Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Benefits and challenges of fortified beverage systems with brewer's spent grain-derived protein isolates
Niamh Aherna,
Laura Nyhan a,
Elke K. Arendt
a,
Elke K. Arendt ab,
Patrick O'Riordanc and
Aylin W. Sahin
ab,
Patrick O'Riordanc and
Aylin W. Sahin *a
*a
aSchool of Food and Nutritional Sciences, University College Cork, Cork, Ireland. E-mail: niamh.ahern@umail.ucc.ie; lnyhan@ucc.ie; e.arendt@ucc.ie; aylin.sahin@ucc.ie; Tel: +353-21-4902880
bAPC Microbiome Ireland, University College Cork, Cork, Ireland
cEverGrain LLC, St. Louis, MO 63118, USA. E-mail: patrick.oriordan@ab-inbev.com
First published on 20th April 2026
Abstract
Plant protein soft drinks align with current consumer demand for hydration, nutrition and sustainability yet faces challenges due to protein aggregation and precipitation in an acidic environment. This study evaluated the effects of two different barley and rice protein isolates, EverPro® Dark Fraction (EDF) and a decolourised version, EverPro® Light Fraction (ELF), at ingredient inclusion levels of 0%, 2.5%, 5% and 10% in diverse beverage systems including water as a control, apple juice, a full sugar and sugar free carbonated drink, a sweetened ice tea, an unsweetened black tea and a non-alcoholic beer. Physicochemical properties were comprehensively assessed. Protein inclusion, particularly at 10%, elevated pH to a more neutral range (from 3.1 and 4.88 to 5.0 and 7.8). Increasing the addition of both ingredients also elevated viscosity (from 1.04 and 1.46 to 2.54 and 5.43 mPa s), density (0.999 and 1.045 to 1.030 and 1.074 g cm−3) as well as lubrication properties of all beverages with ELF exhibiting higher values compared to EDF. Dispersion stability improved dramatically including turbidity and particle size at higher inclusion levels (10%), influenced by changes in pH further from the proteins isoelectric point. Low inclusion levels (2.5%) caused maximum turbidity and separation rate. Principal component analysis revealed distinct clustering by beverage matrix and protein type, with apple juice most affected. These findings demonstrate a step towards soft drinks fortified with upcycled protein from brewers spent grain with added nutritional benefits for future commercially potential.
Sustainability spotlightBrewer's spent grain (BSG) is the most abundant side-stream in the brewing industry. Since BSG is rich in protein, several research has been conducted in extracting and characterising the protein fraction. This research article investigates the potential of BSG-derived protein to be added in different beverage systems resulting in significant insights for the development of high protein soft drinks and in further consideration of sensory attributes. Applications of upcycled proteins scares, but if thoroughly tested, can contribute to transforming our food system towards more sustainable food systems by reducing food loss during food production process. This work aligns with SDG2 (enhancing food security), SDG3 (food fortification), SDG12 (upcycling of by-products), and SDG13 (reducing food losses that cause global warming). |
1 Introduction
Conventional soft drinks offer little to no nutritional value, primarily composed of water, sugar or sweeteners, acidulants and flavourings.1 Other soft drinks, such as juices, teas and non-alcoholic beers, also offer limited nutritional value but can provide essential micronutrients including vitamins, minerals, antioxidants and polyphenols. Protein-based beverages are continuing to grow as popular functional or sports product. Most research has been focused on plant-based milk alternatives and the ongoing challenges they face, such as nutrition, stability and flavour.2 These plant-based beverages or drinks are made from a variety of plant sources including cereals, nuts, and legumes, whereas typical commercial plant-based milk substitutes include the use of almonds, cashews, hazelnuts, rice, quinoa, oat, hemp and soy.3–5 Consumers are seeking a shift away from traditional protein smoothies or shakes to clear beverage options that can address thirst and provide hydration.6 Beverages such as these can be a convenient option to supply a high dose of protein. The functional drinks market is one of the fastest growing industries, with functional waters emerging as one of the most popular beverages in this category.6 However, most soft drinks have an acidic pH range, which can cause proteins to precipitate.7 Furthermore, these beverages are commonly thermally processed to extend their shelf life by reducing harmful microbes and inactivating enzymes. This is typically achieved through methods like pasteurisation or ultra-high temperature (UHT) sterilisation, which may alter their flavour, aroma and colour.4,5 As a result, stabilizing protein in both acidic conditions and high temperatures presents a challenge, as turbidity, phase separation, viscosity and gelation can be affected.8 Additionally, the incorporation of plant-based proteins into beverages is often associated with challenges such as sensorial, imbalanced amino acid profiles, low bioavailability, and the presence of antinutrients. These sensory challenges can include undesirable beany flavours, astringency, bitterness and mouthcoating mainly attributed from the polyphenols.9–11 These hurdles can limit consumer acceptance. Specifically for carbonated beverages, including high levels of protein could also lead to processing issues during production such as excessive foaming.1,12,13 Protein hydrolysates can be an excellent ingredient for fortifying these beverages due to their high solubility and greater thermal and acidic stability when compared to larger molecular size proteins. Methods used for hydrolysing proteins can include biological processes (including fermentation and enzyme treatment), physical, or chemical processes, while each method has its own advantages and disadvantages.14 Brewing is an example of a process that causes protein degradation leading to partially hydrolysed proteins and small peptides in brewers spent grain.15The demand for high protein food and beverage products has risen significantly in recent years with a particular emphasis on plant-based protein options as consumers increasingly identify as vegetarian, vegan or even flexitarian, and due to environmental and ethical considerations.16 As a result, majority of the leading dairy companies are incorporating plant-based alternatives into their product portfolio. Numerous studies have focused on animal-based protein beverages, particularly fortifying various flavoured soft drinks with whey protein.17–20 Many of these whey-based flavoured soft drinks are formulated with low to medium protein contents (ranging from 0.8% to 6% per 100 ml) and are limited to certain beverage types such as juices, while tea and non-alcoholic beers have not been explored. Moreover, there has been limited research on the inclusion of plant-based proteins in soft drinks.
The valorisation of protein ingredients recovered from food processing side streams is critical to reduce food waste and improve resource efficiency for producing alternative proteins to address future protein demands. EverPro™ Dark Fraction (EDF) and Everpro™ Light Fraction (ELF) produced by the company EverGrain Ingredients, are commercially available barley and rice protein isolates derived from upcycled brewers spent grain.21,22 ELF (beige/sandy colour) is a decolourised version of EDF (dark brown colour), with higher L* brightness values and distinctive physiochemical properties.23
This study tested two hypotheses (1) Does EDF and ELF affect techno-functionality of the beverages? And if so, is the impact similar or significantly different? and (2) Does the ingredient addition level change the techno-functionality of the beverages? If yes, are these changes beneficial or do they cause challenges?
2 Materials and methods
2.1 Materials
The barley and rice protein isolates, EDF and ELF were obtained from EverGrain Ingredients (St. Louis, MO, USA). The apple juice from concentrate (AJ), carbonated lemon and lime soft drink (LL), carbonated lemon and lime soft drink sugar free (LL Free), sweetened peach flavoured ice tea (IT), unsweetened black tea bags (BT) and non-alcoholic lager beer (NAB) were all purchased from local supermarkets in Cork, Ireland. The list of ingredients and nutritional composition of each beverage used in the study are shown in Table 1. Chemicals for the analyses were purchased from Sigma-Aldrich (St. Louis, MO, USA) unless stated otherwise.| Soft drink | Calories (kcal) | Carbohydrates (g) | Of which sugar (g) | Ingredients list |
|---|---|---|---|---|
| Water | 0 | 0 | 0 | Water |
| Apple juice | 46 | 11.1 | 11.1 | Apple juice from concentrate |
| Lemon & lime* | 19 | 4.7 | 4.7 | Carbonated water, sugar, acids (citric acid, malic acid), natural lemon and lime flavouring with other natural flavourings, acidity regulator (sodium citrate), sweeteners (acesulfame K, sucralose) |
| Lemon & lime free* | 1 | 0 | 0 | Carbonated water, acids (citric acid, malic acid), natural lemon and lime flavouring, acidity regulator (sodium citrate), sweeteners (aspartame, acesulfame K), preservative (sodium benzoate) |
| Ice tea peach | 13 | 3.1 | 3 | Water, sugars (sucrose, fructose), acids (malic acid, citric acid), black tea extract (0.12%), peach juice from concentrate (0.1%), acidity regulator (trisodium citrate), flavourings, antioxidant (ascorbic acid), sweetener (steviol glycosides from stevia) |
| Black tea | 0 | 0 | 0 | 100% black tea |
| Non-alcoholic beer* | 14 | 3.1 | 0.2 | Water, malted barley, hops, yeast |
2.2 Preparation of protein beverages
EDF and ELF were added singly to a variety of beverage systems (water, AJ, LL, LL Free, IT, BT, and NAB) at four different concentrations, 0%, 2.5%, 5% and 10%. These concentrations were chosen to match the protein content of protein soft drinks that were available on the global market from a previous study performed (i.e. for commercial relevance).24 According to the protein content of the ingredients each beverage had a final protein content of approximately 2%, 4% and 8% with the addition of 2.5%, 5% and 10% (w/w) of ingredient, respectively.23 Protein solubility was done on ingredients exceeding 80% across pH 2–9, relevant to soft drinks acidic value. A range of different drinks were chosen to cover a variety of beverage matrices. Carbonated beverages (LL, LL Free, NAB) were decarbonated before protein addition using ultrasonication as previously described.24 The BT was prepared using package instructions by brewing 2 teabags in 1 L of boiled water for 4 min, followed by the removal of the teabags and cooling until it reached room temperature (∼20 °C). The beverages were mixed using a vortex, then left for 2 h to allow further hydration, followed by a 30 s mixing stage using an Ultra-Turrax equipped with a S10N-10G dispersing element at speed 3 (IKA Labortechnik, Janke and Kunkel GmbH, Staufen, Germany). All beverages were freshly prepared prior to analysis.2.3 pH
The pH of each beverage was measured using a calibrated pH meter (Mettler Toledo, Ohio, United States) at 20 °C.2.4 Apparent viscosity
Rheological behaviour was determined using a rheometer (MCR301, Anton Paar GmbH, Graz, Austria) equipped with a concentric cylinder measuring attachment (Anton Paar GmbH, Graz, Austria) as previously described.24 A shear sweep was performed, measured as a function of shear rate ranging from 0.5 to 100 (1/s) at 20 °C. The apparent viscosity at 80.7 (1/s) was evaluated and used to compare the beverages.2.5 Liquid density
The liquid density (g cm−3) of the soft drinks was determined using the Anton Paar density meter DMA 4500 M (Anton Paar GmbH, Austria). The sample was loaded and measured based on the oscillating U-shaped-tube principle where the sample is excited and oscillated at an oscillation period specific for the sample mass and volume at 20 °C.2.6 Turbidity
Turbidity was measured using a turbidity meter (Thermo Scientific Eutech TN-100 Waterproof Turbidimeter, Singapore, Singapore). Samples were measured at a wavelength of 850 nm at 20 °C. A sample volume of 10 ml was used to determine turbidity ranging from 0–2000 nephelometric turbidity unit (NTU).2.7 Dispersion stability
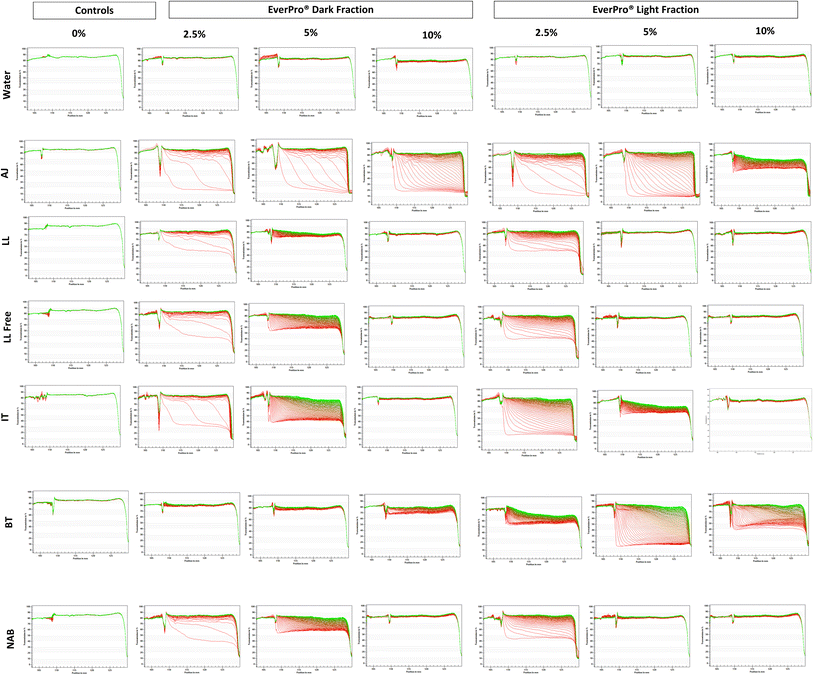
Dispersion stability was determined using an analytical centrifuge (LUMiSizer, LUM GmbH, Berlin, Germany), through phase separation based on light transmission during centrifugation. The separation rate, an indication of stability was reported. The cycle configuration involved taking 100 profiles in 10 s intervals during centrifugation at 1000 rcf for 17 min, at a wavelength of 865 nm and a temperature of 20 °C. The percentage of integrated light transmission increased over time and the separation rate in is expressed as the % transmission per minute, as previously reported.252.8 Particle size distribution
The average particle size (nm) and polydispersity index (PDI) of each soft drink was measured using dynamic light scattering technology with the Zetasizer Nano Z (Malvern Instruments Ltd, Worcestershire, UK), equipped with a 633 nm laser and set size range of 0.3 nm–10 µm. A refractive index of 1.45 and absorbance at 0.001 specific for proteins was used at 20 °C.2.9 Foam capacity and foam stability
Foaming properties of the beverages were determined by frothing 20 ml of samples using an Ultra-Turrax equipped with a S10N-10G dispersing element (IKA Labortechnik, Janke and Kunkel GmbH, Staufen, Germany), at the maximum speed of 6 for 30 s. Sample expansion was calculated using the following equations:262.10 Tribology
Tribological measurements were conducted on a rheometer MCR301 using the BC-12.7 ball-on-3-pins tribology attachment (Anton Paar GmbH, Graz, Austria) at 37 °C as described.27 The attachment contains changeable tribopairs (a glass ball and polydimethylsiloxane (PDMS) pins), which were supplied by Anton Paar (Graz, Austria). New pins were used for each test consisting of one run-in period and two repeated measurements of Stribeck curves. Before starting a test, the pin-holder and ball-holder were washed gently using a dilute detergent solution and rinsed thoroughly as well as being rinsed in acetone and dried with lab-wipes with the tribopairs. The test sequence was as followed: first the glass ball was lowered until a target force of 3 N was reached and held at that height for an equilibration period of 1 min. Furthermore, the following settings of the measurement system were selected: a logarithmic increase in sliding speed from 10−8 to 1 m s−1 was performed recording 80 data points with point duration from 1 to 10 s logarithmic, followed by a resting period of 1 min. Only the 0% and the 10% (w/w) beverages of EDF and ELF were measured using 1 ml aliquots. Three consecutive Stribeck curve measurements were performed for each sample. The first measurement served as a run-in period and was excluded from the data set. Each sample was performed in triplicate, resulting in six evaluated datasets per sample.2.11 Statistics
All analyses were performed in triplicate. Statistical analysis was performed using Origin lab pro2025b (Northampton, MA, USA). A one-way ANOVA with post hoc Tukey test (p < 0.05), principal component analysis, as well as a correlation analysis were performed.3 Results
The results are separated into two parts. First, the changes in techno-functionality due to protein fortification with EDF and ELF at different concentrations are reported. The second part reveals the degree of changes of the beverages due to either EDF or ELF, illustrated in a principal component analysis (PCA) biplot.3.1 Impact of protein fortification on techno-functionality of beverages
| Soft drink and EverPro inclusion level | pH | Apparent viscosity at 80.7 1/s (mPa s) | Density (g cm−3) | Foaming capacity (%) | Foaming stability (%) | |
|---|---|---|---|---|---|---|
| Control | Water control (0%) | 6.71 ± 0.00 a | 1.07 ± 0.13 a | 0.9985 ± 0.0000 a | 0.00 ± 0.00 a | 0.00 ± 0.00 a |
| Water D 2.5% | 8.15 ± 0.02 b | 1.27 ± 0.13 ab | 1.0061 ± 0.0001 b | 120.03 ± 3.53 b | 36.9 ± 0.57 f | |
| Water D 5% | 8.25 ± 0.05 b | 1.57 ± 0.08 bc | 1.0138 ± 0.0000 c | 128.99 ± 1.26 bc | 37.07 ± 1.38 f | |
| Water D 10% | 8.24 ± 0.05 b | 2.56 ± 0.17 d | 1.0295 ± 0.0000 d | 121.92 ± 2.44 b | 28.14 ± 1.94 d | |
| Water L 2.5% | 7.45 ± 0.01 c | 1.36 ± 0.06 ab | 1.0064 ± 0.0001 e | 134.12 ± 3.22 c | 19.89 ± 0.19 b | |
| Water L 5% | 7.45 ± 0.01 c | 1.81 ± 0.09 c | 1.0143 ± 0.0001 f | 128.57 ± 1.10 bc | 23.9 ± 1.33 c | |
| Water L 10% | 7.45 ± 0.01 c | 3.29 ± 0.10 e | 1.0304 ± 0.0001 g | 125.92 ± 7.91 bc | 33.12 ± 0.65 e | |
| Juice | Apple juice control (0%) | 3.51 ± 0.01 a | 1.23 ± 0.05 f | 1.0448 ± 0.0000 e | 26.64 ± 5.17 c | 84.85 ± 5.25 a |
| Apple juice D 2.5% | 4.26 ± 0.08 b | 1.89 ± 0.01 e | 1.0517 ± 0.0001 d | 124.83 ± 2.50 ab | 34.64 ± 1.17 bc | |
| Apple juice D 5% | 4.74 ± 0.02 c | 2.68 ± 0.47 cd | 1.0586 ± 0.0001 c | 130.68 ± 2.54 ab | 36.87 ± 1.70 bc | |
| Apple juice D 10% | 5.49 ± 0.06 d | 4.07 ± 0.25 b | 1.0728 ± 0.0010 b | 133.82 ± 2.37 a | 43.99 ± 1.93 b | |
| Apple juice L 2.5% | 4.12 ± 0.01 e | 2.04 ± 0.06 de | 1.052 ± 0.0001 d | 120.23 ± 7.04 b | 37.99 ± 6.19 bc | |
| Apple juice L 5% | 4.49 ± 0.01 f | 2.88 ± 0.28 c | 1.0594 ± 0.0002 c | 122.91 ± 3.64 b | 32.01 ± 5.59 cd | |
| Apple juice L 10% | 4.98 ± 0.02 g | 5.43 ± 0.12 a | 1.0743 ± 0.0001 a | 122.61 ± 4.90 b | 21.22 ± 1.83 d | |
| Soft drink (full sugar) | Lemon lime control (0%) | 3.30 ± 0.02 f | 1.25 ± 0.06 d | 1.0175 ± 0.0000 e | 0.00 ± 0.00 e | 0.00 ± 0.00 f |
| Lemon lime D 2.5% | 5.03 ± 0.06 d | 1.59 ± 0.05 de | 1.0249 ± 0.0001 d | 118.05 ± 3.77 cd | 38.41 ± 1.49 b | |
| Lemon lime D 5% | 5.68 ± 0.02 b | 1.86 ± 0.17 cd | 1.0325 ± 0.0001 c | 113.57 ± 1.16 d | 35.22 ± 0.62 bc | |
| Lemon lime D 10% | 6.57 ± 0.02 a | 2.81 ± 0.27 b | 1.0473 ± 0.0005 b | 142.64 ± 2.34 a | 45.36 ± 2.03 a | |
| Lemon lime L 2.5% | 4.65 ± 0.0 e | 1.80 ± 0.04 d | 1.0249 ± 0.0001 d | 124.68 ± 2.96 bc | 29.64 ± 1.49 d | |
| Lemon lime L 5% | 5.07 ± 0.01 d | 2.26 ± 0.03 c | 1.0325 ± 0.0003 c | 124.44 ± 2.22 b | 23.23 ± 2.05 e | |
| Lemon lime L 10% | 5.56 ± 0.01 c | 3.98 ± 0.27 a | 1.0482 ± 0.0002 a | 123.74 ± 2.06 bc | 34.31 ± 0.36 c | |
| Soft drink (no sugar) | Free lemon lime control (0%) | 3.30 ± 0.02 f | 1.04 ± 0.03 e | 0.9997 ± 0.0000 e | 0.00 ± 0.00 b | 0.00 ± 0.00 c |
| Free lemon lime D 2.5% | 4.95 ± 0.02 d | 1.39 ± 0.03 d | 1.0072 ± 0.0000 d | 122.17 ± 3.55 a | 36.83 ± 3.21 a | |
| Free lemon lime D 5% | 5.63 ± 0.01 b | 1.72 ± 0.06 c | 1.0149 ± 0.0001 c | 121.74 ± 2.17 a | 36.31 ± 2.59 a | |
| Free lemon lime D 10% | 6.48 ± 0.04 a | 2.54 ± 0.18 b | 1.0303 ± 0.0009 b | 127.25 ± 9.12 a | 38.61 ± 3.13 a | |
| Free lemon lime L 2.5% | 4.65 ± 0.00 e | 1.64 ± 0.08 cd | 1.0073 ± 0.0001 d | 122.46 ± 1.26 a | 17.75 ± 1.79 b | |
| Free lemon lime L 5% | 5.08 ± 0.01 c | 1.82 ± 0.12 c | 1.0153 ± 0.0001 c | 120 ± 6.14 a | 18.95 ± 1.27 b | |
| Free lemon lime L 10% | 5.60 ± 0.01 b | 3.41 ± 0.05 a | 1.0314 ± 0.0003 a | 120.29 ± 6.28 a | 35.04 ± 2.96 a | |
| Sweetened tea-based beverage | Ice tea control (0%) | 3.10 ± 0.00 a | 1.09 ± 0.13 f | 1.0177 ± 0.0000 a | 24.45 ± 2.37 e | 47.22 ± 4.81 a |
| Ice tea D 2.5% | 4.71 ± 0.01 b | 1.54 ± 0.12 e | 1.025 ± 0.0001 b | 117.3 ± 2.56 d | 36.81 ± 1.89 bc | |
| Ice tea D 5% | 5.44 ± 0.02 c | 1.93 ± 0.14 cd | 1.0326 ± 0.0001 c | 118.3 ± 3.69 cd | 30.25 ± 1.20 cd | |
| Ice tea D 10% | 6.68 ± 0.02 d | 3.09 ± 0.06 b | 1.0479 ± 0.0000 d | 122.46 ± 1.26 bcd | 34.92 ± 2.25 bcd | |
| Ice tea L 2.5% | 4.53 ± 0.01e | 1.73 ± 0.10 de | 1.0252 ± 0.0000 e | 125.02 ± 1.63 abc | 29.75 ± 3.05 d | |
| Ice tea L 5% | 4.99 ± 0.01 f | 2.25 ± 0.03 c | 1.0328 ± 0.0001 f | 128.6 ± 3.63 ab | 19.44 ± 0.71 e | |
| Ice tea L 10% | 5.56 ± 0.03 g | 4.21 ± 0.19 a | 1.0485 ± 0.0002 g | 131.16 ± 1.26 a | 39.23 ± 0.84 b | |
| Tea (unsweetened) | Black tea control (0%) | 4.88 ± 0.02 a | 1.10 ± 0.06 e | 0.9995 ± 0.0000 a | 27.95 ± 2.75 d | 63.1 ± 4.29 a |
| Black tea D 2.5% | 7.56 ± 0.01 b | 1.33 ± 0.06 de | 1.0071 ± 0.0000 b | 129.63 ± 3.39 ab | 40.55 ± 1.70 b | |
| Black tea D 5% | 7.72 ± 0.04 c | 1.57 ± 0.08 cd | 1.0144 ± 0.0005 d | 131.2 ± 3.09 a | 39.23 ± 0.84 b | |
| Black tea D 10% | 7.79 ± 0.02 d | 2.30 ± 0.04 b | 1.0303 ± 0.0001 e | 121.25 ± 4.01 bc | 35.69 ± 0.68 bc | |
| Black tea L 2.5% | 6.83 ± 0.02 e | 1.36 ± 0.07 de | 1.0073 ± 0.0001 b | 130.94 ± 1.48 a | 18.14 ± 1.81 d | |
| Black tea L 5% | 7.20 ± 0.01 f | 1.80 ± 0.14 c | 1.0152 ± 0.0000 c | 122.65 ± 3.54 abc | 30.93 ± 2.08 c | |
| Black tea L 10% | 7.32 ± 0.03 g | 3.42 ± 0.27 a | 1.0309 ± 0.0003 f | 118.72 ± 2.62 c | 30.89 ± 1.26 c | |
| Malt-based beverage | Non alcoholic beer control (0%) | 4.32 ± 0.00 a | 1.46 ± 0.05 c | 1.0156 ± 0.0000 d | 121.75 ± 0.47 bc | 39.91 ± 3.27 a |
| Non alcoholic beer D 2.5% | 6.01 ± 0.02 b | 1.77 ± 0.06 c | 1.0227 ± 0.0002 c | 124.09 ± 2.20 bc | 35.32 ± 2.39 a | |
| Non alcoholic beer D 5% | 6.64 ± 0.03 c | 2.06 ± 0.18 c | 1.0306 ± 0.0004 b | 117.39 ± 2.17 c | 26.59 ± 4.31 b | |
| Non alcoholic beer D 10% | 7.27 ± 0.01 d | 3.19 ± 0.32 b | 1.0457 ± 0.0006 a | 133.36 ± 3.58 a | 41.3 ± 1.31 a | |
| Non alcoholic beer L 2.5% | 5.26 ± 0.01 e | 1.89 ± 0.06 c | 1.0231 ± 0.0001 c | 126.35 ± 4.39 ab | 11.55 ± 0.89 c | |
| Non alcoholic beer L 5% | 5.62 ± 0.02 f | 2.30 ± 0.14 c | 1.0309 ± 0.0000 b | 118.15 ± 2.83 c | 5.52 ± 0.06 c | |
| Non alcoholic beer L 10% | 6.08 ± 0.01 g | 4.82 ± 0.33 a | 1.0464 ± 0.0003 a | 125.89 ± 1.88 ab | 25.11 ± 2.04 b |
The addition of EDF and ELF caused an increase in pH, with ELF elevating the value to a higher extend compared to ELF. Increased inclusion of either EP™ ingredient consistently elevated the pH of all beverages significantly. However, in water, the addition of 2.5% of either EDF or ELF caused an increase in pH from 6.71 (0% addition) to 8.15 and 7.45, respectively, but higher addition levels did not further raise the pH. The highest pH values (>7) in beverages fortified with 10% of EDF were observed in water (8.25) followed by BT (7.8) and NAB (7.3). The addition of 10% EDF to acidic beverages elevated their pH to 6.7 in IT, to 6.6 in LL, to 6.5 in LL Free, and to 5.5 in AJ. Addition of ELF followed the same trend in these beverages with slightly lower final pH values. A significant strong positive correlation between the addition level of either EP ingredient in IT and the resulting pH was observed (r = 0.97, p ≤ 0.05). Interestingly, the addition of ELF in AJ showed a strong correlation between addition level and pH (r = 0.97, p ≤ 0.05), but fortifying with EDF did not show significance.
Fortification with both EDF and ELF increased apparent viscosity across all beverages, with ELF addition resulting in higher viscosity values than EDF. Water showed a clear increase in viscosity with EP addition, reaching significantly higher values of 3.19 mPa s (EDF) and 4.82 mPa s (ELF) at 10% inclusion. AJ with 10% inclusion was shown to be the most viscous beverage, particularly with ELF (5.43 mPa s), followed by EDF (4.07 mPa s) with statistical significance. LL showed moderate increases, with 10% addition of EDF and ELF resulting in viscosity values of 2.81 mPa s and 3.98 mPa s, respectively. LL Free increased to 2.54 mPa s (EDF) and 3.41 mPa s (ELF) at 10% inclusion levels. NAB displayed apparent viscosities of 3.19 mPa s (EDF) and 4.82 mPa s (ELF) at 10% ingredient addition, with ELF inclusion resulting in a significantly higher value than EDF. Additionally, the inclusion of either EP ingredient showed a higher impact on IT than on BT.
All beverages demonstrated a significantly strong positive correlation (r = 0.96–1.00, p ≤ 0.05) between protein inclusion level and apparent viscosity, with the exception of EDF addition in IT where the correlation was not significant.
Fortification with both EP ingredients led to elevated density values in all beverages, while ELF showed significantly higher density values compared to EDF. Water with 10% addition of EP ingredients resulted in the lowest density values at 1.0295 g cm−3 and 1.0304 g cm−3, similar to BT at 1.0303 g cm−3 and 1.0309 g cm−3 for EDF and ELF inclusion, respectively. LL and LL Free reached similar densities at 10% inclusion with both EPs (1.05 g cm−3 (EDF) and 1.03 g cm−3 (ELF)). NAB and IT increased to 1.046 g cm−3 and 1.048 g cm−3 at 10% EP inclusion. AJ showed the highest densities at 10% addition level, with a value of 1.0728 g cm−3 determined for EDF addition, and 1.0743 g cm−3 for ELF addition.
All beverages displayed a strong positive correlation between protein inclusion and density (r = 0.99, p ≤ 0.05), revealing higher amounts of soluble solids with increasing EP fortification.
| Soft drink and EverPro inclusion level | Turbidity (NTU) | Particle size (nm) | Polydispersity index | Separation rate (% min−1) | |
|---|---|---|---|---|---|
| Control | Water control (0%) | 0.16 ± 0.04 a | n.a | n.a | 0.00 ± 0.00 a |
| Water D 2.5% | 25.7 ± 0.53 b | 298 ± 16 d | 0.59 ± 0.04 a | 0.09 ± 0.00 b | |
| Water D 5% | 42.73 ± 0.06 c | 306 ± 21 cd | 0.65 ± 0.04 a | 0.14 ± 0.00 c | |
| Water D 10% | 58.53 ± 0.58 d | 417 ± 7 b | 0.57 ± 0.02 a | 0.20 ± 0.00 d | |
| Water L 2.5% | 52.5 ± 0.17 e | 256 ± 9 e | 0.64 ± 0.19 a | 0.06 ± 0.00 e | |
| Water L 5% | 96.93 ± 0.78 f | 338 ± 9 c | 0.55 ± 0.05 a | 0.11 ± 0.00 f | |
| Water L 10% | 184.33 ± 0.58 g | 562 ± 7 a | 0.56 ± 0.08 a | 0.17 ± 0.01 g | |
| Juice | Apple juice control (0%) | 4.26 ± 0.05 c | 2560 ± 48 d | 1.00 ± 0.00 a | 0.04 ± 0.00 d |
| Apple juice D 2.5% | 625 ± 22.07 b | 4607 ± 1228 c | 0.85 ± 0.26 a | 5.26 ± 0.46 b | |
| Apple juice D 5% | 360.67 ± 9.45 d | 2560 ± 491 d | 0.22 ± 0.08 b | 6.09 ± 0.19 ab | |
| Apple juice D 10% | 466.67 ± 6.51 e | 6509 ± 420 bc | 0.83 ± 0.29 a | 5.4 ± 0.13 ab | |
| Apple juice L 2.5% | 670.33 ± 13.32 b | 8050 ± 1247 b | 1.00 ± 0.00 a | 5.82 ± 0.28 ab | |
| Apple juice L 5% | 548.67 ± 12.22 f | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 180 ± 528 a 180 ± 528 a |
1.00 ± 0.00 a | 6.18 ± 0.02 a | |
| Apple juice L 10% | 956.67 ± 41.31 a | 834 ± 46 d | 0.85 ± 0.13 a | 1.21 ± 0.54 c | |
| Soft drink (full sugar) | Lemon lime control (0%) | 0.24 ± 0.11 a | 917 ± 219 b | 0.45 ± 0.13 c | 0.02 ± 0.00 c |
| Lemon lime D 2.5% | 1768.67 ± 13.61 b | 3354 ± 324 a | 1.00 ± 0.00 a | 2.84 ± 0.30 a | |
| Lemon lime D 5% | 617.67 ± 14.01 d | 943 ± 80 b | 0.22 ± 0.03 d | 0.49 ± 0.04 b | |
| Lemon lime D 10% | 770.33 ± 62.31 c | 338 ± 14 b | 0.72 ± 0.06 b | 0.19 ± 0.02 bc | |
| Lemon lime L 2.5% | >2000 | 3977 ± 1342 a | 1.00 ± 0.00 a | 2.68 ± 0.11 a | |
| Lemon lime L 5% | 807.00 ± 37.24 c | 253 ± 11 b | 0.50 ± 0.04 c | 0.13 ± 0.00 c | |
| Lemon lime L 10% | 165.00 ± 37.36 e | 406 ± 20 b | 0.67 ± 0.03 b | 0.20 ± 0.01 bc | |
| Soft drink (no sugar) | Free lemon lime control (0%) | 0.38 ± 0.25 a | 1035 ± 110 cd | 0.47 ± 0.13 c | 0.03 ± 0.00 c |
| Free lemon lime D 2.5% | 927.00 ± 52.85 b | 3981 ± 742 a | 1.00 ± 0.00 a | 3.31 ± 0.05 a | |
| Free lemon lime D 5% | 984.00 ± 70.00 b | 1571 ± 38 c | 0.80 ± 0.04 ab | 1.82 ± 0.10 b | |
| Free lemon lime D 10% | 564.67 ± 39.80 c | 319 ± 18 d | 0.65 ± 0.07 bc | 0.21 ± 0.01 c | |
| Free lemon lime L 2.5% | >2000 | 2668 ± 671 b | 1.00 ± 0.13 a | 3.23 ± 0.14 a | |
| Free lemon lime L 5% | 267.33 ± 13.61 d | 277.07 ± 27 d | 0.54 ± 0.01 c | 0.21 ± 0.02 c | |
| Free lemon lime L 10% | 189.67 ± 32.52 d | 360 ± 46 d | 0.67 ± 0.01 bc | 0.19 ± 0.01 c | |
| Sweetened tea-based beverage | Ice tea control (0%) | 23.67 ± 0.42 d | 187 ± 31 c | 0.30 ± 0.07 bc | 0.04 ± 0.01 e |
| Ice tea D 2.5% | 980.00 ± 3.61 a | 3543 ± 365 a | 0.58 ± 0.43 abc | 4.16 ± 0.27 b | |
| Ice tea D 5% | 953.33 ± 16.86 a | 3621 ± 540 a | 0.94 ± 0.10 a | 2.76 ± 0.23 c | |
| Ice tea D 10% | 82.03 ± 2.80 d | 366 ± 47c | 0.71 ± 0.01 ab | 0.19 ± 0.01 e | |
| Ice tea L 2.5% | 1006.33 ± 54.86 a | 2427 ± 515 b | 0.15 ± 0.05 c | 5.16 ± 0.07 a | |
| Ice tea L 5% | 781.00 ± 12.12 b | 1736 ± 182 b | 0.89 ± 0.20 a | 0.98 ± 0.10 d | |
| Ice tea L 10% | 186.00 ± 4.36 c | 341 ± 4 c | 0.54 ± 0.00 abc | 0.20 ± 0.01 e | |
| Tea (unsweetened) | Black tea control (0%) | 27.47 ± 1.25 f | 309 ± 28 d | 0.68 ± 0.07 a | 0.08 ± 0.01 e |
| Black tea D 2.5% | 789.00 ± 12.29 c | 145 ± 7 d | 0.14 ± 0.01 b | 0.25 ± 0.02 e | |
| Black tea D 5% | 737.67 ± 12.06 de | 191 ± 5 d | 0.15 ± 0.02 b | 0.35 ± 0.01 de | |
| Black tea D 10% | 699.67 ± 13.01 e | 2337 ± 76 a | 0.14 ± 0.01 b | 0.90 ± 0.09 cd | |
| Black tea L 2.5% | 1641.33 ± 13.8 a | 197 ± 6 d | 0.15 ± 0.01 b | 1.31 ± 0.00 c | |
| Black tea L 5% | 765.67 ± 17.62 cd | 607 ± 120 c | 0.26 ± 0.02 b | 5.13 ± 0.29 a | |
| Black tea L 10% | 1151.33 ± 20.50 b | 1809 ± 215 b | 0.51 ± 0.19 a | 3.53 ± 0.43 b | |
| Malt-based beverage | Non alcoholic beer control (0%) | 5.05 ± 0.15 e | 465 ± 47 c | 0.55 ± 0.03 ab | 0.03 ± 0.00 e |
| Non alcoholic beer D 2.5% | 766.33 ± 9.29 a | 2472 ± 305 a | 0.16 ± 0.09 c | 1.03 ± 0.02 a | |
| Non alcoholic beer D 5% | 258.33 ± 23.16 b | 380 ± 40 c | 0.57 ± 0.04 ab | 0.15 ± 0.01 c | |
| Non alcoholic beer D 10% | 112.10 ± 33.05 d | 614 ± 88 bc | 0.51 ± 0.12 b | 0.19 ± 0.01 b | |
| Non alcoholic beer L 2.5% | 88.90 ± 1.73 d | 336 ± 27 c | 0.71 ± 0.05 a | 0.12 ± 0.0 d | |
| Non alcoholic beer L 5% | 92.97 ± 1.96 d | 355 ± 56 c | 0.6 ± 0.12 ab | 0.16 ± 0.01 c | |
| Non alcoholic beer L 10% | 163.33 ± 1.53 c | 973 ± 177 b | 0.44 ± 0.02 b | 0.21 ± 0.00 b |
Addition of both EP ingredients significantly increased turbidity in all beverages. Water samples with EP inclusion consistently exhibited low turbidity across all concentrations, with ELF causing slightly higher values than EDF. The turbidity of most beverages was dependent on concentration levels. The most substantial increases in turbidity were observed in the LL beverages fortified with ELF at 2.5%, giving values out of range (≥2000 NTU). An addition of 2.5% EDF to LL showed higher turbidity (1768 ± 14 NTU) compared to LL Free (927 ± 53 NTU). IT and BT also showed high turbidity levels, especially at 2.5% inclusion of both EP ingredients. However, ELF caused higher turbidity in IT and BT than EDF. In AJ, the fortification with EDF caused the highest turbidity value at an inclusion level of 2.5%, while fortification with ELF reached the highest turbidity at 10% addition level. The fortification of NAB with EP ingredients displayed moderate turbidity increases. The addition of ELF to NAB revealed a significantly strong positive correlation with turbidity (r = 0.95, p ≤ 0.05). Overall lower protein inclusions (2.5%) generally resulted in the most turbid samples (excluding water), while the higher inclusion level of 10% often reduced turbidity, observed in LL (EDF and ELF), LL Free (EDF and ELF), IT (EDF and ELF), BT (EDF) and NAB (EDF).
Water samples containing either EDF or ELF at all inclusion levels also showed similarly low values (≤0.2% min−1). Both EDF and ELF addition increased separation rates across all beverages with no consistent difference between the two ingredients. However, inclusion level showed a strong influence on separation behaviour. AJ showed sedimentation and a high separation rate at every EP concentration (highest in AJ ELF 5% = 6.18 ± 0.02% min−1). Both LL and LL Free followed a pattern similar to IT, where separation rates were the highest at low inclusion levels (2.5%), and significantly lower at 10%, indicating improved stability at higher protein levels. Dispersion stability was also influenced by 2.5% inclusion of both EPs in NAB. BT demonstrated a clear protein concentration-dependent effect for EDF, with a significant positive correlation observed between addition level and separation rate (r = 0.98, p ≤ 0.05).
Across all beverages (excluding water), more than half of the samples displayed their highest separation rates at 2.5% protein inclusion, whereas the lowest rates were consistently observed at 10% addition, indicating greater stability at higher protein levels.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 180 ± 529 nm, respectively). LL soft drinks displayed a peak in particle sizes at 2.5% with both EPs (2700–4000 nm), followed by a marked decrease at 10% inclusion level (320–360 nm). IT showed a similar pattern with significant differences. NAB showed no clear relationship between inclusion level and particle size, with values varying independently of EP concentration.
180 ± 529 nm, respectively). LL soft drinks displayed a peak in particle sizes at 2.5% with both EPs (2700–4000 nm), followed by a marked decrease at 10% inclusion level (320–360 nm). IT showed a similar pattern with significant differences. NAB showed no clear relationship between inclusion level and particle size, with values varying independently of EP concentration.Regarding PDI, the majority of the samples obtained a PDI > 0.5 (90% of all samples) indicating polydisperse systems with a wide particle size distribution.
Inclusion of either EDF or ELF significantly increased the foam capacity and stability in all beverages compared to controls, except for NAB, which showed minimal changes. No consistent differences were observed between EDF and ELF across the beverages. Foam capacity ranged from 113.57% to 142.64% with protein inclusion, showing no clear correlation with EP addition level in most beverages. Overall, lower and higher protein concentrations led to a similar foam capacity, indicating that foam formation was largely dependent on the presence of EP protein rather than the specific addition level. Despite the increases in foam capacity with EP fortification, foam stability remained below 50% in most samples.
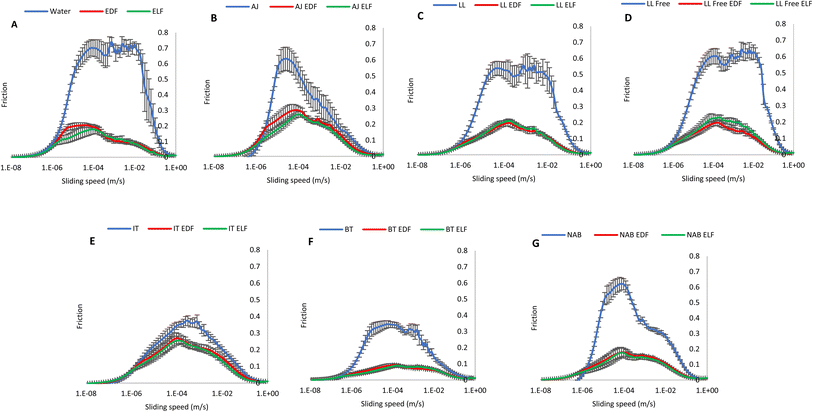 | ||
| Fig. 2 Stribeck curves of soft drinks containing EDF and ELF at 10%. Error bars indicate standard deviation. (A) Water,23, (B) apple juice, (C) lemon and lime, (D) lemon and lime sugar free, (E) ice tea, (F) black tea, (G) non-alcoholic beer. | ||
Water had the highest frictional factor compared to all other beverages, followed by the remaining beverage controls including LL Free, LL, NAB, AJ, IT and BT, showing frictional factors between 10−5 to 10−2 although NAB and AJ showed higher frictional factors between sliding speeds 10−5 and 10−4.
The inclusion of EP ingredients increased lubricating properties. ELF showed higher lubricating properties in the beverages compared to EDF. AJ exhibited the highest frictional properties when either of both ingredients were included. LL soft drinks showed similar Stribeck curves across static and boundary regimes. The addition of ELF, followed by EDF, to BT gave the highest lubricating properties. NAB showed the second highest lubricating properties followed by IT with EP inclusion.
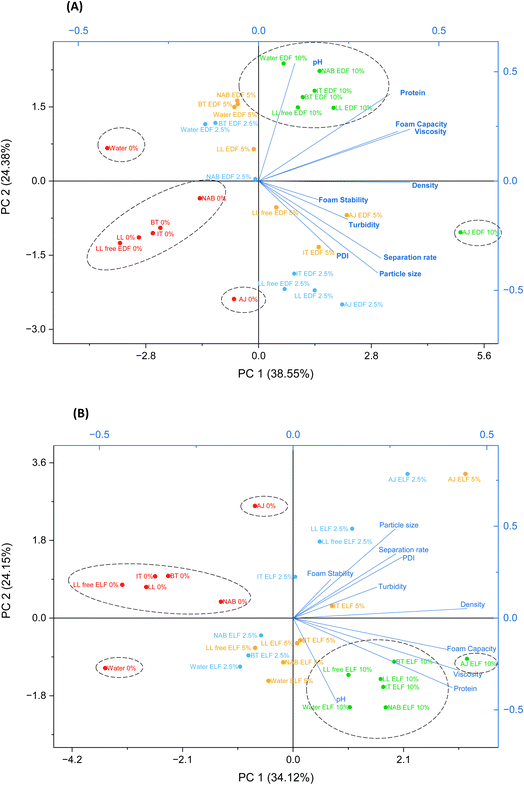
3.2 Principal component analysis biplot
Two principal component analysis (PCA) biplots were performed to visualise the changes in beverage characteristics following fortification with EDF (Fig. 3A) or ELF (Fig. 3B). The biplots captured a total variance of 62.93% for EDF fortification and 58.27% for ELF fortification.The biplot illustrating the influence of EDF (Fig. 3A) revealed five distinct groups including group 1: AJ 0%; group 2: water 0%; group 3: AJ EDF 10%; group 4: the remaining control (0%) beverages; group 5: the remaining 10% EDF beverages. Lower inclusion levels (2.5 and 5%) show the gradual modification of beverage techno functionality, with majority of samples clustering in the region associated with high dispersion instability properties. As EDF increased, a clear progression in sample distribution was observed. The distinct separation of AJ (0%) and water (0%) from the other beverages suggests notable differences in their techno-functional profiles. AJ with 10% EDF stands out distinctly due to its high density, separation rate, particle size, PDI and viscosity, of all which are positively correlating. The remaining 10% EDF beverages cluster together, sharing similar measurements for pH, foam capacity and turbidity, also positively correlating.
Similarly, the biplot in Fig. 3B (influence of ELF) also showed five groups. Group 1: AJ 0%; group 2: water 0%; group 3: AJ ELF 10%; group 4: the remaining control (0%) beverages; group 5: the remaining 10% ELF beverages. Increasing ELF concentration produced clear directional shifts in sample clustering, with higher inclusion levels (5% and 10%) associated with lower dispersion instability properties.
In both biplots, controls (indicated as “0%”) clustered distinctly apart from samples with 10% EP ingredient addition, highlighting the impact of inclusion level on techno-functionality of the beverages. Hence, ingredients both show individual changes in beverage characteristics.
4 Discussion
The protein beverage market continues to expand, fuelled by health-conscious consumers seeking clear beverage alternatives that satisfy both hydration and nutritional requirements without the heaviness of protein smoothies or shakes. This trend highlights importance of developing protein ingredients that perform well in a variety of beverage systems. Numerous studies have explored fortifying fruit juices with protein to enhance nutritional value using primarily whey protein ingredients (i.e. concentrates, isolates and hydrolysates) at levels between ∼0.8 to 6%, as well as collagen hydrolysate at a protein content of ∼2.5%.17–20,28,29 The nutritional quality of EDF and ELF have been previously characterised with both ingredients containing a high protein content (>80%). All essential amino acids meet the FAO requirements for individuals above >3 years old with the exception of lysine, with notable levels of branch chain amino acids. Dynamic in vitro digestibility of EDF also demonstrated favourable amino acid digestibility with a DIAAS of 67%, exceeding values of other plant-based proteins studied.15,24 It was further demonstrated that these ingredients possess techno-functional properties that make them promising candidates for beverage applications.24 However, those findings were limited to testing in water. The present study addresses this gap by investigating both EP ingredients at various inclusion levels (2.5%, 5% and 10%) across a range of commonly consumed beverages, determining whether certain beverage properties pose challenges for incorporation. As these ingredients are upcycled from low cost BSG, they offer sustainable and economic advantages over proteins from dedicated crops like pea, faba or soy. However, ELF's extra processing increases its cost relative to EDF. Higher inclusion levels (2.5–10% w/w; 2–8 g protein per 100 ml) cover cost effective moderate fortification to premium formulations consistent with market data showing higher protein contents and carbonated types were more expensive.24 Pictures of each beverage are displayed in Fig. 4, where clear differences can be seen between the inclusion of EDF and ELF. Fortifying beverages with EDF resulted in solutions with brown shade colours, increasing in darkness with increased addition level. The addition of ELF, on the other hand, caused a yellow shade colour at low concentrations, turning to dark orange and even brown colours at higher inclusion levels. This is, putatively, due to the lighter colour of ELF powder, where the decolourisation process likely removed many of these Maillard reaction compounds (e.g. melanoidin) originating from the brewing process23 However, residual pigments and their concentration showed to proportionally darken the beverages.The majority of soft drinks have low pH values due to the addition of acidulants and, in the case of carbonated beverages, the presence of dissolved carbonic acid.1 EverPro® ingredients possess the unique functional property of being highly soluble over a range of pH values. At pH 4, the pH closest to their isoelectric points, both EDF and ELF exhibited high protein solubility values of 82% and 80% respectively.23 The pH values of the chosen beverages (excluding water) fell within the acidic range of 3.1–4.9, as expected due to the presence of acidulants such as malic and citric acid.1 The beverages ranked by acidity from highest to lowest pH were as follows: IT > LL > LL Free > AJ > NAB > BT. The BT and NAB showed the lowest acidity amongst the beverages (excluding water). BT includes organic acids and polyphenols, such as tannins, developed from the oxidation/fermentation of tea leaves which can possess weakly acidic properties.30 Additionally, NAB contains low amounts of organic acids synthesised during the brewing process.27,31 IT exhibited the highest acidity, attributed to a combination of organic acids, polyphenols from the black tea extract, and added acidulants.1,30 Increased inclusion of both EP ingredients substantially increased the pH value closer to the neutral range. When both EP ingredients were gradually added to water, there was no change in average pH for all inclusion levels of EDF and ELF. Since proteins can act as a buffer, the addition of EP ingredients to a soft drink can interact with its acidic components. Specifically acidic amino acids with negatively charged side chains, such as aspartic and glutamic acid (∼30 g/100 g of EP protein), can bind to the hydrogen ions in an acidic environment.32 This interaction can reduce free hydrogen concentration, leading to an increase in pH within the beverage demonstrating their buffering capacity. Organic acids and ash can also contribute to a higher buffering capacity, where the slightly higher ash content observed in EDF compared to ELF may have influenced its elevated pH.23,32
The liquid density of a beverage can be influenced by the amount of total soluble solids present.33 Additionally, soluble solids in a beverage system can elevate viscosity. Both density and viscosity showed similar trends. Beverage controls containing higher sugar contents (LL, AJ) exhibited higher density and viscosity results compared to those that used artificial sweeteners (IT, LL Free), as reported previously.34,35 When sugar is replaced with artificial sweeteners, the reduction in bulk or density of the beverages leads to a significant decrease in viscosity, whereas if added at equivalent concentrations, these parameters remain comparable.36 As artificial sweeteners can be 200 to 500 times sweeter than sucrose, only very minimal quantities are required37 evident in the reduction of sugar from 4.7 g in LL to 0 g in LL Free, replaced by with aspartame and acesulfame K. Although the NAB is low in sugar, it showed the highest apparent viscosity, which is likely due to the presence of complex carbohydrates, such as beta glucan and dextrin from the malted barley, which contribute to viscosity.27 Higher concentrations of both EP ingredients resulted in increased density and viscosity values with significant positive correlations (with the exception of IT EDF for viscosity), as seen previously in fruit flavoured beverages fortified with whey protein ingredients.19 Specifically, the addition of ELF led to higher density and viscosity values compared to EDF. The PCA biplots clearly demonstrates this by the distinct separate clusters of 0% and 10% inclusions. ELF went through further processing compared to EDF, particularly a decolourisation step. Decolourisation has been shown to alter the functional properties of proteins, particularly increasing water holding capacity, thereby increasing the viscosity.38 This occurs due to an oxidation process expanding the protein structure and allowing for the more efficient binding of water.38 Similar results have been observed for decolorised soy protein isolate, which also demonstrated increased water holding capacity.39 EP originates from brewers spent grain and has been shown to consist of low molecular weight proteins and peptides.23 This lack of large and folded structure of intact proteins can cause the lower water holding capacity leading to a lower change in viscosity.40 Additionally, ELF resulted in larger particle sizes in the water solution compared to EDF, which could be attributed to the different native pH values of the ingredients in water affecting protein charge. EDF has a higher pH than ELF, thus being further away from the isoelectric point (pI ∼3.4), leading to higher repulsion forces and improved dispersion and/or hydration.23 This was shown by Dissanayake et al.36 (2013) where whey protein dispersions exhibited increased viscosity with decreasing pH values, due to increased repulsive forces at higher pHs and enhanced aggregation of protein at lower pHs.
Stability in soft drinks is essential for maintaining consistent quality, appearance, and flavour throughout the products shelf life, ensuring consumer satisfaction. Many ready-to-drink protein beverages with both animal- and plant-based protein sources on the market tend to have neutral pHs with few being acidic.24,41 Stabilizing plant-based protein ingredients in an acidic environment can be a challenge as these proteins tend to precipitate around their isoelectric point, typically within this pH range. The pH of these beverages is important as it has been found that pH values far from their isoelectric point play a key role in the clearness of beverages because of the electrostatically repulsive forces amongst unfolded proteins or aggregated proteins/peptides.42 This was seen in the current study where EP inclusion level at 2.5% in soft drinks resulted in the most acidic pH values and exhibited the highest turbidity and separation rate. On the other hand, 10% addition resulted in the highest pH values, with the clearest appearance and the lowest separation rates with little to no sedimentation. Particle size trends indicated that the largest size was observed at the 2.5% addition, with significant correlations not observed with increased inclusion levels. As seen in the PCA biplots (Fig. 3), AJ is distinctly separated at both 0% and 10% inclusions due to its high particle size and separation rate, likely attributed to its natural composition including residual pulp which increases particle size, thus affecting stability as highlighted.43 One study demonstrated that adjusting the pH to an optimum value resulted in a reduction in the mean particle size for both whey protein isolate/hydrolysate, with beverages with smaller particle sizes showing the clearest appearance.18 One method of trying to stabilize an acidic protein beverage can be the use of different polysaccharides such as beta glucan, low methoxyl pectin and chitosan that can alter the charge characteristics, potentially increasing stability.44 The presence of beta glucan in the NAB beverages may explain the low separation rates observed across the varying EP concentrations. Another study investigated the use of six different hydrocolloids (as stabilizing agents) to prevent phase separation, where particle size decreased with increasing concentrations of some stabilizers.45
While raising the pH can improve solubility and stability of proteins, it may also impact the expected tangy, acidic flavour as well as colour of a soft drink. Additionally, a higher pH can increase susceptibility to microbial spoilage as well as decrease the effectiveness of preservatives (i.e. sorbic and benzoic acid) and therefore measures such as pasteurisation or UHT need to be considered.1
Soft tribology analysis provides insight into the mouthfeel of oral processing of foods and beverages. Higher frictional coefficients are generally associated with increased surface roughness and sensations such as astringency or dryness, while lower friction reflects smoother lubrication behaviour.46 This method has been widely applied to beverages such as beer, wine, tea and soft drinks.27,47–49 In this study, ELF showed higher lubricating properties than EDF. These results are consistent with a previous study23 that reports that the presence of free amino acids and organic acids can influence frictional properties, and ELF showed reduced metabolite.50,51 Compared with their controls, NAB and AJ showed elevated frictional factors at the boundary regime believed to be associated with astringency.52 Comparable findings have been reported for other NABs, where high friction corresponds with sensory descriptors of thin or watery mouthfeel relative to alcoholic ones described as creamy and smooth.27 LL Free had higher frictional properties compared to LL with added sugar. The addition of EDF or ELF decreased frictional factors, therefore may sensorially decrease astringency and increase the viscosity of the mouthfeel. Contrary to expectations, BT and IT, typically perceived as astringent, exhibited the opposite tribological behaviour with the highest lubricating properties, even in control samples with no EP addition. One explanation is that the addition of protein may facilitate the formation of a thin, lubricating interfacial layer, improving sliding between the ball and pin, as previously proposed.49 Although polyphenols generally increase oral friction by forming precipitating complexes with proteins, the behaviour of BT and IT suggests that the nature of protein–polyphenol interactions depend on factors such as molecular size, charge, tannin flexibility and protein conformation.50 For example, large protein–polyphenol aggregates can form lubricating films,51 while tea types also differ in polyphenol composition, with green tea typically containing higher polyphenol levels than black tea.52 However, while this study shows IT exhibiting smaller particle sizes (∼350 nm) and BT showing larger particle sizes (∼2000 nm), the literature shows conflicting findings, with studies reporting that mainly smaller particle sizes have reduced frictional parameters.46,49 However, the tests in the current study were conducted without the addition of saliva which would further influence the lubrication behaviour under real oral conditions. Therefore, this discrepancy needs to be considered and highlights how a combination of factors can be involved in tribological behaviour.
5 Conclusion
Several beverage systems were evaluated following the addition of EDF and ELF at various concentrations. Clear differences were found depending on both the concentration of protein added, and the type of beverage system studied. Increased protein concentration elevated the pH across all beverage systems, leading to a trend whereby beverages became more stable and clearer at higher protein levels due to changes in solubility as a result of shifts in their isoelectric points. Moreover, protein inclusion increased both viscosity and liquid density accordingly. Lubricating properties increased with the addition of EP ingredients in all beverages, with ELF showing more effective lubrication in all beverage systems. Beverages with polyphenols, such as BT and IT, demonstrated the lowest frictional parameters regardless of protein inclusion level. Incorporating both EP ingredients into these beverages enhanced their protein content by offering a convenient ready-to-drink protein soft drink, while also promoting sustainability benefits through the use of upcycled proteins. Nevertheless, the current study highlights the need for further research to address the impact of protein addition, particularly plant-based proteins, on the stability and quality of these beverages. No single beverage system demonstrated a clear advantage over another and it should be noted that protein inclusion can potentially compromise sensory properties, which should be carefully considered during formulation. Future work should investigate the antioxidant properties of these ingredients and their interactions within the beverage systems including polyphenols, particularly in tea based beverages. Furthermore, trials to ensure microbial stability are essential for safe consumption, especially in ready-to-drink soft drinks. Continued research is key for advancing the development of nutritious, stable, safe, consumer accepted protein fortified beverages.Author contributions
Conceptualization, Aylin W. Sahin and Elke K. Arendt; methodology, Niamh Ahern; formal analysis, Niamh Ahern; resources, Aylin W. Sahin and Elke K. Arendt; data curation, Niamh Ahern; writing – original draft preparation, Niamh Ahern; writing – review & editing, Aylin W. Sahin; supervision, Aylin W. Sahin; project administration, Laura Nyhan; funding acquisition, Aylin W. Sahin and Elke K. Arendt.Conflicts of interest
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Patrick O′ Riordan reports a relationship with EverGrain LLC that includes employment. Other authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Abbreviations
| EP | EverPro™ |
| EDF | EverPro™ Dark Fraction |
| ELF | EverPro™ Light Fraction |
| AJ | Apple juice |
| LL | Lemon and lime |
| LL Free | Lemon and lime sugar free |
| IT | Ice tea (sweetened) |
| BT | Black tea (unsweetened) |
| NAB | Non-alcoholic beer |
Data availability
Data will be made available on request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fb00945f.
Acknowledgements
The stipend of Niamh Ahern was funded by the Department of Agriculture, Food and Marine (Project code: 2019R495). The authors would like to thank EverGrain Ingredients for kindly providing EDF and ELF ingredients.References
- P. R. Ashurst, R. Hargitt and F. Palmer, in Soft Drink and Fruit Juice Problems Solved, Elsevier, 2017, pp. 29–66 Search PubMed.
- A. Xie, Y. Dong, Z. Liu, Z. Li, J. Shao, M. Li and X. Yue, Foods, 2023, 3952, DOI:10.3390/foods12213952.
- S. Jeske, E. Zannini and E. K. Arendt, Plant Foods Hum. Nutr., 2017, 72, 26–33 CrossRef CAS PubMed.
- R. Giugliano, N. Musolino, V. Ciccotelli, C. Ferraris, V. Savio, B. Vivaldi, C. Ercolini, D. M. Bianchi and L. Decastelli, Nutrients, 2023, 15(10) DOI:10.3390/nu15102258.
- O. E. Mäkinen, V. Wanhalinna, E. Zannini and E. K. Arendt, Crit. Rev. Food Sci. Nutr., 2016, 56, 339–349 CrossRef PubMed.
- A. Gupta, N. Sanwal, M. A. Bareen, S. Barua, N. Sharma, O. Joshua Olatunji, N. Prakash Nirmal and J. K. Sahu, Food Res. Int., 2023, 170, 113046 CrossRef CAS PubMed.
- D. H. G. Pelegrine and C. A. Gasparetto, LWT--Food Sci. Technol., 2005, 38, 77–80 CrossRef CAS.
- K. N. Ryan, B. Vardhanabhuti, D. P. Jaramillo, J. H. van Zanten, J. N. Coupland and E. A. Foegeding, Food Hydrocoll., 2012, 27, 411–420 CrossRef CAS.
- S. M. F. Bessada, J. C. M. Barreira and M. B. P. P. Oliveira, Trends Food Sci. Technol., 2019, 53–68, DOI:10.1016/j.tifs.2019.08.022.
- S. H. M. Gorissen, J. J. R. Crombag, J. M. G. Senden, W. A. H. Waterval, J. Bierau, L. B. Verdijk and L. J. C. van Loon, Amino Acids, 2018, 50, 1685–1695 CrossRef CAS PubMed.
- K. K. Ma, M. Greis, J. Lu, A. A. Nolden, D. J. McClements and A. J. Kinchla, Foods, 2022, 594, DOI:10.3390/foods11040594.
- Q. Tang, Y. H. Roos and S. Miao, Foods, 2023, 12, 368 CrossRef CAS PubMed.
- M. Cermeño, J. V. C. Silva, M. Arcari and C. Denkel, LWT--Food Sci. Technol., 2024, 198 DOI:10.1016/j.lwt.2024.115948.
- T. J. Ashaolu, Int. J. Food Sci. Technol., 2020, 421–428, DOI:10.1111/ijfs.14380.
- A. Jaeger, N. Ahern, A. W. Sahin, L. Nyhan, J. J. Mes, C. van der Aa, I. Vrasidas and E. K. Arendt, Innov. Food Sci. Emerg. Technol., 2024, 103571 CrossRef CAS.
- M. Henchion, M. Hayes, A. M. Mullen, M. Fenelon and B. Tiwari, Foods, 2017, 6, 1–21 CrossRef PubMed.
- M. Djurić, M. Carić, S. Milanović, M. Tekić and M. Panić, Eur. Food Res. Technol., 2004, 219, 321–328 CrossRef.
- M. Goudarzi, A. Madadlou, M. E. Mousavi and Z. Emam-Djomeh, Int. J. Dairy Technol., 2015, 68, 70–78 CrossRef CAS.
- D. N. Yadav, R. K. Vishwakarma, S. Borad, S. Bansal, A. K. Jaiswal and M. Sharma, J. Food Sci. Technol., 2016, 53, 3844–3852 CrossRef CAS PubMed.
- E. Koffi, R. Shewfelt and L. Wicker, J. Food Qual., 2005, 28, 386–401 CrossRef.
- N. Ahern, T. Boeck, A. Ressa, L. Nyhan, E. K. Arendt, R. Sanchez, P. O'Riordan, S. Münch and A. W. Sahin, Innov. Food Sci. Emerg. Technol., 2025, 102, 103999 CrossRef CAS.
- N. Ahern, E. K. Arendt and A. W. Sahin, Beverages, 2023, 9, 73 CrossRef CAS.
- M. Vogelsang-O’Dwyer, A. W. Sahin, E. Zannini and E. K. Arendt, J. Sci. Food Agric., 2021 DOI:10.1002/jsfa.11230.
- M. Vogelsang-O’Dwyer, J. Bez, I. L. Petersen, M. S. Joehnke, A. Detzel, M. Busch, M. Krueger, L. Ispiryan, J. A. O’Mahony, E. K. Arendt and E. Zannini, Foods, 2020, 9(2) DOI:10.3390/foods9020230.
- D. Fox, A. W. Sahin, D. P. De Schutter and E. K. Arendt, J. Am. Soc. Brew. Chem., 2022, 80, 112–127 CrossRef CAS.
- X. Zhang, X. Du, Y. zheng Li, C. ning Nie, C. ming Wang, J. lin Bian and F. Luo, Food Nutr. Sci., 2022, 10, 2071–2081 CrossRef CAS PubMed.
- L. Nyhan, A. W. Sahin and E. K. Arendt, Eur. Food Res. Technol., 2023, 249, 167–181 CrossRef CAS PubMed.
- M. M. Al-Dabbas, K. Al-Ismail, R. A. Taleb and S. Ibrahim, Am. J. Agric. Biol. Sci., 2010, 5, 154–160 CrossRef CAS.
- M. Jabeen, I. Razzaq, N. Aslam and A. M. Zafar, Pak. J. Anal. Environ. Chem., 2022, 23, 129–140 CrossRef CAS.
- K. Saniah, M. S. Samsiah and M. Sharifah Samsiah, The application of Stevia as sugar substitute in carbonated drinks using Response Surface Methodology (Penggunaan Stevia sebagai pengganti gula di dalam minuman berkarbonat menggunakan Kaedah Gerak Balas Permukaan), J. Trop. Agric. Food Sci., 2012, 40, 23–34 Search PubMed.
- M. M. Al-Dabbas and J. M. Al-Qudsi, Effect of partial replacement of sucrose with the artificial sweetener sucralose on the physico-chemical, sensory, microbial characteristics, and final cost saving of orange nectar, Int. Food Res. J., 2012, 19, 679–683 CAS.
- S. Mittal and U. Bajwa, J. Food Sci. Technol., 2012, 49, 704–712 CrossRef CAS PubMed.
- P. R. Ashurst, Chemistry and Technology of Soft Drinks and Fruit Juices, Wiley, Hoboken, 2016 Search PubMed.
- G. L. Yao, W. He, Y. G. Wu, J. Chen, X. W. Hu and J. Yu, Int. J. Food Prop., 2019, 22, 1283–1295 CrossRef CAS.
- K. Sakai, M. Okada and S. Yamaguchi, Sci. Rep., 2022, 12(1) DOI:10.1038/s41598-022-26883-8.
- M. Dissanayake, L. Ramchandran and T. Vasiljevic, Influence of pH and protein concentration on rheological properties of whey protein dispersions, Int. Food Res. J., 2013, 20, 2167–2171 Search PubMed.
- Y. Liu, R. S. D. Toro-Gipson and M. A. Drake, J. Sensory Stud., 2021, 36(6) DOI:10.1111/joss.12704.
- C. E. Laclair and M. R. Etzel, J. Food Sci., 2010, 75(1) DOI:10.1111/j.1750-3841.2009.01400.x.
- D. Zhu, Y. Shen, L. Wei, L. Xu, X. Cao, H. Liu and J. Li, Food Chem., 2020, 328 DOI:10.1016/j.foodchem.2020.126967.
- Z. Zhang, Y. Yang, X. Huang, Z. Jin and A. Jiao, Food Hydrocoll., 2023, 139 DOI:10.1016/j.foodhyd.2023.108518.
- J. Liu, H. L. Pedersen, L. Knarreborg, R. Ipsen and W. L. P. Bredie, Food Nutr. Sci., 2020, 8, 6433–6444 CrossRef CAS PubMed.
- B. Corvera-Paredes, A. I. Sánchez-Reséndiz, D. I. Medina, R. S. Espiricueta-Candelaria, S. Serna-Saldívar and C. Chuck-Hernández, Front. Nutr., 2022, 874763, DOI:10.3389/fnut.2022.874763.
- P. H. Chong, J. Chen, D. Yin, R. Upadhyay, L. Mo and L. Han, Food Res. Int., 2019, 125 DOI:10.1016/j.foodres.2019.108642.
- L. Laguna and A. Sarkar, Tribol. - Mater. Surf. Interfaces, 2017, 11, 116–123 CrossRef CAS.
- A. Steinbach, B. Guthrie, S. Smith, T. Lindgren and S. Debon, J. Food Meas. Char., 2014, 8, 142–148 CrossRef.
- R. Upadhyay, N. Brossard and J. Chen, J. Phys. D Appl. Phys., 2016, 49(10) DOI:10.1088/0022-3727/49/10/104003.
- L. Laguna and A. Sarkar, Tribol. - Mater. Surf. Interfaces, 2017, 11, 116–123 CrossRef CAS.
- S. Prakash, D. D. Y. Tan and J. Chen, Food Res. Int., 2013, 1627–1635, DOI:10.1016/j.foodres.2013.10.010.
- B. Kew, M. Holmes, M. Stieger and A. Sarkar, Food Hydrocoll., 2021, 116 DOI:10.1016/j.foodhyd.2021.106636.
- M. H. Alu’datt, T. Rababah, M. N. Alhamad, G. J. Al-Rabadi, C. C. Tranchant, A. Almajwal, S. Kubow and I. Alli, Crit. Rev. Food Sci. Nutr., 2018, 1–10, DOI:10.1080/10408398.2017.1391169.
- A. Chojnicka, S. De Jong, C. G. De Kruif and R. W. Visschers, J. Agric. Food Chem., 2008, 56, 1274–1282 CrossRef CAS PubMed.
- A. Rana, M. Samtiya, T. Dhewa, V. Mishra and R. E. Aluko, J. Food Biochem., 2022, 46(10), e14264, DOI:10.1111/jfbc.14264.
| This journal is © The Royal Society of Chemistry 2026 |