Open Access Article
Open Access ArticleStructure-controlled sulfur poisoning and hydrogen-induced regeneration in single Pd nanoparticles probed by nanospectroscopy
Mazal Kostan-Carmiela,
Yehonatan Hovava,
Athanasios Theodoridis b,
Christoph Langhammer
b,
Christoph Langhammer b and
Elad Gross
b and
Elad Gross *a
*a
aInstitute of Chemistry, and the Center for Nanoscience and Nanotechnology, The Hebrew University, Jerusalem 91904, Israel. E-mail: elad.gross@mail.huji.ac.il
bDepartment of Physics, Chalmers University of Technology, Gothenburg 412 96, Sweden
First published on 3rd February 2026
Abstract
Structural order and surface crystallinity of palladium nanoparticles (Pd NPs) significantly affect their chemical and mechanical stability, and impact their resistance to poisoning and efficiency in hydrogen (H2) (de)sorption. This study investigates the impact of sulfuric acid (H2SO4) poisoning on pristine and annealed Pd NPs to identify the influence of structure on the density and stability of poisoners. Electron microscopy and diffraction analyses show that annealing transforms initially amorphous, rough, and aggregated particles into spherical and crystalline NPs with better-defined grain boundaries. Nanoscale AFM-IR analysis shows that pristine NPs rapidly decompose SOx upon H2 exposure, and SOx is mainly located on the rim of the NP. Annealed NPs maintain stable, localized SOx signatures across multiple N2–H2 cycles, and only after the second H2 exposure cycle the SOx is removed from the central part of the NP. Contact potential difference analysis shows the strong affinity of SOx to the rim of the NP. Together, these results reveal that crystallinity and defect density dictate both the spatial distribution and chemical stability of sulfur species on Pd surfaces. Pristine, defect-rich NPs promote rapid H2-induced SOx reduction and selective desorption from their centers, whereas annealed, crystalline NPs stabilize SOx more strongly and exhibit delayed desorption. This direct link between structural order, SOx binding strength, and hydrogen (de)sorption affinity underscores the critical role of nanoscale morphology in controlling poisoning dynamics and highlights nanospectroscopy as a powerful tool for correlating local structure with chemical functionality.
Introduction
Catalytic behavior of metallic nanoparticles (NPs) is influenced by their atomic arrangement, degree of crystallinity, and distribution of structural defects.1–3 Grain size, surface roughness, facet orientation, and defect density all contribute to shaping the electronic structure and adsorption energetics of active sites, thereby governing reaction pathways and catalytic efficiency.4–9 Structural imperfections provide highly reactive adsorption sites, including disordered atomic domains, vacancies, steps, and grain boundaries. However, these imperfections can also contribute to mechanical deformation, chemical restructuring, and long-term degradation of the material.10 Therefore, rational design of catalytic materials requires a clear understanding of the interplay between structural order and chemical functionality.11–13There has been extensive research demonstrating the influence of structure on catalytic performance across a variety of metallic systems. Amorphous or defect-rich materials often exhibit enhanced initial reactivity due to the presence of a high density of reactive surface sites, but may suffer from limited stability or irreversible restructuring.14,15 In contrast, crystalline NPs with well-defined facets provide more predictable adsorption behavior and improved stability.16–19 Small or irregular NPs often exhibit rapid adsorption kinetics, but experience high lattice strain after repeated reaction exposure, while larger or more faceted particles possess greater mechanical stability.20,21 It can be therefore deduced that crystallinity, morphology, and defect density have a crucial impact on the catalytic performance and stability of materials.
Catalyst stability is strongly affected by the susceptibility to surface poisoning. Molecules such as CO, NOx, H2O, and sulfur-oxide species (SOx) can deactivate surface sites, due to strong interaction with the metal surface, preventing reactant adsorption and its activation.10,22–24 The poisoning mechanism is highly sensitive to the surface structure. Surfaces with a high density of adsorption sites can facilitate strong, sometimes irreversible binding of poisoning species.25,26 Consequently, the role and reversibility of catalyst poisoning depend on both the chemical nature of the poisoner and the structural characteristics of the active surface.
Pd NPs are ideal materials for studying structure–function relationships due to their exceptional ability to dissociate and absorb hydrogen. Hydrogen uptake in Pd induces a reversible phase transition accompanied by lattice expansion, making Pd a powerful system for hydrogen storage, catalysis, and sensing.9,22,27–30 However, the reversibility and kinetics of phase transition depend strongly on structural and morphological characteristics of Pd NPs, including size, crystallinity, and surface defect density. Amorphous domains, rough surfaces, and high density of surface defects are common features in pristine Pd NPs, which enhance their hydrogen sorption rates, but also induce irreversible lattice strain upon repeated cycling.30,31 Alternatively, thermal annealing can be used to transform amorphous Pd NPs into polycrystalline nanostructures that are characterized with smooth surfaces and lower defect density. This structural change does not only enhance the particle uniformity but also leads to more predictable and reversible phase transitions during hydrogen (de)sorption cycling.30,32,33 Therefore, structural order is directly related to mechanical robustness, phase-transition reversibility, and hydrogen reactivity in Pd-based nanomaterials.34–37
Pd surfaces are highly vulnerable to poisoning, and specifically to sulfur poisoning.9,38,39 SOx species bind strongly to Pd, blocking H2 dissociation sites and significantly reducing their catalytic activity.9 Defect-rich, pristine Pd NPs provide a significant number of high-energy adsorption sites, which result in rapid SOx accumulation and accelerated deactivation. Annealed Pd NPs, on the other hand, exhibit a lower defect density and well-defined crystallographic facets, resulting in a higher resistance to sulfur adsorption as well as a better recovery following H2 exposure.40
Due to their structural complexity, which induces both inter- and intra-particle variances, a comprehensive understanding of structure–reactivity correlations in Pd NPs requires an in-depth nanoscale analysis.41–43 Such analysis will enable spatial mapping of poisoners’ distribution and reveal how variations in surface coordination influence the adsorption strength, stability and diffusivity of sulfur-based species. Recent advances in high spatial resolution spectroscopy have enabled nanoscale investigations of catalytic nanoparticles.7,8,44–54 In particular, infrared (IR) nanospectroscopy measurements55,56 have shown that the types and adsorption geometries of SOx species vary significantly due to morphological and structural heterogeneity among individual nanoparticles.9,39 However, the connection between crystallinity, location-dependent SOx adsorption (rim vs. center), and the kinetics of H2-induced SOx reduction remains insufficiently resolved.
Herein, we combine AFM-IR, KPFM, and electron microscopy to correlate the structural evolution of pristine versus annealed Pd NPs with their sulfur-poisoning behavior. We show that annealing transforms amorphous, highly corrugated particles into crystalline nanostructures with smoother surfaces, which in turn stabilizes SOx species and delays their H2-induced desorption. In contrast, pristine NPs exhibit rapid SOx reduction and selective desorption from their central regions, with preferential retention at defect-rich rim sites. These insights establish a direct structure–poisoning relationship and highlight how crystallinity governs both SOx stability and hydrogen (de)sorption dynamics.
Experimental
Nanofabrication of Pd NPs samples
Pd nanodisk arrays were fabricated on 1 cm × 1 cm fused silica substrates using Hole-mask Colloidal Lithography (HCL).57 The particles had a nominal size of 210 nm (diameter) and 25 nm (height), covering approx. 10% of the total sample area. For the annealed NPs, subsequent annealing was performed at 500 °C for 18 h under a flow of 2 vol% H2 in Ar.AFM-IR and KPFM measurements
Atomic force microscopy infrared (AFM-IR) and Kelvin probe force microscopy (KPFM) measurements were performed in tapping mode using a nanoIR-3 setup (Anasys, Bruker). AFM-IR setup is equipped with a Bruker Hyperspectral Quantum Cascade Lasers (QCL) laser source (790–1950 cm−1), a gold-coated Si probe with a nominal diameter of ∼25 nm, resonance frequencies of 75 ± 15 kHz, and spring constants of 1–7 N m−1. Spectra were acquired with an average integration time of 5 s per spectrum and a spectral resolution of 2 cm−1. For each measurement point, IR spectra were obtained by averaging five consecutive spectra collected at the same location on the nanoparticle. KPFM measurements were performed using a Pt/Ir probe with a nominal diameter of ∼25 nm, resonance frequency of 62 ± 14 kHz, and spring constants of 1–6 N m−1. Nano-IR measurements were conducted in situ at room temperature under exposure to varying gas environments (1 atm of N2 or 1 atm of 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 N2
1 N2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2 for 60 minutes), with humidity values less than 10%.
H2 for 60 minutes), with humidity values less than 10%.
H2SO4 poisoning
Sulfur poisoning of Pd NPs was performed by immersing the sample in a 10 mM aqueous H2SO4 solution for 10 min at 25 °C. Following exposure, the sample was dried on a hot plate in air at 100–110 °C for 10 min to remove physisorbed surface residues. The poisoning conditions were selected based on previous studies demonstrating that these conditions preserve the structure of both the Pd nanoparticles and the substrate, while harsher conditions induce structural changes.39FIB-TEM measurements
A lamella of Pd NPs for Scanning Transmission Electron Microscopy (STEM) imaging was prepared using a Focused-Ion Beam (FIB) (Helios NanoLab 460F1). STEM images were taken using an aberration probe-corrected Themis Z-G3 (Thermo Fisher Scientific) operated at 300 kV, equipped with an Annular Dark Field Detector (HAADF) for the diffraction pattern of a single Pd NP.Results and discussion
Pd NPs were prepared on a Si (100) wafer with a native oxide layer by using Hole-mask Colloidal Lithography nanofabrication. In order to prepare NPs that are characterized with a better-defined structure, the NPs were annealed to 500 °C for 18 h under a flow of 2% v/v H2 in Ar. The chosen annealing temperature is above the Hüttig temperature (0.3 Tm, Tm = melting temperature of Pd = 1828 K) of Pd (275 °C) and below the Tammann temperature (0.5 Tm, ∼650 °C) and thus leads to surface atom diffusion and smoothing without sintering of nanoparticles due to ripening and migration. High-resolution TEM (HR-TEM) imaging shows the morphology and structure of the pristine and annealed Pd NPs (Fig. 1a, b, e and f, respectively). | ||
| Fig. 1 TEM (a and e), HR-TEM (b and f), FIB-TEM cross-sectional images (c and g) and FFT diffraction patterns (d and h) of pristine and annealed Pd NPs, respectively. | ||
Characterization was conducted by using FIB to extract a lamella and its analysis by TEM imaging and fast Fourier transform (FFT) of the diffraction pattern of the pristine and annealed NPs (Fig. 1c, d, g and h, respectively).
Both samples included NPs with a diameter and height of 190 ± 10 nm and 25 ± 5 nm, respectively. It was also identified that the pristine NPs are characterized with a more corrugated surface that is constructed of nanoscale aggregates (Fig. 1a and b). The annealed NPs showed a more spherical morphology with smoother surfaces, better-defined particle boundaries, and enhanced crystallinity (Fig. 1e and f), which are consistent with thermally driven intra-particle sintering and grain growth.
TEM cross-sections of the pristine NP revealed a largely amorphous structure, as indicated by the diffuse, disordered FFT patterns (Fig. 1c and d). Conversely, annealed NPs displayed a different morphology, while the TEM cross-sections revealed sharply defined crystalline domains, and the corresponding FFT images exhibited a distinct diffraction pattern that is consistent with a well-ordered face-centered cubic (FCC) structure (Fig. 1g and h). These results confirm that thermal annealing promotes the transformation of disordered Pd NPs into nanostructures that are more robust, with defined crystalline domains and lower density of grain boundaries.
The pristine and annealed NPs were immersed in H2SO4 (10 mM, 25 °C, 10 minutes), followed by heating the dried sample in air (110 °C, 10 minutes) to remove physisorbed residues. Localized IR spectra were acquired from the center of the pristine and annealed NPs (Fig. 2a and b, respectively). The pristine NP exhibited characteristic IR absorption bands (Fig. 2a, blue colored spectrum) at 1100, 1194 and 1228 cm−1. The feature at 1100 cm−1 was assigned to sulfates adsorbed with 3-fold geometry on metallic sites (Pd0) of Pd(111) facets, while the peak at 1194 cm−1 is attributed to sulfates adsorbed with 3-fold geometry with the C3v symmetry on electron-deficient Pdx+ sites.58 The peak at 1228 cm−1 is attributed to SO2 species.59 The peak at 1451 cm−1 was assigned to 2-fold or 3-fold sulfates adsorbed on oxidized Pd sites (i.e., Pdx+). The annealed NP (Fig. 2b, blue colored spectrum) showed signals at 1048 and 1095 cm−1, correlated to symmetric and asymmetric S–O stretching of sulfate, respectively, while the minor signal at 1264 cm−1 was correlated to SO2. The detection of a broad absorbance band at 1000–1150 cm−1 with a split to two peaks is correlated to sulfate adsorption in a bidentate adsorption mode with C2V symmetry.58
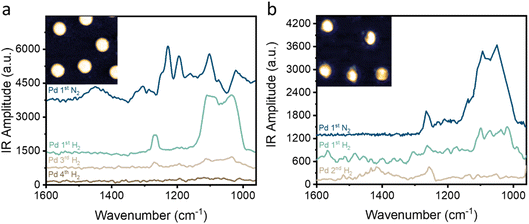 | ||
| Fig. 2 IR nanospectroscopy measurements acquired on the center of pristine (a) and annealed (b) NPs after their exposure to H2SO4. IR spectra were acquired following exposure to cycles of N2 and H2. | ||
After exposure to H2 the IR signal of the pristine sample (Fig. 2a, green-colored spectrum) was similar to that of the annealed sample, with two dominant peaks at 1035 and 1108 cm−1 and a smaller peak at 1267 cm−1. The disappearance of the peaks at 1228 cm−1, coupled with the presence of a new peak at 1267 cm−1, was assigned to structural changes that modified the adsorption geometry.60 Consecutive H2–N2 cycles led to an overall decrease in the SOx signal and after 3 cycles only a minor signature was detected on the pristine NP. No signal was detected on the central part of the NPs after the 4th cycle, indicative of SOx desorption from the center of the NP (Fig. 2a, brown-colored spectrum).
Exposure to H2 reduced the vibrational signals on the annealed NP to about 1/3 of their initial amplitude. After exposure to a second cycle of H2, the IR signals in the 1000–1100 cm−1 range were not detected. The peak at 1264 cm−1 was attributed to SO2 species.59 The peak at 1420 cm−1 was assigned to 2-fold or 3-fold sulfates adsorbed on oxidized Pd sites (i.e., Pdx+). It can be concluded that exposure to H2SO4 led to the formation of different surface chemistry on the pristine and annealed NPs.
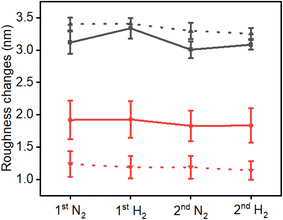
Quantitative analysis of surface roughness was performed based on AFM measurements (Fig. 3). The pristine NPs exhibited an average surface roughness of 3.12 ± 0.17 nm, which increased to 3.40 ± 0.10 nm after exposure to H2SO4 (Fig. 3, black colored curves). Following two cycles of exposure to N2 and H2 the roughness value decreased to 3.25 ± 0.08 nm. The annealed Pd NPs displayed a smoother surface, with an overall roughness of 1.92 ± 0.29 nm before exposure to H2SO4, which decreased to 1.24 ± 0.20 nm after poisoning. Following two cycles of exposure to N2 and H2 the roughness values of both the pristine and annealed samples showed a moderate decrease. It was previously demonstrated by us that due to the high structural similarity of the nanoparticles, analysis of five nanoparticles provides statistically meaningful values that are representative of the behavior of the nanoparticle array.9
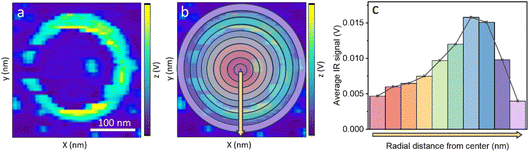
Nanoscale IR mapping was performed to correlate the structural properties with the distribution and stability of poisoners. Nanoscale AFM-IR mapping was performed at 1108 cm−1, correlated to the asymmetric S–O vibration of SO42− (Fig. 4a). Quantitative analysis of the variance in the IR amplitude on single NPs was performed by dividing the NP surface into concentric rings and averaging the IR signal across the ring (Fig. 4b). The averaged IR signal of each ring was then plotted as function of the radial distance of the ring from the center of the NP to provide a quantitative analysis of the IR signal amplitude on different sites of the NP (Fig. 4c).
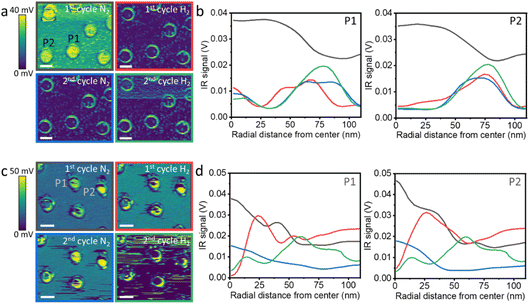
AFM-IR maps were recorded for the pristine and annealed NPs (Fig. 5a and c, respectively) after exposure to H2SO4 and following consecutive N2 and H2 cycles. Quantitative analysis of the IR signal was performed for two pristine and annealed NPs (Fig. 5b and d, respectively), in a similar approach to the one described in Fig. 4. The pristine NPs showed an AFM-IR signal that covered the NPs, indicating that the NPs are coated with SOx molecules (Fig. 5a). Analysis of the AFM-IR maps showed similar signals for the central parts of the NPs with a continuous decrease in the IR signal toward the edges of the NPs (Fig. 5b, black-colored curve). Following exposure to H2, the IR signal in the central part of the pristine NPs decreased and a ring of IR signal was detected on the outer rim of the NP (Fig. 5a). This was also validated by analysis of the IR signals (red-colored curve, Fig. 5b). A second exposure cycle to N2 (blue-colored curve, Fig. 5b) and to H2 (green-colored curve, Fig. 5b) led to sharpening of the IR signal toward the rim of the NP. These results show the selective desorption of SOx from the central part of the NP following exposure to H2.
A different pattern was detected for the annealed NP. AFM-IR imaging probed the selective desorption of SOx species only after the second H2 cycle (Fig. 5c). Quantitative analysis of the IR signal showed that prior to exposure to H2, the IR signal reached its maximum at the center of the NP and moderately decreased toward the edges of the NP (black colored curve, Fig. 5d). Following H2 exposure, the maximum IR signal shifted from the centre toward the rim of the NP (red colored curve, Fig. 5d) and this shift further increased after the second H2 exposure (green colored curve, Fig. 5d).
These observations are consistent with the analysis of surface roughness (Fig. 3). Pristine NPs exhibit higher surface corrugation values, and noticeable changes in surface roughness were measured following exposure to H2, indicating a dynamic and adaptable surface structure. Due to enhanced atomic mobility, SOx diffusion and desorption occurred during H2 exposure, presumably due to dissociation of H2 that enabled the following reduction and desorption of SOx species. The annealed NPs exhibit smaller and more consistent roughness variations, indicating that the surface is structurally more stable. In addition, it is expected that H2 dissociation will have higher kinetic barriers on a surface with a lower density of surface defects. Thus, the rate of SOx reduction, followed by desorption, is expected to be slower on the annealed NPs. For both the pristine and annealed NPs there is higher stability of SOx on the edge of the NP, correlated with the higher density of surface defects on these sites.
KPFM measurements were performed to probe the changes in contact potential difference (CPD), which reflects the difference in work function between the tip and sample (Fig. 6). As the transformation from metal to metal–hydride is expected to influence the work function value, KPFM measurements can shed light on the hydrogenation of the particle’s top layers and the impact of surface poisoning on hydrogen (de)sorption kinetics.
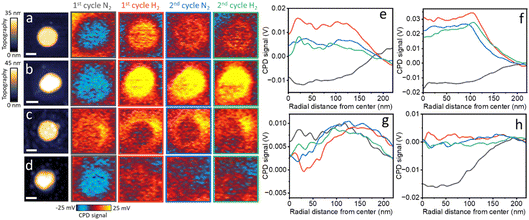
AFM topography and CPD mapping were performed for pristine and annealed NPs before (Fig. 6a and b, respectively) and after (Fig. 6c and d, respectively) exposure to H2SO4 following alternating cycles of N2 and H2. CPD measurements show noticeable differences in the CPD values of the pristine NP following the initial exposure to H2 (Fig. 6a). Quantitative analysis of the CPD signal shows that the CPD values at the center of the NP increased by ∼25 mV after the initial exposure to H2 (red colored curve, Fig. 6e). In the second N2 and H2 exposure cycle relatively minor changes were probed in CPD values. Interestingly, the CPD values at the rim of the NP showed a higher signal than at the center of the NP, demonstrating the high reactivity and affinity of hydrogen to these sites.
The annealed NP displayed noticeable changes in CPD values of up to 40 mV following H2 exposure (Fig. 6b and red colored curve in Fig. 6f), indicative of enhanced hydrogen sorption affinity, correlated to improved crystallinity that led to higher diffusivity of hydrogen in the NP. Local enhancement in CPD values was measured at the rim of the NP, correlated to the higher density of surface defects at these sites. For both the pristine and annealed NPs, smaller changes were probed in the second N2–H2 exposure cycle, in comparison to the first cycle, attributed to the slower kinetics in hydrogen desorption from the top layers of the NPs.
Following H2SO4 poisoning, the CPD values of the pristine NP showed noticeable differences of up to 10 mV between the rim and the center of the NP (Fig. 6c and red colored curve in Fig. 6g). The non-homogeneous CPD signal along the NP’s rim reflects the structural variance and heterogeneity in SOx adsorption pattern that changed the hydrogen sorption affinity. Nevertheless, although the adsorption of SOx species is located at rim sites (Fig. 4a), the magnitude of response to H2 exposure is much smaller compared to the non-poisoned pristine NPs (Fig. 6a). These results show the dominant effect of sulfate adsorption at rim sites on the overall hydrogen sorption affinity. In contrast, the annealed sample that was poisoned by SOx showed a more homogeneous CPD distribution after the first H2 exposure (Fig. 6d and h).
These trends are consistent with the AFM-IR mapping results (Fig. 5a). After the first H2 exposure, the pristine NPs displayed IR signature on the rim of the NP and this IR signature was identified as well in the CPD analysis. The annealed Pd NPs maintained stable IR signatures across the surface until the second H2 exposure (Fig. 5b), which promoted SOx sorption. Therefore, the CPD values do not show the rim signal that was detected for the pristine NP and show a moderate response to the change in the N2 to H2 environment due to surface poisoning of the NP.
Conclusions
This work demonstrates how the structure of Pd NPs governs their chemical reactivity, hydrogen (de)sorption affinity, and susceptibility to sulfur poisoning. Pristine Pd NPs, characterized by rough, defect-rich surfaces and largely amorphous interiors, exhibit rapid adsorption and reduction of SOx species during H2 exposure. In contrast, thermal annealing produces crystalline Pd nanostructures with reduced defect densities, leading to more spatially and chemically uniform SOx adsorption.Nanoscale AFM-IR mapping reveals that SOx species distribute and evolve differently on pristine and annealed NPs. Pristine particles show rapid depletion of SOx from their centers and selective retention at their more defective rims. Annealed NPs exhibit more stable SOx signatures, with desorption occurring only after two cycles of hydrogen exposure. These trends correlate with AFM-derived surface roughness measurements and KPFM results, both of which confirm the more pronounced effect of poisoning on the annealed NPs, correlated to the lower density of surface defects, which limits the dissociative adsorption of H2, followed by reduction and desorption of the adsorbed SOx species.
These results establish a direct link between nanoscale crystallinity, defect density, and sulfur poisoning dynamics in Pd NPs. Annealed NPs are characterized by structural robustness, which induces stronger sulfur adsorption sites, and quenches the hydrogen sorption affinity. In contrast, pristine NPs are characterized by a high density of surface defects, leading to weaker SOx adsorption sites and improved hydrogen (de)sorption affinity, in comparison to the annealed NPs. These nanospectroscopic insights underscore the importance of structural control in designing durable Pd-based catalysts and hydrogen-responsive sensors, and highlights nanoscale spectroscopy as a powerful tool for correlating local structure with chemical functionality.
Conflicts of interest
There are no conflicts to declare.Data availability
The datasets supporting this article, titled “Structure-controlled sulfur poisoning and hydrogen-induced regeneration in single Pd nanoparticles probed by nanospectroscopy” are openly available on Zenodo and can be accessed via the DOI: https://doi.org/10.5281/zenodo.17934083.Acknowledgements
This research was supported by the Swedish Foundation for Strategic Research (SSF) and Israel’s Ministry of Innovation, Science, and Technology (MOST), grant no. 4250, and received funding from the Swedish Foundation for Strategic Research project SIP21-0032 and the Competence Centre TechForH2 (AT, CL). The Competence Centre TechForH2 is hosted by Chalmers University of Technology and is financially supported by the Swedish Energy Agency (P2021-90268) and the member companies Volvo, Scania, Siemens Energy, GKN Aerospace, PowerCell, Oxeon, RISE, Stena Rederier AB, Johnson Matthey, and Insplorion. Part of this work was carried out at the Chalmers MC2 cleanroom facility (µFab).References
- B. Hammer and J. K. Nørskov, Theoretical Surface Science and Catalysis-Calculations and Concepts, Adv. Catal., 2000, 45, 71–129, DOI:10.1016/S0360-0564(02)45013-4.
- H. J. Freund and G. Pacchioni, Oxide Ultra-Thin Films on Metals: New Materials for the Design of Supported Metal Catalysts, Chem. Soc. Rev., 2008, 37(10), 2224–2242, 10.1039/b718768h.
- G. A. Somorjai and Y. Li, Introduction to Surface Chemistry and Catalysis, 2nd edn, Wiley, 2010 Search PubMed.
- N. Nilius and H. J. Freund, Activating Nonreducible Oxides via Doping, Acc. Chem. Res., 2015, 48(5), 1532–1539, DOI:10.1021/acs.accounts.5b00018.
- L. Vattuone, L. Savio and M. Rocca, Influence of Defects on Adsorption Model Studies with Stepped Surfaces, Encyclopedia of Interfacial Chemistry, 2018, pp. 38–165, DOI:10.1016/B978-0-12-409547-2.11461-1.
- M. Carmiel-Kostan, S. Nijem, S. Dery, G. Horesh and E. Gross, Composition-Reactivity Correlations in Platinum-Cobalt Nanoporous Network as Catalyst for Hydrodeoxygenation of 5-Hydroxymethylfurfural, J. Phys. Chem. C, 2019, 123(50), 30274–30282, DOI:10.1021/acs.jpcc.9b07453.
- L. Rikanati, H. Shema, T. Ben-Tzvi and E. Gross, Nanoimaging of Facet-Dependent Adsorption, Diffusion, and Reactivity of Surface Ligands on Au Nanocrystals, Nano Lett., 2023, 23(12), 5437–5444, DOI:10.1021/acs.nanolett.3c00250.
- B. Friedman, L. Giloni, O. M. Gazit and E. Gross, Nanoscale Chemical Imaging of Basic Sites Distribution on Catalytically Active Mg–Al Mixed Oxide Particles, Chem. Biomed. Imaging, 2025, 3(8), 560–568, DOI:10.1021/cbmi.5c00017.
- M. Kostan-Carmiel, A. Theodoridis, H. R. Eisenberg, T. Stein, C. Langhammer and E. Gross, Nanoscale Analysis of Sulfur Poisoning Effects on Hydrogen Sorption in Single Pd Nanoparticles, ACS Nano, 2025, 19(42), 36969–36981, DOI:10.1021/acsnano.5c08917.
- C. H. Bartholomew, Mechanisms of Catalyst Deactivation, Appl. Catal., A, 2001, 212(1–2), 17–60, DOI:10.1016/S0926-860X(00)00843-7.
- B. Coq and F. Figueras, Structure–Activity Relationships in Catalysis by Metals: Some Aspects of Particle Size, Bimetallic and Supports Effects, Coord. Chem. Rev., 1998, 178–180, 1753–1783, DOI:10.1016/S0010-8545(98)00058-7.
- C. Vogt and B. M. Weckhuysen, The Concept of Active Site in Heterogeneous Catalysis, Nat. Rev. Chem., 2022, 6, 89–111, DOI:10.1038/s41570-021-00340-y.
- Y. Wen, F. Wang, J. Zhu, Q. Wen, X. Xia, J. Wen, C. Deng, J. H. Du, X. Ke, Z. Zhang, H. Guan, L. Nie, M. Wang, W. Hou, W. Li, W. Tang, W. Ding, J. Chen and L. Peng, Revealing the Structure-Activity Relationship of Pt1/CeO2 with 17O Solid-State NMR Spectroscopy and DFT Calculations, Nat. Commun., 2025, 16(1), 3537, DOI:10.1038/s41467-025-58709-2.
- E. P. George, D. Raabe and R. O. Ritchie, High-Entropy Alloys, Nat. Rev. Mater., 2019, 4, 515–534, DOI:10.1038/s41578-019-0121-4.
- M. Ibáñez, S. C. Boehme, R. Buonsanti, J. De Roo, D. J. Milliron, S. Ithurria, A. L. Rogach, A. Cabot, M. Yarema, B. M. Cossairt, P. Reiss, D. V. Talapin, L. Protesescu, Z. Hens, I. Infante, M. I. Bodnarchuk, X. Ye, Y. Wang, H. Zhang, E. Lhuillier, V. I. Klimov, H. Utzat, G. Rainò, C. R. Kagan, M. Cargnello, J. S. Son and M. V. Kovalenko, Prospects of Nanosciencewith Nanocrystals: 2025 Edition, ACS Nano, 2025, 19(36), 31969–32051, DOI:10.1021/acsnano.5c07838.
- R. Narayanan and M. A. El-Sayed, Shape-Dependent Catalytic Activity of Platinum Nanoparticles in Colloidal Solution, Nano Lett., 2004, 4(7), 1343–1348, DOI:10.1021/nl0495256.
- F. Ma, S. L. Ma, K. W. Xu and P. K. Chu, Surface Stability of Platinum Nanoparticles Surrounded by High-Index Facets, J. Phys. Chem. C, 2008, 112(9), 3247–3251, DOI:10.1021/jp077324v.
- C. Xiao, B. A. Lu, P. Xue, N. Tian, Z. Y. Zhou, X. Lin, W. F. Lin and S. G. Sun, High-Index-Facet- and High-Surface-Energy Nanocrystals of Metals and Metal Oxides as Highly Efficient Catalysts, Joule, 2020, 4(12), 2562–2598, DOI:10.1016/j.joule.2020.10.002.
- Y. Kang, S. M. João, R. Lin, K. Liu, L. Zhu, J. Fu, W. C. Cheong, S. Lee, K. Frank, B. Nickel, M. Liu, J. Lischner and E. Cortés, Effect of Crystal Facets in Plasmonic Catalysis, Nat. Commun., 2024, 15(1), 3923, DOI:10.1038/s41467-024-47994-y.
- X. F. Yang, A. Wang, B. Qiao, J. Li, J. Liu and T. Zhang, Single-Atom Catalysts: A New Frontier in Heterogeneous Catalysis, Acc. Chem. Res., 2013, 46(8), 1740–1748, DOI:10.1021/ar300361m.
- J. A. Van Bokhoven and S. Vajda, Size Selected Clusters and Particles: From Physical Chemistry and Chemical Physics to Catalysis, Phys. Chem. Chem. Phys., 2014, 16(48), 26418–26420, 10.1039/c4cp90163k.
- I. Darmadi, F. A. A. Nugroho and C. Langhammer, High-Performance Nanostructured Palladium-Based Hydrogen Sensors – Current Limitations and Strategies for Their Mitigation, ACS Sens., 2020, 5(11), 3306–3327, DOI:10.1021/acssensors.0c02019.
- Y. Hayashi, H. Yamazaki, K. Masunishi, T. Ikehashi, N. Nakamura and A. Kojima, Effects of Poisoning Gases on and Restoration of PdCuSi Metallic Glass in a Capacitive MEMS Hydrogen Sensor, Int. J. Hydrogen Energy, 2020, 45(1), 1187–1194, DOI:10.1016/j.ijhydene.2019.10.245.
- S. Zarabi Golkhatmi, M. I. Asghar and P. D. Lund, A Review on Solid Oxide Fuel Cell Durability: Latest Progress, Mechanisms, and Study Tools, Renewable Sustainable Energy Rev., 2022, 161, 112339, DOI:10.1016/j.rser.2022.112339.
- X. Xu, Y. Zhong, M. Wajrak, T. Bhatelia, S. P. Jiang and Z. Shao, Grain Boundary Engineering: An Emerging Pathway toward Efficient Electrocatalysis, InfoMat, 2024, 6(8), e12608, DOI:10.1002/inf2.12608.
- H. Shooshtari Gugtapeh, A. H. Aghaii and A. Simchi, A Critical Review on the Role of Structural Defects in Electrochemical Nitrate Reduction Catalysts: From Mechanisms to Formation Strategies, J. Environ. Chem. Eng., 2025, 13(6), 120221, DOI:10.1016/J.JECE.2025.120221.
- A. Tittl, P. Mai, R. Taubert, D. Dregely, N. Liu and H. Giessen, Palladium-Based Plasmonic Perfect Absorber in the Visible Wavelength Range and Its Application to Hydrogen Sensing, Nano Lett., 2011, 11(10), 4366–4369, DOI:10.1021/nl202489g.
- C. Wadell, S. Syrenova and C. Langhammer, Plasmonic Hydrogen Sensing with Nanostructured Metal Hydrides, ACS Nano, 2014, 8(12), 11925–11940, DOI:10.1021/nn505804f.
- R. Griessen, N. Strohfeldt and H. Giessen, Thermodynamics of the Hybrid Interaction of Hydrogen with Palladium Nanoparticles, Nat. Mater., 2016, 15(3), 311–317, DOI:10.1038/nmat4480.
- C. Andersson, J. Zimmerman, J. Fritzsche, E. Rabkin and C. Langhammer, Hydride Formation Pressures and Kinetics in Individual Pd Nanoparticles with Systematically Varied Levels of Plastic Deformation, Nat. Commun., 2025, 16(1), 9242, DOI:10.1038/s41467-025-64311-3.
- R. Delmelle, B. Amin-Ahmadi, M. Sinnaeve, H. Idrissi, T. Pardoen, D. Schryvers and J. Proost, Effect of Structural Defects on the Hydriding Kinetics of Nanocrystalline Pd Thin Films, Int. J. Hydrogen Energy, 2015, 40(23), 7335–7347, DOI:10.1016/J.IJHYDENE.2015.04.017.
- S. Alekseeva, M. Strach, S. Nilsson, J. Fritzsche, V. P. Zhdanov and C. Langhammer, Grain-Growth Mediated Hydrogen Sorption Kinetics and Compensation Effect in Single Pd Nanoparticles, Nat. Commun., 2021, 12(1), 5427, DOI:10.1038/s41467-021-25660-x.
- X. Geng, S. Li, J. Heo, Y. Peng, W. Hu, Y. Liu, J. Huang, Y. Ren, D. Li, L. Zhang and L. Luo, Grain-Boundary-Rich Noble Metal Nanoparticle Assemblies: Synthesis, Characterization, and Reactivity, Adv. Funct. Mater., 2022, 32(34), 2204169, DOI:10.1002/adfm.202204169.
- N. J. J. Johnson, B. Lam, B. P. MacLeod, R. S. Sherbo, M. Moreno-Gonzalez, D. K. Fork and C. P. Berlinguette, Facets and Vertices Regulate Hydrogen Uptake and Release in Palladium Nanocrystals, Nat. Mater., 2019, 18(5), 454–458, DOI:10.1038/s41563-019-0308-5.
- J. Tang, S. Yamamoto, T. Koitaya, A. Yoshigoe, T. Tokunaga, K. Mukai, I. Matsuda and J. Yoshinobu, Mass Transport in the PdCu Phase Structures during Hydrogen Adsorption and Absorption Studied by XPS under Hydrogen Atmosphere, Appl. Surf. Sci., 2019, 480, 419–426, DOI:10.1016/J.APSUSC.2019.02.180.
- B. Ai, Y. Sun and Y. Zhao, Plasmonic Hydrogen Sensors, Small, 2022, 18(25), 2107882, DOI:10.1002/smll.202107882.
- Z. Su, C. Zhang, X. Zhang, W. Chen, X. Zhang, S. Wang, Y. Yang and Z. Wang, Probe of Nanocatalysts In-Action by Ambient Pressure Photoelectron Spectroscopy, Nano Res., 2025, 18(9), 94907709, DOI:10.26599/NR.2025.94907709.
- Y. Mathieu, L. Tzanis, M. Soulard, J. Patarin, M. Vierling and M. Molière, Adsorption of SOx by Oxide Materials: A Review, Fuel Process. Technol., 2013, 114, 81–100, DOI:10.1016/J.FUPROC.2013.03.019.
- Z. Say, M. Kaya, Ç. Kaderoğlu, Y. Koçak, K. E. Ercan, A. T. Sika-Nartey, A. Jalal, A. A. Turk, C. Langhammer, M. Jahangirzadeh Varjovi, E. Durgun and E. Ozensoy, Unraveling Molecular Fingerprints of Catalytic Sulfur Poisoning at the Nanometer Scale with Near-Field Infrared Spectroscopy, J. Am. Chem. Soc., 2022, 144(19), 8848–8860, DOI:10.1021/jacs.2c03088.
- X. Sun, J. Li, J. Fan, J. Fan, Q. Deng, M. Xu, H. Huang, J. Wu and D. Ye, Study on Sulfur-Resistant Pd-Based Catalysts for High-Efficiency Oxidation of VOCs, J. Hazard. Mater., 2025, 491, 137958, DOI:10.1016/J.JHAZMAT.2025.137958.
- W. Xu, J. S. Kong, Y.-T. E. Yeh and P. Chen, Single-Molecule Nanocatalysis Reveals Heterogeneous Reaction Pathways and Catalytic Dynamics, Nat. Mater., 2008, 7(12), 992–996, DOI:10.1038/nmat2319.
- J. I. J. Choi, T.-S. Kim, D. Kim, S. W. Lee and J. Y. Park, Operando Surface Characterization on Catalytic and Energy Materials from Single Crystals to Nanoparticles, ACS Nano, 2020, 14(12), 16392–16413, DOI:10.1021/acsnano.0c07549.
- I. M. N. Groot, Investigation of Active Catalysts at Work, Acc. Chem. Res., 2021, 54(23), 4334–4341, DOI:10.1021/acs.accounts.1c00429.
- O. M. Busch, W. Brijoux, S. Thomson and F. Schüth, Spatially Resolving Infrared Spectroscopy for Parallelized Characterization of Acid Sites of Catalysts via Pyridine Sorption: Possibilities and Limitations, J. Catal., 2004, 222(1), 174–179, DOI:10.1016/J.JCAT.2003.11.002.
- M. B. J. Roeffaers, G. De Cremer, J. Libeert, R. Ameloot, P. Dedecker, A. J. Bons, M. Bückins, J. A. Martens, B. F. Sels, D. E. De Vos and J. Hofkens, Super-Resolution Reactivity Mapping of Nanostructured Catalyst Particles, Angew. Chem., Int. Ed., 2009, 48(49), 9285–9289, DOI:10.1002/anie.200904944.
- M. A. Karreman, I. L. C. Buurmans, J. W. Geus, A. V. Agronskaia, J. Ruiz-Martínez, H. C. Gerritsen and B. M. Weckhuysen, Integrated Laser and Electron Microscopy Correlates Structure of Fluid Catalytic Cracking Particles to Brønsted Acidity, Angew. Chem., Int. Ed., 2012, 51(6), 1428–1431, DOI:10.1002/anie.201106651.
- J. Ruiz-Martínez, A. M. Beale, U. Deka, M. G. O’Brien, P. D. Quinn, J. F. W. Mosselmans and B. M. Weckhuysen, Correlating Metal Poisoning with Zeolite Deactivation in an Individual Catalyst Particle by Chemical and Phase-Sensitive X-Ray Microscopy, Angew. Chem., Int. Ed., 2013, 52(23), 5983–5987, DOI:10.1002/anie.201210030.
- C.-Y. Wu, W. J. Wolf, Y. Levartovsky, H. A. Bechtel, M. C. Martin, F. D. Toste and E. Gross, High-Spatial-Resolution Mapping of Catalytic Reactions on Single Particles, Nature, 2017, 541(7638), 511–515, DOI:10.1038/nature20795.
- S. Dery, S. Kim, D. Haddad, A. Cossaro, A. Verdini, L. Floreano, F. D. Toste and E. Gross, Identifying Site-Dependent Reactivity in Oxidation Reactions on Single Pt Particles, Chem. Sci., 2018, 9(31), 6523–6531, 10.1039/C8SC01956H.
- X. Mao, C. Liu, M. Hesari, N. Zou and P. Chen, Super-Resolution Imaging of Non-Fluorescent Reactions via Competition, Nat. Chem., 2019, 11(8), 687–694, DOI:10.1038/s41557-019-0288-8.
- Z. Li and D. Kurouski, Plasmon-Driven Chemistry on Mono- and Bimetallic Nanostructures, Acc. Chem. Res., 2021, 54(10), 2477–2487, DOI:10.1021/acs.accounts.1c00093.
- S. Dery, H. Mehlman, L. Hale, M. Carmiel-Kostan, R. Yemini, T. Ben-Tzvi, M. Noked, F. D. Toste and E. Gross, Site-Independent Hydrogenation Reactions on Oxide-Supported Au Nanoparticles Facilitated by Intraparticle Hydrogen Atom Diffusion, ACS Catal., 2021, 11(15), 9875–9884, DOI:10.1021/acscatal.1c01987.
- L. Rikanati, S. Dery and E. Gross, AFM-IR and s-SNOM-IR Measurements of Chemically Addressable Monolayers on Au Nanoparticles, J. Chem. Phys., 2021, 155(20), 204704, DOI:10.1063/5.0072079.
- S. Dery, B. Friedman, H. Shema and E. Gross, Mechanistic Insights Gained by High Spatial Resolution Reactivity Mapping of Homogeneous and Heterogeneous (Electro)Catalysts, Chem. Rev., 2023, 123(9), 6003–6038, DOI:10.1021/acs.chemrev.2c00867.
- A. Dazzi and C. B. Prater, AFM-IR: Technology and Applications in Nanoscale Infrared Spectroscopy and Chemical Imaging, Chem. Rev., 2017, 117(7), 5146–5173, DOI:10.1021/acs.chemrev.6b00448.
- D. Kurouski, A. Dazzi, R. Zenobi and A. Centrone, Infrared and Raman Chemical Imaging and Spectroscopy at the Nanoscale, Chem. Soc. Rev., 2020, 49(11), 3315–3347, 10.1039/C8CS00916C.
- H. Fredriksson, Y. Alaverdyan, A. Dmitriev, C. Langhammer, D. S. Sutherland, M. Zäch and B. Kasemo, Hole–Mask Colloidal Lithography, Adv. Mater., 2007, 19(23), 4297–4302, DOI:10.1002/adma.200700680.
- N. Hoshi, M. Kuroda, T. Ogawa, O. Koga and Y. Hori, Infrared Reflection Absorption Spectroscopy of the Sulfuric Acid Anion Adsorbed on Pd(S)–[n(111) × (111)] Electrodes, Langmuir, 2004, 20(12), 5066–5070, DOI:10.1021/la036149g.
- M. S. Wilburn and W. S. Epling, Formation and Decomposition of Sulfite and Sulfate Species on Pt/Pd Catalysts: An SO2 Oxidation and Sulfur Exposure Study, ACS Catal., 2019, 9(1), 640–648, DOI:10.1021/acscatal.8b03529.
- A. B. Horn and K. Jessica Sully, ATR-IR Spectroscopic Studies of the Formation of Sulfuric Acid and Sulfuric Acid Monohydrate Films, Phys. Chem. Chem. Phys., 1999, 1(16), 3801–3806, 10.1039/A904544I.
| This journal is © The Royal Society of Chemistry 2026 |