furthermore, while the method for chemical induction of colitis associated colorectal cancer that was used in this study is well established, the etiology that is induced is highly specific and may not accurately model other diverse types of colorectal cancer. The microbiota associations that are discussed in this study are hypothesis generating, but association cannot confirm causation without further investigation. Finally, the metagenomics analysis that was conducted in this study utilized 16S rRNA gene sequencing, amplicon sequence variant calling, and taxonomic assignment to the lowest confident level. This method is not 100% accurate in calling species level taxonomy, and thus any species level assignments that are discussed in this study should be considered putative.
5. Conclusions
In our animal model, meat consumption did not significantly increase tumor development, but it did worsen colitis-associated weight loss, indicating that it may have increased intestinal inflammation. The addition of PTSO, L. plantarum, β-carotene, astaxanthin, ALA, DHA, catechins, carnosic acid, α-tocopherol, and ascorbyl palmitate protected against this colitis-associated weight loss, reduced inflammation, modified the gut microbiota, and reduced tumor development. The mixtures in which the ingredients were not all used together, showed no reduction in tumor development. Thus, it is likely that the reduction in tumor development comes from synergistic effects amongst the different nutraceuticals, the observed modulation of the gut microbiota, and a potential prevention of omega-3 fatty acids oxidation in the functional meat matrix. Our previous studies have demonstrated the feasibility of functional meat products to retain specific nutraceutical compounds, deliver them to the host, and achieve a health promoting physiological response. This current study confirms this viability with a wide range of nutraceutical compounds and indicates that mixing diverse compounds within the same functional meat product may be a particularly interesting strategy.Conflicts of interest
PLM, JF, CJV, and FL declare no conflict of interest. JB and MG are employed by AGrowingData. LG-M, AF-P, and AB are employed by DMC research center. ECL and FMR-S are employed by BDI-Biotechnology Development for Industry SL. MCR and IUO are employed by FUDIn. MAG and JFDR are employed by Neoalgae. AMA and DGM are employed by BTSA. FS-M and JPC are employed by ARGAL.Data availability
Data for this article, including raw metagenomics sequences are available uploaded to NCBI SRA under BioProject ID: PRJNA1338236.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo04442a.
Acknowledgements
Authors wish to thank CDTI (Centro para el Desarrollo Tecnológico e Industrial, from Ministerio de Ciencia, Innovación y Universidades (MICIU)) for funding this work (grant MIG-20201012), also to the Programa de Ayudas Predoctorales de la Asociación Española Contra el Cáncer (AECC grant PRDAS233973MCAL to P. L. M.), and the research project PID2021-127812OB-I00 from MICIU (Ministerio de Ciencia, Innovación y Universidades).References
- C. P. Wild, E. Weiderpass and B. W. Stewart, World Cancer Report: Cancer research for cancer prevention, International Agency for Research on Cancer, Lyon, 2020 Search PubMed.
- I. Sadien, R. Davies and J. Wheeler, The genomics of sporadic and hereditary colorectal cancer, Ann. R. Coll. Surg. Engl., 2024, 106, 313–320 CrossRef CAS PubMed.
- G. Grosso, F. Bella and J. Godos, et al., Possible role of diet in cancer: systematic review and multiple meta-analyses of dietary patterns, lifestyle factors, and cancer risk, Nutr. Rev., 2017, 75, 405–419 CrossRef PubMed.
- L. Schwingshackl, C. Schwedhelm, C. Galbete and G. Hoffmann, Adherence to Mediterranean Diet and Risk of Cancer: An Updated Systematic Review and Meta-Analysis, Nutrients, 2017, 9, 1063 CrossRef PubMed.
- S. K. Clinton, E. L. Giovannucci and S. D. Hursting, The World Cancer Research Fund/American Institute for Cancer Research Third Expert Report on Diet, Nutrition, Physical Activity, and Cancer: Impact and Future Directions, J. Nutr., 2020, 150, 663–671 Search PubMed.
- V. Bouvard, D. Loomis and K. Z. Guyton, et al., Carcinogenicity of consumption of red and processed meat, Lancet Oncol., 2015, 16, 1599–1600 CrossRef.
- M. S. Deoula, K. El Kinany and I. Huybrechts, et al., Consumption of meat, traditional and modern processed meat and colorectal cancer risk among the Moroccan population: A large–scale case–control study, Int. J. Cancer, 2020, 146, 1333–1345 CrossRef.
- P. Sans and P. Combris, World meat consumption patterns: An overview of the last fifty years (1961–2011), Meat Sci., 2015, 109, 106–111 Search PubMed.
- J. Fernández, E. Ledesma and J. Monte, et al., Traditional Processed Meat Products Re-designed Towards Inulin-rich Functional Foods Reduce Polyps in Two Colorectal Cancer Animal Models, Sci. Rep., 2019, 9, 14783 CrossRef.
- J. Fernández, L. García, J. Monte, C. J. Villar and F. Lombó, Functional Anthocyanin-Rich Sausages Diminish Colorectal Cancer in an Animal Model and Reduce Pro-Inflammatory Bacteria in the Intestinal Microbiota, Genes, 2018, 9, 133 CrossRef PubMed.
- M. Rebersek, Gut microbiome and its role in colorectal cancer, BMC Cancer, 2021, 21, 1325 Search PubMed.
- P. Paone and P. D. Cani, Mucus barrier, mucins and gut microbiota: the expected slimy partners?, Gut, 2020, 69, 2232–2243 CrossRef CAS.
- F. Zhang and S. Qiao, Research Progress on the Relationship Between Inflammation and Colorectal Cancer, Ann. Gastroenterol. Surg., 2022, 6, 204–211 CrossRef PubMed.
- J. Fernández, S. Redondo-Blanco, I. Gutiérrez-del-Río, E. M. Miguélez, C. J. Villar and F. Lombó, Colon microbiota fermentation of dietary prebiotics towards short-chain fatty acids and their roles as anti-inflammatory and antitumour agents: A review, J. Funct. Foods, 2016, 25, 511–522 CrossRef.
- L. Yang, X. Zhang and N. Ma, et al., Effect of sodium caseinate and sodium alginate W/O/W emulsion gel loaded with Aronia melanocarpa proanthocyanidins as fat substitutes on the quality of fermented sausages, Food Chem.: X, 2025, 29, 102680 CAS.
- J. E. Hayes, V. Stepanyan, P. Allen, M. N. O'Grady and J. P. Kerry, Evaluation of the effects of selected plant-derived nutraceuticals on the quality and shelf-life stability of raw and cooked pork sausages, LWT – Food Sci. Technol., 2011, 44, 164–172 CrossRef CAS.
- T. Pintado, I. Muñoz-González, M. Salvador, C. Ruiz-Capillas and A. M. Herrero, Phenolic compounds in emulsion gel-based delivery systems applied as animal fat replacers in frankfurters: Physico-chemical, structural and microbiological approach, Food Chem., 2021, 340, 128095 CrossRef CAS PubMed.
- S. Stajić and D. Vasilev, Encapsulation of Meat Product Ingredients and Influence on Product Quality, in Encapsulation in Food Processing and Fermentation, ed. S. Lević, V. Nedović and B. Bugarski, Taylor & Francis Group, Boca Raton, 1st edn, 2022 Search PubMed.
- E. J. Johnson and R. M. Russell, Beta-Carotene, in Encyclopedia of Dietary Supplements, ed. P. Coates, M. Blackman, G. M. Cragg, M. A. Levine, J. Moss and J. D. White, USDA, NY, 2004, pp. 81–87 Search PubMed.
- R. Ambati, S.-M. Phang, S. Ravi and R. Aswathanarayana, Astaxanthin: Sources, Extraction, Stability, Biological Activities and Its Commercial Applications—A Review, Mar. Drugs, 2014, 12, 128–152 CrossRef.
- K. E. Lee, M. Kwon and Y. S. Kim, et al., β-carotene regulates cancer stemness in colon cancer in vivo and in vitro, Nutr. Res. Pract., 2022, 16, 161 CrossRef CAS PubMed.
- N. Y. Lee, Y. Kim, Y. S. Kim, J.-H. Shin, L. P. Rubin and Y. Kim, β-Carotene exerts anti-colon cancer effects by regulating M2 macrophages and activated fibroblasts, J. Nutr. Biochem., 2020, 82, 108402 CrossRef CAS.
- P. Nagendraprabhu and G. Sudhandiran, Astaxanthin inhibits tumor invasion by decreasing extracellular matrix production and induces apoptosis in experimental rat colon carcinogenesis by modulating the expressions of ERK-2, NFkB and COX-2, Invest. New Drugs, 2011, 29, 207–224 CrossRef CAS PubMed.
- P. N. Prabhu, P. Ashokkumar and G. Sudhandiran, Antioxidative and antiproliferative effects of astaxanthin during the initiation stages of 1,2-dimethyl hydrazine–induced experimental colon carcinogenesis, Fundam. Clin. Pharmacol., 2009, 23, 225–234 CrossRef CAS PubMed.
- H.-Y. Kim, Y.-M. Kim and S. Hong, Astaxanthin suppresses the metastasis of colon cancer by inhibiting the MYC-mediated downregulation of microRNA-29a-3p and microRNA-200a, Sci. Rep., 2019, 9, 9457 CrossRef CAS.
- T. Kochi, M. Shimizu and T. Sumi, et al., Inhibitory effects of astaxanthin on azoxymethane-induced colonic preneoplastic lesions in C57/BL/KsJ-db/dbmice, BMC Gastroenterol., 2014, 14, 212 CrossRef PubMed.
- Y. Yasui, M. Hosokawa, N. Mikami, K. Miyashita and T. Tanaka, Dietary astaxanthin inhibits colitis and colitis-associated colon carcinogenesis in mice via modulation of the inflammatory cytokines, Chem.-Biol. Interact., 2011, 193, 79–87 CrossRef CAS PubMed.
- Q. Yuan, F. Xie and W. Huang, et al., The review of alpha–linolenic acid: Sources, metabolism, and pharmacology, Phytother. Res., 2022, 36, 164–188 CrossRef CAS PubMed.
- F. Echeverría, R. Valenzuela, M. C. Hernandez-Rodas and A. Valenzuela, Docosahexaenoic acid (DHA), a fundamental fatty acid for the brain: New dietary sources, Prostaglandins, Leukotrienes Essent. Fatty Acids, 2017, 124, 1–10 CrossRef PubMed.
- M. Takahashi, Reduction in formation of 2-amino-1-methyl-6-phenylimidazo[4,5- b]pyridine (PhIP)-induced aberrant crypt foci in the rat colon by docosahexaenoic acid (DHA), Carcinogenesis, 1997, 18, 1937–1941 CrossRef CAS PubMed.
- A. Fluckiger, A. Dumont and V. Derangère, et al., Inhibition of colon cancer growth by docosahexaenoic acid involves autocrine production of TNFα, Oncogene, 2016, 35, 4611–4622 CrossRef CAS.
- D. D. Rosa, F. C. Lourenço and A. C. M. da Fonseca, et al., Fish Oil Improves the Lipid Profile and Reduces Inflammatory Cytokines in Wistar Rats With Precancerous Colon Lesions, Nutr. Cancer, 2012, 64, 569–579 CrossRef CAS PubMed.
- C. V. Rao, J. M. R. Patlolla, I. Cooma, T. Kawamori and V. E. Steele, Prevention of Familial Adenomatous Polyp Development in APC min Mice and Azoxymethane-Induced Colon Carcinogenesis in F344 Rats by ω-3 Fatty Acid Rich Perilla Oil, Nutr. Cancer, 2013, 65, 54–60 CrossRef CAS.
- A. J. Cockbain, G. J. Toogood and M. A. Hull, Omega-3 polyunsaturated fatty acids for the treatment and prevention of colorectal cancer, Gut, 2012, 61, 135–149 CrossRef CAS.
- M. J. González-Fernández, I. Ortea and J. L. Guil-Guerrero, α-Linolenic and γ-linolenic acids exercise differential antitumor effects on HT-29 human colorectal cancer cells, Toxicol. Res., 2020, 9, 474–483 CrossRef.
- A. Fluckiger, A. Dumont and V. Derangère, et al., Inhibition of colon cancer growth by docosahexaenoic acid involves autocrine production of TNFα, Oncogene, 2016, 35, 4611–4622 CrossRef CAS PubMed.
- M. R. Sam, M. Tavakoli-Mehr and R. Safaralizadeh, Omega-3 fatty acid DHA modulates p53, survivin, and microRNA-16-1 expression in KRAS-mutant colorectal cancer stem-like cells, Genes Nutr., 2018, 13, 8 CrossRef PubMed.
- P. Magadán-Corpas, Á. Pérez-Valero and S. Ye, et al., Gut Microbiota and Inflammation Modulation in a Rat Model for Ulcerative Colitis after the Intraperitoneal Administration of Apigenin, Luteolin, and Xanthohumol, Int. J. Mol. Sci., 2024, 25, 3236 Search PubMed.
- Á. Pérez-Valero, P. Magadán-Corpas and S. Ye, et al., Antitumor Effect and Gut Microbiota Modulation by Quercetin, Luteolin, and Xanthohumol in a Rat Model for Colorectal Cancer Prevention, Nutrients, 2024, 16, 1161 CrossRef.
- X. Hao, H. Xiao, J. Ju, M.-J. Lee, J. D. Lambert and C. S. Yang, Green Tea Polyphenols Inhibit Colorectal Tumorigenesis in Azoxymethane-Treated F344 Rats, Nutr. Cancer, 2017, 69, 623–631 CrossRef CAS PubMed.
- D. E. Corpet and S. Tache, Most Effective Colon Cancer Chemopreventive Agents in Rats: A Systematic Review of Aberrant Crypt Foci and Tumor Data, Ranked by Potency, Nutr. Cancer, 2002, 43, 1–21 CrossRef CAS PubMed.
- Z. Zhang, S. Zhang and J. Yang, et al., Integrated transcriptomic and metabolomic analyses to characterize the anti-cancer effects of (−)-epigallocatechin-3-gallate in human colon cancer cells, Toxicol. Appl. Pharmacol., 2020, 401, 115100 CrossRef CAS.
- D. Kim, K. Park and I. G. Chae, et al., Carnosic acid inhibits STAT3 signaling and induces apoptosis through generation of ROS in human colon cancer HCT116 cells, Mol. Carcinog., 2016, 55, 1096–1110 CrossRef CAS.
