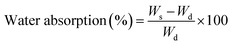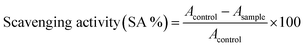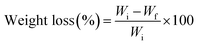Open Access Article
Open Access ArticleChitosan/PVA/gelatin derived biodegradable coatings enriched with Rosa rubiginosa for extended shelf life and preservation of strawberries
Beenish Sarwara,
Muhammad Zubair b,
Asma Yaqoobc,
Faiz Ahmed
b,
Asma Yaqoobc,
Faiz Ahmed d,
Sohail Shahzad*a and
Aman Ullah
d,
Sohail Shahzad*a and
Aman Ullah *b
*b
aDepartment of Chemistry, University of Sahiwal, Sahiwal 57000, Pakistan. E-mail: drsohail@uosahiwal.edu.pk
bLipid Chemistry Utilization Lab, Department of Agricultural, Food & Nutritional Science, University of Alberta, Edmonton, AB T6G 2P5, Canada. E-mail: ullah2@ualberta.ca
cInstitute of Biochemistry, Biotechnology and Bioinformatics, The Islamia University of Bahawalpur, Pakistan
dDepartment of Chemistry, Government College University Faisalabad, Faisalabad 37000, Pakistan
First published on 3rd February 2026
Abstract
This study developed biodegradable composite coatings and films using chitosan (CS), polyvinyl alcohol (PVA), and gelatin enriched with Rosa rubiginosa extract (RRE) to enhance the shelf life of strawberries. The coatings were prepared through solution blending and applied via dip-coating, while the corresponding films were fabricated using solution casting. FTIR and SEM analyses confirmed the effective molecular interactions and uniform microstructure within the composite matrix. Increasing the proportion of RRE improved functional performance by reducing the moisture content, water solubility, water absorption, and oxygen permeability. The formulation with the highest RRE content (BS-03) exhibited the strongest antioxidant activity (78.8 ± 1.01% DPPH inhibition; IC50 = 0.3 mg mL−1) and enhanced antibacterial potential against Staphylococcus aureus, Bacillus subtilis, and Escherichia coli. Soil burial tests revealed that the films were biodegradable, with BS-02 and BS-03 showing mass losses of up to 91.29% and 86.79% after 45 days, respectively. When applied to strawberries, the coatings significantly reduced weight loss and delayed visible spoilage, extending the shelf life by up to 10 days compared to uncoated fruit. Sensory evaluation also confirmed better color, firmness, and acceptability of the coated samples. These findings demonstrate that CS/PVA/gelatin coatings enriched with RRE offer a promising, safe, and eco-friendly approach for prolonging the freshness of perishable fruits.
Sustainability spotlightBiodegradable coatings developed from chitosan, polyvinyl alcohol, gelatin, and Rosa rubiginosa extract present an environmentally friendly technique for fruit preservation. These natural and synthetic biopolymers are combined to produce films with improved antioxidant, antibacterial and barrier properties, providing a sustainable alternative to traditional plastic packaging. By applying these coatings to strawberries, the study shows a significant increase in the shelf life while minimizing food waste and environmental impact. The addition of Rosa rubiginosa, a plant rich in bioactive compounds, enhances the value of underutilized botanical resources. This work aligns with the goals of circular bioeconomy and supports the global transition to sustainable, biodegradable materials for food packaging applications. |
1. Introduction
Approximately 1.3 billion tons of food is wasted annually, making food waste one of the major global challenges of our time, which results in resource depletion and environmental damage.1 Packaging protects food from harmful ultraviolet rays and preserves its quality, aroma, taste, and physicochemical characteristics. By preserving food, we can enhance its use and protect it from microbial decay.2 With the increasing demand for eco-friendly packaging solutions, innovative biopolymer-based coatings play a vital role in preserving food and prolonging the shelf life of perishable items, thereby positively affecting the environment.3 Films containing plasticizers such as glycerol, honey, and propylene glycol are applied to food products to improve their nutritional, antimicrobial, and antioxidant properties.4 The addition of natural extracts, such as essential oils or plant extracts, to chitosan-based films improves their functional properties.5 A variety of semi-synthetic and edible biopolymers are used in food packaging applications, including chitosan, agarose, alginate, agar, starch, cellulose, guar gum, polyvinyl alcohol and carboxymethyl cellulose.6,7 Although plastic materials are lightweight, economical, and flexible, they contribute to waste, raising environmental concerns.3Recently, there has been growing interest in utilizing biodegradable materials for food packaging and coatings.8 Bio-based plastics are favoured for film coatings because they are abundantly available, biodegradable and capable of forming films.9 However, these materials often lack mechanical strength and offer poor resistance.10 To address these limitations, structural modifications11 or blending of bio-based polymers with synthetic polymers have been performed to obtain specific properties.12 Chitosan, a naturally occurring biopolymer with inherent anti-microbial13 and anti-oxidant properties,14–16 has been widely used in food packaging.17 It is non-toxic18 and has chelating ability to bind with vital trace metals selectively and bacteria, and to prevent toxin and bacterial growth,19,20 thus making it sutiable for food packaging applications.21,22 In addition, chitosan has high susceptibility to water and low thermal and mechanical strength, which limits it use in food packaging materials. To address these limitations, different techniques such as cross-linking,23–26 enzymatic treatment,27 graft copolymerization,28–30 complex formation,29 surface coating,31 incorporation of fillers,32 high-energy radiation33 and blending with other biopolymers or synthetic polymers can be used.34,35
PVA is a synthetic and water-soluble polymer that is an ideal biomaterial for making polymer blends. It is biocompatible, flexible, and non-toxic36 and can be used in food packaging applications because of its low cost, eco-friendly nature, durability, transparency, mechanical strength, chemical stability, and film-forming properties.37,38 Blending PVA with chitosan not only enhanced the mechanical strength but also improved the antibacterial activity of the fabricated films.
In contrast, Rosa rugosa, a common petal variety, has been traditionally used in biomedical applications.39 Rosa species contain components including flavonoids, terpenes, glycosides, polyphenols, and anthocyanins. The volatile oil extracted from Rosa flowers possesses therapeutic properties and is primarily used for analgesic, anticonvulsant, cardiovascular, and laxative purposes.40 Given the numerous bioactive components found in Rosa rubiginosa (RR), our objective was to explore the effects of its incorporation and proportion when combined with CS/PVA/gelatine solutions. These coatings and films are expected to exhibit enhanced mechanical strength and improved antibacterial, antioxidant, and biodegradable properties, which are essential for prolonging the shelf life of coated or packaged food products, such as strawberries. Choi et al. developed a ternary blend (CS/PVA/gelatin)-based composite film incorporating Duchesnea indica extract for the preservation of strawberries.41 In another study, Islam et al. fabricated packaging films using a casting methodology with CS/PVA incorporated with zinc oxide nanoparticles and dragon fruit waste extract. The incorporation of ZnO nanoparticles and dragon fruit waste extract enhanced the thermal and mechanical strength and provided antioxidant and antibacterial potential to the films.42 Similarly, Wang et al. synthesized bilayered films with excellent biodegradable, antibacterial, mechanical, and oxygen barrier properties using CS as the outer layer and PVA/gelatin as the inner layer loaded with 2.5% curcumin nanoparticles for the preservation of fish (bigeye tuna).43 However, CS/PVA/gelatin ternary films have not yet been combined with RRE, and their application as edible coatings for strawberries has not been evaluated in terms of antioxidant/antibacterial performance, barrier properties, biodegradability, and sensory acceptance. This study aims to develop and characterize CS/PVA/gelatin coatings enriched with varying concentrations of RRE and evaluate their effectiveness in extending the shelf life of strawberries.
2. Experimental
2.1 Materials
The chemicals and solvents utilized in this study were employed directly as obtained from manufacturers or redistilled when necessary. Macklin Chemicals (China) supplied chitosan (CS) with a deacetylation degree exceeding 95% (molecular weight (MW) 100–200 kDa) and viscosity in the range of 100–200 mPa s. Polyvinyl alcohol (PVA) (MW 72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 with 98% degree of hydrolysis) was procured from Merck (Germany). Gelatin and glacial acetic acid (C2H4O2) were bought from Sigma Aldrich–Germany. A taxonomist identified the plant sample as Rosa rubiginosa (RR), which was obtained from the University of Sahiwal, Pakistan. Strawberries were purchased from a local market in Sahiwal–Pakistan.
000 with 98% degree of hydrolysis) was procured from Merck (Germany). Gelatin and glacial acetic acid (C2H4O2) were bought from Sigma Aldrich–Germany. A taxonomist identified the plant sample as Rosa rubiginosa (RR), which was obtained from the University of Sahiwal, Pakistan. Strawberries were purchased from a local market in Sahiwal–Pakistan.
2.2 Methodology
| Samples codes | CS solution (mL) | PVA solution (mL) | Glycerol solution (mL) | Gelatin solution (mL) | RRE (mL) |
|---|---|---|---|---|---|
| BS-0 | 20 | 20 | 10 | 2 | 0 |
| BS-01 | 20 | 20 | 10 | 2 | 2 |
| BS-02 | 20 | 20 | 10 | 2 | 4 |
| BS-03 | 20 | 20 | 10 | 2 | 6 |
2.3 Characterization of the synthesized films
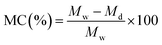 | (1) |
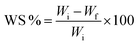 | (2) |
where Ws stands for the swollen weight and Wd stands for the dried weight.
where Acontrol shows the absorbance of the control, that is DPPH solution, and Asample stands for the absorbance of the samples.
where Wi stands for the initial weight of strawberries and Wf stands for the final weight of strawberries.
3. Results and discussion
3.1 FTIR analysis
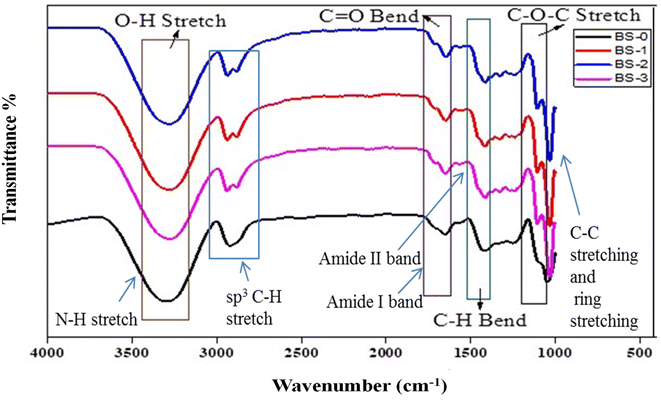
FTIR analyses were performed to determine the various functional groups present in the films synthesized from CS/PVA/gelatin and loaded with RRE and to check their intermolecular interactions. Fig. 2 shows the FTIR spectra of CS/PVA/gelatin films without RRE (BS-0) and with RRE (BS-01, BS-02 and BS-03). FTIR spectra of all films in Fig. 2 show a very broad peak at 3300 cm−1 to 3250 cm−1 due to the overlapping stretching frequencies of hydroxyl O–H and amino N–H groups.46 Alkyl C–H group asymmetric stretching was observed between 2950 cm−1 and 2916 cm−1. The peaks for the amide I and amide II bands appeared at 1669 cm−1 and 1564 cm−1, respectively.56 CH2 bending peaks were seen at 1408 cm−1 and 1338 cm−1. C–O and C–O–C stretching vibrations were observed at 1143 cm−1 and 1099 cm−1. These vibration bands were also reported by Dawei Yun et al.57 Chitosan's ring stretching vibrations appeared in the fingerprint region at 895 cm−1.58The FTIR spectra of the films with RRE exhibited an additional prominent peak at 2970 cm−1 attributed to aromatic C–H stretch,59 which was due to aromatic polyphenolic and flavonoid compounds present in RRE. Similarly, FTIR spectra of BS-01, BS-02 and BS-03 exhibited numerous small peaks from 1800 cm−1 to 1500 cm−1, due to additional components of RRE.60 Minor variations towards lower frequency for certain peaks were observed in the FTIR spectra, possibly due to bonding interactions between CS, PVA, gelatin and RRE. Additional peaks at 1700 cm−1, present in BS-01, BS-02 and BS-03 spectra and absent in the BS-0 spectrum, were attributed to various components like secondary metabolites and polyphenols present in RRE.61 The BS-0 film displayed O–H and N–H stretching at 3330 cm−1, and the band shifted towards lower wavenumber at 3267 cm−1 for films incorporated with RRE (BS-01, BS-02 and BS-03), due to the greater interactions, created by active functional groups O–H and N–H present within CS, PVA, gelatin and RRE. As the OH group present in polyphenols develops hydrogen bonding with the OH group present in CS and PVA, and NH2 group in chitosan, greater hydrogen bonding results in lower frequency ranges. These FTIR results are also supported by literature studies.62 The hydroxyl group (–OH) and amino group (–NH2) stretching vibrations in the BS-0 film spectrum appeared to be greater than those found in BS-01, BS-02, and BS-03 spectra. This was due to the presence of a higher number of unreacted –OH and –NH2 functional groups in the BS-0 film. The reduction in stretching vibrations in the spectra of BS-01, BS-02, and BS-03 films containing RRE suggested the involvement of active –OH and –NH2 groups in PVA and chitosan with the functional groups present in RRE, leading to the formation of hydrogen bonds and other weak electrostatic interactions. Overall, FTIR analysis confirmed the presence of the characteristic peaks for all components under consideration.
3.2 Film thickness
The thickness of the films is a vital factor in attaining desirable optical properties and optimum barrier against water and vapor transfer. An increase in film thickness may reduce the diffusion rates of oxygen and water vapors of the film.63 The addition of plasticizers, antioxidants, and antimicrobial agents can increase the thickness of the films, offering better barrier properties and improved mechanical strength.64 The thickness results shown in Fig. 3 indicate that the BS-0 film (without RRE) showed a minimum thickness of 0.123 ± 0.02 mm, while the maximum thickness was displayed by BS-03 (0.884 ± 0.06 mm), followed by BS-02 (0.771 ± 0.05 mm), and BS-01 (0.439 ± 0.03 mm). Gradually increasing the RRE from 0 mL (BS-0) to 6 mL (BS-03) also enhanced the film thickness, which is consistent with the literature.65 Films with suitable or greater thicknesses offer better barrier properties, and thus lower water and oxygen permeability, making them ideal for prolonged packaging of perishable fruits.3.3 Visual impact by camera images
The visual appearance and color of films are essential parameters that affect the standard and quality of food items. Colored films have more potential to wrap food items and preserve them because they reduce oxidation reactions in packaged foods.66 Fig. 4 shows the camera images of the CS/PVA/gelatin films and CS/PVA/gelatin films loaded with various volumes of RRE. The CS/PVA/gelatin films were completely transparent, whereas the other films incorporating RRE were yellowish-brown. It was noted that, by increasing the RRE volume from 2 mL in BS-01 to 6 mL in BS-03, the color of the films became intense yellowish brown as illustrated in Fig. 4. Additionally, the films were seen smooth due to better mixing, blending and developing certain types of new interactions between film components. The dried films were observed to be smooth, homogeneous, stretchable, bubble free, mechanically strong, and flexible and they were peeled off from the Petri dishes swiftly without cracks.3.4 Morphological analysis using SEM
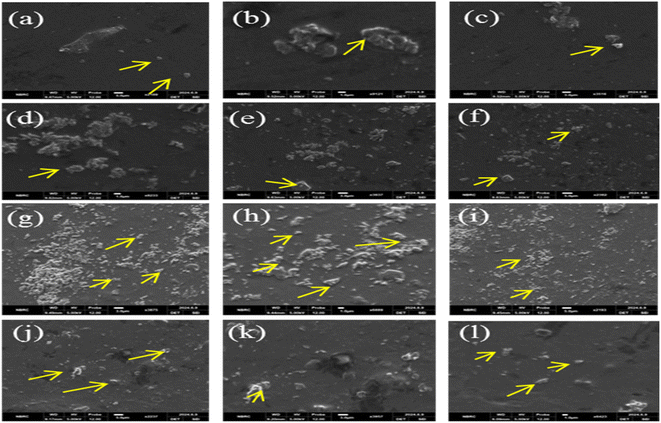
SEM analysis was used to examine the surface morphology of the prepared CS/PVA/gelatin film and CS/PVA/gelatin loaded with RRE films. The SEM images of the composite films are shown Fig. 5. The SEM images in Fig. 5(a–c) of the BS-0 film (composed of only CS/PVA/gelatin) appear clearer and more homogenous without cracks and crystalline particles/aggregates on their surface, indicating the amorphous nature of this film. The micrographs in Fig. 5(d–f) of the BS-01 film show that all components were uniformly distributed and appeared in one phase and were homogeneous. The images show the presence of certain aggregates/crystals within the structure, which were due to the addition of 2 mL of RRE into the CS/PVA/gelatin matrix. The addition of RRE into the CS/PVA/gelatin matrix has induced certain intermolecular interactions, thus producing a few aggregates of some components. The micrographs in Fig. 5(g–i) represent the BS-02 film incorporating CS/PVA/gelatin loaded with 4 mL of RRE. Here also, the images reveal the uniformity, consistency and homogeneity of all the added components. Additionally, the BS-02 film also showed many white crystals, which might have been formed due to the interactions between active functional groups in CS, PVA, gelatin and RRE. Thus, it is evident from the images that increasing the RRE ratio from 2 mL (BS-01) to 4 mL (BS-02) had altered the morphology of the films to some extent. The film with a higher volume ratio of RRE (4 mL in BS-02) exhibited more crystalline molecules/aggregates within its structure, as already reported in the literature.67 One potential explanation is the interaction between the active components of RRE and the active groups in CS, PVA, and gelatin, which leads to the partial release of bioactive compounds such as polyphenols, alkaloids, and amino acids onto the film's surface.68 A similar type of interaction was also reported by Han et al. in Noni (Morinda citrifolia) fruit polysaccharide enriched with blueberry leaf extract.69 It is believed that bridging phenomena occurred inside the bio-composite films in which some molecules of RRE bonded to the CS chain while the rest of the functional groups attached to another CS chain. Thus, the overall surface morphology shows that RRE was effectively incorporated into the CS/PVA/gelatin solution matrix. In contrast to the above results, Fig. 5(j–l) represents the BS-03 film (incorporating 6 mL of RRE), which shows the film structure as clearer, smoother and more homogeneous, without a higher number of crystalline particles/aggregates on its surface as compared to the BS-02 film. Thus, it can be concluded that increasing the RRE to 6 mL in the CS/PVA/gelatin matrix would again make the film texture smooth.70The cross-sectional analysis of the BS-0 film is shown in Fig. 6(a), which shows that the film is homogeneous, uniform, thick, and has no cracks in its structure. Similarly, the cross-sectional SEM image of the BS-01 film is more homogeneous, uniform and without any pores as compared to the BS-02 film, which has certain cracks in its structure.
Further cross-sectional images in Fig. 6(c and d) reveal that BS-02 and BS-03 films incorporating RRE addition to CS/PVA and gelatin were smoother, and their thickness increased, respectively. The increase in thickness was primarily due to the increasing volume ratios of RRE. The BS-02 film cross-sectional image in Fig. 6(c) shows some cracks and irregular cavities, which indicates its changed morphology as compared to other films. This indicated the presence of certain types of intermolecular interactions between functional groups of RRE active constituents and CS, PVA and gelatin.
The BS-03 film also appeared smooth and without cracks/pores. The homogeneous distribution of bioactive components within the matrix of the films ensures that their antibacterial and antioxidant activities remain unaffected.
3.5 Mechanical strength
The mechanical properties of films are crucial for their use in packaging. To assess the mechanical strengths of these films, stress–strain curves were plotted and are depicted in Fig. 7. Two sample films, including BS-0 (incorporating CS/PVA/gelatine) and BS-03 (incorporating CS/PVA/gelatine and RRE) were selected for the determination of mechanical properties and to evaluate the effect of incorporation of RRE into the CS/PVA/gelatin matrix. The BS-0 film stress–strain curve is shown in green colour, while the BS-03 stress–strain curve is shown in red colour. It was noted that the addition of RRE had slightly reduced the mechanical properties of the films. The stress value for the BS-0 film was noted as 0.32 MPa, while the stress value for the BS-03 film was 0.21 MPa. Similarly, the strain % for the BS-01 film was noted as 56%, while for the BS-03 film it was nearly 25%. This phenomenon can be explained as follows: RRE contains many polyphenolic and flavonoid components, which have active functional groups. These active functional groups had created certain interactions with polymeric chains of CS, PVA and gelatin. Thus, polymeric interactions among CS, PVA and gelatin chains were reduced, which ultimately altered the internal structure of the films. It is well known that chitosan has low mechanical strength; thus, it is blended with synthetic polymers like PVA to enhance its mechanical strength. The introduction of aqueous RRE into the CS/PVA/gelatin film (BS-03) has reduced its deformation, made the film stiffer and more rigid, and less flexible. Young's modulus for the BS-0 film was calculated to be 0.57 MPa, while Young's modulus for the BS-03 film was calculated to be 0.86 MPa. Thus the BS-03 film was observed to be less deforming, stiffer and more rigid as compared to the BS-01 film. Thus the BS-03 film is suitable for applications which require more strength and dimensional stability such as packaging applications. However the BS-01 film was deformed more under higher stress, which exhibits its higher flexibility and better suitability for those applications where flexibility and stretchability are required.3.6 XRD analysis
XRD analysis was performed to determine the crystalline or amorphous nature of the composite films. Literature studies of PVA and CS XRD spectra showed that PVA had a prominent peak at 2θ = 39.9°, while CS exhibited a sharp peak at 2θ = 20°. The BS-0 coded film demonstrated two notable peaks, one at 2θ = 19.59° corresponding to the crystalline phase of chitosan, and another at 2θ = 39.9° representing the amorphous phase as a lower intensity shoulder. The CS/PVA film BS-01 containing RRE also showed two amorphous peaks, slightly shifted to 2θ = 20° and 2θ = 41.9°. The BS-02 film exhibited peaks at 19.9° and 39.6°, while the BS-03 film displayed intensified peaks at 20.2° and 42°. Additional peaks were observed in BS-01, BS-02, and BS-03 films due to the inclusion of RRE, as illustrated in Fig. 8. The XRD values obtained for CS/PVA/gelatine films and CS/PVA/gelatine loaded with RRE films were found to be aligned with previously reported values.71 These XRD results confirmed the various interactions between CS, PVA, gelatin and RRE within the film matrix.3.7 Moisture content percent of the films
The moisture content of polymer-based films is a crucial parameter to determine the quality of films, as the purpose of films is to preserve the food from microbial spoilage. Here, CS/PVA/gelatin and CS/PVA/gelatin enriched with RRE composite films were analyzed for their ability to absorb moisture, and the results are displayed in Fig. 9(A), and the values are mentioned in Table 2. The outcomes of this assay revealed that all the films absorbed moisture content from 15% to 17%. Thus, a small variation was observed in moisture content results, which could be attributed to their different compositions. The moisture absorption phenomenon could be explained based on interactions such as hydrogen bonds between active amino and hydroxyl groups of CS, hydroxyl groups of PVA and water. In addition to this, the presence of glycerol had also contributed to moisture absorption, as glycerol is a well-known humectant. Furthermore, it was noted that the incorporation of RRE into the CS/PVA/gelatin matrix had slightly lessened the percent moisture content of the films. As the ratio of RRE was increased in the CS/PVA/gelatin matrix, the moisture content percent was decreased to a small extent. The reported results are in accordance with literature studies, as it has been mentioned in the literature that CS and PVA produced more interactions with active components of RRE and ultimately, their interaction with water was decreased, which led to hydrophobicity and a slight reduction of moisture content.72 The decreased percent moisture content is most favourable for food packaging applications as it offers better water barrier properties by lowering the microbial growth and improving the texture of packaged food.71| S. no. | Sample codes | Film thickness (mm) | Moisture content (%) | Water solubility (%) | Water absorption (%) |
|---|---|---|---|---|---|
| 1 | BS-0 | 0.123 ± 0.04 | 17.42 ± 1.97 | 73 ± 3.6 | 225.3 ± 5.25 |
| 2 | BS-01 | 0.439 ± 0.081 | 16.51 ± 2.52 | 70.2 ± 2.52 | 97.7 ± 3.44 |
| 3 | BS-02 | 0.771 ± 0.087 | 16.41 ± 2.35 | 63.2 ± 3.07 | 71.4 ± 1.97 |
| 4 | BS-03 | 0.884 ± 0.06 | 15.14 ± 1.46 | 60.2 ± 4.35 | 58.23 ± 2.31 |
3.8 Percent water solubility
The percent solubility of composite films of CS/PVA/gelatin and CS/PVA/gelatin loaded with RRE in water is the measure of weight loss of films when fully immersed in water for 24 hours. Solubility percent determination is important to measure as it depicts the stability of films as and when they come in contact with moisture in the environment for a longer time. The solubility percent test results are presented in Fig. 9(B), and their exact values are also given in Table 2. It was noticed that the CS/PVA/gelatin film BS-0 shows a maximum solubility % of 73%, which is mainly due to the presence of hydrophilic polymers of PVA and gelatin. However, CS is hydrophobic, and it makes interactions with both PVA and gelatin within the films to decrease their solubility. The addition of RRE into CS/PVA/gelatin films had decreased their solubility, as the BS-01 film solubility was 70.2%. Furthermore, increasing the volume ratio of RRE in the CS/PVA/gelatin films further decreased the solubility of the films, as observed in BS-02 (63.2% solubility) and BS-03 films (60.2% solubility). The intense interaction between CS and other polymers such as PVA, gelatin, and RRE led to increased intermolecular bonding, which in turn reduced their solubility.73 The films with lower solubility rates are more beneficial for food packaging applications, as these films are more likely to compromise with the moisture present in the surroundings, and their dissolution rate slows down. However, a balance between solubility and biodegradability is important, as excessively low solubility may slow down the degradation process post-use. A similar phenomenon of reducing the solubility of films by incorporating tea extract was also reported by Hafsa et al.743.9 Water absorption
Water absorption percent of films is a vital indicator to check their quality to absorb and retain water within their structure without being dissolved or solubilized. The degree of water absorption was found to be inversely proportional to the degree of interactions or crosslinking among different constituents of films. More interactions or crosslinking between polymeric chains show lesser water absorption and vice versa. The films with lower water absorption percent are frequently opted for food packaging applications. The results of the water absorption percent test are shown in Fig. 9(C) and listed in Table 2. The results exhibited that the BS-0 film absorbed maximum water up to 225.3% for 30 minutes. The BS-01, BS-02 and BS-03 films showed 97.7%, 71.4% and 58.23% of water absorption, respectively. The higher absorption by the BS-0 film can be attributed to the presence of hydrophilic polymers such as PVA and gelatin, which, in the absence of RRE, had made more interactions with water and thus absorbed more. The other films showed less absorption due to the presence of RRE and more interactions/crosslinking of polymeric chains with RRE instead of water molecules. This is evident because as the RRE volume ratio is enhanced, % water absorption is decreased, due to apparent development of a higher degree of crosslinking which resulted in lower % water absorption.753.10 Water drop test
A water drop test was performed on the composite films to check their ability to allow water to pass through or not. The films that hold water appeared to be acceptable for their application in food packaging. The test results for all films are displayed in Fig. 9(D), and the results show that the films did not allow the water drop to pass through them. The films or other materials which show superior water barrier properties are often opted for in food packaging applications.763.11 Oxygen permeability
Oxygen permeability of films has a noteworthy effect on the quality and shelf life of packaged food items. When oxygen enters the films, it causes spoilage of food items by oxidation, microbial growth and colour changes. Thus, lower rates of oxygen permeability can inhibit the oxidative spoilage and growth of aerobic microbes, leading to extended shelf life of perishable food products like strawberries. The oxygen present in the atmosphere can also weaken the bond energy of polymers, and thus unstable peroxides are formed. These unstable species slow down the degradation rate of polymeric films and thus make them unsuitable for food packaging applications. The oxygen permeability of the composite films was evaluated by the method already discussed by Wrinkler, and the results are presented in Fig. 9(E).77The O2-permeability test results for an airtight flask (negative control) and an open flask (positive control) were 4.7 ± 0.43 mg L−1 and 5.21 ± 0.22 mg L−1, respectively. The oxygen permeability values calculated for each film were 5.08 ± 0.23 mg per L (BS-0), 5.05 ± 0.31 mg per L (BS-01), 4.69 ± 0.57 mg per L (BS-02) and 3.03 ± 0.43 mg per L (BS-03). The data demonstrated that incorporating RRE reduced oxygen permeability, due to increasing RRE volume, which caused free volumetric holes in the films to contract, making it more difficult for oxygen molecules to penetrate. Thus, lower oxygen permeability is a desirable characteristic for polymers, particularly in food packaging applications.78
3.12 Antibacterial potential
Natural plant extracts incorporating antibacterial components are more sterile, eco-friendly, and biodegradable. They are regarded as excellent materials to be used in food packaging against food pathogens.79 Plant extracts contain bioactive phenolic and flavonoid components, and the hydroxyl groups in these compounds provide the basis for their antibacterial activity. The hydroxyl groups develop certain interactions, such as hydrogen bonds, with the bacterial cell membrane. Based on their hydrophobic nature, these phenolic compounds may accumulate on the surface of cells or may invade the cytoplasm of microbes. After reaching the cytoplasm, these components interact with the other cellular organelles and cell's components; thus, they change the pH inside the cell membrane.80 The bacterial cell wall has negative carboxylate groups (–COO−) and chitosan carries positive amino groups (–NH3+). The electrostatic interactions of these groups with each other provide the mechanism for antibacterial activity.81 CS/PVA/gelatine and CS/PVA/gelatine incorporating RRE solutions were assessed for antibacterial potential against S. aureus, B. subtilis and E. coli, and the results are shown in Fig. 10(A and B) and Table 3. The effectiveness of each coating solution was determined by measuring the inhibition zone in mm. All the coating solutions exhibited fair to good antibacterial potential against all three bacterial strains in comparison to the control. The results demonstrated that in most tests, the BS-03 sample showed better results than other samples. This was due to the presence of a higher volume ratio of RRE in the BS-03 sample. When we compared the results of all samples, BS-03 showed higher % inhibition against S. aureus with the lowest IC50 value of 78.65 ± 0.13. Similarly, the BS-02 sample exhibited higher activity against E. coli with the lowest IC50 value of 118.6 ± 0.021; while the BS-03 sample exhibited the highest potential against B. subtilis with the lowest IC50 value of 128.75 ± 0.65. Overall, BS-02 and BS-03 samples incorporating higher volume ratios of RRE showed better antibacterial potential against all three bacterial strains as compared to BS-0 and BS-01, attributed to the presence of various active components (phenolic compounds, flavonoids, and others) in the RRE. Notably, the increasing volume of RRE correlated with improved antibacterial activity.| Sample code | Inhibition zone (mm) | IC50 at 100 mg mL−1 | Percent inhibition |
|---|---|---|---|
| S. aureus | |||
| BS-0 | 9.6 | 172.91 ± 0.0542 | 17.12 ± 0.0542 |
| BS-01 | 9.6 | 122.91 ± 0.047 | 31 ± 0.047 |
| BS-02 | 11 | 93.6 ± 0.049 | 38.57 ± 0.049 |
| BS-03 | 8.9 | 78.65 ± 0.13 | 38.57 ± 0.13 |
| Control | 24 | 50 ± 0.9 | 100.714 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||
| E. coli | |||
| BS-0 | 6 | 120 ± 0.014 | 40 ± 0.014 |
| BS-01 | 6 | 115 ± 0.09 | 40 ± 0.09 |
| BS-02 | 12 | 118.6 ± 0.021 | 33.6 ± 0.021 |
| BS-03 | 11 | 120 ± 0.028 | 23 ± 0.028 |
| Control | 22 | 50 ± 0.8 | 100 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||
| B. subtilis | |||
| BS-0 | 8.7 | 138 ± 0.604 | 40 ± 0.234 |
| BS-01 | 3.6 | 225 ± 0.234 | 39.47368421 |
| BS-02 | 11 | 189.64 ± 0.28 | 26.10526316 ± 0.28571 |
| BS-03 | 14 | 128.75 ± 0.65 | 37.89473684 ± 0.65 |
| Control | 21 | 50.85 ± 0.714 | 100.714 |
Based on these findings, the synthesized CS/PVA/gelatine/RRE films, especially BS-02 and BS-03, show promise for food packaging applications. Similar antibacterial results were also reported by Dan et al.82 for a chitosan–gelatin film incorporating hop plant extract. The natural compounds found in plants are considered ideal candidates for food packaging applications due to their antibacterial properties. Two-way ANOVA demonstrated that the treatment type significantly influenced antibacterial activity (p < 0.001), and its effect varied among bacterial species. The positive control produced the strongest inhibition zones, BS-02 and BS-03 had moderate effects, while BS-0 and BS-01 showed minimal inhibition. These results suggest that specific treatments possess promising antibacterial potential depending on the bacterial species tested.
3.13 Antioxidant potential
The enhancement in food shelf life and its preservation applications depend upon the antioxidant potential of the synthesized coating solutions and films. Oxidation reactions cause discoloration of food and reduce its flavor. Antioxidant materials are required to deactivate and scavenge free radicals. The antioxidant potential of solutions was determined by the 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay (Table 4). DPPH is a nitrogen-free stable radical of deep purple color, which turns yellow after reacting with antioxidants. Antioxidant capacity was determined using the IC50 values, which represent the sample mass per mL of DPPH required to reduce the DPPH concentration by 50%. The results in Fig. 11 indicate that the BS-03 film demonstrated superior antioxidant properties (78.8 ± 1.01 with IC50 value of 0.3 mg mL−1) attributed to the presence of plant extract (RRE) in excess. Similarly, the BS-02 sample showed an antioxidant potential of 70.5 ± 0.57 with an IC50 value of 0.5 mg mL−1. The BS-01 film exhibited an antioxidant potential of 58.23 ± 1.3 with an IC50 value of 0.7 mg mL−1. The lowest antioxidant activity was exhibited by the BS-0 sample having an antioxidant potential value of 47.0 ± 0.51 with an IC50 value of 1.1 mg mL−1. The enhanced antioxidant activities of BS-03, BS-02 and BS-01 as compared to BS-0 were clearly attributed to the numerous active components found in RRE, including flavonoids, phenolics, citronellol, nerol, quercetin, catechin, epicatechin, rutin and phenethyl alcohol. These compounds prevent oxidative degradation, thus preventing the food from microbial spoilage and resulting in prolonging freshness. The results were also in accordance with the literature analysis.83 A statistical analysis reveals that both concentration and treatment type have highly significant effects on antioxidant activity. Furthermore, there is a significant interaction between these factors, indicating that the effect of each treatment depends on the concentration used. Thus, higher concentration significantly increases antioxidant activity (p < 0.0001).| Sample code | DPPH assay | IC50 mg mL−1 | |
|---|---|---|---|
| Percent inhibition of the DPPH free radical | |||
| 0.5 mg mL−1 | 1 mg mL−1 | ||
| BS-0 | 23.20 ± 1.3 | 47.0 ± 0.51 | 1.1 |
| BS-01 | 33.1 ± 0.4 | 58.23 ± 1.3 | 0.7 |
| BS-02 | 48.8 ± 1.3 | 70.5 ± 0.57 | 0.5 |
| BS-03 | 55.0 ± 2.0 | 78.8 ± 1.01 | 0.3 |
| Positive control (ascorbic acid) | 70.08 + 1.3 | 87.14 + 0.26 | 0.36 |
3.14 Biodegradation of the composite films
A soil burial degradation test was conducted to evaluate the decomposition rate of the prepared films. The weight loss percentage with respect to the number of days for BS-0, BS-01, BS-02, and BS-03 films was measured. This method effectively indicated film biodegradability due to the presence of microbes and moisture in soil. The biodegradation process is initiated with water absorption, which disrupts hydrogen bonding between polymeric chains, followed by microbial invasion from bacteria, algae, and fungi. Biodegradation is a natural process and is also influenced by the chemical composition of composite films. Various factors affect polymeric film degradation rates, including pollutant quantity, microbial presence, and environmental conditions (oxygen, humidity, pH, and temperature). Fig. 12(A) displays the degraded film images after 18 and 45 days, while Fig. 12(B) shows the % biodegradation after 18 and 45 days. The results showed that the BS-02 film demonstrated the highest % degradation of 91.29%, followed by the BS-03 film at 86.79% and the BS-01 film at 85.91%, while the BS-0 film showed a % degradation of 81.48%. It was observed that incorporation of RRE into CS/PVA/gelatin has improved the % biodegradation of the films. The biodegradation rates were higher as compared to already reported results by Riaz et al.84 The increased % biodegradation, due to the incorporation of RRE, was attributed to the presence of carbon, nitrogen, and oxygen in many components of RRE, which served as food sources for microorganisms. Continuous water addition activates soil bacteria and weakens polymeric interactions between components, thus increasing degradation rates.85 The incorporation of RRE into CS/PVA/gelatin films improved their biodegradability, transforming them into a rapidly degradable and eco-friendly material.84 The faster degradation of these biopolymer films in soil environments provides a sustainable alternative with reduced environmental impact.3.15 Total phenolic contents (TPCs) and total flavonoid contents (TFCs)
Polyphenols constitute the most frequent and broadly distributed group of compounds in the plant kingdom, with more than 8000 currently known compounds.86 The rose flower has high polyphenolic compounds and essential oil, and thus serves as a key source of many raw materials for cosmetics, herbal teas and rose oil.87 The TPCs and TFCs of Rosa rubiginosa extract (RRE) are expressed in µg of gallic acid equivalent (GAE) and µg of quercetin equivalent (QE) per mg of extract, respectively. The TPCs of RRE were found to be 86.74 ± 0.91 µg GAE per mg. These values are close to those described by Murathan et al.88 and Fascella et al.89 Literature studies have demonstrated that among phenolic compounds, p-coumaric acid and chlorogenic acid were present abundantly in RRE extract. It was observed that the phytochemical properties of various rose species are greatly dependent upon their type of species. The RRE was found to be a promising source of natural antioxidants, which are useful in the pharmaceutical and food industries.90 Similarly, TFCs of RRE were found to be 26.73 ± 0.68 µg QE per mg. These values were also in accordance with already reported values by Tabaszewska et al.91 Quercetin and luteolin are key flavonoid aglycons in rose species. Quercetin and luteolin belong to the flavonol and flavone groups, respectively.923.16 Application on strawberries
Coatings offer an effective solution to extend the shelf life of fruits and vegetables. Strawberries, being delicate and water-rich, have a limited shelf life and are prone to spoilage after harvesting due to pathogen attacks. Unpackaged or uncoated foods also deteriorate due to water evaporation and cellular respiration. All the composite coating solutions of BS-0, BS-01, BS-02 and BS-03 were used for application on strawberries to evaluate their potential to protect and enhance the shelf life of strawberries. Uncoated strawberries were used as a negative control for comparison of results. The weight loss % was monitored after the 1st, 2nd, 6th, 7th, 8th and 17th day. Fig. 13 (A) and (B) represent the results in terms of images of strawberries and weight loss % over time (in days), respectively. The results indicated that coating solutions with higher RRE ratios exhibited better protection of strawberries and provided greater shelf life, as indicated by both images and weight loss % graphs. Similar results have also been discussed in the literature by Mukhtar et al.93 The results showed that uncoated strawberries (control) started to deteriorate slowly after the 2nd day and experienced a 60% weight loss after the 7th day. For coated strawberries using BS-0, BS-01, BS-02, and BS-03 coating solutions, the weight loss percentages on day 6 were 39.82%, 34.87%, 32.12%, and 29.28%, respectively, compared to 45.55% for uncoated strawberries. Similarly, after the 7th day, the weight loss % was 45.35% for BS-0, 39.09% for BS-01, 35.05% for BS-02, 32.23% for BS-03 and 60.59% for the negative control. It can be inferred from Fig. 13(A) that the initial weight loss up to 6 days was mainly due to the loss of water content from strawberries, and then after 6 or 7 days, fungal attack began, which started to degrade the strawberries.By day 17, weight loss % reached 71.09% for BS-0, 66.32% for BS-01, 60.07% for BS-02, 57.2% for BS-03 and 89.03% for the uncoated negative control. It was noted that all coating solutions preserved strawberries well and enhanced their shelf life up to 8 days without being deteriorated by fungal attack. After 8 days, the strawberries coated with BS-0, BS-01 and BS-02 started to deteriorate by fungal attack more rapidly as compared to those coated with BS-03 solution, which remained intact up to 10 days. Thus, it can be concluded that increasing the volume ratio of RRE in CS/PVA/gelatin-based coating solutions has enhanced the shelf life of strawberries up to 10 days. The BS-03 coating solution demonstrated the best performance in reducing % weight loss, suggesting its highest potential for preserving delicate food items. The results of fabricated coatings on strawberries were also compared with literature studies, and the results are presented in Table S1 in the SI. The literature studies reported that the strawberries were stored for 5 days at room temperature and for 15 days at 4 °C, but our fabricated coatings protected strawberries for 10 days at room temperature.
3.17 Sensory evaluation
The results of sensory evaluation for each formulation coded as BS-0, BS-01, BS-02 and BS-03 and a control (without any coating) are presented in Tables S2–S6. The results showed that strawberries coated with the BS-03 formulation showed the most favourable and better results in terms of taste, smell, color, juiciness, firmness and acceptability in general as compared to BS-0, BS-01 and BS-02 formulations.This might be due to the presence of the highest amount of RRE in this formulation, which provided enhanced antioxidant and antibacterial properties to this formulation. The BS-0 formulation exhibited results of parameters such as taste, smell, color, juiciness, firmness and general acceptability mostly in “neither good nor bad” and “like somewhat” categories. Similarly, BS-01 showed most of the studied parameters in the “like somewhat” category. The BS-02 coating exhibited parameters in “like somewhat” and “moderate” categories. The BS-03 coating showed the most attractive and convincing results of studied parameters in the “similar to moderate” and “greatly admire” categories. The control strawberries without any coating were subjected to the highest degradation and changes, thus their study parameters mostly fall under “mildly disliked” and “mildly unfavourable” categories.
4. Conclusion
This study demonstrated that CS/PVA/gelatin composite coatings enriched with RRE enhanced the preservation of strawberries. Increasing the RRE volume ratio in CS/PVA/gelatin coatings (from BS-01 to BS-03) thickened the films and improved their barrier performance, while % moisture content, water solubility water absorption, and oxygen permeability were decreased. Antimicrobial tests showed that coatings containing a higher volume of RRE, particularly BS-02 and BS-03, exhibited stronger activities against S. aureus, B. subtilis, and E. coli than the RRE free coating (BS-0). Antioxidant capacity also increased with enhancing RRE ratio, with BS-03 reaching 78.8 ± 1.01% DPPH inhibition (IC50 = 0.3 mg mL−1). Soil burial trials confirmed that all films were biodegradable; the highest 45-day weight loss was observed for BS-02 (91.29%), followed by BS-03 (86.79%). Although the incorporation of RRE reduced ductility and increased stiffness, the mechanical properties of the BS-03 film would be more suitable for handling and packaging applications. When applied to strawberries, all coatings helped reduce weight loss compared to the uncoated control and prolonged their shelf life. Among them, BS-03 was the most effective, preventing visible spoilage for up to 10 days and achieving the highest sensory scores. Overall, the BS-03 formulation emerges as a promising bio-based coating for the short-term extension of strawberries shelf life. Future research should focus on optimizing composition through response surface methodology, conducting antifungal tests, evaluating performance during refrigerated storage, and examining component migration for applications involving direct contact with food.Author contributions
B. S.: conducted experiments, investigations, data analysis and writing – original draft. M. Z.: conceptualization, writing – original draft, reviewing and editing. A. Y.: performed antibacterial assessment. F. A.: reviewing. S. S.: conceptualization, supervision, editing and reviewing. A. U.: conceptualization, supervision, editing and reviewing. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the Supplementary Information (SI). Supplementary information: the tables have been cited in the manuscript. Table S1 cited in Section 3.16 and Tables S2–S6 cited in Section 3.17. While the Fig. S3 is cited in the Section 2.2.1. See DOI: https://doi.org/10.1039/d5fb00934k.References
- L. Kalra, R. Anushree and A. W. Sultani, Indian J. Ecol., 2024, 51, 697–706 Search PubMed.
- FAO, The State of Food and Agriculture 2019: Moving Forward on Food Loss and Waste Reduction, UN, 2019 Search PubMed.
- L. Brennan, S. Langley, K. Verghese, S. Lockrey, M. Ryder, C. Francis, N. T. Phan-Le and A. Hill, J. Clean. Prod., 2021, 281, 125276 CrossRef.
- S. Wardejn, S. Wacławek and G. Dudek, Int. J. Mol. Sci., 2024, 25, 12580 CrossRef CAS PubMed.
- S. Sumnu and L. Bayindirli, J. Sci. Food Agric., 1995, 67, 537–540 CrossRef.
- L. Pavoni, D. R. Perinelli, G. Bonacucina, M. Cespi and G. F. Palmieri, Nanomaterials, 2020, 10, 135 CrossRef CAS PubMed.
- F. Salehi, Int. J. Fruit Sci., 2020, 20, S570–S589 CrossRef.
- A. F. Pires, O. Díaz, A. Cobos and C. D. Pereira, Foods, 2024, 13, 2638 CrossRef CAS PubMed.
- M. Mujtaba, R. E. Morsi, G. Kerch, M. Z. Elsabee, M. Kaya, J. Labidi and K. M. Khawar, Int. J. Biol. Macromol., 2019, 121, 889–904 CrossRef CAS PubMed.
- F. Mohanty and S. Swain, in Nanotechnology Applications in Food, Elsevier, 2017, pp. 363–379 Search PubMed.
- E. Ojogbo, E. O. Ogunsona and T. H. Mekonnen, Mater. Today Sustain., 2020, 7, 100028 Search PubMed.
- Z. Wu, J. Wu, T. Peng, Y. Li, D. Lin, B. Xing, C. Li, Y. Yang, L. Yang and L. Zhang, Polymers, 2017, 9, 102 CrossRef PubMed.
- N. Ruocco, S. Costantini, S. Guariniello and M. Costantini, Molecules, 2016, 21, 551 CrossRef PubMed.
- M. Friedman and V. K. Juneja, J. Food Prot., 2010, 73, 1737–1761 CrossRef CAS PubMed.
- M. Kong, X. G. Chen, K. Xing and H. J. Park, Int. J. Food Microbiol., 2010, 144, 51–63 CrossRef CAS PubMed.
- L. A. van den Broek, R. J. Knoop, F. H. Kappen and C. G. Boeriu, Carbohydr. Polym., 2015, 116, 237–242 CrossRef CAS PubMed.
- M. A. Del Nobile, N. Di Benedetto, N. Suriano, A. Conte, M. R. Corbo and M. Sinigaglia, Food Microbiol., 2009, 26, 587–591 CrossRef CAS PubMed.
- A. Verlee, S. Mincke and C. V. Stevens, Carbohydr. Polym., 2017, 164, 268–283 CrossRef CAS PubMed.
- M. Hosseinnejad and S. M. Jafari, Int. J. Biol. Macromol., 2016, 85, 467–475 CrossRef CAS PubMed.
- D. C. Vodnar, O. L. Pop, F. V. Dulf and C. Socaciu, Not. Bot. Horti Agrobot. Cluj-Napoca, 2015, 43, 302–312 CrossRef CAS.
- N. A. Al-Tayyar, A. M. Youssef and R. Al-Hindi, Food Chem., 2020, 310, 125915 CrossRef CAS PubMed.
- M. Z. Elsabee and E. S. Abdou, Mater. Sci. Eng., C, 2013, 33, 1819–1841 CrossRef CAS PubMed.
- F. Jahan, R. Mathad and S. Farheen, Mater. Today: Proc., 2016, 3, 3689–3696 Search PubMed.
- J. Khouri, A. Penlidis and C. Moresoli, Processes, 2019, 7, 157 CrossRef CAS.
- H. Haghighi, F. Licciardello, P. Fava, H. W. Siesler and A. Pulvirenti, Food Packaging and Shelf Life, 2020, 26, 100551 CrossRef.
- T. Yeamsuksawat and J. Liang, Food Packag. Shelf Life, 2019, 22, 100415 CrossRef.
- E. Aguila-Almanza, R. Salgado-Delgado, Z. Vargas-Galarza, E. García-Hernández and H. Hernández-Cocoletzi, J. Chem., 2019, 2019, 1–8 CrossRef.
- W. M. Argüelles-Monal, J. Lizardi-Mendoza, D. Fernández-Quiroz, M. T. Recillas-Mota and M. Montiel-Herrera, Polymers, 2018, 10, 342 CrossRef PubMed.
- X.-Y. Wang, C.-S. Wang and M.-C. Heuzey, Int. J. Polym. Mater. Polym. Biomater., 2016, 65, 96–104 CrossRef CAS.
- J. Wang, L. Wang, H. Yu, Y. Chen, Q. Chen, W. Zhou, H. Zhang and X. Chen, Int. J. Biol. Macromol., 2016, 88, 333–344 CrossRef CAS PubMed.
- K. Khwaldia, A. H. Basta, H. Aloui and H. El-Saied, Carbohydr. Polym., 2014, 99, 508–516 CrossRef CAS PubMed.
- E. Abdelrazek, I. Elashmawi and S. Labeeb, Phys. B, 2010, 405, 2021–2027 CrossRef CAS.
- M. Shahbazi, G. Rajabzadeh and S. J. Ahmadi, Carbohydr. Polym., 2017, 157, 226–235 CrossRef CAS PubMed.
- A. Muxika, A. Etxabide, J. Uranga, P. Guerrero and K. De La Caba, Int. J. Biol. Macromol., 2017, 105, 1358–1368 CrossRef CAS PubMed.
- H. Wang, J. Qian and F. Ding, J. Agric. Food Chem., 2018, 66, 395–413 CrossRef CAS PubMed.
- E. A. Kamoun, E.-R. S. Kenawy and X. Chen, J. Adv. Res., 2017, 8, 217–233 CrossRef CAS PubMed.
- S. Mohanapriya, Polyvinyl Alcohol-Based Biocomposites and Bionanocomposites, 2023, pp. 59–79 Search PubMed.
- J. Zanela, A. P. Bilck, M. Casagrande, M. V. E. Grossmann and F. Yamashita, Polímeros, 2018, 28, 256–265 CrossRef.
- D. Gu, Y. Yang, M. Bakri, Q. Chen, X. Xin and H. A. Aisa, Phytochem. Anal., 2013, 24, 661–670 CrossRef CAS PubMed.
- B. Hamedi, A. G. Pirbalouti and F. Rajabzadeh, Ind. Crops Prod., 2022, 187, 115470 CrossRef CAS.
- H.-J. Choi, S.-W. Choi, N. Lee and H.-J. Chang, Foods, 2022, 11, 3963 CrossRef CAS PubMed.
- S. Islam, V. Jaiswal, B. Butola and A. Majumdar, Int. J. Biol. Macromol., 2023, 252, 126457 CrossRef PubMed.
- Y. Wang, Y. Yu, C. Shi, Y. Ren, J. Han and R. Wu, Int. J. Biol. Macromol., 2025, 308, 142299 CrossRef CAS PubMed.
- J. F. Rubilar, R. M. Cruz, H. D. Silva, A. A. Vicente, I. Khmelinskii and M. C. Vieira, J. Food Eng., 2013, 115, 466–474 CrossRef CAS.
- V. G. Bhat, S. S. Narasagoudr, S. P. Masti, R. B. Chougale, A. B. Vantamuri and D. Kasai, Int. J. Biol. Macromol., 2022, 200, 50–60 CrossRef CAS PubMed.
- M. Mujahid, M. Zubair, A. Yaqoob, S. Shahzad and A. Ullah, Bioengineering, 2025, 12, 439 CrossRef CAS PubMed.
- A. Hassan, M. B. K. Niazi, A. Hussain, S. Farrukh and T. Ahmad, J. Polym. Environ., 2018, 26, 235–243 CrossRef CAS.
- S. Shahzad, S. Ahmed, A. Yaqoob, S. Saleem, S. Shaheen, K. Mammadova, A. K. Qureshi, M. Arshad, A. Saeed and D. H. M. Alkhalifah, Chem. Pap., 2025, 1–16 Search PubMed.
- S. Vasi, G. Ceccio, A. Cannavò, P. Pleskunov and J. Vacík, Sustainability, 2022, 14, 5863 Search PubMed.
- D. Yun and J. Liu, Foods, 2024, 13, 1174 CrossRef CAS PubMed.
- L. W. Winkler, Ber. Dtsch. Chem. Ges., 1888, 21, 2843–2854 Search PubMed.
- H. Bauer, F. Paronetto, W. A. Burns and A. Einheber, J. Exp. Med., 1966, 123, 1013–1024 CrossRef CAS PubMed.
- M. Abbas, M. Arshad, M. Rafique, A. Altalhi, D. Saleh, M. Ayub, S. Sharif, M. Riaz, S. Alshawwa and N. Masood, Arab. J. Chem., 2022, 15, 103766 Search PubMed.
- T. Wang, X. Zhai, X. Huang, Z. Li, X. Zhang, X. Zou and J. Shi, Food Packag. Shelf Life, 2023, 39, 101133 CrossRef CAS.
- M. Nowacka, K. Rybak, A. Wiktor, A. Mika, P. Boruszewski, J. Woch, K. Przybysz and D. Witrowa-Rajchert, Food Control, 2018, 93, 183–190 CrossRef CAS.
- F. Nugroho, N. Nizardo and E. Saepudin, AIP Conf. Proc., 2020, 2242, 040040 CrossRef CAS.
- D. Yun, Y. Qin, J. Zhang, M. Zhang, C. Qian and J. Liu, Int. J. Biol. Macromol., 2021, 189, 900–909 CrossRef CAS PubMed.
- M. Zubair, S. Hussain, A. Hussain, M. E. Akram, S. Shahzad, Z. Rauf, M. Mujahid and A. Ullah, Biomater. Sci., 2025, 13, 130–160 Search PubMed.
- N. K. Howell, G. Arteaga, S. Nakai and E. C. Li-Chan, J. Agric. Food Chem., 1999, 47, 924–933 CrossRef CAS PubMed.
- M. Cantor, E. Buta, I. Conţiu, R. Ştefan, I. Crişan and T. Buru, Emir. J. Food Agric., 2021, 33, 899–908 Search PubMed.
- R. Różyło, R. Amarowicz, M. A. Janiak, M. Domin, I. Różyło, K. Rząd, A. Matwijczuk, R. Rusinek and M. Gancarz, Molecules, 2024, 29, 4931 CrossRef PubMed.
- D. Hu, X. Liu, Y. Qin, J. Yan, J. Li and Q. Yang, Food Qual. Saf., 2022, 6, fyac028 CrossRef.
- G. Qiao, Z. Xiao, W. Ding and A. Rok, Coatings, 2019, 9, 828 CrossRef CAS.
- D. Lin and Y. Zhao, Compr. Rev. Food Sci. Food Saf., 2007, 6, 60–75 CrossRef CAS.
- Y. Peng, Y. Wu and Y. Li, Int. J. Biol. Macromol., 2013, 59, 282–289 CrossRef CAS PubMed.
- U. Siripatrawan and B. R. Harte, Food Hydrocoll., 2010, 24, 770–775 CrossRef CAS.
- X. Wang, H. Yong, L. Gao, L. Li, M. Jin and J. Liu, Food Hydrocoll., 2019, 89, 56–66 CrossRef CAS.
- F. Bigi, H. Haghighi, H. W. Siesler, F. Licciardello and A. Pulvirenti, Food Hydrocolloids, 2021, 120, 106979 CrossRef CAS.
- H.-S. Han and K. B. Song, Food Hydrocolloids, 2021, 112, 106372 CrossRef CAS.
- T. A. Dinh, Y. N. Le, N. Q. Pham, P. Ton-That, T. Van-Xuan, T. G.-T. Ho, T. Nguyen and H. H. K. Phuong, Prog. Org. Coat., 2023, 183, 107772 CrossRef CAS.
- Annu, A. Ali and S. Ahmed, Heliyon, 2021, 7(3), e06550 CrossRef CAS PubMed.
- L. Wang, Y. Dong, H. Men, J. Tong and J. Zhou, Food Hydrocoll., 2013, 32, 35–41 Search PubMed.
- S. B. Ghelejlu, M. Esmaiili and H. Almasi, Int. J. Biol. Macromol., 2016, 86, 613–621 CrossRef PubMed.
- J. Hafsa, M. ali Smach, M. R. B. Khedher, B. Charfeddine, K. Limem, H. Majdoub and S. Rouatbi, LWT-Food Sci. Technol., 2016, 68, 356–364 CrossRef CAS.
- S. Yadav, G. Mehrotra, P. Bhartiya, A. Singh and P. Dutta, Carbohydr. Polym., 2020, 227, 115348 Search PubMed.
- C. Pagella, G. Spigno and D. M. De Faveri, Food Bioprod. Process., 2002, 80, 193–198 Search PubMed.
- B. Singh, J. Singh, V. Sharma, P. Sharma and R. Kumar, Hybrid Adv., 2023, 4, 100096 Search PubMed.
- N. Nowak, W. Grzebieniarz, A. Cholewa-Wójcik, L. Juszczak, A. Konieczna-Molenda, E. Dryzek, M. Sarnek, M. Szuwarzyński, T. Mazur and E. Jamróz, Food Bioprocess Technol., 2024, 17, 1201–1214 Search PubMed.
- L. K. Winkelströter, E. Bezirtzoglou and F. L. Tulini, Front. Microbiol., 2022, 13, 1–3 Search PubMed.
- D. Altiok, E. Altiok and F. Tihminlioglu, J. Mater. Sci.: Mater. Med., 2010, 21, 2227–2236 Search PubMed.
- Y. Xing, Q. Xu, X. Li, C. Chen, L. Ma, S. Li, Z. Che and H. Lin, Int. J. Polym. Sci., 2016, 2016, 4851730 Search PubMed.
- D. Xu, T. Chen and Y. Liu, Polym. Bull., 2021, 78, 3607–3624 CrossRef CAS.
- M. Moghadam, M. Salami, M. Mohammadian, M. Khodadadi and Z. Emam-Djomeh, Food Hydrocolloids, 2020, 104, 105735 CrossRef CAS.
- A. Riaz, C. Lagnika, H. Luo, Z. Dai, M. Nie, M. M. Hashim, C. Liu, J. Song and D. Li, Int. J. Biol. Macromol., 2020, 150, 595–604 CrossRef CAS PubMed.
- T. Gasti, S. Dixit, S. P. Sataraddi, V. D. Hiremani, S. P. Masti, R. B. Chougale and R. B. Malabadi, J. Polym. Environ., 2020, 28, 2918–2930 CrossRef CAS.
- J. B. Harborne, Phytochemistry, 1983, 22, 1683–1684 CrossRef.
- S. Kayahan, F. Gülbağ, Y. Kaya and H. Altunkanat, Hortic. Stud., 2024, 41, 74–81 CrossRef.
- Z. T. Murathan, M. Zarifikhosroshahi, E. Kafkas and E. Sevindik, Ital. J. Food Sci., 2016, 28, 314–325 CAS.
- G. Fascella, F. D'Angiolillo, M. M. Mammano, M. Amenta, F. V. Romeo, P. Rapisarda and G. Ballistreri, Food Chem., 2019, 289, 56–64 CrossRef CAS PubMed.
- S. Shameh, B. Hosseini, A. Alirezalu and R. Maleki, J. AOAC Int., 2018, 101, 1788–1793 CrossRef CAS PubMed.
- M. Tabaszewska and D. Najgebauer-Lejko, NFS J., 2020, 21, 50–56 CrossRef.
- A. Y. Aldhebiani and W. A. Yaslam, Pak. J. Bot., 2023, 55, 995–999 CAS.
- N. Mukhtar, N. A. Marzuki, N. M. Zain, L. Naher, N. M. Hairin and N. M. Arsab, IOP Conf. Ser.: Earth Environ. Sci., 2024, 1397, 1–9 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |