Open Access Article
Open Access ArticleImpact of pressing process on the chemical and nutritional characteristics of flaxseed and hempseed cakes
Francesca Carrà†
,
Vincenzo Disca† ,
Antonio Colasanto,
Marco Arlorio,
Matteo Bordiga,
Jean Daniel Coïsson,
Monica Locatelli
,
Antonio Colasanto,
Marco Arlorio,
Matteo Bordiga,
Jean Daniel Coïsson,
Monica Locatelli * and
Fabiano Travaglia
* and
Fabiano Travaglia
Dipartimento di Scienze del Farmaco, Università del Piemonte Orientale, Novara, 28100, Italy. E-mail: monica.locatelli@uniupo.it
First published on 27th March 2026
Abstract
The increasing demand for high-quality plant-derived oils has generated large quantities of press cakes as by-products of mechanical extraction. This study investigates the impact of screw-pressing conditions on the chemical, nutritional and functional properties of flaxseed and hempseed cakes. Whole flaxseeds and hempseeds were mechanically pressed at two rotor velocities (20% and 80%), and unprocessed seeds and their press cakes were analyzed for proximate composition, fatty acid profile, total phenolic content (TPC), antioxidant activity (AA) through the DPPH assay with the QUENCHER method, and individual phenolic compounds by HPLC-DAD. Press cakes exhibited significantly higher protein and fiber content compared to the whole seeds. Flaxseed press cakes reached up to approximately 36% protein versus 22.7% in whole flaxseed, and 35.8% fiber for both flaxseed cakes. Hempseed press cakes presented a higher fiber component in the range of 45–50%. AA increased markedly in all press cakes, particularly in flaxseed cakes obtained at high velocity, which showed 34.2 µmol TE g−1 compared to 14.8 µmol TE g−1 in whole flaxseed. TPC and HPLC analyses revealed that lower pressing velocity preserved more free phenolic compounds, while higher velocity occasionally led to degradation, especially of heat-sensitive molecules. Finally, despite the reduced oil fraction, the residual lipid fraction is characterized by favorable fatty acid profiles, dominated by α-linolenic acid (ω-3) in flax and linoleic acid (ω-6) in hemp, maintaining balanced ω-6/ω-3 ratios. These findings highlight the potential of flaxseed and hempseed press cakes as functional food ingredients and emphasize the role of pressing conditions in determining their nutritional and antioxidant properties.
Sustainability spotlightThis study addresses sustainable food systems by valorizing flaxseed and hempseed press cakes, which are byproducts of mechanical oil extraction, into potential nutrient rich, functional food ingredients. By demonstrating that these by-products are high in protein, dietary fiber, and phenolics and have valuable fatty acid profiles, the study directly supports SDG 12 (Responsible Consumption and Production) through waste reduction and circular economy principles. Furthermore, increasing the availability of low-cost, plant-based sources of high-quality protein and fiber supports SDGs 2 (Zero Hunger) and 3 (Good Health and Well-Being) by promoting functional foods. |
1. Introduction
Every year, the food industry produces and discards a large amount of biodegradable waste and by-products, produced at various stages of the supply chain. In Europe, the total waste from food processing amounts to 30.6 million tonnes per year,1 most of it generated by various sub-sectors of the pressing stage.The increasing demand for high-quality plant-derived-oils has led to the generation of significant amounts of oilseed press cakes, obtained during mechanical oil extraction; these by-products are constantly growing,2 as is their use in food and nutraceutical sectors.3,4 In general, the cakes obtained from the extraction of soybean, sunflower, flax, hemp, etc., are rich in residual oil, bioactive compounds,3 and 40–50% of the proteins and fiber found in the original seeds.5 Despite their high nutritional value, traditionally they are mainly used as animal feed. However, in the context of sustainability and a circular economy approach, there is growing interest in valorizing these by-products, transforming them into value-added food ingredients rather than waste.
Flaxseed (Linum usitatissimum L.) and hempseed (Cannabis sativa L.) are examples of high-value oilseeds, whose press cakes show promise as nutrient-rich by-products. The residue from the pressing procedure of flaxseed contains abundant high-quality protein, soluble fiber, minerals and health-promoting lignans, as well as a residual omega-3 fatty acids. In particular, the remaining oil is especially rich in α-linolenic acid and linoleic acid.6 Moreover, the protein fraction of flaxseed cakes can have functional uses in food technology like emulsifying, and their lignans and omega-3 fatty acids add nutraceutical value.2 Different studies have highlighted the antioxidant potential of the bioactive compounds,2 which contribute to the stability and functional properties of the cakes.
Similarly, hempseed cake is notable for its protein and fiber levels as well as for its fatty-acid profile. Hemp seeds contain approximately 30% oil by weight, with a characteristic linoleic:linolenic ratio of approximately 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.7 After mechanical extraction, hemp cakes have been shown to maintain the same balanced omega-6/omega-3 PUFA ratio7 and significant amounts of oil and protein. Moreover, dehulling the seeds prior to pressing has been shown to improve the protein quality and digestibility of the cakes. As flaxseed and hempseed cakes retain these valuable components, they are of great interest for waste valorization.
1.7 After mechanical extraction, hemp cakes have been shown to maintain the same balanced omega-6/omega-3 PUFA ratio7 and significant amounts of oil and protein. Moreover, dehulling the seeds prior to pressing has been shown to improve the protein quality and digestibility of the cakes. As flaxseed and hempseed cakes retain these valuable components, they are of great interest for waste valorization.
Understanding the influence of the press type and its pressing conditions on bioactive compound retention is essential for optimizing processing techniques and improving the functionality of oilseed-derived residues.8–12 Screw pressing, a mechanical pressing method, is widely used, especially on a smaller scale, due to its simplicity and low cost. There are many studies that demonstrate that pressing parameters could influence the characteristics of oils,13,14 but there are few studies that evaluate the effect of screw speed on the obtained cakes. Unlike solvent extraction, which removes more than 99% of the oil, mechanical pressing typically leaves a substantial fraction of oil in the seedcake, often over 10% of the original fat.3 As a result, screw-pressed cakes are rich in residual oil and fiber, and contain slightly less protein than solvent-extracted meals, which may be due to the fact that solvent extraction allows more oil to be extracted than the pressing process, as reported by Ixtaina et al. (2011) for chia seeds.15 Faster pressing often results in higher residual oil in the cake but may reduce efficiency and affect the retention of heat-sensitive compounds such as phenolics and tocopherols.16 The residual oil imparts a high energy value and a PUFA profile to the oilcake. Pressing parameters, seed's moisture, temperature, further influence the moisture content, and nutritional composition of the cake.
From these considerations, the present study evaluates whether screw-pressing conditions could influence the chemical and nutritional composition of flaxseed and hempseed cakes. By characterizing their proximate, fatty-acid profiles, phenolic content, and antioxidant activity, our aim is to highlight their usefulness for food applications, transforming what would otherwise be waste into a nutrient-rich resource.
2. Experimental
2.1. Sample preparation
The analyzed samples consist of flaxseeds, hemp seeds, and their corresponding press cakes, the by-products obtained after the extraction of vegetable oils via mechanical pressing. These samples were kindly provided by F.lli Ruata S.p.A. (Baldissero d’Alba, CN, Italy).The cakes were collected during the production of single-seed oils, using a screw press under two different operating conditions: 20% and 80% speed. For flaxseed, three independent samples were taken during the process, while for hempseed, two independent subsamples were collected. Samples and the speed used are summarized in Table 1. Samples were first ground using a Sterilmixer 12 blade homogenizer, and the resulting product was characterized. Moreover, following the Soxhlet extraction process, the samples were treated in a ball mill.
| Seed type | Sample coding | Sample type |
|---|---|---|
| Flaxseed | F | Flaxseed |
| Flaxseed | FC20 | Flaxseed cake obtained with 20% press speed |
| Flaxseed | FC80 | Flaxseed cake obtained with 80% press speed |
| Hempseed | H | Hempseed |
| Hempseed | HC20 | Hempseed cake obtained with 20% press speed |
| Hempseed | HC80 | Hempseed cake obtained with 80% press speed |
2.2. Proximate composition analysis
The ground samples were analyzed as follows: moisture content by the air oven method (AOAC 925.10), total protein by the Kjeldahl method (Ntot × 6.25), lipid content by the Soxhlet method using dichloromethane as solvent (AOAC 935.38), and ash content by AOAC 935.03; the fiber content analysis, determined through the AOAC 991.43 method, was conducted on the defatted sample. Available carbohydrate content was calculated by difference, applying the following formula:17| Available carbohydrates (%) = 100 – (fat + protein + ash + water + dietary fiber) |
This calculation assumes that any unmeasured components in the sample are carbohydrates, offering a practical and widely accepted method for estimating carbohydrate content.
The energy value of the samples was determined in accordance with EU Directive 1169/2011. Total energy (kcal per 100 g of product and kJ per 100 g of product) was calculated using the following equations:
| Energy (kcal/100 g) = (carbohydrates × 4) + (protein × 4) + (fat × 9) + (fiber × | (2) |
| Energy (kJ/100 g) = (carbohydrates × 17) + (protein × 17) + (fat × 37) + (fiber × 8) |
2.3. Lipid fraction composition
The relative content of the fatty acids was evaluated on the lipid fraction obtained from flax and hemp seeds and their corresponding cakes using two different extraction methods: Soxhlet extraction (S) was performed using dichloromethane at 60 °C for 6 h (continuous reflux cycle). Cold extraction (H) was carried out with n-hexane at room temperature using a solid-to-solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 (g mL−1) on a tilting plate for 1 h; the extraction was repeated twice and the supernatants were pooled. The procedure was reported by Locatelli et al. (2011).18 Briefly 200 µL of sodium methylate (0.5 mol L−1) were added to 0.20 g of lipid. The mixture reacted in a closed vial at 80 °C under stirring for 10 min, 350 rpm (Thermomixer comfort, Eppendorf, Milan, Italy). The content of each vial was then supplied with 250 µL of distilled water and 500 µL of diethyl ether, and fatty acid methyl esters (FAMEs) were gently extracted by inversion of the vial. 50 µL of the organic phase were diluted with 1 mL of CH2Cl2. The analysis was carried out using a Thermo Trace 1300 Gas Chromatograph (Thermo Fisher GmbH, Kandel, Germany), equipped with a flame ionization detector (FID) and a DB-23 J&W column (30 m; i.d. 0.25 mm; film thickness 0.25 µm, J&W Scientific, Folsom, CA, USA). The injector and the FID were set to 250 °C. Hydrogen was utilized as the carrier gas with a flow rate of 1.5 mL min−1 and a split ratio of 50
4 (g mL−1) on a tilting plate for 1 h; the extraction was repeated twice and the supernatants were pooled. The procedure was reported by Locatelli et al. (2011).18 Briefly 200 µL of sodium methylate (0.5 mol L−1) were added to 0.20 g of lipid. The mixture reacted in a closed vial at 80 °C under stirring for 10 min, 350 rpm (Thermomixer comfort, Eppendorf, Milan, Italy). The content of each vial was then supplied with 250 µL of distilled water and 500 µL of diethyl ether, and fatty acid methyl esters (FAMEs) were gently extracted by inversion of the vial. 50 µL of the organic phase were diluted with 1 mL of CH2Cl2. The analysis was carried out using a Thermo Trace 1300 Gas Chromatograph (Thermo Fisher GmbH, Kandel, Germany), equipped with a flame ionization detector (FID) and a DB-23 J&W column (30 m; i.d. 0.25 mm; film thickness 0.25 µm, J&W Scientific, Folsom, CA, USA). The injector and the FID were set to 250 °C. Hydrogen was utilized as the carrier gas with a flow rate of 1.5 mL min−1 and a split ratio of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Fatty acids were identified by comparing their retention times with those of a reference mixture (Supelco 37 Component FAME mix, Sigma-Aldrich, Germany). Quantification was carried out by calculating each fatty acid's contribution to the sum of chromatographic peaks area, and expressing the results as internal relative percentages.
1. Fatty acids were identified by comparing their retention times with those of a reference mixture (Supelco 37 Component FAME mix, Sigma-Aldrich, Germany). Quantification was carried out by calculating each fatty acid's contribution to the sum of chromatographic peaks area, and expressing the results as internal relative percentages.
2.4. Antioxidant activity
The antioxidant activity of the samples was measured by free-radical scavenging against 2,2-dipheny-l-1-picrylhydrazyl (DPPH) radicals, directly on solid powder, adopting the QUENCHER method described by Locatelli et al. (2010).19 Defatted and ground samples were sieved (<250 µm). 0.5 mg was weighed, and then 700 µL of water (hemp samples) or 1.4 mL of water (flax samples) was added and the reaction was started by adding 700 µL or 1.4 mL (for hemp and flax samples, respectively) of DPPH˙ working solution (20 °C, 100 rpm). After 30 minutes the absorbance was measured at 515 nm. All samples were analysed in triplicate, and the antioxidant activity was calculated in µmol of Trolox equivalent (TE) g−1 of dry weight sample (d.w.) through a Trolox calibration curve.2.5. Extraction of phenolic compounds
The extraction of free polyphenols was performed on the defatted and ground samples using a mixture of methanol, acetone and distilled water in a 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7
7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 (v/v/v) ratio. 50 mg of the defatted sample, previously ground, was weighed into 2 mL tubes. One millilitre of solvent was added, and the sample was vortexed for 10 s and sonicated for 2 min (Bransonic 1510). The extract was then centrifuged at 20.800 g for 2 min (centrifuge, Eppendorf 5417 R). The supernatant was collected in a 15 mL conical tube, and the extraction was repeated twice on the residual pellet. The combined extracts were collected and stored at −20 °C until the analysis.
6 (v/v/v) ratio. 50 mg of the defatted sample, previously ground, was weighed into 2 mL tubes. One millilitre of solvent was added, and the sample was vortexed for 10 s and sonicated for 2 min (Bransonic 1510). The extract was then centrifuged at 20.800 g for 2 min (centrifuge, Eppendorf 5417 R). The supernatant was collected in a 15 mL conical tube, and the extraction was repeated twice on the residual pellet. The combined extracts were collected and stored at −20 °C until the analysis.
The bound polyphenol fraction was extracted from the residual pellet obtained after free polyphenol extraction as reported by Colasanto et al. (2025).20 10 mL of 4 mol L−1 NaOH were added to the solid residue and the mixture was left stirring for 210 min. The pH was adjusted to 2.3 using 10 mol L−1 HCl. The polyphenols were subsequently extracted using ethyl acetate in a separatory funnel, performing three successive extractions. The organic phases were combined in a 100 mL round-bottom flask, and the solvent was evaporated under reduced pressure using a rotary evaporator (Rotavapor® BUCHI R-210, Flawil, Switzerland). The resulting residue was dissolved in 2 mL of HPLC-grade methanol and stored at −20 °C until the analysis.
2.6. Total phenolic content
The determination of total phenolic content (TPC) followed the classical Folin–Ciocâlteu assay, as outlined in Papillo et al. (2019) work,21 and was conducted on the extracts containing polyphenols in their free form. Briefly, 50 µL of Folin–Ciocalteu's reagent (Carlo Erba) and 175 µL of aqueous Na2CO3 (5%, w/v) were added to the extracts. The solutions were then diluted with water to a final volume of 1.45 mL. The absorbance was read at 760 nm after one hour, using a Shimadzu UV-1900 spectrophotometer (Shimadzu, Tokyo, Japan). The results were expressed as mg gallic acid equivalent (GAE) g−1 of dry weight (d.w.) samples.2.7. HPLC-DAD analysis
The quantitative analysis of free and bound phenolic compounds was performed using reverse-phase high-performance liquid chromatography with a diode array detector (RP-HPLC-DAD) (Shimadzu LC-20 AD Prominence, with SPD-M20a detector). The separation was performed on a Luna C-18 (2) column (150 × 2 mm, 5 µm, Phenomenex, Torrance, CA, USA) with a silica support and pre-column protection. For the separation, the mobile phase consisted of water with 0.1% formic acid (solvent A) and methanol with 0.1% formic acid (solvent B), with a flow rate of 400 µL min−1. The detection wavelengths were set to 280 and 330 nm, with injection volumes of 5 µL. The gradient program was structured as follows: 5–17.5% B (30 min), 17.5–30% B (10 min), 30–100% B (5 min), isocratic at 100% B (10 min), returning to 5% B in 1 min, followed by an isocratic step at 5% B for 19 min with a total separation time of 75 minutes. Quantification was carried out by interpolation of calibration curves of the correspondent molecule (gallic, protocatechuic, p-hydroxybenzoic, vanillic, caffeic, p-coumaric, ferulic, ellagic acids and epicatechin, myricetin and quercetin).2.8. Statistical analysis
The statistical analysis was conducted using R software 4.3.0 (ref. 22) and GraphPad Prism (10.4.2 version). The one-factor analysis of variance (ANOVA) followed by the Tukey multiple comparison test was used to determine significant differences (p < 0.05) between samples.3. Results and discussion
3.1. Proximate composition
The proximate composition of flaxseed (F, FC20, and FC80) and hempseed (H, HC20, and HC80) samples revealed significant differences in nutritional profiles (Table 2). These differences are particularly pronounced in moisture, lipid, protein, and dietary fiber contents, which influence the energy density of the products. The defatting of flax and hemp seeds by mechanical pressing markedly concentrated the residual nutrients in the press cakes.| Flaxseed | Hempseed | |||||
|---|---|---|---|---|---|---|
| F | FC20 | FC80 | H | HC20 | HC80 | |
| Moisture | 5.78 ± 0.04b | 9.02 ± 0.31a | 8.30 ± 0.50a | 8.43 ± 0.12a | 8.24 ± 0.60a | 8.35 ± 1.19a |
| Ashes | 3.14 ± 0.0c | 4.81 ± 0.01a | 4.63 ± 0.09b | 4.02 ± 0.16c | 5.35 ± 0.06a | 5.06 ± 0.12b |
| Lipids | 30.1 ± 1.3a | 12.6 ± 0.5b | 15.0 ± 0.1b | 26.5 ± 3.5a | 9.14 ± 0.37b | 8.51 ± 0.09c |
| Protein | 22.7 ± 0.04b | 35.9 ± 0.5a | 34.7 ± 0.3a | 22.5 ± 0.5b | 30.6 ± 1.4a | 29.9 ± 0.7a |
| Total dietary fiber | 33.9 ± 0.6a | 35.8 ± 1.0a | 35.8 ± 0.4a | 37.6 ± 0.6c | 45.4 ± 1.5b | 50.2 ± 1.9a |
| Available carbohydrates | 10.1 | 10.9 | 9.77 | 9.44 | 9.49 | 6.29 |
| Energy (kcal/100g) | 470 | 372 | 384 | 373 | 287 | 322 |
| Energy (kJ/100g) | 1943 | 1548 | 1597 | 1824 | 1383 | 1331 |
Moisture content was higher in FC20 and FC80 (9.02% and 8.30%, respectively) compared to unpressed flaxseed (5.78%), in contrast with the results observed for hempseed cakes, where HC20 and HC80 had moisture contents not significantly different from the seeds (H). The results obtained are partially in line with those reported by Jakab et al. (2025), who analyzed flax, hemp, hazelnut and pumpkin seeds and seedcakes and reported that the moisture content was higher in cold-pressed cakes.23 Elevated moisture content may depend on the characteristics of the pressing process itself. Mechanical pressing breaks cell walls, increasing porosity and exposing a larger surface of the cake to the environment, thus making it more hygroscopic and facilitating water absorption. This can potentially affect the stability and the shelf-life of the product, as higher moisture levels are associated with greater susceptibility to microbial spoilage and oxidative degradation. In addition, processing parameters, such as screw-speed can further affect the properties of the cake; for example, it has been shown that increasing the rpm promotes greater moisture retention in the resulting pressed cake.24
The ash content, indicative of total mineral content, was higher in all the cakes types than in raw seeds, especially in HC20 (5.35%) and FC20 (4.81%). A similar trend was observed in the work of Ferreira et al. (2023), who characterized chia seeds and the defatted cake obtained using a screw press, finding that the ash content in the residues obtained was significantly higher (6.42%) than that of the starting raw material (4.67%).25 In addition, focusing on the lipid content it sharply declined in all pressed cakes. HC20 (9.14%) and HC80 (8.51%) showed a lower percentage compared to raw hempseed (26.46%), as did the flaxseed counterparts, where the starting matrix (F) contained 30.1% oil, while the by-products obtained contained 12.6% and 15.0% (FC20 and FC80, respectively). The amount of leftover oil, that remained in the pressed cakes could depend on the screw speed applied as it determines how long the seeds will be pressed.26 Furthermore, reducing the lipid content, in addition to lowering the caloric value, can have an impact on the bioactive lipid profile causing changes in the content of triglycerides, phospholipids and tocopherols key molecules that contribute to nutritional quality and oxidative stability.27
In contrast to the lipid content, the protein content increased markedly in the pressed cakes (35.87% and 34.7%, for FC20 and FC80, and 30.6% and 29.9%, for HC20 and HC80) samples compared to their respective raw materials. This enhancement could be attributed to the relative concentration of proteins following oil removal using screw pressing. No significant differences were observed between the two by-products obtained at different screw speeds, indicating that screw speed did not markedly influence the protein fraction under the tested conditions. Moreover, the results obtained for flaxseed cakes are consistent with the findings reported by Talwar et al. (2025), who highlighted that the protein content in flaxseed press cakes may vary between 27.78% and 38.40%.2 Similarly, the results obtained for hempseed cakes agree with those of Capcanari et al. (2023), who reported a protein content of approximately 31.62% in cold-pressed hempseed cakes.8 Moreover, hemp proteins have high digestibility and for this reason this by-product is widely studied in food products.28 The high protein levels in flaxseed and hempseed cakes support their potential application in high-protein diets.
Finally, a key nutritional feature of these cakes is their total dietary fiber content, which remained high across all samples (33.9–50.2%). Hempseed cakes, particularly HC80, exhibited the highest fiber content (50.2%), which may contribute to delayed gastric emptying, enhanced satiety, and improved glycemic response. The elevated fiber content may exert positive effects on gut microbiota, with benefits for host metabolism. For instance, microbial enzymes can complement the activity of endogenous digestive enzymes present in the gastrointestinal tract and liver, contributing to the production of metabolites such as short-chain fatty acids29 and also inhibiting the proliferation of pathogenic bacteria through competitive exclusion and the secretion of antimicrobial compounds. For these reasons fiber-enriched products are widely recognized for their beneficial effects on gut health and chronic disease prevention.
Despite variations in macronutrient composition, available carbohydrates remained relatively consistent, ranging from 6.29% to 10.9%, with slightly lower values in HC80. Given the generally low carbohydrate levels, these ingredients may be suitable for low-glycemic or ketogenic dietary applications. The proximate composition analysis of flax and hemp seeds and their corresponding press cakes revealed that the seeds presented a significantly higher energy value compared to the cakes, primarily due to the removal of lipids during the screw-pressing process. As a result of macronutrient shifts, the energy values decreased in the pressed cakes (e.g., from 470 kcal/100 g in flaxseed (F) to 372 kcal and 384 kcal in FC20 and FC80, respectively).
Overall, pressing speed had a negligible impact on macronutrients such as protein and fiber.
3.2. Fatty acid composition
Lipids from flax and hemp are rich in polyunsaturated fatty acids, notably α-linolenic acid (ω-3) and linoleic acid (ω-6), which have established roles in cardiovascular and metabolic health.30The fatty (FA) acid composition was first determined on the lipid fraction obtained from lipid Soxhlet extraction using dichloromethane (S), and then the lipid fraction obtained after cold hexane extraction (H), under magnetic stirring, was also evaluated; the results are reported in Table 3. Hempseed samples, obtained through Soxhlet extraction, maintained their characteristic profile, with LA as the predominant PUFA, with values around 55–56%, and α-linolenic acid present in lower quantities, remaining approximately 13% across the seed and its cakes. The profile is in line with what is reported by Montero et al. (2023), who reported that the ALA content for hempseed oil ranges between 15.2% and 26.2%, while LA content varies between 55.1% and 63.7%.31 In contrast, the FA profile of the flaxseed samples showed differences relative to literature reports; however, these discrepancies are most plausibly explained by extraction-related artifacts, as detailed later, rather than genuine compositional variability. Indeed, flaxseed oil is commonly dominated by higher amounts of α-linolenic acid (ALA) and then by the presence of linoleic (LA) and oleic acid.32 Soxhlet-extracted samples, in the present study, exhibited lower relative percentages of ALA than expected, with linoleic acid emerging as the predominant FA. Specifically, the LA content ranged from 38.8% in F–S to 46.0% in FC20-S, while ALA varied between 22.1% (FC20-S) and 29.6% (F–S). As F–S, FC20-S and FC80-S presented an unexpected profile with low ALA content, a second extraction was carried out under cold conditions, using hexane, to minimize thermal stress. The cold hexane extraction (H) of the flaxseed sample yielded FA compositions (Table 4 and Fig. 1) that contrasted with the Soxhlet results. In F–H, ALA reached 37.2%, a higher value than LA content (29.7%), thereby restoring he typical ALA-rich composition that characterizes flaxseed oil. These findings indicate that the reduced levels of ALA observed after Soxhlet extraction are likely an extraction-induced artifact associated with prolonged thermal exposure. This deviation from the normal FA profile is consistent with the known thermal lability of some fatty acids, such as α-linolenic acid, which, by their nature, are more susceptible to oxidation at high temperatures. In fact, Zhen-Shan et al.'s (2013) findings showed that linseed oil subjected to high temperatures over time undergoes a change in its fatty acid composition.33 The FA profile of flaxseed cakes significantly differed from that of flaxseed, with linoleic acid remained the predominant FA component. FC20-H and FC80-H still showed a relative percentage of α-linolenic acid (23.2% and 23.5%, respectively) lower than that of linoleic acid (45.9% and 45.5%, respectively). This finding could be due to the fact that cakes are composed of the residual lipid fraction after the removal of the major oil portion, suggesting a potential impact of screw pressing on the FA profile. As a proof of this, a similar trend was already observed in the flax seed oils extracted through screw pressing. Pedron et al. (2025) showed that flaxseed oils were richer in LA than ALA and this may depend on the temperature of the screw press process, as well as from possible differences in the composition of the seeds.34 Differently, in hempseed samples extracted under cold hexane extraction conditions, the relative abundance of FAMEs did not vary depending on the solvent or extraction conditions. This is in agreement with what is reported by Disca et al. (2025), who observed no significant differences in the fatty acid composition of cocoa bean shell lipids extracted by Soxhlet using four different apolar solvents.35 In fact, focusing on LA and ALA percentages in hempseed samples (Fig. 1), the inversion in the percentage of these two FAs was not observed. This behavior may be attributed to the substantially lower relative abundance of ALA in hempseed oil compared with flaxseed oil, which reduces the impact of extraction temperature on its relative proportion. In contrast, the very high ALA content of flaxseed oil makes it more susceptible to thermal- and solvent-related effects during prolonged extraction, leading to measurable compositional differences between Soxhlet and cold extraction methods.36 Accordingly, no statistical differences were detected between the two different extraction methods (Tables 3 and 4).
| Flaxseed | Hempseed | ||||||
|---|---|---|---|---|---|---|---|
| F | FC20 | FC80 | H | HC20 | HC80 | ||
| a F: flaxseed; FC20: flaxseed cake 20% speed; FC80: flaxseed cake 80% speed; H: hempseed; HC20: hempseed cake 20% speed; HC80: hempseed cake 80% speed. Results are expressed as mean ± standard deviation (n = 3). | |||||||
| Myristic acid | C16:0 | 6.32 ± 0.17c | 7.86 ± 0.07a | 7.54 ± 0.09b | 7.59 ± 0.17b | 8.20 ± 0.05a | 8.24 ± 0.09a |
| Palmitic acid | C18:0 | 5.16 ± 0.11a | 3.92 ± 0.10b | 4.03 ± 0.10b | 3.24 ± 0.20 | 3.15 ± 0.03 | 3.17 ± 0.02 |
| Oleic acid | C18:1n9cis | 19.1 ± 0.2 | 19.1 ± 0.2 | 19.0 ± 0.4 | 14.2 ± 0.7a | 13.3 ± 0.1b | 13.2 ± 0.04b |
| Linoleic acid | C18:2n6cis | 38.8 ± 0.1b | 46.0 ± 0.8a | 45.6 ± 1.3a | 55.9 ± 0.3 | 55.6 ± 0.1 | 55.7 ± 0.1 |
| γ-Linolenic acid | C18:3n6 | — | — | — | 3.69 ± 0.14 | 3.7 ± 0.05 | 3.71 ± 0.04 |
| α-Linolenic acid | C18:3n3 | 29.6 ± 0.6a | 22.1 ± 0.8b | 22.7 ± 1.0b | 12.9 ± 0.5 | 13.4 ± 0.1 | 13.4 ± 0.1 |
| Arachidic acid | C20:0 | 0.18 ± 0.01 | 0.19 ± 0.01 | 0.18 ± 0.01 | 0.99 ± 0.07 | 0.99 ± 0.02 | 1.00 ± 0.03 |
| Eicosenoic acid | C20:1 | 0.12 ± 0.00b | 0.13 ± 0.01a | 0.13 ± 0.00 ab | 0.41 ± 0.02 | 0.44 ± 0.03 | 0.47 ± 0.03 |
| Eicosadienoic acid | C20:2 | — | — | — | 0.10 ± 0.01b | 0.17 ± 0.01a | 0.14 ± 0.03 ab |
| Behenic acid | C22:0 | 0.18 ± 0.00 | 0.19 ± 0.01 | 0.18 ± 0.01 | 0.42 ± 0.04 | 0.45 ± 0.02 | 0.45 ± 0.01 |
| Lignoceric acid | C24:0 | 0.13 ± 0.00b | 0.19 ± 0.01a | 0.18 ± 0.01a | 0.21 ± 0.02 | 0.21 ± 0.01 | 0.21 ± 0.02 |
| ω-6/ω-3 | 1.29 | 2.08 | 2.00 | 4.59 | 4.43 | 4.44 | |
| Flaxseed | Hempseed | ||||||
|---|---|---|---|---|---|---|---|
| F | FC20 | FC80 | H | HC20 | HC80 | ||
| a F: flaxseed; FC20: flaxseed cake 20% speed; FC80: flaxseed cake 80% speed; H: hempseed; HC20: hempseed cake 20% speed; HC80: hempseed cake 80% speed. The results are expressed as mean ± standard deviation (n = 3). | |||||||
| Myristic acid | C16:0 | 6.15 ± 0.15c | 7.44 ± 0.09a | 7.22 ± 0.07b | 7.37 ± 0.06b | 7.95 ± 0.10a | 7.96 ± 0.13a |
| Palmitic acid | C18:0 | 5.52 ± 0.55a | 3.82 ± 0.08b | 4.04 ± 0.10b | 3.03 ± 0.02 | 3.05 ± 0.03 | 3.09 ± 0.09 |
| Oleic acid | C18:1n9cis | 20.4 ± 1.3a | 18.6 ± 0.1b | 18.6 ± 0.2b | 13.9 ± 0.03a | 13.3 ± 0.1b | 13.3 ± 0.2b |
| Linoleic acid | C18:2n6cis | 29.7 ± 0.8b | 45.9 ± 0.7a | 45.5 ± 1.4a | 56.4 ± 0.2 | 56.2 ± 0.1 | 56.0 ± 0.2 |
| γ-Linolenic acid | C18:3n6 | — | — | — | 3.83 ± 0.02 | 3.72 ± 0.01 | 3.72 ± 0.07 |
| α-Linolenic acid | C18:3n3 | 37.2 ± 0.9a | 23.2 ± 0.8b | 23.5 ± 1.1b | 13.2 ± 0.2 | 13.2 ± 0.1 | 13.4 ± 0.2 |
| Arachidic acid | C20:0 | 0.22 ± 0.01a | 0.19 ± 0.02b | 0.18 ± 0.01b | 0.88 ± 0.01b | 0.99 ± 0.02a | 0.98 ± 0.03a |
| Eicosenoic acid | C20:1 | 0.13 ± 0.01 | 0.13 ± 0.01 | 0.12 ± 0.00 | 0.36 ± 0.01b | 0.45 ± 0.04a | 0.46 ± 0.03a |
| Eicosadienoic acid | C20:2 | — | — | — | 0.10 ± 0.02 | 0.10 ± 0.02 | 0.12 ± 0.01 |
| Behenic acid | C22:0 | 0.20 ± 0.02 | 0.17 ± 0.02 | 0.17 ± 0.01 | 0.34 ± 0.03b | 0.43 ± 0.03a | 0.43 ± 0.01a |
| Lignoceric acid | C24:0 | 0.13 ± 0.01b | 0.16 ± 0.01a | 0.15 ± 0.01 ab | 0.17 ± 0.01b | 0.21 ± 0.01a | 0.2 ± 0.01a |
| ω-6/ω-3 | 0.79 | 1.97 | 1.93 | 4.57 | 4.53 | 4.46 | |
 | ||
| Fig. 1 Oleic acid, linoleic acid and α-linolenic acid expressed as relative percentages for (a) flax and (b) hemp samples extracted under cold conditions using hexane. ****, p-value < 0.0001. | ||
When comparing the two seed oils (Table 3), hemp oils exhibited a more complex profile, due to the presence of eicosadienoic acid and, in particular, γ-linoleic acid (GLA). This unsaturated fatty acid is known for its anti-inflammatory properties,37 and its presence resulted in an ω-6/ω-3 ratio ranging from 4.46 to 4.57 (after cold hexane extraction). These ratios are within the range that corresponds with the European Food and Safety Authority's recommendations for health maintenance and chronic disease prevention.38 Flaxseed samples showed a lower ω-6/ω-3 ratio (0.79 to 1.97), reflecting the high contribution of ALA. This lower ratio underlines a more balance lipid profile, as a reduced omega-6/omega-3 ratio has been associated with decrease inflammation. Oils rich in polyunsaturated fatty acids (PUFAs) have been shown to reduce the risk of cardiovascular diseases, as PUFAs play a key role in the reduction of cholesterol levels.39
Regarding the other acids identified in the samples (Table 3), the concentration of minor saturated (palmitic and myristic) and monounsaturated (oleic) fatty acids showed only minor variations among treatments, highlighting that these compounds were not significantly affected by mechanical conditions of pressing. Such behavior is consistent with previous observations reported by Kasote et al. (2013), who found that mechanical pressing altered polyunsaturated fatty acids such as α-linolenic acid, while saturated and monounsaturated fatty acids remained largely unchanged, with slight variations.40 In this sense the pressing process had a limited impact on these fatty acids, and optimized mechanical pressing can produce cakes with a desirable lipid profile comparable to that obtained using solvents.41
3.3. Total phenolic content and antioxidant activity
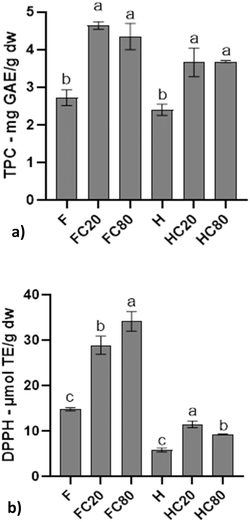
The results of total phenolic content (TPC) and antioxidant activity (AA) of the flax and hemp samples are reported in Fig. 2. During oil extraction, components such as tocopherols and phospholipids are removed, whereas phenolic compounds are concentrated in the residual material, resulting in an increased phenolic content in the oil cake.42 All cake samples exhibited increased total phenolic content and antioxidant capacity compared to whole seeds. In flax samples, TPC rose from 2.72 mg GAE g−1 (F) to 4.64 and 4.35 mg GAE g−1 in FC20 and FC80, respectively, corresponding to an enrichment of approximately 1.7 times. These amounts are consistent with those reported by Alu'datt et al. (2013), who found a phenolic content in defatted flaxseed of 1.20 mg GAE g−1.43 The results differ slightly from those obtained in other studies, but this difference may depend on variety and environmental factors, which play important roles in phenolic content.44 A similar trend was observed for hemp samples, where TPC increased from 2.40 mg GAE g−1 (H) to 3.67 mg GAE g−1 in both HC20 and HC80, and antioxidant activity rose from 5.86 µmol TE g−1 to 11.47 and 9.28 µmol TE g−1, for HC20 and HC80, respectively. The results for HC20 and HC80 were higher than those reported by Leonard et al. (2021b), who measured total phenolic contents ranging from 0.385 to 0.906 mg GAE g−1 using the FC assay.45 Extrusion speed could influence the results as the author used a very high velocity, thus leaving a greater quantity of lipid in the pressing cakes. All these findings corroborate previous observations that oil removal concentrates phenolic antioxidants in the defatted material.Focusing on DPPH radical scavenging activity (expressed as µmol Trolox equivalents per g of seed cake powder, Fig. 2), flaxseed cakes showed an increase, with values doubled compared with the starting amount: 14.8 µmol TE g−1 in F and 28.9 µmol TE g−1 and 34.2 µmol TE g−1, in FC20 and FC80, respectively. The slightly higher antioxidant activity observed in FC80 compared to FC20, may be related to the process-induced changes in the matrix structure. Since the QUENCHER method allows direct evaluation of the matrix as it is, these differences in the antioxidant activity between the two cakes may reflect changes in the structure of the matrix induced by the pressing process. A higher processing speed may enhance the accessibility of bound bioactive compounds, potentially due to increased mechanical stress on the matrix.
In contrast, hemp samples showed higher AA in HC20 than in HC80, suggesting that excessive mechanical stress or high-speed heat may partially degrade some phenolic compounds. This is in agreement with what is reported by Altan et al. (2009), who reported that antioxidant activity decreased in barley-grape pomace extrudates as screw speed increased.46 Furthermore, the opposite trends observed in DPPH antioxidant activity between flaxseed and hempseed cakes can be attributed to species-specific differences in seed composition and structural organization that influence the retention and accessibility of antioxidant compounds during pressing. Flaxseed is rich in lignans and phenolic acids mainly associated with the seed coat and mucilage, which may be better retained in the press cake at higher processing temperatures, whereas hempseed has a distinct phenolic profile whose stability may be favored under milder pressing conditions.47,48 These compositional and matrix-related differences likely affect oil release behavior and the distribution of antioxidant compounds in the resulting cakes, ultimately leading to the contrasting DPPH activity patterns observed between flaxseed and hempseed by-products. Overall, the high TPC and DPPH results for the press cakes suggested significant functional value.
3.4. Chromatographic characterization of free and bound polyphenols
A more detailed characterization study of the phenolic composition was carried out to distinguish the contribution of individual compounds. For this purpose, the phenolic profiles of flax and hemp samples were analyzed to determine their free and bound phenolic fractions, and the results are reported in Tables 4 and 5. The results highlighted many differences between seeds and their correspondent cakes. In the free phenolic fraction (Table 5), flaxseed cakes (FC20 and FC80) showed a significantly higher amount of total phenolic compounds, characterized by epicatechin, present in amounts of 171 and 166 µg g−1 for FC80 and FC20, respectively, values that increased by approximately 48% (FC20) and 73% (FC80) relative to the raw material. This molecule has health effects as it improves vascular function, reduces oxidative stress and has anti-inflammatory properties.49 Similarly, protocatechuic acid, ferulic acid, gallic acid and p-coumaric acid were present in higher quantities in the meals, with amounts around 16 µg g−1 for protocatechuic acid and 7 µg g−1 for ferulic acid, 4 µg g−1 for gallic acid and 9 µg g−1 for p-coumaric acid. These molecules have strong antiradical and anti-inflammatory activities,50 which contribute to increasing the value of these cakes for potential use in food. Their higher amounts in seedcakes suggest that phenols are more concentrated in the residual matrix, probably due to their hydrophilic nature and partial association with the fiber-protein matrix.| Flaxseed | Hempseed | |||||
|---|---|---|---|---|---|---|
| F | FC20 | FC80 | H | HC20 | HC80 | |
| a —: Not detected. | ||||||
| Gallic acid | 2.48 ± 0.09c | 4.47 ± 0.22a | 4.12 ± 0.24b | — | — | — |
| Protocatechuic acid | 6.76 ± 0.37b | 16.9 ± 1.5a | 15.4 ± 0.9a | 10.9 ± 0.7b | 16.1 ± 1.9a | 17.7 ± 1.1a |
| p-Hydroxybenzoic acid | 10.6 ± 0.7b | 8.8 ± 0.8c | 14.8 ± 1.0a | 48.8 ± 3.1b | 80.2 ± 6.5a | 79.8 ± 6.1a |
| Vanillic acid | — | — | — | 4.34 ± 0.28a | 4.28 ± 0.36a | 4.26 ± 0.54a |
| Caffeic acid | 3.09 ± 0.25b | 4.37 ± 0.33a | 4.61 ± 0.34a | 9.75 ± 0.69b | 12.8 ± 1.9a | 14.4 ± 1.1a |
| Chlorogenic acid | — | — | 25.7 ± 2.2b | 33.9 ± 3.6a | 37.9 ± 4.1a | |
| p-Coumaric acid | 6.00 ± 0.42b | 9.73 ± 0.49a | 9.32 ± 0.51a | 2.02 ± 0.17b | 3.17 ± 0.24a | 3.08 ± 0.28a |
| Ferulic acid | 3.93 ± 0.23c | 7.87 ± 0.84a | 6.80 ± 0.58b | 8.01 ± 0.80b | 10.1 ± 0.8a | 9.43 ± 0.70a |
| Ellagic acid | — | — | — | 44.1 ± 6.5b | 72.3 ± 6.4a | 67.6 ± 2.9a |
| Epicatechin | 98.5 ± 4.8b | 166 ± 12a | 171 ± 13a | — | — | — |
| Myricetin | — | — | — | 62.2 ± 6.7b | 91.8 ± 8.5a | 84.3 ± 6.3a |
| Quercetin | — | — | — | 237 ± 37b | 287 ± 29a | 280 ± 19a |
Furthermore, the slightly lower yields observed for some compounds, such as gallic and ferulic acid, in samples subjected to high-speed screw pressing (FC80) may be explained by two factors. First, higher pressing speeds can lead to increased residual oil within the cake matrix, effectively diluting phenolic concentrations. Second, the intensified mechanical and associated thermal stress at high rotational speeds can enhance the degradation of thermolabile phenolics. Polyphenols, including phenolic acids, are well documented to undergo heat- and oxidation-induced decomposition during processing, with elevated temperature and combined stress conditions accelerating oxidative breakdown and structural cleavage, thereby decreasing the extractable content of specific compounds under severe conditions. The thermal instability of polyphenols under heat and processing stress has been observed across a range of matrices and treatments, where higher temperatures promote degradation reactions and consequent loss of individual phenolic constituents.51,52
Hemp seed cakes (HC20 and HC80) showed a broader and more complex phenolic profile than flax, despite the absence of gallic acid and epicatechin.
Compounds such as chlorogenic acid, ellagic acid, quercetin, and myricetin were particularly concentrated in the hempseed cakes, distinguishing their phenolic fingerprint from that of flaxseed and highlighting the species-specific nature of the phenolic composition. For example, chlorogenic acid increased from 25.7 µg g−1 in H to 33.9 and 37.9 µg g−1 in HC20 and HC80, respectively, while quercetin, the main compound present, increased from 237 µg g−1 in H to approximately 280 µg g−1 in HC20 and HC80. Overall, the total free phenolic content was quantitatively higher in hempseed cakes than in flaxseed cakes. This result is in contrast with the trends observed using the FC assay. Such differences may be attributed to the fact that flaxseed is a rich source of lignans,53 which are mainly located in the seed hull and are known to react with the FC reagent.
The bound phenolic fractions (Table 6) followed a similar trend to that of the free fraction, with oilseed cakes generally showing elevated phenolic concentrations compared to whole seeds. However, greater variability was observed for some compounds, likely reflecting matrix heterogeneity and the multistep extraction procedure required for bound phenolics.
| Flaxseed | Hempseed | |||||
|---|---|---|---|---|---|---|
| F | FC20 | FC80 | H | HC20 | HC80 | |
| a —: Not detected. | ||||||
| Gallic acid | 4.30 ± 0.33c | 12.0 ± 1.0a | 10.9 ± 0.8b | — | — | — |
| Protocatechuic acid | 6.29 ± 0.60b | 8.14 ± 0.98a | 8.03 ± 0.78a | — | — | — |
| p-Hydroxybenzoic acid | 27.6 ± 0.5b | 31.2 ± 1.3a | 31.6 ± 3.4a | 14.5 ± 0.5c | 23.2 ± 3.3a | 18.4 ± 2.2b |
| Vanillic acid | 3.15 ± 0.87a | 3.52 ± 0.75a | 3.39 ± 0.44a | 11.3 ± 1.6a | 14.1 ± 3.4a | 12.3 ± 2.1a |
| Syringic acid | — | — | — | 3.24 ± 0.28b | 4.56 ± 0.51a | 4.37 ± 0.35a |
| p-Coumaric acid | 28.7 ± 1.4b | 29.3 ± 4.4 ab | 33.2 ± 4.3a | 99.9 ± 5.5b | 133 ± 14a | 132 ± 15a |
| Ferulic acid | 164±6a | 181 ± 28a | 174 ± 54a | 14.2 ± 1.9a | 11.8 ± 2.1a | 14.4 ± 2.2a |
| Myricetin | — | — | — | 124±9 ab | 141 ± 19a | 107 ± 23b |
| Quercetin | — | — | — | 14.7 ± 1.8a | 15.5 ± 2.4a | 10.3 ± 1.0b |
In flaxseed cake, ferulic acid dominated the bound fraction, with levels ranging from 164 µg g−1 in F to 181 µg g−1 in FC20 and 174 µg g−1 in FC80, with no statistical differences between the three samples. This suggests that most ferulates remain bound to cell wall lignins despite pressing.54 Retained bound phenols, such as ferulic acid in flax, likely contribute to health benefits, as they may be released during digestion. Protocatechuic and p-hydroxybenzoic acids increased in flaxseed cakes, with no statistical differences between the two by-products obtained, while gallic acid increased to a greater extent in FC20 than in FC80. In hempseed samples, the bound phenolic profile was more variable. Among phenolic acids, coumaric acid emerged as the dominant compound, particularly in the cakes, with concentrations increasing from 99.9 µg g−1 in H to 133 µg g−1 in HC20 and 132 µg g−1 in HC80. Myricetin and quercetin, absent in flax samples, reached over 141 µg g−1 in HC20 for myricetin and 15.5 µg g−1 in HC20 for quercetin. In both cases, however, at high speed (HC80), the amount of these molecules decreased (e.g. myricetin decreased from 141 µg g−1 in HC20 to 107 µg g−1 in HC80), suggesting a potential sensitivity of these bound compounds to processing intensity. The discrepancy between the higher TPC values obtained for flaxseed cakes using the Folin–Ciocalteu assay and the higher total free phenolic content quantified by HPLC in hempseed cakes can be explained by methodological differences. However, the limitations of the Folin–Ciocalteu assay must be acknowledged, particularly its lack of specificity, as the reagent responds to the overall reducing capacity of the sample and can react with non-phenolic compounds such as amino acids and other reducing substances present in complex matrices.55 In contrast, HPLC provides a more selective quantification of free phenolic compounds, explaining the observed differences.
Overall, these findings indicate that moderate pressing (20%) may enhance the analytical accessibility and quantification of bound phenolic compounds by promoting matrix disruption, while higher pressing speed could affect structurally sensitive components of the seed matrix.
5. Conclusions
This study showed that flaxseed and hempseed press cakes, residual by-products from oil extraction, have significant nutritional and functional value. They are rich in protein and fiber, but being low lipid content, they present a reduced energy value compared to the raw seeds. The residual lipid fractions retained favorable fatty acid profiles, with flaxseed cakes being richer of α-linolenic acid (ALA) than hempseed cakes, which instead have a higher content of linoleic acid (LA). In addition, the analysis of fatty acid composition demonstrated that the pressing process can alter the lipid profile of the oilseeds. In particular, for flaxseed oil, a significant reduction in α-linolenic acid (ALA) in comparison to the starting material was observed, suggesting that processing conditions such as temperature are important. The antioxidant activity and the phenolic content were significantly higher in the oilseed cakes compared to whole seeds, confirming that oil removal enhances their concentration after pressing. The HPLC profiling revealed species-specific polyphenolic fingerprints, with flax samples rich in epicatechin and ferulic acid, and hemp samples rich in quercetin. The pressing speed had only a modest effect on nutrient and phenolic retention, although excessive speed could slightly reduce labile compounds. Both free and bound phenolic fractions showed an improvement post-extraction, with moderate pressing conditions favoring their preservation. In conclusion, the functional benefits of flax and hempseed cakes are highlighted by the overall preservation of phenols and antioxidant activity, together with the high content of dietary fiber and proteins. Therefore, these by-products are promising sustainable, nutrient-rich substances that can be used to enhance the nutritional and antioxidant profile of food formulations, especially bakery products, in line with circular economy principles.Author contributions
Francesca Carrà: formal analysis, investigation, writing – original draft. Vincenzo Disca: formal analysis, investigation, writing – original draft, visualization. Antonio Colasanto: formal analysis, investigation. Marco Arlorio: writing review – editing, funding acquisition. Matteo Bordiga: writing review – editing. Jean Daniel Coïsson: conceptualization, writing review – editing, funding acquisition. Monica Locatelli: writing review – editing, data curation, supervision. Fabiano Travaglia: methodology, project administration, writing review & editing. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare that there are no conflicts to declare.Data availability
All data are available with in the article.Acknowledgements
Dr Carrà holds a PhD career grant supported by Next Generation EU – MUR (CUP C13C22000490008) and currently holds a post doc position financed by the Agrihealth project, Interreg IT-CH. This publication is part of the project NODES which has received funding from the MUR-M4C2 1.5 of PNRR, funded by the European Union-NextGenerationEU (Grant agreement no. ECS00000036). The research was partially funded by the project “Food Digital Monitoring – FDM” – Accordo di programma MIUR – Regione Piemonte, Azione 3 “Fabbrica Intelligente”, Bando: Piattaforma Tecnologica “Fabbrica Intelligente”.References
- M. Rao, A. Bast and A. de Boer, Sustainability, 2021, 13, 4428 CrossRef.
- B. Talwar, R. Chopra, N. K. Taneja, M. Chand, S. Homroy, A. Dhiman, P. K. Singh and S. Chaudhary, Food Prod., Process. Nutr., 2025, 7, 22 CrossRef.
- S. Rakita, B. Kokić, M. Manoni, S. Mazzoleni, P. Lin, A. Luciano, M. Ottoboni, F. Cheli and L. Pinotti, Foods, 2023, 12, 432 CrossRef CAS PubMed.
- A. Mannucci, A. Castagna, M. Santin, A. Serra, M. Mele and A. Ranieri, LWT, 2019, 104, 84–90 CrossRef CAS.
- N. Grahovac, M. Aleksić, B. Trajkovska, A. Marjanović Jeromela and G. Nakov, Foods, 2025, 14, 2244 CrossRef CAS PubMed.
- J. Yang, C. Wen, Y. Duan, Q. Deng, D. Peng, H. Zhang and H. Ma, Trends Food Sci. Technol., 2021, 118, 252–260 CrossRef CAS.
- K. Ambroziak and A. Wenda-Piesik, Foods, 2025, 14, 1605 CrossRef CAS PubMed.
- T. Capcanari, E. Covaliov, C. Negoița, R. Siminiuc, A. Chirsanova, V. Reşitca and D. Țurcanu, Foods, 2023, 12, 4327 CrossRef CAS PubMed.
- M. Pojić, A. Mišan, M. Sakač, T. Dapčević Hadnađev, B. Šarić, I. Milovanović and M. Hadnađev, J. Agric. Food Chem., 2014, 62, 12436–12442 CrossRef PubMed.
- X. Feng, G. Sun and Z. Fang, Foods, 2022, 11, 211 CrossRef CAS PubMed.
- C. Sanmartin, I. Taglieri, F. Venturi, M. Macaluso, A. Zinnai, S. Tavarini, A. Botto, A. Serra, G. Conte, G. Flamini and L. G. Angelini, Foods, 2020, 9, 204 CrossRef CAS PubMed.
- S.-S. Teh, A. E.-D. Bekhit and J. Birch, Antioxidants, 2014, 3, 67–80 CrossRef PubMed.
- A. Allay, A. B. Moumen, Y. Rbah, M.-L. Fauconnier, J. Nkengurutse, H. S. Caid, A. Elamrani and F. Mansouri, J. Cannabis Res., 2025, 7, 37 CrossRef PubMed.
- Z. Piravi-Vanak, A. Dadazadeh, S. Azadmard-Damirchi, M. Torbati and F. Martinez, Foods, 2024, 13, 1472 CrossRef CAS PubMed.
- V. Y. Ixtaina, M. L. Martínez, V. Spotorno, C. M. Mateo, D. M. Maestri, B. W. K. Diehl, S. M. Nolasco and M. C. Tomás, J. Food Compos. Anal., 2011, 24, 166–174 CrossRef CAS.
- G. Abdilova, Z. Sergibayeva, D. Orynbekov, M. Shamenov, G. Zhumadilova, A. Bakiyeva, N. Mukashev, A. Bayadilova and D. Dukenbayev, Appl. Sci., 2023, 13, 9958 CrossRef CAS.
- D. Gumul, M. Kruczek, K. Szary-Sworst, R. Sabat and A. Wywrocka-Gurgul, Processes, 2025, 13, 1875 CrossRef CAS.
- M. Locatelli, J. D. Coïsson, F. Travaglia, E. Cereti, C. Garino, M. D'Andrea, A. Martelli and M. Arlorio, Food Chem., 2011, 129, 1865–1873 CrossRef CAS.
- M. Locatelli, F. Travaglia, J. D. Coïsson, A. Martelli, C. Stévigny and M. Arlorio, Food Chem., 2010, 119, 1647–1655 CrossRef CAS.
- A. Colasanto, V. Disca, F. Travaglia, M. Bordiga, J. D. Coïsson, M. Arlorio and M. Locatelli, Food Chem., 2025, 472, 142889 CrossRef CAS PubMed.
- V. A. Papillo, M. Locatelli, F. Travaglia, M. Bordiga, C. Garino, J. D. Coïsson and M. Arlorio, Food Res. Int., 2019, 115, 511–518 CrossRef CAS PubMed.
- R Core Team, R: A Language and Environment for Statistical Computing, Computer software, R foundation for Statistical Computing, Vienna, Austria, 2024, https://www.R-project.org/ Search PubMed.
- I. Jakab, M. Mardani, J. Tormási, L. Abrankó and K. Badak-Kerti, Eur. J. Lipid Sci. Technol., 2025, 127, e70017 CrossRef CAS.
- C. F. Lewandoski, R. F. Santos, D. Bassegio, S. S. N. M. de, J. A. C. Siqueira, S. D. M. de, L. S. Reis and P. L. Bueno, Aust. J. Crop Sci., 2021, 15, 594–601 CAS.
- D. M. Ferreira, M. A. Nunes, L. E. Santo, S. Machado, A. S. G. Costa, M. Álvarez-Ortí, J. E. Pardo, M. B. P. P. Oliveira and R. C. Alves, Molecules, 2023, 28, 723 CrossRef CAS PubMed.
- M. Ionescu, G. Voicu, S.-S. Biris, M. Dincă and N. Ungureanu, Parameters Influencing the Screw Pressing Process of Oilseed Materials, 2014 Search PubMed.
- K. P. Luna-Castillo, X. C. Olivares-Ochoa, R. G. Hernández-Ruiz, I. M. Llamas-Covarrubias, S. C. Rodríguez-Reyes, A. Betancourt-Núñez, B. Vizmanos, E. Martínez-López, J. F. Muñoz-Valle, F. Márquez-Sandoval and A. López-Quintero, Nutrients, 2022, 14, 1104 CrossRef CAS PubMed.
- J. Xu, M. Bai, H. Song, L. Yang, D. Zhu and H. Liu, Plant Foods Hum. Nutr., 2022, 77, 504–513 CrossRef CAS PubMed.
- V. Disca, F. Carrà, M. Stampini, M. Arlorio and M. Locatelli, Food Biosci., 2025, 72, 107435 CrossRef CAS.
- A. Mazzocchi, V. De Cosmi, P. Risé, G. P. Milani, S. Turolo, M.-L. Syrén, A. Sala and C. Agostoni, Front. Physiol., 2021, 12 DOI:10.3389/fphys.2021.659551.
- L. Montero, D. Ballesteros-Vivas, A. F. Gonzalez-Barrios and A. del P. Sánchez-Camargo, Front. Nutr., 2022, 9 DOI:10.3389/fnut.2022.1039180.
- M. M. Özcan and N. Uslu, J. Food Process. Preserv., 2022, 46, e17091 Search PubMed.
- Z. Zhen-Shan, L. Dong and Z. Li-Xia, Asian J. Chem., 2013, 25, 10082–10086 CrossRef.
- G. Pedron, Y. Jaouhari, M. Bordiga and M. Locatelli, J. Raw Mater. Process. Food., 2025, 6, 1–13 Search PubMed.
- V. Disca, F. Carrà, Y. Jaouhari, M. Stampini, I. C. Martakos, I. Tzavellas, N. S. Thomaidis, L. Cecchi, N. Mulinacci, P. Ferreira-Santos, M. Locatelli, M. Arlorio and F. Travaglia, Int. J. Food Sci. Technol., 2025, 60, vvaf169 CrossRef CAS.
- C. Da Porto, D. Decorti and F. Tubaro, Ind. Crops Prod., 2012, 36, 401–404 CrossRef CAS.
- Ö. F. Kaçar, T. Kose and H. Kaya Kaçar, Front. Nutr., 2025, 12 DOI:10.3389/fnut.2025.1669375.
- J. C. Callaway, Euphytica, 2004, 140, 65–72 CrossRef.
- E. Tortosa-Caparrós, D. Navas-Carrillo, F. Marín and E. Orenes-Piñero, Crit. Rev. Food Sci. Nutr., 2017, 57, 3421–3429 CrossRef PubMed.
- D. M. Kasote, Y. S. Badhe and M. V. Hegde, Ind. Crops Prod., 2013, 42, 10–13 CrossRef CAS.
- N. Aybar Yalınkılıç, Ş. Çiçek Bayram and S. Başbağ, Life, 2025, 15, 483 CrossRef PubMed.
- J. R. Sarkis, A. P. F. Côrrea, I. Michel, A. Brandeli, I. C. Tessaro and L. D. F. Marczak, J. Am. Oil Chem.' Soc., 2014, 91, 1773–1782 CrossRef CAS.
- M. H. Alu’datt, T. Rababah, K. Ereifej and I. Alli, Food Chem., 2013, 139, 93–99 CrossRef PubMed.
- Q. Deng, X. Yu, F. Ma, J. Xu, F. Huang, Q. Huang and F. Sheng, Int. J. Food Prop., 2017, 20, S2708–S2722 CrossRef CAS.
- W. Leonard, P. Zhang, D. Ying, Y. Xiong and Z. Fang, J. Food Sci., 2021, 86, 3159–3175 CrossRef CAS PubMed.
- A. Altan, K. L. McCarthy and M. Maskan, Int. J. Food Sci. Technol., 2009, 44, 1263–1271 CrossRef CAS.
- W. Leonard, P. Zhang, D. Ying, Y. Xiong and Z. Fang, J. Food Sci., 2021, 86, 3159–3175 CrossRef CAS PubMed.
- P. Sangiorgio, S. Errico, A. Verardi, S. Moliterni, G. Tamasi, C. Rossi and R. Balducchi, Nutraceuticals, 2023, 3, 58–74 CrossRef.
- I. Bernatova, Biotechnol. Adv., 2018, 36, 666–681 CrossRef CAS PubMed.
- S. Kiokias, C. Proestos and V. Oreopoulou, Foods, 2020, 9, 534 CrossRef CAS PubMed.
- A. Antony and M. Farid, Appl. Sci., 2022, 12(4), 2107 CrossRef CAS.
- W. M. Zhang Haolin, W. Minglong and X. Jianbo, in Advances in Food and Nutrition Research, Academic Press, 2022, vol. 102, pp. 1–45 Search PubMed.
- S. F. De Silva and J. Alcorn, Pharmaceuticals, 2019, 12, 68 CrossRef CAS PubMed.
- D. Rabiej-Kozioł and A. Szydłowska-Czerniak, Foods, 2025, 14, 1444 CrossRef PubMed.
- R. Amorati and L. Valgimigli, Free Radical Res., 2015, 49, 633–649 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |