Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Microwave-assisted extraction as a fast and sustainable approach for the recovery of caffeine and chlorogenic acids from coffee silverskin: evaluation of operating conditions and comparison with conventional extraction
Juliana A. Barreto-Peixoto†
 *,
Marlene Machado†
*,
Marlene Machado† ,
Ana L. Silva,
Catarina S. M. Martins,
Susana Machado,
Anabela S. G. Costa,
João A. V. Prior
,
Ana L. Silva,
Catarina S. M. Martins,
Susana Machado,
Anabela S. G. Costa,
João A. V. Prior *,
M. Beatriz P. P. Oliveira
*,
M. Beatriz P. P. Oliveira and
Rita C. Alves
and
Rita C. Alves *
*
LAQV REQUIMTE, Faculty of Pharmacy, University of Porto, R. J. Viterbo Ferreira, 228, 4050-313, Porto, Portugal. E-mail: jpeixoto@ff.up.pt; joaoavp@ff.up.pt; rcalves@ff.up.pt
First published on 9th February 2026
Abstract
The recovery of bioactive compounds in a fast, sustainable, and efficient way from food matrices, particularly from food by-products, is increasingly a key priority for industries. This study aimed to evaluate microwave-assisted extraction (MAE) conditions for the recovery of caffeine and chlorogenic acids (CGAs) from coffee silverskin, the main by-product of coffee roasting industries, and to compare its performance with that of a conventional extraction under selected conditions. Different temperatures and times were tested in MAE to maximize caffeine and CGA yields, which were analyzed by reverse-phase high-performance liquid chromatography-photodiode array detection (RP-HPLC-DAD). MAE evaluation showed that caffeine extraction was stable across temperatures and times, while CGA contents were higher at 80 °C combined with 5 minutes of extraction, with 5-CQA being the most abundant CGA. Above this temperature and for longer extraction times (beyond 10 minutes), isomerization of 5-CQA to 3-CQA and 4-CQA and degradation of CGA were noticed. Comparing the best conventional extract with MAE extracts, MAE provided higher CGA (but not caffeine) yields in a significantly shorter time using only water as a solvent, contrasting with hydroethanolic solvent and 120 minutes of extraction used in conventional extraction. Additionally, this study also found that the grinding degree influenced CGA extraction, with finer grinding improving recovery. Therefore, these results show that MAE has great potential to be used as a sustainable, cost- and time-effective alternative for the recovery of bioactive compounds from coffee silverskin, thus promoting its valorization and enhancing the environmental performance of coffee roasting industries.
Sustainability spotlightCoffee silverskin, the major by-product of coffee roasting industries, is often discarded despite being rich in bioactive compounds with potential health benefits. This study presents a promising sustainable solution for the recovery of these bioactives from coffee silverskin, by evaluating microwave-assisted extraction using only water as the extraction medium, thus eliminating organic solvents and significantly reducing extraction time and environmental impact compared to conventional methods. Hence, this work contributes to reducing waste and promoting a circular economy in the coffee industry and to taking advantage of the potential nutritional and functional value of coffee silverskin, aligning with the UN SDG 3 (Good Health and Well-Being), SDG12 (Responsible Consumption and Production), and SDG9 (Industry, Innovation and Infrastructure). |
1. Introduction
The coffee industry represents one of the largest agri-food value chains worldwide and, as a consequence, it is also responsible for the generation of huge amounts of by-products, from production to consumption.1 Around 7.5 kg of coffee silverskin are generated per ton of roasted coffee, representing the major by-product of the coffee roasting sector.2 Silverskin, although often discarded or used as a firelighter, a fertilizer, and animal feed,3,4 holds significant potential for valorization, due to its stability and composition rich in dietary fiber, proteins, and bioactive compounds, such as melanoidins, vitamins, minerals, caffeine, and phenolic compounds.5,6 In particular, chlorogenic acids (CGAs) are the major phenolics found in this by-product.7,8 This group of compounds is formed by esterification between trans-cinnamic acids (namely caffeic acid, ferulic acid, and p-coumaric acid) and quinic acid, giving rise to three major groups: caffeoylquinic acids (CQAs), feruloylquinic acids (FQAs), and coumaroylquinic acids (CoQAs).8 Among them, 5-caffeoylquinic acid (5-CQA) is the most abundant CGA in coffee silverskin.9,10 This compound has been widely studied for its potential health-promoting properties,7,8 with application in the food, pharmaceutical, and cosmetic industries. Simultaneously, its recovery from coffee by-products offers a sustainable solution for coffee roasting companies within the framework of a circular economy.11 Reported health benefits include the prevention of cardiovascular diseases, type 2 diabetes, and other metabolic disorders and antioxidant, anti-inflammatory, prebiotic, hepatoprotective, neuroprotective, and anticancer effects.10,12–17 These benefits have been closely associated not only with CGAs but also with caffeine,11,15,18 which may act synergistically,19 leading to increased interest in extracting these compounds from coffee silverskin.11The recovery of bioactive compounds from plant matrices, such as coffee silverskin, is dependent on several factors, namely the physical characteristics of the sample, the type of solvent used, temperature and time of extraction, the solid–liquid ratio, and the methodology employed.20,21 Conventional extraction methods have been widely used due to their efficiency and easy execution, but they are solvent-, time- and energy-consuming and usually require the use of organic solvents (e.g., ethanol, methanol, and acetone) that need to be further removed, making these techniques neither profitable nor environmentally friendly.22,23 Consequently, the scientific community has increasingly focused on studying and implementing alternative methodologies that can be as efficient and user-friendly as conventional methods, while being more economical, faster, and sustainable. In this context, microwave-assisted extraction (MAE) and ultrasound-assisted extraction (UAE) have emerged as innovative and environmentally friendly alternatives, offering highly efficient recovery of bioactive compounds in significantly shorter times, with reduced use of non-toxic solvents, such as water.20,22 In fact, previous studies conducted by our research group demonstrated that UAE enabled a higher yield of antioxidant compounds from coffee silverskin in a shorter time, using only water as the extracting medium, compared to a conventional solid–liquid extraction employing a hydroethanolic solvent.24 Besides, this methodology was scalable, making it a promising approach for the sustainable valorization of coffee silverskin at the industrial level.25
As mentioned above, MAE is another modern and green technique applicable to the extraction of bioactive components from natural matrices.26,27 It operates by applying electromagnetic radiation within the microwave frequency range (0.3 GHz to 300 GHz), inducing heating through two mechanisms: ionic conduction and dipole rotation. This increases internal pressure within plant cells and causes cellular disruption, thereby facilitating the release of target compounds into the extraction solvent.21–23 One of the main advantages of MAE is its significantly reduced extraction time compared to conventional methods. The rapid and uniform heating (both internally and externally, without thermal gradients) ensures efficient compound recovery while minimizing the degradation of thermolabile compounds and preventing sample oxidation.20–22 In addition, MAE provides high extraction yields with good reproducibility and requires substantially less solvent, making it a more ecological, efficient, and cost-effective process.20–22 Some studies investigated the extraction of compounds from coffee silverskin by MAE. For example, the extraction of caffeine, CGA and other phenolic and alkaloid compounds using ethanol28 and butylene glycol29 was reported. Furthermore, microwave-assisted alkaline and alkaline-acid extraction methods have been employed to extract proteins.26,30 Besides these solvent-based approaches, water has also been used as an extraction medium to recover dietary fiber27 and proteins31 from this by-product.
To the best of our knowledge, no studies have evaluated the use of water alone to extract caffeine and CGAs from coffee silverskin using MAE. Therefore, the aim of the present study was to evaluate MAE for the recovery of caffeine and CGAs from coffee silverskin by testing different temperatures and times of extraction, using water exclusively as the extraction medium. In parallel, conventional extracts were prepared under different conditions (temperature, time, and type of solvent), and the most effective extract was selected for comparison with the MAE extracts. The influence of the grinding degree on the extraction of bioactive compounds was also explored.
2. Experimental
2.1. Reagents and standards
Caffeine, 5-caffeoylquinic acid (5-CQA), 4-CQA, 3-CQA, 5-feruloylquinic acid (5-FQA), and 4-FQA standards were acquired from Sigma-Aldrich (St. Louis, MO, USA). Glacial acetic acid was from VWR Chemicals (Fontenay-sous-Bois, France). HPLC-grade methanol and absolute ethanol (99.8%) were purchased from Honeywell (Düsseldorf, Germany). Ultrapure water (18.2 MΩ cm) was obtained from a Direct-Pure UP Ultrapure & RO Lab Water System (RephiLe Bioscience ltd, Boston, MA, USA).2.2. Samples
Coffee silverskin, obtained after the roasting of industrial coffee batches and resulting from coffee blends comprising ∼40% arabica (Coffea arabica) and ∼60% robusta (Coffea canephora) coffees, was kindly provided by JMV – José Maria Vieira, S.A. After receipt, the sample was stored at room temperature (21 °C), protected from light in a dry place. Before extract preparation (using both conventional and MAE methods), the sample was homogenized and ground at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 20 seconds using a Grindomix GM 200 (Retsch, Haan, Germany).
000 rpm for 20 seconds using a Grindomix GM 200 (Retsch, Haan, Germany).
To ascertain the influence of the particle size on the bioactive compounds' extraction, a separate experiment was conducted using a new batch of coffee silverskin. In this experiment, sample G1 was prepared using the same grinding procedure described above. In addition, sample G2 underwent a more intensive grinding procedure consisting of three consecutive 20-second cycles at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, with short pauses between cycles to prevent overheating and possible compounds' degradation.
000 rpm, with short pauses between cycles to prevent overheating and possible compounds' degradation.
2.3. Conventional extraction
Different extraction conditions were selected to simulate a range of classical extraction parameters reported in the literature,20,32 enabling comparison of extraction efficiency using different solvents (water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol (1
ethanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and water), temperatures (room temperature (RT, 21 °C), 40 °C, and 60 °C), and times of extraction (10 min, 60 min, and 60 min followed by two re-extraction cycles of 30 min). This allowed the identification of the most effective conventional conditions before comparison with the MAE extracts. Table 1 summarizes the extraction conditions applied for each extract.
1) and water), temperatures (room temperature (RT, 21 °C), 40 °C, and 60 °C), and times of extraction (10 min, 60 min, and 60 min followed by two re-extraction cycles of 30 min). This allowed the identification of the most effective conventional conditions before comparison with the MAE extracts. Table 1 summarizes the extraction conditions applied for each extract.
| Type of extraction | Extract | Medium | Temperature (°C) | Time of extraction (min) | Two further re-extraction steps |
|---|---|---|---|---|---|
| a RT, room temperature (21 °C). | |||||
| Conventional | A | Water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol (1 ethanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
RT | 120 (60 + 30 + 30) | Yes |
| B | Water | RT | 120 (60 + 30 + 30) | Yes | |
| C | Water | RT | 60 | No | |
| D | Water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol (1 ethanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
RT | 60 | No | |
| E | Water | 40 | 120 (60 + 30 + 30) | Yes | |
| F | Water | 40 | 10 | No | |
| G | Water | 60 | 120 (60 + 30 + 30) | Yes | |
| H | Water | 60 | 10 | No | |
| MAE | MW40 | Water | 40 | 10 | No |
| MW50 | Water | 50 | 10 | No | |
| MW60 | Water | 60 | 10 | No | |
| MW70 | Water | 70 | 10 | No | |
| MW80 | Water | 80 | 10 | No | |
| MW90 | Water | 90 | 10 | No | |
| MW110 | Water | 110 | 10 | No | |
| MW130 | Water | 130 | 10 | No | |
| MW150 | Water | 150 | 10 | No | |
| MW170 | Water | 170 | 10 | No | |
| MW2′ | Water | 80 | 2 | No | |
| MW5′ | Water | 80 | 5 | No | |
| MW10′ | Water | 80 | 10 | No | |
| MW20′ | Water | 80 | 20 | No | |
| MW30′ | Water | 80 | 30 | No | |
| MW60′ | Water | 80 | 60 | No | |
| MW90′ | Water | 80 | 90 | No | |
Extracts obtained at room temperature were prepared according to Machado et al.33 with minor modifications. In brief, 500 mg of silverskin were extracted with 25 mL of solvent in a Multi Reax (1100 rpm, Heidolph, Schwabach, Germany) for 60 min and subsequently centrifuged (4500 rpm, 10 min, Megafuge 16 centrifuge, Heraeus, Hanau, Germany). For A and B extracts, the supernatant was collected and the solid residue was subjected to two additional re-extraction cycles of 30 min each using 10 mL of fresh solvent. After each cycle, the samples were centrifuged under the same conditions, and all supernatants were combined and homogenized.
Extractions at 40 °C and 60 °C were prepared according to a method previously optimized by Costa et al.,32 with minor modifications. Briefly, 500 mg of silverskin and 25 mL of solvent were mixed under constant magnetic stirring on a 10-position heating plate (DLAB Scientific, Beijing, China) at either 40 °C or 60 °C. Extractions were performed for 120 min (60 min followed by two additional 30 min re-extraction cycles for E and G extracts) or for 10 min (F and H extracts), after which the extracts were centrifuged using the same protocol as above. The re-extraction procedure for E and G was identical to that used for A and B.
Aliquots of all extracts (A–H) were analyzed by chromatography immediately after preparation, as described in Section 2.5. All extractions were performed in triplicate.
2.4. Microwave-assisted extraction (MAE)
Microwave-assisted hydrothermal extraction was carried out using a CEM Discover SP® microwave synthesizer (Matthews, NC, USA) operated with Synergy™ software v1.58. The same sample-to-solvent ratio (500 mg of coffee silverskin in 25 mL of water) used in the conventional extraction (Section 2.3) was also applied here to ensure direct comparability between methods.The microwave power was set at 300 W, corresponding to the maximum power output of the equipment. The evaluation of MAE conditions was performed using a sequential one-factor-at-a-time (OFAT) approach, in which extraction temperature and extraction time were varied independently to assess their individual influence on the yields of caffeine and CGA, expressed as mg per 100 g of dry weight (Table 1).
The experimental procedure was conducted in two steps. First, the extraction temperature was varied (40, 50, 60, 70, 80, 90, 110, 130, 150, and 170 °C) using a fixed extraction time of 10 minutes. The temperature range used was intended to detect the maximum extraction yields of both CGA and caffeine. After establishing the best temperature of extraction (80 °C), the extraction time was varied (2, 5, 10, 20, 30, 60, and 90 minutes) while keeping the temperature constant.
Aliquots of each extract were analyzed by chromatography immediately after preparation, as detailed in Section 2.5. All extractions were performed in triplicate.
2.5. Determination of caffeine contents and CGA profile by RP-HPLC-DAD
For chromatographic analysis, all extract aliquots were first centrifuged at 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 min (Heraeus Fresco 17 Centrifuge, Thermo Fisher Scientific, Schwerte, Germany) and then transferred to injection vials. The analysis was carried out as previously described by Machado et al.33 using an integrated HPLC system (Jasco, Tokyo, Japan) with an LC-Net II/ADC hardware interface, an AS-950 automated injector (20 µL loop), two Jasco PU-2080 Plus HPLC pumps, an MD-2010 Plus multiwavelength diode array detector, and a CO-2060 Plus column oven. Compound separation was achieved on a reversed-phase Zorbax-SB-C18 (5 µm; 250 mm × 4.6 mm) column from Agilent Technologies (Santa Clara, CA, USA), maintained at 28 °C. The mobile phase consisted of 0.5% acetic acid (A) and methanol (B), applied using the following gradient: 0′, 5% B; 40′, 25% B; 55′, 45% B; 60′, 60% B; 65′ 5% B, with a flow rate of 1.1 mL min−1. Caffeine and CGA were detected at 274 nm and 320 nm, respectively. The identification of compounds was performed by comparing retention times, elution order, and UV absorption spectra with those of authentic standards. The results were expressed as mg of compound per 100 g of dry weight (dw) sample.
000 rpm for 10 min (Heraeus Fresco 17 Centrifuge, Thermo Fisher Scientific, Schwerte, Germany) and then transferred to injection vials. The analysis was carried out as previously described by Machado et al.33 using an integrated HPLC system (Jasco, Tokyo, Japan) with an LC-Net II/ADC hardware interface, an AS-950 automated injector (20 µL loop), two Jasco PU-2080 Plus HPLC pumps, an MD-2010 Plus multiwavelength diode array detector, and a CO-2060 Plus column oven. Compound separation was achieved on a reversed-phase Zorbax-SB-C18 (5 µm; 250 mm × 4.6 mm) column from Agilent Technologies (Santa Clara, CA, USA), maintained at 28 °C. The mobile phase consisted of 0.5% acetic acid (A) and methanol (B), applied using the following gradient: 0′, 5% B; 40′, 25% B; 55′, 45% B; 60′, 60% B; 65′ 5% B, with a flow rate of 1.1 mL min−1. Caffeine and CGA were detected at 274 nm and 320 nm, respectively. The identification of compounds was performed by comparing retention times, elution order, and UV absorption spectra with those of authentic standards. The results were expressed as mg of compound per 100 g of dry weight (dw) sample.
2.6. Statistical analysis
Statistical analysis was performed using IBM SPSS 30 for macOS (SPSS Inc., Chicago, IL, USA). One-way ANOVA was used to evaluate significant differences between samples, followed by Tukey's HSD post hoc test to assess multiple comparisons. The results were considered statistically significant at p < 0.05 and were expressed as mean ± standard deviation (n = 3).3. Results and discussion
3.1. Comparison of conventional extracts
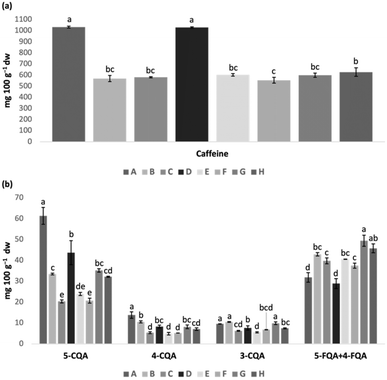
Conventional extracts prepared using different solvents, temperatures, and times of extraction were first compared regarding caffeine and CGA contents in order to select the best conditions to compare with MAE. The results are depicted in Fig. 1 and detailed in Table S1 in the SI.The results showed, as expected, that the type of solvent used for extraction significantly affected caffeine contents, as the only two hydroethanolic extracts prepared (A and D) presented significantly higher amounts of caffeine (almost double) than the aqueous extracts (Fig. 1a). Regarding CGA contents (Fig. 1b), similar findings were also observed for 5-CQA, but for 5-FQA and 4-FQA (which eluted together) opposite results were noticed, that is, the aqueous B and C extracts presented significantly higher amounts of these two combined compounds than the hydroethanolic analogous extracts (A and D, respectively). These results were, in truth, unexpected, as FQA tends to be more hydrophobic than CQA due to the presence of a methoxy (OCH3) group rather than the hydroxyl (OH) group found in CQA.
Furthermore, when comparing the extracts subjected to a single extraction of 60 min (C and D) with those undergoing two additional re-extraction cycles of 30 min (A and B, a total extraction time of 120 min), the re-extraction process did not seem to present advantages regarding caffeine extraction since the composition of A and B extracts (hydroethanolic and aqueous extracts where re-extraction was performed) was similar to that obtained for the homologous extracts without re-extraction (D and C, respectively) (Fig. 1a). Similar findings were also noticed for FQA. However, re-extraction appears to have significantly influenced CQA contents since the composition of A and B was, in general, significantly higher than that obtained with the homologous extracts without re-extraction (D and C, respectively, Fig. 1b).
Regarding the temperature factor, three different extracts prepared under similar conditions (water as solvent and 60 min of extraction with further two re-extractions for 30 min) but at three different temperatures (room temperature – B; 40 °C – E; 60 °C – G) were compared. Caffeine contents were not affected by the temperatures tested (no significant differences found, p > 0.05. Fig. 1a). On the other hand, temperature significantly affected the extraction of CGA (Fig. 1b): the extracts prepared at 60 °C (G) and at room temperature (B) resulted in significantly higher amounts of 5-CQA, 4-CQA, and 3-CQA than those obtained at 40 °C. Regarding 5-FQA and 4-FQA combined isomers, the best extraction temperature was 60 °C (significantly higher contents than those obtained at room temperature and 40 °C). Thus, in general, extraction at 60 °C allowed a greater recovery of CGA.
Considering that extracts prepared by MAE aimed to reduce the time of extraction and use an environmentally friendly solvent (water), two additional conventional extractions were performed, at two different temperatures (40 °C – F – and 60 °C – H) using only water as solvent for 10 min, in order to compare with the conventional extracts prepared under similar conditions but at different extraction times (E and G, respectively) and, further, with microwave-assisted extracts. The results show that caffeine and CGA contents were not affected by extraction time, since the amounts obtained in 120 min (60 + 30 + 30 min, E and G extracts) were similar to those obtained in 10 min (F and H). Nonetheless, contrary to what was observed above for E and G, for shorter extraction times, temperature significantly affected the extraction of caffeine and 5-CQA and FQA, since the extraction at 60 °C for 10 min (H) presented significantly higher amounts of these compounds than the one performed at 40 °C (F).
In general, caffeine extraction was mainly affected by the type of solvent, while both solvent and temperature appeared to be the factors that most influenced the extraction of CGA. In fact, A was the extract with the highest content of CGA, followed by G, B, and H. In addition, A was also the one with the highest amount of caffeine. Based on these achievements, the A extract was deliberately selected as the reference extract for comparison with the extracts obtained by MAE, which is the central focus of the present study.
3.2. Evaluation of microwave-assisted extraction (MAE) conditions
As mentioned in the Introduction section, this study aimed to evaluate MAE conditions for the extraction of caffeine and CGA from coffee silverskin using a safe and sustainable solvent (water). Extraction temperature and time were varied at a fixed microwave power of 300 W. This setting was selected to ensure efficient energy transfer from the microwave field to the solvent, enabling rapid and uniform heating and promoting effective solvent penetration into the dense plant matrix of coffee silverskin. Fixing at 300 W also guarantees consistent energy input across all trials, which is essential for reproducibility and method standardization. This decision is further supported by literature demonstrating that 300 W is a suitable and effective power level for extracting phenolic and antioxidant compounds from complex plant matrices. For example, Cerdá-Bernad et al.34 used 300 W during the microwave-assisted extraction of phenolic compounds from Crocus sativus L. floral by-products, achieving high antioxidant activity with reduced extraction time and energy consumption. Likewise, Oufighou et al.35 reported that a microwave power setting of 300 W was optimal for recovering phenolics from Opuntia ficus-indica cladodes, using a predictive optimization strategy. Both studies demonstrate that this power level ensures high extraction efficiency while preserving the integrity of thermolabile bioactives.The following two subsections present the results and discussion that enabled the selection of the best temperature and time for extraction, respectively, which were further compared in Subsection 3.2.3.
| Extract | Caffeine | 5-CQA | 4-CQA | 3-CQA | 5-FQA + 4-FQA | ΣCGA |
|---|---|---|---|---|---|---|
| a Results are expressed as mean ± standard deviation. For each compound (within each column), different letters denote significant differences between samples at p < 0.05. CQA, caffeoylquinic acid; FQA, feruloylquinic acid. | ||||||
| MW40 | 481.50 ± 18.50e | 26.05 ± 3.70e | 13.64 ± 0.40d | 9.72 ± 0.57 f | 37.73 ± 0.92cd | 87.14 |
| MW50 | 510.68 ± 26.66de | 32.66 ± 4.90cde | 17.44 ± 1.51cd | 11.60 ± 1.04ef | 39.45 ± 5.28cd | 101.15 |
| MW60 | 531.80 ± 11.86cde | 37.75 ± 1.41abcd | 16.98 ± 0.96cd | 13.35 ± 0.42ef | 43.24 ± 3.09abc | 111.32 |
| MW70 | 548.57 ± 9.96cd | 39.10 ± 3.21abc | 18.59 ± 1.63cd | 14.18 ± 0.83e | 44.20 ± 1.22abc | 116.07 |
| MW80 | 546.16 ± 11.36cd | 44.33 ± 3.71a | 23.56 ± 3.37bc | 18.24 ± 2.10d | 48.91 ± 5.62ab | 135.04 |
| MW90 | 560.02 ± 21.74bcd | 41.37 ± 2.37abc | 31.04 ± 2.82a | 24.36 ± 1.93bc | 48.60 ± 3.95ab | 145.37 |
| MW110 | 564.63 ± 2.68bc | 42.14 ± 2.66ab | 34.49 ± 4.09a | 26.08 ± 2.61abc | 50.07 ± 1.30a | 152.78 |
| MW130 | 610.14 ± 14.38ab | 29.24 ± 3.56de | 32.79 ± 3.92a | 29.51 ± 1.39a | 40.05 ± 2.57bcd | 131.59 |
| MW150 | 649.39 ± 6.56a | 27.69 ± 1.50e | 30.16 ± 1.46ab | 27.10 ± 1.55ab | 40.21 ± 0.61bcd | 125.16 |
| MW170 | 577.02 ± 8.14cd | 33.71 ± 0.02bcde | 30.02 ± 1.53ab | 22.62 ± 0.37c | 32.97 ± 1.05d | 119.32 |
Caffeine contents were relatively similar at the temperatures tested, which demonstrates its extractability in water and thermostability, already well documented in the literature.37,38 From 130 °C onwards, significant differences were observed, with better extraction of this compound, which in turn increased at 150 °C. Considering these results, and in order to determine the maximum extraction yields of caffeine, a further extraction at 170 °C was subsequently performed. A significant decrease in the amounts of caffeine was found, thus indicating degradation at this temperature. Thus, the maximum caffeine extraction peak occurred at 150 °C.
Regarding the CGA contents, the major compound obtained at temperatures up to 110 °C was 5-CQA, which is consistent with literature data.24 Furthermore, although it was not possible to separate 5-FQA and 4-FQA, since they eluted together, it is expected that 5-FQA was present in higher amounts than 4-FQA, based on previous studies.25,39 Nonetheless, as the temperature increased (mainly from 80 °C onwards), a decrease in the major CGA (5-CQA) was noticed, with a simultaneous increase in its respective isomers (3-CQA and 4-CQA), which suggests that 5-CQA is converted into their isomers. These findings can be more easily noticed in Fig. S1 in the SI. In fact, several studies have shown that CGAs possess low thermal stability and that 5-CQA is easily isomerized to 3-CQA and 4-CQA and also transformed and/or degraded into products derived from trans-cis isomerization, hydrolysis, esterification, and addition of water molecules to the double bond, including during the early stages of extraction,22,40–43 thus corroborating our results. In addition, it is reported that isomerization to 4-CQA is more predominant than to 3-CQA,41 which was also found in our results. Considering the sum of all CGAs, the temperature at which more CGA was extracted was 110 °C, followed by 90 °C and 80 °C. However, despite the three isomers possessing similar biological effects in vitro,44 it is described in the literature that 3-CQA and 4-CQA are less stable42 and also less bioavailable than 5-CQA.45 For this reason, the preservation of 5-CQA is particularly relevant when aiming to obtain extracts for potential applications in functional foods or pharmaceutical fields. Hence, considering that 5-CQA was found in smaller quantities at 90 °C and 110 °C, 80 °C was considered the most suitable temperature for extraction of all CGAs in reasonable quantities, without potential isomerization and/or degradation of compounds, and, for this reason, this temperature was selected to perform subsequent evaluation of the influence of time on MAE of coffee silverskin. In addition, it is interesting to note that although it is described in the literature that CGAs are thermosensitive, these results demonstrate that they are better extracted at relatively high temperatures, namely at temperatures close to the boiling point of water (100 °C), as occurs in the preparation of infusions, for example. This can be explained by the influence of heating in rupturing the cell membrane, which facilitates the release of membrane-bound phytochemicals (namely CGAs). In particular, as explained in the Introduction section, during MAE, molecules are heated up by ionic conduction and dipole rotation mechanisms, causing the disruption of cell walls and, consequently, the release of bioactive compounds.21 Even though, for temperatures above 110 °C, all CGA contents started to decrease, evidencing their degradation. In general, considering the contents of both CGA and caffeine throughout the different temperatures tested and having in view an industrial context, it can be suggested that MAE should not be used at high temperatures due to the potential degradation of bioactive compounds of interest present in coffee silverskin.
Furthermore, on comparing these extracts with the ones prepared by conventional extraction using the same extraction time (10 min) and using only water as solvent (F and H extracts (Fig. 1 or Table S1 in the SI)), it is possible to notice that MAE allowed, in general, a greater extraction of CGA, at all the temperatures tested. Therefore, this shows that MAE has great potential to be used as an alternative to conventional extraction methods.
| Extract | Caffeine | 5-CQA | 4-CQA | 3-CQA | 5-FQA + 4-FQA | ΣCGA |
|---|---|---|---|---|---|---|
| a Results are expressed as mean ± standard deviation. For each compound (within each column), different letters represent significant differences between samples at p < 0.05. CQA, caffeoylquinic acid; FQA, feruloylquinic acid. | ||||||
| MW2′ | 596.50 ± 20.87a | 66.34 ± 3.87ab | 27.40 ± 3.34bc | 14.26 ± 0.89d | 60.42 ± 6.22ab | 168.43 |
| MW5′ | 606.23 ± 7.66a | 71.37 ± 4.88a | 28.33 ± 1.44bc | 17.67 ± 0.81cd | 70.22 ± 3.60a | 187.60 |
| MW10′ | 602.31 ± 13.97a | 61.52 ± 6.05ab | 24.49 ± 0.84c | 15.44 ± 0.53d | 57.76 ± 3.16b | 159.21 |
| MW20′ | 606.59 ± 23.32a | 57.49 ± 1.67ab | 30.92 ± 0.18b | 20.40 ± 0.44bc | 57.80 ± 2.77b | 166.61 |
| MW30′ | 586.91 ± 25.59a | 54.70 ± 7.40b | 31.25 ± 1.88b | 21.44 ± 0.89b | 59.21 ± 3.48b | 166.60 |
| MW60′ | 596.66 ± 23.30a | 58.20 ± 3.62ab | 39.17 ± 1.96a | 28.57 ± 1.79a | 62.01 ± 2.92ab | 187.95 |
| MW90′ | 609.30 ± 23.80a | 53.36 ± 3.04b | 39.08 ± 0.52a | 29.60 ± 1.11a | 59.60 ± 1.77ab | 181.63 |
Caffeine extraction was very similar at the different times tested (no significant differences found, p > 0.05), which denotes, once again, its stability. Nonetheless, contrary to what was observed with the different temperatures tested, the caffeine concentration did not increase over time. Therefore, caffeine extraction by MAE was shown to be dependent on extraction temperature, but not on extraction time.
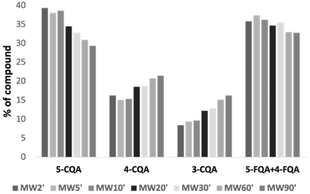
Regarding the CGA contents, the best extraction time was clearly 5 min, since the MW5′ extract presented the highest contents of the major CGA (5-CQA) and the highest levels of total CGA. This time of extraction (5 min) was also the one resulting in the greatest extraction of CGA and caffeine from green coffee beans in a study performed by Upadhyay et al.46 As observed for temperature, increasing extraction time appears to have promoted isomerization of the main CGA into its isomers, as the decrease in 5-CQA started concomitantly with increases in 3-CQA and 4-CQA. However, this phenomenon seems to have been more evident with the temperature factor. In addition, at longer extraction times (60 and 90 min), the amounts of minor CGA (3-CQA and 4-CQA) were significantly higher than those at the remaining times of extraction. Furthermore, the amounts of total CGA at these times were similar to those obtained at the best extraction time (5 min) and greater than those obtained at 10, 20, and 30 min. This may indicate that, in addition to the isomerization of 5-CQA, which leads to an increase of minority compounds over time, a greater extraction of these minor compounds for longer extraction times has also occurred, explaining the greater amounts of total CGA obtained at 60 and 90 min compared to 10, 20, and 30 min. Considering the proportion of CGA (Fig. 2), interesting findings were also noticed: up to 10 min of extraction (MW2′, MW5′, and MW10′ extracts), the proportions of CGA were rather similar, but from 20 min of extraction, they started to vary, with a decrease of 5-CQA and an increase of 3-CQA and 4-CQA. Therefore, at 80 °C (temperature selected and used for time evaluation), isomerization and/or degradation of 5-CQA may start to be more evident after 20 min of extraction.
Overall, extraction of CGA was significantly affected by time: the best yields were achieved at 5 min, and longer extraction times negatively affected CGA contents. In fact, several studies have demonstrated that long extraction times may result in the degradation of phenolic compounds. More concretely, some studies reported maximum extraction yields of total phenolic contents23 and CGA and caffeine46 by MAE at 5 min of extraction, and it was reported that extraction times longer than 10 min did not result in higher total phenolic contents but, instead, in a decrease after 10–15 min of extraction,23 which was also observed in the present study for CGA contents. These findings can be explained by the fast heating and destruction of the plant cell structure achieved by MAE, which allows the efficient extraction of compounds at shorter times.21,23
3.3. Comparison of the best extraction conditions
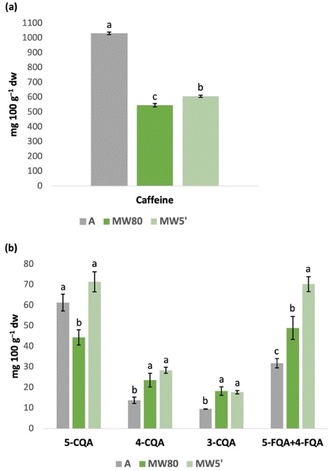
The best extraction conditions selected in the previous sections (A, MW80, and MW5′) were compared (Fig. 3). Clearly, conventional extraction (A) extracted more caffeine (almost double) than extracts obtained by MAE (MW80 and MW5′), which is related to the solvent used, that is, the hydroethanolic solvent used in the A extract allowed better caffeine extraction than the water used in MAE, which was also observed when comparing different conventional extracts (Section 3.1). In fact, extracts obtained by MAE extracted similar amounts of caffeine to those obtained with aqueous extracts prepared by the conventional method, showing that caffeine extraction is mostly dependent on the type of solvent and not so much on other factors such as temperature, the time of extraction, or the extraction method selected. On the other hand, in relation to CGA, the MW5′ extract (5 min, 80 °C) presented the best amounts of CGA and, in general, MAE presented a significantly better CGA extraction performance than conventional extraction. Considering the aim to find more sustainable alternatives for industries to recover bioactive compounds from coffee by-products in an economic and time-costly manner, MAE proved to be a viable option to be considered, as it allowed the recovery of adequate amounts of caffeine and CGA from coffee silverskin using a green solvent (water) and in a significantly faster way (only 5 min) compared to conventional extraction. In fact, MAE is already applied in the food industry,23 so the application particularly in coffee industries is possible and could not only add value to coffee by-products but also reward the stakeholders in this sector.3.4. Influence of the degree of grinding on the extraction of bioactive compounds
Considering the results previously obtained, we found that MW80 and MW10′, which were prepared under the same MAE conditions, presented slight differences in caffeine and CGA contents, particularly in caffeine and 5-CQA levels (whose levels were higher in MW10′ than in MW80) which, at first glance, would not be expected. These differences might be explained, in addition to other factors, by the sample being resultant from roasting the coffee blend comprising both arabica and robusta, which means that the exact percentage of each species present in our sample was not exactly known and, hence, even though the sample was as homogenized as possible before extract preparation, the part of the sample used in the preparation of the MW80 extract might have contained, for example, more arabica than robusta, resulting in an extract with a smaller quantity of caffeine and 5-CQA compared to MW10′, as it is well known that robusta is richer in these compounds.47 In addition, differences in grinding fineness might also have influenced the observed variability. As mentioned above, despite careful sample homogenization prior to extraction, the variations in particle size cannot be excluded, as no additional sieving step was performed before extraction. Therefore, considering that particle size directly affects the contact surface area with the solvent, such heterogeneity could have influenced extraction efficiency, resulting in the observed differences in caffeine and 5-CQA contents. Thus, we further decided to investigate the influence of this factor on the extraction of caffeine and CGA from silverskin. Due to the lack of sample at the end of these experiments, a new coffee silverskin sample was supplied by the same coffee industry to carry out this new study, performed in parallel and à posteriori. This coffee silverskin sample was obtained from a new batch that was available according to the commercial roasts performed in the industry at that moment. After reception, it was subjected to two different degrees of grinding (G1, where the sample was subjected to the same grinding process as the sample used in previous sections, that is, ground at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 20 seconds (see Section 2.2), and G2, where the sample was subjected to a grinding process three times the time of G1) and a granulometry analysis was first performed using four sieves with different pore sizes (500, 200, 180, and 90 µm). The granulometry results are presented in Table 4. The G1 sample exhibited a more heterogeneous feature, as most of the sample possessed pore sizes ranging between 250 µm and 90 µm or lower. On the other hand, around 67% of the G2 sample presented pore sizes smaller than or equal to 90 µm, denoting a more homogeneous feature. These results are interesting and may help explain the differences between MW80 and MW10′: as the degree of grinding used in the preparation of these extracts (equal to that used in G1) presented pore sizes quite heterogeneous, the contact surface area of each pore size with the solvent was different and, hence, in each extraction, the homogeneity of pore sizes was not guaranteed, resulting in different extents of extraction. Therefore, the blend composition of the sample (comprising both arabica and robusta coffee silverskin) and the heterogeneity of ground pore sizes might explain the differences obtained between extracts prepared under the same conditions.
000 rpm for 20 seconds (see Section 2.2), and G2, where the sample was subjected to a grinding process three times the time of G1) and a granulometry analysis was first performed using four sieves with different pore sizes (500, 200, 180, and 90 µm). The granulometry results are presented in Table 4. The G1 sample exhibited a more heterogeneous feature, as most of the sample possessed pore sizes ranging between 250 µm and 90 µm or lower. On the other hand, around 67% of the G2 sample presented pore sizes smaller than or equal to 90 µm, denoting a more homogeneous feature. These results are interesting and may help explain the differences between MW80 and MW10′: as the degree of grinding used in the preparation of these extracts (equal to that used in G1) presented pore sizes quite heterogeneous, the contact surface area of each pore size with the solvent was different and, hence, in each extraction, the homogeneity of pore sizes was not guaranteed, resulting in different extents of extraction. Therefore, the blend composition of the sample (comprising both arabica and robusta coffee silverskin) and the heterogeneity of ground pore sizes might explain the differences obtained between extracts prepared under the same conditions.
| Pore size (µm) | % G1 | % G2 |
|---|---|---|
| 500 | 2.94 | 0.16 |
| 200 | 32.62 | 19.90 |
| 180 | 18.45 | 13.25 |
| 90 | 27.93 | 45.61 |
| <90 | 18.06 | 21.08 |
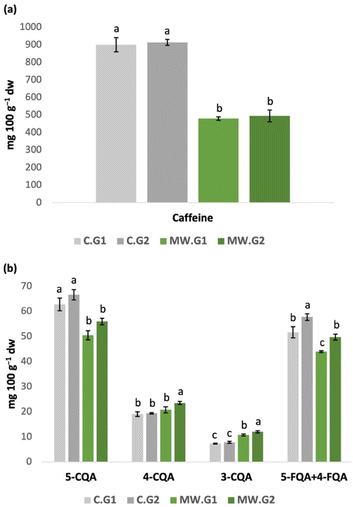
After performing the granulometry test, the best conventional extraction conditions (corresponding to the A extract) and the best MAE conditions (corresponding to the MW5′ extract) previously selected were used to prepare extracts with the different ground samples (G1 and G2). The results of caffeine and CGA contents of the four different extracts are presented in Fig. 4 or, alternatively, in Table S2 in the SI. The degree of grinding did not influence caffeine contents, as no significant differences were observed between C.G1 and C.G2 (conventional extracts) and between MW.G1 and MW.G2 (microwave-assisted extracts). However, in the case of CGA, the increase in the grinding degree positively influenced CGA extraction, as C.G2 and MW.G2 presented, in general, higher amounts of CGA (although not statistically significant for some of them) than the respective homologous extracts, which can be explained by the higher contact surface area of G2 particles with the solvent used, allowing a greater extraction of bioactive compounds. Therefore, these results showed us that, in future studies, it will be important to fractionally grind the sample in order to obtain the highest possible percentage of particles with the same pore size, ensuring not only greater sample homogeneity but also greater extraction of the compounds.
4. Conclusions
The present study demonstrates the potential of MAE as an efficient, time-saving, and environmentally friendly alternative to conventional extraction methods for the recovery of bioactive compounds from coffee silverskin. When compared to conventional extraction methods employing different solvents, temperatures, and extraction times, MAE provided higher CGA yields in a significantly shorter time using only water as solvent (thus avoiding the use of organic solvents), which represents a clear advantage from both environmental and industrial perspectives.More concretely, the comparison of the best conventional extraction conditions with those of MAE revealed that although conventional hydroethanolic extraction achieved higher caffeine contents, MAE was particularly advantageous for the recovery of CGA. Moreover, in the MAE study, it was found that caffeine extraction was stable across different temperatures tested, while CGA extraction was favoured at moderately high temperatures (80–110 °C), with 5-CQA being better preserved at 80 °C. Higher temperatures resulted in lower yields of 5-CQA and increased amounts of its isomers, suggesting isomerization and degradation. Moreover, the best extraction time for CGA was 5 minutes, thus highlighting short extraction time as a great advantage of MAE.
Additionally, the study also found that the degree of grinding influenced the extraction of CGA (but not caffeine), with finer grinding enhancing the recovery of these bioactive compounds.
Overall, this study demonstrates a novel and industry-relevant approach for the coffee roasting sector, showing that water-based MAE is a fast, efficient, and environmentally friendly strategy for the valorization of coffee silverskin, the major by-product of coffee roasting. The exclusive use of water as extraction solvent, together with the short extraction time, makes this technique particularly attractive for large-scale applications in food and pharmaceutical sectors, while also contributing to the circular economy of coffee roasting industries.
In future studies, it will be necessary to scale-up the MAE process and validate its performance under industrial conditions and to evaluate the stability of extracts during storage. Furthermore, having in view the incorporation of these extracts in functional foods or pharmaceutical formulations, further research should also assess the potential bioactivities of the extracts, together with their bioaccessibility and bioavailability.
Author contributions
Conceptualization, J. A. B.-P., M. M. and R. C. A.; methodology, J. A. B.-P., M. M., S. M., J. A. V. P. and R. C. A.; validation, J. A. V. P. and R. C. A.; formal analysis, J. A. B.-P. and M. M.; investigation, J. A. B.-P., M. M., A. L. S., C. S. M. M., S. M. and A. S. G. C.; resources: M. B. P. P. O. and R. C. A.; writing—original draft preparation, J. A. B.-P. and M. M.; writing—review and editing, J. A. B.-P., M. M., J. A. V. P., and R. C. A.; visualization, J. A. B.-P. and M. M.; supervision, J. A. V. P. and R. C. A.; project administration, R. C. A.; funding acquisition, M. B. P. P. O. and R. C. A. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting the findings of this research are available in the article and in the supplementary information (SI). Supplementary information: Table S1 – caffeine and CGA contents of conventional extracts; Table S2 – influence of grinding on caffeine and CGA contents of conventional extracts and MAE; Fig. S1 – influence of temperature on caffeoylquinic acid (CQA) contents. See DOI: https://doi.org/10.1039/d5fb00931f.Acknowledgements
This work was funded by FCT/MCTES (Fundação para a Ciência e Tecnologia/Ministério da Ciência, Tecnologia e Ensino Superior) through the project PTDC/SAU-NUT/2165/2021 (COBY4HEALTH—Can coffee by-products decrease the risk of metabolic syndrome?) A comprehensive approach to reduce waste and valorize health benefits; (https://doi.org/10.54499/PTDC/SAU-NUT/2165/2021). This work also received financial support from the PT national funds (FCT/MECI, Fundação para a Ciência e Tecnologia/Ministério da Educação, Ciência e Inovação) through the project UID/50006/2025 DOI: https://doi.org/10.54499/UID/50006/2025 – Laboratório Associado para a Química Verde – Tecnologias e Processos Limpos. J. A. B.-P., M. M., C. S. M. M. and A. L. S. thank FCT/MCTES (Fundação para a Ciência e Tecnologia/Ministério da Ciência, Tecnologia e Ensino Superior) and ESF (European Social Fund) through NORTE 2020 (Programa Operacional Região Norte) for their PhD grants (ref. SFRH/BD/07329/2021, DOI: https://doi.org/10.54499/2021.07329.BD, ref. SFRH/BD/04907/2021, DOI: https://doi.org/10.54499/2021.04907.BD, SFRH/BD/151097/2021, and 2021.09393.BD, respectively). R. C. A. thanks FCT for funding through the Scientific Employment Stimulus—Individual Call (CEECIND/01120/2017). The authors also thank JMV–José Maria Vieira, S. A., for kindly providing the samples for the study.References
- J. A. Barreto Peixoto, J. F. Silva, M. B. P. P. Oliveira and R. C. Alves, Compr. Rev. Food Sci. Food Saf., 2022, 22, 287–332 Search PubMed.
- R. C. Alves, F. Rodrigues, M. A. Nunes, A. F. Vinha and M. B. P. P. Oliveira, in Handbook of Coffee Processing By-Products, ed. C. M. Galanakis, Academic Press, 2017, ch. 1, pp. 1–26 Search PubMed.
- L. Lorbeer, S. Schwarz, H. Franke and D. W. Lachenmeier, Molecules, 2022, 27, 6839 Search PubMed.
- G. Picca, C. Plaza, E. Madejón and M. Panettieri, Waste Biomass Valorization, 2022, 14, 297–307 CrossRef.
- F. K. Nzekoue, G. Borsetta, L. Navarini, D. Abouelenein, J. Xiao, G. Sagratini, S. Vittori, G. Caprioli and S. Angeloni, Food Chem., 2022, 372, 131188 CrossRef CAS PubMed.
- S. Tores de la Cruz, A. Iriondo-DeHond, T. Herrera, Y. Lopez-Tofiño, C. Galvez-Robleño, M. Prodanov, F. Velazquez-Escobar, R. Abalo and M. D. d. Castillo, Foods, 2019, 8, 68 CrossRef PubMed.
- A. Bejaoui, Y. M'rabet and A. Boulila, Chem. Afr., 2024, 7, 4105–4126 CrossRef CAS.
- H. Lu, Z. Tian, Y. Cui, Z. Liu and X. Ma, Compr. Rev. Food Sci. Food Saf., 2020, 19, 3130–3158 CrossRef CAS PubMed.
- L. Bresciani, L. Calani, R. Bruni, F. Brighenti and D. Del Rio, Food Res. Int., 2014, 61, 196–201 CrossRef CAS.
- F. K. Nzekoue, S. Angeloni, L. Navarini, C. Angeloni, M. Freschi, S. Hrelia, L. A. Vitali, G. Sagratini, S. Vittori and G. Caprioli, Food Res. Int., 2020, 133, 109128 CrossRef CAS PubMed.
- S. E. Koskinakis, C. Stergiopoulos, C. Vasileiou and M. Krokida, Foods, 2025, 14, 615 CrossRef CAS PubMed.
- B. Fernandez-Gomez, A. Lezama, M. Amigo-Benavent, M. Ullate, M. Herrero, M. Á. Martín, M. D. Mesa and M. D. del Castillo, J. Funct. Foods, 2016, 25, 197–207 CrossRef CAS.
- A. Iriondo-DeHond, A. I. Haza, A. Ávalos, M. D. del Castillo and P. Morales, Food Res. Int., 2017, 100, 791–797 CrossRef CAS PubMed.
- A. Iriondo-DeHond, P. Martorell, S. Genovés, D. Ramón, K. Stamatakis, M. Fresno, A. Molina and M. Del Castillo, Molecules, 2016, 21, 721 Search PubMed.
- J. A. B. Peixoto, N. Andrade, S. Machado, A. S. G. Costa, M. B. P. P. Oliveira, F. Martel and R. C. Alves, Foods, 2022, 11, 3902 CrossRef CAS PubMed.
- M. Rebollo-Hernanz, Y. Aguilera, M. A. Martín-Cabrejas and E. Gonzalez de Mejia, Front. Nutr., 2022, 9, 866233 CrossRef PubMed.
- M. Rebollo-Hernanz, Q. Zhang, Y. Aguilera, M. A. Martín-Cabrejas and E. Gonzalez de Mejia, Food Chem. Toxicol., 2019, 132, 110672 CrossRef CAS PubMed.
- A. S. Franca, E. P. Basílio, L. M. Resende, C. A. Fante and L. S. Oliveira, Foods, 2024, 13, 3935 CrossRef CAS PubMed.
- M. Xu, L. Yang, Y. Zhu, M. Liao, L. Chu, X. Li, L. Lin and G. Zheng, Food Funct., 2019, 10, 7489–7497 RSC.
- A. F. Bondam, D. D. da Silveira, J. P. dos Santos and J. F. Hoffmann, Trends Food Sci. Technol., 2022, 123, 172–186 CrossRef CAS.
- N. W. Ismail-Suhaimy, S. S. A. Gani, U. H. Zaidan, M. I. E. Halmi and P. Bawon, Plants, 2021, 10, 682 CrossRef CAS PubMed.
- I. Frosi, I. Montagna, R. Colombo, C. Milanese and A. Papetti, Molecules, 2021, 26, 4515 CrossRef CAS PubMed.
- M. Tsevdou, A. Ntzimani, M. Katsouli, G. Dimopoulos, D. Tsimogiannis and P. Taoukis, Molecules, 2024, 29, 2303 CrossRef CAS PubMed.
- H. Puga, R. C. Alves, A. S. Costa, A. F. Vinha and M. B. P. P. Oliveira, J. Cleaner Prod., 2017, 168, 14–21 CrossRef CAS.
- J. A. B. Peixoto, N. Andrade, S. Machado, A. S. G. Costa, H. Puga, M. B. P. P. Oliveira, F. Martel and R. C. Alves, Foods, 2022, 11, 1671 Search PubMed.
- W. Jirarat, T. Kaewsalud, K. Yakul, P. Rachtanapun and T. Chaiyaso, Foods, 2024, 13, 1230 Search PubMed.
- L. Wen, Z. Zhang, M. Zhao, R. Senthamaraikannan, R. B. Padamati, D. W. Sun and B. K. Tiwari, Int. J. Food Sci. Technol., 2020, 55, 2242–2250 Search PubMed.
- A. Guglielmetti, V. D'Ignoti, D. Ghirardello, S. Belviso and G. Zeppa, Ital. J. Food Sci., 2017, 29, 409–423 Search PubMed.
- N. Khat-udomkiri and S. M. Win, Ind. Crops Prod., 2025, 226, 120647 CrossRef CAS.
- L. Wen, C. Álvarez, Z. Zhang, M. M. Poojary, M. N. Lund, D.-W. Sun and B. K. Tiwari, Biomass Convers. Biorefin., 2020, 11, 1575–1585 Search PubMed.
- V. Biondić Fučkar, A. Božić, A. Jukić, A. Krivohlavek, G. Jurak, A. Tot, S. Serdar, I. Žuntar and A. Režek Jambrak, Foods, 2023, 12, 518 Search PubMed.
- A. S. G. Costa, R. C. Alves, A. F. Vinha, S. V. P. Barreira, M. A. Nunes, L. M. Cunha and M. B. P. P. Oliveira, Ind. Crops Prod., 2014, 53, 350–357 Search PubMed.
- M. Machado, L. Espírito Santo, S. Machado, J. C. Lobo, A. S. G. Costa, M. B. P. P. Oliveira, H. Ferreira and R. C. Alves, Foods, 2023, 12, 2354 Search PubMed.
- D. Cerdá-Bernad, J. P. Baixinho, N. Fernández and M. J. Frutos, Foods, 2022, 11, 2335 Search PubMed.
- A. Oufighou, F. Brahmi, S. Achat, S. Yekene, S. Slimani, Y. Arroul, L. Boulekbache-Makhlouf and F. Blando, Processes, 2025, 13, 724 Search PubMed.
- D. Rente, M. Cvjetko Bubalo, M. Panić, A. Paiva, B. Caprin, I. Radojčić Redovniković and A. R. C. Duarte, J. Cleaner Prod., 2022, 380, 135147 CrossRef CAS.
- J. Grzelczyk, P. Fiurasek, A. Kakkar and G. Budryn, Food Chem., 2022, 387, 132888 CrossRef CAS PubMed.
- F. M. Mehaya and A. A. Mohammad, Heliyon, 2020, 6, e05508 Search PubMed.
- S. M. F. Bessada, R. C. Alves, A. S. G. Costa, M. A. Nunes and M. B. P. P. Oliveira, Sci. Total Environ., 2018, 645, 1021–1028 CrossRef CAS PubMed.
- G. Budryn, E. Nebesny, A. Podsędek, D. Żyżelewicz, M. Materska, S. Jankowski and B. Janda, Eur. Food Res. Technol., 2009, 228, 913–922 CrossRef CAS.
- A. L. Dawidowicz and R. Typek, J. Agric. Food Chem., 2010, 58, 12578–12584 Search PubMed.
- A. L. Dawidowicz and R. Typek, Eur. Food Res. Technol., 2016, 243, 379–390 CrossRef.
- F. Ianni, C. Barola, F. Blasi, S. Moretti, R. Galarini and L. Cossignani, Food Chem., 2022, 374, 131820 Search PubMed.
- J.-G. Xu, Q.-P. Hu and Y. Liu, J. Agric. Food Chem., 2012, 60, 11625–11630 CrossRef CAS PubMed.
- A. Farah, M. Monteiro, C. M. Donangelo and S. Lafay, J. Nutr., 2008, 138, 2309–2315 CrossRef CAS PubMed.
- R. Upadhyay, K. Ramalakshmi and L. Jagan Mohan Rao, Food Chem., 2012, 130, 184–188 CrossRef CAS.
- I. A. Ludwig, M. N. Clifford, M. E. J. Lean, H. Ashihara and A. Crozier, Food Funct., 2014, 5, 1695–1717 RSC.
Footnote |
| † Both authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |