Open Access Article
Open Access ArticleRepurposing grape pomace as a functional ingredient: in-depth analytical characterization and in vitro bioaccessibility evaluation of the phenolic fraction
Filippo Peruccaccia,
Luciano Mangiapelo,
Shahd Ali,
Francesca Blasi ,
Federica Ianni
,
Federica Ianni * and
Lina Cossignani
* and
Lina Cossignani
Department of Pharmaceutical Sciences, University of Perugia, 06126 Perugia, Italy. E-mail: federica.ianni@unipg.it
First published on 22nd April 2026
Abstract
The study investigated the chemical profile of red grape pomace varieties and evaluated various extraction techniques for the recovery of polyphenols. Conventional maceration (MAC) and non-conventional extractions [ultrasound-assisted (UAE), ultraturrax-assisted (UTAE), enzyme-assisted (EAE)] were comparatively assessed on pomace from Merlot variety. UAE-based probe revealed as the most efficient, yielding higher concentrations of bioactives while preserving antioxidant activity. The optimal conditions were applied to pomaces from different grape varieties. Spectrophotometric and chromatographic analyses highlighted significant variation in polyphenol concentrations, with Merlot showing the highest while Sangiovese the lowest content of compounds. HPLC-PDA analysis indicated catechin, epicatechin and peonidin-3-O-glucoside as the most abundant (in the range 96.82–322.15, 115.81–361.16 and 111.72–402.36 µg g−1, respectively), followed by quercetin (41.97–115.35 µg g−1), gallic acid and procyanidin B2 (22.64–43.78 and 32.01–70.20 µg g−1, respectively). Other polyphenols were present in lower concentrations (0.19 to 33.95 µg g−1). Multivariate statistical analysis grouped the samples into three distinct clusters based on their bioactive profiles: cluster 1, comprising Merlot-3, Sagrantino-1 and Sangiovese-1; cluster 2 comprising Merlot-1, Merlot-2 and Cabernet-1; cluster 3 with Cabernet-2 as the sole member. A bioaccessibility study on both raw grape pomace (from Cabernet Sauvignon and Merlot) and their relative polyphenol extracts revealed a different pattern of bioactives during simulated gastrointestinal digestion. In some cases, gastric and intestinal digestion enhanced the concentration of specific compounds, resulting in bioaccessibility values exceeding 100%. The obtained findings suggest that the sample matrix may protect bioactives and promote their release during digestion, whereas isolated extracts may require tailored formulation strategies to preserve compound stability.
Sustainability spotlightThis works has been developed within the framework of promoting sustainability, focusing on the recovery of a food waste, namely grape pomace, and its valorisation for the production of functional foods and nutraceuticals. Grape pomace is widely recognized as one of the major agro-industrial wastes generated worldwide, amounting to nearly 8.5 million tons per year. If non properly managed, this by-product causes environmental pollution and its disposal generates significant economic losses. For these reasons, the extraction of bioactive compounds represents an important opportunity given the well-documented health-promoting properties of these constituents and their potential applications in several fields. Accordingly, this work is aligned with the UN's Sustainable Development Goals 3 (Good Health and Well-Being) and 12 (Responsible Consumption and Production). |
1. Introduction
Winemaking processes generate large amounts of grape pomace, a by-product rich in nutrients and bioactive compounds with potential health benefits.1 Great attention is paid to the polyphenolic fraction which includes phenolic acids, flavonols, flavanols, stilbenes and anthocyanins.2 These molecules stand out for their antioxidant and anti-inflammatory properties and for their potential role in preventing chronic diseases such as cardiovascular and metabolic diseases, and cancer.2Several factors may induce variation in the polyphenolic content of grape pomace, including grape variety, processing conditions, and extraction methods.1,3 Over the past few decades, considerable research has been devoted to the exploration of extraction techniques aimed at obtaining wholesome, natural food components to be used as substitutes for artificial antioxidants and food preservatives, or functional supplements.4,5
Although no standardized approach has yet been established, both traditional processes and non-conventional methods are often applied and compared, with the latter receiving particular attention for their major alignment with green chemistry principles.6 The overall goal of a suitable extraction process is to produce high-quality extracts with minimum structural modification of targeted compounds.
Beyond its significant polyphenolic content, grape pomace possesses functional properties that make it a valuable ingredient for a wide range of food products. Its richness in dietary fiber (accounting 50–75% of its dry matter), as well as the presence of proteins and healthy fats, contribute to improve food texture and sensory qualities. These properties make grape pomace itself a promising candidate for the development of functional foods and nutraceuticals that meet the needs of a health-conscious market.7
However, some limitations including poor solubility, stability under environmental conditions and low bioavailability in the human gastrointestinal tract still hinder the practical application of grape pomace and its polyphenolic extracts in the food industry.8
All these factors are closely interconnected: stability, natural chemical form in food, and absorption characteristics of polyphenols and other functional components directly influence their bioaccessibility and bioavailability. It is enough to consider that, in their glycosidic form, these phytochemicals are not readily absorbed within the upper gastrointestinal tract. Likewise, interaction with proteins or dietary fiber can promote the formation of insoluble protein-polyphenol complexes, thereby affecting their bioaccessibility and absorption.
As emphasized by Hu and co-workers,9 once ingested, phytochemicals undergo processes similar to pharmaceuticals, including liberation, solubilization, absorption, distribution, metabolism, and excretion.
From this perspective, assessing bioaccessibility, defined as the fraction of compounds released from the food matrix in a form that can readily be absorbed, is the first crucial action in the development of bioactive-rich foods or functional formulations. The concept encompasses digestive transformations of food occurring in the gastrointestinal tract to release the fraction of phytochemicals potentially absorbable into the bloodstream and ultimately utilized by tissues and organs.
The present study focused on the characterization of the phenolic profile of different red grape pomace varieties (including Cabernet Sauvignon, Merlot, Sagrantino and Sangiovese) provided by Umbrian wineries. Extracts were obtained by comparing conventional (maceration, MAC) and non-conventional techniques [ultrasound-assisted extraction (UAE), ultraturrax-assisted extraction (UTAE), enzyme-assisted extraction (EAE)].
The most promising extracts were selected following a preliminary screening and subjected to an extensive analytical characterization in terms of antioxidant properties and phenol composition determined by HPLC-PDA. Accordingly, aimed at evaluating the stability of the extracted polyphenols in a simulated gastrointestinal environment, in vitro bioaccessibility was performed and changes in the phenolic composition were analyzed chromatographically. Further insights were gained by applying the bioaccessibility study directly to the starting grape pomaces, allowing assessment of the efficiency of polyphenol release.
Despite recent advances, systematic comparisons of multiple extraction techniques applied to grape pomace matrices are still limited. In this context, the present study provides a structured comparative evaluation of conventional and non-conventional extraction techniques, followed by a more in-depth comparison between bath- and probe-based UAE. This was coupled with a comprehensive characterization, enabling a clearer assessment of their efficiency in recovering phenolic compounds. Furthermore, a distinctive aspect of this work is the evaluation of polyphenol bioaccessibility performed in both a simplified (extract) and native complex (raw grape pomace) systems. This approach allows the direct assessment of the impact of matrix complexity on polyphenol release and stability during simulated gastrointestinal digestion, providing more realistic insights into their potential bioavailability.
In the context of the emerging consumer awareness towards a wholesome life and access to healthier products, the findings of this study pave the way for the development of formulation strategies when it comes to polyphenolic extracts. Moreover, the possibility of repurposing grape pomace itself as an ingredient for functional foods and nutraceuticals points to the increased attention on environmental sustainability, alongside benefits to human health.
2. Materials and methods
2.1. Samples, chemicals and reagents
Pomace of red wine grapes (Vitis vinifera L.) were provided by two local wineries located in the province of Perugia (Central Italy). Merlot (M1 and M3) and Cabernet Sauvignon (C1) were obtained from Chiorri Azienda Agricola, while Merlot (M2), Cabernet (C2), Sagrantino (ST1) and Sangiovese (SG1) varieties were supplied by Tenute Baldo Agricola.All samples obtained after winemaking fermentation and comprising residual grape seeds, stems, pulp and skins were freeze-dried GP (VirTis Benchtop 2 K Freeze Dryer, SP Industries Inc., Gardiner, NY, USA; chamber parameters: condenser temperature, −70 °C; pressure value, 250 mTorr).10
Folin–Ciocalteu reagent, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) diammonium salt (ABTS), 2,4,6-tris(2-pyridyl)-s-triazine (TPTZ), 2,2-diphenyl-1-picrylhydrazyl (DPPH), gallic acid, 6-hydroxy-2,5,7,8-tetramethylchromane-2-carboxylic acid (Trolox), methanol (MeOH), ethanol (EtOH), acetic acid, formic acid, sodium thiosulphate, protocatechuic acid, (+)-catechin, procyanidin B2, chlorogenic acid, caffeic acid, p-coumaric acid, ferulic acid, resveratrol, and quercetin were provided by Sigma-Aldrich (Milan, Italy). (−)-epicatechin, quercetin-3-O-glucoside, kampferol-3-O-glucoside, and peonidin-3-O-glucoside were provided by Extrasynthese (Genay, France).
2.2. Extraction of polyphenols from grape pomace
Based on a preliminary study,10 Ultrasound-assisted extraction (UAE, sonicator bath Model AU-65, Argolab, Carpi, Italy) performed for 30 min with an ethanol/water (80/20, v/v) mixture supplemented with 1% (v) acetic acid was identified as the reference process. Merlot was selected as test variety for the optimization of the extraction process. A sample/solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 (g mL−1) was used.
20 (g mL−1) was used.
On this basis, conventional extraction of polyphenols from Merlot grape pomace was carried out by maceration, using a hydroalcoholic mixture of ethanol and water (10/90, v/v) with 1% (v) acetic acid (AcOH) as additive. The same extraction solvent was also used to compare the non-conventional techniques UAE, carried out for 30 min, and ultraturrax-assisted extraction (UTAE, Silent crusher M, by Heidolph Instruments GmbH & Co. KG, Schwabach, Germany), carried out for 2 min. In addition, both MAC- and UTAE-based extractions were carried out in two consecutive steps (30 × 2 min and 2 × 2 min, respectively).
In addition, plain water was evaluated as the extraction solvent under maceration (MAC) for 4 h. The same experimental conditions were applied for enzyme-assisted extraction (EAE) using two different enzyme preparations, namely Celluclast® (from Trichoderma reesei) and Viscozyme® (from Aspergillus sp), kindly provided by Novozymes A/S (Bagsvaerd, Denmark). A third enzymatic system was prepared by combining the two enzyme systems (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50, w/w).
50, w/w).
Under the optimized conditions, UAE using a probe system (UP200Ht, Hielscher Ultrasonics GmbH, Teltow, Germany) was also evaluated in comparison with sonicator bath. In both cases, the solvent system consisted of EtOH/H2O, 80/20 (v/v) with 1% acetic acid and extraction was carried out for 15 min in three consecutive steps with solvent renewal. A sample-to-solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 (g mL−1) was applied for the first extraction, while a ration of 1
20 (g mL−1) was applied for the first extraction, while a ration of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (g mL−1) for the second and third extraction. Extractions were stopped every 5 minutes and the probe or bath was left to cool for 90 s in order to maintain temperature within 35–40 °C.
10 (g mL−1) for the second and third extraction. Extractions were stopped every 5 minutes and the probe or bath was left to cool for 90 s in order to maintain temperature within 35–40 °C.
Regardless of the extraction method, the obtained extracts were dried under vacuum and subsequently resuspended for further evaluations.
2.3. Evaluation of the total phenol content and in vitro antioxidant activity
Spectrophotometric assays were carried out to evaluate the total phenol content (TPC) and the in vitro antioxidant (AOX) activity of the polyphenol extracts obtained from the investigated grape pomaces. All analyses were performed according to previously described protocols.10,11TPC was determined using the Folin–Ciocalteu's phenol reagent. Absorbance was measured at 765 nm, and quantification was based on a calibration curve prepared with gallic acid. Results were expressed as mg of gallic acid equivalents per gram of grape pomace (mg GAE per g).
The total antioxidant activity of the extracts was evaluated with both the free radical scavenging activity (ABTS and DPPH assays) and the reducing capacity (ferric reducing antioxidant power, FRAP assay).
For ABTS and DPPH assays, the absorbance was measured at 734 and 517 nm, respectively, while for the FRAP assay, the absorbance was measured at 593 nm. For all assays, Trolox was used as the reference standard to build-up calibration curves. Results were expressed as mg of Trolox equivalent per gram of grape pomace (mg TE g−1). All UV-Vis measurements were recorded with a LAMBDA 20™ UV-Vis spectrophotometer (PerkinElmer, Inc., Waltham, MA, USA).
2.4. Total anthocyanin content
Total anthocyanin content (TAC) was determined using the pH differential method by measuring the UV-Vis absorbance of the extract at both a highly acidic pH and a less acidic or neutral pH. Samples were diluted in a pH 3.5 buffer solution made of Na2HPO4 (0.2 M) and citric acid solution (0.1 M) and in a pH 0.6 solution of HCl (2%). The mixture was incubated in the dark for 15 min. Absorbance was measured at 520 and 700 nm, and the difference between the two pH conditions was used to estimate anthocyanin concentration. Results were expressed as milligrams of malvidin-3-glucoside (M3G) equivalents per kilogram of dry weight (mg M3G kg−1 dw).2.5. HPLC-PDA analysis
The quali-quantitative analysis of the phenolic profile was performed by applying a previously optimized method.12 A Waters® Acquity Arc HPLC (High Performance Liquid Chromatography, Waters Corporation, Milford, MA, USA) equipped with a low-pressure mixing quaternary solvent manager-R (QSM-R) as a high-pressure pump, a sample manager flow through needle-R (FTN-R) using a direct-injection mechanism, a column heather/cooler, and a Waters 2998 photodiode array (PDA) detector was used. Waters® Empower 3R Chromatography Data Software (CDS) was employed for data management. Phenolic compound separation and quantification were performed using a X-Bridge column (150 × 4.6 mm, 5 µm, Waters®). The gradient elution program employed eluent A [water containing 0.1% formic acid (v)] and eluent B (MeOH) as follows: 0 min − 100% A, 21 min − 70% A, 27 min − 60% A, 33 min − 40% A, 40 min − 100% A. The flow rate was set at 0.7 mL min−1, and the column temperature was maintained at 25 °C.Samples obtained from the bioaccessibility assessment were filtered with a 0.22 µm nylon syringe filter (ClearLine®, Bernolsheim, France) prior to injection.
Identified phenolic compounds were quantified using the external standard method. Detection wavelengths were selected according to compound class: (+)-catechin, (−)-epicatechin, gallic acid, procyanidin B2 and protocatechuic acid were evaluated at 278 nm; chlorogenic, caffeic, p-coumaric, ferulic acids, and resveratrol were measured at 320 nm; quercetin-3-O-glucoside, kampferol-3-O-glucoside, and quercetin were determined at 360 nm; peonidin-3-glucoside at 520 nm.
2.6. In vitro bioaccessibility – INFOGEST protocol
The in vitro bioaccessibility on both raw grape pomace and polyphenol-rich extracts from both Merlot and Cabernet Sauvignon was assessed according to the harmonized INFOGEST in vitro static digestion protocol.13 Briefly, 500 mg of raw grape pomace or 60 mg of polyphenol extract obtained from grape pomace (500 mg) were mixed with 2 mL of simulated salivary fluid, 12.5 µL of CaCl2 (0.3 M), and 750 µL α-amylase (from Bacillus licheniformis, Merck Life Science Milan, Italy; 500 U mg−1 protein). The volume was adjusted to 5 mL with distilled water, and the mixture incubated at 37 °C for 2 min in a thermostated incubator (Ski4; Argolab; Modena, Italy), under continuous shaking at 100 rpm to simulate the oral digestion.Then, 4 mL of simulated gastric fluid containing 0.5 mL pepsin (from porcine gastric mucosa, Merck Life Science Milan, Italy; 250 U mg−1 protein) and 2.5 µL of CaCl2 (0.3 M) were added. The pH value was adjusted to 3.0 using 6 M HCl and the volume was brought to 10 mL with distilled water. The gastric phase lasted in the incubator at 100 rpm for 120 min at 37 °C.
For the intestinal phase, 4.25 mL of simulated intestinal fluid at pH = 7.0 (adjusted with 1 M NaOH) was added to the chyme, together with 2.5 mL pancreatin (from porcine pancreas, Merck Life Science Milan, Italy; 4 × USP specifications), and 1.25 mL bile salts (bile extract porcine, Merck Life Science Milan, Italy; 65 mg mL−1). The total volume was adjusted to 20 mL, and the samples were incubated for 2 h at 37 °C with shaking (100 rpm). The intestinal digest was finally centrifuged (at 5000 rpm for 15 min), and the supernatant was stored. Liquid aliquots were withdrawn after both the gastric (GP-GD, grape pomace-gastric digested and GPE-GD, grape pomace extract-gastric digested) and the small intestinal (GP-ID, grape pomace-intestinal digested and GPE-ID, grape pomace extract-intestinal digested) digestion phases, filtered, and analyzed by HPLC-PDA to evaluate the phenolic profile.
Two control samples of raw grape pomace, from the two varieties, were prepared under the same digestion conditions as provided by the protocol and described above (in terms of volumes, incubation times and temperature), but using distilled water without the addition of enzymes, salts, bile salts, or pH adjustment. Aliquots were taken from both control samples under gastric [GP-Ctrl(G), grape pomace-control (Gastric)] and intestinal [GP-Ctrl(I), grape pomace-control (Intestinal)] conditions. The results of the bioaccessibility study were expressed as the recovery percentage of each identified polyphenol, as determined by HPLC-PDA, relative to its concentration in the undigested grape pomace extract.
2.7. Color analysis
Both the selected grape pomaces and extracts were analyzed for color using a tristimulus colorimeter (Eoptis, Mod. CLM-196 Benchtop, Trento, Italy). The results were expressed in the CIELAB color space, which encompasses the full spectrum of human color perception and is based on the opponent-process theory of vision. In this system, color is expressed in three dimensions: L* represents lightness, a* the red-green axis, and b* signifies the yellow-blue axis.2.8. Statistical analysis
All analyses were performed in triplicate (n = 3), with results expressed as the mean ± standard deviation (SD). Statistical significance was assessed using one-way analysis of variance (ANOVA), followed by Tukey's HSD (honestly significant difference) post hoc test. A p-value less than 0.05 was considered statistically significant. GraphPad PRISM version 9.3.1 (GraphPad Software, Boston, MA, USA) and Excel Version 16.91 (Microsoft, Redmond, WA, USA) for Windows were used for statistical analysis and graph generation. Pearson's correlation was used to assess the relationship between total phenolic content and antioxidant capacity.Chemometric analysis was obtained using the MetaboAnalyst 6.0 web platform on the normalized dataset. Pareto scaling based on the square root of the standard deviation was applied as the scaling factor, providing an intermediate scaling effect between the no scaling and unit variance scaling.
3. Results and discussion
3.1. Preliminary screening of the extraction conditions on Merlot variety
Previous findings highlighted UAE (ultrasound-assisted extraction) as the elective technique for the extraction of polyphenols from grape pomace.10 The study emphasized the critical role of water content in ethanol and the positive influence of extraction time. Accordingly, based on a preliminary study,10 UAE-based extraction with an ethanol/water (80/20, v/v) mixture supplemented of 1% (v) acetic acid, for 30 min, was adopted as the reference protocol in this preliminary evaluation. Based on this, different extraction procedures (Section 2.2) were adopted, with the aim of focusing on green and sustainable extraction conditions. Grape pomace obtained from the Merlot variety was selected as the model sample for preliminary extraction optimization. This choice was supported by our previous study, which focused on the optimization of an analytical workflow for grape pomace polyphenol characterization, where the Merlot sample provided the best results in terms of TPC (Total Phenol Content) and antioxidant activity.10The different extraction approaches were compared using the spectrophotometric Folin–Ciocalteau assay for the measurement of the TPC. An overview of the extraction conditions applied along with the corresponding TPC results is shown in Table 1.
| Sample | Extraction mode | Extraction solvent | Time | TPC (mg GAE per g) |
|---|---|---|---|---|
| a Different letters in each column indicate significant differences with p-value < 0.01. The symbols *, § and † indicate significant differences with p-value < 0.05. UAE: ultrasound-assisted extraction; MAC: maceration; UTAE: ultraturrax-assisted extraction; EAE: enzyme assisted extraction. TPC: total phenol content; GAE: gallic acid equivalents. | ||||
| A | UAE | EtOH/H2O (80/20) + 1% AcOH | 30 min | 23.45 ± 1.06a |
| B | UAE | H2O/EtOH (90/10) + 1% AcOH | 30 min | 18.01 ± 0.47b,* |
| C | MAC | H2O/EtOH (90/10) + 1% AcOH | 30 min | 17.86 ± 0.11b,* |
| D | UTAE | H2O/EtOH (90/10) + 1% AcOH | 2 min | 16.33 ± 1.02b,c |
| E | MAC | H2O/EtOH (90/10) + 1% AcOH | 2 × 30 min | 15.51 ± 0.16c,d,* |
| F | UTAE | H2O/EtOH (90/10) + 1% AcOH | 2 × 2 min | 16.97 ± 0.40b,d |
| G | MAC | H2O | 4 h | 7.45 ± 0.06e,§ |
| H | EAE (celluclast) | H2O (celluclast 3.6%) | 4 h | 8.74 ± 0.16e,f,† |
| I | EAE (viscozyme) | H2O (viscozyme 3.6%) | 4 h | 11.18 ± 0g,† |
| L | EAE (celluclast/viscozyme) | H2O (celluclast/viscozyme 3.6%) | 4 h | 9.98 ± 0.07f,g,§ |
The results indicated that the ethanol-rich hydroalcoholic mixture used for sample A yielded the extract with the highest phenolic content after 30 minutes of UAE (sonicator bath), which was further confirmed by the significant difference compared to all other samples (p < 0.01). Slightly lower TPC values were observed for sample B (18.01 mg GAE per g), obtained with a water-rich hydroalcoholic mixture, followed by samples C, F, D and E. However, as indicated by the Tukey's multiple test, no statistically significant differences were observed for samples B, C, D and F (p > 0.05), while sample E showed only slight differences, with significance indicated at p < 0.05. This result is particularly interesting, as it highlights how both conventional and non-conventional extraction techniques produced similar outcomes when using excess of water as the extraction solvent.
The second group of samples (G–L) shared similar results, showing approximately halved TPC values, indicating how the use of plain water dramatically affected the extraction efficiency of polyphenols. This trend was consistent for both with MAC (maceration) and EAE (enzyme-assisted extraction), despite the latter being generally recommended for the many advantages it offers.14
3.2. UAE bath vs. probe
In this part of the study the effects of UAE methods, specifically bath and probe, were investigated with the aim of enhancing the extraction efficiency of phenolic compounds from grape pomace. The solvent system consisted of ethanol/water (80/20, v/v) containing 1% (v) acetic acid, with some modifications from the previous method. Specifically, the total extraction time was set to 15 min, applied in three consecutive steps with solvent renewal (see Section 2.2 for details). Both Merlot and Cabernet Sauvignon grape pomace were tested under these conditions: TPC, AOX (antioxidant activity measured by ABTS, DPPH and FRAP assays) and TAC (total anthocyanin content) were evaluated (Table 2).| Extraction | TPC (mg GAE/g) | ABTS (mg TE g−1) | DPPH (mg TE g−1) | FRAP (mg TE g−1) | TAC (mg M3G g−1) | |
|---|---|---|---|---|---|---|
| a Different letters in each column indicate significant differences with p-value < 0.01. The symbols *, §, † and ‡ indicate significant differences with p-value < 0.05. AOX: antioxidant activity (measured by ABTS, DPPH and FRAP assays); TAC: total anthocyanin content. | ||||||
| Cabernet sauvignon | Probe – 1st | 28.35 ± 0.55a,* | 57.39 ± 1.96a | 44.19 ± 2.77a | 50.49 ± 0.68a | 1.15 ± 0.05a |
| Probe – 2nd | 4.80 ± 0.21b,§ | 9.51 ± 0.10b,c,* | 7.17 ± 0.12b,* | 8.64 ± 0.07b,* | 0.26 ± 0.00b,* | |
| Probe – 3rd | 2.19 ± 0.11c,§ | 4.21 ± 0.40d | 2.06 ± 0.05c,* | 3.46 ± 0.36c | 0.11 ± 0.00c,d | |
| Bath – 1st | 12.67 ± 0.92d,† | 33.28 ± 2.30e | 19.44 ± 1.20d | 21.19 ± 2.22d | 0.69 ± 0.03e,§ | |
| Bath – 2nd | 10.30 ± 0.97e,† | 23.78 ± 0.94f | 19.68 ± 1.40d | 22.36 ± 1.21d | 0.56 ± 0.02f | |
| Bath – 3rd | 3.36 ± 0.24b,c,‡ | 7.14 ± 0.06b,d,§ | 7.46 ± 0.66b,* | 7.85 ± 0.14b,e | 0.22 ± 0.02b,g | |
| Merlot | Probe – 1st | 30.79 ± 0.01f,* | 59.59 ± 0.31a | 34.99 ± 2.61e | 73.00 ± 0.01g | 0.63 ± 0.01e,f |
| Probe – 2nd | 5.78 ± 0.10b,‡ | 11.23 ± 0.13c,§ | 7.33 ± 0.63b,* | 12.95 ± 0.15f | 0.17 ± 0.01b,d | |
| Probe – 3rd | 2.48 ± 0.03c,§ | 4.73 ± 0.16d | 2.34 ± 0.02c,* | 5.29 ± 0.09c,e,* | 0.05 ± 0.00c | |
| Bath – 1st | 24.82 ± 1.17g | 47.31 ± 0.58g | 22.02 ± 0.51d | 57.30 ± 0.22h | 0.60 ± 0.02f,§ | |
| Bath – 2nd | 5.98 ± 0.16b,‡ | 11.23 ± 0.14c,§ | 7.45 ± 0.10b,* | 13.10 ± 0.23f | 0.16 ± 0.01d,g,*,† | |
| Bath – 3rd | 2.77 ± 0.06b,c | 5.00 ± 0.07d,* | 2.91 ± 0.01c,* | 6.00 ± 0.05b,c | 0.07 ± 0.00c,† | |
The results showed that UAE probe extraction yielded higher concentrations of bioactive compounds, while also preserving the antioxidant activity. This aligns with the features of probe-type systems, whose higher efficiency arises from the direct application of ultrasonic energy to the sample with minimal energy losses.15 However, despite the enhanced cell disruption and the facilitated release of phytochemicals, probe systems may also induce excessive cavitation resulting in increased number of bubbles, inter-bubble collision phenomena, deformation and collapse, which decrease the impact of bubble implosion. Furthermore, the layer of cavitation bubbles formed around the probe tip may acts as a shield, hindering the transmission of ultrasonic energy into the extraction medium.16 All these events can result in reduced extraction yields and lower antioxidant activities of the phytoextract.17
In general, although the superiority of probe-based systems compared to ultrasonic baths has not always been confirmed, in terms of process efficiency and energy losses, the ultrasonic probe-based system can still be considered preferable for extraction purposes.
As shown in Table 2 and Fig. S1 (SI), the first extraction step was the most effective in isolating the polyphenolic fraction, both with probe and bath systems, and for both evaluated varieties (Cabernet Sauvignon, Fig. S1A; Merlot, Fig. S1D). In contrast, during the second and third extraction (Fig. S1B, C and E, F), a slight improvement was gained with the bath compared to the probe, confirming the stronger ultrasonic effect of the latter. This greater efficiency of the probe reduced the recovery of bioactives in subsequent extractions, making their contribution less significant, and allowing for high phenol recovery in short extraction time.
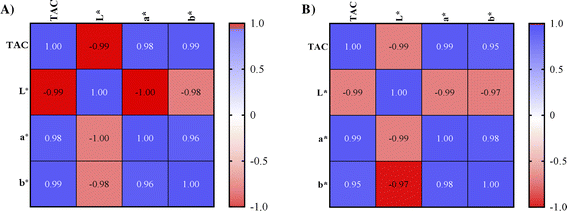
Strong positive correlations were observed between TPC and the antioxidant activity as showed by the Pearson's correlation coefficient (r), regardless of the extraction mode. This outcome strengthens the ability of the polyphenolic pool to scavenge and reduce free radicals (Fig. 1). This positive relationship is consistent with previous findings.10,12
Color analysis of the extracts obtained from the two extraction techniques indicated important color variation according to the extraction step. The lightness (L*) scale, ranging from 0 (black) to 100 (white), showed values between 23.07 (lowest) and 83.05 (highest) (Table 3). Both extremes were recorded for Merlot: the lowest value corresponded to the first extraction by probe, while the highest was obtained during the third extraction by bath. Overall, a significant increase in L* was observed across consecutive extraction steps, with the third step showing the highest values, likely reflecting the progressive depletion of phenolic compounds from the matrix. This trend paralleled the reduction in both a*, which ranges from greenish (−a*) to reddish (+a*), and b*, which ranges from bluish (−b*) to yellowish (+b*). Accordingly, regardless of the extraction technique, the first extract was characterized by the highest redness, consistent with its higher phenolic content. In subsequent steps, the values became smoother, indicating only a slight tendency toward red. With respect to b*, the color measurements revealed a stronger yellowish component in the first extraction, which gradually decreased toward neutrality in the following steps. Statistically significant differences (p-value < 0.01) were observed between the two grape varieties for L*, a*, and b*, reflecting their different polyphenol contents. The only exception was the a* parameter in the second extraction of the Merlot variety, where probe and bath treatments did not differ significantly.
| Extraction | L* | a* | b* | ||
|---|---|---|---|---|---|
| a All values are significantly different (p-value < 0.01). The symbol * indicates non-significant differences (p-value > 0.05). L*: lightness; a*: greenish-reddish; b*: bluish-yellowish. | |||||
| Cabernet sauvignon | Probe – 1st | 29.13 ± 0.01 | 54.95 ± 0.02 | 39.32 ± 0.05 |  |
| Probe – 2nd | 71.53 ± 0.01 | 18.24 ± 0.01 | 6.07 ± 0.04 |  |
|
| Probe – 3rd | 81.2 ± 0.01 | 8.51 ± 0.00 | 2.03 ± 0.03 |  |
|
| Bath – 1st | 44.75 ± 0.01 | 45.01 ± 0.03 | 21.88 ± 0.02 |  |
|
| Bath – 2nd | 59.03 ± 0.01 | 31.12 ± 0.01 | 11.71 ± 0.03 |  |
|
| Bath – 3rd | 77.16 ± 0.01 | 12.15 ± 0.01 | 3.17 ± 0.02 |  |
|
| Merlot | Probe – 1st | 17.31 ± 0.01 | 32.54 ± 0.03 | 25.13 ± 0.03 |  |
| Probe – 2nd | 62.01 ± 0.01 | 13.62 ± 0.02* | 16.06 ± 0.03 |  |
|
| Probe – 3rd | 81.85 ± 0.01 | 4.88 ± 0.01 | 5.22 ± 0.02 |  |
|
| Bath – 1st | 23.07 ± 0.05 | 32.66 ± 0.03 | 28.36 ± 0.04 |  |
|
| Bath – 2nd | 68.12 ± 0.05 | 13.58 ± 0.02* | 12.01 ± 0.03 |  |
|
| Bath – 3rd | 83.05 ± 0.02 | 3.47 ± 0.01 | 3.76 ± 0.03 |  |
|
In line with expectations, a strong negative correlation was observed between anthocyanin content (TAC) and the L* color parameter (Fig. 2). This result reflects the effect of anthocyanins, particularly abundant in the first extracts obtained from both bath and probe, in making the product darker, thereby reducing its overall perceived lightness. Conversely, a strong positive correlation was found with the a* parameter, consistent with the role of anthocyanins as the pigments responsible for red and purple hues in plants. A counterintuitive relationship emerged between TAC and b*: the positive correlation could be indicative of other light-absorbing compounds and matrix components which can interfere with spectrophotometric evaluation. Notably, quercetin, catechin and their derivatives are prominent flavonoids in grape pomace extracts,13 typically colorless or yellow depending on several factors such as pH, light exposure, and the presence of other compounds within the matrix.18 Their contribution to the yellowish appearance of the extract solutions cannot be excluded. In support of this, an interesting study by Li et al.19 highlighted the co-pigmentation process between malvidin-3-O-glucoside, a typical anthocyanin in Vitis vinifera young red wines and a key determinant of wine color, and quercetin. The results of the study underlined the role of intermolecular hydrogen bonds in stabilizing strong co-pigments, whose presence can significantly influence the UV-Vis spectral characteristics, color properties and stability of red wines or other foods.
3.3. Application of probe-UAE extraction to different grape pomace samples
The probe-based UAE was selected as the best performing technique and then used to obtain extracts from seven different grape pomace samples. The first screening of the phenolic content was made by spectrophotometric determination of the TPC, followed by HPLC-PDA (high performance liquid chromatography equipped with photodiode array detector) analysis for the qualitative and quantitative characterization of the pool. The TPC reported in Table 4, indicated variation in polyphenol concentrations according to the grape cultivar, with Merlot pomace (in particular samples 1 and 2) showing the highest content of polyphenolic compounds while Sangiovese pomace the lowest, a trend quite consistent with HPLC results (Table 4).| GP variety | TPC (mg GAE per g) | Gallic acid (µg g−1) | Protocatechuic acid (µg g−1) | Catechin (µg g−1) | Procyanidin B2 (µg g−1) | Epicatechin (µg g−1) | Chlorogenic acid (µg g−1) | Caffeic acid (µg g−1) |
|---|---|---|---|---|---|---|---|---|
| Merlot-1 | 25.31 ± 0.66 | 36.69 ± 2.37 | 12.62 ± 0.66 | 322.15 ± 9.29 | 62.69 ± 1.78 | 361.16 ± 10.40 | 1.99 ± 0.08 | 9.64 ± 0.59 |
| Merlot-2 | 20.58 ± 1.69 | 28.45 ± 1.20 | 11.27 ± 0.32 | 283.68 ± 9.97 | 70.20 ± 0.99 | 308.32 ± 5.80 | 2.62 ± 0.05 | 6.61 ± 0.09 |
| Merlot-3 | 18.56 ± 0.29 | 22.64 ± 0.10 | 5.45 ± 0.13 | 234.11 ± 2.19 | 55.95 ± 0.60 | 248.40 ± 0.68 | 3.08 ± 0.02 | 7.00 ± 0.07 |
| Cabernet-1 | 17.39 ± 0.24 | 42.50 ± 057 | 7.30 ± 0.14 | 245.43 ± 4.57 | 60.66 ± 0.47 | 253.80 ± 0.44 | 2.12 ± 0.01 | 10.85 ± 0.68 |
| Cabernet-2 | 17.30 ± 0.21 | 43.78 ± 0.12 | 2.48 ± 0.07 | 155.84 ± 4.58 | 48.61 ± 0.65 | 185.90 ± 8.78 | 3.54 ± 0.03 | 8.91 ± 0.19 |
| Sagrantino-1 | 19.31 ± 0.15 | 28.66 ± 0.80 | 0.18 ± 0.00 | 215.36 ± 2.07 | 48.67 ± 0.62 | 127.03 ± 3.42 | 3.64 ± 0.2 | 8.77 ± 0.53 |
| Sangiovese-1 | 10.60 ± 0.37 | 29.18 ± 1.02 | 0.19 ± 0.00 | 96.82 ± 1.18 | 32.01 ± 0.57 | 115.81 ± 0.02 | 0.19 ± 0.00 | 2.83 ± 0.16 |
| GP variety | p-Coumaric acid (µg g−1) | Ferulic acid (µg g−1) | Resveratrol (µg g−1) | Quercetin-3-O-glu (µg g−1) | Kaempferol-3-O-glu (µg g−1) | Quercetin (µg g−1) | Peonidin-3-O-glu (µg g−1) | Tot HPLC (µg g−1) |
|---|---|---|---|---|---|---|---|---|
| Merlot-1 | 2.52 ± 0.11 | 16.58 ± 0.85 | 22.77 ± 0.38 | 7.76 ± 0.13 | 7.22 ± 0.02 | 53.25 ± 3.03 | 353.36 ± 8.37 | 1270.39 ± 26.27 |
| Merlot-2 | 1.92 ± 0.08 | 10.92 ± 0.06 | 7.74 ± 0.30 | 12.14 ± 0.22 | 11.96 ± 0.46 | 41.97 ± 1.14 | 382.15 ± 1.90 | 1179.93 ± 16.26 |
| Merlot-3 | 1.97 ± 0.09 | 5.41 ± 0.09 | 7.46 ± 0.13 | 0.20 ± 0.00 | 5.36 ± 0.34 | 45.54 ± 0.74 | 185.65 ± 2.66 | 828.24 ± 0.35 |
| Cabernet-1 | 3.12 ± 0.10 | 14.16 ± 0.41 | 17.89 ± 0.26 | 9.33 ± 0.29 | 11.23 ± 0.48 | 68.66 ± 1.11 | 402.36 ± 14.69 | 1149.42 ± 20.79 |
| Cabernet-2 | 33.95 ± 1.00 | 9.78 ± 0.37 | 9.40 ± 0.00 | 11.50 ± 0.30 | 14.79 ± 0.50 | 115.35 ± 1.93 | 317.82 ± 7.26 | 961.66 ± 6.04 |
| Sagrantino-1 | 1.71 ± 0.10 | 6.35 ± 0.08 | 7.17 ± 0.08 | 5.08 ± 0.33 | 2.81 ± 0.16 | 48.39 ± 1.18 | 129.99 ± 3.44 | 633.83 ± 0.89 |
| Sangiovese-1 | 0.19 ± 0.00 | 4.10 ± 0.24 | 3.33 ± 0.00 | 24.10 ± 0.19 | 1.64 ± 0.08 | 69.74 ± 2.46 | 111.72 ± 6.87 | 491.85 ± 6.68 |
The chromatographic analysis indicated catechin, epicatechin and peonidin-3-O-glucoside as the most abundant compounds (within the range 96.82–322.15, 115.81–361.16 and 111.72–402.36 µg g−1, respectively), followed by quercetin (within the range 41.97–115.35 µg g−1), gallic acid and procyanidin B2 (22.64–43.78 and 32.01–70.20 µg g−1, respectively). Lower concentrations were measured for the remaining compounds (protocatechuic acid, chlorogenic acid, caffeic acid, p-coumaric acid, ferulic acid, resveratrol, quercetin-3-O-glucoside, kaempferol-3-O-glucoside) overally ranging from 0.19 to 33.95 µg g−1.
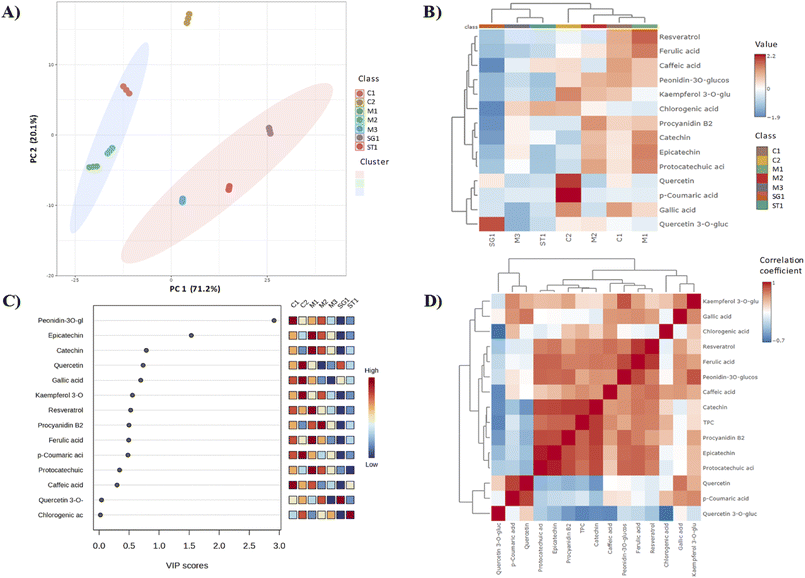
Interrelationships between samples and their chemical profiles were investigated using a chemometric approach. The principal component analysis (PCA) revealed that the first two components (PC1 and PC2) accounted for 91.3% of the total variability, with 71.2% explained by PC1 and 20.2% by PC2. Consistent with the analytical findings, k-means clustering allowed to identify three distinct clusters: cluster 1, comprising Merlot-3 (M3), Sagrantino-1 (ST1) and Sangiovese-1 (SG1); cluster 2 comprising Merlot-1 (M1), Merlot-2 (M2) and Cabernet-1 (C1); cluster 3 with Cabernet-2 (C2) as the sole member (Fig. 3A). This sample organization is reflected enough in the Heatmap hierarchical clustering analysis (HCA) visualization (Fig. 3B), where samples are displayed in columns and compounds in rows. Each colored cell corresponds to a concentration value, with a color scale ranging from red to blue, representing relative intensities from high to low, respectively. The relationship among the identified polyphenols are shown on the left side of the heatmap. What clearly emerges is the well distinct pattern for some bioactive compounds among the grape pomace samples: quercetin-3-O-glucoside was significant only in grape pomace from Sangiovese variety, while quercetin and p-coumaric acid were particularly noticeable in Cabernet-2 pomace. Grape pomaces from Merlot-1, Merlot-2 and Cabernet-1 exhibited a more similar polyphenolic pattern. The variable importance in projection (VIP) scores, with colored boxes on the right indicating the relative concentrations of the corresponding metabolites in each sample, allowed to identify key polyphenols that significantly influenced the dataset outcomes (Fig. 3C). Peonidin 3-O-glucoside and epicatechin, characterized by a high VIP score (>1.0), emerged as the strongest variables in the model, revealing as potential predictive markers for discriminating among different grape pomace varieties. A further insight can be retrieved from the correlation heatmap in Fig. 3D, which allows to visualize the relationship between variables in a dataset. The intensity of the color, ranging from −1 (perfect negative correlation) to 1 (perfect positive correlation), with 0 indicating no correlation, reflects the magnitude of these correlations. Interestingly, a high positive (close to 1) correlation was observed between p-coumaric acid and quercetin, between epicatechin and protocatechuic acid, and between ferulic acid and resveratrol. In contrast, chlorogenic acid and quercetin-3-O-glucoside showed very strong negative correlation. Moreover, quercetin and its glycoside derivative exhibited a negative correlation with TPC, while gallic acid and p-coumaric acid displayed almost no correlation with this assay.
Overall, the results obtained so far highlight not only the critical role of extraction parameters on polyphenol yield, but also the pivotal influence of grape pomace origin in determining the final profile in bioactives, thus impacting the relative biological properties.
4. Bioaccessibility study
One of the most important characteristics of foods, particularly functional foods, is their bioefficiency, that is, their ability to meet the metabolic needs of the consumer. In this context, bioaccessibility and bioavailability are two key parameters used to evaluate the behaviour of bioactive compounds. These can be investigated through in vivo, ex vivo, or in vitro approaches and expressed quantitatively using different measurement units.Among the factors influencing polyphenol bioaccessibility, pH and enzymatic activity play a crucial role, as they can significantly affect the stability and release of these compounds. Simulated digestion models therefore provide a realistic approximation of the chemical transformations that bioactives can undergo from their release from the food matrix to their potential absorption in the gastrointestinal tract.20
This part of the study focused on the assessment of the in vitro bioaccessibility of polyphenols using simulated gastrointestinal fluid systems, to evaluate their potential use as nutraceuticals or the need to develop specific delivery systems to protect bioactives until they reach the small intestine where absorption primarily occurs. The protocol adopted in this work was the INFOGEST one, an in vitro static digestion method based on constant ratios of meal to digestive fluids and a fixed pH values for each step of the digestion process (See Section 2.6 for details). Using this approach, both raw grape pomaces and the extracts from Merlot-1 and Cabernet-1 were subjected to sequential oral, gastric and intestinal digestion, by setting for each fluid the relative electrolyte composition, type and concentration of enzymes, content of bile salts, dilution, pH and digestion time as based on available physiological data. The extent of compound release at each stage was monitored using HPLC-PDA analysis. The results obtained for the gastric and intestinal digestion phases are summarized in Table 5. Recovery percentage of each identified polyphenol at the end of digestion for all sample types [GP-Ctrl(G) and GP-Ctrl(I), GP-GD and GP-ID, GPE-GD and GPE-ID] was calculated in relation to its amount in the undigested grape pomace extract (Table 4), used as the reference affording the maximum polyphenol content obtainable.
| Gallic acid | Protocatechuic acid | Catechin | Procyanidin B2 | Epicatechin | Chlorogenic acid | Caffeic acid | ||
|---|---|---|---|---|---|---|---|---|
| a Different letters in each column, relatively to the extracts from Cabernet Sauvignon or Merlot, indicate significant differences with p-value < 0.01. The symbols *, §, † and ‡ indicate significant differences with p-value < 0.05. GP-Ctrl(G) = grape pomace control in gastric conditions (water); GP-GD = grape pomace subjected to gastric digestion; GPE-GD = grape pomace extract subjected to gastric digestion. GP-Ctrl(I) = grape pomace control in intestinal conditions (water); GP-ID = grape pomace subjected to intestinal digestion; GPE-ID = grape pomace extract subjected to intestinal digestion. nd = not determined. | ||||||||
| Cabernet sauvignon | GP-Ctrl(G) | 108.08 ± 1.00a,b,* | nd | 15.79 ± 0.51a,b | 64.77 ± 4.47a | 9.38 ± 0.64a | 132.35 ± 4.32a,* | nd |
| GP-GD | 88.58 ± 3.06a,§ | nd | 10.12 ± 0.06b,* | nd | nd | 121.22 ± 18.36a | nd | |
| GPE-GD | 120.62 ± 3.78b,c,§ | 155.42 ± 3.68a | 91.71 ± 6.55c | 138.38 ± 6.17b | 83.25 ± 4.49b | 195.20 ± 20.14b,* | 59.27 ± 4.29a | |
| GP-Ctrl(I) | 136.59 ± 12.50c,* | nd | 54.41 ± 4.09d,§ | nd | 70.09 ± 11.82b | 162.78 ± 12.06b | 98.23 ± 8.12b | |
| GP-ID | 41.10 ± 8.09d | 262.91 ± 13.87b | 28.29 ± 1.39a,e,* | nd | nd | 86.74 ± 0.48a | 162.79 ± 6.28c | |
| GPE-ID | < LOD | nd | 38.18 ± 3.11e,§ | 71.02 ± 2.76a | nd | 87.76 ± 3.44a | 149.18 ± 8.14c | |
| Merlot | GP-Ctrl(G) | 150.13 ± 21.37a,* | nd | 43.61 ± 0.69a | 104.19 ± 0.74a | 59.49 ± 3.44a | 147.10 ± 1.87a | nd |
| GP-GD | 106.49 ± 6.08b,*,§ | nd | 25.26 ± 0.02b | nd | 34.79 ± 0.94a,d | 144.10 ± 7.84a | nd | |
| GPE-GD | 116.89 ± 1.10a,b,† | nd | 94.50 ± 1.85c | 149.87 ± 9.51b | 164.08 ± 17.70b | 222.99 ± 64.52a | nd | |
| GP-Ctrl(I) | 157.34 ± 3.82a,§,† | nd | 67.01 ± 0.41d | 90.09 ± 4.83a,* | 119.01 ± 9.19c | 153.47 ± 3.89a | 47.50 ± 5.02a | |
| GP-ID | 44.22 ± 1.93d | nd | 35.90 ± 0.45e | 55.80 ± 0.53c | 29.03 ± 3.96a,d | 246.70 ± 25.56a | 54.84 ± 7.26a | |
| GPE-ID | nd | nd | 27.13 ± 1.31b | 70.74 ± 0.35c,* | 3.19 ± 0.73d | 151.05 ± 14.79a | nd | |
| p-Coumaric acid | Ferulic acid | Resveratrol | Quercetin 3-O-glucoside | Kaempferol 3-O-glucoside | Quercetin | Peonidin 3-O-glucoside | ||
|---|---|---|---|---|---|---|---|---|
| Cabernet sauvignon | GP-Ctrl(G) | nd | 46.74 ± 3.56a,* | nd | 12.88 ± 1.84a | nd | nd | 18.05 ± 0.56a,* |
| GP-GD | nd | 36.40 ± 2.01a | nd | 12.34 ± 1.12a | 11.21 ± 1.26a,* | nd | 17.95 ± 0.58a,§ | |
| GPE-GD | 80.01 ± 2.53a | 142.41 ± 21.24b | 65.34 ± 1.25 | 26.62 ± 0.73b | 48.68 ± 5.73b,c,§ | 2.52 ± 0.08a | 56.38 ± 1.53b | |
| GP-Ctrl(I) | nd | 75.42 ± 2.62a | nd | 23.33 ± 0.59b | 18.21 ± 2.82a,§,† | nd | 28.39 ± 0.75c | |
| GP-ID | 254.47 ± 9.36b | 69.94 ± 14.87a | nd | 83.30 ± 1.70c | 42.67 ± 7.70b,*,†,‡ | nd | 14.38 ± 0.21d,*,§ | |
| GPE-ID | 226.67 ± 21.30b | 107.47 ± 5.47b,* | nd | 37.47 ± 2.84d | 67.27 ± 7.85c,‡ | 4.89 ± 0.10b | 8.62 ± 0.05e | |
| Merlot | GP-Ctrl(G) | nd | 95.80 ± 13.03a,c | nd | 114.75 ± 28.16a,* | 14.17 ± 1.19a,* | nd | 33.82 ± 0.73a |
| GP-GD | nd | 59.86 ± 7.82a,* | nd | 118.93 ± 2.97a,*,§,† | 20.14 ± 0.08b,* | nd | 30.86 ± 1.15a | |
| GPE-GD | nd | 206.66 ± 27.84b | nd | 209.63 ± 34.16b,§,‡ | 73.31 ± 0.50c | nd | 58.46 ± 0.13b | |
| GP-Ctrl(I) | nd | 142.15 ± 16.79c,* | nd | 275.15 ± 25.04b | 29.83 ± 1.26d | nd | 46.07 ± 1.34c | |
| GP-ID | 381.39 ± 5.46a | 134.29 ± 20.69a,c | nd | 240.66 ± 15.80b,† | 40.38 ± 2.19e | nd | 30.72 ± 0.84a | |
| GPE-ID | 611.81 ± 20.49b | 138.88 ± 20.54c,* | nd | 102.87 ± 10.45a,‡ | 52.46 ± 2.28f | nd | 2.72 ± 0.07d |
The comparison between gastric-digested grape pomace (GP-GD) and the control one [GP-Ctrl(G)], treated under the same conditions of time and water volume for each digestive step, indicated a general decrease in the content of identified polyphenols in the GP-GD samples from both Cabernet and Merlot. The only exceptions were kaempferol-3-O-glucoside from both varieties and quercetin-3-O-glucoside from Cabernet, which increased compared to the control sample. The complex interpretation of these results reflects the influence and intricacy of the matrix composition in affecting the release of phytochemicals into the gastrointestinal fluids. However, in most cases, the differences observed were not statistically significant, indicating that the gastric-digestion process on the raw grape pomace had only a limited impact on the overall polyphenol release and content.
Interesting results emerged when samples were subjected to intestinal digestion. The comparison between intestinal-digested grape pomace (GP-ID) and the control [GP-Ctrl(I)] provided remarkable insights (Table 5). Some polyphenols, including gallic acid, chlorogenic acid and peonidin-3-O-glucoside (from Cabernet), epicatechin (from Merlot), decreased during the transition from gastric to intestinal digestion. In contrast, other bioactive compounds increased following intestinal digestion.
Protocatechuic acid from Cabernet, for example, experienced a substantial increase after this digestive step. This compound commonly exists in both free or bound forms in plants, often linked via ester bonds to cell wall components, as described by Weber et al. in 1995 in grapevine leaves.21 Therefore, it is plausible to hypothesize that the action of intestinal enzymes can favour the release of protocatechuic acid, otherwise unquantifiable in the controls [GP-Ctrl(G) and GP-Ctrl(I)]. A similar trend was observed for caffeic acid and p-coumaric acid. In contrast, compounds as catechin, ferulic acid, quercetin3-O-glucoside and kaempferol-3-O-glucoside (from both varieties), as well as chlorogenic acid (from Merlot), exhibited increased levels after intestinal digestion compared to the gastric one, although their concentrations remained lower than those measured in the respective controls. In general, the observed results can be plausibly attributed to the presence of the above bioactive compounds as sugar conjugated or covalently bound to other molecules or structural cell wall components.22
The hydrolysis of these complexed forms under acidic or alkaline conditions, or through enzymatic activity, can affect the efficiency of their release, depending on both the complexity of the cell wall and the chemical nature of the compounds. This is a key point, as increasing the released fraction of bioactives from the food matrix can enhance their bioaccessibility and potentially their absorption in the intestine, thereby maximizing their effects.20 Moreover, it is noteworthy that enzymes, pH, and bile salts used to simulate the gastrointestinal environment can induce structural modifications in phenolic compounds, leading to the formation of new molecules with altered bioavailability and biological activities.
The bioaccessibility was assessed in the extracts obtained from the two grape pomaces. The results revealed a different pattern compared to the whole grape pomace. Firstly, the extraction process led to a higher polyphenol content, with only few exceptions, than those observed in the raw grape pomaces [GP-Ctrl(G) and GP-Ctrl(I)]. This outcome reflects the effectiveness of the extraction in concentrating specific bioactives while minimizing interferences from the food matrix. Interestingly, gastric digestion led to an increase in the content of several molecules, resulting into bioaccessibility values higher than 100% compared to the GP extract (GPE). In line with the observations of Ferreira-Santos et al. this outcome may indicate the biotransformation of high molecular-weight phenols through depolymerisation, yielding low molecular-weight compounds favoured by the acidic conditions of the gastric environment.23 These values underwent drastic decreases when the extracts were subjected to intestinal digestion simulation. For instance, gallic acid, which reached 120.62% after gastric digestion (GPE-GD), dropped to undetectable levels after intestinal digestion (GPE-ID). Similar trends have been described in literature, underlying a decrease in polyphenols standards after intestinal digestion compared to their initial concentrations.23,24 This reduction can be plausibly attributable to the higher instability of these compounds under moderate alkaline conditions.
Interestingly, in a recent study by Calvo et al. carried out on white grape pomace, the authors attributed the dramatic decrease in polyphenol concentration after the intestinal phase to the interaction between bile salts and polyphenols, responsible for the precipitation of approximately 50% of the phenolic compounds. This finding suggests that the observed reduction is not only due to polyphenol degradation under intestinal conditions, but also to their insolubilization. Importantly, such interaction with bile salts suggests that the insolubilized polyphenols can remain longer in the gut, potentially acting as prebiotic compounds for the microbiota.25
On the contrary, compounds such as caffeic acid, quercetin-3-O-glucoside, kaempferol-3-O-glucoside and quercetin (in extracts from Cabernet), as well as p-coumaric acid (in extracts from both varieties) exhibited a notable improvement at the end of the digestion process. This observation supports the hypothesis that complexed or conjugated polyphenols were biotransformed into simpler molecules, thereby enhancing their bioaccessibility and, theoretically, their subsequent absorption.
The obtained results indicate that sample matrix can, in some cases, protect the integrity of bioactives during in vitro gastrointestinal digestion, facilitating their release from more complex structures, as observed in the case of raw grape pomace. Conversely, when considering the extracts, the results generally revealed higher polyphenol levels, compared to the grape pomace, after gastric digestion. However, the significant low recovery observed after the intestinal step suggests that appropriate formulation strategies may be required to preserve the integrity of these compounds throughout digestion. In this context, the study by Martinović et al. evaluated the effect of encapsulating phenol-rich grape pomace extracts using systems based on sodium alginate and gelatin or chitosan on the bioaccessibility of polyphenols during in vitro simulated digestion. The results highlighted that bioaccessibility can be differentially influenced depending on the encapsulation system, emphasizing the importance of selecting a proper delivery method for incorporating the extracts into orally administered products.26
5. Conclusions
This study provides a comprehensive extraction and characterization of grape pomace polyphenols. Probe-based UAE was selected as the best performing extraction technique, as it yielded higher concentrations of bioactive compounds, while preserving the antioxidant activity. Its higher efficiency was readily evident in the first extraction step, eliminating the need for consecutive extractions, as confirmed by colorimetric analysis. The best identified extraction conditions applied to four grape varieties (Merlot, Cabernet Sauvignon, Sangiovese and Sagrantino) indicated Merlot and Cabernet as the richest in polyphenols, as determined by HPLC-PDA analysis.In vitro bioaccessibility evaluations performed on both raw grape pomaces and the corresponding extracts, revealed significant variation of the phenolic composition during simulated digestion, strongly influenced by the sample matrix. The biotransformation of some bioactives into low molecular-weight compounds, favoured by acidic gastric conditions, can potentially increase their bioaccessibility. Interesting differences between the raw grape pomaces and their extracts, suggest a protective effect of the pomace matrix on polyphenol integrity during digestion, whereas extracts exhibited higher susceptibility to degradation after the gastric phase, highlighting the need for appropriate formulation strategies to improve compound stability and bioaccessibility.
Overall, this study highlights the high potential of grape pomace as a valuable material for the development of functional foods and nutraceuticals: understanding the chemical modifications during sample processing and the biological transformations occurring during gastrointestinal digestion is essential to assess the bioavailability of bioactives and their potential contributions to human health. Furthermore, future studies focused on the role of gut microbiota in the biotransformation of both polyphenol-based extracts and native grape pomace would further support the valorization of this matrix, or its extracts, as functional ingredients and help to identify effective strategies to improve their bioavailability.
Author contributions
Filippo Peruccaccia: methodology, investigation, data curation. Luciano Mangiapelo: methodology, investigation. Shahd Ali: methodology. Francesca Blasi: writing – review & editing. Federica Ianni: writing – original draft, writing – review & editing, validation, formal analysis. Lina Cossignani: conceptualization, writing – review & editing, project administration.Conflicts of interest
There are no conflicts to declare.Data availability
All data supporting the findings of this study are included in the article.Supplementary information (SI): figure S1. Graphic representation of TPC, AOX and TAC after consecutive extractions using probe and bath systems for Cabernet Sauvignon extracts (A–C) and Merlot (D–F). See DOI: https://doi.org/10.1039/d5fb00944h.
Acknowledgements
This study was carried out within the AgriBioPack project “Valorizing Agrifood Residues for Bio-based Packaging Solutions”, funded in the frame of the PRIMA call, Section 2, Topic 2.3.1–2023 (RIA) “Assessing novel antimicrobial food packaging and coating materials to reduce food waste to improve safety in the Mediterranean food supply chain”, and received funding from the Italian Ministry of University and Research.References
- F. Blasi, V. Trovarelli, L. Mangiapelo, F. Ianni and L. Cossignani, Foods, 2024, 13, 3541, DOI:10.3390/foods13223541.
- J. D. C. Lopes, J. Madureira, F. M. A. Margaça and S. Cabo Verde, Molecules, 2025, 30, 362, DOI:10.3390/molecules30020362.
- P. Chowdhary, A. Gupta, E. Gnansounou, A. Pandey and P. Chaturvedi, Environ. Pollut., 2021, 278, 116796, DOI:10.1016/j.envpol.2021.116796.
- I. Tomaz, N. Huzanić, D. Preiner, D. Stupić, Ž. Andabaka, E. Maletić, J. K. Kontić and D. Ašperger, in Polyphenols in Plants – Isolation, Purification and Extract Preparation, ed. Ronald Ross Watson, Academic Press, London, 2nd edn, 2019, 10, 151–167. DOI: DOI:10.1016/B978-0-12-813768-0.00010-4.
- D. Pintać, T. Majkić, L. Torović, D. Orčić, I. Beara, N. Simin, N. Mimica-Dukić and M. Lesjak, Ind. Crops Prod., 2018, 111, 379–390, DOI:10.1016/j.indcrop.2017.10.038.
- L. E. N. Castro, W. G. Sganzerla, A. P. G. Silva, O. D. John, T. L. C. Tabosa Barroso, M. A. Rostagno and T. Forster-Carneiro, Phytochem. Rev., 2025, 24, 2059–2086, DOI:10.1007/s11101-024-09991-3.
- M. Temkov, E. Velickova, V. Stamatovska and G. Nakov, Sci. Pap. Ser. Manag. Econom. Eng. Agric. Rural. Dev., 2021, 21, 753–762 Search PubMed.
- J. Grgić, G. Šelo, M. Planinić, M. Tišma and A. Bucić-Kojić, Antioxidants, 2020, 9, 923, DOI:10.3390/antiox9100923.
- Y. Hu, Q. Lin, H. Zhao, X. Li, S. Sang, D. J. McClements, J. Long, Z. Jin, J. Wang and C. Qiu, Bioaccessibility and bioavailability of phytochemicals: Influencing factors, improvements, and evaluations, Food Hydro., 2023, 135, 108165, DOI:10.1016/j.foodhyd.2022.108165.
- L. Mangiapelo, F. Blasi, F. Ianni, C. Suvieri, R. Sardella, C. Volpi and L. Cossignani, Food Bioprocess Technol., 2024, 17, 1942–1957, DOI:10.1007/s11947-023-03249-0.
- L. Mangiapelo, F. Ianni, F. Blasi, M. Bastianini, R. Spogli and L. Cossignani, Food Chem., 2025, 471, 142719, DOI:10.1016/j.foodchem.2024.142719.
- L. Mangiapelo, N. Pinna, F. Blasi, F. Ianni, G. Verducci and L. Cossignani, Molecules, 2025, 30, 693, DOI:10.3390/molecules30030693.
- A. Brodkorb, L. Egger, M. Alminger, P. Alvito, R. Assunção, S. Ballance, T. Bohn, C. Bourlieu-Lacanal, R. Boutrou, F. Carrière, A. Clemente, M. Corredig, D. Dupont, C. Dufour, C. Edwards, M. Golding, S. Karakaya, B. Kirkhus, S. Le Feunteun, U. Lesmes, A. Macierzanka, A. R. Mackie, C. Martins, S. Marze, D. J. McClements, O. Ménard, M. Minekus, R. Portmann, C. N. Santos, I. Souchon, R. P. Singh, G. E. Vegarud, M. S. J. Wickham, W. Weitschies and I. Recio, Nat. Protoc., 2019, 14, 991–1014, DOI:10.1038/s41596-018-0119-1.
- O. Gligor, A. Mocan, C. Moldovan, M. Locatelli, G. Crişan and I. C. F. R. Ferreira, Trends Food Sci. Technol., 2019, 88, 302–315, DOI:10.1016/j.tifs.2019.03.029.
- B. K. Tiwari, TrAC, Trends Anal. Chem., 2015, 71, 100–109, DOI:10.1016/j.trac.2015.04.013.
- K. Kumar, S. Srivastav and V. S. Sharanagat, Ultrason. Sonochem., 2021, 70, 105325, DOI:10.1016/j.ultsonch.2020.105325.
- M. I. Prawira-Atmaja, S. Puangpraphant and J. Saudi, Soc. Agric. Sci., 2025, 24, 10, DOI:10.1007/s44447-025-00014-x.
- L. M. Guardianelli, M. V. Salinas, M. C. Puppo, A. Hidalgo and G. Pasini, Foods, 2025, 14, 2386, DOI:10.3390/foods14132386.
- Y. Li, M. Prejanò, M. Toscano and N. Russo, Front. Chem., 2018, 6, 245, DOI:10.3389/fchem.2018.00245.
- K. Wojtunik-Kulesza, A. Oniszczuk, T. Oniszczuk, M. Combrzyński, D. Nowakowska and A. Matwijczuk, Nutrients, 2020, 12, 1401, DOI:10.3390/nu12051401.
- B. Weber, L. Hoesch and D. M. Rast, Phytochemistry, 1995, 40, 433–437, DOI:10.1016/0031-9422(95)00314-W.
- G. Rocchetti, R. Perez Gregorio, J. M. Lorenzo, F. J. Barba, P. García Oliveira, M. A. Prieto, J. Simal-Gandara, J. I. Mosele, M. J. Motilva, M. Tomas, V. Patrone, E. Capanoglu and L. Lucini, Compr. Rev. Food Sci. Food Saf., 2022, 21, 811–842, DOI:10.1111/1541-4337.12921.
- P. Ferreira-Santos, C. Nobre, R. M. Rodrigues, Z. Genisheva, C. Botelho and J. A. Teixeira, Food Chem., 2024, 436, 137780, DOI:10.1016/j.foodchem.2023.137780.
- D. D. Milinčić, N. S. Stanisavljević, M. M. Pešić, A. Ž. Kostić, S. P. Stanojević and M. B. Pešić, Foods, 2025, 14, 607, DOI:10.3390/foods14040607.
- L. G. Calvo, M. Celeiro, M. Lores, A. G. Abril and T. de Miguel, Food Chem., 2025, 480, 143810, DOI:10.1016/j.foodchem.2025.143810.
- J. Martinović, J. Lukinac, M. Jukić, R. Ambrus, M. Planinić, G. Šelo, A. M. Klarić, G. Perković and A. Bucić-Kojić, Molecules, 2023, 28, 5285, DOI:10.3390/molecules28135285.
| This journal is © The Royal Society of Chemistry 2026 |