- Y. Kim, J. Kim, Y. R. Seo, J. H. Y. Park, M. Choi and M. Sung, Carnosic acid suppresses colon tumor formation in association with antiadipogenic activity, Mol. Nutr. Food Res., 2014, 58, 2274–2285 CrossRef CAS.
- S. Ding, S. Xu, J. Fang and H. Jiang, The Protective Effect of Polyphenols for Colorectal Cancer, Front. Immunol., 2020, 11, 1407 CrossRef CAS PubMed.
- X. La, L. Zhang, Z. Li, H. Li and Y. Yang, (−)-Epigallocatechin Gallate (EGCG) Enhances the Sensitivity of Colorectal Cancer Cells to 5-FU by Inhibiting GRP78/NF-κB/miR-155-5p/MDR1 Pathway, J. Agric. Food Chem., 2019, 67, 2510–2518 CrossRef CAS PubMed.
- K.-W. Luo, J. Xia, B.-H. Cheng, H.-C. Gao, L.-W. Fu and X.-L. Luo, Tea polyphenol EGCG inhibited colorectal-cancer-cell proliferation and migration via downregulation of STAT3, Gastroenterol. Rep., 2021, 9, 59–70 Search PubMed.
- M. Shimizu, A. Deguchi, Y. Hara, H. Moriwaki and I. B. Weinstein, EGCG inhibits activation of the insulin-like growth factor-1 receptor in human colon cancer cells, Biochem. Biophys. Res. Commun., 2005, 334, 947–953 Search PubMed.
- A. Falcón-Piñeiro, D. García-López and L. Gil-Martínez, et al., PTS and PTSO, two organosulfur compounds from onion by-products as a novel solution for plant disease and pest management, Chem. Biol. Technol. Agric., 2023, 10, 76 Search PubMed.
- P. L. McAlpine, J. Fernández, C. J. Villar and F. Lombó, Organosulfur Compounds in Colorectal Cancer Prevention and Progression, Nutrients, 2024, 16, 802 CrossRef CAS.
- A. C. Lira, A. I. Prieto and A. Baños, et al., Safety assessment of propyl-propane-thiosulfonate (PTSO): 90-days oral subchronic toxicity study in rats, Food Chem. Toxicol., 2020, 144, 111612 Search PubMed.
- L. Zhu, A. I. S. Andersen-Civil and J. L. Castro-Meija, et al., Garlic-Derived Metabolites Exert Antioxidant Activity, Modulate Gut Microbiota Composition and Limit Citrobacter rodentium Infection in Mice, Antioxidants, 2022, 11, 2033 Search PubMed.
- L. Zhu, L. J. Myhill, A. I. S. Andersen-Civil, S. M. Thamsborg, A. Blanchard and A. R. Williams, Garlic–Derived Organosulfur Compounds Regulate Metabolic and Immune Pathways in Macrophages and Attenuate Intestinal Inflammation in Mice, Mol. Nutr. Food Res., 2022, 66, e2101004 Search PubMed.
- T. Vezza, J. Garrido-Mesa and P. Diez-Echave, et al., Allium-Derived Compound Propyl Propane Thiosulfonate (PTSO) Attenuates Metabolic Alterations in Mice Fed a High-Fat Diet through Its Anti-Inflammatory and Prebiotic Properties, Nutrients, 2021, 13, 2595 Search PubMed.
- T. Vezza, F. Algieri and J. Garrido-Mesa, et al., The Immunomodulatory Properties of Propyl–Propane Thiosulfonate Contribute to its Intestinal Anti–Inflammatory Effect in Experimental Colitis, Mol. Nutr. Food Res., 2019, 63 DOI:10.1002/mnfr.201800653.
- E. Guillamón, N. Mut-Salud and M. J. Rodríguez-Sojo, et al., In Vitro Antitumor and Anti-Inflammatory Activities of Allium-Derived Compounds Propyl Propane Thiosulfonate (PTSO) and Propyl Propane Thiosulfinate (PTS), Nutrients, 2023, 15, 1363 CrossRef.
- H. A. Seddik, F. Bendali, F. Gancel, I. Fliss, G. Spano and D. Drider, Lactobacillus plantarum and Its Probiotic and Food Potentialities, Probiotics Antimicrob. Proteins, 2017, 9, 111–122 Search PubMed.
- I. M. Fareez, S. M. Lim and K. Ramasamy, Chemoprevention by Microencapsulated Lactiplantibacillus Plantarum LAB12 Against Orthotopic Colorectal Cancer Mice is Associated with Apoptosis and Anti-angiogenesis, Probiotics Antimicrob. Proteins, 2024, 16, 99–112 CrossRef PubMed.
- M. Sun, W. Liu, Y. Song, Y. Tuo, G. Mu and F. Ma, The Effects of Lactobacillus plantarum-12 Crude Exopolysaccharides on the Cell Proliferation and Apoptosis of Human Colon Cancer (HT-29) Cells, Probiotics Antimicrob. Proteins, 2021, 13, 413–421 CrossRef PubMed.
- Q. Wang, B. Jiang and M. Wei, et al., Antitumor effect of exopolysaccharide from Lactiplantibacillus plantarum WLPL09 on melanoma mice via regulating immunity and gut microbiota, Int. J. Biol. Macromol., 2024, 254, 127624 Search PubMed.
- M.-T. Huang, E. E. Deschner, H. L. Newmark, Z.-Y. Wang, T. A. Ferraro and A. H. Conney, Effect of dietary curcumin and ascorbyl palmitate on azoxymethanol-induced colonic epithelial cell proliferation and focal areas of dysplasia, Cancer Lett., 1992, 64, 117–121 CrossRef PubMed.
- D. Albanes, N. Malila and P. R. Taylor, et al., Effects of supplemental α-tocopherol and β-carotene on colorectal cancer: results from a controlled trial (Finland), Cancer Causes Control, 2000, 11, 197–205 CrossRef PubMed.
- A. Azzi, Molecular mechanism of α-tocopherol action, Free Radicals Biol. Med., 2007, 43, 16–21 CrossRef PubMed.
- M. Imran, B. Titilayo and M. Adil, et al., Ascorbyl palmitate: A comprehensive review on its characteristics, synthesis, encapsulation and applications, Process Biochem., 2024, 142, 68–80 CrossRef.
- H. Kohno, R. Suzuki, R. Noguchi, M. Hosokawa, K. Miyashita and T. Tanaka, Dietary Conjugated Linolenic Acid Inhibits Azoxymethane–induced Colonic Aberrant Crypt Foci in Rats, Jpn. J. Cancer Res., 2002, 93, 133–142 CrossRef PubMed.
- W. L. Stone, A. M. Papas, I. O. LeClair, M. Qui and T. Ponder, The influence of dietary iron and tocopherols on oxidative stress and ras-p21 levels in the colon, Cancer Detect. Prev., 2002, 26, 78–84 Search PubMed.
- S. Khanna, S. Walia, K. K. Kondepudi and G. Shukla, Administration of indigenous probiotics modulate high-fat diet-induced metabolic syndrome in Sprague Dawley rats, Antonie Van Leeuwenhoek, 2020, 113, 1345–1359 Search PubMed.
- J. Fernández, F. J. Moreno, A. Olano, A. Clemente, C. J. Villar and F. Lombó, A Galacto-Oligosaccharides Preparation Derived From Lactulose Protects Against Colorectal Cancer Development in an Animal Model, Front. Microbiol., 2018, 9, 2004 Search PubMed.
- A. Ferreira-Lazarte, J. Fernández and P. Gallego-Lobillo, et al., Behaviour of citrus pectin and modified citrus pectin in an azoxymethane/dextran sodium sulfate (AOM/DSS)-induced rat colorectal carcinogenesis model, Int. J. Biol. Macromol., 2021, 167, 1349–1360 CrossRef PubMed.
- B. Parang, C. W. Barrett and C. S. Williams, AOM/DSS Model of Colitis-Associated Cancer, Methods Mol. Biol., 2016, 1422, 297–307 CrossRef PubMed.
- T. Magoč and S. L. Salzberg, FLASH: fast length adjustment of short reads to improve genome assemblies, Bioinformatics, 2011, 27, 2957–2963 CrossRef PubMed.
- S. Chen, Ultrafast one–pass FASTQ data preprocessing, quality control, and deduplication using fastp, iMeta, 2023, 2, e107 Search PubMed.
- T. Rognes, T. Flouri, B. Nichols, C. Quince and F. Mahé, VSEARCH: a versatile open source tool for metagenomics, PeerJ, 2016, 4, e2584 Search PubMed.
- B. J. Callahan, P. J. McMurdie, M. J. Rosen, A. W. Han, A. J. A. Johnson and S. P. Holmes, DADA2: High-resolution sample inference from Illumina amplicon data, Nat. Methods, 2016, 13, 581–583 CrossRef PubMed.
- E. Bolyen, J. R. Rideout and M. R. Dillon, et al., Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2, Nat. Biotechnol., 2019, 37, 852–857 CrossRef PubMed.
- M. Estaki, L. Jiang and N. A. Bokulich, et al., QIIME 2 Enables Comprehensive End–to–End Analysis of Diverse Microbiome Data and Comparative Studies with Publicly Available Data, Curr. Protoc. Bioinf., 2020, 70, e100 CrossRef PubMed.
- D. McDonald, Y. Jiang and M. Balaban, et al., Greengenes2 unifies microbial data in a single reference tree, Nat. Biotechnol., 2024, 42, 715–718 CrossRef PubMed.
- J. Lin, Divergence measures based on the Shannon entropy, IEEE Trans. Inf. Theory, 1991, 37, 145–151 CrossRef.
- C. Ricotta and J. Podani, On some properties of the Bray-Curtis dissimilarity and their ecological meaning, Ecol. Complex., 2017, 31, 201–205 CrossRef.
- N. Halko, P.-G. Martinsson, Y. Shkolnisky and M. Tygert, An Algorithm for the Principal Component Analysis of Large Data Sets, SIAM J. Sci. Comput., 2011, 33, 2580–2594 CrossRef.
- Y. Vázquez-Baeza, M. Pirrung, A. Gonzalez and R. Knight, EMPeror: a tool for visualizing high-throughput microbial community data, Gigascience, 2013, 2, 16 CrossRef.
- M. J. Anderson, A new method for non–parametric multivariate analysis of variance, Austral. Ecol., 2001, 26, 32–46 Search PubMed.
- H. Mallick, A. Rahnavard and L. J. McIver, et al., Multivariable association discovery in population-scale meta-omics studies, PLoS Comput. Biol., 2021, 17, e1009442 CrossRef PubMed.
- J. Fernández, V. G. de la Fuente and M. T. F. García, et al., A diet based on cured acorn-fed ham with oleic acid content promotes anti-inflammatory gut microbiota and prevents ulcerative colitis in an animal model, Lipids Health Dis., 2020, 19, 28 Search PubMed.
- P. Magadán-Corpas, Á. Pérez-Valero and S. Ye, et al., Gut Microbiota and Inflammation Modulation in a Rat Model for Ulcerative Colitis after the Intraperitoneal Administration of Apigenin, Luteolin, and Xanthohumol, Int. J. Mol. Sci., 2024, 25, 3236 CrossRef PubMed.
- E. Mazzon, C. Muià and R. Di Paola, et al., Green tea polyphenol extract attenuates colon injury induced by experimental colitis, Free Radical Res., 2005, 39, 1017–1025 Search PubMed.
- M. Brückner, S. Westphal, W. Domschke, T. Kucharzik and A. Lügering, Green tea polyphenol epigallocatechin-3-gallate shows therapeutic antioxidative effects in a murine model of colitis, J. Crohns Colitis, 2012, 6, 226–235 CrossRef PubMed.
- S. M. Barbalho, H. Bosso, L. M. Salzedas-Pescinini and R. de Alvares Goulart, Green tea: A possibility in the therapeutic approach of inflammatory bowel diseases?, Complement. Ther. Med., 2019, 43, 148–153 CrossRef PubMed.
- C. Du, Z. Li and J. Zhang, et al., The protective effect of carnosic acid on dextran sulfate sodium-induced colitis based on metabolomics and gut microbiota analysis, Food Sci. Hum. Wellness, 2023, 12, 1212–1223 CrossRef.
- J. P. Veenstra, B. Vemu, R. Tocmo, M. C. Nauman and J. J. Johnson, Pharmacokinetic Analysis of Carnosic Acid and Carnosol in Standardized Rosemary Extract and the Effect on the Disease Activity Index of DSS-Induced Colitis, Nutrients, 2021, 13, 773 CrossRef.
- N. Yang, Z. Xia and N. Shao, et al., Carnosic acid prevents dextran sulfate sodium-induced acute colitis associated with the regulation of the Keap1/Nrf2 pathway, Sci. Rep., 2017, 7, 11036 Search PubMed.
- A. Zinger, E. L. Barnes, L. Weisbein, M. Kappelman and D. Micic, High Red Meat Consumption is Associated With Greater Risk of Ulcerative Colitis Flare, Am. J. Gastroenterol., 2023, 118, S17 CrossRef.
- J. Ge, T.-J. Han and J. Liu, et al., Meat intake and risk of inflammatory bowel disease: A meta-analysis, Turk. J. Gastroenterol., 2020, 26, 492–497 CrossRef PubMed.
- S. L. Jowett, Influence of dietary factors on the clinical course of ulcerative colitis: a prospective cohort study, Gut, 2004, 53, 1479–1484 CrossRef PubMed.
- C. Dong, S. S. M. Chan and P. Jantchou, et al., Meat Intake Is Associated with a Higher Risk of Ulcerative Colitis in a Large European Prospective Cohort Studyø, J. Crohns Colitis, 2022, 16, 1187–1196 CrossRef PubMed.
- D. Li, M. Cui, F. Tan, X. Liu and P. Yao, High Red Meat Intake Exacerbates Dextran Sulfate-Induced Colitis by Altering Gut Microbiota in Mice, Front. Nutr., 2021, 8, 646819 CrossRef PubMed.
- T. Vezza, F. Algieri and J. Garrido-Mesa, et al., The Immunomodulatory Properties of Propyl–Propane Thiosulfonate Contribute to its Intestinal Anti–Inflammatory Effect in Experimental Colitis, Mol. Nutr. Food Res., 2019, 63, e1800653 CrossRef.
- R. Atreya and M. F. Neurath, Chemokines in Inflammatory Bowel Diseases, Dig. Dis., 2010, 28, 386–394 CrossRef PubMed.
- T. Vezza, J. Garrido-Mesa and P. Diez-Echave, et al., Allium-Derived Compound Propyl Propane Thiosulfonate (PTSO) Attenuates Metabolic Alterations in Mice Fed a High-Fat Diet through Its Anti-Inflammatory and Prebiotic Properties, Nutrients, 2021, 13, 2595 CrossRef PubMed.
- A. Magryś and M. Pawlik, Postbiotic Fractions of Probiotics Lactobacillus plantarum 299v and Lactobacillus rhamnosus GG Show Immune-Modulating Effects, Cells, 2023, 12, 2538 CrossRef.
- X. Zhang, Y. Li and C. Zhang, et al., Postbiotics derived from Lactobacillus plantarum 1.0386 ameliorate lipopolysaccharide-induced tight junction injury via MicroRNA-200c-3p mediated activation of the MLCK-MLC pathway in Caco-2 cells, Food Funct., 2022, 13, 11008–11020 Search PubMed.
- J. A. Buffa, K. A. Romano and M. F. Copeland, et al., The microbial gbu gene cluster links cardiovascular disease risk associated with red meat consumption to microbiota l-carnitine catabolism, Nat. Microbiol., 2021, 7, 73–86 CrossRef.
- L. J. Rajakovich, B. Fu, M. Bollenbach and E. P. Balskus, Elucidation of an anaerobic pathway for metabolism of l -carnitine–derived γ-butyrobetaine to trimethylamine in human gut bacteria, Proc. Natl. Acad. Sci. U. S. A., 2021, 118, e2101498118 CrossRef.
- R. A. Koeth, B. R. Lam-Galvez and J. Kirsop, et al., l-Carnitine in omnivorous diets induces an atherogenic gut microbial pathway in humans, J. Clin. Invest., 2018, 129, 373–387 CrossRef PubMed.
- L. Cuervo, P. L. McAlpine, C. Olano, J. Fernández and F. Lombó, Low-Molecular-Weight Compounds Produced by the Intestinal Microbiota and Cardiovascular Disease, Int. J. Mol. Sci., 2024, 25, 10397 CrossRef PubMed.
- B. J. Bennett, T. Q. de Aguiar Vallim and Z. Wang, et al., Trimethylamine-N-Oxide, a Metabolite Associated with Atherosclerosis, Exhibits Complex Genetic and Dietary Regulation, Cell Metab., 2013, 17, 49–60 Search PubMed.
- W.-K. Wu, S. Panyod and P.-Y. Liu, et al., Characterization of TMAO productivity from carnitine challenge facilitates personalized nutrition and microbiome signatures discovery, Microbiome, 2020, 8, 162 CrossRef PubMed.
- R. Xu, Q. Wang and L. Li, A genome-wide systems analysis reveals strong link between colorectal cancer and trimethylamine N-oxide (TMAO), a gut microbial metabolite of dietary meat and fat, BMC Genomics, 2015, 16, S4 CrossRef PubMed.
- S. Yang, H. Dai, Y. Lu, R. Li, C. Gao and S. Pan, Trimethylamine N-Oxide Promotes Cell Proliferation and Angiogenesis in Colorectal Cancer, J. Immunol. Res., 2022, 2022, 1–7 Search PubMed.
- J. Fernández, E. Ledesma and J. Monte, et al., Traditional Processed Meat Products Re-designed Towards Inulin-rich Functional Foods Reduce Polyps in Two Colorectal Cancer Animal Models, Sci. Rep., 2019, 9, 14783 CrossRef PubMed.
- J. Fernández, F. J. Moreno, A. Olano, A. Clemente, C. J. Villar and F. Lombó, A Galacto-Oligosaccharides Preparation Derived From Lactulose Protects Against Colorectal Cancer Development in an Animal Model, Front. Microbiol., 2018, 9, 2004 CrossRef.
- C. Rémésy, S. R. Behr, M.-A. Levrat and C. Demigne, Fiber fermentability in the rat cecum and its physiological consequences, Nutr. Res., 1992, 12, 1235–1244 Search PubMed.
- H. Younes, K. Garleb, S. Behr, C. Rémésy and C. Demigné, Fermentable Fibers or Oligosaccharides Reduce Urinary Nitrogen Excretion by Increasing Urea Disposal in the Rat Cecum, J. Nutr., 1995, 125, 1010–1016 Search PubMed.
- P. Paone, D. Latousakis and R. Terrasi, et al., Human milk oligosaccharide 2′-fucosyllactose protects against high-fat diet-induced obesity by changing intestinal mucus production, composition and degradation linked to changes in gut microbiota and faecal proteome profiles in mice, Gut, 2024, 73, 1632–1649 CrossRef PubMed.
- J. M. Campbell, G. C. Fahey and B. W. Wolf, Selected Indigestible Oligosaccharides Affect Large Bowel Mass, Cecal and Fecal Short-Chain Fatty Acids, pH and Microflora in Rats, J. Nutr., 1997, 127, 130–136 CrossRef PubMed.
- P. Ren, H. Yue, Q. Tang, Y. Wang and C. Xue, Astaxanthin exerts an adjunctive anti-cancer effect through the modulation of gut microbiota and mucosal immunity, Int. Immunopharmacol., 2024, 128, 111553 CrossRef PubMed.
- H. Liu, M. Liu and X. Fu, et al., Astaxanthin Prevents Alcoholic Fatty Liver Disease by Modulating Mouse Gut Microbiota, Nutrients, 2018, 10, 1298 CrossRef.
- L. Zhu, Y. Song and H. Liu, et al., Gut microbiota regulation and anti–inflammatory effect of β–carotene in dextran sulfate sodium–stimulated ulcerative colitis in rats, J. Food Sci., 2021, 86, 2118–2130 Search PubMed.
- Z. Dai, Z. Li and E. Shi, et al., Study on the Interaction between Four Typical Carotenoids and Human Gut Microflora Using an in Vitro Fermentation Model, J. Agric. Food Chem., 2022, 70, 13592–13601 Search PubMed.
- H. R. Rocha, M. E. Pintado, A. M. Gomes and M. C. Coelho, Carotenoids and Intestinal Harmony: Exploring the Link for Health, Foods, 2024, 13, 1599 Search PubMed.
- K. Nie, K. Ma and W. Luo, et al., Roseburia intestinalis: A Beneficial Gut Organism From the Discoveries in Genus and Species, Front. Cell. Infect. Microbiol., 2021, 11, 757718 Search PubMed.
- D. Gevers, S. Kugathasan and L. A. Denson, et al., The Treatment-Naive Microbiome in New-Onset Crohn's Disease, Cell Host Microbe, 2014, 15, 382–392 Search PubMed.
- Q. Liang, J. Chiu and Y. Chen, et al., Fecal Bacteria Act as Novel Biomarkers for Noninvasive Diagnosis of Colorectal Cancer, Clin. Cancer Res., 2017, 23, 2061–2070 CrossRef PubMed.
- T. Wang, G. Cai and Y. Qiu, et al., Structural segregation of gut microbiota between colorectal cancer patients and healthy volunteers, ISME J., 2012, 6, 320–329 CrossRef PubMed.
- J. Dong, B. Wang and Y. Xiao, et al., Roseburia intestinalis sensitizes colorectal cancer to radiotherapy through the butyrate/OR51E1/RALB axis, Cell Rep., 2024, 43, 113846 Search PubMed.
- X. Kang, C. Liu and Y. Ding, et al., Roseburia intestinalis generated butyrate boosts anti-PD-1 efficacy in colorectal cancer by activating cytotoxic CD8 + T cells, Gut, 2023, 72, 2112–2122 Search PubMed.
- J. Cheng, A. Laitila and A. C. Ouwehand, Bifidobacterium animalis subsp. lactis HN019 Effects on Gut Health: A Review, Front. Nutr., 2021, 8, 790561 CrossRef PubMed.
- Z. Faghfoori, M. H. Faghfoori, A. Saber, A. Izadi and A. Yari Khosroushahi, Anticancer effects of bifidobacteria on colon cancer cell lines, Cancer Cell Int., 2021, 21, 258 CrossRef PubMed.
- P. Vitheejongjaroen, A. Kasorn, N. Puttarat, F. Loison and M. Taweechotipatr, Bifidobacterium animalis MSMC83 Improves Oxidative Stress and Gut Microbiota in D-Galactose-Induced Rats, Antioxidants, 2022, 11, 2146 Search PubMed.
- E. Pasquereau-Kotula, M. Martins, L. Aymeric and S. Dramsi, Significance of Streptococcus gallolyticus subsp. gallolyticus Association With Colorectal Cancer, Front. Microbiol., 2018, 9, 614 CrossRef PubMed.
- N. Jiménez, I. Reverón, M. Esteban-Torres, F. López de Felipe, B. de las Rivas and R. Muñoz, Genetic and biochemical approaches towards unravelling the degradation of gallotannins by Streptococcus gallolyticus, Microb. Cell Fact., 2014, 13, 154 CrossRef.
- W. L. Stone, K. Krishnan, S. E. Campbell, M. Qui, S. G. Whaley and H. Yang, Tocopherols and the Treatment of Colon Cancer, Ann. N. Y. Acad. Sci., 2004, 1031, 223–233 CrossRef CAS PubMed.
- A. Ismail, G. Bannenberg, H. B. Rice, E. Schutt and D. MacKay, Oxidation in EPA– and DHA–rich oils: an overview, Lipid Technol., 2016, 28, 55–59 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |