Open Access Article
Open Access ArticleCocoa flavanols protect endothelial function during prolonged sitting in healthy older adults
Alessio
Daniele
 a,
Samuel J. E.
Lucas
a,
Samuel J. E.
Lucas
 ab and
Catarina
Rendeiro
ab and
Catarina
Rendeiro
 *ab
*ab
aSchool of Sport, Exercise and Rehabilitation Sciences, University of Birmingham, Birmingham, UK. E-mail: c.rendeiro@bham.ac.uk; Tel: +44 (0)1214147242
bCentre for Human Brain Health, University of Birmingham, Birmingham, UK
First published on 13th January 2026
Abstract
Sitting time is high in older adults and has been shown to temporarily impair endothelial function and blood pressure (BP). Flavanols, plant-derived compounds, acutely enhance endothelial function and reduce BP in older adults. The aim of this study was to investigate whether acute ingestion of cocoa flavanols can improve peripheral endothelial function and BP during prolonged sitting in healthy older adults. In a randomised, double-blinded, within-subject, cross-over, placebo-controlled human study, 20 apparently healthy, older adults (age, 72.4 ± 5.0 years; 7 males, 13 females) consumed a high-flavanol (695 mg) and a low-flavanol (5.6 mg) cocoa beverage immediately before a 2-hour sitting bout. Flow-mediated dilation (FMD) of the superficial femoral (SFA; primary outcome) and brachial (BA) arteries, and BP, were assessed before and after sitting. Microvasculature haemodynamics were assessed in the gastrocnemius before, during, and after sitting. Sitting reduced both SFA FMD (Δ = −0.7%; p = 0.005) and BA FMD (Δ = −0.7%; p = 0.016) in the low-flavanol condition. The high-flavanol intervention prevented the decline in both SFA and BA FMD following sitting, with FMD measures remaining similar to pre-sitting (p > 0.3). Sitting increased both systolic (Δ = 6.1 mm Hg, p = 0.001) and diastolic BP (Δ = 2.6 mm Hg, p = 0.001), with no benefit from flavanol intake. Sitting increased muscle oxygenation resting levels (p < 0.001) and haemoglobin content (p < 0.001), and decreased muscle oxygen consumption during SFA occlusion (p < 0.001). Flavanols had no effect on the muscle microvasculature. These findings indicate that flavanol-rich foods may be efficacious nutritional strategies to counteract sitting-induced endothelial impairments during prolonged sitting in older adults, but do not alleviate sitting-induced increases in BP.
Introduction
Sedentary behaviour is defined as any waking behaviour with an energy expenditure of less than 1.5 metabolic equivalents of task (also known as MET), while in a sitting, reclining or lying posture.1 Sedentarism is more prevalent in older adults compared to all other age groups.2 Older individuals are estimated to spend more than 10 hours in their waking day in sedentary activities,3 which can include sitting bouts lasting up to 5.4 hours per day.4 Greater sedentary time was found to be associated with a significantly increased risk of cardiovascular diseases (CVD) and all-cause mortality by 30% and 49%, respectively.5,6 Critically, for every extra hour per day spent sedentary, there is an increased risk of CVD and all-cause mortality of 5% and 11%, respectively.5,7 Among older adults, sedentary time has additionally been found to be positively associated with hypertension and metabolic disorders:8,9 specifically, higher systolic and diastolic blood pressure (BP), higher triglycerides, decreased levels of high-density lipoprotein cholesterol, and increased glycated haemoglobin (HbA1c).8,10–12 Interestingly, moderate-to-vigorous physical activity (MVPA) can reduce CVD risk and mortality,13,14 yet, sedentary time’s impact on CVD risk/mortality is still apparent among individuals who are physically active.7,15 Therefore, this is an increasing public health concern given that trends indicate the number of older adults to rise more than twofold worldwide between 2021 and 2050.16Numerous experimental studies have shown that even just one isolated episode of prolonged sitting (from 1 to 6 hours) has detrimental effects on human endothelial function, as measured by flow-mediated dilation (FMD).17,18 A recent meta-analysis, which included young, middle-aged and older adults, estimated that sitting-induced declines in lower-limb FMD can reach up to 2.5%.18 However, most of the available literature has demonstrated sitting-induced endothelial/vascular dysfunction in younger healthy adults (e.g., ref. 19–21), with only a limited number of studies focused on older adults. For example, Climie et al.22 have shown that 5 hours of uninterrupted sitting reduced FMD in the lower-limb superficial femoral artery (SFA) in sedentary overweight/obese adults,22 whilst a more recent study reported no differences in SFA FMD following 7 hours of uninterrupted sitting in older adults with type 2 diabetes.23 In addition, prolonged sitting has been shown to impact BP, with a recent study demonstrating that 8 hours of sitting can induce increases in both systolic (10 ± 1 mm Hg) and diastolic BP (5 ± 1 mm Hg) in overweight/obese older adults with type 2 diabetes.24 Alarmingly, these values reach a clinically relevant threshold associated with an increased risk of atherosclerotic CVD and stroke.25,26 This suggests that, if such behaviour persists, the impact of uninterrupted sitting periods in older adults is likely to contribute and/or aggravate long-term health issues.
Recent randomised controlled trials have shown that lowering sedentary time, for a duration of approximately 3 to 4 months, has induced meaningful reductions in systolic BP (down ∼3.5 mm Hg) and lower-limb endothelial function (as measured by FMD).27,28 Physical activity, particularly walking breaks and simple resistance-based activities, has also been shown to ameliorate the effects of sitting on SFA FMD22,23,27 and BP24,29,30 in late middle-aged and older adults. On the other hand, diet has been shown to be protective against cardiovascular disease (e.g., Mediterranean or DASH diets),31,32 but has been poorly explored as a strategy to counteract the negative effects of sitting/sedentarism on vascular health. Flavonoid-rich foods are typically consumed in high amounts in cardioprotective diets, with evidence from both observational and RCTs showing flavonoids to improve biomarkers of cardiovascular health, particularly upper-limb brachial artery (BA) FMD and BP both acutely and chronically.33–42 In particular, flavanols, a subgroup of flavonoids present in unprocessed cocoa, tea, berries, grapes and apples,43 have been extensively shown to act quickly within the vasculature, improving both brachial FMD39,44–46 and lower-limb common femoral artery (CFA) FMD47 within 2 hours of intake in older adults. As such, flavanol-rich foods may be an effective dietary strategy to help minimise the detrimental effects of prolonged sitting on endothelial function and BP in old age, particularly on occasions during which frequent breaks from sitting may not be viable in this population. Importantly, reducing sedentary behaviour remains a primary public health goal. However, when this is not possible due to occupational, environmental, or physical limitations, complementary dietary strategies may be able to minimise sitting-induced vascular deficits.
Therefore, the aim of the current study was to examine whether acute consumption of cocoa flavanols prior to a 2-hour bout of uninterrupted sitting can be beneficial in improving endothelial function (as measured by FMD) in the upper-limb BA and lower-limb SFA in older adults, as well as downstream muscle microvascular function. We further investigated whether cocoa flavanols could prevent sitting-induced increases in BP in older adults. We hypothesised that cocoa flavanols would be effective at rescuing upper- and lower-limb FMD and improve BP during uninterrupted sitting in older age.
Materials and methods
Ethical approval
The study was conducted in accordance with the Declaration of Helsinki, and approved by the University of Birmingham Science, Technology, Engineering and Mathematics ethics committee (ERN_19-0851B). Informed written consent was obtained from all participants before enrolment in the study. The study was retrospectively registered at clinicaltrials.gov (NCT07265869).Participants
Twenty healthy older adults (aged ≥65 years old, males or females) were recruited from the University of Birmingham (Birmingham, England) and the surrounding community, and throughout ‘the Birmingham 1000 Elders group’. Prior to participation in the study, all participants provided a signed informed consent form and completed a general health and lifestyle questionnaire. Excluding criteria included a history or symptoms of cardiovascular, renal, pulmonary, metabolic, or neurologic disease, hypertension (blood pressure higher than 140/90 mm Hg, based on recent guidelines48), diabetes mellitus, anaemia, asthma, immune conditions, or high cholesterol. In addition, smokers, individuals who were on weight-reducing diets or taking anticoagulants, or had recently undergone prolonged bed-rest periods, were also excluded from the study.Study design
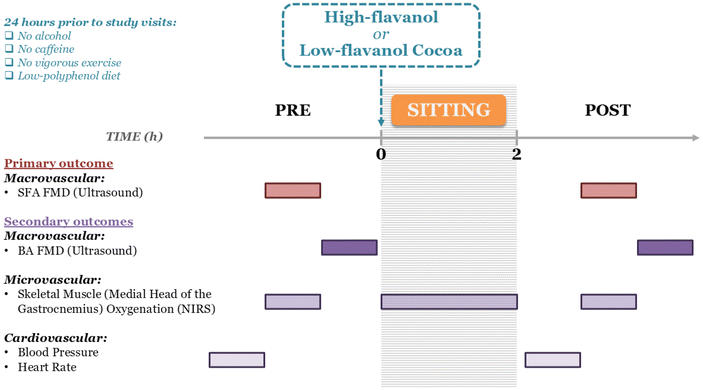
A randomised, double-blinded, counterbalanced, placebo-controlled, within-subject, cross-over intervention study (Fig. 1) was conducted.All participants attended two experimental visits separated by a minimum washout period of seven days, in line with previous studies.49–51 Prior to the experimental visits, participants were asked to fast for at least 12 hours. Participants were also instructed to refrain from caffeine, alcohol and polyphenol-containing foods/beverages (a detailed list of foods/beverages to avoid and that are allowed to consume was provided beforehand), and any form of physical activity for at least 24 hours. Although polyphenol metabolites from some sources can remain in circulation for up to 80 hours after ingestion,52 the majority of metabolites are excreted within the first 24 hours.53 The two experimental visits started in the early morning (between 08:00 and 10:30). Upon arrival at the laboratory, anthropometric characteristics were recorded using a telescopic measuring rod (Seca 220, Seca, Hamburg, Germany) and a flat scale (Seca 880, Seca, Hamburg, Germany) for height and weight, respectively. Participants were invited to rest in a supine position for approximately 15 min. To assess compliance with the 24-hour dietary restrictions, a 24-hour Dietary Recall Questionnaire was performed verbally at the start of each experimental visit. The baseline measures preceding sitting/flavanol intervention were measured in the following sequence: (i) upper arm systolic and diastolic blood pressure, (ii) heart rate, (iii) SFA FMD, (iv) gastrocnemius (medial head) oxygenation during post-occlusive reactive hyperaemia in concomitance with SFA FMD (assessed via near-infrared spectroscopy; NIRS) and (v) BA FMD. Subsequently, the participant sat for 2 hours on a comfortable armchair while consuming (within 10 min) the high- or low-flavanol cocoa beverage (randomly assigned, counterbalanced and double-blinded). In addition, medial gastrocnemius oxygenation (assessed via NIRS) was taken during the sitting trial (Fig. 2).
While sitting, the participants were asked to keep their lower limbs parallel and bent at approx. 90° with both feet in a neutral position with the plantar surface on the floor, while having the trunk resting on the backrest. Participants were only permitted to move their arms/hands to do light activities such as computer typing. Two hours after the commencement of the sitting/flavanol intervention, the aforementioned primary and secondary outcome measures were reassessed (Fig. 2). At the end of each experimental visit, participants were asked to complete a Feasibility Questionnaire to assess their comfort level during each FMD protocol. Experimental visits were performed in a quiet, darkened and temperature-controlled laboratory (22–24 °C), as recommended in guidelines for FMD procedures.54,55 The study adopted a 2-hour sitting period, as this duration is adequate to induce measurable declines in FMD following prolonged sitting21 and coincides with the peak bioavailability of flavanol metabolites in circulation, which generally occurs around 2 hours post-ingestion.46 Our investigation focused on SFA FMD as the designated primary outcome measure. Complementary secondary outcome measures included: BA FMD, BP, heart rate, and NIRS-related measures.
Habitual diet and physical activity
Habitual diet and physical activity were assessed according to the following procedure: briefly, participants were asked to wear a wrist-worn tri-axial accelerometer (GENEActiv, version 1.1, ActivInsights Ltd, Kimbolton, England) for 7 consecutive days and to complete a Food Frequency Questionnaire where they were asked to recall their diet in the previous year. Physical activity data were collected at a frequency of 85.7 Hz, converted into a 60-second epoch and analysed using the GENEActiv software (GENEActiv, version 3.3, ActivInsights Ltd, Kimbolton, England). Habitual diet was assessed using the European Prospective Investigation into Diet and Cancer (EPIC) Norfolk Food Frequency Questionnaire (FFQ),56 which comprises 131 food products to select the intake frequency on a 9-point scale. Nutrient and food group data was estimated using the FFQ EPIC Tool for Analysis (FETA):57 energy (kcal), fat (g), saturated fat (g), carbohydrate (g), sugars (g), fibre (g), protein (g), total flavonoids (mg), and portions of fruit and vegetables (calculated as 1 portion corresponding to 80 g, from National Health Service [NHS] guidelines58) are reported.High- and low-flavanol interventions
Cocoa flavanol beverages were prepared as previously described.49,59 Briefly, 12 g of cocoa powder was dissolved in 350 mL of ‘Buxton’ still natural mineral water (nitrate: <0.1 mg L−1). The two cocoa powders used are commercially available (Barry Callebaut AG, Zurich, Switzerland). The low-flavanol cocoa powder was a fat-reduced alkalized cocoa powder containing less than 6 mg of (−)-epicatechin, and 5.6 mg of total flavanols per beverage. The high-flavanol cocoa powder was a fat-reduced natural cocoa powder containing 150 mg of (−)-epicatechin, and 695 mg of total flavanols per beverage. Concentrations of macronutrient content and flavonoid compounds are reported in Table 1.| Low flavanol | High flavanol | |
|---|---|---|
| a Concentrations expressed as mg of gallic acid equivalents per 12 g of cocoa powder. | ||
| Total polyphenolsa | 260.0 | 1246.8 |
| Total flavanols (mg) | 5.6 | 695.0 |
| Procyanidins (dimers-decamers; mg) | ND | 459.6 |
| (−)-Epicatechin (mg) | <6 | 150.0 |
| (−) and (+)-Catechin (mg) | <6 | 85.4 |
| Theobromine (mg) | 278.4 | 262.8 |
| Caffeine (mg) | 22.2 | 27.6 |
| Fat (g) | 1.3 | 1.7 |
| Carbohydrates (g) | 1.2 | 2.7 |
| Protein (g) | 2.7 | 2.7 |
| Fibre (g) | 4.0 | 1.8 |
| Energy (kcal) | 36.6 | 41.4 |
The two cocoa-based flavanol interventions were matched for all micro- and macronutrients, including methylxanthines (caffeine and theobromine). Cocoa powder concentrations for flavanol monomers, procyanidins, and methylxanthines were measured by high-performance liquid chromatography, as described in previous studies.60,61 Total polyphenol concentrations were estimated by a Folin–Ciocalteu reagent calorimetric assay, as described previously.62 The dose of flavanol monomers used in the present study was chosen based on previous research demonstrating its effectiveness in modulating endothelial function and is consistent with that used in our prior studies.49,51,59 To ensure double-blindness, cocoa beverages were indistinguishable in texture, aroma, and taste, and they were provided in an opaque container (covered on top) with a dark-coloured opaque straw. Cocoa interventions were unblinded after completion of all data analyses.
Flow-mediated dilation of the brachial and superficial femoral arteries
Endothelial-dependent vasodilation of the SFA and BA was assessed using established FMD protocol guidelines.55 Briefly, artery diameter and blood velocity of the SFA and BA were non-invasively assessed by means of a high-resolution duplex ultrasound device (Terason uSmart 3300, Teratech Corporation, Burlington, MA, USA) with a linear array transducer (4.5 MHz) (Terason 15L4 Smart Mark, Teratech Corporation, Burlington, MA, USA) attached to an adjustable stereotactic probe-holding tool (FMD-probe-holder-xyz, Quipu S.r.l., Pisa, Italy). The pulse-wave Doppler signal was corrected at an insonation angle of 60°. The right SFA was located and scanned longitudinally 10–20 cm distal to the inguinal crease. A manual blood pressure cuff was wrapped around the distal end of the right thigh (3–4 cm proximal to the patella), distal to the imaged artery. The right BA was located and scanned longitudinally between 5 and 10 cm proximal to the antecubital fossa. A manual blood pressure cuff was wrapped around the right forearm (∼2 cm distal to the antecubital fossa), distal to the imaged artery. Artery diameter and blood velocity were continuously recorded for 1 min (baseline), 5 min during which the cuff was inflated and maintained at a pressure of 220 mm Hg, and 5 minutes following the rapid cuff deflation. The total duration of the FMD protocol was 11 min. All the FMD protocols were performed by a trained and experienced PhD student (AD; first author), with inter-day coefficients of variation (CV) for arterial diameter (mm) of 2.2% and 2.8% for BA and SFA, respectively, and for FMD (%), 10.9% and 8.3% for BA and SFA, respectively, in healthy young adults.Measurements of arterial diameter and blood velocity were analysed offline by means of an automated edge-detection software (Cardiovascular Suite, version 3.4.0, Quipu S.r.l., Pisa, Italy). All video recordings were analysed by the same researcher (AD) who performed the FMD measurements. The blood velocity calculated by the software (i.e., time-averaged maximum velocity) was adjusted to reflect time-averaged mean velocity as described in previous research,63,64 based on the following formula: Time-averaged mean velocity = time-averaged maximum velocity/2. The resulting blood velocity was used to calculate blood flow using the following formula: Blood flow = [blood velocity × π (baseline diameter/2)2] × 60. Baseline and peak diameters were calculated as the average diameter recorded during the minute preceding cuff inflation and the largest diameter observed following cuff deflation, respectively. Both parameters were then used to calculate the percent change in arterial diameter following the formula: FMD (%) = [(peak diameter − baseline diameter)/baseline diameter] × 100. In addition to the classic FMD (%) calculation, allometric scaling of FMD as proposed by Atkinson and Batterham65 is also presented, in accordance with their previously published guidelines.66 Shear rate, an adequate surrogate measure of shear stress,67 was calculated as follows: 4 × baseline blood velocity/baseline diameter.
Muscle microvasculature haemodynamics
Skeletal muscle microvascular haemodynamics were assessed in the leg using a NIRS device (NIRO-200NX, Hamamatsu Photonics KK, Shizuoka, Japan) during: (i) the 2-hour sitting trial, and (ii) the SFA FMD protocol, according to the following procedure: briefly, the NIRS probe was placed on the right medial gastrocnemius and covered with an opaque cover to block the external ambient light. Tissue oxygenation index (TOI) and normalised tissue haemoglobin index (nTHI) were the outcome measures used, derived via the spatially resolved spectroscopy (SRS) method.68Blood pressure
Blood pressure was measured in the left upper arm in supine position (after more than 10 min of supine resting) using an automatic blood pressure (BP) monitor (Omron M3 [HEM-7131-E], OMRON HEALTHCARE Co., Ltd, Kyoto, Japan) to obtain systolic BP, diastolic BP, and heart rate. Within each time point, BP measurements were taken three consecutive times, which were then averaged.Statistical analysis
All statistical analyses were performed using statistical software IBM SPSS Statistics for Windows, version 29 (IBM Corp., Armonk, New York, USA). Two-way repeated measures ANOVA with sitting time (0; 2 h) and dietary intervention (low flavanol; high flavanol) as within-subject variables was performed to analyse changes in SFA and BA FMD, arterial diameter, shear rate, blood flow, blood pressure and NIRS-related haemodynamic measures. To assess the impact of medication use for SFA FMD (N = 8, on medication; N = 4, free of medication) and BA FMD (N = 6, on medication; N = 8, free of medication), medication use was also added as a between-subjects factor in the analysis. Additionally, as a separate exploratory analysis, sedentary activity volume and vigorous activity volume (only habitual activity variables that had a considerable spread across the sample) were included as covariates in the model, to assess whether habitual levels of physical activity/sedentarism modulated the impact of flavanols on endothelial function during sitting. TOI and nTHI during sitting were analysed by a two-way repeated measures ANOVA with sitting time (0, 10, 60, 120 min) and dietary intervention (low flavanol; high flavanol) as within-subject variables. Paired-samples t-tests were used to assess differences between flavanol interventions (low flavanol; high flavanol) at baseline and to determine differences in comfort level ratings (data not shown) between arteries (SFA; BA) across the two experimental visits, and flavanol interventions (low flavanol; high flavanol). Normality of residuals was assessed using Shapiro–Wilk tests and Q–Q plots. For some outcome measures, the Shapiro–Wilk test indicated significant deviations from normality (p < 0.05), although Q–Q plots showed only mild departures and no substantial skew. To address this, data were normalised (Shapiro–Wilk test: p > 0.05) and statistical analysis in the normalised data confirmed previous statistical results in the raw data, confirming the ANOVA’s robustness to moderate violations of normality.69,70 Sample size estimation was performed using G*Power (version 3.1.9.3) and was based on acute changes in SFA FMD following the same dose of flavanols during sitting in 40 young healthy males.51 This power analysis estimated that 20 participants were needed to determine a significant difference, of at least 1% FMD (SD = 1.2%), in SFA FMD between high and low flavanol post-sitting (p < 0.05; power of 95%, dz = 0.867). The data in the tables and figures are presented as mean ± SD. A p-value less than 0.05 was considered statistically significant. Non-usable data for BA FMD (N = 6/20) and SFA FMD (N = 8/20) were due to the inability to recover a reliable/stable B-mode image post-occlusion in some of the time points. For NIRS during SFA occlusion, the non-usable data (N = 5/20) was due to data files presenting high-amplitude, high-frequency artefacts and also included occasions in which ‘signal underflow’ was detected by the NIRS device, reflecting poor signal quality.Results
Study participants
Participants’ anthropometric characteristics, sedentary time, and physical activity levels are summarised in Table 2. All participants were older adults (age, 72.4 ± 5.0 years; N = 20) who were apparently healthy, with a healthy body mass index (BMI) (24.4 ± 3.6 kg m−2). Seven-day averaged accelerometry data revealed that participants spent an average of 1.8 ± 0.6 hours per day engaging in light physical activity, and had a light physical activity volume of 263.6 ± 95.3 MET min. All participants recorded an average of more than 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 steps per day. Participants’ medication use reflects that of the general older UK population, including medications for cardiovascular risk management, such as statins (N = 4), as well as treatments for hypothyroidism (e.g., levothyroxine; N = 3), dyspepsia (e.g., omeprazole; N = 1), and osteoporosis (e.g., alendronic acid; N = 1).
000 steps per day. Participants’ medication use reflects that of the general older UK population, including medications for cardiovascular risk management, such as statins (N = 4), as well as treatments for hypothyroidism (e.g., levothyroxine; N = 3), dyspepsia (e.g., omeprazole; N = 1), and osteoporosis (e.g., alendronic acid; N = 1).
| All participants | |
|---|---|
| Recommendations for older adults (aerobic physical activity, per week) – at least 150–300 min of moderate intensity, or at least 75–150 min of vigorous intensity.71 Data are presented as mean ± SD. BMI, body mass index; MET, metabolic equivalent of task. | |
| Anthropometric characteristics | |
| N | 20 |
| Age (year) | 72.4 ± 5.0 |
| Height (m) | 1.67 ± 0.10 |
| Weight (kg) | 68.7 ± 15.6 |
| BMI (kg m−2) | 24.4 ± 3.6 |
| Daily physical activity pattern | |
| N | 19 |
| Sedentary activity time (h) | 9.8 ± 1.5 |
| Light activity time (h) | 1.8 ± 0.6 |
| Moderate activity time (h) | 2.5 ± 1.1 |
| Vigorous activity time (h) | 0.0 ± 0.1 |
| Sedentary activity volume (estimated as MET min) | 747.1 ± 138.7 |
| Light activity volume (estimated as MET min) | 263.6 ± 95.3 |
| Moderate activity volume (estimated as MET min) | 533.7 ± 243.6 |
| Vigorous activity volume (estimated as MET min) | 25.2 ± 35.3 |
| Step count | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 651.8 ± 3 651.8 ± 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 883.6 883.6 |
Habitual dietary intake
Estimates of participants’ daily dietary intake of macronutrients, total flavonoids and portions of fruits and vegetables are displayed in Table 3. The percentage of individuals who exceed or fail to meet the daily dietary recommendations (based on NHS guidelines58,72) are also summarised (Table 3). Participants consumed on average 863 mg per day of flavonoids, and 7.4 portions per day of fruit and vegetables. Further breakdown of flavonoid intake revealed that participants consumed approximately 154.9 mg per day of total flavanols, 54.0 mg per day of proanthocyanidin monomers, and 106.6 mg per day of polymers—with a degree of polymerisation of 2–10. Finally, participants consumed on average 60.8 g of fat, 97.1 g of sugars, and 17.9 g of fibre daily, which were 31.6% over, 100.0% over, and 89.5% under the recommended daily intake of fat, sugars, and fibre, respectively.| Sample average | % of participants over/under the recommended daily intake | |
|---|---|---|
| Recommendations for adults – fat: <70 g per day, saturated fat: <30 g per day (male) or <20 g per day (female), sugar: <30 g per day, fibre: >30 g per day, fruit and vegetables: >5 portions per day. *1 portion = 80 g (NHS guidelines58). Data are presented as mean ± SD (N = 19). | ||
| Energy (kcal) | 1530.4 ± 439.8 | N/A |
| Fat (g) | 60.8 ± 23.8 | 31.6% over |
| Saturated fat (g) | 21.2 ± 11.4 | 21.1% over |
| Carbohydrate (g) | 175.3 ± 55.7 | N/A |
| Sugars (g) | 97.1 ± 44.1 | 100.0% over |
| Fibre (g) | 17.9 ± 6.7 | 89.5% under |
| Protein (g) | 70.7 ± 23.5 | N/A |
| Total flavonoids (mg) | 863.3 ± 430.1 | N/A |
| Fruit and vegetables (g) | 592.7 ± 328.6 | N/A |
| Fruit and vegetables (portion) | 7.4 ± 4.1 | 21.1% under |
Vascular and haemodynamic measures
A summary of baseline (pre-intervention) resting vascular parameters—including arterial diameter, shear rate, FMD, blood pressure, and heart rate—during the low- and high-flavanol interventions is presented in Table 4. No significant differences were observed between the flavanol interventions for any parameter.| N | Low flavanol | High flavanol | p-Value | |
|---|---|---|---|---|
| Data are presented as mean ± SD. BP, blood pressure; FMD, flow-mediated dilation. Data analysed via paired-samples t-tests. | ||||
| Brachial Artery | ||||
| Artery diameter (mm) | 20 | 3.9 ± 0.7 | 3.9 ± 0.6 | 0.525 |
| Anterograde shear rate (s−1) | 20 | 92.0 ± 29.1 | 96.4 ± 34.9 | 0.540 |
| Retrograde shear rate (s−1) | 20 | −28.7 ± 23.4 | −28.8 ± 22.5 | 0.998 |
| Anterograde blood flow (mL min−1) | 20 | 31.9 ± 12.6 | 32.9 ± 14.9 | 0.530 |
| Retrograde blood flow (mL min−1) | 20 | −9.6 ± 6.7 | −10.1 ± 8.9 | 0.667 |
| FMD (%) | 15 | 3.4 ± 1.3 | 3.5 ± 1.4 | 0.258 |
| Superficial femoral artery | ||||
| Artery diameter (mm) | 19 | 6.5 ± 1.4 | 6.5 ± 1.6 | 0.415 |
| Anterograde shear rate (s−1) | 19 | 61.4 ± 24.9 | 62.5 ± 21.7 | 0.918 |
| Retrograde shear rate (s−1) | 19 | −37.6 ± 20.6 | −38.7 ± 21.1 | 0.798 |
| Anterograde blood flow (mL min−1) | 19 | 95.7 ± 42.8 | 95.2 ± 38.0 | 0.690 |
| Retrograde blood flow (mL min−1) | 19 | −51.4 ± 20.2 | −55.9 ± 23.5 | 0.183 |
| FMD (%) | 14 | 2.0 ± 1.2 | 1.7 ± 1.2 | 0.144 |
| Blood pressure/heart rate | ||||
| Systolic BP (mm Hg) | 20 | 128.8 ± 14.1 | 128.0 ± 14.1 | 0.798 |
| Diastolic BP (mm Hg) | 20 | 75.0 ± 7.4 | 74.0 ± 7.1 | 0.426 |
| Heart rate (bpm) | 20 | 62 ± 12 | 61 ± 12 | 0.535 |
Resting blood flow and arterial diameter
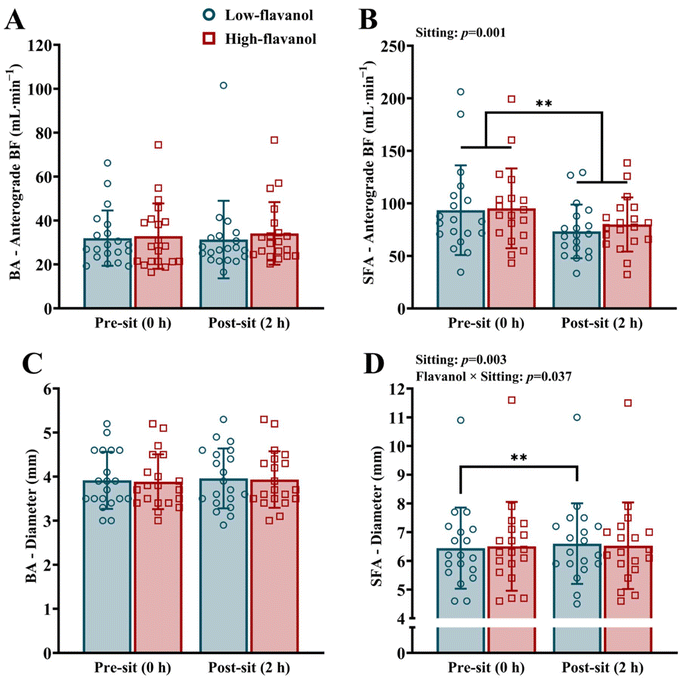
Resting anterograde blood flow and arterial diameter of the BA and SFA are displayed in Fig. 4. In the BA, neither sitting nor flavanol intervention had an effect on anterograde blood flow (Fig. 4A). In the SFA, sitting resulted in a similar decline in anterograde blood flow for both flavanol interventions (p = 0.001; Fig. 4B). No differences in retrograde blood flow were detected in both arteries due to sitting or flavanol intervention (data not shown). Sitting had no impact on arterial diameter in the BA (Fig. 4C). In the SFA, however, sitting increased arterial diameter, but only following consumption of the low-flavanol cocoa (p = 0.003; Fig. 4D).Flow-mediated dilation
Fig. 5 shows BA and SFA FMD as well as allometrically scaled FMD before and after 2 hours of sitting following either a low- or high-flavanol acute intervention.As shown in Fig. 5, there was a significant interaction effect for both BA and SFA FMD responses (p ≤ 0.015). Specifically, while sitting resulted in a decline in BA FMD (Δ = −0.7 ± 1.4%; p < 0.05; Fig. 5A) and SFA FMD (Δ = −0.7 ± 1.2%; p < 0.01; Fig. 5B) following consumption of the low-flavanol cocoa, after the high-flavanol cocoa there were no detectable changes in either the BA (Δ = 0.1 ± 1.3%; p = 0.375) or SFA (Δ = 0.0 ± 1.1%; p = 0.593) FMD response. After sitting, there was a higher FMD following the high-flavanol compared to the low-flavanol in the BA (p < 0.001), but not in the SFA (p = 0.164, ηp2 = 0.168). No significant differences between the flavanol interventions were detected at baseline in both the BA and SFA.
Allometrically scaled BA FMD (Fig. 5C) outcomes showed the same pattern, with sitting-induced reductions following ingestion of the low-flavanol cocoa (p < 0.005), and no changes were observed in response to high-flavanol intake (p = 0.507). Higher FMD was reported after sitting following the high-flavanol compared to the low-flavanol cocoa (p < 0.001). For the SFA FMD (Fig. 5D), a significant effect of sitting was observed (p = 0.004), and a trend towards an interaction with flavanol intake (p = 0.055, ηp2 = 0.090). Further post hoc analyses suggest a significant decline in SFA FMD only in the low-flavanol condition (p < 0.001), whilst FMD is maintained after high-flavanol intake (p = 0.42). No differences between the flavanol interventions were detected at baseline in both the BA and SFA allometrically scaled FMD measures (both p > 0.05).
Participant habitual medication did not significantly affect the impact of sitting or flavanol intervention on endothelial function measures in the BA or SFA. However, individuals on medication had an overall lower BA FMD (p = 0.019). Additionally, sedentary activity volume or vigorous activity volume was included as a covariate in the statistical analysis, with no significant effects of the covariates observed for either BA FMD or SFA FMD, suggesting that habitual levels of vigorous physical activity/sedentarism does not affect benefits of flavanol interventions.
Leg muscle oxygenation haemodynamics during SFA occlusion
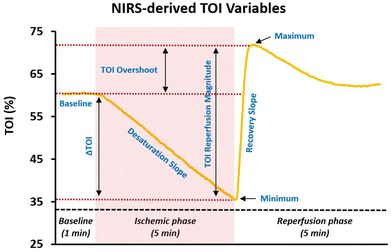
Microvascular tissue oxygenation in the medial gastrocnemius during rest, SFA occlusion (ischaemic period), and post-occlusion reactive hyperaemia are depicted in Fig. 6A. Parameters describing the microvascular haemodynamic response (as represented in Fig. 3) were estimated before and after sitting, following either a low- or high-flavanol intervention, and are summarised in Table 5.| Low Flavanol | High Flavanol | |||||
|---|---|---|---|---|---|---|
| TOI Variable | N | Pre | Post | Pre | Post | Effect |
| Data are presented as mean ± SD. TOI, tissue oxygenation index. Two-way repeated measures ANOVA conducted. | ||||||
| Baseline (%) | 20 | 70.2 ± 5.1 | 70.3 ± 4.8 | 69.5 ± 4.3 | 69.1 ± 4.9 | NS |
| Minimum (%) | 19 | 61.5 ± 9.5 | 63.6 ± 9.5 | 60.7 ± 8.0 | 62.9 ± 7.2 | Sitting (p = 0.006) |
| Maximum (%) | 15 | 73.4 ± 4.1 | 74.0 ± 3.5 | 72.5 ± 4.1 | 72.3 ± 3.2 | Flavanol (p = 0.018) |
| ΔTOI (%) | 19 | 8.3 ± 6.1 | 6.5 ± 5.8 | 8.5 ± 5.1 | 5.9 ± 4.2 | Sitting (p < 0.001) |
| Overshoot (%) | 15 | 3.9 ± 3.0 | 4.5 ± 3.3 | 3.8 ± 3.3 | 4.2 ± 3.4 | NS |
| Reperfusion magnitude (%) | 15 | 13.4 ± 8.9 | 12.2 ± 8.8 | 13.8 ± 7.6 | 11.1 ± 6.8 | Sitting (p = 0.006) |
| Desaturation slope (% s−1) | 19 | −0.03 ± 0.02 | −0.02 ± 0.02 | −0.03 ± 0.02 | −0.02 ± 0.01 | Sitting (p < 0.001) |
| Recovery slope (% s−1) | 15 | 0.38 ± 0.25 | 0.32 ± 0.18 | 0.43 ± 0.25 | 0.38 ± 0.27 | NS |
During the reperfusion period (after the release of the cuff), sitting induced a reduction in reperfusion magnitude (minimum–maximum) (p = 0.006; Fig. 6B), with no significant effect on recovery slope (Fig. 6C), and overshoot (baseline–maximum; see Table 5). During the ischaemic period (when the cuff remains inflated), sitting resulted in a reduction in ΔTOI (baseline–minimum) (p < 0.001; Fig. 6D), a slower/reduced desaturation slope (p < 0.001; Fig. 6E), and an increase in minimum oxygenation (p = 0.006; see Table 5). Sitting did not induce any significant change in resting baseline oxygenation (Table 5). The high-flavanol intervention displayed a lower maximum compared to the low-flavanol (p = 0.018), but this difference was present before (0 h) and after sitting (2 h). Consuming flavanols prior to sitting did not affect any haemodynamic responses during the hyperaemic or occlusion phase (Table 5).
Leg muscle oxygenation haemodynamics during sitting
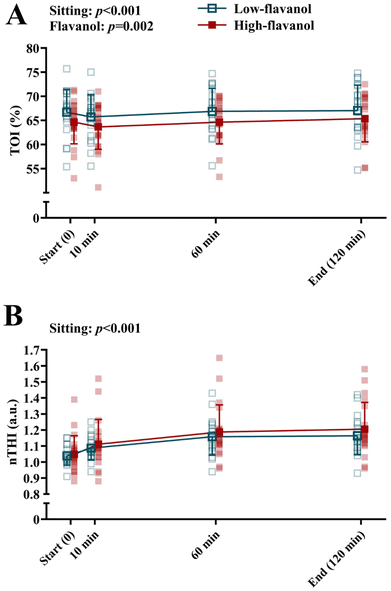
Oxygenation levels (TOI) and total haemoglobin content (nTHI) measured in the medial gastrocnemius during the 2-hour sitting trial are reported in Fig. 7 (A and B, respectively).Sitting modulated both TOI (p < 0.001; Fig. 7A) and nTHI (p < 0.001; Fig. 7B) over the 2-hour period, regardless of the flavanols intervention. Post hoc analysis revealed a decline in TOI after 10 min of sitting (p = 0.035 vs. start [0]), whilst there was an increase from 10 min to 60 min (p = 0.002) and 120 min (p = 0.002 vs. 10 min). No significant differences were observed between start (0) and 60 min or 120 min. In nTHI, post hoc analysis revealed increases from the start (0) at 10 min (p = 0.001), 60 min (p < 0.001), and at 120 min (p < 0.001). In addition, for TOI only, there was a main effect of flavanol intervention (p = 0.002), with the high-flavanol intervention displaying lower TOI during the sitting trial as compared to the low-flavanol.
Blood pressure and heart rate
Sitting resulted in an increase in both systolic (low-flavanol: Δ = 5.2 ± 7.4 mm Hg; high-flavanol: Δ = 7.0 ± 10.1 mm Hg; p = 0.001; Fig. 8A) and diastolic BP (low-flavanol: Δ = 1.8 ± 3.5 mm Hg; high-flavanol: Δ = 3.5 ± 5.0 mm Hg; p = 0.001; Fig. 8B), and a decline in heart rate (low-flavanol: Δ = −5 ± 5 bpm; high-flavanol: Δ = −4 ± 4 bpm; p < 0.001; Fig. 8C). For both BP and HR, these changes were similar following both flavanol interventions (interaction effects: p > 0.05).Discussion
In the current study, we showed for the first time that cocoa flavanols prevented sitting-induced declines in upper- and lower-limb FMD without affecting the microvasculature and blood pressure in apparently healthy, older adults. We further showed that 2 hours of uninterrupted sitting induced detrimental declines in BA and SFA FMD and reduced resting anterograde blood flow. Muscle microvascular reperfusion magnitude and rate of oxygen consumption (desaturation slope) were also impaired in the lower limbs, following sitting. Furthermore, the sitting intervention resulted in increases in both systolic and diastolic BP and declines in heart rate. This is the first study to suggest that dietary flavonoids may be protective in healthy older adults during prolonged sitting.Impact of sitting on the macro and microvasculature
In the present study, we observed for the first time sitting-induced declines in endothelial function in the BA (0.7% FMD) in healthy older adults, in addition to impairments in the SFA (0.7% FMD). One previous study in late middle-aged overweight/obese adults reported no changes in BA and a larger sitting effect in SFA (1.5%) FMD.22 The sample in the current study, was relatively healthy, which may contribute to these differences: for example, our sample had a healthier BMI (24.4 kg m−2), was highly active and had a relatively heathy diet (>7 F&V portions per day). The BA FMD values of 0.7% are moderately close to the clinically relevant threshold of 1%, which has been found to be inversely associated with a risk of a future CVD event of at least 9%.73 This may have clinical significance in this particular population, given that older adults can experience several bouts of prolonged sitting per day.3,4 To date, only a limited number of other sitting studies have been conducted in older adults; specifically, with type 2 diabetes23 and increased cardiovascular risk.27 As such our study adds to the body of evidence in a relatively healthy sample.There were also reductions in anterograde blood flow and shear rate in SFA following the 2-hour sitting trial, with no significant changes in retrograde shear rate/blood flow. This has been extensively shown in young adults21,74–77 but the data in older adults is more inconsistent, with some studies reporting either no significant changes,23 increases22 or decreases27 in shear rate and/or blood flow in the SFA. This may be a reflection of a more heterogeneous population in comparison to a young group. Furthermore, only one other study assessed the retrograde component of blood flow,27 whose modulation can be associated with endothelial dysfunction outcomes.78 Similar to the current study, no changes in retrograde blood flow/shear rate were observed in upper and lower limbs after sitting, indicating that this is unlikely to be driving the negative effects of sitting on endothelial function. It is widely recognised that shear rate/stress plays a substantial role in the regulation of the vascular response in both conduit arteries and microvessels,67,79 as it initiates a cascade that leads to increased vasodilation.80,81 The FMD response, however, is not determined completely by shear rate/stress, as other aspects are involved as potential underlying mechanisms, which include increased hydrostatic pressure, blood viscosity, arterial bending, and modulation of metabolic and vascular factors such as insulin resistance, endothelin-1 (ET-1), and nitric oxide (NO) (as reviewed in Daniele et al.17).
The current study shows, for the first time, that sitting induces a decline in reperfusion magnitude in the lower limb microvasculature during reactive hyperaemia, suggesting impaired microvascular function. This was paralleled by a decline in ΔTOI (baseline–minimum, %) and a higher minimum oxygenation reached during occlusion, indicating reduced oxygen extraction during ischaemia.82 Furthermore, the TOI desaturation slope was less steep after sitting in both flavanol interventions, indicating reduced muscle oxygen consumption during the occlusion phase of the FMD protocol, which is representative of impaired microcirculation.83 Importantly, the TOI desaturation slope has been found to be an important predictor of mortality (i.e., 0.70).83 A recent study has found a negative correlation between TOI desaturation slope (tibial anterior muscle) and SFA FMD (r = −0.509, p = 0.044) in young healthy adults,84 which is consistent with the observations of the current study.
The literature investigating microvascular function in response to prolonged sitting has mainly focused on younger populations, with little to no research in older populations. Similar to the present study, previous investigations in young adults show that exposure to sitting (2.5–3 hours) affects tissue oxygenation parameters, including reductions in baseline oxygenation and reoxygenation rate and slower/reduced desaturation rate.85–89 Furthermore, the continuous recording of muscle oxygenation revealed an increase in oxygenation levels (TOI) and total haemoglobin content (nTHI) during sitting. This is generally in contrast with previous studies in young adults, showing typically a decline in TOI.90,91 Future work is needed to understand the underlying factors associated with these age differences.
Overall, these findings highlight that uninterrupted sitting impairs upper- and lower-limb macrovascular function, as well as leg microvascular function, given that reactive hyperaemia is a measure of peripheral microvascular function closely associated to cardiovascular risk factors, in healthy and at-risk populations.92
Impact of flavanols on the macro and microvasculature during sitting
Cocoa flavanols have shown efficacy in preventing declines in endothelial function in both BA and SFA, with levels post-sitting remaining similar to pre-sitting. The positive effects on the SFA seem less accentuated: this may be partially driven by the limited sample size (N = 12 for the SFA), which was under the sample size estimated (N = 20, Power = 95%). This was due to loss of data as implementing the SFA FMD protocol in this population is more challenging: this may be related to the elevated discomfort experienced during SFA FMD (data not shown). Interestingly, cocoa flavanols enhanced FMD without having an impact on shear rate, suggesting that improvements in endothelial function may be related to other mechanisms. Human studies have shown that flavanols—particularly, (–)-epicatechin—upregulate bioavailability and production of NO,93 a potent vasodilator that largely mediates the FMD response,46,94 whilst reducing concentrations of ET-1, a potent vasoconstrictor.93 Furthermore, flavanols have been recently demonstrated to reduce oxidative stress, proinflammatory mediators, and lipid peroxidation in a dose-dependent manner,95 which are all aspects that are deleterious to vasculature and particularly important in ageing.To date, studies investigating acute cocoa flavanol intake in older adults mainly assessed FMD in the BA, with only one study measuring FMD in the lower-limb CFA. These studies typically showed improvements in FMD ranging from 1% to 4.3% in the BA,34,39,44,47,96 and 2.8–2.9% in the CFA within 1–2 hours after flavanol ingestion.47 Comparatively, the FMD differences observed between high- and low-flavanol interventions after 2 hours post-sitting in the present study were of smaller magnitude. This may be a consequence of sitting itself or related to differences in the conduit arteries’ responsiveness. Importantly, flavanol-induced benefits in vascular function are, in some cases, comparable to other physical activity-based interventions that have been applied in older adults during sitting. For example, breaking prolonged sitting every 30 min (with 2 min light-intensity walking) induces a 1.1% increase in SFA FMD,27 and resistance activities induce 0.4% to 3.1%22,23 improvements in SFA FMD in older adults. Interestingly, the current study is the first to show benefits in endothelial function in the BA during sitting, not observed in the context of regular sitting breaks/resistance training activities.22 As such, flavonoid-based nutritional approaches might be ideally used in combination with physical activity breaks to optimise protection of the vasculature from the deleterious effects of sitting in older adults.
Despite improvements in macrovascular function during sitting, flavanols had no impact on microvascular function (e.g., desaturation slope, reperfusion slope) post-sitting or resting tissue oxygenation during sitting. The only significant differences detected between flavanol treatments during sitting (TOI) and post-sitting reactive hyperaemia (maximum TOI) were present also at baseline (0 h), suggesting a random difference in baseline physiology between visits, rather than an effect of flavanol intake. The lack of modulation of microvascular function by flavanols may indicate that different mechanisms may be at play. Whilst flavanols contribute to enhancements in macrovascular function through NO-mediated mechanisms, other vasoactive compounds seem to be more predominantly involved in microvascular function. For example, there is evidence to suggest that the NO contribution to peak reactive hyperaemia in the forearm and cutaneous microvasculature is limited, with other vasoactive factors, including prostaglandins and endothelium-derived hyperpolarising factors, playing a more prominent role.97 Indeed, the literature investigating the acute effects of cocoa flavanols on microvascular function is scarce and particularly discrepant in older populations. For example, one study has reported an increase in haemoglobin oxygen saturation and blood flow in cutaneous tissue,98 whilst other studies have generally reported no changes in microvascular parameters in cutaneous microvasculature, following intake of flavanols.47,99 Similarly, in young populations, some studies report improvements in measures of the peripheral microcirculation, either cutaneous or muscular,36,100 whilst others reported no significant effects during reactive hyperaemia in the forearm skeletal muscle.101 It is possible that these inconsistencies are due to the methods used and location of assessment, so more well-controlled studies are needed to fully establish the potential benefits of cocoa flavanols on peripheral microcirculation. It is also possible that distinct timeframes of exposure to flavanols are needed to achieve benefits within the microvasculature, for example, chronic exposure. More studies are required to establish this.102 Interestingly, previous studies using physical activity interventions during sitting—e.g., leg fidgeting, foot elliptical exercise, and half squats plus calf raises—showed efficacy in attenuating sitting-induced microvascular dysfunction.87–89 As such, dietary flavanols may be used in combination with physical-activity interventions in older adults to modulate both macro and microvascular function during sitting.
Blood pressure
We have observed sitting-induced increases in both systolic and diastolic BP, and declines in heart rate, following both flavanol interventions. Although the research investigating the impact of sitting in older populations is limited, these findings are in agreement with what other studies have reported. For example, some studies have shown that prolonged sitting (7–8 hours) significantly increased systolic and diastolic BP in early middle-aged overweight/obese hypertensive adults103 and late middle-aged/older adults with type 2 diabetes.24 The observed elevations in BP from the current study may potentially be of clinical significance, considering the positive associations between high BP and CVD, CVD-related mortality, and all-cause mortality.104–106 Furthermore, given that our study participants displayed resting systolic BP of approximately 128 mm Hg, the observed sitting-induced increase in systolic BP of >5 mm Hg would move participants towards a higher BP category (from ‘normal’ to ‘high-normal’),48 and may result in a significantly higher risk of major cardiovascular events and mortality if sitting is persistent.107,108 Research indicates that cocoa flavanols can ameliorate BP as shown in a recent systematic review and meta-analysis of 91 RCTs reporting positive effects of flavanols in reducing systolic and diastolic BP of −1.46 mm Hg and −0.99 mm Hg, respectively.41 Several studies have shown improvements in BP among older adults following short-term interventions (e.g.,36,109,110), however, less research has been conducted following acute interventions. Some experimental studies, but not all,111 have shown that acute intake of cocoa flavanols reduces BP measures in middle-aged healthy adults and those with type 2 diabetes.34,47 However, in the present study, acute flavanol intake did not prevent/attenuate sitting-induced increases in BP, suggesting that those benefits may not translate to prolonged sitting contexts in older adults. Further research is needed to establish the link between cocoa flavanols and BP in the context of sitting, particularly following chronic supplementation aimed at counteracting the impact of sedentary lifestyles with diets rich in flavanols.Lifestyle: habitual diet and physical activity
The habitual diet of the study participants is moderately representative of the UK population. All participants exceeded the recommended daily intake of sugars as compared to 61.3% of the British population.112 In addition, ∼80% of the participants consumed at least 5 portions of fruit and vegetables per day, which is better than the UK average of 28%.113 Importantly, participants consumed 870 mg per day of flavonoids, which is in line with reports in a recent large cross-sectional study (805.7 mg per day).114 Given the current dose administered of 695 mg of flavanols, if this was to be maintained when combined with their habitual diet, it would translate into >1500 mg of flavonoids per day. This would have implications for health, given that doses of 1000–1200 mg per day, have been found to be associated with a 14% lower risk of atherosclerotic CVD (as compared to a lower daily intake).115Participants were generally very active: all participants within our sample performed at least 150 minutes per week at moderate intensity, thus meeting the minimum recommendations for physical activity.71 This is higher than the average prevalence in the UK, where only 37% of older adults, aged 65–74 years, are considered physically active.116 In addition, participants took more than 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 steps per day, which is more than the amount of daily steps that healthy older adults generally perform (i.e., 2000–9000 steps per day),117 and above the number of steps that has been recently found to be associated with a progressively lower risk of mortality (i.e., 6000–8000 steps per day).118 However, habitual levels of physical activity or sedentary time do not seem to affect the impact of flavanols on endothelial function during sitting, in agreement with our previous data in young healthy males.51
000 steps per day, which is more than the amount of daily steps that healthy older adults generally perform (i.e., 2000–9000 steps per day),117 and above the number of steps that has been recently found to be associated with a progressively lower risk of mortality (i.e., 6000–8000 steps per day).118 However, habitual levels of physical activity or sedentary time do not seem to affect the impact of flavanols on endothelial function during sitting, in agreement with our previous data in young healthy males.51
In summary, the benefits of flavanols during sitting were demonstrated in a healthy and physically active group of older adults. Future studies should test the efficacy of dietary flavanols in sedentary older adults, more representative of the older adults in the UK.
Limitations
One limitation of the present study is related to the higher discomfort level associated with SFA FMD (vs. BA FMD), in agreement with previous research.119 This led to worsening the quality of arterial images, especially post-occlusion, leading to higher exclusion of analysis and higher variability in this population (inter-day CVSFA FMD = 23.7%). This may have had negative implications for the statistical analysis (e.g., reduced statistical power): based on the current data in older adults (ηp2 = 0.168), a sample size of N = 12–19 (corresponding to power of 80–95%) is needed to detect a significant difference between high- and low-flavanol interventions 2 hours post sitting. Future studies looking to reduce variability in the SFA FMD should consider applying the cuff around the calf instead of the thigh, which is reported to be more comfortable.119 Secondly, although this study included both males and females, it was not sufficiently powered to make comparisons between sexes for the primary and secondary outcome measures. Preliminary analysis including sex as a between-subjects factor indicated no significant impact of sex in the effect of flavanols on endothelial function during sitting. A balanced representation of sexes would also improve the generalizability of the results. Another important limitation is the inclusion of only apparently healthy older adults, as no formal clinical screening was performed. Therefore, the findings may not be generalisable to older individuals with existing or undiagnosed cardiovascular conditions or risk factors. Finally, the participants in this study were highly physically active for their age group, engaging in an average of 2.5 hours per day of moderate-intensity physical activity, which exceeds typical activity levels reported in older adults.120 Therefore, the results may not be representative of more sedentary populations.Conclusion
To the best of our knowledge, the current study is the first to investigate the acute impact of dietary flavanols on macro and microvascular function in response to 2 hours of uninterrupted prolonged sitting in apparently healthy, older adults. Findings showed that sitting induced adverse effects on BA and SFA FMD, resting physiological measures in the SFA, and measures of peripheral muscle oxygenation and BP. Consuming cocoa flavanols at the onset of sitting was effective at preventing sitting-induced declines in FMD across upper- and lower-limb conduit arteries, without affecting microvascular function and BP. Given that sedentary time is highly prevalent among older populations, and may increase their risk of CVD-related conditions, consuming flavanol-rich foods (e.g., cocoa processed to minimize loss of flavanols, green tea, apples, berries) when sedentary may be an effective strategy to be used alone or in conjunction with other approaches (e.g., regular walking breaks, intermittent leg fidgeting) to limit the detrimental effects of inactivity on vascular health.Author contributions
Conceptualisation and funding acquisition: CR. Data collection and data analysis: AD. Writing of original draft: AD. Project administration, supervision, writing, review and editing: CR and SL.Conflicts of interest
All authors declare no conflict of interest.Data availability
The data that support the findings of this study are available from the corresponding author (CR), upon reasonable request.Supplementary information (SI) is available. The SI includes Fig. S1, which presents the Consolidated Standards of Reporting Trials (CONSORT) flow diagram showing inclusion and exclusion of data points for the primary (femoral FMD) and secondary (brachial FMD) outcome measures. See DOI: https://doi.org/10.1039/d5fo02793d.
Acknowledgements
AD’s PhD studentship was fully funded by the College of Life and Environmental Sciences, University of Birmingham (Birmingham, UK), as part of the start-up funds awarded to the PI (CR). The authors would like to thank Barry Callebaut (Leen Allegaert) for providing the high- and low-flavanol cocoa interventions. The graphical abstract has been designed using resources from Flaticon.com.References
- M. S. Tremblay, S. Aubert, J. D. Barnes, T. J. Saunders, V. Carson, A. E. Latimer-Cheung, S. F. M. Chastin, T. M. Altenburg and M. J. M. Chinapaw, Sedentary behavior research network (SBRN)–terminology consensus project process and outcome, Int. J. Behav. Nutr. Phys. Act., 2017, 14, 75 CrossRef PubMed.
- L. F. M. d. Rezende, J. P. Rey-López, V. K. R. Matsudo and O. d. C. Luiz, Sedentary behavior and health outcomes among older adults: a systematic review, BMC Public Health, 2014, 14, 1–9 CrossRef PubMed.
- D. Rosenberg, R. Walker, M. A. Greenwood-Hickman, J. Bellettiere, Y. Xiang, K. Richmire, M. Higgins, D. Wing, E. B. Larson and P. K. Crane, Device-assessed physical activity and sedentary behavior in a community-based cohort of older adults, BMC Public Health, 2020, 20, 1–13 CrossRef PubMed.
- J. Bellettiere, S. Nakandala, F. Tuz-Zahra, E. A. H. Winkler, P. R. Hibbing, G. N. Healy, D. W. Dunstan, N. Owen, M. A. Greenwood-Hickman and D. E. Rosenberg, CHAP-Adult: A Reliable and Valid Algorithm to classify sitting and measure sitting patterns using data from hip-worn accelerometers in adults aged 35+, J. Meas. Phys. Behav., 2022, 5, 215–223 Search PubMed.
- S. Onagbiye, A. Guddemi, O. J. Baruwa, F. Alberti, A. Odone, H. Ricci, M. Gaeta, D. Schmid and C. Ricci, Association of sedentary time with risk of cardiovascular diseases and cardiovascular mortality: A systematic review and meta-analysis of prospective cohort studies, Prev. Med., 2024, 179, 107812 CrossRef PubMed.
- E. G. Wilmot, C. L. Edwardson, F. A. Achana, M. J. Davies, T. Gorely, L. J. Gray, K. Khunti, T. Yates and S. J. H. Biddle, Sedentary time in adults and the association with diabetes, cardiovascular disease and death: systematic review and meta-analysis, Diabetologia, 2012, 55, 2895–2905 CrossRef PubMed.
- N. Owen, G. N. Healy, C. E. Matthews and D. W. Dunstan, Too much sitting: the population health science of sedentary behavior, Exercise Sport Sci. Rev., 2010, 38, 105–113 CrossRef PubMed.
- T. Koyama, N. Kuriyama, E. Ozaki, S. Tomida, R. Uehara, Y. Nishida, C. Shimanoe, A. Hishida, T. Tamura and M. Tsukamoto, Sedentary time is associated with cardiometabolic diseases in a large Japanese population: a cross-sectional study, J. Atheroscler. Thromb., 2020, 27, 1097–1107 Search PubMed.
- R. T. Mankowski, M. Aubertin-Leheudre, D. P. Beavers, A. Botoseneanu, T. W. Buford, T. Church, N. W. Glynn, A. C. King, C. Liu and T. M. Manini, Sedentary time is associated with the metabolic syndrome in older adults with mobility limitations—The LIFE Study, Exp. Gerontol., 2015, 70, 32–36 CrossRef PubMed.
- K. N. Alme, A.-B. Knapskog, H. Næss, M. Naik, M. Beyer, H. Ellekjaer, C. English, H. I. Hansen, C. S. Kummeneje and R. Munthe-Kaas, Is long-bout sedentary behaviour associated with long-term glucose levels 3 months after acute ischaemic stroke? A prospective observational cohort study, BMJ Open, 2020, 10, e037475 CrossRef PubMed.
- W. Jia, W. He, Q. Cui, X. Ye and H. Qian, The effect of sedentary time on cardiovascular disease risk in middle-aged and elderly patients with type 2 diabetes mellitus, Medicine, 2023, 102, e35901 CrossRef PubMed.
- M. McCarthy, C. L. Edwardson, M. J. Davies, J. Henson, L. Gray, K. Khunti and T. Yates, Change in sedentary time, physical activity, bodyweight, and HbA1c in high-risk adults, Med. Sci. Sports Exercise, 2017, 49, 1120 CrossRef PubMed.
- D. Hupin, F. Roche, V. Gremeaux, J.-C. Chatard, M. Oriol, J.-M. Gaspoz, J.-C. Barthélémy and P. Edouard, Even a low-dose of moderate-to-vigorous physical activity reduces mortality by 22% in adults aged≥ 60 years: a systematic review and meta-analysis, Br. J. Sports Med., 2015, 49, 1262–1267 CrossRef PubMed.
- Q. Liu, F.-C. Liu, K.-Y. Huang, J.-X. Li, X.-L. Yang, X.-Y. Wang, J.-C. Chen, X.-Q. Liu, J. Cao and C. Shen, Beneficial effects of moderate to vigorous physical activity on cardiovascular disease among Chinese adults, J. Geriatr. Cardiol., 2020, 17, 85 Search PubMed.
- P. D. Loprinzi, Sedentary behavior and predicted 10 year risk for a first atherosclerotic cardiovascular disease (ASCVD) event using the pooled cohort risk equations among US adults, Int. J. Cardiol., 2016, 203, 443–444 CrossRef PubMed.
- J. R. Wilmoth, D. Bas, S. Mukherjee and N. Hanif, World social report 2023: Leaving no one behind in an ageing world, UN, 2023 Search PubMed.
- A. Daniele, S. J. E. Lucas and C. Rendeiro, Detrimental effects of physical inactivity on peripheral and brain vasculature in humans: Insights into mechanisms, long-term health consequences and protective strategies, Front. Physiol., 2022, 13, 998380 CrossRef PubMed.
- C. Paterson, S. Fryer, G. Zieff, K. Stone, D. P. Credeur, B. Barone Gibbs, J. Padilla, J. K. Parker and L. Stoner, The effects of acute exposure to prolonged sitting, with and without interruption, on vascular function among adults: a meta-analysis, Sports Med., 2020, 50, 1929–1942 CrossRef PubMed.
- T. Morishima, R. M. Restaino, L. K. Walsh, J. A. Kanaley and J. Padilla, Prior exercise and standing as strategies to circumvent sitting-induced leg endothelial dysfunction, Clin. Sci., 2017, 131, 1045–1053 CrossRef.
- M. W. O'Brien, J. A. Johns, T. D. Williams and D. S. Kimmerly, Sex does not influence impairments in popliteal endothelial-dependent vasodilator or vasoconstrictor responses following prolonged sitting, J. Appl. Physiol., 2019, 127, 679–687 CrossRef PubMed.
- S. S. Thosar, S. L. Bielko, K. J. Mather, J. D. Johnston and J. P. Wallace, Effect of prolonged sitting and breaks in sitting time on endothelial function, 2015 Search PubMed.
- R. E. Climie, M. J. Wheeler, M. Grace, E. A. Lambert, N. Cohen, N. Owen, B. A. Kingwell, D. W. Dunstan and D. J. Green, Simple intermittent resistance activity mitigates the detrimental effect of prolonged unbroken sitting on arterial function in overweight and obese adults, J. Appl. Physiol., 2018, 125, 1787–1794 CrossRef PubMed.
- F. C. Taylor, D. W. Dunstan, A. R. Homer, P. C. Dempsey, B. A. Kingwell, R. E. Climie, N. Owen, N. D. Cohen, R. N. Larsen and M. Grace, Acute effects of interrupting prolonged sitting on vascular function in type 2 diabetes, Am. J. Physiol.: Heart Circ. Physiol., 2021, 320, H393–H403 CrossRef PubMed.
- P. C. Dempsey, J. W. Sacre, R. N. Larsen, N. E. Straznicky, P. Sethi, N. D. Cohen, E. Cerin, G. W. Lambert, N. Owen and B. A. Kingwell, Interrupting prolonged sitting with brief bouts of light walking or simple resistance activities reduces resting blood pressure and plasma noradrenaline in type 2 diabetes, J. Hypertens., 2016, 34, 2376–2382 CrossRef PubMed.
- G. W. Somes, M. Pahor, R. I. Shorr, W. C. Cushman and W. B. Applegate, The role of diastolic blood pressure when treating isolated systolic hypertension, Arch. Intern. Med., 1999, 159, 2004–2009 CrossRef PubMed.
- S. P. Whelton, J. W. McEvoy, L. Shaw, B. M. Psaty, J. A. C. Lima, M. Budoff, K. Nasir, M. Szklo, R. S. Blumenthal and M. J. Blaha, Association of normal systolic blood pressure level with cardiovascular disease in the absence of risk factors, JAMA Cardiol., 2020, 5, 1011–1018 Search PubMed.
- Y. A. W. Hartman, L. C. M. Tillmans, D. L. Benschop, A. N. L. Hermans, K. M. R. Nijssen, T. M. H. Eijsvogels, P. H. G. M. Willems, C. J. Tack, M. T. E. Hopman, J. A. H. R. Claassen and D. H. J. Thijssen, Long-Term and Acute Benefits of Reduced Sitting on Vascular Flow and Function, Med. Sci. Sports Exercise, 2021, 53, 341–350 CrossRef PubMed.
- D. E. Rosenberg, W. Zhu, M. A. Greenwood-Hickman, A. J. Cook, S. F. Acevedo, J. B. McClure, D. E. Arterburn, J. Cooper, N. Owen and D. Dunstan, Sitting time reduction and blood pressure in older adults: a randomized clinical trial, JAMA Netw. Open, 2024, 7, e243234–e243234 CrossRef PubMed.
- R. N. Larsen, B. A. Kingwell, P. Sethi, E. Cerin, N. Owen and D. W. Dunstan, Breaking up prolonged sitting reduces resting blood pressure in overweight/obese adults, Nutr., Metab. Cardiovasc. Dis., 2014, 24, 976–982 CrossRef PubMed.
- Z. S. Zeigler, P. D. Swan, D. M. Bhammar and G. A. Gaesser, Walking workstation use reduces ambulatory blood pressure in adults with prehypertension, J. Phys. Act. Health, 2015, 12, S119–S127 Search PubMed.
- L. Chiavaroli, E. Viguiliouk, S. K. Nishi, S. Blanco Mejia, D. Rahelić, H. Kahleová, J. Salas-Salvadó, C. W. C. Kendall and J. L. Sievenpiper, DASH dietary pattern and cardiometabolic outcomes: an umbrella review of systematic reviews and meta-analyses, Nutrients, 2019, 11, 338 CrossRef PubMed.
- M. A. Martínez-González, A. Gea and M. Ruiz-Canela, The Mediterranean diet and cardiovascular health: A critical review, Circ. Res., 2019, 124, 779–798 Search PubMed.
- M. Ebaditabar, K. Djafarian, N. Saeidifard and S. Shab-Bidar, Effect of dark chocolate on flow-mediated dilatation: Systematic review, meta-analysis, and dose–response analysis of randomized controlled trials, Clin. Nutr. ESPEN, 2020, 36, 17–27 Search PubMed.
- Z. Faridi, V. Y. Njike, S. Dutta, A. Ali and D. L. Katz, Acute dark chocolate and cocoa ingestion and endothelial function: a randomized controlled crossover trial1, Am. J. Clin. Nutr., 2008, 88, 58–63 Search PubMed.
- M. Gröne, R. Sansone, P. Höffken, P. Horn, A. Rodriguez-Mateos, H. Schroeter, M. Kelm and C. Heiss, Cocoa flavanols improve endothelial functional integrity in healthy young and elderly subjects, J. Agric. Food Chem., 2019, 68, 1871–1876 Search PubMed.
- C. Heiss, R. Sansone, H. Karimi, M. Krabbe, D. Schuler, A. Rodriguez-Mateos, T. Kraemer, M. M. Cortese-Krott, G. G. C. Kuhnle and J. P. E. Spencer, Impact of cocoa flavanol intake on age-dependent vascular stiffness in healthy men: a randomized, controlled, double-masked trial, Age, 2015, 37, 56 CrossRef PubMed.
- L. Hooper, C. Kay, A. Abdelhamid, P. A. Kroon, J. S. Cohn, E. B. Rimm and A. Cassidy, Effects of chocolate, cocoa, and flavan-3-ols on cardiovascular health: a systematic review and meta-analysis of randomized trials, Am. J. Clin. Nutr., 2012, 95, 740–751 CrossRef PubMed.
- F. Jalili, S. Moradi, S. Talebi, S. Mehrabani, S. M. Ghoreishy, A. Wong, A. R. Jalalvand, M. A. H. Kermani, C. Jalili and F. Jalili, The effects of citrus flavonoids supplementation on endothelial function: A systematic review and dose–response meta–analysis of randomized clinical trials, Phytother. Res., 2024, 38, 2847–2859 Search PubMed.
- K. D. Monahan, R. P. Feehan, A. R. Kunselman, A. G. Preston, D. L. Miller and M. E. J. Lott, Dose-dependent increases in flow-mediated dilation following acute cocoa ingestion in healthy older adults, J. Appl. Physiol., 2011, 111, 1568–1574 CrossRef PubMed.
- B. H. Parmenter, K. D. Croft, L. Cribb, M. B. Cooke, C. P. Bondonno, A. Lea, G. M. McPhee, M. Komanduri, K. Nolidin and K. Savage, Higher habitual dietary flavonoid intake associates with lower central blood pressure and arterial stiffness in healthy older adults, Br. J. Nutr., 2022, 128, 279–289 CrossRef PubMed.
- G. Raman, E. E. Avendano, S. Chen, J. Wang, J. Matson, B. Gayer, J. A. Novotny and A. Cassidy, Dietary intakes of flavan-3-ols and cardiometabolic health: systematic review and meta-analysis of randomized trials and prospective cohort studies, Am. J. Clin. Nutr., 2019, 110, 1067–1078 CrossRef PubMed.
- K. Ried, T. R. Sullivan, P. Fakler, O. R. Frank and N. P. Stocks, Effect of cocoa on blood pressure, Cochrane Database Syst. Rev., 2012, CD008893 Search PubMed.
- C. Manach, A. Scalbert, C. Morand, C. Rémésy and L. Jiménez, Polyphenols: food sources and bioavailability, Am. J. Clin. Nutr., 2004, 79, 727–747 Search PubMed.
- J. I. Dower, J. M. Geleijnse, P. A. Kroon, M. Philo, M. Mensink, D. Kromhout and P. C. H. Hollman, Does epicatechin contribute to the acute vascular function effects of dark chocolate? A randomized, crossover study, Mol. Nutr. Food Res., 2016, 60, 2379–2386 CrossRef PubMed.
- L. Loffredo, L. Perri, E. Catasca, P. Pignatelli, M. Brancorsini, C. Nocella, E. De Falco, S. Bartimoccia, G. Frati and R. Carnevale, Dark chocolate acutely improves walking autonomy in patients with peripheral artery disease, J. Am. Heart Assoc., 2014, 3, e001072 Search PubMed.
- H. Schroeter, C. Heiss, J. Balzer, P. Kleinbongard, C. L. Keen, N. K. Hollenberg, H. Sies, C. Kwik-Uribe, H. H. Schmitz and M. Kelm, (–)-Epicatechin mediates beneficial effects of flavanol-rich cocoa on vascular function in humans, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 1024–1029 Search PubMed.
- M. Bapir, G. R. Untracht, D. Cooke, J. H. McVey, S. S. Skene, P. Campagnolo, M. B. Whyte, N. Dikaios, A. Rodriguez-Mateos and D. D. Sampson, Cocoa flavanol consumption improves lower extremity endothelial function in healthy individuals and people with type 2 diabetes, Food Funct., 2022, 13, 10439–10448 Search PubMed.
- G. Mancia, R. Kreutz, M. Brunström, M. Burnier, G. Grassi, A. Januszewicz, M. L. Muiesan, K. Tsioufis, E. Agabiti-Rosei and E. A. E. Algharably, 2023 ESH Guidelines for the management of arterial hypertension The Task Force for the management of arterial hypertension of the European Society of Hypertension: Endorsed by the International Society of Hypertension (ISH) and the European Renal Association (ERA), J. Hypertens., 2023, 41, 1874–2071 CrossRef PubMed.
- R. Baynham, J. J. C. S. V. van Zanten and C. Rendeiro, Cocoa flavanols rescue stress-induced declines in endothelial function after a high-fat meal, but do not affect cerebral oxygenation during stress in young, healthy adults, Food Funct., 2024, 15, 11472–11490 RSC.
- D. J. Lamport, D. Pal, C. Moutsiana, D. T. Field, C. M. Williams, J. P. E. Spencer and L. T. Butler, The effect of flavanol-rich cocoa on cerebral perfusion in healthy older adults during conscious resting state: a placebo controlled, crossover, acute trial, Psychopharmacology, 2015, 232, 3227–3234 CrossRef PubMed.
- A. Daniele, S. J. E. Lucas and C. Rendeiro, Dietary flavanols preserve upper- and lower-limb endothelial function during sitting in high- and low-fit young healthy males, J. Physiol., 2026, 604, 145–171 Search PubMed.
- A. González-Sarrías, J. C. Espín and F. A. Tomás-Barberán, Non-extractable polyphenols produce gut microbiota metabolites that persist in circulation and show anti-inflammatory and free radical-scavenging effects, Trends Food Sci. Technol., 2017, 69, 281–288 CrossRef.
- G. Borges, J. I. Ottaviani, J. J. J. van der Hooft, H. Schroeter and A. Crozier, Absorption, metabolism, distribution and excretion of (−)-epicatechin: A review of recent findings, Mol. Aspects Med., 2018, 61, 18–30 Search PubMed.
- R. A. Harris, S. K. Nishiyama, D. W. Wray and R. S. Richardson, Ultrasound assessment of flow-mediated dilation, Hypertension, 2010, 55, 1075–1085 CrossRef CAS PubMed.
- D. H. J. Thijssen, R. M. Bruno, A. C. C. M. van Mil, S. M. Holder, F. Faita, A. Greyling, P. L. Zock, S. Taddei, J. E. Deanfield and T. Luscher, Expert consensus and evidence-based recommendations for the assessment of flow-mediated dilation in humans, Eur. Heart J., 2019, 40, 2534–2547 Search PubMed.
- S. A. Bingham, A. A. Welch, A. McTaggart, A. A. Mulligan, S. A. Runswick, R. Luben, S. Oakes, K. T. Khaw, N. Wareham and N. E. Day, Nutritional methods in the European prospective investigation of cancer in Norfolk, Public Health Nutr., 2001, 4, 847–858 CrossRef PubMed.
- A. A. Mulligan, R. N. Luben, A. Bhaniani, D. J. Parry-Smith, L. O'Connor, A. P. Khawaja, N. G. Forouhi and K.-T. Khaw, A new tool for converting food frequency questionnaire data into nutrient and food group values: FETA research methods and availability, BMJ Open, 2014, 4, e004503 CrossRef PubMed.
- N. H. S. NHS, 5 A Day portion sizes, https://www.nhs.uk/live-well/eat-well/5-a-day/portion-sizes/.
- G. Gratton, S. R. Weaver, C. V. Burley, K. A. Low, E. L. Maclin, P. W. Johns, Q. S. Pham, S. J. E. Lucas, M. Fabiani and C. Rendeiro, Dietary flavanols improve cerebral cortical oxygenation and cognition in healthy adults, Sci. Rep., 2020, 10, 19409 Search PubMed.
- F. A. Alsolmei, H. Li, S. L. Pereira, P. Krishnan, P. W. Johns and R. A. Siddiqui, Polyphenol-enriched plum extract enhances myotubule formation and anabolism while attenuating colon cancer-induced cellular damage in C2C12 cells, Nutrients, 2019, 11, 1077 CrossRef PubMed.
- R. J. Robbins, J. Leonczak, J. Li, J. C. Johnson, T. Collins, C. Kwik-Uribe and H. H. Schmitz, Determination of flavanol and procyanidin (by degree of polymerization 1–10) content of chocolate, cocoa liquors, powder (s), and cocoa flavanol extracts by normal phase high-performance liquid chromatography: collaborative study, J. AOAC Int., 2012, 95, 1153–1160 CrossRef PubMed.
- K. B. Miller, W. J. Hurst, M. J. Payne, D. A. Stuart, J. Apgar, D. S. Sweigart and B. Ou, Impact of alkalization on the antioxidant and flavanol content of commercial cocoa powders, J. Agric. Food Chem., 2008, 56, 8527–8533 CrossRef PubMed.
- R. L. Hoiland, P. N. Ainslie, K. W. Wildfong, K. J. Smith, A. R. Bain, C. K. Willie, G. Foster, B. Monteleone and T. A. Day, Indomethacin–induced impairment of regional cerebrovascular reactivity: implications for respiratory control, J. Physiol., 2015, 593, 1291–1306 Search PubMed.
- E. Seidel, B. M. Eicke, B. Tettenborn and F. Krummenauer, Reference values for vertebral artery flow volume by duplex sonography in young and elderly adults, Stroke, 1999, 30, 2692–2696 Search PubMed.
- G. Atkinson and A. M. Batterham, Allometric scaling of diameter change in the original flow-mediated dilation protocol, Atherosclerosis, 2013, 226, 425–427 Search PubMed.
- G. Atkinson and A. M. Batterham, The percentage flow-mediated dilation index: a large-sample investigation of its appropriateness, potential for bias and causal nexus in vascular medicine, Vasc. Med., 2013, 18, 354–365 Search PubMed.
- K. E. Pyke and M. E. Tschakovsky, The relationship between shear stress and flow–mediated dilatation: implications for the assessment of endothelial function, J. Physiol., 2005, 568, 357–369 CrossRef PubMed.
- D. J. Davies, Z. Su, M. T. Clancy, S. J. E. Lucas, H. Dehghani, A. Logan and A. Belli, Near-infrared spectroscopy in the monitoring of adult traumatic brain injury: a review, J. Neurotrauma, 2015, 32, 933–941 CrossRef PubMed.
- M. J. Blanca Mena, R. Alarcón Postigo, J. Arnau Gras, R. Bono Cabré and R. Bendayan, Non-normal data: Is ANOVA still a valid option?, Psicothema, 2017, 29(4), 552–557 CrossRef PubMed.
- G. Norman, Likert scales, levels of measurement and the “laws” of statistics, Adv. Health Sci. Educ., 2010, 15, 625–632 CrossRef PubMed.
- F. C. Bull, S. S. Al-Ansari, S. Biddle, K. Borodulin, M. P. Buman, G. Cardon, C. Carty, J.-P. Chaput, S. Chastin and R. Chou, World Health Organization 2020 guidelines on physical activity and sedentary behaviour, Br. J. Sports Med., 2020, 54, 1451–1462 CrossRef PubMed.
- N. H. S. NHS, Eating a balanced diet, https://www.nhs.uk/live-well/eat-well/how-to-eat-a-balanced-diet/eating-a-balanced-diet/#:~:text=eat%20at%20least%205%20portions,food%20you%20eat%20each%20day.
- D. J. Green, H. Jones, D. Thijssen, N. T. Cable and G. Atkinson, Flow-mediated dilation and cardiovascular event prediction: does nitric oxide matter?, Hypertension, 2011, 57, 363–369 CrossRef PubMed.
- K. D. Ballard, R. M. Duguid, C. W. Berry, P. Dey, R. S. Bruno, R. M. Ward and K. L. Timmerman, Effects of prior aerobic exercise on sitting-induced vascular dysfunction in healthy men, Eur. J. Appl. Physiol., 2017, 117, 2509–2518 Search PubMed.
- R. M. Restaino, S. W. Holwerda, D. P. Credeur, P. J. Fadel and J. Padilla, Impact of prolonged sitting on lower and upper limb micro–and macrovascular dilator function, Exp. Physiol., 2015, 100, 829–838 CrossRef PubMed.
- S. S. Thosar, S. L. Bielko, C. C. Wiggins and J. P. Wallace, Differences in brachial and femoral artery responses to prolonged sitting, Cardiovasc. Ultrasound, 2014, 12, 1–7 CrossRef PubMed.
- S. S. Thosar, S. L. Bielko, C. S. Wiggins, J. E. Klaunig, K. J. Mather and J. P. Wallace, Antioxidant vitamin C prevents decline in endothelial function during sitting, Med. Sci. Monit., 2015, 21, 1015 CrossRef PubMed.
- S. M. Holder, E. A. Dawson, Á. Brislane, J. Hisdal, D. J. Green and D. H. J. Thijssen, Fluctuation in shear rate, with unaltered mean shear rate, improves brachial artery flow-mediated dilation in healthy, young men, J. Appl. Physiol., 2019, 126, 1687–1693 CrossRef PubMed.
- J. C. Tremblay and K. E. Pyke, Flow-mediated dilation stimulated by sustained increases in shear stress: a useful tool for assessing endothelial function in humans?, Am. J. Physiol.: Heart Circ. Physiol., 2018, 314, H508–H520 Search PubMed.
- D. Lu and G. S. Kassab, Role of shear stress and stretch in vascular mechanobiology, J. R. Soc., Interface, 2011, 8, 1379–1385 CrossRef PubMed.
- J. J. Paszkowiak and A. Dardik, Arterial wall shear stress: observations from the bench to the bedside, Vasc. Endovasc. Surg., 2003, 37, 47–57 CrossRef PubMed.
- D. K. Townsend, D. M. Deysher, E. E. Wu and T. J. Barstow, Reduced insulin sensitivity in young, normoglycaemic subjects alters microvascular tissue oxygenation during postocclusive reactive hyperaemia, Exp. Physiol., 2019, 104, 967–974 CrossRef PubMed.
- N. I. Shapiro, R. Arnold, R. Sherwin, J. O'Connor, G. Najarro, S. Singh, D. Lundy, T. Nelson, S. W. Trzeciak and A. E. Jones, The association of near-infrared spectroscopy-derived tissue oxygenation measurements with sepsis syndromes, organ dysfunction and mortality in emergency department patients with sepsis, Crit. Care, 2011, 15, 1–10 CrossRef PubMed.
- V. d. S. Pinheiro, A. C. F. da Silva Tavares, M. Volino-Souza, G. V. de Oliveira and T. S. Alvares, Association between femoral artery flow-mediated dilation and muscle oxygen saturation parameters in healthy, young individuals, J. Cardiovasc. Dev. Dis., 2023, 10, 63 Search PubMed.
- C. P. Anderson and S.-Y. Park, Attenuated reactive hyperemia after prolonged sitting is associated with reduced local skeletal muscle metabolism: insight from artificial intelligence, Am. J. Physiol.: Regul., Integr. Comp. Physiol., 2023, 325, R380–R388 CrossRef PubMed.
- R. J. Headid, 3rd, E. J. Pekas, T. K. Wooden, W.-M. Son, G. Layec, J. Shin and S.-Y. Park, Impacts of prolonged sitting with mild hypercapnia on vascular and autonomic function in healthy recreationally active adults, Am. J. Physiol.: Heart Circ. Physiol., 2020, 319, H468–H480 CrossRef PubMed.
- M. Horiuchi and L. Stoner, Macrovascular and microvascular responses to prolonged sitting with and without bodyweight exercise interruptions: a randomized cross-over trial, Vasc. Med., 2022, 27, 127–135 Search PubMed.
- S.-Y. Park, T. K. Wooden, E. J. Pekas, C. P. Anderson, S. K. Yadav, D. R. Slivka and G. Layec, Effects of passive and active leg movements to interrupt sitting in mild hypercapnia on cardiovascular function in healthy adults, J. Appl. Physiol., 2022, 132, 874–887 CrossRef PubMed.
- H. Tamiya, K. Hotta, M. Hoshiai, Y. Kurosawa, T. Hamaoka and A. Tsubaki, Leg fidgeting alleviates reduced oxygen extraction in the gastrocnemius muscle caused by prolonged sitting: a randomized crossover trial, Eur. J. Appl. Physiol., 2024, 1–13 Search PubMed.
- W. S. Evans, L. Stoner, Q. Willey, E. Kelsch, D. P. Credeur and E. D. Hanson, Local exercise does not prevent the aortic stiffening response to acute prolonged sitting: a randomized crossover trial, J. Appl. Physiol., 2019, 127, 781–787 CrossRef PubMed.
- L. Stoner, Q. Willey, W. S. Evans, K. Burnet, D. P. Credeur, S. Fryer and E. D. Hanson, Effects of acute prolonged sitting on cerebral perfusion and executive function in young adults: A randomized cross–over trial, Psychophysiology, 2019, 56, e13457 CrossRef PubMed.
- M. Gayda, M. Juneau, J.-C. Tardif, F. Harel, S. Levesque and A. Nigam, Cardiometabolic and traditional cardiovascular risk factors and their potential impact on macrovascular and microvascular function: preliminary data, Clin. Hemorheol. Microcirc., 2015, 59, 53–65 CrossRef PubMed.
- W. M. Loke, J. M. Hodgson, J. M. Proudfoot, A. J. McKinley, I. B. Puddey and K. D. Croft, Pure dietary flavonoids quercetin and (−)-epicatechin augment nitric oxide products and reduce endothelin-1 acutely in healthy men, Am. J. Clin. Nutr., 2008, 88, 1018–1025 CrossRef PubMed.
- D. J. Green, E. A. Dawson, H. M. M. Groenewoud, H. Jones and D. H. J. Thijssen, Is flow-mediated dilation nitric oxide mediated? A meta-analysis, Hypertension, 2014, 63, 376–382 CrossRef PubMed.
- D. Grassi, F. Mai, M. De Feo, R. Barnabei, A. Carducci, G. Desideri, S. Necozione, L. Allegaert, H. Bernaert and C. Ferri, Cocoa consumption decreases oxidative stress, proinflammatory mediators and lipid peroxidation in healthy subjects: A randomized placebo-controlled dose-response clinical trial, High Blood Pressure Cardiovasc. Prev., 2023, 30, 219–225 CrossRef PubMed.
- C. E. Marsh, H. H. Carter, K. J. Guelfi, K. J. Smith, K. E. Pike, L. H. Naylor and D. J. Green, Brachial and cerebrovascular functions are enhanced in postmenopausal women after ingestion of chocolate with a high concentration of cocoa, J. Nutr., 2017, 147, 1686–1692 CrossRef.
- R. Rosenberry and M. D. Nelson, Reactive hyperemia: a review of methods, mechanisms, and considerations, Am. J. Physiol.: Regul., Integr. Comp. Physiol., 2020, 318, R605–R618 CrossRef PubMed.
- K. Neukam, W. Stahl, H. Tronnier, H. Sies and U. Heinrich, Consumption of flavanol-rich cocoa acutely increases microcirculation in human skin, Eur. J. Nutr., 2007, 46, 53–56 CrossRef PubMed.
- A. Hammer, R. Koppensteiner, S. Steiner, A. Niessner, G. Goliasch, M. Gschwandtner and M. Hoke, Dark chocolate and vascular function in patients with peripheral artery disease: A randomized, controlled cross-over trial, Clin. Hemorheol. Microcirc., 2015, 59, 145–153 CrossRef PubMed.
- R. Baynham, J. J. C. S. Veldhuijzen van Zanten, P. W. Johns, Q. S. Pham and C. Rendeiro, Cocoa flavanols improve vascular responses to acute mental stress in young healthy adults, Nutrients, 2021, 13, 1103 CrossRef PubMed.
- K. S. d. Santos, O. J. F. R. Junior, I. R. G. Tavares, M. Volino-Souza, G. V. d. Oliveira and T. d. S. Alvares, A single dose of microencapsulated cocoa supplementation attenuated eccentric exercise-induced endothelial dysfunction, Int. J. Food Sci. Nutr., 2023, 74, 373–381 CrossRef.
- S. Richardson, J. Marshall and C. Rendeiro, The role of cocoa flavanols in modulating peripheral and cerebral microvascular function in healthy individuals and populations at-risk of cardiovascular disease: a systematic review, Nutr. J., 2025, 24, 57 CrossRef PubMed.
- A. B. Alansare, R. J. Kowalsky, M. A. Jones, S. J. Perdomo, L. Stoner and B. B. Gibbs, The Effects of a Simulated Workday of Prolonged Sitting on Seated versus Supine Blood Pressure and Pulse Wave Velocity in Adults with Overweight/Obesity and Elevated Blood Pressure, J. Vasc. Res., 2020, 57, 355–366 CrossRef PubMed.
- R. S. Vasan, M. G. Larson, E. P. Leip, J. C. Evans, C. J. O'Donnell, W. B. Kannel and D. Levy, Impact of high-normal blood pressure on the risk of cardiovascular disease, N. Engl. J. Med., 2001, 345, 1291–1297 CrossRef PubMed.
- C.-Y. Wu, H.-Y. Hu, Y.-J. Chou, N. Huang, Y.-C. Chou and C.-P. Li, High blood pressure and all-cause and cardiovascular disease mortalities in community-dwelling older adults, Medicine, 2015, 94, e2160 CrossRef PubMed.
- J. Wu, X. Han, D. Sun, J. Zhang, J. Li, G. Qin, W. Deng, Y. Yu and H. Xu, Age-specific association of stage of hypertension at diagnosis with cardiovascular and all-cause mortality among elderly patients with hypertension: a cohort study, BMC Cardiovasc. Disord., 2023, 23, 270 CrossRef PubMed.
- K. Rahimi, Z. Bidel, M. Nazarzadeh, E. Copland, D. Canoy, M. Wamil, J. Majert, R. McManus, A. Adler and L. Agodoa, Age-stratified and blood-pressure-stratified effects of blood-pressure-lowering pharmacotherapy for the prevention of cardiovascular disease and death: an individual participant-level data meta-analysis, Lancet, 2021, 398, 1053–1064 CrossRef PubMed.
- Y. Tabara, Prognostic significance of blood pressure in frail older adults, Hypertens. Res., 2022, 45, 378–379 CrossRef PubMed.
- K. Davison, N. M. Berry, G. Misan, A. M. Coates, J. D. Buckley and P. R. C. Howe, Dose-related effects of flavanol-rich cocoa on blood pressure, J. Hum. Hypertens., 2010, 24, 568–576 Search PubMed.
- D. Taubert, R. Roesen, C. Lehmann, N. Jung and E. Schömig, Effects of low habitual cocoa intake on blood pressure and bioactive nitric oxide: a randomized controlled trial, J. Am. Med. Assoc., 2007, 298, 49–60 CrossRef PubMed.
- A. Tanghe, E. Heyman, E. Lespagnol, J. Stautemas, B. Celie, J. Op‘t Roodt, E. Rietzschel, D. Dias Soares, N. Hermans and E. Tuenter, Acute effects of cocoa flavanols on blood pressure and peripheral vascular reactivity in type 2 diabetes mellitus and essential hypertension, Nutrients, 2022, 14, 2692 CrossRef PubMed.
- F. Rauber, M. L. da Costa Louzada, E. M. Steele, L. F. M. de Rezende, C. Millett, C. A. Monteiro and R. B. Levy, Ultra-processed foods and excessive free sugar intake in the UK: a nationally representative cross-sectional study, BMJ Open, 2019, 9, e027546 CrossRef PubMed.
- N. H. S. NHS, Statistics on Obesity, Physical Activity and Diet, England, 2020 Search PubMed.
- A. S. Thompson, A. Jennings, N. P. Bondonno, A. Tresserra-Rimbau, B. H. Parmenter, C. Hill, A. Perez-Cornago, T. Kühn and A. Cassidy, Higher habitual intakes of flavonoids and flavonoid-rich foods are associated with a lower incidence of type 2 diabetes in the UK Biobank cohort, Nutr. Diabetes, 2024, 14, 32 CrossRef PubMed.
- F. Dalgaard, N. P. Bondonno, K. Murray, C. P. Bondonno, J. R. Lewis, K. D. Croft, C. Kyrø, G. Gislason, A. Scalbert and A. Cassidy, Associations between habitual flavonoid intake and hospital admissions for atherosclerotic cardiovascular disease: a prospective cohort study, Lancet Planetary Health, 2019, 3, e450–e459 CrossRef PubMed.
- W. H. O. WHO, Physical activity factsheet, 2014 Search PubMed.
- C. Tudor-Locke, C. L. Craig, Y. Aoyagi, R. C. Bell, K. A. Croteau, I. De Bourdeaudhuij, B. Ewald, A. W. Gardner, Y. Hatano and L. D. Lutes, How many steps/day are enough? For older adults and special populations, Int. J. Behav. Nutr. Phys. Act., 2011, 8, 1–19 CrossRef PubMed.
- A. E. Paluch, S. Bajpai, D. R. Bassett, M. R. Carnethon, U. Ekelund, K. R. Evenson, D. A. Galuska, B. J. Jefferis, W. E. Kraus and I. M. Lee, Daily steps and all-cause mortality: a meta-analysis of 15 international cohorts, Lancet Public Health, 2022, 7, e219–e228 CrossRef PubMed.
- M. Bapir, G. R. Untracht, J. E. A. Hunt, J. H. McVey, J. Harris, S. S. Skene, P. Campagnolo, N. Dikaios, A. Rodriguez-Mateos and D. D. Sampson, Age-dependent decline in common femoral artery flow-mediated dilation and wall shear stress in healthy subjects, Life, 2022, 12, 2023 CrossRef PubMed.
- M. Menai, V. T. Van Hees, A. Elbaz, M. Kivimaki, A. Singh-Manoux and S. Sabia, Accelerometer assessed moderate-to-vigorous physical activity and successful ageing: results from the Whitehall II study, Sci. Rep., 2017, 7, 45772 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |