Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
High-pressure technology: application as a pretreatment technique for de-bittering and fermentation acceleration of green table olives
Varvara Andreou *,
Sofia Chanioti,
Panagiota Stergiou and
George Katsaros*
*,
Sofia Chanioti,
Panagiota Stergiou and
George Katsaros*
Institute of Technology of Agricultural Products, Hellenic Agricultural Organization-DEMETER, ELGO-DEMETER, Sof. Venizelou 1, Str., GR14123, Lykovrissi, 14123, Attica, Greece. E-mail: vandreou@chemeng.ntua.gr; schanioti@gmail.com; ster.panagiota@gmail.com; gkatsaros@elgo.gr; Tel: +30 2102845940
First published on 4th March 2026
Abstract
An alternative approach is proposed through the application of high-pressure (HP) technology for the acceleration of table olive de-bittering processing. HP-assisted de-bittering in 8% NaCl brine for 5–20 min at 100–400 MPa was performed. The conventional “Greek-style” method (immersion in an 8% NaCl brine) and lye treatment (immersion in a 2% NaOH solution for 8 h) were also tested and compared. Oleuropein content, sensory properties, and quality characteristics, including color and texture, were evaluated immediately after treatment. Following de-bittering, all the samples underwent fermentation in an acidified brine (8% NaCl, pH 5) for at least six months to assess fermentation duration and final product quality. HP treatment resulted in up to 80% reduction in oleuropein immediately after processing, whereas NaOH and conventional brine treatments required 24 h and 8 weeks, respectively, to achieve equivalent reduction. The HP process did not alter the initial lactic acid bacteria population, allowing normal fermentation progression. Brine pH dropped below 4.2, and bitterness intensity became acceptable after approximately 101, 45, and 250 days for samples debittered by conventional, HP, and NaOH processes, respectively. No significant differences were observed in the quality characteristics of the final fermented olives. These results demonstrate that HP-assisted de-bittering efficiently minimizes processing time without compromising product quality while simultaneously reducing the use of alkali solutions, a critical concern in the table olive industry. HP technology thus represents a promising alternative for accelerating olive de-bittering and for streamlining production, maintaining desirable sensory and textural attributes in the final product. The study highlights the potential of HP as a rapid, effective, and sustainable approach for table olive processing.
Sustainability spotlightTable olives contain bitter phenolic compounds, such as oleuropein, which require extensive processing to ensure consumer acceptance, traditionally involving long fermentation times and substantial use of chemicals. This study demonstrates that high-pressure (HP) treatment can significantly accelerate olive debittering while preserving sensory and nutritional quality. By reducing processing time and minimizing the use of alkali solutions, HP-assisted debittering decreases chemical consumption, energy use, and environmental impact, providing a more sustainable production method. This approach aligns with multiple UN Sustainable Development Goals, including SDG 2 (Zero Hunger), SDG 6 (Clean Water and Sanitation), SDG 9 (Industry, Innovation and Infrastructure), SDG 12 (Responsible Consumption and Production), and SDG 13 (Climate Action), highlighting its potential contribution to resource-efficient, innovative, and environmentally responsible food processing. |
Introduction
The consumption of table olives has been increasing continuously over the last two decades1,2 due to their popularity and proven health effects as part of the Mediterranean diet.3 Table olives are rich in phenolic compounds, which are well-known for their antioxidant properties.4 However, some of these compounds, such as oleuropein, impart a bitter taste,5,6 rendering fresh-cut olives inedible.7 To make table olives acceptable for consumption, they must undergo a series of processing steps, including de-bittering, fermentation, and acidification.8 The de-bittering is a mandatory process as it aims to (a) reduce the oleuropein and its aglycone contents, phenolic compounds associated with characteristic bitterness, (b) increase the olives' permeability to accelerate the subsequent brining step9 and (c) improve the consumers' acceptance.10 The most well-known and widely used de-bittering methods for table olives are the “Greek-style”, “Spanish-style” and “California-style” processes.4,8 Conventional de-bittering (Greek-style) includes the immersion of olives in brine (5–10% w/w), where the bitter phenolic compounds are removed by diffusion from olive flesh into the brine, and simultaneously, these compounds are hydrolyzed by the presence of endogenous enzymes, such as β-glucosidase, and esterase present in the olives' surface, producing non-bitter compounds.11 This process is extremely time-consuming, requiring approximately 3–6 months.12,13 The natural bitterness can be eliminated, or at least reduced, by the addition of NaOH solution for several hours (lye de-bittering or Spanish-style), rendering the product acceptable for human consumption.14,15 Strong bases can penetrate the olive flesh, promoting the rapid hydrolysis of oleuropein, producing non-bitter compounds, such as hydroxytyrosol or tyrosol.8 Moreover, lye treatment causes structural changes in olives, decreasing their firmness and facilitating the subsequent fermentation step.16 Before the fermentation starts, water-rising steps to remove NaOH from olive flesh are required, and the pH drops below 7. This step produces high volumes (3.9–7.5 m3 per t) of heavily contaminated wastewaters, raising several environmental concerns.4,17,18 Nowadays, growing interest from the olive industry and the preference of consumers for healthier lifestyles have led to the development and exploration of alternative novel technologies to replace or assist the typical processing steps used in table olive production.4,19 The use of novel technologies aims to decrease processing time, thereby reducing energy savings and costs, lowering water usage, promoting sustainability and improving the quality of the final commercial table olives. Ultrasound,20,21 vacuum impregnation,22 carbon dioxide overpressure,23 and the use of enzyme mixtures22 are some of the alternative processes used to reduce the time and cost of the de-bittering of table olives. High Pressure (HP) may also potentially be used to enhance the extraction of oleuropein, decreasing the bitter taste of olives in significantly shorter times. HP has never been explored as a pretreatment process to assist the de-bittering of commercial table olives. However, it has been implemented for the extraction of intracellular compounds from olives,24,25 resulting in increased yields. The HP mechanism includes the alteration of cell wall structure due to pressure-induced cell compression, promoting more effective diffusion of intracellular material into the surrounding extraction media compared to the conventional extraction processes.26–28 HP focuses on the almost-instant and effective removal of the bitter phenolic compounds from the olive flesh. The final process to produce the commercial table olives includes a natural fermentation in brine (5–10% salt) that lasts up to several months. Fermentation is typically a spontaneous process that follows the de-bittering treatment and proceeds through the activity of lactic acid bacteria (LAB) and yeasts that coexist in the olives.29 LAB promotes and continues the de-bittering of the olives through hydrolysis,30,31 lowers the brine pH (below 4), and improves the sensorial characteristics of the final products.32 The aim of the present study was to evaluate the potential of HP treatment as a pretreatment method to accelerate the de-bittering of table olives, followed by the typical fermentation step. Specifically, the study investigated how HP-assisted de-bittering affects oleuropein reduction, the progression and duration of fermentation, and the quality characteristics of the final product, including color, texture, and sensory properties. The results of HP treatment were compared with those obtained using traditional Greek-style fermentation in 8% NaCl brine and lye treatment (2% NaOH for 8 h), to determine whether HP can provide a faster and more efficient alternative without compromising the organoleptic and microbial quality of the olives. In this context, the novelty of the present study lies in the application of HP pre-treatment prior to both de-bittering and the subsequent fermentation, which has not been previously explored, allowing us to evaluate its effects on microbial dynamics, oleuropein reduction, and final product quality within the full industrial processing sequence.Materials and methods
Raw materials
All experiments in this study were carried out using green table olives of the Konservolia variety (Amfissa, Greece) at the first stage of ripening (light-green flesh). Olives were harvested in October 2021 and immediately transferred to the Institute of Technology of Agricultural Products of ELGO-Demeter. Olives were stored at 7 °C until further analysis (for less than one week). Before each experiment, the olives were washed with running tap water. The ripeness index of the olives was determined according to the method of Gutiérrez, Jimenez, Ruiz, and Albi,33 using 100 randomly selected fruits. Based on skin and flesh color classification, the calculated ripeness index was 0.58, indicating an early maturity stage suitable for table olive processing.Experimental design
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio olives:brine). The HP-assisted de-bittering process was performed at 100, 250 and 400 MPa for 5 and 20 min, followed by immersion in acidified (pH 5.5 with CH3COOH) brine (8% w/w NaCl) for 48 h. The lye (2% NaOH, 8 h) de-bittering process was also conducted. A 2% NaOH solution was selected based on literature21,22,34–36 and industrial practices. Higher concentrations of NaOH can cause excessive softening. Oleuropein content of samples debittered by HP, NaOH and conventional processes was monitored along with a sensory evaluation of the samples (intensity of bitter taste) during each de-bittering process. Quality properties were also evaluated, such as the color and texture of de-bittered table olives. Two replicates from each condition were performed.
1 ratio olives:brine). The HP-assisted de-bittering process was performed at 100, 250 and 400 MPa for 5 and 20 min, followed by immersion in acidified (pH 5.5 with CH3COOH) brine (8% w/w NaCl) for 48 h. The lye (2% NaOH, 8 h) de-bittering process was also conducted. A 2% NaOH solution was selected based on literature21,22,34–36 and industrial practices. Higher concentrations of NaOH can cause excessive softening. Oleuropein content of samples debittered by HP, NaOH and conventional processes was monitored along with a sensory evaluation of the samples (intensity of bitter taste) during each de-bittering process. Quality properties were also evaluated, such as the color and texture of de-bittered table olives. Two replicates from each condition were performed.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio olives
1 ratio olives![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) brine) and lasted for 6 months (until the pH dropped below 4.2). Three different samples were assessed: (a) natural fermentation – control, (b) HP-pretreated (optimal HP condition – 250 MPa for 15 min – as assisted de-bittering step) olives, (c) lye de-bittered olives, to evaluate the effect of each de-bittering technique on the duration of the fermentation step and on the quality of the final product. Oleuropein content and pH were also measured throughout fermentation. Quality and sensory evaluations were also performed.
brine) and lasted for 6 months (until the pH dropped below 4.2). Three different samples were assessed: (a) natural fermentation – control, (b) HP-pretreated (optimal HP condition – 250 MPa for 15 min – as assisted de-bittering step) olives, (c) lye de-bittered olives, to evaluate the effect of each de-bittering technique on the duration of the fermentation step and on the quality of the final product. Oleuropein content and pH were also measured throughout fermentation. Quality and sensory evaluations were also performed.High-pressure unit
The HP-assisted de-bittering process was performed using an HP unit (Food Pressure Unit FPU 1.01, Resato International BV, Roden, Holland) with a 1.5 L volume vessel. Olives (∼500 g) were immersed in acidified (pH 5.5) brine with 8% w/w NaCl at a ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (wolives/wbrine) and were packaged using a multilayer PP aluminum material. HP experiments were conducted at room temperature. Pressure increase in the vessel was achieved using a pressure intensifier, and tap water was used as the pressure-transmitting fluid. The temperature and pressure inside the vessel were monitored during HP treatment. Each HP condition was repeated twice.
1 (wolives/wbrine) and were packaged using a multilayer PP aluminum material. HP experiments were conducted at room temperature. Pressure increase in the vessel was achieved using a pressure intensifier, and tap water was used as the pressure-transmitting fluid. The temperature and pressure inside the vessel were monitored during HP treatment. Each HP condition was repeated twice.
Determination of bioactive compounds of table olives
Total phenolic compounds, oleuropein and hydroxytyrosol content, as well as the antioxidant activity, were determined for each sample throughout the de-bittering and fermentation processes. Solid–liquid extraction was performed according to Andreou et al.24 De-pitted olives were freeze-dried using a freeze dryer (Thermo Savant, ModulyoD, USA). One gram of each dried sample was mixed with 80% methanol solution for 1 h in an ultrasonic bath (Sultan Pro-Sonic 300, Sultan Healthcare, Smile Way, NY). The samples were centrifuged at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 min, and the obtained extracts were stored at −18 °C until further analysis. Three replicates of each measurement were performed.
000 rpm for 10 min, and the obtained extracts were stored at −18 °C until further analysis. Three replicates of each measurement were performed.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. The elution of compounds was monitored at 280 nm. The identification and quantification of oleuropein and hydroxytyrosol were performed by the addition of standards and by comparison with their respective standard curves, respectively. The concentration of each compound is expressed as mg/100 g dry matter (d.m.). Three replicates of each measurement were performed.
2. The elution of compounds was monitored at 280 nm. The identification and quantification of oleuropein and hydroxytyrosol were performed by the addition of standards and by comparison with their respective standard curves, respectively. The concentration of each compound is expressed as mg/100 g dry matter (d.m.). Three replicates of each measurement were performed.Analysis of quality characteristics
The main quality characteristics, such as color and texture of the table olives, were monitored throughout both the de-bittering and fermentation steps. Color (CIELab colorscale – Commission International de I'Eclairage) of olives was expressed in terms of L* (luminosity), a* (redness) and b* (yellowness) by using a colorimeter Minolta CR-300 (Minolta Company, Chuo-Ku, Osaka, Japan). Total color difference, ΔE, was also determined based on eqn (1):
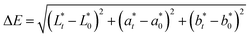 | (1) |
Texture analysis was performed for each studied sample by using a texture analyser HD plus (Stable Micro Systems Ltd, Godalming, UK), equipped with a suitable needle probe (0.5 mm diameter). The velocity of the probe was fixed at 0.5 mm s−1, and the samples were compressed by 2 mm. The maximum peak force was the mechanical parameter considered and is expressed as firmness (N). Nine replicates were performed for each sample studied.
pH determination
The pH of the brine during the fermentation process was measured using an ORION pH meter (ORION 900 ion analyzer model 900 A, ORION-scientific, Boston, USA).Microbial analysis during fermentation of table olives
Total viable count (TVC) (Plate Count Agar-Biokar Diagnostics, Beauvais, France), lactic acid bacteria (LAB), Enterobacteriaceae, and yeasts and molds were determined by using the pour plate method (ISO 15214:1998) for TVC, LAB and Enterobacteriaceae, and the surface plating technique (ISO4833-2:2013) for yeasts and molds. TVC, LAB, Enterobacteriaceae and yeasts/molds were grown on Plate Count Agar Standard (Biokar, Zac de ther, France), De Man–Rogosa–Sharpe Agar (MRS Agar (ISO), Lab M), violet red bile glucose agar (VRBG) (Biokar Diagnostics, Beauvais, France), and Rose-Bengal Chloramphenicol (RBC, Biokar, Zac de ther, France), respectively. The incubations were performed for TVC at 30 °C for 48 h, for LAB at 30 °C for 96 h, for Enterobacteriaceae at 37 °C for 24 h, and for yeasts/molds at 25 °C for 120 h. The measurements were performed twice, and the standard deviations were calculated. Microbial load is expressed as log CFU g−1.Sensory evaluation
Sensory evaluation of table olives was also conducted by eight trained panelists (four males and four females of different ages) throughout the de-bittering and fermentation steps. Each panelist performed an individual blind trial for each studied sample and scored the intensity of bitter taste using a scale ranging from 1 (no bitter taste) to 9 (extremely bitter taste). The mean values of bitter taste were calculated and correlated with the oleuropein content of each sample. The panelists also scored all other organoleptic characteristics of the samples during the fermentation process, such as appearance, aroma, taste, texture and overall acceptability.Statistical analysis
One-way or factorial analysis of variance (ANOVA) was used to evaluate the statistically significant differences and the interaction between the oleuropein content, TPC, quality characteristics and sensory characteristics of all the studied samples from each de-bittering method. The experimental data were analyzed using Statistica 7 (Stat Soft, Tulsa, OK, USA). The comparisons of means were calculated using Duncan's test at the 5% level of significance (p ≤ 0.05).Results and discussion
HP-assisted de-bittering process of table olives – comparison with other techniques
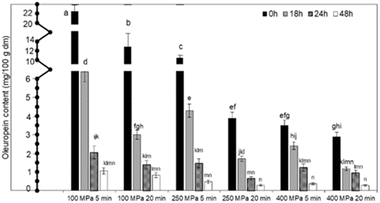
In general, HP led to de-bittered olives in significantly shorter time compared to conventional de-bittering techniques. Oleuropein content was decreased by up to 80% (2.88 mg/100 g d.m.) immediately after HP treatment at the most intense studied conditions (400 MPa for 20 min) compared to the initial oleuropein content (54.94 mg/100 g d.m.) (Fig. 1).HP instantaneously induced the mechanical disruption of the olive cell membranes, leading to higher extractability of intracellular compounds and instant removal of a high proportion of the oleuropein content, thus decreasing the de-bittering time to a few hours. Immediately after HP treatment, total phenolic compounds were also decreased from 30% to 57%, compared to their initial concentration (2.23 mg/g d.m.). Increasing the intensity of HP treatment conditions (from 100 to 400 MPa; from 5 to 20 min) led to higher extraction of oleuropein and TPC. Nevertheless, HP was not a stand-alone process for de-bittering of table olives. It was used as a pretreatment to the conventional Greek-style de-bittering procedure, where the olives were incubated in brine for months.
During incubation of HP pretreated olives in brine (8% w/w NaCl), the oleuropein content continued to decrease, achieving an oleuropein concentration below 1.06 mg/100 g d.m. after 48 h. At the most intense pressures of 400 MPa, oleuropein content was equal to 0.25 mg/100 g d.m. after 48 h incubation in brine. It is noteworthy that the oleuropein content of the conventionally de-bittered samples after 8 weeks (1.96 mg/100 g d.m.) in brine was equally achieved by the HP-assisted de-bittering technique in less than 18 h. TPC of HP de-bittered samples during incubation in brine was stable and ranged from 9.15 to 12.05 mg/g d.m. (Table 1). The antioxidant activity of the samples decreased, reaching a plateau after 24 h incubation in brine at all HP conditions studied, ranging from 8.94 to 13.50 mg Trolox/g. The basic mechanism of olive de-bittering includes the hydrolysis of oleuropein to other non-bitter compounds, such as hydroxytyrosol, via the activity of endogenous enzymes such as β-glycosidase and esterase.41 Cell disruption by HP enhanced the extraction of oleuropein and a part of TPC at zero time and promoted oleuropein hydrolysis to hydroxytyrosol during incubation. This was also confirmed by the hydroxytyrosol content of de-bittered olives, which was increased during incubation at all studied HP conditions. The initial concentration of hydroxytyrosol was 0.19 mg/100 g d.m. and was increased by approximately 40 times (7.28 mg/100 g d.m.) due to oleuropein hydrolysis of HP pretreated samples. In parallel, the lye procedure was performed; the olives were immersed in a solution of 2% NaOH for 8 h, followed by washing with tap water for another 16 h, and the results were compared with those obtained using the HP-assisted de-bittering technique. Although lye de-bittering lasted only 8 h and resulted in decreased oleuropein content (0.5 mg/100 g d.m.), the TPC of de-bittered olives and their antioxidant activity were approximately zero. HP technology has already been used as a pretreatment to assist the extraction process of intracellular compounds, such as phenolic compounds from different plant tissues.24,25,39,42,43 Andreou et al.24 explored HP (600 MPa, 15 min) as a pretreatment to assist the conventional extraction of phenolic compounds from olive pomace, achieving up to 71.8% increase in extraction yield. Chanioti and Tzia,39 successfully combined HP with natural deep eutectic solvents (NADES) and observed that HP enhanced the extractability of oleuropein from olive pomace up to 5 times compared to the conventional extraction method. Andreou et al.25 also used HP (200 MPa, a solid![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water ratio of 1
water ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, and 10 min) to accelerate the de-bittering of olive paste for incorporation in a new food product, which resulted in an oleuropein content reduction of up to 50% compared to the original process. Other novel technologies have also focused on accelerating the de-bittering process and removing phenolic compounds more efficiently. Habibi et al.21 proved that ultrasound-assisted de-bittering was able to decrease de-bittering time (up to 48%) of green table olives, while also reducing NaOH concentration (from 2.0 to 1.5% w/w) and water usage. Tamer et al.22 focused on the combined use of NaOH (1.5% w/w) and NaCl (3% w/w) solutions under vacuum (68 kPa) to accelerate the de-bittering of olives, achieving significant (p < 0.05) shortening of de-bittering duration to 6 h but with a decrease in the TPC (up to 21.43%) and antioxidant activity (up to 59.89%) of the samples.
4, and 10 min) to accelerate the de-bittering of olive paste for incorporation in a new food product, which resulted in an oleuropein content reduction of up to 50% compared to the original process. Other novel technologies have also focused on accelerating the de-bittering process and removing phenolic compounds more efficiently. Habibi et al.21 proved that ultrasound-assisted de-bittering was able to decrease de-bittering time (up to 48%) of green table olives, while also reducing NaOH concentration (from 2.0 to 1.5% w/w) and water usage. Tamer et al.22 focused on the combined use of NaOH (1.5% w/w) and NaCl (3% w/w) solutions under vacuum (68 kPa) to accelerate the de-bittering of olives, achieving significant (p < 0.05) shortening of de-bittering duration to 6 h but with a decrease in the TPC (up to 21.43%) and antioxidant activity (up to 59.89%) of the samples.
| De-bittering technique | Natural | HP-assisted | Lye (2% NaOH for 8 h) | |||||
|---|---|---|---|---|---|---|---|---|
| Incubation in brine (h) | 100 MPa and 5 min | 100 MPa and 20 min | 200 MPa and 5 min | 200 MPa and 20 min | 400 MPa and 5 min | 400 MPa and 20 min | ||
| a “±” represents the standard deviation of triplicates. Different superscript letters indicate significantly different means (p < 0.05) between different incubation times in brine for each de-bittering condition. Different capital letters indicate significantly different means (p < 0.05) between different de-bittering conditions at the same incubation time in brine. | ||||||||
| 0 | 22.35 ± 3.25aA | 15.67 ± 1.08aB | 13.11 ± 1.25aBC | 16.02 ± 2.12aB | 11.60 ± 1.25aCD | 10.26 ± 0.08aCD | 9.50 ± 1.12aD | 0.20 ± 0.08aE |
| 18 | — | 12.05 ± 0.36bAB | 12.77 ± 0.21abA | 11.74 ± 0.76bBC | 11.10 ± 0.85aC | 9.85 ± 0.14aD | 9.48 ± 0.50aD | |
| 24 | — | 12.12 ± 0.15bA | 11.69 ± 0.07abA | 10.97 ± 0.28bB | 10.35 ± 0.14aC | 9.35 ± 0.35aD | 9.12 ± 0.69aD | |
| 48 | — | 11.12 ± 0.07bAB | 11.90 ± 0.21bA | 10.76 ± 0.36bB | 10.22 ± 0.69aB | 9.20 ± 1.12aC | 9.15 ± 0.14aC | |
| 8 weeks | 13.12 ± 2.05b | — | — | — | — | — | — | — |
Quality characteristics of de-bittered table olives
During the de-bittering procedure with all the studied techniques, the brightness (L* value) and yellowness (b* value) of the olives were significantly (p < 0.05) decreased, while the redness was increased (a* value), mainly attributed to the enzymatic activity of polyphenol oxidase (enzymatic browning), which is involved in oxidation reactions producing dark compounds.44 For color parameters of HP de-bittered olives, there were no significant differences among HP conditions; however, there was a color alteration immediately after HP pretreatment, with ΔE values ranging from 8.96 to 13.53 compared to the original raw olives (L* = 59.77, a* = −22.22, b* = 35.52) (Table 2). The findings agree with those reported by Tamer et al.,22 who measured increased a* values but decreased L* and b* values for all the olives processed with NaOH (1.5% w/w) and NaCl (3% w/w) solutions under vacuum (68 kPa). During the incubation in brine, the color of HP de-bittered olives was altered significantly within the first 18 h (ΔE values ranging from 20 to 25) and then remained almost constant for up to 48 h. The color of the control samples altered slowly during the incubation, achieving a ΔE value of 17.75 after 8 weeks. More specifically, the initial bright-green color of raw olives (chroma and hue angle values of 41.90 and 122.03, respectively) was significantly darkened (p < 0.05) immediately after HP, thus the chroma values decreased (32.46–38.40). The chroma value of the conventionally de-bittered olives after 8 weeks of incubation in brine (before the de-bittering process was complete) was significantly decreased (p < 0.05) compared to the initial value (from 41.90 to 34.40); however, it never reached the final dark-green color of the HP-debittered samples. The surface color of the olives darkened over time, which could be attributed to the light-mediated degradation of chlorophyll derivatives such as pheophytin present on olives.| Samples | Color parameters immediately after pretreatment | |||||
|---|---|---|---|---|---|---|
| L* | a* | b* | Chroma | Hue angle | ΔE | |
| a “±” represents the standard deviation of triplicates. Different superscript letters indicate significantly different means (p < 0.05) between different de-bittering conditions at zero time (immediately after pretreatment). | ||||||
| Raw olives | 59.77 ± 3.02a | −22.22 ± 0.53f | 35.52 ± 2.76a | 41.91 ± 2.76a | 122.1 ± 1.2a | 0.00 ± 0.00e |
| 100 MPa/5 min | 51.38 ± 2.16bc | −19.32 ± 1.15de | 32.98 ± 2.36ab | 38.27 ± 1.75ab | 120.4 ± 2.9a | 9.24 ± 0.12cd |
| 250 MPa/5 min | 50.30 ± 2.97c | −15.63 ± 2.01bc | 28.45 ± 3.89b | 32.46 ± 2.33d | 118.8 ± 1.6ab | 13.53 ± 0.25a |
| 400 MPa/5 min | 52.64 ± 1.31bc | −14.71 ± 1.53b | 30.89 ± 1.51ab | 34.21 ± 1.11cd | 115.5 ± 1.3b | 11.35 ± 0.31b |
| 100 MPa/20 min | 50.27 ± 1.95c | −17.35 ± 0.51cd | 30.97 ± 1.30ab | 35.50 ± 0.88bcd | 119.2 ± 0.4a | 11.61 ± 0.08b |
| 250 MPa/20 min | 52.62 ± 1.09bc | −19.74 ± 0.37e | 30.74 ± 0.85ab | 36.52 ± 1.40bc | 122.7 ± 2.3a | 8.96 ± 0.11d |
| 400 MPa/20 min | 55.20 ± 1.20b | −21.10 ± 1.19ef | 35.62 ± 1.81a | 38.40 ± 2.76ab | 120.6 ± 1.2a | 10.71 ± 0.16bc |
| Lye (2% w/w NaOH for 8 h) | 49.52 ± 2.22c | −12.35 ± 1.47a | 32.85 ± 3.45ab | 35.01 ± 2.47bcd | 120.6 ± 2.3a | 12.35 ± 2.50ab |
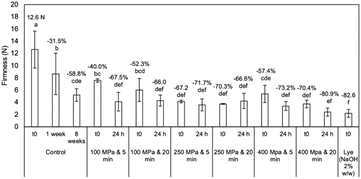
Panagou45 and Tamer et al.22 reported a similar decrease pattern for L* and b* values of green olives within the first 30 days of storage. The firmness of HP-debittered olives decreased (by up to 70.4%) compared to the untreated olives (12.6 N) immediately after HP treatment (Fig. 2).
Within the first 24 h of incubation in brine, a more pronounced decrease in the firmness of HP-debittered olives was observed, while it remained constant for longer durations (>24 h). For pressures of 250 MPa or higher, no significant differences in firmness were observed at zero time and after 24 h of incubation in brine. Similar firmness (no statistically significant difference) reduction was achieved after 8 weeks of conventional de-bittering (5.19 N, −58.9% decrease compared to raw olives). Habibi et al.20 used an ultrasound-assisted (5 kHz, 40 W power, pulse durations 10 min) de-bittering technique to process table olives, and no significant differences were observed for the ΔE and firmness values of ultrasound de-bittered samples compared to the conventional ones. Lye-debittered olives had the lowest firmness after treatment with NaOH, equal to 2.19 N (−82.6% decrease). The results agree with the study of Jimenez, Guillen, Sanchez, Fernandez-Bolanos, and Heredia,46 who stated that the NaOH de-bittering process caused a 35% reduction in olive fruit firmness compared to untreated samples. Regarding the organoleptic characteristics of table olives for all de-bittering techniques, the HP pretreatment reduced the intensity of the bitter taste of olives in significantly shorter times. HP (≥250 MPa) de-bittered samples were characterized as non-bitter (organoleptic scoring < 5) after only 24 h incubation in brine. In contrast, the conventionally de-bittered olives were scored as 8.5 (bitter taste intensity) after 1 week in brine, which was also well related to their oleuropein content (108 mg/100 d.m.). After 8 weeks incubation in brine, the conventionally de-bittered olives may be considered as acceptable (score < 5) based on the panelists' assessment in terms of bitter taste. The results are in accordance with those reported by Habibi et al.,20 who observed that the ultrasound-assisted de-bittering process significantly reduced the essential time for reaching the acceptable threshold of non-bitterness. A positive correlation between oleuropein content reduction and bitterness (R2 = 0.978) was also stated, confirming that the decreased oleuropein level also led to decreased bitterness scores. Pressures equal to or higher than 250 MPa were selected as the HP condition since a significant decrease in oleuropein content (14.57 mg/100 g d.m.) was achieved after only 24 h of incubation in brine. At 100 MPa, the reduction in oleuropein content was not considered satisfactory, considering the corresponding panelists' scores for the olives' bitterness. For longer HP treatment times (20 min), no significant differences were observed (compared to shorter times, i.e. 5 min) in terms of oleuropein content decrease, olives' quality characteristics, and sensory evaluation during the de-bittering step.
Thus, for the subsequent step of olive fermentation, only HP-assisted de-bittering at 250 and 400 MPa for 5 min was considered.
Fermentation of de-bittered table olives
A fermentation step was conducted after each de-bittering technique. The fermentation of table olives was carried out in acidified (pH 6) brine with 8% (w/w) NaCl and lasted 250 days. The end of fermentation was considered when the pH dropped below approximately 4.0, and the product was considered safe. In total, four different samples were studied: (a) natural fermentation (control), (b) HP250 fermentation (HP at 250 MPa for 5 min – as assisted de-bittering step), (c) HP400 fermentation (HP at 400 MPa for 5 min – as assisted de-bittering step) and (d) lye fermentation (2% w/w NaOH for 8 h – as de-bittering step).Population dynamics and pH during the fermentation of table olives
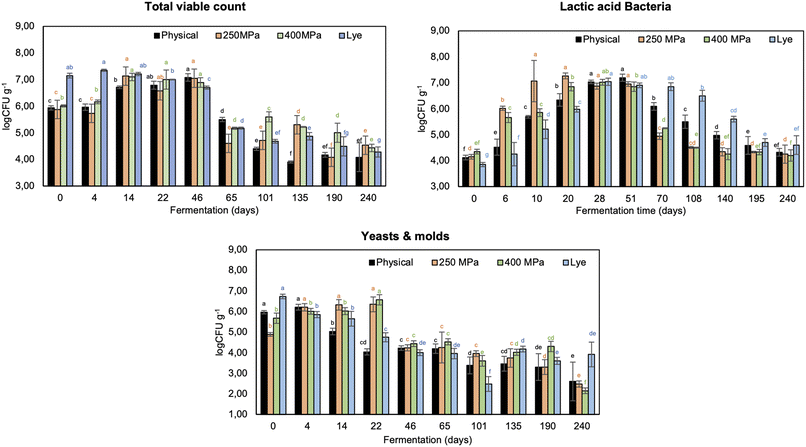
Natural fermentation is a spontaneous complex process, where the activity of the endogenous microflora that pre-exists in olives affects and shapes the quality characteristics and sensory profile of the final fermented table olives.8,47 The initial concentration of NaCl in brine was 8% (w/w), the initial pH was 6.2, and the fermentation temperature was set at 25 °C.48 These parameters are decisive for the progress of fermentation, ensuring that only LAB or yeasts will grow in the brine, avoiding undesired microorganism growth that can cause spoilage.49,50 The initial LAB, yeasts/molds, and Enterobacteriaceae load of the natural fermentation were 4.12, 5.72 and 3.28 log CFU g−1, respectively (Table 3). During the natural fermentation (the de-bittering step is also included), the diffusion phenomena (intracellular nutrients of olives are extracted in the surrounding brine) are slow, thus the microbial growth, and consequently the progress of fermentation, is delayed.4 The initial number of TVC was approximately 6.0 log CFU g−1 and then increased during the first 14 days of fermentation, while a decrease after 46 days was observed (∼5.5–5.0 log CFU g−1). This reduction could be related to the low pH of the brine after the first 30 days of fermentation. Similar trends were observed by Pino, De Angelis, Todaro, Van Hoorde, Randazzo, and Caggia,51 who observed that the initial TVC of brine samples (6.56 log CFU mL−1) was reduced by almost 0.5 and 1.0 log after 30 and 120 days of natural fermentation. At the first stages of natural fermentation, LAB and yeasts coexisted and started to emerge. Typically, under the optimal conditions, the pH would reach 4, and the LAB or yeasts (or both) would consume the nutrients in the medium.49 After the first 14 days of fermentation, LAB prevailed with a mean load of 6.34 log CFU g−1 (Fig. 3).| Zero time of fermentation | ||||
|---|---|---|---|---|
| Microbial load (log CFU g−1) | Natural (control) | Lye-pretreated | HP-pretreated | |
| 250 MPa/5 min | 400 MPa/5 min | |||
| a *ND: not detected; “±” represents the standard deviation of triplicates. Different superscript letters indicate significantly different means (p < 0.05) between different fermentations for each type of microorganism. | ||||
| TVC | 5.96 ± 0.12b | 7.15 ± 0.74a | 5.86 ± 0.26b | 6.01 ± 0.47b |
| LAB | 4.12 ± 0.20ab | 3.85 ± 0.14b | 4.15 ± 0.14ab | 4.35 ± 0.18a |
| Yeasts/molds | 5.72 ± 0.14a | 5.48 ± 0.98a | 5.41 ± 0.69a | 5.15 ± 0.36a |
| Enterobacteriaceae | 3.28 ± 0.08b | 7.02 ± 0.06a | ND* | ND* |
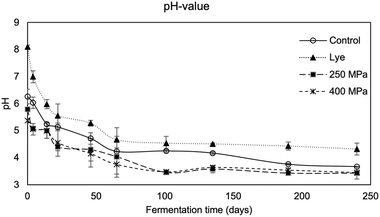
The pH also decreased significantly throughout the fermentation, reaching a value of 4.25 after 101 days (Fig. 4), due to the lactic acid produced by LAB.52,53 This might be attributed to the low NaCl concentration in the brine (<8% w/w), which enhances the growth of LAB.54 Moreover, the yeasts/molds were also associated with the enhancement of LAB growth by their release of nutritive compounds due to their metabolism, such as vitamins or amino acids.55 The yeasts and molds load decreased continuously during fermentation. After 101 d, their population was 3.38 log CFU g−1. The Enterobacteriaceae population was under 3.0 log CFU g−1 throughout the fermentation time. Regarding the lye de-bittering, the initial pH of the brine (pH 8.09) was higher than that observed during natural fermentation (Fig. 4). As a result of this high initial pH, the dominant microorganisms were Enterobacteriaceae, with a load of 7.02 log CFU g−1, at the first stage of fermentation (Fig. 3). The corresponding LAB and yeasts/molds initial loads were 3.85 and 5.48 log CFU g−1, respectively. The first phase of lye fermentation is commonly characterized by a high concentration of Enterobacteriaceae, which is reduced when the pH of the brine is decreased below 6.49 After 30 days of lye fermentation, a significant decrease in the Enterobacteriaceae population (up to 3.5 log CFU g−1 after 248 days) was observed, accompanied by a pH drop (below 5.5), favoring the implantation of LAB. LAB load reached 7.04 log CFU g−1 after 28 days of fermentation (Fig. 3). This last phase of lye fermentation is characterized by the fast growth of LAB and the trend of the pH to reach a value of around 4. LAB communities decreased after 140 days since the fermented substrate was depleted.
The pH decreased continuously, achieving a value of 4.2 after 248 days, which was significantly higher than the corresponding values of other fermentations over the same time (Fig. 4).
The load of yeasts also varied (6.93–3.91 log CFU g−1), which is consistent with results reported by other studies and ranged between 4 and 6 log CFU g−1. The difference in microbial population among the fermentations was reflected in the sensory attributes of the final fermented table olives.56 In the case of HP-assisted de-bittering (250 and 400 MPa, 24 h incubation in 8% w/w brine), followed by fermentation, the results showed that the HP technology did not affect the load of LAB and yeasts/molds for either of the applied pressures. HP induced alterations in the cell membranes of the olives, leading to enhanced mass transfer phenomena. The extractability of intracellular nutrients in brine was more effective and faster than the natural fermentation process, facilitating the LAB and yeasts/molds growth due to the abundance of nutrients in the brine. Nevertheless, HP pretreatment, used as a debittering method to replace lye treatment, promoted the instant release of phenolic compounds into the brine, especially oleuropein and its hydrolysis derivatives, which inhibited LAB growth during the early stages of fermentation because of their antimicrobial and antioxidant activity.4 As a result, at the beginning of the HP400 fermentation, the LAB growth was slower compared to the natural and HP250 fermentations. HP250 fermentation was the fastest compared to the others due to the high LAB growth rate, enhanced by the enriched brine with extracted nutrients from the olives. The increased load of LAB is correlated to the high quality of the final product.48 Despite the slight delay in LAB growth under the more intense high-pressure condition (400 MPa), the decrease in pH during fermentation of both HP-pretreated olives (HP250 and HP400) did not differ significantly (p > 0.05), indicating that the overall fermentation duration was not affected. The pH monitoring showed a clear positive effect for HP-pretreatment. The pH of the brines dropped below 4.2 after approximately 101, 46, 42 and 250 days of fermentation for control, HP250-pretreated, HP400-pretreated and lye-pretreated samples, respectively (Fig. 4). Brines with a pH below 4.5 inhibit the growth of acid-sensitive bacteria and avoid the development of pathogens during fermentation and storage.57 The pH of HP-pretreated samples decreased more rapidly. Therefore, it can be concluded that the different pressures (250 and 400 MPa) did not influence the acidification dynamics and the growth of cultivable microbiota.
Quality parameters of table olives during fermentation
No significant differences in quality characteristics were observed between the final fermented olives (p > 0.05). Immediately after HP pretreatment, there was an initial chroma value and hue angle reduction from 41.9 (control) to ∼32–34 and from 122.1 (control) to ∼115–119, respectively. This reduction in chroma values is attributed to PPO activity. PPO is responsible for enzymatic browning in fruit and vegetables.57 HP treatment disrupted intracellular organelles, resulting in the release of PPO enzyme and its subsequent contact with its phenolic substrates. As a result, the oxidation reactions were accelerated.57 The loss of chroma and hue was higher in lye fermented olives at zero days, confirming faster brownish coloration compared to other physical treatments, making the product less attractive for consumption.58 Nevertheless, at the end of each fermentation, the chroma values ranged from approximately 21.43 to 26.07 for all the final fermented olives. The final fermented olives had a dark-green color that corresponded to hue angles equal to 73.6–74.3° (Table 4). The intensity of enzymatic activity depends on various brine conditions, including sodium concentration and pH.59 Lye de-bittering as a pretreatment step caused the highest decrease in firmness values of olives (5-fold decrease), achieving a firmness of approximately 2.3 N at the end of fermentation (Table 4). A similar trend in the firmness decrease was observed during the lye fermentation of green olives by applying different concentrations of NaOH and KOH.60 Since the lye treatment is a critical stage for determining the firmness of the final fermented table olives, the concentration of the alkali solution and the lye temperature should be controlled.61 HP treatment significantly reduced olive firmness immediately after processing, with decreases of up to 70.4% compared to the untreated sample (12.6 N). However, during fermentation in brine, the firmness of all the samples decreased further, and at the end of the fermentation step, no significant differences were observed between HP-treated and naturally fermented olives (4.89–5.39 N). This indicates that, although HP initially softens the olives, the final texture is preserved after fermentation, maintaining product quality. Regarding the sensory evaluation, all fermented olives had the same scores at the end of each fermentation, in terms of overall impression, flavor and visual appearance. The intensity of the bitter taste was also evaluated by the panelists. The bitter sensation was attributed to the presence of unhydrolyzed oleuropein in the olives. The intensity of the bitter taste of naturally fermented olives was acceptable (<5.0 score) after approximately the first 28 days of fermentation. In the other three fermentations (HP250, HP400 and lye), the intensity of the bitter taste was in the range of acceptable limit from the first day of fermentation since the different pretreatments accelerated the de-bittering process in only a few hours.| Fermentation time (days) | Control | Lye | ||||
|---|---|---|---|---|---|---|
| Chroma | Hue | Firmness | Chroma | Hue | Firmness | |
| a “±” represents the standard deviation of triplicates. Different superscript letters indicate significantly different means (p < 0.05) between de-bittering techniques and fermentation times. | ||||||
| 0 | 41.91 ± 2.76a | 122.1 ± 1.2a | 12.60 ± 2.06a | 26.61 ± 0.25hikl | 99.7 ± 0.2c | 2.48 ± 0.22f |
| 28 | 35.50 ± 2.59bc | 97.6 ± 0.9cd | 9.11 ± 0.69b | 28.61 ± 0.12fghi | 99.4 ± 0.5c | 2.45 ± 0.32f |
| 51 | 34.84 ± 1.59bcd | 92.1 ± 1.3ef | 5.19 ± 1.07cd | 28.96 ± 1.02fghi | 95.5 ± 0.9de | 2.59 ± 0.33f |
| 70 | 37.18 ± 2.23bc | 93.0 ± 1.5e | 5.44 ± 1.13cd | 30.83 ± 0.35efg | 85.5 ± 1.1g | 2.43 ± 0.37f |
| 101 | 28.35 ± 1.97fghik | 88.5 ± 1.4g | 5.52 ± 1.07c | 30.89 ± 0.98efg | 85.9 ± 1.0gi | 2.96 ± 0.32ef |
| 245 | 22.79 ± 0.69mn | 73.6 ± 1.2l | 5.39 ± 0.85cd | 26.07 ± 0.67iklm | 87.1 ± 0.9gi | 2.32 ± 0.36f |
| Fermentation time (days) | HP 250 MPa and 5 min | HP 400 MPa and 5 min | ||||
|---|---|---|---|---|---|---|
| Chroma | Hue | Firmness | Chroma | Hue | Firmness | |
| 0 | 32.46 ± 2.33bc | 118.8 ± 1.6b | 4.15 ± 0.24cde | 34.21 ± 1.11b | 115.5 ± 1.3b | 5.38 ± 0.55cd |
| 28 | 31.80 ± 2.03def | 94.5 ± 2.7de | 4.40 ± 0.52cd | 34.23 ± 1.79cde | 97.6 ± 3.5cd | 4.32 ± 0.52cd |
| 51 | 29.74 ± 1.28fgh | 93.7 ± 1.6e | 5.12 ± 0.68cd | 31.54 ± 1.90defg | 93.9 ± 4.6de | 4.35 ± 0.38cd |
| 70 | 28.09 ± 4.38ghik | 88.8 ± 2.8fg | 4.27 ± 0.54cd | 34.27 ± 0.73cde | 94.9 ± 1.3de | 4.06 ± 0.26de |
| 101 | 25.05 ± 1.65klm | 84.6 ± 3.3i | 4.84 ± 0.77cd | 25.08 ± 0.64klm | 80.0 ± 0.9k | 4.87 ± 0.25cd |
| 245 | 21.43 ± 2.86n | 74.3 ± 4.2l | 5.20 ± 0.45cd | 23.64 ± 2.47lmn | 73.7 ± 0.5l | 4.89 ± 0.12cd |
Selection of optimal HP-assisted de-bittering conditions
The selection of optimal HP conditions for the accelerated de-bittering process was based on: (a) the maximum decrease of oleuropein content in as short a time as possible, (b) minimal effect on the quality of olives, and (c) with maximum organoleptic acceptance in terms of bitter taste.Both HP (250 and 400 MPa for 5 min) pretreatments, as de-bittering steps, significantly accelerated the fermentation step compared to the natural technique. The selected HP conditions were 250 MPa and 5 min at room temperature, which were milder HP conditions compared to 400 MPa. Under these HP conditions, oleuropein hydrolysis was promoted, leading to high-quality final products, while simultaneously shortening the de-bittering step to only 24 h and the fermentation step to 41 days.
Conclusions
The results of this study confirmed that HP processing is efficient and effective for the pretreatment of table olives and accelerates their de-bittering stage. As optimal HP conditions, 250 MPa and 5 min may be selected, based on: (a) the decrease in oleuropein content of the olives in only 24 h, (b) the promotion of LAB growth in brine due to the abundance of extracted nutrients, without affecting the start of fermentation, (c) the decrease in the fermentation duration by half compared to the natural fermentation, and (d) the high quality of the final fermented table olives. Consequently, the application of HP as a de-bittering step is considered to have high potential benefits, minimizing the de-bittering time and fermentation duration of table olives while simultaneously limiting the use of alkali solutions, which is a major drawback for the industry. These findings are directly relevant to table olive producers and food processing industries, who may consider implementing HP technology to accelerate fermentation. The main advantages of HP-assisted de-bittering include its very low energy requirement and environmental sustainability, as it reduces processing time while preserving product quality. HP processing is one of the emerging food technologies that has successfully been scaled up for industrial applications. However, certain limitations must be acknowledged. The high initial investment cost of HP units requires careful techno-economic evaluation to determine whether the time savings justify the expenditure. Additionally, optimization of processing conditions and adaptation to existing production lines are necessary to achieve consistent results at an industrial scale. Despite these considerations, HP technology represents a promising, sustainable approach to accelerating table olive fermentation, balancing environmental and operational efficiency with high-quality product outcomes.Author contributions
Conceptualization: Katsaros, Andreou; methodology: Andreou, Chanioti; software: Stergiou, Andreou, Chanioti; validation: Stergiou, Andreou, Chanioti; formal analysis: Katsaros, Andreou, Chanioti; investigation: Stergiou, Andreou, Chanioti; resources: Katsaros; data curation: Andreou, Chanioti; writing—original draft preparation: Andreou; writing—review and editing: Katsaros, Chanioti; funding acquisition: Katsaros; visualization: Katsaros, supervision: Katsaros; and project administration: Katsaros.Conflicts of interest
There are no conflicts to declare.Data availability
All data supporting the findings of this study are provided within the article. No additional data were generated or deposited in external repositories.Acknowledgements
The authors would like to thank Dr Miltiadis Christopoulos for his generous supply of the raw material-green table olives of Konservolia variety (Amfissa, Greece).References
- K. Mattas and E. Tsakiridou, The functional olive oil market: marketing prospects and opportunities, in Olives and Olive Oil as Functional Foods: Bioactivity, Chemistry and Processing, 2017, p. 647–658 Search PubMed.
- N. Sabatini, E. Perri and V. Marsilio, An investigation on molecular partition of aroma compounds in fruit matrix and brine medium of fermented table olives, Innov. Food Sci. Emerg. Technol., 2009, 10(4), 621–626 CrossRef CAS.
- V. Frisardi, F. Panza, D. Seripa, B. P. Imbimbo, G. Vendemiale and A. Pilotto, et al., Nutraceutical properties of Mediterranean diet and cognitive decline: possible underlying mechanisms, J. Alzheimers Dis., 2010, 22(3), 715–740 CAS.
- R. L. Johnson and A. E. Mitchell, Reducing phenolics related to bitterness in table olives, J. Food Qual., 2018, 2018, 1–8 CrossRef.
- G. Blekas, C. Vassilakis, C. Harizanis, M. Tsimidou and D. G. Boskou, Biophenols in table olives, J. Agric. Food Chem., 2002, 50(13), 3688–3692 CrossRef CAS PubMed.
- G. Bianchi, Lipids and phenols in table olives, Eur. J. Lipid Sci. Technol., 2003, 105(5), 229–242 CrossRef CAS.
- H. J. Wichers, C. Soler-Rivas and J. C. Espín, Oleuropein and related compounds, J. Sci. Food Agric., 2000, 80, 1013–1023 CrossRef.
- P. Conte, C. Fadda, A. Del Caro, P. P. Urgeghe and A. Piga, Table olives: an overview on effects of processing on nutritional and sensory quality, Foods, 2020, 9(4), 514 CrossRef CAS PubMed.
- A. G. Fernández, M. R. Adams and M. J. Fernández-Díez, Table Olives: Production and Processing, Springer, London, 1997 Search PubMed.
- E. Yilmaz and B. Aydeniz, Sensory evaluation and consumer perception of some commercial green table olives, Br. Food J., 2012, 114(8), 1085–1094 CrossRef.
- C. Romero-Segura, C. Sanz and A. G. Perez, Purification and characterization of an olive fruit β-glucosidase involved in the biosynthesis of virgin olive oil phenolics, J. Agric. Food Chem., 2009, 57(17), 7983–7988 CrossRef CAS.
- C. C. Tassou, E. Z. Panagou and K. Z. Katsaboxakis, Microbiological and physicochemical changes of naturally black olives fermented at different temperatures and NaCl levels in the brines, Food Microbiol., 2002, 19(6), 605–615 CrossRef CAS.
- E. Ramírez, E. Medina, P. García, M. Brenes and C. Romero, Optimization of the natural debittering of table olives, LWT--Food Sci. Technol., 2017, 77, 308–313 CrossRef.
- S. Charoenprasert and A. Mitchell, Factors influencing phenolic compounds in table olives (Olea europaea), J. Agric. Food Chem., 2012, 60(29), 7081–7095 CrossRef CAS PubMed.
- L. Cocolin, V. Alessandria, C. Botta, R. Gorra, F. De Filippis and D. Ercolini, et al., NaOH-debittering induces changes in bacterial ecology during table olives fermentation, PLoS One, 2013, 8(7), e69074 CrossRef CAS PubMed.
- M. Brenes and A. de Castro, Transformation of oleuropein and its hydrolysis products during Spanish-style green olive processing, J. Sci. Food Agric., 1998, 77(3), 353–358 CrossRef CAS.
- C. S. Parinos, C. D. Stalikas, T. S. Giannopoulos and G. A. Pilidis, Chemical and physicochemical profile of wastewaters produced from the different stages of Spanish-style green olives processing, J. Hazard. Mater., 2007, 145(1–2), 339–343 CrossRef CAS.
- B. Rincón-Llorente, D. De la Lama-Calvente, M. J. Fernández-Rodríguez and R. Borja-Padilla, Table olive wastewater: problem, treatments and future strategy, Front. Microbiol., 2018, 9, 1641 CrossRef.
- M. Campus, N. Değirmencioğlu and R. Comunian, Technologies and trends to improve table olive quality and safety, Front. Microbiol., 2018, 9, 617 CrossRef.
- M. Habibi, M. T. Golmakani, A. Farahnaky, G. Mesbahi and M. Majzoobi, NaOH-free debittering of table olives using power ultrasound, Food Chem., 2016, 192, 775–781 CrossRef CAS PubMed.
- M. Habibi, M. T. Golmakani, G. Mesbahi, M. Majzoobi and A. Farahnaky, Ultrasound-accelerated debittering of olive fruits, Innov. Food Sci. Emerg. Technol., 2015, 31, 105–115 CrossRef.
- C. E. Tamer, B. İncedayı, B. Yıldız and Ö. U. Çopur, The use of vacuum impregnation for debittering green olives, Food Bioprocess Technol., 2013, 6, 3604–3612 CrossRef CAS.
- V. G. Dourtoglou, A. Mamalos, D. P. Makris and P. Kefalas, Storage of olives under CO2 atmosphere: LC–MS characterization of indices related to changes in polyphenolic metabolism, J. Agric. Food Chem., 2006, 54(6), 2211–2217 CrossRef CAS PubMed.
- V. Andreou, M. Psarianos, G. Dimopoulos, D. Tsimogiannis and P. Taoukis, Effect of pulsed electric fields and high pressure on improved recovery of high-added-value compounds from olive pomace, J. Food Sci., 2020, 85(5), 1500–1512 CrossRef CAS PubMed.
- V. Andreou, S. Chanioti, P. Stergiou and G. Katsaros, Valorization of the olive oil production residue: healthy ingredient for developing high value-added spread, Sustainability, 2021, 13(24), 13984 CrossRef CAS.
- Z. Shouqin, Z. Junjie and W. Changzhen, Novel high pressure extraction technology, Int. J. Pharm., 2004, 278(2), 471–474 CrossRef.
- M. Giannoglou, V. Andreou, I. Thanou, G. Markou and G. Katsaros, High pressure assisted extraction of proteins from wet biomass of Arthrospira platensis: a kinetic approach, Innov. Food Sci. Emerg. Technol., 2022, 81, 103138 CrossRef CAS.
- G. Dimopoulos, A. Limnaios, E. Aerakis, V. Andreou and P. Taoukis, Effect of high pressure on the proteolytic activity and autolysis of yeast Saccharomyces cerevisiae, Innov. Food Sci. Emerg. Technol., 2021, 74, 102865 CrossRef CAS.
- S. Bonatsou, C. C. Tassou, E. Z. Panagou and G. J. E. Nychas, Table olive fermentation using starter cultures with multifunctional potential, Microorganisms, 2017, 5(2), 30 Search PubMed.
- A. I. Doulgeraki, O. Hondrodimou, V. Iliopoulos and E. Z. Panagou, Lactic acid bacteria and yeast heterogeneity during storage of natural black olives, Food Control, 2012, 26(1), 49–57 CrossRef CAS.
- R. Tofalo, G. Perpetuini, M. Schirone, A. Ciarrocchi, G. Fasoli and G. Suzzi, et al., Lactobacillus pentosus dominates spontaneous fermentation of Italian table olives, LWT--Food Sci. Technol., 2014, 57(2), 710–717 CrossRef CAS.
- G. Perpetuini, R. Prete, N. Garcia-Gonzalez, M. K. Alam and A. Corsetti, Table olives more than a fermented food, Foods, 2020, 9(2), 178 CrossRef CAS PubMed.
- F. Gutiérrez, B. Jimenez, A. Ruiz and M. A. Albi, Effect of olive ripeness on oxidative stability of virgin olive oil, J. Agric. Food Chem., 1999, 47(1), 121–127 Search PubMed.
- M. M. Özcan and N. Uslu, Effect of debittering process by lye instead of NaOH on phenolic components and sensory properties of olives, Food Human., 2023, 1, 227–240 CrossRef.
- M. B. Maldonado, C. A. Zuritz, A. D. Gascón and E. Rey, Sodium diffusion in green olives during debittering, Grasas Aceites, 2003, 54(4), 358–364 Search PubMed.
- M. B. Maldonado, C. A. Zuritz, R. G. Wuilloud, C. R. Bageta, J. Terreni and M. J. Sánchez, Diffusion phenomena during debittering of green olives, Grasas Aceites, 2011, 62(1), 39–48 CrossRef CAS.
- O. Folin and V. Ciocalteu, On tyrosine and tryptophane determinations in proteins, J. Biol. Chem., 1927, 73(2), 627–650 Search PubMed.
- W. Brand-Williams, M. E. Cuvelier and C. Berset, Use of a free radical method to evaluate antioxidant activity, LWT--Food Sci. Technol., 1995, 28(1), 25–30 Search PubMed.
- S. Chanioti and C. Tzia, Extraction of phenolic compounds from olive pomace using natural deep eutectic solvents, Innov. Food Sci. Emerg. Technol., 2018, 48, 228–239 Search PubMed.
- V. Andreou, E. Kourmbeti, G. Dimopoulos, M. Psarianos, G. Katsaros and P. Taoukis, Optimization of virgin olive oil yield and quality using nonthermal processing, Food Bioprocess Technol., 2022, 15(4), 891–903 CrossRef CAS.
- E. Ramírez, M. Brenes, P. García, E. Medina and C. Romero, Oleuropein hydrolysis in natural green olives, Food Chem., 2016, 206, 204–209 CrossRef PubMed.
- K. N. Prasad, B. Yang, M. Zhao, N. Ruenroengklin and Y. Jiang, Ultrasonication or high-pressure extraction of flavonoids from litchi pericarp, J. Food Process. Eng., 2009, 32(6), 828–843 CrossRef.
- M. Corrales, S. Toepfl, P. Butz, D. Knorr and B. Tauscher, Extraction of anthocyanins from grape by-products assisted by emerging technologies, Innov. Food Sci. Emerg. Technol., 2008, 9(1), 85–91 Search PubMed.
- V. Marsilio, C. Campestre and B. Lanza, Phenolic compounds change during California-style ripe olive processing, Food Chem., 2001, 74(1), 55–60 CrossRef CAS.
- E. Z. Panagou, Effect of packing treatments on characteristics of untreated green olives, J. Sci. Food Agric., 2004, 84, 757–764 CrossRef CAS.
- A. Jimenez, R. Guillen, C. Sanchez, J. Fernandez-Bolanos and A. Heredia, Changes in texture and cell wall polysaccharides during Spanish-style processing, J. Agric. Food Chem., 1995, 43(8), 2240–2246 Search PubMed.
- B. Lanza, Abnormal fermentations in table-olive processing, Front. Microbiol., 2013, 4, 91 Search PubMed.
- A. Corsetti, G. Perpetuini, M. Schirone, R. Tofalo and G. Suzzi, Application of starter cultures to table olive fermentation, Front. Microbiol., 2012, 3, 248 Search PubMed.
- A. Hurtado, C. Reguant, A. Bordons and N. Rozés, Lactic acid bacteria from fermented table olives, Food Microbiol., 2012, 31, 1–8 Search PubMed.
- M. Alves, T. Gonçalves and C. Quintas, Microbial quality and yeast dynamics in cracked green table olives, Food Control, 2012, 23(2), 363–368 CrossRef CAS.
- A. Pino, M. De Angelis, A. Todaro, K. Van Hoorde, C. L. Randazzo and C. Caggia, Fermentation of Nocellara Etnea olives by functional starters, Front. Microbiol., 2018, 9, 1125 CrossRef.
- G. Perpetuini, G. Caruso, S. Urbani, M. Schirone, S. Esposto and A. Ciarrocchi, et al., Changes in polyphenols of table olives under different irrigation regimes, Front. Microbiol., 2018, 9, 1287 Search PubMed.
- A. H. Sánchez, A. De Castro, L. Rejano and A. Montaño, Chemical changes in olive juice and brine during fermentation, J. Agric. Food Chem., 2000, 48(12), 5975–5980 CrossRef PubMed.
- M. Poiana and F. V. Romeo, Chemical and microbiological changes during natural fermentation of olives, Grasas Aceites, 2006, 57(4), 402–408 CrossRef CAS.
- J. L. Ruiz-Barba, J. C. Piard and R. Jiménez-Díaz, Plasmid profiles of Lactobacillus plantarum from olive fermentation, J. Appl. Bacteriol., 1991, 71, 417–421 Search PubMed.
- A. Mastralexi, F. T. Mantzouridou and M. Z. Tsimidou, Evolution of safety and quality parameters of Greek PDO table olives, Eur. J. Lipid Sci. Technol., 2019, 121(3), 1800171 CrossRef.
- M. De Angelis, D. Campanella, L. Cosmai, C. Summo, C. G. Rizziello and F. Caponio, Microbiota and metabolome of Greek-type olive fermentation, Food Microbiol., 2015, 52, 18–30 CrossRef CAS PubMed.
- E. Ramírez, B. Gandul-Rojas, C. Romero, M. Brenes and L. Gallardo-Guerrero, Pigments and colour changes in green table olives, Food Chem., 2015, 166, 115–124 Search PubMed.
- C. Fadda, A. Del Caro, A. M. Sanguinetti and A. Piga, Texture and antioxidant evolution of green table olives, Grasas Aceites, 2014, 65, e002 CrossRef.
- P. García-Serrano, B. de Los Santos, A. H. Sánchez, C. Romero, A. Aguado and P. García-García, et al., Progress on green table olive processing with KOH, Sci. Total Environ., 2020, 746, 141150 CrossRef PubMed.
- S. J. Carmona, A. de Castro and L. R. Navarro, Traditional process of green table olives: rationalization of alkaline treatment, Grasas Aceites, 2011, 62(4), 375–382 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |