Open Access Article
Open Access ArticleOptimization and greenness evaluation of extraction methods of bioactive compounds from cocoa bean shells (Theobroma cacao L.)
Erick Alvarez-Yanamango *abc,
Milagros Huaytalla-Ramirez
*abc,
Milagros Huaytalla-Ramirez b,
Nereyda Sarmiento-Perez
b,
Nereyda Sarmiento-Perez b,
Gerald Chumpitaz-Huanqui
b,
Gerald Chumpitaz-Huanqui bc,
Luis Napan-Tacca
bc,
Luis Napan-Tacca b,
Daniel Obregon
b,
Daniel Obregon b and
Alfredo Ibáñez
b and
Alfredo Ibáñez c
c
aDepartamento de Ingeniería, Pontificia Universidad Católica Del Perú, San Miguel, Lima, Peru. E-mail: erick.alvarez@pucp.edu.pe
bGrupo de Investigación en Tecnologías y Procesos Agroindustriales (ITEPA PUCP), Pontificia Universidad Católica Del Perú, San Miguel, Lima, Peru
cInstituto de Ciencias Ómicas y Biotecnología Aplicada (ICOBA PUCP), Pontificia Universidad Católica Del Perú, San Miguel, Lima, Peru
First published on 21st April 2026
Abstract
In this study, the efficiency of two green technologies, ultrasound-assisted extraction (CBS-UAE) and microwave-assisted extraction (CBS-MAE), was optimized and compared for the recovery of bioactive compounds from cocoa bean shells (CBSs). Response surface (I-optimal) experimental designs were applied, considering variables such as temperature, time, solid–liquid ratio, particle size, and power. Total phenol content (TPC) and antioxidant capacity (DPPH = 2,2-diphenyl-1-picrylhydrazyl, ABTS = 2,2′-azino-bis(3-ethylbenzothiazoline)-6-sulfonic acid, and FRAP = ferric reducing antioxidant power) were analyzed. CBS-UAE showed higher antioxidant activity, as measured by the ABTS (265.8 ± 5.1 mg TE per g CBS extract b.s.) and FRAP (212.86 ± 4.95 mg TE per g CBS extract b.s.) assays, than CBS-MAE. Optimal conditions obtained for UAE were 65 °C, 45 min, a ratio of 70 mL g−1, and a fine particle size (75–150 µm). While both methods yielded similar TPC and DPPH profiles, UAE achieved the highest antioxidant activity in the ABTS and FRAP assays, whereas MAE drastically reduced the extraction time. UHPLC–HRMS (Q-Exactive–MS/MS) analysis of the optimal CBS-UAE/CBS-MAE extracts mainly identified methylxanthines (theobromine and caffeine), amino acids (L-phenylalanine and arginine), and organic acids (mannitol). The environmental impact of the extraction technique was validated using AGREE metrics, confirming the ecological sustainability of MAE and UAE with AGREE scores of 0.62/1.00 and 0.55/1.00, respectively. These results validate the use of cocoa by-products via clean, sustainable technologies, thereby adding value to these residues.
Sustainability spotlightCocoa bean shells (CBSs) are abundant agro-industrial residues whose valorization is essential to reduce waste and advance circular bioeconomy practices. This study optimizes two green extraction technologies—ultrasound (UAE) and microwave-assisted extraction (MAE)—to recover antioxidant compounds using energy-efficient, low-solvent processes. A response surface design enabled an efficient, robust evaluation of extraction variables, resulting in environmentally favorable conditions. Both methods showed positive AGREE scores (0.62 for MAE and 0.55 for UAE) and produced comparable extracts rich in methylxanthines, organic acids, and amino acids with potential nutraceutical applications. By enabling high-value recovery from cocoa waste and minimizing environmental impact, this work supports SDG 12 (Responsible Consumption and Production), SDG 9 (Industry, Innovation and Infrastructure), and SDG 3 (Good Health and Well-Being). |
Introduction
In recent years, waste generated by the food industry has been valorized to develop circular-economy processes. In the chocolate industry, the most exploited fraction of cocoa is the cotyledon after the bean is subjected to fermentation, drying, roasting, and shelling, generating as waste the shell that represents between 10 and 20% of the cocoa bean.1,2 According to the International Cocoa Organization (ICCO), in the period 2021/2022 cocoa bean production reached 4.8 million tons, and it is estimated that by the period 2022/2023 it will have reached 5 million tons of cocoa.3 Based on these data, the estimated global production of cocoa bean shells (CBSs) is between 480![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 and 960
000 and 960![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons per year (10–20%).
000 tons per year (10–20%).
As shown in Fig. 1, CBSs are an important source of phytochemicals, including bioactive substances such as methylxanthines and polyphenolic compounds, as well as fiber, volatile cocoa compounds, proteins, minerals, and vitamins, which would allow it to be considered as an additive for innovative and functional foods.4 The polyphenols present in CBSs are notable for their high antioxidant activity, as they act as enzyme modulators and inhibitors. These compounds have demonstrated the ability to prevent the progression of various diseases, with potential antidiabetic, anti-inflammatory, and cardioprotective effects.5–7 In particular, methylxanthines are associated with improved cardiovascular function and a lower risk of neurodegenerative disorders, such as Alzheimer's and Parkinson's diseases, as well as metabolic conditions, including obesity and diabetes.8,9 Therefore, CBS extracts represent a promising source of bioactive compounds for the development of nutraceutical products, i.e., concentrated forms of these phytochemicals intended to supplement the diet and provide health benefits beyond basic nutrition, particularly in the prevention and management of chronic diseases.4,10
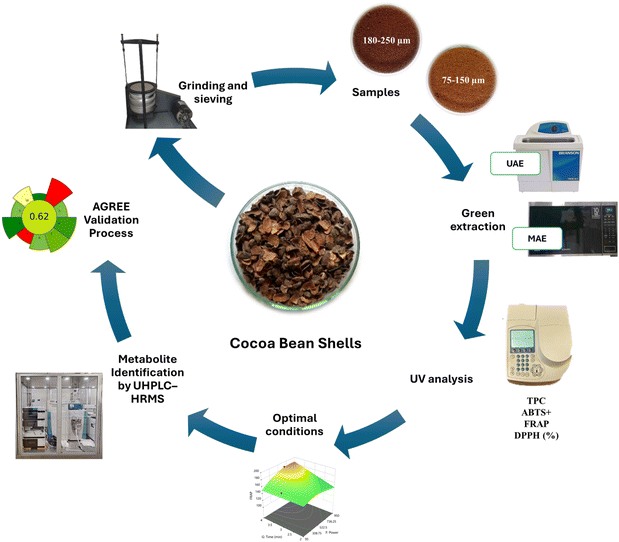 | ||
| Fig. 1 Whole cocoa bean shell treatment from grinding and sieving, green extraction, analysis, optimization, and AGREE validation. | ||
Traditional extraction techniques offer simplicity, widespread availability, and low cost; however, they are constrained by inefficient penetration of solvents into plant cell walls and the potential toxicity of the solvents used. Ultrasonic probe cavitation disrupts cell walls via microjets and shockwaves; however, scaling remains challenging because probes offer high intensity but limited volume and thermal control, whereas ultrasonic baths provide uniformity at lower intensity over large volumes. Meanwhile, microwave-assisted extraction (MAE) uses temperature-induced diffusion and localized heating to rupture cells. Both technologies thus represent effective, scalable options for bioactive compound recovery, each with distinct advantages depending on the process requirements.11
This study optimizes and compares two green extraction technologies—ultrasound-assisted extraction (UAE) and microwave-assisted extraction (MAE), for recovering bioactive compounds from cocoa bean shells (CBSs). Using response surface methodology, key process parameters were evaluated to maximize total phenolic content and antioxidant capacity. In addition to phytochemical profiling by UHPLC–HRMS, environmental sustainability was quantitatively assessed using AGREE metrics. The results enable selection of the most suitable technology based on the target outcome, maximizing antioxidant activity or minimizing processing time, supporting CBS valorization through clean, scalable processes.
Materials and methods
Chemical reagents
The chemicals used in the sample analysis were analytical grade chemicals, including Folin&Ciocalteu's phenol reagent, sodium carbonate (≥99.5%), 2,2′-diphenyl-1-picryl hydrazyl (95%; DPPH), 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (97%; trolox), (+)-catechin hydrate (>98%), methanol (≥99.9%), hydrochloric acid (37%), aluminium chloride (99%), sodium nitrite (≥99%), formic acid (≥98%) and quercetin (≥98.5%) acquired from Sigma-Aldrich (St. Louis, Missouri, USA); isotopically labeled standards for LC-MS/MS (>99%), 3,7-dimethylxanthine-(dimethyl-D6) and caffeine-(trimethyl-13C3) were obtained from Merck (Sigma-Aldrich and Supelco, respectively). In addition, the chemicals used in the extraction process, including food-grade ethanol, were purchased from Colaromo SAC (Lima, Peru).Raw material preparation
The CBS sample was obtained from fine-aroma cocoa of Amazonian origin (Theobroma cacao L.), the Criollo variety, provided by Kuyay Chocolates, located in the Jahuanga zone, Bagua Grande district, Utcubamba province, Amazonas region, Peru (5°46′19.6″ S, 78°32′57.4″ W, 605 m altitude). The cocoa bean shells were obtained after fermentation in handmade wooden crates, drying in the shade, and roasting with a heat ramp from 90 to 120 °C for 50 minutes, followed by mechanical shelling.To obtain the study sample, the shells were ground to a powder using a knife mill (MKM6003, Bosch, Slovenia) for 30 s, and then passed through 60-, 80-, 100-, and 200-mesh sieves (ASTM E11, Retsch, Germany). The obtained fractions were used as samples with particle sizes of 75–150 µm and 180–250 µm. The samples were vacuum-packed in trilaminate bags and stored at −20 °C until further analysis (Fig. 1).
Green extraction process of CBS bioactives
In both cases, the UAE-and MAE-assisted extracts were separated only by centrifugation at 5000 rpm for 10 minutes. The obtained supernatant was made up to 40 mL and stored in amber vials at freezing temperatures. Before analysis, the supernatant was centrifuged again, and filtered through Whatman 41 paper (20–25 µm pore size), and then through a 0.22 µm syringe filter.| Factor name | Type | Min | Max | Coded | |
|---|---|---|---|---|---|
| Low | High | ||||
| Temperature (A) | Numeric | 60 | 80 | −1 | +1 |
| Time (B) | Numeric | 45 | 90 | −1 | +1 |
| Ratio (C) | Numeric | 60 | 80 | −1 | +1 |
| Granulometry (D) | Categoric | A | B | −1 | +1 |
| Factor name | Type | Min | Max | Coded | |
|---|---|---|---|---|---|
| Low | High | ||||
| Ratio (E) | Numeric | 60 | 80 | −1 | +1 |
| Power (F) | Numeric | 95 | 950 | −1 | +1 |
| Time (G) | Numeric | 2 | 4 | −1 | +1 |
| Granulometry (H) | Categoric | A | B | −1 | +1 |
Spectrophotometric assays
Absorbance was measured using a spectrophotometer, Thermo Scientific, Genesys 10S UV-VIS, USA, in all cases.The DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging assay was performed as described by Brand William et al. (1995).13 The DPPH radical activity of the CBS extract was obtained after mixing 100 µL of CBS extract with 3.9 mL of 500 µM DPPH. The mixture was shaken and left to stand in the dark for 30 minutes, and then the absorbance was measured at 515 nm. The radical scavenging activity was calculated using eqn (1), where the control represents the absorbance of the DPPH reagent blank (without extract), and the sample is the absorbance of the DPPH solution in the presence of the CBS extract.
 | (1) |
ABTS (2,20-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid)) radical scavenging was carried out as proposed by Re et al. (1999).14 It consists of mixing 100 µL of CBS extract with 2 mL of ABTS solution (7 mM), incubating in the dark for 6 minutes, and measuring the absorbance at 734 nm. ABTS values were expressed as micrograms of trolox equivalents per gram of CBS extract on a dry-weight basis (mg TE per g CBS d.w), using a trolox standard curve at concentrations of 5 and 150 µg mL−1.
The ferric reducing antioxidant power (FRAP) assay was performed following the methodology proposed by Benzie & Strain (1996).15 To 100 µL of CBS extract, 1.9 mL of FRAP solution is added, and the mixture is incubated in the dark for 30 minutes, with absorbance read at 593 nm. FRAP values were expressed in micrograms of trolox equivalent per gram of CBS extract on a dry weight basis (mg TE per g CBS d.w), using a trolox standard curve at concentrations of 10 and 100 µg mL−1, respectively.
DPPH, ABTS, and FRAP are widely used in vitro electron-transfer-based assays that evaluate chemical reducing power and radical-scavenging capacity rather than direct biological antioxidant activity.16,17 As these methods rely on synthetic radicals and nonphysiological conditions and do not account for bioavailability, their results should be considered preliminary.18,19 Therefore, further validation through cellular antioxidant assays (e.g., ORAC) or in vivo studies is required to confirm their physiological relevance.20
Identification of metabolites by liquid chromatography-mass spectrometry (UHPLC-MS/MS)
The identification of phenolic compounds and methylxanthines in the MAE/UAE extract was carried out by UHPLC–Q-Orbitrap–MS/MS following the methodology adapted from a previous study.21 An ultra-high-performance liquid chromatograph (UHPLC Dionex UltiMate 3000, Thermo Fisher Scientific, Waltham, MA, USA) equipped with an Acclaim™ C18 reversed-phase HPLC column (5 µm, 4.6 × 150 mm) was used, with a flow rate of 0.7 mL min−1. The mobile phases were 0.1% formic acid in water (A) and 90% acetonitrile in water (B). The gradient program was: −3.6–1.0 min, 15% B; 1.0–25.0 min, linear increase to 95% B; 25.0–26.0 min, increase to 100% B; 26.0–27.0 min, hold at 100% B. The ESI source (HESI II, Thermo Fisher Scientific, Waltham, MA, USA) was operated in positive ion mode (ESI+) for alkaloid analysis and in negative ion mode (ESI−) for the phenolic profile, at 2.0 and 1.5 kV, respectively, using data-dependent acquisition (DDA).Environmental impact assessment of the extraction process
The environmental impact of MAE- and UAE-assisted extraction processes was assessed using the innovative quantitative environmental metrics tool AGREEprep, proposed by Wojnowski et al.(2022), which is based on 10 principles of Green Analytical Chemistry, including criteria such as solvent type, sample size, and energy consumption.22,23 The open-access AGREEprep software was available at mostwiedzy.pl/AGREEprep or via the online emulator at the https://www.agreeprep.anvil.app website. The quantitative results of the tool are presented as pictograms, with scores from 0 to 1 represented by colors ranging from red to green. Red indicates a high negative impact, and green indicates a process that meets the green chemistry criteria.The following criteria were considered in the CBS bioactive extraction process. Criterion 1: the CBS extraction process is realized prior to the analytical test; therefore, ex situ preparation was considered. Criterion 2: ethanol was used as a solvent, avoiding the use of methanol and hydrochloric acid, among others. Additionally, up to 75% of the solvent was recovered, enabling a sustainable process (criterion 3). Criterion 4: products derived from the separation of bioproducts after centrifugation are considered waste. Criterion 5: the amount of CBS needed to obtain one liter of extract in one hour is considered under optimal MAE and UAE conditions. Criterion 6: the maximum capacity of the equipment used in an extraction batch (e.g., mL per batch or units per batch). Criterion 7: this consisted of the number of steps in the extraction process, as well as the level of instrumentation from sample preparation to extraction. Criterion 8: the maximum energy output required to obtain a batch of CBS extract was considered. Criterion 9: the analytical technique and instrumentation were defined to validate the presence of metabolites in the extract. Criterion 10: operating conditions based on moderate/high temperatures for extraction, but below the boiling point of the solvents.
Statistical assay
Stat-Ease® software from Stat-Ease, Inc. (MN, USA) was used for statistical analysis. A matrix was generated using optimal response surface methodology (RSM). Optimal designs are recommended when categorical and numeric factors are modeled using a custom model or a cubic or higher-order model.24The optimization of the UAE extraction was achieved using an optimal response surface design with three continuous numerical factors: extraction time (min), temperature (°C), and S–L ratio (mL g−1), and a nominal categorical factor, granulometry of the CBS fraction, with two levels: A (75–150 µm) and B (180–250 µm) (Table 1). On the other hand, for microwave-assisted extraction (MAE), the ratios of the sample to solvent (mL g−1), power (W), time (min), and granulometry with two levels: A (75–150 µm) and B (180–250 µm), were selected (Table 2).
The optimized extraction conditions (UAE and MAE) were validated for maximum TPC, TFC, and in vitro antioxidant activities (DPPH, FRAP, and ABTS) using RSM-derived values. All responses were again determined under the optimized extraction conditions. The experimental values were compared with the model-predicted values to assess the model's validity.
Results and discussion
ANOVA analysis
The experimental design consisted of 24 runs, as shown in Table 3, with the responses TPC, DPPH, ABTS, and FRAP presented. Response values varied widely across all experiments: TPC ranged from 94 to 191 mg GAE per g, DPPH from 35 to 76%, ABTS from 111 to 274 mg TE per g, and FRAP from 124 to 253 mg TE per g, indicating a strong influence of extraction parameters. The influence of the parameters on the response was evaluated using analysis of variance (ANOVA), with the corresponding sum of squares (SS), degrees of freedom (df), and mean squares (MS). A p-value lower than 0.05 means that the model term is significant, whereas values higher than 0.100 are considered not significant. Furthermore, hierarchical ordering is used to assess model terms with p-values > 0.05.| Run order | UAE – factors | Response | MAE – factors | Response | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A | B | C | D | TPC | DPPH | ABTS | FRAP | E | F | G | H | TPC | DPPH | ABTS | FRAP | |
| T (°C) | t (min) | Ratio | Gr (µm) | Ratio | Power | t (min) | Gr (µm) | |||||||||
| 1 | 60 | 90 | 60 | B | 132 | 61 | 179 | 158 | 69 | 665 | 3 | B | 142 | 41 | 158 | 168 |
| 2 | 76 | 80 | 71 | B | 158 | 61 | 247 | 194 | 60 | 665 | 4 | A | 147 | 64 | 188 | 186 |
| 3 | 80 | 45 | 72 | B | 165 | 61 | 260 | 208 | 65 | 950 | 4 | B | 128 | 59 | 153 | 161 |
| 4 | 80 | 45 | 60 | A | 160 | 62 | 221 | 192 | 69 | 665 | 3 | B | 129 | 47 | 130 | 160 |
| 5 | 60 | 59 | 66 | B | 121 | 52 | 170 | 152 | 80 | 950 | 2 | B | 126 | 45 | 134 | 166 |
| 6 | 72 | 45 | 80 | A | 143 | 53 | 211 | 158 | 66 | 570 | 2 | A | 149 | 55 | 187 | 194 |
| 7 | 70 | 68 | 70 | A | 166 | 63 | 241 | 210 | 60 | 95 | 3 | A | 122 | 51 | 141 | 165 |
| 8 | 80 | 90 | 80 | A | 134 | 55 | 210 | 148 | 77 | 95 | 2 | A | 140 | 42 | 154 | 185 |
| 9 | 70 | 90 | 80 | B | 143 | 52 | 219 | 166 | 80 | 570 | 3 | A | 146 | 53 | 202 | 163 |
| 10 | 70 | 45 | 60 | B | 144 | 76 | 211 | 195 | 80 | 570 | 3 | A | 170 | 54 | 200 | 181 |
| 11 | 80 | 90 | 60 | B | 115 | 52 | 179 | 135 | 60 | 950 | 2 | B | 115 | 53 | 134 | 124 |
| 12 | 80 | 64 | 80 | B | 100 | 38 | 130 | 127 | 70 | 950 | 3 | A | 150 | 60 | 196 | 163 |
| 13 | 60 | 83 | 72 | B | 102 | 47 | 132 | 132 | 77 | 760 | 2 | A | 158 | 47 | 134 | 186 |
| 14 | 60 | 90 | 80 | A | 148 | 51 | 206 | 187 | 70 | 950 | 3 | A | 153 | 72 | 194 | 168 |
| 15 | 80 | 73 | 62 | A | 123 | 60 | 193 | 169 | 72 | 190 | 4 | A | 131 | 49 | 115 | 144 |
| 16 | 68 | 90 | 60 | A | 153 | 53 | 223 | 211 | 77 | 950 | 3 | B | 124 | 52 | 111 | 146 |
| 17 | 62 | 46 | 72 | A | 157 | 51 | 240 | 210 | 69 | 665 | 3 | B | 94 | 47 | 147 | 151 |
| 18 | 60 | 45 | 80 | B | 138 | 57 | 203 | 179 | 61 | 190 | 2 | A | 105 | 45 | 145 | 146 |
| 19 | 80 | 90 | 60 | B | 115 | 57 | 171 | 156 | 80 | 950 | 4 | B | 101 | 52 | 161 | 170 |
| 20 | 80 | 90 | 80 | A | 139 | 41 | 201 | 174 | 80 | 190 | 3 | B | 147 | 35 | 127 | 150 |
| 21 | 80 | 45 | 72 | B | 148 | 48 | 209 | 190 | 65 | 190 | 2 | B | 105 | 40 | 136 | 136 |
| 22 | 60 | 45 | 80 | B | 169 | 45 | 238 | 214 | 75 | 475 | 4 | B | 151 | 40 | 131 | 141 |
| 23 | 60 | 90 | 80 | A | 191 | 47 | 274 | 252 | 61 | 190 | 4 | B | 124 | 38 | 129 | 133 |
| 24 | 60 | 45 | 60 | A | 178 | 61 | 262 | 253 | 72 | 190 | 4 | A | 182 | 39 | 164 | 160 |
It can be observed that the operating conditions of UAE and MAE influence the responses of phenol, flavonoid, and FRAP content. On the other hand, variations in DPPH and ABTS responses do not significantly affect the operating conditions. ANOVA evaluation of phenols, flavonoids, and FRAP is presented in Tables 4–11.
| Source | SS | df | MS | F-value | p-value | |
|---|---|---|---|---|---|---|
| Model | 4688.56 | 3 | 1563 | 4.15 | 0.0194 | Significant |
| B-t (min) | 1241.06 | 1 | 1241 | 3.29 | 0.0846 | |
| D-Gr (µm) | 2199.24 | 1 | 2199 | 5.84 | 0.0254 | |
| B2 | 1181.16 | 1 | 1181 | 3.13 | 0.0919 | |
| Residual | 7537.40 | 20 | 377 | |||
| Lack of fit | 5995.69 | 15 | 400 | 1.30 | 0.4151 | Not significant |
| Pure error | 1541.72 | 5 | 308 | |||
| Cor total | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 225.97 225.97 |
23 |
| Source | SS | df | MS | F-value | p-value | |
|---|---|---|---|---|---|---|
| Model | 6872.76 | 6 | 1145.46 | 4.64 | 0.0058 | Significant |
| E-ratio | 1311.57 | 1 | 1311.57 | 5.31 | 0.0341 | |
| F-power | 11.63 | 1 | 11.63 | 0.0471 | 0.8308 | |
| G-t (min) | 560.98 | 1 | 560.98 | 2.27 | 0.1502 | |
| H-Gr (µm) | 3071.26 | 1 | 3071.26 | 12.43 | 0.0026 | |
| EF | 1118.46 | 1 | 1118.46 | 4.53 | 0.0483 | |
| FG | 901.01 | 1 | 901.01 | 3.65 | 0.0732 | |
| Residual | 4200.88 | 17 | 247.11 | |||
| Lack of fit | 1377.89 | 12 | 114.82 | 0.2034 | 0.9889 | Not significant |
| Pure error | 2822.99 | 5 | 564.6 | |||
| Cor total | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 073.64 073.64 |
23 |
| Source | SS | df | MS | F-value | p-value | |
|---|---|---|---|---|---|---|
| Model | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 042 042 |
3 | 3680.66 | 4.73 | 0.0119 | Significant |
| A-T (°C) | 3926.82 | 1 | 3926.82 | 5.05 | 0.0361 | |
| D-Gr (µm) | 4531.66 | 1 | 4531.66 | 5.82 | 0.0255 | |
| AD | 3711.13 | 1 | 3711.13 | 4.77 | 0.0411 | |
15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 566 566 |
20 | 778.32 | ||||
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 155 155 |
15 | 810.35 | 1.19 | 0.4593 | ||
| Residual | 3411.18 | 5 | 682.24 | |||
| Lack of fit | 26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 608 608 |
23 | Not significant | |||
| Pure error | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 042 042 |
3 | 3680.66 | 4.73 | 0.0119 | |
| Cor total | 3926.82 | 1 | 3926.82 | 5.05 | 0.0361 |
| Source | SS | df | MS | F-value | p-value | |
|---|---|---|---|---|---|---|
| Model | 5524.89 | 7 | 789.27 | 5.71 | 0.0019 | Significant |
| E-ratio | 279.86 | 1 | 279.86 | 2.02 | 0.174 | |
| F-power | 689.5 | 1 | 689.5 | 4.99 | 0.0402 | |
| G-t (min) | 98.74 | 1 | 98.74 | 0.7142 | 0.4105 | |
| H-Gr (µm) | 2215.38 | 1 | 2215.38 | 16.02 | 0.001 | |
| EG | 1008.24 | 1 | 1008.24 | 7.29 | 0.0158 | |
| FG | 892.06 | 1 | 892.06 | 6.45 | 0.0218 | |
| F2 | 490.39 | 1 | 490.39 | 3.55 | 0.078 | |
| Residual | 2212.12 | 16 | 138.26 | |||
| Lack of fit | 1761.06 | 11 | 160.1 | 1.77 | 0.2735 | Not significant |
| Pure error | 451.06 | 5 | 90.21 | |||
| Cor total | 7737.01 | 23 |
| Source | SS | df | MS | F-value | p-value | |
|---|---|---|---|---|---|---|
| Model | 5190.87 | 1 | 5190.87 | 4.33 | 0.0492 | Significant |
| D-Gr (µm) | 5190.87 | 1 | 5190.87 | 4.33 | 0.0492 | |
| Residual | 26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 349.5 349.5 |
22 | 1197.7 | |||
| Lack of fit | 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 015.73 015.73 |
17 | 1295.04 | 1.49 | 0.348 | Not significant |
| Pure error | 4333.77 | 5 | 866.75 | |||
| Cor total | 31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 540.37 540.37 |
23 |
| Source | SS | df | MS | F-value | p-value | |
|---|---|---|---|---|---|---|
| Model | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 124.54 124.54 |
5 | 2224.91 | 6.06 | 0.0019 | Significant |
| F-power | 2993.18 | 1 | 2993.18 | 8.15 | 0.0105 | |
| G-t (min) | 230.16 | 1 | 230.16 | 0.6267 | 0.4389 | |
| H-Gr (µm) | 6732.67 | 1 | 6732.67 | 18.33 | 0.0004 | |
| FG | 1246.66 | 1 | 1246.66 | 3.39 | 0.0819 | |
| FH | 1395.37 | 1 | 1395.37 | 3.8 | 0.067 | |
| Residual | 6610.1 | 18 | 367.23 | |||
| Lack of fit | 5005.58 | 13 | 385.04 | 1.2 | 0.4513 | Not significant |
| Pure error | 1604.52 | 5 | 320.9 | |||
| Cor total | 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 734.64 734.64 |
23 |
| Source | SS | df | MS | F-value | p-value | |
|---|---|---|---|---|---|---|
| Model | 724.1 | 3 | 241.37 | 6.2 | 0.0038 | Significant |
| A-T (°C) | 2.38 | 1 | 2.38 | 0.0611 | 0.8073 | |
| C-ratio | 521.51 | 1 | 521.51 | 13.39 | 0.0016 | |
| A2 | 178.64 | 1 | 178.64 | 4.59 | 0.0447 | |
| Residual | 778.98 | 20 | 38.95 | |||
| Lack of fit | 528.24 | 15 | 35.22 | 0.7022 | 0.7281 | Not significant |
| Pure error | 250.74 | 5 | 50.15 | |||
| Cor total | 1503.08 | 23 |
| Source | SS | df | MS | F-value | p-value | |
|---|---|---|---|---|---|---|
| Model | 1489.05 | 5 | 297.81 | 18.83 | < 0.0001 | Significant |
| E-ratio | 148.08 | 1 | 148.08 | 9.36 | 0.0067 | |
| F-power | 1096.56 | 1 | 1096.56 | 69.34 | < 0.0001 | |
| G-t (min) | 51.73 | 1 | 51.73 | 3.27 | 0.0872 | |
| H-Gr (µm) | 622.05 | 1 | 622.05 | 39.34 | < 0.0001 | |
| FG | 66.32 | 1 | 66.32 | 4.19 | 0.0555 | |
| Residual | 284.64 | 18 | 15.81 | |||
| Lack of fit | 135.9 | 13 | 10.45 | 0.3514 | 0.9403 | Not significant |
| Pure error | 148.74 | 5 | 29.75 | |||
| Cor total | 1773.69 | 23 |
An equation in terms of coded factors was generated correlating reaction parameters (independent variables) with the concentration response (dependent variable). In all cases, the final equation in default-coded terms assigns the highest value as 1 and the lowest value as −1. The influence of each factor will be compared according to its coefficient in the equation.
Comparison of TPC extraction methods
Table 3 presents the TPC per gram of dry weight of CBS extracts obtained using UAE and MAE. The TPC values of the CBS obtained by UAE ranged from 100 to 191 mg GAE per g extract d.w., while those obtained by MAE ranged from 94 to 182 mg GAE per g extract d.w.The TPC model equations are expressed in coded factor form for UAE (2) and MAE (3). In the UAE equation, time (B) shows a negative linear effect and a positive quadratic term (B2), indicating an optimal time. Granulometry (D) has a negative coefficient because D is coded (−1 for fine and +1 for coarse), so finer particles increase TPC. In the MAE equation, the ratio (E), power (F), and time (G) exhibit positive linear effects, while granulometry (H) is negative (finer particles are beneficial). The interaction terms EF and FG are negative, suggesting antagonistic effects when these factor pairs are increased simultaneously.
| TPC (UAE) = 129.46 − 8.14B − 9.66D + 19.10B2 | (2) |
| TPC (MAE) = 135.32 + 10.63E + 0.9897F + 6.38G − 11.81H − 12.60EF − 10.33FG | (3) |
Both UAE and MAE showed that finer particles significantly increased TPC, but while UAE exhibited a curvilinear effect of time, indicating an optimum, MAE presented positive linear effects of the ratio, power, and time, along with two negative interactions (EF and FG). These differences reflect the distinct physical principles of each technique, conventional thermal diffusion in UAE versus microwave-induced heating in MAE, underscoring that optimization strategies must be tailored to the extraction method. All factor interpretations were based on statistical significance (p < 0.05) derived from the ANOVA.
Comparison of antioxidant capacity extraction methods
In this study, the FRAP, DPPH, and ABTS methods were employed to assess the antioxidant capacity of CBS extracts obtained via UAE and MAE. The FRAP values for the MAE and UAE extracts ranged from 124–194 and 127–253 mg TE per g extract d.w., respectively. Similarly, the DPPH scavenging effects of MAE and UAE extracts ranged from 35 to 72% and 38 to 76%, respectively. In addition, the ABTS values for MAE and UAE extracts ranged from 111 to 202 and 130 to 274 mg TE per g extract d.w., respectively (Table 3). Each antioxidant model equation (FRAP, DPPH, and ABTS) is expressed in the coded factor form for UAE (4, 6, 8) and MAE (5, 7, 9).For the FRAP equation obtained by UAE, it is evident that temperature (A) is the most significant factor negatively influencing concentration. Also, the effect of particle size (D) produced by the AD interaction is suppressed mainly by the D term. If the AD interaction is negative (i.e., −1 smaller particle size), the D term will be positive, and vice versa. In the case of MAE, the main factors, power (F) and ratio (E), have the most significant positive effects, and the power (F2) quadratic term has the most significant negative effect on FRAP extraction.
| FRAP (UAE) = 183.45 − 14.91A − 13.80D + 14.49AD | (4) |
| FRAP (MAE) = 165.83 + 4.93E + 7.72F − 2.70G − 10.12H − 11.83EG + 10.46FG − 11.63F2 | (5) |
Regarding the UAE-derivate DPPH equation, the quadratic temperature term (A) is the most negative in the extraction, independent of particle size. On the other hand, MAE extraction is primarily influenced by power (F) and, to a lesser extent, by granulometry (H).
| DPPH (UAE) = 59.83 − 0.3654A − 5.60C − 6.75A2 | (6) |
| DPPH (MAE) = 48.47 − 3.56E + 9.61F + 1.94G − 5.31H + 2.80FG | (7) |
Finally, the ABTS equation for UAE and MAE is represented by the a plane without any quadratic term (curvature). Granulometry has a positive effect on the extraction of smaller size (D and H, −1). Power (F) and time (G) in MAE have a positive effect. In contrast, granulometry (H) negatively affects extraction.
| ABTS (UAE) = 210.75 − 14.76D | (8) |
| ABTS (MAE) = 153.51 + 15.66F + 4.08G − 17.65H + 12.18FG − 10.69FH | (9) |
Across the three antioxidant assays, granulometry (particle size) consistently influenced extraction in both methods, with finer particles (D or H coded as −1) favouring higher activity in UAE for FRAP and ABTS, and in MAE for DPPH, FRAP, and ABTS. Other key factors differed between techniques, with UAE showing a dominant role in temperature (A) and its quadratic term in FRAP and DPPH, indicating an optimal temperature, whereas ABTS was affected solely by particle size. In contrast, MAE exhibited a more complex behaviour, with microwave power (F) significant across all three assays, often accompanied by quadratic (FRAP) or interaction terms (FRAP, ABTS, and DPPH), while the solvent-to-sample ratio (E) and time (G) contributed selectively depending on the assay. These differences reflect the distinct energy transfer mechanisms of each method and highlight that the optimal conditions for antioxidant recovery are assay-dependent. The interpretation of model terms was based on their p-values from the ANOVA.
The antioxidant activity values obtained for CBS extracts were higher than those reported for CBS flours (ABTS = 3.39–11.55 and FRAP = 3.84–7.62 mg TE per g sample).25 However, in both studies, particle size influences antioxidant activity, with antioxidant activity inversely proportional to particle size. The smaller the particle size, the greater the recovery of bioactive compounds and the higher the antioxidant activity. Particle size favors mass transfer and solvent accessibility to bioactive compounds, facilitating their permeation and diffusion through the plant matrix.26
Surface response analysis
Graphs represent a compound increase in a colour scale. In the case of TPC, as measured by UAE and MAE, the increase shifts from blue to red, ranging from approximately 120 to 180 mg GAE per g extract d.w. The use of a graphical contrast was proposed to highlight higher and lower extraction concentrations of each compound.The TPC graphs show that MAE-assisted extraction yields higher extraction at higher ratios, with a significant effect of particle size (Fig. 2C). Granulometry is a significant factor influencing extraction concentration in the UAE (Fig. 2A and B). Additionally, increased time reduces the extraction of TPC from samples with larger particles.
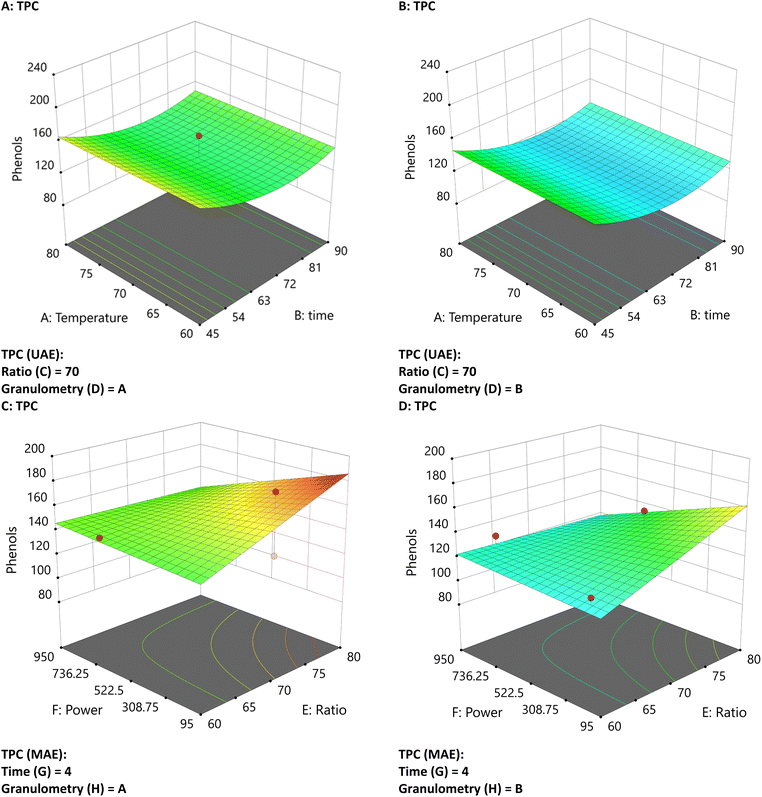 | ||
| Fig. 2 Three-dimensional graphs of UAE and MAE under different conditions for phenols with UAE (A and B) and MAE (C and D) with different particle sizes. | ||
Higher TPC concentrations by MAE can be attributed to microwave heating, which facilitates the direct transfer of electromagnetic energy to the matrix-solvent system, enabling rapid and uniform heating at the molecular level.27,28 This accelerates extraction kinetics, resulting in shorter extraction times than with UAE.
The FRAP graph shows an increase from blue to red, ranging from approximately 120 to 253 mg TE per g extract d.w. As observed in the FRAP graph (Fig. 3), a higher concentration is achieved with MAE at a 60 ratio, high power, and small particle size (Fig. 3C). On the other hand, using UAE, low temperatures, and small granulometry sizes increase FRAP concentration (Fig. 3A). In both cases, a significant reduction in concentration of the compounds is observed with higher particle size.
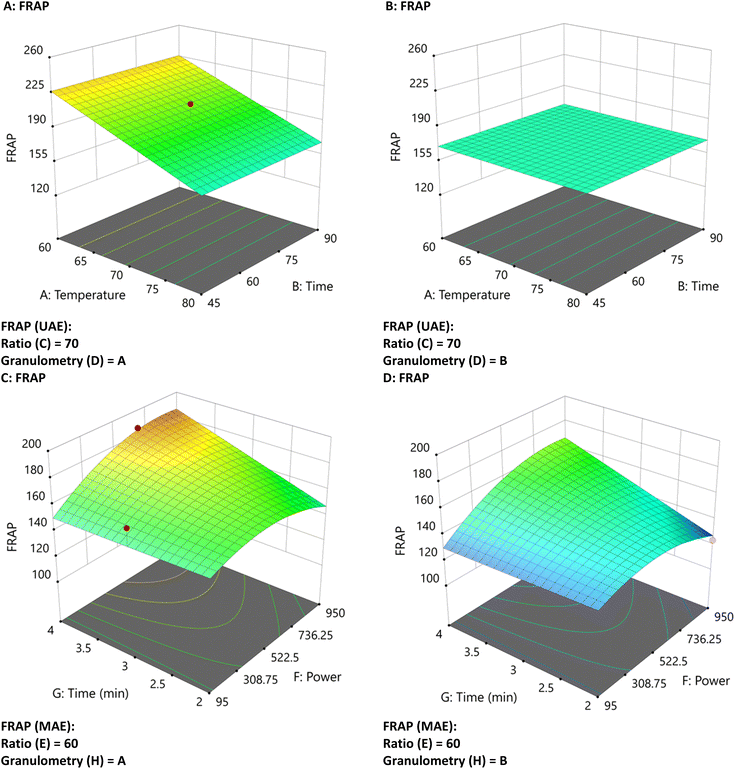 | ||
| Fig. 3 Three-dimensional graphs of UAE and MAE under different conditions for FRAP with UAE (A and B) and MAE (C and D) with different particle sizes. | ||
These FRAP trends closely parallel the TPC results discussed previously, where MAE also yielded higher phenolic extraction under similar conditions (high power and small particle size). This positive correlation suggests that phenolic compounds are major contributors to the antioxidant activity measured by FRAP. However, FRAP values were higher (up to 253 mg TE per g with UAE at A: 60, B: 45, C: 60, and D: A) than those with MAE, indicating that other factors may be influencing the reducing capacity.
Synergistic effects or interference from non-phenolic constituents with reducing power can enhance FRAP values, even when they lack biological relevance as antioxidants. Indeed, sugars and citric acid are recognized as common interferences,18 while organic acids (e.g., tartaric acid) and minerals have also been shown to significantly influence FRAP results.29 In the present study, gluconic acid (a reducing organic acid) was identified by UHPLC, and its presence likely contributed to the elevated FRAP values. Furthermore, the pronounced influence of particle size on both TPC and FRAP supports this, as smaller particles facilitate the release of a broader range of reducing species beyond phenolics.30
The DPPH concentration with MAE increases at high power levels and with smaller particles. On the other hand, in the UAE, DPPH values increase at lower ratios, as indicated by ANOVA analysis (Fig. 4C). In MAE, there is a linear increase with power reduction and time that generates a planar surface. On the other hand, the UAE reaches higher values in the middle of the experimental window (Fig. 4A and B). The UAE exhibits a slight increase with a lower ratio and shows no significant influence of particle size. Slightly higher DPPH values were observed in the MAE extracts than in the UAE (72% vs. 65%). Phenolic compounds, which donate hydrogen from their hydroxyl groups, are potent DPPH radical scavengers, and a positive correlation between DPPH activity and total phenolic content is generally expected.31
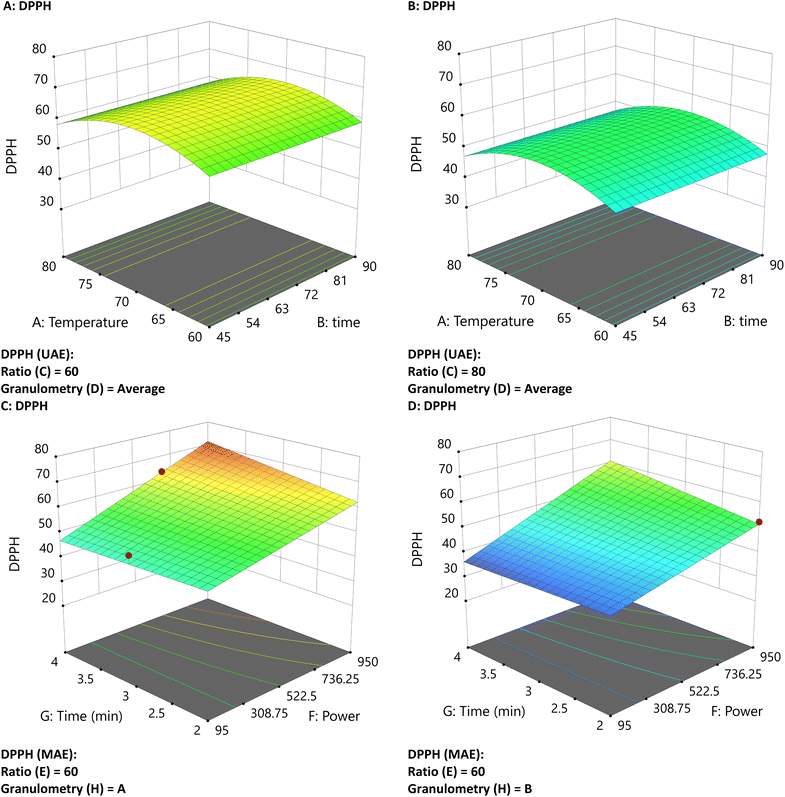 | ||
| Fig. 4 Three-dimensional graphs of UAE and MAE under different conditions for DPPH with UAE (A and B) and MAE (C and D). | ||
However, as with FRAP, DPPH results must be interpreted with caution. The DPPH radical is sterically hindered, making the reaction rate more dependent on the antioxidant's ability to access the radical center than on its intrinsic chemical properties.19 Consequently, large polyphenols may react slowly and be underestimated, whereas small molecules with good accessibility can react quickly regardless of their true antioxidant capacity.19 Moreover, the DPPH assay is susceptible to interference from reducing agents, which can cause decolorization independently of genuine radical-scavenging activity.18,20 In the present study, gluconic acid (identified by UHPLC) likely contributed to the measured radical scavenging activity, leading to an overestimation of the actual antioxidant capacity, a factor often overlooked in conventional DPPH-based analyses.
In ABTS, the UAE values were higher than those for MAE (274 > 202 mg ET per g CBS extract d.w.), as shown in Table 3 (Fig. 5C). Higher extraction was observed in MAE at smaller particle sizes, higher power, and a 4 minute time (Fig. 5C). ABTS extraction shows a linear increase in both cases, with no quadratic terms in their equations.
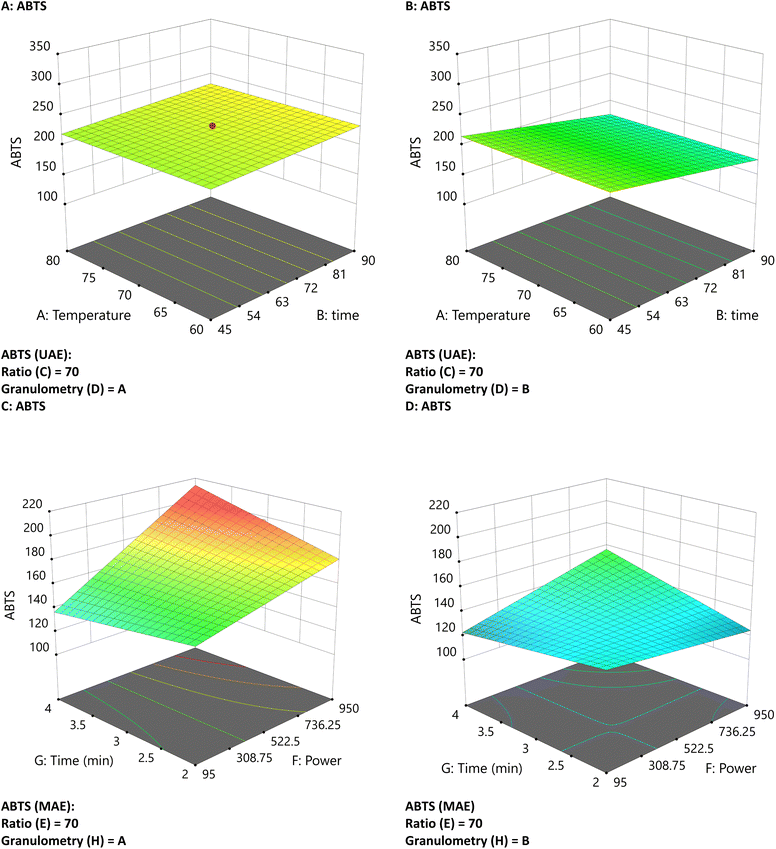 | ||
| Fig. 5 Three-dimensional graphs of UAE and MAE under different conditions for ABTS with UAE (A and B) and MAE (C and D) with different particle sizes. | ||
The discordance among FRAP, DPPH, and ABTS results highlights that electron-transfer-based antioxidant assays measure the combined redox activity of multiple chemical families rather than a single class of compounds.20 The Folin-Ciocalteu reagent is reduced by both phenolic and non-phenolic compounds, while ABTS and FRAP respond to the overall reducing capacity of a sample, often leading to poor correlation when total phenolic content is low or when non-phenolic reductants are abundant.20 In the present study, FRAP and DPPH values were notably influenced by the presence of gluconic acid (identified by UHPLC), a reducing organic acid that can overestimate antioxidant capacity independently of phenolic content. This explains why UAE extracts showed higher FRAP values despite lower TPC, whereas MAE yielded higher phenolic extraction. Since theobromine and caffeine (both alkaloids) were found to be prominent compounds in CBS extracts, their presence accounts for the higher ABTS values observed with UAE, despite the lower phenolic content. Taken together, these findings indicate that MAE favors phenolic extraction (as reflected in higher TPC), whereas UAE may enhance the recovery of alkaloids, organic acids, or other reducing species that interfere with FRAP, DPPH, and ABTS assays. Consequently, antioxidant capacity is not an absolute property but rather depends on both the extraction method and the assay employed, highlighting the need for chemical characterization and multiple assays when evaluating bioactive extracts.
Optimal conditions were established to achieve the highest component concentration in the extract (Tables 12 and 13). Desirability criteria were used to guide the optimization, aiming to maximize the concentration obtained for each component simultaneously. Desirability is an objective function that ranges from (d = 0) to the unit (d = 1).24
| T (°C) | t (min) | Ratio | Gr (µm) | TPC | DPPH | ABTS | FRAP | Desirability |
|---|---|---|---|---|---|---|---|---|
| 65 | 45 | 70 | A | 166 | 58 | 226 | 213 | 0.67 |
| Ratio | Power | t (min) | Gr (µm) | TPC | DPPH | ABTS | FRAP | Desirability |
|---|---|---|---|---|---|---|---|---|
| 60 | 950 | 4 | A | 146 | 72 | 214 | 187 | 0.85 |
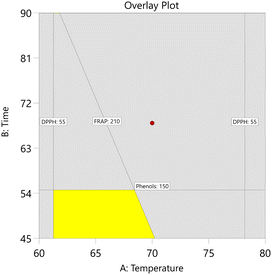
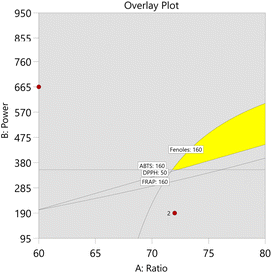
For CBS-UAE, a desirability value of 0.67 was obtained under optimal extraction conditions of 45 min, 65 °C, a ratio of 70 mL g−1, and a particle size of 75–150 µm (Table 12). On the other hand, the optimal conditions for CBS-MAE yielded a desirability of 0.85 with higher power (950 watts), a ratio of 60 mL g−1, and a particle size similar to that of UAE (Table 13).
Ultrasound-assisted extraction (UAE) demonstrated advantages over microwave-assisted extraction (MAE), yielding higher phenol concentrations and greater antioxidant capacity (as measured by ABTS and FRAP).
Graphical optimization plots are shown in Fig. 6 and 7. They indicate the regions where the desired criteria are met. The bright yellow (default) indicates where the entire range of all intervals meets the specified criteria (maximum), and the dark gold shows where the requirements are partially met.
The experimental verification values obtained under optimal extraction conditions, all within the expected range, are shown in Table 14. For the UAE, the experimental values were 160.01 mg GAE per g CBS extract (TPC), 31.99% antioxidant activity (DPPH), and 265.80 and 212.86 mg ET per g CBS extract for ABTS and FRAP, respectively. For MAE, the values were 166.74 mg GAE per g CBS extract (TPC), 42.12% (DPPH), and 253.59 and 183.83 mg ET per g CBS extract for ABTS and FRAP.
| Method | 95% CI | TPC | DPPH | ABTS | FRAP |
|---|---|---|---|---|---|
| a Note: DPPH was diluted 1/4 for the DOE and 1/20 for validation due to the high initial concentration, which explains the lower DPPH values observed during validation. | |||||
| UAE | Low for mean | 134 | 48 | 204 | 199 |
| High for mean | 165 | 58 | 247 | 254 | |
| SD | 19.4 | 6.24 | 34.6 | 27.8 | |
| Experimental | 160.01 ± 0.27 | 31.99 ± 0.77a | 265.8 ± 5.1 | 212.86 ± 4.95 | |
| MAE | Low for mean | 138 | 51 | 159 | 166 |
| High for mean | 157 | 56 | 183 | 186 | |
| SD | 15.7 | 3.97 | 19.1 | 11.7 | |
| Experimental | 166.74 ± 6.47 | 42.12 ± 1.53 | 253.59 ± 4.51 | 183.83 ± 15.03 | |
When comparing the two methods, UAE exhibited higher antioxidant capacity in the ABTS (266 > 254 mg ET per g) and FRAP (213 > 184 mg ET per g) assays, whereas MAE showed higher DPPH radical-scavenging activity (42% vs. 32%). The confidence intervals for TPC overlapped, indicating no statistically significant difference between the methods. Both extraction techniques outperformed traditional systems that rely on mechanical agitation.32
Table 15 compares the extraction of bioactive compounds from cocoa bean shells using different techniques. Among the reported UAE and MAE methods, our approach achieved competitive results while using ethanol as a simpler, conventional solvent. Under optimal MAE conditions (95 W, 2 min, 80 mL g−1), we obtained 42.12% DPPH and 166.74 mg g−1 TPC, with UAE yielding 31.99% DPPH and 160.01 mg g−1 TPC. Notably, our TPC values substantially outperformed water-based extractions (22.1–35.9 mg g−1),32 demonstrating ethanol's superior efficiency for phenolic compound recovery. In comparison, deep eutectic solvents have shown high DPPH scavenging (74.81%),33 though they require complex solvent preparation, whereas UAE with water has been reported to be efficient for isolating bioactive compounds from CBSs.34
| Method | Optimal conditions | Obtained compounds (d.b.) | Reference |
|---|---|---|---|
| a TPC (mg GAE per g extract bs) = total phenolic content; scavenging activity, DPPH (%) = 2,2-diphenyl-1-picryl-hydrazyl-hydrate, and ABTS (mg ET per g extract bs) = 2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonic acid); antioxidant capacity: FRAP (mg ET per g extract bs) = ferric reducing antioxidant power (*). | |||
| UAE | 45 min, 64 °C, ratio 70 mL g−1, and a particle size of 75–150 µm | TPC: 160.01 | This study |
| % DPPH: 31.99 | |||
| ABTS: 265.80 | |||
| FRAP: 212.86 | |||
| MAE | 95 W, 2 min, 80 mL g−1, and a particle size of 75–150 µm | TPC: 166.74 | This study |
| % DPPH: 42.12 | |||
| ABTS: 253.59 | |||
| FRAP: 183.83 | |||
| UAE | 69.45 °C; S/L 50 mL g−1; 37 kHz; UAE power (70%), and extraction time, 44.26 min | TPC: 118.38 | N. Pavlović et al., 2021 (ref. 34) |
| % DPPH: 81.846 | |||
| UAE | 40 kHz; 296 W; 50 mL g−1; 80% v/v ethanol; 55 °C; 45 min; extracto liofilizado | % DPPH: 30 | Md Yusof et al., 2019 (ref. 35) |
| FRAP: 319.20 µM Fe II per g extract | |||
| MAE | 5 min; pH 12, 97 °C; 25 mL g−1; water solvent; extracto seco a 40 °C | TPC: 35.9 | Mellinas et al., 2020 (ref. 32) |
| FRAP: 35.5 | |||
| ABTS: 33.6 | |||
| Conventional extraction | 22.2 mL g−1; water solvent; 100 °C; 90 min; extracto seco a 40 °C | TPC 22.1 | |
| FRAP 16.0 | |||
| ABTS 22.2 | |||
| MAE | Stirring; 63% v/v ethanol; 399.88 W; 217.42 s; 69.71 mL g−1; extracto diluido a 100 mL | TPC: 45.68 | Rincón Soledad, 2023.27 |
| FRAP*: 41.29 | |||
It can be assumed that there is a more efficient mass transfer mechanism in MAE than in UAE, which, unlike localized acoustic cavitation in UAE, activates a mechanism known as temperature-induced diffusion (TID), in which thermal gradients alter the internal chemical potential of the biomass, promoting greater water absorption in the cells and generating high internal pressures that weaken or break the cell walls.36,37 This effect facilitates the release of intracellular bioactive compounds, thereby increasing extraction yield and significantly reducing processing time compared to the UAE. Furthermore, microwave-induced heating produces localized temperature increases within the plant matrix that exceed the average temperature of the extraction system, thereby accelerating the solubilization of compounds in the solvent and improving overall extraction efficiency.37
In addition, MAE efficiency is linked to the dielectric properties of the system (solvent-plant matrix), especially the dielectric constant (ε′) and the dielectric loss factor (ε″), which determines the material's ability to absorb energy and convert it into heat.38 In the case of CBS, its heterogeneous composition, moisture, minerals, and fibers significantly influence its dielectric response. The higher the humidity and ions, the greater the dielectric loss (ε″), which translates into greater microwave energy absorption and an increase in the heating rate.38,39
The use of polar solvents, such as ethanol–water (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50), improves microwave energy absorption by adjusting the medium's dielectric constant, increasing internal temperature, and facilitating cell disruption.40 Factors such as humidity, volumetric density, and the presence of CBS ions can increase the dielectric loss factor, intensify the heating rate, and release bioactive compounds, including polyphenols and methylxanthines, present in this residue.38,41 While this temperature increase is beneficial for cell disruption and extraction yield, it also introduces a critical process control consideration: if dielectric and thermal parameters are not precisely regulated, localized overheating may occur, leading to partial degradation of thermolabile antioxidant compounds. For this reason, beyond this inherent risk, the primary technical restriction to the use of MAE in industry is process scalability, as limitations in heat and mass transfer at large volumes must be addressed.
50), improves microwave energy absorption by adjusting the medium's dielectric constant, increasing internal temperature, and facilitating cell disruption.40 Factors such as humidity, volumetric density, and the presence of CBS ions can increase the dielectric loss factor, intensify the heating rate, and release bioactive compounds, including polyphenols and methylxanthines, present in this residue.38,41 While this temperature increase is beneficial for cell disruption and extraction yield, it also introduces a critical process control consideration: if dielectric and thermal parameters are not precisely regulated, localized overheating may occur, leading to partial degradation of thermolabile antioxidant compounds. For this reason, beyond this inherent risk, the primary technical restriction to the use of MAE in industry is process scalability, as limitations in heat and mass transfer at large volumes must be addressed.
Chromatography identification of metabolites in optimized CBS extracts
Although the chemical profile obtained by UHPLC-Q-Orbitrap-MS/MS reveals no significant differences between both extraction methods, the analyzed extracts contain a complex combination of primary and secondary metabolites resulting from fermentation and drying. Metabolites were identified using deuterated standards; others were putatively identified based on matching retention times, exact masses, and MS2 spectra (Table 16). Retention times ranged from 1.50 to 6.00 min, with a predominance of highly polar molecules characteristic of lignocellulosic matrices, such as CBSs, and oxidized carbohydrates, amino acids, and alkaloids as a significant bioactive fraction. Table 16 summarizes the identified compounds, ordered based on retention time (RT).| Component name | Formula | Reference ion | Retention time (min) | Theoretical mass | Observed m/z | ppma | Fragments |
|---|---|---|---|---|---|---|---|
| a Parts per million, unit of mass precision of the mass spectrometer.b Available commercial standard (STD) for validation. | |||||||
| Arginine | C6H14N4O2 | [M + H]+ | 1.88 | 175.1190 | 175.1191 | 0.85 | 60.0562, 68.05019, 70.06577, 71.04976 |
| Adenine | C5H5N5 | [M + H]+ | 2.08 | 136.0617 | 136.0619 | 0.95 | 55.0297, 65.01408, 67.02972, 77.01400, 82.0404, 92.02483, 94.04040, 119.035949 |
| Mannitol | C6H14O6 | [M–H]− | 2.11 | 181.0718 | 181.0713 | 2.54 | 59.0128, 71.01301 |
| Gluconic acid | C6H12O7 | [M–H]− | 2.13 | 195.0510 | 195.0507 | 1.69 | 59.0128, 75.00780, 71.01285, 72.99220 |
| Aminoundecanoic acid | C11H23NO2 | [M + H]+ | 2.25 | 202.1802 | 202.1807 | 2.70 | 55.05489, 57.0705, 58.06578, 65.0391, 67.05479, 79.05473, 81.07034, 84.08130, 85.06528 |
| Theobromineb | C7H8N4O2 | [M + H]+ | 3.38 | 181.0720 | 181.0720 | 0.00 | 54.0345, 56.0501, 67.02970, 69.04534, 81.0452, 83.06092, 94.0404, 108.05600, 110.0715 |
| Phenylethanimine | C8H9N | [M + H]+ | 3.46 | 120.0807 | 120.0810 | 1.92 | 51.02362, 53.03927, 63.0235, 65.03922, 67.0548, 75.02357, 77.03918, 91.05474, 103.05458 |
| Phenylalanine | C9H11NO2 | [M + H]+ | 3.46 | 166.0863 | 166.0863 | 0.30 | 95.04965, 103.05461 |
| Caffeineb | C8H10N4O2 | [M + H]+ | 5.86 | 195.0877 | 195.0876 | 0.26 | 69.04535, 83.06095, 109.0400, 110.07171, 138.06639 |
On the one hand, two of the most intense signals found, in positive mode, for the CBS extracts assisted by MAE/UAE were theobromine (RT 3.38 min; m/z 181.0720) and caffeine (RT 5.86 min; m/z 195.0876), suggesting their high tolerance to energy-assisted extraction treatments, consistent with previous reports highlighting their thermal resistance and their high solubility in hydroalcoholic matrices.42 Their stability during MAE and UAE treatments suggests that both processes are suitable for obtaining alkaloid-enriched extracts, as reported by other authors who used MAE and UAE to extract alkaloids from cocoa husk.27,34 The presence of these methylxanthines reinforces the extract's potential for neurostimulant, antioxidant, and nutraceutical applications. It serves as a reliable chemical marker for cocoa crop authenticity, since the theobromine/caffeine ratio varies according to the cocoa genotype.43
On the other hand, polyalcohols and organic acids, such as mannitol (RT 2.11 min; m/z 181.0713) and gluconic acid (RT 2.13 min; m/z 195.0507), were identified in negative mode. These are typical products of carbohydrate transformation during cocoa fermentation, so their presence in the extract suggests disruption of the cell wall and release of partially or totally oxidized sugars during dielectric heating (MAE) and cavitation (UAE). On the one hand, mannitol is produced primarily by yeasts and lactic acid bacteria (LAB), especially Lactobacillus fermentum, which uses fructose as an alternative electron acceptor, allowing its reduction to mannitol during anaerobic fermentation.44–46 Mannitol is associated with bitterness, astringency, and fruity and floral descriptors in chocolate.46 Gluconic acid, on the other hand, is an undesirable product of cocoa fermentation, associated with a vinegary or unpleasant flavor in chocolate.47 Its production is attributed to Gluconobacter and Acetobacter pasteurianus, with Gluconobacter preferentially oxidizing glucose to gluconic acid when glucose has not been depleted by acetic acid bacteria (AAB), whereas Acetobacter acts at the end of fermentation, after ethanol depletion.47,48 Finally, nitrogen-rich and Maillard reaction metabolites were detected, such as arginine (RT 1.88 min, m/z 175.1191), phenylalanine (RT 3.46 min, m/z 166.0863), phenylethanimine (RT 3.46 min, m/z 120.0810), and aminoundecanoic acid (RT 2.25 min, m/z 202.1807). The presence of these compounds may indicate early Maillard reactions during roasting and the release of free amino acids during metabolite extraction.49 Notably, the roasting conditions employed (a gradual ramp from 90 to 120 °C over 50 min) are relatively mild, which is consistent with the detection of early-stage Maillard products rather than advanced browning compounds.
Arginine was also identified, mainly due to proteolysis during grain fermentation, where this amino acid contributes to the slightly bitter taste.50 Phenylalanine plays a structural role in proteins and is among the predominant amino acids following cocoa fermentation.51–53 It is also a key precursor in Maillard reactions, contributing to the aromas and color of cocoa, as well as to the formation of biogenic amines such as phenylethanimine, a product of its decarboxylation.54,55 The co-detection of phenylethanimine with phenylalanine in the extract suggests active degradation and conversion pathways during post-harvest processing of cocoa beans. Aminoundecanoic acid is associated with lipid derivatives formed during processing through the modification of fatty acids.56,57 Finally, adenine was identified (RT 2.08 min; m/z 136.0619), suggesting possible changes in another nitrogen-rich metabolic pathway.58
Environmental impact assessment
AGREEprep has been used to select processes with lower environmental impact or to identify problem points in terms of “greenness” during the design and development of new extractive processes. AGREEprep uses a scale of 0 to 1, with scores above 0.5 indicating an acceptable method or above 0.6, indicating an environmentally friendly process.59 The results of the AGREEprep assessments are shown in Fig. 8, and the weight criterion is shown in Table 17. The extraction procedures using UAE and MAE yielded overall scores of 0.58/1.00 and 0.65/1.00, respectively. Although both extraction processes exceed the threshold for ecological or eco-friendly treatments,59 MAE would be more sustainable. Likewise, the UAE score technique compared to MAE shows notable advantages in performance (criterion 6) and energy consumption (criterion 8). At the same time, both techniques shared weaknesses in the use of hazardous substances in the bioactive recovery process and in the number of steps required (criteria 2 and 7). Nevertheless, ethanol is considered one of the most environmentally friendly solvents, enabling more sustainable, scalable extraction of bioactives on an industrial scale.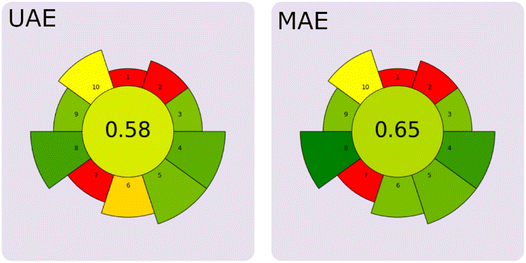 | ||
| Fig. 8 Comparison of the results of the AGREEprep evaluation of CBS bioactive extraction techniques. | ||
| Assessment criteria | Weight | Score | ||
|---|---|---|---|---|
| UAE | MAE | |||
| 1 | Sample preparation: ex situ | 1 | 0.00 | 0.00 |
| 2 | Volume [mL] of problematic substances | 2 | 0.00 | 0.00 |
| 3 | Use of sustainable or renewable materials | 2 | 0.75 | 0.75 |
| 4 | Mass [g] of waste generated | 4 | 0.82 | 0.90 |
| 5 | Mass [g] of the sample | 4 | 0.77 | 0.79 |
| 6 | Number of samples per hour | 3 | 0.42 | 0.76 |
| 7 | Integrate steps and automation | 2 | 0.00 | 0.00 |
| 8 | Energy consumption | 4 | 0.87 | 1.00 |
| 9 | Post-sample preparation configuration for analysis | 2 | 0.75 | 0.75 |
| 10 | Number of identified hazards | 3 | 0.50 | 0.50 |
| Total | 0.58 | 0.65 | ||
To distinguish between the two processes using AGREEprep criteria, greater weight (4) was assigned to criteria 4, 5, and 8, as these directly reflect critical differences in operational efficiency and resource utilization.
In criterion 4 (waste generation), MAE achieved a higher score (0.90) compared to UAE (0.82), reflecting its more efficient use of materials and lower waste output per batch. For criterion 5 (sample mass), both methods performed similarly (UAE: 0.77; MAE: 0.79), indicating that they handle sample sizes efficiently and show no meaningful difference. Criterion 8 (energy consumption) showed the most pronounced disparity, with the value for MAE at 1.00 compared to that of UAE at 0.76, confirming that the shorter extraction time of MAE translates into substantially lower energy demand.
For criterion 1 (ex situ sample preparation), both UAE and MAE were assigned a score of 0.00, as sample preparation is performed prior to extraction, as shown in Fig. 1 (grinding and sieving of the dried sample). Regarding criterion 3 (use of sustainable or renewable materials), both methods received a score of 0.75, reflecting a good commitment to renewable sources. In criterion 6 (number of samples per hour), MAE (0.76) substantially outperformed UAE (0.42). For UAE, each extraction batch required 45 minutes (including setup and cooling), allowing approximately 6 batches per hour, which yielded a score of 0.42. For MAE, the extraction time was 4 minutes per batch, enabling up to 25 batches per hour and resulting in a score of 0.76. These scores reflect the tool's normalization based on throughput efficiency. For criterion 9 (post-sample preparation configuration for analysis), both methods received a score of 0.75, indicating a moderate scope for direct coupling or reduced handling. Finally, criteria 2 (volume of problematic samples) and 10 (number of identified hazards) yielded a score of 0.50 for both UAE and MAE, primarily due to the use of ethanol, which, despite being one of the most environmentally friendly solvents for scalable extractions, remains flammable and requires careful safety measures.
In summary, the higher overall AGREEprep score for MAE (0.65 vs. 0.58) reflects its better performance in throughput (criterion 6), waste generation (criterion 4), and energy consumption (criterion 8). The results suggest that future efforts should focus primarily on reducing waste generation (criterion 4), improving extraction yield per unit mass (criterion 5), and optimizing energy consumption (criterion 8), as these factors were most decisive in distinguishing between the two techniques.
Conclusions
Both MAE and UAE demonstrated high extraction efficiency for bioactive compounds from cocoa husk at fine particle sizes (75–150 µm), with similar results for total phenolic content (TPC) and DPPH radical-scavenging activity. The selection of the most suitable method depends on the primary objective: if the goal is to maximize total phenolic content (TPC), MAE is preferred, as it yields higher phenolic extraction. Conversely, if the objective is to obtain extracts with high overall reducing capacity (as measured by FRAP and ABTS), UAE may be more suitable, though these assays are susceptible to interference from non-phenolic compounds such as gluconic acid and alkaloids, identified by HPLC. The UAE yielded significantly higher ABTS and FRAP values (indicating antioxidant activity), supporting its viability for industrial-scale applications. MAE is better suited when processing speed is the main driver, offering a comparable phytochemical profile while requiring careful management of its inherent risks. The temperature increase inherent to MAE is beneficial for cellular disruption and yield, but if not precisely regulated, localized overheating due to uneven dielectric absorption can degrade thermolabile antioxidant compounds. Furthermore, the primary technical limitation for MAE remains its scalability, given the complexities of achieving uniform microwave distribution in large-volume systems.UHPLC–HRMS analysis showed that UAE and MAE produce extracts with highly comparable chemical profiles, characterized by intense signals for key methylxanthines, including theobromine and caffeine, as well as polyalcohols, organic acids, amino acids, and nitrogen-rich metabolites derived from cocoa fermentation and post-harvest processing. These findings confirm that both green technologies effectively release diverse and stable bioactive compounds from cocoa bean shells. Moreover, the similarity of the identified metabolites, predominantly alkaloids such as theobromine and caffeine, highlights the potential of these extracts for applications in nutraceutical formulations, antioxidant systems, functional beverages, and the development of micro- and nano-encapsulated products, given their compatibility with polymeric matrices.
Finally, the environmental assessment using the AGREEprep method validated both technologies as clean for the recovery of high-value metabolites, thereby supporting agro-industrial waste utilization and the cocoa value chain. However, MAE achieved a higher overall score (0.65 vs. 0.58) due to better throughput, lower waste generation, and reduced energy consumption, the key differentiating factors (criteria 4, 5, and 8). Therefore, future efforts should prioritize minimizing waste, improving extraction yield per unit mass, and optimizing energy use.
Author contributions
Erick Alvarez-Yanamango: conceptualization, methodology, investigation, resources, writing – original draft, writing – review & editing, visualization, supervision, project administration, funding acquisition. Milagros Huaytalla-Ramirez, Nereyda Sarmiento-Perez, Gerald Chumpitaz-Huanqui, and Luis Napan-Tacca: investigation, formal analysis. Daniel Obregon-Valencia: methodology, formal analysis, validation, writing – review & editing, visualization. Alfredo Ibañez: investigation, supervision, formal analysis, writing – review & editing.Conflicts of interest
The authors declare that no conflicts of interest or known personal connections could have influenced the work presented in this article.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this study.Acknowledgements
The authors acknowledge the financial support of the National Program for Scientific Research and Advanced Studies (PROCIENCIA) of the National Council of Science, Technology and Technological Innovation (CONCYTEC) through the project with contract PE501086489-2024-PROCIENCIA, and the Vice-Rectorate of Research of the Pontifical Catholic University of Peru for its financial support in the CAP 2024 - PI1115 project. Erick Alvarez-Yanamango acknowledges the Doctoral Scholarship in Engineering from Pontificia Universidad Católica del Perú (PUCP).Notes and references
- J. C. Hashimoto, J. C. Lima, R. M. S. Celeghini, A. B. Nogueira, P. Efraim, R. J. Poppi and J. A. L. Pallone, Quality Control of Commercial Cocoa Beans (Theobroma cacao L.) by Near-infrared Spectroscopy, Food Anal. Methods, 2018, 11, 1510–1517 CrossRef.
- M. Sánchez, A. Laca, A. Laca and M. Díaz, Cocoa Bean Shell: A By-Product with High Potential for Nutritional and Biotechnological Applications, Antioxidants, 2023, 12(5), 1028, DOI:10.3390/antiox12051028.
- ICCO, Quarterly Bulletin of Cocoa Statistics - Production, 2024 Search PubMed.
- O. Rojo-Poveda, L. Barbosa-Pereira, G. Zeppa and C. Stévigny, Cocoa bean shell—a by-product with nutritional properties and biofunctional potential, Nutrients, 2020, 12(4), 1123, DOI:10.3390/nu12041123.
- Z. Ö. Cinar, M. Atanassova, T. B. Tumer, G. Caruso, G. Antika, S. Sharma, J. Sharifi-Rad and R. Pezzani, Cocoa and cocoa bean shells role in human health: An updated review, J. Food Compos. Anal., 2021, 103, 104115, DOI:10.1016/j.jfca.2021.104115.
- M. C. Castro, H. Villagarcía, A. Nazar, L. G. Arbeláez, M. L. Massa, H. Del Zotto, J. L. Ríos, G. R. Schinella and F. Francini, Cacao extract enriched in polyphenols prevents endocrine-metabolic disturbances in a rat model of prediabetes triggered by a sucrose rich diet, J. Ethnopharmacol., 2020, 247, 112263, DOI:10.1016/j.jep.2019.112263.
- J. Li, H. D. Sesso, E. Kim, J. E. Manson, G. Friedenberg, A. Clar, T. Copeland, A. H. Shadyab, J. Wactawski-Wende, L. Tinker and S. Liu, Cocoa Extract Supplementation and Risk of Type 2 Diabetes: The Cocoa Supplement and Multivitamin Outcomes Study (COSMOS) Randomized Clinical Trial, Diabetes Care, 2023, 46, 2278–2284 CrossRef PubMed.
- L. Goya, J. E. Kongor and S. de Pascual-Teresa, From Cocoa to Chocolate: Effect of Processing on Flavanols and Methylxanthines and Their Mechanisms of Action, Int. J. Mol. Sci., 2022, 23(22), 14365, DOI:10.3390/ijms232214365.
- D. Janitschke, A. A. Lauer, C. M. Bachmann, J. Winkler, L. V. Griebsch, S. M. Pilz, E. L. Theiss, H. S. Grimm, T. Hartmann and M. O. W. Grimm, Methylxanthines Induce a Change in the AD/Neurodegeneration-Linked Lipid Profile in Neuroblastoma Cells, Int. J. Mol. Sci., 2022, 23(4), 2295, DOI:10.3390/ijms23042295.
- M. Sánchez, A. Laca, A. Laca and M. Díaz, Cocoa Bean Shell: A By-Product with High Potential for Nutritional and Biotechnological Applications, Antioxidants, 2023, 12(5), 1028, DOI:10.3390/antiox12051028.
- E. Alvarez-Yanamango, D. Obregon and A. Ibañez, A Comprehensive Review of the Development of Green Extraction Methods and Encapsulation of Theobromine from Cocoa Bean Shells for Nutraceutical Applications, Food Eng. Rev., 2025, 17, 1083–1104 CrossRef CAS.
- V. L. Singleton and J. A. Rossi, Colorimetry Of Total Phenolics With Phosphomolybdic-Phosphotungstic Acid Reagents, Am. J. Enol. Vitic., 1965, 16, 144–158, DOI:10.5344/ajev.1965.16.3.144.
- W. Brand-Williams, M. E. Cuvelier and C. Berset, Use of a Free Radical Method to Evaluate Antioxidant Activity, LWT – Food Sci. Technol., 1995, 28(1), 25–30, DOI:10.1016/S0023-6438(95)80008-5.
- R. Re, N. Pellegrini, A. Proteggente, A. Pannala, M. Yang and C. Rice-Evans, Original Contribution Antioxidant Activity Applying An Improved ABTS Radical Cation Decolorization Assay, Free Radical Biol. Med., 1999, 26(9–10), 1231–1237, DOI:10.1016/S0891-5849(98)00315-3.
- I. F. F. Benzie and J. J. Strain, The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay, Anal. Biochem., 1996, 239(1), 70–76, DOI:10.1006/abio.1996.0292.
- N. B. Sadeer, D. Montesano, S. Albrizio, G. Zengin and M. F. Mahomoodally, The versatility of antioxidant assays in food science and safety—chemistry, applications, strengths, and limitations, Antioxidants, 2020, 9(8), 709, DOI:10.3390/antiox9080709.
- M. Minarti, N. Ariani, M. Megawati, A. Hidayat, M. Hendra, G. Primahana and A. Darmawan, in E3S Web of Conferences, EDP Sciences, 2024, vol. 503 Search PubMed.
- R. L. Prior, X. Wu and K. Schaich, Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements, J. Agric. Food Chem., 2005, 53(10), 4290–4302, DOI:10.1021/jf0502698.
- K. M. Schaich, X. Tian and J. Xie, Hurdles and pitfalls in measuring antioxidant efficacy: A critical evaluation of ABTS, DPPH, and ORAC assays, J. Funct. Foods, 2015, 14, 111–125, DOI:10.1016/j.jff.2015.01.043.
- F. Shahidi and A. Samarasinghe, How to assess antioxidant activity? Advances, limitations, and applications of in vitro, in vivo, and ex vivo approaches, Food Prod., Process. Nutr., 2025, 7(1), 1, DOI:10.1186/s43014-025-00326-z.
- Y. Rodríguez-Carrasco, A. Gaspari, G. Graziani, A. Santini and A. Ritieni, Fast analysis of polyphenols and alkaloids in cocoa-based products by ultra-high performance liquid chromatography and Orbitrap high resolution mass spectrometry (UHPLC-Q-Orbitrap-MS/MS), Food Res. Int., 2018, 111, 229–236 CrossRef PubMed.
- W. Wojnowski, M. Tobiszewski, F. Pena-Pereira and E. Psillakis, AGREEprep – Analytical greenness metric for sample preparation, TrAC, Trends Anal. Chem., 2022, 149, 116553, DOI:10.1016/j.trac.2022.116553.
- F. Pena-Pereira, M. Tobiszewski, W. Wojnowski and E. Psillakis, A Tutorial on AGREEprep an Analytical Greenness Metric for Sample Preparation, Adv. Sample Prep., 2022, 149, 116553, DOI:10.1016/j.sampre.2022.100025.
- M. J. Anderson and P. J. Whitcomb, Optimizing Processes Using Response Surface Methods for Design of Experiments Second Edition RSM Simplified, Boca Raton, FL, 2016 Search PubMed.
- C. Botella-Martínez, R. Lucas-Gonzalez, C. Ballester-Costa, J. Á. Pérez-álvarez, J. Fernández-López, J. Delgado-Ospina, C. Chaves-López and M. Viuda-Martos, Ghanaian cocoa (Theobroma cacao l.) bean shells coproducts: Effect of particle size on chemical composition, bioactive compound content and antioxidant activity, Agronomy, 2021, 11(2), 401, DOI:10.3390/agronomy11020401.
- A. Meullemiestre, E. Petitcolas, Z. Maache-Rezzoug, F. Chemat and S. A. Rezzoug, Impact of ultrasound on solid-liquid extraction of phenolic compounds from maritime pine sawdust waste. Kinetics, optimization and large scale experiments, Ultrason. Sonochem., 2016, 28, 230–239 CrossRef CAS PubMed.
- M. C. Rincón Soledad, Extracción asistida por microondas de compuestos fenólicos de los subproductos del beneficio del cacao (Theobroma cacao L.), 2023 Search PubMed.
- S. R. Dewi, L. Stevens, D. Irvine, R. Ferrari and E. Binner, Microwave-Assisted Extraction Of Phenolic From Cacao Pod Husks-An Alternative For Valorisation, Zenodo, 2021 Search PubMed.
- I. R. N. de Oliveira, R. F. Teófilo, E. B. de Oliveira, A. M. Ramos, F. A. R. de Barros, M. P. Maia and P. C. Stringheta, Evaluation of potential interfering agents on in vitro methods for the determination of the antioxidant capacity in anthocyanin extracts, Int. J. Food Sci. Technol., 2017, 52, 511–518 CrossRef CAS.
- V. Lavelli, P. S. C. Sri Harsha, L. Torri and G. Zeppa, Use of winemaking by-products as an ingredient for tomato puree: The effect of particle size on product quality, Food Chem., 2014, 152, 162–168 CrossRef CAS PubMed.
- A. Itam, M. S. Wati, V. Agustin, N. Sabri, R. A. Jumanah and M. Efdi, Comparative Study of Phytochemical, Antioxidant, and Cytotoxic Activities and Phenolic Content of Syzygium aqueum (Burm. f. Alston f.) Extracts Growing in West Sumatera Indonesia, Sci. World J., 2021 DOI:10.1155/2021/5537597.
- A. C. Mellinas, A. Jiménez and M. C. Garrigós, Optimization of microwave-assisted extraction of cocoa bean shell waste and evaluation of its antioxidant, physicochemical and functional properties, LWT, 2020, 127, 109361, DOI:10.1016/j.lwt.2020.109361.
- N. Pavlović, S. Jokić, M. Jakovljević, M. Blažić and M. Molnar, Green extraction methods for active compounds from food waste - Cocoa bean shell, Foods, 2020, 9(2), 140, DOI:10.3390/foods9020140.
- N. Pavlović, M. Jakovljević, M. Molnar and S. Jokić, Ultrasound-Assisted Extraction Of Active Compounds From Cocoa Bean Shell, Hrana u zdravlju i bolesti, 2021, 10(2), 77–88 Search PubMed.
- A. H. Md Yusof, S. S. Abd Gani, U. H. Zaidan, M. I. E. Halmi and B. H. Zainudin, Optimization of an Ultrasound-Assisted Extraction Condition for Flavonoid Compounds from Cocoa Shells (Theobroma cacao) Using Response Surface Methodology, Molecules, 2019, 24(4), 711, DOI:10.3390/molecules24040711.
- A. Taqi, E. Farcot, J. P. Robinson and E. R. Binner, Understanding microwave heating in biomass-solvent systems, Chem. Eng. J., 2020, 393, 124741, DOI:10.1016/j.cej.2020.124741.
- Y. Mao, J. Robinson and E. Binner, Understanding heat and mass transfer processes during microwave-assisted and conventional solvent extraction, Chem. Eng. Sci., 2021, 233, 116418, DOI:10.1016/j.ces.2020.116418.
- X. Zhu, W. Guo, X. Wu and S. Wang, Dielectric properties of chestnut flour relevant to drying with radio-frequency and microwave energy, J. Food Eng., 2012, 113, 143–150 CrossRef CAS.
- D. Wang, X. Li, X. Hao, J. Lv and X. Chen, The Effects of Moisture and Temperature on the Microwave Absorption Power of Poplar Wood, Forests, 2022, 13(2), 309, DOI:10.3390/f13020309.
- M. D. Pavlović, A. V. Buntić, S. S. Šiler-Marinković and S. I. Dimitrijević-Branković, Ethanol influenced fast microwave-assisted extraction for natural antioxidants obtaining from spent filter coffee, Sep. Purif. Technol., 2013, 118, 503–510 CrossRef.
- Q. Hu, Y. He, F. Wang, J. Wu, Z. Ci, L. Chen, R. Xu, M. Yang, J. Lin, L. Han and D. Zhang, Microwave technology: a novel approach to the transformation of natural metabolites, Chin. Med., 2021, 16(1), 87, DOI:10.1186/s13020-021-00500-8.
- J. Zhong, N. Tang, B. Asadzadeh and W. Yan, Measurement and Correlation of Solubility of Theobromine, Theophylline, and Caffeine in Water and Organic Solvents at Various Temperatures, J. Chem. Eng. Data, 2017, 62, 2570–2577 CrossRef CAS.
- A. C. Aprotosoaie, S. V. Luca and A. Miron, Flavor Chemistry of Cocoa and Cocoa Products-An Overview, Compr. Rev. Food Sci. Food Saf., 2016, 15, 73–91 CrossRef CAS.
- T. Lefeber, M. Janssens, F. Moens, W. Gobert and L. De Vuyst, Interesting starter culture strains for controlled cocoa bean fermentation revealed by simulated cocoa pulp fermentations of cocoa-specific lactic acid bacteria, Appl. Environ. Microbiol., 2011, 77, 6694–6698 CrossRef CAS.
- H. Nishimura, Y. Shiwa, S. Tomita and A. Endo, Microbial composition and metabolic profiles during machine-controlled intra-factory fermentation of cocoa beans harvested in semitropical area of Japan, Biosci. Microbiota, Food Health, 2024, 43, 29–42 CrossRef CAS.
- Y. Kitani, S. P. Putri and E. Fukusaki, Investigation of the effect of processing on the component changes of single-origin chocolate during the bean-to-bar process, J. Biosci. Bioeng., 2022, 134, 138–143 CrossRef CAS PubMed.
- C. Xin Hui, N. Lindley, Y. Kanagasundaram and Y. Chow, in Theobroma Cacao - Past, Present and Future Insights [Working Title], IntechOpen, 2025 Search PubMed.
- F. Moens, T. Lefeber and L. De Vuyst, Oxidation of metabolites highlights the microbial interactions and role of Acetobacter pasteurianus during cocoa bean fermentation, Appl. Environ. Microbiol., 2014, 80, 1848–1857 CrossRef PubMed.
- R. F. Peña-Correa, B. A. Mogol, C. Fryganas and V. Fogliano, Fluidized-Bed-Roasted Cocoa Has Different Chemical Characteristics than Conventionally Roasted Cocoa, J. Agric. Food Chem., 2023, 71, 10203–10211 CrossRef.
- C. R. Balcázar-Zumaeta, E. Fernández-Romero, A. S. Lopes, N. R. Ferreira, G. C. A. Chagas-Júnior, I. Yoplac, H. A. López-Trigoso, M. L. Tuesta-Occ, I. Maldonado-Ramirez, J. L. Maicelo-Quintana, I. S. Cayo-Colca and E. M. Castro-Alayo, Amino acid profile behavior during the fermentation of Criollo cocoa beans, Food Chem.: X, 2024, 22, 101486, DOI:10.1016/j.fochx.2024.101486.
- V. Barišić, M. Kopjar, A. Jozinović, I. Flanjak, Đ. Ačkar, B. Miličević, D. Šubarić, S. Jokić and J. Babić, The chemistry behind chocolate production, Molecules, 2019, 24(17), 3163, DOI:10.3390/molecules24173163.
- M. Granvogl, S. Bugan and P. Schieberle, Formation of amines and aldehydes from parent amino acids during thermal processing of cocoa and model systems: New insights into pathways of the strecker reaction, J. Agric. Food Chem., 2006, 54, 1730–1739 CrossRef CAS.
- C. Hue, Z. Gunata, A. Breysse, F. Davrieux, R. Boulanger and F. X. Sauvage, Impact of fermentation on nitrogenous compounds of cocoa beans (Theobroma cacao L.) from various origins, Food Chem., 2016, 192, 958–964 CrossRef CAS.
- K. Purbaningrum, C. Hidayat, L. D. Witasari and T. Utami, Flavor Precursors and Volatile Compounds Improvement of Unfermented Cocoa Beans by Hydrolysis Using Bromelain, Foods, 2023, 12(4), 820, DOI:10.3390/foods12040820.
- C. Q. Chen and E. Robbins, Chocolate flavor via the Maillard reaction, ACS Symp. Ser., 2000, 754, 286–292 CrossRef CAS.
- T. F. Soares and M. B. P. P. Oliveira, Cocoa By-Products: Characterization of Bioactive Compounds and Beneficial Health Effects, Molecules, 2022, 27(5), 1625, DOI:10.3390/molecules27051625.
- X.-Q. Zheng, Y. Koyama, C. Nagai and H. Ashihara, Biosynthesis, accumulation and degradation of theobromine in developing Theobroma cacao fruits, J. Plant Physiol., 2004, 161(4), 363–369, DOI:10.1078/0176-1617-01253.
- P. H. Pour, F. M. Suzaei and S. M. Daryanavard, Greenness assessment of microextraction techniques in therapeutic drug monitoring, Bioanalysis, 2024, 16, 249–278 CrossRef CAS PubMed.
- S. S. Ferreira, T. A. Brito, A. P. R. Santana, T. G. S. Guimarães, R. S. Lamarca, K. C. Ferreira, P. C. F. L. Gomes, A. Oliveira, C. D. B. Amaral and M. H. Gonzalez, Greenness of procedures using NADES in the preparation of vegetal samples: Comparison of five green metrics, Talanta Open, 2022, 6, 100131, DOI:10.1016/j.talo.2022.100131.
| This journal is © The Royal Society of Chemistry 2026 |