Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Persimmon by-products, rich in fibre, promote beneficial gut bacteria
Carlos
Sabater†
 a,
Lucía
López-Bermudo†
a,
Lucía
López-Bermudo†
 bc,
Bryan
Moreno-Chamba
bc,
Bryan
Moreno-Chamba
 de,
Julio
Salazar-Bermeo
de,
Nicholas J.
Hayward
a,
Amanda
Morris
a,
Gary J.
Duncan
a,
Wendy R.
Russell
a,
Genoveva
Berná
bc,
Blanca
Escudero-López
bc,
Nuria
Martí Bruña
d,
Sylvia H.
Duncan
a,
Madalina
Neacsu
de,
Julio
Salazar-Bermeo
de,
Nicholas J.
Hayward
a,
Amanda
Morris
a,
Gary J.
Duncan
a,
Wendy R.
Russell
a,
Genoveva
Berná
bc,
Blanca
Escudero-López
bc,
Nuria
Martí Bruña
d,
Sylvia H.
Duncan
a,
Madalina
Neacsu
 *a and
Franz
Martin
*bc
*a and
Franz
Martin
*bc
aRowett Institute, University of Aberdeen, Foresterhill, Aberdeen AB25 2ZD, UK. E-mail: m.neacsu@abdn.ac.uk
bAndalusian Center of Molecular Biology and Regenerative Medicine (CABIMER), Pablo de Olavide University, University of Seville, CSIC, 41092 Seville, Spain. E-mail: lucia.lopez@cabimer.es; besclop@upo.es; gberamo@upo.es; fmarber@upo.es
cBiomedical Research Network on Diabetes and Related Metabolic Diseases (CIBERDEM), Instituto de Salud Carlos III, 28029 Madrid, Spain
dInstitute of Research, Development and Innovation in Sanitary Biotechnology of Elche, Miguel Hernández University, 03202 Elche, Spain. E-mail: bryan.morenoc@umh.es; julio.salazar@goumh.umh.es; nmarti@umh.es
eUniversity Institute of Food Engineering-FoodUPV, Universitat Politècnica de València, Valencia, 46022 Valencia, Spain
First published on 17th February 2026
Abstract
Persimmon by-products are a promising source of bioactive ingredients that could be used for functional food formulation and health promotion. The aim of this study was to investigate the microbiota modulatory properties of persimmon soluble and insoluble fibre fractions. For this purpose, an in vitro faecal fermentation experiment was carried out and microbiota profiles were analysed by amplicon sequencing. According to the results obtained, different persimmon fractions selectively promoted the growth of potentially probiotic genera including Bacteroides, Megasphaera, Oscillibacter and Lachnospiraceae members. Members of these taxa are important short-chain fatty acid (SCFA) producers showing statistical associations with other members of the gut microbiota. The influence of raw material composition on the fermentative profiles was also determined. Soluble non-starch polysaccharide (NSP) fractions rich in arabinose and fucose promote Oscillibacter while insoluble NSP fractions led to an increase in Megasphaera. The effect of phenolic molecules on the fermentative properties of persimmon substrates was also investigated. Faecal fermentation of soluble persimmon fractions led to higher production of indole-3-propionic acid compared to insoluble fractions. These findings underscore the potential health benefits of soluble fibre-rich persimmon by-products. Results presented here highlight the potential applications of persimmon by-products to formulate prebiotic ingredients that selectively stimulate the growth of beneficial gut commensals including next-generation probiotics.
Introduction
The popularity of persimmon (Diospyros kaki Thunb.) fruits has increased markedly due to their potential health benefits,1 with over four million tons produced annually2 in countries including China, Japan, and Spain. While comprehensive global data on persimmon waste are limited, it is estimated that approximately one-third of all food produced worldwide is lost or wasted along the agri-food chain.3 In particular, the losses of persimmon during harvest and postharvest processes were estimated at 29.5% of the total produced volume.4Persimmon fruits, particularly the Rojo Brillante variety, are sold as fresh fruit and processed into preserves. Persimmon juice is also a valuable product and a growing market. During processing, approximately 20–30% of the fruit mass, mainly peel and fruit fibre, is discarded as a by-product.5,6 Given the high volume of persimmon waste, there is growing interest in optimizing and valorising these by-products to create value-added products, aligning with circular economy principles. Persimmon by-products are rich in dietary fibre, particularly insoluble fractions, and contain significant amounts of bound polyphenols. According to Salazar-Bermeo et al.,5 the main fibre components in persimmon by-products include cellulose, hemicellulose, and pectin, with cellulose being the predominant fraction. This composition contributes to both the structural integrity and fermentability of the fibre matrix, making it a promising substrate for gut microbial fermentation.
Dietary fibre constitutes a fundamental component of human nutrition and is increasingly recognized for its multifaceted health benefits. It comprises non-digestible carbohydrates and lignin that resist enzymatic digestion in the small intestine and undergo fermentation in the colon.7 Fiber is broadly categorised into soluble and insoluble types. Soluble fibre dissolves in water to form viscous gels that modulate glucose and lipid absorption, while insoluble fibre adds bulk to stool and facilitates intestinal transit.8 Both types contribute to gut health, metabolic regulation, and chronic disease prevention, including cardiovascular disease, type 2 diabetes, and colorectal cancer9 supporting the growth of beneficial colonic microbes, the majority of which are anaerobes that ferment fibre mainly to short chain fatty acids (SCFAs).10
Despite its importance, dietary fibre intake remains below recommended levels in many populations. According to the Dietary Guidelines for Americans 2020–2025, adults should consume 14 grams of fibre per 1000 calories, which translates to approximately 25 grams per day for women and 38 grams per day for men.11 In Europe, the European Food Safety Authority (EFSA) recommends a minimum of 25 grams of fibre per day for adults to support normal bowel function and reduce disease risk.12 However, most European adults consume significantly less, with average intakes ranging from 18–24 g day−1 for men and 16–20 g day−1 for women, depending on the country.13
Fermentation of dietary fibre by colonic bacteria produces short-chain fatty acids (SCFAs) such as acetate, propionate, and butyrate, which play key roles in maintaining intestinal barrier integrity, modulating immune responses, and reducing inflammation.14 Moreover, certain dietary fibres, including those derived from persimmon fruits, are rich in bioactive molecules such as polyphenols. These compounds, including gallic and protocatechuic acids, delphinidin, and cyanidin, are often bound to fibre matrices and released during microbial fermentation in the colon.15,16
The interaction between dietary fibre and polyphenols is increasingly recognised as a critical determinant of their bioavailability and biological activity. These interactions are largely governed by non-covalent bonds, such as hydrogen bonding and hydrophobic interactions, between polyphenolic compounds and the polysaccharide components of dietary fibre.17 This binding can protect polyphenols from early degradation in the upper gastrointestinal tract, delaying their release until they reach the colon, where microbial fermentation liberates these compounds.15 Once released, polyphenols can exert local anti-inflammatory and antioxidant effects, contributing to gut barrier integrity and immune modulation. Fibre–polyphenol complexes also influence digestive enzyme activity, potentially reducing carbohydrate and lipid absorption, and modulate microbial metabolism, enhancing the production of bioactive metabolites such as phenolic acids and short-chain fatty acids (SCFAs).18 These biochemical interactions are essential to understanding the impact of persimmon fibre on gut microbiota. Polyphenols can selectively inhibit pathogenic bacteria while promoting beneficial taxa such as Faecalibacterium prausnitzii, Roseburia, and Bifidobacterium, contributing to a more favourable microbial profile and improved gut health.19 Our previous work has shown that fibre-rich fractions prepared from persimmon by-products are important sources of phytochemicals and promote the growth of beneficial human gut bacteria, including Faecalibacterium prausnitzii, while significantly decreasing the inflammatory effect of interleukin-1β in Caco-2 cells, and interleukin-6 and tumour necrosis factor-α in RAW 264.7 cells.16
There is strong evidence, therefore, that persimmon by-products could be valuable ingredients for the development of functional foods to promote metabolic and gut health. However, there is limited evidence on the modulatory effect of persimmon fibre on human gut microbiota. Here, we investigated the fermentative properties of high-fibre persimmon fractions (Rojo Brillante variety) when incubated with mixed faecal microbiota from healthy human volunteers. Structure–activity relationships of persimmon fractions and their role in the modulation of gut microbiota were also investigated. This work aims therefore to bring key information to promote the use of persimmon fibre-rich by-products as a novel source for prebiotics development.
Materials and methods
Standards and reagents
Standards and general laboratory reagents were purchased from Sigma-Aldrich (Gillingham, UK) and Fisher Scientific UK Ltd (Loughborough, UK) or synthesised as described previously.20,21Sample preparation
Fresh persimmon fruits (Red Brilliant variety) were purchased from a local market in Elche, Spain. The fruits were processed to separate juice and solid by-products. The by-products, made up of pulp and peels, were hydrated with water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 w/v) at room temperature prior to fermentation or hydrolysis treatments (Fig. 1) as described elsewhere.16,22
10 w/v) at room temperature prior to fermentation or hydrolysis treatments (Fig. 1) as described elsewhere.16,22
For the batches subjected to fermentation treatment, 1 L of by-products suspension was inoculated with 1 mL of fresh suspensions (107 CFU mL−1) of each Streptococcus salivarius subsp. thermophilus CECT 7207 and Lactobacillus casei CECT 475. The suspensions were incubated at 37 °C for 24 h at 150 rpm in a benchtop incubator shaker. After incubation, the suspensions were filtered, discarding the liquid. The solid fractions were vacuum dried at 60 °C overnight. The powder obtained from each sample was further subjected to hydrolysis. The bacterial strains were selected because they are commonly used in food fermentation and/or naturally present in fermented products, and previous studies showed their growth in persimmon fibres under various pre-treatments.23 Their use aimed to reduce free sugar content while increasing the bioaccesibility of non-extractable phytochemicals in NSP fractions, ensuring the effects on the gut microbiome could be attributed primarily to persimmon fibres.
For the hydrolysis treatment, the pH of the non-fermented (untreated) and fermented by-product suspensions was alkaline hydrolysed by adjusting the pH of each suspension to 12 with 5 M NaOH (colour change of suspension from clear to deep dark) using a pH meter (Hanna Instruments edge®, Daselab S.L., Valencia, Spain). The mixtures were heated at 40 °C for 24 h under constant shaking (150 rpm) in a benchtop incubator shaker; then, the pH was lowered to 2.5 with 5 M HCl (acidic hydrolysis, colour change from deep dark to red). The suspensions were filtered to separate the solids and liquids from both untreated and fermented batches. The obtained solid and liquid fractions were freeze dried separately (LyoQuest, Telstar, Barcelona, Spain) to obtain the red untreated soluble and insoluble fractions, as well as the red fermented soluble and insoluble fractions from persimmon by-products. The subsequent hydrolysis was applied to obtain both soluble and insoluble fractions, allowing analysis of extractable and non-extractable polyphenols. This process was performed in all samples consistently evaluate complete fractionation and characterization of persimmon fibre components.
Non starch polysaccharides (NSPs) analysis of persimmon fractions
We have used previous published method,24 to determine the soluble and insoluble NSP. The analysis of NSP composition was performed by gas chromatography using a Hewlett Packard HP7890N chromatograph equipped with a SP2330 30 m × 0.75 mm column.Phytochemical analyses of persimmon samples by HPLC and LC-MS/MS
The persimmon fractions were measured for derivatives and metabolites of the simple phenols, benzoic acids, phenolic acids, phenylacetic acids, pheny-propionic acids, phenylpyruvic acids, phenyllactic acids, mandellic acids, phenolic dimers, acetophenones, benzaldehydes, cinnamal-dehydes, benzyl alcohols, cinnamyl alcohols, indoles, isoflavones, coumarins, chalcones, flavanones, flavones, and flavonols using LC-MS/MS analysis and anthocyanidins by HPLC analysis.The method used for the extraction of the phenolic compounds prior LC-MS/MS analysis was consisting in a three-step extraction generating three different fractions, as previously described.25 Briefly, the samples were initially suspended in hydrochloric acid and extracted into ethyl acetate. This extraction was repeated three times, and the ethyl acetate extracts combined and were then evaporated to dryness (representing the “free fraction” or extractable polyphenols-EPP) and was stored at −70 °C prior to analysis by LC–MS/MS. The remaining aqueous fraction was first alkaline hydrolysed and then acid hydrolysed at room temperature for 4 h and respectively 30 minutes at 95 °C and following each the samples were then extracted into ethyl acetate (pH 2) and processed as described above. The extracts obtained after alkaline and acid hydrolysis represent the “bound fractions” or non-extractable polyphenols (NEPP).
The LC–MS/MS analysis of phytochemicals: for the LC–MS/MS analysis methods published previously have been used.33–35 Liquid chromatography separation of the metabolites was performed on an Agilent 1100 LC–MS system (Agilent Technologies, Wokingham, UK) using a Zorbax Eclipse 5 μm, 150 mm × 4.6 mm C18 column (Agilent Technologies). Three distinct gradients were used to separate the different categories of metabolites and the mobile phase solvents in each case were water containing 0.1% acetic acid and acetonitrile containing 0.1% acetic acid. The eluent was then directed without splitting into an ABI 3200 triple quadrupole mass spectrometer (Applied Biosystems, Warrington, UK) fitted with a Turbo Ion Spray™ (TIS) source. All the metabolites were quantified using multiple reaction monitoring (MRM). For all the phytochemical quantifications, standard calibrations curves were prepared in concentration intervals of 2 ng μl−1 up to 10 pg μl−1. The threshold used for quantification had a signal to noise ratio of 3 to 1. All the ion transitions for each of the metabolites were determined based upon their molecular ions and strong fragment ions; their voltage parameters; declustering potential, collision energy and cell entrance/exit potentials were optimized individually for each metabolite and have been previously described.25–27
To measure the anthocyanidin content of the persimmon fractions was used the extraction and hydrolysis methods adapted from Zhang et al.28 Briefly, samples (n = 3) of persimmon samples (0.1 g) were extracted with methanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water
water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) hydrochloric acid (ratio of 50
hydrochloric acid (ratio of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33
33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 17; v/v/v; 3 mL) three times, and the supernatants and the pellet combined and hydrolysed at 100 °C for 60 min. Hydrolysed samples were then immediately cooled to room temperature, filtered using 0.2 µm filters and analysed by HPLC.
17; v/v/v; 3 mL) three times, and the supernatants and the pellet combined and hydrolysed at 100 °C for 60 min. Hydrolysed samples were then immediately cooled to room temperature, filtered using 0.2 µm filters and analysed by HPLC.
The quantification of the anthocyanidins was performed using a 1260 Infinity HPLC from Agilent (Wokingham, UK) and a Synergi 4 µm Polar-RP 80A (250 × 4.6 mm) column with a Polar-RP 4 × 3 mm pre-column from Phenomenex (Macclesfield, UK). The DAD spectra were recorded between 200 and 700 nm and the chromatograms were monitored at 530 nm.
Analysis of free sugars composition in persimmon fractions
The quantification of mono- and disaccharides from the persimmon fraction was performed using a 1260 Infinity HPLC from Agilent (Wokingham, UK) equipped with a RI detection and an Asahipak NH2P-50 4E (5 μm; 25 cm × 0.46 cm), (Shodex, Japan) column connected to an Asahipak NH2P-50G 4A pre-column (4.6 mm × 10 mm, Shodex, Japan). Persimmon fractions (approx. 0.05 g, n = 3) were dissolved in 250 μl of water and filtered using 0.2 µm filters prior to the HPLC analysis using an isocratic solvent program consisting of 70% acetonitrile at a constant flow of 1 mL min−1. Quantification of the free sugars was performed using external calibration curves using validated standards for each sugar analysed.Incubations of persimmon fractions with mixed microbiota prepared from healthy human volunteers’ faecal samples
The incubation of persimmon fractions with mixed microbiota was prepared from six human faecal samples; one sample from three volunteers (D1, D2 and D3) for soluble persimmon fraction (untreated and fermented) incubations and respectively one sample from three volunteers (D2, D3 and D4) for insoluble persimmon fraction (untreated and fermented) incubations. For this study, we recruited four healthy males and females, which consume an omnivore diet, with a BMI between 18.5 and 30 kg m−2, with age between 18- and 65-year-old. The volunteer did not need to follow any specific dietary restriction prior to the donation of their fecal samples, and the samples were used as a source of mixed microbiota for the microbial incubation for this study. In the recruitment of the volunteers, we excluded people who have taken antibiotics within the 3 months prior fecal sample donation, were under medication for high blood pressure, were under treatment for severe IBD (inflammatory bowel disease) and IBS (irritable bowel syndrome), were suffering from autoimmune disorders or cancer and undergone any type of cancer treatment within the 12 months prior fecal sample donation. This study was conducted in accordance with the Declaration of Helsinki and good clinical practice, and all procedures involving human subjects were reviewed and approved by the Human Studies Management Committee of the Rowett Institute, University of Aberdeen, UK, and the Rowett Institute Ethics Panel. Informed consent was obtained from all subjects involved in the study. The study was carried out from March until July 2023.These experiments were carried out as previously described (Zhang et al., 2023).29 Briefly, 20 ± 1 mg persimmon fractions were weighed in six duplicates into pre-weighed Hungate tubes sealed with butyl rubber stoppers and screw caps (Bellco Glass, Shrewsbury, UK). Under an anaerobic condition CO2 maintained, 7.5 mL M2 basal medium (containing no soluble starch, glucose, and cellobiose, M2SGC) were added into those Hungate tubes, following by subsequent autoclaving and cooling to room temperature. The faecal slurry was achieved by anaerobically mixing 2 g of fresh feces with 8 mL of anaerobic 50 mM phosphate buffer containing 0.05% cysteine and vortex mixing. Three tubes of six duplicates were inoculated with the 100 µL faecal slurry under CO2 and incubated at 37 °C, while three control tubes were added with 100 µL sterile buffer. Therefore, we had several controls for this experimental setting, the media plus slurry control (without persimmon matrix), and the matrix control (the media and persimmon matrix without fecal slurry). Samples recovered at 0 h and 24 h were also used for short chain fatty acid (SCFA) analysis, DNA extractions and microbial metabolite analyses.
Analysis of short-chain fatty acid (SCFAs) formed during faecal incubation experiments
The concentrations of SCFA formed during the faecal incubations of the persimmon fractions with faecal samples of human volunteers (at 0 h and 24 h) were measured by gas chromatography as previously described.30 Following conversion to tert-butyldimethylsilyl derivatives, 1 μl of sample was analysed using a Hewlett-Packard (Palo Alto, CA, USA) gas chromatograph.Analysis of microbial metabolites analysis formed during faecal incubation experiments
Samples recovered at 0 h and 24 h during faecal incubations experiments were mixed with internal standard for negative mode mass spectrometry 13C benzoic acid at 2 µg mL−1 and, respectively, 2-amino-3,4,7,8-tetramethylimidazo(4,5-f) quinoxaline at 0.5 µg mL−1, for positive mode mass spectrometry prepared in methanol and centrifuged at 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g for 10 min. The supernatant was used for LC-MS/MS analysis as described in section 2.4.
000g for 10 min. The supernatant was used for LC-MS/MS analysis as described in section 2.4.
DNA extraction and high-throughput sequencing of 16S rRNA
Total DNA was extracted using the FastDNA® SPIN Kit for Feces (MP Biomedicals 116570200, MP Biomedicals SARL, Illkirch, France). All the steps of the protocol were performed according to the manufacturer's instructions. A total of 72 samples were selected for partial 16S rRNA sequencing including different substrates (fermentation times 0 h and 24 h): (i) soluble fraction control (faecal slurry + media; 3 donors, 2 time points per donor and 2 replicates per time point; n = 12), (ii) insoluble fraction control (n = 12), (ii) untreated soluble fraction (3 donors, 2 time points per donor and 2 replicates per time point; n = 12), (iii) treated insoluble fraction (n = 12), (iv) fermented soluble fraction (n = 12), (v) fermented insoluble fraction (n = 12).Amplicon sequencing of the V1–V2 region of bacterial 16S rRNA genes employing barcoded fusion primers MiSeq-27F (5′-AATGATACGGCGACCACCGAGATCTACACTATGGTAATTCCAGMGTTYGATYMTGGCTCAG-3′) and MiSeq-338R (5′-CAAGCAGAAGACGGCATACGAGAT-barcode-AGTCAGTCAGAAGCTGCCTCCCGTAGGAGT-3′) were carried out in an Illumina MiSeq platform at the in-house facilities at the Center for Genomic Enabled Biology and Medicine (CGEBM), University of Aberdeen, UK.
Sequence reads were quality filtered, and the resulting ones were processed using a personalized script of QIIME2 v.2021.8 software,31 matched by pair-ends. Quality control filtering was performed, keeping sequences with a mean sequence quality score >20. To analyse the taxonomic profiles of faecal fermentation samples, the 16S rRNA reads were clustered in amplicon sequence variants (ASVs) using Reference database SILVA 138 release.32–34 Raw sequence reads have been deposited in the Short Reads Archive (SRA) of the National Center for Biotechnology Information (NCBI) under BioProject accession number PRJNA1266485.
Bioinformatics and statistical analysis
Statistical analysis of microbiota composition results was performed on R (v.4.4.1). To characterise microbial diversity, alpha (Chao1 and Shannon) and beta (Bray Curtis dissimilarity metric) diversity estimators were calculated using microbiome (v.1.26.0)35 and Phyloseq (v.1.48.0)36 R packages. Comparative plots describing the distribution of microbial communities across groups of samples were generated using microbiome (v.1.26.0) R package:35 hierarchical cluster, principal coordinate analysis (PCoA) and microbiota composition barplots.The microbiota modulatory effect of different persimmon-derived fractions on specific microbial genera was calculated using several microbiome-specific statistical methods: ANCOM, ANCOMBC, ALDEx2, LEfSe and metagenomeSeq and Limma-Voom implemented in microbiomeMarker (v.1.10.0) R package.37–43 For this purpose, statistically significant (p < 0.05 and padj < 0.25) differences in microbiota composition (i.e., normalized microbial read counts) at initial (0 h) and final (24 h) fermentation times were calculated for each substrate. It should be noted that sequencing reads were normalised prior differential analysis using total sum scaling (TSS) method implemented in microbiomeMarker (v.1.10.0) R package. In this regard, data were normalized by dividing the corresponding sample library size. Potential associations between microbial genera modulated by persimmon fractions were elucidated in a correlation network computed using corrr (v.0.4.4) R package.44
On the other hand, general statistical analysis of phytochemical and SCFAs data included Shapiro–Wilk and Levene to test normality and variance homogeneity, ANOVA and Tukey post-hoc test for parametric data, Kruskal–Wallis and Dunn post-hoc test for non-parametric data using base R (v.4.4.1.) functions. Statistically significant (p < 0.05) correlations between microbiota composition profiles, and raw material composition were calculated and expressed as Pearson correlation coefficients using base R (v.4.4.1.) function and mixOmics (v.6.28.0) R package.45
Results
Characterisation of persimmon fractions
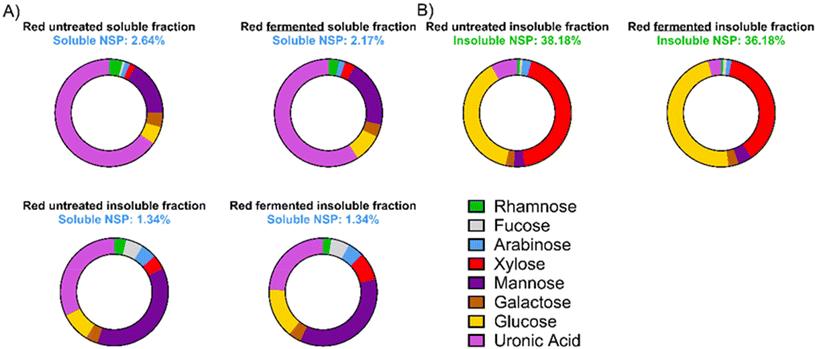
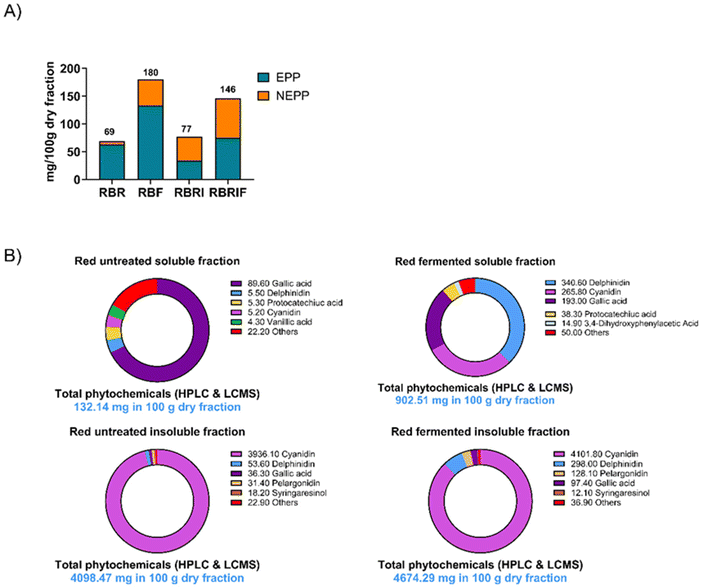
Novel persimmon fractions that may exert microbiota modulatory effects have been obtained. Soluble and insoluble fractions were first characterised. Soluble non-starch polysaccharides (NSPs) fraction comprised 2.64 and 2.17% of untreated and treated products, respectively (Fig. 2). Uronic acids, mannose, glucose and galactose were the most abundant monosaccharides in these fractions. Regarding the insoluble fraction, soluble NSPs comprised 1.34% of untreated and treated products. These fractions were characterised by higher mannose and glucose contents compared to the soluble fractions (Fig. 2). Finally, insoluble NSPs comprised 38.18 and 36.18% of untreated and treated products, respectively. Glucose and xylose were the predominant monosaccharides in these insoluble NSPs. As expected, the amount of free glucose and fructose was higher in the soluble fraction compared to the insoluble fraction (Fig. 2).Phytochemical composition of persimmon products was also determined (Fig. 3, SI Table S1). Major differences were observed between treated and untreated fractions. Gallic acid, delphinidin, protocatechuic acid were the major phytochemicals of the untreated soluble fraction. The proportion of delphinidin and cyanidin increased significantly in the treated soluble fraction (Fig. 3, Table S1). Cyanidin was the most abundant compound in the untreated insoluble fraction, and the treatment of the insoluble fraction increased significantly the delphinidin content (Table S1).
Fermentation treatment significantly increased the extractability of the molecules in free and respectively bound form, respectively EPP and NEPP (measured by LC-MS), having a higher impact on the soluble fraction compared with the insoluble fractions (Fig. 3B). The PCA analysis (SI Fig. S1) also shows that the profile of the metabolites changes following the fermentation, especially in the molecules extractable in free form (EPP) from the soluble fraction. Furthermore, the profile of the molecules measured in the free and bound form (EPP and NEPP) from the soluble fractions (with and without fermentation) also is different as suggested by their distribution in different quadrants in the PCA plot (SI Fig. S1).
Microbiota modulatory effect of persimmon fractions
Microbiota modulatory properties of soluble and insoluble persimmon fibre fractions have been investigated. For this purpose, faecal fermentation experiments using healthy volunteers have been carried out. Microbial diversity of the faecal slurry was first characterised (SI Fig. S2–S4). Alpha diversity estimators expressed as Chao1 and Shannon coefficients measure the variability of microbial genera within faecal samples (SI Fig. S2). In general, donor 1 showed significantly (p < 0.05) lower alpha diversity values compared to the other donors. Beta diversity was calculated to compare microbial diversity among groups of samples (soluble and insoluble fractions at different fermentation times) and expressed as Bray Curtis dissimilarity distances showing no significant (p < 0.05) differences (SI Fig. S3). The absence of significant differences might be attributed to the interindividual variability in the fermentation profiles. In addition, PCoA revealed that samples corresponding to the same donor and same fermentation time were grouped together highlighting the role of interindividual variability in the microbiota composition profiles. PCoA analysis also discriminated different groups of substrates (SI Fig. S4). These results reveal that the fermentative properties of persimmon soluble and insoluble fractions may depend on the initial microbiota composition of each participant.To illustrate microbial differences between donors and substrates, microbiota composition bar plots were generated for initial and final fermentation times (SI Fig. S5). These plots highlighted major differences in the microbial abundances of several genera including Bacteroides, Prevotella and other commensals like Lachnoclostridium and Ruminococcus and novel taxon Ruminococcaceae UCG-02. These results indicate that the modulatory effect of the persimmon fractions were, at least, in part donor-driven.
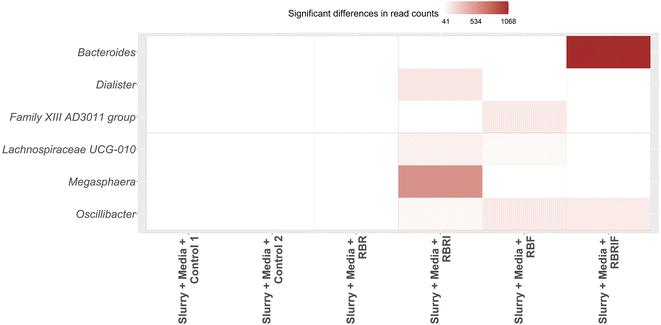
To assess these modulatory properties, statistically significant differences (p < 0.05 and padj < 0.25) in microbial genera composition after fermentation were calculated using several microbiota-specific statistical tests (ANCOM, ANCOMBC, ALDEx2, LEfSe and metagenomeSeq and Limma-Voom). A total of six microbial genera showed statistically significant increments (p < 0.05 and padj < 0.25) in their read counts and abundances after 24 h of faecal microbial fermentation of persimmon samples (Fig. 4). It should be noted that these taxa were not increased in control samples. The highest increments were observed for Bacteroides in the microbiota of donors 4 and 2. Interestingly, Bacteroides was selectively modulated by pretreated insoluble fibre fraction. Similarly, non-treated insoluble fractions selectively promoted Megasphaera in the microbiota of donor 4 (Fig. 4). On the other hand, pretreated soluble fraction enhanced the growth of Lachnospiraceae UCG-010 in all donors and Anaerovoraceae family XIII AD3011 group in donor 2. Less selective effects were observed for Oscillibacter, promoted in the microbiota of several donors after faecal fermentation of different substrates including pretreated soluble fraction and pretreated and non-treated insoluble fractions (Fig. 4).
A correlation network was generated to elucidate the potential interactions between microbial taxa modulated by persimmon fractions and other gut commensals (SI Fig. S6). Two main groups of bacteria were observed: (ii) Megasphaera showed positive correlations with Lachnospira and novel Clostridia taxa, (i) Anaerovoraceae family XIII AD3011 group and Lachnospiraceae UCG-010 showed positive correlations with Coprococcus, Eubacterium ruminatum group and Rikenellaceae RC9 groups. The first group of bacteria was more promoted by untreated insoluble fraction while the second group also modulated by soluble treated fraction.
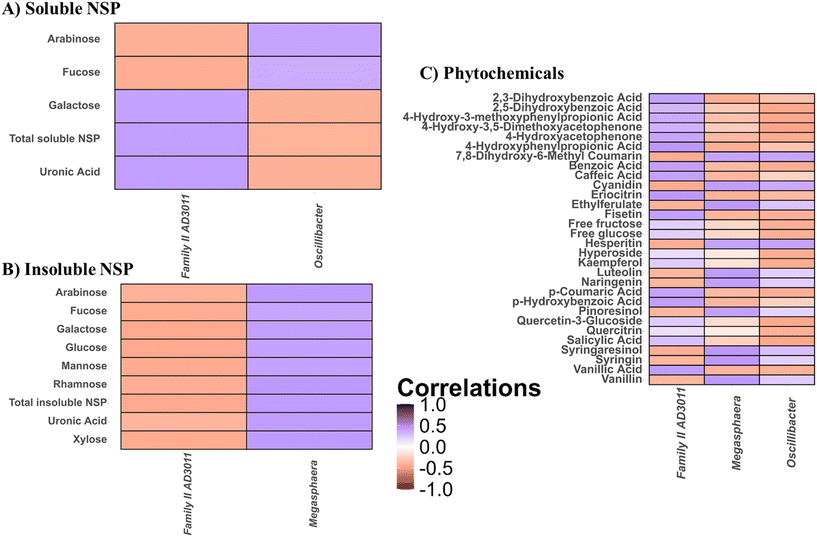
The effect of raw material composition on fermentative profiles of persimmon
To establish structure-activities of microbiota-modulating persimmon fractions, the effect of raw material composition on fermentative profiles of persimmon was investigated (Fig. 5). For this purpose, statistically significant (p < 0.05) correlations between soluble and insoluble NSPs composition and microbial genera modulated by persimmon were calculated (Fig. 5A and B). Oscillibacter showed positive associations with arabinose and fucose levels in the soluble NSP fraction and negative correlations with galactose and uronic acid (Fig. 5A). The opposite behaviour was observed for Anaerovoraceae family XIII AD3011 group. Similarly, Megasphaera and Anaerovoraceae family XIII AD3011 group showed positive and negative correlations with the insoluble NSP fraction, respectively (Fig. 5B).Similarly, associations between raw material phytochemical composition and microbial groups promoted by persimmon were determined (Fig. 5C). Anaerovoraceae family XIII AD3011 group showed positive correlations with a wide variety of phytochemicals including acetophenones (4-hydroxy-3,5-dimethoxyacetophenone), benzenes (4-hydroxyacetophenone), benzoic acids (salicylic acid, p-hydroxybenzoic acid, 2,3-dihydroxybenzoic acid, 2,5-dihydroxybenzoic acid, vanillic acid), cinnamic acids (caffeic acid, p-coumaric acid) and phenylpropionic acids (4-hydroxy-3-methoxyphenylpropionic acid, 4-hydroxyphenylpropionic acid), and negative with flavanoids/coumarins (hesperitin, luteolin, naringenin) and lignans (pinoresinol, syringaresinol). The opposite behaviour was observed for Megasphaera and Oscillibacter (Fig. 5C).
Microbial metabolite profiles
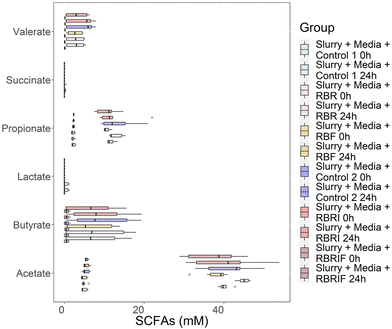
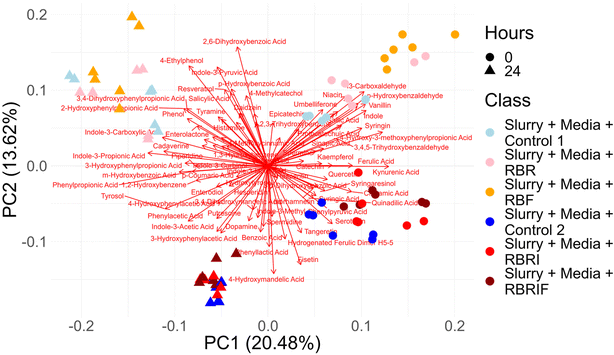
To further characterise the fermentative behaviour of persimmon samples, SCFAs production and the modification of phytochemicals were also investigated (Fig. 6 and 7). Acetate, butyrate, propionate and valerate levels significantly (p < 0.05) increased in all fermentation experiments at 24 h, showing no major differences between substrates (Fig. 6). In contrast, lactate increased only in untreated soluble fraction at 24 h. Interestingly, samples showing the highest increments in Megasphaera (red untreated insoluble fraction) also showed high increments in several SCFAs (butyrate, iso-butyrate and iso-valerate). In addition, statistical correlations were found between insoluble NSP found in the raw substrates and iso-butyrate and iso-valerate production (SI Fig. S7). Similarly, positive correlations between soluble NSP and lactate levels were also determined (SI Fig. S7).The profile of the microbial metabolites after faecal fermentation of persimmon substrates were also determined (Fig. 7 and SI Table S2). Principal Component Analysis (PCA) revealed a distinct separation between the metabolite profiles of persimmon substrates prior (0 h) and after 24 h of faecal fermentation, for both soluble and insoluble fractions. This segregation reflects substantial metabolic transformations induced by microbial activity. Furthermore, biplot analysis indicates that metabolites such as 4-hydroxymandelic acid and phenyl lactic acid, detected after 24 h of fermentation, primarily drive the separation of the insoluble fibre fraction. In contrast, compounds including 2-hydroxyphenylpropionic acid, 3,4-dihydroxyphenylpropionic acid, 4-ethylphenol, and indole-3-carboxylic acid contribute predominantly to the segregation of the soluble fibre fraction following microbial fermentation (Fig. 7). The Table S2 is showing the microbial metabolites composition of persimmon fractions after 24 h of faecal fermentation. Notable is the fact that the indole 3-propionic acid was produced in significant higher amounts following the faecal fermentation of the soluble persimmon fractions in comparison with the insoluble fractions (Table S2).
Discussion
This study successfully demonstrated microbiota modulatory properties of different persimmon by-product fractions from food industry. The treatment applied to obtain the persimmon fractions significantly influenced their carbohydrate composition, particularly the profile of NSP. As expected, all free glucose and fructose were depleted during fermentation, indicating rapid microbial utilisation of simple sugars. Interestingly, while the total quantity of NSPs remained unchanged, the monosaccharide composition was notably altered. Specifically, glucose levels increased while uronic acid decreased in both soluble and insoluble NSP fractions. Additionally, arabinose and xylose were reduced, whereas mannose and galactose increased in the insoluble NSP fraction.These compositional shifts are critical, as the structure and monosaccharide makeup of dietary fibre directly influence its fermentability and the resulting SCFA profile. Jensen et al.46 demonstrated that the monosaccharide content of dietary fibres, particularly glucose, xylose, and arabinose, can predict fermentation outcomes, including SCFA concentrations and microbial diversity. In our study, the altered NSP composition had a clear impact on SCFA production. Total soluble NSPs were positively associated with lactate levels, while arabinose and mannose contents of soluble NSPs correlated with increased iso-butyrate and iso-valerate (SI Fig. S7A) production. Similarly, total insoluble NSPs showed positive correlations with these branched-chain fatty acids (SI Fig. S7B).
These findings align with previous research showing that mannose-rich substrates promote the growth of Bifidobacterium and Faecalibacterium, and the latter is a major producer of butyrate.47 Moreover, arabinose and xylose, typically found in arabinoxylans, have been shown to support the production of propionate and butyrate, depending on their degree of branching and feruloylation.48
The positive association between soluble NSPs and lactate production is particularly noteworthy. Lactate is a key intermediate in microbial cross-feeding networks and can be converted into SCFAs such as propionate and butyrate by specialized bacteria, including Megasphaera species.49 The accumulation of lactate may indicate active fermentation by Lactobacillus and Bifidobacterium, which are known to thrive on soluble fibres and produce lactate as a primary metabolite.50
Comparative studies have shown that the fermentation of NSPs varies significantly depending on their source and structural complexity. For example, Wisker et al.51 reported that NSPs from citrus fibre and rye bread exhibited fermentation rates between 54% and 96%, with SCFA profiles influenced by particle size and solubility. Similarly, Zhang et al.52 found that structurally diverse soluble dietary fibres, including pectins and arabinogalactans, induced distinct microbial shifts and SCFA production patterns in the TIM-2 in vitro colon system.
The fermentation treatment applied to persimmon fibre-rich fractions had a pronounced impact also on the extractability and distribution of phytochemicals, significantly increasing both the extractable polyphenol (EPP) and non-extractable polyphenol (NEPP) content. This finding is particularly noteworthy, as fermentation is typically associated with enhanced release of soluble compounds due to microbial enzymatic activity, but an increase in NEPPs is less commonly reported and warrants further mechanistic exploration.
Fermentation is known to facilitate the breakdown of plant cell wall structures through microbial enzymatic action, including cellulases, pectinases, esterases, and glycosidases, which liberate bound phenolics from complex matrices.53,54 These enzymes can hydrolyse glycosidic bonds and ester linkages, releasing phenolic acids and flavonoids that were previously inaccessible. In our study, the most noticeable increases were observed in cyanidin, delphinidin, protocatechuic acid, and gallic acid in the soluble fraction, and cyanidin, delphinidin, gallic acid, and pelargonidin in the insoluble fraction. These compounds are known for their potent antioxidant and anti-inflammatory properties, with gallic acid and delphinidin particularly well-studied for their cardiovascular and metabolic health benefits.55,56
The unexpected increase in NEPPs may be explained by microbial biotransformation pathways that not only release but also rebind or polymerise phenolic intermediates during fermentation. Ding et al.57 highlighted that NEPPs can form through interactions with polysaccharides and proteins during microbial metabolism, resulting in new bound phenolic structures that are retained in the insoluble fraction. This phenomenon suggests that fermentation may not only liberate existing phenolics but also generate novel NEPPs through secondary binding or condensation reactions.
Moreover, fermentation has been shown to enhance the antioxidant potential of food matrices by increasing the concentration and diversity of phenolic compounds.58,59 In our study, the increase in bioactive compounds following fermentation suggests improved functional potential of the persimmon fractions. This is consistent with findings from Liu et al.,60 who reported that fermentation combined with enzymatic hydrolysis significantly increased polyphenol content and antioxidant activity in Astragalus membranaceus stems.
The implications of these findings are substantial. Enhanced extractability of polyphenols, particularly in both soluble and insoluble fractions, suggests that fermented persimmon by-products may offer dual-phase bioactivity: early release of soluble compounds in the upper gastrointestinal tract and delayed release of NEPPs in the colon, where they undergo microbial fermentation. This dual-phase release could improve systemic bioavailability and local gut health effects, supporting the development of next-generation prebiotics enriched with bioavailable bioactives.
Furthermore, anthocyanins such as cyanidin and delphinidin, and phenolic acids like gallic and protocatechuic acid, have demonstrated beneficial effects on endothelial function, oxidative stress, and inflammation in human studies.61,62 Their increased presence in fermented persimmon fractions reinforces the potential of this treatment to enhance the health-promoting properties of agri-food by-products.
The microbiota modulatory properties of persimmon fractions have also been investigated. The highest microbial increments were observed for the Bacteroides genus, which was selectively modulated by pre-treated insoluble fibre fractions. Bacteroides in one of the major generalist commensals and potentially beneficial bacterial groups tracked while evaluating the prebiotic in previous in vitro models.63 These in vitro fermentation studies report the ability of fruit and vegetable-derived prebiotics to modulate the growth of Bacteroides such as pectin, modified pectins and pectic oligosaccharides from orange peel wastes,64 and other renewable bioresources including pectin-rich artichoke and sunflower by-products.65 In this regard, some Bacteroides species have been proposed as next-generation probiotics. Previous studies reporting genome mining of probiotic candidates highlight the metabolic capabilities of B. dorei, B. ovatus, B. thetaiotaomicron, B. uniformis and B. xylanisolvens to utilize all vegetable carbohydrates (xylan, arabinoxylan, pectin and their derived oligosaccharides). Bacteroides species tend to have larger genomes than many other gut bacterial species and are considered as generalists, showing the widest range of activities such as polygalacturonases, rhamnosidases, rhamnogalacturonases, pectin lyases and pectin methyl- and acetyl esterases compared to other potentially probiotic gut commensals.63
The ability of persimmon fractions to promote Bacteroides species was reported previously in both single and mixed culture experiments.16 Single culture experiments of B. thetaiotaomicron B5482 revealed similar growth rates with persimmon fractions and glucose resulting in the production of formate, acetate, butyrate, and lactate. However, mixed culture experiments using several of strains belonging to the Bacteroidetes and Firmicutes phyla resulted in significantly (p < 0.001) lower growth rates. The present study describes the behaviour of Bacteroides and other key symbionts in the presence of persimmon fractions, in the context of their natural communities.
The level of Shigella–Escherichia after 24 h incubation is an unfavourable outcome (Fig. S4). This is to some extent is donor dependent and will depend on the composition of each donor's microbiota. One of the donors, Donor 3, was shown to have an increase in proportional abundance of Shigella–Escherichia after 24 h incubation. This in part may come from metabolism of the peptides in the growth medium, as this bacterial group can grow well on peptides, using these as a carbon and energy source. Other unfavourable bacterial genera however, such as Sutterella however declined in proportional abundance.
Many bacterial generalist groups that can utilise simple sugars and oligosaccharides as growth substrates may depend on cross feeding following the release of soluble sugars or on metabolism of short chain fatty acids. Interestingly, beneficial bacteria including Parabacteroides, Acidaminococcus and Blautia increased in proportional abundance.
Lactobacillus species are predominantly present in the small intestine, so it is not surprising that this genus was absent. Bifidobacteria are usually present in low abundance in adults often less than 5% of total microbiota and are not key players in fibre metabolism, therefore not surprising that proportional abundance was low. Interestingly, key beneficial bacterial genera belonging to the Lachnospiraceae family, in addition to Faecalibacterium were largely maintained following incubations with the persimmon fibres.
Other potentially probiotic bacteria selectively promoted by persimmon fractions include Megasphaera, a known as a lactate utiliser, converting lactate to acetate and propionate,66 and under carbon-limited conditions can convert lactate to acetate and butyrate. This organism can establish metabolic cross-feeding interactions to convert acetate or lactate produced by other fibre-degrading gut commensals to butyrate.67
Similarly, Oscillibacter promoted by different persimmon fractions has been proposed as a next-generation probiotic capable of producing SCFAs such as butyrate and may exert positive health effects including anti-inflammatory activities.68,69 The other fibre-degrading organisms modulated by persimmon fractions included Lachnospira members that could be of special interest for the development of novel probiotics or biotherapies.70 Previous studies suggest that daily supplementation with rhamnogalacturonan-I modulates the growth of Lachnospiraceae UCG-010 on the gut microbiota in healthy adults and correlates with reduced common cold symptoms.71 Previous faecal fermentation experiments reported that Lachnospiraceae UCG-010 was selectively modulated by apple pomace and pectin fractions.72 Similarly, increments in Dialister were observed after faecal fermentation of persimmon fractions. In this regard, human intervention studies revealed that whole-grain barley promoted the growth of Dialister.73 This taxon was also promoted by apple pomace and pectin after faecal fermentation in vitro.72 Increases of Dialister have been reported during prebiotic supplementation.74,75 Similarly, members of the Anaerovoraceae family are known to produce SCFA, and experience increased growth when participants consume fermented foods or prebiotic ingredients.76 Several statistical associations between these bacteria were also determined highlighting metabolic synergies. It should be noted that cross-feeding interactions between SCFAs-producing gut anaerobes have been widely reported.77
Cross-feeding interactions between Megasphaera and other gut commensals have been reported.78 In this regard, Megasphaera converts lactate into butyrate and valerate in agreement with the results from our correlation analysis. In the current study, our results also highlight positive associations between Lachnospiraceae UCG-010 and Dialister and SCFAs levels. Bacterial species belonging to both these genera can form lactate as a fermentation end products which may be used by cross feeding Megasphaera and as shown here all genera increased in abundance, and in particular Megasphaera, on the RBRIF substrate (Fig. 4). It has previously been reported that Lachnospira members produce SCFA as a product of the fermentation of complex polysaccharides.70,79 The influence of Dialister on SCFA production has also been reported.80,81
Structure–activity-relationships determined for persimmon substrates revealed that soluble NSP fractions rich in arabinose and fucose stimulate the growth of Oscillibacter while insoluble NSP fractions promote Megasphaera. The role of soluble and insoluble fibre on microbial fermentation has been extensively studied.82 The ability of different NSPs structures to promote the growth of different microbial groups including Bacteroides, Lachnospiraceae, Oscillibacter, Megasphaera and Dialister, has been reported.83,84
Previous studies reported the ability of phenolic compounds to promote gut microbiota diversity and maintain colonic health.85 A few bacterial genera involved in the metabolism of phenolic compounds have been identified and their degradative pathways elucidated.85,86 Medium doses of persimmon tannin promote Bacteroides,87 while proanthocyanidins extracts led to an increase in Dialister.88 Other studies dealing with different substrates highlight the potential of orange juice and chenpi tea rich in flavonoids to promote Lachnospira and Oscillibacter, respectively.89,90 Similarly, it has been reported that several gut commensals including Megasphaera, Dialister and Lachnospiraceae members might contribute to polyphenol liberation and metabolism in fermented quinoa.91 Results here presented agree with those reported by previous studies. In this regard, tea phenolics rich in benzoic acids may inhibit the growth of Bacteroides in the gut microbiota,92 in agreement with our correlation analysis. Phenylacetic acid is a metabolic product of Bacteroides and is formed from the metabolism of the amino acid phenylalanine, but its function remains poorly understood.93 In contrast, caffeine may decrease the relative abundance of Bacteroides.94
Comparative studies on fibre types indicate that soluble fibres generally promote the production of beneficial metabolites, including indole 3-propionic acid (IPA), indolelactic acid, and short-chain fatty acids, whereas insoluble fibres primarily contribute to stool bulking and transit time with limited fermentability.95,96 Recent work by Sinha et al.97 demonstrated that fermentable fibres enhance IPA production by influencing microbial competition for tryptophan, favouring pathways that yield protective indole derivatives over potentially harmful ones like indole itself. This mechanistic insight underscores the importance of soluble fibre-rich diets in shaping a metabolome conducive to health. Furthermore, has been associated with multiple health-promoting effects. It acts as a potent antioxidant, surpassing melatonin in hydroxyl radical scavenging capacity, and protects against oxidative stress without generating pro-oxidant intermediates.98,99 IPA also contributes to gut barrier integrity by upregulating tight junction proteins and modulating immune responses, thereby reducing systemic inflammation.99 Moreover, IPA has demonstrated neuroprotective properties, including inhibition of β-amyloid fibril formation and attenuation of neuronal damage in ischemic models, suggesting a role in mitigating neurodegenerative disorders such as Alzheimer's disease.100,101
Conclusions
The study findings suggest that the structural modification of persimmon NSPs through processing not only alters their monosaccharide composition but also modulates their fermentability and metabolic output. These changes have implications for the development of functional ingredients targeting gut health, particularly through the selective stimulation of SCFA-producing and anti-inflammatory microbial taxa.We observed an increase in both EPP and NEPP content following fermentation, which is likely due to a combination of enzymatic release, microbial biotransformation, and secondary binding reactions. These findings validate the use of fermentation as a strategic tool to enhance the nutritional and functional value of persimmon by-products and support their application in functional food development targeting metabolic and gut health.
The microbiota modulatory properties of persimmon soluble and insoluble fibre fractions have been determined. Pretreated insoluble fraction selectively promoted the growth of Bacteroides while non-treated insoluble fraction stimulated the growth of Megasphaera. On the other hand, pretreated soluble fraction enhanced the growth of Lachnospiraceae UCG-010 and Anaerovoraceae family XIII AD3011. Soluble and insoluble fractions modulated the growth of Oscillibacter. Some of these taxa include important SCFA producers and next-generation probiotics that showed a wide range of statistical associations with other members of the microbiota. In addition, structure–activity relationships for persimmon fractions have been determined. In this regard, arabinose and fucose-rich soluble NSPs promote Oscillibacter while insoluble NSP stimulate the growth of Megasphaera. Similarly, phenolic compound composition exerted a great influence on the fermentative properties of persimmon fractions highlighting the role of flavonoids, benzoic and phenylacetic acids. Persimmon by-products are a promising source of potentially prebiotic substrates that could be of special interest to design novel prebiotic formulations with enhanced selectivity targeting next-generation probiotics.
The present study demonstrates also that faecal fermentation of soluble persimmon fractions leads to significantly higher production of indole-3-propionic acid (IPA) compared to insoluble fractions. Given IPA's established antioxidant, anti-inflammatory, and neuroprotective properties, this finding underscores the potential health benefits of incorporating soluble fibre-rich persimmon by-products into the diet. These results align with previous evidence that soluble fibres are more fermentable and capable of generating bioactive metabolites that support gut and systemic health. Future research should explore the mechanistic pathways linking soluble fibre fermentation to IPA biosynthesis and evaluate its implications for metabolic and neurodegenerative disease prevention.
Author contributions
Conceptualisation, N. M. B., S. H. D., M. N. and F. M.; methodology, L. L.-B., B. M.-C., J. S.-B., N. J. H., A. M., G. J. D., G. B. and B. E.-L.; validation, W. R. R., N. M. B., S. H. D., M. N. and F. M.; formal analysis, C. S., L. L.-B., B. M.-C., B. E.-L., G. B., N. M. B., S. H. D., M. N. and F. M.; investigation, C. S., L. L.-B., B. M.-C., J. S.-B., N. J. H., A. M., G. J. D. and B. E.-L.; software, C. S.; resources, M. N. and F. M.; data curation, C. S., L. L.-B., G. B., S. H. D., M. N. and F. M.; writing – original draft preparation, L. L.-B., N. M. B., S. H. D., M. N., C. S. and F. M.; visualization, N. M. B., S. H. D., M. N., C. S. and F. M.; supervision, M. N. and F. M.; project administration, F. M.; funding acquisition, F. M. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare that there is no conflict of interest.Data availability
Raw sequences generated have been deposited in the SRA. Accession number has been incorporated in the material and methods section (PRJNA1266485).Supplementary information (SI) is available. Supplementary Tables and Figures showing phytochemical composition content and microbial metabolites composition of persimmon fractions after 24 h of faecal fermentation, and several microbial diversity indicators and correlation plots. See DOI: https://doi.org/10.1039/d5fo03495g.
Acknowledgements
Carlos Sabater is funded by UKRI Engineering and Physical Sciences Research Council (EP/Z001064/1). This research was funded by the Spanish Ministerio de Ciencia e Innovación (grant number PID2020-116731RB-C21 to F. M.). LL-B was a recipient of an FPU contract (FPU19/04497) from the Ministerio de Educación y Formación Profesional (Spain). LL-B's stay at The Rowett Institute was supported by a mobility fellowship from the Training Program of the Centro de Investigación Biomédica en Red de Diabetes y Enfermedades Metabólicas Asociadas (CIBERDEM), which is funded by the Instituto de Salud Carlos III (Spain). The Rowett Institute, University of Aberdeen co-authors acknowledge support from the Scottish Government Rural and Environmental Sciences and Analytical Services Division (RESAS) Strategic Research Programme (projects RI-B1-01 and RI-B5-03). For the purpose of open access, the authors have applied a Creative Commons Attribution (CC BY) licence to any Author Accepted Manuscript version arising from this submission.References
- S. Pérez-Burillo, M. J. Oliveras, J. Quesada, J. A. Rufián-Henares and S. Pastoriza, Relationship between composition and bioactivity of persimmon and kiwifruit, Food Res. Int., 2018, 105, 461–472, DOI:10.1016/j.foodres.2017.11.022.
- Food and Agriculture Organization (FAO), FAOSTAT Statistical Database, Available online: https://www.fao.org/faostat/es/#home (accessed on 10 November 2025).
- Food and Agriculture Organization (FAO), Global Food Loss and Waste Facts, Available online: https://www.fao.org/platform-food-loss-waste/flw-events/events/events-detail/2024-food-loss-and-waste-masterclass–the-basics/en (accessed on 10 November 2025).
- C. Conesa, N. Laguarda-Miró, P. Fito and L. Seguí, Evaluation of Persimmon (Diospyros kaki Thunb. cv. Rojo Brillante) industrial residue as a source for value added products. Waste and biomass valorization, Waste Biomass Valorization, 2020, 11, 3749–3760, DOI:10.1007/s12649-019-00621-0.
- J. Salazar-Bermeo, B. Moreno-Chamba, M. C. Martínez-Madrid, D. Saura, M. Valero and N. Martí, Potential of persimmon dietary fiber obtained from byproducts as antioxidant, prebiotic and modulating agent of the intestinal epithelial barrier function, Antioxidants, 2021, 10, 1668, DOI:10.3390/antiox10111668.
- S. Yaqub, U. Farooq, A. Shafi, K. Akram, M. A. Murtaza, T. Kausar and F. Siddique, Chemistry and functionality of bioactive compounds present in persimmon, J. Chem., 2016, 2016, 3424025, DOI:10.1155/2016/3424025.
- A. M. Stephen, M. M. Champ, S. J. Cloran, M. Fleith, L. V. Lieshout, H. Mejborn and V. J. Burley, Dietary fibre in Europe: current state of knowledge on definitions, sources, recommendations, intakes and relationships to health, Nutr. Res. Rev., 2017, 30, 149–190, DOI:10.1017/S095442241700004X.
- D. E. Threapleton, D. C. Greenwood, C. E. L. Evans, C. L. Cleghorn, C. Nykjaer, C. Woodhead, J. E. Cade, C. P. Gale and V. J. Burley, Dietary fibre intake and risk of cardiovascular disease: systematic review and meta-analysis, Br. Med. J., 2013, 347, f6879, DOI:10.1136/bmj.f6879.
- A. Reynolds, J. Mann, J. Cummings, N. Winter, E. Mete and L. T. Morenga, Carbohydrate quality and human health: a series of systematic reviews and meta-analyses, Lancet, 2019, 393, 434–445, DOI:10.1016/S0140-6736(18)31809-9.
- S. H. Duncan, E. Conti, L. Ricci and A. W. Walker, Links between diet, intestinal anaerobes, microbial metabolites and health, Biomedicines, 2023, 11, 1338, DOI:10.3390/biomedicines11051338.
- US Department of Agriculture and US Department of Health and Human Services, Dietary guidelines for Americans, 2020–2025, Dietary Guidelines for Americans website, Available online: https://www.dietaryguidelines.gov (accessed on 10 November 2025).
- EFSA Panel on Dietetic Products, Nutrition, and and Allergies (NDA), Scientific opinion on dietary reference values for carbohydrates and dietary fibre, EFSA J., 2010, 8(3) DOI:10.2903/j.efsa.2010.1462.
- EUFIC, Recommended daily intake of fibre and fibre-rich foods to help you achieve it, Available online: https://www.eufic.org/en/?ACT=115&path=global/pdf/recommended-daily-intake-of-fibre-and-fibre-rich-foods-to-help-you-achieve-it (accessed on 10 November 2025).
- S. Facchin, L. Bertin, E. Bonazzi, G. Lorenzon, C. De Barba, B. Barberio, F. Zingone, D. Maniero, M. Scarpa, C. Ruffolo, I. Angriman and E. V. Savarino, Short-chain fatty acids and human health: from metabolic pathways to current therapeutic implications, Life, 2024, 14, 559, DOI:10.3390/life14050559.
- B. J. Kesler, Emergent science on fiber-bound polyphenols and the gut microbiome. Naturally Informed Conference Presentation, 2025, Available online: https://naturallyinformed.net/wp-content/uploads/2025/06/Brendan-Presentation-Emergent-Science-on-Fiber-Bound-Polyphenols-1.pdf (accessed on 10 November 2025).
- L. López-Bermudo, B. Moreno-Chamba, J. Salazar-Bermeo, N. J. Hayward, A. Morris and G. J. Duncan, et al., Persimmon Fiber-Rich Ingredients Promote Anti-Inflammatory Responses and the Growth of Beneficial Anti-Inflammatory Firmicutes Species from the Human Colon, Nutrients, 2024, 16, 2518, DOI:10.3390/nu16152518.
- V. Sharma, L. Devkota, N. Kishore and S. Dhital, Understanding the interplay between dietary fiber, polyphenols, and digestive enzymes, Food Hydrocolloids, 2025, 166, 111310, DOI:10.1016/j.foodhyd.2025.111310.
- C. A. Edwards, J. Havlik, W. Cong, W. Mullen, T. Preston, D. J. Morrison and E. Combet, Polyphenols and health: Interactions between fibre, plant polyphenols and the gut microbiota, Nutr. Bull., 2017, 42, 356–360, DOI:10.1111/nbu.12296.
- F. Meiners, A. Ortega-Matienzo, G. Fuellen and I. Barrantes, Gut microbiome-mediated health effects of fiber and polyphenol-rich dietary interventions, Front. Nutr., 2025, 12, 1647740, DOI:10.3389/fnut.2025.1647740.
- W. R. Russell, M. J. Burkitt, L. Scobbie and A. Chesson, Radical formation and coupling of hydroxycinnamic acids containing 1,2-dihydroxy substituents, Bioorg. Chem., 2003, 31, 206–215, DOI:10.1016/S0045-2068(03)00042-7.
- W. R. Russell, A. R. Forrester, A. Chesson and M. J. Burkitt, Oxidative Coupling during Lignin Polymerization Is Determined by Unpaired Electron Delocalization within Parent Phenylpropanoid Radicals, Arch. Biochem. Biophys., 1996, 332, 357–366, DOI:10.1006/abbi.1996.0353.
- B. Moreno-Chamba, L. López-Bermudo, G. Lo, J. Salazar-Bermeo, N. Hayward and A. Morris, et al., Efficacy of pre-treatment of insoluble fibres derived from persimmon (Diospyros kaki) fruit by-products to promote growth of beneficial human colonic bacteria, Food Biosci., 2025, 68, 106737, DOI:10.1016/j.fbio.2025.106737.
- B. Moreno-Chamba, J. Salazar-Bermeo, M. C. Martínez-Madrid, V. Lizama, F. Martín-Bermudo, G. Berná, M. Neacsu, D. Saura, N. Martí and M. Valero, Bound galloylated compounds in persimmon upcycled dietary fiber modulate microbial strains associated to human health after in vitro digestion, LWT–Food Sci. Technol., 2022, 156, 113011, DOI:10.1016/j.lwt.2021.113011.
- H. N. Englyst, S. M. Kingman and J. H. Cummings, Classification and measurement of nutritionally important starch fractions, Eur. J. Clin. Nutr., 1992, 46, 33 Search PubMed.
- M. Neacsu, J. McMonagle, R. J. Fletcher, L. Scobbie, G. J. Duncan and L. Cantlay, et al., Bound phytophenols from ready-to-eat cereals: comparison with other plant-based foods, Food Chem., 2013, 141, 2880–2886, DOI:10.1016/j.foodchem.2013.05.023.
- M. Neacsu, N. Vaughan, V. Raikos, S. Multari, G. J. Duncan and G. G. Duthie, et al., Phytochemical profile of commercially available food plant powders: their potential role in healthier food reformulations, Food Chem., 2015, 179, 159–169, DOI:10.1016/j.foodchem.2015.01.128.
- W. R. Russell and S. H. Duncan, Advanced analytical methodologies to study the microbial metabolome of the human gut, TrAC, Trends Anal. Chem., 2013, 52, 54–60, DOI:10.1016/j.trac.2013.08.004.
- Z. Zhang, X. Kou, K. Fugal and J. McLaughlin, Comparison of HPLC methods for determination of anthocyanins and anthocyanidins in bilberry extracts, J. Agric. Food Chem., 2004, 52, 688–691, DOI:10.1021/jf034596w.
- Z. Zhang, S. Fan, G. J. Duncan, A. Morris, D. Henderson, P. Morrice, W. R. Russell, S. H. Duncan and M. Neacsu, Buckwheat (Fagopyrum esculentum) Hulls Are a Rich Source of Fermentable Dietary Fibre and Bioactive Phytochemicals, Int. J. Mol. Sci., 2023, 24, 16310, DOI:10.3390/ijms242216310.
- A. J. Richardson, A. G. Calder, C. S. Stewart and A. Smith, Simultaneous determination of volatile and non–volatile acidic fermentation products of anaerobes by capillary gas chromatography, Lett. Appl. Microbiol., 1989, 9, 5–8, DOI:10.1111/j.1472-765X.1989.tb00278.x.
- E. Bolyen, J. R. Rideout, M. R. Dillon, N. A. Bokulich, C. C. Abnet and G. A. Al-Ghalith, et al., Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2, Nat. Biotechnol., 2019, 37, 852–857, DOI:10.1038/s41587-019-0209-9.
- C. Quast, E. Pruesse, P. Yilmaz, J. Gerken, T. Schweer and P. Yarza, et al., The SILVA ribosomal RNA gene database project: improved data processing and web-based tools, Nucleic Acids Res., 2013, 41, 590, DOI:10.1093/nar/gks1219.
- F. O. Glöckner, P. Yilmaz, C. Quast, J. Gerken, A. Beccati and A. Ciuprina, et al., 25 years of serving the community with ribosomal RNA gene reference databases and tools, J. Biotechnol., 2017, 261, 169–176, DOI:10.1016/j.jbiotec.2017.06.1198.
- P. Yilmaz, L. W. Parfrey, P. Yarza, J. Gerken, E. Pruesse and C. Quast, et al., The SILVA and “All-species Living Tree Project (LTP)” taxonomic frameworks, Nucleic Acids Res., 2014, 42, 643, DOI:10.1093/nar/gkt1209.
- L. Lahti and S. Shetty, microbiome R package, 2019 Search PubMed.
- P. J. McMurdie and S. Holmes, phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data, PLoS One, 2013, 8, e61217, DOI:10.1371/journal.pone.0061217.
- Y. Cao, Q. Dong, D. Wang, P. Zhang, Y. Liu and C. Niu, microbiomeMarker: an R/Bioconductor package for microbiome marker identification and visualization, Bioinformatics, 2022, 38, 4027–4029, DOI:10.1093/bioinformatics/btac438.
- H. Lin and S. D. Peddada, Analysis of compositions of microbiomes with bias correction, Nat. Commun., 2020, 11, 3514, DOI:10.1038/s41467-020-17041-7.
- S. Mandal, W. Van Treuren, R. A. White, M. Eggesbø, R. Knight and S. D. Peddada, Analysis of composition of microbiomes: a novel method for studying microbial composition, Microb. Ecol. Health Dis., 2015, 26, 27663, DOI:10.3402/mehd.v26.27663.
- A. D. Fernandes, J. N. Reid, J. M. Macklaim, T. A. McMurrough, D. R. Edgell and G. B. Gloor, Unifying the analysis of high-throughput sequencing datasets: characterizing RNA-seq, 16S rRNA gene sequencing and selective growth experiments by compositional data analysis, Microbiome, 2014, 2, 15, DOI:10.1186/2049-2618-2-15.
- C. W. Law, Y. Chen, W. Shi and G. K. Smyth, voom: precision weights unlock linear model analysis tools for RNA-seq read counts, Genome Biol., 2014, 15, R29, DOI:10.1186/gb-2014-15-2-r29.
- J. N. Paulson, O. C. Stine, H. C. Bravo and M. Pop, Differential abundance analysis for microbial marker-gene surveys, Nat. Methods, 2013, 10, 1200–1202, DOI:10.1038/nmeth.2658.
- N. Segata, J. Izard, L. Waldron, D. Gevers, L. Miropolsky and W. S. Garrett, et al., Metagenomic biomarker discovery and explanation, Genome Biol., 2011, 12, R60, DOI:10.1186/gb-2011-12-6-r60.
- M. Kuhn, S. Jackson and J. Cimentada, corrr R package, 2022 Search PubMed.
- F. Rohart, B. Gautier, A. Singh and K. L. Cao, mixOmics: An R package for ‘omics feature selection and multiple data integration, PLoS Comput. Biol., 2017, 13, e1005752, DOI:10.1371/journal.pcbi.1005752.
- N. Jensen, M. Maldonado-Gomez, N. Krishnakumar, C. Weng, J. Castillo, D. Razi, K. Kalanetra, J. B. German, C. B. Lebrilla, D. A. Mills and D. H. Taft, Dietary fiber monosaccharide content alters gut microbiome composition and fermentation, Appl. Environ. Microbiol., 2024, 90, e0096424, DOI:10.1128/aem.00964-24.
- L. K. Leung, L. S. Po, T. Y. Lau and Y. M. Yuen, Br. J. Nutr., 2004, 91, 831–839, DOI:10.1079/bjn20041122.
- Z. Li, H. Zhang, L. He, Y. Hou, Y. Che, T. Liu, S. Xiong, X. Zhang, S. Luo, C. Liu and T. Chen, Influence of structural features and feruloylation on fermentability and ability to modulate gut microbiota of arabinoxylan in in vitro fermentation, Front. Microbiol., 2023, 13, 1113601, DOI:10.3389/fmicb.2022.1113601.
- P. Louis, S. H. Duncan, P. O. Sheridan, A. W. Walker and H. J. Flint, Microbial lactate utilisation and the stability of the gut microbiome, Gut Microbiome, 2022, 3, e3, DOI:10.1017/gmb.2022.3.
- M. Calatayud, P. Van den Abbeele, J. Ghyselinck, M. Marzorati, E. Rohs and A. Birkett, Comparative effect of 22 dietary sources of fiber on gut microbiota of healthy humans in vitro, Front. Nutr., 2021, 8, 700571, DOI:10.3389/fnut.2021.700571.
- E. Wisker, M. Daniel, G. Rave and W. Feldheim, Fermentation of non-starch polysaccharides in mixed diets and single fibre sources: comparative studies in human subjects and in vitro, Br. J. Nutr., 1998, 80, 253–261, DOI:10.1017/S0007114598001305.
- Y. Zhang, N. Belkassem, A. Umanets, A. P. de Santana-Filho, K. Venema, H. A. Schols, J. Penders, D. Jonkers and K. Y. Abboud, A head-to-head comparison of soluble dietary fibers on microbiota composition and short-chain fatty acid production using the in vitro TIM-2 system, Carbohydr. Polym., 2025, 368, 124090, DOI:10.1016/j.carbpol.2025.124090.
- R. Sharma, B. Diwan, B. P. Singh and S. Kulshrestha, Probiotic fermentation of polyphenols: Potential sources of novel functional foods, Food Prod., Process. Nutr., 2022, 4, 21, DOI:10.1186/s43014-022-00101-4.
- A. Kumar, S. Saranyadevi, S. K. Thirumalaisamy, T. T. D. Durage, S. G. Jaiswal, D. Kavitake and S. Wei, Phenolic acids in fermented foods: microbial biotransformation, antioxidant mechanisms, and functional health implications, Front. Mol. Biosci., 2025, 12, 1678673, DOI:10.3389/fmolb.2025.1678673.
- K. Goszcz, S. J. Deakin, G. G. Duthie, D. Stewart and I. L. Megson, Bioavailable concentrations of delphinidin and its metabolite, gallic acid, induce antioxidant protection associated with increased intracellular glutathione in cultured endothelial cells, Oxid. Med. Cell. Longevity, 2017, 2017, 9260701, DOI:10.1155/2017/9260701.
- A. Husain, H. Chanana, S. A. Khan, U. M. Dhanalekshmi, M. Ali, A. A. Alghamdi and A. Ahmad, Chemistry and pharmacological actions of delphinidin, a dietary purple pigment in anthocyanidin and anthocyanin forms, Front. Nutr., 2022, 9, 746881, DOI:10.3389/fnut.2022.746881.
- Y. Ding, K. Morozova, M. Scampicchio and G. Ferrentino, Non-Extractable polyphenols from food by-products: Current knowledge on recovery, characterisation, and potential applications, Processes, 2020, 8, 925, DOI:10.3390/pr8080925.
- S. Sarıtaş, A. C. M. Portocarrero, J. M. M. López, M. Lombardo, W. Koch, A. Raposo, H. R. El-Seedi, J. L. de Brito Alves, T. Esatbeyoglu, S. Karav and A. M. Witkowska, The impact of fermentation on the antioxidant activity of food products, Molecules, 2024, 29, 3941, DOI:10.3390/molecules29163941.
- R. Gan, N. P. Shah, M. Wang, W. Lui and H. Corke, Fermentation alters antioxidant capacity and polyphenol distribution in selected edible legumes, Int. J. Food Sci. Technol., 2016, 51, 875–884, DOI:10.1111/ijfs.13062.
- N. Liu, X. An, Y. Wang, J. Qi, Y. Jia, X. Li, Z. Zhang and X. Chen, Polyphenol composition and antioxidant activity of fermentation combined with enzymatic hydrolysis modified Astragalus membranaceus stems, Chem. Biol. Technol. Agric., 2024, 11, 153, DOI:10.1186/s40538-024-00674-x.
- A. Liang, W. Leonard, J. T. Beasley, Z. Fang, P. Zhang and C. S. Ranadheera, Anthocyanins-gut microbiota-health axis: A review, Crit. Rev. Food Sci. Nutr., 2024, 64, 7563–7588, DOI:10.1080/10408398.2023.2187212.
- A. Rauf, A. Olatunde, R. Ajaj, N. Hafeez, M. Y. Zaky, M. R. Islam, M. M. Islam and G. Ribaudo, The Medicinal Chemistry of Cyanidin and its Glycoside Derivatives: Focus on the Antiproliferative and Potential Anticancer Activity, Planta Med., 2025, 91, 635–644, DOI:10.1055/a-2634-4300.
- C. Sabater, I. Calvete-Torre, M. Villamiel, F. J. Moreno, A. Margolles and L. Ruiz, Vegetable waste and by-products to feed a healthy gut microbiota: Current evidence, machine learning and computational tools to design novel microbiome-targeted foods, Trends Food Sci. Technol., 2021, 118, 399–417, DOI:10.1016/j.tifs.2021.10.002.
- B. Gómez, B. Gullón, C. Remoroza, H. A. Schols, J. C. Parajó and J. L. Alonso, Purification, characterization, and prebiotic properties of pectic oligosaccharides from orange peel wastes, J. Agric. Food Chem., 2014, 62, 9769–9782, DOI:10.1021/jf503475b.
- A. Ferreira-Lazarte, V. Kachrimanidou, M. Villamiel, R. A. Rastall and F. J. Moreno, In vitro fermentation properties of pectins and enzymatic-modified pectins obtained from different renewable bioresources, Carbohydr. Polym., 2018, 199, 482–491, DOI:10.1016/j.carbpol.2018.07.041.
- R. Prabhu, E. Altman and M. A. Eiteman, Lactate and acrylate metabolism by Megasphaera elsdenii under batch and steady-state conditions, Appl. Environ. Microbiol., 2012, 78, 8564–8570, DOI:10.1128/AEM.02443-12.
- E. Mutuyemungu, H. A. Motta-Romero, Q. Yang, S. Liu, S. Liu and M. Singh, et al., Megasphaera elsdenii, a commensal member of the gut microbiota, is associated with elevated gas production during in vitro fermentation, Gut Microbiome, 2024, 5, e1, DOI:10.1017/gmb.2023.18.
- U. Gophna, T. Konikoff and H. B. Nielsen, Oscillospira and related bacteria - From metagenomic species to metabolic features, Environ. Microbiol., 2017, 19, 835–841, DOI:10.1111/1462-2920.13658.
- J. Yang, Y. Li, Z. Wen, W. Liu, L. Meng and H. Huang, Oscillospira - a candidate for the next-generation probiotics, Gut Microbes, 2021, 13, 1987783, DOI:10.1080/19490976.2021.1987783.
- R. Abdugheni, W. Wang, Y. Wang, M. Du, F. Liu and N. Zhou, et al., Metabolite profiling of human-originated Lachnospiraceae at the strain level, iMeta, 2022, 1, e58, DOI:10.1002/imt2.58.
- C. Jian, N. Sorensen, R. Lutter, R. Albers, W. de Vos and A. Salonen, et al., The impact of daily supplementation with rhamnogalacturonan-I on the gut microbiota in healthy adults: A randomized controlled trial, Biomed. Pharmacother., 2024, 174, 116561, DOI:10.1016/j.biopha.2024.116561.
- I. Calvete-Torre, C. Sabater, M. J. Antón, F. J. Moreno, S. Riestra and A. Margolles, et al., Prebiotic potential of apple pomace and pectins from different apple varieties: Modulatory effects on key target commensal microbial populations, Food Hydrocoll., 2022, 133, 107958, DOI:10.1016/j.foodhyd.2022.107958.
- I. Martínez, J. M. Lattimer, K. L. Hubach, J. A. Case, J. Yang and C. G. Weber, et al., Gut microbiome composition is linked to whole grain-induced immunological improvements, ISME J., 2013, 7, 269–280, DOI:10.1038/ismej.2012.104.
- R. Xiao, W. Liao, G. Luo, Z. Qin, S. Han and Y. Lin, Modulation of Gut Microbiota Composition and Short-Chain Fatty Acid Synthesis by Mogroside V in an In Vitro Incubation System, ACS Omega, 2021, 6, 25486–25496, DOI:10.1021/acsomega.1c03485.
- J. Mou, Q. Li, W. Shi, X. Qi, W. Song and J. Yang, Chain conformation, physicochemical properties of fucosylated chondroitin sulfate from sea cucumber Stichopus chloronotus and its in vitro fermentation by human gut microbiota, Carbohydr. Polym., 2020, 228, 115359, DOI:10.1016/j.carbpol.2019.115359.
- B. Bhaskar, A. Bhattacharya, A. Adak, S. Das and M. R. Khan, A Human and Animal Based Study Reveals That a Traditionally Fermented Rice Beverage Alters Gut Microbiota and Fecal Metabolites for Better Gut Health, Fermentation, 2023, 9, 126, DOI:10.3390/fermentation9020126.
- M. Vacca, G. Celano, F. M. Calabrese, P. Portincasa, M. Gobbetti and M. De Angelis, The Controversial Role of Human Gut Lachnospiraceae, Microorganisms, 2020, 8, 573, DOI:10.3390/microorganisms8040573.
- S. Zhao, R. Lau, Y. Zhong and M. Chen, Lactate cross-feeding between Bifidobacterium species and Megasphaera indica contributes to butyrate formation in the human colonic environment, Appl. Environ. Microbiol., 2024, 90, e0101923, DOI:10.1128/aem.01019-23.
- S. Bang, G. Kim, M. Y. Lim, E. Song, D. Jung and J. Kum, et al., The influence of in vitro pectin fermentation on the human fecal microbiome, AMB Express, 2018, 8, 98, DOI:10.1186/s13568-018-0629-9.
- L. Moles, S. Delgado, M. Gorostidi-Aicua, L. Sepúlveda, A. Alberro and L. Iparraguirre, et al., Microbial dysbiosis and lack of SCFA production in a Spanish cohort of patients with multiple sclerosis, Front. Immunol., 2022, 13, 960761, DOI:10.3389/fimmu.2022.960761.
- S. Yu, L. Wang, X. Jing, Y. Wang and C. An, Features of gut microbiota and short-chain fatty acids in patients with first-episode depression and their relationship with the clinical symptoms, Front. Psychol., 2023, 14, 1088268, DOI:10.3389/fpsyg.2023.1088268.
- Z. Guan, E. Yu and Q. Feng, Soluble Dietary Fiber, One of the Most Important Nutrients for the Gut Microbiota, Molecules, 2021, 26, 6802, DOI:10.3390/molecules26226802.
- D. Zhang, J. Liu, H. Cheng, H. Wang, Y. Tan and W. Feng, et al., Interactions between polysaccharides and gut microbiota: A metabolomic and microbial review, Food Res. Int., 2022, 160, 111653, DOI:10.1016/j.foodres.2022.111653.
- C. Thomson, A. L. Garcia and C. A. Edwards, Interactions between dietary fibre and the gut microbiota, Proc. Nutr. Soc., 2021, 80, 398–408, DOI:10.1017/S0029665121002834.
- J. A. Domínguez-Avila, J. A. Villa-Rodriguez, M. Montiel-Herrera, R. Pacheco-Ordaz, D. E. Roopchand and K. Venema, et al., Phenolic Compounds Promote Diversity of Gut Microbiota and Maintain Colonic Health, Dig. Dis. Sci., 2021, 66, 3270–3289, DOI:10.1007/s10620-020-06676-7.
- S. Alqudah and J. Claesen, Mechanisms of gut bacterial metabolism of dietary polyphenols into bioactive compounds, Gut Microbes, 2024, 16, 2426614, DOI:10.1080/19490976.2024.2426614.
- W. Zhu, K. Lin, K. Li, X. Deng and C. Li, Reshaped fecal gut microbiota composition by the intake of high molecular weight persimmon tannin in normal and high-cholesterol diet-fed rats, Food Funct., 2018, 9, 541–551, 10.1039/C7FO00995J.
- X. Chen, S. Liu, H. Song, C. Yuan and J. Li, Evaluation of biological activity and prebiotic properties of proanthocyanidins with different degrees of polymerization through simulated digestion and in vitro fermentation by human fecal microbiota, Food Chem., 2024, 447, 139015, DOI:10.1016/j.foodchem.2024.139015.
- E. Brasili, N. M. A. Hassimotto, F. Del Chierico, F. Marini, A. Quagliariello and F. Sciubba, et al., Daily Consumption of Orange Juice from Citrus sinensis L. Osbeck cv. Cara Cara and cv. Bahia Differently Affects Gut Microbiota Profiling as Unveiled by an Integrated Meta-Omics Approach, J. Agric. Food Chem., 2019, 67, 1381–1391, DOI:10.1021/acs.jafc.8b05408.
- J. Wang, J. Hao, D. Miao, P. Xiao, X. Jiang and L. E-Hu, Compound chenpi tea consumption reduces obesity-related metabolic disorders by modulating gut microbiota and serum metabolites in mice, J. Sci. Food Agric., 2024, 104, 431–442, DOI:10.1002/jsfa.12940.
- K. Huang, G. Chu, P. Yang, Y. Liu, Y. Zhang and X. Guan, et al., Benefits of Monascus anka solid-state fermentation for quinoa polyphenol bioaccessibility and the anti-obesity effect linked with gut microbiota, Food Funct., 2024, 15, 2208–2220, 10.1039/D3FO04555B.
- H. C. Lee, A. M. Jenner, C. S. Low and Y. K. Lee, Effect of tea phenolics and their aromatic fecal bacterial metabolites on intestinal microbiota, Res. Microbiol., 2006, 157, 876–884, DOI:10.1016/j.resmic.2006.07.004.
- M. Jiao, W. He, Z. Ouyang, Q. Shi and Y. Wen, Progress in structural and functional study of the bacterial phenylacetic acid catabolic pathway, its role in pathogenicity and antibiotic resistance, Front. Microbiol., 2022, 13, 964019, DOI:10.3389/fmicb.2022.964019.
- L. Chen, X. Wang, J. Chen, J. Yang, X. Cai and Y. Chen, Caffeine ameliorates the metabolic syndrome in diet-induced obese mice through regulating the gut microbiota and serum metabolism, Diabetol. Metab. Syndr., 2023, 15, 37, DOI:10.1186/s13098-023-00993-3.
- Z. Guan, E. Yu and Q. Feng, Soluble dietary fiber, one of the most important nutrients for the gut microbiota, Molecules, 2021, 26, 6802, DOI:10.3390/molecules26226802.
- D. Mudgil, The interaction between insoluble and soluble fiber, in Dietary Fiber for the Prevention of Cardiovascular Disease, ed. R. A. Samaan, Academic Press, Los Angeles, CA, United States, 2017, pp. 35–59. DOI:10.1016/B978-0-12-805130-6.00003-3.
- A. K. Sinha, M. F. Laursen, J. E. Brinck, M. L. Rybtke, A. P. Hjørne, N. Procházková, M. Pedersen, H. M. Roager and T. R. Licht, Dietary fibre directs microbial tryptophan metabolism via metabolic interactions in the gut microbiota, Nat. Microbiol., 2024, 9, 1964–1978, DOI:10.1038/s41564-024-01737-3.
- P. Konopelski and I. Mogilnicka, Biological effects of indole-3-propionic acid, a gut microbiota-derived metabolite, and its precursor tryptophan in mammals’ health and disease, Int. J. Mol. Sci., 2022, 23, 1222, DOI:10.3390/ijms23031222.
- H. Jiang, C. Chen and J. Gao, Extensive summary of the important roles of indole propionic acid, a gut microbial metabolite in host health and disease, Nutrients, 2023, 15, 151, DOI:10.3390/nu15010151.
- D. Agircan, S. Taskin, M. Cekic and H. Celik, The neuroprotective role of indole-3-propionic acid in migraine pathophysiology, Medicina, 2024, 60, 1417, DOI:10.3390/medicina60091417.
- I. K. Hwang, K. Yoo, H. Li, O. K. Park, C. H. Lee, J. H. Choi, Y. Jeong, Y. L. Lee, Y. Kim, Y. Kwon and M. Won, Indole-3-propionic acid attenuates neuronal damage and oxidative stress in the ischemic hippocampus, J. Neurosci. Res., 2009, 87, 2126–2137, DOI:10.1002/jnr.22030.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |