 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Impact of anthocyanin-rich black rice consumption on cognitive function, inflammation and microvascular function in older adults: a crossover intervention trial
Chusana
Mekhora
 *ab,
Daniel J.
Lamport
*ab,
Daniel J.
Lamport
 c and
Jeremy P. E.
Spencer
c and
Jeremy P. E.
Spencer
 *a
*a
aDepartment of Food and Nutritional Sciences, School of Chemistry, Food and Pharmacy, University of Reading, Reading, RG2 6AP, UK. E-mail: j.p.e.spencer@reading.ac.uk; c.mekhora@pgr.reading.ac.uk; chusana.m@ku.th; Tel: +44 (0) 118 378 8724
bInstitute of Food Research and Product Development, Kasetsart University, Bangkok 10900, Thailand
cSchool of Psychology and Clinical Language Sciences, University of Reading, Earley Gate, Reading, Berkshire RG6 6AL, UK
First published on 13th January 2026
Abstract
Typical and atypical declines in cognitive function, as well as increases in chronic, low-grade inflammation and impaired vascular function are all impacted by the ageing process. Flavonoid-rich foods/beverages have been extensively shown to impact human cognition and to modulate immune and/or vascular function, although the cause-and-effect relationship between these factors is unclear. Here, we examine the acute (2 hours) and short-term (8 days) effects of anthocyanin-rich black rice on cognition, inflammation, and vascular function in older adults. Twenty-four older adults (65 ± 7 years) participated in a randomized, single-blind, crossover trial with one-week washout periods. Participants consumed either 210 g of anthocyanin-rich black rice (208 mg of anthocyanins) or the brown rice control (0 mg of anthocyanins) daily for 9 days. Acute effects were assessed 2 hours after consumption on days 1 and 9, and short-term effects were evaluated after completing 8 days of intake. Cognitive performance (RAVLT, digit span, Stroop, and digit symbol substitution), microvascular blood flow, and blood pressure were measured for both acute and short-term interventions, while serum inflammatory biomarkers were assessed for the short-term intervention. Anthocyanins and phenolic acids in rice were identified by using liquid chromatography-mass spectrometry (LC-MS). Data were analyzed using linear mixed models with Bonferroni-corrected comparisons. Eight days of black rice intake significantly improved verbal memory (RAVLT final recall: 12.64 vs. 11.92, p = 0.04; total recall: 52.57 vs. 49.54, p = 0.02) and enhanced digit span backward (change from baseline (CFB) = 0.83, p = 0.03) compared with brown rice. In parallel, black rice significantly reduced interleukin-6 (IL-6) levels (CFB: −0.67, p = 0.03), an effect not seen with the control. Acute black rice consumption attenuated declines in delayed recall (CFB: −1.17, p = 0.09) and recognition (CFB: −0.67, p = 0.19), while significant reductions were observed following brown rice intake. No significant treatment effects were observed for microvascular blood flow or blood pressure. Consumption of anthocyanin-rich black rice for 8 days improved verbal memory and reduced blood IL-6 in older adults. These data suggest for the first time that cognitive benefits induced by anthocyanin-rich black rice may be mediated by anti-inflammatory mechanisms. The clinical trial registry number is NCT06583785 (https://clinicaltrials.gov).
Introduction
Cognitive decline, whether typical as part of normal aging or atypical due to underlying pathological mechanisms, is a defining feature of the aging process.1,2 It significantly contributes to increased morbidity and reduced quality of life, and often leads to the need for long-term supportive care.3 Maintaining cognitive health is essential not only for preserving independence and functional abilities but also for promoting overall well-being and reducing the risk of age-related neurodegenerative diseases.1 Cognitive decline results from a complex interplay of biological, genetic, and environmental factors that collectively contribute to the gradual deterioration of brain function.4 Among the underlying mechanisms, chronic low-grade inflammation and vascular dysfunction are particularly prominent in older adults, associated with neurodegenerative processes and age-related cognitive decline.5–8 Observational studies reported that increased concentrations of pro-inflammatory blood mediators, including interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and C-reactive protein (CRP), have all been associated with diminished cognitive performance in older individuals and patients with neurodegenerative diseases.9,10 Elevated levels of circulating inflammatory cytokines can lead to their crossing of the blood–brain barrier, leading to disrupted neuronal signaling, synaptic dysfunction, and neuronal loss, which contribute to the decline of cognitive function and neurodegenerative disorders.7,11,12 Furthermore, endothelial function, particularly its impact on vasodilatory capacity, plays a vital role in maintaining cognitive performance by ensuring adequate delivery of oxygen and nutrients to brain tissue.13,14 Current evidence indicates that impaired peripheral blood flow, resulting from vascular dysfunction or endothelial impairment, is associated with poorer cognitive performance and an increased risk of neurodegenerative disorders,15 with data suggesting that lower flow-mediated dilation has been linked to declines in executive function and working memory in older adults.16 Therefore, interventions aimed at mitigating inflammation and improving endothelial function may provide a promising approach for preserving cognitive function in aging populations.There is strong clinical evidence that both acute and chronic consumption of flavonoid-rich foods and supplements, such as cocoa, citrus and berries, have a beneficial impact on human cognitive function.17–25 For example, acute consumption of citrus flavanones,19,22 cocoa flavanols17 and berry anthocyanins20 may improve episodic memory and processing speed in older adults. Chronic intake of berry anthocyanins over 5–12 weeks led to the enhancement of working memory and verbal memory in healthy aging individuals and those with dementia.21,23 Furthermore, the evidence for their impact on the immune system, inflammatory status26,27 and vascular function24,28–30 is very well established. However, most research to date has focused on high-cost berry interventions or functional extracts and supplements, which may limit their practicality for widespread application. As such, staple crops such as anthocyanin-rich grains like the black rice (Oryza sativa Linn.) used here are more promising for encouraging higher intake of flavonoids at the population level. In animal models, anthocyanin-rich black rice has been shown to improve spatial memory, reduce hippocampal inflammation, and modulate vascular function.31–33 Clinical studies report that acute intake of pigmented rice varieties, such as black and red rice, significantly reduced inflammatory biomarkers and enhanced antioxidant activity in both healthy and obese individuals.34,35 Furthermore, supplementation of anthocyanin extracts derived from black rice has been shown to attenuate postprandial glycemic responses and improve plasma antioxidants and inflammation in healthy and overweight individuals.36–38 These findings suggest that staple crops like black rice, which are as rich in anthocyanins and phenolic acids as berries,39,40 may offer anti-inflammatory and beneficial vascular outcomes, which may contribute to potential cognitive improvements. The aim of this investigation is two-fold: (1) to investigate the acute and short-term effects of black rice consumption on cognitive performance; and (2) to assess whether modulation of the inflammatory and vascular systems is involved in cognitive improvements. We hypothesize that the consumption of black rice will enhance cognitive performance relative to brown rice, through the potential of anthocyanins to modulate inflammatory status and vasodilatory mechanisms.
Methods
Rice preparation and anthocyanin analysis
Black and brown rice samples were purchased from Orientalmart UK Ltd and Healthy Supplies Ltd. All rice samples were stored away from light in a cool and dry environment during the experiment. Brown rice was selected as a control due to its comparable quantities of carbohydrates, protein, fat, and fibre, but negligible anthocyanin content (Table 1). For the acute study, the rice meal was prepared freshly on the morning of each study visit. 100 g of raw rice was rinsed and subsequently cooked in a rice cooker with 250 ml of water. Rice was cooked for 35 min, with a maximum temperature of 100 °C. The post-cooking yield was approximately 210 g and represented the serving of rice. A portion of cooked rice was served with scrambled egg (2 medium eggs, 1 tablespoon of vegetable oil). The scrambled eggs provided 263 kcal, 14.6 g of protein, 22 g of fat, and 0.8 g of carbohydrate (McCance and Widdowson's Composition of Foods Integrated Dataset 2021). For the short-term intake study, volunteers were provided with individual packages of raw rice (100 g per pack) and a rice cooker to prepare the rice meal at home for the subsequent seven days after the acute study. The rice cooker provided was similar to that used in the acute study to control the cooking method for anthocyanin content. The flavonoids cyanidin-3-glucoside (C3G), cyanidin-3-O-rutinoside (C3R), peonidin-3-O-glucoside (P3G), cyanidin chloride and quercetin, and the related phenolic acids protocatechuic acid and vanillic acid identified in the cooked black and brown rice interventions by LC-MS are presented in Table 1. Details of sample preparation, extraction, and LC-MS conditions, and the complete analytical procedure for the raw and cooked black and brown rice samples are provided in SI1.| Composition | Content (per 210 g of cooked rice) | |
|---|---|---|
| Black rice | Brown rice | |
| Nutrient content was obtained from the nutritional labeling on the product packaging, while flavonoid and phenolic acid contents were analyzed using LC-MS. ND = not detected. For acute study, the black rice intervention meal provided total energy of 623 kcal, 78.1 g of carbohydrate, 22.6 g of protein, and 24.7 g of fat, whereas the brown rice intervention meal provided total energy of 630 kcal, 76.8 g of carbohydrate, 22.1 g of protein, and 25.2 g of fat. | ||
| Nutrients | ||
| Energy (kcal) | 360 | 367 |
| Carbohydrate (g) | 77.3 | 76.0 |
| Protein (g) | 8.0 | 7.5 |
| Fat (g) | 2.7 | 3.2 |
| Fiber (g) | 2.7 | 3.6 |
| Flavonoids | ||
| Cyanidin-3-O-glucoside | 189.31 | ND |
| Cyanidin-3-O-rutinoside | 0.47 | ND |
| Peonidin-3-O-glucoside | 14.60 | ND |
| Cyanidin chloride | 4.10 | ND |
| Total anthocyanins | 208.48 | — |
| Quercetin | 0.75 | ND |
| Phenolic acids | ||
| Protocatechuic acid | 43.57 | ND |
| Vanillic acid | 7.85 | ND |
| Total polyphenols | 260.65 | — |
Participants
Twenty-four subjects (13 females; 11 males) with general healthy status, aged 50–77 years (mean age: 65.0 ± 7.2 years), a body mass index (BMI) between 18.5 and 35.0 kg m−2, and a Mini–Mental State Examination (MMSE) score of ≥26 were recruited from the local communities in Reading, UK. Exclusion criteria were the diagnosis of cardiovascular disease, diabetes, hyperlipidemia, hypertension (blood pressure above 140/90 mmHg), cancer, liver disease, kidney diseases bleeding disorders or blood-related diseases, neurodegenerative diseases (e.g., dementia, Alzheimer's, Parkinson's, or current stroke), psychotic disorders (e.g., schizophrenia, bipolar depression, or eating disorder), or major surgery within the past 6 months. Exclusion criteria also included currently taking anti-inflammatory drugs (e.g., aspirin, warfarin, or ibuprofen) or antibiotics within the past 3 months; having signs of infections or acute inflammation; taking medication to lower blood fats or to stabilize blood glucose or blood pressure; taking hormone replacement therapy; taking high doses (more than 200% of the UK's reference nutrient intakes) of dietary supplements; having a pacemaker; being a heavy smoker (more than 20 cigarettes per day); heavy alcohol drinking (more than 14 units of alcohol a week); substance abuse; allergies; hypersensitivity; or food intolerances. A power calculation was done based on similar studies investigating the acute effect of flavonoid/anthocyanin-rich food intake on cognitive function in middle-aged to older adults using G* power, assuming an average effect size of d = 0.64.19,20,41 To allow for a 10% attribution rate, a total of 24 volunteers were required to achieve a statistical power of 0.8 with an alpha level of 0.05.Study design and procedure
All procedures were subjected to ethical review in accordance with the guidelines established in the Declaration of Helsinki, and all procedures involving human participants received approval from the University of Reading Research Ethics Committee, UREC 23/30. This trial was registered at clinicaltrials.gov, unique identifier: NCT06583785.This study employed a single blind, randomized controlled crossover design with a washout period of at least one week to investigate the acute (2 h) and short-term (8 d) effects of anthocyanin-rich black rice consumption on cognitive performance, inflammation, and microvascular function compared to a brown rice control in older adults. The eligible volunteers were invited to attend one screening and four study visits at the Hugh Sinclair Unit of Human Nutrition in the Department of Food and Nutritional Sciences, University of Reading. Informed consent was acquired, and individuals were randomly assigned to the black or brown rice intervention using computer-generated block randomization, with allocation concealed until assignment. Allocation and randomization were performed by an independent researcher not involved in data collection or analysis. The participants were informed that the study aimed to investigate the effects of two different types of unpolished (black and brown) rice on the measured outcomes as controlling the blinding procedure. To minimize potential practice effects during cognitive assessment, volunteers were required to complete a practice version of the cognitive test battery one week prior to their first study visit. For each study visit, cognitive performance, laser Doppler imaging with iontophoresis (LDI), and blood pressure (BP) were assessed at baseline, 2 h post-acute rice meal consumption, and repeated on day 9 following short-term rice intake. Blood samples were collected at baseline on day 1 and day 9. Volunteers were instructed to fast for 8 h prior to each study visit and to follow a polyphenol-restricted diet for 24 h preceding the initiation of each intervention, in accordance with previous research examining bioavailability, vasodilatory, and cognitive function following acute anthocyanin-rich food interventions.20,42–44 A one-week washout period, together with a 24-hour polyphenol-restricted diet prior to study visits, was considered sufficient to minimize any acutely elevated circulating metabolites that could influence the outcomes.45,46
Upon arrival at the nutrition unit, 15 mL of venous blood was collected, followed by cognitive assessments, and then baseline measurements of BP and LDI were performed. Participants were then provided with 210 g of either cooked black rice (208 mg of anthocyanins) or control cooked brown rice (0 mg of anthocyanins) for breakfast, in a random sequence, with scrambled egg (2 medium eggs and 1 tablespoon of vegetable oil) and one glass of water (200 ml), and instructed to complete the meal within 20 min. Table salt or soy sauce was provided ad libitum. Two hours after meal consumption, cognitive performance was reassessed, followed by measurements of BP and LDI. For the short-term intervention, participants were provided with uncooked rice and a rice cooker for home preparation for the subsequent seven days. They were instructed to consume cooked rice for one meal per day, avoid other anthocyanin-rich foods and maintain their usual physical activity levels throughout the trial. To monitor compliance, participants were also asked to complete a daily checklist confirming adherence to the study protocol. A schematic representation of the study design and procedure is shown in Fig. 1.
Cognitive performance measures
The cognitive assessment batteries were selected based on previous studies in which cognitive domains have been shown to be affected by inflammatory status, including short-term memory, working memory, attention, processing speed and executive function.5,47 The standardized cognitive assessments were conducted at baseline and 2 h post-consumption of the rice meal intervention on days 1 and 9. The computer-based cognitive batteries, including the Rey auditory verbal learning test (RAVLT) for immediate recall, delayed recall and recognition, forward and backward digit span, and Stoop task, were administered via the Gorilla platform, with the exception of the Digit Symbol Substitution Task (DSST), which was conducted using a paper-based format (see SI2 for task description and scoring). New versions of all cognitive tasks were generated and randomized for each visit to further mitigate practice and version effects.Blood sample collection and analysis
The blood inflammatory markers were measured at each study visit with 8 h fasting in advance. 15 ml blood samples were taken via venipuncture by registered nurses and drawn into an SST BD tube. To separate the serum, the blood sample was centrifuged at 3000 rpm and 4 °C for 15 min. The serum was aliquoted into vials, each containing 500 µL and stored in a −80 °C freezer for analysis. High sensitivity C-reactive protein (hs-CRP) was analyzed by using a Daytona clinical chemistry analyzer (Randox, Crumlin, UK). IL-6, TNF-α, IL-1β, intercellular adhesion molecule-1 (ICAM-1), and vascular cell adhesion molecule-1 (VCAM-1) were analyzed by using automated ELISA (Ella™, Bio-Techne, UK).Microvascular blood flow
Laser Doppler Imaging (LDI) was employed to assess microvascular blood flow in response to iontophoresis of acetylcholine (ACh) and sodium nitroprusside (SNP) vasodilators. ACh was used to evaluate endothelium-dependent vasodilation, reflecting the capacity of the endothelium cell to produce nitric oxide (NO), whereas SNP was used to assess endothelium-independent vasodilation, acting directly on vascular smooth muscle via NO release.48 To ensure consistency across repeated test sessions, participants rested in a supine position for 30 min in a temperature-controlled room (22–24 °C) before LDI assessment. Peripheral microvascular function was evaluated using a validated method that measures vasodilatory responses to 1% acetylcholine (ACh) and 1% sodium nitroprusside (SNP).49 For vascular reactivity testing, participants were placed in a semi-recumbent position with the right arm supported. A temperature probe and iontophoresis chambers were applied to the volar surface of the forearm. Freshly prepared solutions of acetylcholine chloride (2.5 ml; 1% w/v in 0.5% w/v NaCl) and sodium nitroprusside (2.5 ml; 1% w/v in 0.5% w/v NaCl) were introduced into the anodal and cathodal chambers, respectively. After baseline skin perfusion was recorded, an incremental current was applied in 5 mA steps (5, 10, 15, and 20 mA), producing a total charge (current × time) of 8000 coulombs over a 20-minute period. Fifteen scans were obtained as the current increased from 0 to 20 mA, followed by five additional scans after current cessation. Skin perfusion, expressed as erythrocyte flux, was measured using a laser Doppler imager (Moor Instruments Ltd, Axminster, Devon, UK). Within- and between-day coefficients of variation (CV) were consistently below 10%, as previously reported.50 The results are presented as incremental area under the curve (iAUC), calculated using the baseline value as the reference point and including only the area above this line, using a method modified from the study protocol developed for the unit by Trevor George under the guidance of William Ferrell's group in Glasgow.51Blood pressure
BP was measured at each study visit using a clinically validated measurement. To maintain consistency between repeat test sessions, the volunteers were rested in a seated position for 5 min before each measurement. BP was recorded at baseline and after 2 h in a seated position, with the cuff placed on the left arm, and with the arm resting on an adjacent desk throughout the measurement.Sensory evaluation
Liking scores for appearance, color, taste/flavor, adhesiveness, and softness, and overall preference were evaluated using a 9-point hedonic scale (1 = dislike extremely, 9 = like extremely) following the completion of each treatment. Additionally, volunteers were asked whether they would be willing to consume black or brown rice instead of white rice.Statistical analysis
All data were analyzed using IBM SPSS statistics version 28 (IBM, NY, USA). A comparable statistical approach was employed for all outcomes, including cognitive assessments, BP, microvascular blood flow and serum inflammatory mediators. For reaction time data, only correct responses were included in mean values. z-Score analysis was applied to detect the outliers in the data; data points with a z-score over 3.29 were excluded prior to statistical analysis.52 A linear mixed-effects model (LMM) with an unstructured covariance matrix for repeated measurements was established to evaluate the effects of treatment, time, and their interaction (treatment × time) on the outcomes for varying correlations between time points. Treatment (black rice or brown rice control), time (baseline, day 1 – 2 h, day 9, and day 9 – 2 h), and treatment × time interaction were entered as fixed factors in the model. Participant ID was included as a random effect to account for within-subject correlations. Estimated marginal means (EMMEANS) were calculated and adjusted for multiple comparisons using the Bonferroni correction. Post hoc comparisons were used to investigate any significant effects of treatment, time and their interactions. The restricted maximum likelihood (REML) method was used for parameter estimation. This approach aligns with an intention-to-treat principle, as it incorporates all randomized participants in the analysis and appropriately handles missing data under the missing-at-random assumption. Therefore, data from all 24 participants were included in the LMM model, including one participant with an incomplete period 2 intervention. The outcomes were analyzed separately for the acute (acute analysis 1; baseline and day 1 – 2 h), short-term (baseline on day 1 and day 9), and acute-on-short-term (acute analysis 2; day 9 and day 9 – 2 h) studies, as described in Fig. 1. Effect size (d) was interpreted as small (d = 0.2), medium (d = 0.5), and large (d > 0.8) according to Cohen's criteria.53 Sensory evaluation outcomes were not measured over time, therefore analysis of these ratings only included a comparison between the two treatment conditions by using paired sample t tests.Results
Participant flowchart and baseline characteristics
A total of 42 individuals were recruited for the study, of whom 18 were excluded and 24 were included and randomly assigned to the intervention (Fig. 2). Among the 24 participants, 22 identified as White (91.7%) and 2 as Asian British (8.3%). Educational attainment included GCSE (8.3%), A level (8.3%), higher education (20.8%), and university degree or equivalent (62.5%). One volunteer dropped out after completing one treatment due to personal reasons unrelated to the trial. The initial study commenced in January 2024 and concluded in October 2024, with no adverse or unintended events reported during this period. Baseline characteristics of the participants are presented in Table 2. The analysis of the Food Frequency Questionnaire (FFQ) for macro- and micronutrient intake before the commencement of the study visit is presented in SI3.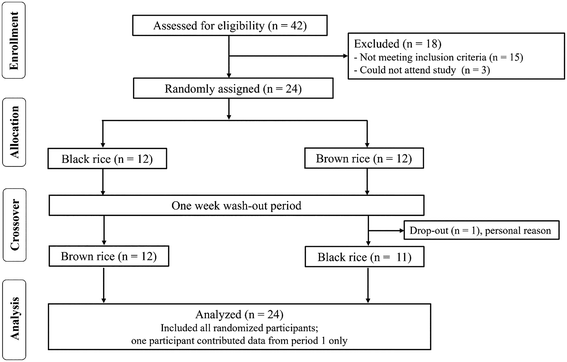 | ||
| Fig. 2 CONSORT flow diagram outlining the number of participants at each stage of a randomized controlled crossover study. | ||
| Characteristics | Mean | SD | Range |
|---|---|---|---|
| Age (year) | 65 | 7 | 50–77 |
| BMI (kg m−2) | 24.6 | 3.9 | 19–34 |
| MMSE score (30) | 29.5 | 0.9 | 27–30 |
| Systolic BP (mmHg) | 123 | 9 | 103–140 |
| Diastolic BP (mmHg) | 80 | 6 | 64–90 |
| Pulse rate (bpm) | 72 | 9 | 57–91 |
| Waist/hip circumference ratio | 0.9 | 0.1 | 0.8–1.0 |
| % Body fat | 27.4 | 10.1 | 7.1–49.2 |
| Vegetable consumption (portions per day) | 2.7 | 1.2 | 1–6 |
| Fruit consumption (portions per day) | 2.0 | 0.9 | 1–5 |
Short-term analysis (8 days)
Consumption of black rice for 8 days led to significant improvements in final recall (12.64 vs. 11.92; F(1, 22) = 4.66, p = 0.04) and total recall (52.57 vs. 49.54; F(1, 24) = 5.97, p = 0.02), in comparison with the brown rice control (Fig. 3a and b). In addition, black rice intake significantly increased backward digit span from baseline (CFB: 0.83, p = 0.03, 95% CI: 0.08, 1.57), while no change was observed for the brown rice control (CFB: 0.0, p = 1.0, 95% CI: −0.73, 0.73). For DSST scores, a measure of processing speed, a significant increase from baseline was observed after consumption of both black rice and the brown rice control (CFB: 8.74, p ≤ 0.001, 95% CI: 5.75, 11.73 vs. 4.92, p = 0.002, 95% CI: 1.99, 7.85), with no significant differences between groups.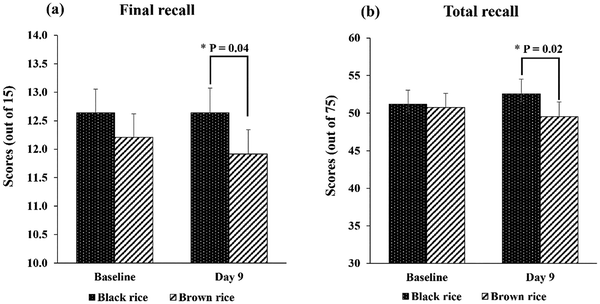 | ||
| Fig. 3 The scores of final recall (a) and total recall (b) improved after 8 days of black rice consumption compared to the brown rice control. | ||
In parallel, black rice intake led to a significant reduction in IL-6 levels from baseline (CFB −0.67, p = 0.03, 95% CI: −1.26, −0.08), a change not observed with brown rice (CFB 0.27, p = 0.36, 95% CI: −0.32, 0.87) (Fig. 4). No significant changes were observed for hsCRP, TNF-α, ICAM-1, and VCAM-1; however, a marginal change from baseline was noted for hsCRP following black rice intake (CFB: −0.33, p = 0.051, 95% CI: −0.65, 0.002). Furthermore, no significant treatment impact was seen for microvascular blood flow and blood pressure following the short-term intervention (summary data from the LMM analysis are provided in Table 3).
 | ||
| Fig. 4 Change from baseline in serum IL-6 concentration after 8 days of black rice and brown rice intake. | ||
| Black rice | Brown rice control | LMM fixed effects F, [p-values] | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline | Day 9 | Baseline | Day 9 | Treatment | Time | Treatment × time | |||||
| Mean | SE | Mean | SE | Mean | SE | Mean | SE | ||||
| Estimates from LMM with mean (estimated marginal mean) ± SE for cognitive, inflammatory, and microvascular outcomes across 2 time points. * = P < 0.05. ND = non-detected, as the concentrations in most two-fold diluted samples were below the assay's limit of detection (0.064 pg ml−1). | |||||||||||
| Cognitive assessments | |||||||||||
| Immediate recall (out of 15) | 7.00 | 0.39 | 6.92 | 0.38 | 6.83 | 0.39 | 6.50 | 0.38 | 2.12, [0.16] | 0.83, [0.37] | 0.28, [0.60] |
| Final recall (out of 15) | 12.64 | 0.41 | 12.64 | 0.43 | 12.21 | 0.41 | 11.92 | 0.43 | 6.50, [0.02]* | 0.45, [0.51] | 0.45, [0.51] |
| Total recall (out of 75) | 51.18 | 1.88 | 52.57 | 1.95 | 50.75 | 1.87 | 49.54 | 1.94 | 5.29, [0.03]* | 0.01, [0.91] | 2.44, [0.13] |
| Learning | 5.62 | 0.41 | 5.71 | 0.38 | 5.38 | 0.40 | 5.42 | 0.37 | 0.95, [0.34] | 0.06, [0.81] | 0.01, [0.93] |
| Proactive interference | 1.38 | 0.35 | 0.72 | 0.31 | 1.50 | 0.34 | 0.54 | 0.31 | 0.02, [0.89] | 7.36, [0.01]* | 0.27, [0.61] |
| Retroactive interference | 1.31 | 0.32 | 1.66 | 0.39 | 1.13 | 0.31 | 0.92 | 0.38 | 4.62, [0.04]* | 0.05, [0.83] | 0.79, [0.38] |
| Delayed recall | 10.86 | 0.62 | 10.86 | 0.65 | 11.13 | 0.61 | 10.79 | 0.65 | 0.12, [0.73] | 0.22, [0.64] | 0.22, [0.64] |
| Recognition (out of 50) | 47.52 | 0.66 | 47.44 | 0.59 | 47.42 | 0.65 | 47.38 | 0.59 | 0.09, [0.77] | 0.02, [0.88] | 0.00, [0.96] |
| Digit span forward | 5.99 | 0.25 | 6.34 | 0.28 | 5.83 | 0.24 | 6.21 | 0.27 | 0.84, [0.37] | 4.86, [0.03]* | 0.01, [0.93] |
| Digit span backward | 4.53 | 0.29 | 5.36 | 0.28 | 5.13 | 0.28 | 5.13 | 0.27 | 0.70, [0.41] | 2.55, [0.12] | 2.55, [0.12] |
| Stroop effect_reaction time (ms) | 179.41 | 73.36 | 116.12 | 73.06 | 91.22 | 73.20 | 118.67 | 72.28 | 0.80, [0.38] | 0.22, [0.64] | 1.41, [0.24] |
| DSST | 52.23 | 2.63 | 60.97 | 2.27 | 55.42 | 2.60 | 60.33 | 2.25 | 0.55, [0.47] | 43.11, [<0.01]* | 3.38, [0.73] |
| Inflammatory markers | |||||||||||
| hsCRP (mg dL−1) | 1.84 | 0.39 | 1.51 | 0.43 | 1.44 | 0.38 | 1.65 | 0.42 | 0.11, [0.74] | 0.27, [0.61] | 5.82, [0.02]* |
| IL-6 (pg mL−1) | 3.05 | 0.35 | 2.39 | 0.26 | 2.02 | 0.35 | 2.29 | 0.26 | 3.45, [0.08] | 0.89, [0.35] | 5.06, [0.03]* |
| TNF-α (pg mL−1) | 10.44 | 0.44 | 10.37 | 0.44 | 10.17 | 0.44 | 10.23 | 0.44 | 0.63, [0.44] | 0.002, [0.97] | 0.31, [0.58] |
| IL-1β (pg mL−1) | ND | — | — | — | |||||||
| ICAM-1 (ng mL−1) | 377.49 | 14.86 | 373.03 | 18.34 | 358.48 | 14.60 | 366.67 | 18.03 | 0.65, [0.43] | 0.07, [0.80] | 0.79, [0.38] |
| VCAM-1 (ng mL−1) | 655.80 | 28.88 | 653.64 | 27.29 | 656.07 | 28.65 | 657.16 | 26.85 | 0.01, [0.94] | 0.002, [0.97] | 0.01, [0.91] |
| Microvascular blood flow | |||||||||||
| Ach_iACU | 195.76 | 46.42 | 136.47 | 31.27 | 170.96 | 46.37 | 94.26 | 32.60 | 0.66, [0.43] | 3.70, [0.06] | 0.06, [0.81] |
| SNP_iAUC | 389.94 | 82.97 | 276.12 | 84.25 | 312.36 | 79.82 | 257.31 | 84.21 | 0.45, [0.51] | 1.36, [0.25] | 0.16, [0.69] |
| Blood pressure | |||||||||||
| Systolic | 123.61 | 2.39 | 121.22 | 2.28 | 123.38 | 2.37 | 121.38 | 2.28 | 0.00, [0.96] | 6.80, [0.01]* | 0.05, [0.82] |
| Diastolic | 70.11 | 1.35 | 68.76 | 1.38 | 68.96 | 1.34 | 68.17 | 1.37 | 1.97, [0.17] | 2.98, [0.09] | 0.20, [0.66] |
Acute analysis 1 (day 1)
Following both acute rice intakes, there was a trend toward lower scores in RAVLT, a verbal memory measure, including immediate recall, total recall, proactive interference, delayed recall and recognition. However, black rice consumption attenuated the decline in delayed recall from baseline (CFB: −1.17, p = 0.09, 95% CI: −2.54, 0.19), whereas a significant reduction was observed for brown rice (CFB: −1.42, p = 0.04, 95% CI: −2.75, −0.08) (Fig. 5a). A significant increase from baseline in DSST scores was observed after consuming both black rice and the brown rice control (CFB: 5.44, p ≤ 0.001, 95% CI: 3.47, 7.40 vs. 2.38, p = 0.02, 95% CI: 0.45, 4.30). The digit span backward and Stroop effect showed a tendency to improve following black rice consumption; however, the results were not statistically significant. | ||
| Fig. 5 Change from baseline in delayed recall (a) and recognition (b) following acute consumption of black rice and the brown rice control. | ||
Meanwhile, a reduction in microvascular blood flow was observed after consuming both types of rice. A significant decrease in SNP, a measure of vascular smooth muscle responsiveness to NO, from baseline was observed for both black and brown rice treatments (CFB: −221.45, p = 0.01, 95% CI: −391.92, −50.98 vs. −196.01, p = 0.02, 95% CI: −359.77, −32.25). In contrast, a significant decrease in Ach, a measure of endothelial dependent NO-producing capacity, was observed only for black rice (CFB: −97.15, p = 0.04, 95% CI: −190.49, −3.81). For blood pressure, a significant reduction from baseline following black rice and brown rice intake was observed in systolic pressure (CFB: −3.91, p = 0.02, 95% CI: −7.07, −0.75 vs. −5.41, p ≤ 0.001, 95% CI: −8.51, −2.32) and diastolic pressure (CFB: −5.17, p < 0.01, 95% CI: −8.45, −1.90 vs. −3.91, p = 0.02, 95% CI: −7.13, −0.71). However, no significant differences between treatments for cognitive function, microvascular blood flow and blood pressure were observed during day 1 of acute rice consumption (summary data from the LMM analysis are provided in Table 4).
| Black rice | Brown rice control | LMM fixed effects F, [p-values] | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline | 2 h | Baseline | 2 h | Treatment | Time | Treatment × time | |||||
| Mean | SE | Mean | SE | Mean | SE | Mean | SE | ||||
| Estimates from LMM with mean (estimated marginal mean) ± SE for cognitive, inflammatory, and microvascular outcomes across 2 time points. * = P < 0.05. | |||||||||||
| Cognitive assessments | |||||||||||
| Immediate recall (out of 15) | 6.98 | 0.42 | 6.11 | 0.42 | 6.83 | 0.41 | 5.79 | 0.41 | 0.99, [0.33] | 14.24, [<0.01]* | 0.12, [0.74] |
| Final recall (out of 15) | 12.63 | 0.39 | 11.89 | 0.47 | 12.21 | 0.38 | 11.83 | 0.46 | 0.64, [0.43] | 3.56, [0.07] | 0.38, [0.54] |
| Total recall (out of 75) | 51.10 | 1.90 | 47.45 | 2.17 | 50.75 | 1.89 | 47.50 | 2.15 | 0.03, [0.87] | 8.84, [0.01]* | 0.03, [0.86] |
| Learning | 5.59 | 0.39 | 5.72 | 0.46 | 5.38 | 0.39 | 6.04 | 0.45 | 0.02, [0.89] | 1.01, [0.32] | 0.46, [0.50] |
| Proactive interference | 1.35 | 0.34 | −0.17 | 0.47 | 1.50 | 0.33 | −0.67 | 0.46 | 0.26, [0.62] | 17.24, [<0.01]* | 0.53, [0.47] |
| Retroactive interference | 1.32 | 0.31 | 2.54 | 0.54 | 1.13 | 0.31 | 2.50 | 0.53 | 0.09, [0.76] | 12.02, [<0.01]* | 0.04, [0.83] |
| Delayed recall | 10.83 | 0.61 | 9.65 | 0.77 | 11.12 | 0.60 | 9.71 | 0.76 | 0.24, [0.63] | 7.50, [0.01]* | 0.07, [0.80] |
| Recognition (out of 50) | 47.49 | 0.70 | 46.27 | 0.65 | 47.42 | 0.69 | 45.87 | 0.64 | 0.26, [0.62] | 11.79, [<0.01]* | 0.17, [0.68] |
| Digit span forward | 5.94 | 0.23 | 5.94 | 0.22 | 5.83 | 0.23 | 6.00 | 0.22 | 0.02, [0.88] | 0.32, [0.57] | 0.32, [0.57] |
| Digit span backward | 4.53 | 0.30 | 5.05 | 0.29 | 5.13 | 0.29 | 4.96 | 0.28 | 1.16, [0.29] | 0.72, [0.40] | 2.70, [0.11] |
| Stroop effect_ reaction time (ms) | 183.17 | 81.49 | 117.64 | 81.96 | 88.64 | 81.42 | 165.34 | 81.40 | 0.34, [0.57] | 0.02, [0.88] | 3.73, [0.06] |
| DSST | 52.14 | 2.47 | 57.57 | 2.28 | 55.42 | 2.43 | 57.79 | 2.25 | 0.65, [0.43] | 32.78, [<0.01]* | 5.03, [0.03]* |
| Microvascular blood flow | |||||||||||
| Ach_iACU | 196.41 | 47.37 | 99.25 | 34.60 | 181.97 | 47.22 | 116.92 | 34.48 | 0.001, [0.97] | 6.11, [0.02]* | 0.24, [0.63] |
| SNP_iAUC | 393.39 | 86.11 | 171.94 | 32.81 | 312.36 | 82.57 | 116.35 | 32.78 | 1.12, [0.30] | 12.66, [<0.01]* | 0.05, [0.83] |
| Blood pressure | |||||||||||
| Systolic | 123.79 | 2.31 | 119.87 | 2.71 | 123.37 | 2.29 | 117.96 | 2.68 | 0.50, [0.49] | 18.04, [<0.01]* | 0.47, [0.50] |
| Diastolic | 70.21 | 1.30 | 65.04 | 1.93 | 68.96 | 1.29 | 65.04 | 1.90 | 0.26, [0.61] | 15.93, [<0.01]* | 0.31, [0.58] |
Acute analysis 2 (day 9)
Acute black rice consumption, following 8 days of daily intake, attenuated the decline in word recognition from baseline (CFB: −0.67, p = 0.19, 95% CI: −1.69, 0.35), whereas a significant decrease was observed for brown rice (CFB: −1.30, p = 0.01, 95% CI: −2.27, −0.31) (Fig. 5b). A significant increase in DSST scores was seen for the brown rice control (CFB: 3.50, p ≤ 0.001, 95% CI: 1.90, 5.10).Concurrently, a significant reduction from baseline following black rice and brown rice intake was found in systolic pressure (CFB: −4.13, p = 0.01, 95% CI: −6.97, −1.29 vs. −3.46, p = 0.02, 95% CI: −6.24, −0.68) and diastolic pressure (CFB: −6.61, p ≤ 0.001, 95% CI: −9.44, −3.78 vs. −4.00, p = 0.006, 95% CI: −6.78, −1.22). In addition, a non-significant reduction in microvascular blood flow in response to ACh and SNP was observed after both rice treatments. No significant differences between treatments were detected for any outcomes during this period (summary data from the LMM analysis are provided in Table 5).
| Black rice | Brown rice control | LMM fixed effects F, [p-values] | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Day 9 | Day 9 – 2 h | Day 9 | Day 9 – 2 h | Treatment | Time | Treatment × time | |||||
| Mean | SE | Mean | SE | Mean | SE | Mean | SE | ||||
| Estimates from LMM with mean (estimated marginal mean) ± SE for cognitive, inflammatory, and microvascular outcomes across 2 time points. * = P < 0.05. | |||||||||||
| Cognitive assessments | |||||||||||
| Immediate recall (out of 15) | 6.87 | 0.39 | 6.39 | 0.40 | 6.50 | 0.38 | 6.54 | 0.40 | 0.31, [0.58] | 0.76, [0.39] | 1.08, [0.31] |
| Final recall (out of 15) | 12.64 | 0.42 | 12.59 | 0.59 | 11.92 | 0.42 | 12.21 | 0.58 | 2.20, [0.15] | 0.11, [0.74] | 0.21, [0.65] |
| Total recall (out of 75) | 52.56 | 1.80 | 49.60 | 1.91 | 49.54 | 1.79 | 48.75 | 1.89 | 4.07, [0.06] | 2.97, [0.09] | 0.99, [0.33] |
| Learning | 5.71 | 0.37 | 6.14 | 0.57 | 5.42 | 0.37 | 5.67 | 0.56 | 0.89, [0.35] | 0.77, [0.39] | 0.06, [0.81] |
| Proactive interference | 0.69 | 0.32 | 0.69 | 0.38 | 0.54 | 0.31 | 0.46 | 0.37 | 0.61, [0.44] | 0.02, [0.90] | 0.02, [0.90] |
| Retroactive interference | 1.64 | 0.39 | 3.12 | 0.71 | 0.92 | 0.38 | 3.13 | 0.70 | 0.50, [0.49] | 13.93, [<0.01]* | 0.55, [0.46] |
| Delayed recall | 10.89 | 0.64 | 9.71 | 0.68 | 10.79 | 0.63 | 9.29 | 0.67 | 0.51, [0.48] | 15.28, [<0.01]* | 0.23, [0.64] |
| Recognition (out of 50) | 47.40 | 0.59 | 46.73 | 0.60 | 47.37 | 0.59 | 46.08 | 0.58 | 0.78, [0.39] | 7.80, [0.01]* | 0.79, [0.38] |
| Digit span forward | 6.31 | 0.27 | 6.53 | 0.25 | 6.21 | 0.26 | 6.33 | 0.24 | 1.02, [0.33] | 0.88, [0.35] | 0.06, [0.80] |
| Digit span backward | 5.36 | 0.29 | 5.27 | 0.31 | 5.12 | 0.28 | 4.75 | 0.30 | 1.84, [0.19] | 1.62, [0.21] | 0.63, [0.43] |
| Stroop effect_ reaction time (ms) | 119.67 | 62.75 | 140.50 | 59.59 | 118.67 | 61.61 | 110.70 | 58.52 | 0.07, [0.80] | 0.03, [0.87] | 0.13, [0.72] |
| DSST | 60.89 | 2.24 | 62.11 | 2.23 | 60.33 | 2.22 | 63.83 | 2.21 | 0.17, [0.68] | 17.25, [<0.01]* | 4.04, [0.05] |
| Microvascular blood flow | |||||||||||
| Ach_iACU | 135.97 | 31.87 | 90.64 | 28.15 | 81.48 | 33.17 | 58.95 | 28.69 | 2.90, [0.11] | 1.57, [0.22] | 0.18, [0.68] |
| SNP_iAUC | 280.98 | 77.48 | 142.32 | 41.65 | 245.14 | 77.47 | 131.51 | 41.65 | 0.14, [0.72] | 4.31, [0.04]* | 0.04, [0.84] |
| Blood pressure | |||||||||||
| Systolic | 121.25 | 2.29 | 117.12 | 2.49 | 121.38 | 2.28 | 117.92 | 2.48 | 0.20, [0.66] | 14.76, [<0.01]* | 0.12, [0.74] |
| Diastolic | 68.67 | 1.42 | 62.06 | 1.77 | 68.17 | 1.41 | 64.17 | 1.74 | 0.60, [0.45] | 29.02, [<0.01]* | 1.76, [0.19] |
Sensory analysis
The mean liking scores for all attributes of cooked black and brown rice characteristics are presented in Table 6; these results indicate moderate to extreme preference for both types of rice. No significant differences in liking scores were seen for all attributes except for softness, where volunteers indicated a significant preference for black rice over brown rice (p < 0.001). In terms of acceptability, 69% of volunteers were willing to consume black rice and 76% were willing to consume brown rice, instead of white rice.| Attributes | Black rice | Brown rice |
|---|---|---|
| Mean and standard deviation values in the same row with different superscripts differ significantly (p < 0.05). | ||
| Appearance | 7.26 ± 1.42a | 7.09 ± 1.28a |
| Color | 7.04 ± 1.74a | 6.96 ± 1.22a |
| Taste | 7.83 ± 0.78a | 7.39 ± 1.23a |
| Adhesiveness | 7.13 ± 1.46a | 6.83 ± 1.37a |
| Softness | 7.70 ± 0.97a | 6.43 ± 1.62b |
| Overall | 7.62 ± 0.93a | 7.22 ± 1.20a |
Discussion
This is the first study investigating the acute and short-term effects of anthocyanin-rich black rice consumption on cognitive performance, inflammatory markers, and microvascular function in older adults. Daily consumption of black rice, providing 208 mg of anthocyanins or 261 mg of total polyphenols for 8 days significantly improved verbal memory, with small to medium effect sizes (d), for final recall (d = 0.32) and total recall (d = 0.33) compared with the brown rice control. In parallel, a significant reduction in serum IL-6 levels (d = 0.39) relative to baseline was observed following black rice intake. No significant changes were detected in microvascular blood flow or blood pressure during this period. These data hint at the fact that anti-inflammatory effects of black rice polyphenols could potentially underpin cognitive effects behaviorally. According to acute analysis, a trend toward reduced verbal memory (RAVLT), microvascular blood flow, and blood pressure was observed for both treatments. However, black rice consumption was associated with attenuated declines in delayed recall on day 1 (d = 0.40) and word recognition on day 9 (d = 0.33) from baseline, but there was a lack of significant difference between treatments. Although acute intake showed a trend toward cognitive benefits, regular consumption may be required to achieve more substantial and sustained enhancements in cognitive function among older adults. These findings contribute to the growing body of evidence that flavonoid-rich foods may beneficially influence both cognitive performance and inflammatory status in humans.26,54–57The levels and compositions of anthocyanins, the pigments responsible for red, blue, and purple colors in plants, vary widely among different food sources.58 Black rice contains between 19.7 and 572 mg of cyanidin-3 glucoside per 100 g of uncooked grain, depending on the variety;40,59 however, cooking can reduce the available anthocyanin content by 40–60%.59–61 Therefore, the concentration is slightly lower than that of well-known anthocyanin-rich foods, such as blueberries, which typically contain 57–503 mg of total anthocyanins per 100 g of fresh weight.62 In our study, the anthocyanin content in cooked black rice was quantified at 208 mg per serving (210 g), comparable to the effective doses reported in other anthocyanin-rich foods or supplements in human cognitive trials, which have ranged from 16 to 598 mg.63 During acute intake, a transient decline in verbal memory, as measured by the RAVLT, was observed for both interventions. The observed cognitive changes may reflect a rapid postprandial rise in blood glucose and insulin, followed by a sharp decline, after ingestion of a carbohydrate-rich meal. Such fluctuations can induce drowsiness and fatigue, potentially impairing cognitive performance, particularly in tasks requiring sustained attention and memory.64 However, acute ingestion of black rice attenuated declines in specific memory measures, such as delayed recall (d = 0.40) and word recognition (d = 0.33) from baseline. The findings indicate that the acute cognitive effects of black rice consumption on both day 1 and day 9 consistently preserve episodic memory. This aligns with previous acute anthocyanin studies in older adults; for example, consumption of wild blueberries providing 475 mg of anthocyanins preserved word recognition performance over a 2-hour period in middle-aged to older adults.65 Similarly, the consumption of a blueberry beverage containing 508 mg of anthocyanins alleviated the decline in immediate word recognition (d = 0.44) over a 2 h period.20 Furthermore, a single dose of wild blueberry extract containing 222 mg of anthocyanins preserved executive function relative to placebo during the post-lunch dip.66 Here, black rice consumption similarly showed acute cognitive benefits by attenuating declines in verbal memory and recognition among older adults, although, higher-dose interventions may be required for larger effect sizes.
Compared with a single intake, daily consumption for 8 days produced more pronounced cognitive gains, with improvements in verbal memory, measured by final recall (d = 0.32) and total recall (d = 0.33) relative to the brown rice control. Additionally, a significant improvement from baseline was observed for digit span backward (d = 0.63) following black rice intake, suggesting a potential facilitative effect on working memory. These cognitive domains rely on the integrity and functional capacity of the prefrontal cortex, a region previously shown to benefit from flavonoid-rich diets.67–69 Given the low bioavailability, rapid metabolism and excretion of anthocyanins, regular consumption may maintain circulating levels of their metabolites, potentially sustaining cognitive function.70,71 Our findings suggest that regular anthocyanin-rich black rice consumption may provide cumulative benefits across memory domains, with effects comparable to those reported in other chronic berry anthocyanin interventions, administered at doses of 138–248 mg over 5–12 weeks.21,23,72 For example, a 5-week intake of a berry beverage containing 248 mg of anthocyanins significantly enhanced working memory task (d = 0.37).21 Similarly, daily consumption of cherry juice providing 138 mg of anthocyanins for 12 weeks resulted in significant improvements in verbal memory (d = 0.94), as well as short-term (d = 0.52) and long-term (d = 0.59) memory, in individuals with mild to moderate dementia.23 In addition, consuming 24 g of freeze-dried blueberry, equivalent to 230 mg of anthocyanins, daily for 90 days significantly reduced repetition errors in the RAVLT (d = 0.49) and decreased switch cost compared to the controls.72 Notably, variations in study design, population, anthocyanin composition and dosage, intervention duration, and the cognitive assessments employed have limited the comparability of findings across studies. Nevertheless, converging evidence suggests that certain cognitive domains, particularly verbal and working memory, appear to be responsive to anthocyanin interventions in older adults, with extended intervention durations and dosage likely producing larger effect sizes.
The potential mechanisms by which anthocyanins enhance cognitive performance, including anti-inflammation and vascular function, were investigated in this study. A significant decrease in IL-6 levels was observed, occurring in parallel with improvements in memory recall and working memory after 8 days of black rice intake. However, no significant changes in microvascular blood flow or blood pressure were detected over this timeframe. Therefore, the anti-inflammatory mechanism could potentially be linked to cognitive improvements following black rice intake. Notably, brown rice intake improved DSST scores, a measure of processing speed, although no reduction in IL-6 was observed. This distinction becomes obvious when considering domain-specific effects, as improvements in verbal memory following black rice intake appear to be related to anti-inflammatory mechanisms,73 whereas enhancements in processing speed after brown rice intake may occur through mechanisms independent of inflammation, such as regulated glucose metabolism, enhanced synaptic plasticity, increased neurotrophic factors, and reduced oxidative stress.74,75 IL-6, a pivotal cytokine, has been negatively correlated with global cognitive performance, particularly in domains related to memory and executive function among non-dementia older adults.5,76 A recent systematic review and meta-analysis of human randomized controlled trials supports our findings, showing that chronic polyphenols intake significantly reduces blood IL-6 levels while improving verbal memory and executive function.73 In addition to the reduction in IL-6, no significant changes were observed in serum TNF-α, hsCRP, ICAM-1, or VCAM-1 following black rice intake. It is possible that the relatively short intervention period limited the detection of broader inflammatory changes, consistent with previous evidence that dietary polyphenols often require longer durations to exert broader anti-inflammatory effects.77 Alternatively, the selective effect on IL-6 may suggest a more targeted mechanism of action by black rice anthocyanins, which warrants further mechanistic exploration.
Regarding the vascular system, a temporary decline in microvascular blood flow during the postprandial phase in the acute setting was observed following intake of both types of rice. In the same window, the timing of microvascular changes coincides with transient declines in cognitive performance, particularly verbal memory, suggesting a possible link between impaired microvascular function and cognitive dysfunction.15,78 Additionally, reductions in both systolic and diastolic blood pressure were also observed during the postprandial phase of both rice treatments, suggesting that vascular responsiveness, particularly via nitric oxide (NO)-mediated pathways, may be more sensitive to dietary compounds rather than specifically to the effects of anthocyanins. It has been similarly reported that acute anthocyanin intake from a blueberry beverage led to reduced blood pressure in older adults; however, the control drink produced a comparable effect.20 We are unable to exclude the possibility that acute vascular responses are a result of other bioactive compounds identified in both rice varieties, including γ-oryzanol, peptides, and phenolic acids.40 These compounds have been reported to promote vascular relaxation and modulate the renin–angiotensin system.79 Although no specific vascular effects of black rice were detected in the acute postprandial or short-term phases of this study, it is important to recognize that anthocyanin-rich foods have been linked to cardiovascular benefits such as improved endothelial function and reduced blood pressure, both of which are associated with enhanced cognitive performance.24 The positive vascular outcomes of anthocyanin-rich food may require longer duration or higher dose interventions to become measurable at the microvascular level, especially in older adults. Overall, the observed cognitive improvements were accompanied by reductions in the IL-6 inflammatory marker but not by changes in vasodilatory function, supporting the hypothesis that anti-inflammatory mechanisms may contribute to enhanced cognitive performance. However, anthocyanin-derived metabolites may also exert cognitive effects through other neuroprotective pathways, including upregulation of neurotrophic factors, modulation of neuronal signaling, enhancement of cerebral blood flow, activation of cognitive brain regions, and regulation of the gut–brain axis.80–82 Future research should consider these additional mechanisms to better elucidate the effects of anthocyanin-rich foods on cognitive function.
While many cognitive interventions focus on high-cost supplements or expensive anthocyanin-rich food such as blueberries, these may not be practical or sustainable at a population level. In contrast, this study investigated a staple-based food, black rice, which is culturally recognized and frequently consumed in many parts of Asia and around the world. The effect sizes for cognitive performance observed in this study are lower than those reported in studies using concentrated berry extracts, whole fruits, or beverages, likely due to lower anthocyanin concentrations, differences in anthocyanin composition, the influence of the food matrix, and the shorter intervention period. However, the cost-effectiveness and habitual nature of rice consumption make it a compelling choice for population-level dietary intervention. Although cooking partially degrades the anthocyanin content of black rice,59–61 the residual amount, approximately 208 mg per serving shown in our study, remains sufficient to exert cognitive function and anti-inflammatory activity, even over a relatively short intervention period. Furthermore, black rice contains a variety of nutrients and non-nutrients including, dietary fiber, peptides, vitamin E, vitamin B1, γ-oryzanol, and phenolic acids, providing additional benefits beyond anthocyanins.40 In this context, regular consumption of anthocyanin-rich black rice could offer meaningful public health benefits, particularly in populations with limited access to dietary supplements or fresh fruit. Importantly, our sensory data indicate that black rice was well accepted across all attributes, with 69% of participants expressing a willingness to substitute black rice for white rice, highlighting its feasibility for broader dietary integration. Thus, incorporating black rice into the daily diet, or substituting it for white rice, represents a practical strategy for populations seeking a cost-effective source of anthocyanins with potential health-promoting effects.
Conclusion
This study is the first to demonstrate the effects of anthocyanin-rich black rice on cognitive function, inflammation, and microvascular function in older adults. Our findings indicate that 8 days of daily black rice consumption improved verbal and working memory, alongside a reduction in the inflammatory marker IL-6, suggesting that the cognitive benefits may be mediated by anti-inflammatory pathways. These findings suggest that incorporating anthocyanin-rich foods into the diet could support cognitive health and reduce inflammatory burden, with potential implications for aging populations and individuals at risk of cognitive decline.Author contributions
C. M.: conceptualization, investigation, data curation, formal analysis and writing – original draft. D. J. L.: supervision, formal analysis, and writing – review & editing. J. P. E. S.: conceptualization, supervision, resources, writing – review and editing, and approval of the final version of the manuscript.Conflicts of interest
The authors declare no conflicts of interest.Data availability
All data supporting the results of this study are provided in the article. The raw data are not publicly available due to confidentiality restrictions, but may be provided by the corresponding author upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo04351d.
Acknowledgements
This research was conducted as part of Chusana Mekhora's PhD scholarship program, supported by a grant from the Royal Thai Government. The funding body had no role in the study design, data collection, analysis, interpretation, or manuscript preparation. The authors gratefully acknowledge all participants, staff, and colleagues who contributed to this study.References
- C. N. Harada, M. C. Natelson Love and K. L. Triebel, Normal cognitive aging, Clin. Geriatr. Med., 2013, 29, 737–752 CrossRef PubMed.
- E. R. Harrell, J. W. King, L. E. Stoeckel and M. Treviño, National institute on aging's 50th anniversary: Advancing cognitive aging research and the cognitive health of older adults, J. Gerontol. B. Psychol. Sci. Soc. Sci., 2024, 79, gbae120 CrossRef PubMed.
- A. Comas-Herrera, R. Wittenberg, L. Pickard and M. Knapp, Cognitive impairment in older people: Future demand for long-term care services and the associated costs, Int. J. Geriatr. Psychiatry, 2007, 22, 1037–1045 CrossRef PubMed.
- M. Baumgart, H. M. Snyder, M. C. Carrillo, S. Fazio, H. Kim and H. Johns, Summary of the evidence on modifiable risk factors for cognitive decline and dementia: A population-based perspective, Alzheimer's Dementia, 2015, 11, 718–726 CrossRef PubMed.
- A. C. Sartori, D. E. Vance, L. Z. Slater and M. Crowe, The impact of inflammation on cognitive function in older adults: Implications for healthcare practice and research, J. Neurosci. Nurs., 2012, 44, 206–217 CrossRef PubMed.
- A. Thomas, J. Guo, D. Reyes-Dumeyer, D. Sanchez, N. Scarmeas, J. J. Manly, A. M. Brickman, R. A. Lantigua, R. Mayeux and Y. Gu, Inflammatory biomarkers profiles and cognition among older adults, Sci. Rep., 2025, 15, 2265 CrossRef PubMed.
- C. Mekhora, D. J. Lamport and J. P. E. Spencer, An overview of the relationship between inflammation and cognitive function in humans, molecular pathways and the impact of nutraceuticals, Neurochem. Int., 2024, 181, 105900 Search PubMed.
- A. A. Simen, K. A. Bordner, M. P. Martin, L. A. Moy and L. C. Barry, Cognitive dysfunction with aging and the role of inflammation, Ther. Adv. Chronic Dis., 2011, 2, 175–195 CrossRef PubMed.
- L. Feng, Y. Wang, D. Zeng, M. Wang and X. Duan, Predictors of cognitive decline in older individuals without dementia: An updated meta-analysis, Ann. Clin. Transl. Neurol., 2023, 10, 497–506 CrossRef PubMed.
- X. N. Shen, L. D. Niu, Y. J. Wang, X. P. Cao, Q. Liu, L. Tan, C. Zhang and J. T. Yu, Inflammatory markers in Alzheimer's disease and mild cognitive impairment: A meta-analysis and systematic review of 170 studies, J. Neurol., Neurosurg. Psychiatry, 2019, 90, 590–598 CrossRef PubMed.
- X. Huang, B. Hussain and J. Chang, Peripheral inflammation and blood-brain barrier disruption: Effects and mechanisms, CNS Neurosci. Ther., 2021, 27, 36–47 CrossRef.
- F. Takata, S. Nakagawa, J. Matsumoto and S. Dohgu, Blood–brain barrier dysfunction amplifies the development of neuroinflammation: understanding of cellular events in brain microvascular endothelial cells for prevention and treatment of BBB dysfunction, Front. Cell. Neurosci., 2021, 15, 661838 Search PubMed.
- H. L. Zhou, X. Z. Jiang and Y. Ventikos, Role of blood flow in endothelial functionality: A review, Front. Cell Dev. Biol., 2023, 11, 1259280 Search PubMed.
- S. Ogoh, Relationship between cognitive function and regulation of cerebral blood flow, J. Physiol. Sci., 2017, 67, 345–351 Search PubMed.
- T. M. De Silva and F. M. Faraci, Microvascular dysfunction and cognitive impairment, Cell. Mol. Neurobiol., 2016, 36, 241–258 Search PubMed.
- M. R. Naiberg, D. F. Newton and B. I. Goldstein, Flow-mediated dilation and neurocognition: Systematic review and future directions, Psychosom. Med., 2016, 78, 192–207 CrossRef PubMed.
- D. Mastroiacovo, C. Kwik-Uribe, D. Grassi, S. Necozione, A. Raffaele, L. Pistacchio, R. Righetti, R. Bocale, M. C. Lechiara, C. Marini, C. Ferri and G. Desideri, Cocoa flavanol consumption improves cognitive function, blood pressure control, and metabolic profile in elderly subjects: The Cocoa, Cognition, and Aging (CoCoA) Study—a randomized controlled trial14, Am. J. Clin. Nutr., 2015, 101, 538–548 CrossRef.
- R. J. Kean, D. J. Lamport, G. F. Dodd, J. E. Freeman, C. M. Williams, J. A. Ellis, L. T. Butler and J. P. E. Spencer, Chronic consumption of flavanone-rich orange juice is associated with cognitive benefits: An 8-wk, randomized, double-blind, placebo-controlled trial in healthy older adults, Am. J. Clin. Nutr., 2015, 101, 506–514 CrossRef PubMed.
- M. H. Alharbi, D. J. Lamport, G. F. Dodd, C. Saunders, L. Harkness, L. T. Butler and J. P. E. Spencer, Flavonoid-rich orange juice is associated with acute improvements in cognitive function in healthy middle-aged males, Eur. J. Nutr., 2016, 55, 2021–2029 CrossRef PubMed.
- G. F. Dodd, C. M. Williams, L. T. Butler and J. P. E. Spencer, Acute effects of flavonoid-rich blueberry on cognitive and vascular function in healthy older adults, Nutr. Healthy Aging, 2019, 5, 119–132 CrossRef.
- A. Nilsson, I. Salo, M. Plaza and I. Björck, Effects of a mixed berry beverage on cognitive functions and cardiometabolic risk markers; A randomized cross-over study in healthy older adults, PLoS One, 2017, 12, e0188173 CrossRef.
- D. J. Lamport, D. Pal, A. L. Macready, S. Barbosa-Boucas, J. M. Fletcher, C. M. Williams, J. P. E. Spencer and L. T. Butler, The effects of flavanone-rich citrus juice on cognitive function and cerebral blood flow: An acute, randomised, placebo-controlled cross-over trial in healthy, young adults, Br. J. Nutr., 2016, 116, 2160–2168 CrossRef.
- K. Kent, K. Charlton, S. Roodenrys, M. Batterham, J. Potter, V. Traynor, H. Gilbert, O. Morgan and R. Richards, Consumption of anthocyanin-rich cherry juice for 12 weeks improves memory and cognition in older adults with mild-to-moderate dementia, Eur. J. Nutr., 2017, 56, 333–341 CrossRef.
- E. Wood, S. Hein, R. Mesnage, F. Fernandes, N. Abhayaratne, Y. Xu, Z. Zhang, L. Bell, C. Williams and A. Rodriguez-Mateos, Wild blueberry (poly)phenols can improve vascular function and cognitive performance in healthy older individuals: a double-blind randomized controlled trial, Am. J. Clin. Nutr., 2023, 117, 1306–1319 CrossRef.
- L. Bell, D. J. Lamport, L. T. Butler and C. M. Williams, A review of the cognitive effects observed in humans following acute supplementation with flavonoids, and their associated mechanisms of action, Nutrients, 2015, 7, 10290–10306 CrossRef.
- A. A. Fallah, E. Sarmast, P. Fatehi and T. Jafari, Impact of dietary anthocyanins on systemic and vascular inflammation: systematic review and meta-analysis on randomised clinical trials, Food Chem. Toxicol., 2020, 135, 110922 CrossRef.
- S. K. Bøhn, M. C. W. Myhrstad, M. Thoresen, I. Erlund, A. K. Vasstrand, A. Marciuch, M. H. Carlsen, N. E. Bastani, K. Engedal, K. M. Flekkøy and R. Blomhoff, Bilberry/red grape juice decreases plasma biomarkers of inflammation and tissue damage in aged men with subjective memory impairment – a randomized clinical trial, BMC Nutr., 2021, 7, 75 CrossRef.
- D. Grassi, G. Desideri, S. Necozione, P. di Giosia, R. Barnabei, L. Allegaert, H. Bernaert and C. Ferri, Cocoa consumption dose-dependently improves flow-mediated dilation and arterial stiffness decreasing blood pressure in healthy individuals, J. Hypertens., 2015, 33, 294–303 CrossRef CAS.
- C. Heiss, R. Sansone, H. Karimi, M. Krabbe, D. Schuler, A. Rodriguez-Mateos, T. Kraemer, M. M. Cortese-Krott, G. G. Kuhnle, J. P. Spencer, H. Schroeter, M. W. Merx and M. Kelm, Impact of cocoa flavanol intake on age-dependent vascular stiffness in healthy men: a randomized, controlled, double-masked trial, Age, 2015, 37, 9794 CrossRef.
- G. Istas, E. Wood, M. Le Sayec, C. Rawlings, J. Yoon, V. Dandavate, D. Cera, S. Rampelli, A. Costabile, E. Fromentin and A. Rodriguez-Mateos, Effects of aronia berry (poly)phenols on vascular function and gut microbiota: a double-blind randomized controlled trial in adult men, Am. J. Clin. Nutr., 2019, 110, 316–329 CrossRef PubMed.
- S. N. Hwang, J. C. Kim, M. I. H. Bhuiyan, J. Y. Kim, J. S. Yang, S. H. Yoon, K. D. Yoon and S. Y. Kim, Black Rice (Oryza sativa L., Poaceae) extract reduces hippocampal neuronal cell death induced by transient global cerebral ischemia in mice, Exp. Neurobiol., 2018, 27, 129–138 CrossRef PubMed.
- A. Y. Lee, J. M. Choi, Y. A. Lee, S. H. Shin and E. J. Cho, Beneficial effect of black rice (Oryza sativa L. var. japonica) extract on amyloid β-induced cognitive dysfunction in a mouse model, Exp. Ther. Med., 2020, 20, 64 Search PubMed.
- W. Pannangrong, J. Wattanathorn, S. Muchimapura, S. Tiamkao and T. Tong-Un, Purple rice berry is neuroprotective and enhances cognition in a rat model of Alzheimer's disease, J. Med. Food, 2011, 14, 688–694 CrossRef CAS.
- E. T. Callcott, C. L. Blanchard, P. Snell and A. B. Santhakumar, The anti-inflammatory and antioxidant effects of acute consumption of pigmented rice in humans, Food Funct., 2019, 10, 8230–8239 RSC.
- E. T. Callcott, C. L. Blanchard, P. Snell and A. B. Santhakumar, The anti-inflammatory and antioxidant effects of pigmented rice consumption in an obese cohort, Food Funct., 2019, 10, 8016–8025 RSC.
- T. Anuyahong, C. Chusak and S. Adisakwattana, Riceberry rice beverage decreases postprandial glycemic response, inflammatory markers and antioxidant status induced by a high-carbohydrate and moderate-fat meal in overweight and obese men, Food Funct., 2022, 13, 834–845 RSC.
- C. Chusak, P. Pasukamonset, P. Chantarasinlapin and S. Adisakwattana, Postprandial glycemia, insulinemia, and antioxidant status in healthy subjects after ingestion of bread made from anthocyanin-rich riceberry rice, Nutrients, 2020, 12, 782 Search PubMed.
- N. Muangchan, B. Khiewvan, S. Chatree, K. Pongwattanapakin, N. Kunlaket, T. Dokmai and R. Chaikomin, Riceberry rice (Oryza sativa L.) slows gastric emptying and improves the postprandial glycaemic response, Br. J. Nutr., 2022, 128, 424–432 CrossRef CAS.
- S. Das, T. Kumari, S. Babu, S. C. N. Kumar and S. C. Deka, Bioactive compounds, functional properties, health benefits, and food applications of black rice: A comprehensive review, Food Chem. Adv., 2025, 7, 101028 CrossRef.
- P. Goufo and H. Trindade, Rice antioxidants: phenolic acids, flavonoids, anthocyanins, proanthocyanidins, tocopherols, tocotrienols, γ-oryzanol, and phytic acid, Food Sci. Nutr., 2014, 2, 75–104 CrossRef CAS PubMed.
- A. W. Watson, C. F. Haskell-Ramsay, D. O. Kennedy, J. M. Cooney, T. Trower and A. Scheepens, Acute supplementation with blackcurrant extracts modulates cognitive functioning and inhibits monoamine oxidase-B in healthy young adults, J. Funct. Foods, 2015, 17, 524–539 Search PubMed.
- A. Rodriguez-Mateos, C. Rendeiro, T. Bergillos-Meca, S. Tabatabaee, T. W. George, C. Heiss and J. P. E. Spencer, Intake and time dependence of blueberry flavonoid-induced improvements in vascular function: A randomized, controlled, double-blind, crossover intervention study with mechanistic insights into biological activity, Am. J. Clin. Nutr., 2013, 98, 1179–1191 CrossRef CAS.
- L. Bell, D. J. Lamport, L. T. Butler and C. M. Williams, A study of glycaemic effects following acute anthocyanin-rich blueberry supplementation in healthy young adults, Food Funct., 2017, 8, 3104–3110 RSC.
- A. Rodriguez-Mateos, R. P. Feliciano, T. Cifuentes-Gomez and J. P. E. Spencer, Bioavailability of wild blueberry (poly)phenols at different levels of intake, J. Berry Res., 2016, 6, 137–148 CrossRef CAS.
- R. M. de Ferrars, C. Czank, Q. Zhang, N. P. Botting, P. A. Kroon, A. Cassidy and C. D. Kay, The pharmacokinetics of anthocyanins and their metabolites in humans, Br. J. Pharmacol., 2014, 171, 3268–3282 Search PubMed.
- C. Del Bó, P. Riso, J. Campolo, P. Møller, S. Loft, D. Klimis-Zacas, A. Brambilla, A. Rizzolo and M. Porrini, A single portion of blueberry (Vaccinium corymbosum L) improves protection against DNA damage but not vascular function in healthy male volunteers, Nutr. Res., 2013, 33, 220–227 CrossRef.
- T. Lin, G. A. Liu, E. Perez, R. D. Rainer, M. Febo, Y. Cruz-Almeida and N. C. Ebner, Systemic inflammation mediates age-related cognitive deficits, Front. Aging Neurosci., 2018, 10, 236 CrossRef PubMed.
- J. Turner, J. J. Belch and F. Khan, Current concepts in assessment of microvascular endothelial function using laser Doppler imaging and iontophoresis, Trends Cardiovasc. Med., 2008, 18, 109–116 Search PubMed.
- D. Vauzour, E. J. Houseman, T. W. George, G. Corona, R. Garnotel, K. G. Jackson, C. Sellier, P. Gillery, O. B. Kennedy, J. A. Lovegrove and J. P. E. Spencer, Moderate champagne consumption promotes an acute improvement in acute endothelial-independent vascular function in healthy human volunteers, Br. J. Nutr., 2010, 103, 1168–1178 Search PubMed.
- J. E. Ramsay, W. R. Ferrell, I. A. Greer and N. Sattar, Factors critical to iontophoretic assessment of vascular reactivity: implications for clinical studies of endothelial dysfunction, J. Cardiovasc. Pharmacol., 2002, 39, 19–17 Search PubMed.
- S. Jadhav, N. Sattar, J. R. Petrie, S. M. Cobbe and W. R. Ferrell, Reproducibility and repeatability of peripheral microvascular assessment using iontophoresis in conjunction with laser Doppler imaging, J. Cardiovasc. Pharmacol., 2007, 50, 343–349 Search PubMed.
- L. Bell, A. R. Whyte, D. J. Lamport, J. P. E. Spencer, L. T. Butler and C. M. Williams, Grape seed polyphenol extract and cognitive function in healthy young adults: A randomised, placebo-controlled, parallel-groups acute-on-chronic trial, Nutr. Neurosci., 2022, 25, 54–63 Search PubMed.
- J. Cohen, Statistical Power Analysis for the Behavioral Sciences, Lawrence Erlbaum Associates, Hillsdale, NJ, 1988 Search PubMed.
- E. Lorzadeh, K. Weston-Green, S. Roodenrys, V. do Rosario, K. Kent and K. Charlton, The effect of anthocyanins on cognition: A systematic review and meta-analysis of randomized clinical trial studies in cognitively impaired and healthy adults, Curr. Nutr. Rep., 2025, 14, 23 Search PubMed.
- V. Socci, D. Tempesta, G. Desideri, L. De Gennaro and M. Ferrara, Enhancing human cognition with cocoa flavonoids, Front. Nutr., 2017, 4, 00019 Search PubMed.
- N. Cheng, L. Bell, D. J. Lamport and C. M. Williams, Dietary flavonoids and human cognition: a meta-analysis, Mol. Nutr. Food Res., 2022, 66, e2100976 Search PubMed.
- A. F. Curtis, M. Musich, A. N. Costa, J. Gonzales, H. Gonzales, B. J. Ferguson, B. Kille, A. L. Thomas, X. Wei, P. Liu, C. M. Greenlief, J. I. Shenker and D. Q. Beversdorf, Feasibility and preliminary efficacy of american elderberry juice for improving cognition and inflammation in patients with mild cognitive impairment, Int. J. Mol. Sci., 2024, 25, 4352 Search PubMed.
- M. Lakshmikanthan, S. Muthu, K. Krishnan, A. B. Altemimi, N. N. Haider, L. Govindan, J. Selvakumari, Z. T. Alkanan, F. Cacciola and Y. M. Francis, A comprehensive review on anthocyanin-rich foods: Insights into extraction, medicinal potential, and sustainable applications, J. Agric. Food Res., 2024, 17, 101245 Search PubMed.
- M. Hiemori, E. Koh and A. E. Mitchell, Influence of cooking on anthocyanins in black rice (Oryza sativa L. japonica var. SBR), J. Agric. Food Chem., 2009, 57, 1908–1914 Search PubMed.
- A. Colasanto, F. Travaglia, M. Bordiga, J. D. Coïsson, M. Arlorio and M. Locatelli, Impact of traditional and innovative cooking techniques on Italian black rice (Oryza sativa L., Artemide cv) composition, Food Res. Int., 2024, 194, 114906 Search PubMed.
- J. Surh and E. Koh, Effects of four different cooking methods on anthocyanins, total phenolics and antioxidant activity of black rice, J. Sci. Food Agric., 2014, 94, 3296–3304 Search PubMed.
- D. Stevenson and J. Scalzo, Anthocyanin composition and content of blueberries from around the world, J. Berry Res., 2012, 2, 179–189 Search PubMed.
- S. Ahles, P. J. Joris and J. Pla, Effects of berry anthocyanins on cognitive performance, vascular function and cardiometabolic risk markers: a systematic review of randomized placebo-bontrolled intervention studies in humans, Int. J. Mol. Sci., 2021, 22, 6482 Search PubMed.
- M. R. Rizzo, R. Marfella, M. Barbieri, V. Boccardi, F. Vestini, B. Lettieri, S. Canonico and G. Paolisso, Relationships between daily acute glucose fluctuations and cognitive performance among aged type 2 diabetic patients, Diabetes Care, 2010, 33, 2169–2174 Search PubMed.
- A. R. Whyte, S. Rahman, L. Bell, I. Edirisinghe, R. Krikorian, C. M. Williams and B. Burton-Freeman, Improved metabolic function and cognitive performance in middle-aged adults following a single dose of wild blueberry, Eur. J. Nutr., 2021, 60, 1521–1536 Search PubMed.
- N. Cheng, K. L. Barfoot, R. Le Cozannet, P. Fança-Berthon, D. J. Lamport and C. M. Williams, Wild blueberry extract intervention in healthy older adults: A multi-study, randomised, controlled investigation of acute cognitive and cardiovascular effects, Nutrients, 2024, 16, 1180 Search PubMed.
- J. P. E. Spencer, The impact of fruit flavonoids on memory and cognition, Br. J. Nutr., 2010, 104, 40–47 Search PubMed.
- A. L. Macready, O. B. Kennedy, J. A. Ellis, C. M. Williams, J. P. E. Spencer and L. T. Butler, Flavonoids and cognitive function: A review of human randomized controlled trial studies and recommendations for future studies, Genes Nutr., 2009, 4, 227–242 Search PubMed.
- J. L. Bowtell, Z. Aboo-Bakkar, M. E. Conway, A. R. Adlam and J. Fulford, Enhanced task-related brain activation and resting perfusion in healthy older adults after chronic blueberry supplementation, Appl. Physiol., Nutr., Metab., 2017, 42, 773–779 Search PubMed.
- M. G. Miller and B. Shukitt-Hale, Berry fruit enhances beneficial signaling in the brain, J. Agric. Food Chem., 2012, 60, 5709–5715 Search PubMed.
- A. Rodriguez-Mateos, D. Vauzour, C. G. Krueger, D. Shanmuganayagam, J. Reed, L. Calani, P. Mena, D. Del Rio and A. Crozier, Bioavailability, bioactivity and impact on health of dietary flavonoids and related compounds: An update, Arch. Toxicol., 2014, 88, 1803–1853 Search PubMed.
- M. G. Miller, D. A. Hamilton, J. A. Joseph and B. Shukitt-Hale, Dietary blueberry improves cognition among older adults in a randomized, double-blind, placebo-controlled trial, Eur. J. Nutr., 2018, 57, 1169–1180 Search PubMed.
- C. Mekhora, D. J. Lamport and J. P. E. Spencer, Effect of polyphenols on inflammation related to cognitive function: A systematic review and meta-analysis of human randomized controlled trials, Nutr. Healthy Aging, 2024, 9, 113–131 Search PubMed.
- M. Uenobe, T. Saika, N. Waku, M. Ohno and H. Inagawa, Efficacy of continuous ingestion of dewaxed brown rice on the cognitive functions of the residents of elderly welfare facilities: A pilot test using crossover trial, Food Sci. Nutr., 2019, 7, 3520–3526 CrossRef PubMed.
- W. Rungratanawanich, G. Cenini, A. Mastinu, M. Sylvester, A. Wilkening, G. Abate, S. A. Bonini, F. Aria, M. Marziano, G. Maccarinelli, M. Memo, W. Voos and D. Uberti, γ-Oryzanol improves cognitive function and modulates hippocampal proteome in mice, Nutrients, 2019, 11, 753 CrossRef PubMed.
- K. Yaffe, K. Lindquist, B. W. Penninx, E. M. Simonsick, M. Pahor, S. Kritchevsky, L. Launer, L. Kuller, S. Rubin and T. Harris, Inflammatory markers and cognition in well-functioning African-American and white elders, Neurology, 2003, 61, 76–80 CrossRef PubMed.
- I. Peluso, A. Raguzzini and M. Serafini, Effect of flavonoids on circulating levels of TNF-α and IL-6 in humans: A systematic review and meta-analysis, Mol. Nutr. Food Res., 2013, 57, 784–801 CrossRef PubMed.
- S. P. Rensma, T. T. van Sloten, A. J. H. M. Houben, S. Köhler, M. P. J. van Boxtel, T. T. J. M. Berendschot, J. F. A. Jansen, F. R. J. Verhey, A. A. Kroon, A. Koster, W. H. Backes, N. Schaper, G.-J. Dinant, C. G. Schalkwijk, R. M. A. Henry, E. M. L. Wolfs, M. J. A. van Heumen, M. T. Schram and C. D. A. Stehouwer, Microvascular dysfunction is associated with worse cognitive performance, Hypertension, 2020, 75, 237–245 CrossRef PubMed.
- N. Shobako and K. Ohinata, Anti-hypertensive effects of peptides derived from rice bran protein, Nutrients, 2020, 12, 3060 CrossRef PubMed.
- G. A. Rutledge, A. K. Sandhu, M. G. Miller, I. Edirisinghe, B. B. Burton-Freeman and B. Shukitt-Hale, Blueberry phenolics are associated with cognitive enhancement in supplemented healthy older adults, Food Funct., 2021, 12, 107–118 RSC.
- F. Hedayatikatouli, M. Kalyn, D. Elsaid, H. A. Mbesha and M. Ekker, Neuroprotective effects of ascorbic acid, vanillic acid, and ferulic acid in dopaminergic neurons of zebrafish, Biomedicines, 2024, 12, 2497 CrossRef PubMed.
- K. Krzysztoforska, D. Mirowska-Guzel and E. Widy-Tyszkiewicz, Pharmacological effects of protocatechuic acid and its therapeutic potential in neurodegenerative diseases: Review on the basis of in vitro and in vivo studies in rodents and humans, Nutr. Neurosci., 2019, 22, 72–82 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |

