Open Access Article
Open Access ArticleL-Arabinose attenuates loperamide-induced constipation through the regulation of gut microbiota in mice
Simin
Zheng
a,
Hao
Zheng
b,
Yangru
Huang
a,
Bowen
Liang
a,
Yingfei
Gong
c,
Jinhai
Shen
a,
Wenrui
Liu
d,
Xiaona
Xu
a,
Yuliang
Ma
e,
Zhen
Liu
f and
Yun
Wang
 *abc
*abc
aState Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing, Jiangsu 211198, PR China. E-mail: wangyun@cpu.edu.cn; Fax: +(86)-25-86185622; Tel: +(86)-25-86185619
bSchool of Basic Medicine and Clinical Pharmacy, China Pharmaceutical University, Nanjing, Jiangsu 211198, PR China
cCenter for New Drug Safety Evaluation and Research, China Pharmaceutical University, Nanjing, Jiangsu 211198, PR China
dSchool of Life Science and Technology, China Pharmaceutical University, Nanjing, Jiangsu 211198, PR China
eSchool of Pharmacy, China Pharmaceutical University, Nanjing, Jiangsu 211198, PR China
fSchool of Pharmacy, Jiangxi University of Chinese Medicine, Nanchang, Jiangxi 330004, PR China
First published on 3rd February 2026
Abstract
L-Arabinose is a plant-specific pentaose with various functional properties. Here, the effects of L-arabinose on long-term constipation were investigated. According to the results, L-arabinose significantly attenuated loperamide-induced constipation by enhancing the gastrointestinal motility and relieving intestinal injuries. In addition, L-arabinose reduced neutrophil proportion and inflammatory cytokine concentrations in constipated mice, exhibiting immunomodulatory activity. Furthermore, the 16S rRNA analysis showed that L-arabinose ameliorated the gut microbiota dysbiosis induced by loperamide, and Lactobacillus was the most significant dominant strain at the genus level in the L-arabinose group. More importantly, the effect of L-arabinose was diminished by gut microbiota depletion, further confirming the necessity of gut microbiota in L-arabinose-initiated constipation treatment. Mechanistically, L-arabinose increased the abundance of Lactobacillus, ameliorating the gut microbiota dysbiosis, which further attenuated the constipation by enhancing the gastrointestinal motility and relieving intestinal injuries. In summary, L-arabinose can ameliorate loperamide-induced constipation in mice through the regulation of gut microbiota.
1. Introduction
Constipation is a common gastrointestinal disorder that is prevalent worldwide, characterized by hard stools, infrequent bowel movements, and incomplete bowel evacuation.1,2 Regardless of affecting the quality of life, constipation may lead to gastrointestinal nerve dysfunction, gut microbiota disorders, hepatic encephalopathy, cardio-cerebrovascular diseases, and even fatal pulmonary embolism (PE) or colorectal cancer (CRC).3–6 Although the global cost of treating constipation is high every year, more than 50% of people with constipation cannot be cured.7,8 Thus, developing effective strategies for the management of constipation is essential.Intestinal microbiota dysbiosis has been associated with constipation, and rebuilding or restoring gut microbiota homeostasis is a feasible strategy for the treatment of constipation.7,9–13 Prebiotics, non-digestible food ingredients that beneficially affect the host by selectively stimulating the growth and/or activity of beneficial microorganisms, have emerged as key modulators of this complex microbial community.14–17 Currently, more and more studies have shown that the prebiotics can alleviate disease symptoms by regulating the gut microbiota.18L-Arabinose (C5H10O5), a plant-specific pentaose, is a potential prebiotic with multiple effects on alleviating lipid metabolic disorder, improving insulin resistance, and anti-inflammation.19 Besides these effects, a previous study has shown that L-arabinose can inhibit DSS-induced colitis by regulating gut microbiota.20 Some studies have shown that L-arabinose can promote the production rate of SCFAs (acetic acid, propionic acid, butyric acid, lactic acid, malic acid and succinic acid) in the intestine21 and promote the proliferation of some beneficial bacteria (such as Bifidobacterium), which are more conducive to the maintenance of intestinal acidic environment and intestinal microecological balance. Based on these activities of L-arabinose on gut microbiota regulation, we hypothesize that L-arabinose can treat long-term constipation by rescuing the microbiota dysbiosis.
In this study, we used a mouse model of long-term constipation induced by loperamide to evaluate the effect of L-arabinose on intestinal motility, inflammation, intestinal pathological changes and gastrointestinal peptide secretion. The changes in the gut microbiota of the mice were detected by 16S rRNA amplifier sequencing, and the effect of L-arabinose on the proliferation of the differential bacteria Lactobacillus was further investigated. In addition, an animal intestinal bacterial deprivation experiment was designed to verify the key role of gut microbiota in L-arabinose in the treatment of constipation. In summary, the findings of this study can lay a foundation for the in-depth development of L-arabinose as a safe and effective functional food for alleviating long-term constipation.
2. Materials and methods
2.1. Main materials
L-Arabinose (purity >99.0%) was obtained from Tangchuan Biotechnology (Xiamen) Co., Ltd. Polyethylene glycol with electrolyte lavage solution (PEG-ELS) was purchased from Shutaishen Biopharmaceutical Co., Ltd. Loperamide hydrochloride was obtained from Aladdin. Lactobacillus plantarum TWK10, isolated from Taiwanese fermented cabbage,22 was obtained from SynbioTech Biotechnology (Yangzhou) Co., Ltd (Yangzhou, China) with the Lot number 25310201.2.2. Animal experiments
Male Balb/c mice (aged 6 to 8 weeks) were purchased from Zhejiang Weitong Lihua Experimental Animal Technology Co., Ltd. The mice were housed in a specific pathogen-free animal facility, under standard conditions at 23 ± 2 °C and a relative humidity of 55 ± 5% with a 12 h light/dark cycle, with free food and water. All animal experiments were performed according to the National Institutes of Health Guide for the Care and Use of Laboratory Animals and approved by the Center for New Drug Safety Evaluation and Research, China Pharmaceutical University (B20210219-1).According to the average daily intake of 30 g of sucrose by adults, adding L-arabinose at a ratio of 3.5% is equivalent to a daily intake of 17.5 mg per kg BW (based on a 60 kg body weight) of L-arabinose by the human body. The recommended amount is converted into the dosage for mice, which is 0.21 g kg−1 day−1. After acclimating for at least 3 days, the experimental animals were randomly divided into four groups (n = 10–12): control, model, L-arabinose (L-ara) and PEG-ELS. The mice were pretreated with purified water with or without L-arabinose (0.21 g kg−1 day−1) and PEG-ELS (5.25 g kg−1 day−1) for two weeks. Then, the mice in the model group, L-arabinose group, and PEG-ELS group received loperamide hydrochloride (LOP) at 10 mg kg−1 day−1 for two weeks, and the control group received an equivalent volume of drinking water. Meanwhile, all the mice continued to be fed with drinking water, L-arabinose or PEG-ELS. The feces of the mice were collected before the beginning of the experiment and at the endpoint of the experimental cycle for gut microbiota analysis.
For gut microbiota depletion experiments, male BALB/c mice (aged six to seven weeks) were randomly divided into four groups (n = 6): LOP, LOP + L-ara, LOP + Antibiotic mixture (Abx) and LOP + L-ara + Abx. The mice in Abx groups were treated with Abx (1.0 mg mL−1 ampicillin sodium, 1.0 mg mL−1 neomycin sulfate, 1.0 mg kg−1 metronidazole and 0.5 mg mL−1 vancomycin) with or without L-arabinose (0.21 g kg−1) orally for 2 weeks. In comparison, the mice without microbiota intervention were administered a gavage of drinking water or L-arabinose (0.21 g kg−1) for 2 weeks. Then, all the groups were treated with loperamide hydrochloride (10 mg kg−1) in addition to the former treatment for 2 weeks. At the endpoint, the mice were euthanized, and the blood, gastrointestinal tract and spleen were collected. All the samples were stored under appropriate conditions for further analysis.
2.3. Measurement of constipation indices
The mice were fasted overnight. At the end of the drug treatment, all the mice were given 0.2 mL of ink by intragastric administration. For intestinal propulsion evaluation, the gastrointestinal tracts of the mice were collected 30 min after the ink treatment. The number and weight of black feces were recorded within 4 h. The gastrointestinal (GI) propulsion rate was calculated as the percentage of the ink movement distance from the stomach to the length of the whole small intestine.2.4. Histopathology and glycogen staining
After dewaxing and rehydration, the morphological and quantitative changes of goblet cells in the colon tissue of the mice were measured using a glycogen staining kit (Nanjing Senbega Biotechnology Co., Ltd).Formalin-fixed proximal ileum or colon tissues were embedded in paraffin and cut into 4 μm sections. For H&E staining, the sections were dewaxed in xylene and rehydrated in a series of descending percentages of ethanol. After staining with hematoxylin and eosin (Beyotime), the slides were dehydrated and mounted. For glycogen staining, the sections were dewaxed in xylene, rehydrated in a series of descending percentages of ethanol and boiled in Tris-EDTA solution for antigen retrieval. After blocking, the slides were incubated with a primary antibody for Ki67 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000, Proteintech), CD3 (1
1000, Proteintech), CD3 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000, Proteintech), or CD8 (1
1000, Proteintech), or CD8 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200, Abcam) at 4 °C overnight. After washing, the sections were immersed with biotin-conjugated immunoglobulin G secondary antibody from the immunohistochemistry kit for 20 min, stained with DAB (MXB Biotechnologies) for 2 min according to the manufacturer's protocol and counter-stained with hematoxylin for another 2 min. The nuclei were counterstained with hematoxylin (Beyotime). The scores for the IHC and H&E staining were independently evaluated at 400× magnification using light microscopy by two pathologists who were blinded to the experiment data.
200, Abcam) at 4 °C overnight. After washing, the sections were immersed with biotin-conjugated immunoglobulin G secondary antibody from the immunohistochemistry kit for 20 min, stained with DAB (MXB Biotechnologies) for 2 min according to the manufacturer's protocol and counter-stained with hematoxylin for another 2 min. The nuclei were counterstained with hematoxylin (Beyotime). The scores for the IHC and H&E staining were independently evaluated at 400× magnification using light microscopy by two pathologists who were blinded to the experiment data.
2.5. Enzyme-linked immunosorbent assay (ELISA)
In order to determine the level of TNF-α and IL-6 in the spleen of the mice, half of the spleen tissue was added to a 300 μL 1x phosphate buffer solution (PBS) buffer and 3 μL protease inhibitor homogenate with a tissue grinder (Shanghai Jingxin). The homogenate was placed for 5 min at the end of grinding, and the mixture was centrifuged at 5000g for 10 min at 4 °C. The supernatant was collected to detect the cytokine concentration using TNF-α and IL-6 kits (MLBio), and the protein content of the spleen homogenate was determined using a BCA kit (Vazyme).In order to determine the levels of the gastrointestinal peptides and inflammatory factors in the serum of the mice, blood samples were collected from the orbital venous plexus during dissection. The collected blood was kept at room temperature for 2 h and centrifuged at 3000 rpm for 10 min at 4 °C. The serum was collected for detection by TNF-α, IL-6, 5-HT, SP, GAS and SS kits (MLBio).
2.6. Analyses based on flow cytometry
The spleen of the mice was grinded with 2 mL PBS, centrifuged at 400g for 5 min at 4 °C, and the precipitates were collected. After a 1x red blood cell lysate (Shanghai Beyotime Biotechnology Co., Ltd) treatment, the cells were blocked with mouse FcR Blocking Reagent (Miltenyi Biotec) at 4 °C for 15 min. The cells were then washed with PBS and resuspended for surface staining at 4 °C for 30 min. After a PBS rinse and centrifugation at 350g for 5 min at 4 °C, the cells were then re-suspended in cold PBS and analyzed using an FACSymphony A5 SORP instrument (BD Biosciences). Flow cytometry analysis was conducted with only viable cells using the FlowJo software. The following antibodies were used: Live/dead Fixable near-IR (Invitrogen), anti-CD45 (30-F11) (Biolegend), anti-CD11b (M1/70), anti-Ly6G (1A8-Ly6g) (eBioscience).2.7. 16S rRNA sequencing of intestinal bacteria
The feces of Day 0 and 28 were collected using a DNA/RNA free tube and immediately frozen at −80 °C. 16S rRNA sequencing and analysis were performed by Novogene (Tianjin, China). Briefly, genomic DNA was extracted using a DNA extraction kit (TIANGEN, China). The V3–V4 region of the 16S rRNA was amplified. A MetaVx Library Preparation kit was used to prepare a library. The DNA was then sequenced using the Illumina MiSeq method. The 16S rRNA data were analyzed using the QIIME2 data package (Version QIIME2-202202). Amplicon Sequence Variants (ASVs) were obtained using the DADA2 module in the QIIME2 software and mapped to the SILVA 138.1 database (SILVA 138.1) to obtain taxonomic information (https://www.arb-silva.de/). Alpha diversity metrics were calculated with the “vegan” R package (version 2.5–7). The identification of species with significant differences in each classification level was performed using MetaStat and t tests in R software v3.5.3. Linear Discriminant Analysis (LDA) Effect Size (LEfSe) was performed to reveal the community structure differentiation among three groups; the threshold LDA score of >4 was used. Functional prediction was achieved using Tax4Fun (V0.3.1) (https://tax4fun.gobics.de/).2.8. Lactobacillus bacterial proliferation test in vitro
In order to verify the effect of L-arabinose on the proliferation of Lactobacillus, Lactobacillus plantarum TWK10, obtained from SynbioTech Biotechnology (Yangzhou) Co., Ltd (Yangzhou, China) with the Lot number 25230101, was cultured in MRS broth (de Man, Rogosa, and Sharpe) with or without 0.2%–2% L-arabinose at 37 °C under anaerobic conditions for 60 h. The absorption at 600 nm and the pH of the bacteria culture solution were measured continuously at several intervals within 60 h. At the endpoint of the experiment, the bacteria DNA was extracted using a DNA extraction kit (TIANGEN, China), and quality-controlled in a NanoDrop®-300 spectrophotometer. A real-time PCR assay was carried out using a Lactobacillus spp. Probe qPCR Kit (ml107706, MLBio, China) according to the manufacturer's instructions by Thermo Fisher Scientific QuantStudioTM3 Real-time PCR Instrument (Applied Biosystems, USA). The concentration of the test samples was calculated based on the accompanying standard curve.2.9. Statistical analysis
All values are presented as the mean ± SEM. The statistical analyses were conducted using GraphPad Prism 8 (GraphPad Software, Inc.). Unless otherwise indicated, a two-tailed Student's t-test (for two groups) or one-way ANOVA with Turkey's post hoc test (for multiple groups) was used to establish statistical significance. P values of <0.05 were considered statistically significant.3. Results
3.1. L-Arabinose improved the defecation parameters and relieved intestinal injury in mice with constipation
To investigate the effect of L-arabinose on constipation, the mice were administered a gavage of loperamide for 2 weeks after L-arabinose or PEG-ELS treatment (Fig. 1A). The results showed that the constipation model was successful. Compared with the control group, loperamide significantly prolonged the first black feces time (P < 0.001), reduced the number and weight of black feces within 4 h after the administration of ink (P < 0.001), and decreased the intestinal transit rate (P < 0.05) (Fig. 1B–E). All these effects were observed with L-arabinose or PEG-ELS (drug used in clinic as positive control) treatment, indicating that L-arabinose had a therapeutic effect on long-term constipation (Fig. 1B–E).To evaluate the effect of L-arabinose on pathological injury in the intestine, pathological samples of the ileum from the mice were prepared and stained using hematoxylin–eosin (H&E) staining. As shown in Fig. 1F, the villi of the ileum were tightly arranged and had normal height; the apical epithelial cells were monolayer, orderly and regular; and the lamina propria had no infiltration of inflammatory cells in the control group. Conversely, in the loperamide-induced long-term constipation model group, the height of the ileum villi significantly decreased (P < 0.01). The villus spaces increased, and the villi appeared disrupted and irregularly arranged. Some apical epithelial cells were disorganized, multilayered and exhibited localized proliferation. Inflammatory cell infiltration was observed in the lamina propria. All these phenomena were alleviated when L-arabinose was applied (Fig. 1F).
3.2. L-Arabinose alleviated inflammation in mice with constipation
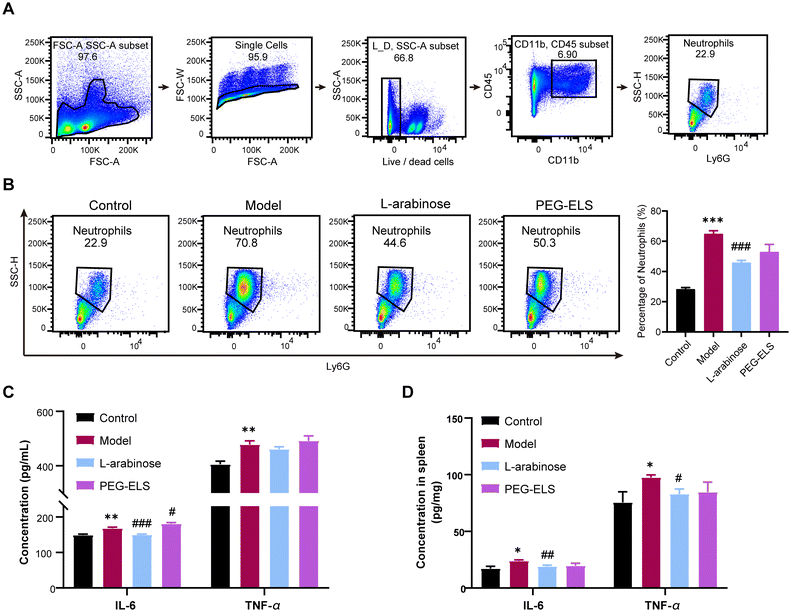
Our previous results showed that the L-arabinose group had fewer inflammatory cells infiltrating the lamina propria and less change of apical epithelial cells than the model group (Fig. 1F). Thus, we further explored the effect of L-arabinose on immune regulation. The spleen is a large immune organ of the human body and is full of lymphocytes, neutrophils, monocytes, etc. These cells are all important participants in the inflammatory response. Neutrophils (NEU) are the first-line cells in the immune system to detect the invasion of pathogenic microorganisms and attack them. The more the neutrophils, the more serious the infection. The flow cytometry results showed that the proportion of NEU in the spleen of the model group was significantly higher than that in the spleen of the control group (P < 0.001), while L-arabinose treatment mitigated the phenomena (P < 0.001) (Fig. 2B). Moreover, the concentration of IL-6 and TNF-α in the spleen was significantly higher in the model group than in the control group (P < 0.05), and these changes were significantly reversed by L-arabinose (P < 0.01, P < 0.05). PEG-ELS also down-regulated the levels of IL-6 and TNF-α, but there was no statistical difference (Fig. 2D). Similarly, the serum levels of IL-6 and TNF-α in the model group were significantly higher than those in the control group (P < 0.01). Compared with the model group, the levels of IL-6 in the L-arabinose group decreased significantly (P < 0.001), and the concentration of TNF-α exhibited a downward trend, but there was no statistical difference (Fig. 2C). In summary, these results show that L-arabinose can reduce inflammation in constipated mice.3.3. L-Arabinose regulated gastrointestinal peptide secretion and the function of goblet cells in the colon tissue of mice with constipation
Chronic constipation is associated with abnormalities of gastrointestinal peptides, which are involved in gastrointestinal motility, secretion, absorption and regulation of food intake. In order to explore the mechanism of L-arabinose in the treatment of constipation, we detected the level of gastrointestinal peptides in the serum of experimental mice. The results showed that the level of excitatory gastrointestinal peptide (5-hydroxytryptamine (5-HT), substance P (SP) and Gastrin (GAS)) in the model group was significantly lower than that in the control group (P < 0.01). L-Arabinose significantly increased the level of 5-HT, SP and GAS, compared to those in the model group (P < 0.01, P < 0.05, P < 0.001) (Fig. 3A–C). Considering the crucial role of 5-HT in gut motility, we further detected the mRNA expression of its synthesis restriction enzymes, tryptophan hydroxylase 1 (Tph1) and major transporter serotonin transporter (Sert), in the ileum. Although the L-arabinose treatment tended to rescue the down-regulation of Tph1 and Sert by loperamide, only the Tph1 expression level had a statistical difference between the model and L-arabinose groups (P < 0.05) (SI Fig. 1A and B). On the other hand, the level of Somatostatin (SS) in the L-arabinose group decreased significantly compared with that in the model group (P < 0.05) (Fig. 3D). Taken together, L-arabinose can treat constipation by enhancing gastrointestinal motility through regulating the gastrointestinal peptides.Since ileal H&E staining showed that L-arabinose could reduce the injury of the ileum in mice with constipation (Fig. 1F), we further examined the effect of L-arabinose on the functions of colonic goblet cells, which play an important role in the intestinal mucus barrier. Compared with the control group, the goblet cell morphology of the model group shrank, while L-arabinose rescued this pathological change (Fig. 3E). Next, we analyzed the number of goblet cells in the sigmoid colon by periodic acid-Schiff (PAS) staining. The results showed that compared with the control group, the number of goblet cells in the model group decreased (P < 0.001), and the number of goblet cells in the L-arabinose group increased significantly compared with that in the model group (P < 0.01) (Fig. 3F). These results suggested that L-arabinose prevents constipation by maintaining the function of goblet cells and mucus secretion in mice.
3.4. L-Arabinose regulated the gut microbiota in mice with constipation
Alteration of the gut microbiota composition is considered a major cause of impaired intestinal barrier function. Thus, we analyzed the effects of L-arabinose on the gut microbiome in the mice through the metagenomic shotgun sequencing of fecal samples (n ≥ 7). The feces were collected before the experiment (Day 0) and at the endpoint of the experiment (Day 28) for further analysis by 16S rRNA. At the baseline, there was no significant difference in the gut microbiota composition among different groups regarding α-diversity. However, the Simpson index of the L-arabinose and PEG-ELS groups all significantly decreased on Day 28 after treatment (SI Fig. 2A). The Venn diagram showed that the unique microbiota in the control, model, L-arabinose and PEG-ELS groups on Day 28 were 832, 218, 361, and 590, respectively, which indicated that the bacteria community was changed after different treatments (Fig. 4A). We further analyzed the composition of gut microbiota in the mice after the L-arabinose treatment. Compared with the control group, the level of Firmicutes, the most important butyric acid synthesizer, decreased significantly at the phylum level as well as the ratio of Firmicutes/Bacteroidota in the model group (Fig. 4B and SI Fig. 2B). At the genus level, Lactobacillus abundance was dramatically decreased in the constipated mice, compared to the control group. The level of Helicobacter that reduces SCFA production was increased under constipation (Fig. 4C). As expected, L-arabinose diminished these phenomena (Fig. 4B and C). In addition, the LEfSe results validated that the Lactobacillales, Lactobacillaceae and Lactobacillus levels were significantly higher in the L-arabinose group (Fig. 4D and E). MetaStat analysis results showed that compared with the control group, the level of Lactobacillus abundance in the model group decreased significantly (P < 0.05), while L-arabinose totally reversed the changes (P < 0.05) (Fig. 4F). These results suggested that Lactobacillus may be the most important genus regulated by L-arabinose to relieve constipation in mice. To further investigate whether Lactobacillus abundance was increased by L-arabinose, an in vitro proliferation study was applied. Considering the widely reported anti-constipation effects of Lactobacillus plantarum, we selected Lactobacillus plantarum TWK10 for the in vitro experiments.22–25 The results showed that 0.2%–2% L-arabinose could directly promote the proliferation of Lactobacillus (P < 0.001), and the pH value of culture media with L-arabinose decreased significantly (P < 0.001) (SI Fig. 2C–E). Lactic acid, the main metabolic product of Lactobacillus, can promote intestinal peristalsis and maintain an acidic environment to inhibit the reproduction of pathogenic and decaying bacteria. Besides, Lactobacillus can activate AhR on gut neurons to promote intestinal peristalsis by metabolizing tryptophan.26 Our data showed that the expression level of Ahr in the model group showed a downward trend, while L-arabinose increased Ahr expression in the model group (SI Fig. 2F). Taken together, L-arabinose could regulate the gut microbiota dysbiosis in constipated mice and especially increase Lactobacillus abundance.3.5. Antibiotics diminished the protective effects of L-arabinose on constipated mice
To verify whether L-arabinose exerted its effect by regulating the gut microbiota of mice, we used an antibiotic cocktail (Abx, 1.0 g L−1 ampicillin sodium, 1.0 g L−1 metronidazole, 0.5 g L−1 vancomycin, 1.0 g L−1 neomycin sulfate) on constipated mice to interfere with the microbiota. Consistent with our previous findings, L-arabinose significantly improves bowel movement according to the shorter first black feces time (P < 0.01), more and heavier black feces (P < 0.01, P < 0.05) and higher gut propulsion rate in the mice with long-term constipation (P < 0.01). Abx application dramatically diminished the protective effect of L-arabinose (Fig. 5B–E). Compared to the L-arabinose group, additional Abx intervention dramatically increased the first black feces time (P < 0.001) and decreased the number and weight of black feces (P < 0.001, P = 0.0546). Intriguingly, compared to the loperamide group, the first feces time was prolonged (P < 0.001) and the number of black feces was decreased (P < 0.05) in the gut microbiota-depletion mice with constipation. This indicated that microbiota still has a certain function of intestinal regulation (Fig. 5B and C). The above results confirmed that L-arabinose attenuated loperamide-induced constipation through the regulation of gut microbiota in mice.4. Discussion
Chronic constipation is a gastrointestinal disorder affecting millions of people of all ages worldwide, with a huge financial burden each year.27–29 A survey of patient perspectives recorded that 43% of people with constipation were not completely satisfied with the current pharmacological treatments for constipation.30 Hence, it is necessary to develop new and effective strategies for the treatment of chronic constipation. With the in-depth study of intestinal microecology and metabolic diseases, restoring intestinal microbiota homeostasis seems a promising strategy against constipation.13,31,32 Recently, several studies have proved that the regulation of intestinal microbiota could relieve constipation.33,34L-Arabinose, a kind of plant-specific aldopentose, is known to have beneficial effects on anti-hyperglycemia activity, improving insulin sensitivity and reducing triglycerides, fat accumulation and colitis, which are related to its regulation on gut microbiota.19,20,35,36 However, whether L-arabinose could relieve long-term constipation needs to be further elucidated. In this study, we systematically investigated the therapeutic effect of L-arabinose against loperamide-induced long-term constipation in mice. Besides enhancing intestinal peristalsis and improving defecation parameters, L-arabinose also alleviates gut histological injury by reducing constipation-induced inflammation and maintaining mucus secretion. Notably, its effect was comparable to or better than that of PEG-ELS. Mechanistically, L-arabinose ameliorates long-term constipation by restoring gut microbiota dysbiosis, which, in turn, regulates gastrointestinal peptide levels. Importantly, the gut microbiota depletion experiment confirmed that the therapeutic effect of L-arabinose on long-term constipation was mostly dependent on its regulation of gut microbiota.In most cases, constipation is considered to be closely related to intestinal motility disorders. Two main intestinal movements drive intestinal contents into the rectum: peristalsis and colon propulsion. These movements cause regular contraction of the colon wall and push the contents forward, facilitating normal bowel evacuation.37 Fecal and defecation characteristics are considered to be directly reflective of constipation, and peristalsis is detected by measuring the intestinal transit rate. Here, we used fecal wet weight, fecal number, time to first black stool defecation, and the intestinal transit rate to evaluate the effect of L-arabinose on constipation. The osmotic laxative, PEG-ELS, was used as a positive control. As expected, L-arabinose improved the defecation parameters in the constipated mice. It is gratifying that, apart from the fecal weight, the effect of L-arabinose is comparable to that of PEG-ELS. Osmotic laxatives work by absorbing water in the intestine and increasing stool volume. Therefore, it is understandable that the feces weight in the PEG-ELS group is heavier than that in the L-arabinose group. Taking into account the different doses administered to each group, our results indicate that arabinose could be a more effective treatment for constipation.
Gastrointestinal (GI) motility is generally considered to be multifactorial, involving inflammation and secretory dysfunction.38 A breakdown of the intestinal epithelial barrier due to loperamide administration leads to impaired intestinal function and subsequent constipation, which, in turn, increases the expression of pro-inflammatory cytokines.39 The overexpression of TNF-α, IL-1β, and IL-6 induces the disruption of connexins between cells, leading to the collapse of intestinal structural integrity.40 In clinics, elevated levels of IL-6 and TNF-α are often present in people with constipation.41 Therefore, we detected the pathology of gut and inflammatory cytokines in the constipated mice. The results showed that L-arabinose improved the intestinal histopathological changes caused by constipation, such as reducing inflammatory cell infiltration and maintaining the mucus secretion and gut integrity (Fig. 1F and Fig. 3E, F). Flow cytometry and ELISA showed that L-arabinose had a good inhibitory effect on inflammation in mice with constipation, which is in accordance with a previous study20 (Fig. 2).
Gastrointestinal hormones and neurotransmitters often influence intestinal fluid secretion and gastric motility.42 For instance, 5-hydroxytryptamine (5-HT) plays a key role in regulating gastrointestinal motility by activating a variety of 5-HT excitatory receptors43–45 or directly acting on smooth muscle and promoting tissue contraction. Substance P (SP) promotes gastrointestinal smooth muscle contraction and stimulates gastrointestinal peristalsis through the acetylcholine effect.46 Besides, SP can also down-regulate inflammatory factors to reduce intestinal wall injury and maintain the integrity of the intestinal barrier.47 Gastrin (GAS) is the only brain-gut peptide that can stimulate gastrointestinal motility not only at the level of the enteric nervous system and central nervous system, but also at the level of the peripheral nervous system. GAS has a direct stimulating effect on parietal cells to secrete gastric acid by producing histamine and promotes gastrointestinal motility and gastric emptying to relieve constipation.10 Somatostatin (SS), which is mainly distributed from the gastric antrum to the colon, can inhibit smooth muscle contraction and substance P (SP) release, reduce gastrointestinal motility, increase gastrointestinal transport time, and thereby aggravate constipation.48 The imbalance between the excitatory and inhibitory neurotransmitters performs a critical function in the pathogenesis of constipation. In this study, we observed the decreased SP, GAS and 5-HT levels, and the increased SS levels in the serum of constipated mice, indicating that the disruption of balance led to defecation dysfunction. L-Arabinose administration reversed the abnormal levels of those neurotransmitters, thereby relieving the constipation symptoms. Moreover, L-arabinose increases the Tph1 mRNA level, which in turn, enhances 5-HT synthesis. Currently, commonly used therapeutic drugs for constipation often cause adverse reactions. For example, serotonin 5-HT4 receptor agonists may cause nausea and vomiting, and linaclotide may lead to diarrhea.49,50L-Arabinose derived from food may be a safer and more effective treatment option. Unexpectedly, the mRNA expression of major transporter Sert doesn't change significantly. Further studies need to investigate the underlying mechanism at multiple endpoints.
Previous studies have shown that the gut microbiota is severely disturbed in both people with constipation and related animal models.10,11 The specific manifestations are as follows: compared with healthy individuals, the intestinal beneficial bacteria (such as Lactobacillus and Bifidobacterium) are decreased, while potential pathogens are increased in people with constipation.10,51–53 In alignment with these reports, our data showed that the gut microbiota in constipated mice was dramatically disturbed, while L-arabinose reversed the changes in the gut flora at different levels. In particular, L-arabinose can significantly increase the bacterial abundance of Lactobacillus (Fig. 4). It has been proven that Lactobacilli bacteria stimulate intestinal chromaffin cells to synthesize serotonin and accelerate intestinal peristalsis.54 Moreover, the proliferation assay confirmed that Lactobacillus plantarum TWK10 could promote its proliferation by L-arabinose in vitro (SI Fig. 2C–E). In order to verify whether L-arabinose works through the mechanism of intestinal bacteria, we designed the experiment of intestinal microbiota deprivation in mice (Fig. 5). By interfering with the intestinal bacteria of mice with antibiotics, it was confirmed that L-arabinose did treat constipation by regulating the intestinal microecology.
The metabolic process of intestinal microorganisms produces short-chain fatty acids (SCFAs), endotoxins and other products, which have different effects on the intestinal motility of the host.55 Among them, SCFAs are the main metabolites of dietary fiber fermented by intestinal microorganisms, which can stimulate enterochromaffin cells (ECs) to release 5-hydroxytryptophan (5-HT) and stimulate 5-HT receptors on the sensory fibers of the vagus nerve, resulting in muscle contraction, promoting intestinal movement and relieving constipation.48,56,57 A limitation of our study is the lack of SCFA assays to further confirm the gut microbiota-dependent role of L-arabinose in relieving long-term constipation. Additionally, future research should test more Lactobacillus strains to evaluate the effect of L-arabinose on their proliferation. Given that prebiotics may have multiple targets and mechanisms, unlike small-molecule drugs, the underlying mechanism explored in this study remains to be further elucidated.
5. Conclusions
This study confirmed the therapeutic effect of L-arabinose on long-term constipation induced by loperamide in mice, which could improve the physiological parameters of constipation, reduce ileal injury, inhibit inflammatory response and maintain mucus secretion in the gut. In addition, L-arabinose reshapes the structure of the intestinal microbiota. Through the intestinal bacteria depletion mouse model, we proved that the therapeutic effect of L-arabinose on constipation was dependent on the regulation of gut microbiota. The therapeutic effect of L-arabinose was analyzed systematically for the first time, and several possible mechanisms of L-arabinose were explored. This study provides a new candidate for the treatment of clinical constipation. Compared with osmotic laxatives, L-arabinose is a safer and longer-acting microecologic with the potential of multi-target and multi-mechanism therapy.Author contributions
Conception and design: Y. Wang and S. Zheng. Development of methodology: S. Zheng, H. Zheng, Y. Huang, B. Liang, Y. Gong, J. Shen, W. Liu, X. Xu, and Y. Ma. Acquisition of data: S. Zheng, H. Zheng, Y. Huang, B. Liang, Y. Gong, W. Liu, and X. Xu. Analysis and interpretation of data: S. Zheng, H. Zheng, Y. Huang, Y. Gong, X. Xu, and Z. Liu. Writing, review, and/or revision of the manuscript: Y. Wang and S. Zheng. Administrative, technical, or material support: Y. Wang. Study supervision: Y. Wang.Conflicts of interest
The authors declare that they have no competing interests.Data availability
All data supporting the findings of this study are available within the article. Supplementary information (SI) is available. The supplementary information contains: Supplementary Table. 1 Primer sequence. Supplementary Fig. 1 Relative mRNA expression levels of Tph1 and Sert in ileum of experimental mice. Supplementary Fig. 2 L-arabinose regulated gut microbiota and relevant genes expression of constipated mice. See DOI: https://doi.org/10.1039/d4fo03554b.The fecal sample data used in the study were deposited in the NCBI Sequence Read Archive (SRA) (https://www.ncbi.nlm.nih.gov/sra) under the project accession PRJNA1300306.
Acknowledgements
We thank Tangchuan Biotechnology (Xiamen) Co., Ltd. for providing L-arabinose and SynbioTech Biotechnology (Yangzhou) Co., Ltd for providing Lactobacillus plantarum TWK10. This work was supported by the National Natural Science Foundation of China (No. 82330119) and Fundamental Research Funds for the Central Universities (2632023TD08).References
- A. E. Bharucha and B. E. Lacy, Mechanisms, Evaluation, and Management of Chronic Constipation, Gastroenterology, 2020, 158, 1232–1249.e3 CrossRef CAS PubMed.
- M. Camilleri, A. C. Ford, G. M. Mawe, P. G. Dinning, S. S. Rao, W. D. Chey, M. Simrén, A. Lembo, T. M. Young-Fadok and L. Chang, Chronic constipation, Nat. Rev. Dis. Primers, 2017, 3, 17095 CrossRef PubMed.
- L. Wang, L. Hu, Q. Xu, T. Jiang, S. Fang, G. Wang, J. Zhao, H. Zhang and W. Chen, Bifidobacteria exert species-specific effects on constipation in BALB/c mice, Food Funct., 2017, 8, 3587–3600 RSC.
- J. Yin, Y. Liang, D. Wang, Z. Yan, H. Yin, D. Wu and Q. Su, Naringenin induces laxative effects by upregulating the expression levels of c-Kit and SCF, as well as those of aquaporin 3 in mice with loperamide-induced constipation, Int. J. Mol. Med., 2018, 41, 649–658 Search PubMed.
- I. Payne and L. M. Grimm Jr., Functional Disorders of Constipation: Paradoxical Puborectalis Contraction and Increased Perineal Descent, Clinics in colon and rectal surgery, 2017, vol. 30, pp. 22–29 Search PubMed.
- Y. Gan, J. Liang, W. Diao, X. Zhou, J. Mu, L. Pang, F. Tan and X. Zhao, Lactobacillus plantarum KSFY06 and geniposide counteract montmorillonite-induced constipation in Kunming mice, Food Sci. Nutr., 2020, 8, 5128–5137 Search PubMed.
- X. Zhang, H. Yang, J. Zheng, N. Jiang, G. Sun, X. Bao, A. Lin and H. Liu, Chitosan oligosaccharides attenuate loperamide-induced constipation through regulation of gut microbiota in mice, Carbohydr. Polym., 2021, 253, 117218 CrossRef CAS PubMed.
- A. D. Sperber, S. I. Bangdiwala, D. A. Drossman, U. C. Ghoshal, M. Simren, J. Tack, W. E. Whitehead, D. L. Dumitrascu, X. Fang, S. Fukudo, J. Kellow, E. Okeke, E. M. M. Quigley, M. Schmulson, P. Whorwell, T. Archampong, P. Adibi, V. Andresen, M. A. Benninga, B. Bonaz, S. Bor, L. B. Fernandez, S. C. Choi, E. S. Corazziari, C. Francisconi, A. Hani, L. Lazebnik, Y. Y. Lee, A. Mulak, M. M. Rahman, J. Santos, M. Setshedi, A. F. Syam, S. Vanner, R. K. Wong, A. Lopez-Colombo, V. Costa, R. Dickman, M. Kanazawa, A. H. Keshteli, R. Khatun, I. Maleki, P. Poitras, N. Pratap, O. Stefanyuk, S. Thomson, J. Zeevenhooven and O. S. Palsson, Worldwide Prevalence and Burden of Functional Gastrointestinal Disorders, Results of Rome Foundation Global Study, Gastroenterology, 2021, 160, 99–114 CrossRef PubMed.
- M. H. Vriesman, I. J. N. Koppen, M. Camilleri, C. Di Lorenzo and M. A. Benninga, Management of functional constipation in children and adults, Nat. Rev. Gastroenterol. Hepatol., 2020, 17, 21–39 Search PubMed.
- J. Huang, B. Lin, Y. Zhang, Z. Xie, Y. Zheng, Q. Wang and H. Xiao, Bamboo shavings derived O-acetylated xylan alleviates loperamide-induced constipation in mice, Carbohydr. Polym., 2022, 276, 118761 CrossRef CAS PubMed.
- Y. X. Liang, P. Wen, Y. Wang, D. M. OuYang, D. Wang, Y. Z. Chen, Y. Song, J. Deng, Y. M. Sun and H. Wang, The Constipation-Relieving Property of d-Tagatose by Modulating the Composition of Gut Microbiota, Int. J. Mol. Sci., 2019, 20(22), 5721 Search PubMed.
- R. Pan, L. Wang, X. Xu, Y. Chen, H. Wang, G. Wang, J. Zhao and W. Chen, Crosstalk between the Gut Microbiome and Colonic Motility in Chronic Constipation: Potential Mechanisms and Microbiota Modulation, Nutrients, 2022, 14(18), 3704 Search PubMed.
- L. Yang, Y. Wang, Y. Zhang, W. Li, S. Jiang, D. Qian and J. Duan, Gut microbiota: a new avenue to reveal pathological mechanisms of constipation, Appl. Microbiol. Biotechnol., 2022, 106, 6899–6913 Search PubMed.
- G. R. Gibson and M. B. Roberfroid, Dietary modulation of the human colonic microbiota: introducing the concept of prebiotics, J. Nutr., 1995, 125, 1401–1412 CrossRef CAS PubMed.
- E. M. Dewulf, P. D. Cani, S. P. Claus, S. Fuentes, P. G. Puylaert, A. M. Neyrinck, L. B. Bindels, W. M. de Vos, G. R. Gibson, J. P. Thissen and N. M. Delzenne, Insight into the prebiotic concept: lessons from an exploratory, double blind intervention study with inulin-type fructans in obese women, Gut, 2013, 62, 1112–1121 CrossRef CAS PubMed.
- L. B. Bindels, N. M. Delzenne, P. D. Cani and J. Walter, Towards a more comprehensive concept for prebiotics, Nat. Rev. Gastroenterol. Hepatol., 2015, 12, 303–310 CrossRef CAS PubMed.
- G. R. Gibson, R. Hutkins, M. E. Sanders, S. L. Prescott, R. A. Reimer, S. J. Salminen, K. Scott, C. Stanton, K. S. Swanson, P. D. Cani, K. Verbeke and G. Reid, Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics, Nat. Rev. Gastroenterol. Hepatol., 2017, 14, 491–502 CrossRef PubMed.
- S. Yoo, S. C. Jung, K. Kwak and J. S. Kim, The Role of Prebiotics in Modulating Gut Microbiota: Implications for Human Health, Int. J. Mol. Sci., 2024, 25(9), 4834 CrossRef CAS PubMed.
- L. Zhao, Y. Wang, G. Zhang, T. Zhang, J. Lou and J. Liu, L-Arabinose Elicits Gut-Derived Hydrogen Production and Ameliorates Metabolic Syndrome in C57BL/6J Mice on High-Fat-Diet, Nutrients, 2019, 11(12), 3054 Search PubMed.
- Y. Li, H. Pan, J. X. Liu, T. Li, S. Liu, W. Shi, C. Sun, M. Fan, L. Xue, Y. Wang, C. Nie, H. Zhang, H. Qian, H. Ying and L. Wang, L-Arabinose Inhibits Colitis by Modulating Gut Microbiota in Mice, J. Agric. Food Chem., 2019, 67, 13299–13306 Search PubMed.
- U. P. Tiwari, A. K. Singh and R. Jha, Fermentation characteristics of resistant starch, arabinoxylan, and β-glucan and their effects on the gut microbial ecology of pigs: A review, Anim. Nutr. (Zhongguo xu mu shou yi xue hui), 2019, 5, 217–226 Search PubMed.
- T. H. Liu, G. L. Chen, C. H. Lin, T. Y. Tsai and M. C. Cheng, Lactobacillus plantarum TWK10 relieves loperamide-induced constipation in rats fed a high-fat diet via modulating enteric neurotransmitters, short-chain fatty acids and gut microbiota, Food Funct., 2025, 16, 181–194 RSC.
- R. Yi, X. Zhou, T. Liu, R. Xue and Z. Yang, Amelioration effect of Lactobacillus plantarum KFY02 on low-fiber diet-induced constipation in mice by regulating gut microbiota, Front. Nutr., 2022, 9, 938869 CrossRef PubMed.
- C. Li, S. P. Nie, K. X. Zhu, T. Xiong, C. Li, J. Gong and M. Y. Xie, Effect of Lactobacillus plantarum NCU116 on loperamide-induced constipation in mice, Int. J. Food Sci. Nutr., 2015, 66, 533–538 CrossRef CAS PubMed.
- X. Zhao, R. Yi, Y. Qian and K. Y. Park, Lactobacillus plantarum YS-3 Prevents Activated Carbon-Induced Constipation in Mice, J. Med. Food, 2018, 21, 575–584 CrossRef CAS PubMed.
- Y. Obata, Á. Castaño, S. Boeing, A. C. Bon-Frauches, C. Fung, T. Fallesen, M. G. de Agüero, B. Yilmaz, R. Lopes, A. Huseynova, S. Horswell, M. R. Maradana, W. Boesmans, P. Vanden Berghe, A. J. Murray, B. Stockinger, A. J. Macpherson and V. Pachnis, Neuronal programming by microbiota regulates intestinal physiology, Nature, 2020, 578, 284–289 CrossRef CAS PubMed.
- A. E. Bharucha and A. Wald, Chronic Constipation, Mayo Clin. Proc., 2019, 94, 2340–2357 CrossRef PubMed.
- D. L. Tran and P. Sintusek, Functional constipation in children: What physicians should know, World J. Gastroenterol., 2023, 29, 1261–1288 CrossRef CAS PubMed.
- L. A. Harris and C. H. Chang, Burden of Constipation: Looking Beyond Bowel Movements, Am. J. Gastroenterol., 2022, 117, S2–S5 Search PubMed.
- G. Basilisco and G. Italian, Society of Neurogastroenterology Motility Study, Patient dissatisfaction with medical therapy for chronic constipation or irritable bowel syndrome with constipation: analysis of N-of-1 prospective trials in 81 patients, Aliment. Pharmacol. Ther., 2020, 51, 629–636 CrossRef PubMed.
- K. Hou, Z. X. Wu, X. Y. Chen, J. Q. Wang, D. Zhang, C. Xiao, D. Zhu, J. B. Koya, L. Wei, J. Li and Z. S. Chen, Microbiota in health and diseases, Signal Transduction Targeted Ther., 2022, 7, 135 CrossRef PubMed.
- Y. Fan and O. Pedersen, Gut microbiota in human metabolic health and disease, Nat. Rev. Microbiol., 2021, 19, 55–71 CrossRef CAS PubMed.
- B. Chandrasekharan, B. J. Saeedi, A. Alam, M. Houser, S. Srinivasan, M. Tansey, R. Jones, A. Nusrat and A. S. Neish, Interactions Between Commensal Bacteria and Enteric Neurons, via FPR1 Induction of ROS, Increase Gastrointestinal Motility in Mice, Gastroenterology, 2019, 157, 179–192 CrossRef CAS PubMed.
- S. Jayasimhan, N. Y. Yap, Y. Roest, R. Rajandram and K. F. Chin, Efficacy of microbial cell preparation in improving chronic constipation: a randomized, double-blind, placebo-controlled trial, Clin. Nutr., 2013, 32, 928–934 CrossRef PubMed.
- Y. Wang, Y. Guan, L. Xue, J. Liu, Z. Yang, C. Nie, Y. Yan, S. Liu, J. Sun, M. Fan, H. Qian, H. Ying, Y. Li and L. Wang, L-Arabinose suppresses gluconeogenesis through modulating AMP-activated protein kinase in metabolic disorder mice, Food Funct., 2021, 12, 1745–1756 RSC.
- K. Pol, M. L. Puhlmann and M. Mars, Efficacy of L-Arabinose in Lowering Glycemic and Insulinemic Responses: The Modifying Effect of Starch and Fat, Foods, 2022, 11(2), 157 CrossRef CAS PubMed.
- H. L. Wang, Understanding the Pathogenesis of Slow-Transit Constipation: One Step Forward, Dig. Dis. Sci., 2015, 60, 2216–2218 Search PubMed.
- E. A. Mayer, T. Savidge and R. J. Shulman, Brain-gut microbiome interactions and functional bowel disorders, Gastroenterology, 2014, 146, 1500–1512 CrossRef CAS PubMed.
- J. E. Kim, H. J. Song, Y. J. Choi, Y. J. Jin, Y. J. Roh, A. Seol, S. H. Park, J. M. Park, H. G. Kang and D. Y. Hwang, Improvement of the intestinal epithelial barrier during laxative effects of phlorotannin in loperamide-induced constipation of SD rats, Lab. Anim. Res., 2023, 39, 1 CrossRef CAS PubMed.
- X. Zhang, J. Zheng, N. Jiang, G. Sun, X. Bao, M. Kong, X. Cheng, A. Lin and H. Liu, Modulation of gut microbiota and intestinal metabolites by lactulose improves loperamide-induced constipation in mice, Eur. J. Pharm. Sci., 2021, 158, 105676 Search PubMed.
- M. Mokhtare, R. Alimoradzadeh, S. Agah, H. Mirmiranpour and N. Khodabandehloo, The Association between Modulating Inflammatory Cytokines and Constipation of Geriatrics in Iran, Middle East J. Dig. Dis., 2017, 9, 228–234 CrossRef PubMed.
- Q. Zhao, Y. Y. Chen, D. Q. Xu, S. J. Yue, R. J. Fu, J. Yang, L. M. Xing and Y. P. Tang, Action Mode of Gut Motility, Fluid and Electrolyte Transport in Chronic Constipation, Front. Pharmacol., 2021, 12, 630249 CrossRef CAS PubMed.
- Y. Reynaud, J. Fakhry, L. Fothergill, B. Callaghan, M. Ringuet, B. Hunne, D. M. Bravo and J. B. Furness, The chemical coding of 5-hydroxytryptamine containing enteroendocrine cells in the mouse gastrointestinal tract, Cell Tissue Res., 2016, 364, 489–497 CrossRef CAS PubMed.
- T. K. Smith, K. J. Park and G. W. Hennig, Colonic migrating motor complexes, high amplitude propagating contractions, neural reflexes and the importance of neuronal and mucosal serotonin, J. Neurogastroenterol. Motil., 2014, 20, 423–446 Search PubMed.
- Z. Zheng, J. Tang, Y. Hu and W. Zhang, Role of gut microbiota-derived signals in the regulation of gastrointestinal motility, Front. Med., 2022, 9, 961703 CrossRef PubMed.
- Z. Yang, S. Ye, Z. Xu, H. Su, X. Tian, B. Han, B. Shen, Q. Liao, Z. Xie and Y. Hong, Dietary synbiotic ameliorates constipation through the modulation of gut microbiota and its metabolic function, Food Res. Int., 2021, 147, 110569 CrossRef CAS PubMed.
- H. S. Hong, D. Y. Hwang, J. H. Park, S. Kim, E. J. Seo and Y. Son, Substance-P alleviates dextran sulfate sodium-induced intestinal damage by suppressing inflammation through enrichment of M2 macrophages and regulatory T cells, Cytokine, 2017, 90, 21–30 Search PubMed.
- J. Wang, Q. Liang, Q. Zhao, Q. Tang, A. F. Ahmed, Y. Zhang and W. Kang, The effect of microbial composition and proteomic on improvement of functional constipation by Chrysanthemum morifolium polysaccharide, Food Chem. Toxicol., 2021, 153, 112305 Search PubMed.
- J. Tack, M. van Outryve, G. Beyens, R. Kerstens and L. Vandeplassche, Prucalopride (Resolor) in the treatment of severe chronic constipation in patients dissatisfied with laxatives, Gut, 2009, 58, 357–365 CrossRef CAS PubMed.
- G. Bassotti, P. Usai-Satta and M. Bellini, Linaclotide for the treatment of chronic constipation, Expert Opin. Pharmacother., 2018, 19, 1261–1266 CrossRef CAS PubMed.
- T. Ohkusa, S. Koido, Y. Nishikawa and N. Sato, Gut Microbiota and Chronic Constipation: A Review and Update, Front. Med., 2019, 6, 19 CrossRef PubMed.
- L. Mancabelli, C. Milani, G. A. Lugli, F. Turroni, M. Mangifesta, A. Viappiani, A. Ticinesi, A. Nouvenne, T. Meschi, D. van Sinderen and M. Ventura, Unveiling the gut microbiota composition and functionality associated with constipation through metagenomic analyses, Sci. Rep., 2017, 7, 9879 Search PubMed.
- L. Zhu, W. Liu, R. Alkhouri, R. D. Baker, J. E. Bard, E. M. Quigley and S. S. Baker, Structural changes in the gut microbiome of constipated patients, Physiol. Genomics, 2014, 46, 679–686 CrossRef CAS PubMed.
- C. M. Theriot, A. A. Bowman and V. B. Young, Antibiotic-Induced Alterations of the Gut Microbiota Alter Secondary Bile Acid Production and Allow for Clostridium difficile Spore Germination and Outgrowth in the Large Intestine, mSphere, 2016, 1(1), e00045–15 Search PubMed.
- A. Segers, L. Desmet, T. Thijs, K. Verbeke, J. Tack and I. Depoortere, The circadian clock regulates the diurnal levels of microbial short-chain fatty acids and their rhythmic effects on colon contractility in mice, Acta Physiol., 2019, 225, e13193 CrossRef PubMed.
- S. Fukumoto, M. Tatewaki, T. Yamada, M. Fujimiya, C. Mantyh, M. Voss, S. Eubanks, M. Harris, T. N. Pappas and T. Takahashi, Short-chain fatty acids stimulate colonic transit via intraluminal 5-HT release in rats, Am. J. Physiol.: Regul., Integr. Comp. Physiol., 2003, 284, R1269–76 Search PubMed.
- D. J. Morrison and T. Preston, Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism, Gut Microbes, 2016, 7, 189–200 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |