Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Influence of light spectrum and photoperiod on early seedling development and microgreen yield in cabbage (Brassica oleracea var. capitata) and arugula (Eruca sativa)
Christos Vatistas *,
Dafni Despoina Avgoustaki and
Thomas Bartzanas
*,
Dafni Despoina Avgoustaki and
Thomas Bartzanas
Department of Natural Resources and Agricultural Engineering, Agricultural University of Athens, Iera Odos 75, Athens 118 55, Greece. E-mail: vatistasx@aua.gr
First published on 24th April 2026
Abstract
Controlled Environment Agriculture (CEA) and vertical farming (VF) enable urban food production through artificial lighting and soilless cultivation, reducing transport distances and supporting year-round supply. However, sustainability is strongly affected by the energy demand of LED lighting, making it critical to improve yield per unit of supplied light and energy. Microgreens are well suited to VF systems because they are harvested at an early developmental stage, typically 7 to 21 days after sowing, allowing short production cycles. In this study, nine LED spectra (UV, blue, green, orange, red, far-red, blue-red, blue-red-far-red, and cool-white) were tested under 4, 8, and 16 h photoperiods at constant light intensity, resulting in different daily light integral (DLI) levels, to evaluate their effects on seedling emergence, early development, biomass accumulation, and final microgreen yield in cabbage and arugula. Higher emergence values for cabbage were observed under 16 h blue light, while arugula showed the highest emergence under 8 h green light. In both species, 16 h blue-red-far-red treatment was associated with more compact seedlings and higher dry to fresh biomass ratios. In a second phase, emergence enhancing spectra were combined with the biomass enhancing blue-red-far-red treatment to evaluate microgreen production on soil. This two-stage lighting strategy yielded the highest fresh biomass in both species, 2.189 g for cabbage and 12.56 g for arugula. These findings indicate that tailoring the early light environment through species-specific emergence-enhancing spectra can improve microgreen uniformity and yield in vertical farming systems, while simultaneously enhancing energy-use efficiency and light-use efficiency.
Sustainability spotlightEnergy demand from artificial lighting remains a key challenge for the sustainability of urban vertical farming. This study demonstrates that appropriate lighting during the early cultivation stage can increase the number of successfully established seedlings, while targeted lighting during subsequent growth can accelerate development and increase microgreens biomass production in cabbage and arugula. By combining an emergence-enhancing light treatment with a biomass-enhancing spectrum in a stage-specific strategy, higher yields were achieved compared with conventional production practices. These productivity gains were associated with improved light-use and energy-use efficiency, highlighting the potential of targeted lighting management to support more sustainable and resource-efficient urban food production. |
1. Introduction
The development of agriculture has enabled stable food production and supported population growth. Controlled-Environment Agriculture (CEA) systems, including traditional greenhouses and more recent vertical farming (VF) systems, enable crop production independent of external climate conditions. These systems support year-round cultivation and expand agricultural production to regions with unfavorable environments.1,2The ease of mass production has led to inefficient management of production volumes, resulting in the overexploitation of natural resources, increased energy consumption, and the accumulation of waste. These effects contribute to ecosystem degradation, greenhouse gas emissions, and water and soil pollution.3 According to the Food and Agriculture Organization (FAO), 14% of food produced by agriculture is lost between harvest and before reaching consumers, mainly due to inefficiencies in harvesting, transportation, storage and processing.4 A promising approach to addressing these challenges and supporting urban food production is vertical farming (VF).5 This method employs hydroponic systems and vertically stacked cultivation units, allowing efficient use of space. The use of artificial lighting enables precise control of the Daily Light Integral (DLI) which can enhance biomass production and shorten growth cycles compared with open-field or greenhouse systems.6 In addition, automation and environmental control systems allow stable cultivation conditions and improved resource use efficiency.7
Microgreens constitute a category of crops with high commercial and research interest in VF systems. They are tender seedlings grown mainly from vegetable seeds and harvested once the cotyledons are fully developed, typically before the first true leaves appear. Their rising popularity is due to several factors that make them attractive for production.8 These include their suitability for cultivation under artificial lighting, their adaptability to urban environments, their year-round production potential, and their use for both personal consumption and educational purposes.9 Additional advantages include their high nutritional value, intense flavor, and wide diversity in color, taste, and morphology, which contribute to their increasing demand and added value.10 Microgreen cultivation requires relative low inputs, such as water, substrate, and space, while minimizing waste by producing mainly edible biomass. Furthermore, local production close to consumers, reduces post-harvest losses and transportation-related emissions, supporting more sustainable food systems.8,11
In microgreens production, early-stage growth is particularly critical due to the very short cultivation cycle. Uniform and rapid seedling emergence is essential for achieving homogeneous canopies and synchronized harvest, while hypocotyl elongation directly affects product quality and marketability. In addition, maximizing biomass production within a limited time frame is a key objective, closely linked to light use efficiency and overall energy consumption in controlled-environment systems. Therefore, optimizing light conditions during the early developmental stages represents an important challenge for improving both productivity and resource efficiency in microgreens cultivation.9,11
In this context, lighting management becomes a critical factor in microgreens production systems, particularly in vertical farming environments. One of the main challenges for the sustainability, profitability and viability of VFs is the intense energy demand required for lighting operation. Although the use of LED lamps significantly reduces energy consumption, lighting costs remain high and can reach up to 80% of the total production cost in a VF.12 Research on the optimal light intensity and spectrum for each crop, the optimal photoperiod, and improving LEDs efficiency are key areas for reducing costs, increasing production, and enhancing the sustainability of these cultivation systems.13
Plants utilize light in the 400–700 nm range, known as Photosynthetically Active Radiation (PAR), to support growth and development.14 Light perception is mediated by photoreceptors, which regulate molecular and physiological processes throughout the plant life cycle.15 In plants, major photoreceptor groups include phytochromes (detect light at red/far-red parts of spectrum), cryptochromes (detect light at blue/UV-A parts of spectrum), phototropins (blue light receptors associated with stomatal opening), UV-B receptors (UVR8) (detect light in the UV-B range) and ZTL-like proteins, which detect light in the blue range of the spectrum and are associated with the regulation of the circadian clock.16 In seeds, phytochromes play a key role in regulating germination by controlling hormonal balance, promoting gibberellin (GA) synthesis and reducing abscisic acid (ABA) levels under favorable light conditions.15,17 Photoreceptor activity influences signaling pathways involved in photomorphogenesis and seed germination, although responses vary among species and wavelengths.11,14,18–20 While some species exhibit light-sensitive germination, many cultivated vegetables germinate equally well in darkness, with light primarily affecting seedling emergence and early post-germinative development. Seed size has also been linked to light sensitivity, as smaller seeds, with limited nutrient reserves, tend to respond more strongly to light cues that facilitate emergence and the transition to photosynthetic growth.21,22
Although previous studies have investigated the effects of light spectrum and photoperiod on plant growth, limited information is available on how early-stage light conditions affect seedling emergence and subsequent microgreen yield, particularly in controlled-environment systems. In addition, the potential benefits of applying different lighting conditions during successive developmental stages remain insufficiently explored. Understanding seedling responses not only to blue and red wavelengths, which are strongly associated with photomorphogenic regulation and photosynthetic activity, but also to other spectral regions such as green, orange, UV, and far-red radiation, is of particular interest. While green and orange wavelengths fall within the PAR range, UV and far-red lie outside it and are known to influence plant development through distinct photoreceptor-mediated pathways.15–17
The aim of this study was to evaluate the effects of different light spectra and photoperiods, applied under constant light intensity (different DLI), on seedling emergence, early seedling development, and subsequent microgreen production of cabbage and arugula under controlled-environment conditions. Faster and more uniform emergence can shorten the overall production cycle and improve cultivation process efficiency, while early-stage light conditions strongly influence seedling morphology and subsequent biomass accumulation. In this context, the second phase of the study focused on identifying the light spectrum that maximizes biomass production and on evaluating whether a two-stage lighting strategy, in which the emergence-enhancing spectrum is applied during the first days after sowing and the biomass-enhancing spectrum is used during subsequent growth, can enhance final microgreen yield while enabling a more efficient use of supplied light and energy. Based on current knowledge of plant responses to light, it was expected that both spectral quality and daily light exposure would influence seedling emergence and early development, and that combining different lighting conditions across developmental stages could provide advantages over the use of a single lighting regime.
2. Material and methods
2.1. Seedling growth
The selected leafy greens studied were cabbage (Brassica oleracea var.capitata) and arugula (Eruca sativa), both cultivated in two identical growth chambers with controlled environmental conditions. The chambers used were the CL 600 model from ELVEM. The external dimensions of the chamber are height × width × depth (H × W × D): 210 × 72 × 90 cm, and the internal dimensions are H × W × D: 155 × 60 × 67 cm. Temperature inside the chambers ranged from 19–21 °C during the day and 16–18 °C at night. Twenty seeds of each species were placed in small square plastic containers (12 × 12 × 5 cm) containing water-saturated absorbent paper as a substrate. The relative humidity (RH%) in the growth area fluctuated between 80% and 90% and was maintained by covering the containers with transparent film. These two species were selected because they are widely used as microgreens, and previous experiments have shown that they exhibit enhanced seedling emergence under light conditions.11 The experiments were conducted at the Laboratory of Farm Structures of the Agricultural University of Athens, Greece, from October 2023 to April 2024.2.2. Lighting treatments
An initial sowing was performed under continuous darkness to assess its effect on seedling emergence. Experiments under light conditions were conducted using 4 h, 8 h, and 16 h photoperiods across nine different light spectra. For each photoperiod, a total of 180 seeds were tested, distributed across the nine spectra. Each spectrum × photoperiod combination was applied to one container containing 20 seeds, and the container was considered the experimental unit. A 16 h photoperiod was selected because it is commonly used to promote the growth of microgreens and other leafy plants in vertical farming systems.23,24 Shorter photoperiods (8 h and 4 h) were included to examine the effect of progressively reduced daily light exposure under a constant PPFD, resulting in corresponding differences in daily light integral (DLI) among treatments.Artificial lighting was provided using LED light sources emitting relatively narrow-band radiation centered on specific wavelengths. Spectral measurements were conducted using a spectroradiometer (UPRTEK PG200N, measurement range 350–800 nm), which was used to determine the peak wavelength and approximate emission range of each LED spectrum. Although LED sources were selected to target specific spectral regions, measured emission profiles indicated partial overlap between adjacent wavelength bands; however, each treatment was characterized by a dominant peak within the intended spectral region. The spectra studied were: (1) UV-A (measured emission ∼350–400 nm, peak 364 nm), (2) blue (∼410–500 nm, peak 448 nm), (3) green (∼470–570 nm, peak 516 nm), (4) orange (∼550–640 nm, peak 594 nm), (5) red (∼580–670 nm, peak 630 nm), (6) far-red (∼650–770 nm, peak 729 nm), (7) a combination of blue and red (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), (8) a combination of blue-red-far-red (1
1), (8) a combination of blue-red-far-red (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), and (9) cool-white LED (∼410–750 nm, peak 448 nm) (Table 1). Mixed spectra were generated using the corresponding monochromatic LED sources described above. The photosynthetic photon flux density (PPFD) was set to 30 µmol m−2 s−1 for all PAR spectra (400–700 nm). For the UV and far-red treatments, which fall outside the PAR range, intensity was measured as photon flux density (PFD). The UV spectrum had a PFD value of 0.29 µmol m−2 s−1, while the far-red treatment was applied at a photon flux level comparable to the PPFD used for PAR spectra. Each treatment lasted for 7 days (168 h).
1), and (9) cool-white LED (∼410–750 nm, peak 448 nm) (Table 1). Mixed spectra were generated using the corresponding monochromatic LED sources described above. The photosynthetic photon flux density (PPFD) was set to 30 µmol m−2 s−1 for all PAR spectra (400–700 nm). For the UV and far-red treatments, which fall outside the PAR range, intensity was measured as photon flux density (PFD). The UV spectrum had a PFD value of 0.29 µmol m−2 s−1, while the far-red treatment was applied at a photon flux level comparable to the PPFD used for PAR spectra. Each treatment lasted for 7 days (168 h).
| Photoperiod | Spectra | DLIa (mol per m2 per day) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| UV | Blue | Green | Orange | Red | Far-red | Blue-red | Blue-red-farred | Cool-white | ||
a DLI values refer to the daily light integral corresponding to a PPFD of 30 µmol m−2 s−1 across the three photoperiods applied. In the case of UV lighting, DLI values were significantly lower due to the very low PFD, measuring 0.0042, 0.0084, and 0.0167 mol per m2 per day for the 4-, 8-, and 16 h photoperiods, respectively. All PAR spectra were applied at a constant PPFD of 30 µmol m−2 s−1, while UV and far-red treatments were expressed as PFD. Mixed spectra were applied at equal photon ratios (blue![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) red = 1 red = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and blue 1 and blue![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) red red![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) far-red = 1 far-red = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). 1). |
||||||||||
| 4 hours | 4hUV | 4hB | 4hG | 4hOr | 4hR | 4hFR | 4hBR | 4hBRFR | 4hCW | 0.432 |
| 8 hours | 8hUV | 8hB | 8hG | 8hOr | 8hR | 8hFR | 8hBR | 8hBRFR | 8hCW | 0.864 |
| 16 hours | 16hUV | 16hB | 16hG | 16hOr | 16hR | 16hFR | 16hBR | 16hBRFR | 16hCW | 1.728 |
2.3. Microgreens production
The second phase of the experiment involved the production of microgreens from the two studied species, using four different cultivation strategies. Microgreens were grown on a soil substrate composed of peat, plant residues, and organic fertilizer, placed in plastic containers with holes at the bottom to allow drainage, which were positioned above a second container to collect excess irrigation water. For each species, 0.85 g of seeds were sown per container, and each cultivation method was replicated four times.For the first method (M1), seeds were kept in darkness for 3 days and then transferred to artificial lighting with a blue-red-far-red (BRFR) spectrum under a 16 h photoperiod, a regime commonly used for microgreens cultivation.25,26 In the second method (M2), seeds were placed under the same BRFR spectrum with a 16 h photoperiod from the first day of cultivation. This spectrum was selected because, in the first phase of the experiment, it produced seedlings with the highest dry-to-fresh biomass ratio in both species. In the third method (M3), cabbage seeds were exposed to blue light under a 16 h photoperiod for the first five days. Arugula seeds received green light under an 8 h photoperiod for the first five days. After this initial stage, both species were transferred to BRFR lighting under a 16 h photoperiod for the remainder of the cultivation period. These initial lighting conditions were selected because they produced the highest seedling emergence and the fastest cotyledon-stage development during the first experimental phase. In the fourth method (M4), seeds were exposed to the same initial spectrum used in M3, but only for the first day. On the following day, seedlings were transferred to BRFR lighting under a 16 h photoperiod for the remainder of the cultivation period. This treatment was included to examine whether a brief, 1 day exposure to the emergence-enhancing spectrum could provide an early light cue that supports the initiation of germination and subsequent seedling emergence. The light intensity applied during the initial lighting stage (prior to exposure to the growth spectrum) was 30 µmol m−2 s−1 (M3 and M4). The growth-spectrum intensity was set to 120 µmol m−2 s−1, and the total cultivation period lasted 12 days.
2.4. Measurements
The initial measurements included recordings every 24 h of the number of seeds that developed both a root and a shoot (seedling emergence). The emergence percentage was calculated using the following formula:
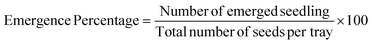 | (1) |
At the end of each lighting treatment, the developed seedlings were placed on graph millimeter paper to measure shoot length. Measurements were taken in millimeters (mm) and included hypocotyl and cotyledon length, as well as cotyledon area, which was quantified in square millimeters (mm2). Morphological measurements were obtained from the seedlings that emerged within each container. Seedlings within the same container were treated as subsamples rather than independent biological replicates, and sample size varied among treatments according to emergence percentage.
Fresh and dry biomass of the developed shoots and cotyledons were measured for each lighting treatment. Biomass values presented in the results correspond to the seedlings that successfully emerged within each container. Seedlings were weighed using the aeAdam LTB 3602i precision scale, which has a measurement error of ±30 mg. Because some of the samples had masses close to the instrument's error margin, each sample was weighed five times to reduce random measurement variability. The mean of these repeated measurements was used as the final value for each sample. The drying of the fresh biomass was carried out in a drying oven at 80 °C for 24 h. Then the value of the dry to fresh biomass was divided to calculate the DW![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) FW biomass ratio. The same procedure was followed during the second experimental phase involving microgreens production. The mean value was calculated from the biomass measurements of the four containers per cultivation method. This mean value represents the fresh and dry biomass of the above-ground parts (shoots and cotyledons).
FW biomass ratio. The same procedure was followed during the second experimental phase involving microgreens production. The mean value was calculated from the biomass measurements of the four containers per cultivation method. This mean value represents the fresh and dry biomass of the above-ground parts (shoots and cotyledons).
2.5. Light and energy use efficiency
Light use efficiency (LUE) and lighting energy use efficiency (EUE) were calculated for the four microgreens cultivation methods applied to cabbage and arugula. LUE (g DW/mol) was calculated as the ratio between the shoot dry weight produced per unit cultivation area (g DW per m2) and the cumulative light integral (mol m−2). The cumulative light integral was obtained by multiplying the DLI values by the number of days during which lighting was applied. EUE (g FW per kW h) was calculated as the ratio between the fresh shoot biomass (g FW) and the total energy consumption of the lighting system (kW h) over the entire cultivation period.These calculations were conducted solely to facilitate a direct comparison among the four cultivation methods assessed in this study. In practical large-scale microgreens production, overall efficiency depends on multiple factors – including light intensity, lamp efficiency, light mounting height, cultivation area, and seeding density – which may lead to substantially different EUE and LUE values. Therefore, the values reported here should be interpreted as method-to-method comparative indicators rather than absolute measures of production efficiency.
2.6. Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics 25. For the seedling-stage experiment, the number of the emerged seedlings differed among treatments. Due to the experimental design and variability in emergence, results from this phase were evaluated descriptively based on mean values and standard deviation, and no inferential statistical comparisons were performed. For the microgreens production experiment, one-way ANOVA followed by Tukey's Honestly Significant Difference (HSD) test (P < 0.05) was performed on the fresh and dry biomass, as well as on the calculated light use efficiency LUE and energy use efficiency (EUE), to assess differences among the four cultivation methods (n = 4 containers per treatment).3. Results
3.1. Seedling emergence of cabbage and arugula under different light treatments
Diagrams in Fig. 1 and 2 present the daily progression of seedling emergence for cabbage and arugula, respectively, over the 7 days evaluation period. In both cases, the first diagram illustrates the emergence percentage of seeds maintained under continuous darkness.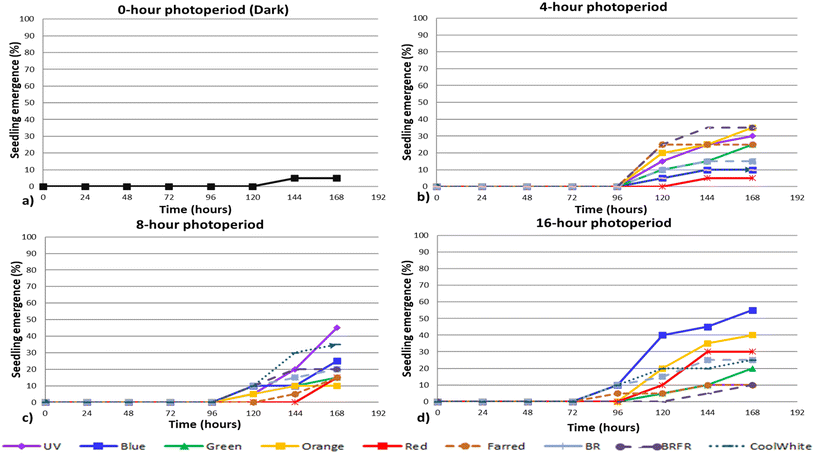 | ||
| Fig. 1 Seedling emergence (%) of cabbage under different lighting conditions: (a) continuous darkness (0 h photoperiod), (b) 4 h photoperiod, (c) 8 h photoperiod and (d) 16 h photoperiod. | ||
 | ||
| Fig. 2 Seedling emergence (%) of arugula under different lighting conditions: (a) continuous darkness (0 h photoperiod), (b) 4 h photoperiod, (c) 8 h photoperiod and (d) 16 h photoperiod. | ||
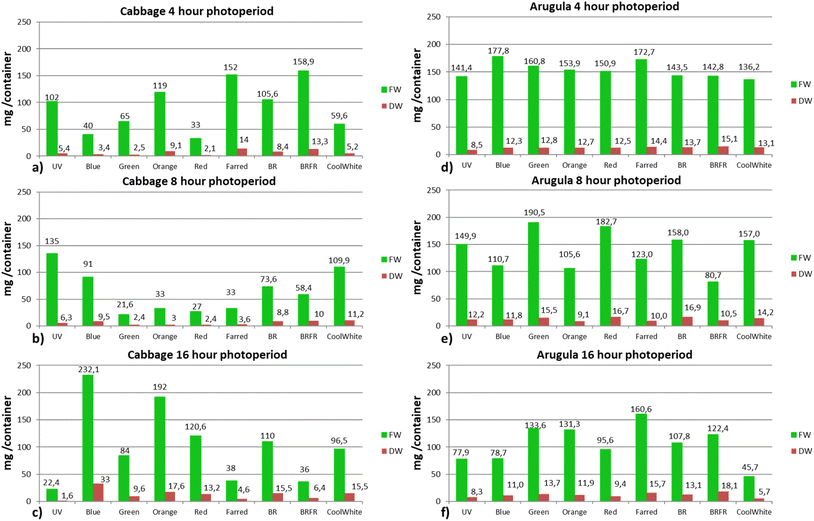
Under continuous darkness, only 5% of the seeds developed both a root and a shoot by the end of the 7 days period, representing the lowest emergence percentage among all treatments. A similarly low seedling emergence percentage (5%) was also observed under the 4 hR treatment. During the 4 h lighting application, seedling emergence ranged between 5% and 35%, with the 4hOr and 4hBRFR treatments showing the highest emergence (35%). Under the 8 h photoperiod, emergence varied from 10% (8hOr) to 45%, with the 8hUV treatment exhibiting the highest value. Under the 16 h photoperiod, the UV, far-red and blue-red-far-red spectrum produced the lowest emergence (10%), whereas blue light resulted in the highest percentage (55%). The remaining spectra within this photoperiod produced emergence levels ranging from 20% to 40% (Fig. 1).
Under the 4 h and 8 h photoperiods, the first seedlings emerged at 120 h, whereas under the 16 h photoperiod – specifically in the 16 hB, 16hFR, 16hBR and 16hCW treatments – the first seedlings emerged earlier, at 96 h.
In the case of arugula under dark conditions, the seedling emergence rate was 50%. The lowest seedling emergence rate among the lighting treatments was observed under the 16hCW spectrum (25%). All other light treatments resulted in emergence rates above 50%, with the highest value (85%) recorded under the 8 hG treatment. Specifically, the 4 h photoperiod produced emergence rates ranging from 65% to 75%, the 8 h photoperiod from 50% to 85%, and the 16 h photoperiod from 25% to 80% (16hFR) (Fig. 2).
Under the 4 h and 16 h photoperiods, seedling emergence was observed as early as 48 h after sowing, whereas under the 8 h photoperiod the first seedlings appeared at 72 h, similar to the dark treatment.
3.2. Produced seedlings under different lighting treatments
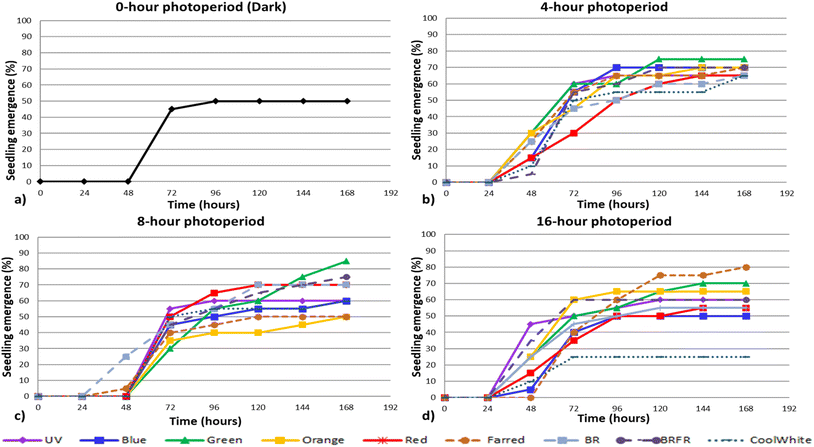
Fig. 3 presents the seedlings developed under each lighting treatment. For cabbage, seedlings grown under the 4 h photoperiod developed the longest shoots, with a mean length of 25.8 mm, whereas those grown under the 8 h and 16 h photoperiods reached mean shoot lengths of 14.1 mm and 18.4 mm, respectively (Table 2). The largest mean cotyledon area was observed under the 16 h photoperiod (45.2 mm2), while the smallest was recorded under the 8 h photoperiod (21.3 mm2). For arugula, seedlings grown under the 4 h photoperiod also exhibited the longest shoots, with a mean length of 25.4 mm, whereas those grown under the 16 h photoperiod developed the shortest shoots (21.3 mm). However, the 16 h photoperiod resulted in the largest mean cotyledon area (42.2 mm2), while the smallest cotyledon area was recorded under the 4 h photoperiod (32.4 mm2).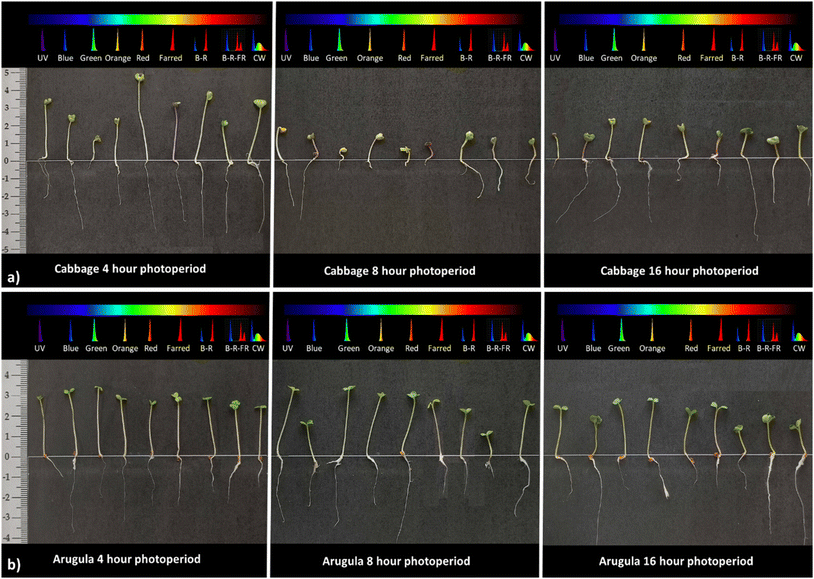 | ||
| Fig. 3 Morphological characteristics of the developed (a) cabbage and (b) arugula seedlings, under the different lighting treatments. | ||
| Photoperiod | N | Mean SL ± STDEV (mm) | Mean LA ± STDEV (mm2) |
|---|---|---|---|
| a LA corresponds to cotyledon area. | |||
| Cabbage | |||
| 4 hours | 38 | 25.8 ± 9.1 | 27.4 ± 12.1 |
| 8 hours | 40 | 14.1 ± 5.0 | 21.3 ± 9.6 |
| 16 hours | 45 | 18.4 ± 3.9 | 45.2 ± 14.5 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||
| Arugula | |||
| 4 hours | 123 | 25.4 ± 7.6 | 32.4 ± 10.9 |
| 8 hours | 116 | 24.0 ± 9.1 | 41.3 ± 12.5 |
| 16 hours | 104 | 21.3 ± 7.8 | 42.2 ± 11.7 |
Differences among the applied light spectra were observed in the morphology of cabbage and arugula seedlings (Table 3). For cabbage, under the 4 h photoperiod, the greatest shoot length was recorded under the red spectrum (47 mm – only one seedling developed under this treatment), while the largest mean cotyledon area occurred under the cool-white spectrum (47.5 mm2). Under the 8 h photoperiod, the longest mean shoot length was observed under UV light (17.7 mm), although cotyledon development was minimal under this spectrum (10.7 mm2). The BR spectrum produced the largest cotyledon area within this photoperiod (34 mm2). For the 16 h photoperiod, the green spectrum resulted in the longest mean shoot length (22 mm), whereas the BRFR spectrum produced the largest cotyledon area (58 mm2), representing the highest value recorded for cabbage across all lighting treatments.
| Light treatments | Cabbage | Arugula | ||||
|---|---|---|---|---|---|---|
| N | Mean SL ± STDEV (mm) | Mean LA ± STDEV (mm2) | N | Mean SL ± STDEV (mm) | Mean LA ± STDEV (mm2) | |
| a One-way ANOVA indicated significant differences in shoot length and leaf area among light spectra within each photoperiod (P < 0.05). However, in some treatments, emergence rates were too low to yield enough seedlings for valid post hoc comparisons. LA corresponds to cotyledon area. | ||||||
| 4hUV | 6 | 31.2 ± 7.9 | 17.7 ± 6.2 | 13 | 24.6 ± 6.9 | 15.4 ± 2.5 |
| 4 hB | 2 | 27.5 ± 0.7 | 39.0 ± 1.4 | 14 | 30.3 ± 6.1 | 39.7 ± 7.9 |
| 4 hG | 5 | 13.6 ± 2.1 | 15.8 ± 5.5 | 15 | 30.9 ± 6.3 | 24.9 ± 8.3 |
| 4hOr | 7 | 20.7 ± 5.5 | 15.1 ± 4.7 | 14 | 24.7 ± 6.6 | 34.0 ± 4.3 |
| 4 hR | 1 | 47.0 ± 0.0 | 42.0 ± 0.0 | 13 | 23.4 ± 6.9 | 21.3 ± 5.9 |
| 4hFR | 5 | 31.0 ± 2.9 | 36.0 ± 3.4 | 14 | 26.6 ± 5.9 | 38.0 ± 6.8 |
| 4hBR | 3 | 37.0 ± 2.6 | 38.0 ± 4.0 | 13 | 25.2 ± 8.3 | 39.5 ± 8.6 |
| 4hBRFR | 7 | 21.1 ± 5.3 | 34.7 ± 6.2 | 14 | 21.9 ± 5.8 | 43.4 ± 5.5 |
| 4hCW | 2 | 32.0 ± 4.2 | 47.5 ± 3.5 | 13 | 19.9 ± 6.4 | 35.0 ± 5.3 |
| 8hUV | 9 | 17.7 ± 6.2 | 10.7 ± 3.9 | 12 | 31.5 ± 4.7 | 27.0 ± 2.0 |
| 8hB | 5 | 16.4 ± 3.6 | 26.4 ± 7.9 | 12 | 15.9 ± 3.4 | 49.3 ± 7.0 |
| 8hG | 3 | 7.3 ± 0.6 | 12.7 ± 5.0 | 17 | 31.4 ± 8.7 | 46.9 ± 9.1 |
| 8hOr | 2 | 17.4 ± 2.8 | 26.0 ± 5.7 | 10 | 27.7 ± 6.6 | 19.5 ± 4.4 |
| 8hR | 3 | 8.3 ± 5.1 | 27.3 ± 2.5 | 14 | 27.5 ± 7.1 | 45.1 ± 4.3 |
| 8hFR | 3 | 8.6 ± 1.5 | 25.3 ± 7.6 | 10 | 25.1 ± 7.2 | 37.1 ± 6.7 |
| 8hBR | 4 | 16.3 ± 2.9 | 34.0 ± 9.1 | 14 | 22.5 ± 7.9 | 44.7 ± 8.6 |
| 8hBRFR | 4 | 14.3 ± 1.0 | 27.0 ± 7.4 | 15 | 12.4 ± 5.0 | 53.3 ± 9.5 |
| 8hCW | 7 | 13.0 ± 1.4 | 18.6 ± 6.5 | 12 | 22.3 ± 5.9 | 42.3 ± 4.4 |
| 16hUV | 2 | 16.0 ± 2.8 | 6.0 ± 1.4 | 12 | 23.9 ± 3.4 | 37.4 ± 3.4 |
| 16hB | 11 | 15.7 ± 2.8 | 47.5 ± 9.2 | 10 | 17.7 ± 4.9 | 50.3 ± 8.9 |
| 16hG | 4 | 22.0 ± 2.4 | 21.3 ± 5.4 | 14 | 25.8 ± 5.4 | 29.5 ± 7.7 |
| 16hOr | 8 | 21.4 ± 3.4 | 27.0 ± 8.5 | 13 | 28.1 ± 2.5 | 32.0 ± 8.6 |
| 16hR | 6 | 20.3 ± 3.7 | 29.8 ± 4.5 | 11 | 21.6 ± 4.5 | 40.3 ± 6.7 |
| 16hFR | 2 | 16.5 ± 3.5 | 16.0 ± 5.7 | 16 | 23.1 ± 3.7 | 47.6 ± 5.3 |
| 16hBR | 5 | 16.8 ± 2.2 | 44.0 ± 8.9 | 11 | 12.9 ± 3.4 | 38.4 ± 3.9 |
| 16hBRFR | 2 | 14.5 ± 0.7 | 58.0 ± 2.8 | 12 | 19.8 ± 4.6 | 61.2 ± 6.7 |
| 16hCW | 5 | 19.2 ± 5.1 | 40.6 ± 7.6 | 5 | 14.4 ± 2.1 | 53.4 ± 4.7 |
For arugula, under the 4 h photoperiod, the monochromatic blue and green spectra produced the longest seedlings, with mean shoot lengths of 30.3 mm and 30.9 mm, respectively. Under the 8 h photoperiod, the UV and green spectra were associated with the greatest shoot elongation, with mean lengths of 31.5 mm and 31.4 mm, respectively. Under the 16 h photoperiod, the orange spectrum produced the longest seedlings, with a mean shoot length of 28.1 mm. Across all photoperiods, the BRFR spectrum generated seedlings with the largest cotyledon area, measuring 43.4 mm2 under the 4 h photoperiod, 53.3 mm2 under the 8 h photoperiod, and 61.2 mm2 under the 16 h photoperiod.
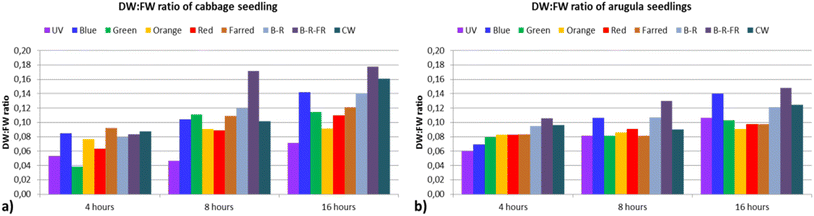 | ||
Fig. 5 Ratio of dry weight to fresh weight (DW![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) FW) biomass of seedlings which was produced from each lighting treatment for (a) cabbage and (b) arugula seeds. FW) biomass of seedlings which was produced from each lighting treatment for (a) cabbage and (b) arugula seeds. | ||
The differences in biomass values are attributed not only to variations in successful germination but also to distinct biochemical activities occurring in the seedlings, modulated by the lighting conditions. This is also reflected in the differences observed in the DW![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) FW biomass ratios across treatments.
FW biomass ratios across treatments.
DW![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) FW ratios ranged from 0.04 under the 4 hG treatment to nearly 0.18 under the 16hBRFR treatment. The BRFR spectrum also produced the highest ratio under the 8 h photoperiod (0.17). Under the 4 h photoperiod, the highest ratio was achieved with the FR spectrum (0.09). Ratios above 0.10 were recorded for all spectra under the 16 h photoperiod except UV and Orange, whereas under the 4 h photoperiod, ratios for all spectra remained below 0.10.
FW ratios ranged from 0.04 under the 4 hG treatment to nearly 0.18 under the 16hBRFR treatment. The BRFR spectrum also produced the highest ratio under the 8 h photoperiod (0.17). Under the 4 h photoperiod, the highest ratio was achieved with the FR spectrum (0.09). Ratios above 0.10 were recorded for all spectra under the 16 h photoperiod except UV and Orange, whereas under the 4 h photoperiod, ratios for all spectra remained below 0.10.
Regarding the DW![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) FW biomass ratio, the lowest value was observed under the 4hUV treatment (0.06), while the highest was recorded in the 16hBRFR treatment (0.15). Across all spectra, the DW
FW biomass ratio, the lowest value was observed under the 4hUV treatment (0.06), while the highest was recorded in the 16hBRFR treatment (0.15). Across all spectra, the DW![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) FW ratio tended to be higher under the 16 h photoperiod.
FW ratio tended to be higher under the 16 h photoperiod.
3.4. Microgreen production with different cultivation methods
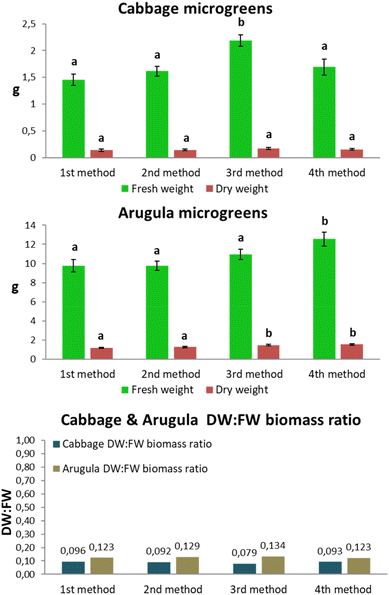
Fig. 6 presents the FW (green bars) and DW (brown bars) biomass values for cabbage and arugula microgreens grown under the four cultivation methods. For cabbage, the first method – beginning with an initial period of darkness – resulted in the lowest fresh and dry biomass, measuring 1.455 g and 0.139 g, respectively. The third method, which included an initial exposure to the light spectrum that produced the highest seedling emergence in the first phase of the experiment, yielded the highest FW and DW biomass values, measuring 2.189 g and 0.173 g, respectively. The FW biomass of the third method was 50.44% higher than that of the first method and 35.54% higher than that of the second method, while the corresponding DW biomass was 24.46% and 16.9% higher, respectively. The fourth method also produced higher FW and DW biomass compared to the first and second methods, although to a lesser extent than the third. The DW![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) FW ratio for the third method was the lowest among the four (0.079), whereas the ratios for the other three methods ranged narrowly between 0.092 and 0.096.
FW ratio for the third method was the lowest among the four (0.079), whereas the ratios for the other three methods ranged narrowly between 0.092 and 0.096.
For arugula, the fourth method resulted in the highest FW and DW biomass production. A one-day exposure to the spectrum associated with the highest emergence rate yielded 12.56 g of fresh biomass and 1.54 g of dry biomass. The third method also produced higher biomass than the first two methods; however, its FW biomass was 12.98% lower than that of the fourth method, and its DW biomass was 5.2% lower. Fresh biomass in the fourth method was 22.4% higher than in the first method and 22.5% higher than in the second, while DW biomass was 22.1% and 18.2% higher, respectively. The DW![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) FW ratios of the first, second, and fourth methods were similar (0.123–0.129), whereas the third method showed a slightly higher ratio of 0.134.
FW ratios of the first, second, and fourth methods were similar (0.123–0.129), whereas the third method showed a slightly higher ratio of 0.134.
4. Discussion
4.1. Seedling emergence
The present study examined the effects of different lighting spectra and photoperiods on seedling emergence, early morphological development, and biomass production of cabbage (Brassica oleracea var.capitata) and arugula (Eruca sativa). The results indicated that exposure to light was associated with an increased proportion of seeds that successfully emerged and developed shoots within the 7 days experimental period. Although cabbage and arugula are not strictly light-requiring species for germination, light has been reported to enhance or accelerate the transition from germination to visible seedling emergence in several small-seeded species. These results agree with previous studies showing that light can enhance seed responsiveness during the transition from germination to early seedling emergence, especially in species with small seeds and limited endogenous reserves.11,21,22 In microgreens cultivation, seeds are typically placed in darkness for the first few days after sowing and are subsequently transferred to the growth lighting. This dark phase is widely used to maintain high humidity around the seeds and to support uniform sprouting before exposure to light.25,26 However, in some species, appropriate light treatments can act as an early stimulus that promotes faster and more synchronized sprouting, leading to earlier shoot development and potentially higher seedling numbers.15,17 For this reason, in addition to the nine lighting spectra tested, a dark treatment was included in the experimental design to assess whether continuous light exposure from the first day could provide advantages over the commonly applied dark-start practice.For cabbage, all light treatments resulted in higher seedling emergence percentages (except 4hR) compared to those observed under dark conditions (5%). For each light spectrum tested, differences were observed among the three photoperiods: in some cases, longer exposure increased the number of emerged seedlings, whereas in others, shorter photoperiods were more favorable. Overall, when the emergence values were summed across all spectra, the 16 h photoperiod was associated with the highest total number of seedlings, exceeding both the 4 h and 8 h treatments. Among all treatments, 16 h of blue light showed the highest emergence percentage, reaching 55%. The increased number of emerged seedlings is directly associated with the fact that this treatment also yielded the highest number of germinated seeds (55%), all of which successfully developed shoots by the end of the treatment. This response is consistent with previous studies reporting that exposure to blue light can enhance germination in various species, including cabbage (Brassica oleracea) seeds and stevia (Stevia rebaudiana Bertoni) seeds.27,28
For arugula, only the 16hCW treatment (25%) produced an emergence percentage lower than that recorded under darkness (50%), while all other treatments yielded equal or higher values. The highest overall seedling output was observed under the 4 h photoperiod, where all spectra produced emergence percentages between 65% and 75%. All treatments exhibited high germination percentages (above 65% and higher than in darkness), which contributed to the elevated emergence values. However, not all germinated seeds completed shoot development within the 7 days period. Although several spectra resulted in high emergence percentages, the highest value (85%) was observed under an 8 h application of green light. Green light is not commonly used, nor widely investigated, as a germination-promoting wavelength; nevertheless, similarly positive responses have been reported by Vogel & Makedo in their study on orchid (Cyrtopodium glutiniferum Raddi) seeds.29 Opposing findings have also been documented. In their work on Vigna unguiculata (L.) Walp seeds, Lal and Sachan found that red light supported the highest germination percentage, whereas green light did not promote germination.18 Conversely, Hernandez-Adasme et al. reported that blue light enhanced germination in two lettuce cultivars (‘Levistro’ and ‘Carmoli’), while red light reduced it.19 They additionally observed that a mixed spectrum containing 52% blue, 27% green, and 21% red accelerated germination and increased the proportion of germinated seeds, resulting in a greater number of developed seedlings.
4.2. Seedling morphology and growth responses
Although specific photoreceptors in seeds and plants are traditionally associated with certain wavelengths of light, the results of this study suggest that photoreceptors may also respond to a broader range of wavelengths than previously understood. Different plant species might rely on distinct wavelengths to trigger seed germination and early-stage seedling development, indicating species-specific photoreceptive mechanisms. In particular, very few studies have investigated the effects of green light spectrum on plants, from the seed stage and throughout their life cycle. The prevailing perception was that, since green light is mostly reflected rather than absorbed by plants, it does not contribute to their growth. However, recent studies have demonstrated its significant role. Research shows that green light can penetrate deeper into the leaf canopy compared to blue and red wavelengths, thereby enhancing photosynthesis in the lower layers of the leaves.30,31 Experiments with lettuce plants have shown that green light promotes stem elongation and stimulates stomatal opening. Furthermore, these studies suggest that green wavelengths enhance the early-stage growth of seedlings by increasing biomass and leaf expansion; however, their influence diminishes as the plant matures.30,32 Experiments with Arabidopsis thaliana seedlings have shown that the addition of low-intensity (>10 µmol m−2 s−1) green light promotes hypocotyl elongation and counteracts the effects of red and far-red light. This phenomenon was primarily attributed to the mediation of phototropin 1 and cryptochromes.33 Similar shoot elongation characteristics were observed in the experiments of this study, where arugula seedlings, across all photoperiods, exhibited longer stems under green light treatments. For cabbage, this effect was only observed under the 16 h photoperiod (16 hG).In the experiments with mixed light spectra, arugula seedlings had shorter stem lengths compared to monochromatic spectra (except 8 hB and 16 hB), across all photoperiods. For cabbage, this was observed only under the 16 h lighting application. In most experiments, mixed light spectra were associated with higher DW![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) FW biomass ratio compared to single spectrum treatments, indicating lower water content and increased solid compound concentration, which are desirable characteristics for microgreens. Similar characteristics were also observed in seedlings grown under blue light. The blue spectrum has been reported to reduce water retention, enhance the accumulation of phenolic compounds, and consequently increase dry biomass production.19,34 In the case of cabbage, far-red light produced seedlings with morphological characteristics similar to those grown under the blue spectrum, while the stem and leaves developed a reddish coloration. This reddish pigmentation is likely due to the activation and production of anthocyanins.35 The DW
FW biomass ratio compared to single spectrum treatments, indicating lower water content and increased solid compound concentration, which are desirable characteristics for microgreens. Similar characteristics were also observed in seedlings grown under blue light. The blue spectrum has been reported to reduce water retention, enhance the accumulation of phenolic compounds, and consequently increase dry biomass production.19,34 In the case of cabbage, far-red light produced seedlings with morphological characteristics similar to those grown under the blue spectrum, while the stem and leaves developed a reddish coloration. This reddish pigmentation is likely due to the activation and production of anthocyanins.35 The DW![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) FW ratio under far-red light was higher than blue light for the 4 and 8 h photoperiods.
FW ratio under far-red light was higher than blue light for the 4 and 8 h photoperiods.
Monochromatic light treatments, in both species examined, generally produced seedlings with elongated stems and reduced cotyledon expansion. Because microgreens have a very short growth cycle, light exposure strongly influences photomorphogenesis, which in turn regulates key developmental traits such as stem elongation and leaf enlargement – both directly linked to photosynthetic performance.10,36 When post-germination light conditions fail to adequately activate the relevant photoreceptors, seedlings exhibit characteristics resembling skotomorphogenesis: they prioritize stem elongation and produce small, pale leaves, a morphological strategy that enables them – under natural conditions – to reach light above the soil surface.37,38 This phenomenon was clearly reflected in the contrasting morphological patterns observed between the 4 h and 16 h photoperiods. Under the 4 h photoperiod, both cabbage and arugula developed longer shoots yet smaller cotyledons, whereas the 16 h photoperiod produced seedlings with more expanded cotyledons and shorter stems. These opposite trends demonstrate that targeted manipulation of early-stage light quality and duration can be used strategically to direct seedling morphology – either promoting shoot elongation or enhancing cotyledon growth, depending on the desired production traits. In short-cycle crops such as microgreens, improvements in seedling emergence and early morphological development directly determine the number and uniformity of plants entering the growth phase, and therefore strongly influence final yield and production efficiency.
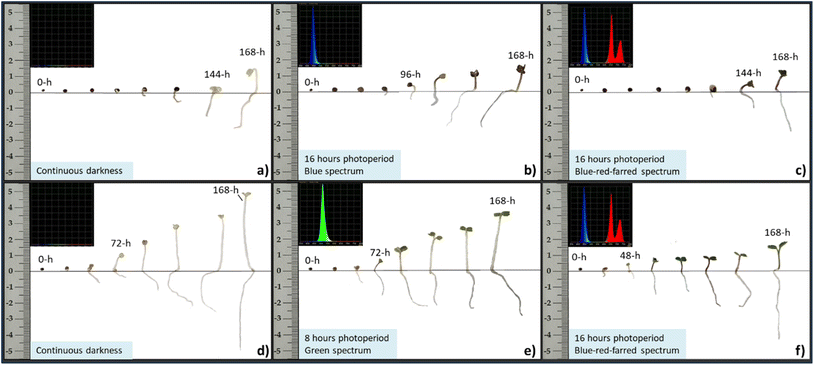
Fig. 8 illustrates the developmental progression of cabbage (top row) and arugula (bottom row) seedlings under three representative treatments: continuous darkness, the light spectrum that produced the highest number of emerged seedlings (16 h blue light for cabbage and 8 h green light for arugula), and the 16 h blue-red-far-red spectrum, which yielded the highest DW![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) FW biomass ratio. Although growth in darkness does not represent a typical developmental pathway, it is included here because it reflects a common practice in microgreens production, where seeds are kept without light for the first days after sowing until radicle emergence and early shoot formation occur. Presenting these treatments side by side provides a visual comparison that complements the quantitative results and highlights how light quality and photoperiod shape early seedling development.
FW biomass ratio. Although growth in darkness does not represent a typical developmental pathway, it is included here because it reflects a common practice in microgreens production, where seeds are kept without light for the first days after sowing until radicle emergence and early shoot formation occur. Presenting these treatments side by side provides a visual comparison that complements the quantitative results and highlights how light quality and photoperiod shape early seedling development.
In both species, seedlings that developed under complete darkness exhibited pale, undersized cotyledons and elongated hypocotyls-morphological traits characteristic of skotomorphogenesis and the absence of photosynthetic light. When seeds are intentionally kept in darkness until the radicle and hypocotyl emerge, as is common practice in microgreens production, it is essential to monitor the timing of shoot emergence carefully so that seedlings can be transferred promptly to growth lighting, preventing excessive elongation and weakened early development.
Cabbage seedlings exposed to blue light exhibited faster shoot development, shorter and more compact hypocotyls, and larger, intensely green cotyledons. These traits reflect the strong influence of blue wavelengths on photomorphogenesis and photosynthetic activation during early seedling growth. In arugula exposed to green light, an intermediate pattern was observed: seedlings displayed moderate hypocotyl elongation – resembling a partial skotomorphogenic response – while cotyledons exhibited a more distinct green coloration and greater surface area compared to those developed in darkness. This suggests that, although green light is generally considered less effective than blue or red wavelengths in driving seedling development, it still contributes to early-stage growth to some extent. Under the mixed BRFR spectrum, both species produced seedlings with compact morphology, well-expanded cotyledons, and strong green pigmentation. These traits correspond with the higher DW![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) FW ratios measured under this treatment, indicating a more balanced allocation of biomass and improved structural development during the early seedling stage.
FW ratios measured under this treatment, indicating a more balanced allocation of biomass and improved structural development during the early seedling stage.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) FW biomass ratio, microgreens production increased in both species. This sequential lighting approach (emergence-enhancing spectrum during the first days followed by a biomass-enhancing growth spectrum) was more effective than the conventional practice of keeping seeds in darkness for several days before exposure to growth lighting. Specifically, for cabbage, the third method (M3), which included a five-day initial exposure to blue light under a 16 h photoperiod, produced the highest yield (2.189 g). The fourth method (M4), involving only one day of initial exposure to the emergence-enhancing spectrum, resulted in a yield of 1.692 g. In arugula, the green-light treatment applied for 5 days in M3 (8 h photoperiod) produced 10.93 g of microgreens, whereas M4 resulted in the highest yield (12.56 g). The conventional approach used in microgreens production (M1) – in which seeds are kept in darkness for approximately three days before exposure to growth lighting, produced lower yields in both species.25,26 For cabbage, M1 resulted in a yield approximately 34% lower than the recommended M3 and around 14% lower than M4. For arugula, the yield obtained with M1 was about 11% lower than M3 and roughly 22% lower than M4. The second method (M2), where seeds were placed under the growth spectrum from the start, also improved microgreens production compared to M1 for cabbage. For arugula, however, M1 and M2 produced similar yields.
FW biomass ratio, microgreens production increased in both species. This sequential lighting approach (emergence-enhancing spectrum during the first days followed by a biomass-enhancing growth spectrum) was more effective than the conventional practice of keeping seeds in darkness for several days before exposure to growth lighting. Specifically, for cabbage, the third method (M3), which included a five-day initial exposure to blue light under a 16 h photoperiod, produced the highest yield (2.189 g). The fourth method (M4), involving only one day of initial exposure to the emergence-enhancing spectrum, resulted in a yield of 1.692 g. In arugula, the green-light treatment applied for 5 days in M3 (8 h photoperiod) produced 10.93 g of microgreens, whereas M4 resulted in the highest yield (12.56 g). The conventional approach used in microgreens production (M1) – in which seeds are kept in darkness for approximately three days before exposure to growth lighting, produced lower yields in both species.25,26 For cabbage, M1 resulted in a yield approximately 34% lower than the recommended M3 and around 14% lower than M4. For arugula, the yield obtained with M1 was about 11% lower than M3 and roughly 22% lower than M4. The second method (M2), where seeds were placed under the growth spectrum from the start, also improved microgreens production compared to M1 for cabbage. For arugula, however, M1 and M2 produced similar yields.Early light exposure also helps explain why methods M3 and M4, which applied an emergence-enhancing spectrum before switching to BRFR growth lighting, resulted in higher microgreens yields compared with method M2, where seeds were placed directly under 16hBRFR. The initial wavelength–photoperiod combination used in M3 and M4 likely provided a stimulus that triggered earlier germination and supported faster early development, increasing the number of seedlings entering the growth stage and ultimately enhancing total microgreens yield.
Additionally, the large difference observed between cabbage and arugula microgreens yield is partly explained by the sowing strategy: in all treatments, seeds were distributed based on weight (0.85 g per tray). Because arugula seeds have lower individual seed mass – and also showed higher emergence capacity – each tray contained a substantially greater number of viable arugula seeds, resulting in a higher number of seedlings and, consequently, greater total microgreens production.
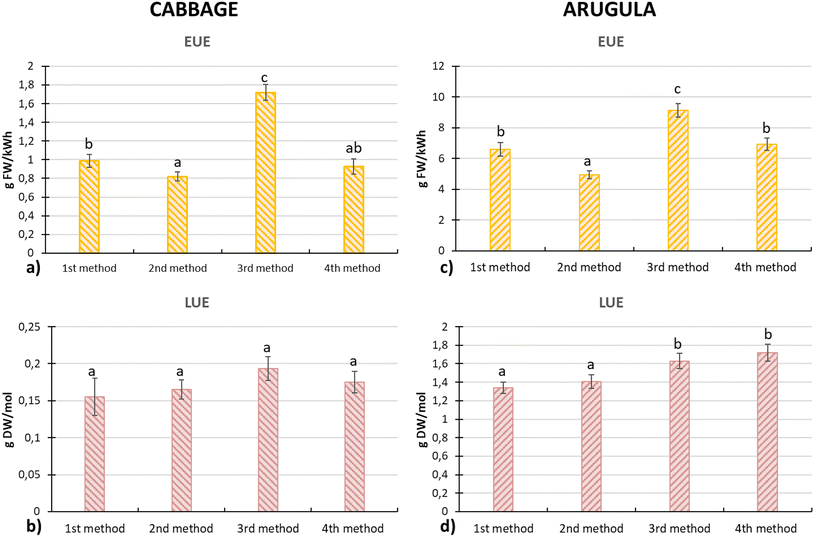
4.4. Energy and light use efficiency
The increased seedling production observed in methods M3 and M4 also led to more efficient use of both energy and light. In cabbage, the highest EUE value was recorded in method M3 (1.72 g FW per kW h), which was approximately 79% higher than that of the conventional method M1. Similarly, for arugula, method M3 achieved an EUE of 9.14 g FW per kW h, representing an increase of about 38% compared to the conventional method M1 (6.61 g FW per kW h). Although M1 involved 9 days of lighting while M3 applied lighting for the full 12 days cultivation period (higher energy consumption), the greater number of seeds that successfully developed shoots in M3 resulted in a higher final microgreens yield, which in turn contributed to improved EUE. For cabbage, the highest LUE value was obtained with method M3, reaching 0.19 g DW per mol, which represents an improvement of approximately 19% compared with the conventional method M1. Method M4 also showed an increased LUE relative to M1, with a value about 13% higher. For arugula, the highest LUE was recorded under method M4, at 1.72 g DW/mol, representing an increase of approximately 28% over M1. Method M3 achieved an LUE of 1.63 g DW per mol, which was about 23% higher than that of M1.The reason why method M4 (1 day initial lighting) produced the highest LUE in arugula, whereas cabbage reached its highest LUE under method M3 (5 days initial lighting), is linked to species-specific differences in the speed of early development. Arugula seeds germinated within the first 24 h and initiated shoot development as early as 48–72 h, meaning that even a single day of exposure to the emergence-enhancing spectrum was sufficient for seedlings to receive the necessary stimulus before transitioning to the growth spectrum (16hBRFR). As a result, arugula entered the growth-lighting phase earlier, enabling faster biomass accumulation under method M4. In contrast, cabbage exhibited slower radicle and shoot development; therefore, a longer initial exposure period, as applied in M3, was required for the spectrum to exert its beneficial effects before seedlings were transferred to the growth phase. Consequently, cabbage achieved higher LUE under M3 rather than M4.
4.5. Limitations and future research
Although several studies have been conducted on the effect of different light spectra for various seeds and seedlings, there is a lack of research concerning the combination of lighting strategies that could achieve faster and more efficient seedling development during their early growth stages.9,10,15,22 In the present study, the observed differences among lighting strategies highlight the need for species-specific optimization. Both light quality and photoperiod influenced not only the proportion of emerged seedlings but also their early shoot growth, which is important for achieving uniform and reliable seedling production. However, the number of observations per treatment was constrained by the proportion of seeds that successfully emerged, resulting in variable and, in some cases, limited sample sizes. Under these conditions, the results should be considered indicative of general trends rather than definitive comparisons.Additionally, because PPFD was constant across treatments, these observed differences reflect the combined effect of lighting duration and daily light integral (DLI), rather than photoperiod alone. This is particularly relevant for the UV treatment, where the substantially lower applied light intensity resulted in correspondingly lower DLI values compared to the PAR spectra, limiting direct comparability with the other lighting conditions. Therefore, the morphological characteristics observed in seedlings developed under UV radiation should be interpreted within the context of the applied irradiation level, and not as an indication of reduced biological relevance for seedling development.
Future studies would benefit from the use of a greater number of seeds per treatment and increased replication, as well as the evaluation of a broader range of environmental conditions (e.g., temperature and relative humidity), in order to more comprehensively assess the effects of light spectrum on seedling emergence and development. Further research across a wider range of light intensities and spectral combinations would contribute to refining species-specific lighting strategies and improving early seedling performance in CEAs.
5. Conclusions
In this study, nine different light spectra were evaluated under three photoperiods (4 h, 8 h, and 16 h) applied under constant light intensity to identify the conditions that most effectively increased seedling emergence and reduced the time required for shoot development in cabbage and arugula. The results indicated that a 16 h blue-light treatment resulted in the highest seedling emergence in cabbage, whereas an 8 h green-light treatment produced the highest emergence values in arugula. The mixed blue-red-far-red spectrum generated seedlings with more compact morphology and greater dry biomass accumulation in both species. Applying a combined lighting strategy, beginning with the spectrum that most effectively promoted seedling emergence and followed by the spectrum that enhanced dry biomass accumulation, proved more effective than using a single lighting condition throughout cultivation. This staged approach increased the number of seedlings that successfully entered the growth phase and led to higher final microgreens yields for both cabbage and arugula. Based on the experimental results, this staged lighting approach was associated with improved energy-use efficiency and light-use efficiency compared with the conventional microgreens production practice, within the conditions of the present study.These findings suggest that identifying species-specific lighting requirements is particularly important for improving production efficiency in microgreens cultivation, which is characterized by short production cycle. Optimizing early-stage lighting may contribute to shorter cultivation time and increased yield uniformity. Moreover, targeted manipulation of light quality may provide a practical means to guide seedling morphology toward desirable traits without increasing overall energy input. Together, these findings highlight the potential of stage-specific lighting strategies to support more sustainable microgreens production in urban controlled-environment systems.
Author contributions
Christos Vatistas: conceptualization, methodology, project administration, investigation, data curation, formal analysis, visualization, validation, writing – original draft, writing – review & editing. Dafni Despoina Avgoustaki: writing – review & editing. Thomas Bartzanas: supervision.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
All data generated or analyzed during this study are included in this article.Acknowledgements
This research received no external funding.References
- L. Graamans, E. Baeza, A. Van Den Dobbelsteen, I. Tsafaras and C. Stanghellini, Agric. Syst., 2018, 160, 31–43, DOI:10.1016/j.agsy.2017.11.003.
- O. Akaeze and D. Nandwani, Urban Agric. Reg. Food Syst., 2020, 5, e20002, DOI:10.1002/uar2.20002.
- A. Zaręba, A. Krzemińska and R. Kozik, Resources, 2021, 10, 109, DOI:10.3390/resources10110109.
- Food and Agriculture Organization of the United Nations, The State of Food and Agriculture 2019: Moving Forward on Food Loss and Waste Reduction, FAO, Rome, 2019, Retrieved from http://www.fao.org/3/ca6030en/ca6030en.pdf Search PubMed.
- D. Despommier, Trends Biotechnol., 2013, 31, 388–389, DOI:10.1016/j.tibtech.2013.03.008.
- O. Voutsinos, M. Mastoraki, G. Ntatsi, G. Liakopoulos and D. Savvas, Agriculture, 2021, 11, 503, DOI:10.3390/agriculture11060503.
- C. Vatistas, D. D. Avgoustaki and T. Bartzanas, Atmosphere, 2022, 13, 1258, DOI:10.3390/atmos13081258.
- Y. Zhang, Z. Xiao, E. Ager, L. Kong and L. Tan, J. Future Foods, 2021, 1, 58–66, DOI:10.1016/j.jfutfo.2021.07.001.
- R. N. Maru, J. Wesonga, H. Okazawa, A. Kavoo, J. O. Neondo, D. M. Mazibuko, S. Maskey and F. Orsini, Horticulturae, 2024, 10, 436, DOI:10.3390/horticulturae10050436.
- S. Toscano, V. Cavallaro, A. Ferrante, D. Romano and C. Patané, Plants, 2021, 10, 1584, DOI:10.3390/plants10081584.
- C. Vatistas, D. D. Avgoustaki, G. Monedas and T. Bartzanas, Sci. Hortic., 2024, 331, 113118, DOI:10.1016/j.scienta.2024.113118.
- J. Balasus, J. Blank, S. Babilon, T. Hegemann and T. Q. Khanh, Agronomy, 2021, 11, 2570, DOI:10.3390/agronomy11122570.
- C. Nájera, V. M. Gallegos-Cedillo, M. Ros and J. A. Pascual, Biol. Life Sci. Forum, 2022, 16, 24, DOI:10.3390/IECHo2022-12514.
- R. Paradiso and S. Proietti, J. Plant Growth Regul., 2022, 41, 742–780, DOI:10.1007/s00344-021-10337-y.
- Y. Wei, S. Wang and D. Yu, Plants, 2023, 12, 2746, DOI:10.3390/plants12142746.
- I. Paik and E. Huq, Cell Dev. Biol., 2019, 92, 114–121, DOI:10.1016/j.semcdb.2019.03.007.
- M. Seo, E. Nambara, G. Choi and S. Yamaguchi, Plant Mol. Biol., 2009, 69, 463–472, DOI:10.1007/s11103-008-9429-y.
- N. Lal and P. Sachan, Indian J. Biol., 2017, 4, 2, DOI:10.21088/ijb.2394.1391.4217.10.
- C. Hernández-Adasme, H. Silva and V. Escalona, Ital. J. Agron., 2022, 17, 1982, DOI:10.4081/ija.2022.1982.
- R. Nabipour Sanjbod, Y. P. Hir, E. Chamani and A. Estaji, Russ. J. Plant Physiol., 2023, 70, 121, DOI:10.1134/S1021443723601052.
- J. Flores, C. González-Salvatierra and E. Jurado, J. Plant Ecol., 2016, 9, 174–179, DOI:10.1093/jpe/rtv046.
- H. Bu, W. Ge, X. Zhou, W. Qi, K. Liu, D. Xu, X. Wang and G. Du, Plant Species Biol., 2017, 32, 263–269, DOI:10.1111/1442-1984.12147.
- K. Liu, M. Gao, H. Jiang, S. Ou, X. Li, R. He, Y. Li and H. Liu, Molecules, 2022, 27, 883, DOI:10.3390/molecules27030883.
- D. Filatov and I. Olonin, E3S Web Conf., 2023, 383, 04074, DOI:10.1051/e3sconf/202338304074.
- P. I. Moraru, T. Rusu and O. S. Mintas, Foods, 2022, 11, 1327, DOI:10.3390/foods11091327.
- A. Singh, J. Singh, S. Kaur, M. Gunjal, J. Kaur, V. Nanda, R. Ullah, S. Ercisli and P. Rasane, Food Chem. X, 2024, 23, 101527, DOI:10.1016/j.fochx.2024.101527.
- Y. Liu, J. Xue, G. Che, L. Guo and J. Tang, Agric. Food Sci., 2022, 31, 78–88, DOI:10.23986/afsci.119416.
- M. Simlat, P. Ślęzak, M. Moś, M. Warchoł, E. Skrzypek and A. Ptak, Sci. Hortic., 2016, 211, 295–304, DOI:10.1016/j.scienta.2016.09.009.
- I. N. Vogel and A. F. Macedo, Plant Cell, Tissue Organ Cult., 2011, 104, 147–155, DOI:10.1007/s11240-010-9810-9.
- C. E. Wong, Z. W. N. Teo, L. Shen and H. Yu, Trends Food Sci. Technol., 2020, 106, 48–63, DOI:10.1016/j.tifs.2020.09.031.
- Y. Chen, Z. Bian, L. F. M. Marcelis, E. Heuvelink, Q. Yang and E. Kaiser, J. Exp. Bot., 2024, 75, 5655–5666, DOI:10.1093/jxb/erae259.
- T. Zhang and K. M. Folta, Plant Signaling Behav., 2012, 7, 75–78, DOI:10.4161/psb.7.1.18635.
- Y. Wang, S. A. Maruhnich, M. H. Mageroy, J. R. Justice and K. M. Folta, Planta, 2013, 237, 225–237, DOI:10.1007/s00425-012-1767-y.
- R. Vrkić, J. Šic Žlabur, M. Dujmović and B. Benko, Horticulturae, 2024, 10, 249, DOI:10.3390/horticulturae10030249.
- Y. Ma, X. Ma, X. Gao, W. Wu and B. Zhou, Int. J. Mol. Sci., 2021, 22, 11116, DOI:10.3390/ijms222011116.
- L. G. Izzo, B. Hay Mele, L. Vitale, E. Vitale and C. Arena, Environ. Exp. Bot., 2020, 179, 104195, DOI:10.1016/j.envexpbot.2020.104195.
- S. Tripathi, Q. T. N. Hoang, Y.-J. Han and J.-I. Kim, Int. J. Mol. Sci., 2019, 20, 6165, DOI:10.3390/ijms20246165.
- Y. Luo, J. Liang and G. Zeng, Chemosphere, 2020, 242, 125261, DOI:10.1016/j.chemosphere.2019.125261.
| This journal is © The Royal Society of Chemistry 2026 |